- Department of Internal Medicine, The 2nd Xiangya Hospital of Central South University, Changsha, China
A Corrigendum on
Iguratimod as a New Drug for Rheumatoid Arthritis: Current Landscape
by Xie S, Li S, Tian J and Li F (2020). Front. Pharmacol. 11:73. doi: 10.3389/fphar.2020.00073
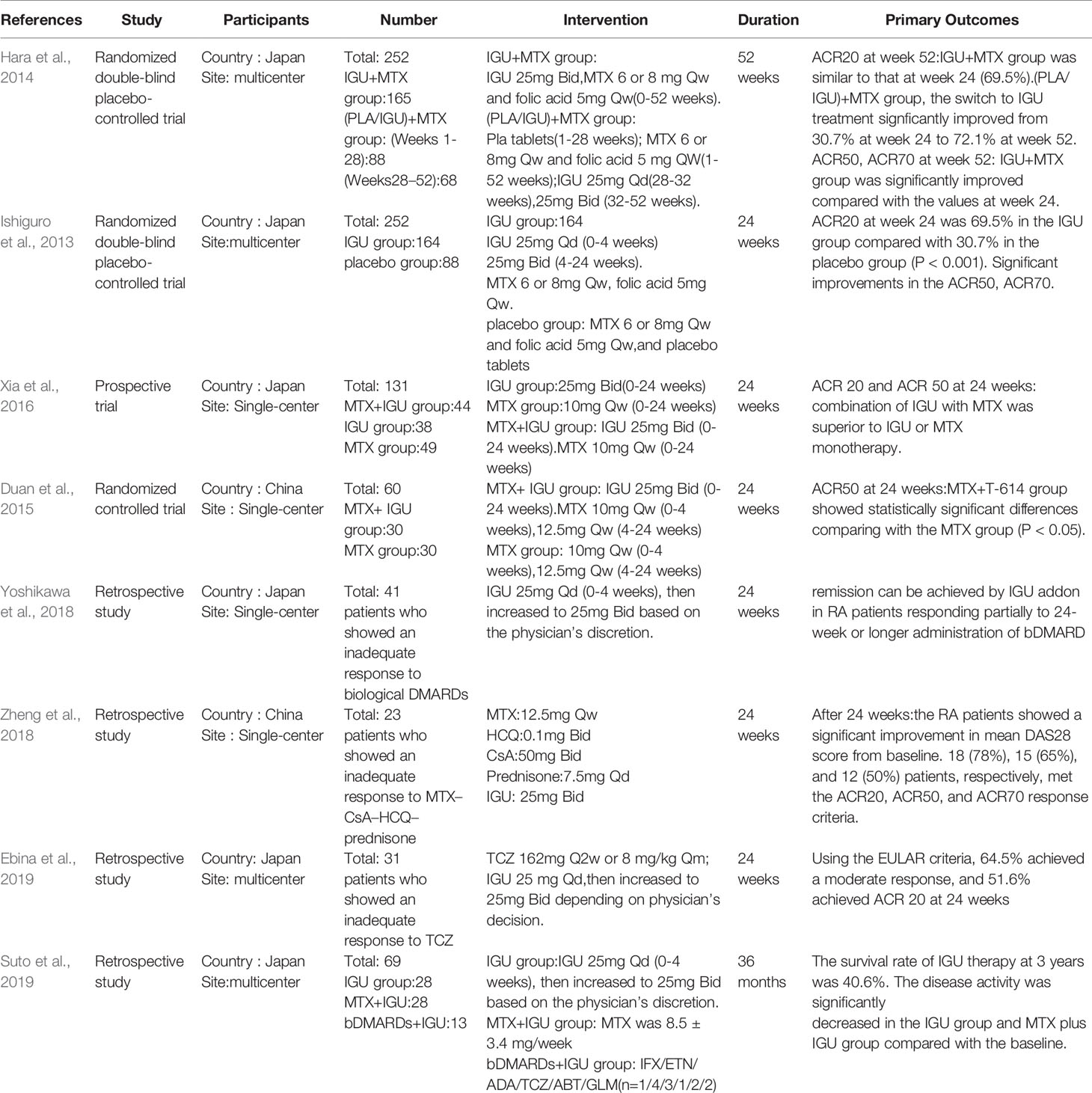
In the original article, there was a mistake in Table 2 as published. The row headers of Table 2 in the article are missing. The corrected Table 2 appears below.
The authors apologize for this error and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
References
Duan, X. W., Zhang, X. L., Mao, S. Y., Shang, J. J., Shi, X. D. (2015). Efficacy and safety evaluation of a combination of iguratimod and methotrexate therapy for active rheumatoid arthritis patients: a randomized controlled trial. Clin. Rheumatol. 34 (9), 1513–1519. doi: 10.1007/s10067-015-2999-6
Ebina, K., Miyama, A., Tsuboi, H., Kaneshiro, S., Nishikawa, M., Owaki, H., et al. (2019). The add-on effectiveness and safety of iguratimod in patients with rheumatoid arthritis who showed an inadequate response to tocilizumab. Mod. Rheumatol. 29 (4), 581–588. doi: 10.1080/14397595.2018.1486939
Hara, M., Ishiguro, N., Katayama, K., Kondo, M., Sumida, T., Mimori, T., et al. (2014). Safety and efficacy of combination therapy of iguratimod with methotrexate for patients with active rheumatoid arthritis with an inadequate response to methotrexate: an open-label extension of a randomized, double-blind, placebo-controlled trial. Mod. Rheumatol. 24 (3), 410–418. doi: 10.3109/14397595.2013.843756
Ishiguro, N., Yamamoto, K., Katayama, K., Kondo, M., Sumida, T., Mimori, T., et al. (2013). Concomitant iguratimod therapy in patients with active rheumatoid arthritis despite stable doses of methotrexate: a randomized, double-blind, placebo-controlled trial. Mod. Rheumatol. 23 (3), 430–439. doi: 10.1007/s10165-012-0724-8
Suto, T., Yonemoto, Y., Okamura, K., Sakane, H., Takeuchi, K., Tamura, Y., et al. (2019). The three-year efficacy of iguratimod in clinical daily practice in patients with rheumatoid arthritis. Mod. Rheumatol. 29 (5), 775–781. doi: 10.1080/14397595.2018.1510879
Xia, Z., Lyu, J., Hou, N., Song, L., Li, X., Liu, H. (2016). Iguratimod in combination with methotrexate in active rheumatoid arthritis: Therapeutic effects. Z. Rheumatol. 75 (8), 882–833. doi: 10.1007/s00393-015-1641-y
Yoshikawa, A., Yoshida, S., Kimura, Y., Tokai, N., Fujiki, Y., Kotani, T., et al. (2018). Add-on iguratimod as a therapeutic strategy to achieve remission in patients with rheumatoid arthritis inadequately responding to biological DMARDs: A retrospective study. Mod. Rheumatol. 28 (2), 227–234. doi: 10.1080/14397595.2017.1336865
Keywords: iguratimod, rheumatoid arthritis, NF-kappa B, randomized controlled trial, pharmacology
Citation: Xie S, Li S, Tian J and Li F (2020) Corrigendum: Iguratimod as a New Drug for Rheumatoid Arthritis: Current Landscape. Front. Pharmacol. 11:488. doi: 10.3389/fphar.2020.00488
Received: 23 March 2020; Accepted: 27 March 2020;
Published: 08 April 2020.
Edited and reviewed by: Gerard Bannenberg, GOED, United States
Copyright © 2020 Xie, Li, Tian and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Fen Li, TGlmZW4wNzMxQGNzdS5lZHUuY24=
 Sisi Xie
Sisi Xie Shu Li
Shu Li Fen Li
Fen Li