- 1Nutrition Programme, Faculty of Health Sciences, National University of Malaysia, Kuala Lumpur, Malaysia
- 2Faculty of Health and Medical Science, School of BioSciences, Taylor’s University Lakeside Campus, Selangor, Malaysia
- 3Dietetics Programme, Faculty of Health Sciences, National University of Malaysia, Kuala Lumpur, Malaysia
- 4Medical Department, Faculty of Medicine, Universiti Kebangsaan Malaysia Medical Centre, Kuala Lumpur, Malaysia
- 5Acute and Chronic Renal Failure Unit, Department of Clinical and Experimental Medicine, University of Parma, Parma, Italy
- 6Malaysian Palm Oil Council (MPOC), Petaling Jaya, Malaysia
A Corrigendum on
Composition and Functionality of Lipid Emulsions in Parenteral Nutrition: Examining Evidence in Clinical Applications
by Sadu Singh, B. K., Narayanan, S. S., Khor, B. H., Sahathevan, S., Abdul Gafor, A. H., Fiaccadori, E., Sundram, K., and Karupaiah, T. (2020). Front. Pharmacol. 11:506. doi: 10.3389/fphar.2020.00506
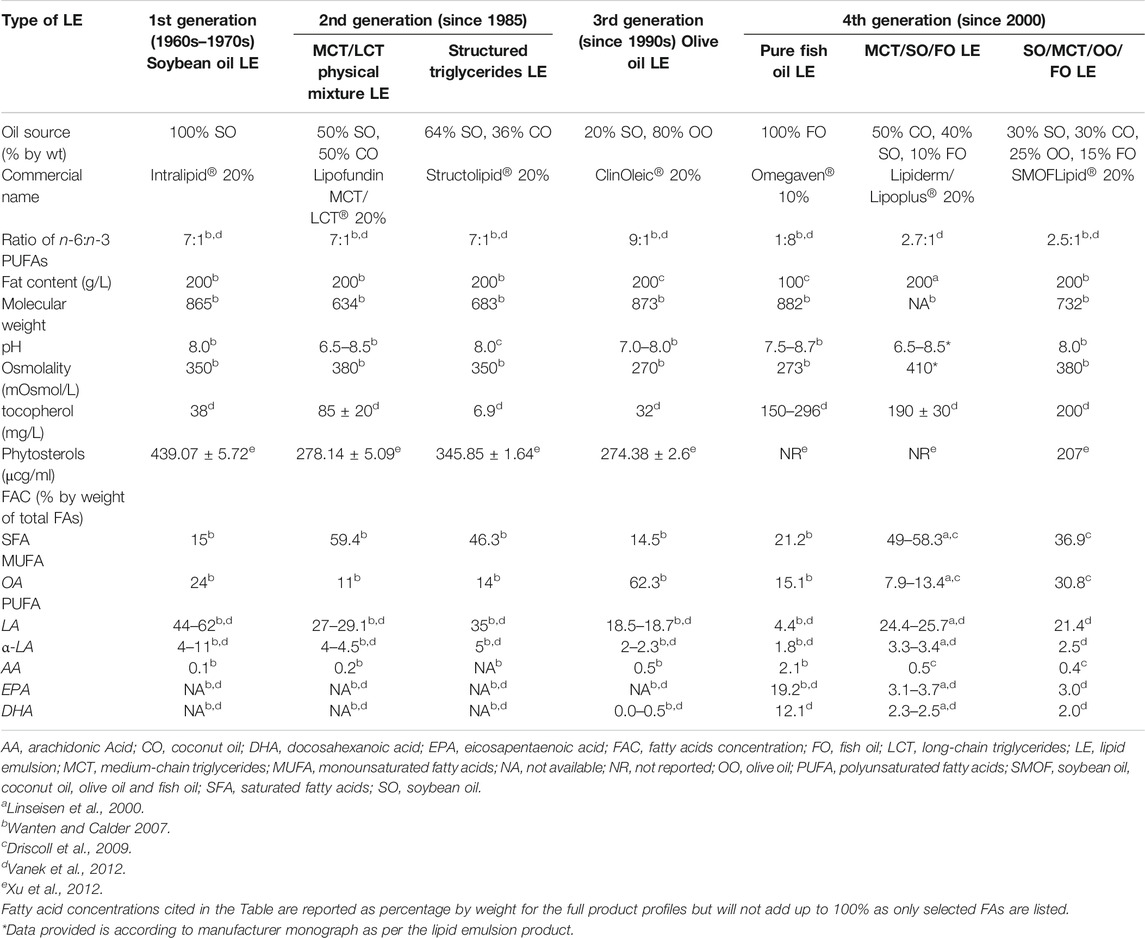
In the original article, there was a mistake in the legend for Table 2 as published. Referring to the references cited in the original article Table 2, Linseisen et al., 2000 and Driscoll et al., 2009 were missing, while Waitzberg et al., 2006 has to be removed. Therefore, Linseisen et al., 2000 and Driscoll et al., 2009 were added as the reference for the corrected Table 2. We added a sentence for the footnote of corrected Table 2: “Fatty acids concentration cited in the Table are reported as percentage by weight for the full product profiles but will not add up to 100% as only selected FAs are listed. In addition, a sentence “Data provided is according to manufacturer monograph as per the lipid emulsion product” was added as the footnote for the corrected Table 2. The correct legend appears below. The authors apologize for this error and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
References: “aLinseisen et al., 2000; bWanten and Calder 2007; cDriscoll et al., 2009; dVanek et al., 2012; eXu et al., 2012.”
In the original article, there was a mistake in Table 2 as published. The Lipiderm/Lipoplus® detailed fatty acids composition was referred from Linseisen et al., 2000 and this reference was missed out in the original Table 2. The authors cross-checked and recalculated the data on fatty acid composition presented in Table 2 based on one particular cited paper (Vanek et al., 2012), which indicated there are errors in the extrapolation of the data from the primary source (Waitzberg et al., 2006). Therefore the authors acknowledged these errors and amended the data accordingly to reflect percent composition of fatty acids by full profile (Driscoll et al., 2009). The corrected Table 2 appears below. The authors apologize for this error and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
Keywords: fatty acids, lipid emulsions, triglycerides, triacylglycerols, parenteral nutrition
Citation: Sadu Singh BK, Narayanan SS, Khor BH, Sahathevan S, Abdul Gafor AH, Fiaccadori E, Sundram K and Karupaiah T (2021) Corrigendum: Composition and Functionality of Lipid Emulsions in Parenteral Nutrition: Examining Evidence in Clinical Applications. Front. Pharmacol. 12:663960. doi: 10.3389/fphar.2021.663960
Received: 04 February 2021; Accepted: 16 February 2021;
Published: 16 April 2021.
Edited and reviewed by:
Brian Godman, University of Strathclyde, United KingdomCopyright © 2021 Sadu Singh, Narayanan, Khor, Sahathevan, Abdul Gafor, Fiaccadori, Sundram and Karupaiah. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Tilakavati Karupaiah, dGlsbHlfa2FydUB5YWhvby5jby51aw==
 Birinder Kaur Sadu Singh
Birinder Kaur Sadu Singh Sreelakshmi Sankara Narayanan
Sreelakshmi Sankara Narayanan Ban Hock Khor
Ban Hock Khor Sharmela Sahathevan
Sharmela Sahathevan Abdul Halim Abdul Gafor
Abdul Halim Abdul Gafor Enrico Fiaccadori
Enrico Fiaccadori Kalyana Sundram6
Kalyana Sundram6 Tilakavati Karupaiah
Tilakavati Karupaiah