1 Introduction
Methylenetetrahydrofolate dehydrogenase 2 (MTHFD2) is a mitochondrial one-carbon (1C) metabolism enzyme that is overexpressed in cancer cells and barely expressed in most healthy adult tissues (Nilsson et al., 2014; Jha et al., 2023). The overexpression of MTHFD2 could provide the basis for biosynthesis of pyrimidine and purine during rapid proliferation of cancer cells which is widely needed for the growth of all tumors (Kim et al., 2016; Zhao et al., 2021; Bonagas et al., 2022; Zhao et al., 2022). Inhibition of MTHFD2 leads to imbalance of NADPH and redox homeostasis, which inhibits tumorigenic proliferation and growth, and increases cancer cell death under hypoxia (Ju et al., 2019). The knockdown of MTHFD2 leads to decreased expression of cell cycle genes suggesting interference with cell cycle progression (Yu et al., 2020). Because of the low expression of MTHFD2 in most adult tissues, targeting MTHFD2 is unlikely to produce significant side effects and MTHFD2 could be as a novel target for cancer therapy (Nishimura et al., 2019; Cuthbertson et al., 2021; Yang et al., 2021).
Recent research found that MTHFD2 was consistently overexpressed in many diseases, including ulcerative colitis, Celiac’s disease, systemic lupus erythematosus (SLE), psoriatic arthritis, Sjogren’s syndrome, multiple sclerosis (MS) and so on (Sugiura et al., 2022). Inhibition of MTHFD2 promotes regulatory CD4 T cell (Treg) activity, which suppresses the immune response. Does MTHFD2 play a new role from anticancer targets to inti-inflammatory disease?
2 A new role on anti-inflammatory disease and proposed mechanisms
In fact, what we are more interested in is that MTHFD2 deficiency can reduce disease degree in various inflammatory condition models. T-cell dependent Delayed Type Hypersensitivity (DTH) mouse models trials showed that MTHFD2 inhibitors did not increase inflammatory symptoms in mice, and increase animal weight, suggesting that the inhibitor has a protective effect on inflammation extending to B cell function (Sugiura et al., 2022). MS is an inflammatory demyelinating disease originating in the central nervous system. Compared to control group, Experimental Autoimmune Encephalomyelitis (EAE) model using with MTHFD2 inhibitors (MTHFD2i) resulted in significantly lower disease degree and cumulative clinical score. The infiltration of CD4st, CD4+ and CD8+ cells in the spinal cord of mice was significantly reduced after MTHFD2i treatment (Sugiura et al., 2022). In two other different inflammatory models-inflammatory bowel disease (IBD) and allergic airway disease, mice receptor of CD4ΔMthfd2 T cells continued gaining body weight, the number and frequency of CD4ΔMthfd2 T cells in spleen and mesenteric lymph nodes (MLNs) were significantly reduced. Meanwhile, the neutrophil richness in the bronchioalveolar lavage fluid (BALF) of CD4ΔMthfd2 mice affected by Alternaria-induced allergic airway disease showed a decreasing tendency (Sugiura et al., 2022). The sensitivity of T-cell to MTHFD2i might provide an efficacious strategy of immunotherapy for CD4+ T-cell-driven inflammation, and produce fewer adverse reactions than presently usable therapeutics. It should be worth studying whether T cell nucleus carries MTHFD2 and whether MTHFD2 is a therapeutic target for inflammatory disease.
CD4+ T cells are the key mediators and adaptive immunity which play a crucial role in host defense against pathogens (Candia and Matarese, 2022). CD4+ T cell subpopulations need MTHFD2 to varying degrees for activation, proliferation, survival, and cytokine production (Sugiura et al., 2022). Sugiura et al. (2022) have found that MTHFD2 in patients with inflammatory disease continues to upregulate combined with cell CRISPR-based screening and genetic test. The research showed that MTHFD2 may function as a metabolic checking point for the Th17/Treg cell axis and highlight its potential as a target for anti-inflammatory immunotherapeutic treatment. Meanwhile, MTHFD2i raised the basal and maximal oxygen consumption rate (OCR) of Th17 cells and decreased the expression of interferon-gamma (IFN-g) and interleukin (IL)-17 in Th1 and Th17 cells, which appears to alter the counterbalance between the pathogenic and anti-inflammatory state.
MTHFD2 has been shown to regulate de novo purine synthesis and signal transduction in activated T cells, promoting proliferation and the production of inflammatory cytokine (Ducker et al., 2016). MTHFD2 has been reported to transport to the nucleus and is presumed regulate gene expression (Gustafsson Sheppard et al., 2015). The lack of MTHFD2 could lead to the accumulation of intermediates in the purine synthesis pathway, which activates AMP-activated protein kinase to inhibit the mechanistic target of rapamycin (mTORC)1 (Su et al., 2019). The mTORC1 pathway plays a crucial role in promoting synthetic metabolism, driving a mass of the transcription factor ATF4 and inducing the expression of MTHFD2 (Ben-Sahra et al., 2016). Inhibition of mTORC1 signaling transduction might lead to changes in the metabolic process from glycolysis to mitochondrial respiration, and alter T cell receptor cycle metabolites (Shang et al., 2021).
3 Novel MTHFD2 inhibitors
One possible mechanism is that MTHFD2i damages T cell expansion through inadequate nucleotide production. Scientists have been working on the design and development of MTHFD2i as anticancer drugs (Table 1). Tricyclic coumarins and xanthine compounds are the only selective inhibitors of MTHFD2 reported to date (Jha et al., 2023). Comprehensive searches of English databases, including PubMed, Scopus, and Web of Science, and the time of index was from inception to 30 April 2023 for each database. Full-text searches were performed using “MTHFD2 inhibitors” in all fields (Figure 1). The dual MTHFD1/2 inhibitor LY345899 synthesized in 2017 has been demonstrated efficacy in improving disease conditions in the EAE model (Gustafsson et al., 2017; Ju et al., 2019). A simplification of the tricyclic core of LY345899 shows that TH9028, TH9619 and TH7299 are actually more active against MTHFD1 and MTHFD2L (Bonagas et al., 2022; Scaletti et al., 2022; Green et al., 2023). A novel isozyme-selective MTHFD2 inhibitor DS44960156 might provide further optimization options due to its >18-fold selectivity for MTHFD2 over MTHFD1, with a smaller molecular weight and favorable ligand efficiency (Kawai et al., 2019a). Subsequently, the same team developed an effective, selective, and oral MTHFD2i (DS18561882) which has favorable oral pharmacokinetic characteristics with the strongest cell activity and tumor growth inhibition (Kawai et al., 2019b; Lee et al., 2021). Most importantly, DS18561882 has been shown to reduce disease degree in variety of inflammatory disease models in vivo (Sugiura et al., 2022), which leads us to believe that MTHFD2 may be an anti-inflammatory and autoimmune target in vivo in the future.
TABLE 1
| Name | Target | Structure | Pathology |
|---|---|---|---|
| LY345899 | Dual MTHFD1/2 inhibitor |

|
LY345899 treatment statistically significantly suppresses tumor growth and decreases the tumor weight in CRC patient-derived xenograft models |
| TH7299 | Dual MTHFD1/2 inhibitor |

|
(−) |
| TH9028 | Dual MTHFD1/2 inhibitor |

|
TH9028 and TH9619 showed overall strong antiproliferative efficacy in acute myeloid leukemia (AML) cells and T-ALL Jurkat cells comparable to standard-of-care compounds, with reduced effect on lymphoblastoid cell line (LCL) viability |
| TH9619 | Dual MTHFD1/2 inhibitor |

|
|
| DS44960156 | MTHFD2 inhibitor |

|
DS44960156has > 18-fold selectivity for MTHFD2 over MTHFD1, with a low molecular weight and a good ligand efficiency |
| DS18561882 | MTHFD2 inhibitor |

|
DS18561882, combined with enzalutamide can signifcantly inhibit CRPC cell proliferation in vitro and tumor growth in vivo. DS18561882 has also been shown to reduce disease degree in variety of inflammatory disease models in vivo |
Novel MTHFD2 inhibitors.
FIGURE 1
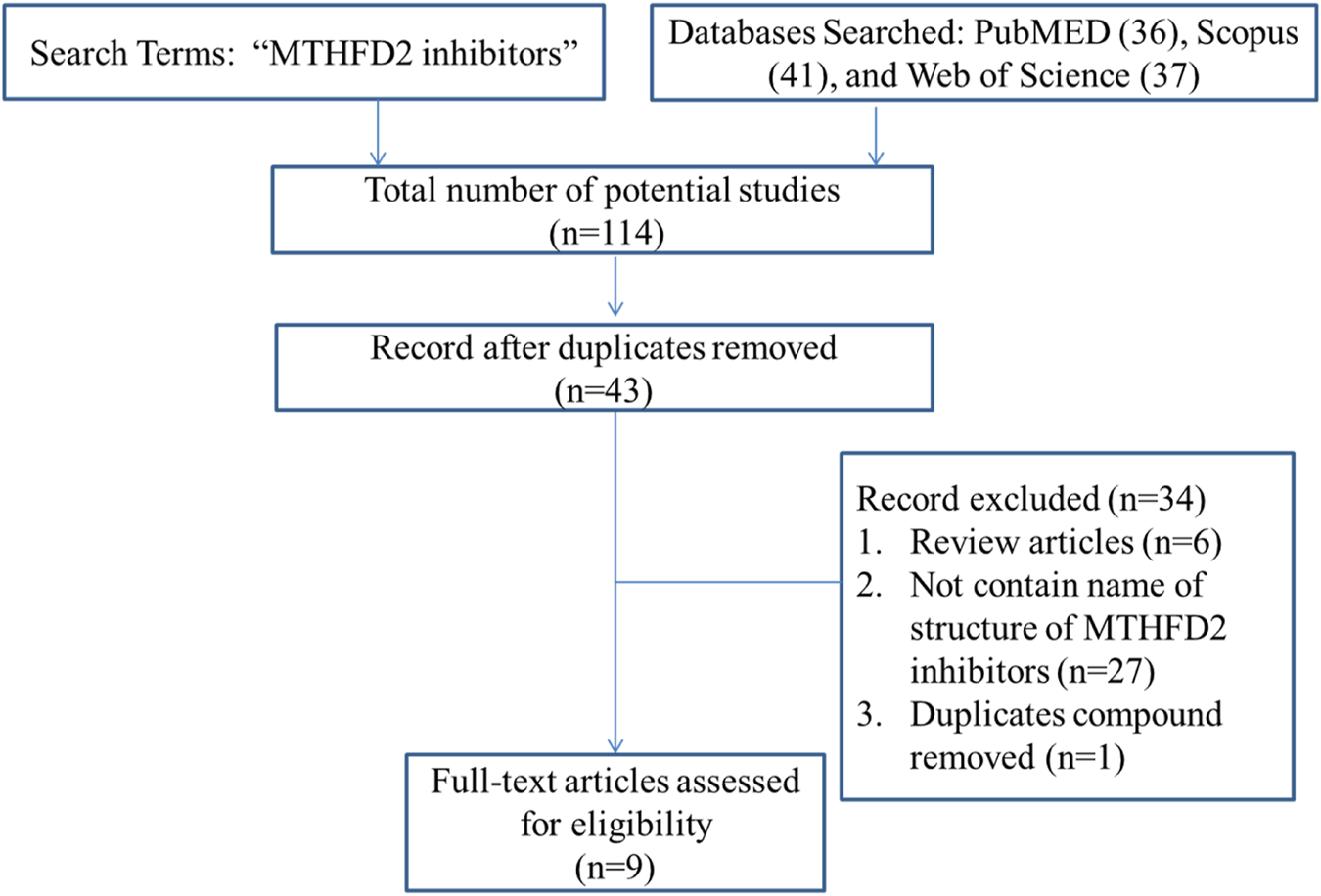
Study flow diagram of literature search.
4 Conclusion
MTHFD2 is a mitochondrial one-carbon metabolism enzyme highly expressed in several human tumors, and targeting MTHFD2 has been used as the target of tumor therapy. Recent research suggests that MTHFD2 inhibitors appear to reduce inflammatory disease severity and alter the counterbalance between the pathogenic and anti-inflammatory state, which may serve as an anti-inflammatory and autoimmune target in vivo in the future. The research of anti-inflammatory drugs is expected to be promoted and developed.
Statements
Author contributions
HT: Conceptualization, Writing–original draft. NH: Conceptualization, Writing–review and editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1
Ben-Sahra I. Hoxhaj G. Ricoult S. J. H. Asara J. M. Manning B. D. (2016). mTORC1 induces purine synthesis through control of the mitochondrial tetrahydrofolate cycle. Science351, 728–733. 10.1126/science.aad0489
2
Bonagas N. Gustafsson N. M. S. Henriksson M. Marttila P. Gustafsson R. Wiita E. et al (2022). Pharmacological targeting of MTHFD2 suppresses acute myeloid leukemia by inducing thymidine depletion and replication stress. Nat. Cancer3, 156–172. 10.1038/s43018-022-00331-y
3
Candia P. D. Matarese G. (2022). The folate way to T cell fate. Immunity55, 1–3. 10.1016/j.immuni.2021.12.009
4
Cuthbertson C. R. Arabzada Z. Bankhead A. Kyani A. Neamati N. (2021). A review of small-molecule inhibitors of one-carbon enzymes: SHMT2 and MTHFD2 in the spotlight. ACS Pharmacol. Transl. Sci.4, 624–646. 10.1021/acsptsci.0c00223
5
Ducker G. S. Chen L. Morscher R. J. Ghergurovich J. M. Esposito M. Teng X. et al (2016). Reversal of cytosolic one-carbon flux compensates for loss of the mitochondrial folate pathway. Cell Metab.23, 1140–1153. 10.1016/j.cmet.2016.04.016
6
Green A. C. Marttila P. Kiweler N. Chalkiadaki C. Wiita E. Cookson V. et al (2023). Formate overflow drives toxic folate trapping in MTHFD1 inhibited cancer cells. Nat. Metab.5, 642–659. 10.1038/s42255-023-00771-5
7
Gustafsson R. Jemth A.-S. Gustafsson N. M. S. Färnegårdh K. Loseva O. Wiita E. et al (2017). Crystal structure of the emerging cancer target MTHFD2 in complex with a substrate-based inhibitor. Cancer Res.77, 937–948. 10.1158/0008-5472.CAN-16-1476
8
Gustafsson Sheppard N. Jarl L. Mahadessian D. Strittmatter L. Schmidt A. Madhusudan N. et al (2015). The folate-coupled enzyme MTHFD2 is a nuclear protein and promotes cell proliferation. Sci. Rep.5, 15029. 10.1038/srep15029
9
Jha V. Holmelin F. L. Eriksson L. A. (2023). Binding analysis and structure-based design of tricyclic coumarin-derived MTHFD2 inhibitors as anticancer agents: insights from computational modeling. ACS Omega8, 14440–14458. 10.1021/acsomega.2c08025
10
Ju H.-Q. Lu Y.-X. Chen D.-L. Zuo Z.-X. Liu Z.-X. Wu Q.-N. et al (2019). Modulation of redox homeostasis by inhibition of MTHFD2 in colorectal cancer: mechanisms and therapeutic implications. J. Natl. Cancer Inst.111, 584–596. 10.1093/jnci/djy160
11
Kawai J. Ota M. Ohki H. Toki T. Suzuki M. Shimada T. et al (2019a). Structure-based design and synthesis of an isozyme-selective MTHFD2 inhibitor with a tricyclic coumarin scaffold. ACS Med. Chem. Lett.10, 893–898. 10.1021/acsmedchemlett.9b00069
12
Kawai J. Toki T. Ota M. Inoue H. Takata Y. Asahi T. et al (2019b). Discovery of a potent, selective, and orally available MTHFD2 inhibitor (DS18561882) with in vivo antitumor activity. J. Med. Chem.62, 10204–10220. 10.1021/acs.jmedchem.9b01113
13
Kim J. Yang G. Kim Y. Kim J. Ha J. (2016). AMPK activators: mechanisms of action and physiological activities. Exp. Mol. Med.48, e224. 10.1038/emm.2016.16
14
Lee J. Chen X. Wang Y. Nishimura T. Li M. Ishikawa S. et al (2021). A novel oral inhibitor for one-carbon metabolism and checkpoint kinase 1 inhibitor as a rational combination treatment for breast cancer. Biochem. Biophys. Res. Commun.584, 7–14. 10.1016/j.bbrc.2021.11.001
15
Nilsson R. Jain M. Madhusudhan N. Sheppard N. G. Strittmatter L. Kampf C. et al (2014). Metabolic enzyme expression highlights a key role for MTHFD2 and the mitochondrial folate pathway in cancer. Nat. Commun.5, 3128. 10.1038/ncomms4128
16
Nishimura T. Nakata A. Chen X. Nishi K. Meguro-Horike M. Sasaki S. et al (2019). Cancer stem-like properties and gefitinib resistance are dependent on purine synthetic metabolism mediated by the mitochondrial enzyme MTHFD2. Oncogene38, 2464–2481. 10.1038/s41388-018-0589-1
17
Scaletti E. R. Gustafsson Westergren R. Andersson Y. Wiita E. Henriksson M. Homan E. J. et al (2022). The first structure of human MTHFD2L and its implications for the development of isoform-selective inhibitors. ChemMedChem17, e202200274. 10.1002/cmdc.202200274
18
Shang M. Yang H. Yang R. Chen T. Fu Y. Li Y. et al (2021). The folate cycle enzyme MTHFD2 induces cancer immune evasion through PD-L1 up-regulation. Nat. Commun.12, 1940. 10.1038/s41467-021-22173-5
19
Su C.-C. Hsieh K.-L. Liu P.-L. Yeh H.-C. Huang S.-P. Fang S.-H. et al (2019). AICAR induces apoptosis and inhibits migration and invasion in prostate cancer cells through an AMPK/mTOR-Dependent pathway. Int. J. Mol. Sci.20, 1647. 10.3390/ijms20071647
20
Sugiura A. Andrejeva G. Voss K. Heintzman D. R. Xu X. Madden M. Z. et al (2022). MTHFD2 is a metabolic checkpoint controlling effector and regulatory T cell fate and function. Immunity55, 65–81.e9. 10.1016/j.immuni.2021.10.011
21
Yang C. Zhang J. Liao M. Yang Y. Wang Y. Yuan Y. et al (2021). Folate-mediated one-carbon metabolism: a targeting strategy in cancer therapy. Drug Discov. Today26, 817–825. 10.1016/j.drudis.2020.12.006
22
Yu C. Yang L. Cai M. Zhou F. Xiao S. Li Y. et al (2020). Down‐regulation of MTHFD2 inhibits NSCLC progression by suppressing cycle‐related genes. J. Cell. Mol. Med.24, 1568–1577. 10.1111/jcmm.14844
23
Zhao L. N. Björklund M. Caldez M. J. Zheng J. Kaldis P. (2021). Therapeutic targeting of the mitochondrial one-carbon pathway: perspectives, pitfalls, and potential. Oncogene40, 2339–2354. 10.1038/s41388-021-01695-8
24
Zhao R. Feng T. Gao L. Sun F. Zhou Q. Wang X. et al (2022). PPFIA4 promotes castration-resistant prostate cancer by enhancing mitochondrial metabolism through MTHFD2. J. Exp. Clin. Cancer Res. CR41, 125. 10.1186/s13046-022-02331-3
Summary
Keywords
methylenetetrahydrofolate dehydrogenase 2, T cells, inflammatory disease, therapeutic target, one-carbon metabolism enzyme
Citation
Tang H and Hou N (2023) Whether MTHFD2 plays a new role: from anticancer targets to anti-inflammatory disease. Front. Pharmacol. 14:1257107. doi: 10.3389/fphar.2023.1257107
Received
12 July 2023
Accepted
16 October 2023
Published
23 October 2023
Volume
14 - 2023
Edited by
Yan Huang, Anhui Medical University, China
Reviewed by
Giorgia Colombo, University of Eastern Piedmont, Italy
Gulam M. Rather, Rutgers, The State University of New Jersey, United States
Updates
Copyright
© 2023 Tang and Hou.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ning Hou, houning196@163.com
ORCID:Hui Tang, orcid.org/0000-0002-7274-1487; Ning Hou, orcid.org/0000-0001-5494-8460
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.