Abstract
Bai Hua Qian Hu (Qianhu; Peucedanum praeruptorum Dunn) is a classical medicinal plant traditionally prescribed for respiratory ailments, including cough, pulmonary hypertension, and asthma. In this review, we summarize the research progress of the toxicology, pharmacokinetics, pharmacology, phytochemistry, botany, quality control, and traditional uses of P. praeruptorum in order to support future investigations into the scientific and therapeutic promise of this important medicinal plant. Information pertaining to P. praeruptorum was collected from scientific databases (ScienceDirect, Springer, SciFinder, PubMed, Baidu Scholar, Google Scholar, Web of Science), as well as toxicology papers from local conferences, M. Sc. and Ph.D. theses and dissertations, local magazines, classic texts on Chinese botanical drugs, and peer-reviewed journals. The Plant List (www.theplantlist.org) was utilized to verify the taxonomy of P. praeruptorum. P. praeruptorum was found to contain more than 119 distinct phytochemicals, including simple coumarins, pyranocoumarins, furanocoumarins, flavonoids, ketones, organic acids, and sterols, among others (e.g., praeruptorins A and B). Both crude plant extracts and purified metabolites of P. praeruptorum have been reported as treatments for hypertension, osteoporosis, Huntington’s disease, and cancer. In addition, extracts of P. praeruptorum are reported to exhibit diverse pharmacological activities, including osteogenic, anti-osteoclastogenic, antidepressant, neuroprotective, antitumor, and anti-inflammatory effects. Research into the pharmacology and phytochemistry of P. praeruptorum partially support both traditional uses and extraction methods. However, further research is required to elucidate the relationships between these metabolites, their molecular mechanisms, their structure-function roles, and their antagonistic and synergistic effects.
1 Introduction
Peucedanum L. (Umbelliferae) consists of 120 species of herbaceous perennial plants which are distributed widely across the globe (Editorial Committee of Flora of China, 1992). One member of the genus, Peucedanum praeruptorum Dunn, is cultivated in montane habitats at an altitude between 250 and 2000 m. In traditional Chinese medicine (TCM), the root tissues of P. praeruptorum (Qianhu) have been utilized for hundreds of years to address diverse respiratory ailments, including cough, asthma, and pulmonary hypertension (Zhou et al., 2013). The roots of P. praeruptorum demonstrate diverse pharmacological activities, including anti-inflammatory, neuroprotective, antitumor, anti-osteoclastogenic, antidepressant, and osteogenic effects (Song et al., 2022). In addition, P. praeruptorum has been found to contain an array of useful phytochemicals, including simple coumarins, pyranocoumarins, furanocoumarins, flavonoids, ketones, sterols, and organic acids, and others (Song et al., 2022).
However, to date, there has been no comprehensive and systematic evaluation of the bioactivities, pharmacology, structures, functions, and toxicities of these phytochemicals, or of P. praeruptorum crude extracts. Moreover, the traditional uses of P. praeruptorum and their pharmacological evidence have not been critically evaluated. Here, we systematically summarized the toxicology, molecular mechanisms, pharmacology, phytochemistry, botany, quality control, and traditional uses of P. praeruptorum to validate the medicinal use of this species. To further clarify the material basis of P. praeruptorum’s medicinal effect, identifying the structures of metabolites will provide a certain theoretical basis for the further development and utilization of Qianhu. The information presented here can aid the planning of clinical trials and the development of novel medicines containing P. praeruptorum or its active constituents.
2 Materials and methods
Information pertaining to P. praeruptorum was sourced from scientific databases (ScienceDirect, Web of Science, Springer, Google Scholar, SciFinder, PubMed, Baidu Scholar), as well as toxicology papers from local conferences, M. Sc. and Ph.D. theses and dissertations, local magazines, classic texts on Chinese botanical drugs, and peer-reviewed journals. We utilized the following, as well as related, keywords to perform the literature review: P. praeruptorum Dunn, secondary metabolites, toxicology, safety, ethnobotanical survey, quality control, pharmacology, medicinal uses, phytochemistry, and biological activity. The Plant List (www.theplantlist.org) was utilized to verify the taxonomy of P. praeruptorum and verify subspecies and cultivars. The chemical structures were drawn using ChemDraw.
3 Botany
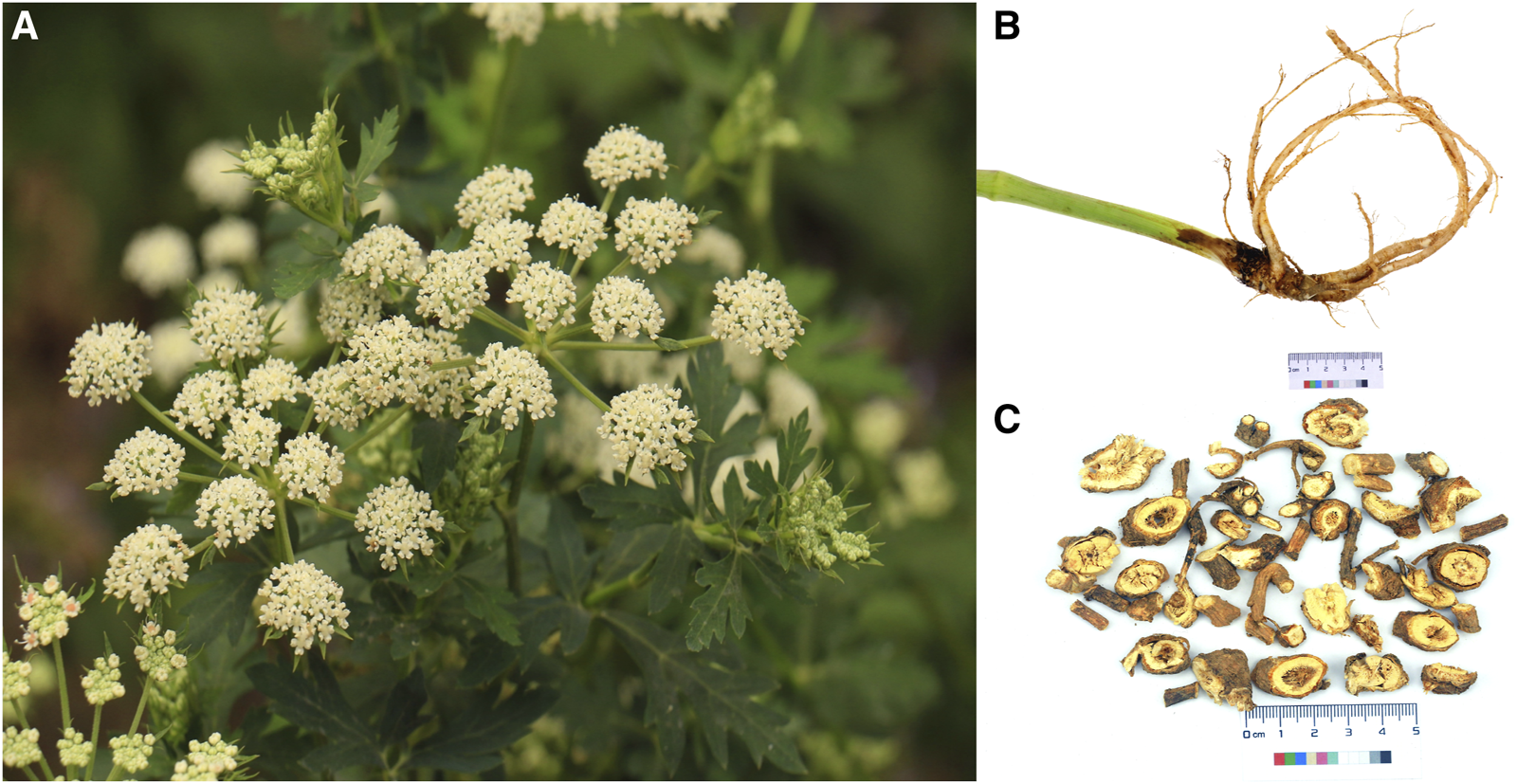
P. praeruptorum Dunn (Figure 1) is an herbaceous perennial in the Umbelliferae family. P. praeruptorum Dunn is the only accepted name for the species (www.theplantlist.org), although it has two other synonyms: P. praeruptorum var. grande K.T. Fu and P. praeruptorum subsp. hirsutiusculum Ma. P. praeruptorum is found in the wild in south China, including in Zhejiang, Anhui, Jiangxi, Hubei, Hunan, Guizhou, Sichuan, and Yunnan provinces (Figure 2). The traditional production areas are northwest Zhejiang, southeast Anhui, and northeast Jiangxi, where the plant is called “Zhe Qianhu,” “Ning Qianhu,” and “Xin Qianhu,” respectively (Zhou et al., 2021).
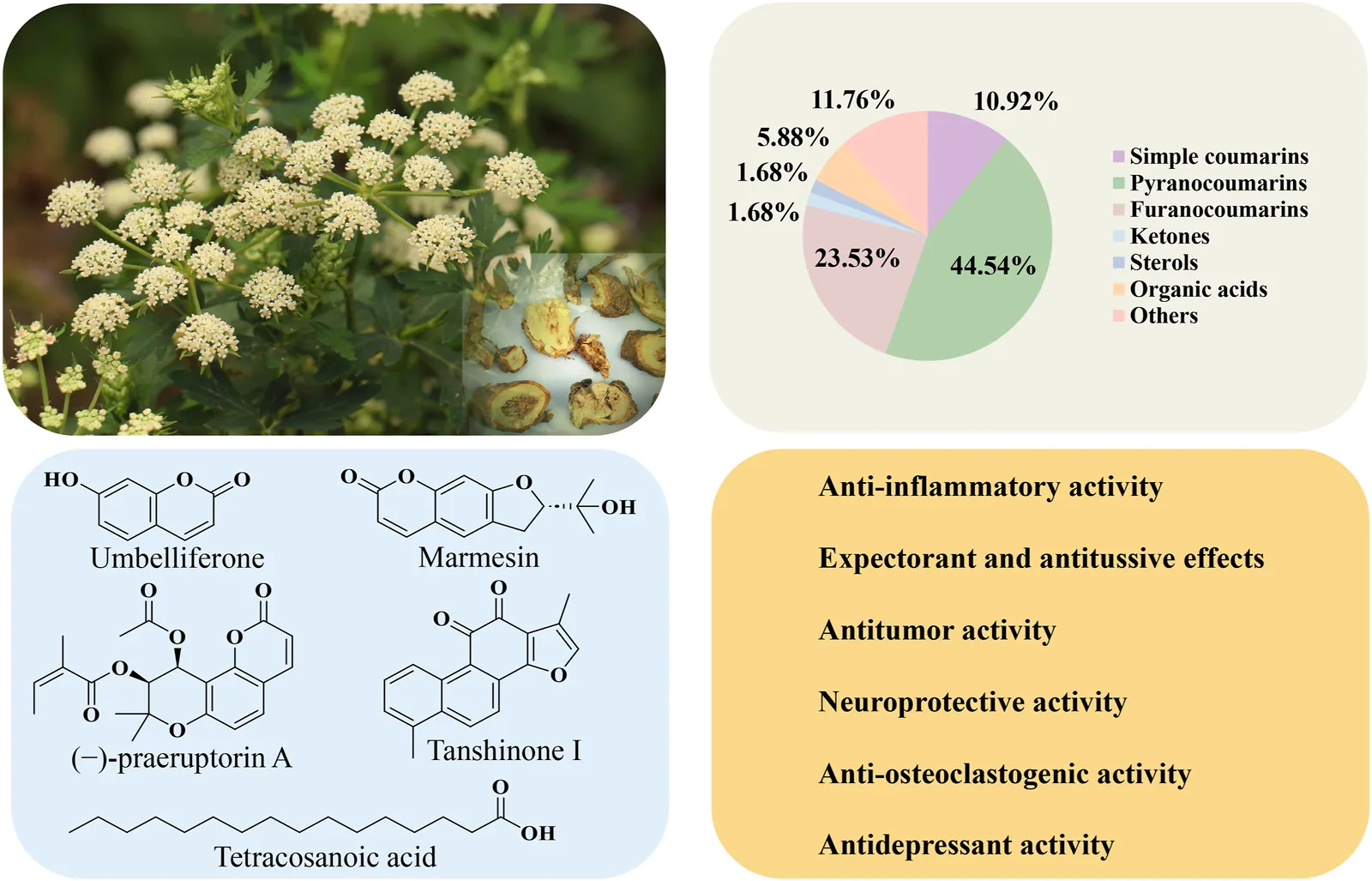
FIGURE 1
The aerial tissues (A), medicinal root tissues (B), and commercial presentation (C) of Peucedanum praeruptorum Dunn.
FIGURE 2
The natural distribution of Peucedanum praeruptorum Dunn across southern China.
According to the Flora of China (Editorial Committee of Flora of China, 1992), P. praeruptorum grows along forest edges, near roadsides, and in semi-open grassy areas within montane habitats at an altitude of 250–2000 m (Liu et al., 2021). P. praeruptorum can reach a height of 60–100 cm. The plant has a cylindrical stem, with a glabrous lower part and piliferous branches on the upper part. The medulla is solid. Leaves are wide ovate or triangular ovate. The compound umbel is terminal or lateral, and 3.5–9 cm in diameter. The fruits are oval, 4 mm in length and 3 mm in width. The back of the fruit is flat. The rhizomes are strong, brown, and 1–1.5 cm in diameter. The roots are conical and branched, with thin root ends. For medicinal use, roots are collected from winter to spring. Abluent, fresh, thin slices are dried prior to medicinal use.
4 Traditional uses
The List of Mingyi Bielu (《名医别录》), which dates to the Wei-Jin and South-North Dynasties (A.D. 220-450), was the first to record the roots of P. praeruptorum as TCM. Many ancient texts, such as the Rihuazi Bencao (《日华子本草》) (Five Dynasties, A.D. 908–923), the Compendium of Materia Medica (《本草纲目》) (Ming Dynasty, A.D. 1578), and the Illustrated Classics of Materia Medica (《本草图经》) (Song Dynasty, A.D. 1061) also record that P. praeruptorum was widely used to treat colds, headaches, coughs, asthma, and chest congestion (He et al., 2007). In the Chinese Pharmacopoeia 2000 (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2000), Qianhu is defined as the roots of either P. decursivum (Miq.) Maxim (Zihuaqianhu) or P. praeruptorum Dunn. However, P. decursivum is not traditionally used as a source of Qianhu, and thus, was removed from the Chinese Pharmacopoeia 2005 (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2005). More recently, the Chinese Pharmacopoeia 2010, 2015, and 2020 (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2010; Chinese Pharmacopoeia Committee of People’s Repulic of China, 2015; Chinese Pharmacopoeia Committee of People’s Repulic of China, 2020) define Qianhu as the roots of P. praeruptorum exclusively, while Zihuaqianhu is defined as the roots of P. decursivum.
P. praeruptorum has many folk names, including yimacai, luoguicai, shuiqianhu, shuifangfeng, shanyuansui, guanqianhu, and shanduhuo. In TCM, Qianhu is used to treat chronic respiratory failure, acute bronchitis, and iridocyclitis after cataract surgery (Wang, 2016). The roots of P. praeruptorum have been utilized in a variety of traditional preparations, and are often used in combination with Mentha haplocalyx Briq., Arctium lappa L., and Platycodon grandiflorum (Jacq.) A. DC. to treat external wind-heat, body heat, headache, cough, and phlegm. In addition, P. praeruptorum is used in combination with Citrus reticulata Blanco, Pinellia ternata (Thunb.) Breit., and Prunus armeniaca L. var. ansu Maxim. to treat cough, chest congestion, vomiting, and nausea. The roots of P. praeruptorum have been used in more than 189 TCM preparations and 525 classical prescriptions (https://db.yaozh.com, accessed 7th December, 2023). Examples of TCM prescriptions containing P. praeruptorum are listed in Table 1. “Bai He Qian Hu Tang,” “Da Qian Hu Tang,” “Fu Ling Qian Hu Tang,” “Jiu Wei Qian Hu Tang,” and “Xing Su San” are Chinese classical prescriptions recorded in many ancient books. The botanical drugs “Tong Xuan Li Fei Ke Li” and “Tong Xuan Li Fei Pian” (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2020), which are accredited by the National Medical Products Administration, are produced and marketed in China to treat cough. However, further research is required to clarify any potential synergisms or interactions between the bioactive phytochemicals in P. praeruptorum and those of other medicinal plants, as well as to elucidate their mechanisms of action. According to the Chinese Pharmacopoeia 2020 (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2020), although Qianhu can disperse wind-heat, reduce cough and phlegm, and dissipate adverse Qi, comprehensive studies of its constitutive bioactive monomers should be conducted.
TABLE 1
| Prescription | Ingredients | Role played by qianhu in formulation | Clinical and traditional uses | References |
|---|---|---|---|---|
| Jin Fei Cao San | Inula japonica Thunb., Ephedra sinica Stapf, Peucedanum praeruptorum Dunn, Schizonepeta tenuisfolia Briq., Glycyrrhiza uralensis Fisch., Pinellia ternate (Thunb.) Breit., Paeonia lactiflora Pall | Leading role | Phlegmatic heat cough, wind-heat cough | Bojifang (《博济方》) |
| Bai He Qian Hu Tang | Lilium lancifolium Thunb., Peucedanum praeruptorum Dunn, Ephedra sinica Stapf, Pueraria lobata (Willd.) Ohwi, Ophiopogon japonicus (L.f.) KerGawl., CaSO4·2H2O | Leading role | Wind-heat cough | General Medical Collection of Royal Benevolence (《圣济总录》) |
| Da Qian Hu Tang | Peucedanum praeruptorum Dunn, Zingiber officinale Rosc., Pinellia ternata (Thunb.) Breit., Ephedra sinica Stapf, Scutellaria baicalensis Georgi, Paeonia lactiflora Pall., Ziziphus jujuba Mill., Citrus aurantium L | Leading role | Wind-heat cough | Waitai Miyao (《外台秘要》), Gujin Luyan (《古今录验》) |
| Fu Ling Qian Hu Tang | Poria cocos (Schw.) Wolf, Peucedanum praeruptorum Dunn, Chrysanthemum morifolium Ramat., Atractylodes macrocephala Koidz., Aconitum carmichaelii Debx., Asarum heterotropoides Fr. Schmidt var. mandshuricum (Maxim.) Kitag, Ephedra sinica Stapf | Leading role | Phlegmatic heat cough, wind-heat cough | General Medical Collection of Royal Benevolence (《圣济总录》) |
| Jia Wei Qian Hu Tang | Glycyrrhiza uralensis Fisch., Platycodon grandiflorum (Jacq.) A.DC., Morus alba L., Eriobotrya japonica (Thunb.) Lindl., Tussilago farfara L., Prunus armeniaca L.var.ansu Maxim., Lonicera japonica Thunb., Scutellaria baicalensis Georgi, Ophiopogon japonicus (L.f) Ker-Gawl., Anemarrhena asphodeloides Bge., Peucedanum praeruptorum Dunn | Leading role | Phlegmatic heat cough, wind-heat cough | Zhengqiaofang (《郑侨方》) |
| Jiu Wei Qian Hu Tang | Crataegus pinnatifida Bge. var. major N. E. Br., Schizonepeta tenuifolia Briq., Carthamus tinctorius L., Citrus aurantium L., Prunus armeniaca L.var.ansu Maxim., Platycodon grandiflorum (Jacq.) A.DC., Angelica sinensis (Oliv.) Diels, Saposhnikovia divaricata (Turcz.) Schischk., Peucedanum praeruptorum Dunn | Leading role | Phlegmatic heat cough | Zhizhen Quanshu (《治疹全书》) |
| Jie Geng Qian Hu Tang | Platycodon grandiflorum (Jacq.) A.DC., Perilla frutescens (L.) Britt., Prunus armeniaca L.var.ansu Maxim., Peucedanum praeruptorum Dunn, Paeonia lactiflora Pall., Morus alba L., Glycyrrhiza uralensis Fisch., Citrus reticulata Blanco, Bambusa tuldoides Munro | Leading role | Phlegmatic heat cough | Bi Hua Yi Jing (《笔花医镜》) |
| Pi Pa Ye Qian Hu San | Citrus aurantium L., Trionyx sinensis Wiegmann, Paeonia suffruticosa Andr., Glycyrrhiza uralensis Fisch., Paeonia lactiflora Pall., Angelica sinensis (Oliv.) Diels, Zingiber officinale Rosc., Magnolia officinalis Rehd.et Wils., Saposhnikovia divaricata (Turcz.) Schischk., Atractylodes macrocephala Koidz., Schisandra chinensis (Turcz.) Baill., Peucedanum praeruptorum Dunn, Zingiber officinale Rosc., Eriobotrya japonica (Thunb.) Lindl., Poria cocos (Schw.) Wolf, Angelica dahurica (Fisch.ex Hoffm.) Benth.et Hook.f., Pinellia ternate (Thunb.) Breit., Anemarrhena asphodeloides Bge. Pogostemon cablin (Blanco) Benth., Panax ginseng C. A. Mey., Alisma orientale (Sam.) Juzep., Platycodon grandiflorum (Jacq.) A.DC., Aucklandia lappa Decne., Areca catechu L., Akebia quinata (Thunb.) Decne., Scirpus yagara Ohwi, Terminalia chebula Retz | Leading role | Phlegmatic heat cough, wind-heat cough | Chuanjia Mibao (《传家秘宝》) |
| Qian Hu San | Peucedanum praeruptorum Dunn, Scutellaria baicalensis Georgi, Gardenia jasminoides Ellis, Saposhnikovia divaricata (Turcz.) Schischk., Chrysanthemum morifolium Ramat., Adenophora stricta Miq., Glycyrrhiza uralensis Fisch., Saiga tatarica Linnaeus, Ophiopogon japonicus (L.f.) KerGawl., Citrus aurantium L., CaSO4·2H2O | Leading role | Phlegmatic heat cough, wind-heat cough | Qixiao Liangfang (《奇效良方》) |
| Zhi Qiao Qian Hu Tang | Platycodon grandiflorum (Jacq.) A.DC., Glycyrrhiza uralensis Fisch., Peucedanum praeruptorum Dunn, Saposhnikovia divaricata (Turcz.) Schischk., Citrus aurantium L., Poria cocos (Schw.) Wolf, Perilla frutescens (L.) Britt | Leading role | Phlegmatic heat cough, wind-heat cough | Make Huoren Quanshu (《麻科活人全书》) |
| Xing Su San | Citrus reticulata Blanco, Prunus armeniaca L.var.ansu Maxim., Ziziphus jujuba Mill., Platycodon grandiflorum (Jacq.) A.DC., Zingiber officinale Rosc., Citrus aurantium L., Peucedanum praeruptorum Dunn, Poria cocos (Schw.) Wolf, Pinellia ternate (Thunb.) Breit., Glycyrrhiza uralensis Fisch., Perilla frutescens (L.) Britt | Leading role | Phlegmatic heat cough, wind-heat cough | Detailed analysis of epidemic warm diseases (《温病条辨》) |
| Jie Ji Tou Sha Tang | Forsythia suspensa (Thunb.) Vahl, Schizonepeta tenuisfolia Briq., Peucedanum praeruptorum Dunn, Glycine max (L.) Merr., Arctium lappa L., Bambusa tuldoides Munro, Cryptotympana pustulata Fabricius, Belamcanda chinensis (L.) DC., Platycodon grandiflorum (Jacq.) A.DC., Glycyrrhiza uralensis Fisch., Pueraria lobata (Willd.) Ohwi, Lasiosphaera fenzlii Reich., Bombyx mori Linnaeus, Spirodela polyrrhiza (L.) Schleid | Leading role | Phlegmatic heat cough, wind-heat cough | Dinshi Yian (《丁氏医案》) |
| Bai Du San | Mentha haplocalyx Briq., Panax ginseng C. A. Mey., Notopterygium incisum Ting ex H. T. Chang, Zingiber officinale Rosc., Angelica pubescens Maxim.f. biserrata Shan et Yuan, Glycyrrhiza uralensis Fisch., Citrus aurantium L., Poria cocos (Schw.) Wolf, Ligusticum chuanxiong Hort., Peucedanum praeruptorum Dunn, Bupleurum chinense DC., Platycodon grandiflorum (Jacq.) A.DC | Leading role | Phlegmatic heat cough, wind-heat cough | Taiping Huimin Hejiju Fang (《太平惠民和剂局方》), Direct Formula of Pediatric Medicine Syndrome (《小儿药证直诀》) |
| Shen Su Yin | Citrus reticulata Blanco, Aucklandia lappa Decne., Platycodon grandiflorum (Jacq.) A.DC., Poria cocos (Schw.) Wolf, Peucedanum praeruptorum Dunn, Pinellia ternate (Thunb.) Breit., Citrus aurantium L., Pueraria lobata (Willd.) Ohwi, Perilla frutescens (L.) Britt., Glycyrrhiza uralensis Fisch., Panax ginseng C. A. Mey | Leading role | Phlegmatic heat cough, wind-heat cough | Taiping Huimin Hejiju Fang (《太平惠民和剂局方》) |
| Cang Lin San | Notopterygium incisum Ting ex H. T. Chang, Panax ginseng C. A. Mey., Mentha haplocalyx Briq., Zingiber officinale Rosc., Oryza sativa L., Citrus aurantium L., Poria cocos (Schw.) Wolf, Angelica pubescens Maxim.f. biserrata Shan et Yuan, Ligusticum chuanxiong Hort., Bupleurum chinense DC., Peucedanum praeruptorum Dunn, Platycodon grandiflorum (Jacq.) A.DC., Glycyrrhiza uralensis Fisch | Leading role | Phlegmatic heat cough, wind-heat cough | Prescriptions for Universal Relief (《普济方》) |
| Qing Yan Shuang He Yin | Schizonepeta tenuisfolia Briq., Pueraria lobata (Willd.) Ohwi, Lonicera japonica Thunb., Platycodon grandiflorum (Jacq.) A.DC., Peucedanum praeruptorum Dunn, Glycyrrhiza uralensis Fisch., Juncus effusus L., Poria cocos (Schw.) Wolf, Scrophularia ningpoensis Hemsl., Fritillaria cirrhosa D.Don, Paeonia suffruticosa Andr., Paeonia lactiflora Pall., Angelica sinensis (Oliv.) Diels, Rehmannia glutinosa Libosch | Leading role | Phlegmatic heat cough, wind-heat cough | Houke Zizhen Ji (《喉科紫珍集》) |
| Jing Fang Bai Du San | Ligusticum chuanxiong Hort., Platycodon grandiflorum (Jacq.) A.DC., Saposhnikovia divaricata (Turcz.) Schischk., Glycyrrhiza uralensis Fisch., Schizonepeta tenuisfolia Briq., Poria cocos (Schw.) Wolf, Citrus aurantium L., Angelica pubescens Maxim.f. biserrata Shan et Yuan, Notopterygium incisum Ting ex H. T. Chang, Peucedanum praeruptorum Dunn, Bupleurum chinense DC. | Leading role | Plegmatic heat cough, wind-heat cough | Shesheng Zongmiao Fang (《摄生众妙方》) |
| Xuan Du Fa Biao Tang | Mentha haplocalyx Briq., Zingiber officinale Rosc., Oryza sativa L., Bupleurum chinense DC., Citrus aurantium L., Platycodon grandiflorum (Jacq.) A.DC., Notopterygium incisum Ting ex H. T. Chang, Ligusticum chuanxiong Hort., Panax ginseng C. A. Mey., Peucedanum praeruptorum Dunn, Glycyrrhiza uralensis Fisch., Angelica pubescens Maxim.f. biserrata Shan et Yuan, Poria cocos (Schw.) Wolf | Supporting role | Phlegmatic heat cough, wind-heat cough | Golden Mirror of Medicine (《医宗金鉴》) |
| Su Zi Jiang Qi Tang | Zingiber officinale Rosc., Perilla frutescens (L.) Britt., Pinellia ternate (Thunb.) Breit., Ziziphus jujuba Mill., Citrus reticulata Blanco, Magnolia officinalis Rehd.et Wils., Cinnamomum cassia Presl, Peucedanum praeruptorum Dunn, Glycyrrhiza uralensis Fisch., Angelica sinensis (Oliv.) Diels | Supporting role | Phlegmatic heat cough, wind-heat cough | Taiping Huimin Hejiju Fang (《太平惠民和剂局方》) |
| Qiang Huo Sheng Feng Tang | Scutellaria baicalensis Georgi., Atractylodes macrocephala Koidz., Bupleurum chinense DC., Glycyrrhiza uralensis Fisch., Citrus aurantium L., Schizonepeta tenuisfolia Briq., Notopterygium incisum Ting ex H. T. Chang, Angelica dahurica (Fisch.ex Hoffm.) Benth.et Hook.f., Mentha haplocalyx Briq., Platycodon grandiflorum (Jacq.) A.DC., Peucedanum praeruptorum Dunn, Saposhnikovia divaricate (Turcz.) Schischk., Angelica pubescens Maxim.f. biserrata Shan et Yuan, Ligusticum chuanxiong Hort | Supporting role | Phlegmatic heat cough, wind-heat cough | Yuanji Qiwei (《原机启微》) |
Traditional Chinese medicine (TCM) prescriptions utilizing Peucedanum praeruptorum Dunn.
5 Phytochemistry
P. praeruptorum is reported to contain a diverse array of phytochemicals, including simple coumarins (1–13), pyranocoumarins (14–66), furanocoumarins (67–94), ketones (95, 96), sterols (97, 98), and organic acids (99–105), and others (106–119) (Table 2). The majority of these phytochemicals were isolated from root tissues, which are the traditional medicinal material. Among these isolated metabolites, angular pyranocoumarins (e.g., praeruptorins A and B) are the most abundant bioactive metabolites in P. praeruptorum tissues (Song et al., 2015).
TABLE 2
| Molecular class | Phytochemical | Tissue | Identification and isolation methodsa | Extract type | References |
|---|---|---|---|---|---|
| Simple coumarins | Umbelliferone 1 | Root | MRCC, NMR, OCC, MS, SGCC | Ethanol | Zhang et al. (2011) |
| Scopoletin 2 | Root | HPLC, HREIMS, IR, UV, NMR, SGCC | Petroleum ether | Kong et al. (1994b) | |
| Isoscopoletin 3 | Root | MRCC, SGCC, OCC, NMR, MS | Ethanol | Zhang et al. (2011) | |
| Isofraxidin 4 | Root | OHPLCc18/c30, NMR, EIMS | Water | Ishii et al. (2008) | |
| 8-carboxy-7-hydroxy coumarin 5 | Root | OHPLCc18/c30, NMR, EIMS | Water | Ishii et al. (2008) | |
| Skimmin 6 | Root | HPLC, NMR, EIMS | N-butanol | Okuyama et al. (1989) | |
| Scopolin 7 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Osthenol 8 | Root | HPLC | Ethanol | Chen et al. (2021) | |
| Praeroside VI 9 | Root | EIMS, OHPLCc18/c30, NMR | Water | Ishii et al. (2008) | |
| Apiosylskimmin 10 | Root | EIMS, OHPLCc18/c30, NMR | Water | Ishii et al. (2008) | |
| Hymexelsin 11 | Root | EIMS, OHPLCc18/c30, NMR | Water | Ishii et al. (2008) | |
| Eleutheroside B1 12 | Root | MS, NMR, OCC, SGCC | Ethanol | Zhang et al. (2009) | |
| (−)-peucedanol 13 | Root | EIMS, NMR, SGCC | Petroleum ether | Kong et al. (1993a) | |
| Pyranocoumarin | Praeruptorin C 14 | Root | FC/ACC, NMR | Diethyl ether/petroleum ether | Chen et al. (1979) |
| Praeruptorin E 15 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Qianhucoumarin D 16 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Qianhucoumarin A 17 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Khellactone 18 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Qianhucoumarin B 19 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| (9R,10R)-9-acetoxy-8,8-dimethyl-9,10-dihydro-2H,8H-benzo [1,2-b:3,4-b’] dipyran-2-one-10-yl-ester 20 | Root | HPLC | Ethanol | Chen et al. (2021) | |
| (±) cis-4′-acetyl-3′-crotonoykhellactone 21 | Root | MS, HPLC, NMR | Ethanol | Chen et al. (2021) | |
| Qianhucoumarin E 22 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| Hyuganin D 23 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| Qianhucoumarin I 24 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| Hyuganin C 25 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| Qianhucoumarin J 26 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| Praeruptorin B 27 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| (Chen et al.)-Praeruptorin A 28 | Root | HPLC, NMR | Boiling light petroleum | Xiong et al. (2012) | |
| Cis-3′-isovaleryl-4′-senecioylkhellactone 29 | Root | MS, SGCC, HPLC, NMR | Ethanol extract | Jong et al. (1992) | |
| Decursinol angelate 30 | Root | OCC, HR-TOF-MS, GPC, NMR, PHPLC, SGCC | Ethanol | Liu (2020) | |
| 3′(S),4′(S)-3′,4′-disenecioyl-3′, 4′-dihydroseselin 31 | Root | LCC, HPLC, NMR, ACC, SGCC | Petroleum ether | Chang (1998) | |
| 3′(R)-O-acetyl-4′(S)-O-angeloylkhellact 32 | Root | MS, PHPLC, NMR | Ethanol | Lou et al. (2004) | |
| 3′, 4′-disenecioyl-cis-khellactone 33 | Root | AC, MS, HPLC, NMR | Crude | Cheong et al. (2002) | |
| Pteryxin 34 | Root | OCC, NMR, PHPLC, HR-TOF-MS, SGCC, GPC | Ethanol | Liu (2020) | |
| Selinidin 35 | Root | HR-ESI-MS NMR, FC/SNAP | Ethanol | Lee et al. (2015) | |
| Isobocconin 36 | Root | ACC, HPLC, NMR | Ethanol | Chang and Li (1999a) | |
| Aegelinol 37 | Root | ACC, NMR, HPLC | Ethanol | Chang and Li (1999a) | |
| Suksdorfin 38 | Root | FC/SNAP, NMR, HR-ESI-MS | Ethanol | Lee et al. (2015) | |
| D-laserpitin 39 | Root | FC/SNAP, NMR, HR-ESI-MS | Ethanol | Lee et al. (2015) | |
| (−)-trans-khellactone 40 | Root | EIMS, HPLC, NMR, SGCC | Petroleum ether | Kong et al. (1993b) | |
| (+)-cis-khellactone 41 | Root | EIMS, NMR, SGCC, HPLC | Petroleum ether | Kong et al. (1993b) | |
| Neopeucedalactone 42 | Root | SCC-LH20, NMR, sPHPLC, SGCC | Ethanol | Li et al. (2020) | |
| Decursitin D 43 | Root | NMR, SGCC, EIMS | CHCl3 | Wang et al. (2018) | |
| Praeroside V 44 | Root | OHPLCc18, NMR, MS | Acetone | Takata et al. (1988) | |
| Cis-3′,4′-diisovalerylkhellactone 45 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Praeroside III 46 | Root | NMR, OHPLCc18, MS | Acetone | Takata et al. (1988) | |
| Praeroside II 47 | Root | NMR, OHPLCc18, MS | Acetone | Takata et al. (1988) | |
| (±)-peuformosin 48 | Root | NMR, HPLC, MS | Ethanol | Chen et al. (2021) | |
| Praeruptorin D 49 | Root | FC/ACC, NMR | Diethyl ether/petroleum ether | Chen et al. (1979) | |
| Peucedanocoumarin II 50 | Root | EIMS, NMR, SSHPLC | Acetone | Takata et al. (1990) | |
| Isoepoxypteryxin 51 | Root | HPLC | Ethanol | Chen et al. (2021) | |
| Qianhucoumarin H 52 | Root | EIMS, NMR. SGCC, IR | Petrol | Kong et al. (1996) | |
| Praeroside IV 53 | Root | MS, NMR, OHPLCc18 | Acetone | Takata et al. (1988) | |
| (+)-Praeruptorin A 54 | Root | NMR, HPLC, MS | Boiling light petroleum | Xiong et al. (2012) | |
| (±)-cis-4′-ethy-3′-tigloylkhellactone 55 | Root | NMR, OCC, HR-TOF-MS, SGCC, PHPLC, GPC | Ethanol | Liu (2020) | |
| (3S′,4S′)-3-angeloyloxy-4-hydroxy-3,4-dihydroSeselin 56 | Root | HR-TOF-MS, SGCC, NMR, PHPLC, OCC, GPC | Ethanol | Liu (2020) | |
| Hyuganin B 57 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| Corymbocoumarin 58 | Root | HR-TOF-MS, NMR, OCC, GPC, PHPLC, SGCC | Ethanol | Liu (2020) | |
| Pd-C-II 59 | Root | EIMS, NMR, PHPLC, SGCC | Ethanol | Wang et al. (2018) | |
| Peucedanocoumarin I 60 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| (+)-samidin 61 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| (3′S,4′S)-3′-O-isobutyroyl-4′-O-isovaleroylkhellactone 62 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| Pd-Ib 63 | Root | HPLC, MS | Methanol | Okuyama and Shibata (1981) | |
| Qianhucoumarin C 64 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| Pd-C-I 65 | Root | SGCC, NMR, MS | Petroleum ether | Kong et al. (1994a) | |
| Peucedanocoumarin III 66 | Root | SSHPLC, NMR, EIMS | Acetone | Takata et al. (1990) | |
| Furanocoumarins | Psoralen 67 | Root, stem, leaf | HPLC-EIMS | Methanol | Jian et al. (2020) |
| Angelicin 68 | Root, stem, leaf | HPLC-EIMS | Methanol | Jian et al. (2020) | |
| Xanthotoxin 69 | Cork, phloem, cambium, xylem, whole root | MS, HPLC-DAD | Methanol | Chen et al. (2019) | |
| Bergapten 70 | Cork, phloem, cambium, xylem, whole root | MS, HPLC-DAD | Methanol | Chen et al. (2019) | |
| Imperatorin 71 | Cork, phloem, cambium, xylem, whole root | MS, HPLC-DAD | Methanol | Chen et al. (2019) | |
| Deltoin 72 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| Isopimpinellin 73 | Root | UHPLC/ToFMS, MS | Methanol | Chen et al. (2019) | |
| Rutaretin 74 | Root | HPLC, NMR, MS | Ethanol | Chen et al. (2021) | |
| Arnocoumarin 75 | Root | MS, NMR, ACC, SGCC, HPLC | Ethanol | Chang and Li (1999b) | |
| Qianhucoumarin G 76 | Root | SGCC, IR, NMR, EIMS | Petrol | Kong et al. (1996) | |
| Nodakenetin 77 | Root | MS, NMR, UV, RLCC, MRCC, SGCC | N-butanol | Asahara et al. (1984) | |
| Nodakenetin tiglate 78 | Root | HPLC, 2DHCCC, NMR, ESI-MS | Ethanol | Liu et al. (2014) | |
| Marmesinin 79 | Root | HPLC, NMR, MS | Ethanol | Chen et al. (2021) | |
| Oxypeucedanin 80 | Root | SGCC, OCC, NMR, MS | Ethanol | Zhang et al. (2011) | |
| Marmesin-11-O-β-D-glucopyranosyl (1→6)-β-D-glucopyranoside 81 | Root | ESI-MS, NMR, HPLC | Ethanol | Wang et al. (2018) | |
| Rutarin 82 | Root | EIMS, NMR, HPLC | N-butanol | Okuyama et al. (1989) | |
| Oxypeucedanin hydrate 83 | Root | SGCC, OCC, NMR, MS | Ethanol | Zhang et al. (2011) | |
| Marmesin 84 | Root | HPLC, NMR, EIMS | N-butanol | Okuyama et al. (1989) | |
| Sphondin 85 | Root | MS, NMR, OCC, SGCC | Ethanol | Zhang et al. (2011) | |
| Oroselol 86 | Root | EIMS, NMR, SGP, SGCC | Ethanol | Wang et al. (2018) | |
| Peucedanoside A 87 | Root | MS, NMR, sPHPLC, SGCC, TLC | Methanol | Chang et al. (2007) | |
| Peucedanoside B 88 | Root | MS, NMR, sPHPLC, SGCC, TLC | Methanol | Chang et al. (2007) | |
| Apterin 89 | Root | MS, NMR, sPHPLC, SGCC, TLC | Methanol | Chang et al. (2007) | |
| Praeroside VII 90 | Root | NMR, TLC, sPHPLC, SGCC | Methanol | Chang et al. (2008) | |
| Isorutarin 91 | Root | EIMS, NMR, HPLC | N-butanol | Okuyama et al. (1989) | |
| Nodakenin 92 | Root | MS, UHPLC/ToFMS | Methanol | Chen et al. (2019) | |
| Praeroside I 93 | Root | SGCC, GPC, PHPLC, OCC, NMR, HR-TOF-MS | Ethanol | Liu (2020) | |
| (2′S)-rutaretin-4′-O-(6-p-hydroxybenzoyl-β-D-glucopyranoside) 94 | Root | MS, NMR, HPLC | Ethanol | Chen et al. (2021) | |
| Ketone | Tanshinone I 95 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2005) |
| Tanshinone IIA 96 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2005) | |
| Sterol | β-sitosterol 97 | Stem, leaf | IR, SGCC, NMR, EIMS | Ethanol | Kong et al. (1993b) |
| Daucosterol 98 | Stem, leaf | IR, SGCC, NMR, EIMS | Ethanol | Kong et al. (1993a) | |
| Organic acid | Vanillic acid 99 | Root | SGCC, PCC, NMR, MS | Ethyl acetate | Kong et al. (1994a) |
| Gallic acid 100 | Root | SGCC, PCC, NMR, MS | Ethyl acetate | Kong et al. (1994a) | |
| Butyric acid 101 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2009) | |
| Palmitic acid 102 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2006) | |
| 4H-1-benzopyran-4-one,5-hydroxy-6-methoxy-2-phenyl-7-O-α-D-glucuronyl acid 103 | Root | MS, NMR, SGCC | Ethanol | Zhang et al. (2012) | |
| Tetracosanoic acid 104 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2006) | |
| 9,10-dihydrophenanthrinic acid 105 | Root | UV, IR, SIMS | Ethyl acetate | Zhang et al. (2010a) | |
| Others | 2,6-dimethyl quinoline 106 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2006) |
| 3-(4′-for mylphenoxy)-4-methoxybenzaldehyde 107 | Root | HPLC, NMR, MS | Ethanol | Chen et al. (2021) | |
| 3-(4′-formylphenoxy)-4-methoxybenzaldehyde 108 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| Bis(2-ethylhexyl) phthalate 109 | Root | HR-TOF-MS, NMR, PHPLC, OCC, GPC, SGCC | Ethanol | Liu (2020) | |
| 4-[β-D-apiofuranosyl-(1→6)-β-D-glucopyranosyloxy]-3-methox ypropiophenone 110 | Root | HPLC, NMR, MS | Ethanol | Chen et al. (2021) | |
| Baihuaqianhuoside 111 | Root | MS, NMR, PHPLC, SGCC | Ethanol | Asahara et al. (1984) | |
| Galactitol 112 | Root | EIMS, NMR, SGCC | Ethanol | Kong et al. (1993a) | |
| (−)-sclerodin 113 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2006) | |
| Adenoside 114 | Root | MRCC, SGCC, OCC, NMR, MS | Ethanol | Zhang et al. (2009) | |
| Acetylatractylodinol 115 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2005) | |
| 4H-1-benzopyran-4-one,5-hydroxy-6-methoxy-2-phenyl-7-O-α-D-glucuronyl methyl ester 116 | Root | MS, NMR, OCC, SGCC | Ethanol | Zhang et al. (2012) | |
| Polyacetylene 117 | Root | CS, NMR, HR-ESI-MS | Ethanol | Lee et al. (2015) | |
| D-mannitol monohexadecanoate 118 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2009) | |
| α-D-glucopyranose-1-hexadecanoate 119 | Root | MS, NMR, OCC, SGCC, MRCC | Ethanol | Zhang et al. (2009) |
Phytochemicals isolated from Peucedanum praeruptorum Dunn.
Note: adsorption chromatography (AC); alumina column chromatography (ACC); chromatographic separation (CS); electrospray ionization mass spectrometry (ESI-MS); flash chromatography using SNAP, Ultra cartridge (FC/SNAP); fractional crystallization/alumina column chromatography (FC/ACC); gel permeation chromatography (GPC); high performance liquid chromatography with octadecylsilyl (c18) (OHPLCc18); high performance liquid chromatography (HPLC); high performance liquid chromatography with octadecylsilyl (c18)/develosil (c30) (OHPLCc18/c30); high performance liquid chromatography-diode-array detector (HPLC-DAD); high performance liquid chromatography-electrospray ionization mass spectrometry (HPLC-EIMS); high-resolution electron ionization mass spectrometry (HREIMS); high-resolution-electrospray ionization-mass spectrometry (HR-ESI-MS); high-resolution-time-of-flight mass spectrometry (HR-TOF-MS); infrared spectroscopy (IR); lobar column chromatography (LCC); macroporous resin column chromatography (MRCC); mass spectrometry (MS); nuclear magnetic resonance (NMR); octadecylsilyl column chromatography (OCC); polyphthalamide column chromatography (PCC); preparative high performance liquid chromatography (PHPLC); Rp-8, reversed lobar column chromatography (RLCC); secondary ion mass spectroscopy (SIMS); semi-preparative high performance liquid chromatography (sPHPLC); Senshu scientific high performance liquid chromatography (SSHPLC); Sephadex gel purification (SGP); Sephadex LH-20, column chromatography (SCC-LH20); silica gel column chromatography (SGCC); thin-layer chromatography (TLC); two-dimensional hyphenation of counter-current chromatography (2DHCCC); ultra-high performance liquid chromatography/time of flight mass spectrometry (UHPLC/ToFMS); ultraviolet spectrum (UV).
5.1 Simple coumarins
Thirteen simple coumarins (Figure 3) have been isolated from P. praeruptorum root tissues, including umbelliferone 1, scopoletin 2, isoscopoletin 3, isofraxidin 4, 8-carboxy-7-hydroxy coumarin 5, skimmin 6, scopolin 7, osthenol 8, praeroside VI 9, apiosylskimmin 10, hymexelsin 11, eleutheroside B1 12, and (−)-peucedanol 13. However, the pharmacological activities of these simple coumarins have rarely been reported.
FIGURE 3
Simple coumarins isolated from Peucedanum praeruptorum Dunn.
5.2 Pyranocoumarins
Fifty-five pyranocoumarins (Figure 4) have been isolated from P. praeruptorum root tissues, including praeruptorin C 14, praeruptorin E 15, qianhucoumarin D 16, qianhucoumarin A 17, khellactone 18, qianhucoumarin B 19, (9R,10R)-9-acetoxy-8,8-dimethyl-9,10-dihydro-2H, 8H-benzo [1,2-b:3,4-b′]dipyran-2-one-10-yl-ester 20, (±)-cis-4′-acetyl-3′-crotonoykhellactone 21, qianhucoumarin E 22, hyuganin D 23, qianhucoumarin I 24, hyuganin C 25, qianhucoumarin J 26, praeruptorin B 27, (−)-praeruptorin A 28, cis-3′-isovaleryl-4′-senecioylkhellactone 29, decursinol angelate 30, 3′(S),4′(S)-3′,4′-disenecioyl-3′,4′-dihydroseselin 31, 3′-O-acetyl-4′(S)-O-angeloyl-khellact 32, 3′,4′-disenecioyl-cis-khellactone 33, pteryxin 34, selinidin 35, isobocconin 36, aegelinol 37, suksdorfin 38, D-laserpitin 39, (−)-trans-khellactone 40, (+)-cis-khellactone 41, neopeucedalactone 42, decursitin D 43, praeroside V 44, cis-3′,4′-diisovalerylkhellactone 45, praeroside III 46, praeroside II 47, (±)-peuformosin 48, praeruptorin D 49, peucedanocoumarin II 50, isoepoxypteryxin 51, qianhucoumarin H 52, praeroside IV 53, (+)-praeruptorin A 54, (±)-cis-4′-ethy-3′-tigloylkhellactone 55, (3S′,4S′)-3-angeloyloxy-4-hydroxy-3,4-dihydroseselin 56, hyuganin B 57, corymbocoumarin 58, Pd-C-II 59, peucedanocoumarin I 60, (+)-samidin 61, (3′S, 4′S)-3′-O-isobutyroyl-4′-O-isovaleroylkhellactone 62, Pd-Ib 63, qianhucoumarin C 64, Pd-C-I 65, and peucedanocoumarin III 66. Research suggests that praeruptorin B possesses antitumor activity, and praeruptorin E possesses anti-inflammatory activity (Yu et al., 2012; Lin et al., 2020).
FIGURE 4
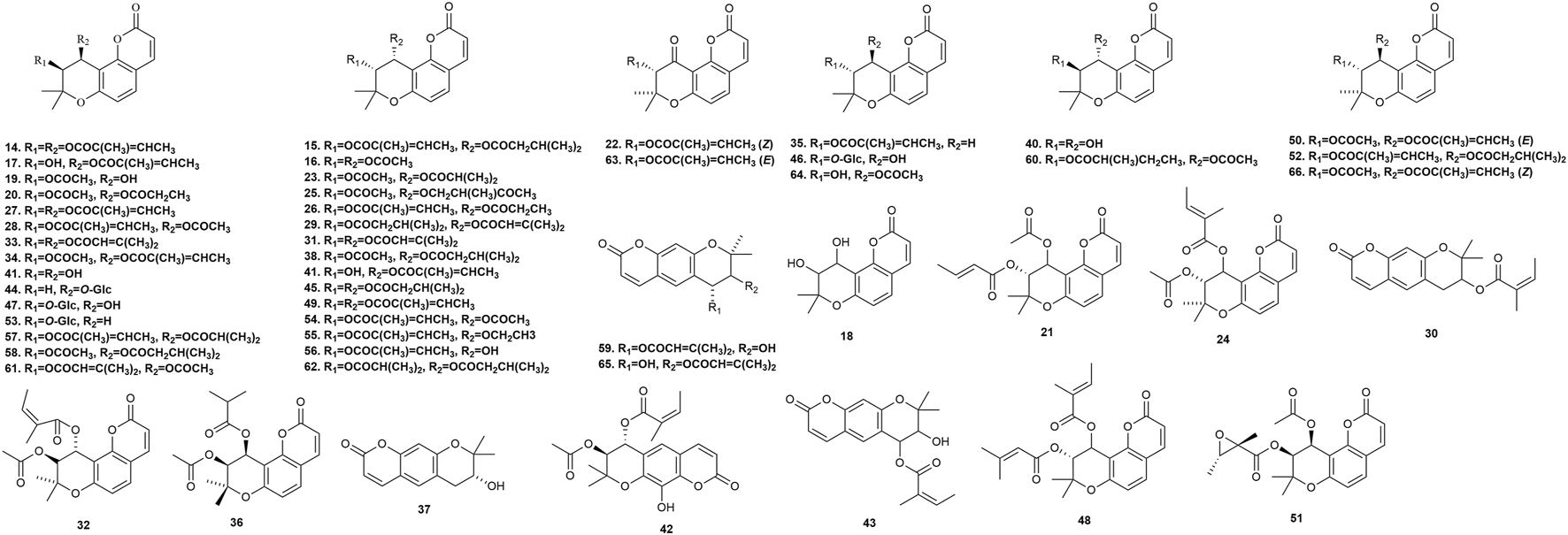
Pyranocoumarins isolated from Peucedanum praeruptorum Dunn.
5.3 Furanocoumarins
Furanocoumarins possess neuroprotective, anti-inflammatory, and anticancer activities in animals, and serve as phytotoxins and allelochemicals in plants (Jian et al., 2020). Twenty-nine furanocoumarins (Figure 5) have been isolated from P. praeruptorum root tissues, including psoralen 67, angelicin 68, xanthotoxin 69, bergapten 70, imperatorin (IMP) 71, deltoin 72, isopimpinellin 73, rutaretin 74, arnocoumarin 75, qianhucoumarin G 76, nodakenetin 77, nodakenetin tiglate 78, marmesinin 79, oxypeucedanin 80, marmesin-11-O-β-D-glucopyranosyl (1→6)-β-D-glucopyranoside 81, rutarin 82, oxypeucedanin hydrate 83, marmesin 84, sphondin 85, oroselol 86, peucedanoside A 87, peucedanoside B 88, apterin 89, praeroside VII 90, isorutarin 91, nodakenin 92, praeroside I 93, and (2′S)-rutaretin-4′-O-(6-p-hydroxybenzoyl-β-D-glucopyranoside) 94.
FIGURE 5
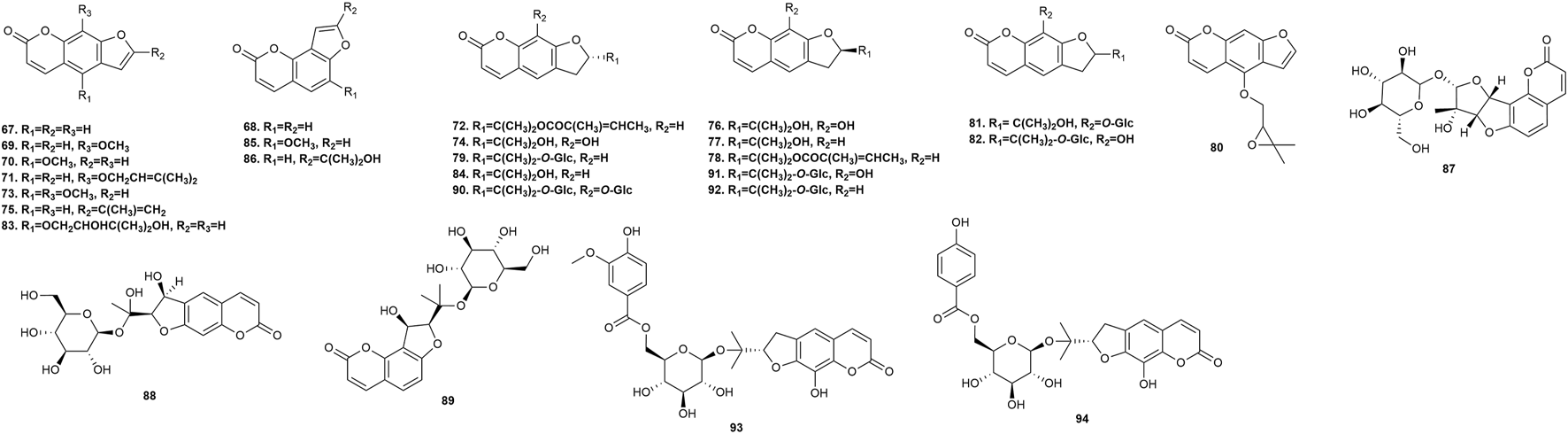
Furanocoumarins isolated from Peucedanum praeruptorum Dunn.
5.4 Ketones, sterols, and organic acids
Two ketones (tanshinone I 95 and tanshinone IIA 96) were confirmed in the roots of P. praeruptorum. Two sterols (β-sitosterol 97 and daucosterol 98) and seven organic acids (vanillic acid 99, gallic acid 100, butyric acid 101, palmitic acid 102, 4H-1-benzopyran-4-one,5-hydroxy-6-methoxy-2-phenyl-7-O-α-D-glucuronyl acid 103, tetracosanoic acid 104, and 9,10-dihydrophenanthrinic acid 105) were confirmed in the stem and leaves of P. praeruptorum. However, the pharmacological activities of these ketones, sterols, and organic acids were not found in the available studies. Figure 6 shows the chemical structures of these phytochemicals.
FIGURE 6
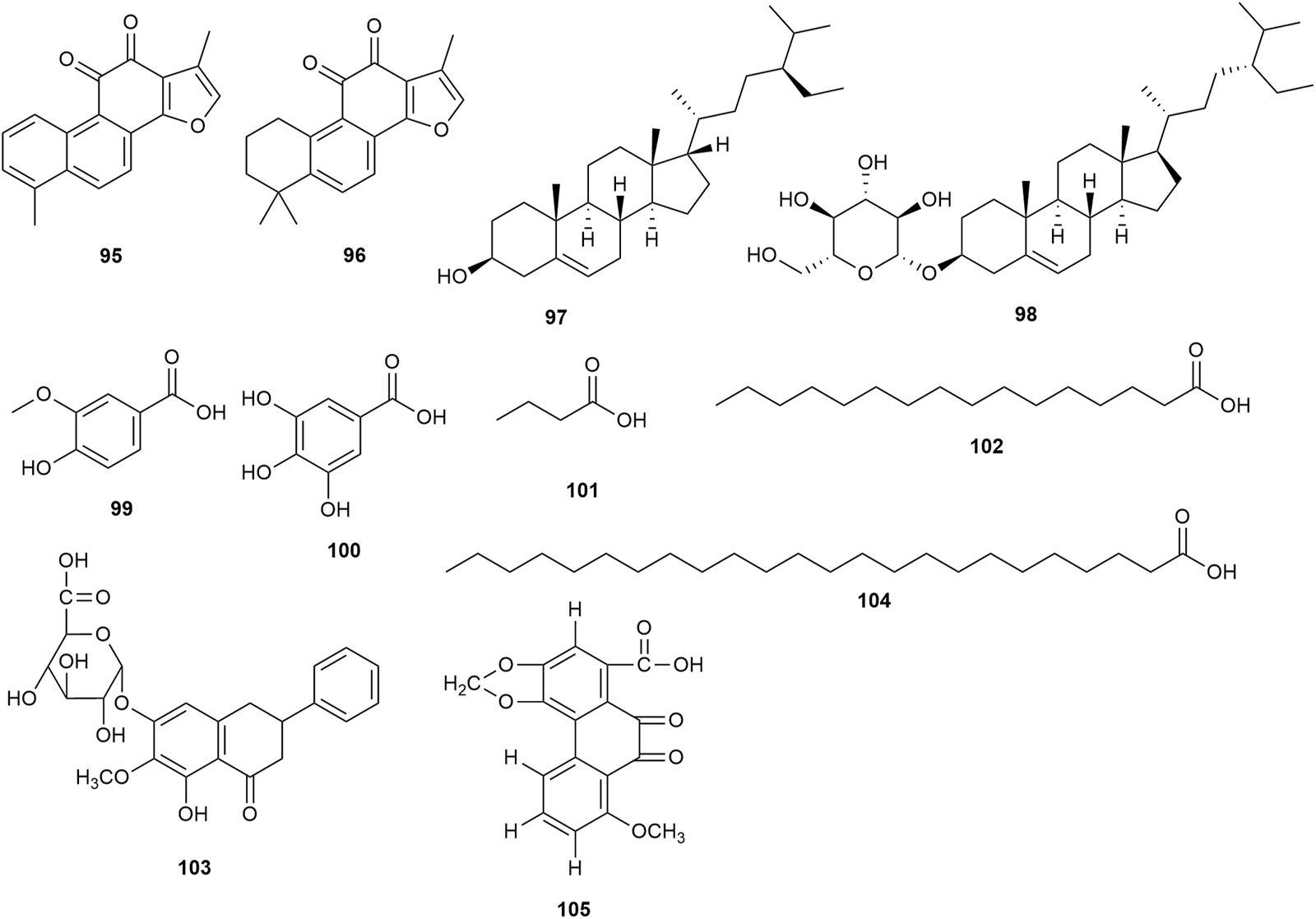
Ketones, sterols, and organic acids isolated from Peucedanum praeruptorum Dunn.
5.5 Other metabolites
Other metabolites (Figure 7), such as 2,6-dimethyl quinoline 106, 3-(4′-formylphenoxy)-4-methoxybenzaldehyde 107, 3-(4′-formylphenoxy)-4-methoxybenzaldehyde 108, bis(2-ethylhexyl) phthalate 109, 4-[β-D-apiofuranosyl-(1→6)-β-D-glucopyranosyloxy]-3-methoxypropiophenone 110, baihuaqianhuoside 111, galactitol 112, (−)-sclerodin 113, adenoside 114, acetylatractylodinol 115, 4H-1-benzopyran-4-one,5-hydroxy-6-methoxy-2-phenyl-7-O-α-D-glucuronyl methyl ester 116, polyacetylene 117, D-mannitol monohexadecanoate 118, and α-D-glucopyranose-1-hexadecanoate 119, have been isolated from P. praeruptorum root tissues. However, the pharmacological activities of these phytochemicals were not found in the available studies.
FIGURE 7
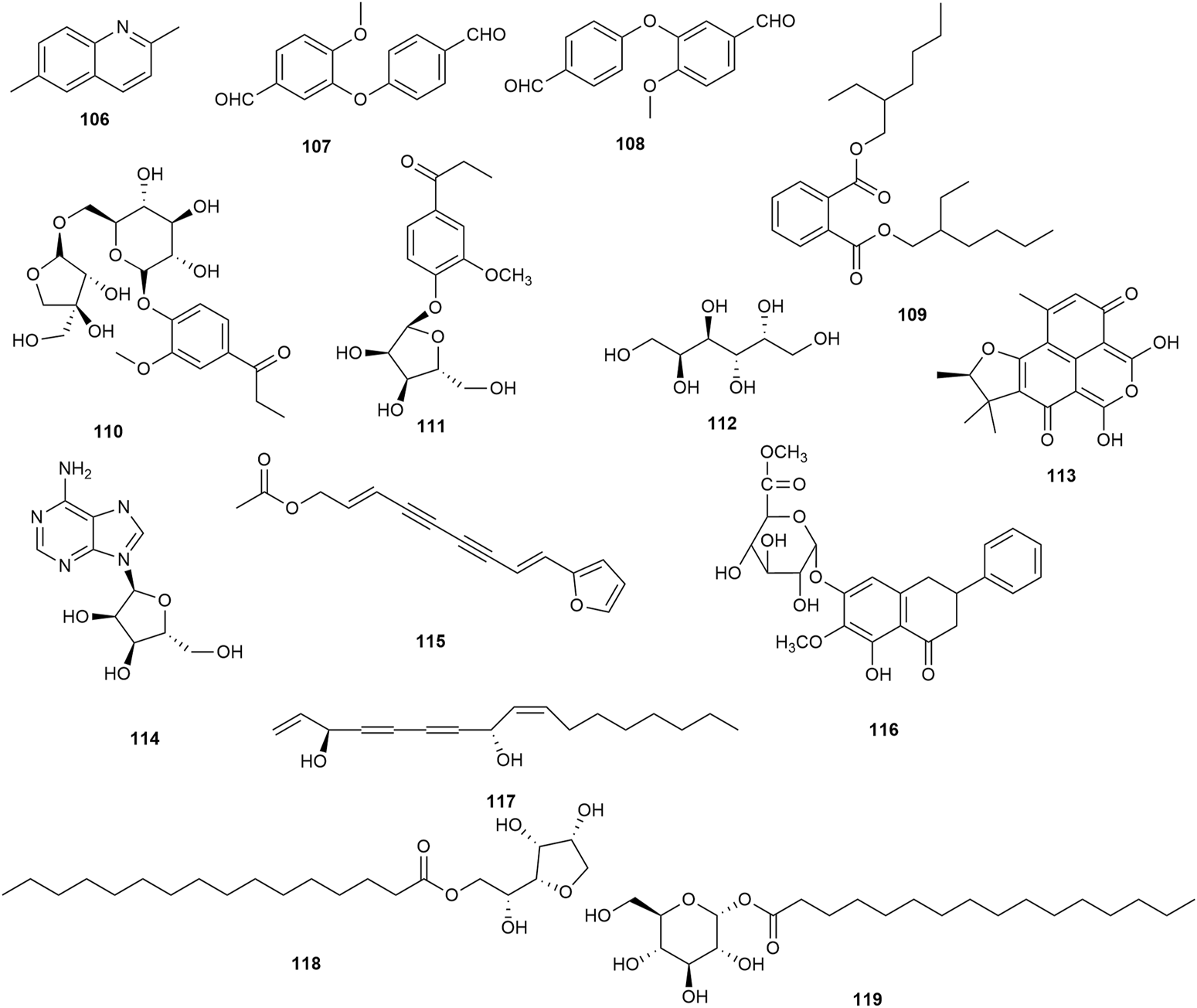
Additional phytochemicals isolated from Peucedanum praeruptorum Dunn.
6 Pharmacological activities
P. praeruptorum exhibits diverse pharmacological activities (Table 3), including anti-inflammatory, expectorant, antitussive, antitumor, neuroprotective, anti-osteoclastogenic, and antidepressant effects. The antitumor, immunoregulatory, and anti-inflammatory activities are the most notable, and putative molecular mechanisms are shown in Figure 8.
TABLE 3
| Pharmacological activity | Tested substance | Model | Experimental system | Type of study | Results | Dose range | Application period | References |
|---|---|---|---|---|---|---|---|---|
| Anti-inflammatory activity | Praeruptorin C | Mouse | Left paw | In vivo | Inhibited microglial activation; attenuated proinflammatory cytokine release; regulated excitatory transmission in ACC of CFA-injected mice | 3 mg/kg | 21 d | Su et al. (2021) |
| (±)-praeruptorin A | Murine model of chronic asthma | Lung | In vivo | Decreased expression of IgE (serum) and IL-4/-13 (BALF); suppressed airway inflammation, hyperresponsiveness, and remodeling; constrained TGF-β1 and pSmad2/3 expression and promoted Smad7 expression (lung tissue) and INF-γ (BALF) | 30, 60, 120 mg/kg | 56 d | Xiong et al. (2012) | |
| Praeruptorin D | Inflammatory Periodontal membrane cells | Cell culture | In vitro | Constrained TNF-ɑ and IL-1β expression | 10, 20, 30, 40 μg/mL | 1, 2, 3, 5 d | Yu (2022) | |
| Praeruptorin C | LPS-stimulated raw264.7 macrophage cells | Cell culture | In vitro | Inhibited NF-κB and STAT3 activation | 2, 4, 8, 16 μg/mL | 18 h | Yu et al. (2012) | |
| Praeruptorin D | LPS-stimulated raw264.7 macrophage cells | Cell culture | In vitro | Inhibited NF-κB and STAT3 activation | 2, 4, 8, 16 μg/mL | 18 h | Yu et al. (2012) | |
| Praeruptorin E | LPS-stimulated raw264.7 macrophage cells | Cell culture | In vitro | Inhibited NF-κB and STAT3 activation | 2, 4, 8, 16 μg/mL | 18 h | Yu et al. (2012) | |
| Imperatorin | RBL-2H3 allergic inflammatory cell | Cell culture | In vitro | Inhibited the degranulation rate of RBL-2H3 cells; inhibited the release of histamine, IL-3/-4/-6, TNF-α, and COX-2; promoted the expression of IFN-γ | 5, 10, 15 μmol/L | 1 h | Long et al. (2019) | |
| P. praeruptorum polysaccharides | RAW264.7 macrophages | Cell culture | In vitro | Increased accessory and costimulatory molecule expression, the secretion of chemokines and inflammatory factors, and phagocytosis/pinocytosis | 25, 50, 100, 200 μg/mL | 24 h | Zhao et al. (2022) | |
| Dl- praeruptorin A | LPS mouse model of acute lung injury | Lung | In vivo | Reduced lung inflammation | 10 μg/g | 24 h | Zhou et al. (2016) | |
| Dl- praeruptorin A | LPS-induced HUVECs | Cell culture | In vitro | Inhibited LPS-induced endothelial inflammation | 10, 20, 40 μmol/L | 24 h | Wang et al. (2012) | |
| Expectorant and antitussive effects | Peucedanum praeruptorum Dunn water extract | Mouse | Trachea | In vivo | Reduced phlegm | 45 g/kg | 1 h | Liu et al. (1997), Meng et al. (1997) |
| Praeruptorin C | Mouse | Trachea | In vivo | Reduced phlegm | 3 mg/kg, 10 mg/kg | 1 h | Liu et al. (2009) | |
| Nodakenin | Mouse | Trachea | In vivo | Reduced phlegm | 3 mg/kg, 10 mg/kg | 1 h | Liu et al. (2009) | |
| Nodakenin | BALB/c mouse | Mouse | In vivo | Decreased expression of IgE (serum) and IL-4/-13/-5 (BALF); suppressed airway inflammation and hyperresponsiveness; constrained nuclear P65/p-P65; promoted cytoplasmic P65 and IκBα; promoted DNA binding activity of NF-κB | 10 mg/kg | 5 d | Xiong et al. (2014) | |
| Raw Qianhu | Mouse | Trachea | In vivo | Strong expectorant and antitussive effects | 2.5, 5.0, 10.0 g/kg | 6 d | Zhang et al. (2010b) | |
| Honey-roasted Qianhu | Mouse | Trachea | In vivo | Strong expectorant and antitussive effects | 2.5, 5.0, 10.0 g/kg | 6 d | Zhang et al. (2010b) | |
| Raw Qianhu | Guinea pigs | Guinea pig | In vivo | Asthma relief | 2, 4, 8 g/kg | 3 d | Zhang et al. (2010b) | |
| Honey-roasted Qianhu | Guinea pigs | Guinea pig | In vivo | Asthma relief | 2, 4, 8 g/kg | 3 d | Zhang et al. (2010b) | |
| Antitumor activity | Praeruptorin B | Human RCC cell lines 786-O and ACHN | Cell culture | In vitro | Inhibited migrability and invasibility; inhibited cathepsin C and cathepsin V expression in ACHN and 786-O cell lines | 0, 10, 20, 30 μmol/L | 24 h | Lin et al. (2020) |
| Praeruptorin B | Ovarian cancer SK-OV-3 cells | Cell culture | In vitro | Inhibited SK-OV-3 cellular proliferation and migration; reduced the expression of c-myc, cyclind1, srebp-1c, and fasn mRNA/protein | 20, 40, 60 μmol/L | 24 h | Xue et al. (2021) | |
| Praeruptorin A | Ovarian cancer A2780/TAX cells | Cell culture | In vitro | Inhibited A2780/TAX cellular proliferation, viability, and migration via promoting apoptosis | 25 μmol/L | 48 h | Chen et al. (2022) | |
| Praeruptorin A | HeLa cells | Cell culture | In vitro | Induction of G0/G1 phase cell cycle arrest; upregulated expression of tissue inhibitor of metalloproteinase-2, Rb, and p16/21/27; downregulated expression of matrix metalloproteinase-2, S-phase kinase-associated protein 2, and cyclin D1 | 0, 10, 20, 30 μmol/L | 24 h | Wu et al. (2017) | |
| Praeruptorin A | HeLa cells | Cell culture | In vitro | Enhanced the ability of MEK1/2 inhibitor PD98059 to downregulate metalloproteinase-2; suppressed the activation of SERK1/2; upregulated expression of tissue inhibitor of metalloproteinase-2 | 0, 20 μmol/L | 24 h | Wu et al. (2017) | |
| Praeruptorin A | SiHa cells | Cell culture | In vitro | Upregulated the expression of tissue inhibitor of metalloproteinase-2; downregulated the expression of matrix metalloproteinase-2 | 0, 10, 20, 30 μmol/L | 24 h | Wu et al. (2017) | |
| Praeruptorin A | Human HCC cells | Cell culture | In vitro | Reduced the migrability and invasibility of human HCC cells; activated extracellular signal-regulated kinase signaling; downregulated matrix metalloproteinase-1 expression | 0, 10, 20, 30 μmol/L | 24 h | Yu et al. (2021) | |
| Praeruptorin A | LS174T cells | Cell culture | In vitro | Pregnane X receptor-mediated induction of cytochrome P450 3A4 expression and activity | 2.5, 10, 40 μmol/L | 48 h | Huang et al. (2013) | |
| Praeruptorin A | SGC7901 human gastric cancer cells | Cell culture | In vitro | Cytotoxicity toward SGC7901 cells | 10, 50, 100 μmol/L | 24 h | Liang et al. (2010) | |
| Praeruptorin B | SGC7901 human gastric cancer cells | Cell culture | In vitro | Cytotoxicity toward SGC7901 cells | 10, 50, 100 μmol/L | 24 h | Liang et al. (2010) | |
| Praeruptorin A | SGC7901 human gastric cancer cells | Cell culture | In vitro | Complemented the effect of Doxorubincin on SGC7901 cells | 50, 100 μmol/L | 24 h | Liang et al. (2010) | |
| Praeruptorin A | HepG2 cells | Cell culture | In vitro | Constitutive androstane receptor-mediated upregulation of multidrug resistance-associated protein 2 in vitro | 10, 25, 50 μmol/L | 24 or 28 h | Zhou et al. (2013) | |
| Praeruptorin C | HepG2 cells | Cell culture | In vitro | Constitutive androstane receptor-mediated upregulation of multidrug resistance-associated protein 2 in vitro | 10, 25, 50 μmol/L | 24 or 28 h | Zhou et al. (2013) | |
| Praeruptorin A | H1975 (EGFR L858R/T790M double-mutant, EGFR TKI-resistant) human non-small-cell lung cancer cells | Cell culture | In vitro | Induced apoptosis in H1975 cells | 0, 50, 100 μg/mL | 72 h | Park et al. (2022) | |
| Pteryxin | H1975 (EGFR L858R/T790M double-mutant, EGFR TKI-resistant) human non-small-cell lung cancer cells | Cell culture | In vitro | Induced apoptosis in H1975 cells | 0, 50, 100 μg/mL | 72 h | Park et al. (2022) | |
| Praeruptorin A | H1975 (EGFR L858R/T790M double-mutant, EGFR TKI-resistant) human non-small-cell lung cancer cells, PC9/ER (erlotinib-resistant) human non-small-cell lung cancer cells | Cell culture | In vitro | Suppressed HGF-induced phosphorylation of MET. | 0, 50, 100 μg/mL | 2 h | Park et al. (2022) | |
| Pteryxin | H1975 (EGFR L858R/T790M double-mutant, EGFR TKI-resistant) human non-small-cell lung cancer cells, PC9/ER human non-small-cell lung cancer cells | Cell culture | In vitro | Suppressed HGF-induced phosphorylation of MET. | 0, 50, 100 μg/mL | 2 h | Park et al. (2022) | |
| Praeruptorin A | H1975 (EGFR L858R/T790M double-mutant, EGFR TKI-resistant) human non-small-cell lung cancer cells | Cell culture | In vitro | Dephosphorylated AKT. | 0, 50, 100 μg/mL | 2 h | Park et al. (2022) | |
| (±)-4′-O- acetyl-3′-O-angeloyl- cis- khellactone | U266 cells | Cell culture | In vitro | Induced apoptosis to suppress cell proliferation | 0, 10, 20, 30, 40 μg/mL | 24 h | Yu et al. (2015) | |
| Neopeuceda-lactone | Human leukemic HL-60 cell lines | Cell culture | In vitro | Inhibited cell growth in vitro | - | 3 d | Li et al. (2020) | |
| Neopeuceda-lactone | Human leukemic THP-1 cell lines | Cell culture | In vitro | Inhibited cell growth in vitro | - | 3 d | Li et al. (2020) | |
| Neopeuceda-lactone | Human prostate cancer PC-3 cell lines | Cell culture | In vitro | Inhibited cell growth in vitro | - | 3 d | Li et al. (2020) | |
| Neuroprotective activity | Praeruptorin C | Primary neurons | Cell culture | In vitro | Reversed N-methyl-D-aspartate-induced upregulation of GluN2B-containing N-methyl-D-aspartate receptors | 0, 1, 10 μmol/L | 24 h | Yang et al. (2013) |
| Praeruptorin C | Primary neurons | Cell culture | In vitro | Inhibited N-methyl-D-aspartate-induced neuronal apoptosis via reversing intracellular Ca2+ overload and balancing the Bcl-2/Bax ratio | 0, 1, 10 μmol/L | 24 h | Yang et al. (2013) | |
| Praeruptorin C | 3-nitropropionic-treated acid mouse | Mouse | In vivo | Alleviated excitotoxicity, motor deficits, and depressive behavior in 3-nitropropionic acid-treated mice | 1.5, 3.0 mg/kg | 3 d | Wang et al. (2017) | |
| Anti-osteoclastogenic activity | Praeruptorin C | Osteoclasts | Cell culture | In vitro | Attenuated the formation of osteoclasts via inhibition of JNK and NF-κB pathways, without altering p38 or ERK. | 0, 20 μmol/L | 4 h | Liu et al. (2017) |
| Praeruptorin C | OVX mouse | Mouse | In vivo | Constrained osteoclastic bone resorption and F-actin ring formation | 5, 10 μmol/L | 28 d | Liu et al. (2017) | |
| Praeruptorin A | Bone marrow–derived macrophages | Cell culture | In vitro | Constrained Akt and p 38 signaling, and RANKL-mediated osteoclast differentiation | 10 μmol/L | 30 min | Yeon et al. (2014) | |
| Peucedanum praeruptorum Dunn | Inflammatory periodontal membrane cells | Cell culture | In vitro | Induced the expression of osteogenic genes RUNX-2, ALP, and OCN, and differentiation of inflammatory periodontal membrane cells | 10, 20, 30, 40 μg/mL | 1, 2, 3, 5 d | Yu (2022) | |
| Antidepressant activity | Dl- praeruptorin A | Chronic unpredicted mildly stressed rat | Rat | In vivo | Improved the depressive behavior of chronic mildly-stressed rats | 10, 30, 60 mg/kg | 28 d | Wang and Xu (2014) |
| Imperatorin | Rat (male offspring) | Rat | In vivo | Enhanced 5-HT1AR expression, 5-HT level, and sucrose preference; increased the incidence of grooming, rearing, and crossing behaviors; reduced immobility; and decreased 5-HTT expression | 15, 30 mg/kg | 28 d | Zheng et al. (2019) | |
| Other activity | P. praeruptorum alcohol extracts | Rat | Left ventrical | In vivo | Affected ventricular remodeling and apoptosis-related proteins in different ways, and had a positive influence on ventricular remodeling | 1, 2, 4 g/mL | 28 d | Tu et al. (2004) |
| Total courmarins | Mouse | Mouse | In vivo | Prolonged the hypnotic duration of pentobarbital sodium in a dose-dependent manner; inhibited the activities of aniline hydroxylase and aminopyrine N-demethylase; minimally influenced the hypnotic effect of barbital sodium; inhibited the activity of hepatic microsomal drug-metabolizing enzymes | 50, 100, 200 mg/kg | 1 h | Wang et al. (2004) | |
| Nodakenin | Mouse | Mouse | In vivo | Reversed scopolamine-induced cognitive impairments | 0, 2.5, 5, 10, 20 mg/kg | 4 d | Kim et al. (2007) | |
| Nodakenin | Mouse | Mouse | In vivo | Inhibited acetylcholinesterase activity | 10 mg/kg | 24 h | Kim et al. (2007) | |
| Dl-praeruptorin A | Rat | Cell culture | In vitro | Prevented postischemic cell death in rat heart | 0.5, 1.0, 2.0 mg/kg | 2 h | Chang et al. (2003) | |
| Praeruptorin C | Rat | Myocardial tissue | In vitro | Reduced ischemia/reperfusion injury induced by coronary ligation | 5, 15, 30 mg/kg | 3 d | Liu and Wang (2009) |
Pharmacology of phytochemicals extracted from Peucedanum praeruptorum Dunn.
“-” denotes no useful information found in the study.
FIGURE 8
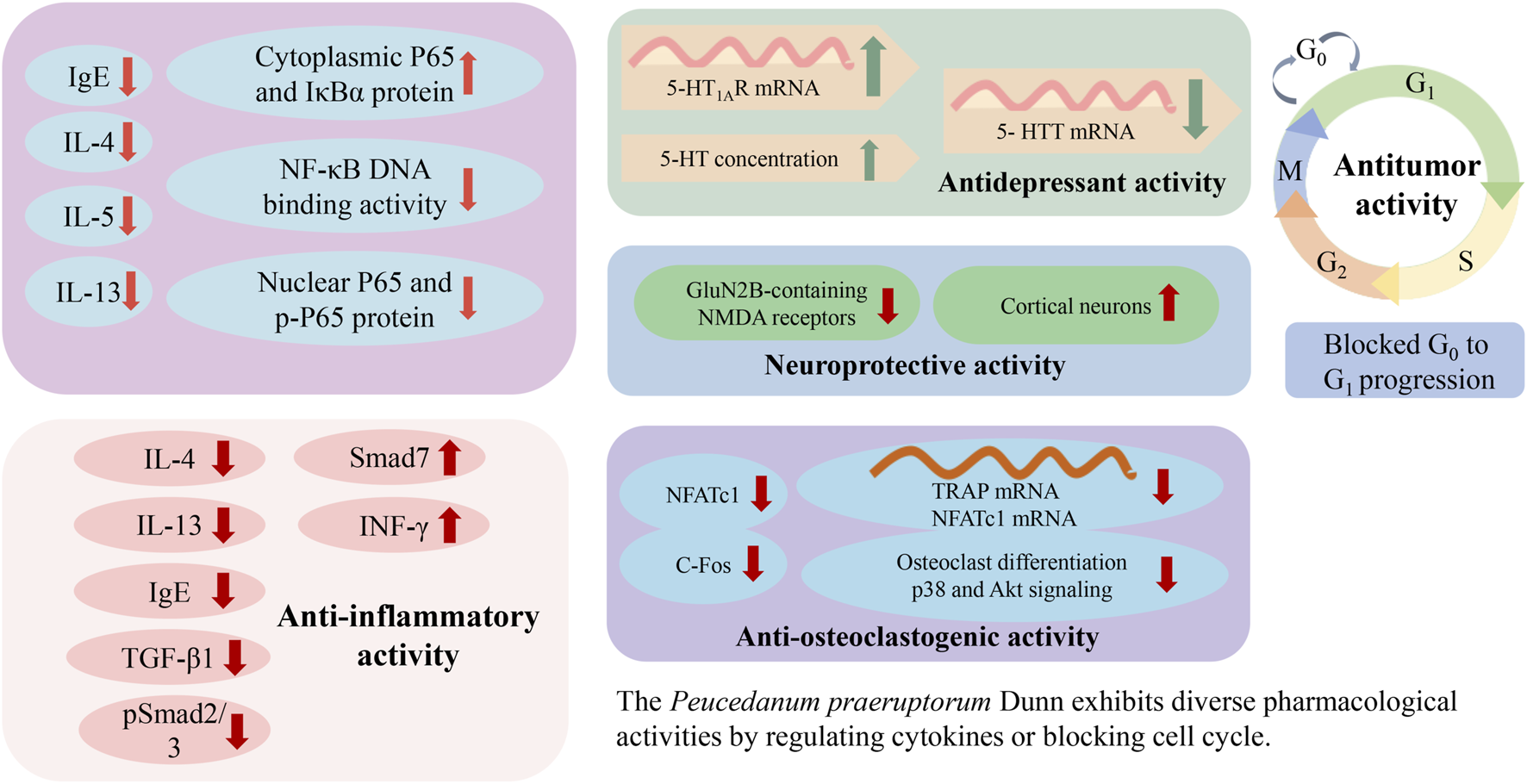
Possible mechanisms associated with the pharmacology of Peucedanum praeruptorum Dunn.
6.1 Anti-inflammatory activity
Praeruptorin C (pyranocoumarins) was traditionally used as an antibechic and antibronchitic drug. In one study, praeruptorin C treatment was found to regulate excitatory synaptic proteins in the anterior cingulate cortex, attenuate the release of proinflammatory cytokines, and inhibit the activation of microglia (Su et al., 2021). However, further studies need to assess the effects of praeruptorin C in other pain models. In another study, administration of (±)-praeruptorin A (30, 60, 120 mg/kg) to ovalbumin-sensitized BALB/c mice for 56 days increased the level of INF-γ and reduced the expression of IL-13/-4 in bronchoalveolar lavage fluid (BALF); decreased the level of immunoglobulin (Ig) E in serum; and suppressed airway inflammation, hyperresponsiveness, and remodeling. In the study, levels of cytokines in BALF, immunoglobulin (Ig) E in serum as well as expression of TGF-β1 and Smad proteins in lung tissue were measured by enzyme-linked immunosorbent assay, immunohistochemistry or Western blot analysis. (Xiong et al., 2012). Treatment of inflamed human periodontal membrane cells with praeruptorin D (10, 20, 30, 40 μg/mL) inhibited TNF-α and IL-1β expression. According to experimental requirements, the negative control group was healthy cells group and positive control group was minocycline hydrochloride group (Yu, 2022). Administration of praeruptorins C, D, and E (2, 4, 8, 16 μg/mL) to RAW264.7 macrophages stimulated with lipopolysaccharide (LPS) for 24 h inhibited NF-κB and STAT3 activation. Although all three had anti-inflammatory activities, praeruptorins D and E exhibited greater anti-inflammatory activities than praeruptorin C and in vivo pharmacological potencies need to be further evaluated. (Yu et al., 2012). One study demonstrated that the administration of IMP (furanocoumarins) at doses of 5, 10, and 15 μmol/L for 1 h in the RBL-2H3 allergic inflammatory cell model promoted IFN-γ expression; decreased the expression of TNF-α, COX-2, IL-6, IL-4, and IL-3; and inhibited RBL-2H3 cell degranulation. These results indicate that IMP is effective for inhibiting the inflammatory response in the RBL-2H3 allergic inflammatory cell model mediated by IgE immunoregulation (Long et al., 2019). However, the authors did not evaluate the effectiveness of IMP using animal models. Moreover, P. praeruptorum polysaccharide treatments ranging from 25 to 200 μg/mL increased the expression of costimulatory and accessory factors, increased the secretion of chemokines and inflammatory factors, and enhanced phagocytosis and pinocytosis. In this way, P. praeruptorum polysaccharides modulated the inflammatory response of macrophages via the NF-κB and TLR2/TLR4-dependent MAPK pathways (Zhao et al., 2022). Another study demonstrated that Dl-praeruptorin A reduced inflammation in an LPS-induced acute lung injury mouse model, specifically inhibiting endothelial inflammation (Wang et al., 2012; Zhou et al., 2016). Nonetheless, the mechanism of Dl-praeruptorin A against acute lung injury needs further study prior to its use in clinical treatment.
6.2 Expectorant and antitussive effects
P. praeruptorum water extract treatment (45 g/kg) was found to resolve phlegm (Liu et al., 1997; Meng et al., 1997). The administration of praeruptorin C and nodakenin (10 mg/kg) to mice, with phenolsulfonphthalein as an expectorant indicator, increased phenolsulfonphthalein excretion in tracheal tissues. Both praeruptorin C and nodakenin showed expectorant effects. And ammonium chloride served as a positive control (Liu et al., 2009). However, phenolsulfonphthalein is mainly excreted from the urine by the kidneys, and drugs that affect renal function are prone to false positive results. Similarly, treating BALB/c mice for 5 days with nodakenin (10 mg/kg) promoted the DNA binding activity of NF-κB, increased the levels of IκBα and P65 in the cytoplasm, decreased the expression of p-P65 and P65 in the nucleus, reduced the level of IgE in serum and IL-13/-5/-4 in BALF, and suppressed airway hyperreactivity and inflammation (Xiong et al., 2014). Notably, the pharmacological properties of P. praeruptorum before and after honey roasting were found to be different. The 5.0 and 10.0 g/kg doses of honey-roasted products showed stronger expectorant and antitussive effects than raw products. However, the 2.5 g/kg dose of honey-roasted and raw products was more effective as relieving asthma (Zhang et al., 2010b). Although P. praeruptorum has long been used to resolve phlegm, descend Qi, clear heat, and dissipate wind in TCM (Zhao et al., 2022), comprehensive studies of its constitutive bioactive monomers and their molecular mechanisms, as well as clinical trials, are necessary to improve its expectorant and antitussive activities with minimal side effects.
6.3 Antitumor activity
More than 85% of all kidney cancers worldwide are characterized as renal cell carcinoma (RCC). In one study, treating RCC cells for 24 h with praeruptorin B (0–30 μmol/L) inhibited both migrability and invasibility, as well as downregulated the expression of cathepsins V and C in ACHN and 786-O cells (Lin et al., 2020). Similarly, praeruptorin B (20, 40, 60 μmol/L) was reported to inhibit both the proliferation and migration of SK-OV-3 ovarian cancer cells, as well as downregulate the expression of FASN, c-Myc, SREBP-1c, and cyclin D1. The likely mechanism is that the SREBP-1c/FASN signaling pathway regulates the energy metabolism of SK-OV-3 ovarian cancer cells, thus inhibiting their proliferation. Real-time fluorescence quantitative polymerase chain reaction (RT-qPCR) was used to detect the mRNA expressions of proliferating genes such as c-Myc and CyclinD1 and the mRNA expressions of key genes of energy metabolism such as SREBP-1c and FASN in tumor cells and Western blot was used to detect the expressions of SREBP-1c, FASN proteins (Xue et al., 2021).
A 48 h treatment with praeruptorin A (25 μmol/L) combined with taxol (125 nmol/L) induced apoptosis in A2780/TAX ovarian cancer cells, and inhibited their migration by downregulating MMP9 and MMP2 expression. However, it remains to be seen whether praeruptorin A can enhance the chemotherapeutic efficacy of taxol, or whether it retains its curative effect against ovarian cancer in vivo (Chen et al., 2022). Another study on HeLa and SiHa cell lines reported that praeruptorin A could increase the levels of tissue inhibitors of metalloproteinase-2 and decrease the expression of matrix metalloproteinase-2; downregulate S-phase kinase-associated protein 2 and cyclin D1; upregulate p27, p21, p16, and Rb; and induce G0/G1 phase cell cycle arrest. However, praeruptorin A could not inhibit cell viability in IgG-treated cells, the effect of IgG interference with praeruptorin A in HeLa cells (Wu et al., 2017). Additionally, praeruptorin A has been found effective at inhibiting the migrability and invasibility of hepatocellular carcinoma (HCC) cells by activating extracellular signal-regulated kinase signaling and inhibiting matrix metalloproteinase-1 expression. However, in vivo metastasis animal model is even worthier for further investigation to examine the antimetastatic effect and safety evaluation of praeruptorin A (Yu et al., 2021). In LS174T cells, praeruptorin A (2.5, 10, 40 μmol/L) could significantly upregulate cytochrome P450 3A4 levels and activity via a pregnane X receptor-mediated pathway. However, siRNA knockdown of the pregnane X receptor resulted in suppressed expression of cytochrome P450 3a11 in mouse primary hepatocytes (Huang et al., 2013).
Administration of praeruptorins A and B to SGC7901 human gastric cancer cells for 24 h produced cytotoxic effects in SGC7901 cells, resulting in antiproliferation. praeruptorin A could also enhance the action of doxorubicin on SGC7901 cells (Liang et al., 2010). Administration of praeruptorins A and C (10, 25, 50 μmol/L) to HepG2 cells in vitro for 24 or 48 h increased the expression of multidrug resistance-associated protein 2 by way of the constitutive androstane receptor-mediated pathway (Zhou et al., 2013). In H1299, PC9, H1975, and PC9/ER human non-small-cell lung cancer (NSCLC) cell lines, praeruptorin A and pteryxin restricted the HGF-induced phosphorylation of MET in PC9/ER and H1975 cells, increased PARP cleavage in H1975 cells and the proportion of annexin V-positive cells, and overall induced apoptosis and reduced cell viability. However, praeruptorin A and pteryxin could not inhibit HGF-induced AKT phosphorylation and prompted apoptosis in NSCLC cells regardless of EGFR TKI resistance or epidermal growth factor receptor (EGFR) mutation status (Park et al., 2022).
In another study, 24 h of treatment with the angular pyranocoumarin (±)-4′-O-acetyl-3′-O-angeloyl-cis-khellactone (0, 10, 20, 30, 40 μg/mL) was found to promote apoptosis in U266 cells, thereby constraining proliferation. The most likely mechanism involved the upregulation of caspase-3/-8 expression and the downregulation of hTERT, p-AKT, and pERK (Yu et al., 2015). Neopeucedalactone, a pyranocoumarin isolated from P. praeruptorum roots, was found to inhibit the growth of human leukemic HL-60, prostate cancer PC-3, and THP-1 cell lines in vitro (Li et al., 2020).
6.4 Neuroprotective activity
One study demonstrated that 24 h of treatment with praeruptorin C (0, 1, and 10 μmol/L) could partially reverse the upregulated expression of GluN2B-containing N-methyl-D-aspartate receptors, inhibit neuronal apoptosis, and balance Bax and Bcl-2 expression. Although it was suggested that praeruptorin C exerted its neuroprotective effects by reversing intracellular Ca2+ overload, it is possible other pathways or mechanisms were involved (Yang et al., 2013). In another study, praeruptorin C (1.5, 3.0 mg/kg) was found to alleviate depressive behavior, motor deficit, and neuronal excitotoxicity in 3-nitropropionic acid (3-NP)-treated mice via upregulating the expression of HTT, DARPP32, and BDNF in striatum tissue. Motor behavior was tested using the open field test and rotarod test, while psychiatric symptoms were tested using the forced swimming test and tail suspension test. We suggest that, based on these findings, praeruptorin C may prove therapeutic for cognitive, psychiatric, and movement disorders associated with Huntington’s disease (Wang et al., 2017).
6.5 Anti-osteoclastogenic activity
Osteoporosis results in an elevated risk of fracture, compromised bone strength due to low bone density, and metabolic defects. Bone homeostasis depends on the resorption of bone by osteoclasts and formation of bone by osteoblasts. Imbalance of this tightly coupled process can cause diseases such as osteoporosis (Chen et al., 2018). In one study, exposure of RAW264.7 cells for 4 h to praeruptorin C (0, 20 μmol/L) reduced osteoclast formation by obstructing the JNK and NF-κB pathways, without disturbing the p38 and ERK pathways. In ovariectomized (OVX) mice, a model for post-menopausal bone loss, praeruptorin C was found to increase bone mass and decrease osteoclast activity. The antiresorptive properties of praeruptorin C suggest that it may be an effective treatment for osteoporosis, although further research is required (Liu et al., 2017). In bone marrow-derived macrophages, praeruptorin A (10 μmol/L) treatment for 30 min inhibited RANKL-stimulated osteoclast differentiation and p38 and Akt signaling (Yeon, Jeong-Tae, et al., 2014). Praeruptorin D could promote the osteogenic differentiation and proliferation of inflammatory periodontal membrane cells, and upregulate osteogenic gene (RUNX-2, ALP, and OCN) expression at doses of 10, 20, 30, and 40 μg/mL by using RT-qPCR and Alizarin red S staining (Yu, 2022).
6.6 Antidepressant activity
Clinical depression is characterized by sustained depressive mood and cognitive dysfunction, including restlessness, anhedonia, sleep disorders, guilt, and repeated thoughts of suicide (Liu et al., 2011). The occurrence of depression is closely related to damage to hippocampal neurons. Chronic stress can damage the hippocampus, resulting in atrophy, apoptosis, or reduced regeneration of hippocampal neurons (Wang and Xu, 2014). Dl-praeruptorin A can protect the nervous and cardio-cerebrovascular systems. For example, 28 days of treatment with Dl-praeruptorin A (10, 30, 60 mg/kg) improved the synaptic ultrastructure of hippocampal CA1 region and increased neurotrophic factors and nerve growth factors in the hippocampus (Wang and Xu, 2014). In addition, 28 days of treatment with IMP (15, 30 mg/kg) significantly enhanced 5-HT1AR expression, 5-HT level, and sucrose preference; increased the incidence of grooming, rearing, and crossing behaviors; reduced immobility; and decreased 5-HTT expression. These results indicate that IMP exhibits antidepressant effects in rats, likely due to changes in the concentration of 5-HT and 5-HTT, and in the expression of 5-HT1AR, in the hippocampus and prefrontal cortex (Zheng et al., 2019). However, the mechanism responsible for the antidepressant activity of P. praeruptorum extracts remains unknown, and clinical pharmacological experiments are lacking.
6.7 Other activities
P. praeruptorum has been shown to exhibit other therapeutic activities, including ventricular remodeling, inhibiting hepatic microsomal drug-metabolizing enzymes, ameliorating memory disruption, and alleviating ischemia/reperfusion injury. Administration of P. praeruptorum alcohol extracts to rats for 28 days affected ventricular remodeling and apoptosis-related proteins in different ways, and had a positive influence on ventricular remodeling (Tu et al., 2004). Treatment of mice with total coumarins (50, 100, 200 mg/kg) prolonged the hypnotic duration of pentobarbital sodium in a dose-dependent manner, inhibited the activities of aniline hydroxylase and aminopyrine N-demethylase, minimally influenced the hypnotic effect of barbital sodium, and inhibited the activity of hepatic microsomal drug-metabolizing enzymes (Wang et al., 2004). In addition, administration of nodakenin (10 mg/kg) reduced scopolamine-induced cognitive impairments associated with the Y-maze test and passive avoidance test, as well as minimized escape latency in the Morris water maze test. Nodakenin has been shown to block acetylcholinesterase activity in a dose-dependent manner in vitro (Kim et al., 2007). Administration of Dl-praeruptorin A (0.5, 1.0, 2.0 mg/kg) reduced the levels of bcl-2, bax, Fas, and IL-6, and raised the bcl-2/bax ratio, under hypotension without bradycardia. A positive, linear correlation has been demonstrated between bax, Fas, bcl-2, and IL-6, where neutrophil infiltration was minimal. Dl-praeruptorin A was also found to prevent postischemic cell death in rat heart, likely due to the automodulation of immediate-early gene expression of bax, Fas, bcl-2, and IL-6 during myocardial ischemia/reperfusion (Chang et al., 2003). In rats, praeruptorin C (5, 15, 30 mg/kg) was found to reduce ischemia/reperfusion injury induced by coronary ligation, most likely by reducing oxygen free radicals (Liu and Wang, 2009).
7 Pharmacokinetic studies
Praeruptorins A, B, and C, the primary metabolites of P. praeruptorum, exhibit diverse biological activities, including neuroprotective, antitumor, anti-inflammatory, immunoregulatory, anti-osteoclastogenic, and antidepressant effects. Recently, researchers developed a liquid chromatography–selected ion monitoring–mass spectrometry (LC–SIM–MS) method to conduct a pharmacokinetic study of WaiGan KeSou Formula decoction administered to rats at a dose of 20 mL. The monarch drug of WaiGan KeSou Formula was Qianhu, and its index compound was Praeruptorin A. Within 24 h, the peak concentration (Cmax) of praeruptorin A in plasma was 172.697 ± 17.254 ng/mL, the peak time was 1.50 h, the elimination half-life (t1/2) was 1.02 h, the mean retention time was 3.42 h, the AUC0–τ (area under the curve) was 504.866 ± 50.317 h ng/mL, and the AUC0–∞ was 514.401 ± 36.950 h ng/mL (He and Wu, 2021). In addition, LC-MS/MS was utilized to evaluate the plasma concentrations of praeruptorin A in rats after a single intragastric dose of 8 g/kg body weight. The researchers found that praeruptorin A was detectable up to 24 h after administration, with an AUC0–t of 311.80 ± 42.38 ng h/mL, a Cmax of 31.09 ± 4.84 ng/mL, and a t1/2 of 7.52 ± 1.00 h (Zhou et al., 2015).
A sensitive, selective, and rapid online solid phase extraction-chiral LC–MS/MS method was developed to conduct a pharmacokinetic study of praeruptorins B and C after orally administering P. praeruptorum extract to rats. Praeruptorins B and C were detectable in rat plasma up to 24 h after administration, with AUC0–t values of 187.29 ± 15.02 (B) and 91.64 ± 9.37 h ng/mL (C), Cmax values of 19.66 ± 4.25 (B) and 7.59 ± 1.98 ng/mL (C), and t1/2 values of 8.20 ± 1.21 h (B) and 14.97 ± 3.66 h (C) (Zhou et al., 2015). Nonetheless, additional pharmacokinetic studies should be conducted on the other bioactive metabolites present in P. praeruptorum, including praeruptorin D, praeruptorin E, qianhucoumarin B, and praeroside I.
8 Quality control
Qianhu is typically processed by washing and immediately drying at low temperature, which must be kept below 60°C. Fresh slices should be thicker than 6 mm (Ren et al., 2021). To maintain medicinal quality, the Chinese Pharmacopoeia dictates the use of microscopic, morphological, HPLC, and TLC detection and identification, as well as ethanol extraction and cold-dipping. By utilizing cold-dipping, the ethanol extract must be more than 20.0% for P. praeruptorum. According to the requirements of the Chinese Pharmacopoeia (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2020), the moisture content (after drying) must not exceed 12.0% and the ash content must not exceed 8.0%. Moreover, different extraction methods have different effects on the index metabolites of P. praeruptorum (praeruptorins A and B). The reflux method is favored for the extraction of praeruptorin A, producing a much higher praeruptorin A content than ultrasonic methods. Conversely, the ultrasonic method is favored for the extraction of praeruptorin B, producing a much higher praeruptorin B content than the reflux method (Xu et al., 2022). However, it is inadvisable to utilize only one crude, quantitative marker when assessing the quality of P. praeruptorum extracts. An array of bioactive metabolites has been detected in P. praeruptorum by HPLC, UV, gas chromatography (GC), NMR, high-speed counter-current chromatography coupled with electrospray ionization multi-stage MS (prep-HSCCC/ESI-MS(n)) (Zhou et al., 2015).
The medicinal quality of P. praeruptorum is affected by the altitude at which it is produced. According to reports by Luo et al. (2022), an altitude of 900 m improved the praeruptorin A content, while an altitude of 650 m improved the praeruptorin B content. Moreover, the influence of altitude was greater on praeruptorin B content than on praeruptorin A content (Luo et al., 2022). In another study, praeruptorin A and B contents were higher in plants cultivated at high altitudes than in plants cultivated at low altitudes by using HPLC-DAD method to determine the contents of praeruptorin A and B in the 24 batches of Qianhu from different producing areas. According to the experimental results, cluster analysis and principal component analysis were carried out (Yang et al., 2021). In addition, the key climatic factors affecting the praeruptorin content are average relative humidity, average maximum temperature in July, average annual temperature, and average temperature in July (Xu et al., 2021). However, the relationships between Qianhu quality and climatic factors have not been widely investigated, and warrant further study.
Praeruptorin A and B contents are also influenced by plant organ, harvesting time, cultivation environment, and fertilization strategy. According to reports by Li et al. (2022), the HPLC method was used to determine the content of praeruptorin A and B in the cultured P. praeruptorum at different harvesting periods, as well as to analyze the fluctuation of praeruptorin A and B at different harvesting periods. Praeruptorin A and B contents are highest in P. praeruptorum roots, followed by stems, and are lowest in leaves. In the 14 samples, the praeruptorin A content was found to be much higher in 2-year-old P. praeruptorum than in 1-year-old P. praeruptorum. Moreover, plants cultivated on southern slopes exhibited an approximately 80% higher content of praeruptorin A than plants grown on slopes of other orientations. However, no significant differences in the contents of praeruptorin A or B were observed in P. praeruptorum harvested before or after bolting (Li et al., 2022). Finally, the reasonable application of P and K fertilizers has been found to improve both the yield and medicinal quality P. praeruptorum, although the application of N fertilizer should be controlled (Zhou et al., 2022).
9 Safety
Praeruptorin C exerted no toxicity on primary cultures of mouse neurons at doses of 0 and 10 μg/mol (Yang et al., 2013). Moreover, an emulsion of praeruptorin C did not exert any toxicity or induce any behavioral changes at doses of 5 and 40 μg/mol in OVX mice by performing a CCK-8 assay (Liu et al., 2017). In BMMs, praeruptorin A was found to be atoxic at doses under 10 mmol/L, but significantly cytotoxic at doses over 20 mmol/L (Yeon et al., 2014). In HepG2 cells, praeruptorins A and C were found to be atoxic at doses of 10 and 100 μg/mol, respectively (Zhou et al., 2013). However, a maximum dose of 200 μg/mol dramatically increased cell toxicity. The toxicity of praeruptorin B on SK-OV-3 ovarian cancer cells was not obvious at doses of 20 and 60 μmol/L (Xue et al., 2021). HCC cell line and normal liver THLE-2 cells were treated with different concentrations of praeruptorin A (0, 10, 20, 30, 40 μg/mL) for 24 h. The cell viability was assessed through 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide assay. The results implied that praeruptorin A did not induce cytotoxicity in an HCC cell line at doses of 10 and 40 μg/mL (Yu et al., 2021). In addition, praeruptorin A was atoxic to normal liver THLE-2 cells under at concentrations of 10–30 μg/mol. The Chinese Pharmacopoeia 2020 (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2020) recommends a Qianhu dosage of 3–10 g/d. In addition, Qianhu can safely be used with Pinelliae Rhizoma, but not with Gleditsia sinensis Lam. or Veratrum nigrum L. (“Bencaojing Jizhu” 《本草经集注》). Finally, Qianhu is not considered suitable for people with Yin deficiency syndrome (a series of symptoms caused by the deficiency of yin essence or fluid in the human body. Yin: the dark, not active, female principle of the Universe in Chinese philosophy) (Wen, 2023), cough, cold, or cold fluid syndrome. According to cell studies, P. praeruptorum appears to be atoxic at a low dose but cytotoxic at higher doses (Zhou et al., 2013; Yeon et al., 2014).
10 Conclusion and future perspectives
P. praeruptorum is a classical medicinal plant commonly used in TCM preparations. Here, we systematically evaluated the toxicology, molecular mechanisms, pharmacology, phytochemistry, botany, quality control, and traditional uses of P. praeruptorum in order to validate the medicinal use of this species. According to the Chinese Pharmacopoeia and classical Chinese botanical drugs, P. praeruptorum has been historically prescribed to treat a wide spectrum of diseases including cough, asthma, and pulmonary hypertension. Pharmacological studies suggest that P. praeruptorum exhibits anti-inflammatory, expectorant, antitussive, antitumor, neuroprotective, anti-osteoclastogenic, and antidepressant effects, and largely support the traditional uses of this plant. To date, more than 119 distinct phytochemicals have been identified in P. praeruptorum extracts, the most common of which are pyranocoumarins and furanocoumarins.
Although there has been considerable progress in evaluating the phytochemistry and pharmacology of P. praeruptorum, there are still gaps in our knowledge. First, according to the Chinese Pharmacopoeia 2020 (Chinese Pharmacopoeia Committee of People’s Repulic of China, 2020), Qianhu can disperse wind-heat, reduce cough and phlegm, and dissipate adverse Qi, indicating that Qianhu may alleviate the effects of wind-heat on the lungs (Song et al., 2015). However, most of the active metabolites of P. praeruptorum effective against cough and wind-heat are currently administered as crude extracts. Therefore, comprehensive investigations should be conducted to identify the effective metabolites and elucidate their modes of action in order to facilitate clinical trials. Second, we found few reports on the toxicity of P. praeruptorum extracts or potential botanical drugs interactions. The potential adverse effects, contraindications, and toxicities of P. praeruptorum extracts and their bioactive metabolites should therefore be studied in vitro, in vivo, and in clinical trials. Third, the majority of the reviewed research was conducted in cell cultures or animal models. Clinical trials in humans will be required to truly evaluate the efficacy P. praeruptorum in addressing depression, osteoporosis, cancer, and inflammation, among other diseases. Forth, most of the P. praeruptorum containing health products are mainly derived from its root rich in chemical compounds, while non-medicinal parts are rarely exploited. Therefore, it may be interesting to extend the research to the non-medicinal parts of the inexpensive flowers, leaves, and stems of P. praeruptorum to ensure the fully utilization of its edible and medicinal values. Finally, new and updated analytical and quality control methods will be required to identify novel markers of quality for the assessment of TCM preparations.
In conclusion, P. praeruptorum is rich in medicinal materials, and its pharmacological effects are extensive. With the advantages of modern instruments and data analysis technology in identifying chemical components and separation, Qianhu medicinal materials can be better developed and new drug discovery (Liu, 2020). Future research should be conducted to investigate the mode of action responsible for the pharmacological activities of P. praeruptorum extracts, as well as to comprehensively evaluate the potential toxicities, adverse effects, and contraindications of this botanical drugs. Alongside updated quality control measures, these investigations will facilitate clinical trials. Updated in vivo pharmacological studies must be performed to validate the traditional uses of P. praeruptorum. Finally, the clinical safety and efficacy of P. praeruptorum-derived phytochemicals in the treatment of depression, osteoporosis, cancer, and other diseases, require validation.
Statements
Author contributions
QW: Writing–review and editing, Writing–original draft, Visualization, Investigation, Data curation. QS: Writing–review and editing. QH: Writing–review and editing. LQ: Writing–review and editing. BZ: Writing–review and editing, Supervision.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study was supported by the Talent Projects of Zhejiang Chinese Medical University (2021ZR09), Young Innovative Talents Project of Zhejiang Medical Health Science and Technology (2022RC052), Natural Science Foundation of Zhejiang Province (LQ21H280003), National Natural Science Foundation of China (82003896), Ningbo Natural Science Foundation (202003N4334).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
BALF, bronchoalveolar lavage fluid; LPS, lipopolysaccharide; CFA, complete Freund’s adjuvant; ER, erlotinib-resistant; OVX, ovariectomized; HCC, hepatocellular carcinoma; HR-ESI-MS, high-resolution-electrospray ionization-mass spectrometry; Ig, immunoglobulin; IL, interleukin; IMP, imperatorin; IR, infrared spectroscopy; LC–MS/MS, liquid chromatography-mass spectrometry/-mass spectrometry; LC–SIM–MS, liquid chromatography-selected ion monitoring-mass spectrometry; MS, mass spectrometry; NMR, nuclear magnetic resonance; NO, nitric oxide; NSCLC, epidermal growth factor receptor; NSCLC, non-small-cell lung cancer; RCC, renal cell carcinoma; RT-qPCR, real-time fluorescence quantitative polymerase chain reaction; SIMS, secondary ion mass spectroscopy; TCM, traditional Chinese medicine; TNF-α, tumor necrosis factor αtumor necrosis factor α; UV, ultraviolet.
References
1
Asahara T. Sakakibara I. Okuyama T. Shibata S. (1984). Studies on coumarins of a Chinese drug “Qian-Hu” V. Coumarin-Glycosides from “Zi-Hua qian-hu”1. Planta Med.50 (6), 488–492. 10.1055/s-2007-969780
2
Chang H. Okada Y. Okuyama T. Tu P. (2007). 1H and 13C NMR assignments for two new angular furanocoumarin glycosides from Peucedanum praeruptorum. Magn. Reson. Chem.45 (7), 611–614. 10.1002/mrc.2005
3
Chang H. T. (1998). Studies on the components from the root of Peucedanum praeruptorum in reducing pulmonary hypertension. Shenyang: Master, Shenyang Pharmaceutical University.
4
Chang H. T. Li X. (1999a). Coumarins from peucedanum praeruptorum dunn. J. Shenyang Pharm. Univ.16 (02), 28–31. 10.3969/j.issn.1006-2858.1999.02.007
5
Chang H. T. Li X. (1999b). Studies on chemical constituents from roots of Peucedanum praeruptorum Ⅴ. Chin. Tradit. Herb. Drugs30 (06), 414–416. 10.3321/j.issn:0253-2670.1999.06.007
6
Chang H. T. Okada Y. Ma T. J. Okuyama T. Tu P. F. (2008). Two new coumarin glycosides from Peucedanum praeruptorum. J. Asian Nat. Prod. Res.10 (5-6), 577–581. 10.1080/10286020801966740
7
Chang T. H. Liu X. Y. Zhang X. H. Xing J. Wang H. L. (2003). Effects of Peucedanum Praeruptorum Dunn and dl-praeruptorin A on IL-6 level and Fas, bax, bcl-2 protein expressions in ischemia-reperfusion myocardium of rats. J. China Med. Univ.32 (01), 5–7+10. 10.3969/j.issn.0258-4646.2003.01.001
8
Chen F.-Y. Tu L.-F. Liu D.-P. Ma J. Luo Y.-M. (2021). Chemical constituents from the roots of Peucedanum praeruptorum Dunn and their chemotaxonomic significance. Biochem. Syst. Ecol.99 (2021), 104355. 10.1016/j.bse.2021.104355
9
Chen L. L. Chu S. S. Zhang L. Xie J. Dai M. Wu X. et al (2019). Tissue-specific metabolite profiling on the different parts of bolting and unbolting peucedanum praeruptorum dunn (Qianhu) by laser microdissection combined with UPLC-Q/TOF(-)MS and HPLC(-)DAD. Molecules24 (7), 1439. 10.3390/molecules24071439
10
Chen Q. H. Liu X. L. Pan L. X. OuYang J. M. (2022). Study on the effects of praeruptorin A combined with low concentration of taxol on taxol-resistant ovarian cancer cell line A2780/TAX. Chin. J. Hosp. Pharm.42 (17), 1755–1759+1765. 10.13286/j.1001-5213.2022.17.03
11
Chen X. Wang Z. Duan N. Zhu G. Schwarz E. M. Xie C. (2018). Osteoblast-osteoclast interactions. Connect. Tissue Res.59 (2), 99–107. 10.1080/03008207.2017.1290085
12
Chen Z. X. Huang B. S. She Q. L. Zeng G. F. (1979). The chemical constituents of Bai-Hua-Qian-Hu, the root of Peucedanum praeruptorum Dunn. (Umbelliferae)--four new coumarins (author's transl). Acta Pharm. Sin.14 (08), 486–496. 10.16438/j.0513-4870.1979.08.007
13
Cheong C. T. Kim W. C. Jin M. H. Kim H. J. Kang S. J. Kang S. H. et al (2002). Inhibitors of melanogenesis from the roots of peucedanum praeruptorum. Kor. J. Pharmacogn.33 (4), 395–398.
14
Chinese Pharmacopoeia Committee of People's Repulic of China (2000). Chinese Pharmacopoeia Ⅰ. Beijing: China Medical Science Press.
15
Chinese Pharmacopoeia Committee of People's Repulic of China (2005). Chinese Pharmacopoeia Ⅰ. Beijing: China Medical Science Press.
16
Chinese Pharmacopoeia Committee of People's Repulic of China (2010). Chinese Pharmacopoeia Ⅰ. Beijing: China Medical Science Press.
17
Chinese Pharmacopoeia Committee of People's Repulic of China (2015). Chinese Pharmacopoeia Ⅰ. Beijing: China Medical Science Press.
18
Chinese Pharmacopoeia Committee of People's Repulic of China (2020). Chinese Pharmacopoeia Ⅰ. Beijing: China Medical Science Press.
19
Editorial Committee of Flora of China (1992). Flora of China. Beijing: Chinese Academy of Sciences, Science Press.
20
He D. M. Wu F. H. Kong L. Y. (2007). Review on pharmacological action of Peucedanum praeruptorum. Pharm. Clin. Res.15 (03), 167–170. 10.13664/j.cnki.pcr.2007.03.001
21
He G. X. Wu S. H. (2021). Rat plasma pharmacokinetic research of praeruptorin A in waigan kesou formula. Strait Pharm. J.33 (08), 19–22. 10.3969/j.issn.1006-3765.2021.08.008
22
Huang L. Bi H. C. Li Y. H. Zhang J. Q. Kuang S. Y. Zhang L. et al (2013). Regulation of human pregnane X receptor and its target gene cytochrome P450 3A by praeruptorin A isolated from the herbal medicine Peucedanum praeruptorum. Planta Med.79 (16), 1509–1515. 10.1055/s-0033-1350795
23
Ishii H. Okada Y. Baba M. Okuyama T. (2008). Studies of coumarins from the Chinese drug Qianhu, XXVII: structure of a new simple coumarin glycoside from Bai-Hua Qianhu, Peucedanum praeruptorum. Chem. Pharm. Bull. (Tokyo)56 (9), 1349–1351. 10.1248/cpb.56.1349
24
Jian X. Zhao Y. Wang Z. Li S. Li L. Luo J. et al (2020). Two CYP71AJ enzymes function as psoralen synthase and angelicin synthase in the biosynthesis of furanocoumarins in Peucedanum praeruptorum Dunn. Plant Mol. Biol.104 (3), 327–337. 10.1007/s11103-020-01045-4
25
Jong T. T. Hwang H. C. Jean M. Y. Wu T. S. Teng C. M. (1992). An antiplatelet aggregation principle and X-ray structural analysis of cis-khellactone diester from Peucedanum japonicum. J. Nat. Prod.55 (10), 1396–1401. 10.1021/np50088a002
26
Kim D. H. Kim D. Y. Kim Y. C. Jung J. W. Lee S. Yoon B. H. et al (2007). Nodakenin, a coumarin compound, ameliorates scopolamine-induced memory disruption in mice. Life Sci.80 (21), 1944–1950. 10.1016/j.lfs.2007.02.023
27
Kong L. Y. Li X. Pei Y. H. Yu R. M. Min Z. D. Zhu T. R. (1994a). Isolation and structure elucidation of Baihuaqianhuside and Pd-C-I from Peucedanum praeruptorum. Acta Pharm. Sin.29 (04), 276–280. 10.16438/j.0513-4870.1994.04.007
28
Kong L. Y. Li X. Pei Y. H. Zhu T. R. (1994b). Isolation and structural elucidation of qianhucoumarin D and qianhucoumarin E from peucedanum praeruptorum. Acta Pharm. Sin.29 (01), 49–54. 10.16438/j.0513-4870.1994.01.010
29
Kong L. Y. Li Y. Min Z. D. Li X. Zhu T. R. (1996). Coumarins from Peucedanum praeruptorum. Phytochemistry41 (5), 1423–1426. 10.1016/0031-9422(95)00783-0
30
Kong L. Y. Pei Y. H. Li X. Zhu T. R. (1993a). On the chemical constituents of the root of Peucedanum praeruptorum. Chin.Tradit. Herb. Drugs24 (08), 401–404+446.
31
Kong L. Y. Pei Y. H. Li X. Zhu T. R. Okuyama T. (1993b). Isolation and structure elucidation of qianhucoumarin A. Acta Pharm. Sin.28 (06), 432–436. 10.16438/j.0513-4870.1993.06.007
32
Lee J. Lee Y. J. Kim J. Bang O. S. (2015). Pyranocoumarins from root extracts of peucedanum praeruptorum dunn with multidrug resistance reversal and anti-inflammatory activities. Molecules20 (12), 20967–20978. 10.3390/molecules201219738
33
Li X. L. Song C. Jia B. Liu L. Ou J. M. Han B. X. (2022). Analysis of factors affecting the content of praeruptorin A and B in Peucedanum praeruptorum Dunn. Chin. Wild Plant Resour.41 (01), 26–32. 10.3969/j.issn.1006-9690.2022.01.005
34
Li X. Y. Zu Y. Y. Ning W. Tang M. X. Gong C. Niu S. L. et al (2020). A new xanthyletin-type coumarin from the roots of Peucedanum praeruptorum. J. Asian Nat. Prod. Res.22 (3), 287–294. 10.1080/10286020.2018.1551887
35
Liang T. Yue W. Li Q. (2010). Chemopreventive effects of Peucedanum praeruptorum DUNN and its major constituents on SGC7901 gastric cancer cells. Molecules15 (11), 8060–8071. 10.3390/molecules15118060
36
Lin C. L. Hung T. W. Ying T. H. Lin C. J. Hsieh Y. H. Chen C. M. (2020). Praeruptorin B mitigates the metastatic ability of human renal carcinoma cells through targeting CTSC and CTSV expression. Int. J. Mol. Sci.21 (8), 2919. 10.3390/ijms21082919
37
Liu D. P. (2020). Studies on the chemical constituents of Peucedanum praeruptorum Dunn. Master. Nanchang: Jiangxi University of Traditional Chinese Medicine.
38
Liu J. L. Wang S. S. Li X. F. Lu X. Y. Li H. Hong C. X. (2021). Investigation and analysis on the present situation of Peucedanum praeruptorum resources in its producing area. Mod. Agric. Sci. Technol.2021 (01), 89–92+99. 10.3969/j.issn.1007-5739.2021.01.035
39
Liu J. L. Wang X. Y. Zhang L. L. Fang M. J. Wu Y. L. Wu Z. et al (2014). Two-dimensional countercurrent chromatography×high performance liquid chromatography with heart-cutting and stop-and-go techniques for preparative isolation of coumarin derivatives from Peucedanum praeruptorum Dunn. J. Chromatogr. A1374 (2014), 156–163. 10.1016/j.chroma.2014.11.053
40
Liu J. L. Yuan Y. H. Chen N. H. (2011). Research progress in treatment of depression. Chin. Pharmacol. Bull.27 (09), 1193–1196. 10.3969/j.issn.1001-1978.2011.09.003
41
Liu X. Chin J. F. Qu X. Bi H. Liu Y. Yu Z. et al (2017). The beneficial effect of praeruptorin C on osteoporotic bone in ovariectomized mice via suppression of osteoclast formation and bone resorption. Front. Pharmacol.8, 627. 10.3389/fphar.2017.00627
42
Liu X. Y. Wang G. X. (2009). Effects of praeruptorin C in rats with myocardial ischemia reperfusion injury. Chin. J. Cardiovasc. Res.7 (2), 146–148. 10.3969/j.issn.1672-5301.2009.02.024
43
Liu Y. Li X. Y. Song Z. Z. Wei H. Y. (2009). Expectorant action of Praeruptofin C and nodakenin. Lishizhen Med. Mater. Med. Res.20 (05), 1049. 10.3969/j.issn.1008-0805.2009.05.012
44
Liu Y. Wei H. Y. Yao S. H. Zheng X. Z. (1997). Comparison of the expectorant pharmacological effects of Peucedanum. Guid. J. Tradit. Chin. Med.3 (01), 41–43.
45
Long T. Song P. Liang P. Y. Ou S. J. Wang S. X. (2019). Immunomodulatory effect of imperatorin on RBL-2H3 allergic inflammatory cell model induced by IgE. J. Guangzhou Univ. Tradit. Chin. Med.36 (12), 2001–2006. 10.13359/j.cnki.gzxbtcm.2019.12.026
46
Lou H. X. Sun L. R. Yu W. T. Fan P. H. Cui L. Gao Y. H. et al (2004). Absolute configuration determination of angular dihydrocoumarins from Peucedanum praeruptorum. J. Asian Nat. Prod. Res.6 (3), 177–184. 10.1080/10286020310001653255
47
Luo M. Xu G. Tang Q. S. Guo Y. Deng C. F. Luo S. et al (2022). Response of medicinal quality to altitude gradient in Peucedanum praeruptorum Dunn. Chin. Wild Plant Resour.41 (11), 20–24. 10.3969/j.issn.1006-9690.2022.11.004
48
Meng X. L. Jia M. R. Zhang Y. Wu P. Tang M. C. Tang S. W. et al (1997). Pharmacological studies of variet of Qian-hu. Pharmacol. Clin. Chin. Mater. Med.13 (01), 36–39.
49
Okuyama T. Shibata S. (1981). Studies on coumarins of a Chinese drug “Qian-Hu”. Planta Med.42 (1), 89–96. 10.1055/s-2007-971551
50
Okuyama T. Takata M. Shibata S. (1989). Structures of linear furano- and simple-coumarin glycosides of Bai-Hua Qian-Hu. Planta Med.55 (1), 64–67. 10.1055/s-2006-961828
51
Park H. J. Jeong J. H. Park S. H. (2022). The root extract of peucedanum praeruptorum dunn exerts anticancer effects in human non-small-cell lung cancer cells with different EGFR mutation statuses by suppressing MET activity. Molecules27 (7), 2360. 10.3390/molecules27072360
52
Ren J. J. Sun J. Yu X. P. Shen X. X. Wang Z. A. (2021). Effects of different habitat processing methods on quality of Peucedani Radix. Mod. Chin. Med.23 (05), 844–848. 10.13313/j.issn.1673-4890.20200526009
53
Song Y. Jing W. Yan R. Wang Y. (2015). Research progress of the studies on the roots of Peucedanum praeruptorum dunn (Peucedani radix). Pak. J. Pharm. Sci.28 (1), 71–81.
54
Song Z. Q. Li B. Tian K. Y. Hong L. Wu W. Zhang H. Y. (2022). Research progress on chemical constituents and pharmacological activities of peucedani radix and peucedani decursivi radix. Chin. Tradit. Herb. Drugs53 (03), 948–964. 10.7501/j.issn.0253-2670.2022.03.035
55
Su D. J. Li L. F. Wang S. Y. Yang Q. Wu Y. J. Zhao M. G. et al (2021). Pra-C exerts analgesic effect through inhibiting microglial activation in anterior cingulate cortex in complete Freund's adjuvant-induced mouse model. Mol. Pain17, 1744806921990934. 10.1177/1744806921990934
56
Takata M. Okuyama T. Shibata S. (1988). Studies on coumarins of a Chinese drug, “qian-hu”; VIII. Structures of new coumarin-glycosides of “Bai-hua qian-hu”. Planta Med.54 (4), 323–327. 10.1055/s-2006-962446
57
Takata M. Shibata S. Okuyama T. (1990). Structures of angular pyranocoumarins of Bai-hua qian-hu, the root of peucedanum praeruptorum1. Planta Med.56 (3), 307–311. 10.1055/s-2006-960966
58
Tu X. Wang J. M. Tu Q. Tu J. W. (2004). Effects of Peucedanum praeruptorum Dunn extract on left ventricular remodeling and Bcl-2, Bax protein expression in aorta coarctation rats. Chin. J. Clin. Pharmacol. Ther.9 (04), 394–398. 10.3969/j.issn.1009-2501.2004.04.009
59
Wang D. C. Zhao X. M. Li T. D. Ma J. Kong Z. F. Li K. et al (2004). Effect of total coumarins from Peucedanum Praeruptorum Dunn on the activity hepatic drug-metabolizing enzymes in mice. Her. Med.23 (08), 522–524. 10.3870/j.issn.1004-0781.2004.08.002
60
Wang K. Nie Z. X. Sun Y. P. Liu S. Z. Wang G. K. (2018). Current surgical strategies and techniques of aortic valve diseases in children. J. Anhui Univ. Chin. Med.37 (05), 83–90. 10.21037/tp.2018.02.03
61
Wang L. Wang J. Yang L. Zhou S. M. Guan S. Y. Yang L. K. et al (2017). Effect of Praeruptorin C on 3-nitropropionic acid induced Huntington's disease-like symptoms in mice. Biomed. Pharmacother.86 (2017), 81–87. 10.1016/j.biopha.2016.11.111
62
Wang M. (2016). Pharmacological analysis and clinical application of Qianhu. Asia-Pacific Tradit. Med.12 (18), 75–76. 10.11954/ytctyy.201618032
63
Wang X. M. Xu H. H. (2014). Studies on the antidepressant effect and mechanism of Praeruptorin A on CUMS rats. J. Chin. Med. Mater.37 (12), 2259–2262. 10.13863/j.issn1001-4454.2014.12.035
64
Wang Y. Yang C. Chang H. Li G. Jin X. Zou J. (2012). Pd-Ia inhibited inflammation in LPS-induced HUVECs via PPARα. Chin. Pharmacol. Bull.28 (11), 1594–1597. 10.3969/j.issn.1001-1978.2012.11.027
65
Wen H. (2023). Research on intelligent diagnosis method of Yin deficiency syndrome based on multimodal information fusion. Beijing: Beijing University of Chemical Technology. Master.
66
Wu M. H. Lin C. L. Chiou H. L. Yang S. F. Lin C. Y. Liu C. J. et al (2017). Praeruptorin A inhibits human cervical cancer cell growth and invasion by suppressing MMP-2 expression and ERK1/2 signaling. Int. J. Mol. Sci.19 (1), 10. 10.3390/ijms19010010
67
Xiong Y. Y. Shi W. J. Yu H. Zhang X. L. (2014). Inhibitory effects of nodakenin on the airway inflammation and NF-κB signaling pathway in a murine asthmatic model. Basic Clin. Med.34 (05), 690–694. 10.16352/j.issn.1001-6325.2014.05.012
68
Xiong Y. Y. Wang J. S. Wu F. H. Li J. Kong L. Y. (2012). The effects of (±)-Praeruptorin A on airway inflammation, remodeling and transforming growth factor-β1/Smad signaling pathway in a murine model of allergic asthma. Int. Immunopharmacol.14 (4), 392–400. 10.1016/j.intimp.2012.08.019
69
Xu G. Tan Q. S. Yang D. Fu Y. Guo Y. Luo C. et al (2022). Effect of index components in Peucedanum Praerupterum by different extraction methods. Asia-Pacific Tradit. Med.18 (03), 42–44. 10.11954/ytctyy.202203010
70
Xu P. Liang W. Q. Zhang H. J. Hu Y. J. Chen R. B. Pu J. B. (2021). Correlation analysis between active ingredients and climate factors in Radix Peucedanum. China J. Tradit. Chin. Med. Pharm.36 (09), 5614–5618.
71
Xue N. M. Chai M. Y. Yang W. S. Zhou X. C. (2021). Inhibitory effect of praeruptorin B on SK-OV-3 cell proliferation in ovarian cancer by SREBP-1c/FASN signaling pathway. Chin. J. Gerontol.41 (13), 2773–2777. 10.3969/j.issn.1005-9202.2021.13.026
72
Yang L. Kang X. J. Du W. F. Hong H. Ge W. H. Wang L. G. et al (2021). Effect of altitude of producing area on contents of praeruptorin A and praeruptorin B in Qianhu. Chin. Arch. Tradit. Chin. Med.39 (08), 14–18. 10.13193/j.issn.1673-7717.2021.08.004
73
Yang L. Li X. B. Yang Q. Zhang K. Zhang N. Guo Y. Y. et al (2013). The neuroprotective effect of praeruptorin C against NMDA-induced apoptosis through down-regulating of GluN2B-containing NMDA receptors. Toxicol. Vitro27 (2), 908–914. 10.1016/j.tiv.2013.01.001
74
Yeon J. T. Kim K. J. Choi S. W. Moon S. H. Park Y. S. Ryu B. J. et al (2014). Anti-osteoclastogenic activity of praeruptorin A via inhibition of p38/Akt-c-Fos-NFATc1 signaling and PLCγ-independent Ca2+ oscillation. PLoS One9 (2), e88974. 10.1371/journal.pone.0088974
75
Yu C. L. Yu Y. L. Yang S. F. Hsu C. E. Lin C. L. Hsieh Y. H. et al (2021). Praeruptorin A reduces metastasis of human hepatocellular carcinoma cells by targeting ERK/MMP1 signaling pathway. Environ. Toxicol.36 (4), 540–549. 10.1002/tox.23059
76
Yu C. Y. (2022). Study on the effect of PD on the proliferation and osteogenic activity of human inflammatory periodontal membrane cells. Lanzhou: LanZhou University. Master.
77
Yu P. J. Jin H. Zhang J. Y. Wang G. F. Li J. R. Zhu Z. G. et al (2012). Pyranocoumarins isolated from Peucedanum praeruptorum Dunn suppress lipopolysaccharide-induced inflammatory response in murine macrophages through inhibition of NF-κB and STAT3 activation. Inflammation35 (3), 967–977. 10.1007/s10753-011-9400-y
78
Yu Q. H. Ma L. Shen Y. P. Zhai W. Zhou Y. H. (2015). Effect of angular pyranocoumarin isolated from peucedanum praeruptorum on the proliferation and apoptosis of U266 cells. Chin. J. Hematol.36 (11), 937–941. 10.3760/cma.j.issn.0253-2727.2015.10.010
79
Zhang C. Li L. Xiao Y. Q. Li W. Yin X. J. Tian G. F. et al (2010a). A new phenanthraquinone from the roots of Peucedanum praeruptorum. Chin. Chem. Lett.21 (7), 816–817. 10.1016/j.cclet.2010.03.020
80
Zhang C. Li L. Xiao Y. Q. Massahiko T. Kimiye B. (2011). Coumarins from the roots of Peucedanum praeruptorum. Chin. J. Tradit. Chin. Med.26 (09), 1995–1997.
81
Zhang C. Xiao Y. Q. Li L. Massahiko T. Kimiye B. (2012). Chemical constituents from roots of Peucedanum praeruptorum (Ⅴ). China J. Chin. Materia Medica37 (23), 3573–3576. 10.4268/cjcmm20122314
82
Zhang C. Xiao Y. Q. Massahiko T. Kimiye B. (2005). Studies on chemical constituents in roots of Peucedanum praeruptorum (Ⅰ). China J. Chin. Mater. Med.30 (09), 675–677. 10.3321/j.issn:1001-5302.2005.09.009
83
Zhang C. Xiao Y. Q. Massahiko T. Kimiye B. (2006). Studies on chemical constituents from roots of Peucedanum praeruptorum II. China J. Chin. Mater. Med.31 (16), 1333–1335. 10.3321/j.issn:1001-5302.2006.16.008
84
Zhang C. Xiao Y. Q. Massahiko T. Kimiye B. (2009). Studies on chemical constituents from roots of Peucedanum praeruptorum Ⅲ. China J. Chin. Mater. Med.34 (08), 1005–1006. 10.3321/j.issn:1001-5302.2009.08.019
85
Zhang C. Yin X. J. Li L. Li W. Xiao Y. Q. Yu D. R. et al (2010b). Comparative studies on pharmacological effects for processed pieces from peucedanum praeruptorum. Chin. J. Exp. Tradit. Med. Formulae16 (15), 146–148. 10.13422/j.cnki.syfjx.2010.15.053
86
Zhao M. Hou J. Zheng S. Ma X. Fu X. Hu S. et al (2022). Peucedanum praeruptorum Dunn polysaccharides regulate macrophage inflammatory response through TLR2/TLR4-mediated MAPK and NF-κB pathways. Biomed. Pharmacother.152 (2022), 113258. 10.1016/j.biopha.2022.113258
87
Zheng X. X. Liu J. H. Liu M. H. Cheng Y. Cao Y. J. (2019). Antidepressant effect and mechanism of imperatorin. Chin. Pharmacol. Bull.35 (01), 101–105. 10.3969/j.issn.1001-1978.2019.01.020
88
Zhou G. Chen G. Liu H. (2015). Simultaneous quantification of three pyranocoumarins of Peucedanum praeruptorum in rat plasma by liquid chromatography-tandem mass spectrometry: application to pharmacokinetic study. J. Chromatogr. Sci.53 (4), 511–518. 10.1093/chromsci/bmu077
89
Zhou J. R. Sun J. Ren J. J. Shao Q. S. Wang Z. A. (2022). Effects of combined application of N, P, and K in autumn on yield and quality of Peucedanum praeruptorum. Mod. Chin. Med.24 (12), 2443–2449. 10.13313/j.issn.1673-4890.20220222003
90
Zhou J. R. Sun J. Shen X. X. Shao Q. S. Wang L. Y. Wang Z. A. (2021). Study on genetic diversity of peucedanum praeruptorum germplasm resources in traditional production areas. J. Chin. Med. Mater.44 (11), 2543–2548. 10.13863/j.issn1001-4454.2021.11.009
91
Zhou X. Bi H. Jin J. Niu L. Cai D. Deng R. et al (2013). Effects of praeruptorin A and praeruptorin C, a racemate isolated from Peucedanum praeruptorum, on MRP2 through the CAR pathway. Planta Med.79 (17), 1641–1647. 10.1055/s-0033-1350955
92
Zhou X. X. Zhang J. Q. Liu C. X. Chang H. Zou J. Xue M. Q. (2016). Protective effect of Pd-Ia on LPS-induced mouse acute lung injury and its mechanism. Chin. Pharmacol. Bull.32 (08), 1165–1170. 10.3969/j.issn.1001-1978.2016.08.026
Summary
Keywords
Peucedanum praeruptorum , pharmacology, phytochemistry, traditional uses, quality control
Citation
Wang Q, Sun Q, Huang Q, Qin L and Zhu B (2024) The traditional uses, pharmacology, and phytochemistry of Peucedanum praeruptorum Dunn. Front. Pharmacol. 15:1352657. doi: 10.3389/fphar.2024.1352657
Received
08 December 2023
Accepted
04 March 2024
Published
03 April 2024
Volume
15 - 2024
Edited by
Mohammad Reza Khazdair, Birjand University of Medical Sciences, Iran
Reviewed by
Noor Zulfiqar, University of Agriculture, Faisalabad, Pakistan
Neeraj Khatri, Institute of Microbial Technology (CSIR), India
Ravishankar Ramesh Patil, Amity University, India
Updates
Copyright
© 2024 Wang, Sun, Huang, Qin and Zhu.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Luping Qin, lpqin@zcmu.edu.cn; Bo Zhu, zhubo@zcmu.edu.cn
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.