- 1Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, Sichuan, China
- 2Chengdu Medical College, Chengdu, China
- 3Chengdu First People’s Hospital, Chengdu, Sichuan, China
A Corrigendum on
Efficacy of hyaluronic acid in the treatment of nasal inflammatory diseases: a systematic review and meta-analysis
by Liu H, Chen Y, Wang H, Luo X, Xie D, Ji Q and Tian L (2024). Front. Pharmacol. 15:1350063. doi: 10.3389/fphar.2024.1350063
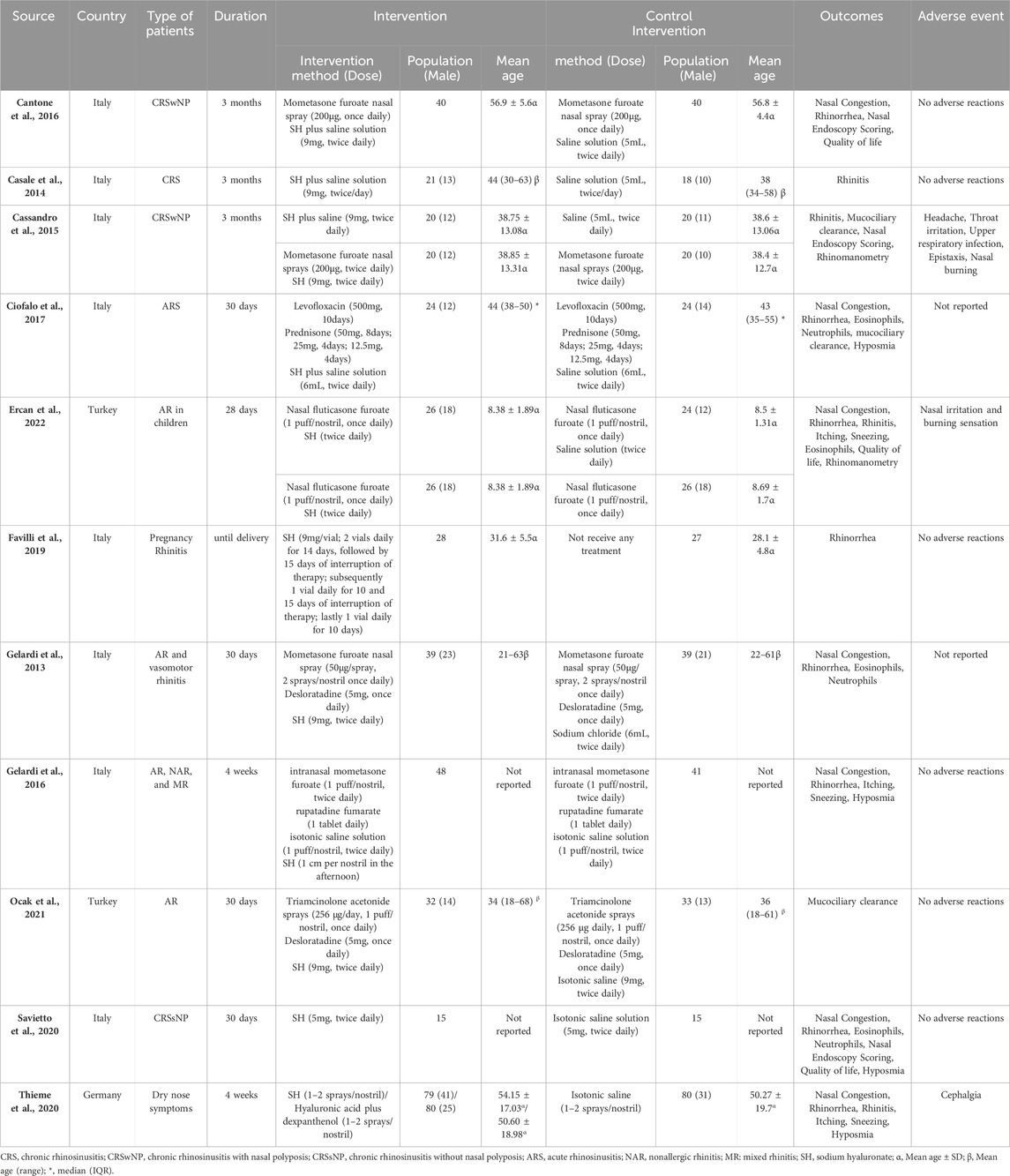
In the published article, there was an error in Table 1 as published. The authors found that the occurrence of cephalgia was possibly related the use of nasal spray (Thieme et al., 2020), however, this was omitted in the meta-analysis. The final row of Table 1 (source Thieme et al., 2020) has been corrected to reflect this. The corrected Table 1 and its caption appear below.
In the published article, there was an error. The authors found that the occurrence of cephalgia was possibly related the use of nasal spray (Thieme et al., 2020), however, this was omitted in the meta-analysis.
A correction has been made to 3 Results, 3.4 Outcomes, 3.4.4 Adverse events, Paragraph Number 1. The sentences previously stated:
“These studies (Cassandro et al., 2015; Ercan et al., 2022) reporting adverse events included nasal burning, headaches, throat irritation, upper respiratory tract infections, epistaxis, and nasal irritation. One trial (Cassandro et al., 2015) reported no difference in the incidence of adverse events between the HA and control groups, and the other trial (Ercan et al., 2022) showed only mild adverse events in the control group.”
The corrected sentence appears below:
“These studies (Cassandro et al., 2015; Ercan et al., 2022) reported adverse events including nasal burning, headache, throat irritation, upper respiratory tract infection, epistaxis, and nasal irritation. One patient reported thrice about the occurrence of cephalgia in hyaluronic acid plus dexpanthenol group, rated as possibly related to the application of the nasal spray (Thieme et al., 2020). One trial (Cassandro et al., 2015) reported no difference in the incidence of adverse events between the HA and control groups, and the other trial (Ercan et al., 2022) showed only mild adverse events in the control group.”
In the published article, there was an error in Table 1. In the final row, “α” was not in superscript. The corrected table and its caption appear above.
The authors apologize for these errors and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Keywords: hyaluronic acid, sodium hyaluronate, allergic rhinitis, sinusitis, nasal inflammatory diseases, meta-analysis
Citation: Liu H, Chen Y, Wang H, Luo X, Xie D, Ji Q and Tian L (2024) Corrigendum: Efficacy of hyaluronic acid in the treatment of nasal inflammatory diseases: a systematic review and meta-analysis. Front. Pharmacol. 15:1404434. doi: 10.3389/fphar.2024.1404434
Received: 21 March 2024; Accepted: 25 October 2024;
Published: 08 November 2024.
Edited and reviewed by:
Pablo Andres Evelson, University of Buenos Aires, ArgentinaCopyright © 2024 Liu, Chen, Wang, Luo, Xie, Ji and Tian. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Qing Ji, cWh5ODIwOTE1QDE2My5jb20=; Li Tian, Y3RjbWRhbkBzdHUuY2R1dGNtLmVkdS5jbg==
†These authors have contributed equally to this work
 Huixia Liu1†
Huixia Liu1† Qing Ji
Qing Ji Li Tian
Li Tian