- 1Department of Pharmacology, Shandong Provincial Hospital Affiliated to Shandong First Medical University, Jinan, China
- 2Cheeloo College of Medicine, Shandong University, Jinan, China
- 3Department of Pharmacology, Hospital for Skin Diseases, Shandong First Medical University, Jinan, China
- 4Department of Pharmacology, Shandong Provincial Institute of Dermatology and Venereology, Shandong Academy of Medical Sciences, Jinan, China
Background: Sunitinib is approved for the treatment of metastatic renal cell carcinoma (mRCC), imatinib-resistant gastrointestinal stromal tumors (GIST), and advanced pancreatic neuroendocrine tumors (PNET). This study aims to investigate the safety profiles of sunitinib through data mining of the US Food and Drug Administration Adverse Event Reporting System (FAERS).
Methods: The individual case safety reports (ICSRs) on sunitinib from 2006 Q1 to 2024 Q1 were collected from the ASCII data packages in the Food and Drug Administration Adverse Event Reporting System (FAERS). After standardizing the data, a variety of disproportionality analyses, including the reporting odds ratio (ROR), the proportional reporting ratio (PRR), the bayesian confidence propagation neural network (BCPNN), and the multi-item gamma Poisson shrinker (MGPS) were employed to identify the potential safety signals of sunitinib-associated AEs.
Results: A total of 35,923 ICSRs of sunitinib as the “primary suspected” drug were identified within the reporting period. The search detected 276 disproportionate preferred terms (PTs). The most common AEs, including diarrhea, asthenia, decreased appetite, hypertension, and dysgeusia, were consistent with the drug label and clinical trials. Unexpected significant AEs, such as uveal melanocytic proliferation, salivary gland fistula, yellow skin, eyelash discoloration, scrotal inflammation, were detected. The median onset time of sunitinib-related AEs was 57 days (interquartile range [IQR]16–170 days), with most of the ICSRs developing within the first month (n = 4,582, 39.73%) after sunitinib therapy as initiated.
Conclusion: The results of our study were consistent with routine clinical observations, and some unexpected AEs signals were also identified for sunitinib, providing valuable evidence for the safe use of sunitinib in the real-world and contributing to the clinical monitoring and risk identification of sunitinib.
1 Introduction
Sunitinib, an oral small-molecule multitargeted tyrosine kinase inhibitors (TKIs), exerts dual effects on anti-tumor angiogenesis and anti-tumor cell proliferation via inhibiting the vascular endothelial growth factor-1, 2, 3 (VEGF-1, 2, 3), the platelet-derived growth factor-α, β (PDGFr-α, β), the stem-cell growth factor receptor (KIT), colony-stimulating factor (CSF)-1R, FMS-like tyrosine kinase-3 (FLT-3), and the rearranged during transfection (RET) (Mena et al., 2010). The agent has been approved by the Food and Drug Administration (FDA) for the treatment of metastatic renal cell carcinoma (mRCC), gastrointestinal stromal tumor (GIST) refractory to imatinib therapy, and advanced pancreatic neuroendocrine tumor (PNET) (Raymond et al., 2011; Savard et al., 2020; Li et al., 2024). The available data suggests that it can significantly prolong the median progression-free survival and overall survival of patients with mRCC or GIST (Hopkins et al., 2008; Moran et al., 2019). Encouraging results have also been shown in clinical trials of sunitinib alone or in combination with other anti-tumour agents for the treatment of solid tumours such as lung and breast cancer (Crown et al., 2013; Tanday, 2015).
However, despite its significant clinical benefits, the widespread clinical use of sunitinib inevitably leads to adverse effects in patients. In clinical phase II and phase III studies of sunitinib, the most common adverse events (AEs) occurring in at least 25% of recipients include fatigue, rash, diarrhea, mucositis, loss of appetite, hand-foot syndrome, hypertension, hemorrhages, taste disturbances, and dyspepsia (Cella et al., 2008; Escudier et al., 2009). Additionally, sunitinib has an FDA “black box” warning for hepatotoxicity, which has the potential to be fatal (Amaya et al., 2018). Therefore, it is highly desirable to utilize data mining algorithms to identify potential safety signals of sunitinib in real-world settings.
The Food and Drug Administration Adverse Event Reporting System (FAERS) is one of the largest post-marketing safety monitoring databases that document real-world standardized data, which can be used for identifying and analyzing potential drug-AEs associations (Michel et al., 2017). We performed a retrospective pharmacovigilance study to detect the AEs signals of sunitinib based on disproportionality analysis methods. This research aimed to find unexpected AEs that were not described on the drug’s label and offer valuable reference for clinical practices.
2 Materials and methods
2.1 Data source and processing
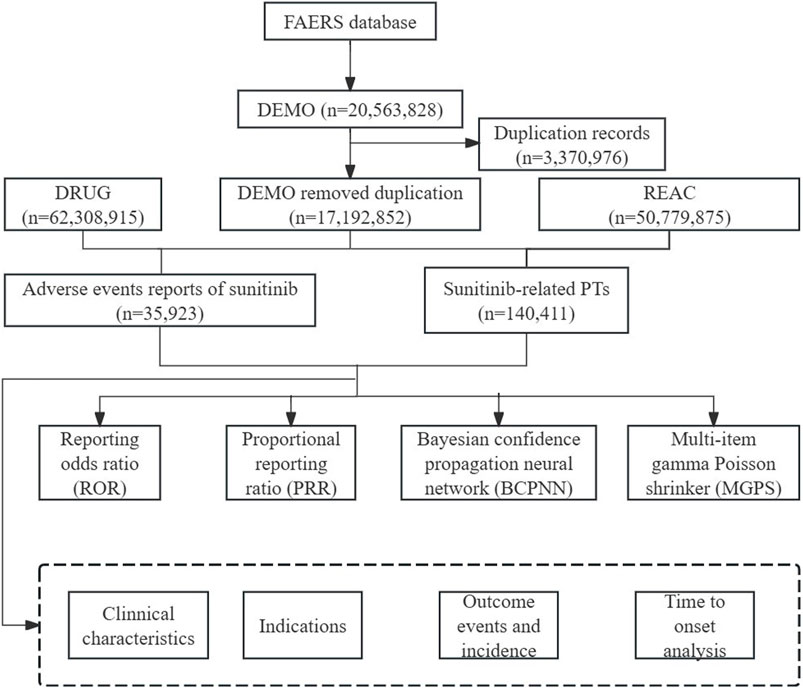
FAERS is the primary system in the United States for conducting post-marketing adverse drug reaction surveillance and is one of the main avenues for current pharmacovigilance research (Dhodapkar et al., 2022). In our pharmacovigilance study, the ASCII data packages submitted from the first quarter of 2006 to the first quarter of 2024 were retrieved from the database. All data analyses were imported into SAS 9.4 and Excel software for data cleaning and analyses. The sample group was chosen by screening for DRUGNAME and PROD_AI, using both the generic and brand names (sunitinib, Sutent®) as keywords, with the suspicion level for reporting limited to “primary suspect” drugs. Based on the latest version of the Medical Dictionary for Regulatory Activities (MedDRA) 26.0 dictionary, AEs of sunitinib are coded on preferred terms (PT) and system organ class (SOC) levels, and the toxicity spectrum of sunitinib was investigated. According to the recommended approach by the FDA to remove duplicate reports, choose the PRIMARYID, CASEID, and FDA_DT fields of the DEMO table, sort them by CASEID, FDA_DT, and PRIMARYID, and keep the report with the highest FDA_DT value for individual case safety reports (ICSRs) with the same CASEID. Additionally, keep the one with the highest PRIMARYID value for reports with both the same CASEID and FDA_DT. Ensure retention of the one with the largest PRIMARYID value. Since Q1 2019, a roster of erased reports has been included in every quarterly packet. Following data de-duplication, reports are deleted using the CASEID included in the roster above. The comprehensive screening procedure was depicted in Figure 1.
2.2 Onset time analysis
The onset time of sunitinib-related events was calculated by subtracting the date of AE occurrence from the date of sunitinib initiation. It is crucial to highlight that the study excluded erroneous ICSRs whose occurrence date preceded the initiation of the drug or ICSRs of unknown time of onset. The median and interquartile ranges were employed to characterize the time to onset.
2.3 Statistical analysis
Descriptive analyses were used to present all sunitinib-related AEs reporting characteristics. In our study, disproportionate analyses, including reporting odds ratio (ROR) (Sakaeda et al., 2013), proportional reporting ratio (PRR) (Kelly et al., 2007), bayesian confidence propagation neural network (BCPNN) (Bate et al., 1998), and multinomial gamma Poisson shrinkage (MGPS) (Szarfman et al., 2004), were performed to identify signals indicating a possible increased risk of sunitinib-related AEs. The ROR and PRR are classified as frequency methods, and the methods demonstrate high sensitivity but low specificity. The BCPNN and MGPS are categorized as Bayesian methods which are suitable for handling complex variables, but with low sensitivity (Zou et al., 2023). Accordingly, multiple algorithms are combined to ensure the stability and reliability of the research results. The larger the value of the four parameters, the stronger the signal strength is. The relevant numerical values are the signal strength. The formulas and criteria for the four algorithms are shown in Table 1. (Liu et al., 2024; Tang et al., 2024). The AEs uncovered in the latest version of the sunitinib label issued by the FDA were defined as unexpected AEs. (FDA LABEL, 2021).
3 Results
3.1 Descriptive analysis
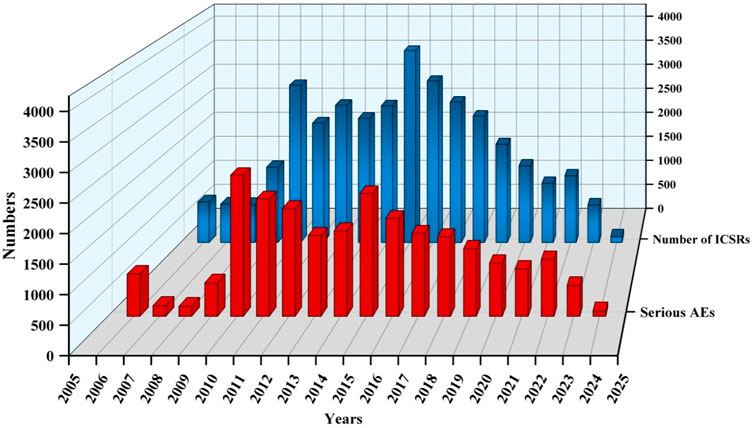
A total of 20,563,828 ICSRs were submitted to the FAERS database during the study period, among which there were 35,923 ICSRs on sunitinib of PS. The clinical characteristics of events regarding sunitinib were presented in Table 2 and Figure 2. Among all ICSRs, more males (59.35%) than females (31.85%) were reported. In terms of age, patients aged>65 contributed to the majority of ICSRs (38.55%). Death (28.92%) and hospitalization-initial or prolonged (28.38%) were the most common serious outcomes, and the high proportion of deaths might be more related to cancer progression in AEs. The country that reported the most was the United States (48.87%), and the primary ICSRs were consumers (38.13%) and physicians (30.93%). In terms of year of reporting, ICSRs were concentrated in 2010 (3085 cases), 2015 (3,760 cases), and 2016 (3,167 cases).
3.2 Potential safety signal detection results
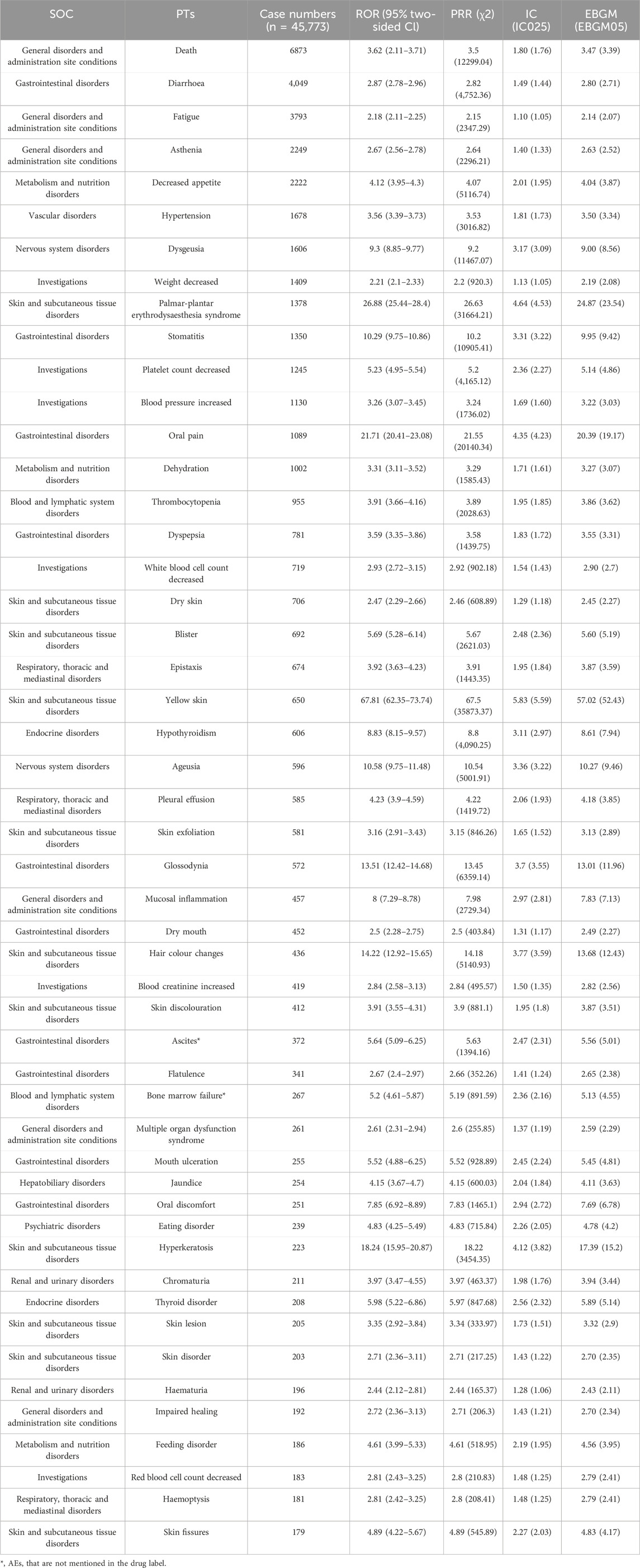
We performed signal detection of sunitinib-associated AEs at the SOC level which was shown in Table 3. AEs of sunitinib involved a total of 27 organ systems. Statistically, the SOC that met all four criteria simultaneously and showed significant association with sunitinib AEs were gastrointestinal disorders (n = 24452, ROR 2.27, PRR 2.04, IC 1.03, EBGM 2.04) and endocrine disorders (n = 1128, ROR 3.21, PRR 3.21, IC 1.67, EBGM 3.18). Additionally, general disorders and administration site conditions (n = 28170) were the most common SOCs. Of note, skin and subcutaneous tissue disorders (n = 10290), blood and lymphatic system disorders (n = 4,123), and vascular disorders (n = 3864) were also common and noteworthy SOC categories.
Based on the signal frequency and the signal strength (adopting the most sensitive ROR algorithm results), we respectively ranked these AEs which satisfied all four screening methods, as detailed in Tables 4, 5. Excepting in AEs of death (n = 6873), in our results, diarrhea (n = 4,049), fatigue (n = 3793), asthenia (n = 2249), decreased appetite (n = 2222), hypertension (n = 1678), and dysgeusia (n = 1606) were the most common AEs, which were consistent with the label and clinical trials. The AEs with significant potential risk signals included diffuse uveal melanocytic proliferation (ROR = 131.15, PRR = 131.15, IC = 6.59, EBGM = 96.44), salivary gland fistula (ROR = 98.36, PRR = 98.36, IC = 6.28, EBGM = 77.50), yellow skin (ROR = 67.81, PRR = 67.50, IC = 5.83, EBGM = 57.02), eyelash discoloration (ROR = 64.03, PRR = 64.02, IC = 5.77, EBGM = 54.52), scrotal inflammation (ROR = 39.20, PRR = 39.20, IC = 5.15, EBGM = 35.46), among others. Furthermore, comparing with the latest package insert of released by the FDA, unexpected significant AEs were discovered, such as including diffuse uveal melanocytic proliferation (ROR = 131.15, PRR = 131.15, IC = 6.59, EBGM = 96.44), salivary gland fistula (ROR = 98.36, PRR = 98.36, IC = 6.28, EBGM = 77.50), scrotal inflammation (ROR = 39.20, PRR = 39.20, IC = 5.15, EBGM = 35.46), thyroid atrophy (ROR = 36.07, PRR = 36.07, IC = 5.04, EBGM = 32.88), esophagobronchial fistula (ROR = 36.07, PRR = 36.07, IC = 5.04, EBGM = 32.88), perihepatic discomfort (ROR = 15.03, PRR = 15.03, IC = 3.85, EBGM = 14.47) and vena cava injury (ROR = 14.24, PRR = 14.24, IC = 3.78, EBGM = 13.73). Of note, nausea, vomiting, malaise, pain in extremity, pyrexia, constipation, and abdominal pain in the drug label did not meet the criteria for at least one of the four algorithms.
3.3 Time-to-onset analysis
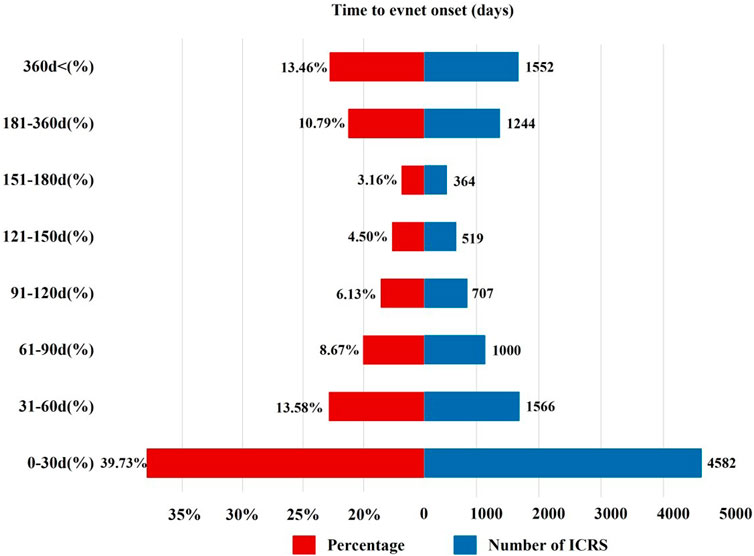
The onset time of sunitinib-related events was collected, with unreported onset time reports or erroneous reports excluded from the analysis. A total of 11,534 ICSRs were eligible for the inclusion criteria, and the mean time to onset was 207 days, with a median onset time of 51 days (interquartile range [IQR] 16–170 days). Our data revealed that 39.73% of ICSRs occurred within the first month following sunitinib administration (n = 4,582). Notably, AEs might still occur after 1 year of sunitinib treatment, accounting for 13.39% of the total cases, as illustrated in Figure 3.
4 Discussion
In recent years, several studies have carried out pharmacovigilance analyses of TKIs on electrolyte abnormalities, psychiatric disorders, and neuropsychiatric events (Raschi et al., 2022; Barbieri et al., 2023; Wang et al., 2023; She et al., 2024). However, the comprehensive safety profiles of sunitinib-induced AEs have not been reported. In this study, we systematically collect and assess the post-marketing AEs of sunitinib based on real-world pharmacovigilance data. The study aimed to identify new and significant risk signals and enhance the safety of clinical drug therapy. The ICSRs of sunitinib occurred more commonly in males (59.53%) than in females (31.85%), and a higher ICSRs proportion in elderly individuals over the age of 65 (38.83%), which could be attributed to the higher incidence of RCC in elderly males. It has been reported that the prevalence of renal cell carcinoma in males is approximately twice that in females and tends to increase with age (Scelo et al., 2018; Scelo and Larose, 2018). Besides, the median age of most patients at diagnosis is around 75 years old (Bukavina et al., 2022). Regarding reporting countries, the United States stands out as the most prominent reporting country (49.16%), possibly due to the earlier availability and higher prescription volume of the drug in the country. Notably, consumers (38.20%) were the predominant group reporting ICSRs, followed by physicians (30.98%). This indicates the need for heightening vigilance among clinical doctors and pharmacists in monitoring sunitinib-related AEs, particularly in the elderly population, to reduce the occurrence of life-threatening AEs.
Our disproportionality analyses identified that the significant SOCs were gastrointestinal disorders and endocrine disorders, and the most common SOC was general disorders and administration site conditions. As shown in Table 4, common AEs included diarrhea, fatigue, asthenia, decreased appetite, hypertension, dysgeusia, hand-foot syndrome, and stomatitis, which were mostly consistent with the insert and clinical trials (Saltz et al., 2007; Baumann et al., 2012; Jonasch et al., 2018). Hypertension is most common upon treatment with sunitinib (Pal et al., 2021). All-grade hypertension has been documented in up to 30% of mRCC patients receiving sunitinib, with grade 3 hypertension in 12% of cases (Motzer et al., 2009). Besides, hypertension was also a relatively common AE among other vascular endothelial growth factor TKIs (Zhu et al., 2009; Bæk Møller et al., 2019). The underlying mechanism is currently believed to be the activation of the endothelin-1 pathway and the disruption of endothelial cell survival signaling, resulting in reduced capillary density and diminished nitric oxide secretion (Kappers et al., 2010; Rini et al., 2011). It has been reported that hypertension often occurs as a complication in the early stages of treatment with TKIs, such as sorafenib and axitinib (Motzer et al., 2009). Therefore, it is recommended that blood pressure is regularly monitored at least once a week during the initial 6 weeks of sunitinib treatment (Zhu et al., 2009). In case of severe or persistent hypertension, it is advisable to withdraw sunitinib immediately and initiate antihypertensive treatment.
Some AEs with high signal intensity were also examined, such as yellow skin, eyelash discoloration, hand-foot syndrome (HFS), genital injury (including anal injuries, genital rashes, desquamation, ulcers, etc.), and bleeding events. Approximately 24% of patients suffered from skin discoloration, including yellow skin and pigmentation after sunitinib therapy (Rosenbaum et al., 2008). Of note, the yellow color of the medication may be a contributing factor to the development of yellow skin. Hair depigmentation occurred in 15.2% of patients, and the inhibition of multiple signaling pathways, such as platelet-derived growth factor receptors (PDGFR), seemed to have a role in the pathogenesis (Joensuu et al., 2011). The incidence of HFS was reported between 13.5% and 25%, of which 4%–7% of cases were grade 3 to 4. The onset of HFS was 2–4 weeks. The potential mechanism behind HFS is not completely understood, but it is hypothesized to involve the inhibition of VEGFR and PDGFR, resulting in vascular deformation and cell apoptosis in the dermis (Terada et al., 2015). Billemont et al. reported that 12.5% of patients experienced genital rash and anal injuries, with a median onset time of 66 days (Billemont et al., 2008). The mechanism of this AE is still largely unclear, and it may be related to VEGF and hypoxia-inducible factor-1a (Billemont et al., 2008; Chou et al., 2013). Lastly, various bleeding events such as splinter haemorrhage, umbilical haemorrhage, mediastinal haemorrhage, and scleral haemorrhage were also detected in our study. In a meta-analysis of patients with sunitinib and sorafenib, the incidence was 16.7% for all-grade bleeding events and 2.4% for grade 3/4 toxicity (Je et al., 2009). The increased risk of bleeding may be mediated by VEGF inhibition and concomitant administration with antiplatelet agents (Je et al., 2009). In addition, sunitinib-associated thrombocytopenia, with an incidence of 22% (Lee et al., 2014), was hypothesized to be an intriguing explanation for bleeding events.
Furthermore, unexpected and significant safety signals, such as diffuse uveal melanocytic proliferation, vena cava injury, esophagobronchial fistula, perihepatic discomfort, and thyroid atrophy, were detected in our analysis. Regardless, medical staff should note that patients on sunitinib are at risk of these unexpected AEs. So far, there have been no documented reports about diffuse uveal melanocytic proliferation and vena cava injury Therefore, it is necessary to further investigate the pathogenesis of these incidents. Basille et al. described a 40-year-old male with renal cell carcinoma who developed an esophagotracheal fistula after the administration of sunitinib for 2 months (Basille et al., 2010). However, the exact mechanism of esophagotracheal fistula remained unclear. In our study, we observed that sunitinib-induced hepatoxicity AEs mainly included perihepatic discomfort, liver failure, and jaundice. Approximately 40% of patients with sunitinib experienced elevated liver enzymes, and 3% of patients developed grade 3 or 4 hepatotoxicity (Ibrahim et al., 2013). Additionally, similar AEs have also been observed in other VEGF-TKIs (Teo et al., 2013). The onset time is 1–3 weeks after initiation of treatment or even several months later (Lammert et al., 2008). Toxic intermediate metabolites, mitochondrial dysfunction, and glycolysis inhibition have been described as possible mechanisms (Teo et al., 2013; Paech et al., 2017). Furthermore, several cases of sunitinib-related fulminant acute liver failure have been reported (Aqsa et al., 2021; Casas Deza et al., 2021). In 2010, FDA issued a black box warning that sunitinib-associated hepatotoxicity may be severe, and even fatal. Clinicians should be aware that liver failure is a complication unrelated to dosage or tumor progression, and liver function should be regularly monitored during the first year of treatment. According to a prospective study, the incidence of hypothyroidism as a common complication following sunitinib treatment was found to be 36% (Desai et al., 2006). However, our research findings suggested that sunitinib-induced thyroid atrophy represented a novel, unexpected, and rarely reported thyroid toxicity. A long-term study has indicated that the thyroid volume decreased by approximately 30% in metastatic renal cell carcinoma patients treated with sunitinib, and those with a reduction of over 50% in thyroid volume experienced a dramatically increased incidence of hypothyroidism (Shinohara et al., 2011). Thyroid atrophy induced by sunitinib may be attributed to the induction of follicular atrophy and apoptosis of thyroid cells, with its toxicity being a temporal relationship (Sakurai et al., 2010; Grosse et al., 2014). It has been observed that patients with sunitinib-induced hypothyroidism may show improvement upon suspension of the medication and initiation of levothyroxine therapy. However, in cases where severe thyroid atrophy caused by sunitinib leads to secondary hypothyroidism, it may be deemed irreversible (Desai et al., 2006). Therefore, regular monitoring of thyroid toxicity, particularly thyroid volume, is crucial during sunitinib treatment.
In this study, there is a wide variance between the onset of AEs and sunitinib application. The median onset time of reported AEs was 51 days, and 39.73% of ICSRs occurred within the first month after sunitinib initiation. However, 13.39% of ICSRs developed with a delayed onset, occurring 1 year after sunitinib therapy. Consistently, in a phase III clinical trial involving 312 GIST patients, the median onset time of AEs caused by sunitinib was 56 days, and the onset time of some AEs was longer. For example, the average onset time of hypothyroidism was 350 days (Joensuu et al., 2011). Therefore, it is imperative to be vigilant about the AEs throughout the whole course of treatment with sunitinib, and long-term follow-up for some AEs may be needed.
The pre-marketing drug safety studies are characterized by the small number of enrolled cases, brief observation periods, stringent medication usage conditions, etc. Thus, the AEs observed in pre-marketing trials of a drug may not reflect all AEs observed in practice. The spontaneous reporting database, with a wide monitoring range and early detection of suspected AE signals, was extensively used to carry out post-marketing pharmacovigilance on drug safety. In recent years, with the wide use of sunitinib in clinical practice there is an emerging need to make further evaluation of its safety profiles in a real-world environment. Our study first comprehensively and scientifically explores the post-marketing safety profiles of sunitinib based on AE reports from the FAERS database. Furthermore, some unexpected AEs were detected and the onset time of all AEs was analyzed, which guides the safe clinical use of sunitinib.
5 Conclusion
In conclusion, our study conducted a systematic and comprehensive exploration of the signals associated with sunitinib based on the FAERS database. The common AEs detected in this study were consistent with the manufacturer’s labeling and clinic trials. Some unexpected AEs were also revealed, such as diffuse uveal melanocytic proliferation, thyroid atrophy, esophagobronchial fistula, vena cava injury, and perihepatic discomfort. Furthermore, the median onset time of AEs was analyzed, which enables clinicians and pharmacists to make informed decisions regarding sunitinib. However, given the exploratory character of our work, future prospective clinical trials and long-term data are needed to validate these results and provide valuable evidence for the safety profile of sunitinib.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding authors.
Author contributions
XZ: Data curation, Formal Analysis, Investigation, Methodology, Writing–original draft. XR: Data curation, Formal Analysis, Investigation, Methodology, Writing–original draft. TZ: Data curation, Investigation, Writing–original draft. WZ: Data curation, Writing–original draft. CS: Conceptualization, Formal Analysis, Supervision, Writing–review and editing. CL: Conceptualization, Formal Analysis, Supervision, Writing–review and editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was supported by the Natural Science Foundation of Shandong Province, China (Nos ZR2022QH109, ZR2023LSW013, and ZR2023MH077).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Amaya, G. M., Durandis, R., Bourgeois, D. S., Perkins, J. A., Abouda, A. A., Wines, K. J., et al. (2018). Cytochromes P450 1A2 and 3A4 catalyze the metabolic activation of sunitinib. Chem. Res. Toxicol. 31, 570–584. doi:10.1021/acs.chemrestox.8b00005
Aqsa, A., Droubi, S., Amarnath, S., Al-Moussawi, H., and Abergel, J. (2021). Sunitinib-induced acute liver failure. Case Rep. Gastroenterol. 15, 17–21. doi:10.1159/000511249
Bæk Møller, N., Budolfsen, C., Grimm, D., Krüger, M., Infanger, M., Wehland, M., et al. (2019). Drug-induced hypertension caused by multikinase inhibitors (sorafenib, sunitinib, lenvatinib and axitinib) in renal cell carcinoma treatment. Int. J. Mol. Sci. 20, 4712. doi:10.3390/ijms20194712
Barbieri, M. A., Sorbara, E. E., Russo, G., Cicala, G., Franchina, T., Santarpia, M., et al. (2023). Neuropsychiatric adverse drug reactions with tyrosine kinase inhibitors in gastrointestinal stromal tumors: an analysis from the European spontaneous adverse event reporting system. Cancers 15 (6), 1851. doi:10.3390/cancers15061851
Basille, D., Andrejak, M., Bentayeb, H., Kanaan, M., Fournier, C., Lecuyer, E., et al. (2010). Bronchial fistula associated with sunitinib in a patient previously treated with radiation therapy. Ann. Pharmacother. 44, 383–386. doi:10.1345/aph.1M469
Bate, A., Lindquist, M., Edwards, I. R., Olsson, S., Orre, R., Lansner, A., et al. (1998). A Bayesian neural network method for adverse drug reaction signal generation. Eur. J. Clin. Pharmacol. 54 (4), 315–321. doi:10.1007/s002280050466
Baumann, K. H., du Bois, A., Meier, W., Rau, J., Wimberger, P., Sehouli, J., et al. (2012). A phase II trial (AGO 2.11) in platinum-resistant ovarian cancer: a randomized multicenter trial with sunitinib (SU11248) to evaluate dosage, schedule, tolerability, toxicity and effectiveness of a multitargeted receptor tyrosine kinase inhibitor monotherapy. Ann. Oncol. 23, 2265–2271. doi:10.1093/annonc/mds003
Billemont, B., Barete, S., and Rixe, O. (2008). Scrotal cutaneous side effects of sunitinib. N. Engl. J. Med. 359, 975–976. doi:10.1056/NEJMc0802736
Bukavina, L., Bensalah, K., Bray, F., Carlo, M., Challacombe, B., Karam, J. A., et al. (2022). Epidemiology of renal cell carcinoma: 2022 update. Eur. Urol. 82, 529–542. doi:10.1016/j.eururo.2022.08.019
Casas Deza, D., Gascón Ruiz, M., Lamuela Calvo, L. J., Sierra Gabarda, O., Betoré Glaria, E., and Bernal Monterde, V. (2021). Fulminant liver failure in a patient treated with Sunitinib for metastatic gastrointestinal stromal tumor. Gastroenterol. Hepatol. 44, 424–425. doi:10.1016/j.gastrohep.2020.08.006
Cella, D., Li, J. Z., Cappelleri, J. C., Bushmakin, A., Charbonneau, C., Kim, S. T., et al. (2008). Quality of life in patients with metastatic renal cell carcinoma treated with sunitinib or interferon alfa: results from a phase III randomized trial. J. Clin. Oncol. 26, 3763–3769. doi:10.1200/JCO.2007.13.5145
Chou, C. Y., Wang, K. H., Lin, Y. H., Lin, Y. T., and Tsai, H. H. (2013). Sunitinib-induced scrotal cutaneous side-effect. J. Dermatol 40, 67–68. doi:10.1111/j.1346-8138.2012.01681.x
Crown, J. P., Diéras, V., Staroslawska, E., Yardley, D. A., Bachelot, T., Davidson, N., et al. (2013). Phase III trial of sunitinib in combination with capecitabine versus capecitabine monotherapy for the treatment of patients with pretreated metastatic breast cancer. J. Clin. Oncol. 31 (23), 2870–2878. doi:10.1200/JCO.2012.43.3391
Desai, J., Yassa, L., Marqusee, E., George, S., Frates, M. C., Chen, M. H., et al. (2006). Hypothyroidism after sunitinib treatment for patients with gastrointestinal stromal tumors. Ann. Intern. Med. 145, 660–664. doi:10.7326/0003-4819-145-9-200611070-00008
Dhodapkar, M. M., Shi, X., Ramachandran, R., Chen, E. M., Wallach, J. D., and Ross, J. S. (2022). Characterization and corroboration of safety signals identified from the US Food and drug administration adverse event reporting system, 2008-19: cross sectional study. BMJ 379, e071752. doi:10.1136/bmj-2022-071752
Escudier, B., Roigas, J., Gillessen, S., Harmenberg, U., Srinivas, S., Mulder, S. F., et al. (2009). Phase II study of sunitinib administered in a continuous once-daily dosing regimen in patients with cytokine-refractory metastatic renal cell carcinoma. J. Clin. Oncol. 27, 4068–4075. doi:10.1200/JCO.2008.20.5476
FDA Label (2021). SUTENT® (sunitinib malate). Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/021938s039lbledt.pdf (Accessed June 25, 2024).
George, S., Reichardt, P., Lechner, T., Li, S., Cohen, D. P., and Demetri, G. D. (2012). Hypertension as a potential biomarker of efficacy in patients with gastrointestinal stromal tumor treated with sunitinib. Ann. Oncol. 23, 3180–3187. doi:10.1093/annonc/mds179
Grosse, J., Warnke, E., Wehland, M., Pietsch, J., Pohl, F., Wise, P., et al. (2014). Mechanisms of apoptosis in irradiated and sunitinib-treated follicular thyroid cancer cells. Apoptosis 19, 480–490. doi:10.1007/s10495-013-0937-0
Hopkins, T. G., Marples, M., and Stark, D. (2008). Sunitinib in the management of gastrointestinal stromal tumours (GISTs). Target. Oncol. 34 (8), 844–850. doi:10.1016/j.ejso.2007.10.011
Ibrahim, E. M., Kazkaz, G. A., Abouelkhair, K. M., Bayer, A. M., and Elmasri, O. A. (2013). Sunitinib adverse events in metastatic renal cell carcinoma: a meta-analysis. Int. J. Clin. Oncol. 18, 1060–1069. doi:10.1007/s10147-012-0497-2
Je, Y., Schutz, F. A., and Choueiri, T. K. (2009). Risk of bleeding with vascular endothelial growth factor receptor tyrosine-kinase inhibitors sunitinib and sorafenib: a systematic review and meta-analysis of clinical trials. Lancet. Oncol. 10, 967–974. doi:10.1016/S1470-2045(09)70222-0
Joensuu, H., Trent, J. C., and Reichardt, P. (2011). Practical management of tyrosine kinase inhibitor-associated side effects in GIST. Cancer Treat. Rev. 37, 75–88. doi:10.1016/j.ctrv.2010.04.008
Jonasch, E., Slack, R. S., Geynisman, D. M., Hasanov, E., Milowsky, M. I., Rathmell, W. K., et al. (2018). Phase II study of two weeks on, one week off sunitinib scheduling in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 36, 1588–1593. doi:10.1200/JCO.2017.77.1485
Kappers, M. H., van Esch, J. H., Sluiter, W., Sleijfer, S., Danser, A. H., and van den Meiracker, A. H. (2010). Hypertension induced by the tyrosine kinase inhibitor sunitinib is associated with increased circulating endothelin-1 levels. Hypertension 56, 675–681. doi:10.1161/HYPERTENSIONAHA.109.149690
Kelly, W. N., Arellano, F. M., Barnes, J., Bergman, U., Edwards, R. I., Fernandez, A. M., et al. (2007). Guidelines for submitting adverse event reports for publication. Drug Saf. 30 (5), 367–373. doi:10.2165/00002018-200730050-00001
Lammert, C., Einarsson, S., Saha, C., Niklasson, A., Bjornsson, E., and Chalasani, N. (2008). Relationship between daily dose of oral medications and idiosyncratic drug-induced liver injury: search for signals. Hepatology 47, 2003–2009. doi:10.1002/hep.22272
Lee, S. H., Bang, Y. J., Mainwaring, P., Ng, C., Chang, J. W., Kwong, P., et al. (2014). Sunitinib in metastatic renal cell carcinoma: an ethnic Asian subpopulation analysis for safety and efficacy. Asia Pac. J. Clin. Oncol. 10, 237–245. doi:10.1111/ajco.12163
Li, J., Zhang, J., Zhang, Y., Qiu, H., Zhou, Y., Zhou, Y., et al. (2024). Efficacy and safety of ripretinib vs. sunitinib in patients with advanced gastrointestinal stromal tumor previously treated with imatinib: a phase 2, multicenter, randomized, open-label study in China. Eur. J. cancer (Oxford, Engl. 1990) 196, 113439. doi:10.1016/j.ejca.2023.113439
Liu, M., Gu, L., Zhang, Y., Zhou, H., Wang, Y., and Xu, Z. X. (2024). A real-world disproportionality analysis of mesalazine data mining of the public version of FDA adverse event reporting system. Front. Pharmacol. 15, 1290975. doi:10.3389/fphar.2024.1290975
Mena, A. C., Pulido, E. G., and Guillén-Ponce, C. (2010). Understanding the molecular-based mechanism of action of the tyrosine kinase inhibitor: sunitinib. Anticancer. Drugs. 21 (Suppl. 1), S3–S11. doi:10.1097/01.cad.0000361534.44052.c5
Michel, C., Scosyrev, E., Petrin, M., and Schmouder, R. (2017). Can disproportionality analysis of post-marketing case reports be used for comparison of drug safety profiles? Clin. Drug Investig. 37 (5), 415–422. doi:10.1007/s40261-017-0503-6
Moran, M., Nickens, D., Adcock, K., Bennetts, M., Desscan, A., Charnley, N., et al. (2019). Sunitinib for metastatic renal cell carcinoma: a systematic review and meta-analysis of real-world and clinical trials data. Target. Oncol. 14 (4), 405–416. doi:10.1007/s11523-019-00653-5
Motzer, R. J., Hutson, T. E., Tomczak, P., Michaelson, M. D., Bukowski, R. M., Oudard, S., et al. (2009). Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 27 (22), 3584–3590. doi:10.1200/JCO.2008.20.1293
Paech, F., Bouitbir, J., and Krähenbühl, S. (2017). Hepatocellular toxicity associated with tyrosine kinase inhibitors: mitochondrial damage and inhibition of glycolysis. Front. Pharmacol. 8, 367. doi:10.3389/fphar.2017.00367
Pal, S. K., Tangen, C., Thompson, I. M., Balzer-Haas, N., George, D. J., Heng, D. Y. C., et al. (2021). A comparison of sunitinib with cabozantinib, crizotinib, and savolitinib for treatment of advanced papillary renal cell carcinoma: a randomised, open-label, phase 2 trial. Lancet 397, 695–703. doi:10.1016/S0140-6736(21)00152-5
Raschi, E., Fusaroli, M., Giunchi, V., Repaci, A., Pelusi, C., Mollica, V., et al. (2022). Adrenal insufficiency with anticancer tyrosine kinase inhibitors targeting vascular endothelial growth factor receptor: analysis of the FDA adverse event reporting system. Cancers 14 (19), 4610. doi:10.3390/cancers14194610
Raymond, E., Dahan, L., Raoul, J. L., Bang, Y. J., Borbath, I., Lombard-Bohas, C., et al. (2011). Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 501–513. doi:10.1056/NEJMoa1003825
Rini, B. I., Cohen, D. P., Lu, D. R., Chen, I., Hariharan, S., Gore, M. E., et al. (2011). Hypertension as a biomarker of efficacy in patients with metastatic renal cell carcinoma treated with sunitinib. J. Natl. Cancer Inst. 103, 763–773. doi:10.1093/jnci/djr128
Rosenbaum, S. E., Wu, S., Newman, M. A., West, D. P., Kuzel, T., and Lacouture, M. E. (2008). Dermatological reactions to the multitargeted tyrosine kinase inhibitor sunitinib. Support. Care Cancer. 16, 557–566. doi:10.1007/s00520-008-0409-1
Sakaeda, T., Tamon, A., Kadoyama, K., and Okuno, Y. (2013). Data mining of the public version of the FDA adverse event reporting system. Int. J. Med. Sci. 10 (7), 796–803. doi:10.7150/ijms.6048
Sakurai, K., Fukazawa, H., Arihara, Z., and Yoshida, K. (2010). Sunitinib-induced thyrotoxicosis followed by persistent hypothyroidism with shrinkage of thyroid volume. Tohoku. J. Exp. Med. 222 (1), 39–44. doi:10.1620/tjem.222.39
Saltz, L. B., Rosen, L. S., Marshall, J. L., Belt, R. J., Hurwitz, H. I., Eckhardt, S. G., et al. (2007). Phase II trial of sunitinib in patients with metastatic colorectal cancer after failure of standard therapy. J. Clin. Oncol. 25, 4793–4799. doi:10.1200/JCO.2007.12.8637
Savard, M. F., Wells, J. C., Graham, J., Dudani, S., Steinharter, J. A., McGregor, B. A., et al. (2020). Real-world assessment of clinical outcomes among first-line sunitinib patients with clear cell metastatic renal cell carcinoma (mRCC) by the international mRCC database consortium risk group. Oncologist 25 (5), 422–430. doi:10.1634/theoncologist.2019-0605
Scelo, G., and Larose, T. L. (2018). Epidemiology and risk factors for kidney cancer. J. Clin. Oncol. 36 (36), JCO2018791905. doi:10.1200/JCO.2018.79.1905
Scelo, G., Li, P., Chanudet, E., and Muller, D. C. (2018). Variability of sex disparities in cancer incidence over 30 Years: the striking case of kidney cancer. Eur. Urol. Focus 4, 586–590. doi:10.1016/j.euf.2017.01.006
She, X., Yin, D., Guo, Q., Tang, Y., Wang, S., and Wang, X. (2024). Electrolyte disorders induced by six multikinase inhibitors therapy for renal cell carcinoma: a large-scale pharmacovigilance analysis. Sci. Rep. 14 (1), 5592. doi:10.1038/s41598-024-56335-4
Shinohara, N., Takahashi, M., Kamishima, T., Ikushima, H., Otsuka, N., Ishizu, A., et al. (2011). The incidence and mechanism of sunitinib-induced thyroid atrophy in patients with metastatic renal cell carcinoma. Br. J. Cancer. 104 (2), 241–247. doi:10.1038/sj.bjc.6606029
Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., et al. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Ca. Cancer J. Clin. 71, 209–249. doi:10.3322/caac.21660
Szarfman, A., Tonning, J. M., and Doraiswamy, P. M. (2004). Pharmacovigilance in the 21st century: new systematic tools for an old problem. Pharmacotherapy 24 (9), 1099–1104. doi:10.1592/phco.24.13.1099.38090
Tanday, S. (2015). Sunitinib benefits patients with small-cell lung cancer. Lancet. Oncol. 16 (4), e164. doi:10.1016/S1470-2045(15)70120-8
Tang, L., Sun, C., Liu, W., Wu, H., and Ding, C. (2024). A pharmacovigilance study on antibody-drug conjugate (ADC)-related neurotoxicity based on the FDA adverse event reporting system (FAERS). Front. Pharmacol. 15, 1362484. doi:10.3389/fphar.2024.1362484
Teo, Y. L., Ho, H. K., and Chan, A. (2013). Risk of tyrosine kinase inhibitors-induced hepatotoxicity in cancer patients: a meta-analysis. Cancer Treat. Rev. 39, 199–206. doi:10.1016/j.ctrv.2012.09.004
Terada, T., Noda, S., and Inui, K. (2015). Management of dose variability and side effects for individualized cancer pharmacotherapy with tyrosine kinase inhibitors. Pharmacol. Ther. 152, 125–134. doi:10.1016/j.pharmthera.2015.05.009
Wang, X., Yin, D., Tang, Y., Xiao, F., and Wang, S. (2023). Psychiatric adverse reactions to non-selective RET multi-kinase inhibitors: a large-scale pharmacovigilance analysis. Front. Pharmacol. 14, 1209933. doi:10.3389/fphar.2023.1209933
Zhu, X., Stergiopoulos, K., and Wu, S. (2009). Risk of hypertension and renal dysfunction with an angiogenesis inhibitor sunitinib: systematic review and meta-analysis. Acta. Oncol. 48 (1), 9–17. doi:10.1080/02841860802314720
Keywords: sunitinib, FDA adverse event reporting system, disproportionality analyses, pharmacovigilance, adverse event
Citation: Zhang X, Ren X, Zhu T, Zheng W, Shen C and Lu C (2024) A real-world pharmacovigilance study of FDA adverse event reporting system (FAERS) events for sunitinib. Front. Pharmacol. 15:1407709. doi: 10.3389/fphar.2024.1407709
Received: 27 March 2024; Accepted: 12 July 2024;
Published: 24 July 2024.
Edited by:
Jianqiang Xu, Dalian University of Technology, ChinaReviewed by:
Maria Antonietta Barbieri, University of Messina, ItalyJing Peng, Affiliated Hospital of Jining Medical University, China
Copyright © 2024 Zhang, Ren, Zhu, Zheng, Shen and Lu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chengwu Shen, c2N3ODEwQDEyNi5jb20=; Cuicui Lu, Y2VjZTIwMjFAMTYzLmNvbQ==
†These authors have contributed equally to this work and share first authorship
 Xusheng Zhang
Xusheng Zhang Xiuli Ren1†
Xiuli Ren1† Cuicui Lu
Cuicui Lu