- 1College of Traditional Chinese Medicinal Material, Jilin Agricultural University, Changchun, China
- 2School of Medicine, Changchun Sci-Tech University, Changchun, China
- 3Jilin Provincial International Joint Research Center for the Development and Utilization of Authentic Medicinal Materials, Changchun, China
- 4Jilin Province Faw General Hospital, Changchun, China
Depression is a prevalent mental disorder that significantly diminishes quality of life and longevity, ranking as one of the primary causes of disability globally. Contemporary research has explored the potential pathogenesis of depression from various angles, encompassing genetics, neurotransmitter systems, neurotrophic factors, the hypothalamic-pituitary-adrenal axis, inflammation, and intestinal flora, among other contributing factors. In addition, conventional chemical medications are plagued by delayed onset of action, persistent adverse effects, and restricted therapeutic efficacy. In light of these limitations, the therapeutic approach of traditional Chinese medicine (TCM) has gained increasing recognition for its superior effectiveness. Numerous pharmacological and clinical studies have substantiated TCM’s capacity to mitigate depressive symptoms through diverse mechanisms. This article attempts to summarize the mechanisms involved in the pathogenesis of depression and to describe the characteristics of herbal medicines (including compounded formulas and active ingredients) for the treatment of depression. It further evaluates their effectiveness by correlating with the multifaceted pathogenesis of depression, thereby furnishing a reference for future research endeavors.
1 Introduction
Major Depressive Disorder (MDD), or depression for short, is a chronic, recurrent, and potentially life-threatening mental disorder that not only reduces people’s quality of life, but also imposes a heavy burden on individuals and their families in terms of health, finances, work, interpersonal relationships, etc., (Sim et al., 2015). MDD is characterized by symptoms such as low mood, cognitive impairment, anhedonia, worthlessness, social phobia, and suicide. Based on the most recent statistics provided by the World Health Organization, it is estimated that over 350 million individuals globally are afflicted by MDD, which accounts for approximately 5% of the global population, making it the fourth most common disease in the world (World Health Organization, 2022). Furthermore, under the influence of the new coronavirus pandemic, the prevalence of depression has risen by 28% (COVID-19 Mental Disorders Collaborators, 2021). In addition, the occurrence of MDD also significantly increases the risk of patients developing other diseases such as cardiovascular disease, stroke, Alzheimer’s disease, and epilepsy (Mathers and Loncar, 2006).
MDD is mainly treated with traditional antidepressants of the monoamine class (López-Muñoz and Alamo, 2009). Such drugs suffer from long onset of action, persistent side effects, and limited therapeutic efficacy (only 50% of patients achieve complete remission) (Deussing and Arzt, 2018) Therefore, it is particularly urgent to find antidepressant drugs that are effective and have fewer side effects. TCM is a natural product with a long history of medication, and it is widely accepted that it is safe and has few side effects. Screening antidepressant formulas and active ingredients from TCM is a promising alternative and may become a major trend in the treatment of MDD in the future. To facilitate research on MDD and the development of new antidepressants, we have compiled an overview of the pathogenesis of MDD and the corresponding therapeutic modalities offered by TCM. This compilation aims to provide a valuable reference for further studies in this field.
2 Hypotheses on the pathogenesis of MDD
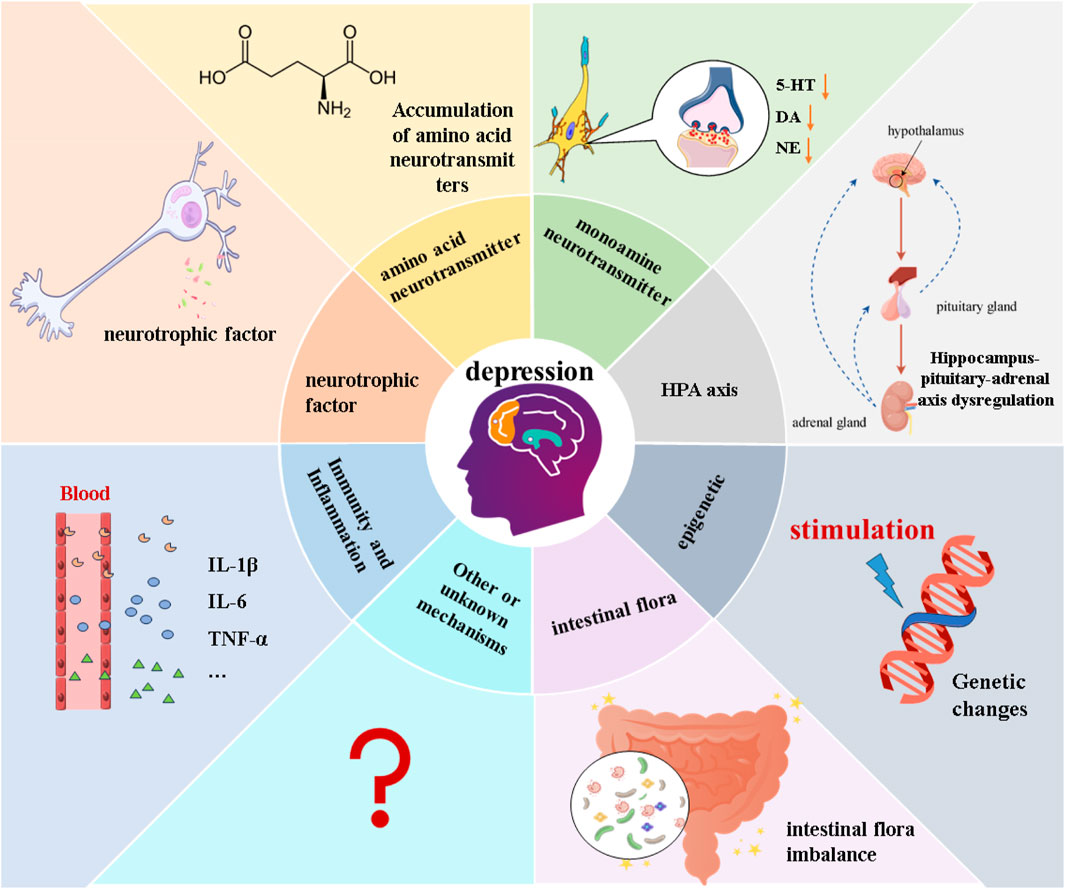
The etiology and pathogenesis of MDD are extremely complex, involving genetic, environmental, biochemical, and psychological factors (Jesulola et al., 2018). Despite numerous in-depth studies, there is no clear concept to explain the causes and mechanisms of MDD development. Based on this, hypotheses explaining the pathogenesis of MDD from various aspects have been proposed, such as epigenetic, monoamine neurotransmitters, Hypothalamus-Pituitary-Adrenal (HPA) axis, neuroplasticity and neurotrophic factors, intestinal microbiota, and inflammatory hypotheses (Dobrek and Głowacka, 2023). Figure 1.
3 Antidepressant theory and application of TCM
Chinese medical theory has a long history of understanding MDD-related diseases, and there are many MDD-related diseases recorded in classical Chinese medical texts, such as baihe disease, globus hystericus, renalmass, and depressive psychosis, which belong to the category of “yv zheng” in Chinese medical theory (Zhou, 2007; Yu et al., 2009). According to Chinese medicine theory, MDD is mostly characterized by emotional disorders and stagnation of qi, which leads to a loss of regulation of the liver, a loss of function of the spleen, and a loss of nourishment of the heart. There are internal and external causes for MDD, externally because of emotional factors such as worry, fear, and anger, and internally because the qi of the organs is easily disturbed. The disease is mainly located in the liver, involving the heart, spleen, and kidney; the disease mechanism is mainly stagnation of qi and dysfunction of internal organs. At the beginning of the disease, most of the evidence is solid, due to the stagnation of qi, resulting in food stagnation, phlegm coagulation, blood stasis; as the disease progresses from solid to virtual, it ultimately leads to deficiency of the five viscera, yin and yang imbalance. Common symptoms include liver stagnation, qi stagnation and fire, loss of heart and spirit, deficiency of heart and spleen, and phlegm and qi stagnation (Li and Liu, 2017).
With the continuous development of theoretical research on TCM and the advantages of low side effects and low recurrence rate of Chinese medicine, significant progress has been made in the research on TCM for MDD (Yeung et al., 2014). Currently, TCM antidepressant studies are available in the form of monomers isolated from TCM, individual Chinese medicine extracts, and formulas. In this review, we review and discuss the effects of TCM on depression through different mechanisms of action (Figure 2).
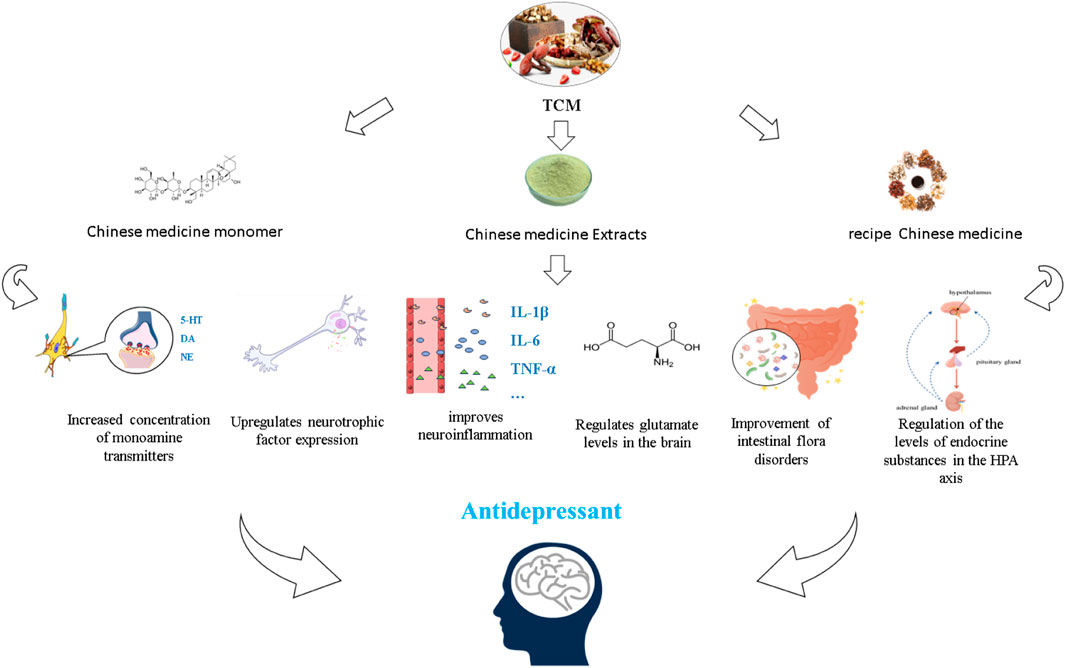
Figure 2. Different forms of TCM exert antidepressant effects based on the hypothesis of the pathogenesis of depression.
4 Substance basis of antidepressant efficacy and its mechanism of action in Chinese medicine
With the in-depth exploration of Chinese medicine, many active ingredients or parts with significant antidepressant effects have been found, which can be used as supplements to traditional antidepressants. They can affect depression by modulating monoamine transmitters, glutamate levels, the HPA axis, neurotrophic factors, the immune system, and gut flora.
4.1 By modulating monoamine transmitters
The monoamine hypothesis is the first theory in the history of MDD, and this hypothesis suggests that the possible pathogenesis of MDD is a decrease in monoamine neurotransmitter metabolism in the synaptic gap (Schildkraut et al., 1972). Monoamine neurotransmitters are an important class of excitatory neurotransmitters that include two major groups, catecholamines and indoleamines. Catecholamines include norepinephrine (NE) and dopamine (DA), and indoleamines are mainly serotonin (5-HT) (Jovanovic et al., 2022). Many functions in the body are regulated by the monoaminergic system. For instance, anxiety and fear disorders involve 5-HT, which is also related to pain sensitivity control and neurohormonal regulation (Bazian and Grigor’ian, 2006). NE and DA are associated with mood and eroticism, respectively, and are involved in the regulation of selective attentional processes, learning, locomotion, and reward activities (Goddard et al., 2010; Shen et al., 2012). Whereas the monoamine hypothesis is a classic prediction of depression, the underlying pathophysiology of depression is based on decreased levels of monoamine neurotransmitters in the synaptic gap (Delgado, 2000). It has also been found that drugs that act through various mechanisms to increase synaptic monoamine concentrations can improve symptoms of depression (Hirschfeld, 2000). However, it is important to note that the monoamine hypothesis is not without its criticisms. Some researchers argue that the hypothesis oversimplifies the complex etiology of depression and fails to account for other neurotransmitter systems, such as glutamate and GABA, which may also play a role in the development of MDD (Cryan et al., 2008). Additionally, not all individuals with depression respond positively to traditional monoamine-based antidepressant medications, suggesting that other factors may be at play in the development of depression (Pariante, 2017).
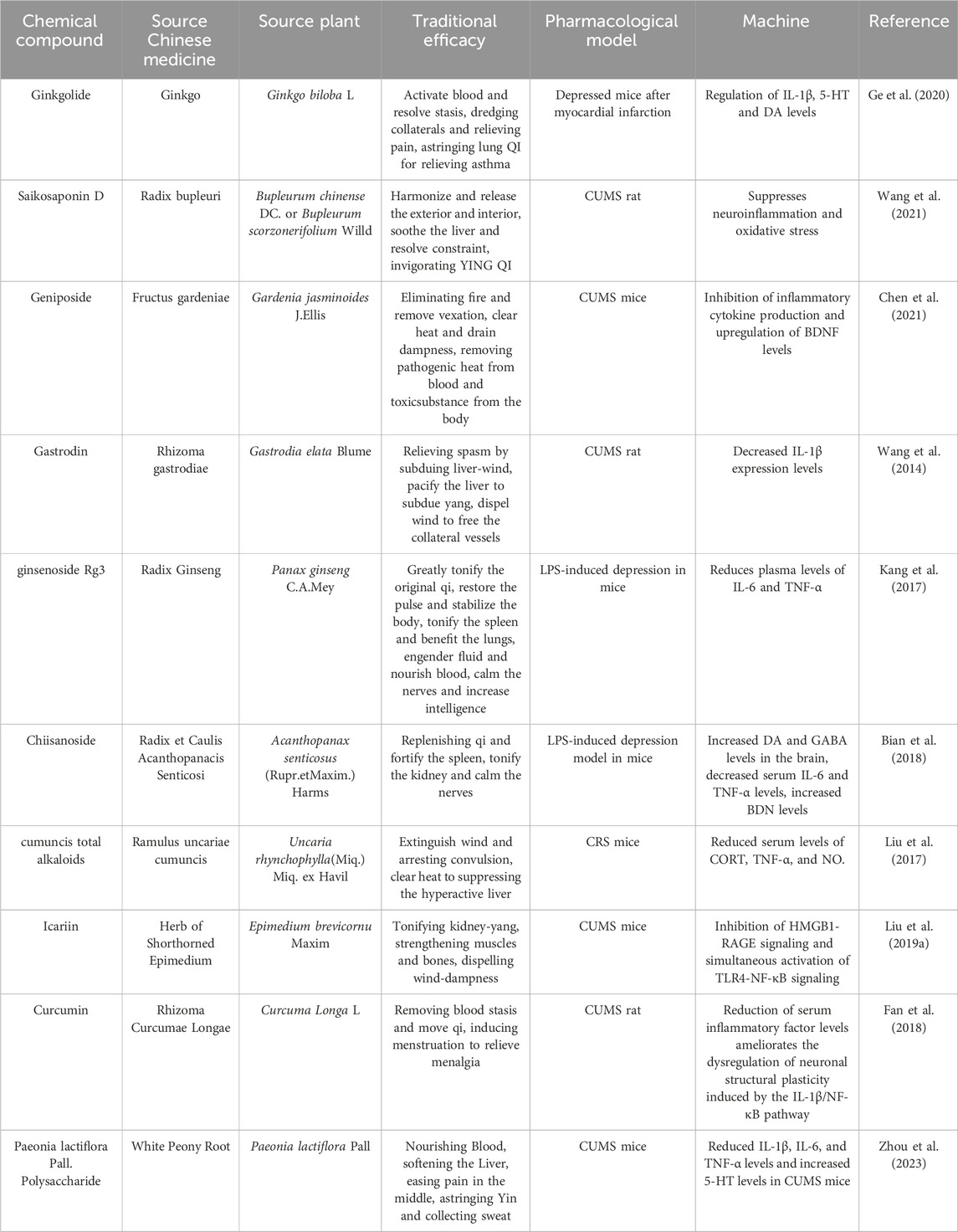
Overall, while the monoamine hypothesis has been influential in shaping our understanding of depression and guiding treatment approaches, it is important to continue to research and explore other potential mechanisms involved in the pathology of MDD in order to develop more effective treatments for this debilitating condition. Modulation of synaptic gap monoamine neurotransmitter concentrations and inhibition of monoamine oxidase may be one of the antidepressant mechanisms in Traditional Chinese Medicine (TCM) (Xin-xi, 2011). Research has shown that the active ingredients in TCM can improve the levels of different monoamine transmitters. For example, in a study involving a chronic mild stress (CMS) depressed mouse model, administration of ginkgolide at varying doses (3 mg/kg/d, 6 mg/kg/d, 12 mg/kg/d) was able to increase the levels of dopamine (DA) and serotonin (5-HT) in the hippocampal tissues of the mice and downregulate the levels of monoamine oxidase-A, thereby improving the depression-like behavior of the mice (Yang et al., 2020). Another active ingredient, crocin, found in the Chinese medicine saffron, was also studied in a chronic unpredictable stress (CUMS) mouse model, where crocin at a dosage of 25 mg/kg/d was found to alleviate depressive symptoms by modulating the metabolic level of tryptophan, the precursor of 5-hydroxytryptophan, as demonstrated in metabolomics studies (Luo et al., 2023). TCM active ingredients are known to regulate the levels of different monoamine transmitters through various channels, targets, and levels. For instance, Yuanzhi-1 was found to block synaptic reuptake of monoamine transmitters and simultaneously increase the levels of 5-HT, DA, and norepinephrine (NE) in the synaptic gap.
Monoamine oxidase inhibitors (MAOIs) were one of the first drugs used to treat depression but were phased out because of large side effects (Finberg, 2014). In contrast, a recent study found that curcumin at 50 mg/kg/d could improve the despairing behavior of rifampicin- and buprenorphine-induced mice by acting similarly to MAOIs, whereas curcumin did not have a direct activating effect on monoamines and did not have a significant inhibitory effect on the reuptake of monoamine neurotransmitters (Chen et al., 2006). More studies of herbal active ingredients or extracts to increase the inter-synaptic concentration of monoamines in response to depression are shown in Table 1.
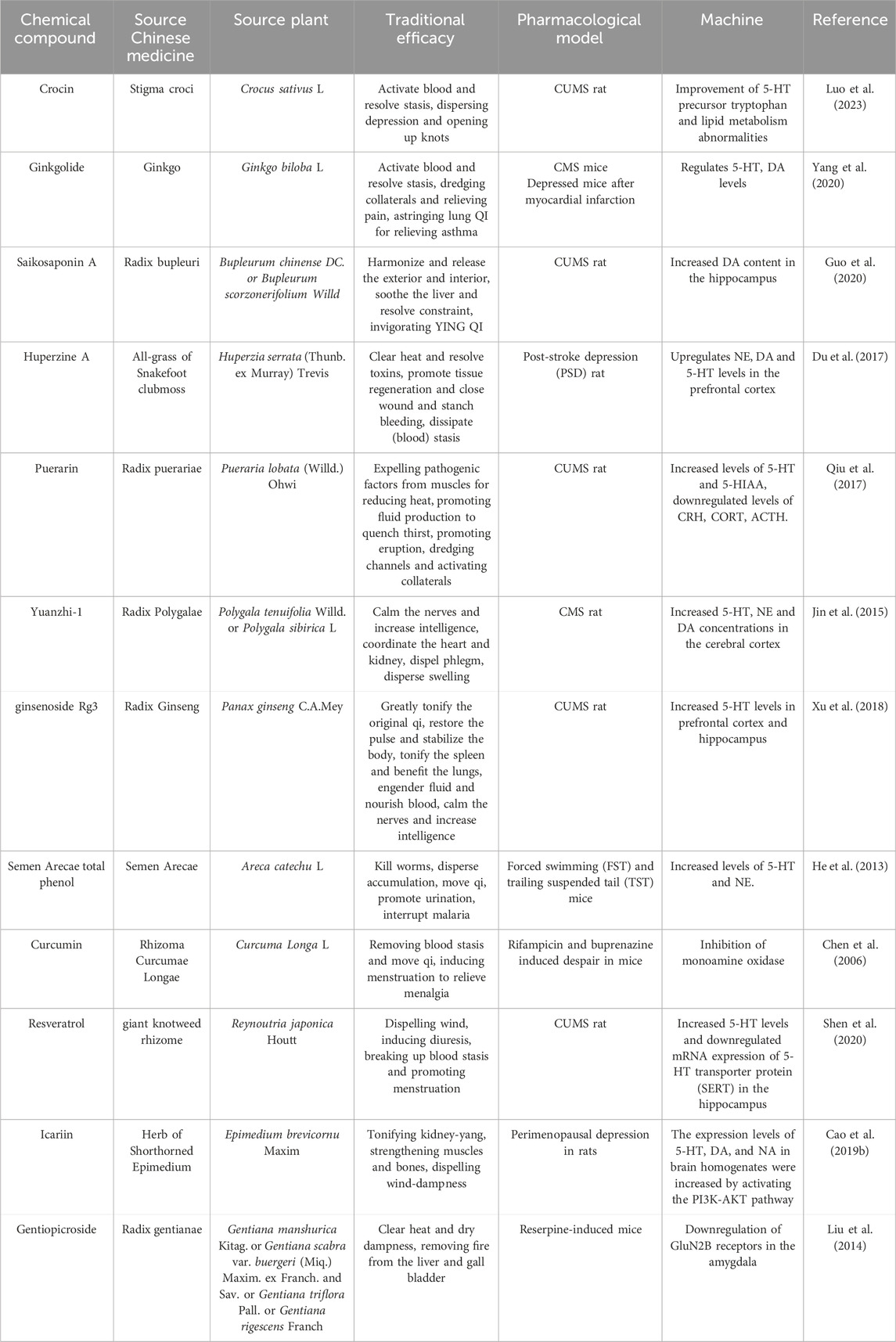
Table 1. Antidepressant mechanisms of Chinese herbal active ingredients modulating monoamine transmitter levels.
4.2 By regulating glutamatergic transmitter levels
Glutamate is the predominant excitatory neurotransmitter in the central nervous system and is involved in neuronal excitability, synaptic plasticity, and neurogenesis by activating various receptors, such as N-methyl-D-aspartic acid (NMDA) receptors, AMPA receptors, and metabotropic glutamate receptors (Orrego and Villanueva, 1993). Glutamate (Glu) and a small amount of another inhibitory amino acid neurotransmitter, gamma-aminobutyric acid (GABA), synergistically maintain the balance of excitatory/inhibitory circuits (Douglas and Martin, 2007; Sanacora et al., 2012). However, the balance of excitatory and inhibitory circuits has been found to be characteristically disturbed in MDD, mainly by dysfunction of the glutamatergic system, dysregulation of glutamatergic clearance, and metabolic regulatory mechanisms, which cause persistent glutamate accumulation. Structural and functional changes in regions related to mood and cognition and reduced synaptic activity are caused by excitotoxicity (e.g., excessive stimulation of glutamate receptors), which may lead to core symptoms of MDD, such as depressed mood, anhedonia, and cognitive dysfunction (Patel et al., 2015).
Glial glutamate transporter protein-1 (GLT-1) is one of the major Na-driven glutamate transporter proteins and plays an important role in maintaining control of synaptic glutamate concentration, and dysregulated expression of GLT-1 induces depressive symptoms (Weng et al., 2014). Schisantherin B is a lignan substance isolated from the traditional Chinese medicine Schisandra chinensis (Turcz.). Recent studies have shown that a single dose of Schisantherin B (15 mg/kg/d) can increase GLT-1 levels and alleviate symptoms of FST-induced depression in FST-induced depressed mice by promoting PI3K/AKT/mTOR pathways. Excessive accumulation of glutamate in brain tissues is a direct manifestation of the glutamate hypothesis of depression (Weng et al., 2014), so inhibiting excessive elevation of glutamate levels in the brain may be one of the approaches to treating depression with traditional Chinese medicines. Saikosaponin D and Gentiopicroside are the active ingredients in Chai Hu and Gentian, respectively, in which Saikosaponin D (1.50 mg/kg/d, 0.75 mg/kg/d) reduced glutamate levels in the CA1 region of the hippocampus of CUMS-exposed rats by modulating the Homer1-mGluR5 and downstream mTOR signaling pathways, and improved the behavioral performance of the rats in behavioral tests, proving its antidepressant effect (Liu et al., 2022). In contrast, Gentiopicroside (50 mg/kg/d, 100 mg/kg/d, 200 mg/kg/d) significantly reduced the immobility time during the forced swim test and increased the time in the center area in rifampicin-induced pain/depression dichotomy mice and the open-field test by down-regulating the GluN2B receptor in the amygdala, total distance traveled (Liu et al., 2014). More antidepressant mechanisms of herbal active ingredients by modulating glutamate transmitter levels are shown in Table 2.
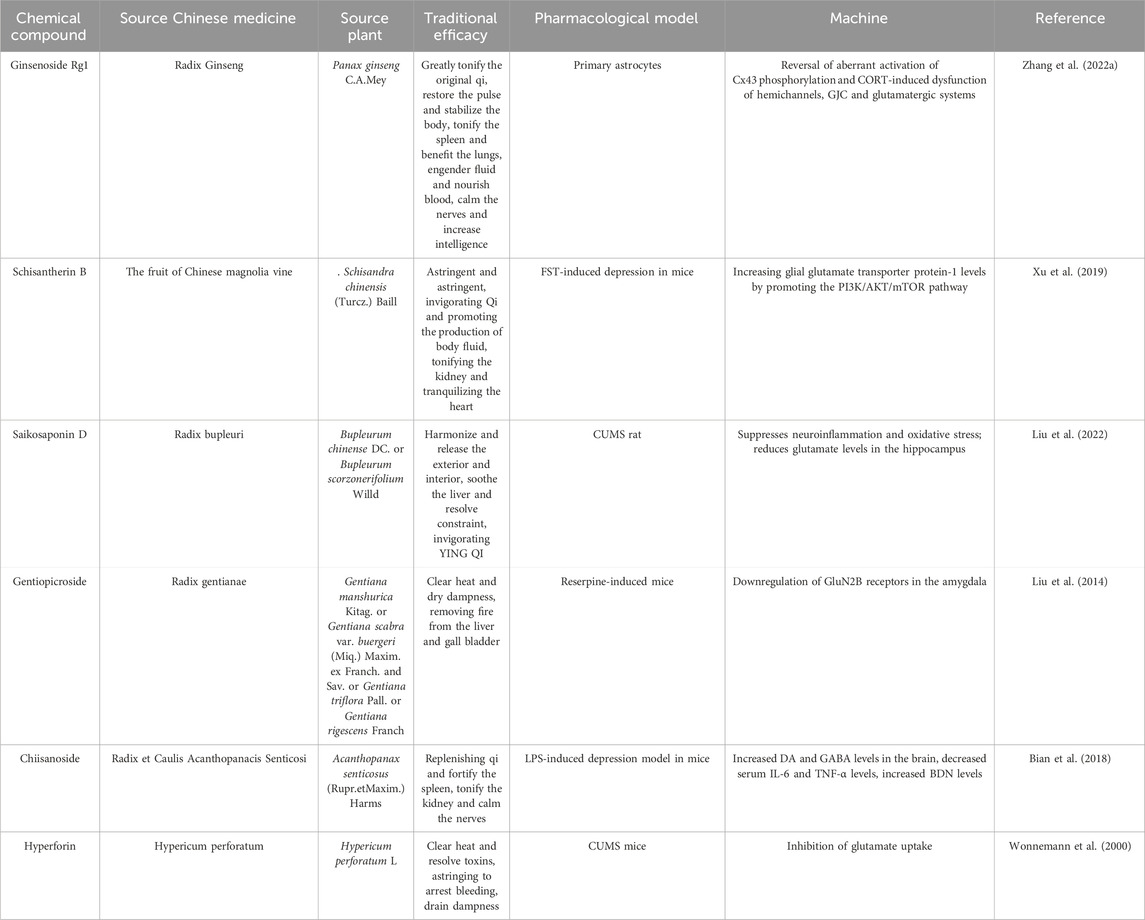
Table 2. Antidepressant mechanisms of Chinese herbal medicine active ingredients regulating glutamate levels.
In summary, abnormalities in the glutamatergic system are one of the recognized hypotheses in the pathogenesis of depression. Although there are fewer studies on the antidepressant effects of Chinese herbal medicines through glutamate, there is indeed some evidence of the effects of the active ingredients of Chinese herbal medicines on the glutamatergic system in the depressive process. This calls for more research to find the exact mechanism in order to develop new approaches and drugs in the treatment of depression.
4.3 By improving HPA axis dysfunction
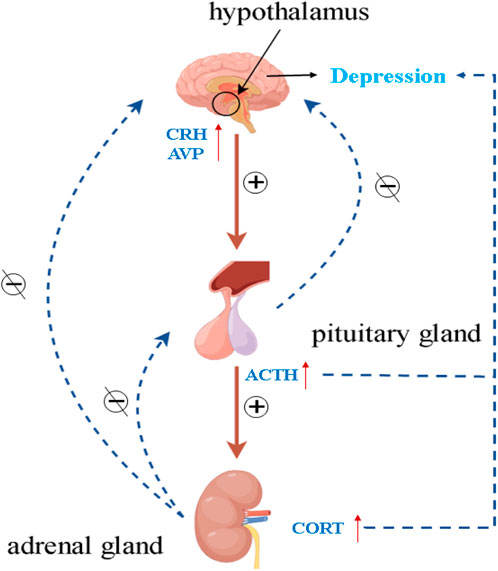
Various mood and cognitive disorders have been associated with the pathophysiology of the HPA axis. The HPA axis may be hyperactive in MDD patients, as studies have demonstrated (Keller et al., 2017). The regulation of the HPA axis primarily occurs through the influence of the hippocampus, which governs the secretion of corticotropin-releasing hormone (CRH) and arginine vasopressin (AVP) from the hypothalamic paraventricular nucleus (PVN). The anterior pituitary is stimulated by CRH to produce adrenocorticotropic hormone (ACTH), which then prompts the adrenal cortex to generate and release glucocorticoids (cortisol in humans and corticosterone in rodents) into the blood. Exogenous or endogenous stress may impair certain functions in the HPA axis, such as glucocorticoid resistance, i.e., dysfunction of the glucocorticoid receptor (GR), which impairs the negative feedback it mediates, and cause the pituitary gland and adrenal glands to enlarge, resulting in the disruption of the overstimulated HPA axis and increased levels of cortisol (CORT) in patients with depression (Figure 3) (Anacker et al., 2011; Nikkheslat et al., 2020). Therefore, the search for active ingredients of herbal medicines that can improve hippocampus-pituitary-adrenal axis dysfunction is a focus that requires urgent attention. Encouragingly, a large number of studies have been reported to elucidate the active ingredients of herbal medicines to alleviate depression by modulating the HPA axis, as well as their mechanisms of action.
Icariin, a flavonoid constituent isolated from Epimedium, was investigated in a chronic social defeat stress (CSDS)-induced mouse model of depression. The results showed that oral administration of 25 mg/kg and 50 mg/kg of Icariin reversed the CSDS-induced reduction in the duration of social interactions and attenuated the downregulation of serum GR and the increase in corticosterone levels in mice. The overexpression of CRH and ACTH as well as chronically elevated serum levels of CORT may contribute to the depressive-like behaviors (Vincent and Jacobson, 2014). Salidroside is a key active ingredient isolated from Rhodiola rosea. Salidroside, a key active ingredient isolated from Rhodiola rosea, was studied in a rat model of olfactory bulbectomy-induced depression. The results showed that oral administration of Salidroside (20 mg/kg/d/d, 40 mg/kg/d/d) significantly shortened the immobilization time of FST and TST and significantly increased the expression of GR and brain-derived neurotrophic factor (BDNF). In addition, Salidroside attenuated the expression of hypothalamic CRH and significantly reduced serum corticosterone levels. Suggesting that the antidepressant activity possessed by Salidroside may be mediated primarily by ameliorating abnormal HPA axis function (Yang et al., 2014). Rats were and exposed to a series of unpredictable mild stressors to establish a CUMS depression model, and after 19 days of gavage administration of Puerarin (a flavonoid glycoside extracted from the traditional Chinese medicine Pueraria lobata), it was found that concentrations of 60 and 120 mg/kg/d/d of Puerarin blocked the increase in serum CRH, CORT, and ACTH, implying that Puerarin’s antidepressant mechanism may be related to the regulation of HPA axis function (Qiu et al., 2017). The antidepressant mechanisms of the active ingredients of Chinese medicines by alleviating the dysfunction of the hypothalamic-pituitary-adrenal (HPA) axis are shown in Table 3.
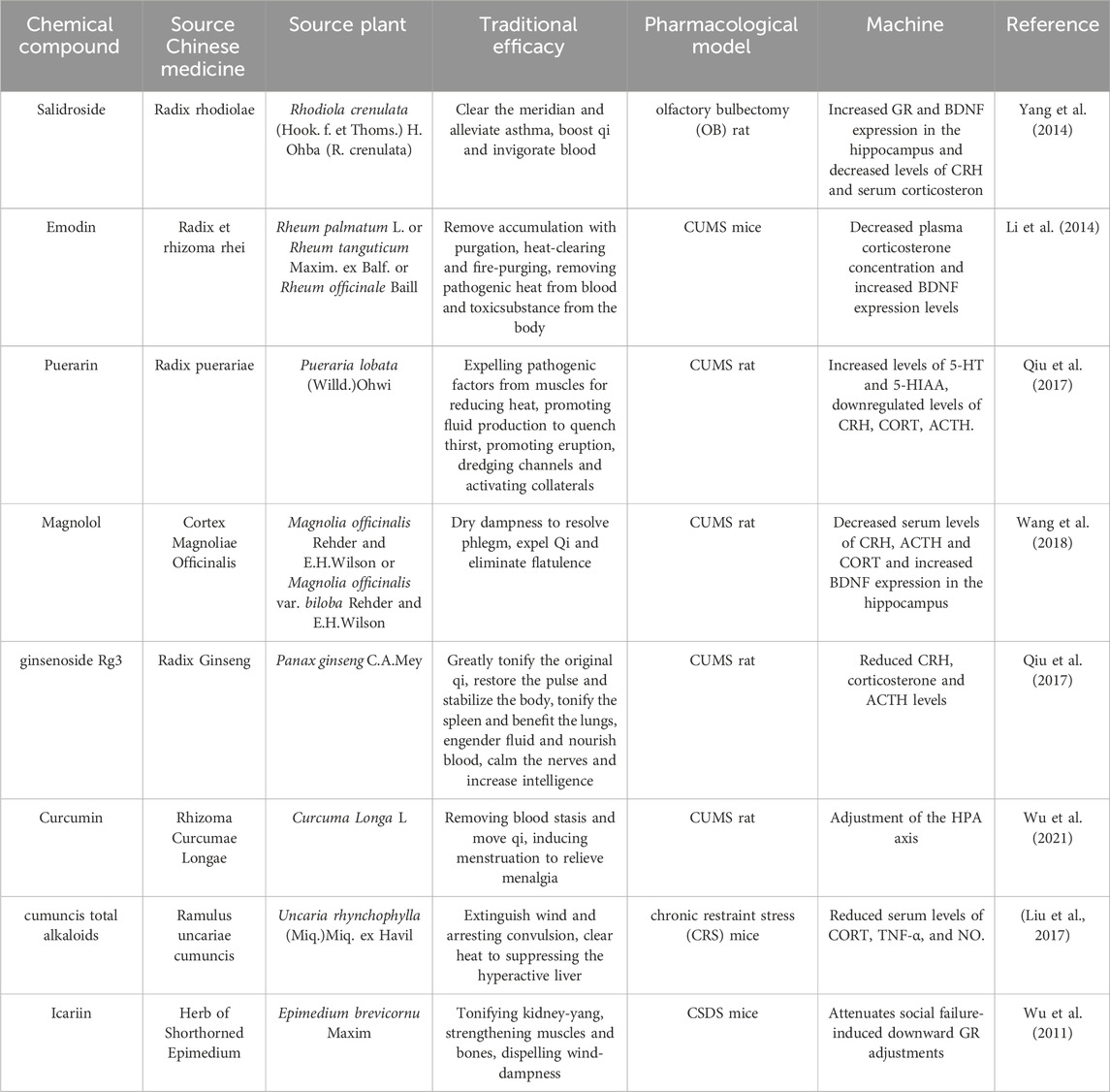
Table 3. Antidepressant mechanism of improving HPA axis dysfunction by Chinese herbal active ingredients.
4.4 By regulating neurotrophic factor levels
Neuroplasticity, the process of growth and adaptation at the neuronal level, plays a crucial role in the modification of neurogenesis and the morphology of mature neurons. However, restricted neurogenesis and disturbances in neuronal morphology can contribute to the development of MDD-like symptoms (Castrén, 2013; Allen and Lyons, 2018). Neuroplasticity, including synaptic plasticity, neurotransmission, neuronal survival, growth, and differentiation, is regulated by a combination of neurotrophic factors such as BDNF, glial cell line-derived neurotrophic factor (GDNF), vascular endothelial growth factor (VEGF), and nerve growth factor (NGF) (Song et al., 2017). These factors work synergistically to enhance and support neuroplasticity processes (Thoenen, 1995; Wang et al., 2022). Therefore, modulation of neurotrophic factor levels may be the direction of herbal medicine to improve depression.
There has been a large body of evidence demonstrating the effects of active ingredients in herbal medicines on the levels of neurotrophic factors. For example, resveratrol, a natural polyphenol, was found to improve CUMS-induced depressive-like behavior and cognitive deficits in mice by activating the Sirt1/miR-134 pathway and upregulating downstream cAMP response element-binding protein (CREB)/BDNF levels in the hippocampus (Shen et al., 2018). Long-term administration of ginsenoside Rg1 (40 mg/kg/d for 5 weeks) significantly ameliorated neuronal structural abnormalities and biochemical changes induced by CUMS stress and prevented depressive-like behaviors in rats with CUMS, which may be based on the mechanism of neuroprotective and antidepressant-like effects exhibited through activation of the CREB-BDNF system in the basolateral amygdala (Zhu et al., 2016). Ethanolic extracts of Dendrobium flowers increased the expression of NGF and BDNF at both transcriptional and protein levels, decreased immobilization time and increased sucrose consumption in a forced swimming test in mice (Zhu et al., 2021). It suggests that the antidepressant effects of the Dendrobium flowers may be related to the modulation of neurotrophic factor levels. Table 4 exhibits other Central Asian active ingredients and extracts that target neurotrophic factors.
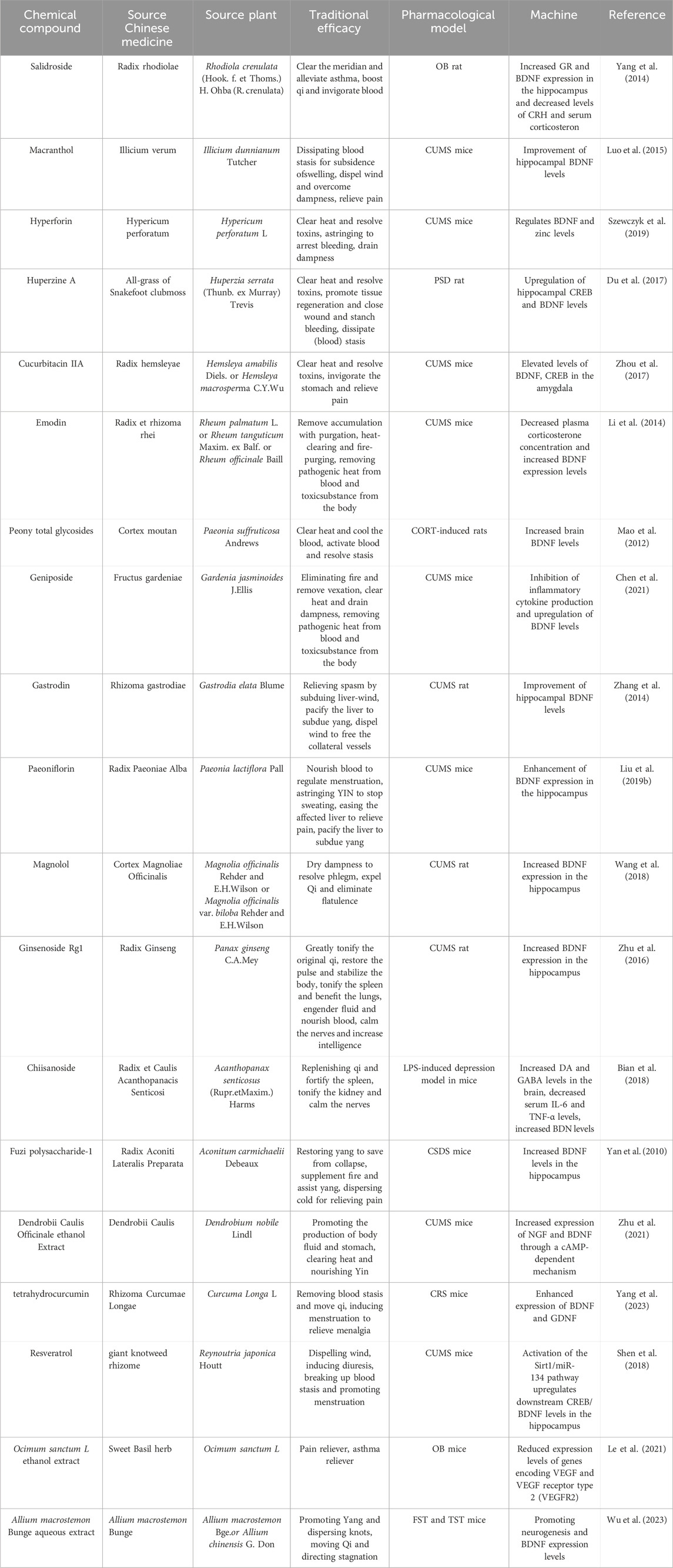
Table 4. Antidepressant mechanism of Chinese herbal medicine active ingredients regulating the level of neurotrophic factor.
4.5 By improving inflammation
In the past few decades, a growing body of evidence has highlighted the involvement of systemic immune activation and inflammatory processes in the development of MDD (Maes, 1999; Gibney and Drexhage, 2013). Inflammation, which arises from the activation of the immune system, typically manifests as a localized response triggered by irritation, injury, or infection. However, when dysregulated, immune cells and mechanisms within the body can contribute to the onset of diseases like MDD (Murphy et al., 2007). There is evidence that patients with depression have elevated levels of inflammatory cytokines such as tumor necrosis factor alpha (TNF-α), interleukin 1β (IL-1β), and interleukin 6 (IL-6) (Beurel et al., 2020). This may indicate the presence of inflammatory processes in patients with MDD.
Using LPS-induced depressed mice, after gavage administration of Chiisanoside (5.0 mg/kg/d), it effectively reduced serum IL-6 and TNF-α levels, as well as improved oxidative stress-related indices, and significantly increased immobility time of TST and FST, suggesting that Chiisanoside may improve depression by modulating the levels of inflammatory factors. Under stress, NF-κB is activated to promote the production of IL-1β proximate (Bian et al., 2018). Fan et al. (2018) found that pretreatment with Curcumin (40 mg/kg/d) for 5 weeks ameliorated the depressive-like effects and suppressed the inflammatory response and neuronal structural abnormalities in CUMS-exposed mice, with a mechanism of action similar to that of IL-1β or NF-κB antagonists. There is evidence that High Mobility Group Box 1 (HMGB1) can play an important role in neurodegenerative diseases by mediating neuroinflammation (Chi et al., 2015). Icariin was found to promote HMGB1 translocation to the nucleus and, on the one hand, inhibit HMGB1-RAGE anti-inflammatory signaling and, on the other hand, may activate TLR4-NF-κB signaling and increase p65 expression in the nucleus to promote nerve regeneration, effectively improving social avoidance behavior in the social interaction test and time spent in the central area in the open field test in rats (Liu L. et al., 2019). Other studies involving the improvement of immune and inflammatory dysregulation mechanisms are shown in Table 5.
4.6 By regulating the intestinal flora
The human gut flora is a diverse and complex ecosystem consisting of trillions of microorganisms, including bacteria, viruses, archaea, and fungi (Kc et al., 2020), that have a role in human health. Gut flora participate in a bidirectional communication pathway with the central nervous system (CNS), known as the microbiota-gut-brain axis (Dinan and Cryan, 2017; Chang et al., 2022). In contrast, dysbiosis disrupts the brain-gut-microbiota axis, induces a neuroimmune inflammatory response, disrupts the function of the intestinal mucosa and the blood-brain barrier, and directly stimulates the vagus nerve, enteric nervous system, spinal nerves, and HPA axis. These interactions have been implicated in the development of neurological disorders, including MDD (Tang and Cao, 2021).
As the link between gut flora and the onset of depression becomes more deeply understood, more and more studies are finding that herbs can improve depression by regulating gut flora. Patchouli essential oil, the main active ingredient in patchouli, is used in aromatherapy for stress relief. Oral administration of patchouli essential oil (0.8 mL/kg) significantly attenuated the depression-like behavior induced by CUMS stress in mice in FST. It was found to increase the relative abundance of several probiotics (including Bacteroides and Blautia) and improve the levels of metabolites short-chain fatty acids (SCFAs) and restore hippocampal 5-HT levels regarding the gut microbiota as analyzed by 16S rRNA gene sequencing (Ouyang et al., 2024). In addition, changes in gut flora affect the immune system and generate an inflammatory response, which in turn induces or exacerbates the depressive response (Peirce and Alviña, 2019). The establishment of a CUMS rat model of depression significantly increased the abundance of beneficial bacteria (Lactobacillus and Oscillospira) in the rat gut after the intervention of administering a certain amount of Astragaloside IV, as well as modulating the imbalance of Th17/Treg cells and the abnormal levels of anti-inflammatory and pro-inflammatory factors. This resulted in improved depression-like behavior in rats, as evidenced by weight gain, upregulation of sucrose preference, and decreased immobility time. This implies that Astragaloside IV is able to improve the inflammatory response by regulating intestinal flora, which in turn reduces depressive symptoms (Liu et al., 2024). We have compiled a list of active ingredients of some Chinese herbal medicines (Table 6), which have antidepressant effects by modulating the intestinal flora.
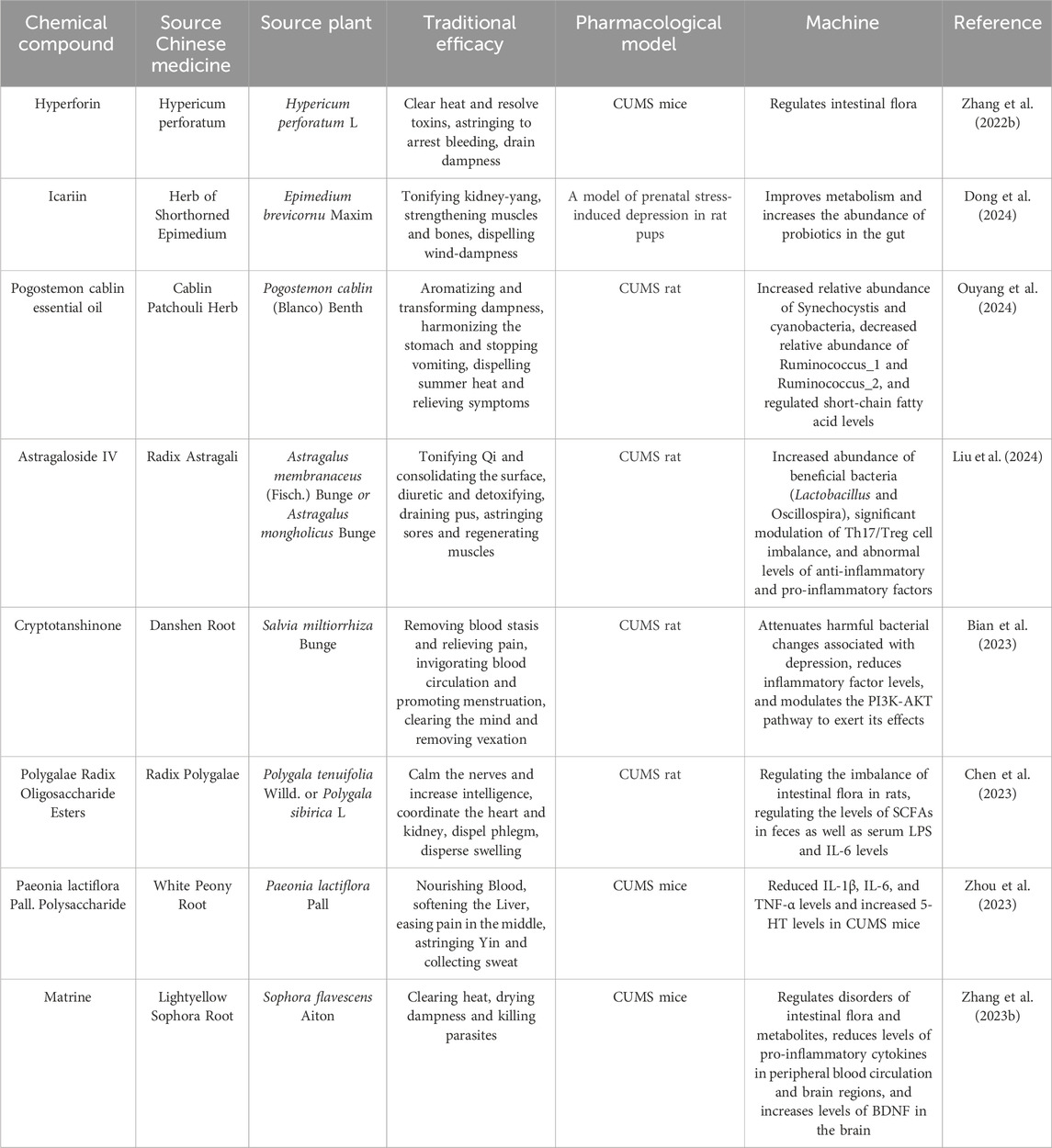
Table 6. Antidepressant mechanism of Chinese herbal medicine active ingredients regulating intestinal flora.
In summary, intestinal flora-associated depression has received much attention in recent years. As the interactions and relationships between changes in intestinal flora and depression become clearer and clearer, the mechanism of antidepressant effects of traditional Chinese medicines by regulating intestinal flora will continue to be revealed.
5 Application of classical and famous Chinese medicine formulas in antidepressants
Depression has a complex and diverse etiology and pathogenesis, and single-targeted therapeutic strategies with traditional antidepressants may be ineffective. In contrast, Chinese herbal formulas, with their systemic properties and multi-targeting characteristics, may provide advantages for the treatment and prevention of depression. In traditional Chinese medicine, Chinese herbal formulas are commonly used drug therapies, consisting of a combination of multiple Chinese medicines, providing a material basis for the diversity of antidepressant mechanisms. They can adapt to the characteristics of the diversity of causative factors and the complexity of lesions in depression through the integration of the regulating effects of multi-link, multi-level, and multi-targets. At the same time, different Chinese medicines can synergistically enhance antidepressant efficacy through mutual promotion and enhancement (Zhuang et al., 2023).
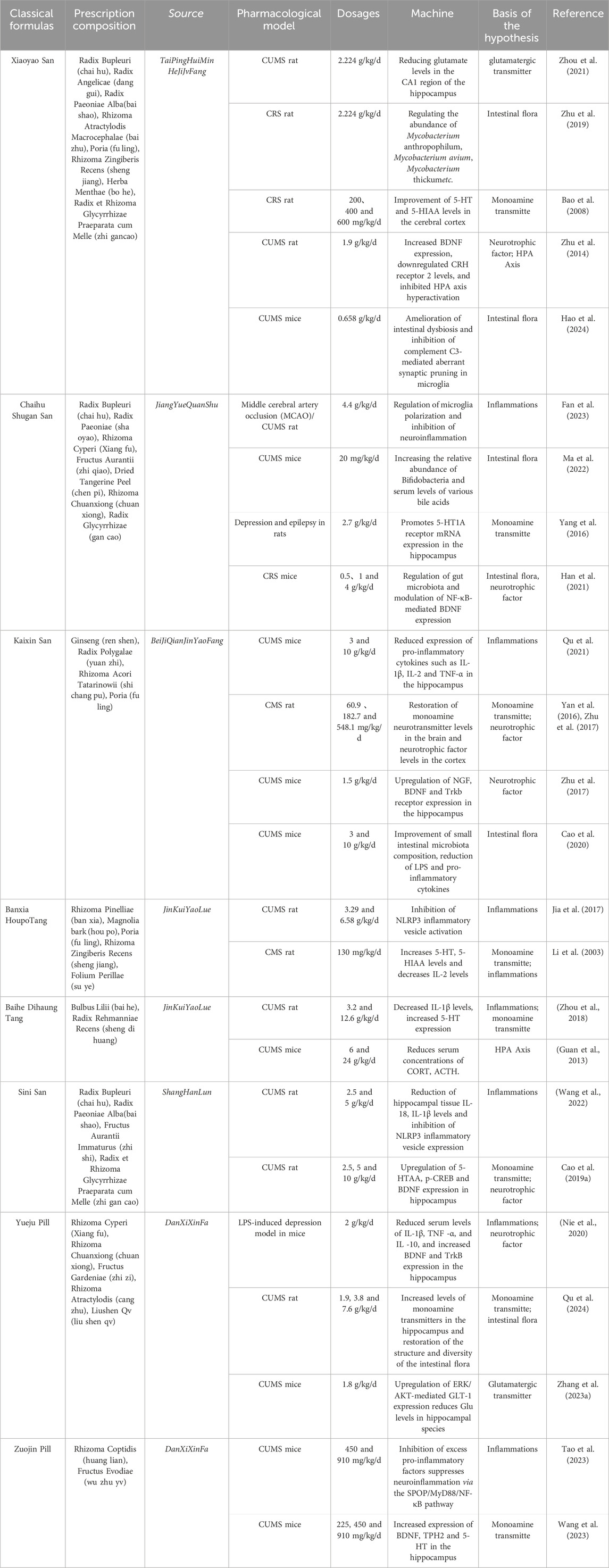
In the records of traditional Chinese medicine, many Chinese herbal formulas have been proposed and applied to treat depression (“yu-shen” in Chinese medicine theory). Some of the classical formulas are hundreds or even thousands of years old and are still used in current clinical practice. In this paper, we summarize eight classic TCM formulas (Table 7), which have been shown to have multiple mechanisms of action and targets. Among them, Xiaoyao San (XYS), Chaihu Shuogan San (CHSGS), and Kaixin San (KXS) have received strong attention for their remarkable efficacy. We will focus on a systematic review of these three classic prescriptions.
5.1 XYS
XYS is a classic formula in TCM, which was first developed in the Song Dynasty (960–1127 A.D.) from the “TaiPingHuiMin HeJiJvFang” and has been used in the treatment of psychiatric disorders for thousands of years. XYS is made up of eight commonly used Chinese herbs: Chai Hu, Dang Gui, Bai Shao, Bai Zhu, Fu Ling, Sheng Jiang, Bo He, and Zhi Gan Cao. In recent years, a large number of studies have demonstrated that XYS can exert antidepressant effects through various mechanisms. Zhou et al. (2021) found that glutamate and serum CORT levels were abnormally high in the hippocampal CA1 region of CUMS-induced depressed rats, and that the administration of XYS (2.224 g/kg/d) significantly reduced glutamate and CORT levels, and improved the depression-like symptoms of the rats. Moreover, the antidepressant effect of XYS may be exerted through the NR2B and PI3K/Akt signaling pathway. In order to treat MDD, Zhu et al. (2019) explored the effects of XYS on depressive behaviors. A CRS rat model of MDD was established, and a 16S rDNA high-throughput method was used to sequence fecal specimens to detect the structure and changes of intestinal flora. The results showed that at the phylum level, XYS regulated the abundance of Bacteroidetes, Proteobacteria, Firmicutes, Chloroflexi, and Planctomycetes. At the genus level, XYS decreased the abundance of Prevotellaceae_Ga6A1_group, Prevotellaceae_UCG-001, and Desulfovibrio, and increased the abundance of Ruminococcaceae in order to improve depression-like behavior. It was also found that this process involves mechanisms that may be related to short-chain fatty acids, lipopolysaccharides, and intestinal inflammation. Recent studies have found that dysbiosis of gut flora can induce the development of depressive-like behaviors through abnormal synaptic pruning of microglia mediated by complement C3, and that XYS was able to inhibit abnormal synaptic pruning by regulating gut flora to improve depressive-like behaviors in CUMS mice (Hao et al., 2024). In addition, other studies have shown that XYS can also ameliorate MDD by modulating monoamine neurotransmitters, BDNF, and neuroendocrine levels (Bao et al., 2008; Zhu et al., 2019).
5.2 CHSGS
CHSGS was first introduced in the classic medical text “JingYueQuanShu” from the Ming Dynasty. It is composed of Chai Hu, Shao Yao, Xiang Fu, Zhi Ke, Chen Pi, Chuan Xiong, and Gan Cao. In Chinese medicine, it is used to relieve liver qi stagnation. Modern research on it has found that CHSGS is able to alleviate MDD by regulating intestinal flora, reducing inflammation, and increasing monoamine transmitter levels. For example, post-stroke MDD was modeled by subjecting male rats to middle cerebral artery occlusion and chronic unpredictable mild stress. CHSGS (4.4 g/kg/d) was found to modulate microglia polarization by activating the JAK/STAT3-GSK3β/PTEN/Akt pathway, suggesting that CHSGS can exert antidepressant effects by inhibiting neuroinflammation (Fan et al., 2023). Ma et al. (2022) transplanted gut microbes from CHSGS-treated mice into untreated CUMS mice, restored serum levels of hyocholic acid and 7-ketodeoxycholic acid in depressed mice, increased BDNF levels, and alleviated depression-like symptoms. The comorbidity of epilepsy and MDD is common in neuropsychiatry (Alhashimi et al., 2022). A rat model of depressive epilepsy was established by inducing epilepsy with chronic hairy fructosamine followed by exposure to chronic mild stress, and it was found that CHSGS (2.7 g/kg/d) increased the expression of 5-HT1A receptor mRNA and cell proliferation in the hippocampal dentate gyrus of depressive epileptic rats, which was effective in ameliorating the depressive symptoms (Yang et al., 2016).
5.3 KXS
KXS was initially described in the ancient Chinese book “BeiJiQianJinYaoFang” written by Sun Simiao in the Tang Dynasty around the year 652 AD. Initially used to treat dementia and forgetfulness, it is composed of ginseng, Yuan Zhi, shi chang pu, and fu ling. In traditional Chinese medicine theory, MDD is believed to be caused by qi, dampness, and phlegm, ultimately leading to brain dysfunction (Pei et al., 2020). KXS functions to tonify qi, nourish the heart, expel dampness, and resolve phlegm, thus aiding in the treatment of MDD. More and more research indicates that KXS can effectively treat MDD by regulating neurotrophic factors, neurotransmitters, gut microbiota, and inflammation (Fu et al., 2019). In a preclinical study using fluoxetine (7.2 mg/kg/d) as a positive control, administering KXS extract orally at doses of 3 or 10 g/kg/d significantly improved depressive-like behaviors in chronic unpredictable mild stress (CUMS) rats in sucrose preference, forced swimming, and tail suspension tests, thereby alleviating symptoms of anhedonia, despair, and anxiety. Additionally, KXS inhibits the activation of microglial cells and significantly reduces the expression of pro-inflammatory cytokines such as IL-1β, IL-2, and TNF-α in the mouse hippocampus by inhibiting the TLR4/IKK/NF-κB pathway (Qu et al., 2021). KXS also helps regulate neurotrophic factor metabolic pathways; after oral administration of KXS (1.5 g/kg/d) daily for several days, it significantly alleviates depressive symptoms in mice subjected to chronic unpredictable mild stress, as evidenced by increased sucrose consumption, reduced immobility time in forced swimming, and increased locomotor activity. It was found that KXS achieves this effect by upregulating the expression of NGF, BDNF, and Trkb receptors in the hippocampus, which was confirmed by treatment with respective inhibitors tPA-stop and K252a (Zhu et al., 2017).
6 Discussion
MDD’s pathogenesis is multifaceted and convoluted. Despite the fact that present studies have shed light on its possible pathogenesis from various angles, the extent to which each pathway contributes differs among individuals and still fails to explain the full range of pathogenic mechanisms. Based on this dilemma, new treatments are scarce, and the mechanisms studied are relatively homogeneous, lacking integration and systematic research. MDD is a disease with multiple pathways and factors, which requires in-depth study of the interactions among pathogenic mechanisms and exploration of the intrinsic linkages and molecular pathways through which various pathogenic factors affect each other, rather than just following the causal logic of the association.
With the development of TCM, TCM has received more and more attention due to its outstanding therapeutic effect and fewer side effects. TCM natural monomers are also supplements to traditional antidepressants to minimize adverse effects. These single components have higher biological activity and are more likely to enter cells to exert medicinal effects. They have advantages of fast onset of action and low side effects. In addition, as summarized in this paper, it was found that, compared with traditional synthetic drugs, many of the active ingredients of Chinese medicines do not have a single antidepressant mechanism and can achieve antidepressant effects through multiple pathways. For example, Icariin and Curcumin are able to act on MDD through a variety of mechanisms, such as neurotrophic factor, HPA axis, and inflammation. This may be an advantage of the natural active ingredients of herbal medicine instead of traditional antidepressant drugs. Overall, the natural active ingredients in TCM provide a promising direction for the development of novel antidepressant drugs.
Formulas are one of the most commonly used drug therapies in TCM, especially the classical formulas, which have been in use for a long time and have clear therapeutic effects. To this day, they are still widely used in clinical practice, and a range of herbal combinations provide the material basis for diverse antidepressant mechanisms. However, due to the complexity of the ingredients, current studies have mainly focused on their overall effects on MDD, and the exact relationship between the active ingredients in the formula and the pathogenesis of MDD has rarely been reported. It is also not clear how the multiple ingredients synergize with each other to treat MDD through different mechanisms. In addition, there are many traditional antidepressant formulas in China, and the antidepressant effects of some of them have not yet been fully explored, including many classical formulas that have been determined to have significant antidepressant effects but have been neglected due to a lack of research. Therefore, more comprehensive and in-depth studies are needed to elucidate the exact active ingredients in classical prescriptions and their molecular pharmacology related to the prevention and treatment of MDD.
In conclusion, this review summarizes the latest information and insights from research on the use of herbal medicines in the treatment of MDD. Much evidence suggests that the antidepressant effects of TCM are definitive and have great potential for development, but the research does contain shortcomings that may seriously constrain the development of novel antidepressants. Firstly, the hypothesis of the pathogenesis of MDD mentioned above still does not provide an adequate explanation for the nature of MDD, although current studies have elaborated the possible pathogenesis of MDD from different perspectives. At the same time, the insufficiency of basic mechanism research also restricts the deeper study of TCM antidepressants, and it is not known through which different unknown pathways TCM can still act on MDD. Secondly, TCM antidepressant studies have mainly focused on the traditional mechanisms of monoamine transmitters, HPA axis, and neurotrophic factors, and the effects of Glu and brain-gut axis on the occurrence of MDD have been well demonstrated by the studies. However, the studies of TCM in treating MDD through these pathways are still lacking in reports. In addition, compared with other drugs that have been widely used, most of the studies on TCM still remain in the preclinical research stage, and there is a lack of clinical trials to elucidate the exact antidepressant effects of TCM.
Therefore, to address the above problems, further development of basic mechanism studies is urgently needed, with a view to developing a unified understanding of the etiology and pathogenesis of MDD, exploring the antidepressant effects of TCM in depth and comprehensively, in order to elucidate the nature of MDD and the moderating effects of TCM, and providing a scientific basis for new means of treating MDD.
Author contributions
WD: Writing–original draft, Writing–review and editing. LW: Writing–review and editing. LL: Data curation, Writing–review and editing. HL: Visualization, Writing–review and editing. JaW: Visualization, Writing–review and editing. JZ: Conceptualization, Supervision, Writing–review and editing. JnW: Supervision, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This research is supported by Jilin Science and Technology Development Program Project (20240404007YY).
Acknowledgments
The author would like to thank Figdraw for their support in making the map.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
5-HT, serotonin; ACTH, adrenocorticotropic hormone; AVP, arginine vasopressin; BDNF, brain-derived neurotrophic factor; CMS, chronic mild stress; CNS, central nervous system; CORT, cortisol; CRH, corticotropin-releasing hormone; CRS, chronic restraint stress; CSDS, chronic socially frustrating stress; CUMS, chronic unpredictable stress; DA, dopamine; FST, forced swimming; GABA, gamma-aminobutyric acid; GDNF, glial cell line-derived neurotrophic factor; GLT-1, Glial glutamate transporter protein-1; GR, glucocorticoid recepto; HPA, Hypothalamus-Pituitary-Adrenal; IL-1, interleukin-1; IL-6, interleukin-6; MCAO, middle cerebral artery occlusion; MDD, Major Depressive Disorder/Depression; MS, maternal separation; NE, norepinephrine; NGF, nerve growth factor; NMDA, N-methyl-D-aspartic acid OB, olfactory bulbectomy; PSD, post-stroke depression; PVN, paraventricular nucleus; SCFAs, short-chain fatty acids; TCM, Traditional Chinese Medicine; TNF-α, Tumor necrosis factor-alpha; TrkB, tyrosine kinase B; TST, trailing suspended tail; VEGF, vascular endothelial growth factor.
References
Alhashimi, R., Thoota, S., Ashok, T., Palyam, V., Azam, A. T., Odeyinka, O., et al. (2022). Comorbidity of epilepsy and depression: associated pathophysiology and management. Cureus 14 (1), e21527. doi:10.7759/cureus.21527
Allen, N. J., and Lyons, D. A. (2018). Glia as architects of central nervous system formation and function. Science 362 (6411), 181–185. doi:10.1126/science.aat0473
Anacker, C., Zunszain, P. A., Carvalho, L. A., and Pariante, C. M. (2011). The glucocorticoid receptor: pivot of depression and of antidepressant treatment? Psychoneuroendocrinology 36 (3), 415–425. doi:10.1016/j.psyneuen.2010.03.007
Bao, L., Chen, J., Huang, L., Chen, W., Lin, Q., Yao, X. S., et al. (2008). Effects of Xiaoyao Wan on the behavioral despair and stress depression mice. Zhong Yao Cai 31 (9), 1360–1364. doi:10.13863/j.issn1001-4454.2008.09.027
Bazian, A. S., and Grigor'ian, G. A. (2006). Molecular-chemical basis of emotional states and reinforcement. Usp. Fiziol. Nauk. 37 (1), 68–83.
Beurel, E., Toups, M., and Nemeroff, C. B. (2020). The bidirectional relationship of depression and inflammation: double trouble. Neuron 107 (2), 234–256. doi:10.1016/j.neuron.2020.06.002
Bian, L. H., Wang, S. Q., Li, W. J., Li, J., Yin, Y., Ye, F. F., et al. (2023). Cryptotanshinone regulates gut microbiota and PI3K-AKT pathway in rats to alleviate CUMS induced depressive symptoms. Biomed. Pharmacother. 169, 115921. doi:10.1016/j.biopha.2023.115921
Bian, X., Liu, X., Liu, J., Zhao, Y., Li, H., Cai, E., et al. (2018). Study on antidepressant activity of chiisanoside in mice. Int. Immunopharmacol. 57, 33–42. doi:10.1016/j.intimp.2018.02.007
Cao, C., Liu, M., Qu, S., Huang, R., Qi, M., Zhu, Z., et al. (2020). Chinese medicine formula Kai-Xin-San ameliorates depression-like behaviours in chronic unpredictable mild stressed mice by regulating gut microbiota-inflammation-stress system. J. Ethnopharmacol. 261, 113055. doi:10.1016/j.jep.2020.113055
Cao, K., Shen, C., Yuan, Y., Bai, S., Yang, L., Guo, L., et al. (2019a). SiNiSan ameliorates the depression-like behavior of rats that experienced maternal separation through 5-ht1a receptor/CREB/BDNF pathway. Front. Psychiatry 10, 160. doi:10.3389/fpsyt.2019.00160
Cao, L. H., Qiao, J. Y., Huang, H. Y., Fang, X. Y., Zhang, R., Miao, M. S., et al. (2019b). PI3K-AKT signaling activation and icariin: the potential effects on the perimenopausal depression-like rat model. Molecules 24 (20), 3700. doi:10.3390/molecules24203700
Castrén, E. (2013). Neuronal network plasticity and recovery from depression. JAMA Psychiatry 70 (9), 983–989. doi:10.1001/jamapsychiatry.2013.1
Chang, L., Wei, Y., and Hashimoto, K. (2022). Brain-gut-microbiota axis in depression: a historical overview and future directions. Brain Res. Bull. 182, 44–56. doi:10.1016/j.brainresbull.2022.02.004
Chen, Q., Jia, T., Wu, X., Chen, X., Wang, J., and Ba, Y. (2023). Polygalae radix oligosaccharide esters may relieve depressive-like behavior in rats with chronic unpredictable mild stress via modulation of gut microbiota. Int. J. Mol. Sci. 24 (18), 13877. doi:10.3390/ijms241813877
Chen, T., Liu, S., Zheng, M., Li, Y., and He, L. (2021). The effect of geniposide on chronic unpredictable mild stress-induced depressive mice through BTK/TLR4/NF-κB and BDNF/TrkB signaling pathways. Phytother. Res. 35 (2), 932–945. doi:10.1002/ptr.6846
Chen, W. X., Liu, L. P., Li, L., Lu, Y., Cheng, G., Li, N., et al. (2006). Study on the antidepressant effect of curcumin and its mechanism. New Chin. Med. Clin. Pharmacol. 17 (05), 317–320. doi:10.19378/j.issn.1003-9783.2006.05.001
Chi, W., Chen, H., Li, F., Zhu, Y., Yin, W., and Zhuo, Y. (2015). HMGB1 promotes the activation of NLRP3 and caspase-8 inflammasomes via NF-κB pathway in acute glaucoma. J. Neuroinflammation 12, 137. doi:10.1186/s12974-015-0360-2
COVID-19 Mental Disorders Collaborators (2021). Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet 398 (10312), 1700–1712. doi:10.1016/s0140-6736(21)02143-7
Delgado, P. L. (2000). Depression: the case for a monoamine deficiency. J. Clin. Psychiatry 61 (Suppl. 6), 7–11.
Deussing, J. M., and Arzt, E. (2018). P2X7 receptor: a potential therapeutic target for depression? Trends Mol. Med. 24 (9), 736–747. doi:10.1016/j.molmed.2018.07.005
Dinan, T. G., and Cryan, J. F. (2017). Brain-gut-microbiota Axis and mental health. Psychosom. Med. 79 (8), 920–926. doi:10.1097/psy.0000000000000519
Dobrek, L., and Głowacka, K. (2023). Depression and its phytopharmacotherapy-A narrative review. Int. J. Mol. Sci. 24 (5), 4772. doi:10.3390/ijms24054772
Dong, Y., Wang, L., Yang, M., Zhou, X., Li, G., Xu, K., et al. (2024). Effect of icariin on depressive behaviour in rat pups. Evidences for its mechanism of action by integrating network pharmacology, metabolomics and gut microbiota composition. Phytomedicine 126, 155422. doi:10.1016/j.phymed.2024.155422
Douglas, R. J., and Martin, K. A. (2007). Mapping the matrix: the ways of neocortex. Neuron 56 (2), 226–238. doi:10.1016/j.neuron.2007.10.017
Du, Y., Liang, H., Zhang, L., and Fu, F. (2017). Administration of Huperzine A exerts antidepressant-like activity in a rat model of post-stroke depression. Pharmacol. Biochem. Behav. 158, 32–38. doi:10.1016/j.pbb.2017.06.002
Fan, C., Song, Q., Wang, P., Li, Y., Yang, M., Liu, B., et al. (2018). Curcumin protects against chronic stress-induced dysregulation of neuroplasticity and depression-like behaviors via suppressing IL-1β pathway in rats. Neuroscience 392, 92–106. doi:10.1016/j.neuroscience.2018.09.028
Fan, Q., Liu, Y., Sheng, L., Lv, S., Yang, L., Zhang, Z., et al. (2023). Chaihu-Shugan-San inhibits neuroinflammation in the treatment of post-stroke depression through the JAK/STAT3-GSK3β/PTEN/Akt pathway. Biomed. Pharmacother. 160, 114385. doi:10.1016/j.biopha.2023.114385
Finberg, J. P. (2014). Update on the pharmacology of selective inhibitors of MAO-A and MAO-B: focus on modulation of CNS monoamine neurotransmitter release. Pharmacol. Ther. 143 (2), 133–152. doi:10.1016/j.pharmthera.2014.02.010
Fu, H., Xu, Z., Zhang, X. L., and Zheng, G. Q. (2019). Kaixinsan, a well-known Chinese herbal prescription, for alzheimer's disease and depression: a preclinical systematic review. Front. Neurosci. 13, 1421. doi:10.3389/fnins.2019.01421
Ge, Y., Xu, W., Zhang, L., and Liu, M. (2020). Ginkgolide B attenuates myocardial infarction-induced depression-like behaviors via repressing IL-1β in central nervous system. Int. Immunopharmacol. 85, 106652. doi:10.1016/j.intimp.2020.106652
Gibney, S. M., and Drexhage, H. A. (2013). Evidence for a dysregulated immune system in the etiology of psychiatric disorders. J. Neuroimmune Pharmacol. 8 (4), 900–920. doi:10.1007/s11481-013-9462-8
Goddard, A. W., Ball, S. G., Martinez, J., Robinson, M. J., Yang, C. R., Russell, J. M., et al. (2010). Current perspectives of the roles of the central norepinephrine system in anxiety and depression. Depress Anxiety 27 (4), 339–350. doi:10.1002/da.20642
Guan, J. Q., Sun, Y., and Chen, H. W. (2013). Effects of baihedihuang soup on a mouse model of depression. China J. Traditional Chin. Med. Pharm. 28 (06), 1875–1877. 1877.
Guo, J., Zhang, F., Gao, J., Guan, X., Liu, B., Wang, X., et al. (2020). Proteomics-based screening of the target proteins associated with antidepressant-like effect and mechanism of Saikosaponin A. J. Cell. Mol. Med. 24 (1), 174–188. doi:10.1111/jcmm.14695
Han, S. K., Kim, J. K., Park, H. S., Shin, Y. J., and Kim, D. H. (2021). Chaihu-Shugan-San (Shihosogansan) alleviates restraint stress-generated anxiety and depression in mice by regulating NF-κB-mediated BDNF expression through the modulation of gut microbiota. Chin. Med. 16 (1), 77. doi:10.1186/s13020-021-00492-5
Hao, W., Ma, Q., Wang, L., Yuan, N., Gan, H., He, L., et al. (2024). Gut dysbiosis induces the development of depression-like behavior through abnormal synapse pruning in microglia-mediated by complement C3. Microbiome 12 (1), 34. doi:10.1186/s40168-024-01756-6
He, J. Y., Huang, B., Xin, Z. T., Chen, J. T., Chen, X. Y., Chen, X. J., et al. (2013). Antidepressant effects of total phenols from areca seed. J. Chin. Med. Mater. 36 (08), 1331–1334. doi:10.13863/j.issn1001-4454.2013.08.035
Hirschfeld, R. M. (2000). History and evolution of the monoamine hypothesis of depression. J. Clin. Psychiatry 61 (Suppl. 6), 4–6.
Jesulola, E., Micalos, P., and Baguley, I. J. (2018). Understanding the pathophysiology of depression: from monoamines to the neurogenesis hypothesis model - are we there yet? Behav. Brain Res. 341, 79–90. doi:10.1016/j.bbr.2017.12.025
Jia, K. K., Zheng, Y. J., Zhang, Y. X., Liu, J. H., Jiao, R. Q., Pan, Y., et al. (2017). Banxia-houpu decoction restores glucose intolerance in CUMS rats through improvement of insulin signaling and suppression of NLRP3 inflammasome activation in liver and brain. J. Ethnopharmacol. 209, 219–229. doi:10.1016/j.jep.2017.08.004
Jin, Z. L., Gao, N., Li, X. R., Tang, Y., Xiong, J., Chen, H. X., et al. (2015). The antidepressant-like pharmacological profile of Yuanzhi-1, a novel serotonin, norepinephrine and dopamine reuptake inhibitor. Eur. Neuropsychopharmacol. 25 (4), 544–556. doi:10.1016/j.euroneuro.2015.01.005
Jovanovic, P., Wang, Y., Vit, J. P., Novinbakht, E., Morones, N., Hogg, E., et al. (2022). Sustained chemogenetic activation of locus coeruleus norepinephrine neurons promotes dopaminergic neuron survival in synucleinopathy. PLoS One 17 (3), e0263074. doi:10.1371/journal.pone.0263074
Kang, A., Xie, T., Zhu, D., Shan, J., Di, L., and Zheng, X. (2017). Suppressive effect of ginsenoside Rg3 against lipopolysaccharide-induced depression-like behavior and neuroinflammation in mice. J. Agric. Food Chem. 65 (32), 6861–6869. doi:10.1021/acs.jafc.7b02386
Kc, D., Sumner, R., and Lippmann, S. (2020). Gut microbiota and health. Postgrad. Med. 132 (3), 274. doi:10.1080/00325481.2019.1662711
Keller, J., Gomez, R., Williams, G., Lembke, A., Lazzeroni, L., Murphy, G. M., et al. (2017). HPA axis in major depression: cortisol, clinical symptomatology and genetic variation predict cognition. Mol. Psychiatry 22 (4), 527–536. doi:10.1038/mp.2016.120
Le, X. T., Nguyen, H. T., Nguyen, T. V., Pham, H. T. N., Nguyen, P. T., Nguyen, K. M., et al. (2021). Ocimum sanctum linn. Extract improves cognitive deficits in olfactory bulbectomized mice via the enhancement of central cholinergic systems and VEGF expression. Evid. Based Complement. Altern. Med. 2021, 6627648. doi:10.1155/2021/6627648
Li, J. M., Kong, L. D., Wang, Y. M., Cheng, C. H., Zhang, W. Y., and Tan, W. Z. (2003). Behavioral and biochemical studies on chronic mild stress models in rats treated with a Chinese traditional prescription Banxia-houpu decoction. Life Sci. 74 (1), 55–73. doi:10.1016/j.lfs.2003.06.030
Li, M., Fu, Q., Li, Y., Li, S., Xue, J., and Ma, S. (2014). Emodin opposes chronic unpredictable mild stress induced depressive-like behavior in mice by upregulating the levels of hippocampal glucocorticoid receptor and brain-derived neurotrophic factor. Fitoterapia 98, 1–10. doi:10.1016/j.fitote.2014.06.007
Li, Y. X., and Liu, X. Z. (2017). Research progress on traditional Chinese medicine treatment of depression. Chin. Med. Res. 30 (08), 78–80.
Liu, C. Y., Chen, J. B., Liu, Y. Y., Zhou, X. M., Zhang, M., Jiang, Y. M., et al. (2022). Saikosaponin D exerts antidepressant effect by regulating Homer1-mGluR5 and mTOR signaling in a rat model of chronic unpredictable mild stress. Chin. Med. 17 (1), 60. doi:10.1186/s13020-022-00621-8
Liu, L., Zhao, Z., Lu, L., Liu, J., Sun, J., and Dong, J. (2019a). Icariin and icaritin ameliorated hippocampus neuroinflammation via mediating HMGB1 expression in social defeat model in mice. Int. Immunopharmacol. 75, 105799. doi:10.1016/j.intimp.2019.105799
Liu, S. B., Zhao, R., Li, X. S., Guo, H. J., Tian, Z., Zhang, N., et al. (2014). Attenuation of reserpine-induced pain/depression dyad by gentiopicroside through downregulation of GluN2B receptors in the amygdala of mice. Neuromolecular Med. 16 (2), 350–359. doi:10.1007/s12017-013-8280-8
Liu, S. C., Hu, W. Y., Zhang, W. Y., Yang, L., Li, Y., Xiao, Z. C., et al. (2019b). Paeoniflorin attenuates impairment of spatial learning and hippocampal long-term potentiation in mice subjected to chronic unpredictable mild stress. Psychopharmacol. Berl. 236 (9), 2823–2834. doi:10.1007/s00213-019-05257-5
Liu, S. L., Xv, Y. X., Huang, Y. J., and Zeng, C. Q. (2017). Antidepressant effects of total alkaloids from Uncaria rhynchophylla on chronic restraint stress mice. J. Guangdong Pharm. Univ. 33 (01), 72–76. doi:10.16809/j.cnki.2096-3653.2016101703
Liu, X., Wang, S., Wu, X., Zhao, Z., Jian, C., Li, M., et al. (2024). Astragaloside IV alleviates depression in rats by modulating intestinal microbiota, T-immune balance, and metabolome. J. Agric. Food Chem. 72 (1), 259–273. doi:10.1021/acs.jafc.3c04063
López-Muñoz, F., and Alamo, C. (2009). Monoaminergic neurotransmission: the history of the discovery of antidepressants from 1950s until today. Curr. Pharm. Des. 15 (14), 1563–1586. doi:10.2174/138161209788168001
Luo, L., Liu, X. L., Li, J., Mu, R. H., Liu, Q., Yi, L. T., et al. (2015). Macranthol promotes hippocampal neuronal proliferation in mice via BDNF-TrkB-PI3K/Akt signaling pathway. Eur. J. Pharmacol. 762, 357–363. doi:10.1016/j.ejphar.2015.05.036
Luo, Y., Zhong, Z., Li, H., Wang, L., Guo, D., Dong, X., et al. (2023). Integrating serum metabolomics and network analysis to explore the antidepressant activity of crocin in rats with chronic unexpected mild stress-induced depression. Pharm. Biol. 61 (1), 1414–1430. doi:10.1080/13880209.2023.2246531
Ma, C., Yuan, D., Renaud, S. J., Zhou, T., Yang, F., Liou, Y., et al. (2022). Chaihu-shugan-san alleviates depression-like behavior in mice exposed to chronic unpredictable stress by altering the gut microbiota and levels of the bile acids hyocholic acid and 7-ketoDCA. Front. Pharmacol. 13, 1040591. doi:10.3389/fphar.2022.1040591
Maes, M. (1999). Major depression and activation of the inflammatory response system. Adv. Exp. Med. Biol. 461, 25–46. doi:10.1007/978-0-585-37970-8_2
Mao, Q. Q., Huang, Z., Ip, S. P., Xian, Y. F., and Che, C. T. (2012). Peony glycosides reverse the effects of corticosterone on behavior and brain BDNF expression in rats. Behav. Brain Res. 227 (1), 305–309. doi:10.1016/j.bbr.2011.11.016
Mathers, C. D., and Loncar, D. (2006). Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 3 (11), e442. doi:10.1371/journal.pmed.0030442
Murphy, K. P., Weaver, C. T., Mowat, A. M., Berg, L. J., Chaplin, D. D., Janeway, C. A. J., et al. (2007). Janeway's immunobiology.
Nie, C. Y., Wang, J. H., Zhang, H. L., Zhou, T., Sun, Y., Ge, R., et al. (2020). Mechanism of Yueju pill on LPS depression model mice. Lishizhen Med. Materia Medica Res. 31 (04), 774–778. doi:10.3969/j.issn.1008-0805.2020.04.002
Nikkheslat, N., McLaughlin, A. P., Hastings, C., Zajkowska, Z., Nettis, M. A., Mariani, N., et al. (2020). Childhood trauma, HPA axis activity and antidepressant response in patients with depression. Brain Behav. Immun. 87, 229–237. doi:10.1016/j.bbi.2019.11.024
Orrego, F., and Villanueva, S. (1993). The chemical nature of the main central excitatory transmitter: a critical appraisal based upon release studies and synaptic vesicle localization. Neuroscience 56 (3), 539–555. doi:10.1016/0306-4522(93)90355-j
Ouyang, P., Kang, D., You, W., Shen, X., Mo, X., and Liu, Y. (2024). Pogostemon cablin essential oil affects anxiety- and depressive-like behaviors and the gut microbiota in chronic unpredictable mild stress model rats. Front. Nutr. 11, 1303002. doi:10.3389/fnut.2024.1303002
Patel, D., Kharkar, P. S., and Nandave, M. (2015). Emerging roles of system [Formula: see text] antiporter and its inhibition in CNS disorders. Mol. Membr. Biol. 32 (4), 89–116. doi:10.3109/09687688.2015.1096972
Pei, H., Ma, L., Cao, Y., Wang, F., Li, Z., Liu, N., et al. (2020). Traditional Chinese medicine for alzheimer's disease and other cognitive impairment: a review. Am. J. Chin. Med. 48 (3), 487–511. doi:10.1142/s0192415x20500251
Peirce, J. M., and Alviña, K. (2019). The role of inflammation and the gut microbiome in depression and anxiety. J. Neurosci. Res. 97 (10), 1223–1241. doi:10.1002/jnr.24476
Qiu, Z. K., Zhang, G. H., Zhong, D. S., He, J. L., Liu, X., Chen, J. S., et al. (2017). Puerarin ameliorated the behavioral deficits induced by chronic stress in rats. Sci. Rep. 7 (1), 6266. doi:10.1038/s41598-017-06552-x
Qu, S., Liu, M., Cao, C., Wei, C., Meng, X. E., Lou, Q., et al. (2021). Chinese medicine formula kai-xin-san ameliorates neuronal inflammation of CUMS-induced depression-like mice and reduces the expressions of inflammatory factors via inhibiting TLR4/IKK/NF-κB pathways on BV2 cells. Front. Pharmacol. 12, 626949. doi:10.3389/fphar.2021.626949
Qu, Z., Wu, S., Zheng, Y., Bing, Y., Liu, X., Li, S., et al. (2024). Fecal metabolomics combined with metagenomics sequencing to analyze the antidepressant mechanism of Yueju Wan. J. Pharm. Biomed. Anal. 238, 115807. doi:10.1016/j.jpba.2023.115807
Sanacora, G., Treccani, G., and Popoli, M. (2012). Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neuropharmacology 62 (1), 63–77. doi:10.1016/j.neuropharm.2011.07.036
Schildkraut, J. J., Draskoczy, P. R., Gershon, E. S., Reich, P., and Grab, E. L. (1972). Catecholamine metabolism in affective disorders. IV. Preliminary studies of norepinephrine metabolism in depressed patients treated with amitriptyline. J. Psychiatr. Res. 9 (3), 173–185. doi:10.1016/0022-3956(72)90019-2
Shen, J., Xu, L., Qu, C., Sun, H., and Zhang, J. (2018). Resveratrol prevents cognitive deficits induced by chronic unpredictable mild stress: Sirt1/miR-134 signalling pathway regulates CREB/BDNF expression in hippocampus in vivo and in vitro. Behav. Brain Res. 349, 1–7. doi:10.1016/j.bbr.2018.04.050
Shen, J. D., Zhang, Y. W., Wang, B. Y., Bai, L., Lu, S. F., Zhu, L. L., et al. (2020). Effects of resveratrol on the levels of ATP, 5-HT and GAP-43 in the hippocampus of mice exposed to chronic unpredictable mild stress. Neurosci. Lett. 735, 135232. doi:10.1016/j.neulet.2020.135232
Shen, L. H., Liao, M. H., and Tseng, Y. C. (2012). Recent advances in imaging of dopaminergic neurons for evaluation of neuropsychiatric disorders. J. Biomed. Biotechnol. 2012, 259349. doi:10.1155/2012/259349
Sim, K., Lau, W. K., Sim, J., Sum, M. Y., and Baldessarini, R. J. (2015). Prevention of relapse and recurrence in adults with major depressive disorder: systematic review and meta-analyses of controlled trials. Int. J. Neuropsychopharmacol. 19 (2), pyv076. doi:10.1093/ijnp/pyv076
Song, M., Martinowich, K., and Lee, F. S. (2017). BDNF at the synapse: why location matters. Mol. Psychiatry 22, 1370–1375. doi:10.1038/mp.2017.144
Szewczyk, B., Pochwat, B., Muszyńska, B., Opoka, W., Krakowska, A., Rafało-Ulińska, A., et al. (2019). Antidepressant-like activity of hyperforin and changes in BDNF and zinc levels in mice exposed to chronic unpredictable mild stress. Behav. Brain Res. 372, 112045. doi:10.1016/j.bbr.2019.112045
Tang, Q., and Cao, L. (2021). Intestinal flora and neurological disorders. Sheng Wu Gong Cheng Xue Bao 37 (11), 3757–3780. doi:10.13345/j.cjb.210253
Tao, W., Su, K., Huang, Y., Lu, Z., Wang, Y., Yang, L., et al. (2023). Zuojinwan ameliorates CUMS-induced depressive-like behavior through inducing ubiquitination of MyD88 via SPOP/MyD88/NF-κB pathway. J. Ethnopharmacol. 312, 116487. doi:10.1016/j.jep.2023.116487
Thoenen, H. (1995). Neurotrophins and neuronal plasticity. Science 270 (5236), 593–598. doi:10.1126/science.270.5236.593
Vincent, M. Y., and Jacobson, L. (2014). Glucocorticoid receptor deletion from the dorsal raphé nucleus of mice reduces dysphoria-like behavior and impairs hypothalamic-pituitary-adrenocortical axis feedback inhibition. Eur. J. Neurosci. 39 (10), 1671–1681. doi:10.1111/ejn.12538
Wang, C., Gan, D., Wu, J., Liao, M., Liao, X., and Ai, W. (2018). Honokiol exerts antidepressant effects in rats exposed to chronic unpredictable mild stress by regulating brain derived neurotrophic factor level and hypothalamus-pituitary-adrenal Axis activity. Neurochem. Res. 43 (8), 1519–1528. doi:10.1007/s11064-018-2566-z
Wang, C. S., Kavalali, E. T., and Monteggia, L. M. (2022). BDNF signaling in context: from synaptic regulation to psychiatric disorders. Cell. 185 (1), 62–76. doi:10.1016/j.cell.2021.12.003
Wang, H., Zhang, R., Qiao, Y., Xue, F., Nie, H., Zhang, Z., et al. (2014). Gastrodin ameliorates depression-like behaviors and up-regulates proliferation of hippocampal-derived neural stem cells in rats: involvement of its anti-inflammatory action. Behav. Brain Res. 266, 153–160. doi:10.1016/j.bbr.2014.02.046
Wang, W., Zhou, Y. Y., Yv, X. M., Jiang, X. C., Tian, D. Z., and Xiao, M. (2022). Effects of sini powder on NLRP3 inflammasome and depression-like behavior in rats with depression. Chin. J. Exp. Traditional Med. Formulae 28 (12), 22–30. doi:10.13422/j.cnki.syfjx.20221002
Wang, Y., Huang, Y., Zhao, M., Yang, L., Su, K., Wu, H., et al. (2023). Zuojin pill improves chronic unpredictable stress-induced depression-like behavior and gastrointestinal dysfunction in mice via the theTPH2/5-HT pathway. Phytomedicine 120, 155067. doi:10.1016/j.phymed.2023.155067
Wang, Z., Li, J., Wu, W., Qi, T., Huang, Z., Wang, B., et al. (2021). Saikosaponin D rescues deficits in sexual behavior and ameliorates neurological dysfunction in mice exposed to chronic mild stress. Front. Pharmacol. 12, 625074. doi:10.3389/fphar.2021.625074
Weng, H. R., Gao, M., and Maixner, D. W. (2014). Glycogen synthase kinase 3 beta regulates glial glutamate transporter protein expression in the spinal dorsal horn in rats with neuropathic pain. Exp. Neurol. 252, 18–27. doi:10.1016/j.expneurol.2013.11.018
Wonnemann, M., Singer, A., and Müller, W. E. (2000). Inhibition of synaptosomal uptake of 3H-L-glutamate and 3H-GABA by hyperforin, a major constituent of St. John's Wort: the role of amiloride sensitive sodium conductive pathways. Neuropsychopharmacology 23 (2), 188–197. doi:10.1016/s0893-133x(00)00102-0
World Health Organization (2022). Depressive disorder (depression). Available at: https://www.who.int/news-room/fact-sheets/detail/depression.
Wu, J., Du, J., Xu, C., Le, J., Xu, Y., Liu, B., et al. (2011). Icariin attenuates social defeat-induced down-regulation of glucocorticoid receptor in mice. Pharmacol. Biochem. Behav. 98 (2), 273–278. doi:10.1016/j.pbb.2011.01.008
Wu, J., Wang, L., Cui, Y., Liu, F., and Zhang, J. (2023). Allii macrostemonis bulbus: a comprehensive review of Ethnopharmacology, phytochemistry and pharmacology. Molecules 28 (6), 2485. doi:10.3390/molecules28062485
Wu, Y., Sun, F., Guo, Y., Zhang, Y., Li, L., Dang, R., et al. (2021). Curcumin relieves chronic unpredictable mild stress-induced depression-like behavior through the PGC-1α/FNDC5/BDNF pathway. Behav. Neurol. 2021, 2630445. doi:10.1155/2021/2630445
Xin-xi, L. (2011). Research progress of traditional Chinese medicines in single use as antidepressants. Med. Recapitulate 17 (23), 3624–3627.
Xu, J. N., Chen, L. F., Su, J., Liu, Z. L., Chen, J., Lin, Q. F., et al. (2018). The anxiolytic-like effects of ginsenoside Rg3 on chronic unpredictable stress in rats. Sci. Rep. 8 (1), 7741. doi:10.1038/s41598-018-26146-5
Xu, M., Xiao, F., Wang, M., Yan, T., Yang, H., Wu, B., et al. (2019). Schisantherin B improves the pathological manifestations of mice caused by behavior desperation in different ages-depression with cognitive impairment. Biomol. Ther. Seoul. 27 (2), 160–167. doi:10.4062/biomolther.2018.074
Yan, H. C., Qu, H. D., Sun, L. R., Li, S. J., Cao, X., Fang, Y. Y., et al. (2010). Fuzi polysaccharide-1 produces antidepressant-like effects in mice. Int. J. Neuropsychopharmacol. 13 (5), 623–633. doi:10.1017/s1461145709990733
Yan, L., Hu, Q., Mak, M. S., Lou, J., Xu, S. L., Bi, C. W., et al. (2016). A Chinese herbal decoction, reformulated from Kai-Xin-San, relieves the depression-like symptoms in stressed rats and induces neurogenesis in cultured neurons. Sci. Rep. 6, 30014. doi:10.1038/srep30014
Yang, P., Li, L., Liu, X. J., Cai, X., Sun, M. Z., He, J. F., et al. (2016). Effect of Chaihu-Shugan-San on the mRNA expression of the 5-HT1A receptor and cellular proliferation in the hippocampus of epileptic rats with depression. Exp. Ther. Med. 11 (1), 124–130. doi:10.3892/etm.2015.2867
Yang, S. J., Yu, H. Y., Kang, D. Y., Ma, Z. Q., Qu, R., Fu, Q., et al. (2014). Antidepressant-like effects of salidroside on olfactory bulbectomy-induced pro-inflammatory cytokine production and hyperactivity of HPA axis in rats. Pharmacol. Biochem. Behav. 124, 451–457. doi:10.1016/j.pbb.2014.07.015
Yang, T., Wang, X., Liu, L., Chen, H., and Wang, N. (2020). Ginkgolide-Platinum(II) complex GPt(II) exhibits therapeutic effect on depression in mice via upregulation of DA and 5-HT neurotransmitters. Med. Sci. Monit. 26, e922052. doi:10.12659/msm.922052
Yang, Y., Yang, J., Ma, T., Yang, X., Yuan, Y., and Guo, Y. (2023). The role and mechanism of TGF-β1 in the antidepressant-like effects of tetrahydrocurcumin. Eur. J. Pharmacol. 959, 176075. doi:10.1016/j.ejphar.2023.176075
Yeung, W. F., Chung, K. F., Ng, K. Y., Yu, Y. M., Ziea, E. T., and Ng, B. F. (2014). A systematic review on the efficacy, safety and types of Chinese herbal medicine for depression. J. Psychiatr. Res. 57, 165–175. doi:10.1016/j.jpsychires.2014.05.016
Yu, L. Y., Wang, X. D., Yv, S., and Shi, L. W. (2009). Comparison of “bentun” in “nan jing” and “jin kui Yao lue”. Lishizhen Med. Materia Medica Res. 20 (11), 2848.
Zhang, B., Su, D., Song, Y., Li, H., Chen, C., Liao, L., et al. (2023a). Yueju volatile oil plays an integral role in the antidepressant effect by up-regulating ERK/AKT-mediated GLT-1 expression to clear glutamate. Fitoterapia 169, 105583. doi:10.1016/j.fitote.2023.105583
Zhang, M., Li, A., Yang, Q., Li, J., Zheng, L., Wang, G., et al. (2023b). Matrine alleviates depressive-like behaviors via modulating microbiota-gut-brain axis in CUMS-induced mice. J. Transl. Med. 21 (1), 145. doi:10.1186/s12967-023-03993-z
Zhang, N., Jiang, H., Wang, H., Wang, Y., Peng, Y., Liu, Y., et al. (2022a). Novel antidepressant mechanism of ginsenoside Rg1 in regulating the dysfunction of the glutamatergic system in astrocytes. Int. J. Mol. Sci. 24 (1), 575. doi:10.3390/ijms24010575
Zhang, R., Peng, Z., Wang, H., Xue, F., Chen, Y., Wang, Y., et al. (2014). Gastrodin ameliorates depressive-like behaviors and up-regulates the expression of BDNF in the hippocampus and hippocampal-derived astrocyte of rats. Neurochem. Res. 39 (1), 172–179. doi:10.1007/s11064-013-1203-0
Zhang, Z., Yao, C., Li, M., Wang, L. C., Huang, W., and Chen, Q. J. (2022b). Prophylactic effects of hyperforin on anhedonia-like phenotype in chronic restrain stress model: a role of gut microbiota. Lett. Appl. Microbiol. 75 (5), 1103–1110. doi:10.1111/lam.13710
Zhou, S. M., Guan, S. Y., Yang, L., Yang, L. K., Wang, L., Nie, H. F., et al. (2017). Cucurbitacin IIa exerts antidepressant-like effects on mice exposed to chronic unpredictable mild stress. Neuroreport 28 (5), 259–267. doi:10.1097/wnr.0000000000000747
Zhou, X. (2007). Exploration of the name and visceral location of baihe disease. Liaoning J. Traditional Chin. Med. 13 (07), 901–902. doi:10.13192/j.ljtcm.2007.07.41.zhoux.022.houx.022
Zhou, X. L., Xiao, B. Y., Wang, H. L., and Tan, T. (2018). Effects of baihe Dihuang Tang on changes in the levels of inflammatory factor IL-1β and neurotransmitter 5-HT in rats with depression model. Guid. J. Traditional Chin. Med. Pharm. 24 (16), 30–33. doi:10.13862/j.cnki.cn43-1446/r.2018.16.009
Zhou, X. M., Liu, C. Y., Liu, Y. Y., Ma, Q. Y., Zhao, X., Jiang, Y. M., et al. (2021). Xiaoyaosan alleviates hippocampal glutamate-induced toxicity in the CUMS rats via NR2B and PI3K/akt signaling pathway. Front. Pharmacol. 12, 586788. doi:10.3389/fphar.2021.586788
Zhou, Z., Wang, Y., Sun, S., Zhang, K., Wang, L., Zhao, H., et al. (2023). Paeonia lactiflora Pall. Polysaccharide alleviates depression in CUMS mice by inhibiting the NLRP3/ASC/Caspase-1 signaling pathway and affecting the composition of their intestinal flora. J. Ethnopharmacol. 316, 116716. doi:10.1016/j.jep.2023.116716
Zhu, H. Z., Liang, Y. D., Ma, Q. Y., Hao, W. Z., Li, X. J., Wu, M. S., et al. (2019). Xiaoyaosan improves depressive-like behavior in rats with chronic immobilization stress through modulation of the gut microbiota. Biomed. Pharmacother. 112, 108621. doi:10.1016/j.biopha.2019.108621
Zhu, X., Gao, R., Liu, Z., Cheng, Z., Qi, Y., Fan, C., et al. (2016). Ginsenoside Rg1 reverses stress-induced depression-like behaviours and brain-derived neurotrophic factor expression within the prefrontal cortex. Eur. J. Neurosci. 44 (2), 1878–1885. doi:10.1111/ejn.13255
Zhu, X., Xia, O., Han, W., Shao, M., Jing, L., Fan, Q., et al. (2014). Xiao Yao san improves depressive-like behavior in rats through modulation of β-arrestin 2-mediated pathways in Hippocampus. Evid. Based Complement. Altern. Med. 2014, 902516. doi:10.1155/2014/902516
Zhu, Y., Chao, C., Duan, X., Cheng, X., Liu, P., Su, S., et al. (2017). Kai-Xin-San series formulae alleviate depressive-like behaviors on chronic mild stressed mice via regulating neurotrophic factor system on hippocampus. Sci. Rep. 7 (1), 1467. doi:10.1038/s41598-017-01561-2
Zhu, Y., Liu, M., Cao, C., Qu, S., Zheng, J., Zhu, Z., et al. (2021). Dendrobium officinale flos increases neurotrophic factor expression in the hippocampus of chronic unpredictable mild stress-exposed mice and in astrocyte primary culture and potentiates NGF-induced neuronal differentiation in PC12 cells. Phytother. Res. 35 (5), 2665–2677. doi:10.1002/ptr.7013
Keywords: depression, pathogenesis hypothesis, antidepressant potential of traditional Chinese medicine, herbal monomers, classical Chinese medicine prescriptions
Citation: Ding W, Wang L, Li L, Li H, Wu J, Zhang J and Wang J (2024) Pathogenesis of depression and the potential for traditional Chinese medicine treatment. Front. Pharmacol. 15:1407869. doi: 10.3389/fphar.2024.1407869
Received: 27 March 2024; Accepted: 05 June 2024;
Published: 25 June 2024.
Edited by:
Wenda Xue, Nanjing University of Chinese Medicine, ChinaCopyright © 2024 Ding, Wang, Li, Li, Wu, Zhang and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jing Zhang, emhqaW5nMDcwMUAxNjMuY29t; Jing Wang, NzI2NzM1MUBxcS5jb20=
†These authors have contributed equally to this work
 Weixing Ding
Weixing Ding Lulu Wang
Lulu Wang Lei Li1
Lei Li1 Hongyan Li
Hongyan Li Jianfa Wu
Jianfa Wu