- 1Department of Traditional Chinese Medicine, Jiading Branch of Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- 2Medical Experiment Center, Jiading Branch of Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- 3The Affiliated Cancer Hospital of Zhengzhou University and Henan Cancer Hospital, Zhengzhou, China
- 4Oncology Department, Yueyang Hospital of Integrated of Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai, China
- 5Jiading Branch of Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- 6Center for Research and Graduate Studies, University of Cyberjaya, Selangor, Malaysia
Classic Formulas (Jing fang) are considered the essence and authority of Traditional Chinese Medicine (TCM) due to their long history and proven efficacy. These formulas play a pivotal role in all kinds of different disease prevention and therapeutic strategies. Yiyi Fuzi Baijiang San (YYFZBJS), one of the Classic Formulas, was originally developed for the treatment of chronic intestinal abscess. With the accumulation of clinical experience and the exploration of modern pharmacological research in recent years, YYFZBJS has been extensively employed to address a broad spectrum of conditions such as colorectal cancer. Although numerous studies have explored the clinical efficacy and underlying mechanisms of YYFZBJS, no comprehensive review summarizing these findings exists to date. This study aims to systematically review and critically assess the current clinical and mechanistic research on YYFZBJS, with the objective of providing valuable insights and guidance for TCM research in the future.
1 Introduction
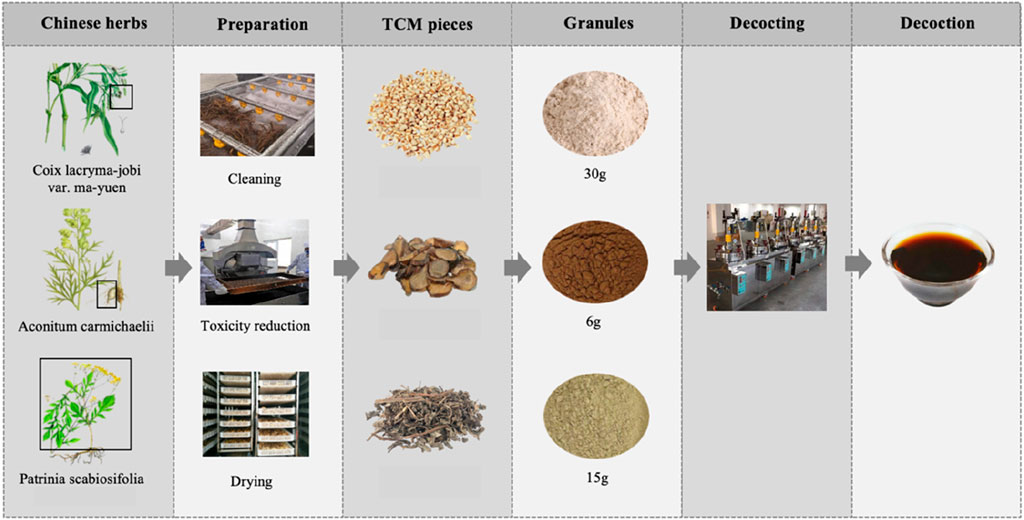
Traditional Chinese Medicine (TCM), with its millennia-long history, is widely practiced in China and increasingly gaining recognition worldwide. Unlike modern medicine, TCM is distinguished by its approach to diagnosis and treatment, which emphasizes syndrome differentiation and holistic principles. By integrating clinical symptoms with an understanding of the body’s internal environment, TCM seeks to restore balance and address the underlying causes of illness. This holistic perspective informs its therapeutic strategies, providing a nuanced approach to prevention and treatment (Oravecz and Mészáros, 2012). YYFZBJS, one of the Classic Formulas first recorded in the ancient Chinese medical text Synopsis of Prescriptions of the Golden Chamber. It comprises three botanical drugs: Coix lacryma-jobi L. var. mayuen (Roman.) Stapf. (Coix seed), Aconitum carmichaeli Debx. (Radix Aconiti Lateralis), and Patrinia villosa (Thunb.) Juss. (Patrinia villosa). These botanical drugs undergo a process of washing, detoxification, drying, powdering, and are then decocted in the proportions of 30 g: 6 g: 15 g to produce a medicinal solution typically administered orally (Twice daily, 200 mL each time) (Figure 1). According to the original documentation, YYFZBJS was originally used to treat chronic intestinal abscess resulting from persistent pathogenic factors including yang deficiency, cold, dampness and blood stasis. With advancements in modern pharmacological research, the clinical applications of YYFZBJS are gradually expanding to various areas including tumor treatment. Previous research has elucidated several potential pharmacological mechanisms of YYFZBJS and its components in anti-inflammation, immune regulation, and disease prevention. Nevertheless, due to challenges such as its complex composition, the full scope of its clinical applications and underlying mechanisms remains unclear. This article provides a comprehensive review of its pharmacological effects and clinical applications, with the hope to deepen understanding and encourage further research on classical ancient prescriptions in TCM.
2 Review methodology
To investigate the clinical efficacy and underlying mechanisms of YYFZBJS, we conducted a comprehensive search of articles in PubMed, Web of Science, and China National Knowledge Infrastructure from January 2000 to May 2024. The following keywords were used: ‘Yiyi Fuzi Baijiang San,’ ‘Yiyi Fuzi Baijiang Powder,’ ‘YYFZBJS,’ ‘YYFZBJP,’‘Coix seed,’ ‘Adlay,’ ‘C. lacryma-jobi L. var. mayuen (Roman.) Stapf.’‘Aconitum carmichaeli Debx.’‘Radix Aconiti Lateralis,’‘Patrinia villosa,’‘P. villosa (Thunb.) Juss.’ ‘Traditional Chinese Medicine,’ ‘mechanism,’ and ‘clinical application.’ Articles published in peer-reviewed journals were considered. The retrieved articles were reviewed by two independent reviewers based on their titles, abstracts, and full texts, adhering to specific inclusion and exclusion criteria. The inclusion criteria were: 1) Original articles written in English or Chinese; 2) Articles that examined the relevant mechanisms or clinical application of YYFZBJS and the active components or herbs it contains. Exclusion criteria were as follows: 1) Articles written in any language other than English and Chinese; 2) Literature that is unrelated to YYFZBJS or its active components or herbs; 3) Editorials; 4) Conference abstracts; 5) Studies lacking adequate discussion on the review topic; 6) Duplicate publications.
3 Traditional applications of YYFZBJS
The botanical drugs comprising YYFZBJS each possess distinct and irreplaceable therapeutic properties, and their synergistic combination forms a solid foundation for the clinical application of YYFZBJS. Coix seed, the dried mature kernel of C. lacryma-jobi var. ma-yuen (Rom.Caill.) Stapf, is a widely utilized culinary and medicinal plant in Southeast Asia. In TCM theory, Coix seed is characterized by its cool nature, sweet and light flavour, and its association with the spleen, stomach and lung meridians (Hou J et al., 2018). It is widely recognized for its properties in clearing heat, expelling pus, relaxing tendons, relieving arthralgia, dispersing knots, strengthening the spleen, and dispelling dampness (Han X et al., 2017). Historically, Coix seed has been used to treat inflammatory conditions accompanied by abscesses. Radix Aconiti Lateralis, the lateral root tuber of Aconitum carmichaeli Debx, is worm in nature, pungent and sweet, and associated with the heart, kidney and spleen meridians. It is important to note that Radix Aconiti Lateralis must undergo standardized and meticulous processing before it can be safely applied in clinical practice (Yang M et al., 2018). Secondly, Radix Aconiti Lateralis is known for its functions in restoring yang, relieving collapse, tonifying fire, dispelling cold, and alleviating pain. Over time, its clinical applications have expanded to include the treatment of various conditions such as cancer, heart failure, colitis, and rheumatoid arthritis (Fu Y et al., 2022; Tai C et al., 2021). Thirdly, Patrinia villosa is cool in nature, pungent and bitter in taste, and is linked to the liver, stomach and large intestine meridians. As a classical heat-clearing and detoxifying herb, Patrinia villosa is used to eliminate carbuncles, expel pus, dispel blood stasis, and relieve pain. Consequently, it is commonly employed in treating intestinal abscess, lung carbuncles, gynecological epigastric pain, postpartum blood stasis, and eczema (He et al., 2019; Gong et al., 2021).
4 Active ingredients of YYFZBJS
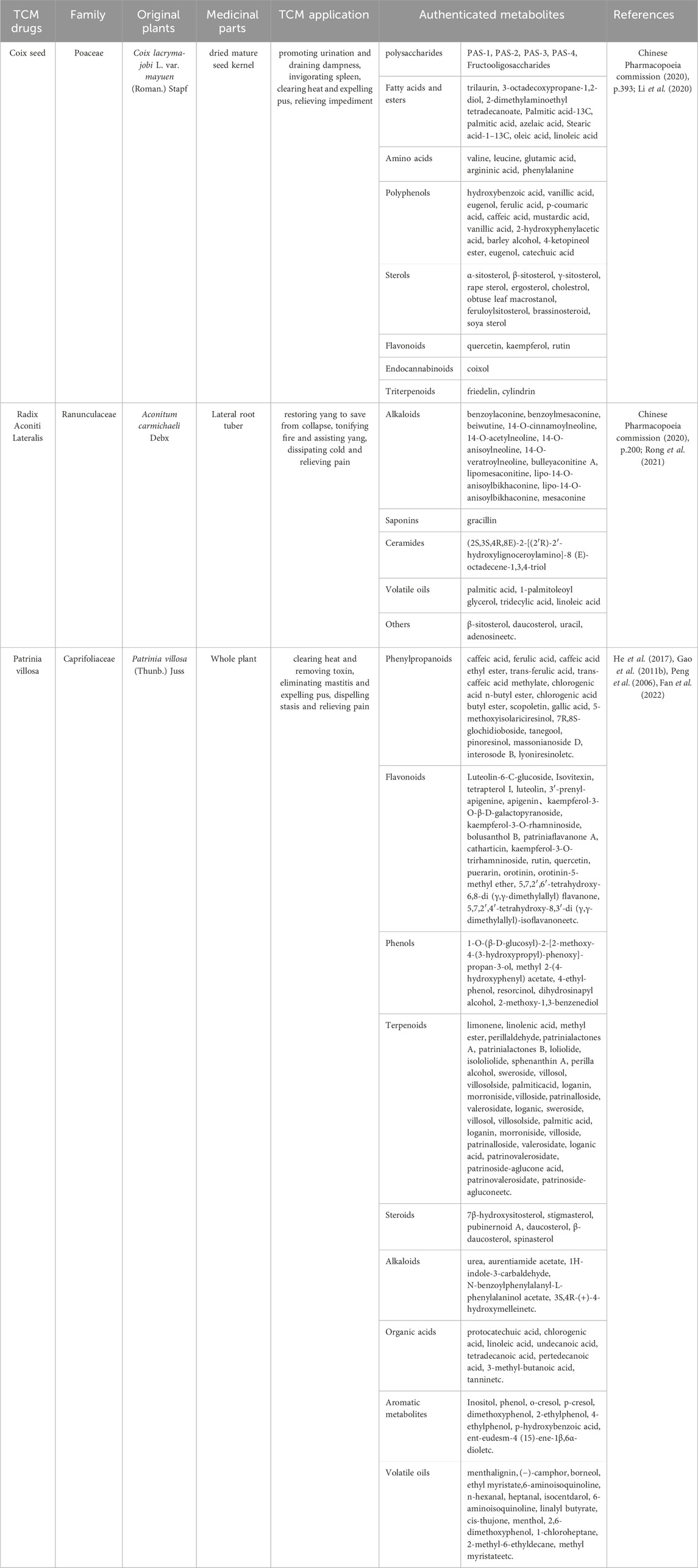
Analyzing the active ingredients of YYFZBJS is essential for understanding its pharmacological mechanisms, as it facilitates the identification of key bioactive compounds and their interactions with various targets. Advanced analytical techniques, such as UPLC-MS, have been employed to identify several significant ingredients in YYFZBJS, including liquiritigenin, aconitine, hypaconitine, luteolin, and puerarin (Zhang et al., 2022). Furthermore, additional components such as Coix seed oil (CSO), flavonoids, and Aconitum alkaloids have also been isolated from YYFZBJS (Table 1; Figure 2). Beyond merely detecting these ingredients, network analysis methods play a pivotal role in clarifying the synergistic effects and mechanisms underlying the multi-component, multi-target nature of YYFZBJS. These methods help in mapping the complex interactions among the bioactive compounds and their respective targets, providing a comprehensive understanding of how the formula exerts its therapeutic effects (Sui et al., 2020). This approach has revealed the diverse biological activities of YYFZBJS, highlighting its anti-inflammatory, anti-cancer, and antioxidant properties (Sun et al., 2020; Fang et al., 2018a). Such integrated analyses provide researchers with valuable insights into the therapeutic potential of YYFZBJS and its potential clinical applications.
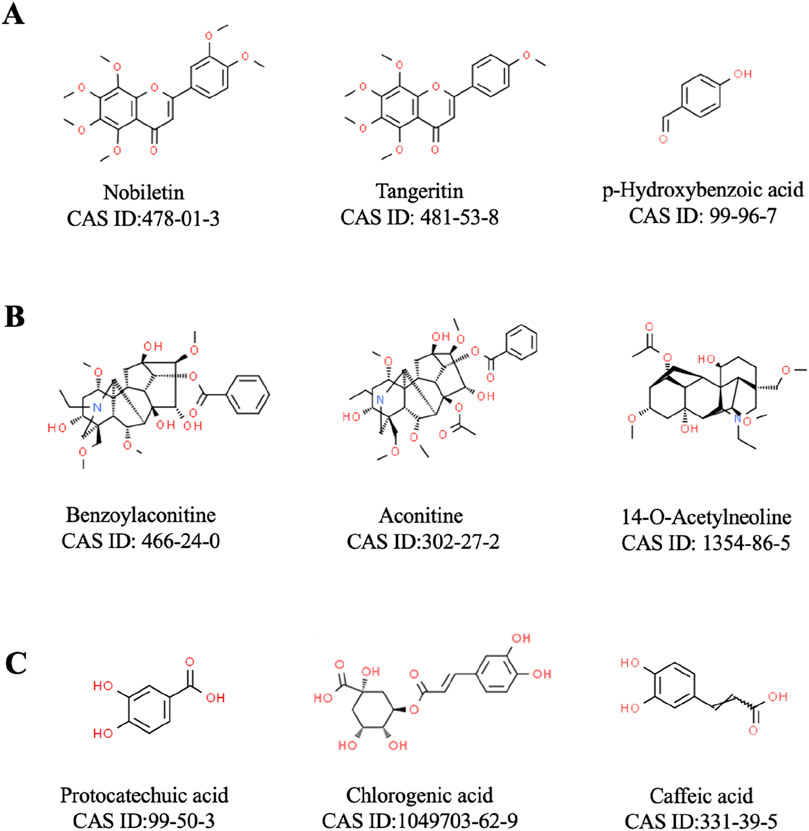
Figure 2. Representative metabolites included in YYFZBJS. (A): Coix seed; (B) Radix Aconiti Lateralis; (C) Patrinia villosa).
5 Pharmacological activities of YYFZBJS and its constituents
The clinical application of YYFZBJS is strongly supported by an abundance of in-depth modern pharmacological research. Although the content of constituents in YYFZBJS decoction liquid may vary with different extraction methods, the active components with high content are mainly CSO and flavonoids, and their pharmacological effects have been continuously recognized (Zhou et al., 2016).
5.1 Signaling pathway regulation in inflammation and metabolism
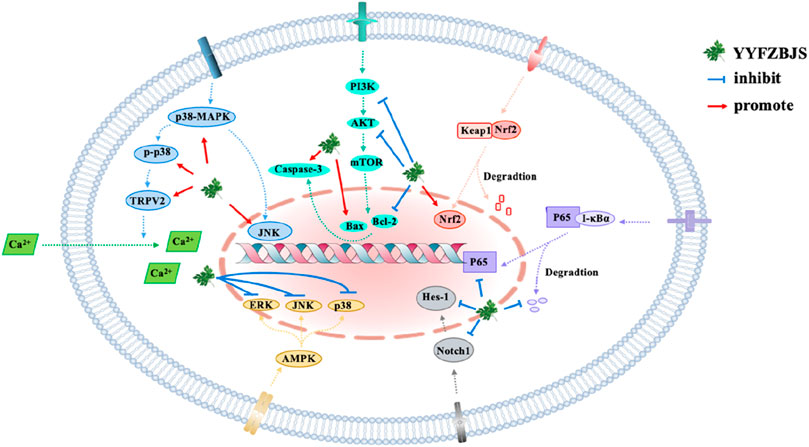
The crucial role of signaling pathways in metabolic disorders and infections lies in their influence on cellular responses. These pathways are intricately linked to various cellular processes, including metabolism, gene expression, alterations in intracellular enzyme activities, cytoskeletal structure, and DNA synthesis. In keeping with these, numerous studies have demonstrated that YYFZBJS plays a therapeutic role in modulating specific signaling pathways (Figure 3).
For instance, the Nrf2 signaling pathway is recognized as one of the most essential mechanisms in the regulation of antioxidant stress responses. YYFZBJS has been shown to increase the expression of Nrf2 and its downstream antioxidant protein HO-1 in rodents with UC, resulting in a decreased inflammatory response (Fang et al., 2018b). However, the precise metabolites responsible for this effect remain unidentified.
One of the principal active components of YYFZBJS, CSO, has been reported to modulate signaling pathways involved in lipid metabolism (Qin et al., 2023). Studies have identified that CSO primarily consists of oleic acid (50.54%), linoleic acid (33.76%), palmitic acid (11.74%), and stearic acid (2.45%), with its fatty acid profile closely matching that of vegetable oil (Ni et al., 2021). CSO has been shown to influence the p-AMPK/SePP1/apoER2 signaling pathway, which regulates lipid accumulation in liver tissue. Researchers demonstrated that CSO could inhibit the phosphorylation of adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK), leading to a reduction in the expression of SePP1/apoER2, thereby decreasing lipid accumulation, as observed in both in vivo and in vitro experiments (Gu et al., 2021). Similarily, Benzoylaconitine, another active metabolite of YYFZBJS, has emerged as a potential therapeutic agent for synovial inflammation in rheumatoid arthritis. The underlying mechanism appears to involve the inhibition of the PI3K/Akt signaling pathway, through the suppression of IL-1-induced expression of IL-6 and IL-8, inflammatory cytokines associated with rheumatoid arthritis (Yu et al., 2020). Additionally, Benzoylaconitine targets the MAPK and NF-κB signaling pathways to exert its anti-inflammatory effects. In summary, the regulation of signaling pathways is a critical mechanism by which YYFZBJS exerts its therapeutic effects.
5.2 Gene and protein modulation in tumor and muscle atrophy
Researchers have identified that the active metabolites of YYFZBJS have the potential to regulate gene and protein expression, which is crucial in addressing the abnormal expression contributing to the onset and progression of various diseases. For instance, Chen et al. discovered that CSO significantly upregulated the expression of stress-inducible genes, such as daf-16, sod-3, hsp-16.2, and gst-4 in Caenorhabditis elegans (Chen X. Y. et al., 2020). In another study, CSO was shown to increase the binding of the NF-κB p65 subunit to the promoter regions of IL-2- and Bcl-2-encoding genes in tumor-bearing mice (Huang X. et al., 2014). Radix Aconiti Lateralis has also been found to exert therapeutic effects through the modulation of protein expression. The specific mechanisms include the significant suppression of dexamethasone-induced mRNA expressions of muscle atrophy F-box protein (MAFbx)/atrogin1, Casitas B-lineage lymphoma-b (Cbl-b), troponin, and branched-chain amino acid aminotransferase 2 (BCAT2), thereby inhibiting muscle atrophy (Kondo et al., 2022). Additionally, Aconitum alkaloids, the main metabolites of Radix Aconiti Lateralis, have been observed to increase the expression of multidrug resistance-associated protein 2 (MRP2), providing insights into the clinical application of plants in the Aconitum family (Wu et al., 2018). It is well recognized that tumor patients are prone to muscle atrophy and weakness as their illness progresses; however, the role of YYFZBJS’s active metabolites in treating muscular dystrophy in these patients remains unclear and warrants further exploration and validation (Williams et al., 2021).
5.3 Apoptosis-based therapeutic and potential toxic effects
Apoptosis is a genetically regulated process by which cells autonomously undergo programmed death, playing a crucial role in maintaining a stable internal environment. Mitochondrial proteins such as Bcl-2, Bax, and Cyt-c are activated and oligomerize on the outer mitochondrial membrane, mediating its permeability-a critical step in the apoptotic process. One study found that CSO can regulate mitochondrial apoptotic pathways by downregulating Bcl-2 and upregulating Bax, cleaved caspase-9, cleaved caspase-3, and Cyt-c proteins (Yang et al., 2022). Another active metabolite, SPVJ, extracted from Patrinia villosa, has been reported to significantly increase the number of apoptotic cells (from 9.42% to 28.9%) in U14 cervical cancer-bearing mice when administered at a dose of 100 mg/kg body weight (p.o.), compared to the control group receiving distilled water (p.o.) (Zhang et al., 2008). Additionally, Aconitum alkaloids have been demonstrated to induce apoptosis in various tumor cells without affecting normal cells, as confirmed by several studies (Fan et al., 2016; Qu et al., 2020). However, it is important to note that apoptosis is also implicated in the cardiotoxicity and neurotoxicity associated with aconitum plants. Xia et al. conducted a developmental toxicity assay of Aconitine on zebrafish embryos and found that high doses (7.27 and 8.23 μM) of Aconitine increased the levels of reactive oxygen species (ROS) and induced apoptosis in embryonic heart and brain regions (Xia et al., 2021). Furthermore, Aconitine was found to promote intracellular Ca2+ accumulation and cardiomyocyte apoptosis through the p38 MAPK signaling pathway in a dose-dependent manner (Yang et al., 2021). These findings suggest that apoptosis is not only related to the therapeutic effects of Aconitine but also to its potential toxicity. Therefore, the anti-apopotic function of YYFZBJS may be achieved through the regulation of mitochondrial proteins, remodeling ion channels, and activating signaling pathways.
5.4 Modulation of immune response for diverse effects
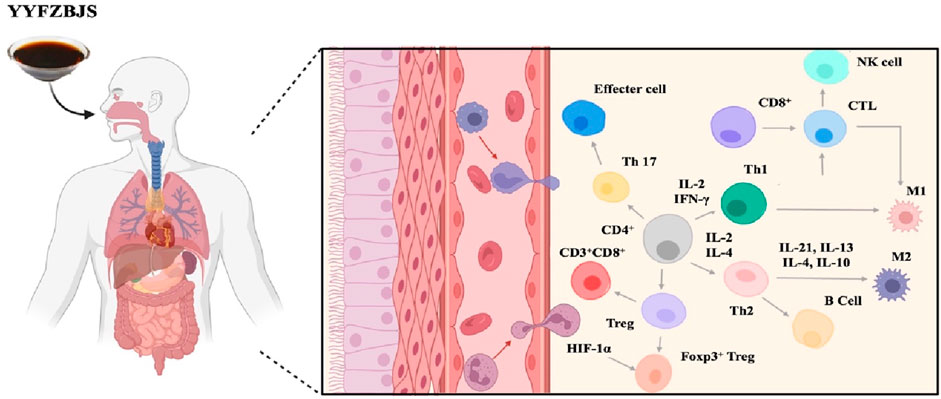
As is well known, the immune system plays a crucial role in defending the body against pathogens and maintaining overall health by identifying and neutralizing harmful microorganisms and abnormal cells. Along with the development in molecular biology and immunology, the mechanism of the therapeutic effect on YYFZBJS has improved quickly in recent years. (Figure 4).
It is widely recognized that regulatory T (Treg) cells can inhibit anti-tumor immune responses, thereby facilitating tumor progression and invasion. Preliminary studies conducted by our research team have confirmed that the effects of oral administration of YYFZBJS on azoxymethane (AOM)/dextran sulfate sodium (DSS)-induced tumorigenesis in C57BL/6J mice. YYFZBJS treatment was found to reduce tumor load, tumor number, histological severity, and disease activity index (DAI) scores. Additionally, the study observed that the tumor-inhibiting effects of YYFZBJS were diminished in a Treg-deficient mouse model, compared to mice treated with YYFZBJS alone. This suggests that the enhanced immune response mediated by peripheral Tregs (pTregs) plays a crucial role in the anticancer activities of YYFZBJS (Sui et al., 2020; Zhang et al., 2022). The immune-regulating effects of YYFZBJS are closely related to its herbal components, as confirmed by another clinical study where Coix seed was the main intervention method (Jinnouchi et al., 2021). In animal models, the ethyl-acetate fraction of the ethanolic extract of Coix seed (ABE-EtOAc) has been shown to treat reversible increases in Th1/Th2 immunity by upregulating the expression of IL-2 and IL-4, reducing the release of histamines and cytokines such as IL-6 and TNF, and decreasing Akt production (Chen et al., 2012a; Chen et al., 2012b). Additionally, alkali-extractable polysaccharides from Coix seed have been shown to stimulate the production of molecules such as NO, TNF, and IL-6 in RAW264.7 murine macrophages a dose-dependent manner (Yao et al., 2015). Moreover, strong evidence indicated that the water-soluble polysaccharide fractions of Radix Aconiti Lateralis can stimulate lymphocyte proliferation, enhance antibody production, and promote macrophage phagocytosis, thereby boosting immune responses in the host (Zhao et al., 2006; Gao T. et al., 2011).
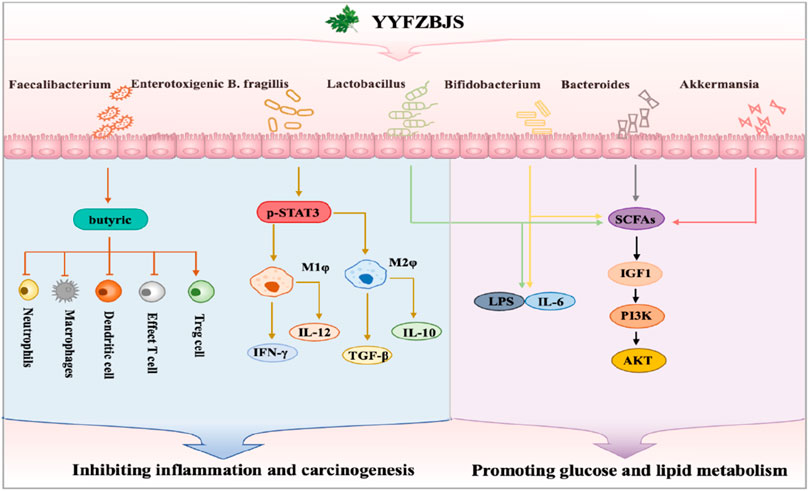
5.5 Therapeutic effects via gut microbiota modulation
An increasing number of studies are highlighting the role of TCM formulas in modulating the microbiota, reflecting growing interest in their impact on microbial balance and health (Hwang et al., 2016). It is well-established that the intestinal flora represents a complex microecosystem involved in numerous pathological processes, which can lead to diseases such as inflammatory bowel disease (IBD), cancer, diabetes, and tumors (De Vos et al., 2022). Modern pharmacological research has demonstrated that YYFZBJS possesses remarkable properties in modulating the intestinal flora, thereby exerting therapeutic effects on various diseases (Figure 5). Our previous studies revealed that the intestinal flora of APCMin/+ mice treated with YYFZBJS exhibited significant alterations, including an increase in beneficial bacterial species such as Bifidobacterium and a decrease in harmful species such as Bacteroides, norank_f_ Erysipelotrichaceae (Zhang L. et al., 2020). The dysbiosis caused by enterotoxigenic Bacteroides fragilis has also been found to contribute to the development of colorectal cancer (CRC) by activating the p-STAT3 receptor, which influences M2 macrophage polarization. However, YYFZBJS can inhibit this process, thereby preventing chronic inflammation and the malignant transformation of adenomas (Chai et al., 2021). Coix seed was found to promote the spontaneous regression of viral cutaneous infections in healthy adult males by increasing the abundance of gastrointestinal Faecalibacterium (Li et al., 2021b). Additionally, the hypoglycemic efficacy of polysaccharides from Coix seed was also demonstrated through their ability to modulate the gastrointestinal microbiota, specifically by increasing the production of short-chain fatty acid (SCFA)-producing bacteria (Ge et al., 2024).
6 Clinical application and therapeutic potential of YYFZBJS
6.1 Clinical efficacy in the management of inflammatory diseases
Traditionally, YYFZBJS was used to treat chronic intestinal abscesses, which are localized inflammations of the intestines. However, with the accumulation of clinical experience and the advancement of pharmacological research, YYFZBJS and its constituent herbs have been proven to possess broader anti-inflammatory effects, demonstrating significant efficacy in treating various intestinal and extra-intestinal diseases.
6.1.1 Decoction of YYFZBJS
The anti-inflammatory properties of YYFZBJS are primarily attributed to its constituents, Patrinia villosa and Coix seed. With the adjunctive support of Radix Aconiti Lateralis, YYFZBJS is more widely utilized in the management of chronic inflammatory diseases, including UC, Crohn’s disease, and anal sinusitis. Notably, YYFZBJS is typically administered via oral ingestion or retention enemas as a novel approach to the UC treatment, and the clinical efficacy has been confirmed in randomized controlled trials involving modest sample sizes (Hu et al., 2020; Li and Chen, 2021; Lv and Zhang, 2017; Zhang S. X. et al., 2019; Liu et al., 2023). Some clinical trials also demonstrated that patients receiving YYFZBJS have a higher effective rate and a lower recurrence rate compared to those treated with antibiotics such as norfloxacin and metronidazole for anal sinusitis (Wei et al., 2013; Guo et al., 2009). In addition, YYFZBJS is often combined with other treatments for inflammatory diseases. Single-center randomized controlled studies have shown that combining YYFZBJS with other TCM prescriptions, such as Dachaihu Decoction or Guizhi Fuling Pills, can significantly alleviate the clinical symptoms of chronic prostatitis with few adverse effects and reduced costs (Kong, 2016;Liu and Liu, 2018). Furthermore, it is reported that YYFZBJS may be effective in treating chronic gynecological inflammatory diseases like chronic pelvic inflammatory disease (PID) and chronic skin conditions like acne (Wu et al., 2016; Zhang and Chen, 2021). Overall, YYFZBJS has broad applications in the treatment of inflammatory diseases, with one of its most prominent advantages being its ability to significantly reduce the recurrence of chronic infectious diseases.
6.1.2 Constituents in YYFZBJS
Recent studies have elucidated that the anti-inflammatory mechanisms of YYFZBJS are closely associated with its constituent metabolites. Coix seed, the botanical component with the highest concentration in YYFZBJS, exhibits significant anti-inflammatory properties. Notably, ethyl acetate extracts of Coix seed have been found to contain anti-inflammatory flavonoids and phenolic compounds, such as tangeretin, nobiletin, and p-hydroxybenzoic acid. These compounds are thought to exert their effects through potent inhibition of NO production, suppression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) expression, and reduction of pro-inflammatory cytokines, IL-6 and TNF-α (Huang D. W. et al., 2014; Seo et al., 2000; Huang et al., 2009).
Additionally, other metabolites within YYFZBJS demonstrate substantial anti-inflammatory effects. 14-O-acetylneoline, a diterpenoid alkaloid derived from Radix Aconiti Lateralis, has been shown to protect rodents from various forms of intestinal inflammation by reducing IFN-γ production (Wangchuk et al., 2015). In an in vivo study, colonic IFN-γ mRNA levels were significantly lower in mice treated with 14-O-acetylneoline compared to control mice administered trinitrobenzoylsulfonic acid. Although further research is necessary, this alkaloid is currently considered a promising candidate for the development of anti-colitis therapeutics. Moreover, hydroalcoholic extracts of Radix Aconiti Lateralis have been reported to inhibit dense inflammatory cell infiltration in the lamina propria of animals with gastric ulcers by mitigating detrimental free radical cascades and oxidative stress (Rajakrishnan et al., 2020). The anti-inflammatory effect of Patrinia villosa is equally noteworthy. Methanol extracts of Patrinia villosa roots have demonstrated anti-inflammatory effects at colorectal sites by inhibiting NF-κB p65 activation and reducing levels of inflammatory mediators such as IL-6 and TNF (Cho et al., 2011; Lee et al., 2012). In rodent models of PID, Patrinia villosa significantly reduced inflammatory cell infiltration in the pelvis. The underlying mechanisms may involve the downregulation of lactate dehydrogenase and pyruvate carboxylase, coupled with the upregulation of arachidonic acid esters (Zou et al., 2015). Furthermore, Patrinia villosa has been reported to mitigate cutaneous inflammation in rodents with atopic dermatitis by increasing filaggrin expression and reducing inflammation-related cytokines and IgE levels, potentially through inhibition of JNK1/2 phosphorylation (Cha et al., 2017).
6.2 Clinical efficacy in the management of cancer
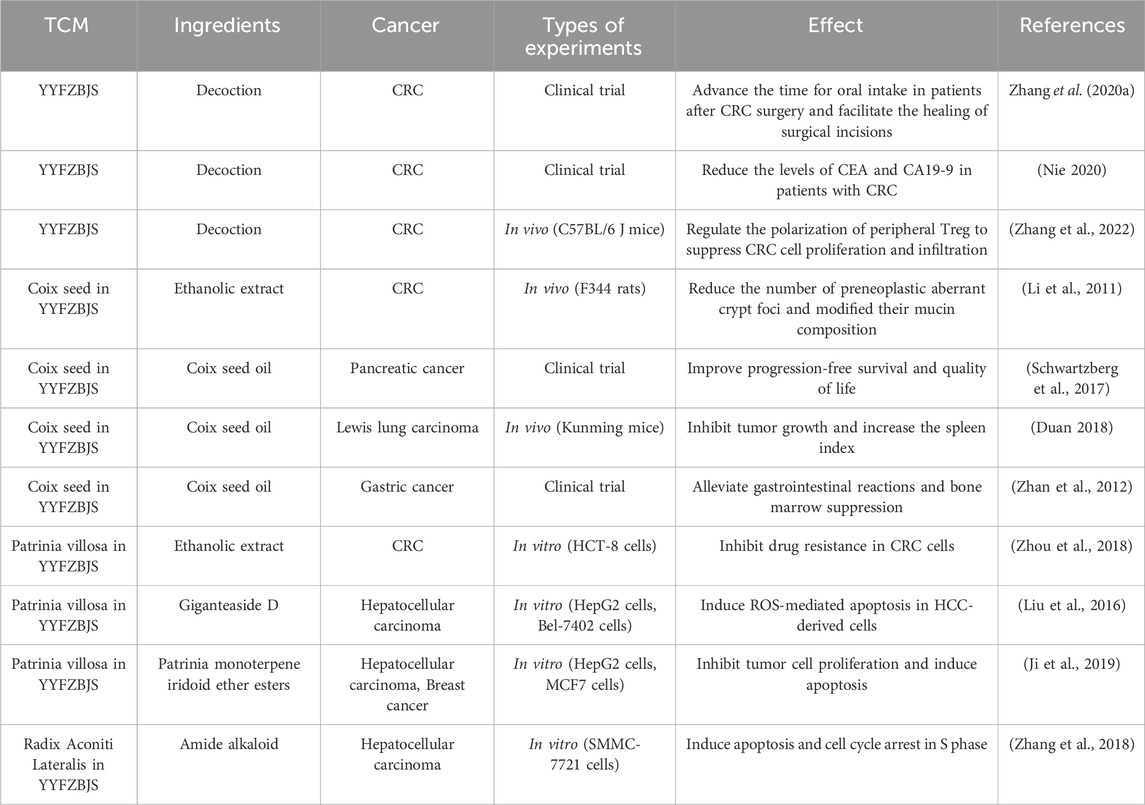
Recent research has increasingly highlighted the link between inflammation and various stages of cancer, including its onset, progression, and recurrence (Wang et al., 2024). This recognition underscores the importance of inflammation inhibition as a critical strategy in cancer treatment. With an understanding of the close relationship between inflammation and tumorigenesis, the potential anti-cancer effect of YYFZBJS has also become a research topic of significant interest (Table 2).
6.2.1 Multiple cancers including colorectal cancer
TCM has traditionally emphasized the observation and summary of disease symptoms, which complicates the identification of a specific diagnosis directly corresponding to CRC. However, the characteristic clinical manifestations of CRC suggest a close association with the TCM concept of “intestinal abscess,” which is characterized by severe symptoms and treatment difficulties, and relevant to the indications for YYFZBJS (Liu, 2017).
Clinical studies have indicated that the combination of YYFZBJS with chemotherapy may improve treatment outcomes for CRC and facilitate postoperative recovery. Patients with postoperative CRC who received YYFZBJS experienced significantly shorter times to first defecation, first bowel movement, and initiation of oral feeding (p < 0.05), along with notably higher plasma albumin levels compared to those who did not receive YYFZBJS (Nie, 2020; Zhang Y. et al., 2020). Additionally, administration of Coix seed and its ethanolic extract in rodent models has been shown to reduce preneoplastic aberrant crypt foci (ACF) and alter mucin composition, potentially preventing colonic preneoplastic lesions (Li et al., 2011). Preliminary research also suggests that Patrinia villosa may decrease the resistance of intestinal cancer cells to 5-fluorouracil (5-FU), indicating that it could enhance the effectiveness of CRC chemotherapy (Zhou et al., 2018).
Beyond CRC, emerging evidence suggests that YYFZBJS and its constituent botanical drugs may also be beneficial in other cancer types (Han et al., 2019; Song et al., 2018; Hu et al., 2023). A randomized, open-label study on pancreatic cancer patients demonstrated that Kanglaite (a pharmaceutical-grade emulsion of Coix seed oil) combined with gemcitabine significantly improved progression-free survival compared to gemcitabine alone (Schwartzberg et al., 2017). The addition of Coix seed to chemotherapy regimens for lung and gastric cancers has also yielded positive outcomes (Duan, 2018; Zhan et al., 2012). The therapeutic efficacy of Coix seed may be attributed to its high solubility and bioavailability. Coix seed oil (CSO), due to its unique texture and anti-tumor effects, has been developed into an oil phase component of microemulsions for anti-tumor treatment, showing promising results in breast, cervical, and lung cancers (Qu et al., 2017b; Chen Y. et al., 2020; Qu et al., 2017a). Moreover, Kanglaite alone has been reported to alleviate cancer-related pain and improve the quality of life for cancer patients (Zhang P. et al., 2019).
6.2.2 Antitumor potential of the constituents in YYFZBJS
As a crucial component of YYFZBJS, Radix Aconiti Lateralis has demonstrated significant potential in anti-tumor activity. The diterpene alkaloids, which are categorized into C18-, C19-, C20-, and bis-diterpenoid alkaloids, are the principal components responsible for its therapeutic effects. These compounds exhibit notable cytotoxicity against various tumors, including lung cancer, prostate cancer, and triple-negative breast cancer (Wada and Yamashita, 2019; Thawabteh et al., 2021). In addition to the naturally alkaloids extracted from Radix Aconiti Lateralis, synthetic alkaloids derived from its metabolites have also demonstrated significant antitumor activity. For instance, ITPD, a metabolite from Radix Aconiti Lateralis, exerts its antitumor effects by activating caspase-3 and caspase-9, inducing Bax/Bcl-2 imbalance, leading to DNA damage and subsequent cell apoptosis (Zhang et al., 2018). In Patrinia villosa, the main antitumor components identified include polysaccharides, giganteaside D (GD), and Patrinia monoterpene iridoid ether esters (PMIEE), which has been reported to inhibit cancer cell proliferation and induce apoptosis in liver, breast, and cervical cancer cells through the downregulation of Bcl-2, CDC2, and Cyclin B1, and the upregulation of Bax and caspase-3 (Gong et al., 2021; Ji et al., 2019; He et al., 2019). Additionally, GD has been found to induce reactive oxygen species (ROS) production, leading to mitochondria-mediated apoptosis in hepatoma cells, and its cytotoxicity is associated with modulation of the MAPK signaling pathway (Liu et al., 2016; Xie et al., 2017).
7 Conclusion and perspectives
The use of YYFZBJS has a history spanning over 1800 years, but the concept of evidence-based medicine is relatively recent. As a result, there is currently limited support from large-scale clinical studies and historical data. Furthermore, the broad clinical application of YYFZBJS, as discussed in this study, is not arbitrary or widespread. Aligned with the TCM principle of pattern identification and treatment, YYFZBJS is primarily used to treat patients with a cold-heat complex pattern as determined by competent TCM practitioners.
As highlighted in this review, the exact ingredients of YYFZBJS have not yet been identified, and the exact action mechanisms of YYFZBJS are still unclear. Therefore, extensive research and rational standardization are crucial for the successful clinical application and promotion of YYFZBJS. Additionally, more clinical trials and cohort studies are needed to establish the therapeutic benefits of these herbs.
Author contributions
YZ: Writing–original draft. LZ: Writing–review and editing. NC: Writing–original draft, Visualization. ZW: Data curation, Writing–original draft. HS: Project administration, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study was funded by “Jiaotong University Star” Program (No. YG2022QN082), the National Natural Science Foundation of China (No. 82204887), the Science Foundation for Shanghai Committee of Science Project (No. 23S21901200), Special Disease Construction Project of Jiading District Health System (No. ZB202406), Special Project of Traditional Chinese Medicine Scientific Research of Henan Province (No. 2024ZY2145), Project of Scientific and technological breakthroughs in Henan Province (No.242102310493), and Henan Province Science and Technology Research and Development Plan Joint Fund Project (No.242301420116).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer QJ declared a shared affiliation with the author NC to the handling editor at the time of the review.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Cha, K. J., Im, M. A., Gu, A., Kim, D. H., Lee, D., Lee, J. S., et al. (2017). Inhibitory effect of Patrinia scabiosifolia Link on the development of atopic dermatitis-like lesions in human keratinocytes and NC/Nga mice. J. Ethnopharmacol. 206, 135–143. doi:10.1016/j.jep.2017.03.045
Chai, N., Xiong, Y. B., Zhang, Y. L., Cheng, Y. L., Shi, W. F., Yao, Y. Q., et al. (2021). YYFZBJS inhibits colorectal tumorigenesis by remodeling gut microbiota and influence on M2 macrophage polarization in vivo and in vitro. Am. J. Cancer Res. 11, 5338–5357.
Chen, H. J., Hsu, H. Y., and Chiang, W. (2012a). Allergic immune-regulatory effects of adlay bran on an OVA-immunized mice allergic model. Food Chem. Toxicol. 50, 3808–3813. doi:10.1016/j.fct.2012.07.011
Chen, H. J., Lo, Y. C., and Chiang, W. (2012b). Inhibitory effects of adlay bran (Coix lachryma-jobi L. var. ma-yuen Stapf) on chemical mediator release and cytokine production in rat basophilic leukemia cells. J. Ethnopharmacol. 141, 119–127. doi:10.1016/j.jep.2012.02.009
Chen, X. Y., Liao, D. C., Yu, Y. T., Wei, C. M., Xuan, L. Y., Li, S., et al. (2020a). Coix seed oil prolongs lifespan and enhances stress resistance in Caenorhabditis elegans. Biogerontology 21, 245–256. doi:10.1007/s10522-020-09857-z
Chen, Y., Guo, M., Qu, D., Liu, Y., Guo, J., and Chen, Y. (2020b). Furin-responsive triterpenine-based liposomal complex enhances anticervical cancer therapy through size modulation. Drug Deliv. 27, 1608–1624. doi:10.1080/10717544.2020.1827086
Chinese Pharmacopoeia commission (2020). Pharmacopoeia of the people's Republic of China(I). BeiJing: China Medical Science Press.
Cho, E., Shin, J., Noh, Y., Cho, Y., Hong, S., Park, J., et al. (2011). Anti-inflammatory effects of methanol extract of Patrinia scabiosaefolia in mice with ulcerative colitis. J. Ethnopharmacol. 136, 428–435. doi:10.1016/j.jep.2010.04.047
De Vos, W. M., Tilg, H., Van Hul, M., and Cani, P. D. (2022). Gut microbiome and health: mechanistic insights. Gut 71, 1020–1032. doi:10.1136/gutjnl-2021-326789
Duan, G. C. (2018). The effects of combination of coix seed extract and cisplatin on TAM and expression of HIF-1α in vivo in lewis lung carcinoma. Iran. J. Public Health 47, 838–843.
Fang, J., Chen, J., and Jin, Y. (2018a). Clinical application and research review of Yiyi Fuzi Baijiang powder. Shaanxi J. Traditional Chin. Med. 39 (03), 383–386.
Fang, J., Chen, J., Peng, J. W., Li, H. L., and Jiang, C. F. (2018b). Mechanism of action of yiyi Fuzi Baijiang san on ulcerative colitis based on Nrf2 pathway. Chin. J. Exp. Traditional Med. Formulae 24 (13), 85–92. doi:10.13422/j.cnki.syfjx.20181052
Fu, Y., Zou, Y., Lei, F., Wangensteen, H., and Inngjerdingen, K. (2022). Aconitum carmichaelii Debeaux: A. systematic review on traditional use, and the chemical structures and pharmacological properties of polysaccharides and phenolic compounds in the roots. J. Ethnopharmacol. 291, 115148. doi:10.1016/j.jep.2022.115148
Gao, L., Zhang, L., Li, N., Liu, J. Y., Cai, P. L., and Yang, S. L. (2011b). New triterpenoid saponins from Patrinia scabiosaefolia. Carbohydr. Res. 346 (18), 2881–2885. doi:10.1016/j.carres.2011.10.008
Gao, T., Ma, S., Song, J., Bi, H., and Tao, Y. (2011a). Antioxidant and immunological activities of water-soluble polysaccharides from Aconitum kusnezoffii Reichb. Int. J. Biol. Macromol. 49, 580–586. doi:10.1016/j.ijbiomac.2011.06.017
Ge, Q., Hou, C., Rao, X., Zhang, A., Xiao, G., Wang, L., et al. (2024). In vitro. fermentation characteristics of polysaccharides from coix seed and its effects on the gut microbiota. Int. J. Biol. Macromol. 262 (Pt 2), 129994. doi:10.1016/j.ijbiomac.2024.129994
Gong, L., Zou, W., Zheng, K., Shi, B., and Liu, M. (2021). The Herba Patriniae (Caprifoliaceae): a review on traditional uses, phytochemistry, pharmacology and quality control. J. Ethnopharmacol. 265, 113264. doi:10.1016/j.jep.2020.113264
Han, X., Ji, X., Zhao, H., Zhang, Y., Liu, G., Wang, Y., et al. (2017). Mechanisms of coix. Seed compositions in the treatment of spleen deficiency and wet dampness zheng. African journal of traditional. complementary, Altern. Med. 14 (4), 239–246. doi:10.21010/ajtcam.v14i4.26
Han, X., Song, S. S., Zhao, H. J., Bao, Y. R., and Zhao, L. (2019). The study of the antitumor effect of Yiyi Fuzi Baijiang Powder effective components of optimal compatibility on H22 tumor-bearing mice. Lishizhen Med. Material Medica Res. 30 (7), 1551–1553.
He, X., Luan, F., Zhao, Z., Ning, N., Li, M., Jin, L., et al. (2017). The genus Patrinia: a review of traditional uses, phytochemical and pharmacological studies. Am. J. Chin. Med. 45 (4), 637–666. doi:10.1142/S0192415X17500379
He, X. Y., Wei, L. Z., Wang, H. L., Sun, X. T., and Duan, L. J. (2019). Effects of patrina villosa Juss on poptosis of human cervical cancer cell line Hela and it’s mechanism. Gansu Med. J. 38 (7), 584–585+611. doi:10.15975/j.cnki.gsyy.2019.07.002
Hou, J., Guo, J., Cao, C., Yao, S., Long, H., Cai, L., et al. (2018). Green quantification. Strategy combined with chemometric analysis for triglycerides in seeds used in traditional Chinese medicine. Planta medica. 84 (6-07), 457–464. doi:10.1055/s-0044-100723
Hu, J. W., Feng, Q. H., and Liang, X. X. (2020). Effect of modified yiyi Fuzi Baijiang san for retention enema in the treatment of ulcerative colitis, and its influence on serum of SOD, TNF-α and IL-β. Chin. J. Coloproctol. 40 (8), 25–28.
Hu, Q., Liu, Y., Yu, J., Yang, X., Yang, M., He, Y., et al. (2023). The protective effect and antitumor. activity of Aconiti Lateralis Radix Praeparata (Fuzi) polysaccharide on cyclophosphamide-induced immunosuppression in H22 tumor-bearing mice. Front. Pharmacol. 14, 1151092. doi:10.3389/fphar.2023.1151092
Huang, D. W., Kuo, Y. H., Lin, F. Y., Lin, Y. L., and Chiang, W. (2009). Effect of Adlay (Coix lachryma-jobi L. var. ma-yuen Stapf) Testa and its phenolic components on Cu2+-treated low-density lipoprotein (LDL) oxidation and lipopolysaccharide (LPS)-induced inflammation in RAW 264.7 macrophages. J. Agric. Food Chem. 57, 2259–2266. doi:10.1021/jf803255p
Huang, D. W., Wu, C. H., Shih, C. K., Liu, C. Y., Shih, P. H., Shieh, T. M., et al. (2014a). Application of the solvent extraction technique to investigation of the anti-inflammatory activity of adlay bran. Food Chem. 145, 445–453. doi:10.1016/j.foodchem.2013.08.071
Huang, X., Qin, J., and Lu, S. (2014b). Kanglaite stimulates anticancer immune responses and inhibits HepG2 cell transplantation-induced tumor growth. Mol. Med. Rep. 10, 2153–2159. doi:10.3892/mmr.2014.2479
Hwang, I. Y., Koh, E., Kim, H. R., Yew, W. S., and Chang, M. W. (2016). Reprogrammable microbial cell-based therapeutics against antibiotic-resistant bacteria. Drug Resist. Updat. 27, 59–71. doi:10.1016/j.drup.2016.06.002
Ji, L. Q., Wu, J. M., Min, W., Chen, Y. F., and Xie, M. H. (2019). Effects of PMIEE on proliferation and apoptosis of HepG2 and MCF7 cells. Chin. J. Mod. Appl. Pharm. 36 (6), 692–697. doi:10.13748/j.cnki.issn1007-7693.2019.06.009
Jinnouchi, M., Miyahara, T., and Suzuki, Y. (2021). Coix seed consumption affects the gut microbiota and the peripheral lymphocyte subset profiles of healthy male adults. Nutrients 13 (11), 4079. doi:10.3390/nu13114079
Kondo, T., Ishida, T., Ye, K., Muraguchi, M., Tanimura, Y., Yoshida, M., et al. (2022). Suppressive effects of processed aconite root on dexamethasone-induced muscle ring finger protein-1 expression and its active ingredients. J. Nat. Med. 76 (3), 594–604. doi:10.1007/s11418-022-01606-5
Kong, L. Q. (2016). Pain reduction on category III prostatitis patients through the application of DaChaiHuTang combined with YiYiFuZiBaiJiangSan decoction. J. Basic Chin. Med. 22 (9), 1270–1272. doi:10.19945/j.cnki.issn.1006-3250.2016.09.050
Lee, E. J., Kim, C., Kim, J. Y., Kim, S. M., Nam, D., Jang, H. J., et al. (2012). Inhibition of LPS-induced inflammatory biomarkers by ethyl acetate fraction of Patrinia scabiosaefolia through suppression of NF-κB activation in RAW 264.7 cells. Immunopharmacol. Immunotoxicol. 34, 282–291. doi:10.3109/08923973.2011.602412
Li, H. L., Xue, Y., and Chen, J. (2021). A Meta-analysis of therapeutic effect of Yiyi Fuzi Baijiang powder on ulcerative colitis. Chin. J. Integr. Trad. West Med. Dig. 29 (2), 132–137.
Li, H. L., Xue, Y., Chen, J., and Liang, G. Q. (2021b). Effect of Yiyi Fuzi Baijiang powder on intestinal flora of acute ulcerative colitis mice based on 16S rDNA sequencing. Chin. J. Microecol. 33 (12), 1365–1373. doi:10.13381/j.cnki.cjm.202112001
Li, S. C., Chen, C. M., Lin, S. H., Chiang, W., and Shih, C. K. (2011). Effects of adlay bran and its ethanolic extract and residue on preneoplastic lesions of the colon in rats. J. Sci. Food Agric. 91, 547–552. doi:10.1002/jsfa.4219
Li, X. K., Gu, K., Liang, M. W., Zhang, Y. T., Wang, Y. M., and Li, Y. B. (2020). Research progress on chemical constituents and pharmacological effects of Coicis Semen. Chin. Traditional Herb. Drugs 51 (21), 5645–5657.
Liu, J., Wei, X., Wu, Y., Wang, Y., Qiu, Y., Shi, J., et al. (2016). Giganteaside D induces ROS-mediated apoptosis in human hepatocellular carcinoma cells through the MAPK pathway. Cell Oncol. (Dordr) 39, 333–342. doi:10.1007/s13402-016-0273-9
Liu, J. R., and Liu, S. Y. (2018). Observation on the effect of Yiyi Fuzi Baijiang powder combined with Guizhi Fuling Pill in the treatment of chronic prostatitis of damp-heat stasis type. J. Clin. Med. 5 (37), 165–166. doi:10.16281/j.cnki.jocml.2018.37.124
Liu, M. (2017). Analysis of clinical application of YYFZBJS. Chiness J. Ethnomedicine Ethnopharmacy 26 (4), 6–7.
Liu, M., Wang, Z., Liu, X., Xiao, H., Liu, Y., Wang, J., et al. (2023). Therapeutic effect of Yiyi Fuzi Baijiang formula on TNBS-induced ulcerative colitis via metabolism and Th17/Treg cell balance. J. Ethnopharmacol. 309, 116301. doi:10.1016/j.jep.2023.116301
Lv, L. Y., and Zhang, L. (2017). Randomized control trail in the treatment of ulcerative colitis with retention enema of Chinese herabal medicine and moxibustion. World J. Integr. Traditional Weatern Med. 12 (7), 970–973. doi:10.13935/j.cnki.sjzx.170720
Nie, D. (2020). Clinical study of modified Yiyi Fuzi Baijiang San combined with XELOX regimen in the treatment of stage IV colorectal cancer (cold-heat complex syndrome) [D]. Chengdu University of Traditional Chinese Medicine. doi:10.26988/d.cnki.gcdzu.2020.000129
Oravecz, M., and Mészáros, J. (2012). Traditional Chinese medicine: theoretical background and its use in China. Orv. Hetil. 153, 723–731. doi:10.1556/OH.2012.29365
Qin, B., Yao, Y., Zhang, J., and Wang, L. (2023). Bioavailability of coix seed polyphenols in a MKN28/caco-2. Continuous transport model and their lipid-lowering effects via modulating adipocyte differentiation of 3T3-L1 cells. J. Agric. Food Chem71 71 (22), 8425–8436. doi:10.1021/acs.jafc.2c08388
Qu, D., Guo, M., Qin, Y., Wang, L., Zong, B., Chen, Y., et al. (2017a). A multicomponent microemulsion using rational combination strategy improves lung cancer treatment through synergistic effects and deep tumor penetration. Drug Deliv. 24, 1179–1190. doi:10.1080/10717544.2017.1365394
Qu, D., Ma, J., Song, N., Hui, L., Yang, L., Guo, Y., et al. (2020). Lappaconitine sulfate induces apoptosis and G0/G1 phase cell cycle arrest by PI3K/AKT signaling pathway in human non-small cell lung cancer A549 cells. Acta histochem. 122, 151557. doi:10.1016/j.acthis.2020.151557
Qu, D., Wang, L., Liu, M., Shen, S., Li, T., Liu, Y., et al. (2017b). Oral nanomedicine based on multicomponent microemulsions for drug-resistant breast cancer treatment. Biomacromolecules 18, 1268–1280. doi:10.1021/acs.biomac.7b00011
Rajakrishnan, R., Alfarhan, A. H., Al-Ansari, A. M., Lekshmi, R., Sreelakshmi, R., Benil, P. B., et al. (2020). Therapeutic efficacy of the root tubers of Aconitum heterophyllum and its substitute Cyperus rotundus in the amelioration of pylorus ligation induced ulcerogenic and oxidative damage in rats. Saudi J. Biol. Sci. 27, 1124–1129. doi:10.1016/j.sjbs.2019.12.025
Rong, B. S., Huang, K. L., Yuan, L. Y., Wang, J. H., Li, X., and Ma, C. J. (2021). Research progress on chemical composition and pharmacological action of aconitum. Chin. Pharm. Aff. 35 (08), 932–947. doi:10.16153/j.1002-7777.2021.08.012
Schwartzberg, L. S., Arena, F. P., Bienvenu, B. J., Kaplan, E. H., Camacho, L. H., Campos, L. T., et al. (2017). A randomized, open-label, safety and exploratory efficacy study of kanglaite injection (KLTi) plus gemcitabine versus gemcitabine in patients with advanced pancreatic cancer. J. Cancer 8, 1872–1883. doi:10.7150/jca.15407
Seo, W. G., Pae, H. O., Chai, K. Y., Yun, Y. G., Kwon, T. H., and Chung, H. T. (2000). Inhibitory effects of methanol extract of seeds of Job's Tears (Coix lachryma-jobi L. var. ma-yuen) on nitric oxide and superoxide production in RAW 264.7 macrophages. Immunopharmacol. Immunotoxicol. 22, 545–554. doi:10.3109/08923970009026011
Song, S. S., Bao, Y. R., Zhao, H. J., Xiang, Z., Meng, X. S., Wang, S., et al. (2018). Effective components compatibility of yiyi Fuzi Baijiang powder against hepatoma cells SMMC-7721. Cent. South Pharm. 16 (2), 161–166.
Sui, H., Zhang, L., Gu, K. J., Chai, N., Ji, Q., Zhou, L. H., et al. (2020). YYFZBJS ameliorates colorectal cancer progression in ApcMin/+ mice by remodeling gut microbiota and inhibiting regulatory T-cell generation. Cell Commun. Signal. CCS 18 (1), 113. doi:10.1186/s12964-020-00596-9
Tai, C., El-Shazly, M., Tsai, Y., Csupor, D., Hohmann, J., Wu, Y., et al. (2021). Uncovering. Modern clinical applications of Fuzi and fuzi-based formulas: a nationwide descriptive study with market basket analysis. Front. Pharmacol. 12, 641530. doi:10.3389/fphar.2021.641530
Thawabteh, A. M., Thawabteh, A., Lelario, F., Bufo, S. A., and Scrano, L. (2021). Classification, toxicity and bioactivity of natural diterpenoid alkaloids. Molecules 26 (13), 4103. doi:10.3390/molecules26134103
Wada, K., and Yamashita, H. (2019). Cytotoxic effects of diterpenoid alkaloids against human cancer cells. Molecules 24 (12), 2317. doi:10.3390/molecules24122317
Wang, M., Chen, S., He, X., Yuan, Y., and Wei, X. (2024). Targeting inflammation as cancer therapy. J. Hematol. Oncol. 17 (1), 13. doi:10.1186/s13045-024-01528-7
Wangchuk, P., Navarro, S., Shepherd, C., Keller, P. A., Pyne, S. G., and Loukas, A. (2015). Diterpenoid alkaloids of Aconitum laciniatum and mitigation of inflammation by 14-O-acetylneoline in a murine model of ulcerative colitis. Sci. Rep. 5, 12845. doi:10.1038/srep12845
Wei, J. Q., Yan, W. j., Ning, G. L., and Cui, X. H. (2013). 120 cases of anal sinusitis treated by yiyi Fuzi Baijiang powder with enema. CJGMCM 28 (5), 957–958.
Williams, G. R., Dunne, R. F., Giri, S., Shachar, S. S., and Caan, B. J. (2021). Sarcopenia in the older adult with cancer. J. Clin. Oncol. official J. Am. Soc. Clin. Oncol. 39 (19), 2068–2078. doi:10.1200/JCO.21.00102
Wu, J. J., Zhu, Y. F., Guo, Z. Z., Lou, Y. M., He, S. G., Guan, Y., et al. (2018). Aconitum alkaloids, the major components of Aconitum species, affect expression of multidrug resistance-associated protein 2 and breast cancer resistance protein by activating the Nrf2-mediated signalling pathway. Phytomedicine 44, 87–97. doi:10.1016/j.phymed.2017.12.007
Wu, M., Zhang, H. Y., Liu, T. F., Sun, H. B., Wang, J. F., Cao, Y., et al. (2016). Clinical observation on the treatment of phlegm and blood stasis interrelated acne with Yiyi Fuzi Baijiang Powder. J. ANHUI Univ. Chin. Med. 35 (6), 41–43.
Xia, Q., Gao, S., Rapael Gnanamuthu, S. R., Zhuang, K., Song, Z., Zhang, Y., et al. (2021). Involvement of nrf2-HO-1/JNK-erk signaling pathways in aconitine-induced developmental toxicity, oxidative stress, and ROS-mitochondrial apoptosis in zebrafish embryos. Front. Pharmacol. 12, 642480. doi:10.3389/fphar.2021.642480
Xie, S. W., Wang, Y. N., Luo, H. Y., Lu, Z. B., Yu, L. Z., and Liu, J. S. (2017). Inhibitory effect of giganteaside D on hepatocellular carcinoma Hep 3b cells and the underlying mechanisms. J. South Med. Univ. 37 (9), 1211–1216. doi:10.3969/j.issn.1673-4254.2017.09.12
Yang, C., Zeng, X., Cheng, Z., Zhu, J., and Fu, Y. (2021). Aconitine induces TRPV2-mediated Ca(2+) influx through the p38 MAPK signal and promotes cardiomyocyte apoptosis. Evid. Based Complement. Altern. Med. 2021, 9567056. doi:10.1155/2021/9567056
Yang, J., Liu, Y., Lu, S., Sun, X., Yin, Y., Wang, K., et al. (2022). Coix seed oil regulates mitochondrial functional damage to induce apoptosis of human pancreatic cancer cells via the PTEN/PI3K/AKT signaling pathway. Mol. Biol. Rep. 49 (7), 5897–5909. doi:10.1007/s11033-022-07371-8
Yang, M., Ji, X., and Zuo, Z. (2018). Relationships between the toxicities of Radix Aconiti Lateralis. Preparata (Fuzi) and the toxicokinetics of its main diester-diterpenoid alkaloids. Toxins 10 (10), 391. doi:10.3390/toxins10100391
Yao, Y., Zhu, Y., Gao, Y., and Ren, G. (2015). Effect of ultrasonic treatment on immunological activities of polysaccharides from adlay. Int. J. Biol. Macromol. 80, 246–252. doi:10.1016/j.ijbiomac.2015.06.033
Yu, H. H., Li, M., Li, Y. B., Lei, B. B., Yuan, X., Xing, X. K., et al. (2020). Benzoylaconitine inhibits production of IL-6 and IL-8 via MAPK, Akt, NF-κB signaling in IL-1β-induced human synovial cells. Biol. Pharm. Bull. 43, 334–339. doi:10.1248/bpb.b19-00719
Zhan, Y. P., Huang, X. E., Cao, J., Lu, Y. Y., Wu, X. Y., Liu, J., et al. (2012). Clinical safety and efficacy of Kanglaite® (Coix Seed Oil) injection combined with chemotherapy in treating patients with gastric cancer. Asian Pac J. Cancer Prev. 13, 5319–5321. doi:10.7314/apjcp.2012.13.10.5319
Zhang, L., Sui, H., Fu, J., Xu, R. J., Chen, J. H., Zhang, W. X., et al. (2020a). Effects of classical prescription on intestinal adenoma and intestinal microflora in conditional gene knockout APCMin/+mice. CJTCMP 35 (5), 2327–2332.
Zhang, P., Meng, X., Tang, X., Ren, L., and Liang, J. (2019b). The effect of a coix seed oil injection on cancer pain relief. Support Care Cancer 27, 461–465. doi:10.1007/s00520-018-4313-z
Zhang, S., and Chen, J. (2021). Exploration on the clinical application of modified yiyi Fuzi Baijiang powder. China J. Traditional Chin. Med. Pharm. 36 (01), 263–266.
Zhang, S. X., Song, X. F., Zhang, X. A., and An, Y. K. (2019a). Clinical effects of Yiyi Fuzi Baijiang Powder combined with mesalazine on patients with ulcerative colitis. Chin. Tradit. Pat. Med. 41 (11), 2642–2646. doi:10.3969/j.issn.1001-1528.2019.11.017
Zhang, T., Li, Q., Li, K., Li, Y., Li, J., Wang, G., et al. (2008). Antitumor effects of saponin extract from Patrinia villosa (Thunb.) Juss on mice bearing U14 cervical cancer. Phytother. Res. 22, 640–645. doi:10.1002/ptr.2354
Zhang, X., Li, D., Xue, X., Zhang, Y., Zhang, J., Huang, C., et al. (2018). First total synthesis of a novel amide alkaloid derived from Aconitum taipeicum and its anticancer activity. Nat. Prod. Res. 32, 128–132. doi:10.1080/14786419.2017.1340283
Zhang, Y., Chai, N., Wei, Z., Li, Z., Zhang, L., Zhang, M., et al. (2022). YYFZBJS inhibits colorectal tumorigenesis by enhancing Tregs-induced immunosuppression through HIF-1α mediated hypoxia in vivo and in vitro. Phytomedicine 98, 153917. doi:10.1016/j.phymed.2021.153917
Zhang, Y., Gong, H. J., Han, G., Cao, Y., Zhang, Y. Y., and Zhang, X. (2020b). Clinical efficacy of YYFZBJS to accelerate the recovery of colorectal cancer surgery patients. J. Colorectal and Anal Surg. 26, 13–14. doi:10.19668/j.cnki.issn1674-0491.2020.S2.0013
Zhou, Z., Liu, L. P., Liu, H. T., and Lei, P. (2016). Optimization of extraction technology of yiyi Fuzi Baijiang powder by orthogonal design. J. Chin. Med. Mater. 39 (4), 829–832. doi:10.13863/j.issn1001-4454.2016.04.033
Zhou, Z., Liu, W. Y., Lou, Y. Y., Tang, J. Z., Wang, J. R., and Chen, J. D. (2018). Study on the inhibition of drug resistance of colorectal cancer HCT-8/5-FU cells by Herba Patriniae. Fujian J. TCM 49 (2), 33–35.
Zou, W., Wen, X., Zheng, Y., Xiao, Z., Luo, J., Chen, S., et al. (2015). Metabolomic study on the preventive effect of Patrinia scabiosaefolia fisch on multipathogen induced pelvic inflammatory disease in rats. Evid. Based Complement. Altern. Med. 2015, 170792. doi:10.1155/2015/170792
Glossary
TCM traditional Chinese medicine
YYFZBJS Yiyi Fuzi Baijiang San
CSO Coix Seed Oil
Nrf2 Nuclear Factor erythroid 2-Related Factor 2
HO-1 heme oxygenase 1
Bax BCL2-associated X protein
Bcl-2 B-cell lymphoma/leukemia-2 gene
PI3K Phosphoinositide-3 kinase
MAPK mitogen-activated protein kinase
NF-κB nuclear factor kappa-B
AMPK Adenosine 5‘-monophosphate -activated protein kinase
SePP1 selenoprotein
P plasma, 1
apoER2 apolipoprotein E receptor 2
MAFbx muscle atrophy F-box protein
Cbl-b Casitas B-lineage lymphoma-b
BCAT2 branched-chain amino acid aminotransferase 2
MRP2 multidrug resistance-associated protein 2
ROS reactive oxygen species
UC Ulcerative colitis
INOS Inducible Nitric Oxide Synthase
COX Cyclooxygenase
JNK c-Jun N-terminal kinase
CRC colorectal cancer
HIF-1α hypoxia inducible factor-1α
ACF preneoplastic aberrant crypt foci
EMT epithelial-mesenchymal transition
TNF tumor necrosis factor
PMIEE Patrinia monoterpene iridoid ether esters
PID Pelvic inflammatory disease
NAFLD Nonalcoholic Fatty Liver Disease
TC total cholesterol
TG triacylglycerol
HDL high-density lipoprotein
RyR2 Rabbit Ryanodine receptor 2
SERCA sarco/endoplasmic reticulum Ca2+-ATPase
LPC lysophosphatidylcholine
Treg regulatory T cell
PCNA proliferating cell nuclear antigen
GSH-Px Glutathione peroxidase
CAT Catalase
SOD Super Oxide Dimutese
VEGF vascular endothlial growth factor
MDA malonaldehyde
STAT3 signal transducer and activator of transcription 3
Keywords: Yiyi Fuzi Baijiang San, traditional Chinese medicine, mechanism, active ingredients, clinical application
Citation: Zhang Y, Zhang L, Chai N, Wan Z and Sui H (2024) Inspired by an ancient Chinese Medicine prescription: the modern significance and potential of Yiyi Fuzi Baijiang San in treating diseases. Front. Pharmacol. 15:1465387. doi: 10.3389/fphar.2024.1465387
Received: 16 July 2024; Accepted: 24 October 2024;
Published: 07 November 2024.
Edited by:
Chien-shan Cheng, Shanghai Jiao Tong University, ChinaReviewed by:
Jinjun Wu, Guangzhou University of Chinese Medicine, ChinaQing Ji, Shanghai University of Traditional Chinese Medicine, China
Chiwei Chen, Guangzhou University of Chinese Medicine, China
Robert David Hoffman, Zhejiang Chinese Medical University, China
Copyright © 2024 Zhang, Zhang, Chai, Wan and Sui. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hua Sui, c3loMDgwOEAxNjMuY29t
†These authors have contributed equally to this work
 Yuli Zhang
Yuli Zhang Lu Zhang3†
Lu Zhang3† Hua Sui
Hua Sui