- 1Department of Agriculture and Animal Health, College of Agriculture and Environmental Sciences, University of South Africa, Florida, South Africa
- 2Department of Plant and Soil Sciences, Faculty of Science, Engineering and Agriculture, University of Venda, Thohoyandou, South Africa
- 3Department of Food Science and Technology, Faculty of Science, Engineering and Agriculture, University of Venda, Thohoyandou, South Africa
- 4Department of Biochemistry and Microbiology, Faculty of Science, Engineering and Agriculture, University of Venda, Thohoyandou, South Africa
- 5School of Agricultural, Earth and Environmental Sciences, University of Kwa-Zulu Natal, Pietermaritzburg, South Africa
- 6Biochemistry and Biotechnology, Sefako Makgatho Health Sciences University, Ga-Rankuwa, South Africa
Herbal tea is a rich source of secondary metabolites which are reputed to have medicinal and nutritional efficacy. These secondary metabolites are influenced by the abiotic and biotic stresses that improve the production of herbal teas in terms of biomass production, accumulation and partitioning of assimilates of compounds. In this study, various examples of herbal teas have been shown to respond differently to secondary metabolites affected by environmental factors. Thus, the meta-analysis of this study confirms that different herbal teas' response to environmental factors depends on the type of species, cultivar, and the degree of shade that the plant is exposed. It is also evident that the metabolic processes are also known to optimize the production of secondary metabolites which can thus be achieved by manipulating agronomic practices on herbal teas. The different phenolic compound in herbal teas possesses the antioxidant, antimicrobial, antiatherosclerosis, anti-inflammatory, antimutagenic, antitumor, antidiabetic and antiviral activities that are important in managing chronic diseases associated with lifestyle. It can be precluded that more studies should be conducted to establish interactive responses of biotic and abiotic environmental factors on quality attributes of herbal teas.
Introduction
In its 2014–2023 strategy, the World Health Organization (WHO) aims to promote utilization of traditional medicines, including herbal medicine, with the goal of keeping populations healthy by providing access to efficient and reasonably priced alternatives to medicine and by offering healthcare options that are consistent with people's cultural practices (World Health Organization, 2013). Therefore, there is a need to develop new herbal teas from the underutilized herbal plants if we are to achieve WHO strategy. The consumption of herbal teas is gaining attraction in recent years because most of them are rich in natural bioactive components such as alkaloids, carotenoids, coumarins, flavonoids, polyacetylenes, and terpenoids (Chandrasekara and Shahidi, 2018).
Herbal tea is an aqueous infusion of different plant materials in hot or cold water to extract the phytochemical constituents (Poswal et al., 2019). It is produced from different plant parts, such as flowers, roots, barks, and seeds, compared to the traditional leaves of black tea. Herbal teas exist in different taste, color, and smell depending on the blend's preparation of other plants (Phelan and Rees, 2003; Malongane et al., 2020) and are referred to as “tisanes” (Sehgal, 2022). Its brewing process only takes 5–10 min, and it is served immediately (Omogbai and Ikenebomeh, 2013, Sharpe et al., 2016). Herbal tea has low calories and can be used as a relaxing drink.
A high intake of herbal tea as a beverage reduces the risk of chronic diseases due to antioxidant, antiatherosclerosis, anti-inflammatory, antimutagenic, antitumor, and antiviral activities (Jurendić and Ščetar, 2021). Herbal tea has the potential to scavenge free radicals from sticking together (Prajapati et al., 2022) because of its low tannin content. The tea contains a high concentration of polyphenols (Shao et al., 2018). According to McGraw et al. (2007), herbal tea is a healthier beverage as it does not contain caffeine and pyridoxine compared to tea.
Abiotic and biotic stresses such as light intensity, temperature, water availability, type, and soil composition have an impact on the productivity and the quality attributes of herbal teas (Akula and Ravishankar, 2011). Environmental stresses which include inappropriate radiation or temperature as well as drought trigger the over-production of reactive oxidative species (ROS), which involves superoxide (O2−), singlet oxygen (•O2), hydroxyl ion (OH–), and H2O2 (Smirnoff, 1993). Hence, plants can potentially divert their photosynthetic resources to defense mechanisms against detrimental environmental factors to promote vegetative (biomass) and reproductive stages (secondary metabolites) (Wahid et al., 2007; Pezzani et al., 2017). Therefore, herbal teas use secondary metabolites as defense mechanisms to scavenge and detoxify ROS using enzymes such as CAT, peroxidase or superoxide dismutase and decompose H2O2 to H2O at different cellular locations.
Currently, the vast majority of herbal teas are still collected in the wild for various medicinal attributes and variation in quality characteristics is of great concern. According to reports, seasonal variations as well as varying climatic conditions affect the quality of herbal teas throughout the year (Ghasemzadeh et al., 2010; Mudau and Makunga, 2018). Carbon-based secondary metabolites have been discovered to increase in Labisia pumila plants only when climatic conditions promote the production, accumulation, and partitioning of non-structural carbohydrates (Ibrahim et al., 2011). There is a need for the domestication of herbal teas for commercialization as a healthy beverage to protect the plants from becoming extinct (Mashimbye et al., 2006) and maximize the quality of secondary metabolites (Mudau et al., 2006). Previous studies have documented four major factors that affect parameters that determine quality in herbal tea production, namely: environmental conditions (Tshivhandekano et al., 2013; MacAlister et al., 2020; Ramphinwa et al., 2022), cultural practices (Yilmaz et al., 2004; Mudau et al., 2006; Hlahla, 2010; Bandara, 2012; Mphangwe, 2012; Mohale et al., 2018), cultivars (Owuor et al., 2000) and seasonal variation (Mudau and Makunga, 2018). Thus, environmental factors and agronomic practices have an impact on the quality parameters of tea (Zhang et al., 2014).
Although drinking black, green, and oolong tea (Camellia sinensis) has been linked to various health advantages, there is relatively little research on the effects of most herbal teas. Despite being particularly rich sources of polyphenols, recommendations for the intake of plant-based beverages such as herbal teas are still lacking. Recently, Malongane et al. (2017), claimed that polyphenolic compounds in herbal tea might have synergistic potential, which aids in managing medical conditions when used alongside conventional medicines. The present review gathers data on eco-physiological response of secondary metabolites of herbal tea to better understand the interrelationships between herbal tea and health considering the shift toward integrated medicine and increased interest in herbal tea and health. Drinking herbal tea has been linked to some therapeutic and preventative advantages and this is according to a new scoping review that compiled data from 21 studies, including observational studies (Poswal et al., 2019).
The overall main objective of the present work is to provide a review of production, health benefits and antioxidant properties and how eco-physiological parameters and environmental factors influence accumulation and distribution of secondary metabolites of herbal tea. Thus, the use of locally available herbal plants to develop new herbal tea or infusion could have a significant socio-economic impact on the herbal tea producers in terms of job creation, introducing the product into local and international market as well as improved nutrition and health of consumers since herbal tea is rich in secondary metabolites. Therefore, there is a need to develop strategies to optimize biotic and abiotic factors to promote effective secondary metabolites on a large scale of herbal tea production.
Methodology
The methodology of the current study is divided into four phases to achieve the research objectives, viz: definition of key terms, a mixed methods review to determine the current state of herbal teas and identify gaps in their mainstreaming, a review to quantify the amount of knowledge on how light, shading, season, soil composition, and temperature affect secondary metabolite accumulation in herbal teas. The following sections outline the details of four different stages.
Phase 1: Definitions of key phrases.
Four definitions have been used in this article to describe herbal tea's medicinal properties and functions. Key phrases to be defined include herbal teas, medicinal plants, antioxidants, and polyphenols. Original papers and review articles have been used to gather information to describe the key terms.
Phase 2: Identifying medicinal properties and their functions in herbal teas. Google was used throughout the study to gather information.
Phase 3: Identifying bioactive compounds and their functions in herbal teas.
Phase 4: A mixed-method review approach including quantitative and qualitative research has been used in this phase to gather information about production, processing, biological activities and how environmental factors affect the accumulation of secondary metabolites of herbal teas.
Search strategy
This section was created using a two-step approach to provide an in-depth assessment of herbal teas' nutraceutical, medicinal properties, and priorities.
First stage: Planning the review
Research questions were developed in this stage and were as follows:
1. The current review aims to gather information about nutraceutical properties and environmental factors that influence the quality of herbal teas.
2. What are the medicinal and antioxidant characteristics of herbal teas?
3. Which bioactive compounds are related to the functional characteristics of herbal teas?
4. How do environmental factors affect the quality of herbal teas?
The second stage: A review
Information about the nutraceutical and medicinal benefits of herbal teas was collected through reviewing different research articles. The main objective was to identify herbal teas' medicinal properties and bioactive compounds. Therefore, it was significant to include nutraceutical and medicinal properties attributes of herbal teas. Different attributes identified included: antimicrobial, antioxidant, hepatoprotection, anti-allergy, antigenotoxic, antiplasmodial, cytotoxic, antispasmodic, cardioactive, anticough, anti-diabetic, anti-inflammatory and antinociceptive, antifungal, toxicological, anti-diabetes, anti-cancer, antinociceptive, antimelanogenic, wound healing, memory enhancing, antibacterial, antimycotic, antitumor, antiviral, gastrointestinal protective, hepatoprotective, and chemopreventive, anti-diarrhea, neuro-protective, anti-allergic, improves cardiac health antiseptic and stimulant, antimicrobial, sedative properties, anti-cholesterol, anti-hypertensive, analgesic, chemoprotective activities and anti-aging.
Results and discussion
Definitions of key phrases
Herbal teas are an aqueous mixture of different plant materials in hot or cold water to extract the phytochemical constituents for an unspecified period. They are a popular worldwide beverage and are utilized as a therapeutic vehicle in different forms of traditional medicine (Poswal et al., 2019).
A medicinal plant contains active compounds or therapeutic properties in one or more organs that can benefit the human body pharmacologically (Rasool Hassan, 2012; Namdeo, 2018). Since prehistoric times, medicinal plants, also known as medicinal herbs, have been discovered and used in traditional medicine practices. These herbs are preferred by non-industrialized societies, owing to their lower cost compared to modern medications.
Antioxidants are groups of compounds that act in the cell to neutralize free radicals and reactive oxygen species (ROS) (Göçer et al., 2013; Çakmakçi et al., 2015). A free radical is a highly charged and unstable carbon or oxygen atom with an unpaired electron. Lipids, proteins, and carbohydrates may all produce free radicals.
Polyphenols are secondary metabolites produced by higher plants that have potential health benefits for humans, primarily as antioxidants, anti-allergic, anti-inflammatory, anticancer, antihypertensive, and antimicrobial agents (Fraga et al., 2019). They are essential in plant-pathogen and animal defense, herbivore aggression, and stress response to a wide range of biotic and abiotic stressors.
Characteristics of searched literature
Diversity of functional herbal teas
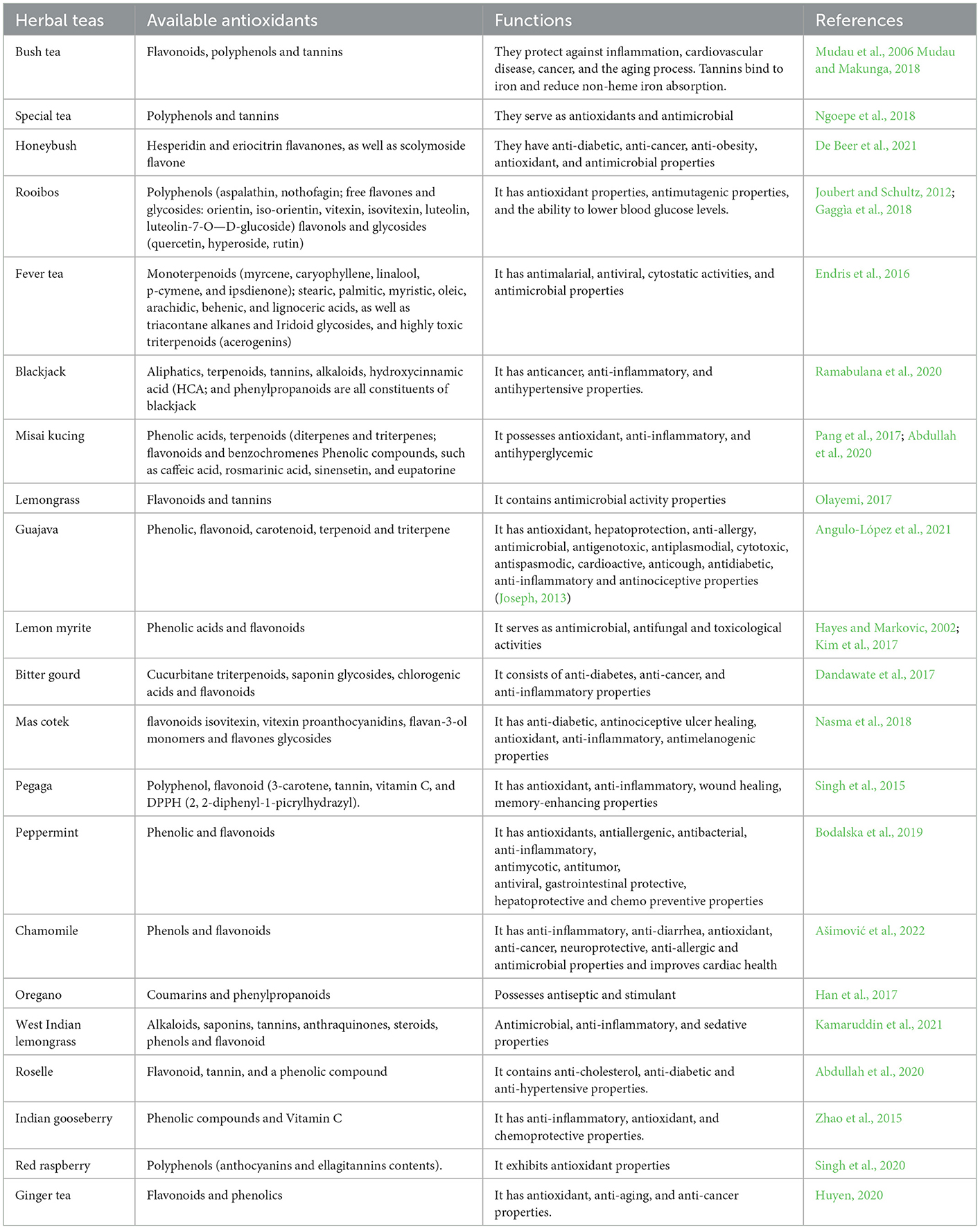
The research study listed 26 herbal teas belonging to 17 different species sampled worldwide (Table 1). The regions were presented by the sampled articles which include South Africa (0.15%), Africa (0.04%), South America (0.04%), South-East America (0.04%), South East Asia (0.12%), Algeria (0.04%), Germany (0.04%), Asia (0.15%), Europe (0.08%), North America, India, South-East Europe, Asia, Mexico (0.04%), Euro Siberian (0.04%), Irano Siberian (0.04%), North Asia, Saudi Arabia, East Europe (0.04%), North Asia (0.04), China (0.04%), South India (0.04%), and Sri-Lanka (0.04%).
Processing of herbal teas
Total concentration of polyphenol, antioxidants and tannin content of herbal tea have been reported to be determined by extraction conditions, variety, extracting solvent and processing methods (Villa-Rodriguez et al., 2018; Vural et al., 2020). Differences in chemical constituents and biological activity of plant products are caused by different processing methods and determining how to assess the impact of post-harvest treatment. Chemical and biological testing are essential for improving the quality control of herbal tea products (Chao et al., 2017). However, Turkmen et al. (2009), discovered the high concentration of polyphenols and antioxidants activity on black tea when extracted over extended period using aqueous acetone compared to lower concentration of polyphenols and antioxidant which were influenced by short period of time during extraction using absolute acetone.
Drying is a thermal process in the production of herbal teas which may result in significant losses of bioactive ingredients, reducing the health benefits and quality of the products (Mbondo et al., 2018; Monika and Selvakumar, 2019). The content of the desired components in tea infusion may be affected significantly by the brewing conditions such as quality, volume, and temperature of water and brewing time (Nikniaz et al., 2016; Sharpe et al., 2016).
Polyphenols of herbal teas
Different polyphenols substances, such as flavonoids, tannins, phenolic acids, chlorogenic acids etc., have been reported in herbal teas; however, different parts of herbal teas react differently to different polyphenols (Table 2). Herbal teas have medicinal attributes due to their high concentration of polyphenols (Koch et al., 2020), which is well known to have a wide range of advantageous biochemical and physiological properties (Koch et al., 2020; Afrin et al., 2022). Herbal teas use these bioactive compounds as their defense against environmental stresses, herbivores, and pathogen attack. Hence, the nature of growing conditions and the amount of stress that the plant is subjected to, determine the concentration of secondary metabolites (Akula and Ravishankar, 2011; Mohale et al., 2018). Polyphenols of herbal teas are found on leaves, flowers, twigs, roots, and rhizomes.
Bush tea has been reported to contain 5-hydroxy-6,7,8,3′,4′,5′-hexamethoxy flavon-3-ol (Mashimbye et al., 2006), 3-0-demethyldigicitrin, 5,6,7,8,3′,4′-hexamethoxyflavone, and quercetin, as well as other polyphenols, tannins, and antioxidants (Mudau et al., 2006, 2007a; Tshivhandekano et al., 2018). The special tea' leaves and fruits has been reported to have high concentration of total phenolic compound and total antioxidant activity (Mamphiswana et al., 2010; Suleman et al., 2022; Mfengu et al., 2021). The unfermented leaves of honey bush tea have been discovered to contain polyphenol compounds which include pinitol, shikimic acid, p-coumaric acid, 4-glucosyltyrosol, epigallocatechin gallate, the isoflavone orobol, flavanones hesperedin, narirutin, and eriocitrin, a glycosylated flavan, the flavones luteolin, 5-deoxyluteolin, and scolymoside, the xanthone mangiferin, and the flavonol C-6-glucosylkaempferol (Dube et al., 2017; Ajuwon et al., 2018). Rooibos has been characterized by a diverse range of phenolic compounds, including dihydrochalcones (aspalathin, aspalalinin, and nothofagin), flavones (orientin, iso-orientin, vitexin, isovitexin, chrysoeriol, and luteolin), flavonols (quercetin, isoquercitrin, hyperoside, and rutin), and phenolic (Ajuwon et al., 2018). Fever tea has been reported to have volatile oil as well as various monoterpenoids which include myrcene, caryophyllene, linalool, p-cymene and ipsdienone (Mokoka, 2007; Endris et al., 2016).
Endris et al. (2016) and Chawafambira (2021), discovered that leaves of fever tea produce numerous polyphenolic compouds such as of stearic, palmitic, myristic, oleic, arachidic, behenic, and lignoceric acids and triacontane alkanes. Misai kucing possess pharmacological properties due to the presence of different groups of phenolic acids including terpenoids (diterpenes and triterpenes) (Abdullah et al., 2020), flavonoids (Pang et al., 2017), and benzochromenes (Abdullah et al., 2020). Lemon grass possess different compounds as hydrocarbon terpenes, alcohols, ketones, esters due to different geographical origin (Majewska et al., 2019). Guajava contains significant biological activities caused by the presence of phenolic, flavonoid, carotenoid, terpenoid and triterpene (Angulo-López et al., 2021).
The high levels of antioxidant activity influenced by phenolic compounds contents as phenolic acids and flavonoids are found on the leaves of lemon myrite (Kim et al., 2017). Triterpene, proteid, steroid, alkaloid, inorganic, lipid, and phenolic compounds are the major compounds of bitter gourd which are responsible for antidiabetic activities (Grover and Yadav, 2004; Chung et al., 2018). Mascotek has been characterized by various phenolic compounds including flavonoids, saponins, tannins, steroids, and terpenes (Nasma et al., 2018). Peppermint has been discovered to possess flavonoids such as flavanone aglycone eriodictyol and glycosides eriocitrin (eriodictyol-7-O-rutinoside), hesperidin (hesperetin-7-O-rutinoside) and naringenin-7-O glucoside, the flavone aglycone luteolin and the glycosides isorhoifolin (apigenin-7-O-rutinoside) and luteolin-7-O-glucoside (Bodalska et al., 2019). The extract of pegaga consist of bioactive compounds such as plant sterols, flavonoids, and other components with no known pharmacological activity (Nazmi and Sarbon, 2020).
Chamomile have been reported to contain numerous bioactive phenolic compounds which are coumarins: (herniarin, umbelliferone; phenylpropanoids: chlorogenic acid, caffeic acid; flavones: apigenin, apigenin, 7-O-glucoside, luteolin, luteolin-7-O-glucoside; flavonols: quercetin, rutin, and flavanone: naringenin) and are found in chamomile extract (Gupta et al., 2010; Bayliak et al., 2021). The major compounds of oregano are caryophyllene, spathulenol, germacrene-D, and aterpineol (Sahin et al., 2004). The volatile oil from Cymbopogon citratus consist of volatile oil which is characterized by monoterpene hydrocarbons (Adesegun et al., 2013; Oladeji et al., 2019). The fraction of monoterpene has been classified by a high percentage content of geranial (39.53%), neral (33.31%), myrecene (11.41%), and other sesquiterpene (0.78%) (Wright et al., 2009; Moreira et al., 2010).
Da-Costa-Rocha et al. (2014), discovered that the extracts of roselle consist of a high percentage of organic acids, including citric acid, hydroxycitric acid, hibiscus acid, malic and tartaric acids as major compounds, and oxalic and ascorbic acid as minor compounds. Goose berry extracts are rich source of polyphenols which are responsible for cytotoxic activity against cervical and ovarian cancer cells (De Wet et al., 2012). Red raspberry possesses two major polyphenols such as anthocyanin and ellagitannins content (Singh et al., 2020). Ginger is a rich source of phenolic components which are important food material and can be served as cheap (Gbenga-Fabusiwa et al., 2018).
Biological activities of herbal teas
Medicinal properties of herbal teas are summarized in Table 2 and has been reported by different researchers.
Antioxidant activity
Herbal teas, which are herb extracts, are popular because of their fragrance and antioxidative properties (Aoshima et al., 2007; Farzaneh and Carvalho, 2015; Jin et al., 2016). Antioxidants are substances that have been shown to significantly neutralize reactive oxygen species (ROS), which are oxygen-derived free radicals that cause degenerative diseases such as superoxide anion, hydroxyl radicals, and nitric oxides (Malongane et al., 2017). In vitro studies revealed that ethanolic bush tea extract has strong antioxidant activity and inhibition of DPPH was found to be 81.6% when the lowest concentration was used to test antioxidant activity (Mavundza et al., 2007). Moreover, Bahadori et al. (2020) demonstrated that Stachys byzantina and Stachys iberica exhibited DPPH scavenging activity ranging from 26 to 125 mg TE/g extracts.
Antidiabetic activity
According to Martínez-Solís et al. (2021), diabetes mellitus has been treated by herbal teas and infusions. Diabetes mellitus is a chronic condition in which the body's ability to control the amount of glucose in the blood is impaired (DeFronzo et al., 2015). This is due to pancreatic insulin resistance or a lack of insulin secretion. Phenolic compounds in herbal teas such as chlorogenic acid, 1,3-dicaffeoylquinic acid, and hydroxylcinnamic acid, have been linked to diabetes and obesity prevention (Joubert and Schultz, 2012). Chellan et al. (2012) reported that herbal teas could be used to treat metabolic irregularities associated with diabetes by increasing glucose utilization in insulin-responsive tissues. Previous studies have also demonstrated anti-diabetic, antioxidant, antilipidemic, and antinociceptive effects using aqueous and alcoholic extracts of Annona muricata, Annona squamosa, Annona stenophylla, Annona macroprophyllata, and Annona diversifolia and this is attributed to the presence of phenolic compounds in their leaves (Martínez-Solís et al., 2021).
Antimicrobial activities
Herbal teas have been reported to have antimicrobial activities against gram-positive and -negative bacteria and yeast when they are used alone (Hacioglu et al., 2017). Depending on the antibiotic or type of tea, the synergistic, additive, or antagonistic effects of herbal teas with antibiotics were observed. Thus, using herbal teas alone or in combination with chemical antimicrobials may be a viable alternative treatment strategy for a wide range of pathogenic microorganisms. Generally, antimicrobial activities decrease with the extent of tea fermentation, implying that green tea is more active than black tea (Nibir et al., 2017). Green tea catechins, particularly epigallocatechingallate (EGCG) and epicatechingallate (ECG), have antibacterial properties against Gram-positive and Gram- bacteria negative bacteria (Bancirova, 2010; Ignasimuthu et al., 2019).
Green tea can help prevent tooth decay by inhibiting oral bacteria (Zayed et al., 2021). Xia et al. (2021) discovered that combining white tea and pepper mint resulted in synergistic antibacterial activity against four strains, two of which were gram-positive (S. argenteus and B. halotolerans) and two of which were gram-negative (E. coli and P. aeruginosa). Bush tea inhibits microorganisms such as Staphylococcus aureus, Bacillus cereus, Enterococcus, Escherichia coli, and Mycobacterium smegmatis.
Anti-mutagenic/anti-carcinogenic activities
The antimutagenic assay revealed that unfermented rooibos and honey bush tea extracts had a strong antimutagenic effect against both metabolically activated carcinogens, antimutagenic activity against 2-acetylaminofluorene (2-AAF) and (aflatoxin) AFB1 (Marnewick et al., 2000; Chaudhary et al., 2021). Carabajal et al. (2017), also discovered that antimutagenic activity of the freeze-dried plant infusions tested against a direct-acting mutagen (4-NPD), with at least three herbal mixtures resulting to a positive response.
Environmental factors that affect the production and accumulation of secondary metabolites of herbal teas
Abiotic and biotic factors, including ultraviolet irradiation, high light, temperature, pathogen attack, wounding, herbicide, and nutrient deficiency have been reported to determine the concentration of different metabolites in herbal plants (Akula and Ravishankar, 2011; Mohale et al., 2018). Plants are a rich source of natural compounds with diverse bioactivities (Masike et al., 2017). These organic compounds are divided into two categories: primary and secondary metabolites and used by plants as defense mechanisms against biotic and abiotic (Akula and Ravishankar, 2011; Mohale et al., 2018). The quality of herbal teas remains one of the constant critical aspects in determining tea's price (Ravichandran and Parthiban, 1998; Tshivhandekano et al., 2018), industrialization, and exportation (Mudau et al., 2007b). Herbal tea's quality is determined by active secondary metabolites present, viz., flavonoids, polyphenols, and tannins (Mathivha et al., 2020). Thus, compounds such as polyphenols, flavonols, and tannins are the main indicators of the medicinal potential of herbal teas due to their antioxidant activities (Hirasawa et al., 2002; Mudau et al., 2007b; Poswal et al., 2019).
Light
Light is a critical resource for plants (Ghasemzadeh et al., 2010; Rihan et al., 2020) and competition for light under shade affect growth and development of herbal teas (Kumar et al., 2013a; Ramphinwa et al., 2022). Plant growth and development, photosynthetic rate, and production of both primary and secondary metabolites are all influenced by light (Zhang et al., 2014). Currently, the light requirements of herbal teas are not well documented. Although it is difficult to define details of the ideal range of shadow required by tea plants, 50% of diffused sunshine is generally necessary for optimal physiological activity of Camellia sinensis tea (den Braber et al., 2011; Tshivhandekano et al., 2013). Shade/low irradiance enhances flavonoid synthesis and other bioactive compounds in ginger production (Ghasemzadeh et al., 2010).
Photosynthetically Active Radiation (PAR) is a waveband that has a significant influence on plants' growth, development (Proutsos et al., 2022) and accumulation of secondary metabolites (Ghasemzadeh et al., 2010). It is also known as the intercepted radiation that ranges between 400 to 700-nanometre wavebands (Mubvuma, 2018; Rihan et al., 2020). Appropriate intercepted radiation and utilization are of great importance in herbal tea production. Bush tea plants exposed to 80% white shade net accumulated more chlorogenic acid (CGAs) than plants exposed to other shade nets (i.e., 80% black, green shade nets and full sunlight) (Ramphinwa et al., 2022). These results could be attributed to the accumulation of CGAs caused by low temperatures and light intensities under 80% white shade net. The white shade net might have induced a higher number of chlorogenic acids than other shade nets due to the amount of light penetrating it. These findings are consistent with those of Karimi et al. (2013), who found that when different varieties of Labisia pumila Benth are exposed to high light intensities, tend to accumulate more phenolic compounds such as gallic acid, caffeic acid, and flavonoids such as quercetin, rutin, myricetin, kaempferol, and naringin.
Contradictory, higher oxygenated monoterpene and sesquiterpene components such as citronellol, geranyl acetate, linalool and trans-rose oxide were recorded in an open field compared to 25 and 50% shade levels. Kumar et al. (2014) and Rezai et al. (2018), also reported that oxygenated monoterpenes and sesquiterpene components decreased with shade levels in S. sclarea and T. minuta. Light might have influenced these results as a critical component that plays a significant role in producing secondary metabolites that vary due to the plant's metabolic processes and physiology. Variation of light intensities revealed plant morphological and physiological changes, which significantly impacted the herb's medicinal compounds (Idrees et al., 2018). Therefore, different plants respond differently to light intensity, resulting in differences in secondary metabolite production (Ibrahim et al., 2011; Ni et al., 2020).
Herbal plants utilized in traditional therapeutic techniques are frequently produced in places with significant UV exposure, and there are no defined conventional agricultural practices (Makola et al., 2016a). In such conditions, photoisomerization reactions of bioactive compounds are expected to be simple, putting active molecules at risk. As a result, it is critical to investigate the environmental effects of plants with therapeutic properties. Plants naturally produce cinnamic acids in a trans-configuration, which changes to a cis configuration when exposed to UV light (Makola et al., 2016b). These results concur with those of Masike et al. (2017) and Nobela et al. (2018), who reported the increasing of the content of secondary metabolites containing cinnamic acids through UV light. Chrysanthemum's concentrations of flavonoids and phenolic acids increased in response to increased UV-B radiation (Ma et al., 2016). These results might have been attributed to an increase in UV radiation associated with the amount of solar radiation received by the plants (Naghiloo et al., 2012).
Shading
Plant shading is caused primarily by dense plant populations, intercropping, planting geometry, and excessive vegetative growth, and it has an impact on crop performance by reducing plant photosynthetic capacity (Kumar et al., 2013b; Zaman et al., 2022). Irradiance is one of the crucial environmental factors that affect many physiological processes in plants, such as plant growth and development, reproduction, and distribution of secondary metabolites (Kumar et al., 2013a; Zhang et al., 2015). Photoselective films, which regulate light conditions and influence medicinal plant growth and secondary metabolism, are important for optimizing secondary metabolite accumulation (Khandaker et al., 2010; Grbic et al., 2016). Black tea exposed to artificial shade resulted in a higher concentration of theaflavin, lower concentration of thearubigin, higher flavor index and taster's evaluation compared to tea grown in an open field. This hypothesis holds that removing the shade from tea gardens results in a loss of quality (Owuor et al., 1988; Zaman et al., 2022).
These findings concur with those of Morita and Tuji (2002), who reported that shading produces high-quality Gukyo and Tencha green teas in Japan. The results might have been influenced by low light intensity under shades. Hence, shading could define tea's characteristics by slowing the photosynthesis process and thus increasing the chlorophyll content (Lehlohonolo et al., 2013). These might be due to the leaves that turn dark green, and the tannin content decreases, resulting in a sweeter flavor rather than the astringent taste common in green teas under shades. These results contradict with Kumar et al. (2013b), who reported that shade regimes did not affect the accumulation of stevioside and Rebaudioside-A in the stevia plant. Thus, accumulation and partitioning of the secondary metabolite are impacted by plant species, different plant organs, and environmental conditions to which the plants are subjected (Eko et al., 2012; Mohd Yusof et al., 2021).
Seasons
Producing tea of the same quality is impractical throughout the year due to different climatic conditions and seasonal variations (Owour and Obanda, 1998; Mudau et al., 2007a, 2016). The results are in line with those of Lin-Wang et al. (2011), who reported that significant climatic changes throughout the cropping season have a significant impact on tea quality and value. These findings might have been influenced by harsh climatic conditions, which are unsuitable for producing high-quality herbal tea (Mudau et al., 2016). According to Mudau et al. (2006), variation in seasonal temperatures and vapor pressure deficit affect the quality and antioxidants in bush tea. The concentration of polyphenols in bush tea leaves collected from the wild has been recorded to be lowest in autumn, spring and highest in winter (Mudau et al., 2006; Nchabeleng et al., 2012).
Furthermore, hydrolysable tannins concentration was lowest during summer compared to autumn, spring and winter. The results might have been attributed due to drought stress (Hamilton et al., 2001; Lv et al., 2021) and low temperatures (Mudau et al., 2006) during autumn and winter. The results are in line with those of Caruso et al. (2020), who reported the highest value of hydrophilic antioxidant activity under unshaded fields, but there was no significant difference between autumn and winter cropping seasons; hence lowest was recorded in the last cropping season.
Turkmen et al. (2009) and Soni et al. (2015), further reported variation of catechin content and distribution in fresh tea leaves are influenced by harvesting season. Similar results were also discovered by Chou et al. (1999) and Salman et al. (2022), who reported that catechin content variation due to harvesting season and higher antimicrobial activity during summer cropping season resulted in a higher concentration of catechin. In Australia, higher epicatechin gallate (ECG) and epigallocatechin gallate (EGCG) levels were recorded during warmer months (Yao et al., 2005; Kashchenko et al., 2021), while EGC recorded higher levels in cooler months. As a result, the warmer season is recommended for producing high-quality black tea (Lin et al., 1996; Hossain et al., 2017). Understanding the metabolic process of herbal teas will elucidate how the variation of seasons may impact the formation and its quality.
Temperature
The effect of temperature on plant biosynthesis and phytochemical accumulation is significant (Cheynier et al., 2013; Pola et al., 2020). The physiological and biochemical processes that result in the formation of plant metabolites are influenced by temperature (Tshivhandekano et al., 2013). Low temperature restricts the accumulation of alkaloids (morphinane, phthalisoquinoline and benzylisoquinoline) in dry Papaver somniferum) (Yang et al., 2018). Similarly, Dutta et al. (2007) and Jan et al. (2021), reported the reduction of vindoline and catharanthine levels in Catharanthus roseus leaves due to low temperatures. Contradictory, different cultivars of Lupinus angustifolius increased accumulation of alkaloid when exposed to high temperature. Thakur et al. (2019), also reported that higher temperatures promote leaf senescence and concentration of the secondary metabolite in the roots of Panax quinquefolius.
Soil type and composition
The production, accumulation, and partitioning of secondary metabolites in plants have been reported to rest on a few hypotheses: carbon nutrient balance (CNB) and growth differentiation. Verma and Shukla (2015) discovered that plant growth and cell development take precedence over secondary metabolite production. Before distributing carbon and nitrogen to produce secondary metabolites, the growth requirement must be met. Nutrient-deficient in plants promotes a higher concentration of carbon-based secondary metabolites at the expense of plant growth and development than plants with enough nutrients (Radušiene et al., 2019). These findings imply that herbal tea could accumulate more secondary metabolites when plants are deprived of nutrients.
The most important determinant of the rate of secondary metabolite production is the collection of precursor molecules (Waterman and Mole, 2019). As a result, it suggests that the status of carbon-nutrient balance in plants, as determined by resource availability, has a significant influence on secondary metabolite allocation. Hence, the palatability and resistance of the plant to herbivores are affected by the allocation of secondary metabolites. The more nitrogen is invested in soil, the less the accumulation of phenolics and flavonoids in Labisia pumila Benth (Ibrahim et al., 2011). Contradictory, previous studies reported that N shortage increased the content of carbon-based secondary metabolites (CBSMs) (e.g., polyphenols, flavonoids, and triterpenoids) (Ibrahim et al., 2013; Strzemski et al., 2021; Sun et al., 2021).
Water stress
Water stress is one of the most important environmental stresses affecting plant morphological growth and development and their biochemical properties (Ashraf et al., 2018). It can increase the accumulation of secondary metabolites in a wide range of plant species. Endogenous levels of plant secondary metabolites increased in response to drought stress in several medicinal plants, including C. roseus, H. perforatum, and Artemisia annua. Drought stress, for example, increased phenolics and photosynthetic pigments while decreasing plant fresh and dry biomass in T. ammi (Azhar et al., 2011). Similar results were reported by Verma and Shukla (2015), that improved quality of secondary metabolites such as rutin, quercetin, and betulinic acid in Hypericum brasiliense, Artemisinin and Artemisia resulted in water stress. Contradictory, essential oil content (%) and yield decrease significantly with increasing the level of water stress in Origanum vulgare and Melissa officinalis (Said-Al Ahl and Hussein, 2010). Drought tolerance in plants differs from species to species (Thakur et al., 2019).
Limitation and future perspective of herbal teas
Over the past two decades, the safety and medicinal potency of herbal teas have been subject of interest. Results from different studies show that herbal teas contain phytochemicals that are beneficial while other are toxic to human health (Upadhya, 2004; Krushna et al., 2009; Gohil et al., 2010; Singh et al., 2013). There is a very limited information from the literature with regards to the safety of herbs and herbal teas, herb-herb and interconnections of herb-therapeutic drug (Chandrasekara and Shahidi, 2018). Therefore, there is a need that future research determines the toxicity and bioactivity of herbal teas.
Another challenge of herbal teas is that their market is unstructured and there is risk of poisonous products entering the market. For example, Jin et al. (2018) found 14 poisonous species used for herbal teas in the ethnobotanical surveys of traditional medicinal markets on the Dragon Boat Festival, China. Poor sourcing standards could result in such risk since there might be no quality measurement of original plant materials. Therefore, there is a need for sector to provide products that meet medical standard and not only concentrating on the amount of phytochemicals in the plant materials, but to guarantee quality and reassure consumers that the products are not contaminated (Booker and Heinrich, 2016). Moreover, authors indicated that there might be adulteration of herbal teas in the same market. A recent quality evaluation of marketed chamomile tea found that other plant materials are more likely to adulterate crude flowers than German chamomile tea bags (Guzelmeric et al., 2017).
The labeling and promotion claim for most of herbal teas are questionable, with claims of superior antioxidant activity compared with tea or coffee and the potential to treat various health disorders (McKay and Blumberg, 2007). Such claims should be supported by evidence that is sound and research based (Balentine et al., 1999). Most of these herbal teas have not been well documented especially for studies that involve human trials, therefore, such health benefit claims are not justifiable even though most of these teas have history of utilization in some traditional medicines. Poswal et al. (2019) suggested that there might be relationships between the consumption of herbal teas and low risk of thyroid disease and liver. Therefore, there is a need to conduct further research on analytical and clinical trials to evaluate the phytochemicals of herbal beverages that act in reducing the risk of disease in human as well as verifying how they promote human health (Chandrasekara and Shahidi, 2018).
Other areas of interest include synergistic combination and microencapsulation of herbal teas. Moreover, non-thermal treatments, such as high-pressure processing should also be exploited. The synergistic effect of various combinations of herbal teas has been studied. The mixture of tea with other herbs has demonstrated to synergistically improve antioxidant activities and this leads to the development of various blends of tea blends. The antioxidant activity of the synergistic mixture might offer concomitant health benefit since polyphenolic compounds available in the tea and herbs play role in the prevention of cardiovascular diseases and type 2 diabetes mellitus (Malongane et al., 2017). Mathivha et al. (2020) determined the synergistic effect of South African herbal teas namely bush tea—Athrixia phylicoides DC and special tea—Monsonia burkeana Planch. Ex Harv and reported that the mixture has high antioxidant activities that might play a role in managing lifestyle diseases such as diabetes (Etheridge and Derbyshire, 2020).
Microencapsulation is an emerging technique which leads to the protection of various components of food or functional ingredients against different processing methods since it covers them inside a polymeric or non-polymeric material and allow their controlled release under specific conditions (Choudhury et al., 2021). Moreover, microencapsulation improves the organoleptic attributes of food by camouflaging the unpleasant smell and taste as well as preventing the growth of microorganisms (Sengupta et al., 2001; Hasanvand et al., 2015). Spray and freeze drying are the extensively used methods for microencapsulation. Pasrija et al. (2015) microencapsulated polyphenolic compounds of green tea with walls of maltodextrin, b-cyclodextrin and combination of both and determined its effect on the quality of bread. Green tea extract and encapsulates added bread maintained their quality characteristics with regards to loaf volume and crumb firmness and they were almost similar to control sample. The total polyphenolic content of extract of green tea and microencapsulated bread did not differ significantly.
High pressure processing (HPP) is applied to food to eliminate vegetative microorganisms, thereby warranting microbial safety and improved shelf life of food (Guerrero-Beltrán et al., 2005). During HPP process, hydrostatic pressure varying from 100 to 800 MPa, is applied to beverages or food which leads to inactivation of microorganism by denaturing protein or cell injury (Guerrero-Beltrán et al., 2005). Moreover, HPP also retains bioactive components such as polyphenols, antioxidant activity and it does not affect small molecules such as pigments, vitamins and volatile compounds (Patras et al., 2009; Medina-Meza et al., 2015; Marszałek et al., 2017).
Kieling and Prudencio (2019) evaluated the quality characteristics of lemongrass-lime mixed beverage processed under the optimal HPP conditions during 8 weeks of storage at 4°C. The beverage was compared with the control and pasteurized sample. High pressure processed beverage retained its phenolic compounds, no significant losses of ascorbic acid observed, and physicochemical properties were closer to control sample compared with pasteurized sample. Moreover, HPP of 250 MPa for 1 min at 25°C resulted in microbiological safety, based on the inactivation tests with Listeria innocua as the target microorganism. This shows that HPP can be utilized as an alternative or substitute of thermal processing of herbal teas since it extended the shelf life of lemongrass-lime mixed beverage.
Conclusions
Herbal tea is a rich source of secondary metabolites. Environmental factors have been studied herein to determine their influence on the quality of herbal teas. Generally, abiotic and biotic stresses increase the production, accumulation and partitioning of secondary metabolites on plants at the expense of plant growth, development, and yield. In this study, various examples of herbal teas have been shown to respond differently to secondary metabolites affected by environmental factors. Therefore, this study confirms that different herbal teas' response to environmental factors depends on the type of species, cultivar, and the degree of shade that the plant is exposed to. Quality assurance is a critical step required to be achieved for the potential market for herbal teas. Hence, metabolic processes known to optimize the production of secondary metabolites can thus be achieved by manipulating agronomic practices on herbal teas. It is thus concluded that light, shading, season, soil composition, water stress and temperature play a significant role in cultivating herbal teas. The health benefits of herbal tea necessitate more effort to understand the complex interaction of biotic and abiotic environmental factors with the plant to produce secondary metabolites.
Author contributions
Conceptualization, validation, and writing-original draft preparation: MLR, MEM, and FNM. Methodology: MLR, MEM, NEM, VST, and FNM. Resources and project administration: GRAM, NEM, and FNM. Funding acquisition: MLR, NEM, GRAM, and FNM. Writing-review and editing: MLR, MEM, NEM, GRAM, VGPC, TAM, TM, VST, and FMN. All authors contributed to the article and approved the published version.
Funding
This study was funded by the University of South Africa, University of Venda, and National Research Foundation (Thuthuka Grant No. UID 118069).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Abdullah, F. I., Chua, L. S., Mohd Bohari, S. P., and Sari, E. (2020). Rationale of Orthosiphon aristatus for healing diabetic foot ulcer. Nat. Prod. Commun. 15, 1–13. doi: 10.1177/1934578X20953308
Adesegun, A. S., Folorunso, O. S., Raimi, G. O., and Funmilola, S. A. (2013). Antioxidant activity of the volatile oil of Cymbopogon citratus and its inhibition of the partially purified and characterized extracellular protease of Shigella sonnei. Department of Biochemistry, Faculty of Science, Lagos State University, Lagos, Nigeria.
Afrin, T., Haque, M. N., Mohibbullah, M., Hossain, M. A., Mortuza, M. G., Islam, A. M., et al. (2022). Antioxidant, Anticholinesterase, and Neurotrophic Potentials of Indigenous Medicinal Herbs of Bangladesh. doi: 10.5455/jabet.2022.d148
Ajuwon, O. R., Ayeleso, A. O., and Adefolaju, G. A. (2018). The potential of South African herbal tisanes, rooibos and honeybush in the management of type 2 diabetes mellitus. Molecule. 23, 3207. doi: 10.3390/molecules23123207
Akula, R., and Ravishankar, G. A. (2011). Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal. Behav. 6, 1720–1731. doi: 10.4161/psb.6.11.17613
Angulo-López, J. E., Flores-Gallegos, A. C., Torres-León, C., Ramírez-Guzmán, K. N., Martínez, G. A., and Aguilar, C. N. (2021). Guava (Psidium guajava L.) fruit and valorization of industrialization by-products. Processes. 9, 075. doi: 10.3390/pr9061075
Anwar, F., Abbas, A., and Alkharfy, K. M. (2016). “Cardamom (Elettaria cardamomum Maton) Oils,” in Essential oils in food preservation, flavor and safety, eds. V. R. Preedy (London: Academic Press) 295–301. doi: 10.1016/B978-0-12-416641-7.00033-X
Aoshima, H., Hirata, S., and Ayabe, S. (2007). Antioxidative and anti-hydrogen peroxide activities of various herbal teas. Food Chem. 103, 617–622. doi: 10.1016/j.foodchem.2006.08.032
Ashraf, M. A., Iqbal, M., Rasheed, R., Hussain, I., Riaz, M., and Arif, M. S. (2018). “Environmental stress and secondary metabolites in plants: an overview,” in Plant Metabolites and regulation Under Environmental Stress 153–167. doi: 10.1016/B978-0-12-812689-9.00008-X
Ašimović, Z., Sirbubalo, E., Cengić, L., Muminović, Š., and Jurković, J. (2022). “Phenolic Content and Antioxidant Activity Determination in Chamomile (Matricaria Recutita) and Sage (Salvia Officinalis) Teas,” in 10th Central European Congress on Food. CE-Food 2020 (Cham, Springer). doi: 10.1007/978-3-031-04797-8_31
Azhar, N., Hussain, B., Ashraf, M. Y., and Abbasi, K. Y. (2011). Water stress mediated changes in growth, physiology and secondary metabolites of desi ajwain (Trachyspermum ammi L.). Pak. J. Bot. 43, 15–19.
Bahadori, M. B., Zengin, G., Dinparast, L., and Eskandani, M. (2020). The health benefits of three Hedgenettle herbal teas (Stachys byzantina, Stachys inflata, and Stachys lavandulifolia)-profiling phenolic and antioxidant activities. Eur. J. Integr. Med. 36, 101134. doi: 10.1016/j.eujim.2020.101134
Balentine, D. A., Albano, M. C., and Nair, M. G. (1999). Role of medicinal plants, herbs, and spices in protecting human health. Nutr. Rev. 57, 41–45. doi: 10.1111/j.1753-4887.1999.tb01806.x
Bancirova, M. (2010). Comparison of the antioxidant capacity and the antimicrobial activity of black and green tea. Food Res. Int. 43, 1379–1382. doi: 10.1016/j.foodres.2010.04.020
Bandara, S. N. (2012). Agronomy of irrigated tea in low elevation growing areas of Sri Lanka. PhD thesis, Univ. Adelaide School Agriculture, Food Wine, Adelaide, Australia.
Bayliak, M. M., Dmytriv, T. R., Melnychuk, A. V., Strilets, N. V., Storey, K. B., and Lushchak, V. I. (2021). Chamomile as a potential remedy for obesity and metabolic syndrome. EXCLI. J. 20, 1261. doi: 10.17179/excli2021-4013
Bilanda, D. C., Dzeufiet, P. D. D., Kouakep, L., Aboubakar, B. F. O., Tedong, L., Kamtchouing, P., et al. (2017). Bidens pilosa ethylene acetate extract can protect against L-NAME-induced hypertension on rats. BMC Compl. Altern. Med. 17, 1–7. doi: 10.1186/s12906-017-1972-0
Bodalska, A., Kowalczyk, A., Włodarczyk, M., and Fecka, I. (2019). Analysis of polyphenolic composition of a herbal medicinal product—peppermint tincture. Molecules. 25, 69. doi: 10.3390/molecules25010069
Booker, A., and Heinrich, M. (2016). Value chains of botanical and herbal medicinal products: A European perspective. HerbalGram. 112, 40–45.
Boonyarikpunchai, W., Sukrong, S., and Towiwat, P. (2014). Antinociceptive and anti-inflammatory effects of rosmarinic acid isolated from Thunbergia laurifolia Lindl. Pharmacol. Biochem. Behav. 124, 67–73. doi: 10.1016/j.pbb.2014.05.004
Çakmakçi, S., Topdas, E. F., Kalin, P., Han, H., Sekerci, P., Köse, L., et al. (2015). Antioxidant capacity and functionality of oleaster (E laeagnus angustifolia L.) flour and crust in a new kind of fruity ice cream. Int. J. Food Sci. 50, 472–481. doi: 10.1111/ijfs.12637
Carabajal, M. P. A., Isla, M. I., and Zampini, I. C. (2017). Evaluation of antioxidant and antimutagenic activity of herbal teas from native plants used in traditional medicine in Argentina. S. Afr. J. Bot. 110, 258–265. doi: 10.1016/j.sajb.2016.10.006
Caruso, G., Formisano, L., Cozzolino, E., Pannico, A., El-Nakhel, C., Rouphael, Y., et al. (2020). Shading affects yield, elemental composition and antioxidants of perennial wall rocket crops grown from spring to summer in southern Italy. Plants. 9, 933. doi: 10.3390/plants9080933
Chandrasekara, A., and Shahidi, F. (2018). Herbal beverages: Bioactive compounds and their role in disease risk reduction-A review. J. Tradit. Complement. Med. 8, 451–458. doi: 10.1016/j.jtcme.2017.08.006
Chao, J., Dai, Y., Cheng, H. Y., Lam, W., Cheng, Y. C., Li, K., et al. (2017). Improving the concentrations of the active components in the herbal tea ingredient, Uraria crinita: The effect of post-harvest oven-drying processing. Sci. Rep. 7, 1–16. doi: 10.1038/srep38763
Chaudhary, S. K., Sandasi, M., Makolo, F., van Heerden, F. R., and Viljoen, A. M. (2021). Aspalathin: A rare dietary dihydrochalcone from Aspalathus linearis (rooibos tea). Phytochem Rev. 20, 1161–1192 doi: 10.1007/s11101-021-09741-9
Chawafambira, A. (2021). The effect of incorporating herbal (Lippia javanica) infusion on the phenolic, physicochemical, and sensorial properties of fruit wine. Food Sci. Nutr. 9, 4539–4549. doi: 10.1002/fsn3.2432
Chellan, P., Land, K. M., Shokar, A., Au, A., An, S. H., Clavel, C. M., et al. (2012). Exploring the versatility of cycloplatinated thiosemicarbazones as antitumor and antiparasitic agents. Organometallics. 31, 5791–5799. doi: 10.1021/om300334z
Cheynier, V., Comte, G., Davies, K. M., Lattanzio, V., and Martens, S. (2013). Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol. Biochem. 72, 1–20. doi: 10.1016/j.plaphy.2013.05.009
Chou, C. C., Lin, L. L., and Chung, K. T. (1999). Antimicrobial activity of tea as affected by the degree of fermentation and manufacturing season. Int. J. Food Microbiol. 48, 125–130. doi: 10.1016/S0168-1605(99)00034-3
Choudhury, N., Meghwal, M., and Das, K. (2021). Microencapsulation: An overview on concepts, methods, properties and applications in foods. Food Front. 2, 426–442. doi: 10.1002/fft2.94
Chung, I. M., Rekha, K., Rajakumar, G., and Thiruvengadam, M. (2018). Elicitation of silver nanoparticles enhanced the secondary metabolites and pharmacological activities in cell suspension cultures of bitter gourd. Biotech. 8, 1–12. doi: 10.1007/s13205-018-1439-0
Da-Costa-Rocha, I., bonnlaender, B., sievers, H, Pischel, I., and Heinrich, M. (2014). Hibiscus sabdariffa L.-A pharmacological and phytochemical. Food Chem. 165, 424–443 doi: 10.1016/j.foodchem.2014.05.002
Dandawate, P. R., Subramaniam, D., Padhye, S. B., and Anant, S. (2017). “Physiology, I,” in City K., City K., City K. and Campus A 913–945.
De Beer, D., du Preez, B. V. P., and Joubert, E. (2021). Development of HPLC method for quantification of phenolic compounds in Cyclopia intermedia (honeybush) herbal tea infusions. J. Food Compos. Anal.104, 104154. doi: 10.1016/j.jfca.2021.104154
De Wet, H., Nzama, V. N., and Van Vuuren, S. F. (2012). Medicinal plants used for the treatment of sexually transmitted infections by lay people in northern Maputaland, KwaZulu–Natal Province, South Africa. S. Afr. J. Bot. 78, 12–20. doi: 10.1016/j.sajb.2011.04.002
DeFronzo, R. A., Ferrannini, E., Groop, L., Henry, R. R., Herman, W. H., Holst, J. J., et al. (2015). Type 2 diabetes mellitus. Nat. Rev. Dis. Primers. 1, 15019. doi: 10.1038/nrdp.2015.19
den Braber, K., Sato, D., and Lee, E. (2011). “Farm and forestry production and marketing profile for tea (Camellia sinensis),” Specialty Crops for Pacific Island Agroforestry (Hawai: Permanent Agriculture Resources) 1–33.
Dube, P., Meyer, S., and Marnewick, J. L. (2017). Antimicrobial and antioxidant activities of different solvent extracts from fermented and green honeybush (Cyclopia intermedia) plant material. S. A. J. Bot. 110, 184–193. doi: 10.1016/j.sajb.2016.10.010
Dutta, A., Sen, J., and Deswal, R. (2007). Downregulation of terpenoid indole alkaloid biosynthetic pathway by low temperature and cloning of a AP2 type C-repeat binding factor (CBF) from Catharanthus roseus (L). G. Don. Plant Cell Rep. 26, 1869–1878. doi: 10.1007/s00299-007-0383-y
Eko, H., Hardhina, W., Triana, H., and Bambang, H. S. (2012). Effect of shading, nitrogen and magnesium fertilizer on phyllanthin and total flavonoid yield of Phyllanthus niruri in Indonesia soil. J. Med. Plant Res. 6, 4586–4592. doi: 10.5897/JMPR12.591
Endris, A., Asfaw, N., and Bisrat, D. (2016). Chemical composition, antimicrobial and antioxidant activities of the essential oil of Lippia javanica leaves from Ethiopia. J. Essent. Oil Res. 28, 221–226. doi: 10.1080/10412905.2015.1108880
Etheridge, C. J., and Derbyshire, E. (2020). Herbal infusions and health: A review of findings from human studies, mechanisms and future research directions. Nutr. Food Sci. 50, 969–985. doi: 10.1108/NFS-08-2019-0263
Farzaneh, V., and Carvalho, I. S. (2015). A review of the health benefit potentials of herbal plant infusions and their mechanism of actions. Ind. Crops Prod. 65, 247–258. doi: 10.1016/j.indcrop.2014.10.057
Fraga, C. G., Croft, K. D., Kennedy, D. O., and Tomás-Barberán, F. A. (2019). The effects of polyphenols and other bioactives on human health. Food Funct. 10, 514–528. doi: 10.1039/C8FO01997E
Gaggìa, F., Baffoni, L., Galiano, M., Nielsen, D. S., Jakobsen, R. R., Castro-Mejía, J. L., et al. (2018). Kombucha beverage from green, black and rooibos teas: a comparative study looking at microbiology, chemistry and antioxidant activity. Nutrients. 11, 1. doi: 10.3390/nu11010001
Gbenga-Fabusiwa, F. J., Oladele, E. P., Oboh, G., Adefegha, S. A., and Oshodi, A. A. (2018). Polyphenol contents and antioxidants activities of biscuits produced from ginger-enriched pigeon pea–wheat composite flour blends. J. Food Biochem. 42, e12526. doi: 10.1111/jfbc.12526
Ghasemzadeh, A., Jaafar, H. Z., Rahmat, A., Wahab, P. E. M., and Halim, M. R. A. (2010). Effect of different light intensities on total phenolics and flavonoids synthesis and antioxidant activities in young ginger varieties (Zingiber officinale Roscoe). Int. J. Mol. Sci.11, 3885–3897. doi: 10.3390/ijms11103885
Göçer, H., Akincioglu, A., Öztaşkin, N., Göksu, S., and Gülçin, I. (2013). Synthesis, Antioxidant, and Antiacetylcholinesterase Activities of Sulfonamide Derivatives of Dopamine-R elated Compounds. Arch. Pharm. 346, 783–792. doi: 10.1002/ardp.201300228
Gohil, K. J., Patel, J. A., and Gajjar, A. K. (2010). Pharmacological review on Centella asiatica: a potential herbal cure-all. Indian J. Pharm. Sci. 72, 546. doi: 10.4103/0250-474X.78519
Grbic, N., Paschko, K., Pinker, I., and Böhme, M. H. (2016). Effect of different light spectra by using coloured plastic films on growth, fresh and dry matter, nutrient solution uptake and secondary metabolites of Perilla frutescens (L.). Britt. Sci. Hortic. 210, 93–98. doi: 10.1016/j.scienta.2016.07.016
Grover, J. K., and Yadav, S. P. (2004). Pharmacological actions and potential uses of Momordica charantia: a review. J. Ethnopharmacol. 93, 123–132. doi: 10.1016/j.jep.2004.03.035
Guerrero-Beltrán, J. A., Barbosa-Cánovas, G. V., and Swanson, B. G. (2005). High hydrostatic pressure processing of fruit and vegetable products. Food Rev. Int. 21, 411–425. doi: 10.1080/87559120500224827
Gupta, V., Mittal, P., Bansal, P., Khokra, S. L., and Kaushik, D. (2010). Pharmacological potential of Matricaria recutita-A review. Int. J. Pharm Sci. Drug Res. 2, 12–16.
Guzelmeric, E., Ristivojević, P., Vovk, I., Milojković-Opsenica, D., and Yesilada, E. (2017). Quality assessment of marketed chamomile tea products by a validated HPTLC method combined with multivariate analysis. J. Pharm. Biomed. Anal. 132, 35–45. doi: 10.1016/j.jpba.2016.09.030
Hacioglu, M., Dosler, S., Tan, A. S. B., and Otuk, G. (2017). Antimicrobial activities of widely consumed herbal teas, alone or in combination with antibiotics: an in vitro study. Peer J. 5, e3467. doi: 10.7717/peerj.3467
Hamilton, J. G., Zangerl, A. R., DeLucia, E. H., and Berenbaum, M. R. (2001). The carbon–nutrient balance hypothesis: its rise and fall. Ecol. Lett. 4, 86–95. doi: 10.1046/j.1461-0248.2001.00192.x
Han, F., Ma, G. Q., Yang, M., Yan, L., Xiong, W., Shu, J. C., et al. (2017). Chemical composition and antioxidant activities of essential oils from different parts of the oregano. J. Zhejiang Univ. Sci. B. 18, 79–84. doi: 10.1631/jzus.B1600377
Hasanvand, E., Fathi, M., Bassiri, A., Javanmard, M., and Abbaszadeh, R. (2015). Novel starch based nanocarrier for vitamin D fortification of milk: Production and characterization. Food Bioprod. Process. 96, 264–277. doi: 10.1016/j.fbp.2015.09.007
Hayes, A. J., and Markovic, B. (2002). Toxicity of Australian essential oil Backhousia citriodora (Lemon myrtle). Part 1. Antimicrobial activity and in vitro cytotoxicity. Food Chem. Toxicol. 40, 535–543. doi: 10.1016/S0278-6915(01)00103-X
Hirasawa, M., Takada, K., Makimura, M., and Otake, S. (2002). Improvement of periodontal status by green tea catechin using a local delivery system: a clinical pilot study. J. Periodontal Res. 37, 433–438. doi: 10.1034/j.1600-0765.2002.01640.x
Hlahla, L. N. (2010). Effect of fermentation temperature and duration on chemical composition of Bush tea (Athrixia phylicoides DC.). PhD thesis, Univ. of Limpopo, Turfloop Campus, Mankweng, South Africa.
Hossain, M. F., Islam, M. T., Islam, M. A., and Akhtar, S. (2017). Cultivation and uses of stevia (Stevia rebaudiana Bertoni): A review. African J. Food, Agric. Nutr. Dev. 17, 12745–12757. doi: 10.18697/ajfand.80.16595
Hui Gan, S., Chai Tham, T., Xiang Ng, M., Suan Chua, L., Aziz, R., Redza Baba, M., et al. (2017). Study on retention of metabolites composition in misai kucing (Orthosiphon stamineus) by heat pump assisted solar drying. J. Food Process. Preserv. 41, e13262. doi: 10.1111/jfpp.13262
Huyen, T. T. (2020). Optimization of microwave-assisted extraction of phenolic compounds from ginger (Rosc.) using response surface methodology. Herba Pol. 66, 19–27. doi: 10.2478/hepo-2020-0007
Ibrahim, M. H., Jaafar, H. Z., Karimi, E., and Ghasemzadeh, A. (2013). Impact of organic and inorganic fertilizers application on the phytochemical and antioxidant activity of Kacip Fatimah (Labisia pumila Benth). Molecules. 18, 10973–10988. doi: 10.3390/molecules180910973
Ibrahim, M. H., Jaafar, H. Z., Rahmat, A., and Rahman, Z. A. (2011). Effects of nitrogen fertilization on synthesis of primary and secondary metabolites in three varieties of Kacip Fatimah (Labisia pumila Blume). Int. J. Mol. Sci. 12, 5238–5254. doi: 10.3390/ijms12085238
Idrees, M., Sania, B., Hafsa, B., Kumari, S., Khan, H., Fazal, H., et al. (2018). Spectral lights trigger biomass accumulation and production of antioxidant secondary metabolites in adventitious root cultures of Stevia rebaudiana (Bert.). C. R. Biol. 341, 334–342. doi: 10.1016/j.crvi.2018.05.003
Ignasimuthu, K., Prakash, R., Murthy, P. S., and Subban, N. (2019). Enhanced bioaccessibility of green tea polyphenols and lipophilic activity of EGCG octaacetate on gram-negative bacteria. Lwt. 105, 103–109. doi: 10.1016/j.lwt.2019.01.064
Jan, R., Asaf, S., Numan, M., and Kim, K. M. (2021). Plant secondary metabolite biosynthesis and transcriptional regulation in response to biotic and abiotic stress conditions. Agronomy. 11, 968. doi: 10.3390/agronomy11050968
Jin, B., Liu, Y., Xie, J., Luo, B., and Long, C. (2018). Ethnobotanical survey of plant species for herbal tea in a Yao autonomous county (Jianghua, China): results of a 2-year study of traditional medicinal markets on the Dragon Boat Festival. J. Ethnobiol. Ethnomedicine. 14, 58. doi: 10.1186/s13002-018-0257-0
Jin, L., Li, X. B., Tian, D. Q., Fang, X. P., Yu, Y. M., Zhu, H. Q., et al. (2016). Antioxidant properties and color parameters of herbal teas in China. Ind. Crops Prod. 87, 198–209. doi: 10.1016/j.indcrop.2016.04.044
Joseph, B., and Jini, D. (2013). Antidiabetic effects of Momordica charantia (bitter melon) and its medicinal potency. Asian Pac. J. Trop. Dis. 3, 93–102. doi: 10.1016/S2222-1808(13)60052-3
Joubert, E., and Schultz, H. (2012). Production and quality aspects of rooibos tea and related products. A review. J. Appl. Bot. Food Qual. 80, 138–144.
Jurendić, T., and Ščetar, M. (2021). Aronia melanocarpa products and by-products for health and nutrition: A Review. Antioxidants. 10, 1052. doi: 10.3390/antiox10071052
Kamaruddin, Z. H., Jumaidin, R., Selamat, M. Z., and Ilyas, R. A. (2021). Characteristics and properties of lemongrass (Cymbopogan Citratus): A comprehensive review. J. Nat. Fibers. 1–18. doi: 10.1080/15440478.2021.1958439
Karimi, E., Jaafar, H. Z., Ghasemzadeh, A., and Ibrahim, M. H. (2013). Light intensity effects on production and antioxidant activity of flavonoids and phenolic compounds in leaves, stems and roots of three varieties of Labisia pumila Benth. Aust. J. Crop Sci. 7, 1016.
Kashchenko, N. I., Olennikov, D. N., and Chirikova, N. K. (2021). Metabolites of Siberian raspberries: LC-MS profile, seasonal variation, antioxidant activity and, thermal stability of Rubus matsumuranus phenolome. Plants. 10, 2317. doi: 10.3390/plants10112317
Khandaker, L., Akond, A. M., Ali, M. B., and Oba, S. (2010). Biomass yield and accumulations of bioactive compounds in red amaranth (Amaranthus tricolor L.) grown under different colored shade polyethylene in spring season. Sci. Hortic. 123, 289–294. doi: 10.1016/j.scienta.2009.09.012
Kieling, D. D., and Prudencio, S. H. (2019). Antioxidants properties, physicochemical, and sensory quality attributes of a new soy-based beverage with lemongrass and lime juice. J. Culin. Sci. Technol. 17, 103–117. doi: 10.1080/15428052.2017.1405859
Kim, P. K., Jung, K. I., Choi, Y. J., and Gal, S. W. (2017). Anti-inflammatory effects of lemon myrtle (Backhousia citriodora) leaf extracts in LPS-induced RAW 264.7 cells. J. Life Sci. 27, 986–993. doi: 10.5352/JLS.2017.27.9.986
Koch, W., Kukuła-Koch, W., Czop, M., Helon, P., and Gumbarewicz, E. (2020). The role of extracting solvents in the recovery of polyphenols from green tea and its antiradical activity supported by principal component analysis. Molecules. 25, 2173. doi: 10.3390/molecules25092173
Krushna, N. S., Shiny, C., Dharanya, S., Sindhu, A., Aishwarya, S., and Narayanan, R. B. (2009). Immunolocalization and serum antibody responses to Brugia malayi pepsin inhibitor homolog (Bm-33). Microbiol. Immunol. 53, 173–183. doi: 10.1111/j.1348-0421.2009.00114.x
Kumar, R., Sharma, S., and Pathania, V. (2013a). Effect of shading and plant density on growth, yield and oil composition of clary sage (Salvia sclarea L.) in North Western Himalaya. J. Essent. Oil Res. 25, 23–32. doi: 10.1080/10412905.2012.742467
Kumar, R., Sharma, S., Ramesh, K., Pathania, V., and Prasad, R. (2014). Irradiance stress and plant spacing effect on growth, biomass and quality of wild marigold (Tagetes minuta L.)–an industrial crop in western Himalaya. J. Essent. Oil Res. 26, 348–358. doi: 10.1080/10412905.2014.935497
Kumar, R., Sharma, S., Ramesh, K., and Singh, B. (2013b). Effects of shade regimes and planting geometry on growth, yield and quality of the natural sweetener plant stevia (Stevia rebaudiana Bertoni) in North-Western Himalaya. Arch. Agron. Soil Sci. 59, 963–979. doi: 10.1080/03650340.2012.699676
Lehlohonolo, N., Mariga, I. K., Ngezimana, W., and Mudau, F. N. (2013). Bush tea (Athrixia phylicoides dc.) Success stories in South Africa A review. Crop Prod. 2, 37–43.
Liang, Y. C., Yang, M. T., Lin, C. J., Chang, C. L. T., and Yang, W. C. (2016). Bidens pilosa and its active compound inhibit adipogenesis and lipid accumulation via down-modulation of the C/EBP and PPARγ pathways. Sci. Rep. 6, 24285. doi: 10.1038/srep24285
Lin, Y. L., Juan, I. M., Chen, Y. L., Liang, Y. C., and Lin, J. K. (1996). Composition of polyphenols in fresh tea leaves and associations of their oxygen-radical-absorbing capacity with antiproliferative actions in fibroblast cells. J. Agric. Food Chem. 44, 1387–1394. doi: 10.1021/jf950652k
Lin-Wang, K. U. I., Micheletti, D., Palmer, J., Volz, R., Lozano, L., Espley, R., et al. (2011). High temperature reduces apple fruit colour via modulation of the anthocyanin regulatory complex. Plant Cell Environ. 34, 1176–1190. doi: 10.1111/j.1365-3040.2011.02316.x
Lv, Z., Zhang, C., Shao, C., Liu, B., Liu, E., Yuan, D., et al. (2021). Research progress on the response of tea catechins to drought stress. J. Sci. Food Agric. 101, 5305–5313. doi: 10.1002/jsfa.11330
Ma, C. H., Chu, J. Z., Shi, X. F., Liu, C. Q., and Yao, X. Q. (2016). Effects of enhanced UV-B radiation on the nutritional and active ingredient contents during the floral development of medicinal chrysanthemum. J. Photochem. Photobiol. B, Biol.158, 228–234. doi: 10.1016/j.jphotobiol.2016.02.019
MacAlister, D., Muasya, A. M., Crespo, O., Ogola, J. B., Maseko, S. T., Valentine, A. J., et al. (2020). Effect of temperature on plant growth and stress tolerant traits in rooibos in the Western Cape, South Africa. Sci. Hortic. 263, 109137. doi: 10.1016/j.scienta.2019.109137
Majewska, E., Kozlowska, M., Gruszczynska-Sekowska, E., Kowalska, D., and Tarnowska, K. (2019). Lemongrass (Cymbopogon citratus) essential oil: extraction, composition, bioactivity and uses for food preservation-a review. Polish J. Food Nutr. Sci. 69, 327–341. doi: 10.31883/pjfns/113152
Makola, M. M., Dubery, I. A., Koorsen, G., Steenkamp, P. A., Kabanda, M. M., du Preez, L. L., et al. (2016b). The effect of geometrical isomerism of 3, 5-dicaffeoylquinic acid on its binding affinity to HIV-integrase enzyme: A molecular docking study. Evid. Based Complement. Alternat. Med. 2016, 4138263. doi: 10.1155/2016/4138263
Makola, M. M., Steenkamp, P. A., Dubery, I. A., Kabanda, M. M., and Madala, N. E. (2016a). Preferential alkali metal adduct formation by cis geometrical isomers of dicaffeoylquinic acids allows for efficient discrimination from their trans isomers during ultra-high-performance liquid chromatography/quadrupole time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 30, 1011–1018. doi: 10.1002/rcm.7526
Malongane, F., Mc,GA., W L. J, and Mudau, F. N. (2017). The synergistic potential of various teas, herbs and therapeutic drugs in health improvement: a review. J. Sci. Food Agric. 97, 4679–4689. doi: 10.1002/jsfa.8472
Malongane, F., McGaw, L. J., Debusho, L. K., and Mudau, F. N. (2020). Sensory characteristics and volatile compounds of herbal teas and mixtures of bush tea with other selected herbal teas of South Africa. Foods. 9, 496. doi: 10.3390/foods9040496
Mamphiswana, N. D., Mashela, P. W., and Mdee, L. K. (2010). Distribution of total phenolics and antioxidant activity in fruit, leaf, stem and root of Monsonia burkeana. Afr. J. Agric. Res. 5, 2570–2575.
Marnewick, J. L., Gelderblom, W. C., and Joubert, E. (2000). An investigation on the antimutagenic properties of South African herbal teas. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 471, 157–166. doi: 10.1016/S1383-5718(00)00128-5
Marszałek, K., Wozniak, Ł., Skapska, S., and Mitek, M. (2017). High pressure processing and thermal pasteurization of strawberry purée: quality parameters and shelf-life evaluation during cold storage. J. Food Sci. Technol. 54, 832–841. doi: 10.1007/s13197-017-2529-4
Martínez-Solís, J., Calzada, F., Barbosa, E., and Valdés, M. (2021). Antihyperglycemic and antilipidemic properties of a tea infusion of the leaves from Annona cherimola miller on streptozocin-induced type 2 diabetic mice. Molecules. 26, 2408. doi: 10.3390/molecules26092408
Mashimbye, M. J., Mudaua, F. N., Soundy, P., and Van Ree, T. (2006). A new flavonol from Athrixia phylicoides (bush tea). S.A. J. Chem. 59, 1–2.
Masike, K., Tugizimana, F., Ndlovu, N., Smit, E., du Preez, L., Dubery, I., et al. (2017). Deciphering the influence of column chemistry and mass spectrometry settings for the analyses of geometrical isomers of L-chicoric acid. J. Chromatogr. B Biomed. Appl. 1052, 73–81. doi: 10.1016/j.jchromb.2017.03.023
Mathivha, P. L., Msagati, T. A., Thibane, V. S., and Mudau, F. N. (2020). “Phytochemical analysis of herbal teas and their potential health, and food safety benefits: a review,” in Herbal Medicine in India, eds. S., Sen, R., Chakraborty (Singapore: Springer) 281–301. doi: 10.1007/978-981-13-7248-3_20
Mavundza, E. J., Tshikalange, T. E., Mudau, F. N., and Meyer, J. J. (2007). Evaluation of antibacterial and antioxidant activities in ethanol extract of wild bush tea (Athrixia phylicoides (DC.). Med. Aromat. Plant Sci. Biotechnol. 1, 240–242
Mbondo, N. N., Owino, W. O., Ambuko, J., and Sila, D. N. (2018). Effect of drying methods on the retention of bioactive compounds in African eggplant. Food Sci. Nutr. 6, 814–823. doi: 10.1002/fsn3.623
McGraw, L. J., Steenkamp, V., and Eloff, J. N. (2007). Evaluation of Athrixia bush tea for cytotoxicity, antioxidant activity, caffeine content and presence of pyrrolizidine alkaloids. J. Ethnopharmacol. 110, 16–22. doi: 10.1016/j.jep.2006.08.029
McKay, D. L., and Blumberg, J. B. (2007). A review of the bioactivity of South African herbal teas: rooibos (Aspalathus linearis) and honeybush (Cyclopia intermedia). Phytother. Res. 21, 1–16. doi: 10.1002/ptr.1992
Medina-Meza, I. G., Barnaba, C., Villani, F., and Barbosa-Cánovas, G. V. (2015). Effects of thermal and high-pressure treatments in color and chemical attributes of an oil-based spinach sauce. LWT-Food Sci. Technol. 60, 86–94. doi: 10.1016/j.lwt.2014.09.033
Mfengu, M. O., Shauli, M., Engwa, G. A., Musarurwa, H. T., and Sewani-Rusike, C. R. (2021). Lippia javanica (Zumbani) herbal tea infusion attenuates allergic airway inflammation via inhibition of Th2 cell activation and suppression of oxidative stress. BMC Complement Altern. Med. 21, 192. doi: 10.1186/s12906-021-03361-8
Mkwanazi, M. V., Ndlela, S. Z., and Chimonyo, M. (2021). Indigenous knowledge to mitigate the challenges of ticks in goats: A systematic review. Vet. Anim. Sci. 13, 100190. doi: 10.1016/j.vas.2021.100190
Mohale, K. C., Hintsa, A. T., Emanuel, M. A., and Mudau, F. N. (2018). Metabolic profiling of cultivated bush tea (Athrixia phylicoides DC.) in response to different pruning types. HortSci. 53, 993–998. doi: 10.21273/HORTSCI13023-18
Mohd Yusof, F. F., Yaacob, J. S., Osman, N., Ibrahim, M. H., Wan-Mohtar, W. A. A. Q.I., Berahim, Z., et al. (2021). Shading effects on leaf gas exchange, leaf pigments and secondary metabolites of Polygonum minus Huds., an aromatic medicinal herb. Plants. 10, 608. doi: 10.3390/plants10030608
Mokoka, N. N. (2007). Indigenous knowledge of fever tea (Lippia javanica) and effect of shade netting on plant growth, oil yield and compound composition. PhD thesis, Univ. of Pretoria, Pretoria, South Africa.
Monika, M., and Selvakumar, S. (2019). Recent developments in direct C–H functionalization of quinoxalin-2 (1H)-ones via radical addition processes. Synthesis. 1, 4113–4136. doi: 10.1055/s-0037-1611910
Moreira, F. V., Bastos, J. F., Blank, A. F., Alves, P. B., and Santos, M. R. (2010). Chemical composition and cardiovascular effects induced by the essential oil of Cymbopogon citratus DC. Stapf, Poaceae, in rats. Rev. Bras. Farmacogn. 20, 904–909. doi: 10.1590/S0102-695X2010005000012
Morita, A., and Tuji, M. (2002). Nitrate and oxalate contents of tea plants (Camellia sinensis L.) with special reference to types of green tea and effect of shading. J. Soil Sci. Plant Nutr. 48, 547–553. doi: 10.1080/00380768.2002.10409237
Mphangwe, N. I. K. (2012). “Lung pruning: A review of practice,” in Tea Research Foundation of Central Africa (TRFCA) News. 18–23.
Mubvuma, M. T. (2018). Planting date as an adaptive strategy to improve yield of Chickpea (Cicer arietinum) under under climate change condition in Southern Africa. PhD thesis, Department of Plant Production, School of Agriculture, University of Venda, Thohoyandou, South Africa.
Mudau, F. N., and Makunga, N. P. (2018). Effect of seasonal variations and growth conditions on carbohydrate partitioning in different organs and the quality of bush tea. HortSci. 53, 999–1005. doi: 10.21273/HORTSCI12972-18
Mudau, F. N., Mudau, A. R., Nkomo, M., and Ngezimana, W. (2016). Variation in carbohydrate reserves and dry matter production of bush tea (Athrixia phylicoides) grown under different environmental conditions. HortSci. 51, 1537–1541. doi: 10.21273/HORTSCI11197-16
Mudau, F. N., Ngele, A., Mashela, P. W., and Soundy, P. (2007a). Seasonal variation of tannin content in wild bush tea. Med. Aromat. Plant Sci. Biotechnol. 1, 74–76.
Mudau, F. N., Soundy, P., and Du Toit, E. S. (2007b). Effects of nitrogen, phosphorus, and potassium nutrition on total polyphenol content of bush tea (Athrixia phylicoides L.) leaves in shaded nursery environment. HortSci. 42, 334–338. doi: 10.21273/HORTSCI.42.2.334
Mudau, F. N., Soundy, P., Du Toit, E. S., and Olivier, J. (2006). Variation in polyphenolic content of Athrixia phylicoides (L.) (bush tea) leaves with season and nitrogen application. S. A. J. Bot. 72, 398–402. doi: 10.1016/j.sajb.2005.10.007
Muronga, M., Quispe, C., Tshikhudo, P. P., Msagati, T. A. M., Mudau, F. N., Martorell, M., et al. (2021). Three selected edible crops of the genus momordica as potential sources of phytochemicals: biochemical, nutritional, and medicinal values. Front. Pharmacol. 12, 625546. doi: 10.3389/fphar.2021.625546
Naghiloo, S., Movafeghi, A., Delazar, A., Nazemiyeh, H., Asnaashari, S., and Dadpour, M. R. (2012). Ontogenetic variation of volatiles and antioxidant activity in leaves of Astragalus compactus Lam. (Fabaceae). EXCLI. J. 11, 436.
Namdeo, A. G. (2018). “Cultivation of medicinal and aromatic plants,” in Natural Products and Drug Discovery, eds. S. C. Mandal, V. Mandal, T. Konishi (London: Elsevier) 525–553. doi: 10.1016/B978-0-08-102081-4.00020-4
Nasma, A., Aishath, N., Azilah, A., and Sulaiman, A. Z. (2018). Optimization of vitexin and isovitexin compounds extracted from dried Mas Cotek leaves using one-factor-at-a-time (OFAT) approach in aqueous extraction. Int. Food Res. J. 25, 2562–2573.
Nazmi, N. N. M., and Sarbon, N. M. (2020). Characterization on antioxidant and physical properties of gelatin based composite films with incorporation of Centella asiatica (pegaga) extract. Food Res. 4, 224–233. doi: 10.26656/fr.2017.4(1).243
Nchabeleng, L., Mudau, F. N., and Mariga, I. K. (2012). Effects of chemical composition of wild bush tea (Athrixia phylicoides DC.) growing at locations differing in altitude, climate and edaphic factors. J. Med. Plants Res. 6, 1662–1666. doi: 10.5897/JMPR11.1453
Ngoepe, N. M., Mbita, Z., Mathipa, M., Mketo, N., Ntsendwana, B., and Hintsho-Mbita, N. C. (2018). Biogenic synthesis of ZnO nanoparticles using Monsonia burkeana for use in photocatalytic, antibacterial and anticancer applications. Ceram. Int. 44, 16999–17006. doi: 10.1016/j.ceramint.2018.06.142
Ni, Y. W., Lin, K. H., Chen, K. H., Wu, C. W., and Chang, Y. S. (2020). Flavonoid compounds and photosynthesis in Passiflora plant leaves under varying light intensities. Plants. 9, 633. doi: 10.3390/plants9050633
Nibir, Y. M., Sumit, A. F., Akhand, A. A., Ahsan, N., and Hossain, M. S. (2017). Comparative assessment of total polyphenols, antioxidant and antimicrobial activity of different tea varieties of Bangladesh. Asian Pac. J. Trop. Biomed. 7, 352–357. doi: 10.1016/j.apjtb.2017.01.005
Nikniaz, Z., Mahdavi, R., Ghaemmaghami, S. J., Yagin, N. L., and Nikniaz, L. (2016). Effect of different brewing times on antioxidant activity and polyphenol content of loosely packed and bagged black teas (Camellia sinensis L.). Avicenna J. Phytomedicine. 6, 313–310.
Nnzeru, R. L., Ntushelo, K., and Mudau, F. N. (2016). Physical appraisal and attributes of Monsonia burkeana (special tea): the perspective of tea users. Indilinga African J. Indig. Knowl. Syst. 15, 111–122.
Nobela, O., Renslow, R. S., Thomas, D. G., Colby, S. M., Sitha, S., Njobeh, P. B., et al. (2018). Efficient discrimination of natural stereoisomers of chicoric acid, an HIV-1 integrase inhibitor. J. Photochem. Photobiol. B, Biol. 189, 258–266. doi: 10.1016/j.jphotobiol.2018.10.025
Oladeji, O. S., Adelowo, F. E., Ayodele, D. T., and Odelade, K. A. (2019). Phytochemistry and pharmacological activities of Cymbopogon citratus: A review. Sci. Afr. 6, e00137. doi: 10.1016/j.sciaf.2019.e00137
Olayemi, R. F. (2017). Comparative study of root, stalk and leaf essential oils of Cymbopogon Citratus (lemon grass). Chem. Search. J. 8, 20–28. doi: 10.4314/csj.v8i1.3
Omogbai, B. A., and Ikenebomeh, M. (2013). Microbiological characteristics and phytochemical screening of some herbal teas in Nigeria. Eur. Sci. J. 9, 149.
Owour, P. O., and Obanda, M. (1998). Clonal selection criteria for quality in Kenya black tea production: achievements, problems and perspectives: review. Tea. 19, 49–58.
Owuor, P. O., Ng'etich, W. K., and Obanda, M. (2000). Quality response of clonal black tea to nitrogen fertiliser, plucking interval and plucking standard. J. Sci. Food Agric. 80, 439–446. doi: 10.1002/(SICI)1097-0010(200003)80:4<439:AID-JSFA546>3.0.CO;2-2
Owuor, P. O., Tsushida, T., Horita, H., and Murai, T. (1988). Effects of geographical area of production on the composition of the volatile flavour compounds in Kenyan clonal black CTC teas. Exper. Agric. 24, 227–235. doi: 10.1017/S0014479700015970
Pang, S. F., Lau, M. Z., Yusoff, M. M., and Gimbun, J. (2017). Microwave-irradiation induced fast simultaneous extraction of methoxylated and hydroxylated phenolic compounds from Orthosiphon stamineus leaves. Mater. Sci. Forum. 890, 155–158. doi: 10.4028/www.scientific.net/MSF.890.155
Pasrija, D., Ezhilarasi, P. N., Indrani, D., and Anandharamakrishnan, C. (2015). Microencapsulation of green tea polyphenols and its effect on incorporated bread quality. LWT-Food Sci. Technol. 6, 289–296. doi: 10.1016/j.lwt.2015.05.054
Patras, A., Brunton, N. P., Gormely, T. R., and Butler, F. (2009). Impact of high-pressure processing on antioxidant activity, ascorbic acid, anthocyanins and instrumental colour of blackberry and strawberry puree. Innov. Food Sci. Emerg. Technol. 10, 308–313. doi: 10.1016/j.ifset.2008.12.004
Pezzani, R., Vitalini, S., and Iriti, M. (2017). Bioactivities of Origanum vulgare L.: An update. Phytochem Rev. 1253–1268. doi: 10.1007/s11101-017-9535-z
Phelan, J., and Rees, J. (2003). The erosive potential of some herbal teas. J. Dent. 31, 241–246. doi: 10.1016/S0300-5712(03)00048-4
Pola, W., Sugaya, S., and Photchanachai, S. (2020). Influence of postharvest temperatures on carotenoid biosynthesis and phytochemicals in mature green chili (Capsicum annuum L.). Antioxidants. 9, 203. doi: 10.3390/antiox9030203
Poswal, F. S., Russell, G., Mackonochie, M., MacLennan, E., Adukwu, E. C., and Rolfe, V. (2019). Herbal teas and their health benefits: a scoping review. Plant Foods Hum. Nutr. 74, 266–276. doi: 10.1007/s11130-019-00750-w
Prajapati, S. K., Jain, D., Parveen, S., Maji, S., and Deb, P. K. (2022). Nanodelivery of Antioxidant Herbal Extracts, Spices, and Dietary Constituents. Phytoantioxidants Nanotherap. 8, 145–171. doi: 10.1002/9781119811794.ch8
Proutsos, N. D., Liakatas, A., Alexandris, S. G., Tsiros, I. X., Tigkas, D., and Halivopoulos, G. (2022). Atmospheric factors affecting global solar and photosynthetically active radiation relationship in a mediterranean forest site. Atmosphere. 13, 1207. doi: 10.3390/atmos13081207
Radušiene, J., Marksa, M., Ivanauskas, L., Jakštas, V., Çalişkan, Ö., Kurt, D., et al. (2019). Effect of nitrogen on herb production, secondary metabolites and antioxidant activities of Hypericum pruinatum under nitrogen application. Ind. Crops Prod. 139, 111519. doi: 10.1016/j.indcrop.2019.111519
Ramabulana, A. T., Petras, D., Madala, N. E., and Tugizimana, F. (2021). Metabolomics and molecular networking to characterize the chemical space of four momordica plant species. Metabolites. 11, 763. doi: 10.3390/metabo11110763
Ramabulana, A. T., Steenkamp, P., Madala, N., and Dubery, I. A. (2020). Profiling of chlorogenic acids from Bidens pilosa and differentiation of closely related positional isomers with the aid of UHPLC-QTOF-MS/MS-based in-source collision-induced dissociation. Metabolites. 10, 178. doi: 10.3390/metabo10050178
Ramphinwa, M. L., Mchau, G. R., Madala, N. E., Nengovhela, N., Ogola, J. B., and Mudau, F. N. (2022). Response of Plant Growth and Development, and Accumulation of Hydroxyl-cinnamoyl Acid Derivatives to Selected Shade Nets and Seasonality of Field-grown Bush Tea (Athrixia phylicoides DC.). Hort Sci. 57, 87–96. doi: 10.21273/HORTSCI16171-21
Rasool Hassan, B. A. (2012). Medicinal plants (importance and uses). Pharmaceut Anal Acta. 3, 2153–2435. doi: 10.4172/2153-2435.1000e139
Ravichandran, R., and Parthiban, R. (1998). Changes in enzyme activities (polyphenol oxidase and phenylalanine ammonialyase) with type of tea leaf and during black tea manufacture and the effect of enzyme supplementation of dhool on black tea quality. Food Chem. 62, 277–281. doi: 10.1016/S0308-8146(97)00220-3
Rezai, S., Etemadi, N., Nikbakht, A., Yousefi, M., and Majidi, M. M. (2018). Effect of light intensity on leaf morphology, photosynthetic capacity, and chlorophyll content in sage (Salvia officinalis L.). Korean J. Hortic. Sci. Technol. 36, 46–57 doi: 10.12972/kjhst.20180006
Rihan, H. Z., Aldarkazali, M., Mohamed, S. J., McMulkin, N. B., Jbara, M. H., and Fuller, M. P. (2020). A novel new light recipe significantly increases the growth and yield of sweet basil (Ocimum basilicum) grown in a plant factory system. Agronomy. 10, 934. doi: 10.3390/agronomy10070934
Sahin, F., Güllüce, M., Daferera, D., Sökmen, A., Sökmen, M., Polissiou, M., et al. (2004). Biological activities of the essential oils and methanol extract of Origanum vulgare ssp. vulgare in the Eastern Anatolia region of Turkey. Food Control. 15, 549–557. doi: 10.1016/j.foodcont.2003.08.009
Said-Al Ahl, H. A. H., and Hussein, M. S. (2010). Effect of water stress and potassium humate on the productivity of oregano plant using saline and freshwater irrigation. Ozean. J. Appl. Sci. 3, 125–141.
Salman, S., Öz, G., Felek, R., Haznedar, A., Turna, T., and Özdemir, F. (2022). Effects of fermentation time on phenolic composition, antioxidant and antimicrobial activities of green, oolong, and black teas. Food Biosci. 49, 101884. doi: 10.1016/j.fbio.2022.101884
Sehgal, A. (2022). An update on disease preventing potential of green tea in comparison with some tisanes. S. Afr. J. Bot. 144, 92–96. doi: 10.1016/j.sajb.2021.08.027
Sengupta, A., Nielsen, K. E., Barinshteyn, G., Li, K., and 3M Innovative Properties Co. (2001). Encapsulation process and encapsulated products. U.S. Patent. 6, 248–364.
Shao, P., Zhang, H., Niu, B., and Jin, W. (2018). Physical stabilities of taro starch nanoparticles stabilized Pickering emulsions and the potential application of encapsulated tea polyphenols. Int. J. Biol. Macromol. 118, 2032–2039. doi: 10.1016/j.ijbiomac.2018.07.076
Sharpe, E., Hua, F., Schuckers, S., Andreescu, S., and Bradley, R. (2016). Effects of brewing conditions on the antioxidant capacity of twenty-four commercial green tea varieties. Food chem. 192, 380–387. doi: 10.1016/j.foodchem.2015.07.005
Singh, R., De, S., and Belkheir, A. (2013). Avena sativa (Oat), a potential neutraceutical and therapeutic agent: an overview. Crit. Rev. Food Sci. Nutr. 53, 126–144. doi: 10.1080/10408398.2010.526725
Singh, R., Shushni, M. A., and Belkheir, A. (2015). Antibacterial and antioxidant activities of Mentha piperita L. Arabian J. Chem. 8, 322–328. doi: 10.1016/j.arabjc.2011.01.019
Singh, S., Virmani, T., and Kohli, K. (2020). Phytochemicals and medicinal uses of red raspberry: a review. J. Pharmaceut Res. 5, 48–52. doi: 10.33140/JPR.05.02.03
Smirnoff, N. (1993). Tansley Review No. 52. The role of active oxygen in the response of plants to water deficit and desiccation. New Phytol. 52, 27–58.
Soni, U., Brar, S., and Gauttam, V. K. (2015). Effect of seasonal variation on secondary metabolites of medicinal plants. Int. J. Pharm. Sci. Res. 6, 3654–3662. doi: 10.13040/IJPSR.0975-8232.6(9).3654-62
Strzemski, M., Dzida, K., Dresler, S., Sowa, I., Kurzepa, J., Szymczak, G., et al. (2021). Nitrogen fertilisation decreases the yield of bioactive compounds in Carlina acaulis L. grown in the field. Ind. Crops. Prod. 170, 113698. doi: 10.1016/j.indcrop.2021.113698
Suleman, Z., Engwa, G. A., Shauli, M., Musarurwa, H. T., Katuruza, N. A., and Sewani-Rusike, C. R. (2022). Neuroprotective effects of Lippia javanica (Burm. F.) Spreng. Herbal tea infusion on Lead-induced oxidative brain damage in Wistar rats. BMC Complement. Altern. Med. 22, 4. doi: 10.1186/s12906-021-03471-3
Sun, T., Zhang, J., Zhang, Q., Li, X., Li, M., Yang, Y., et al. (2021). Integrative physiological, transcriptome, and metabolome analysis reveals the effects of nitrogen sufficiency and deficiency conditions in apple leaves and roots. Environ. Exp. Bot. 192, 104633. doi: 10.1016/j.envexpbot.2021.104633
Thakur, M., Bhatt, V., and Kumar, R. (2019). Effect of shade level and mulch type on growth, yield and essential oil composition of damask rose (Rosa damascena Mill.) under mid hill conditions of Western Himalayas. PLoS ONE. 14, e0214672. doi: 10.1371/journal.pone.0214672
Tshivhandekano, I., Ngezimana, W., Tshikalange, T. E., Makunga, N. P., and Mudau, F. N. (2018). Nitrogen application influences quality, pharmacological activities and metabolite profiles of Athrixia phylicoides DC. (Bush tea) cultivated under greenhouse and field conditions. Acta Agric. Scand. - B Soil Plant Sci. 68, 388–400. doi: 10.1080/09064710.2017.1412496
Tshivhandekano, T., Mudau, F. N., Soundy, P., and Ngezimana, W. N. (2013). Effect of cultural practices and environmental conditions on yield and quality of herbal plants: Prospects leading to research on influence of nitrogen fertilization, planting density and eco-physiological parameters on yield and quality of field-grown bush tea (Athrixia phylicoides DC.). J. Med. Plants Res. 7, 2489–2493
Turkmen, N., Ferda, S. A. R. I., and Velioglu, Y. S. (2009). Factors affecting polyphenol content and composition of fresh and processed tea leaves. Akademik Gida. 7, 29–40
Upadhya, C. (2004). A new transnational capitalist class? Capital flows, business networks and entrepreneurs in the Indian software industry. Econ. Political Wkly. 39, 5141–5151.
Verma, N., and Shukla, S. (2015). Impact of various factors responsible for fluctuation in plant secondary metabolites. J. Appl. Res. Med. Aromat. Plants. 2, 105–113. doi: 10.1016/j.jarmap.2015.09.002
Villa-Rodriguez, J. A., Kerimi, A., Abranko, L., Tumova, S., Ford, L., Blackburn, R. S., et al. (2018). Acute metabolic actions of the major polyphenols in chamomile: An in vitro mechanistic study on their potential to attenuate postprandial hyperglycaemia. Sci. Rep. 8, 1–14. doi: 10.1038/s41598-018-23736-1
Vural, N., Algan Cavuldak, Ö., Akay, M. A., and Anl,i, R. E. (2020). Determination of the various extraction solvent effects on polyphenolic profile and antioxidant activities of selected tea samples by chemometric approach. J. Food Meas. Charact. 14, 1286–1305. doi: 10.1007/s11694-020-00376-6
Wahid, A., Gelani, S., Ashraf, M., and Foolad, M. R. (2007). Heat tolerance in plants: an overview. Environ. Exp. Bot. 61, 199–223. doi: 10.1016/j.envexpbot.2007.05.011
Waterman, P. G., and Mole, S. (2019). “Extrinsic factors influencing production of secondary metabolites in plants,” in Insect-Plant Interactions 107–134. CRC press. doi: 10.1201/9780429290916-4
World Health Organization (2013). WHO traditional medicine strategy 2014–2023. WHO. Available online at: https://www.who.int/medicines/publications/traditional/trm_strategy14_23/en/ (accessed June 11, 2022).
Wright, S. C., Maree, J. E., and Sibanyoni, M. (2009). Treatment of oral thrush in HIV/AIDS patients with lemon juice and lemon grass (Cymbopogon citratus) and gentian violet. Phytomedicine. 16, 118–124. doi: 10.1016/j.phymed.2008.07.015
Xia, X., Lin, Z., Shao, K., Wang, X., Xu, J., Zhai, H., et al. (2021). Combination of white tea and peppermint demonstrated synergistic antibacterial and anti-inflammatory activities. J. Sci. Food Agr. 106, 2500–2510. doi: 10.1002/jsfa.10876
Yang, L., Wen, K. S., Ruan, X., Zhao, Y. X., Wei, F., and Wang, Q. (2018). Response of plant secondary metabolites to environmental factors. Molecules. 23, 762. doi: 10.3390/molecules23040762
Yao, L., Caffin, N., D'arcy, B., Jiang, Y., Shi, J., Singanusong, R., et al. (2005). Seasonal variations of phenolic compounds in Australia-grown tea (Camellia sinensis). J. Agr. food Chem. 53, 6477–6483. doi: 10.1021/jf050382y
Yilmaz, G., Kandemir, N., and Kinalioglu, K. (2004). Effects of different pruning intervals on fresh shoot yield and some quality properties of tea (Camellia sinensis L. Kuntze) in Turkey. Pak. J. Biol. Sci. 7:1208–1212, doi: 10.3923/pjbs.2004.1208.1212
Yu, X., Liang, C., Chen, J., Qi, X., Liu, Y., and Li, W. (2015). The effects of salinity stress on morphological characteristics, mineral nutrient accumulation and essential oil yield and composition in Mentha canadensis L. Sci. Hortic. 197, 579–583. doi: 10.1016/j.scienta.2015.10.023
Zaman, S., Shen, J., Wang, S., Wang, Y., Ding, Z., Song, D., et al. (2022). Effects of Shading Nets on Reactive Oxygen Species Accumulation, Photosynthetic Changes, and Associated Physiochemical Attributes in Promoting Cold-Induced Damage in Camellia sinensis (L.) Kuntze. Hortic. 8, 637. doi: 10.3390/horticulturae8070637
Zayed, S. M., Aboulwafa, M. M., Hashem, A. M., and Saleh, S. E. (2021). Biofilm formation by Streptococcus mutans and its inhibition by green tea extracts. AMB Express. 11, 1–10. doi: 10.1186/s13568-021-01232-6
Zhang, L. X., Guo, Q. S., Chang, Q. S., Zhu, Z. B., Liu, L., and Chen, Y. H. (2015). Chloroplast ultrastructure, photosynthesis and accumulation of secondary metabolites in Glechoma longituba in response to irradiance. Photosynthetica. 53, 144–153. doi: 10.1007/s11099-015-0092-7
Zhang, Q., Shi, Y., Ma, L., Yi, X., and Ruan, J. (2014). Metabolomic analysis using ultra-performance liquid chromatography-quadrupole-time of flight mass spectrometry (UPLC-Q-TOF MS) uncovers the effects of light intensity and temperature under shading treatments on the metabolites in tea. PLoS ONE. 9, e112572. doi: 10.1371/journal.pone.0112572
Keywords: herbal tea, medicinal properties, polyphenolic compounds, health benefits, environmental factors
Citation: Ramphinwa ML, Mchau GRA, Mashau ME, Madala NE, Chimonyo VGP, Modi TA, Mabhaudhi T, Thibane VS and Mudau FN (2023) Eco-physiological response of secondary metabolites of teas: Review of quality attributes of herbal tea. Front. Sustain. Food Syst. 7:990334. doi: 10.3389/fsufs.2023.990334
Received: 09 July 2022; Accepted: 19 January 2023;
Published: 10 February 2023.
Edited by:
Paras Sharma, National Institute of Nutrition, IndiaReviewed by:
Tabussam Tufail, University of Lahore, PakistanPramod K. Prabhakar, National Institute of Food Technology Entrepreneurship and Management, India
Copyright © 2023 Ramphinwa, Mchau, Mashau, Madala, Chimonyo, Modi, Mabhaudhi, Thibane and Mudau. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Fhatuwani Nixwell Mudau, 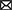 bXVkYXVmQHVrem4uYWMuemE=
bXVkYXVmQHVrem4uYWMuemE=
 Maanea Lonia Ramphinwa1,2
Maanea Lonia Ramphinwa1,2 Mpho Edward Mashau
Mpho Edward Mashau Ntakadzeni Edwin Madala
Ntakadzeni Edwin Madala Vimbayi Grace Petrova Chimonyo
Vimbayi Grace Petrova Chimonyo Thembinkosi Albert Modi
Thembinkosi Albert Modi Tafadzwanashe Mabhaudhi
Tafadzwanashe Mabhaudhi Fhatuwani Nixwell Mudau
Fhatuwani Nixwell Mudau