- 1Department of Food Science and Human Nutrition, College of Agriculture and Food, Qassim University, Buraydah, Saudi Arabia
- 2Department of Food Science and Nutrition, College of Science, Taif University, Taif, Saudi Arabia
- 3Department of Food and Nutrition, Faculty of Human Sciences and Design, King Abdulaziz University, Jeddah, Saudi Arabia
- 4Department of Food Science and Nutrition, College of Food and Agricultural Sciences, King Saud University, Riyadh, Saudi Arabia
Objectives: Fermented cereal beverages are vital for the nutrition of resource-poor populations, as they offer affordable, energy-rich, and safe beverage options. These beverages are integral to traditional food cultures, particularly in Africa and Asia, and they help support local economies through small-scale production and consumption. Probiotic beverages received great attention in last decades due to their positive impacts on the prevention of diseases and promotion of human health. The improvement of traditionally cereal-based fermented beverages is one of the main approaches for production and commercialization of probiotic beverages. Barely-based Sobia beverage (BBSB), is one of the most popular fermented cereal-based probiotic beverages with rising demand in various countries, however, its traditional production method is time-consuming and eliminates scaling up production. Therefore, this study aimed at investigating the impacts of processing methods [traditional (TM) and improved (IM)] on the physicochemical, microbiological and nutritional quality of traditional and improved concentrated BBSB.
Methods: Modification of the TM was done by pasteurization process and incorporation of butter milk, date powder, and ABT-5 probiotic starter culture (Lactobacillus acidophilus LA-5, Bifidobacterium spp. BB-12, and Streptococcus thermophilus) in the formulas. Concentrated BBSB in water or buttermilk (9% total solids) were examined for microbiological quality, total phenolic content (TPC), antioxidant activity (AOA), γ-aminobutyric acid (GABA) content, β-glucan content, volatiles by HS-GC–MS, and in vitro hydrolysis (HI) and glycemic (GI) indices.
Results: Results indicated that concentrated BBSB in buttermilk following the IM showed high levels of lactic acid bacteria (LAB), TPC content, DPPH and ABTS radical scavenging activities, improved microbial quality, high GABA content, and absence of coliform bacteria compared to concentrated BBSB following TM. The highest range of β-glucan was remarked in concentrated BBSB in buttermilk fermented by combining ABT-5 with backer’s yeast regardless of sugar source. The HS-GC–MS analysis identified and quantified 28 volatile compounds. The predominant volatiles in concentrated BBSB were terpenes (49.67–77.56%) followed by alcohols (8.08–32.65%). The HI and GI values were increased for concentrated BBSB in buttermilk (IM) compared to that in water (TM). Application of ABT-5 and bakers’ yeast mixture in the presence of date powder increased HI and GI values of concentrated BBSB.
Conclusion: In conclusion, using the IM enhanced the microbiological and nutritional qualities, increased the health-promoting compounds, improved the in vitro Glycemic index and could be recommended for scaling up commercial applications to produce concentrated BBSB.
1 Introduction
Fermented grain-based beverages are gaining widespread recognition for their capacity to promote food sustainability while delivering enhanced nutritional value. These drinks are produced using microbial fermentation processes applied to various grains, offering multiple advantages such as prolonged storage life, better nutrient bioavailability, and adaptability to diverse dietary requirements. The production techniques and health-beneficial characteristics of these beverages establish their importance in both conventional and contemporary food systems (Phiri et al., 2019; Adebo and Medina-Meza, 2020; Ilango and Antony, 2021; Pswarayi and Gänzle, 2022; Yan et al., 2024). Current research efforts focus on creating novel starter cultures, refining fermentation substrates, and strengthening the functional characteristics of these beverages (Waters et al., 2015; Ignat et al., 2020; Garrido-Galand et al., 2021).
Consequently, functional foods have attracted worldwide attention due to their health-beneficial effects. Functional beverages are recognized for reducing cholesterol and glucose levels, boosting fiber content, strengthening immune function, and supporting digestive processes (Nazhand et al., 2020). Cost-effective probiotic dairy and plant-based products have gained considerable attention in recent years. Various strains of beneficial microorganisms, including yeasts and lactic acid bacteria (LAB), are utilized to create fermented beverages with diverse sensory characteristics, making these fermented drinks appealing to consumers (de Matos Reis et al., 2021). Probiotics can enhance immune function, boost infection resistance, and support digestive health maintenance. They can also prevent the proliferation of harmful food microorganisms. For example, probiotic whey beverages containing Lactobacillus acidophilus and Lacticaseibacillus casei demonstrate inhibitory effects against pathogens including Escherichia coli, Salmonella typhi and Staphylococcus aureus (Turkmen et al., 2019). Bifidobacterium spp. and Lactobacillus spp. represent the primary species utilized in fermented beverages (He et al., 2022).
Grain fermentation processes have recently gained recognition as valuable functional food systems due to their ability to enhance nutritional quality and improve the bioavailability of food compounds. Multiple research studies have demonstrated the health-beneficial characteristics of fermented grain-based products, validating their contribution to nutritionally improved foods (Şanlier et al., 2019). Grains, including barley, maize, millet, oats, rice, rye, sorghum, and wheat, serve as the primary ingredients for fermented beverage production (Salmerón, 2017; Schwan and Ramos, 2019). Spontaneous grain-based fermentation can occur through yeasts, which produce ethanol by transforming grain-derived sugars into ethanol and CO2 (Borai et al., 2021), LAB, and fungi, sometimes creating intricate microbial communities. Examples of grain-based beverages include “Boza,” a spontaneously fermented Bulgarian drink prepared from hulless barley cakes and fermented using yeast and Lactobacilli, resulting in an alcoholic beverage (4% alcohol). Barley-based Sobia beverage (BBSB), commonly known as “Sobia” represents a traditional sweet–sour fermented drink (pH 3.44–4.00) extensively consumed throughout Saudi Arabia and neighboring Middle Eastern regions (Gassem, 2003). BBSB preparation involves mixing malted barley and wheat flour in water, then incorporating sugar and aromatic spices like cardamom and cinnamon. Fermentation proceeds naturally through environmental microorganisms, usually at temperatures ranging from 30 °C to 40 °C for approximately 24 h.
LAB naturally present during BBSB fermentation serve an essential function by preserving and producing nutrient-dense foods/beverages while imparting a characteristic natural sweet–sour flavor to BBSB (Roberfroid, 2002; Borai et al., 2022). The primary probiotic LAB typically identified in BBSB include Lactobacillus cellobiosus, Lactobacillus buchneri, Lactobacillus plantarum, Lactobacillus brevis, Lactobacillus delbrueckii subsp. delbrueckii, Leuconostoc Lactis, and Pedicoccus pentosaceus (Gassem, 2003; Enujiugha and Badejo, 2017).
BBSB has been traditionally consumed primarily for its taste, aroma, and health benefits. Sucrose is typically employed to sweeten BBSB in conventional preparation methods. While numerous studies have examined the microbiological quality and safety of BBSB (Gassem, 2003; El-Said, 2019), its probiotic potential (Enujiugha and Badejo, 2017; Meybodi et al., 2021; Raungrusmee et al., 2022), its alcohol concentration (Borai et al., 2022), and its physicochemical characteristics (Gassem, 2003), no published research has yet focused on improving its nutritional value beyond its inherent nutritional content and sensory attributes (taste and flavor). By substituting regular sucrose with date powder (DP) and utilizing sweet buttermilk as a preparation medium instead of water, a reduced-calorie BBSB can be developed. Additionally, the nutritional profile of buttermilk provides an appropriate food matrix for probiotic microorganism utilization. Meanwhile, the buffering properties of milk proteins can maintain pH stability, thereby supporting probiotic bacteria’s survival throughout processing.
The conventionally uncontrolled BBSB preparation and/or storage methods may present potential public health risks in the final product, including inadequate hygiene and elevated contamination risk. Moreover, its preparation process can be lengthy. Therefore, this research seeks to develop a precise and controlled BBSB preparation method requiring shorter fermentation time while preserving and enhancing its health-promoting compounds by adding natural DP and sweet buttermilk. The improved methodology also aims to regulate alcohol production by minimizing its formation.
2 Materials and methods
2.1 Materials
Organic barley, cinnamon, cardamom, sugar, date powder, and commercial yeast were obtained from the local store in Buraydah, Saudi Arabia. Buttermilk powder (nutritional facts per 100 g; 400 calories, 33.3 g protein, 6.66 f fat, 53.28 g carbohydrates, 33.3 mg cholesterol, 466.2 mg sodium, 880 mg calcium, 1,772 mg potassium, and 20 mg vitamin D) was purchased online from iherb (manufactured by Bob’s Red Mill natural foods, Milwaukie, USA). The Direct VAT Set (DVS) ABT-5 starter culture, which contains Lactobacillus acidophilus LA-5, Bifidobacterium spp. BB-12, and Streptococcus thermophilus (Chr. Hansen Laboratories, Copenhagen, Denmark) was purchased from Misr Food Additives (MIFAD), Badr City, Egypt. Additionally, a mixed-linkage β-glucan kit was acquired from Megazyme International in Bray, Ireland, and the γ-aminobutyric acid (GABA) analytical standard was purchased from Sigma-Aldrich in Saint Louis, Missouri, USA.
2.2 Preparation of concentrated Sobia beverages
Under the current study, eight different formulas of fermented concentrated BBSB were prepared according to the ingredients’ combinations in Table 1 and the processing flow chart in Figure 1. The formulations were divided into water based BBSB (4 formulations) and buttermilk based BBSB (4 formulations). Two water-based formulations were prepared using the conventional method by adding Baker’s yeast and two by adding yeast and ABT-5 starter culture using normal sugar (WBS-Y or WBS-Y-ABT-5) or DP (WBD-Y or WBD-Y-ABT-5). The other four formulations were buttermilk-based and were prepared by adding ABT-5 culture only or both yeast and ABT-5 starter culture using normal sugar (BMS-ABT-5 and BMS-Y-ABT-5) or DP (BMD-ABT-5 and BMD-Y-ABT-5). The purpose of varying the starter culture was to understand the effect of using probiotic culture only or in combination with baker’s yeast.
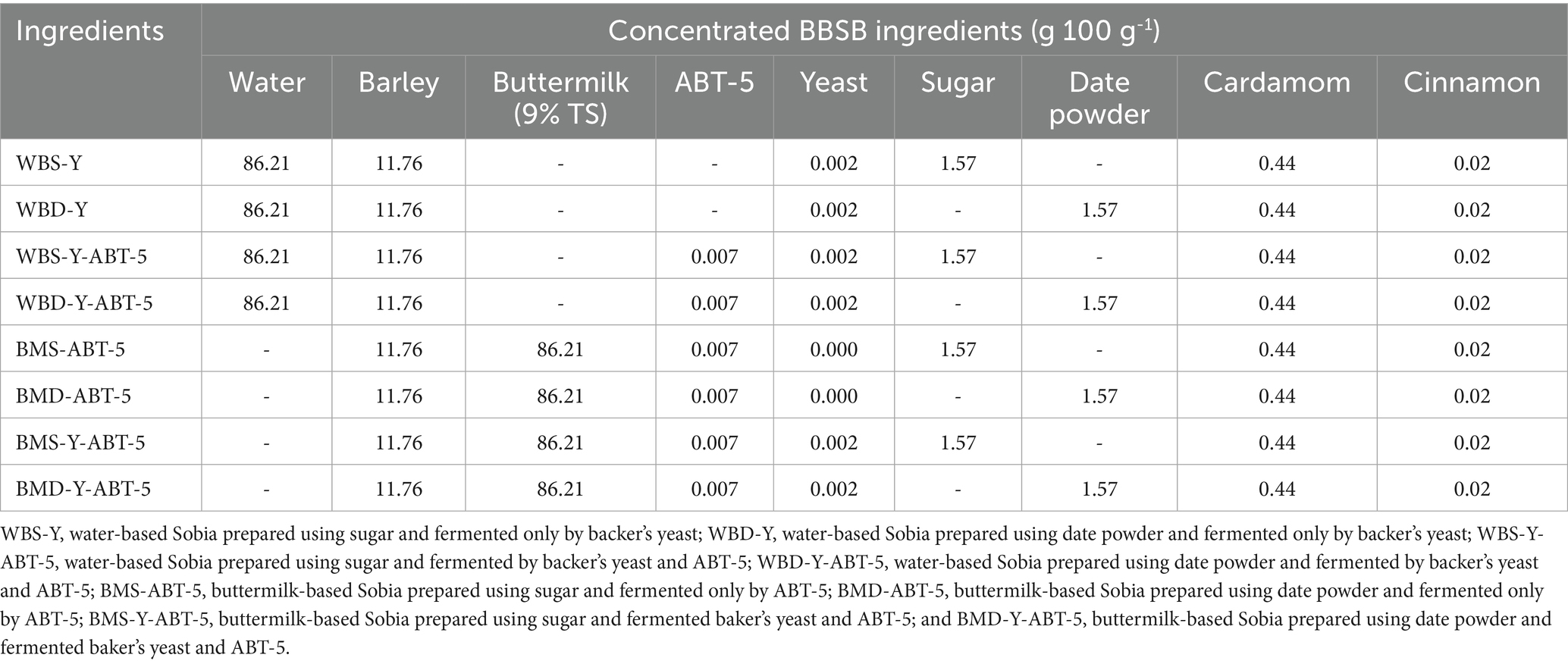
Table 1. Different formulas of concentrated Sobia beverages prepared using the traditional and developed method.
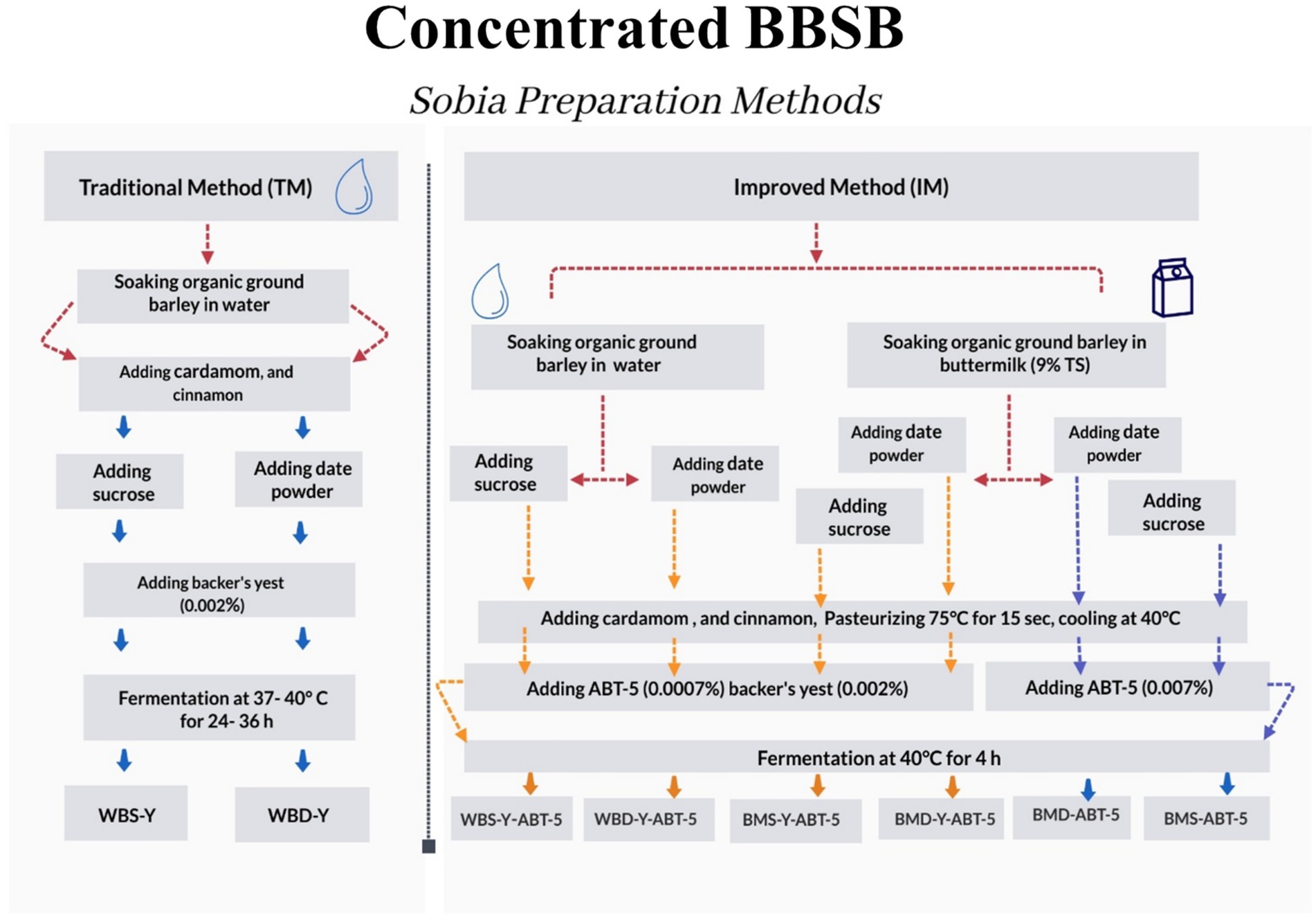
Figure 1. Processing flow chart of different concentrated BBSB formulas: see the tail of Table 1. The blue dashed arrows indicate the flowchart for BBSB beverages fermented with ABT-5 only, while the yellow dashed arrows show the steps for those fermented with both ABT-5 and baker’s yeast.
2.3 Methods of analysis
2.3.1 Total phenolic content (TPC) determination
The total phenolic content (TPC) of BBSB extracts was determined using the Folin–Ciocalteu colorimetric assay, as described by Nsimba et al. (2008). Briefly, 150 μL of the sample was mixed with 300 μL of Folin–Ciocalteu reagent and allowed to react for 5 min. Subsequently, 300 μL of 7.5% (w/v) sodium carbonate solution was added. The reaction mixture was incubated in the dark at 23 °C for 60 min prior to measurement. After incubation, the mixture was centrifuged at 10,000 × g for 10 min at 4 °C. The supernatant (120 μL) was transferred to a new plate, and the absorbance was measured at 765 nm using a microplate reader (BioTek, Winooski, VT, USA). Finally, the concentration of TPC was calculated by a standard calibration curve prepared using gallic acid (GA) solution (R2 = 0.99), and TPC content was expressed as milligrams of gallic acid equivalents (GAE) per 100 g (mg of GAE g − 1 dry weight).
2.3.2 Total antioxidant capacity (TAC) determination
According to DPPH radical bleaching (Nsimba et al., 2008), the TAC values of BBSB extracts were determined spectrophotometrically. In summary, 600 μL of 0.2 mM DPPH solution was added to 120 μL of the sample in an Eppendorf tube and incubated in the dark at 23 °C for 60 min. After centrifugation at 10,000 × g for 10 min at 4 °C, 120 μL of supernatant was transferred to a plate reader (BioTek, Winooski, VT, USA) and measured at 517 nm. Trolox calibration curve is plotted using the DPPH radical scavenging activity percentage. Radical scavenging activity against ABTS radicals was tested using the method described by Lu et al. (2007). 0.1 mL of the beverage was mixed with 2.9 mL of diluted ABTS radical cation solution. The ABTS radical cation solution was prepared by mixing 7 mmol/L ABTS with 2.45 mmol/L potassium persulfate in distilled water. This mixture was incubated in the dark at room temperature for 12 to 16 h to generate the ABTS radical solution. Prior to analysis, the ABTS solution was diluted with ethanol to achieve an absorbance of 0.70 ± 0.02 at 734 nm and was allowed to equilibrate at 30 °C. For the assay, appropriate aliquots of the sample were added to the diluted solution and allowed to react at 30 °C for 20 min. The decrease in absorbance was then measured at 734 nm using a UV–Vis spectrophotometer. Antioxidant activity was expressed as micromoles of Trolox equivalents per gram of sample (μmol TE g−1), based on a standard curve constructed with known concentrations of Trolox.
2.3.3 β-glucan content determination
The total β-glucan content of concentrated BBSB samples was determined in duplicate using a mixed-linkage β-glucan assay kit from Megazyme International (Wicklow, Ireland), following the manufacturer’s instructions. In summary, the samples were washed multiple times with ethanol to eliminate residual sugars and lipids. The ethanol-washed residues were then enzymatically hydrolyzed using lichenase at 50 °C for 1 h, followed by treatment with β-glucosidase at the same temperature for 10 min. Afterwards, glucose concentrations were measured by incubating the hydrolysates with glucose oxidase/peroxidase (GOPOD) reagent at 50 °C for 20 min. The absorbance was recorded at 510 nm, and the β-glucan content was expressed as a percentage on a dry weight (DW) basis (g/100 g DW).
2.3.4 γ-aminobutyric acid (GABA) content determination
The γ-aminobutyric acid (GABA) content in concentrated BBSB was determined using a spectrophotometric method based on the procedure described by Yuwa-Amornpitak et al. (2020). Briefly, 0.5 mL of the sample was mixed with 0.5 mL of borate buffer, 0.5 mL of 6% (w/v) phenol reagent, and 1.5 mL of 6% (v/v) sodium hypochlorite (NaClO). The reaction mixture was heated in a boiling water bath at 100 °C for 10 min, then immediately cooled in an ice bath. Absorbance was measured at 630 nm using a spectrophotometer. GABA concentration was calculated using a standard calibration curve and expressed as milligrams per 100 grams (mg/100 g).
2.3.5 Headspace gas chromatography–mass spectrometry analysis (HS-GC–MS)
The volatile flavor compounds (area %) in different concentrated BBSB were detected using Head Space-Gas Chromatography–Mass Spectrometry analysis (HS-GC–MS). The GC–MS system (Agilent Technologies) was equipped with a gas chromatograph (7890B) and mass spectrometer detector (5977A). Headspace temperature program: oven temperature 80 °C, needle temperature 120 °C, transfer line temperature 140 °C and incubation time 20 min. The GC had a DB-624 column (30 m × 320 μm internal diameter and 1.80 μm film thickness). Analyses were carried out using hydrogen as the carrier gas at a flow rate of 3 mL/min at a splitless injection volume of 1 μL and the following temperature program: 40 °C for 1 min; rising at 7 °C/min to 250 °C and held for 5 min. The injector and detector were held at 250 °C. Mass spectra were obtained by electron ionization (EI) at 70 eV, using a spectral range of mass to charge (m/z) ratio of 30–550. Different constituents were identified by comparing the spectrum fragmentation pattern with those stored in Wiley and NIST Mass Spectral Library data.
2.3.6 In vitro glycemic index (GI) and hydrolysis index (HI) analysis
For the determination of in vitro GI, different concentrated BBSB was used. The method described by Aribas et al. (2020) was used after following the modification of Alwohaibi et al. (2023).
In order to calculate the GI, it is necessary to determine the HI for each sample. This is achieved by employing the following equation.
Then the in vitro GI was determined by using the following equation
2.3.7 Mineral content determination
The mineral content of concentrated BBSB was analyzed using ICP-OES with an Optima 4,300 DV system (PerkinElmer, MA, USA), following the method by Milani et al. (2018). A 1 g sample of dried BBSB was digested in 5 mL of concentrated sulfuric acid at room temperature for 24 h. Then, 3 mL of 35% hydrogen peroxide was added, and the mixture was thermally digested at 400 °C for 2 h using a closed Kjeldahl system. The sample volume was adjusted to 50 mL with distilled water before analysis. The ICP-OES parameters included radio frequency power of 0.7 to 1.5 kW, plasma gas flow rates of 10.5 to 15 L/min (radial) and 15 L/min (axial), and an axial viewing height of 5 to 12 mm. Calibration used a certified reference mineral solution from the National Institute of Standards and Technology (Gaithersburg, MD, USA).
2.3.8 Microbiological quality of different concentrated BBSB
After preparation, samples of BBSB were immediately collected in sterilized bottles and analyzed for their microbiological quality. To perform microbial examination, 10 mL of the sample was mixed with 90 mL of 0.1% w/v sterile peptone water (Oxoid, Hampshire, UK) and appropriate serial dilutions in 0.1% peptone water were prepared. The total plate count was determined by the pour plate method using Standard Plate Count Agar, PCA (Oxoid, UK). The plates were incubated at 37° C for 48 h. The viable starter culture strains of ABT were counted on de Man Rogosa and Sharpe (MRS) and M17 agar media. To inhibit the growth of S. thermophilus, a lower pH value (5.5) MRS agar medium was used for both L. acidophilus and Bifidobacteria. Anaerobic incubation was performed for 72 h at 37 °C to count viable bacterial strains. The enumeration of viable S. thermophilus was carried out on M17 agar after aerobic incubation at 43 °C for 48 h.
2.3.9 Statistical analysis
The statistical analysis was carried out by applying one-way ANOVA using SPSS (Ver. 22.0 for Windows). The experimental results were expressed as mean ± standard error. Multiple comparisons were carried out by applying Tukey’s test, and the significance level was set at p < 0.05. Data were treated as a complete randomization design (Steel et al., 1997).
3 Results and discussion
One traditionally produced and consumed cereal-based beverages in Saudi Arabia is BBSB, a traditional fermented barley beverage probably consumed during Ramadan. Traditional BBSB uses barley, wheat, sugar, water, yeast, spices, cinnamon, and cardamom. Usually, ingredients are combined, fermented for around 24 h at 30–40 °C, strained, cooled then served (Gassem, 2002; Borai et al., 2021). Unfortunately, the preparation method is time-consuming, uncontrolled, and generates alcohol in addition to safety and hygiene issues (Borai et al., 2021). Therefore, the current research was designed to develop a controlled method of preparing concentrated BBSB by maintaining and improving the product’s nutritional value and increasing prebiotics and probiotics properties. As lactic acid bacteria were reported as the most commonly used starters in the food industry (Hole et al., 2012; Mubarak et al., 2013), and fermented milk is currently the most popular commercial functional beverage, and it has nutritional and therapeutic properties (Hamed et al., 2018a; Hamed et al., 2018b) supported by WHO recommendations as it contains probiotics, which positively improve human health when consumed regularly (Kobyliak et al., 2018; Infusino et al., 2020). ABT-5 starter was currently used in preparing concentrated BBSB in water or buttermilk with sucrose or date powder as a sweetening agent, as well as a thermal treatment that had been used to ensure safe and hygienic conditions.
3.1 Phytochemicals and antioxidant capacity of different concentrated BBSB
The TPC and relative antioxidant activities using DPPH and ABTS assays of different concentrated BBSB formulas were investigated, and data are tabulated in Table 2. TPC significantly differed among proposed treatments. The highest TPC content was presented in the BMD-Y-ABT-5 formula, which significantly differed from other treatments, while the lowest TPC content was remarked in the WBS-Y formula. Interestingly, adding DP increased the TPC considerably, as recorded in WBD-Y, WBD-Y-ABT-5, BMD-ABT-5, and BMD-Y-ABT-5 compared to WBS-Y, WBS-Y-ABT-5, BMS-ABT-5, and BMS-Y-ABT-5. Concentrated BBSB formulas fermented by ABT-5 alone or fermented by combined backer’s yeast and ABT-5 showed higher TPC fermented by backer’s yeast alone. Accordingly, the antioxidant capacity increased with increased TPC, correlated to the presence of buttermilk and date powder. The highest values of DPPH and ABTS radical scavenging activities were recorded in formulated concentrated BBSB in buttermilk incorporated date powder and fermented by either ABT-5 or combined ABT-5 with backer’s yeast. The effect of fermentation on the TPC and antioxidant activities in cereals was previously reported (Đorđević et al., 2010). They noted that fermentation led to an increase in TPC and antioxidant activities in four cereals, namely buckwheat, wheat germ, barley and rye, with higher antioxidant activity in cereals fermented with L. rhamnosus, compared with cereals fermented with S. cerevisiae. The authors reported this to be due to the release of bound phenolics during fermentation due to structural breakdown of cereal cell walls, leading to the liberation and/or synthesis of various bioactive compounds (Katina et al., 2007). The authors also reported a positive influence on DPPH inhibitory effect in each cereal species after fermentation with L. rhamnosus however, fermentation with S. cerevisiae had no significant influence on antioxidant activity. Interestingly, yeast preparations themselves might show different antioxidant activities, as using different yeasts (Moore et al., 2007). Fermentation of cereals can alter the composition and availability of bioactive compounds as a result of enzymatic activities originating from both the grains and associated microbial communities (Đorđević et al., 2010). Microbial enzymes including glycosidases, hydrolases, esterases, and β-glucosidases produced by various bacterial strains play a key role in enhancing the bioaccessibility of phenolic compounds present in the grain matrix (Chen et al., 2020). Alharbi et al. (2022) and Algonaiman et al. (2022) showed that fermentation by L. plantarum significantly altered the composition of the oats’ extracts with a significant increase in TPC and related antioxidant activity, which agreed with many other studies. The explanation of the higher degree of TPC in cereals fermented by LAB than those fermented by yeast was related to differences in pH of different fermentations, which may affect the liberation of enzymes responsible for the degradation of cell wall, owing to changes in optimum pH (Boskov Hansen et al., 2002).
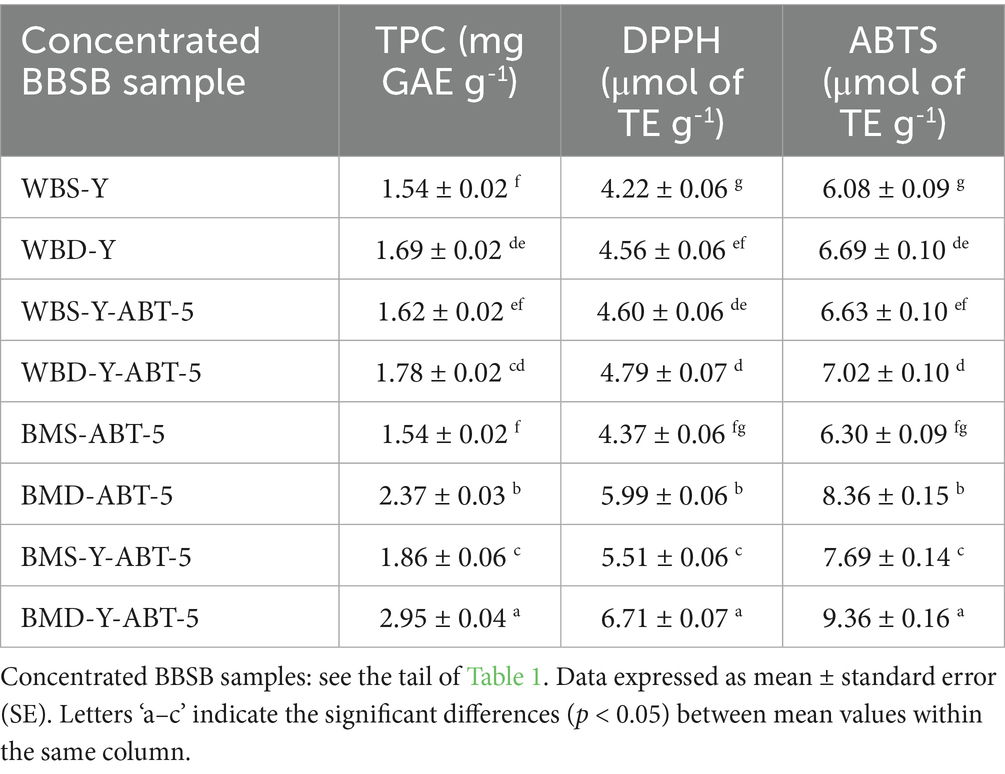
Table 2. Total phenolic content (TPC), potential antioxidant activities, γ aminobutyric acid, and β-glucan contents in different concentrated Sobia beverages (mean ± SE).
Interestingly, adding DP increased the TPC considerably, as recorded in WBD-Y, WBD-Y-ABT-5, BMD-ABT-5, and BMD-Y-ABT-5 compared to WBS-Y-ABT-5, BMS-ABT-5, and BMS-Y-ABT-5. The highest values of DPPH and ABTS radical scavenging activities were recorded in beverages formulated in buttermilk incorporated DP and fermented by either ABT-5 or combined ABT-5 with backer’s yeast. This may be due to the prebiotic effect the DP may have which accelerate the growth and activity of the starter and release a large amount of phenolics. In addition, TPC is supposed to be obtained from the decomposition of DP. Aljutaily et al. (2022) demonstrated that the Sukkari date represents phenolics and minerals such as K, Mg, Zn, Fe, and Cu. TPC, DPPH antiradical activity and probiotic potential (> 7.8 CFU/mL of LAB) were improved when date flesh extracts were incorporated into yogurt formulations (Ghafoor et al., 2023). It was previously reported that a 1.2-fold of increase in TPC upon LAB fermentation of the buttermilk using P. acidilactici BD16 (alaD+) from 5.98 ± 0.16 to 6.25 ± 0.04 mg GAE/mL when compared with the unfermented buttermilk (Sharma et al., 2021).
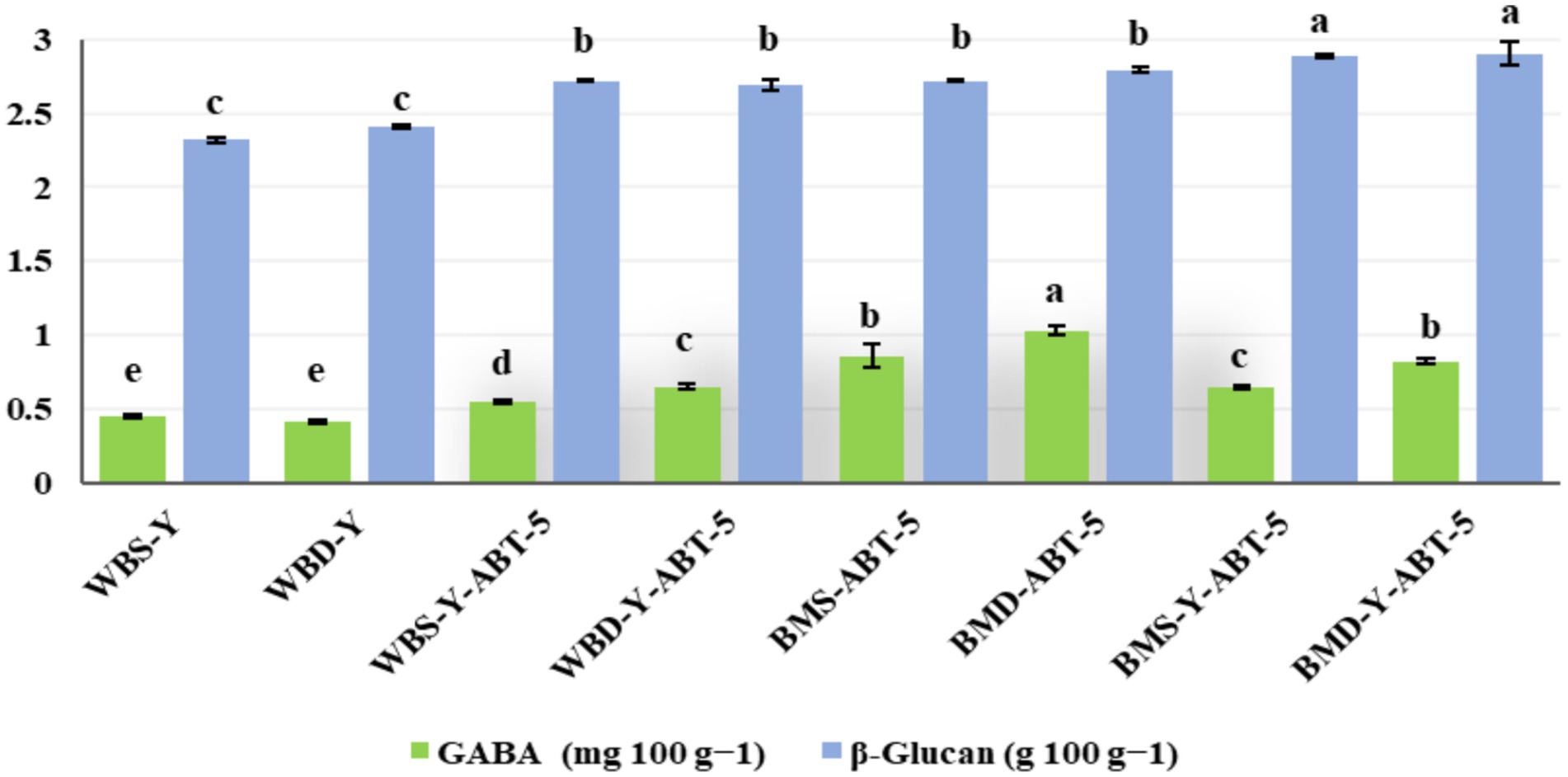
3.2 GABA and β-glucan contents during fermentation
The GABA and β-glucan contents in different concentrated BBSB samples are presented in Figure 2. Formulation and fermentation significantly changed the GABA content. The GABA content was increased considerably when formulated concentrated BBSB in water was fermented by combining ABT-5 with backer’s yeast. A significant difference was found between WBS-Y, WBD-Y, and WBS-Y-ABT-5, even adding date powder accelerated GABA content significantly, as remarked in the WBD-Y-ABT-5 formula. Significant increases in GABA content were recorded in concentrated BBSB formulas prepared in buttermilk. Among these formulas, BMD-ABT-5 presented the highest GABA content, and BMS-Y-ABT-5 gave the lowest GABA content. As remarked, combining fermentation of concentrated BBSB with ABT-5 and backer’s yeast affected the GABA content, whereas fermenting concentrated BBSB with ABT-5 alone with adding date powder presented the highest GABA content.
GABA is a non-protein amino acid that works as a neurotransmitter in the mammals’ brain (Mohd Ali et al., 2013) and has been demonstrated to play several roles in health-promoting activities such as anti-hypertension and antidiabetic activities (Soltani et al., 2011). GABA synthesis is catalyzed by glutamate decarboxylase (GAD) and therefore, biochemical properties of the enzyme may be affected by optimal fermentation conditions (Dhakal et al., 2012). The GABA content increased considerably when formulated beverages were fermented by ABT-5 than by combining ABT-5 with backer’s yeast. The ABT-5 culture contains LAB (Streptococcus thermophilus, Lactobacillus acidophilus, Bifidobacterium bifidus), which are known for their GAD enzyme activity (Sahab et al., 2020). Although GAD has been also isolated from yeast species such as Saccharomyces cerevisiae and Kluyveromyces marxianus, these normally have lower GAD activity than LAB (Perpetuini et al., 2020). Furthermore, yeasts such as S. cerevisiae may need to utilize the GABA as nitrogen source during ethanol fermentation which may explain the lower amount detected in beverages formulated by combining ABT-5 and yeasts (Ando and Nakamura, 2016). De Barber et al. (1989) reported similar results in sourdough in which LAB produced GABA however, it was assimilated by yeast in the dough. Tsolmonbaatar et al. (2016) isolated a yeast mutant defective in assimilation of GABA from a strain of baker’s yeast which consumed less GABA. Furthermore, yeasts may consume GABA using semialdehyde dehydrogenase (SSADH) during fermentation, thereby reducing the amount of GABA produced (Ando and Nakamura, 2016).
DP addition was shown to result in a significantly higher GABA production rate than sucrose, either in water or milk-based beverages. Sukkari dates have a wide profile of simple sugars such as glucose (10.07 ± 0.28) and disaccharides such as fructose (10.09 ± 0.07) and sucrose (43.51 ± 0.34), as previously reported (Zhang et al., 2015). As GABA is normally produced from glucose, this means that, in formulas containing sucrose as the sweetening agent, sucrose needs to be firstly broken into simple sugars for it to be used by the bacteria to multiply and to produce GABA. In a recent study, Hussin et al. (2021) compared the ability of simple sugars and commercial prebiotics to enhance natural GABA production in yoghurt. The authors reported that simple sugars enhanced GABA production (42.83–58.56 mg/100 g) compared to the prebiotics (34.19–40.51 mg/100 g). This is expected as glucose is readily phosphorylated to glucose-6-phosphate to achieve bacterial cell growth. On the other hand, sucrose and fructose need to go through additional conversion steps in the phosphoenolpyruvate-dependent phosphotransferase system before conversion to pyruvate (Li et al., 2020), which then either splits into the GABA-shunt pathway to make GABA or continues to be decarboxylated to generate ATP, NADH and NADPH for cell growth through the glycolytic pathway (Shan et al., 2012). Direct metabolism of glucose may result in the rapid bacterial growth to reach an exponential phase in a shorter time which then contributes to increased GAD secretion and glutamate conversion into GABA. The remark that could be made is that combining fermentation with ABT-5 and backer’s yeast affected the GABA content, whereas fermenting with ABT-5 alone with adding DP presented the highest GABA content.
Cereal β-glucan is a water-soluble bioactive polysaccharide and dietary fiber which can be found in cereals such as oat and barley. It has a prebiotic effect and can be completely fermented by gut microbiota (Aljutaily et al., 2025). The β-glucan content is shown in Figure 2. The results have shown no significant effect of the medium used (water or buttermilk) or the sugar source (sucrose or DP). On the other hand, the type of the starter culture used significantly influenced β-glucan content. In this sense, the beverages formulated using both ABT-5 and baker’s yeast had significantly higher amount of β-glucan than those formulated using ABT-5 only. This might be due to the ability of the yeast to produce β-glucan, above what is already present in barley, which has been reported elsewhere (Rizal et al., 2021). It was also reported that prolonged fermentation time when using only LAB, compared with using LAB and yeasts, in barley-based dairy products reduces β-glucan content (Tiwari and Cummins, 2009). Also, the research conducted by Zhang et al. (2022) revealed that both the concentration and characteristics of β-glucan in fermented barley beverages are significantly influenced by the choice of starter culture and the specifics of the fermentation process. Their findings indicate that the use of Lactobacillus plantarum as the starter culture decreases its molecular weight and enhances its antioxidant properties, enzyme-inhibitory effects, and cholesterol-adsorbing capabilities, as the reduction in the molecular weight of barley β-glucan, accompanied by a transformation in its microstructure from predominantly rod-like shapes to more sheet-like configurations. Although there are no studies that specifically investigate ABT-5, similar fermentation processes indicate that the health-related properties of barley β-glucan improve after fermentation.
3.3 Volatile flavor compounds of BBSB
It is well known that flavor compounds produced during fermentation are important for the acceptability of the final product. HS-GC–MS was used to detect volatile components in formulated BBSB beverages. Alcohols, terpenes, acids, aldehydes and ketones, and other secondary metabolites produced during fermentation and processing are presented in Table 3 and are graphically represented in Figure 3. A total of 26 volatile flavor compounds were identified under conditions of the current study. Total terpenes represent the highest relative content in all samples compared to other volatile aroma groups, followed by the alcohols and aldehydes then ketones. The distribution of volatile compounds and the dominant ones in each group are clearly affected by different factors (preparation medium, sweetening agent and type of fermenting culture).
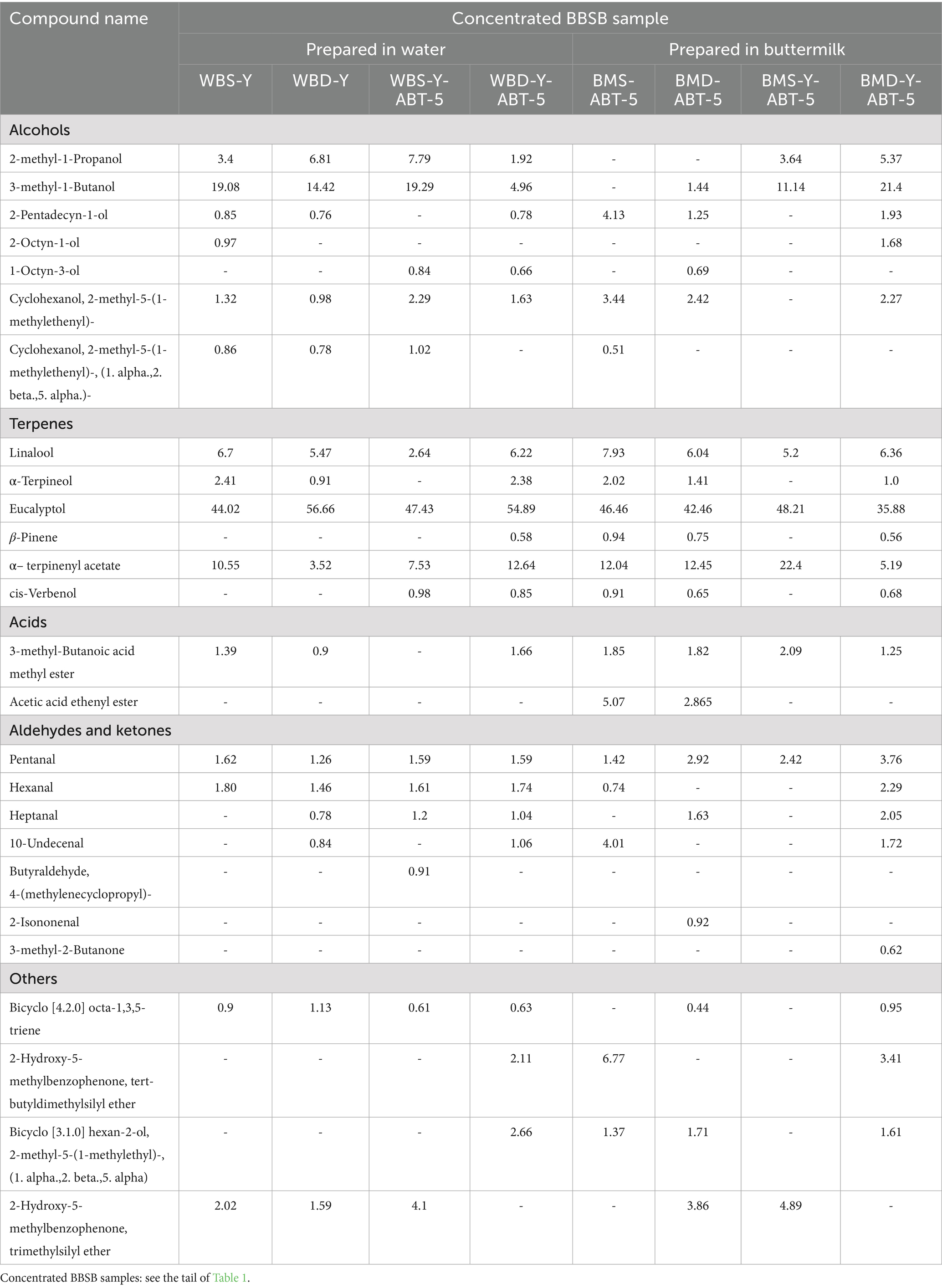
Table 3. Volatile flavor compounds (area %) of different concentrated Sobia beverages quantitated by HS-GC–MS.
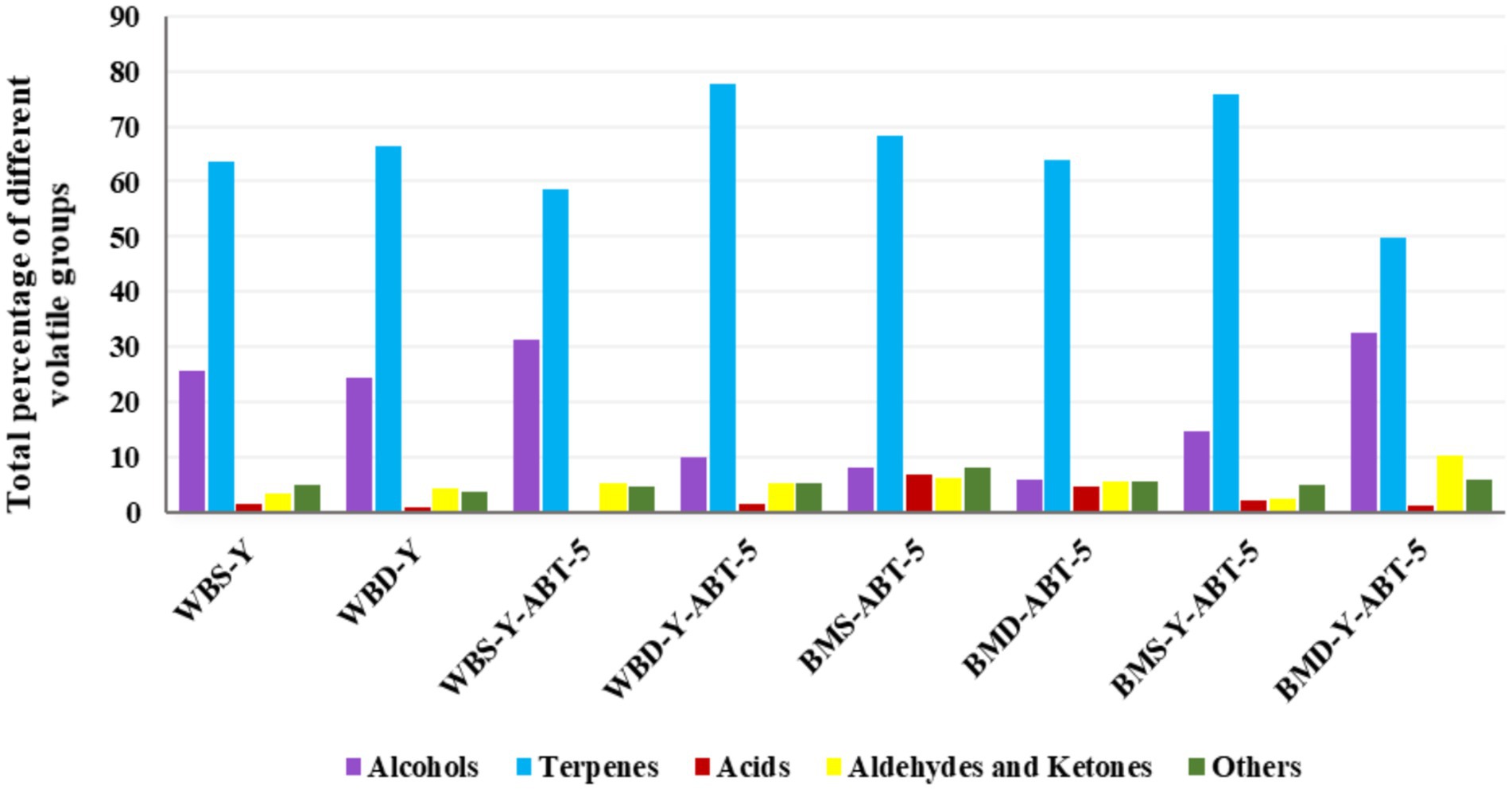
Figure 3. Graphical representation of volatile flavor compounds (Area %) of different concentrated Sobia beverages quantitated by HS-GC–MS. Concentrated BBSB samples: see the tail of Table 1.
Regarding terpenes, the dominant terpenes compound in all samples was Eucalyptol (1,8-cineole), especially those samples prepared in water. In contrast, Eucalyptol was lowered in samples prepared in buttermilk. Regarding the samples prepared in buttermilk, the Eucalyptol relative amounts were higher in samples containing white sugar (BMS-ABT-5:46.46% and BMS-Y-ABT-5: 48.21%). The α–terpinenyl acetate was distributed in the second level among the flavor compounds. The highest amount of α–terpinenyl acetate was for BMS-Y-ABT-5 (22.4%), while the lowest amount was for BBSB-containing date powder and prepared traditionally (WBD-Y: 3.52). Several aromatic plants naturally contain terpenes in high amounts. Thus, the reason behind increasing terpenes amounts in all prepared formulations could be due to the addition of both cinnamon and cardamom in the formulation of the samples especially for eucalyptol (mint, sweet aroma) and α-terpenil acetate, which represent the higher amounts in cardamom (Tambe and Gotmare, 2019). Monoterpenes like α-terpineol are also found in plants, contributing to their fruity aromas (Melgarejo et al., 2011). Linalool may be formed during fermentation through the enzymatic cleavage action of bacterial glycosidase on glycosides (Michlmayr et al., 2012). Glycosides were reported in cereals (Maier et al., 1995).
Most BBSB samples prepared in water had a higher percentage of total alcohol than those prepared in buttermilk. The samples prepared in buttermilk and fermented only by ABT-5 had the lowest alcohol-relative amounts (BMS-ABT-5: 8.08%, and BMD-ABT-5: 5.8%). At the same time, it is observed that fermentation of those samples with both yeast and ABT-5 highly increased the amounts of released alcohols (BMS-Y-ABT-5: 14.78%, and BMD-Y-ABT-5: 32.65%). Moreover, higher levels of alcohols in BMS-Y-ABT-5 (14.78%) and BMD-Y-ABT-5 (32.65%) samples result from the hetero fermentation of sugars by yeasts which yields a high level of alcohols.
Total alcohol contents became in the second level after terpenes in flavor compound groups. The low total alcohol content was for samples fermented only by ABT-5, which was noted to be 5.8 and 8.08% for BMD-ABT-5 and BMS-ABT-5 samples, respectively. The 19.08 and 19.29% of 1-Butanol, 3-methyl in WBS-Y and WBS-Y-ABT-5 were detected as the highest among acids in the other BBSB samples. The higher level of alcohols in sample containing DP and fermented with both yeast and ABT-5 may be due the high level of fermentable sugars in Sukkari date as previously mentioned, which increases the availability of those sugars. The most detected abundant volatile alcohols were 2-methyl-1-Propanol and 3-methyl-1-Butanol. In a study to investigate the volatiles produced by different Rhizopus oligosporus strains and Lactobacillus Plantarum grown on malt extract agar, barley and soybean, ethanol, acetone, ethyl acetate, 2-butanone, 2-methyl-1-propanol, 3-methyl-1-butanol and 2-methyl-1-butanol were dominant compounds on three substrates.
Total aldehydes and ketones relative amounts were detected in higher percent-ages for treatments containing date powder and fermented with yeast and ABT-5 than other BBSB samples. The relative amounts of aldehydes and ketones in BMD-Y-ABT-5 (10.44%) was almost double their content in the WBD-Y-ABT-5 (5.43%) sample. Acetaldehyde, 2-methyl-propanal, ethanol, 2-methyl-1-butanol and 3-methyl-1-butanol were the most abundant volatile compounds produced on MEA and barley (Feng et al., 2007). Cereal-fermented beverages were historically produced to effectively conserve and utilize various types of cereals and crops in a financially sustainable way. Traditionally, these alcoholic beverages have been transformed into non-alcoholic and soft beverages, which are now commercially available. In a product similar to Sobia, the Boza traditional beverage was familiar in different countries. Boza’s alcohol content differs by country (Turkey <1% ABV, Egypt up to 7%) due to varied fermentation microorganisms (Ignat et al., 2020).
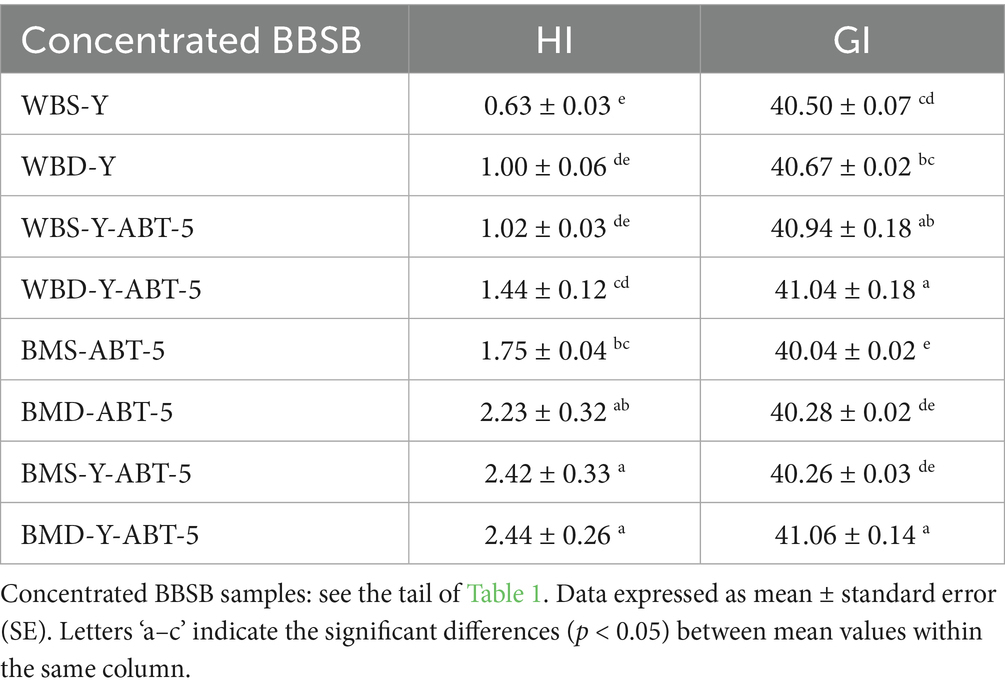
Regarding the total acidic compounds, they were detected in low relative amounts compared to other volatile compounds for all concentrated BBSB samples. However, there was no detection of the acetic acid ethenyl ester in most of the BBSB samples. But it was detected in samples prepared in buttermilk and fermented by ABT-5 alone, whether it contained white sugar (BMS-ABT-5: 5.07%) or date powder (BMD-ABT-5:2.87%). Thus, it could be said that the fermentation of beverages by starter cultures, especially when containing dairy ingredients like buttermilk, is highly characterized by the presence of organic acid compounds, aldehydes, and ketones (Maarse, 2017). Literature evidence supports the fact that organic acids, such as lactic acid, produced during barley fermentation, has the ability to reduce the overall GI and, therfore, to prevent sugar implication on health (Alminger and Eklund-Jonsson, 2008; Maarse, 2017). In this study, all formulas made with buttermilk and DP can be categorized as functional low-GI beverages ranging between 40 and 41, as shown in Table 4. As these concentrated beverages are normally diluted before consumption, which will further reduce GI, this makes these findings of particular interest in formulation of healthy products.
3.4 In vitro GI amounts of different concentrated BBSB samples
Table 4 illustrates the HI and GI of different concentrated BBSB formulas. Regarding the HI, it could be noticed that adding date powder accelerated the hydrolysis and increased HI value when compared WBS-Y with WBD-Y. Consequently, adding ABT-5 to prepared concentrated BBSB in water continuously increased the HI values, as remarked in WBS-Y-ABT-5 and WBD-Y-ABT-5. The HI values were increased for prepared concentrated BBSB in buttermilk compared to prepared concentrated BBSB in water. As recorded, using ABT-5 as a fermenting agent was accelerated in the presence of date powder instead of sucrose. However, combining ABT-S with Backers’ yeast improved the HI by adding sugar. The most efficient treatments were spontaneously preparing concentrated BBSB in buttermilk, with date powder, fermented by ABT-5 and bakers’ yeast. In the same table, data from GI were tabulated. The GI value increased when concentrated BBSB was prepared in buttermilk, with date powder, and fermented by ABT-5 and bakers’ yeast. The table shows that WBD-Y-ABT-5 and BMD-Y-ABT-5 significantly increased compared to other treatments. The GI value in-creased when concentrated BBSB was prepared in BM, with DP, and fermented by ABT-5 and bakers’ yeast along with. The table shows that WBD-Y-ABT-5 and BMD-Y-ABT-5 significantly increased compared to other treatments. The enhanced mineral profile of sample containing buttermilk and DP is likely due to the fact that milk and date products are considered as rich sources of macro and micro minerals (Vayalil, 2012; Zwierzchowski and Ametaj, 2018). In some samples, the content of some minerals in-creased when a combination of yeast and ABT-5 was used compared to the use of ABT-5 culture alone, the opposite was seen in other samples for other minerals. It is also apparent that fermenting microbes (yeast and LAB) could contribute to the increase or reduction of mineral content in fermented BBSB. This is because those minerals are consumed by fermenting microbes during growth and metabolism; however, these microbes could also degrade complex anti-nutritional factors in barely and thereby in-crease the release and extractability of minerals (Samtiya et al., 2021). Overall, the findings of this study revealed that incorporation of buttermilk and DP into BBSB formulation may shorten the fermentation time and improve the nutritional quality of the product by enhancing the mineral contents of the final product.
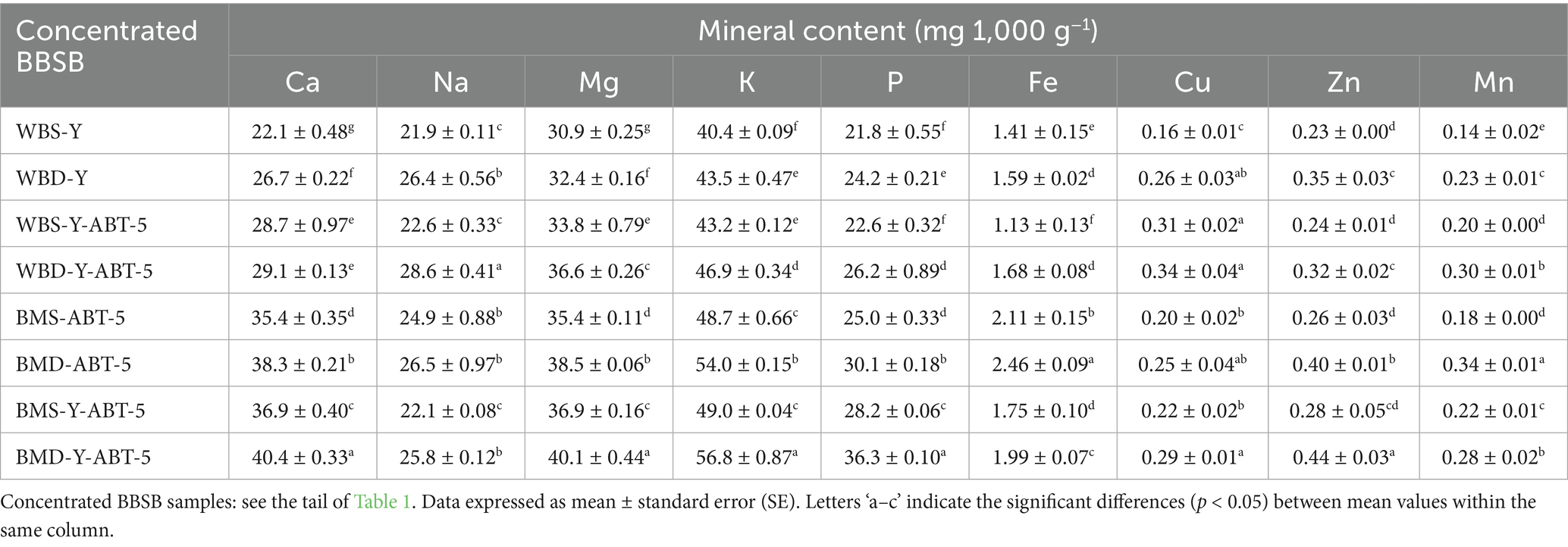
3.5 Mineral contents of different concentrated BBSB samples
The results of macro-and micro-mineral of different BBSB samples were presented in Table 5. The macro mineral contents greatly differed among the formulas, with the highest values of macro minerals of BBSB formulated using the improved method with the addition of barley, buttermilk, and date powder regardless of fermentation microbe. At the same time, microminerals showed slight variations among the BBSB samples. The highest values of calcium, potassium, phosphorous, and magnesium were observed in the BMD-Y-ABT-5 BBSB sample. In contrast, the least values of these minerals were seen in WBS-Y, indicating that the incorporation of date powder and buttermilk improved the contents of these minerals in fermented BBSB. The highest sodium level was observed in WBD-Y-ABT-5, whereas the lowest values were found in WBS-Y, WBS-Y-ABT-5, and BMS-Y-ABT-5 formulas. The highest value of iron was seen in the BMD-ABT-5 sample, followed by the BMS-ABT-5 sample. In contrast, the least value was observed in the WBS-Y formula suggesting that the incorporation of date powder and buttermilk improved the iron content of BBSB fermented with ABT-5; however, fermentation with a combination of yeast and ABT-5 resulted in the reduction of iron content. The highest zinc content was observed in the BMD-Y-ABT-5 formula, whereas the lowest values were seen in different BBSB formulas. WBD-Y-ABT-5 and BMD-ABT-5 possessed the highest values of copper and manganese, respectively, whereas WBS-Y showed the least values of these micro minerals.
The enhanced mineral profile of sample containing BM and DP is likely due to the fact that milk and date products are considered as rich sources of macro and micro minerals (Vayalil, 2012; Zwierzchowski and Ametaj, 2018). In some samples, the con-tent of some minerals increased when a combination of yeast and ABT-5 was used compared to the use of ABT-5 culture alone, the opposite was seen in other samples for other minerals. It is also apparent that fermenting microbes (yeast and LAB) could con-tribute to the increase or reduction of mineral content in fermented BBSB. This is because those minerals are consumed by fermenting microbes during growth and metabolism; however, these microbes could also degrade complex anti-nutritional factors in barely and thereby increase the release and extractability of minerals (Samtiya et al., 2021). Overall, the findings of this study revealed that incorporation of BM and DP into BBSB formulation may shorten the fermentation time and improve the nutritional quality of the product by enhancing the mineral contents of the final product.
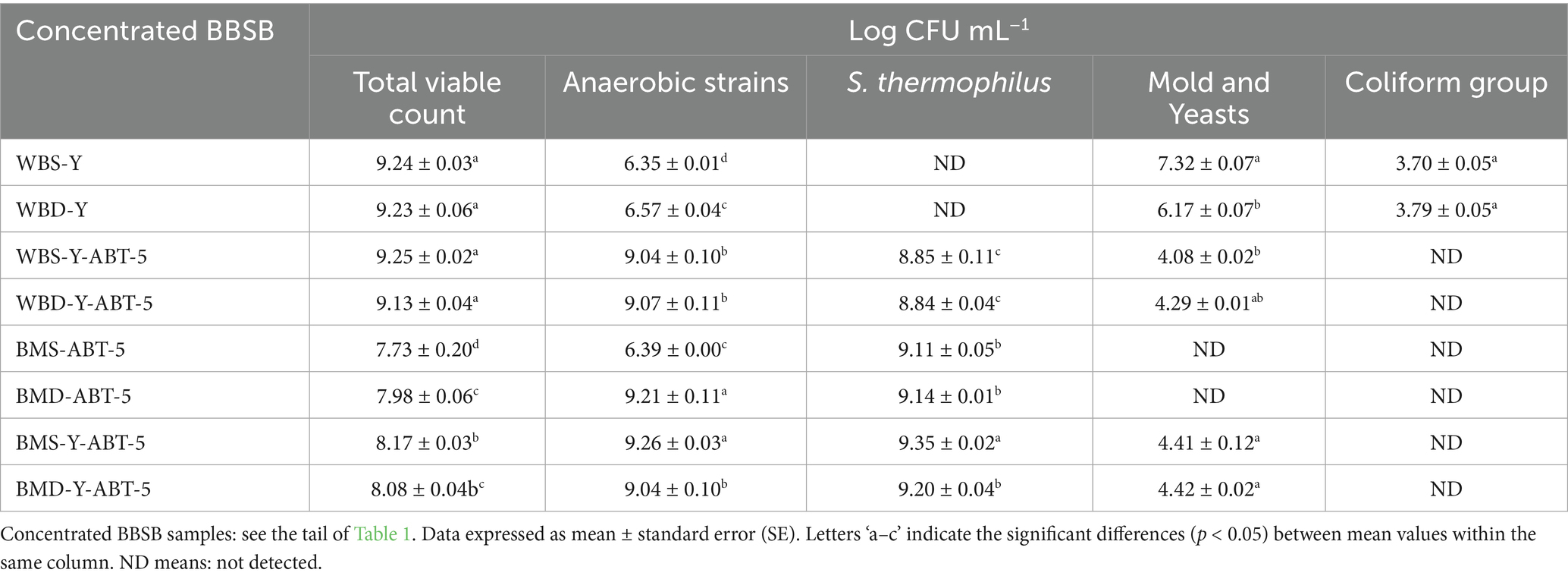
3.6 Viable microbial counts (log CFU mL−1) in concentrated BBSB enumerated on different media
The microbiological quality of different BBSB samples was examined, and the results are illustrated in Table 6. As a general observation, water-based beverages produced using the TM and without a proper heat treatment have shown the highest numbers of total bacterial counts (>9 CFU/mL), which is reflected by the presence of a high number of molds, yeasts and coliform bacteria. Similarly, water-based beverages, those produced using the IM, with a proper pasteurization treatment and added LAB starter culture, presented high bacterial load. However, those high numbers are reflected by the LAB culture added. Unlike BBSB traditional beverages, BBSBs produced using the IM had fewer counts of molds and yeasts (~ 2–3 log reduction) and no coliform numbers at all. Buttermilk-based BBSBs generally exhibited a lower number of total bacterial counts and a higher number of both anaerobic and S. thermophilus strain than those BBSB water-based beverages. Molds and yeasts were not detected in samples based on ABT-5 culture alone and coliforms were not detected in all BM-based beverages developed with IM. The anaerobic viable count varied significantly between the different treatments. The samples BMD-ABT-5 and BMS-Y-ABT-5 had the highest anaerobic viable count, which was significantly different from the other treatments, while the WBS-Y sample had the lowest anaerobic viable count. The concentrated BBSB formula BMS-Y-ABT-5 showed the highest count of S. thermophilus, while the samples WBS-Y-ABT-5 and WBD-Y-ABT-5 showed the lowest count of S. thermophilus. Only BMS-ABT-5 had the lowest total bacterial count. There were no significant differences in the total bacterial counts among WBS-Y, WBD-Y, WBS-Y-ABT-5, and WBD-Y-ABT-5, while BMS-ABT-5 showed the lowest total bacterial count. The highest content of molds and yeast was found in WBS-Y. Coliform bacteria were observed only in WBS-Y and WBD-Y. In recent times, probiotics have been found to contain beneficial bacteria that promote human health when used in probiotic beverages. It is important to consider specific strains based on their functional criteria and the environment in which they are used.
The quality of fermented beverages can be measured by the number of viable bacteria present in them. If microbial cell counts are between 7 and 8 log CFU/mL, then the product is said to have probiotic properties (Ewa and Małgorzata, 2020). In a recent investigation (El-Said, 2019), 50 coliforms (E. coli, Enterobacter cloacae, Klebsiella pneumoniae, Cronobacter sakazakii, Pseudomonas fluorescens, and Streptococcus parasanguinis) were detected in 10 Sobia samples purchased from vendors in Makkah city. The apparent higher count of coliforms can be a sign of potential faecal contamination due to poor sanitary conditions existed during the beverage’s manufacture. Studies have shown counts of coliforms to disappear after 24 h of storage. The World Health Organization published helpful guidelines concerning the safety of street food in 2015 to help prevent foodborne infections as a precaution to avoid negative health impacts, of which heat pasteurization has been proven to be a successful safety measure. In addition, yeast and lactic acid producing bacteria, mostly identified as Lactococcus spp. were detected. Yeast was the most predominant in the tested samples, followed by lactic acid producing bacteria, with the coliforms showing lower counts. More recently, probiotics contain beneficial bacteria that benefit human health when developing probiotic beverages. Specific strains should be considered based on functional criteria and the underlying environment. The number of viable bacteria is an important indicator to measure the quality of fermented beverages. In a previous study, it was found that Sobia, a popular beverage sold by street vendors and shops in Saudi Arabia, was contaminated with a significant number of microorganisms (Borai et al., 2022), They said that, although none of the identified bacteria or yeast species are known to cause food poisoning, their presence raises concerns about the risk of enteric infection. The acidity and microorganism levels in Sobia can vary depending on the storage conditions.
4 Conclusion
In conclusion, this study established a controlled method for producing concentrated BBSB while preserving and enhancing its nutritional, prebiotic, and probiotic properties. By utilizing the ABT-5 starter culture with different bases (water or buttermilk) and sweetening agents (sucrose or date powder), along with thermal treatment to ensure microbial safety and hygiene, the formulated beverages demonstrated improved total phenolic content, antioxidant activity, and functional potential. The incorporation of buttermilk and date powder, in particular, significantly enhanced bioactive compounds and probiotic viability, highlighting the synergistic effect of optimized fermentation and careful ingredient selection. These findings offer a promising framework for developing novel cereal-based probiotic beverages with enhanced health-promoting properties, addressing the growing demand for functional foods.
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: all data is included in the uploaded files.
Author contributions
RA: Conceptualization, Formal analysis, Methodology, Resources, Supervision, Writing – original draft, Writing – review & editing. AA: Formal analysis, Methodology, Project administration, Resources, Writing – original draft. HA: Formal analysis, Funding acquisition, Methodology, Resources, Writing – original draft. TB: Funding acquisition, Methodology, Software, Writing – original draft. KA: Funding acquisition, Methodology, Software, Writing – original draft. WA: Funding acquisition, Methodology, Software, Writing – original draft. IM: Data curation, Formal analysis, Methodology, Resources, Software, Writing – original draft. HB: Data curation, Formal analysis, Investigation, Methodology, Resources, Software, Writing – original draft. SS: Software, Writing – review & editing, Formal analysis, Investigation, Methodology, Resources, Supervision, Visualization, Writing – original draft.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Acknowledgments
The researchers would like to thank the Deanship of Graduate Studies and Scientific Research at Qassim University for financial support (QU-APC-2025).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Adebo, O., and Medina-Meza, I. G. (2020). Impact of fermentation on the phenolic compounds and antioxidant activity of whole cereal grains: a mini review. Molecules 25:927. doi: 10.3390/molecules25040927
Algonaiman, R., Alharbi, H. F., and Barakat, H. (2022). Antidiabetic and hypolipidemic efficiency of Lactobacillus plantarum fermented oat (Avena sativa) extract in streptozotocin-induced diabetes in rats. Fermentation 8:267. doi: 10.3390/fermentation8060267
Alharbi, Y. M., Sakr, S. S., Albarrak, S. M., Almundarij, T. I., Barakat, H., and Hassan, M. F. Y. (2022). Antioxidative, antidiabetic, and Hypolipidemic properties of probiotic-enriched fermented camel Milk combined with Salvia officinalis leaves Hydroalcoholic extract in Streptozotocin-induced diabetes in rats. Antioxidants 11:668. doi: 10.3390/antiox11040668
Aljutaily, T., Aladhadh, M., Alsaleem, K. A., Alharbi, H. F., Barakat, H., Aljumayi, H., et al. (2025). Gut microbiota diversity in obese rats treated with intermittent fasting, probiotic-fermented camel milk with or without dates and their combinations. Sci. Rep. 15:14204. doi: 10.1038/s41598-025-96893-9
Aljutaily, T., Barakat, H., Moustafa, M. M. A., and Rehan, M. (2022). Incorporation of Sukkari date in probiotic-enriched fermented camel Milk improves the nutritional, physicochemical, and Organoleptical characteristics. Fermentation 8:5. doi: 10.3390/fermentation8010005
Alminger, M., and Eklund-Jonsson, C. (2008). Whole-grain cereal products based on a high-fibre barley or oat genotype lower post-prandial glucose and insulin responses in healthy humans. Eur. J. Nutr. 47, 294–300. doi: 10.1007/s00394-008-0724-9
Alwohaibi, A. A. A., Ali, A. A., Sakr, S. S., Mohamed Ahmed, I. A., Alhomaid, R. M., Alsaleem, K. A., et al. (2023). Valorization of different dairy by-products to produce a functional dairy–millet beverage fermented with Lactobacillus paracasei as an adjunct culture. Fermentation 9:927. doi: 10.3390/fermentation9110927
Ando, A., and Nakamura, T. (2016). Prevention of GABA reduction during dough fermentation using a baker’s yeast dal81 mutant. J. Biosci. Bioeng. 122, 441–445. doi: 10.1016/j.jbiosc.2016.03.006
Aribas, M., Kahraman, K., and Koksel, H. (2020). In vitro glycemic index, bile acid binding capacity and mineral bioavailability of spaghetti supplemented with resistant starch type 4 and wheat bran. J. Funct. Foods 65:103778. doi: 10.1016/j.jff.2020.103778
Borai, A., Sebaih, S., Alshargi, A., Albarzan, F., Jaafari, R., Julaidan, L., et al. (2022). The effect of storage temperature and duration on the microbial growth in sobia; a Saudi Arabian traditional drink. J. Saudi Soc. Food Nutr. 15, 1–10.
Borai, A., Sebaih, S., Alshargi, A., Albarzan, F., Al-Ghamdi, A., Al-Ghamdi, S., et al. (2021). Ethanol content of a traditional Saudi beverage Sobia. Int. J. Food Prop. 24, 1790–1798. doi: 10.1080/10942912.2021.2005089
Boskov Hansen, H., Andreasen, M., Nielsen, M., Larsen, L., Knudsen, B. K., Meyer, A., et al. (2002). Changes in dietary fibre, phenolic acids and activity of endogenous enzymes during rye bread-making. Eur. Food Res. Technol. 214, 33–42. doi: 10.1007/s00217-001-0417-6
Chen, L., Wu, D., Schlundt, J., and Conway, P. L. (2020). Development of a dairy-free fermented oat-based beverage with enhanced probiotic and bioactive properties. Front. Microbiol. 11:609734. doi: 10.3389/fmicb.2020.609734
De Barber, C. B., Prieto, J., and Collar, C. (1989). Reversed-phase high-performance liquid chromatography analysis of changes in free amino acids during wheat bread dough fermentation. Cereal Chem. 66, 283–288.
de Matos Reis, S., Mendes, G. R. L., Mesquita, B. M. A. C., Lima, W. J. N., Pinheiro, C. A. F. D., Ruas, F. A. O., et al. (2021). Development of milk drink with whey fermented and acceptability by children and adolescents. J. Food Sci. Technol. 58, 2847–2852. doi: 10.1007/s13197-021-05003-w
Dhakal, R., Bajpai, V. K., and Baek, K. H. (2012). Production of gaba (γ - aminobutyric acid) by microorganisms: a review. Braz. J. Microbiol. 43, 1230–1241. doi: 10.1590/s1517-83822012000400001
Đorđević, T. M., Šiler-Marinković, S. S., and Dimitrijević-Branković, S. I. (2010). Effect of fermentation on antioxidant properties of some cereals and pseudo cereals. Food Chem. 119, 957–963. doi: 10.1016/j.foodchem.2009.07.049
El-Said, H. (2019). Popular fermented beverage (Sobia) as a potential risk factor for aquiring MDR infection. Asian J. Microbiol. Biotechnol. Environ. Sci. 21, 532–536.
Enujiugha, V. N., and Badejo, A. A. (2017). Probiotic potentials of cereal-based beverages. Crit. Rev. Food Sci. Nutr. 57, 790–804. doi: 10.1080/10408398.2014.930018
Ewa, K., and Małgorzata, Z. (2020). “The possibility of obtaining buckwheat beverages fermented with lactic acid Bacteria and Bifidobacteria” in Milk Substitutes. ed. Z. Małgorzata (Rijeka: IntechOpen).
Feng, X. M., Larsen, T. O., and Schnürer, J. (2007). Production of volatile compounds by Rhizopus oligosporus during soybean and barley tempeh fermentation. Int. J. Food Microbiol. 113, 133–141. doi: 10.1016/j.ijfoodmicro.2006.06.025
Garrido-Galand, S., Asensio-Grau, A., Calvo-Lerma, J., Heredia, A., and Andrés, A. (2021). The potential of fermentation on nutritional and technological improvement of cereal and legume flours: a review. Food Res. Int. 145:110398. doi: 10.1016/j.foodres.2021.110398
Gassem, M. A. A. (2002). A microbiological study of Sobia: a fermented beverage in the Western province of Saudi Arabia. World J. Microbiol. Biotechnol. 18, 173–177. doi: 10.1023/A:1014916702466
Gassem, M. A. A. (2003). Physico-chemical properties of sobia: a traditional fermented beverage in western province of Saudi Arabia. Ecol. Food Nutr. 42, 25–35. doi: 10.1080/03670240303115
Ghafoor, K., Sarker, M. Z. I., Al-Juhaimi, F. Y., Mohamed Ahmed, I. A., Babiker, E. E., Alkaltham, M. S., et al. (2023). Bioactive compounds extracted from Saudi dates using green methods and utilization of these extracts in functional yogurt. Foods 12:847. doi: 10.3390/foods12040847
Hamed, H., Chaari, F., Ghannoudi, Z., ElFeki, A., Ellouz, S. C., and Gargouri, A. (2018a). Beneficial effects of fermented camel milk by lactococcus lactis subsp cremoris on cardiotoxicity induced by carbon tetrachloride in mice. Biomed. Pharmacother. 97, 107–114. doi: 10.1016/j.biopha.2017.10.057
Hamed, H., Gargouri, M., Boulila, S., Chaari, F., Ghrab, F., Kallel, R., et al. (2018b). Fermented camel milk prevents carbon tetrachloride induced acute injury in kidney of mice. J. Dairy Res. 85, 251–256. doi: 10.1017/s0022029918000250
He, Y., Liu, G., Xia, C., Chen, J., Zhao, J., Li, X., et al. (2022). Laxative effect of mulberry ferment on two models of constipated mice. J. Funct. Foods 90:104971. doi: 10.1016/j.jff.2022.104971
Hole, A. S., Rud, I., Grimmer, S., Sigl, S., Narvhus, J., and Sahlstrøm, S. (2012). Improved bioavailability of dietary phenolic acids in whole grain barley and oat groat following fermentation with probiotic Lactobacillus acidophilus, Lactobacillus johnsonii, and Lactobacillus reuteri. J. Agric. Food Chem. 60, 6369–6375. doi: 10.1021/jf300410h
Hussin, F. S., Chay, S. Y., Hussin, A. S. M., Wan Ibadullah, W. Z., Muhialdin, B. J., Abd Ghani, M. S., et al. (2021). GABA enhancement by simple carbohydrates in yoghurt fermented using novel, self-cloned Lactobacillus plantarum Taj-Apis362 and metabolomics profiling. Sci. Rep. 11:9417. doi: 10.1038/s41598-021-88436-9
Ignat, M., Salanță, L., Pop, O., Pop, C., Tofană, M., Mudura, E., et al. (2020). Current functionality and potential improvements of non-alcoholic fermented cereal beverages. Foods 9:31. doi: 10.3390/foods9081031
Ilango, S., and Antony, U. (2021). Probiotic microorganisms from non-dairy traditional fermented foods. Trends Food Sci. Technol. 118, 617–638. doi: 10.1016/j.tifs.2021.05.034
Infusino, F., Marazzato, M., Mancone, M., Fedele, F., Mastroianni, C. M., Severino, P., et al. (2020). Diet supplementation, probiotics, and nutraceuticals in SARS-CoV-2 infection: a scoping review. Nutrients 12:718. doi: 10.3390/nu12061718
Katina, K., Liukkonen, K.-H., Kaukovirta-Norja, A., Adlercreutz, H., Heinonen, S.-M., Lampi, A.-M., et al. (2007). Fermentation-induced changes in the nutritional value of native or germinated rye. J. Cereal Sci. 46, 348–355. doi: 10.1016/j.jcs.2007.07.006
Kobyliak, N., Falalyeyeva, T., Boyko, N., Tsyryuk, O., Beregova, T., and Ostapchenko, L. (2018). Probiotics and nutraceuticals as a new frontier in obesity prevention and management. Diabetes Res. Clin. Pract. 141, 190–199. doi: 10.1016/j.diabres.2018.05.005
Li, X., Dai, L., Liu, H., Liu, W., Pan, B., Wang, X., et al. (2020). Molecular mechanisms of furanone production through the EMP and PP pathways in Zygosaccharomyces rouxii with D-fructose addition. Food Res. Int. 133:109137. doi: 10.1016/j.foodres.2020.109137
Lu, J., Zhao, H., Chen, J., Fan, W., Dong, J., Kong, W., et al. (2007). Evolution of phenolic compounds and antioxidant activity during malting. J. Agric. Food Chem. 55, 10994–11001. doi: 10.1021/jf0722710
Maier, W., Peipp, H., Schmidt, J., Wray, V., and Strack, D. (1995). Levels of a terpenoid glycoside (blumenin) and cell wall-bound phenolics in some cereal mycorrhizas. Plant Physiol. 109, 465–470. doi: 10.1104/pp.109.2.465
Melgarejo, P., Calín-Sánchez, Á., Vázquez-Araújo, L., Hernández, F., Martínez, J. J., Legua, P., et al. (2011). Volatile composition of pomegranates from 9 Spanish cultivars using headspace solid phase microextraction. J. Food Sci. 76, S114–S120. doi: 10.1111/j.1750-3841.2010.01945.x
Meybodi, N. M., Nasab, S. S., Khorshidian, N., and Mortazavian, A. M. (2021). “Chapter 7- probiotic beverages: health benefits and current trends in the Middle East” in Probiotic beverages. eds. S. K. Panda, J. Kellershohn, and I. Russell (London, United Kingdom: Academic Press), 99–126.
Michlmayr, H., Nauer, S., Brandes, W., Schümann, C., Kulbe, K. D., del Hierro, A. M., et al. (2012). Release of wine monoterpenes from natural precursors by glycosidases from Oenococcus oeni. Food Chem. 135, 80–87. doi: 10.1016/j.foodchem.2012.04.099
Milani, R. F., Morgano, M. A., and Cadore, S. (2018). A simple and reliable method to determine 16 trace elements by ICP OES in ready to drink beverages. Food Anal. Methods 11, 1763–1772. doi: 10.1007/s12161-018-1172-7
Mohd Ali, N., Mohd Yusof, H., Long, K., Yeap, S. K., Ho, W. Y., Beh, B. K., et al. (2013). Antioxidant and hepatoprotective effect of aqueous extract of germinated and fermented mung bean on ethanol-mediated liver damage. Biomed. Res. Int. 2013:693613. doi: 10.1155/2013/693613
Moore, J., Cheng, Z., Hao, J., Guo, G., Liu, J.-G., Lin, C., et al. (2007). Effects of solid-state yeast treatment on the antioxidant properties and protein and fiber compositions of common hard wheat bran. J. Agric. Food Chem. 55, 10173–10182. doi: 10.1021/jf071590o
Mubarak, A., Hodgson, J. M., Considine, M. J., Croft, K. D., and Matthews, V. B. (2013). Supplementation of a high-fat diet with chlorogenic acid is associated with insulin resistance and hepatic lipid accumulation in mice. J. Agric. Food Chem. 61, 4371–4378. doi: 10.1021/jf400920x
Nazhand, A., Souto, E. B., Lucarini, M., Souto, S. B., Durazzo, A., and Santini, A. (2020). Ready to use therapeutical beverages: focus on functional beverages containing probiotics, prebiotics and synbiotics. Beverages 6:26. doi: 10.3390/beverages6020026
Nsimba, R. Y., Kikuzaki, H., and Konishi, Y. (2008). Antioxidant activity of various extracts and fractions of Chenopodium quinoa and Amaranthus spp. seeds. Food Chem. 106, 760–766. doi: 10.1016/j.foodchem.2007.06.004
Perpetuini, G., Tittarelli, F., Battistelli, N., Suzzi, G., and Tofalo, R. (2020). Γ-Aminobutyric acid production by Kluyveromyces marxianus strains. J. Appl. Microbiol. 129, 1609–1619. doi: 10.1111/jam.14736
Phiri, S., Schoustra, S., Van Den Heuvel, J., Smid, E., Shindano, J., and Linnemann, A. (2019). Fermented cereal-based Munkoyo beverage: processing practices, microbial diversity and aroma compounds. PLoS One 14:e0223501. doi: 10.1371/journal.pone.0223501
Pswarayi, F., and Gänzle, M. (2022). African cereal fermentations: a review on fermentation processes and microbial composition of non-alcoholic fermented cereal foods and beverages. Int. J. Food Microbiol. 378:109815. doi: 10.1016/j.ijfoodmicro.2022.109815
Raungrusmee, S., Kumar, S. R., and Anal, A. K. (2022). “Probiotic Cereal-based Food and Beverages, their Production and Health Benefits” in Probiotics, Prebiotics and Synbiotics: Technological advancements towards safety and industrial applications. (New Jersey, USA: John Wiley & Sons Ltd). 186–212.
Rizal, S., Kustyawati, M. E., and Hasanudin, U. (2021). The growth of yeast and fungi, the formation of β-glucan, and the antibacterial activities during soybean fermentation in producing tempeh. Int. J. Food Sci. 2021:6676042. doi: 10.1155/2021/6676042
Roberfroid, M. B. (2002). Functional foods: concepts and application to inulin and oligofructose. Br. J. Nutr. 87, S139–S143. doi: 10.1079/bjnbjn/2002529
Sahab, N. R., Subroto, E., Balia, R. L., and Utama, G. L. (2020). γ-Aminobutyric acid found in fermented foods and beverages: current trends. Heliyon 6:526. doi: 10.1016/j.heliyon.2020.e05526
Salmerón, I. (2017). Fermented cereal beverages: from probiotic, prebiotic and synbiotic towards nanoscience designed healthy drinks. Lett. Appl. Microbiol. 65, 114–124. doi: 10.1111/lam.12740
Samtiya, M., Aluko, R. E., Puniya, A. K., and Dhewa, T. (2021). Enhancing micronutrients bioavailability through fermentation of plant-based foods: a concise review. Fermentation 7:63. doi: 10.3390/fermentation7020063
Şanlier, N., Gökcen, B. B., and Sezgin, A. C. (2019). Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 59, 506–527. doi: 10.1080/10408398.2017.1383355
Schwan, R. F., and Ramos, C. L. (2019). “10 - Functional Beverages from Cereals” in Functional and Medicinal Beverages. eds. A. M. Grumezescu and A. M. Holban (London, United Kingdom: Academic Press), 351–379.
Shan, Y., Lai, Y., and Yan, A. (2012). Metabolic reprogramming under microaerobic and anaerobic conditions in bacteria. eds. X. Wang, J. Chen, and P. Quinn Reprogram. Microb. Metab. Pathways, (Dordrecht, Netherlands: Springer) 159–179. doi: 10.1007/978-94-007-5055-5_8
Sharma, M., Wasan, A., and Sharma, R. K. (2021). Recent developments in probiotics: An emphasis on Bifidobacterium. Food Biosci. 41, 100993. doi: 10.1016/j.fbio.2021.100993
Soltani, N., Qiu, H., Aleksic, M., Glinka, Y., Zhao, F., Liu, R., et al. (2011). GABA exerts protective and regenerative effects on islet beta cells and reverses diabetes. Proc. Natl. Acad. Sci. USA 108, 11692–11697. doi: 10.1073/pnas.1102715108
Steel, R. G. D., Torrie, J. H., and Dickey, D. A. (1997). Principles and procedures of statistics: A biometrical approach. New York. USA: McGraw-Hill.
Tambe, E. A., and Gotmare, S. (2019). Chemical characterization of three cardamom oils (ELETTARIACARDAMOMUM)BYGCMS. World J. Pharmacy Pharm. Sci. 8, 876–885. doi: 10.20959/wjpps201911-14879
Tiwari, U., and Cummins, E. (2009). Factors influencing β-glucan levels and molecular weight in cereal-based products. Cereal Chem. 86, 290–301. doi: 10.1094/CCHEM-86-3-0290
Tsolmonbaatar, A., Hashida, K., Sugimoto, Y., Watanabe, D., Furukawa, S., and Takagi, H. (2016). Isolation of baker’s yeast mutants with proline accumulation that showed enhanced tolerance to baking-associated stresses. Int. J. Food Microbiol. 238, 233–240. doi: 10.1016/j.ijfoodmicro.2016.09.015
Turkmen, N., Akal, C., and Özer, B. (2019). Probiotic dairy-based beverages: a review. J. Funct. Foods 53, 62–75. doi: 10.1016/j.jff.2018.12.004
Vayalil, P. K. (2012). Date fruits (Phoenix dactylifera Linn): an emerging medicinal food. Crit. Rev. Food Sci. Nutr. 52, 249–271. doi: 10.1080/10408398.2010.499824
Waters, D., Mauch, A., Coffey, A., Arendt, E., and Zannini, E. (2015). Lactic acid Bacteria as a cell factory for the delivery of functional biomolecules and ingredients in cereal-based beverages: a review. Crit. Rev. Food Sci. Nutr. 55, 503–520. doi: 10.1080/10408398.2012.660251
Yan, X., McClements, D., Luo, S., Ye, J., and Liu, C. (2024). A review of the effects of fermentation on the structure, properties, and application of cereal starch in foods. Crit. Rev. Food Sci. Nutr. 20:269. doi: 10.1080/10408398.2024.2334269
Yuwa-Amornpitak, T., Butkhup, L., and Yeunyaw, P.-N. (2020). Amino acids and antioxidant activities of extracts from wild edible mushrooms from a community forest in the Nasrinual District, Maha Sarakham. Thailand. Food Sci. Technol. 40, 712–720. doi: 10.1590/fst.18519
Zhang, C.-R., Aldosari, S. A., Vidyasagar, P., Shukla, P., and Nair, M. G. (2015). Determination of the variability of sugars in date fruit varieties. J. Plant. Crop. 43, 53–61.
Zhang, J., Wang, P., Tan, C., Zhao, Y., Zhu, Y., Bai, J., et al. (2022). Effects of L. plantarum dy-1 fermentation time on the characteristic structure and antioxidant activity of barley β-glucan in vitro. Curr. Res. Food Sci. 5, 125–130.
Keywords: sustainable food, barley beverage, buttermilk, probiotics, glycemic index, antioxidant activity, γ-aminobutyric acid, β-glucan
Citation: Algheshairy RM, Ali AA, Alharbi HF, Bushnaq T, Alsaleem KA, Alsanei WA, Mohamed Ahmed IA, Barakat H and Sakr SS (2025) The nutritional and bioactive properties of probiotic-enriched barley-buttermilk beverages. Front. Sustain. Food Syst. 9:1646160. doi: 10.3389/fsufs.2025.1646160
Edited by:
Malgorzata Ziarno, Warsaw University of Life Sciences, PolandReviewed by:
Shiqi Li, Northwest A&F University, ChinaZeinab Mohamed, Animal Production Research Institute (APRI), Egypt
Copyright © 2025 Algheshairy, Ali, Alharbi, Bushnaq, Alsaleem, Alsanei, Mohamed Ahmed, Barakat and Sakr. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sally S. Sakr, cy5zYWtyQHF1LmVkdS5zYQ==
 Reham M. Algheshairy
Reham M. Algheshairy Asmahan A. Ali
Asmahan A. Ali Hend F. Alharbi
Hend F. Alharbi Taqwa Bushnaq
Taqwa Bushnaq Khalid A. Alsaleem1
Khalid A. Alsaleem1 Woroud A. Alsanei
Woroud A. Alsanei Isam A. Mohamed Ahmed
Isam A. Mohamed Ahmed Hassan Barakat
Hassan Barakat Sally S. Sakr
Sally S. Sakr