Abstract
The variation in animal coloration patterns has evolved in response to different visual strategies for reducing the risk of predation. However, the perception of animal coloration by enemies is affected by a variety of factors, including morphology and habitat. We use the diversity of Australian chrysomeline leaf beetles to explore relationships of visual ecology to beetle morphology and color patterns. There is impressive color pattern variation within the Chrysomelinae, which is likely to reflect anti-predatory strategies. Our phylogenetic comparative analyses reveal strong selection for beetles to be less distinct from their host plants, suggesting that the beetle color patterns have a camouflage effect, rather than the widely assumed aposematic function. Beetles in dark habitats were significantly larger than beetles in bright habitats, potentially to avoid detection by predators because it is harder for large animals to be cryptic in bright habitats. Polyphagous species have greater brightness contrast against their host plants than monophagous species, highlighting the conflict between a generalist foraging strategy and the detection costs of potential predators. Host plant taxa—Eucalyptus and Acacia—interacted differently with beetle shape to predict blue pattern differences between beetle and host plant, possibly an outcome of different predator complexes on these host plants. The variety of anti-predator strategies in chrysomelines may explain their successful radiation into a variety of habitats and, ultimately, their speciation.
Introduction
The pervasive risk of attack by natural enemies has favored the evolution of a variety of anti-predator strategies (Stevens, 2013). Protective color patterns are arguably among the most widespread of these strategies, as they can reduce prey detection and/or warn predators that the prey is unpalatable (Cott, 1940; Ruxton et al., 2004; Stevens, 2007). Color patterns that resemble an animal's background or mask an animal's outline may reduce detection by predators (Endler, 1984; Cuthill et al., 2005; Stevens et al., 2006). On the other hand, conspicuous color patterns are used to advertise the distastefulness of an organism to visually searching enemies (Roper, 1990; Prudic et al., 2007b; Skelhorn et al., 2016). However, these functions of color patterns may not be mutually exclusive, as some color patterns may combine warning coloration at a close range, with crypsis at a longer range (Tullberg et al., 2005; Honma et al., 2015). As a consequence, a diversity of color patterns may derive from these different visual defense mechanisms. While the adaptive significance of these color patterns are reasonably well documented, how their evolution has been shaped by ecological (Prudic et al., 2007a; Mappes et al., 2014) and life history factors (Ojala et al., 2007; Tan et al., 2016) is less clearly understood.
Animal color patterns are rarely “matched” with their background, because habitats are usually heterogeneous, and therefore provide backgrounds that vary visually in time and space (Merilaita and Dimitrova, 2014). Nevertheless, background matching is especially important for individuals that are exposed while feeding (Pellissier et al., 2011; Kjernsmo and Merilaita, 2012), since the risk of predation can increase by up to a 100-fold (Bernays, 1997). One solution is for animals to have patches on the body surface with a mixture of contrasting colors or luminance that match different patches in the immediate adjacent surroundings (Pellissier et al., 2011; Wilts et al., 2012; Espinosa and Cuthill, 2014). For herbivorous animals, apart from being a food source, the host plant can also affect how animals are perceived by their enemies. For instance, host plants can provide visual signals to deter a herbivore's predators (Keasar et al., 2013). While most species of phytophagous insects are associated with a small range of closely related host plant species, other species may have multiple associations with unrelated plants (Novotny et al., 2002, 2007; Jurado-Rivera et al., 2009; Baker et al., 2012). Increasing the number of host plants is likely to decrease background matching, and so generalist species may face a compromise between a preferred host plant and their capacity to be conspicuous or not.
Visual signals may be constrained by morphology, which is also under selection by natural enemies. Larger animals may be under greater selection pressure because of their higher profitability and detectability (Krebs et al., 1977; Utne-Palm, 2000; Sandre et al., 2007; Smith et al., 2016). Thus, the detection of an animal can be affected by a combination of body size and color pattern (Karpestam et al., 2014). The size of an animal can also affect the efficacy of color patterns, and thus the strength of selection. A positive correlation between warning efficacy and animal body size suggests that warning coloration may evolve more readily in larger animals (Gamberale and Tullberg, 1998), but larger insects may not necessarily be more conspicuous (Nilsson and Forsman, 2003). For instance, larger-sized signaling pattern elements can accelerate the learning rate of great tits (Parus major) to unpalatable prey (Lindström et al., 1999). Larger-sized prey can also induce greater avoidance behavior in potential predators (Gamberale and Tullberg, 1996b, 1998; Forsman and Merilaita, 1999; Lindstedt et al., 2008). Other studies reveal the advantages of gregarious aposematic prey (Gamberale and Tullberg, 1996a, 1998; Tan et al., 2016), which suggest that the efficacy of the warning signal may be an effect of the total signaling area.
In addition to size, the shape of an animal affects how it is perceived. An important, but frequently neglected consideration is that the shape of prey will differ according to the perspective of the viewer. For instance, aerial predators such as, birds or predatory hymenoptera will take a predominantly dorsal view of the prey, while terrestrial predators such as carnivorous insects and spiders will assess prey size from a lateral view. The effect of predation on prey shape is highly variable among animal taxa (Brönmark and Miner, 1992; Nilsson et al., 1995; Outomuro and Johansson, 2015), potentially affecting the mobility and thus the ability to escape predators. For instance, damselflies develop short and broad hind wings in response to selection by bird predators (Outomuro and Johansson, 2015) while freshwater carp develop deeper bodies in the presence of pike predators, which either render the carp less vulnerable to predation (Brönmark and Miner, 1992), or increase handling time and thus the opportunity to escape (Nilsson et al., 1995). The distribution of color intensities on a convex shape, like a beetle, would vary in a natural landscape because of multiple illumination sources, such as light scattering from forest gaps or surrounding objects (Khang et al., 2006). Studies of humans, pigeons and starlings show that the perceived shape and depth of an object is affected by the amount of illumination on it (Ramachandran, 1988; Cook et al., 2012; Qadri et al., 2014). As the shape and depth of an organism may be correlated with its nutritional return as a prey item, the shape and depth may also reflect an animal's value to a predator (Stephens and Krebs, 1986; Rychlik, 1999).
Chrysomeline leaf beetles are a good system for investigating the evolution of color patterns because of their phenotypic diversity and the variety of habitats in which they occur (Reid, 2014). Australia includes 25% of the world's diversity of this subfamily: ~750 species in 43 endemic genera (Reid et al., 2009; Reid, 2017). Leaf beetles cover a range of color patterns such as iridescence, contrasting, and mottled coloration (see Reid, 2006 for details on the diversity of the color patterns of Australian chrysomelines). Chrysomeline beetles are also chemically defended, producing noxious secretions of metabolites derived from their host plants (Pasteels et al., 1982, 1983; Selman, 1985a; Schulz et al., 1997; Termonia et al., 2002), or synthesized de novo (Blum et al., 1972; Hilker and Schulz, 1994). The conspicuous appearance of many chrysomelines, together with their defensive chemical secretions and diurnal lifestyle, have led to the widespread assumption that these color patterns are aposematic and deter potential predators (Selman, 1985b, 1994; Pasteels et al., 1989; Matthews and Reid, 2002).
However, attributing an aposematic role to contrasting color patterns commonly associated with toxicity may not accurately reflect the adaptive significance of the beetle color patterns (Pawlik et al., 1995). Chrysomelines are phytophagous, with diverse body shapes and color patterns that, to varying degrees, match or contrast with their environments that range from rainforests to sandy deserts (Reid, 2014). Most chrysomelines are highly specialized on a single host plant genus for their entire life cycle (Jolivet and Hawkeswood, 1995; Reid, 2006; Jurado-Rivera et al., 2009). Indeed, recent analyses suggest that chemical defenses in juvenile chrysomelines is closely linked to the chemistry of their hosts (Rahfeld et al., 2015). Despite the presence of toxic glands in the chrysomelines, the diversity of chrysomeline color patterns suggests that these color patterns may have evolved through mechanisms other than aposematism. Leaf beetles are likely to have a large number of visual predators that will view the beetles from a dorsal and a lateral view, thus exerting selection pressures on chrysomeline size and shape, respectively. Cursorial arachnids are potential predators exerting a selection pressure on chrysomelines from lateral view, while aerial predators such as birds and wasps are potential predators of beetles taking a dorsal view (Recher et al., 1987; Recher, 1989; Recher and Majer, 2006). Documented predators of chrysomelines include wasps (Evans and Hook, 1986) and birds (de Little, 1979; Matthews and Reid, 2002).
We investigate how beetle morphology, habitat and host plant range influence the evolution of diverse color patterns within a single clade of chrysomeline beetles from Australia. We assemble a database of chrysomeline color pattern and morphology by collecting live beetles and drawing on published records, and perform phylogenetic comparative analyses to ask specific questions. First, are beetles visually different from their host plants, and is this distinctiveness predicted by the host plant specificity and light environment. We predict that polyphagous species are visually different from their host plants, because of the inherent differences of host plant species. We also predict that beetles that feed on similar host plant species may have broadly similar color patterns. For instance, beetles that feed on one clade of host plants may have specific color pattern differences that are not evident among beetles that feed on a different clade. Next, we asked if beetle morphology affects how beetles are perceived: we predict that larger beetles may be more commonly found in darker habitats than smaller beetles because larger beetles, which face higher predation risks, may be less conspicous in these habitats. We also predict that more convex beetles that may appear larger to predators, will be visually more distinct from their host plants, thereby providing a greater deterence signal.
Materials and methods
Taxon sampling
Deceased non-iridescent chrysomeline leaf beetles tend to fade to brown (Moore, 1980; Selman, 1985b; Balsbaugh, 1988; Lawrence and Britton, 1994; also see Figure 1), so we included live beetles only as a meaningful record of body color patterns. Museum collections of specimens, while plentiful, furnish little, if any, information about color patterns unless the beetles possess structural coloration.
Figure 1

Live and pinned specimens of chrysomelines. (A) Live specimen of an adult Paropsisterna gloriosa; (B) Corresponding pinned specimen of Paropsisterna gloriosa (Image by Sue Lindsay, © Australian Museum); (C) Live specimen of an adult Calomela pallida; (D) Corresponding pinned specimen of Calomela pallida (Image by Sue Lindsay, © Australian Museum). The white bars represent 5 mm in each image.
Fifty-one species of chrysomeline beetles were collected from 32 locations across four states in Australia from November 2012 to March 2014 (Table S1). Where possible, we collected species representative of the diversity of phenotypes in each genus. We collected an additional 39 species of beetles during our field trips, but they were not represented in the molecular phylogeny (Jurado-Rivera (2014), and so were not included in this analysis. Our study comprises of 143 specimens collected from a range of habitats—rainforests, dry, and wet sclerophyll forests and heath. Specimens have been deposited at the Australian Museum, Sydney. Live beetles were photographed with their host plant in the field at a distance of ~0.1 m during daylight hours, generally under clear skies. We obtained digital images of the dorsal surface of each beetle, as it was assumed to be the clearest exposition of its appearance to visual predators. These beetles are all dorsally convex, and predators are assumed to view them dorsally or laterodorsally as only the dorsal surface is exposed during foraging, mating, and resting.
Phylogenetic tree inference
We obtained a molecular phylogeny for Australian chrysomelines, previously documented in an unpublished Ph.D. Thesis (Jurado-Rivera, 2014). Details of the molecular phylogeny construction are described in the Supplementary Material (Chrysomeline molecular phylogeny; Table S2). The phylogenetic tree was visualized and modified in Mesquite 3.02 (Maddison and Maddison, 2014). The original phylogeny by Jurado-Rivera (2014) was pruned to include only the species in our analysis. The node ages of the tree were estimated using a semi-parametric method based on penalized likelihood (Paradis et al., 2004) and the smoothing parameter used was 0.1. All subsequent analyses were performed in R Development Core Team (2016).
Beetle morphology
We investigated whether adult morphology constrains the color patterns of chrysomelines by obtaining measures of the length, height, and width of each species in the sample. Additional specimens were measured from the collection at the Australian Museum, Sydney. Where available, up to 10 individuals were measured per species, over as wide a range of collection years as possible. The length, width and height dimensions of beetles are defined in Figure 2, and 470 beetles were measured for this purpose (see details in Table S3). To obtain a measure of a beetle's elevation from its substrate—and thus its perceived size by natural enemies approaching from the same substrate—we obtained the ratio of the beetle height to the length (hereafter referred to as convexity). Beetle height was measured as the furthest extremes of the anatomy—i.e., if the elytra were not enlarged and overhanging (see Figure 2A), then the maximum projection of the metaventrite was considered the deepest point.
Figure 2

Measurements of the dimensions of a beetle. (A) Lateral view of beetle depicting height measurement; (B) Dorsal view of beetle depicting length and width measurements. Convexity is defined as the ratio of the beetle height to length. Images by Kindi Smith, © Australian Museum.
Assessment of color pattern difference
Raw image files (CR2 format) with minimally processed data from the image sensor of the camera (Canon EOS 600D) were acquired to prevent information loss during the image compression process (Stevens et al., 2007). Due to limitations of the camera and lens, UV coloration of the beetles and their backgrounds were not recorded. Each photograph was taken in the presence of a card with 1 cm graduations to allow for subsequent scaling of images. All the images were then scaled in Adobe Photoshop CS6 based on the graduations present in each image, such that the number of pixels per cm was consistent for all the images. The beetle in each image was manually defined following Cheney et al. (2014) using a tablet and stylus. Four samples of the background were also obtained by shifting the beetle outline to separate, arbitrarily chosen positions within the background area of the same image.
First, we compared the brightness contrasts of the beetles to their background samples, by measuring the distribution of brightness contrast within each image. Contrasts between prey and background can be important in perception and predator avoidance learning (Gittleman and Harvey, 1980; Roper and Wistow, 1986; Roper and Redston, 1987; Aronsson and Gamberale-Stille, 2009; Llandres et al., 2011). The pixel intensity variance (PIV) is a measure of the distribution of brightness contrast within an image. In order to compare the brightness contrasts of the beetles to their background samples, we measured the PIV of each beetle and background sample as:
where for each sample (beetle or background), N is the length of the data vector, is the mean of the data vector, and xi is element i of the data vector (Zylinski et al., 2011). This was calculated separately on the red, green and blue layers for each sample in MATLAB, using the Image Processing Toolbox. To compare the beetle samples with their respective background samples, we performed two-tailed t-tests of the PIV of beetles against their respective backgrounds. Where the results of the t-test indicated a significant difference between the PIV of the beetle and its background, the beetle was deemed different from its background in terms of brightness contrast in that particular color layer (for convenience, we refer to the brightness contrasts within the specific color layers as either red, green or blue contrasts).
Next, we quantified and compared the color patterns of the beetles and their backgrounds, by obtaining measures of color pattern differences. This allowed comparison of the red, green, and blue patterns of the beetles and their backgrounds. The underlying assumption of this analysis is that a color pattern can be considered cryptic if it resembles a random sample of its background, while it must differ from its background in order to be conspicuous (Endler, 1978). This analysis does not take into account the vision of the viewer, but it nonetheless yields useful empirical information about the differences of both color and pattern between an animal and its background, and allows us to investigate the evolutionary development of color patterns in these toxin-protected beetles. The degree of resemblance between beetle and visual background allows us to examine the presence of any selection pressure because of the visual background signals.
Following Zylinski et al. (2011), the spatial frequency of patterns within each sample (beetle and background) were analyzed using two-dimensional Fast Fourier Transformation (FFT). This was performed separately on the red, green and blue layers of each sample in MATLAB, using the Image Processing Toolbox. A log-scaled power spectrum curve was obtained from the FFT, followed by a rotational average of the amplitudes produced (Cheney et al., 2014). The absolute difference in area between the power spectrum curves of the beetle and each background sample provided a quantification of the difference in color pattern between the beetle and its background. This difference in color pattern between the beetle and its background was obtained separately for red, blue and green (hereafter referred to as red, blue and green pattern differences, respectively). As the values for pattern differences were of a greater range than that described by Cheney et al. (2014), we did not classify them as “different” or “not different.” Rather, we used the values of pattern differences as a continuous measure to compare against the ecological data. These analyses cannot be used to compare patterns between images because of edge effects from the border between the beetle outline and the edge of the image (Cheney et al., 2014). Although image intensity variance and power spectrum analysis can be affected by changes in the light exposure, within-image comparisons of the beetle and the backgrounds would control for such effects.
Variation in color differences between beetle and host plant
To summarize the variation in color differences between beetle and host plant, we performed a phylogenetic Principle Components Analysis using the phytools package (Revell, 2012) in R. This procedure takes into account the phylogenetic non-independence among species means.
Ecological data collection
Ecological data were collated for the 51 species of chrysomeline beetles from a range of habitats. Ecological data of the 51 species were based on conditions in the field and, where necessary, supplemented by information from the literature (details in Table S3). Chrysomeline beetles are herbivores that usually only feed on related host plants within a genus (Jurado-Rivera et al., 2009; Reid, 2017), and host plant identities were used to examine if selection pressures on color patterns were specific to the host plants.
Phylogenetic generalized least squares regression
We used phylogenetic generalized least-squares (PGLS) regression, which controls for the non-independence of species data, to examine the relationship of response variable (color pattern differences) with predictor variables (beetle morphology, host plants, and light environment). For continuous variables (beetle length, height, convexity, color pattern difference), we calculated Pagel's λ (Pagel, 1997, 1999), which indicates the level of association between the trait and phylogeny. λ adopts a value between 0 and 1, where λ = 0 indicates that closely related species do not have similar trait values, and λ = 1 indicates that closely related species have very similar trait values.
We use the caper package (Orme et al., 2013) in R to perform PGLS. We used a model selection approach using Akaike's Information Criterion (AIC). To evaluate how much more likely each of our models were than the null model, we calculated the Evidence Ratio (ER) (Symonds and Moussalli, 2011). The alternative models were compared to the null model and alternative models with less than two AIC units better (lower) than the null model were not considered distinguishable from a null model (Symonds and Moussalli, 2011). We present the ER of the best model compared with the null model, to evaluate how the AIC values of models were better (lower) than that of the null model. In addition, to clarify how we selected our models, we present the number of parameters (k), Δi-values derived from AICi−AICmin, Akaike weights, and adjusted R2 values, following Burnham and Anderson (2002).
Effect of light environment on beetle morphology
Since the distribution of light in forests can vary depending on the vegetation structure (Ross et al., 1986; Johansson, 1987; Constabel and Lieffers, 1996; Montgomery and Chazdon, 2001), we scored the light environment of each beetle specimen, based on the habitats and vegetation structure of the host plants, following the phyto-sociological classification of Groves (1999). The light environment was classified and scored in terms of habitat (rainforest: 1, wet sclerophyll: 2, dry sclerophyll: 3, heath: 4) and vegetation strata (understorey: 1, tree species: 2), such that darker habitats and vegetation strata have lower scores. We then recorded the sum of these two scores for each beetle species, and deemed darker environments for scores ≤ 4 (the median score across all species), and brighter environments for scores ≥ 5. While this definition of light environment is arbitrary, our intention is to keep the analysis simple with a dichotomy that captures the combined effects of vegetation strata and habitat type on the amount of light incident on the beetle, and thus perception by potential predators. Beetles located in the rainforest understorey would be in a very much darker environment than those found on dry sclerophyll trees. Importantly, these values are derived independent of our study. Thus, our brighter/darker habitat dichotomy is not based on an arbitrary threshold, but rather reflects values that are less or greater than the median value. This binary classification of the environment was subsequently used in the comparative analysis (Figure 3).
Figure 3
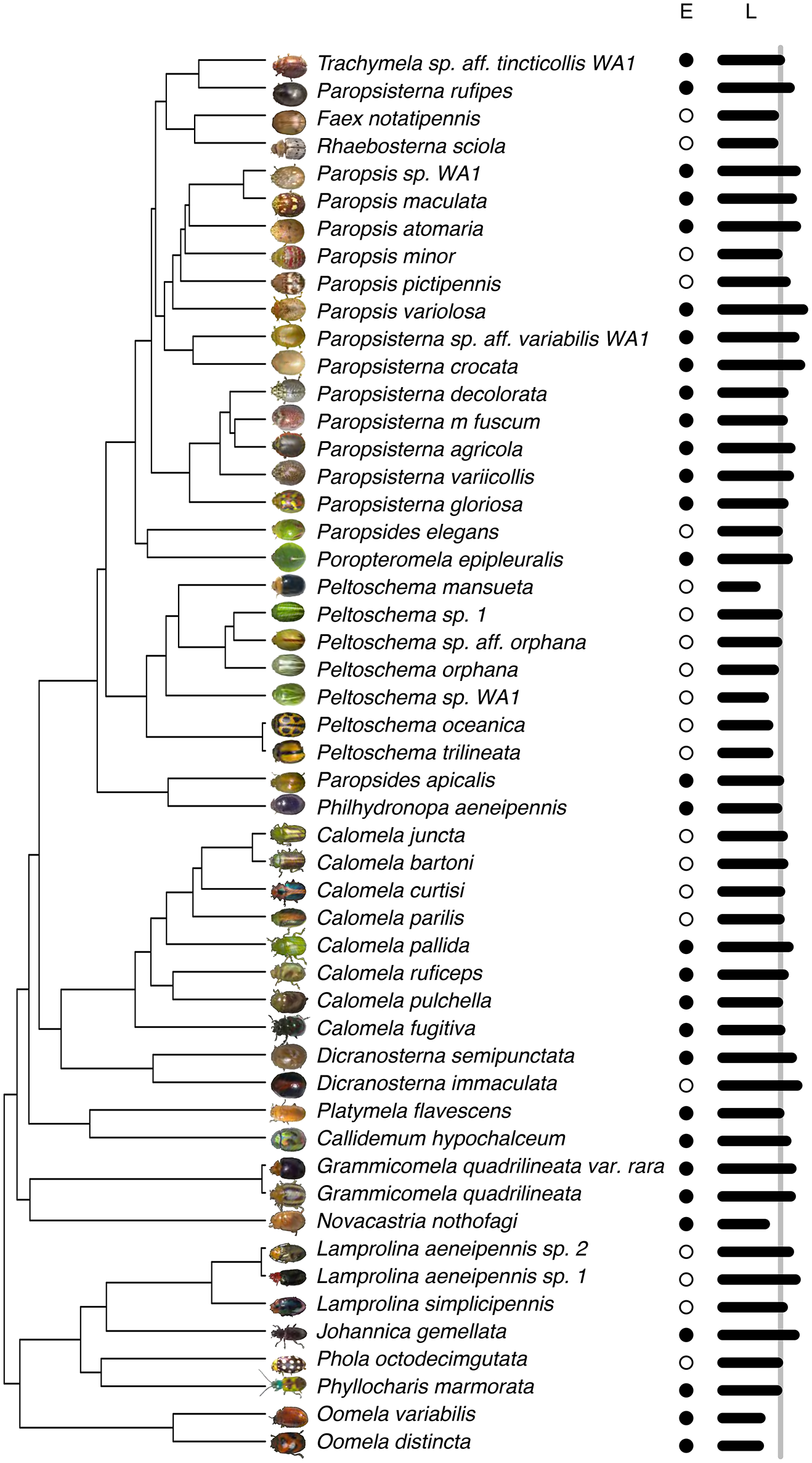
Molecular phylogeny of chrysomelines. Corresponding beetle images (not to scale) are placed at the tips, before the species names. The light environment (E) is represented by open circles (light environments) or closed circles (dark environments). Beetle length (L) is represented by the horizontal lines adjacent to species names and the gray vertical line illustrates the average size of the chrysomelines.
Effect of diet specialization
As the host plant also affects how enemies perceive their animal prey, we examined whether diet specialization (monophagous or polyphagous) affects how beetle color patterns vary with their host plants. Using the ecological information (Table S4), we defined beetles as monophagous if only one species of host plant was recorded, and polyphagous if more than one species of host plant was recorded.
Effect of host plant genera
We examined the effect of host plant genera on the color patterns and beetle morphology in more detail by selecting beetle species that feed on Eucalyptus (n = 13) and Acacia (n = 17), since these were the two dominant genera of host plants within our sample. As Eucalyptus and Acacia dominate almost all of the plant associations of the Australian continent (Barlow, 1981), it is unsurprising that a large proportion of our sampled chrysomeline species feed on these host plants.
As beetle morphology could affect the perception of the beetle against their host plants, we then performed PGLS to test whether beetle length, height, and convexity interacted with host genera to predict color pattern differences in beetles. To assess the value of the interaction terms, we compared the models with and without the interaction term, using the AIC values and the calculation of ER as described above.
Results
Variation in color differences between beetle and host plant
Phylogenetic Principle Components Analysis of the red, green, and blue patterns yielded two principle components (Table 1). PC1 explained 70.6% of the variance (S.D. = 4.13), while PC2 explained 26.0% of the variance (S.D. = 2.50). Green and blue patterns are more important to PC1 while red pattern is more important to PC2. Principal Component 1 (PC1) values of sister taxa are generally similar (Figure 4A), and a similar trend is seen for principal component 2 (PC2) values (Figure 4B).
Table 1
| PC1 | PC2 | PC3 | |
|---|---|---|---|
| Red pattern | −0.529 | 0.842 | −0.107 |
| Green pattern | −0.932 | 0.114 | 0.344 |
| Blue pattern | −0.940 | −0.322 | −0.110 |
Loadings from phylogenetic principal component analysis of differences in red, green, and blue patterns between beetles and host plants.
Figure 4
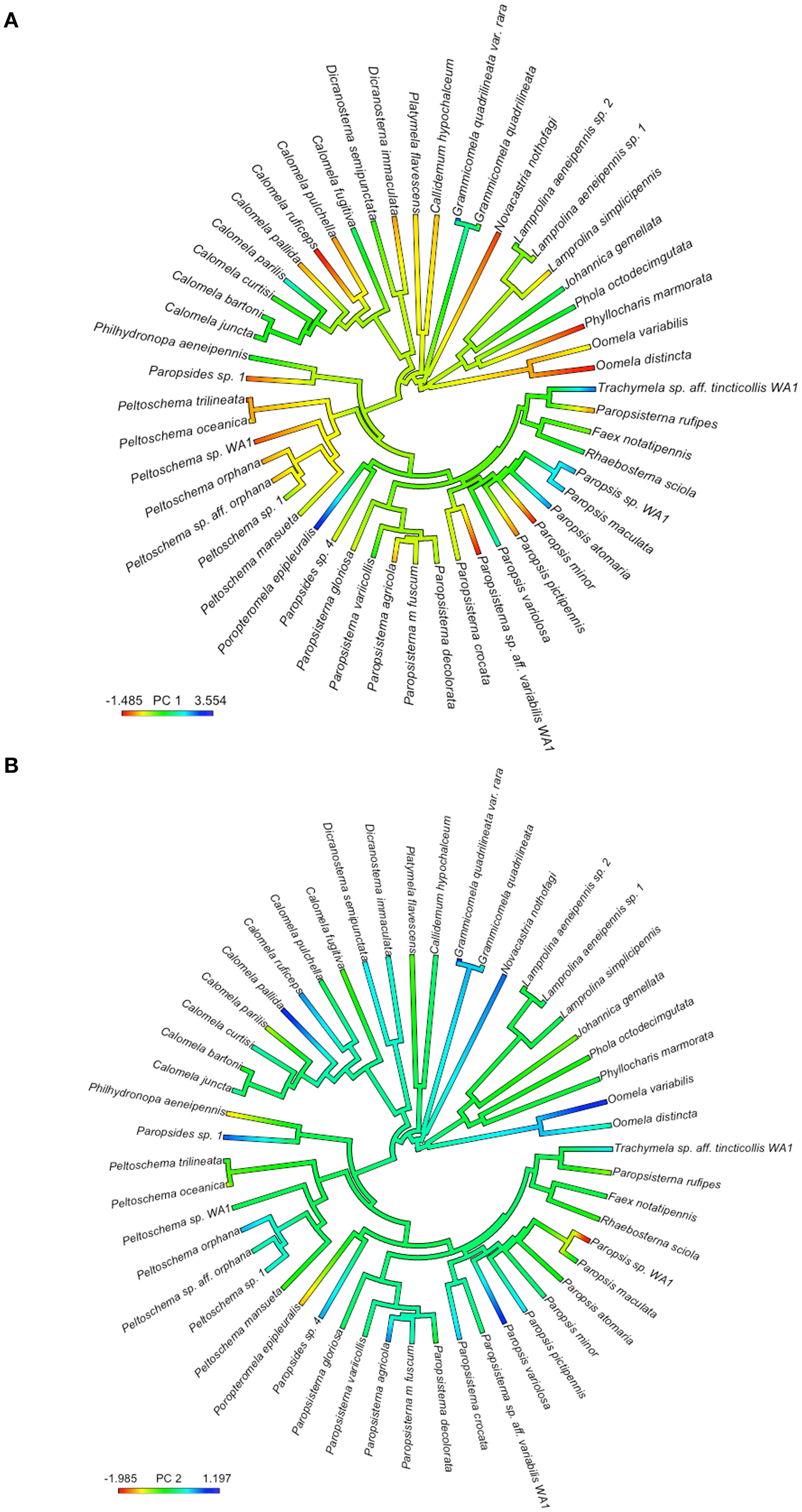
Species values of principal components from the phylogenetic principal components analysis of differences in red, green and blue patterns between beetles and host plants, mapped onto the phylogeny of the chrysomelines. Color on the heat map represent PC1 (A) and PC2 (B) values of species (see legend for scale) but do not represent the actual color of the beetles.
Effect of light environment on beetle morphology
The light environment is a good predictor of beetle length and height. Species that were found in darker environments tend to be larger than beetles in brighter environments (λ = 0.98; β = −0.11 ± 0.04; ER = 347.23; Table 2; Figure 5A). Similarly, species that were found in darker environments tend to be higher than beetles in brighter environments (λ = 0.98; β = −0.11 ± 0.04; ER = 9.79; Table 2; Figure 5B). However, the light environment did not predict beetle convexity (Table 2).
Table 2
| Candidate models | k | Adjusted R2 | AIC value | Δi | wi |
|---|---|---|---|---|---|
| Height | 3 | 0.10 | −72.63 | 0 | 1 |
| Null model | – | 0 | −68.07 | 4.56 | 0 |
| Length | 3 | 0.13 | −81.51 | 0 | 1 |
| Null model | – | 0 | −75.65 | 5.86 | 0 |
| Convexity | 3 | −0.02 | −162.85 | 1.99 | 0 |
| Null model | – | 0 | −164.84 | 0 | 1 |
Effect of light environment on beetle morphology.
Figure 5
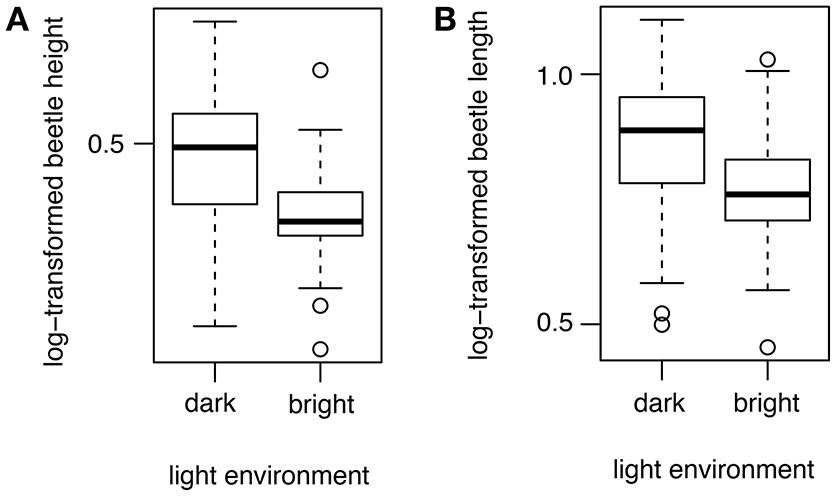
Boxplots illustrating the effect of light environment on beetle morphology. The x-axes represent the light environments as dark or bright. Beetles with greater length (A) and height (B) tend to be in dark environments instead of bright environments.
Effect of beetle morphology on color patterns and contrasts
The green color pattern difference between beetle and the host plant background, a measure or conspicousness, increased with beetle size (λ = 0; β = 2.36 ± 0.82; R2 = 0.15; ER = 539.15; Table 3). Likewise, larger beetles have a greater blue pattern difference compared with their host plant (λ = 0; β = 2.16 ± 0.92; R2 = 0.10; ER = 31.50; Table 3). However, there was no correlation between beetle size and red pattern difference.
Table 3
| Candidate models | k | Adjusted R2 | AIC value | Δi | wi | |
|---|---|---|---|---|---|---|
| Blue | Height | 3 | 0.08 | 142.04 | 0.20 | 0.332 |
| Length | 3 | 0.08 | 141.84 | 0 | 0.366 | |
| Convexity | 3 | 0.01 | 147.01 | 5.17 | 0.028 | |
| Diet specialization | 3 | −0.02 | 147.19 | 5.35 | 0.025 | |
| Height + length | 5 | 0.05 | 143.63 | 1.79 | 0.132 | |
| Height * length | 5 | 0.07 | 145.41 | 3.57 | 0.045 | |
| Null model | – | 0 | 145.29 | 3.45 | 0.071 | |
| Green | Height | 3 | 0.09 | 132.02 | 2.24 | 0.177 |
| Length | 3 | 0.13 | 129.78 | 0 | 0.543 | |
| Convexity | 3 | −0.02 | 138.06 | 8.28 | 0.009 | |
| Diet specialization | 3 | −0.02 | 138.04 | 8.26 | 0.009 | |
| Height + length | 5 | 0.11 | 131.72 | 1.94 | 0.181 | |
| Height * length | 5 | 0.09 | 133.72 | 3.94 | 0.056 | |
| Null model | – | 0 | 136.07 | 6.29 | 0.026 | |
| Red | Height | 3 | −0.01 | 130.02 | 1.64 | 0.133 |
| Length | 3 | 0.00 | 129.39 | 1.01 | 0.184 | |
| Convexity | 3 | −0.01 | 130.03 | 1.65 | 0.133 | |
| Diet specialization | 3 | −0.02 | 130.36 | 1.98 | 0.113 | |
| Height + length | 5 | −0.01 | 130.94 | 2.56 | 0.074 | |
| Height * length | 5 | −0.02 | 132.20 | 3.82 | 0.033 | |
| Null model | – | 0 | 128.38 | 0 | 0.330 |
Effect of beetle morphology and diet specialization on color pattern differences.
The degree of brightness contrast between the beetle and its host plant background, important for predator perception of potential prey, was not consistently predicted by the length, height, and convexity of the beetle. Beetle length did not influence the degree of contrast between beetle and background (Table 4). For all three colors, the null model was better than these models at predicting brightness contrasts between beetle and host plant. Beetle height influences the degree of brightness contrast between beetle and background in blue but not in green and red. Beetles with greater height tend to have a significant blue contrast compared with their host plants (λ = 0; β = 0.82 ± 0.40; R2 = 0.08; ER = 26.17; Table 4; Figure 6A). This was not observed for red contrasts and green contrasts, where the null model was better at predicting the contrast between beetle and host plant (Table 4). However, the convexity of beetles influences the degree of brightness contrast between beetle and background in blue and red colors but not in green. Beetles with greater convexity tend to have a significant blue contrast (λ = 0; β = 4.02 ± 0.98; R2 = 0.26; ER = 5.24 × 105; Table 4; Figure 6B) and red contrast (λ = 0; β = 3.15 ± 1.00; R2 = 0.17; ER = 1,450.99; Table 4; Figure 6C) compared with their host plants. This was not observed for green contrasts (Table 4), where the null model was better at predicting the contrast between beetle and host plant.
Table 4
| Candidate models | k | Adjusted R2 | AIC value | Δi | wi | |
|---|---|---|---|---|---|---|
| Blue | Height | 3 | 0.06 | 70.44 | 11.01 | 0.003 |
| Length | 3 | −0.02 | 74.35 | 14.92 | 0.000 | |
| Convexity | 3 | 0.24 | 59.43 | 0 | 0.722 | |
| Diet specialization | 3 | 0.01 | 72.96 | 13.53 | 0.001 | |
| Height + length | 5 | 0.21 | 62.39 | 2.96 | 0.144 | |
| Height * length | 5 | 0.23 | 62.26 | 2.83 | 0.128 | |
| Null model | – | 0 | 72.60 | 13.17 | 0.001 | |
| Green | Height | 3 | 0.00 | 76.69 | 0.84 | 0.172 |
| Length | 3 | 0.00 | 76.97 | 1.12 | 0.150 | |
| Convexity | 3 | −0.01 | 77.43 | 1.58 | 0.119 | |
| Diet specialization | 3 | −0.02 | 77.77 | 1.92 | 0.100 | |
| Height + length | 5 | −0.02 | 78.69 | 2.84 | 0.056 | |
| Height * length | 5 | 0.04 | 76.84 | 0.99 | 0.117 | |
| Null model | – | 0 | 75.85 | 0 | 0.285 | |
| Red | Height | 3 | 0.06 | 68.38 | 5.64 | 0.041 |
| Length | 3 | −0.01 | 71.41 | 8.67 | 0.009 | |
| Convexity | 3 | 0.15 | 62.74 | 0 | 0.692 | |
| Diet specialization | 3 | −0.02 | 71.84 | 9.1 | 0.007 | |
| Height + length | 5 | 0.12 | 65.70 | 2.96 | 0.138 | |
| Height * length | 5 | 0.12 | 66.13 | 3.39 | 0.093 | |
| Null model | – | 0 | 70.02 | 7.28 | 0.020 |
Effect of beetle morphology on brightness contrasts.
Figure 6
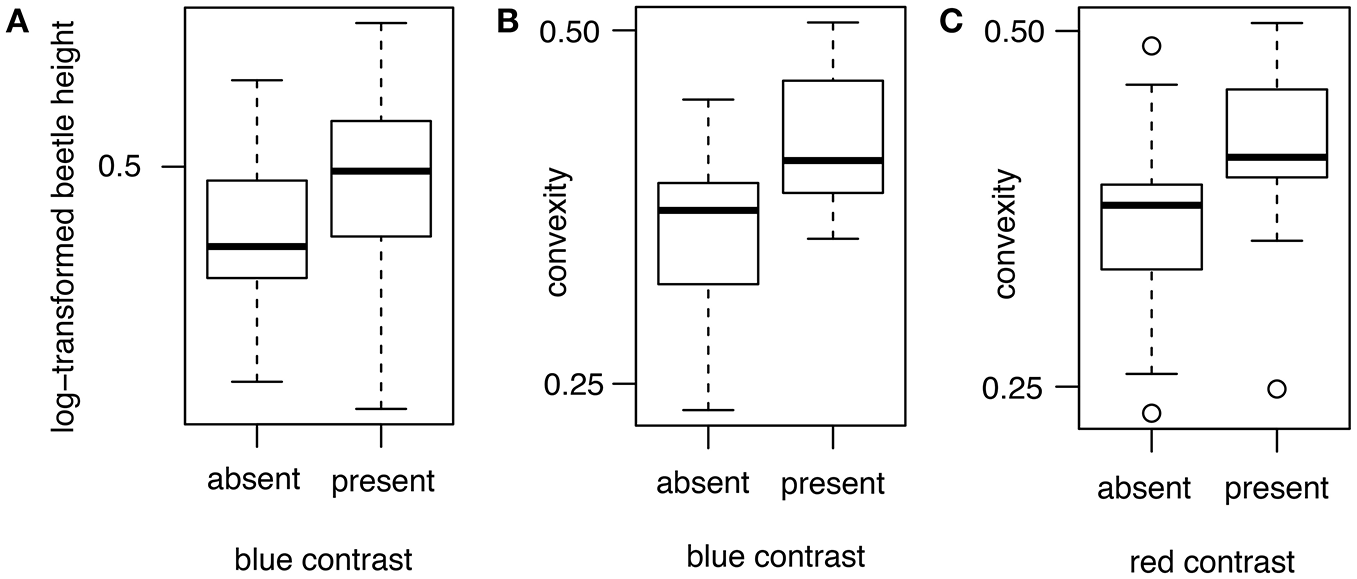
Boxplots illustrating the effect of beetle height on the brightness contrast between beetle and host plant. The x-axis represents the absence or presence of contrast between the beetle and its background. (A) Beetles with greater height tend to have a significant difference in blue contrast with the host plant. Beetles with greater convexity tend to have a significant difference in (B) blue and (C) red contrasts with the host plant.
Effect of diet specialization
Polyphagous beetles had greater green color pattern difference compared with monophagous beetles (λ = 1; β = −0.64 ± 0.27; R2 = 0.10; ER = 6.04; Table 4). Polyphagous beetles also had less red color pattern difference compared with monophagous beetles (λ = 1; β = −1.53 ± 0.25; R2 = 0.42; ER = 6.47 × 105; Table 4). However, there was no effect of diet (i.e., monophagous or polyphagous) on blue color pattern differences (Table 4).
Effect of host plant genera
Host genera and beetle convexity predicted color pattern differences, but to a varying degree. There was a significant interaction between host genera—Acacia and Eucalyptus—and beetle convexity for blue pattern difference (λ = 1; β = 0.69; R2 = 0.32; ER = 17.70; Table 5). For Acacia-feeding beetle species, the blue pattern difference decreased as convexity increased (Figure 7). This contrasted with Eucalyptus-feeding species, as the blue pattern difference increased with increasing convexity. The interaction between host genera and beetle convexity did not better predict the differences in green and red patterns compared with the null model (Table 5). The interaction between host genera and beetle length did not provide a better prediction of color pattern difference compared with the null model, nor did the interaction between host genera and beetle height provide a better prediction of color pattern difference compared with the null model (Table 5).
Table 5
| Candidate models | k | Adjusted R2 | AIC value | Δi | wi | |
|---|---|---|---|---|---|---|
| Blue | Host plant | 3 | 0.04 | 83.37 | 6.69 | 0.048 |
| Height | 3 | 0.04 | 83.41 | 6.73 | 0.047 | |
| Length | 3 | 0.06 | 82.65 | 5.97 | 0.068 | |
| Convexity | 3 | −0.03 | 85.54 | 8.86 | 0.016 | |
| Host plant * height | 5 | 0.02 | 85.73 | 9.05 | 0.008 | |
| Host plant * length | 5 | 0.00 | 86.20 | 9.52 | 0.006 | |
| Host plant * convexity | 5 | 0.27 | 76.68 | 0 | 0.757 | |
| Null model | – | 0 | 83.57 | 6.89 | 0.050 | |
| Green | Host plant | 3 | 0.05 | 67.96 | 0 | 0.290 |
| Height | 3 | −0.03 | 70.12 | 2.16 | 0.099 | |
| Length | 3 | −0.03 | 70.18 | 2.22 | 0.095 | |
| Convexity | 3 | −0.03 | 70.14 | 2.18 | 0.098 | |
| Host plant * height | 5 | 0.01 | 70.83 | 2.87 | 0.039 | |
| Host plant * length | 5 | 0.01 | 70.60 | 2.64 | 0.043 | |
| Host plant * convexity | 5 | 0.04 | 70.36 | 2.40 | 0.049 | |
| Null model | – | 0 | 68.27 | 0.31 | 0.288 | |
| Red | Host plant | 3 | −0.01 | 77.45 | 1.21 | 0.177 |
| Height | 3 | −0.03 | 78.18 | 1.94 | 0.122 | |
| Length | 3 | −0.03 | 78.18 | 1.94 | 0.123 | |
| Convexity | 3 | −0.04 | 78.24 | 2 | 0.119 | |
| Host plant * height | 5 | −0.02 | 79.49 | 3.25 | 0.036 | |
| Host plant * length | 5 | −0.03 | 79.95 | 3.71 | 0.028 | |
| Host plant * convexity | 5 | −0.06 | 80.68 | 4.44 | 0.020 | |
| Null model | – | 0 | 76.24 | 0 | 0.376 |
Effect of host plant and beetle morphology on color pattern differences.
Figure 7
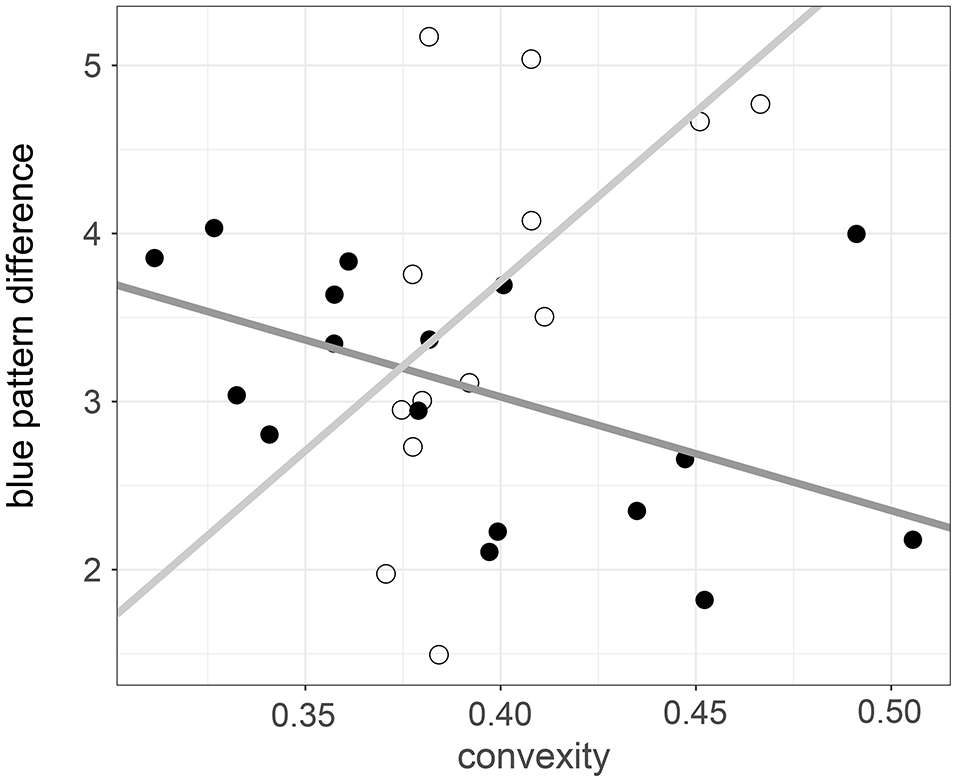
Blue pattern differences between Acacia and Eucalyptus feeding species. Closed circles represent the raw data of Acacia-feeding species while open circles represent the raw data points of Eucalyptus-feeding species. The dark gray line represents the phylogenetically-corrected regression for Acacia-feeding species while the light gray line represents the phylogenetically-corrected regression for Eucalyptus-feeding species.
Discussion
Our results reveal considerable variation in the color pattern difference—quantitative measures of the difference of both color and pattern—between Australian chrysomeline beetles and their host plants, and that this variation is predicted by the host plant specificity of the beetles and their light environment. We found that beetle morphology effects how beetles may be perceived in two ways: (i) larger beetles have a greater green and blue pattern difference compared with their host plants, and (ii) beetles with greater convexity tend to have a significant difference in red and blue contrasts compared with their host plants. Diet and host plant choices also influence beetle color pattern differences. Polyphagous species had significantly greater green difference compared with monophagous beetles, but significantly less red difference compared with monophagous beetles. Finally, host plant taxa influence beetle color patterns, with blue color pattern differences significantly lower for Acacia-feeding species but significantly greater for the more convex, Eucalyptus-feeding species.
Cott (1940) predicted that larger animals should evolve conspicuous coloration because it is harder for large animals to be cryptic. Our findings are broadly consistent with this prediction—larger beetles were more different from the background in blue and green but not red. Red color is a form of aposematic coloration for a range of other organisms, including lepidopterans, coleopterans, and hemipterans (Jones, 1932; Lindstedt et al., 2011; Wee and Monteiro, 2017). However, the difference in red pattern does not differ with chrysomeline beetle length, as might be expected if red was the major component of conspicuous coloration. Indeed, a study of a European chrysomeline indicates that the blue-green metallic color of the beetles are a form of warning coloration (Borer et al., 2010). There are several explanations for the variation in the colors that may be involved in warning coloration in these beetles. First, chrysomeline beetles do not vary much in red; second, physiological constraints may limit the beetles from exhibiting red patterns; third, differences in red may generate consequences that have a counteractive effect, such as attracting potential predators. The beetles in our study ranged from individuals that were entirely red (Oomela variabilis) to beetles that possessed little or no red (Peltoschema orphana). Physiological constraints are unlikely to limit red patterns of beetles through the sequestration or production of red patterns, since species across the phylogeny—Peltoschema oceanica, Paropsisterna nobilitata, and Oomela distincta—possess red patterns, as do other chrysomelid species (for instance, see Kurachi et al., 2002; Keasar et al., 2013; Martínez and Plata-Rueda, 2014). The third explanation is more likely, where differences in red pattern between the beetles and their background may attract potential predators. Thus, instead of increasing inconspicuousness through red patterns, an alternative anti-predator strategy for larger animals is to favor darker environments, which is what we found: species in darker environments tend to be larger, while beetles in lighter environments tending to be smaller. Whether selection on increased body length caused a move to darker environments, or whether a move to darker environments relaxed selection on greater body length and the color patterns, remains unknown.
There is a potential conflict between dietary preferences and avoiding detection by predators. For example, species with a generalist diet may encounter a range of backgrounds and thus be at times conspicuous or inconspicuous. Indeed, Arenas and Stevens (2017) found that aposematic ladybird species that were specialists had evolved the optimal signal of being more contrasting against the background that they were most commonly on. For leaf beetles in our study, polyphagous species had significantly greater green pattern differences to their host plants, compared with monophagous species. However, light reflected from leaves and soil is dominated by green and yellow (Menzel, 1979; Chittka et al., 1994) and the ubiquity of green in the environment may explain the persistence of greater green pattern differences between polyphagous beetles and their host plants. Interestingly, polyphagous beetles had significantly less red difference compared with monophagous beetles. Together with our findings that red pattern does not differ with beetle length, this suggests that the less common adoption of a typical conspicuous color, red, by polyphagous species, could be to limit conspicuousness when they feed on diverse backgrounds. Recent studies of lizards (Marshall et al., 2016) and crabs (Uy et al., 2017) indicate that animals actively choose to match their background. As the spectral sensitivities of chrysomelids appear to be limited to UV, blue and green but not red (Sharkey et al., 2017), active background matching may not fully explain color pattern differences of beetles with their host plant.
Most studies that investigate the antipredator function of color patterns in insects focus predominantly on avian predators (e.g., Greenwood et al., 1981; Roper and Cook, 1989; Roper, 1990; Gamberale and Tullberg, 1996a; Marples et al., 1998; Gamberale-Stille and Tullberg, 1999; Lindström et al., 2001; Thomas et al., 2003; Forsman and Herrström, 2004; Exnerová et al., 2006; Rowland et al., 2007; Sandre et al., 2010; Ihalainen and Lindstedt, 2012). Yet, insect prey species such as chrysomeline beetles, may also be subject to predation by much smaller animals, including spiders and predatory insects. Different predators will view the beetles from different perspectives, and this is likely to have an impact on the selection pressures favoring anti-predator responses. For example, avian predators will view the beetles from above and identify the length of the animal from the dorsal perspective, while smaller invertebrates such as predatory insects and spiders may be approaching from the same substrate and will take a dorsal perspective.
We found no evidence that beetle length and host genera predicted color patterns. On the other hand, the convexity of the beetles and host genera appear to be more important in predicting color pattern differences. In addition, our studies indicate that beetles with greater convexity tend to have a significant red and blue contrast compared with their host plants, but this was not observed for green contrasts. Thus, it is surprising to see a lack of interaction between color pattern and contrasts, with beetle length. The influence of body convexity on conspicuousness is not widely recognized, but our results reveal there was a significant interaction between the convexity of beetles and host genera for blue pattern differences. As convexity increased, the blue pattern difference decreased for Acacia-feeding species, but increased for Eucalyptus-feeding species. This trend is expected for both beetle length and convexity if there are consistent differences in both the color and nutritional value of the leaves of Acacia compared with Eucalyptus species. However, a pattern emerges for convexity only, suggesting that the selection pressure comes from predators that view the beetles laterally. The dissimilar trends observed in blue pattern difference between Acacia and Eucalyptus feeding species suggest that the beetles are subjected to different predation pressures on these host genera, likely because of different guilds of predators on the Acacia and Eucalyptus host genera.
Leaf beetles are likely to have a large number of visual predators that will view the beetles from a lateral perspective. Such predators would have to be on the same substrate as the beetles. Active hunting arachnids are potential predators exerting a selection pressure on chrysomelines, as arachnids would view the beetles at a lateral perspective without succumbing to their chemical defenses (Hilbeck and Kennedy, 1996; Nahrung et al., 2008; Lundgren et al., 2009). Most arachnid species are sensitive to green, while a few taxa are sensitive to blue and red wavelengths (Dahl and Granda, 1989; Peaslee and Wilson, 1989; Zurek et al., 2015). Combined, the visual system of potential arachnid predators, and fewer arachnids on Eucalypts (Woinarski and Cullen, 1984) may result in low selection pressure from arachnid predators, explaining why beetles feeding on Eucalyptus can evolve different blue patterns from their background.
In contrast, selection pressures on beetle length are likely to be influenced by predators taking a dorsal perspective, such as, insectivorous birds. Birds are important predators of beetles in Eucalypt forests (Recher and Majer, 2006), and potential predators of chrysomeline beetles are thornbills (Acanthiza) that forage on arthropods on Eucalypt foliage (Recher et al., 1987; Recher, 1989). Insectivorous birds can be highly selective between different species of Eucalypts and Acacia (Recher, 1989; Recher et al., 1996; Dean et al., 2002), which may reflect arthropod abundance on Eucalyptus species (Recher and Majer, 1994). Indeed, avian species richness is correlated with vegetation structure complexity and species (Shurcliff, 1980; Nally et al., 2008; Brady and Noske, 2010), and with rainfall (Tischler et al., 2013), and the greater the ratio of Eucalypts to Acacia, the higher the avian species richness (Brady and Noske, 2010). However, as avian species and individuals may differ in their reactions to aposematic prey (Exnerová et al., 2008; Svádová et al., 2010), there may be varying selection pressures on leaf beetle color pattern. Together, the background and the predator community can affect the direction and strength of selection for warning coloration (Prudic et al., 2007a; Lindstedt et al., 2011). Many avian species are sensitive to red, in addition to green and blue (see review by Jones et al., 2007), and leaf beetles may have evolved weak aposematic signaling because of the variety of avian predators and the varying tendency of avian predators to attack defended prey (Endler and Mappes, 2004).
Comparative studies like this can be a tool to identify broad trends of selection that can inform future experimental studies (Harvey and Pagel, 1991). Chrysomelines possess a diverse defensive chemistry, including compounds from various chemical classes such as, phenolic, cyanogenic, and cardiac glycosides, pyrrolizidine alkaloids, and cucurbitacins (reviewed by Opitz and Müller, 2009; Morgan, 2010). As de novo synthesis of defensive compounds is expected to be energetically more costly (Kirsch et al., 2011), species that use host-derived defensive compounds could potentially invest more on costly color patterns. A significant correlation in the evolution of toxicity and conspicuous coloration is found in the dendrobatid frogs (Summers and Clough, 2001), and a similar process may influence the evolution of coloration in the Australian chrysomelines. A good system to investigate the effect of defensive compounds on color patterns would be the chrysomelines of the genus Phratora (Köpf et al., 1998). The color patterns of species within this genus, which have been extensively studied in Europe because of their economic significance, have not been quantified, but there is considerable variation in their chemical defenses and host plants.
The common wisdom is that bright coloration, such as that found in leaf beetles, reflects defense through aposematic coloration, but this singular explanation is not supported by our data. The variation in color patterns of chrysomelines is influenced, and possibly limited, by morphological and ecological factors, including beetle length, light environment, and host plant use. Importantly, this indicates that when closely related, chemically defended species radiate into similar ecological habitats and hosts, their color patterns change as a result of novel selection pressures. For example, diet specialization affected the color difference between beetle and background, with polyphagous species having significantly greater green difference but less red difference compared with monophagous beetles. The joint evolution of color pattern and habitat choice could lead to species divergence, with selection through natural enemies driving different sized species into different habitats of darker and lighter environments. Our study reveals the complexities involved in the evolution of color patterns in a single, relatively isolated clade of Australian chrysomelines, which could reflect selection pressures exerted by diverse predators.
Statements
Ethics statement
The research reported in this paper, which involved insects only, was exempt from ethical approval procedures.
Author contributions
ET, CR, MS, and ME conceived and designed the work; ET and CR collected the live specimens; JJ-R collected and analyzed the data for the molecular phylogeny; ET, MS, and ME analyzed and interpreted the comparative data; ET and ME drafted the work; ET, CR, MS, JJ-R, and ME revised the work critically for important intellectual content.
Funding
The authors thank the Holsworth Wildlife Endowment for funding the research, and the Australian Postgraduate Award and the International Postgraduate Research Scholarship for funding ET's postgraduate candidacy.
Acknowledgments
The authors thank Nik Tatarnic, Kate Umbers, Antoni Lunn, Daisy Lunn, Daniel Dobrosak, Evi Reid, and Ewan Reid for advice and help with beetle collections.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fevo.2017.00140/full#supplementary-material
References
1
Arenas L. M. Stevens M. (2017). Diversity in warning coloration is easily recognized by avian predators. J. Evol. Biol.30, 1288–1302. 10.1111/jeb.13074
2
Aronsson M. Gamberale-Stille G. (2009). Importance of internal pattern contrast and contrast against the background in aposematic signals. Behav. Ecol.20, 1356–1362. 10.1093/beheco/arp141
3
Baker M. R. Kitching R. L. Reid C. A. M. Sheldon F. (2012). Coleoptera (Chrysomelidae, Coccinellidae, Curculionoidea) in sclerophyll woodland: variation in assemblages among host plants, and host specificity of phytophagous and predatory beetles. Aust. J. Entomol.51, 145–153. 10.1111/j.1440-6055.2011.00851.x
4
Balsbaugh E. U. Jr. (1988). Mimicry and the chrysomelidae, in Biology of Chrysomelidae, eds JolivetP.PetitpierreE.HsiaoT. H. (Dordrecht: Kluwer Academic Publishers), 261–284.
5
Barlow B. A. (1981). The Australian flora: its origin and evolution, in The Flora of Australia, eds RobertsonR.BriggsB. G.EichlerH.PedleyL.RossJ. H.SymonD. F.WilsonP. G.McCuskerA.GeorgeA. S. (Canberra, ACT: Australian Government Publishing Service), 25–75.
6
Bernays E. A. (1997). Feeding by lepidopteran larvae is dangerous. Ecol. Entomol.22, 121–123. 10.1046/j.1365-2311.1997.00042.x
7
Blum M. S. Brand J. M. Wallace J. B. Fales H. M. (1972). Chemical characterization of the defensive secretion of a chrysomelid larva. Life Sci.11(10 Pt 2), 525–531. 10.1016/0024-3205(72)90286-X
8
Borer M. Van Noort T. Rahier M. Naisbit R. E. (2010). Positive frequency-dependent selection on warning color in alpine leaf beetles. Evolution64, 3629–3633. 10.1111/j.1558-5646.2010.01137.x
9
Brady C. J. Noske R. A. (2010). Succession in bird and plant communities over a 24-year chronosequence of mine rehabilitation in the australian monsoon tropics. Restor. Ecol.18, 855–864. 10.1111/j.1526-100X.2008.00511.x
10
Brönmark C. Miner J. G. (1992). Predator-induced phenotypical change in body morphology in Crucian carp. Science258, 1348–1350. 10.1126/science.258.5086.1348
11
Burnham K. P. Anderson D. R. (2002). Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach. New York, NY: Springer-Verlag.
12
Cheney K. L. Cortesi F. How M. J. Wilson N. G. Blomberg S. P. Winters A. E. et al . (2014). Conspicuous visual signals do not coevolve with increased body size in marine sea slugs. J. Evol. Biol.27, 676–687. 10.1111/jeb.12348
13
Chittka L. Shmida A. Troje N. Menzel R. (1994). Ultraviolet as a component of flower reflections, and the colour perception of Hymenoptera. Vis. Res.34, 1489–1508. 10.1016/0042-6989(94)90151-1
14
Constabel A. J. Lieffers V. J. (1996). Seasonal patterns of light transmission through boreal mixedwood canopies. Can. J. For. Res.26, 1008–1014. 10.1139/x26-111
15
Cook R. G. Qadri M. A. J. Kieres A. Commons-Miller N. (2012). Shape from shading in pigeons. Cognition124, 284–303. 10.1016/j.cognition.2012.05.007
16
Cott H. B. (1940). Adaptive Coloration in Animals. London: Methuen.
17
Cuthill I. C. Stevens M. Sheppard J. Maddocks T. Parraga C. A. Troscianko T. S. (2005). Disruptive coloration and background pattern matching. Nature434, 72–74. 10.1038/nature03312
18
Dahl R. D. Granda A. M. (1989). Spectral sensitivities of photoreceptors in the ocelli of the tarantula Aphonopelma chalcodes (Araneae, Theraphosidae). J. Arachnol.17, 195–205.
19
Dean W. R. J. Anderson M. D. Milton S. J. Anderson T. A. (2002). Avian assemblages in native Acacia and alien Prosopis drainage line woodland in the Kalahari, South Africa. J. Arid Environ.51, 1–19. 10.1006/jare.2001.0910
20
de Little D. W. (1979). Taxonomic and Ecological Studies of the Tasmanian Eucalypt-Defoliating Paropsids. Ph.D., University of Tasmania.
21
Endler J. A. (1978). A predator's view of animal color patterns. Evol. Biol.11, 319–364. 10.1007/978-1-4615-6956-5_5
22
Endler J. A. (1984). Progressive background matching in moths, and a quantitative measure of crypsis. Biol. J. Linn. Soc.22, 187–231. 10.1111/j.1095-8312.1984.tb01677.x
23
Endler J. A. Mappes J. (2004). Predator mixes and the conspicuousness of aposematic signals. Am. Nat.163, 532–547. 10.1086/382662
24
Espinosa I. Cuthill I. C. (2014). Disruptive colouration and perceptual grouping. PLoS ONE9:e87153. 10.1371/journal.pone.0087153
25
Evans H. E. Hook A. W. (1986). Prey selection by Australian wasps of the genus Cerceris (Hymenoptera, Sphecidae). J. Nat. Hist.20, 1297–1307. 10.1080/00222938600770861
26
Exnerová A. Svádová K. Fousová P. Fučíková P. JeŽová D. Niederlová A. et al . (2008). European birds and aposematic Heteroptera: review of comparative experiments. Bull. Insectol.61, 163–165.
27
Exnerová A. Svadová K. Štys P. Barcalová S. Landová E. Prokopová M. et al . (2006). Importance of colour in the reaction of passerine predators to aposematic prey: experiments with mutants of Pyrrhocoris apterus (Heteroptera). Biol. J. Linn. Soc.88, 143–153. 10.1111/j.1095-8312.2006.00611.x
28
Forsman A. Herrström J. (2004). Asymmetry in size, shape, and color impairs the protective value of conspicuous color patterns. Behav. Ecol.15, 141–147. 10.1093/beheco/arg092
29
Forsman A. Merilaita S. (1999). Fearful symmetry: pattern size and asymmetry affects aposematic signal efficacy. Evol. Ecol.13, 131–140. 10.1023/A:1006630911975
30
Gamberale G. Tullberg B. S. (1996a). Evidence for a more effective signal in aggregated aposematic prey. Anim. Behav.52, 597–601. 10.1006/anbe.1996.0200
31
Gamberale G. Tullberg B. S. (1996b). Evidence for a peak-shift in predator generalization among aposematic prey. Proc. Biol. Sci.263, 1329–1334. 10.1098/rspb.1996.0195
32
Gamberale G. Tullberg B. S. (1998). Aposematism and gregariousness: the combined effect of group size and coloration on signal repellence. Proc. Biol. Sci.265, 889–894. 10.1098/rspb.1998.0374
33
Gamberale-Stille G. Tullberg B. (1999). Experienced chicks show biased avoidance of stronger signals: an experiment with natural colour variation in live aposematic prey. Evol. Ecol.13, 579–589. 10.1023/A:1006741626575
34
Gittleman J. L. Harvey P. H. (1980). Why are distasteful prey not cryptic?Nature286, 149–150. 10.1038/286149a0
35
Greenwood J. Wood E. Batchelor S. (1981). Apostatic selection of distasteful prey. Heredity47, 27–34. 10.1038/hdy.1981.56
36
Groves R. H. (1999). Present vegetation types, in Flora of Australia (Melbourne, VIC: Australian Biological Resources Study/CSIRO Publishing).
37
Harvey P. H. Pagel M. (1991). The Comparative Method in Evolutionary Biology. Oxford: Oxford University Press.
38
Hilbeck A. Kennedy G. G. (1996). Predators feeding on the colorado potato beetle in insecticide-free plots and insecticide-treated commercial potato fields in Eastern North Carolina. Biol. Control6, 273–282. 10.1006/bcon.1996.0034
39
Hilker M. Schulz S. (1994). Composition of larval secretion of Chrysomela lapponica (Coleoptera, Chrysomelidae) and its dependence on host plant. J. Chem. Ecol.20, 1075–1093. 10.1007/BF02059744
40
Honma A. Mappes J. Valkonen J. K. (2015). Warning coloration can be disruptive: aposematic marginal wing patterning in the wood tiger moth. Ecol. Evol.5, 4863–4874. 10.1002/ece3.1736
41
Ihalainen E. Lindstedt C. (2012). Do avian predators select for seasonal polyphenism in the European map butterfly Araschnia levana (Lepidoptera: Nymphalidae)?Biol. J. Linn. Soc.106, 737–748. 10.1111/j.1095-8312.2012.01922.x
42
Johansson T. (1987). Irradiance in thinned Norway spruce (Picea abies) stands and the possibilities to prevent suckers of broadleaved trees. For. Ecol. Manage.20, 307–319. 10.1016/0378-1127(87)90087-9
43
Jolivet P. H. Hawkeswood T. J. (1995). Host-Plants of Chrysomelidae of the World: An Essay about the Relationships between the Leaf-Beetles and their Food-Plants. Leiden: Backhuys.
44
Jones F. M. (1932). Insect coloration and the relative acceptability of insects to birds. Trans. R. Entomol. Soc. Lond.80, 345–371. 10.1111/j.1365-2311.1932.tb03313.x
45
Jones M. P. Pierce K. E. Jr. Ward D. (2007). Avian vision: a review of form and function with special consideration to birds of prey. J. Exot. Pet Med. 16, 69–87. 10.1053/j.jepm.2007.03.012
46
Jurado-Rivera J. A. (2014). Filogenia Molecular, Sistemática y Evolución de los Chrysomelinae Australianos (Coleoptera, Chrysomelidae). Ph.D. thesis, University of the Balearic Islands, Spain.
47
Jurado-Rivera J. A. Vogler A. P. Reid C. A. M. Petitpierre E. Gomez-Zurita J. (2009). DNA barcoding insect-host plant associations. Proc. Biol. Sci.276, 639–648. 10.1098/rspb.2008.1264
48
Karpestam E. Merilaita S. Forsman A. (2014). Body size influences differently the detectabilities of colour morphs of cryptic prey. Biol. J. Linn. Soc.113, 112–122. 10.1111/bij.12291
49
Keasar T. Kishinevsky M. Shmida A. Gerchman Y. Chinkov N. Koplovich A. et al . (2013). Plant-derived visual signals may protect beetle herbivores from bird predators. Behav. Ecol. Sociobiol.67, 1613–1622. 10.1007/s00265-013-1572-z
50
Khang B. G. Koenderink J. J. Kappers A. M. L. (2006). Perception of illumination direction in images of 3-D convex objects: influence of surface materials and light fields. Perception35, 625–645. 10.1068/p5485
51
Kirsch R. Vogel H. Muck A. Reichwald K. Pasteels J. M. Boland W. (2011). Host plant shifts affect a major defense enzyme in Chrysomela lapponica. Proc. Natl. Acad. Sci. U.S.A.108, 4897–4901. 10.1073/pnas.1013846108
52
Kjernsmo K. Merilaita S. (2012). Background choice as an anti-predator strategy: the roles of background matching and visual complexity in the habitat choice of the least killifish. Proc. Biol. Sci.279, 4192–4198. 10.1098/rspb.2012.1547
53
Köpf A. Rank N. E. Roininen H. Julkunen-Tiitto R. Pasteels J. M. Tahvanainen J. (1998). The evolution of host-plant use and sequestration in the leaf beetle genus Phratora (Coleoptera: Chrysomelidae). Evolution52, 517–528. 10.1111/j.1558-5646.1998.tb01651.x
54
Krebs J. R. Erichsen J. T. Webber M. I. Charnov E. L. (1977). Optimal prey selection in the great tit (Parus major). Anim. Behav. 25 (Pt 1), 30–38. 10.1016/0003-3472(77)90064-1
55
Kurachi M. Takaku Y. Komiya Y. Hariyama T. (2002). The origin of extensive colour polymorphism in Plateumaris sericea (Chrysomelidae, Coleoptera). Naturwissenschaften89, 295–298. 10.1007/s00114-002-0332-0
56
Lawrence J. F. Britton E. B. (1994). Australian Beetles. Melbourne, VIC: Melbourne University Press.
57
Lindstedt C. Eager H. Ihalainen E. Kahilainen A. Stevens M. Mappes J. (2011). Direction and strength of selection by predators for the color of the aposematic wood tiger moth. Behav. Ecol.22, 580–587. 10.1093/beheco/arr017
58
Lindstedt C. Lindström L. Mappes J. (2008). Hairiness and warning colours as components of antipredator defence: additive or interactive benefits?Anim. Behav.75, 1703–1713. 10.1016/j.anbehav.2007.10.024
59
Lindström L. Alatalo R. V. Lyytinen A. Mappes J. (2001). Predator experience on cryptic prey affects the survival of conspicuous aposematic prey. Proc. Biol. Sci.268, 357–361. 10.1098/rspb.2000.1377
60
Lindström L. Alatalo R. V. Mappes J. Riipi M. Vertainen L. (1999). Can aposematic signals evolve by gradual change?Nature397, 249–251. 10.1038/16692
61
Llandres A. L. Gawryszewski F. M. Heiling A. M. Herberstein M. E. (2011). The effect of colour variation in predators on the behaviour of pollinators: australian crab spiders and native bees. Ecol. Entomol.36, 72–81. 10.1111/j.1365-2311.2010.01246.x
62
Lundgren J. G. Ellsbury M. E. Prischmann D. A. (2009). Analysis of the predator community of a subterranean herbivorous insect based on polymerase chain reaction. Ecol. Appl. 19, 2157–2166. 10.1890/08-1882.1
63
Maddison W. P. Maddison D. R. (2014). Mesquite: A Modular System for Evolutionary Analysis. Version 3.0. Available online at: http://mesquiteproject.org
64
Mappes J. Kokko H. Ojala K. Lindström L. (2014). Seasonal changes in predator community switch the direction of selection for prey defences. Nat. Commun.5, 5016. 10.1038/ncomms6016
65
Marples N. Roper T. J. Harper D. G. C. (1998). Responses of wild birds to novel prey: evidence of dietary conservatism. Oikos83, 161–165. 10.2307/3546557
66
Marshall K. L. A. Philpot K. E. Stevens M. (2016). Microhabitat choice in island lizards enhances camouflage against avian predators. Sci. Rep.6:19815. 10.1038/srep19815
67
Martínez L. C. Plata-Rueda A. (2014). Biological aspects and food consumption of oil palm fruit scraper, Demotispa neivai (Coleoptera: Chrysomelidae). J. Oil Palm Res.26, 47–53.
68
Matthews E. G. Reid C. A. M. (2002). A guide to the Genera of Beetles of South Australia Part 8. Adelaide: South Australian Museum.
69
Menzel R. (1979). Spectral sensitivity and color vision in invertebrates, in Comparative Physiology and Evolution of Vision in Invertebrates: A: Invertebrate Photoreceptors, eds AutrumH.BennettM. F.DiehnB.HamdorfK.HeisenbergM.JärvilehtoM.KunzeP.MenzelR.MillerW. H.SnyderA. W.StavengaD. G.YoshidaM.AutrumH. (Berlin; Heidelberg: Springer Berlin Heidelberg), 503–580.
70
Merilaita S. Dimitrova M. (2014). Accuracy of background matching and prey detection: predation by blue tits indicates intense selection for highly matching prey colour pattern. Funct. Ecol.28, 1208–1215. 10.1111/1365-2435.12248
71
Montgomery R. A. Chazdon R. L. (2001). Forest structure, canopy architecture, and light transmittance in tropical wet forests. Ecology82, 2707–2718. 10.1890/0012-9658(2001)082[2707:FSCAAL]2.0.CO;2
72
Moore B. P. (1980). A Guide to the Beetles of South-Eastern Australia. Greenwich: Australian Entomological Press.
73
Morgan D. E. (2010). Plant substances altered and sequestered by insects, in Biosynthesis in Insects (Cambridge: Royal Society of Chemistry), 315–331.
74
Nahrung H. F. Duffy M. P. Lawson S. A. Clarke A. R. (2008). Natural enemies of Paropsis atomaria Olivier (Coleoptera: Chrysomelidae) in south-eastern Queensland eucalypt plantations. Aust. J. Entomol.47, 188–194. 10.1111/j.1440-6055.2008.00656.x
75
Nally R. M. Fleishman E. Thomson J. R. Dobkin D. S. (2008). Use of guilds for modelling avian responses to vegetation in the Intermountain West (USA). Glob. Ecol. Biogeogr.17, 758–769. 10.1111/j.1466-8238.2008.00409.x
76
Nilsson M. Forsman A. (2003). Evolution of conspicuous colouration, body size and gregariousness: a comparative analysis of lepidopteran larvae. Evol. Ecol.17, 51–66. 10.1023/A:1022417601010
77
Nilsson P. A. Brönmark C. Pettersson L. B. (1995). Benefits of a predator-Induced morphology in crucian carp. Oecologia104, 291–296. 10.1007/BF00328363
78
Novotny V. Basset Y. Miller S. E. Drozd P. Cizek L. (2002). Host specialization of leaf-chewing insects in a New Guinea rainforest. J. Anim. Ecol.71, 400–412. 10.1046/j.1365-2656.2002.00608.x
79
Novotny V. Miller S. E. Hulcr J. Drew R. A. I. Basset Y. Janda M. et al . (2007). Low beta diversity of herbivorous insects in tropical forests. Nature448, 692–695. 10.1038/nature06021
80
Ojala K. Lindström L. Mappes J. (2007). Life-history constraints and warning signal expression in an arctiid moth. Funct. Ecol.21, 1162–1167. 10.1111/j.1365-2435.2007.01322.x
81
Opitz S. W. Müller C. (2009). Plant chemistry and insect sequestration. Chemoecology19, 117–154. 10.1007/s00049-009-0018-6
82
Orme D. Freckleton R. Thomas G. Petzoldt T. Fritz S. Isaac N. et al . (2013). caper: Comparative Analyses of Phylogenetics and Evolution in R. R package version 0.5.2. Available online at: https://CRAN.R-project.org/package=caper
83
Outomuro D. Johansson F. (2015). Bird predation selects for wing shape and coloration in a damselfly. J. Evol. Biol.28, 791–799. 10.1111/jeb.12605
84
Pagel M. (1997). Inferring evolutionary processes from phylogenies. Zool. Scr.26, 331–348. 10.1111/j.1463-6409.1997.tb00423.x
85
Pagel M. (1999). Inferring the historical patterns of biological evolution. Nature401, 877–884. 10.1038/44766
86
Paradis E. Claude J. Strimmer K. (2004). APE: analyses of phylogenetics and evolution in R language. Bioinformatics20, 289–290. 10.1093/bioinformatics/btg412
87
Pasteels J. M. Braekman J. C. Daloze D. Ottinger R. (1982). Chemical defence in chrysomelid larvae and adults. Tetrahedron38, 1891–1897. 10.1016/0040-4020(82)80038-0
88
Pasteels J. M. Rowell-Rahier M. Brackman J. C. Daloze D. Duffey S. (1989). Evolution of exocrine chemical defense in leaf beetles (Coleoptera: Chrysomelidae). Experientia45, 295–300. 10.1007/BF01951815
89
Pasteels J. M. Rowell-Rahier M. Braekman J. C. Dupont A. (1983). Salicin from host plant as precursor of salicylaldehyde in defensive secretion of Chrysomeline larvae. Physiol. Entomol.8, 307–314. 10.1111/j.1365-3032.1983.tb00362.x
90
Pawlik J. Chanas B. Toonen R. Fenical W. (1995). Defenses of Caribbean sponges against predatory reef fish. I. Chemical deterrency. Mar. Ecol. Prog. Ser.127, 183–194. 10.3354/meps127183
91
Peaslee A. G. Wilson G. (1989). Spectral sensitivity in jumping spiders (Araneae, Salticidae). J. Comp. Physiol. A164, 359–363. 10.1007/BF00612995
92
Pellissier L. Wassef J. Bilat J. Brazzola G. Buri P. Colliard C. et al . (2011). Adaptive colour polymorphism of Acrida ungarica H. (Orthoptera: Acrididae) in a spatially heterogeneous environment. Acta Oecologica37, 93–98. 10.1016/j.actao.2010.12.003
93
Prudic K. L. Oliver J. C. Sperling F. A. H. (2007a). The signal environment is more important than diet or chemical specialization in the evolution of warning coloration. Proc. Natl. Acad. Sci. U.S.A.104, 19381–19386. 10.1073/pnas.0705478104
94
Prudic K. L. Skemp A. K. Papaj D. R. (2007b). Aposematic coloration, luminance contrast, and the benefits of conspicuousness. Behav. Ecol.18, 41–46. 10.1093/beheco/arl046
95
Qadri M. A. J. Romero M. L. Cook R. G. (2014). Shape from shading in starlings (Sturnus vulgaris). J. Comp. Psychol.127, 343–356. 10.1037/a.0036848
96
R Core Team (2016). R: A Language and Environment for Statistical Computing.Vienna: R Foundation for Statistical Computing. Available online at: http://www.R-project.org/
97
Rahfeld P. Haeger W. Kirsch R. Pauls G. Becker T. Schulze E. et al . (2015). Glandular β-glucosidases in juvenile Chrysomelina leaf beetles support the evolution of a host-plant-dependent chemical defense. Insect Biochem. Mol. Biol.58, 28–38. 10.1016/j.ibmb.2015.01.003
98
Ramachandran V. S. (1988). Perception of shape from shading. Nature331, 163–166. 10.1038/331163a0
99
Recher H. F. (1989). Foraging segregation of australian warblers (Acanthizidae) in open forest near Sydney, New South Wales. Emu89, 204–215. 10.1071/MU9890204
100
Recher H. F. Davis W. E. Holmes R. T. (1987). Ecology of brown and striated thornbills in forests of South-eastern New South Wales, with comments on forest management. Emu87, 1–13. 10.1071/MU9870001
101
Recher H. F. Majer J. D. (1994). On the selection of tree species by Acanthizidae in open-forest near Sydney, New South Wales. Emu94, 239–245. 10.1071/MU9940239
102
Recher H. F. Majer J. D. (2006). Effects of bird predation on canopy arthropods in wandoo Eucalyptus wandoo woodland. Austral Ecol.31, 349–360. 10.1111/j.1442-9993.2006.01555.x
103
Recher H. F. Majer J. D. Ganesh S. (1996). Eucalypts, arthropods and birds: on the relation between foliar nutrients and species richness. For. Ecol. Manage.85, 177–195. 10.1016/S0378-1127(96)03758-9
104
Reid C. A. M. (2006). A taxonomic revision of the Australian Chrysomelinae, with a key to the genera (Coleoptera: Chrysomelidae). Zootaxa1292, 1–119. 10.11646/zootaxa.1292.1.1
105
Reid C. A. M. (2014). Chrysomelinae Latreille 1802, in Coleoptera, Beetles Morphology and Systematics (Phytophaga), Vol. 3, eds LeschenR. A. B.BeutelR. G. (Berlin: De Gruyter), 243–251.
106
Reid C. A. M. (2017). Australopapuan leaf beetle diversity: the contributions of hosts plants and geography. Austral Entomol.56, 123–137. 10.1111/aen.12251
107
Reid C. A. M. Jurado-Rivera J. A. Beatson M. (2009). A new genus of Chrysomelinae from Australia (Coleoptera: Chrysomelidae). Zootaxa2207, 53–66.
108
Revell L. J. (2012). Phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol.3, 217–223. 10.1111/j.2041-210X.2011.00169.x
109
Roper T. J. (1990). Responses of domestic chicks to artificially coloured insect prey: effects of previous experience and background colour. Anim. Behav.39, 466–473. 10.1016/S0003-3472(05)80410-5
110
Roper T. J. Cook S. E. (1989). Responses of chicks to brightly coloured insect prey. Behaviour110, 276–293. 10.1163/156853989X00510
111
Roper T. J. Redston S. (1987). Conspicuousness of distasteful prey affects the strength and durability of one-trial avoidance learning. Anim. Behav.35, 739–747. 10.1016/S0003-3472(87)80110-0
112
Roper T. J. Wistow R. (1986). Aposematic colouration and avoidance learning in chicks. Q. J. Exp. Psychol. B38, 141–149.
113
Ross M. S. Flanagan L. B. Roi G. H. L. (1986). Seasonal and successional changes in light quality and quantity in the understory of boreal forest ecosystems. Can. J. Bot.64, 2792–2799. 10.1139/b86-373
114
Rowland H. M. Ihalainen E. Lindstrom L. Mappes J. Speed M. P. (2007). Co-mimics have a mutualistic relationship despite unequal defences. Nature448, 64–67. 10.1038/nature05899
115
Ruxton G. D. Sherratt T. N. Speed M. P. (2004). Avoiding Attack: the Evolutionary Ecology of Crypsis, Warning Signals and Mimicry.Oxford: Oxford University Press.
116
Rychlik L. (1999). Changes in prey size preferences during successive stages of foraging in the Mediterranean water shrew Neomys anomalus. Behaviour136, 345–365. 10.1163/156853999501360
117
Sandre S.-L. Stevens M. Mappes J. (2010). The effect of predator appetite, prey warning coloration and luminance on predator foraging decisions. Behaviour147, 1121–1143. 10.1163/000579510X507001
118
Sandre S.-L. Tammaru T. Mänd T. (2007). Size-dependent colouration in larvae of Orgyia antiqua (Lepidoptera: Lymantriidae): a trade-off between warning effect and detectability?Eur. J. Entomol.104, 745–752. 10.14411/eje.2007.095
119
Schulz S. Gross J. Hilker M. (1997). Origin of the defensive secretion of the leaf beetle Chrysomela lapponica. Tetrahedron53, 9203–9212. 10.1016/S0040-4020(97)00618-2
120
Selman B. J. (1985a). The evolutionary biology and taxonomy of the Australian Eucalyptus beetles. Entomography3, 451–454.
121
Selman B. J. (1985b). The use and significance of color in the separation of paropsine chrysomelid species. Entomography3, 477–479.
122
Selman B. J. (1994). The biology of the paropsine eucalyptus beetles of Australia, in Novel Aspects of the Biology of Chrysomelidae, eds JolivetP.CoxM. L.PetitpierreE. (Dordrecht: Kluwer Academic Publishers), 555–565.
123
Sharkey C. R. Fujimoto M. S. Lord N. P. Shin S. McKenna D. D. Suvorov A. et al . (2017). Overcoming the loss of blue sensitivity through opsin duplication in the largest animal group, beetles. Sci. Rep.7, 8. 10.1038/s41598-017-00061-7
124
Shurcliff K. S. (1980). Vegetation and bird community characteristics in an Australian arid mountain range. J. Arid Environ.3, 331–348.
125
Skelhorn J. Halpin C. G. Rowe C. (2016). Learning about aposematic prey. Behav. Ecol.27, 955–964. 10.1093/beheco/arw009
126
Smith K. E. Halpin C. G. Rowe C. (2016). The benefits of being toxic to deter predators depends on prey body size. Behav. Ecol.27, 1650–1655. 10.1093/beheco/arw086
127
Stephens D. W. Krebs J. R. (1986). Foraging Theory. Princeton, NJ: Princeton University Press.
128
Stevens M. (2007). Predator perception and the interrelation between different forms of protective coloration. Proc. Biol. Sci.274, 1457–1464. 10.1098/rspb.2007.0220
129
Stevens M. (2013). Sensory Ecology, Behaviour, and Evolution.Oxford: Oxford University Press.
130
Stevens M. Cuthill I. C. Windsor A. M. M. Walker H. J. (2006). Disruptive contrast in animal camouflage. Proc. Biol. Sci.273, 2433–2438. 10.1098/rspb.2006.3614
131
Stevens M. Parraga C. A. Cuthill I. C. Partridge J. C. Troscianko T. S. (2007). Using digital photography to study animal coloration. Biol. J. Linn. Soc.90, 211–237. 10.1111/j.1095-8312.2007.00725.x
132
Summers K. Clough M. E. (2001). The evolution of coloration and toxicity in the poison frog family (Dendrobatidae). Proc. Natl. Acad. Sci. U.S.A.98, 6227–6232. 10.1073/pnas.101134898
133
Svádová K. H. Exnerová A. Kopećková M. Štys P. (2010). Predator dependent mimetic complexes: do passerine birds avoid Central European red-and-black Heteroptera?Eur. J. Entomol.107, 349–355. 10.14411/eje.2010.044
134
Symonds M. R. E. Moussalli A. (2011). A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using Akaike's information criterion. Behav. Ecol. Sociobiol.65, 13–21. 10.1007/s00265-010-1037-6
135
Tan E. J. Reid C. A. M. Elgar M. A. (2016). Colour pattern variation affects predation in chrysomeline larvae. Anim. Behav.118, 3–10. 10.1016/j.anbehav.2016.05.019
136
Termonia A. Pasteels J. M. Windsor D. M. Milinkovitch M. C. (2002). Dual chemical sequestration: a key mechanism in transitions among ecological specialization. Proc. Biol. Sci.269, 1–6. 10.1098/rspb.2001.1859
137
Thomas R. J. Marples N. M. Cuthill I. C. Takahashi M. Gibson E. A. (2003). Dietary conservatism may facilitate the initial evolution of aposematism. Oikos101, 458–466. 10.1034/j.1600-0706.2003.12061.x
138
Tischler M. Dickman C. R. Wardle G. M. (2013). Avian functional group responses to rainfall across four vegetation types in the Simpson Desert, central Australia. Austral. Ecol.38, 809–819. 10.1111/aec.12065
139
Tullberg B. S. Merilaita S. Wiklund C. (2005). Aposematism and crypsis combined as a result of distance dependence: functional versatility of the colour pattern in the swallowtail butterfly larva. Proc. Biol. Sci.272, 1315–1321. 10.1098/rspb.2005.3079
140
Utne-Palm A. C. (2000). Prey visibility, activity, size and catchability's (evasiveness) influence on Gobiusculus flavescens prey choice. Sarsia85, 157–165. 10.1080/00364827.2000.10414565
141
Uy F. M. K. Ravichandran S. Patel K. S. Aresty J. Aresty P. P. Audett R. M. et al . (2017). Active background choice facilitates crypsis in a tropical crab. Biotropica49, 365–371. 10.1111/btp.12429
142
Wee J. L. Q. Monteiro A. (2017). Yellow and the novel aposematic signal, red, protect delias butterflies from predators. PLoS ONE12:e0168243. 10.1371/journal.pone.0168243
143
Wilts B. D. Michielsen K. Kuipers J. De Raedt H. Stavenga D. G. (2012). Brilliant camouflage: photonic crystals in the diamond weevil, Entimus imperialis. Proc. Biol. Sci.279, 2524–2530. 10.1098/rspb.2011.2651
144
Woinarski J. C. Z. Cullen J. M. (1984). Distribution of invertebrates on foliage in forests of south-eastern Australia. Aust. J. Ecol.9, 207–232. 10.1111/j.1442-9993.1984.tb01359.x
145
Zurek D. B. Cronin T. W. Taylor L. A. Byrne K. Sullivan M. L. G. Morehouse N. I. (2015). Spectral filtering enables trichromatic vision in colorful jumping spiders. Curr. Biol.25, R403–R404. 10.1016/j.cub.2015.03.033
146
Zylinski S. How M. J. Osorio D. Hanlon R. T. Marshall N. J. (2011). To be seen or to hide: visual characteristics of body patterns for camouflage and communication in the Australian giant cuttlefish Sepia apama. Am. Nat.177, 681–690. 10.1086/659626
Summary
Keywords
color pattern, anti-predator strategies, phylogenetic comparative methods, signaling conflicts, chrysomelines
Citation
Tan EJ, Reid CAM, Symonds MRE, Jurado-Rivera JA and Elgar MA (2017) The Role of Life-History and Ecology in the Evolution of Color Patterns in Australian Chrysomeline Beetles. Front. Ecol. Evol. 5:140. doi: 10.3389/fevo.2017.00140
Received
14 July 2017
Accepted
31 October 2017
Published
14 November 2017
Volume
5 - 2017
Edited by
Wayne Iwan Lee Davies, University of Western Australia, Australia
Reviewed by
Floria Mora-Kepfer Uy, University of Miami, United States; Sean John Blamires, University of New South Wales, Australia
Updates
Copyright
© 2017 Tan, Reid, Symonds, Jurado-Rivera and Elgar.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mark A. Elgar m.elgar@unimelb.edu.au
†Present Address: Eunice J. Tan, Division of Science, Yale-NUS College, Singapore, Singapore
This article was submitted to Behavioral and Evolutionary Ecology, a section of the journal Frontiers in Ecology and Evolution
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.