- 1International Bird Conservation Partnership, Monterey, CA, United States
- 2Department of Ecology, Swedish University of Agricultural Sciences, Uppsala, Sweden
- 3Crane Trust, Wood River, NE, United States
- 4Cooperative Wildlife Research Laboratory, Southern Illinois University, Carbondale, IL, United States
- 5U.S. Fish and Wildlife Service, Pierre, SD, United States
- 6Center for Great Plains Studies, University of Nebraska–Lincoln, Lincoln, NE, United States
Few empirical studies have quantified relationships between changing weather and migratory songbirds, but such studies are vital in a time of rapid climate change. Climate change has critical consequences for avian breeding ecology, geographic ranges, and migration phenology. Changing precipitation and temperature patterns affect habitat, food resources, and other aspects of birds’ life history strategies. Such changes may disproportionately affect species confined to rare or declining ecosystems, such as temperate grasslands, which are among the most altered and endangered ecosystems globally. We examined the influence of changing weather on the dickcissel (Spiza americana), a migratory songbird of conservation concern that is an obligate grassland specialist. Our study area in the North American Great Plains features high historic weather variability, where climate change is now driving higher precipitation and temperatures as well as higher frequencies of extreme weather events including flooding and droughts. Dickcissels share their breeding grounds with brown-headed cowbirds (Molothrus ater), brood parasites that lay their eggs in the nests of other songbirds, reducing dickcissel productivity. We used 9 years of capture-recapture data collected over an 18-year period to test the hypothesis that increasing precipitation on dickcissels’ riparian breeding grounds is associated with abundance declines and increasing vulnerability to cowbird parasitism. Dickcissels declined with increasing June precipitation, whereas cowbirds, by contrast, increased. Dickcissel productivity appeared to be extremely low, with a 3:1 ratio of breeding male to female dickcissels likely undermining reproductive success. Our findings suggest that increasing precipitation predicted by climate change models in this region may drive future declines of dickcissels and other songbirds. Drivers of these declines may include habitat and food resource loss related to flooding and higher frequency precipitation events as well as increased parasitism pressure by cowbirds. Positive correlations of June-July precipitation, temperature, and time since grazing with dickcissel productivity did not mitigate dickcissels’ declining trend in this ecosystem. These findings highlight the importance of empirical research on the effects of increasing precipitation and brood parasitism vulnerability on migratory songbird conservation to inform adaptive management under climate change.
Introduction
Global climate change is influencing birds’ breeding ecology, geographic ranges, and migration phenology. Changing precipitation and temperature patterns affect birds directly as well as indirectly through their impacts on habitat, food resources, and other factors critical to avian life history strategies and population dynamics (Moss et al., 2001; Cotton, 2003; Niemuth et al., 2008; Knudsen et al., 2011; McDonald et al., 2012; López-Calderón et al., 2019). For example, precipitation declines on the breeding grounds of yellow-headed blackbirds (Xanthocephalus xanthocephalus) have been correlated with density declines and reproductive failure (Fletcher and Koford, 2004). For burrowing owls (Athene cunicularia), precipitation declines and higher temperatures on the breeding grounds were associated with delayed arrival dates and lower abundance and productivity, resulting in population collapse (Cruz-McDonnell and Wolf, 2016). For whooping cranes (Grus americana), precipitation declines and higher temperatures on the breeding grounds are predicted to drive declines in juvenile recruitment by increasing their vulnerability to mammalian nest predators, driving population-wide declines (Butler et al., 2017). In addition to the influence of changing precipitation and temperature on the breeding grounds, climatic conditions on birds’ migration routes and wintering grounds may also influence bird population dynamics, including through carry-over effects that exert influence in multiple seasons (Marra et al., 1998; Finch et al., 2014; O’Connor et al., 2014; Akresh et al., 2019). Climate change effects may vary considerably within and across geographic regions (e.g., USGCRP, 2018), carrying the potential to affect local bird populations differently across geographic gradients (e.g., Jensen and Cully, 2005).
Climate change may disproportionately affect species restricted to rare or declining ecosystems, such as temperate grasslands, which are among the most endangered and least protected ecosystems worldwide (White et al., 2000; Blancher, 2003). In North America, >80% of temperate grasslands have been converted to agriculture and other human uses (With et al., 2008), driving bird declines (Pietz and Granfors, 2000; Brennan and Kuvlesky, 2005; Stanton et al., 2018). North American grassland birds have declined >50% in abundance in the past 50 years (Askins et al., 2007; Sauer et al., 2012; Rosenberg et al., 2019), warranting urgent conservation attention. Few empirical studies have examined impacts of weather on grassland bird populations (Amburgey et al., 2018; Kleinhesselink and Adler, 2018; Scridel et al., 2018). However, some research to date has identified positive correlations between precipitation levels and grassland bird abundance, productivity, and recruitment (e.g., Patterson and Best, 1996; Rahmig et al., 2009). For example, winter grassland songbird density in desert grasslands in Mexico has been positively correlated with precipitation the preceding year, which in turn was associated with higher grass productivity (Macías-Duarte et al., 2009). In riparian grasslands, however, while increased precipitation may be associated with increased habitat quality in uplands, it may also be correlated with local flooding and lower bird abundance (Kim et al., 2008; Glass et al., 2020), lower arthropod food availability (Siikamäki, 1996; Plum, 2005; Sienkiewicz and Żmihorski, 2012), and lower productivity (Skagen and Yackel Adams, 2012; Jarzyna et al., 2016).
Far removed from the ocean’s moderating effects on weather, the North American Great Plains are prone to dramatic climate variability (Skagen and Yackel Adams, 2012; Conant et al., 2018; USGCRP, 2018), making this an ideal region in which to explore the effects of weather on grassland birds. The “climate bottleneck hypothesis” predicts that precipitation extremes should not influence grassland breeding bird abundance as long as grasslands retain sufficient structural complexity because obligate grassland birds have evolved to tolerate extreme weather events (Wiens, 1974; Zimmerman, 1992). A 10-year study found support for this hypothesis, showing that obligate grassland bird abundance did not change in response to precipitation levels during a period when extreme weather events such as floods or droughts occurred approximately once every 4 years (Zimmerman, 1992). In riparian grasslands, however, which are drought-resistant but flood-prone, studies have found higher levels of precipitation correlated with lower local abundances of grassland birds (Kim et al., 2008; Glass et al., 2020). Climate change is introducing unprecedented variability in extreme weather events in the eastern Great Plains, with expected increases in the number of days with heavy (>2.5 cm) precipitation and an increasing number (15–40) of very hot (>32°C) days (USGCRP, 2018). Examining how precipitation and temperature influence grassland breeding bird populations in this region is crucial for advancing our understanding of their ecology and informing conservation efforts under future climate change.
Obligate grassland specialists, dickcissels (Spiza americana) are sensitive to temperature and moisture changes throughout the annual cycle, making them excellent indicators of weather and climate change effects on grassland bird populations (DeSante et al., 2015; Culp et al., 2017). Neotropical migratory birds, dickcissels’ core nesting grounds are in the North American Great Plains and their core wintering grounds are in Venezuela (Sauer et al., 2005; Temple, 2020). Dickcissels underwent severe declines in the mid-20th century and have not recovered their previous numbers (Temple, 2020). Recent (1992–2006) data from both the North American Breeding Bird Survey (BBS) and the Monitoring Avian Productivity and Survivorship (MAPS) program showed dickcissel populations as stable, albeit with a non-significant declining trend (DeSante et al., 2015). However, dickcissels remain highly vulnerable to extinction due to habitat loss and other anthropogenic impacts that have already reduced and fragmented their populations (Culp et al., 2017). Dickcissel nest success tends to be low, with past studies reporting that 29–44% of nesting females produced only ∼1 fledgling/season (Zimmerman, 1982; Basili et al., 1997). Females take sole responsibility for nest building, incubation, and caring for young (Temple, 2020). Research priorities for dickcissels include investigating potential drivers of their low productivity and low adult survivorship (DeSante et al., 2015). Breeding females build bulky cup nests in dense, tall grasslands with scattered forbs; females typically place nests low (22–46 cm aboveground) in dense vegetation with overhead cover, but occasionally may build nests up to 4 m high in trees (Temple, 2020). Common grassland management strategies such as grazing, haying (mowing), and controlled burning prevent woody encroachment in the long term, but in the short term they simplify grassland structure, which may reduce dickcissels’ habitat quality and ability to adjust to fluctuating climatic conditions (Zimmerman, 1992; Travis, 2003; Jarzyna et al., 2016). Since dickcissel nests are often attached loosely to supporting vegetation (Long et al., 1965; Gross, 1968), they are vulnerable to being knocked down during heavy precipitation events, which can cause loss of eggs and nestlings and reduce dickcissel breeding success (Zimmerman, 1966).
Obligate brood parasites, brown-headed cowbirds (Molothrus ater) lay their eggs in the nests of >220 avian host species rather than building their own nests (Lowther, 2020). In transferring the costs of raising their young to their hosts, cowbirds are entirely dependent on their hosts for their reproductive success (Smith et al., 2000; Croston and Hauber, 2010). Cowbirds remove eggs from nests they parasitize, reducing their hosts’ nesting success, and are known to intensely parasitize dickcissels (Jensen and Cully, 2005; Temple, 2020). Studies have estimated that cowbird parasitism affects 48–90% of dickcissel nests (Basili et al., 1997; Temple, 2020) and significantly reduces dickcissel productivity (Zimmerman, 1983; Jensen and Cully, 2005). Before European settlement of the Great Plains, cowbirds followed migratory herds of American bison (Bison bison), and their impacts on their hosts during the breeding season were limited by their ephemeral presence at any given location. The extirpation of migratory bison and subsequent practice of confining sedentary livestock into fenced areas has transformed these patterns such that cowbirds now tend to concentrate in high densities where they remain throughout the breeding season. Their rates of parasitism on their hosts are correlated with their densities (Jensen and Cully, 2005). Birds with remarkable memories (Guigueno et al., 2014), female cowbirds may learn the whereabouts of most or all nests in their vicinity and parasitize them repeatedly. Previous research has suggested that cowbird parasitism may increase as dickcissel breeding densities decline, because the remaining hosts bear increasing pressure from the relatively higher proportion of cowbirds and their eggs (Zimmerman, 1982). The resulting pattern of inverse density dependence may further reduce dickcissel nest success and productivity (Temple, 2020). Few empirical studies to date have addressed whether or how weather influences cowbirds’ interactions with their hosts (Colón et al., 2017; Buxton et al., 2018). Improving conservation strategies for dickcissels will include an increased understanding of the effects of weather and climate change on cowbird populations (DeSante et al., 2015). Dickcissels’ particular vulnerability to cowbird parasitism also makes their populations valuable indicators of relationships with cowbirds and weather.
We took advantage of a rare opportunity to use 9 years of capture-recapture data collected systematically over an 18-year period to investigate whether dickcissel and cowbird abundance and productivity changed in relation to changes in weather in the eastern Great Plains. We quantified changes in dickcissel abundance and productivity in response to changes in precipitation and temperature as well as grassland management including grazing by cattle (Bos taurus) and bison, haying, and burning. We expected moderate levels of precipitation to benefit birds in this riparian ecosystem by increasing primary productivity and grassland structural complexity. However, we expected increasing precipitation to be associated with an increased number of heavy precipitation days and flooding that would in turn negatively affect dickcissels’ abundance and productivity, including by reducing habitat and food resource availability. Cowbirds, on the other hand, are not known to decline in response to higher precipitation and therefore may be resilient to the negative effects it has on dickcissels and other grassland songbirds. Thus, we hypothesized that increases in precipitation would drive dickcissel declines and increase their vulnerability to cowbird parasitism.
Materials and Methods
Study Area
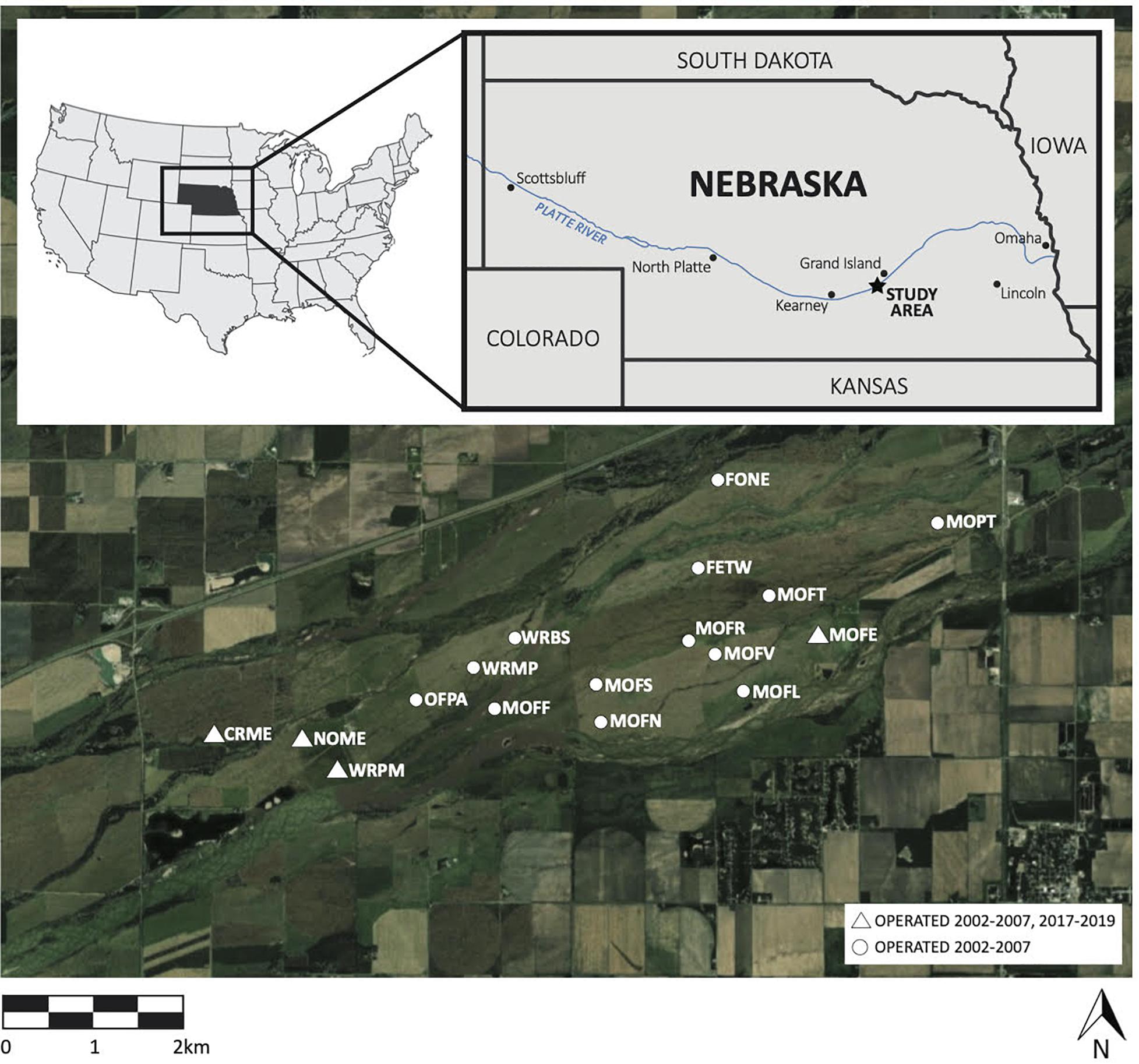
We sampled birds at 17 sites in a 2430 ha private conservation area comprised of riparian upland prairies and wet meadows in Nebraska’s Platte River Valley (Figure 1), where much of the surrounding land has been converted to industrial-scale agriculture, especially corn (Zea mays). In 1978, this fragment of the critically endangered North American Central and Southern Mixed Grasslands ecoregion was designated as protected habitat for the conservation of whooping cranes, sandhill cranes (Antigone canadensis), and other migratory birds. These grasslands comprise disturbance-dependent ecosystems (e.g., Glass et al., 2020). Ephemeral disturbances such as bison grazing, seasonal flooding, and wildfires historically structured this region, controlling woody encroachment and maintaining early successional vegetation (Williams, 1978; Anderson, 2006). The Crane Trust, a non-profit organization, now maintains these grasslands through managed disturbances including grazing, haying, and burning, which control woody encroachment. Cattle, brought in seasonally, were the sole grazers until 2015, when a semi-domesticated bison herd was introduced year-round to part of our study area (King et al., 2019). Cattle and bison grazing often overlap with the songbird breeding season, while haying and burning typically take place during the non-breeding season, in the fall and spring, respectively.
Bird Sampling
We used protocols standardized by the MAPS program (DeSante and Kaschube, 2009; DeSante et al., 2017) to sample birds using constant-effort mist-netting at ∼20 ha stations (hereafter, sites) during the breeding season within the 80-day period from late May to early August. Between 2002 and 2007, we sampled birds at 17 sites, of which 3 were sampled for 6 consecutive years and 14 were sampled for periods ranging between 1 and 5 years. From 2017 to 2019, we replicated bird sampling at 4 sites used in 2002–2007 with different management histories for 3 consecutive years. Standardized in 1992, the MAPS program includes more than 1000 bird banding (ringing) stations (DeSante et al., 2015) that provide capture-recapture data from mist net sampling to assess avian population parameters and vital rates (DeSante et al., 2015; Foster et al., 2016; Saracco et al., 2016, 2019; Ahrestani et al., 2017; Glass et al., 2020). We identified captured birds to species and fitted them with a uniquely numbered aluminum band (ring) issued by the USGS Bird Banding Laboratory. We recorded demographic information and biometric measurements and then released birds at the site of capture (Pyle, 1997). We deployed 10–12 mist nets (12 × 3 m, 30 mm mesh) within a ∼8 ha area at each site (DeSante et al., 2017); we used 12 mist nets during the 2002–2007 data collection period and 10 mist nets during the 2017–2019 data collection period. Each sampling event consisted of a capture period of 6 h following sunrise (∼0600) and operated approximately once every 10 days. Effort was consistent for each sampling event, except when nets were occasionally closed during periods of high winds, heavy precipitation, or lightning storms, in which case we compensated by adding equal effort to a subsequent sampling occasion.
Capture-recapture sampling is a powerful tool for estimating animal abundance, and novel approaches to capture-recapture analyses are increasingly used to estimate abundance of open-population sizes from mark-recapture data on animals (Williams et al., 2002; Manly et al., 2003; Amstrup et al., 2005; Thomson et al., 2009; Gopalaswamy et al., 2012), expanding earlier uses of capture-recapture analyses that historically focused on estimating survival (Williams et al., 2002; Barker et al., 2004). Habitat types, seasons, and species’ behavior and life histories may influence mist net capture rates (Remsen and Good, 1996; DeSante et al., 2015; Martin et al., 2017). Bearing in mind these caveats, appropriate use of mist net capture-recapture data allows us to standardize quantitative data on bird populations, including information on birds’ sex, age, and biometrics, enabling the analysis of demographic parameters to generate insight into ecological processes.
We used MAPS protocols to collect data on songbirds during their breeding season. During this time, both male and female dickcissels make frequent, short flights (Temple, 2020) that facilitate their capture using mist nets (Remsen and Good, 1996). DeSante et al. (2015) analyzed dickcissel demographric data collected through the MAPS program between 1992 and 2006, a period that overlaps with our study period, providing a useful point of reference. Here, we follow DeSante et al. (2015) in quantifying avian productivity through our captures of post-fledging juvenile (hatch-year) birds. Productivity is influenced by the proportion of breeding adults, clutch size, nest survival, and nestling and juvenile survival after independence (DeSante et al., 2015) as well as cowbird parasitism (Temple, 2020). Previous research has indicated that dickcissel juveniles can disperse at least 600 m from nests within 10 days after fledging (Gross, 1921; Temple, 2020). We captured dickcissel juveniles still dependent on their mothers (i.e., adult females and juveniles were captured together) as well as dispersing juveniles that were independent.
Weather and Climate Parameters
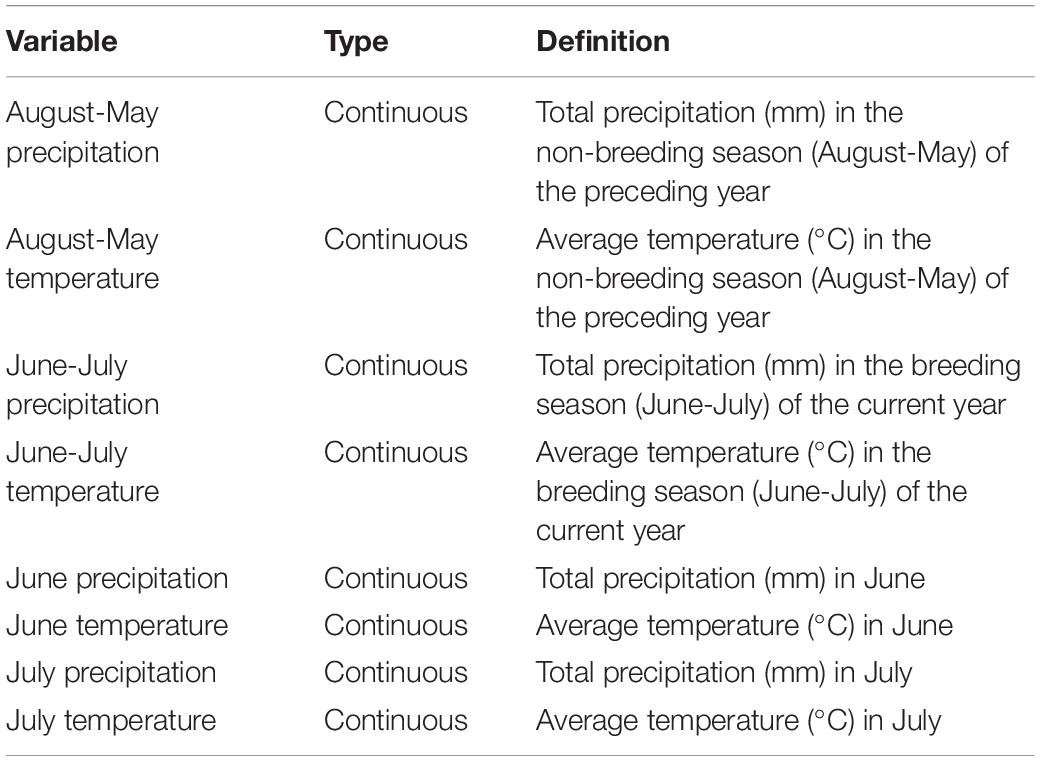
We obtained weather data from the National Oceanic and Atmospheric Administration (NOAA, 2019) online database for South-Central Nebraska (station KGRI, 40.968°N, −98.340°W, Central Nebraska Regional Airport in Grand Island, Nebraska; approximately 21 km northeast of our research site). We created eight variables (Table 1) representing total precipitation (mm) and average temperature (°C) over different temporal scales. We used total precipitation, as provided in NOAA’s database, as a measurement standard for modeling, as it accurately represents differences in monthly precipitation levels. We calculated total precipitation and average temperature for the main migratory songbird breeding season (June-July) of the current year, when precipitation and temperature directly affect breeding birds and their productivity. We also calculated total precipitation and average temperature for the previous non-breeding season (August-May), when precipitation and average temperature indirectly affect breeding birds and productivity through their effects on habitat, plant phenology, food resources, predators, and other factors. We also examined temperature and precipitation variables for June and July separately to determine whether conditions during either month of the breeding season affected dickcissels differently.
Land Management and Habitat Parameters
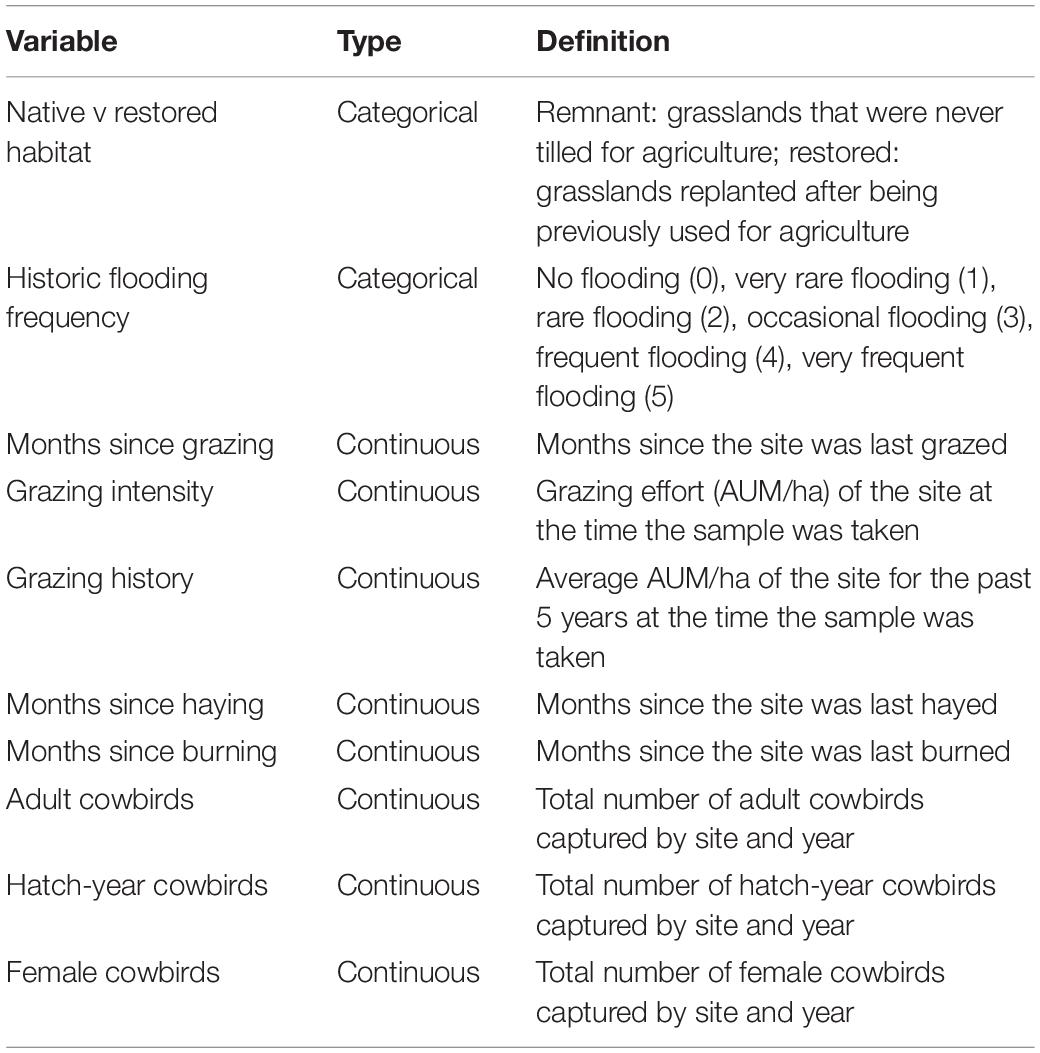
We created seven habitat and land management parameters (Table 2). Three variables represent time (months) since managed disturbances (grazing, haying, and burning) at sampling sites. We set the maximum time since disturbance value for grazed, hayed, and burned pastures at 180 months (15 years) because management actions that occurred earlier were unlikely to have a predictable effect on habitat structure (Collins, 2000). In addition, we created two management parameters (grazing intensity and grazing history) representing livestock (cattle or bison) stocking rate for grazed pastures in animal unit months per hectare (AUM/ha), where one AUM equals the forage requirement for one adult and calf pair for a 1-month period (Hamilton, 2007; Johnson et al., 2011). Grazing intensity is represented by AUM/ha at each study site at the time of data collection; a value of 0 represents sites that had no grazing during MAPS data sampling. Grazing history is represented by the average AUM/ha at each site for the current year plus the 4 years preceding data collection (Glass et al., 2020). In addition, we created a categorical variable to distinguish native remnant grasslands with no major disturbance history from restored grasslands on land previously used for row agriculture or planted with exotic grasses. We created another categorical variable representing historic flooding frequency by averaging flooding frequency of soil (none, very rare, rare, occasional, frequent, very frequent) within the sampling footprint as indicated by USDA soil maps. When this footprint included over three soil types, we averaged the top three values.
Statistical Analyses
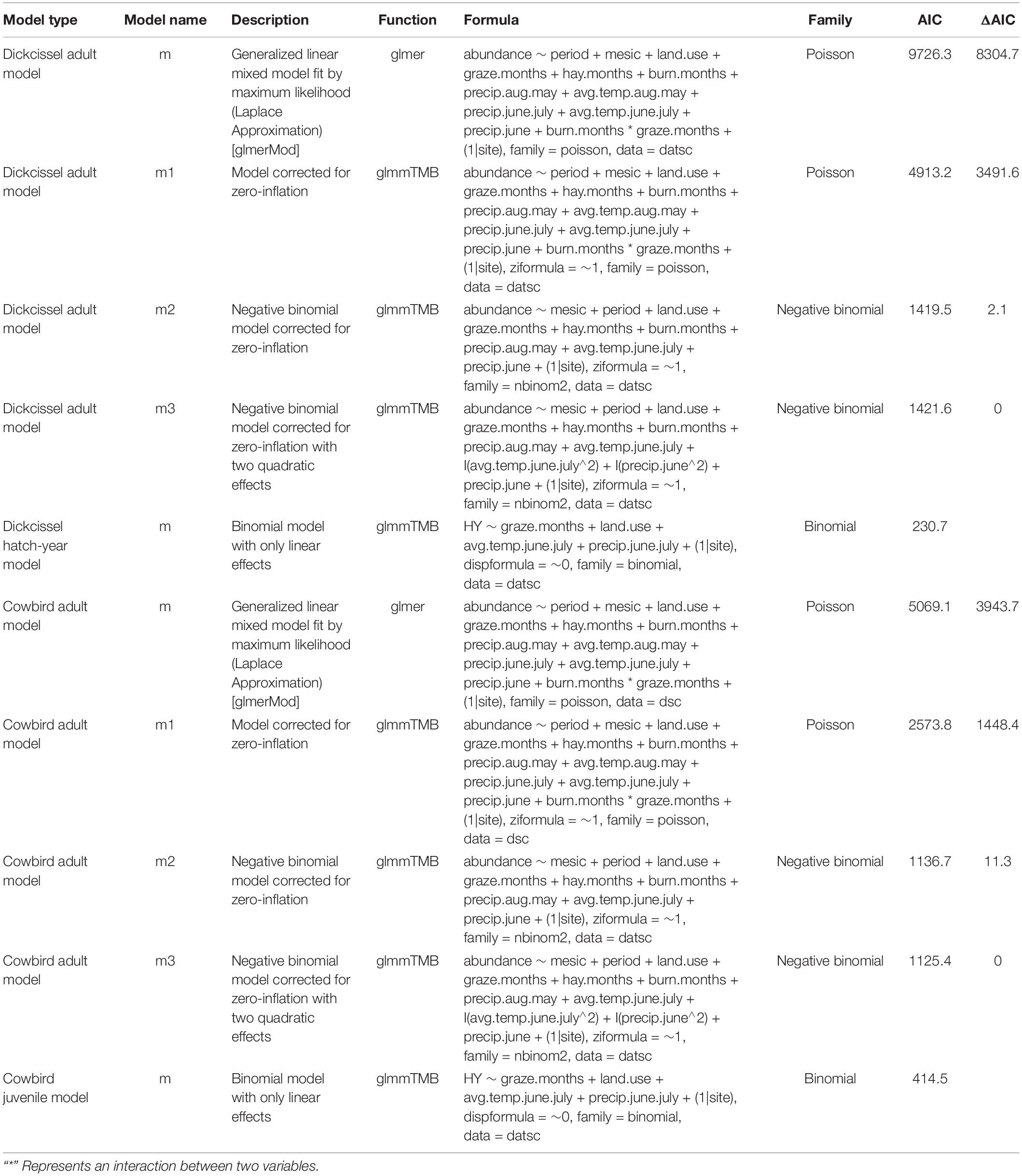
We conducted all statistical analyses using R Studio version 3.6.2 (R Core Team, 2019). We used dickcissel capture data across all 17 sites, removing recaptures of individuals that occurred at the same site during the same year to avoid double-counting. In addition to quantifying dickcissel numbers, we used the same methods to quantify adult, juvenile, and female cowbirds in all sites and all years sampled. We corrected for effort by dividing the number of captures by the cumulative mist net-meter-hours for the given year. We divided the banding season into two halves for data analysis, in which the first half comprised adults arriving and establishing breeding territories and nests, and the second half comprised both adults and juvenile birds that had fledged from successful nests (Glass et al., 2020). The first half of the banding season included MAPS periods 3–6 (May 21-June 29), and the second half included MAPS periods 7–10 (June 30-August 8) (DeSante et al., 2017).
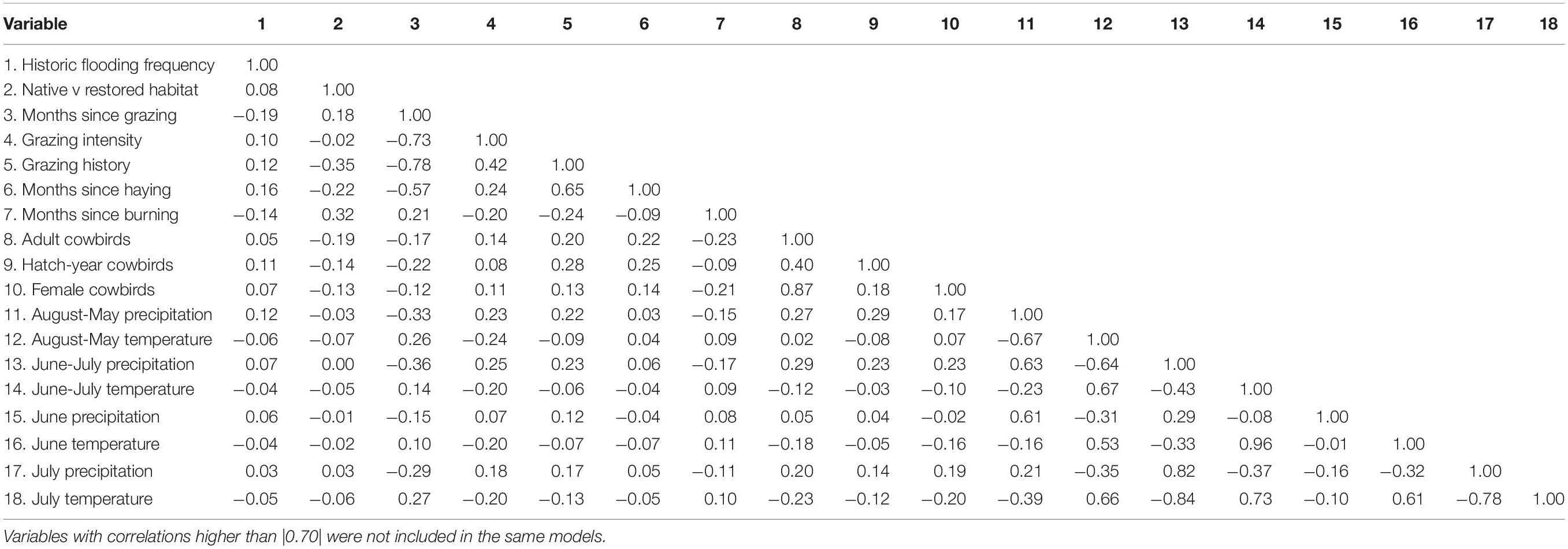
Pastures were often grazed for the first or second half of the summer, such that in some cases values for variables such as time (months) since grazing and current stocking rate changed for a given pasture within a banding season. We tested for correlations between climate and management parameters with a Spearman’s correlation test to account for the non-normal distribution of parameters, using the package psych (Revelle, 2018). We used the uncorrelated variables (r < |0.7|; Table 3) to perform two independent generalized linear mixed models (GLMMS), one for adult and another for juvenile dickcissels. We log-transformed all of the continuous parameters prior to analysis for better convergence. After analyzing different possible combinations including linear, quadratic, and interaction terms, we used Akaike’s Information Criterion (AIC) to select the models that best fit the data (Table 4). For analysis of adults, we used a negative binomial distribution to account for overdispersion of the data using the package glmmTMB (Brooks et al., 2017). The final model includes both linear and quadratic terms for temperature and precipitation as fixed factors, and site as a random factor to account for non-independent observations within each site. We also used the package glmmTMB to run models for juvenile dickcissels after we converted these data into a binomial response with a binomial distribution.
We modeled annual variation in captured dickcissels and cowbirds using GLMMs. For each species, we developed two models. The first model included the number of adults captured, an index of adult abundance (hereafter, abundance) as a response variable. The second model included whether a captured bird was a hatch-year bird (hereafter, productivity) as a response variable (DeSante et al., 2015; Foster et al., 2016; Glass et al., 2020). We created a binomial index to quantify productivity, assigning juvenile birds a value of 1 and adults a value of 0. Lastly, we ran a GLMM with adult cowbird abundance as a response variable and a GLMM with cowbird productivity as a response variable, using the same methods described above for dickcissel abundance and productivity, respectively.
We also ran a principal component analysis (PCA) to create a set of uncorrelated climate and management parameters. From the 10 original variables introduced by the PCA, we selected the first three principal components, which together explained 64.5% of the total variation. We performed the same analyses as above with the principal components and the three categorical variables with site as a random factor to check for consistency in our results. As our results were consistent with both approaches, we modeled the uncorrelated variables independently to allow us to interpret each variable using non-linear effects not shown by the PCA.
Results
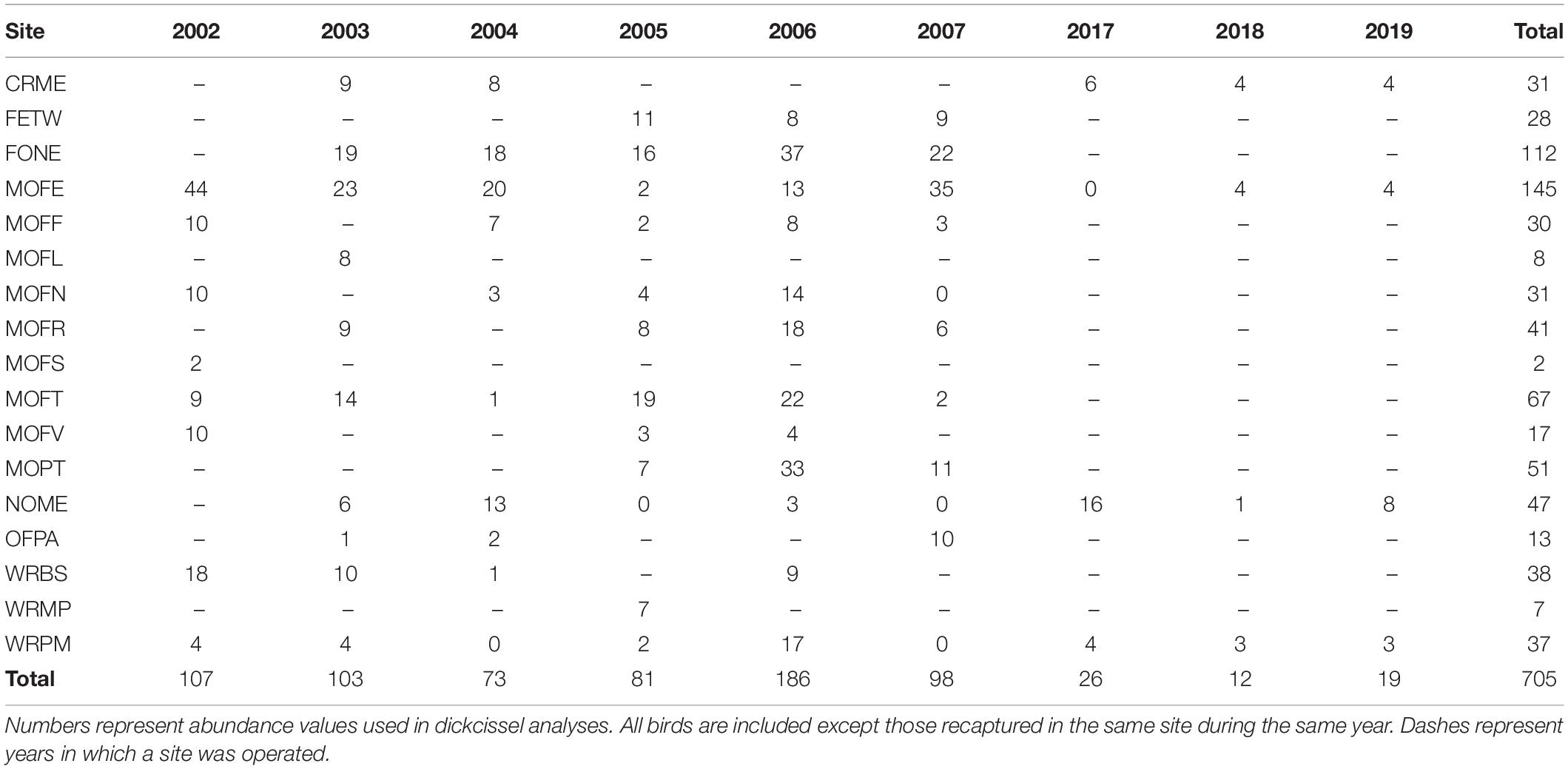
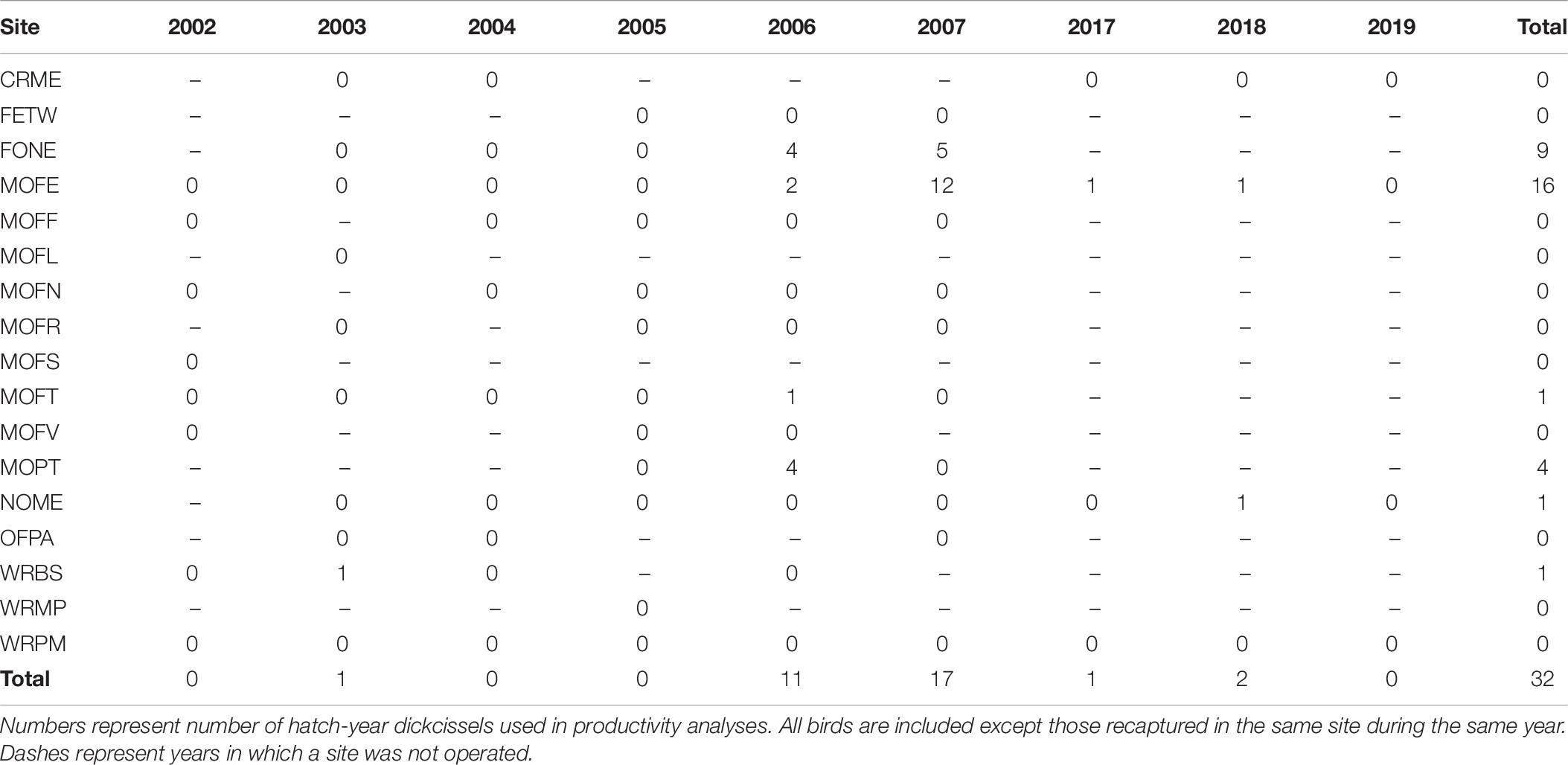
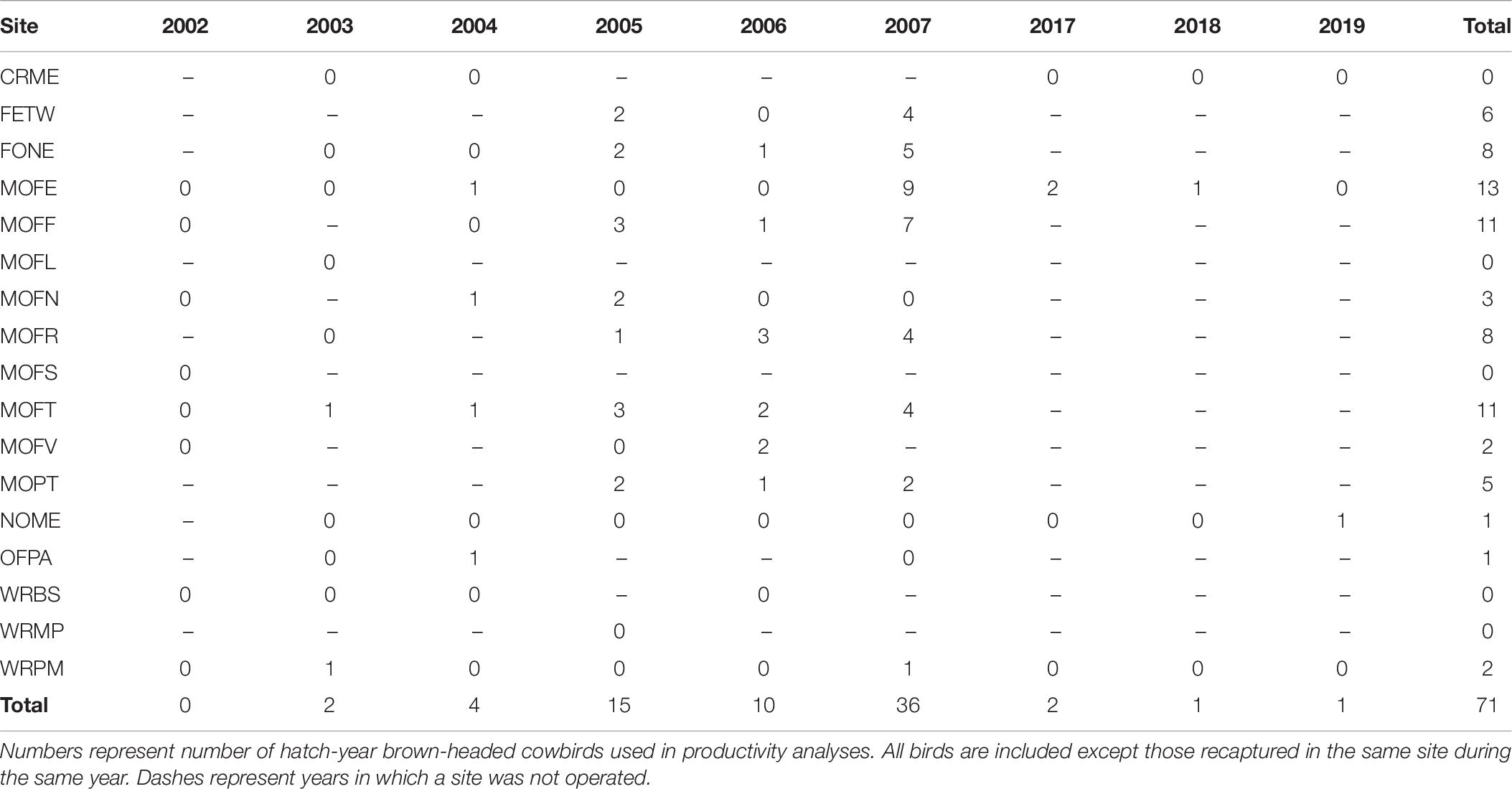
Between 2002 and 2019, we captured a total of 737 dickcissels (Tables 5, 6), excluding recaptures from the same year and site. The number of adult dickcissels captured and banded per year ranged from 12 from four active sites in 2018 to 186 from 12 active sites in 2006 (mean = 78.3 ± 18.3 SE). Of adult dickcissels captured, 76% (n = 528) were male and 24% (n = 165) were female (the sex of 12 adult dickcissels was not recorded). We recaptured 50 adult dickcissels. Of same-year recaptures, 93% were at the same site as their initial capture and the remainder at adjacent sites within <10 km. Of subsequent-year recaptures, 70% were at the same site as their initial capture and the remainder at sites within <10 km. Juveniles (hatch-year birds) made up 4% of total dickcissel captures, which was the equivalent of one juvenile for every five adult female dickcissels. Juvenile numbers per year ranged from 0 in 2002, 2004, 2005, and 2019 to 17 in 2007 (mean = 3.6 ± 2.0 SE). We captured half of all dickcissel juveniles at a single site (MOFE) and over half in a single year (2007; Table 6).
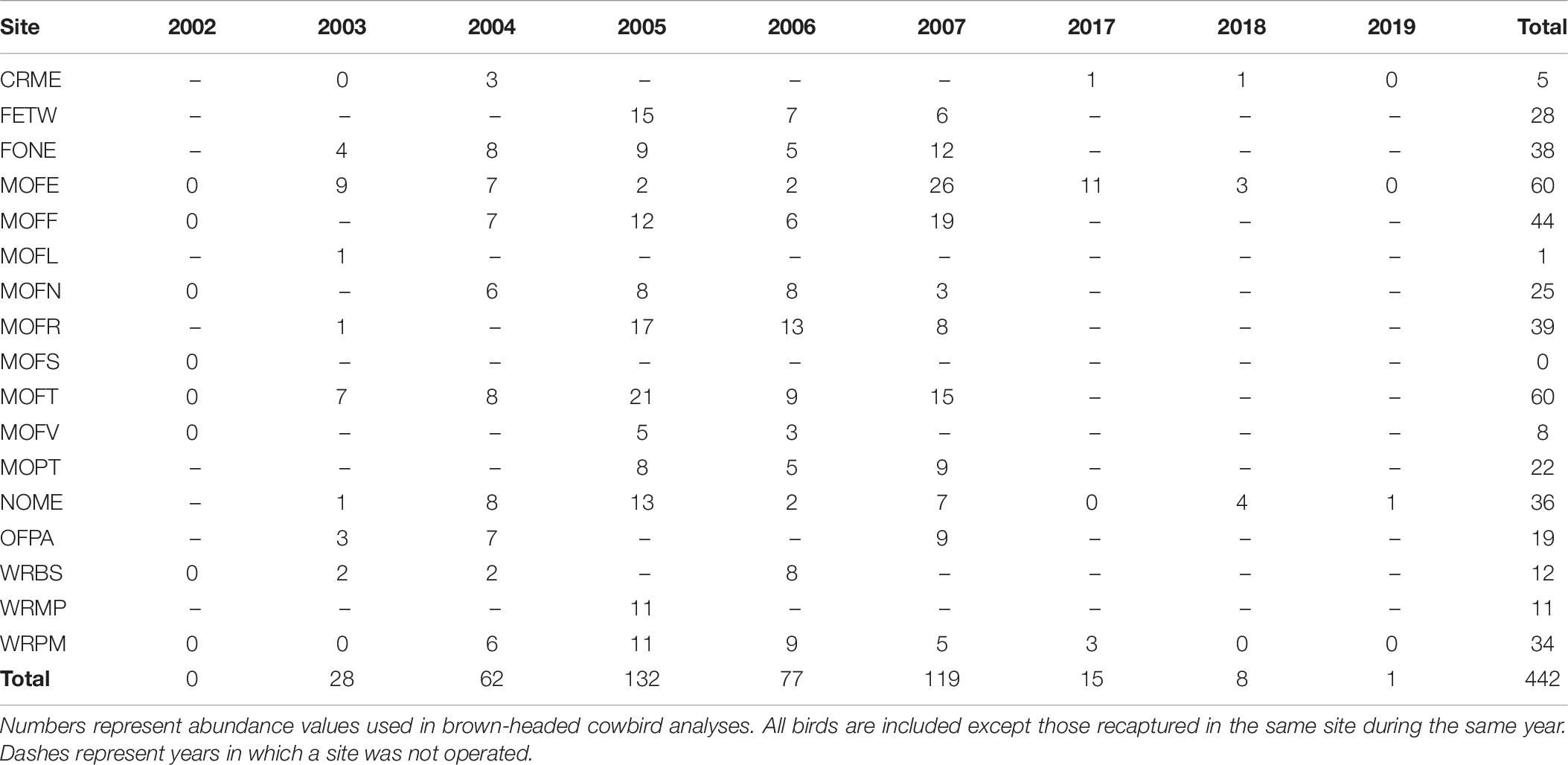
We found a weak positive correlation between numbers of dickcissels and cowbirds, for both abundance (Spearman’s rank correlation test: rho = 0.237, p = 0.004) and productivity (rho = 0.294, p = 0.0002). Between 2002 and 2019, we captured a total of 442 adult cowbirds (Table 7), excluding recaptures from the same year and site. The number of adult cowbirds captured and banded per year ranged from 0 in 2002 to 132 in 2005 (mean = 49.1 ± 17.0 SE). Of adult cowbirds we banded, 49% (n = 206) were female and 51% (n = 199) were male (the sex of 37 cowbirds was not recorded). Juveniles represented 14% of our total cowbird captures (Table 8), which was the equivalent of one juvenile for every three adult female cowbirds. For both species, we made more captures of adult birds during the first half of the breeding season, when new arrivals were establishing territories and breeding (dickcissels: Z = 5.09, p = 3.54e-07; cowbirds: Z = 7.93, p = 2.21e-15; Table 9). This pattern in part reflects trap-shyness of birds that avoided nets after being trapped and banded although they remained on site (Simons et al., 2015).
Dickcissel Responses to Weather, Land Management, and Habitat Parameters
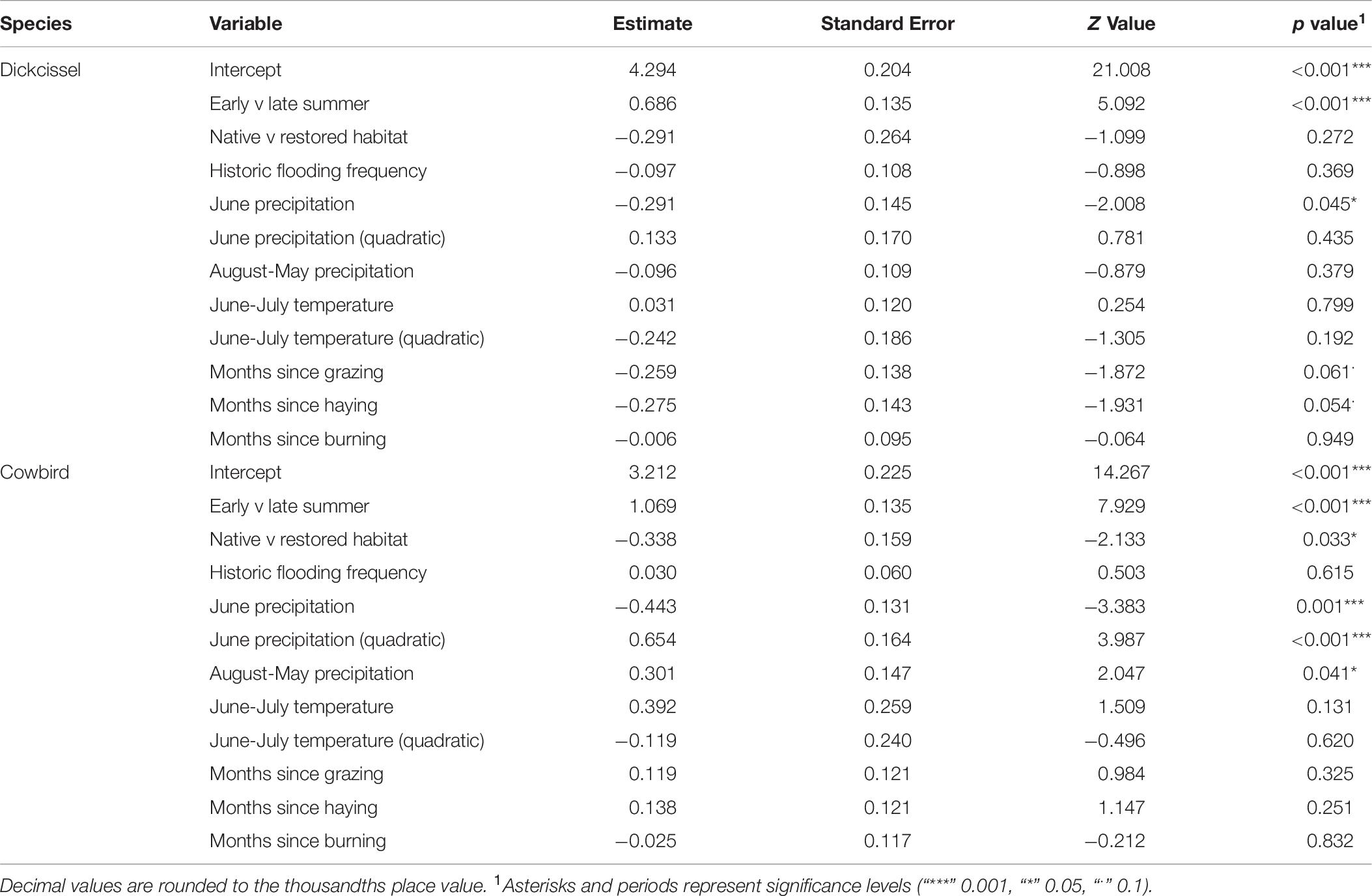
Dickcissel abundance was negatively correlated with June precipitation (Z = 2.01, p = 0.04; Table 9 and Figure 2). Of the parameters we tested, June precipitation was the only significant predictor of dickcissel abundance, although we found near-significant positive correlations with both time since grazing (graze months: p = 0.061) and time since haying (hay months: p = 0.054), indicating that dickcissel abundance may increase with increasing time since grazing and haying for at least 15 years (>180 months) in this ecosystem. Dickcissel productivity was positively correlated with June-July precipitation (Z = 4.156, p = 3.23e-05), June-July temperature (Z = 2.622, p = 0.009), and months since grazing (Z = 2.236, p = 0.03; Table 10 and Figure 3).
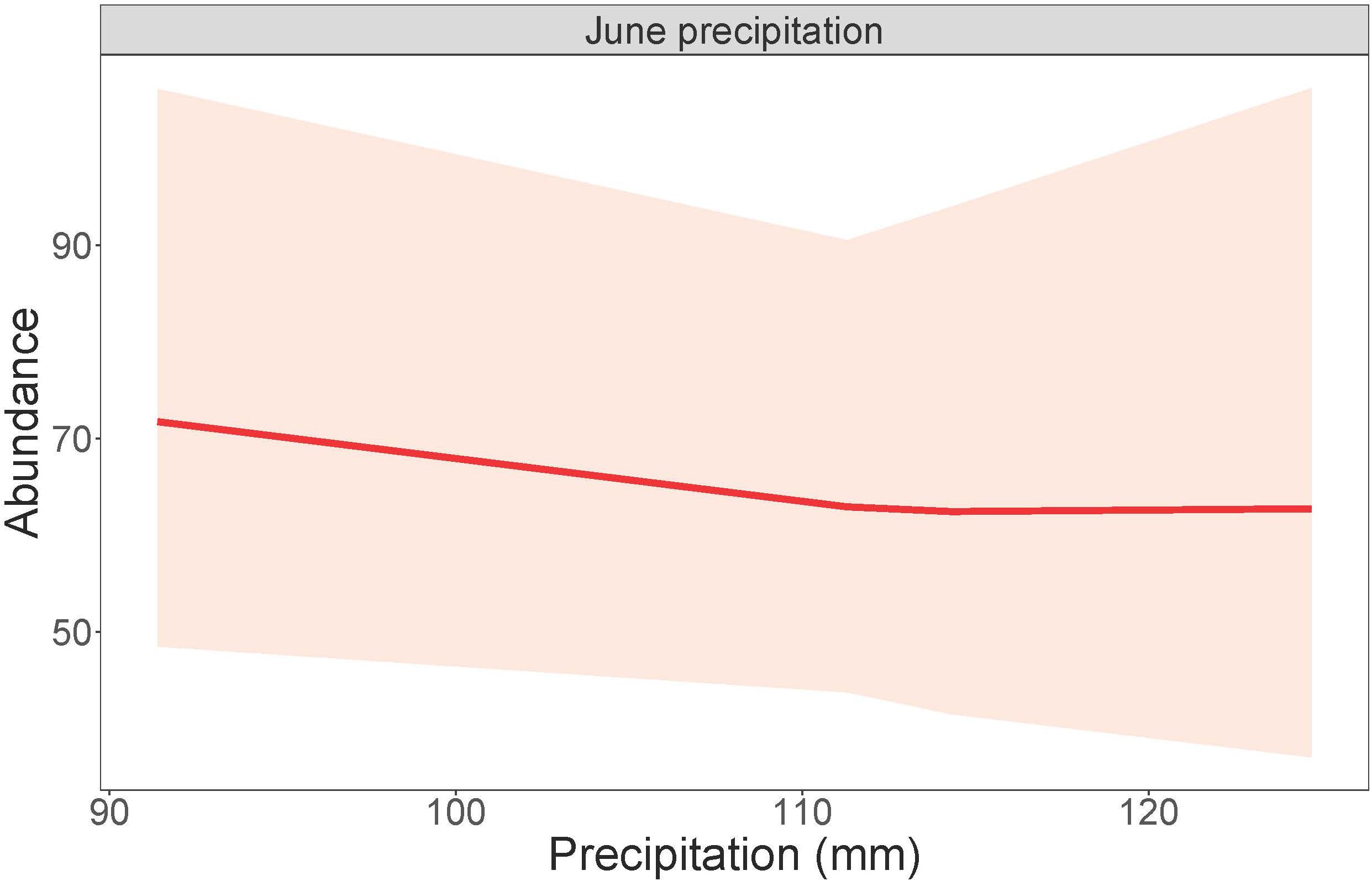
Figure 2. Dickcissel abundance in response to June precipitation. Total June precipitation plotted against numbers of individual adult birds, within a 95% confidence interval.
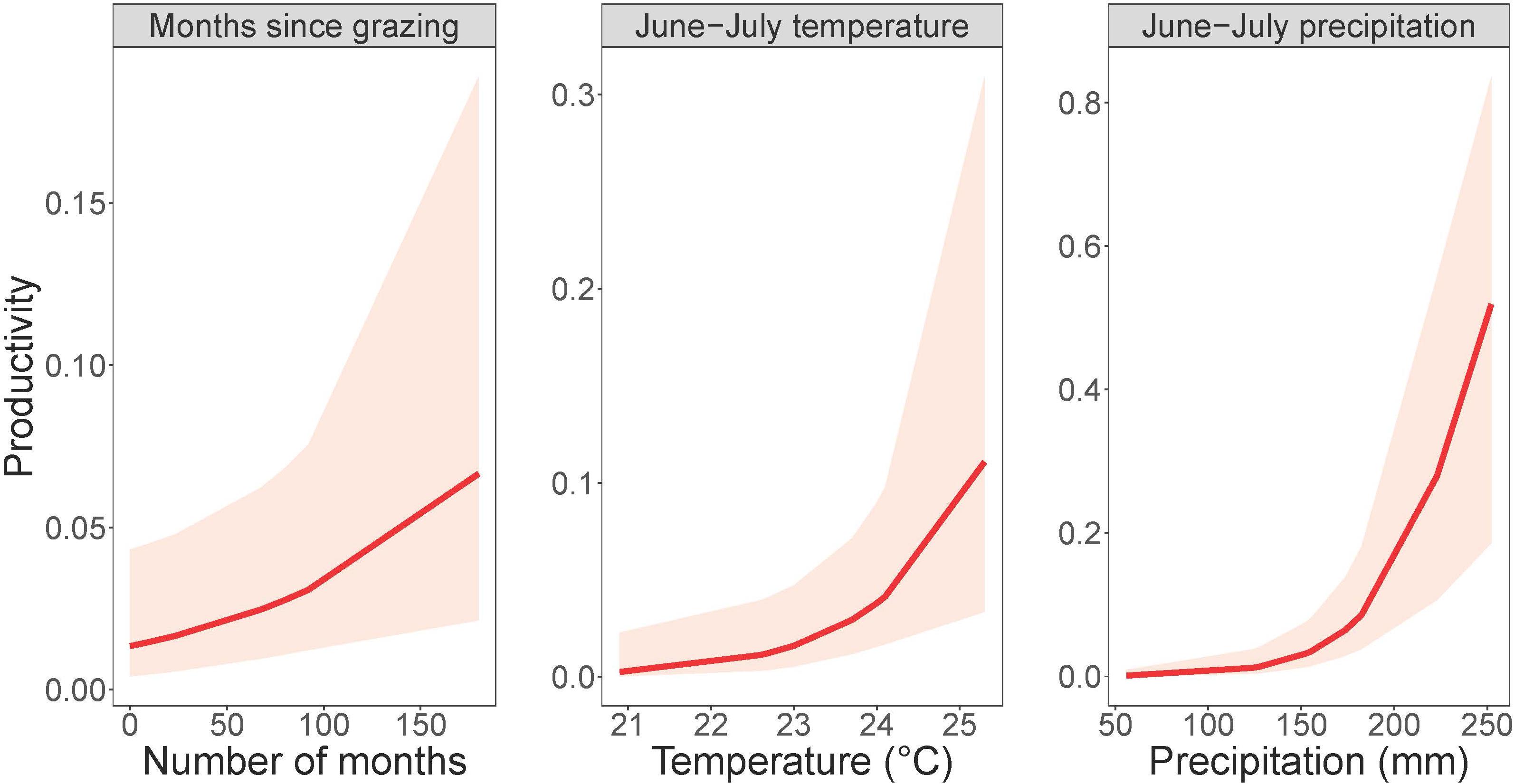
Figure 3. Dickcissel productivity in response to grazing and summer precipitation and temperature. Plots depict (from left to right) months since grazing, mean June-July temperature, and total June-July precipitation plotted against dickcissel productivity, or the probability of encountering a juvenile bird, within 95% confidence intervals.
Cowbird Responses to Weather, Land Management, and Habitat Parameters
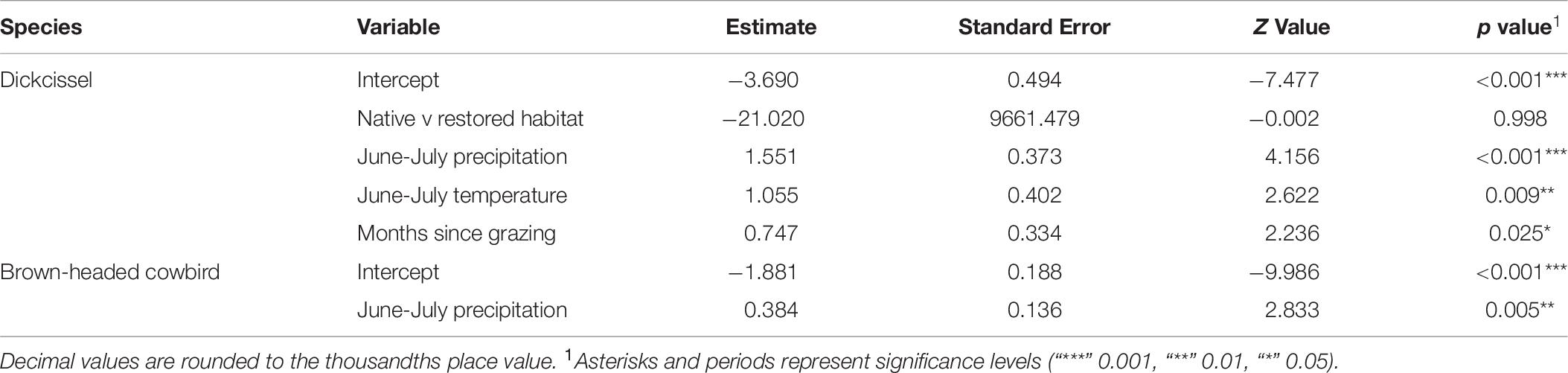
Cowbird abundance exhibited a quadratic positive relationship with June precipitation (Z = 3.99, p = 6.70e-05; Table 9 and Figure 4). In addition, cowbird abundance had a positive relationship with non-breeding season (August-May) precipitation (Z = 2.05, p = 0.04). We captured more cowbirds in remnant prairie fragments compared to grasslands restored after they were planted with invasive grasses or agricultural crops (Z = −2.13, p = 0.03, Table 9). Cowbird productivity was positively correlated with June-July precipitation (Z = 2.833, p = 0.01; Table 10 and Figure 5).
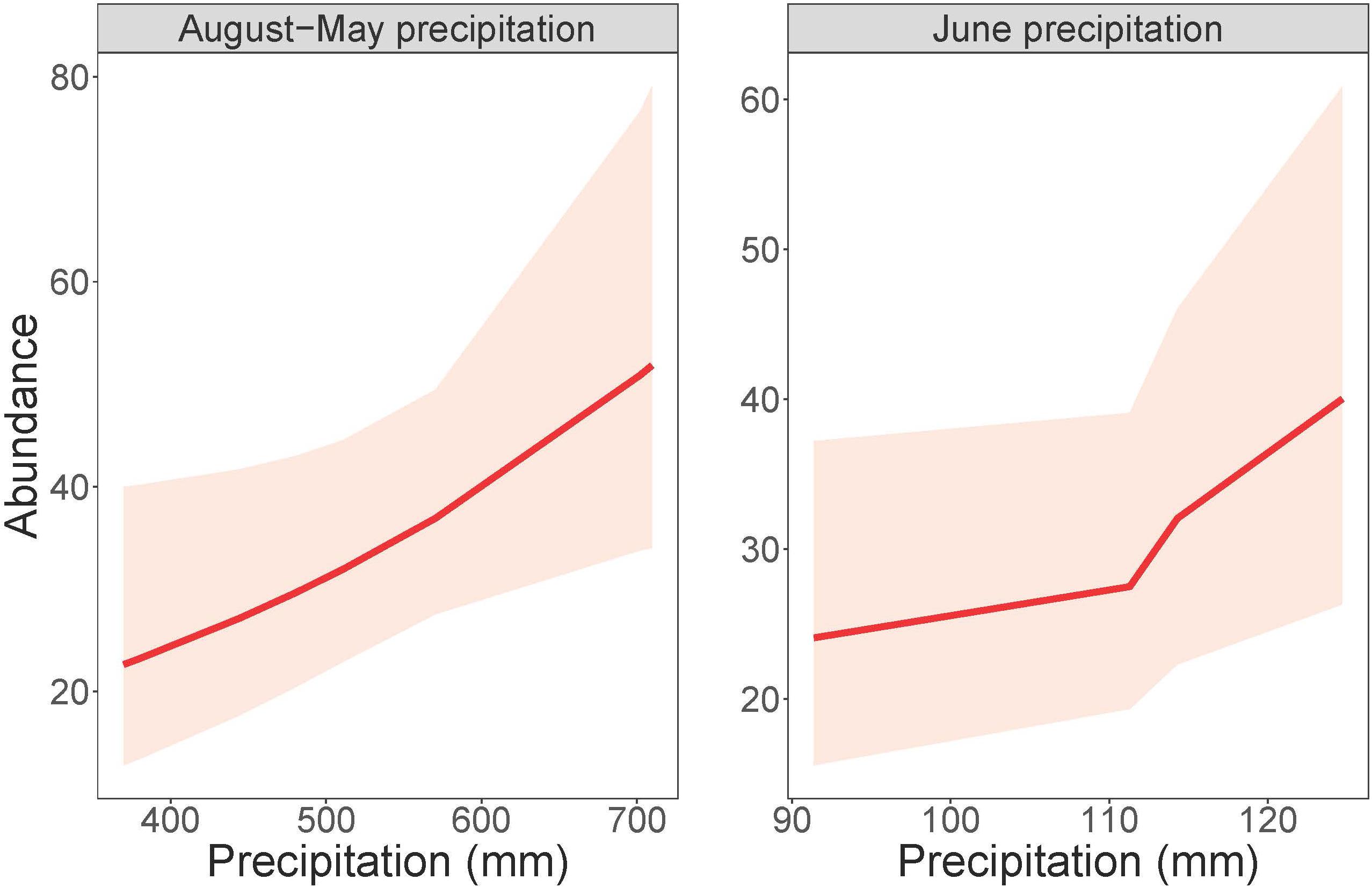
Figure 4. Cowbird abundance in response to precipitation during both the non-breeding season (August-May) and June. Total August-May precipitation (left) and total June precipitation (right) plotted against numbers of individual adult birds, within 95% confidence intervals.
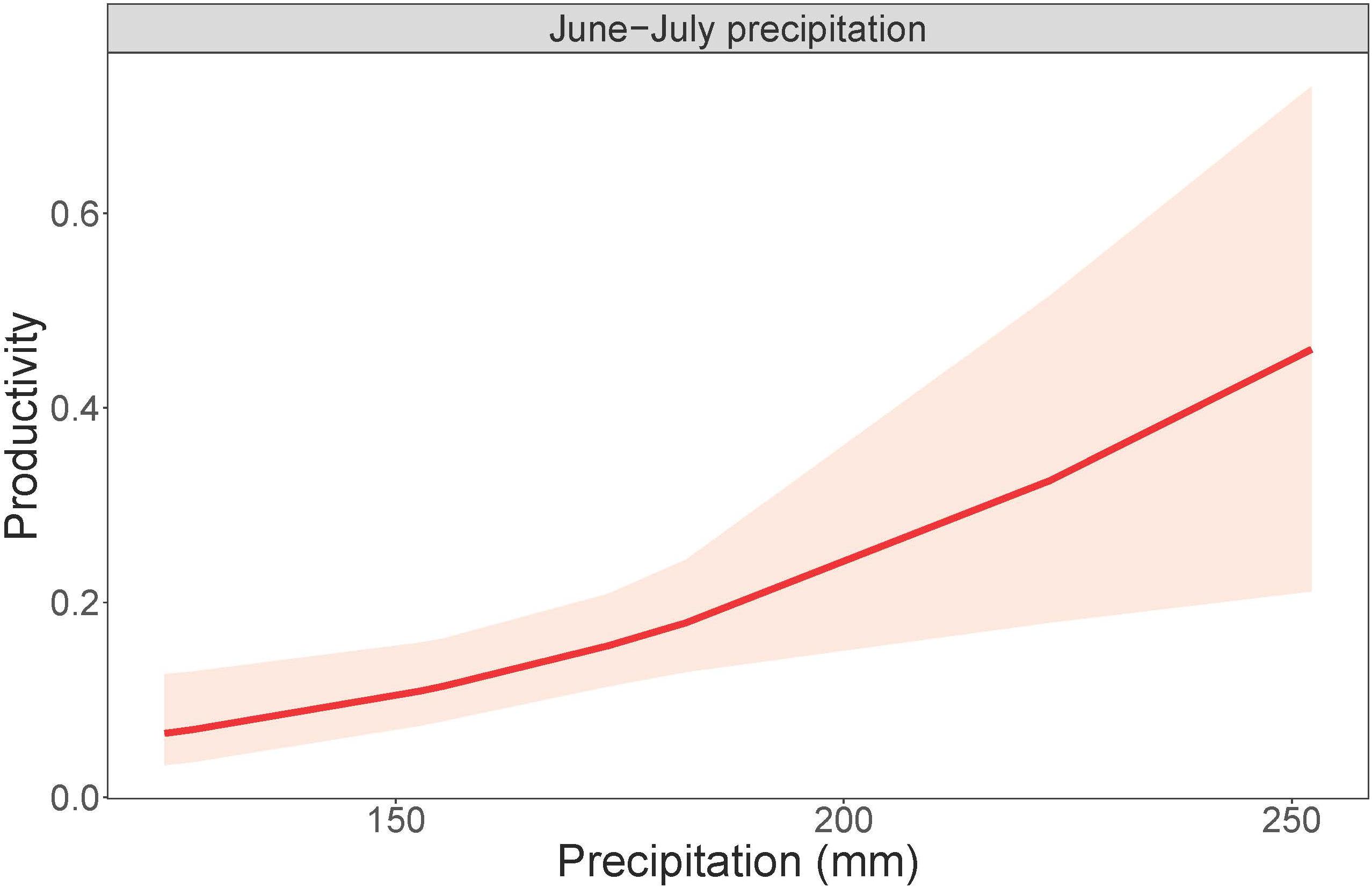
Figure 5. Cowbird productivity in response to summer precipitation. Total June-July precipitation plotted against cowbird productivity, or the probability of encountering a juvenile bird, within a 95% confidence interval.
Discussion
Dickcissel Relationships With Precipitation and Grassland Management
Dickcissel abundance declined with increasing June precipitation, corroborating previous research showing that dickcissel and other breeding songbird densities in this area decreased with increasing levels of moisture (Kim et al., 2008). These findings contrast with previous research elsewhere in the Great Plains testing the “climate bottleneck hypothesis” that found that grassland breeding bird abundance did not significantly change in response to changes in precipitation (Wiens, 1973; Zimmerman, 1992), with the important caveat that our study areas differed in their proximity to riparian corridors and therefore flooding potential. Our findings also contrast with a pattern of increasing bird abundance with increasing precipitation for wetland bird species elsewhere in the Great Plains (Mantyka-Pringle et al., 2019). Dickcissel abundance decreased with increased precipitation during the breeding season but not the non-breeding season (when they are away), implying that negative effects of increased precipitation on dickcissels stem from direct effects during the breeding season, such as emigration due to heavy precipitation and related extreme events including flooding and summer hailstorms. High precipitation and extreme precipitation events may also cause direct mortality of adults and nestlings as well as nest failures, and changes in precipitation may influence exposure to predators (e.g., Takagi, 2001; Mattsson and Cooper, 2009; Robinson et al., 2017; Godwin et al., 2019).
Dickcissels showed high site fidelity, with 70–93% of recaptures occurring at the same site as that of initial capture. This is consistent with findings from other mark-recapture studies demonstrating dickcissels’ fidelity to breeding sites in successive years in their core breeding range (Zimmerman and Finck, 1989; Temple, 2020). The male-biased sex ratio (3 males: 1 female) we found is consistent with male-biased sex ratios detected in other studies of this species on both its breeding and wintering grounds (ffrench, 1967; Fretwell and Calver, 1969; Fretwell, 1977; Basili and Temple, 1999). Fretwell (1977) hypothesized that the conversion of natural grasslands to crops has resulted in a novel winter food supply with larger (crop) seeds rather than smaller (native grass) seeds that in turn favors higher survival of larger (male) birds whose bills are better equipped to crush the larger seeds. Previous research has suggested that male-biased sex ratios may be more pronounced in dickcissel populations in the northern portions of their breeding range, such as in our study area, and have negative implications for dickcissels’ breeding success, particularly given that dickcissels evolved polygyny, in which males typically mate with multiple females (Zimmerman, 1966; Fretwell, 1977; Temple, 2020). Female densities were low in our study area, and low female densities are associated with low productivity, underlining their vulnerability to declines (Fretwell and Calver, 1969; Fretwell, 1977; Hixon and Johnson, 2009).
Dickcissel abundance showed near-significant positive correlations with both time since grazing and time since haying, increasing with time since disturbance for at least 180 months (15 years), the maximum length of post-disturbance grassland regeneration considered in this study. Dickcissels’ preference for breeding sites with tall, dense grasses and scattered forbs, and their positive responses to management that maintains these conditions, are well-established (e.g., Dechant et al., 2002). Dickcissels and other grassland birds consume some arthropods, such as Hymenoptera, that may be more abundant in ungrazed areas since cattle may disturb or trample them (Hoernemann et al., 2001). Dickcissels also consume Orthopteran species (Gross, 1921; Kobal, 1990), which some studies have shown to decline as a result of grazing (Capinera and Sechrist, 1982; Fielding and Brusven, 1995; but see Horn and Dowell, 1974; Hoernemann et al., 2001). Other research has found negative relationships between dickcissel abundance and recent grassland disturbance (Winter, 1998). Multi-year periods between haying events allow the development of dense vegetation including grasses, forbs, and woody plants that provide food, shelter, and nest sites (Temple, 2020). While hayed grasslands may provide nesting habitat for dickcissels and other grassland nesting birds when haying occurs after the breeding season, they create ecological traps when haying occurs during the breeding season because haying destroys nests, killing nestlings and reducing habitat quality (Luscier and Thompson, 2009; Prestby and Anich, 2013). Haying also reduces food availability for dickcissels and other insectivorous birds, as Orthopterans, ants, millipedes, and other invertebrates are sensitive to the mechanical disturbance associated with haying (Evans, 1988; Hoernemann et al., 2001; Jonas et al., 2002; Humbert et al., 2009). Arthropod declines resulting from haying may cause dickcissels and other birds to leave for more suitable habitat (James and Neal, 1986; Zalik and Strong, 2008). Dickcissels and other grassland obligate species may benefit from access to grasslands featuring a mosaic of successional times since disturbance (e.g., Hamilton, 2007; Murray et al., 2016).
While land management is clearly important for dickcissel and other grassland bird conservation (Dechant et al., 2002; Temple, 2020), our findings show that precipitation exerts a stronger influence than management in this riparian ecosystem. Broader-scale climate effects can also influence dickcissels’ settlement decisions (Jensen and Cully, 2005), for example if dickcissels concentrate in the southern part of their breeding range in wetter years and in the northern part of their range in drier years or vice versa. Climate change may thus influence bird species on a broader scale, as geographic shifts in climate envelope may sometimes outweigh effects of local land management (Barbet-Massin et al., 2012; Sohl, 2014; Mantyka-Pringle et al., 2019). Other factors not captured in this study may also influence breeding bird populations, such as carry-over effects, which recent students have suggested are widespread and important in many bird species (Harrison et al., 2011; O’Connor et al., 2014) and can be sex- and age-specific (Saino et al., 2017; López-Calderón et al., 2019). Wintering area precipitation, habitat, and resources may thus influence arrival times and productivity of migratory songbirds on their breeding grounds (e.g., Marra et al., 1998; Robb et al., 2008; McKinnon et al., 2015; Akresh et al., 2019; López-Calderón et al., 2019). For example, Eurasian blue tits (Cyanistes caeruleus) with higher winter food availability laid eggs earlier and exhibited higher chick fledging rates (Robb et al., 2008). Previous research has suggested that winter mortality of dickcissels may have driven their past population declines and/or their male-biased sex ratios (Fretwell, 1973; Basili and Temple, 1999). Dickcissel breeding densities may also be negatively correlated with adult survival, which tends to be substantially lower than that of other songbird species comparable in size (DeSante et al., 2015). Whether winter-driven mortality and/or carry-over effects currently limit dickcissel populations is an important question for future research (Temple, 2020).
Cowbird Relationships With Precipitation and Dickcissels
While dickcissels declined during years with higher precipitation, cowbird abundance increased. Because increasing precipitation contributes to declining dickcissel abundance and increasing cowbird abundance simultaneously, dickcissels may be exposed to greater parasitism pressure from larger proportions of cowbirds in the same breeding area. In addition, we captured higher numbers of cowbirds in native remnant grasslands compared to restored grasslands, while dickcissel numbers did not appear to differ between remnant and restored grasslands. This finding suggests that cowbird parasitism on dickcissels and other hosts may be more frequent and intense in native remnant grasslands, driving ecological traps if dickcissels and other hosts are attracted to habitats that function as population sinks (Hale and Swearer, 2016). Cowbird parasitism intensity varies considerably at landscape scales (Jensen and Cully, 2005). Previous research found that cowbird densities mirrored dickcissel densities, either due to cowbirds tracking dickcissels, similar habitat, and/or similar foraging preferences (Kim et al., 2008). Several studies have suggested that cowbird parasitism may contribute to inverse density dependence in species including dickcissels (Zimmerman, 1966; Fretwell, 1977; Temple, 2020) and can therefore accelerate declines of small and fragmented host populations (Courchamp et al., 1999; Lowther, 2020), such as in our study area.
Dickcissels sustain high rates of cowbird parasitism through much or all of their breeding range, both in terms of the number of dickcissel nests affected and the number of cowbird eggs placed in each nest. Multiple studies have shown that dickcissels are the most parasitized grassland breeding bird species (Patterson and Best, 1996; Rivers et al., 2010) and that dickcissel productivity is reduced in parasitized nests (Fretwell, 1977; Jensen and Cully, 2005; Temple, 2020). Whereas only June precipitation influenced dickcissel abundance, cowbird abundance increased with both June and non-breeding season precipitation, suggesting that cowbirds benefit indirectly from higher precipitation and its relationship with greater plant growth and habitat complexity, which in turn can provide for food birds in the form of seeds and arthropods. When more frequent and heavy June rain may flood dickcissel habitat in this riparian area and in extreme cases (such as summer hail, which occurred multiple times during the study period), knock down nests, causing dickcissels to emigrate, cowbirds are not constrained by breeding territories or particular nests in the same way. In addition to their relationships with dickcissels, cowbird densities are locally related to other host densities, including their fellow Icterids, red-winged blackbirds (Agelaius phoeniceus) and bobolinks (Dolichonyx oryzivorus). Thus, in years with higher precipitation they have the flexibility to switch to these and other avian hosts that may be more tolerant of high June precipitation than dickcissels. For both dickcissels and cowbirds, precipitation was the only significant predictor of abundance of the weather and management factors we tested, highlighting the importance of precipitation in mediating interactions between cowbirds, dickcissels, and other hosts on their breeding grounds.
Dickcissel and Cowbird Productivity and Implications for Population Trends
Dickcissel productivity was positively correlated with breeding season precipitation, temperature, and time since grazing. Higher precipitation may benefit dickcissel productivity and juvenile survival by promoting greater grassland productivity (Rotenberry and Wiens, 1991; Chase et al., 2005), structural complexity, and food abundance (Rotenberry and Wiens, 1991; Skagen and Yackel Adams, 2012). Like that of dickcissels, cowbird productivity increased with increased June-July precipitation, meaning that increasing numbers of cowbirds were raised by hosts including dickcissels. In other riparian ecosystems, higher avian productivity has also been found to be positively correlated with precipitation, such as in Louisiana waterthrush (Parkesia motacilla; Mattsson and Cooper, 2009) and Mississippi kites (Ictinia mississippiensis; Welch-Acosta et al., 2019). By contrast, in other ecosystems, higher precipitation resulted in higher losses of nestlings to predators in bull-headed shrikes (Lanius bucephalus) (Takagi, 2001) and lower productivity in peregrine falcons (Falco peregrinus; Robinson et al., 2017) and tree swallows (Tachycineta bicolor; Godwin et al., 2019).
Dickcissel productivity was also positively correlated with breeding season temperature. The relatively high temperatures (36–40.5°C) at which optimal songbird egg development occurs may help explain this (DuRant et al., 2013). This finding contrasts with studies of other avian species in hotter climates in which higher temperatures (33–39°C, in contrast with July temperatures of 20–31°C in our study area) were associated with lower productivity (e.g., van de Ven et al., 2020). In addition, the positive correlation we found between dickcissel productivity and time since grazing may derive in part from greater predation risks to juvenile dickcissels in recently disturbed fields due to lack of vegetative cover (Bollinger et al., 1990; Suedkamp Wells et al., 2007). Past research has found that predation may cause half of dickcissel nest failures (Zimmerman, 1966; Fretwell, 1977). Nest predators include snakes and ground squirrels (Spermophilus sp.; Temple, 2020), both of which were present at our sites. Longer periods of time since grazing may also favor productivity in that grasslands with taller vegetation provide birds and their nests and nestlings with shade and shelter that have a buffering effect against higher, more variable temperatures (Dechant et al., 2002; Geiger et al., 2009; Villegas et al., 2010; Jarzyna et al., 2016).
In dickcissel populations with male-biased sex ratios, such as in our study system, low female densities may drive low productivity (Fretwell, 1977). Although the extremely low ratio of juvenile to adult dickcissels we found likely underestimates dickcissels’ true productivity, DeSante et al. (2015) likewise found extremely low dickcissel productivity, averaging 80–90% lower than that of most other North American songbirds. Although dickcissel breeding densities peak in their core breeding range, male territoriality appears to limit them (Fretwell, 1977; Temple, 2020). Studies elsewhere have suggested that dickcissel productivity declines at lower densities, possibly due to higher cowbird parasitism levels (Zimmerman, 1966; Fretwell, 1977; Temple, 2020). However, here we found that although dickcissel adult abundance declined with increasing precipitation, their productivity increased. This finding corroborates other research suggesting that dickcissel populations may be regulated through density dependence (DeSante et al., 2015; Temple, 2020), for example if reduced competition for resources among fewer breeding adults may enable higher population growth due to higher survival rates of their young (Hixon and Johnson, 2009). As losses of adults are partially offset by higher numbers of juveniles, this response may be mitigating a significant decline of dickcissels in this area as precipitation has increased over time. However, dickcissels’ low productivity overall may not be sufficient to counteract the declining trend in their abundance, as survival appears to be a stronger driver of population change than productivity in dickcissels (DeSante et al., 2015). Moreover, our findings suggest that cowbird abundance and parasitism rates may increase in the future wetter conditions predicted by climate change models for this region.
Conclusion and Future Directions
Our findings highlight dickcissels’ vulnerability to population declines due to increasing precipitation associated with climate change in this ecosystem, together with increasing cowbird abundance and parasitism that may further reduce dickcissel productivity. Precipitation has significantly increased in frequency and volume in the northeastern Great Plains, including our study area, in recent years (Wuebbles et al., 2014), and levels of precipitation are predicted to continue increasing in the coming decades (USGCRP, 2018). Our research reveals significant changes in avian population parameters related to changing weather and grassland management. Identifying the mechanisms driving these patterns will require additional research. How cowbird parasitism affects dickcissels and other breeding birds in relation to climate change merits particular attention (Lowther, 2020), given cowbirds’ potential to reduce their hosts’ productivity and contribute to population declines, especially in small and fragmented host populations common in temperate grasslands. Adaptive management for conservation (e.g., Dechant et al., 2002) that incorporates habitat heterogeneity remains essential to conserve dickcissels and other grassland specialists. In turn, targeted research to understand how grassland birds are responding to climate change and predict the consequences for their future populations is crucial to inform adaptive management strategies that mitigate the ongoing declines of grassland birds.
Understanding the interactions between cowbirds, dickcissels, and other host species, and how shifting weather patterns mediate these interactions, is an important consideration for grassland management and bird conservation under climate change. Future studies focused on dickcissels’ and other species’ responses to climate and management factors will complement our findings and elucidate whether their responses resemble or differ from those we describe here (Reed et al., 2006; Ficetola and Maiorano, 2016; Bruckerhoff et al., 2020; Glass et al., 2020). While we have focused here on local patterns of dickcissel population change, regional weather patterns also have important influences on migratory bird breeding population dynamics, and carry-over effects may also play a role. Thus, we encourage future studies of dickcissels and other migratory birds that track individuals through telemetry and other technologies throughout the annual cycle, as well as further inquiries into variation in the sex ratios of breeding dickcissels and their relationships to avian survival, productivity, and population dynamics. Finally, grassland conversion to agriculture remains a foremost threat to obligate grassland species, and protecting and managing grasslands is therefore an utmost conservation priority on dickcissels’ and other migratory birds’ breeding grounds as well as their migratory stopover and wintering areas.
Data Availability Statement
All datasets generated for this study are included in the article/Supplementary Material.
Ethics Statement
The animal study was reviewed and approved by the USGS Bird Banding Laboratory.
Author Contributions
NA designed and supervised the project. DHK, NA, RHK, and AG collected field data. KMR and RHK organized the database with contributions from AG. KMR, SG, AS, and RHK analyzed data and produced tables and figures. NA and KMR wrote the manuscript with input from all other authors. All authors contributed to the article and approved the submitted version.
Funding
The Crane Trust funded field data collection, management, analysis, and publication. The International Bird Conservation Partnership provided supplemental funding for data management, analysis, research, and writing of this article.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Tim Smith for providing land management data and Steve Holzman for contributing maps. We are grateful to many people who contributed to making this study happen, including: Madison Sutton, Kelsey King, Joel Popp, Ravin Grove, Mary Ware, Kelsey Brown, Cathy Cargill, Sandy Douglas, Craig Thompson, Brad Korell, Brice Krohn, Andrew Caven, Josh Wiese, Jacob Salter, and all those who contributed bird, habitat, and land management data from 2002–2019. We thank the USGS Bird Banding Lab, including Danny Bystrak, for their support and the Institute for Bird Populations, including Rodney Siegel, Peter Pyle, Jim Saracco, Danielle Kaschube, Ron Taylor, and Dave DeSante. Crystal Stiles and Natalie Umphlett of the University of Nebraska’s High Plains Regional Climate Center provided advice and guidance on climate data sources and analysis. The editor MM, two reviewers, and Ola Svensson read the manuscript and made comments to help us improve it. This manuscript is dedicated to all who work to protect native grasslands and the migratory birds that depend on them.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fevo.2020.536769/full#supplementary-material
References
Ahrestani, F. S., Saracco, J. F., Sauer, J. R., Pardieck, K. L., and Royle, J. A. (2017). An integrated population model for bird monitoring in North America. Ecol. Appl. 27, 916–924. doi: 10.1002/eap.1493
Akresh, M. E., King, D. I., and Marra, P. P. (2019). Examining carry-over effects of winter habitat on breeding phenology and reproductive success in prairie warblers Setophaga discolor. J. Avian Biol. 50:e02025. doi: 10.1111/jav.02025
Amburgey, S. M., Miller, D. A. W., Campbell Grant, E. H., Rittenhouse, T. A. G., Benard, M. F., Richardson, J. L., et al. (2018). Range position and climate sensitivity: the structure of among-population demographic responses to climatic variation. Glob. Change Biol. 24, 439–454. doi: 10.1111/gcb.13817
Amstrup, S. C., McDonald, T. L., and Manly, B. F. J. (eds) (2005). Handbook of Capture-Recapture Analysis. Princeton: Princeton University Press.
Anderson, R. C. (2006). Evolution and origin of the Central Grassland of North America: climate, fire, and mammalian grazers. J. Torrey Bot. Soc. 133, 626–647. doi: 10.3159/1095-5674(2006)133[626:EAOOTC]2.0.CO;2
Askins, R. A., Chávez-Ramírez, F., Dale, B. C., Haas, C. A., Herkert, J. R., Knopf, F. L., et al. (2007). Conservation of grassland birds in North America: understanding ecological processes in different regions. Ornithol. Monogr. 64, 1–46. doi: 10.2307/40166905
Barbet-Massin, M., Thuiller, W., and Jiguet, F. (2012). The fate of European breeding birds under climate, land-use and dispersal scenarios. Glob. Change Biol. 18, 881–890. doi: 10.1111/j.1365-2486.2011.02552.x
Barker, R. J., Burnham, K. P., and White, G. C. (2004). Encounter history modeling of joint mark-recapture, tag-resighting and tag-recovery data under temporary emigration. Statistica Sinica 14, 1037–1055.
Basili, G. D., Brown, S. L., Finck, E. J., Reinking, D., Steigman, K. L., Temple, S. A., et al. (1997). “Breeding biology of Dickcissels across their range and over time,” in Continental-scale Ecology and Conservation of the Dickcissel, ed. G. D. Basili (Madison: University of Wisconsin-Madison), 108–151.
Basili, G. D., and Temple, S. A. (1999). Winter ecology, behavior, and conservation needs of dickcissels in Venezuela. Stud. Avian Biol. 19, 289–299.
Blancher, P. (2003). Importance of North America’s Grasslands to Birds. Montreal, MON: Commission for Environmental Cooperation and Bird Studies.
Bollinger, E. K., Bollinger, P. B., and Gavin, T. A. (1990). Effects of hay-cropping of eastern populations of the bobolink. Wildl. Soc. Bull. 18, 142–150.
Brennan, L. A., and Kuvlesky, W. P. Jr. (2005). North American grassland birds: an unfolding conservation crisis? J. Wildl. Manage 69, 1–13. doi: 10.2193/0022-541X(2005)069<0001:NAGBAU>2.0.CO;2
Brooks, M. E., Kristensen, K., van Benthem, K. J., Magnusson, A., Berg, C. W., Nielsen, A., et al. (2017). glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R. J. 9, 378–400. doi: 10.32614/RJ-2017-066
Bruckerhoff, L. A., Connell, R. K., Guinnip, J. P., Adhikari, E., Godar, A., Gido, K. B., et al. (2020). Harmony on the prairie? Grassland plant and animal community responses to variation in climate across land-use gradients. Ecology 101:e02986. doi: 10.1002/ecy.2986
Butler, M. J., Metzger, K. L., and Harris, G. M. (2017). Are whooping cranes destined for extinction? climate change imperils recruitment and population growth. Ecol. Evol. 7, 2821–2834. doi: 10.1002/ece3.2892
Buxton, V. L., Schelsky, W. M., Boves, T. J., Summers, S., Weatherhead, P. J., and Sperry, J. H. (2018). Effects of drought on brood parasite body condition, follicle development, and parasitism: implications for host–parasite dynamics. Auk 135, 908–918. doi: 10.1642/AUK-18-6.1
Capinera, J. L., and Sechrist, T. S. (1982). Grasshopper (Acrididae) – host plant associations: response of grasshopper populations to cattle grazing intensity. Can. Entomol. 114, 1055–1062. doi: 10.4039/Ent1141055-11
Chase, M. K., Nur, N., and Geupel, G. R. (2005). Effects of weather and population density on reproductive success and population dynamics in a song sparrow (Melospiza melodia) population: a long-term study. Auk 122, 571–592. doi: 10.1093/auk/122.2.571
Collins, S. L. (2000). Disturbance frequency and community stability in native tallgrass prairie. Am. Nat. 155, 311–325. doi: 10.1086/303326
Colón, M. R., Long, A. M., and Morrison, M. L. (2017). Responses of an endangered songbird to an extreme drought event. Southeas. Nat. 16, 195–214. doi: 10.1656/058.016.0207
Conant, R. T., Kluck, D., Anderson, M., Badger, A., Boustead, B. M., Derner, J. D., et al. (2018). “Northern great plains,” in Impacts, Risks, and Adaptation in the United States: Fourth National Climate Assessment, eds D. Reidmiller, C. W. Avery, D. R. Easterling, K. E. Kunkel, K. L. M. Lewis, T. K. Maycock, et al. (Washington, DC: U.S. Global Change Research Program), 941–986. doi: 10.7930/NCA4.2018.CH22
Cotton, P. A. (2003). Avian migration phenology and global climate change. PNAS 100, 12219–12222. doi: 10.1073/pnas.1930548100
Courchamp, F., Clutton-Brock, T., and Grenfell, B. (1999). Inverse density dependence and the Allee effect. Trends Ecol. Evol. 14, 405–410. doi: 10.1016/S0169-5347(99)01683-3
Croston, R., and Hauber, M. E. (2010). The ecology of avian brood parasitism. Nat. Educ. Knowl. 1:3.
Cruz-McDonnell, K. K., and Wolf, B. O. (2016). Rapid warming and drought negatively impact population size and reproductive dynamics of an avian predator in the arid southwest. Glob. Change Biol. 22, 237–253. doi: 10.1111/gcb.13092
Culp, L. A., Cohen, E. B., Scarpignato, A. L., Thogmartin, W. E., and Marra, P. P. (2017). Full annual cycle climate change vulnerability assessment for migratory birds. Ecosphere 8:e01565. doi: 10.1002/ecs2.1565
Dechant, J. A., Sondreal, M. L., Johnson, D. H., Igl, L. D., Goldade, C. M., Zimmerman, A. L., et al. (2002). Effects of management practices on grassland birds: dickcissel. USGS Northern Prairie Wildlife Res. Center 114.
DeSante, D. F., and Kaschube, D. R. (2009). The Monitoring Avian Productivity and Survivorship (MAPS) program 2004, 2005, and 2006 report. Bird Populations 9, 86–169.
DeSante, D. F., Burton, K. M., Velez, P., Froelich, D., Kaschube, D., and Albert, S. (2017). MAPS Manual 2017 Protocol. Institute for Bird Populations. available online at: http://www.birdpop.org/pages/maps.php (accessed October 9, 2020).
DeSante, D. F., Kaschube, D. R., and Saracco, J. F. (2015). Vital Rates of North American Landbirds. Available online at: http://www.VitalRatesOfNorthAmericanLandbirds.org: The Institute for Bird Populations (accessed October 9, 2020).
DuRant, S. E., Hopkins, W. A., Hepp, G. R., and Walters, J. R. (2013). Ecological, evolutionary, and conservation implications of incubation temperature-dependent phenotypes in birds. Biol. Rev. 88, 499–509. doi: 10.1111/brv.12015
Evans, E. W. (1988). Grasshopper (Insecta: Orthoptera: Acrididae) assemblages of tallgrass prairie: influences of fire frequency, topography, and vegetation. Can. J. Zoo. 66, 1495–1501. doi: 10.1139/z88-219
Ficetola, G. F., and Maiorano, L. (2016). Contrasting effects of temperature and precipitation change on amphibian phenology, abundance and performance. Oecologia 181, 683–693. doi: 10.1007/s00442-016-3610-9
Fielding, D. J., and Brusven, M. A. (1995). Grasshopper densities on grazed and ungrazed rangeland under drought conditions in southern Idaho. Great Basin Nat. 55, 352–358.
Finch, T., Pearce-Higgins, J. W., Leech, D. I., and Evans, K. L. (2014). Carry-over effects from passage regions are more important than breeding climate in determining the breeding phenology and performance of three avian migrants of conservation concern. Biodivers. Conserv. 23, 2427–2444. doi: 10.1007/s10531-014-0731-5
Fletcher, R. J. Jr., and Koford, R. R. (2004). Consequences of rainfall variation for breeding wetland blackbirds. Can. J. Zool. 82, 1316–1325. doi: 10.1139/z04-107
Foster, K. R., Godwin, C. M., Pyle, P., and Saracco, J. F. (2016). Reclamation and habitat-disturbance effects on landbird abundance and productivity indices in the oil sands region of northeastern Alberta, Canada. Restor. Ecol. 25, 532–538. doi: 10.1111/rec.12478
Fretwell, S. D. (1973). “The regulation of bird populations on Konza Prairie: the effects of events off of the prairie,” in Proceedings of the Third Midwest Prairie Conference Proceedings, ed. L. C. Hulbert (Manhattan: Kansas State Univ), 37–44.
Fretwell, S. D., and Calver, J. S. (1969). On territorial behavior and other factors influencing habitat distribution in birds. Acta Biotheor. 19, 37–44. doi: 10.1007/BF01601955
Geiger, R., Aron, R. H., and Todhunter, P. (2009). The Climate Near the Ground. Lanham: Rowman & Littlefield Publishing Group.
Glass, A. J., Caven, A. J., Kim, D., Sutton, M. O., and Arcilla, N. (2020). Climate change and land management implications for a declining Neotropical migratory songbird breeding in the North American Great Plains. Avian Conserv. Ecol. 15:4. doi: 10.5751/ACE-01515-150104
Godwin, C. M., Barclay, R. M. R., and Smits, J. E. G. (2019). Tree Swallow (Tachycineta bicolor) nest success and nestling growth near oil sands mining operations in northeastern Alberta, Canada. Can. J. Zool. 97, 547–557. doi: 10.1139/cjz-2018-0247
Gopalaswamy, A. M., Royle, J. A., Hines, J. E., Singh, P., Jathanna, D., Kumar, N. S., et al. (2012). Program SPACECAP: software for estimating animal density using spatially explicit capture-recapture models. Methods Ecol. Evol. 3, 1067–1072. doi: 10.1111/j.2041-210X.2012.00241.x
Gross, A. O. (1921). The dickcissel (Spiza americana) of the Illinois prairies. Auk 38, 163–184. doi: 10.2307/4074023
Gross, A. O. (1968). “Dickcissel,” in Life Histories of North American Cardinals, Grosbeaks, Buntings, Towhees, Finches, Sparrows and Their Allies, ed. O.L. Austin Jr. New Delhi: U S. National Museum Bulletin. 158–191.
Guigueno, M. F., Snow, D. A., MacDougall-Shackleton, S. A., and Sherry, D. F. (2014). Female cowbirds have more accurate spatial memory than males. Biol. Lett. 10:20140026. doi: 10.1098/rsbl.2014.0026
Hale, R., and Swearer, S. E. (2016). Ecological traps: current evidence and future directions. Proc. R. Soc. B 283:20152647. doi: 10.1098/rspb.2015.2647
Hamilton, R. G. (2007). “Restoring heterogeneity on the Tallgrass Prairie Preserve: applying the fire–grazing interaction model,” in Proceedings of the 23rd Tall Timbers Fire Ecology Conference: Fire in Grassland and Shrubland Ecosystems, eds R. E. Masters and K. E. M. Galley (Tallahassee, FL: Tall Timbers Research Station), 163–169.
Harrison, X. A., Blount, J. D., Inger, R., Norris, D. R., and Bearhop, S. (2011). Carry-over effects as drivers of fitness differences in animals. J. Anim. Ecol. 80, 4–18. doi: 10.1111/j.1365-2656.2010.01740.x
Hixon, M. A., and Johnson, D. W. (2009). “Density dependence and independence,” in Encyclopedia of Life Sciences (ELS), (ed.) R. Johnson (Chichester: John Wiley & Sons, Ltd), doi: 10.1002/9780470015902.a0021219
Hoernemann, C. K., Johnson, P. J., and Higgins, K. F. (2001). Effects of grazing and haying on arthropod diversity in North Dakota conservation reserve program grasslands. Proc. S. Dak. Acad. Sci. 80, 283–308.
Horn, D. J., and Dowell, R. V. (1974). A comparison of insect faunas in grazed and ungrazed grasslands. Proceedings North Central Branch-E.S.A. 29, 103–106.
Humbert, J. Y., Ghazoul, J., and Walter, T. (2009). Meadow harvesting techniques and their impacts on field fauna. Agric. Ecosyst. Environ. 130, 1–8. doi: 10.1016/j.agee.2008.11.014
James, D. A., and Neal, J. C. (1986). Arkansas Birds: their Distribution and Abundance. Fayetteville, AR: University of Arkansas Press.
Jarzyna, M. A., Zuckerberg, B., Finley, A. O., and Porter, W. F. (2016). Synergistic effects of climate and land cover: grassland birds are more vulnerable to climate change. Landsc. Ecol. 31, 2275–2290. doi: 10.1007/s10980-016-0399-1
Jensen, W. E., and Cully, J. F. Jr. (2005). Geographic variation in brown-headed cowbird (Molothrus ater) parasitism on dickcissels (Spiza americana) in Great Plains tallgrass prairie. Auk 122, 648–660. doi: 10.1093/auk/122.2.648
Johnson, T. N., Kennedy, P. L., DelCurto, T., and Taylor, R. V. (2011). Bird community responses to cattle stocking rates in a Pacific Northwest bunchgrass prairie. Agric. Ecosyst. Environ. 144, 338–346. doi: 10.1016/j.agee.2011.10.003
Jonas, J. L., Whiles, M. R., and Charlton, R. E. (2002). Aboveground invertebrate responses to land management differences in a central Kansas grassland. Environ. Entomol. 31, 1142–1152. doi: 10.1603/0046-225X-31.6.1142
Kim, D. H., Newton, W. E., Lingle, G. R., and Chávez-Ramírez, F. (2008). Influence of grazing and available moisture on breeding densities of grassland birds in the Central Platte River Valley, Nebraska. Wilson J. Ornithol. 120, 820–829. doi: 10.1676/07-153.1
King, K. C., Caven, A. J., Leung, K. G., Ranglack, D. H., and Arcilla, N. (2019). High society: behavioral patterns as a feedback loop to social structure in plains bison (Bison bison bison). Mammal Res. 64, 365–376. doi: 10.1007/s13364-019-00416-7
Kleinhesselink, A. R., and Adler, P. B. (2018). The response of big sagebrush (Artemisia tridentata) to interannual climate variation changes across its range. Ecology 99, 1139–1149. doi: 10.1002/ecy.2191
Knudsen, E., Lindén, A., Both, C., Jonzén, N., Pulido, F., Saino, N., et al. (2011). Challenging claims in the study of migratory birds and climate change. Biol. Rev. 86, 928–946. doi: 10.1111/j.1469-185X.2011.00179.x
Kobal, S. N. (1990). Abundance, Habitat Selection, and Food Habits of Grassland Birds in 3 Non-native Grasslands in Northern Illinois. Master’s thesis University of Wisconsin-Stevens Point: Stevens Point, WI.
Long, C. A., Long, C. F., Knops, J., and Matulionis, D. H. (1965). Reproduction in the dickcissel. Wilson Bull. 77, 251–256.
López-Calderón, C., Balbontín Arenas, J., Hobson, K. A., and Møller, A. P. (2019). Age-dependent carry-over effects in a long-distance migratory bird. Sci. Rep. 9:12032. doi: 10.1038/s41598-019-47374-3
Lowther, P. E. (2020). “Brown-headed cowbird (Molothrus ater), version 1.0,” in Birds of the World, eds A. F. Poole and F. B. Gill (Ithaca, NY: Cornell Lab of Ornithology), doi: 10.2173/bow.bnhcow.01
Luscier, J. D., and Thompson, W. L. (2009). Short-term responses of breeding birds of grassland and early successional habitat to timing of haying in northwestern Arkansas. Condor 111, 538–544. doi: 10.1525/cond.2009.080019
Macías-Duarte, A., Montoya, A. B., Méndez-González, C. E., Rodríguez-Salazar, J. R., Hunt, W. G., and Krannitz, P. G. (2009). Factors influencing habitat use by migratory grassland birds in the state of Chihuahua, Mexico. Auk 126, 896–905. doi: 10.1525/auk.2009.08251
Manly, B. F. J., McDonald, T. L., Amstrup, S. C., and Regehr, E. V. (2003). Improving size estimates of open animal populations by incorporating information on age. BioScience 53, 666–669. doi: 10.1641/0006-3568(2003)053[0666:ISEOOA]2.0.CO;2
Mantyka-Pringle, C., Leston, L., Messmer, D., Asong, E., Bayne, E. M., Bortolotti, L. E., et al. (2019). Antagonistic, synergistic and direct effects of land use and climate on Prairie wetland ecosystems: ghosts of the past or present? Divers. Distrib. 25, 1924–1940. doi: 10.1111/ddi.12990
Marra, P. P., Hobson, K. A., and Holmes, R. T. (1998). Linking winter and summer events in a migratory bird by using stable-carbon isotopes. Science 282, 1884–1886. doi: 10.1126/science.282.5395.1884
Martin, T. E., Nightingale, J., Baddams, J., Monkhouse, J., Kaban, A., Sastranegara, H., et al. (2017). Variability in the effectiveness of two ornithological survey methods between tropical forest ecosystems. PLoS One 12:e0169786. doi: 10.1371/journal.pone.0169786
Mattsson, B. J., and Cooper, R. J. (2009). Multiscale analysis of the effects of rainfall extremes on reproduction by an obligate riparian bird in urban and rural landscapes. Auk 126, 64–76. doi: 10.1525/auk.2009.08001
McDonald, K. W., McClure, C. J. W., Rolek, B. W., and Hill, G. E. (2012). Diversity of birds in eastern North America shifts north with global warming. Ecol. Evol. 2, 3052–3060. doi: 10.1002/ece3.410
McKinnon, E. A., Stanley, C. Q., and Stutchbury, B. J. M. (2015). Carry-over effects of nonbreeding habitat on start-to-finish spring migration performance of a songbird. PLoS One 10:e0141580. journal.pone.0141580 doi: 10.1371/journal.pone.0141580
Moss, R., Oswald, J., and Baines, D. (2001). Climate change and breeding success: decline of the capercaillie in Scotland. J. Anim. Ecol. 70, 47–61. doi: 10.1111/j.1365-2656.2001.00473.x
Murray, C., Minderman, J., Allison, J., and Calladine, J. (2016). Vegetation structure influences foraging decisions in a declining grassland bird: the importance of fine-scale habitat and grazing regime. Bird Study 63, 1–10. doi: 10.1080/00063657.2016.1180342
Niemuth, N. D., Solberg, J. W., and Shaffer, T. L. (2008). Influence of moisture on density and distribution of grassland birds in North Dakota. Condor 110, 211–222. doi: 10.1525/cond.2008.8514
NOAA (2019). National Centers for Environmental information, Climate at a Glance: County Time Series. Available online at: https://www.ncdc.noaa.gov/cag/ (accessed October 9, 2020).
O’Connor, C. M., Norris, D. R., Crossin, G. T., and Cooke, S. J. (2014). Biological carryover effects: linking common concepts and mechanisms in ecology and evolution. Ecosphere 5, 1–11. doi: 10.1890/ES13-00388.1
Patterson, M. P., and Best, L. B. (1996). Bird abundance and nesting success in Iowa CRP fields: the importance of vegetation structure and composition. Am. Midl. Nat. 135, 153–167. doi: 10.2307/2426881
Pietz, P. J., and Granfors, D. A. (2000). Identifying predators and fates of grassland passerine nests using miniature video cameras. J. Wildl. Manag. 64, 71–87. doi: 10.2307/3802976
Plum, N. (2005). Terrestrial invertebrates in flooded grassland: a literature review. Wetlands 25, 721–737. doi: 10.1672/0277-5212(2005)025[0721:TIIFGA]2.0.CO;2
Prestby, T. G., and Anich, N. M. (2013). The summer of the Dickcissel: 2012. Passen. Pigeon 75, 155–168.
R Core Team (2019). R: A Language and Environment for Statistical Computing. Vienna: R Foundation for Statistical Computing.
Rahmig, C. J., Jensen, W. E., and With, K. A. (2009). Grassland bird responses to land management in the largest remaining tallgrass prairie. Cons. Biol. 23, 420–432. doi: 10.1111/j.1523-1739.2008.01118.x
Reed, A. W., Kaufman, G. A., and Kaufman, D. W. (2006). Species richness-productivity relationship for small mammals along a desert-grassland continuum: differential responses of functional groups. J. Mammal. 87, 777–783. doi: 10.1644/05-MAMM-A-253R2.1
Remsen, J. V. Jr., and Good, D. A. (1996). Misuse of data from mist-net captures to assess relative abundance in bird populations. Auk 113, 381–398. doi: 10.2307/4088905
Revelle, W. R. (2018). Psych: Procedures for Personality and Psychological Research. Evanston, Ill: Northwestern University.
Rivers, J. W., Jensen, W. E., Kosciuch, K. L., and Rothstein, S. I. (2010). Community-level patterns of host use by the brown-headed cowbird (Molothrus ater), a generalist brood parasite. Auk 127, 263–273. doi: 10.1525/auk.2009.09053
Robb, G. N., McDonald, R. A., Chamberlain, D. E., Reynolds, S. J., Harrison, T. J. E., and Bearhop, S. (2008). Winter feeding of birds increases productivity in the subsequent breeding season. Biol. Lett. 4, 220–223. doi: 10.1098/rsbl.2007.0622
Robinson, B. G., Franke, A., and Derocher, A. E. (2017). Weather-mediated decline in prey delivery rates causes food-limitation in a top avian predator. J. Av. Biol. 48, 748–758. doi: 10.1111/jav.01130
Rosenberg, K. V., Dokter, A. M., Blancher, P. J., Sauer, J. R., Smith, A. C., Smith, P. A., et al. (2019). Decline of the North American avifauna. Science 366, 120–124. doi: 10.1126/science.aaw1313
Rotenberry, J. T., and Wiens, J. A. (1991). Weather and reproductive variation in Shrubsteppe Sparrows: a hierarchical analysis. Ecology 72, 1325–1335. doi: 10.2307/1941105
Saino, N., Ambrosini, R., Caprioli, M., Romano, A., Romano, M., Rubolini, D., et al. (2017). Sex-dependent carry-over effects on timing of reproduction and fecundity of a migratory bird. J. Anim. Ecol. 86, 239–249. doi: 10.1111/1365-2656.12625
Saracco, J. F., Radley, P., Pyle, P., Rowan, E., Taylor, R., and Helton, L. (2016). Linking vital rates of landbirds on a tropical island to rainfall and vegetation greenness. PLoS One 11:e0148570. doi: 10.1371/journal.pone.0148570
Saracco, J. F., Siegel, R. B., Helton, L., Stock, S. L., and DeSante, D. F. (2019). Phenology and productivity in a montane bird assemblage: trends and responses to elevation and climate variation. Glob. Change Biol. 25, 985–996. doi: 10.1111/gcb.14538
Sauer, J. R., Hines, J. E., and Fallon, J. (2005). The North American Breeding Bird Survey, Results and Analysis 1966–2005. Version 6.2.2006. Laurel: U.S. Geological Survey, Patuxent Wildlife Research Center.
Sauer, J. R., Hines, J. E., Fallon, J. E., Pardieck, K. L., and Ziolkowski, D. R. Jr., et al. (2012). The North American Breeding Bird Survey, Results and Analysis 1966-2011. Version 12.13.2011. Laurel: U.S. Geological Survey, Patuxent Wildlife Research Center.
Scridel, D., Brambilla, M., Martin, K., Lehikoinen, A., Iemma, A., Matteo, A., et al. (2018). A review and meta-analysis of the effects of climate change on Holarctic mountain and upland bird populations. Ibis 160, 489–515. doi: 10.1111/ibi.12585
Sienkiewicz, P., and Żmihorski, M. (2012). The effect of disturbance caused by rivers flooding on ground beetles (Coleoptera: Carabidae). Eur. J. Entomol. 109, 535–541. doi: 10.14411/eje.2012.067
Siikamäki, P. (1996). Nestling growth and mortality of Pied Flycatchers (Ficedula hypoleuca) in relation to weather and breeding effort. Ibis 138, 471–478. doi: 10.1111/j.1474-919X.1996.tb08067.x
Simons, M. J. P., Winney, I., Nakagawa, S., Burke, T., and Schroeder, J. (2015). Limited catching bias in a wild population of birds with near-complete census information. Ecol. Evol. 5, 3500–3506. doi: 10.1002/ece3.1623
Skagen, S. K., and Yackel Adams, A. A. (2012). Weather effects on avian breeding performance and implications of climate change. Ecol. Appl. 22, 1131–1145. doi: 10.1890/11-0291.1
Smith, J. N. M., Cook, T. L., Rothstein, S. I., Robinson, S. K., and Sealy, S. G. (eds) (2000). Ecology and Management of Cowbirds and Their Hosts. Austin: University of Texas Press.
Sohl, T. L. (2014). The relative impacts of climate and land-use change on conterminous United States bird species from 2001 to 2075. PLoS One 9:e112251. doi: 10.1371/journal.pone.0112251
Stanton, R. L., Morrissey, C. A., and Clark, R. G. (2018). Analysis of trends and agricultural drivers of farmland bird declines in North America: a review. Agric. Ecosyst. Environ. 254, 244–254. doi: 10.1016/j.agee.2017.11.028
Suedkamp Wells, K. M., Ryan, M. R., Millspaugh, J. J., and Hubbard, M. W. (2007). Survival of postfledgling grassland birds in Missouri. Condor 109, 781–794. doi: 10.1093/condor/109.4.781
Takagi, M. (2001). Some effects of inclement weather conditions on the survival and condition of bull-headed shrike nestlings. Ecol. Res. 16, 55–63. doi: 10.1046/j.1440-1703.2001.00371.x
Temple, S. A. (2020). “Dickcissel (Spiza americana), version 1.0,” in Birds of the World, eds A. F. Poole and F. B. Gill (Ithaca, NY: Cornell Lab of Ornithology), doi: 10.2173/bow.dickci.01
Thomson, D. L., Conroy, M. J., Anderson, D. R., Burnham, K. P., Cooch, E. G., Francis, C. M., et al. (2009). Standardising terminology and notation for the analysis of demographic processes in marked populations. Environ. Ecol. Stat. 3, 1099–1106. doi: 10.1007/978-0-387-78151-8_51
Travis, J. M. J. (2003). Climate change and habitat destruction: a deadly anthropogenic cocktail. Proc. R. Soc. B. Biol. Sci. 270, 467–473. doi: 10.1098/rspb.2002.2246
USGCRP (2018). “Impacts, risks, and adaptation in the United States,” in Fourth National Climate Assessment, eds D. R. Reidmiller, C. W. Avery, D. R. Easterling, K. E. Kunkel, K. L. M. Lewis, et al. (Washington, DC: U.S. Global Change Research Program), doi: 10.7930/NCA4.2018
van de Ven, T. M. F. N., McKechnie, A. E., Er, S., and Cunningham, S. J. (2020). High temperatures are associated with substantial reductions in breeding success and offspring quality in an arid-zone bird. Oecologia 193, 225–235. doi: 10.1007/s00442-020-04644-6
Villegas, J. C., Breshears, D. D., Zou, C. B., and Royer, P. D. (2010). Seasonally pulsed heterogeneity in microclimate: phenology and cover effects along deciduous grassland–forest continuum. Vadose Zone J. 9, 537–547. doi: 10.2136/vzj2009.0032
Welch-Acosta, B. C., Skipper, B. R., and Boal, C. W. (2019). Comparative breeding ecology of Mississippi Kites in urban and exurban areas of West Texas. J. Field Ornithol. 90, 248–257. doi: 10.1111/jofo.12303
White, R. P., Murray, S., and Rohweder, M. (2000). Pilot Analysis of Global Ecosystems: Grassland Ecosystems. Washington, DC: World Resources Institute.
Wiens, J. A. (1973). Pattern and process in grassland bird communities. Ecol. Monog. 43, 237–270. doi: 10.2307/1942196
Wiens, J. A. (1974). Climatic instability and the “ecological saturation” of bird communities in North American grasslands. Condor 76, 385–400. doi: 10.2307/1365813
Williams, B. K., Nichols, J. D., and Conroy, M. J. (2002). Analysis and Management of Animal Populations: Modeling, Estimation, and Decision Making. San Diego, CA: Academic Press.
Williams, G. P. (1978). The Case of the Shrinking Channels: The North Platte and Platte Rivers in Nebraska. U.S. Geological Survey Circular. Washington, DC: U.S. Government Printing Office. doi: 10.3133/cir781
Winter, M. (1998). Effect of Habitat Fragmentation on Grassland-nesting Birds in Southwestern Missouri. Ph.D. dissertation University of Missouri.
With, K. A., King, A. W., and Jensen, W. E. (2008). Remaining large grasslands may not be sufficient to prevent grassland bird declines. Biol. Conserv. 141, 3152–3167. doi: 10.1016/j.biocon.2008.09.025
Wuebbles, D. J., Kunkel, K., Wehner, M., and Zobel, Z. (2014). Severe weather in United States under a changing climate. Eos Trans. Am. Geophys. Union 95, 149–150. doi: 10.1002/2014EO180001
Zalik, N. J., and Strong, A. M. (2008). Effects of hay cropping on invertebrate biomass and the breeding ecology of Savannah Sparrows (Passerculus sandwichensis). Auk 125, 700–710. doi: 10.1525/auk.2008.07106
Zimmerman, J. L. (1982). Nesting success of dickcissels (Spiza americana) in preferred and less preferred habitats. Auk 99, 292–298. doi: 10.1093/auk/99.2.292
Zimmerman, J. L. (1983). Cowbird parasitism of dickcissels in different habitats and at different nest densities. Wils. Bull. 95, 7–22.
Zimmerman, J. L. (1992). Density-independent factors affecting the avian diversity of the tallgrass prairie community. Wils. Bull. 104, 85–94.
Keywords: climate change, weather, grassland breeding birds, Monitoring Avian Productivity and Survivorship (MAPS), grazing, haying, dickcissel (Spiza americana), brown-headed cowbird (Molothrus ater)
Citation: Rosamond KM, Goded S, Soultan A, Kaplan RH, Glass A, Kim DH and Arcilla N (2020) Not Singing in the Rain: Linking Migratory Songbird Declines With Increasing Precipitation and Brood Parasitism Vulnerability. Front. Ecol. Evol. 8:536769. doi: 10.3389/fevo.2020.536769
Received: 21 February 2020; Accepted: 29 September 2020;
Published: 26 November 2020.
Edited by:
Mark C. Mainwaring, University of Montana, United StatesReviewed by:
Jose A. Masero, University of Extremadura, SpainJennifer Anne Border, British Trust for Ornithology, United Kingdom
Copyright © 2020 Rosamond, Goded, Soultan, Kaplan, Glass, Kim and Arcilla. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nico Arcilla, bmljby5hcmNpbGxhQGF5YS55YWxlLmVkdQ==
 Kristen M. Rosamond
Kristen M. Rosamond Sandra Goded
Sandra Goded Alaaeldin Soultan
Alaaeldin Soultan Rachel H. Kaplan
Rachel H. Kaplan Alex Glass
Alex Glass Daniel H. Kim
Daniel H. Kim Nico Arcilla
Nico Arcilla