Abstract
Background:
Numerous investigations have underscored the causal effect between chronic pain (CP) and gut microbiota, jointly contributing to the onset and development of widespread CP. Nonetheless, there was still uncertainty about the causal effect between gut microbiota and chronic regional pain (CRP).
Methods:
Genome-wide association study (GWAS) summary data of gut microbial taxa (MiBioGen Consortium: 211 microbiotas and the Dutch Microbiome Project: 207 microbiotas) and eight types of CRP were used to reveal the causal effect between persistent pain in a specific region of the body and gut microbiota. A two-sample bidirectional Mendelian randomization (MR) design was used. In order to ensure the accuracy of the results, multiple sensitivity analyses were employed.
Results:
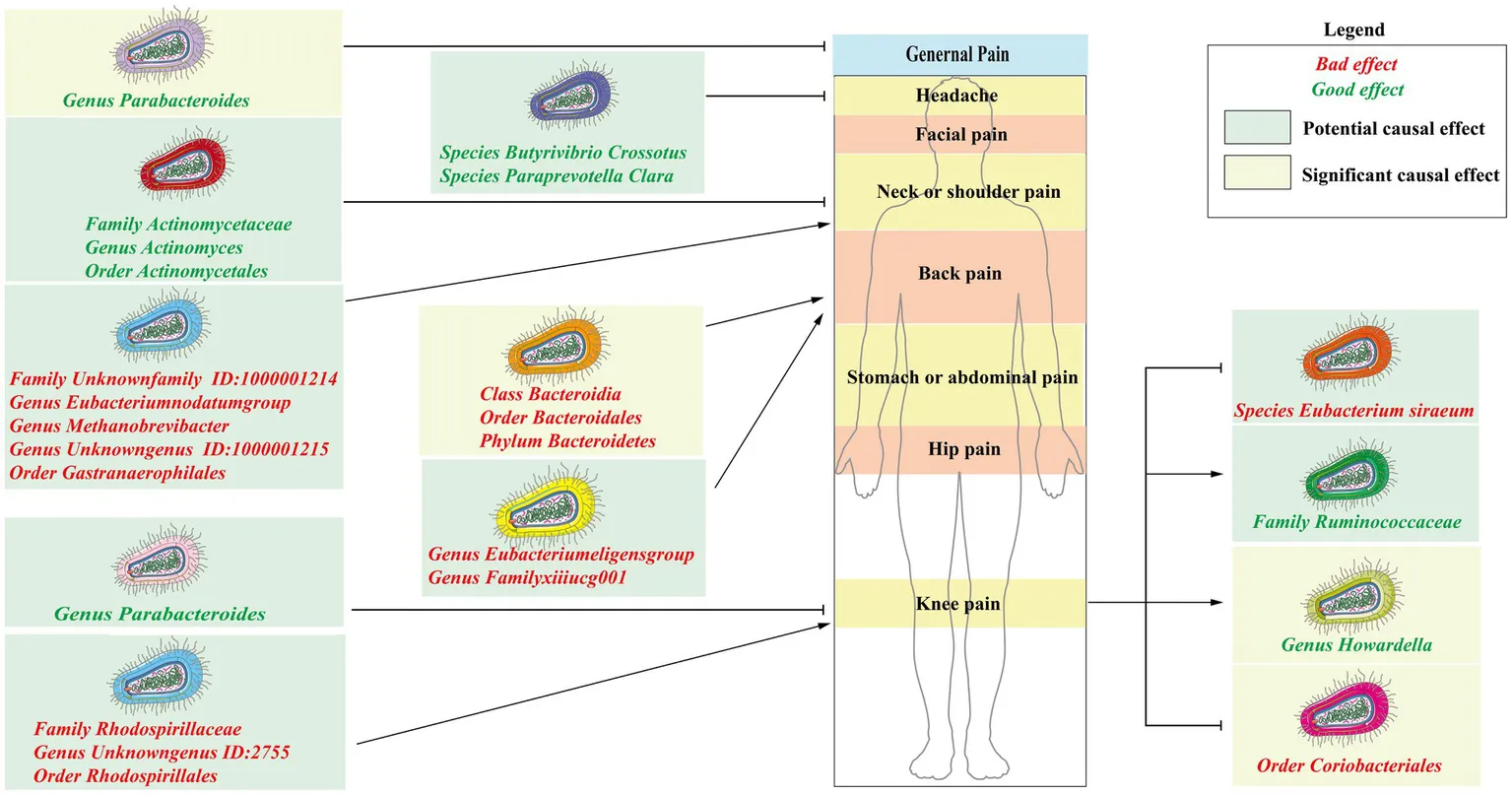
This study uncovered significant causal associations between six gut microbial taxa and three types of CRP (forward: Genus Parabacteroides for general pain; Class Bacteroidia, Order Bacteroidales, and Phylum Bacteroidetes for back pain. Reverse: knee pain for Genus Howardella and Order Coriobacteriales) by forward and reverse MR analysis. These findings had been verified by a rigorous Bonferroni correction. Furthermore, this research identified 19 microbial taxa that exhibited potential correlations with four types of CRP. There are no significant or potential gut microbiotas that were associated with other types of CRP, including fascial pain, stomach or abdominal pain, and hip pain.
Conclusion:
This two-sample bidirectional MR analysis unveiled the causality between gut microbial taxa and eight CRP conditions. The findings reveal the interplay between CRP and 6 gut microbiotas while also delineating 19 potential specific microbial taxa corresponding to diverse locations of persistent pain.
Highlights
Gut microbes play a causal role in chronic regional pain (CRP).
Six gut microbes had a significant impact on three subtypes of CRP, including general pain, back pain, and knee pain.
Nineteen microbial taxa are potentially associated with four subtypes of CRP, including headache, neck or shoulder pain, back pain, and knee pain.
Introduction
Chronic pain (CP), according to the definition of the International Association for the Study of Pain (IASP), is defined as persistent pain lasting for more than 3 months (Cohen et al., 2021). CP encompasses an unfavorable sensory and emotional encounter linked with, or similar to, real or potential tissue harm (Cohen et al., 2021). CP emerges as a clinical dilemma on the global horizon. Contemporary research underscores that over 30% of the global population is affected by CP (Cohen et al., 2021). However, until now, effective therapeutic approaches and interventional differentiation for CP remain lacking (Liu et al., 2023).
In the past decade, studies have progressively shed light on the crucial role of gut dysbiosis in the origin and advancement of various diseases (Tilg et al., 2016). Emerging evidence hints at the potential and pivotal role of the gut microbiota in distinct subtypes of CP, including headaches (Chen et al., 2019), abdominal pain (Newlove-Delgado et al., 2019), chronic widespread musculoskeletal pain (Freidin et al., 2021), and neuropathic discomfort (Yang et al., 2019). The research suggests that a dysregulation in the balance between beneficial and harmful bacteria in the gut can lead to an increase in intestinal permeability, allowing inflammatory substances and immune antigens to enter the body. This, in turn, results in the occurrence of systemic inflammation (Di Vincenzo et al., 2023). Such persistent and low-intensity chronic inflammation is considered a significant factor in the development of CP (Zhou et al., 2021). Nevertheless, currently, there is a lack of comprehensive causal association analysis between gut microbiota and chronic regional pain (CRP), such as neck pain and shoulder pain. In addition, ongoing research is susceptible to diverse confounding elements, spanning factors such as nutritional status, sleep cycles, and physical activity (Gonzalez-Alvarez et al., 2023). The intricate associations between distinct gut microbiota compositions and CP further complicate the exploration of their interplay. Based on the above background, we propose a hypothesis: there is an association between gut microbiota and distinct subtypes of CRP.
Mendelian randomization (MR) is a novel method of genetic epidemiology. It used instrumental variables (IVs), which are strongly associated with exposure, and IVs to make causal inferences and excluded confounding factors (Emdin et al., 2017; Davies et al., 2018). Utilizing MR to investigate causality necessitates satisfying three fundamental assumptions. First, IVs must exhibit a robust correlation with the exposure; second, IVs should not be correlated with confounding factors; and third, IVs can only impact outcomes through exposure (Figure 1; Bowden and Holmes, 2019). Furthermore, single-nucleotide polymorphisms (SNPs) could influence phenotypes, and the inability of phenotypes to alter SNPs allowed MR to eliminate interference from reverse causation (Bowden et al., 2015). In its succinct essence, MR can efficiently alleviate the influence of confounding and reverse causation (Emdin et al., 2017; Davies et al., 2018).
Figure 1
In conclusion, our study systematically investigated the associations between eight types of CRP and individual gut microbial communities. We provided a novel interpretative perspective for unraveling the intricate causal relationships between CRP and the gut microbiota.
Methods
Study design
In this study, SNPs strongly associated with exposure were used as IVs to explore the causal association between CRP and the gut microbiome. Due to allele frequency, disease prevalence may be different in different populations. Therefore, exposure and outcome need to be from the same race in MR analysis, which can rule out interference caused by population differences. At present, the publicly available intestinal flora databases are the main source of European populations, so this study selected European populations as the study population. The genome-wide association study (GWAS) summary data from two sets of gut microbiomes were employed (the MiBioGen Consortium [MC] and the Dutch Microbiome Project [DMP]). Initially, gut microbiomes were identified as the exposure, while eight distinct subtypes of CRP were designated as the outcomes for the MR analysis. Then, reverse MR analysis was employed to detect any reverse causal effect of CRP on gut microbiota. The flowchart is shown in Figure 2. The Strengthening the Reporting of Observational Studies in Epidemiology-Mendelian Randomization (STROBE-MR) checklist was used and interpreted in our study (Supplementary Table S1; Skrivankova et al., 2021).
Figure 2
GWAS data of gut microbiome
First, GWAS summary data were obtained from MC (Kurilshikov et al., 2021). This study included 18,340 individuals from 24 cohorts (Kurilshikov et al., 2021). To the best of our knowledge, this GWAS summary data represent the largest dataset of gut microbiome available at the current stage. Three distinct regions (V1–V2, V3–V4, and V4) of the 16S rRNA gene were employed to analyze the gut microbiota composition (Kurilshikov et al., 2021). Genetic variants influencing the relative abundance of microbial taxa were identified using microbiota quantitative trait loci (mbQTL) mapping (Kurilshikov et al., 2021). Finally, 131 genera, 35 families, 20 orders, 16 classes, and 9 phyla were defined. Nonetheless, this GWAS data lack species-level gut microbiota data. Consequently, we integrated data from the DMP to augment our analysis (Lopera-Maya et al., 2022). The GWAS summary data for DMP comprised a comprehensive set of 5 phyla, 10 classes, 13 orders, 26 families, 48 genera, and 105 species. In this study, 7,738 individuals of European descent were included (Lopera-Maya et al., 2022). The detailed information is shown in Table 1.
Table 1
| Trait | Sample size | Population | Data source |
|---|---|---|---|
| Gut microbiome | |||
| MiBioGen Consortium | 18,340 | European (N = 13,266), American Hispanic/Latin (N = 1,097), East Asian (N = 811), Middle-Eastern (N = 481), African American (N = 114), Complex ancestry (N = 2,571) | https://www.mibiogen.org |
| Phylum | |||
| Class | |||
| Order | |||
| Family | |||
| Genus | |||
| Dutch microbiome project | 7,738 | European (7,738 people) | https://dutchmicrobiomeproject.molgeniscloud.org/ |
| Phylum | |||
| Class | |||
| Order | |||
| Family | |||
| Genus | |||
| Species | |||
| Chronic pain | https://gwas.mrcieu.ac.uk/datasets | ||
| Back pain | 117,404 | European | Ben Elsworth |
| 84,489 | European | Neale lab | |
| Facial pain | 6,510 | European | Neale lab |
| General pain | 5,473 | European | Neale lab |
| Headache | 65,583 | European | Neale lab |
| 91,269 | European | Ben Elsworth | |
| Hip pain | 51,516 | European | Ben Elsworth |
| Knee pain | 97,889 | European | Ben Elsworth |
| 70,992 | European | Neale lab | |
| Neck or shoulder pain | 105,396 | European | Ben Elsworth |
| 75,651 | European | Neale lab | |
| Stomach or abdominal pain | 38,911 | European | Ben Elsworth |
Details of GWAS summary data.
The detailed information about the subtypes of chronic regional pain are found in Supplementary Table S2.
GWAS data of chronic regional pain
The data for CRP were sourced from the IEU open GWAS project (v7.5.5-2023-08-09, n = 42,348). Pain persisting for more than 3 months was meticulously screened, encompassing general pain, headaches, facial pains, neck or shoulder pain, stomach or abdominal pain, back pain, knee pain, and hip pain. All individuals included in this study were of European ancestry, and comprehensive details regarding CRP are provided in Table 1 and Supplementary Table S2.
The selection of IVs
For screening IVs, we rigorously adhere to the three fundamental assumptions of MR to ensure the accuracy of causality inference. SNPs, based on the criterion that a p-value of <1 × 10−5 was selected, were significantly associated with gut microbiota as potential eligible IVs (Yu et al., 2023). Then, to exclude the effect of linkage disequilibrium (LD), these SNPs, based on the European-based 1,000 Genome Projects reference panel, were clumped (r2 = 0.01, window size = 500 kb) (Xu et al., 2021). Finally, palindromic alleles were removed. For the reverse MR analysis, IVs of CRP were filtered. We chose a p-value of ≤5 × 10−6 as the criterion since the p-value of ≤5 × 10−8 was too strict. Then, the criterion of clump is reset (r2 = 0.001, window size = 10,000 kb) (Yu et al., 2023). Based on the following equation, we computed the F-values for each microbial community, which effectively mitigates potential bias from weak instruments (Xu et al., 2021):
R2 is to explain the exposure variance of the IVs, n is the sample size, and k is the number of IVs (Xu et al., 2021). When the F-value exceeds 10, it indicates the absence of a weak instrument bias (Pierce et al., 2011). Microbial communities with F-values less than 10 will be excluded.
MR and reverse MR analysis
Five main MR analysis methods, including inverse-variance weighted (IVW), MR Egger, weighted median, simple mode, and weighted mode, were used to make causal inferences between gut microbiota and CRP. It was important to note that the accuracy of the IVW results depended on the validity of all IVs. Thus, the result of IVW could be affected by the pleiotropy and heterogeneity of IVs (Burgess et al., 2013). Nonetheless, IVW was still the most accurate method where these influences were absent (Chen et al., 2020). For evaluating horizontal pleiotropy and heterogeneity, we employed three methods, including the MR-Egger Intercept Test, Mendelian Randomization Pleiotropy RESidual Sum and Outlier (MR-PRESSO) global test, and Cochran’s Q-test (Greco et al., 2015; Long et al., 2023). For the MR-PRESSO method, it could detect the overall horizontal pleiotropy of IVs; furthermore, it could detect the abnormal SNPs that caused the existence of pleiotropy (Verbanck et al., 2018). The p-value of two methods (MR-Egger Intercept Test and MR-PRESSO) greater than 0.05 displayed that pleiotropy was absent. In addition, Cochran’s Q-test was employed to detect heterogeneity, which indicated non-heterogeneity with p-values above 0.05. Microbial communities exhibiting horizontal pleiotropy or heterogeneity will be excluded. Therefore, we excluded interference from heterogeneity and horizontal pleiotropy.
The positive result mainly depended on the method of IVW (Burgess et al., 2013). When the estimated value (beta) of the IVW aligns with that of the other four methods, even if only IVW < 0.05, we still consider this result to be meaningful (Wang et al., 2023). After harmonization, it is required to have a minimum match with at least three IVs for subsequent analysis. Therefore, if the matched SNPs are fewer than three, we refrain from conducting MR analysis, as this could lead to inaccurate results. Meanwhile, leave-one-out analysis was conducted to assess whether the estimate value of MR analysis was influenced or biased by a single SNP.
Additionally, to mitigate the impact of false positives in multiple tests, we employed Bonferroni correction, setting distinct statistically significant p-value thresholds based on microbial classification (phylum, class, order, family, and genus). For MC, the significant p-value thresholds were as follows: 0.00038 (genera), 0.0014 (families), 0.0025 (orders), 0.0031 (classes), and 0.0056 (phyla). For DMP, the significant p-values were as follows: 0.01 (phyla), 0.005 (classes), 0.0038 (orders), 0.0019 (families), 0.001 (genera), and 0.00048 (species). Subsequently, when the estimated directions of the five MR analysis methods are consistent and the IVW is below the threshold set by Bonferroni correction, the microbial community is considered to have yielded a positive result. To further disclose this potential correlation, microbial taxa that were associated with at least two different datasets in one phenotype, under the condition of IVW < 0.05, were considered to have a potentially more reliable and robust correlation. Furthermore, the analytical method of reverse MR analysis was the same as MR analysis. All analyses were accomplished by R.4.2.31 and implemented by the packages TwoSampleMR (version 0.5.6), MR (version 0.7.0), and MRPRESSO (version 1.0).
Results
Instrumental variables
For MC, 231, 509, 1740, 289, and 126 SNPs associated with gut microbiota at different levels were identified (class, family, genus, order, and phylum; Supplementary Table S3). For DMP, 101, 257, 447, 118, 58, and 983 SNPs at different levels were also identified (class, family, genus, order, phylum, and species; Supplementary Table S4). For CRP, 111 SNPs were screened (Supplementary Table S5).
Causal effects of MC on eight types of CRP
Before Bonferroni correction, our analysis revealed an association between 58 microbial taxa and CP (Supplementary Table S6). However, after applying the Bonferroni correction, only one was observed as a statistically significant microbial taxa (Genus Parabacteroides for general pain, OR: 0.82, 95% CI: 0.74–0.91, p = 0.00013, Figures 3; 4A).
Figure 3
Figure 4
Causal effects of DMP on eight types of CRP
For the DMP, before Bonferroni correction, our analysis identified a correlation between 40 microbial taxa and CP (Supplementary Table S7). Three microbial taxa were still significantly associated with CP (back pain) after Bonferroni correction, including Class Bacteroidia (OR: 1.02, 95% CI:1.01–1.04, p = 0.00027), Order Bacteroidales (OR:1.02, 95% CI:1.01–1.04, p = 0.00027), and Phylum Bacteroidetes (OR:1.02, 95% CI:1.01–1.04, p = 0.00027). This result was replicated across the two GWAS databases for back pain (Figures 3, 4B–G).
Causal effects of eight types of CRP on gut microbiota (MC and DMP)
In the GWAS data for MC, we identified 58 gut microbiotas potentially associated with CP, while in the DMP GWAS data, there were 48 gut microbiotas with potential links to CP (Supplementary Tables S8, S9). During the reverse MR analysis, only two SNPs, after harmonization, exhibited a significant association between facial pain and the gut microbiota (MC). Thus, according to our criteria, the analysis between gut microbiota (MC) and facial pain was unable to be executed. After Bonferroni correction, two microbiotas had a reverse causal effect on CRP (knee pain), as shown in Figure 3. Genus Howardella (OR:0.01, 95% CI:0.00–0.07, p = 1.6 × 10−5) was a protective factor for knee pain. For DMP, after Bonferroni correction, Order Coriobacteriales (OR:9.03, 95% CI:2.07–39.43, p = 0.0034) was found as a risk factor for knee pain (Figures 3, 4H,I).
Potential correlation between gut microbiota and CRP
We also identified, under the condition of IVW < 0.05, several potentially significant microbial taxa that are linked to CP (Figures 5A,B, 6A,B). Further analysis revealed that 19 microbial taxa showed a correlation with both distinct datasets (Figure 7).
Figure 5
Figure 6
Figure 7
For back pain, Genus Eubacterium eligens group (GWASID: ukb-b-8463, OR:1.03, 95% CI:1.00–1.06, p = 0.027; GWSID: ukb-a-346, OR:1.03, 95CI%: 1.00–1.06, p = 0.026) and Genus FamilyXIIIUCG001 (GWASID: ukb-b-8463, OR:1.03, 95% CI: 1.00–1.06, p = 0.026; GWASID: ukb-a-346, OR:1.04, 95% CI: 1.01–1.07, p = 0.0047) were potential risk factors (Figure 7).
For neck or shoulder pain, the different gut microbiomes also display that association (Figure 7). In gut microbiome from MC, the results showed that Family Unknown family ID:1000001214 (GWASID: ukb-b-16118, OR:1.02, 95% CI: 1.00–1.03, p = 0.039; GWASID: ukb-a-341, OR:1.02, 95% CI: 1.00–1.04, p = 0.026), Genus Eubacterium nodatum group (GWASID: ukb-b-16118, OR:1.01, 95% CI: 1.00–1.02, p = 0.026; GWASID: ukb-a-341, OR:1.01, 95% CI: 1.00–1.03, p = 0.027), Genus Methanobrevibacter (GWASID: ukb-b-16118, OR:1.02, 95% CI: 1.00–1.04, p = 0.015; GWASID: ukb-a-341, OR:1.02, 95% CI: 1.01–1.04, p = 0.0085), and Genus Unknown genus ID:1000001215 (GWASID: ukb-b-16118, OR:1.02, 95% CI: 1.00–1.03, p = 0.039; GWASID: ukb-a-341, OR:1.02, 95% CI: 1.00–1.04, p = 0.026) were risk factors. Family Actinomycetaceae (GWASID: ukb-b-16118, OR:0.98, 95% CI: 0.95–1.00, p = 0.027; GWASID: ukb-a-341, OR:0.96, 95% CI: 0.94–0.99, p = 0.004), Genus Actinomyces (GWASID: ukb-b-16118, OR:0.98, 95% CI: 0.96–1.00, p = 0.045; GWASID: ukb-a-341, OR:0.97, 95% CI: 0.95–0.99, p = 0.0076), Order Gastranaerophilales (GWASID: ukb-b-16118, OR: 1.02, 95% CI: 1.00–1.03, p = 0.039; GWASID: ukb-a-341, OR: 1.02, 95% CI: 1.00–1.04, p = 0.026), and Order Actinomycetales (GWASID: ukb-b-16118, OR:0.98, 95% CI: 0.95–1.00, p = 0.027; GWASID: ukb-a-341, OR:0.96, 95% CI: 0.94–0.99, p = 0.004) were potentially protective factors for neck or shoulder pain. In gut microbiome from DMP, we found that genetically predicted neck or shoulder pain was potentially related to a low abundance of Species Roseburia Unclassified (GWASID: ukb-b-16118, OR:0.99, 95% CI: 0.98–1.00, p = 0.03; GWASID: ukb-a-341, OR:0.99, 95% CI: 0.97–1.00, p = 0.01), Family Enterobacteriaceae (GWASID: ukb-b-16118, OR:0.98, 95% CI:0.96–1.00, p = 0.014; GWASID: ukb-a-341, OR:0.97, 95% CI:0.95–0.99, p = 0.016), and Order Enterobacteriales (GWASID: ukb-b-16118, OR:0.98, 95% CI: 0.96–1.00, p = 0.014; GWASID: ukb-a-341, OR:0.97, 95% CI: 0.95–0.99, p = 0.016).
For knee pain, we found that genetically predicted knee pain was potentially related to higher abundance of Family Rhodospirillaceae (GWASID: ukb-b-8906, OR:1.02, 95% CI: 1.00–1.03, p = 0.0082; GWASID: ukb-a-353, OR:1.02, 95% CI: 1.00–1.03, p = 0.025), Genus Unknown genus ID:2755 (GWASID: ukb-b-8906, OR:1.02, 95% CI: 1.00–1.03, p = 0.019; GWASID: ukb-a-353, OR:1.02, 95% CI: 1.00–1.04, p = 0.03), and Order Rhodospirillales (GWASID: ukb-b-8906, OR:1.02, 95% CI: 1.01–1.03, p = 0.0044; GWASID: ukb-a-353, OR:1.02, 95% CI: 1.00–1.04, p = 0.015). Notably, we found that genetically predicted knee pain was potentially related to a low abundance of Genus Parabacteroides (GWASID: ukb-b-8906, OR:0.97, 95% CI:0.94–0.99, p = 0.011; GWASID: ukb-a-353, OR:0.96, 95% CI:0.93–0.99, p = 0.022).
For headache, Species Butyrivibrio crossotus (GWASID: ukb-b-13092, OR:0.99, 95% CI: 0.98–1.00, p = 0.032; GWASID: ukb-a-354, OR:0.99, 95% CI: 0.97–1.00, p = 0.044), Species Paraprevotella Clara (GWASID: ukb-b-13092, OR:0.99, 95% CI: 0.97–1.00, p = 0.033; GWASID: ukb-a-354, OR:0.98, 95% CI: 0.96–0.99, p = 0.00084).
In reverse MR analysis, several microbial taxa were potentially influenced by CRP (Figures 6A,B). Further analysis of this association revealed that the Family Ruminococcaceae (GWASID: ukb-b-8906, OR:0.34, 95% CI: 0.13–0.88, p = 0.026; GWASID: ukb-a-353, OR:0.28, 95% CI: 0.11–0.70, p = 0.0071) was potentially correlated with both of knee’s distinct datasets (Figure 7). Species Eubacterium siraeum (GWASID: ukb-b-8906, OR:15.34, 95% CI: 2.17–108.52, p = 0.0062; GWASID: ukb-a-353, OR:10.23, 95% CI: 1.49–70.11, p = 0.018) also exhibited a correlation with both of knee’s distinct datasets (Figure 7).
Sensitivity analysis
The result of MR-PRESSO and MR Egger regression did not display horizontal pleiotropy among the significant and potential microbiota (Table 2; Supplementary Tables S10–S13). In addition, the result of Cochran’s Q-test also did not indicate heterogeneity (Table 2; Supplementary Tables S10–S13). Moreover, there is no single SNP, in leave-one-out analysis, that strongly violates the overall effect between the gut microbiome and CRP (Figure 8). Furthermore, all F-statistical values of significant and potential gut microbiotas exceeded 10 (Table 2; Supplementary Tables S10–S13).
Table 2
| Exposure | Outcome | Source | GWAS ID | MR Egger intercept test | MR-PRESSO Global test | Cochran’s Q-test | R2 | F-value |
|---|---|---|---|---|---|---|---|---|
| Forward MR analysis | ||||||||
| Genus Parabacteroides | General pain | MC | ukb-d-2956 | 0.513 | 0.855 | 0.787 | 0.019 | 32.88 |
| Class Bacteroidia | Back pain | DMP | ukb-a-346 | 0.549 | 0.445 | 0.415 | 0.037 | 23.08 |
| Order Bacteroidales | Back pain | DMP | ukb-a-346 | 0.549 | 0.446 | 0.415 | 0.037 | 23.08 |
| Phylum bacteroidetes | Back pain | DMP | ukb-a-346 | 0.551 | 0.445 | 0.415 | 0.037 | 23.07 |
| Class Bacteroidia | Back pain | DMP | ukb-b-8463 | 0.236 | 0.584 | 0.540 | 0.037 | 23.08 |
| Order Bacteroidales | Back pain | DMP | ukb-b-8463 | 0.236 | 0.584 | 0.540 | 0.037 | 23.08 |
| Phylum bacteroidetes | Back pain | DMP | ukb-b-8463 | 0.236 | 0.584 | 0.539 | 0.037 | 23.07 |
| Reverse MR analysis | ||||||||
| Knee pain | Genus Howardella | MC | ukb-a-353 | 0.798 | 0.649 | 0.552 | 0.002 | 22.77 |
| Knee pain | Order Coriobacteriales | DMP | ukb-a-353 | 0.450 | 0.405 | 0.331 | 0.002 | 22.98 |
Significant results of sensitivity analysis.
MR, Mendelian randomization; MC, MiBioGen Consortium; DMP, Dutch Microbiome Project.
Figure 8
Discussion
To our knowledge, this is the first systematic study that utilizes MR analysis to detect the causal effect between gut microbiota and eight types of CRP. While previous studies have explored the association between CP and gut microbiotas, these studies could be influenced by potential confounding factors, such as socioeconomic status and dietary habits (Davies et al., 2018). Additionally, these studies did not delve into the relationship between individual microbial taxa and CRP. Our study bridges this gap and provides new insights from a new perspective.
Our research reveals that Genus Parabacteroides is considered a protective factor against chronic widespread pain. It is worth noting that recent studies have suggested that Genus Parabacteroides may represent a novel type of gut probiotic (Wu, 2023). It has therapeutic effects on tumors, metabolic diseases, and inflammatory conditions (Wu, 2023). A recent study also displayed a reduction in the abundance of Genus Parabacteroides in the gut of rheumatoid arthritis patients. Genus Parabacteroides may alleviate rheumatoid arthritis by directly or indirectly affecting the differentiation of T helper 17 cells through secondary bile acids. Interestingly, rheumatoid arthritis patients often experience CP (Sun et al., 2023). Class Bacteroidia, Order Bacteroidales, and Phylum Bacteroidetes have not been reported to be associated with CRP. Our study, for the first time, demonstrated their relationship with chronic back pain. Importantly, these three microbial taxa passed the Bonferroni correction and showed consistency across two different GWAS datasets for the phenotype of persistent back pain. Therefore, we consider this conclusion to be highly reliable. Perhaps regulating the abundance of these three microbial taxa in the gut could be a means of preventing back pain. In reverse MR analysis, we found that knee pain leads to a higher abundance of Genus Howardella, while the abundance of Order Coriobacteriales decreases. Interestingly, previous research did not uncover the associations between these two microbial taxa and chronic knee pain.
In addition to significant microbiota, we also identified 19 microbial taxa associated with CRP. We observed some intriguing phenomena. Previous research has suggested that the Genus Eubacterium nodatum group and Genus Methanobrevibacter may be associated with abdominal pain (Kumpitsch et al., 2021; Jiang et al., 2022). In fecal samples from patients with nodular lymphoid hyperplasia, the abundance of the Genus Eubacterium nodatum group, compared to the control group, was significantly higher, and nodular lymphoid hyperplasia is often accompanied by persistent abdominal pain (Jiang et al., 2022). The genus Methanobrevibacter is considered a major contributor to methane production in the human gut. Individuals with high methane levels in their guts can have Genus Methanobrevibacter abundances up to 1,000 times higher than normal individuals (Kumpitsch et al., 2021). Elevated methane in the gut has been linked to symptoms such as abdominal pain, bloating, and constipation (Kumpitsch et al., 2021). However, our study revealed that an increased abundance of these microbial groups is more likely to be associated with neck and shoulder pain. Additionally, our study unearthed an intriguing phenomenon. We observed that chronic knee pain has an impact on four distinct gut microbiotas, including Genus Howardella, Order Coriobacteriales, Family Ruminococcaceae, and Species Eubacterium siraeum. Surprisingly, we did not uncover any supporting evidence for this in the existing literature. This could potentially be attributed to various factors, including mediating effects such as dietary habits, sleep patterns, and other contributing factors. Nevertheless, another paradoxical observation is the absence of alterations in the gut microbiota associated with other forms of CRP. This suggests the possibility of additional mechanisms at play in influencing the gut microbiota beyond those related to chronic knee pain. One plausible hypothesis is that these microbial communities participate in the occurrence and development of CP through boron metabolism. Recent studies have indicated that prebiotic boron complexes (PBCs), rich in boron, can engage in signal transmission between the host and microbial communities, maintaining a balance between beneficial and harmful bacteria in the gut (Fang et al., 2021). Simultaneously, they contribute to improving the permeability of the intestinal mucosa, preventing inflammatory substances from entering the bloodstream, and thereby impeding the onset of low-intensity chronic inflammation (Bita et al., 2023). Further research could shed light on their underlying mechanisms.
We must acknowledge several limitations in our study. First, to secure a sufficient number of IVs, we employed different selection criteria for gut microbiota and CRP. Additionally, we relaxed the screening threshold for CRP (p < 5 × 10−6), compared to the more stringent standard commonly used in the literature (p < 5 × 10−8). This decision may have potentially weakened the first assumption of MR. Second, the GWAS summary data in our study are derived from European ancestry, including both gut microbiota and CP data. Therefore, our findings for other ethnic groups might not be suitable. Third, while most of the GWAS data for MC are from individuals of European ancestry, the racial composition of the two datasets we employed is not entirely congruent. This incongruence may introduce some level of inconsistency in LD correlations. Fourth, in light of the limited availability of GWAS data for certain site-specific CP conditions, our established criteria were unable to reveal a potential association between microbial communities and the corresponding type of pain. Due to the limitations of MR studies, it is also not clear whether the gut microbiome and CRP are involved in the regulation of disease through certain substances acting as vectors. In addition, the description of pain data does make it impossible to identify the specific cause of pain, which may have a potential impact on our results. Finally, we were unable to access detailed individual-level information such as age, gender, inclusion, and exclusion criteria. Consequently, we could not conduct further stratified analyses.
Despite these limitations, we believe that our study provides novel insights into the potential associations between the gut microbiota and site-specific CP. It lays a solid foundation for future research endeavors, even as we acknowledge the need for caution in generalizing our conclusions, particularly to diverse populations and under varying circumstances.
In conclusion, through the utilization of publicly accessible genetic databases, we have uncovered reciprocal causal connections between particular gut microbiotas and CRP. Our discoveries strongly imply that there could be a direct correlation between distinct CRP and specific gut microbiota compositions. By modulating the biological abundance of the corresponding microbiota, precise treatment goals may be achieved.
Statements
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding authors.
Author contributions
Q-AX: Data curation, Formal analysis, Investigation, Writing – original draft. LQ: Methodology, Software, Writing – review & editing. JY: Resources, Visualization, Writing – review & editing. Y-TH: Formal analysis, Resources, Writing – review & editing. L-FA: Formal analysis, Methodology, Software, Writing – review & editing. D-CW: Conceptualization, Funding acquisition, Writing – review & editing. XX: Funding acquisition, Project administration, Supervision, Writing – original draft. X-LZ: Conceptualization, Supervision, Validation, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was supported by grants from the National Natural Science Foundation of China (no. 31772709) and the Hubei Natural Science Foundation (2015CFB316) of China.
Acknowledgments
The authors thank the MiBioGen Consortium and Dutch Microbiome Project, and the IEU Open GWAS project. The authors thank for the help of Servier Medical Art. Servier Medical Art by Servier is licensed under a Creative Commons Attribution 3.0 Unported License (https://creativecommons.org/license-s/by/3.0/). The authors thank https://www.chiplot.online/ for its help with heatmap. The author Q-AX thanks Wen-bin Bai and her help with the figure layout of the graphical abstract.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2024.1329521/full#supplementary-material
Footnotes
References
1
BitaC. E.ScoreiI. R.VrejuA. F.MusetescuA. E.MogosanuG. D.BitaA.et al. (2023). Microbiota-accessible boron-containing compounds in complex regional pain syndrome. Medicina (Kaunas)59. doi: 10.3390/medicina59111965
2
BowdenJ.Davey SmithG.BurgessS. (2015). Mendelian randomization with invalid instruments: effect estimation and Bias detection through Egger regression. Int. J. Epidemiol.44, 512–525. doi: 10.1093/ije/dyv080
3
BowdenJ.HolmesM. V. (2019). Meta-analysis and Mendelian randomization: a review. Res. Synth. Methods10, 486–496. doi: 10.1002/jrsm.1346
4
BurgessS.ButterworthA.ThompsonS. G. (2013). Mendelian randomization analysis with multiple genetic variants using summarized data. Genet. Epidemiol.37, 658–665. doi: 10.1002/gepi.21758
5
ChenX.KongJ.DiaoX.CaiJ.ZhengJ.XieW.et al. (2020). Depression and prostate Cancer risk: a Mendelian randomization study. Cancer Med.9, 9160–9167. doi: 10.1002/cam4.3493
6
ChenJ.WangQ.WangA.LinZ. (2019). Structural and functional characterization of the gut microbiota in elderly women with migraine. Front. Cell. Infect. Microbiol.9:470. doi: 10.3389/fcimb.2019.00470
7
CohenS. P.VaseL.HootenW. M. (2021). Chronic pain: an update on burden, best practices, and new advances. Lancet397, 2082–2097. doi: 10.1016/s0140-6736(21)00393-7
8
DaviesN. M.HolmesM. V.DaveyS. G. (2018). Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. BMJ362:k601. doi: 10.1136/bmj.k601
9
Di VincenzoF.Del GaudioA.PetitoV.LopetusoL. R.ScaldaferriF. (2023). Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern. Emerg. Med. doi: 10.1007/s11739-023-03374-w
10
EmdinC. A.KheraA. V.KathiresanS. (2017). Mendelian randomization. JAMA318, 1925–1926. doi: 10.1001/jama.2017.17219
11
FangJ.WangH.ZhouY.ZhangH.ZhouH.ZhangX. (2021). Slimy partners: the mucus barrier and gut microbiome in ulcerative colitis. Exp. Mol. Med.53, 772–787. doi: 10.1038/s12276-021-00617-8
12
FreidinM. B.StalteriM. A.WellsP. M.LachanceG.BaleanuA. F.BowyerR. C. E.et al. (2021). An association between chronic widespread pain and the gut microbiome. Rheumatology (Oxford)60, 3727–3737. doi: 10.1093/rheumatology/keaa847
13
Gonzalez-AlvarezM. E.Sanchez-RomeroE. A.TurroniS.Fernandez-CarneroJ.VillafaneJ. H. (2023). Correlation between the altered gut microbiome and lifestyle interventions in chronic widespread pain patients: a systematic review. Medicina (Kaunas)59:256. doi: 10.3390/medicina59020256
14
GrecoM. F.MinelliC.SheehanN. A.ThompsonJ. R. (2015). Detecting pleiotropy in Mendelian randomisation studies with summary data and a continuous outcome. Stat. Med.34, 2926–2940. doi: 10.1002/sim.6522
15
JiangQ. L.LuY.ZhangM. J.CuiZ. Y.PeiZ. M.LiW. H.et al. (2022). Mucosal bacterial Dysbiosis in patients with nodular lymphoid hyperplasia in the terminal ileum. World J. Gastroenterol.28, 811–824. doi: 10.3748/wjg.v28.i8.811
16
KumpitschC.FischmeisterF. P. S.MahnertA.LacknerS.WildingM.SturmC.et al. (2021). Reduced B12 uptake and increased gastrointestinal Formate are associated with Archaeome-mediated breath methane emission in humans. Microbiome9:193. doi: 10.1186/s40168-021-01130-w
17
KurilshikovA.Medina-GomezC.BacigalupeR.RadjabzadehD.WangJ.DemirkanA.et al. (2021). Large-scale association analyses identify host factors influencing human gut microbiome composition. Nat. Genet.53, 156–165. doi: 10.1038/s41588-020-00763-1
18
LiuL.WuQ.ChenY.RenH.ZhangQ.YangH.et al. (2023). Gut microbiota in chronic pain: novel insights into mechanisms and promising therapeutic strategies. Int. Immunopharmacol.115:109685. doi: 10.1016/j.intimp.2023.109685
19
LongY.TangL.ZhouY.ZhaoS.ZhuH. (2023). Causal relationship between gut microbiota and cancers: a two-sample Mendelian randomisation study. BMC Med.21:66. doi: 10.1186/s12916-023-02761-6
20
Lopera-MayaE. A.KurilshikovA.van der GraafA.HuS.Andreu-SanchezS.ChenL.et al. (2022). Effect of host genetics on the gut microbiome in 7,738 participants of the Dutch microbiome project. Nat. Genet.54, 143–151. doi: 10.1038/s41588-021-00992-y
21
Newlove-DelgadoT.AbbottR. A.MartinA. E. (2019). Probiotics for children with recurrent abdominal pain. JAMA Pediatr.173, 183–184. doi: 10.1001/jamapediatrics.2018.4575
22
PierceB. L.AhsanH.VanderweeleT. J. (2011). Power and instrument strength requirements for Mendelian randomization studies using multiple genetic variants. Int. J. Epidemiol.40, 740–752. doi: 10.1093/ije/dyq151
23
SkrivankovaV. W.RichmondR. C.WoolfB. A. R.YarmolinskyJ.DaviesN. M.SwansonS. A.et al. (2021). Strengthening the reporting of observational studies in epidemiology using Mendelian randomization: the Strobe-Mr statement. JAMA326, 1614–1621. doi: 10.1001/jama.2021.18236
24
SunH.GuoY.WangH.YinA.HuJ.YuanT.et al. (2023). Gut commensal Parabacteroides Distasonis alleviates inflammatory arthritis. Gut72, 1664–1677. doi: 10.1136/gutjnl-2022-327756
25
TilgH.CaniP. D.MayerE. A. (2016). Gut microbiome and liver diseases. Gut65, 2035–2044. doi: 10.1136/gutjnl-2016-312729
26
VerbanckM.ChenC. Y.NealeB.DoR. (2018). Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat. Genet.50, 693–698. doi: 10.1038/s41588-018-0099-7
27
WangS.ZhuH.PanL.ZhangM.WanX.XuH.et al. (2023). Systemic inflammatory regulators and risk of acute-on-chronic liver failure: a bidirectional Mendelian-randomization study. Front. Cell Dev. Biol.11:1125233. doi: 10.3389/fcell.2023.1125233
28
WuW. K. K. (2023). Parabacteroides Distasonis: an emerging probiotic?Gut72, 1635–1636. doi: 10.1136/gutjnl-2022-329386
29
XuQ.NiJ. J.HanB. X.YanS. S.WeiX. T.FengG. J.et al. (2021). Causal relationship between gut microbiota and autoimmune diseases: a two-sample Mendelian randomization study. Front. Immunol.12:746998. doi: 10.3389/fimmu.2021.746998
30
YangC.FangX.ZhanG.HuangN.LiS.BiJ.et al. (2019). Key role of gut microbiota in anhedonia-like phenotype in rodents with neuropathic pain. Transl. Psychiatry9:57. doi: 10.1038/s41398-019-0379-8
31
YuH.WanX.YangM.XieJ.XuK.WangJ.et al. (2023). A large-scale causal analysis of gut microbiota and delirium: a Mendelian randomization study. J. Affect. Disord.329, 64–71. doi: 10.1016/j.jad.2023.02.078
32
ZhouW. B. S.MengJ.ZhangJ. (2021). Does low grade systemic inflammation have a role in chronic pain?Front. Mol. Neurosci.14:785214. doi: 10.3389/fnmol.2021.785214
Summary
Keywords
gut microbiome, Mendelian randomization, chronic regional pain, causality, genome-wide association study
Citation
Xiao Q-A, Qin L, Yu J, Hu Y-T, Ai L-F, Wang D-C, Xia X and Zhang X-L (2024) The causality between gut microbiome and chronic regional pain: a Mendelian randomization analysis. Front. Microbiol. 15:1329521. doi: 10.3389/fmicb.2024.1329521
Received
13 November 2023
Accepted
05 February 2024
Published
29 February 2024
Volume
15 - 2024
Edited by
Giuseppe Murdaca, University of Genoa, Italy
Reviewed by
Gopinath Venugopal, University of Arkansas for Medical Sciences, United States
Shikha Sharma, National Institutes of Health (NIH), United States
Updates
Copyright
© 2024 Xiao, Qin, Yu, Hu, Ai, Wang, Xia and Zhang.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiao-Lin Zhang, zhangxiaolin5800@163.comXuan Xia, xiaxuan_2004@163.com
†ORCID: Qing-Ao Xiao, https://orcid.org/0000-0002-7853-7794Lei Qin, https://orcid.org/0009-0000-4440-5884Jin Yu, https://orcid.org/0009-0007-1123-280XYin-Tao Hu, https://orcid.org/0009-0006-8293-7623Lin-Feng Ai, https://orcid.org/0009-0004-7459-6449De-Cheng Wang, https://orcid.org/0000-0002-1742-7460Xuan Xia, https://orcid.org/0000-0002-4788-1971Xiao-Lin Zhang, https://orcid.org/0000-0002-5189-9701
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.