- 1Institute of Gastroenterology, Guangzhou University of Chinese Medicine, Guangzhou, China
- 2Science and Technology Innovation Center, Guangzhou University of Chinese Medicine, Guangzhou, China
- 3Medical College of Acu-Moxi and Rehabilitation, Guangzhou University of Chinese Medicine, Guangzhou, China
Background/Aims: Traditional Herbal Medicines (THM) have been being used for gastroesophageal reflux disease (GERD) for a long time, but clinical evidence is still scarce. We evaluated different THM prescriptions for GERD in adults.
Methods: Data added to nine online databases from their inception to November 30, 2019, were systematically searched. All relevant randomized controlled trials (RCTs) were included and were combined with Bayesian network analysis. The Cochrane Collaboration’s risk of bias tool and GRADE profiler version 3.6 were respectively employed to evaluate the quality of evidence of outcomes.
Results: Seventeen publications involving 1441 participants were retrieved. The results of our analysis suggested that Jianpi therapy+proton pump inhibitors (PPIs) and Ligan Hewei therapy respectively ranked first in overall clinical efficacy and efficacy under gastroscope; Ligan Hewei therapy+PPIs was the optimum intervention in the improvement of acid regurgitation and heartburn.
Conclusion: This research indicates that Ligan Hewei therapy and Jianpi therapy, or these therapies separately combined with PPIs, should be recommended as appropriate complementary and alternative treatments based on the specific characteristics of GERD. However, additional well-designed RCTs with high methodological quality are still needed for future research.
Introduction
Gastroesophageal reflux disease (GERD) is a common chronic disorder characterized by an imbalance of the barrier between the stomach and the esophagus, resulting in the regurgitation of gastric contents into the esophagus amd even the hypopharynx (DeVault and Castell, 2005). Based on the Montreal definition published in 2006, it is subclassified into non-erosive reflux disease (NERD), reflux esophagitis (RE), and Barrett esophagus (BE) (Vakil et al., 2006). Moreover, epidemiological investigation showed that this disease affected approximately 20%~30% of the population around the world and 7.8%~8.8% in East Asia (El-Serag et al., 2014). Without timely treatment, patients with the condition will suffer from numerous complications including esophageal stricture, ulceration, and even BE (Freston et al., 1995; Schwizer and Fried, 1997), thereby leading to huge psychological burden and poor work productivity (Wahlqvist et al., 2008; Nocon et al., 2009).
Currently, the first-line medical drug for GERD is proton pump inhibitors (PPIs). They are estimated to provide about a 56%~76% rate of relief of related symptoms and an 80%~85% recovery rate for esophageal lesions (Katz et al., 2006) as well as reducing the incidence of complications (Savarino et al., 2009). However, approximately 30% GERD sufferers, who had unsatisfactory responses to PPIs still remained symptomatic and had high risk of complications, including BE (Fass et al., 2005). Therefore, in order to seek other effective therapies and improve their quality of life, many patients put their attention on alternative medicine (Patrick, 2011).
The use of traditional Chinese medicine (TCM) has a long history and was first documented by the Sheng Nong Classic of Materia Medica. Currently, traditional Herbal Medicines (THM) are widely used in cardio-cerebrovascular, endocrine, gastrointestinal, neuropsychiatric, and respiratory disorders (Dai et al., 2018; Jiang et al., 2019; Kong et al., 2019; Qin et al., 2019; Gao et al., 2020; Liu et al., 2020; Zhang et al., 2020). Several studies have evaluated the efficacy and safety of THM in treating GERD (Ling et al., 2015; Dai et al., 2017; Shih et al., 2019). However, these findings were obtained from pairwise comparisons between a prescription and conventional Western medicine(s). No comparison within different THM was conducted in the treatment of GERD. Consequently, to obtain up-to-date information regarding the effectiveness of different prescriptions in treating this disorder, a Bayesian network analysis that integrated direct with indirect evidence for multiple intervention comparisons was performed in this study.
Methods
This study was performed based on the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISRMA) (Liberati et al., 2009) statement and the Cochrane Handbook for the Systematic Review of interventions (details via http://training/cochrane.org/handbook).
Data Sources and Search Strategy
We systematically searched the following databases from their inception to November 30, 2019: PubMed, MEDLINE, EMBASE, Cochrane Library, Scopus, Clarivate, and the Chinese databases of CNKI, WanFang, and VIP for relevant literature. The pre-established search terms consisted of three parts: strategies for GRED, THM treatment, and a specific filter for randomized controlled trials. Both Medical Subject Headings (MeSH) terms and text words were used for keywords. The detailed search strategies for each database are shown in Supplementary Table S1. No limitation was placed on language of article. Any omission of publications was remedied by manual retrieval. To obtain eligible trials, the reference lists of the included studies were checked for verification and further assessment.
Study Selection
Following the PICOS (participants, interventions, comparisons, outcomes, and study design) criteria, two investigators (Yun-kai Dai, Yun-bo Wu) preliminarily screened the relevant titles and abstracts. Randomized, parallel-group clinical trials of THM for GERD were initially included. The full texts of these studies were then scanned for further evaluation. Briefly, participants over the age of 18 should meet the diagnosis criteria of GERD (DeVault and Castell, 2005). Any prescription of THM interventions and certain positive controls (PPIs, or gastrointestinal motility drugs (GMD), or combinations) were selected. Meanwhile, the sample size of each trial should not be less than 30/arm, and the duration of treatment should be at least 4 weeks. In order to obtain superior quality literature, works with a Jadad score above 1 was screened.
However, some participants or publications were excluded: pregnant women, patients with comorbidities such as severe cardio-cerebro-vascular diseases and cancers, published meeting abstracts, non-research articles and cross-over studies, and THM as positive control.
Data Abstraction and Quality Evaluation
Using a prepiloted data extraction sheet, two researchers (Yun-kai Dai, Yun-bo Wu) independently conducted data abstraction and quality assessment. Relevant characteristics of participants (gender, age, and sample size), details of interventions and comparisons (regimen for treatment and duration), course of disease, primary outcomes (overall clinical efficacy and efficacy under gastroscope) and secondary outcomes (improvements of acid regurgitation and heartburn, reflux diagnostic questionnaire (RDQ) scores), side effects, and study design were extracted, as was the classification of GERD. Moreover, relevant missing information could be acquired if necessary through telephoning the corresponding authors.
On the basis of the Cochrane Collaboration Recommendations assessment tool (Savovic et al., 2018), the quality of the included trials was independently evaluated by two reviewers (Yun-kai Dai, Yun-bo Wu). Overall evaluation of methodological quality had seven aspects: (i) random sequence generation; (ii) allocation concealment; (iii) blinding of participants and personnel; (iv) blinding (or masking) of outcomes assessment; (v) incomplete outcome data; (vi) selective reporting; (vii) other bias. Disagreements were resolved by further discussion or negotiation. For the methodological quality attribute of each study, the value “high quality” (or low risk), “uncertain quality” (or unclear risk), or “low quality” (or high risk) was assigned to calculate the overall score, which ranged from 0 to 6 points (from worst to best methodological quality). In view of this, the distributions of the methodological qualities on different comparisons across the evidence network were assessed. In addition, the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) profiler version 3.6 was used to analyze the overall evidence quality of this network analysis.
Statistical Analysis
Evidence of direct and indirect multiple-intervention comparisons is obtained by network meta-analysis, and performing this analysis with the Bayesian framework can improve the accuracy of the results. WinBUGS version 1.4.3 (MRC Biostatistics Unit, Cambridge, UK), based on the Bayesian framework and the Markov chain Monte Carlo (MCMC) method, was used to assess and process research data a priori. We used non-informative uniform and normal prior distributions (Ades et al., 2006; Sutton et al., 2008) and three Markov chains to fit the model. Meanwhile, 50,000 simulation iterations and 10 thining intervals per chain were set to gain the posterior distributions of model parameters. The first 20,000 iterations were used for burn-in so as to eliminate the effects of initial value scaling, while the last 30,000 were applied to sampling. A relationship between direct and indirect multiple-intervention comparisons was drawn as a network figure using Stata version 13.0 software. The Brooks-Gelman-Rubin statistic was calculated to evaluate model convergence. The closer the potential scale reduction factor (PSRF) value was to 1, the better the convergence. Of course, a PSRF value of less than 1.2 was still acceptable. The node-splitting analysis was evaluated to test the consistency (Dias et al., 2010). If the p-value was greater than 0.05, a consistency model would be used. Otherwise, an inconsistency model was used. Accordingly, sensitivity analysis was used to test the source of heterogeneity. To summarize the probabilities for all interventions, the surface under the cumulative ranking curve (SUCRA) was used as a summary statistic for the cumulative ranking (Salanti et al., 2011). Based on the definition, the larger SUCRA scores are, the more effective the interventions are. In this study, the effect sizes of all outcomes were analyzed by a fixed or random effect model depending on indexes of statistical heterogeneity including the p-value and inconsistency index statistic (I2) (Higgins et al., 2003). Dichotomous outcomes were calculated using the odds ratio (OR) and 95% credible intervals (CrIs). Continuous variable data were evaluated using the mean difference (MD) and with their corresponding 95% CrIs.
Results
Study Identification and Selection
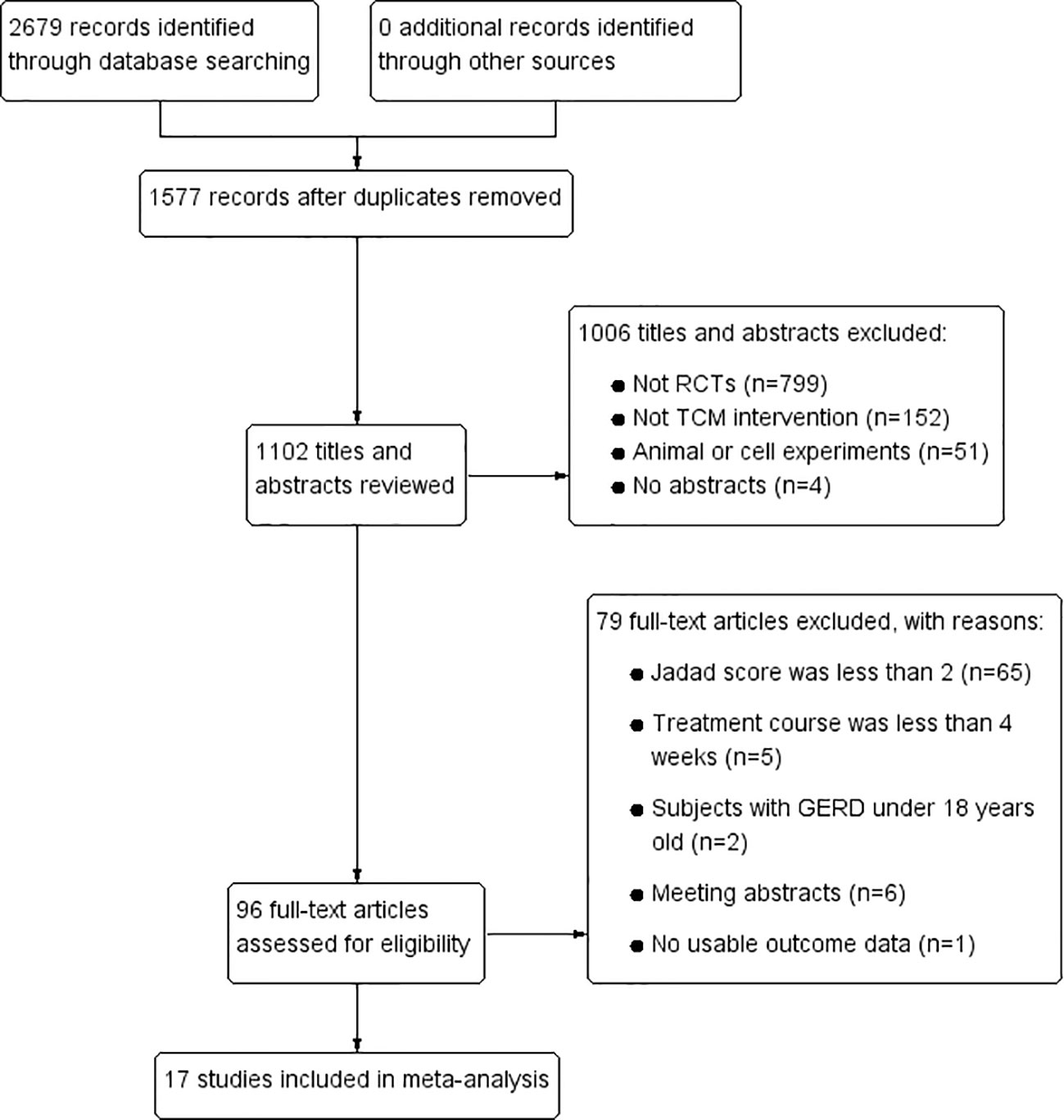
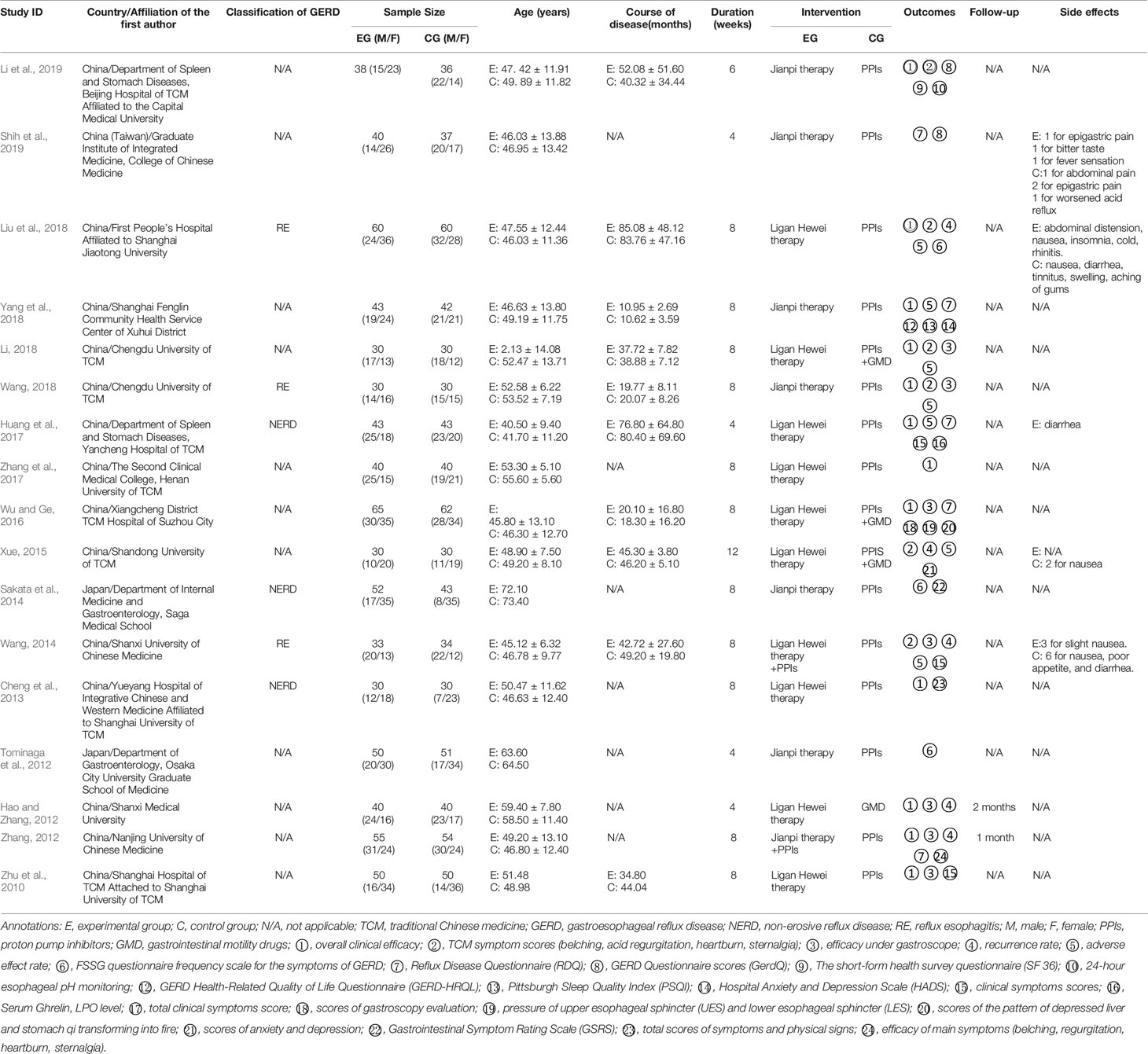
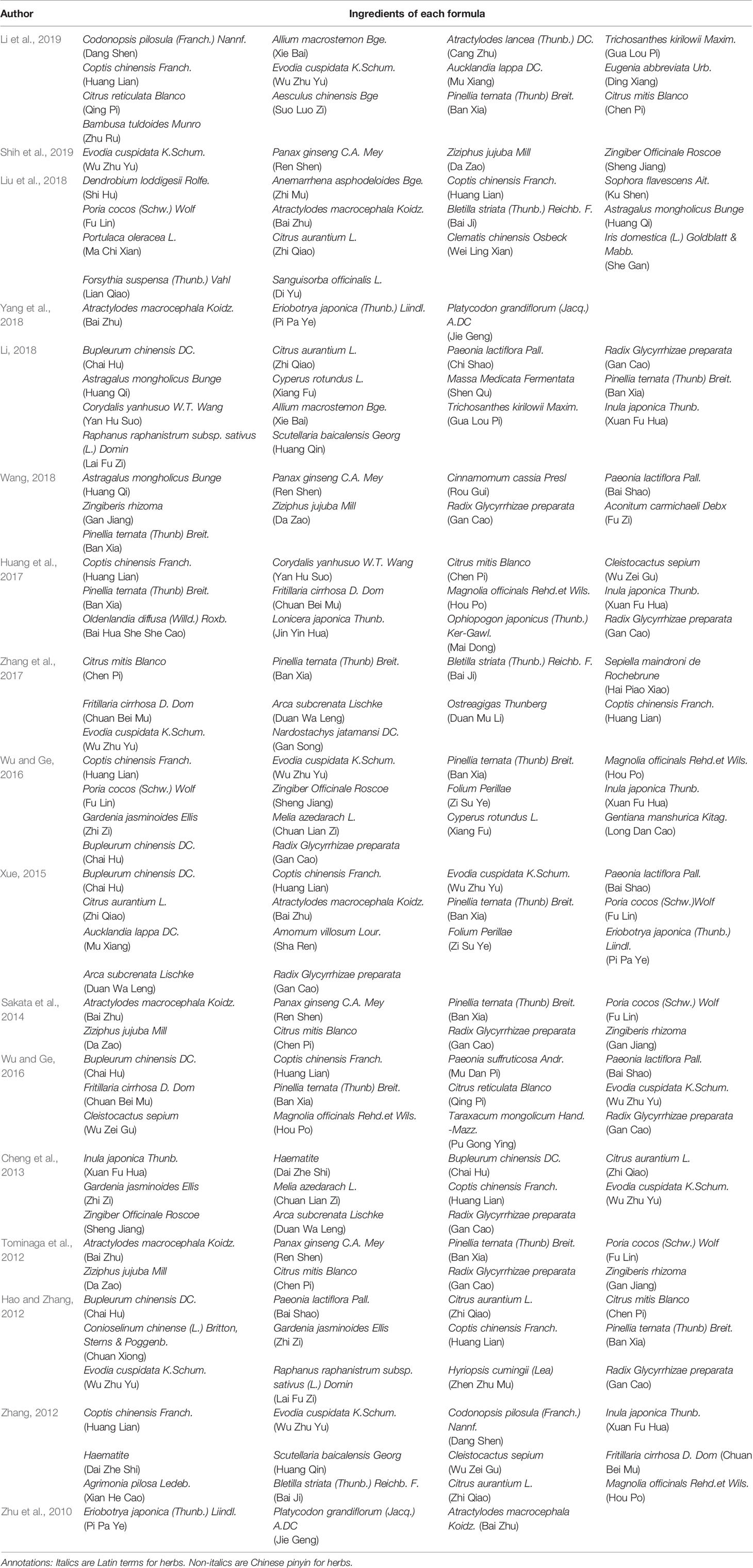
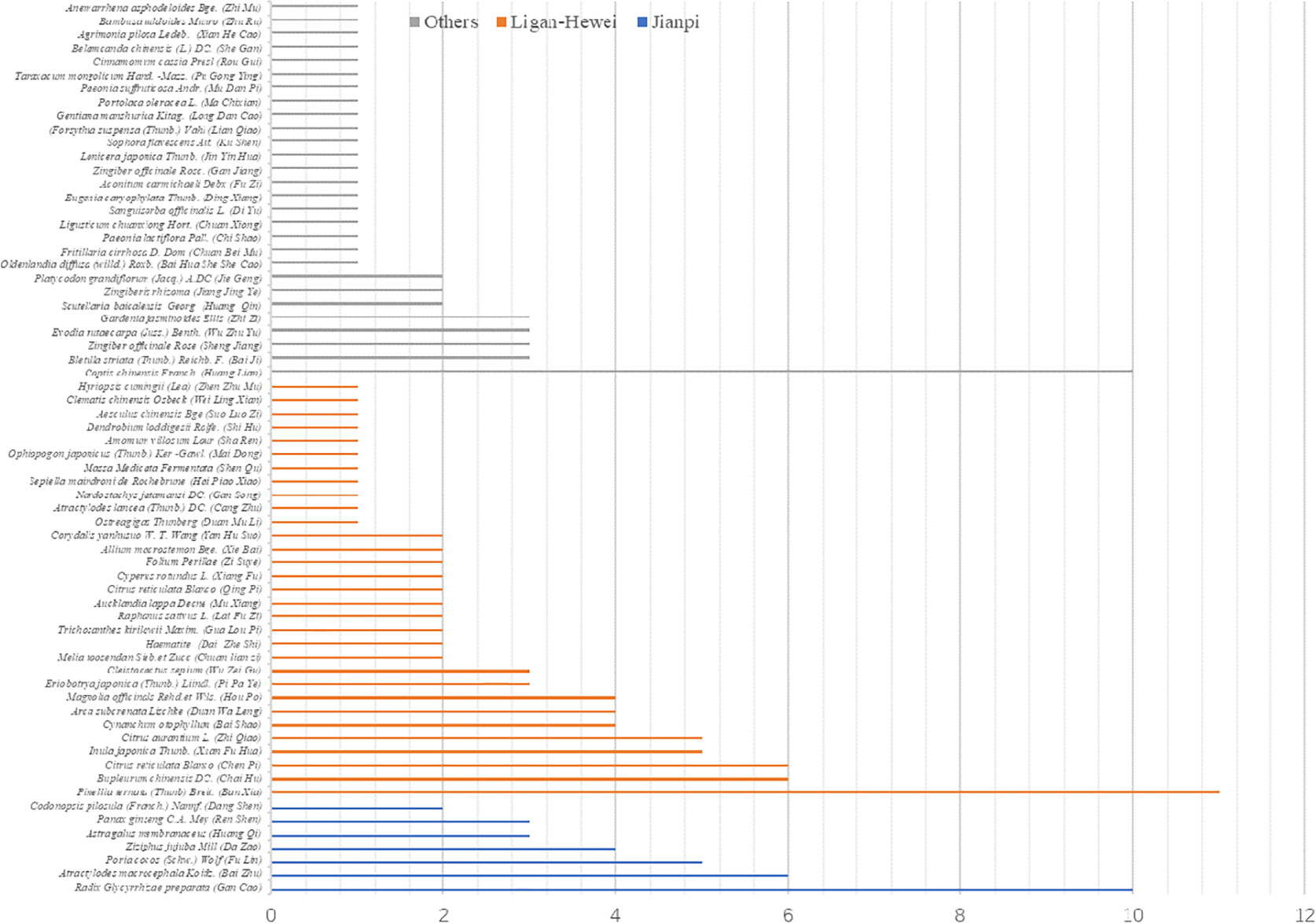
In total, 2679 articles were retrieved from the nine databases according to the corresponding search strategies. After removing duplicates and irrelevant publications, 17 randomized controlled trials (RCTs) including 1441 participants were selected for further quantitative analyses. A flow diagram of the specific retrieval process is shown in Figure 1. The baseline characteristics of the included trials are displayed in Table 1. The classification of herbal medicines and usage frequency of the included herbs can be found in Table 2 and Figure 2. Accordingly, we could draw a rough conclusion that herbs with the function of regulating the liver and harmonizing the stomach (TCM jargon: Ligan Hewei) and invigorating spleen (TCM jargon: Jianpi) had higher frequencies among the included herbs.
Risk of Bias Evaluation
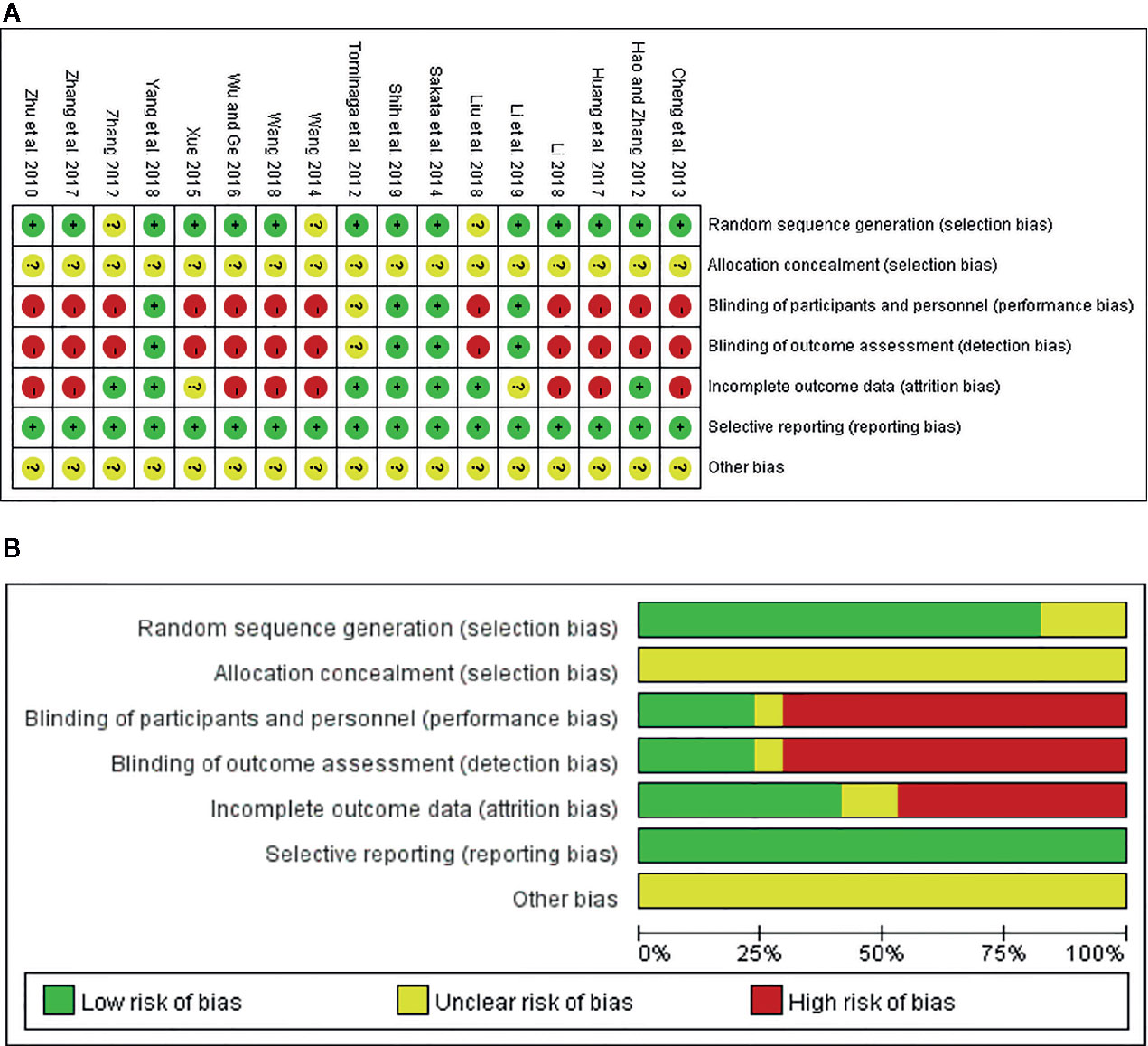
On the basis of the Cochrane Collaboration Recommendations evaluation tools (Savovic et al., 2014), the quality of the included RCTs was assessed. Of all the studies, 82.35% (14/17) gave a specific description of the random-allocation process, such as the use of a random number table or a computer-generated randomization list. The others only used the word “randomization” without any explanation. Because of insufficient information about allocation concealment, all included trials were judged as of “unclear risk.” In performance bias, only four studies (23.53%) described double or single blinding. As for detection bias, 13 trials (76.47%) either could not be blinded or it was unclear whether they had been. In addition, eight RCTs (47.06%) were at low risk of attrition bias because they provided detailed explanations or statistical estimations of dropout rates. However, two trials (11.76%) failed to provide adequate information for the judgment of missing data risk. Moreover, there was insufficient information on other risks for all 17 studies. In sum, among all trials, 4 were viewed as low risk, 2 as unclear risk, and 11 as high risk. A detailed quality evaluation is shown in Figures 3A, B.
Network Evidence
This study included seven regimens as follows: Ligan Hewei therapy, Ligan Hewei therapy+PPIs, Jianpi therapy, Jianpi therapy+PPIs, PPIs, PPIs+GMD, and GMD. The results of the network analysis suggested that the number of GERD patients treated with PPIs was the largest, followed by Ligan Hewei therapy and then Jianpi therapy, while the number of GERD patients treated with Ligan Hewei therapy+PPIs was the smallest (Figure 4).
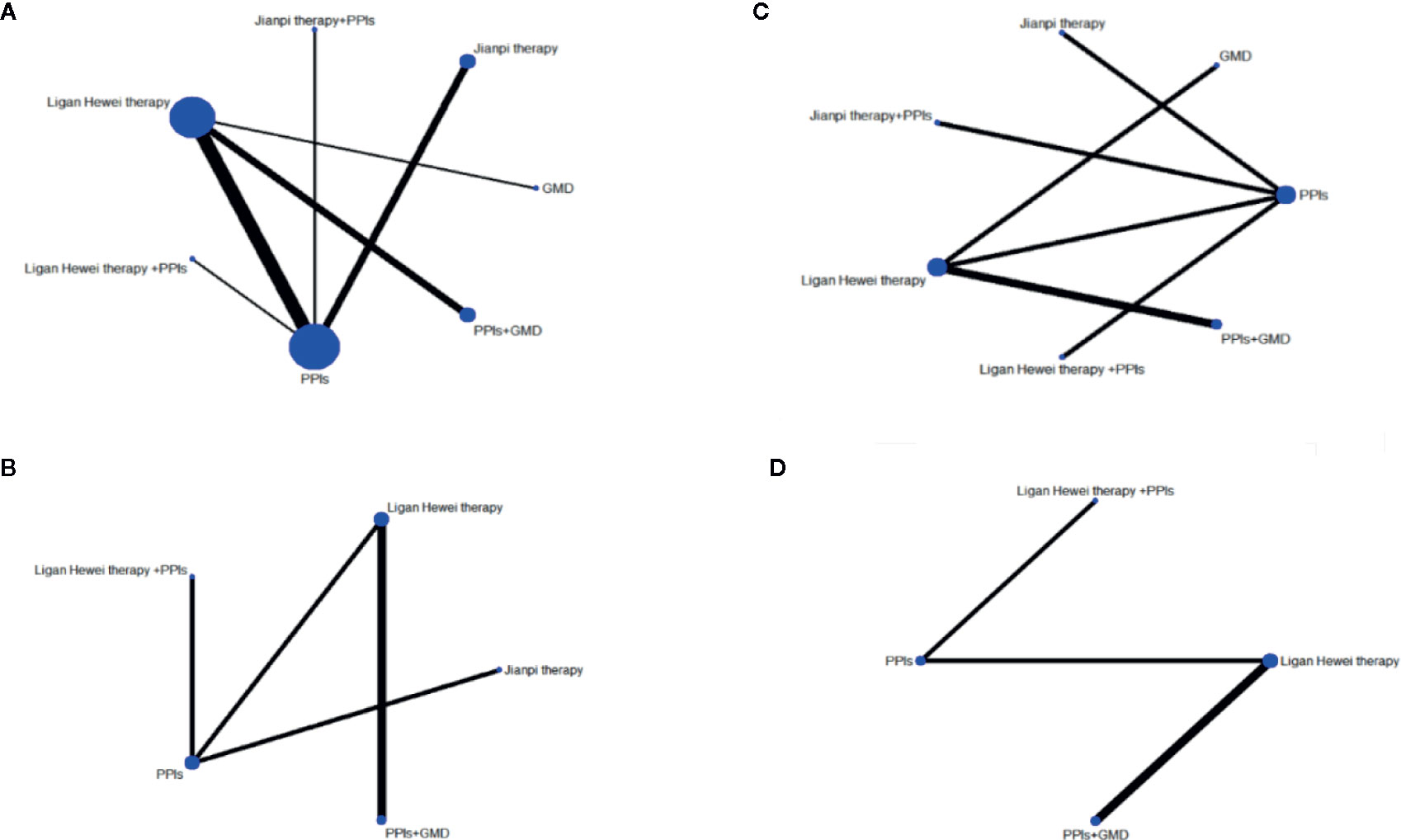
Figure 4 Network evidence diagrams of four endpoints. (A) Overall clinical efficacy. (B) Improvement of acid regurgitation. (C) Efficacy under gastroscope. (D) Improvement of heartburn.
Major Results of Network Analysis
In this study, there were 14 publications reporting overall clinical efficacy, 7 reporting the improvement of gastroesophageal mucosal lesions when viewed under a gastroscope (namely efficacy under gastroscope), 5 reporting improvement of acid regurgitation, and 4 reporting heartburn improvement. As shown in Table S2, the results of node-splitting between the direct and indirect effects showed no inconsistency for the four outcomes (P>0.05). Meanwhile, the PSRF value with 1 or 1.01 indicated good convergence and a stable result. Therefore, a model of consistency was built. As displayed in Table 3, Ligan Hewei therapy was significantly better overall clinical efficacy than PPIs (OR 2.36, 95%CrIs 1.16 to 5.24) and GMD+PPIs (OR 2.99, 95%CrIs 1.09 to 8.60). For the improvement of acid regurgitation, Ligan Hewei therapy+PPIs (MD -1.51, 95%CrIs -3.45 to 0.48), Ligan Hewei therapy (MD -1.07, 95%CrIs -3.10 to 0.93), and PPIs (MD -0.93, 95%CrIs -2.47 to 0.56) were superior to Jianpi therapy, Ligan Hewei therapy+PPIs (MD -0.58, 95%CrIs -1.84 to 0.74) was better than PPIs, and PPIs+GMD was superior to Jianpi therapy (MD -1.25, 95%CrIs -3.49 to 0.92) and Ligan Hewei therapy (MD -0.18, 95%CrIs -1.13 to 0.71). For heartburn improvement, Ligan Hewei therapy+PPIs (MD -0.76, 95%CrIs -1.73 to 0.24), PPIs+GMD (MD -0.34, 95%CrIs -1.56 to 0.85), and Ligan Hewei therapy (MD -0.19, 95%CrIs -1.18 to 0.78) were better than PPIs, and Ligan Hewei therapy+PPIs (MD -0.56, 95%CrIs -1.91 to 0.83) and PPIs+GMD (MD -0.15, 95%CrIs -0.84 to 0.52) were superior to Ligan Hewei therapy.
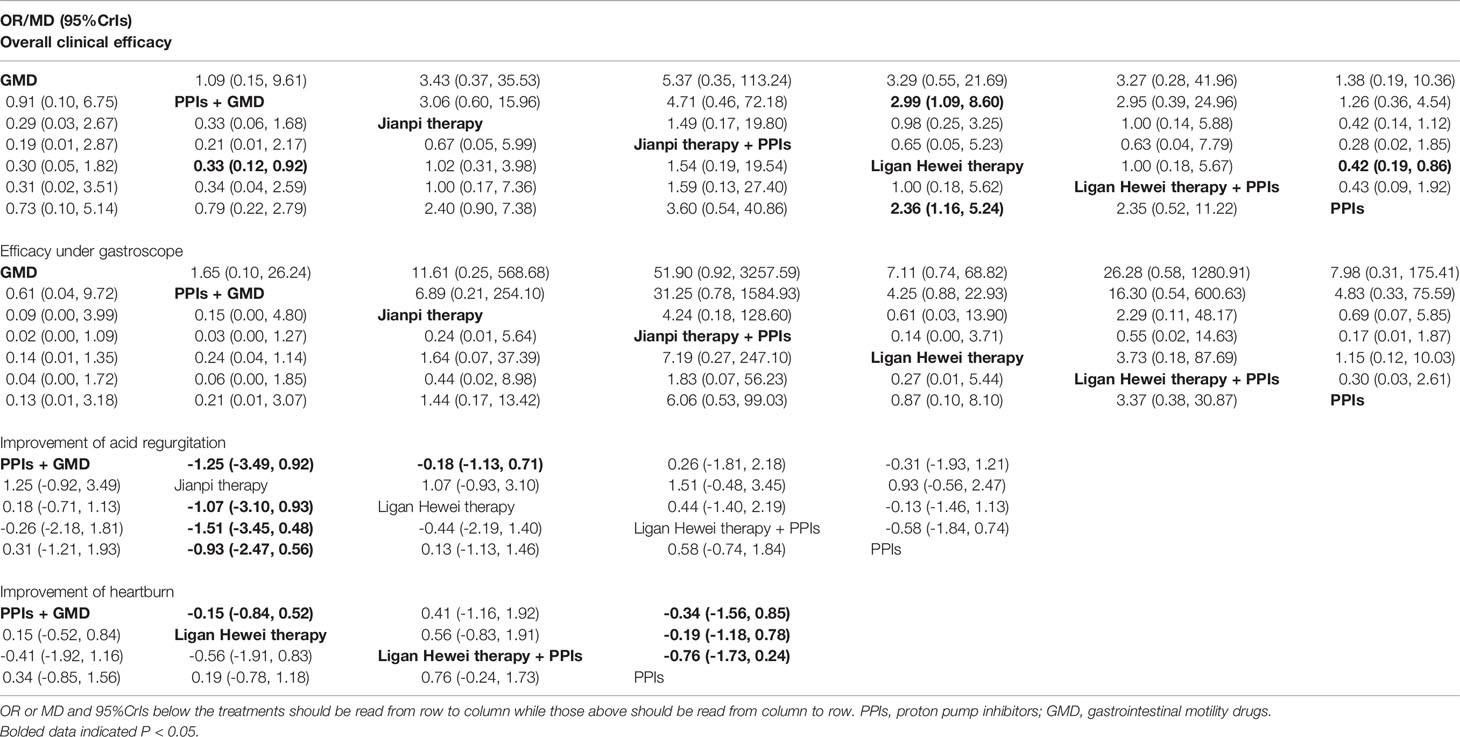
Table 3 Odds ratios (OR) or mean difference and 95% credible intervals (CrIs) of seven regimes with four endpoints.
SUCRA Value
The SUCRA-based rankings of all treatments are displayed in Figure 5. In terms of overall clinical efficacy, Jianpi therapy+PPIs (78.7%) ranked first, followed by Ligan Hewei therapy (71.1%), Ligan Hewei therapy+PPIs (69.2%), Jianpi therapy (67.8%), PPIs (25.4%), GMD (19.6%), and PPIs+GMD (18.3%). However, viewed as a whole, the SUCRA value of efficacy under gastroscope was higher for Ligan Hewei therapy (92.0%) than for any other treatments. As for the improvement of acid regurgitation and heartburn, the highest SUCRA value was found for Ligan Hewei therapy+PPIs (acid regurgitation: 79.4%; heartburn: 90.0%).
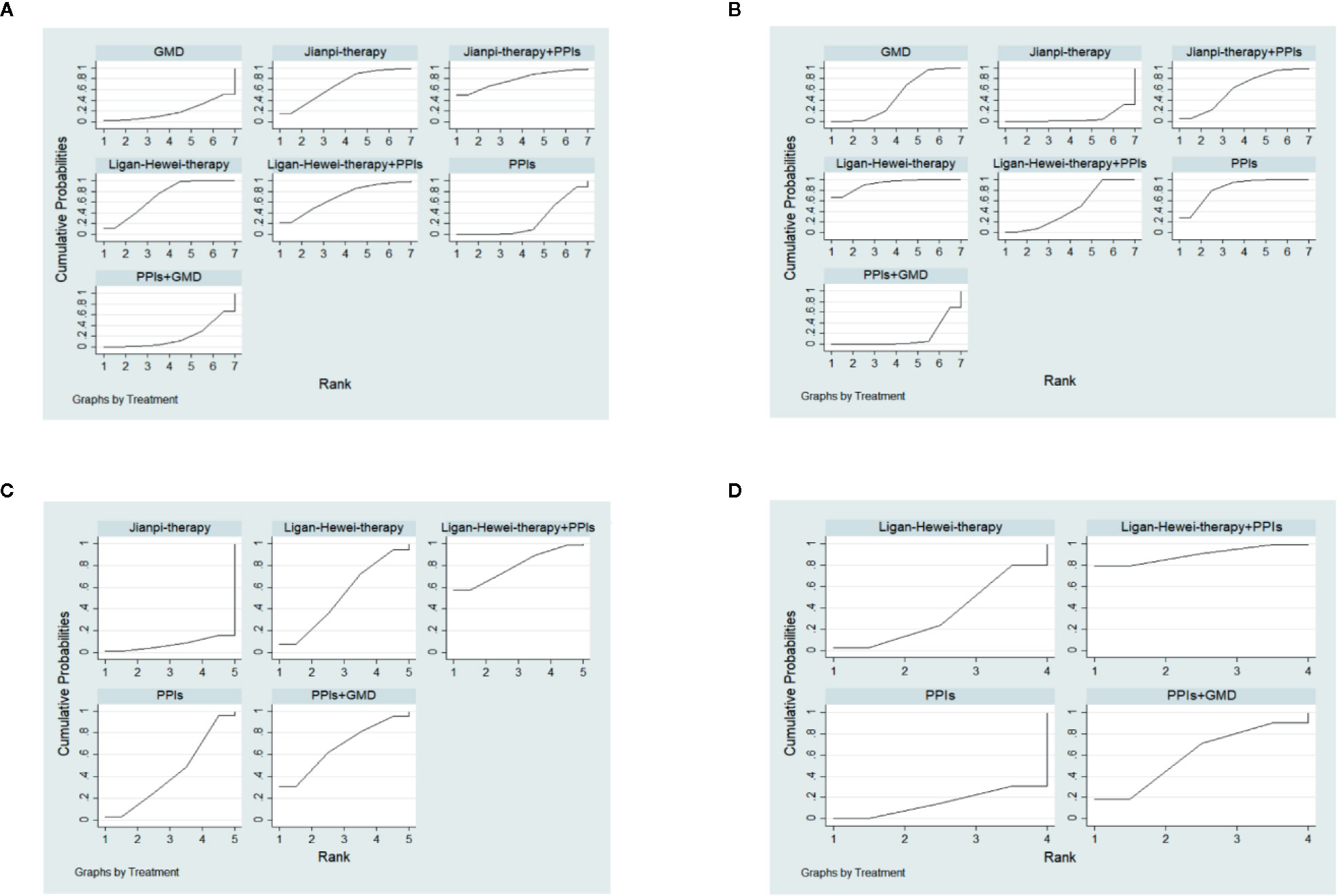
Figure 5 The surface under the cumulative ranking curve (SUCRA) plots of four endpoints. (A) Overall clinical efficacy. (B) Efficacy under gastroscope.(C) Improvement of acid regurgitation. (D) Improvement of heartburn.
Sensitivity Analysis
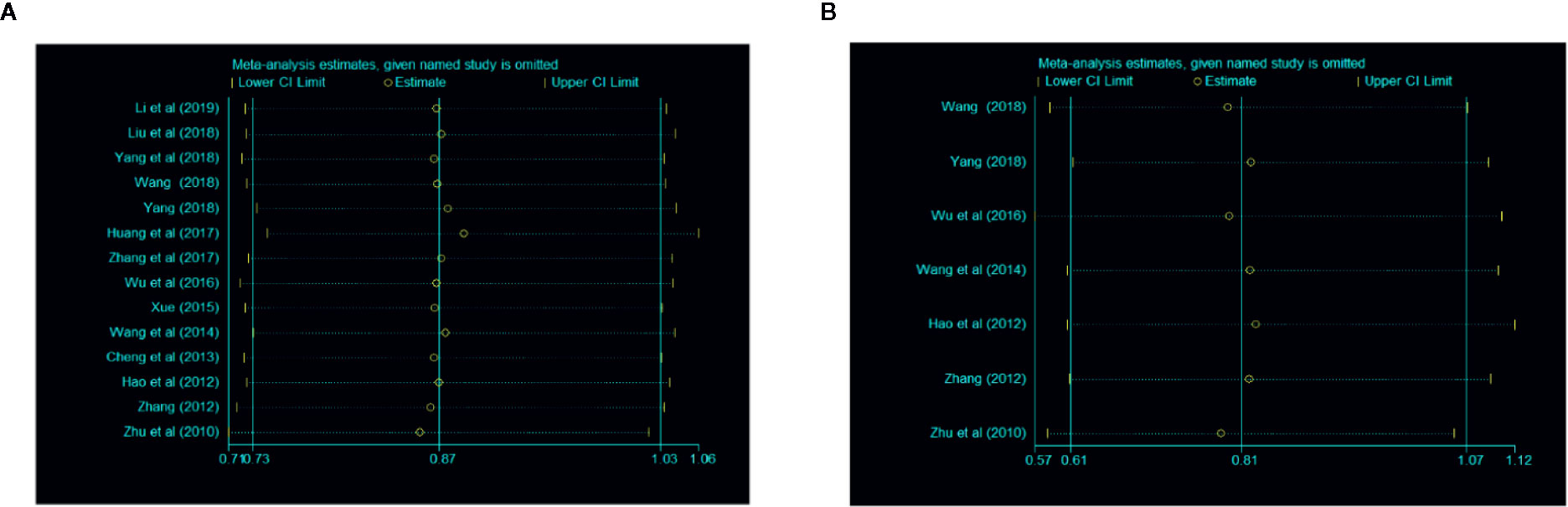
A sensitivity analysis was conducted through omitting studies one by one. The result of this analysis showed that there were no significant differences in overall clinical efficacy and efficacy under gastroscope (Figure 6).
GRADE Evidence of Quality
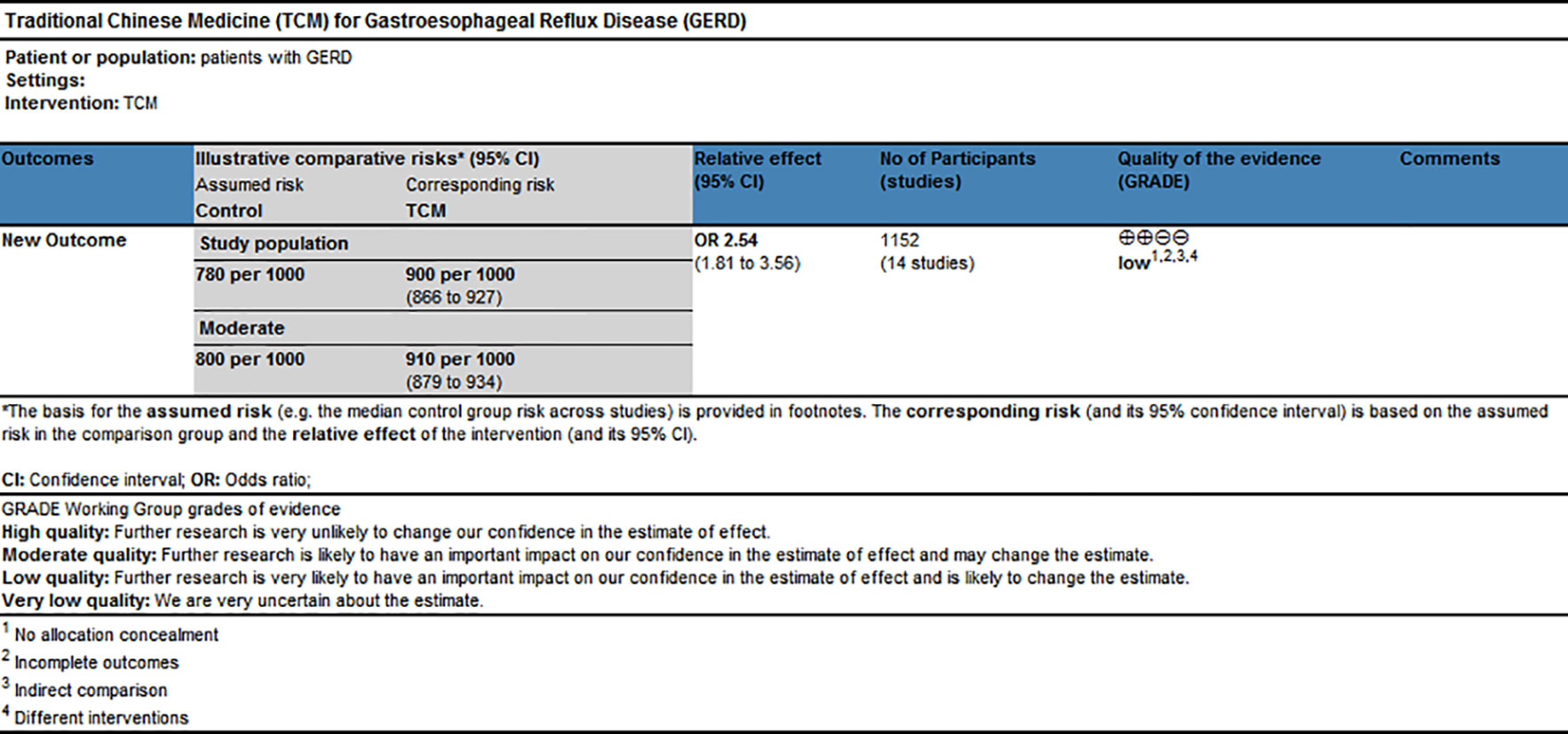
GRADE profiler software, which includes the elements of GRADE criteria such as study design, risk of bias, inconsistency, indirectness, imprecision, and publication bias, was used to rate the quality of evidence and grade strength of recommendations for this network meta-analysis. The results shown in Figure 7 suggested that the evidence quality of overall clinical efficacy was “Low,” which could be related to high risk of bias and indirectness within RCTs.
Discussion
Network meta-analysis is used to analyze studies with multiple interventions and provide rankings for them (Naci et al., 2014). Our findings from the comprehensive network analyses demonstrated the overall synthesis of data for currently available GERD treatments in terms of different THM. Regarding the usage frequency of each herb, a rough conclusion was drawn that herbs with the function of Ligan Hewei and Jianpi were used more frequently among the included herbs. In terms of outcomes, we found that Jianpi therapy+PPIs ranked first in overall clinical efficacy. Ligan Hewei therapy might be a better choice for healing gastroesophageal mucosal lesions according to gastroscope observations. In addition, in the improvement of acid regurgitation and heartburn, Ligan Hewei therapy+PPIs was superior to other interventions. Therefore, Ligan Hewei therapy and Jianpi therapy could be promising complementary and alternative therapies in the management of GERD, which potentially provides TCM practitioners with more suggestions and guidance in clinical decisions, as well as for treatments based on syndrome differentiation.
The pathogenesis of GERD is poorly understood so far. Currently, some acknowledged potential mechanisms are not only involved in hiatal hernia (Dore et al., 2016), anti-reflux barrier dysfunction (Xie et al., 2017), esophageal inflammation (Dunbar et al., 2016), and transient lower esophageal sphincter relaxation (TLESR) (Banovcin et al., 2016) but have also been associated with psychological factors (Baker et al., 1995; Wright et al., 2005) and obesity (Nadaleto et al., 2016). However, in the modern pharmacological field, complementary and alternative medicine (CAM), especially TCM, could potentially intervene in these mechanisms. A clinical study showed that wu chu yu tang (affiliated to Jianpi therapy) could improve the symptoms of GERD through anti-inflammation, antioxidant activity, acid suppression, reduction in pepsin secretion, and mucosal protection (Shih et al., 2019). In the treatment of gastrointestinal (GI) reflux diseases, another study indicated that Wendan decoction (WDD, affiliated with Ligan Hewei therapy) could reduce unhealthy emotions in patients via normalizing behaviors and up-regulating orexin-A, orexin receptor 1, and leptin and its receptor in the brain (Ling et al., 2015). Additionally, WDD could solve phlegm-related problems and recover GI homeostasis through dual action on acid and bile secretion (Xu et al., 2015). Meanwhile, acupuncture regulating qi based on the compatibility of the five meridians (affiliated to Ligan Hewei therapy) could also play an important role in treating GERD with disharmony between liver and stomach syndrome, whose mechanisms were possibly related to its regulation in the neuro-endocrine-immune system, thereby alleviating TLESR, promoting GI motility, suppressing acid secretion, and protecting gastric mucosa (Pan et al., 2017). Besides, acupoint drug finger pressing, based on the TCM theory of Jianpi therapy, also showed good therapeutic effects on GERD, which is probably attributable to lower esophageal sphincter pressure promotion and decrease in acid reflux in esophagus, as well as the improvement of coordination of gastroesophageal movement (Xie et al., 2007). In sum, CAM, especially TCM, may be multi-target treatments of GERD that are worth studying further in vitro and vivo.
Generally speaking, non-randomized trials are susceptible to many biases that affect the weaker forms of evidence. However, in RCTs, certain deficits in their design, conduct, analysis, and reporting may result in bias (Savovic et al., 2018). In this study, the methodological quality of the included trials was generally moderate, and the quality level of evidence for overall clinical efficacy, according to GRADE evidence classification, was “Low.” Analyzing from the above two results, the potential risk of bias in our study was possibly rooted in three aspects. First, there were 13/17 (76.47%) RCTs in which blinding as not implemented, which may lead to the occurrence of performance and detection biases. Next, due to the absence of allocation concealment in all of the included studies, the subjects could easily recognize which treatment they were allocated to, inevitably resulting in selection bias. Last, although 8/17 (47.06%) studies reported detailed withdrawals or dropouts, another 2 (11.76%) failed to provide an adequate explanation for missing data, which may also increase the risk of attrition bias.
In network analysis, consistency is characterized as a single comparison of the relationship between direct and indirect sources of evidence (Madan et al., 2011). When consistency is not good in a statistical analysis, it could be short of transitivity. In our study, for primary and secondary outcomes, based on the “node-splitting” method, it showed good convergence and strong stability, thereby further proving high reliability in our results. Nevertheless, clinical heterogeneity, for example, regarding the improvement of symptoms (acid regurgitation and heartburn), which were assessed by the standard excessively subjective judgments by doctors or patients, cannot be ruled out. Also, it should be taken into consideration that overall clinical efficacy and efficacy under gastroscope were described as comprehensive evaluation of the improvement of both many types of GERD symptoms and histopathological changes of gastroesophageal mucosa.
There are several potential limitations to our study. First, the included studies were only in Chinese and Japanese. Evidence with this geographically limited distribution needs more multicenter and large-scale research around the world to support it. Second, discrepancies in traditional herbal medicines (specifically, the interventions mentioned in our study) may exist because of their source and preparation, which could influence the strength of the evidence. Third, missing data could pose a threat to the validity of RCTs because it means that the observed outcomes of an RCT are not representative of all RCTs in the trial. Meanwhile, there was no corresponding evidence to verify its impact on the overall results in our study. Fourth, there was no unified criterion for the classification of interventions. Accordingly, we categorized them by the functions of herbs or prescriptions in the literature. Last, high quality of RCTs plays a key role in the production of optimal sources of evidence.
Therefore, we are looking forward to further standardized research and superior methodology, such as multicenter, large sample sizes, and well-designed (including the implementation of allocation concealment and blinding) RCTs to update and perfect the current body of evidence. Furthermore, strictly following the Consolidated Standards of Reporting Trials (CONSORT) or Standards for Reporting Interventions in Controlled Trials (STRICTA) statement is also essential to improve the reporting quality of future research.
Conclusion
Evidence from this network analysis indicates that Ligan Hewei therapy and Jianpi therapy could be the most suitable complementary and alternative interventions for GERD. According to different evaluation outcomes, Jianpi therapy+PPIs could be an optimum treatment in terms of overall clinical efficacy. Ligan Hewei therapy might be suitable for improving gastroesophageal mucosal lesions as seen under a gastroscope. Ligan Hewei therapy+PPIs could be a better choice for patients with acid regurgitation and heartburn. These findings could provide physicians and patients with appropriate treatments based on the specific characteristics of GERD. However, additional high-quality RCTs should be conducted to offer more powerful evidence for future research.
Data Availability Statement
All datasets presented in this study are included in the article/Supplementary Material.
Author Contributions
Conceived and designed the study: LH and LL. Performed the experiment: Y-KD, Y-BW, and HW. Analyzed the data: Y-KD and W-BW. Wrote the paper: Y-KD. Study supervision: RL-L, W-JC, CT, LL, and LH. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81774238, 81373563 and 30772689), Guangdong Science and Technology Project South China Cooperative Innovation Center for Chinese Medicine (No. 2014B090902002), Construction of Chinese First-Class Discipline Research of Key Project of Guangzhou University of Chinese Medicine ([2020] No. 62, [2019] No. 5 and [2018] No. 6), Construction of Chinese First-Class Discipline of Guangzhou University of Chinese Medicine (2017, No. 70), Construction of High-level University of Guangzhou University of Chinese Medicine (2016, No. 64), and Innovation Team to Foster Scientific Research Projects of Guangzhou University of Chinese Medicine (No. 2016KYTD07).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2020.00884/full#supplementary-material
References
Ades, A. E., Sculpher, M., Sutton, A., Abrams, K., Cooper, N., Welton, N., et al. (2006). Bayesian methods for evidence synthesis in cost-effectiveness analysis. Pharmacoeconomics 24 (1), 1–19. doi: 10.2165/00019053-200624010-00001
Baker, L. H., Lieberman, D., Oehlke, M. (1995). Psychological distress in patients with gastroesophageal reflux disease. Am. J. Gastroenterol. 90 (10), 1797–1803.
Banovcin, P. J., Halicka, J., Halickova, M., Duricek, M., Hyrdel, R., Tatar, M., et al. (2016). Studies on the regulation of transient lower esophageal sphincter relaxations (TLESRs) by acid in the esophagus and stomach. Dis. Esophagus 29 (5), 484–489. doi: 10.1111/dote.12357
Cheng, Y. M., Zhang, X. L., Liu, C. F., Sun, Y. S., Cao, H. J., Zhu, S. L. (2013). Clinical observation of “Shugan Hewei Decoction” for non-erosive gastroesophageal reflux disease. SH.J.TCM 47 (06), 48–49. doi: 10.16305/j.1007-1334.2013.06.037
Dai, Y., Zhang, Y., Li, D., Ye, J., Chen, W., Hu, L. (2017). Efficacy and Safety of Modified Banxia Xiexin Decoction (Pinellia Decoction for Draining the Heart) for Gastroesophageal Reflux Disease in Adults: A Systematic Review and Meta-Analysis. Evid. Based Complement. Alternat. Med. 2017:9591319. doi: 10.1155/2017/9591319
Dai, Y. K., Li, D. Y., Zhang, Y. Z., Huang, M. X., Zhou, Y. L., Ye, J. T., et al. (2018). Efficacy and safety of Modified Tongxie Yaofang in diarrhea-predominant irritable bowel syndrome management: A meta-analysis of randomized, positive medicine-controlled trials. PloS One 13 (2), e192319. doi: 10.1371/journal.pone.0192319
DeVault, K. R., Castell, D. O. (2005). Updated guidelines for the diagnosis and treatment of gastroesophageal reflux disease. Am. J. Gastroenterol. 100 (1), 190–200. doi: 10.1111/j.1572-0241.2005.41217.x
Dias, S., Welton, N. J., Caldwell, D. M., Ades, A. E. (2010). Checking consistency in mixed treatment comparison meta-analysis. Stat. Med. 29 (7-8), 932–944. doi: 10.1002/sim.3767
Dore, M. P., Pes, G. M., Bassotti, G., Farina, M. A., Marras, G., Graham, D. Y. (2016). Risk factors for erosive and non-erosive gastroesophageal reflux disease and Barrett’s esophagus in Nothern Sardinia. Scand. J. Gastroenterol. 51 (11), 1281–1287. doi: 10.1080/00365521.2016.1200137
Dunbar, K. B., Agoston, A. T., Odze, R. D., Huo, X., Pham, T. H., Cipher, D. J., et al. (2016). Association of Acute Gastroesophageal Reflux Disease With Esophageal Histologic Changes. JAMA 315 (19), 2104–2112. doi: 10.1001/jama.2016.5657
El-Serag, H. B., Sweet, S., Winchester, C. C., Dent, J. (2014). Update on the epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut 63 (6), 871–880. doi: 10.1136/gutjnl-2012-304269
Fass, R., Shapiro, M., Dekel, R., Sewell, J. (2005). Systematic review: proton-pump inhibitor failure in gastro-oesophageal reflux disease–where next? Aliment. Pharmacol. Ther. 22 (2), 79–94. doi: 10.1111/j.1365-2036.2005.02531.x
Freston, J. W., Malagelada, J. R., Petersen, H., McCloy, R. F. (1995). Critical issues in the management of gastroesophageal reflux disease. Eur. J. Gastroenterol. Hepatol. 7 (6), 577–586.
Gao, S., Zhang, Q., Tian, C., Li, C., Lin, Y., Gao, W., et al. (2020). The roles of Qishen granules recipes, Qingre Jiedu, Wenyang Yiqi and Huo Xue, in the treatment of heart failure. J. Ethnopharmacol. 249, 112372. doi: 10.1016/j.jep.2019.112372
Hao, W. J., Zhang, J. Y. (2012). Clinical observation on shugan-yiwei granules for treatment of gastroesophageal reflux disease. Proc. Clin. Med. 21 (04), 246–249.
Higgins, J. P., Thompson, S. G., Deeks, J. J., Altman, D. G. (2003). Measuring inconsistency in meta-analyses. BMJ 327 (7414), 557–560. doi: 10.1136/bmj.327.7414.557
Huang, H., Huang, F. B., Zhang, Y., Liu, Z. C., Qiu, G. (2017). Clinical study of Guan Yanling Granules in the treatment of non-erosive reflux disease with syndrome of stomach heat and qi stagnation. China Med. Herbal 14 (30), 139–142.
Jiang, C., Wang, T., Ma, Z., Fang, B. J. (2019). Effectiveness of Fuyuan Xingnao Decoction for patients with diabetes mellitus combined cerebral infarction. Med. (Baltimore) 98 (39), e17273. doi: 10.1097/MD.0000000000017273
Katz, P. O., Scheiman, J. M., Barkun, A. N. (2006). Review article: acid-related disease–what are the unmet clinical needs? Aliment. Pharmacol. Ther. 23 Suppl 2, 9–22. doi: 10.1111/j.1365-2036.2006.02944.x
Kong, Z., Liang, N., Yang, G. L., Zhang, Z., Liu, Y., Li, J., et al. (2019). Xiao Chai Hu Tang, a herbal medicine, for chronic hepatitis B. Cochrane Database Syst. Rev. 2019 (11), CD013090. doi: 10.1002/14651858.CD013090.pub2
Li, X., Chen, S., Peng, M. Z., Lou, Y., Wang, X. R., Wang, W., et al. (2019). Treatment of invigorating spleen and harmonizing stomach with warming Yang and dredging diaphragm. Beijing J. Tradit. Chin. Med. 38 (06), 519–524. doi: 10.16025/j.1674-1307.2019.06.002
Li, Y. (2018). Clinical study of Chaiqi decoction combined with western medicine in the treatment of gastroesophageal reflux disease of liver and stomach disharmony (Chengdu, Sichuan, China: Chengdu University of TCM), 4–15.
Liberati, A., Altman, D. G., Tetzlaff, J., Mulrow, C., Gotzsche, P. C., Ioannidis, J. P., et al. (2009). The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700. doi: 10.1136/bmj.b2700
Ling, W., Huang, Y., Xu, J. H., Li, Y., Huang, Y. M., Ling, H. B., et al. (2015). Consistent Efficacy of Wendan Decoction for the Treatment of Digestive Reflux Disorders. Am. J. Chin. Med. 43 (5), 893–913. doi: 10.1142/S0192415X15500524
Liu, Q. L., Han, H. J., Zhou, P., Lu, Y. Y., Xu, G. Y., Liang, S. P. (2018). Clinical trial of refractory reflux esophagitis treated with qingwei granules. World J. Integr. Tradit. Western Med. 13 (09), 1267–1270.
Liu, Y., Wang, S., Kan, J., Zhang, J., Zhou, L., Huang, Y., et al. (2020). Chinese Herbal Medicine Interventions in Neurological Disorder Therapeutics by Regulating Glutamate Signaling. Curr. Neuropharmacol. 18 (4), 260–276. doi: 10.2174/1570159X17666191101125530
Madan, J., Stevenson, M. D., Cooper, K. L., Ades, A. E., Whyte, S., Akehurst, R. (2011). Consistency between direct and indirect trial evidence: is direct evidence always more reliable? Value Health 14 (6), 953–960. doi: 10.1016/j.jval.2011.05.042
Naci, H., van Valkenhoef, G., Higgins, J. P., Fleurence, R., Ades, A. E. (2014). Evidence-based prescribing: combining network meta-analysis with multicriteria decision analysis to choose among multiple drugs. Circ. Cardiovasc. Qual. Outc. 7 (5), 787–792. doi: 10.1161/CIRCOUTCOMES.114.000825
Nadaleto, B. F., Herbella, F. A., Patti, M. G. (2016). Gastroesophageal reflux disease in the obese: Pathophysiology and treatment. Surgery 159 (2), 475–486. doi: 10.1016/j.surg.2015.04.034
Nocon, M., Labenz, J., Jaspersen, D., Leodolter, A., Richter, K., Vieth, M., et al. (2009). Health-related quality of life in patients with gastro-oesophageal reflux disease under routine care: 5-year follow-up results of the ProGERD study. Aliment. Pharmacol. Ther. 29 (6), 662–668. doi: 10.1111/j.1365-2036.2008.03921.x
Pan, S., Li, J., Zhang, X., Li, Y., Yan, J., Zhang, W., et al. (2017). Clinical trial of gastroesophageal reflux disease with the disharmony between liver and stomach syndrome treated with acupuncture regulating qi based on the compatibility of the five meridians. Zhongguo Zhen Jiu 37 (12), 1255–1260. doi: 10.13703/j.0255-2930.2017.12.001
Patrick, L. (2011). Gastroesophageal reflux disease (GERD): a review of conventional and alternative treatments. Altern. Med. Rev. 16 (2), 116–133.
Qin, H., Wen, H. T., Gu, K. J., Hu, X. D., Yang, T., Yan, X. F., et al. (2019). Total extract of Xin Jia Xuan Bai Cheng Qi decoction inhibits pulmonary fibrosis via the TGF-beta/Smad signaling pathways in vivo and in vitro. Drug Des. Dev. Ther. 13, 2873–2886. doi: 10.2147/DDDT.S185418
Sakata, Y., Tominaga, K., Kato, M., Takeda, H., Shimoyama, Y., Takeuchi, T., et al. (2014). Clinical characteristics of elderly patients with proton pump inhibitor-refractory non-erosive reflux disease from the G-PRIDE study who responded to rikkunshito. BMC Gastroenterol. 14, 116. doi: 10.1186/1471-230X-14-116
Salanti, G., Ades, A. E., Ioannidis, J. P. (2011). Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J. Clin. Epidemiol. 64 (2), 163–171. doi: 10.1016/j.jclinepi.2010.03.016
Savarino, V., Di Mario, F., Scarpignato, C. (2009). Proton pump inhibitors in GORD An overview of their pharmacology, efficacy and safety. Pharmacol. Res. 59 (3), 135–153. doi: 10.1016/j.phrs.2008.09.016
Savovic, J., Weeks, L., Sterne, J. A., Turner, L., Altman, D. G., Moher, D., et al. (2014). Evaluation of the Cochrane Collaboration’s tool for assessing the risk of bias in randomized trials: focus groups, online survey, proposed recommendations and their implementation. Syst. Rev. 3:37. doi: 10.1186/2046-4053-3-37
Savovic, J., Turner, R. M., Mawdsley, D., Jones, H. E., Beynon, R., Higgins, J., et al. (2018). Association Between Risk-of-Bias Assessments and Results of Randomized Trials in Cochrane Reviews: The ROBES Meta-Epidemiologic Study. Am. J. Epidemiol. 187 (5), 1113–1122. doi: 10.1093/aje/kwx344
Shih, Y. S., Tsai, C. H., Li, T. C., Yu, C. J., Chou, J. W., Feng, C. L., et al. (2019). Effect of wu chu yu tang on gastroesophageal reflux disease: Randomized, double-blind, placebo-controlled trial. Phytomedicine 56, 118–125. doi: 10.1016/j.phymed.2018.09.185
Sutton, A., Ades, A. E., Cooper, N., Abrams, K. (2008). Use of indirect and mixed treatment comparisons for technology assessment. Pharmacoeconomics 26 (9), 753–767. doi: 10.2165/00019053-200826090-00006
Tominaga, K., Iwakiri, R., Fujimoto, K., Fujiwara, Y., Tanaka, M., Shimoyama, Y., et al. (2012). Rikkunshito improves symptoms in PPI-refractory GERD patients: a prospective, randomized, multicenter trial in Japan. J. Gastroenterol. 47 (3), 284–292. doi: 10.1007/s00535-011-0488-5
Vakil, N., van Zanten, S. V., Kahrilas, P., Dent, J., Jones, R. (2006). The Montreal definition and classification of gastroesophageal reflux disease: a global evidence-based consensus. Am. J. Gastroenterol. 101 (8), 1900–1920, 1943. doi: 10.1111/j.1572-0241.2006.00630.x
Wahlqvist, P., Karlsson, M., Johnson, D., Carlsson, J., Bolge, S. C., Wallander, M. A. (2008). Relationship between symptom load of gastro-oesophageal reflux disease and health-related quality of life, work productivity, resource utilization and concomitant diseases: survey of a US cohort. Aliment. Pharmacol. Ther. 27 (10), 960–970. doi: 10.1111/j.1365-2036.2008.03671.x
Wang, C. (2014). Flavored the liver decoction combined western medicine in treatment of reflux esophagitis liver stomach heat syndromes of clinical research (Taiyuan, Shanxi, China: Shanxi University of Chinese Medicine), 4–8.
Wang, Y. (2018). Clinical Study on the Treatment of Reflux Esophagitis (middle-jiao deficiency and Qi reverse) by the Principle of Pungent-Sugariness Restricting Acid (Chengdu, Sichuan, China: Chengdu University).
Wright, C. E., Ebrecht, M., Mitchell, R., Anggiansah, A., Weinman, J. (2005). The effect of psychological stress on symptom severity and perception in patients with gastro-oesophageal reflux. J. Psychosom. Res. 59 (6), 415–424. doi: 10.1016/j.jpsychores.2005.05.012
Wu, Y. H., Ge, H. N. (2016). Clinical Study on Zuojin Pill Combined with Banxia Houpu Decoction in Treatment of gastroesophageal Reflux Disease. Acta Chin. Med. 31 (11), 1782–1785. doi: 10.16368/j.issn.1674-8999.2016.11.497
Xie, S., Liang, J., Yan, C. Y. (2007). [Therapeutic effects of acupoint drug-finger pressing on gastroesophageal reflux]. Zhongguo Zhong Xi Yi Jie He Za Zhi 27 (4), 355–358.
Xie, C., Wang, J., Li, Y., Tan, N., Cui, Y., Chen, M., et al. (2017). Esophagogastric Junction Contractility Integral Reflect the Anti-reflux Barrier Dysfunction in Patients with Gastroesophageal Reflux Disease. J. Neurogastroenterol. Motil. 23 (1), 27–33. doi: 10.5056/jnm16008
Xu, J. H., Huang, Y. M., Ling, W., Li, Y., Wang, M., Chen, X. Y., et al. (2015). Wen Dan Decoction for hemorrhagic stroke and ischemic stroke. Complement. Ther. Med. 23 (2), 298–308. doi: 10.1016/j.ctim.2015.01.001
Xue, Y. (2015). The Theoretical and Clinical Research on Simultaneous Treatment of Liver Lung and Stomach Method in Gastroesophageal Reflux Disease with Syndrome of Incoordination between Liver and Stomach (Jinan, Shandong, China: Shandong University of TCM), 11–20.
Yang, Y. F., Xiao, J., Qin, Y., Zhu, L. Y. (2018). Clinical study of Hezhong Jianpi Decoction in the treatment of gastroesophageal reflux disease: A random, double-blind and double-dummy trial. SH.J.TCM 52 (03), 39–42. doi: 10.16305/j.1007-1334.2018.03.010
Zhang, X. Y., Li, Z., Dang, Z. Q. (2017). Lizhong-Hewei Decoction in the Treatment of Gastroesophageal Reflux Disease for 40 Cases. Chin. Med. Modern Distance Educ. China 15 (03), 92–93. doi: 10.3969/j.issn.1672-2779.2017.03.040
Zhang, Z., Yi, P., Yang, J., Huang, J., Xu, P., Hu, M., et al. (2020). Integrated network pharmacology analysis and serum metabolomics to reveal the cognitive improvement effect of Bushen Tiansui formula on Alzheimer’s disease. J. Ethnopharmacol. 249, 112371. doi: 10.1016/j.jep.2019.112371
Zhang, J. (2012). Clinical study on jiangni-humo recipe in treating (Nanjing, Jiangsu, China: Nanjing University of TCM).
Keywords: traditional herbal medicines, gastroesophageal reflux disease, randomized controlled trials, network analysis, adults
Citation: Dai Y-k, Wu Y-b, Wen H, Li R-l, Chen W-j, Tang C, Lu L and Hu L (2020) Different Traditional Herbal Medicines for the Treatment of Gastroesophageal Reflux Disease in Adults. Front. Pharmacol. 11:884. doi: 10.3389/fphar.2020.00884
Received: 05 April 2020; Accepted: 28 May 2020;
Published: 16 July 2020.
Edited by:
Adolfo Andrade-Cetto, National Autonomous University of Mexico, MexicoReviewed by:
Andy Wai Kan Yeung, The University of Hong Kong, Hong KongSubhash Chandra Mandal, Government of West Bengal, India
Copyright © 2020 Dai, Wu, Wen, Li, Chen, Tang, Lu and Hu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ling Hu, ZHJodWxpbmdAMTYzLmNvbQ==; Liming Lu, bHVsaW1pbmdsZW9uQGd6dWNtLmVkdS5jbg==
 Yun-kai Dai1,2
Yun-kai Dai1,2 Chunzhi Tang
Chunzhi Tang Liming Lu
Liming Lu Ling Hu
Ling Hu