Abstract
Ethnopharmacological relevance: The genus Alternanthera (Amaranthaceae) comprises 139 species including 14 species used traditionally for the treatment of various ailments such as hypertension, pain, inflammation, diabetes, cancer, microbial and mental disorders.
Aim of the review: To search research gaps through critical assessment of pharmacological activities not performed to validate traditional claims of various species of Alternanthera. This review will aid natural product researchers in identifying Alternanthera species with therapeutic potential for future investigation.
Materials and methods: Scattered raw data on ethnopharmacological, morphological, phytochemical, pharmacological, toxicological, and clinical studies of various species of the genus Alternanthera have been compiled utilizing search engines like SciFinder, Google Scholar, PubMed, Science Direct, and Open J-Gate for 100 years up to April 2021.
Results: Few species of Alternanthera genus have been exhaustively investigated phytochemically, and about 129 chemical constituents related to different classes such as flavonoids, steroids, saponins, alkaloids, triterpenoids, glycosides, and phenolic compounds have been isolated from 9 species. Anticancer, antioxidant, antibacterial, CNS depressive, antidiabetic, analgesic, anti-inflammatory, and immunomodulator effects have been explored in the twelve species of the genus. A toxicity study has been conducted on 3 species and a clinical study on 2 species.
Conclusions: The available literature on pharmacological studies of Alternanthera species reveals that few species have been selected based on ethnobotanical surveys for scientific validation of their traditional claims. But most of these studies have been conducted on uncharacterized and non-standardized crude extracts. A roadmap of research needs to be developed for the isolation of new bioactive compounds from Alternanthera species, which can emerge out as clinically potential medicines.
Introduction
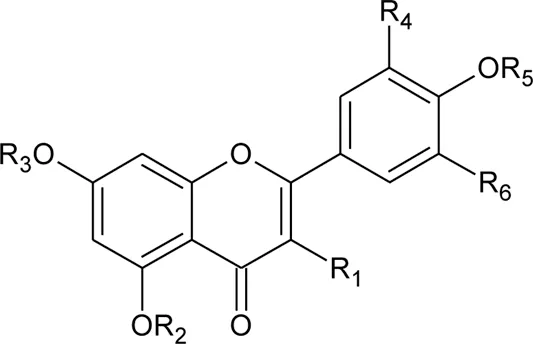
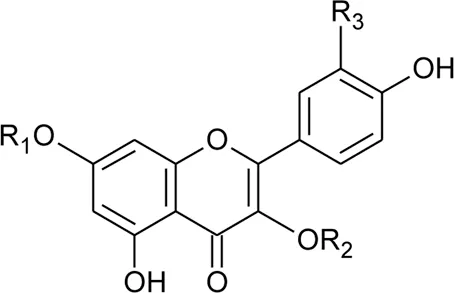
The family Amaranthaceae comprises 65 genera and about 850 species (Hundiwale et al., 2012; Chandrashekhar, 2019). These species are mainly distributed in tropical regions of the United States of America, Africa, and India. Amongst 65 genera and 850 species, only 17 genera and 50 species have been recorded to be found in India. The plants from this family include herbs, shrubs, and universal weeds. The genus Alternanthera, a significant delegate of the family Amaranthaceae was coined by by Forsskal in 1775. The genus Alternanthera comprises roughly 139 species which are distributed in India, China, Sri Lanka, the United States of America, and Africa (Figure 1). Though not complete and exhaustive, but phytochemical characterization was found to be reported that of Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera philoxeroides (Mart.) Griseb., Alternanthera brasiliana (L.) Kuntze, Alternanthera hirtula (Mart.) R.E.Fr., Alternanthera praelonga A.St.-Hil., Alternanthera littoralis P.Beauv., Alternanthera bettzickiana (Regel) G.Nicholson, and Alternanthera pungens Kunth (Table 1 with complete details).
FIGURE 1
TABLE 1
| S.No | Name | Structure | Source | Plant part | References |
|---|---|---|---|---|---|
| Benzopyran | |||||
| 1 | 3,3′-(Propane-2,2diyl)-bis-3,4,5,6,7,8-hexahydro-1H-isochromene | 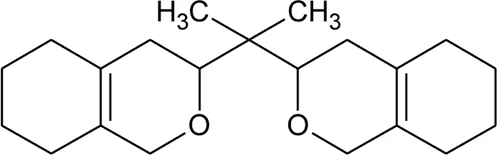 | Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Sundar et al. (2019) |
| Flavonoids | |||||
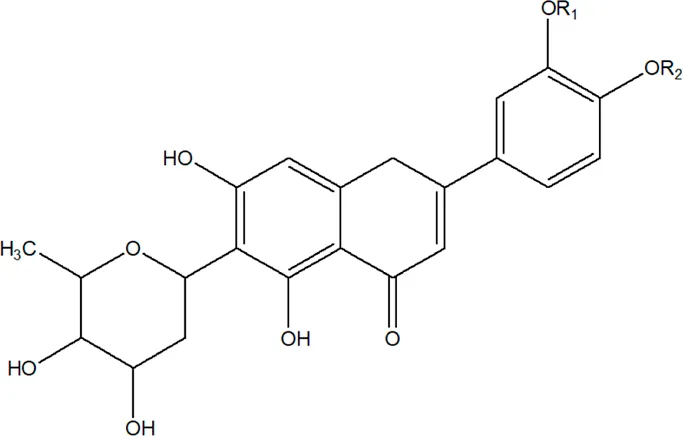 | |||||
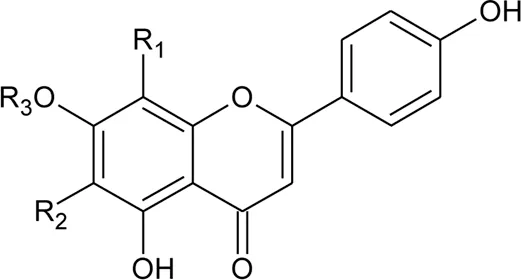
| 2 | Luteolin-6-C-β-D-boivinopyranosyl-3′-O-β-D-glucopyranoside | R1 = Glu; R2 = H | Alternanthera philoxeroides (Mart.) Griseb | Not specified | Li et al. (2016) |
| 3 | Chrysoeriol-6-C-β-D-boivinopyranosyl-4′-O-β-D-glucopyranoside | R1 = CH3; R2 = Glu | Alternanthera philoxeroides (Mart.) Griseb | Not specified | Li et al. (2016) |
| 4 | Luteolin-6-C-β-D-boivinopyranosyl-4′-O-β-D-glucopyranoside | R1 = H; R2 = Glu | Alternanthera philoxeroides (Mart.) Griseb | Not specified | Li et al. (2016) |
| 5 | Luteolin-6-C-β-D-boivinopyranoside or Alternanthin B or Demethyl-torosaflavone B | R1 = H; R2 = H | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Khamphukdee et al. (2018) |
| 6 | Chrysoeriol-6-C-β-D-boivinopyranoside or Alternanthin A | R1 = CH3; R2 = H | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Zhou et al. (1988) |
| Fan, (2008) | |||||
| Li et al. (2016) | |||||
| Khamphukdee et al. (2018) | |||||
| 7 | Chrysoeriol 6-C-β-boivinopyranosyl-7-O-β-glucopyranoside | 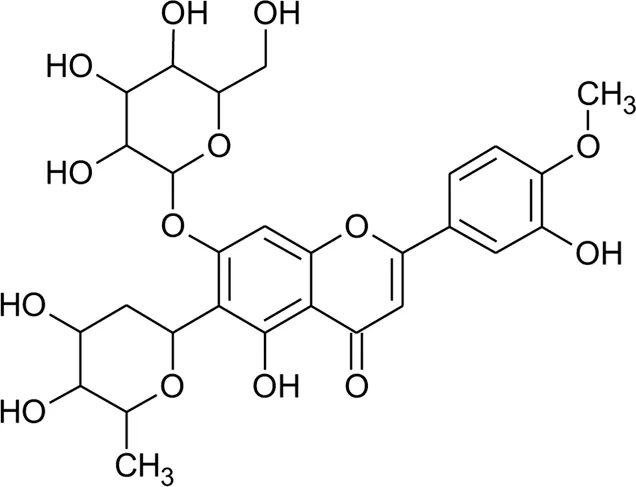 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
 | |||||
| 8 | 2′′-O-Ramnosylvitexin | R1 = Glucoslyl (1→6) ramnoside; R2 = R3 = R4 = H | Alternanthera brasiliana (L.) Kuntze | Aerial parts | Araujo et al. (2014) |
| 9 | 4′,5,7-trimethoxy-2′′-O-ramnosylvitexin | R1 = Glucoslyl (1→6) ramnoside; R2 = R3 = R4 = CH3 | Alternanthera brasiliana (L.) Kuntze | Aerial parts | Araujo et al. (2014) |
| 10 | Ligustroflavone | R1 = H; R2 = Glucoslyl (2→1) ramnoside, (6→1) ramnoside; R3 = R4 = H | Alternanthera brasiliana (L.) Kuntze | Aerial parts | Araujo et al. (2014) |
| 11 | Vitexin or Apigenin-8-C-glucoside | 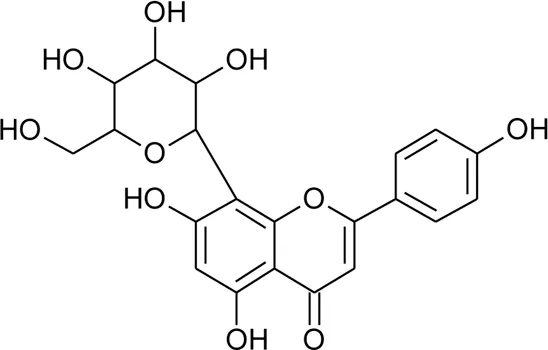 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera philoxeroides (Mart.) Griseb., Alternanthera hirtula (Mart.) R.E.Fr., Alternanthera praelonga A.St.-Hil., Alternanthera littoralis P.Beauv | Aerial parts; Leaves | Salvador and Dias, (2004) |
| Correa et al. (2016) | |||||
| Deladino et al. (2017) | |||||
  | |||||
| (β-D-glucopyranosyl) | |||||
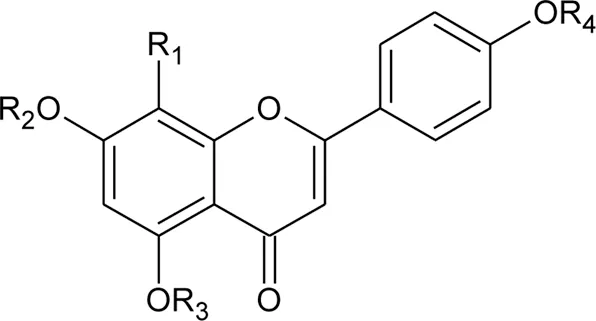
| 12 | 7-O-β-D-glucopyranosyl-6-C-β- D-glucopyranosyl-apigenin | R1 = H; R2 = R3 = β-D-glucopyranosyl | Alternanthera bettzickiana (Regel) G.Nicholson | Flower | Petrus et al. (2014b) |
| 13 | 6-C-β- D-glucopyranosyl-apigenin | R1 = R3 = H; R2 = β-D-glucopyranosyl | Alternanthera bettzickiana (Regel) G.Nicholson | Flower | Petrus et al. (2014b) |
| 14 | 8-C-β- D-glucopyranosyl-apigenin | R1 = β-D-glucopyranosyl; R2 = R3 = H | Alternanthera bettzickiana (Regel) G.Nicholson | Flower | Petrus et al. (2014b) |
| 15 | 5,7,4′-trihydroxyflavone | R1 = R2 = R3 = H | Alternanthera bettzickiana (Regel) G.Nicholson | Flowers | Petrus et al. (2014a) |
| 16 | Isovitexin | 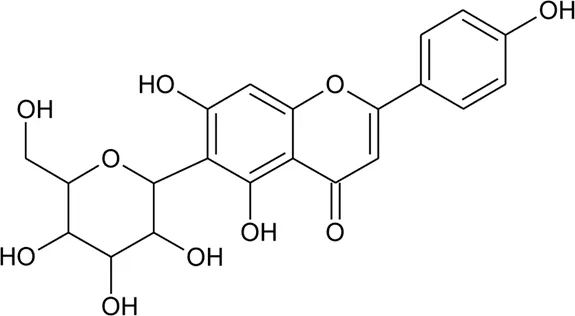 | Alternanthera littoralis P.Beauv | Aerial parts | Salvador and Dias, (2004) |
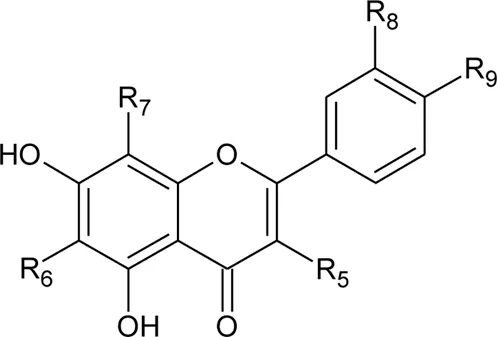 | |||||
| 17 | Kaempferol | R5 = R9 = OH; R6 = R7 = R8 = H | Alternanthera brasiliana (L.) Kuntze, Alternanthera littoralis P.Beauv., Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts; Leaves; Whole Plant | Salvador and Dias, (2004) |
| Salvador et al. (2006) | |||||
| Salvador et al. (2009) | |||||
| Deladino et al. (2017) | |||||
| 18 | Quercetin-3-methyl ether | R5 = OCH3; R6 = R7 = H; R8 = R9 = OH | Alternanthera littoralis P.Beauv.; Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts | Salvador and Dias, (2004) |
| Souza et al. (2007) | |||||
| Salvador et al. (2009) | |||||
| 19 | Quercetin | R5 = R8 = R9 = OH; R6 = R7 = H | Alternanthera brasiliana (L.) Kuntze, Alternanthera littoralis P.Beauv.; Alternanthera sessilis (L.) R.Br. ex DC.; Alternanthera hirtula (Mart.) R.E.Fr.; Alternanthera philoxeroides (Mart.) Griseb | Aerial parts; Whole plant | Salvador and Dias, (2004) |
| Salvador et al. (2006) | |||||
| Souza et al. (2007) | |||||
| Fan, (2008) | |||||
| Salvador et al. (2009) | |||||
| Correa et al. (2016) | |||||
| Deladino et al. (2017) | |||||
| Vani et al. (2018) | |||||
| Zhang et al. (2018) | |||||
| 20 | Luteolin | R5 = R6 = R7 = H; R8 = R9 = OH | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 21 | 2″-O-α-L-rhamnopyranosyl vitexin | R5 = R6 = R8 = H; R7 = C-Glu′′′→2′′ Rha (d); R9 = OH | Alternanthera brasiliana (L.) Kuntze, Alternanthera littoralis P.Beauv.; Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts; whole plant | Salvador and Dias, (2004) |
| Salvador et al. (2006) | |||||
| Souza et al. (2007) | |||||
| Salvador et al. (2009) | |||||
| Deladino et al. (2017) | |||||
| 22 | 2″-O-β-D-glucopyranosyl vitexin | R5 = R6 = R8 = H; R7 = C-Glu′′′→2′′ Glu (d); R9 = OH | Alternanthera brasiliana (L.) Kuntze, Alternanthera littoralis P.Beauv.; Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts; whole plant | Salvador and Dias, (2004) |
| Salvador et al. (2006) | |||||
| Souza et al. (2007) | |||||
| Salvador et al. (2009) | |||||
| Deladino et al. (2017) | |||||
| 23 | Acacetin 8-c-[α-L-rhamnopyranoyl-(1→2)-β-D-glucopyranoside] | R5 = R6 = R8 = H; R7 = C-Glu′′′→2′′ Rha (d); R9 = OCH3 | Alternanthera littoralis P.Beauv., Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts; whole plant | Salvador et al. (2006) |
| Souza et al. (2007) | |||||
| Salvador et al. (2009) | |||||
| 24 | Quercetin 3-O-α-L-rhamnosyl-(1→6)-β-D-glucopyranoside | R5 = d; R6 = H; R7 = H; R8 = OH; R9 = OH | Alternanthera littoralis P.Beauv | Aerial parts | Souza et al. (2007) |
| 25 | Isorhamnetin 3-O-α-L-rhamnosyl-(1→6)-β-D-glucopyranoside | R5 = d; R6 = H; R7 = H; R8 = OH; R9 = OCH3 | Alternanthera littoralis P.Beauv | Aerial parts | Souza et al. (2007) |
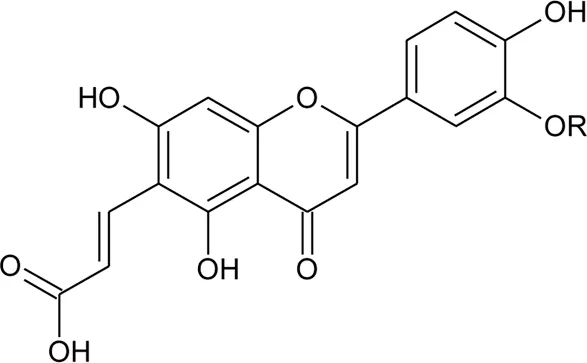 | |||||
| 26 | Torosaflavone E | R = CH3 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Khamphukdee et al. (2018) |
| 27 | Demethyl torosaflavone D | R = H | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Khamphukdee et al. (2018) |
| 28 | Luteolin-8-C-E-propenoic acid | 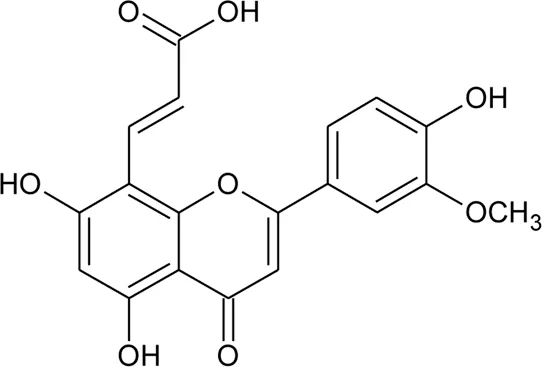 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Khamphukdee et al. (2018) |
| 29 | Chrysoeriol-7-O-rhamnoside | 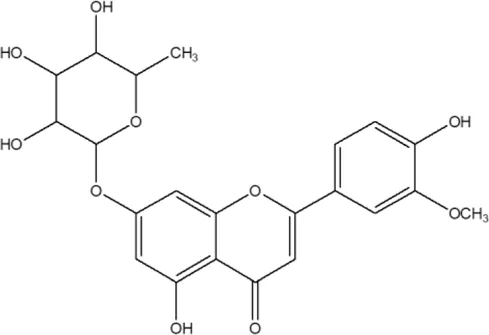 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Khamphukdee et al. (2018) |
 | |||||
| 30 | Crysoeriol (5,7,4′-trihydroxy-3′-methoxyflavone) | R1 = R2 = R3 = R4 = R5 = H; R6 = OCH3 | Alternanthera brasiliana (L.) Kuntze | Flowers | Facundo et al. (2012) |
| 31 | Tricin (5,7,4 -trihydroxy-3′,5′ -dimethoxyflavone) | R1 = R2 = R3 = R5 = H; R4 = R6 = OCH3 | Alternanthera brasiliana (L.) Kuntze | Flowers | Facundo et al. (2012) |
| 32 | 7-O-β-D-glucopyranoside-5,4′-dihydroxy-3′-methoxyflavone | R1 = R2 = R4 = R5 = H; R6 = OCH3; R3 = O-β-D-glucopyranoside | Alternanthera brasiliana (L.) Kuntze | Flowers | Facundo et al. (2012) |
  | |||||
  | |||||
| 33 | Kaempferol-3-O-robinobioside-7-O-α-L-rhamnopyranoside or Robinin or Kaempferol-3-O-rutinoside-7-O-α-L-rhamnopyranoside | R1 = a; R2 = b; R3 = H | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Brochado et al. (2003) |
| Deladino et al. (2017) | |||||
| 34 | Kaempferol-7- O-glucoside | R1 = c; R2 = H; R3 = H | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 35 | Quercetin 3-β-D-glucoside | R1 = H; R2 = c; R3 = H | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 36 | Quercetin-3-O-robinobioside-7-O-α-L-rhamnopyranoside or Clovin | R1 = a; R2 = b; R3 = OH | Alternanthera brasiliana (L.) Kuntze | Leaves | Brochado et al. (2003) |
| 37 | Quercetin-3-O-robinobioside or Quercetin-3-O-rutinoside or Rutin | R1 = H; R2 = b; R3 = OH | Alternanthera brasiliana (L.) Kuntze, Alternanthera littoralis P.Beauv., Alternanthera sessilis (L.) R.Br. ex DC. | Leaves; Aerial parts | Brochado et al. (2003) |
| Salvador and Dias, (2004) | |||||
| Deladino et al. (2017) | |||||
| 38 | Kaempferol-3-O-robinobioside or Kaempferol-3-O-rutinoside | R1 = H; R2 = b; R3 = H | Alternanthera brasiliana (L.) Kuntze | Leaves | Brochado et al. (2003) |
| 39 | Isorhamnetin-3-O-robinobioside or Isorhamnetin-3-O-rutinoside | R1 = H; R2 = b; R3 = OCH3 | Alternanthera littoralis P.Beauv., Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves; Aerial parts | Salvador and Dias, (2004) |
| Deladino et al. (2017) | |||||
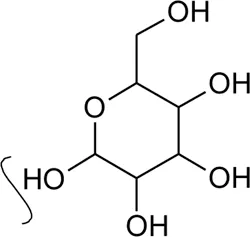
| 40 | Kaempferol-rhamnosyl- rhamnosyl-glycoside | 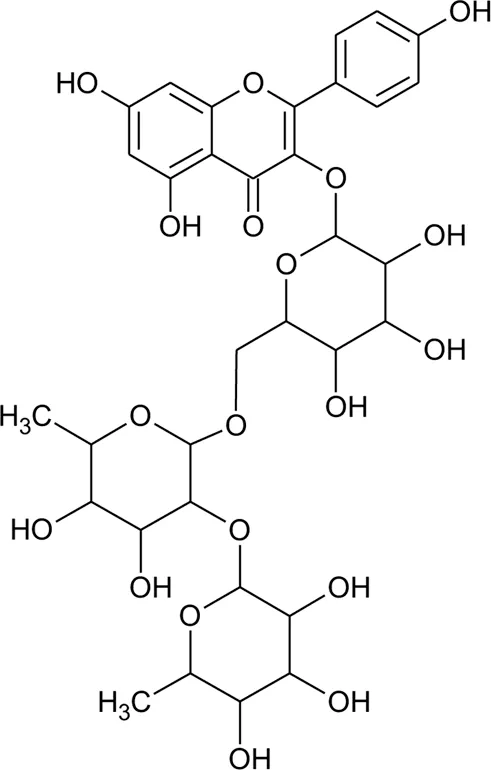 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| Volatile oil | |||||
| 41 | Limonene | 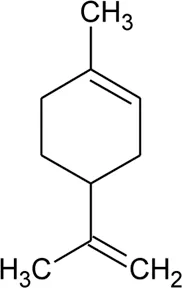 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 42 | α-Curcumene | 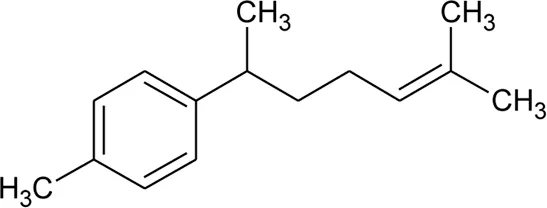 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 43 | Geraniol | 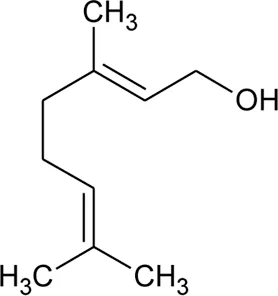 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 44 | Linalool | 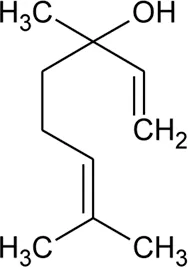 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 45 | Camphor | 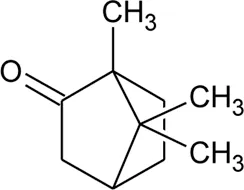 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 46 | Myrcene | 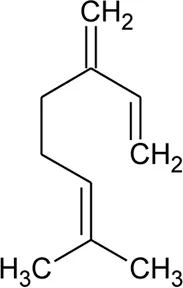 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 47 | Camphene | 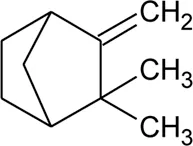 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 48 | α-pinene | 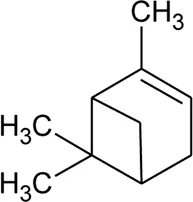 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| Sterols | |||||
| 49 | Stigmasterol | 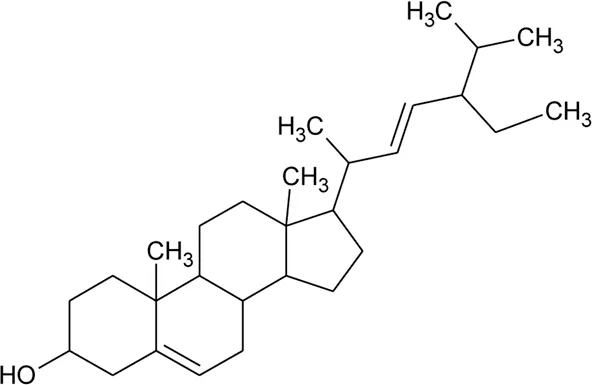 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Pereira et al. (2013) |
| Walter et al. (2014) | |||||
| 50 | Campesterol | 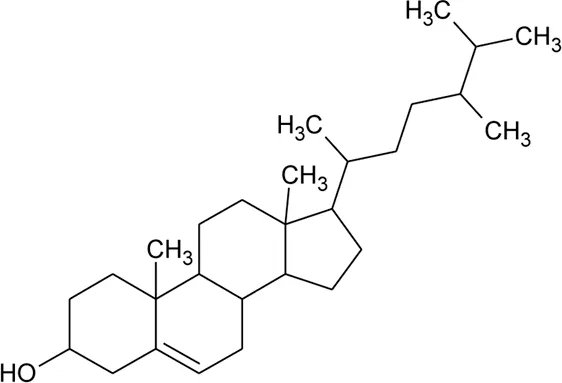 | Alternanthera sessilis (L.) R.Br. ex DC. | — | Walter et al. (2014) |
| 51 | β-Sitosterol | 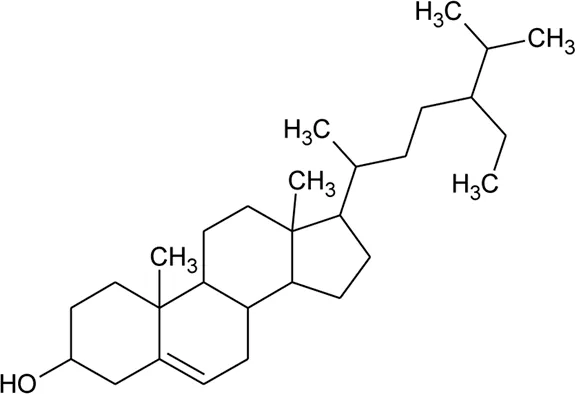 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera philoxeroides (Mart.) Griseb | Leaves | Fang et al. (2006) |
| Gupta and Singh, (2012b) | |||||
| Pereira et al. (2013) | |||||
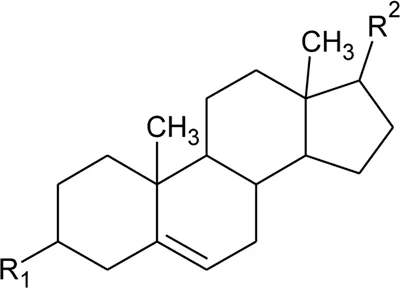 |  | ||||
  |  | ||||
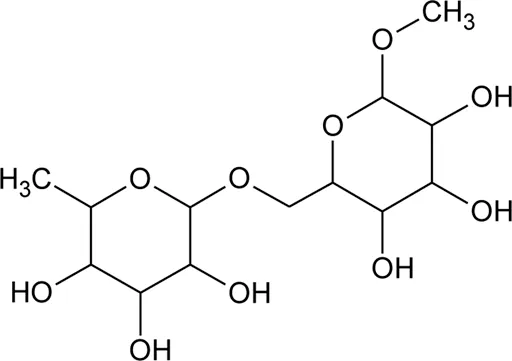 | |||||
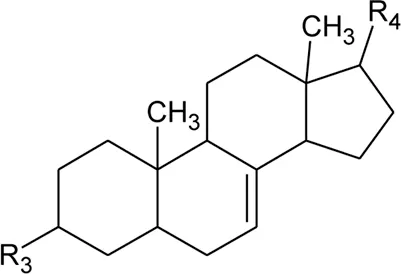
| 52 | Δ5-Stigmasterol or Stigmasteryl or Stigmasta-5, 22-dien-3-β-ol | R1 = OH; R2 = a | Alternanthera littoralis P.Beauv., Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera philoxeroides (Mart.) Griseb | Aerial parts; Whole plant | Salvador and Dias, (2004) |
| Fan, (2008) | |||||
| Salvador et al. (2009) | |||||
| 53 | β-Sitosterol | R1 = OH; R2 = b | Alternanthera sessilis (L.) R.Br. ex DC. | Whole plant | Salvador et al. (2009) |
| 54 | Campesterol | R1 = OH; R2 = c | Alternanthera sessilis (L.) R.Br. ex DC. | Whole plant | Salvador et al. (2009) |
| 55 | Δ7-Spinasterol or α-Spinasterol | R3 = OH; R4 = a | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera philoxeroides (Mart.) Griseb | Aerial parts; Whole plant | Salvador and Dias, (2004) |
| Fang et al. (2006) | |||||
| Fan, (2008) | |||||
| Salvador et al. (2009) | |||||
| Pereira et al. (2013) | |||||
| Walter et al. (2014) | |||||
| 56 | Δ7-Stigmasterol or Stigmast-7en-3-β-ol | R3 = OH; R4 = b | Alternanthera littoralis P.Beauv., Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts; Whole plant | Salvador and Dias, (2004) |
| Salvador et al. (2009) | |||||
| 57 | Stigmast-7enyl-3-β-ol-3-O-β-D-glucopyranoside or 3-O-β-D-Glucopyranosyl β-sitosterol | R1 = O-Glu; R2 = b | Alternanthera littoralis P.Beauv., Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts; Whole plant | Salvador and Dias, (2004) |
| Salvador et al. (2009) | |||||
| 58 | 3-O-β-D-Glucopyranosyl stigmasterol | R1 = O-Glu; R2 = a | Alternanthera sessilis (L.) R.Br. ex DC. | Whole plant | Salvador et al. (2009) |
| 59 | 3-O-β-D-Glucopyranosyl Δ7-stigmasterol | R3 = O-Glu; R4 = b | Alternanthera sessilis (L.) R.Br. ex DC. | Whole plant | Salvador et al. (2009) |
| 60 | 3-O-β-D-Glucopyranosyl spinasterol | R3 = O-Glu; R4 = a | Alternanthera sessilis (L.) R.Br. ex DC. | Whole plant | Salvador et al. (2009) |
| 61 | 6S,7E,9R-6,9-Di-hydroxymegastigma-4,7-dien-3-one-9-O-beta-D-glucopyranoside | 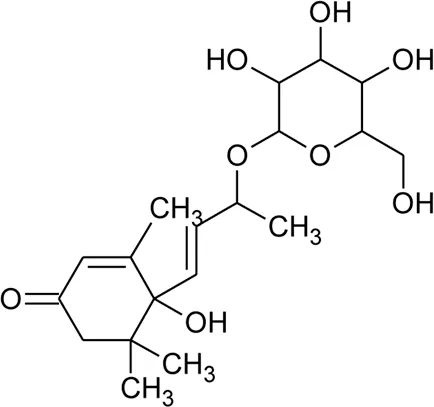 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2009b) |
| 62 | 3β-Hydroxystigmast-5-en-7-one | 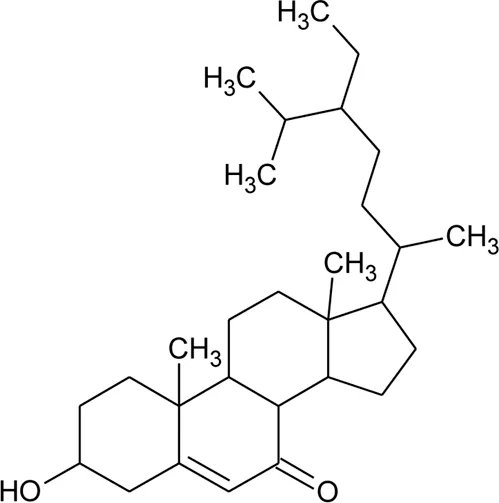 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
 | |||||
| 63 | Sitosterol-3-O-β-D-glucopyranoside | R = β-D-glucopyranoside | Alternanthera brasiliana (L.) Kuntze | Flowers | Facundo et al. (2012) |
| Triterpenoid/Saponins | |||||
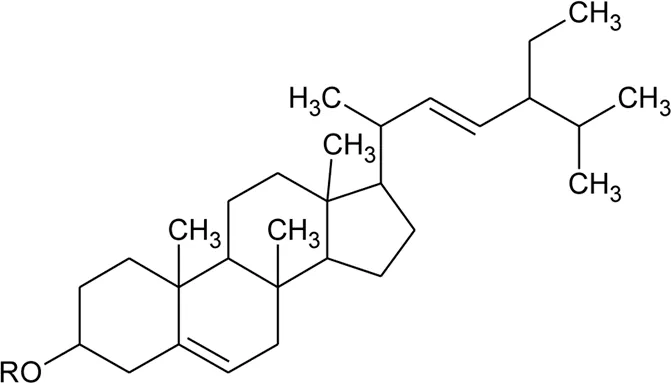
| 64 | Ursolic acid | 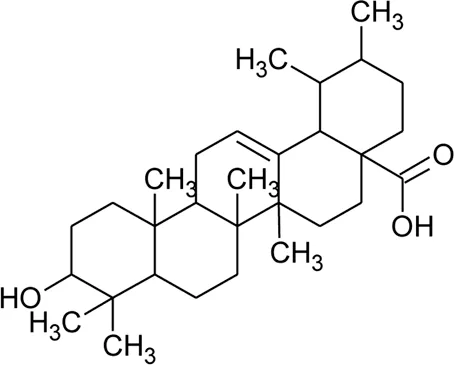 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 65 | Oleanolic acid 28-O-beta-D-glucopyranoside | 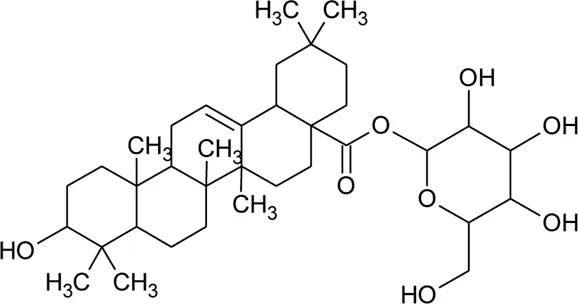 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2009b) |
 | |||||
| 66 | Oleanolic acid 3-O-beta-D-glucuronopyranoside-6′-O-methyl ester | R1 = CH3COO; R2 = CH3 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2009b) |
| 67 | Hederagenin 3-O-beta-D-glucuronopyranoside-6′-O-methyl ester | R1 = CH3COO; R2 = CH2OH | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2009b) |
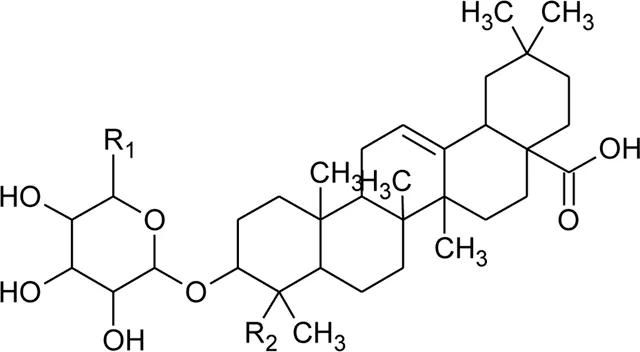
| 68 | Hederagenin-3-O-beta-D-glucuronopyranoside (HN-Saponin K) | R1 = R2 = CH2OH | Alternanthera philoxeroides (Mart.) Griseb | — | Guo et al. (2011) |
| 69 | Philoxeroideside A | 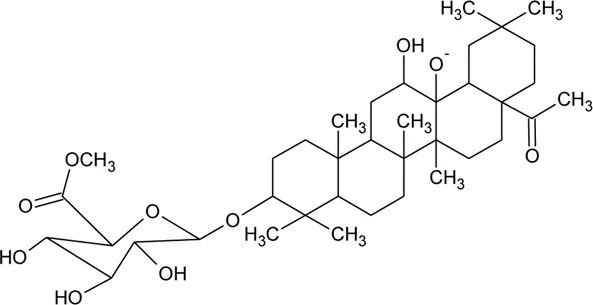 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2009a) |
| 70 | Philoxeroideside B | 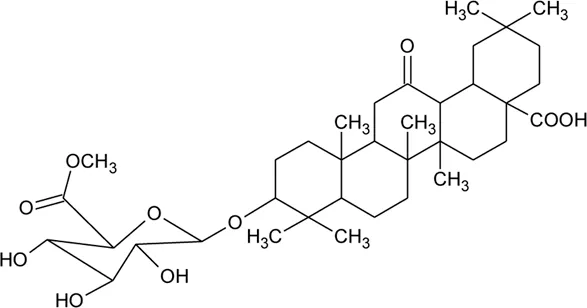 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2009a) |
| 71 | Philoxeroideside C | 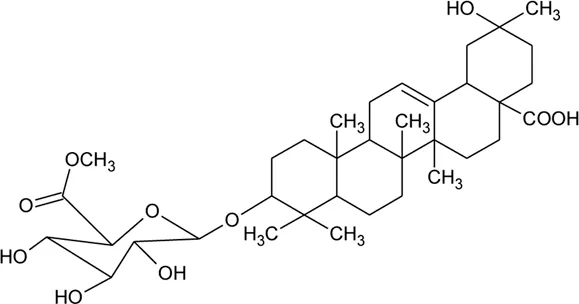 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2009a) |
| 72 | Philoxeroideside D | 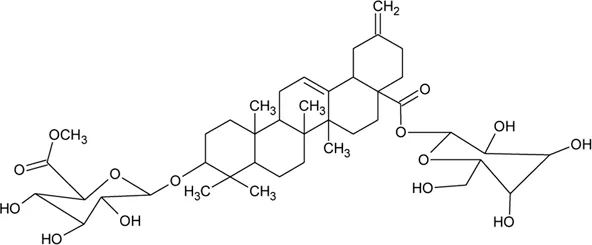 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2009a) |
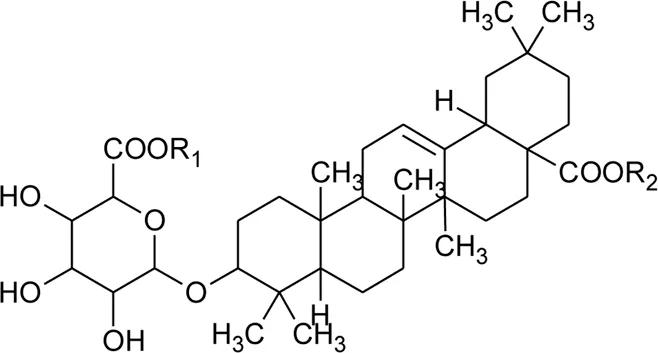  | |||||
| 73 | Chikusetsusaponin IVa or Oleanolic acid-3-O-beta-D-glucopyranosyl-28-Obeta-D-glucopyranosyl ester | R1 = H; R2 = a | Alternanthera philoxeroides (Mart.) Griseb | Whole plant | Rattanathongkom et al. (2009) |
| 74 | Chikusetsusaponin IV a methyl ester | R1 = CH3; R2 = a | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2009b) |
| 75 | Oleanolic acid 3-O-beta-D-glucuronopyranoside or Calenduloside E | R1 = R2 = H | Alternanthera philoxeroides (Mart.) Griseb | Whole plant | Fang et al. (2009b) |
| Rattanathongkom et al. (2009) | |||||
| Guo et al. (2011) | |||||
| 76 | Oleanolic acid | 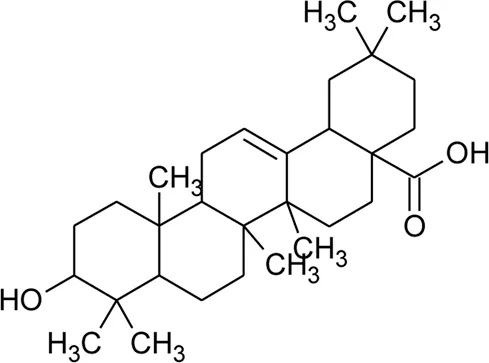 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2006) |
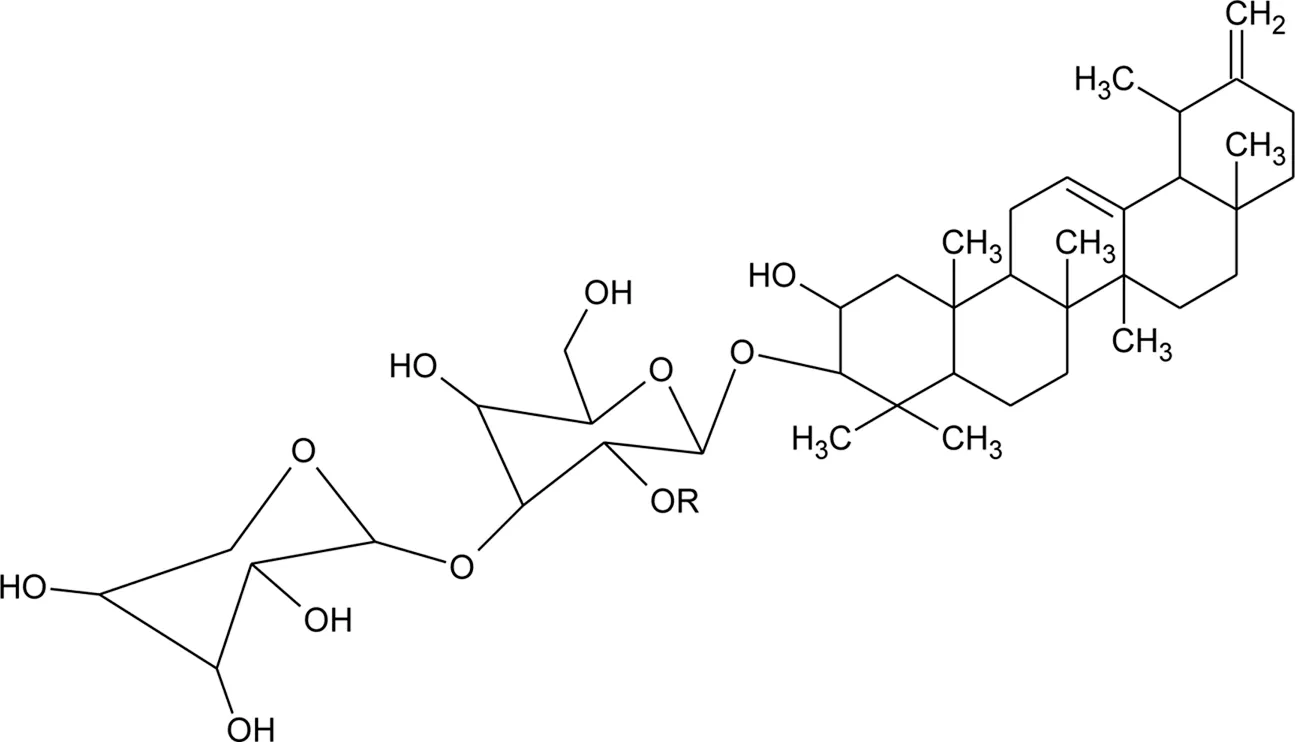 | |||||
| 77 | 2α, 3β-dihydroxyurs-12,20(30)-dien-28-oic acid | 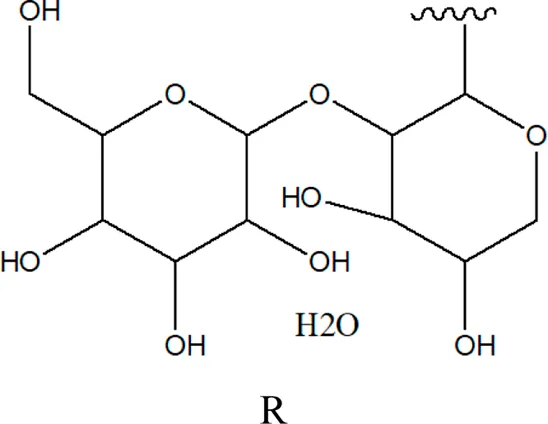 | Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts | Sanoko et al. (1999) |
| 78 | 2α,3β-dihydroxy urs-12,20(30)-dien-28-oic acid 3-O-{O-β -D-quinovopyranosyl-(1→2)-O-α-L- arabinopyranosyl- (1→2)-O-[β-D- xylopyranosyl-(1→3)] β-D-glucopyranoside} | 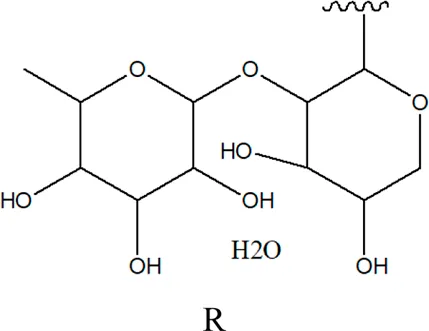 | Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts | Sanoko et al. (1999) |
| 79 | 2α,3β-dihydroxy urs-12,20(30)-dien-28-oic acid 3-O-{O-α -L- arabinopyranosyl -(1→2)-O-[β-D- xylopyranosyl-(1→3)] β-D-glucopyranoside} |  R R | Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts | Sanoko et al. (1999) |
| 80 | 2α,3β-dihydroxy urs-12,20(30)-dien-28-oic acid 3-O-{[O-β-D- xylopyranosyl-(1→3)] β-D-glucopyranoside} | R = H | Alternanthera sessilis (L.) R.Br. ex DC. | Aerial parts | Sanoko et al. (1999) |
| Phenolic compounds | |||||
| 81 | Ellagic acid | 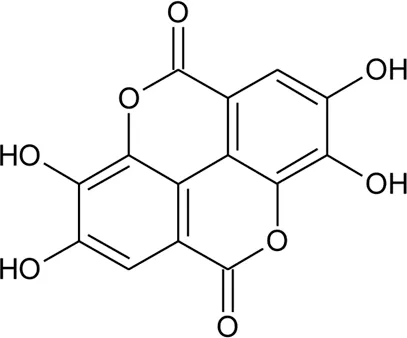 | Alternanthera sessilis (L.) R.Br. ex DC. | Whole plant | Mondal et al. (2015) |
| 82 | Caffeic acid | 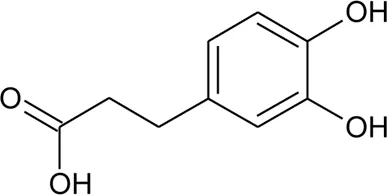 | Alternanthera philoxeroides (Mart.) Griseb., Alternanthera hirtula (Mart.) R.E.Fr., Alternanthera praelonga A.St.-Hil | Whole plant | Correa et al. (2016) |
| 83 | Quinic acid | 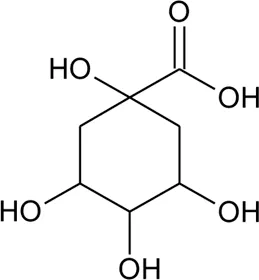 | Alternanthera philoxeroides (Mart.) Griseb., Alternanthera hirtula (Mart.) R.E.Fr., Alternanthera praelonga A.St.-Hil | Whole plant | Correa et al. (2016) |
| 84 | Ferulic acid | 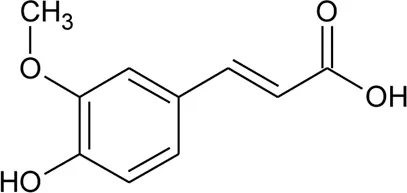 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera hirtula (Mart.) R.E.Fr., Alternanthera praelonga A.St.-Hil | Whole plant; leaves | Correa et al. (2016) |
| Deladino et al. (2017) | |||||
| 85 | p-Coumaric acid | 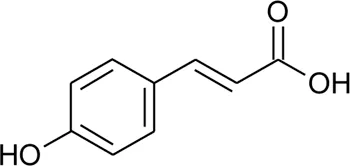 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera philoxeroides (Mart.) Griseb | Leaves; Aerial parts | Fan, (2008) |
| Deladino et al. (2017) | |||||
| 86 | 4-Hydroxybenzoic acid | 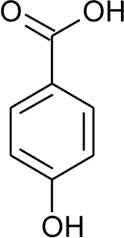 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 87 | 2,5-Dihydroxybenzoic acid or gentisic acid | 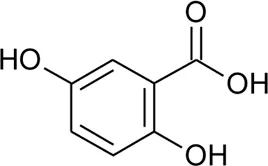 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 88 | Hydroxytyrosol | 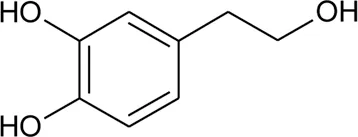 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 89 | Chlorogenic acid | 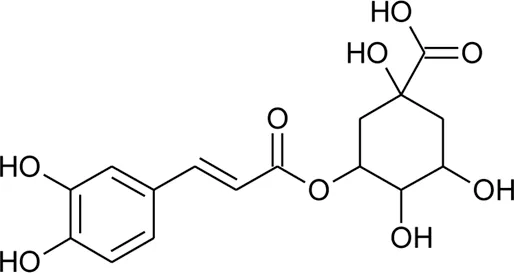 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 90 | 2,5-Dihydroxybenzoic acid 5-O-β-D-glucoside | 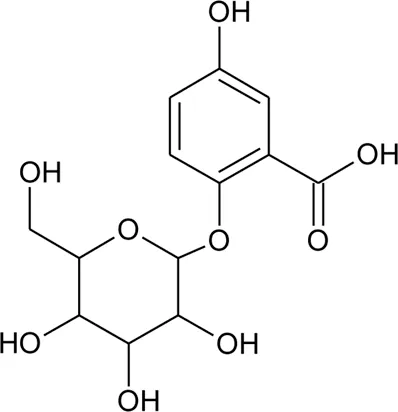 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| Ionone | |||||
| 91 | Ionone (Alcoholic derivative) | 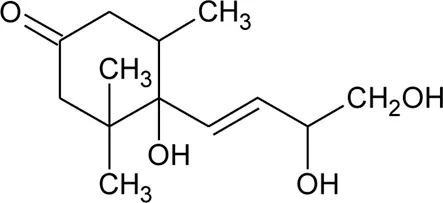 | Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Ragasa et al. (2010) |
| 92 | α-Ionone | 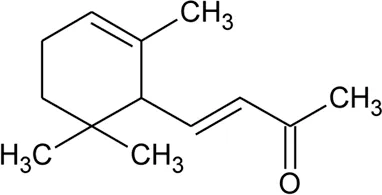 | Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Ragasa et al. (2010) |
| 93 | Ionone (Aldehyde derivative) | 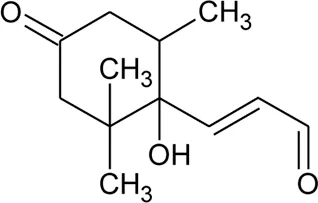 | Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Ragasa et al. (2010) |
| Anthraquinone | |||||
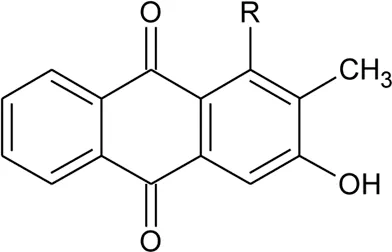 | |||||
| 94 | Rubiadin | R = OH | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| Collett and Taylor, (2019) | |||||
| 95 | Rubiadin l-methyl ether | R = OCH3 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 96 | 2-Hydroxy-3-methylanthraquinone | R = H | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 97 | Rhein | 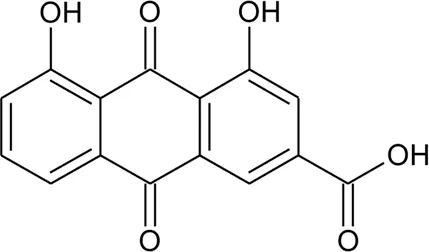 | Alternanthera pungens Kunth | Flowers | Gupta and Saxena, (1987) |
| Hydroxycinnamic acids | |||||
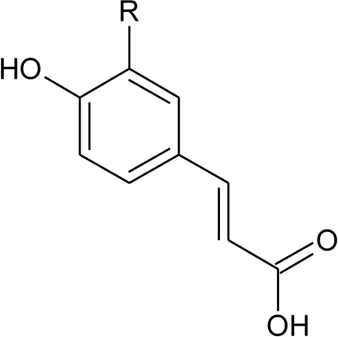 | |||||
| 98 | (E)-3-(4-hydroxyphenyl)prop-2-enoic acid | R = H | Alternanthera bettzickiana (Regel) G.Nicholson | Leaves | Petrus et al. (2014a) |
| 99 | (E)-3-(3,4-dihydroxyphenyl) prop-2-enoic acid | R = OH | Alternanthera bettzickiana (Regel) G.Nicholson | Leaves | Petrus et al. (2014a) |
| 100 | (E)-3-(4-hydroxy-3-methoxyphenyl) prop-2-enoic acid | R = OCH3 | Alternanthera bettzickiana (Regel) G.Nicholson | Leaves | Petrus et al. (2014a) |
| Alkaloids | |||||
| 101 | Alternamide A (7,8-dihydroxy-1,2,4,5-tetrahydro-3H -1,5-ethano[c]azepin-3-one) | 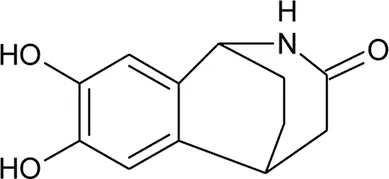 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 102 | Alternamide B (6,7-dihydroxy-3,4- dihydroquinoline-1-one) | 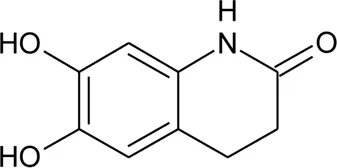 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 103 | Alternamine A [(R)-1-(3,4-dihydroxyphenyl)-1,2,3,4-tetrahydroisoquinoline-6,7-diol)] | 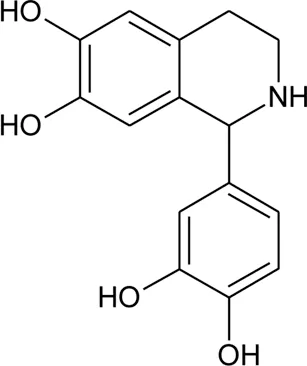 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 104 | N -(3,4-Dihydroxyphenethyl) formamide | 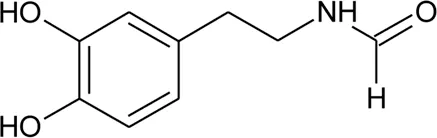 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 105 | Alternamine B {4-(2-aminoethyl) benzene-1,2-diol-4-(2-aminoethyl)benzene-1,2-diol-b -D –glucopyranose} | 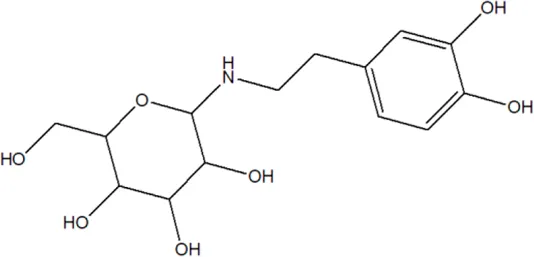 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 106 | Uridine | 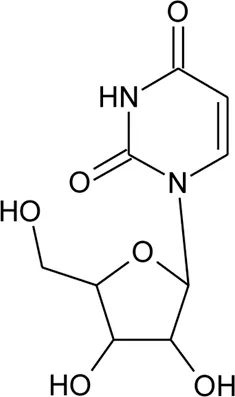 | Alternanthera littoralis P.Beauv | Aerial parts | Koolen et al. (2017) |
| 107 | N-trans-feruloyl tyramine | 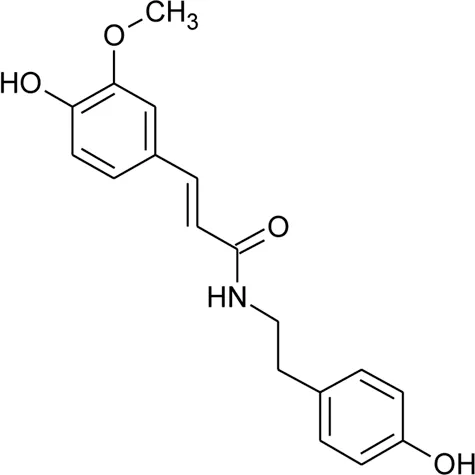 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
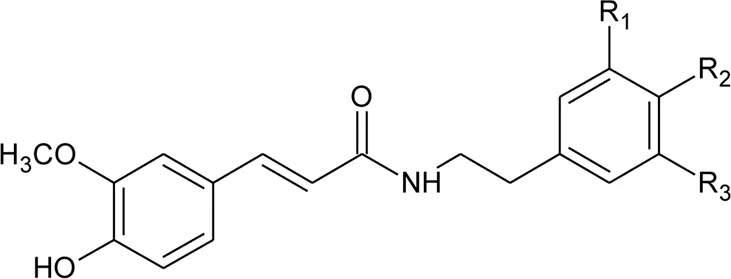 | |||||
| 108 | N-trans-feruloyl-3,5-dimethoxytyramine | R1 = OCH3; R2 = OH; R3 = OCH3 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2007) |
| 109 | N-trans-feruloyl-3-methyldopamine | R1 = OCH3; R2 = OH; R3 = H | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2007) |
| 110 | N-trans-feruloyl tyramine | R1 = H; R2 = OH; R3 = H | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2007) |
| 111 | N-cis-feruloyl tyramine | 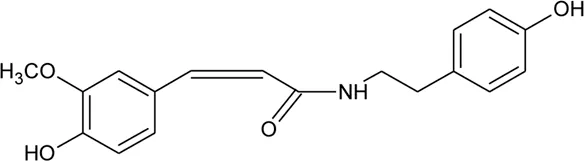 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fang et al. (2007) |
| 112 | β-Carboline | 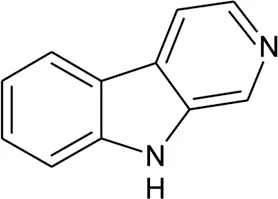 | Alternanthera philoxeroides (Mart.) Griseb | Leaves | Zhang et al. (2018) |
| Miscellaneous | |||||
| 113 | β-Carotene | 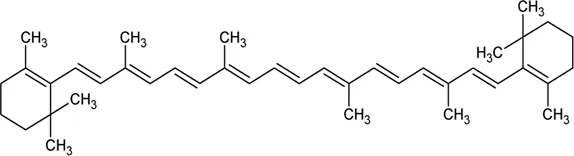 | Alternanthera sessilis (L.) R.Br. ex DC. | — | Walter et al. (2014) |
| 114 | Ricinoleic acid | 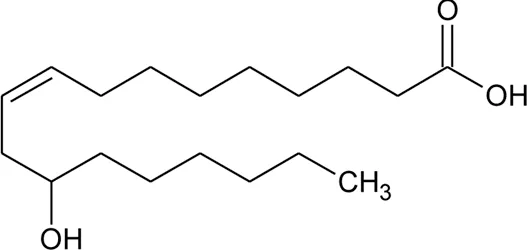 | Alternanthera sessilis (L.) R.Br. ex DC. | Seeds | Hosamani et al. (2004) |
| 115 | Malic acid | 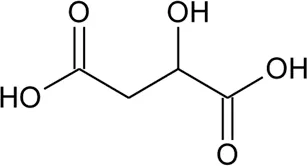 | Alternanthera philoxeroides (Mart.) Griseb., Alternanthera hirtula (Mart.) R.E.Fr | Leaves | Correa et al. (2016) |
 | |||||
| 116 | Indole-3-carboxaldehyde | R = CHO | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 117 | Indole-3-carboxylic acid | R = COOH | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 118 | Azelaic acid | 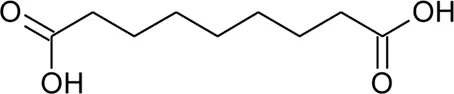 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 119 | Blumenol A | 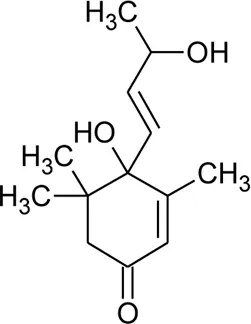 | Alternanthera philoxeroides (Mart.) Griseb | Aerial parts | Fan, (2008) |
| 120 | 4,5-Dihydroblumenol | 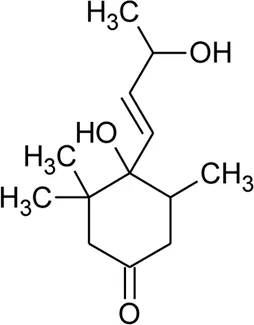 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2009b) |
| 121 | Cycloeucalenol | 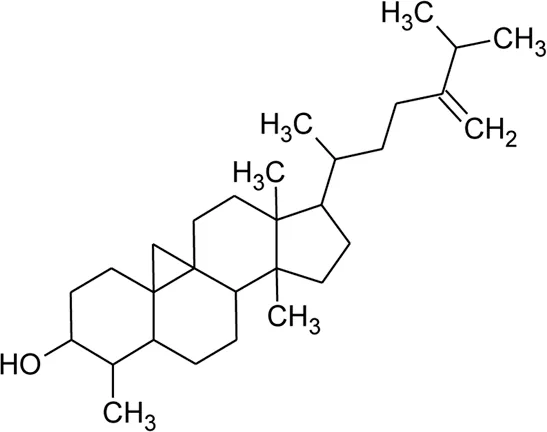 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2006) |
| 122 | Phytol | 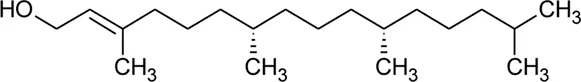 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2006) |
| 123 | Phaeophytin A | 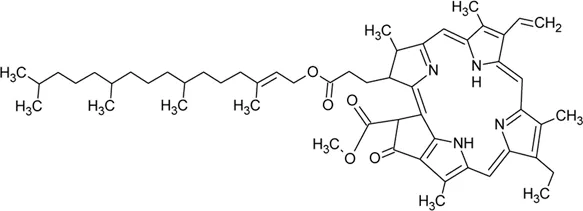 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2006) |
| 124 | Pheophytin A | 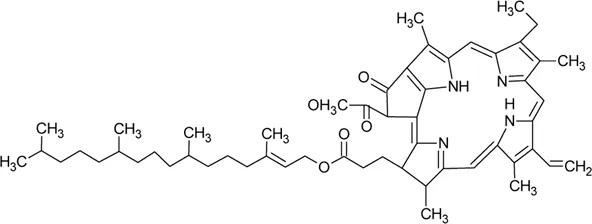 | Alternanthera philoxeroides (Mart.) Griseb | — | Fang et al. (2006) |
| 125 | 24-Methylene-cycloartanol | 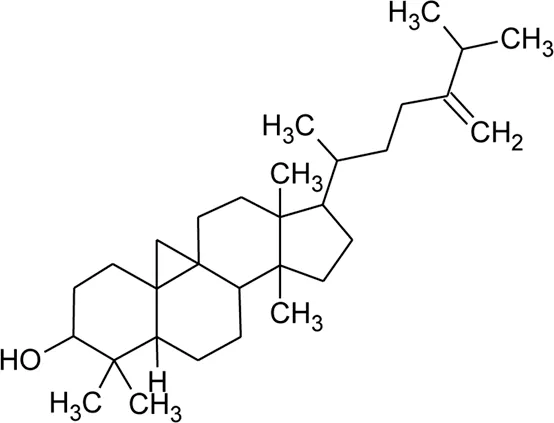 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
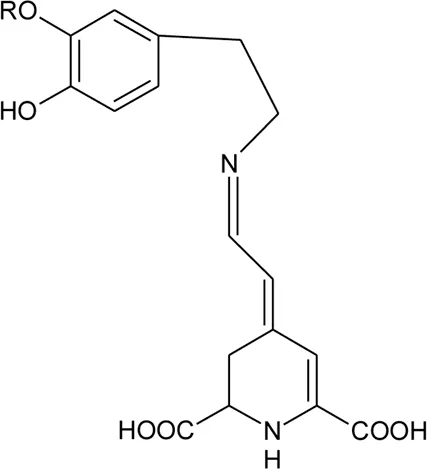 | |||||
| 126 | Dopamine-betaxanthin | R = H | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 127 | 3-Methoxytyramine-betaxanthin | R = CH3 | Alternanthera brasiliana (L.) Kuntze, Alternanthera sessilis (L.) R.Br. ex DC. | Leaves | Deladino et al. (2017) |
| 128 | Choline | 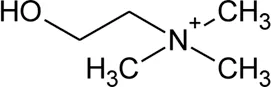 | Alternanthera pungens Kunth | — | De Ruiz et al. (1993) |
| 129 | Leucoantocyanidin | 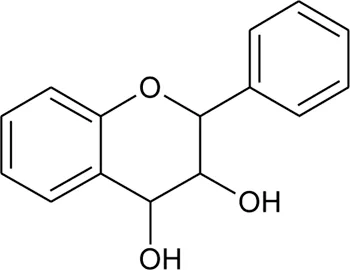 | Alternanthera pungens Kunth | Aerial parts | Petrus and Seetharaman, (2005) |
Chemical constituents isolated from genus Alternanthera.
The present review emphasizes traditional uses, chemical constituents, pharmacological actions, clinical potential, and safety profile of Alternanthera species. The current work has been compiled to fulfill the following goals: 1) to explore if traditional claims of Alternanthera species have been scientifically justified by pharmacological and clinical studies, and also to assess critically if their mechanism of actions is established, 2) to explore whether detailed phytochemical investigations have been conducted to detect and isolate main/bioactive constitutes of various species, 3) to reveal whether appropriate analytical methods have been developed for standardization of plant materials based on marker compounds, 4) to analyze whether isolated compounds from Alternanthera species have potential to be developed as lead molecules unaltered or needs derivatization to develop semisynthetic drugs through proper SAR studies and 5) to check if the safety and toxicity profiles of Alternanthera species have been studied. The scattered raw data has been compiled from online databases such as SciFinder, Google Scholar, PubMed, Science Direct, and Open J-Gate for 100 years up to April 2021 and offline databases such as Aromatic Plants Abstract, scientific journals, and books from different libraries of National repute. Keywords selected were based on various species of Alternanthera genus, and different biological activities. The articles which were in English and available with full text were included. Manuscript written in non-English versions were excluded. A total of 156 articles related to Alternanthera genus were finally studied and cited. But the cross-sectional literature review led us to cover a total of around 500 articles in this review article. The review article is categorized into six sections: 1) morphology emphasizes morphological characters of different Alternanthera species; 2) ethnopharmacology covers traditional uses of different Alternanthera species; 3) phytoconstituents includes name and structure of chemicals constituents isolated from various species of the genus; 4) biological activities focus on different pharmacological activities reported in various species and presented in the table; 5) toxicity studies include scientific reports of toxicity studies of different Alternanthera species and 6) clinical studies describe clinical trials conducted on humans.
Morphology
The morphological profile of various species of the genus was found to be similar with some variations. A. brasiliana (L.) Kuntze (a perennial herb mainly distributed in Brazil) is prostrate, 7.5–45 cm long branches, introducing a round stem, long internodes, and swollen nodes, at which inverse leaves connect (Kumar S. et al., 2011). Branches are glabrous, two lines of hair, nodes frequently villous; leaves are 2.5–7.5 cm, considerably longer when developing in watery spots, rather plump, at some point indefinitely denticulate; flowers are white, found in the form of bunches; seeds are 1.25–1.5 mm, sub-orbicular.
A. denticulata R. Br. and A. nahui Heenan and de Lange comprise stem of 100 mm height and located in an upright position (Heenan et al., 2009). The uniform spreading of minute hairs is present on the stems of both plants. The dark green-colored leaves (length—30 mm and breadth—6 mm) of both plants are linear, entire, narrow, elliptic, denticulate margins, and oblong in appearance. The abaxial surface of the tepals (length: 2.0–4.2 mm) is described by keeled, a character that is presented at the base of mature and dried tepals.
A. philoxeroides (Mart.) Griseb., a perennial herb, has stems crawling or gliding rising towards pinnacle, establishing at the lower hubs, branched, empty, with a longitudinal hairy groove score on two inverse sides (Pulipati et al., 2015). The fresh and delicious stems can develop on a level plane and float on the outside of the water, framing pontoons, or structure tangled bunches that develop onto banks. The leaves are inverse two by two, with an unmistakable midrib, and ranges from 5–10 cm. The plant consists of leaf, lanceolate shape, intense pinnacle, whole edge, glabrous surface, graduate base, and short strong petiole.
A. pungens Kunth is a perennial herb with a stem of 10–15 cm long with hair. The leaves are green in color and ovate in a shape of about 0.5–4.5 cm long and 0.3–2 cm in width (Naidu, 2012). It is native to the Southern American continent generally found in South Carolina, Florida, and California spreading around the road sides (Gupta et al., 2012). In 1918 it was first reported in the Southern parts of India (Rao, 2000).
A. sessilis (L.) R.Br. ex DC. is a perennial herb with purple-colored and glabrous branches grown from the root bases about 50 cm in length (Anitha and Kanimozhi, 2012). The fresh leaves are shiny, 1.3–3.0 cm long and 0.5–1.0 cm wide however the leaves are bigger in wet living spaces, direct elliptic, oval or obovate, zenith adjusted and base cuneate. The blossoms are subtle, white, borne in little, axillary heads; bracts are obovate and 1 mm long. The bracteoles are shorter, persevering; subequal, and intense. Utricleare cordi-structure and are unequivocally compacted. The seeds are orbicular. The plant bears blossoms and natural products consistently.
Ethnopharmacology and Traditional Uses
The infusion of inflorescences of A. Brasiliana (L.) Kuntze with water is used in headaches, coughs, colds, and grippe (Hundiwale et al., 2012). The infusion of leaves with a cup of water has been used in the treatment of fever while a decoction of roots is used in diarrhea. Traditionally, the various plant parts (stems, leaves, flowers, roots) of A. caracasana Kunth have been used to treat dysentery, diarrhea, and fever. The infusion of the plant is used as lavage or beverage in the traditional system of medicines (Canales-Martínez et al., 2008). The aerial parts of A. Brasiliana (L.) Kuntze are indicated in the treatment of inflammation, pain, and various infections (Hundiwale et al., 2012). The leaves of A. ficoidea (L.) P.Beauv. has been used in the treatment of heart and cancer problems (Patil and Kore, 2019). A. littoralis P. Beauv. has a long tradition of use in the treatment of infectious and inflammatory diseases (Koolen et al., 2017). The old texts indicated the use of A. littoralis P. Beauv. in the treatment of inflammatory, infectious diseases (de Santana Aquino et al., 2015), viral infections, immunity problems, cancer, malaria, and diarrhea (Hundiwale et al., 2012; Sekar, 2012). A. nodiflora R.Br. has been in the treatment of skin, degenerative and microbial infections (Feka et al., 2014). A. paronychioides A.St.-Hil. has been used in the treatment of hyperuricemia, rheumatic arthritis, uremia, nephritis, gout, cystitis, diabetes, and systemic neuralgia in TCM (Wu et al., 2013). In Ayurveda, the syrup of the whole plant of A. philoxeroides (Mart.) Griseb. has been employed in the treatment of influenza (Hundiwale et al., 2012). The aqueous infusion of leaf and flower of A. porrigens (Jacq.) Kuntze has been recorded in old texts for the treatment of hepatic pain, kidney problems, and influenza. A. pungens Kunth has been employed as folk medicine in Argentina, commonly known as Yerba del pollo, recorded in the Pharmacopeia National Argentina (1978) for various medicinal purposes. It has been traditionally used in the treatment of swelling, nasopharyngeal infections, as a painkiller in labor pain, and also for lactation stimulus in veterinary-related cases (Burkill, 1985). It is also used in the treatment of gonorrhea (Semenya and Potgieter, 2014), menstrual disorder, miscarriage (Lucky and Diame, 2010) and to treat dysentery, cholera, and many parasitic diseases (Grønhaug et al., 2008; Guede et al., 2010). In Sudan, it is used in aqueous form for the treatment of cough. In Brazil, the aerial parts are used against grippe and vermifuge (Agra et al., 2007). It is used for crushing kidney stones or renal calculi in the form of decoction. The whole plant of A. sessilis (L.) R.Br. ex DC. has been used as green vegetable for maintain the nutrient balance in body (Astudillo-Vázquez et al., 2008). The roasted leaves and stems (p.o.) of A. sessilis (L.) R.Br. ex DC. have been in the treatment of stomach pain, ulcer, and gastric problems (Kumar S. M. et al., 2011). The aerial parts of A. sessilis (L.) R.Br. ex DC. have been used as a diuretic in the Ayurvedic system of medicines (Hundiwale et al., 2012). The leaves of A. sessilis (L.) R.Br. ex DC. are used as a diuretic, antipyretic and antiseptic and roots are used as amenorrhea, inflammations, ovarian diseases, and female sterility. The young shoots of A. sessilis (L.) R.Br. ex DC. have been used as lactagogue and febrifuge (Hosamani et al., 2004). Keeping these in mind, the most common traditional uses for the Alternanthera species were recorded for the treatment and management of inflammation, pain, infectious diseases, and gastric problems.
Phytoconstituents Isolated and Identified in Alternanthera Species
GC–MS of n-hexane extract of A. philoxeroides (Mart.) Griseb. leaves showed the presence of 25 compounds. Among this Acetic acid, 2-(2-methoxycarbonylamino-5-nitrophenylthio)-, methyl ester (31.92%); 1,4-Benzenediol, 2,5-bis(1,1-dimethylethyl) (15.06%); 4-Pyridinecarboxamide, 6-bromo-4,5-dicyano-1,2,3,4-tetrahydro-3,3-dimethyl-2-[[(1methylethyamino] oxy] (8.53%); L-Cysteine, N-(trifluoroacetyl)-, butyl ester, trifluoroacetate (ester) (6.59%); Cyclopentaneundecanoic acid, methyl ester (5.4%) and 3-Bromo-N-(2-thiazolyl) benzamide (3.49%) are dominant (Akbar et al., 2021). LC-MS/MS and GC-MS analysis of an ethanolic extract of A. brasiliana (L.) Kuntze aerial parts were performed (Alencar Filho et al., 2019). Five compounds (luteolin-8-C-rhamnosylglucoside, 2″-O-rhamnosylvitexin, 2″-O-rhamnosyl-6-C-glucosyl methyl-luteolin, rutin, and 2″-O-rhamnosylswertisin) were identified by LC-MS/MS whereas twenty-two compounds were identified by GC-MS but major proportions were n-hexadecanoic acid with 16.61% followed by linoleic acid, clionasterol, α-tocopherol, stigmast-7-en-3-ol, and α-amyrin. The GC-MS analysis of volatile oil obtained from leaves of A. pungens Kunth showed the presence of 12 compounds and the major compound was β-ionone (42.18%) (Ogunmoye et al., 2020). Other compounds identified were Hexahydrofarnesyl acetone (15.53%), Methyl palmitate (6.13%), 1-Octadecyne (4.72%), Undecane (3.73%), p-Metha-1,3,8-triene (3.65%), Isophytol (3.21%), δ-Cadinene (3.06%), 1,2-Dimethyl cyclooctene (3.05%), p-Cymene (2.96%), Phytol (2.67%) and Neophytadiene (2.50%).
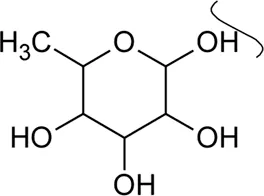
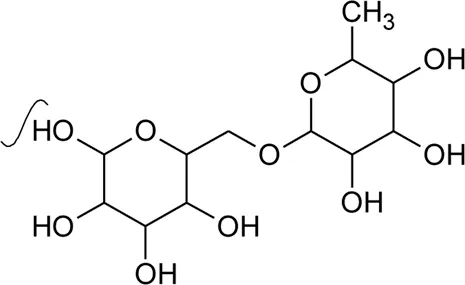
The phytoconstituents—benzopyran, flavonoids, volatile oil, sterols, triterpenoid/saponins, phenolic compounds, ionone, anthraquinone, hydroxycinnamic acids, alkaloids, etc. have been scientifically reported from 9 species of Alternanthera. The chemical constituents (along with their structure) isolated from different species of the Alternanthera genus are shown in Table 1.
Referring to the data tabulated in Table 1 covering the isolated phytoconstituents from 9 species of Alternanthera genus, we have prepared an interactive mapping (Figure 2) to give some quick insight about it to the readers. Notably, it has also been observed that some of the phytocompounds like kaempferol, stigmasterol, quercetin, vitexin, ferulic acid, caffeic acid, etc have been isolated from various species of Alternanthera genus. This somehow lead us to suggest that these phytocompounds could serve as standardization of these markers could be helpful in identifying Alternanthera species, and avoid adulteration. Some of the compounds isolated from the species of Alternanthera genus are very common and usually been reported from multiple biological sources and well known for many pharmacological activities. For instance, kaempferol has been isolated from various other sources including Euonymus alatus (Thunb.) Siebold (Fang et al., 2008; Singla et al., 2021), Vachellia nilotica (L.) P.J.H.Hurter and Mabb.(Singh et al., 2008), etc, with multiple therapeutic potential, including but not limited to antiproliferative (Park et al., 2021), antiviral (Arabyan et al., 2021), hepatoprotective (Alshehri et al., 2021), antioxidant (Sharma et al., 2021), etc. Similarly, chlorogenic acid had been reported from multiple resources, including Cocos nucifera L. (Bankar et al., 2011), apple fruit (Hulme, 1953), Neolamarckia cadamba (Roxb.) Bosser (Kapil et al., 1995), etc with multiple therapeutic potential like neuroprotective (Hung et al., 2021), antihepatotoxic (Kapil et al., 1995), etc. Since species of Alternanthera genus containing other compounds also along with these common phytomolecules, there could be a possibility of synergistic potential and enhanced activity. Thus, we suggest the researchers to explore the therapeutic potential based on the common bioactive compounds.
FIGURE 2
Pharmacological Activities
Several scientific investigations were conducted to validate traditional claims of various species of Alternanthera. Uncharacterized/non-standardized crude extracts of various species of Alternanthera were used in most of these scientific pharmacological studies. Alternanthera species have been observed to display analgesic, anticancer, anti-inflammatory, antimicrobial, antioxidant, hepatoprotective, hypotensive, allelopathic, α-glucosidase inhibitory, anthelmintic, anti-allergic, antianxiety, sedative, antiapoptotic, antiarthritic, antiasthmatic, anticataract, anticonvulsant, antidepressant, antidiabetic, antidiarrhoeal, antifungal, antibacterial, anti-HBV, antiparkinsonian, antiprotozoal, antispasmodic, antiviral, gastrointestinal protective, immunomodulatory and wound healing activities. The plant species, extract/fraction/isolate, dose tested/route of administration, bioactive dose, positive control, negative control, In vivo/in vitro models, and mechanism of action have been summarized in Table 2.
TABLE 2
| S. No | Pharmacological activity | Species | Extract/fraction/isolate | Dose tested/route of administration | Bioactive dose (mg/kg, IC50, etc) | Positive control | Negative control | Animals | Experimental model (In vivo/in vitro) | Mechanism of action | References |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Analgesic | Alternanthera brasiliana (L.) Kuntze | Aqueous extract of aerial parts | 25, 50, 100, 200 and 400 mg/kg, p.o | 400 mg/kg, p.o | Dipyrone (100 mg/kg, p.o.) | Distilled water (10 ml/kg, p.o.) | Male Swiss mice | In vivo - Abdominal contractions induced by acetic acid | Inhibition of the synthesis of prostaglandins and avoid the sensitization of receptors | Pelisoli Formagio et al. (2012) |
| Ethanolic extract of leaves | 25, 50 and 100 mg/kg, p.o | 50 and 100 mg/kg, p.o | Indomethacin (10 mg/kg, p.o.) | 0.9% saline solution | Mice of the Mus musculus strain of the Swiss line | In vivo—Formalin test | Suppression of proinflammatory cytokine expression and inhibition of NFκB pathway and the mitogen-activated protein kinase pathway | Coutinho et al. (2017) | |||
| Alternanthera littoralis P.Beauv | Ethanolic extract of aerial parts and 2″-O-α-L-rhamnopyranosylvitexin | 100, 300, 500 mg/kg, p.o. and 1, 10, 20, 50 mg/kg, p.o.; 0.3–300 μg/paw local | 100, 300 mg/kg, p.o. and 50 mg/kg, p.o.; 0.3–300 μg/paw local | Dexamethasone (1 mg/kg, s.c.) | 0.9% saline solution | Adult male and female Swiss mice | In vivo—carrageenan, TNF, or L-DOPA-induced hyperalgesia model | Act via prevented a Cg-induced decrease in the threshold of mechanical sensitivity | de Santana Aquino et al. (2015) | ||
| Alternanthera philoxeroides (Mart.) Griseb | Methanolic extract of the whole plant | 50, 100, 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Aspirin (200 and 400 mg/kg, p.o.) | Acetic acid (1%, 10 ml/kg, i.p.) and 1% Tween 80 in water, (10 ml/kg) | Swiss albino mice | In vivo—Acetic acid-induced constriction method | Act via inhibition of prostaglandin synthesis, cyclooxygenases, and lipo-oxygenases expression | Khatun et al. (2012) | ||
| Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of leaves | 250 and 500 mg/kg, p.o | 500 mg/kg, p.o | Diclofenac sodium (25 mg/kg, p.o.) and morphine (5 mg/kg, i.p.) | Acetic acid (0.7%, 10 ml/kg, i.p.) | Young Swiss Albino mice | In vivo—acetic acid-induced writhing and hot-plate tests | Act via inhibition of IL-4, IL-5, and IL-13 | Mondal et al. (2014) | ||
| Hydroethanolic extract of leaves | 250 and 500 mg/kg, p.o | 250 and 500 mg/kg, p.o | Morphine (0.5 mg/kg, i.p.) and analgin (50 mg/kg, i.p.) | Acetic acid (3%, 0.1 ml/10 g, i.p.) | Swiss Albino mice | In vivo—Acetic acid-induced writhing and Eddy’s hot plate methods | Act via centrally modulating mechanisms involving opiate, dopaminergic descending noradrenergic, and serotonergic receptor systems or maybe by peripherally inhibiting the prostaglandins, leukotrienes, and other endogenous substances | Mohapatra et al. (2018) | |||
| Methanolic extract of aerial parts | 50, 100, 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Aspirin (200 and 400 mg/kg, p.o.) | 1% Tween 80 in water, 10 ml/kg and 1% acetic acid (10 ml/kg i.p.) | Swiss albino mice | In vivo - acetic acid-induced pain model | Act via preventing prostaglandin production through inhibition of lipooxygenase and cyclooxygenase | Hossain et al. (2014) | |||
| Ethanolic extract of aerial parts | 200 and 400 mg/kg, p.o | 400 mg/kg, p.o | Diclofenac sodium (100 mg/kg, po.) | Saline (10 mg/kg, p.o.) | Female Swiss albino mice | In vivo: Acetic acid-induced writhing test and hot plate test | _____ | Mohaimenul et al. (2020) | |||
| 2 | Anthelmintic | Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous, methanolic, and acetone extracts of leaves | 25, 50, 75 and 100 mg | 25, 50, 75 and 100 mg | Albendazole (15 mg/ml) | 10% propylene glycol in normal saline | Indian adult earthworms Pheretima Posthuma | In vitro - Pheretima Posthuma method | Act via lysis of mucopolysaccharide membrane and cause paralysis or death of the worm | Vennila and Nivetha, (2015) |
| Methanol extract showed potent activity | |||||||||||
| Ethanolic extract of the whole plant and ellagic acid | 1.56–50 mg/ml and 0.09–3 mg/ml | 1.56–50 mg/ml and 0.09–3 mg/ml | Albendazole (1.5 mg/ml) | 1.0% tween-80 in phosphate-buffered saline | Haemonchus contortus strain | In vitro - Adult motility test | Act via disrupting cell membrane permeability through pore formation, the disintegration of integuments at a specific site, inhibition of cAMP phosphodiesterase and Na+/K+ ATPase | Mondal et al. (2015) | |||
| 3 | Antiallergic | Alternanthera sessilis (L.) R.Br. ex DC. | 95% Ethanolic extract of aerial parts | 25, 50 and 100 μg/ml | 25, 50 and 100 μg/ml | — | — | Rat basophilic leukemia cells | In vitro—Estimation of calcium, IL-6, TNF-α, IL-13, IL-4, lactate dehydrogenase release, β-hexosaminidase secretion assay, and Western Blot Analysis | Act via inhibition of antigen-stimulated secretion of TNF-α and IL-6 production and attenuates activation of NF-κB | Rayees et al. (2013) |
| 4 | Antianxiety | Alternanthera brasiliana (L.) Kuntze | Aqueous extract of leaves | 100, 200 and 400 mg/kg, p.o | 400 mg/kg, p.o | — | Distilled water (10 ml/kg, p.o.) | Male adult Wistar rats | In vivo—Elevated plus-maze model | --------- | Pelisoli Formagio et al. (2012) |
| Ethanolic extract of leaves | 250, 500 and 1,000 mg/kg, p.o | 1,000 mg/kg, p.o | Diazepam (1 mg/kg, i.p.) | Saline (10 ml/kg, p.o.) | Albino mice | In vivo—Hole board test and Elevated plus maze test | Benzodiazepine-like or GABA receptor-related action or 5-HT partial agonists like buspirone | Oyemitan et al. (2015) | |||
| Methanolic extract of leaves | 100, 300 and 600 mg/kg, i.p | 100, 300 and 600 mg/kg, i.p | Diazepam (1 mg/kg, i.p.) | Distilled water (10 ml/kg, p.o.) | Adult male Swiss albino mice | In vivo—Hole board test, open field test, elevated plus maze test, light/dark exploration test, and actophotometer test | Direct activation of GABA receptors | Barua et al. (2013) | |||
| Alternanthera philoxeroides (Mart.) Griseb | Ethanolic extract of leaves | 250 and 500 mg/kg, p.o | 250 and 500 mg/kg, p.o | 17β-Estradiol (1 mg/kg, i.p.) | Distilled water (10 ml/kg, p.o.) | Female ICR mice | In vivo—Elevated plus-maze test, Light/Dark transition test, and Locomotor activity test | Act via estrogenic activity | Khamphukdee et al. (2017) | ||
| 5 | Antiapoptotic | Alternanthera bettzickiana (Regel) G.Nicholson | Ethanolic extract of the whole plant | 20 and 50 μg/ml | 20 and 50 μg/ml | Quercetin (10, 20 mΜ) | High glucose (25 mmol/L) | — | In vitro—high glucose (25 mmol/l)-induced pancreatic β-cell apoptosis and dysfunction | Act via maintaining β-cell viability; suppressing reactive oxygen species production; inhibiting the activation of caspase-9, caspase-3, cleavage of poly (ADP-ribose) polymerase; upregulating pancreatic expression and the insulin secretagogue action of pancreatic β-cells | Wu et al. (2013) |
| 6 | Antiarthritic | Alternanthera bettzickiana (Regel) G.Nicholson | Ethanolic extract of aerial parts | In vivo study: 250, 500 and 1,000 mg/kg, p.o. for 28 days | Dose dependently significantly decreased paw swelling, MDA level, improved biochemical and hematological parameters, increased SOD and CAT levels | In vivo study: Diclofenac sodium (10 mg/kg, p.o.) | Distilled water | Wistar rats | In vivo: Paw swelling, Complete Freund’s Adjuvant induced arthritis | Downregulation of nuclear factor (NF)-kB, COX-2, interleukin (IL)-6, tumor necrosis factor (TNF)-α, and IL-1β and upregulation of IL-10, I-kB, and IL-4 | Manan et al. (2020) |
| In vitro study: 50–6,400 μg/ml | In vitro study: Diclofenac sodium (50–6,400 μg/ml) | In vitro: Egg Albumin Denaturation Inhibition, Inhibition of Protein Denaturation Using Bovine Serum Albumin | |||||||||
| Alternanthera philoxeroides (Mart.) Griseb., Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of leaves | 100–500 μg/ml | 500 μg/ml | Diclofenac sodium (100–500 μg/ml) | — | — | In vitro—Bovine Serum protein denaturation method and Egg albumin denaturation method | Inhibits thermally-induced protein denaturation | Sunmathi et al. (2016) | ||
| 7 | Antiasthmatic | Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of leaves | 500 mg/kg, p.o | 500 mg/kg, p.o | Mepyramine (8 mg/kg, p.o.) | Saline (1 ml/kg, p.o.) | Guinea pigs | In vivo—Bronchial hyperreactivity by Histamine aerosol induced bronchospasm in guinea pigs and broncho-alveolar lavage fluid (BALF) in egg albumin sensitized guinea pigs | Act via inhibition of antigen-induced histamine release or reduction in leucocyte count | Fathima et al. (2016) |
| 70% Ethanolic extract of the whole plant and its dichloromethane and aqueous fractions | — | 1–10 mg/kg i.p | Verapamil (1–10 m g/kg, i.p.) | Acetylcholine (1 μg/kg) | Sprague-Dawley albino rats | In vivo—acetylcholine chloride (Ach)-induced-bronchospasm | Act via calcium channel blocking potential | Saqib and Janbaz, (2016) | |||
| 8 | Anticancer/Cytotoxic | Alternanthera bettzickiana (Regel) G.Nicholson | Aqueous extract of leaves | 10–100 μM | 50–100 μM | β-actin antibody | — | Lung cancer (A549) cell lines | In vitro—MTT assay | Act via decline in cell proliferation, disturbances in the activity of mitochondrial membrane, the process of DNA fragmentation and apoptosis in cell line | Nagalingam et al. (2018) |
| Aqueous extract of leaves and silver nanoparticles and Ag-mesoporous MnO2 nanocomposite | 2.5–30 μM | 10, 30 μM | — | A group without extract/drug | Human HT-29 and SW620 colon cancer cell lines | In vitro—MTT assay | Cell death through the generation of intracellular oxidative stress | Jothi Ramalingam et al. (2017) | |||
| Alternanthera brasiliana (L.) Kuntze | Aqueous fraction of the ethanolic extract from the leaves, robinin, clovin, quercetin 3-O-robinobioside, kaempferol 3-O-robinobioside, kaempferol 3-O-rutinoside-7-O-α-L-rhamnopyranoside and kaempferol 3-O-rutinoside | 10–100 μg/ml | Kaempferol 3-O-robinobioside and kaempferol 3-O-rutinoside: IC50 = 25 μg/ml | Azathioprine | — | Human peripheral blood mononuclear cells | In vitro—Lymphocyte proliferation assay | Inhibition of the proliferative response of human T-cells | Brochado et al. (2003) | ||
| Ethyl acetate extract of leaves | 4, 8, 16, 32 and 64 μg/ml | IC50 = 33.54 and 33.69 μg | 5-Fluorouracil (20 mg/kg i.p.) | Distilled water, p.o., and Ehrlich ascites carcinoma cells (2 × 106 cells/mouse i.p.) | Ehrlich ascites carcinoma cells | In vitro—Trypan blue dye exclusion method and MTT assay | Decreased the levels of lipid peroxidation and significantly increased the levels of GSH, SOD, and catalase | Samudrala et al. (2015) | |||
| 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Swiss albino mice | In vivo—Ehrlich ascites carcinoma method | ||||||||
| Alternanthera philoxeroides (Mart.) Griseb | Alternanthin B; N-trans-feruloyl-3,5-dimethoxytyramine; alternanthin; N-trans-feruloyl-3-methyldopamine and N-trans-feruloyl tyramine | 10 and 30 μg/ml | 30 μg/ml | — | — | Hela and L929 cancer cell lines | In vitro—MTT assay | Cytotoxic effect against Hela and L929 cancer cell lines | Fang et al. (2007) | ||
| Philoxeroideside A-D | — | Philoxeroideside D IC50 = 37.29 (SK-N-SH) and 45.93 (HL60) μg/ml | — | — | SK-N-SH and HL60 cell lines | In vitro—3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide colorimetric assay | Cytotoxic effect against SK-N-SH and HL60 cell lines | Fang et al. (2009b) | |||
| Methanolic extract of leaves | 10, 20, 40, 80, 160 mg/ml | 160 mg/ml | — | Cardiomyocyte apoptosis induced by doxorubicin | H9c2 cell lines | In vitro—MTT assay and annexin V-FITC/PI staining assay | Decreased the cell apoptosis induced by doxorubicin | Zhang et al. (2018) | |||
| Alternanthera philoxeroides (Mart.) Griseb.; Alternanthera hirtula (Mart.) R.E.Fr. and Alternanthera praelonga A.St.-Hil | Ethanolic extract of the whole plant | 0.25, 2.5, 25 and 250 μg/ml | Exhibited mild activity | Doxorubicin | DMSO | UACC-62 (melanoma); MCF-7 (mamma); 786-O (kidney); NCI-H460 (lung); PC-3 (prostate); OVCAR-3 (ovary); HT-29 (colon); K562 (leukemia). Non cancer cell line: VERO (epithelial cell from green monkey kidney) | In vitro—MTT assay | Toxicity against cell lines | Correa et al. (2016) | ||
| Alternanthera sessilis (L.) R.Br. ex DC. | Methanolic extract of leaves | 0.05–10 mg/ml | IC50 = 6.5 mg/ml | — | — | Vero cell lines | In vitro—3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay method | — | Jain et al. (2016) | ||
| Silver nanoparticles of the aqueous extract | 1.56, 3.12, 6.25, 12.5, 25 μg/ml | IC50 = 6.85 μg/ml | — | Normal saline/DMSO | PC3 human prostate cancer cell line | In vitro—MTT assay | Apoptosis dependent pathway | Firdhouse and Lalitha, (2013) | |||
| Gold nanoparticles of the aqueous extract of leaves | 1–15 mg/ml | 10–15 mg/ml | — | A group without extract/drug | HeLa cervical cancer cell lines | In vitro—MTT assay | Act via modulating intrinsic apoptotic mechanisms in cervical cancer cells | Qian et al. (2019) | |||
| Aqueous extract of leaves and stems | 20–100 μg/ml | 20–100 μg/ml | — | A group without extract/drug | SIRC rabbit corneal cell line | In vitro—MTT assay | Act via inhibiting cytotoxic nature of the pathogen causing ocular diseases | Suganya et al. (2019) | |||
| n-hexane and methanolic extracts of aerial parts | 7.81, 15.625, 31.25, 62.5, 125 and 250 μg/ml | LC50values of methanol and n-hexane extracts are 255.4 and 925.68 μg/ml respectively | — | DMSO (2.5 ml) | — | In vitro Brine Shrimp lethality assay | — | Pathak et al. (2020) | |||
| Ethanolic extract of aerial parts | 800, 400, 200 and 100 μg/ml | LC50–1,364 μg/ml | Vincristine sulphate (LC50–0.93 μg/ml) | DMSO (1%) | — | In vitro Brine Shrimp lethality assay | — | Mohaimenul et al. (2020) | |||
| Ethanolic, 70% ethanolic, 80% methanolic, ethyl acetate, and aqueous extracts of the whole plant | 5–40 μg/ml | Ethanol and water extracts exhibited potent activity in a concentration-dependent manner | fenofibrate (0.1 mM) | A group without extract/drug | HepG2, a human hepatic cancer cell line | In vitro—preventive and ameliorative effects against palmitate-induced lipid accumulation in HepG2 | Act via preventing steatosis (intracellular lipid content reduced) | Yap et al. (2019) | |||
| Alternanthera sessilis (L.) R.Br. ex DC. | Silver nanoparticles of the aqueous extract of leaves | 25 and 50 μg/ml | IC50 = 42.5 μg/ml | Quercetin | Group without extract/drug | Human breast adenocarcinoma (MCF-7) cell line | In vitro - MTT assay | Act via decreasing expression of MMP- 9 in the cancer cells and inhibit cancer cell migration and reduce the chances of metastasis in human breast cancer | Sathishkumar et al. (2016) | ||
| Ethanolic extract of aerial parts, stem, and leaves | 25–500 μg/ml | 25–500 μg/ml | Paclitaxel (50 ng/ml) | A group without extract/drug | HT-29 and 3T3 human colon cancer cell line | In vitro—MTT assay and colony formation assay | Act via damage of plasma membrane causing necrosis of cancer cell | Arulselvan et al. (2018) | |||
| Aqueous extract of aerial parts | 5 and 50 mg/kg, i.p | 50 mg/kg, i.p | — | Normal saline | Male albino Swiss mice | In vivo—Ehrlich ascites carcinoma model | Act via potentially reducing the number of tumor cells | Guerra et al. (2003) | |||
| 9 | Anticataract | Alternanthera sessilis (L.) R.Br. ex DC. | Ethyl acetate extract of leaves | 100, 200, and 400 mg | 100, 200, and 400 mg | Malondialdehyde and Inorganic Phosphorus | Cataract induced lenses | Lenses tissue | In vitro—lipid peroxidation and Na+ - K+ ATPase assays | Significant increase in the activity of Na+ - K+ ATPase in the lens tissue | Kota et al. (2017) |
| 10 | Anticonvulsant | Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | 250, 500 and 1,000 mg/kg, p.o | Mild activity at a higher dose | Diazepam (1 mg/kg, i.p.) | Pentylenetetrazole (85 mg/kg, i.p.); Strychnine (2 mg/kg, i.p.) | Albino mice | In vivo—Pentylenetetrazole (PTZ)-induced convulsions, Strychnine-induced convulsions, and Maximal electroshock seizures | Act via inhibition of blocking GABA– BZD receptor-mediated neurotransmission, regulation or stimulation of glycine in the spinal cord and blockade the entry of Ca2+, Na+ into the cells | Oyemitan et al. (2015) |
| Phenytoin sodium (25 mg/kg, i.p.) | |||||||||||
| Ethanolic extract of leaves | 20, 100 and 500 mg/kg, i.p | 20 mg/kg, i.p | — | Distilled water (10—ml/kg, p.o.) and PTZ (60 mg/kg, i.p.) | Wistar rats | In vivo—Pentylenetetrazole-induced seizures in rats test | Act via activation of GABA-ergic system | Schallenberger et al. (2017) | |||
| Methanolic extract of leaves | 100, 300 and 600 mg/kg, i.p | 100, 300 and 600 mg/kg, i.p | Diazepam (1 mg/kg, i.p.) | Distilled water (10—ml/kg, p.o.) and PTZ (80 mg/kg, i.p.) | Adult male Swiss albino mice | In vivo—Maximal electroshock-induced seizures and pentylenetetrazole induced seizures | Act via enhancing GABA mediated inhibition in the brain | Barua et al. (2013) | |||
| 11 | Antidepressant | Alternanthera philoxeroides (Mart.) Griseb | Ethanolic extract of leaves | 250 and 500 mg/kg/day | 250 and 500 mg/kg, p.o | 17β-Estradiol (1 μg/kg, i.p.) | Distilled water (0.2—ml/mice, p.o.) | Female ICR mice | In vivo—forced swimming and tail suspension tests | Act via estrogenic activity | Khamphukdee et al. (2018) |
| Alternanthera sessilis (L.) R.Br. ex DC. | Methanolic extract of leaves | 100 and 200 mg/kg, p.o | 100 and 200 mg/kg | Diazepam (2 mg/kg | Distilled water | Adult Swiss albino Wistar mice | In vivo—Tail suspension test and Forced swim test | Act via interaction with adrenergic, dopaminergic serotonergic, and GABAnergic system | Gupta and Singh, (2014) | ||
| 12 | Antidiabetic | Alternanthera brasiliana (L.) Kuntze | 80% Ethanolic extract of stem and leaves | 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Metformin (600 μg/kg, i.p.) | Distilled water (1 ml, p.o.) | Male Swiss albino mice | In vivo—alloxan-induced diabetes model | Significantly decreased the elevated levels of blood glucose, lipid peroxidation, and various free radicals in experimental animals | Reza et al. (2019) |
| Alternanthera philoxeroides (Mart.) Griseb | Methanolic extract of whole plant | 50, 100, 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Glibenclamide (10 mg/kg, p.o.) | 1% Tween-80 in water, 10 ml/kg | Swiss albino mice | In vivo—oral glucose tolerance test | Act via regeneration of β-cells of the pancreas and inhibiting glucose absorption from the gut | Khatun et al. (2012) | ||
| Methanol-soluble fraction | 20, 40 and 60 μg/ml | 60 μg/ml | — | — | In vitro—α-glucosidase inhibitory test | Act via inhibition of α-glucosidase enzyme | Bhattacherjee et al. (2014) | ||||
| Alternanthera pungens Kunth | Aqueous and ethanolic extracts of the whole plant | 200 and 400 mg/kg, p.o | Dose-dependent activity | Metformin (150 mg/kg, p.o.) | Distilled water | Wistar rats | In vivo: Alloxan-induced hyperglycemia | — | Mourya et al. (2020) | ||
| Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous and ethanolic extracts of aerial parts | 125, 250 and 500 mg/kg, p.o | 125, 250 and 500 mg/kg, p.o | Glibenclamide (0.6 mg/kg, p.o.) | 1% Acacia solution, p.o. and alloxan monohydrate (110 mg/kg, p.o.) | Male Wistar albino rats | In vivo—Alloxan induced diabetes model | Act via potentiating the existing β-cells of islets of Langerhan’s in diabetic rats | Kumar et al. (2011b) | ||
| Hexane, ethyl acetate, and aqueous fractions of aerial parts | 500 mg/kg, p.o | 500 mg/kg, p.o. of ethyl acetate fraction | Glibenclamide (10 mg/kg, p.o.) | 1% CMC (2 ml/kg) and streptozotocin monohydrate (40 mg/kg, i.p.) and pioglitazone (30 mg/kg, i.p.) | Male Sprague-Dawley rats | In vivo—Streptozotocin-induced diabetic rat test | Act via improvements in peripheral insulin sensitivity which reduces blood glucose concentration | Tan and Kim, (2013) | |||
| Methanolic extract of aerial parts | 50, 100, 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Glibenclamide ide (10 mg/kg, p.o.) | 1% Tween 80 in water, 10 ml/kg, and Glucose (2 g/kg, p.o.) | Swiss albino mice | In vivo—oral glucose tolerance tests | Act via potentiating pancreatic insulin secretion or by increasing glucose uptake | Hossain et al. (2014) | |||
| Petroleum ether extract of leaves | 25–100 μg/ml | 25 μg/ml | Acarbose (25–100 μg/ml) | — | — | In vitro - α-amylase inhibition assay | — | Sundar et al. (2019) | |||
| 95% Ethanolic extract of the whole plant | 200 and 400 mg/kg, p.o | 200 and 400 mg/kg, p.o | Glibenclamide (10 mg/kg, p.o.) | Saline (2 ml/kg, p.o.) and streptozotocin monohydrate (50 mg/kg, i.p.) | Wistar albino rats | In vivo - Streptozotocin-induced diabetes | Act via protective action on lipid peroxidation, enhancing effects on cellular antioxidant defense and protection against oxidative damage | Das et al. (2015) | |||
| Ethanolic extract of the whole plant | 200 mg/kg, p.o | 200 mg/kg, p.o | Glibenclamide (90 μg/kg, p.o.) | Tween 20 (0.2 ml, p.o.) and streptozotocin monohydrate (50 mg/kg, i.p.) | Male Albino Wistar rats | In vivo - Streptozotocin-induced diabetes | Reduction in blood glucose levels | Rao et al. (2011) | |||
| n-hexane, ethyl acetate, and water fractions of the Methanolic extract of leaves | Up to 20 mg/ml | Ethyl acetate fraction IC50 α-amylase—0.52 mg/ml IC50 α-glucosidase—2.82 mg/ml | Acarbose IC50 α-amylase—0.0025 mg/ml IC50 α-glucosidase—0.36 mg/ml | — | — | In vitro α-amylase and α-glucosidase inhibitory activities | — | Manalo et al. (2020) | |||
| Ethanolic extract of aerial parts | 200 mg/kg, p.o | 200 mg/kg, p.o | Metformin (150 mg/kg, p.o.) | Saline (10 mg/kg, p.o.) | Female Swiss albino mice | In vivo: Alloxan-induced hyperglycemia | — | Mohaimenul et al. (2020) | |||
| Juice | 20 and 100 μl | 100 μl | — | — | Male adult Wistar rats | In vitro—pancreatic α-amylase inhibition assay and rat intestinal α-glucosidase inhibition assay | Act via lysis of cell membrane and inhibiting protein synthesis | Tiwari et al. (2013) | |||
| Hexane, chloroform, ethyl acetate, butanol, and aqueous fractions of methanolic extracts of leaves and callus | — | Leaf ethyl acetate fraction and Callus ethyl acetate fraction exhibited potent anti-glucosidase | Acarbose | — | — | In vitro—α-glucosidase inhibitory test | Act via inhibition of α-glucosidase enzyme | Chai et al. (2016) | |||
| 13 | Antidiarrhoeal | Alternanthera sessilis (L.) R.Br. ex DC. | Hexane, chloroform, methanolic, and aqueous extracts of the whole plant | 50 and 100 mg/kg, p.o. of each extract | Methanol and aqueous extracts exhibit potent activity | Diphenoxylate (2.5 mg/kg) | Polyvinylpyrrolidone, Castor oil (0.1 ml/mice; 1 ml/rats) or MgSO4 (2 g/kg) | Male Wistar rats and CD1 strain male mice | In vivo—Diarrhoea induced by castor oil and MgSO4 | Inhibition of water and electrolyte transport through the intestinal mucosa or enhancing peristalsis in the intestine | Zavala et al. (1998) |
| 14 | Antigout | Alternanthera sessilis (L.) R.Br. ex DC. | Methanolic extract of aerial parts | 100–1,000 μg/ml | IC50–557.77 μg/ml | Allopurinol (IC50–6.1 μg/ml) | DMSO | — | In vitro: Xanthine oxidase inhibitory assay | Xanthine oxidase inhibition | Chong and Loh, (2020) |
| 15 | Anti-HBV | Alternanthera philoxeroides (Mart.) Griseb | luteolin-6-C-β-D-boivinopyranosyl-3′-O- β-D-glucopyranoside; chrysoeriol-6-C- β D-boivinopyranosyl-4′-O- β D-glucopyranoside; luteolin-6-C-β-D-boivinopyranosyl-4′-O- β-D-glucopyranoside; luteolin-6-C-β-D-boivinopyranoside and chrysoeriol-6-C- β-D-boivinopyranoside | — | — | — | DMEM with 0.2% DMSO | HepG2.2.15 cells | In vitro—Inhibition of HBsAg and HBeAg secretions HepG and MTT assay | Act via inhibiting the secretion of HBsAg in HepG2.2.15 | Li et al. (2016) |
| 16 | Antihypertensive | Alternanthera sessilis (L.) R.Br. ex DC. | 70% Ethanolic extract of the whole plant and its dichloromethane and aqueous fractions | 1–10 mg/kg, i.p | Ethanol extract: 1–10 mg/kg, i.p | Verapamil (1–10 m g/kg, i.p.) | Adrenaline (1 μg/kg) | Sprague-Dawley albino rats | In vivo—ketamine (50–80 mg/kg, i.p.) –diazepam (5 mg/kg, i.p.) anaesthetized normotensive rats | Decreased both systolic and diastolic blood pressure of the anesthetized rats | Saqib and Janbaz, (2016) |
| 17 | Anti-inflammatory | Alternanthera brasiliana (L.) Kuntze | Aqueous extract of leaves | 200 or 400 mg/kg, p.o | 400 mg/kg, p.o | Indomethacin (10 mg/kg, p.o.) | Distilled water (10 ml/kg, p.o.) | Male adult Wistar rats | In vivo—carrageenan-induced pleurisy | Reduction of polymorphonuclear cells and the increase of mononuclear cells in the exudate of animals | Pelisoli Formagio et al. (2012) |
| Methanolic extract of leaves | 300, 600 and 900 mg/kg, p.o | 600 mg/kg, p.o | Sulfasalazine (360 mg/kg, p.o.) | Normal saline and 4% acetic acid (1 ml, t.r.) | Adult Wistar albino rats | In vivo—acetic acid-induced colitis model of inflammatory bowel disease | Significantly reduced colon weight and decreased macroscopic and microscopic score | P et al. (2016) | |||
| Alternanthera littoralis P.Beauv | Ethanolic extract of aerial parts 2″-O-α-L-rhamnopyranosylvitexin | 30, 100, 300 mg/kg, p.o. 1, 10, 20 mg/kg, p.o | 100, 300 mg/kg, p.o. 1, 10, 20 mg/kg, p.o | Dexamethasone (1 mg/kg, s.c.) | 0.9% saline solution | Adult male and female Swiss mice | In vivo—carrageenan-induced paw edema and carrageenan-induced pleurisy method | Act via inhibiting TrpV1, oxidative stress, cytokines | de Santana Aquino et al. (2015) | ||
| Alternanthera philoxeroides (Mart.) Griseb., Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of leaves | 100–500 μg/ml | 500 μg/ml | Diclofenac sodium (100–500 μg/ml) | — | — | In vitro—% Membrane stabilization and % Haemolysis | Act via by inhibiting hypotonicity induced lysis of erythrocyte membrane and inhibition of the release of phospholipases | Sunmathi et al. (2016) | ||
| Alternanthera pungens Kunth | Aqueous extract of leaves | 200 mg kg, i.p | 200 mg kg, i.p | Indomethacin (10 mg/kg, i.p.) | 1% Carrageenan (0.1 ml, i.p.) | Wistar strain rats | In vivo—carrageenan-induced inflammatory test | Decreased level of release of histamine serotonin and kinin, prostaglandin, proteases, lysosomes, and protein C-reactive | Franck et al. (2016) | ||
| Alternanthera sessilis (L.) R.Br. ex DC. | 90% ethanolic extract of stems | 25, 50, 100, 200, 300, 400 and 500 μg/ml | 200 or 500 μg/ml | Dexamethasone (0.5 μg/ml) | Untreated cells | RAW 264.7 murine macrophage cell line | In vitro—cell viability assay, quantifying the nitric oxide, proinflammatory cytokine production, nuclear translocation of NF-κB p65, and protein expression analysis | Reduced the level of proinflammatory cytokines and mediators in LPS- stimulated RAW 264.7 macrophages by inactivating their corresponding genes at the transcriptional level and by preventing the activation of the NF- κB pathway | Muniandy et al. (2018a) | ||
| Petroleum ether and methanolic extracts of leaves | 100, 200 and 300 μg/ml | Methanol extract (100 μg/ml) | Aspirin (100, 200 and 300 μg/ml) | Group without extract/drug | — | In vitro—protein denaturation method | — | Sundar et al. (2019) | |||
| Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of whole plant (EEAT) 2″-O-β-D-glucopyranosyl-vitexin | 30, 100 and 300 mg/kg, p.o. 0.1, 1 and 10 mg/kg, p.o | 100 mg/kg, p.o. 1 mg/kg, p.o | Prednisolone (3 mg/kg, p.o.) | Swiss mice | In vivo: Carrageenan-induced paw edema, zymosan-articular inflammation, carrageenan pleurisy, and complete Freund’s adjuvant | Significantly inhibited (i) edema, mechanical hyperalgesia in carrageenan-induced paw inflammation; (ii) leukocyte migration and protein extravasation in carrageenan-induced pleurisy; (iii) knee edema, mechanical hyperalgesia, and leukocyte migration in articular inflammation induced by zymosan | Kassuya et al. (2021) | |||
| Aqueous extract of the whole plant | 200 and 400 mg/kg, i.p | 200 and 400 mg/kg, i.p | Indomethacin (5 mg/kg, i.p.) | Sterile saline (0.2 ml, i.p.) | Male BALB/c mice | In vivo—Carrageenan-induced edema method | Cyclooxygenase -1 and -2 inhibition | Biella et al. (2008) | |||
| 18 | Antimicrobial | Alternanthera bettzickiana (Regel) G.Nicholson | Hexane, chloroform, ethyl acetate, methanolic, and aqueous extracts of leaves | 125, 250, 500 and 1,000 μg/ml | Mild activity | Cotrimoxazole (23.75µg/disc), Ciproflaxocin (5µg/disc), Chloramphenicol (30µg/disc) and Piperacillin (100µg/disc) | Sterile distilled water | Various bacterial strains | In vitro - Kirby-Bauer disc diffusion method | Act via lysis of bacterial cell wall and inhibiting protein synthesis | Vidhya et al. (2015) |
| Aqueous extract of leaves and silver nanoparticles and Ag-mesoporous MnO2 nanocomposite | 5–100 μg/ml | 100 μg/ml of Silver nanoparticles and Ag-mesoporous MnO2 nanocomposite | — | DMSO | Various bacterial strains | In vitro—Agar well diffusion assay | Act via inhibition of DNA replication and blocking cellular respiration | Jothi Ramalingam et al. (2017) | |||
| Aqueous extract of leaves (Au-NP) | 10, 20, 30 or 40 μl | 10, 20, 30 or 40 μl | Ciprofloxacin | — | Various bacterial strains | In vitro - Agar well diffusion method | Act via inhibiting DNA gyrase, topoisomerase II, topoisomerase IV | Nagalingam et al. (2018) | |||
| Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | MIC = ≥1,024 μg/ml | MIC = ≥1,024 μg/ml | Gentamicin (1,024 μg/ml) | — | Various bacterial strains | In vitro - disk diffusion method | Act via methylation of the aminoglycoside-binding site and targeted mutations in the 30S ribosomal subunit | Coutinho et al. (2017) | ||
| Ethanolic extract of aerial parts | 7.8–1,000 μg/ml | Inactive | Amphotericin-B | DMSO | Various murine macrophages and fungal strains | In vitro - broth microdilution method | — | Johann et al. (2010) | |||
| Alternanthera caracasana Kunth | Hexane, chloroform, methanolic, acetone, and ethyl acetate extract of aerial parts and 7-methoxycoumarin | — | Acetone and ethyl acetate extracts and 7-methoxycoumarin | Kanamycin and chloramphenicol (25 μg) | DMSO | Various bacterial strains | In vitro - disk diffusion method | Act via lysis of microbial cell wall and inhibiting protein synthesis | Canales-Martínez et al. (2008) | ||
| Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | 250 mg | Mild activity | Ciprofloxacin | — | Various bacterial strains | In vitro - disk diffusion method | Act via lysis of microbial cell wall and inhibit protein synthesis | Akachukwu and Uchegbu, (2016) | ||
| Silver nanoparticles from aqueous extract of leaves | 20–100 μg/ml | 20–100 μg/ml | — | DMSO | Various bacterial strains | In vitro—Agar well diffusion assay | Act via inhibition of DNA replication and blocking cellular respiration | Kumar et al. (2014) | |||
| Alternanthera littoralis P.Beauv | Hexane and ethanolic extract of leaves | 25 mg/ml (Final reactive concentration: 2,625 µg/105 µl) | 25 mg/ml(Final reactive concentration: 2,625 µg/105 µl) | ketoconazole (0.20 mg/ml) and methylene blue (0.05 mg/ml) | Propylene glycol/distilled sterilized water (5:95) | Various fungal strains | In vitro - agar-well diffusion method | Microbial membrane lysis and protein degradation | Gasparetto et al. (2010) | ||
| Alternanthera nodiflora R.Br | Aqueous and methanolic extracts of the whole plant | 25–100 mg/ml | Methanol extract (100 mg/ml) | — | — | Various bacterial and fungal strains | In vitro - agar well diffusion method | Act via lysis of microbial cell wall and inhibiting protein synthesis | Feka et al. (2014) | ||
| Alternanthera philoxeroides (Mart.) Griseb | Methanol-soluble fraction of leaves | 20, 40 and 60 μg/ml | 60 μg/ml | — | — | Various bacterial strains | In vitro - disc diffusion assay | Act via lysis of bacterial cell wall and inhibit protein synthesis | Bhattacherjee et al. (2014) | ||
| Aqueous and chloroform: methanol (1:1) extracts of leaves | 35.25–80 μg/ml | 35.25–80 μg/ml | — | Distilled water and DMSO | Various bacterial strains | In vitro—disc diffusion method | — | Rawani et al. (2011) | |||
| Ethanolic extract of leaves | 500, 750 and 1,000 μg/ml | 1,000 μg/ml | Tetracycline (30 μg/ml) for bacteria and fluconazole (100 μg/ml) for fungi | DMSO | Various bacterial and fungal strains | In vitro—Agar well diffusion assay | Act via lysis of microbial cell wall and inhibit protein synthesis | Pulipati et al. (2016) | |||
| Methanolic extract of leaves, stem and roots n-hexane, chloroform and ethyl acetate fractions | 100 mg/ml | 100 mg/ml | Penicillin (100 mg/ml) | DMSO (166 µl) | Bacterial phytopathogens (Erwinia carotovora, Ralstonia solanacearum, and Xanthomonas axonopodis) | In vitro; Disk diffusion method n-hexane fraction maximum zone of inhibition | — | Akbar et al. (2021) | |||
| Methanolic extract of leaves | 500, 750 and 1,000 μg/ml | 1,000 μg/ml | Nitrofurantoin (300 µg/disc) | DMSO | Multidrug-resistant uropathogens (Staphylococcus aureus, Staphylococcus saprophyticus, Enterococcus faecalis, Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus vulgaris, and Proteus mirabilis) | In vitro; Agar well diffusion method Inhibition rate observed in the following order: S. saprophyticus > S. aureus > K. pneumoniae E. coli, P. vulgaris > E. faecalis, P. aeruginosa > P. Mirabilis | — | Pulipati and Babu, (2020) | |||
| Alternanthera philoxeroides (Mart.) Griseb. and Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous extract of leaves | — | Both plants exhibited antibacterial only | — | — | Various bacterial and fungal strains | In vitro - agar well diffusion method | Act via lysis of microbial cell wall and inhibit protein synthesis | Kumari and Krishnan, (2016) | ||
| Ethanolic extract of leaves | 10, 25, and 50 μg | 10, 25, and 50 μg | Gentamycin/Nystatin | Ethanol | Various bacterial and fungal strains | In vitro - Well diffusion assay | — | Sivakumar and Sunmathi, (2016) | |||
| Alternanthera pungens Kunth | Aqueous, acetone, ethanolic, and petroleum ether extracts of aerial parts | 25–200 mg/ml | All extracts exhibited antibacterial potential but the antifungal profile was shown by only acetone and aqueous extracts | Ampicillin (100 μg/ml) and Miconazole (100 μg/ml) | DMSO | Various bacterial and fungal strains | In vitro—Agar well diffusion assay | Act via inhibition of DNA replication and blocking cellular respiration | Jakhar and Dahiya, (2017) | ||
| Alternanthera sessilis (L.) R.Br. ex DC. | Hexane and methanolic extracts of aerial parts | 2–16 mg/ml | Mild action | Cefotaxime (2–16 μg/ml) | — | Various bacterial strains | In vitro—agar dilution method | Act via lysis of microbial cell wall and inhibiting protein synthesis | Osuna et al. (2008) | ||
| Petroleum ether (40–60°C), chloroform, acetone, methanolic, and aqueous extracts of leaves | 5–75 µg | Chloroform extract exhibited a potent antibacterial profile | Ciprofloxacin (5–75 µg) and fluconazole (5–75 µg) | — | Various bacterial and fungal strains | In vitro - cup plate and turbidimetric methods | — | Jalalpure et al. (2008) | |||
| Aqueous, ethanolic, and acetone extracts of leaves | 1,000 μg/ml | 25.7 and 252.5 μg/ml | Tetracycline and ketoconazole | — | Various bacterial and fungal strains | In vitro - Kirby-Bauer method | Act via lysis of microbial cell wall and inhibit protein synthesis | Monroy and Limsiaco, (2016) | |||
| Silver nanoparticles of aqueous extract of leaves | 100 μg/ml | 100 μg/ml | --------- | — | Various bacterial strains | In vitro - Well diffusion assay | --------- | Niraimathi et al. (2013) | |||
| Ethanolic extract of leaves | 25, 100, 250 and 500 μg/ml | 500 μg/ml | — | — | Various bacterial strains | In vitro - agar-well diffusion method | Act via inhibition of extracellular microbial enzymes, proteins, deprivation of iron as substances for microbial growth or destroy its membranes | Rajamurugan et al. (2013) | |||
| Aqueous extract of leaves and stems | 250, 500 and 1,000 μg/μl | 250, 500 and 1,000 μg/μl | — | — | Various bacterial strains | In vitro - agar well diffusion method | Act via lysis of microbial cell wall and inhibit protein synthesis | Suganya et al. (2019) | |||
| Petroleum ether, ethyl acetate, chloroform, and methanolic extract of leaves | 50 mg/ml | Ethyl acetate and methanol extract exhibited maximum activity | — | DMSO | Various bacterial strains | In vitro—Agar well diffusion assay | Act via inhibition of DNA replication and blocking cellular respiration | Kota et al. (2017) | |||
| Petroleum ether and methanolic extracts of leaves | 25, 50, and 100 µg | 100 µg | Streptomycin (10 µg) | DMSO | Various bacterial strains | In vitro—Agar well diffusion assay | — | Sundar et al. (2019) | |||
| Petroleum ether and methanolic extracts of leaves | 10 mg | 10 mg | Fluconazole (10 mg) | DMSO | Various bacterial and fungal strains | In vitro—Agar well diffusion assay | — | Sundar et al. (2019) | |||
| Hexane and ethanolic extracts of the adult plants | MIC = 50–500 μg/ml | MIC = 50–500 μg/ml | Fluconazole (10 mg) | DMSO | Various bacterial and fungal strains | In vitro—Agar well diffusion assay | Act via destroying the cell membrane and prevent the protein synthesis | Salvador et al. (2009) | |||
| 19 | Antioxidant | Alternanthera bettzickiana (Regel) G.Nicholson | Four fractions of 80% aqueous methanolic extract of flowers | 200 mg/l | 8.2–67.2% Scavenging of ABTS radical; 6.9–63.8% scavenging as per FRAP assay | Rutin (10 mg/l) | Solution of stable free radicals | — | In vitro - ABTS, FRAP, and metal ion chelation assay | Inhibition of free radicals | Petrus et al. (2014b) |
| Hexane, chloroform, ethyl acetate, methanolic, and aqueous extracts of leaves | 125, 250, 500, and 1,000 μg/ml | Methanol extract exhibited strong activity IC50 = 293.44 μg/ml | — | Solution of stable free radicals | — | In vitro - DPPH radical scavenging, reducing power and total antioxidant (Ammonium molybdate) activities | Inhibition of free radicals | Vidhya et al. (2015) | |||
| Alternanthera brasiliana (L.) Kuntze | 80% Ethanolic extract of stem and leaves | 1–1,000 μg/ml | IC50 = 52.02–140.05 μg/ml | Ascorbic acid | Solution of stable free radicals | — | In vitro - DPPH radical scavenging, reducing power, nitric oxide (NO) radical inhibition, and scavenging of hydrogen peroxide assay | Act via inhibition of free radicals | Reza et al. (2019) | ||
| Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | 0.1–1,000 μg/ml | — | Ascorbic acid | — | — | In vitro—1,1-diphenyl- 2-picrylhydrazyl (DPPH) radical-scavenging, iron (II)-chelating, nitric oxide radical-scavenging, ferrous sulfate, and carbon tetrachloride-induced lipid peroxidation assays | Inhibition/inactive free radicals | Enechi et al. (2013) | ||
| Methanolic extract of leaves | 50–1,000 μg/ml | 50–1,000 μg/ml | Butylated hydroxyanisole | DPPH stable free radicals | — | In vitro—DPPH assay | Inhibition of stable DPPH free radicals | Chandran, (2017) | |||
| Ethanolic extract of leaves | 0–1 mg/ml | Concentration-dependent activity | Vitamin C | Solution of stable free radicals | — | In vitro—DPPH assay, Ferric oxide reducing power assay, and Nitric oxide scavenging assay | Inhibition of stable free radicals | Attaugwu and Uvere, (2017) | |||
| Ethanolic extract and its dichloromethane, ethyl acetate, n-butanolic fractions of leaves | Ethyl acetate fraction exhibited strong activity (IC50 = 163 mg/ml) | Ethyl acetate fraction exhibited strong activity (IC50 = 163 mg/ml) | Ascorbic acid (IC50 = 6.48 μg/ml) | Solution of stable free radicals | — | In vitro—DPPH assay | Inhibition of free radicals | Pereira et al. (2013) | |||
| Ethanolic extract of aerial parts and its hexane, chloroform, and ethyl acetate fractions | --------- | Ethanol extract and its ethyl acetate fraction exhibited maximum activity | Ascorbic acid, butylated hydroxyanisole, and butylated hydroxytoluene | Solution of stable free radicals | — | In vitro –DPPH and β-carotene assay | Act via inhibition of stable free radicals | Araujo et al. (2014) | |||
| Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | 25–400 μg/ml | 400 mg/ml | Ascorbic acid (25–400 μg/ml) | Solution of stable free radicals | — | In vitro - 2, 2-diphenyl-1-picrylhydrazyl (DPPH) and ferric reducing antioxidant power assay | Inhibition of stable free radicals | Akachukwu and Uchegbu, (2016) | ||
| Alternanthera ficoidea (L.) P.Beauv | Methanolic extract of leaves, stem, and roots | IC50 = 442.5, 423.75 and 390.66 μg/ml, respectively, for leaves, stems and roots | IC50 = 442.5, 423.75 and 390.66 μg/ml, respectively, for leaves, stems and roots | Ascorbic acid | Solution of stable free radicals | — | In vitro—DPPH assay | Inhibition of stable free radicals | Patil and Kore, (2019) | ||
| Alternanthera littoralis P.Beauv | Alternamide A-B, Alternamine A-B | — | Alternamide B (1.10 relative Trolox equivalent) | Quercetin and caffeic acid | Solution of stable free radicals | — | In vitro—ORAC assay | Inhibition of stable free radicals | Koolen et al. (2017) | ||
| Alternanthera paronychioides A.St.-Hil | Methanolic, ethanolic, and aqueous extracts of the whole plant | Ethanolic extract | — | — | Solution of stable free radicals | — | In vitro - Trolox equivalent antioxidant capacity, oxygen radical absorbance capacity, and cellular antioxidant activity | Inhibition of stable free radicals | Wu et al. (2013) | ||
| Aqueous extract of leaves | 100 μg/ml | Mild activity | Trolox (0–80 nmol/μl) | — | — | In vitro—1,1-diphenyl- 2-picrylhydrazyl (DPPH) | Inhibition/inactive free radicals | Tukun et al. (2014) | |||
| Alternanthera philoxeroides (Mart.) Griseb | Methanol-soluble fraction from leaves | 20, 40 and 60 μg/ml | 60 μg/ml | — | — | — | In vitro - DPPH and ABTS radical scavenging assay | Inhibition of stable free radicals | Bhattacherjee et al. (2014) | ||
| Alternanthera philoxeroides (Mart.) Griseb.; Alternanthera hirtula (Mart.) R.E.Fr. and Alternanthera praelonga A.St.-Hil | Ethanolic extracts of the whole plant | --------- | Exhibited mild activity | Quercetin, vitexin, caffeic acid, chlorogenic acid, and Trolox | Solution of stable free radicals | — | In vitro –DPPH assay | Inhibition of stable free radicals | Correa et al. (2016) | ||
| Alternanthera pungens Kunth | Ethanolic and aqueous extracts of leaves | 20–100 mg/ml | 100 mg/ml | Butylated hydroxytoluene and ascorbic acid | Azinobis-3-ethybenzothiazoline-6-sulfonic acid radical and DPPH radical | — | In vitro—2, 2-azinobis-3-ethylbenzothiazoline-6-sulfonic acid radical scavenging assay and DPPH radical scavenging assay | Act via proton- donating ability and could serve as free radical inhibitors | Mourya et al. (2019) | ||
| Aqueous extract of leaves | 200 mg kg, i.p | 200 mg kg, i.p | Vitamin C (100 mg/kg, i.p.) | 1% Carrageenan (0.1 ml, i.p.) | Wistar strain rats | In vivo—estimation of thiobarbiturates Acid Reactive Substances assay | Act via significant reduction of serum concentration levels of TBARS | Franck et al. (2016) | |||
| Aqueous, acetone, ethanolic, and petroleum ether extracts of aerial parts | 100–1,000 μg/ml | IC50 = 324.43, 203.56, 100.79 and 931.63 μg/ml, respectively for extracts | Ascorbic acid (100–1,000 μg/ml) | Solution of stable free radicals | — | In vitro—DPPH assay | Act via inhibition of stable free radicals | Jakhar and Dahiya, (2017) | |||
| Alternanthera sessilis (L.) R.Br. ex DC. | 90% Methanolic, 70% acetone, 80% ethanolic extracts of leaves and stems | 100–1,000 μg/ml | All extracts are active | Ascorbic acid and rutin | — | — | In vitro - phosphomolybdate, DPPH scavenging, superoxide scavenging, nitric oxide scavenging, and iron-chelating methods | Act via inhibition of various oxidative stress-producing species | Borah et al. (2011) | ||
| Hexane, chloroform, ethyl acetate, butanolic, and aqueous fractions of leaves and callus methanol extracts | 100 μg/ml | Ethyl acetate fraction of leaves exhibited potent antioxidant activity | Quercetin | — | — | In vitro - 1,1-diphenyl-2-picrylhydrazyl (DPPH) scavenging assay | Inhibition of free radicals | Chai et al. (2016) | |||
| 30% Hydroethanolic extract of the whole plant | 100 μg/ml | — | Mannitol, ascorbic acid, quercetin and sodium pyruvate | Solution of respective free radicals | — | In vitro - scavenging of hydroxyl radicals, superoxide radical scavenging, hydrogen peroxide radical scavenging, and metal chelating tests | Inhibition of free radicals | Sharma et al. (2013) | |||
| Separate Methanolic and hexane extracts of leaves and stems | 0.05–0.20 mg/ml | Methanolic extract of leaves | Butylated hydroxytoluene | Solution of stable DPPH free radicals | — | In vitro - DPPH radical scavenging activity | Inhibition of DPPH free radicals | Khan et al. (2018) | |||
| Ethanolic and aqueous extracts of aerial parts | — | The ethanolic extract exhibited strong activity | — | Solution of stable free radicals | — | In vitro - β-carotene bleaching, DPPH, ABTS, ORAC, and FRAP assay | Inhibition of free radicals | Azizah et al. (2015) | |||
| 90% hydroethanolic extract of stem | 100–1,000 μg/ml | 100–1,000 μg/ml | Gallic acid | DPPH stable free radicals | — | In vitro - DPPH assay | Inhibition of stable DPPH free radicals | Muniandy et al. (2018b) | |||
| Ethanolic and aqueous extracts of aerial parts | 100–1,000 μg/ml | — | β-carotene, ascorbic acid, Trolox, iron (II) sulfate heptahydrate | Solution of stable free radicals | — | In vitro - β-carotene bleaching assay, 2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay, 2,2′-azinobis-3-ethylbenzothiazoline-6-sulphonic acid (ABTS) assay, Oxygen radical absorbance capacity (ORAC) assay, and Ferric reducing antioxidant power (FRAP) assay | Inhibition of stable free radicals | Othman et al. (2016) | |||
| Juice | 25, 40, and 100 μl | 25, 40, and 100 μl | Ascorbic acid | Solution of stable free radicals | — | In vitro - 2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay, 2,2′-azinobis-3-ethylbenzothiazoline-6-sulphonic acid (ABTS) assay, hydrogen peroxide (HO) scavenging assay, ferric chloride reducing assay | Inhibition of stable free radicals | Tiwari et al. (2013) | |||
| Ethanolic extract of leaves | 10, 50, 100, 250 and 500 μg/ml | IC50 = 364, 522 μg/ml | Ascorbic acid | Solution of stable free radicals | — | In vitro - DPPH radical scavenging assay, ABTS radical cation-scavenging assay, and Reducing power assay | Inhibition of stable free radicals | Rajamurugan et al. (2013) | |||
| Methanolic extract of leaves | 100–1,200 μg/ml | IC50 = 400 μg/ml | Ascorbic acid | Solution of stable free radicals | — | In vitro - DPPH radical scavenging method | Inhibition of stable free radicals | Jain et al. (2016) | |||
| Aqueous extract of leaves and stems | 10–100 μg/ml | 10–100 μg/ml | Quercetin (10–100 μg/ml) | Solution of stable free radicals | — | In vitro - DPPH radical scavenging and Ferric reducing antioxidant power assay | Inhibition of stable free radicals | Suganya et al. (2019) | |||
| Hexane, ethyl acetate, ethanolic, and aqueous extracts of leaves and stem | 0–1,000 μg/ml | The ethanolic extract exhibited potent activity | Ascorbic acid, gallic acid, rutin, and butylated hydroxytoluene | Solution of stable free radicals | — | In vitro—DPPH test, Trolox equivalent antioxidant capacity, and ferric reducing antioxidant power assay | Inhibition of stable free radicals | Mohd Hazli et al. (2019) | |||
| Silver nanoparticles from aqueous extract of leaves | 100–500 μg/ml | IC50 = 300.6 μg/ml | Gallic acid | Solution of stable free radicals | — | In vitro—DPPH test | Inhibition of stable free radicals | Niraimathi et al. (2013) | |||
| 100% Ethanolic, 70% ethanolic, 80% methanolic, ethyl acetate, and aqueous extracts of the whole plant | 0–1,000 μg/ml | Ethanolic extracts exhibited maximum activity (DPPH IC50: 82.6 μg/ml; TEAC: 0.51 mmol TE/g; FRAP: 1.95 mmol Fe2+/g) | Ascorbic acid, gallic acid, rutin, and butylated hydroxytoluene | Solution of stable free radicals | — | In vitro—DPPH test, Trolox equivalent antioxidant capacity, and ferric reducing antioxidant power assay | Inhibition of stable free radicals | Yap et al. (2019) | |||
| Petroleum ether, ethyl acetate, chloroform, and methanolic extract of leaves | 100–600 μg/ml | 100–600 μg/ml | Ascorbic acid (100–600 μg/ml) | Solution of stable free radicals | — | In vitro—Reducing power and DPPH assay | Inhibition of stable free radicals | Kota et al. (2017) | |||
| Petroleum ether and methanolic extracts of leaves | 50, 100 and 150 μg/ml | 50, 100 and 150 μg/ml | Ascorbic acid (50–150 μg/ml) | Solution of stable free radicals | — | In vitro—Reducing power and DPPH assay | Inhibition of stable free radicals | Sundar et al. (2019) | |||
| n-hexane and methanolic extracts of aerial parts | 10, 30, 50, 70, 90 and 110 μg/ml | Methanol extract (IC50–71.10 μg/ml) n-hexane extract (IC50–92.54 μg/ml) | Ascorbic acid (IC50–39.53 μg/ml) | — | — | In vitro DPPH assay | — | Pathak et al. (2020) | |||
| The volatile oil of leaves and flowers | 50–250 μg/ml | Flower (IC50 = 170 μg/ml) and leaves (IC50 = 179 μg/ml) | Butylated hydroxytoluene (IC50 = 88 μg/ml) | Solution of stable free radicals | — | In vitro—DPPH assay | Inhibition of stable free radicals | Khan et al. (2016) | |||
| Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract and its four fractions; Acacetin 8-c-[α-L-rhamnopyranoyl-(1→2)-β-D-glucopyranoside]; 2″-O-α-L-rhamnopyranosyl-vitexin; 2″-O-β-D-glucopyranosyl vitexin and Vitexin | — | Extract, fractions, and isolates exhibited significant activity | Quercetin, isoquercitrin, caffeic acid and chlorogenic acid | Solution of stable free radicals | — | In vitro—ORAC assay | Inhibition of stable free radicals | Salvador et al. (2006) | ||
| 20 | Antiparkinsonism/Antidementia | Alternanthera philoxeroides (Mart.) Griseb | Ethanolic extract of the whole plant | In vivo study: 250 and 500 mg/kg, p.o. once daily for 8 weeks | Dose dependently improved cognitive deficits-like behavior of the estrogen-deprived mice | 17β-estradiol 1 μg/kg, p.o. once daily for 8 weeks | Distilled water | OVX Female ICR mice | In vivo: Morris water maze task, novel object recognition task, and Y-maze task | Inhibition of lipid peroxidation in the whole brain, downregulation of neuroinflammatory cytokines (IL-1β, IL-6, and TNF-α) and upregulation of estrogen receptor-mediated facilitation genes (PI3K and AKT) in both frontal cortex and hippocampus | Khamphukdee et al. (2021) |
| In vitro study: 100 μg/ml | In vitro: Amyloid aggregation inhibition and cholinesterase inhibitory activity | ||||||||||
| Alternanthera sessilis (L.) R.Br. ex DC. | Silver nanoparticles and ethanolic extract of the whole plant | 20 and 200 mg/kg, p.o | 20 and 200 mg/kg, p.o | Syndopa (10 mg/kg, p.o.) | Distilled water and Rotenone (1.5 mg/kg, s.c.) | Male Wistar rats | In vivo—rotenone model of parkinsonism | Act via the reduction in the lipid peroxidation, increase in reduced glutathione, and reduction in oxidative stress in the brain of animals | Ittiyavirah and Hameed, (2015) | ||
| 21 | Antiprotozoal | Alternanthera littoralis P.Beauv | Alternamide A-B, Alternamine A-B | — | Alternamine A (IC50 = 0.16 μM) and Alternamine B (IC50 = 0.82 μM) | Amphotericin B and crystal violet | DMSO | Various protozoal strains | In vitro—Trypanocidal and leishmanicidal assays | — | Koolen et al. (2017) |
| 22 | Antispasmodic | Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous, hexane, methanolic extract, and fractions of methanol extract (F1-F6) of leaves | — | Methanolic extract and fractions of the methanolic extract (F2-F4) | — | — | Adult male Wistar rats | In vivo—Smooth muscle preparation, Inhibition of dose-response curves to CaCl2, Relaxant effect on K+-induced contractions, Inhibition of dose-response curves to 5-HT, and inhibition of concentration-response curve to acetylcholine (ACh) | Act via inhibition of serotonergic and Ca2+ influx blockade, the peristaltic movement of the rat ileum, and reduction of the intestinal transit of food in rats | Garín-Aguilar et al. (2013) |
| Alternanthera sessilis (L.) R.Br. ex DC. | 70% Ethanolic extract of the whole plant and its dichloromethane, aqueous fractions | — | Ethanolic extract: (0.01–1.0 mg/ml), aqueous fraction (0.01–0.3 mg/ml) and dichloromethane (0.01–0.1 mg/ml) | Verapamil (1–10 mg/kg, i.p.) | — | White albino rabbits | In vitro—isolated rabbit tissue preparations (i.e., jejunum, trachea, and aorta) | Decreased the contractions in terms of both frequency and magnitude | Saqib and Janbaz, (2016) | ||
| 23 | Antiviral | Alternanthera philoxeroides (Mart.) Griseb | Chikusetsusaponin IV a | — | IC50 = 29, 30, 73, 25, and 25 | — | No drug group | HSV-1, HSV-2, human cytomegalovirus, measles virus, mumps virus, and Female BALB/c mice | In vitro—various viral cell | Suppressed both the intracellular virus levels and the release of the virus in a concentration-dependent manner and prevent the viral protein synthesis | Rattanathongkom et al. (2009) |
| In vivo—mouse model of genital herpes caused by HSV-2 | |||||||||||
| 24 | Central-stimulating | Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of leaves | 250 and 500 mg/kg, p.o | 500 mg/kg, p.o | Caffeine (20 mg/kg, i.p.) | Pentobarbitone (50 mg/kg, i.p.) | Young Swiss Albino mice | In vivo—Pentobarbitone induced sleeping time, open field and hole cross tests | Act via stimulating the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) mediate d postsynaptic inhibition through allosteric modification of GABA-A receptors | Mondal et al. (2014) |
| 25 | Gastrointestinal protective | Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous and ethanolic extract of the whole plant | 1–300 mg/kg, p.o | 1–300 mg/kg, p.o | Atropine (1 mg/kg) | — | Swiss mice | In vitro—charcoal meal method | Act via decreasing gastrointestinal content | Astudillo-Vázquez et al. (2008) |
| 26 | Hepatoprotective | Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous extract of entire plant | 300 mg/kg, p.o | 300 mg/kg, p.o | — | Corn oil/0.9% physiological saline/polyethylene glycol 400 Carbon tetrachloride (31.25 μL/kg, i.p.) or acetaminophen/paracetamol (600 mg/kg, i.p.) in mice and D(+)-galactosamine (188 mg/kg, i.p.) in rats | Male ICR strain mice and male Wistar strain albino rats | In vivo—Carbon tetrachloride-induced hepatotoxicity | Act via inhibition of cytochrome P450, or promotion of its glucuronidation | Lin et al. (1994) |
| Methanolic extract of the whole plant | 50, 200 and 250 mg/kg, p.o | 200 and 250 mg/kg, p.o | Silymarin (100 mg/kg, p.o.) | 2% w/v Gum acacia suspension (1 ml/kg, p.o.) and carbon tetra chloride (1.25 ml/kg, i.p.) | Male Wistar rats | In vivo—carbon tetrachloride-induced hepatotoxicity | Act via significant reversal of degeneration marked by a prominent decrease of necrosis, cell integrity restoration | Bhuyan et al. (2017) | |||
| 27 | Immunomodulatory | Alternanthera sessilis (L.) R.Br. ex DC. | Aqueous extract of the whole plant | 50, 100 and 200 mg/kg, i.p | 50, 100 and 200 mg/kg, i.p | Sheep red blood cells (0.1 mL, 25% suspension in saline, i.p.) | Sterile saline (0.2 ml, i.p.) | Male BALB/c mice, adult guinea pigs, and adult sheep | In vivo—Enzyme-linked immunosorbent assay | Act via increasing production of mitogen-induced antibodies and inhibiting the production of antibodies to T-dependent antigens | Biella et al. (2008) |
| Aqueous extract of aerial parts | 5 and 50 mg/kg, i.p | 50 mg/kg, i.p | — | Normal saline | Male albino Swiss mice | In vivo—mice immunized with sheep red blood cells (SRBC 10%, i.p.) as T-dependent antigen, or in mice stimulated with mitogens (10 μg, Escherichia coli lipopolysaccharide, LPS, i.p.) | Act via immune activation either by inhibiting or stimulating antibody production, depending on its concentration | Guerra et al. (2003) | |||
| Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera brasiliana (L.) Kuntze and Alternanthera littoralis P.Beauv | Aqueous and ethanolic extract of leaves; tetrahydrofuran, dichloromethane, aqueous, petroleum ether soluble fraction | 0–200 μg/ml | 0–200 μg/ml | — | — | Peripheral blood mononuclear cells | In vitro—Natural Killer Assay | Inhibition of lymphocyte activation | Moraes et al. (1994) | ||
| Act via activation of the cells of the immune system | |||||||||||
| 28 | Insecticide | Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | 10, 20 and 40 μg/ml | 10, 20 and 40 μg/ml | — | 1% sucrose | Adult flies (Drosophila melanogaster) | In vivo—Toxicity against Drosophila melanogaster and locomotor assays | Act via inhibition of nucleic acid synthesis, DNA gyrase | Coutinho et al. (2017) |
| 29 | Lithotriptic/Antiurolithiatic | Alternanthera sessilis (L.) R.Br. ex DC. | Kalka - fine paste of macerated fresh plant material | 0.054 g/100g, 0.108 g/100g, and 0.216 g/100 g | 0.054 g/100g, 0.108 g/100g, and 0.216 g/100 g | Cystone (67.5 mg/kg) | 0.75% (v/v) ethylene glycol in drinking water and coconut water (0.86 ml/200 g) | Healthy adult albino rats | In vivo—Ethylene glycol induced urolithiasis | Act via diuretic activity, crystallization inhibition activity, improving renal function and antioxidant activity of the drugs | Dhanya et al. (2017) |
| Ethanolic extract of the whole plant | 10, 20, and 40 mg | 40 mg | Cystone (10 mg) | — | — | In vitro; Titrimetry, simultaneous flow static model, turbidimetry, and gravimetric methods | — | Babu et al. (2021) | |||
| 30 | Larvicidal | Alternanthera sessilis (L.) R.Br. ex DC. | Ethanolic extract of the whole plant | 20, 40, 60, 80 and 100 μg/ml | LC50–66.84 μg/ml | — | — | — | Percent mortality | — | Babu et al. (2021) |
| 31 | Locomotor | Alternanthera brasiliana (L.) Kuntze | Aqueous extract of leaves | 100, 200 and 400 mg/kg, p.o | 400 mg/kg, p.o | — | Distilled water (10 ml/kg, p.o.) | Male adult Wistar rats | In vivo—Open field exposure test | Act via an increase in their exploratory activities | Pelisoli Formagio et al. (2012) |
| Ethanolic extract of leaves | 250, 500 and 1,000 mg/kg, p.o | 500 and 1,000 mg/kg, p.o | Diazepam (1 mg/kg, i.p.) | Saline (10 ml/kg, p.o.) | Albino mice | In vivo—Novelty-induced behaviors | Act via regulation of different neurotransmitters such as GABA, ACh, noradrenaline, serotonin, glutamate, and dopamine | Oyemitan et al. (2015) | |||
| Alternanthera philoxeroides (Mart.) Griseb | Ethanolic extract of leaves | 250 and 500 mg/kg/day | Inactive | 17β-Estradiol (1 μg/kg, i.p.) | Distilled water (0.2 ml/mice, p.o.) | Female ICR mice | In vivo—Y-maze test | — | Khamphukdee et al. (2018) | ||
| 32 | Nootropic | Alternanthera sessilis (L.) R.Br. ex DC. | Methanolic extract of leaves | 100 or 200 mg/kg, p.o | 200 mg/kg, p.o | Bacopa monniera extract (40 mg/kg, p.o.) | Scopolamine (0.4 mg/kg, i.p.) | Adult Swiss albino Wistar mice | In vivo—rectangular maze and Y maze tests | Act via evoking pronounced alteration behavior and better learning assessments | Gupta and Singh, (2012a) |
| 33 | Photoprotective | Alternanthera brasiliana (L.) Kuntze | 5% w/w Gel from extract enriched with flavonoids | — | 5% w/w flavonoids rich gel | — | Gel base | — | In vitro—Mansur method | Act via the ability to stabilize reactive oxygen species, due to the presence of hydroxyl groups attached to the aromatic rings, allowing the resonance | Alencar Filho et al. (2020) |
| 34 | Sedative | Alternanthera brasiliana (L.) Kuntze | Ethanolic extract of leaves | 250, 500 and 1,000 mg/kg, p.o | 250, 500 and 1,000 mg/kg, p.o | Diazepam (1 mg/kg, i.p.) | Saline (10 ml/kg, p.o.) | Albino mice | In vivo—ketamine-induced hypnosis test | Act via stimulatory or central excitatory effect | Oyemitan et al. (2015) |
| Ketamine (100 mg/kg, i.p.) | |||||||||||
| 35 | Wound healing | Alternanthera brasiliana (L.) Kuntze | Methanolic extract of leaves | 5% ointment applied topically; 200 and 400 μg | 5% ointment applied topically; 400 μg | Himax ointment | Vaseline ointment and methylcellulose | Sprague Dawley rats | In vivo—Excision and incision wound model and Chorioallantoic membrane model | Act via an increase in collagen concentration and stabilization of fibers | Barua et al. (2009) |
| Methanolic extract of leaves | 5% w/w ointment applied topically | 5% w/w ointment applied topically | Himax ointment | Soft white petroleum jelly | Sprague Dawley rats | In vivo—burn wound model | Act via formation of the epidermis with keratin layer and deposition of collagen fibers | Barua et al. (2012a) | |||
| Methanolic extract of leaves | 2.5, 5.0 and 7.5% (w/w) ointment | 2.5, 5.0 and 7.5% (w/w) ointment | Himax ointment | Soft white petroleum jelly | Adult Sprague Dawley rats | In vitro—immunocompromised wound model | Act via collagen deposition, fibroblast proliferation, angiogenesis, and development of basement membrane | Barua et al. (2012b) | |||
| Methanolic extract of leaves | 5% (w/w) ointment | 5% (w/w) ointment | — | Soft white petroleum jelly | Healthy Sprague Dawley rats | In vivo—excision wound model | Act via wound contraction, fibroblastic deposition | Baru et al. (2012) | |||
| Alternanthera sessilis (L.) R.Br. ex DC. | 90% hydroethanolic extract of stem | 12.5–500 μg/ml | 50 and 300 μg/ml | Allantoin (50 μg/ml) | The natural rate of migration and viability of cells without extract | NHDF, HDF-D, and HaCaT cells | In vitro—wound scratch and MTT assay | Act via formation of the epidermis with keratin layer and deposition of collagen fibers | Muniandy et al. (2018b) | ||
| Chloroform extract of leaves | 200 μg/ml | 200 μg/ml | — | Saline water | Albino rats | In vivo—excision wound, incision wound, and dead space wound model | Act via increase collagen content, degree of collagen cross-linkage within the wound and promotes cell division, growth of bone, cartilage, and other connective tissues | Jalalpure et al. (2008) |
Pharmacological activities of genus Alternanthera.
Referring to the data tabulated in Table 2, and interactive Figure 3, it is quite evident that the Alternanthera genus is having tremendous potential having polypharmacological effects. 35 different types of pharmacological effects were elicited by different species of Alternanthera genus. While the species like Alternanthera sessilis (L.) R.Br. ex DC., Alternanthera brasiliana (L.) Kuntze, and Alternanthera philoxeroides (Mart.) Griseb. were most widely explored, it opens up the opportunity for the researchers to explore other species of this genus.
FIGURE 3
Analgesic Activity
Pelisoli Formagio and the team had evaluated the aqueous extract from the aerial parts of Alternanthera brasiliana (L.) Kuntze for its analgesic potential. 90.35% reduction of acetic acid induced contractions were observed in mice, when treated with 25 mg/kg of the aqueous extract (Pelisoli Formagio et al., 2012). Coutinho and the team had performed the formalin test in mice for assessment of analgesic effect of ethanolic extract from the leaves of Alternanthera brasiliana (L.) Kuntze. At 100 mg/kg, ethanolic extract was capable of reducing the edematogenic process by 64.17% (Coutinho et al., 2017). Phytoconstituents like kaempferol (Parveen et al., 2007), quercetin (Anjaneyulu and Chopra, 2003), vitexin (Zhu et al., 2016), etc may be responsible for the analgesic potential of Alternanthera brasiliana (L.) Kuntze.
de Santana Aquino and the team had evaluated ethanolic extract as well as isolated compound, 2″-O-α-L-rhamnopyranosylvitexin from the aerial parts of Alternanthera littoralis P.Beauv. for analgesic potential. Results suggested that the ethanolic extract as well as 2″-O-α-L-rhamnopyranosylvitexin are capable of exerting significant analgesic effect, most probably through the TNF pathway (de Santana Aquino et al., 2015). Since kaempferol, quercetin, and vitexin were also been reported from Alternanthera littoralis P.Beauv. (Figure 2), so these compounds could also attribute in analgesic potential of the extract.
Khatun and the team had prepared the methanolic extract from the whole plant part of Alternanthera philoxeroides (Mart.) Griseb. and evaluated for its analgesic potential in the acetic acid induced mice. They found that 400 mg/kg dose of methanolic extract was capable of reducing constrictions by 44.8%. Phytoconstituents like kaempferol (Parveen et al., 2007), quercetin (Anjaneyulu and Chopra, 2003), vitexin (Zhu et al., 2016), caffeic acid (Gamaro et al., 2011), ursolic acid (Vasconcelos et al., 2006), etc may be responsible for the analgesic potential of Alternanthera philoxeroides (Mart.) Griseb.
Various research teams have independently assessed the analgesic potential of Alternanthera sessilis (L.) R.Br. ex DC.: Mondal and the team used ethanolic extract of the leaves (Mondal et al., 2014); Mohapatra and the team used hydroethanolic extract of leaves (Mohapatra et al., 2018); Hossain and the team used methanolic extract of aerial parts (Hossain et al., 2014); while Mohaimenul and the team used ethanolic extract of aerial parts (Mohaimenul et al., 2020). It is thus quite validated that aerial parts especially leaves of Alternanthera sessilis (L.) R.Br. ex DC. have the analgesic potential. Various mechanisms observed by those researchers for this activity. Some of them are like inhibition of interleukins like IL-4, IL-5, and IL-13, dopaminergic and serotonergic pathways, inhibition of lipoxygenase and cyclooxygenase, etc. Along with kaempferol, vitexin, and quercetin, compounds like stigmasterol (Walker et al., 2017) may also be responsible for such analgesic effect.
Anthelmintic Activity
Vennila and Nivetha had prepared various extracts from the leaves of Alternanthera sessilis (L.) R.Br. ex DC. and performed In vitro—Pheretima Posthuma method for assessment of anthelmintic activity. They observed that methanolic extract was the most potent and active at all the tested concentrations. The possible mechanism proposed by them was membrane lysis which subsequently led to paralysis or death of the worm (Vennila and Nivetha, 2015). On the other hand, Mondal and the team had assessed anthelmintic activity of ethanolic extract of the whole plant as well as the isolated ellagic acid by using In vitro—Adult motility test. They had also indicated the disruption of cell permeability, along with various other pathways and found ellagic acid a key responsible compound (Mondal et al., 2015). Other compounds that may be responsible for this pharmacological effects could be quercetin (Borges et al., 2020), β-sitosterol (Deepak et al., 2002), etc.
Antiallergic Activity
Rayees and the team checked the antiallergic activity of 95% ethanolic extract from aerial parts of Alternanthera sessilis (L.) R.Br. ex DC. Studies were conducted in rat basophilic leukemia (RBL-2H3) cells. They found that the treatment with ethanolic extract resulted in nuclear factor-KB (NF-kB) dependent inhibition of cytokines like IL-6, TNF-α, IL-13, and IL-4, along with the decrease in β-hexosaminidase release (Rayees et al., 2013). Compounds like β-sitosterol (Yuk et al., 2007; Mahajan and Mehta, 2011), kaempferol (Oh et al., 2013), quercetin (Mlcek et al., 2016), vitexin (Venturini et al., 2018), stigmasterol (Antwi et al., 2018), etc may be responsible for the antiallergic activity of Alternanthera sessilis (L.) R.Br. ex DC.
Antianxiety Property
Various research teams have independently assessed the antianxiety potential of Alternanthera brasiliana (L.) Kuntze: Pelisoli Formagio had used the aqueous extract of the leaves (Pelisoli Formagio et al., 2012); Oyemitan and the team had used the ethanolic extract of the leaves (Oyemitan et al., 2015); while Barua and the team had used the methanolic extract of the leaves (Barua et al., 2013). It is thus quite validated that the leaves of Alternanthera brasiliana (L.) Kuntze have the antianxiety potential. Various mechanisms observed by those researchers for this activity. Some of them are like activation of GABA receptor and 5-HT partial agonistic action. Phytomolecules like stigmasterol (Karim et al., 2021), kaempferol (Kaur et al., 2017), quercetin (Singh et al., 2013), p-coumaric acid (He Y. et al., 2021), etc may be responsible for this antianxiety property of Alternanthera brasiliana (L.) Kuntze.
Khamphukdee and the team had assessed ethanolic extract from the leaves of Alternanthera philoxeroides (Mart.) Griseb. for antianxiety potential by performing In vivo—Elevated plus-maze test, Light/Dark transition test, and Locomotor activity test in female mice. They observed that both the test doses i.e. 250 and 500 mg/kg/day of the extract was able to reduce the anxiety, most probably through the esterogenic pathway. Quercetin and kaempferol were detected in this plant also, so may be responsible for such antianxiety behavior.
Antiapoptotic Activity
Wu and the team had studied the antiapoptotic potential of ethanolic extract from the whole plant of Alternanthera bettzickiana (Regel) G.Nicholson. They found that ethanolic extract has strong tendency to reduce apoptosis which was modulated via multiple mechanisms including reduction of reactive oxygen species, inhibition of caspase-3 and caspase-9 activation, etc. They had reported quercetin as the major compound in that extract, and they found same mechanisms when evaluated quercetin for antiapoptotic potential.
Antiarthritic Activity
Manan and the team had studied antiarthritic potential of the ethanolic extract obtained from the aerial parts of Alternanthera bettzickiana (Regel) G.Nicholson using in silico, in vitro and in vivo methodologies. HPLC analysis indicated the presence of catechin, gallic acid, sinapic acid, chlorogenic acid, alpha-tocopherol, gamma-tocopherol, and quercetin. They have found that even the 250 mg/kg/day of the ethanolic extract was able to modulate the parameters suggesting the antiarthritic potential when compared with standard drug and disease control. In silico analysis suggested the strong interaction between the HPLC-analysed phytomolecules and cyclooxygenases (Manan et al., 2020).
Sunmathi and the team had studied the antiarthritic activity of ethanolic extracts obtained from the leaves of Alternanthera philoxeroides (Mart.) Griseb. and Alternanthera sessilis (L.) R.Br. ex DC. using in vitro methodologies. They found that 500 μg/ml of ethanolic extract of Alternanthera philoxeroides (Mart.) Griseb. and Alternanthera sessilis (L.) R.Br. ex DC. were able to stabilize the membrane by 64.92 and 75.43%, respectively. Phytomolecules like vitexin (Yang et al., 2019) and quercetin (Mamani-Matsuda et al., 2006) may be responsible for the antiarthritic activity of Alternanthera philoxeroides (Mart.) Griseb. and Alternanthera sessilis (L.) R.Br. ex DC.
Antiasthmatic Activity
Various research teams have independently assessed the antiasthmatic potential of Alternanthera sessilis (L.) R.Br. ex DC.: Fathima and the team had used ethanolic extract of leaves (Fathima et al., 2016) while Saqib and Janbaz had used 70% Ethanolic extract of the whole plant and its dichloromethane and aqueous fractions (Saqib and Janbaz, 2016). This validates the applicability of Alternanthera sessilis (L.) R.Br. ex DC. in the treatment management of asthma. Ethanolic extract obtained from the leaves was found to reduce the leucocyte count and significantly inhibited the histamine release (Fathima et al., 2016). 70% ethanolic extract of the whole plant was found to act via calcium channel blocking mechanism (Saqib and Janbaz, 2016). Phytomolecules like kaempferol (Gong et al., 2012), vitexin (Venturini et al., 2018), quercetin (Fortunato et al., 2012), stigmasterol (Antwi et al., 2017a), chlorogenic acid (Kim et al., 2010), etc. may be key components for the antiasthamatic activity of Alternanthera sessilis (L.) R.Br. ex DC.
Anticancer/Cytotoxic Property
Various research teams have independently assessed the anticancer property of Alternanthera bettzickiana (Regel) G.Nicholson: M Nagalingam and the team had used aqueous extract of the leaves (Nagalingam et al., 2018) while R Jothi Ramalingam and the team had used aqueous extract of leaves and silver nanoparticles and Ag-mesoporous MnO2 nanocomposite (Jothi Ramalingam et al., 2017). This validates the potential of leaves from Alternanthera bettzickiana (Regel) G.Nicholson and their nanoparticles in colon cancer and lung cancer. Apigenin analogues present in the Alternanthera bettzickiana (Regel) G.Nicholson may be responsible for the anticancer property (Madunić et al., 2018; Imran et al., 2020).
Similarly, various research teams have independently assessed the anticancer property of Alternanthera brasiliana (L.) Kuntze: Brochado and the team had used aqueous fraction of the ethanolic extract from the leaves. They had also isolated 6 bioactive compounds from this fraction viz. robinin, clovin, quercetin 3-O-robinobioside, kaempferol 3-O-robinobioside, kaempferol 3-O-rutinoside-7-O-a-L-rhamnopyranoside, and kaempferol 3-O-rutinoside (Brochado et al., 2003); Samudral and the team had used ethyl acetate extract obtained from the leaves (Samudrala et al., 2015). These pieces of evidence validates the anticancer potential of Alternanthera brasiliana (L.) Kuntze leaves. Brochado and the team found Kaempferol 3-O-robinobioside and kaempferol 3-O-rutinoside as the active phytomolecules (Brochado et al., 2003).
Independently several researches had also been conducted from various labs to assess the potential of Alternanthera philoxeroides (Mart.) Griseb. as anticancer agent: Zhang and the team had used the methanolic extract of the leaves and checked cytotoxicity against H9c2 cell lines. They found that even at 20 mg/ml, the methanolic extract was able to inhibit the doxorubicin induced cardiomyocyte apoptosis by more than 50%. They had also observed the presence of -carboline and quercetin (Zhang et al., 2018). Fang and the team had isolated 5 phytomolecules from the aerial parts of Alternanthera philoxeroides (Mart.) Griseb., and checked their inhibitory activity against Hela and L929 cell lines. While N-trans-feruloyl-3,5-dimethoxytyramine, alternanthin, N-trans-feruloyl-3-methyldopamine, and N-trans-feruloyl tyramine were found to have more than 50% inhibition at 30 μg/ml against Hela cell line, only Alternanthin B, and alternanthin were having more than 50% inhibition at 30 μg/ml against L929 cell line (Fang et al., 2007). Fang and the team had further isolated 4 more compounds from the aerial parts of Alternanthera philoxeroides (Mart.) Griseb. The triterpenoidal saponins, Philoxeroidesides A, B, C, and D were found to inhibit SK-N-SH cell line with an IC50 of 51, 118.69, 60.6, and 37.29 μg/ml, respectively, while inhibited HL60 cell line with an IC50 of 185.29, 185.57, 271.45, and 45.93 μg/ml, respectively. Philoxeroidesides D was found to be quite potential against both the cell lines (Fang J.-B. et al., 2009). In another study performed by Correa and the team where they had used ethanolic extracts obtained from the whole plant of Alternanthera philoxeroides (Mart.) Griseb.; Alternanthera hirtula (Mart.) R.E.Fr., and Alternanthera praelonga A.St.-Hil. They tested the ethanolic extracts against various human cancer cells lines including that from melanoma, breast, kidney, lung, prostate, ovary, colon, leukemia, along with non-cancer cell line from green monkey kidney. Out of all the cancer cell lines, these ethanolic extracts were being able to be found potent only against the leukemia cell line, K562 (Correa et al., 2016).
Several researchers have independently assessed the potential of Alternanthera sessilis (L.) R.Br. ex DC. for the management of cancer: Jain and the team had used the methanolic extract of leaves (Jain et al., 2016); Firdhouse and Lalitha had used silver nanoparticles of the aqueous extract (Firdhouse and Lalitha, 2013); Qian and the team had used gold nanoparticles of the aqueous extract of leaves (Qian et al., 2019); D Suganya and the team had used aqueous extract of leaves and stems (Suganya et al., 2019); Pathak and the team had used n-hexane and methanolic extracts of aerial parts (Pathak et al., 2020); Mohaimenul and the team had used ethanolic extract of aerial parts (Mohaimenul et al., 2020); Yap and the team had used ethanolic, 70% ethanolic, 80% methanolic, ethyl acetate, and aqueous extracts of the whole plant (Yap et al., 2019); Sathishkumar and the team had used silver nanoparticles of the aqueous extract of leaves (Sathishkumar et al., 2016); Arulselvan and the team had used ethanolic extract of aerial parts, stem, and leaves (Arulselvan et al., 2018); while Guerra and the team aqueous extract of aerial parts (Guerra et al., 2003). All these studies indicated the true potential of Alternanthera sessilis (L.) R.Br. ex DC. for the treatment and management of cancer, with leaving no doubt in it. Phytomolecules present in the Alternanthera sessilis (L.) R.Br. ex DC. like kaempferol (Imran et al., 2019), vitexin (Liu et al., 2019; Lee et al., 2020), quercetin (Rauf et al., 2018), stigmasterol (Ali et al., 2015), chlorogenic acid (Barahuie et al., 2017), campesterol (Bae et al., 2021), and β-sitosterol (Pradhan et al., 2016), etc. may be responsible for this anticancer property.
Anticataract Property
Kota and the team had checked the anticataract property of ethyl acetate extract obtained from the leaves of Alternanthera sessilis (L.) R.Br. ex DC. Cataract induced in eye lenses of the chicks were subjected for the treatment with 100, 200, and 400 mg of ethyl acetate extract, followed by analysis of lipid peroxidation and Na+- K+ ATPases. They found that 100 and 200 mg ethyl acetate treatment will lead to decrease in malondialdehyde and increase in the inorganic phosphorous content (Kota et al., 2017). Phytomolecules like quercetin (Lan et al., 2020), chlorogenic acid (Kim et al., 2011), and β-sitosterol (Haroon et al., 2020) may be responsible for this anticataract property of Alternanthera sessilis (L.) R.Br. ex DC.
Anticonvulsant Activity
Independently several researches had also been conducted from various labs to assess the potential of Alternanthera brasiliana (L.) Kuntze as anticonvulsant agent: Oyemitan and the team had used the ethanolic extract of leaves (Oyemitan et al., 2015); Schallenberger and the team had also used the ethanolic extract of leaves (Schallenberger et al., 2017); while Barua and the team had used the methanolic extract of leaves (Barua et al., 2013). This had validated the anticonvulsant potential of the leaves of Alternanthera brasiliana (L.) Kuntze. Various mechanisms elucidated by them are like modulation of GABAergic system, controlling the entry of calcium and sodium ions in the cells, and glycine regulation in spinal cord (Oyemitan et al., 2015). Phytomolecules like vitexin (de Oliveira et al., 2020), quercetin (Nassiri-Asl et al., 2014; Nieoczym et al., 2014), stigmasterol (Karim et al., 2021), chlorogenic acid (Aseervatham et al., 2016), and ferulic acid (Hassanzadeh et al., 2017) may be responsible for the antiepileptic effect of Alternanthera brasiliana (L.) Kuntze.
Antidepressant Activity
Khamphukdee and the team had assessed the antidepressant effect of the ethanolic extract obtained from the leaves of Alternanthera philoxeroides (Mart.) Griseb. They found that the extract was having significant antidepressant effect modulated through the estrogenic pathway (Khamphukdee et al., 2018). Phytomolecules like quercetin (Anjaneyulu and Chopra, 2003), vitexin (Can et al., 2013), β-sitosterol (Zhao et al., 2016), p-coumaric acid (Lee et al., 2018), caffeic acid (Monteiro et al., 2020), ursolic acid (Machado et al., 2012; Singla et al., 2017), and malic acid (Gómez-Moreno et al., 2013) may be responsible for the antidepressant activity of Alternanthera philoxeroides (Mart.) Griseb.
Gupta and K. Singh had evaluated the antidepressant activity of methanolic extract obtained from the leaves of Alternanthera sessilis (L.) R.Br. ex DC. They had observed that the antidepressant effect of the methanolic extract was acting via interaction with adrenergic, dopaminergic serotonergic, and GABAergic system (Gupta and Singh, 2014). Phytomolecules like quercetin, vitexin, and p-coumaric acid had also been reported from Alternanthera sessilis (L.) R.Br. ex DC., along with other antidepressant agents like kaempferol (Park et al., 2010b), ferulic acid (Chen et al., 2014) and chlorogenic acid (Park et al., 2010a). These phytomolecules may be responsible for the antidepressant activity of Alternanthera sessilis (L.) R.Br. ex DC.
Antidiabetic Activity
Reza and the team had assessed the antidiabetic potential of 80% ethanolic extracts obtained from the stem and leaves of Alternanthera brasiliana (L.) Kuntze. They found that the ethanolic extracts were being able to significantly modulate the biochemical parameters like blood glucose, lipid peroxidation, and free radicals in the alloxan-induced diabetic Swiss albino mice (Reza et al., 2019). Phytomolecules like kaempferol (Ibitoye et al., 2018), quercetin (Vessal et al., 2003), stigmasterol (Wang et al., 2017; Singla and Shen, 2020), p-coumaric acid (Amalan et al., 2016), ferulic acid (Narasimhan et al., 2015), and chlorogenic acid (Ong et al., 2013) may be responsible for the antidiabetic potential of Alternanthera brasiliana (L.) Kuntze.
Khatun and the team as well as Bhattacherjee and the team had independently assessed the antidiabetic activity of Alternanthera philoxeroides (Mart.) Griseb. Various important mechanisms had been observed by them including regeneration of the β-cells of the pancreas, alpha-glucosidase inhibition, as well as the inhibition of the glucose absorption from the gut wall (Khatun et al., 2012; Bhattacherjee et al., 2014). Compounds like quercetin and p-coumaric acid had been reported from Alternanthera philoxeroides (Mart.) Griseb., and may be responsible for such antidiabetic effect.
Mourya and the team had used aqueous and ethanolic extracts obtained from the whole plant of Alternanthera pungens Kunth for the assessment of antidiabetic potential. Dose dependent antidiabetic activity was observed by them when studied in alloxan-induced diabetic Wistar rats. Phytocompounds like camphene (Hachlafi et al., 2021), camphor (Drikvandi et al., 2020), geraniol (Babukumar et al., 2017), and limonene (Murali and Saravanan, 2012) may be responsible for such antidiabetic property of Alternanthera pungens Kunth.
Independently several researches had also been conducted from various labs to assess the potential of Alternanthera sessilis (L.) R.Br. ex DC. as antidiabetic agent: Kumar and the team had used aqueous and ethanolic extracts of aerial parts (Kumar S. M. et al., 2011); Tan and Kim had used hexane, ethyl acetate, and aqueous fractions of aerial parts (Tan and Kim, 2013); Hossain and the team had used methanolic extract of aerial parts (Hossain et al., 2014); Sundar and the team had used petroleum ether extract of leaves (Sundar et al., 2019); Das and the team had used 95% ethanolic extract of the whole plant (Das et al., 2015); Rao and the team had used ethanolic extract of the whole plant (Rao et al., 2011); Manalo and the team had used n-hexane, ethyl acetate, and water fractions of the methanolic extract of leaves (Manalo et al., 2020); Mohaimenul and the team had used ethanolic extract of aerial parts (Mohaimenul et al., 2020); Tiwari and the team had used the juice (Tiwari et al., 2013); Chai and the team had used hexane, chloroform, ethyl acetate, butanol, and aqueous fractions of methanolic extracts of leaves and callus (Chai et al., 2016). Plenty of evidences obtained from the above researches leaved no doubt in that fact that Alternanthera sessilis (L.) R.Br. ex DC. possesses antidiabetic properties. Various mechanisms demonstrated by different preparations from Alternanthera sessilis (L.) R.Br. ex DC., including but not limited to modulation of insulin sensitivity, improvement in pancreatic insulin secretion, reduction in blood glucose level, inhibition of α-glucosidase enzyme, etc. Phytomolecules like kaempferol (Ibitoye et al., 2018), quercetin (Vessal et al., 2003), stigmasterol (Wang et al., 2017; Singla and Shen, 2020), 4-hydroxybenzoic acid (Peungvicha et al., 1998), β-sitosterol (Ponnulakshmi et al., 2019), ellagic acid (Fatima et al., 2015), ferulic acid (Narasimhan et al., 2015), and chlorogenic acid (Ong et al., 2013) may be responsible for the antidiabetic potential of Alternanthera sessilis (L.) R.Br. ex DC.
Antidiarrheal Activity
Zavala and the team had evaluated the antidiarrheal property of hexane, chloroform, methanolic, and aqueous extracts obtained from the whole plant of Alternanthera sessilis (L.) R.Br. ex DC. They had observed that out of all extracts, methanolic and aqueous extracts had shown significant inhibition of castor oil-induced diarrhea. Methanolic extract was further found to inhibit normal defecation in mice also. Peristaltic movement was also modulated by the methanolic extract (Zavala et al., 1998). Phytomolecules like quercetin (Lozoya et al., 1994; Song et al., 2011; Shi et al., 2020), β-sitosterol (Ding et al., 2018), ellagic acid (Chen et al., 2020), ferulic acid (Hu et al., 2021), and chlorogenic acid (Zhang et al., 2017; Chen et al., 2018) may be responsible for the antidiarrheal property of Alternanthera sessilis (L.) R.Br. ex DC.
Antigout Activity
Chong and Loh had assessed the antigout potential of methanolic extract obtained from the aerial parts of Alternanthera sessilis (L.) R.Br. ex DC. Methanolic extract was able to inhibit xanthine oxidase enzyme with an IC50 of 557.77 μg/ml (Chong and Loh, 2020). Phytomolecules like kaempferol (Wang et al., 2015d), quercetin (Bindoli et al., 1985), stigmasterol (Chiang and Chen, 2008), ellagic acid (Sun et al., 2021), ferulic acid (Nile et al., 2016), and chlorogenic acid (Wang et al., 2009) may be responsible for the antigout potential of Alternanthera sessilis (L.) R.Br. ex DC.
Anti-Hepatitis B Virus Activity
Li and the team had isolated C-boivinopyranosyl flavones from Alternanthera philoxeroides (Mart.) Griseb. and found that luteolin-6-C-β-d-boivinopyranosyl-3′-O-β-d-glucopyranoside, chrysoeriol-6-C-β-d-Boivinopyranosyl-4′-O-β-d-glucopyranoside, and luteolin-6-C-β-d-boivinopyranosyl-4′-O-β-d-glucopyranoside were strongly inhibiting the viral antigen, HBsAg in HBV-infected HepG2.2.15 with an IC50 of 28.65, 22.20, and 31.54 µM, respectively (Li et al., 2016).
Antihypertensive Activity
Saqib and Janbaz had evaluated the antihypertensive effect of 70% Ethanolic extract of the whole plant and its dichloromethane and aqueous fractions from Alternanthera sessilis (L.) R.Br. ex DC. The in vivo studies suggested that the ethanolic extract was capable to reducing both the systolic and the diastolic pressure. Phytomolecules like kaempferol (Ahmad et al., 1993; Binang and Takuwa, 2021), quercetin (Perez-Vizcaino et al., 2009; Binang and Takuwa, 2021), vitexin (Xue et al., 2020), β-sitosterol (Olaiya et al., 2014), ellagic acid (Berkban et al., 2015), ferulic acid (Li et al., 2020), and chlorogenic acid (Zhao et al., 2011) may be responsible for the antihypertensive potential of Alternanthera sessilis (L.) R.Br. ex DC.
Anti-Inflammatory Activity
Pelisoli Formagio and the team had performed the in vivo studies to assess the anti-inflammatory activity of the aqueous extract obtained from the leaves of Alternanthera brasiliana (L.) Kuntze while P Shivashankar and the team had used the methanolic extract obtained from the leaves. Pelisoli Formagio and the team had observed the significant decrease in the polymorphonuclear cells as well as increase in the mononuclear cells in rat’s exudate after treated with the aqueous extract, while P Shivashankar and the team found the reduction in the colon weight in acetic acid-induced colitis model of adult Wistar albino rats after treatment with the methanolic extract (Pelisoli Formagio et al., 2012; P et al., 2016). Phytomolecules like kaempferol (Devi et al., 2015), quercetin (Lesjak et al., 2018), stigmasterol (Morgan et al., 2021), p-coumaric acid (Pragasam et al., 2012), ferulic acid (Ozaki, 1992), and chlorogenic acid (Hwang et al., 2013) may be responsible for the anti-inflammatory potential of Alternanthera brasiliana (L.) Kuntze.
de Santana Aquino and the team had evaluated anti-inflammatory activity of ethanolic extract of aerial parts and the isolated compound, 2″-O-α-L-rhamnopyranosylvitexin from Alternanthera littoralis P.Beauv. They found that the ethanolic extract was able to reduce the paw edema as well as capable to reducing leukocyte migration. In addition to these, the isolated compound was also able to reduce protein leakage into the pleural cavity (de Santana Aquino et al., 2015). Other phytomolecules that could be responsible for the anti-inflammatory activity of the ethanolic extract will be kaempferol, quercetin, stigmasterol, etc.
Sunmathi and the team had evaluated anti-inflammatory activity of ethanolic extract obtained from the leaves of Alternanthera philoxeroides (Mart.) Griseb. Dose dependent membrane stabilization was observed. Phytomolecules like quercetin (Lesjak et al., 2018), vitexin (Rosa et al., 2016), β-sitosterol (Loizou et al., 2010), p-coumaric acid (Pragasam et al., 2012), caffeic acid (da Cunha et al., 2009), ursolic acid (Baricevic et al., 2001), and malic acid (Obertreis et al., 1996) may be responsible for the anti-inflammatory activity of Alternanthera philoxeroides (Mart.) Griseb.
Franck and the team had evaluated the anti-inflammatory activity of aqueous extract obtained from the leaves of Alternanthera pungens Kunth. They had observed the decreased level of histamine release, serotonin and kinin, prostaglandin, proteases, lysosomes, and protein C-reactive. Phytomolecules like α-pinene (Kim et al., 2015), myrcene (Rufino et al., 2015), limonene (Rufino et al., 2015), choline (Rowley et al., 2010), rhein (Gao et al., 2014), linalool (Peana et al., 2002), geraniol (Ye et al., 2019), and camphor (Ehrnhöfer-Ressler et al., 2013) which were reported earlier in Alternanthera pungens Kunth., may be responsible for this anti-inflammatory effect.
Independently several researches had also been conducted from various labs to assess the potential of Alternanthera sessilis (L.) R.Br. ex DC. as anti-inflammatory agent: Sunmathi and the team had used ethanolic extract obtained from the leaves (Sunmathi et al., 2016); Muniandy and the team had used 90% ethanolic extract of stems (Muniandy et al., 2018a); Sundar and the team had used petroleum ether and methanolic extracts of leaves (Sundar et al., 2019); Kassuya and the team had used Ethanolic extract of whole plant (EEAT) as well as the isolated molecule, 2″-O-β-D-glucopyranosyl-vitexin (Kassuya et al., 2021); Biella and the team had used aqueous extract of the whole plant (Biella et al., 2008). Plenty of evidences obtained from the above researches leaved no doubt in that fact that Alternanthera sessilis (L.) R.Br. ex DC. possesses anti-inflammatory properties. Various mechanisms demonstrated by different preparations from Alternanthera sessilis (L.) R.Br. ex DC., including but not limited to cyclooxygenase -1 and -2 inhibition (Biella et al., 2008), modulating NF- κB pathway (Muniandy et al., 2018a), leukocyte migration (Kassuya et al., 2021), etc. Phytomolecules like kaempferol (Devi et al., 2015; Pizzo et al., 2018), quercetin (Lesjak et al., 2018), vitexin (Rosa et al., 2016), stigmasterol (Morgan et al., 2021), β-sitosterol (Loizou et al., 2010), 4-hydroxybenzoic acid (Winter et al., 2017), ellagic acid (Corbett et al., 2010), ferulic acid (Ozaki, 1992), campesterol (Moreno-Anzúrez et al., 2017), spinasterol (Jeong et al., 2010), β-carotene (Uteshev et al., 2000), p-coumaric acid (Pragasam et al., 2012), ricinoleic acid (Vieira et al., 2001), and chlorogenic acid (Hwang et al., 2013) may be responsible for the anti-inflammatory potential of Alternanthera sessilis (L.) R.Br. ex DC.
Antimicrobial Activity
Independently, several research teams had evaluated the antimicrobial effects of the leaves of Alternanthera bettzickiana (Regel) G.Nicholson: Vidhya and the team had used hexane, chloroform, ethyl acetate, methanolic, and aqueous extracts of leaves (Vidhya et al., 2015); R, Jothi Ramalingam and the team had used aqueous extract of leaves and silver nanoparticles and Ag-mesoporous MnO2 nanocomposite (Jothi Ramalingam et al., 2017); Nagalingam and the team had used the aqueous extract obtained from leaves (Au-NP) (Nagalingam et al., 2018). These research were focused on leaves and somehow validated the antimicrobial property of it. Various mechanisms elucidated were like cell wall lysis, protein synthesis inhibition, and topoisomerase inhibition, etc (Vidhya et al., 2015; Jothi Ramalingam et al., 2017; Nagalingam et al., 2018). Phytocompounds like apigenin analogs (Koo, 2003; Thirukumaran et al., 2019) may be responsible for this antimicrobial property of Alternanthera bettzickiana (Regel) G.Nicholson.
Coutinho and the team had evaluated the antimicrobial property of ethanolic extract obtained from the leaves of Alternanthera brasiliana (L.) Kuntze. They had observed that though the ethanolic extract as such was having insignificant potential, but it elicited significant synergetic potential when combined with gentamycin and tested against Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa (Coutinho et al., 2017). Johann and the team had also performed the antimicrobial experiments on the ethanolic extract obtained from the aerial parts of Alternanthera brasiliana (L.) Kuntze, and they had also observed that the extract was inactive against various murine macrophages and fungal strains (Johann et al., 2010). Other research team like that of Akachukwu and Uchegbu had also reported mild activity of the ethanolic extract obtained from its leaves (Akachukwu and Uchegbu, 2016) while Kumar and the team noticed significant activity elicited by the silver nanoparticles obtained from the leaves aqueous extract (Kumar et al., 2014).
Canales-Martínez and the team had evaluated the antimicrobial effect of the hexane, chloroform, methanolic, acetone, and ethyl acetate extracts obtained from the aerial parts of Alternanthera caracasana Kunth and also isolated a bioactive compound, 7-methoxycoumarin. They observed that the ethyl acetate extract as well as 7-methoxycoumarin were active against various Gram-positive and Gram-negative bacterial strains, but inactive against Candida albicans (Canales-Martínez et al., 2008). Phytochemical profiling of Alternanthera caracasana Kunth is still not done, leaving a scope for the researchers.
Gasparetto and the team had used crude hexane and ethanolic extract obtained from the leaves of Alternanthera littoralis P.Beauv., and assessed them for their antimicrobial potential. They noticed that the antifungal activity was exhibited by the crude extracts only when combined with photo-irradiation by a diode laser (Gasparetto et al., 2010). Phytocompounds like kaempferol (del Valle et al., 2016), stigmasterol (Alawode et al., 2021), hydroxytyrosol (Bisignano et al., 1999), quercetin (Gatto et al., 2002), vitexin (Das et al., 2016), and uridine (Wiegmann et al., 2016) which were reported earlier from Alternanthera littoralis P.Beauv., may be responsible for such antimicrobial effects.
Feka and the team had studied the antimicrobial property of the aqueous and methanolic extracts obtained from the whole plant of Alternanthera nodiflora R.Br. They found that the methanolic extract was having significant antimicrobial activity against bacterial and yeast strains, but inactive against mould test strain (Feka et al., 2014). Phytochemical profiling of Alternanthera nodiflora R.Br. is still not done, leaving a scope for the researchers.
Independently several research teams had evaluated the antimicrobial potential of Alternanthera philoxeroides (Mart.) Griseb.: Bhattacherjee and the team had used methanol-soluble fraction obtained from the leaves (Bhattacherjee et al., 2014); Rawani and the team had used aqueous and chloroform: methanol (1:1) extracts of leaves (Rawani et al., 2011); Pulipati and the team had used ethanolic extract obtained from the leaves (Pulipati et al., 2016); Akbar and the team had used methanolic extract of leaves, stem and roots as well as their n-hexane, chloroform and ethyl acetate fractions (Akbar et al., 2021); while Pulipati and Babu had used the methanolic extract of leaves (Pulipati and Babu, 2020). These independent researches left no doubt and validated the antimicrobial feature of Alternanthera philoxeroides (Mart.) Griseb. They had reported multiple mechanisms of actions like bacterial cell wall lysis and protein synthesis inhibition (Bhattacherjee et al., 2014; Pulipati et al., 2016; Pulipati and Babu, 2020). Phytomolecules like quercetin (Gatto et al., 2002), vitexin (Das et al., 2016), β-sitosterol (Ododo et al., 2016), stigmasterol (Alawode et al., 2021), p-coumaric acid (Boz, 2015), caffeic acid (Lima et al., 2016), luteolin analogs (Chiruvella et al., 2007; Qian et al., 2020), chrysoeriol analogs (Jang et al., 2020), malic acid (Raybaudi-Massilia et al., 2009), β-carboline (Arshad et al., 2008; Suzuki et al., 2018), ursolic acid (Collins and Charles, 1987), oleanolic acid (Horiuchi et al., 2007), azelaic acid (Leeming et al., 1986), phytol (Pejin et al., 2014), and rubiadin (Marioni et al., 2016) which were earlier reported from Alternanthera philoxeroides (Mart.) Griseb., may be responsible for this antimicrobial property.
Jakhar and Dahiya had studied the aqueous, acetone, ethanolic, and petroleum ether extracts obtained from the aerial parts of Alternanthera pungens Kunth for assessment of antimicrobial effect against various bacterial and fungal strains. They found that all the extracts were having potential as antibacterial, but the antifungal property was exhibited by only acetone and aqueous extracts. Noticed mechanisms were inhibition of DNA replication as well as blocking of cellular respiration. Phytochemicals like choline (Siopa et al., 2016), rhein (Joung et al., 2012), limonene (Vuuren and Viljoen, 2007), α-curcumene (Santos da Silva et al., 2015), geraniol (Lira et al., 2020), linalool (Park S.-N. et al., 2012), camphor (Masry et al., 2021), myrcene (Chaves-Quirós et al., 2020), and α-pinene (Dhar et al., 2014; Cloeckaert et al., 2015) which were earlier reported from Alternanthera pungens Kunth, may be responsible for such antimicrobial action.
Plenty of independent researches have been extracted from the literature, covering evaluation of antimicrobial activity of Alternanthera sessilis (L.) R.Br. ex DC.: Osuna and the team had used hexane and methanolic extracts obtained from the aerial parts (Osuna et al., 2008); Jalalpure and the team had used petroleum ether (40–60°C), chloroform, acetone, methanolic, and aqueous extracts of leaves (Jalalpure et al., 2008); Monroy and Limsiaco had used aqueous, ethanolic, and acetone extracts obtained from leaves (Monroy and Limsiaco, 2016); Niraimathi and the team had used silver nanoparticles of aqueous extract of leaves (Niraimathi et al., 2013); Rajamurugan and the team had used ethanolic extract obtained from the leaves (Rajamurugan et al., 2013); D Suganya and the team had used aqueous extract of leaves and stems (Suganya et al., 2019); Kota and the team had used petroleum ether, ethyl acetate, chloroform, and methanolic extract obtained from the leaves (Kota et al., 2017); Sundar and the team had used petroleum ether and methanolic extracts of leaves (Sundar et al., 2019); while Salvador and the team had used hexane and ethanolic extracts obtained from the adult plants (Salvador et al., 2009). These studies clearly concluded that Alternanthera sessilis (L.) R.Br. ex DC. possesses antimicrobial properties. Several mechanisms elucidated by them are like cell membrane lysis, prevention of protein synthesis, blocking cellular respiration, inhibition of DNA replication, deprivation of iron for microbial growth, etc (Osuna et al., 2008; Salvador et al., 2009; Rajamurugan et al., 2013; Monroy and Limsiaco, 2016; Kota et al., 2017; Suganya et al., 2019). Phytomolecules like Vitexin (Das et al., 2016), Kaempferol (del Valle et al., 2016), Quercetin (Gatto et al., 2002), Kaempferol-7- O-glucoside (Singh et al., 2011), Stigmasterol (Alawode et al., 2021), β-Sitosterol (Ododo et al., 2016), Ellagic acid (Abuelsaad et al., 2013; De et al., 2018), Ferulic acid (Shi et al., 2016), p-Coumaric acid (Boz, 2015), 4-Hydroxybenzoic acid (Cho J.-Y. et al., 2014), 2,5-Dihydroxybenzoic acid (Kim et al., 2007), Chlorogenic acid (Li et al., 2013; Kabir et al., 2014), Ionone (Mikhlin et al., 1983), β-Carotene (Hayashi et al., 2012), and Ricinoleic acid (Novak et al., 1961) which were earlier reported from Alternanthera sessilis (L.) R.Br. ex DC. may be responsible for its antimicrobial property.
Antioxidant Activity
Petrus and the team had evaluated the antioxidant activity of the 80% aqueous methanolic extract obtained from the flowers of Alternanthera bettzickiana (Regel) G.Nicholson. They had observed that the extract possessed radical scavenging and ferrous ion chelating properties (Petrus A. et al., 2014). On the other hand, Vidhya and the team had evaluated the antioxidant activity of the hexane, chloroform, ethyl acetate, methanolic, and aqueous extracts obtained from the leaves Alternanthera bettzickiana (Regel) G.Nicholson. They observed that out of all, methanolic extract was exhibiting stronger radical scavenging activity (Vidhya et al., 2015). Phytomolecules like apigenin analogs (Prince Vijeya Singh et al., 2004) which were earlier reported from Alternanthera bettzickiana (Regel) G.Nicholson, may be responsible for this antioxidant potential.
Independently, several research teams had investigated the antioxidant potential of Alternanthera brasiliana (L.) Kuntze: Reza and the team had used 80% ethanolic extract of stem and leaves (Reza et al., 2019); Enechi and the team had used ethanolic extract of leaves (Enechi et al., 2013); Chandran R had used methanolic extract of leaves (Chandran, 2017); Attaugwu and Uvere had used ethanolic extract of leaves (Attaugwu and Uvere, 2017); Pereira and the team had used ethanolic extract and its dichloromethane, ethyl acetate, n-butanolic fractions of leaves (Pereira et al., 2013); Araujo and the team had used ethanolic extract of aerial parts and its hexane, chloroform, and ethyl acetate fractions (Araujo et al., 2014); while Akachukwu and Uchegbu had used ethanolic extract of leaves (Akachukwu and Uchegbu, 2016). These pieces of evidence increase the credibility of Alternanthera brasiliana (L.) Kuntze as antioxidant. Phytoconstituents like Ligustroflavone (Kang et al., 2021), Vitexin (An et al., 2012), Kaempferol (Park et al., 2006), Quercetin (Zhang et al., 2011), Tricin (Duarte-Almeida et al., 2007), Quercetin 3-β-D-glucoside (Niranjan Panat et al., 2015), Isorhamnetin-3-O-robinobioside (Boubaker et al., 2012), Stigmasterol (Liang et al., 2020), β-Sitosterol (Gupta et al., 2011), Ferulic acid (Graf, 1992), p-Coumaric acid (Kiliç and Yeşiloğlu, 2013), 4-Hydroxybenzoic acid (Velika and Kron, 2012), 2,5-Dihydroxybenzoic acid (Calderón Guzmán et al., 2007), Chlorogenic acid (Sato et al., 2011), Dopamine-betaxanthin (Cai et al., 2003), and 3-Methoxytyramine-betaxanthin (Cai et al., 2003) which were earlier reported from Alternanthera brasiliana (L.) Kuntze, may be responsible for its antioxidant property.
Patil and Kore had evaluated the antioxidant property of methanolic extracts obtained from different parts viz. leaves, stem, and roots of Alternanthera ficoidea (L.) P.Beauv. They had observed that out of all, the methanolic extract from the roots was having most potent antioxidant activity (Patil and Kore, 2019). To the best of our knowledge, the phytochemial characterization of Alternanthera ficoidea (L.) P.Beauv. was not yet done, leaving an ample scope for the researchers.
Koolen and the team had isolated seven phytoconstituents from the aerial sections of Alternanthera littoralis P.Beauv. and evaluated them for the antioxidant potential using In vitro—ORAC assay. They had observed that out of all compounds, Alternamide B was the most significant one as antioxidant. Researchers had further suggested the catechol scaffold as a pharmacophore for this activity (Koolen et al., 2017).
Two independent research teams had evaluated the antioxidant potential of Alternanthera paronychioides A.St.-Hil.: Wu and the team had used methanolic, ethanolic, and aqueous extracts of the whole plant (Wu et al., 2013) while Tukun and the team had used aqueous extract obtained from the leaves (Tukun et al., 2014). These preliminary studies signifies the role of Alternanthera paronychioides A.St.-Hil. as antioxidant. To the best of our knowledge, the phytochemial characterization of Alternanthera paronychioides A.St.-Hil. was not yet done, leaving an ample scope for the researchers.
Bhattacherjee and the team had evaluated the antioxidant activity of methanol soluble fraction obtained from the leaves of Alternanthera philoxeroides (Mart.) Griseb. (Bhattacherjee et al., 2014). while Correa and the team had used ethanolic extracts of the whole plant (Correa et al., 2016). These preliminary studies suggested that the Alternanthera philoxeroides (Mart.) Griseb. is worthy of further investigation as antioxidant. Phytomolecules like Luteolin and luteolin analogs (Romanova et al., 2001), Chrysoeriol analogs (Mishra et al., 2003), Vitexin (An et al., 2012), Quercetin (Zhang et al., 2011), β-Sitosterol (Gupta et al., 2011), Δ5-Stigmasterol (Liang et al., 2020), Ursolic acid (Bobé et al., 2012; do Nascimento et al., 2014), Oleanolic acid and Oleanolic acid analogs (Wang et al., 2010), Calenduloside E (Tang et al., 2019), Caffeic acid (Gulcin, 2006), Quinic acid (Pero et al., 2009), p-Coumaric acid (Kiliç and Yeşiloğlu, 2013), Rubiadin (Tripathi et al., 1997), β-Carboline (Moura et al., 2007), Malic acid (Jin et al., 2016), Azelaic acid (Muthulakshmi and Saravanan, 2013), Cycloeucalenol (Wang W. et al., 2015), Phytol (Santos et al., 2013), and Pheophytin A (Endo et al., 1985) which were previously been reported from Alternanthera philoxeroides (Mart.) Griseb., may be responsible for this antioxidant property.
Several research teams have independently assessed the antioxidant potential of Alternanthera pungens Kunth: Mourya and the team had used ethanolic and aqueous extracts obtained from the leaves (Mourya et al., 2019); Franck and the team had used aqueous extract of leaves (Franck et al., 2016); while Jakhar and Dahiya had used aqueous, acetone, ethanolic, and petroleum ether extracts of aerial parts (Jakhar and Dahiya, 2017). These studies validated the antioxidant potential of Alternanthera pungens Kunth. Various phytochemicals like Limonene (Roberto et al., 2009), Geraniol (Aytac et al., 2016), Linalool (Duarte et al., 2016), Camphor (Drikvandi et al., 2020), Myrcene (Khalili et al., 2020), Camphene (Tiwari and Kakkar, 2009), and α-pinene (Aydin et al., 2013) which were reported earlier from Alternanthera pungens Kunth, may be responsible for its antioxidant action.
While going through literature, we have found enough pieces of evidences reporting and validating the antioxidant property of Alternanthera sessilis (L.) R.Br. ex DC.: Borah and the team had used 90% methanolic, 70% acetone, 80% ethanolic extracts of leaves and stems (Borah et al., 2011); Chai and the team had used hexane, chloroform, ethyl acetate, butanolic, and aqueous fractions of leaves and callus methanol extracts (Chai et al., 2016); Sharma and the team 30% hydroethanolic extract of the whole plant (Sharma et al., 2013); Khan and the team had used separate Methanolic and hexane extracts of leaves and stems (Khan et al., 2018); Azizah and the team had used ethanolic and aqueous extracts of aerial parts (Azizah et al., 2015); Muniandy and the team had used 90% hydroethanolic extract of stem (Muniandy et al., 2018b); Othman and the team had used ethanolic and aqueous extracts of aerial parts (Othman et al., 2016); Tiwari and the team had used juice (Tiwari et al., 2013); Rajamurugan and the team had used ethanolic extract of leaves (Rajamurugan et al., 2013); Jain and the team had used methanolic extract of leaves (Jain et al., 2016); Suganya and the team had used aqueous extract of leaves and stems (Suganya et al., 2019); Mohd Hazli and the team had used hexane, ethyl acetate, ethanolic, and aqueous extracts of leaves and stem (Mohd Hazli et al., 2019); Niraimathi and the team had used silver nanoparticles from aqueous extract of leaves (Niraimathi et al., 2013); Yap and the team had used 100% ethanolic, 70% ethanolic, 80% methanolic, ethyl acetate, and aqueous extracts of the whole plant (Yap et al., 2019); Kota and the team had used petroleum ether, ethyl acetate, chloroform, and methanolic extract of leaves (Kota et al., 2017); Sundar and the team had used petroleum ether and methanolic extracts of leaves (Sundar et al., 2019); Pathak and the team had used n-hexane and methanolic extracts of aerial parts (Pathak et al., 2020); Khan and the team had used the volatile oil of leaves and flowers (Khan et al., 2016); while Salvador and the team had used ethanolic extract and its four fractions; Acacetin 8-c-[α-L-rhamnopyranoyl-(1→2)-β-D-glucopyranoside]; 2″-O-α-L-rhamnopyranosyl-vitexin; 2″-O-β-D-glucopyranosyl vitexin and Vitexin (Salvador et al., 2006). Results from these researches left no doubt in the credibility and applicability of Alternanthera sessilis (L.) R.Br. ex DC. in reducing oxidative stress. Phytomolecules like Vitexin and vitexin analogs (An et al., 2012), Kaempferol and kaempferol analogs (Park et al., 2006), Quercetin and quercetin analogs (Zhang et al., 2011), Acacetin analogs (Li et al., 2019), Isorhamnetin-3-O-robinobioside (Boubaker et al., 2012), Stigmasterol (Liang et al., 2020), Campesterol (Yoshida and Niki, 2003), β-Sitosterol (Gupta et al., 2011), Spinasterol (Adebiyi et al., 2018), Ellagic acid (Priyadarsini et al., 2002), Ferulic acid (Graf, 1992), p-Coumaric acid (Kiliç and Yeşiloğlu, 2013), 4-Hydroxybenzoic acid (Velika and Kron, 2012), 2,5-Dihydroxybenzoic acid (Calderón Guzmán et al., 2007), Chlorogenic acid (Sato et al., 2011), Ionone (Liu et al., 2009), β-Carotene (Paiva and Russell, 1999), Ricinoleic acid (Park et al., 2020), Dopamine-betaxanthin (Cai et al., 2003), and 3-Methoxytyramine-betaxanthin (Cai et al., 2003) which were earlier been reported from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible for its antioxidant action.
Antiparkinsonism/Antidementia Property
Khamphukdee and the team had evaluated the antidementia activity of the ethanolic extract obtained from the whole plant of Alternanthera philoxeroides (Mart.) Griseb. They had noticed various mechanisms behind it like inhibition of lipid peroxidation in the whole brain, downregulation of neuroinflammatory cytokines (IL-1β, IL-6, and TNF-α), etc (Khamphukdee et al., 2021). Phytomolecules like Luteolin and luteolin analogs (Delgado et al., 2021), Vitexin (Malar et al., 2020; Zhang et al., 2021), Quercetin (Yao et al., 2010), Torosaflavone E (Khamphukdee et al., 2021), Demethyl torosaflavone D (Khamphukdee et al., 2021), β-Sitosterol (Kim et al., 2008), Stigmasterol (Park S. J. et al., 2012; Pratiwi et al., 2021), Ursolic acid (Habtemariam, 2019), Oleanolic acid and oleanolic acid analogs (Lin et al., 2021), Caffeic acid (Khan et al., 2013; Deshmukh et al., 2016), Quinic acid (Liu et al., 2020), p-Coumaric acid (Kim H.-B. et al., 2017), β-Carboline (Zhao et al., 2013; Li et al., 2018), Malic acid (Tian et al., 2021), Blumenol A (Emir et al., 2019), Phytol (Sathya et al., 2020), and Pheophytin A (Park et al., 2014) which were earlier reported from Alternanthera philoxeroides (Mart.) Griseb., may be responsible for this antidementia property.
Ittiyavirah and Hameed had evaluated the antiparkinsonian activity of silver nanoparticles and ethanolic extract obtained from the whole plant of Alternanthera sessilis (L.) R.Br. ex DC. They had observed that the silver nanoparticles as well as the ethanolic extract were able to impart neuroprotection with decrease in catalepsy as well as in muscle rigidity, along with locomotion improvement (Ittiyavirah and Hameed, 2015). Phytomolecules like Vitexin and vitexin analogs (Hu et al., 2018), Kaempferol and kaempferol analogs (Filomeni et al., 2012), Quercetin-3-methyl ether (Kim et al., 2009), Quercetin (Lv et al., 2012), Acacetin analogs (Kim S. M. et al., 2017), Stigmasterol (Haque and Moon, 2018), β-Sitosterol (Kim et al., 2008), Spinasterol (Jeong et al., 2010), Ellagic acid (Baluchnejadmojarad et al., 2017), Ferulic acid (Haque et al., 2015), p-Coumaric acid (Vauzour et al., 2010), 4-Hydroxybenzoic acid (Winter et al., 2017), Chlorogenic acid (Singh et al., 2018), and Ionone (Ma et al., 2014) which were previously been reported from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible for the antiparkinsonian activity.
Antiprotozoal Activity
Koolen and the team had isolated compounds like Alternamide A-B and Alternamine A-B from the aerial parts of Alternanthera littoralis P.Beauv. and evaluated for their antiprotozoal activity againt protozoal strains viz. Trypanosoma cruzi trypomastigotes and Leishmania amazonensis. They had observed that out of all the tested compounds, Alternamine A was the most efficient one (Koolen et al., 2017).
Antispasmodic Activity
Garín-Aguilar and the team had antispasmodic activity of aqueous, hexane, methanolic extract, and fractions of methanol extract (F1-F6) obtained from the leaves of Alternanthera sessilis (L.) R.Br. ex DC. (Garín-Aguilar et al., 2013). while Saqib and Janbaz had used 70% ethanolic extract of the whole plant and its dichloromethane, aqueous fractions (Saqib and Janbaz, 2016). They had observed that Alternanthera sessilis (L.) R.Br. ex DC. possesses significant antispasmolytic activity. Phytomolecules like Vitexin and vitexin analogs (Ragone et al., 2007), Quercetin and quercetin analogs (Lozoya et al., 1994; Morales et al., 1994), Acacetin analogs (González-Trujano et al., 2012), Stigmasterol (Ammar et al., 2009), β-Sitosterol (Rehman et al., 2012), and Ellagic acid (Krenn et al., 2011) which were previously been reported from Alternanthera sessilis (L.) R.Br. ex DC., may be the contributors towards the antispasmodic activity of the extracts.
Antiviral Activity
Rattanathongkom and the team had isolated Chikusetsusaponin IVa isolated from the whole plant of Alternanthera philoxeroides (Mart.) Griseb. and evaluated antiviral activity against various viral cell lines through in vitro and in vivo assays. They had observed the dose-dependent activity along with the potential of Chikusetsusaponin IVa in inhibiting the viral protein synthesis (Rattanathongkom et al., 2009).
Central-Stimulating Activity
Mondal and the team had evaluated the central stimulating potential of the ethanolic extract obtained from the leaves of Alternanthera sessilis (L.) R.Br. ex DC. Results were quite significant (Mondal et al., 2014). Phytoconstituents acting on GABA receptors like Ricinoleic acid (Witt et al., 2002), Chlorogenic acid (Hara et al., 2014), p-Coumaric acid (Scheepens et al., 2014), Ferulic acid (Cheng et al., 2010; Sonar et al., 2019), Ellagic acid (Girish et al., 2013), Spinasterol (Socała et al., 2015), Stigmasterol (Karim et al., 2021), Acacetin analogs (Gálvez et al., 2015), Vitexin and vitexin analogs (Zhu et al., 2016; de Oliveira et al., 2020), and Quercetin and quercetin analogs (Goutman and Calvo, 2004; Kim et al., 2014) which were previously been reported from Alternanthera sessilis (L.) R.Br. ex DC., may be behind this GABA receptor mediated central-stimulating activity.
Gastrointestinal Protective Activity
Astudillo-Vázquez and the team had evaluated the gastrointestinal protective potential of the aqueous and ethanolic extracts obtained from the whole plant of Alternanthera sessilis (L.) R.Br. ex DC. They noticed that the antidiarrheal property i.e. decreasing the gastrointestinal content is the major factor behind the gastrointestinal protective activity of Alternanthera sessilis (L.) R.Br. ex DC. (Astudillo-Vázquez et al., 2008). Phytomolecules like Vitexin and vitexin analogs (Figer et al., 2017), Kaempferol and kaempferol analogs (Beber et al., 2017; Campos-Vidal et al., 2021), Quercetin and quercetin analogs (de la Lastra et al., 1994), Stigmasterol (Sánchez-Mendoza et al., 2008), β-Sitosterol (Sánchez-Mendoza et al., 2008), Ellagic acid (Beserra et al., 2011), Ferulic acid (Shahid et al., 2018), p-Coumaric acid (Panda and Suresh, 2015), Chlorogenic acid (Ahmed et al., 2021), and β-Carotene (Mózsik et al., 1996) which were earlier reported from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible for this gastrointestinal protective potential.
Hepatoprotective Activity
Lin and the team had evaluated the hepatoprotective activity of the aqueous extract obtained from the whole plant of Alternanthera sessilis (L.) R.Br. ex DC. (Lin et al., 1994). while Bhuyan and the team had evaluated the hepatoprotective potential of the methanolic extract obtained from the whole plant (Bhuyan et al., 2017). Both these independent researches finally concluded that the Alternanthera sessilis (L.) R.Br. ex DC. is hepatoprotective. Phytomolecules like Vitexin and vitexin analogs (Duan et al., 2020), Kaempferol and kaempferol analogs (Wang M. et al., 2015; Wang et al., 2015c), Quercetin-3-methyl ether (Tseng et al., 2012), Quercetin and quercetin analogs (Miltonprabu et al., 2017), Acacetin analogs (Cho H.-I. et al., 2014), Stigmasterol (Carter et al., 2007), β-Sitosterol (Abdou et al., 2019), Ellagic acid (Girish and Pradhan, 2012), Ferulic acid (Rukkumani et al., 2004), p-Coumaric acid (Parvizi et al., 2020), 2,5-Dihydroxybenzoic acid (Pujari and Bandawane, 2021), Chlorogenic acid (Chen et al., 2019), and β-Carotene (Manda and Bhatia, 2003) which were previously reported from Alternanthera sessilis (L.) R.Br. ex DC., may be the contributory constituents towards the elicited hepatoprotective activity.
Immunomodulatory Activity
Several research teams had independently assessed the immunomodulatory potential of Alternanthera sessilis (L.) R.Br. ex DC.: Biella and the team had used aqueous extract of the whole plant (Biella et al., 2008); Guerra and the team had used aqueous extract of aerial parts (Guerra et al., 2003); while Moraes and the team had used aqueous and ethanolic extract of leaves as well as tetrahydrofuran, dichloromethane, aqueous, petroleum ether soluble fraction (Moraes et al., 1994). These studies validated the immunomodulatory property of Alternanthera sessilis (L.) R.Br. ex DC. Phytomolecules like Vitexin and vitexin analogs (Rosa et al., 2016), Kaempferol and kaempferol analogs (Lin et al., 2011; Swarnalatha et al., 2015), Quercetin-3-methyl ether (Martino et al., 2016), Quercetin and quercetin analogs (Manjunath and Thimmulappa, 2021), Acacetin analogs (Zhao et al., 2014), Stigmasterol (Antwi et al., 2017b), β-Sitosterol (Desai et al., 2009), Ellagic acid (Abuelsaad et al., 2013), Ferulic acid (He F. et al., 2021), p-Coumaric acid (Pragasam et al., 2012), Chlorogenic acid (Guo et al., 2021), and β-Carotene (Jyonouchi et al., 2009) which were previously been reported from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible for this immunomodulatory potential.
Moraes and the team had also evaluated the immunomodulatory activity of aqueous and ethanolic extract of leaves as well as tetrahydrofuran, dichloromethane, aqueous, petroleum ether soluble fractions obtained from Alternanthera brasiliana (L.) Kuntze and Alternanthera littoralis P.Beauv. (Moraes et al., 1994). Phytomolecules like Vitexin and vitexin analogs (Rosa et al., 2016), Kaempferol and kaempferol analogs (Lin et al., 2011; Swarnalatha et al., 2015), Quercetin and quercetin analogs (Manjunath and Thimmulappa, 2021), Tricin (Santos et al., 2017), Stigmasterol (Antwi et al., 2017b), β-Sitosterol (Desai et al., 2009), Ferulic acid (He F. et al., 2021), p-Coumaric acid (Pragasam et al., 2012), and Chlorogenic acid (Guo et al., 2021) which were previously reported from Alternanthera brasiliana (L.) Kuntze, may be responsible towards its immunomodulatory activity. Phytomolecules like Vitexin and vitexin analogs (Rosa et al., 2016), Kaempferol (Lin et al., 2011; Swarnalatha et al., 2015), Quercetin-3-methyl ether (Martino et al., 2016), Quercetin and quercetin analogs (Manjunath and Thimmulappa, 2021), Acacetin analogs (Zhao et al., 2014), Stigmasterol (Antwi et al., 2017b), and Hydroxytyrosol (Shan and Miao, 2022) which were previously reported from Alternanthera littoralis P.Beauv., may be responsible for its immunomodulatory activity.
Insecticidal Property
Coutinho and the team had evaluated the insecticidal potential of the ethanolic extract obtained from the leaves of Alternanthera brasiliana (L.) Kuntze. against Drosophila melanogaster (Harwich strain). They found that the tested concentrations of the ethanolic extract were having a mild insecticidal effect, and that too after 24–48 h exposure (Coutinho et al., 2017). Phytomolecules like Kaempferol and kaempferol analogs (Zhang et al., 2016), Quercetin and quercetin analogs (Mesbah et al., 2007), Stigmasterol (Gade et al., 2017), β-Sitosterol (Zolotar et al., 2002), Spinasterol (Ahmed et al., 2020), and Ferulic acid (Yang et al., 2017) which were previously isolated from Alternanthera brasiliana (L.) Kuntze., may be responsible for this insecticidal property.
Lithotriptic/Antiurolithiatic Activity
Dhanya and the team had evaluated the antiurolithiatic activity of Kalka—fine paste of macerated fresh plant material of Alternanthera sessilis (L.) R.Br. ex DC. while Babu and the team had used ethanolic extract of the whole plant for the assessment of antiurolithiatic activity (Dhanya et al., 2017; Babu et al., 2021). Results obtained by both these independent studies are quite significant and reflects the potential of Alternanthera sessilis (L.) R.Br. ex DC. as lithotriptic agent. Phytomolecules like Kaempferol and kaempferol analogs (Cechinel-Zanchett et al., 2020), Quercetin and quercetin analogs (Dinnimath et al., 2017), Stigmasterol (Lobine et al., 2020), and Ferulic acid (Zhao et al., 2019) which were previously been reported from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible for this antiurolithiatic activity.
Larvicidal Activity
Babu and the team had also evaluated the larvicidal property of ethanolic extract obtained from the whole plant of Alternanthera sessilis (L.) R.Br. ex DC. They found that the ethanolic extract was having a dose dependent percent mortality against mosquito larvae (Babu et al., 2021). Phytomolecules like Stigmasterol (Gade et al., 2017), β-Sitosterol (Angajala and Subashini, 2018), and Ferulic acid (Pavela, 2011), which were earlier isolated from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible behind this larvicidal activity.
Nootropic Activity
Gupta and Singh had evaluated the nootropic activity of methanolic extract obtained from the leaves of Alternanthera sessilis (L.) R.Br. ex DC. And results were quite promising (Gupta and Singh, 2012b). Phytomolecules like Kaempferol and kaempferol analogs (Das et al., 2018), Quercetin and quercetin analogs (Halder et al., 2015), Ellagic acid (Bansal et al., 2017; Kiasalari et al., 2017), and Ferulic acid (Yang et al., 2016; Mhillaj et al., 2017) which had been previously isolated from Alternanthera sessilis (L.) R.Br. ex DC., may be the contributing phytomolecules towards this nootropic activity.
Photoprotective Activity
Alencar Filho and the team had evaluated the photoprotective effect of the gel prepared from 5% w/w of extract Alternanthera brasiliana (L.) Kuntze enriched in flavonoids. They had observed that the stabilization of the ROS and resonating permission are the mechanisms behind this photoprotective activity of the gel extract (Alencar Filho et al., 2020). Phytomolecules like Kaempferol and kaempferol analogs (Monici et al., 1994), Quercetin and quercetin analogs (Saija, 2003; Gonçalves et al., 2019), Tricin (Moon et al., 2018), Stigmasterol (Bayer et al., 2011), β-Sitosterol (Bayer et al., 2011), Ferulic acid (Lin et al., 2005; Peres et al., 2018), p-Coumaric acid (Biswas et al., 2021), and Chlorogenic acid (Wang et al., 2021) which were earlier reported from Alternanthera brasiliana (L.) Kuntze, may be responsible for this photoprotective property of the gel extract.
Sedative Property
Oyemitan and the team had evaluated the sedative action of the ethanolic extract obtained from the leaves of Alternanthera brasiliana (L.) Kuntze. They had observed that the ethanolic extract was expressing the sedative property by acting on stimulatory or central excitatory channels (Oyemitan et al., 2015). Phytomolecules like Quercetin and quercetin analogs (Nakhaee et al., 2021), β-Sitosterol (Aguirre-Hernández et al., 2007), and Ferulic acid (Tu et al., 2012) which were previously been reported from Alternanthera brasiliana (L.) Kuntze., may be responsible for this sedative action.
Wound Healing Property
Barua and the team had reported several studies validating the wound healing property of Alternanthera brasiliana (L.) Kuntze (Barua et al., 2009; Barua C. et al., 2012; Baru et al., 2012; Barua C. C. et al., 2012). Phytomolecules like Vitexin and vitexin analogs (Bektas et al., 2020), Kaempferol and kaempferol analogs (Petpiroon et al., 2015; Özay et al., 2019), Quercetin and quercetin analogs (Gomathi et al., 2003), Tricin (Han et al., 2016), β-Sitosterol (Abbas et al., 2019), Ferulic acid (Ghaisas et al., 2014), p-Coumaric acid (Kong et al., 2013; Boeing et al., 2020), and Chlorogenic acid (Bagdas et al., 2015) which had been isolated from Alternanthera brasiliana (L.) Kuntze previously, may be responsible for this wound healing property.
Muniandy and the team had evaluated the wound healing action of the 90% hydroethanolic extract obtained from the stem of Alternanthera sessilis (L.) R.Br. ex DC. while Jalalpure and the team had used chloroform extract obtained from the leaves Alternanthera sessilis (L.) R.Br. ex DC. Both these teams had independently ascertained the wound healing property of Alternanthera sessilis (L.) R.Br. ex DC. (Jalalpure et al., 2008; Muniandy et al., 2018b). Phytomolecules like Vitexin and vitexin analogs (Bektas et al., 2020), Kaempferol and kaempferol analogs (Petpiroon et al., 2015; Özay et al., 2019), Quercetin and quercetin analogs (Gomathi et al., 2003), Acacetin analogs (Bhat et al., 2013), β-Sitosterol (Abbas et al., 2019), Ellagic acid (Mo et al., 2014), Ferulic acid (Ghaisas et al., 2014), p-Coumaric acid (Kong et al., 2013; Boeing et al., 2020), and Chlorogenic acid (Bagdas et al., 2015), β-Carotene (Gerber and Erdman, 1982), and Ricinoleic acid (Nada et al., 2018) which had earlier reported from Alternanthera sessilis (L.) R.Br. ex DC., may be responsible for this wound healing property.
After this exhaustive cross-literature review for the bioactive compounds that may be responsible elements behind the potent pharmacological actions elicited by the extracts, we have summarized those in a smart interactive illustration (Figure 4).
FIGURE 4
It is indispensable to confirm if traditional claims of
Alternantheraspecies have been proven by systematic scientifically designed pharmacological (preclinical or clinical) studies. Traditional claims and reported pharmacological activities of various species are presented in
Table 3, and observations are as follows:
a) Traditional claims of some species (Alternanthera brasiliana (L.) Kuntze, Alternanthera caracasana Kunth, A. dentata (now reclaimed as Alternanthera brasiliana (L.) Kuntze), A. ficoides (now reclaimed as Alternanthera sessilis (L.) R.Br. ex DC.), Alternanthera littoralis P.Beauv., A. maritima (now reclaimed as Alternanthera littoralis P.Beauv.), Alternanthera nodiflora R.Br., Alternanthera paronychioides A.St.-Hil., Alternanthera porrigens (Jacq.) Kuntze, Alternanthera pungens Kunth, Alternanthera sessilis (L.) R.Br. ex DC., A. tenella (now reclaimed as Alternanthera sessilis (L.) R.Br. ex DC.), and A. triandra (now reclaimed as Alternanthera sessilis (L.) R.Br. ex DC.)) have not been validated scientifically.
b) Traditionally used species like Alternanthera caracasana Kunth and Alternanthera porrigens (Jacq.) Kuntze have not been investigated for any pharmacological activities. These species hold great potential for future research intending to validate traditional claims.
c) Species (Alternanthera brasiliana (L.) Kuntze, Alternanthera paronychioides A.St.-Hil., Alternanthera philoxeroides (Mart.) Griseb., and Alternanthera sessilis (L.) R.Br. ex DC.) have been screened for those pharmacological actions which are not claimed traditionally. These species may have been chosen following a chemotaxonomical or ecological approach.
d) Literature did not reveal any traditional use of three species (Alternanthera bettzickiana (Regel) G.Nicholson, Alternanthera hirtula (Mart.) R.E.Fr., and Alternanthera praelonga A.St.-Hil.) but evaluated for varied pharmacological activities.
TABLE 3
| Sr No | Species name | Traditional uses | Scientifically validated traditional claims | Traditional claims not validated scientifically | Other pharmacological activities |
|---|---|---|---|---|---|
| 1 | Alternanthera bettzickiana (Regel) G.Nicholson | — | — | — | Antibacterial, anticancer, antimicrobial, antioxidant |
| 2 | Alternanthera brasiliana (L.) Kuntze | In the treatment of headaches, cough, colds, grippe, fever, and diarrhea | Analgesic, antioxidant | Antidiarrhoeal, antipyretic | Allelopathic, antianxiety, antibacterial, anticancer, anticonvulsant, antifungal, anti-inflammatory, insecticide, sedative, and wound healing |
| 3 | Alternanthera caracasana Kunth | In the treatment of dysentery, diarrhea, and fever | — | Anti-dysentery, antidiarrhoeal, and antipyretic | — |
| 4 | Alternanthera dentata (Now reclaimed as Alternanthera brasiliana (L.) Kuntze) | In the treatment of inflammation, pain | — | Analgesic, anti-inflammatory | Antimicrobial, antioxidant |
| 5 | Alternanthera ficoidea (L.) P.Beauv | In the treatment of heart and cancer problems | Antioxidant | Anticancer, cardiotonic | — |
| 6 | Alternanthera hirtula (Mart.) R.E.Fr | — | — | — | Anticancer, antioxidant |
| 7 | Alternanthera littoralis P.Beauv | In the treatment of infectious and inflammatory diseases | Antioxidant | Anti-inflammatory | — |
| 8 | Alternanthera maritima (now reclaimed as Alternanthera littoralis P.Beauv.) | In the treatment of inflammation, viral infections, cancer, malaria, and diarrhea | Anti-inflammatory, antimicrobial | Antiviral, antidiarrhoeal, and anticancer | — |
| 9 | Alternanthera nodiflora R.Br | In the treatment of skin problems, degenerative and microbial infections | Antimicrobial | Skin protection | — |
| 10 | Alternanthera paronychioides A.St.-Hil | In the treatment of hyperuricemia, rheumatic arthritis, nephritis, gout, cystitis, diabetes, and systemic neuralgia | Antioxidant | Antihyperuricemia, antiarthritic, antigout, renal protective, antidiabetic, anti-inflammatory, and analgesic | Antiapoptotic |
| 11 | Alternanthera philoxeroides (Mart.) Griseb | In the treatment of influenza | Antioxidant, antiviral | — | α-glucosidase, inhibitory, analgesic, antianxiety, antiarthritic, anticancer, antidepressant, antidiabetic, anti-HBV, anti-inflammatory, antimicrobial |
| 12 | Alternanthera porrigens (Jacq.) Kuntze | In the treatment of hepatitis, kidney problems, influenza | — | Hepatoprotective, analgesic, antiviral, renal protective | — |
| 13 | Alternanthera praelonga A.St.-Hil | — | — | — | Anticancer, antioxidant |
| 14 | Alternanthera pungens Kunth | In the treatment of nasopharyngeal infections, pain, gonorrhea, menstrual disorder, dysentery, cholera, and many parasitic diseases | Anti-inflammatory, antimicrobial, antioxidant | Analgesic, anti- dysentery | — |
| 15 | Alternanthera repens (now reclaimed as Alternanthera sessilis (L.) R.Br. ex DC.) | — | — | — | Antibacterial, antidiarrhoeal, antispasmodic, gastrointestinal protective |
| 16 | Alternanthera sessilis (L.) R.Br. ex DC.) | In the treatment of stomach pain, ulcer, and gastric problems | Analgesic, antioxidant | Antiulcer, gastroprotective | α-glucosidase inhibitory, anthelmintic, anti-allergic, antiarthritic, antiasthmatic, antibacterial, anticancer, anticataract, antidepressant antidiabetic, antifungal, antihypertensive, anti-inflammatory, antimicrobial, anti-parkinsonism, hepatoprotective, nootropic, and wound healing |
| 17 | Alternanthera tenella (Now reclaimed as Alternanthera sessilis (L.) R.Br. ex DC.)) | In the treatment of urinary problems, fever, menstruation problem, inflammations, and ovarian diseases | Anti-inflammatory, antimicrobial, antioxidant | Renal protective, antipyretic | Immunomodulatory, inhibition of lymphocyte activation, and anticancer |
| 18 | Alternanthera triandra (Now reclaimed as Alternanthera sessilis (L.) R.Br. ex DC.)) | In the treatment of fever, lactation problem | — | Antipyretic | — |
Relationship between reported scientific pharmacological activities of Alternanthera species and their traditional claims.
Toxicological Studies
Hydroalcoholic extract of Alternanthera brasiliana (L.) Kuntze and Alternanthera bettzickiana (Regel) G.Nicholson leaves was orally administered (200 mg/kg dose) for 14 days in mice to observe any change in behavior of animals (Kasthuri and Ramesh, 2018). Further, hematological and histopathological changes were also observed. Sub-acute toxicity study suggested that both extracts samples did not show any harmful side effects. Hydroethanolic leaf extract of Alternanthera bettzickiana (Regel) G.Nicholson displayed a progressively powerful cytotoxic impact on DLA cell lines than Alternanthera brasiliana (L.) Kuntze extract.
The oral acute toxicity study was conducted on 95% ethanolic extract of Alternanthera philoxeroides (Mart.) Griseb. at the dose of 500 mg/kg in male and female rodents (Thanabhorn et al., 2005). The ethanolic extract did not show mortality and gross morphological alterations in the organs of rodents. Oral administration of 1,000 mg/kg/day for 14 days showed no significant changes in the body and inner organs weights, hematological and clinical parameters.
Clinical Studies
The studies have shown antiretroviral activity of Alternanthera pungens Kunth herbal tea due to antioxidant potential when administered to HIV patients (Djohan et al., 2009). Blood samples were taken from fasted patients who received an Alternanthera pungens Kunth tea for 12 months every day before dinner. The markers of oxidative stress (malondialdehyde and advanced oxidation protein end products), plasma T lymphocytes, transaminases, and creatinine were determined in the blood sample. A significant decrease in concentrations of markers of oxidative stress and an increase in plasma levels of CD4 and CD8 T cells after this period were observed. Further, no signs of hepatic and renal toxicity were seen in HIV patients.
In another case study, the potential of Alternanthera sessilis (L.) R.Br. ex DC., Momordica charantia L., and Colocasia esculenta (L.) Schott were investigated in reducing postprandial blood glucose levels in healthy human subjects and patients with type II diabetes (Bachok et al., 2014). The results of the clinical report suggested that Alternanthera sessilis (L.) R.Br. ex DC. reduced the non-significant glucose level in 3 h in comparison to standard control diet in healthy and diseased subjects. This case study was conducted in India with eight healthy subjects and six diabetic subjects.
Conclusion
Scrutiny of available literature reveals that out of 139 species of the genus
Alternanthera:a) Nine species have been investigated phytochemically,
b) Fifteen species possess strong ethnopharmacological records,
c) Twelve species have been scientifically evaluated in the in vitro or in vivo experimental models for various pharmacological activities,
d) Three species have been subjected to toxicity studies for establishing safety profiles,
e) Two species have been examined for clinical studies.
To date, 129 compounds have been isolated from 9 species of Alternanthera. 129 bioactive compounds were classified in 11 phytochemical classes, covering information about 40 flavonoids, 17 triterpenoid/saponins, 15 sterols, 12 alkaloids, 10 phenolic compounds, 3 ionone, 1 benzopyran, 3 hydroxycinnamic acids, 4 anthraquinone, 8 volatile oils and 17 miscellaneous compounds. Flavonoids (∼32%) constitute the main class of phytoconstituents in the genus Alternanthera followed by triterpenoids (∼13%). The isolated triterpenoids such as oleanolic acid, ursolic acid, and flavonoids such as luteolin, apigenin, vitexin, kaempferol, quercetin aglycones and their glycosides from the genus have proven therapeutic value. In terms of the phytochemical exploration, the most explored species of Alternanthera genus were Alternanthera philoxeroides (Mart.) Griseb. (52 compounds), Alternanthera sessilis (L.). R.Br. ex DC. (45 compounds), Alternanthera brasiliana (L.). Kuntze (32 compounds), and Alternanthera littoralis P.Beauv (24 compounds). Alternanthera sessilis (L.) R.Br. ex DC. has so far yielded a diverse class of compounds, like benzopyran, flavonoids, sterols, triterpenoid/saponin, phenolic compounds, ionone, and miscellaneous compounds. Similarly, Alternanthera philoxeroides (Mart.) Griseb. has also yielded a diverse class of compounds like flavonoids, sterols, triterpenoid/saponins, phenolic compounds, anthraquinone, alkaloids, and miscellaneous compounds.While volatile oil related compounds were extracted only from Alternanthera pungens Kunth, ionone analogues were isolated from Alternanthera sessilis (L.) R.Br. ex DC. only and hydroxycinnamic acids were reported only from Alternanthera bettzickiana (Regel) G.Nicholson. Researchers could explore rest of the species of Alternanthera genus to check if containing ionone analogues, volatile oils, and hydroxycinnamic acids. Further, the species of Alternanthera genus which were least explored in terms of phytochemical characterization is also leading for possible opportunities for the researchers.
To the best of our knowledge, the phytochemial characterization of Alternanthera paronychioides A.St.-Hil., Alternanthera caracasana Kunth, Alternanthera nodiflora R.Br., and Alternanthera ficoidea (L.) P.Beauv. was not yet done, leaving an ample scope for the researchers.
Some phytoconstituents like quercetin, vitexin, chlorogenic acid, kaempferol, ferulic acid, β-sitosterol, p-coumaric acid, caffeic acid, quinic acid, etc had been reported from more than one species of Alternanthera. Probably, we could say that these phytoconstituents may be common secondary metabolites in Alternanthera genus. So, we recommend the researchers to explore the rest of the Alternanthera species for these common metabolites. These metabolites could serve as biomarkers for them.
As twelve species of Alternanthera have been investigated scientifically for pharmacological activities, only 9 species of the genus have been explored phytochemically. Few medicinally promising Alternanthera species have not been taken into consideration for phytochemical studies. The existing literature demonstrates that 5 species of genus Alternanthera such as.
Alternanthera brasiliana (L.) Kuntze, Alternanthera caracasana Kunth, Alternanthera ficoidea (L.) P.Beauv., Alternanthera nodiflora R.Br., and Alternanthera paronychioides A.St.-Hil. have been scientifically reported to exhibit various pharmacological activities, but these species have never been subjected to bioactivity directed fractionation to isolate bioactive phytoconstituents using appropriate chromatographic techniques. Therefore, natural product scientists should expand their research activities on Alternanthera species to isolate more bioactive compounds which can be developed as safer and efficacious lead molecules or potent analogs of bioactive markers. Further, it seems necessary to mention a major research gap in phytochemical studies that no emphasis has been given to standardizing these plants based on marker compounds. Appropriate analytical methods need to be developed using HPLC, HPTLC, or LC-MS for the standardization of Alternanthera species. Molecular docking and QSAR studies on selective bioactive markers of these species are also lacking. It has been observed that crude uncharacterized extracts of Alternanthera species have been used in most pharmacological studies. This observation attracts attention towards the isolation of bioactive compounds from Alternanthera following the bioactivity-guided fractionation approach. Highlighting a mechanistic approach for pharmacological activities is another area of research to be covered. Alternamide A-B and Alternamine A-B were evaluated only for antiprotozoal activity while Chikusetsusaponin IVa was checked for antiviral activity only, leaving a wide scope for the researchers.
Amongst 139 species of Alternanthera, only 12 species have shown medicinal value in preclinical studies, and out of these only Alternanthera pungens Kunth and Alternanthera sessilis (L.) R.Br. ex DC. have been investigated clinically for antiretroviral and antidiabetic activities, respectively. The toxicity studies have been conducted on 3 species such as Alternanthera bettzickiana (Regel) G.Nicholson, Alternanthera brasiliana (L.) Kuntze, and Alternanthera philoxeroides (Mart.) Griseb. to establish their safety profile. Please be noted that as per the latest guidelines and recommendations of the ethnopharmacology team, the scientific names of the plants have been reassessed and considered the name given on https://mpns.science.kew.org/mpns-portal/. So the universally recognized name has been mentioned rather than the synonym indicated in the cited articles.
It is finally concluded that a well-planned roadmap of research activities is needed to be designed on traditionally used and medicinally promising plants of genus Alternanthera, so that their products and preparations may emerge out to be clinically potential and safe medicines in the treatment of various ailments.
Statements
Author contributions
RM and BS contributed to the conception and design of the study. RS, VD, DK, SB, MB, SK, AD, and SS wrote sections of the manuscript. All authors contributed to manuscript revision, read, and approved the submitted version.
Funding
This work was supported by the National Natural Science Foundation of China (32070671), the COVID-19 Research Projects of West China Hospital Sichuan University (Grant no. HX-2019-nCoV-057), and the Regional Innovation Cooperation between Sichuan and Guangxi Provinces (2020YFQ0019).
Acknowledgments
The authors acknowledge the financial support received from the National Natural Science Foundation of China, the West China Hospital Sichuan University, and the Regional Innovation Cooperation between Sichuan and Guangxi Provinces.
Conflict of interest
RS and SS are having honorary based association with iGlobal Research and Publishing Foundation, New Delhi India, who declare that there are no conflicts of interest.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Author disclaimer
The scientific name of plants was mentioned as per the universally accepted nomenclature, specified and recommended by the Ethnopharmacology team. So, the names specified in the manuscript will seems to be different from that of cited articles. To cross-check the nomenclature, refer https://mpns.science.kew.org/mpns-portal/.
References
1
AbbasM. M.Al-RawiN.AbbasM. A.Al-KhateebI. (2019). Naringenin Potentiated β-sitosterol Healing Effect on the Scratch Wound Assay. Res. Pharm. Sci.14 (6), 566–573. 10.4103/1735-5362.272565
2
AbdouE. M.FayedM. A. A.HelalD.AhmedK. A. (2019). Assessment of the Hepatoprotective Effect of Developed Lipid-Polymer Hybrid Nanoparticles (LPHNPs) Encapsulating Naturally Extracted β-Sitosterol against CCl4 Induced Hepatotoxicity in Rats. Sci. Rep.9 (1), 19779. 10.1038/s41598-019-56320-2
3
AbuelsaadA. S.MohamedI.AllamG.Al-SolumaniA. A. (2013). Antimicrobial and Immunomodulating Activities of Hesperidin and Ellagic Acid against Diarrheic Aeromonas Hydrophila in a Murine Model. Life Sci.93 (20), 714–722. 10.1016/j.lfs.2013.09.019
4
AdebiyiO. E.OlopadeJ. O.OlayemiF. O. (2018). Sodium Metavanadate Induced Cognitive Decline, Behavioral Impairments, Oxidative Stress and Down Regulation of Myelin Basic Protein in Mice hippocampus: Ameliorative Roles of β-spinasterol, and Stigmasterol. Brain Behav.8 (7), e01014. 10.1002/brb3.1014
5
AgraM. F.BarachoG. S.NuritK.BasílioI. J.CoelhoV. P. (2007). Medicinal and Poisonous Diversity of the flora of "Cariri Paraibano", Brazil. J. Ethnopharmacol111 (2), 383–395. 10.1016/j.jep.2006.12.007
6
Aguirre-HernándezE.Rosas-AcevedoH.Soto-HernándezM.MartínezA. L.MorenoJ.González-TrujanoM. E. (2007). Bioactivity-guided Isolation of Beta-Sitosterol and Some Fatty Acids as Active Compounds in the Anxiolytic and Sedative Effects of Tilia Americana Var. Mexicana. Planta Med.73 (11), 1148–1155. 10.1055/s-2007-981593
7
AhmadM.GilaniA.-U. -H.AftabK.AhmadV. U. (1993). Effects of Kaempferol-3-O-Rutinoside on Rat Blood Pressure. Phytother. Res.7 (4), 314–316. 10.1002/ptr.2650070411
8
AhmedM.QinP.JiM.AnR.GuoH.ShafiJ. (2020). Spinasterol, 22,23-Dihydrospinasterol and Fernenol from Citrullus Colocynthis L. With Aphicidal Activity against Cabbage Aphid Brevicoryne Brassicae L. Molecules25 (9), 1. 10.3390/molecules25092184
9
AhmedM. A. E.MohanadM.AhmedA. A. E.AboulhodaB. E.El-AwdanS. A. (2021). Mechanistic Insights into the Protective Effects of Chlorogenic Acid against Indomethacin-Induced Gastric Ulcer in Rats: Modulation of the Cross Talk between Autophagy and Apoptosis Signaling. Life Sci.275, 119370. 10.1016/j.lfs.2021.119370
10
AkachukwuD.UchegbuR. (2016). GC-MS, Antimicrobial and In Vitro Antioxidant Assay of the Leaf Extract of Alternanthera Dentata. Jamps11 (2), 1–7. 10.9734/jamps/2016/29855
11
AkbarM.AminA.KhalilT.IqbalM. S.NazirA.TaswarA. (2021). Antibacterial Activity of Alternanthera Philoxeroides (Mart.) Griseb. Against Bacterial Phytopathogens: Erwinia Carotovora, Ralstonia Solanacearum and Xanthomonas Axonopodis. Aj53 (1), 83–92. 10.26651/allelo.j/2021-53-1-1329
12
AlawodeT. T.LajideL.OlaleyeM.OwolabiB. (2021). Stigmasterol and β-Sitosterol: Antimicrobial Compounds in the Leaves of Icacina Trichantha Identified by GC-MS. Beni-suef Univ. J. Basic Appl. Sci.10 (1), 1. 10.1186/s43088-021-00170-3
13
Alencar FilhoJ. M. T.TeixeiraH. A. P.SampaioP. A.PereiraE. C. V.AmarizI. A. E.Rolim NetoP. J.et al (2019). Phytochemical Analysis in Alternanthera Brasiliana by LC-MS/MS and GC-MS. Nat. Prod. Res.34 (3), 429–433. 10.1080/14786419.2018.1533827
14
Alencar FilhoJ. M. T. d.SampaioP. A.CarvalhoI. S. d.GuimarãesA. L.AmarizI. A. e.PereiraE. C. V.et al (2020). Flavonoid Enriched Extract of Alternanthera Brasiliana with Photoprotective Effect: Formulation Development and Evaluation of Quality. Ind. Crops Prod.149, 112371. 10.1016/j.indcrop.2020.112371
15
AliH.DixitS.AliD.AlqahtaniS. M.AlkahtaniS.AlarifiS. (2015). Isolation and Evaluation of Anticancer Efficacy of Stigmasterol in a Mouse Model of DMBA-Induced Skin Carcinoma. Drug Des. Devel Ther.9, 2793–2800. 10.2147/dddt.S83514
16
AlshehriA. S.El-KottA. F.El-GerbedM. S. A.El-KenawyA. E.AlbadraniG. M.KhalifaH. S. (2021). Kaempferol Prevents Cadmium Chloride-Induced Liver Damage by Upregulating Nrf2 and Suppressing NF-Κb and Keap1. Environ. Sci. Pollut. Res.29, 13917–13929. 10.1007/s11356-021-16711-3
17
AmalanV.VijayakumarN.IndumathiD.RamakrishnanA. (2016). Antidiabetic and Antihyperlipidemic Activity of P-Coumaric Acid in Diabetic Rats, Role of Pancreatic GLUT 2: In Vivo Approach. Biomed. Pharmacother.84, 230–236. 10.1016/j.biopha.2016.09.039
18
AmmarS.EdziriH.MahjoubM. A.ChatterR.BouraouiA.MighriZ. (2009). Spasmolytic and Anti-inflammatory Effects of Constituents from Hertia Cheirifolia. Phytomedicine16 (12), 1156–1161. 10.1016/j.phymed.2009.03.012
19
AnF.YangG.TianJ.WangS. (2012). Antioxidant Effects of the Orientin and Vitexin in Trollius Chinensis Bunge in D-Galactose-Aged Mice. Neural Regen. Res.7 (33), 2565–2575. 10.3969/j.issn.1673-5374.2012.33.001
20
AngajalaG.SubashiniR. (2018). Evaluation of Larvicidal Potential of β-sitosterol Isolated from Indigenous Aegle Marmelos Correa Crude Leaf Extracts against Blood Feeding Parasites and its Binding Affinity Studies towards Sterol Carrier Protein. Biocatal. Agric. Biotechnol.16, 586–593. 10.1016/j.bcab.2018.10.005
21
AnithaR.KanimozhiS. (2012). Pharmacognostic Evaluation of Alternanthera Sessilis (L.) R.Br.Ex.DC. Pharmacognosy J.4 (28), 31–34. 10.5530/pj.2012.28.6
22
AnjaneyuluM.ChopraK. (2003). Quercetin, a Bioflavonoid, Attenuates thermal Hyperalgesia in a Mouse Model of Diabetic Neuropathic Pain. Prog. Neuropsychopharmacol. Biol. Psychiatry27 (6), 1001–1005. 10.1016/s0278-5846(03)00160-x
23
AntwiA. O.ObiriD. D.OsafoN.EsselL. B.ForkuoA. D.AtobigaC. (2018). Stigmasterol Alleviates Cutaneous Allergic Responses in Rodents. Biomed. Res. Int.2018. 10.1155/2018/3984068
24
AntwiA. O.ObiriD. D.OsafoN. (2017a). Stigmasterol Modulates Allergic Airway Inflammation in Guinea Pig Model of Ovalbumin-Induced Asthma. Mediators Inflamm.2017, 2953930–2954011. 10.1155/2017/2953930
25
AntwiA. O.ObiriD. D.OsafoN.ForkuoA. D.EsselL. B. (2017b). Stigmasterol Inhibits Lipopolysaccharide-Induced Innate Immune Responses in Murine Models. Int. Immunopharmacol53, 105–113. 10.1016/j.intimp.2017.10.018
26
ArabyanE.HakobyanA.HakobyanT.GrigoryanR.IzmailyanR.AvetisyanA.et al (2021). Flavonoid Library Screening Reveals Kaempferol as a Potential Antiviral Agent against African Swine Fever Virus. Front. Microbiol.12, 736780. 10.3389/fmicb.2021.736780
27
AraujoE. C. C.SilvaE. E. S.Alencar-FiJ. M. T.OliveiraA. P.GuimaraesA. L.Siqueira-FJ. A.et al (2014). Identification of Glycosil Flavones and Determination In Vitro of Antioxidant and Photoprotective Activities of Alternanthera Brasiliana L. Kuntze. Res. J. Phytochemistry8 (4), 148–154. 10.3923/rjphyto.2014.148.154
28
ArshadN.Zitterl-EglseerK.HasnainS.HessM. (2008). Effect of Peganum Harmala or its Beta-Carboline Alkaloids on Certain Antibiotic Resistant Strains of Bacteria and Protozoa from Poultry. Phytother Res.22 (11), 1533–1538. 10.1002/ptr.2528
29
ArulselvanP.GothaiS.MuniandyK.Mohd EsaN.SubbiahS. (2018). Anticancer Potential of Alternanthera Sessilis Extract on HT-29 Human colon Cancer Cells. Asian Pac. J. Trop. Biomed.8 (8), 394. 10.4103/2221-1691.239427
30
AseervathamG. S.SuryakalaU.DoulethunishaSundaramS.SundaramS.BoseP. C.SivasudhaT. (2016). Expression Pattern of NMDA Receptors Reveals Antiepileptic Potential of Apigenin 8-C-Glucoside and Chlorogenic Acid in Pilocarpine Induced Epileptic Mice. Biomed. Pharmacother.82, 54–64. 10.1016/j.biopha.2016.04.066
31
Astudillo-VázquezA.Dávalos ValleH.De JesúsL.HerreraG.NavarreteA. (2008). Investigation of Alternanthera Repens and Bidens Odorata on Gastrointestinal Disease. Fitoterapia79 (7-8), 577–580. 10.1016/j.fitote.2008.07.001
32
AttaugwuR. N.UvereP. O. (2017). Health Promoting Properties of Alternanthera Brasiliana Leaves and Hibiscus sabdariffa Calyces Used in Fortification of maize-Bambara Groundnut Malt and maize-cowpea Malt Complementary Foods. Food Res.1 (4), 133–139. 10.26656/fr.2017.4.058
33
AydinE.TürkezH.GeyikoğluF. (2013). Antioxidative, Anticancer and Genotoxic Properties of α-pinene on N2a Neuroblastoma Cells. Biologia68 (5), 1004–1009. 10.2478/s11756-013-0230-2
34
AytacZ.YildizZ. I.Kayaci-SenirmakF.San KeskinN. O.TekinayT.UyarT. (2016). Electrospinning of Polymer-free Cyclodextrin/geraniol-Inclusion Complex Nanofibers: Enhanced Shelf-Life of Geraniol with Antibacterial and Antioxidant Properties. RSC Adv.6 (52), 46089–46099. 10.1039/c6ra07088d
35
AzizahO.AminI.FouadA. R. (2015). Antioxidant Properties ofAlternanthera Sessilisred and green. Acta Hortic.1106, 131–136. 10.17660/ActaHortic.2015.1106.20
36
BabuM.JosephK. H.SreeA.ScariyaS. (2021). In-Vitro Evaluation of Anti-urolithiatic and Larvicidal Activity of Alternanthera Sessilis. Biomed. Pharmacol. J.14 (02), 671–680. 10.13005/bpj/2169
37
BabukumarS.VinothkumarV.SankaranarayananC.SrinivasanS. (2017). Geraniol, a Natural Monoterpene, Ameliorates Hyperglycemia by Attenuating the Key Enzymes of Carbohydrate Metabolism in Streptozotocin-Induced Diabetic Rats. Pharm. Biol.55 (1), 1442–1449. 10.1080/13880209.2017.1301494
38
BachokM. F.YusofB. N.IsmailA.HamidA. A. (2014). Effectiveness of Traditional Malaysian Vegetables (Ulam) in Modulating Blood Glucose Levels. Asia Pac. J. Clin. Nutr.23 (3), 369–376. 10.6133/apjcn.2014.23.3.01
39
BaeH.ParkS.YangC.SongG.LimW. (2021). Disruption of Endoplasmic Reticulum and ROS Production in Human Ovarian Cancer by Campesterol. Antioxidants10 (3), 1. 10.3390/antiox10030379
40
BagdasD.EtozB. C.GulZ.ZiyanokS.InanS.TuracozenO.et al (2015). In Vivo systemic Chlorogenic Acid Therapy under Diabetic Conditions: Wound Healing Effects and Cytotoxicity/genotoxicity Profile. Food Chem. Toxicol.81, 54–61. 10.1016/j.fct.2015.04.001
41
BaluchnejadmojaradT.RabieeN.ZabihnejadS.RoghaniM. (2017). Ellagic Acid Exerts Protective Effect in Intrastriatal 6-hydroxydopamine Rat Model of Parkinson’s Disease: Possible Involvement of ERβ/Nrf2/HO-1 Signaling. Brain Res.1662, 23–30. 10.1016/j.brainres.2017.02.021
42
BankarG. R.NayakP. G.BansalP.PaulP.PaiK. S. R.SinglaR. K.et al (2011). Vasorelaxant and Antihypertensive Effect of Cocos Nucifera Linn. Endocarp on Isolated Rat Thoracic Aorta and DOCA Salt-Induced Hypertensive Rats. J. Ethnopharmacology134 (1), 50–54. 10.1016/j.jep.2010.11.047
43
BansalN.YadavP.KumarM. (2017). Ellagic Acid Administration Negated the Development of Streptozotocin-Induced Memory Deficit in Rats. Drug Res.67 (07), 425–431. 10.1055/s-0043-108552
44
BarahuieF.SaifullahB.DornianiD.FakuraziS.KarthivashanG.HusseinM. Z.et al (2017). Graphene Oxide as a Nanocarrier for Controlled Release and Targeted Delivery of an Anticancer Active Agent, Chlorogenic Acid. Mater. Sci. Eng. C74, 177–185. 10.1016/j.msec.2016.11.114
45
BaricevicD.SosaS.Della LoggiaR.TubaroA.SimonovskaB.KrasnaA.et al (2001). Topical Anti-inflammatory Activity of Salvia Officinalis L. Leaves: the Relevance of Ursolic Acid. J. Ethnopharmacology75 (2-3), 125–132. 10.1016/s0378-8741(00)00396-2
46
BaruC. C.TalukdarA.BegumS. A.BuragohainB.RoyJ. D.PathakD. C.et al (2012). Effect of Alternanthera Brasiliana (L) Kuntze on Healing of Dermal Burn Wound. Indian J. Exp. Biol.50 (1), 56–60.
47
BaruaC.BegumS.SarmaD.PathakD.BorahR. (2012a). Healing Efficacy of Methanol Extract of Leaves of Alternanthera Brasiliana Kuntze in Aged Wound Model. J. Basic Clin. Pharm.3 (4), 1. 10.4103/0976-0105.105336
48
BaruaC. C.Ara BegumS.TalukdarA.Datta RoyJ.BuragohainB.Chandra PathakD.et al (2012b). Influence of Alternanthera Brasiliana (L.) Kuntze on Altered Antioxidant Enzyme Profile during Cutaneous Wound Healing in Immunocompromised Rats. ISRN Pharmacol.2012, 1–8. 10.5402/2012/948792
49
BaruaC. C.BegumS. A.BaruaA. G.BorahR. S.LahkarM. (2013). Anxiolytic and Anticonvulsant Activity of Methanol Extract of Leaves of Alternanthera Brasiliana (L.) Kuntze (Amaranthaceae) in Laboratory Animals. Indian J. Exp. Biol.51 (6), 450–457.
50
BaruaC. C.TalukdarA.BegumS. A.SarmaD. K.FathakD. C.BaruaA. G.et al (2009). Wound Healing Activity of Methanolic Extract of Leaves of Alternanthera Brasiliana Kuntz Using In Vivo and In Vitro Model. Indian J. Exp. Biol.47 (12), 1001–1005.
51
BayerM.ProkschP.FelsnerI.BrendenH.KohneZ.WalliR.et al (2011). Photoprotection against UVAR: Effective Triterpenoids Require a Lipid Raft Stabilizing Chemical Structure. Exp. Dermatol.20 (11), 955–958. 10.1111/j.1600-0625.2011.01350.x
52
BeberA. P.de SouzaP.BoeingT.SomensiL. B.MarianoL. N. B.CuryB. J.et al (2017). Constituents of Leaves from Bauhinia Curvula Benth. Exert Gastroprotective Activity in Rodents: Role of Quercitrin and Kaempferol. Inflammopharmacology26 (2), 539–550. 10.1007/s10787-017-0313-8
53
BektasN.ŞenelB.YenilmezE.ÖzatikO.ArslanR. (2020). Evaluation of Wound Healing Effect of Chitosan-Based Gel Formulation Containing Vitexin. Saudi Pharm. J.28 (1), 87–94. 10.1016/j.jsps.2019.11.008
54
BerkbanT.BoonpromP.BunbuphaS.WelbatJ.KukongviriyapanU.KukongviriyapanV.et al (2015). Ellagic Acid Prevents L-NAME-Induced Hypertension via Restoration of eNOS and P47phox Expression in Rats. Nutrients7 (7), 5265–5280. 10.3390/nu7075222
55
BeserraA. M. S. e. S.CalegariP. I.SouzaM. d. C.dos SantosR. A. N.LimaJ. C. d. S.SilvaR. M.et al (2011). Gastroprotective and Ulcer-Healing Mechanisms of Ellagic Acid in Experimental Rats. J. Agric. Food Chem.59 (13), 6957–6965. 10.1021/jf2003267
56
BhatT. A.NambiarD.TailorD.PalA.AgarwalR.SinghR. P. (2013). Acacetin Inhibits In Vitro and In Vivo Angiogenesis and Downregulates Stat Signaling and VEGF Expression. Cancer Prev. Res.6 (10), 1128–1139. 10.1158/1940-6207.Capr-13-0209
57
BhattacherjeeA.GhoshT.SilR.DattaA. (2014). Isolation and Characterisation of Methanol-Soluble Fraction of Alternanthera Philoxeroides (Mart.) – Evaluation of Their Antioxidant, α-glucosidase Inhibitory and Antimicrobial Activity in In Vitro Systems. Nat. Product. Res.28 (23), 2199–2202. 10.1080/14786419.2014.930857
58
BhuyanB.BaishyaK.RajakP. (2017). Effects of Alternanthera Sessilis on Liver Function in Carbon Tetra Chloride Induced Hepatotoxicity in Wister Rat Model. Indian J. Clin. Biochem.33 (2), 190–195. 10.1007/s12291-017-0666-1
59
BiellaC. d. A.SalvadorM. J.DiasD. A.Dias-BaruffiM.Pereira-CrottL. S. (2008). Evaluation of Immunomodulatory and Anti-inflammatory Effects and Phytochemical Screening of Alternanthera Tenella Colla (Amaranthaceae) Aqueous Extracts. Memórias do Instituto Oswaldo Cruz103 (6), 569–577. 10.1590/s0074-02762008000600010
60
BinangK.TakuwaD. T. (2021). Development of Reverse Phase-High Performance Liquid Chromatography (RP-HPLC) Method for Determination of Selected Antihypertensive Active Flavonoids (Rutin, Myricetin, Quercetin, and Kaempferol) in Medicinal Plants Found in Botswana. Phys. Sci. Rev.0 (0), 1. 10.1515/psr-2020-0209
61
BindoliA.ValenteM.CavalliniL. (1985). Inhibitory Action of Quercetin on Xanthine Oxidase and Xanthine Dehydrogenase Activity. Pharmacol. Res. Commun.17 (9), 831–839. 10.1016/0031-6989(85)90041-4
62
BisignanoG.TomainoA.CascioR. L.CrisafiG.UccellaN.SaijaA. (1999). On the In-Vitro Antimicrobial Activity of Oleuropein and Hydroxytyrosol. J. Pharm. Pharmacol.51 (8), 971–974. 10.1211/0022357991773258
63
BiswasS.MukherjeeP. K.KarA.BannerjeeS.JanaS. N.HaldarP. K.et al (2021). Enhanced Permeability and Photoprotective Potential of Optimized P-Coumaric Acid-Phospholipid Complex Loaded Gel against UVA Mediated Oxidative Stress. J. Photochem. Photobiol. B: Biol.221. 10.1016/j.jphotobiol.2021.112246
64
BobéP.CheckerR.SandurS. K.SharmaD.PatwardhanR. S.JayakumarS.et al (2012). Potent Anti-inflammatory Activity of Ursolic Acid, a Triterpenoid Antioxidant, Is Mediated through Suppression of NF-Κb, AP-1 and NF-AT. PLoS ONE7 (2), 1. 10.1371/journal.pone.0031318
65
BoeingT.CostaP.VenzonL.MeurerM.MarianoL. N. B.FrançaT. C. S.et al (2020). Gastric Healing Effect of P-Coumaric Acid Isolated from Baccharis Dracunculifolia DC on Animal Model. Naunyn-Schmiedeberg's Arch. Pharmacol.394 (1), 49–57. 10.1007/s00210-020-01928-9
66
BorahA.YadavR. N. S.UnniB. G. (2011). In Vitro antioxidant and Free Radical Scavenging Activity of Alternanthera Sessilis. Int. J. Pharm. Sci. Res.2 (6), 1502–1506. 10.13040/IJPSR.0975-8232.2(6).1502-06
67
BorgesD. G. L.de AraújoM. A.CarolloC. A.CarolloA. R. H.LifschitzA.CondeM. H.et al (2020). Combination of Quercetin and Ivermectin: In Vitro and In Vivo Effects against Haemonchus contortus. Acta Tropica201. 10.1016/j.actatropica.2019.105213
68
BoubakerJ.SghaierM. B.SkandraniI.GhediraK.Chekir-GhediraL. (2012). Isorhamnetin 3-O-Robinobioside from Nitraria Retusa Leaves Enhance Antioxidant and Antigenotoxic Activity in Human Chronic Myelogenous Leukemia Cell Line K562. BMC Complement. Altern. Med.12 (1), 1. 10.1186/1472-6882-12-135
69
BozH. (2015). p-Coumaric Acid in Cereals: Presence, Antioxidant and Antimicrobial Effects. Int. J. Food Sci. Tech.50 (11), 2323–2328. 10.1111/ijfs.12898
70
BrochadoC. d. O.AlmeidaA. P. d.BarretoB. P.CostaL. P.RibeiroL. S.PereiraR. L. d. C.et al (2003). Flavonol Robinobiosides and Rutinosides from Alternanthera Brasiliana (Amaranthaceae) and Their Effects on Lymphocyte Proliferation In Vitro. J. Braz. Chem. Soc.14 (3), 449–451. 10.1590/s0103-50532003000300018
71
BurkillH. M. (1985). The Useful Plants of West Tropical Africa. Kew, UK: Royal Botanic Gardens.
72
CaiY.SunM.CorkeH. (2003). Antioxidant Activity of Betalains from Plants of the Amaranthaceae. J. Agric. Food Chem.51 (8), 2288–2294. 10.1021/jf030045u
73
Calderón GuzmánD.Trujillo JiménezF.Hernández GarcíaE.Juárez OlguínH. (2007). Assessment of Antioxidant Effect of 2,5-Dihydroxybenzoic Acid and Vitamin A in Brains of Rats with Induced Hyperoxia. Neurochem. Res.32 (6), 1036–1040. 10.1007/s11064-006-9269-6
74
Campos-VidalY.Herrera-RuizM.Trejo-TapiaG.Gonzalez-CortazarM.AparicioA. J.ZamilpaA. (2021). Gastroprotective Activity of Kaempferol Glycosides from Malvaviscus Arboreus Cav. J. Ethnopharmacology268. 10.1016/j.jep.2020.113633
75
CanÖ. D.Demir ÖzkayÜ.ÜçelU. İ. (2013). Anti-depressant-like Effect of Vitexin in BALB/c Mice and Evidence for the Involvement of Monoaminergic Mechanisms. Eur. J. Pharmacol.699 (1-3), 250–257. 10.1016/j.ejphar.2012.10.017
76
Canales-MartínezM.Hernández-DelgadoT.Flores-OrtizC.Durán-DíazA.García-BoresA. M.Avila-AcevedoG. (2008). Antimicrobial Activity of Alternanthera Caracasana. Pharm. Biol.43 (4), 305–307. 10.1080/13880200590951685
77
CarterB. A.TaylorO. A.PrendergastD. R.ZimmermanT. L.Von FurstenbergR.MooreD. D.et al (2007). Stigmasterol, a Soy Lipid–Derived Phytosterol, Is an Antagonist of the Bile Acid Nuclear Receptor FXR. Pediatr. Res.62 (3), 301–306. 10.1203/PDR.0b013e3181256492
78
Cechinel-ZanchettC. C.Bolda MarianoL. N.BoeingT.da CostaJ. d. C.Da SilvaL. M.BastosJ. K.et al (2020). Diuretic and Renal Protective Effect of Kaempferol 3-O-Alpha-L-Rhamnoside (Afzelin) in Normotensive and Hypertensive Rats. J. Nat. Prod.83 (6), 1980–1989. 10.1021/acs.jnatprod.0c00274
79
ChaiT. T.KhooC. S.TeeC. S.WongF. C. (2016). Alpha-glucosidase Inhibitory and Antioxidant Potential of Antidiabetic Herb Alternanthera Sessilis: Comparative Analyses of Leaf and Callus Solvent Fractions. Pharmacogn Mag.12 (48), 253–258. 10.4103/0973-1296.192202
80
ChandranR. P. (2017). Analysis of Proximate, Phytochemical, Elemental Compositions and Antioxidant Property of Leaf of Alternanthera Brasiliana (L.) Kuntze. MOJ Food Process. Tech.4 (3), 1. 10.15406/mojfpt.2017.04.00090
81
ChandrashekharK. (2019). Ethnobotanical and Phyto-Pharmacological Overview of Matsyakshi (Alternanthera Sessilis R. Br. Ex DC.). J. Ayu. Her. Med.5 (4), 152–155.
82
Chaves-QuirósC.Usuga-UsugaJ.Morales-UchimaS.Tofiño-RiveraA.Tobón-ArroyaveS.Martínez-PabónM. (2020). Assessment of Cytotoxic and Antimicrobial Activities of Two Components of Cymbopogon Citratus Essential Oil. J. Clin. Exp. Dentistry12, e749–e754. 10.4317/jced.56863
83
ChenJ.LiY.YuB.ChenD.MaoX.ZhengP.et al (2018). Dietary Chlorogenic Acid Improves Growth Performance of Weaned Pigs through Maintaining Antioxidant Capacity and Intestinal Digestion and Absorption Function. J. Anim. Sci.96 (3), 1108–1118. 10.1093/jas/skx078
84
ChenJ.LinD.ZhangC.LiG.ZhangN.RuanL.et al (2014). Antidepressant-like Effects of Ferulic Acid: Involvement of Serotonergic and Norepinergic Systems. Metab. Brain Dis.30 (1), 129–136. 10.1007/s11011-014-9635-z
85
ChenJ.YangH.ShengZ. (2020). Ellagic Acid Activated PPAR Signaling Pathway to Protect Ileums against Castor Oil-Induced Diarrhea in Mice: Application of Transcriptome Analysis in Drug Screening. Front. Pharmacol.10. 10.3389/fphar.2019.01681
86
ChenZ.YangY.MiS.FanQ.SunX.DengB.et al (2019). Hepatoprotective Effect of Chlorogenic Acid against Chronic Liver Injury in Inflammatory Rats. J. Funct. Foods62. 10.1016/j.jff.2019.103540
87
ChengC.-y.SuS.-y.TangN.-y.HoT.-y.LoW.-y.HsiehC.-l. (2010). Ferulic Acid Inhibits Nitric Oxide-Induced Apoptosis by Enhancing GABAB1 Receptor Expression in Transient Focal Cerebral Ischemia in Rats. Acta Pharmacologica Sinica31 (8), 889–899. 10.1038/aps.2010.66
88
ChiangH.-C.ChenY.-Y. (2008). Xanthine Oxidase Inhibitors from the Roots of Eggplant (Solanum MelongenaL.). J. Enzyme Inhib.7 (3), 225–235. 10.3109/14756369309040765
89
ChiruvellaK. K.MohammedA.DampuriG.GhantaR. G.RaghavanS. C. (2007). Phytochemical and Antimicrobial Studies of Methyl Angolensate and Luteolin-7-O-Glucoside Isolated from Callus Cultures of Soymida Febrifuga. Int. J. Biomed. Sci.3 (4), 269–278.
90
ChoH.-I.ParkJ.-H.ChoiH.-S.KwakJ. H.LeeD.-U.LeeS. K.et al (2014a). Protective Mechanisms of Acacetin against D-Galactosamine and Lipopolysaccharide-Induced Fulminant Hepatic Failure in Mice. J. Nat. Prod.77 (11), 2497–2503. 10.1021/np500537x
91
ChoJ.-Y.MoonJ.-H.SeongK.-Y.ParkK.-H. (2014b). Antimicrobial Activity of 4-Hydroxybenzoic Acid Andtrans4-Hydroxycinnamic Acid Isolated and Identified from Rice Hull. Biosci. Biotechnol. Biochem.62 (11), 2273–2276. 10.1271/bbb.62.2273
92
ChongS.LohK. E. (2020). Xanthine Oxidase Inhibitory Activity of Methanolic Extract of Alternanthera Sessilis. Sains Malaysiana49 (2), 405–410. 10.17576/jsm-2020-4902-19
93
CloeckaertA.KovačJ.ŠimunovićK.WuZ.KlančnikA.BucarF.et al (2015). Antibiotic Resistance Modulation and Modes of Action of (-)-α-Pinene in Campylobacter Jejuni. Plos One10 (4), 1. 10.1371/journal.pone.0122871
94
CollettM. G.TaylorS. M. (2019). Photosensitising Toxins in alligator weed (Alternanthera Philoxeroides) Likely to Be Anthraquinones. Toxicon167, 172–173. 10.1016/j.toxicon.2019.06.218
95
CollinsM. A.CharlesH. P. (1987). Antimicrobial Activity of Carnosol and Ursolic Acid: Two Anti-oxidant Constituents of Rosmarinus Officinalis L. Food Microbiol.4 (4), 311–315. 10.1016/s0740-0020(87)80005-9
96
CorbettS.DanielJ.DraytonR.FieldM.SteinhardtR.GarrettN. (2010). Evaluation of the Anti-inflammatory Effects of Ellagic Acid. J. PeriAnesthesia Nurs.25 (4), 214–220. 10.1016/j.jopan.2010.05.011
97
CorreaW. R.Hernandez TascoA. J.MarinhoJ. V. N. (2016). Antioxidant and Cytotoxic Activities and Chemical Profile of Five Amaranthaceae Plants Collected in the South of Brazil. Nat. Prod. Chem. Res.4 (5), 1–7. 10.4172/2329-6836.1000230
98
CoutinhoH. D. M.de Morais Oliveira-TintinoC. D.TintinoS. R.PereiraR. L. S.de FreitasT. S.da SilvaM. A. P.et al (2017). Toxicity against Drosophila melanogaster and Antiedematogenic and Antimicrobial Activities of Alternanthera Brasiliana (L.) Kuntze (Amaranthaceae). Environ. Sci. Pollut. Res.25 (11), 10353–10361. 10.1007/s11356-017-9366-x
99
da CunhaF. M.DumaD.AssreuyJ.BuzziF. C.NieroR.CamposM. M.et al (2009). Caffeic Acid Derivatives: In Vitro and In Vivo Anti-inflammatory Properties. Free Radic. Res.38 (11), 1241–1253. 10.1080/10715760400016139
100
DasD.BiswalS.BarhwalK. K.ChaurasiaO. P.HotaS. K. (2018). Kaempferol Inhibits Extra-synaptic NMDAR-Mediated Downregulation of TRkβ in Rat Hippocampus during Hypoxia. Neuroscience392, 77–91. 10.1016/j.neuroscience.2018.09.018
101
DasM. C.SandhuP.GuptaP.RudrapaulP.DeU. C.TribediP.et al (2016). Attenuation of Pseudomonas aeruginosa Biofilm Formation by Vitexin: A Combinatorial Study with Azithromycin and Gentamicin. Scientific Rep.6 (1), 1. 10.1038/srep23347
102
DasM.KumarA. D.MastanaiahK.DasA. (2015). Evaluation of Anti-diabetic Activity of Ethanolic Extract of Alternanthera Sessilis Linn. In Streptozotocin-Induced Diabetic Rats. Int. J. Pharma Sci. Res.6 (7), 1027–1032.
103
de la LastraA.oacuteMartinM. J.MotilvaV. (1994). Antiulcer and Gastroprotective Effects of Quercetin: A Gross and Histologic Study. Pharmacology48 (1), 56–62. 10.1159/000139162
104
de OliveiraD. D.da SilvaC. P.IglesiasB. B.BeleboniR. O. (2020). Vitexin Possesses Anticonvulsant and Anxiolytic-like Effects in Murine Animal Models. Front. Pharmacol.11. 10.3389/fphar.2020.01181
105
DeR.SarkarA.GhoshP.GangulyM.KarmakarB. C.SahaD. R.et al (2018). Antimicrobial Activity of Ellagic Acid against Helicobacter pylori Isolates from India and during Infections in Mice. J. Antimicrob. Chemother.73 (6), 1595–1603. 10.1093/jac/dky079
106
De RuizR. E.FuscoM.RuizS. O. (1993). Constituents of Alternanthera Pungens. Fitoterapia64, 95–99.
107
de Santana AquinoD. F.PiccinelliA. C.SoaresF. L. P.ArenaA. C.SalvadorM. J.KassuyaC. A. L. (2015). Anti-hyperalgesic and Anti-inflammatory Activity of Alternanthera Maritima Extract and 2″-O-α-L-Rhamnopyranosylvitexin in Mice. Inflammation38 (6), 2057–2066. 10.1007/s10753-015-0187-0
108
DeepakM.DipankarG.PrashanthD.AshaM. K.AmitA.VenkataramanB. V. (2002). Tribulosin and β-sitosterol-D-glucoside, the Anthelmintic Principles of Tribulus Terrestris. Phytomedicine9 (8), 753–756. 10.1078/094471102321621395
109
del ValleP.García-ArmestoM. R.de ArriagaD.González-DonquilesC.Rodríguez-FernándezP.RúaJ. (2016). Antimicrobial Activity of Kaempferol and Resveratrol in Binary Combinations with Parabens or Propyl Gallate against Enterococcus faecalis. Food Control61, 213–220. 10.1016/j.foodcont.2015.10.001
110
DeladinoL.AlvarezI.De AncosB.Sánchez-MorenoC.Molina-GarcíaA. D.Schneider TeixeiraA. (2017). Betalains and Phenolic Compounds of Leaves and Stems of Alternanthera Brasiliana and Alternanthera Tenella. Food Res. Int.97, 240–249. 10.1016/j.foodres.2017.04.017
111
DelgadoA.CholevasC.TheoharidesT. C. (2021). Neuroinflammation in Alzheimer's Disease and Beneficial Action of Luteolin. BioFactors47 (2), 207–217. 10.1002/biof.1714
112
DesaiF.RamanathanM.FinkC. S.WildingG. E.Weinstock-GuttmanB.AwadA. B. (2009). Comparison of the Immunomodulatory Effects of the Plant Sterol β-sitosterol to Simvastatin in Peripheral Blood Cells from Multiple Sclerosis Patients. Int. Immunopharmacology9 (1), 153–157. 10.1016/j.intimp.2008.10.019
113
DeshmukhR.KaundalM.BansalV.Samardeep (2016). Caffeic Acid Attenuates Oxidative Stress, Learning and Memory Deficit in Intra-cerebroventricular Streptozotocin Induced Experimental Dementia in Rats. Biomed. Pharmacother.81, 56–62. 10.1016/j.biopha.2016.03.017
114
DeviK. P.MalarD. S.NabaviS. F.SuredaA.XiaoJ.NabaviS. M.et al (2015). Kaempferol and Inflammation: From Chemistry to Medicine. Pharmacol. Res.99, 1–10. 10.1016/j.phrs.2015.05.002
115
DhanyaV.JollykuttyE.PremlalS. (2017). Lithotriptic Effect of Combination of Matsyakshi (Alternanthera Sessilis Linn. R.Br.,) and Tender Coconut Water in Albino Rats. Int. Res. J. Pharm.8 (9), 56–64. 10.7897/2230-8407.089158
116
DharP.ChanP.CohenD. T.KhawamF.GibbonsS.Snyder-LeibyT.et al (2014). Synthesis, Antimicrobial Evaluation, and Structure–Activity Relationship of α-Pinene Derivatives. J. Agric. Food Chem.62 (16), 3548–3552. 10.1021/jf403586t
117
DingK.TanY. Y.DingY.FangY.YangX.FangJ.et al (2018). β‐Sitosterol Improves Experimental Colitis in Mice with a Target against Pathogenic Bacteria. J. Cell Biochem.120 (4), 5687–5694. 10.1002/jcb.27853
118
DinnimathB. M.JalalpureS. S.PatilU. K. (2017). Antiurolithiatic Activity of Natural Constituents Isolated from Aerva Lanata. J. Ayurveda Integr. Med.8 (4), 226–232. 10.1016/j.jaim.2016.11.006
119
DjohanY. F.CamaraC.MondéA. A.KoffiG.NiamkéG.DéréL.et al (2009). Intérêt des antioxydants dans la prise en charge des patients infectés par le VIH: apport de la consommation régulière de tisane d’Alternanthera pungens. Ann. de biologie clinique67 (5), 563–568. 10.1684/abc.2009.0362
120
do NascimentoP.LemosT.BizerraA.ArriagaÂ.FerreiraD.SantiagoG.et al (2014). Antibacterial and Antioxidant Activities of Ursolic Acid and Derivatives. Molecules19 (1), 1317–1327. 10.3390/molecules19011317
121
DrikvandiP.BahramikiaS.AlirezaeiM. (2020). Modulation of the Antioxidant Defense System in Liver, Kidney, and Pancreas Tissues of Alloxan‐induced Diabetic Rats by Camphor. J. Food Biochem.44 (12), 1. 10.1111/jfbc.13527
122
DuanS.DuX.ChenS.LiangJ.HuangS.HouS.et al (2020). Effect of Vitexin on Alleviating Liver Inflammation in a Dextran Sulfate Sodium (DSS)-induced Colitis Model. Biomed. Pharmacother.121. 10.1016/j.biopha.2019.109683
123
DuarteA.LuísÂ.OleastroM.DominguesF. C. (2016). Antioxidant Properties of Coriander Essential Oil and Linalool and Their Potential to Control Campylobacter Spp. Food Control61, 115–122. 10.1016/j.foodcont.2015.09.033
124
Duarte-AlmeidaJ. M.NegriG.SalatinoA.de CarvalhoJ. E.LajoloF. M. (2007). Antiproliferative and Antioxidant Activities of a Tricin Acylated Glycoside from Sugarcane (Saccharum Officinarum) Juice. Phytochemistry68 (8), 1165–1171. 10.1016/j.phytochem.2007.01.015
125
Ehrnhöfer-ResslerM. M.FrickeK.PignitterM.WalkerJ. M.WalkerJ.RychlikM.et al (2013). Identification of 1,8-Cineole, Borneol, Camphor, and Thujone as Anti-inflammatory Compounds in a Salvia Officinalis L. Infusion Using Human Gingival Fibroblasts. J. Agric. Food Chem.61 (14), 3451–3459. 10.1021/jf305472t
126
EmirC.EmirA.BozkurtB.SomerN. U. (2019). Phytochemical Constituents from Galanthus Alpinus Sosn. Var. Alpinus and Their Anticholinesterase Activities. South Afr. J. Bot.121, 63–67. 10.1016/j.sajb.2018.10.021
127
EndoY.UsukiR.KanedaT. (1985). Antioxidant Effects of Chlorophyll and Pheophytin on the Autoxidation of Oils in the Dark. I. Comparison of the Inhibitory Effects. J. Am. Oil Chemists' Soc.62 (9), 1375–1378. 10.1007/bf02545962
128
EnechiO. C.OdoC. E.WuaveC. P. (2013). Evaluation of the In Vitro Anti-oxidant Activity of Alternanthera Brasiliana Leaves. J. Pharm. Res.6 (9), 919–924. 10.1016/j.jopr.2013.09.006
129
FacundoV. A.AzevedoM. S.RodriguesR. V.NascimentoL. F. d.MilitãoJ. S. L. T.SilvaG. V. J. d.et al (2012). Chemical Constituents from Three Medicinal Plants: Piper Renitens, Siparuna Guianensis and Alternanthera Brasiliana. Revista Brasileira de Farmacognosia22 (5), 1134–1139. 10.1590/s0102-695x2012005000040
130
FanW.-Q. (2008). Chemical Constituents of Alternanthera philoxeroides</I>. Chin. J. Nat. Medicines6 (2), 112–115. 10.3724/sp.J.1009.2008.00112
131
FangJ.-B.JiaW.GaoW.-Y.YaoZ.TengJ.ZhaoA.-H.et al (2007). Antitumor Constituents from Alternanthera Philoxeroides. J. Asian Nat. Prod. Res.9 (6), 511–515. 10.1080/10286020600782231
132
FangJ.-B.YaoZ.ChenJ.-C.LiuY.-W.TakaishiY.DuanH.-Q. (2009a). Cytotoxic Triterpene Saponins fromAlternanthera Philoxeroides. J. Asian Nat. Prod. Res.11 (3), 261–266. 10.1080/10286020802684656
133
FangJ. B.DuanH. Q.ZhangY. W.YoshihisaT. (2006). Chemical Constituents from Herb of Alternanthera Philoxeroides. Zhongguo Zhong Yao Za Zhi31 (13), 1072–1075.
134
FangJ.ChenJ.LiuY.DuanH. (2009b). Constituents from Alternanthera Philoxeroides and Their Antitumor Activity. Zhongguo Zhong Yao Za Zhi34 (19), 2473–2476.
135
FangX.-K.GaoJ.ZhuD.-N. (2008). Kaempferol and Quercetin Isolated from Euonymus Alatus Improve Glucose Uptake of 3T3-L1 Cells without Adipogenesis Activity. Life Sci.82 (11-12), 615–622. 10.1016/j.lfs.2007.12.021
136
FathimaS. N.SalwaA.AnushaS.FatimaS. (2016). Study of Antiasthmatic Activity of Ethanolic Extract of Alternanthera Sessilis Leaves. Int. J. Pharma Res. Health Sci.4 (6), 1478–1482.
137
FatimaN.HafizurR. M.HameedA.AhmedS.NisarM.KabirN. (2015). Ellagic Acid in Emblica Officinalis Exerts Anti-diabetic Activity through the Action on β-cells of Pancreas. Eur. J. Nutr.56 (2), 591–601. 10.1007/s00394-015-1103-y
138
FekaP. D.MohammedS. Y.ShaibuM. A.SolomonR. S. (2014). Phytochemical Screening and Antimicrobial Efficacy of <i>Alternanthera Nodiflora Extracts. Bayero J. Pure Appl. Sci.6 (2), 1. 10.4314/bajopas.v6i2.20
139
FigerB.PissurlenkarR.AmbreP.KalekarS.MunshiR.GatneM.et al (2017). Treatment of Gastric Ulcers with Fenugreek Seed Extract; In Vitro, In Vivo and In Silico Approaches. Indian J. Pharm. Sci.79 (5), 1. 10.4172/pharmaceutical-sciences.1000285
140
FilomeniG.GrazianiI.De ZioD.DiniL.CentonzeD.RotilioG.et al (2012). Neuroprotection of Kaempferol by Autophagy in Models of Rotenone-Mediated Acute Toxicity: Possible Implications for Parkinson's Disease. Neurobiol. Aging33 (4), 767–785. 10.1016/j.neurobiolaging.2010.05.021
141
FirdhouseM. J.LalithaP. (2013). Biosynthesis of Silver Nanoparticles Using the Extract of Alternanthera Sessilis—Antiproliferative Effect against Prostate Cancer Cells. Cancer Nanotechnology4 (6), 137–143. 10.1007/s12645-013-0045-4
142
FortunatoL. R.AlvesC. d. F.TeixeiraM. M.RogerioA. P. (2012). Quercetin: a Flavonoid with the Potential to Treat Asthma. Braz. J. Pharm. Sci.48 (4), 589–599. 10.1590/s1984-82502012000400002
143
FranckA. M.MassaraC. C.InnocentK. K.FrancoisM. G.GogahyK.AbsalomeM. A.et al (2016). Phytochemical Screening, Anti-inflammatory and Antioxidant Effects of Aqueous Extract of Alternanthera Pungens (Amaranthaceae) in Rats. AJBBL5 (1), 1–10.
144
GadeS.RajamanikyamM.VadlapudiV.NukalaK. M.AluvalaR.GiddigariC.et al (2017). Acetylcholinesterase Inhibitory Activity of Stigmasterol & Hexacosanol Is Responsible for Larvicidal and Repellent Properties of Chromolaena Odorata. Biochim. Biophys. Acta (Bba) - Gen. Subjects1861 (3), 541–550. 10.1016/j.bbagen.2016.11.044
145
GálvezJ.Estrada-ReyesR.Benítez-KingG.AraujoG.OrozcoS.Fernández-MasR.et al (2015). Involvement of the GABAergic System in the Neuroprotective and Sedative Effects of Acacetin 7-O-Glucoside in Rodents. Restorative Neurol. Neurosci.33 (5), 683–700. 10.3233/rnn-140486
146
GamaroG. D.SuyenagaE.BorsoiM.LermenJ.PereiraP.ArdenghiP. (2011). Effect of Rosmarinic and Caffeic Acids on Inflammatory and Nociception Process in Rats. ISRN Pharmacol.2011, 1–6. 10.5402/2011/451682
147
GaoY.ChenX.FangL.LiuF.CaiR.PengC.et al (2014). Rhein Exerts Pro- and Anti-inflammatory Actions by Targeting IKKβ Inhibition in LPS-Activated Macrophages. Free Radic. Biol. Med.72, 104–112. 10.1016/j.freeradbiomed.2014.04.001
148
Garín-AguilarM. E.Benavides-CatalánD.Segura CobosD.Ramírez SoteloG.Piña GuzmánA. B.Valencia-del ToroG. (2013). Spasmolytic Effect ofAlternanthera Repenson Isolated Rat Ileum. Pharm. Biol.52 (4), 479–485. 10.3109/13880209.2013.844716
149
GasparettoA.LapinskiT. F.ZamunerS. R.KhouriS.AlvesL. P.MuninE.et al (2010). Extracts from Alternanthera Maritima as Natural Photosensitizers in Photodynamic Antimicrobial Chemotherapy (PACT). J. Photochem. Photobiol. B: Biol.99 (1), 15–20. 10.1016/j.jphotobiol.2010.01.009
150
GattoM. T.FalcocchioS.GrippaE.MazzantiG.BattinelliL.NicolosiG.et al (2002). Antimicrobial and Anti-lipase Activity of Quercetin and its C2-C16 3-O-Acyl-Esters. Bioorg. Med. Chem.10 (2), 269–272. 10.1016/s0968-0896(01)00275-9
151
GerberL. E.ErdmanJ. W. (1982). Effect of Dietary Retinyl Acetate, β-Carotene and Retinoic Acid on Wound Healing in Rats. J. Nutr.112 (8), 1555–1564. 10.1093/jn/112.8.1555
152
GhaisasM. M.KshirsagarS. B.SahaneR. S. (2014). Evaluation of Wound Healing Activity of Ferulic Acid in Diabetic Rats. Int. Wound J.11 (5), 523–532. 10.1111/j.1742-481X.2012.01119.x
153
GirishC.PradhanS. C. (2012). Hepatoprotective Activities of Picroliv, Curcumin, and Ellagic Acid Compared to Silymarin on Carbon-Tetrachloride-Induced Liver Toxicity in Mice. J. Pharmacol. Pharmacother.3 (2), 149–155. 10.4103/0976-500X.95515
154
GirishC.RajV.AryaJ.BalakrishnanS. (2013). Involvement of the GABAergic System in the Anxiolytic-like Effect of the Flavonoid Ellagic Acid in Mice. Eur. J. Pharmacol.710 (1-3), 49–58. 10.1016/j.ejphar.2013.04.003
155
GomathiK.GopinathD.Rafiuddin AhmedM.JayakumarR. (2003). Quercetin Incorporated Collagen Matrices for Dermal Wound Healing Processes in Rat. Biomaterials24 (16), 2767–2772. 10.1016/s0142-9612(03)00059-0
156
Gómez-MorenoG.Aguilar-SalvatierraA.GuardiaJ.Uribe-MarioniA.Cabrera-AyalaM.Delgado-RuizR. A.et al (2013). The Efficacy of a Topical Sialogogue Spray Containing 1% Malic Acid in Patients with Antidepressant-Induced Dry Mouth: A Double-Blind, Randomized Clinical Trial. Depress. Anxiety30 (2), 137–142. 10.1002/da.22017
157
GongJ.-H.ShinD.HanS.-Y.KimJ.-L.KangY.-H. (2012). Kaempferol Suppresses Eosionphil Infiltration and Airway Inflammation in Airway Epithelial Cells and in Mice with Allergic Asthma. J. Nutr.142 (1), 47–56. 10.3945/jn.111.150748
158
González-TrujanoM.Ventura-MartínezR.ChávezM.Díaz-RevalI.PellicerF. (2012). Spasmolytic and Antinociceptive Activities of Ursolic Acid and Acacetin Identified in Agastache Mexicana. Planta Med.78 (08), 793–796. 10.1055/s-0031-1298416
159
GonçalvesM.SantosV.TaylorJ.PerasoliF.SantosO.RabeloA.et al (2019). Preparation and Characterization of a Quercetin-Tetraethyl Ether-Based Photoprotective Nanoemulsion. Química Nova. 10.21577/0100-4042.20170345
160
GoutmanJ. D.CalvoD. J. (2004). Studies on the Mechanisms of Action of Picrotoxin, Quercetin and Pregnanolone at the GABAρ1 Receptor. Br. J. Pharmacol.141 (4), 717–727. 10.1038/sj.bjp.0705657
161
GrafE. (1992). Antioxidant Potential of Ferulic Acid. Free Radic. Biol. Med.13 (4), 435–448. 10.1016/0891-5849(92)90184-i
162
GrønhaugT. E.GlæserudS.SkogsrudM.BalloN.BahS.DialloD.et al (2008). Ethnopharmacological Survey of Six Medicinal Plants from Mali, West-Africa. J. Ethnobiol. Ethnomedicine4 (1), 1. 10.1186/1746-4269-4-26
163
GuedeN. Z.N’guessanK.DibiéT. E.GrellierP. (2010). Ethnopharmacological Study of Plants Used to Treat Malaria, in Traditional Medicine, by Bete Populations of Issia. J. Pharm. Sci. Res.2 (4), 216–227.
164
GuerraR. N. M.PereiraH. A. W.SilveiraL. M. S.OleaR. S. G. (2003). Immunomodulatory Properties of Alternanthera Tenella Colla Aqueous Extracts in Mice. Braz. J. Med. Biol. Res.36 (9), 1215–1219. 10.1590/s0100-879x2003000900011
165
GulcinI. (2006). Antioxidant Activity of Caffeic Acid (3,4-dihydroxycinnamic Acid). Toxicology217 (2-3), 213–220. 10.1016/j.tox.2005.09.011
166
GuoC.BiJ.LiX.LyuJ.LiuX.LiuJ.et al (2021). Effects of Isomerisation and Oxidation on the Immunomodulatory Activity of Chlorogenic Acid in RAW264.7 Macrophages. Int. J. Food Sci. Technol.. 10.1111/ijfs.15442
167
GuoQ. L.LiB.LiJ.LiJ. J.XiaL. Y.DongJ. X. (2011). Triterpenoid Saponins of Alternanthera Philoxeroides (Mart.) Griseb. Yao Xue Xue Bao46 (4), 428–431.
168
GuptaH. C.RajJ.RathiA.SundaramE. N.ManchandaR. K. (2012). Morpho-anatomy of Leaf, Stem and Root of Alternanthera Sessilis (L.) R. Br. Ex DC and Alternanthera Pungens Kunth (Amaranthaceae) and its Significance in Drug Identification. Indian J. Res. Homoeopathy6, 1–7.
169
GuptaR. K.SaxenaV. K. (1987). Volatile Constituents from the Flowers of Althernanthera Pungens HBK (Amaranthaceae). Indian Perfumer31, 366–369.
170
GuptaR.SharmaA. K.DobhalM. P.SharmaM. C.GuptaR. S. (2011). Antidiabetic and Antioxidant Potential of β-sitosterol in Streptozotocin-Induced Experimental Hyperglycemia. J. Diabetes3 (1), 29–37. 10.1111/j.1753-0407.2010.00107.x
171
GuptaR.SinghH. K. (2012a). Detection and Quantitation of SS-Sitosterol in Clerodendrum Infortunatum and Alternanthera Sessilis by HPTLC. Pharmacognosy Commun.2 (1), 31–36. 10.5530/pc.2012.1.6
172
GuptaR.SinghH. K. (2012b). Nootropic Potential of Alternanthera Sessilis and Clerodendrum Infortunatum Leaves on Mice. Asian Pac. J. Trop. Dis.2, S465–S470. 10.1016/s2222-1808(12)60204-7
173
GuptaR.SinghK. H. (2014). Antidepressant like Effects of Alternanthera Sessilis and Clerodendrum Infortunatum Leaves Extract in Immobility Models. Nat. Prod. J.4 (1), 33–37. 10.2174/2210315504666140515004826
174
HabtemariamS. (2019). Antioxidant and Anti-inflammatory Mechanisms of Neuroprotection by Ursolic Acid: Addressing Brain Injury, Cerebral Ischemia, Cognition Deficit, Anxiety, and Depression. Oxidative Med. Cell Longevity2019, 1–18. 10.1155/2019/8512048
175
HachlafiN. E. L.AannizT.MenyiyN. E.BaabouaA. E.OmariN. E.BalahbibA.et al (2021). In Vitro and In Vivo Biological Investigations of Camphene and its Mechanism Insights: A Review. Food Rev. Int.28, 1–28. 10.1080/87559129.2021.1936007
176
HalderS.KarR.GalavV.MehtaA. K.BhattacharyaS. K.MedirattaP. K.et al (2015). Cadmium Exposure during Lactation Causes Learning and Memory-Impairment in F1 Generation Mice: Amelioration by Quercetin. Drug Chem. Toxicol.39 (3), 272–278. 10.3109/01480545.2015.1092042
177
HanJ. M.KwonH. J.JungH. J. (2016). Tricin, 4′,5,7-Trihydroxy-3′,5′-Dimethoxyflavone, Exhibits Potent Antiangiogenic Activity In Vitro. Int. J. Oncol.49 (4), 1497–1504. 10.3892/ijo.2016.3645
178
HaqueE.JavedH.AzimullahS.Abul KhairS. B.OjhaS. (2015). Neuroprotective Potential of Ferulic Acid in the Rotenone Model of Parkinson’s Disease. Drug Des. Dev. Ther.9, 5499–5510. 10.2147/dddt.S90616
179
HaqueM. N.MoonI. S. (2018). Stigmasterol Upregulates Immediate Early Genes and Promotes Neuronal Cytoarchitecture in Primary Hippocampal Neurons as Revealed by Transcriptome Analysis. Phytomedicine46, 164–175. 10.1016/j.phymed.2018.04.012
180
HaraK.HaranishiY.KataokaK.TakahashiY.TeradaT.NakamuraM.et al (2014). Chlorogenic Acid Administered Intrathecally Alleviates Mechanical and Cold Hyperalgesia in a Rat Neuropathic Pain Model. Eur. J. Pharmacol.723, 459–464. 10.1016/j.ejphar.2013.10.046
181
HaroonH. B.PerumalsamyV.NairG.AnandD. K.KolliR.MonichenJ.et al (2020). Repression of Polyol Pathway Activity by Hemidesmus Indicus Var. Pubescens R.Br. Linn Root Extract, an Aldose Reductase Inhibitor: An In Silico and Ex Vivo Study. Nat. Prod. Bioprospecting11 (3), 315–324. 10.1007/s13659-020-00290-w
182
HassanzadehP.ArbabiE.AtyabiF.DinarvandR. (2017). Ferulic Acid Exhibits Antiepileptogenic Effect and Prevents Oxidative Stress and Cognitive Impairment in the Kindling Model of Epilepsy. Life Sci.179, 9–14. 10.1016/j.lfs.2016.08.011
183
HayashiM.NaknukoolS.HayakawaS.OgawaM.Ni’matulahA.-B. A. (2012). Enhancement of Antimicrobial Activity of a Lactoperoxidase System by Carrot Extract and β-carotene. Food Chem.130 (3), 541–546. 10.1016/j.foodchem.2011.07.067
184
HeF.ChouC. J.ScheinerM.PoetaE.Yuan ChenN.GuneschS.et al (2021a). Melatonin- and Ferulic Acid-Based HDAC6 Selective Inhibitors Exhibit Pronounced Immunomodulatory Effects In Vitro and Neuroprotective Effects in a Pharmacological Alzheimer’s Disease Mouse Model. J. Med. Chem.64 (7), 3794–3812. 10.1021/acs.jmedchem.0c01940
185
HeY.ChenS.TsoiB.QiS.GuB.WangZ.et al (2021b). Alpinia Oxyphylla Miq. And its Active Compound P-Coumaric Acid Promote Brain-Derived Neurotrophic Factor Signaling for Inducing Hippocampal Neurogenesis and Improving Post-cerebral Ischemic Spatial Cognitive Functions. Front. Cel Dev. Biol.8. 10.3389/fcell.2020.577790
186
HeenanP. B.de LangeP. J.KeelingJ. (2009). Alternanthera Nahui, a New Species of Amaranthaceae Indigenous to New Zealand. New Zealand J. Bot.47 (1), 97–105. 10.1080/00288250909509795
187
HoriuchiK.ShiotaS.HatanoT.YoshidaT.KurodaT.TsuchiyaT. (2007). Antimicrobial Activity of Oleanolic Acid from Salvia Officinalis and Related Compounds on Vancomycin-Resistant Enterococci (VRE). Biol. Pharm. Bull.30 (6), 1147–1149. 10.1248/bpb.30.1147
188
HosamaniK. M.GanjihalS. S.ChavadiD. V. (2004). Alternanthera Triandra Seed Oil: A Moderate Source of Ricinoleic Acid and its Possible Industrial Utilisation. Ind. Crops Prod.19 (2), 133–136. 10.1016/j.indcrop.2003.07.009
189
HossainA. I.FaisalM.RahmanS.JahanR.RahmatullahM. (2014). A Preliminary Evaluation of Antihyperglycemic and Analgesic Activity of Alternanthera Sessilis Aerial Parts. BMC Complement. Altern. Med.14, 169. 10.1186/1472-6882-14-169
190
HuM.LiF.WangW. (2018). Vitexin Protects Dopaminergic Neurons in MPTP-Induced Parkinson’s Disease through PI3K/Akt Signaling Pathway. Drug Des. Dev. Ther.12, 565–573. 10.2147/dddt.S156920
191
HuR.WuS.LiB.TanJ.YanJ.WangY.et al (2021). Dietary Ferulic Acid and Vanillic Acid on Inflammation, Gut Barrier Function and Growth Performance in Lipopolysaccharide-Challenged Piglets. Anim. Nutr.. 10.1016/j.aninu.2021.06.009
192
HulmeA. C. (1953). The Isolation of Chlorogenic Acid from the Apple Fruit. Biochem. J.53 (3), 337–340. 10.1042/bj0530337
193
HundiwaleJ. C.PatilA. V.KulkarniM. V.PatilD. A.MaliR. G. (2012). A Current Update on Phytopharmacology of the Genus Alternanthera. J. Pharm. Res.5 (4), 1924–1929.
194
HungY. C.KuoY. H.HsiehP. W.HsiehT. Y.KuoJ. R.WangS. J. (2021). Chlorogenic Acid Decreases Glutamate Release from Rat Cortical Nerve Terminals by P/Q-Type Ca(2+) Channel Suppression: A Possible Neuroprotective Mechanism. Int. J. Mol. Sci.22 (21), 1. 10.3390/ijms222111447
195
HwangS. J.KimY.-W.ParkY.LeeH.-J.KimK.-W. (2013). Anti-inflammatory Effects of Chlorogenic Acid in Lipopolysaccharide-Stimulated RAW 264.7 Cells. Inflamm. Res.63 (1), 81–90. 10.1007/s00011-013-0674-4
196
IbitoyeO. B.UwazieJ. N.AjiboyeT. O. (2018). Bioactivity-guided Isolation of Kaempferol as the Antidiabetic Principle from Cucumis Sativus L. Fruits. J. Food Biochem.42 (4), 1. 10.1111/jfbc.12479
197
ImranM.Aslam GondalT.AtifM.ShahbazM.Batool QaisaraniT.Hanif MughalM.et al (2020). Apigenin as an Anticancer Agent. Phytotherapy Res.34 (8), 1812–1828. 10.1002/ptr.6647
198
ImranM.SalehiB.Sharifi-RadJ.Aslam GondalT.SaeedF.ImranA.et al (2019). Kaempferol: A Key Emphasis to its Anticancer Potential. Molecules24 (12), 1. 10.3390/molecules24122277
199
IttiyavirahS. P.HameedJ. (2015). Protective Role of Alternanthera Sessilis (Linn.) Silver Nanoparticles and its Ethanolic Extract against Rotenone Induced Parkinsonism. IOSR J. Pharm. Biol. Sci.10 (5), 25–32.
200
JainA.RoyS.Joshi*A.JoshiN. (2016). Evaluation of In-Vitro Cytotoxic and Antioxidant Activity of Methanolic Extracts of Ipomoea Carnea and Alternanthera Sessilis. Int. J. Bioassays5 (08), 1. 10.21746/ijbio.2016.08.008
201
JakharS.DahiyaP. (2017). Antimicrobial, Antioxidant and Phytochemical Potential of Alternanthera Pungens HB&K. J. Pharm. Sci. Res.9 (8), 1305–1311.
202
JalalpureS. S.AgrawalN.PatilM. B.ChimkodeR.TripathiA. (2008). Antimicrobial and Wound Healing Activities of Leaves of Alternanthera Sessilis Linn. Int. J. Green Pharm.2 (3), 141–144. 10.4103/0973-8258.42729
203
JangY.-H.ParkJ.-R.KimK.-M. (2020). Antimicrobial Activity of Chrysoeriol 7 and Chochlioquinone 9, White-Backed Planthopper-Resistant Compounds, against Rice Pathogenic Strains. Biology9 (11), 1. 10.3390/biology9110382
204
JeongG.-S.LiB.LeeD.-S.KimK. H.LeeI. K.LeeK. R.et al (2010). Cytoprotective and Anti-inflammatory Effects of Spinasterol via the Induction of Heme Oxygenase-1 in Murine Hippocampal and Microglial Cell Lines. Int. Immunopharmacology10 (12), 1587–1594. 10.1016/j.intimp.2010.09.013
205
JinX.YangR.YanX.ZhouY.WangX.GuZ. (2016). Malic Acid and Oxalic Acid Spraying Enhances Phytic Acid Degradation and Total Antioxidant Capacity of Mung Bean Sprouts. Int. J. Food Sci. Tech.51 (2), 370–380. 10.1111/ijfs.12941
206
JohannS.CisalpinoP. S.WatanabeG. A.CotaB. B.de SiqueiraE. P.PizzolattiM. G.et al (2010). Antifungal Activity of Extracts of Some Plants Used in Brazilian Traditional Medicine against the Pathogenic fungusParacoccidioides Brasiliensis. Pharm. Biol.48 (4), 388–396. 10.3109/13880200903150385
207
Jothi RamalingamR.Vaali-MohammedM.-A.Al-LohedanH. A.AppaturiJ. N. (2017). Synthesis and Bio-Physical Characterization of Silver Nanoparticle and Ag-Mesoporous MnO2 Nanocomposite for Anti-microbial and Anti-cancer Activity. J. Mol. Liquids243, 348–357. 10.1016/j.molliq.2017.08.037
208
JoungD.-K.JoungH. E. E.YangD.-W.KwonD.-Y.ChoiJ.-G.WooS. E. O.et al (2012). Synergistic Effect of Rhein in Combination with Ampicillin or Oxacillin against Methicillin-Resistant Staphylococcus aureus. Exp. Ther. Med.3 (4), 608–612. 10.3892/etm.2012.459
209
JyonouchiH.HillR. J.TomitaY.GoodR. A. (2009). Studies of Immunomodulating Actions of Carotenoids. I. Effects Ofβ‐carotene and Astaxanthin on Murine Lymphocyte Functions and Cell Surface Marker Expression Inin Vitroculture System. Nutr. Cancer16 (2), 93–105. 10.1080/01635589109514148
210
KabirF.KatayamaS.TanjiN.NakamuraS. (2014). Antimicrobial Effects of Chlorogenic Acid and Related Compounds. J. Korean Soc. Appl. Biol. Chem.57 (3), 359–365. 10.1007/s13765-014-4056-6
211
KangR.TianW.CaoW.SunY.ZhangH.-N.FengY.-D.et al (2021). Ligustroflavone Ameliorates CCl4-Induced Liver Fibrosis through Down-Regulating the TGF-β/Smad Signaling Pathway. Chin. J. Nat. Medicines19 (3), 170–180. 10.1016/s1875-5364(21)60018-3
212
KapilA.KoulI. B.SuriO. P. (1995). Antihepatotoxic Effects of Chlorogenic Acid fromAnthocephalus Cadamba. Phytotherapy Res.9 (3), 189–193. 10.1002/ptr.2650090307
213
KarimN.KhanI.AbdelhalimA.HalimS. A.KhanA.Al-HarrasiA. (2021). Stigmasterol Can Be New Steroidal Drug for Neurological Disorders: Evidence of the GABAergic Mechanism via Receptor Modulation. Phytomedicine90. 10.1016/j.phymed.2021.153646
214
KassuyaR. M.dos SantosE.BossoF. H.PedrosoT. F.MarinhoJ. V. N.SalvadorM. J.et al (2021). Anti-inflammatory Properties of Ethanolic Extract and 2″-O-β-D-Glucopyranosyl-Vitexin Obtained from Alternanthera Tenella Colla Whole Plant. Inflammation44 (4), 1540–1552. 10.1007/s10753-021-01438-7
215
KasthuriO. R.RameshB. (2018). Toxicity Studies on Leaf Extracts of Alternanthera Brasiliana (L.) Kuntze and Alternanthera Bettzickiana (Regel) Voss. J. Appl. Pharm. Sci.8 (10), 82–89. 10.7324/japs.2018.81011
216
KaurS.SharmaA.BediP. M. S. (2017). Bioactivity Guided Isolation, Characterization and Quantification of an Anxiolytic Constituent - Kaempferol, from Melilotus Officinalis Aerial Parts. J. Biologically Active Prod. Nat.7 (5), 379–390. 10.1080/22311866.2017.1378923
217
KhaliliM.AttarM.AmirlatifiR.MalekiZ. N.HoseiniS. M. (2020). Effects of Dietary Myrcene Administration on Antioxidant Gene Responses in Common Carp (Cyprinus carpio), Exposed to Copper Sulphate. Aquac. Res.51 (4), 1653–1659. 10.1111/are.14511
218
KhamphukdeeC.ChulikhitY.DaodeeS.MonthakantiratO. (2017). Potential of Alternanthera Philoxeroides on Improvement of Anxiety-like Behavior Induced by Ovariectomized Mice Model. Indian J. Pharm. Edu. Res.51 (3s2), s493–s497. 10.5530/ijper.51.3s.73
219
KhamphukdeeC.MonthakantiratO.ChulikhitY.BoonyaratC.DaodeeS.Aon-imP.et al (2021). Antidementia Effects of Alternanthera Philoxeroides in Ovariectomized Mice Supported by NMR-Based Metabolomic Analysis. Molecules26 (9), 1. 10.3390/molecules26092789
220
KhamphukdeeC.MonthakantiratO.ChulikhitY.ButtachonS.LeeM.SilvaA.et al (2018). Chemical Constituents and Antidepressant-like Effects in Ovariectomized Mice of the Ethanol Extract of Alternanthera Philoxeroides. Molecules23 (9), 1. 10.3390/molecules23092202
221
KhanK. A.KumarN.NayakP. G.NampoothiriM.ShenoyR. R.KrishnadasN.et al (2013). Impact of Caffeic Acid on Aluminium Chloride-Induced Dementia in Rats. J. Pharm. Pharmacol.65 (12), 1745–1752. 10.1111/jphp.12126
222
KhanM. S.YusufzaiS. K.YingL. Y.ZulnashriqW. (2018). Gc-Ms Based Chemical Profiling and Evaluation of Antioxidant Potential of Leaves and Stems of Alternanthera Sessilis Red from Sabah, Malaysia. Int. J. Pharm. Pharm. Sci.10 (7), 1. 10.22159/ijpps.2018v10i7.25204
223
KhanM.YusufzaiS.KaunL.ShahM.IdrisR. (2016). Chemical Composition and Antioxidant Activity of Essential Oil of Leaves and Flowers of Alternanthera Sessilis Red from Sabah. J. Appl. Pharm. Sci.6, 157–161. 10.7324/japs.2016.601222
224
KhatunF.ZamanF.MosaiabT.MostafaF.ZamanM.RehanaF.et al (2012). Evaluation of Antinociceptive and Antihyperglycemic Activities in Methanol Extracts of Whole Plants of Alternanthera Philoxeroides (Mart.) Griseb. (Amaranthaceae) in Mice. Pak J. Pharm. Sci.25 (3), 583–587.
225
KiasalariZ.HeydarifardR.KhaliliM.Afshin-MajdS.BaluchnejadmojaradT.ZahediE.et al (2017). Ellagic Acid Ameliorates Learning and Memory Deficits in a Rat Model of Alzheimer’s Disease: an Exploration of Underlying Mechanisms. Psychopharmacology234 (12), 1841–1852. 10.1007/s00213-017-4589-6
226
KiliçI.YeşiloğluY. (2013). Spectroscopic Studies on the Antioxidant Activity of P-Coumaric Acid. Spectrochimica Acta A: Mol. Biomol. Spectrosc.115, 719–724. 10.1016/j.saa.2013.06.110
227
KimC.-S.KimJ.LeeY. M.SohnE.JoK.KimJ. S. (2011). Inhibitory Effects of Chlorogenic Acid on Aldose Reductase Activity In Vitro and Cataractogenesis in Galactose-Fed Rats. Arch. Pharmacal Res.34 (5), 847–852. 10.1007/s12272-011-0519-z
228
KimD.-S.LeeH.-J.JeonY.-D.HanY.-H.KeeJ.-Y.KimH.-J.et al (2015). Alpha-Pinene Exhibits Anti-inflammatory Activity through the Suppression of MAPKs and the NF-Κb Pathway in Mouse Peritoneal Macrophages. Am. J. Chin. Med.43 (04), 731–742. 10.1142/s0192415x15500457
229
KimH.-B.LeeS.HwangE.-S.MaengS.ParkJ.-H. (2017a). p-Coumaric Acid Enhances Long-Term Potentiation and Recovers Scopolamine-Induced Learning and Memory Impairments. Biochem. Biophysical Res. Commun.492 (3), 493–499. 10.1016/j.bbrc.2017.08.068
230
KimH.-J.LeeB.-H.ChoiS.-H.JungS.-W.KimH.-S.LeeJ.-H.et al (2014). Differential Effects of Quercetin Glycosides on GABAC Receptor Channel Activity. Arch. Pharmacal Res.38 (1), 108–114. 10.1007/s12272-014-0409-2
231
KimH.-R.LeeD.-M.LeeS.-H.SeongA.-R.GinD.-W.HwangJ.-A.et al (2010). Chlorogenic Acid Suppresses Pulmonary Eosinophilia, IgE Production, and Th2-type Cytokine Production in an Ovalbumin-Induced Allergic Asthma: Activation of STAT-6 and JNK Is Inhibited by Chlorogenic Acid. Int. Immunopharmacology10 (10), 1242–1248. 10.1016/j.intimp.2010.07.005
232
KimH. J.FanX.GabbiC.YakimchukK.PariniP.WarnerM.et al (2008). Liver X Receptor (LXR ): A Link between -sitosterol and Amyotrophic Lateral Sclerosis-Parkinson's Dementia. Proc. Natl. Acad. Sci.105 (6), 2094–2099. 10.1073/pnas.0711599105
233
KimJ. H.CampbellB. C.MahoneyN.ChanK. L.MolyneuxR. J.MayG. S. (2007). Enhancement of Fludioxonil Fungicidal Activity by Disrupting Cellular Glutathione Homeostasis with 2,5-dihydroxybenzoic Acid. FEMS Microbiol. Lett.270 (2), 284–290. 10.1111/j.1574-6968.2007.00682.x
234
KimS. H.Naveen KumarC.KimH. J.KimD. H.ChoJ.JinC.et al (2009). Glucose-containing Flavones—Their Synthesis and Antioxidant and Neuroprotective Activities. Bioorg. Med. Chem. Lett.19 (21), 6009–6013. 10.1016/j.bmcl.2009.09.062
235
KimS. M.ParkY. J.ShinM.-S.KimH.-R.KimM. J.LeeS. H.et al (2017b). Acacetin Inhibits Neuronal Cell Death Induced by 6-hydroxydopamine in Cellular Parkinson’s Disease Model. Bioorg. Med. Chem. Lett.27 (23), 5207–5212. 10.1016/j.bmcl.2017.10.048
236
KongC.-S.JeongC.-H.ChoiJ.-S.KimK.-J.JeongJ.-W. (2013). Antiangiogenic Effects of P-Coumaric Acid in Human Endothelial Cells. Phytotherapy Res.27 (3), 317–323. 10.1002/ptr.4718
237
KooH. (2003). Inhibition of Streptococcus Mutans Biofilm Accumulation and Polysaccharide Production by Apigenin and Tt-Farnesol. J. Antimicrob. Chemother.52 (5), 782–789. 10.1093/jac/dkg449
238
KoolenH. H. F.PralE. M. F.AlfieriS. C.MarinhoJ. V. N.SerainA. F.Hernández-TascoA. J.et al (2017). Antiprotozoal and Antioxidant Alkaloids from Alternanthera Littoralis. Phytochemistry134, 106–113. 10.1016/j.phytochem.2016.11.008
239
KotaS.GovadaV. R.AnanthaR. K.VermaM. K. (2017). An Investigation into Phytochemical Constituents, Antioxidant, Antibacterial and Anti-cataract Activity of Alternanthera Sessilis, a Predominant Wild Leafy Vegetable of South India. Biocatal. Agric. Biotechnol.10, 197–203. 10.1016/j.bcab.2017.03.008
240
KrennL.BeyerG.PertzH.KarallE.KremserM.GalambosiB.et al (2011). In Vitro Antispasmodic and Antiinflammatory Effects of Drosera Rotundifolia. Arzneimittelforschung54 (07), 402–405. 10.1055/s-0031-1296991
241
KumarD. A.PalanichamyV.RoopanS. M. (2014). Green Synthesis of Silver Nanoparticles Using Alternanthera Dentata Leaf Extract at Room Temperature and Their Antimicrobial Activity. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc.127, 168–171. 10.1016/j.saa.2014.02.058
242
KumarS. M.RaniS. G.AstalakshmiN.ManasaG.VanajaP.SirishaG.et al (2011b). Screening of Aqueous and Ethanolic Extracts of Aerial Parts of Alternanthera Sessilis Linn. R.Br.ex.DC (Amaranthaceae) for Antidiabetic Activity. J. Pharm. Sci. Res.4 (5), 1528–1530.
243
KumarS.SinghP.MishraG.SrivastavS.JhaK. K.KhosaR. L. (2011a). Phytopharmacological Review of Alternanthera Brasiliana (Amaranthaceae). Asian J. Plant Sci. Res.1 (1), 41–47.
244
KumariE. V. N.KrishnanV. (2016). Antimicrobial Activity of Alternanthera Sessilis (L) R. BR. Ex. DC and Alternanthera Philoxeroides (Mart). Griseb. World J. Res. Rev.3 (3), 78–81.
245
LanQ.DiD.WangS.ZhaoQ.GaoY.ChangD.et al (2020). Chitosan-N-acetylcysteine Modified HP-β-CD Inclusion Complex as a Potential Ocular Delivery System for Anti-cataract Drug: Quercetin. J. Drug Deliv. Sci. Tech.55. 10.1016/j.jddst.2019.101407
246
LeeJ. H.MohanC. D.ShanmugamM. K.RangappaS.SethiG.SiveenK. S.et al (2020). Vitexin Abrogates Invasion and Survival of Hepatocellular Carcinoma Cells through Targeting STAT3 Signaling Pathway. Biochimie175, 58–68. 10.1016/j.biochi.2020.05.006
247
LeeS.KimH.-B.HwangE.-S.KimE.-s.KimS.-S.JeonT.-D.et al (2018). Antidepressant-like Effects of P-Coumaric Acid on LPS-Induced Depressive and Inflammatory Changes in Rats. Exp. Neurobiol.27 (3), 189–199. 10.5607/en.2018.27.3.189
248
LeemingJ. P.HollandK. T.BojarR. A. (1986). The In Vitro Antimicrobial Effect of Azelaic Acid. Br. J. Dermatol.115 (5), 551–556. 10.1111/j.1365-2133.1986.tb05764.x
249
LesjakM.BearaI.SiminN.PintaćD.MajkićT.BekvalacK.et al (2018). Antioxidant and Anti-inflammatory Activities of Quercetin and its Derivatives. J. Funct. Foods40, 68–75. 10.1016/j.jff.2017.10.047
250
LiB.GuoQ.-L.TianY.LiuS.-J.WangQ.ChenL.et al (2016). New Anti-HBV C-Boivinopyranosyl Flavones from Alternanthera Philoxeroides. Molecules21 (3), 1. 10.3390/molecules21030336
251
LiG.WangX.XuY.ZhangB.XiaX. (2013). Antimicrobial Effect and Mode of Action of Chlorogenic Acid on Staphylococcus aureus. Eur. Food Res. Tech.238 (4), 589–596. 10.1007/s00217-013-2140-5
252
LiP.PengY.MaQ.LiZ.ZhangX. (2020). p>Study on the Formation of Antihypertensive Twin Drugs by Caffeic Acid and Ferulic Acid with Telmisartan</p>. Drug Des. Dev. Ther.Vol. 14, 977–992. 10.2147/dddt.S225705<
253
LiS.-P.WangY.-W.QiS.-L.ZhangY.-P.DengG.DingW.-Z.et al (2018). Analogous β-Carboline Alkaloids Harmaline and Harmine Ameliorate Scopolamine-Induced Cognition Dysfunction by Attenuating Acetylcholinesterase Activity, Oxidative Stress, and Inflammation in Mice. Front. Pharmacol.9. 10.3389/fphar.2018.00346
254
LiX.OuyangX.CaiR.ChenD. (2019). 3′,8″-Dimerization Enhances the Antioxidant Capacity of Flavonoids: Evidence from Acacetin and Isoginkgetin. Molecules24 (11), 1. 10.3390/molecules24112039
255
LiangQ.YangJ.HeJ.ChenX.ZhangH.JiaM.et al (2020). Stigmasterol Alleviates Cerebral Ischemia/reperfusion Injury by Attenuating Inflammation and Improving Antioxidant Defenses in Rats. Biosci. Rep.40 (4), 1. 10.1042/bsr20192133
256
LimaV. N.Oliveira-TintinoC. D. M.SantosE. S.MoraisL. P.TintinoS. R.FreitasT. S.et al (2016). Antimicrobial and Enhancement of the Antibiotic Activity by Phenolic Compounds: Gallic Acid, Caffeic Acid and Pyrogallol. Microb. Pathogenesis99, 56–61. 10.1016/j.micpath.2016.08.004
257
LinF.-H.LinJ.-Y.GuptaR. D.TournasJ. A.BurchJ. A.Angelica SelimM.et al (2005). Ferulic Acid Stabilizes a Solution of Vitamins C and E and Doubles its Photoprotection of Skin. J. Invest. Dermatol.125 (4), 826–832. 10.1111/j.0022-202X.2005.23768.x
258
LinK.SzeS. C.-W.LiuB.ZhangZ.ZhangZ.ZhuP.et al (2021). 20(S)-protopanaxadiol and Oleanolic Acid Ameliorate Cognitive Deficits in APP/PS1 Transgenic Mice by Enhancing Hippocampal Neurogenesis. J. Ginseng Res.45 (2), 325–333. 10.1016/j.jgr.2020.07.003
259
LinM.-K.YuY.-L.ChenK.-C.ChangW.-T.LeeM.-S.YangM.-J.et al (2011). Kaempferol from Semen Cuscutae Attenuates the Immune Function of Dendritic Cells. Immunobiology216 (10), 1103–1109. 10.1016/j.imbio.2011.05.002
260
LinS.-C.LinY.-H.ShyuuS.-J.LinC.-C. (1994). Hepatoprotective Effects of Taiwan Folk Medicine:Alternanthera Sessilis on Liver Damage Induced by Various Hepatotoxins. Phytotherapy Res.8 (7), 391–398. 10.1002/ptr.2650080703
261
LiraM. H. P. d.Andrade JúniorF. P. d.MoraesG. F. Q.MacenaG. d. S.PereiraF. d. O.LimaI. O. (2020). Antimicrobial Activity of Geraniol: an Integrative Review. J. Essent. Oil Res.32 (3), 187–197. 10.1080/10412905.2020.1745697
262
LiuJ.-R.DongH.-W.SunX.-R.WangQ.SunW.-G.ParryJ. W.et al (2009). Effects of β-Ionone on Mammary Carcinogenesis and Antioxidant Status in Rats Treated with DMBA. Nutr. Cancer62 (1), 58–65. 10.1080/01635580903191510
263
LiuL.LiuY.ZhaoJ.XingX.ZhangC.MengH. (2020). Neuroprotective Effects of D-(-)-Quinic Acid on Aluminum Chloride-Induced Dementia in Rats. Evidence-Based Complement. Altern. Med.2020, 1–10. 10.1155/2020/5602597
264
LiuX.JiangQ.LiuH.LuoS. (2019). Vitexin Induces Apoptosis through Mitochondrial Pathway and PI3K/Akt/mTOR Signaling in Human Non-small Cell Lung Cancer A549 Cells. Biol. Res.52 (1), 1. 10.1186/s40659-019-0214-y
265
LobineD.AhmedS.AschnerM.KhanH.MirzaeiH.MahomoodallyM. F. (2020). Antiurolithiatic Effects of Pentacyclic Triterpenes: The Distance Traveled from Therapeutic Aspects. Drug Dev. Res.81 (6), 671–684. 10.1002/ddr.21670
266
LoizouS.LekakisI.ChrousosG. P.MoutsatsouP. (2010). β-Sitosterol Exhibits Anti-inflammatory Activity in Human Aortic Endothelial Cells. Mol. Nutr. Food Res.54 (4), 551–558. 10.1002/mnfr.200900012
267
LozoyaX.MeckesM.Abou-ZaidM.TortorielloJ.NozzolilloC.ArnasonJ. T. (1994). Quercetin Glycosides in Psidium Guajava L. Leaves and Determination of a Spasmolytic Principle. Arch. Med. Res.25 (1), 11–15.
268
LuckyG.DiameA. (2010). Ethnobotany and Ecological Studies of Plants Used for Reproductive Health: A Case Study at Bia Biosphere Reserve in the Western Region of Ghana [Online]. France: The Division of Ecological Sciences: UNESCO. Available: http://www.unesco.org/science/doc/mab/MAB_Ghana2008.pdf (Accessed 04 01, 2021).
269
LvC.HongT.YangZ.ZhangY.WangL.DongM.et al (2012). Effect of Quercetin in the 1-Methyl-4-Phenyl-1, 2, 3, 6-Tetrahydropyridine-Induced Mouse Model of Parkinson's Disease. Evidence-Based Complement. Altern. Med.2012, 1–6. 10.1155/2012/928643
270
MaB.YouX.LuF. (2014). Inhibitory Effects of β-ionone on Amyloid Fibril Formation of β-lactoglobulin. Int. J. Biol. Macromolecules64, 162–167. 10.1016/j.ijbiomac.2013.12.003
271
MachadoD. G.NeisV. B.BalenG. O.CollaA.CunhaM. P.DalmarcoJ. B.et al (2012). Antidepressant-like Effect of Ursolic Acid Isolated from Rosmarinus Officinalis L. In Mice: Evidence for the Involvement of the Dopaminergic System. Pharmacol. Biochem. Behav.103 (2), 204–211. 10.1016/j.pbb.2012.08.016
272
MadunićJ.MadunićI. V.GajskiG.PopićJ.Garaj-VrhovacV. (2018). Apigenin: A Dietary Flavonoid with Diverse Anticancer Properties. Cancer Lett.413, 11–22. 10.1016/j.canlet.2017.10.041
273
MahajanS. G.MehtaA. A. (2011). Suppression of Ovalbumin-Induced Th2-Driven Airway Inflammation by β-sitosterol in a guinea Pig Model of Asthma. Eur. J. Pharmacol.650 (1), 458–464. 10.1016/j.ejphar.2010.09.075
274
MalarD. S.PrasanthM. I.JeyakumarM.BalamuruganK.DeviK. P. (2020). Vitexin Prevents Aβ Proteotoxicity in Transgenic Caenorhabditis elegans Model of Alzheimer's Disease by Modulating Unfolded Protein Response. J. Biochem. Mol. Toxicol.35 (1), 1. 10.1002/jbt.22632
275
Mamani-MatsudaM.KaussT.Al-KharratA.RambertJ.FawazF.ThiolatD.et al (2006). Therapeutic and Preventive Properties of Quercetin in Experimental Arthritis Correlate with Decreased Macrophage Inflammatory Mediators. Biochem. Pharmacol.72 (10), 1304–1310. 10.1016/j.bcp.2006.08.001
276
ManaloR. A. M.ArolladoE. C.HeraldeF. M.Iii (2020). Alternanthera Sessilis Leaf Fractions Possess In Vitro Inhibitory Activities in Mammalian α-amylase and α-glucosidase. Pharm. Sci. Asia47 (3), 279–286. 10.29090/psa.2020.03.019.0076
277
MananM.SaleemU.AkashM. S. H.QasimM.HayatM.RazaZ.et al (2020). Antiarthritic Potential of Comprehensively Standardized Extract of Alternanthera Bettzickiana: In Vitro and In Vivo Studies. ACS Omega5 (31), 19478–19496. 10.1021/acsomega.0c01670
278
MandaK.BhatiaA. L. (2003). Role of β-carotene against Acetaminophen-Induced Hepatotoxicity in Mice. Nutr. Res.23 (8), 1097–1103. 10.1016/s0271-5317(03)00103-9
279
ManjunathS. H.ThimmulappaR. K. (2021). Antiviral, Immunomodulatory, and Anticoagulant Effects of Quercetin and its Derivatives: Potential Role in Prevention and Management of COVID-19. J. Pharm. Anal.. 10.1016/j.jpha.2021.09.009
280
MarioniJ.da SilvaM. A.CabreraJ. L.MontoyaS. C. N.ParajeM. G. (2016). The Anthraquinones Rubiadin and its 1-methyl Ether Isolated from Heterophyllaea Pustulata Reduces Candida tropicalis Biofilms Formation. Phytomedicine23 (12), 1321–1328. 10.1016/j.phymed.2016.07.008
281
MartinoR.ArcosM. L. B.AlonsoR.SülsenV.CremaschiG.AnesiniC. (2016). Polyphenol-Rich Fraction fromLarrea Divaricataand its Main Flavonoid Quercetin-3-Methyl Ether Induce Apoptosis in Lymphoma Cells through Nitrosative Stress. Phytotherapy Res.30 (7), 1128–1136. 10.1002/ptr.5615
282
MasryS. H. D.TahaT. H.BotrosW. A.MahfouzH.Al-KahtaniS. N.AnsariM. J.et al (2021). Antimicrobial Activity of Camphor Tree Silver Nano-Particles against Foulbrood Diseases and Finding Out New Strain of Serratia marcescens as a Secondary Infection on Honeybee Larvae. Saudi J. Biol. Sci.28 (4), 2067–2075. 10.1016/j.sjbs.2021.02.038
283
MesbahH. A.SaadA. S.MouradA. K.TamanF. A.MohamedI. B. (2007). Joint Action of Quercetin with Four Insecticides on the Cotton Leaf-Worm Larvae, Spodoptera Littoralis Boisd. (Lep. : Noctuidae) in Egypt. Commun. Agric. Appl. Biol. Sci.72 (3), 445–457.
284
MhillajE.CatinoS.MiceliF. M.SantangeloR.TrabaceL.CuomoV.et al (2017). Ferulic Acid Improves Cognitive Skills through the Activation of the Heme Oxygenase System in the Rat. Mol. Neurobiol.55 (2), 905–916. 10.1007/s12035-017-0381-1
285
MikhlinE. D.RadinaV. P.DmitrovskiiA. A.BlinkovaL. P.ButovaL. G. (1983). Antifungal and Antimicrobial Activity of Beta-Ionone and Vitamin A Derivatives. Prikl Biokhim Mikrobiol19 (6), 795–803.
286
MiltonprabuS.TomczykM.Skalicka-WoźniakK.RastrelliL.DagliaM.NabaviS. F.et al (2017). Hepatoprotective Effect of Quercetin: From Chemistry to Medicine. Food Chem. Toxicol.108, 365–374. 10.1016/j.fct.2016.08.034
287
MishraB.PriyadarsiniK. I.KumarM. S.UnnikrishnanM. K.MohanH. (2003). Effect of O -glycosilation on the Antioxidant Activity and Free Radical Reactions of a Plant Flavonoid, Chrysoeriol. Bioorg. Med. Chem.11 (13), 2677–2685. 10.1016/s0968-0896(03)00232-3
288
MlcekJ.JurikovaT.SkrovankovaS.SochorJ. (2016). Quercetin and its Anti-allergic Immune Response. Molecules21 (5), 1. 10.3390/molecules21050623
289
MoJ.PanichayupakaranantP.KaewnopparatN.NitiruangjarasA.ReanmongkolW. (2014). Wound Healing Activities of Standardized Pomegranate Rind Extract and its Major Antioxidant Ellagic Acid in Rat Dermal Wounds. J. Nat. Medicines68 (2), 377–386. 10.1007/s11418-013-0813-9
290
MohaimenulM. D.DuttaK.FerdiousiN.Nath RoyD. (2020). Comparative Studies on Antidiabetic, Analgesic, and Cytotoxic Effect of Ethanolic Extracts of Amaranthus Gangeticus L. And Alternanthera Sessilis L. Asian J. Pharm. Clin. Res.7, 113–117. 10.22159/ajpcr.2020.v13i11.39232
291
MohapatraS. S.KafleA.ChatterjeeJ.MohanP.RoyR. K.ReddyI. (2018). Analgesic Activity of Hydroethanolic Extract of Alternanthera Sessilis in Mice. J. Pharmacognosy Phytochemistry7 (4), 1836–1839. 10.20546/ijcmas.2018.701.120
292
Mohd HazliU. H. A.Abdul-AzizA.Mat-JunitS.CheeC. F.KongK. W. (2019). Solid-liquid Extraction of Bioactive Compounds with Antioxidant Potential from Alternanthera Sesillis (Red) and Identification of the Polyphenols Using UHPLC-QqQ-MS/MS. Food Res. Int.115, 241–250. 10.1016/j.foodres.2018.08.094
293
MondalH.HossainH.AwangK.SahaS.Mamun-Ur-RashidS.IslamM. K.et al (2015). Anthelmintic Activity of Ellagic Acid, a Major Constituent of Alternanthera Sessilis against Haemonchus contortus. Pakistan Vet. J.35 (1), 58–62.
294
MondalH.SahaS.AwangK.HossainH.AblatA.IslamM. K.et al (2014). Central-stimulating and Analgesic Activity of the Ethanolic Extract of Alternanthera Sessilis in Mice. BMC Complement. Altern. Med.14 (1), 1. 10.1186/1472-6882-14-398
295
MoniciM.BaglioniP.MulinacciN.BaldiA.VincieriF. F. (1994). A Research Model on Flavonoids as Photoprotectors: Studies on the Photochemistry of Kaempferol and Pelargonidin. Acta Horticulturae381, 340–347. 10.17660/ActaHortic.1994.381.41
296
MonroyA. E. M.LimsiacoC. L. (2016). Phytochemical and Antimicrobial Analysis of “Lupo” (Alternanthera Sessilis L.R.BR.). West Visayas State. Univ. Res. J.5 (2), 21–34.
297
MonteiroÁ. B.Kelly de Souza RodriguesC.Petícia do NascimentoE.SalesV. d. S.de Araújo DelmondesG.Nogueira da CostaM. H.et al (2020). Anxiolytic and Antidepressant-like Effects of Annona Coriacea (Mart.) and Caffeic Acid in Mice. Food Chem. Toxicol.136, 1. 10.1016/j.fct.2019.111049
298
MoonJ.-M.ParkS.-H.JheeK.-H.YangS.-A. (2018). Protection against UVB-Induced Wrinkle Formation in SKH-1 Hairless Mice: Efficacy of Tricin Isolated from Enzyme-Treated Zizania Latifolia Extract. Molecules23 (9), 1. 10.3390/molecules23092254
299
MoraesV. L. G.SantosL. F. M.CastroS. B.LoureiroL. H.LimaO. A.SouzaM. L. M.et al (1994). Inhibition of Lymphocyte Activation by Extracts and Fractions of Kalanchoe, Alternanthera, Paullinia and Mikania Species. Phytomedicine1 (3), 199–204. 10.1016/s0944-7113(11)80065-6
300
MoralesM. A.TortorielloJ.MeckesM.PazD.LozoyaX. (1994). Calcium-antagonist Effect of Quercetin and its Relation with the Spasmolytic Properties of Psidium Guajava L. Arch. Med. Res.25 (1), 17–21.
301
Moreno-AnzúrezN.MarquinaS.AlvarezL.ZamilpaA.Castillo-EspañaP.Perea-ArangoI.et al (2017). A Cytotoxic and Anti-inflammatory Campesterol Derivative from Genetically Transformed Hairy Roots of Lopezia Racemosa Cav. (Onagraceae). Molecules22 (1), 1. 10.3390/molecules22010118
302
MorganL. V.PetryF.ScatolinM.de OliveiraP. V.AlvesB. O.ZilliG. A. L.et al (2021). Investigation of the Anti-inflammatory Effects of Stigmasterol in Mice: Insight into its Mechanism of Action. Behav. Pharmacol.32 (8), 640–651. 10.1097/fbp.0000000000000658
303
MouraD. J.RichterM. F.BoeiraJ. M.Pegas HenriquesJ. A.SaffiJ. (2007). Antioxidant Properties of -carboline Alkaloids Are Related to Their Antimutagenic and Antigenotoxic Activities. Mutagenesis22 (4), 293–302. 10.1093/mutage/gem016
304
MouryaP.RohitS.NeeteshK. J. (2020). A Study of Antihyperglycaemic Activity of Alternanthera Pungens Kunth on Alloxan Induced Diabetic Rats. Int. J. Pharm. Life Sci.11 (7), 44.
305
MouryaP.SharmaN. K.GuptaM. K. (2019). Antioxidant Activity of Ethanolic and Aqueous Extracts of Alternanthera Pungens Kunth. Asian J. Pharm. Pharmacol.5 (6), 1091–1096. 10.31024/ajpp.2019.5.6.3
306
MózsikG.Abdel-SalamO. M. E.BódisB.KarádiO.KirályÁ.SütőG.et al (1996). Gastric Mucosal Preventive Effects of Prostacyclin and β-carotene, and Their Biochemical Effects in Rats Treated with Ethanol and HCl at Different Doses and Time Intervals after Administration of Necrotizing Agents. Inflammopharmacology4 (4), 361–378. 10.1007/bf02755789
307
MuniandyK.GothaiS.BadranK. M. H.Suresh KumarS.EsaN. M.ArulselvanP. (2018a). Suppression of Proinflammatory Cytokines and Mediators in LPS-Induced RAW 264.7 Macrophages by Stem Extract of Alternanthera Sessilis via the Inhibition of the NF-Κb Pathway. J. Immunol. Res.2018, 1–12. 10.1155/2018/3430684
308
MuniandyK.GothaiS.TanW. S.KumarS. S.Mohd EsaN.ChandramohanG.et al (2018b). Vitro Wound Healing Potential of Stem Extract of Alternanthera Sessilis. Evidence-Based Complement. Altern. Med.2018, 1–13. 10.1155/2018/3142073
309
MuraliR.SaravananR. (2012). Antidiabetic Effect of D-Limonene, a Monoterpene in Streptozotocin-Induced Diabetic Rats. Biomed. Prev. Nutr.2 (4), 269–275. 10.1016/j.bionut.2012.08.008
310
MuthulakshmiS.SaravananR. (2013). Protective Effects of Azelaic Acid against High-Fat Diet-Induced Oxidative Stress in Liver, Kidney and Heart of C57BL/6J Mice. Mol. Cell Biochem.377 (1-2), 23–33. 10.1007/s11010-013-1566-1
311
NadaA. A.ArulM. R.RamosD. M.KronekováZ.MosnáčekJ.RudraiahS.et al (2018). Bioactive Polymeric Formulations for Wound Healing. Polym. Adv. Tech.29 (6), 1815–1825. 10.1002/pat.4288
312
NagalingamM.KalpanaV. N.RajeswariD. R.PanneerselvamA. (2018). Biosynthesis, Characterization, and Evaluation of Bioactivities of Leaf Extract-Mediated Biocompatible Gold Nanoparticles from Alternanthera Bettzickiana. Biotechnol. Rep.19, 1. 10.1016/j.btre.2018.e00268
313
NaiduV. S. G. R. (2012). Hand Book on Weed Identification. Jaipur, India: Directorate of Weed Science Research.
314
NakhaeeS.FarrokhfallK.Miri-MoghaddamE.FoadoddiniM.AskariM.AmirabadizadehA.et al (2021). The Effects of Naloxone, Diazepam, and Quercetin on Seizure and Sedation in Acute on Chronic Tramadol Administration: an Experimental Study. Behav. Brain Functions17 (1), 1. 10.1186/s12993-021-00178-w
315
NarasimhanA.ChinnaiyanM.KarundeviB. (2015). Ferulic Acid Exerts its Antidiabetic Effect by Modulating Insulin-Signalling Molecules in the Liver of High-Fat Diet and Fructose-Induced Type-2 Diabetic Adult Male Rat. Appl. Physiol. Nutr. Metab.40 (8), 769–781. 10.1139/apnm-2015-0002
316
Nassiri-AslM.HajialiF.TaghilooM.AbbasiE.MohseniF.YousefiF. (2014). Comparison between the Effects of Quercetin on Seizure Threshold in Acute and Chronic Seizure Models. Toxicol. Ind. Health32 (5), 936–944. 10.1177/0748233713518603
317
NieoczymD.SocałaK.RaszewskiG.WlaźP. (2014). Effect of Quercetin and Rutin in Some Acute Seizure Models in Mice. Prog. Neuro-Psychopharmacology Biol. Psychiatry54, 50–58. 10.1016/j.pnpbp.2014.05.007
318
NileS. H.KoE. Y.KimD. H.KeumY.-S. (2016). Screening of Ferulic Acid Related Compounds as Inhibitors of Xanthine Oxidase and Cyclooxygenase-2 with Anti-inflammatory Activity. Revista Brasileira de Farmacognosia26 (1), 50–55. 10.1016/j.bjp.2015.08.013
319
NiraimathiK. L.SudhaV.LavanyaR.BrindhaP. (2013). Biosynthesis of Silver Nanoparticles Using Alternanthera Sessilis (Linn.) Extract and Their Antimicrobial, Antioxidant Activities. Colloids Surf. B: Biointerfaces102, 288–291. 10.1016/j.colsurfb.2012.08.041
320
Niranjan PanatA.Bhushan AmruteK.ShateeshB.Santosh HaramK.Geeta SharmaK.Saroj GhaskadbiS. (2015). Antioxidant Profiling of C3 Quercetin Glycosides: Quercitrin, Quercetin 3-β-D-Glucoside and Quercetin 3-O-(6”-O-Malonyl)-β-Dglucoside in Cell Free Environment. Free Radicals Antioxid.5 (2), 90–100. 10.5530/fra.2015.2.7
321
NovakA. F.ClarkG. C.DupuyH. P. (1961). Antimicrobial Activity of Some Ricinoleic Acid Oleic Acid Derivatives. J. Am. Oil Chemists Soc.38 (6), 321–324. 10.1007/bf02638439
322
ObertreisB.GillerK.TeucherT.BehnkeB.SchmitzH. (1996). Anti-inflammatory Effect of Urtica Dioica Folia Extract in Comparison to Caffeic Malic Acid. Arzneimittelforschung46 (1), 52–56.
323
OdodoM. M.ChoudhuryM. K.DekeboA. H. (2016). Structure Elucidation of β-sitosterol with Antibacterial Activity from the Root Bark of Malva Parviflora. SpringerPlus5 (1), 1. 10.1186/s40064-016-2894-x
324
OgunmoyeA. O.Atewolara-OduleO. C.OlubomehinO. O.OgundareS. A.YussufS. T. (2020). The Chemical Constituents of the Leaves Essential Oil of Alternanthera Pungens (Kunth). Afr. J. Sci. Nat.10, 123–130. 10.46881/ajsn.v10i0.185
325
OhH.-A.HanN.-R.KimM.-J.KimH.-M.JeongH.-J. (2013). Evaluation of the Effect of Kaempferol in a Murine Allergic Rhinitis Model. Eur. J. Pharmacol.718 (1-3), 48–56. 10.1016/j.ejphar.2013.08.045
326
OlaiyaC. O.EsanA. M.AlabiT. D. (2014). Ameliorative Effects of Beta-Sitosterol on Some Biochemical Indices of Hypertension in Wistar Albino Rats. Afr. J. Med. Med. Sci.43 (Suppl. 1), 157–166.
327
OngK. W.HsuA.TanB. K. H. (2013). Anti-diabetic and Anti-lipidemic Effects of Chlorogenic Acid Are Mediated by Ampk Activation. Biochem. Pharmacol.85 (9), 1341–1351. 10.1016/j.bcp.2013.02.008
328
OsunaL.Tapia-PérezM. E.Jiménez-FerrerJ. E.Carrillo-QuirózB. A.Silva-SánchezJ. (2008). Screening ofAlternanthera repens.,Boerhavia coccinea.,Flaveria trinervia.,Tournefortia densiflora., andVitex Mollis. Extracts to Evaluate Their Antibacterial Activity and Effect on Smooth Muscle. I. Pharm. Biol.43 (9), 749–753. 10.1080/13880200500406412
329
OthmanA.IsmailA.HassanF. A.YusofB. N. M.KhatibA. (2016). Comparative Evaluation of Nutritional Compositions, Antioxidant Capacities, and Phenolic Compounds of Red and green Sessile Joyweed ( Alternanthera Sessilis ). J. Funct. Foods21, 263–271. 10.1016/j.jff.2015.12.014
330
OyemitanI. A.BelloO. A.AkinpeluL. A. (2015). Neuropharmacological Evaluation of Ethanolic Leaf Extract of Alternanthera Brasiliana (L.) Kuntze (Amaranthaceae) in Mice. Int. J. Pharm. Sci. Res.6 (9), 3796–3806.
331
OzakiY. (1992). Antiinflammatory Effects of Tetramethylpyrazine and Ferulic Acid. Chem. Pharm. Bull.40 (4), 954–956. 10.1248/cpb.40.954
332
ÖzayY.GüzelS.YumrutaşÖ.PehlivanoğluB.Erdoğduİ. H.YildirimZ.et al (2019). Wound Healing Effect of Kaempferol in Diabetic and Nondiabetic Rats. J. Surg. Res.233, 284–296. 10.1016/j.jss.2018.08.009
333
PS.KP.LahkarM. (2016). Effect of Alternanthera Brasiliana in Experimentally Induced Inflammatory Bowel Disease in Albino Rats. Int. J. Basic Clin. Pharmacol.18, 1809–1815. 10.18203/2319-2003.ijbcp20162789
334
PaivaS. A. R.RussellR. M. (1999). β-Carotene and Other Carotenoids as Antioxidants. J. Am. Coll. Nutr.18 (5), 426–433. 10.1080/07315724.1999.10718880
335
PandaV.SureshS. (2015). Gastro-protective Effects of the Phenolic Acids of Macrotyloma Uniflorum (Horse Gram) on Experimental Gastric Ulcer Models in Rats. Food Biosci.12, 34–46. 10.1016/j.fbio.2015.07.004
336
ParkC. G.KimJ. J.KimH. K. (2020). Lipase-mediated Synthesis of Ricinoleic Acid Vanillyl Ester and Evaluation of Antioxidant and Antibacterial Activity. Enzyme Microb. Tech.133. 10.1016/j.enzmictec.2019.109454
337
ParkJ.LeeG. E.AnH. J.LeeC. J.ChoE. S.KangH. C.et al (2021). Kaempferol Sensitizes Cell Proliferation Inhibition in Oxaliplatin-Resistant colon Cancer Cells. Arch. Pharm. Res.44 (12), 1091–1108. 10.1007/s12272-021-01358-y
338
ParkJ. S.RhoH. S.KimD. H.ChangI. S. (2006). Enzymatic Preparation of Kaempferol from Green Tea Seed and its Antioxidant Activity. J. Agric. Food Chem.54 (8), 2951–2956. 10.1021/jf052900a
339
ParkS.-H.SimY.-B.HanP.-L.LeeJ.-K.SuhH.-W. (2010a). Antidepressant-like Effect of Chlorogenic Acid Isolated fromArtemisia capillarisThunb. Anim. Cell Syst.14 (4), 253–259. 10.1080/19768354.2010.528192
340
ParkS.-H.SimY.-B.HanP.-L.LeeJ.-K.SuhH.-W. (2010b). Antidepressant-like Effect of Kaempferol and Quercitirin, Isolated from Opuntia Ficus-Indica Var. Saboten. Exp. Neurobiol.19 (1), 30–38. 10.5607/en.2010.19.1.30
341
ParkS.-N.LimY. K.FreireM. O.ChoE.JinD.KookJ.-K. (2012a). Antimicrobial Effect of Linalool and α-terpineol against Periodontopathic and Cariogenic Bacteria. Anaerobe18 (3), 369–372. 10.1016/j.anaerobe.2012.04.001
342
ParkS.ChoiJ. J.ParkB.-K.YoonS. J.ChoiJ. E.JinM. (2014). Pheophytin a and Chlorophyll a Suppress Neuroinflammatory Responses in Lipopolysaccharide and Interferon-γ-Stimulated BV2 Microglia. Life Sci.103 (2), 59–67. 10.1016/j.lfs.2014.04.003
343
ParkS. J.KimD. H.JungJ. M.KimJ. M.CaiM.LiuX.et al (2012b). The Ameliorating Effects of Stigmasterol on Scopolamine-Induced Memory Impairments in Mice. Eur. J. Pharmacol.676 (1-3), 64–70. 10.1016/j.ejphar.2011.11.050
344
ParveenZ.DengY.SaeedM. K.DaiR.AhamadW.YuY. H. (2007). Antiinflammatory and Analgesic Activities of Thesium Chinense Turcz Extracts and its Major Flavonoids, Kaempferol and Kaempferol-3-O-Glucoside. Yakugaku Zasshi127 (8), 1275–1279. 10.1248/yakushi.127.1275
345
ParviziF.YaghmaeiP.Haeri RohaniS. A.MardS. A. (2020). Hepatoprotective Properties of P-Coumaric Acid in a Rat Model of Ischemia-Reperfusion. Avicenna J. Phytomed10 (6), 633–640.
346
PathakI.BudhathokiR.YadavN.NiraulaM.KalauniS. K. (2020). Phytochemical Screening, Cytotoxic and Antioxidant Activity of Alternathera Sessilis and Moringa Oleifera. Amrit Res. J.1 (1), 65–71. 10.3126/arj.v1i1.32456
347
PatilR. B.KoreB. A. (2019). Potential of an Invasive weed Alternanthera ficoidea (L.) P. Beauv. As Resource of Antioxidants. Int. J. Scientific Res. Rev.8 (2), 4041–4046.
348
PavelaR. (2011). Antifeedant and Larvicidal Effects of Some Phenolic Components of Essential Oils Lasp Lines of Introduction AgainstSpodoptera littoralis(Boisd.). J. Essent. Oil Bearing Plants14 (3), 266–273. 10.1080/0972060x.2011.10643932
349
PeanaA. T.D'AquilaP. S.PaninF.SerraG.PippiaP.MorettiM. D. L. (2002). Anti-inflammatory Activity of Linalool and Linalyl Acetate Constituents of Essential Oils. Phytomedicine9 (8), 721–726. 10.1078/094471102321621322
350
PejinB.SavicA.SokovicM.GlamoclijaJ.CiricA.NikolicM.et al (2014). Furtherin Vitroevaluation of Antiradical and Antimicrobial Activities of Phytol. Nat. Product. Res.28 (6), 372–376. 10.1080/14786419.2013.869692
351
Pelisoli FormagioE. L.MendelM. T.FracassoR.KnoblochJ. G.TeixeiraP. W.KehlL.et al (2012). Evaluation of the Pharmacological Activity of theAlternanthera Brasilianaaqueous Extract. Pharm. Biol.50 (11), 1442–1447. 10.3109/13880209.2012.688058
352
PereiraD. F.ZanonR. B.dos SantosM.BoligonA. A.AthaydeM. L. (2013). Antioxidant Activities and Triterpenoids Isolated fromAlternanthera brasiliana(L.) Kuntze Leaves. Nat. Product. Res.27 (18), 1660–1663. 10.1080/14786419.2012.750313
353
PeresD. D. A.SarrufF. D.de OliveiraC. A.VelascoM. V. R.BabyA. R. (2018). Ferulic Acid Photoprotective Properties in Association with UV Filters: Multifunctional Sunscreen with Improved SPF and UVA-PF. J. Photochem. Photobiol. B: Biol.185, 46–49. 10.1016/j.jphotobiol.2018.05.026
354
Perez-VizcainoF.DuarteJ.JimenezR.Santos-BuelgaC.OsunaA. (2009). Antihypertensive Effects of the Flavonoid Quercetin. Pharmacol. Rep.61 (1), 67–75. 10.1016/s1734-1140(09)70008-8
355
PeroR. W.LundH.LeandersonT. (2009). Antioxidant Metabolism Induced by Quinic Acid. Increased Urinary Excretion of Tryptophan and Nicotinamide. Phytotherapy Res.23 (3), 335–346. 10.1002/ptr.2628
356
PetpiroonN.SuktapC.PongsamartS.ChanvorachoteP.SukrongS. (2015). Kaempferol-3-O-rutinoside from Afgekia Mahidoliae Promotes Keratinocyte Migration through FAK and Rac1 Activation. J. Nat. Medicines69 (3), 340–348. 10.1007/s11418-015-0899-3
357
PetrusA. A.SeetharamanT. R. (2005). Antioxidant Flavone C-Biosides from the Aerial Parts of Alternanthera Pungens. Indian J. Pharm. Sci.67 (2), 187–192.
358
PetrusA. J. A.KalpanaK.DeviA. B. (2014b). Antioxidant Capacity and Lipophilic Constitution of Alternanthera Bettzickiana Flower Extract. Oriental J. Chem.30 (2), 491–499. 10.13005/ojc/300212
359
PetrusA.KalpanaK.DeviA. (2014a). Foliar Biophenolic Antioxidant Metabolites of Alternanthera Bettzickiana. Oriental J. Chem.30 (3), 1197–1203. 10.13005/ojc/300334
360
PeungvichaP.TemsiririrkkulR.PrasainJ. K.TezukaY.KadotaS.ThirawarapanS. S.et al (1998). 4-Hydroxybenzoic Acid: a Hypoglycemic Constituent of Aqueous Extract of Pandanus Odorus Root. J. Ethnopharmacology62 (1), 79–84. 10.1016/s0378-8741(98)00061-0
361
PizzoS. V.WangJ.FangX.GeL.CaoF.ZhaoL.et al (2018). Antitumor, Antioxidant and Anti-inflammatory Activities of Kaempferol and its Corresponding Glycosides and the Enzymatic Preparation of Kaempferol. Plos One13 (5), 1. 10.1371/journal.pone.0197563
362
PonnulakshmiR.ShyamaladeviB.VijayalakshmiP.SelvarajJ. (2019). In Silicoandin Vivoanalysis to Identify the Antidiabetic Activity of Beta Sitosterol in Adipose Tissue of High Fat Diet and Sucrose Induced Type-2 Diabetic Experimental Rats. Toxicol. Mech. Methods29 (4), 276–290. 10.1080/15376516.2018.1545815
363
PradhanM.SuriC.ChoudharyS.NaikP. K.LopusM. (2016). Elucidation of the Anticancer Potential and Tubulin Isotype-specific Interactions of β-sitosterol. J. Biomol. Struct. Dyn.36 (1), 195–208. 10.1080/07391102.2016.1271749
364
PragasamS. J.VenkatesanV.RasoolM. (2012). Immunomodulatory and Anti-inflammatory Effect of P-Coumaric Acid, a Common Dietary Polyphenol on Experimental Inflammation in Rats. Inflammation36 (1), 169–176. 10.1007/s10753-012-9532-8
365
PratiwiR.NantasenamatC.RuankhamW.SuwanjangW.PrachayasittikulV.PrachayasittikulS.et al (2021). Mechanisms and Neuroprotective Activities of Stigmasterol against Oxidative Stress-Induced Neuronal Cell Death via Sirtuin Family. Front. Nutr.8. 10.3389/fnut.2021.648995
366
Prince Vijeya SinghJ.SelvendiranK.Mumtaz BanuS.PadmavathiR.SakthisekaranD. (2004). Protective Role of Apigenin on the Status of Lipid Peroxidation and Antioxidant Defense against Hepatocarcinogenesis in Wistar Albino Rats. Phytomedicine11 (4), 309–314. 10.1078/0944711041495254
367
PriyadarsiniK. I.KhopdeS. M.KumarS. S.MohanH. (2002). Free Radical Studies of Ellagic Acid, a Natural Phenolic Antioxidant. J. Agric. Food Chem.50 (7), 2200–2206. 10.1021/jf011275g
368
PujariR. R.BandawaneD. D. (2021). Hepatoprotective Activity of Gentisic Acid on 5-Fluorouracil-Induced Hepatotoxicity in Wistar Rats. Turkish J. Pharm. Sci.18 (3), 332–338. 10.4274/tjps.galenos.2020.95870
369
PulipatiS.BabuP. S. (2020). In-vitro Antibacterial Potential of Alternanthera Phyloxeroides (Mart) Griseb. Against Multi-Drug Resistant Uropathogens. Int. J. Pharm. Sci. Res.11 (8), 3834–3840. 10.13040/ijpsr.0975-8232.11(8).3834-40
370
PulipatiS.BabuS. P.SreeN. B.KumarU. E.ShaheelaS.KrishnaJ. M.et al (2016). Phytochemical Analysis and Antimicrobial Investigations of Ethanolic Leaf Extract of Alternanthera Philoxeroides (Mart.) Griseb. World J. Pharm. Pharm. Sci.5 (2), 1122–1129.
371
PulipatiS.BabuS. P.Sri DeviB.Rama DeviG.BhanujaM. (2015). Pharmacognostic Studies of Alternanthera Philoxeroides (Mart.) Griseb. J. Pharmacognosy Phytochemistry4 (2), 202–204.
372
QianL.SuW.WangY.DangM.ZhangW.WangC. (2019). Synthesis and Characterization of Gold Nanoparticles from Aqueous Leaf Extract of Alternanthera Sessilis and its Anticancer Activity on Cervical Cancer Cells (HeLa). Artif. Cell Nanomedicine, Biotechnol.47 (1), 1173–1180. 10.1080/21691401.2018.1549064
373
QianW.LiuM.FuY.ZhangJ.LiuW.LiJ.et al (2020). Antimicrobial Mechanism of Luteolin against Staphylococcus aureus and Listeria Monocytogenes and its Antibiofilm Properties. Microb. Pathogenesis142. 10.1016/j.micpath.2020.104056
374
RagasaC. Y.TremorN.RideoutJ. A. (2010). Ionone Derivatives from Alternanthera Sessilis. J. Asian Nat. Prod. Res.4 (2), 109–115. 10.1080/10286020290027380
375
RagoneM. I.SellaM.ConfortiP.VolontéM. G.ConsoliniA. E. (2007). The Spasmolytic Effect of Aloysia Citriodora, Palau (South American Cedrón) Is Partially Due to its Vitexin but Not Isovitexin on Rat Duodenums. J. Ethnopharmacology113 (2), 258–266. 10.1016/j.jep.2007.06.003
376
RajamuruganR.DeepaV.SivashanmugamM.RaghavanC. M. (2013). Phytochemistry, Antioxidant and Antibacterial Activities of Medicinal Plants - A Comparative Study. Int. J. Curr. Res. Rev.5, 8–19.
377
RaoK. V. R.RaoK. R. S. S.NelsonR.NagaiahK.ReddyV. J. S. (2011). Hypoglycaemic and Antidiabetic Effect of Alternanthera Sessilis in normal and Streptozotocin (STZ)-induced Rat. J. Glob. Trends Pharm. Sci.2 (3), 325–335.
378
RaoR. R. (2000). Synoptic Flora of Mysore District. Mysore, India. Today and Tomorrow’s Printer and Publisher.
379
RattanathongkomA.LeeJ.-B.HayashiK.SripanidkulchaiB.-o.KanchanapoomT.HayashiT. (2009). Evaluation of Chikusetsusaponin IV a Isolated fromAlternanthera Philoxeroidesfor its Potency against Viral Replication. Planta Med.75 (08), 829–835. 10.1055/s-0029-1185436
380
RaufA.ImranM.KhanI. A.ur-RehmanM.GilaniS. A.MehmoodZ.et al (2018). Anticancer Potential of Quercetin: A Comprehensive Review. Phytotherapy Res.32 (11), 2109–2130. 10.1002/ptr.6155
381
RawaniA.PalS.ChandraG. (2011). Evaluation of Antimicrobial Properties of Four Plant Extracts against Human Pathogens. Asian Pac. J. Trop. Biomed.1 (1), S71–S75. 10.1016/s2221-1691(11)60127-5
382
Raybaudi-MassiliaR. M.Mosqueda-MelgarJ.Martín-BellosoO. (2009). Antimicrobial Activity of Malic Acid against Listeria Monocytogenes, Salmonella Enteritidis and Escherichia coli O157:H7 in Apple, Pear and Melon Juices. Food Control20 (2), 105–112. 10.1016/j.foodcont.2008.02.009
383
RayeesS.KumarA.RasoolS.KaiserP.SattiN. K.SangwanP. L.et al (2013). Ethanolic Extract ofAlternanthera sessilis(AS-1) Inhibits IgE-Mediated Allergic Response in RBL-2H3 Cells. Immunological Invest.42 (6), 470–480. 10.3109/08820139.2013.789909
384
RehmanN.-u.MehmoodM. H.AlkharfyK. M.GilaniA.-H. (2012). Studies on Antidiarrheal and Antispasmodic Activities of Lepidium Sativum Crude Extract in Rats. Phytotherapy Res.26 (1), 136–141. 10.1002/ptr.3642
385
RezaH. M.AlamM. A.SarkerS. D.NaharL.HossainH.ShillM. C.et al (2019). Alternanthera Bicolor Produces Hypoglycemic Effect in Alloxan-Induced Diabetic Mice through its Antioxidant Activity. Dhaka Univ. J. Pharm. Sci.18 (1), 49–60. 10.3329/dujps.v18i1.41431
386
RobertoD.MicucciP.SebastianT.GracielaF.AnesiniC. (2009). Antioxidant Activity of Limonene on Normal Murine Lymphocytes: Relation to H2O2Modulation and Cell Proliferation. Basic Clin. Pharmacol. Toxicol.106 (1), 38–44. 10.1111/j.1742-7843.2009.00467.x
387
RomanovaD.VachalkovaA.CipakL.OvesnaZ.RaukoP. (2001). Study of Antioxidant Effect of Apigenin, Luteolin and Quercetin by DNA Protective Method. Neoplasma48 (2), 104–107.
388
RosaS. I. G.Rios-SantosF.BalogunS. O.MartinsD. T. d. O. (2016). Vitexin Reduces Neutrophil Migration to Inflammatory Focus by Down-Regulating Pro-inflammatory Mediators via Inhibition of P38, ERK1/2 and JNK Pathway. Phytomedicine23 (1), 9–17. 10.1016/j.phymed.2015.11.003
389
RowleyT. J.McKinstryA.GreenidgeE.SmithW.FloodP. (2010). Antinociceptive and Anti-inflammatory Effects of Choline in a Mouse Model of Postoperative Pain. Br. J. Anaesth.105 (2), 201–207. 10.1093/bja/aeq113
390
RufinoA. T.RibeiroM.SousaC.JudasF.SalgueiroL.CavaleiroC.et al (2015). Evaluation of the Anti-inflammatory, Anti-catabolic and Pro-anabolic Effects of E-Caryophyllene, Myrcene and Limonene in a Cell Model of Osteoarthritis. Eur. J. Pharmacol.750, 141–150. 10.1016/j.ejphar.2015.01.018
391
RukkumaniR.ArunaK.Suresh VarmaP.Padmanabhan MenonV. (2004). Hepatoprotective Role of Ferulic Acid: A Dose-dependent Study. J. Med. Food7 (4), 456–461. 10.1089/jmf.2004.7.456
392
SaijaA. (2003). In Vitro' Antioxidant and Photoprotective Properties and Interaction with Model Membranes of Three New Quercetin Esters. Eur. J. Pharmaceutics Biopharmaceutics56 (2), 167–174. 10.1016/s0939-6411(03)00101-2
393
SalvadorM. J.DiasD. A. (2004). Flavone C-Glycosides from Alternanthera Maritima (Mart.) St. Hil. (Amaranthaceae). Biochem. Syst. Ecol.32 (1), 107–110. 10.1016/s0305-1978(03)00180-7
394
SalvadorM. J.FerreiraE. O.Mertens-TalcottS. U.De CastroW. V.ButterweckV.DerendorfH.et al (2006). Isolation and HPLC Quantitative Analysis of Antioxidant Flavonoids from Alternanthera Tenella Colla. Z. für Naturforschung C61 (1-2), 19–25. 10.1515/znc-2006-1-204
395
SalvadorM. J.PereiraP. S.FrançaS. C.CandidoR. C.ItoI. Y.DiasD. A. (2009). Bioactive Chemical Constituents and Comparative Antimicrobial Activity of Callus Culture and Adult Plant Extracts from Alternanthera Tenella. Z. für Naturforschung C64 (5-6), 373–381. 10.1515/znc-2009-5-612
396
SamudralaP.AugustineB.KasalaE.BodduluruL.BaruaC.LahkarM. (2015). Evaluation of Antitumor Activity and Antioxidant Status of Alternanthera Brasiliana against Ehrlich Ascites Carcinoma in Swiss Albino Mice. Pharmacognosy Res.7 (1), 1. 10.4103/0974-8490.147211
397
Sánchez-MendozaM. E.ArrietaJ.NavarreteA. (2008). Role of Prostaglandins, Nitric Oxide, Sulfhydryls and Capsaicin-Sensitive Neurons in Gastroprotection of Stigmasterol and β-Sitosterol. Nat. Product. Commun.3 (4), 1. 10.1177/1934578x0800300406
398
SanokoR.SperanzaG.PizzaC.DetommasiN. (1999). Triterpene Saponins from Alternanthera Repens. Phytochemistry51 (8), 1043–1047. 10.1016/s0031-9422(99)00046-1
399
SantosA. L.YamamotoE. S.PasseroL. F. D.LaurentiM. D.MartinsL. F.LimaM. L.et al (2017). Antileishmanial Activity and Immunomodulatory Effects of Tricin Isolated from Leaves of Casearia Arborea (Salicaceae). Chem. Biodiversity14 (5), 1. 10.1002/cbdv.201600458
400
SantosC. C. d. M. P.SalvadoriM. S.MotaV. G.CostaL. M.de AlmeidaA. A. C.de OliveiraG. A. L.et al (2013). Antinociceptive and Antioxidant Activities of Phytol In Vivo and In Vitro Models. Neurosci. J.2013, 1–9. 10.1155/2013/949452
401
Santos da SilvaG. N.PozzattiP.RigattiF.HörnerR.Hartz AlvesS.MallmannC. A.et al (2015). Antimicrobial Evaluation of Sesquiterpene Alpha-Curcumene and its Synergism with Imipenem. J. Microbiol. Biotechnol. Food Sci.04 (05), 434–436. 10.15414/jmbfs.2015.4.5.434-436
402
SaqibF.JanbazK. H. (2016). Rationalizing Ethnopharmacological Uses of Alternanthera Sessilis: A Folk Medicinal Plant of Pakistan to Manage Diarrhea, Asthma and Hypertension. J. Ethnopharmacology182, 110–121. 10.1016/j.jep.2016.02.017
403
SathishkumarP.VennilaK.JayakumarR.YusoffA. R. M.HadibarataT.PalvannanT. (2016). Phyto-synthesis of Silver Nanoparticles Using Alternanthera Tenella Leaf Extract: an Effective Inhibitor for the Migration of Human Breast Adenocarcinoma (MCF-7) Cells. Bioproc. Biosyst. Eng.39 (4), 651–659. 10.1007/s00449-016-1546-4
404
SathyaS.ManogariB. G.ThamaraiselviK.VaideviS.RuckmaniK.DeviK. P. (2020). Phytol Loaded PLGA Nanoparticles Ameliorate Scopolamine-Induced Cognitive Dysfunction by Attenuating Cholinesterase Activity, Oxidative Stress and Apoptosis in Wistar Rat. Nutr. Neurosci.8, 1–17. 10.1080/1028415x.2020.1764290
405
SatoY.ItagakiS.KurokawaT.OguraJ.KobayashiM.HiranoT.et al (2011). In Vitro and In Vivo Antioxidant Properties of Chlorogenic Acid and Caffeic Acid. Int. J. Pharmaceutics403 (1-2), 136–138. 10.1016/j.ijpharm.2010.09.035
406
SchallenbergerC.VieiraV.KraiJ. S.MorissoF.SuyenagaE.TavaresR. G.et al (2017). Anticonvulsant Effect of Alternanthera Brasiliana Extract on Pentylenetetrazole-Induced Seizures in Rats. J. Neurosci. Clin. Res.2 (1), 1–3.
407
ScheepensA.BissonJ.-F.SkinnerM. (2014). p-Coumaric Acid Activates the GABA-A Receptor In Vitro and Is Orally Anxiolytic In Vivo. Phytotherapy Res.28 (2), 207–211. 10.1002/ptr.4968
408
SekarK. C. (2012). Invasive Alien Plants of Indian Himalayan Region—Diversity and Implication. Am. J. Plant Sci.03 (02), 177–184. 10.4236/ajps.2012.32021
409
SemenyaS. S.PotgieterM. J. (2014). Bapedi Traditional Healers in the Limpopo Province, South Africa: Their Socio-Cultural Profile and Traditional Healing Practice. J. Ethnobiol. Ethnomedicine10 (1), 1. 10.1186/1746-4269-10-4
410
ShahidF.FarooquiZ.KhanF. (2018). Cisplatin-induced Gastrointestinal Toxicity: An Update on Possible Mechanisms and on Available Gastroprotective Strategies. Eur. J. Pharmacol.827, 49–57. 10.1016/j.ejphar.2018.03.009
411
ShanC.MiaoF. (2022). Immunomodulatory and Antioxidant Effects of Hydroxytyrosol in Cyclophosphamide-Induced Immunosuppressed Broilers. Poult. Sci.101 (1), 1. 10.1016/j.psj.2021.101516
412
SharmaA.SanadhyaI.BhotM.VargheseJ. (2013). Evaluation of Antioxidant Potential of Alternanthera Sessilis (L.) DC. Res. J. Pharmacognosy Phytochemistry5 (4), 194–198. 10.4103/0974-8490.118767
413
SharmaN.BiswasS.Al-DayanN.AlhegailiA. S.SarwatM. (2021). Antioxidant Role of Kaempferol in Prevention of Hepatocellular Carcinoma. Antioxidants (Basel)10 (9), 1. 10.3390/antiox10091419
414
ShiC.ZhangX.SunY.YangM.SongK.ZhengZ.et al (2016). Antimicrobial Activity of Ferulic Acid AgainstCronobacter Sakazakiiand Possible Mechanism of Action. Foodborne Pathog. Dis.13 (4), 196–204. 10.1089/fpd.2015.1992
415
ShiT.BianX.YaoZ.WangY.GaoW.GuoC. (2020). Quercetin Improves Gut Dysbiosis in Antibiotic-Treated Mice. Food Funct.11 (9), 8003–8013. 10.1039/d0fo01439g
416
SinghB.SharmaV.Singh IsharM.SharmaA. (2013). Bioactivity Guided Isolation of Quercetin as Anxiolytic Compound from Elaeocarpus Ganitrus Beads. Nat. Prod. J.3 (3), 224–229. 10.2174/22103155113039990010
417
SinghD.SharmaS. K.RaniR.MishraS.SharmaR. A. (2011). Kaempferol-7-O-Glucoside and Their Antimicrobial Screening Isolate from Cassia Renigera Wall. Int. J. Pharm. Clin. Res.3 (2), 30–34.
418
SinghR.SinghB.SinghS.KumarN.KumarS.AroraS. (2008). Anti-free Radical Activities of Kaempferol Isolated from Acacia Nilotica (L.) Willd. Ex. Del. Toxicol. Vitro22 (8), 1965–1970. 10.1016/j.tiv.2008.08.007
419
SinghS. S.RaiS. N.BirlaH.ZahraW.KumarG.GeddaM. R.et al (2018). Effect of Chlorogenic Acid Supplementation in MPTP-Intoxicated Mouse. Front. Pharmacol.9. 10.3389/fphar.2018.00757
420
SinglaR. K.HeX.ChopraH.TsagkarisC.ShenL.KamalM. A.et al (2021). Natural Products for the Prevention and Control of the COVID-19 Pandemic: Sustainable Bioresources. Front. Pharmacol.12, 758159. 10.3389/fphar.2021.758159
421
SinglaR. K.ScottiL.DubeyA. K. (2017). In Silico Studies Revealed Multiple Neurological Targets for the Antidepressant Molecule Ursolic Acid. Curr. Neuropharmacology15 (8), 1. 10.2174/1570159x14666161229115508
422
SinglaR. K.ShenB. (2020). In Silico ADMET Evaluation of Natural DPP-IV Inhibitors for Rational Drug Design against Diabetes. Curr. Drug Metab.21 (10), 768–777. 10.2174/1389200221999200901202945
423
SiopaF.FigueiredoT.FradeR. F. M.NetoI.MeirinhosA.ReisC. P.et al (2016). Choline-Based Ionic Liquids: Improvement of Antimicrobial Activity. ChemistrySelect1 (18), 5909–5916. 10.1002/slct.201600864
424
SivakumarR.SunmathiD. (2016). Phytochemical Screening and Antimicrobial Activity of Ethanolic Leaf Extract of Alternanthera Sessilis (L.) R.BR. EX DC and Alternanthera Philoxeroides (Mart.) Griseb. Eur. J. Pharm. Med. Res.3 (3), 409–412.
425
SocałaK.NieoczymD.PierógM.WlaźP. (2015). α-Spinasterol, a TRPV1 Receptor Antagonist, Elevates the Seizure Threshold in Three Acute Seizure Tests in Mice. J. Neural Transm.122 (9), 1239–1247. 10.1007/s00702-015-1391-7
426
SonarV. P.FoisB.DistintoS.MaccioniE.MeledduR.CottigliaF.et al (2019). Ferulic Acid Esters and Withanolides: In Search of Withania Somnifera GABAA Receptor Modulators. J. Nat. Prod.82 (5), 1250–1257. 10.1021/acs.jnatprod.8b01023
427
SongJ. H.ShimJ. K.ChoiH. J. (2011). Quercetin 7-rhamnoside Reduces Porcine Epidemic Diarrhea Virus Replication via Independent Pathway of Viral Induced Reactive Oxygen Species. Virol. J.8 (1), 1. 10.1186/1743-422x-8-460
428
SouzaJ. G.TomeiR. R.KanashiroA.KabeyaL. M.AzzoliniA. E. C. S.DiasD. A.et al (2007). Ethanolic Crude Extract and Flavonoids Isolated from Alternanthera Maritima: Neutrophil Chemiluminescence Inhibition and Free Radical Scavenging Activity. Z. für Naturforschung C62 (5-6), 339–347. 10.1515/znc-2007-5-604
429
SuganyaD.BanupriyaR.maheswari AU.ElumalaiS. (2019). Studies on Biological Activity of Aqueous Extract of Alternanthera Sessilis (Linn) for Developing Potential Herbal Drug Formulation of Ocular Diseases. Med. Aromatic Plants08 (01), 1. 10.35248/2167-0412.19.8.327
430
SunZ.-R.LiuH.-R.HuD.FanM.-S.WangM.-Y.AnM.-F.et al (2021). Ellagic Acid Exerts Beneficial Effects on Hyperuricemia by Inhibiting Xanthine Oxidase and NLRP3 Inflammasome Activation. J. Agric. Food Chem.69 (43), 12741–12752. 10.1021/acs.jafc.1c05239
431
SundarR. D. V.RaviL.MythiliS. (2019). Discovery of New Anti-fungal Phytochemical PDHC (Propane-diyl-bis-hexahydro-isochromene) Isolated from Alternanthera Sessilis Leaves. Int. J. Pharm. Sci. Res.10 (3), 1148–1159. 10.13040/ijpsr.0975-8232.10(3).1136-47
432
SunmathiD.SivakumarR.RavikumarK. (2016). In Vitro anti-inflammatory and Antiarthritic Activity of Ethanolic Leaf Extract of Alternanthera sessilis(L.) R.BR. Ex DC and Alternanthera Philoxeroides (Mart.) Griseb. Int. J. Adv. Pharm. Biol. Chem.5 (2), 109–115.
433
SuzukiK.NomuraI.NinomiyaM.TanakaK.KoketsuM. (2018). Synthesis and Antimicrobial Activity of β-carboline Derivatives with N2-Alkyl Modifications. Bioorg. Med. Chem. Lett.28 (17), 2976–2978. 10.1016/j.bmcl.2018.06.050
434
SwarnalathaS.UmamaheswariA.PuratchikodyA. (2015). Immunomodulatory Activity of Kaempferol 5-O-β-D-Glucopyranoside from Indigofera Aspalathoides Vahl Ex DC. (Papilionaceae). Med. Chem. Res.24 (7), 2889–2897. 10.1007/s00044-015-1341-9
435
TanK. K.KimK. H. (2013). Alternanthera sessilisRed Ethyl Acetate Fraction Exhibits Antidiabetic Potential on Obese Type 2 Diabetic Rats. Evidence-Based Complement. Altern. Med.2013, 1–8. 10.1155/2013/845172
436
TangT.WangS.CaiT.ChengZ.QiS.QiZ. (2019). Calenduloside E Inhibits Lipopolysaccharide-Induced Inflammatory Response by Inhibiting Activation of ROS-Mediated JAK1-Stat3 Signaling Pathway in RAW264.7 Cells. Nan Fang Yi Ke Da Xue Xue Bao39 (8), 904–910. 10.12122/j.issn.1673-4254.2019.08.05
437
ThanabhornS.JaijoyK.ThamareeS.IngkaninanK.PanthongA. (2005). Acute and Subacute Toxicities of the Ethanol Extract from Alternanthera Philoxeroides Griseb. Pharm. Sci. Asia31 (1-2), 7–14.
438
ThirukumaranP.ManoharanR. K.ParveenA. S.AtchudanR.KimS.-C. (2019). Sustainability and Antimicrobial Assessments of Apigenin Based Polybenzoxazine Film. Polymer172, 100–109. 10.1016/j.polymer.2019.03.048
439
TianD.GaoQ.LinJ.ChangZ.WangY.ShiY.et al (2021). Uncovering the Mechanism of the Shenzhi Jiannao Formula against Vascular Dementia Using a Combined Network Pharmacology Approach and Molecular Biology. Phytomedicine90. 10.1016/j.phymed.2021.153637
440
TiwariA.JyothiA.TejeswiniV.MadhusudanaK.KumarD.ZehraA.et al (2013). Mitigation of Starch and Glucose-Induced Postprandial Glycemic Excursion in Rats by Antioxidant-Rich green-leafy Vegetables′ Juice. Pharmacognosy Mag.9 (36), 1. 10.4103/0973-1296.117872
441
TiwariM.KakkarP. (2009). Plant Derived Antioxidants – Geraniol and Camphene Protect Rat Alveolar Macrophages against T-BHP Induced Oxidative Stress. Toxicol. Vitro23 (2), 295–301. 10.1016/j.tiv.2008.12.014
442
TripathiY. B.SharmaM.ManickamM. (1997). Rubiadin, a New Antioxidant from Rubia Cordifolia. Indian J. Biochem. Biophys.34 (3), 302–306.
443
TsengH.-L.LiC.-J.HuangL.-H.ChenC.-Y.TsaiC.-H.LinC.-N.et al (2012). Quercetin 3-O-Methyl Ether Protects FL83B Cells from Copper Induced Oxidative Stress through the PI3K/Akt and MAPK/Erk Pathway. Toxicol. Appl. Pharmacol.264 (1), 104–113. 10.1016/j.taap.2012.07.022
444
TuY.ChengS.-x.SunH.-t.MaT.-z.ZhangS. (2012). Ferulic Acid Potentiates Pentobarbital-Induced Sleep via the Serotonergic System. Neurosci. Lett.525 (2), 95–99. 10.1016/j.neulet.2012.07.068
445
TukunA. B.ShaheenN.BanuC. P.MohiduzzamanM.IslamS.BegumM. (2014). Antioxidant Capacity and Total Phenolic Contents in Hydrophilic Extracts of Selected Bangladeshi Medicinal Plants. Asian Pac. J. Trop. Med.7, S568–S573. 10.1016/s1995-7645(14)60291-1
446
UteshevD. B.KostriukovE. B.KarabinenkoA. A.KovalevaV. L.MakarovaO. V.StorozhakovG. I. (2000). The Anti-inflammatory Activity of Intal and Beta-Carotene in a Model of Experimental Granulomatous Lung Inflammation. Patol Fiziol Eksp Ter2, 19–22.
447
VaniM.RahamanS. A.Prameela RaniA. (2018). Detection and Quantification of Major Phytochemical Markers for Standardization of Talinum Portulacifolium, Gomphrena Serrata, Alternanthera Sessilis and Euphorbia Heterophylla by HPLC. Pharmacognosy J.10 (3), 439–446. 10.5530/pj.2018.3.72
448
VasconcelosM. A. L.RoyoV. A.FerreiraD. S.CrottiA. E. M.e SilvaM. L. A.CarvalhoJ. C. T.et al (2006). In Vivo Analgesic and Anti-inflammatory Activities of Ursolic Acid and Oleanoic Acid from Miconia Albicans (Melastomataceae). Z. für Naturforschung C61 (7-8), 477–482. 10.1515/znc-2006-7-803
449
VauzourD.CoronaG.SpencerJ. P. E. (2010). Caffeic Acid, Tyrosol and P-Coumaric Acid Are Potent Inhibitors of 5-S-Cysteinyl-Dopamine Induced Neurotoxicity. Arch. Biochem. Biophys.501 (1), 106–111. 10.1016/j.abb.2010.03.016
450
VelikaB.KronI. (2012). Antioxidant Properties of Benzoic Acid Derivatives against Superoxide Radical. Free Radicals Antioxid.2 (4), 62–67. 10.5530/ax.2012.4.11
451
VennilaV.NivethaR. (2015). Screening the In Vitro Anthelmintic Activity of Alternanthera Sessilis Leaves. World J. Pharm. Pharm. Sci.4 (4), 1402–1415.
452
VenturiniC. L.MachoA.ArunachalamK.de AlmeidaD. A. T.RosaS. I. G.PavanE.et al (2018). Vitexin Inhibits Inflammation in Murine Ovalbumin-Induced Allergic Asthma. Biomed. Pharmacother.97, 143–151. 10.1016/j.biopha.2017.10.073
453
VessalM.HemmatiM.VaseiM. (2003). Antidiabetic Effects of Quercetin in Streptozocin-Induced Diabetic Rats. Comp. Biochem. Physiol. C: Toxicol. Pharmacol.135 (3), 357–364. 10.1016/s1532-0456(03)00140-6
454
VidhyaT.SujiT.DhatchayaniR.PriyaC. L.Bhaskara RaoK. V. (2015). Evaluation of In-Vitro Antioxidant, Antimicrobial Activities and GC-MS Analysis of Alternanthera Bettzickiana Linn. Leaf Extracts. Int. J. Pharmacognosy Phytochem. Res.7 (6), 1072–1079. 10.9734/ijbcrr/2015/17241
455
VieiraC.FetzerS.SauerS.EvangelistaS.AverbeckB.KressM.et al (2001). Pro- and Anti-inflammatory Actions of Ricinoleic Acid: Similarities and Differences with Capsaicin. Naunyn-Schmiedeberg's Arch. Pharmacol.364 (2), 87–95. 10.1007/s002100100427
456
VuurenS. F. v.ViljoenA. M. (2007). Antimicrobial Activity of Limonene Enantiomers and 1,8-cineole Alone and in Combination. Flavour Fragrance J.22 (6), 540–544. 10.1002/ffj.1843
457
WalkerC. I. B.OliveiraS. M.TonelloR.RossatoM. F.da Silva BrumE.FerreiraJ.et al (2017). Anti-nociceptive Effect of Stigmasterol in Mouse Models of Acute and Chronic Pain. Naunyn-Schmiedeberg's Arch. Pharmacol.390 (11), 1163–1172. 10.1007/s00210-017-1416-x
458
WalterT. M.MerishS.TamizhamuthuM. (2014). Review of Alternanthera Sessilis with Reference to Traditional Siddha Medicine. Int. J. Pharmacognosy Phytochem. Res.6 (2), 249–254.
459
WangJ.HuangM.YangJ.MaX.ZhengS.DengS.et al (2017). Anti-diabetic Activity of Stigmasterol from Soybean Oil by Targeting the GLUT4 Glucose Transporter. Food Nutr. Res.61 (1), 1. 10.1080/16546628.2017.1364117
460
WangM.ShiY.GuoY.ChenY.ZhaoC.ZhouY.et al (2021). Nonadiabatic Dynamics Mechanisms of Natural UV Photoprotection Ompounds Chlorogenic Acid and Isochlorogenic Acid a: Double Conjugated Structures but Single Photoexcited Channel. J. Mol. Liquids324. 10.1016/j.molliq.2020.114725
461
WangM.SunJ.JiangZ.XieW.ZhangX. (2015a). Hepatoprotective Effect of Kaempferol against Alcoholic Liver Injury in Mice. Am. J. Chin. Med.43 (02), 241–254. 10.1142/s0192415x15500160
462
WangS.-H.ChenC.-S.HuangS.-H.YuS.-H.LaiZ.-Y.HuangS.-T.et al (2009). Hydrophilic Ester-Bearing Chlorogenic Acid Binds to a Novel Domain to Inhibit Xanthine Oxidase. Planta Med.75 (11), 1237–1240. 10.1055/s-0029-1185521
463
WangW.GuoJ.ZhangJ.PengJ.LiuT.XinZ. (2015b). Isolation, Identification and Antioxidant Activity of Bound Phenolic Compounds Present in rice Bran. Food Chem.171, 40–49. 10.1016/j.foodchem.2014.08.095
464
WangX.YeX.-l.LiuR.ChenH.-L.BaiH.LiangX.et al (2010). Antioxidant Activities of Oleanolic Acid In Vitro: Possible Role of Nrf2 and MAP Kinases. Chemico-Biological Interactions184 (3), 328–337. 10.1016/j.cbi.2010.01.034
465
WangY.TangC.ZhangH. (2015c). Hepatoprotective Effects of Kaempferol 3-O-Rutinoside and Kaempferol 3-O-Glucoside from Carthamus tinctorius L. On CCl4-Induced Oxidative Liver Injury in Mice. J. Food Drug Anal.23 (2), 310–317. 10.1016/j.jfda.2014.10.002
466
WangY.ZhangG.PanJ.GongD. (2015d). Novel Insights into the Inhibitory Mechanism of Kaempferol on Xanthine Oxidase. J. Agric. Food Chem.63 (2), 526–534. 10.1021/jf505584m
467
WiegmannD.KoppermannS.WirthM.NiroG.LeyererK.DuchoC. (2016). Muraymycin Nucleoside-Peptide Antibiotics: Uridine-Derived Natural Products as lead Structures for the Development of Novel Antibacterial Agents. Beilstein J. Org. Chem.12, 769–795. 10.3762/bjoc.12.77
468
WinterA. N.BrennerM. C.PunessenN.SnodgrassM.ByarsC.AroraY.et al (2017). Comparison of the Neuroprotective and Anti-inflammatory Effects of the Anthocyanin Metabolites, Protocatechuic Acid and 4-Hydroxybenzoic Acid. Oxidative Med. Cell Longevity2017, 1–13. 10.1155/2017/6297080
469
WittM. R.Westh-HansenS. E.RasmussenP. B.HastrupS.NielsenM. (2002). Unsaturated Free Fatty Acids Increase Benzodiazepine Receptor Agonist Binding Depending on the Subunit Composition of the GABAA Receptor Complex. J. Neurochem.67 (5), 2141–2145. 10.1046/j.1471-4159.1996.67052141.x
470
WuC.-H.HsiehH.-T.LinJ.-A.YenG.-C. (2013). Alternanthera Paronychioides Protects Pancreatic β-cells from Glucotoxicity by its Antioxidant, Antiapoptotic and Insulin Secretagogue Actions. Food Chem.139 (1-4), 362–370. 10.1016/j.foodchem.2013.01.026
471
XueW.WangX.TangH.SunF.ZhuH.HuangD.et al (2020). Vitexin Attenuates Myocardial Ischemia/reperfusion Injury in Rats by Regulating Mitochondrial Dysfunction Induced by Mitochondrial Dynamics Imbalance. Biomed. Pharmacother.124. 10.1016/j.biopha.2020.109849
472
YangH.HuangJ.MaoY.WangL.LiR.HaC. (2019). Vitexin Alleviates Interleukin‐1β‐induced Inflammatory Responses in Chondrocytes from Osteoarthritis Patients: Involvement of HIF‐1α Pathway. Scand. J. Immunol.90 (2), 1. 10.1111/sji.12773
473
YangH.QuZ.ZhangJ.HuoL.GaoJ.GaoW. (2016). Ferulic Acid Ameliorates Memory Impairment in D-Galactose-Induced Aging Mouse Model. Int. J. Food Sci. Nutr.67 (7), 806–817. 10.1080/09637486.2016.1198890
474
YangJ.SunX.-Q.YanS.-Y.PanW.-J.ZhangM.-X.CaiQ.-N. (2017). Interaction of Ferulic Acid with Glutathione S-Transferase and Carboxylesterase Genes in the Brown Planthopper, Nilaparvata Lugens. J. Chem. Ecol.43 (7), 693–702. 10.1007/s10886-017-0859-3
475
YaoY.HanD. D.ZhangT.YangZ. (2010). Quercetin Improves Cognitive Deficits in Rats with Chronic Cerebral Ischemia and Inhibits Voltage-dependent Sodium Channels in Hippocampal CA1 Pyramidal Neurons. Phytotherapy Res.24 (1), 136–140. 10.1002/ptr.2902
476
YapC. H.Mat JunitS.Abdul AzizA.KongK. W. (2019). Multiple Extraction Conditions to Produce Phytochemical- and Antioxidant-Rich Alternanthera Sessilis (Red) Extracts that Attenuate Lipid Accumulation in Steatotic HepG2 Cells. Food Biosci.32. 10.1016/j.fbio.2019.100489
477
YeC.-J.LiS.-A.ZhangY.LeeW.-H. (2019). Geraniol Targets KV1.3 Ion Channel and Exhibits Anti-inflammatory Activity In Vitro and In Vivo. Fitoterapia139. 10.1016/j.fitote.2019.104394
478
YoshidaY.NikiE. (2003). Antioxidant Effects of Phytosterol and its Components. J. Nutr. Sci. Vitaminology49 (4), 277–280. 10.3177/jnsv.49.277
479
YukJ. E.WooJ. S.YunC.-Y.LeeJ.-S.KimJ.-H.SongG.-Y.et al (2007). Effects of Lactose-β-Sitosterol and β-sitosterol on Ovalbumin-Induced Lung Inflammation in Actively Sensitized Mice. Int. Immunopharmacology7 (12), 1517–1527. 10.1016/j.intimp.2007.07.026
480
ZavalaM. A.PérezS.PérezC.VargasR.PérezR. M. (1998). Antidiarrhoeal Activity of Waltheria Americana, Commelina Coelestis and Alternanthera Repens. J. Ethnopharmacology61 (1), 41–47. 10.1016/s0378-8741(98)00014-2
481
ZhangM.SwartsS. G.YinL.LiuC.TianY.CaoY.et al (2011). “Antioxidant Properties of Quercetin,” in Oxygen Transport to Tissue XXXII, 283–289. 10.1007/978-1-4419-7756-4_38
482
ZhangQ.FanZ.XueW.SunF.ZhuH.HuangD.et al (2021). Vitexin Regulates Epac and NLRP3 and Ameliorates Chronic Cerebral Hypoperfusion Injury. Can. J. Physiol. Pharmacol.99 (10), 1079–1087. 10.1139/cjpp-2021-0034
483
ZhangX.-Y.ShenJ.ZhouY.WeiZ.-P.GaoJ.-M. (2016). Insecticidal Constituents from Buddlej Aalbiflora Hemsl. Nat. Product. Res.31 (12), 1446–1449. 10.1080/14786419.2016.1247080
484
ZhangX.LiP.GuoS.WangS.LiuD. (2018). Quantitation of β-carboline and Quercetin in alligator weed (Alternanthera Philoxeroides (Mart.) Griseb.) by LC-MS/MS and Evaluation of Cardioprotective Effects of the Methanol Extracts. Drug Discoveries Ther.12 (6), 341–346. 10.5582/ddt.2018.01070
485
ZhangZ.WuX.CaoS.CromieM.ShenY.FengY.et al (2017). Chlorogenic Acid Ameliorates Experimental Colitis by Promoting Growth of Akkermansia in Mice. Nutrients9 (7), 1. 10.3390/nu9070677
486
ZhaoB.SuB.ZhangH.LiuW.DuQ.LiY. (2019). Antiurolithiatic Effect of Ferulic Acid on Ethylene Glycolinduced Renal Calculus in Experimental Rats. Trop. J. Pharm. Res.18 (1), 1. 10.4314/tjpr.v18i1.16
487
ZhaoD.ZhengL.QiL.WangS.GuanL.XiaY.et al (2016). Structural Features and Potent Antidepressant Effects of Total Sterols and β-sitosterol Extracted from Sargassum Horneri. Mar. Drugs14 (7), 1. 10.3390/md14070123
488
ZhaoN.DongQ.FuX.-X.DuL.-L.ChengX.DuY.-M.et al (2014). Acacetin Blocks Kv1.3 Channels and Inhibits Human T Cell Activation. Cell Physiol. Biochem.34 (4), 1359–1372. 10.1159/000366343
489
ZhaoT.DingK.-m.ZhangL.ChengX.-m.WangC.-h.WangZ.-t. (2013). Acetylcholinesterase and Butyrylcholinesterase Inhibitory Activities Ofβ-Carboline and Quinoline Alkaloids Derivatives from the Plants of GenusPeganum. J. Chem.2013, 1–6. 10.1155/2013/717232
490
ZhaoY.WangJ.BallevreO.LuoH.ZhangW. (2011). Antihypertensive Effects and Mechanisms of Chlorogenic Acids. Hypertens. Res.35 (4), 370–374. 10.1038/hr.2011.195
491
ZhouB.-N.BlaskòG.CordellG. A. (1988). Alternanthin, A C-Glycosylated Flavonoid from Alternanthera Philoxeroides. Phytochemistry27 (11), 3633–3636. 10.1016/0031-9422(88)80781-7
492
ZhuQ.MaoL.-N.LiuC.-P.SunY.-H.JiangB.ZhangW.et al (2016). Antinociceptive Effects of Vitexin in a Mouse Model of Postoperative Pain. Scientific Rep.6 (1), 1. 10.1038/srep19266
493
ZolotarR. M.BykhovetsA. I.SokolovS. N.KovgankoN. V. (2002). Structure-Activity Relationship of Insecticidal Steroids. IV. 3β-Chlorosubstituted Derivatives of Cholesterol and β-Sitosterol. Chem. Nat. Comp.38 (1), 70–73. 10.1023/a:1015789917352
Summary
Keywords
alternanthera, anticancer, antidiabetic, antimicrobial, flavonoids, triterpenoid saponins, natural products (NP)
Citation
Singla RK, Dhir V, Madaan R, Kumar D, Singh Bola S, Bansal M, Kumar S, Dubey AK, Singla S and Shen B (2022) The Genus Alternanthera: Phytochemical and Ethnopharmacological Perspectives. Front. Pharmacol. 13:769111. doi: 10.3389/fphar.2022.769111
Received
01 September 2021
Accepted
21 February 2022
Published
11 April 2022
Volume
13 - 2022
Edited by
Patrícia Mendonça Rijo, Universidade Lusófona, Portugal
Reviewed by
Mohamed L. Ashour, Ain Shams University, Egypt
Swapnil Sharma, Banasthali Vidyapith, India
Updates
Copyright
© 2022 Singla, Dhir, Madaan, Kumar, Singh Bola, Bansal, Kumar, Dubey, Singla and Shen.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bairong Shen, bairong.shen@scu.edu.cn; Reecha Madaan, reecha.madan@chitkara.edu.in
†These authors have contributed equally to this work and share first authorship
This article was submitted to Ethnopharmacology, a section of the journal Frontiers in Pharmacology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.