Abstract
Background:
Postpartum depression (PPD) poses a major global public health challenge. PPD is the most common complication associated with childbirth and exerts harmful effects on children. Although hundreds of PPD studies have been published, we lack accurate global or national PPD prevalence estimates and have no clear account of why PPD appears to vary so dramatically between nations. Accordingly, we conducted a meta-analysis to estimate the global and national prevalence of PPD and a meta-regression to identify economic, health, social, or policy factors associated with national PPD prevalence.
Methods:
We conducted a systematic review of all papers reporting PPD prevalence using the Edinburgh Postnatal Depression Scale. PPD prevalence and methods were extracted from each study. Random effects meta-analysis was used to estimate global and national PPD prevalence. To test for country level predictors, we drew on data from UNICEF, WHO, and the World Bank. Random effects meta-regression was used to test national predictors of PPD prevalence.
Findings:
291 studies of 296284 women from 56 countries were identified. The global pooled prevalence of PPD was 17.7% (95% confidence interval: 16.6–18.8%), with significant heterogeneity across nations (Q = 16,823, p = 0.000, I2 = 98%), ranging from 3% (2–5%) in Singapore to 38% (35–41%) in Chile. Nations with significantly higher rates of income inequality (R2 = 41%), maternal mortality (R2 = 19%), infant mortality (R2 = 16%), or women of childbearing age working ≥40 h a week (R2 = 31%) have higher rates of PPD. Together, these factors explain 73% of the national variation in PPD prevalence.
Interpretation:
The global prevalence of PPD is greater than previously thought and varies dramatically by nation. Disparities in wealth inequality and maternal-child-health factors explain much of the national variation in PPD prevalence.
Introduction
Maternal mental health problems pose major public health challenges for societies across the globe. For example, psychiatric illness (often associated with suicidality) is one of the leading causes of maternal death in the UK (1), as well as a leading killer of women of childbearing age in both India and China (2). The most common psychiatric malady following childbirth is postpartum depression (PPD), a devastating mental illness that can impair maternal behaviors (3, 4) and adversely affect the cognitive, emotional, and behavioral development of children (5).
Three decades of interdisciplinary research have produced thousands of studies investigating the characteristics, measurement, consequences, treatment, and predictors of PPD. Despite these efforts, the global prevalence of PPD remains unknown. The widely cited PPD prevalence rate of 13% ascertained two-decades ago is based on a meta-analysis of overwhelmingly Western samples (6) and most likely do not reflect the incidence of PPD in the majority of the world’s population. For example, a systematic review and meta-analysis that focused exclusively on low- and lower-middle income countries found a higher incidence of postpartum mental health disorders (7). However, this review, too, did not include wealthy nations for purposes of comparison, leaving open the possibility that the apparently inflated incidence of PPD in the developing world was an artifact of the different study methods employed in those societies (7). For example, low-income countries are more likely than high-income countries to rely on self-report PPD measures (rather than interviews) in the first weeks after birth (7), and we know that self-reported PPD measures taken earlier postpartum tend to yield higher PPD prevalence than interview tools given later. Accordingly, a meta-analysis comparing PPD prevalence, and taking into account divergent research methods used in high-, medium-, and low-income countries, is required to determine the true global and cross-national variation of PPD prevalence.
Further, to our knowledge, no prior large-scale meta-analysis has considered potential cross-national differences in PPD, despite qualitative evidence suggesting that PPD may vary dramatically from nation to nation even between nations of comparable economic standing (8, 9). Reliable national PPD estimates could help to illumine particular economic, health, and policy factors that inflate or reduce PPD prevalence, thereby informing prevention efforts. Further, generating reliable national estimates of PPD could aid policy-makers in decisions about where to allocate limited resources, and alert global health agencies to direct aid to those countries most impacted.
Motivated by the potential health benefits of filling these knowledge gaps, we conducted the largest meta-analysis and meta-regression to date of global PPD prevalence. The present meta-analysis contains four times more studies, 22 times more women, and data from an additional 36 nations compared to the largest previous meta-analysis of PPD prevalence (6). We aimed to estimate PPD prevalence both globally and by nation and to explore whether divergent methodologies or disparities in health, economic, policy, or sociodemographic factors explain cross-national differences in PPD.
Methods
This study was comprised of three phases: (1) conducting a systematic review in accordance with PRISMA guidelines (10), (2) performing a meta-analyses to estimate PPD prevalence both globally and for each nation, and (3) using meta-regression to investigate whether methodological, economic, health, and/or policy factors predict cross-national variation in PPD.
Search Strategy and Selection Criteria
To identify potentially eligible articles, we searched PubMed, PsychINFO, and CINAHL using a combination of the following MeSH terms in the abstract: (“postpartum depression” or “postnatal depression”) and (“incidence” or “prevalence”). In addition, we used the measures and instruments qualifier “edinburgh postnatal depression scale.” We further limited our search by only including studies of human females published in English between 1985 (just before the EPDS scale was published) and 2015. The exact Boolean searches used for each database are provided in Section “Boolean Search Information” in the Appendix. Additionally, we reviewed three previously published comprehensive literature reviews of PPD prevalence (7–9).
To be eligible for inclusion in this meta-analysis, studies were required to report PPD prevalence using the Edinburgh Postpartum Depression Scale (a 10-item self-report, widely used tool specially designed to measure PPD; EPDS) (11) on samples of mothers ≤1 year postpartum with a sample size >20. We chose to include studies conducted anytime in the first year postpartum because this is a convention used in the empirical literature (12) [despite the fact that the American Psychological Association categorizes PPD as occurring anytime in the first 4 weeks postpartum (13), whereas PPD is defined as depression occurring anytime within the first 6 weeks by the World Health Organization (14)]. To address the important issue of timing, we examined whether the timing of assessment influenced PPD prevalence through meta-regression in this paper. We also excluded studies reporting PPD prevalence in samples unlikely to be representative of the general population (e.g., studies that exclusively recruited women with a history of depression, teen mothers, immigrant mothers, abused mothers, mothers seeking treatment, mothers of high-risk infants, etc.).
291 studies (of 487 full-text articles assessed for eligibility) met these criteria and were included in this meta-analysis (see Figure 1 for a PRISMA flow diagram reporting identification and selection of studies for the meta-analysis).
Figure 1
PRISMA flow diagram reporting identification and selection of studies for the meta-analysis.
Studies using the EPDS to estimate PPD prevalence were the focus of this meta-analysis and meta-regression for several reasons. First, a recent systematic review of the validated screening tools for common mental disorder strongly recommended the use of the EPDS because it consistently performs well on metrics of internal and external validity, is easy enough to administer in resource-limited settings, and does not include the word “depression” which is stigmatized in some cultures (15). Second, there are advantages to keeping the type of screening tool used consistent across countries when trying to quantify and illuminate the causes of cross-national variability. For example, the wealth of a country strongly determines the type of PPD screening tool used (16) (e.g., it is harder to use time-intensive clinical interviews in resource-poor settings yet easier in resource-rich settings), and the type of screening tool used can influence PPD prevalence (6, 17). Had we included multiple screening tools that differed on ease of administration (e.g., self-report vs. clinical interviews), it would have been difficult to determine whether any observed cross-national variance in PPD prevalence was due to disparities in national wealth or merely an artifact of the assessment tool used. Third, the EPDS had been widely translated and validated for use in at least 18 languages and exhibits good cross-cultural reliability (18). In addition, an examination of previously published systematic reviews showed that roughly 70% of studies used the EPDS to assess PPD prevalence (6, 8, 9). Therefore, the use of the EPDS allowed us to include the majority of studies while limiting confounding variables associated with different types of measurement (8). Finally, because the EPDS is specifically designed for administration in the postpartum period, the scale does not include items assessing changes in appetite, sleep, or weight. Changes in these factors are normal in the postpartum period, yet these somatic items are included as indicators of depression by other self-report screening tools designed to assess depression outside of the postpartum window (e.g., Patient Health Questionnaire-PHQ-9, The Hamilton Rating Scale for Depression-HAM-D, Center for Epidemiologic Studies Depression Scale-CESD, Beck Depression Inventory-BDI, and Zung’s Self-Rating Depression Scale-SDS).
Data Extraction
The following methodological variables were coded from each study: PPD prevalence, total sample size, EPDS cutoff score employed, and the timeframe postpartum in which PPD was assessed. Because meta-analysis requires one estimate of PPD prevalence per study, data from longitudinal studies reporting PPD in the same women at multiple time points were consolidated by averaging the PPD prevalence over the time points weighted by the sample size at each time point. Also, if multiple prevalence rates were reported in the same study using different EPDS cutoffs, the prevalence rate from the lowest EPDS cutoff was chosen by default. This decision could cause a bias toward higher estimates of PPD incidence; therefore, we also used meta-regression to estimate PPD prevalence at the standard recommended EPDS cutoffs for possible (9/10) and probable (12/13) PPD (11).
To investigate whether studies including women earlier or later in the postpartum period report higher PPD prevalence, we created scores for each study reflecting the range of the timeframes postpartum during which PPD was assessed.
National Data
Various methodological, health, economic, policy, and sociodemographic variables were explored as potential predictors of cross-national variation in PPD. Potential cross-national predictors of PPD were chosen because they had been previously hypothesized to predict PPD and reliable national data were available for the majority of counties represented in this meta-analysis. See Data Sheet S1 in Supplementary Material for an Excel file containing all of the national data used.
Methodological Variables
A previous meta-analysis of PPD suggested that it is important to rule out the possibility that cross-national variation in PPD prevalence is explained by methodological conventions used in different countries (7). For example, it is important to know whether systematic methodological differences like assessing PPD earlier postpartum or using higher/lower EPDS cutoff scores are employed in some countries more often than others. Further, if methodological conventions do differ across countries, we need to know the extent to which these explain the apparent cross-national variation in PPD prevalence. To explore this possibility, country sample-size-weighted national averages for each methodological variable were calculated for use in meta-regression models. In addition, we used meta-regression to assess whether the number of studies conducted in a country predicted cross-national PPD prevalence.
Health Variables
Health variables were obtained from UNICEF (19) unless otherwise noted and included infant mortality rate (the probability of dying between birth and age one, expressed per 1,000 live births), lifetime risk of maternal death (the annual number of deaths of women from pregnancy-related causes per 100,000 live births), total fertility rate (the number of children that would be born per woman if she were to live to the end of her childbearing years and bear children in accordance with prevailing age-specific fertility rates), and percentage of low-birthweight infants (born weighing <2,500 g). Percentage of cesarean births was obtained from the World Health Report (20).
Economic and Policy Variables
GINI index (an index of the income distribution of a nation’s residents wherein higher values indicate greater wealth inequality) data were obtained from Ortiz and Cummins (21). Gross Domestic Product (GDP) per capita (in adjusted US dollars) and percentage of women working ≥40 h a week (aged 25–30) data were obtained from the Annual labor force statistics (22). Additionally, we investigated national provisions for paid and unpaid maternity leave available from the international labor office (23).
Sociodemographic Predictors
The percentage of children living in single parent homes and the percentage of infants born outside of marriage data were obtained from the World Family Map (24). The percentage of urbanized population data were also obtained from UNICEF.
Data Analysis
Following the recommendations for meta-analysis of prevalence (25), we used a double-arcsine transformation of the PPD prevalence data before calculating the study weights and 95% confidence intervals (CIs) to avoid the undue large weights obtained for studies with low or high prevalence (prevalence close to 0 or 1). To test for heterogeneity in the data, both the Cochran Q test statistic and the I2 statistic were consulted (26). The same procedure was followed to create meta-analytically derived national estimates of PPD prevalence based solely on the studies available from each country. Meta-analytic estimates of PPD prevalence could not be calculated in countries with fewer than two studies (N = 16) (27). All meta-analyses were conducted using the program MetaXL and the “prev” command (25).
Two sets of meta-regressions were performed, the first addressing which methodological factors predicted variation in PPD across all studies, regardless of the nation in which the study was conducted, and the second addressing predictors of PPD variation across nations. All meta-regression analyses were performed with STATA 14 (28) using the “metareg” command with random-effects models (because all tests indicated significant heterogeneity). To obtain the SEs needed to weight studies (or nations) for meta-regression in STATA, we transformed the 95%-CIs provided by MetaXL using the following formula (upper 95% CI − lower 95% CI)/3.92. Because national data were not available for all variables, the number of countries included is reported for each meta-regression result using national variables.
Funnel plots, Doi plot analysis, and the LFK index were used to assess potential publication bias. Specifically, to test whether papers are more or less likely to be published due to higher/lower PPD prevalence.
Statistical significance was evaluated using 2-tailed 0.05-level tests.
Results
Meta-Analysis of Global PPD Prevalence
296,284 women from 291 studies were included in this meta-analysis. Table 1 presents the data extracted from each study. The global pooled prevalence of PPD was 17.7% (95% CI: 16.6 to 18.8%; see Figure S1 in Supplementary Material). There was a significant degree of heterogeneity between studies (Q = 16,823, p = 0.000, I2 = 98%). Adjusting for the recommended EPDS cutoffs yielded a global PPD prevalence of 21.0% (CI: 19.1 to 23.0%) for possible PPD and 16.7% (CI: 14.9 to 18.6%) for probable PPD. See Figure S1 in Supplementary Material for meta-analytically derived PPD estimates for each individual study. There was evidence of publication bias based on sample size (LFK = 1.98; see Funnel Plot in Figure 2).
Table 1
| Reference | n | Depression prevalence (%) | Cut-off used | Postpartum assessment (weeks) | Country |
|---|---|---|---|---|---|
| Affonso et al. (29) | 102 | 15.8 | 10 | 1–6 | Australia |
| Alcorn et al. (30) | 866 | 14.4 | 12 | 4–24 | |
| Armstrong et al. (31) | 114 | 26.4 | 12 | ||
| Astbury et al. (32) | 790 | 15.4 | 13 | 32–36 | |
| Bilszta et al. (33) | 1,966 | 7.6 | 13 | 6–8 | |
| Boyce and Hickey (34) | 425 | 9.1 | 12 | 6–24 | |
| Boyce et al (35) | 103 | 12.7 | 13 | 4–6 | |
| Brooks et al. (36) | 3,853 | 6.0 | 13 | 1–52 | |
| Brown and Lumley (37) | 1,331 | 19.6 | 13 | 4–6 | |
| Buist et al. (38) | 12,361 | 15.5 | 10 | 6–8 | |
| Condon and Corkindale (39) | 212 | 6.1 | 13 | 4–6 | |
| Eastwood et al. (40) | 25,455 | 12.0 | 10 | 1–12 | |
| Eastwood et al. (41) | 15,389 | 16.9 | 10 | 2–3 | |
| Edwards et al. (42) | 421 | 29.7 | 10 | ||
| Griepsma et al. (43) | 185 | 57.8 | 13 | 12 | |
| Leigh and Milgrom (44) | 161 | 11.2 | 13 | 10–12 | |
| Maloney (45) | 399 | 18.0 | 13 | 4–6 | |
| Miller et al. (46) | 325 | 25.0 | 9 | 6–24 | |
| Stamp and Crowther (47) | 222 | 9.4 | 13 | 6–24 | |
| Stamp et al. (48) | 108 | 17.0 | 13 | 4–6 | |
| White et al. (49) | 316 | 20.3 | 10 | 6–52 | |
| Willinck and Cotton (50) | 358 | 7.0 | 13 | 6–8 | |
| Wynter et al. (51) | 172 | 12.2 | 9 | 24 | |
| Kohl et al. (52) | 95 | 9.5 | 12 | 1 | Austria |
| Al Dallal and Grant (53) | 237 | 37.1 | 12 | 8 | Bahrain |
| Edhborg et al. (54) | 674 | 14.0 | 10 | 8–12 | Bangladesh |
| Gausia et al. (55) | 346 | 22.0 | 10 | 6–8 | |
| Da-Silva et al. (56) | 21 | 42.8 | 13 | 4 | Brazil |
| de Almeida et al. (57) | 222 | 16.2 | 13 | ||
| Filha et al. (58) | 12,764 | 25.8 | 13 | 24–36 | |
| Lobato et al. (59) | 811 | 24.3 | 12 | 0–20 | |
| Lobato et al. (60) | 456 | 24.8 | 12 | 6–8 | |
| Matijasevich et al. (61) | 4,109 | 13.3 | 13 | 12–52 | |
| Melo et al. (62) | 555 | 10.8 | 12 | 4–6 | |
| Morais et al. (63) | 87 | 19.1 | 12 | 16–52 | |
| Pinheiro et al. (64) | 207 | 20.3 | 13 | 6–12 | |
| Silva et al. (65) | 1,109 | 16.5 | 13 | 4–8 | |
| Bernazzani et al. (66) | 213 | 12.7 | 13 | 24 | Canada |
| Bowen et al. (67) | 649 | 8.1 | 12 | 4 | |
| DaCosta et al. (68) | 78 | 63.0 | 12 | 4–38 | |
| Dennis and Letourneau (69) | 498 | 8.0 | 13 | 8 | |
| Dennis and Ross (70) | 425 | 14.1 | 10 | 8 | |
| Dennis and Vigod (71) | 497 | 20.7 | 10 | 8 | |
| Dennis et al. (72) | 498 | 24.8 | 10 | 1–8 | |
| Dennis et al. (73) | 315 | 7.0 | 13 | 12 | |
| Malta et al. (74) | 972 | 10.0 | 10 | 16 | |
| McDonald et al. (75) | 1,578 | 13.0 | 10 | 16 | |
| Sword et al. (76) | 2,560 | 7.6 | 12 | 6 | |
| Verreault et al. (77) | 226 | 16.4 | 10 | 12 | |
| Vigod et al. (78) | 6,126 | 7.5 | 13 | ||
| Florenzano et al. (79) | 88 | 50.0 | 0–2 | Chile | |
| Jadresic et al. (80) | 108 | 28.7 | 10 | 8–12 | |
| Jadresic et al. (81) | 542 | 36.7 | 8–12 | ||
| Risco et al. (82) | 103 | 37.6 | 1–12 | ||
| Gao et al. (83) | 130 | 13.8 | 13 | 6–8 | China |
| Gao et al. (84) | 126 | 14.3 | 13 | 6–8 | |
| Leung et al. (85) | 694 | 7.2 | 10 | 6 | |
| Xie et al. (86) | 300 | 17.3 | 13 | 6 | |
| Xie et al. (87) | 534 | 19.3 | 13 | 2 | |
| Nielsen Forman et al. (88) | 5,091 | 5.5 | 13 | 6 | Denmark |
| Affonso et al. (29) | 58 | 21.8 | 10 | 1–6 | Finland |
| Hiltunen et al. (89) | 185 | 14.7 | 13 | 1–36 | |
| Luoma et al. (90) | 147 | 10.0 | 13 | 8 | |
| de Tychey et al. (91) | 277 | 11.1 | 12 | 4–8 | France |
| Frossey et al. (93) | 126 | 11.0 | 12 | 1 | |
| Gaillard et al. (94) | 264 | 16.7 | 12 | 6–8 | |
| Glangeaud-Freudenthal and Kaminski (95) | 604 | 11.0 | 13 | 8 | |
| Guendeney and Fermanian (96) | 87 | 73.5 | 11 | 16 | |
| Sutter-Dallay et al. (97) | 497 | 5.8 | 12 | 6 | |
| Ballestrem et al. (98) | 772 | 17.0 | 10 | 6–8 | Germany |
| Bergant et al. (99) | 110 | 19.0 | 10 | 1 | |
| Mehta et al. (100) | 419 | 11.2 | 9 | 1–32 | |
| Reck et al. (101) | 891 | 23.6 | 10 | 2–6 | |
| Zaers et al. (102) | 50 | 21.7 | 10 | 6–24 | |
| Chatzi et al. (103) | 529 | 14.0 | 13 | 8–10 | Greece |
| Gonidakis et al. (104) | 402 | 19.8 | 12 | 1–24 | |
| Koutra et al. (105) | 438 | 13.0 | 13 | 8 | |
| Lambrinoudaki et al. (106) | 57 | 23.5 | 11 | 1–6 | |
| Thorpe et al. (107) | 165 | 13.0 | 12 | 4 | |
| Affonso et al. (29) | 106 | 53.3 | 10 | 1–6 | Guyana |
| Lau and Chan (108) | 1,200 | 34.4 | 9 | 1 | Hong Kong |
| Lee et al. (109) | 145 | 11.3 | 13 | 6 | |
| Lee et al. (110) | 244 | 24.2 | 10 | 6 | |
| Leung et al. (111) | 269 | 19.8 | 13 | 6 | |
| Tiwari et al. (112) | 3,036 | 69.9 | 10 | 1 | |
| Nagy et al. (113) | 988 | 10.8 | 13 | 3–26 | Hungary |
| Thome (114) | 734 | 14.0 | 13 | 8–12 | Iceland |
| Affonso et al. (29) | 110 | 33.4 | 10 | 1–6 | India |
| Dubey et al. (92) | 293 | 6.1 | 10 | 1 | |
| Ghosh and Goswami (115) | 6,000 | 25.08 | 13 | 1 | |
| Jain et al. (116) | 1,537 | 7.31 | 12 | 1 | |
| Mariam and Srinivasan (117) | 132 | 30.0 | 12 | 6–10 | |
| Patel et al. (118) | 134 | 48.5 | 11 | 1 | |
| Patel et al. (119) | 270 | 23.0 | 12 | 6–24 | |
| Andajani-Sutjahjo et al. (120) | 274 | 7.4 | 12 | 6–24 | Indonesia |
| Abbasi et al. (121) | 416 | 34.1 | 13 | 12 | Iran |
| Abdollahi et al. (122) | 2,083 | 19.4 | 12 | 8 | |
| Goshtasebi et al. (123) | 281 | 5.5 | 13 | 4–6 | |
| Kheirabadi and Maracy (124) | 1,291 | 26.3 | 14 | 6–8 | |
| Montazeri et al. (125) | 100 | 20.0 | 13 | 6–14 | |
| Ahmed et al. (126) | 1,000 | 28.4 | 10 | 6–8 | Iraq |
| Crotty and Sheehan (127) | 625 | 27.0 | 12 | 6 | Ireland |
| Cryan et al. (128) | 377 | 28.6 | 13 | 1–52 | |
| Lane et al. (129) | 242 | 11.0 | 13 | 6 | |
| Leahy-Warren et al. (130) | 410 | 12.3 | 11 | 6 | |
| Alfayumi-Zeadna et al. (131) | 564 | 31.0 | 10 | 4–28 | Israel |
| Bloch et al. (132) | 210 | 33.0 | 10 | 1 | |
| Bloch et al. (133) | 1,286 | 6.8 | 10 | 1 | |
| Dankner et al. (134) | 327 | 11.0 | 9 | 6–10 | |
| Eilat-Tsanani et al. (135) | 574 | 9.9 | 13 | 8 | |
| Fisch et al. (136) | 327 | 5.2 | 13 | 6–12 | |
| Glasser et al. (137) | 288 | 22.6 | 10 | 6 | |
| Glasser et al. (138) | 104 | 43.0 | 10 | 1–36 | |
| Affonso et al. (29) | 100 | 55.5 | 10 | 1–6 | Italy |
| Benvenuti et al. (139) | 113 | 38.9 | 13 | 8–12 | |
| Carpiniello et al. (140) | 61 | 29.5 | 10 | 4–6 | |
| Elisei et al. (141) | 54 | 13.9 | 13.00 | 1–12 | |
| Giardinelli et al. (142) | 590 | 13.2 | 10 | 12 | |
| Gorman et al. (143) | 21 | 9.5 | 13 | 24 | |
| Grussu and Quantraro (144) | 297 | 13.0 | 9 | 6–8 | |
| Mauri et al. (145) | 751 | 10.4 | 13 | 4–52 | |
| Oppo et al. (146) | 600 | 6.7 | 13 | 4–24 | |
| Matsumoto (147) | 675 | 14.8 | 9 | 12 | Japan |
| Miyake et al. (148) | 865 | 14.0 | 9 | 8–36 | |
| Nishigori et al. (149) | 677 | 21.3 | 9 | 24–36 | |
| Nishizono-Maher et al. (150) | 1,048 | 13.9 | 9 | 12–16 | |
| Ohoka et al. (151) | 388 | 10.3 | 9 | 4 | |
| Shimizu et al. (152) | 65 | 12.3 | 9 | 4–16 | |
| Tamaki et al. (153) | 627 | 18.2 | 13 | 4 | |
| Ueda et al. (154) | 70 | 27.0 | 9 | 1–52 | |
| Watanabe et al. (155) | 235 | 12.8 | 9 | 1–12 | |
| Yamashita et al. (156) | 75 | 16.0 | 9 | 4 | |
| Affonso et al. (29) | 97 | 36.7 | 10 | 1–6 | Korea |
| Bang (157) | 137 | 22.6 | 4 | ||
| Kim et al. (158) | 239 | 12.6 | 10 | 6 | |
| Chaaya et al. (159) | 396 | 21.0 | 13 | 12–20 | Lebanon |
| El-Hachem et al. (160) | 228 | 33.3 | 9 | 1 | |
| Dow et al. (161) | 154 | 8.1 | 13 | 10–14 | Malawi |
| Azidah et al. (162) | 377 | 22.8 | 12 | 1 | Malaysia |
| Kadir et al. (163) | 293 | 24.9 | 12 | 1–6 | |
| Kit et al. (164) | 154 | 3.9 | 14 | 6 | |
| Yusuf et al. (165) | 1,362 | 14.3 | 12 | 1–24 | |
| Felice et al. (166) | 229 | 8.7 | 8 | Malta | |
| deCastro et al. (167) | 298 | 14.8 | 13 | 1–36 | Mexico |
| Flores-Quijano et al. (168) | 163 | 24.5 | 13 | 2–12 | |
| Agoub et al. (169) | 144 | 20.1 | 12 | 2–3 | Morocco |
| Alami et al. (170) | 100 | 21.0 | 12 | 0–36 | |
| Dørheim Ho-Yen et al. (171) | 426 | 4.9 | 13 | 5–10 | Nepal |
| Regmi et al. (172) | 100 | 12.0 | 13 | 8–12 | |
| Blom et al. (173) | 4,941 | 8.0 | 12 | 8 | Netherlands |
| Verkerk et al. (174) | 277 | 8.2 | 12 | 12–52 | |
| Abbott and Williams (175) | 1,376 | 16.4 | 13 | 6 | New Zealand |
| Holt (176) | 121 | 14.0 | 13 | 6 | |
| McGill et al. (177) | 1,330 | 20.0 | 12 | 24–36 | |
| Webster et al. (178) | 206 | 7.8 | 13 | 4 | |
| Abiodun (179) | 360 | 18.6 | 9 | 6 | Nigeria |
| Adewuya et al. (180) | 478 | 20.9 | 13 | 0–8 | |
| Adewuya et al. (181) | 876 | 14.6 | 10 | 6 | |
| Bakare et al. (182) | 408 | 24.8 | 9 | 1–52 | |
| Dørheim et al. (183) | 2,791 | 16.5 | 10 | 7 | Norway |
| Dørheim et al. (184) | 2,088 | 23.9 | 10 | 8 | |
| Eberhand-Gran et al. (185) | 56 | 26.8 | 10 | 6 | |
| Eberhand-Gran et al. (186) | 2,370 | 8.9 | 10 | 6 | |
| Eberhard-Gran et al. (187) | 473 | 9.1 | 10 | 1–52 | |
| Glavin et al. (188) | 2,227 | 10.1 | 10 | 6 | |
| Haga et al. (189) | 737 | 13.6 | 10 | 6–26 | |
| Markhus et al. (190) | 43 | 6.9 | 10 | 13 | |
| Nordeng et al. (191) | 1,984 | 8.1 | 13 | 17 | |
| Ahmad and Khan (192) | 876 | 14.6 | 9 | 6 | Pakistan |
| Husain et al. (193) | 149 | 36.0 | 12 | 12 | |
| Ayoub (194) | 235 | 17.0 | 10 | 2–12 | Palestine |
| Duedek et al. (195) | 344 | 16.0 | 13 | 6–12 | Poland |
| Augusto et al. (196) | 588 | 12.5 | 13 | 8–20 | Portugal |
| Figueiredo and Conde (197) | 260 | 14.4 | 10 | 0–12 | |
| Figueiredo and Costa (198) | 91 | 26.7 | 10 | 13 | |
| Figueiredo et al. (199) | 108 | 17.6 | 13 | 8–12 | |
| Gorman et al. (143) | 48 | 9.5 | 13 | 24 | |
| Chee et al. (200) | 278 | 6.8 | 7 | 6 | Singapore |
| Kok et al. (201) | 200 | 0.5 | 16 | 12 | |
| Lawrie et al. (202) | 180 | 36.2 | 12 | 6 | South Africa |
| Lawrie et al. (203) | 103 | 36.9 | 13 | 6 | |
| Escriba-Aguir and Artazcoz (204) | 420 | 9.8 | 11 | 12–52 | Spain |
| Garcia-Esteve et al. (205) | 1,201 | 21.7 | 9 | 6 | |
| Sebastián Romero et al. (206) | 190 | 13.2 | 12 | 6–8 | |
| Affonso et al. (29) | 108 | 13.9 | 13 | 1–6 | Sweden |
| Agnafors et al. (207) | 1,707 | 12.0 | 10 | 12 | |
| Bågedahl-Strindlund and Börjesson (208) | 309 | 14.5 | 13 | 12 | |
| Josefsson et al. (209) | 1,192 | 13.0 | 10 | 6–8 | |
| Lundh and Gyllang (210) | 258 | 8.0 | 10 | 6 | |
| Rubertsson et al. (211) | 2,430 | 12.4 | 13 | 8–52 | |
| Seimyr et al. (212) | 326 | 14.6 | 10 | 8–52 | |
| Sylven et al. (213) | 2,318 | 10.6 | 12 | 1–24 | |
| Wickberg and Hwang (214) | 1,655 | 12.0 | 12 | 8 | |
| Burgut et al. (215) | 1,379 | 17.6 | 12 | 1–24 | Qatar |
| Alharbi and Abdulghani (216) | 352 | 33.2 | 10 | 8–12 | Saudi Arabia |
| Al-Modayfer et al. (217) | 571 | 13.7 | 13 | 5 | |
| Gorman et al. (143) | 60 | 6.7 | 13 | 24 | Switzerland |
| Gürber et al. (218) | 219 | 13.4 | 10 | 1–3 | |
| Righetti-Veltema et al. (219) | 570 | 10.2 | 13 | 12 | |
| Affonso et al. (29) | 99 | 67.3 | 10 | 1–6 | Taiwan |
| Chen et al. (220) | 226 | 18.2 | 10 | 4–24 | |
| Chien et al. (221) | 190 | 8.4 | 10 | 1–52 | |
| Heh et al. (222) | 186 | 21.0 | 10 | 4 | |
| Heh et al. (223) | 400 | 23.0 | 10 | 4 | |
| Huang and Mathers (224) | 101 | 19.0 | 13 | 24 | |
| Huang and Mathers (225) | 106 | 25.5 | 13 | 24 | |
| Lee et al. (226) | 60 | 25.0 | 14 | 5–8 | |
| Teng et al. (227) | 203 | 10.3 | 13 | 6 | |
| Tsao et al. (228) | 162 | 24.1 | 13 | 6 | |
| Limlomwongse and Liabsuetrakul (229) | 525 | 16.8 | 10 | 6–8 | Thailand |
| Akman et al. (230) | 60 | 13.6 | 13 | 4 | Turkey |
| Alkar and Gençöz (231) | 151 | 74.0 | 10 | 1 | |
| Aydin et al. (232) | 728 | 34.6 | 13 | 0–52 | |
| Aydin et al.(233) | 341 | 35.8 | 12.5 | 0–52 | |
| Ayvaz et al. (234) | 152 | 21.1 | 13 | 6–24 | |
| Bugdayci et al. (235) | 1,447 | 37.4 | 13 | 0–52 | |
| Danaci et al. (236) | 257 | 14.0 | 13 | 4–24 | |
| Dindar and Erdogan (237) | 679 | 32.7 | 12 | 8–52 | |
| Ege et al. (238) | 364 | 33.2 | 13 | 6–48 | |
| Ekuklu et al. (239) | 178 | 40.4 | 12 | 6 | |
| Goker et al. (240) | 318 | 31.4 | 13 | 6 | |
| Gulseren et al. (241) | 125 | 13.6 | 5–26 | ||
| Inandi et al. (242) | 2,514 | 27.2 | 13 | 1–52 | |
| Inandi et al. (243) | 1,350 | 31.1 | 13 | 1–52 | |
| Kirpinar et al. (244) | 479 | 15.9 | 13 | 1–6 | |
| Orhon et al. (245) | 103 | 27.2 | 12 | 4 | |
| Poçan et al. (246) | 187 | 28.9 | 13 | 4–6 | |
| Tezel and Gözüm (247) | 567 | 12.9 | 11 | 1 | |
| Yagmur and Ulukoca (248) | 785 | 21.0 | 13 | 1–52 | |
| Ghubash and Abou-Saleh (249) | 94 | 18.0 | 12 | 1 | United Arab Emirates |
| Green et al. (250) | 86 | 39.4 | 10 | 12–24 | |
| Hamdan and Tamim (251) | 137 | 16.8 | 10 | 8 | |
| Brugha et al. (252) | 190 | 17.4 | 11 | 12 | United Kingdom |
| Cooper et al. (253) | 5,124 | 31.8 | 9 | 6–8 | |
| Edge et al. (254) | 301 | 32.0 | 12 | 6 | |
| Evans et al. (255) | 9,028 | 9.1 | 13 | 8 | |
| Hearn et al. (256) | 176 | 17.0 | 12 | 7 | |
| Heron et al. (257) | 207 | 14.0 | 13 | 1–8 | |
| Honey et al. (258) | 223 | 17.0 | 13 | 6 | |
| Huang and Mathers (224) | 50 | 18.0 | 13 | 12 | |
| Matijasevich et al. (61) | 13,798 | 9.6 | 13 | 8–24 | |
| Morrell et al. (259) | 3,449 | 17.3 | 12 | 6 | |
| O’Higgins et al. (260) | 2,048 | 13.9 | 13 | 4 | |
| Ramchandani et al. (261) | 11,833 | 10.0 | 12 | 8 | |
| Shelton and Herrick (262) | 394 | 24.4 | 10 | 1–52 | |
| Thompson et al. (263) | 149 | 18.8 | 13 | 12 | |
| Thorpe et al. (107) | 101 | 12.0 | 12 | 4 | |
| Warner et al. (264) | 2,375 | 11.8 | 13 | 6–8 | |
| Abbasi et al. (265) | 2,972 | 5.1 | 12 | 4 | United States of America |
| Affonso et al. (29) | 119 | 34.1 | 10 | 1–6 | |
| Beck and Gable (266) | 150 | 14.6 | 12 | 2–12 | |
| Birkeland et al. (267) | 149 | 29.0 | 13 | 8–52 | |
| Certain et al. (268) | 1,519 | 10.1 | 13 | ||
| Chaudron et al. (269) | 60 | 27.0 | 10 | 0–52 | |
| Dagher and Shenassa (270) | 526 | 6.5 | 8 | ||
| Dagher et al. (271) | 638 | 4.7 | 13 | 11 | |
| Demissie et al. (272) | 652 | 7.0 | 13 | 12 | |
| Doering Runquist et al. (273) | 43 | 24.3 | 13 | 4–24 | |
| Eisenach et al. (274) | 939 | 11.2 | 13 | 8 | |
| Gaffney et al. (275) | 1,447 | 24.1 | 10 | 8 | |
| Georgiopoulos et al. (276) | 909 | 11.4 | 12 | 6 | |
| Glynn and Sandman (277) | 170 | 20.0 | 10 | 12 | |
| Gorman et al. (143) | 41 | 5.0 | 13 | 24 | |
| Hahn-Holbrook et al. (278) | 200 | 20.0 | 10 | 13 | |
| Herring et al. (279) | 850 | 4.0 | 13 | 24 | |
| Horowitz (280) | 1,071 | 19.7 | 10 | 2–4 | |
| Horowitz et al. (281) | 5,169 | 13.0 | 10 | 4 | |
| Howell et al. (282) | 242 | 14.1 | 13 | 12–24 | |
| Howell et al. (283) | 251 | 5.5 | 10 | 3–24 | |
| Hunker et al. (284) | 123 | 21.0 | 9 | 2 | |
| Kim et al. (285) | 324 | 17.0 | 10 | 1 | |
| Kuo et al. (286) | 139 | 25.4 | 13 | 1–24 | |
| McGrath et al. (287) | 114 | 13.1 | 12 | 9–24 | |
| Mercier et al. (288) | 688 | 6.7 | 13 | 12–52 | |
| Miller et al. (46) | 280 | 8.0 | 13 | 0–16 | |
| Morris-Rush et al. (289) | 121 | 22.0 | 10 | 6 | |
| Mosack and Shore (290) | 98 | 14.3 | 12 | 24 | |
| Mott et al. (291) | 147 | 7.5 | 13 | 52 | |
| Murphy et al. (292) | 97 | 12.0 | 9 | 4–6 | |
| Park et al. (293) | 25 | 12.0 | 13 | 2–14 | |
| Paul et al. (294) | 1,123 | 4.2 | 12 | 1–24 | |
| Reighard and Evans (295) | 181 | 19.9 | 12 | ||
| Rich-Edwards et al. (296) | 1,278 | 8.0 | 13 | 24 | |
| Roy et al. (297) | 185 | 17.4 | 12 | 6 | |
| Schaper et al. (298) | 287 | 7.0 | 13 | 24 | |
| Silverman and Loudon (299) | 439 | 21.4 | 9 | 6 | |
| Watkins et al. (300) | 2,586 | 8.6 | 13 | 8 | |
| Wisner et al. (301) | 10,000 | 14.0 | 10 | 4–6 | |
| Yim et al. (302) | 100 | 22.0 | 10 | 8 | |
| Yonkers et al. (303) | 802 | 16.0 | 12 | 4 | |
| Chibanda et al. (304) | 210 | 35.5 | 12 | 6–7 | Zimbabwe |
| Fisher et al. (305) | 506 | 33.0 | 12 | 6 | Vietnam |
Studies included in meta-analysis.
Figure 2
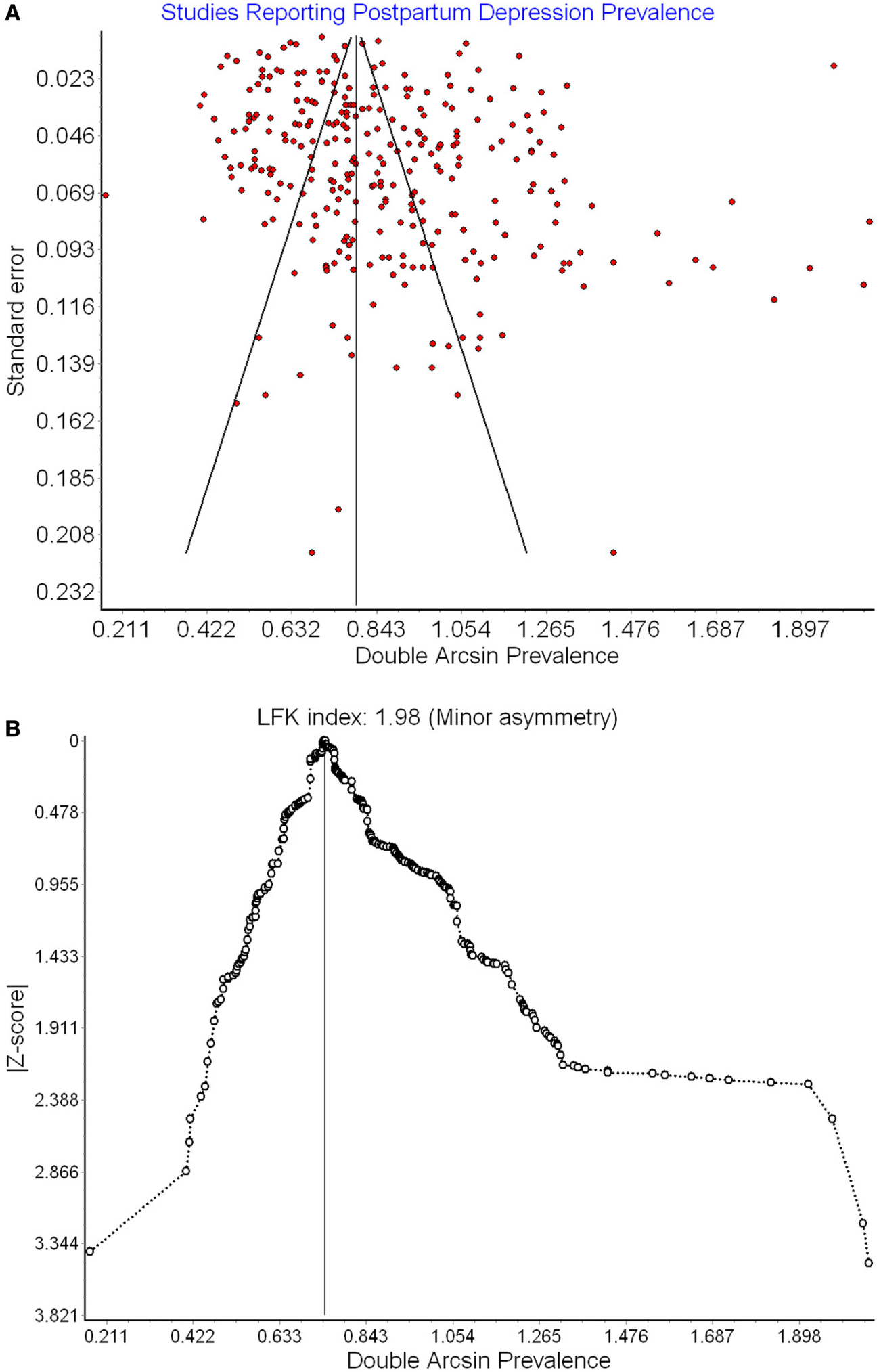
Funnel plot (A) and Doi plot (B) of postpartum depression (PPD) prevalence as a function of prevalence estimate SE.
Meta-Regression of Between-Study Variation
Studies that used lower cutoffs of the EPDS reported significantly higher prevalence (Coef. = −1.44, SE = 0.455, p = 0.002; CI: −2.333 to −0.542, R2 = 3.08%). Studies that measured PPD later postpartum tended to report slightly lower levels of PPD (Coef. = −0.373, SE = 0.109, p = 0.001, 95% CI: −0.587 to −0.159, R2 = 3.65%). No other methodological variables predicted between-study variation in PPD. Together timing of PPD assessment and cutoff used accounted for 5.21% of the variance in PPD prevalence between studies [F(2, 293) = 6.44, p < 0.002].
Meta-Analyses of National PPD Prevalence
See Figure 3 for meta-analytically derived estimates of PPD prevalence in 40 countries. National sample sizes ranged from 244 to 65,634 women (M = 7,229.76; SD = 13,502.69). National estimates of PPD ranged from 3.1% in Singapore to 37.7% in Chile. Meta-analysis suggested that there was significant heterogeneity in PPD prevalence between nations (Q = 3,489.09, p < 0.001, I2 = 99%).
Figure 3
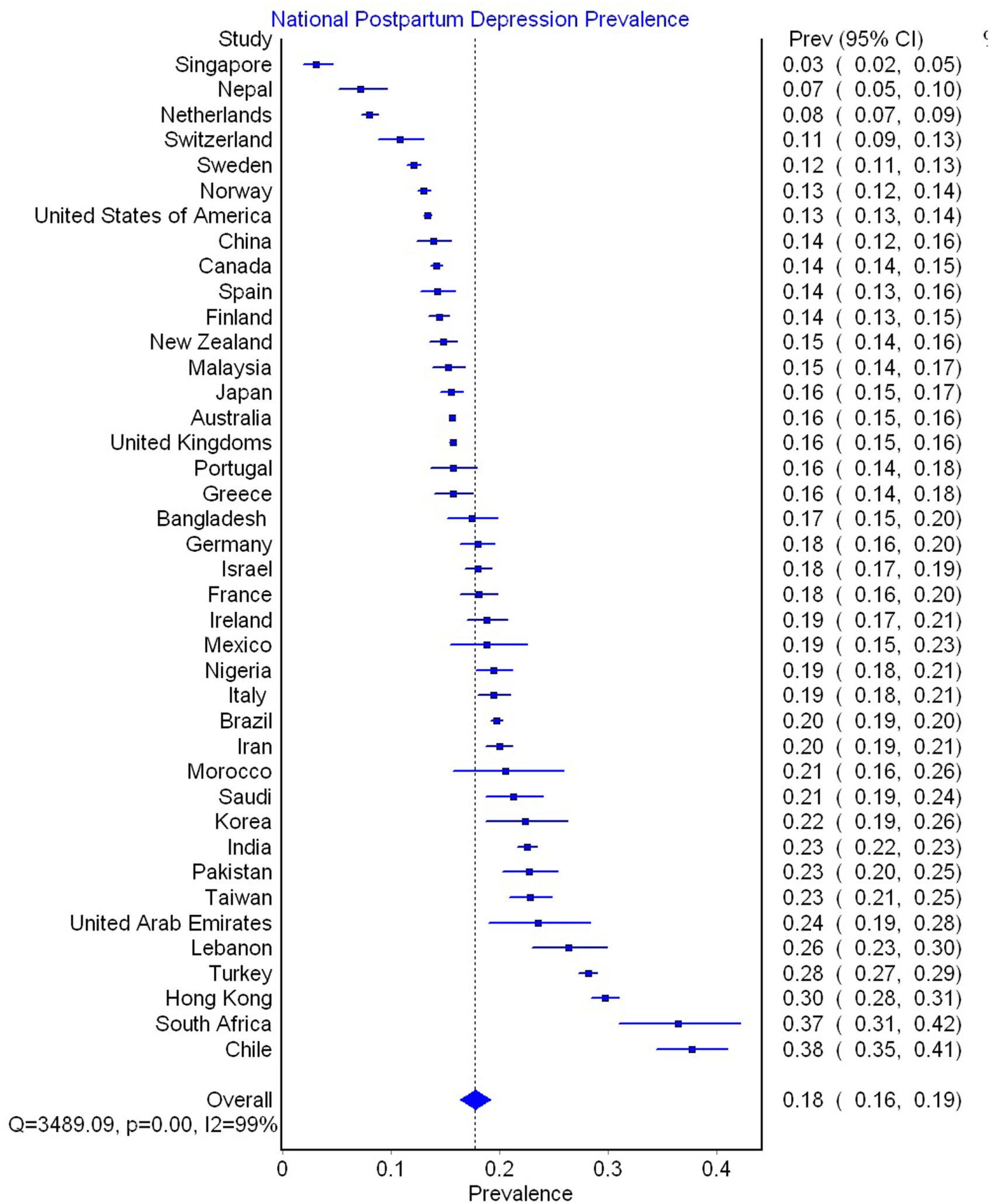
Meta-analytically derived postpartum depression prevalence in 40 Countries.
Meta-Regression of Predictors of Cross-National Variation
Methodological Predictors
None of the methodological variables predicted cross-national variation in PPD prevalence (all ps > 0.15). Therefore, no methodological variables were included as covariates in subsequent models.
Economic and Policy Predictors
GINI index explained 41% of the cross-national variation in PPD prevalence. Nations with higher wealth inequality had higher levels of PPD (N = 38; Coef. = 0.039, SE = 0.009, p < 0.000, CI: 0.020 to 0.058) (see Figure 4A). GDP per capita was also inversely related to PPD prevalence (N = 39; Coef. = −0.033, SE = 0.009, p = 0.002, CI: −0.053 to −0.014, R2 = 30.4%). However, when GDP per capital and GINI index were modeled together, GINI index remained statistically significant while GDP per capita did not. In addition, countries with higher percentages of young women who were working ≥40 h a week had higher PPD prevalence (N = 24; Coef. = 0.038, SE = 0.013, p < .01, CI: 0.012 to 0.065, R2 = 30.9%; see Figure 4B). National paid and unpaid maternity leave policies did not predict PPD prevalence (ps > 0.60). Together, economic predictors (GINI index, GDP per capita, and women working >40 h per week) accounted for 73.1% of the cross-national variation in PPD prevalence, although GINI index was the only unique economic predictor in a multivariate model.
Figure 4
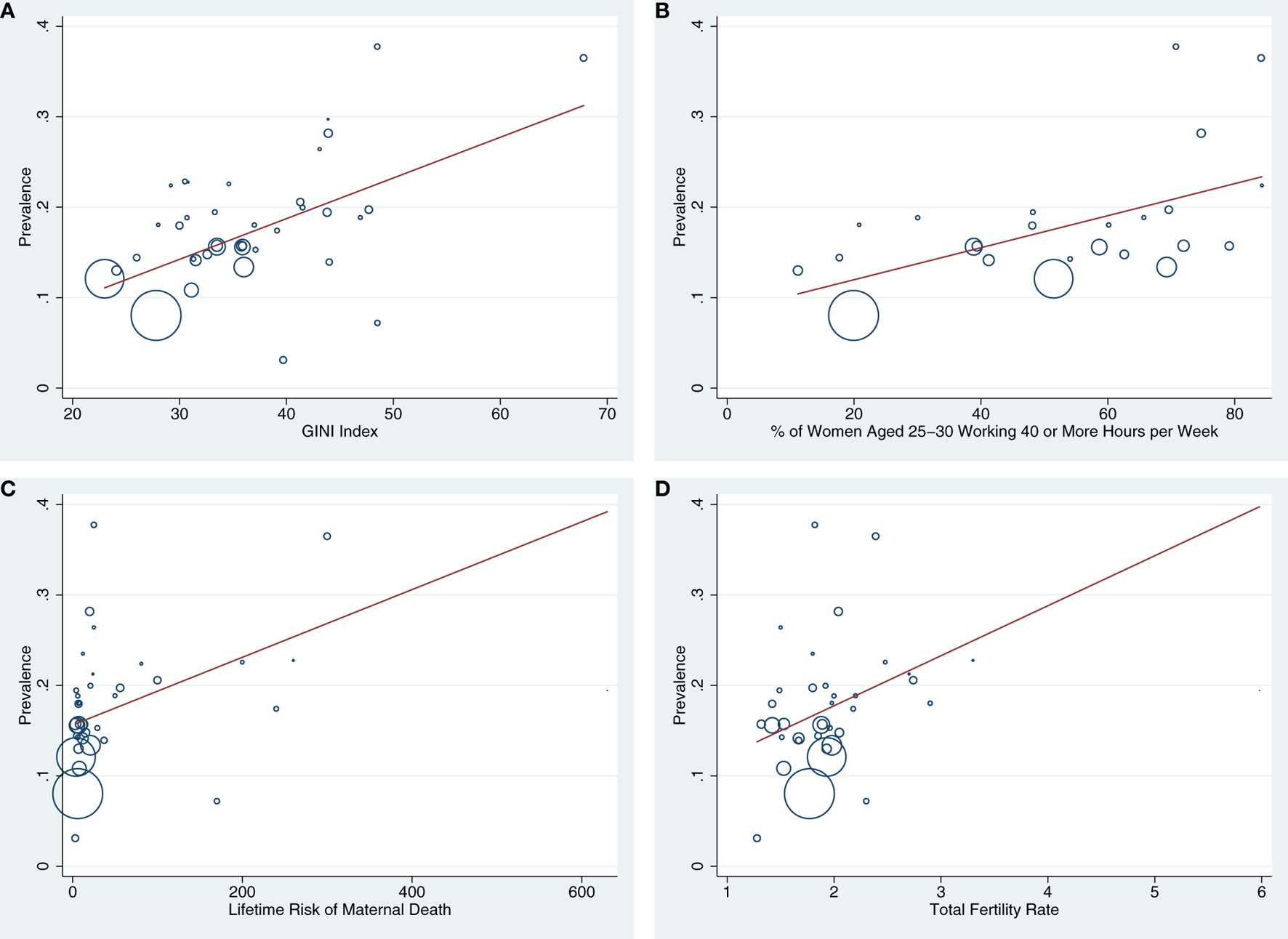
(A–D) Bubble plots are presented showing the associations between GINI index (A), % of women aged 25–30 working ≥40 h a week (B), lifetime risk of maternal death (C), and total fertility rate (D) with national postpartum depression (PPD) prevalence. Countries with larger bubbles had larger sample sizes and were weighted accordingly in meta-regression models.
Health Predictors
Rates of maternal mortality and total fertility in Nigeria were more than 4 SDs above the mean, therefore Nigeria was excluded from analyses involving these factors. Higher prevalence of PPD was reported in countries with higher risk of maternal or infant mortality (maternal mortality: N = 36; Coeff. = 0.045, SE = 0.019, p = 0.024, CI = 0.006 to 0.085), R2 change = 18.73%, see Figure 4C; infant mortality: N = 36; Coeff. = 0.039, SE = 0.018, p = 0.034, CI: 0.003 to 0.074; R2 change = 15.56%). There were also statistical trends suggesting that higher national PPD prevalence was associated with higher total fertility rates (N = 36; Coeff. = 0.040, SE = 0.024, p = 0.102, CI: −0.008 to 0.088; R2 change = 6.33%, see Figure 4D) and higher percentages of infants born low birth weight (N = 36; Coeff. = 0.023, SE = 0.014, p = 0.094, CI: −0.004 to 0.051; R2 change = 9.99%). National cesarean rates did not predict PPD prevalence. Together, these health factors predicted 26.03% of the variance in PPD prevalence, although maternal mortality rate was the only unique predictor in multivariate models when all health variables were included.
Sociodemographic Predictors
The percentages of infants born outside of marriage, living in single parent homes or in urbanized areas did not predict cross-national PPD prevalence.
In sum, economic and health variables explained 73.87% percent of the cross-national variation in PPD [N = 24; F(3, 20) = 13.27, p < 0.001]. Notably, GINI index was the only significant independent predictor of cross-national PPD incidence when all health and economic predictors were included together in the model.
Discussion
In the largest meta-analysis and meta-regression of PPD to date, the global prevalence of PPD was found to be approximately 17.7% (95% CI: 16.6–18.8%). Adjusting for the recommended cutoffs provided by the EPDS for possible (≥10) and probable depression (≥13) yielded prevalence estimates of 21.3 and 16.7%, respectively. These estimates are significantly higher than the widely cited prevalence of 13% (95% CI: 12.3–13.4%), derived from a meta-analysis of studies from developed countries (6). Our estimate is more similar to the 19% prevalence for PPD derived from studies of relatively low- and middle-income countries (7). We found some evidence of publication bias wherein larger studies reported lower PPD prevalence (R2 = 0.8%). However, this effect was small and most likely a byproduct of the fact that countries with more wealth inequality tend to produce studies with smaller sample sizes and wealth inequality (GINI index) between nations predicted 41% of the cross-national variation in PPD in this meta-analysis and meta-regression.
The current meta-analysis also revealed large disparities in PPD prevalence across nations. The countries with the highest rates of PPD were Chile (38%, 95% CI: 35–41%), South Africa (37%; 95% CI: 31–42%), Hong Kong (30%, CI: 28–31%), and Turkey (28%, CI: 27–29%). In contrast, countries with the lowest rates included Singapore (3%; 95% CI: 2–5%), Nepal (7%; 95% CI: 5–10%), the Netherlands (8%; 95% CI: 7–9%), and Switzerland (11%; 95% CI: 7–13%). Surprisingly, these national differences in PPD prevalence could not be explained by methodological conventions used in different counties, for example, the typical EPDS cutoff used, sample size, or the timing of PPD assessment. Instead, the vast majority (73%) of the cross-national variation in PPD prevalence could be explained by economic and health disparities between nations.
Notably, national disparities in PPD appear to exist even among countries that fall within similar economic strata. For example, Chile evinced the highest rates of PPD whereas another high-income nation, the Netherlands, had among the lowest. As many scholars have pointed out (306–308), aggregate wealth metrics like GDP give only a very limited picture of the circumstances of large portions of the population. Instead, we found that wealth disparities (i.e., GINI coefficients) was the most robust predictor of cross-national variation in PPD. Countries with higher GINI coefficients have a greater proportion of citizens living in abject poverty, which is a potent predictor of many mental and physical health problems (309). As previous investigators have also noted, living below the material standards of one’s society equates to possessing low social status—regardless of objective income—which can limit access to less tangible resources like education, opportunity, and security (308). Loss of these forms of social capital is thought to contribute to family dysfunction, health problems, and mood disorders (28).
Relatedly, countries with higher rates of wealth inequality in this meta-analysis also tended to have a higher percentage of women of childbearing age working full-time (Coef. = 0.553, SE = 0.126, p = 0.001, CI: = 0.250 to 856, R2 = 36.9%). This fact may partially explain why countries in which higher proportions of women of childbearing age work full-time have a higher prevalence of PPD. Working full-time while caring for young children can place multiple demands on new mothers (310, 311), causing stress and family discord linked to PPD. These findings militate for PPD intervention efforts focusing on providing support for working mothers.
Our finding that maternal mortality predicts 19% of the cross-national variation in PPD prevalence can be interpreted in several ways. First, suicide linked to mental illness is a major cause of maternal mortality in many countries (1, 2). However, maternal mortality is also a reliable proxy of poor access to medical care, consistent with our finding that higher rates of infant mortality and low birth weight also predicted higher national PPD prevalence. The relationship between maternal mortality and PPD is likely bidirectional, with PPD driving maternal mortality rates and poor healthcare driving both maternal mortality and PPD risk. Therefore, efforts to improve either of these outcomes are likely to evince spillover benefits improving the other. Relatedly, high total fertility rates predicted elevated PPD prevalence, suggesting that improved access to contraception associated with healthcare services may also reduce national PPD prevalence.
Limitations
Several methodological limitations should be considered when interpreting the results of this meta-analysis and meta-regression. First, clinical interviews are the gold standard for PPD diagnosis, whereas our analysis focused on a widely used self-report measure. Self-report measures tend to yield higher estimates of PPD than clinical interviews, therefore, our estimates are likely higher than if we had focused on interview methods (6). However, given the serious consequences of PPD, we felt it was better to potentially overestimate than to underestimate national prevalence. Second, several countries had few studies (e.g., Finland, Mexico, and Nepal), rendering those national estimates less reliable relative to countries where the bulk of PPD research has been done (e.g., the United States, the United Kingdom, and Australia). Finally, many potential predictors of cross-national PPD prevalence were beyond the scope of this study ranging from degree of cultural collectivism to rates of vitamin D deficiency (311–313). We hope that the data set provided in this study will allow future researchers to uncover additional structural, cultural and health predictors of cross-national variation in PPD prevalence.
Conclusion
In sum, our findings reveal that the global prevalence of PPD is both higher and more variable than previously thought, and that wealth inequality, maternal-child health indexes, and employment patterns explain most of the cross-national variation. Creating meaningful improvements in these areas presents enormous social challenges, yet the potential benefits of reducing PPD for mothers, families, and infants are equally great.
Statements
Author contributions
JH-H conceptualized the research questions, conducted the analysis, wrote the paper, and approved this manuscript. TC-H and IA helped to compile the data set, write the manuscript, and approved this manuscript.
Acknowledgments
The authors would like to thank Taylor Delaney, Lilly Murphy, Holly Rankin, Ian Nel, and Nicole Wright for assistance in compiling the data set. The authors would also like to acknowledge the work of Halbreich U, Karkun S, Norhayati MN, Hazlina NN, Asrenee AR, Emilin WW, Fisher A, Cabral de Mello C, Patel V, Rahman A, Tran T, Holton S, and Holmes W for creating the careful systematic reviews on which this work heavily relied.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at https://www.frontiersin.org/articles/10.3389/fpsyt.2017.00248/full#supplementary-material.
References
1
OatesM. Perinatal psychiatric disorders: a leading cause of maternal morbidity and mortality. Br Med Bull (2003) 67(1):219–29.10.1093/bmb/ldg011
2
MirandaJJPatelV. Achieving the millennium development goals: does mental health play a role?PLoS Med (2005) 2(10):e291.10.1371/journal.pmed.0020291
3
FieldT. Postpartum depression effects on early interactions, parenting, and safety practices: a review. Infant Behav. Dev. (2010) 33(1):1–6.10.1016/j.infbeh.2010.04.005
4
PaulsonJFDauberSLeifermanJA. Individual and combined effects of postpartum depression in mothers and fathers on parenting behavior. Pediatrics (2006) 118(2):659–68.10.1542/peds.2005-2948
5
GraceSLEvindarAStewartD. The effect of postpartum depression on child cognitive development and behavior: a review and critical analysis of the literature. Arch Womens Ment Health (2003) 6(4):263–74.10.1007/s00737-003-0024-6
6
O’haraMWSwainAM. Rates and risk of postpartum depression—a meta-analysis. Int Rev Psychiatry (1996) 8(1):37–54.10.3109/09540269609037816
7
FisherJCabral de MelloMelloMPatelVRahmanATranTHoltonSet alPrevalence and determinants of common perinatal mental disorders in women in low-and lower-middle-income countries: a systematic review. Bull WHO (2012) 90(2):139–49.10.2471/BLT.11.091850
8
HalbreichUKarkunS. Cross-cultural and social diversity of prevalence of postpartum depression and depressive symptoms. J Affect Disord (2006) 91(2):97–111.10.1016/j.jad.2005.12.051
9
NorhayatiMHazlinaNNAsreneeAEmilinW. Magnitude and risk factors for postpartum symptoms: a literature review. J Affect Disord (2015) 175:34–52.10.1016/j.jad.2014.12.041
10
MoherDLiberatiATetzlaffJ. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med (2009) 6(7):e1000097.10.1371/journal.pmed.1000097
11
CoxJLHoldenJMSagovskyR. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry (1987) 150(6):782–6.10.1192/bjp.150.6.782
12
YimSTanner StapletonLGuardinoCHahn-HolbrookJDunkel SchetterC. Biological and psychosocial predictors of postpartum depression: systematic review and call for integration. Ann Rev Clin Psychol (2015) 11:99–37.10.1146/annurev-clinpsy-101414-020426
13
American Psychiatric Association. Diagnositic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: Am. Psychiatr. Publ.
14
World Health Organization. International Statistical Classification of Diseases and Related Health Problems. 10th ed. Geneva: World Health Organisation (2004).
15
AliG-CRyanGDe SilvaMJ. Validated screening tools for common mental disorders in low and middle income countries: a systematic review. PLoS One (2016) 11(6):e0156939.10.1371/journal.pone.0156939
16
EvagorouOArvanitiASamakouriM. Cross-cultural approach of postpartum depression: manifestation, practices applied, risk factors and therapeutic interventions. Psychiatr Q (2016) 87(1):129–54.10.1007/s11126-015-9367-1
17
GaynesBNGavinNMeltzer-BrodySet al (2005). Perinatal depression: prevalence, screening accuracy, and screening outcomes: Summary.
18
MarshallJBethellK. Edinburgh Postnatal Depression Scale (EPDS): Translated Versions – Validated. Perth, Western Australia: Department of Health, Government of Western Australia (2006).
19
UNICEF. The State of the World’s Children 2015: Reimagine the Future: Innovation for Every Child. New York, NY: UNICEF (2015).
20
GibbonsLBelizánJMLauerJABetránAMerialdiMAlthabeF. The global numbers and costs of additionally needed and unnecessary caesarean sections performed per year: overuse as a barrier to universal coverage. World Health Rep (2010) 30:1–31.
21
OrtizICumminsM. (2011). Global Inequality: Beyond the bottom billion – a rapid review of income distribution in 141 countries.
22
Development OfEC-Oa. (2015). Annual labour force statistics: LFS by sex and age…
23
AddatiLCassirerNGilchristK. Maternity and Paternity at Work: Law and Practice Across the World. Geneva: International Labour Office (2014).
24
TrendsC. World Family Map: Mapping Family Change and Child Well-Being Outcomes. Child Trends (2014).
25
BarendregtJJDoiSALeeYYNormanREVosT. Meta-analysis of prevalence. J Epidemiol Community Health (2013) 67(11):974–8.10.1136/jech-2013-203104
26
Huedo-MedinaTBSánchez-MecaJMarín-MartínezFBotellaJ. Assessing heterogeneity in meta-analysis: Q statistic or I2 index?Psychol Methods (2006) 11(2):193.10.1037/1082-989X.11.2.193
27
ValentineJCPigottTDRothsteinHR. How many studies do you need? A primer on statistical power for meta-analysis. J Educ Behav Stat (2010) 35(2):215–47.10.3102/1076998609346961
28
StataCorpL. Intercooled Stata 9.1 for Windows. College Station: Stata-Corp LP (2005).
29
AffonsoDDDeAKHorowitzJAMayberryLJ. An international study exploring levels of postpartum depressive symptomatology. J Psychosom Res (2000) 49(3):207–16.10.1016/S0022-3999(00)00176-8
30
AlcornKLO’DonovanAPatrickJCCreedyDDevillyGJ. A prospective longitudinal study of the prevalence of post-traumatic stress disorder resulting from childbirth events. Psychol Med (2010) 40(11):1849–59.10.1017/S0033291709992224
31
ArmstrongKLHaeringenAVDaddsMRCashR. Sleep deprivation or postnatal depression in later infancy: separating the chicken from the egg. J Paediatr Child Health (1998) 34(3):260–2.10.1046/j.1440-1754.1998.00213.x
32
AstburyJBrownSLumleyJSmallR. Birth events, birth experiences and social differences in postnatal depression. Aust J Public Health (1994) 18(2):176–84.10.1111/j.1753-6405.1994.tb00222.x
33
BilsztaJCGuYZMeyerDBuistAE. A geographic comparison of the prevalence and risk factors for postnatal depression in an Australian population. Aust N Z J Public Health (2008) 32(5):424–30.10.1111/j.1753-6405.2008.00274.x
34
BoycePHickeyA. Psychosocial risk factors to major depression after childbirth. Soc Psychiatry Psychiatr Epidemiol (2005) 40(8):605–12.10.1007/s00127-005-0931-0
35
BoycePMStubbsJToddAL. The Edinburgh Postnatal Depression Scale: validation for an Australian sample. Aust N Z J Psychiatry (1993) 27(3):472–6.10.3109/00048679309075805
36
BrooksJNathanESpeelmanCSwalmDJacquesADohertyD. Tailoring screening protocols for perinatal depression: prevalence of high risk across obstetric services in Western Australia. Arch Womens Ment Health (2009) 12(2):105–12.10.1007/s00737-009-0048-7
37
BrownSLumleyJ. Changing childbirth: lessons from an Australian survey of 1336 women. Br J Obstet Gynaecol (1998) 105(2):143–55.
38
BuistAEAustinMPHayesBASpeelmanCBilsztaJLGemmillAWet alPostnatal mental health of women giving birth in Australia 2002-2004: findings from the beyondblue National Postnatal Depression Program. Aust N Z J Psychiatry (2008) 42(1):66–73.10.1080/00048670701732749
39
CondonJTCorkindaleC. The correlates of antenatal attachment in pregnant women. Br J Med Psychol (1997) 70(4):359–72.10.1111/j.2044-8341.1997.tb01912.x
40
EastwoodJGPhungHBarnettB. Postnatal depression and socio-demographic risk: factors associated with Edinburgh Depression Scale scores in a metropolitan area of New South Wales, Australia. Aust N Z J Psychiatry (2011) 45(12):1040–6.10.3109/00048674.2011.619160
41
EastwoodJGJalaludinBBKempLAPhungHNBarnettBW. Relationship of postnatal depressive symptoms to infant temperament, maternal expectations, social support and other potential risk factors: findings from a large Australian cross-sectional study. BMC Pregnancy Childbirth (2012) 12:148.10.1186/1471-2393-12-148
42
EdwardsBGalletlyCSemmler-BoothTDekkerG. Antenatal psychosocial risk factors and depression among women living in socioeconomically disadvantaged suburbs in Adelaide, South Australia. Aust N Z J Psychiatry (2008) 42(1):45–50.10.1080/00048670701732673
43
GriepsmaJMarcolloJCaseyCCherryFVaryEWaltonV. The incidence of postnatal depression in a rural area and the needs of affected women. Aust J Adv Nurs (1994) 11(4):19–23.
44
LeighBMilgromJ. Risk factors for antenatal depression, postnatal depression and parenting stress. BMC Psychiatry (2008) 8:24.10.1186/1471-244X-8-24
45
MaloneyDM. Postnatal depression: a study of mothers in the metropolitan area of Perth, Western Australia. Aust Coll Midwives Inc J (1998) 11(2):18–23.10.1016/S1031-170X(98)80030-5
46
MillerRLPallantJFNegriLM. Anxiety and stress in the postpartum: is there more to postnatal distress than depression?BMC Psychiatry (2006) 6:12.10.1186/1471-244X-6-12
47
StampGECrowtherCA. Postnatal depression: a South Australian prospective survey. Aust N Z J Obstet Gynaecol (1994) 34(2):164–7.10.1111/j.1479-828X.1994.tb02681.x
48
StampGEWilliamsASCrowtherCA. Predicting postnatal depression among pregnant women. Birth (1996) 23(4):218–23.10.1111/j.1523-536X.1996.tb00498.x
49
WhiteTMattheySBoydKBarnettB. Postnatal depression and post-traumatic stress after childbirth: prevalence, course and co-occurrence. J Reprod Infant Psychol (2006) 24(2):107–20.10.1080/02646830600643874
50
WillinickLACottonSM. Risk factors for postnatal depression. Aust Midwifery (2004) 17(2):10–5.10.1016/S1448-8272(04)80004-X
51
WynterKRoweHFisherJ. Common mental disorders in women and men in the first six months after the birth of their first infant: a community study in Victoria, Australia. J Affect Disord (2013) 151(3):980–5.10.1016/j.jad.2013.08.021
52
KohlCWalchTHuberRKemmlerGNeurauterGFuchsDet alMeasurement of tryptophan, kynurenine and neopterin in women with and without postpartum blues. J Affect Disord (2005) 86(2):135–42.10.1016/j.jad.2004.12.013
53
Al DallalFHGrantIN. Postnatal depression among Bahraini women: prevalence of symptoms and psychosocial risk factors. East Mediterr Health J (2012) 18(5):439–45.10.4236/ojog.2015.511086
54
EdhborgMNasreenHKabirZN. Impact of postpartum depressive and anxiety symptoms on mothers’ emotional tie to their infants 2–3 months postpartum: a population-based study from rural Bangladesh. Arch Womens Ment Health (2011) 14(4):307–16.10.1007/s00737-011-0221-7
55
GausiaKFisherCAliMOosthuizenJ. Magnitude and contributory factors of postnatal depression: a community-based cohort study from a rural subdistrict of Bangladesh. Psychol Med (2009) 39(6):999–7.10.1017/S0033291708004455
56
Da-SilvaVAMoraes-SantosARCarvalhoMSMartinsMLTeixeiraNA. Prenatal and postnatal depression among low income Brazilian women. Braz J Med Biol Res (1998) 31(6):799–804.10.1590/S0100-879X1998000600012
57
de AlmeidaLSJansenKKöhlerCAPinheiroRTda SilvaRABoniniJS. Working and short-term memories are impaired in postpartum depression. J Affect Disord (2012) 136(3):1238–42.10.1016/j.jad.2011.09.031
58
FilhaMMAyersSda GamaSGdo Carmo LealM. Factors associated with postpartum depressive symptomatology in Brazil: the Birth in Brazil National Research Study, 2011/2012. J Affect Disord (2016) 194:159–67.10.1016/j.jad.2016.01.020
59
LobatoGMoraesCLDiasASReichenheimME. Postpartum depression according to time frames and sub-groups: a survey in primary health care settings in Rio de Janeiro, Brazil. Arch Womens Ment Health (2011) 14(3):187–93.10.1007/s00737-011-0206-6
60
LobatoGBrunnerMADiasMAMoraesCLReichenheimME. Higher rates of postpartum depression among women lacking care after childbirth: clinical and epidemiological importance of missed postnatal visits. Arch Womens Ment Health (2012) 15(2):145–6.10.1007/s00737-012-0256-4
61
MatijasevichAGoldingJSmithGDSantosISBarrosAJVictoraCG. Differentials and income-related inequalities in maternal depression during the first two years after childbirth: birth cohort studies from Brazil and the UK. Clin Pract Epidemiol Ment Health (2009) 5(1):1.10.1186/1745-0179-5-12
62
MeloEFCecattiJGPacagnellaRCLeiteDFVulcaniDEMakuchMY. The prevalence of perinatal depression and its associated factors in two different settings in Brazil. J Affect Disord (2012) 136(3):1204–8.10.1016/j.jad.2011.11.023
63
MoraisMLucciTKOttaE. Postpartum depression and child development in first year of life. Estudos de Psicologia (Campinas) (2013) 30(1):7–17.10.1590/S0103-166X2013000100002
64
PinheiroRTCoelhoFMSilvaRAPinheiroKAOsesJPQuevedo LdeÁet alAssociation of a serotonin transporter gene polymorphism (5-HTTLPR) and stressful life events with postpartum depressive symptoms: a population-based study. J Psychosom Obstet Gynecol (2013) 34(1):29–33.10.3109/0167482X.2012.759555
65
SilvaRJansenKSouzaLQuevedoLBarbosaLMoraesIet alSociodemographic risk factors of perinatal depression: a cohort study in the public health care system. Rev Bras Psiquiatr (2012) 34(2):143–8.10.1590/S1516-44462012000200005
66
BernazzaniOSaucierJDavidHBorgeatF. Psychosocial predictors of depressive symptomatology level in postpartum women. J Affect Disord (1997) 46(1):39–49.10.1016/S0165-0327(97)00077-3
67
BowenABowenRButtPRahmanKMuhajarineN. Patterns of depression and treatment in pregnant and postpartum women. Can J Psychiatry (2012) 57(3):161–7.10.1177/070674371205700305
68
DaCostaDDritsaMRippenNLowensteynIKhaliféS. Health-related quality of life in postpartum depressed women. Arch Womens Ment Health (2006) 9(2):95–102.10.1007/s00737-005-0108-6
69
DennisCLetourneauN. Global and relationship-specific perceptions of support and the development of postpartum depressive symptomatology. Soc Psychiatry Psychiatr Epidemiol (2007) 42(5):389–95.10.1007/s00127-007-0172-5
70
DennisCRossL. Women’s perceptions of partner support and conflict in the development of postpartum depressive symptoms. J Adv Nurs (2006) 56(6):588–99.10.1111/j.1365-2648.2006.04059.x
71
DennisCVigodS. The relationship between postpartum depression, domestic violence, childhood violence, and substance use: epidemiologic study of a large community sample. Violence Against Women (2013) 19(4):503–17.10.1177/1077801213487057
72
DennisCEJanssenPASingerJ. Identifying women at-risk for postpartum depression in the immediate postpartum period. Acta Psychiatr Scand (2004) 110(5):338–46.10.1111/j.1600-0447.2004.00337.x
73
DennisCHodnettEKentonLWestonJZupancicJStewartDEet alEffect of peer support on prevention of postnatal depression among high risk women: multisite randomised controlled trial. BMJ (2009) 77:280–4.
74
MaltaLAMcDonaldSWHegadorenKMWellerCAToughSC. Influence of interpersonal violence on maternal anxiety, depression, stress and parenting morale in the early postpartum: a community based pregnancy cohort study. BMC Pregnancy Childbirth (2012) 12:153.10.1186/1471-2393-12-153
75
McDonaldSWallJForbesKKingstonDKehlerHVekvedMet alDevelopment of a prenatal psychosocial screening tool for post-partum depression and anxiety. Paediatr Perinat Epidemiol (2012) 26(4):316–27.10.1111/j.1365-3016.2012.01286.x
76
SwordWLandyCKThabaneLWattSKruegerPFarineDet alIs mode of delivery associated with postpartum depression at 6 weeks: a prospective cohort study. BJOG (2011) 118(8):966–77.10.1111/j.1471-0528.2011.02950.x
77
VerreaultNDa CostaDMarchandAIrelandKDritsaMKhaliféS. Rates and risk factors associated with depressive symptoms during pregnancy and with postpartum onset. J Psychosom Obstet Gynecol (2014) 35(3):84–91.10.3109/0167482X.2014.947953
78
VigodSNTarasoffLABryjaBDennisCYudinMHRossLE. Relation between place of residence and postpartum depression. Can Med Assoc J (2013) 185(13):1129–35.10.1503/cmaj.122028
79
FlorenzanoRBottoAMuñizCRojasJAstorquizaJGutierrezL. Frecuencia de síntomas depresivos medidos con el EPDS en puérperas hospitalizadas en el hospital del Salvador [Frequency of depressive symptoms measured with the EPDS in postpartum women hospitalized at the Hospital del Salvador]. Revista Chilena de Neuro-Psiquiatría (2002) 40(Suppl 4):10.10.2147/IJWH.S51436
80
JadresicEJaraCMirandaMArrauBArayaR. Trastornos emocionales en el embarazo y el puerperio: estudio prospectivo de 108 mujeres [Emotional disorders in pregnancy and postpartum: prospective study of 108 women]. Revista Chilena de Neuro-Psiquiatría (1992) 30(2):99–106.
81
JadresicEArayaRJaraC. Validation of the Edinburgh Postnatal Depression Scale (EPDS) in Chilean postpartum women. J Psychosom Obstet Gynaecol (1995) 16(4):187–91.10.3109/01674829509024468
82
RiscoLJadresicEGalleguillosTGarayJLGonzálezMHasbúnJ. Depresión posparto: alta frecuencia en puérperas chilenas, detección precoz, seguimiento y factores de riesgo [Postpartum depression: high frequency in Chilean postpartum, early detection, follow-up and risk factors]. Psiquiatría y Salud Integral (2002) 2:61–6.10.4067/S0034-98872008000100006
83
GaoLChanSWMaoQ. Depression, perceived stress, and social support among first-time Chinese mothers and fathers in the postpartum period. Res Nurs Health (2009) 32(1):50–8.10.1002/nur.20306
84
GaoLLChanSWYouLLiX. Experiences of postpartum depression among first-time mothers in mainland China. J Adv Nurs (2010) 66(2):303–12.10.1111/j.1365-2648.2009.05169.x
85
LeungWCKungFLamJLeungTWHoPC. Domestic violence and postnatal depression in a Chinese community. Int J Gynaecol Obstet (2002) 79(2):159–66.10.1016/S0020-7292(02)00236-9
86
XieRHeGLiuABradwejnJWalkerMWenSW. Fetal gender and postpartum depression in a cohort of Chinese women. Soc Sci Med (2007) 65(4):680–4.10.1016/j.socscimed.2007.04.003
87
XieRHLeiJWangSXieHWalkerMWenSW. Cesarean section and postpartum depression in a cohort of Chinese women with a high cesarean delivery rate. J Womens Health (2011) 20(12):1881–6.10.1089/jwh.2011.2842
88
Nielsen FormanDVidebechPHedegaardMDalby SalvigJSecherNJ. Postpartum depression: identification of women at risk. BJOG (2000) 107(10):1210–7.10.1111/j.1471-0528.2000.tb11609.x
89
HiltunenPRaudaskoskiTEbelingHMoilanenI. Does pain relief during delivery decrease the risk of postnatal depression?Acta Obstet Gynecol Scand (2004) 83(3):257–61.10.1111/j.0001-6349.2004.0302.x
90
LuomaITamminenTKaukonenPLaippalaPPuuraKSalmelinRet alLongitudinal study of maternal depressive symptoms and child well-being. J Am Acad Child Adolesc Psychiatry (2001) 40(12):1367–74.10.1097/00004583-200112000-00006
91
de TycheyCSpitzEBriançonSLighezzoloJGirvanFRosatiAet alPre- and postnatal depression and coping: a comparative approach. J Affect Disord (2005) 85(3):323–6.10.1016/j.jad.2004.11.004
92
DubeyCGuptaNBhasinSMuthalRAAroraR. Prevalence and associated risk factors for postpartum depression in women attending a tertiary hospital, Delhi, IndiaInt J Soc Psychiatry (2012) 58(6):577–80.10.1177/0020764011415210
93
FosseyLPapiernikEBydlowskiM. Postpartum blues: a clinical syndrome and predictor of postnatal depression?J Psychosom Obstet Gynaecol (1997) 18(1):17–21.10.3109/01674829709085564
94
GaillardALe StratYMandelbrotLKeïtaHDubertretC. Predictors of postpartum depression: prospective study of 264 women followed during pregnancy and postpartum. Psychiatry Res (2014) 215(2):341–6.10.1016/j.psychres.2013.10.003
95
Glangeaud-FreudenthalNMCKaminskiM. Severe post-delivery blues: associated factors. Arch Womens Ment Health (1999) 2(1):37–44.10.1007/s007370050033
96
GuedeneyNFermanianJ. Validation study of the French version of the Edinburgh Postnatal Depression Scale (EPDS): new results about use and psychometric properties. Eur Psychiatry (1998) 13(2):83–9.10.1016/S0924-9338(98)80023-0
97
Sutter-DallayALGiaconne-MarcescheVGlatigny-DallayEVerdouxH. Women with anxiety disorders during pregnancy are at increased risk of intense postnatal depressive symptoms: a prospective survey of the MATQUID cohort. Eur Psychiatry (2004) 19(8):459–63.10.1016/j.eurpsy.2004.09.025
98
BallestremCLStraussMKächeleH. Contribution to the epidemiology of postnatal depression in Germany – implications for the utilization of treatment. Arch Womens Ment Health (2005) 8(1):29–35.10.1007/s00737-005-0068-x
99
BergantAMNguyenTHeimKUlmerHDapuntO. Deutschsprachige Fassung und Validierung der Edinburgh postnatal depression scale [German language version and validation of the Edinburgh postnatal depression scale]. Dtsch Med Wochenschr (1998) 123(3):35–40.10.1055/s-2007-1023895
100
MehtaDQuastCFaschingPASeifertAVoigtFBeckmannMWet alThe 5-HTTLPR polymorphism modulates the influence on environmental stressors on peripartum depression symptoms. J Affect Disord (2012) 136(3):1192–7.10.1016/j.jad.2011.11.042
101
ReckCStrubenKBackenstrassMStefenelliUReinigKFuchsTet alPrevalence, onset and comorbidity of postpartum anxiety and depressive disorders. Acta Psychiatr Scand (2008) 118(6):459–68.10.1111/j.1600-0447.2008.01264.x
102
ZaersSWaschkeMEhlertU. Depressive symptoms and symptoms of post-traumatic stress disorder in women after childbirth. J Psychosom Obstet Gynecol (2008) 29(1):61–71.10.1080/01674820701804324
103
ChatziLMelakiVSarriKApostolakiIRoumeliotakiTGeorgiouVet alDietary patterns during pregnancy and the risk of postpartum depression: the mother-child ’Rhea’ cohort in Crete, Greece. Public Health Nutr (2011) 14(9):1663–70.10.1017/S1368980010003629
104
GonidakisFRabavilasADVarsouEKreatsasGChristodoulouGN. A 6-month study of postpartum depression and related factors in Athens Greece. Compr Psychiatry (2008) 49(3):275–82.10.1016/j.comppsych.2007.05.018
105
KoutraKVassilakiMGeorgiouVKoutisABitsiosPChatziLet alAntenatal maternal mental health as determinant of postpartum depression in a population based mother – child cohort (Rhea Study) in Crete, Greece. Soc Psychiatry Psychiatr Epidemiol (2014) 49(5):711–21.10.1007/s00127-013-0758-z
106
LambrinoudakiIRizosDArmeniEPliatsikaPLeonardouASygelouAet alThyroid function and postpartum mood disturbances in Greek women. J Affect Disord (2010) 121(3):278–82.10.1016/j.jad.2009.07.001
107
ThorpeKJDragonasTGoldingJ. The effects of psychosocial factors on the emotional well-being of women during pregnancy: a cross-cultural study of Britain and Greece. J Reprod Infant Psychol (1992) 10(4):191–204.10.1080/02646839208403953
108
LauYChanKS. Influence of intimate partner violence during pregnancy and early postpartum depressive symptoms on breastfeeding among Chinese women in Hong Kong. J Midwifery Womens Health (2007) 52(2):e15–20.10.1016/j.jmwh.2006.09.001
109
LeeDSYipSKChiuHKLeungTSChanKMChauILet alDetecting postnatal depression in Chinese women: validation of the Chinese version of the Edinburgh Postnatal Depression Scale. Br J Psychiatry (1998) 172:433–7.10.1192/bjp.172.5.433
110
LeeDSChungTH. Postnatal depression: an update. Best Pract Res Clin Obstet Gynaecol (2007) 21(2):183–91.10.1016/j.bpobgyn.2006.10.003
111
LeungSKMartinsonIMArthurD. Postpartum depression and related psychosocial variables in Hong Kong Chinese women: findings from a prospective study. Res Nurs Health (2005) 28(1):27–38.10.1002/nur.20053
112
TiwariAChanKLFongDLeungWCBrownridgeDALamHet alThe impact of psychological abuse by an intimate partner on the mental health of pregnant women. BJOG (2008) 115(3):377–84.10.1111/j.1471-0528.2007.01593.x
113
NagyEMolnarPPalAOrvosH. Prevalence rates and socioeconomic characteristics of post-partum depression in Hungary. Psychiatry Res (2011) 185(1):113–20.10.1016/j.psychres.2010.05.005
114
ThomeM. Predictors of postpartum depressive symptoms in Icelandic women. Arch Womens Ment Health (2000) 3(1):7–14.10.1007/PL00010326
115
GhoshAGoswamiS. Evaluation of post partum depression in a tertiary hospital. J Obstet Gynaecol India (2011) 61(5):528–30.10.1007/s13224-011-0077-9
116
JainATyagiPKaurPPuliyelJSreenivasV. Association of birth of girls with postnatal depression and exclusive breastfeeding: an observational study. BMJ Open (2014) 4(6):e003545.10.1136/bmjopen-2013-003545
117
MariamKASrinivasanK. Antenatal psychological distress and postnatal depression: a prospective study from an urban clinic. Asian J Psychiatry (2009) 2(2):71–3.10.1016/j.ajp.2009.04.002
118
PatelHLGanjiwaleJDNimbalkarASVaniSNVasaRNimbalkarSM. Characteristics of Postpartum Depression in Anand District, Gujarat, India. J Trop Pediatr (2015) 61(5):364–9.10.1093/tropej/fmv046
119
PatelVRodriguesMDeSouzaN. Gender, poverty, and postnatal depression: a study of mothers in Goa, India. Am J Psychiatry (2002) 159(1):43–7.10.1176/appi.ajp.159.1.43
120
Andajani-SutjahjoSMandersonLAstburyJ. Complex emotions, complex problems: understanding the experiences of perinatal depression among new mothers in urban Indonesia. Cult Med Psychiatry (2007) 31(1):101–22.10.1007/s11013-006-9040-0
121
AbbasiMVan den AkkerOBewleyC. Persian couples’ experiences of depressive symptoms and health-related quality of life in the pre- and perinatal period. J Psychosom Obstet Gynaecol (2014) 35(1):16–21.10.3109/0167482X.2013.865722
122
AbdollahiFRohaniSSazlinaGSZarghamiMAzharMZLyeMSet alBio-psycho-socio-demographic and obstetric predictors of postpartum depression in pregnancy: a prospective cohort study. Iran J Psychiatry Behav Sci (2014) 8(2):11–21.
123
GoshtasebiAAlizadehMGandevaniSB. Association between maternal anaemia and postpartum depression in an urban sample of pregnant women in Iran. J Health Popul Nutr (2013) 31(3):398–402.10.3329/jhpn.v31i3.16832
124
KheirabadiGRMaracyMR. Perinatal depression in a cohort study on Iranian women. J Res Med Sci (2010) 15(1):41–9.
125
MontazeriATorkanBOmidvariS. The Edinburgh Postnatal Depression Scale (EPDS): translation and validation study of the Iranian version. BMC Psychiatry (2007) 7:11.10.1186/1471-244X-7-11
126
AhmedHMAlalafSKAl-TawilNG. Screening for postpartum depression using Kurdish version of Edinburgh Postnatal Depression Scale. Arch Gynecol Obstet (2012) 285(5):1249–55.10.1007/s00404-011-2165-6
127
CrottyFSheehanJ. Prevalence and detection of postnatal depression in an Irish community sample. Ir J Psychol Med (2004) 21(4):117–21.10.1017/S0790966700008533
128
CryanEKeoghFConnollyECodySQuinlanADalyI. Depression among postnatal women in an urban Irish community. Ir J Psychol Med (2001) 18(1):5–10.10.1017/S0790966700006145
129
LaneAKevilleRMorrisMKinsellaATurnerMBarryS. Postnatal depression and elation among mothers and their partners: prevalence and predictors. Br J Psychiatry (1997) 171(6):550–5.10.1192/bjp.171.6.550
130
Leahy-WarrenPMcCarthyGCorcoranP. First-time mothers: social support, maternal parental self-efficacy and postnatal depression. J Clin Nurs (2012) 21(3–4):388–97.10.1111/j.1365-2702.2011.03701.x
131
Alfayumi-ZeadnaSKaufman-ShriquiVZeadnaALaudenAShoham-VardiI. The association between sociodemographic characteristics and postpartum depression symptoms among Arab-Bedouin women in southern Israel. Depress Anxiety (2015) 32(2):120–8.10.1002/da.22290
132
BlochMRotenbergNKorenDKleinE. Risk factors associated with the development of postpartum mood disorders. J Affect Disord (2005) 88(1):9–18.10.1016/j.jad.2005.04.007
133
BlochMRotenbergNKorenDKleinE. Risk factors for early postpartum depressive symptoms. Gen Hosp Psychiatry (2006) 28(1):3–8.10.1016/j.genhosppsych.2005.08.006
134
DanknerRGoldbergRPFischRZCrumRM. Cultural elements of postpartum depression. A study of 327 Jewish Jerusalem women. J Reprod Med (2000) 45(2):97–104.
135
Eilat-TsananiSMeromARomanoSReshefALaviITabenkinH. The effect of postpartum depression on women’s consultations with physicians. Isr Med Assoc J (2006) 8(6):406–10.
136
FischRZTadmorOPDanknerRDiamantYZ. Postnatal depression: a prospective study of its prevalence, incidence and psychosocial determinants in an Israeli sample. J Obstet Gynaecol Res (1997) 23(6):547–54.10.1111/j.1447-0756.1997.tb00885.x
137
GlasserSBarellVShohamAZivABoykoVLuskyAet alProspective study of postpartum depression in an Israeli cohort: prevalence, incidence and demographic risk factors. J Psychosom Obstet Gynaecol (1998) 19(3):155–64.10.3109/01674829809025693
138
GlasserSStoskiEKnelerVMagneziR. Postpartum depression among Israeli Bedouin women. Arch Womens Ment Health (2011) 14(3):203–8.10.1007/s00737-011-0216-4
139
BenvenutiPFerraraMNiccolaiCValorianiVCoxJL. The Edinburgh Postnatal Depression Scale: validation for an Italian sample. J Affect Disord (1999) 53(2):137–41.10.1016/S0165-0327(98)00102-5
140
CarpinielloBParianteCMSerriFCostaGCartaMG. Validation of the Edinburgh Postnatal Depression Scale in Italy. J Psychosom Obstet Gynaecol (1997) 18(4):280–5.10.3109/01674829709080700
141
EliseiSLucariniEMurgiaNFerrantiLAttademoL. Perinatal depression: a study of prevalence and of risk and protective factors. Psychiatr Danub (2013) 25(Suppl 2):S258–62.
142
GiardinelliLInnocentiABenniLStefaniniMCLinoGLunardiCet alDepression and anxiety in perinatal period: prevalence and risk factors in an Italian sample. Arch Womens Ment Health (2012) 15(1):21–30.10.1007/s00737-011-0249-8
143
GormanLLO’HaraMWFigueiredoBHayesSJacquemainFKammererMHet alAdaptation of the Structured Clinical interview for DSM-IV Disorders for assessing depression in women during pregnancy and post-partum across countries and cultures. Br J Psychiatry (2004) 184(Suppl 46):s17–23.10.1192/bjp.184.46.s17
144
GrussuPQuatraroRM. Prevalence and risk factors for a high level of postnatal depression symptomatology in Italian women: a sample drawn from ante-natal classes. Eur Psychiatry (2009) 24(5):327–33.10.1016/j.eurpsy.2009.01.006
145
MauriMOppoAMontagnaniMSBorriCBantiSCamilleriVet alBeyond “postpartum depressions:” Specific anxiety diagnoses during pregnancy predict different outcomes: results from PND-ReScU. J Affect Disord (2010) 127(1):177–84.10.1016/j.jad.2010.05.015
146
OppoAMauriMRamacciottiDCamilleriVBantiSBorriCet alRisk factors for postpartum depression: the role of the Postpartum Depression Predictors Inventory-Revised (PDPI-R): results from the Perinatal Depression-Research & Screening Unit (PNDReScU) study. Arch Womens Ment Health (2009) 12(4):239–49.10.1007/s00737-009-0071-8
147
MatsumotoKTsuchiyaKJItohHKanayamaNSudaSMatsuzakiHet alAge-specific 3-month cumulative incidence of postpartum depression: the Hamamatsu Birth Cohort (HBC) Study. J Affect Disord (2011) 133(3):607–10.10.1016/j.jad.2011.04.024
148
MiyakeYSasakiSTanakaKYokoyamaTOhyaYFukushimaWet alDietary folate and vitamins B12, B6, and B2 intake and the risk of postpartum depression in Japan: the Osaka maternal and child health study. J Affect Disord (2006) 96(1–2):133–8.10.1016/j.jad.2006.05.024
149
NishigoriHSugawaraJObaraTNishigoriTSatoKSugiyamaTet alSurveys of postpartum depression in Miyagi, Japan, after the great east Japan earthquake. Arch Womens Ment Health (2014) 17(6):579–81.10.1007/s00737-014-0459-y
150
Nishizono-MaherAKishimotoJYoshidaHUrayamaKMiyatoMOtsukaYet alThe role of self-report questionnaire in the screening of postnatal depression a community sample survey in central Tokyo. Soc Psychiatry Psychiatr Epidemiol (2004) 39(3):185–90.10.1007/s00127-004-0727-7
151
OhokaHKoideTGotoSMuraseSKanaiAMasudaTet alEffects of maternal depressive symptomatology during pregnancy and the postpartum period on infant-mother attachment. Psychiatry Clin Neurosci (2014) 68(8):631–9.10.1111/pcn.12171
152
ShimizuANishiumiHOkumuraYWatanabeK. Depressive symptoms and changes in physiological and social factors 1 week to 4 months postpartum in Japan. J Affect Disord (2015) 179:175–82.10.1016/j.jad.2015.03.036
153
TamakiRMurataMOkanoT. Risk factors for postpartum depression in Japan. Psychiatry Clin Neurosci (1997) 51(3):93–8.10.1111/j.1440-1819.1997.tb02368.x
154
UedaMYamashitaHYoshidaK. Impact of infant health problems on postnatal depression: pilot study to evaluate a health visiting system. Psychiatry Clin Neurosci (2006) 60(2):182–9.10.1111/j.1440-1819.2006.01484.x
155
WatanabeMWadaKSakataYAratakeYKatoNOhtaHet alMaternity blues as predictor of postpartum depression: a prospective cohort study among Japanese women. J Psychosom Obstet Gynecol (2008) 29(3):211–7.10.1080/01674820801990577
156
YamashitaHYoshidaKNakanoHTashiroN. Postnatal depression in Japanese women: detecting the early onset of postnatal depression by closely monitoring the postpartum mood. J Affect Disord (2000) 58(2):145–54.10.1016/S0165-0327(99)00108-1
157
BangKS. Infants’ temperament and health problems according to maternal postpartum depression. J Korean Acad Nurs (2011) 41(4):444–50.10.4040/jkan.2011.41.4.444
158
KimJJGordonTJLa PorteLMAdamsMKuendigJMSilverRK. The utility of maternal depression screening in the third trimester. Am J Obstet Gynecol (2008) 199(5): 509.e1–5.10.1016/j.ajog.2008.04.018
159
ChaayaMCampbellOMEl KakFShaarDHarbHKaddourA. Postpartum depression: prevalence and determinants in Lebanon. Arch Womens Ment Health (2002) 5(2):65–72.10.1007/s00737-002-0140-8
160
El-HachemCRohayemJKhalilRBRichaSKesrouaniAGemayelRet alEarly identification of women at risk of postpartum depression using the Edinburgh Postnatal Depression Scale (EPDS) in a sample of Lebanese women. BioMed Cent Psychiatry (2014) 14(1):1–16.10.1186/s12888-014-0242-7
161
DowADubeQPenceBWVan RieA. Postpartum depression and HIV infection among women in Malawi. J Acquir Immune Defic Syndr (1999) 65(3):359–65.10.1097/QAI.0000000000000050
162
AzidahAKShaifulBIRusliNJamilMY. Postnatal depression and socio-cultural practices among postnatal mothers in Kota Bahru, Kelantan, Malaysia. Med J Malays (2006) 61(1):76–83.
163
KadirAADaudMNYaacobMJHussainNH. Relationship between obstetric risk factors and postnatal depression in Malaysian women. Int Med J (2009) 16(2):101–6.
164
KitLKJanetGJegasothyR. Incidence of postnatal depression in Malaysian women. J Obstet Gynaecol Res (1997) 23(1):85–9.10.1111/j.1447-0756.1997.tb00811.x
165
YusuffASTangLBinnsCWLeeAH. Prevalence and risk factors for postnatal depression in Sabah, Malaysia: a cohort study. Women Birth (2015) 28(1):25–9.10.1016/j.wombi.2014.11.002
166
FeliceESalibaJGrechVCoxJ. Prevalence rates and psychosocial characteristics associated with depression in pregnancy and postpartum in Maltese women. J Affect Disord (2004) 82(2):297–301.10.1016/j.jad.2003.11.011
167
deCastroFHinojosa-AyalaNHernandez-PradoB. Risk and protective factors associated with postnatal depression in Mexican adolescents. J Psychosom Obstet Gynecol (2011) 32(4):210–7.10.3109/0167482X.2011.626543
168
Flores-QuijanoMECórdovaAContreras-RamírezVFarias-HernándezLCruz TolentinoMCasanuevaE. Risk for postpartum depression, breastfeeding practices, and mammary gland permeability. J Hum Lact (2008) 24(1):50–7.10.1177/0890334407310587
169
AgoubMMoussaouiDBattasO. Prevalence of postpartum depression in a Moroccan sample. Arch Womens Ment Health (2005) 8(1):37–43.10.1007/s00737-005-0069-9
170
AlamiKMKadriNBerradaS. Prevalence and psychosocial correlates of depressed mood during pregnancy and after childbirth in a Moroccan sample. Arch Womens Ment Health (2006) 9(6):343–6.10.1007/s00737-006-0154-8
171
Dørheim Ho-YenSTschudi BondevikGEberhard-GranMBjorvatnB. The prevalence of depressive symptoms in the postnatal period in Lalitpur district, Nepal. Acta Obstet Gynecol Scand (2006) 85(10):1186–92.10.1080/00016340600753158
172
RegmiSSliglWCarterDGrutWSeearM. A controlled study of postpartum depression among Nepalese women: validation of the Edinburgh Postpartum Depression Scale in Kathmandu. Trop Med Int Health (2002) 7(4):378–82.10.1046/j.1365-3156.2002.00866.x
173
BlomEAJansenPWVerhulstFCHofmanARaatHJaddoeVVet alPerinatal complications increase the risk of postpartum depression: the generation R Study. BJOG (2010) 117(11):1390–8.10.1111/j.1471-0528.2010.02660.x
174
VerkerkGJDenolletJVan HeckGLVan SonMJPopVJ. Patient preference for counselling predicts postpartum depression: a prospective 1-year follow up study in high-risk women. J Affect Disord (2004) 83(1):43–8.10.1016/j.jad.2004.04.011
175
AbbottMWWilliamsMM. Postnatal depressive symptoms among Pacific mothers in Auckland: prevalence and risk factors. Aust N Z J Psychiatry (2006) 40(3):230–8.10.1080/j.1440-1614.2006.01779.x
176
HoltWJ. The detection of postnatal depression in general practice using the Edinburgh Postnatal Depression Scale. N Z Med J (1995) 108(994):57–9.
177
McGillHBurrowsVLHollandLALangerHJSweetMA. Postnatal depression: a Christchurch study. N Z Med J (1995) 108(999):162–5.
178
WebsterMLThompsonJDMitchellEAWerryJS. Postnatal depression in a community cohort. Aust N Z J Psychiatry (1994) 28(1):42–9.10.3109/00048679409075844
179
AbiodunOA. Postnatal depression in primary care populations in Nigeria. Gen Hosp Psychiatry (2006) 28(2):133–6.10.1016/j.genhosppsych.2005.11.002
180
AdewuyaAOOlaBADadaAOFasotoOO. Validation of the Edinburgh Postnatal Depression Scale as a screening tool for depression in late pregnancy among Nigerian women. J Psychosom Obstet Gynecol (2006) 27(4):267–72.10.1080/01674820600915478
181
AdewuyaAOFatoyeFOOlaBAIjaodolaORIbigbamiSM. Sociodemographic and obstetric risk factors for postpartum depressive symptoms in Nigerian women. J Psychiatr Pract (2015) 11(5):353–8.10.1097/00131746-200509000-00009
182
BakareMOOkoyeJOObindoJT. Introducing depression and developmental screenings into the National Programme on Immunization (NPI) in southeast Nigeria: an experimental cross-sectional assessment. Gen Hosp Psychiatry (2014) 36(1):105–12.10.1016/j.genhosppsych.2013.09.005
183
DørheimSKBondevikGTEberhard-GranMBjorvatnB. Sleep and depression in postpartum women: a population-based study. Sleep (2009) 32(7):847–55.10.1093/sleep/32.7.847
184
DørheimSKBjorvatnBEberhard-GranM. Can insomnia in pregnancy predict postpartum depression? A longitudinal, population-based study. PLoS One (2014) 9(4):e94674.10.1371/journal.pone.0094674
185
Eberhard-GranMEskildATambsKOpjordsmoenSSamuelsenSO. Review of validation studies of the Edinburgh Postnatal Depression Scale. Acta Psychiatr Scand (2001) 104(4):243–9.10.1034/j.1600-0447.2001.00187.x
186
Eberhard-GranMEskildATambsKSamuelsenSOOpjordsmoenS. Depression in postpartum and non-postpartum women: prevalence and risk factors. Acta Psychiatr Scand (2002) 106(6):426–33.10.1034/j.1600-0447.2002.02408.x
187
Eberhard-GranMTambsKOpjordsmoenSSkrondalAEskildA. Depression during pregnancy and after delivery: a repeated measurement study. J Psychosom Obstet Gynecol (2004) 25(1):15–21.10.1080/01674820410001737405
188
GlavinKSmithLSørumR. Prevalence of postpartum depression in two municipalities in Norway. Scand J Caring Sci (2009) 23(4):705–10.10.1111/j.1471-6712.2008.00667.x
189
HagaSMUllebergPSlinningKKraftPSteenTBStaffA. A longitudinal study of postpartum depressive symptoms: multilevel growth curve analyses of emotion regulation strategies, breastfeeding self-efficacy, and social support. Arch Womens Ment Health (2012) 15(3):175–84.10.1007/s00737-012-0274-2
190
MarkhusMWSkotheimSGraffIEFrøylandLBraarudHCStormarkKMet alLow omega-3 index in pregnancy is a possible biological risk factor for postpartum depression. PLoS One (2013) 8:7.10.1371/journal.pone.0067617
191
NordengHHansenCGarthus-NiegelSEberhard-GranM. Fear of childbirth, mental health, and medication use during pregnancy. Arch Womens Ment Health (2012) 15(3):203–9.10.1007/s00737-012-0278-y
192
AhmadIKhanM. Risk factors associated with post-natal depression in Pakistani women. Pak J Soc Clin Psychol (2005) 3:41–50.10.1186/s13104-015-1074-3
193
HusainNBevcIHusainMChaudhryIBAtifNRahmanA. Prevalence and social correlates of postnatal depression in a low income country. Arch Womens Ment Health (2006) 9(4):197–202.10.1007/s00737-006-0129-9
194
AyoubKA. Prevalence of Postpartum Depression among Recently Delivering Mothers in Nablus District and Its Associated Factors (Unpublished Master’s Thesis). Nablus, Palestine: An-Najah National University (2014).
195
DudekDJaeschkeRSiwekMMaczkaGTopór-MadryRRybakowskiJ. Postpartum depression: identifying associations with bipolarity and personality traits. Preliminary results from a cross-sectional study in Poland. Psychiatry Res (2014) 215(1):69–74.10.1016/j.psychres.2013.10.013
196
AugustoAKumarRCalheirosJMMatosEFigueiredoE. Post-natal depression in an urban area of Portugal: comparison of childbearing women and matched controls. Psychol Med (1996) 26(1):135–41.10.1017/S0033291700033778
197
FigueiredoBCondeA. Anxiety and depression in women and men from early pregnancy to 3-months postpartum. Arch Womens Ment Health (2011) 14(3):247–55.10.1007/s00737-011-0217-3
198
FigueiredoBCostaR. Mother’s stress, mood and emotional involvement with the infant: 3 months before and 3 months after childbirth. Arch Womens Ment Health (2009) 12(3):143–53.10.1007/s00737-009-0059-4
199
FigueiredoBPachecoACostaR. Depression during pregnancy and the postpartum period in adolescent and adult Portuguese mothers. Arch Womens Ment Health (2007) 10(3):103–9.10.1007/s00737-007-0178-8
200
CheeCILeeDSChongYSTanLKNgTRFonesCL. Confinement and other psychosocial factors in perinatal depression: a transcultural study in Singapore. J Affect Disord (2005) 89(1–3):157–66.10.1016/j.jad.2005.09.004
201
KokLPChanPSRatnamSS. Postnatal depression in Singapore women. Singapore Med J (1994) 35(1):33–5.
202
LawrieTAHofmeyrGJDe JagerMBerkMPaikerJViljoenE. A double-blind randomised placebo controlled trial of postnatal norethisterone enanthate: the effect on postnatal depression and serum hormones. Br J Obstet Gynaecol (1998) 105(10):1082–90.10.1111/j.1471-0528.1998.tb09940.x
203
LawrieTAHofmeyrGJDe JagerMBerkM. Validation of the Edinburgh Postnatal Depression Scale on a cohort of South African women. S Afr Med J (1998) 88(10):1340–4.
204
Escribà-AgüirVArtazcozL. Gender differences in postpartum depression: a longitudinal cohort study. J Epidemiol Community Health (2011) 65(4):320–6.10.1136/jech.2008.085894
205
Garcia-EsteveLAscasoCOjuelJNavarroP. Validation of the Edinburgh Postnatal Depression Scale (EPDS) in Spanish mothers. J Affect Disord (2003) 75(1):71–6.10.1016/S0165-0327(02)00020-4
206
Sebastián RomeroEMas LodoNMartín BlázquezMRaja CasillasMIIzquierdo ZamarriegoMJValles FernándezNet alDepresión postparto en el área de salud de Toledo [Postpartum depression in the health area of Toledo]. Atención primaria (1992) 24(4):215–9.
207
AgnaforsSSydsjöGdeKeyserLSvedinCG. Symptoms of depression postpartum and 12 years later-associations to child mental health at 12 years of age. Matern Child Health J (2013) 17(3):405–14.10.1007/s10995-012-0985-z
208
Bågedahl-StrindlundMMonsen BörjessonK. Postnatal depression: a hidden illness. Acta Psychiatr Scand (1998) 98(4):272–5.10.1111/j.1600-0447.1998.tb10083.x
209
JosefssonABergGNordinCSydsjöG. Prevalence of depressive symptoms in late pregnancy and postpartum. Acta Obstet Gynecol Scand (2001) 80(3):251–5.10.1034/j.1600-0412.2001.080003251.x
210
LundhWGyllangC. Use of the Edinburgh Postnatal Depression Scale in some Swedish child health care centres. Scand J Caring Sci (1993) 7(3):149–54.10.1111/j.1471-6712.1993.tb00190.x
211
RubertssonCWickbergBGustavssonPRådestadI. Depressive symptoms in early pregnancy, two months and one year postpartum-prevalence and psychosocial risk factors in a national Swedish sample. Arch Womens Ment Health (2005) 8(2):97–104.10.1007/s00737-005-0078-8
212
SeimyrLEdhborgMLundhWSjögrenB. In the shadow of maternal depressed mood: experiences of parenthood during the first year after childbirth. J Psychosom Obstet Gynecol (2004) 25(1):23–34.10.1080/01674820410001737414
213
SylvénSMEkseliusLSundström-PoromaaISkalkidouA. Premenstrual syndrome and dysphoric disorder as risk factors for postpartum depression. Acta Obstet Gynecol Scand (2013) 92(2):178–84.10.1111/aogs.12041
214
WickbergBHwangCP. Counselling of postnatal depression: a controlled study on a population based Swedish sample. J Affect Disord (1996) 39(3):209–16.10.1016/0165-0327(96)00034-1
215
BurgutFTBenerAGhuloumSSheikhJ. A study of postpartum depression and maternal risk factors in Qatar. J Psychosom Obstet Gynecol (2013) 34(2):90–7.10.3109/0167482X.2013.786036
216
AlharbiAAAbdulghaniHM. Risk factors associated with postpartum depression in the Saudi population. Neuropsychiatr Dis Treat (2014) 10:311–6.10.2147/NDT.S57556
217
Al-ModayferOAlatiqYKhairOAbdelkawiS. Postpartum depression and related risk factors among Saudi females. Int J Cult Ment Health (2015) 8(3):316–24.10.1080/17542863.2014.999691
218
GürberSBielinski-BlattmannDLemolaSJaussiCvon WylASurbekDet alMaternal mental health in the first 3-week postpartum: the impact of caregiver support and the subjective experience of childbirth–a longitudinal path model. J Psychosom Obstet Gynecol (2012) 33(4):176–84.10.3109/0167482X.2012.730584
219
Righetti-VeltemaMConne-PerréardEBousquetAManzanoJ. Risk factors and predictive signs of postpartum depression. J Affect Disord (1998) 49(3):167–80.10.1016/S0165-0327(97)00110-9
220
ChenHHHwangFMTaiCJChienLY. The interrelationships among acculturation, social support, and postpartum depression symptoms among marriage-based immigrant women in Taiwan: a cohort study. J Immigr Minor Health (2013) 15(1):17–23.10.1007/s10903-012-9697-0
221
ChienLYTaiCJYehMC. Domestic decision-making power, social support, and postpartum depression symptoms among immigrant and native women in Taiwan. Nurs Res (2012) 61(2):103–10.10.1097/NNR.0b013e31824482b6
222
HehSCoombesLBartlettH. The association between depressive symptoms and social support in Taiwanese women during the month. Int J Nurs Stud (2004) 41(5):573–9.10.1016/j.ijnurstu.2004.01.003
223
HehSHuangLHoSFuYWangL. Effectiveness of an exercise support program in reducing the severity of postnatal depression in Taiwanese women. Birth (2008) 35(1):60–5.10.1111/j.1523-536X.2007.00192
224
HuangYCMathersNJ. A comparison of sexual satisfaction and post-natal depression in the UK and Taiwan. Int Nurs Rev (2006) 53(3):197–204.10.1111/j.1466-7657.2006.00459.x
225
HuangYCMathersNJ. Postnatal depression and the experience of South Asian marriage migrant women in Taiwan: survey and semi-structured interview study. Int J Nurs Stud (2008) 45(6):924–31.10.1016/j.ijnurstu.2007.02.006
226
LeeSHLiuLCKuoPCLeeMS. Postpartum depression and correlated factors in women who received in vitro fertilization treatment. J Midwifery Womens Health (2011) 56(4):347–52.10.1111/j.1542-2011.2011.00033.x
227
TengHWHsuCSShihSMLuMLPanJJShenWW. Screening postpartum depression with the Taiwanese version of the Edinburgh Postnatal Depression Scale. Compr Psychiatry (2005) 46(4):261–5.10.1016/j.comppsych.2004.10.003
228
TsaoYCreedyDKGambleJ. Prevalence and psychological correlates of postnatal depression in rural Taiwanese women. Health Care Women Int (2015) 36(4):457–74.10.1080/07399332.2014.946510
229
LimlomwongseNLiabsuetrakulT. Cohort study of depressive moods in Thai women during late pregnancy and 6-8 weeks of postpartum using the Edinburgh Postnatal Depression Scale (EPDS). Arch Womens Ment Health (2006) 9(3):131–8.10.1007/s00737-005-0115-7
230
AkmanIKuscuMKYurdakulZÖzdemirNSolakoğluMOrhonLet alBreastfeeding duration and postpartum psychological adjustment: role of maternal attachment styles. J Paediatr Child Health (2008) 44(6):369–73.10.1111/j.1440-1754.2008.01336.x
231
AlkarÖYGençözT. Critical factors associated with early postpartum depression among Turkish women. Contemp Fam Ther (2005) 27(2):263–75.10.1007/s10591-005-4043-5
232
AydinNInandiTKarabulutN. Depression and associated factors among women within their first postnatal year in Erzurum province in Eastern Turkey. Women Health (2005) 41(2):1–12.10.1300/J013v41n02_01
233
AydinNInandiTYigitAHodoglugilNN. Validation of the Turkish version of the Edinburgh Postnatal Depression Scale among women within their first postpartum year. Soc Psychiatry Psychiatr Epidemiol (2004) 39(6):483–6.10.1007/s00127-004-0770-4
234
AyvazSHocaoğluCTiryakiAAkI. Incidence of postpartum depression in Trabzon province and risk factors at gestation. Turk Psikiyatri Derg (2006) 17(4):243–51.
235
BugdayciRSasmazCTTezcanHKurtAÖÖnerS. A cross-sectional prevalence study of depression at various times after delivery in Mersin province in Turkey. J Womens Health (2004) 13(1):63–8.10.1089/154099904322836465
236
DanaciAEDinçGDeveciAŞenFSİçelliİ. Postnatal depression in Turkey: epidemiological and cultural aspects. Soc Psychiatry Psychiatr Epidemiol (2002) 37(3):125–9.10.1007/s001270200004
237
DindarIErdoganS. Screening of Turkish women for postpartum depression within the first postpartum year: the risk profile of a community sample. Public Health Nurs (2007) 24(2):176–83.10.1111/j.1525-1446.2007.00622.x
238
EgeETimurSZincirHGeçkilESunar-ReederB. Social support and symptoms of postpartum depression among new mothers in Eastern Turkey. J Obstet Gynaecol Res (2008) 34(4):585–93.10.1111/j.1447-0756.2008.00718.x
239
EkukluGTokucBEskiocakMBerberogluUSaltikA. Prevalence of postpartum depression in Edirne, Turkey, and related factors. J Reprod Med (2004) 49(11):908–14.
240
GokerAYanikkeremEDemetMMDikayakSYildirimYKoyuncuFM. Postpartum depression: is mode of delivery a risk factor?ISRN Obstet Gynecol (2012) 2012:1–6.10.5402/2012/616759
241
GulserenLErolAGulserenSKueyLKilicBErgorG. From antepartum to postpartum: a prospective study on the prevalence of peripartum depression in a semiurban Turkish community. J Reprod Med (2006) 51(12):955–60.
242
InandiTElciOCOzturkAEgriMPolatASahinTK. Risk factors for depression in postnatal first year, in eastern Turkey. Int J Epidemiol (2002) 31(6):1201–7.10.1093/ije/31.6.1201
243
InandiTBugdayciRDundarPSumerHSasmazT. Risk factors for depression in the first postnatal year: a Turkish study. Soc Psychiatry Psychiatr Epidemiol (2005) 40(9):725–30.10.1007/s00127-005-0949-3
244
KirpinarİGözümSPasinlioğluT. Prospective study of postpartum depression in eastern Turkey prevalence, socio-demographic and obstetric correlates, prenatal anxiety and early awareness. J Clin Nurs (2010) 19(3–4):422–31.10.1111/j.1365-2702.2009.03046.x
245
OrhonFSUlukolBSoykanA. Postpartum mood disorders and maternal perceptions of infant patterns in well-child follow-up visits. Acta Paediatr (2007) 96(12):1777–83.10.1111/j.1651-2227.2007.00557.x
246
PoçanAGAkiÖEParlakgümüsAHGerekliogluÇDolgunAB. The incidence of and risk factors for postpartum depression at an urban maternity clinic in Turkey. Int J Psychiatry Med (2013) 46(2):179–94.10.2190/PM.46.2.e
247
TezelAGözümS. Comparison of effects of nursing care to problem solving training on levels of depressive symptoms in post-partum women. Patient Educ Couns (2006) 63(1–2):64–73.10.1016/j.pec.2005.08.011
248
YağmurYUlukocaN. Social support and postpartum depression in low-socioeconomic level postpartum women in Eastern Turkey. Int J Public Health (2010) 55(6):543–9.10.1007/s00038-010-0182-z
249
GhubashRAbou-SalehMT. Postpartum psychiatric illness in Arab culture: prevalence and psychosocial correlates. Br J Psychiatry (1997) 171:65–8.10.1192/bjp.171.1.65
250
GreenKBroomeHMirabellaJ. Postnatal depression among mothers in the United Arab Emirates: socio-cultural and physical factors. Psychol Health Med (2006) 11(4):425–31.10.1080/13548500600678164
251
HamdanATamimH. Psychosocial risk and protective factors for postpartum depression in the United Arab Emirates. Arch Womens Ment Health (2011) 14(2):125–33.10.1007/s00737-010-0189-8
252
BrughaTSWheatleySTaubNACulverwellAFriedmanTKirwanPet alPragmatic randomized trial of antenatal intervention to prevent post-natal depression by reducing psychosocial risk factors. Psychol Med (2000) 30(6):1273–81.10.1017/S0033291799002937
253
CooperPJMurrayLHooperRWestA. The development and validation of a predictive index for postpartum depression. Psychol Med (1996) 26(3):627–34.10.1017/S003329170003569
254
EdgeDBakerDRogersA. Perinatal depression among black Caribbean women. Health Soc Care Community (2004) 12(5):430–8.10.1111/j.1365-2524.2004.00513.x
255
EvansJHeronJFrancombHOkeSGoldingJ. Cohort study of depressed mood during pregnancy and after childbirth. BMJ (2001) 323(7307):257–60.10.1136/bmj.323.7307.257
256
HearnGIliffAJonesIKirbyAOrmistonPParrPet alPostnatal depression in the community. Br J Gen Pract (1998) 48(428):1064–6.
257
HeronJHaqueSOyebodeFCraddockNJonesI. A longitudinal study of hypomania and depression symptoms in pregnancy and the postpartum period. Bipolar Disord (2009) 11(4):410–7.10.1111/j.1399-5618.2009.00685.x
258
HoneyKLBennettPMorganM. Predicting postnatal depression. J Affect Disord (2003) 76(1):201–10.10.1016/S0165-0327(02)00085-X
259
MorrellCJSladePWarnerRPaleyGDixonSWaltersSJet alClinical effectiveness of health visitor training in psychologically informed approaches for depression in postnatal women: pragmatic cluster randomised trial in primary care. BMJ (2009) 15(338):276–80.10.1136/bmj.a3045
260
O’HigginsMRobertsIJGloverVTaylorA. Mother-child bonding at 1 year: associations with symptoms of postnatal depression and bonding in the first few weeks. Arch Womens Ment Health (2013) 16(5):381–9.10.1007/s00737-013-0354-y
261
RamchandaniPSteinAEvansJO’ConnorTG. Paternal depression in the postnatal period and child development: a prospective population study. Lancet (2005) 365(9478):2201–5.10.1016/S0140-6736(05)66778-5
262
SheltonNJHerrickKG. Comparison of scoring methods and thresholds of the General Health Questionnaire-12 with the Edinburgh Postnatal Depression Scale in English women. Public Health (2009) 123(12):789–93.10.1016/j.puhe.2009.09.012
263
ThompsonWMHarrisBLazarusJRichardsC. A comparison of the performance of rating scales used in the diagnosis of postnatal depression. Acta Psychiatr Scand (1998) 98(3):224–7.10.1111/j.1600-0447.1998.tb10071.x
264
WarnerRApplebyLWhittonAFaragherB. Demographic and obstetric risk factors for postnatal psychiatric morbidity. Br J Psychiatry (1996) 168(5):607–11.10.1192/bjp.168.5.607
265
AbbasiSChuangCHDagherRZhuJKjerulffK. Unintended pregnancy and postpartum depression among first-time mothers. J Womens Health (2013) 22(5):412–6.10.1089/jwh.2012.3926
266
BeckCTGableRK. Comparative analysis of the performance of the postpartum depression screening scale with two other depression instruments. Nurs Res (2001) 50(4):242–50.10.1097/00006199-200107000-00008
267
BirkelandRThompsonJKPharesV. Adolescent motherhood and postpartum depression. J Clin Child Adolesc Psychol (2005) 34(2):292–300.10.1207/s15374424jccp3402_8
268
CertainHEMuellerMJagodzinskiTFlemingM. Domestic abuse during the previous year in a sample of postpartum women. J Obstet Gynecol Neonatal Nurs (2008) 37(1):35–41.10.1111/j.1552-6909.2007.00200.x
269
ChaudronLHSzilagyiPGKitzmanHJWadkinsHMConwellY. Detection of postpartum depressive symptoms by screening at well-child visits. Pediatrics (2004) 113(3 Pt 1):551–8.10.1542/peds.113.3.551
270
DagherRKShenassaED. Prenatal health behaviors and postpartum depression: is there an association?Arch Womens Ment Health (2012) 15(1):31–7.10.1007/s00737-011-0252-0
271
DagherRKMcGovernPMAlexanderBHDowdBEUkestadLKMcCaffreyDJ. The psychosocial work environment and maternal postpartum depression. Int J Behav Med (2009) 16(4):339–46.10.1007/s12529-008-9014-4
272
DemissieZSiega-RizAMEvensonKRHerringAHDoleNGaynesBN. Physical activity during pregnancy and postpartum depressive symptoms. Midwifery (2013) 29(2):139–47.10.1016/j.midw.2011.12.006
273
Doering RunquistJJMorinKStetzerFC. Severe fatigue and depressive symptoms in lower-income urban postpartum women. West J Nurs Res (2009) 31(5):599–612.10.1177/0193945909333890
274
EisenachJCPanPHSmileyRLavand’hommePLandauRHouleTT. Severity of acute pain after childbirth, but not type of delivery, predicts persistent pain and postpartum depression. Pain (2008) 140(1):87–94.10.1016/j.pain.2008.07.011
275
GaffneyKFKitsantasPBritoASwamidossCS. Postpartum depression, infant feeding practices, and infant weight gain at six months of age. J Pediatr Health Care (2014) 28(1):43–50.10.1016/j.pedhc.2012.10.005
276
GeorgiopoulosAMBryanTLYawnBPHoustonMSRummansTATherneauTM. Population-based screening for postpartum depression. Obstet Gynecol (1999) 93(5 Pt 1):653–7.10.1016/S0029-7844(98)00543-2
277
GlynnLMSandmanCA. Evaluation of the association between placental corticotrophin-releasing hormone and postpartum depressive symptoms. Psychosom Med (2014) 76(5):355–62.10.1097/PSY.0000000000000066
278
Hahn-HolbrookJHaseltonMGSchetterCDGlynnLM. Does breastfeeding offer protection against maternal depressive symptomatology?Arch Womens Ment Health (2013) 16(5):411–22.10.1007/s00737-013-0348-9
279
HerringSJRich-EdwardsJWOkenERifas-ShimanSLKleinmanKPGillmanMW. Association of postpartum depression with weight retention 1 year after childbirth. Obesity (2008) 16(6):1296–301.10.1038/oby.2008.71
280
HorowitzJA. Community-based postpartum depression screening within the first month after delivery. Contemp Nurse (2006) 21(1):85–93.10.5172/conu.2006.21.1.85
281
HorowitzJAMurphyCAGregoryKEWojcikJ. A community-based screening initiative to identify mothers at risk for postpartum depression. J Obstet Gynecol Neonatal Nurs (2011) 40(1):52–61.10.1111/j.1552-6909.2010.01199.x
282
HowellEABalbierzAWangJParidesMZlotnickCLeventhalH. Reducing postpartum depressive symptoms among black and Latina mothers: a randomized controlled trial. Obstet Gynecol (2012) 119(5):942–9.10.1097/AOG.0b013e318250ba48
283
HowellEABodnar-DerenSBalbierzALoudonHMoraPAZlotnickCet alAn intervention to reduce postpartum depressive symptoms: a randomized controlled trial. Arch Womens Ment Health (2014) 17(1):57–63.10.1007/s00737-013-0381-8
284
HunkerDFPatrickTEAlbrechtSAWisnerKL. Is difficult childbirth related to postpartum maternal outcomes in the early postpartum period?Arch Womens Ment Health (2009) 12(4):211–9.10.1007/s00737-009-0068-3
285
KimHGGeppertJQuanTBrachaYLupoVCuttsDB. Screening for postpartum depression among low-income mothers using an interactive voice response system. Matern Child Health J (2012) 16(4):921–8.10.1007/s10995-011-0817-6
286
KuoSChenSTzengY. Depression and anxiety trajectories among women who undergo an elective cesarean section. PLoS One (2014) 9(1).10.1371/journal.pone.0086653
287
McGrathJMRecordsKRiceM. Maternal depression and infant temperament characteristics. Infant Behav Dev (2008) 31(1):71–80.10.1016/j.infbeh.2007.07.001
288
MercierRJGarrettJThorpJSiega-RizAM. Pregnancy intention and postpartum depression: secondary data analysis from a prospective cohort. BJOG (2013) 120(9):1116–22.10.1111/1471-0528.12255
289
Morris-RushJKFredaMCBernsteinPS. Screening for postpartum depression in an inner-city population. Am J Obstet Gynecol (2003) 188(5):1217–9.10.1067/mob.2003.279
290
MosackVShoreER. Screening for depression among pregnant and postpartum women. J Community Health Nurs (2006) 23(1):37–47.10.1207/s15327655jchn2301_4
291
MottSLSchillerCERichardsJGO’HaraMWStuartS. Depression and anxiety among postpartum and adoptive mothers. Arch Womens Ment Health (2011) 14(4):335–43.10.1007/s00737-011-0227-1
292
MurphyPKMuellerMHulseyTCEbelingMDWagnerCL. An exploratory study of postpartum depression and vitamin D. J Am Psychiatry (2010) 16:170–7.10.1177/1078390310370476
293
ParkEMMeltzer-BrodySStickgoldR. Poor sleep maintenance and subjective sleep quality are associated with postpartum maternal depression symptom severity. Arch Womens Ment Health (2013) 16(6):539–47.10.1007/s00737-013-0356-9
294
PaulIMDownsDSSchaeferEWBeilerJSWeismanCS. Postpartum anxiety and maternal-infant health outcomes. Pediatrics (2013) 131(4):e1218–24.10.1542/peds.2012-2147
295
ReighardFTEvansML. Use of the Edinburgh Postnatal Depression Scale in a southern, rural population in the United States. Prog Neuropsychopharmacol Biol Psychiatry (1995) 19(7):1219–24.10.1016/0278-5846(95)00238-3
296
Rich-EdwardsJWKleinmanKAbramsAHarlowBLMcLaughlinTJJoffeHet alSociodemographic predictors of antenatal and postpartum depressive symptoms among women in a medical group practice. J Epidemiol Community Health (2006) 60(3):221–7.10.1136/jech.2005.039370
297
RoyAGangPColeKRutskyMReeseLWeisbordJ. Use of Edinburgh Postnatal Depression Scale in a North American population. Prog Neuropsychopharmacol Biol Psychiatry (1993) 17(3):501–4.10.1016/0278-5846(93)90082-4
298
SchaperAMRooneyBLKayNRSilvaPD. Use of the Edinburgh Postnatal Depression Scale to identify postpartum depression in a clinical setting. J Reprod Med (1994) 39(8):620–4.
299
SilvermanMELoudonH. Antenatal reports of pre-pregnancy abuse is associated with symptoms of depression in the postpartum period. Arch Womens Ment Health (2010) 13(5):411–5.10.1007/s00737-010-0161-7
300
WatkinsSMeltzer-BrodySZolnounDStuebeA. Early breastfeeding experiences and postpartum depression. Obstet Gynecol (2011) 118(2 Pt 1):214–21.10.1097/AOG.0b013e3182260a2d
301
WisnerKLSitDKMcSheaMCRizzoDMZoretichRAHughesCLet alOnset timing, thoughts of self-harm, and diagnoses in postpartum women with screen-positive depression findings. JAMA Psychiatry (2013) 70(5):490–8.10.1001/jamapsychiatry.2013.87
302
YimISGlynnLMSchetterCDHobelCJChicz-DeMetASandmanCA. Risk of postpartum depressive symptoms with elevated corticotropin-releasing hormone in human pregnancy. Arch Gen Psychiatry (2009) 66(2):162–9.10.1001/archgenpsychiatry.2008.533
303
YonkersKARaminSMRushAJNavarreteCACarmodyTMarchDet alOnset and persistence of postpartum depression in an inner-city maternal health clinic system. Am J Psychiatry (2001) 158(11):1856–63.10.1176/appi.ajp.158.11.1856
304
ChibandaDMangeziWTshimangaMWoelkGRusakanikoPStranix-ChibandaLet alValidation of the Edinburgh Postnatal Depression Scale among women in a high HIV prevalence area in urban Zimbabwe. Arch Womens Ment Health (2010) 13(3):201–6.10.1007/s00737-009-0073-6
305
FisherJFeekeryCRoweH. Treatment of maternal mood disorder and infant behaviour disturbance in an Australian private mothercraft unit: a follow-up study. Arch Womens Ment Health (2004) 7(1):89–93.10.1007/s00737-003-0041-5
306
KubiszewskiICostanzaRFrancoCLawnPTalberthJJackstonTet alBeyond GDP: measuring and achieving global genuine progress. Ecol Econ (2013) 93:57–68.10.1016/j.ecolecon.2013.04.019
307
BarroRJ. Inequality and growth in a panel of countries. J Econ Growth (2000) 5(1):5–32.10.1023/A:1009850119329
308
CoburnD. Beyond the income inequality hypothesis: class, neo-liberalism, and health inequalities. Soc Sci Med (2004) 58(1):41–56.10.1016/S0277-9536(03)00159-X
309
JohnsNECowlingKGakidouE. The wealth (and health) of nations: a cross-country analysis of the relation between wealth and inequality in disease burden estimation. Lancet (2013) 381:S66.10.1016/S0140-6736(13)61320-3
310
HillEJ. Work-family facilitation and conflict, working fathers and mothers, work-family stressors and support. J Fam Issues (2005) 26(6):793–819.10.1177/0192513X05277542
311
BinaR. The impact of cultural factors upon postpartum depression: a literature review. Health Care Women Int (2008) 29(6):568–92.10.1080/07399330802089149
312
Hahn-HolbrookJHaseltonM. Is postpartum depression a disease of modern civilization?Curr Dir Psychol Sci (2014) 23(6):395–400.10.1177/0963721414547736
313
ZambranaREScrimshawSCollinsNDunkel-SchetterC. Prenatal health behaviors and psychosocial risk factors in pregnant women of Mexican origin: the role of acculturation. Am J Public Health (1997) 87(6):1022–6.10.2105/AJPH.87.6.1022
Appendix
Boolean Search Information
CINAHL
Boolean search that yields 142 records:
(IN edinburgh postnatal depression scale) AND (AB (postpartum depression OR postnatal depression)) AND (AB (incidence OR prevalence))
With the additional limiters:
- (1)
Narrow to 1985–2015
- (2)
Narrow to English articles only
- (3)
Narrow to Humans only
- (4)
Narrow to Females only
PsychInfo
Boolean search that yields 236 records:
AB (incidence or prevalence) AND AB (postnatal depression or postpartum depression) AND TM edinburgh postnatal depression scale
With the additional limiters:
- (1)
Narrow to 1985–2015
- (2)
Narrow to English articles only
- (3)
Narrow to females only
PubMed
Boolean search yields 338 records:
((“postpartum depression”[All Fields] OR “postnatal depression”[All Fields]) AND “prevalence”[All Fields]) AND “edinburgh postnatal depression scale”[All Fields] AND ((“1985/01/01”[PDAT]:?“2015/12/31”[PDAT]) AND “humans”[MeSH Terms])
Summary
Keywords
depression, prevalence, gini index, postpartum, gross domestic product, pregnancy, infant mortality
Citation
Hahn-Holbrook J, Cornwell-Hinrichs T and Anaya I (2018) Economic and Health Predictors of National Postpartum Depression Prevalence: A Systematic Review, Meta-analysis, and Meta-Regression of 291 Studies from 56 Countries. Front. Psychiatry 8:248. doi: 10.3389/fpsyt.2017.00248
Received
08 August 2017
Accepted
07 November 2017
Published
01 February 2018
Volume
8 - 2017
Edited by
Gianluca Serafini, University of Genoa, Italy
Reviewed by
Erika Comasco, Uppsala University, Sweden; Taolin Chen, West China Hospital of Sichuan University, China
Updates
Copyright
© 2018 Hahn-Holbrook, Cornwell-Hinrichs and Anaya.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jennifer Hahn-Holbrook, jhahn-holbrook@ucmerced.edu
Specialty section: This article was submitted to Mood and Anxiety Disorders, a section of the journal Frontiers in Psychiatry
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.