- 1Department of Psychology, University of Roehampton, London, UK
- 2CIBER en Salud Mental (CIBERSAM), Barcelona, Spain
- 3Parc Sanitari Sant Joan de Déu, Sant Boi de Llobregat, Barcelona, Spain
- 4Opole University, Opole, Poland
The aim of the present study was to examine the factor structure and psychometric properties of the short version of the Children’s Somatization Inventory (CSI-24) in Poland. The CSI-24 is a self-report questionnaire designed to assess somatic symptoms in children and adolescents. A total of 733 children and adolescents, aged 12–17 years, participated in this research. The participants for this study were recruited from urban and suburban schools of Opole province in South Western Poland. In addition to the CSI-24, all participants completed the Spence Children’s Anxiety Scale (SCAS) and the Strength and Difficulties Questionnaire (SDQ). The correlated four-factor model that included four-correlated dimensions (pain/weakness, gastrointestinal problems, cardiovascular symptoms, and pseudoneurological problems) showed a better fit compared to the single-factor model. The Cronbach’s Alpha for the CSI-24 was 0.91. Somatic symptoms correlated significantly highly with the SCAS total scores and the SDQ emotional subscale, suggesting good construct validity. Somatic symptoms had low correlation with the SDQ behavioral problems symptoms, suggesting adequate discriminant validity. The CSI-24 reliably measured somatic symptoms in children and adolescents in Poland.
Introduction
Somatic symptoms such as stomach aches and headaches occur frequently in children and adolescents in the general population (1, 2). Somatic symptoms are more prevalent in girls than in boys, and they tend to increase with age (3). Youths with somatic symptoms tend to experience impairment in academic and social functioning including a high level of absenteeism, poor academic achievement, and psychosocial difficulties (4). Furthermore, somatic symptoms are linked to an increase in health care services utilization, thus, they tend to exert a heavy financial burden on both the families and the health care system (4).
Somatic symptoms co-occur frequently with anxiety and depression (5) and that they tend to be stable over time (6). Chronic somatic symptoms in childhood significantly predicts psychopathology in adulthood (7).These observations highlight the importance of understanding and screening somatic symptoms in children and adolescents.
The Children’s Somatization Inventory [CSI; (8)] is a self-report questionnaire for assessing somatic symptoms in children and adolescents. Its original version consists of 35 items (referred to as CSI-35) from which items were taken from the symptoms of somatization disorder (9), the somatization factor of the Hopkins Symptom Checklist (10), and common symptoms of functional gastrointestinal (GI) disorder (i.e., constipation). Children and adolescents report the extent to which they experience each symptom in the past 2 weeks on a 5-point scale ranging from “not at all” (0) to “a whole lot” (4). Somatization can be defined as a psychological distress that is manifested in the form of somatic symptoms.
Studies that examined the factor structure of the CSI-35 have reported considerable inconsistency regarding the factor structure of this instrument. For example, studies conducted in the US (11) and in the Ukraine (12) reported a four-factor structure on the CSI: pseudoneurological, cardiovascular, GI, and pain/weakness symptoms. However, in a study in the Netherlands (13) and the UK (3), only three factors were found: pain/weakness, GI, and pseudoneurological. In all of these studies, some of the CSI items are rarely endorsed and the item-total correlations were low. This is not surprising because some of the items were taken from the symptoms criteria for somatization disorders in adults which may not be applicable to children (14).
In a recent study by Walker and colleagues (14) that examined the dimensionality of the CSI-35, 11 statistically weak items (lump in throat, deafness, double vision, blindness, fainting/passing out, memory loss/amnesia, seizures, convulsion, trouble walking, paralysis/muscle weakness, difficulty urinating, pain-urinating) were deleted, leaving 24 items (referred to as CSI-24). The authors conducted a principal component analysis and found a single large component which explained almost 30% of the total variance and containing symptoms of various organs; a second factor containing symptoms related to GI symptoms was also found, but it was regarded as weak. The authors also conducted a confirmatory factor analysis (CFA) and found that the CSI-24 did not fit a single-factor model. Because of its better psychometric properties, shorter time completion and containing more appropriate items for children and adolescents, the authors suggested that the CSI-24 is preferable to the CSI-35.
In view of the above findings, it remains unclear whether a general somatization factor exists or whether somatic symptoms would be better represented by multidimensionality. Therefore, the main aim of this study was to test the factor structure of the Polish translation of the CSI-24: one-factor model (14), three-factor model (3, 13), four-factor model (11, 12), and two models including a general factor and specific group factors (i.e., bifactor models). A bifactor model would include a general factor that accounts for relationships between items, but also specific factors that account for the unique variance among the items above and beyond the general factor (15). Other aims of this study were to examine the relationship of somatic symptoms with other psychopathology, and to examine gender and age patterns of somatic symptoms.
Materials and Methods
Participants
The participants for this study were recruited from five urban and suburban schools of the Opole province in South Western Poland. A total of 733 adolescents participated in the study and of these 51.1% were girls. They ranged in age from 12 to 17 years (mean = 15.4, SD = 1.59). About 89.5% of the participants indicated Christianity as their religion.
Consent from the parent was obtained before the youths could participate in the study. Each participating school was sent a letter that described the aim of the present study and the procedure involved. The youths were informed about the aim of the study as well as about the voluntary nature and anonymity of their participation in this study. The questionnaires were completed by the participants in groups in the designated classrooms. A research assistant was available to provide answers if necessary and to ensure independent responding. Approval to conduct this study was obtained from the Psychology Ethic Committee at the University of Roehampton.
Measures
In addition to the CSI-24, the participants also completed a brief questionnaire to obtain demographic characteristics, the Strengths and Difficulties Questionnaire (SDQ) (16), and the Spence Children’s Anxiety Scale (SCAS) (17). The English version of these questionnaires was adapted and translated to Polish according to guidelines that are widely accepted for the successful translation of instruments in cross-cultural research (18).
The Strengths and Difficulties Questionnaire (16) was used to assess general difficulties and positive attributes. Its 25 items are divided into 5 scales, which generate scores for conduct problems, hyperactivity-inattention, emotional symptoms, peer problems, and pro-social behavior; the “emotional symptoms” subscale assesses anxiety and depressive symptoms. Each of the items is rated on a 3-point scale, ranging from “not true” (0) to “certainly true” (2). In the present study, the Cronbach’s Alpha for the total SDQ scores was 0.79.
The Spence Children’s Anxiety Scale (17) is a 38-item measure of anxiety symptoms in children and adolescents. The items reflect symptoms of the main DSM-IV anxiety disorders, including separation anxiety, social phobia, obsessive-compulsive disorder (OCD), panic/agoraphobia, physical injury fears, and generalized anxiety disorder (GAD). Each item is rated on a 4-point scale in terms of its frequency from “never” (0) to “always” (3). In the present study, the Cronbach’s Alpha for the SCAS was 0.93.
Statistical Analysis
The Statistical Package for Social Science (SPSS) version 19.0 and the Structural Equation Modeling package EQS version 6.1 were used to perform the statistical analyses. The CSI-24 data were 99.2% complete, with 96% of cases having no missing items. With less than the 5% of cases having any missing data, any reasonable method of dealing with missing values could be used (19). Missing values were replaced using expectation maximization (EM) algorithm (20) to fulfill the 4% of missing items based on non-missing responses (21). This imputation method is thought to be better than the mean imputation since it preserves the variance. The average item mean and standard deviation were the same both before and after imputation (M = 0.62, SD = 0.93).
Corrected item-total correlations were calculated to examine how each item contributed to the overall scale. The Cronbach’s alpha was calculated to determine the internal consistency of the CSI-24.
Confirmatory factor analysis was performed to compare the one, three, and four-factor structure of the CSI (Figures 1A–C). The last two factors were considered to be factor-correlated. The Lagrange Multiplier tests (LM test) were also conducted to determine unspecified parameters in the model. Statistically significant LM Chi-square values would argue for the presence of factor cross-loadings and error covariances (22). Two bifactor models were specified in which each item loaded onto a general somatization factor, and items also loaded onto one of the three or the four domain-specific factors (Figures 1D,E).
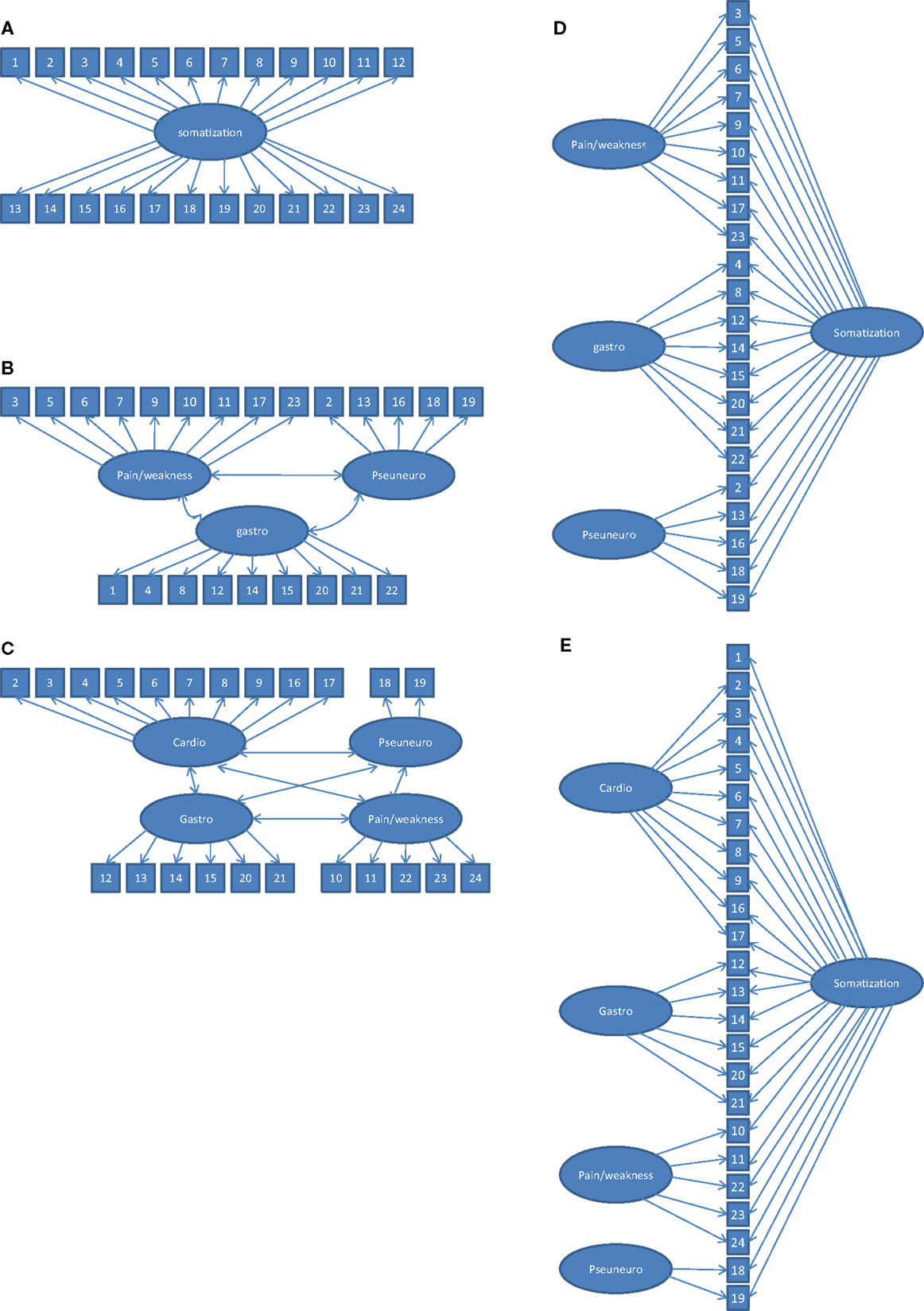
Figure 1. Five factor models tested with CFA. (A) unidimensional model (14). (B) three-correlated factor model (3, 13). Item 16 (“heart beating”) was considered as part of the pseudoneurological symptoms (3). (C) four- correlated factor model (11, 12). Item 1 (“headaches”) was not included as was proposed by the authors. (D) bifactor model with one general factor (somatization) and three specific factors (pain/weakness, gastrointestinal problems, and pseudoneurological problems). (E) bifactor model with one general factor (somatization) and four factors (Pain/weakness, cardiovascular problems, gastrointestinal problems, and pseudoneurological problems). This model included item 1 (“headaches”) as being part of the general factor.
Since the observed variables included in the model were ordinal and presented a certain level of skewness, a maximum likelihood robust method based on polychoric correlations was used (23). Satorra–Bentler scaled Chi-square, the Normed Fit Index (NFI), the Non-Normed Fit Index (NNFI), the Comparative Fit Index (CFI), the Root Mean Square Error of Approximation (RMSEA), the Standardized Root Mean Square Residual (SRMR) were used as goodness-of-fit indices. Values of NFI, NNFI, and CFI higher than 0.90 indicate adequate fit (24). RMSEA of 0.05 or lower is indicative of an adequate fit (25); a favorable value of the SRMR is less than 0.10 (26). Robust versions of all fit statistics were computed except for the SRMR, which has no robust counterpart but summarizes the fit in a way the other (robust) indices do not. The Akaike’s Information Criterion (AIC), the consistent AIC (CAIC), and the Expected Cross-Validation Index (ECVI) were used as parsimony indices: smaller model AIC, CAIC, and ECVI than the comparison model indicate better fit.
Non-parametric correlation tests (Spearman rank) were computed between the CSI and the SCAS and the SDQ to assess the convergent and divergent validity of the CSI. Gender and age effects were examined with Mann–Whitney and Kruskal–Wallis tests.
Results
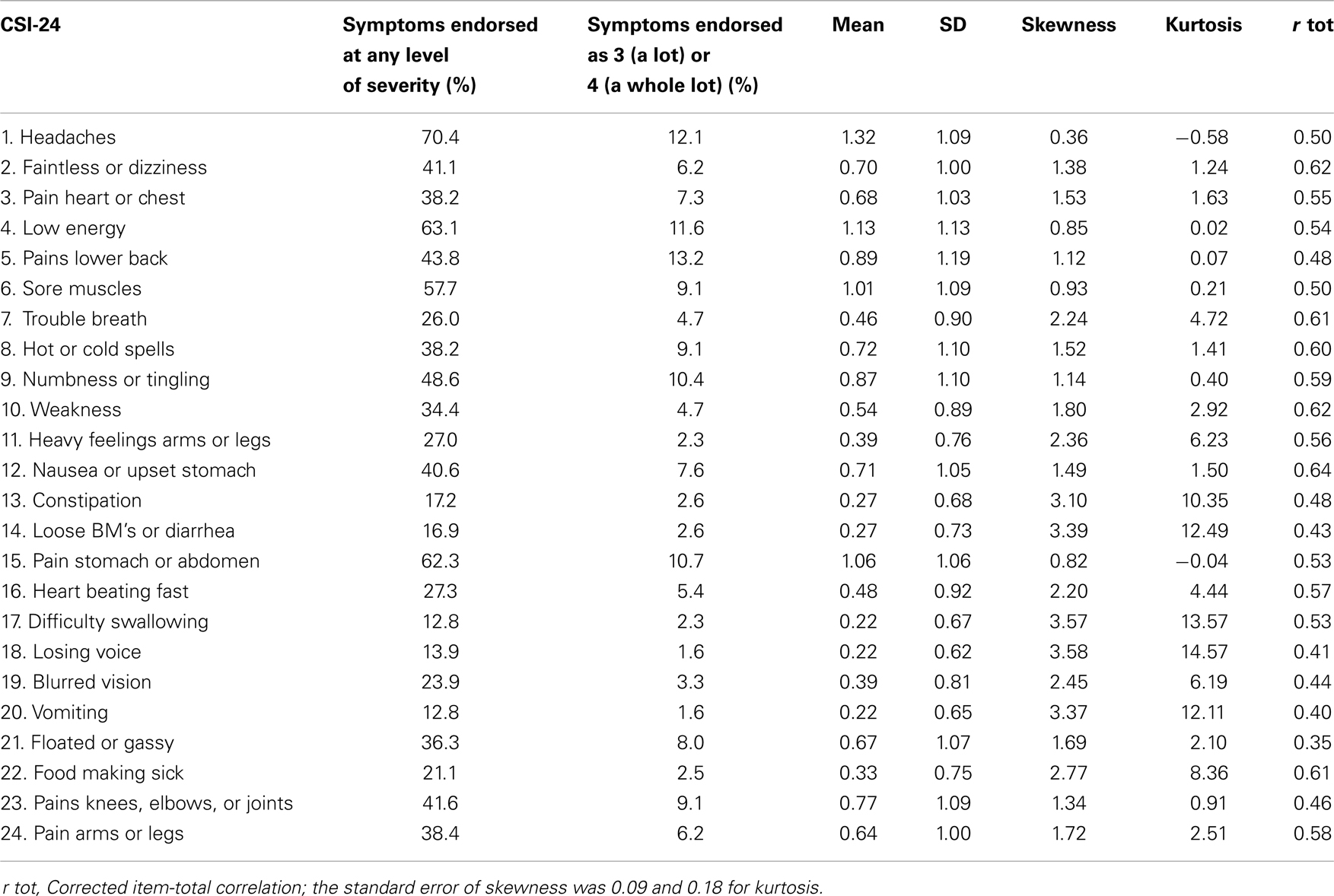
Descriptive analyses of the CSI-24 items (Table 1) showed the lowest mean for items “difficulty swallowing,” “losing voice,” and “vomiting.” The highest mean was obtained for “headaches,” followed by “low energy,” “pain in stomach or abdomen.” There was a marked skewness or kurtosis for the following items: “constipation,” “loose BM’s or diarrhea,” “difficulty swallowing,” “losing voice,” “vomiting,” and “food making sick.” All items obtained a corrected item-total correlation greater than the rule of thumb minimum value of 0.20 (27).
Confirmatory Factor Analyses
Lagrange multiplier test in the one-factor model showed that the highest LM Test incremental multivariate Chi-square values represent error covariances between items 13 (“constipation”) and 14 (“Loose BM’s or diarrhea”), items 14 (“diarrhea”) and 22 (“Food intolerance”), items 13 (“constipation”) and 21 (“floated or gassy”), items 14 (“diarrhea”) and 21 (“floated or gassy”), and between item 23 (“pain joints”) and item 24 (“pain in arms and legs”). To understand these results, it is important to look at the content of these items. It seems that there might be an overlap between them (i.e., pain in joints and pain in arms or legs), and in other cases, items refer to symptoms that occur generally at the same time (i.e., constipation and being gassy). Therefore, the one-factor model was re-specified including these parameters.
For the three-factor model, the LM-test showed the following misspecified parameters (error covariances): items 13 (“constipation”) and 14 (“diarrhea”), items 14 (“diarrhea”) and 21 (“floated or gassy”), items 13 (“constipation”) and 21 (“floated or gassy”), and items 23 (“pain joints”) and 24 (“pain in arms and legs”). The LM-test in the four-factor model included as misspecified parameters the following error covariances: 23 (“Pain in knees, elbows, or joints”) and 24 (“Pain in arms and legs”), items 13 (“constipation”) and 14 (“Loose BM’s or diarrhea”), items 14 (“diarrhea”) and 21 (“floated or gassy”), and items 13 (“constipation”) and 21 (“floated or gassy”). Moreover, the LM-test showed as misspecified a cross-loading from factor 4 (pseudoneurological) to item 17 (“difficulty swallowing”), and from factor 2 (GI problems) to item 22 (“Food intolerance”). After reviewing the content of each item, it was considered that these error covariances and cross-loadings should be kept in both the three- and four-factor models including these new parameters. The goodness-of-fit indices of the new re-specified models were better than the models without these items, and the different values of the Satorra–Bentler Chi-Squared [calculated with the computation process from (28)] were also significant (p < 0.001).
Table 2 includes the goodness-of-fit indices with these new parameters in the three models. Overall, the goodness-of-fit indices were good in all models under test. However, the model which yielded better fit indices as well as better parsimonious indices was the four-factor model. Bifactor model including the three domain-specific factors (i.e., GI, pseudoneurological, and pain/weakness) showed better indices than the three-factor model, whereas goodness-of-fit of the bifactor model with the four domain-specific factors was in general poorer than the four-correlated factors.
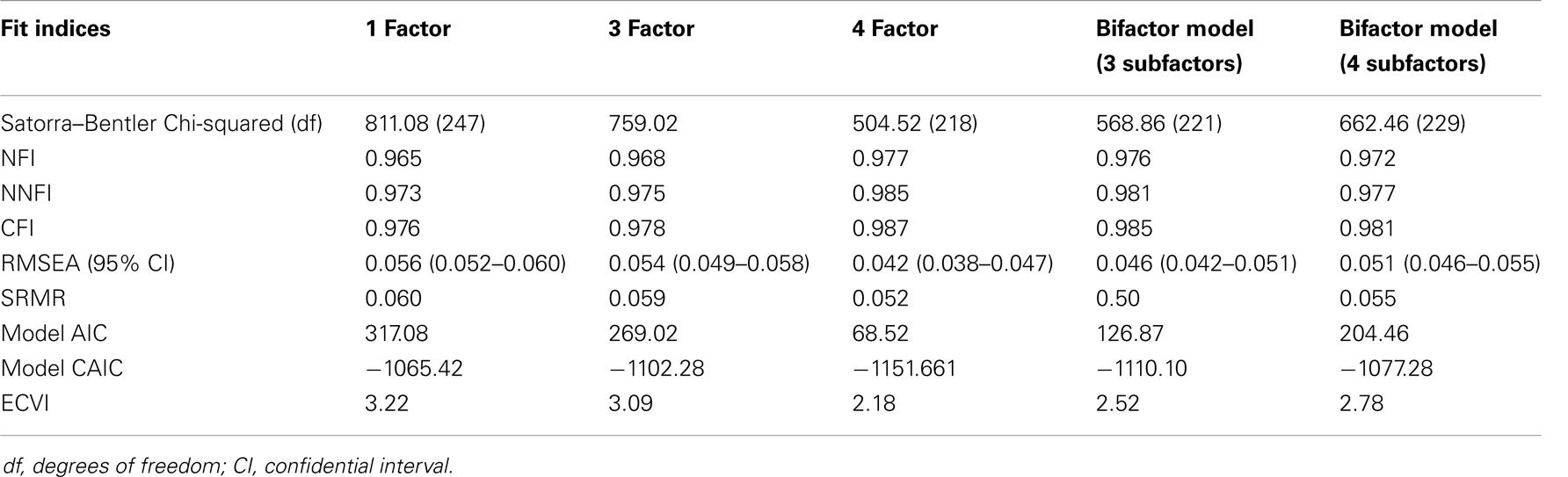
Table 2. Goodness-of-fit indices for the one-, three-, four-factors, and bifactor models of the CSI-24.
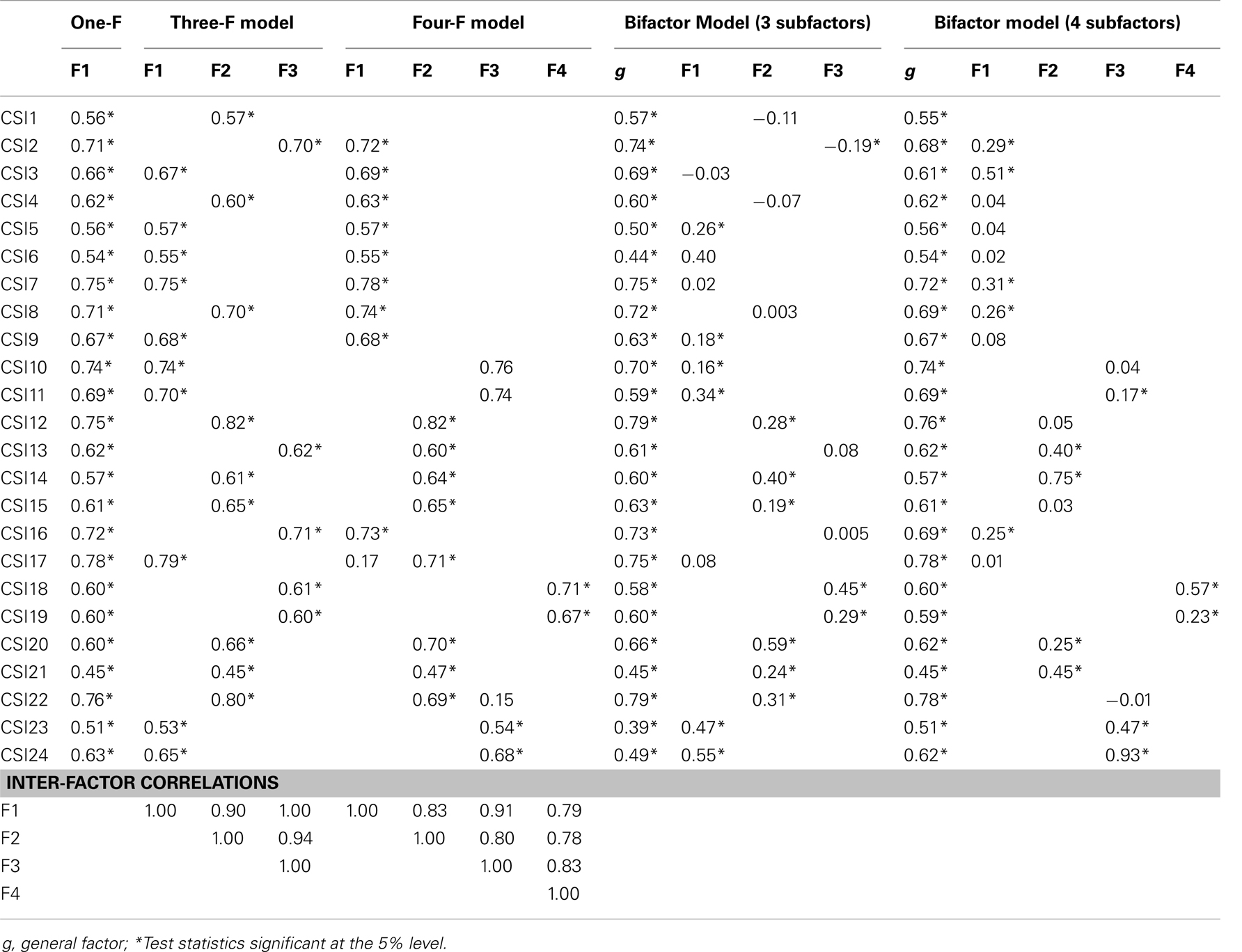
Factors loadings in the one-factor model ranged from 0.45 (item 21) to 0.78 (item 17) (Table 3). In the three-factor and four-factor models, items showed high loadings on their respective hypothesized factors. In the four-factor model, however, items 17 (“difficulty swallowing”) and 22 (“food intolerance”) had low values in their cross-factor loading (0.17 and 0.15, respectively), which were marginally significant. Factor loadings to the general factor in the bifactor models tend to go down slightly compared with the loadings in the one-factor solution. This might occur because the factor in the unidimensional model is likely a conglomeration of the common variance present through all items due to shared content (29). In the bifactor model with three specific group factors, 15 out of 24 items had significant factor loadings on the general factor as well as on the specific factors. Particular symptoms did not share symptom specific variances and only loaded significantly on the general factor (e.g., “trouble breathing” and “heart beating fast”). Similarly, in the bifactor model with four specific group factors, 14 out of 24 items showed significant factor loadings on both the general factor and the specific group factor. Again, some items did not significantly load onto the specific factor (e.g., “sore muscles” or “difficulty swallowing”). Inter-factor correlations were very high, both in the three-factor and the four-factor model.
Internal Consistency and Validity
The Cronbach’s alpha of the CSI-24 was 0.91. Internal consistency of the four-factor model was good in three of its subscales. Cronbach’s alpha coefficient of the GI factor was 0.72, for the cardiovascular problems was 0.84, and for the pain/weakness was 0.76. The internal consistency of the pseudoneurological factor was poor, with a Cronbach’s alpha of 0.48.
The CSI-24 total scores correlated highly with the SCAS total scores and all its subscales, as well as with the SDQ total scores and all its subscales except for the pro-social behavior subscale (Table 4). Correlations between the CSI-24 and the SDQ conduct problems and ADHD subscales were high (both equal to 0.40). In order to determine whether correlations between the CSI-24 and the SCAS and the SDQ emotional problems were significantly stronger (convergent validity) than correlations between the CSI-24 and the SDQ conduct problems and ADHD subscales (divergent validity), we used the Steiger’s Z test (30), following the recommendation of Meng et al. (31) for correlated correlations. Results showed that the relationship between the CSI-24 and the SCAS OCD, panic, GAD, SCAS total score, and SDQ emotional problems were significantly greater than the correlations between the CSI-24 and the SDQ conduct problems and the correlations between the CSI-24 and the SDQ hyperactivity (z > 1.96, p < 0.05). However, results showed that correlations between the CSI-24 and the SAD, Social phobia, and fears subscales were not significantly different from correlations between the CSI-24 and both the SDQ conduct problems and hyperactivity subscales (z < 1.96, p > 0.5).
Gender and Age Effects
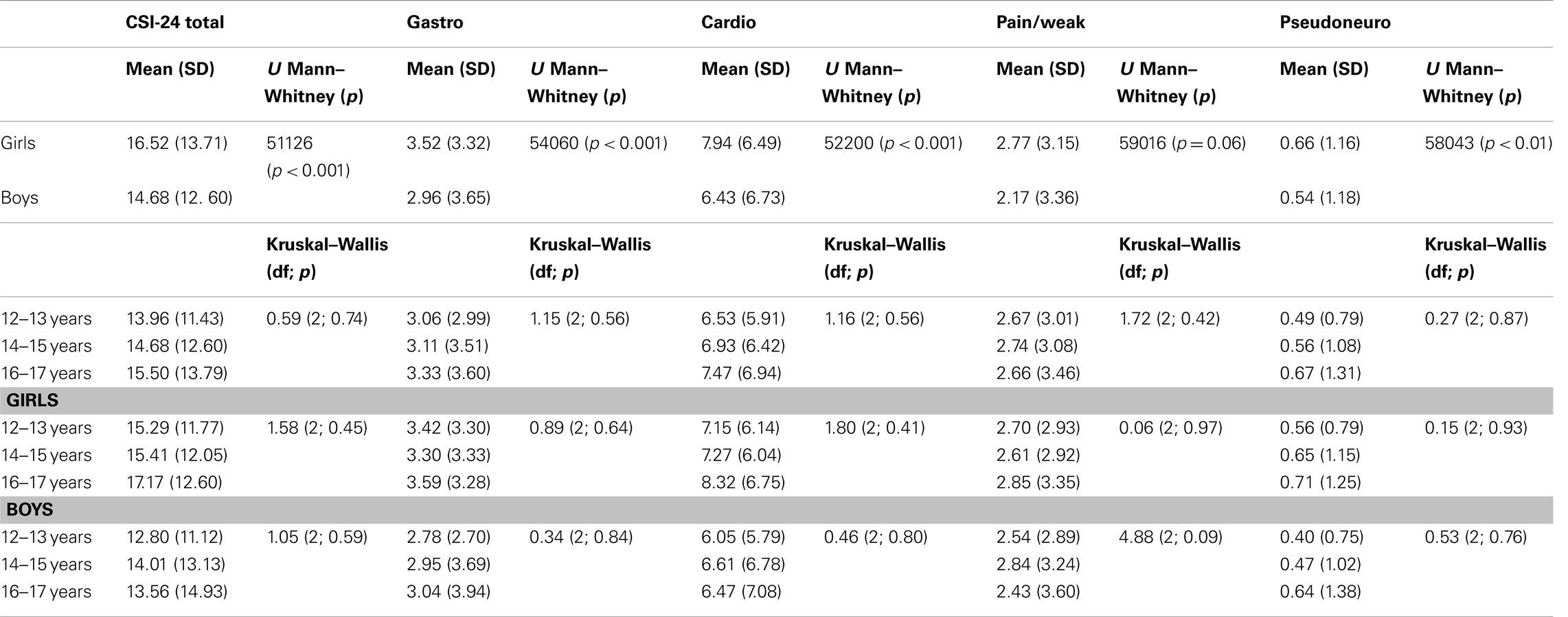
The mean of the CSI-24 total score on the whole sample was 14.95 (SD = 12.99). We also calculated the means and standard errors of the four factors: GI factor (mean = 3.21, SD = 3.47); cardiovascular (mean = 7.14, SD = 6.60), pain/weakness (mean = 2.67, SD = 3.24), and pseudoneurological factor (mean = 0.60, SD = 1.16). Girls had significantly higher mean scores than boys on the CSI-24 total score, GI, cardiovascular, and pseudoneurological symptoms (Table 5). Comparisons between three groups (12–13, 14–15, 16–17 years) in terms of the mean scores on the CSI-24 and the four subscales yielded no significant differences.
Due to the violation of the normality assumption, Age × Gender interaction was not examined. However, we examined the mean scores of the CSI-24 among boys and girls separately within the three age groups. There were no significant differences in the different CSI scores between age groups according to the participants’ gender (Table 5).
Discussion
The main purpose of this article was to examine the factor structure, reliability, and the validity of the Polish version of the CIS-24. Another aim was to examine the effects of gender and age on the frequency of somatic symptoms. Our main findings are summarized as follows: first, the better fit of the correlated four-factor model compared to the single-factor model supports the multicomponential nature of the CSI-24 with four-correlated dimensions: pain/weakness, GI problems, cardiovascular symptoms, and pseudoneurological problems. This factor structure corresponded to the factor structure reported by Garber et al. (11) and Litcher et al. (12) when the original CIS-35 was used with children and adolescents in the US and Ukraine. However, our findings were inconsistent with those by Walker et al. (14) in which one dominant general factor was found when a PCA was conducted. However, the bifactor models under test yielded good fit indices and the inspection of the factor loadings showed that many symptoms significantly loaded onto both the general factor and the specific factors. This would suggest that the variability of a CSI-24 symptom is determined by two sources of systematic variance, one from a general somatization factor and the other from a specific factor. Some authors have suggested that this could be explained in the light of the presence of an affective component associated to the symptom experience as the general factor, whereas the specific factors would correspond to the sensory part of the symptom experience (32). Another point of concern is the high inter-factor correlations between the three and the four factors, which could be suggestive of the presence of a second-order factor. In such a model, correlations between factors would be accounted for by the general somatization factor. The authors conducted a CFA with second-order factor models (with both the three and the four factors), although results did not convergent to admissible solution. If this model provides better fit to the data, the domain would consist of a single broad somatization construct that can be broken down into specific manifestations or facets. Thus, it is possible to calculate a total score of somatic symptoms by adding up all the items as well as to use different dimensions that might better reflect the complexity of the somatization construct in children and adolescents.
Second, the Cronbach’s alpha of the CSI-24 was 0.91. Studies conducted in the US and Turkey (14, 33) have similarly shown high internal consistency for the CSI-24, with Cronbach’s Alphas ranging from 0.90 to 0.91. Third, similar to previous studies (3), girls reported significantly higher number of somatic symptoms than boys. The reason for this gender difference is unclear although biological factors related to puberty (e.g., menstruation) have been considered as contributing to higher somatic symptoms in girls (34). The role of age in somatic symptoms is less clear. In the study by Vila et al. (3) in the UK, the 11- to 12-year-olds had significantly higher scores than the 13- to 14-year-olds. It was argued that the 11- to 12-year-olds were more stressed than the 13- to 14-year-olds as they needed to adapt to a new school.
Fourth, in agreement with several studies (3), the Polish CSI-24 has a good convergent validity. Specifically, the CSI-24 total score correlated significantly with the SCAS, and the SDQ, suggesting that a high level of somatic symptoms was associated with a high level of anxiety and emotional problems. The CSI-24 showed good construct validity as evidenced by the significant positive correlations with the anxiety symptoms as measured by the SCAS and on the emotional subscale of the SDQ. These findings are consistent with previous studies that showed significant correlation between somatic and two internalizing problems [i.e., anxiety, and emotional symptoms (35)]. In agreement with previous studies (3), somatic symptoms had low correlation with behavioral problems (which is an example of an externalizing problem), suggesting adequate discriminant validity. These findings seem to suggest that the CSI-24 can be used to distinguish internalizing from externalizing problems. Internalizing problems are inner-directed which cause distress to oneself, while as externalizing problems are outer-directed which cause distress to others (36).
There are several limitations to the present study, which need to be taken into consideration when interpreting our results. First, our participants were 12- to 17-year-olds who were recruited from school settings. The use of a community sample may have implications for the generalizability of our findings to other populations such as those in clinical setting or younger age groups. Second, the data were based on self-report; however children and adolescents have been reported to be better informants than parents because of the internalizing experiences of some somatic symptoms. Indeed studies have reported disagreement between children and their parents when reporting somatic symptoms (37), with mothers reporting more somatic symptoms than the children. Third, because the CSI-24 does not provide information on possible medical explanations for symptoms, it is unclear whether or not the symptoms reported on the CSI-24 are an expression of organic problems. These limitations notwithstanding, our findings support the usefulness of the CSI-24 as an efficient way of screening for somatic symptoms in children and adolescents in Poland.
Future studies should replicate the present study in other settings (e.g., clinical setting) and age groups.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Ghandour RM, Overpeck MD, Huang ZJ, Kogan MD, Scheidt PC. Headache, stomachache, backache, and morning fatigue among adolescent girls in the United States: associations with behavioral, sociodemographic, and environmental factors. Arch Pediatr Adolesc Med (2004) 158:797–803. doi:10.1001/archpedi.158.8.797
2. Watson KD, Papageorgiou AC, Jones GT, Taylor S, Symmons DPM, Silman AJ, et al. Low back pain in school children: occurrence and characteristics. Pain (2002) 97:87–92. doi:10.1016/S0304-3959(02)00008-8
3. Vila M, Kramer T, Hickey N, Dattani M, Jefferis H, Singh M, et al. Assessment of somatic symptoms in British secondary school children using the Children’s Somatization Inventory (CSI). J Pediatr Psychol (2009) 34:989–98. doi:10.1093/jpepsy/jsp005
4. Walker LS, Williams SE, Smith CA, Garber J, Van Slyke DA, Lipani TA. Parent attention versus distraction: impact on symptom complaints by children with and without chronic functional abdominal pain. Pain (2006) 122:43–52. doi:10.1016/j.pain.2005.12.020
5. Vaccarino AL, Evans KR, Sills TL, Kalali AH. Symptoms of anxiety in depression: assessment of item performance of the Hamilton Anxiety Rating Scale in patients with depression. Depress Anxiety (2008) 25:1006–13. doi:10.1002/da.20435
6. Walker LS, Garber J, Van Slyke DA, Greene JW. Long-term health outcomes in patients with recurrent abdominal pain. J Pediatr Psychol (1995) 20:233–45. doi:10.1093/jpepsy/20.2.233
7. Gureje O, Simon GE, Von Korff M. A cross-national study of the course of persistent pain in primary care. Pain (2001) 92:195–200. doi:10.1016/S0304-3959(00)00483-8
8. Walker LS, Garber J, Green JW. Somatization symptoms in pediatric abdominal pain patients: relation to chronicity of abdominal pain and parent somatization. J Abnorm Child Psychol (1991) 19:379–94. doi:10.1007/BF00919084
9. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd ed. Washington, DC: American Psychiatric Association (1987).
10. Derogatis LR, Lipman RS, Rickels K, Uhlenhuth EH, Covi L. The Hopkins Symptom Checklist (HSCL): a self-report symptom inventory. Behav Sci (1974) 19:1–15. doi:10.1002/bs.3830190102
11. Garber J, Walker LS, Zeman J. Somatization symptoms in a community sample of children and adolescents: further validation of the Children’s Somatization Inventory. Psychol Assess (1991) 3:588–95. doi:10.1037/1040-3590.3.4.588
12. Litcher L, Bromet E, Carlson G, Gilbert T, Panina N, Golovakha E, et al. Ukrainian application of the Children’s Somatization Inventory: psychometric properties and associations with internalizing symptoms. J Abnorm Child Psychol (2001) 29:165–75. doi:10.1023/A:1005240214564
13. Meesters C, Muris P, Ghys A, Reumerman T, Rooijmans M. The Children’s Somatization Inventory: further evidence for its reliability and validity in a pediatric and community sample of Dutch children and adolescents. J Pediatr Psychol (2003) 28:413–22. doi:10.1093/jpepsy/jsg031
14. Walker LS, Beck JE, Garber J, Lambert W. Children’s Somatization Inventory: psychometric properties of the revised form (CSI-24). J Paediatr Psychol (2009) 34:430–40. doi:10.1093/jpepsy/jsn093
15. Chen FF, West SG, Sousa KH. A comparison of bifactor and second-order models of quality of life. Multivariate Behav Res (2006) 41:189–225. doi:10.1207/s15327906mbr4102_5
16. Goodman R. The strengths and difficulties questionnaire: a research note. J Child Psychol Psychiatry (1997) 38:581–6. doi:10.1111/j.1469-7610.1997.tb01545.x
17. Spence SH. The structure of anxiety symptoms among children: a confirmatory factor analytic study. J Abnorm Psychol (1997) 106:280–97. doi:10.1037/0021-843X.106.2.280
18. Brislin RW. Back translation for cross-cultural research. J Cross Cult Psychol (1970) 1:185–216. doi:10.1177/135910457000100301
19. Harrell FE. Regression Modeling Strategies with Applications to Linear Models, Logistic Regression, and Survival Analysis. New York: Springer (2001).
21. Little RJA, Rubin DB. Statistical Analysis with Missing Data. New York: John Wiley & Sons (1987).
22. Byrne BM. Structural Equation Modeling with EQS: Basic Concepts, Applications and Programming. 2nd ed. Mahwah, NJ: Erlbaum (2006).
23. Satorra A, Bentler PM. Corrections to test statistics and standard errors in covariance structure analysis. In: von Eye A, Clogg CC, editors. Latent Variables Analysis: Applications for Developmental Research. Thousand Oaks, CA: Sage (1994). p. 399–419.
24. Marsh HW, Hau KT. Assessing goodness of fit: is parsimony always desirable? J Exp Educ (1996) 64:364–90.
25. Browne MW, Cudeck R. Alternative ways of assessing model fit. In: Bollen KA, Long JS, editors. Testing Structural Equation Models. Newbury Park, CA: Sage (1993). p. 136–62.
26. Hu LT, Bentler PM. Cutoff criteria for fit indexes in covariance structure analysis: conventional criteria versus new alternatives. Struct Equ Modeling (1999) 6:1–55. doi:10.1080/10705519909540118
28. Satorra A, Bentler PM. A scaled difference chi-square test statistic for moment structure analysis. Psychometrika (2001) 66:507–14. doi:10.1007/BF02296192
29. Reise SP, Morizot J, Hays R. The role of the bifactor model in resolving dimensionality issues in health outcomes measures. Qual Life Res (2007) 16:19–31. doi:10.1007/s11136-007-9183-7
30. Steiger JH. Test for comparing elements of correlations matrix. Psychol Bull (1980) 87:245–81. doi:10.1037/0033-2909.87.2.245
31. Meng XL, Rosenthal R, Rubin DB. Comparing correlated correlation coefficients. Psychol Bull (1992) 111:172–5. doi:10.1037/0033-2909.111.1.172
32. Witthöft M, Basfeld C, Steinhoff M, Gerlach AL. Can’t suppress this feeling: automatic negative evaluations of somatosensory stimuli are related to the experience of somatic symptom distress. Emotion (2012) 12:640–9. doi:10.1037/a0024924
33. Kadioglu H, Sisman FN, Ergun A. Reliability and validity of the Turkish Version of Children’s Somatization Inventory. Asian Nurs Res (2012) 6:9–12. doi:10.1016/j.anr.2012.02.004
34. Eminson M, Benjamin S, Shortall A, Woods T. Physical symptoms and illness attitudes in adolescents: an epidemiological study. J Child Psychol Psychiatry (1996) 37:519–28. doi:10.1111/j.1469-7610.1996.tb01438.x
35. Essau CA, Conradt J, Petermann F. Frequency, comorbidity, and psychosocial impairment of anxiety disorders in adolescents. J Anxiety Disord (2000) 14:263–79. doi:10.1016/S0887-6185(99)00039-0
36. Achenbach TM. Manual for the Youth Self-Report and 1991 Profile. Burlington: University of Vermont (1991).
Keywords: Children’s Somatization Inventory, anxiety disorders, somatic symptoms, Poland, children and adolescents
Citation: Essau CA, Olaya B, Bokszczanin A, Gilvarry C and Bray D (2013) Somatic symptoms among children and adolescents in Poland: a confirmatory factor analytic study of the Children Somatization Inventory. Front. Public Health 1:72. doi: 10.3389/fpubh.2013.00072
Received: 10 September 2013; Paper pending published: 23 September 2013;
Accepted: 09 December 2013; Published online: 24 December 2013.
Edited by:
Ilknur Aydin Avci, Ondokuz Mayis University, TurkeyReviewed by:
Lucas Giner, University of Seville, SpainLaura Nabors, University of Cincinnati, USA
Lynn M. Breau, Glenrose Rehabilitation Hospital, Canada
Copyright: © 2013 Essau, Olaya, Bokszczanin, Gilvarry and Bray. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Cecilia A. Essau, Department of Psychology, University of Roehampton, Whitelands College, Holybourne Avenue, London SW15 4JD, UK e-mail:Yy5lc3NhdUByb2VoYW1wdG9uLmFjLnVr
 Cecilia A. Essau
Cecilia A. Essau Beatriz Olaya
Beatriz Olaya Anna Bokszczanin
Anna Bokszczanin Catherine Gilvarry1
Catherine Gilvarry1 Diane Bray
Diane Bray