- 1School of Pharmacy, Monash University Malaysia, Bandar Sunway, Malaysia
- 2Faculty of Pharmacy, Social and Administrative Pharmacy Department, Rangsit University, Muang Pathum Thani, Thailand
- 3National Vaccine Institute (Public Organization), Ministry of Public Health, Nonthaburi, Thailand
- 4Center of Pharmaceutical Outcomes Research (CPOR), Faculty of Pharmaceutical Sciences, Department of Pharmacy Practice, Naresuan University, Phitsanulok, Thailand
- 5School of Pharmacy, University of Wisconsin, Madison, WI, United States
- 6Asian Centre for Evidence Synthesis in Population, Implementation and Clinical Outcomes (PICO), Health and Well-being Cluster, Global Asia in the 21st Century (GA21) Platform, Monash University Malaysia, Bandar Sunway, Malaysia
Current study aimed to estimate clinical and economic outcomes of providing the Haemophilus influenzae type b (Hib) vaccination as a national vaccine immunization program in Thailand. A decision tree combined with Markov model was developed to simulate relevant costs and health outcomes covering lifetime horizon in societal and health care payer perspectives. This analysis considered children aged under 5 years old whom preventive vaccine of Hib infection are indicated. Two combined Hib vaccination schedules were considered: three-dose series (3 + 0) and three-dose series plus a booster does (3 + 1) compared with no vaccination. Budget impact analysis was also performed under Thai government perspective. The outcomes were reported as Hib-infected cases averted and incremental cost-effectiveness ratios (ICERs) in 2014 Thai baht (THB) ($) per quality-adjusted life year (QALY) gained. In base-case scenario, the model estimates that 3,960 infected cases, 59 disability cases, and 97 deaths can be prevented by national Hib vaccination program. The ICER for 3 + 0 schedule was THB 1,099 ($34) per QALY gained under societal perspective. The model was sensitive to pneumonia incidence among aged under 5 years old and direct non-medical care cost per episode of Hib pneumonia. Hib vaccination is very cost-effective in the Thai context. The budget impact analysis showed that Thai government needed to invest an additional budget of 110 ($3.4) million to implement Hib vaccination program. Policy makers should consider our findings for adopting this vaccine into national immunization program.
Introduction
Haemophilus influenzae type b (Hib) is a leading cause of childhood bacterial meningitis, pneumonia, and other serious infections, associated with substantial morbidity and mortality among children under 5 years of age (1). About 3–6% among Hib cases are fatal and up to 20% of patients who survive Hib meningitis have permanent hearing loss or other long-term neurological sequelae (2). Routine Hib vaccination program led to a near eradication of Hib diseases in high-income countries (3).
Recommended by the World Health Organization (WHO), Hib vaccine has been introduced into the Expanded Program on Immunization (EPI) in some low- and middle-income countries (LMICs) with the support from the Gavi, the Vaccine Alliance (4–8). Despite Gavi support, including Hib vaccine into EPI in the LMICs was delaying far behind from all high-income countries (9). Thailand and China are the only two countries in the whole world that has not introduced Hib vaccine as part of EPI (10). This is due to uncertainties around low Hib disease burden and high vaccine price, although it has been available in Thailand since 2001 (11, 12). With substantial financial barriers to implementing EPI, economic evaluation on vaccine is essential and highly recommended to inform vaccine decision-making (6, 13). As of now, there was only one economic evaluation of universal Hib vaccination based on cost-benefit analysis (CBA) for Thailand conducted in year 2010 (14). It showed that the Hib vaccine was only cost-effective if intangible benefits were included in the model. The other factors contributing to unfavorable outcomes were vaccine unit price and Hib disease incidence.
There are a number of reasons that highlight the need for updated and more comprehensive economic evaluation of this vaccine. First, the previous economic evaluation did not capture the non-meningitis non-pneumonia infection, which is one of important consequences of Hib infection. Second, the previous economic evaluation was CBA under provider and client perspectives which was not the requirement by Thai government health decision makers that need cost-utility analysis (CUA) under societal perspective to compare Hib vaccine with other health interventions. Last, the vaccine decision-making process in Thailand has undergone major changes. Before 2003, EPI committee directly informed Thai Ministry of Public Health (MOPH) decides vaccines to be included in EPI. After 2003, vaccine incorporation into EPI needed to go through two more appraisal bodies including the National Health Security Office (NHSO) currently taking hold of national vaccine fund and the National List of Essential Medicines (NLEM). In addition, since 2011, new information has been established, e.g., reduction of vaccine acquisition cost (price), vaccine effectiveness (VE) from a recent meta-analysis, and the cost-effectiveness threshold of Thai baht (THB) 160,000 ($4,878) per QALY gained in Thailand, which is useful in guiding reimbursement of medicines using a CUA recommended in the Thailand’s Health Technology Assessment (HTA) guideline (15–17). To better understand the simultaneous impact of those factors on the health economic value of Hib vaccine, we performed a CUA of universal Hib vaccination requested by National Vaccine Institute to inform decisions in including Hib vaccine as a part of this country’s EPI using the most updated available information.
Materials and Methods
Overall Description
A CUA was undertaken to estimate costs and health outcomes of routine Hib vaccination (DTP-HepB-Hib: diphtheria, tetanus, pertussis, hepatitis B, and Hib infections) with three-dose primary series at 2, 4, and 6 months with or without booster dose (3 + 0 and 3 + 1 schedules) compared to “no vaccination” (DTP-HepB) among children in Thailand. Since some cases of Hib meningitis have long-term sequelae, i.e., hearing loss, epilepsy, the lifetime time horizon was used. Discount rate of 3% was used for costs and health outcomes. Results were presented incremental cost-effectiveness ratio (ICER) in terms of monetary value (THB) per quality-adjusted life year (QALY) gained. Societal perspective was used recommended by Thailand’s HTA Guideline (18).
Decision tree combined with Markov model was constructed based on the natural history of diseases from Hib infection (Figure 1). Decision tree captures Hib infections occurred during the first 5-year of life, while Markov captures long-term outcomes throughout their life. The decision tree model captures the incidence of Hib meningitis, Hib pneumonia, Hib non-pneumonia non-meningitis (NPNM, including sepsis/bacteremia, cellulitis, and epiglottitis) during the first 5-year of life since Hib infections occur in this period of time. Hib meningitis leads to three clinical consequences including full recovery, recovery with sequelae, and death. Sequalae included hearing loss, epilepsy, and developmental deficit. The Markov model captures long-term consequences after 5-year period in decision tree model. Patients who survived from acute Hib infection were moved to Markov states which were classified into four heath statuses including (1) healthy (full recovery), (2) survived with hearing loss, (3) survived with epilepsy, and (4) survived with developmental deficit. Patients could die with different mortality rate depending on their health status after acute Hib infection. The herd effect and the vaccine adverse effects were not considered in this model. Cycle length of 1 year was used. We assumed that Hib infection could occur only once for each individual.
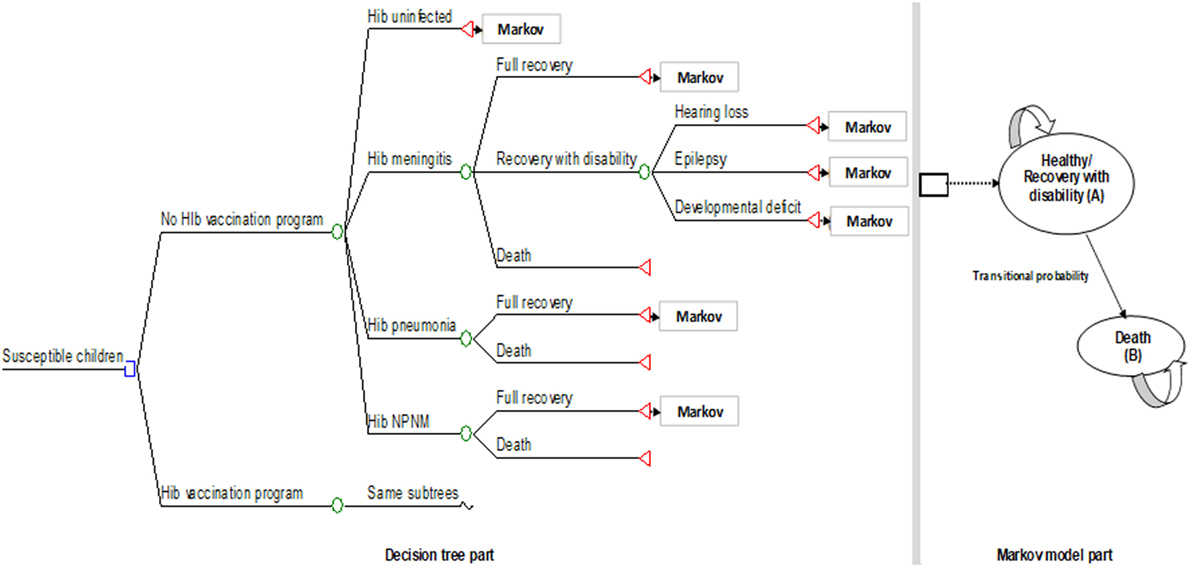
Figure 1. Decision tree combined with Markov model. Model used for evaluating costs and health-related outcomes of DTP-HepB-Hib vaccination national program compared to no vaccination (The structure of the vaccination program node is same to no vaccination node). NPNM, non-pneumonia non-meningitis; Hib, Haemophilus influenzae type b.
Model Input Parameters
Model input parameters were obtained from retrospective electronic hospital database of NHSO and published evidence (Tables 1–3). Five types of stakeholders, including health economist, health care providers (pediatrician and pediatric infectious disease specialist), patients’ representative, third-party payers, and representatives from pharmaceutical industry have participated in two stakeholder consultation meetings in order to define study population, intervention, model structure, outcome, and comparator, assumptions used, validating data inputs, providing comments on interim findings.
Epidemiological Data
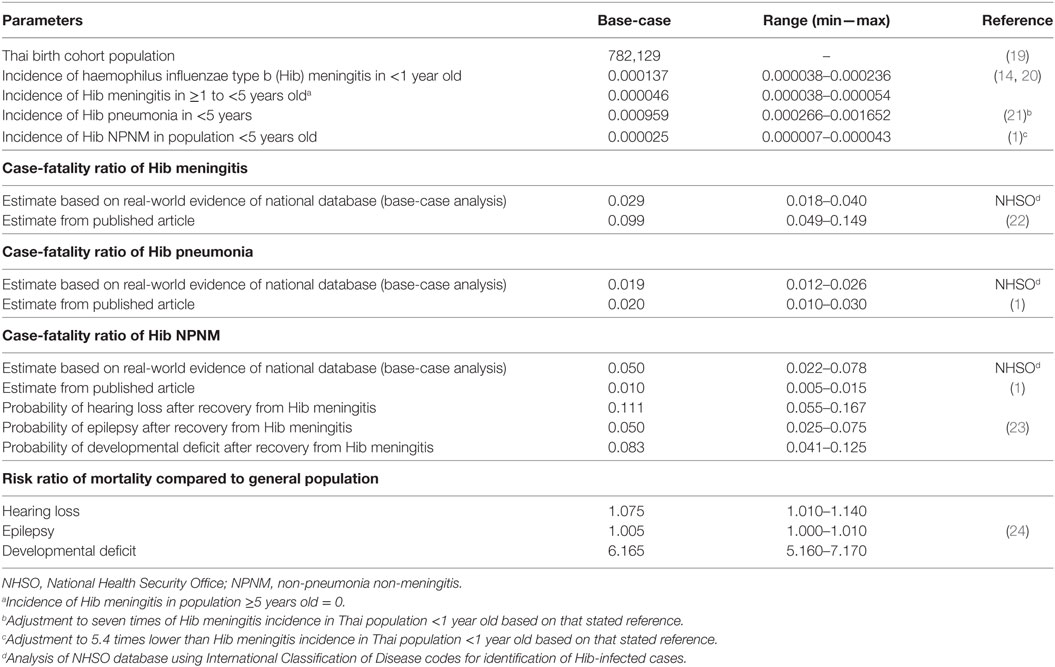
The disease incidences were derived from prospective studies of Hib meningitis incidence in Thailand and a previously published economic evaluation study (14, 20, 25). While incidence of other Hib infections was not available, the incidence of Hib pneumonia was assumed as 7 times (range of 4–10 times) of Hib meningitis incidence based on WHO estimation (21). The incidence of Hib NPNM infection consisting mainly of bacteremia was estimated to be 5.4 times less than that of Hib meningitis based on global estimates (1).
Case-fatality ratios (CFRs) associated with Hib infections were derived using electronic hospital database of NHSO covering 80% of insured Thai population during January 2008 and December 2013. CFR was calculated based on patients less than 5 years of age with primary diagnosis code of Hib meningitis [International Classification of Disease (ICD)-10: G00.0], Hib pneumonia (ICD-10: J14), or sepsis due to Hib (ICD-10: A41.3)]. The CFR of Hib meningitis, pneumonia, and sepsis that assumed to represent Hib NPNM were 2.9% (1.8–4.0%), 1.9% (1.2–2.6%), and 5.0% (2.2–7.8%), respectively. In addition, CFR from international estimates were alternatively used in a scenario analysis (1, 22). The probabilities of developing hearing loss, epilepsy, and development deficit after Hib meningitis were based on estimates in previous CBA and meta-analysis studies (14, 23). The relative risks of dying among those with sequalae were based on previous CUA study (24). These relative risks were multiplied with Thai age-specific mortality to derive the likelihood of dying from all health states in the Markov model (26) (Table 1).
VE and Coverage Rate
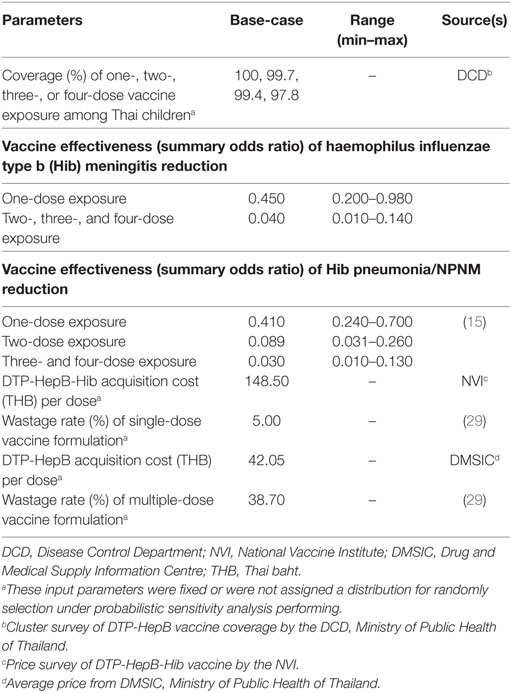
Vaccine effectiveness against vaccine-type Hib meningitis and invasive Hib disease (pneumonia and NPNM) was based on a systematic review and meta-analysis (Table 1) (15, 27, 28). It was assumed that the vaccination effect lasted over a 5-year period (15). Vaccination coverage rate was based on 2013 cluster survey study of DTP-HepB vaccine among Thai children conducted by Department of Disease Control of Thai MOPH (personal communication with expert in EPI committee) (Table 2).
Costs and Outcomes
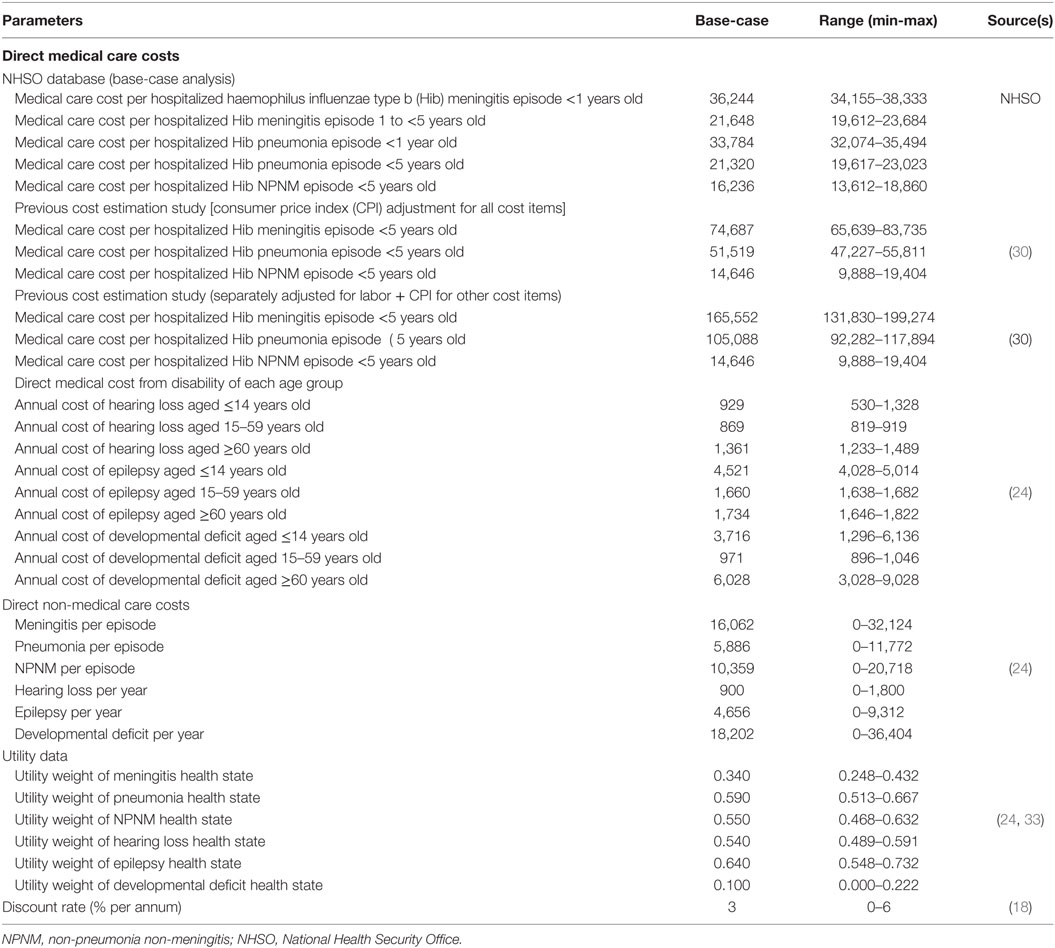
Both direct medical and direct non-medical costs were estimated. It was assumed that lost or impaired ability to work or engage in leisure activities due to morbidity would be captured in the disutility of QALY. Indirect costs were thus not included to evade double counting (18). The cost of the vaccination program included vaccine acquisition cost and wastage cost based on wastage rate of single-dosed DTP-HepB-Hib and multiple-dosed DTP-HepB vaccines from Thailand’s survey study (29). Direct medical costs for hospitalized episode of Hib infection were obtained from three cost estimation approaches in order to provide a broader possible range of cost estimates. The first approach was to derive cost estimates based on DRG using the abovementioned NHSO database (2008–2013). The adjusted relative weights (adjRW) of Hib-specific infections were multiplied with base-rate reimbursement of THB 8,200 ($250) for 1 adjRW. These costs were used in base-case analysis. The second approach was based on cost estimates obtained from a study determining cost estimates for Hib-specific infection, conducted in Thailand in year 2002 in 42 patients (30). We adjusted the estimates using medical care consumer price index (CPI) for the whole aggregated value from 2002 to 2014. The third approach was similar to the second approach where both were based on cost estimates from a study in Thailand (30). However, in the third approach, we adjusted labor cost item with the ratio of the aggregated amount for wage and salary of health care professionals of 2002 and 2014 by Thai Ministry of Health, while other cost items were adjusted using CPI (Table 1) (31). The rationale for the separated adjustment for labor cost was the observed substantial increase of health care professionals’ compensation rates in the last decade. The cost estimates from the second and third approach were used in scenario analyses. Direct non-medical costs, including transportation, meals, accommodation, facilities, caregivers’ productivity loss for hospital visits, or providing informal care, were derived from a previous study (24).
The budget impact analysis was analyzed to estimate the whole budget required and the real incremental budget for implementing of national DTP-HepB-Hib vaccination program compared to DTP-HepB in EPI among 782,129 Thai children (the number of cohort of children in year 2013). All cost values are presented in 2014 and could be converted to US dollars ($) using exchange rates THB 32.8 = $1 for international comparison (32) (Table 3).
Utility
Utility reduction was not applied for healthy individuals in our model. For infected individuals, we applied their utility weights deriving from published studies that conducted among Thai patients in the same health states of Hib infection (Table 3) (24, 33). The utility weights were based on Health Utility Index mark 3, which they were used to analyze the cost-utility of pneumococcal vaccine under the Thai context (24).
Base-Case Analysis
Primary outcomes were the difference of Hib infections with/without sequelae and death over 5 years, incremental costs, life year gained, QALYs gained, and ICER. For base-case analysis, we calculated the expected lifetime costs and outcomes for each program. The results are presented as ICER of DTP-HepB-Hib versus DTP-HepB as a part of EPI. An official willingness-to-pay (WTP) of the Thai Health Economic Working Group threshold (THB 160,000; $4,878 per QALY gained) for drug listing in NLEM year 2012 was used as cost-effectivenessthreshold (16, 17).
Sensitivity Analysis
One-way sensitivity analysis was performed to evaluate the uncertainties surrounding each input within plausible ranges of 95% CI including disease incidence, VE, costs, utilities, and discounting rate at 0 and 6% per annum and were presented using tornado diagram (Figure 2). Scenario analyses were performed when a four-dose of DPT-HB-Hib regimen (at 2, 4 and 6 months of age plus a booster dose at age between 12 and 18 months) or 3 + 1 schedule was considered, assuming the similar effectiveness between 3 + 0 and 3 + 1 dose regimens (15, 34). Scenario analyses were also performed using another two different cost estimating approaches as described previously CFR of Hib-specific infections based on published article was used in another scenario analysis. We also provide threshold analysis for vaccine acquisition cost.
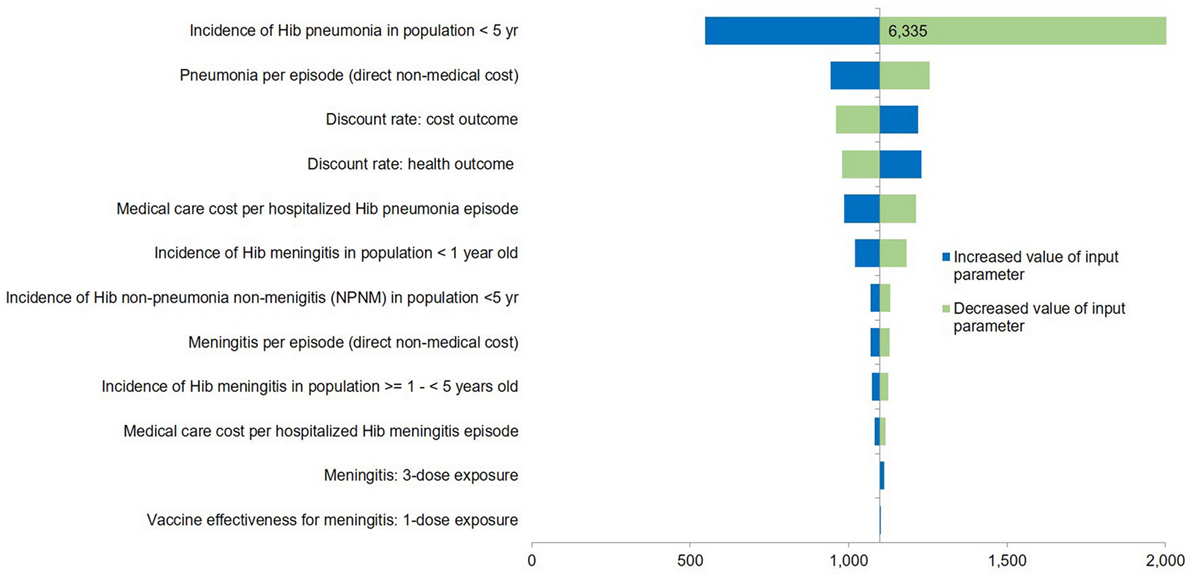
Figure 2. Tornado diagram of one-way sensitivity analysis (3 + 0 vaccination schedule with NHSO costing data). Tornado diagram summarized the results of one-way sensitivity analysis to evaluate the input uncertainties within plausible ranges of 95% confidence interval. The influential factors were listed descending with the variation of value. The vertical line represents the base-case incremental cost-effectiveness ratio (ICER). X-axis indicates the ICER. NHSO, National Health Security Office; Hib, Haemophilus influenzae type b.
Multivariate probabilistic sensitivity analysis (PSA) was conducted to simultaneously examine the effects of all parameter uncertainty using a Monte Carlo simulation performed by Microsoft Excel 2003 (Microsoft Corp., Redmond, WA) (35). The various probability distributions were defined: (a) probability and utility ranging between zero and one would follow beta-distributions, (b) costs and length of stay normally positively skewed and positive would follow gamma-distributions, and (c) VE and relative risk of dying parameters would follow a log-normal distribution. 1,000 Monte Carlo simulations were run and results were presented as cost-effectiveness acceptability curve (Figure 3) (36). The expected net monetary benefit was calculated for WTP of NLEM 2012 threshold in Thailand in order to show the probability that DPT-HB-Hib vaccination program is cost-effective.
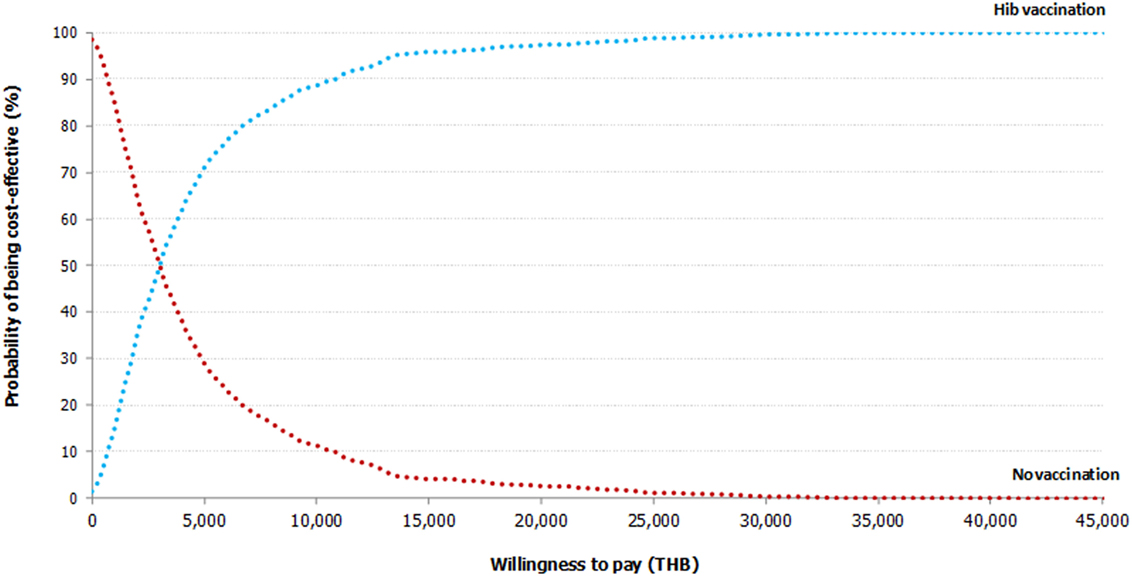
Figure 3. Cost-effectiveness acceptability curve. The cost-effectiveness acceptability curve shows the result of multivariate probabilistic sensitivity analysis based on 1,000 Monte Carlo simulations. If the willingness-to-pay threshold is set at THB 160,000 per QALY gained, the three-dose universal Hib vaccination program will have 100% probability of being cost-effective from societal perspective. THB, Thai baht; Hib, Haemophilus influenzae type b.
Results
Base-Case Analysis
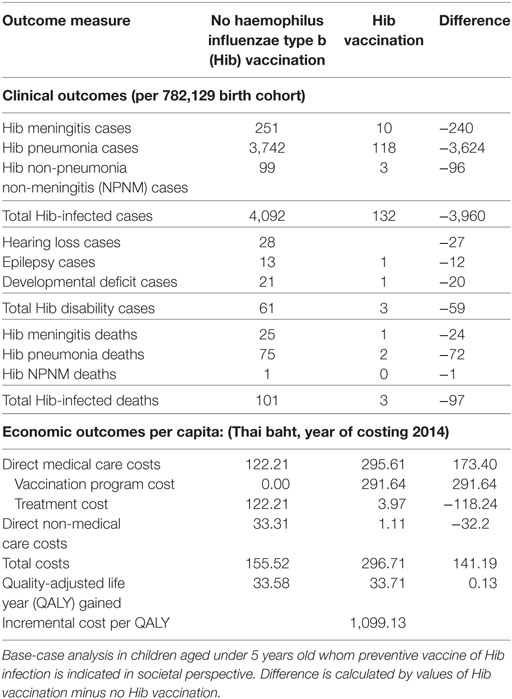
Haemophilus influenzae type b vaccination program among 782,129 Thai children could avert 3,960 Hib-infected cases, which includes 240 Hib meningitis, 3,624 Hib pneumonia, and 96 Hib NPNM over 5 years (Table 4). It could prevent 27 hearing losses, 12 epilepsies, and 20 developmental deficits which are long-term Hib meningitis-related sequelae. The vaccination would avert 89 Hib-related deaths (13 due to Hib meningitis, 71 due to Hib pneumonia, and 5 due to Hib NPNM). Table 4 also presents incremental cost per capita of implementing both three-dose universal Hib vaccination program as THB 141.19 ($4.3). The incremental vaccination program cost of a three-dose vaccination schedule would require is THB 291.64 ($8.9) based on a unit cost of THB 148.5 ($4.5) per dose and wastage cost of 5% for DTP-HepB-Hib single-dose dosage form (39% for 10-dose vial), compared to THB 42.05 ($1.3) per dose of DTP-HepB multiple-dose preparation.
Our model estimates that vaccinating children with Hib vaccine would save nearly THB 118.24 ($3.6) in direct medical care costs and THB 32.20 ($1) in direct non-medical care costs per vaccinated child. Therefore, a universal Hib infant vaccination with three-dose regimen is expected to lead to an ICER value of THB 1,099.13 ($33.5) per QALY gained under societal perspective.
Sensitivity Analyses
In Figure 2, the one-way sensitivity analyses showed that the uncertainty around incidence of Hib pneumonia for those below 5 years of age led to the largest variation of ICER from THB 479 ($14.6) to THB 2,511 ($76.6) per QALY gained. All the remaining parameters seemed to have moderate to minimal impact on cost-effectiveness results. Results of the PSA based on 1,000 Monte Carlo simulations are presented in cost-effectiveness acceptability curve (Figure 3). Despite variation in base-case parameter inputs, around 1.40% of the simulated ICERs were in the lower-right hand quadrant, indicating that Hib vaccination program is always less costly and more effective than no vaccination (dominant). Another 98.6% of ratios which lie in the upper-right hand quadrant indicate that the vaccination program is more effective but also costlier than no vaccination. However, all simulated outcomes were well below the cost-effectiveness threshold line. As shown in Figure 3, the three-dose universal Hib vaccination program from societal perspective will have 100% probability of being cost-effective if the WTP threshold is set at THB 160,000 ($4,878) per QALY gained.
Scenario Analyses
Based on current evidence of VE and coverage, 3 + 1 vaccination schedule provide costlier than 3 + 0 with the ICER was THB 1,835.53 ($56.5) per QALY gained. Two scenarios mentioned in the methodology section of higher cost estimates from previous costing study were also analyzed in order to provide an implication of varying Hib-infected direct medical care cost. For cost estimates using CPI for all cost items of previous costing study, the ICER was reduced to THB 56 ($1.7) per QALY gained, while it was cost-saving (less incremental cost with higher QALY gained) under separated labor cost adjustment using wage and salary with CPI adjustment for other cost items of previous costing study. In addition, when we used CFR from published article, which had higher CFR among Hib meningitis cases and lower CFR among Hib NPNM (Table 1), the ICER was slightly reduced to THB 1,098.23 ($33.5) per QALY gained. When analysis considered the effect of Hib vaccine only on Hib meningitis, ICER was increased to THB 31,753.21 ($968.1) per QALY gained.
Threshold Analysis
At current pricing, universal Hib infant vaccination is unlikely to be cost-saving compared to no vaccination under societal perspective. The price of DTP-HepB-Hib vaccine should be THB ≤101 ($3.1) per dose for being cost-saving intervention. For cost estimates using CPI for all cost items of previous costing study, the maximum price per dose are THB 149 ($4.5) for cost-saving. In addition, prices should be THB 232 ($7.1) under separated labor cost adjustment using wage and salary with CPI adjustment for other cost items.
Budget Impact Analysis
Based on a birth cohort of 782,129 children under base-case analysis, Thai government should invest about THB 228 ($7.0) million to implement 3 + 0 Hib vaccination program. However, total numbers of cases prevented can provide a cost reduction about THB 118 ($3.6) million. Therefore, an extra budget needed is about THB 110 ($3.4) million.
Discussion
Haemophilus influenzae type b vaccination on children below five would substantially reduce the incidence of Hib-infected cases, leading to more than 100,000 QALYs gained. Thailand has had very low Hib infection which is believed to be a contributing factor to the former decision of not including Hib vaccination in EPI for Thai children (20, 25). However, our CUA incorporating most updated information showed that the preventive vaccine affirms a good value for money from societal perspective. This CUA study indicates that DTP-HepB-Hib vaccine offers a good value for money compared with existing EPI vaccine (DTP-HepB) at current market price. The ICER lies far below the Thailand’s standard ceiling threshold of THB 160,000 ($4,878) per QALY gained (16, 17). The break-even analysis also shows that the vaccination program would be cost-saving if the current vaccine price is reduced by one-thirds at THB 101 ($3.1) per dose where vaccination cost was offset by reduced treatment costs due to fewer Hib-infected cases. The budget impact analysis accounting for vaccine coverage and wastage under Thai context points to a reasonable incremental investment at current market price with routine Hib vaccine being included in EPI. The incremental investment is THB 141 ($4.3) for three doses compared to $8.9 (THB 292) per child vaccinated in Indonesia (37).
Haemophilus influenzae type b vaccine was found to be either cost-saving or cost-effective using country-specific cost-effectiveness threshold in most of the other countries such as Indonesia, India, Belarus, and Uzbekistan (37–40). In the same region, two CUA studies in Indonesia found that Hib vaccine is a highly cost-effective intervention. The confirmed Hib meningitis incidence used in both studies was low and was from prospective study in the selected catchment area as representative of all Indonesia, like we used the Thai prospective incidence study in our analysis (20, 41). However, one CBA study published in South Korea presented non-cost-effective results similar to Thai study, main factors for showing non-cost-effectiveness discussed in the two CBA studies were vaccine price, which was quite high at the time of analysis and the low incidence of Hib-related infection in both South Korea and Thailand (14, 42).
Our study did not incorporate herd immunity into the model because of a paucity of information related to herd immunity of Hib vaccine. This assumption is in line with previous cost-effectiveness analyses, which also did not incorporate herd immunity into the model. Since our findings showed that Hib vaccine was cost-effective, inclusion of herd effect would make it even more favorable. Therefore, this lack of herd effect incorporation will not change the overall conclusion of our study.
Even though our findings were sensitive to Hib pneumonia incidence, however it did not reverse our conclusion. It is worth noting that Hib pneumonia incidence was assumed to be 4–10 times of that of meningitis same as previous CBA study in Thailand (14). We performed a more conservative analysis considering only the effect of Hib vaccine on Hib meningitis and found that ICER was still below the cost-effectiveness threshold. This reflects the robustness of our findings. Furthermore, the results in our analysis were most sensitive to Hib disease incidence, vaccine price, and discount rate. This is in line with the report from the systematic review of Hib vaccine economic evaluations which indicated that previous studies were also most sensitive to these three parameters (43). In addition, the PSA results of all 1,000 iterations fall below the national cost-effectiveness threshold.
Input parameters used in this model were Thailand-specific and obtained from high-quality sources, including systematic reviews and meta-analyses. We used Hib incidences from local prospective population-based study to estimate number of infected cases (20, 25). VE was obtained from a meta-analysis (15). Contrary to most of the vaccine CEA studies that used vaccine efficacy data for projection, the use of VE is more realistic because it was obtained from real-world evidence. Furthermore, the use of domestic inputs in the model makes health authorities better informed in introducing Hib vaccine into EPI.
This study has some limitations. First, this study adopted a static model rather than transmission dynamic. The static model used in the current analysis is unable to estimate herd effect. Thus, the current results should be deemed conservative. Using dynamic transmission model would expect to generate more favorable outcomes. Second, we used Hib infection incidence from five provinces to estimate cases for the whole country. Although all included provinces are representative of all regions of the country; however, we do not exactly know whether this figure is an over- or under-estimate of the true incidence at national level. This uncertainty was addressed in 1-way sensitivity analysis and it showed that Hib pneumonia incidence is the most influential parameter. However, it still showed cost-effectiveness when the Hib pneumonia incidence was not considered in scenario analysis. Finally, this model used one year as the model’s cycle length. This limits the recurrence of Hib infection within one year. However, the cycle length was agreed by our stakeholders, which included clinicians, researchers, and health payers. The cycle length is also similar to previous cost-effectiveness analyses of Hib vaccine.
There has been one economic evaluation of universal Hib vaccination program in Thailand using CBA approach where tangible and intangible costs and benefits were captured (14). They measured and valued intangible costs and benefits by surveying the WTP among parents who want their children to get Hib vaccine. It was shown that Hib vaccination program produced negative net benefit thus was not cost-effective compared to no vaccination program without including intangible costs. However, the intangible cost itself is difficult to quantify in monetary unit and is dependent on eliciting approach used (44). In addition to this uncertainty of CBA, HTA guideline of Thailand recommended CUA as a national preferred approach for economic evaluation. Therefore, this study was performed in line with current HTA guideline and our results are thus useful to enable comparison with CUAs of other health interventions as effectiveness is standardized as QALYs. This also allows the allocative efficiency among competed health measures under a limited health care budget. It is also important to note that the cost-effectiveness ceiling threshold used in our analysis is based on the local preference of decision makers/bodies (16, 17). Under other circumstances, they may have their own preference regarding health investment. Therefore, it is encouraged that audiences compare the results to any threshold considered as appropriate. As aforementioned, Thailand is in the minority among many countries that have already implemented Hib vaccine for their citizens. The main reason for this vaccination program not being adopted in Thailand is due to low Hib-infected incidence. However, our results using the updated information show that this vaccination program would be cost-effective even with low disease burden. Our analysis showed reconsidering of Hib vaccine to be included in EPI may be warrant based on these economic evaluation results along with other important aspects as well. In summary, DTP-HepB-Hib vaccination program for Thai children would be a very cost-effective intervention and is likely to produce a budget impact affordable to government. Furthermore, its delivery schedule is also aligned with the implemented DTP-HepB vaccine. The inclusion of DTP-HepB-Hib in Thailand’s EPI is highly encouraged.
Author Contributions
SK and NC contributed in the study conception, model conceptualization, critical review of input parameters, model analyses and interpretation, consultation-meeting arrangement, and manuscript writing. CM and ST participated in study conception, reviewed the model outputs from the clinical viewpoint and consultation-meeting arrangement, and participated in manuscript drafting. PD and DW participated in critical review of input parameters, model analyses and interpretation, and manuscript writing. All authors reviewed the final draft and collectively decided to submit for publication.
Conflict of Interest Statement
CM and ST are employees of the NVI—a not-for-profit organization. The rest of authors had funding support from NVI for conducting the study and had no other relationships or activities that could appear to have influenced the submitted work.
Acknowledgments
The authors would like to thank all participants in the two experts-stakeholders’ meetings and all staff at the National Vaccine Institute (NVI) for their administrative supports. We also thank to Mr. Kittinop Kittirotruchi for his clerical assistance.
Funding
CM and ST are employees of the National Vaccine Institute (NVI, public organization), Ministry of Public Health, Nonthaburi, Thailand – a not-for-profit organization. The rest of authors had funding support from NVI for conducting the study and had no other relationships or activities that could appear to have influenced the submitted work.
Supplementary Material
The Supplementary Material for this article can be found online at http://www.frontiersin.org/article/10.3389/fpubh.2017.00289/full#supplementary-material.
References
1. Watt JP, Wolfson LJ, O’Brien KL, Henkle E, Deloria-Knoll M, McCall N, et al. Burden of disease caused by Haemophilus influenzae type b in children younger than 5 years: global estimates. Lancet (2009) 374:903–11. doi:10.1016/s0140-6736(09)61203-4
2. MacNeil JR, Cohn AC, Farley M, Mair R, Baumbach J, Bennett N, et al. Current epidemiology and trends in invasive Haemophilus influenzae disease – United States, 1989–2008. Clin Infect Dis (2011) 53:1230–6. doi:10.1093/cid/cir735
3. Crawford SE, Daum RS. Haemophilus influenzae. In: Kliegman R, Jenson H, Stanton B, editors. Nelson Textbook of Pediatrics. Philadelphia, PA: Saunders Elsevier (2004).
4. Baqui AH, El Arifeen S, Saha SK, Persson L, Zaman K, Gessner BD, et al. Effectiveness of Haemophilus influenzae type B conjugate vaccine on prevention of pneumonia and meningitis in Bangladeshi children: a case-control study. Pediatr Infect Dis J (2007) 26:565–71. doi:10.1097/INF.0b013e31806166a0
5. Cowgill KD, Ndiritu M, Nyiro J, Slack MP, Chiphatsi S, Ismail A, et al. Effectiveness of Haemophilus influenzae type b conjugate vaccine introduction into routine childhood immunization in Kenya. JAMA (2006) 296:671–8. doi:10.1001/jama.296.6.671
6. Gessner BD, Duclos P, Deroeck D, Nelson EA. Informing decision makers: experience and process of 15 National Immunization Technical Advisory groups. Vaccine (2010) 28(Suppl 1):A1–5. doi:10.1016/j.vaccine.2010.02.025
7. Lee EH, Lewis RF, Makumbi I, Kekitiinwa A, Ediamu TD, Bazibu M, et al. Haemophilus influenzae type b conjugate vaccine is highly effective in the Ugandan routine immunization program: a case-control study. Trop Med Int Health (2008) 13:495–502. doi:10.1111/j.1365-3156.2008.02027.x
8. UNICEF. UNICEF’s Engagement with the GAVI Alliance. (2015). Available from: http://www.unicef.org/partners/Partnership_profile_2012_GAVI_Alliance_V7_approved.pdf
9. Global Alliance for Vaccines and Immunisation. Hib Initiative: A GAVI Success Story. (2015). Available from: http://www.gavi.org/library/news/roi/2010/hib-initiative--a-gavi-success-story/
10. Centre, Johns Hopkins Bloomberg School of Public Health I. V. A. IVAC (Johns Hopkins Bloomberg School of Public Health). (2016).
11. Griffiths UK, Clark A, Hajjeh R. Cost-effectiveness of Haemophilus influenzae type b conjugate vaccine in low- and middle-income countries: regional analysis and assessment of major determinants. J Pediatr (2013) 163:S50–9. doi:10.1016/j.jpeds.2013.03.031
12. Bureau of Drug Control, Food and Drug Administration & Ministry of Public Health of Thailand. Hib Vaccine (Vaxem-Hib) Registration (2003). Available from: http://drug.fda.moph.go.th/zone_search/files/1C_128_44_N_Vaxem-Hib.pdf
13. World Health Organization. Conclusions and recommendations from the Immunization Strategic Advisory Group. Wkly Epidemiol Rec (2006) 81:2–11.
14. Muangchana C, Warinsatian P. Incorporation of private demand into cost-benefit analysis of a universal Hib vaccination program in Thailand. Southeast Asian J Trop Med Public Health (2011) 42:376–87.
15. Jackson C, Mann A, Mangtani P, Fine P. Effectiveness of Haemophilus influenzae type b vaccines administered according to various schedules: systematic review and meta-analysis of observational data. Pediatr Infect Dis J (2013) 32:1261–9. doi:10.1097/INF.0b013e3182a14e57
16. Sangmala P, Chaikledkaew U, Tanwandee T, Pongchareonsuk P. Economic evaluation and budget impact analysis of the surveillance program for hepatocellular carcinoma in Thai chronic hepatitis B patients. Asian Pac J Cancer Prev (2014) 15:8993–9004. doi:10.7314/APJCP.2014.15.20.8993
17. Thavorncharoensap M, Teerawattananon Y, Natanant S, Kulpeng W, Yothasamut J, Werayingyong P. Estimating the willingness to pay for a quality-adjusted life year in Thailand: does the context of health gain matter? Clinicoecon Outcomes Res (2013) 5:29–36. doi:10.2147/ceor.s38062
18. Chaikledkaew U, Teerawattananon Y, Kongpittayachai S, Suksomboon N, editors. Thailand’s National Health Technology Assessment Guidelines. 1st ed. Nonthaburi: The Graphico Systems (2009).
19. National Statistical Office of Thailand. The Number of Births by Sex, Region, and Province in Thailand: 2004–2013 (2017). Available from: http://service.nso.go.th/nso/web/statseries/statseries01.html
20. Rerks-Ngarm S, Treleaven SC, Chunsuttiwat S, Muangchana C, Jolley D, Brooks A, et al. Prospective population-based incidence of Haemophilus influenzae type b meningitis in Thailand. Vaccine (2004) 22:975–83. doi:10.1016/j.vaccine.2003.09.013
21. World Health Organization. Estimating the Local Burden of Haemophilus influenzae Type b (Hib) Disease Preventable by Vaccination: A Rapid Assessment Tool (2015). Available from: http://whqlibdoc.who.int/hq/2001/WHO_V&B_01.27.pdf
22. Ahmed AS, Khan NZ, Hussain M, Amin MR, Hanif M, Mahbub M, et al. Follow-up of cases of Haemophilus influenzae type b meningitis to determine its long-term sequelae. J Pediatr (2013) 163:S44–9. doi:10.1016/j.jpeds.2013.03.030
23. Baraff LJ, Lee SI, Schriger DL. Outcomes of bacterial meningitis in children: a meta-analysis. Pediatr Infect Dis J (1993) 12:389–94. doi:10.1097/00006454-199305000-00008
24. Kulpeng W, Leelahavarong P, Rattanavipapong W, Sornsrivichai V, Baggett HC, Meeyai A, et al. Cost-utility analysis of 10- and 13-valent pneumococcal conjugate vaccines: protection at what price in the Thai context? Vaccine (2013) 31:2839–47. doi:10.1016/j.vaccine.2013.03.047
25. Muangchana C, Chunsuttiwat S, Rerks-Ngarm S, Kunasol P. Bacterial meningitis incidence in Thai children estimated by a rapid assessment tool (RAT). Southeast Asian J Trop Med Public Health (2009) 40:553–62. doi:10.1017/S0950268812000957
26. World Health Organization. Global Health Observatory Data Repository: Life Tables by Country (Thailand) (2017). Available from: http://apps.who.int/gho/data/?theme=main&vid=61640
27. Griffiths UK, Clark A, Gessner B, Miners A, Sanderson C, Sedyaningsih ER, et al. Dose-specific efficacy of Haemophilus influenzae type b conjugate vaccines: a systematic review and meta-analysis of controlled clinical trials. Epidemiol Infect (2012) 140:1343–55. doi:10.1017/s0950268812000957
28. Bar-On ES, Goldberg E, Hellmann S, Leibovici L. Combined DTP-HBV-HIB vaccine versus separately administered DTP-HBV and HIB vaccines for primary prevention of diphtheria, tetanus, pertussis, hepatitis B and Haemophilus influenzae B (HIB). Cochrane Database Syst Rev (2012) 4:CD005530. doi:10.1002/14651858.CD005530.pub3
29. Techathawat S, Yoochareon P, Varinsathien P, Rasjarmrearnsook A. Wastage of multi-dose measles vaccine survey in public health facilities. Dis Control J (2008) 34:311–26.
30. Wisasa W, Jiamton S, Kongsin S. Measurement of the Hib Disease Burden in Thailand in Phase 1b: Prospective Study of Meningitis and Pneumonia Attributable to Hib. Nonthaburi, Thailand: Department of Communicable Disease Control, Ministry of Public Health (2002).
31. Office of the National Economic and Social Development Board. The Budget of the Ministry of Health of Thailand: 2003–2014 (2015). Available from: http://social.nesdb.go.th/SocialStat/StatReport_FullScreen.aspx?reportid=257&template=1R1C&yeartype=M&subcatid=18
32. OANDA. Historical Exchange Rates: USD to THB Year 2014 (2017). Available from: http://www.oanda.com/currency/historical-rates/
33. Kulpeng W, Sornsrivichai V, Chongsuvivatwong V, Rattanavipapong W, Leelahavarong P, Cairns J, et al. Variation of health-related quality of life assessed by caregivers and patients affected by severe childhood infections. BMC Pediatr (2013) 13:122. doi:10.1186/1471-2431-13-122
34. Low N, Redmond SM, Rutjes AW, Martínez-González NA, Egger M, di Nisio M, et al. Comparing Haemophilus influenzae type b conjugate vaccine schedules: a systematic review and meta-analysis of vaccine trials. Pediatr Infect Dis J (2013) 32:1245–56. doi:10.1097/INF.0b013e31829f0a7e
35. Briggs A, Sculpher M, Claxton K. Decision Modelling for Health Economic Evaluation. New York: Oxford University Press (2006).
36. Limwattananon S. Handling uncertainty of the economic evaluation result: sensitivity analysis. J Med Assoc Thai (2008) 91:S59–65.
37. Gessner BD, Sedyaningsih ER, Griffiths UK, Sutanto A, Linehan M, Mercer D, et al. Vaccine-preventable haemophilus influenza type B disease burden and cost-effectiveness of infant vaccination in Indonesia. Pediatr Infect Dis J (2008) 27:438–43. doi:10.1097/INF.0b013e318165f1ba
38. Broughton EI. Economic evaluation of Haemophilus influenzae type B vaccination in Indonesia: a cost-effectiveness analysis. J Public Health (Oxf) (2007) 29:441–8. doi:10.1093/pubmed/fdm055
39. Clark AD, Griffiths UK, Abbas SS, Rao KD, Privor-Dumm L, Hajjeh R, et al. Impact and cost-effectiveness of Haemophilus influenzae type b conjugate vaccination in India. J Pediatr (2013) 163:S60–72. doi:10.1016/j.jpeds.2013.03.032
40. Griffiths UK, Clark A, Shimanovich V, Glinskaya I, Tursunova D, Kim L, et al. Comparative economic evaluation of Haemophilus influenzae type b vaccination in Belarus and Uzbekistan. PLoS One (2011) 6:e21472. doi:10.1371/journal.pone.0021472
41. Gessner BD, Sutanto A, Linehan M, Djelantik IG, Fletcher T, Gerudug IK, et al. Incidences of vaccine-preventable Haemophilus influenzae type b pneumonia and meningitis in Indonesian children: hamlet-randomised vaccine-probe trial. Lancet (2005) 365:43–52. doi:10.1016/S0140-6736(04)17664-2
42. Shin S, Shin YJ, Ki M. Cost-benefit analysis of Haemophilus influenzae type B immunization in Korea. J Korean Med Sci (2008) 23:176–84. doi:10.3346/jkms.2008.23.2.176
43. Griffiths UK, Miners A. Economic evaluations of Haemophilus influenzae type b vaccine: systematic review of the literature. Expert Rev Pharmacoecon Outcomes Res (2009) 9:333–46. doi:10.1586/erp.09.38
Keywords: economic evaluation, budget impact analysis, Haemophilus influenzae, Hib, Haemophilus vaccines, vaccination, Thailand
Citation: Kotirum S, Muangchana C, Techathawat S, Dilokthornsakul P, Wu DB-C and Chaiyakunapruk N (2017) Economic Evaluation and Budget Impact Analysis of Vaccination against Haemophilus influenzae Type b Infection in Thailand. Front. Public Health 5:289. doi: 10.3389/fpubh.2017.00289
Received: 26 July 2017; Accepted: 19 October 2017;
Published: 20 November 2017
Edited by:
Nemanja Rancic, Military Medical Academy, SerbiaReviewed by:
Guenka Ivanova Petrova, Medical University, Sofia, BulgariaZoltán Vokó, Eötvös Loránd University, Hungary
Copyright: © 2017 Kotirum, Muangchana, Techathawat, Dilokthornsakul, Wu and Chaiyakunapruk. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nathorn Chaiyakunapruk, bmF0aG9ybi5jaGFpeWFrdW5hcHJ1a0Btb25hc2guZWR1
†See Author’s contribution on the last page.
 Surachai Kotirum
Surachai Kotirum Charung Muangchana3†
Charung Muangchana3† Piyameth Dilokthornsakul
Piyameth Dilokthornsakul David Bin-Chia Wu
David Bin-Chia Wu Nathorn Chaiyakunapruk
Nathorn Chaiyakunapruk