- 1Department of Civil and Environmental Engineering, University of Vermont, Burlington, VT, United States
- 2Gund Institute for Environment, University of Vermont, Burlington, VT, United States
- 3Department of Agriculture, Landscape and Environment, University of Vermont, Burlington, VT, United States
- 4Casella Center for Circular Economy and Sustainability, University of Vermont, Burlington, VT, United States
- 5Rubenstein School of Environment and Natural Resources, University of Vermont, Burlington, VT, United States
Plastic has become a prominent material type used for numerous purposes since the 1950s and persists in waterbodies, sediments, and terrestrial soils worldwide. Through time, plastics break apart into smaller fragments that become dispersed throughout the environment. Organic waste derived soil amendments, such as compost, may serve as a transport vector for plastics into terrestrial soils. A Vermont-wide survey was conducted to provide a range of plastic abundance, mass, and types found in composts and contextualize future research on plastic effects. Twenty composts were analyzed including composts derived from feedstocks with both high (15% or more by volume) and low/no (5% or less by volume) food waste inclusion. Plastics were isolated in multiple size classes (> 5 mm, 1–5 mm, and 0.5–1 mm) with sieving, 30% hydrogen peroxide digestion, and microscopy. Fourier transform infrared (FTIR) spectrum similarity was also used to identify plastic polymers. Ranges of 0 to 1,201 plastic particles per dry kg of compost and 0 to 0.056% w/w plastic contamination on a dry mass basis were found across the composts. Plastic abundance was not a predictor of plastic mass. No statistical differences were found between the high and low/no food waste compost groups due to variability and relatively low sample sizes, although the five most contaminated composts (> 0.02% w/w) were all in the high food waste group. Methodological challenges and recommendations are also discussed, including emphasis on establishing confidence levels for putative microplastic confirmation under the uncertainty that is inherent to analyzing complex organic matrices using microscopy and FTIR.
1 Introduction
Over the last 70 years, plastic has become ubiquitous throughout manufactured products. Plastic is relatively easy to produce, and its lightweight and versatile qualities make it a prime material across many industries (Veidis et al., 2022). Approximately 8.3 billion metric tonnes of plastic were produced between 1950 and 2015, nearly 60% of which has been discarded (Geyer et al., 2017). Plastics entering the environment do not reliably degrade but instead typically break apart into smaller fragments (Thompson et al., 2004). Sizes of plastics are typically categorized as macroplastics (larger than 10 or 25 mm), mesoplastics (5 to 10 or 25 mm), microplastics (1 μm to 5 mm or simply < 5 mm), and nanoplastics (smaller than 1 μm) (Cole et al., 2011; Braun et al., 2021; Romeo et al., 2015). Size ranges for these categories, however, vary between studies and applications (Hartmann et al., 2019). Primary microplastics are direct-manufactured plastics and are common in cosmetics and some agricultural applications (ECHA, 2020; Napper et al., 2015). Secondary microplastics are produced from the fragmentation of primary plastics, including primary microplastics that enter the environment (Thompson et al., 2004; Rillig et al., 2021).
Research on environmental microplastic pollution began in the 1970s and has focused most prominently on aquatic environments (Colton et al., 1974). Soils, however, receive an estimated 4–23 times more mismanaged plastic by mass than oceans annually (Horton et al., 2017). Nizzetto et al. (2016) estimated that roughly 44,000–300,000 metric tonnes of microplastics enter North American croplands each year due to application of wastewater sludge/biosolids. In some cases, compost can be a dominant pathway of microplastic introduction into soils (e.g., Zhang et al., 2023). To date, however, there has been limited research on the abundance and fate of microplastics in food waste streams and composts (Porterfield et al., 2023a), and related data are scarce in the United States. Such studies are necessary to inform end-users of organic amendments and to inform research on the effects of microplastic pollution on environmental quality.
To address knowledge gaps in the current understanding of plastic contamination in compost, we analyzed commercially produced composts across the US state of Vermont. Specific research questions included: (1) What levels of compost plastic contamination exist in terms of both particle counts and mass? (2) Are composts derived from feedstocks including a substantial fraction of food waste significantly more contaminated with plastic than those derived solely from manure, bedding, and green waste mixtures? (3) Are microplastic abundance and mass correlated in composts? (4) What particle shapes and polymer types account for the greatest proportions of plastic counts and mass? By answering these questions, our larger objective was to contribute to the small but growing pool of data on microplastic contamination in organic residuals in the United States and globally and thus inform efforts to understand ecological effects of microplastics in agricultural soils, mitigate future contamination, and protect environmental quality.
2 Materials and methods
2.1 Compost sampling
Composting facilities were selected from the Vermont Agency of Natural Resources’ (VT ANR) current records of permitted non-farm composting facilities (VT DEC, 2021), and a 2018 study (including on- and off-farm sites) by the Composting Association of Vermont in partnership with VT ANR. Facilities that are permitted as waste management facilities were prioritized in site selection because these facilities are regulated by VT ANR and must meet compost operational and product standards. A composter questionnaire was approved by the University of Vermont Institutional Review Board (STUDY00001988). Names and locations are not provided here to maintain the anonymity of facilities. The questionnaire was used to gather basic information about the composting process at each facility, including composting method, feedstock characteristics, sieve sizes, and existing protocols for minimizing contaminants.
A total of 20 finished composts were sampled across 15 different composting facilities between February 23 and April 22, 2022. At 5 of the 15 facilities, two samples were collected due to the existence of two distinct composts based on food waste inclusion in the feedstock. If the facility sieved their compost prior to sale, then sieved compost was collected. If sieved compost was unavailable or the facility did not sieve their compost, then unsieved compost was collected. For compost sampling, a representative windrow was selected and divided into quadrants. For each quadrant, a random location was chosen near the middle of the quadrant and the top ~15 cm of compost was removed with a stainless-steel spade to avoid sampling surface material that might not be representative of the windrow. A clean (i.e., washed with soap followed by ASTM Type II water rinse) stainless-steel scoop (473 mL) was then used to take upper, middle, and lower stratified samples at that location. At each of these 3 depths, 1.5 scoops were taken (approximately 750 mL). This was repeated for each quadrant to yield a composite sample with a total volume of ~9,100 mL. Samples were transported in pre-washed and DI-rinsed food-grade HDPE buckets with sealing lids applied directly after collection of each sample. A field blank bucket was taken when sampling and no plastic particles were detected. After collection, samples were stored at 4°C prior to analysis.
2.2 Isolation of larger plastics and microplastics
Compost samples were mixed in their respective buckets using a metal spatula cleaned between samples. After mixing, a 200-gram sample of each compost was dried in aluminum containers with loosely fitted foil covers at 60°C until a constant dry mass was achieved. Compost total solids (TS) contents were measured according to ASTM D2974.
A 25-gram sample of dried compost was then sieved through a clean 5-mm stainless-steel mesh sieve. Materials retained on the 5-mm sieve were then weighed in an aluminum weigh boat and recorded, and this mass was then subtracted from the total mass of 25 g to calculate the mass of material < 5 mm. This was repeated for three replicates of each of the 20 compost samples, plus 6 procedural no-compost controls (1 per every 10 samples). The material retained by each 5-mm sieve was then cleaned by adding ~200 mL of ASTM Type II water to each sample, and the water-compost mixture was gently stirred with a glass stir rod. After 7 days, samples were poured over a 1 mm metal sieve. The wet retained materials on the 1 mm sieve were inspected visually and putative larger plastics (> 5 mm) were extracted according to dichotomous key criteria adapted from Hidalgo-Ruz et al. (2012) and used previously by Porterfield et al. (2023b). Key characteristics of potential plastics include unnatural colors, size regularity, and presence of patterns or multiple colors on the particle. A ruler was used to ensure each extracted particle was > 5 mm in at least one dimension. Extracted particles were air-dried under aluminum foil, weighed, photographed, and stored in labeled glass vials. Additionally, the shape and color of macroparticles were recorded along with a timestamp for identification. No putative plastics of a size of < 5 mm were found in the materials retained on the 5 mm sieve.
Next, a 2.50 (± 0.005) g dried, < 5 mm sieved compost sample was placed into an acid washed beaker and 200 mL of 30% hydrogen peroxide (H2O2) (or ASTM Type II water for the laboratory blanks) was added to each sample (Gui et al., 2021; Meixner et al., 2020; Schwinghammer et al., 2020; Porterfield et al., 2023b). After the addition of H2O2, each sample was stirred to submerge buoyant compost particles with an acid-washed magnetic stir bar and a stir plate. A squeeze bottle filled with ASTM Type II water was used to rinse the stir bar and recovery wand over each beaker. Stirring was repeated after 3–5 days if particles again appeared to be emergent. Beakers were left in a fume hood for 7 days (Nuelle et al., 2014; Ruggero et al., 2020). While not actively being worked with, all samples were covered with a watch glass top to prevent intrusion of plastics from the surrounding environment.
Custom-built sieves were used to strain and extract plastic from digested compost samples (Porterfield et al., 2023b). A 1-mm sieve was stacked on top of a 0.5-mm sieve and placed in a large (10 cm) Buchner funnel. The funnel was placed over a liquid-collecting beaker. The digested compost samples were gently mixed to suspend any particulates that had settled, and the contents were poured through the stacked sieves (Masura et al., 2015). A squeeze bottle was then used to wash the particles on the stacked sieves with 500 mL Type II ASTM water. Sieves were then placed on Kim Wipes and covered with aluminum foil to dry at room temperature. Method blanks (similar procedure followed without a compost sample) were tested and confirmed no plastic contamination.
A light stereo microscope (AmScope 20X-80X Compact Multi-Lens) was used to analyze each sieve for potential microplastics. Each sieve was placed on the microscope stage under 40x magnification, and the entire surface was dissected with metal forceps twice. The same dichotomous key used for particles > 5 mm was again used to identify potential microplastics < 5 mm. Particles that disintegrated during processing but were in proximity on the sieve were counted as one particle. A photograph of each particle was taken, and identifying characteristics (shape, color, length) were recorded. Then, forceps were used to transfer the particle from the sieve to a scintillation vial. An ASTM Type II water dampened laboratory wipe was used to decrease static on the forceps to aid in particle transfer. After analysis, each sieve was covered with aluminum foil and placed back into a covered aluminum tin for storage.
A microbalance was used to measure the mass of particles that could be recovered from their glass storage vials. Vials were opened and examined under light, and then gently tapped onto an aluminum weigh boat. The median mass of measured particles for each size fraction and shape was assumed for any particles lost due to transfer or inability to remove from the scintillation vials, and half of the detection limit (0.0005 mg) was assumed for particles that measured below detection.
2.3 Plastic identification
Three methods were used to evaluate plastic identification for each particle: (1) visual analysis of photos collected during microscopy, (2) spectroscopy with an attenuated total reflection (ATR) Fourier-transform infrared (FTIR) spectrometer, and (3) peak analysis of FTIR generated spectra. First, photos of each particle from the microscope were reviewed by three individuals and classified in binary fashion as plastic or non-plastic based on a dichotomous key (Porterfield et al., 2023b). Second, all recoverable putative plastic particles were measured with a Bruker Alpha II Compact FTIR spectrometer with a diamond ATR accessory. Prior to each analysis, the instrument was cleaned with 70% ethanol. Particle and background measurements were taken with the Bruker OPUS 8.5 software at a resolution of 4 cm−1 and recorded for the mid-IR spectrum range of 4,000 cm−1 to 400 cm−1 and 24 scans. Spectral results were compared to the Bruker Polymers library and a custom library of common plastics (Löder et al., 2015) for a percent spectral match with the standard “Library Search” function, and a minimum Hit Quality (HQ, unitless) of 200 was used. HQ is a numeric measure of the similarity of the measured spectrum to a reference spectrum and has a maximum value of 1,000 (absolute match). The top two highest HQ results were saved for each particle, and the HQ and material match were recorded for further analysis. Third, the spectrum of each measured particle from FTIR analysis was analyzed for the presence of peaks in key locations (Veerasingam et al., 2021). If a particle had a hit quality of a polymer above 200 out of 1,000, the spectrum was analyzed for similarity to the closest matching particle. If the particle had no hits, the spectral peaks were analyzed for location and shape of key plastic or organic peaks (Jung et al., 2018).
Plastic status of particles was finalized according to a ranking with four confidence levels (CL). Particles only meeting one of the following criteria were designated as having the lowest confidence level (CL0): positive visual plastic identification by all three analysts, FTIR HQ for a plastic polymer < 600, spectrum matching a plastic spectrum for FTIR. Particles meeting two of those criteria were assigned CL1. If the FTIR test reported a HQ > 600 and one of the other criteria was also met, then CL2 was used. The greatest confidence level (CL3) required meeting all three criteria with FTIR HQ > 600.
2.4 Calculations and statistics
Plastic contamination was quantified in terms of both abundance (particle count per dry kg) and mass (% w/w dry basis) based on three replicates per sample, including both microplastics (0.5–1 mm and 1–5 mm) and larger plastics (> 5 mm). Particles with CL0 were excluded from final count and mass estimates, but were included in related graphs to help visualize uncertainty. Wilcoxon rank-sum tests were used to compare composts derived from feedstocks characterized by high food waste content (≥ 15% by volume) and low/no food waste content (≤ 5% by volume), and simple linear regression was used to determine whether there was a predictive relationship between plastic count and mass in composts (counts and masses of zero were excluded). Data for each food waste group are reported as ranges and mean values ± 1 standard deviation. Data analyses were performed in Microsoft Excel and RStudio (Posit Team, 2025).
3 Results
Characteristics of the composting process at each facility are provided in Supplementary Table 1 and photographs of example particles separated from compost samples in this study are provided in Supplementary Figure 1.
3.1 Plastic abundance in composts
Total plastic particle count per dry kg of compost for all three size fractions (0.5–1 mm, 1–5 mm, and > 5 mm) ranged from 0 to 1,201 (Figure 1A) when only particles meeting criteria for higher levels of confidence (CL1, CL2, and CL3) were included. This range expands to 0 to 1,692 particles per dry kg with the inclusion of particles meeting only criteria associated with the lowest confidence level (CL0) (Figure 1A). The distribution of confidence levels for plastic counts within each compost sample was variable, ranging from composts only containing CL0 particles to those only containing CL3 particles (Figure 1B). For composts derived from feedstocks containing ≥ 15% food waste by volume (n = 14), mean total plastic count (± 1 standard deviation) was 190 ± 362 per dry kg of compost, with most particles being in the 1–5 mm size fraction on average (Table 1). Mean total plastic count for composts derived from feedstocks having low/no food waste content (≤ 5% by volume, n = 6) was 65 ± 101 particles per dry kg, with a plurality of particles in the 0.5–1 mm size fraction (Table 1). Total plastic particle counts were highly variable for both the high and low/no food waste groups with no detectable statistical difference between groups (p = 0.647) (Table 1).
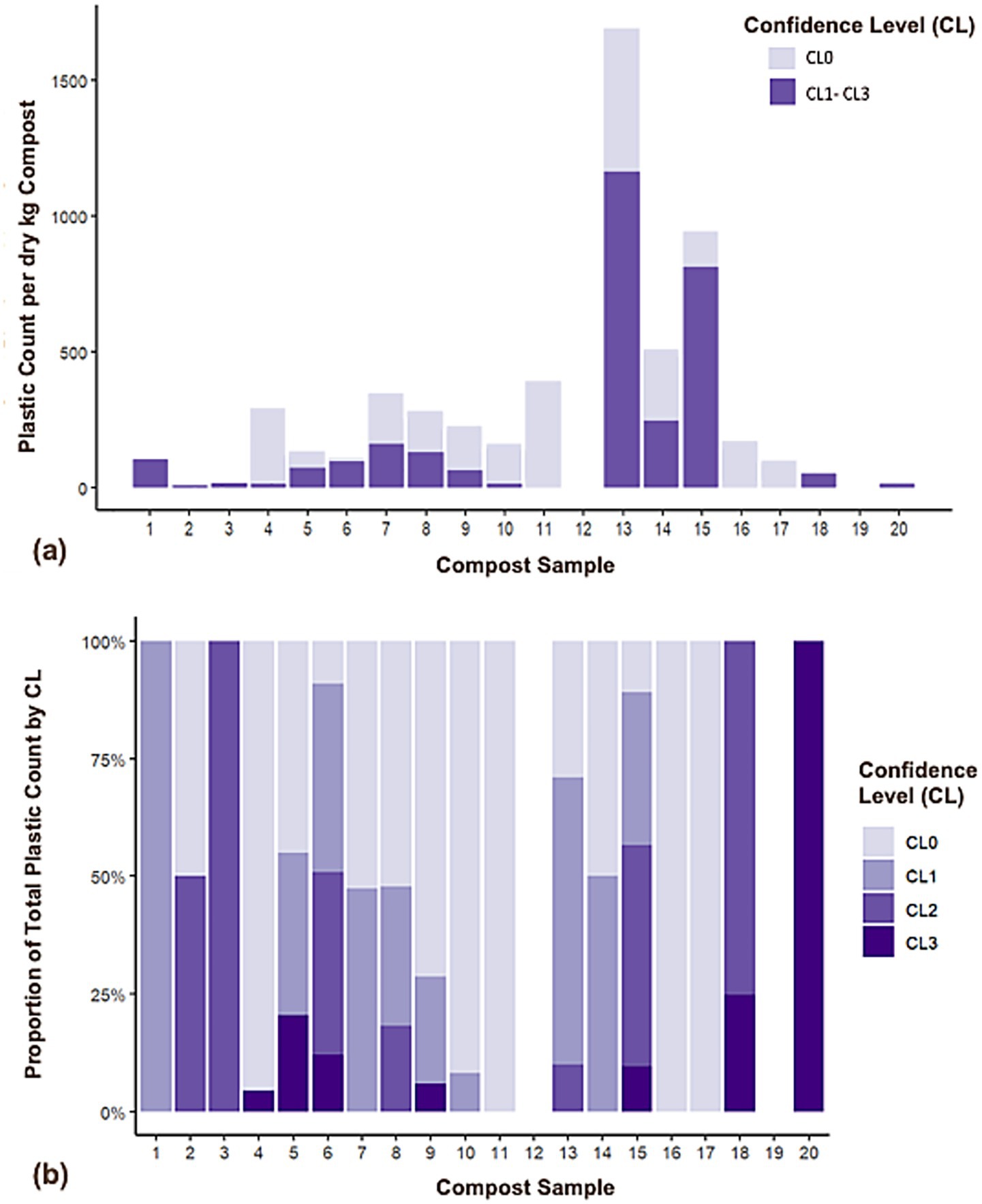
Figure 1. (a) Plastic count per kg dry mass compost, all sizes with CL0 particles distinguished from CL1–CL3 particles, and (b) proportion of total plastic count by confidence level for each sample, including all particle sizes. Data are mean values.
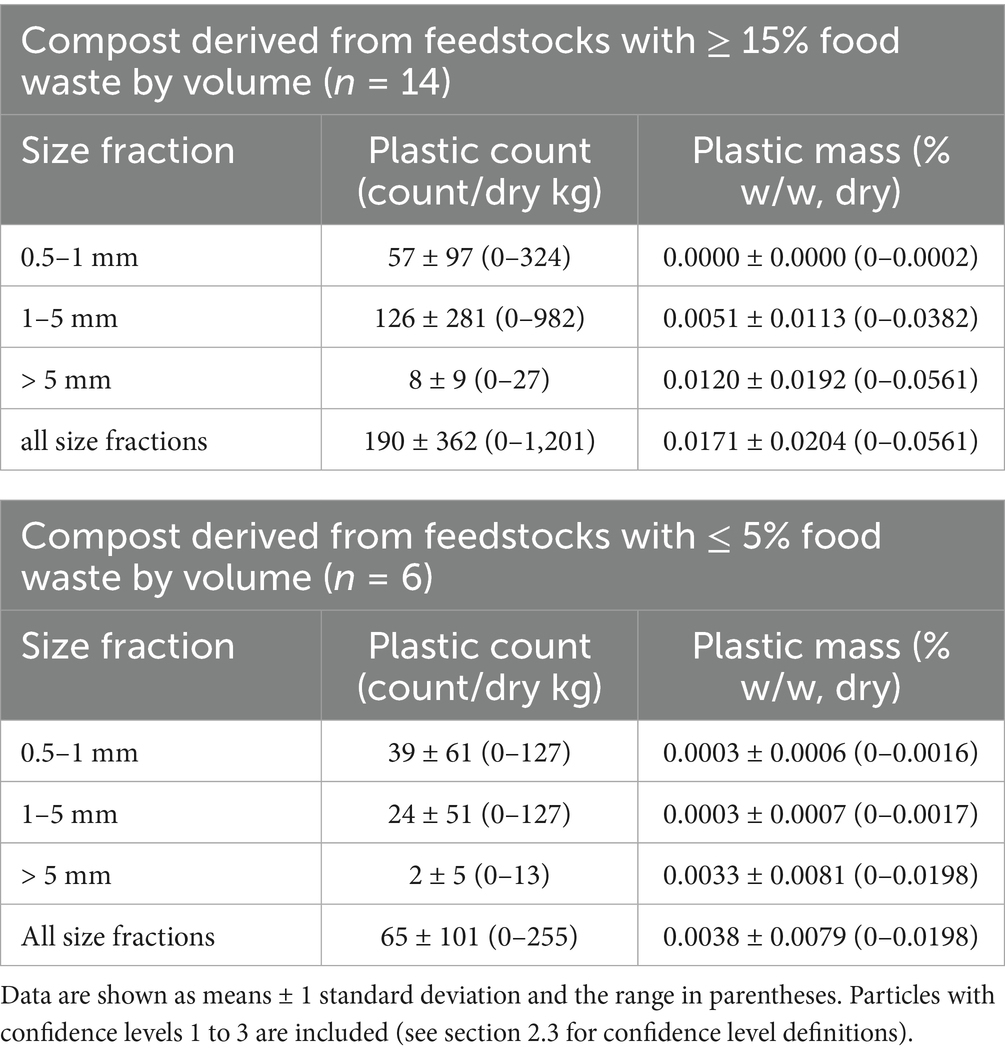
Table 1. Compost plastic contamination particle counts and mass by plastic size fractions and high (≥ 15%) versus low/no (≤ 5%) food waste in feedstock recipe by volume.
3.2 Plastic mass in composts
The percentage mass of plastic per dry mass of compost ranged from 0 to 0.0561% w/w across the 20 composts (Table 1). The upper end of this range would increase to 0.2241% w/w with the inclusion of particles identified with the lowest confidence level (CL = 0). Excluding lowest confidence particles, half of the compost samples analyzed had negligible plastic mass (i.e., not visible in Figure 2A). Five compost samples (all containing food waste as a feedstock) contained plastic accounting for ≥ 0.02% of dry compost mass (Figure 2A). As with counts, the distribution of confidence levels for mass within each compost sample was variable, again ranging from composts only containing CL0 particles to those only containing CL3 particles (Figure 2B). Total plastic particle masses were highly variable for both the high and low/no food waste groups with no detectable statistical difference between groups (p = 0.198) (Table 1). Plastic counts were unable to predict plastic mass in composts [mass = (1.878e-02) - (6.399e-06) * count, r2 = 0.013, p = 0.688].
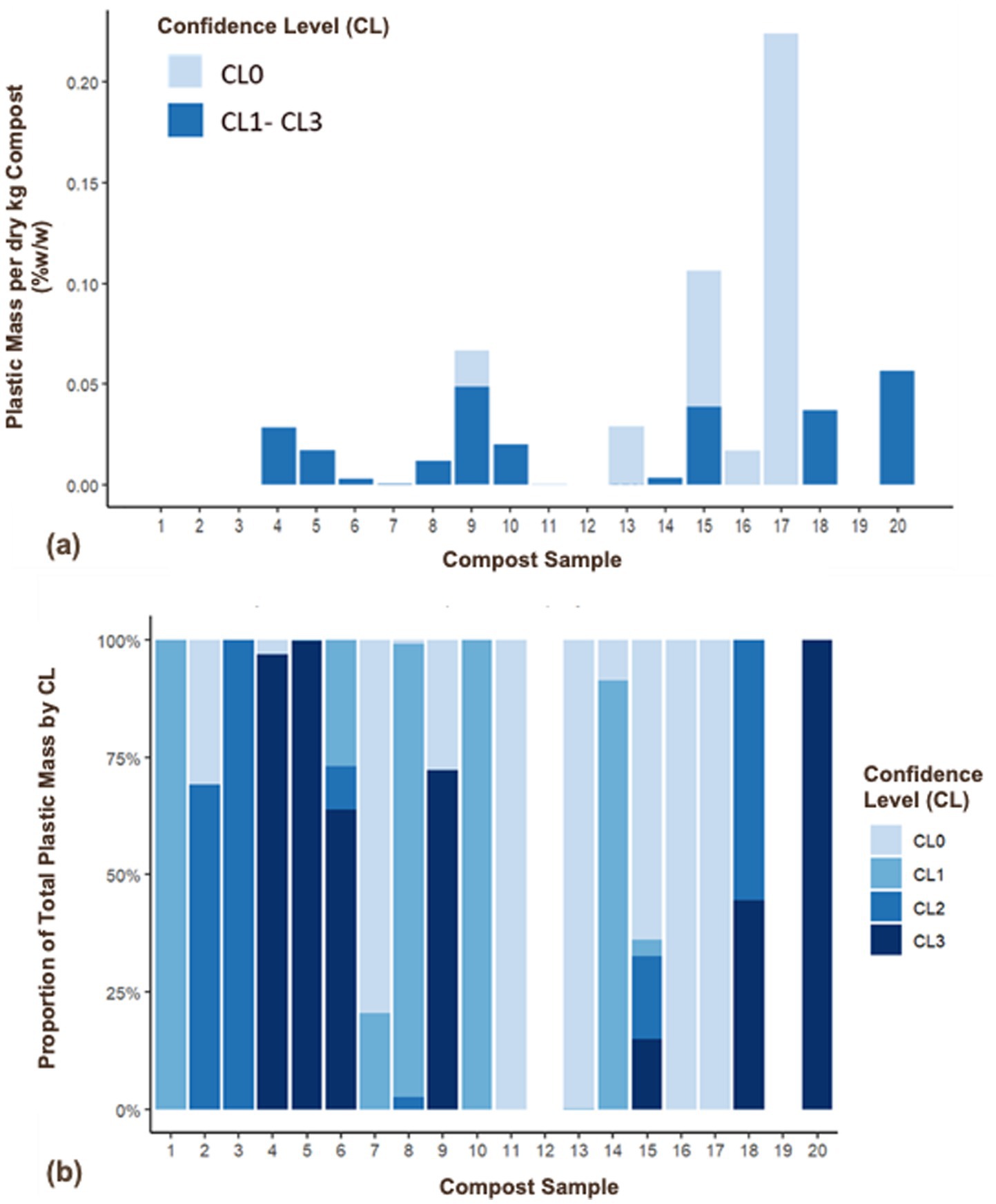
Figure 2. (a) Plastic mass per dry mass compost (% w/w, dry basis), all sizes with CL0 particles distinguished from CL1–CL3 particles, and (b) proportion of total plastic mass per dry kg by confidence level for each sample. Data are mean values.
3.3 Compost plastic characteristics
Plastic shapes varied across the three size fractions of considered, with relative abundances of films as highest and fragments as lowest on both a count- and mass-basis (CL1 to CL3). Particle counts illustrate the diversity of shapes (Figure 3A), while particle mass measurements show the relative contribution of shapes toward total plastic mass (Figure 3B). Fibers, which often had mass below microbalance detection limits (< 0.001 mg), contribute relatively little mass while fragments, which were less common but have higher masses, contributed more substantially to total plastic mass.
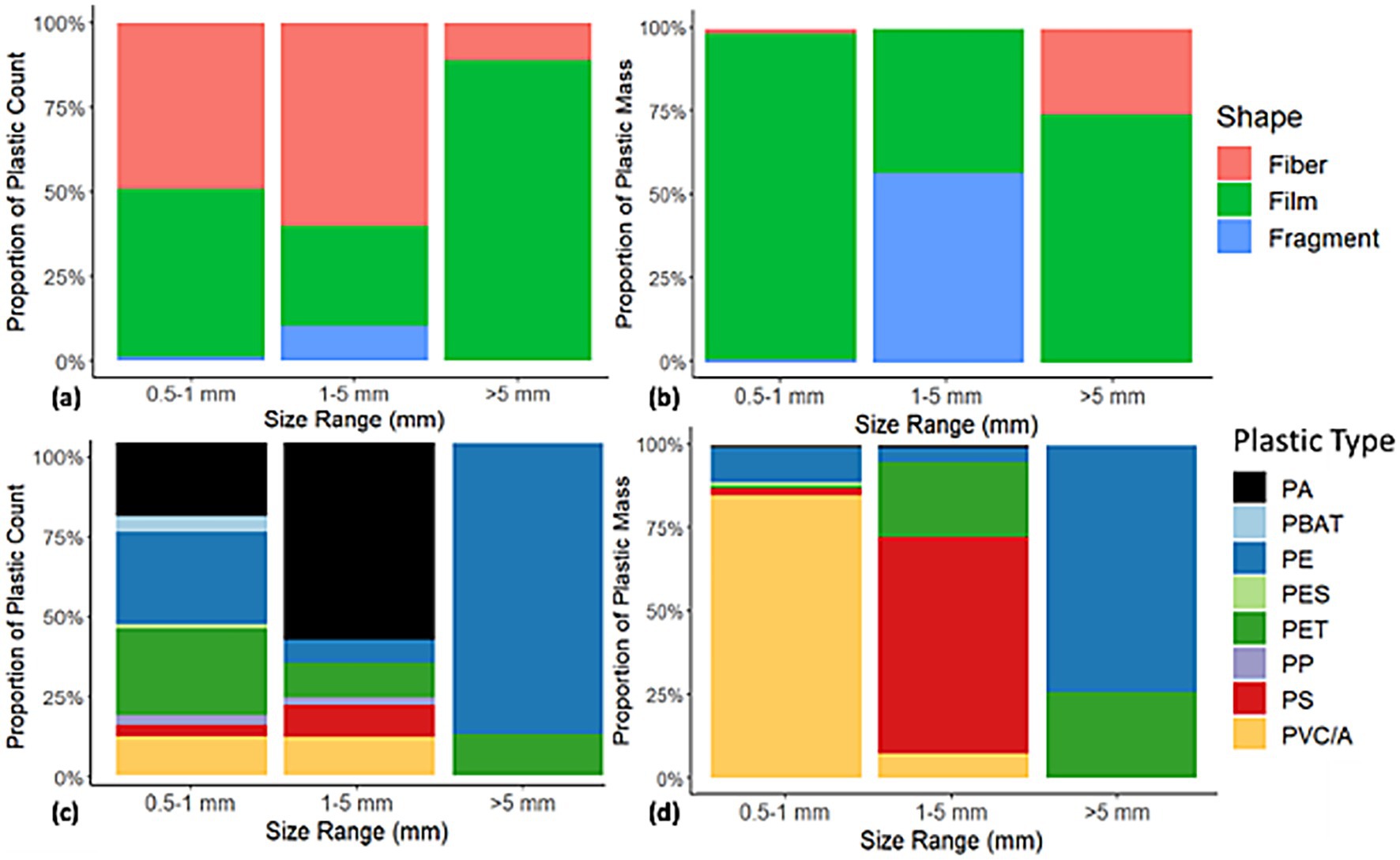
Figure 3. Percentage of (a) plastic shape counts by size fraction, (b) plastic shape mass by size fraction, (c) plastic type counts by size fraction, and (d) plastic type mass by size fraction. Only particles at confidence levels CL1 to CL3 are included. Plastic types are Polyamides (PA), Polybutylene Terephthalate (PBT), Polyethylene (PE) (including Low- and High-Density Polyethylene [LDPE, HDPE] and Ethylene-Vinyl Acetate), Other Polyesters (PES), Polyethylene Terephthalate (PET), Polypropylene (PP), Polystyrene (PS), and Polyvinyl Chloride (PVC) and Polyvinyl Acetate (PVA). Note that a single relatively heavy PVC particle influenced the 0.5-1 mm data shown in panel (d).
Eleven plastic types were detected across isolated particles. These can be grouped into eight main types of polymers: (1) Polyamides (PA), (2) Polybutylene Terephthalate (PBT), (3) Polyethylene (PE) (including Low- and High-Density Polyethylene [LDPE, HDPE]), and Ethylene-Vinyl Acetate (EVA), (4) Other Polyesters (PES), (5) Polyethylene Terephthalate (PET), (6) Polypropylene (PP), (7) Polystyrene (PS), and (8) Polyvinyl Chloride (PVC) and Polyvinyl Acetate (PVA). Plastic types varied greatly across the three particle size ranges on both a count and mass basis (CL1 to CL3). PA, PE, and PET were most abundant across the three size classes on a count basis (Figure 3c), while PVC, PS, PE, and PET were most abundant across the 3 size classes on a mass basis (Figure 3d). PA and PE have relatively low densities in comparison to PVC, and the prevalence of PS mass in the 1–5 mm size group (Figure 3d, middle column) may be due to the high proportion of heavier PS films and fragments.
4 Discussion
4.1 Methodological challenges and recommendations
As reported previously in Porterfield et al. (2023b), the similar methods we used here for composts were time consuming and less suitable for particles < 1 mm. Therefore, we suggest that future studies employing similar digestion, microscopy, and FTIR techniques focus on particles ≥ 1 mm. Doing so will undoubtedly miss smaller plastic particles < 1 mm that are present, which may constitute a substantial fraction of the overall count of plastic particles in a sample (Table 1). However, such particles, while potentially high in number, appear to make up a minor proportion of plastic mass in samples of compost (Table 1), as well as digestate and food waste (Porterfield et al., 2023b). Macroplastics can become microplastics (and then nanoplastics) through time (Thompson et al., 2004; Rillig et al., 2021). Thus, it is most prudent for monitoring and policy to focus on the bulk mass of plastic entering soils in organic amendments, as opposed to the total number of particles. Furthermore, our results in this study illustrate that plastic particle counts can be a poor predictor of plastic mass in compost.
Even in the 1–5 mm size class, visual isolation of plastic particles using microscopy was particularly challenging in this study of composts. This was likely due to a combination of relatively low plastic abundance and presence of substantial amounts of recalcitrant organic material and minerals after H2O2 digestion, including particles similar in appearance to plastic polymers. While inclusion of a density separation step has proven effective for some solid materials (Hanvey et al., 2017), it is usually not suitable for compost samples that contain abundant lightweight organic matter and as a result some researchers have explored alternatives including use of olive oil (Scopetani et al., 2020). Additionally, FTIR analysis of isolated putative microplastics was also frequently challenging, due to small particle size, particles consisting of multiple layered plastic polymers, and/or potential weathering of microplastics during composting or methodological steps (e.g., H2O2 digestion) (Nuelle et al., 2014). Our presentation of confidence levels (Figures 1, 2) was necessary in this study to communicate uncertainty in results given the challenges we encountered. We suggest that researchers measuring microplastics in organic residuals using microscopy and FTIR consider presenting confidence levels with their results. Moving forward, microscopy and FTIR-based screening methods for particles > 1 mm coupled with GC–MS methods capable of direct plastic mass measurements for smaller particles likely offers a robust monitoring approach (Wiesner et al., 2023).
4.2 Comparison with prior studies
Our plastic count results for twenty Vermont composts (0–1,201 particles per dry kg compost) were within range of mean values reported in other studies of composts derived at least in part from food waste (typically between 10’s to 1,000’s of particles per dry kg compost) compiled in a review by Porterfield et al. (2023a). This was also the case on a mass basis, with our range in this study (0 to 0.0561% w/w dry) comparable to the range in literature means (< 0.0001 to 0.1358% w/w dry) for composts derived from food waste (Porterfield et al., 2023a). Note that nearly all the compost studies included in the review by Porterfield et al. (2023a) occurred in Europe. Our results also overlap with ranges reported in more recent studies. For example, Ruffell et al. (2025) found that bulk and bagged composts in New Zealand from five facilities contained 1,060 to 2,990 and 480 to 2,610 particles per dry kg compost, respectively. Lu et al. (2025) found that plastic contamination in nine composts derived from food waste and other feedstocks in Australia ranged from 0.0016 to 0.0529% w/w dry on average. In another study from Australia, Kaur et al. (2025) found PE and PVC contamination in composts ranging from 0.033 to 0.217% w/w dry and 0.048 to 0.114% w/w dry, respectively, with additional conventional plastic contamination (excluding PE and PVC) ranging from below detection to 0.030% w/w dry on average. Some past studies have reported relatively high counts of plastic particles < 0.5 mm (Lu et al., 2025), however our similar % w/w results to those studies, our mass results by size class, and prior findings (Porterfield et al., 2023b) all suggest that abundant smaller plastic particles do not necessarily increase the mass of plastic contamination substantially.
When available, plastic polymer type information can help with source identification and inform ecotoxicity potential of a given substrate (Lambert et al., 2017). The polymers identified most prominently in our study (PE, PET, PA, PVC/A, and PS) are all commonly reported in surveys of environmental microplastics (Duis and Coors, 2016). Past studies of composts have reported detection of notable amounts of PE, PVC, PP, PET, poly(methyl methacrylate) (PMMA), polyethersulfone (PES), and PS (Kaur et al., 2025; Ruffell et al., 2025; Lu et al., 2025). Method limitations may lead to underestimation of plastic diversity in collected compost samples (Abel et al., 2021), especially in the 0.5 to 1 mm size class. Potential sources of these polymers include food packaging, plastic covers used on compost windrows, other plastic used on farms, and litter (Porterfield et al., 2023a). Based on prior characterization of food wastes (Porterfield et al., 2023a; Porterfield et al., 2023b), we hypothesize that food packaging is a primary source of the relatively high plastic mass contamination (≥ 0.02% w/w dry) in five of the compost samples included in the high food waste group in this study. This is also supported by the relatively high abundance and mass of films in our samples (Figure 3). While comparison of particle morphology with prior studies is challenging due to variation in characterization schemes, our observation that films and fragments (as opposed to fibers) accounted for most of the plastic contamination mass in composts is in line with prior reports (e.g., Ruffell et al., 2025; Lu et al., 2025).
4.3 Conclusion
This study is among the first in the United States to report measurements of microplastics and larger plastic particles in compost samples, including twenty composts in Vermont derived from feedstocks with and without food waste. Total plastic in compost for all three size fractions (0.5–1 mm, 1–5 mm, and > 5 mm) ranged from 0 to 1,201 particles per dry kg and 0 to 0.0561% w/w for particles at higher confidence levels (CL1, CL2, and CL3). No statistical differences were found between the groups with and without food waste feedstocks for plastic counts or mass in this initial study due to variability across composts and relatively low sample sizes for each group, although all five samples containing ≥ 0.02% w/w plastic on a dry mass basis were derived in part from food waste. Furthermore, plastic particle counts were a poor predictor of plastic mass in composts when considering all particles > 0.5 mm. PA, PE, and PET were most abundant on a count basis and PVC, PS, PE, and PET were most abundant on a mass basis. Compost samples include complex organic matrices that present multiple methodological challenges for microplastics analysis. We recommend that future studies employ a confidence level approach similar the one used here, and work toward monitoring methods that couple microscopy and FTIR-based screening methods for particles > 1 mm with GC-MS methods used to measure mass directly for smaller plastic particles.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author/s.
Ethics statement
The studies involving humans were approved by the Committees on Human Subjects Serving the University of Vermont and the UVM Medical Center. The studies were conducted in accordance with the local legislation and institutional requirements. The ethics committee/institutional review board waived the requirement of written informed consent for participation from the participants or the participants’ legal guardians/next of kin via a Waiver of Documentation of Consent under 46.117(c)(1).
Author contributions
SH: Visualization, Formal analysis, Writing – review & editing, Data curation, Writing – original draft, Investigation, Conceptualization, Methodology. KP: Writing – review & editing, Writing – original draft, Methodology, Investigation, Conceptualization. DN: Writing – review & editing, Conceptualization, Writing – original draft. MS: Writing – original draft, Supervision, Writing – review & editing. ER: Writing – original draft, Resources, Funding acquisition, Conceptualization, Supervision, Project administration, Methodology, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by Casella Waste Systems, Inc. (Sustainable Materials Management Graduate Fellowship for SH) and the Gund Institute for Environment at the University of Vermont (Gund Barrett PhD Fellowship for KP).
Acknowledgments
The authors acknowledge Tiffany Chin for assistance in the laboratory.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fsufs.2025.1617039/full#supplementary-material
References
Abel, S. M., Primpke, S., Int-Veen, I., Brandt, A., and Gerdts, G. (2021). Systematic identification of microplastics in abyssal and hadal sediments of the Kuril Kamchatka trench. Environ. Pollut. 269:116095. doi: 10.1016/j.envpol.2020.116095
Braun, M., Mail, M., Heyse, R., and Amelung, W. (2021). Plastic in compost: prevalence and potential input into agricultural and horticultural soils. Sci. Total Environ. 760:143335. doi: 10.1016/j.scitotenv.2020.143335
Cole, M., Lindeque, P., Halsband, C., and Galloway, T. S. (2011). Microplastics as contaminants in the marine environment: a review. Mar. Pollut. Bull. 62, 2588–2597. doi: 10.1016/j.marpolbul.2011.09.025
Colton, J. B., Knapp, F. D., and Burns, B. R. (1974). Plastic particles in surface waters of the northwestern Atlantic. Science 185, 491–497. doi: 10.1126/science.185.4150.491
Duis, K., and Coors, A. (2016). Microplastics in the aquatic and terrestrial environment: sources (with a specific focus on personal care products), fate and effects. Environ. Sci. Eur. 28:2. doi: 10.1186/s12302-015-0069-y
ECHA. (2020). Background document to the opinion on the annex XV report proposing restrictions on intentionally added microplastics (Report No. ECHA/RAC/RES-O-0000006790-71-01/F). Helsinki, Finland: European Chemicals Agency (ECHA). Available at: https://echa.europa.eu/documents/10162/b56c6c7e-02fb-68a4-da69-0bcbd504212b
Geyer, R., Jambeck, J. R., and Law, K. L. (2017). Production, use, and fate of all plastics ever made. Sci. Adv. 3:e1700782. doi: 10.1126/sciadv.1700782
Gui, J., Sun, Y., Wang, J., Chen, X., Zhang, S., and Wu, D. (2021). Microplastics in composting of rural domestic waste: abundance, characteristics, and release from the surface of macroplastics. Environ. Pollut. 274:116553. doi: 10.1016/j.envpol.2021.116553
Hanvey, J. S., Lewis, P. J., Lavers, J. L., Crosbie, N. D., Pozo, K., and Clarke, B. O. (2017). A review of analytical techniques for quantifying microplastics in sediment. Anal. Methods 9, 1369–1383. doi: 10.1039/C6AY02707E
Hartmann, N., Hüffer, T., Thompson, R., Hassellöv, M., Verschoor, A., Daugaard, A. E., et al. (2019). Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris. Environ. Sci. Technol. 53, 1039–1047. doi: 10.1021/acs.est.8b05297
Hidalgo-Ruz, V., Gutow, L., Thompson, R. C., and Thiel, M. (2012). Microplastics in the marine environment: a review of the methods used for identification and quantification. Environ. Sci. Technol. 46, 3060–3075. doi: 10.1021/es2031505
Horton, A. A., Walton, A., Spurgeon, D. J., Lahive, E., and Svendsen, C. (2017). Microplastics in freshwater and terrestrial environments: evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 586, 127–141. doi: 10.1016/j.scitotenv.2017.01.190
Jung, M. R., Horgen, F. D., Orski, S. V., Rodriguez, C. V., Beers, K. L., Balazs, G. H., et al. (2018). Validation of ATR FT-IR to identify polymers of plastic marine debris, including those ingested by marine organisms. Mar. Pollut. Bull. 127, 704–716. doi: 10.1016/j.marpolbul.2017.12.061
Kaur, S., Okoffo, E. D., Thomas, K. V., and Rauert, C. (2025). Unearthing the hidden plastic in garden compost. Sci. Total Environ. 973:179153. doi: 10.1016/j.scitotenv.2025.179153
Lambert, S., Scherer, C., and Wagner, M. (2017). Ecotoxicity testing of microplastics: considering the heterogeneity of physicochemical properties. Integr. Environ. Assess. Manag. 13, 470–475. doi: 10.1002/ieam.1901
Löder, M. G. J., Kuczera, M., Mintenig, S., Lorenz, C., Gerdts, G., Löder, M. G. J., et al. (2015). Focal plane array detector-based micro-Fourier-transform infrared imaging for the analysis of microplastics in environmental samples. Environ. Chem. 12, 563–581. doi: 10.1071/EN14205
Lu, H. C., Cao, W., Jones, S., Leusch, F. D., Slynkova, N., Trevorah, R., et al. (2025). Microplastic abundance in Australian processed organics: abundance, characteristics and potential transport to soil ecosystem. J. Environ. Manag. 375:124359. doi: 10.1016/j.jenvman.2025.124359
Masura, J., Baker, J., Foster, G., and Arthur, C. (2015). Laboratory methods for the analysis of microplastics in the marine environment: recommendations for quantifying synthetic particles in waters and sediments. NOAA Technical Memorandum NOS-OR&R-48.
Meixner, K., Kubiczek, M., and Fritz, I. (2020). Microplastic in soil–current status in Europe with special focus on method tests with Austrian samples. AIMS Environ Sci 7, 174–191. doi: 10.3934/environsci.2020011
Napper, I. E., Bakir, A., Rowland, S. J., and Thompson, R. C. (2015). Characterisation, quantity and sorptive properties of microplastics extracted from cosmetics. Mar. Pollut. Bull. 99, 178–185. doi: 10.1016/j.marpolbul.2015.07.029
Nizzetto, L., Futter, M., and Langaas, S. (2016). Are agricultural soils dumps for microplastics of urban origin? Environ. Sci. Technol. 50, 10777–10779. doi: 10.1021/acs.est.6b04140
Nuelle, M. T., Dekiff, J. H., Remy, D., and Fries, E. (2014). A new analytical approach for monitoring microplastics in marine sediments. Environ. Pollut. 184, 161–169. doi: 10.1016/j.envpol.2013.07.027
Porterfield, K. K., Hobson, S. A., Neher, D. A., Niles, M. T., and Roy, E. D. (2023a). Microplastics in composts, digestates, and food wastes: a review. J. Environ. Qual. 52, 225–240. doi: 10.1002/jeq2.20450
Porterfield, K. K., Scarborough, M. J., and Roy, E. D. (2023b). Organics recycling tradeoffs: biogas potential and microplastic content of mechanically Depackaged food waste. ACS Sustain. Chem. Eng. 11, 10422–10429. doi: 10.1021/acssuschemeng.3c01710
Posit Team (2025). RStudio: Integrated development environment for R. Boston, MA: Posit Software, PBC.
Rillig, M. C., Kim, S. W., Kim, T. Y., and Waldman, W. R. (2021). The global plastic toxicity debt. Environ. Sci. Technol. 55, 2717–2719. doi: 10.1021/acs.est.0c07781
Romeo, T., Pietro, B., Peda, C., Consoli, P., Andaloro, F., and Fossi, M. C. (2015). First evidence of presence of plastic debris in stomach of large pelagic fish in the Mediterranean Sea. Mar. Pollut. Bull. 95, 358–361. doi: 10.1016/j.marpolbul.2015.04.048
Ruffell, H., Pantos, O., Robinson, B., and Gaw, S. (2025). Quantification of microplastics in biowastes including biosolids, compost, and vermicompost destined for land application. Water Emerging Contaminants Nanoplastics 4:1. doi: 10.20517/wecn.2024.65
Ruggero, F., Gori, R., and Lubello, C. (2020). Methodologies for microplastics recovery and identification in heterogeneous solid matrices: a review. J. Polym. Environ. 28, 739–748. doi: 10.1007/s10924-019-01644-3
Schwinghammer, L., Krause, S., and Schaum, C. (2020). Determination of large microplastics: wet-sieving of dewatered digested sludge, co-substrates, and compost. Water Sci. Technol. 84, 384–392. doi: 10.2166/wst.2020.582
Scopetani, C., Chelazzi, D., Mikola, J., Leino, V., Heikkinen, R., Cincinelli, A., et al. (2020). Olive oil-based method for the extraction, quantification, and identification of microplastics in soil and compost samples. Sci. Total Environ. 733:139338. doi: 10.1016/j.scitotenv.2020.139338
Thompson, R., Olsen, Y., Mitchell, R., Davis, A., Rowland, S., John, A., et al. (2004). Lost at sea: where is all the plastic? Science 304:838. doi: 10.1126/science.1094559
Veerasingam, S., Ranjani, M., and Venkatachalapathy, R. (2021). Contributions of Fourier transform infrared spectroscopy in microplastic pollution research: a review. Crit. Rev. Environ. Sci. Technol. 51, 2681–2743. doi: 10.1080/10643389.2020.1807450
Veidis, E. M., LaBeaud, A. D., Phillips, A. A., and Barry, M. (2022). Tackling the ubiquity of plastic waste for human and planetary health. American J Tropical Medicine Hygiene 106, 12–14. doi: 10.4269/ajtmh.21-0968
VT DEC. (2021). Vermont certified food scrap management facilities. Vermont Agency of Natural Resources. Available online at: https://dec.vermont.gov/sites/dec/files/wmp/SolidWaste/Documents/Food-Scrap-Facilities.pdf
Wiesner, Y., Bednarz, M., Braun, U., Bannick, C. G., Ricking, M., and Altmann, K. (2023). A promising approach to monitor microplastic masses in composts. Front. Environ. Chem. 4:1281558. doi: 10.3389/fenvc.2023.1281558
Keywords: microplastics, compost, organics recycling, food waste, contamination
Citation: Hobson SA, Porterfield KK, Neher DA, Scarborough MJ and Roy ED (2025) Plastic contamination of composts derived from feedstocks with and without food waste. Front. Sustain. Food Syst. 9:1617039. doi: 10.3389/fsufs.2025.1617039
Edited by:
João Frias, Atlantic Technological University, IrelandReviewed by:
Francis Okori, Makerere University, UgandaHelena Ruffell, University of Canterbury, New Zealand
Copyright © 2025 Hobson, Porterfield, Neher, Scarborough and Roy. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Eric D. Roy, ZXJpYy5yb3lAdXZtLmVkdQ==
 Sarah A. Hobson1,2
Sarah A. Hobson1,2 Katherine K. Porterfield
Katherine K. Porterfield Deborah A. Neher
Deborah A. Neher Eric D. Roy
Eric D. Roy