- 1Centre for Health, Activity and Rehabilitation Research, School of Physiotherapy, University of Otago, Dunedin, New Zealand
- 2Department of Surgical Sciences, Otago Medical School, University of Otago, Dunedin, New Zealand
- 3The Health Boutique, Napier, New Zealand
- 4Biostatistics Centre, University of Otago, Dunedin, New Zealand
- 5Department of Anatomy, School of Biomedical Sciences, University of Otago, Dunedin, New Zealand
- 6Otago Medical School, University of Otago, Dunedin, New Zealand
- 7Department of General Practice, University of Otago, Christchurch, New Zealand
- 8Department of Anesthesiology, School of Medicine, University of California, San Diego, CA, United States
- 9Pain Research and Intervention Center of Excellence, Clinical and Translational Science Institute, College of Dentistry, University of Florida, Gainesville, FL, United States
Introduction: Knee osteoarthritis (OA) is a chronic secondary musculoskeletal pain condition resulting in disability, reduced quality of life, and high societal costs. Pain associated with knee OA is linked to increased sensitivity in sensory, cognitive, and emotional areas of the brain. Self-regulation training targeting brain functioning related to pain experience could reduce pain and its associated disability. Self-regulatory treatments such as mindfulness meditation (MM) and electroencephalography neurofeedback (EEG-NF) training improve clinical outcomes in people with knee OA. A feasibility clinical trial can address factors that could inform the design of the full trial investigating the effectiveness of self-regulation training programmes in people with knee OA. This clinical trial will evaluate the feasibility, safety, acceptability, experience and perceptions of the self-regulatory training programmes.
Methods: The proposed feasibility trial is based on a double-blind (outcome assessor and investigators), three-arm (MM usual care, EEG-NF + usual care and usual care control group) randomised controlled parallel clinical trial. Participants with knee OA will be recruited from the community and healthcare practices. A research assistant (RA) will administer both interventions (20-min sessions, four sessions each week, and 12 sessions over three successive weeks). Feasibility measures (participant recruitment rate, adherence to interventions, retention rate), safety, and acceptability of interventions will be recorded. An RA blinded to the group allocation will record secondary outcomes at baseline, immediately post-intervention (4th week), and 3 months post-intervention. The quantitative outcome measures will be descriptively summarised. The qualitative interviews will evaluate the participants' experiences and perceptions regarding various aspects of the trial, which includes identifying the barriers and facilitators in participating in the trial, evaluating their opinions on the research procedures, such as their preferences for the study site, and determining the level of acceptability of the interventions as potential clinical treatments for managing knee OA. Māori participant perceptions of how assessment and training practices could be acceptable to a Māori worldview will be explored. The interviews will be audio-recorded and analysed thematically.
Discussion: This trial will provide evidence on the feasibility, safety, and acceptability of the MM and EEG-NF training in people with knee OA, thus informing the design of a full randomised clinical control trial.
Introduction
Knee osteoarthritis (OA) is a chronic secondary musculoskeletal pain condition resulting in disability, reduced quality of life, and high societal costs (1, 2). Knee OA was among the top-ranked conditions in terms of the number of years lived with disability (3). Chronic pain in knee OA is often attributed to structural changes in joint structures. However, there is an apparent discordance between the nature and intensity of the pain and the degree of structural changes in the knee joints (4, 5). Pain associated with knee OA is linked to increased sensitivity in sensory, cognitive, and emotional cortices of the brain and psychological contributions (6–17). Self-regulation training targeting normalising brain functioning related to pain experience could improve clinical outcomes (18–24).
Mindfulness meditation training (MMT), a form of self-regulation training, ‘involves focussed attention to the changing sensations of the body (usually the breath) and non-reactive monitoring of arising sensory events’ (19). Several mechanisms that mediate pain relief following MMT have been identified (25). Neuroimaging studies confirm that MMT can modulate activity in the cortical areas processing sensory, cognitive, and emotional components of the pain experience. MMT improves mood, increases autonomic modulation, and engages non-opioid mechanisms that could mediate pain outcomes (26–38).
Electroencephalography neurofeedback (EEG-NF) is a biofeedback training designed to improve health outcomes. The EEG-NF training involves assisting people to learn to self-regulate the activity of specific brain areas that are involved in key functions related to pain perception and disease. In the EEG-NF training, a real-time feedback (reward) is provided when the participant's brain activity reaches the set threshold frequency level of the targeted cortical electrical activity (39–48). Evidence demonstrates a clinically significant reduction in pain following the EEG-NF training (49–55). NF also improves anxiety, emotional regulation, brain activity, and autonomic modulation, potentially mediating mechanisms of pain reduction (56–63). Pilot EEG-NF training targeting infraslow frequency bands demonstrated encouraging patterns for pain outcomes in people with chronic musculoskeletal pain (64, 65).
Both MMT and NF are stand-alone, promising self-regulatory interventions demonstrated to improve pain outcomes. However, evidence of the effectiveness of these two self-regulation training methods is lacking in people with knee OA, a common chronic secondary musculoskeletal pain condition. Although both interventions appear to rely on self-regulatory principles, the physiological and psychological mechanisms through which these two interventions improve pain outcomes may be different (63–69). Therefore, identifying the mechanisms for how these self-regulatory interventions improve outcomes can assist in further optimising protocols to enhance outcomes. Thus, the primary aim of the full clinical trial will be to assess the clinical effectiveness and cost-effectiveness of MMT + usual care and EEG-NF training + usual care against usual care for improving pain and functional outcomes in individuals with knee OA. The secondary aim of the full randomised clinical trial will be to identify the psychological and physiological mechanisms by which training reduces pain and improves function.
However, prior to conducting this full RCT, we propose conducting a feasibility trial to address the factors that could influence the design of the full clinical trial (64). The feasibility issues/questions that require understanding prior to conducting a full-powered RCT are as follows: Are mindfulness meditation and neurofeedback training feasible, safe, and acceptable interventions for people with knee OA? What are the participant recruitment, enrolment, adherence, and dropout rates? What are the adverse side effects of these two self-regulation-based training programmes? What are the participant's experiences of the treatment and research procedures, particularly Māori perceptions of how assessment and training practices in the trial are acceptable to cultural perspectives and values, including Te Ao Māori (a Māori worldview)? What modifications are required for the full trial? What sample size is required to conduct an adequately powered trial?
Therefore, the objectives of this feasibility randomised clinical trial are as follows:
• Objective 1: To determine the feasibility (i.e., rates of participant recruitment, enrolment, training compliance, retention/dropouts) of conducting the full randomised clinical trial.
• Objective 2: To examine the safety of the administered interventions.
• Objective 3: To explore the participants' perceptions regarding the study procedures, including the acceptability of interventions.
• Objective 4: To explore Māori perceptions of how assessment and training practices in the trial are acceptable to cultural perspectives and values, including Te Ao Māori (a Māori worldview).
• Objective 5: To derive the central tendency and variability of the clinical outcome measures to inform the sample size of a full clinical trial.
Research design and methods
This protocol was reported in accordance with the CONSORT statement on pilot/feasibility studies (69). This trial is prospectively registered in the Australian New Zealand Clinical Trials Registry (ANZCTR 12621001741875). The trial has been documented following the recommendations outlined in the Interventional Trials (SPIRIT) statement and the template for intervention description and replication (TIDieR) checklist (Table 1) (70).
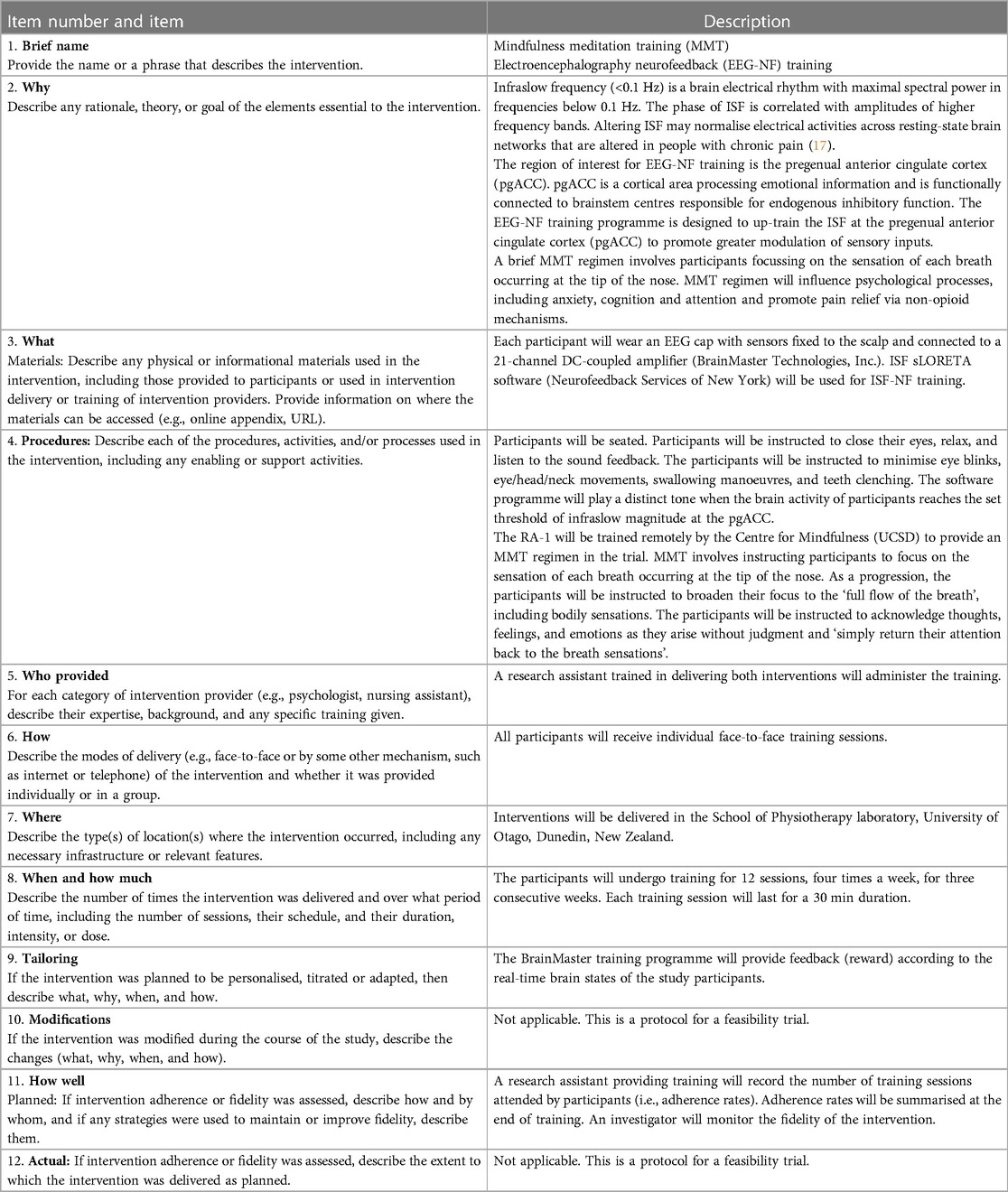
Table 1. Description of the EEG-NF and MMT, based on the template for intervention description and replication.
Study design
This feasibility study (71, 72) will be a double-blind (outcome assessor and all investigators), three-arm randomised control parallel trial with participants having an equal probability of being assigned to each arm. The study outcome measures will be collected at baseline, immediately post-intervention (4th week), and 3 months post-intervention.
Ethical approval
Ethical approval was obtained from the NZ Health & Disability Ethics Committee. The Ngāi Tahu Research Consultation was obtained. The School of Physiotherapy health and safety committee will act as an internal Data and Safety Monitoring Board.
Establishing whakawhanaungatanga (relationships) with Māori
Osteoarthritis is an increasing health condition among the Māori population. Improving the OA treatment will help in reducing health inequalities between Māori and non-Māori (17, 73–80). Combining our consultation with the Ngāi Tahu Research Committee, Māori researchers, and research work conducted by Māori on chronic pain, we will implement practices acceptable to a Māori worldview, thus being responsive towards the cultural and health needs of the Māori people. The clinical trial will be guided by a Māori-centred research approach (i.e., Kaupapa Māori research) that incorporates cultural considerations and provides an interpretive framework aligned to Māori customs (tīkanga) base. A Māori researcher (SA) with expertise in issues about kaumātua living with OA will oversee the implementation of tīkanga principles and administer culturally appropriate strategies to recruit Māori participants. We will adhere to Māori rituals of engagement, such as pōwhiri and hui, on Marae and wharenui (meeting house in Marae). This will involve presenting the background and procedures of the study, as well as the findings from previous work on lived experiences of pain among Māori. In addition, we will discuss culturally appropriate pain assessment tools for Māori with OA, as well as involve their family and whānau in the community. Hui will be carried out face-to-face (kanohi-ki-te-kanohi) to enable the community to see who the researchers are and include whanaungatanga (strengthening connections). Engaging participants with respectful behaviour closely related to Māori customs, such as ‘aroha’ and ‘manākitanga’. The priority will be getting the community talking about interventions for reducing pain, improving function, and strengthening referral networks of Māori living with OA into the research. A Māori researcher will conduct interviews with Māori participants. In hui and interviews, as well as assessment and intervention sessions, the importance of whānau support means that Māori participants can bring families and friends for the assessment and treatment sessions. Furthermore, koha (such as food, drink, and a voucher) will be offered as a mark of appreciation at each interview. In prioritising mana (respect) for the participants, Māori culture considers the head sacred (‘he tapu te upoko’). Therefore, permission must be obtained at every EEG-NF session before touching the participant's head. Finally, the research outcomes will be fed back to the community to offer nurturing guidance (awhi) and community relationship building. In this way, the research process and outcomes form lasting relationships beneficial to both the research and the community.
Study eligibility criteria
The inclusion criteria of the study are as follows: Adults (45–85 years) will be eligible to participate if they meet the clinical criteria for the diagnosis of knee OA according to the guidelines established by the American College of Rheumatology (ACR) and with knee pain that persisted for longer than 3 months, (81). The exclusion criteria of the study are as follows: Recent soft tissue injuries of the knee (e.g., ligaments, muscles, meniscus, tendon) in the last 3 months, infective/inflammatory arthritis, scheduled for joint replacement surgery within the next 4 months, joint injections (hyaluronic acid) in the last 6 months, joint injections (steroids) in the last 3 months, brain injury or diseases (e.g., stroke, multiple sclerosis, Parkinson's disease), spinal cord injury or diseases, nerve injuries or neuropathy in the legs, migraine or recurrent headaches, cognitive illness (dementia)—any difficulty thinking, reasoning, or remembering, major psychiatric illnesses and former neurosurgical procedures of the brain, and recent/current pregnancy.
Study location and settings
Recruitment strategy
We will recruit participants from the wider Dunedin community. Community newspapers [The Star (a free newspaper published weekly) and the Otago Daily Times (a paid newspaper published daily)] will periodically publish study advertisements, reaching out to the urban area of Dunedin city, its suburbs, and other nearby villages. Sponsored Facebook adverts will also be implemented. Targeted advertisements will be published in community organisations, including arthritis advocacy groups and community-based exercise classes. Patients attending primary healthcare practices based in Dunedin will also be invited to participate.
The participants in the experimental groups will get a reimbursement of $200 in the form of supermarket/travel vouchers as a compensation for their time dedicated to this study. It is anticipated that each participant will contribute approximately 20 h to the study, including three 2-h assessments [baseline, post-intervention (4th week), and 3 months post-intervention], 12 1-h training sessions, and 1 h of a qualitative interview.
The participants in the usual care group will get a reimbursement of $80 in the form of supermarket/travel vouchers as a compensation for their time dedicated to this study. It is anticipated that each participant will contribute approximately 8 h to the study, which includes three 2-h assessments and 1 h of a qualitative interview.
Māori sampling
The purposeful sampling strategy will be conducted with people identifying as being of Māori ancestry and ethnicity that meet our sampling criteria. Key informants will initially be identified through the Māori researcher and research teams' whānaungatanga (networks and relationships) and Māori healthcare providers (73). The snowball sampling method will involve asking the initial respondents to suggest others, precisely who they know in the target group and whom to contact and invite to take part in the research. A Māori-specific participant information sheet will clearly outline the purpose of the study and emphasise that the information provided by Māori participants, their family, and their whānau could contribute to the acceptability of the study as a potential clinical treatment for the management of knee OA from a Māori perspective. A Māori investigator will train the research staff on Tikanga Māori to create a safe and respectful atmosphere for all participants during the trial.
Screening and enrolment
Volunteers who expressed interest in participating in the study will undergo initial screening via a digital survey or phone. The participants will undergo screening for cognitive involvement at the beginning of the baseline assessment, and if found eligible, they continue to complete the baseline assessment. The following procedures will be conducted at the baseline assessment.
Baseline assessment
Written informed consent will be obtained, and the participants will be subjected to a baseline assessment to complete the questionnaires (age, sex, ethnicity, education, income, employment status, OA duration, pain severity, interference and quality, co-morbidity, psychological states, medication use, and other treatments for OA pain-management activities) (82–84) and undergo anthropometric measurements.
Allocation
After the baseline assessment, the participants will be allocated into one of the three study groups with equal probabilities. Blocks of unequal length, with the measurements and the possibilities of these lengths unknown to other investigators, will be used to promote allocation concealment. The senior biostatistician, using non-informative group allocation codes, will generate the allocation sequence using a standard computer software.
Allocation concealment
The allocation of groups will be concealed using opaque envelopes to conceal the sequence until interventions are assigned. The senior biostatistician will prepare the envelopes and provide them to the assistant research fellow (ARF). The ARF will give the opaque sealed envelope to the participants following the baseline assessment. The participants open the envelope to reveal the group they have been allocated to after the baseline assessment. The participants inform the ARF about the group, and the ARF administers one of the active interventions to the participant. If a participant is identified to be allocated to the usual care control group, they would be advised to continue managing their health condition as usual and attend the follow-up assessment sessions.
Blinding
The outcome assessor and all investigators will be blind to the group allocation. The participants and the intervention provider will not be blinded to the intervention because the content will be evident. The biostatistician will be blinded to the study groups until all planned analyses are completed, thus minimising bias during data analysis. All the participants will be requested to keep their treatment allocation group private from the outcome assessor during the duration of the study. This information is detailed and indicated to the participants within the study information sheet.
Interventions
Following the baseline assessment and group allocation, the participants will receive one of the active interventions or the usual care control group. The number of sessions and duration for both interventions will be identical. A trained research assistant will administer both active interventions. A research assistant will be trained to administer the study interventions. Both interventions will consist of 12 20-min sessions which will be administered in four consecutive sessions/week for three successive weeks. The interventions described in this study adhered to the Template for Intervention Description and Replication guide.
MMT
An MMT regimen (85) will be used. An RA will be trained remotely by the Centre for Mindfulness, University of California San Diego, to conduct the intervention. During each session, the participants will be instructed to focus on the sensation of each breath occurring at the tip of the nose. On progression, the participants will be instructed to broaden their focus to the ‘full flow of the breath’, including bodily sensations. We will instruct the participants to acknowledge thoughts, feelings, and emotions as they arise without judgment and ‘simply return their attention back to the breath sensations’.
EEG-NF training
An RA will be trained to administer the EEG-NF training using the BrainMaster Inc. system (86). Comby EEG Caps with sensors (Ag/AgCl) are used to record EEG signals (Figure 1). The participants will be instructed to minimise eye blinks, eye/head/neck movements, swallowing manoeuvres, and teeth clenching. The software programme will play a distinct tone when the brain activity of the participants reaches the set threshold of infraslow (0.0–0.1 Hz) frequency band at the pregenual anterior cingulate cortex (pgACC). The participants will be seated and instructed to close their eyes, relax, and listen to the sound feedback. The reward threshold will be adjusted in real time between 60%–80%, which means that the sound feedback will be delivered 60%–80% of the time.
We will use the EEG- NF programme administered in our pilot studies in people with chronic musculoskeletal pain, including knee OA (64, 65). Infraslow frequency (<0.1 Hz) is a brain electrical rhythm with maximal spectral power in frequencies below 0.1 Hz. Influencing the ISF may normalise electrical activities across resting-state brain networks that are altered in people with chronic pain (87–90). The pgACC is the chosen region of interest (ROI) for the training due to its connectivity with the periaqueductal grey matter (PAG), which promotes endogenous pain inhibition (17, 90, 91). Up-training ISF at the pgACC improved the pain outcomes in people with chronic pain (65). Therefore, the EEG-NF training programme was designed to up-train the ISF at the pgACC to improve pain outcomes through promoting greater sensory modulation, altering attention and cognitive controllability of pain.
The participants in the control group who are receiving usual care will continue to receive treatments as usual. However, they will refrain from practising meditation during the study period. Following the trial completion, they will be offered to receive one of the trainings.
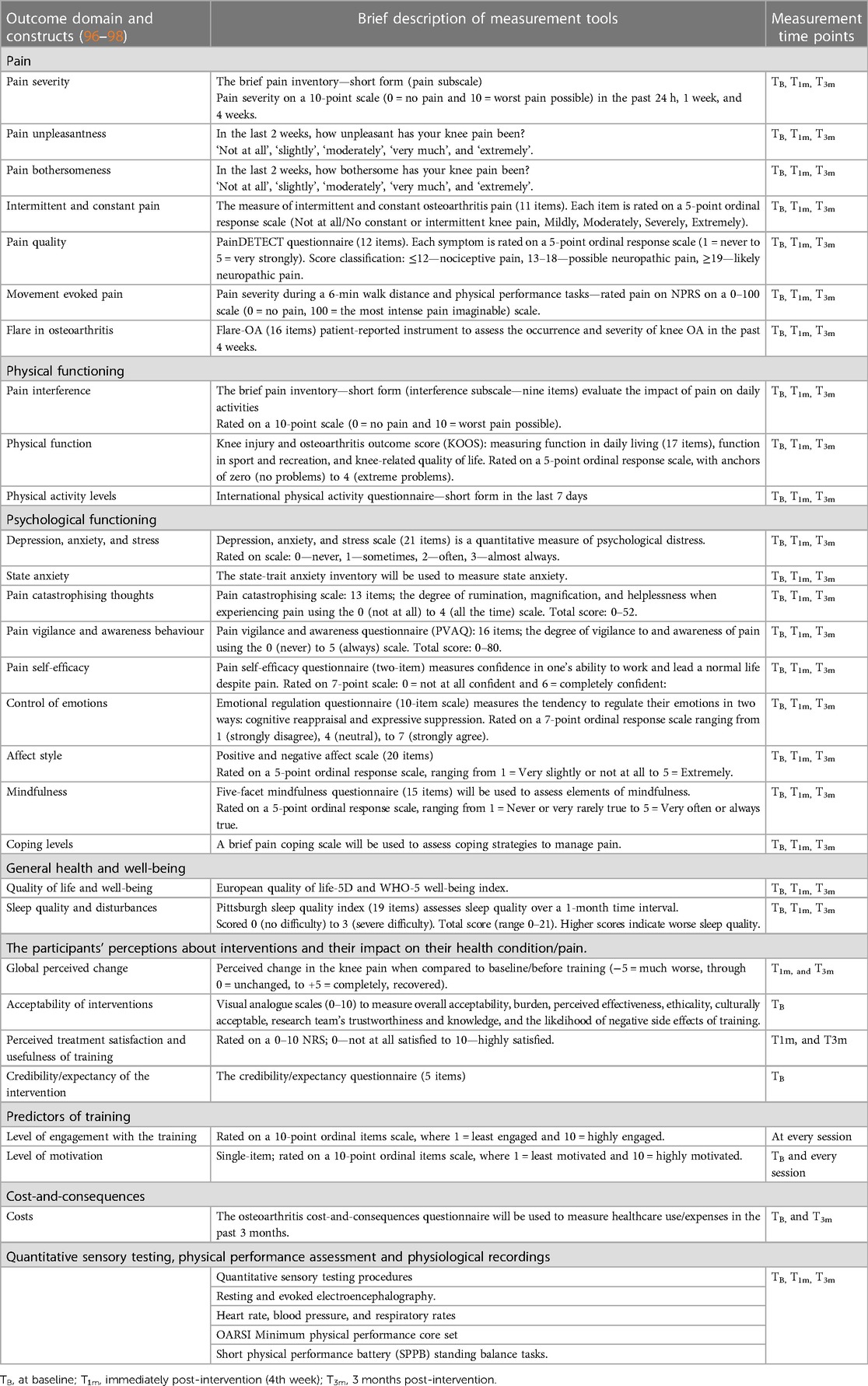
Outcome measures
The assistant research fellow will collect primary outcome measures throughout the study period. All secondary outcome measures will be collected at baseline, immediately post-intervention (4th week), and 3 months post-intervention by a research assistant (RA-2) blinded to group allocation. The RA will be adequately trained to collect quantitative sensory testing procedures, electrocardiogram, and electroencephalography recordings (Table 2).
Primary outcomes
Feasibility data
The feasibility data (71) will be collected by the assistant research fellow.
• Recruitment rate and enrolment: The percentage of the number of participants recruited from the total number of participants screened over 1 year will be calculated. We aim to recruit 20 participants in each arm of the trial over 1 year in a single centre.
• Compliance rate to training sessions: A research assistant providing training will record the number of training sessions attended by the participants (i.e., adherence rates). The adherence rates will be summarised as percentages. The reasons for non-compliance (e.g., adverse effects) will be documented. A targeted compliance rate of 80% will be considered acceptable.
• Participant retention rate (dropout rates): The percentage of the allocated participants in each group who completed the follow-up assessment will be measured. A targeted retention rate of 80% will be considered acceptable.
Safety data
The training administrator will record symptoms using the Discontinuation-Emergent Sign and Symptom (DESS) (93) checklist. The 43-item checklist includes emotional, behavioural, cognitive, and physical symptoms, and the participants will be required to compare their current status with their status prior to the training or the previous session.
Qualitative study
An assistant research fellow will coordinate with the investigators in conducting semi-structured in-depth interviews following the intervention to evaluate participant experiences in the trial (94, 95). As they finish the trial, the participants will be asked if they would consider this additional interview and be enrolled sequentially until numbers are met. The interview will explore experiences and perceptions regarding interventions based on the theoretical framework of acceptability. These would be recorded with the consent of the participants. The aims of this interview include exploring the participants’ experiences regarding the barriers and facilitators in participating in the trial. In addition, the interview aims to assess the acceptability of the research procedures, including the intervention, preferences for the study site, the perceived value of the study, and the acceptability of interventions as potential clinical treatments for managing knee OA. Furthermore, the interview seeks to explore the perspectives of Māori people on assessment and treatment practices that align with their cultural worldview (95). Māori is treated as an important subgroup and are selected and interviewed separately. We aim to interview five people per group with a purposeful sample of Māori participants from each group. The participants in the usual care group will also be interviewed to explore their experiences participating in the trial, including the acceptability of the assessment procedures.
Secondary outcomes
Table 2 presents the outcome domain and constructs, together with a brief description of the measurement tools used to measure the constructs and the measurement time points (96–127). All outcomes are well-established, reliable, and validated constructs in people with persistent pain (predictors of pain and disability) and are recommended for clinical trials of chronic pain and knee osteoarthritis (96–98, 120). Each assessment session will last for a maximum of 2 h, including completing the questionnaires and undergoing testing.
Outcome assessor and blinding
A research assistant who will be blinded to the groups will collect the secondary outcomes at baseline, immediately post-intervention, and 3 months post-intervention. The effectiveness of blinding will be measured. The self-report questionnaires will be administered using the Qualtrics data management system. The participants will complete the questionnaires in two parts: at home and in person during the assessment sessions.
Pain and physical function measures will be measured using validated questionnaires in people with knee OA (99–102). All participants will perform the minimum physical performance core set (sit-to-stand, 30-s chair stand test, and walking short distances) as recommended by the Osteoarthritis Research Society International (102). A short physical performance battery (SPPB) will be administered that includes standing balance tasks. Pain intensity during each physical performance will be recorded using a numerical 0–100 numeric rating scale (0 = no pain, 100 = the most intense pain imaginable) (102, 132): All participants will be asked to perform a 6-min walk test (6MWT) to derive the Sensitivity to Physical Activity (SPA) index. The participants will be asked to rate their discomfort before the 6MWT and every minute of walking. The SPA index will be derived by subtracting baseline pain ratings from their greatest pain ratings for each trial. The European Quality of Life-5 Dimensions scale will be used to assess health-related quality of life (104). Depression, anxiety, and stress will be recorded, as well as the occurrence of pain catastrophising thoughts, pain self-efficacy, positive and negative affect, emotion regulation, and elements of mindfulness and coping levels (106–112). Sleep quality and physical activity levels will be measured. Following the training, the participants' perceptions of change (−5 = much worse, through 0 = unchanged, to +5 = completely recovered) in their symptoms will be measured using the global rate of change scale (99) Visual analogue scales (0–10) will be used to measure the overall acceptability, burden, perceived effectiveness, ethicality, culturally acceptable, the research team's trustworthiness and knowledge, and the likelihood of negative side effects of training. The credibility/expectancy questionnaire will measure the treatment expectancy and rationale credibility of the interventions tested (113, 114). The level of engagement with the training sessions will be recorded using a 10-point ordinal items scale, where 1 = least engaged and 10 = highly engaged—level of motivation. For the EEG-NF group, an adapted version of the Current Motivation-Brain Computer Interference (QCM-BCI) questionnaire will be used to assess the level of motivation for participation in the training (114). The Osteoarthritis Cost-and-Consequences questionnaire will be used to record healthcare use/expenses during the 3-month period prior to the study and the 3-month period post-intervention (105).
Cardiovascular measures
The resting heart rate and beat-to-beat R-R interval will be measured using a Polar V800 HR monitor and a Polar H10 chest Pro Strap for a duration of 7 min, with a resolution of 2 ms. Blood pressure and respiratory rate will be measured. Raw heart rate and R-R interval time series data will be downloaded from the Polar flow software for further processing and analysis. Kubios software will be used to derive time and frequency domains of heart rate variability (HRV). Peripheral oxygen saturation will be monitored and recorded (123, 124).
Quantitative sensory testing procedures will be administered following the guidelines (115–122).
An algometer (AlgoMed, Medoc, Ramat Yishai, Israel) will be used to measure three trials of pressure pain threshold (PPT) over the two regions (index knee and non-dominant wrist) in random order. The mechanical temporal summation (MTS) procedure will be assessed twice using a nylon monofilament (Semmes monofilament 6.65, 300 g) under two contexts: at rest and during EEG recordings. Ten repetitive contacts will be delivered at 1 Hz. The participants rate the pain severity on an 11-point NPRS (0 = no pain to 100 = extreme pain) immediately after the first contact and rate the greatest pain intensity they experienced during 10 contacts. MTS is the difference between the NPRS scores after the first contact and the greatest pain scores. The average of two trials will be calculated. MTS will also be administered while EEG is being recorded. Pain intensity (NPRS 0–100) will be recorded following a single contact, and then the MTS will be administered in the following order: three trials of 10 contacts with 20 s rest period between trials. The overall pain intensity (NPRS 0–100) experienced during the three trials will be recorded following the completion of EEG recordings.
The conditioned pain modulation (CPM) procedure will be administered according to the published recommendations (98, 115) to measure the efficiency of the endogenous pain modulatory system. The conditioning stimulus involves the participants submerging their dominant hand in a cold water (−5°) bath for 2 min (maximum period) or until it is too uncomfortable. Pain intensity during the conditioning stimulus will be recorded on a 11-point NPRS (0 = no pain to 100 = extreme pain) at every 15 s intervals (98). The test stimulus involves measuring the suprathreshold PPT (pain40) at the tibialis anterior muscle over the non-dominant leg region. Two PPT (pain40) trials prior to exposure to the conditioning stimulus and three trials of PPT (pain40) trials at 30, 60, and 90 s following the conditioning stimulus will be recorded. The CPM will be determined by calculating the percent change score for each time point. Heart rate and blood pressure will be monitored for safety reasons.
For the collection, processing, and analysis of EEG data (125–127), the SynAmps RT Amplifier (Compudemics Neuroscan) will be used to capture resting-state EEG data for 10 min while the participant is seated in an upright position in a quiet room with closed eyes. The raw EEG signals will be processed according to our previous work (125–127). Exact low-resolution brain electromagnetic tomography (eLORETA) software will be used to estimate the intracerebral electrical sources in the following frequency bands: infraslow (0.01–0.10 Hz), slow (0.2–1.5 Hz), delta (2–3.5 Hz), theta (4–7.5 Hz), alpha1 (8–10 Hz), alpha2 (10.5–12 Hz), beta1 (12.5–18 Hz), beta2 (18.5–21 Hz), beta3 (21.5–30 Hz), and gamma (30.5–44 Hz). The current source density (CSD) and functional connectivity (FC) will be calculated for the selected regions of interest (ROIs), namely, pregenual, sub-genual, prefrontal, cingulate, insular, and somatosensory cortices. MM and NF interventions have been studied previously and have demonstrated altering the activity of the ROIs chosen in this trial. The changes in the CSD of the selected ROIs and FC between ROIs will be analysed within and between groups.
Quantitative data analyses plan
Descriptive statistics will be derived for primary and secondary outcome measures. The sample size estimation was not determined for primary outcomes since it is a feasibility trial. However, we aim to recruit a sample (20 participants per group, 60 in total) in 1 year in a single centre to provide sufficient data for assessing adverse events, feasibility outcomes, identifying operational issues, and estimating variability estimates of clinical pain outcomes.
In the full clinical trial, linear mixed models will be used to examine the differences in changes in the pain and physical function between groups to determine the effectiveness of interventions against usual care based on the randomised group (i.e., using an intention-to-treat approach). Group–time interactions will be used to assess between-group differences at 1 and 3 months following the intervention, with 3 months being the primary endpoint. Potential explanatory factors (which could include age, sex, BMI, duration, baseline pain severity, and depression scores) will be adjusted in the analysis. In this feasibility study, we will still perform these analyses (with no stratification or competing exposure variables included) to assess the study protocol, focusing on the uncertainty in estimated effects, not statistical significance. The following parameters will be used to determine the sample size in the full clinical trial: standard deviations (SD) and correlations between repeated measures of outcome measures derived from the feasibility trial. The full trial will have 80% power to detect a meaningful difference in the primary outcome (i.e., pain severity at 3 months). The minimal clinically important difference for pain severity (NPRS) is 1.8 using a two-sided (0.05) significance level. An attrition rate informed by the results of this feasibility study will be incorporated.
Qualitative data analysis (72): The interviews will be audio-recorded and fully transcribed. The General Inductive Approach will guide the analysis. The qualitative findings will provide important information regarding recruitment and retention in the trial and the acceptability of interventions. The results of the qualitative study will be published separately.
Māori analysis (70, 128–131): The journal and the interview data will be subjected to qualitative and inductive thematic analysis and Māori analysis, field notes collected by the Māori researcher, and interviews with Māori participants to ensure that the study reflects the values, beliefs, and cultural practices of the Tangata Whenua community towards broader objectives for Māori development. This will support the researcher in identifying factors related to the perceptions of the Māori participants, including the study site and setting, the overall value of the study, and the level of acceptability of interventions as potential clinical treatments for managing pain associated with knee OA. Overall, the interviews will provide deeper insights into whether assessment and training practices could be acceptable to a Māori worldview.
Criteria for termination of the study
If any unexpected serious adverse medical event or other incidents occur, the study or one of the arms of the trial will be discontinued. A serious adverse medical event is defined as any occurrence or effect that is life-threatening, including death, requiring hospitalisation, and significant disability. These medical events are considered very unlikely in this study due to the nature of the interventions.
Discussion
Self-regulatory interventions can improve health outcomes in people with chronic pain (20, 22). The proposed feasibility clinical trial will provide evidence on the feasibility, safety, and acceptability of self-regulatory interventions such as mindfulness meditation training and electroencephalography neurofeedback training in people with knee OA. It is essential to establish the feasibility of conducting a randomised control trial to investigate the effectiveness of these novel interventions for managing chronic secondary musculoskeletal pain. Therefore, a feasibility trial has been designed to assess the factors that could influence the conduct of a fully powered clinical trial.
The recruitment rate is one of the primary outcomes of this feasibility trial. Based on our previous experience in EEG-NF studies (56, 64, 65), we anticipate a favourable response from the community of people with knee OA living in an urban centre in this trial. However, predicting the recruitment rate/enrolment in this trial is not straightforward. Several factors can contribute to a less-than-optimal recruitment rate in this trial. Some of these factors that may affect recruitment include the presence of concurrent trials recruiting people with knee OA; the biomedical beliefs (wear and tear) in people with knee OA (135) could preclude participation in a trial focused on mind–body interventions such as mindfulness meditation; the potential of planned study advertisements and community engagements in reaching the population of interest, including the Māori and Pacific populations, and the anticipated level of acceptability and perceived usefulness of these self-regulatory brain-based interventions by the community of people with knee OA (64) are also important considerations. Moreover, this trial involves a significant time commitment over a short period, which may be a barrier to participation, affecting recruitment rates. Moreover, other barriers, such as parking availability and time away from work/family/childcare, can influence participation rates (133, 134). As hypothesised, good compliance and retention rates (80%) in the active training groups are expected.
Improving the treatment of knee osteoarthritis and access to treatments will help reduce health inequity between the Māori and non-Māori population (128). A Māori investigator will apply a Māori-centric approach to research, drawing on Kaupapa Māori theory (130). The proposed clinical trial will foster whakawhanaungatanga (building relationships) with Māori stakeholders and incorporate practices aligned with a Māori worldview, ensuring culturally relevant and sensitive interventions. Qualitative interview data will guide researchers in developing and testing culturally sensitive interventions in a future clinical trial (95), including addressing the barriers to participating in a clinical trial, thus addressing the specific needs of the Māori community in the New Zealand context. There is still a risk of inadequate representation of Māori in this trial despite this clinical trial being designed to implement culturally appropriate study procedures.
Since it is a feasibility clinical trial (137), and the sample size for this trial was not estimated, the secondary clinical outcomes were not powered to detect treatment effects. However, as supported by previous literature (64, 65), positive trends in the secondary outcomes, including pain severity, interference, and physical function, are anticipated in people undergoing active training. We hypothesise that the up-training pgACC training protocol will increase the current density across the frequency spectrum in the pregenual anterior cingulate cortex in people undergoing neurofeedback training (56). We hypothesise a reduction in the somatosensory cortex activity following mindfulness meditation training, whereas greater activation at the rostral anterior cingulate, anterior insula, and orbitofrontal cortices is expected. We also hypothesise a reduction in the mechanical temporal summation pain scores, an increased pressure pain threshold, and positive conditioned pain modulation responses in the active training groups compared with those in the control group. The dosage of interventions tested in this trial was similar to that of previous trials, but it may not be enough to demonstrate long-term clinical outcomes (64, 65). A future fully powered clinical trial could consider having booster training sessions to assess medium- to long-term training effects on clinical outcomes.
Estimating a range of side effects (minor to adverse events) associated with exposure to the proposed self-regulatory training procedures is critical. We will record symptoms weekly using the Discontinuation-Emergent Sign and Symptom checklist (93). Based on the previous pilot trial, we observed no significant adverse events associated with NF training (64, 65). The brief mindfulness meditation protocol used in this trial has been thoroughly investigated in experimental acute pain conditions and people with pain. This is the first trial that will provide signs and symptoms that may be associated with this specific brief mindfulness meditation training protocol used in this trial (136).
The experienced research team trained the research assistants in administering the assessment procedures. The research team has trained the research assistant to provide training and help in troubleshooting as it arises. The trained RAs will offer training as reported in the TIDieR checklist. An expert investigator will assess the fidelity of the NF intervention administration (138). A clinical psychologist associated with the Centre for Mindfulness, University of Southern California trained the research assistant to offer MM to the participants. The MM training protocol is scripted and was adapted slightly for cultural sensitivity by the Māori investigator; hence, the fidelity assessment of delivering MM may not be essential.
The biopsychosocial outcome measures in this trial have been selected based on the recommended core outcomes for clinical trials of chronic pain (97, 98). In addition, a range of physiological measures were used alongside the clinical outcomes to explore the potential mediating mechanisms contributing to the effect of intervention (120). A future fully powered clinical trial could assess the psychological and physiological markers as mediators and moderators of treatment effects. Due to a range of outcome measures utilised in the trial, we anticipate that the participants will face a burden in terms of completing the questionnaires and undergo test procedures. However, this issue could be addressed by scheduling for some questionnaires to be completed at home and administering specific questionnaires in person (134).
The neurofeedback training protocol in this trial has been previously used and demonstrated a clinically meaningful descriptive trend in the clinical outcomes (65). High-quality evidence (55) suggests that EEG-NF training effectively induces clinically meaningful analgesia in people with chronic pain. The observed clinical effect following NF training could result from active ingredients, such as NF-induced alterations in brain area activity and the placebo effects associated with NF training (e.g., wearing a cap or listening to auditory feedback). Therefore, having a placebo group (wearing an EEG cap only) is not required as this is a feasibility trial assessing the clinical effectiveness of NF training against mindfulness meditation and usual care. In our trial, we believe that the active ingredient (change in the brain activity) induced by the EEG-NF training protocol and the placebo effects would collectively produce the treatment effects.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics statement
Ethical approval was obtained from the NZ Health & Disability Ethics Committee. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
RM: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. DA: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Writing – original draft, Writing – review & editing, Visualization. SA: Conceptualization, Funding acquisition, Investigation, Methodology, Supervision, Validation, Visualization, Writing – review & editing, Data curation, Formal Analysis, Project administration, Writing – original draft. AG: Funding acquisition, Investigation, Methodology, Resources, Validation, Writing – review & editing, Conceptualization, Data curation, Formal Analysis, Writing – original draft, Project administration. JM: Investigation, Methodology, Validation, Writing – review & editing, Project administration, Visualization. LW: Methodology, Project administration, Visualization, Writing – review & editing, Funding acquisition, Investigation, Resources, Supervision, Validation. AS: Data curation, Investigation, Validation, Writing – review & editing, Project administration. DJ: Data curation, Investigation, Project administration, Resources, Supervision, Writing – review & editing, Validation, Visualization. BH: Investigation, Methodology, Writing – review & editing, Funding acquisition. FZ: Funding acquisition, Investigation, Methodology, Supervision, Validation, Visualization, Writing – review & editing, Resources. RF: Conceptualization, Investigation, Methodology, Validation, Writing – review & editing, Funding acquisition, Visualization. DD: Conceptualization, Investigation, Methodology, Resources, Writing – review & editing, Funding acquisition, Software, Supervision, Validation, Visualization.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article:
Health Research Council of New Zealand (Feasibility Study Grant 21/616).
Acknowledgements
The authors would like to thank Mark Smith, who owns Neurofeedback Therapy Services in New York, New York. Mark designed and provided the EEG neurofeedback training protocol in the current trial. The authors would also like to thank the Centre for Mindfulness, University of Southern California, for training the research assistant to provide mindfulness meditation in this trial.
Conflict of interest
Mark Smith owns Neurofeedback Therapy Services in New York, New York. Our industry partner, Mark, provided the EEG neurofeedback training software protocol in the current trial.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Wilson R, Abbott JH. The projected burden of knee osteoarthritis in New Zealand: healthcare expenditure and total joint replacement provision. N Z Med J. (2019) 132(1503):53–65.31581182
2. Ministry of Health. Annual Data Explorer 2021/22: New Zealand Health Survey [Data File]. (2022). Available at: https://minhealthnz.shinyapps.io/nz-health-survey-2021-22-annual-data-explorer/ (Accessed July 25, 2023).
3. Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M, et al. The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis. (2014) 73(7):1323–30. doi: 10.1136/annrheumdis-2013-204763
4. Perrot S, Cohen M, Barke A, Korwisi B, Rief W, Treede RD, et al. The IASP classification of chronic pain for ICD-11: chronic secondary musculoskeletal pain. Pain. (2019) 160(1):77–82. doi: 10.1097/j.pain.0000000000001389
5. Finan PH, Buenaver LF, Bounds SC, Hussain S, Park RJ, Haque UJ, et al. Discordance between pain and radiographic severity in knee osteoarthritis: findings from quantitative sensory testing of central sensitisation. Arthritis Rheum. (2013) 65(2):363–72. doi: 10.1002/art.34646
6. Neogi T. The epidemiology and impact of pain in osteoarthritis. Osteoarthritis Cartilage. (2013) 21(9):1145–53. doi: 10.1016/j.joca.2013.03.018
7. Fingleton C, Smart K, Moloney N, Fullen BM, Doody C. Pain sensitisation in people with knee osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage. (2015) 23(7):1043–56. doi: 10.1016/j.joca.2015.02.163
8. Neogi T, Frey-Law L, Scholz J, Niu J, Arendt-Nielsen L, Woolf C, et al. Sensitivity and sensitisation in relation to pain severity in knee osteoarthritis: trait or state? Ann Rheum Dis. (2015) 74(4):682–8. doi: 10.1136/annrheumdis-2013-204191
9. Cardoso JS, Riley JL, Glover T, Sibille KT, Bartley EJ, Goodin BR, et al. Experimental pain phenotyping in community-dwelling individuals with knee osteoarthritis. Pain. (2016) 157(9):2104–14. doi: 10.1097/j.pain.0000000000000625
10. Bartley EJ, King CD, Sibille KT, Cruz-Almeida Y, Riley JL 3rd, Glover TL, et al. Enhanced pain sensitivity among individuals with symptomatic knee osteoarthritis: potential sex differences in central sensitisation. Arthritis Care Res (Hoboken). (2016) 68(4):472–80. doi: 10.1002/acr.22712
11. Cruz-Almeida Y, Cardoso J, Riley JL 3rd, Goodin B, King CD, Petrov M, et al. Physical performance and movement-evoked pain profiles in community-dwelling individuals at risk for knee osteoarthritis. Exp Gerontol. (2017) 98:186–91. doi: 10.1016/j.exger.2017.08.026
12. French HP, Smart KM, Doyle F. Prevalence of neuropathic pain in knee or hip osteoarthritis: a systematic review and meta-analysis. Semin Arthritis Rheum. (2017) 47(1):1–8. doi: 10.1016/j.semarthrit.2017.02.008
13. Cottam WJ, Condon L, Alshuft H, Reckziegel D, Auer DP. Associations of limbic-affective brain activity and severity of ongoing chronic arthritis pain are explained by trait anxiety. NeuroImage Clin. (2016) 12:269–76. doi: 10.1016/j.nicl.2016.06.022
14. Hiramatsu T, Nakanishi K, Yoshimura S, Yoshino A, Adachi N, Okamoto Y, et al. The dorsolateral prefrontal network is involved in pain perception in knee osteoarthritis patients. Neurosci Lett. (2014) 581:109–14. doi: 10.1016/j.neulet.2014.08.027
15. Kulkarni B, Bentley DE, Elliott R, Julyan PJ, Boger E, Watson A, et al. Arthritic pain is processed in brain areas concerned with emotions and fear. Arthritis Rheum. (2007) 56(4):1345–54. doi: 10.1002/art.22460
16. Cruz-Almeida Y, King CD, Goodin BR, Sibille KT, Glover TL, Riley JL, et al. Psychological profiles and pain characteristics of older adults with knee osteoarthritis. Arthritis Care Res (Hoboken). (2013) 65(11):1786–94. doi: 10.1002/acr.22070
17. Mathew J, Adhia DB, Hall M, De Ridder D, Mani R. EEG-based cortical alterations in individuals with chronic knee pain secondary to osteoarthritis: a cross-sectional investigation. J Pain. (2023):S1526-5900(23)00619-3. doi: 10.1016/j.jpain.2023.11.012
18. Lee AC, Harvey WF, Price LL, Morgan LP, Morgan NL, Wang C. Mindfulness is associated with psychological health and moderates pain in knee osteoarthritis. Osteoarthritis Cartilage. (2017) 25(6):824–31. doi: 10.1016/j.joca.2016.06.017
19. Reddan MC, Wager TD. Brain systems at the intersection of chronic pain and self-regulation. Neurosci Lett. (2019) 702:24–33. doi: 10.1016/j.neulet.2018.11.047
20. Weiss S, Sack M, Henningsen P, Pollatos O. On the interaction of self-regulation, interoception and pain perception. Psychopathology. (2014) 47(6):377–82. doi: 10.1159/000365107
21. Gard T, Hölzel BK, Sack AT, Hempel H, Lazar SW, Vaitl D, et al. Pain attenuation through mindfulness is associated with decreased cognitive control and increased sensory processing in the brain. Cerebral cortex. (2012) 22(11):2692–702. doi: 10.1093/cercor/bhr352
22. Kabatzinn J, Lipworth L, Burney R. The clinical use of mindfulness meditation for the self-regulation of chronic pain. J Behav Med. (1985) 8(2):163–90. doi: 10.1007/BF00845519
23. Hilton L, Hempel S, Ewing BA, Apaydin E, Xenakis L, Newberry S, et al. Mindfulness meditation for chronic pain: systematic review and meta-analysis. Ann Behav Med. (2017) 51(2):199–213. doi: 10.1007/s12160-016-9844-2
24. Swain N, Lennox Thompson B, Gallagher S, Paddison J, Mercer S. Gratitude enhanced mindfulness (GEM): a pilot study of an internet-delivered programme for self-management of pain and disability in people with arthritis. J Posit Psychol. (2020) 15(3):420–6. doi: 10.1080/17439760.2019.1627397
25. Zeidan F, Vago DR. Mindfulness meditation-based pain relief: a mechanistic account. Ann N Y Acad Sci. (2016) 1373:114–27. doi: 10.1111/nyas.13153
26. Zeidan F, Baumgartner JN, Coghill RC. The neural mechanisms of mindfulness-based pain relief: a functional magnetic resonance imaging-based review and primer. Pain Rep. (2019) 4(4):e759. doi: 10.1097/PR9.0000000000000759
27. Zeidan F, Martucci KT, Kraft RA, Gordon NS, McHaffie JG, Coghill RC. Brain mechanisms supporting the modulation of pain by mindfulness meditation. J Neurosci. (2011) 31(14):5540–8. doi: 10.1523/JNEUROSCI.5791-10.2011
28. Chiesa A, Serretti A, Jakobsen JC. Mindfulness: top-down or bottom-up emotion regulation strategy? Clin Psychol Rev. (2013) 33(1):82–96. doi: 10.1016/j.cpr.2012.10.006
29. Kerr CE, Sacchet MD, Lazar SW, Moore CI, Jones SR. Mindfulness starts with the body: somatosensory attention and top-down modulation of cortical alpha rhythms in mindfulness meditation. Front Hum Neurosci. (2013) 7(12):1–15.23355817
30. Zeidan F, Emerson NM, Farris SR, Ray JN, Jung Y, McHaffie JG, et al. Mindfulness meditation-based pain relief employs different neural mechanisms than placebo and sham mindfulness meditation-induced analgesia. J Neurosci. (2015) 35(46):15307–25. doi: 10.1523/JNEUROSCI.2542-15.2015
31. Zeidan F, Grant JA, Brown CA, McHaffie JG, Coghill RC. Mindfulness meditation-related pain relief: evidence for unique brain mechanisms in the regulation of pain. Neurosci Lett. (2012) 520(2):165–73. doi: 10.1016/j.neulet.2012.03.082
32. Creswell JD, Taren AA, Lindsay EK, Greco CM, Gianaros PJ, Fairgrieve A, et al. Alterations in resting-state functional connectivity link mindfulness meditation with reduced interleukin-6: a randomized controlled trial. Biol Psychiatry. (2016) 80(1):53–61. doi: 10.1016/j.biopsych.2016.01.008
33. Lehmann D, Faber PL, Tei S, Pascual-Marqui RD, Milz P, Kochi K. Reduced functional connectivity between cortical sources in five meditation traditions detected with lagged coherence using EEG tomography. NeuroImage. (2012) 60(2):1574–86. doi: 10.1016/j.neuroimage.2012.01.042
34. Zeidan F, Martucci KT, Kraft RA, McHaffie JG, Coghill RC. Neural correlates of mindfulness meditation-related anxiety relief. Soc Cogn Affect Neursci. (2014) 9(6):751–9. doi: 10.1093/scan/nst041
35. Zeidan F, Johnson SK, Gordon NS, Goolkasian P. Effects of brief and sham mindfulness meditation on mood and cardiovascular variables. J Altern Complement Med. (2010) 16(8):867–73. doi: 10.1089/acm.2009.0321
36. Zeidan F, Johnson SK, Diamond BJ, David Z, Goolkasian P. Mindfulness meditation improves cognition: evidence of brief mental training. Conscious Cogn. (2010) 19(2):597–605. doi: 10.1016/j.concog.2010.03.014
37. Adler-Neal AL, Waugh CE, Garland EL, Shaltout HA, Diz DI, Zeidan F. The role of heart rate variability in mindfulness-based pain relief. J Pain. (2020) 21(3–4):306–23. doi: 10.1016/j.jpain.2019.07.003
38. Wells RE, Collier J, Posey G, Morgan F, Auman T, Strittameter B, et al. Attention to breath sensations does not engage endogenous opioids to reduce pain. Pain. (2020) 161(8):1884–93. doi: 10.1097/j.pain.0000000000001865
39. Jacobs EH, Jensen MP. EEG neurofeedback in the treatment of chronic pain: a case series. NeuroRegulation. (2015) 2(2):86. doi: 10.15540/nr.2.2.86
40. Koberda JL. LORETA Z-score neurofeedback in chronic pain and headaches. Z score neurofeedback: Clinical applications. Amsterdam: Elsevier Inc., Academic Press (2014). p. 115–39. doi: 10.1016/b978-0-12-801291-8.00006-6
41. Koberda JL, Koberda P, Bienkiewicz AA, Moses A, Koberda L. Pain management using 19-electrode Z-score LORETA neurofeedback. J Neurother. (2013) 17(3):179–90. doi: 10.1080/10874208.2013.813204
42. Jensen MP, Sherlin LH, Askew RL, Fregni F, Witkop G, Gianas A, et al. Effects of non-pharmacological pain treatments on brain states. Clin Neurophysiol. (2013) 124(10):2016–24. doi: 10.1016/j.clinph.2013.04.009
43. Jensen MP, Hakimian S, Sherlin LH, Fregni F. New insights into neuromodulatory approaches for the treatment of pain. J Pain. (2008) 9(3):193–9. doi: 10.1016/j.jpain.2007.11.003
44. Jensen MP, Sherlin LH, Hakimian S, Fregni F. Neuromodulatory approaches for chronic pain management: research findings and clinical implications. J Neurother. (2009) 13(4):196–213. doi: 10.1080/10874200903334371
45. Jensen MP, Gertz KJ, Kupper AE, Braden AL, Howe JD, Hakimian S, et al. Steps toward developing an EEG biofeedback treatment for chronic pain. Appl Psychophysiol Biofeedback. (2013) 38(2):101–8. doi: 10.1007/s10484-013-9214-9
46. Hammond DC. What is neurofeedback: an update. J Neurother. (2011) 15(4):305–36. doi: 10.1080/10874208.2011.623090
47. Vuckovic A, Altaleb MKH, Fraser M, McGeady C, Purcell M. EEG Correlates of self-managed neurofeedback treatment of central neuropathic pain in chronic spinal cord injury. Front Neurosci. (2019) 13:762. doi: 10.3389/fnins.2019.00762
48. Omejc N, Rojc B, Battaglini PP, Marusic U. Review of the therapeutic neurofeedback method using electroencephalography: EEG neurofeedback. Bosn J Basic Med. (2019) 19(3):213–20. doi: 10.17305/bjbms.2018.3785
49. Kubik A, Biedron A. Neurofeedback therapy in patients with acute and chronic pain syndromes–literature review and own experience. Przegl Lek. (2013) 70(7):440–2.24167944
50. Jensen MP, Grierson C, Tracy-Smith V, Bacigalupi SC, Othmer S. Neurofeedback treatment for pain associated with complex regional pain syndrome type I. J Neurother. (2007) 11(1):45–53. doi: 10.1300/J184v11n01_04
51. Kayıran S, Dursun E, Dursun N, Ermutlu N, Karamürsel S. Neurofeedback intervention in fibromyalgia syndrome; a randomised, controlled, rater blind clinical trial. Appl Psychophysiol Biofeedback. (2010) 35(4):293–302. doi: 10.1007/s10484-010-9135-9
52. Mayaud L, Wu H, Barthélemy Q, Favennec P, Delpierre Y, Congedo M, et al. Alpha-phase synchrony EEG training for multi-resistant chronic low back pain patients: an open-label pilot study. Eur Spine J. (2019) 28(11):2487–501. doi: 10.1007/s00586-019-06051-9
53. Patel K, Sutherland H, Henshaw J, Taylor J, Brown C, Casson A, et al. Effects of neurofeedback in the management of chronic pain: a systematic review and meta-analysis of clinical trials. Eur J Pain. (2020) 24(8):1440–57. doi: 10.1002/ejp.1612
54. Al-Taleb MKH, Purcell M, Fraser M, Petric-Gray N, Vuckovic A. Home used, patient self-managed, brain-computer interface for the management of central neuropathic pain post spinal cord injury: usability study. J Neuroeng Rehabil. (2019) 16(1):128. doi: 10.1186/s12984-019-0588-7
55. Hesam-Shariati N, McAuley JH, … , Gustin SM. The analgesic effect of electroencephalographic neurofeedback for people with chronic pain: a systematic review and meta-analysis. Eur J Neurol. (2022) 29(3):921–36. doi: 10.1111/ene.15189
56. Adhia DB, Mani R, Turner PR, Vanneste S, De Ridder D. Infraslow neurofeedback training alters effective connectivity in individuals with chronic low back pain: a secondary analysis of a pilot randomized placebo-controlled study. Brain Sci. (2022) 12(11):1514. doi: 10.3390/brainsci12111514
57. Tolin DF, Davies CD, Moskow DM, Hofmann SG. Biofeedback and neurofeedback for anxiety disorders: a quantitative and qualitative systematic review. Adv Exp Med Biol. (2020) 1191:265–89. doi: 10.1007/978-981-32-9705-0_16
58. Balt K, Du Toit P, Smith ML, van Rensburg CJ. The effect of infraslow frequency neurofeedback on autonomic nervous system function in adults with anxiety and related diseases. NeuroRegulation. (2020) 7(2):64. doi: 10.15540/nr.7.2.64
59. Grin-Yatsenko VA, Ponomarev VA, Kara O, Wandernoth B, Gregory M, Ilyukhina VA, et al. Effect of infra-low frequency neurofeedback on infra-slow EEG fluctuations. Biofeedback: IntechOpen (2018) p. 75–7. doi: 10.5772/intechopen.77154
60. Dobrushina OR, Vlasova RM, Rumshiskaya AD, Litvinova LD, Mershina EA, Sinitsyn VE, et al. Modulation of intrinsic brain connectivity by implicit electroencephalographic neurofeedback. Front Hum Neurosci. (2020) 14(192):1–13. doi: 10.3389/fnhum.2020.00192
61. Shtark MB, Kozlova LI, Bezmaternykh DD, Mel’nikov MY, Savelov AA, Sokhadze EM. Neuroimaging study of alpha and beta EEG biofeedback effects on neural networks. Appl Psychophysiol Biofeedback. (2018) 43(2):169–78. doi: 10.1007/s10484-018-9396-2
62. Morgenroth E, Saviola F, Gilleen J, Allen B, Lührs M, Eysenck MW, et al. Using connectivity-based real-time fMRI neurofeedback to modulate attentional and resting state networks in people with high trait anxiety. NeuroImage Clin. (2020) 25:102191. doi: 10.1016/j.nicl.2020.102191
63. Linhartová P, Látalová A, Kóša B, Kašpárek T, Schmahl C, Paret C. fMRI neurofeedback in emotion regulation: a literature review. NeuroImage. (2019) 193:75–92. doi: 10.1016/j.neuroimage.2019.03.011
64. Mathew J, Adhia DB, Smith ML, De Ridder D, Mani R. Source localized infraslow neurofeedback training in people with chronic painful knee osteoarthritis: a randomized, double-blind, sham-controlled feasibility clinical trial. Front Neurosci. (2022) 16:899772. doi: 10.3389/fnins.2022.899772
65. Adhia DB, Mani R, Mathew J, O’Leary F, Smith M, Vanneste S, et al. Exploring electroencephalographic infraslow neurofeedback treatment for chronic low back pain: a double-blinded safety and feasibility randomized placebo-controlled trial. Sci Rep. (2023) 13(1):1177. doi: 10.1038/s41598-023-28344-2
66. Burns JW. Mechanisms, mechanisms, mechanisms: it really does all boil down to mechanisms. Pain. (2016) 157(11):2393. doi: 10.1097/j.pain.0000000000000696
67. Day MA, Ward LC, Thorn BE, Burns J, Ehde DM, Barnier AJ, et al. Mechanisms of mindfulness meditation, cognitive therapy, and mindfulness-based cognitive therapy for chronic low back pain. Clin J Pain. (2020) 36(10):740–9. doi: 10.1097/AJP.0000000000000862
68. Cunningham NR, Kashikar-Zuck S, Coghill RC. Brain mechanisms impacted by psychological therapies for pain: identifying targets for optimisation of treatment effects. Pain Rep. (2019) 4(4):e767. doi: 10.1097/PR9.0000000000000767
69. Eldridge SM, Chan CL, Campbell MJ, Bond CM, Hopewell S, Thabane L, et al. CONSORT 2010 statement: extension to randomised pilot and feasibility trials. Br Med J. (2016) 355:i5239. doi: 10.1136/bmj.i5239
70. Hoffmann TC, Glasziou PP, Boutron I, Milne R, Perera R, Moher D, et al. Better reporting of interventions: template for intervention description and replication (TIDieR) checklist and guide. Bmj-Brit Med J. (2014) 348:g1687. doi: 10.1136/bmj.g1687
71. Craig P, Dieppe P, Macintyre S, Michie S, Nazareth I, Petticrew M. Developing and evaluating complex interventions: the new medical research council guidance. Br Med J. (2008) 337:a1655. doi: 10.1136/bmj.a1655
72. Moore GF, Audrey S, Barker M, Bond L, Bonell C, Hardeman W, et al. Process evaluation of complex interventions: medical research council guidance. BMJ. (2015) 350:h1258. doi: 10.1136/bmj.h1258
73. Awatere SA. “I like to be treated like a person, a little smile never costs a thing”: weaving kaumātua experiences of living with osteoarthritis in Hawkes’s bay, Aotearoa New Zealand into a collaborative osteoarthritis-management toolkit: a thesis presented for the requirements for the degree of doctor of philosophy, public health, Massey university, Palmerston North. New Zealand: Massey University (2018).
74. Hoeta TB, Baxter GD, Bryant K, Mani R. Evidence for pain assessment tools sensitive for Māori—a systematic review. New Zealand: Health Research Council (2018).
75. Hoeta TJ, Baxter GD, Pōtiki Bryant KA, Mani R. Māori pain experiences and culturally valid pain assessment tools for Māori: a systematic narrative review. N Z J Physiother. (2020) 48(1):37–50. doi: 10.15619/NZJP/48.1.05
76. McGruer N, Baldwin JN, Ruakere BT, Lamer PJ. Māori lived experience of osteoarthritis: a qualitative study guided by Kaupapa Māori principles. J Prim Health Care. (2019) 11(2):128–37. doi: 10.1071/HC18079
77. McGavock Z, Moewaka-Barnes H, McCreanor T. Māori and pain: a literature review. AlterNative. (2012) 8(2):163–75. doi: 10.1177/117718011200800205
78. McGavock ZC. A life transformed: the lived experiences of Māori with chronic pain. Wellington, New Zealand: Massey University (2011).
79. Magnusson JE, Fennell JA. Understanding the role of culture in pain: Māori practitioner perspectives of pain descriptors. N Z Med J. (2011) 124(1328):30–40. PMID: 21475337.
80. Fennell JA. Understanding the experience of pain from a Māori perspective. Auckland: The University of Auckland (2005).
81. Altman R. Criteria for classification of clinical osteoarthritis. J Rheumatol. (1991) 18(Suppl. 27):10–2. PMID: 2027107.
82. McAlindon TE, Driban J, Henrotin Y, Hunter D, Jiang G-L, Skou ST, et al. OARSI clinical trials recommendations: design, conduct, and reporting of clinical trials for knee osteoarthritis. Osteoarthritis Cartilage. (2015) 23(5):747–60. doi: 10.1016/j.joca.2015.03.005
83. de Rooij M, van der Leeden M, Heymans MW, Holla JF, Häkkinen A, Lems WF, et al. Prognosis of pain and physical functioning in patients with knee osteoarthritis: a systematic review and meta-analysis. Arthrit Care Res. (2016) 68(4):481–92. doi: 10.1002/acr.22693
84. Altman R, Asch E, Bloch D, Bole G, Borenstein D, Brandt K, et al. Development of criteria for the classification and reporting of osteoarthritis: classification of osteoarthritis of the knee. Arthritis Rheum. (1986) 29(8):1039–49. doi: 10.1002/art.1780290816
85. Zeidan F, Gordon NS, Merchant J, Goolkasian P. The effects of brief mindfulness meditation training on experimentally induced pain. J Pain. (2010) 11(3):199–209. doi: 10.1016/j.jpain.2009.07.015
86. Leong SL, Vanneste S, Lim J, Smith M, Manning P, De Ridder D. A randomised, double-blind, placebo-controlled parallel trial of closed-loop infraslow brain training in food addiction. Sci Rep. (2018) 8(1):11659. doi: 10.1038/s41598-018-30181-7
87. Mathew J, Perez TM, Adhia DB, De Ridder D, Mani R. Is there a difference in EEG characteristics in acute, chronic, and experimentally induced musculoskeletal pain states? A systematic review. Clin EEG Neurosci. (2022) 55(1):101–20. doi: 10.1177/15500594221138292
88. Watson BO. Cognitive and physiologic impacts of the infraslow oscillation. Front Syst Neurosci. (2018) 12:44. doi: 10.3389/fnsys.2018.00044
89. Grooms JK, Thompson GJ, Pan WJ, Billings J, Schumacher EH, Epstein CM, et al. Infraslow electroencephalographic and dynamic resting state network activity. Brain Connect. (2017) 7(5):265–80. doi: 10.1089/brain.2017.0492
90. Hiltunen T, Kantola J, Abou Elseoud A, Lepola P, Suominen K, Starck T, et al. Infra-slow EEG fluctuations are correlated with resting-state network dynamics in fMRI. J Neurosci. (2014) 34(2):356–62. doi: 10.1523/JNEUROSCI.0276-13.2014
91. De Ridder D, Vanneste S. The Bayesian brain in imbalance: medial, lateral and descending pathways in tinnitus and pain: A perspective. Prog Brain Res. (2021) 262:309–34. doi: 10.1016/bs.pbr.2020.07.012
92. Zhang BL, Jung M, Tu YH, Gollub R, Lang C, Ortiz A, et al. Identifying brain regions associated with the neuropathology of chronic low back pain: a resting-state amplitude of low-frequency fluctuation study. Br J Anaesth. (2019) 123(2):E303–11. doi: 10.1016/j.bja.2019.02.021
93. Rogel A, Guez J, Getter N, Keha E, Cohen T, Amor T, et al. Transient adverse side effects during neurofeedback training: a randomised, sham-controlled, double blind study. Appl Psychophysiol Biofeedback. (2015) 40(3):209–18. doi: 10.1007/s10484-015-9289-6
94. Locke HN, Brooks J, Arendsen LJ, Jacob NK, Casson A, Jones AK, et al. Acceptability and usability of smartphone-based brainwave entrainment technology used by individuals with chronic pain in a home setting. Br J Pain. (2020) 14(3):161–70. doi: 10.1177/2049463720908798
95. Reid P, Paine SJ, Curtis E, Jones R, Anderson A, Willing E, et al. Achieving health equity in Aotearoa: strengthening responsiveness to Māori in health research. N Z Med J. (2017) 130(1465):96–103.29121628
96. Bellamy N, Kirwan J, Boers M, Brooks P, Strand V, Tugwell P, et al. Recommendations for a core set of outcome measures for future phase III clinical trials in knee, hip, and hand osteoarthritis. Consensus development at OMERACT III. J Rheumatol. (1997) 24(4):799–802. PMID: 9101522.
97. Taylor AM, Phillips K, Patel KV, Turk DC, Dworkin RH, Beaton D, et al. Assessment of physical function and participation in chronic pain clinical trials: IMMPACT/OMERACT recommendations. Pain. (2016) 157(9):1836–50. doi: 10.1097/j.pain.0000000000000577
98. Yarnitsky D, Bouhassira D, Drewes AM, Fillingim RB, Granot M, Hansson P, et al. Recommendations on practice of conditioned pain modulation (CPM) testing. Eur J Pain. (2015) 19(6):805–6. doi: 10.1002/ejp.605
99. Alghadir AH, Anwer S, Iqbal A, Iqbal ZA. Test–retest reliability, validity, and minimum detectable change of visual analog, numerical rating, and verbal rating scales for measurement of osteoarthritic knee pain. J Pain Res. (2018) 11:851. doi: 10.2147/JPR.S158847
100. Lee AC, Driban JB, Price LL, Harvey WF, Rodday AM, Wang C. Responsiveness and minimally important differences for 4 patient-reported outcomes measurement information system short forms: physical function, pain interference, depression, and anxiety in knee osteoarthritis. J Pain. (2017) 18(9):1096–110. doi: 10.1016/j.jpain.2017.05.001
101. Roos EM, Lohmander LS. The Knee injury and Osteoarthritis Outcome Score (KOOS): from joint injury to osteoarthritis. Health Qual Life Out. (2003) 1(1):1–8. doi: 10.1186/1477-7525-1-64
102. Pham T, Van Der Heijde D, Lassere M, Altman RD, Anderson JJ, Bellamy N, et al. Outcome variables for osteoarthritis clinical trials: the OMERACT-OARSI set of responder criteria. J Rheumatol. (2003) 30(7):1648–54. PMID: 12858473.
103. Jaeschke R, Singer J, Guyatt GH. Measurement of health status: ascertaining the minimal clinically important difference. Control Clin Trials. (1989) 10(4):407–15. doi: 10.1016/0197-2456(89)90005-6
104. Balestroni G, Bertolotti G. EuroQol-5D (EQ-5D): an instrument for measuring quality of life. Monaldi Arch Chest Dis. (2012) 78(3):155–9. doi: 10.4081/monaldi.2012.121
105. Pinto D, Robertson M, Abbott J, Hansen P, Campbell A, Team MT. Manual therapy, exercise therapy, or both, in addition to usual care, for osteoarthritis of the hip or knee. 2: economic evaluation alongside a randomised controlled trial. Osteoarthritis Cartilage. (2013) 21(10):1504–13. doi: 10.1016/j.joca.2013.06.014
106. Campbell CM, Buenaver LF, Finan P, Bounds SC, Redding M, McCauley L, et al. Sleep, pain catastrophizing, and central sensitization in knee osteoarthritis patients with and without insomnia. Arthritis Care Res. (2015) 67(10):1387–96. doi: 10.1002/acr.22609
107. Stubbs B, Aluko Y, Myint PK, Smith TO. Prevalence of depressive symptoms and anxiety in osteoarthritis: a systematic review and meta-analysis. Age Ageing. (2016) 45(2):228–35. doi: 10.1093/ageing/afw001
108. Kittelson AJ, Stevens-Lapsley JE, Schmiege SJ. Determination of pain phenotypes in knee osteoarthritis: a latent class analysis using data from the osteoarthritis initiative. Arthritis Care Res. (2016) 68(5):612–20. doi: 10.1002/acr.22734
109. Riddle DL, Jensen MP. Construct and criterion-based validity of brief pain coping scales in persons with chronic knee osteoarthritis pain. Pain Med. (2013) 14(2):265–75. doi: 10.1111/pme.12007
110. Scopaz KA, Piva SR, Wisniewski S, Fitzgerald GK. Relationships of fear, anxiety, and depression with physical function in patients with knee osteoarthritis. Arch Phys Med Rehabil. (2009) 90(11):1866–73. doi: 10.1016/j.apmr.2009.06.012
111. Day MA, Thorn BE, Ehde DM, Burns JW, Barnier A, Mattingley JB, et al. Moderators of mindfulness meditation, cognitive therapy, and mindfulness-based cognitive therapy for chronic low back pain: a test of the limit, activate, and enhance model. J Pain. (2020) 36(10):740–9. doi: 10.1016/j.jpain.2019.06.006
112. Nicholas MK, McGuire BE, Asghari A. A 2-item short form of the pain self-efficacy questionnaire: development and psychometric evaluation of PSEQ-2. J Pain. (2015) 16(2):153–63. doi: 10.1016/j.jpain.2014.11.002
113. Devilly GJ, Borkovec TD. Psychometric properties of the credibility/expectancy questionnaire. J Behav Ther Exp Psychiatry. (2000) 31(2):73–86. doi: 10.1016/S0005-7916(00)00012-4
114. Weber LA, Ethofer T, Ehlis A-C. Predictors of neurofeedback training outcome: a systematic review. NeuroImage Clin. (2020) 27:102301. doi: 10.1016/j.nicl.2020.102301
115. Mani R, Adhia DB, Leong SL, Vanneste S, De Ridder D. Sedentary behaviour facilitates conditioned pain modulation in middle-aged and older adults with persistent musculoskeletal pain: a cross-sectional investigation. Pain Rep. (2019) 4(5):e773. doi: 10.1097/PR9.0000000000000773
116. Alqarni AM, Manlapaz D, Baxter D, Tumilty S, Mani R. Test procedures to assess somatosensory abnormalities in individuals with peripheral joint pain: a systematic review of psychometric properties. Pain Pract. (2018) 18(7):895–924. doi: 10.1111/papr.12680
117. Othman R, Dassanayake S, Jayakaran P, Tumilty S, Swain N, Mani R. Relationships between psychological, social, physical activity, and sleep measures and somatosensory function in individuals with spinal pain: a systematic review and meta-analysis. Clin J Pain. (2020) 36(2):124–34. doi: 10.1097/AJP.0000000000000775
118. Othman R, Jayakaran P, Swain N, Dassanayake S, Tumilty S, Mani R. Relationships between psychological, sleep, and physical activity measures and somatosensory function in people with peripheral joint pain: a systematic review and metaanalysis. Pain Pract. (2021) 21(2):226–61. doi: 10.1111/papr.12943
119. Rolke R, Baron R, Maier C, Tolle TR, Treede RD, Beyer A, et al. Quantitative sensory testing in the German research network on neuropathic pain (DFNS): standardized protocol and reference values. Pain. (2006) 123(3):231–43. doi: 10.1016/j.pain.2006.01.041
120. Edwards RR, Dworkin RH, Turk DC, Angst MS, Dionne R, Freeman R, et al. Patient phenotyping in clinical trials of chronic pain treatments: IMMPACT recommendations. Pain. (2016) 157(9):1851. doi: 10.1097/j.pain.0000000000000602
121. Goodin BR, Bulls HW, Herbert MS, Schmidt J, King CD, Glover TL, et al. Temporal summation of pain as a prospective predictor of clinical pain severity in adults aged 45 years and older with knee osteoarthritis: ethnic differences. Psychosom Med. (2014) 76(4):302–10. doi: 10.1097/PSY.0000000000000058
122. Suokas AK, Walsh DA, McWilliams DF, Condon L, Moreton B, Wylde V, et al. Quantitative sensory testing in painful osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage. (2012) 20(10):1075–85. doi: 10.1016/j.joca.2012.06.009
123. Camm AJ, Malik M, Bigger JT, Breithardt G, Cerutti S, Cohen RJ, et al. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation. (1996) 93(5):1043–65. PMID: 8598068.
124. Van Den Houte M, Van Oudenhove L, Bogaerts K, Van Diest I, Van den Bergh O. Endogenous pain modulation: association with resting heart rate variability and negative affectivity. Pain Med. (2018) 19(8):1587–96. doi: 10.1093/pm/pnx165
125. Vanneste S, Ost J, Van Havenbergh T, De Ridder D. Resting state electrical brain activity and connectivity in fibromyalgia. PLoS One. (2017) 12(6):e0178516. doi: 10.1371/journal.pone.0178516
126. Ploner M, May ES. Electroencephalography and magnetoencephalography in pain research—current state and future perspectives. Pain. (2018) 159(2):206–11. doi: 10.1097/j.pain.0000000000001087
127. Faber PL, Lehmann D, Tei S, Tsujiuchi T, Kumano H, Pascual-Marqui RD, et al. EEG source imaging during two qigong meditations. Cogn Process. (2012) 13(3):255–65. doi: 10.1007/s10339-012-0441-4
128. Health Research Council of New Zealand. Guidelines for researchers on health research involving Māori. Auckland, New Zealand: Health Research Council of New Zealand. (2010).
129. Cunningham C. A framework for addressing Māori knowledge in research, science and technology. Pac Health Dialog. (2000) 7(1):62–9. PMID: 11709883.
130. Smith LT. Decolonising methodologies: research and indigenous peoples. London & New York: Zed Books Ltd. (2013).
131. Durie M. Exploring the interface between science and indigenous knowledge. 5th APEC Research and Development Leaders Forum; 2004; Christchurch, New Zealand (2004).
132. Overton M, Swain N, Falling C, Gwynne-Jones D, Fillingim R, Mani R. Activity-related pain predicts pain and functional outcomes in people with knee osteoarthritis: a longitudinal study. Front Pain Res. (2023) 3:1082252. doi: 10.3389/fpain.2022.1082252
133. Cameron D, Willoughby C, Messer D, Lux M, Aitken M, Getz K. Assessing participation burden in clinical trials: introducing the patient friction coefficient. Clin Ther. (2020) 42(8):e150–9. doi: 10.1016/j.clinthera.2020.06.015
134. Rolstad S, Adler J, Rydén A. Response burden and questionnaire length: is shorter better? A review and meta-analysis. Value Health. (2011) 14(8):1101–8. doi: 10.1016/j.jval.2011.06.003
135. Darlow B, Brown M, Thompson B, Hudson B, Grainger R, McKinlay E, et al. Living with osteoarthritis is a balancing act: an exploration of patients’ beliefs about knee pain. BMC Rheumatol. (2018) 2(1):1–9. doi: 10.1186/s41927-018-0023-x
136. Binda DD, Greco CM, Morone NE. What are adverse events in mindfulness meditation? Glob Adv Health Med. (2022) 11:2164957X221096640. doi: 10.1177/2164957X221096640
137. Abbott JH. The distinction between randomized clinical trials (RCTs) and preliminary feasibility and pilot studies: what they are and are not. J Orthop Sports Phys Ther. (2014) 44(8):555–8. doi: 10.2519/jospt.2014.0110
Keywords: chronic pain, musculoskeletal pain, mindfulness meditation (MM), mindfulness—pain intervention, neurofeedback (NF), self-regulation training, knee osteoarthritis, feasibility and acceptability
Citation: Mani R, Adhia DB, Awatere S, Gray AR, Mathew J, Wilson LC, Still A, Jackson D, Hudson B, Zeidan F, Fillingim R and De Ridder D (2024) Self-regulation training for people with knee osteoarthritis: a protocol for a feasibility randomised control trial (MiNT trial). Front. Pain Res. 4:1271839. doi: 10.3389/fpain.2023.1271839
Received: 2 August 2023; Accepted: 7 December 2023;
Published: 8 January 2024.
Edited by:
Man-Kyo Chung, University of Maryland, United StatesReviewed by:
Tatsunori Ikemoto, Aichi Medical University, JapanDokyoung Sophia You, Stanford University, United States
© 2024 Mani, Adhia, Awatere, Gray, Mathew, Wilson, Still, Jackson, Hudson, Zeidan, Fillingim and De Ridder. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ramakrishnan Mani cmFtYWtyaXNobmFuLm1hbmlAb3RhZ28uYWMubno=
 Ramakrishnan Mani
Ramakrishnan Mani Divya Bharatkumar Adhia
Divya Bharatkumar Adhia Sharon Awatere
Sharon Awatere Andrew Robert Gray
Andrew Robert Gray Jerin Mathew
Jerin Mathew Luke Charles Wilson
Luke Charles Wilson Amanda Still
Amanda Still David Jackson
David Jackson Ben Hudson7
Ben Hudson7 Roger Fillingim
Roger Fillingim Dirk De Ridder
Dirk De Ridder