- 1The Second Clinical College, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong, China
- 2Guangzhou University of Chinese Medicine, Guangzhou, Guangdong, China
- 3Guangdong Provincial Hospital of Chinese Medicine, Guangzhou, China
- 4The First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou, Guangdong, China
- 5KM Science Research Division, Korea Institute of Oriental Medicine (KIOM), Daejeon, Republic of Korea
- 6Department of Pediatrics, King Saud University Medical City, Riyadh, Saudi Arabia
- 7Research Chair for Evidence-Based Health Care and Knowledge Translation, Family and Community Medicine Department, College of Medicine, King Saud University, Riyadh, Saudi Arabia
- 8Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo, Ribeirão Preto, Brazil
- 9Department for Evidence-Based Medicine and Evaluation, University for Continuing Education Krems, Krems an der Donau, Lower Austria, Austria
- 10Oral Medicine, Oral Diagnosis, and Periodontology Department, Faculty of Dentistry, Cairo University, Cairo, Egypt
- 11State Key Laboratory of Traditional Chinese Medicine Syndrome, Research Group of Standardization of Chinese Medicine, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou, China
Background: To develop a rational and standardized traditional Chinese medicine (TCM) good practice recommendation (GPR) methodology model that guides the formulation of recommendations grounded in clinical experience.
Methods: We adopted an exploratory sequential mixed-method to develop a methodology model by coding systematically collected literature on methodology and TCM guidelines related to TCM GPR using a best-fit framework synthesis. Then based on real-world data (published TCM guidelines), saturation tests, structural rationality validation, and discriminability tests were conducted to validate methodology model.
Results: A total of 35 methodological literature and 190 TCM guidelines were included. A TCM GPR methodology model was developed, including 3 themes, 10 sub-themes, and the relationships between themes and subthemes. The information of TCM GPR methodology model achieved data saturation. The fit indices were within the acceptable range, and were able to distinguish the overall differences between guidelines from different literature sources, development organizations, guideline types, discipline categories, and funding categories.
Conclusion: The study developed a TCM GPR methodology model which describes the definition of a TCM GPR, how to formulate it, and how to report it. The methodology modeldemonstrates good fit, discriminability, and data saturation. It can standardize the specific formulation of TCM GPRs, facilitate the scientific and rational formation of TCM GPRs, and provide theoretical and methodological guidance for the formation of TCM GPRs.
1 Introduction
In 1992, the concept of evidence-based medicine was introduced and rapidly spread (Guyatt et al., 1992; Djulbegovic and Guyatt, 2017). Currently, the formulation of recommendations in traditional Chinese medicine (TCM) guidelines emphasizes evidence from systematic reviews (Verwoerd et al., 2021). However, when the evidence is insufficient, recommendations and guidance are still needed for some important and urgent questions (Ge et al., 2022; Liu et al., 2021). In response to such situations, guideline developers tend to use good practice approaches to introduce clinical experience, one of the three elements of evidence-based medicine, into guidelines to address the issues (Diekemper et al., 2018; Vermeulen et al., 2019; Guyatt et al., 2015; Guyatt et al., 2016; Dewidar et al., 2023).
Currently, there are two main research approaches in the field of TCM for incorporating clinical experience into clinical practice guidelines: The first approach is to integrate clinical experience into the evidence system, constructing a TCM-specific evidence body and subsequently developing recommendations based on this evidence system (Wang et al., 2013; Liu, 2007; Chen et al., 2019). The second approach involves translating clinical experience into recommendations outside the evidence system (Chen et al., 2013; Guo et al., 2008; Chang et al., 2022). These approaches represent active explorations for incorporating clinical experience into TCM guidelines. However, it is important to note that expert clinical experience should not be considered a research design and should not be included within the evidence system (Guyatt et al., 2008; Balshem et al., 2011; Oxman et al., 2006). In addition, the methods that operate outside of the evidence system lack systematic and standardized procedures, which undermines the quality of the recommendations and prevents alignment with recognized guideline methodologies.
In light of the issues discussed above, it is necessary to establish a systematic and standardized methodological system to standardize the formation of recommendations based on expert clinical experience in TCM guidelines. This study adopts an exploratory sequential mixed-methods approach to develop a TCM Good Practice Recommendation (GPR) methodology model, providing theoretical and methodological support for the formation of TCM GPR.
2 Methods
We adopted a mixed-methods approach grounded in an exploratory sequential design, where qualitative research based on best-fit framework synthesis was used to develop the TCM GPR methodology model (Carroll et al., 2011; Carroll et al., 2013). Then, quantitative research was conducted to validate this new model. Subsequently, an expert consensus meeting was held to review the results.
2.1 Systematically identify research samples
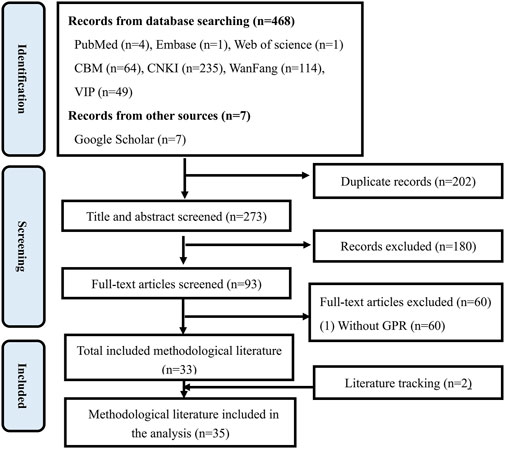
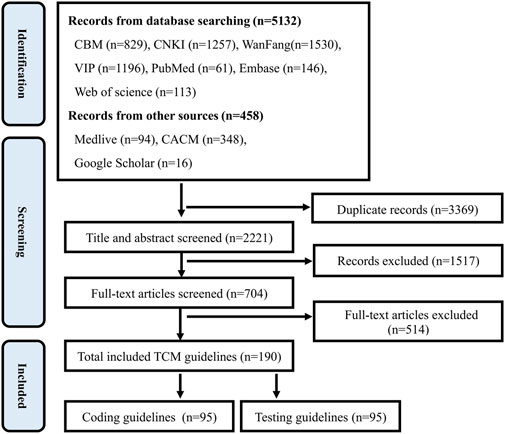
In our study, methodological literature on TCM GPR methods and TCM guidelines that mentioned TCM GPR methods were used as research samples. A comprehensive search was conducted using PubMed, EMBASE, Web of Science, China Biology Medicine Database (CBM), China National Knowledge Infrastructure (CNKI), Chinese Scientific Journal Database (VIP), and the Wanfang Database to search for methodological literature and TCM guidelines. Additionally, searches were performed on the China Association of Chinese Medicine (CACM) and Medlive (http://www.medlive.cn) websites for relevant TCM guidelines. Gray literature was also searched using Google Scholar. All relevant articles from their inception to February2023 were retrieved. The search strategy is provided in Supplementary Material 1.
2.1.1 Inclusion criteria for methodological literature
Inclusion Criteria: Studies related to methods or theories that provide recommendations based on clinical experience when there is a lack of evidence during the development of TCM guidelines.
Exclusion Criteria: (1) Duplicated literature; (2) Unavailability of full-text electronic version; (3) Conference abstracts.
2.1.2 Inclusion criteria for TCM guidelines
Inclusion Criteria: Including recommendations or opinions for clinical guidance based on clinical experience when there is a lack of evidence.
Exclusion Criteria: (1) Duplicated published literature; (2) Unavailability of electronic full text; (3) Interpretation, and evaluation of guidelines.
Two researchers independently screened the full-text articles. The results were then discussed with the research team to reach a consensus on the final inclusion of the methodological literature and TCM guidelines. SPSS 17.0 software was used to randomly select half of the TCM guidelines as coding guidelines for developing the methodology model. The remaining half of the TCM guidelines were used as testing guidelines to evaluate the saturation of the methodology model.
2.2 Model development
2.2.1 Thematic analysis to generate a priori framework
Using the logical framework method, we began with the problem of TCM GPR as the core issue and gradually extrapolated downward, constructing a problem tree, which was then transformed into the corresponding initial analytical framework based on the causal relationships illustrated in the problem tree.
According to the initial analytical framework, the thematic analysis (Thomas and Harden, 2008) method was used to code the methodology literature line-by-line. The codes were then categorized and grouped to form an a priori framework with a three-tier structure of themes, subthemes, and categories. Two researchers independently coded and cross-checked codes, and inconsistencies were discussed and agreed upon.
2.2.2 Synthesizing TCM GPR methodology model
Based on the a priori framework, we employed a line-by-line coding format to code the relevant information from the TCM guidelines into the corresponding themes/subthemes/categories in the priori framework. Information that could not be coded into the a priori framework was subjected to secondary thematic analysis to form new themes/subthemes/categories.
Ultimately, by merging the a priori frameworks with new themes and exploring potential linkages between the themes at each level, the TCM GPR methodology model was developed.
2.2.3 Dissonance exploration
Dissonance exploration aims to identify any differences between the a priori framework and the TCM GPR methodology model.
2.3 Model validation
2.3.1 Saturation test
A thematic saturation test was conducted using a theme saturation table. If five testing guidelines in succession do not yield any new themes, this indicates that the study has reached theme saturation (Yang et al., 2022; Constantinou et al., 2017).
2.3.2 Structural rationality validation
SPSS 17.0 and AMOS 21.0 software were used to assess the reliability and validity of the methodology model. Three themes were treated as latent variables, while ten subthemes were considered as observed variables. A matrix coding table was generated for specific data analysis, with each subtheme of each guideline serving as a reference point.
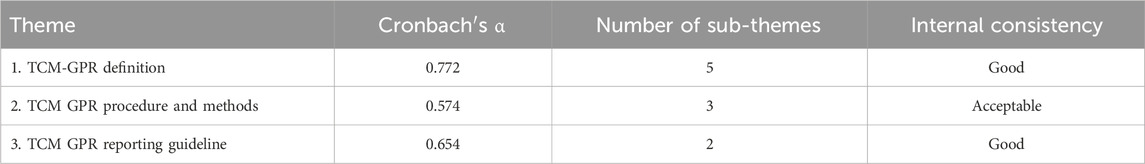
Reliability: The reliability of the model was assessed using Cronbach’s α. Cronbach’s α ≥0.6 was considered indicative of good reliability (Pires et al., 2022).
Validity: Several indices were used to evaluate the goodness of fit, including χ2/df, root mean square error of approximation (RMSEA), goodness of fit index (GFI), and adjusted goodness of fit index (AGFI). For χ2/df <3 was considered a good fit. For RMSEA <0.1 was considered an acceptable model fit (Pires et al., 2022; Yan et al., 2023). For GFI >0.90 or AGFI >0.8 was considered a good fit (Lopez et al., 2023; Marsh et al., 1988).
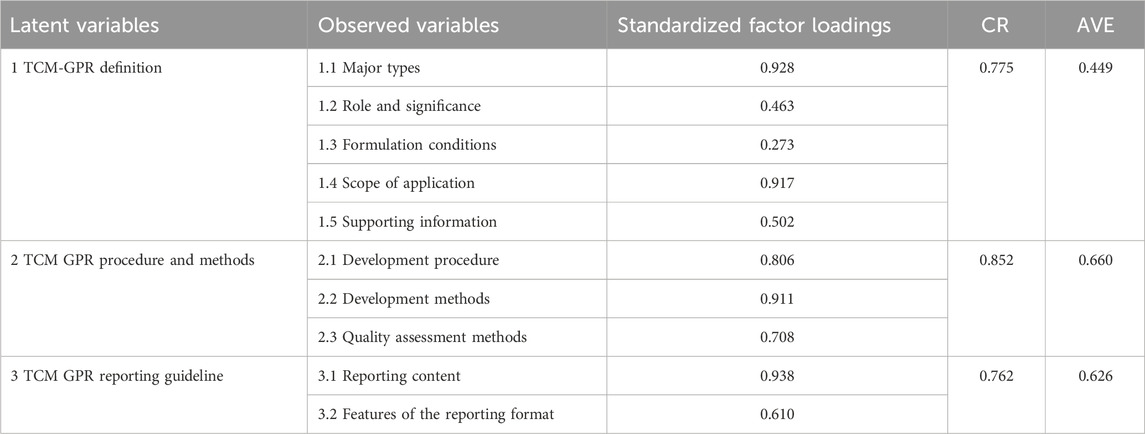
Composite reliability (CR) and average variance extracted (AVE) were used to assess the convergent validity and discriminant validity. For CR >0.60 and AVE >0.5 was considered a good convergent validity (Zhou et al., 2022).
2.3.3 Discriminability test
2.3.3.1 TCM GPR evaluation
After discussions and consensus among the research group, the TCM GPR evaluation was based on the three themes of the methodology model as domains and nine sub-themes as items. The sub-theme “1.1 Main types” was excluded. Points were assigned to the initial codes under the items, with 1 point assigned to the presence of the initial code and 0 points assigned to the absence of the initial code. The item score was the sum of the scores of the initial codes under the item, and the domain score was the sum of the item scores under the domain. The total score was the sum of the three domain scores. According to the assignment rules, the matrix coding table in NVivo 12 Plus software was exported for statistical analysis.
The score rate (%) was calculated using Microsoft Office Excel 2019 software. Item score = actual total item score/theoretical maximum item score. Domain score rate = actual domain score/theoretical maximum domain score. Total score rate = total theoretical maximum score.
Statistically significant differences in the scoring rates of the subgroups under different basic feature classifications indicate that the TCM GPR methodology model has discriminability and is capable of distinguishing between different types of TCM guidelines.
2.3.3.2 Subgroup analysis
Statistical analysis of subgroup scores was conducted based on the classification of basic features such as publication year, literature sources, development organizations, guideline types, discipline categories, and funding categories.
3 Results
3.1 Systematically identify research samples
A total of 35 methodological literature and 190 TCM guidelines were included. Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) Flowchart (Page et al., 2021) for methodological literature and TCM guidelines was shown in Figures 1, 2.
The first methodological literature about TCM GPR was published in 2008. Since then, the annual average number of publications has stayed low but stable. 68.57% (24/35) of the methodological literature received funding support. The number of funded articles has shown a general upward trend.
The first TCM guidelines that mentioned the TCM GPR method were published in 2011, and the number of publications has generally been on the rise. Among the 190 guidelines, 60.00% (114/190) received funding support.
3.2 Model development
3.2.1 Thematic analysis to generate an a priori framework
Through the logical framework analysis, a problem tree containing three key issues (unclear definitions, non-standardized procedures and methods, and non-uniformity of reporting forms) and nine phenomenon issues (role in terms of significance, conditions of use, scope of application, supporting information, procedures, methods, quality assessment, reporting content, and reporting presentation) was constructed. This was transformed into an initial analytical framework based on causal relationships (themes including definitions, procedures and methods, and reporting guideline, and sub-themes including role in terms of meaning, conditions of use, scope of application, supporting information, procedures, methods, quality assessment methods, reporting content, and reporting presentation).
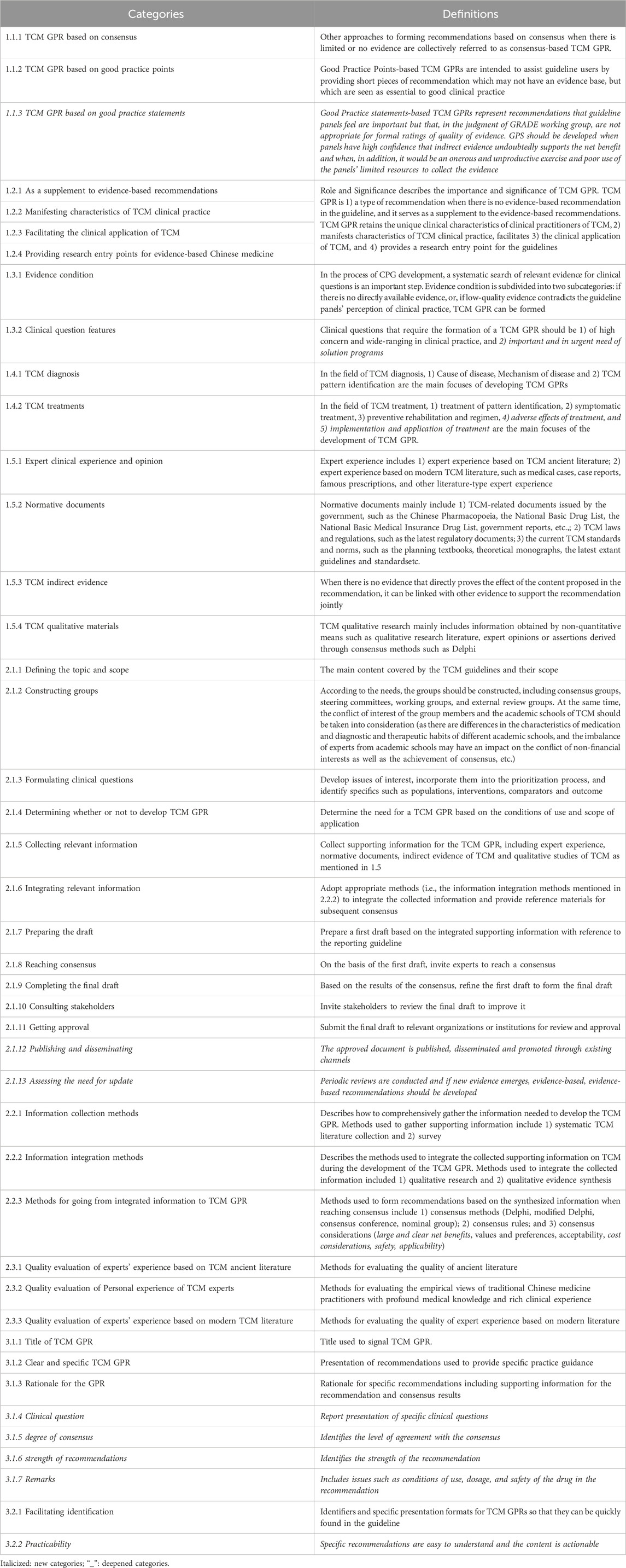
Through thematic analysis, a priori framework comprising three main themes and ten sub-themes was extracted from the literature, as summarized in Table 1.
Definition refers to what the TCM GPR is. 97.14% (34/35) of the methodological literature reported on this theme. Main Types describes the categories of methods integrated into the methodology model. Role and Significance describes the importance and significance of developing TCM GPR. Conditions of Use describes the conditions necessary for the development of TCM GPRs. Scope of Application describes the main areas where TCM GPRs can be developed. Supporting Information describes information that supports the formation of TCM GPR. The coding support of methodological literature for themes at all levels of TCM GPR Definition (the proportion of methodological literature mentioning the theme to the overall methodological literature) is detailed in Figure 3.

Figure 3. Coding support of methodological literature and coding guidelines for themes at all levels of TCM GPR definition.
Procedures and Methods refer to how to develop TCM GPR. 85.71% (30/35) of the methodological literature reported on this theme. Procedures describe the specific steps involved in developing the TCM GPR in chronological order. Methods describe the technical methods involved in developing the TCM GPR, and the quality assessment methodology describes the methods used to assess the quality of the supporting information, ensuring the credibility of the TCM GPR. The specific levels of support are shown in Figure 4.

Figure 4. Coding support of methodological literature and coding guidelines for themes at all levels of TCM GPR procedures and methods.
Reporting guideline refers to how to report TCM GPR. It plays an important role in clearly expressing the content of TCM GPR and quickly identifying TCM GPR. 17.14% (6/35) of the methodological literature reported on this theme. Report Content describes the essential elements that should be included in the report. Features of the reporting format describe how the report should be presented for clarity and comprehensiveness. Specific levels of support are shown in Figure 5.
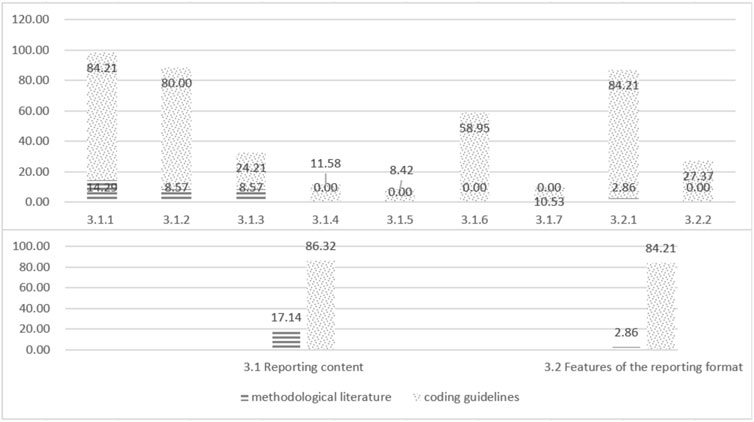
Figure 5. Coding support of methodological literature and coding guidelines for themes at all levels of TCM GPR reporting guideline.
3.2.2 Synthesizing the TCM GPR methodological model
The three main themes in the a priori framework were all supported by the TCM coding guidelines, with 100% (95/95), 78.95% (75/95), and 86.32% (82/95) levels of support respectively. All ten sub-themes were also supported by the coding guidelines. Specific levels of support are shown in Figures 3–5.
What could not be coded into the a priori framework was thematically analyzed to form new themes (sub-themes/categories). Themes and sub-themes remained unchanged, with eight new categories and four prior categories deepened. Ultimately, the a priori framework and new themes were merged to develop TCM GPR methodology model, including 3 themes, 10 sub-themes, and 42 categories, as shown in Table 2.
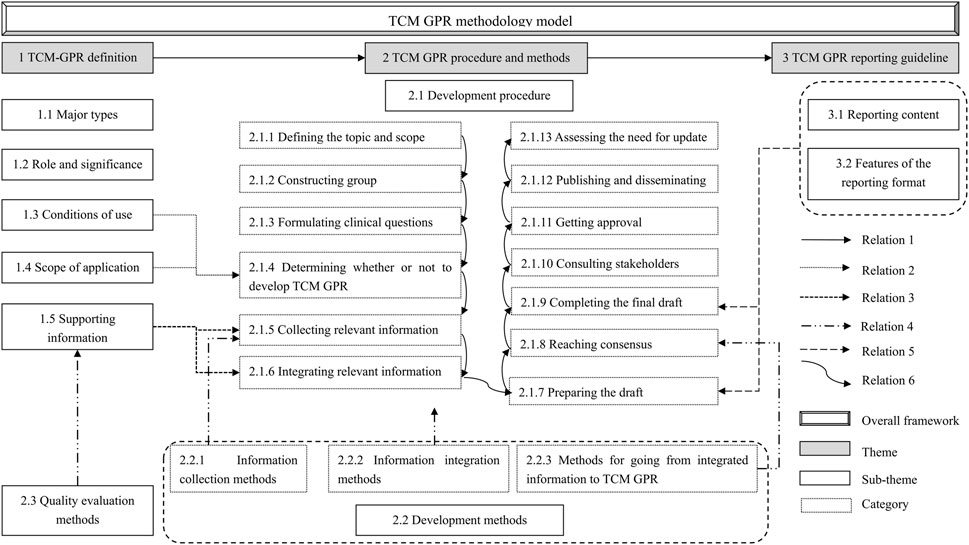
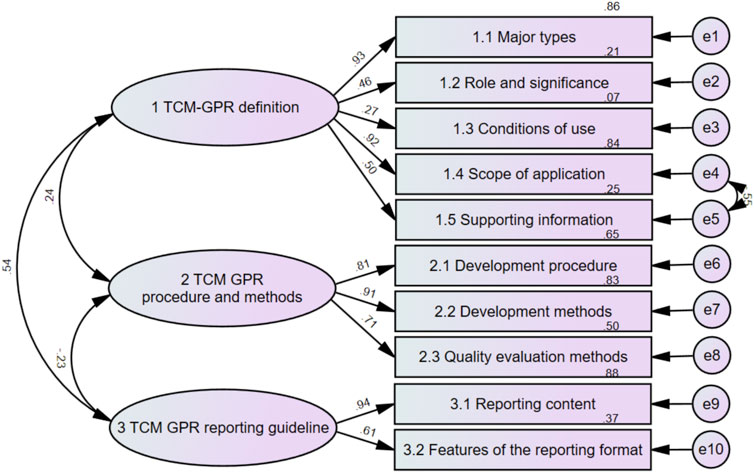
The relations between themes, sub-themes, and categories are presented in a thematic relationship diagram, as shown in Figure 6. The relationships are as follows:
Relation 1: Definition, procedures and methods, and reporting guideline are the main themes of the TCM GPR methodology model. The need for a TCM GPR is determined based on its definition. Once established, the GPR is developed and reported according to the specified procedures and methods, and reporting guideline.
Relation 2: “Conditions of Use” and “Scope of Application” are key determinants for the applicability of a TCM GPR. When both conditions are met, a TCM GPR can be developed.
Relation 3: The information collected and integrated during the “Collecting relevant information” and “Integrating relevant information” steps is provided by the “Supporting information”.
Relation 4: In the “Collecting relevant information” step, information is gathered using specified “Information collection methods”. During the “Integrating relevantinformation” step, the collected information is synthesized using “Information integration methods”. Concurrently, a quality assessment method is employed to evaluate the collected supporting information. The “Methods for going from integrated information to TCM GPR” provides methodological support for the “Reaching consensus” step.
Relation 5: At the steps of the “preparing the draft” and the “Completing the final draft”, TCM GPR could be reported according to the “Reporting guideline”.
Relation 6: The sequence of development steps.
3.2.3 Dissonance exploration
Compared with the a priori framework, the themes and sub-themes of the final TCM GPR methodology model remained unchanged, with eight new categories and four existing categories being further refined. The details are shown in Table 2.
3.3 Model validation
3.3.1 Saturation test
None of the test guidelines indicated any newly emerging themes or sub-themes, leading to the conclusion that the study had reached theme saturation. See Supplementary Material 2 for details.
3.3.2 Structural rationality test
TCM GPR methodology model includes 10 observational variables and requires at least 50 samples (Naidoo and Fields, 2019). With 190 TCM guidelines included in this study, the sample size is considered sufficient.
3.3.2.1 Reliability
The Cronbach’s α of the 3 themes were 0.772, 0.574, and 0.654 in that order. As shown in Table 3.
3.3.2.2 Validity
According to the data characteristics, a validation factor analysis was conducted by selecting Asymptotically distribution-free using the Analysis Properties column (Analysis Properties) in AMOS 21.0 software.The results are shown in Table 4: χ2/df was 2.988 (<3); the RMSEA was 0.103 (<0.11), indicating acceptable fit (Warlick et al., 2018); GFI was 0.932 (>0.9); and AGFI was 0.860 (>0.8). As shown in Figure 7, the standardized factor loadings of the subthemes were all >0.4 except for the (1.3) conditions of use.The CRs of the latent variables were 0.775, 0.852, and 0.762, which were all greater than 0.6 in that order, and the AVEs were 0.449, 0.660, and 0.662, in that order, except for “1 Definition of GPR in TCM”, which was greater than 0.5, See Table 5 for details.
3.3.3 Discriminability test
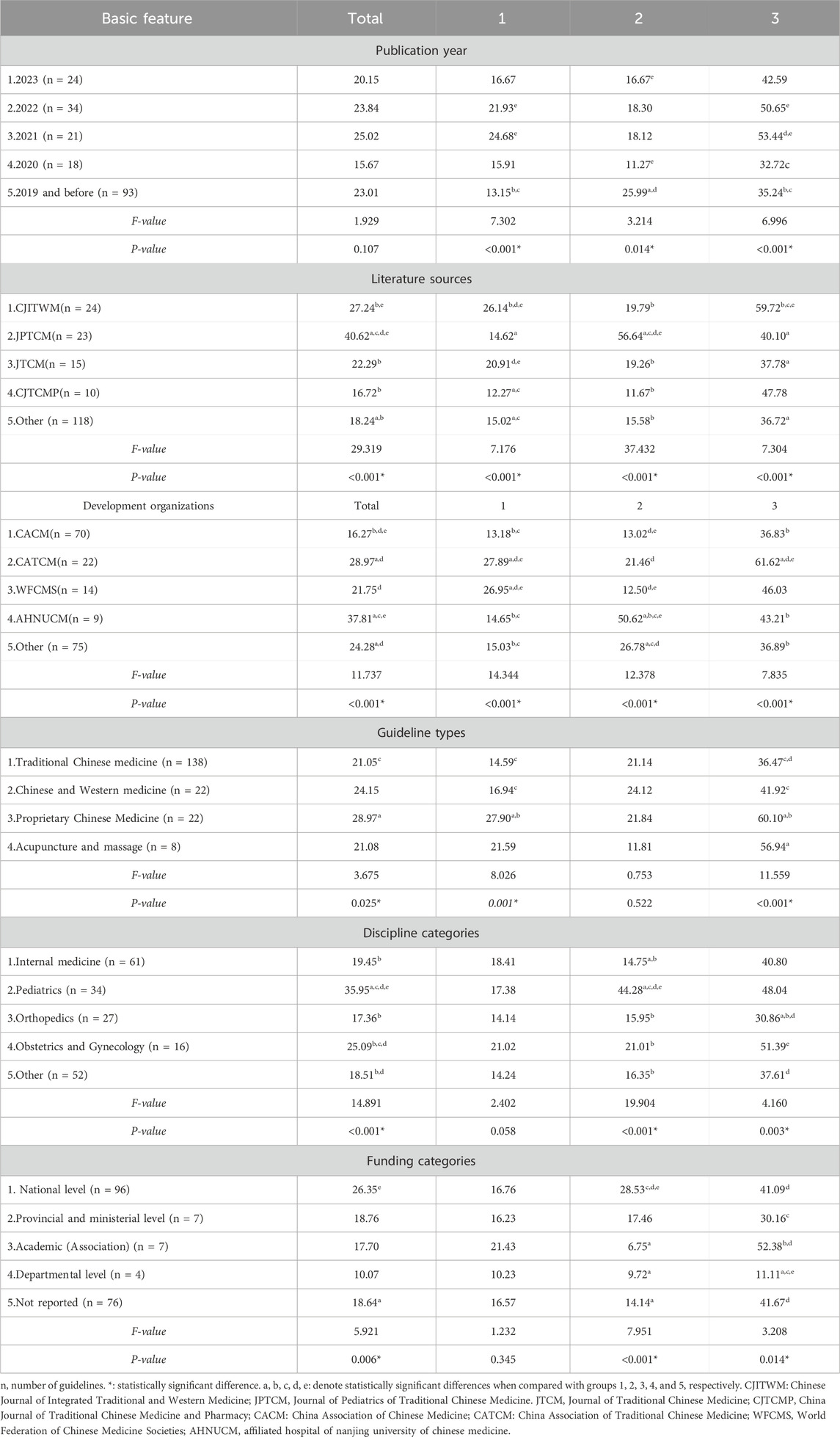
The results showed statistically significant differences in the total score rates of guidelines published by different literature sources, different development organizations, different guideline types, different discipline categories, and different funding categories. Statistically significant differences in the scoring rates of guidelines published in different years, different literature sources, and different development organizations on the definition, procedures and methods, and reporting guideline of TCM GPR, statistically significant differences in the scoring rates of guidelines published in different guideline types on the definition and reporting guideline of TCM GPR, and statistically significant differences in the scoring rates of guidelines published in different disciplinary categories, and different funding categories on the definition, procedures and methods, and reporting guideline of TCM GPR. As shown in Table 6.
4 Discussion
4.1 Increasing attention to TCM-GPR standardization
A total of 35 methodological papers and 190 TCM guidelines were analyzed in this study. Both showed an increasing trend in numbers, partly due to enhanced financial support, with most funded projects receiving national-level funding. This indicates an increasing emphasis on TCM-GPR standardization.
4.2 Model development
4.2.1 Definition of TCM GPR
According to the “TCM GPR Definition”, the concept of TCM GPR can be outlined as follows: When clinical evidence is of low (or very low) quality and it is inconsistent with the experience of clinical practice of TCM, or no direct evidence is in place, TCM guidelines are needed for important and urgent questions that are of high concern and wide-ranging in clinical practice. Recommendations that reflect the characteristics of clinical practice of TCM can be formed based on expert clinical opinion and experience, normative documents, indirect evidence or qualitative research of TCM to guide and facilitate application of TCM treatments. Such recommendations are named TCM GPR.
4.2.2 Quality evaluation methods
Expert experience is a crucial component in formulating TCM guidelines, necessitating a comprehensive evaluation of its reliability, authority, completeness, and source. Various methods for evaluating these aspects are mentioned in the collected literature. However, these methods typically serve as evidence-quality grading methods, and since expert experience is not derived from clinical research, these grading methods should not be directly applied. Instead, they can be used as quality grading methods for the literature to better guide clinical decision-making for TCM practitioners.
4.2.3 Dissonance exploration
The Dissonance Exploration highlights the differences between the a priori framework and TCM GPR methodology model. These differences are categorized into two main scenarios: (i) themes (including sub-themes and categories) in the a priori framework that are not supported by coding in the TCM guidelines, and (ii) new themes (including sub-themes and sub-sub-themes) developed based on the coding guideline. The first scenario suggests that the theoretical approaches related to TCM GPR mentioned in methodological literature have not been systematically applied in practice. The second scenario indicates the limitations of these theoretical approaches. Both scenarios reflect a disconnection between theory and practice, underscoring the need for a TCM GPR methodology model.
Compared to the a priori framework, eight new sub-themes have been integrated into the TCM GPR methodology model, including five additional reporting guideline. While the methodological literature primarily focuses on the specific formulation of TCM GPR, the guidelines emphasize the standardization and normalization of reporting. Therefore, the TCM guidelines include increasingly detailed specifications for TCM GPR reporting and their presentation.
4.3 Model validation
4.3.1 Saturation test
Data saturation is commonly employed to assess the adequacy of research data in qualitative studies and serves as a critical criterion for evaluating article quality (Yang et al., 2022; Constantinou et al., 2017). In this study, the themes of the TCM GPR methodology model reached saturation. This indicates that the research data was sufficient and validates the rigor and qualitative robustness of the model development.
4.3.2 Structural rationality test
Confirmatory factor analysis was used to fit the real-world data from published TCM guidelines. The overall fit of the corrected final methodology model fell within an acceptable range, indicating a well-developed model.
This TCM GPR methodology model incorporates three themes as latent variables. The low intrinsic reliability of “TCM GPR procedures and methods” may stem from categorizing information from the same reference into different sub-themes. For instance, a reference using a particular method might be categorized under both “Development methods” and “Development procedures”, affecting intrinsic reliability. Additionally, the factor loading of “Conditions of Use” was below 0.4, possibly due to limited data in this sub-theme. At the same time, the AVE of “1 TCM GPR definition” was poor, probably due to the low factor loading of “conditions of use”.
Additionally, the weights of the three themes were calculated based on the standardized factor loadings between each theme and its subthemes, using the absolute value method. The specific approach involved summing the absolute values of the standardized factor loadings for all subthemes within each theme, then calculating the ratio of each theme’s total factor loading to the total factor loadings of all themes, which was used as the theme’s weight. The results showed that the weight of Theme 1 was 43.63%, the weight of Theme 2 was 34.43%, and the weight of Theme 3 was 21.96%. The theme weights reflect the relative importance or contribution of each theme within the overall model. Among the themes, the most important was “TCM GPR Definition,” followed by “TCM GPR Procedures and Methods,” and lastly “TCM GPR Reporting Guidelines.” The definition serves as the foundation for any practice recommendation and is the premise for all subsequent work. Without a clear definition, the formulation and reporting process will lose direction and consistency. Procedures and methods, on the other hand, are at the core of practice recommendations. Only through systematic procedures and scientific methods can the scientific and practical validity of the recommendations be ensured. Lastly, reporting guidelines are essential for the effective dissemination and implementation of the recommendations. Standardized reporting not only facilitates the execution of the recommendations but also provides crucial support for continuous improvement and monitoring evaluation.
4.3.3 Discriminability test
The results of the discriminability test revealed significant statistical differences in the scoring rates of guideline subgroups based on different basic features. This indicates that the TCM GPR methodology model can effectively differentiate between various types of TCM guidelines, demonstrating its discriminability.
4.4 Shortcomings and prospects
This study employed a qualitative method to develop the TCM GPR methodology model, but qualitative methods are prone to subjective biases. To mitigate this, a mixed-methods approach combining qualitative and quantitative research was adopted, utilizing quantitative methods to validate the developed model and ensure its scientific rigor.
Currently, the TCM GPR methodology model provides a general theoretical framework for TCM GPR. However, it lacks specific and clear operational specifications. Future research should focus on developing a more detailed, specific, and operational TCM GPR methodology tool, integrating existing guideline methodology tools to guide the precise formulation and application of TCM GPR (Zhou et al., 2022; Xie et al., 2020).
Data availability statement
The original data presented in the study can be found in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Author contributions
SL: Conceptualization, Data curation, Validation, Writing–original draft, Writing–review and editing, Software. LZ: Data curation, Methodology, Validation, Writing–original draft, Writing–review and editing, Software. YW: Data curation, Writing–review and editing, Investigation. RX: Data curation, Investigation, Writing–review and editing. WC: Conceptualization, Writing–review and editing. ML: Writing–review and editing. YA: Writing–review and editing. AS: Writing–review and editing. HH: Writing–review and editing. HL: Conceptualization, Methodology, Supervision, Writing–original draft, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This work was supported by National Natural Science Foundation of China (72204061), the Special Research Fund for Traditional Chinese Medicine Science and Technology of Guangdong Provincial Hospital of Chinese Medicine (YN2023QN05) and Research Project of State Key Laboratory of Traditional Chinese Medicine Syndrome (QZ2023ZZ04).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2025.1501634/full#supplementary-material
Abbreviations
TCM, traditional Chinese medicine; GPR, good practice recommendation; CACM, China Association of Chinese Medicine; RMSEA, root mean square error of approximation; GFI, goodness of fit index; AGFI, adjusted goodness of fit index; CR, composite reliability; AVE, average variance extracted.
References
Balshem, H., Helfand, M., Schünemann, H. J., Oxman, A. D., Kunz, R., Brozek, J., et al. (2011). GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 64 (4), 401–406. doi:10.1016/j.jclinepi.2010.07.015
Carroll, C., Booth, A., and Cooper, K. (2011). A worked example of “best fit” framework synthesis: a systematic review of views concerning the taking of some potential chemopreventive agents. BMC Med. Res. Methodol. 11, 29–9. doi:10.1186/1471-2288-11-29
Carroll, C., Booth, A., Leaviss, J., and Rick, J. (2013). “Best fit” framework synthesis: refining the method. BMC Med. Res. Methodol. 13, 37–16. doi:10.1186/1471-2288-13-37
Chang, W. J., Liu, J. P., and Chen, W. (2022). The role of expert opinion of prestigious Chinese medicine doctors in the development of clinical practice guidelines for Chinese medicine. CJITWM 42, 1118–1123. doi:10.7661/j.cjim.20220809.087
Chen, G. Z., Wang, S. C., Xu, S., and Luo, H. (2013). Methodological study on integrating the ancient and modern doctor’s experience into evidence-based clinical practice guidelines of traditional Chinese medicine. CJTCMP 28, 3609–3611.
Chen, W., Fang, S., and Liu, J. (2019). Recommendations for clinical evidence grading on traditional Chinese medicine based on evidence body. CJITWM 39 (3), 358–364. doi:10.7661/j.cjim.20190123.062
Constantinou, C. S., Georgiou, M., and Perdikogianni, M. (2017). A comparative method for themes saturation (CoMeTS) in qualitative interviews. Qual. Res. 17 (5), 571–588. doi:10.1177/1468794116686650
Dewidar, O., Lotfi, T., Langendam, M. W., Parmelli, E., Parkinson, Z. S., Solo, K., et al. (2023). Good or best practice statements: proposal for the operationalisation and implementation of GRADE guidance. BMJ evidence-based Med. 28 (3), 189–196. doi:10.1136/bmjebm-2022-111962
Diekemper, R. L., Patel, S., Mette, S. A., Ornelas, J., Ouellette, D. R., and Casey, K. R. (2018). Making the GRADE: CHEST updates its methodology. Chest 153 (3), 756–759. doi:10.1016/j.chest.2016.04.018
Djulbegovic, B., and Guyatt, G. H. (2017). Progress in evidence-based medicine: a quarter century on. lancet 390 (10092), 415–423. doi:10.1016/S0140-6736(16)31592-6
Ge, L. L., Cao, X., and Wu, D. R. (2022). Evidence-based clinical practice guidelines on tuina for children with anorexia (2021 edition). J. Tradit. Chin. Med. 63, 1295–1300. doi:10.13288/j.11-2166/r.2022.13.018
Guo, X., Liu, B. Y., Liu, Z. S., and Zhao, H. (2008). Discussing the characteristics of structure and methodology in the evidence-based guidelines of acupuncture for clinical practice. J. Evid. Based Med. 8, 105–108. doi:10.3969/j.issn.1671-5144.2008.02.012
Guyatt, G., Cairns, J., Churchill, D., Cook, D., Haynes, B., Hirsh, J., et al. (1992). Evidence-based medicine: a new approach to teaching the practice of medicine. Jama 268 (17), 2420–2425. doi:10.1001/jama.1992.03490170092032
Guyatt, G. H., Alonso-Coello, P., Schünemann, H. J., Djulbegovic, B., Nothacker, M., Lange, S., et al. (2016). Guideline panels should seldom make good practice statements: guidance from the GRADE Working Group. J. Clin. Epidemiol. 80, 3–7. doi:10.1016/j.jclinepi.2016.07.006
Guyatt, G. H., Oxman, A. D., Vist, G. E., Kunz, R., Falck-Ytter, Y., Alonso-Coello, P., et al. (2008). GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. Bmj 336 (7650), 924–926. doi:10.1136/bmj.39489.470347.AD
Guyatt, G. H., Schünemann, H. J., Djulbegovic, B., and Akl, E. A. (2015). Guideline panels should not GRADE good practice statements. J. Clin. Epidemiol. 68 (5), 597–600. doi:10.1016/j.jclinepi.2014.12.011
Liu, J. P. (2007). Composition of the evidence body in traditional medicine and recommendations for its evidence grading. Chin. J. Integr. Tradit. West Med. 12, 1061–1065. doi:10.3321/j.issn:1003-5370.2007.12.001
Liu, Q. Q., Chen, T. F., Zhao, G. Z., and Liu, J. P. (2021). Clinical practice guideline of traditional Chinese medicine for influenza (2021). J. Tradit. Chin. Med. 24, 4155–4159. doi:10.13288/j.11-2166/r.2022.01.016
Lopez, V. K., Nika, A., Blanton, C., Talley, L., and Garfield, R. (2023). Can severity of a humanitarian crisis be quantified? Assessment of the INFORM severity index. Glob. health 19 (1), 7. doi:10.1186/s12992-023-00907-y
Marsh, H. W., Balla, J. R., and McDonald, R. P. (1988). Goodness-of-fit indexes in confirmatory factor analysis: the effect of sample size. Psychol. Bull. 103 (3), 391–410. doi:10.1037/0033-2909.103.3.391
Naidoo, L., and Fields, Z. (2019). Critical success factors for the successful initiation of Lean in public hospitals in KwaZulu-Natal: a factor analysis and structural equation modelling study. Hum. Resour. health 17, 69–11. doi:10.1186/s12960-019-0405-1
Oxman, A. D., Schünemann, H. J., and Fretheim, A. (2006). Improving the use of research evidence in guideline development: 7. Deciding what evidence to include. Health Res. Policy Syst. 4, 19–27. doi:10.1186/1478-4505-4-19
Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., Mulrow, C. D., et al. (2021). The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Bmj-British Med. J. 372, n71. doi:10.1136/bmj.n71
Pires, MRGM, Almeida, A.N. D., Gottems, L. B. D., Mundim, G. D. A., and Oliveira, R. N. G. D. (2022). Validation of Ludic-Quest to the playfulness of health games: gameplay and emotions on the field. Rev. Bras. Enferm. 75 (04), e20210822. doi:10.1590/0034-7167-2021-0822
Thomas, J., and Harden, A. (2008). Methods for the thematic synthesis of qualitative research in systematic reviews. BMC Med. Res. Methodol. 8, 45–10. doi:10.1186/1471-2288-8-45
Vermeulen, N., Le Clef, N., Veleva, Z., D'Angelo, A., and Tilleman, K. (2019). European Recommendations for good practice in addition to an evidence-based guidelines programme: rationale and method of development. Bmj Evidence-Based Med. 24 (1), 30–34. doi:10.1136/bmjebm-2018-111032
Verwoerd, A., Armbrust, W., Cowan, K., van den Berg, L., de Boer, J., Bookelman, S., et al. (2021). Dutch patients, caregivers and healthcare professionals generate first nationwide research agenda for juvenile idiopathic arthritis. Pediatr. Rheumatol. 19, 52–10. doi:10.1186/s12969-021-00540-2
Wang, S., Chen, Z., Xu, S., Zhao, X., and Yu, S. (2013). Study on evidence grading system in evidence-based clinical practice guidelines of traditional Chinese medicine. World Sci. Technology-Modernization Traditional Chin. Med., 1488–1492. doi:10.11842/wst.2013.07.001
Warlick, C., Richter, K. P., Catley, D., Gajewski, B. J., Martin, L. E., and Mussulman, L. M. (2018). Two brief valid measures of therapeutic alliance in counseling for tobacco dependence. J. Subst. Abuse Treat. 86, 60–64. doi:10.1016/j.jsat.2017.12.010
Xie, R., Xia, Y., Chen, Y., Li, H., Shang, H., Kuang, X., et al. (2020). The RIGHT extension statement for Traditional Chinese Medicine: development, recommendations, and explanation. Pharmacol. Res. 160, 105178. doi:10.1016/j.phrs.2020.105178
Yan, W., Caihong, H., Xuefeng, Y., and Jiayu, Z. (2023). Evaluation of the nutrition literacy assessment questionnaire for college students and identification of the influencing factors of their nutrition literacy. BMC Public Health 23 (1), 2127. doi:10.1186/s12889-023-17062-z
Yang, L., Qi, L., and Zhang, B. (2022). Concepts and evaluation of saturation in qualitative research. Adv. Psychol. Sci. 30 (3), 511–521. doi:10.3724/sp.j.1042.2022.00511
Keywords: traditional chinese medicine (TCM), guidelines, good practice recommendation (GPR), methodology model, mixed method
Citation: Li S, Zhang L, Wang Y, Xie R, Chen W, Lee MS, Amer YS, Sharifan A, Hussein H and Li H (2025) Development and validation a methodology model for traditional Chinese medicine good practice recommendation: an exploratory sequential mixed methods study. Front. Pharmacol. 16:1501634. doi: 10.3389/fphar.2025.1501634
Received: 25 September 2024; Accepted: 13 January 2025;
Published: 31 January 2025.
Edited by:
Anthony Booker, University of Westminster, United KingdomReviewed by:
Selen Yeğenoğlu, Hacettepe University, TürkiyeJingjing Wei, China Academy of Chinese Medical Sciences, China
Copyright © 2025 Li, Zhang, Wang, Xie, Chen, Lee, Amer, Sharifan, Hussein and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hui Li, bGlodWl0Y21AMTI2LmNvbQ==
 Su Li1,2,3
Su Li1,2,3 Luan Zhang
Luan Zhang Yangyang Wang
Yangyang Wang Hui Li
Hui Li