- 1Department of Microbiology, ICMR-Regional Medical Research Centre (Department of Health Research, Ministry of Health and Family Welfare, Government of India), Bhubaneswar, Odisha, India
- 2Division of Epidemiology and Communicable Diseases, Indian Council of Medical Research, New Delhi, India
Background: Antimicrobial resistance (AMR) has emerged as one of the major public health issues globally. This cross-sectional study determined knowledge, attitudes and practices (KAP) regarding antimicrobial use and AMR among rural communities of Tigiria (Odisha), India.
Methods: A semi-structured questionnaire based on socio-demographic characteristics, antibiotics usage, awareness of antimicrobial resistance, healthcare utilization and quality of life were asked to the participants using an electronic device with Open Data Kit. Descriptive statistics, independent t-test and ANOVA were performed to analyze the variables.
Results: A total of 1,003 participants were surveyed in the study from 25 villages of Tigiria. About 44.47% (95% CI: 41.36–47.60) of study participants have heard about antimicrobial medicines and 14.75% (95% CI: 12.65–17.13) of participants were involved in buying antibiotics without prescription over the counter. Around 20.14% (95% CI: 17.72–22.78) of participants, stopped taking antibiotics before completing the full course. The physical domain was the most affected with low scores compared to other domains of quality of life (QOL). The QOL scores were found significant (p < 0.05) across age, gender, education and ethnicity.
Conclusion: The study documented a significant level of KAP regarding antimicrobial (mis)use in the study. It is essential that antimicrobial stewardship programs for various stakeholders and educational programmes must be initiated to increase awareness of people on antimicrobial resistance.
1. Introduction
The development of antibiotics has dramatically transformed medical care and resulted in a global decline in mortality and morbidity from infectious diseases (1). Due to the rise of antimicrobial resistance (AMR), the efficiency of currently used antimicrobial drugs is declining, making it more difficult and expensive to treat illnesses and harder to manage outbreaks. This has emerged as one of the major public health issues globally (2). Antimicrobial-resistant organisms (AMRO) have emerged globally as a result of microbial evolution, antimicrobial misuse and abuse in veterinary, human, and agricultural settings and poor infection control procedures, among other factors as key contributors to the global rise of AMR. This has resulted in difficult-to-treat infections, prolonged hospital stays, higher healthcare costs, and poor health outcomes (3, 4).
Developing nations in Asia and Africa are at greater risk of antimicrobial resistance (AMR), even though it's a matter of global concern. It is predicted that by 2050 over 10 million people would die from the disease worldwide including roughly 4 million people each from Asia and Africa, if no action is taken to stop the spread of AMR (5). The 68th World Health Assembly, which took place in May 2015, saw the adoption of a Global Action Plan (GAP) against AMR by its member nations (6). This strategy mandated that each nation create and carry out a National Action Plan for Antimicrobial Resistance (NAPAR) for combating AMR. The World Health Organization (WHO) is urging its member nations to adopt and put into practice the updated Model List of Essential Medicines, which divided antibiotics into the categories of Access, Watch, and Reserve (AWaRe) (7). The AWaRe categorization aims to increase access to antibiotics that can save lives and avoid the development of resistance from overuse of the selected priority medications.
In the Low and Middle-Income Countries (LMICs), a systematic study has revealed an increase of 77% in the rate of antibiotic consumption and an overall 114% rise in antibiotic consumption from 2000 to 2015 (8). During the year 2000–2010, a 76% rise in global antibiotic consumption was collectively contributed by BRICS countries, i.e., Brazil, Russia, India, China and South Africa (9). Unfortunately, India has one of the highest rates of antibiotic consumption with a massive 23% when compared to the other BRICS nations (9). According to a recent study in India, it is reported that the total defined daily doses (DDD) was 5,071 million in 2019 and the defined daily doses per 1,000 people per day (DID) was 10.4. Azithromycin was the most consumed antibiotic molecule (12.6%) followed by cefixime (10.2%) (10). As per the scoping report in 2017, more than 70% of the isolates of the Gram-negative bacteria, i.e., Escherichia coli, Klebsiella pneumoniae, and Acinetobacter baumannii as well as almost half of all Pseudomonas aeruginosa were resistant to fluoroquinolones and third generation cephalosporins. Gram-positive bacteria such as Staphylococcus aureus had a methicillin resistance rate of 42.6% while Enterococcus faecium had a vancomycin resistance rate of 10.5%. For ciprofloxacin, resistance rates among Salmonella typhi and Shigella species were 28% and 82%, respectively. Tetracycline resistance rates for Vibrio cholerae ranged from 17 to 75% in different parts of the country (11).
There is a lack of AMR research examining the complexity of upstream and downstream issues in a country like India, particularly in areas with limited resources. There is a need for generating evidence from well-designed studies from the community to execute policy development and implementation to prevent the irrational use of antibiotics and reduce the spread of AMR. The following study is a quantitative study that was conducted to map the knowledge relating to antibiotics, antibiotic resistance and antibiotic usage practices among the rural communities of Tigiria, Cuttack district of Odisha. Healthcare utilization and the quality of life from the study participants were also recorded during the survey.
2. Methodology
2.1. Study design
It is a cross-sectional study conducted to assess the knowledge, attitude and practices prevalent among rural communities regarding antibiotic usage and antimicrobial resistance in the state of Odisha, India. This study was conducted from November 2021 to January 2021.
2.2. Study settings
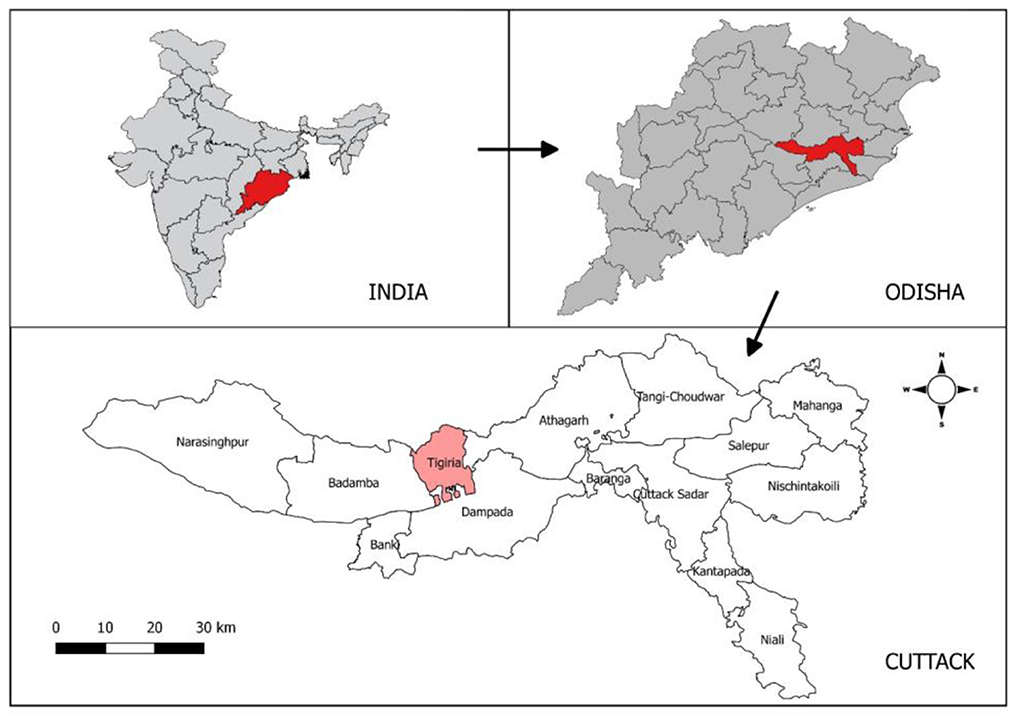
The study was undertaken in the rural settings of Tigiria block (“Block” is a district subdivision), Cuttack district which is situated in the coastal area of Odisha with 50 villages under it. Tigiria block is located at 20°29′0″N latitude and 85°31′0″E longitude. Tigiria has a tropical savanna type of climate where the average summer temperature is 45°C and the average winter temperature is 10°C. Tigiria receives 964 mm of rainfall annually. Average relative humidity varies from 55 to 79% throughout the year. The total population of the Tigiria block is 75,000 according to 2011 census. The study area map is given in Figure 1.
2.3. Sample size
The sample size was calculated using the formula (4pq/d2)*DE, where p = awareness of antibiotics, q = (1 – p), d = relative precision and DE = design effect. Assuming an awareness of antibiotics to be 50%, design effect of 1.3, relative precision of 4% and a non-response rate of 10%, the sample size was calculated to be 972 which was rounded off to 1,000.
2.4. Sampling method
Cluster sampling method was adopted for selecting study participants from the Tigiria block. There are 50 villages in Tigiria block out of which 25 villages were selected from the cluster sampling method. From each village, 40 households were selected for the survey. In each village, households were chosen through systematic random sampling method and only one adult individual was enrolled from each selected house. Simple random selection was used to choose a participant for the study if there were two or more adults living in a single household.
2.5. Data collection
An 86-item based questionnaire was developed to collect information from the study participants. The questionnaire consisted of semi-open questions based on socio-demographic characteristics, antibiotics usage, awareness of antimicrobial resistance, healthcare utilization and quality of life (WHO-QOL BREF scale) (12). The questionnaire was uploaded to electronic devices using an Open Data Kit. Questionnaire was developed in English and translated into the local language for a better understanding of the rural people. Hands-on training was provided to the field staff for collecting the data through electronic devices.
2.6. Data analysis
Data analysis was done using Statistical Package for Social Sciences (SPSS) version 21 (IBM®, USA). Distribution of frequency, percentages of variables and 95% confidence intervals (CI) were calculated for categorical variables. Continuous variables were calculated as mean with standard deviation (SD). Independent t-test and one-way analysis of variance (ANOVA) were used to determine the association of socio-demographic variables with WHOQOL-BREF domains. All tests of significance were two-tailed with a p < 0.05 indicating a statistically significant difference. Study area map was made using QGIS v3.10 which is an open-source software freely available on the internet.
2.7. Ethical concerns
Ethical clearance was obtained from the Institutional Human Ethics Committee of ICMR-Regional Medical Research Centre (RMRC), Bhubaneswar (ICMR-RMRCB/IHEC-2019/034) and the State Ethical Committee of Odisha. Each participant was explained about the aims and objectives of the study and provided with the participant information sheet for reference. Written informed consent was taken from every participant before taking part in the study.
3. Results
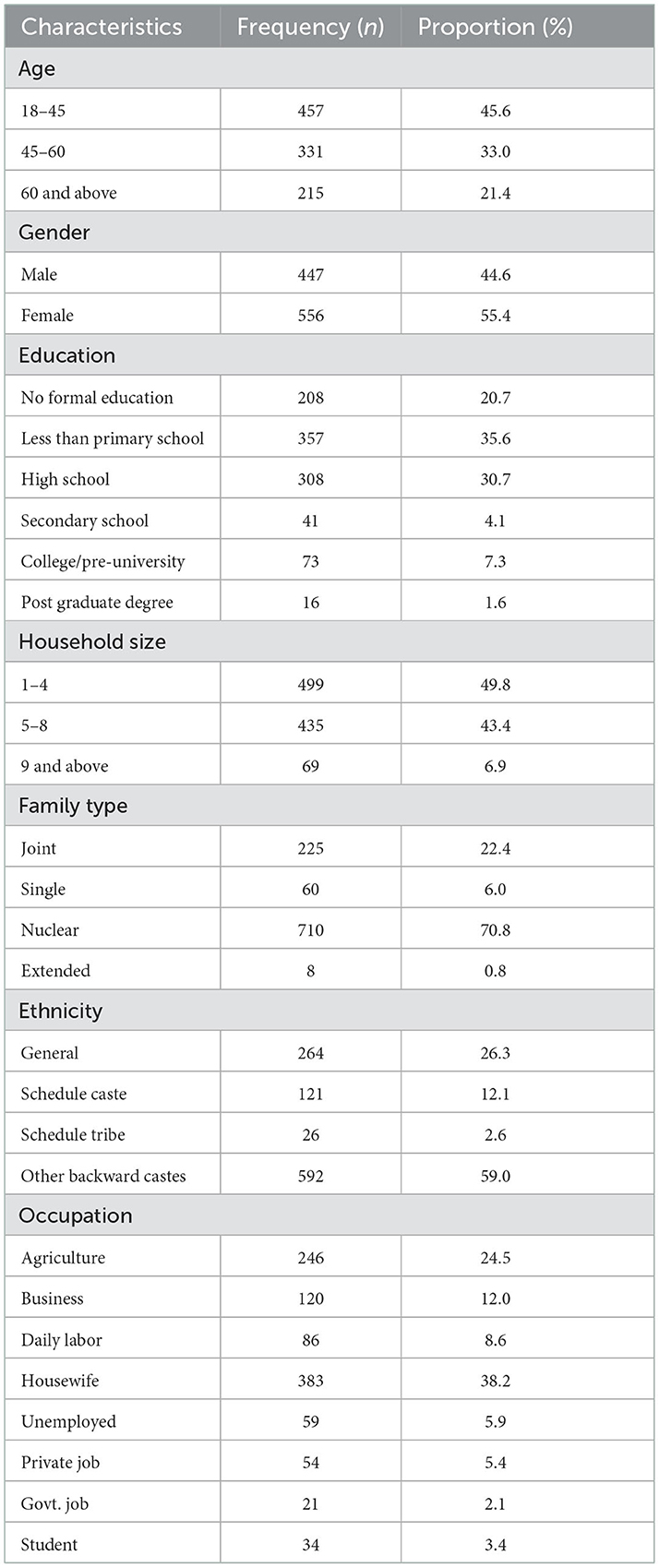
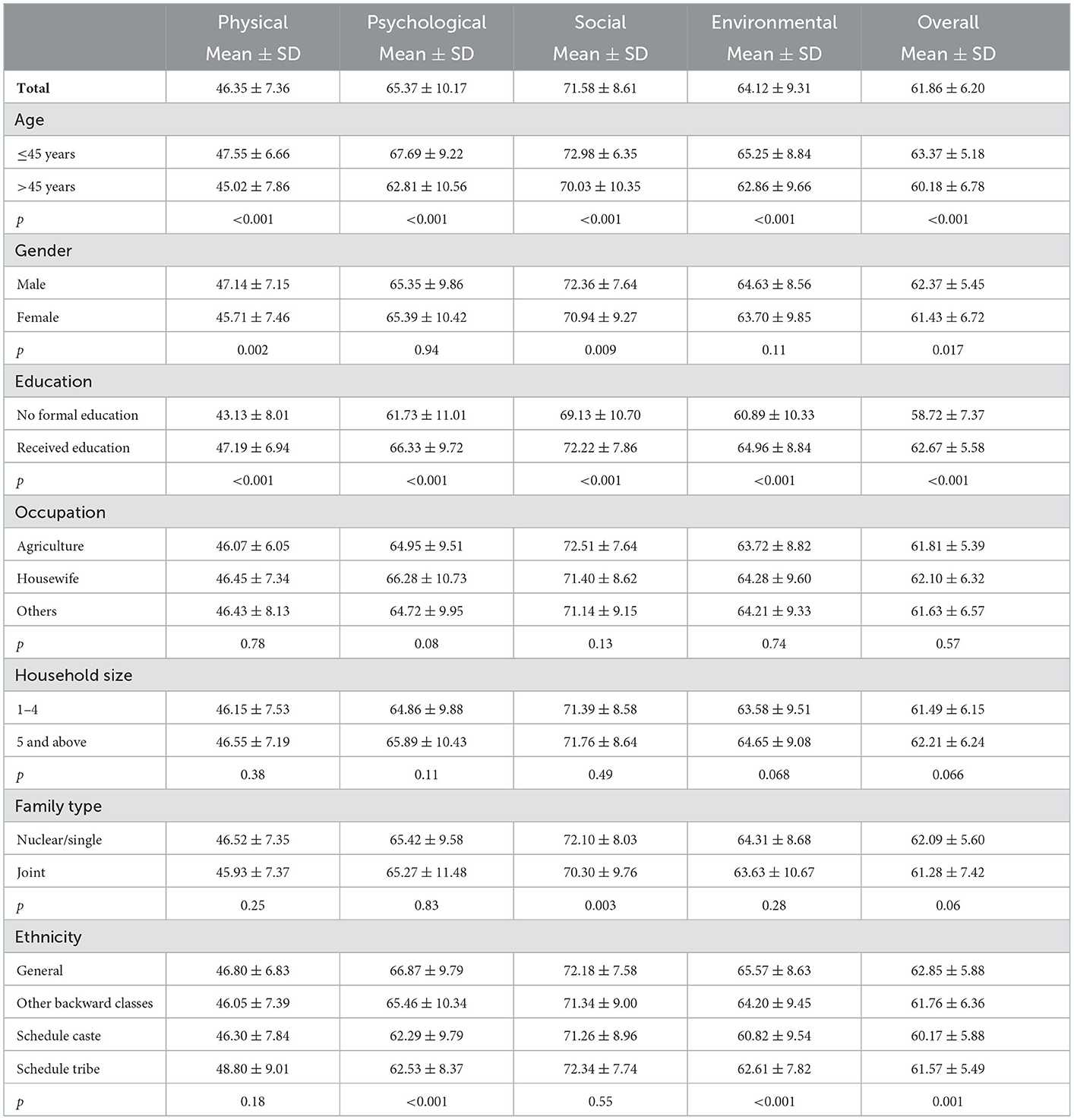
A total of 1003 participants were surveyed in the study and most of the participants were from the 18 to 45 years age group. The proportion of female participants was more than the male participants. Around 20.7% (n = 208) of the study population received no formal education, whereas 30.7% of them have completed schooling (Table 1). About 50% (n = 499) of the houses consist of household members around 1–4. More than 70% (n = 710) of the houses had nuclear family structure, followed by joint family (22.4%). About 59% of the study population belonged to other backward castes, followed by the general population (26.3%). Occupation of these study participants were as follows in increasing order of their proportion, housewife (38.2%), agriculture (24.5%) and business (12%).
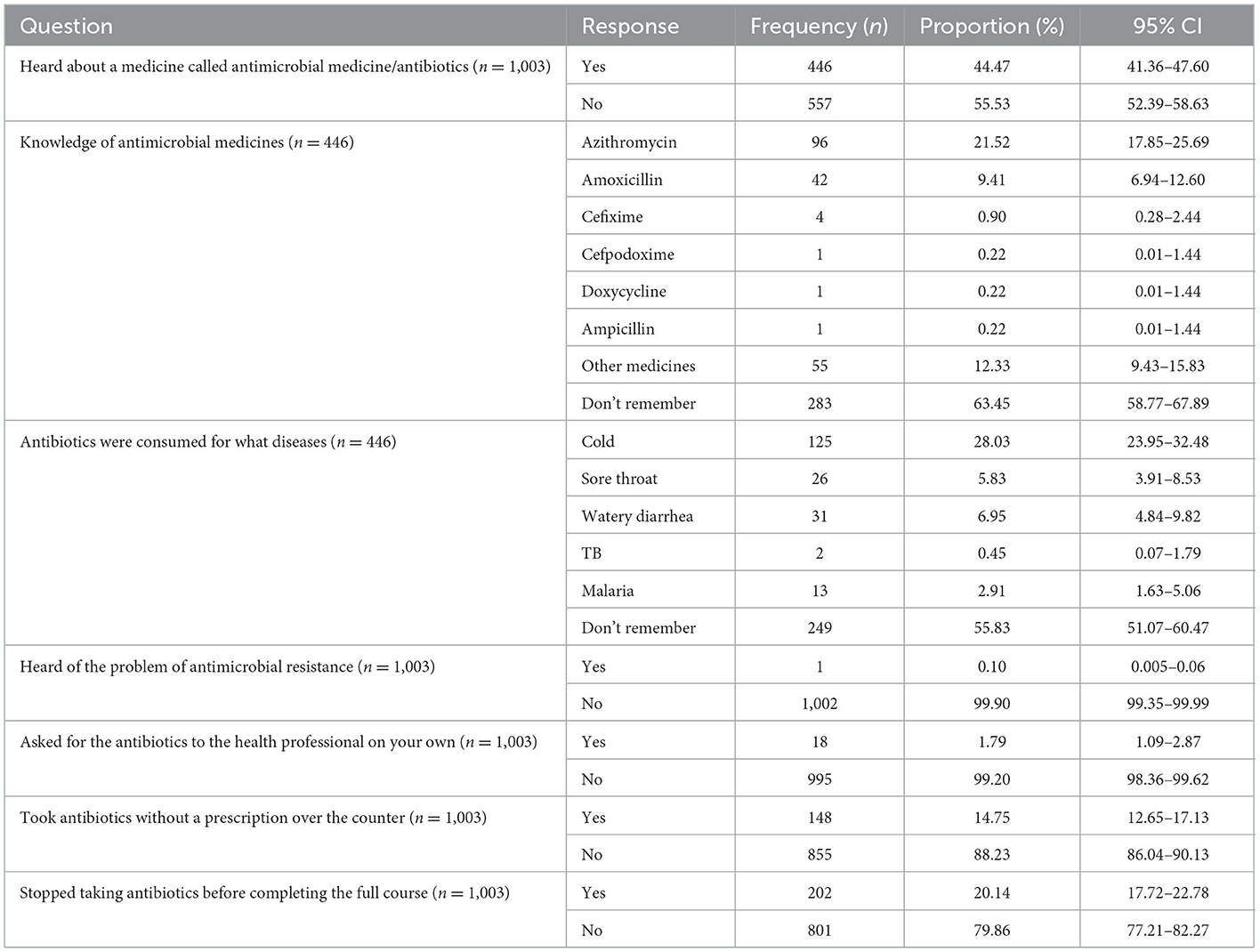
Almost 44.47% (n = 446) of study participants have heard about antimicrobial medicines (Table 2). About 21.52% (n = 96) of participants knew about azithromycin and 9.41% (n = 42) about amoxicillin among the participants who have heard about antimicrobial medicines. About 28.03% (95% CI: 23.95–32.48) of participants took antibiotics for cold, 5.83% (95% CI: 3.91–8.53) for sore throat, 6.95% (95% CI: 4.84–9.82) for watery diarrhea, and 2.91% (95% CI: 1.63–5.06) for malaria. However, 14.75% (95% CI: 12.65–17.13) of participants were involved in buying antibiotics without prescription over-the-counter and 20.14% (95% CI: 17.72–22.78) of participants, stopped taking antibiotics before completing the full course. Similarly, 1.79% (95% CI: 1.09–2.87) of participants ask for antibiotics from health professionals on their own. Only 0.10% (95% CI: 0.005–0.06) of study participants knew about antimicrobial resistance.
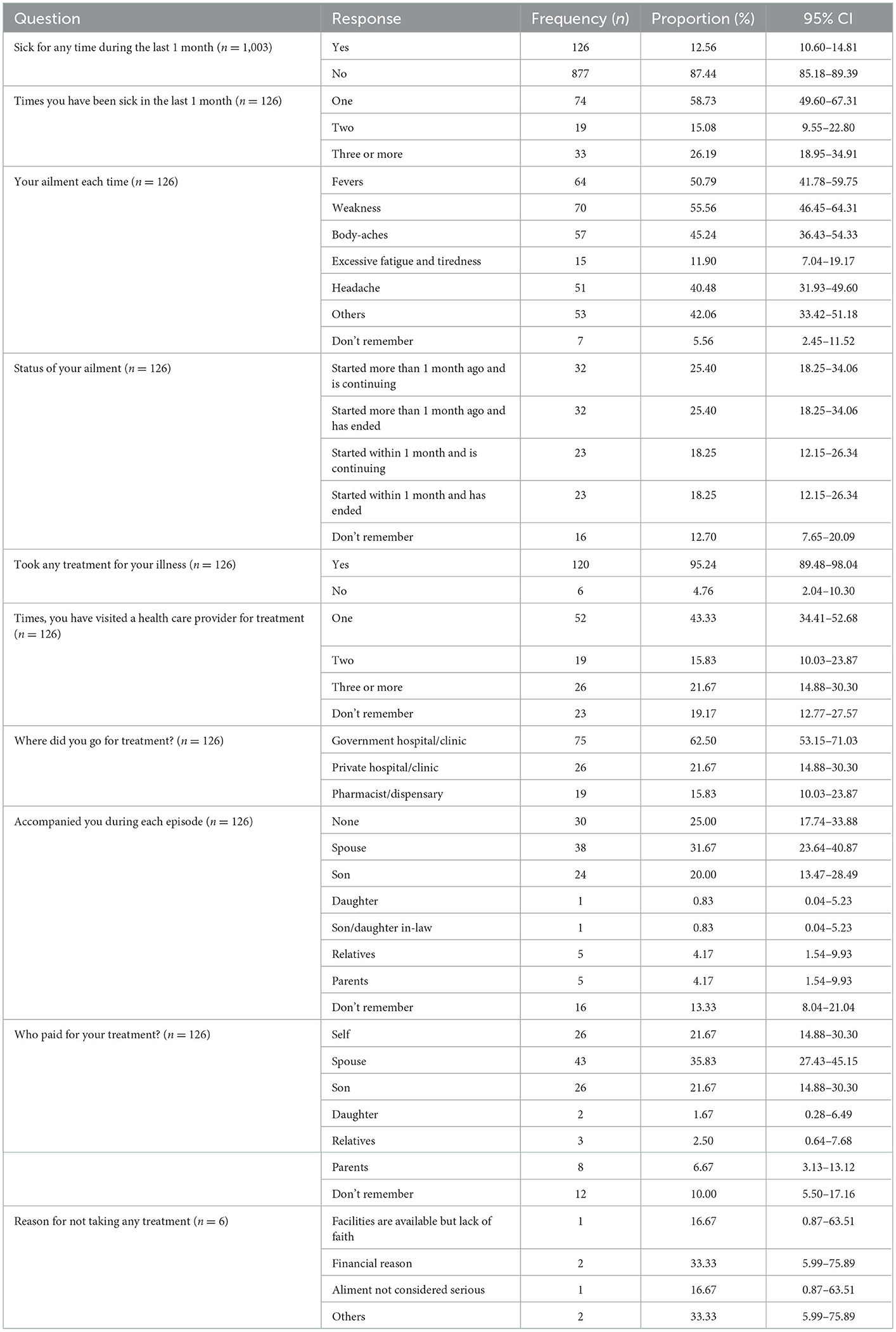
Participants were also asked about their healthcare utilization, and it was seen that 12.56% (n = 126) of the participants were sick during the last month of the interview (Table 3). Out of those 126 participants, 74 (58.73%) got sick once, 19 (15.08%) got twice and 33 (26.19%) got sick thrice in the last month. Participants had different ailments such as fever 64 (50.79%), weakness 70 (55.56%), body aches 57 (45.24%) and headaches 51 (40.48%). About 120 (95.24%) of the participants went for pre-hospitalization and 75 (62.50%) went to a government hospital/clinic for treatment.
The mean score across the four domains and the overall domain of WHOQOL-BREF based on sociodemographic factors was shown in Table 4. QOL of younger age participants (≤45 years) were observed with better scores significant (p < 0.05) in all their domains. Male participants had better QOL in physical, social and overall domains which were statistically significant (p < 0.05). With regard to education, illiterate participants had poor QOL in all domains when compared to educated participants (p < 0.05). Similarly, nuclear/single families displayed better QOL than joint families only in the social domain (p < 0.05). The QOL scores were found significant (p < 0.05) across ethnicity.
4. Discussion
This study represents one of the attempts to assess the knowledge, attitude and practices of antimicrobial use and antimicrobial resistance in a rural setting in the eastern region of India. It is expected that the findings from the present study will provide baseline information related to understanding the use or misuse of antibiotics in rural Indian communities. However, there is insufficient knowledge about antimicrobial resistance in the rural community. The level of knowledge observed in our study about antimicrobials and their use is similar to the findings from Jordan and Nepal (13, 14). The lack of knowledge regarding antibiotic use and antimicrobial resistance is in accordance with the previous findings (13, 15). An increase in participants' education is an important criterion that depicts their knowledge gain about antibiotic use and antimicrobial resistance which was reported in the findings from earlier studies (13, 15–17). More than 50% of the participants were unaware of AMR similar to the finding reported in Nepal (14). The level of knowledge on AMR seen in our study, however, is lower than that reported in Norway (18). Very few studies have been done among the general population to determine their knowledge of AMR as seen in the literature. Studies have primarily focused on individuals or specific groups such as medical professionals, veterinary or students (17, 19, 20).
In the study, it was observed that participants have heard about antibiotics but as many of them have not received complete education, they were not able to tell the antibiotic medicines, they had used. Nearly one-seventh of the participants preferred taking antimicrobial medicines without a prescription over the counter or without consultation with healthcare workers. This was an unhealthy method of taking antibiotics in the rural population. Participants directly asking the health professionals to prescribe antibiotics was minimal. Very few people stopped taking the antibiotics before completing the full course which might be due to their compliant nature with the healthcare workers or for the fear of not getting completely cured. However, the knowledge of antimicrobial resistance in the rural community is almost negligible when compared with the other studies (13–16).
In the community, AMR is primarily caused by self-medication or inappropriate use of antimicrobials (21). However, in these settings, evidence-based antimicrobial stewardship programmes are very limited. Education must be a key element of antimicrobial stewardship programmes in order for them to be successful. Healthcare workers must be involved in antimicrobial stewardship programmes so that antibiotics can be preserved, AMR and substantial morbidity and mortality can be reduced. The distinctive legal and cultural traits of the study populations should be taken into account while designing intervention plans. The intervention can only be successful when both the healthcare workers and community work as a whole to reduce AMR.
Inadequate awareness of antibiotic use and AMR should be seen as worrying issues and possible signs of AMR that must be addressed right away. The data from this study might be used by policy- and decision-makers as one of the inputs to monitor drug usage policy and recommendations and reduce the risk of AMR. Our findings advocate for customized treatments, such as community-focused education campaigns about optimal antibiotic usage and AMR prevention strategies. For example, persons with less formal education, who have shown less understanding and poor practices about the use of antibiotics and steps to combat their resistance, should be the focus of education programmes.
The healthcare system was efficiently utilized by the study population across all age groups. It shows that the patients had better access to the healthcare system showing higher utilization in the rural area which might be due to multiple factors such as better transportation, etc. The physical domain of life analysis, which addressed working ability, daily activities, bodily discomfort, sleep and rest, mobility and energy received the lowest score as per the WHO-QOL BREF scale. The QOL scores in other domains were higher than the physical domain and were found to be significant across age, gender, education and ethnicity.
There are very few studies have been conducted on antimicrobial use and AMR in rural settings, especially in India which is one of the major advantages of our study. The sample size is also big and generalizable inferences can also be drawn from a similar setting. However, the study has some limitations such as the data might have been affected by recall bias or fear among rural study participants of not sharing undesirable practices. To draw causal inferences from this study is not possible as the study is cross-sectional in nature. Besides these limitations, the data from the study can provide baseline information that can help to carry out future research and track the efficiency of intervention studies.
5. Conclusion
This study provides information on knowledge, attitude and practices on antimicrobial usage and AMR from rural settings of Tigiria, Odisha. The study documented a low level of knowledge and a high level of practice on antimicrobial usage and AMR. The findings are crucial for directing policy development, programme planning, in execution of antimicrobial use and AMR-related initiatives focusing the community-based awareness, education, and sensitization.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving human participants were reviewed and approved by Institutional Ethical Committee, RMRC, Bhubaneswar. The participants provided their written informed consent to participate in this study.
Author contributions
SPat, DB, KW, and TS were involved in the concept, planning, and formulation of the study. AN, SKar, SS, DP, and HC were involved in data collection. Data analysis and the initial draft were prepared by MP. SPal, SKan, and JK reviewed the manuscript. All the authors have read and approved the final version of the manuscript.
Funding
The study received funding from the Indian Council of Medical Research, New Delhi, India [File No./AMR/194/2019/ECD-II (Pt) dated 12.01.2021].
Acknowledgments
We would like to acknowledge and thank our field research staff including Santosh Kumar Behuria, Dasharatha Majhi, Asit Biswal, Jeevan Kumar Mohanta, Partha Sarathi Patra and Pooja Pattanayak. We are thankful to the State Health Department, Odisha for their timely support and coordination. We also acknowledge all the stakeholders from different departments who contributed their time and efforts for their help in our study. Finally, we are grateful to our funders; the Indian Council of Medical Research, New Delhi, India.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Aminov RI. A brief history of the antibiotic era: lessons learned and challenges for the future. Front Microbiol. (2010) 1:134. doi: 10.3389/fmicb.2010.00134
2. Byarugaba DK. Antimicrobial resistance in developing countries and responsible risk factors. Int J Antimicrob Agents. (2004) 24:105–10. doi: 10.1016/j.ijantimicag.2004.02.015
3. Friedman ND, Temkin E, Carmeli Y. The negative impact of antibiotic resistance. Clin Microbiol Infect. (2016) 22:416–22. doi: 10.1016/j.cmi.2015.12.002
4. World Health Organisation. Antibiotic Resistance (2020). Available online at: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance (accessed August 3, 2022).
5. O'Neill J. Antimicrobial Resistance: Tackling a Crisis for the Health Wealth of Nations (2014). Available online at: https://amr-review.org/sites/default/files/AMR.Reviewpaper-tacklingacrisisforthehealthandwealthofnations_1.pdf (accessed August 3, 2022).
6. World Health Organisation. Global Action Plan on Antimicrobial Resistance. Geneva: World Health Organization (2016). Available online at: http://www.who.int/antimicrobial-resistance/global-action-plan/en/ (accessed August 3, 2022).
7. World Health Organisation. Report of the 21st WHO Expert Committee on the Selection and Use of Essential Medicines. Geneva: WHO Headquarters (2017). Available online at: http://www.who.int/selection_medicines/committees/subcommittee/2/eeb1098%5B1%5D.pdf (accessed August 9, 2022).
8. Ventola CL. The antibiotic resistance crisis: part 1: causes and threats. Pharm Ther. (2015) 40:277.
9. Patel I, Hussain R, Khan A, Ahmad A, Khan MU, Hassalai MA. Antimicrobial resistance in India. J Pharm Policy Pract. (2017) 10:1–2. doi: 10.1186/s40545-017-0118-6
10. Koya SF, Ganesh S, Selvaraj S, Wirtz VJ, Galea S, Rockers PC. Consumption of systemic antibiotics in India in 2019. Lancet Regional Health-Southeast Asia. (2022) 4:100025. doi: 10.1016/j.lansea.2022.100025
11. Gandra S, Joshi J, Trett A, Lamkang A, Laxminarayan R. Scoping Report on Antimicrobial Resistance in India. Washington, DC: Center for Disease Dynamics, Economics Policy (2017). Available online at: https://dbtindia.gov.in/sites/default/files/ScopingreportonAntimicrobialresistanceinIndia.pdf (accessed December 18, 2022).
12. Skevington SM, Lotfy M, O'Connell KA, WHOQOL Group. The World Health Organization's WHOQOL-BREF quality of life assessment: psychometric properties and results of the international field trial A report from the WHOQOL group. Qual Life Res. (2004) 13:299–310. doi: 10.1023/B:QURE.0000018486.91360.00
13. Nepal A, Hendrie D, Robinson S, Selvey LA. Knowledge, attitudes and practices relating to antibiotic use among community members of the Rupandehi District in Nepal. BMC Public Health. (2019) 19:1558. doi: 10.1186/s12889-019-7924-5
14. Shehadeh M, Suaifan G, Darwish RM, Wazaify M, Zaru L, Alja'fari S. Knowledge, attitudes and behavior regarding antibiotics use and misuse among adults in the community of Jordan. A pilot study. Saudi Pharm J. (2012) 20:125–33. doi: 10.1016/j.jsps.2011.11.005
15. Goodluck M, Basiliana E, Maseke RM, Geofrey NS, Debora CK. Community knowledge and attitudes on antibiotic use in Moshi Urban, Northern Tanzania: findings from a cross sectional study. Afr J Microbiol Res. (2017) 11:1018–26. doi: 10.5897/AJMR2017.8583
16. Gebeyehu E, Bantie L, Azage M. Inappropriate use of antibiotics and its associated factors among urban and rural communities of Bahir Dar city administration, Northwest Ethiopia. PLoS ONE. (2015) 10:e0138179. doi: 10.1371/journal.pone.0138179
17. Azevedo MM, Pinheiro C, Yaphe J, Baltazar F. Portuguese students' knowledge of antibiotics: a cross-sectional study of secondary school and university students in Braga. BMC Public Health. (2009) 9:359. doi: 10.1186/1471-2458-9-359
18. Waaseth M, Adan A, Røen IL, Eriksen K, Stanojevic T, Halvorsen KH, et al. Knowledge of antibiotics and antibiotic resistance among Norwegian pharmacy customers—a cross-sectional study. BMC Public Health. (2019) 19:66. doi: 10.1186/s12889-019-6409-x
19. Jamshed SQ, Elkalmi R, Rajiah K, Al-Shami AK, Shamsudin SH, Siddiqui MJA, et al. Understanding of antibiotic use and resistance among final year pharmacy and medical students: a pilot study. J Infect Dev Ctries. (2014) 8:780–5. doi: 10.3855/jidc.3833
20. Seid MA, Hussen MS. Knowledge and attitude towards antimicrobial resistance among final year undergraduate paramedical students at University of Gondar, Ethiopia. BMC Infect Dis. (2018) 18:312. doi: 10.1186/s12879-018-3199-1
Keywords: knowledge, attitude, practices, antibiotics, antimicrobial resistance
Citation: Pattnaik M, Nayak AK, Karna S, Sahoo SK, Palo SK, Kanungo S, Kshatri JS, Parai D, Walia K, Singh T, Choudhary HR, Pati S and Bhattacharya D (2023) Perception and determinants leading to antimicrobial (mis)use: A knowledge, attitude, and practices study in the rural communities of Odisha, India. Front. Public Health 10:1074154. doi: 10.3389/fpubh.2022.1074154
Received: 19 October 2022; Accepted: 28 December 2022;
Published: 13 January 2023.
Edited by:
Sandul Yasobant, Indian Institute of Public Health Gandhinagar (IIPHG), IndiaReviewed by:
Germán Peñalva, Institute of Biomedicine of Seville (CSIC), SpainShahzad Ali, University of Veterinary and Animal Sciences, Pakistan
Copyright © 2023 Pattnaik, Nayak, Karna, Sahoo, Palo, Kanungo, Kshatri, Parai, Walia, Singh, Choudhary, Pati and Bhattacharya. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sanghamitra Pati,  ZHJzYW5naGFtaXRyYTEyQGdtYWlsLmNvbQ==; Debdutta Bhattacharya,
ZHJzYW5naGFtaXRyYTEyQGdtYWlsLmNvbQ==; Debdutta Bhattacharya,  ZHJkZWJkdXR0YS5iaGF0dGFjaGFyeWFAeWFob28uY28uaW4=
ZHJkZWJkdXR0YS5iaGF0dGFjaGFyeWFAeWFob28uY28uaW4=
†These authors have contributed equally to this work
 Matrujyoti Pattnaik1†
Matrujyoti Pattnaik1† Subrat Kumar Sahoo
Subrat Kumar Sahoo Subrata Kumar Palo
Subrata Kumar Palo Srikanta Kanungo
Srikanta Kanungo Jaya Singh Kshatri
Jaya Singh Kshatri Debaprasad Parai
Debaprasad Parai Kamini Walia
Kamini Walia Taru Singh
Taru Singh Hari Ram Choudhary
Hari Ram Choudhary Sanghamitra Pati
Sanghamitra Pati Debdutta Bhattacharya
Debdutta Bhattacharya