- 1Amref Health Africa, Nairobi, Kenya
- 2Amref Health Africa, Dar es Salaam, Tanzania
- 3Amref Health Africa, Kampala, Uganda
Introduction: Coronavirus Disease 2019 (COVID-19) presents a massive challenge in Africa due to overwhelmed and underresourced health systems, as well as the existing burden of communicable and non- communicable diseases. Self-inoculation may occur when an individual touches their mucous membrane following direct contact between their hands and severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2)-contaminated surfaces. Therefore, handwashing can be used along with COVID-19 vaccines to reduce the spread of SARS-CoV-2 and the burden of COVID-19. We were interested in investigating the levels of handwashing and vaccine uptake to control and prevent COVID-19 in Kenya, Uganda and Tanzania using a systematic review and meta-analysis.
Methods: We searched PubMed, African Index Medicus and African Journals Online for studies published since inception to 31st January 2023. We included all studies that assessed: the levels of COVID-19 vaccine acceptance and hesitance as indicators of vaccine uptake; and studies on the levels of handwashing to prevent and control COVID-19 in Kenya, Uganda and Tanzania. Study findings were synthesized by meta-analysis to get the pooled effect measure. Three studies were synthesized qualitatively due to high level of heterogeneity in effect measure precluding a quantitative meta-analysis.
Results: Our search identified 128 articles of which 17 studies with 18,305 participants and 622 vaccination sites were reviewed with 14 of them being meta-analyzed. This systematic review and meta-analysis reports high levels of COVID-19 vaccine acceptance and handwashing in Kenya, Uganda and Tanzania at 67% (95% CI: 55, 78) and 88% (95% CI: 73, 97), respectively. Vaccine hesitance among the participants was low at 31% (95% CI: 15, 49).
Discussion: Despite their importance in the control and prevention of COVID-19, some countries do not implement handwashing and vaccination effectively. There is a need for public health strategies to raise awareness about the importance of handwashing and the uptake of the COVID-19 vaccine.
Systematic Review Registration: https://www.crd.york.ac.uk/PROSPERO/, PROSPERO ID CRD42023394698.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causal agent of Coronavirus Disease 2019 (COVID-19), is transmitted via respiratory droplets (1). Self-inoculation may occur when one touches their mucous membranes of the nose, mouth, or eyes after direct contact with contaminated surfaces (2). Further, SARS-CoV-2 can be transmitted indirectly when contaminated hands spread the virus to other surfaces (2). As of 16th March 2023, the World Health Organization (WHO) had reported over 760 million verified cases of COVID-19 including more than six million deaths (3). Coronavirus Disease 2019 (COVID-19) presents a massive challenge in Africa due to overwhelmed and underresourced health systems, as well as the existing burden of communicable and non- communicable diseases (4).
Studies have reported that handwashing is beneficial in preventing a range of infections, including respiratory infections, gastrointestinal illnesses, and soil helminth infections (5–7). The WHO reported handwashing with soap and water as an effective and affordable way to prevent the transmission of SARS-CoV-2 (8). Similarly, studies show that handwashing reduces chances of self-contamination and subsequent nasal inoculation (8–10). According to a study, the practice of handwashing was found to reduce the transmission of respiratory viruses by approximately 45–55% (11). In addition, Saunders-Hastings et al. reported that hand hygiene decreased the spread of H1N1 influenza in humans by 38% (12). Evaluation of adult participants showed that handwashing was effectual in decreasing the spread of influenza virus (13). Despite the critical need for working handwashing stations at community and structural levels, it was estimated that the majority of people living in sub-Saharan Africa lacked access to handwashing stations before the COVID-19 pandemic (14).
Vaccination prevents SARS-CoV-2 infection and lowers the risk of severe health outcomes linked to COVID-19 (15, 16). Clinical trials along with observational studies have reported numerous COVID-19 vaccines to be harmless and efficacious in averting severe illness and risk of death (17–19). More than 13 billion vaccine doses have been administered globally to control and prevent COVID-19 (3). It is important to achieve high coverage of COVID-19 vaccination to reduce the adverse economic and health impacts associated with the pandemic. Furthermore, in the face of new variants, booster doses as well as vaccines with updated formulations may be required (20).
Implementing public health and social measures such as handwashing and vaccination against COVID-19 can slow down the spread of SARS-CoV-2 (21). However, few studies have investigated the link between handwashing and vaccine uptake to control and prevent COVID-19 in East Africa and their findings are inconclusive. Therefore, our objective was to conduct a systematic review and meta-analysis of levels of handwashing and COVID-19 vaccine uptake in Kenya, Uganda and Tanzania to prevent and control COVID-19. The outcomes of interest included vaccine hesitance and vaccine acceptance as indicators of levels of vaccine uptake, and levels of handwashing. Consequently, this research seeks to address the following fundamental research question: In residents of Kenya, Uganda, and Tanzania, what is the prevalence of handwashing practices, vaccine hesitancy, and vaccine acceptance in the context of COVID-19 prevention and control?
Materials and methods
Study design
We conducted this study according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and the Centre for Reviews and Dissemination (CRD) guidelines (22, 23). The systematic review and meta-analysis was registered under registration number CRD42023394698 on the International Prospective Register of Systematic Reviews (PROSPERO) database.
Eligibility criteria
Inclusion criteria: (1) studies on the levels of COVID-19 vaccine acceptance as an indicator of vaccine uptake in Kenya, Uganda and Tanzania; (2) studies on the levels of COVID-19 vaccine hesitance as an indicator of vaccine uptake in Kenya, Uganda and Tanzania; (3) studies on the levels of handwashing to prevent and control COVID-19 in Kenya, Uganda and Tanzania; (4) studies published in any language were considered for inclusion.
Exclusion criteria: (1) literature reviews, conference abstracts, and case series; (2) preprints; (3) articles with unclear measures of vaccine uptake; (4) studies conducted in countries other than Kenya, Uganda and Tanzania; (4) studies on COVID-19 vaccine side effects; (5) studies without the relevant exposure or treatment were excluded.
Literature search
We conducted a systematic literature search in PubMed, African Index Medicus and African Journals Online to select plausibly eligible articles published since database inception to 31st January 2023. Moreover, we manually screened citations of eligible articles to identify additional studies. We formulated a search strategy based on the PECOS framework by combining the terms handwashing, vaccine, COVID-19, prevention, policy, Kenya, Uganda, and Tanzania. The search approach used in the PubMed database was modified to suit other databases. The detailed approach of literature search is available in Supplementary Tables 1, 2.
Study selection
We used the Mendeley reference manager to manage the articles identified during the search. Identical studies were initially excluded using Mendeley after which irrelevant articles were removed by screening the titles and abstracts for relevance. We then reviewed full texts of the potentially relevant studies to determine eligibility for inclusion. The eligible studies were selected by two independent reviewers and discordant outcomes were resolved through discussion.
Data extraction
Two independent researchers extracted data from the eligible articles using a predefined and standardized excel sheet. Variables that were extracted from these studies included: (1) the name of the first author(s); (2) the title of the study; (3) the year of publication; (4) study objective(s); (5) the publishing journal; (6) the study design; (7) the sample size; (8) participants’ characteristics including age; (9) the inclusion and exclusion criteria; (10) indicators of hand washing levels; (11) indicators of vaccine uptake levels; (12) main finding and other findings. Missing data were obtained by contacting authors of the eligible studies.
Quality assessment
We assessed the potential for bias in the eligible articles based on the Quality Assessment Tool for Observational Cohort and Cross-sectional studies (24).1 This checklist encompasses 14 crucial criteria regarded as fundamental for ensuring the quality of reporting in cohort and cross-sectional studies. These recommendations focus on various aspects, including the article’s objectives, the study population, exposure measures and potential confounders, among others.
Statistical analysis
We conducted meta-analyses of single proportions to calculate the overall proportion using the metaprop function of the Meta package in R (version 4.1.2). Heterogeneity between studies was assessed using I2 statistics, with an I2 of more than 75% indicating substantial heterogeneity. We used a funnel plot to check for publication bias and the Eggers test for assessing funnel plot asymmetry. We implemented random effect meta-analysis because of the high levels of heterogeneity between study populations. Heterogeneous studies were synthesized through a narrative summary based on the specific outcome indicator.
Results
Study selection
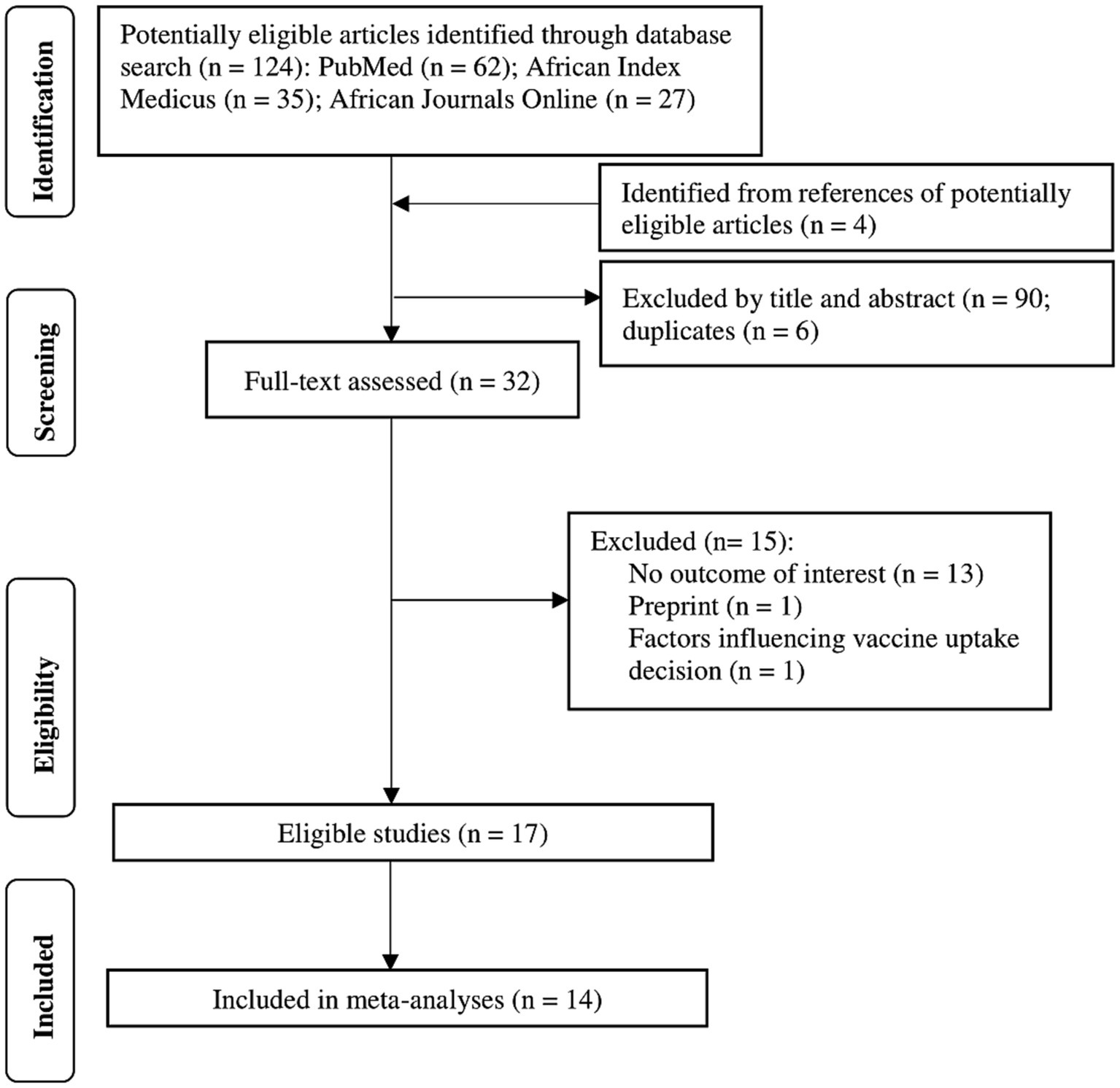
Our literature search yielded 124 articles and an additional four papers from potentially eligible articles (Figure 1). We excluded 6 duplicates and 90 studies that were irrelevant after screening titles and abstracts. We excluded 15 papers after a full-text review of the remaining articles, including one preprint and 14 others without the outcome of interest. A total of 17 studies with 18,305 participants and 622 vaccination sites were included in the systematic review. Only 14 of the 17 studies were meta-analyzed. Some studies could not be meta-analyzed because they reported heterogeneous effect estimates, including vaccination rates and odds ratios of getting the COVID-19 vaccine.
Basic characteristics of eligible studies
We included observational studies published between 2021 and 2023 in this systematic review and meta-analysis. The eligible studies included 15 cross-sectional studies, one mixed-method study and one longitudinal study. Seven of these studies were conducted in Kenya, with Uganda and Tanzania having eight and two studies, respectively. The sample sizes varied across studies ranging from 33 to 4,136 participants. Only three of four studies that reported handwashing levels in Kenya, Uganda and Tanzania had handwashing as the primary outcome.
Quality evaluation
According to the Quality Assessment Tool for Observational Cohort and Cross-sectional studies checklist, all the studies met the recommendations for conducting observational studies (Supplementary Table 3). This finding indicates high overall methodological quality and low risk of bias of these studies.
Meta-analysis
Levels of handwashing in Kenya, Uganda and Tanzania
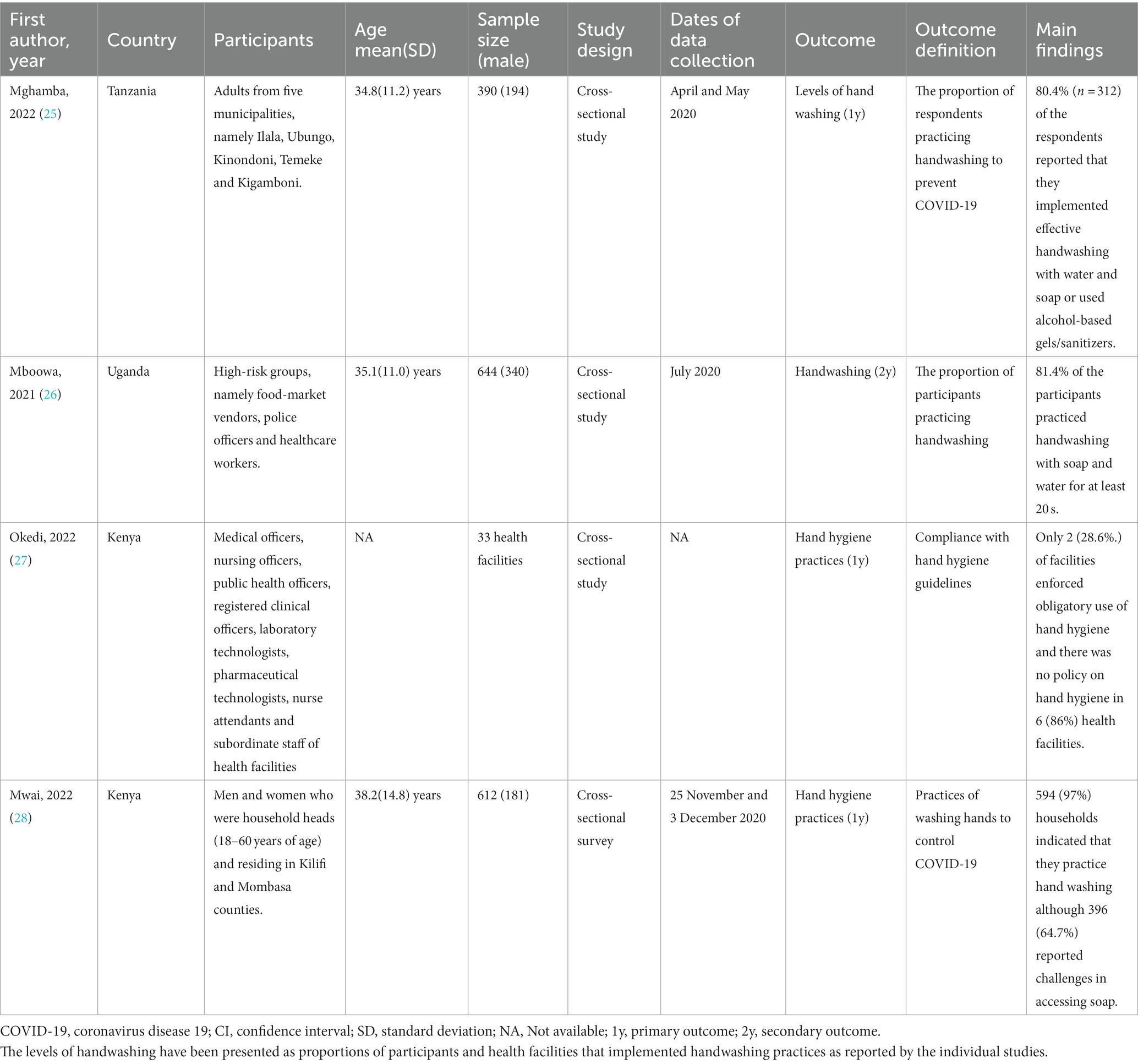
The participants of the studies that reported handwashing levels as a way of preventing and controlling COVID-19 were adults with a mean (SD) ages ranging from 34.8(11.2) years to 38.2(14.8) years. These studies reported that approximately 80.4 to 97% of the respondents practiced handwashing and 28.6% of facilities enforced obligatory use of hand hygiene (Table 1). Further meta-analysis of three of those studies (N = 1,646) showed that the pooled estimate proportion was 0.88 (95% CI: 0.73, 0.97) (Figure 2).
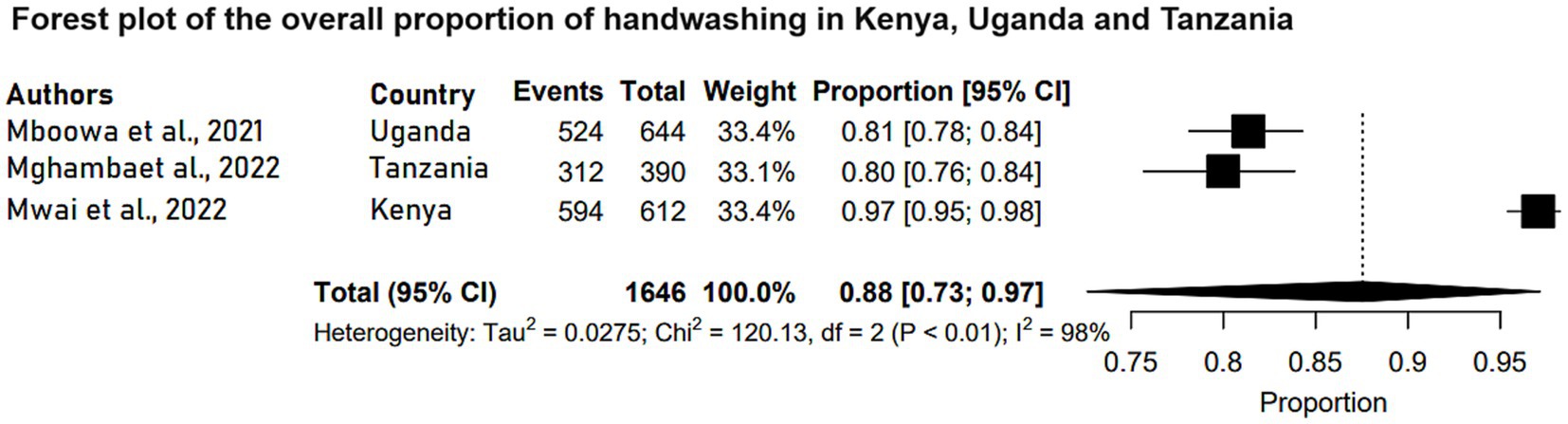
Figure 2. Forest plot of the overall proportion of handwashing in Kenya, Uganda and Tanzania. Each study is represented by a black box and a horizontal line, which correspond to the proportion and 95% confidence interval, respectively. I2 shows the degree of heterogeneity with value of p indicating whether there was statistically significance heterogeneity between the studies and among the groups.
Levels of vaccine acceptance in Kenya and Uganda
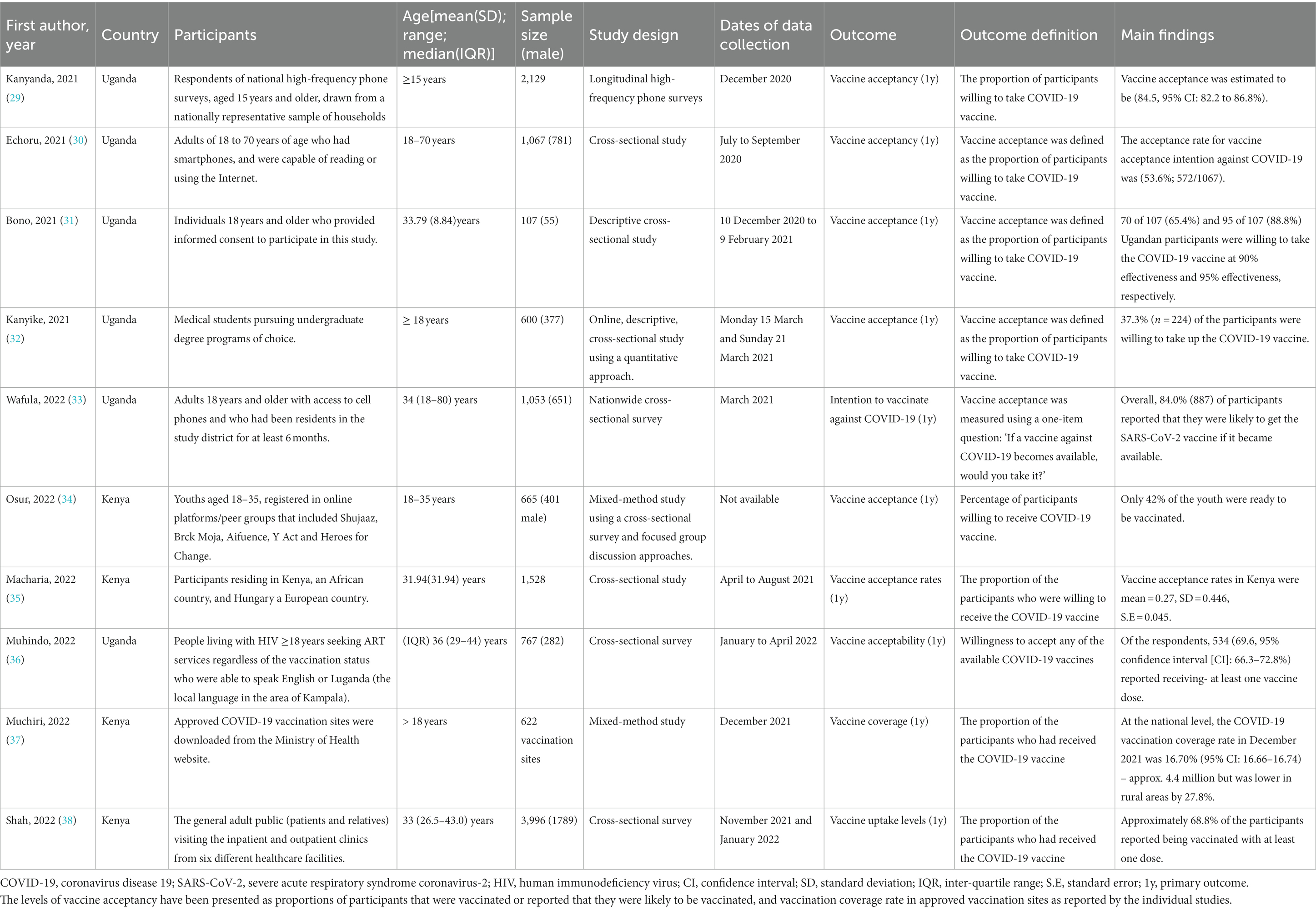
On the other hand, 10 studies reported on levels of vaccine acceptance in Kenya and Uganda with Tanzania lacking a study with this outcome. Six of the 10 studies were conducted among Ugandan respondents. All the studies were conducted among participants 15 years old and above. The vaccine acceptance rate ranged between 37.3 and 84.5% across studies. Table 2 shows the characteristics of the studies that focused on vaccine acceptance to prevent and control COVID-19 in Kenya and Uganda. Meta-analysis of eight of those studies (N = 10,384) reported that the pooled estimate proportion for vaccine acceptance in Kenya and Uganda was 0.67 (95% CI: 0.55, 0.78) (Figure 3).
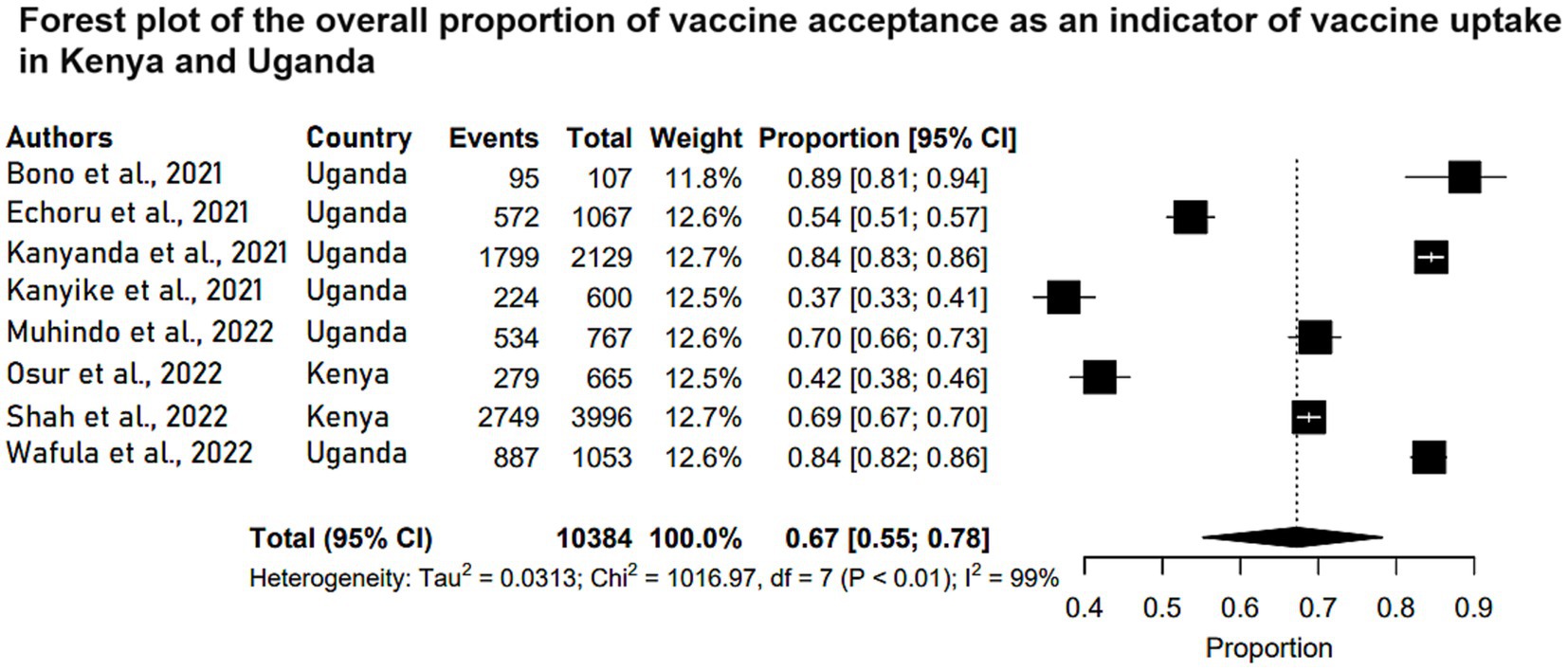
Figure 3. Forest plot of the overall proportion of vaccine acceptance as an indicator of vaccine uptake in Kenya and Uganda. Studies are plotted alphabetically. Each study is represented by a black box and a horizontal line, which correspond to the proportion and 95% confidence interval, respectively. I2 shows the degree of heterogeneity with value of p indicating whether there was statistically significance heterogeneity between the studies and among the groups.
Levels of vaccine hesitance in Kenya, Uganda and Tanzania
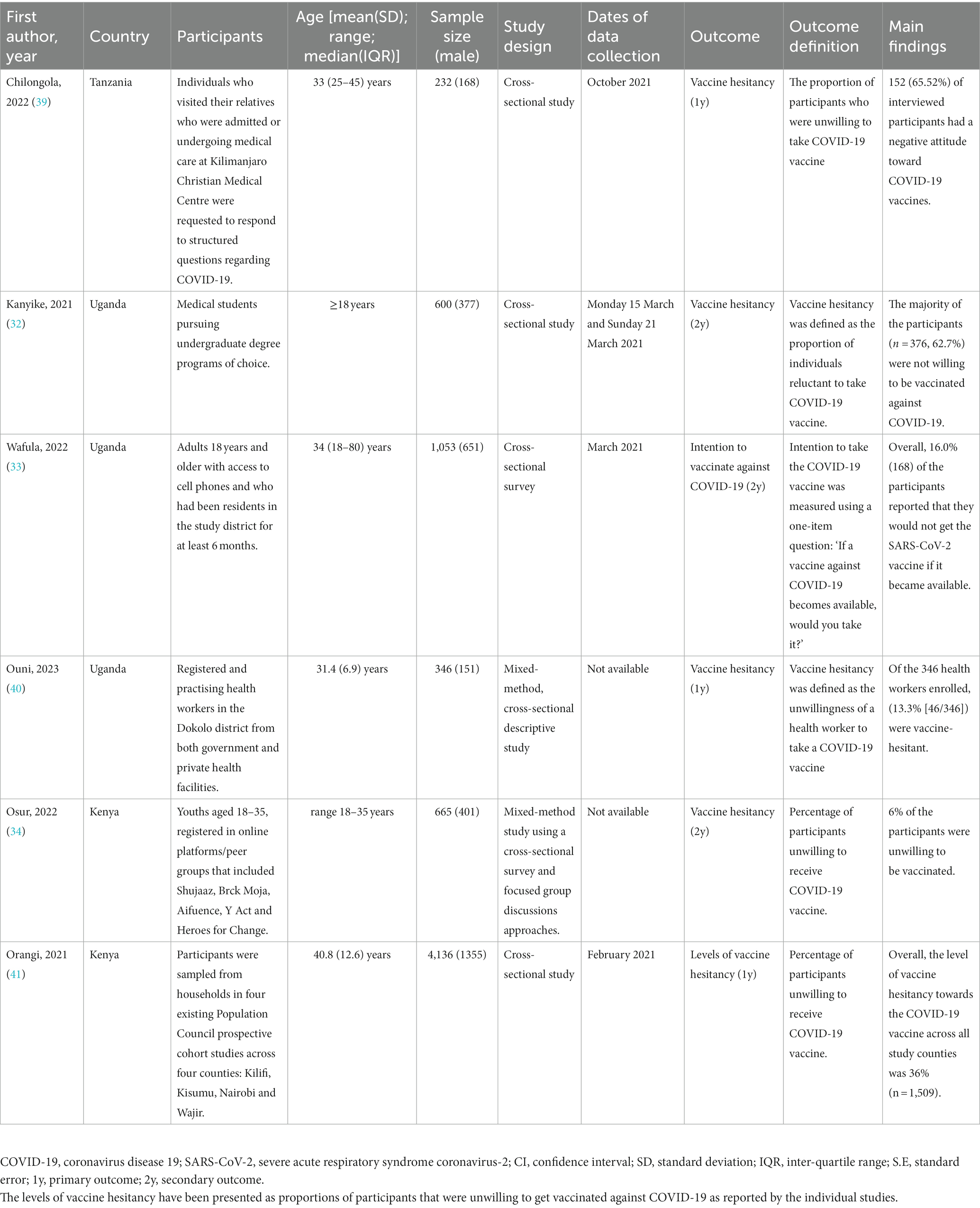
Six observational studies reported the levels of COVID-19 vaccine hesitance among the study participants in Kenya, Uganda and Tanzania. These studies were conducted among adult respondents above 18 years of age. Three of the studies reported vaccine hesitance as a secondary outcome indicating vaccine uptake levels in the three countries. The COVID-19 vaccine hesitance levels across studies were between 6 and 65.5% among the study participants. The characteristics of studies of vaccine hesitance in Kenya, Uganda and Tanzania are depicted in Table 3. A meta-analysis of the six studies (N = 7,032) reported that the pooled estimate proportion was 0.31 (95% CI: 0.15, 0.49) in Kenya, Uganda and Tanzania (Figure 4).
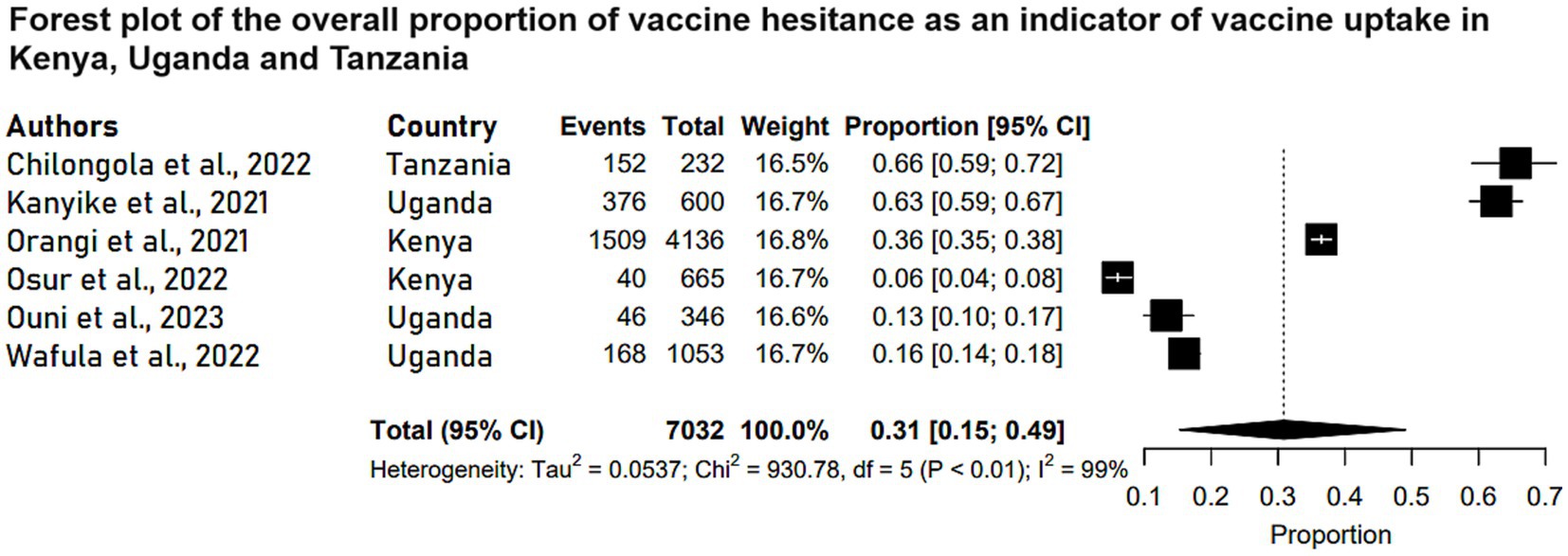
Figure 4. Forest plot of the overall proportion of vaccine hesitance as an indicator of vaccine uptake in Kenya, Uganda and Tanzania. Each study is represented by a black box and a horizontal line, which correspond to the proportion and 95% confidence interval, respectively. I2 shows the degree of heterogeneity with value of p indicating whether there was statistically significance heterogeneity between the studies and among the groups.
Publication bias
A funnel plot was evaluated for all three outcomes and the results are shown on Supplementary Figures 1–3. The two sides of each funnel plot were symmetrical, and no significant publication bias was found in this study.
Heterogeneity
The meta-analyzed studies revealed substantial heterogeneity, with I2 statistics indicating 98% for handwashing levels, 99% for vaccine acceptance levels, and 99% for vaccine hesitance levels, as depicted in Figures 1–3, respectively.
Qualitative analysis
Levels of handwashing in Kenya, Uganda and Tanzania
The participants of the studies that reported handwashing levels as a way of preventing and controlling COVID-19 were adults with a mean (sd) ages ranging from 34.8(11.2) years to 38.2(14.8) years. These studies reported that approximately 80.4 to 97% of the respondents practiced handwashing and 28.6% of facilities enforced obligatory use of hand hygiene (Table 1).
Levels of vaccine acceptance in Kenya and Uganda
On the other hand, 10 studies reported on levels of vaccine acceptance in Kenya and Uganda with Tanzania lacking a study with this outcome. Six of the 10 studies were conducted among Ugandan respondents. All the studies were conducted among participants 15 years old and above. The vaccine acceptance rate ranged between 37.3 and 84.5% across studies. Table 2 shows the characteristics of the studies that focused on vaccine acceptance to prevent and control COVID-19 in Kenya and Uganda.
Levels of vaccine hesitancy in Kenya, Uganda and Tanzania
Six observational studies reported the levels of COVID-19 vaccine hesitance among the study participants in Kenya, Uganda and Tanzania. These studies were conducted among adult respondents above 18 years of age. Three of the studies reported vaccine hesitancy as a secondary outcome indicating vaccine uptake levels in the three countries. The COVID-19 vaccine hesitancy levels across studies were between 6 and 65.5%mong the study participants. The characteristics of studies of vaccine hesitance in Kenya, Uganda and Tanzania are depicted in Table 3.
Discussion
To date, there are scarce data on the levels of handwashing and COVID-19 vaccine uptake in East Africa. Information regarding these interventions to contain the spread of SARS-CoV-2 is helpful in developing public health strategies of managing COVID-19. This study involved conducting a systematic review and meta-analysis of handwashing and vaccine uptake levels among Kenyan, Ugandan, and Tanzanian study participants.
In the current study, a total of 17 articles were systematically reviewed after which 14 were included in the final analysis conducted according to the outcome measure. Overall, the included studies had high methodological quality and the pooled handwashing levels in Kenya, Uganda and Tanzania were 88% (95% CI: 73, 97). The level of vaccine uptake in the three countries was fairly high as indicated by vaccine acceptance and vaccine hesitance at 67% (95% CI: 55, 78) and 31% (95% CI: 15, 49), respectively.
Handwashing was associated with reduced levels of COVID-19 suggesting that this strategy could be effective in the control and prevention of COVID-19. These findings are concordant with those reported by Beale et al. showing that handwashing (6–10 times per day) predicted a decreased susceptibility to coronavirus infection (13).
Despite clinical trials reporting most COVID-19 vaccines to be harmless and efficacious (42), the findings of this study suggest that there are considerable levels of vaccine hesitance in Kenya, Uganda and Tanzania. These findings are concordant with those of Afolabi and Ilesanmi, and Mutombo et al. (43, 44) which reported considerable vaccine hesitancy in Africa in addition to low COVID-19 vaccine coverage in the continent.
In our meta-analyses, we acknowledge the presence of substantial heterogeneity, with I2 statistics ranging from 98 to 99%. This observation warrants careful consideration, as it underscores the need for a nuanced interpretation of our findings. The sources of this heterogeneity are multifaceted. One contributing factor could be the inherent regional differences within the study populations, spanning Kenya, Uganda, and Tanzania. Additionally, variability in outcome definitions employed across the studies may have added to this heterogeneity. Therefore, the results of this systematic review and meta-analysis should be interpreted with caution.
To the best of our knowledge, this is the first systematic review and meta-analysis exploring levels of handwashing and vaccine uptake in the prevention and control of COVID-19 in Kenya, Uganda and Tanzania. Moreover, our study is comprehensive as it includes a large number of relevant articles published to date. Our systematic review and meta-analysis has a few limitations: we may have missed some studies since some African journals are not indexed in PubMed. Lastly, only observational studies, which tend to provide weaker evidence compared to randomized clinical trials, were included in the meta-analysis section and some studies could not be meta-analyzed.
Conclusion
Evidence of the levels of handwashing and vaccine uptake in Kenya, Uganda and Tanzania is limited and inconsistent. Leaders should champion awareness and COVID-19 vaccine uptake and improve handwashing facilities. Our findings warrant further investigation to determine the levels of handwashing and vaccine uptake to control and prevent COVID-19.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author.
Author contributions
JoM: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. JaM: Data curation, Methodology, Writing – review & editing. HM: Data curation, Methodology, Writing – review & editing. KK: Data curation, Methodology, Writing – review & editing. SK: Data curation, Methodology, Writing – review & editing. LK: Data curation, Methodology, Writing – review & editing. RM: Data curation, Methodology, Writing – review & editing. MN: Data curation, Methodology, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study was funded by Foreign, Commonwealth & Development Office (FCDO). The funding body did not have any role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpubh.2023.1256007/full#supplementary-material
Abbreviations
COVID-19, Coronavirus disease-19; SARS-CoV-2, Severe acute respiratory syndrome coronavirus-2; WHO, World Health Organization; CI, Confidence interval; SD, standard deviation; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; CRD, Centre for Reviews and Dissemination guidelines; PROSPERO, International Prospective Register of Systematic Reviews; PECO, population, exposure, comparator, and outcome
Footnotes
References
1. Lai, C-C , Shih, T-P , Ko, W-C , Tang, H-J , and Hsueh, P-R . Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents. (2020) 55:105924. doi: 10.1016/j.ijantimicag.2020.105924
2. Ramesh, N , Siddaiah, A , and Joseph, B . Tackling corona virus disease 2019 (COVID-19) in workplaces. Indian J Occup Environ Med. (2020) 24:16. doi: 10.4103/ijoem.IJOEM_49_20
3. World Health Organization . WHO coronavirus (COVID-19) dashboard. Geneva: World Health Organization (2023).
4. Ozili, P . COVID-19 in Africa: socio-economic impact, policy response and opportunities. Int J Sociol Soc Policy. (2020). doi: 10.2139/ssrn.3574767
5. Sangalang, SO , Prado, NO , Lemence, ALG , Cayetano, MG , Lu, JLDP , Valencia, JC, et al. Diarrhoea, malnutrition, and dehydration associated with school water, sanitation, and hygiene in Metro Manila, Philippines: A cross-sectional study. Sci Total Environ. (2022) 838:155882. doi: 10.1016/j.scitotenv.2022.155882
6. Xun, Y , Shi, Q , Yang, N , Li, Y , Si, W , Shi, Q, et al. Associations of hand washing frequency with the incidence of illness: a systematic review and meta-analysis. Ann Transl Med. (2021) 9:395. doi: 10.21037/atm-20-6005
7. Hadaway, A . Handwashing: clean hands save lives. J Consum Health Internet. (2020) 24:43–9. doi: 10.1080/15398285.2019.1710981
8. Chaudhary, NK , Chaudhary, N , Dahal, M , Guragain, B , Rai, S , Chaudhary, R, et al. Fighting the SARS CoV-2 (COVID-19) pandemic with soap. (2020). doi: 10.20944/preprints202005.0060.v1
9. Valsamatzi-Panagiotou, A , and Penchovsky, R . Environmental factors influencing the transmission of the coronavirus 2019: a review. Environ Chem Lett. (2022) 20:1603–10. doi: 10.1007/s10311-022-01418-9
10. Khatib, MN , Sinha, A , Mishra, G , Quazi, SZ , Gaidhane, S , Saxena, D, et al. WASH to control COVID-19: a rapid review. Front public Heal. (2022) 10:976423. doi: 10.3389/fpubh.2022.976423
11. Jefferson, T , Foxlee, R , Del Mar, C , Dooley, L , Ferroni, E , Hewak, B, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses: systematic review. BMJ. (2008) 336:77–80. doi: 10.1136/bmj.39393.510347.BE
12. Saunders-Hastings, P , Crispo, JAG , Sikora, L , and Krewski, D . Effectiveness of personal protective measures in reducing pandemic influenza transmission: a systematic review and meta-analysis. Epidemics. (2017) 20:1–20. doi: 10.1016/j.epidem.2017.04.003
13. Smith, SMS , Sonego, S , Wallen, GR , Waterer, G , Cheng, AC , and Thompson, P . Use of non-pharmaceutical interventions to reduce the transmission of influenza in adults: a systematic review. Respirology. (2015) 20:896–903. doi: 10.1111/resp.12541
14. Brauer, M , Zhao, JT , Bennitt, FB , and Stanaway, JD . Global access to handwashing: implications for COVID-19 control in low-income countries. Environ Health Perspect. (2020) 128:57005. doi: 10.1289/EHP7200
15. Gupta, S , Cantor, J , Simon, KI , Bento, AI , Wing, C , and Whaley, CM . Vaccinations against COVID-19 may have averted up to 140,000 deaths in the United States: study examines role of COVID-19 vaccines and deaths averted in the United States. Health Aff. (2021) 40:1465–72. doi: 10.1377/hlthaff.2021.00619
16. Andrews, N , Tessier, E , Stowe, J , Gower, C , Kirsebom, F , Simmons, R, et al. Duration of protection against mild and severe disease by COVID-19 vaccines. N Engl J Med. (2022) 386:340–50. doi: 10.1056/NEJMoa2115481
17. Saadh, MJ , and Jaber, SA . Efficacy of COVID-19 vaccines. Microb Pathog. (2022) 171:105729. doi: 10.1016/j.micpath.2022.105729
18. Lipsitch, M , and Dean, NE . Understanding COVID-19 vaccine efficacy. Science. (2020) 370:763–5. doi: 10.1126/science.abe5938
19. Fiolet, T , Kherabi, Y , MacDonald, C-J , Ghosn, J , and Peiffer-Smadja, N . Comparing COVID-19 vaccines for their characteristics, efficacy and effectiveness against SARS-CoV-2 and variants of concern: a narrative review. Clin Microbiol Infect. (2022) 28:202–21. doi: 10.1016/j.cmi.2021.10.005
20. Xiao, C , Su, J , Zhang, C , Huang, B , Mao, L , Ren, Z, et al. Effectiveness of booster doses of the SARS-CoV-2 inactivated vaccine KCONVAC against the mutant strains. Viruses. (2022) 14. doi: 10.3390/v14092016
21. Talic, S , Shah, S , Wild, H , Gasevic, D , Maharaj, A , Ademi, Z, et al. Effectiveness of public health measures in reducing the incidence of covid-19, SARS-CoV-2 transmission, and COVID-19 mortality: systematic review and meta-analysis. BMJ. (2021):375. doi: 10.1136/bmj-2021-068302
22. Moher, D , Liberati, A , Tetzlaff, J , and Altman, DG, PRISMA Group . Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. (2009) 6:e1000097. doi: 10.1371/journal.pmed.1000097
23. Akers, J , and York, U . Systematic reviews: CRD’s guidance for undertaking reviews in health care. York: CRD, University of York (2009).
24. Heart, N, Blood Institute L. Quality assessment tool for observational cohort and cross-sectional studies [internet]. Natl Institutes Heal. (2021)
25. Mghamba, JM , Oriyo, NM , Bita, AAF , Shayo, E , Kagaruki, G , Katsande, R, et al. Compliance to infection prevention and control interventions for slowing down COVID-19 in early phase of disease transmission in Dar es Salaam. Tanzania Pan Afr Med J. (2022) 41:174. doi: 10.11604/pamj.2022.41.174.31481
26. Mboowa, G , Musoke, D , Bulafu, D , and Aruhomukama, D . Face-masking, an acceptable protective measure against COVID-19 in ugandan high-risk groups. Am J Trop Med Hyg. (2021) 104:502–13. doi: 10.4269/ajtmh.20-1174
27. Okedi, W , Wakoli, C , and Emojong, I . The impact and sustainability of hand-hygiene practices on health facilities in the context of the COVID-19 pandemic. A case study of Busia County (Kenya). Int J Med Public Heal. (2022) 12:116–30. doi: 10.5530/ijmedph.2022.3.23
28. Mwai, J , Nyole, D , Abdi, M , Ahmed, I , Mutai, J , Kaduka, L, et al. Assessment of water, sanitation and hygiene practices for prevention and control of COVID-19 in Kenya. Int Health. (2022) 14:597–603. doi: 10.1093/inthealth/ihab077
29. Kanyanda, S , Markhof, Y , Wollburg, P , and Zezza, A . Acceptance of COVID-19 vaccines in sub-Saharan Africa: evidence from six national phone surveys. BMJ Open. (2021) 11:e055159–8. doi: 10.1136/bmjopen-2021-055159
30. Echoru, I , Ajambo, PD , Keirania, E , and Bukenya, EEM . Sociodemographic factors associated with acceptance of COVID-19 vaccine and clinical trials in Uganda: a cross-sectional study in western Uganda. BMC Public Health. (2021) 21:1–9. doi: 10.1186/s12889-021-11197-7
31. Bono, SA , Villela Ef De, M , Siau, CS , Chen, WS , Pengpid, S , Hasan, MT, et al. Factors affecting COVID-19 vaccine acceptance: an international survey among low-and middle-income countries. Vaccine. (2021) 9:1–19. doi: 10.3390/vaccines9050515
32. Kanyike, AM , Olum, R , Kajjimu, J , Ojilong, D , Akech, GM , Nassozi, DR, et al. Acceptance of the coronavirus disease-2019 vaccine among medical students in Uganda. Trop Med Health. (2021) 49:1–11. doi: 10.1186/s41182-021-00331-1
33. Wafula, ST , Mugume, IB , Sensasi, B , Okware, S , Chimbaru, A , Nanyunja, M, et al. Intention to vaccinate against COVID-19 and adherence to non-pharmaceutical interventions against COVID-19 prior to the second wave of the pandemic in Uganda: a cross-sectional study. BMJ Open. (2022) 12:e057322. doi: 10.1136/bmjopen-2021-057322
34. Osur, JO , Chengo, R , Muinga, E , Kemboi, J , Sidibe, M , and Rarieya, M . Determinants of COVID-19 vaccine behaviour intentions among the youth in Kenya: a cross-sectional study. Arch Public Health. (2022) 80:159. doi: 10.1186/s13690-022-00904-4
35. Macharia, JM , Gakenye, GW , Rozmann, N , Onchonga, D , Mwangi, RW , Kaposztas, Z, et al. An empirical assessment of the factors influencing acceptance of COVID-19 vaccine uptake between Kenyan and Hungarian residing populations: a cross-sectional study. Sci Rep. (2022) 12:22262. doi: 10.1038/s41598-022-26824-5
36. Muhindo, R , Okoboi, S , Kiragga, A , King, R , Arinaitwe, WJ , and Castelnuovo, B . COVID-19 vaccine acceptability, and uptake among people living with HIV in Uganda. PLoS One. (2022) 17:e0278692. doi: 10.1371/journal.pone.0278692
37. Muchiri, SK , Muthee, R , Kiarie, H , Sitienei, J , Agweyu, A , Atkinson, PM, et al. Unmet need for COVID-19 vaccination coverage in Kenya. Vaccine. (2022) 40:2011–9. doi: 10.1016/j.vaccine.2022.02.035
38. Shah, J , Abeid, A , Sharma, K , Manji, S , Nambafu, J , Korom, R, et al. Perceptions and knowledge towards COVID-19 vaccine hesitancy among a subpopulation of adults in Kenya: an English survey at six healthcare facilities. Vaccine. (2022) 10:1–15. doi: 10.3390/vaccines10050705
39. Chilongola, JO , Rwegoshola, KM , Balingumu, OH , Semvua, HS , and Kwigizile, ET . COVID-19 knowledge, attitudes, practices, and vaccination hesitancy in Moshi, Kilimanjaro Region, Northern Tanzania. J Health Res. (2022) 23:1–12. doi: 10.4314/thrb.v23i1
40. Ouni, PD , Namulondo, R , Wanume, B , Okia, D , Olupot, PO , Nantale, R, et al. COVID-19 vaccine hesitancy among health workers in rural Uganda: a mixed methods study. Vaccine X. (2023) 13:100260. doi: 10.1016/j.jvacx.2023.100260
41. Orangi, S , Pinchoff, J , Mwanga, D , Abuya, T , Hamaluba, M , Warimwe, G, et al. Assessing the level and determinants of COVID-19 vaccine confidence in Kenya. Vaccine. (2021) 9. doi: 10.3390/vaccines9080936
42. Liu, Q , Qin, C , Liu, M , and Liu, J . Effectiveness and safety of SARS-CoV-2 vaccine in real-world studies: a systematic review and meta-analysis. Infect Dis Poverty. (2021) 10:1–15. doi: 10.1186/s40249-021-00915-3
43. Afolabi, AA , and Ilesanmi, OS . Dealing with vaccine hesitancy in Africa: the prospective COVID-19 vaccine context. Pan Afr Med J. (2021) 38:3.
Keywords: handwashing, vaccine, COVID-19, prevention, control, policy, Kenya, Uganda
Citation: Muchangi JM, Mturi J, Mukasa H, Kithuki K, Kosgei SJ, Kanyangi LM, Mararo R and Nankanja M (2023) Levels of handwashing and vaccine uptake in Kenya, Uganda, and Tanzania to prevent and control COVID-19: a systematic review and meta-analysis. Front. Public Health. 11:1256007. doi: 10.3389/fpubh.2023.1256007
Edited by:
Nicola Luigi Bragazzi, University of Parma, ItalyReviewed by:
Fnu Kajal, University of Arizona, United StatesChiranjivi Adhikari, Pokhara University, Nepal
Copyright © 2023 Muchangi, Mturi, Mukasa, Kithuki, Kosgei, Kanyangi, Mararo and Nankanja. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Josphat Martin Muchangi, TWFydGluLk11Y2hhbmdpQGFtcmVmLm9yZw==
 Josphat Martin Muchangi
Josphat Martin Muchangi James Mturi2
James Mturi2 Hajra Mukasa
Hajra Mukasa