- 1Center for Infectious Disease Research and Policy, University of Minnesota, Minneapolis, MN, United States
- 2Department of Anesthesiology, University of Washington, Seattle, WA, United States
- 3Department of Medicine, McGill University, Montreal, QC, Canada
- 4Departments Medicine and Population Medicine, Harvard Medical School, Boston, MA, United States
- 5Department of Medicine, University of California San Diego, San Diego, CA, United States
- 6Department of Medicine, University of California San Francisco, San Francisco, CA, United States
Introduction
In order to protect patients and healthcare workers from aerosol transmissible diseases, healthcare facilities should improve ventilation and air purification and in addition should consider universal use of respirators (e.g., N95, FFP2 or equivalent) when aerosol-transmissible pathogens are widespread in the community. A study of SARS-CoV-2 within 288 United States hospitals documented more than 14,000 infections potentially acquired in the hospital over a 2-year period and found that more than 8% of patients hospitalized with SARS-CoV-2 may have acquired their infection in the hospital (1). Despite the frequency of nosocomial respiratory viral transmission most countries have no national mandate for masks or respirators in healthcare facilities. We propose that healthcare facilities should anticipate that aerosol transmissible disease will continue to be of major importance to public health for the foreseeable future.
Aerosol transmissible disease
Early in the COVID pandemic, some argued that SARS CoV-2 was transmitted primarily by larger respiratory particles known as “droplets,” produced during coughing and sneezing and propelled into the mouth or nose of someone nearby (2). Surgical masks were thought by many to provide adequate protection; respirators were only recommended for healthcare workers performing a limited and variable set of procedures.
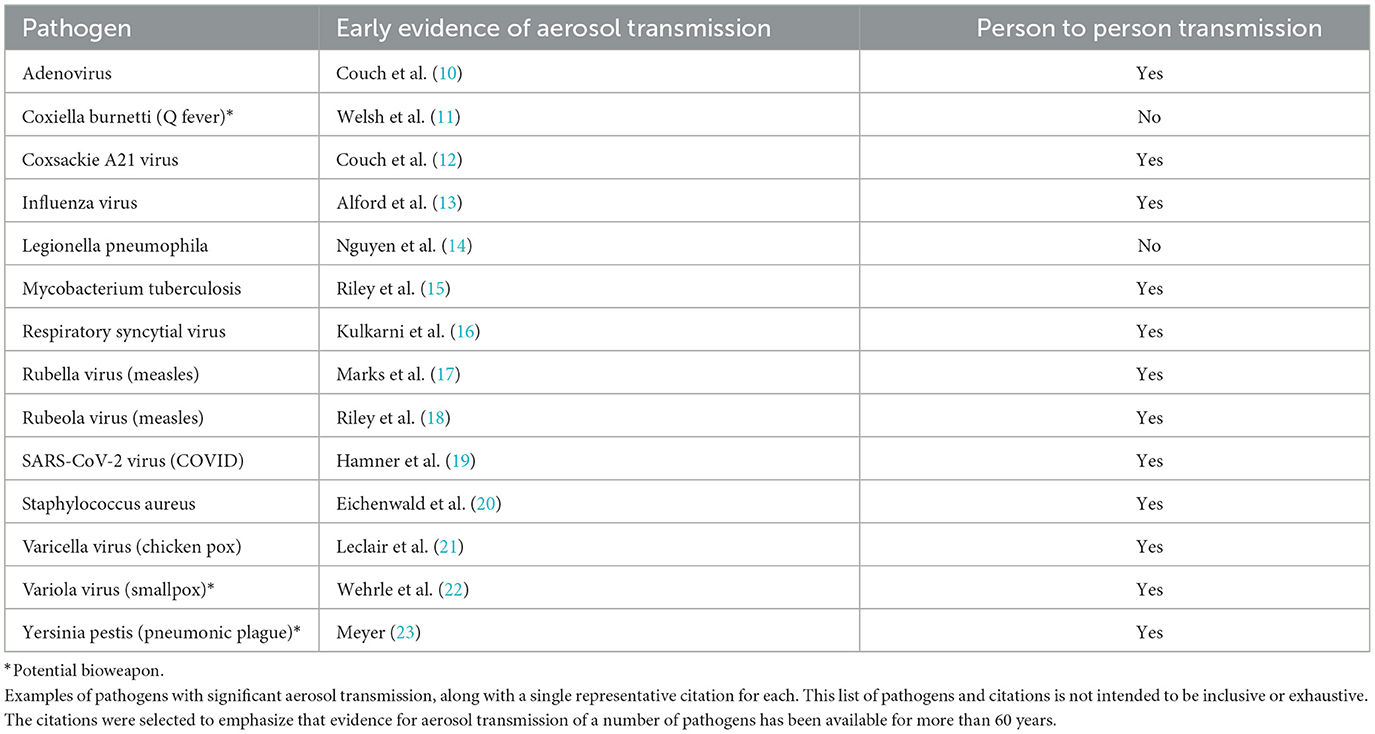
Examination of a large body of evidence has shown that transmission of SARS-CoV-2 is primarily through inhalation of smaller respiratory particles generated by breathing, talking, singing and other ordinary respiratory activities (3–7). These smaller particles are predominantly < 5 microns in size, can remain suspended in air for many minutes or even hours, diffuse or move by air currents throughout an indoor space, and are easily inhaled both near and far from a source (8). Optimal protection from inhaling these small particles requires respirators, not surgical masks (4, 9). While recent attention has been focused on SARS-CoV-2, the importance of aerosol transmission for a wide variety of pathogens has been recognized for many years; examples of aerosol transmissible viruses and bacteria are shown in the Table 1, along with examples of pertinent literature.
Variable performance of face coverings
Recognition of the importance of aerosol transmission has critical implications for healthcare workers and patients. However, the conversation about appropriate protection has been obscured by the widespread use of the terms “mask” and “masking” to encompass anything worn on the face. These terms lack precision and suggest that everything worn on the face provides similar levels of source control and personal protection. In fact, there are large differences in the performance characteristics and effectiveness of different face coverings.
The most common face coverings in health care facilities are surgical masks (a term which encompasses procedure masks). Most surgical masks are not designed to fit tightly against the face and thus have a limited impact on inward and outward movement of smaller particles because air can move freely around the edges of the mask instead of through the filtering material. In addition, surgical masks are typically constructed of filtering material that is not as effective as the material used for respirators. Surgical masks have been worn during surgery with the intention of preventing bacterial infection of surgical wounds from droplets generated by surgical personnel, although efficacy for this purpose is questionable (24). Since the HIV epidemic, surgical masks have also been deployed as protection against splashes with blood or other body fluids. Efficacy of surgical masks for protection against aerosol transmission is limited (25–27).
Respirators (which should not be referred to as “masks”) are designed to fit tightly to the face, are constructed of highly effective filtering material and can provide substantial protection against aerosol transmission. They typically undergo a rigorous testing and approval process supervised by governmental agencies. The most common disposable “filtering facepiece” respirators are designated as N95 in the United States and Canada and FFP2 in the United Kingdom, European Union, Australia and New Zealand.
There are some in healthcare who question the relative effectiveness of respirators in comparison to surgical masks. Laboratory and workplace measurements have clearly and consistently demonstrated the superior performance of respirators for all types of hazardous aerosols (26–28). However, some have suggested that evidence from randomized clinical trials is needed. This is problematic because most trials have only assigned healthcare workers to wear respirators when caring for patients with known or suspected respiratory viral infections, ignoring the fact that workers are continuously exposed to viruses in other contexts, at home, in the community and from exposure to co-workers and pre-symptomatic or asymptomatic patients. Nevertheless, the limited clinical evidence we have suggests that respirators reduce the risk of infection to a greater degree than surgical masks (29–31), consistent with the strong evidence from laboratory and workplace measurements of respirator performance.
Discussion
The HIV epidemic transformed healthcare worker behavior, making contact between the healthcare worker and patient body fluids something to be strictly avoided. Similarly, the COVID-19 pandemic has heightened awareness about the importance of preventing transmission of aerosolized pathogens (32). Prior to COVID-19, precautions against aerosol-transmissible pathogens were considered important for only a few specific pathogens, such as tuberculosis and measles and most healthcare workers seldom if ever donned a respirator. In fact, evidence for aerosol transmission of influenza has been accumulating since the 1960s (33) along with evidence for aerosol transmission of a variety of viruses (10, 34), bacteria (35) and fungi (36). Coronaviruses and influenza viruses are especially noteworthy because of their proven pandemic potential (37), but their mode of transmission is not unique. Some pathogens, not classically thought to spread by aerosols, can become airborne pathogens in some circumstances, for example Yersinia pestis in primary plague pneumonia (23, 38). While not all aerosol transmissible diseases result in a significant incidence of hospitalization and death, many are serious threats to public health.
The importance of aerosol transmission has fundamental importance for health policy, because traditional droplet precautions, such as staying six feet away from a source or wearing a surgical mask, will not provide adequate protection from aerosols. Prevention of aerosol transmission requires attention to indoor air quality through adequate ventilation and air purification and the use of respirators rather than surgical masks for personal protection and source control.
Since universal masking with either respirators or surgical masks has been largely abandoned by healthcare facilities, it is critical to understand the appropriate triggers for reinstating universal respiratory protection. This has been the subject of considerable discussion but unfortunately remains unclear (39). Knowing when to upgrade or relax precautions depends upon reliable and timely assessment of transmission and the consequences of infection. This is not a trivial problem. For instance, in many places around the world including some parts of the United States, testing and reporting of COVID-19 infection, hospitalization and death has lapsed, and wastewater monitoring has become the main source of data used to infer prevalence.
We need to develop better ways to monitor our environment for indicators of respiratory pathogen risk in near real time and geolocatable terms, and to use this information in quantitative ways to assess respiratory risk. For example, Puthussery et al. (40) recently reported a technology for near real-time analysis of air samples for SARS-CoV-2 or other viruses that might be used to estimate the risk of transmission from indoor air for a specific time and location. Similar technologies might also be used to perform near real-time testing of exhaled air from individuals to identify infection and the need for source control (41). While SARS-CoV-2 has been of greatest concern in recent times, the risks posed by other aerosol transmissible pathogens, or the combined risks from several pathogens circulating simultaneously in a community may warrant elevated precautionary measures; such protocols must become part of preparedness for future pandemics, some of which will involve, no doubt, airborne pathogens.
Lacking methods for sampling infectious aerosol concentrations in indoor spaces, it may be possible to assess risk qualitatively. Important factors include indoor air quality, based on ventilation and air purification, the likely number of encounters with potential sources (while noting that many infected persons can be asymptomatic or presymptomatic) and duration of exposure. When community transmission of a respiratory pathogen is widespread, exposure to healthcare workers and visitors with occult infections, in addition to infected patients, becomes a risk factor.
Universal masking policies in healthcare facilities have to consider potential obstacles to compliance including the available supply of masks and respirators. In some countries, regulators require periodic fit testing of respirators for employees who may be required to use them in the workplace. While the use of fit tested respirators is more likely to provide optimal protection from aerosol transmission than when respirators are worn without fit testing, a well-designed respirator that fits most people well is likely to provide better protection than surgical masks or other relatively less effective face coverings (26). Thus, providing respirators for use by patients, even when not fit tested, may be a rational protective measure. Whether healthcare workers or patients can or should be compelled to use respirators, surgical masks, or other face coverings is a complex legal, political and administrative problem with no easy answers.
Some have expressed practical concerns about the supply of respirators. At the current time, respirators are in abundant supply, however early in the COVID-19 pandemic this was not the case. When respirators are in short supply, it is important to realize that a single filtering facepiece respirator (e.g., N95, FFP2) can be worn at least a few times without losing its ability to fit and filter effectively. Once trapped in the filtering material of the respirator, particles remain bound indefinitely (42, 43). Greater routine use of respirators might well stimulate improvements in design that improve comfort and ease of donning and doffing; and a steady demand for respirators might make the supply chain more robust. Reusable elastomeric respirators offer advantages in comparison to disposable respirators (44) because they can be cleaned and their filters are very long lasting. Especially in pandemic or surge situations, elastomeric respirators have considerable strategic value.
In conclusion, there is convincing evidence for aerosol transmission of many pathogens, including some with pandemic potential, such as influenza and corona viruses. Healthcare facilities should endeavor to improve ventilation and air purification to reduce exposure of healthcare workers and patients to dangerous aerosols. When the risk of aerosol transmission is elevated, especially when transmission in the community is widespread, masking healthcare workers and patients, preferably with respirators rather than surgical masks, will make healthcare safer for all.
Author contributions
LB: Conceptualization, Writing – original draft, Writing – review & editing. AB: Conceptualization, Writing – original draft, Writing – review & editing. RT: Writing – review & editing. MK: Writing – review & editing. RS: Writing – review & editing. RH: Writing – review & editing. SJ: Writing – review & editing. MO: Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Hatfield KM, Baggs J, Maillis A, Warner S, Jernigan JA, Kadri SS, et al. Assessment of hospital-onset SARS-CoV-2 infection rates and testing practices in the US, 2020-2022. JAMA Netw Open. (2023) 6:e2329441. doi: 10.1001/jamanetworkopen.2023.29441
2. Jimenez JL, Marr LC, Randall K, Ewing ET, Tufekci Z, Greenhalgh T, et al. What were the historical reasons for the resistance to recognizing airborne transmission during the COVID-19 pandemic? Indoor Air. (2022) 32:e13070. doi: 10.1111/ina.13070
3. Morawska L, Milton DK. It is time to address airborne transmission of coronavirus disease 2019 (COVID-19). Clin Infect Dis. (2020) 71:2311–3. doi: 10.1093/cid/ciaa939
4. Goldberg L, Levinsky Y, Marcus N, Hoffer V, Gafner M, Hadas S, et al. SARS-CoV-2 Infection among health care workers despite the use of surgical masks and physical distancing-the role of airborne transmission. Open Forum Infect Dis. (2021) 8:ofab036. doi: 10.1093/ofid/ofab036
5. Alsved M, Nygren D, Thuresson S, Fraenkel CJ, Medstrand P, Londahl J. Size distribution of exhaled aerosol particles containing SARS-CoV-2 RNA. Infect Dis. (2023) 55:158–63. doi: 10.1080/23744235.2022.2140822
6. Alsved M, Nyström K, Thuresson S, Nygren D, Patzi-Churqui M, Hussein T, et al. Infectivity of exhaled SARS-CoV-2 aerosols is sufficient to transmit COVID-19 within minutes. Sci Rep. (2023) 13:21245. doi: 10.1038/s41598-023-47829-8
7. Tellier R. COVID-19: the case for aerosol transmission. Interface Focus. (2022) 12:20210072. doi: 10.1098/rsfs.2021.0072
8. Wang CC, Prather KA, Sznitman J, Jimenez JL, Lakdawala SS, Tufekci Z, et al. Airborne transmission of respiratory viruses. Science. (2021) 373:eabd9149. doi: 10.1126/science.abd9149
9. Klompas M, Baker MA, Griesbach D, Tucker R, Gallagher GR, Lang AS, et al. Transmission of SARS-CoV-2 from asymptomatic and presymptomatic individuals in healthcare settings despite medical masks and eye protection. Clin Infect Dis. (2021) 73:1693–5. doi: 10.1093/cid/ciab218
10. Couch RB, Knight V, Douglas RG Jr, Black SH, Hamory BH. The minimal infectious dose of adenovirus type 4; the case for natural transmission by viral aerosol. Trans Am Clin Climatol Assoc. (1969) 80:205–11.
11. Welsh HH, Lennette EH, Abinanti FR, Winn JF. Air-borne transmission of Q fever: the role of parturition in the generation of infective aerosols. Ann N Y Acad Sci. (1958) 70:528–40. doi: 10.1111/j.1749-6632.1958.tb35409.x
12. Couch RB, Douglas RG Jr, Lindgren KM, Gerone PJ, Knight V. Airborne transmission of respiratory infection with coxsackievirus A type 21. Am J Epidemiol. (1970) 91:78–86. doi: 10.1093/oxfordjournals.aje.a121115
13. Alford RH, Kasel JA, Gerone PJ, Knight V. Human influenza resulting from aerosol inhalation. Proc Soc Exp Biol Med. (1966) 122:800–4. doi: 10.3181/00379727-122-31255
14. Nguyen TM, Ilef D, Jarraud S, Rouil L, Campese C, Che D, et al. A community-wide outbreak of legionnaires disease linked to industrial cooling towers–how far can contaminated aerosols spread? J Infect Dis. (2006) 193:102–11. doi: 10.1086/498575
15. Riley RL, Mills CC, O'Grady F, Sultan LU, Wittstadt F, Shivpuri DN. Infectiousness of air from a tuberculosis ward. Ultraviolet irradiation of infected air: comparative infectiousness of different patients. Am Rev Respir Dis. (1962) 85:511–25.
16. Kulkarni H, Smith CM, Lee Ddo H, Hirst RA, Easton AJ, O'Callaghan C. Evidence of respiratory syncytial virus spread by aerosol. Time to revisit infection control strategies? Am J Respir Crit Care Med. (2016) 194:308–16. doi: 10.1164/rccm.201509-1833OC
17. Marks JS, Serdula MK, Halsey NA, Gunaratne MV, Craven RB, Murphy KA, et al. Saturday night fever: a common-source outbreak of rubella among adults in Hawaii. Am J Epidemiol. (1981) 114:574–83. doi: 10.1093/oxfordjournals.aje.a113223
18. Riley EC, Murphy G, Riley RL. Airborne spread of measles in a suburban elementary school. Am J Epidemiol. (1978) 107:421–32. doi: 10.1093/oxfordjournals.aje.a112560
19. Hamner L, Dubbel P, Capron I, Ross A, Jordan A, Lee J, et al. High SARS-CoV-2 attack rate following exposure at a choir practice - Skagit County, Washington, March 2020. MMWR Morb Mortal Wkly Rep. (2020) 69:606–10. doi: 10.15585/mmwr.mm6919e6
20. Eichenwald HF, Kotsevalov O, Fasso LA. The “cloud baby”: an example of bacterial-viral interaction. Am J Dis Child. (1960) 100:161–73. doi: 10.1001/archpedi.1960.04020040163003
21. Leclair JM, Zaia JA, Levin MJ, Congdon RG, Goldmann DA. Airborne transmission of chickenpox in a hospital. N Engl J Med. (1980) 302:450–3. doi: 10.1056/NEJM198002213020807
22. Wehrle PF, Posch J, Richter KH, Henderson DA. An airborne outbreak of smallpox in a German hospital and its significance with respect to other recent outbreaks in Europe. Bull World Health Organ. (1970) 43:669–79.
24. Burdick H, Maibach H. Clinical relevance of masks in the operating room? A systematic review. Clin Infect Pract. (2021) 12:100087. doi: 10.1016/j.clinpr.2021.100087
25. Adenaiye OO, Lai J, de Mesquita PJB, Hong F, Youssefi S, German J, et al. Infectious severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in exhaled aerosols and efficacy of masks during early mild infection. Clin Infect Dis. (2022) 75:e241–8. doi: 10.1093/cid/ciab797
26. Lai J, Coleman KK, Tai S-HS, German J, Hong F, Albert B, et al. Relative Efficacy of Masks and Respirators as Source Control for Viral Aerosol Shedding from People Infected with SARS-CoV-2: A Human Controlled Trial. (2023). doi: 10.2139/ssrn.4631479
27. Grinshpun SA, Haruta H, Eninger RM, Reponen T, McKay RT, Lee SA. Performance of an N95 filtering facepiece particulate respirator and a surgical mask during human breathing: two pathways for particle penetration. J Occup Environ Hyg. (2009) 6:593–603. doi: 10.1080/15459620903120086
28. Lee SA, Grinshpun SA, Reponen T. Respiratory performance offered by N95 respirators and surgical masks: human subject evaluation with NaCl aerosol representing bacterial and viral particle size range. Ann Occup Hyg. (2008) 52:177–85. doi: 10.1093/annhyg/men005
29. Lawton T, Butler M, Peters C. Airborne protection for staff is associated with reduced hospital-acquired COVID-19 in English NHS trusts. J Hosp Infect. (2022) 120:81–4. doi: 10.1016/j.jhin.2021.11.018
30. MacIntyre CR, Chughtai AA, Rahman B, Peng Y, Zhang Y, Seale H, et al. The efficacy of medical masks and respirators against respiratory infection in healthcare workers. Influenza Other Respir Viruses. (2017) 11:511–7. doi: 10.1111/irv.12474
31. Andrejko KL, Pry JM, Myers JF, Fukui N, DeGuzman JL, Openshaw J, et al. Effectiveness of face mask or respirator use in indoor public settings for prevention of SARS-CoV-2 infection - California, February-December 2021. MMWR Morb Mortal Wkly Rep. (2022) 71:212–6. doi: 10.15585/mmwr.mm7106e1
32. Tellier R, Li Y, Cowling BJ, Tang JW. Recognition of aerosol transmission of infectious agents: a commentary. BMC Infect Dis. (2019) 19:101. doi: 10.1186/s12879-019-3707-y
33. Tellier R. Review of aerosol transmission of influenza A virus. Emerg Infect Dis. (2006) 12:1657–62. doi: 10.3201/eid1211.060426
34. Tang JW, Tellier R, Li Y. Hypothesis: all respiratory viruses (including SARS-CoV-2) are aerosol-transmitted. Indoor Air. (2022) 32:e12937. doi: 10.1111/ina.12937
35. Sherertz RJ, Bassetti S, Bassetti-Wyss B. “Cloud” health-care workers. Emerg Infect Dis. (2001) 7:241–4. doi: 10.3201/eid0702.010218
36. Vonberg RP, Gastmeier P. Nosocomial aspergillosis in outbreak settings. J Hosp Infect. (2006) 63:246–54. doi: 10.1016/j.jhin.2006.02.014
37. Klompas M, Milton DK, Rhee C, Baker MA, Leekha S. Current insights into respiratory virus transmission and potential implications for infection control programs : a narrative review. Ann Intern Med. (2021) 174:1710–8. doi: 10.7326/M21-2780
38. Hinckley AF, Biggerstaff BJ, Griffith KS, Mead PS. Transmission dynamics of primary pneumonic plague in the USA. Epidemiol Infect. (2012) 140:554–60. doi: 10.1017/S0950268811001245
39. Landelle C, Birgand G, Price JR, Mutters NT, Morgan DJ, Lucet JC, et al. Considerations for de-escalating universal masking in healthcare centers. Antimicrob Steward Healthc Epidemiol. (2023) 3:e128. doi: 10.1017/ash.2023.200
40. Puthussery JV, Ghumra DP, McBrearty KR, Doherty BM, Sumlin BJ, Sarabandi A, et al. Real-time environmental surveillance of SARS-CoV-2 aerosols. Nat Commun. (2023) 14:3692. doi: 10.1038/s41467-023-39419-z
41. Ghumra DP, Shetty N, McBrearty KR, Puthussery JV, Sumlin BJ, Gardiner WD, et al. Rapid direct detection of SARS-CoV-2 aerosols in exhaled breath at the point of care. ACS Sens. (2023) 8:3023–31. doi: 10.1021/acssensors.3c00512
42. Birkner JS, Fung D, Hinds WC, Kennedy NJ. Particle release from respirators, part I: determination of the effect of particle size, drop height, and load. J Occup Environ Hyg. (2011) 8:1–9. doi: 10.1080/15459624.2011.534975
43. Fisher EM, Richardson AW, Harpest SD, Hofacre KC, Shaffer RE. Reaerosolization of MS2 bacteriophage from an N95 filtering facepiece respirator by simulated coughing. Ann Occup Hyg. (2012) 56:315–25. doi: 10.1093/annhyg/mer101
Keywords: respirator, pandemic, SARS-CoV-2, respiratory protection, mask, aerosol transmission, indoor air, N95
Citation: Brosseau LM, Bowdle A, Tellier R, Klompas M, Schooley RT, Harrison R, Jelacic S and Osterholm MT (2024) The time has come to protect healthcare workers and patients from aerosol transmissible disease. Front. Public Health 12:1378567. doi: 10.3389/fpubh.2024.1378567
Received: 29 January 2024; Accepted: 09 April 2024;
Published: 23 April 2024.
Edited by:
Kingston Rajiah, Ulster University, United KingdomReviewed by:
Christopher Bradburne, Johns Hopkins University, United StatesCopyright © 2024 Brosseau, Bowdle, Tellier, Klompas, Schooley, Harrison, Jelacic and Osterholm. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Andrew Bowdle, Ym93ZGxlQHUud2FzaGluZ3Rvbi5lZHU=
 Lisa M. Brosseau
Lisa M. Brosseau Andrew Bowdle
Andrew Bowdle Raymond Tellier3
Raymond Tellier3 Michael Klompas
Michael Klompas Robert T. Schooley
Robert T. Schooley Robert Harrison
Robert Harrison Srdjan Jelacic
Srdjan Jelacic Michael T. Osterholm
Michael T. Osterholm