- 1Guangdong Province Hospital for Occupational Disease Prevention and Treatment, Guangzhou, China
- 2School of Public Health, Shanxi Medical University, Taiyuan, China
- 3Center for Public Health and Epidemic Preparedness and Response, School of Public Health, Peking University Health Science Center, Beijing, China
- 4School of Public Health, Southern Medical University, Guangzhou, China
- 5Department of Medical Statistics, School of Public Health, Sun Yat-sen University, Guangzhou, China
- 6Tianjin Key Laboratory of Radiation Medicine and Molecular Nuclear Medicine, Institute of Radiation Medicine, Chinese Academy of Medical Sciences and Peking Union Medical College, Tianjin, China
Background: Although radiation workers’ exposure levels consistently remained below established safety thresholds, accumulating evidence demonstrates that chronic low-dose ionizing radiation exposure may still pose significant health risks to humans. We aimed to explore the relationship between the years of low-dose radiation work and dyslipidemia.
Methods: We collected occupational and physical examination data of 10,338 radiation workers from 1,200 workplaces during 2019–2020 in Guangdong Province, China. After controlling for social demographic and health behavior confounders, we used a mixed-effects model to assess the association of ionizing radiation exposure duration with blood lipid biomarkers as well as the prevalence of dyslipidemia. We further comprehensively evaluated the modifying effects of various demographic characteristics, health behavior factors, and air pollutant concentrations.
Results: We found that participants with prolonged ionizing radiation exposure tended to have 8–40% higher levels of total cholesterol (TC) compared to those with < 10 years of exposure. The estimates were 9–23% for triglycerides (TG) and 5–26% for low-density lipoprotein cholesterol (LDL-C). Similar disparities were observed for the prevalence of overall dyslipidemia, abnormal TC or TG, hypercholesterolemia, hypertriglyceridemia, and high β-lipoproteinemia, with odds being 1.51–2.45 times higher in the group with > 30 years of ionizing radiation exposure compared to others. Our estimates further indicated greater effect estimates for prolonged ionizing radiation exposure and the prevalence of lipid abnormalities (p < 0.05) among the females, unmarried ones, and the workers with normal BMI.
Conclusion: These findings suggest a deleterious effect of prolonged ionizing radiation exposure on lipid metabolism, with certain groups of workers being particularly vulnerable.
1 Introduction
Dyslipidemia is a common medical condition characterized by abnormal lipid profiles in the bloodstream. It is highly prevalent among Chinese adults (1), and has been responsible for the increased risk of a variety of cardiovascular diseases such as ischemic heart disease (IHD) and stroke (2). According to the estimates released by the WHO in 2012, dyslipidemia contributed to 18% of cases of IHD and 56% of strokes, resulting in over 4 million deaths globally each year (3).
It is widely recognized that chronic exposure to ionizing radiation increases the risk of cardiovascular diseases, likely mediated through the development of dyslipidemia. The association between ionizing radiation and disturbed lipid metabolism is biologically plausible. For example, Erica Werner et al. exposed human bronchial epithelial cells and lung cancer cell lines to ionizing radiation (5 Gy X radiation) and found a notable elevation in the activity of cholesterol biosynthesis enzymes and cellular cholesterol levels (10–30%) (4). Another study by Chukwuemeka N et al. exposed rats to 3 Gy of total body irradiation. After the initial exposure (5 days), serum triglyceride and total cholesterol levels increased significantly, with further increments observed after the second (10 days) and third (15 days) exposures (5). Changes in the level of cholesterol and triglycerides as observed in these experimental studies suggest the potential of ionizing radiation to disturb lipid metabolism (6). Moreover, emerging epidemiological evidence has shed light on the association between ionizing radiation exposure and dyslipidemia with its subclinical manifestations. Wen et al. recently demonstrated a significant correlation between chronic occupational radiation exposure and dyslipidemia among radiologists (7). Complementing these findings, Oslina et al. identified molecular alterations in blood samples of radiation-exposed workers that may indicate radiation-associated dyslipidemia linked to atherogenic progression (8).
Occupational exposure to ionizing radiation is believed to be higher than regular radiation from the environment among the general population (9). However, radiation workers are typically well-protected and exposed to minimal ionizing radiation, which remain significantly below the International Commission on Radiological Protection (ICRP)‘s safety threshold of an annual effective dose limit of 20 mSv/year, averaged over 5 years, with no single year exceeding 50 mSv for occupational exposure (10). Existing studies indicate that the vast majority of occupational radiation workers receive annual effective doses in the range of 1–5 mSv, well below the ICRP’s recommended limit (11–13). Although low-dose ionizing radiation generally would not lead to an apparent health impact in the short term, it is still unclear whether prolonged exposure to such low-dose ionizing radiation remains safe for humans. In situations where dose may not be a concern, the duration of such exposure may become a potential predictor for adverse health outcomes (14–17). However, existing research on the relationship between the duration of long-term low-dose exposure and blood lipid metabolism indicators remains extremely limited.
To address this important knowledge gap, we investigated a large group of radiation workers to clarify the relationship between their durations of ionizing radiation and the levels of multiple lipid biomarkers, as well as the prevalence of dyslipidemia.
2 Methods
2.1 Study design and population
This study retrospectively reviewed health examination data from Guangdong Provincial Occupational Disease Prevention and Control Hospital, including 11,663 radiation workers from 1,200 institutions across the province between 2019 and 2020. Study participants were recruited from occupational sectors exposed to ionizing radiation, including medical radiology, nuclear industry operations, and industrial radiography. According to local regulations, radiation workers are required to participate in a health examination every 2 years. The study utilized retrospectively collected anonymized health examination records, thus requiring neither active recruitment nor direct interviews. Participant consent was waived under ethical approval (GDHOD MEC 2023034) in accordance with regulations governing research involving de-identified medical data. All the participants had at least 1 year of working experience and had no major health issues such as cancer or severe chronic conditions. Participants who 1. lacked information on lipid profiles, 2. had a family history of cardiovascular and cerebrovascular diseases, or 3. worked outside of Guangdong Province were excluded, resulting in a total of 10,338 participants in the analysis (Figure 1). The research protocol was approved by the Medical Ethics Committee of the Guangdong Provincial Hospital for Occupational Disease Prevention and Control (Approval No. GDHOD MEC 2023034).

Figure 1. Cohort diagram illustrating the study population. The missing concentration of lipid index in the 210 participants was due to the lack of blood samples available for use in lipid analysis; The lack of information on smoking, drinking and marital status was due to incomplete information collection.
2.2 Exposure measurement and covariates
Information on the duration of ionizing radiation exposure was collected from the individuals and confirmed by the employers. Occupational exposure duration was assessed using a dual-verification approach combining worker self-reported data with official employer records, thereby minimizing potential recall bias and ensuring data reliability. Participants were divided into four groups (in 10-year intervals): 1–10 years, 10–20 years, 20–30 years, and > 30 years. Before the physical examination, all participants underwent measurements of height and weight, and body mass index (BMI) was calculated from them. Information on basic demographic characteristics (i.e., age, gender, and marital status) and health behaviors (i.e., smoking and drinking) of the participants was obtained from the health examination database.
Since existing studies suggested a link between ambient air pollution and lipid indicators (18), we incorporated the concentration of major air pollutants, such as PM2.5, O3, SO2, and NO2 as potential confounders. The concentrations were determined for each participant by accessing the China High-resolution Air Pollution (CHAP) dataset according to the geocoded address of the workplace. The CHAP dataset is a well-validated, high-quality dataset on near-surface air pollution covering 10 km × 10 km spatially (19). It has been extensively utilized in previous research (20, 21). The average air pollution concentration over the year before the physical examination date was defined as the exposure variable. The distribution of pollutant concentrations (mean ± standard error) was as follows: 27.26 ± 2.66 μg/m3 for PM2.5, 103.35 ± 9.59 μg/m3 for O3, 8.58 ± 1.33 μg/m3 for SO2, and 41.76 ± 10.05 μg/m3 for NO2 (See Supplementary material).
2.3 Outcome definition
Blood samples were obtained following a fasting period of at least 10 h overnight. We used Mindray automatic analyzer (BS2000, China) to obtain the concentrations of lipid biomarkers, including TC, TG, high-density lipoprotein cholesterol (HDL-C), and LDL-C. Dyslipidemia was defined based on the established standards (22): 1. hypercholesterolemia (TC ≥ 6.2 mmol/L); 2. hypertriglyceridemia (TG ≥ 2.3 mmol/L); 3. hypolipoproteinemia (HDL-C < 1.0 mmol/L); 4. high β lipoproteinemia (LDL-C ≥ 4.1 mmol/L). The definition of overall dyslipidemia encompassed the occurrence of at least one of the four lipid abnormalities mentioned above.
2.4 Statistical analysis
We utilized mean (standard deviation, SD) for continuous variables, frequency (percentage) for categorical variables and using chi-square tests or analysis of variance to examine the disparities across different exposure duration groups, as appropriate. To assess the association between the duration of ionizing radiation exposure and lipid profiles, we developed three models using mixed-effects models.
Model 1. A crude model, only adjusted for a workplace-level random intercept.
Model 2. Further adjusted for age, gender, and marital status (i.e., married vs. unmarried/widowed/single) based on Model 1.
Model 3. Further adjusted for health behavior indicators, including BMI (kg/m2), smoking (i.e., non-smokers, light smokers with <100 cigarettes/year, moderate smokers with 100–200 cigarettes/year, heavy smokers with > 200 cigarettes/year), and drinking (i.e., yes or no), and additionally adjusted for the concentrations of air pollutants including PM2.5, SO2, O3, and NO2.
To evaluate the collinearity in the final model, we computed the variance inflation factor (VIF) to confirm that all variables in the model had VIF values < 5. Furthermore, we performed a comprehensive set of stratified analyses by baseline characteristics, health behaviors, and air pollution concentrations based on the final model. This allowed us to estimate how these factors modified the relationship between exposure duration and lipid levels, as well as dyslipidemia types.
To ensure the reliability of the findings, we carried out sensitivity analyses by (1) excluding the participants with an extremely short period of radiation working years (i.e., last 5%, n = 571) and (2) excluding those with hypertension (n = 1,344). Statistically significant estimates were identified with p-values below 0.05. All statistical analyses were performed utilizing R version 4.1.2.
3 Results
3.1 Participant characteristics
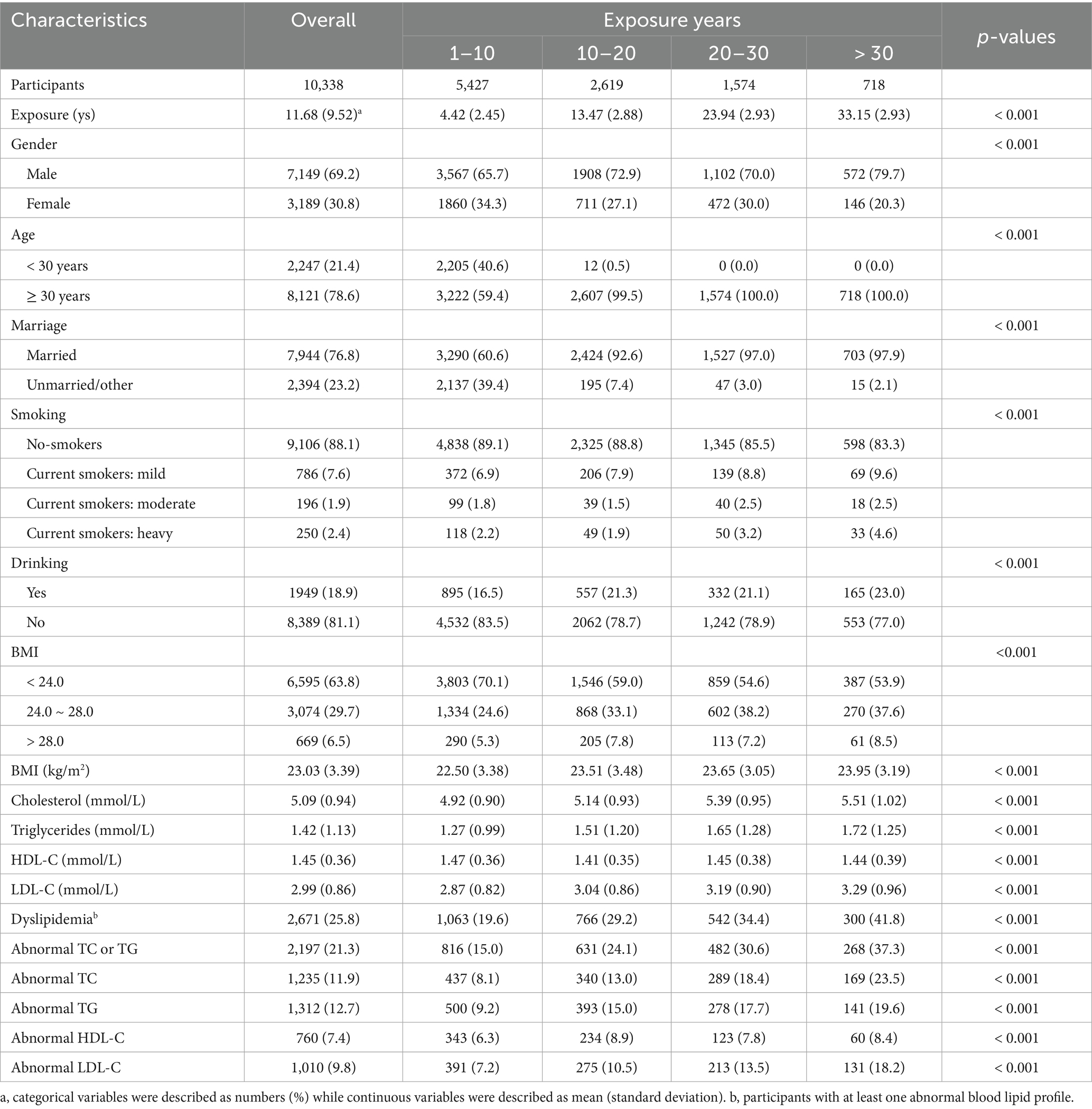
Among the 10,338 participants, 3,189 (30.8%) were female, and 8,121 (78.6%) were aged ≥ 30 years. The overall average BMI was 23.03 ± 3.39 kg/m2. Among the participants, 29.7% were overweight and 6.5% were obese. Additionally, we observed a significantly higher proportion of married ones than unmarried ones (76.8% vs. 23.2%). A total of 1,232 individuals (11.9%) were smokers, with 786 (7.6%) being classified as light smokers, 196 (1.9%) as moderate, and 250 (2.4%) as heavy smokers. A total of 1,949 individuals (18.9%) drank alcohol (Table 1). There were 2,671 individuals with dyslipidemia, among which 29.2% had 10–20 exposure years, 34.4% had 20–30 exposure years, and 41.8% had >30 exposure years. Compared to participants with normal blood lipids, those with dyslipidemia were likely to have more exposure years. Similar disparities were also observed with the criteria of abnormal blood lipids based on cholesterol, triglycerides, and LDL-C levels (Table 2).
3.2 Relationship of radiation exposure duration with blood lipid levels and dyslipidemia
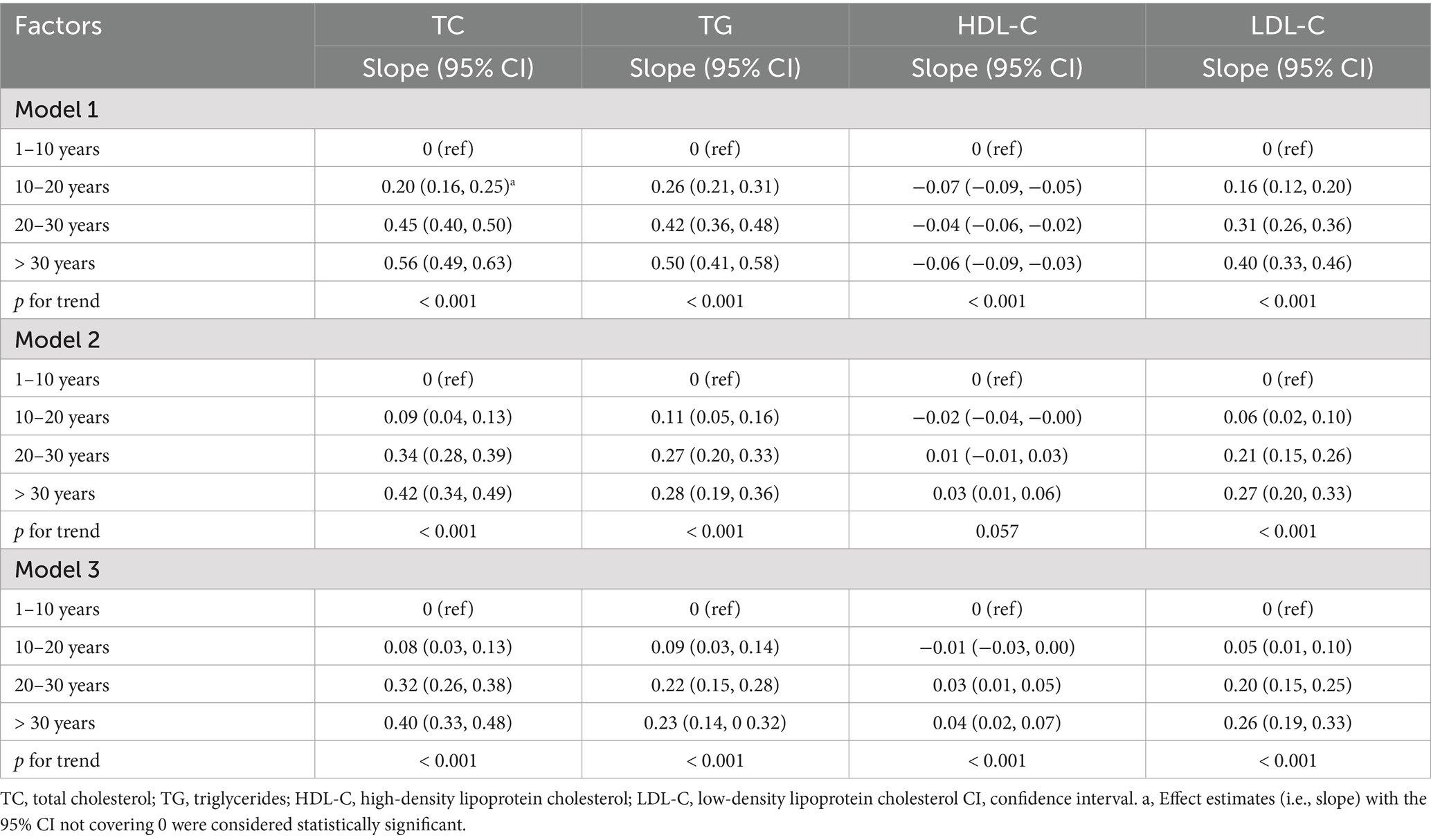
We found that as exposure years increased, levels of TC, TG, and LDL-C increased in all models. After adjusting for covariates, the regression coefficient (β) slightly decreased but remained statistically significant (p < 0.05). However, radiation exposure had no significant influence on HDL-C levels. According to the results of the final model, compared to participants with 1–10 exposure years, those with >30 exposure years were observed with an increase of 0.40 (95% CI 0.33–0.48) mmol/L, 0.23 (95% CI 0.14–0.32) mmol/L and 0.26 (95% CI 0.19–0.33) mmol/L in serum concentrations of TC, TG, and LDL-C, respectively (Table 3).
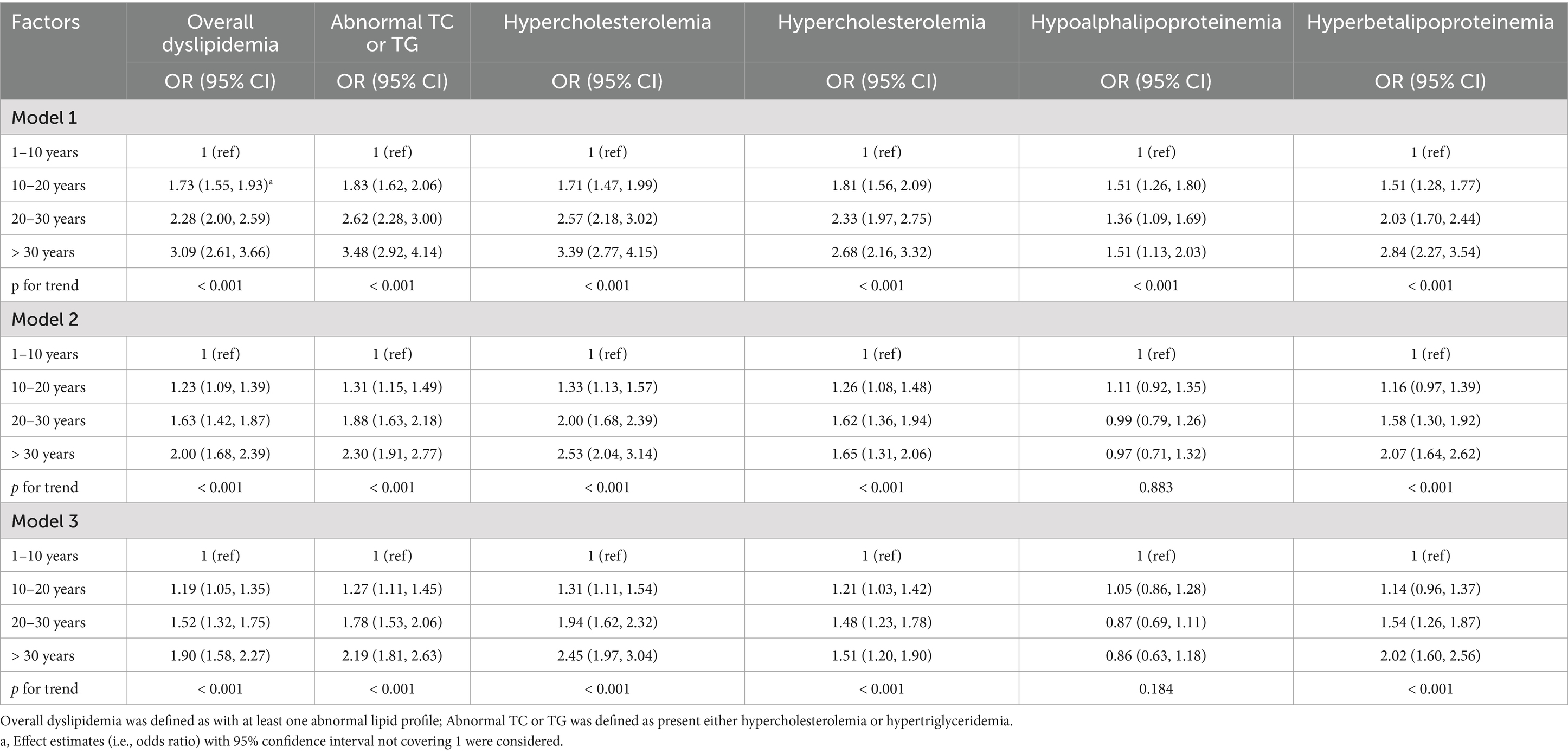
Consistent with the findings from continuous lipid spectrum observations, there was a positive exposure-response relationship between low-dose ionizing radiation exposure duration and the prevalence of dyslipidemia. Overall, the prevalence of dyslipidemia increased with increasing radiation exposure years. Compared to the group with the shortest exposure duration (1–10 years, the reference group), participants with prolonged radiation exposure tended to have a 73% (95% CI 1.55–1.93), 128% (95% CI 2.00–2.59), and 209% (95% CI 2.61–3.66) greater risk of dyslipidemia. After adjusting for important covariates, the odds ratios (95% CI) of dyslipidemia for groups with 10–20 years, 20–30 years, and > 30 years of exposure were 1.19 (95% CI 1.05–1.35), 1.52 (95% CI 1.32–1.75), and 1.90 (95% CI 1.58–2.27), respectively, relative to the reference group. We also found that the highest radiation exposure group (> 30 years) had significantly increased risks of overall dyslipidemia, abnormal TC or TG, hypercholesterolemia, hypertriglyceridemia, and high β-lipoproteinemia, with odds ratios of 1.90 (95% CI 1.58–2.27), 2.19 (95% CI 1.81–2.63), 2.45 (95% CI 1.97–3.04), 1.51 (95% CI 1.20–1.90), and 2.02 (95% CI 1.60–2.56), respectively. There was no significant association of radiation exposure years with the prevalence of hypo α-lipoproteinemia (p > 0.05) (Table 2).
3.3 Factors modifying the relationship between radiation exposure and blood lipid metabolism
We observed that the association of radiation exposure years with the prevalence of dyslipidemia significantly varied by gender (p for interaction < 0.001). Results in Tables 4, 5 suggested that female radiation workers were more susceptible to the effects of radiation exposure than males in terms of the levels of TC, LDL-C, and the prevalence of dyslipidemia. There was a significant interaction between marital status and radiation exposure years in the prevalence of dyslipidemia (p for interaction = 0.001). Unmarried/other participants in the high-exposure group (> 30 years) had a greater impact on the estimated effects (total cholesterol β = 0.95, 95% CI 0.51–1.39, p for interaction < 0.001; LDL-C β = 0.82, 95% CI 0.42–1.22, p for interaction = 0.006; overall dyslipidemia OR = 10.07, 95% CI 3.24–31.30, p for interaction = 0.001; abnormal TC or TG OR = 11.74, 95% CI 3.82–36.07, p for interaction < 0.001; hypercholesterolemia OR = 9.60, 95% CI 3.08–29.94, p for interaction = 0.007). A significant difference in the association between radiation exposure and overall dyslipidemia was found among BMI categories (p for interaction = 0.005). Obesity (BMI ≥ 28.0) and overweight (24.0 ≤ BMI < 28.0) appeared to be protective factors, with the lowest prevalence of overall dyslipidemia observed in obesity participants with low radiation exposure (10–20 years) (risk ratio 0.96, 95% CI 0.63–1.47). There was no significant interaction between smoking, drinking, and exposure years in the prevalence of dyslipidemia (p for interaction were 0.149 and 0.640, respectively). Within each smoking and drinking category, increasing radiation exposure duration was associated with an increased prevalence of overall dyslipidemia. Participants with 10–20 exposure years and smoking had the lowest risk (OR = 1.15, 95% CI 0.83–1.61). Non-drinkers in the 10–20 exposure years group had the lowest prevalence of dyslipidemia (OR = 1.16, 95% CI 1.01–1.33) (Tables 4, 5). Additionally, we observed that participants with long-term exposure to high concentrations of PM2.5, O3, and NO2 were more susceptible to the effects of radiation exposure years on overall dyslipidemia relative to their counterparts, although the interaction terms may not be statistically significant.
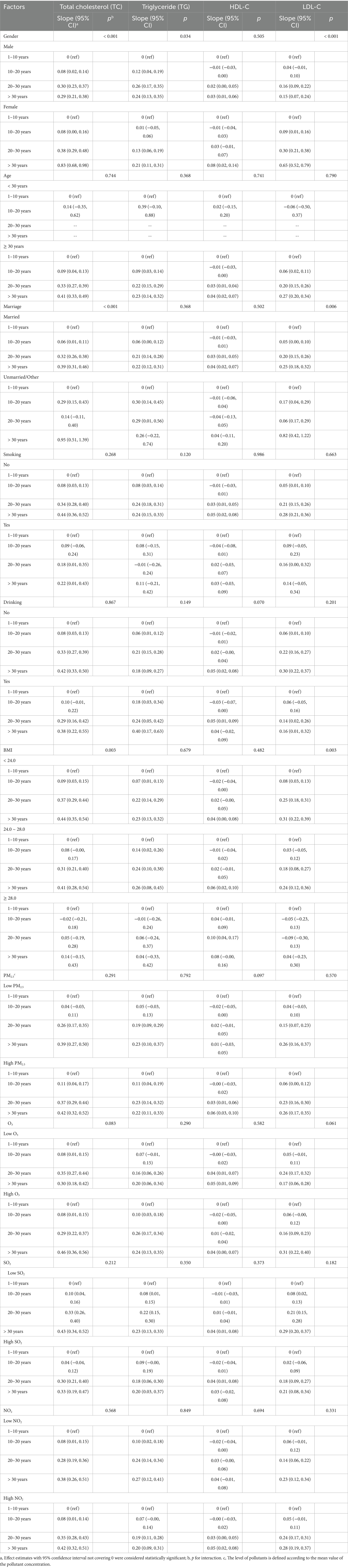
Table 4. The modification effect of basic participant characteristics on the association between ionizing radiation exposure duration and blood lipid profiles.
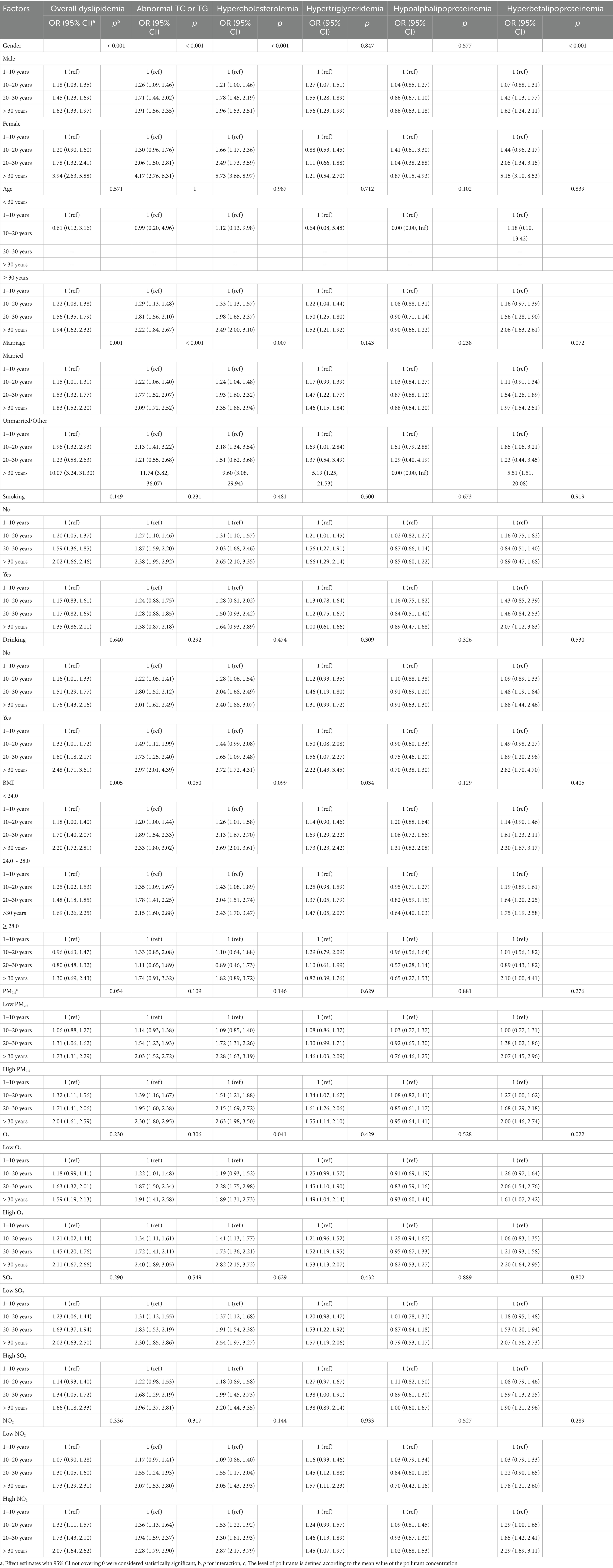
Table 5. The modification effect of basic participant characteristics on the association between ioniziang radiation exposure duration and dyslipidemia.
We observed similar effect estimates when excluding participants with a working experience of 1 year or less or excluding those with hypertension (See Supplementary material).
4 Discussion
Our results confirmed that occupational exposure duration to ionizing radiation was significantly associated with higher levels of TC, TG, and LDL-C, as well as a higher risk of blood lipid abnormalities. We found the association differed by gender and marriage, with females and those unmarried being more susceptible. Participants with obesity (≥ 28.0 kg/m2) tended to have a lower risk of blood lipid abnormalities following exposure.
4.1 Interpretation of the results
Recent evidence has consistently demonstrated the potential link between ionizing radiation exposure and dyslipidemias. In a rat model, ionizing radiation exposure altered lipid metabolism, resulting in significant increases in TC, TG, and LDL-C, as well as decreases in HDL-C concentration. Furthermore, Amorim et al. and Daniel et al. found elevated levels of triglycerides and LDL-C, and free fatty acids decades after radiation (23, 24), indicating impaired lipid metabolism related to the radiation exposure (25). An increased risk of dyslipidemia and coronary heart disease was also observed after radiation therapy (26). In epidemiological studies, Wen et al. observed a dose-dependent increase in dyslipidemia risk and elevated TG levels, with the systemic immune-inflammation index (SII) potentially mediating the association between cumulative radiation dose and TG concentrations (7). Research by Oslina et al. revealed blood molecular alterations in individuals with occupational exposure to dual radiation sources (α-particles internally and γ-rays externally), potentially marking dyslipidemia connected to atherogenic processes (8). Although the link between ionizing radiation exposure and dyslipidemias has been investigated, the epidemiological evidence is still limited (7, 8). Therefore, our study significantly contributes to the epidemiological evidence regarding the association between ionizing radiation exposure and dyslipidemias. It is noteworthy that the OR of dyslipidemia for the groups with 20–30 exposure years and > 30 exposure years could increase by 52% (95% CI 1.32–1.75) and 90% (95% CI 1.58–2.27), relative to the reference group.
Our study suggested multiple effect modifiers underlying the association between ionizing radiation exposure and serum lipid profile, including gender, marital status and BMI. Specifically, we observed greater effect estimates among the females, the unmarried, and the workers with normal BMI. Although existing evidence is limited on the human vulnerability toward the association between ionizing radiation and dyslipidemia, some studies suggested that these subgroups were more susceptible to the impact of ionizing radiation on other outcomes. Similar interpretations may help understanding the observed phenomenon of this study. For example, a study reviewed the primary human studies concerning the health risks associated with radiation exposure, revealing that females exposed to the same dose of radiation may face a significantly higher risk of experiencing and succumbing to radiation-induced cancer compared to males (27). Sex leads to genetic differences, hormonal regulation and DNA damage, which are considered possible driving mechanisms of radiation sensitivity and radiation-induced risk of dyslipidemia (28). Furthermore, a cohort study of industrial radiographers in Xinjiang, China, found that the long-term low-dose radiation exposure posed a risk for liver injury in the unadjusted analysis, with unmarried workers at increased risk compared to married workers. The mechanisms behind this may involve differences in lifestyle, hormonal roles, stress levels, or access to healthcare between married and single individuals, which interact with radiation exposure (29). Furthermore, the existing research on the impact of BMI on worker radiation exposure estimates is inconsistent. A cross-sectional study on the association between BMI and abdominal fat in preoperative liver CT scans of potential live donor liver and radiation dose showed that obese patients typically receive higher effective doses of ionizing radiation in imaging examinations compared to normal weight individuals, as the thickness of the surveyed area in obese individuals is larger, leading to higher radiation exposure (30, 31). The discrepancy between these findings and the outcomes of our study may be partly attributable to our use of BMI as a practical indicator of general obesity, derived from routinely collected occupational health records. However, abdominal obesity, particularly visceral fat, has been shown to play a more significant role in lipid metabolism dysregulation than BMI alone (32–34). More studies are needed in the future to replicate these findings and clarify the underlying causes.
4.2 Biological rationality
Ionizing radiation (IR) is commonly employed in both diagnostic and therapeutic applications. However, it is important to note that healthcare workers may inadvertently be exposed to IR (35). Although the levels of radiation experienced by healthcare workers are typically low, the cumulative ionizing dose over several years of work may have adverse effects on human health (13).
Several potential mechanisms have been suggested for the link between exposure to ionizing radiation and changes in the lipid spectrum. Ionizing radiation could induce excessive lipid peroxidation and reduce antioxidant levels, leading to oxidative stress and thereby disrupting lipid metabolism. This is also a key mechanism for regulating a programmed form of cell death in ferroptosis (36). The second mechanism is that ionizing radiation may disrupt the feedback regulation of cholesterol biosynthesis, leading to elevated levels of cholesterol in lung and possibly other possible post-mitotic tissues (4). This could affect the function and cellular processes of cholesterol-sensitive proteins. Furthermore, ionizing radiation can directly interact with and damage cell membranes, leading to lipid peroxidation and subsequent apoptosis and necrosis (37). Furthermore, radiation exposure could cause skin lipid remodeling, including changes in fatty acid and lipid composition in the skin, which may lead to radiation-induced skin lipid damage (38). Another possible mechanism is that radiation-induced increases in mitochondrial damage and insulin resistance could also dysregulate the lipid metabolism and insulin resistance profiles (39). Moreover, the epigenetic changes induced by IR may contribute to the changes in the expression of genes involved in lipid metabolism (40). Together, the existing evidence suggests that the deleterious impact of prolonged radiation exposure on lipid metabolism we observed is biologically plausible.
4.3 Strengths and limitations
Population-based epidemiological evidence demonstrated significant associations between chronic low-dose ionizing radiation exposure and dyslipidemia indicators, providing crucial scientific support for health risk evaluation in long-term radiation exposure settings. The study covered participants from 1,200 workplaces in Guangdong Province which enhances the applicability of our studies. The extensive sample size ensures ample statistical power to test moderate correlations and estimate variations in those association among various subgroups. Additionally, we established various lipid variables to comprehensively investigate the effect of radiation on lipid metabolism. Our findings have been validated through multiple sensitivity analyses.
However, some limitations should be acknowledged. First, we used occupational exposure duration to represent the ionizing radiation exposure since the individual-level radiation dose tended to be low and remains unknown. While duration of employment serves as a critical exposure metric under chronic low-dose conditions, individual dose data are also essential. However, due to limitations in data accessibility, this study did not incorporate individual dose measurements. Future studies should prioritize the collection of individual dose data to refine exposure assessments. Obtaining individual exposure data through dosimeters is feasible, as radiation workers could be required to wear personal monitoring devices. However, challenges include ensuring consistent device use, standardizing data collection across workplaces, and integrating historical dose records. Future studies should prioritize collaboration with occupational health agencies to access these data, enabling more precise dose–response analyses (41). Second, as this is a cross-sectional study, we could not establish the causal link between ionizing radiation exposure and lipid metabolism. The cross-sectional study design is susceptible to numerous confounding factors that can bias results. For instance, background prevalence, which varies by geographical location, age, or socioeconomic status of the study population, may skew findings. Other confounders include selection bias (e.g., underrepresentation in the sample), information bias (e.g., recall bias or measurement errors), temporal confounding (e.g., difficulty distinguishing the sequence of exposure and outcome), and genetic factors that may simultaneously influence the exposure-outcome relationship. These issues can compromise the reliability of causal inferences. Future research may require more rigorous designs with stronger causal inference capabilities, such as cohort studies. Third, we did not collect individual-level data on dietary habits and lifestyle factors, including physical activity, diet and exercise levels. These factors are known to influence lipid metabolism and may also modulate biological responses to ionizing radiation. The lack of such data introduces the possibility of residual confounding that may affect the interpretation of our results. Due to the retrospective design of the study, it was not feasible to collect this information. Future investigations should incorporate validated tools such as food frequency questionnaires, physical activity surveys, or relevant biomarkers to comprehensively assess lifestyle and behavioral factors. These approaches will help to reduce residual confounding and enhance the validity and generalizability of the findings.
5 Conclusion
Our study suggests that radiation workers with longer-term occupational ionizing radiation exposure have a higher risk of disturbed lipid levels. Females, unmarried individuals, and workers with normal BMI, appear to be more vulnerable. Further studies are still warranted to validate our results. Further validation in other radiation worker cohorts is needed to confirm its occupational health implications.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by the Medical Ethics Committee of the Guangdong Provincial Hospital for Occupational Disease Prevention and Control. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin because this study utilized only previously collected and fully anonymized clinical data/samples, with no possibility of identifying individuals through either direct or indirect means. The study protocol was approved by the Medical Ethics Committee of the Guangdong Provincial Hospital for Occupational Disease Prevention and Control with a waiver of written informed consent (Approval No. GDHOD MEC 2023034).
Author contributions
MZ: Conceptualization, Formal analysis, Methodology, Software, Validation, Visualization, Writing – original draft. XW: Conceptualization, Formal analysis, Methodology, Validation, Visualization, Writing – original draft. QL: Formal analysis, Software, Validation, Writing – original draft. ZW: Formal analysis, Software, Validation, Writing – original draft. XH: Conceptualization, Data curation, Investigation, Writing – original draft. XLi: Conceptualization, Data curation, Writing – original draft. YL: Conceptualization, Investigation, Writing – original draft. YP: Conceptualization, Investigation, Writing – original draft. ZLi: Conceptualization, Data curation, Writing – original draft. ZLiu: Data curation, Investigation, Writing – original draft. QW: Data curation, Investigation, Writing – original draft. YGo: Conceptualization, Data curation, Writing – original draft. PH: Conceptualization, Data curation, Writing – original draft. XLiu: Data curation, Investigation, Writing – original draft. SZ: Data curation, Investigation, Writing – original draft. SW: Data curation, Investigation, Writing – original draft. FZ: Data curation, Investigation, Writing – original draft. YQ: Conceptualization, Data curation, Writing – original draft. QN: Conceptualization, Data curation, Writing – original draft. JW: Conceptualization, Data curation, Writing – original draft. YGu: Conceptualization, Data curation, Writing – original draft. YH: Conceptualization, Data curation, Writing – original draft. WZ: Funding acquisition, Supervision, Validation, Visualization, Writing – review & editing. NZ: Supervision, Validation, Visualization, Writing – review & editing, Funding acquisition.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the National Natural Science Foundation of China (82204162); The CAMS Innovation Fund for Medical Science (2021-I2M-1-042); Young Elite Scientist Sponsorship Program by China Association for Science and Technology (YESS20230500); Natural Science Foundation of Guangdong Province, China (2025A1515010794, 2023A1515010085); Guangdong Provincial Pearl River Talents Program (0920220207); Basic and Applied Basic Research Foundation of Guangdong Province (2022A1515010823); Science and Technology Planning Project of Guangzhou (2023A04J2471); Guangzhou Municipal Science and Technology Bureau (2023A04J2072); Fundamental Research Funds for the Central Universities, Sun Yat-sen University (23qnpy108); Biomedical Industry Innovation Subsidy of Guangzhou Science and Technology Bureau (2023-02); The school of Public Health, Shanxi Medical University “233” Cooperation Project, Shanxi Province Higher Education “Billion Project” Science and Technology Guidance Project, and MOE Key Laboratory of Coal Environmental Pathogenicity and Prevention.
Acknowledgments
We thank Guangdong Provincial Occupational Disease Prevention and Treatment Hospital’s Key Laboratory Medicine Specialty for technical support. Thanks to the Radiation Epidemiology Joint Laboratory of the Institute of Radiation Medicine, Chinese Academy of Medical Sciences for its support of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpubh.2025.1651676/full#supplementary-material
Abbreviations
TC, Total cholesterol; TG, Triglycerides; LDL-C, Low-density lipoprotein cholesterol; IHD, Ischemic heart disease; BMI, The body mass index; CHAP, The China High-resolution Air Pollution dataset; HDL-C, High-density lipoprotein cholesterol; VIF, The variance inflation factor; IR, Ionizing radiation.
References
1. Pan, L, Yang, Z, Wu, Y, Yin, RX, Liao, Y, Wang, J, et al. The prevalence, awareness, treatment and control of dyslipidemia among adults in China. Atherosclerosis. (2016) 248:2–9. doi: 10.1016/j.atherosclerosis.2016.02.006
2. Hedayatnia, M, Asadi, Z, Zare-Feyzabadi, R, Yaghooti-Khorasani, M, Ghazizadeh, H, Ghaffarian-Zirak, R, et al. Dyslipidemia and cardiovascular disease risk among the MASHAD study population. Lipids Health Dis. (2020) 19:42. doi: 10.1186/s12944-020-01204-y
3. Addisu, B, Bekele, S, Wube, TB, Hirigo, AT, and Cheneke, W. Dyslipidemia and its associated factors among adult cardiac patients at ambo university referral hospital, Oromia region, West Ethiopia. BMC Cardiovasc Disord. (2023) 23:321. doi: 10.1186/s12872-023-03348-y
4. Werner, E, Alter, A, Deng, Q, Dammer, EB, Wang, Y, Yu, DS, et al. Ionizing radiation induction of cholesterol biosynthesis in lung tissue. Sci Rep. (2019) 9:12546. doi: 10.1038/s41598-019-48972-x
5. Chukwuemeka, N, Philippe, M, Magdalene, N, and Onyezuligbo, O. Effects of total body irradiation on fatty acid and total lipid content of rats. Pak J Pharm Sci. (2012) 25:169–73. Available at: https://pubmed.ncbi.nlm.nih.gov/22186326 (Accessed May 19, 2014).
6. Rosenson, RS, and Cannon, CP. (2021). Patient education: high cholesterol and lipid treatment options (beyond the basics). Available online at: https://savannahendocrinology.com/wp-content/uploads/2024/05/High-cholesterol-and-lipids.pdf (Accessed February 16, 2021).
7. Wen, C, Liu, X, Lian, Y, Guo, W, Zhang, L, Chen, Y, et al. Analysis of the association between long-term exposure to low-dose ionizing radiation and dyslipidemia and its components in medical radiologists: the mediating role of inflammatory markers. Int J Cardiol Cardiovasc Risk Prev. (2025) 25:200406. doi: 10.1016/j.ijcrp.2025.200406
8. Oslina, D, Rybkina, V, Adamova, G, Zhuntova, G, Bannikova, M, and Azizova, T. Biomarkers of atherosclerotic vascular disease in workers chronically exposed to ionizing radiation. Health Phys. (2021) 121:92–101. doi: 10.1097/hp.0000000000001416
9. Wong, Y-S, Cheng, Y-Y, Cheng, T-J, Huang, C-C, Yeh, J-J, and Guo, H-R. The relationship between occupational exposure to low-dose ionizing radiation and changes in thyroid hormones in hospital workers. Epidemiology. (2019) 30:S32–8. doi: 10.1097/ede.0000000000001004
10. ICRP. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Ann ICRP. (2007) 37:1–332. doi: 10.1016/j.icrp.2007.10.003
11. Stewart, FA, Akleyev, AV, Hauer-Jensen, M, Hendry, JH, Kleiman, NJ, Macvittie, TJ, et al. ICRP publication 118: ICRP statement on tissue reactions and early and late effects of radiation in normal tissues and organs--threshold doses for tissue reactions in a radiation protection context. Ann ICRP. (2012) 41:1–322. doi: 10.1016/j.icrp.2012.02.001
12. Boice, J Jr, Dauer, LT, Kase, KR, Mettler, FA Jr, and Vetter, RJ. Evolution of radiation protection for medical workers. Br J Radiol. (2020) 93:20200282. doi: 10.1259/bjr.20200282
13. Baudin, C, Vacquier, B, Thin, G, Chenene, L, Guersen, J, Partarrieu, I, et al. Occupational exposure to ionizing radiation in medical staff: trends during the 2009-2019 period in a multicentric study. Eur Radiol. (2023) 33:5675–84. doi: 10.1007/s00330-023-09541-z
14. Tang, FR. Health effect of low-dose-rate irradiation with cumulative threshold dose: a promising area to explore in nuclear emergency and environmental contamination. Cells. (2024) 13:1521. doi: 10.3390/cells13181521
15. Vaiserman, A, Koliada, A, Zabuga, O, and Socol, Y. Health impacts of low-dose ionizing radiation: current scientific debates and regulatory issues. Dose Response. (2018) 16:1559325818796331. doi: 10.1177/1559325818796331
16. Dimitrova, T, Hristova, E, and Petrova, N. Low-dose ionizing radiation exposure on human male gametes: damage or benefit. Life. (2024) 14:830. doi: 10.3390/life14070830
17. Frangione, B, Hinton, P, and Villeneuve, PJ. Low-dose ionizing radiation and adverse birth outcomes: a systematic review and meta-analysis. Int Arch Occup Environ Health. (2023) 96:77–92. doi: 10.1007/s00420-022-01911-2
18. Zhang, Y, Shi, J, Ma, Y, Yu, N, Zheng, P, Chen, Z, et al. Association between air pollution and lipid profiles. Toxics. (2023) 11:894. doi: 10.3390/toxics11110894
19. Wei, J, Li, Z, Wang, J, Li, C, Gupta, P, and Cribb, M. Ground-level gaseous pollutants (NO 2, SO2, and CO) in China: daily seamless mapping and spatiotemporal variations. Atmos Chem Phys. (2023) 23:1511–32. doi: 10.5194/acp-23-1511-2023
20. Jiang, H, Zhang, S, Yao, X, Meng, L, Lin, Y, Guo, F, et al. Does physical activity attenuate the association between ambient PM (2.5) and physical function? Sci Total Environ. (2023) 874:162501. doi: 10.1016/j.scitotenv.2023.162501
21. Liu, C, and Qiao, Y. The association between long-term exposure to ambient PM (2.5) and high-density lipoprotein cholesterol level among chinese middle-aged and older adults. BMC Cardiovasc Disord. (2024) 24:173. doi: 10.1186/s12872-024-03835-w
22. Zhao, SP, Lu, GP, Zhao, D, and Li, JJ. 2016 Chinese guidelines for the management of dyslipidemia in adults. J Geriatr Cardiol. (2018) 15:1–29. doi: 10.11909/j.issn.1671-5411.2018.01.011
23. Amorim, NML, Kee, A, Coster, ACF, Lucas, C, Bould, S, Daniel, S, et al. Irradiation impairs mitochondrial function and skeletal muscle oxidative capacity: significance for metabolic complications in cancer survivors. Metabolism. (2020) 103:154025. doi: 10.1016/j.metabol.2019.154025
24. Daniel, S, Nylander, V, Ingerslev, LR, Zhong, L, Fabre, O, Clifford, B, et al. T cell epigenetic remodeling and accelerated epigenetic aging are linked to long-term immune alterations in childhood cancer survivors. Clin Epigenetics. (2018) 10:138. doi: 10.1186/s13148-018-0561-5
25. Nylander, V, Ingerslev, LR, Andersen, E, Fabre, O, Garde, C, Rasmussen, M, et al. Ionizing radiation potentiates high-fat diet-induced insulin resistance and reprograms skeletal muscle and adipose progenitor cells. Diabetes. (2016) 65:3573–84. doi: 10.2337/db16-0364
26. Cheng, YJ, Nie, XY, Ji, CC, Lin, XX, Liu, LJ, Chen, XM, et al. Long-term cardiovascular risk after radiotherapy in women with breast Cancer. J Am Heart Assoc. (2017) 6:5633. doi: 10.1161/jaha.117.005633
27. Narendran, N, Luzhna, L, and Kovalchuk, O. Sex difference of radiation response in occupational and accidental exposure. Front Genet. (2019) 10:260. doi: 10.3389/fgene.2019.00260
28. Tong, J, and Hei, TK. Aging and age-related health effects of ionizing radiation. Radiat Med Prot. (2020) 1:15–23. doi: 10.1016/j.radmp.2020.01.005
29. Sun, Q, Mao, W, Jiang, H, Zhang, X, Xiao, J, and Lian, Y. The effect of protracted exposure to radiation on liver injury: a cohort study of industrial radiographers in Xinjiang, China. Int J Environ Res Public Health. (2018) 15:71. doi: 10.3390/ijerph15010071
30. Cornacchia, S, La Tegola, L, Maldera, A, Pierpaoli, E, Tupputi, U, Ricatti, G, et al. Radiation protection in non-ionizing and ionizing body composition assessment procedures. Quant Imaging Med Surg. (2020) 10:1723–38. doi: 10.21037/qims-19-1035
31. Lee, S, Kim, KW, Kwon, HJ, Lee, J, Koo, K, Song, GW, et al. Relationship of body mass index and abdominal fat with radiation dose received during preoperative liver CT in potential living liver donors: a cross-sectional study. Quant Imaging Med Surg. (2022) 12:2206–12. doi: 10.21037/qims-21-977
32. Aragón-Herrera, A, Moraña-Fernández, S, Otero-Santiago, M, Anido-Varela, L, Campos-Toimil, M, García-Seara, J, et al. The lipidomic and inflammatory profiles of visceral and subcutaneous adipose tissues are distinctly regulated by the SGLT2 inhibitor empagliflozin in Zucker diabetic fatty rats. Biomed Pharmacother. (2023) 161:114535. doi: 10.1016/j.biopha.2023.114535
33. Neeland, IJ, Ross, R, Després, JP, Matsuzawa, Y, Yamashita, S, Shai, I, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes Endocrinol. (2019) 7:715–25. doi: 10.1016/s2213-8587(19)30084-1
34. Von Krüchten, R, Lorbeer, R, Müller-Peltzer, K, Rospleszcz, S, Storz, C, Askani, E, et al. Association between adipose tissue depots and dyslipidemia: the KORA-MRI population-based study. Nutrients. (2022) 14:797. doi: 10.3390/nu14040797
35. Chartier, H, Fassier, P, Leuraud, K, Jacob, S, Baudin, C, Laurier, D, et al. Occupational low-dose irradiation and cancer risk among medical radiation workers. Occup Med (Lond). (2020) 70:476–84. doi: 10.1093/occmed/kqaa130
36. Yuan, ZH, Liu, T, Wang, H, Xue, LX, and Wang, JJ. Fatty acids metabolism: the bridge between Ferroptosis and ionizing radiation. Front Cell Dev Biol. (2021) 9:675617. doi: 10.3389/fcell.2021.675617
37. Einor, D, Bonisoli-Alquati, A, Costantini, D, Mousseau, TA, and Møller, AP. Ionizing radiation, antioxidant response and oxidative damage: a meta-analysis. Sci Total Environ. (2016) 548-549:463–71. doi: 10.1016/j.scitotenv.2016.01.027
38. Xiao, Y, Mo, W, Jia, H, Yu, D, Qiu, Y, Jiao, Y, et al. Ionizing radiation induces cutaneous lipid remolding and skin adipocytes confer protection against radiation-induced skin injury. J Dermatol Sci. (2020) 97:152–60. doi: 10.1016/j.jdermsci.2020.01.009
39. Yoshida, T, Goto, S, Kawakatsu, M, Urata, Y, and Li, TS. Mitochondrial dysfunction, a probable cause of persistent oxidative stress after exposure to ionizing radiation. Free Radic Res. (2012) 46:147–53. doi: 10.3109/10715762.2011.645207
40. Laiakis, EC, Strassburg, K, Bogumil, R, Lai, S, Vreeken, RJ, Hankemeier, T, et al. Metabolic phenotyping reveals a lipid mediator response to ionizing radiation. J Proteome Res. (2014) 13:4143–54. doi: 10.1021/pr5005295
Keywords: ionizing radiation, radiation worker, dyslipidemia, lipid biomarkers, mixed-effect model
Citation: Zhong M, Wang X, Li Q, Wu Z, Huang X, Li X, Lian Y, Peng Y, Li Z, Liu Z, Wang Q, Gong Y, Hu P, Liu X, Zhu S, Wu S, Zeng F, Qin Y, Nong Q, Wang J, Gu Y, Huang Y, Zhang W and Zhao N (2025) Association between occupational ionizing radiation exposure duration and the increased risk of dyslipidemia: evidence from a large group of radiation workers. Front. Public Health. 13:1651676. doi: 10.3389/fpubh.2025.1651676
Edited by:
Yi Xie, Chinese Academy of Sciences (CAS), ChinaReviewed by:
Maurizio Tomasi, New Mexico State University, United StatesJianxiang Liu, Chinese Center for Disease Control and Prevention, China
Copyright © 2025 Zhong, Wang, Li, Wu, Huang, Li, Lian, Peng, Li, Liu, Wang, Gong, Hu, Liu, Zhu, Wu, Zeng, Qin, Nong, Wang, Gu, Huang, Zhang and Zhao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wangjian Zhang, emhhbmd3ajIyN0BtYWlsLnN5c3UuZWR1LmNu; Na Zhao, emhhb25hQGdkcGNjLmNvbQ==
†These authors have contributed equally to this work
 Meimei Zhong1,2†
Meimei Zhong1,2† Xiaowen Wang
Xiaowen Wang Yiru Qin
Yiru Qin Jinhan Wang
Jinhan Wang Yeqing Gu
Yeqing Gu Yongshun Huang
Yongshun Huang Wangjian Zhang
Wangjian Zhang Na Zhao
Na Zhao