- College of Natural and Health Sciences, Zayed University, Dubai, United Arab Emirates
Background: The use of perfumes and cosmetic products is widespread, serving personal hygiene, aesthetic, and olfactory functions. However, concerns have been raised regarding the potential health impacts associated with long-term exposure to various ingredients used in these products.
Objectives: This narrative review aims to synthesize evidence on the health risks associated with perfumes and cosmetics, focusing on specific health concerns, including fertility, respiratory health, cancer risk, allergies, skin disorders, endocrine disruption, and neurological effects. It also discusses the presence of heavy metals in cosmetics, regulatory challenges, and the need for transparency in ingredient disclosure.
Methods: A comprehensive review of literature published between 2005 and 2025 was conducted, examining findings from interdisciplinary studies relevant to the health impacts of cosmetic and fragrance products. The review highlights health concerns linked to specific chemical components, including synthetic chemicals such as phthalates, parabens, and volatile organic compounds (VOCs).
Results: The findings indicate that many synthetic chemicals in perfumes and cosmetics are associated with adverse health outcomes. These include allergies, respiratory issues, endocrine disruption, reproductive problems, and potentially cancer. Heavy metals in cosmetics also pose significant health risks. Despite regulatory guidelines, the cumulative and long-term effects of combined exposure to multiple cosmetic ingredients remain poorly understood and inadequately addressed.
Conclusion: There is a pressing need for stricter regulatory oversight and improved transparency in ingredient disclosure to safeguard consumer health. Further research is required to clarify the long-term health risks associated with the daily use of cosmetic products and to develop safer alternatives.
1 Introduction
Perfumes and cosmetic products have become an integral part of daily life, transcending cultural and demographic boundaries (Park and Hong, 2024a). These products, which include an extensive array of items such as perfumes, deodorants, lotions, shampoos, soaps, and makeup, are valued not only for their practical purposes—such as personal hygiene and skin protection—but also for their capacity to enhance aesthetic appeal and provide sensory pleasure (Srinivasulu et al., 2022). With a history dating back to ancient civilizations, where natural extracts and oils were used for beautification and ritualistic purposes, the use of these products has evolved dramatically. Today, the cosmetics industry stands as a global powerhouse, worth billions of dollars and driven by relentless innovation and consumer demand (McMullen and Dell’Acqua, 2023).
The modern cosmetics industry is characterized by the use of sophisticated formulations that combine natural and synthetic ingredients. These innovations aim to achieve properties such as long-lasting fragrance, enhanced texture, vibrant color, and improved shelf life (Rico et al., 2024). While these advancements have undoubtedly elevated consumer experiences, they have also brought about a host of concerns regarding the potential health impacts of prolonged exposure to the chemical components of these products. Ingredients such as phthalates, parabens, volatile organic compounds (VOCs), and heavy metals are commonly used to achieve desired cosmetic effects, but growing evidence suggests that they may pose significant health risks.
Research in recent decades has linked certain cosmetic ingredients to a range of adverse health outcomes, including skin irritation, respiratory issues, allergic reactions, endocrine disruption, reproductive problems, and even an elevated risk of certain cancers. For instance, phthalates and parabens, often used as preservatives and stabilizers, have been associated with hormone mimicry, which can interfere with endocrine functions (Ahmed et al., 2024). Similarly, VOCs—emitted as gases from fragrances and aerosol products—have been implicated in respiratory disorders and neurological effects. The presence of heavy metals such as lead, cadmium, and mercury in certain cosmetics further exacerbates health concerns, as these elements are known to accumulate in the body and exert toxic effects over time (Witkowska et al., 2021).
Despite the potential health implications, the regulatory landscape governing cosmetics and personal care products remains fragmented and, in some cases, insufficiently stringent. While agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have implemented guidelines to ensure product safety, these regulations often fall short in addressing the cumulative and long-term effects of repeated exposure to multiple chemical ingredients (Bopp et al., 2018). Additionally, the lack of transparency in ingredient disclosure—with many formulations protected under the guise of trade secrets—complicates efforts to assess and mitigate risks effectively. This regulatory gap has sparked growing public concern and calls for stricter oversight to safeguard consumer health (Iqbal and Biller-Andorno, 2022). The average consumer is exposed to a multitude of personal care products daily, ranging from toothpaste and shampoo in the morning to moisturizers and perfumes throughout the day. This habitual use results in compounded exposure to a cocktail of chemicals, the interactions and combined effects of which remain poorly understood (Elcombe et al., 2022). Furthermore, vulnerable populations such as pregnant women, children, and individuals with pre-existing health conditions may be at heightened risk, underscoring the need for targeted research and tailored safety recommendations.
Existing reviews on the safety of perfumes and cosmetics have primarily focused on specific aspects, such as dermatological effects or toxicological impacts, often leaving critical gaps in the broader understanding of their implications for human health (Manful et al., 2024). This narrative review seeks to bridge these gaps by synthesizing evidence from diverse disciplines, including dermatology, toxicology, endocrinology, and environmental health (Nunes et al., 2024). By doing so, it aims to provide a holistic perspective on the health risks associated with the use of cosmetic products and fragrances (Alnuqaydan, 2024).
Specifically, this review will explore the impact of cosmetic ingredients on fertility, respiratory health, cancer risk, allergies, skin disorders, endocrine function, neurological well-being, and the presence of heavy metals. It will also examine emerging evidence on potential risks, offering insights into areas requiring further investigation. Ultimately, this review underscores the urgent need for more comprehensive regulatory frameworks, greater transparency in ingredient disclosure, and heightened consumer awareness to address the multifaceted health challenges posed by the widespread use of cosmetic and fragrance products.
Historically, discussions surrounding the safety of cosmetics have primarily focused on well-documented health issues, such as their effects on fertility, respiratory health, cancer risks, and allergic reactions (Tong et al., 2023). However, a growing body of research has begun to uncover a broader spectrum of potential health implications that extend beyond these commonly examined areas. This expanding field of inquiry emphasizes the need to explore other conditions that may arise due to the complex interactions between cosmetic ingredients and the human body (Alnuqaydan, 2024).
Among these emerging concerns are skin disorders, such as dermatitis, eczema, and allergic reactions, which affect a significant number of individuals following the use of certain cosmetic products. Additionally, there is increasing evidence that specific chemicals in cosmetics may disrupt endocrine function, potentially interfering with hormonal regulation and leading to adverse effects on reproductive and metabolic systems. Furthermore, some studies suggest a link between exposure to fragrances and neurological outcomes, including headaches, mood disturbances, and other cognitive effects (Li et al., 2024).
As the scope of research widens, it becomes essential to consider these additional conditions to gain a more comprehensive understanding of the potential health risks associated with cosmetics (Pistollato et al., 2021). This exploration underscores the importance of rigorous testing, informed consumer choices, and regulatory measures to ensure the safe use of these everyday products. The following sections delve into these emerging concerns in greater detail, highlighting the need for heightened awareness and ongoing scientific investigation (Pastor-Nieto and Gatica-Ortega, 2021). The main objectives are to assess the impacts of perfumes and cosmetics on fertility, lung health, cancer risk, allergies, and other conditions such as skin disorders, endocrine disruption, and neurological effects, and the effects of heavy metals.
2 Methodology
The methodology for this narrative review on the health impacts of perfumes and cosmetic products encompasses a literature collection based on inclusion and exclusion criteria, and qualitative synthesis of peer-reviewed studies. The goal is to consolidate findings that examine potential health risks associated with cosmetic and fragrance use, particularly focusing on fertility, lung health, cancer risk, allergies, skin disorders, endocrine disruption, neurological effects, and effects of heavy metals. To comprehensively analyze the health impacts of perfumes and cosmetic products, peer-reviewed studies were sourced from databases including PubMed, Google Scholar, and ResearchGate. Keywords such as “perfume toxicity,” “cosmetic ingredients health effects,” “fragrance VOCs,” “allergies and cosmetics,” “endocrine disruption cosmetics,” and “cancer risk cosmetics” were employed to ensure a wide scope of relevant literature. All articles used were peer-reviewed and sourced from reputable journals to ensure scientific rigor. Studies that were published in the years between 2005 and 2025 were included to provide a well rounded and comprehensive perspective on the topic.
2.1 Inclusion criteria
• Studies that examine the effects of perfumes and cosmetics on human health.
• Both observational and experimental research were included.
• Focus on fertility, lung health, cancer risk, allergies, skin disorders, endocrine disruption, neurological effects, and the effects of heavy metals in cosmetics.
2.2 Exclusion criteria
• Non-peer-reviewed articles.
• Opinion pieces and anecdotal evidence.
• Studies with limited relevance or non-generalizable results (e.g., small sample sizes).
3 Results and discussion
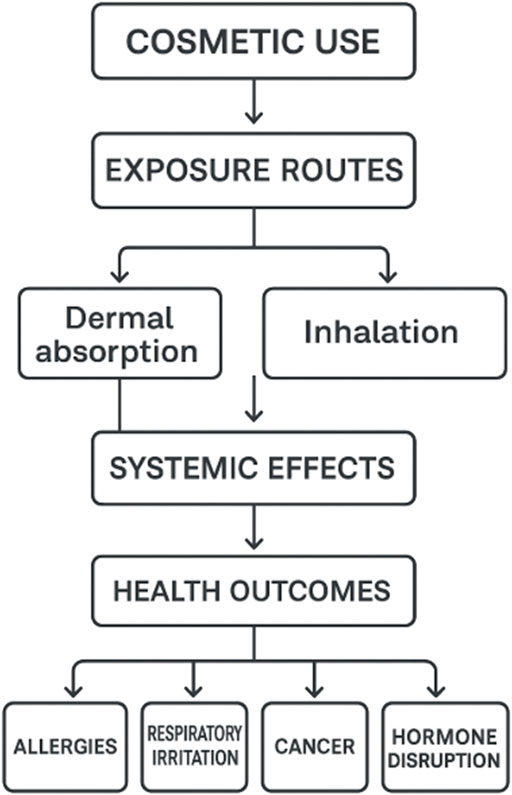
Figure 1 illustrates the pathway through which cosmetic use can lead to adverse health outcomes, emphasizing the mechanisms of exposure and subsequent systemic effects. It begins with the application of cosmetic products, which introduces potential contaminants into the body via two primary exposure routes: dermal absorption and inhalation. Dermal absorption occurs when substances penetrate the skin, while inhalation involves breathing in volatile compounds, sprays, or powders. Both routes can result in systemic distribution of these chemicals throughout the body, potentially disrupting normal physiological functions. This systemic exposure may lead to various health outcomes, including allergic reactions, respiratory irritation, cancer, and hormonal disruption. The diagram highlights the importance of understanding not just the ingredients in cosmetic products but also how they interact with the body over time, particularly through repeated use, which can amplify cumulative exposure and health risks.
The use of cosmetics, while an integral component of personal care and beauty practices, introduces a multifaceted array of potential health risks stemming from their chemical compositions (Abiodun and Ayeleru, 2025). This discussion explores key areas of concern, highlighting evidence from diverse studies and emphasizing the need for increased public awareness, enhanced research efforts, and stringent regulatory measures to ensure consumer safety.
3.1 Endocrine disruption
Endocrine-disrupting chemicals (EDCs) such as parabens, phthalates, bisphenols, and other synthetic compounds frequently found in cosmetics pose significant threats to human health (Thompson, 2018). These compounds, prevalent in personal care products including perfumes, lotions, nail polishes, and hair sprays, have been shown to interfere with the endocrine system, thereby disrupting hormonal homeostasis (Anderson et al., 2022). A biomonitoring study involving 144 Norwegian adults found widespread urinary detection of parabens, bisphenols, phthalates, and UV filters, with exposure levels linked to hormone-related toxicity and metabolic disruption (Husøy et al., 2019). These substances can mimic, inhibit, or interfere with endogenous hormone signaling, thereby disrupting critical regulatory pathways associated with reproduction, metabolism, and thyroid function. EDCs can interfere with hormones such as estrogen, testosterone, thyroid hormones, and insulin, provoking physiological disturbances across multiple systems. See Table 1 for health risks associated with common cosmetic ingredients and contaminants across multiple bodily systems and functions.
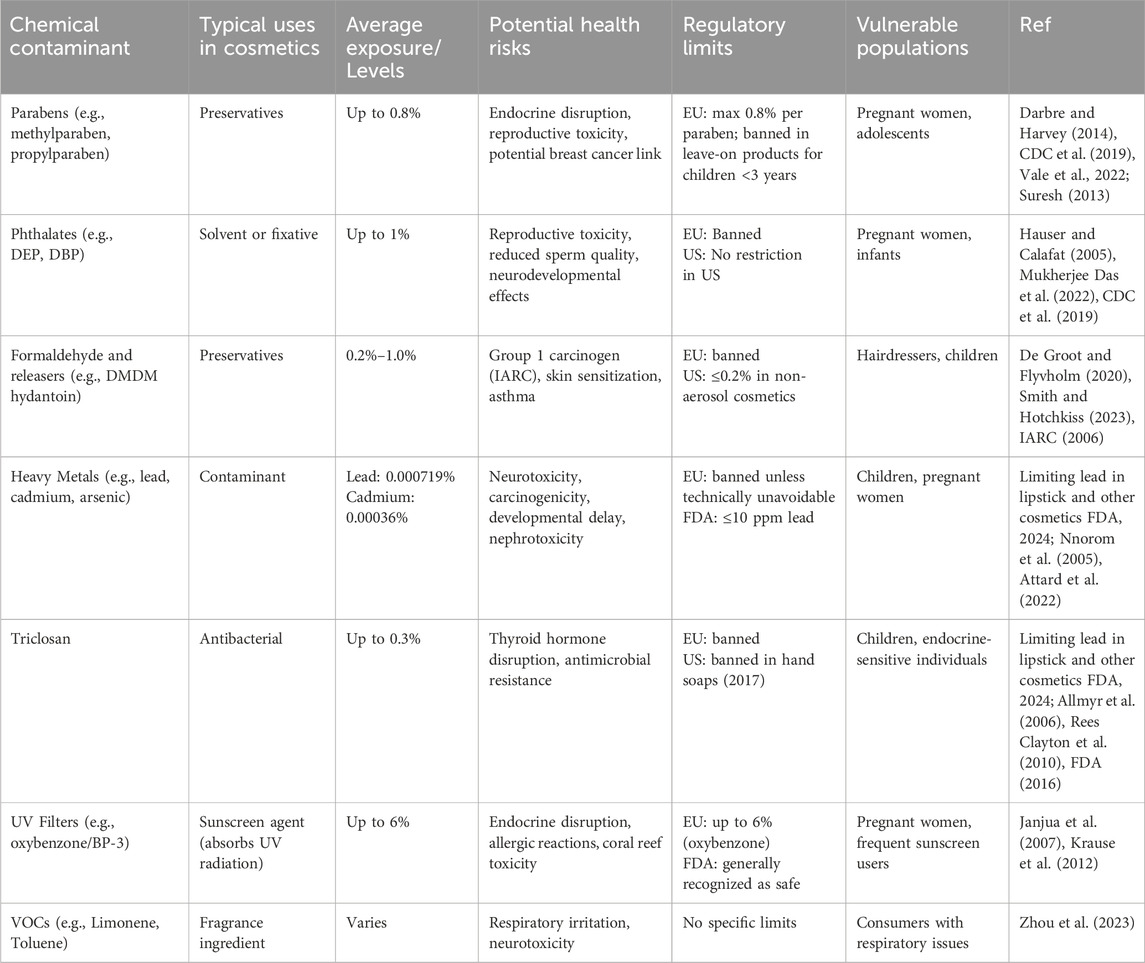
Table 1. Common cosmetic contaminants: uses, exposure, health risks, regulations, and vulnerable populations.
Additionally, a substantial body of research has correlated EDC exposure with conditions such as obesity, diabetes, polycystic ovarian syndrome (PCOS), and infertility. Phthalates and bisphenols are particularly implicated in obesity through their impact on adipocyte differentiation, metabolic rate, and appetite regulation (Hauser and Calafat, 2005). These chemicals contribute to increased lipid accumulation and impaired appetite signaling, facilitating excessive weight gain. Phthalates have also been shown to impair insulin sensitivity and alter glucose metabolism, contributing to the pathogenesis of type 2 diabetes. Critically, the timing of exposure plays a pivotal role in determining health outcomes. Prenatal and early childhood periods represent windows of heightened vulnerability, during which EDCs can induce lasting developmental and physiological alterations. For example, bisphenol A (BPA), found in plastic packaging and some cosmetic containers, can cross the placental barrier and disrupt fetal endocrine function (Limiting lead in lipstick and other cosmetics FDA, 2024). Emerging evidence indicates that the effects of EDCs may extend across generations through epigenetic mechanisms. Maternal exposure to EDCs can induce modifications in gene expression that are heritable, even in the absence of continued exposure. These transgenerational effects may increase the risk of metabolic dysfunction, endocrine disorders, and reproductive abnormalities in descendants (Nnorom et al., 2005).
Given their widespread presence in personal care products, urgent regulatory action is warranted. While some jurisdictions have initiated restrictions on EDCs in cosmetics, enforcement remains fragmented, and many formulations still contain potentially hazardous chemicals (Allmyr et al., 2006). Regulatory frameworks must prioritize the establishment of concentration limits, pre-market safety assessments, and transparent labeling. Policies encouraging safer, non-toxic alternatives can help protect public health and reduce long-term exposure risks. Consumer engagement is also essential in mitigating EDC exposure. By selecting products free of parabens, phthalates, and bisphenols, individuals can reduce personal risk. Supporting brands that prioritize safety, and transparency encourages broader industry reform. The rising demand for clean, non-toxic cosmetics is already catalyzing a shift toward safer formulations and more sustainable manufacturing practices (Rees Clayton et al., 2010; Janjua et al., 2007). In summary, the endocrine-disrupting potential of numerous cosmetic ingredients necessitates comprehensive action from regulatory bodies, industry stakeholders, and consumers. A coordinated approach involving regulatory reform, scientific innovation, and public awareness is essential to reduce exposure and safeguard hormonal health across the lifespan.
3.2 Fertility
As discussed in Section 3.1, endocrine-disrupting chemicals such as phthalates and parabens impair hormonal regulation, leading to fertility issues. In females, these compounds can compromise ovarian function, oocyte quality, and hormonal signaling pathways necessary for conception and gestation. In males, EDCs can impair spermatogenesis, reduce sperm motility, and lower sperm counts, collectively undermining fertility. Additionally, exposure to these chemicals has been associated with early-onset puberty in both sexes, which may disrupt growth trajectories, sexual maturation, and long-term reproductive outcomes (De Groot and Flyvholm, 2020).
Mechanistically, phthalates disrupt the hypothalamic-pituitary-gonadal (HPG) axis by acting as exogenous ligands that can mimic, antagonize, or modulate the activity of endogenous hormones (Di Cicco et al., 2021). Disruption of the HPG axis alters the synthesis and secretion of key reproductive hormones, leading to documented fertility impairments (Gould et al., 2024). Epidemiological studies have linked phthalate exposure to reduced semen quality in men and menstrual disturbances in women (Elsaid and Ahmed, 2021). Importantly, their reproductive toxicity extends to developmental windows of heightened vulnerability, particularly during gestation. Prenatal exposure has been linked to adverse birth outcomes, such as cryptorchidism in male infants, suggesting interference with utero-androgen signaling pathways (Khalid and Abdollahi, 2021). Similarly, Octyl Methoxycinnamate (OMC), a UV filter commonly used in sunscreens and cosmetics, has been shown to affect fetal development. In an in vivo animal study, pregnant Wistar rats exposed to OMC had significantly reduced maternal thyroxine (T4) levels, and their male offspring displayed decreased testosterone levels, lower sperm counts, and reduced weights of reproductive organs such as the prostate and testes (Axelstad et al., 2011). These effects persisted into adulthood, though the study’s applicability is limited by the lack of human data. Furthermore, disruptions in the timing of pubertal onset, either precocious or delayed puberty, have been documented in populations with elevated early-life phthalate exposure, indicating that these chemicals may perturb developmental endocrine pathways with long-term consequences for reproductive health (Lazou, 2024).
Parabens, another class of EDCs, exert estrogenic effects by binding to estrogen receptors and mimicking the activity of endogenous estrogens (Akano et al., 2024). By exhibiting estrogenic activity, they can overstimulate estrogen receptors and disrupt endocrine homeostasis (Darbre and Harvey, 2014). This interaction has raised concerns about their potential role in impairing female fertility. In vitro studies show that butylparaben and propylparaben strongly mimicked estrogen, activating both ERα and ERβ, while ethylparaben had weaker effects, and methylparaben was inactive (Gomez et al., 2005). UV filters such as OMC, 4-Methylbenzylidene camphor (4-MBC), Octyl-Dimethyl PABA (OD-PABA), and Homosalate (HMS) selectively activated ERα, indicating a risk of tissue-specific endocrine disruption. Musk ketone and galaxolide weakly activated ERα; however, they raise concern as they persist in the environment and bioaccumulate. The study used a validated receptor-specific assay and benchmarking against 17β-estradiol, the primary endogenous estrogen. While this approach characterizes receptor binding and activation potential, it does not account for the metabolic transformation of these compounds within the human body, limiting its applicability to real-world exposure scenarios.
Addressing this gap, a single-blinded human exposure study involving thirty-two healthy adults (15 young men and 17 postmenopausal women) exposed participants to sunscreen formulations containing benzophenone-3 (BP-3), OMC, and 4-MBC (Janjua et al., 2004). Despite systemic absorption confirmed by detectable levels of these UV filters in both blood and urine samples, the study found no biologically significant alterations in reproductive hormones, including FSH, LH, estradiol, testosterone, and inhibin B. The study was limited in its short exposure period, relatively small sample size, and lack of long-term follow-up. It also did not evaluate cumulative exposure effects or possible delayed endocrine outcomes. Nonetheless, the detection of UV-filters in the blood raises concern, and safety thresholds should account for systemic bioavailability of these ingredients during repeated daily use.
Inverse associations have been observed between urinary paraben concentrations and ovarian reserve markers, including antral follicle count and anti-Müllerian hormone levels (Alnuqaydan, 2024). These suggest that parabens may impair reproductive potential by compromising oocyte quality, with implications for fertility and assisted reproductive outcomes. Genetic susceptibility may also mediate individual responses to EDCs. A biomonitoring study that looked at 195 healthy young men in Denmark, with and without filaggrin (FLG) gene mutations (a gene that affects the skin’s natural barrier function) found that in men with FLG mutations, higher urinary concentrations of bisphenol A (BPA) and UV filters such as BP-1, BP-3, and 4-HBP were associated with increased serum testosterone and estradiol levels, decreased follicle-stimulating hormone (FSH), and reduced sperm motility (Joensen et al., 2018). These effects were not observed in non-carriers, showing that endocrine response could be subject to the role of genetic predisposition. No significant associations were found between urinary paraben levels and hormone levels or semen health in either group. These findings underscore the importance of personalized risk assessment based on genetic susceptibility.
The reproductive risks associated with cosmetic EDCs are compounded by widespread and repeated exposure. Daily application of multiple personal care products results in cumulative exposure, contributing to systemic bioaccumulation over time (Marusic Kristina, 2023). This risk is especially concerning for susceptible subgroups, such as pregnant individuals, adolescents undergoing pubertal development, and those with pre-existing endocrine disorders, who may exhibit heightened biological sensitivity to these compounds. Given the persistence and biological activity of EDCs in cosmetics, a precautionary approach is warranted. Regulatory frameworks should enforce rigorous safety assessments of cosmetic ingredients, mandate clear labeling of EDC content, and incentivize the development of non-disruptive alternatives. Public health campaigns must also play a role in disseminating evidence-based guidance to help consumers make informed choices about personal care product use (Mukherjee Das et al., 2022). In summary, converging evidence from mechanistic, epidemiological, and developmental studies supports a causal link between chronic exposure to phthalates, parabens, and other EDCs in cosmetics and adverse reproductive health outcomes (Hager et al., 2022). These findings underscore the need for proactive policy interventions and consumer education to mitigate risks and safeguard reproductive potential across the lifespan and for future generations (Beyuo et al., 2024).
3.3 Lung health and asthma
The respiratory consequences of cosmetic product use, particularly those containing synthetic fragrances and aerosolized components, have emerged as a critical area of public health concern. Numerous products, such as perfumes, deodorants, hair sprays, and air fresheners, emit Volatile Organic Compounds (VOCs), which are recognized contributors to both indoor and outdoor air pollution (Srinivasulu et al., 2022). These VOCs not only act as primary airborne irritants but also undergo chemical reactions with other indoor pollutants, especially nitrogen oxides, to form secondary toxicants, including ozone and ultrafine particulate matter. This dynamic significantly elevates respiratory health risks, particularly in enclosed or poorly ventilated environments where pollutant concentrations can accumulate rapidly (Srinivasulu et al., 2022).
VOCs are a chemically diverse group of compounds frequently present in scented cosmetic products. They include terpenes (e.g., limonene), alcohols, aldehydes, and esters, many of which have known irritant or sensitizing properties (Dhiman et al., 2024). Inhalation of these compounds can provoke acute respiratory symptoms such as coughing, wheezing, and dyspnea by irritating the mucosal linings of the upper and lower respiratory tract (Singh et al., 2023). Of particular concern is their role in generating ground-level ozone when they interact with other ambient pollutants. Ozone is a well-established pulmonary irritant associated with airway inflammation, reduced lung function, and increased hospital admissions for respiratory conditions (Bird et al., 2021). Chronic exposure to elevated VOC and ozone levels has been implicated in the development and exacerbation of asthma, particularly among sensitive subpopulations.
Persistent exposure to aerosolized cosmetics, including spray deodorants, dry shampoos, and setting sprays is associated with structural changes in lung tissue, impaired gas exchange, and a heightened risk of respiratory infections. Vulnerable groups such as children whose lungs are still maturing and elderly individuals with pre-existing conditions like asthma or COPD are especially susceptible to these effects, with exposure linked to increased frequency and severity of exacerbations, hospital visits, and longterm morbidity (Attard et al., 2022; Ullah et al., 2023).
The implications of cosmetic-related respiratory distress extend beyond personal health, impacting healthcare systems and occupational environments. Increased incidence of asthma and other chronic respiratory diseases due to VOC and particulate exposure translates into greater demand for medical care, productivity losses, and elevated healthcare expenditures (Raza-Naqvi et al., 2022). Occupational exposure in high-risk settings, such as beauty salons, spas, and cosmetic manufacturing facilities, is particularly concerning due to the intensity and duration of exposure. Workers in these environments often experience prolonged inhalation of concentrated chemical mixtures, frequently without adequate personal protective equipment or ventilation systems, placing them at risk for chronic respiratory conditions and occupational asthma (Manful et al., 2024).
3.4 Cancers
Cosmetic products, while widely used for enhancing appearance and hygiene, can also expose users to harmful substances linked to carcinogenesis (Scopelliti et al., 2021). Mounting scientific evidence implicates several common cosmetic ingredients in cancer development, underscoring the need for stricter regulation, safer formulations, and informed consumer choices. Understanding these associations is crucial to public health protection (Lohmann, 2024).
3.4.1 Hormone-mimicking chemicals: parabens and phthalates
Given their endocrine-disrupting properties (see Section 3.1), parabens and phthalates can also stimulate cell proliferation in estrogen-sensitive tissues, raising concern for hormone-related cancers such as breast and ovarian cancer (Lazzarini et al., 2018). Notably, even low-dose, chronic exposure to these chemicals has been associated with biological effects due to their capacity for bioaccumulation (Groot, 2021). Widespread use in items like deodorants, moisturizers, nail polish, and hair products ensures near-continuous human contact (Aparicio-Soto et al., 2022). In the absence of enforceable limits or mandatory disclosure, consumers are often unaware of their exposure. Implementing precautionary regulations and promoting the development of endocrine-safe alternatives is imperative to reduce carcinogenic risks.
3.4.2 Talc and asbestos contamination
Talc, valued in cosmetics for its texture-enhancing and moisture-absorbing properties, has drawn concern due to potential asbestos contamination, a result of talc and asbestos often co-occurring geologically (Mawhirt and Fonacier, 2022). Asbestos fibers, when inhaled or used in the genital area, have been strongly associated with mesothelioma and ovarian cancer, respectively (Lejding, 2024).
Although regulatory agencies require asbestos-free talc, inconsistencies in enforcement and testing practices have left gaps in consumer protection (Tammaro et al., 2012). In particular, some regions lack mandatory, standardized testing for asbestos contamination. Strengthening quality assurance procedures and ensuring transparent ingredient sourcing are critical steps to safeguard public health. Safer talc alternatives and clear labeling would further mitigate exposure risks (Fonacier et al., 2022).
3.4.3 Heavy metals
Heavy metals such as lead and arsenic, often present as unintentional contaminants in cosmetics, pose another significant carcinogenic threat (Line et al., 2023). Lead, found in some lipsticks, can accumulate in the body and is linked to DNA damage and endocrine disruption. Arsenic, detected in certain skin-lightening creams, is classified as a Group 1 carcinogen and associated with cancers of the skin, lungs, and bladder (Sicherer et al., 2022). A toxicological analysis of nine widely available fairness creams showed mercury and zinc at concentrations up to 141 mg/kg and 138 mg/kg, mercury levels exceeding international safety thresholds and being associated with elevated hazard quotients (HQ) and lifetime cancer risk (LCR) scores (Irfan et al., 2022). The products also contained lead, cadmium, nickel, and chromium, all linked to carcinogenic effects. These metals often enter cosmetic formulations through contaminated raw materials or inadequate quality control during manufacturing. Because of their ability to persist in tissues and their cumulative toxicity, even low-level, chronic exposure raises serious health concerns (Sicherer et al., 2022). To minimize risks, enhanced contaminant screening, supply chain monitoring, and regulatory oversight are urgently required. Educating the public about these hazards will further reduce unnecessary exposure.
A laboratory-based analysis of 95 cosmetic products reported that all samples contained multiple toxic metals, including arsenic, lead, cadmium, chromium, and nickel (Salles et al., 2023). Arsenic was the dominant contributor to lifetime cancer risk (LCR), especially among children exposed via ingestion (LCR up to 10−5) and adults via dermal contact (LCR up to 10−3). Further raising these concerns, laboratory-based toxicological risk assessment of 14 facial cosmetics revealed that several lipsticks and eye shadows contained lead, chromium, and mercury at concentrations far exceeding international safety limits. Lead levels reached 27.0 mg/kg in lipstick and 40.9 mg/kg in eye shadow, while chromium was as high as 149 mg/kg and mercury up to 138 mg/kg (Voica et al., 2023). These metals are associated with carcinogenicity. On the other hand, another large-scale laboratory risk assessment analyzed 200 cosmetic products from the Korean market and found that most heavy metals, including chromium, lead, cadmium, mercury, and arsenic, posed negligible cancer and systemic toxicity risks under normal use (Lim et al., 2018). Lifetime cancer risk (LCR) and hazard indices (HI) for all metals remained below safety thresholds. However, a subset of lip and eye products used by heavy consumers exceeded acceptable daily intake levels for Cr6+, Mn, and Pb, suggesting targeted concern for frequent users. These findings highlight the importance of product-specific use patterns and support routine monitoring to ensure safety across varying levels of cosmetic use.
3.4.4 Broader implications and the path forward
The recurring presence of carcinogens in cosmetics highlights fundamental gaps in global regulatory frameworks (Aleksandra and Lelieveld, 2022). Unlike drugs, many cosmetic products bypass pre-market safety evaluations. This regulatory void allows ingredients with limited toxicological data to enter the market unchecked. A harmonized global approach involving ingredient testing, safety thresholds, and post-market surveillance is needed to ensure consistent consumer protection. Public health initiatives should prioritize awareness campaigns, transparent labeling, and investment in green chemistry solutions (El Maghraby and Arafa, 2023). Collaborative regulatory efforts across borders can help align standards and enforcement mechanisms, fostering a safer cosmetics landscape. Such actions are vital not only to reduce cancer incidence but also to promote accountability and sustainability in the personal care industry (El Maghraby and Arafa, 2023).
In summary, several commonly used cosmetic ingredients, parabens, phthalates, talc, and heavy metals, pose potential carcinogenic risks that warrant immediate attention (Amutha et al., 2024). A combined effort by regulators, manufacturers, and consumers is essential to transition toward safer formulations that protect long-term health. Reinforcing trust in cosmetic safety requires science-driven policy, rigorous oversight, and a shift toward transparency and prevention (Lohmann, 2024). Mitigating these risks requires a comprehensive public health and regulatory response. Public education initiatives are essential to inform consumers about the potential hazards posed by fragranced and aerosolized cosmetic products, especially for those with existing respiratory vulnerabilities. Improved labeling standards that indicate the presence and potential effects of VOCs and particulate matter are needed to facilitate informed consumer decision-making (Alurame Eruaga, 2024). Regulatory agencies should implement stricter limits on VOC emissions in cosmetic formulations and mandate routine safety testing for inhalation risks, particularly for spray products.
Advances in formulation science also offer pathways to reduce respiratory hazards. Reformulating products to use low- or zero-VOC ingredients, transitioning to solid or water-based cosmetic formats, and increasing the availability of unscented or naturally derived fragrance options can significantly lower exposure levels (Anagnosti et al., 2021). For occupational settings, enforcing indoor air quality standards, providing appropriate ventilation systems, and ensuring access to protective gear are critical strategies for protecting worker health (Smith and Hotchkiss, 2023).
In conclusion, while cosmetics are widely used and culturally embedded, their potential to harm respiratory health especially through VOCs and fine particulate emissions warrants significant attention (Lohmann, 2024). Individuals with asthma, COPD, or other respiratory conditions, along with children and the elderly, face heightened risks. Through targeted regulation, innovation in product development, and greater public awareness, meaningful progress can be made in reducing these exposures and protecting the population’s lung health (Lohmann, 2024).
3.5 Allergies
Allergic reactions to cosmetics, particularly allergic contact dermatitis (ACD), represent a significant proportion of adverse events associated with personal care products (Lin et al., 2024). These hypersensitivity reactions are typically triggered by specific chemical constituents such as fragrances, formaldehyde-releasing preservatives, and synthetic dyes. These allergens can provoke a range of responses from localized inflammation to, in rare cases, systemic effects like urticaria and anaphylaxis (Novak-Bilić et al., 2018).
3.5.1 Pathophysiology of cosmetic-induced allergies
ACD results primarily from type IV delayed-type hypersensitivity, mediated by T lymphocytes. After initial exposure, small allergenic molecules (haptens) penetrate the skin barrier and bind to carrier proteins, forming immunogenic complexes (Khan and Alam, 2019). These are processed by Langerhans cells, which migrate to lymph nodes to activate T cells. Upon re-exposure, memory T cells initiate a robust inflammatory cascade, resulting in the clinical features of dermatitis (Maden, 2024). In contrast, irritant contact dermatitis (ICD) arises from cumulative damage to the skin barrier by repeated exposure to irritants. Although not immune-mediated, ICD can exacerbate or coexist with ACD, complicating diagnosis and treatment. Accurate differentiation is essential, as management strategies vary (Parkes, 2023).
3.5.2 Common allergenic components
• Fragrances: Among the most prevalent sensitizers, fragrance ingredients like cinnamic aldehyde, eugenol, and isoeugenol are frequent triggers of ACD. Cross-reactivity is common, particularly in individuals allergic to balsam of Peru, which shares chemical constituents with many fragrance compounds (Martini, 2022).
• Preservatives: Widely used to prevent microbial contamination, preservatives such as formaldehyde-releasing agents, parabens, and isothiazolinones pose notable allergenic risks. Formaldehyde may act as a direct sensitizer or induce cross-sensitization in predisposed individuals (Inderbinen et al., 2022).
• Synthetic Dyes: Para-phenylenediamine (PPD), a component in many hair dyes, is a potent allergen that can provoke severe localized dermatitis and, occasionally, systemic reactions. Diagnosis is typically confirmed via targeted patch testing (Dalamaga et al., 2024).
• Trace Metals: Nickel, cobalt, and chromium—often present as impurities in pigments, applicators, or packaging—can provoke allergic reactions, especially in sensitized individuals. Nickel allergy remains one of the most common contact allergies globally (Ghosh et al., 2022).
3.5.3 Diagnostic challenges
Diagnosis of cosmetic-related allergies is frequently complicated by several factors:
• Cross-reactivity: Chemical similarities between cosmetic allergens and compounds in foods or the environment can trigger reactions in sensitized individuals. For instance, those allergic to balsam of Peru may also react to cinnamon or citrus oils (Sakali et al., 2021).
• Poly-allergenicity: Cosmetics often contain multiple potential sensitizers, making it difficult to isolate the exact trigger, especially when using multiple products simultaneously (Koshko et al., 2022).
• Delayed Reaction Onset: ACD symptoms may not appear until 24–72 h post-exposure, complicating retrospective identification of the causative agent. Patch testing remains the diagnostic gold standard. Comprehensive panels like the TRUE Test or customized tests with patient-provided products are typically employed (Wu et al., 2023).
3.5.4 Management and prevention
• Avoidance: Once allergens are identified, strict avoidance is paramount. Patients should be educated on ingredient label interpretation and guided toward suitable alternatives devoid of their specific allergens (Pistollato et al., 2021).
• Product Reformulation: Advances in cosmetic science have led to hypoallergenic formulations that minimize sensitizer use. While these reduce overall risk, no product can be universally non-allergenic (Ajayi et al., 2024).
• Regulatory Oversight: Regulatory frameworks like those enforced by the EU and FDA mandate ingredient disclosure and allergen labeling. However, inconsistencies in enforcement and scope remain barriers to comprehensive consumer protection (Debora et al., 2025).
• Barrier Support: Restoring the skin’s natural defense mechanisms is key. Emollients containing ceramides, hyaluronic acid, or occlusives support skin barrier repair and reduce vulnerability to irritants and allergens (Park and Hong, 2024b).
In conclusion, cosmetic-induced allergic reactions, especially ACD, pose significant dermatological challenges driven by both immune and non-immune mechanisms. A proactive approach emphasizing precise diagnosis, allergen avoidance, product innovation, and improved regulatory enforcement is essential to enhance safety and reduce the burden of allergic diseases associated with cosmetic use (Chen et al., 2022).
3.6 Skin disorders
Cosmetic-induced skin disorders are widespread, particularly among individuals with sensitive skin or pre-existing conditions such as eczema, rosacea, and psoriasis. These individuals often have compromised skin barriers, making them more susceptible to irritation and allergic reactions from various cosmetic ingredients. Contact dermatitis, one of the most frequent adverse reactions, results from direct exposure to irritants or allergens (Clouston et al., 2022). It can be categorized into irritant contact dermatitis (ICD) and allergic contact dermatitis (ACD), both of which cause redness, swelling, and discomfort. People with compromised immune systems or a history of eczema are particularly vulnerable to these reactions.
Recent research suggests that certain endocrine-disrupting chemicals (EDCs) found in personal care products may also contribute to inflammatory skin conditions. A longitudinal panel study that involved 18 boys aged 3–7 with diagnosed atopic dermatitis found that elevated urinary levels of mono-n-butyl phthalate (MnBP) and bisphenol A glucuronide (BPAG) were significantly associated with worsened atopic dermatitis (AD) on both the same day and the following day (Kim et al., 2017). Although the study was limited in its small sample size and its inclusion of only boys, it employed a robust time series design using 460 pooled urine samples and daily symptom diaries, while controlling for temperature and humidity. These findings reinforce that phthalates and BPA have inflammatory effects; hence, young children’s exposure through personal care products and packaging should be limited.
Another prospective birth cohort study involving 413 Korean mother-infant pairs found that higher maternal urinary concentrations Di(2-ethylhexyl) phthalate (DEHP) metabolites were associated with significantly increased risk of AD in infants at 6 months of age, especially among girls (Lee et al., 2021). BPA did not show a strong independent association in single-pollutant models, but it contributed to increased AD risk when considered as part of a chemical mixture. The study’s use of repeated urine sampling and Bayesian Kernel Machine Regression (BKMR) strengthened its ability to evaluate interactions between exposures. These results suggest that pregnant women should limit their exposure to products that contain phthalates and BPA to reduce the risk of AD in their babies.
Long-term exposure to certain cosmetic ingredients has been linked to more persistent skin issues, such as perioral dermatitis (a facial rash around the mouth and nose) and acneiform eruptions, which resemble acne but are often triggered by specific cosmetic products like heavy moisturizers or topical steroids (Goel et al., 2024). A case-control study found that urinary BPA concentrations were significantly higher in adults with acne (mean: 7.94 μg/g creatinine) compared to healthy controls (mean: 5.62 μg/g) (Kaya Ozden and Karadag, 2021). Higher BPA levels were also correlated with increased acne severity and earlier onset. Frequent use of products with high alcohol content or astringents can exacerbate dry skin, leading to further complications like irritation and peeling (Preller et al., 2024). The use of heavy or occlusive ingredients in skincare can also block pores and contribute to the development of cystic acne, especially in those predisposed to the condition. Over time, the cumulative effect of exposure to harsh ingredients can worsen the skin’s condition, leading to chronic inflammatory disorders that may require medical intervention. Furthermore, in one analysis, 44% of creams exhibited microbial contamination, surpassing EU safety thresholds 1 month after opening, suggesting poor preservative control and a heightened risk of skin infections (Irfan et al., 2022).
Given these risks, it is increasingly important to prioritize dermatologically tested formulations, especially for those with sensitive or reactive skin. Transparent ingredient lists are crucial in helping consumers identify known irritants or allergens. Dermatologists often recommend patch testing new products to minimize the risk of adverse reactions. Formulations free of parabens, artificial fragrances, sulfates, and other common irritants are essential for maintaining skin health and avoiding unnecessary flare-ups of existing conditions. Furthermore, consumers should be aware that even seemingly “gentle” products can trigger irritation if used inappropriately or in combination with other potent ingredients. Therefore, mindful product selection and a more cautious approach to skincare can significantly reduce the likelihood of cosmetic-induced skin disorders (CDC et al., 2019).
3.7 Neurological effects
Cosmetic products containing synthetic fragrances and volatile organic compounds (VOCs) have increasingly come under scrutiny for their potential neurotoxic effects. Neurological outcomes associated with such exposure span a continuum from acute symptoms such as headaches, migraines, and mood fluctuation to longer-term impacts involving cognitive dysfunction and emotional dysregulation. Although the exact molecular mechanisms remain incompletely understood, growing evidence suggests that certain VOCs can traverse the blood-brain barrier (BBB), a selective membrane that protects the central nervous system from harmful exogenous substances. Once within the brain, these compounds may disrupt neuronal communication, alter neurotransmitter dynamics, and induce oxidative stress, ultimately contributing to neuroinflammation and cellular injury (Mukherjee Das et al., 2022).
Acute exposure to elevated concentrations of synthetic fragrances or VOCs, particularly in confined or inadequately ventilated environments, has been correlated with immediate symptoms including vertigo, nausea, and sensory hypersensitivity. These effects are thought to arise from the stimulation of the olfactory and trigeminal nerve pathways, which are intricately connected to brain regions regulating emotion, pain, and autonomic function. In individuals with underlying vulnerabilities such as multiple chemical sensitivity (MCS) or chronic migraine syndromes, such exposures may precipitate severe exacerbations, often necessitating medical intervention and long-term symptom management (Attard et al., 2022).
Prolonged or chronic exposure to neuroactive VOCs presents a more covert threat, with potential implications for mood disorders, cognitive impairment, and even neurodegenerative conditions. Research has implicated VOCs in the dysregulation of key processes such as synaptic plasticity and neurogenesis, both of which are essential for learning, memory, and adaptive behavior. Persistent oxidative stress and inflammation triggered by VOC exposure may undermine neural integrity, resulting in behavioral disturbances, anxiety, and depressive symptoms (Vale et al., 2022). Moreover, chronic inhalation of these compounds has been shown to elevate cortisol levels and disrupt the hypothalamic-pituitary-adrenal (HPA) axis, further compromising emotional regulation and increasing the risk for affective disorders (Mukherjee Das et al., 2022). Prenatal exposure to BPA has been associated with neurodevelopmental disorders such as autism, ADHD, and learning deficits, likely due to its interference with brain and endocrine development (Chen et al., 2022). These early exposures may also impair organogenesis in systems such as the immune, reproductive, and nervous systems, with consequences that persist into adulthood.
The neurobehavioral risks associated with cosmetic VOC exposure necessitate a multidisciplinary investigative approach. Current evidence remains fragmented, and a comprehensive understanding of the dose-response relationship, susceptibility thresholds, and critical periods of exposure is lacking. Individual factors such as genetic polymorphisms in detoxification enzymes, pre-existing neurological or psychiatric conditions, and cumulative chemical exposures may significantly modulate vulnerability. Advances in neuroimaging techniques (e.g., functional MRI, PET scans) and the identification of neurotoxic biomarkers hold promise in elucidating the structural and functional brain changes attributable to cosmetic-related VOC exposure (Smith and Hotchkiss, 2023).
In conclusion, while the neurological effects of synthetic fragrances and VOCs in cosmetics are still being delineated, available evidence points to their potential role in both acute neurophysiological responses and long-term neurobehavioral dysfunction. The convergence of toxicological, neurological, and environmental research is essential to delineate causal pathways and to inform safer cosmetic formulation standards, especially for individuals with heightened neurological sensitivity.
3.8 Common cosmetic contaminants
Table 1 offers a detailed synthesis of prevalent chemical contaminants found in a wide range of cosmetic products, summarizing their common uses, typical exposure levels, associated health effects, existing regulatory limits, and the most vulnerable population groups. Substances such as parabens, phthalates, formaldehyde and its releasers, heavy metals (including lead and cadmium), triclosan, volatile organic compounds (VOCs), and UV filters like oxybenzone are frequently detected in everyday cosmetic items such as lotions, shampoos, sunscreens, deodorants, nail products, and makeup. These compounds are often added intentionally for functions like preservation, fragrance stabilization, or UV protection, while others, such as heavy metals, appear as unintentional contaminants due to raw material impurities or manufacturing processes.
Despite the often-low concentrations at which these chemicals are present, ranging from parts per million to percentages, their health implications can be significant, especially with chronic use and simultaneous exposure to multiple products. For instance, parabens and phthalates are recognized endocrine disruptors with links to reproductive toxicity and developmental issues. Formaldehyde, classified as a Group 1 carcinogen by the International Agency for Research on Cancer (IARC), is known to cause respiratory irritation and skin sensitization. Heavy metals like lead have no known safe exposure level, particularly for children, and have been associated with neurodevelopmental delays and carcinogenicity. Triclosan has been linked to antimicrobial resistance and hormonal disruption, while certain UV filters such as oxybenzone have shown estrogenic activity and potential environmental toxicity.
The table also highlights notable disparities in regulatory frameworks. While the European Union has established strict bans or concentration limits for many of these substances, the United States often permits their use with minimal restrictions, relying instead on industry self-regulation. This variation underscores a regulatory gap that may lead to increased consumer exposure in less strictly regulated markets. Vulnerable populations such as pregnant women, children, individuals with pre-existing hormonal conditions, and occupationally exposed groups like hairdressers are particularly susceptible to the harmful effects of these chemicals. Overall, the information presented in the table calls attention to the pressing need for comprehensive toxicological assessments, harmonized international regulations, and increased transparency in cosmetic product labeling. It also highlights the importance of raising public awareness about the potential health risks associated with long-term and cumulative exposure to chemical contaminants in personal care products.
3.9 Regulatory frameworks for cosmetic products
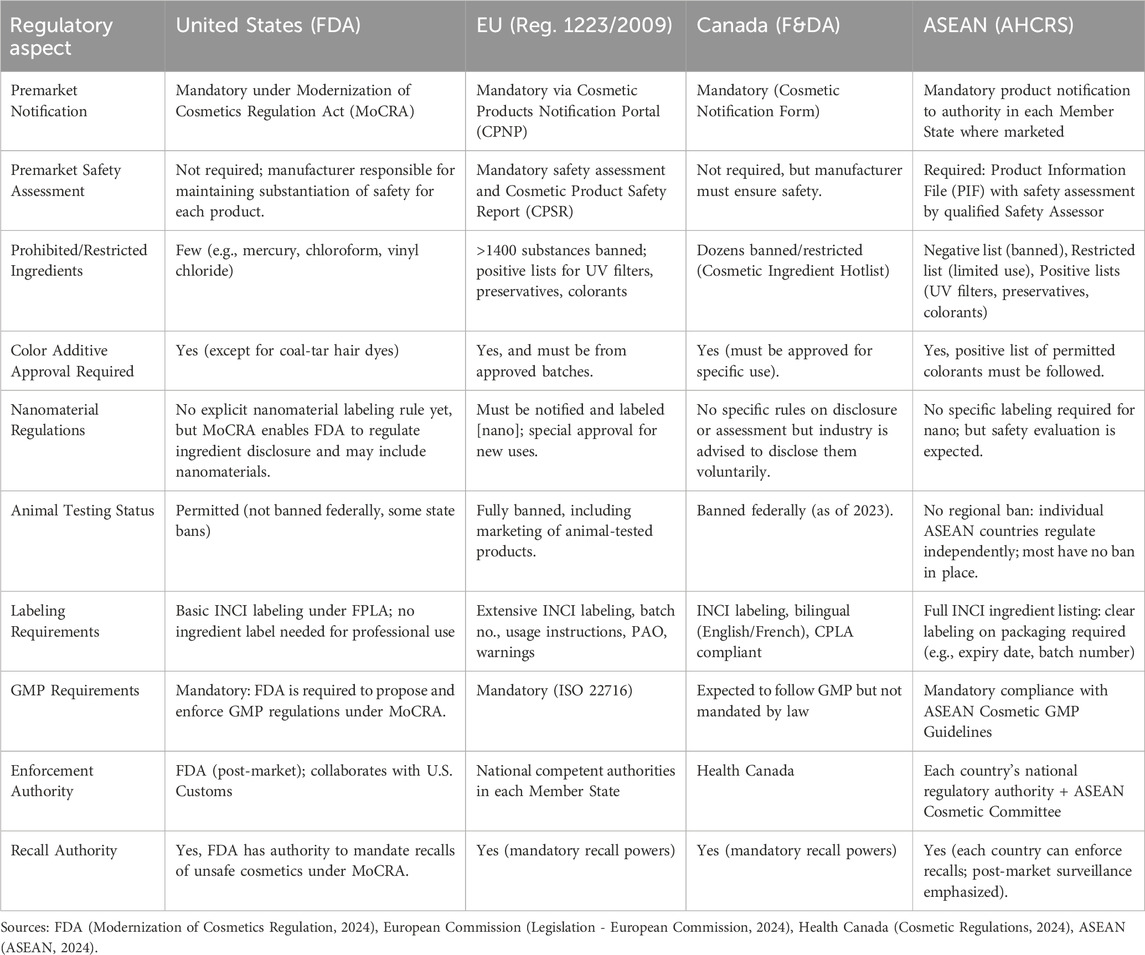
Regulatory frameworks for cosmetics may vary across regions, affecting how products are evaluated, labeled, and monitored. Table 2 compares key regulatory components across the United States, European Union, Canada, and ASEAN, highlighting differences in premarket requirements, safety assessments, ingredient restrictions, labeling, and enforcement mechanisms.
4 Limitations and future research directions
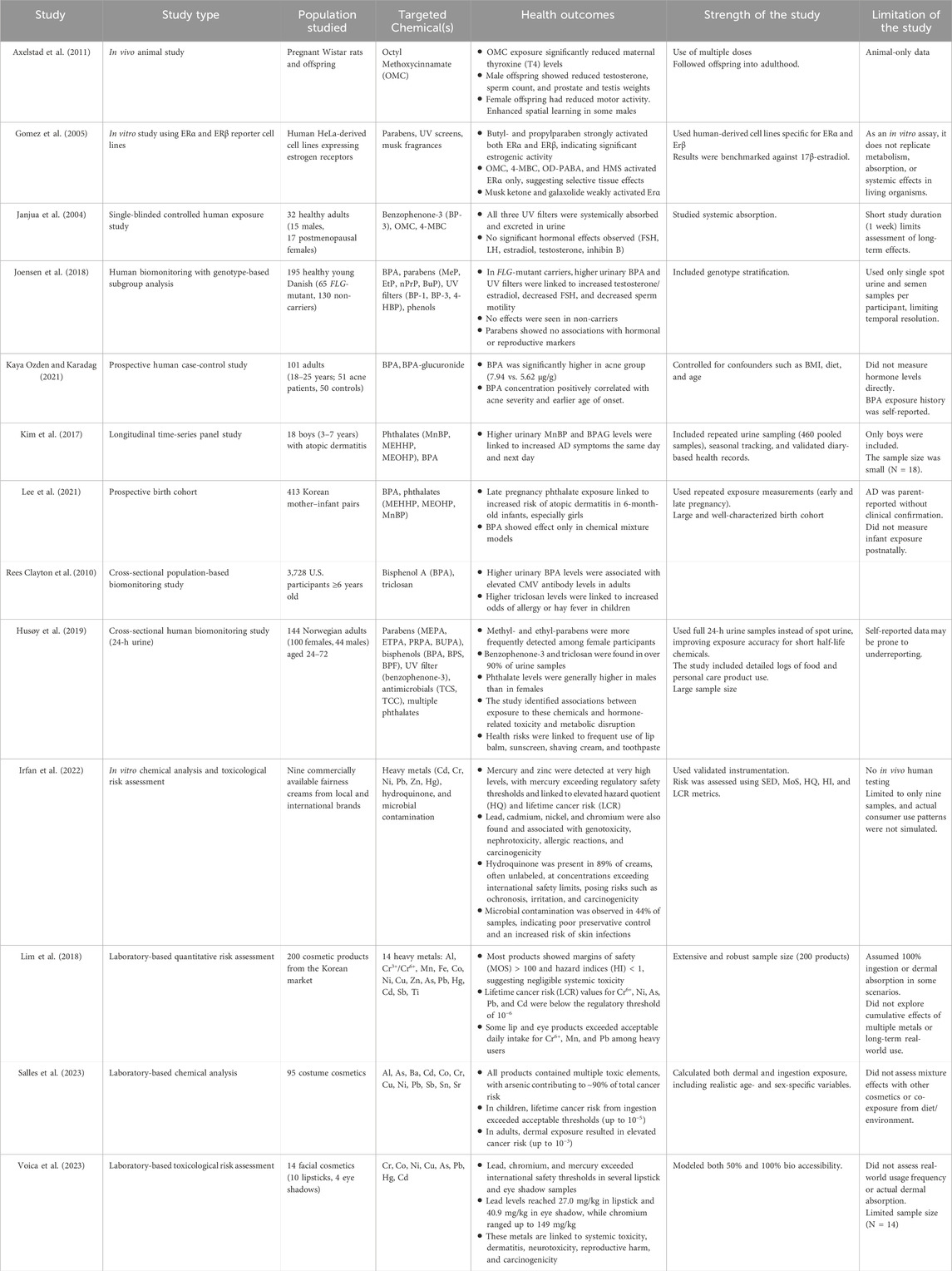
Despite growing evidence on the health risks of cosmetic and personal care products, several limitations hinder a conclusive understanding. The summary of evidence included in this study is presented in Table 3, alongside study population, the methodological strengths and limitations. Most existing studies are observational, limiting causal inference due to potential confounding and exposure misclassification. Long-term effects of chronic, low-dose, and cumulative exposure to multiple cosmetic chemicals remain underexplored, particularly in real-world settings where mixtures of substances are used daily. Vulnerable populations, including pregnant women, infants, and individuals with underlying sensitivities, are often underrepresented in research, despite their increased susceptibility. There is also a lack of standardized biomarkers and biomonitoring data specific to cosmetic-related compounds, making it difficult to assess internal exposure levels and biological responses. Regulatory inconsistencies across countries further complicate risk evaluation, with some harmful chemicals still permitted in certain markets. To address these gaps, future research should focus on longitudinal studies, advanced exposure assessment tools, mechanistic investigations using ‘omics’ technologies, and randomized controlled trials. A more harmonized, multidisciplinary approach is essential to inform safer cosmetic formulations and effective public health policies.
5 Conclusion
In light of the mounting evidence, the cosmetic industry stands at a critical crossroads between innovation and accountability. Ensuring product safety without sacrificing efficacy requires an integrated strategy involving rigorous ingredient screening, investment in green chemistry, and adherence to scientifically validated safety thresholds. Regulatory bodies should establish and enforce clear limits on toxic ingredients, and regulatory frameworks must evolve to include mandatory pre-market safety assessments. Continual post-market surveillance and routine testing are needed to ensure product safety. Current evaluations often overlook the real-world impacts of chronic, low-dose exposure to chemical mixtures. More longitudinal human studies, especially in vulnerable populations such as children, pregnant women, and individuals with genetic susceptibilities are needed. Safety margins should be reassessed for chemicals that are systemically absorbed, such as UV filters. Equally important is fostering a culture of transparency, where consumers are empowered through clear labeling and access to ingredient safety information. Cosmetic manufacturers should actively avoid combining multiple weak estrogenic compounds, especially in products intended for long-term dermal application. Educational campaigns targeting both consumers and healthcare providers can facilitate better recognition of adverse reactions and promote safer product choices. Ultimately, safeguarding public health demands a coordinated effort among scientific communities, regulatory authorities, manufacturers, and consumers. By embracing a proactive, evidence-based approach, the cosmetics sector can transition toward more sustainable and health-conscious practices, ensuring that beauty does not come at the cost of well-being.
Author contributions
SA: Conceptualization, Data curation, Investigation, Methodology, Resources, Validation, Visualization, Writing – original draft, Writing – review and editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Correction note
A correction has been made to this article. Details can be found at: 10.3389/ftox.2025.1713750.
Generative AI statement
The author(s) declare that Generative AI was used in the creation of this manuscript. AI was used to write some parts of the paper.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Abiodun, O. A., and Ayeleru, O. O. (2025). Transport, fate, and detection of personal care products (PCPs) in the environment. Smart Nanomater. Environ. Appl., 487–525. doi:10.1016/b978-0-443-21794-4.00010-7
Ahmed, T. M. K., Bakr, M. M., and Ahmed, Q. A. (2024). Side effects of preservatives on human life. Sci. Res. J. Pharm. doi:10.47310/srjp.2022.v02i02.002
Ajayi, S. A., Olaniyi, O. O., Oladoyinbo, T. O., Ajayi, N. D., and Olaniyi, F. G. (2024). Sustainable sourcing of organic skincare ingredients: a critical analysis of ethical concerns and environmental implications. Asian J. Adv. Res. Rep. 18 (1), 65–91. doi:10.9734/ajarr/2024/v18i1598
Akano, O. A., Hanson, E., Nwakile, C., and Esiri, A. E. (2024). Improving worker safety in confined space entry and hot work operations: best practices for high-risk industries. Glob. J. Adv. Res. Rev. 2 (2), 031–039. doi:10.58175/gjarr.2024.2.2.0056
Aleksandra, M., and Lelieveld, H. L. M. (2022). Ensuring global food safety: exploring global harmonization.
Allmyr, M., Adolfsson-Erici, M., McLachlan, M. S., and Sandborgh-Englund, G. (2006). Triclosan in plasma and milk from Swedish nursing mothers and their exposure via personal care products. Sci. Total Environ. 372 (1), 87–93. doi:10.1016/j.scitotenv.2006.08.007
Alnuqaydan, A. M. (2024). The dark side of beauty: an in-depth analysis of the health hazards and toxicological impact of synthetic cosmetics and personal care products. Front. Public Health 12, 1439027. doi:10.3389/fpubh.2024.1439027
Alurame Eruaga, M. (2024). Enhancing global food safety standards through international collaboration and policy harmonization. Int. J. Sch. Res. Multidiscip. Stud. 4 (1), 020–032. doi:10.56781/ijsrms.2024.4.1.0027
Amutha, C., Gopan, A., Pushbalatatha, I., Ragavi, M., and Reneese, J. A. (2024). Nanotechnol. Gov. Regul. Framew. Responsible Innovation, 481–503. doi:10.1007/978-981-97-6184-5_14
Anagnosti, L., Varvaresou, A., Pavlou, P., Protopapa, E., and Carayanni, V. (2021). Worldwide actions against plastic pollution from microbeads and microplastics in cosmetics focusing on European policies. Has the issue been handled effectively? Mar. Pollut. Bull. 162, 111883. doi:10.1016/j.marpolbul.2020.111883
Anderson, J. T., Cowan, J., Condino-Neto, A., Levy, D., and Prusty, S. (2022). Health-related quality of life in primary immunodeficiencies: impact of delayed diagnosis and treatment burden. Clin. Immunol. 236, 108931. doi:10.1016/j.clim.2022.108931
Aparicio-Soto, M., Curato, C., Riedel, F., Thierse, H. J., Luch, A., and Siewert, K. (2022). In vitro monitoring of human t cell responses to skin sensitizing Chemicals—a systematic review. Cells 11 (1), 83. doi:10.3390/cells11010083
ASEAN (2024). ASEAN Cosmetic Regulation | ChemLinked. Available online at: https://cosmetic.chemlinked.com/cosmepedia/asean-cosmetic-regulation.
Attard, T., Attard, E., Attard, T., and Attard, E. (2022). Heavy metals in cosmetics. Environ. Impact Remediat. Heavy Metals. doi:10.5772/intechopen.102406
Axelstad, M., Boberg, J., Hougaard, K. S., Christiansen, S., Jacobsen, P. R., Mandrup, K. R., et al. (2011). Effects of pre- and postnatal exposure to the UV-filter Octyl Methoxycinnamate (OMC) on the reproductive, auditory and neurological development of rat offspring. Toxicol. Appl. Pharmacol. 250 (3), 278–290. doi:10.1016/j.taap.2010.10.031
Beyuo, J., Sackey, L. N. A., Yeboah, C., Kayoung, P. Y., and Koudadje, D. (2024). The implications of pesticide residue in food crops on human health: a critical review. Discov. Agric. 2 (1), 123. doi:10.1007/s44279-024-00141-z
Bird, T., Steffen, J. E., Tran, T. H., and Egilman, D. S. (2021). A review of the talc industry’s influence on federal regulation and scientific standards for asbestos in talc. New Solut. 31 (2), 152–169. doi:10.1177/1048291121996645
Bopp, S. K., Barouki, R., Brack, W., Dalla Costa, S., Dorne, JLCM, Drakvik, P. E., et al. (2018). Current EU research activities on combined exposure to multiple chemicals. Environ. Int. 120, 544–562. doi:10.1016/j.envint.2018.07.037
CDC, Nceh, DLS, Od (2019). Fourth national report on human exposure to environmental chemicals: updated tables. Available online at: https://stacks.cdc.gov/view/cdc/75822.
Chen, B., Zhao, J., Zhang, R., Zhang, L., Zhang, Q., Yang, H., et al. (2022). Neuroprotective effects of natural compounds on neurotoxin-induced oxidative stress and cell apoptosis. Nutr. Neurosci. 25 (5), 1078–1099. doi:10.1080/1028415X.2020.1840035
Clouston, S. A. P., Hall, C. B., Kritikos, M., Bennett, D. A., DeKosky, S., Edwards, J., et al. (2022). Cognitive impairment and World Trade Centre-related exposures. Nat. Rev. Neurol. 18 (2), 103–116. doi:10.1038/s41582-021-00576-8
Cosmetic Regulations (2024). Cosmetic regulations. Available online at: https://laws-lois.justice.gc.ca/eng/regulations/C.R.C.,_c._869/.
Dalamaga, M., Kounatidis, D., Tsilingiris, D., Vallianou, N. G., Karampela, I., Psallida, S., et al. (2024). The role of endocrine disruptors bisphenols and phthalates in obesity: current evidence, perspectives and controversies. Int. J. Mol. Sci. 25 (1), 675. doi:10.3390/ijms25010675
Darbre, P. D., and Harvey, P. W. (2014). Parabens can enable hallmarks and characteristics of cancer in human breast epithelial cells: a review of the literature with reference to new exposure data and regulatory status. J. Appl. Toxicol. 34 (9), 925–938. doi:10.1002/jat.3027
De Groot, A. C., and Flyvholm, M. A. (2020). Formaldehyde and formaldehyde-releasers. Kanerva’s Occup. Dermatol., 521–542. doi:10.1007/978-3-319-68617-2_37
Debora, S. P., Graduand, S., and Santoro, C. (2025). Sustainability and transparency in the cosmetic industry: the clean beauty movement and consumers’ consciousness. Available online at: https://unitesi.unive.it/handle/20.500.14247/11811.
Dhiman, J., Anupam, K., and Kumar, V. (2024). Saruchi. Varieties, characteristics, and applications of nanofillers in pulp and paper industry. Handb. Nanofillers. doi:10.1007/978-981-99-3516-1_43-1
Di Cicco, M., Kantar, A., Masini, B., Nuzzi, G., Ragazzo, V., and Peroni, D. (2021). Structural and functional development in airways throughout childhood: children are not small adults. Pediatr. Pulmonol. 56 (1), 240–251. doi:10.1002/ppul.25169
El Maghraby, G. M., and Arafa, M. F. (2023). “Skin ceramides based systems in transdermal drug delivery and skin care,” in Green sustainable process for chemical and environmental engineering and science: biomedical applications of green composites, 35–54.
Elcombe, C. S., Evans, N. P., and Bellingham, M. (2022). Critical review and analysis of literature on low dose exposure to chemical mixtures in mammalian in vivo systems. Crit. Rev. Toxicol. 52 (3), 221–238. doi:10.1080/10408444.2022.2091423
Elsaid, A. M., and Ahmed, M. S. (2021). Indoor air quality strategies for air-conditioning and ventilation systems with the spread of the global Coronavirus (COVID-19) epidemic: improvements and recommendations. Environ. Res. 199, 111314. doi:10.1016/j.envres.2021.111314
FDA (2016). FDA issues final rule on safety and effectiveness of antibacterial soaps Rule removes triclosan and triclocarban from overthecounter antibacterial hand and body washes for Immediate Release. Available online at: http://www.cdc.gov/handwashing/.
Fonacier, L., Frankel, D., and Mawhirt, S. (2022). Contact allergens for the allergist. Ann. Allergy Asthma Immunol. 128 (6), 629–644. doi:10.1016/j.anai.2022.03.022
Ghosh, A., Tripathy, A., and Ghosh, D. (2022). Impact of endocrine disrupting chemicals (EDCs) on reproductive health of human. Proc. Zool. Soc. 75 (1), 16–30. doi:10.1007/s12595-021-00412-3
Goel, M., Mittal, A., Jain, V. R., Bharadwaj, A., Modi, S., Ahuja, G., et al. (2024). Integrative functions of the hypothalamus: linking cognition, emotion and physiology for well-being and adaptability. Ann. Neurosci. 32, 128–142. doi:10.1177/09727531241255492
Gomez, E., Pillon, A., Fenet, H., Rosain, D., Duchesne, M. J., Nicolas, J. C., et al. (2005). Estrogenic activity of cosmetic components in reporter cell lines: Parabens, UV screens, and musks. J. Toxicol. Environ. Health A 68 (4), 239–251. doi:10.1080/15287390590895054
Gould, C. F., Heft-Neal, S., Johnson, M., Aguilera, J., Burke, M., and Nadeau, K. (2024). Health effects of wildfire smoke exposure. Annu. Rev. Med. 75, 277–292. doi:10.1146/annurev-med-052422-020909
Groot, A. C. de (2021). Fragrances and essential oils. Boca Raton, FL: CRC Press. Available online at: https://www.routledge.com/Monographs-in-Contact-Allergy-Volume-2-Fragrances-and-Essential-Oils/deGroot/p/book/9781032078946.
Hager, E., Chen, J., and Zhao, L. (2022). Minireview: Parabens exposure and breast cancer. Int. J. Environ. Res. Public Health 19 (3), 1873. doi:10.3390/ijerph19031873
Hauser, R., and Calafat, A. M. (2005). Phthalates and human health. Occup. Environ. Med. 62 (11), 806–818. doi:10.1136/oem.2004.017590
Husøy, T., Andreassen, M., Hjertholm, H., Carlsen, M. H., Norberg, N., Sprong, C., et al. (2019). The Norwegian biomonitoring study from the EU project EuroMix: levels of phenols and phthalates in 24-hour urine samples and exposure sources from food and personal care products. Environ. Int. 132, 105103. doi:10.1016/j.envint.2019.105103
IARC (2006). Formaldehyde, 2-Butoxyethanol and 1-tert-Butoxypropan-2-ol. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Lyon, France: International Agency for Research on Cancer. 88. Available online at: https://www.ncbi.nlm.nih.gov/books/NBK326468/.
Inderbinen, S. G., Kley, M., Zogg, M., Sellner, M., Fischer, A., Kędzierski, J., et al. (2022). Activation of retinoic acid-related orphan receptor γ(t) by parabens and benzophenone UV-filters. Toxicology 471, 153159. doi:10.1016/j.tox.2022.153159
Iqbal, J. D., and Biller-Andorno, N. (2022). The regulatory gap in digital health and alternative pathways to bridge it. Health Policy Technol. 11 (3), 100663. doi:10.1016/j.hlpt.2022.100663
Irfan, M., Shafeeq, A., Siddiq, U., Bashir, F., Ahmad, T., Athar, M., et al. (2022). A mechanistic approach for toxicity and risk assessment of heavy metals, hydroquinone and microorganisms in cosmetic creams. J. Hazard Mater 433, 128806. doi:10.1016/j.jhazmat.2022.128806
Janjua, N. R., Mogensen, B., Andersson, A. M., Petersen, J. H., Henriksen, M., Skakkebæk, N. E., et al. (2004). Systemic absorption of the sunscreens benzophenone-3, octyl-methoxycinnamate, and 3-(4-methyl-benzylidene) camphor after whole-body topical application and reproductive hormone levels in humans. J. Investigative Dermatology 123 (1), 57–61. doi:10.1111/j.0022-202X.2004.22725.x
Janjua, N. R., Kongshoj, B., Petersen, J. H., and Wulf, H. C. (2007). Sunscreens and thyroid function in humans after short-term whole-body topical application: a single-blinded study. Br. J. Dermatology;156, 1080, 1082. doi:10.1111/j.1365-2133.2007.07803.x
Joensen, U. N., Jørgensen, N., Thyssen, J. P., Szecsi, P. B., Stender, S., Petersen, J. H., et al. (2018). Urinary excretion of phenols, parabens and benzophenones in young men: associations to reproductive hormones and semen quality are modified by mutations in the Filaggrin gene. Environ. Int. 121 (Pt 1), 365–374. doi:10.1016/j.envint.2018.09.020
Kaya Ozden, H., and Karadag, A. S. (2021). Could endocrine disruptors be a new player for acne pathogenesis? The effect of bisphenol A on the formation and severity of acne vulgaris: a prospective, case-controlled study. J. Cosmet. Dermatol 20 (11), 3573–3579. doi:10.1111/jocd.14364
Khalid, M., and Abdollahi, M. (2021). Environmental distribution of personal care products and their effects on human health. Iran. J. Pharm. Res. 20 (1), 216–253. doi:10.22037/ijpr.2021.114891.15088
Khan, A. D., and Alam, M. N. (2019). Cosmetics and their associated adverse effects: a review. J. Appl. Pharm. Sci. Res., 1–6. doi:10.31069/japsr.v2i1.1
Kim, E. H., Jeon, B. H., Kim, J., Kim, Y. M., Han, Y., Ahn, K., et al. (2017). Exposure to phthalates and bisphenol A are associated with atopic dermatitis symptoms in children: a time-series analysis. Environ. Health 16 (1), 24. doi:10.1186/s12940-017-0225-5
Koshko, L., Scofield, S., Mor, G., and Sadagurski, M. (2022). Prenatal pollutant exposures and hypothalamic development: early life disruption of metabolic programming. Front. Endocrinol. (Lausanne) 13, 938094. doi:10.3389/fendo.2022.938094
Krause, M., Klit, A., Blomberg Jensen, M., Søeborg, T., Frederiksen, H., Schlumpf, M., et al. (2012). Sunscreens: are they beneficial for health? An overview of endocrine disrupting properties of UV-filters. Int. J. Androl. 35 (3), 424–436. doi:10.1111/j.1365-2605.2012.01280.x
Lazou, A. E. (2024). Food extrusion: an advanced process for innovation and novel product development. Crit. Rev. Food Sci. Nutr. 64 (14), 4532–4560. doi:10.1080/10408398.2022.2143474
Lazzarini, R., de Figueiredo Silva Hafner, M., Rocha, V. B., and Lorenzini, D. (2018). Eczemas. Dermatology Public Health Environ., 389–414. doi:10.1007/978-3-319-33919-1_18
Lee, S., Park, S. K., Park, H., Lee, W., Lee, J. H., Hong, Y. C., et al. (2021). Joint association of prenatal bisphenol-A and phthalates exposure with risk of atopic dermatitis in 6-month-old infants. Sci. Total Environ. 789, 147953. doi:10.1016/j.scitotenv.2021.147953
Legislation - European Commission (2024). Legislation - European Commission. Available online at: https://single-market-economy.ec.europa.eu/sectors/cosmetics/legislation_en.
Lejding, T. (2024). Contact allergy to hexavalent chromium clinical and experimental studies focusing on cement.
Li, T., Sun, H., Wang, M., Dai, W., and Qian, X. (2024). An empirical Study on the effect of blended scents in driving environments from a Neuro-Cognitive perspective. Brain Behav. 14 (10), e70082. doi:10.1002/brb3.70082
Lim, D. S., Roh, T. H., Kim, M. K., Kwon, Y. C., Choi, S. M., Kwack, S. J., et al. (2018). Non-cancer, cancer, and dermal sensitization risk assessment of heavy metals in cosmetics. J. Toxicol. Environ. Health - Part A Curr. Issues 81 (11), 432–452. doi:10.1080/15287394.2018.1451191
Limiting lead in lipstick and other cosmetics FDA (2024). Limiting lead in lipstick and other cosmetics | FDA Available online at: https://www.fda.gov/cosmetics/cosmetic-products/limiting-lead-lipstick-and-other-cosmetics
Lin, R. R., Lin, D. A., and Maderal, A. D. (2024). Toxic ingredients in personal care products: a dermatological perspective. Dermatitis 35 (2), 121–131. doi:10.1089/derm.2023.0215
Line, J., Saville, E., Meng, X., and Naisbitt, D. (2023). Why drug exposure is frequently associated with T-cell mediated cutaneous hypersensitivity reactions. Front. Toxicol. 5, 1268107. doi:10.3389/ftox.2023.1268107
Lohmann, R. M. (2024). Drug-induced skin hypersensitivity reactions: a review of clinical cases and literature. Master's thesis, Vilnius, Lithuania: Vilnius University. Available online at: https://epublications.vu.lt/object/elaba:210545000/.
Maden, S. (2024). Epidermal skin barrier and skin care in Rosacea: a narrative review. Dermis 4 (2). doi:10.35702/derm.10015
Manful, M. E., Ahmed, L., and Barry-Ryan, C. (2024). Cosmetic formulations from natural sources: safety considerations and legislative frameworks in the European Union. Cosmetics 11 (3), 72. doi:10.3390/cosmetics11030072
Martini, A. M. (2022). Yeast functional genomics as a tool to better understand the mode of action of food preservative chemicals. Available online at: https://hdl.handle.net/1813/112008.
Marusic Kristina (2023). A new war on cancer: the unlikely heroes revolutionizing prevention. Available online at: https://islandpress.org/books/new-war-cancer.
Mawhirt, S. L., and Fonacier, L. (2022). Patch testing and the evaluation of contact allergy. Allerg. Immunol. Dis. A Pract. Guide Eval. Diagnosis Manag. Allerg. Immunol. Dis., 511–541. doi:10.1016/b978-0-323-95061-9.00018-7
McMullen, R. L., and Dell’Acqua, G. (2023). History of natural ingredients in cosmetics. Cosmetics 10, 71. doi:10.3390/cosmetics10030071
Modernization of Cosmetics Regulation (2024). Modernization of cosmetics regulation act of 2022 (MoCRA) | FDA. Available online at: https://www.fda.gov/cosmetics/cosmetics-laws-regulations/modernization-cosmetics-regulation-act-2022-mocra.
Mukherjee Das, A., Gogia, A., Janardhanan, R., Babu-Rajendran, R., and Das, B. C. (2022). Environmental contamination and chronic exposure to endocrine-disrupting phthalates: an overlooked and emerging determinant for hormone-sensitive cancers. J. Indian Inst. Sci. 102 (2), 731–742. doi:10.1007/s41745-022-00319-8
Nnorom, I., Igwe, J., and Oji-Nnorom, C. (2005). Trace metal contents of facial (make-up) cosmetics commonly used in Nigeria. Afr. J. Biotechnol. 4 (10), 1133–1138. Available online at: https://www.ajol.info/index.php/ajb/article/view/71343.
Novak-Bilić, G., Vučić, M., Japundžić, I., Meštrović-štefekov, J., Stanić-Duktaj, S., and Lugović-Mihić, L. (2018). Irritant and allergic contact dermatitis - skin lesion characteristics. Acta Clin. Croat. 57 (4), 713–720. doi:10.20471/acc.2018.57.04.13
Nunes, Y. C., Mendes, N. M., Pereira de Lima, E., Chehadi, A. C., Lamas, C. B., Haber, J. F. S., et al. (2024). Curcumin: a golden approach to healthy aging: a systematic review of the evidence. Nutrients 16 (16), 2721. doi:10.3390/nu16162721
Park, Y. W., and Hong, P. (2024a). Cosmetics marketing strategy in the era of the digital ecosystem. Available online at: https://www.springerprofessional.de/en/cosmetics-marketing-strategy-in-the-era-of-the-digital-ecosystem/50067328.
Park, Y. W., and Hong, P. (2024b). Beauty reimagined: navigating the cosmetic industry’s digital transformation. Springer Books. Available online at: https://ideas.repec.org/h/spr/sprchp/978-981-97-3674-4_3.html.
Pastor-Nieto, M. A., and Gatica-Ortega, M. E. (2021). Ubiquity, hazardous effects, and risk assessment of fragrances in consumer products. Curr. Treat. Options Allergy 8 (1), 21–41. doi:10.1007/s40521-020-00275-7
Pistollato, F., Madia, F., Corvi, R., Munn, S., Grignard, E., Paini, A., et al. (2021). Current EU regulatory requirements for the assessment of chemicals and cosmetic products: challenges and opportunities for introducing new approach methodologies. Arch. Toxicol. 95 (6), 1867–1897. doi:10.1007/s00204-021-03034-y
Preller, K. H., Scholpp, J., Wunder, A., and Rosenbrock, H. (2024). Neuroimaging biomarkers for drug discovery and development in schizophrenia. Biol. Psychiatry 96 (8), 666–673. doi:10.1016/j.biopsych.2024.01.009
Raza-Naqvi, S. A., Idrees, F., Sherazi, T. A., Anjum-Shahzad, S., Ul-Hassan, S., Ashraf, N., et al. (2022). Toxicology of heavy metals used in cosmetics. J. Chil. Chem. Soc. 67 (3), 5615–5622. doi:10.4067/s0717-97072022000305615
Rees Clayton, E. M., Todd, M., Dowd, J. B., and Aiello, A. E. (2010). The impact of bisphenol A and triclosan on immune parameters in the U.S. population, NHANES 2003–2006. Environ. Health Perspect. 119 (3), 390–396. doi:10.1289/ehp.1002883
Rico, F., Mazabel, A., Egurrola, G., Pulido, J., Barrios, N., Marquez, R., et al. (2024). Meta-Analysis and analytical methods in cosmetics formulation: a review. Cosmetics 11, 1. doi:10.3390/cosmetics11010001
Sakali, A. K., Bargiota, A., Fatouros, I., Jamurtas, A., Macut, D., Mastorakos, G., et al. (2021). Effects on puberty of nutrition-mediated endocrine disruptors employed in agriculture. Nutrients 13 (11), 4184. doi:10.3390/nu13114184
Salles, F. J., Paniz, F. P., Batista, B. L., Nardocci, A. C., and Olympio, K. P. K. (2023). Potentially toxic elements in costume cosmetics used by children and adults are associated with cancer risk. Int. J. Environ. Res. Public Health 20 (1), 531. doi:10.3390/ijerph20010531
Scopelliti, F., Dimartino, V., Cattani, C., and Cavani, A. (2021). “Mechanisms in allergic contact dermatitis,” in Clinical contact dermatitis: a practical approach, 41–48.
Sicherer, S. H., Abrams, E. M., Nowak-Wegrzyn, A., and Hourihane, J. O. B. (2022). Managing food allergy when the patient is not highly allergic. J. Allergy Clin. Immunol. Pract. 10 (1), 46–55. doi:10.1016/j.jaip.2021.05.021
Singh, S., Roy Pradhan, S., Yadav, A., and Singh, P. K. (2023). Banning asbestos in talcum powder: time for action in India. Dialogues Health 3, 100158. doi:10.1016/j.dialog.2023.100158
Smith, C. K., and Hotchkiss, S. A. M. (2023). Allergic contact dermatitis: Chemical and metabolic mechanisms. Allerg. Contact Dermat. Chem. Metabolic Mech. doi:10.1201/9780429121463
Srinivasulu, M., Chandra, M. S., Gooty, J. M., and Madhavi, A. (2022). Personal care products—fragrances, cosmetics, and sunscreens—in the environment. Environ. Micropollutants A Volume Adv. Pollut. Res., 131–149. doi:10.1016/b978-0-323-90555-8.00015-5
Suresh (2013). Scientific Committee on Consumer Safety SCCS OPINION ON Parabens Updated request for a scientific opinion on propyl-and butylparaben COLIPA n° P82. Available online at: http://ec.europa.eu/health/scientific_committees/consumer_safety/index_en.htm.
Tammaro, A., Narcisi, A., Di Russo, P. P., Abruzzese, C., De Marco, G., Persechino, F., et al. (2012). Contact allergy to limonene from a home-made cosmetic. Eur. J. Inflamm. 10 (2), 243–245. doi:10.1177/1721727X1201000211
Thompson, J. E. (2018). Airborne particulate matter: human exposure and health effects. J. Occup. Environ. Med. 60 (5), 392–423. doi:10.1097/JOM.0000000000001277
Tong, D. Q., Gill, T. E., Sprigg, W. A., Van Pelt, R. S., Baklanov, A. A., Barker, B. M., et al. (2023). Health and safety effects of airborne soil dust in the americas and beyond. Rev. Geophys. 61 (2), e2021RG000763. Available online at:. doi:10.1029/2021RG000763
Ullah, H., Aslam, S., Mustafa, G., Waseem, A., de Freitas Marques, M. B., Gul, Z., et al. (2023). Potential toxicity of heavy metals in cosmetics: fake or fact: a review. Int. J. Environ. Anal. Chem. 104, 8878–8909. doi:10.1080/03067319.2023.2217406
Vale, F., Sousa, C. A., Sousa, H., Santos, L., and Simões, M. (2022). Parabens as emerging contaminants: environmental persistence, current practices and treatment processes. J. Clean. Prod. 347, 131244. doi:10.1016/j.jclepro.2022.131244
Voica, C., Iordache, A. M., Roba, C., and Nechita, C. (2023). Determination of toxic elements in facial cosmetics from the Romanian market and their health risk assessment. Anal. Lett. 56 (2), 244–256. doi:10.1080/00032719.2022.2053699
Witkowska, D., Słowik, J., and Chilicka, K. (2021). Heavy metals and human health: possible exposure pathways and the competition for protein binding sites. Molecules 26 (19), 6060. doi:10.3390/molecules26196060
Wu, Y. L., Lin, Z. J., Li, C. C., Lin, X., Shan, S. K., Guo, B., et al. (2023). Epigenetic regulation in metabolic diseases: mechanisms and advances in clinical study. Signal Transduct. Target Ther. 8 (1), 98. doi:10.1038/s41392-023-01333-7
Keywords: Perfumes, cosmetics, health risks, synthetic chemicals, regulatory guidelines, endocrine disruption, heavy metals, consumer safety
Citation: Alblooshi S (2025) The impact of perfumes and cosmetic products on human health: a narrative review. Front. Toxicol. 7:1646075. doi: 10.3389/ftox.2025.1646075
Received: 12 June 2025; Accepted: 11 August 2025;
Published: 29 August 2025; Corrected: 16 December 2025.
Edited by:
Jaroslav Semerád, Academy of Sciences of the Czech Republic (ASCR), CzechiaReviewed by:
Mohammed Alzuhairi, University of Technology, IraqOpoku Gyamfi, Akenten Appiah-Menka University of Skills Training and Entrepreneurial Development, Ghana
Copyright © 2025 Alblooshi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sharifa Alblooshi, c2hhcmlmYS5hbGJsb29zaGlAenUuYWMuYWU=
 Sharifa Alblooshi
Sharifa Alblooshi