Abstract
Diabetes is affecting the social and economic developments in developing countries like Sri Lanka. Siddha Medicine (Tamil Medicine) is mostly practiced in the Eastern and Northern Provinces of Sri Lanka. Our recent review of Sri Lankan Siddha historical documents identified 171 plant species used to prepare anti-diabetic preparations. On the other hand, there is no study of plants currently used to treat diabetes in Sri Lankan Siddha Medicine. Hence, the aim of this study is to identify and document the plant species currently used in anti-diabetic preparations in Eastern Province, also enabling a comparative analysis with historical uses. Further, assessing the level of scientific evidence (in vitro, in vivo, and clinical studies) available for recorded species. A systematically prepared questionnaire was used to conduct an ethnobotanical survey with 27 Siddha healers residing in Eastern Province to identify the currently used anti-diabetic plants. Furthermore, Web of Science electronic database was used to assess the level of scientific evidence available excluding widespread and very well studied species. On average 325 diabetic patients were seen by 27 healers per week. Interestingly, inorganic substances, and animal parts used as ingredients in historical anti-diabetic preparations are currently not used in Eastern Province. A total of 88 plant species from 46 families were reported in this study. Syzygium cumini (L.) Skeels was the most frequently recorded species and the largest number of taxa are from Fabaceae. Remarkably, one third of reported species were not stated in Sri Lankan Siddha historical documents. The highest number of plant species (59%) have been studied up to an in vivo level followed by no scientific evidence for anti-diabetic activity found (27%), clinical evidence (10%), and in vitro (2%). This is the first ethnobotanical study of plants used to treat diabetes by Siddha healers in the Eastern Province in Sri Lanka. Moreover, awareness should be created to the diabetics about the side effects of herb-drug interactions and complications caused by taking both herbal preparations and biomedical drugs.
Introduction
There are an estimated 1.16 million people (20–79 years old) with diabetes in Sri Lanka with 0.60 million undiagnosed cases. Furthermore, in 2015 16,319 (20–79 years old) deaths caused by diabetes were recorded (IDF, 2015). About 70% of rural population relies on traditional medicinal system as their primary health care (Perera, 2012). While there are two types of diabetes: Type 1 (insulin deficiency) and type 2 (insulin resistance), globally the majority of cases are type 2 diabetes. Heart attacks, lower limb amputation, blindness, and kidney failure are mostly caused by diabetes. The number of diabetics is rising fast in middle- and low-income countries (WHO, 2017).
Sri Lanka is a South Asian island situated in the Indian Ocean. It has 65,610 km2 area with a population of 21.2 million (Department of Census and Statistics Sri Lanka, 2017). The largest (75%) ethnic group is Sinhalese, followed by Sri Lankan Tamil (11%), Sri Lanka Moor (9%), and Indian Tamil (4%). The official and national languages of Sri Lanka are Sinhala and Tamil. The major and official religion is Buddhism (70%) followed by Saivism (13%), Islam (10%), and Christianity (7%) (Department of Census and Statistics Sri Lanka, 2012). There are three climatic zones (dry, intermediate, and wet) in Sri Lanka (Department of Meteorology Sri Lanka, 2016).
A total of 4,143 plant species of 214 families identified in Sri Lanka were listed in a work by Senaratna (2001). Furthermore, 75% of these species were indigenous whereas 25% of them were introduced/exotics, with 32% having become naturalized and 68% being under cultivation (Senaratna, 2001). In 2010 nearly 29.7% of Sri Lankan land area is covered by natural forests (Forest Department Sri Lanka, 2017). The vegetation of Sri Lanka can be categorized as follows according to the impact of soil and elevation: montane, sub montane, lowland rain, moist monsoon, dry monsoon, riverine dry, sparse and open, and mangrove forests (Forest Department Sri Lanka, 2017). The most frequently identified plant families in the montane forests were Lauraceae (Actinodaphne, Cinnamomum, and Litsea) and Myrtaceae (Eugenia, Rhodomyrtus, and Syzygium) (Werner and Balasubramaniam, 1992). The main Sri Lankan export crops are Camellia sinensis (tea), Hevea brasiliensis (rubber), and Cocos nucifera (coconut) (Department of Census and Statistics Sri Lanka, 2017).
Four traditional medicinal systems (Ayurveda, Siddha, Unani, and Deshiya Chikitsa) are currently practiced in Sri Lanka (Weragoda, 1980). Siddha (or Tamil) Medicine is mostly practiced in the Eastern and Northern Provinces of Sri Lanka (Sivashanmugarajah, 2000). So far, only two ethnobotanical surveys have been carried out in Jaffna (Rajamanoharan, 2014) and Vavuniya (Rajamanoharan, 2016), Northern Province. However, so far, no ethnobotanical survey has been carried out in Eastern Province. A recent review of Sri Lankan Siddha historical documents revealed 171 species in 73 families were used to treat diabetes in Sri Lankan Siddha Medicine (Sathasivampillai et al., 2017). However, there is no documentation of species currently used to treat diabetes in Sri Lankan Siddha Medicine.
For centuries a large number of disorders including diabetes have been managed and treated using traditional herbal preparations (Nearing, 1985). Ethnobotanical surveys focusing on the use of medicinal substances play an important role in safeguarding and understanding local/traditional and local knowledge and are seen a starting point for drug discovery (Fabricant and Farnsworth, 2001). Safeguarding is, however, only one element of such research. Modern use of “traditional” medicines are embedded in complex expectations both by those who are the keepers of this knowledge and by the wider society. However, outcomes of ethnobotanical surveys are not frequently compared with the available published scientific reports to study the pharmacological properties of plants used in the preparations. Recent surveys on anti-diabetic plants have been published for many countries such as India (Vidyasagar and Siddalinga, 2013), South Africa (Davids et al., 2016), Nigeria (Salihu Shinkafi et al., 2015), México (Andrade-Cetto and Heinrich, 2005), Turkey (Durmuşkahya and Öztürk, 2013), and China (Guo et al., 2017).
Sri Lankan local and traditional medicine is a practice which has developed as a part of the historical development of Sri Lanka incorporating not only autochthonous traditions but also the multiple impacts of diverse cultures. Our previous research (Sathasivampillai et al., 2017) identified a large number of taxa documented in historical texts which were a part of the formal curriculum for training Siddha medical practitioners for conditions associated with diabetes.
Therefore, the aim of this work is to identify and document the species currently used to treat diabetes in Eastern Province, Sri Lanka in Siddha Medicine, enabling a comparative analysis with the historical uses. With this we want to contribute to an understanding of continuity and change of practice as it relates to diabetes in this culture. This also forms a basis for a systematic, comparison with medicinal plants usage in other regions of Sri Lanka. In addition, we assess the levels of scientific evidence (in vitro, in vivo, and clinical studies) of the reported species based on a bibliographic assessment.
Background and methods
The study region
This ethnobotanical study was conducted with Siddha healers residing in the Eastern Province in the dry climatic zone of Sri Lanka which consists of three districts (Batticaloa, Ampara, and Trincomalee) (Figure 1). Tamil is the major language spoken in this area. Siddha Medicine is mostly practiced in Tamil speaking regions around the world (AYUSH, 2016).
Figure 1
Ethical approval of the research
Research ethical approval was obtained from UCL Research Ethics Committee (9141/001) on 13.06.2016 prior starting the investigation in Sri Lanka. The purpose of this study and informed consent page were read to each healer before beginning the interview. The interview was carried out only after receiving the verbal consent from each informant. Healers participated voluntarily in this study, were free to withdraw at any point in time and no compensation was provided.
This study was conducted recognizing the relevant obligations under the Convention on Biological Diversity and subsequent agreements, most notably the Nagoya Protocol which had at the time of writing (07.01.2018) not yet been signed or ratified by Sri Lanka, while the UK ratified it on 22.02.2016 (https://www.cbd.int/abs/doc/protocol/nagoya-protocol-en.pdf 07.01.2018). Also, the right to authorship and use the traditional knowledge of all informants were preserved. Using this information except scientific publication requires permission from the traditional owners of knowledge. However, no permission is required from the government to collect and preserve plant material samples within Sri Lanka. It was started prior to the publication of the ConsEFS statement on best practice in ethnopharmacological research (Heinrich et al., 2018), but has followed these guidelines.
Ethnobotanical data collection and interviews
This ethnobotanical investigation was conducted from July to September 2016. SVS conducted the interviews jointly with PRR (community health medical officer at Planning Unit, Eastern Provincial Department of Indigenous Medicine and in charge of Eastern Provincial Herbal Garden, Trincomalee) being present during the interviews.
The interviews were carried out in Tamil. Only Siddha healers whose families have been practicing Siddha Medicine for at least two generations and are registered at the Sri Lanka Ministry of Indigenous Medicine were included in this study. Siddha healers were actively chosen by the community health medical officer from the database of registered healers. Permission and appointments were obtained verbally from each healer by the community health medical officer. Then, the interviews were held at Siddha healers' homes.
Initially, 33 Siddha healers were approached. However, 6 of them do not practice any treatment for diabetes. Hence, they were excluded from this study. Interviews using a questionnaire including semi-structured questions were conducted with the healers (Appendix). The questions focused on social and demographic data such as gender, age, and experience and species currently used to treat diabetes. With each healer, the interview lasted for a minimum of 15 minutes.
Voucher specimens and plant identification
Reported species which are available locally or cultivated were collected. The fieldwork for collecting plant part samples was conducted during September 2016 to June 2017, a period ideal for collecting flowering and fruiting specimens. Specimens are deposited at the Herbarium of the Eastern Provincial Herbal Garden, Trincomalee (for voucher specimen numbers see Table 1) and identified by PRR. Siddha healers mentioned Tamil names of the species. Scientific names and families are based on Sugathadasa et al. (2008) and validated using The Plant List (2013) and the Royal Botanic Gardens Kew, Medicinal Plant Service (2018).
Table 1
| Category | No. of healers | Percentage/% |
|---|---|---|
| GENDER | ||
| Male | 25 | 93 |
| Female | 2 | 7 |
| AGE/YEAR | ||
| 21–30 | 0 | 0 |
| 31–40 | 6 | 22 |
| 41–50 | 0 | 0 |
| 51–60 | 0 | 0 |
| 61–70 | 17 | 63 |
| 71–80 | 3 | 11 |
| 81–90 | 1 | 4 |
| 90–100 | 0 | 0 |
| EXPERIENCE/YEARS | ||
| 1–10 | 1 | 4 |
| 11–20 | 5 | 19 |
| 21–30 | 0 | 0 |
| 31–40 | 0 | 0 |
| 41–50 | 17 | 63 |
| 51–60 | 3 | 11 |
| 60–70 | 1 | 4 |
| Total | 27 | 100 |
Demographic data of the Siddha healers (N = 27).
Data analysis
A database of reported species including family, scientific name, part used, and number of participants citing it as being of medicinal importance was created. Plant species currently used to treat diabetes by Siddha healers (this study) were compared with the species historically used to treat diabetes in Sri Lankan Siddha Medicine (Sathasivampillai et al., 2017). Also, species confirmed in this work were compared with the species used to treat diabetes reported in the ethnobotanical studies previously carried out in the other areas where Siddha Medicine is mostly practiced in Jaffna (Rajamanoharan, 2014) and Vavuniya (Rajamanoharan (2016). Other comparisons with ethnobotanical information were carried out based on local knowledge of Eastern Province of Sri Lanka by the authors SVS and PRR. Furthermore, an assessment of the levels of scientific evidence of each species was carried out using the electronic database Web of Science until September 2017. The method described in Sathasivampillai et al. (2017) was followed to identify the relevant published studies. Species listed in Brendler (2010), Upton et al. (2016), American Herbal Pharmacopoeia (2017), European Medicines Agency (2018), and WHO (1999, 2004, 2007, 2009) were considered to be very well studied and global plants. Therefore, they were excluded from the literature search to identify the level of scientific evidence.
Results and discussion
Socio-demographic characteristics of the siddha healers participated in this study
A total of 27 Siddha healers residing in Eastern Province and currently treating diabetes were interviewed in detail for this study. The majority of participants were men. The highest number of participants were in the 61–70 age group. Also, the majority of the healers had 41–50 years of experience practicing Siddha Medicine (Table 1).
Current diagnosis methods employed by siddha healers
This study relies on the self-reporting by the healers on their practice. While some of the cases of diabetes treated may well be confirmed using bio-medical diagnosis, it was not possible in the context of this study to ascertain any diagnosis. Diabetes is termed as Neerilivu (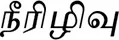 ) in Siddha Medicine (Anonymous, 2000). The common symptoms of diabetes in Siddha Medicine mentioned include dry tongue, chest and throat; feeling lazy, ants and flies gather around the urine, weight loss, feeling thirsty, and excessive urination (Anonymous, 2000, 2003). Diabetes is categorized into 24 types referring to the color and taste of the urine in Siddha Medicine (Sithamparthanuppillai, 1982). In Siddha Medicine eight diagnostic methods (pulse examination, touch, tongue examination, body color, speech, eye, stool, and urine) are used to diagnose a disorder (Narayanaswami, 1975). However, in this investigation Siddha healers mentioned they only use pulse reading to diagnose diabetic cases combination with above mentioned Siddha symptoms of diabetes.
) in Siddha Medicine (Anonymous, 2000). The common symptoms of diabetes in Siddha Medicine mentioned include dry tongue, chest and throat; feeling lazy, ants and flies gather around the urine, weight loss, feeling thirsty, and excessive urination (Anonymous, 2000, 2003). Diabetes is categorized into 24 types referring to the color and taste of the urine in Siddha Medicine (Sithamparthanuppillai, 1982). In Siddha Medicine eight diagnostic methods (pulse examination, touch, tongue examination, body color, speech, eye, stool, and urine) are used to diagnose a disorder (Narayanaswami, 1975). However, in this investigation Siddha healers mentioned they only use pulse reading to diagnose diabetic cases combination with above mentioned Siddha symptoms of diabetes.
Number of diabetic patients seen by a siddha healer
According to the information provided by the Siddha healers, on average 12 diabetics are seen by a healer per week. However, they did not show any written evidences of number of patients consulting them. Also, they did not have any information of how many of the patients come back for a regular consultation. Usually preparations sufficient for 1 month was given to each patient. Anti-diabetic preparations are prepared by the healers at home.
Types of ingredients currently used
Herbal, inorganic (such as metals and minerals), and animal products have been recorded commonly as elements of Siddha Medicine (NIS, 2018). Also, these three types of ingredients were included in Siddha historical documents analyzed for anti-diabetic preparations (Sathasivampillai et al., 2017). Siddha healers in this study reported only botanical ingredients to prepare anti-diabetic preparations.
Species reported by siddha healers
Overall, 88 species from 46 families was documented in this study (Table 2). Syzygium cumini was the most cited species (cited by 21 Siddha healers) followed by Gymnema sylvestre, Artocarpus heterophyllus, Salacia reticulata, and Achyranthes aspera. The largest number of reported taxa are from the Fabaceae. Leaves were cited as the most frequently used plant part followed by fruits, whole plant, root, and bark. The majority of the plants documented in this study were South Asian medicinal plants, like Withania somnifera, Typhonium trilobatum, Tribulus terrestris, Toddalia asiatica, and Tinospora sinensis followed by food plants including Achyranthes aspera, Borassus flabellifer, Cinnamomum verum, Eleusine coracana, and Limonia acidissima.
Table 2
| Family, scientific name, voucher specimen identification | Tamil name | Part used | Local use, number of times cited | ||
|---|---|---|---|---|---|
| Food | Medicine | Other | |||
| ACANTHACEAE | |||||
| Hygrophila auriculata (Schumach.) Heine | 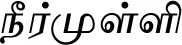 (Neermulli) | Leaf | 4 | ||
| Andrographis paniculata (Burm.f.) Nees (VS006)*# | 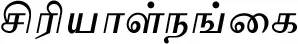 (Siriyaalnangai) | Whole plant | 3 | ||
| AMARANTHACEAE | |||||
| Achyranthes aspera L. (VS001) | 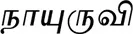 (Naayuruvi) | Whole plant | 11 | ||
| Aerva lanata (L.) Juss. (VS003)# | 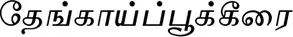 (Thengaaippookkeerai) | Whole plant | 2 | ||
| Alternanthera sessilis (L.) R.Br. ex DC. (VS004) | 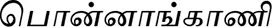 (Ponnaangkaani) | Leaf | 2 | ||
| AMARYLLIDACEAE | |||||
| Allium sativum L.* | 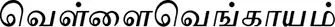 (Vellai vengaayam) | Bulb | 3 | ||
| ANACARDIACEAE | |||||
| Anacardium occidentale L. (VS005)* | 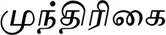 (Munthirihai) | Fruit | 1 | ||
| APIACEAE | |||||
| Anethum graveolens L.* | 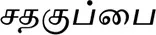 (Sathahuppai) | Seed | 1 | ||
| Coriandrum sativum L.*# | 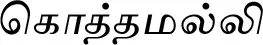 (Koththamalli) | Seed | 5 | ||
| Cuminum cyminum L.* | 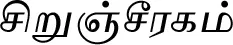 (Sirunjcheeraham) | Fruit | 5 | ||
| Ferula assa-foetida L.* | 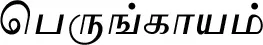 (Perungkaayam) | Resin | 1 | ||
| Foeniculum vulgare Mill.* | 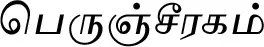 (Perunjcheeraham) | Fruit | 2 | ||
| Trachyspermum roxburghianum (DC.) H. Wolff$ | 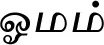 (Omam) | Fruit | 3 | ||
| APOCYNACEAE | |||||
| Catharanthus roseus (L.) G.Don (VS013)*# | 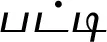 (Patti) | Root | 1 | ||
| Gymnema sylvestre (Retz.) R.Br. ex Sm. (VS024)*# | 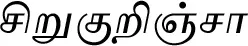 (Sirukurinjaa) | Leaf, root | 18 | ||
| ARACEAE | |||||
| Typhonium trilobatum (L.) Schott#$ | 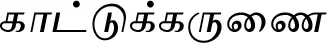 (Kaattukkarunai) | Rhizome | 1 | ||
| ARECACEAE | |||||
| Borassus flabellifer L. | 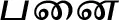 (Panai) | Fruit | 2 | ||
| ARISTOLOCHIACEAE | |||||
| Aristolochia bracteolata Lam.# | 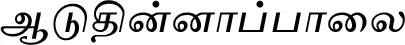 (Aaduthinnaappaalai) | Whole plant | 1 | ||
| ASCLEPIADACEAE | |||||
| Calotropis procera (Aiton) Dryand.# |  (Vellerukku) | Root | 1 | ||
| Dregea volubilis (L.f.) Benth. ex Hook.f. (VS018)# | 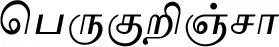 (Perukurinjaa) | Leaf | 4 | ||
| ASTERACEAE | |||||
| Cyanthillium cinereum (L.) H.Rob. (VS016) | 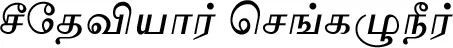 (Seetheviyaar sengkaluneer) | Whole plant | 2 | ||
| Eclipta prostrata (L.) L. (VS019) | 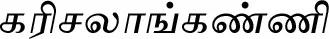 (Karisalaangkanni) | Whole plant | 3 | ||
| BASELLACEAE | |||||
| Basella alba L. (VS009)#$ | 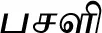 (Pasali) | Leaf | 3 | ||
| BIGNONIACEAE | |||||
| Stereospermum chelonoides (L.f.) DC. | 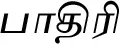 (Paathiri) | Root | 2 | ||
| CALOPHYLLACEAE | |||||
| Mesua ferrea L.$ | 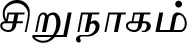 (Sirunaaham) | Flower | 2 | ||
| CAPPARACEAE | |||||
| Crateva adansonii DC.#$ | 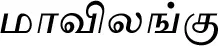 (Maavilangu) | Bark | 1 | ||
| CARICACEAE | |||||
| Carica papaya L. (VS011)*# |  (Pappaasi) | Leaf | 3 | ||
| CELASTRACEAE | |||||
| Salacia reticulata Wight (VS036) | 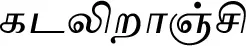 (Kadaliraanji) | Bark | 12 | ||
| COMBRETACEAE | |||||
| Terminalia arjuna (Roxb. ex DC.) Wight & Arn. (VS041)* | 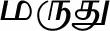 (Maruthu) | Bark | 1 | ||
| Terminalia bellirica (Gaertn.) Roxb. (VS042)* | 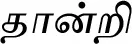 (Thaandri) | Fruit | 2 | ||
| Terminalia chebula Retz.* | 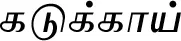 (Kadukkaai) | Fruit | 5 | ||
| CONVOLVULACEAE | |||||
| Evolvulus nummularius (L.) L. (VS020)#$ |  (Vellai vittunukiraanthi) | Whole plant | 1 | ||
| Ipomoea aquatica Forssk. (VS025) | 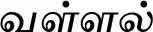 (Vallal) | Leaf | 4 | ||
| Merremia emarginata (Burm. f.) Hallier f.# | 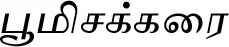 (Poomisakkarai) | Rhizome | 3 | ||
| COSTACEAE | |||||
| Cheilocostus speciosus (J.Koenig) C.D.Specht (VS014) | 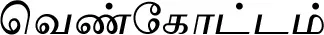 (Venkottam) | Rhizome | 3 | ||
| CUCURBITACEAE | |||||
| Coccinia grandis (L.) Voigt (VS015) | 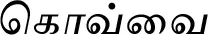 (Kovvai) | Leaf | 9 | ||
| Momordica charantia L. (VS027)* | 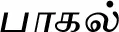 (Paahal) | Fruit, leaf | 3 | ||
| Mukia maderaspatana (L.) M.Roem. (VS028) | 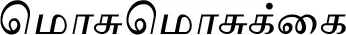 (Mosumosukkai) | Leaf | 6 | ||
| FABACEAE | |||||
| Cassia fistula L. (VS012)* | 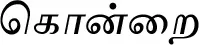 (Kondrai) | Bark | 1 | ||
| Indigofera aspalathoides DC.#$ | 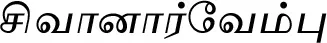 (Sivanaarvembu) | Whole plant | 2 | ||
| Indigofera tinctoria L.$ | 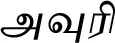 (Avuri) | Root | 2 | ||
| Pongamia pinnata (L.) Pierre (VS035)#$ | 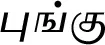 (Pungu) | Root | 3 | ||
| Senna auriculata (L.) Roxb. (VS037) | 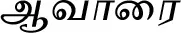 (Aavaarai) | Bark, flower, leaf, root, seed | 5 | ||
| Senna sophera (L.) Roxb. (VS038) | 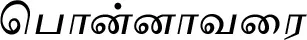 (Ponnaavarai) | Flower | 2 | ||
| Sesbania grandiflora (L.) Pers. (VS039)# | 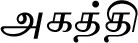 (Ahaththi) | Leaf | 3 | ||
| Trigonella foenum- graecum L.* | 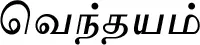 (Venthayam) | Seed | 6 | ||
| LAMIACEAE | |||||
| Ocimum tenuiflorum L. (VS030)*# | 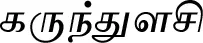 (Karunthulasi) | Whole plant | 3 | ||
| LAURACEAE | |||||
| Cinnamomum verum J.Presl* | 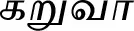 (Karuvaa) | Bark, leaf | 1 | ||
| MALVACEAE | |||||
| Abutilon indicum (L.) Sweet | 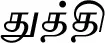 (Thuththi) | Whole plant | 3 | ||
| Thespesia populnea (L.) Sol. ex Corrêa (VS043) | 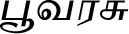 (Poovarasu) | Bark | 1 | ||
| MELIACEAE | |||||
| Azadirachta indica A.Juss. (VS008)* | 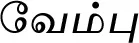 (Vembu) | Bark, tender, leaf | 4 | ||
| MENISPERMACEAE | |||||
| Coscinium fenestratum (Goetgh.) Colebr. | 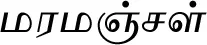 (Maramanjal) | Stem | 4 | ||
| Tinospora sinensis (Lour.) Merr.* | 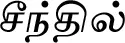 (Seenthil) | Stem | 1 | ||
| MORACEAE | |||||
| Artocarpus heterophyllus Lam. (VS007) | 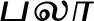 (Palaa) | Mature | 13 | ||
| Ficus benghalensis L. (VS021) | 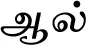 (Aal) | Bark | 2 | ||
| Ficus racemosa L. (VS022) | 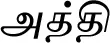 (Aththi) | Bark | 2 | ||
| Ficus religiosa L. (VS033) | 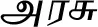 (Arasu) | Bark | 3 | ||
| MYRISTICACEAE | |||||
| Myristica fragrans Houtt. | 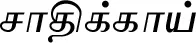 (Saathikkaai) | Leaf, mace | 3 | ||
| MYRTACEAE | |||||
| Syzygium aromaticum (L.) Merr. & L.M.Perry* |  (Karaambu) | Flower, bud | 4 | ||
| Syzygium cumini (L.) Skeels (VS040) | 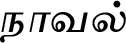 (Naaval) | Bark, root, seed | 2 | ||
| NYCTAGINACEAE | |||||
| Boerhavia diffusa L.# | 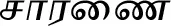 (Saaranai) | Leaf | 2 | ||
| OXALIDACEAE | |||||
| Averrhoa carambola L.# | 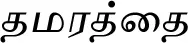 (Thamaraththai) | Fruit | 1 | ||
| PASSIFLORACEAE | |||||
| Passiflora edulis Sims (VS032)*# | 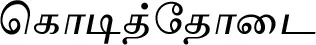 (Kodiththodai) | Leaf | 2 | ||
| PEDALIACEAE | |||||
| Pedalium murex L.#$ | 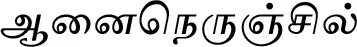 (Aanainerunjil) | Whole plant | 2 | ||
| PHYLLANTHACEAE | |||||
| Phyllanthus emblica L. (VS033)* | 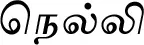 (Nelli) | Fruit, root | 8 | ||
| PIPERACEAE | |||||
| Piper longum L. (VS034)* | 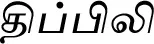 (Thippili) | Fruit | 2 | ||
| Piper nigrum L.* | 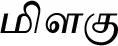 (Milahu) | Fruit | 2 | ||
| PLANTAGINACEAE | |||||
| Bacopa monnieri (L.) Wettst.*# |  (Piraammi) | Leaf | 1 | ||
| Scoparia dulcis L. | 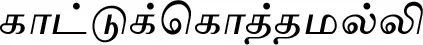 (Kaattukkoththamalli) | Leaf | 1 | ||
| POACEAE | |||||
| Chrysopogon zizanioides (L.) Roberty | 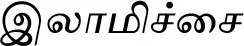 (Ilaamichchai) | Root | 1 | ||
| Cynodon dactylon (L.) Pers. (VS017)*# | 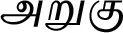 (Aruhu) | Whole plant | 3 | ||
| Eleusine coracana (L.) Gaertn. | 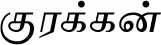 (Kurakkan) | Seed | 1 | ||
| Oryza sativa L. (VS031)* | 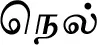 (Nel) | Seed | 4 | ||
| Setaria italica (L.) P.Beauv.# | 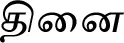 (Thinai) | Seed | 2 | ||
| RANUNCULACEAE | |||||
| Nigella sativa L.* | 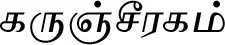 (Karunjcheeraham) | Seed | 2 | ||
| RUBIACEAE | |||||
| Pavetta indica L.#$ |  (Paavattai) | Leaf | 2 | ||
| RUTACEAE | |||||
| Aegle marmelos (L.) Corrêa (VS002)* | 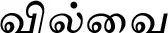 (Vilvai) | Bark, fruit | 7 | ||
| Limonia acidissima Groff (VS026)$ | 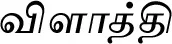 (Vilaaththi) | Fruit | 2 | ||
| Murraya koenigii (L.) Spreng. (VS029)* | 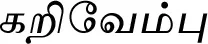 (Karivembu) | Leaf | 10 | ||
| Toddalia asiatica (L.) Lam.*# | 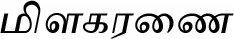 (Milaharanai) | Root | 1 | ||
| SAPINDACEAE | |||||
| Cardiospermum halicacabum L. (VS010) | 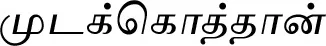 (Mudakkoththaan) | Leaf | 5 | ||
| SCHISANDRACEAE | |||||
| Illicium verum Hook.f.*# | 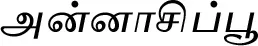 (Annaasippoo) | Fruit | 1 | ||
| SOLANACEAE | |||||
| Withania somnifera (L.) Dunal* | 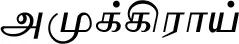 (Amukkiraai) | Rhizome | 4 | ||
| ZINGIBERACEAE | |||||
| Curcuma longa L.* | 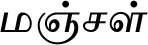 (Manjal) | Rhizome | 5 | ||
| Elettaria cardamomum (L.) Maton* | 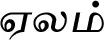 (Elam) | Fruit | 5 | ||
| Kaempferia galanga L.$ | 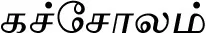 (Kachcholam) | Rhizome | 2 | ||
| Zingiber officinale Roscoe* | 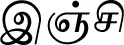 (Inji) | Rhizome | 4 | ||
| ZYGOPHYLLACEAE | |||||
| Tribulus terrestris L. (VS044)* | 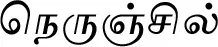 (Nerunjchil) | Whole plant | 1 | ||
Reported plant species used to treat diabetes in Siddha Medicine in Eastern Province (n = 27).
Local use, Food, medicinal, and other uses of plant species were categorized according to the most common local use of the particular plant species.
Plant species excluded from further analysis in this study.
Plant species had not been reported in either anti-diabetic preparations in Sri Lankan Siddha historical documents or ethnobotanical surveys carried out in Siddha Medicine practicing regions in Sri Lanka.
Plant species without any bioscientific evidence for anti-diabetic activity.
Remarkably, one third of the species reported in this study had not been recorded before either in anti-diabetic preparations of Sri Lankan Siddha historical documents or in the ethnobotanical surveys carried out in regions of Sri Lanka where Siddha Medicine is practiced (marked as “#” in Table 2). For example, Sesbania grandiflora and Pedalium murex are reported for the first time in this study as ingredients in Siddha anti-diabetic preparations in Sri Lanka. At the same time there is a strong overlap with the historical Siddha documents with 61% of the recorded species also stated as anti-diabetic preparations including Ficus racemosa and Salacia reticulata. Since these are used as textbooks for training Siddha healers this indicates that both written records (Leonti, 2011) and oral transmission / local innovations form the basis for the species used. The most frequently used species in this study (S. cumini) was also used in the anti-diabetic preparations in the historical documents. Senna auriculata is the most frequently stated in the historical preparations used to treat diabetes in Sri Lankan Siddha Medicine (Sathasivampillai et al., 2017) but only five healers in this work consider it to be a useful anti-diabetic therapy.
A total of eight species were recorded previously to treat diabetes in Northern Province. Seven species were reported in Vavuniya and only Scoparia dulcis was recorded in Jaffna (Rajamanoharan, 2014). Species such as Momordica charantia and G. sylvestre were reported in both Eastern and Northern Provinces. Also, species such as Andrographis paniculata and Toddalia asiatica had been only confirmed in Eastern Province. Interestingly, the most frequently mentioned species (S. cumini) in this study and cited in historical documents (S. auriculata) had not been recorded in Northern Province (Rajamanoharan, 2014, 2016; Sathasivampillai et al., 2017).
Side effects and toxicity studies of plant extracts and herbal preparations play a very important role in assessing the safety and efficacy of drugs purposes. Some species are clearly toxic, like Aristolochia bracteolata (cf. Michl et al., 2016) and their use cannot be endorsed. More complex is the situation with regards to potential herb-drug interactions. However, a detailed analysis of such interactions would be limited by the evidence for specific interactions, for example, with the multiple steps of the detoxification system of xenobiotics that could be affected and the specific composition of the preparation which might be active. This therefore, should be the focus of specific studies of the most important plants using well-characterized extracts.
Levels of scientific evidence of reported species
Based on the Web of Science electronic database of the pharmacological evidence linked to anti-diabetic activities of the species were assessed [excluding 39 (44%) very well studied and globally distributed species, marked with an “
*” in Table
2]. The levels of scientific evidence were established, including information on the plant part used, active extract or compound, model, dose, and duration for the remaining 49 (56%) species (
Appendix). Four levels of scientific evidence were established:
There is no reported bioscientific evidence
In vitro evidence only
In vivo evidence and active compound identified and
Clinical evidence and active compound identified.
Species with no bioscientific evidence
There was no scientific evidence for anti-diabetic activity found for 13 species (27%). Hence, Anti-diabetic and toxicity studies should be carried out in the future focusing on the most frequently used species such as Limonia acidissima, Crateva adansonii, Evolvulus nummularius, P. murex, and Mesua ferrea.
In vitro evidence reported
Only for Mukia maderaspatana and Setaria italica in vitro bioassays form the basis for as an evidence base. Ethanol extract of S. italica seeds showed inhibitory activity in the α-glucosidase inhibition assay with an IC50 1.1 to 1.4 μg/ml (Kim et al., 2011). The active compounds have so far not been isolated from either of these species calling for further phytochemical studies, especially for S. italica (foxtail millet) seeds since these had higher α-glucosidase inhibitory activity at a low dose (IC50 1.1 to 1.4 μg/ml). This species would also be of particular interest because of its wide distribution and use as a (specialist) food.
In vivo evidence and active compound found
The majority (29 out of 49, 59%) have in vivo evidence including Coccinia grandis, Sesbania grandiflora, Cardiospermum halicacabum, Thespesia populnea, and Coscinium fenestratum. Compounds with anti-diabetic activity have been isolated from eight species (Eclipta prostrata, Cheilocostus speciosus, S. auriculata, F. benghalensis, Myristica fragrans, S. cumini, Averrhoa carambola, and S. dulcis) studied in in vivo models. Costunolide (5 mg/kg) isolated from C. speciosus roots was orally administered to Streptozotocin induced diabetic rats daily for 30 days. This treatment significantly reduced blood glucose concentration (Eliza et al., 2009b). In another study 2-(3-acetoxy-4,4,14-trimethylandrost- 8-en-17-yl) (5 mg/kg) identified in S. auriculata flowers orally administered to Alloxan induced diabetic rats for 15 days decreased elevated blood glucose levels (Venkatachalam et al., 2013). Further phytochemical, in vivo, clinical, and toxicity studies should be carried out to identify the active compounds and further evaluate the anti-diabetic activities of the species with reported in vivo evidence. Species such as Borassus flabellifer, Alternanthera sessilis, Ipomoea aquatica, Senna sophera, and Chrysopogon zizanioides are potential candidates for clinical studies.
Clinical evidence and active compound found
Clinical evidence is available for 5 out of 49 species (10%): Cyanthillium cinereum, S. reticulata, Artocarpus heterophyllus, Eleusine coracana, and F. racemosa. So far anti-diabetic compounds have only been isolated from F. racemosa, but these compounds have not been studied in clinical trials. F. racemosa bark water extract (1.2 g/d) orally administered to type 2 diabetic patients (18 men and 12 women) for 1 month showed 15% of reduction of fasting blood glucose levels and 22% of reduction of postprandial blood glucose levels (Ahmed et al., 2011). However, the authors did not state whether this clinical trial was a controlled trail or not. In another study F. racemosa bark extract (100 mg twice a day) was administered orally to diabetics (25 male and 25 female) for 15 days and reduced serum glucose concentrations (Gul-e-Rana et al., 2013).
Conclusion
This is the first ethnobotanical study of plants used to treat diabetes by Siddha healers in the Eastern Province in Sri Lanka. This study aimed at documenting and comparing the current ethnobotanical knowledge from the Siddha healers linked to the treatment of diabetes with plant species recorded in the historical documents and in the few other studies which exist on Siddha medicine. Overall, the bioscientific evidence is limited and priority should be given to the most widely used species. While we excluded globally distributed species, some of them have a better evidence base in terms of safety and pharmacology / clinical effectiveness, and thus they may be better suited in primary health care projects. With this medical tradition's importance also as an element of primary health care, scientific evidence is needed first and foremost on the Siddha medicines' safety and lack of toxicity [e.g., Aristolochia species like A. bracteolate clearly not be endorsed as a phytomedicine (Michl et al., 2016)]. This study demonstrates that there is a wealth of knowledge among Siddha healers about managing diabetes, and that there is an urgent need for more studies providing a better evidence-base for these uses. In many cases the chemistry of the species is known relatively well and thus further bibliographic assessments can form a starting point for such an assessment. In further steps observational and ideally intervention studies are essential.
Ethnobotanical surveys should be carried out in the other regions of Sri Lanka for documenting the useful species utilized by Siddha healers before this knowledge may disappear in the future. Importantly, as many diabetics are currently taking combination of herbal preparations with biomedical medications such as Metformin, ethnobotanical surveys should be carried out with diabetics to create awareness of potential herb-drug interactions, side effects and complications caused by taking both biomedical and traditional medicinal preparations.
Furthermore, the Sri Lankan government also needs to advice traditional healers, biomedical doctors, and the public on the species safety and potential uses. Also, potential interactions between “Western” and traditional medicinal preparations need to be assessed and then should communicated to a wider public. Last but not least, this work provides new opportunities to discover novel compounds which could be used as active compounds in future drug discovery.
Statements
Author contributions
The first author SS has contributed 50% and the rest 50% have been equally contributed by both authors PR and MH to this work.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2018.01022/full#supplementary-material
References
1
AdisakwattanaS.PudhomK.Yibchok-AnunS. (2009). Influence of the methanolic extract from Abutilon indicum leaves in normal and streptozotocin-induced diabetic rats. Afr. J. Biotechnol.8, 2011–2015. 10.5897/AJB08.987
2
AgrawalR.SethiyaN. K.MishraS. H. (2013). Antidiabetic activity of alkaloids of Aerva lanata roots on streptozotocin-nicotinamide induced type-II diabetes in rats. Pharm. Biol.51, 635–642. 10.3109/13880209.2012.761244
3
AhmedF.HudedaS.UroojA. (2011). Antihyperglycemic activity of Ficus racemosa bark extract in type 2 diabetic individuals. J. Diabetes3, 318–319. 10.1111/j.1753-0407.2011.00152.x
4
AhmedF.UroojA. (2010). Effect of Ficus racemosa stem bark on the activities of carbohydrate hydrolyzing enzymes: an in vitro study. Pharm. Biol.48, 518–523. 10.3109/13880200903190993
5
AjiboyeB. O.OjoO. A.AdeyonuO.ImiereO.OlayideI.FadakaA.et al. (2016). Inhibitory effect on key enzymes relevant to acute type-2 diabetes and antioxidative activity of ethanolic extract of Artocarpus heterophyllus stem bark. J. Acute Dis.5, 423–429. 10.1016/j.joad.2016.08.011
6
AkhtarM. S.IqbalJ. (1991). Evaluation of the hypoglycaemic effect of Achyranthes aspera in normal and alloxan-diabetic rabbits. J. Ethnopharmacol.31, 49–57. 10.1016/0378-8741(91)90143-2
7
AlrheamA. I. A. A.Saad-Al ShehriZ. (2015). Ethanopharmacologicalstudy of the aqueous, chloroform, ethanol leaves extracts and latex of Calotropis procera in diabetic rats. Biomed Res. Ther.2, 396–401. 10.7603/s40730-015-0027-8
8
American Herbal Pharmacopoeia (2017). Botanical Reference Standards. Available online at: http://www.herbal-ahp.org/order_online.htm (Accessed January 4, 2018).
9
Andrade-CettoA.HeinrichM. (2005). Mexican plants with hypoglycemic effect used in the treatment of diabetes. J. Ethnopharmacol.99, 325–348. 10.1016/j.jep.2005.04.019
10
Anonymous (2000). Seharaasasehara Treatment (
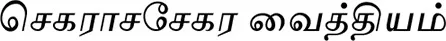 -Seharaasasehara Vaiththiyam). Jaffna: Provincial Department of Indigenous Medicine, Ministry of Health Northern and Eastern Provinces.
-Seharaasasehara Vaiththiyam). Jaffna: Provincial Department of Indigenous Medicine, Ministry of Health Northern and Eastern Provinces.11
Anonymous (2003). Pararasaseharam (Fifth Part)(
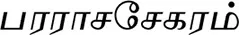
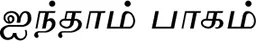 ) - Pararaasaseharam (Ainthaam Paaham). Jaffna: Niyanthree Publication.
) - Pararaasaseharam (Ainthaam Paaham). Jaffna: Niyanthree Publication.12
ArulmozhiD. K.KurianR.VeeranjaneyuluA.BodhankarS. L. (2007). Antidiabetic and antihyperlipidemic effects of Myristica fragrans in animal models. Pharm. Biol.45, 64–68. 10.1080/13880200601028339
13
AttanayakeA. P.JayatilakaK.PathiranaC.MudduwaL. K. B. (2015). Antihyperglycemic activity of Coccinia grandis (L.) Voigt in streptozotocin induced diabetic rats. Indian J. Tradit. Know. 14, 376–381.
14
AYUSH (Department of Ayurveda, Yoga and Naturopathy, Unani, Siddha and Homoeopathy). (2016). Introduction and Origin of Siddha Medicine. Available online at: http://ayush.gov.in/about-the-systems/siddha/introduction-and-origin (Accessed February 4, 2018).
15
BalasubramanianT.LalM. S.SarkarM.ChatterjeeT. K. (2009). Antihyperglycemic and antioxidant activities of medicinal plant Stereospermum suaveolens in streptozotocin-induced diabetic rats. J. Diet Suppl.6, 227–251. 10.1080/19390210903070780
16
BelhekarS. N.ChaudhariP. D.SaryawanshiJ. S.MaliK. K.PandhareR. B. (2013). Antidiabetic and antihyperlipidemic effects of Thespesia populnea fruit pulp extracts on alloxan-induced diabetic rats. Indian J. Pharm. Sci.75, 217–221. 10.4103/0250-474X.115491
17
BrendlerT. (2010). African Herbal Pharmacopoeia. Port Louis: Association for African Medicinal Plants Standards.
18
CazarolliL. H.KappelV. D.PereiraD. F.MorescoH. H.BrighenteI. M. C.PizzolattiM. G.et al. (2012). Anti-hyperglycemic action of apigenin-6-C-β-fucopyranoside from Averrhoa carambola. Fitoterapia83, 1176–1183. 10.1016/j.fitote.2012.07.003
19
ChackrewarthyS.ThabrewM. I.WeerasuriyaM. K. B.JayasekeraS. (2010). Evaluation of the hypoglycemic and hypolipidemic effects of an ethyl acetate fraction of Artocarpus heterophyllus (jak) leaves in streptozotocin-induced diabetic rats. Pharmacogn. Mag.6, 186–190. 10.4103/0973-1296.66933
20
CherianS.AugustiK. T. (1993). Antidiabetic effects of a glycoside of leucopelargonidin isolated from Ficus bengalensis Linn. Indian J. Exp. Biol.31, 26–29.
21
CherianS.AugustiK. T. (1995). Insulin sparing action of leucopelargonin derivative isolated from Ficus bengalensis Linn. Indian J. Exp. Biol.33, 608–611.
22
DavidsD.GibsonD.JohnsonQ. (2016). Ethnobotanical survey of medicinal plants used to manage high blood pressure and type 2 diabetes mellitus in bitterfontein, Western Cape Province, South Africa. J. Ethnopharmacol. 194, 755–766. 10.1016/j.jep.2016.10.063
23
DebnathT.RadhakrishnanR.MurugananthanG.TalwarS. K. N. (2013). Hypoglycaemic effects of alcoholic root extract of Borassus flabellifer (Linn.) in normal and diabetic rats. Pak. J. Pharm. Sci.26, 673–679.
24
Department of Census and Statistics of Sri Lanka (2017). Agriculture and Environment Statistics Division. Available online at: http://www.statistics.gov.lk/agriculture/index.htm (Accessed December 14, 2017).
25
Department of Census and Statistics Sri Lanka (2012). Population and Housing. Available online at: http://www.statistics.gov.lk/page.asp?page=Population%20and%20Housing (Accessed April 29, 2018).
26
Department of Census and Statistics Sri Lanka (2017). Population and Housing. Available online at: http://www.statistics.gov.lk/page.asp?page=Population%20and%20Housing (Accessed April 29, 2018).
27
Department of Meteorology Sri Lanka (2016). Climate of Sri Lanka. Available online at: http://www.meteo.gov.lk/index.php?option=com_content&view=article&id=94&Itemid=310&lang=en (Accessed April 29, 2018).
28
DeshmukhT.YadavB. V.BadoleS. L.BodhankarS. L.DhaneshwarS. R. (2008). Antihyperglycaemic activity of alcoholic extract of Aerva lanata (L.) AL Juss. Ex JA Schultes leaves in alloxan induced diabetic mice. J. Appl. Biomed.6, 81–87.
29
DurmuşkahyaC.ÖztürkM. (2013). Ethnobotanical survey of medicinal plants used for the treatment of diabetes in Manisa, Turkey. Sains Malays.42, 1431–1438. 10.2174/2210289201304010288
30
ElizaJ.DaisyP.IgnacimuthuS.DuraipandiyanV. (2009b). Normo-glycemic and hypolipidemic effect of costunolide isolated from Costus speciosus (Koen ex. Retz.)Sm. in streptozotocin-induced diabetic rats. Chem. Biol. Interact. 179, 329–334. 10.1016/j.cbi.2008.10.017
31
European Medicines Agency (2018). Committee on Herbal Medicinal Products (HMPC). Available online at: http://www.ema.europa.eu/ema/index.jsp?curl=pages/about_us/general/general_content_000264.jsp (Accessed February 4, 2018).
32
FabricantD. S.FarnsworthN. R. (2001). The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect.109, 69–75. 10.1289/ehp.01109s169
33
FarswanM.MazumderP. M.ParchaV.UpaganlawarA. (2009). Modulatory effect of Syzygium cumini seeds and its isolated compound on biochemical parameters in diabetic rats. Phcog. Mag.5:127.
34
Forest Department Sri Lanka (2017). Forest Cover. Available online at: http://www.forestdept.gov.lk/index.php/en/# (Accessed November 14, 2017).
35
GandhiG. R.SasikumarP. (2012). Antidiabetic effect of Merremia emarginata Burm. F. in streptozotocin induced diabetic rats. Asian Pac. J. Trop. Biomed.2, 281–286. 10.1016/S2221-1691(12)60023-9
36
Gul-e-Rana.KarimS.KhurhsidR.Saeed-ul-HassanS.TariqI.SultanaM.et al. (2013). Hypoglycemic activity of Ficus racemosa bark in combination with oral hypoglycemic drug in diabetic human. Acta Pol. Pharm.70, 1045–1049.
37
GuoJ.HanJ.WangS.LiuJ. (2017). An ethnobotanical survey of medicinal plants used for the ailment of diabetes mellitus in Changzhi city of Shanxi Province, China. Biomed. Res.28, 1370–1377.
38
GuptaS.SharmaS. B.PrabhuK. M. (2009a). Ameliorative effect of Cassia auriculata L. leaf extract on glycemic control and atherogenic lipid status in alloxan-induced diabetic rabbits. Indian J. Exp. Biol.47, 974–980.
39
GuptaS.SharmaS. B.PrabhuK. M.BansalS. K. (2009b). Protective role of Cassia auriculata leaf extract on hyperglycemia-induced oxidative stress and its safety evaluation. Indian J. Biochem. Biophys.46, 371–377.
40
HasanN.ShirinF.KhanM. A. J.MamunM. A.BelalM. H.HasanM. M.et al. (2017). Hypoglycemic, hypolipidemic and antibacterial activity of Ficus racemosa fruit extract. Br. J. Pharm. Res.16, 1–9. 10.9734/BJPR/2017/33056
41
HatapakkiB. C.SureshH. M.BhoomannavarV.ShivkumarS. I. (2005). Effect of Cassia auriculata Linn flowers against alloxan-induced diabetes in rats. J. Nat. Rem.5, 132–136. 10.18311/jnr/2005/398
42
HeinrichM.LardosA.LeontiM.WeckerleC.WillcoxM.ConSEFS Advisory Groupet al. (2018). Best practice in research: Consensus Statement on Ethnopharmacological Field Studies-ConSEFS. J. Ethnopharmacol.211, 329–339. 10.1016/j.jep.2017.08.015
43
IDF (International Diabetes Federation) (2015). Diabetes Atlas. Available online at: https://www.idf.org/e-library/epidemiology-research/diabetes-atlas/13-diabetes-atlas-seventh-edition.html (Accessed April 29, 2018).
44
ImR.ManoH.MatsuuraT.NakataniS.ShimizuJ.WadaM. (2009). Mechanisms of blood glucose-lowering effect of aqueous extract from stems of Kothala himbutu (Salacia reticulata) in the mouse. J. Ethnopharmacol.121, 234–240. 10.1016/j.jep.2008.10.026
45
JahanI. A.NaharN.MosihuzzamanM.RokeyaB.AliL.Azad KhanA. K.et al. (2009). Hypoglycaemic and antioxidant activities of Ficus racemosa Linn. fruits. Nat. Prod. Res.23, 399–408. 10.1080/14786410802230757
46
JuvekarA. R.HaladeG. V. (2006). Hypoglycemic activity of Cassia auriculata in neonatal streptozotocin-induced non-insulin dependent diabetes mellitus in rats. J. Nat. Rem.6, 14–18. 10.18311/jnr/2006/273
47
KarA.ChoudharyB. K.BandyopadhyayN. G. (2003). Comparative evaluation of hypoglycaemic activity of some Indian medicinal plants in alloxan diabetic rats. J. Ethnopharmacol.84, 105–108. 10.1016/S0378-8741(02)00144-7
48
KarthicK.KirthiramK. S.SadasivamS.ThayumanavanB. (2008). Identification of alpha amylase inhibitors from Syzygium cumini Linn seeds. Indian J. Exp. Biol.46, 677–680.
49
KazeemM. I.MayakiA. M.OgungbeB. F.OjekaleA. B. (2016). In vitro studies on Calotropis procera leaf extracts as inhibitors of key enzymes linked to diabetes mellitus. Iran J. Pharm. Res.15, 37–44.
50
KeshariA. K.KumarG.KushwahaP. S.BhardwajM.KumarP.RawatA.et al. (2016). Isolated flavonoids from Ficus racemosa stem bark possess antidiabetic, hypolipidemic and protective effects in albino Wistar rats. J. Ethnopharmacol.181, 252–262. 10.1016/j.jep.2016.02.004
51
KimJ. S.HyunT. K.KimM. J. (2011). The inhibitory effects of ethanol extracts from sorghum, foxtail millet and proso millet on α-glucosidase and α-amylase activities. Food Chem.124, 1647–1651. 10.1016/j.foodchem.2010.08.020
52
KotowarooM. I.MahomoodallyM. F.Gurib-FakimA.SubrattyA. H. (2006). Screening of traditional antidiabetic medicinal plants of Mauritius for possible alpha-amylase inhibitory effects in vitro. Phytother. Res.20, 228–231. 10.1002/ptr.1839
53
KrisanapunC.LeeS.-H.PeungvichaP.TemsiririrkkulR.BaekS. J. (2011). Antidiabetic activities of Abutilon indicum (L.) sweet are mediated by enhancement of adipocyte differentiation and activation of the GLUT1 promoter. J. Evid. Based Complement. Altern. Med.2011:167684. 10.1093/ecam/neq004
54
KumarA.IlavarasanR.DeecaramanM.AravindanP.PadmanabhanN.KrishanM. R. V. (2013). Anti-diabetic activity of Syzygium cumini and its isolated compound against streptozotocin-induced diabetic rats. J. Med. Plants Res.2, 246–249. 10.5897/JMPR
55
KumarV. L.PadhyB. M. (2011). Protective effect of aqueous suspension of dried latex of Calotropis procera against oxidative stress and renal damage in diabetic rats. Biocell35, 63–69.
56
KushwahaP. S.RajV.SinghA. K.KeshariA. K.SarafS. A.MandalS. C.et al. (2015). Antidiabetic effects of isolated sterols from Ficus racemosa leaves. RSC Adv.5, 35230–35237. 10.1039/C5RA00790A
57
LathaM.PariL. (2003). Antihyperglycaemic effect of Cassia auriculata in experimental diabetes and its effects on key metabolic enzymes involved in carbohydrate metabolism. Clin. Exp. Pharmacol. Physiol.30, 38–43. 10.1046/j.1440-1681.2003.03785.x
58
LathaM.PariL. (2004). Effect of an aqueous extract of Scoparia dulcis on blood glucose, plasma insulin and some polyol pathway enzymes in experimental rat diabetes. Braz. J. Med. Biol. Res.37, 577–586. 10.1590/S0100-879X2004000400015
59
LathaM.PariL.RamkumarK. M.RajaguruP.SureshT.DhanabalT.et al. (2009). Antidiabetic effects of scoparic acid D isolated from Scoparia dulcis in rats with streptozotocin-induced diabetes. Nat. Prod. Res.23, 1528–1540. 10.1080/14786410902726126
60
LathaM.PariL.SitasawadS.BhondeR. (2004). Scoparia dulcis, a traditional antidiabetic plant, protects against streptozotocin induced oxidative stress and apoptosis in vitro and in vivo. J. Biochem. Mol. Toxicol.18, 261–272. 10.1002/jbt.20035
61
LeontiM. (2011). The future is written: Impact of scripts on the cognition, selection, knowledge and transmission of medicinal plant use and its implications for ethnobotany and ethnopharmacology. J. Ethnopharmacol.134, 542–555. 10.1016/j.jep.2011.01.017
62
LiJ.WeiX.XieQ.Hoa PhamT. T.WeiJ.HeP.et al. (2016). Protective effects of 2-dodecyl-6-methoxycyclohexa-2,5 -diene-1,4-dione isolated from Averrhoa Carambola L. (Oxalidaceae) roots on high-fat diet-induced obesity and insulin resistance in mice. Cell. Physiol. Biochem.40, 993–1004. 10.1159/000453156
63
MalalavidhaneT. S.WickramasingheS. M. D. N.PereraM. S. A.JanszE. R. (2003). Oral hypoglycaemic activity of Ipomoea aquatica in streptozotocin-induced, diabetic wistar rats and Type II diabetics. Phytother. Res.17, 1098–1100. 10.1002/ptr.1345
64
MichlJ.KiteG. C.WankeS.ZierauO.VollmerG.NeinhuisC.et al. (2016). LC-MS- and (1)H NMR-based metabolomic analysis and in vitro toxicological assessment of 43 aristolochia species. J. Nat. Prod.79, 30–37. 10.1021/acs.jnatprod.5b00556
65
MishraM. R.MishraA.PradhanD. K.PandaA. K.BeheraR. K.JhaS. (2013). Antidiabetic and antioxidant activity of Scoparia dulcis Linn. Indian J. Pharm. Sci.75, 610–614. 10.4103/0250-474X.122887
66
NarayanaswamiV. (1975). Introduction to the Siddha System of Medicine. Chennai: A. Anandakumar, Pandit S. S. Anandam Research Institute of Siddha Medicine.
67
NatarajanV.Arul Gnana DhasA. S. (2013). Effect of active fraction isolated from the leaf extract of Dregea volubilis [Linn.] Benth. on plasma glucose concentration and lipid profile in streptozotocin-induced diabetic rats. Springerplus2:394. 10.1186/2193-1801-2-394
68
NearingM. (1985). The green pharmacy. Herbal medicines in modern usage. IDRC Rep.14, 10–11.
69
NetoL.MárioC.de VasconcelosC. F.ThijanV. N.CaldasG. F.AraújoA. V.et al. (2013). Evaluation of antihyperglycaemic activity of Calotropis procera leaves extract on streptozotocin-induced diabetes in Wistar rats. Rev. Bras. Farmacogn.23, 913–919. 10.1590/S0102-695X2013000600008
70
NIS (National Institute of Siddha, Chennai, India) (2018). About Siddha Medicine. Available online at: http://nischennai.org/siddhamedicine.html (Accessed February 4, 2018).
71
OkonkwoC. C.AguC. V.NjokuO. U.AbonyiU.VictorA.AnaduakaE. G.et al. (2015). Hypoglycaemic and haematinic properties of ethanol leaf extract of Artocarpus heterophyllus in alloxan induced diabetic rats. Afr. J. Tradit. Complement. Altern. Med.12, 144–148. 10.4314/ajtcam.v12i2.21
72
OliveiraA. C. P.EndringerD. C.AmorimL. A. S.das Graças L BrandãoM.CoelhoM. M. (2005). Effect of the extracts and fractions of Baccharis trimera and Syzygium cumini on glycaemia of diabetic and non-diabetic mice. J. Ethnopharmacol.102, 465–469. 10.1016/j.jep.2005.06.025
73
PanditR.PhadkeA.JagtapA. (2010). Antidiabetic effect of Ficus religiosa extract in streptozotocin-induced diabetic rats. J. Ethnopharmacol.128, 462–466. 10.1016/j.jep.2010.01.025
74
PanigrahiG.PandaC.PatraA. (2016). Extract of Sesbania grandiflora ameliorates hyperglycemia in high fat diet-streptozotocin induced experimental diabetes mellitus. Scientifica2016:4083568. 10.1155/2016/4083568
75
PariL.Amarnath SatheeshM. (2004). Antidiabetic effect of Boerhavia diffusa: effect on serum and tissue lipids in experimental diabetes. J. Med. Food7, 472–476. 10.1089/jmf.2004.7.472
76
PariL.LathaM. (2005). Antihyperglycaemic effect of Scoparia dulcis.: effect on key metabolic enzymes of carbohydrate metabolism in streptozotocin-induced diabetes. Pharm. Biol.42, 570–576. 10.1080/13880200490901799
77
PariL.LathaM. (2006). Antihyperlipidemic effect of Scoparia dulcis (sweet broomweed) in streptozotocin diabetic rats. J. Med. Food9, 102–107. 10.1089/jmf.2006.9.102
78
PariL.VenkateswaranS. (2002). Hypoglycaemic activity of Scoparia dulcis L. extract in alloxan induced hyperglycaemic rats. Phytother. Res.16, 662–664. 10.1002/ptr.1036
79
PereraP. K. (2012). Current scenario of herbal medicine in Sri Lanka, in Conference proceeding, ASSOCHAM, 4th annual Herbal International Summit cum Exhibition on Medicinal & Aromatic Products, Spices and Finished Products (HIMAPS), New Delhi: NSIC, Okhla Industrial Estate.
80
PhamH. T. T.HuangW.HanC.LiJ.XieQ.WeiJ.et al. (2017). Effects of Averrhoa carambola L. (Oxalidaceae) juice mediated on hyperglycemia, hyperlipidemia, and its influence on regulatory protein expression in the injured kidneys of streptozotocin-induced diabetic mice. Am. J. Transl. Res.9, 36–49.
81
PunithaI. S. R.RajendranK.ShirwaikarA.ShirwaikarA. (2005). Alcoholic stem extract of Coscinium fenestratum regulates carbohydrate metabolism and improves antioxidant status in streptozotocin-nicotinamide induced diabetic rats. Evid. Based Complement. Alternat. Med.2, 375–381. 10.1093/ecam/neh099
82
RafiqM.MitraS. K. (2010). Purified methanolic extract of Salacia reticulata Wight. Ameliorates insulin resistance and metabolic alterations in rats fed high fructose diet. Int. J. Pharmacol. 6, 386–392. 10.3923/ijp.2010.386.392
83
RahmanM. S.RahmanM. Z.BegumB.ChowdhuryR.IslamS. N.RashidM. A. (2011). Antidiabetic principle from Eclipta prostrata. Latin Am. J. Pharm.30, 1656–1660.
84
RajamanoharanP. R. S. (2014). An ethnobotanical survey of medicinal plants in Sillalai, Jaffna, Northern Province, Sri Lanka. Int. J. Herb. Med.1, 22–30.
85
RajamanoharanP. R. S. (2016). Existence and survey of medicinal plants of thiru palani murugan kovil, Asikulam, Vavuniya District, Northern Province, Sri Lanka. World J. Pharm. Res.5, 161–182. 10.20959/wjpr201611-6702
86
RajeshR.ChitraK.PaarakhP. M. (2012). Antihyperglycemic and antihyperlipidemic activity of aerial parts of Aerva lanata Linn Juss in streptozotocin induced diabetic rats. Asian Pac. J. Trop. Biomed.2, S924–S929. 10.1016/S2221-1691(12)60338-4
87
RajuM. G.ReddyT. H. S. (2017). Anti-diabetic and hypolipidemic activity of methanolic extract of Aristolochia bracteolata on streptozotocin induced diabetic rat model. Int. J. Pharm. Sci. Res.8, 1173–1177. 10.13040/IJPSR.0975-8232.8(3).1173-77
88
RiyaM. P.AntuK. A.PalS.ChandrakanthK. C.AnilkumarK. S.TamrakarA. K.et al. (2015). Antidiabetic property of Aerva lanata (L.) Juss. ex Schult. is mediated by inhibition of alpha glucosidase, protein glycation and stimulation of adipogenesis. J. Diabetes7, 548–561. 10.1111/1753-0407.12216
89
RoyS.SehgalR.PadhyB. M.KumarV. L. (2005). Antioxidant and protective effect of latex of Calotropis procera against alloxan-induced diabetes in rats. J. Ethnopharmacol.102, 470–473. 10.1016/j.jep.2005.06.026
90
Royal Botanic Gardens Kew (2018). Medicinal Plant Names Services. Available online at: https://www.kew.org/science/data-and-resources/tools-and-services/medicinal-plant-names-services (Accessed January 4, 2018).
91
SabuM. C.SubburajuT. (2002). Effect of Cassia auriculata Linn. on serum glucose level, glucose utilization by isolated rat hemidiaphragm. J. Ethnopharmacol.80, 203–206. 10.1016/S0378-8741(02)00026-0
92
Salihu ShinkafiT.BelloL.Wara HassanS.AliS. (2015). An ethnobotanical survey of antidiabetic plants used by Hausa-Fulani tribes in Sokoto, Northwest Nigeria. J. Ethnopharmacol.172, 91–99. 10.1016/j.jep.2015.06.014
93
SathasivampillaiS. V.RajamanoharanP. R. S.MundayM.HeinrichM. (2017). Plants used to treat diabetes in Sri Lankan Siddha Medicine-An ethnopharmacological review of historical and modern sources. J. Ethnopharmacol.198, 531–599. 10.1016/j.jep.2016.07.053
94
SchoenfelderT.WarmlinC. Z.ManfrediniM. S.PaveiL. L.RéusJ. V.TristãoT. C.et al. (2010). Hypoglycemic and hypolipidemic effect of leaves from Syzygium cumini (L.) Skeels, Myrtaceae. in diabetic rats. Rev. Bras. Farmacogn.20, 222–227. 10.1590/S0102-695X2010000200014
95
SenaratnaL. K. (2001). A Check List of the Flowering Plants of Sri Lanka. Colombo: National Science Foundation of Sri Lanka.
96
ShahreenS.BanikJ.HafizA.RahmanS.ZamanA. T.ShoyebM. A.et al. (2012). Antihyperglycemic activities of leaves of three edible fruit plants (Averrhoa carambola, Ficus hispida and Syzygium samarangense) of Bangladesh. Afr. J. Tradit. Complement. Altern. Med.9, 287–291. 10.4314/ajtcam.v9i2.16
97
SharmaS.PathakS.GuptaG.SharmaS. K.SinghL.SharmaR. K.et al. (2017). Pharmacological evaluation of aqueous extract of Syzigium cumini for its antihyperglycemic and antidyslipidemic properties in diabetic rats fed a high cholesterol diet-Role of PPARγ and PPARα. Biomed. Pharmacother.89, 447–453. 10.1016/j.biopha.2017.02.048
98
ShindeJ.TaldoneT.BarlettaM.KunaparajuN.HuB.KumarS.et al. (2008). Alpha-glucosidase inhibitory activity of Syzygium cumini (Linn.) skeels seed kernel in vitro and in Goto-Kakizaki (GK) rats. Carbohydr. Res.343, 1278–1281. 10.1016/j.carres.2008.03.003
99
ShirwaikarA.RajendranK.PunithaI. S. R. (2005). Antidiabetic activity of alcoholic stem extract of Coscinium fenestratum in streptozotocin-nicotinamide induced type 2 diabetic rats. J. Ethnopharmacol.97, 369–374. 10.1016/j.jep.2004.11.034
100
ShobanaS.HarshaM. R.PlatelK.SrinivasanK.MalleshiN. G. (2010). Amelioration of hyperglycaemia and its associated complications by finger millet (Eleusine coracana L.) seed coat matter in streptozotocin-induced diabetic rats. Br. J. Nutr.104, 1787–1795. 10.1017/S0007114510002977
101
SinghN.GuptaM. (2006). Effect of alcoholic extract of Ficus bengalensis (L.) on the pancreatic islets of alloxan diabeticalbino rats. J. Exp. Zool. India9, 275–284.
102
SinghN.GuptaM. (2007). Effects of ethanolic extract of Syzygium cumini (Linn) seed powder on pancreatic islets of alloxan diabetic rats. Indian J. Exp. Biol.45, 861–867.
103
SithamparthanuppillaiS. (1982). Disorder Explanation(
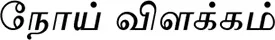 –Noi Vilakkam). Chennai: Siddha Medicine Book Research Centre. [In Tamil]
–Noi Vilakkam). Chennai: Siddha Medicine Book Research Centre. [In Tamil]104
SivashanmugarajahS. (2000). Sri Lankan Siddha Medicine in 20th century(
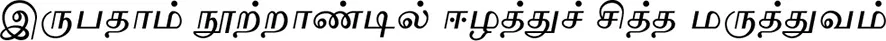 – Irupathaam Nootraandil Eelaththuchchiththa Maruththuvam). Jaffna: Siddha Medicine Development Society [In Tamil].
– Irupathaam Nootraandil Eelaththuchchiththa Maruththuvam). Jaffna: Siddha Medicine Development Society [In Tamil].105
SrilathaB. R.AnandaS. (2014). Antidiabetic effects of Mukia maderaspatana and its phenolics: an in vitro study on gluconeogenesis and glucose uptake in rat tissues. Pharm. Biol.52, 597–602. 10.3109/13880209.2013.858268
106
SugathadasaK. S. S.JeevandaraP. M.DevanarayanaA.PushpakumaraD. (2008). A Checklist of Medicinal Plants in Sri Lanka. Colombo: Bandaranaike Memorial Ayurvedic Research Institute.
107
SuranaS. J.GokhaleS. B.JadhavR. B.SawantR. L.WadekarJ. B. (2008). Antihyperglycemic activity of various fractions of Cassia auriculata Linn. in alloxan diabetic rats. Ind. J. Pharm. Sci.70, 227–229. 10.4103/0250-474X.41461
108
SwamiU.RishiP.SoniS. K. (2017). Antidiabetic, hypolipidemic and hepato-renal protective effect of a novel fermented beverage from Syzygium Cumini Stem. Int. J. Pharm. Sci. Res.8, 1336–1345. 10.13040/IJPSR.0975-8232.8(3).1336-45
109
TalukderF. Z.KhanK. A.UddinR.JahanN.AlamM. A. (2012). In vitro free radical scavenging and anti-hyperglycemic activities of Achyranthes aspera extract in alloxan-induced diabetic mice. Drug Discov Ther. 6, 298–305. 10.5582/ddt.2012.v6.6.298
110
TanK. K.KimK. H. (2013). Alternanthera sessilis red ethyl acetate fraction exhibits antidiabetic potential on obese type 2 diabetic rats. Evid. Based Complement. Alternat. Med.2013:845172. 10.1155/2013/845172
111
The Plant List (2013). The Plant List. Available online at: http://www.theplantlist.org/ (Accessed January 5, 2018).
112
TrinhB. T. D.JägerA. K.StaerkD. (2017). High-resolution inhibition profiling combined with HPLC-HRMS-SPE-NMR for identification of PTP1B inhibitors from vietnamese plants. Molecules22:E1228. 10.3390/molecules22071228
113
TrinhB. T. D.StaerkD.JägerA. K. (2016). Screening for potential α-glucosidase and α-amylase inhibitory constituents from selected Vietnamese plants used to treat type 2 diabetes. J. Ethnopharmacol.186, 189–195. 10.1016/j.jep.2016.03.060
114
UptonR.GraffA.JolliffeG.LängerR.WilliamsonE. (2016). American Herbal Pharmacopoeia: Botanical Pharmacognosy-Microscopic Characterization of Botanical Medicines. Florida, FL: CRC Press.
115
VeeramaniC.PushpavalliG.PugalendiK. V. (2008). Antihyperglycaemic effect of Cardiospermum halicacabum Linn. leaf extract on STZ-induced diabetic rats. J. Appl. Biomed.6, 19–26.
116
VeerapurV. P.PrabhakarK. R.ThippeswamyB. S.BansalP.SrinivasanK. K.UnnikrishnanM. K. (2012). Antidiabetic effect of Ficus racemosa Linn. stem bark in high-fat diet and low-dose streptozotocin-induced type 2 diabetic rats: a mechanistic study. Food Chem.132, 186–193. 10.1016/j.foodchem.2011.10.052
117
VenkatachalamM.SingaraveluG.GovindarajuK.AhnJ. S. (2013). PTP 1B inhibitory action of a phytochemical propanoic acid, 2-(3-acetoxy-4,4,14-trimethylandrost-8-en-17-yl). Curr. Sci.105, 827–832.
118
VetrichelvanT.JegadeesanM. (2002). Anti-diabetic activity of alcoholic extract of Aerva lanata (L.) Juss. ex Schultes in rats. J. Ethnopharmacol.80, 103–107. 10.1016/S0378-8741(01)00412-3
119
VidyasagarG. M.SiddalingaM. S. M. (2013). Medicinal plants used in the treatment of diabetes mellitus in Bellary district, Karnataka. Indian J. Tradit. Know.12:4.
120
VijayakumarM.GovindarajanR.RaoG. M. M.RaoC. V.ShirwaikarA.MehrotraS.et al. (2006). Action of Hygrophila auriculata against streptozotocin-induced oxidative stress. J. Ethnopharmacol.104, 356–361. 10.1016/j.jep.2005.09.030
121
WeragodaP. B. (1980). The traditional system of medicine in Sri Lanka. J. Ethnopharmacol.2, 71–73. 10.1016/0378-8741(80)90033-1
122
WernerW. L.BalasubramaniamS. (1992). Structure and Dynamics of the Upper Montane Rain Forests of Sri Lanka, in Tropical Forests in Transition Advances in Life Sciences, ed GoldammerJ. (Basel: Birkhäuser), 165–172.
123
WHO (World Health Organization) (1999). Monographs on Selected Medicinal Plants, Vol. 1. Available online at: http://apps.who.int/medicinedocs/en/d/Js2200e/ (Accessed October 4, 2017).
124
WHO (World Health Organization) (2004). Monographs on Selected Medicinal Plants, Vol. 2. Available online at: http://apps.who.int/medicinedocs/en/d/Js4927e/ (Accessed October 4, 2017).
125
WHO (World Health Organization) (2007). Monographs on Selected Medicinal Plants, Vol. 3. Available online at: http://apps.who.int/medicinedocs/en/m/abstract/Js14213e/ (Accessed October 4, 2017).
126
WHO (World Health Organization) (2009). Monographs on Selected Medicinal Plants, Vol. 4. Available online at: http://apps.who.int/medicinedocs/en/m/abstract/Js16713e/ (Accessed October 4, 2017).
127
WHO (World Health Organization) (2017). Diabetes. Available online at: http://www.who.int/news-room/fact-sheets/detail/diabetes (Accessed October 4, 2017).
128
YadavS. K.NagoriB. P.DesaiP. K. (2014). Pharmacological characterization of different fractions of Calotropis procera (Asclepiadaceae) in streptozotocin induced experimental model of diabetic neuropathy. J. Ethnopharmacol.152, 349–357. 10.1016/j.jep.2014.01.020
129
YoshinoK.MiyauchiY.KanetakaT.TakagiY.KogaK. (2009). Anti-diabetic activity of a leaf extract prepared from Salacia reticulata in mice. Biosci. Biotechnol. Biochem.73, 1096–1104. 10.1271/bbb.80854
130
YousafS.HussainA.RehmanS.AslamM. S.AbbasZ. (2016). Hypoglycemic and hypolipidemic effects of Lactobacillus fermentum, fruit extracts of Syzygium cumini and Momordica charantia on diabetes induced mice. Pak. J. Pharm. Sci.29, 1535–1540.
Summary
Keywords
Sri Lanka, diabetes mellitus, Tamil medicine, Syzygium cumini, Fabaceae, Eastern Province, ethnobotany, Siddha Medicine
Citation
Sathasivampillai SV, Rajamanoharan PRS and Heinrich M (2018) Siddha Medicine in Eastern Sri Lanka Today–Continuity and Change in the Treatment of Diabetes. Front. Pharmacol. 9:1022. doi: 10.3389/fphar.2018.01022
Received
06 May 2018
Accepted
23 August 2018
Published
10 October 2018
Volume
9 - 2018
Edited by
Adolfo Andrade-Cetto, Universidad Nacional Autónoma de México, Mexico
Reviewed by
Zsuzsanna Hajdú, University of Szeged, Hungary; Michał Tomczyk, Medical University of Bialystok, Poland
Updates
Copyright
© 2018 Sathasivampillai, Rajamanoharan and Heinrich.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Michael Heinrich m.heinrich@ucl.ac.uk
This article was submitted to Ethnopharmacology, a section of the journal Frontiers in Pharmacology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.