- Pengiran Anak Puteri Rashidah Sa’adatul Bolkiah (PAPRSB) Institute of Health Sciences, Universiti Brunei Darussalam, Gadong, Brunei
The human gut is home to trillions of microbes that interact with host cells to influence and contribute to body functions. The number of scientific studies focusing on the gut microbiome has exponentially increased in recent years. Studies investigating factors that may potentially affect the gut microbiome and may be used for therapeutic purposes in diseases where dysbioses in the gut microbiome have been shown are of particular interest. This review compiles current evidence available in the scientific literature on the use of probiotics to treat metabolic diseases and autism spectrum disorders (ASDs) to analyze the efficacy of probiotics in these diseases. To do this, we must first define the healthy gut microbiome before looking at the interplay between the gut microbiome and diseases, and how probiotics affect this interaction. In metabolic diseases, such as obesity and diabetes, probiotic supplementation positively impacts pathological parameters. Conversely, the gut–brain axis significantly impacts neurodevelopmental disorders such as ASDs. However, manipulating the gut microbiome and disease symptoms using probiotics has less pronounced effects on neurodevelopmental diseases. This may be due to a more complex interplay between genetics and the environment in these diseases. In conclusion, the use of microbe-based probiotic therapy may potentially have beneficial effects in ameliorating the pathology of various diseases. Validation of available data for the development of personalized treatment regimens for affected patients is still required.
Introduction
The gut is a natural habitat for trillions of diverse microbes (anaerobic bacteria, yeasts, viruses, and bacteriophages) where the phyla Firmicutes, Bacteroidetes, and Actinobacteria are the most common (1). The gut microbiome is a complex ecosystem where microbes and their metabolites interact with host cells to influence body functions. General health is associated with a “healthy” microbiome, defined by the diversity and types of species of bacteria within the gut.
Fecal microbiome analysis has shown that the gut microbiota composition is influenced by various factors such as age, genetics, types of food consumed, economic development, and immediate environment (2–7). Dysbiosis of the microbiome is associated with a reduction in the diversity of microbes within the gut. The altered diversity of gut microbes is correlated with various diseases such as metabolic diseases, autism spectrum disorders (ASDs), and other brain disorders (8–16). Changes in the microbiome have been also linked with infection risk and susceptibility (17), including COVID-19 (18).
Recently, products containing supposedly “healthy” bacteria are touted as being beneficial to health by restoring balance to the microbiome within the gut. These products are generally termed “probiotics,” but have also generated other related products, all of which are proposed to act to enhance healthy bacteria within the gut. The term “probiotics” was coined in the 1970s while food containing beneficial bacteria have been consumed even earlier. Recently added interest in the commercialization of probiotic foods meant that there has been a need to define what can be claimed as a probiotic.
The generally accepted definition of probiotic was generated together by the Food and Agriculture Organization of the United Nations (FAO) and WHO—“live microorganisms which when administered in adequate amounts confer a health benefit on the host” (19). Other related products include “prebiotics,” “synbiotics,” “postbiotics,” and “metabiotics” (20–23). The term “live and active cultures” is sometimes used for fermented or functional foods with live microorganisms within them but those microorganisms may not prove to be probiotic yet (19).
With the plethora of probiotics and associated products now available commercially, it is no wonder that there have been many misconceptions regarding probiotics, their usage, and their health benefits, which we will address in the subsequent sections of this review.
Constituents of Probiotics
Specific health benefits have been ascribed to particular probiotic strains, and therefore, not all probiotic supplements are equal, even if they list the same species of probiotic bacteria. Therefore, it is essential to ensure that the correct strain is used to treat the underlying clinical issue. Additionally, supplements that contain multiple strains of bacteria may also lack the scientific evidence for the claimed benefits.
The most common bacterial species used in current probiotic products are lactic acid bacteria such as Bifidobacterium and Lactobacillus strains (24). However, recent studies have identified other species of bacteria that may also confer benefits when used as probiotics, such as Akkermansia muciniphila and Faecalibacterium prausnitzii (25), and the use of these bacteria in probiotic products is rising as well (26). The effectiveness of probiotic supplementation can be measured through the bacterial load in the feces, or other measures within the body (27, 28), and is essential to establish the efficacy of any treatment.
Evidence of Therapeutic Effects of Probiotics
The benefits of probiotic supplementation result from either inhibition of pathogen growth in the large intestine or augmented immune response and intestinal barrier function in both small and large intestines (29). As most probiotics are beneficial bacteria found naturally within the gut, ingested probiotics within the gut interact with immune cells to sustain an immunologic balance within the gastrointestinal tract (30, 31). Therefore, the interplay between the gut microbiome, probiotics, and human health is via the modulation of immune responses at the epithelial cells constituting the mucosal interface between host and microorganisms.
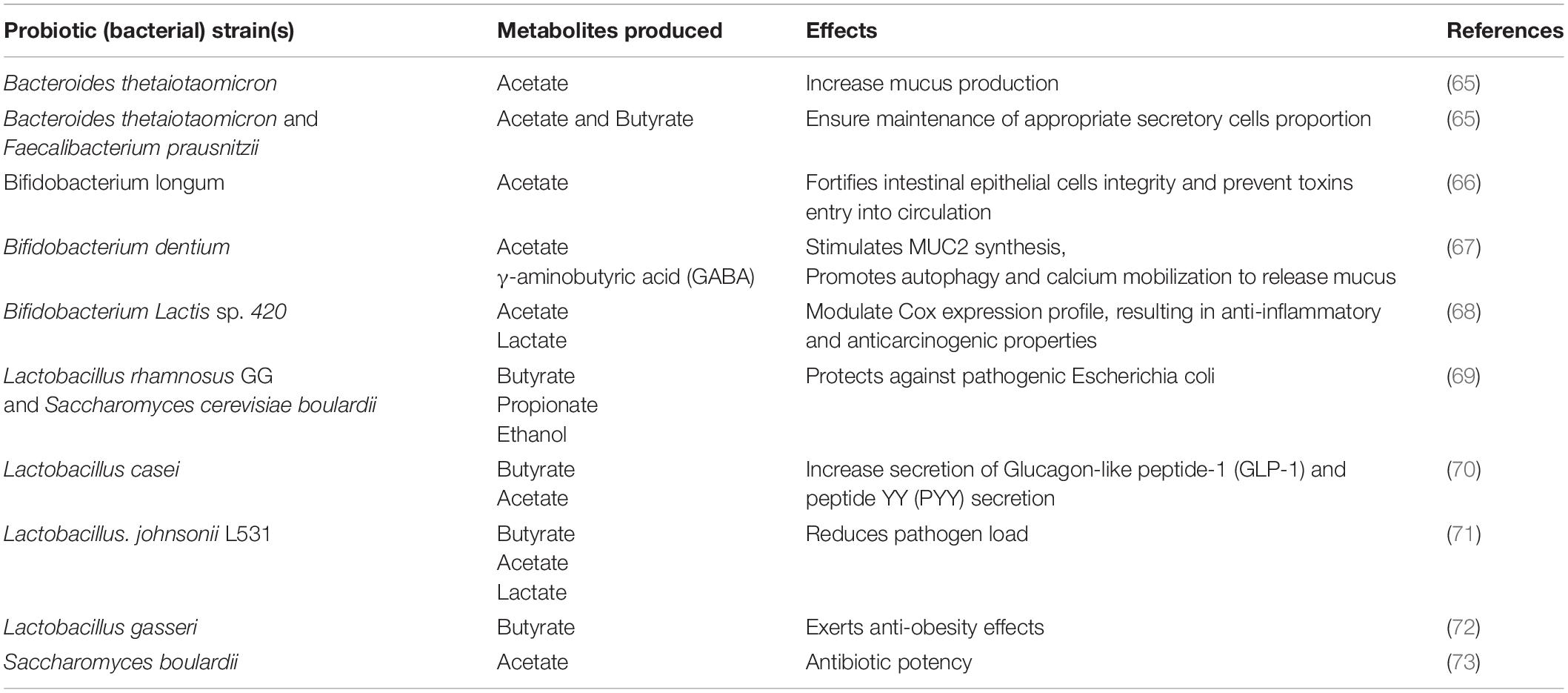
The gut microbiome also produces a wide range of metabolites due to the anaerobic fermentation of undigested materials and endogenous compounds found within the microbes and host. The metabolites produced by the microbiome serve as agents that modulate the host cells’ responses, thus its immune system and disease probability. Rooks and Garrett have reviewed how these metabolites modulate the immune responses and disease risk (32). We have summarized probiotic strains, their resulting metabolites, and their effects on health in Table 1.
Regular consumption of probiotic supplements and foods has ascribed numerous scientifically backed benefits, including effects on the gut such as amelioration of diarrhea and other digestive symptoms (33–38), reduction of inflammation (33, 39), as well as benefits to various conditions ranging from emotional imbalance to autoimmune diseases (40–45). Some groups have even shown the benefits of consuming probiotics for patients with cancer (28, 46, 47). However, it must be noted that while many clinical benefits have been rigorously tested, in many cases, probiotics cannot be considered an alternative to medicine, particularly in severe diseases.
Healthy Gut Microbiome Profiles and Changes in Disease
Knowledge of a healthy gut microbiome is necessary before addressing the diseases triggered by the dysregulation of the gut microbiome. Hou et al. (51) established three enterotypes comprising specific species and functional composition: Bacteroides, Blautia, and Prevotella enterotypes. These different gut microbiome diversity signatures have different risks for different diseases (48–50). Additionally, the efficacy of probiotic supplementation is also affected by enterotype (51). Therefore, these enterotypes may form a basal gut microbiome that is independent of geographical location as well as nutrition.
Gut community profiles have also shown that healthy pre-adolescents have more significant numbers of species and greater diversity than adults, with increased Firmicutes and Actinobacteria (52). Both Bacteroidetes and Firmicutes bacteria are SCFAs producers, specifically acetic acid and propionic acid by Bacteroidetes and butyric acid by Firmicutes (53). Functionally, the diversity of microbial genes detected in the gut microbiome in children was responsible for the ensuing growth and development, such as vitamin synthesis. In contrast, the enriched microbial genes detected in the gut microbiome of adults are associated with inflammation and fat deposition (52). Findings from a further study to understand the gut microbiome of pre-adolescents in different geographical areas and conditions showed that the distal guts of children living in the Bangladeshi slum have significantly higher bacterial gut microbiome diversity with enrichment in Prevotella, Butyrivibrio, and Oscillospira together with a depletion in Bacteroides (54). However, this microbial diversity was more prone to changes, unlike the microbiota found in children living in the suburban community.
A reference profile comprising the abundance and list of microbes in a healthy human was constructed, with 157 organisms classified as healthy gut microbes in the Fecal Biome Population Report (55). Additionally, Kong et al. (56) studied the gut microbiome of healthy centenarians as a benchmark for a healthy microbiome model. They found that short-chain fatty acids (SCFAs)-producing bacteria were more abundant in the long-living Chinese cohort. SCFAs such as butyrate, propionate, and acetate, produced by the gut microbiome, are beneficial for health. SCFAs act by stimulating the expansion of regulatory T cells, inhibiting inflammation via reducing histone deacetylase-9 gene expression (57). Thus, SCFAs maintain the gut barrier’s integrity, stimulate immunity in the intestines, and prevent pathogen infection (32, 58). Hence, metabolites produced by the gut microbiome can also modulate a person’s health status (59).
Interestingly, the follow-up study revealed that the long-living healthy people in the study (both Chinese and Italian cohorts) had more diverse microbiota structures than younger age groups (60). This result contrasts with previous studies whose results have suggested that gut microbiome diversity in a person tends to decrease as the person ages (61, 62). This suggests that the changes in your gut microbiome are not set in stone and can be modulated with environmental factors and diet.
The potential of the dysbiosis of the gut microbiome in the establishment of metabolic diseases should be obvious. However, the gut microbiome is also able to communicate with the nervous system via the gut-brain axis (GBA) and thus affects neurological diseases as well. The GBA involves bidirectional interaction between the central and the enteric nervous systems, connecting the cognitive and emotional centers of the brain with peripheral intestinal functions. Bacteria in the gastrointestinal (GI) tract influence the signaling of neural pathways and the central nervous system (CNS) (63–67). Evidence of microbiota-GBA communications emerged from the association of dysbiosis with central nervous disorders (63, 68, 69). From this, we note that healthy gut microbiota is essential for brain development and function.
Consequently, a healthy gut microbiome is essential for both metabolic and neurological health. In the following sub-sections, we will be addressing the use of probiotics in metabolic diseases (obesity and type II diabetes) and neurodegenerative diseases.
Gut Dysbiosis and Probiotics and Obesity
Obesity is defined by excessive fat accumulation in the body, which may increase the risk of non-communicable diseases such as diabetes, cardiovascular diseases, some cancers, and hypertension (70). The gut microbiome and the composition of dietary intake are profoundly linked (71). For example, the intake of animal-based foods provided up to 5 consecutive days of increased bile-tolerant microbes (Alistipes, Bilophila, and Bacteroides) and reduced the amount of fiber-fermenting bacteria (72).
Gut microbiota profiles in overweight and obese individuals show higher amounts of Bacteroides, Bifidobacteria, Staphylococcus aureus, and Lactobacilli Clostridia (73, 74). Among overweight individuals, the baseline ratio of gut microflora, Firmicutes to Bacteroidetes was disturbed (75). Firmicutes bacteria potentially are able to affect the modulation of gene expression and hormones involved in metabolism (76). Therefore, the change in the ratio of different bacteria species may affect human metabolism, leading to obesity.
Probiotics may act as anti-obesity agents by various modes of action, including modulation of specific gut microbiota strains, gastrointestinal and immune system modulation, lowering insulin resistance, and greater satiety. The use of probiotics containing Lactobacillus and Bifidobacterium species in obesity treatment is promising (77). Some of the positive changes which resulted from the intake of probiotics include lower waist circumference, lower body fat deposition, lower body weight, lower weight gain, and improved lipid profile. However, Vajro et al. showed that L. salivalis supplementation in obese adolescents led to no improvement in obesity parameters (78). Another study with the consumption of one capsule of L. rhamnosus G showed a lower weight gain at 1 year of life and up to 4 years old in children but observed no weight changes after that period (79). This difference in weight gain patterns may be due to the colonization of the gut microflora, which begins during the first few years of life (80, 81). Unless various scientific groups consistently match the age of controls and subjects, together with consistent bacterial strains utilized in probiotics, the conclusion derived from the comparison of these studies remains murky.
A. muciniphila is negatively correlated to obesity development, as well as other diseases such as type-2 diabetes and hypertension (82). A human clinical trial looking at the impact of A. muciniphila supplementation for over 3 months showed that the treatment led to improved insulin sensitivity, insulinemic, and reduction of total cholesterol (83). The evidence of A. muciniphila as a probiotic that confers a protective effect against metabolic disorders has been accumulating over the past few years (84) and may merit further study.
Hence, probiotics positively impact the reduction of relevant obesity parameters, although the effect varies across the different age groups and genders. More standardized studies are needed to investigate how the different mixtures of bacterial species in probiotics affect different age groups and genders.
Gut Dysbiosis, Probiotics, and Diabetes
Type-2 diabetes is a metabolic disorder in which individuals display abnormally high blood glucose, resulting from inadequate insulin secretion and resistance (85). Type 2 diabetes results from the interaction between environmental factors and genetic factors (86). One of the primary risk factors of type-2 diabetes is being overweight or obese (87).
A change in the composition of the gut microbiota may result in increased susceptibility to prediabetic conditions such as insulin resistance (87–89). Reports have revealed that the intestinal microbiome of individuals with type-2 diabetes has reduced butyrate-producing bacteria (87, 90), a lower frequency of Firmicutes, and a higher frequency of Bacteroidetes and Proteobacteria (88). The metabolites produced by gut microbes also affect insulin sensitivity and glucose homeostasis, with metabolites like SCFA improving insulin secretion (91). Therefore, butyrate-producing bacteria affect insulin secretion and therefore, the blood sugar level of a person. Further exploration of the bacterial strain or administration of butyrate may be beneficial to a diabetic.
Probiotic intake, such as Lactobacillus rhamnosus GG, leads to improvement in intestinal integrity, reduced lipopolysaccharide level, reduced endoplasmic reticulum stress, and improved insulin sensitivity (91–93). Animal and clinical trials have shown that both single probiotic strains or mixtures of probiotics have the potential to improve type-2 diabetes parameters (87, 94). More research is required to dissect the most suitable species impacting gut metabolism, as well as exposure time, and dose.
Gut Dysbiosis, Probiotics, and Autism Spectrum Disorder
Autism spectrum disorder is a group of neurodevelopmental disorders defined by deficits in communication and social interaction, and stereotyped behaviors (65). GI abnormalities are common among individuals with ASD (95, 96), with a strong correlation of GI symptoms with ASD severity (97).
The gut microbiota of children with ASD is less diverse, with decreased levels of health-promoting gut bacteria, and an increased abundance of species that produce neurotoxins (65). Metabolites from the gut microbiota may play vital roles in the pathogenesis of ASD (95, 96). Altered fecal SCFAs have been linked to constipation in ASD (97), where lower levels of acetic acid and butyrate and an elevated level of valeric acid have been reported in subjects with ASD (96). It has also been shown that SCFAs can induce autistic-like symptoms upon injection into rats (98).
Maternal immune activation (MIA) mouse models that display features of ASD have altered microbiota and GI barrier defects. Oral treatment of MIA offspring with the human commensal bacteria Bacteroides fragilis improves gut permeability, alters the microbial composition, and corrects behavioral defects in MIA animals. Therefore, it has been proposed that targeting the gut microbiota may be a potential therapy for specific symptoms in ASDs (95).
Probiotics potentially impact gut microbiota communities to alter the levels of harmful metabolites in ASD children, reducing GI inflammation and intestinal permeability (1, 99). However, the results of probiotic supplementation in individuals with ASD remain inconclusive and controversial. Current probiotics are mainly aerobic, short-lived, milk-derived cultures, which are not usually a significant part of the primarily anaerobic human gut microbiome (1). A review based on four studies concluded that current evidence does not support the use of probiotics to modify behavior in patients with ASD (100). Probiotics did not exert a significant effect to restore most of the beneficial bacteria upon assessment of stool samples from 58 individuals with ASD and 39 age-matched typically developing children (97). On the other hand, it has also been reported that probiotics treatment seems to improve ASD-associated behavioral symptoms (101).
Autism spectrum disorder individuals are highly selective eaters (102, 103); therefore dietary factors remain a strong confounding factor for these individuals. The complex interplay between host genetics, environment, and the microbiome although challenging to dissect are important factors to consider. Larger longitudinal trials as well as optimizing dosage, formulation (single vs. multispecies probiotics), timing (101), route of administration as well as toxicity concerns remain to be addressed to validate the efficacy of probiotics for ASD, taking into consideration age and population-specific differences in gut microbiota/metabolites produced (6, 7).
Gut Dysbiosis and Probiotics in Neurodegenerative Diseases
It is well-established that age is a primary risk factor for neurodegenerative diseases due to increased insults including decreased neurotransmitter levels, chronic inflammation, oxidative stress, and apoptosis (104). There is also a high prevalence of GI comorbidities among patients with Parkinson’s and Alzheimer’s diseases (105, 106). Dysbiosis in the intestinal microbiota in the elderly may result in a leaky gut, and subsequently, promote systemic and neuroinflammation (107).
Gut microbiota secretes neurometabolites, which include neurotransmitters that regulate the signaling cascades of the CNS. A comprehensive review of neurotransmitters directly secreted by various probiotics has been published (105). Altered levels of neurotransmitters result in behavioral changes in neurodegenerative diseases. Restoring the balance of neurotransmitters by targeting gut microbiota is therefore central to the management of neurodegenerative disease.
Parkinson’s disease (PD) is characterized by loss of dopaminergic neurons and intraneuronal alpha-synuclein accumulation, in the basal ganglia and at peripheral sites, including the gut (108). GI dysfunction has been reported to be a potential contributor to the pathogenesis of PD with evidence that alpha-synuclein inclusions appear early in the enteric nervous system and travel to the brain via the vagal nerves (109, 110). A review on altered gut microbiota compositions in patients with PD is available (111). Probiotics administration in independent studies improves GI symptoms and the metabolic profile of patients with PD (108, 111, 112).
Alzheimer’s disease (AD) is one of the most common irreversible, neurodegenerative disorders in the elderly, which leads to cognitive decline and dementia. Inflammatory response at the site of beta-amyloid (one of the hallmark features of AD) accumulation in the brain has been linked to the gut microbiota (66). Current studies on the efficacy of probiotics in AD, although limited, seems promising. In a transgenic mice model of Alzheimer’s Disease (AD), modulation of the gut microbiota through exercise and probiotic treatment alleviated the progress of AD (113). Rats injected with probiotics (L. acidophilus, L. fermentum, B. lactis, B. longum) for 8 weeks elicit an improvement in memory deficit and AD-associated pathology (114). However, it remains to be determined whether these findings are replicable in humans. Another randomized, double-blind, and controlled clinical trial among 60 patients with AD revealed that a 12-week probiotic (L. acidophilus, L. casei, B. bifidum, and L. fermentum) consumption improved cognitive function and certain metabolic markers (115). There is also an ongoing clinical trial (randomized, placebo-controlled) to investigate the effect of probiotics on 58 participants with dementia (116). Therefore, the efficacy of probiotics to restore gut dysbiosis in patients with AD awaits further validation.
A key limitation of current probiotic studies for PD and AD is the small sample sizes (n < 100). Consistent study designs in larger human trials with validated safety and efficacy are required before translation into clinical settings.
Future Directions
Manipulation of the gut microbiota and microbial metabolites to address challenging questions in metabolic and brain disorders is difficult due to the complex relationship between host genetics and environmental factors to influence the gut microbiota. A healthy diet and exercise positively modify the gut microbiota (117–119), therefore it remains inevitable to tackle these key modifiable factors to ensure a healthy community of microbes.
Utilizing data from the NIH Human Microbiome Project (HMP) for resources and insights on the human microbiome provides an opportunity to further understand the complex relationship between human health and diseases, which will serve as a pedestal for novel approaches toward the development of therapeutics to tackle relevant diseases. Large scale, harmonized multi-center studies, and freely accessible data are imperative to validate the role of probiotics as potential therapeutics before translating research into clinical practice.
The long-term effects of probiotics and their corresponding metabolites/substances on health are needed to fully understand the mechanisms of each probiotic strain on health (120). Delineation of the precise role and effect of each probiotic strain may just be the beginning of introducing precise probiotic strain for an exact clinical disease. This delineation may be followed by combined efforts of various strains of probiotics. In short, the journey into the gut microbiome is just the tip of the iceberg at the moment.
Author Contributions
IZ provided the concept of the manuscript and finalized the manuscript. All authors wrote, provided revisions to the manuscript, read and approved the final manuscript, reviewed the manuscript, and consented for it to be sent for publication.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Navarro F, Liu Y, Rhoads JM. Can probiotics benefit children with autism spectrum disorders? World J Gastroenterol. (2016) 22:10093–102. doi: 10.3748/wjg.v22.i46.10093
2. Karl PJ, Hatch AM, Arcidiacono SM, Pearce SC, Pantoja-Feliciano IG, Doherty LA, et al. Effects of psychological, environmental and physical stressors on the gut microbiota. Front Microbiol. (2018) 9:2013. doi: 10.3389/fmicb.2018.02013
3. Magrone T, Jirillo E. The interaction between gut microbiota and age-related changes in immune function and inflammation. Immun Ageing. (2013) 10:31. doi: 10.1186/1742-4933-10-31
4. Singh RK, Chang HW, Yan D, Lee KM, Ucmak D, Wong K, et al. Influence of diet on the gut microbiome and implications for human health. J Transl Med. (2017) 15:1–17. doi: 10.1186/s12967-017-1175-y
5. Biesalski HK. Nutrition meets the microbiome: micronutrients and the microbiota. Ann N Y Acad Sci. (2016) 1372:53–64. doi: 10.1111/nyas.13145
6. Odamaki T, Kato K, Sugahara H, Hashikura N, Takahashi S, Xiao J-Z, et al. Age-related changes in gut microbiota composition from newborn to centenarian: a cross-sectional study. BMC Microbiol. (2016) 16:90. doi: 10.1186/s12866-016-0708-5
7. Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, et al. Human gut microbiome viewed across age and geography. Nature. (2012) 486:222–7. doi: 10.1038/nature11053
8. Ai D, Pan H, Han R, Li X, Liu G, Xia LC. Using decision tree aggregation with random forest model to identify gut microbes associated with colorectal cancer. Genes (Basel). (2019) 10:112. doi: 10.3390/genes10020112
9. Vivarelli S, Salemi R, Candido S, Falzone L, Santagati M, Stefani S, et al. Gut microbiota and cancer: from pathogenesis to therapy. Cancers (Basel). (2019) 11:1–26. doi: 10.3390/cancers11010038
10. Mills S, Stanton C, Lane JA, Smith GJ, Ross RP. Precision nutrition and the microbiome, part I: current state of the science. Nutrients. (2019) 11:1–45. doi: 10.3390/nu11040923
11. Mills S, Lane JA, Smith GJ, Grimaldi KA, Ross RP, Stanton C. precision nutrition and the microbiome part II: potential opportunities and pathways to commercialisation. Nutrients. (2019) 11:1468. doi: 10.3390/nu11071468
12. Dhar D, Mohanty A. Gut microbiota and covid-19– possible link and implications. Virus Res. (2020) 285:198018. doi: 10.1016/j.virusres.2020.198018
13. Ramalho R, Rao M, Zhang C, Agrati C, Ippolito G, Wang F, et al. Immunometabolism: new insights and lessons from antigen-directed cellular immune responses. Semin Immunopathol. (2020) 42:279–313. doi: 10.1007/s00281-020-00798-w
14. Garza DR, Taddese R, Wirbel J, Zeller G, Boleij A, Huynen MA, et al. Metabolic models predict bacterial passengers in colorectal cancer. Cancer Metab. (2020) 8:1–13. doi: 10.1186/s40170-020-0208-9
15. Dai Z, Zhang J, Wu Q, Chen J, Liu J, Wang L, et al. The role of microbiota in the development of colorectal cancer. Int J Cancer. (2019) 145:2032–41. doi: 10.1002/ijc.32017
16. John GK, Mullin GE. The gut microbiome and obesity. Curr Oncol Rep. (2016) 18:2–8. doi: 10.1007/s11912-016-0528-7
17. Sood U, Bajaj A, Kumar R, Khurana S, Kalia VC. Infection and microbiome: impact of tuberculosis on human gut microbiome of indian cohort. Indian J Microbiol. (2018) 58:123–5. doi: 10.1007/s12088-018-0706-4
18. Hussain I, Cher GLY, Abid MA, Abid MB. Role of gut microbiome in COVID-19: an insight into pathogenesis and therapeutic potential. Front Immunol. (2021) 12:765965. doi: 10.3389/fimmu.2021.765965
19. Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, et al. Expert consensus document: the international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. (2014) 11:506–14. doi: 10.1038/nrgastro.2014.66
20. Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, et al. Expert consensus document: the international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. (2017) 14:491–502. doi: 10.1038/nrgastro.2017.75
21. Lee HL, Shen H, Hwang IY, Ling H, Yew WS, Lee YS, et al. Targeted approaches for in situ gut microbiome manipulation. Genes (Basel). (2018) 9:351. doi: 10.3390/genes9070351
22. Patil S, Sawant S, Hauff K, Hampp G. Validated postbiotic screening confirms presence of physiologically-active metabolites, such as short-chain fatty acids, amino acids and vitamins in hylak® forte. Probiotics Antimicrob Proteins. (2019) 11:1124–31. doi: 10.1007/s12602-018-9497-5
23. Sharma M, Shukla G. Metabiotics: one step ahead of probiotics; an insight into mechanisms involved in anticancerous effect in colorectal cancer. Front Microbiol. (2016) 7:1940. doi: 10.3389/fmicb.2016.01940
24. Luo G, Li B, Yang C, Wang Y, Bian X, Li W, et al. Major traditional probiotics: comparative genomic analyses and roles in gut microbiome of eight cohorts. Front Microbiol. (2019) 10:712. doi: 10.3389/fmicb.2019.00712
25. Kumari M, Singh P, Nataraj BH, Kokkiligadda A, Naithani H, Azmal Ali S, et al. Fostering next-generation probiotics in human gut by targeted dietary modulation: an emerging perspective. Food Res Int. (2021) 150:110716. doi: 10.1016/j.foodres.2021.110716
26. Saarela MH. Safety aspects of next generation probiotics. Curr Opin Food Sci. (2019) 30:8–13. doi: 10.1016/j.cofs.2018.09.001
27. Yang J, McDowell A, Kim EK, Seo H, Yum K, Lee WH, et al. Consumption of a Leuconostoc holzapfelii-enriched synbiotic beverage alters the composition of the microbiota and microbial extracellular vesicles. Exp Mol Med. (2019) 51:87. doi: 10.1038/s12276-019-0288-1
28. Liu Z, Qin H, Yang Z, Xia Y, Liu W, Yang J, et al. Randomised clinical trial: the effects of perioperative probiotic treatment on barrier function and post-operative infectious complications in colorectal cancer surgery – a double-blind study. Aliment Pharmacol Ther. (2011) 33:50–63. doi: 10.1111/j.1365-2036.2010.04492.x
29. Madsen K. Probiotics and the immune response. J Clin Gastroenterol. (2006) 40:232–4. doi: 10.1097/00004836-200603000-00014
30. Ricci A, Tagliacarne SC, Valsecchi C, Boggini T, Cattaneo F, Licari A, et al. PROBIOTICS AND INFLAMMATORY BOWEL DISEASES. J Biol Regul Homeost Agents. (2015) 29:96–113.
31. Wilkins T, Sequoia J. Probiotics for gastrointestinal conditions: a summary of the evidence. Am Fam Physician. (2017) 96:170–8.
32. Rooks MG, Garrett WS. Gut microbiota, metabolites and host immunity. Nat Rev Immunol. (2016) 16:341–52. doi: 10.1038/nri.2016.42
33. Lai HH, Chiu CH, Kong MS, Chang CJ, Chen CC. Probiotic Lactobacillus casei: effective for managing childhood diarrhea by altering gut microbiota and attenuating fecal inflammatory markers. Nutrients. (2019) 11:1105. doi: 10.3390/nu11051150
34. Eskesen D, Jespersen L, Michelsen B, Whorwell PJ, Müller-Lissner S, Morberg CM. Effect of the probiotic strain Bifidobacterium animalis subsp. lactis, BB-12®, on defecation frequency in healthy subjects with low defecation frequency and abdominal discomfort: a randomised, double-blind, placebo-controlled, parallel-group trial. Br J Nutr. (2015) 114:1638–46. doi: 10.1017/S0007114515003347
35. Hilton E, Kolakowski P, Singer C, Smith M. Efficacy of Lactobacillus GG as a diarrheal preventive in travelers. J Travel Med. (1997) 4:41–3. doi: 10.1111/j.1708-8305.1997.tb00772.x
36. Aggarwal S, Upadhyay A, Shah D, Teotia N, Agarwal A, Jaiswal V. Lactobacillus GG for treatment of acute childhood diarrhoea: an open labelled, randomized controlled trial. Indian J Med Res. (2014) 139:379–85.
37. Salazar-Lindo E, Miranda-Langschwager P, Campos-Sanchez M, Chea-Woo E, Sack RB. Lactobacillus casei strain GG in the treatment of infants with acute watery diarrhea: a randomized, double-blind, placebo controlled clinical trial [ISRCTN67363048]. BMC Pediatr. (2004) 4:18. doi: 10.1186/1471-2431-4-18
38. Lahtinen SJ, Forssten S, Aakko J, Granlund L, Rautonen N, Salminen S, et al. Probiotic cheese containing Lactobacillus rhamnosus HN001 and Lactobacillus acidophilus NCFM§modifies subpopulations of fecal lactobacilli and Clostridium difficile in the elderly. Age (Dordr). (2012) 34:133–43. doi: 10.1007/s11357-011-9208-6
39. Vaisberg M, Paixão V, Almeida EB, Santos JMB, Foster R, Rossi M, et al. Daily intake of fermented milk containing Lactobacillus casei shirota (lcs) modulates systemic and upper airways immune/inflammatory responses in marathon runners. Nutrients. (2019) 11:1678. doi: 10.3390/nu11071678
40. Takada M, Nishida K, Gondo Y, Kikuchi-Hayakawa H, Ishikawa H, Suda K, et al. Beneficial effects of Lactobacillus casei strain Shirota on academic stress-induced sleep disturbance in healthy adults: a double-blind, randomised, placebo-controlled trial. Benef Microbes. (2017) 8:153–62. doi: 10.3920/BM2016.0150
41. Chao L, Liu C, Sutthawongwadee S, Li Y, Lv W, Chen W, et al. Effects of probiotics on depressive or anxiety variables in healthy participants under stress conditions or with a depressive or anxiety diagnosis: a meta-analysis of randomized controlled trials. Front Neurol. (2020) 11:421. doi: 10.3389/fneur.2020.00421
42. Nishida K, Sawada D, Kuwano Y, Tanaka H, Rokutan K. Health benefits of Lactobacillus gasseri cp2305 tablets in young adults exposed to chronic stress: a randomized, double-blind, placebo-controlled study. Nutrients. (2019) 11:1859. doi: 10.3390/nu11081859
43. Ansari F, Pourjafar H, Tabrizi A, Homayouni A. The effects of probiotics and prebiotics on mental disorders: a review on depression, anxiety, Alzheimer, and Autism spectrum disorders. Curr Pharm Biotechnol. (2020) 21:555–65. doi: 10.2174/1389201021666200107113812
44. Parker EA, Roy T, D’Adamo CR, Wieland LS. Probiotics and gastrointestinal conditions: an overview of evidence from the cochrane collaboration. Nutrition. (2018) 45:125–134.e11. doi: 10.1016/j.nut.2017.06.024
45. Pham M, Lemberg DA, Day AS. Probiotics: sorting the evidence from the myths. Med J Aust. (2008) 188:304–8. doi: 10.5694/j.1326-5377.2008.tb01627.x
46. Bajramagic S, Hodzic E, Mulabdic A, Holjan S, Smajlovic SV, Rovcanin A. Usage of probiotics and its clinical significance at surgically treated patients sufferig from colorectal carcinoma. Med Arch. (2019) 73:316–20. doi: 10.5455/medarh.2019.73.316-320
47. Sasidharan BK, Ramadass B, Viswanathan P, Samuel P, Gowri M, Pugazhendhi S, et al. A phase 2 randomized controlled trial of oral resistant starch supplements in the prevention of acute radiation proctitis in patients treated for cervical cancer. J Cancer Res Ther. (2019) 15:1383–91. doi: 10.4103/jcrt.JCRT_152_19
48. Saji N, Niida S, Murotani K, Hisada T, Tsuduki T, Sugimoto T, et al. Analysis of the relationship between the gut microbiome and dementia: a cross-sectional study conducted in Japan. Sci Rep. (2019) 9:1–9. doi: 10.1038/s41598-018-38218-7
49. Vascellari S, Melis M, Palmas V, Pisanu S, Serra A, Perra D, et al. Clinical phenotypes of Parkinson’s disease associate with distinct gut microbiota and metabolome enterotypes. Biomolecules. (2021) 11:1–16. doi: 10.3390/biom11020144
50. Yang TW, Lee WH, Tu SJ, Huang WC, Chen HM, Sun TH, et al. Enterotype-based analysis of gut microbiota along the conventional adenoma-carcinoma colorectal cancer pathway. Sci Rep. (2019) 9:1–13. doi: 10.1038/s41598-019-45588-z
51. Hou Q, Zhao F, Liu W, Lv R, Khine WWT, Han J, et al. Probiotic-directed modulation of gut microbiota is basal microbiome dependent. Gut Microbes. (2020) 12:1736974. doi: 10.1080/19490976.2020.1736974
52. Hollister EB, Riehle K, Luna RA, Weidler EM, Rubio-Gonzales M, Mistretta TA, et al. Structure and function of the healthy pre-adolescent pediatric gut microbiome. Microbiome. (2015) 3:36. doi: 10.1186/s40168-015-0101-x
53. Macfarlane S, Macfarlane GT. Bacterial diversity in the human gut. Adv Appl Microbiol. (2004) 54:261–89. doi: 10.1016/S0065-2164(04)54010-8
54. Lin A, Bik EM, Costello EK, Dethlefsen L, Haque R, Relman DA, et al. Distinct distal gut microbiome diversity and composition in healthy children from Bangladesh and the United States. PLoS One. (2013) 8:e53838. doi: 10.1371/journal.pone.0053838
55. King CH, Desai H, Sylvetsky AC, LoTempio J, Ayanyan S, Carrie J, et al. Baseline human gut microbiota profile in healthy people and standard reporting template. PLoS One. (2019) 14:e0206484. doi: 10.1371/journal.pone.0206484
56. Kong F, Hua Y, Zeng B, Ning R, Li Y, Zhao J. Gut microbiota signatures of longevity. Curr Biol. (2016) 26:R832–3. doi: 10.1016/j.cub.2016.08.015
57. Kieffer DA, Martin RJ, Adams SH. Impact of dietary fibers on nutrient management and detoxi fi cation organs?: gut. Am Soc Nutr. (2016) 7:1111–21. doi: 10.3945/an.116.013219.fiber
58. Venegas DP, De La Fuente MK, Landskron G, González MJ, Quera R, Dijkstra G, et al. Short chain fatty acids (SCFAs)mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front Immunol. (2019) 10:277. doi: 10.3389/fimmu.2019.00277
59. Kadosh E, Snir-Alkalay I, Venkatachalam A, May S, Lasry A, Elyada E, et al. The gut microbiome switches mutant p53 from tumour-suppressive to oncogenic. Nature. (2020) 586:133–8. doi: 10.1038/s41586-020-2541-0
60. Kong F, Deng F, Li Y, Zhao J. Identification of gut microbiome signatures associated with longevity provides a promising modulation target for healthy aging. Gut Microbes. (2019) 10:210–5. doi: 10.1080/19490976.2018.1494102
61. Claesson MJ, Cusack S, O’Sullivan O, Greene-Diniz R, De Weerd H, Flannery E, et al. Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc Natl Acad Sci USA. (2011) 108(Suppl. 1):4586–91. doi: 10.1073/pnas.1000097107
62. Claesson MJ, Jeffery IB, Conde S, Power SE, O’connor EM, Cusack S, et al. Gut microbiota composition correlates with diet and health in the elderly. Nature. (2012) 488:178–84. doi: 10.1038/nature11319
63. Carabotti M, Scirocco A, Maselli MA, Severi C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Ann Gastroenterol Q Publ Hell Soc Gastroenterol. (2015) 28:203.
64. Foster JA, McVey Neufeld K-A. Gut–brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. (2013) 36:305–12. doi: 10.1016/j.tins.2013.01.005
65. Li Q, Han Y, Dy ABC, Hagerman RJ. The gut microbiota and autism spectrum disorders. Front Cell Neurosci. (2017) 11:120. doi: 10.3389/fncel.2017.00120
66. Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. (2012) 13:701–12. doi: 10.1038/nrn3346
67. Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, et al. Host-gut microbiota metabolic interactions. Science. (2012) 336:1262–7. doi: 10.1126/science.1223813
68. Mulak A, Bonaz B. Brain-gut-microbiota axis in Parkinson’s disease. World J Gastroenterol. (2015) 21:10609. doi: 10.3748/WJG.V21.I37.10609
69. Friedland RP. Mechanisms of molecular mimicry involving the microbiota in neurodegeneration. J Alzheimers Dis. (2015) 45:349–62. doi: 10.3233/JAD-142841
70. World Health Organization. Fact Sheet Obesity and Overweight. Geneva: World Health Organization (2011).
71. Dabke K, Hendrick G, Devkota S. The gut microbiome and metabolic syndrome. J Clin Invest. (2019) 129:4050–7. doi: 10.1172/JCI129194
72. David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. (2014) 505:559–63. doi: 10.1038/nature12820
73. Collado MC, Isolauri E, Laitinen K, Salminen S. Distinct composition of gut microbiota during pregnancy in overweight and normal-weight women. Am J Clin Nutr. (2008) 88:894–9. doi: 10.1093/ajcn/88.4.894
74. Kalliomäki M, Carmen Collado M, Salminen S, Isolauri E. Early differences in fecal microbiota composition in children may predict overweight. Am J Clin Nutr. (2008) 87:534–8. doi: 10.1093/ajcn/87.3.534
75. Daniali M, Nikfar S, Abdollahi M. A brief overview on the use of probiotics to treat overweight and obese patients. Expert Rev Endocrinol Metab. (2020) 15:1–4. doi: 10.1080/17446651.2020.1719068
76. Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. (2006) 444:1027. doi: 10.1038/nature05414
77. Abenavoli L, Scarpellini E, Colica C, Boccuto L, Salehi B, Sharifi-Rad J, et al. Gut microbiota and obesity: a role for probiotics. Nutrients. (2019) 11:2690. doi: 10.3390/nu11112690
78. Vajro P, Mandato C, Licenziati MR, Franzese A, Vitale DF, Lenta S, et al. Effects of Lactobacillus rhamnosus strain GG in pediatric obesity-related liver disease. J Pediatr Gastroenterol Nutr. (2011) 52:740–3. doi: 10.1097/MPG.0b013e31821f9b85
79. Luoto R, Kalliomäki M, Laitinen K, Isolauri E. The impact of perinatal probiotic intervention on the development of overweight and obesity: follow-up study from birth to 10 years. Int J Obes. (2010) 34:1531–7. doi: 10.1038/ijo.2010.50
80. Palmer C, Bik EM, DiGiulio DB, Relman DA, Brown PO. Development of the human infant intestinal microbiota. PLoS Biol. (2007) 5:e177. doi: 10.1371/journal.pbio.0050177
81. Jiménez E, Marín ML, Martín R, Odriozola JM, Olivares M, Xaus J, et al. Is meconium from healthy newborns actually sterile? Res Microbiol. (2008) 159:187–93. doi: 10.1016/j.resmic.2007.12.007
82. Zhang X, Shen D, Fang Z, Jie Z, Qiu X, Zhang C, et al. Human Gut Microbiota Changes Reveal the Progression of Glucose Intolerance. PLoS One. (2013) 8:e71108. doi: 10.1371/journal.pone.0071108
83. Depommier C, Everard A, Druart C, Plovier H, Van Hul M, Vieira-Silva S, et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med. (2019) 25:1096–103. doi: 10.1038/s41591-019-0495-2
84. Zhang T, Li Q, Cheng L, Buch H, Zhang F. Akkermansia muciniphila is a promising probiotic. Microb Biotechnol. (2019) 12:1109–25. doi: 10.1111/1751-7915.13410
85. Yazdani B, Shidfar F, Salehi E, Baghbani-arani F, Razmpoosh E, Asemi Z, et al. Probiotic plus low-calorie diet increase gene expression of Toll-like receptor 2 and FOXP3 in overweight and obese participants. J Funct Foods. (2018) 43:180–5. doi: 10.1016/j.jff.2018.02.013
86. Hansen T. Genetics of type 2 diabetes. Curr Sci. (2002) 83:1477–82. doi: 10.5005/jp/books/12626_22
87. Salgaço MK, Oliveira LGS, Costa GN, Bianchi F, Sivieri K. Relationship between gut microbiota, probiotics, and type 2 diabetes mellitus. Appl Microbiol Biotechnol. (2019) 103:9229–38. doi: 10.1007/s00253-019-10156-y
88. Roager HM, Vogt JK, Kristensen M, Hansen LBS, Ibrügger S, Mærkedahl RB, et al. Whole grain-rich diet reduces body weight and systemic low-grade inflammation without inducing major changes of the gut microbiome: a randomised cross-over trial. Gut. (2019) 68:83–93. doi: 10.1136/gutjnl-2017-314786
89. Woldeamlak B, Yirdaw K, Biadgo B. Role of gut microbiota in type 2 diabetes mellitus and its complications: novel insights and potential intervention strategies. Korean J Gastroenterol. (2019) 74:314–20. doi: 10.4166/kjg.2019.74.6.314
90. Sabatino A, Regolisti G, Cosola C, Gesualdo L, Fiaccadori E. Intestinal microbiota in type 2 diabetes and chronic kidney disease. Curr Diab Rep. (2017) 17:16. doi: 10.1007/s11892-017-0841-z
91. Zhai L, Wu J, Lam YY, Kwan HY, Bian ZX, Wong HLX. Gut-microbial metabolites, probiotics and their roles in type 2 diabetes. Int J Mol Sci. (2021) 22:12846. doi: 10.3390/ijms222312846
92. Park KY, Kim B, Hyun CK. Lactobacillus rhamnosus GG improves glucose tolerance through alleviating ER stress and suppressing macrophage activation in db/db mice. J Clin Biochem Nutr. (2015) 56:240–6. doi: 10.3164/jcbn.14-116
93. Lim SM, Jeong JJ, Woo KH, Han MJ, Kim DH. Lactobacillus sakei OK67 ameliorates high-fat diet–induced blood glucose intolerance and obesity in mice by inhibiting gut microbiota lipopolysaccharide production and inducing colon tight junction protein expression. Nutr Res. (2016) 36:337–48. doi: 10.1016/j.nutres.2015.12.001
94. Kobyliak N, Conte C, Cammarota G, Haley AP, Styriak I, Gaspar L, et al. Probiotics in prevention and treatment of obesity: a critical view. Nutr Metab (Lond). (2016) 13:14. doi: 10.1186/s12986-016-0067-0
95. Hsiao EY, McBride SW, Hsien S, Sharon G, Hyde ER, McCue T, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. (2013) 155:1451–63. doi: 10.1016/J.CELL.2013.11.024
96. Liu S, Li E, Sun Z, Fu D, Duan G, Jiang M, et al. Altered gut microbiota and short chain fatty acids in Chinese children with autism spectrum disorder. Sci Rep. (2019) 9:287. doi: 10.1038/s41598-018-36430-z
97. Adams JB, Johansen LJ, Powell LD, Quig D, Rubin RA. Gastrointestinal flora and gastrointestinal status in children with autism – comparisons to typical children and correlation with autism severity. BMC Gastroenterol. (2011) 11:22. doi: 10.1186/1471-230X-11-22
98. Macfabe DF. Short-chain fatty acid fermentation products of the gut microbiome: implications in autism spectrum disorders. Microb Ecol Health Dis. (2012) 23:19260. doi: 10.3402/mehd.v23i0.19260
99. Doenyas C. Novel personalized dietary treatment for autism based on the gut-immune-endocrine-brain axis. Front Endocrinol (Lausanne). (2019) 10:508. doi: 10.3389/fendo.2019.00508
100. Srinivasjois R, Rao S, Patole S. Probiotic supplementation in children with autism spectrum disorder. Arch Dis Child. (2015) 100:505–6. doi: 10.1136/archdischild-2014-308002
101. Umbrello G, Esposito S. Microbiota and neurologic diseases: potential effects of probiotics. J Transl Med. (2016) 14:298. doi: 10.1186/s12967-016-1058-7
102. Cermak SA, Curtin C, Bandini LG. Food selectivity and sensory sensitivity in children with autism spectrum disorders. J Am Diet Assoc. (2010) 110:238–46. doi: 10.1016/j.jada.2009.10.032
103. Bandini LG, Anderson SE, Curtin C, Cermak S, Evans EW, Scampini R, et al. Food selectivity in children with autism spectrum disorders and typically developing children. J Pediatr. (2010) 157:259–64. doi: 10.1016/j.jpeds.2010.02.013
104. Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL, et al. Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol. (2019) 15:565–81. doi: 10.1038/s41582-019-0244-7
105. Westfall S, Lomis N, Kahouli I, Dia SY, Singh SP, Prakash S. Microbiome, probiotics and neurodegenerative diseases: deciphering the gut brain axis. Cell Mol Life Sci. (2017) 74:3769–87. doi: 10.1007/s00018-017-2550-9
106. Xiao J, Wang T, Xu Y, Gu X, Li D, Niu K, et al. Long-term probiotic intervention mitigates memory dysfunction through a novel H3K27me3-based mechanism in lead-exposed rats. Transl Psychiatry. (2020) 10:25. doi: 10.1038/s41398-020-0719-8
107. Leblhuber F, Steiner K, Schuetz B, Fuchs D, Gostner JM. Probiotic supplementation in patients with Alzheimer’s dementia – an explorative intervention study. Curr Alzheimer Res. (2018) 15:1106–13. doi: 10.2174/1389200219666180813144834
108. Magistrelli L, Amoruso A, Mogna L, Graziano T, Cantello R, Pane M, et al. Probiotics may have beneficial effects in Parkinson’s disease: in vitro evidence. Front Immunol. (2019) 10:969. doi: 10.3389/fimmu.2019.00969
109. Braak H, Rüb U, Gai WP, Del Tredici K. Idiopathic Parkinson’s disease: possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen. J Neural Transm. (2003) 110:517–36. doi: 10.1007/s00702-002-0808-2
110. Shannon KM, Keshavarzian A, Dodiya HB, Jakate S, Kordower JH. Is alpha-synuclein in the colon a biomarker for premotor Parkinson’s disease? Evidence from 3 cases. Mov Disord. (2012) 27:716–9. doi: 10.1002/mds.25020
111. Gazerani P. Probiotics for Parkinson’s disease. Int J Mol Sci. (2019) 20:4121. doi: 10.3390/ijms20174121
112. Barichella M, Pacchetti C, Bolliri C, Cassani E, Iorio L, Pusani C, et al. Probiotics and prebiotic fiber for constipation associated with Parkinson disease: an RCT. Neurology. (2016) 87:1274–80. doi: 10.1212/WNL.0000000000003127
113. Abraham D, Feher J, Scuderi GL, Szabo D, Dobolyi A, Cservenak M, et al. Exercise and probiotics attenuate the development of Alzheimer’s disease in transgenic mice: role of microbiome. Exp Gerontol. (2019) 115:122–31. doi: 10.1016/j.exger.2018.12.005
114. Athari Nik Azm S, Djazayeri A, Safa M, Azami K, Ahmadvand B, Sabbaghziarani F, et al. Lactobacilli and bifidobacteria ameliorate memory and learning deficits and oxidative stress in β-amyloid (1–42) injected rats. Appl Physiol Nutr Metab. (2018) 43:718–26. doi: 10.1139/apnm-2017-0648
115. Akbari E, Asemi Z, Daneshvar Kakhaki R, Bahmani F, Kouchaki E, Tamtaji OR, et al. Effect of probiotic supplementation on cognitive function and metabolic status in Alzheimer’s disease: a randomized, double-blind and controlled trial. Front Aging Neurosci. (2016) 8:256. doi: 10.3389/fnagi.2016.00256
117. Monda V, Villano I, Messina A, Valenzano A, Esposito T, Moscatelli F, et al. Exercise modifies the gut microbiota with positive health effects. Oxid Med Cell Longev. (2017) 2017:3831972. doi: 10.1155/2017/3831972
118. Choi JJ, Eum SY, Rampersaud E, Daunert S, Abreu MT, Toborek M. Exercise attenuates PCB-induced changes in the mouse gut microbiome. Environ Health Perspect. (2013) 121:725–30. doi: 10.1289/ehp.1306534
119. Daniel H, Gholami AM, Berry D, Desmarchelier C, Hahne H, Loh G, et al. High-fat diet alters gut microbiota physiology in mice. ISME J. (2014) 8:295–308. doi: 10.1038/ismej.2013.155
Keywords: therapy, microbes, obesity, probiotics, diabetes, neurodegenerative diseases
Citation: Lee SHF, Ahmad SR, Lim YC and Zulkipli IN (2022) The Use of Probiotic Therapy in Metabolic and Neurological Diseases. Front. Nutr. 9:887019. doi: 10.3389/fnut.2022.887019
Received: 01 March 2022; Accepted: 29 March 2022;
Published: 03 May 2022.
Edited by:
Surasak Saokaew, University of Phayao, ThailandReviewed by:
Anchalee Rawangkan, University of Phayao, ThailandEric Banan-Mwine Daliri, Kangwon National University, South Korea
Copyright © 2022 Lee, Ahmad, Lim and Zulkipli. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ihsan N. Zulkipli, bmF6dXJhaC56dWxraXBsaUB1YmQuZWR1LmJu
 Shirley H. F. Lee
Shirley H. F. Lee Siti R. Ahmad
Siti R. Ahmad Ya C. Lim
Ya C. Lim Ihsan N. Zulkipli
Ihsan N. Zulkipli