Abstract
The devastating COVID-19 pandemic has caused more than six million deaths worldwide during the last 2 years. Effective therapeutic agents are greatly needed, yet promising magic bullets still do not exist. Numerous natural products (cordycepin, gallinamide A, plitidepsin, telocinobufagin, and tylophorine) have been widely studied and play a potential function in treating COVID-19. In this paper, we reviewed published studies (from May 2021 to April 2022) relating closely to bioactive natural products (isolated from medicinal plants, animals products, and marine organisms) in COVID-19 therapy in vitro to provide some essential guidance for anti-SARS-CoV-2 drug research and development.
1 Introduction
The ongoing coronavirus disease 2019 (COVID-19) pandemic, the sixth public health emergency of international concern, has resulted in 505,035,185 cases and 6,210,719 deaths worldwide during the last 2 years (at the time of writing). (World Health Organization, 2022). The Alpha, Beta, Gamma, and Delta variants of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) responsible for COVID-19 have created recurrent pandemic alerts. (Nasreen et al., 2022). Alarmingly, the novel Omicron (South Africa) variant was firstly confirmed on 24 November 2021. Still, it became the most predominant strain internationally within months because of its increased transmissibility and extensive immune evasion ability. (Scott et al., 2021; Del Rio et al., 2022). Up to now, the devastating Omicron variant has spread to almost all countries. Effective measures, such as vaccines, (Andrews et al., 2022; Chandrashekar et al., 2022) traditional medicine, (Liu et al., 2020; Alam et al., 2021) and small-molecule inhibitors, (Wang and Yang, 2020a; Reis et al., 2022; Sourimant et al., 2022) are greatly needed to reduce human-to-human transmission.
However, promising magic bullets still do not exist. (Kozlov, 2022). As an indispensable resource for promising compounds, natural products have attracted significant attention in countering SARS-CoV-2 infection via targeting its main protease (Mpro, also called 3CLpro), (Jin et al., 2020; Mengist et al., 2020) RNA-dependent RNA polymerase (RdRp), (Hillen et al., 2020; Wang et al., 2021a) papain-like protease (PLpro), (Yin et al., 2020; Gao et al., 2021) and spike (S) glycoprotein. (Toelzer et al., 2020; Walls et al., 2020). Building on our previously published work, (Wang and Yang, 2020b; Yang and Wang, 2021) we systematically discuss the landmark studies (published between May 2021 and April 2022) relating to bioactive natural products in COVID-19 therapy in vitro to support anti-SARS-CoV-2 drugs research and development.
2 Promising bioactive natural products in COVID-19 therapy
Bioactive natural products, isolated from medicinal plants, animal products, and marine organisms, are widely studied (in in vitro, animal models, and clinical trials) and play an important role in COVID-19 therapy. (Wei et al., 2020; Sahoo et al., 2021; Alqathama et al., 2022). Natural products are still considered one of the most positive and practical approaches to defeating the ongoing pandemic.
Tylophorine, a remarkable tylophora alkaloid, is an active pharmaceutical ingredient of the medicinal plant Cynanchum komarovii AL (Figure 1A) (An et al., 2001). NK007(S,R), a racemate of tylophorine malate, was prepared from S-tylophorine to improve its poor solubility. (Wang et al., 2010). NK007(S,R) displays significant inhibitory activity against SARS-CoV-2 at a half maximal effective concentration (EC50) of 0.030 μM in Vero cells, with an excellent selectivity profile (selectivity index, [SI] = 868). (Wang et al., 2021b). Hossain et al. (2022) found that tylophorine showed binding affinity (−8.5 kcal/mol) against abelson murine leukemia viral oncogene homolog one protein. Additionally, NK007(S,R) exhibits excellent in vivo antiviral efficacy in the COVID-19 golden hamster rat model by significantly reducing viral loads in the lungs. NK007(S,R) could protect against lung injury by decreasing lung inflammation with a dose of 5 mg/kg. (Wang et al., 2021b). Briefly, the abovementioned evidence has highlighted the superior activity of NK007(S,R) against SARS-CoV-2 infection in in vitro and in the rat model. (Wang et al., 2021b). Numerous natural product-based nanomedicines have been sprung up during the past several decades in the field of medicinal chemistry, providing a valuable reference for anti-COVID-19 therapeutics. (Sharma et al., 2021). To evaluate the potential of the candidate NK007(S,R), Wang et al. (Wang et al., 2021b) prepared self-assembled poly (ethylene glycol)–poly (lactide-co-glycolide) nanoparticles, NP-NK007 and LP-NK007. The optimized NP-NK007 exhibited small particle size (145.8 nm), high NK007(S,R) loading (13.10%), maximized encapsulation efficiency (87.47%), and sustained release (66.51% in 48 h). The optimal lung-targeted liposome LP-NK007 exhibited smaller particle size (75 nm), higher drug loading (36.7%), and excellent encapsulation efficiency (62.4%). Subsequent experiments implied that the nanoparticles NP-NK007 and LP-NK007 are effective SARS-CoV-2 inhibitors with higher EC50 values of 0.007 and 0.014 μM, respectively, because they improve the accumulation and delivered efficiency of NK007(S,R) in the lung. (Wang et al., 2021b). Collectively, NK007(S,R) NPs could provide a workable strategy for overcoming the lack of COVID-19-targeting treatment. Theoretically, more validation studies in vivo are needed to systematically assess the anti-SARS-CoV-2 potential of NK007(S,R)-based nanoparticles.
FIGURE 1
Venenum Bufonis (Chinese name: ChanSu), a well-known secretion of a traditional medicine animal (toad Bufo bufo gargarizans), is commonly used in China to treat various diseases, including heart failure, infections, toothaches, and cancers. (Tian et al., 2017; Shen et al., 2022). For example, Huachansu injection, a valuable anticancer agent, has been used in tumour treatment in China for more than 30 years. (Wu et al., 2022a). ChanSu’s main active constituents are bufadienolides that have an unusual 2-pyrone ring, which contributes to their pharmacological activities via inhibiting Na+/K+ ATPase. (Prassas and Diamandis, 2008). Recently, Jin et al. (2021) demonstrated that six bufadienolides (bufalin, bufotalin, cinobufagin, cinobufotalin, resibufogenin, and telocinobufagin) have potent broad-spectrum antiviral activities in vitro (Figure 1B). Experiments showed that bufalin could inhibit virus replication in the nanomolar range, including MERS-CoV at a half-maximal inhibitory concentration (IC50) of 0.018 μM, SARS-CoV at an IC50 of 0.016 μM, and SARS-CoV-2 at an IC50 of 0.019 μM; cinobufagin can inhibit MERS-CoV, SARS-CoV, and SARS-CoV-2 replication at IC50 values of 0.017, 0.060, and 0.072 μM; telocinobufagin can inhibit MERS-CoV, SARS-CoV and SARS-CoV-2 replication with IC50 values of 0.027, 0.071, and 0.142 μM; bufotalin, cinobufotalin and resibufogenin can inhibit the MERS-CoV, SARS-CoV and SARS-CoV-2 replication in vitro with high IC50 values (0.027–1.612 µM). (Jin et al., 2021). This study showed that the unusual 2-pyrone ring in bufadienolides plays an essential role in inhibiting SARS-CoV-2 replication. Subsequent dose toxicity studies (10 mg/kg/day, 5 days) revealed that bufalin and cinobufagin have strong toxicity in the mouse model, while the pharmacokinetic model predicts that telocinobufagin has lower toxicity, better metabolic stability, excellent oral bioavailability, and proper anti-SARS-CoV-2 activity. (Jin et al., 2021). Taken together, telocinobufagin might be a more promising broad‐spectrum inhibitor among the bufadienolides, and thus worthy of multifaceted properties investigation from in vitro studies to clinical practice.
Gallinamide A, possessing an α,β-unsaturated imide moiety, is a novel linear depsipeptide first isolated in 2008 from the marine cyanobacteria Schizothrix genus and Symploca sp. with critical pharmacological effects (Figure 1C) (Linington et al., 2009; Taori et al., 2009). Gallinamide A is a highly selective covalent inhibitor targeting human cathepsin L-like cysteine proteases, which is a promising drug target. (Barbosa Da Silva et al., 2022). Gerwick’s group showed that gallinamide A had a 28- to 320-fold higher affinity and selectivity towards cathepsin L than cathepsin V or B. (Miller et al., 2014). In vitro, gallinamide A demonstrates significant bioactivity against Trypanosoma cruzi at an IC50 of 0.005 μM by irreversible Michael addition. (Miller et al., 2014). It has been reported that gallinamide A can decrease viral load in VeroE6 cells with an IC90 of 0.088 µM and inhibit SARS-CoV-2 cathepsin L-mediated endosomal entry with an EC50 value of 0.028 µM in a dose-dependent manner. (Ashhurst et al., 2022). Angiotensin-converting enzyme 2 (ACE2) and transmembrane serine protease 2 (TMPRSS2) are two essential host determinants for SARS-CoV-2 infection and pathogenesis in vivo. (Hoffmann et al., 2020). Specifically, the S glycoprotein helps the virus enter inside the host cell via cellular receptor ACE2 binding; then TMPRSS2 helps SARS-CoV-2 contents fuse and release into the host cell cytosol via enzymatical activation of the S glycoprotein. (Liu et al., 2022). Based on combination drug therapies, Payne et al. (Ashhurst et al., 2022) recently demonstrated that the combined use of the cathepsin L inhibitor gallinamide A and the TMPRSS2 protease inhibitor nafamostat mesylate exerts a synergistic inhibitory effect in HEK-ACE2-TMPRSS2 cells via inhibiting multiple routes of SARS-CoV-2 entry. Taking gallinamide A as the lead, Payne et al. (Ashhurst et al., 2022) further explored and synthesized 32 analogues for the assessment of SARS-CoV-2 cathepsin L inhibitory activities; the study revealed two lead analogues of gallinamide A with EC50 values in the nanomolar range. Taken together, gallinamide A is a highly selective SARS-CoV-2 cathepsin L inhibitor, thus worthy of further investigation via combination therapies and lead optimization.
Natural products with broad‐spectrum bioactivities and multi-organ protection are an essential class of anti‐SARS‐CoV‐2 agents that play vital roles in COVID-19 therapy. (Wang and Yang, 2021). RdRp could regulate viral replication through catalyzing the RNA template–dependent development of phosphodiester bonds. (Wang et al., 2021a). The adenosine analogue cordycepin (3′-deoxyadenosine) is a unique fungal product isolated from the traditional medicine fungi Cordyceps militaris (Cunningham et al., 1950) and Ophiocordyceps sinensis (Figure 1D) (Zhou et al., 2008). Interestingly, cordycepin is known to have broad‐spectrum pharmacological properties against several diseases (e.g., virulent RNA viruses) and multi-organ protective effects (e.g. acute lung injury). Specifically, cordycepin is a promising therapeutic against several viruses in vitro, including dengue virus, (Panya et al., 2021) Epstein-Barr virus, (Choi et al., 2019) and hepatitis C virus. (Ueda et al., 2014). Because of its close structural similarity to the cellular nucleoside adenosine (except for the absence of a hydroxyl group at the 3′-position of the five-membered ring), cordycepin is a possible potent anti-SARS-CoV-2 agent. Rabie et al. (Rabie, 2022) showed that cordycepin could inhibit SARS-CoV-2 replication in Vero E6 cells with an EC50 value of 2.01 µM and without observable cytotoxicity (SI > 49.8) in a time-dependent manner. It is worth noting that cordycepin is a long-acting antiviral for SARS-CoV-2 prevention with high metabolic stability, reaching maximal anti-SARS-CoV-2 potency within 1.5–2.0 days of treatment. (Rabie, 2022). With respect to the activation mechanism, cordycepin is rapidly converted in vivo to its mono-, di-, and triphosphate forms; then, the active form cordycepin triphosphate can serve as a substrate for the RNA-dependent RNA polymerase (RdRp) to terminate the synthesis of viral RNA sequences. (Rabie, 2022). Bibi et al. (2022) revealed that two pivotal amino acid residues (Asp760 and Asp761) play critical roles in the binding of cordycepin with RdRp. Notably, SARS-CoV-2 infection—even in mild cases—can increase the long-term risk of a broad range of cardiovascular and cerebrovascular complications in COVID-19 patients. (Wang and Yang, 2022c). In terms of organ protection, cordycepin has unique advantages. For example, cordycepin plays a key role in long-term neuroprotection for traumatic brain injury (through inhibiting neutrophil infiltration and preserving neuroinflammation), (Wei et al., 2021) protecting diabetic hearts from ischemia/reperfusion injury (via up-regulating AMPK/Mfn2-dependent mitochondrial fusion and expression), (Yu et al., 2021) and ameliorating cerebral ischemic damage (via improving the memory ability, up-regulating the level of adenosine A1 receptors, and reducing dendritic morphology scathing). (Chen et al., 2021). Thus, cordycepin has its advantages in organ protection and broad-spectrum antiviral activities. Further study is still needed, however, to evaluate its antiviral potency in vitro.
The marine environment is a valuable source of structurally unique natural products with diverse bioactivity targeted at life-threatening diseases, including the emerging COVID-19. (Panggabean et al., 2022; Pokharkar et al., 2022; Zhang et al., 2022). Homofascaplysin A, isolated from the marine sponge Fascaplysinopsis reticulata (Figure 1E), is a well-established β-carboline alkaloid reported to exhibit promising activity against many viruses, including hepatitis C virus, (Ishida et al., 2001) human coronavirus NL63, (Tsai et al., 2020) and dengue virus. (Quintana et al., 2016). Kubanek et al. (Chhetri et al., 2022) revealed that homofascaplysin A can inhibit SARS-CoV-2 replication in Calu-3 cells at an EC50 value of 1.1 µM with relatively slight cytotoxicity (SI ∼4.55). Additionally, Kubanek et al. (Chhetri et al., 2022) found that the viral load was substantially reduced (by >90%) for infections in harvested SARS-CoV-2 RNA after administration of 2.8 μM of homofascaplysin A. Therefore, homofascaplysin A could be used as a unique lead compound for the rapid screening of novel analogues with promising anti-SARS-CoV-2 activity and minimal cytotoxicity.
Chirality is a critical attribute of natural products. (Wang, 2019). Wallichin C and wallichin D, isolated from the medicinal fern Dryopteris wallichiana (Figure 1F), exhibit potent anti-SARS-CoV-2 activities in Vero-E6 cells at EC50 values of 4.5 and 12.1 μM, respectively. (Socolsky et al., 2012; Hou et al., 2022). The corresponding SI values of wallichins C and D were >35 and >11. (Hou et al., 2022). Furthermore, phloroglucinol-terpenoids wallichins C and D exhibit potent inhibitory activities in SARS-CoV-2-infected Calu-3 cells at EC50 values of 20.2 and 30.0 μM, with moderate cytotoxicity (SI values were 4.88 and 2.14 μM, respectively). (Hou et al., 2022). Notably, both wallichins C and D have the same core structure except for the chirality at C-14 position. The study has demonstrated that the slight differences in the chirality at C-14″ S (wallichin C) or C-14″ R (wallichin D) account for differences in their antiviral activities. (Hou et al., 2022). As for the activation mechanism, Zhou et al. (Hou et al., 2022) unambiguously showed that wallichins C and D have higher selectivity and stronger interaction toward the 3CLpro with Kd values of 12.0–16.6 μM, while not active against the TMPRSS2, spike glycoprotein, and ACE2 proteins. Taken together, wallichin C might be the more promising 3CLpro inhibitor, thus worthy of further investigation.
Among pharmacological interventions, traditional medicine plays a positive role in the prevention and treatment of the COVID-19 pandemic. (Lyu et al., 2021; Zhan et al., 2022). For example, the Qingfei Paidu decoction has shown amazing clinical efficacy in treating COVID-19 patients. (Li Y. et al., 2021). It is crucial to support scientific foundations for the clinical use of Chinese herbal medicine by exploring the underlying molecular mechanisms. (Cui et al., 2021; De Jin et al., 2021). Ye and co-workers (Yi et al., 2022) recently indicated that licorice-saponin A3 and its genine aglycone glycyrrhetinic acid, famous triterpenoids that could be isolated from the most frequently used medicinal plant Glycyrrhiza uralensis Fisch. (Figure 1G), show a remarkably different inhibitory potency against SARS-CoV-2 infection in Vero E6 cells at EC50 values of 0.075 μM (targeting SARS-CoV-2 nsp7 protein) and 3.17 μM (targeting the S protein receptor-binding domain [RBD]), respectively in a dose-dependent manner. Interestingly, licorice-saponin A3 and glycyrrhetinic acid were effective in inhibiting the SARS-CoV-2 spike RBD activities, with similar IC50 values of 8.3 and 10.9 μM, respectively. (Yi et al., 2022). To elucidate the remarkable difference between S-RBD inhibitory effects and their antiviral activities, the underlying molecular mechanisms were further explored by Ye and co-workers. Based on molecular docking analysis of licorice-saponin A3 with nsp7 (PDB ID:7JIT), Yi et al. (2022) propose that nsp7 is another vital target for licorice-saponin A3 via seven hydrogen bond interactions (binding energy −8.7 kcal/mol). Qingfei Paidu decoction extracted from 21 types of traditional Chinese medicines (including Glycyrrhiza uralensis Fisch.) could effectively treat COVID-19, highlighting an important contributor to the active components (such as licorice-saponin A3, glycyrrhetinic acid, and so on) in herbal medicine treatment. (Wu et al., 2022b). Importantly, the results provided valuable data on the “multi-components, multiple-pathways, and multi-targets” feature of traditional herbal medicine.
Glycosylation is an important structural modification that increases water solubility, enhances pharmacological activity, and improves the bioavailability of natural products. 11) In fact, GA mainly exists in the form of functional glycosides in licorice. At present, more than 43 saponins have been identified in licorice, many of which are glycosylated derivatives of GA. 12) These glycosylated derivatives have different sugar numbers and types and display various pharmacological activities.
3 Other promising natural products for treating SARS-CoV-2 infection
Innovative drug development is an arduous process; bioactive natural products greatly expedite the development of antiviral drugs. (Abdelmohsen et al., 2017). In addition to the abovementioned agents, numerous other natural products (Table 1) have exhibited highly efficacious anti-SARS-CoV-2 activities in vitro and clinical practice. For example, plitidepsin (Aplidin®), a eukaryotic translation elongation factor 1A (eEF1A) inhibitor of marine origin, was initially approved to treat multiple myeloma. (Rodon et al., 2021). Sachse et al. (2022) showed that plitidepsin is highly effective at inhibiting SARS‐CoV‐2 replication in a dose-dependent manner in Vero E6 cells at IC50 values of 0.0052 μM for D614G variants, 0.0039 μM for Delta variants, and 0.0043 μM for Omicron variants. Furthermore, White et al. (2021) showed that plitidepsin can inhibit SARS-CoV-2 replication in Vero E6 cells, hACE2-293T cells and pneumocyte-like cells at IC50 values of 0.00070, 0.00073, and 0.0016 μM, respectively, via targeting the host protein eEF1A. Notably, Guisado-Vasco et al. (2022) showed that plitidepsin is well-tolerated in humans and can lower viral load in SARS-CoV-2-infected chronic lymphocytic leukemia patients. Clinical trials of plitidepsin have been registered (NCT04382066 and NCT05121740) and will be reported shortly. Further study is still needed to evaluate its anti-SARS-CoV-2 potency in vivo and in vitro.
TABLE 1
| No. | Name | Structure | EC50 or IC50 (μM) | Strain | Refs |
|---|---|---|---|---|---|
| 1 | Aloin A |  | 15.68 | Vero E6 cells | Lewis et al. (2022) |
| 2 | Aloin B | 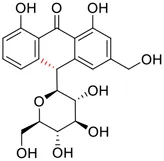 | 17.51 | Vero E6 cells | Lewis et al. (2022) |
| 3 | Andrographolide |  | 0.034 | Calu-3 cells | Sa-Ngiamsuntorn et al. (2021), Schulte et al. (2022) |
| 4 | Aspulvinone D |  | 10.3 | J774A.1 cells | Liang et al. (2022) |
| 5 | Aspulvinone M |  | 9.4 | J774A.1 cells | Liang et al. (2022) |
| 6 | Aspulvinone R |  | 7.7 | J774A.1 cells | Liang et al. (2022) |
| 7 | (+)-Aureol |  | 4.00 | Calu-3 cells | Chhetri et al. (2022) |
| 8 | Baicalein |  | 1.11 | E. coli BL21 cells | Wu et al., 2022a, Xiao et al. (2021) |
| 9 | Baicalin |  | 8.8 | Vero E6 cells | Ngwe Tun et al. (2022) |
| 10 | Bromophycolide A |  | 6.90 | Calu-3 cells | Chhetri et al. (2022) |
| 11 | Bufotalin |  | 0.072 | Vero E6 cells | Jin et al. (2021) |
| 12 | Cannabidiol |  | 1.24 | A549-ACE2 cells | Corpetti et al. (2021), Nguyen et al. (2022) |
| 13 | Cannabidiolic acid |  | 24 μg/mL | Vero E6 cells | van Breemen et al. (2022) |
| 14 | Cannabigerolic acid |  | 37 μg/mL | Vero E6 cells | van Breemen et al. (2022) |
| 15 | Chebulagic acid |  | 9.76 | Vero E6 cells | Du et al. (2021) |
| 16 | Cinobufagin |  | 0.072 | Vero E6 cells | Corpetti et al. (2021), Nguyen et al. (2022) |
| 17 | Cinobufotalin |  | 0.399 | Vero E6 cells | Jin et al. (2021) |
| 18 | Corilagin |  | 24.9 | HEK293 cells | Yang et al. (2021a) |
| 19 | Curcumin |  | 11.9 | Vero E6 cells | Bahun et al. (2022) |
| 20 | Cyclopeniol |  | 0.39 | RAW264.7 cells | Thissera et al. (2021) |
| 21 | Cyclopeptin |  | 0.40 | RAW264.7 cells | Thissera et al. (2021) |
| 22 | Dehydrocyclopeptin |  | 0.89 | RAW264.7 cells | Thissera et al. (2021) |
| 23 | Dieckol |  | 4.50 | Vero E6 cells | Yan et al. (2021) |
| 24 | Digitoxin |  | 0.059 | Vero E6 cells | Caohuy et al. (2021), Jin et al. (2021), Caohuy et al. (2022) |
| 25 | Dihydromyricetin |  | 1.14 | Vero E6 cells | Su et al. (2021), Xiao et al. (2021) |
| 26 | Dihydrotanshinone I |  | 8.14 | Vero E6 cells | Ma and Wang, (2022) |
| 27 | Dithymoquinone |  | 0.275 μg/mL | Vero E6 cells | Esharkawy et al. (2022) |
| 28 | Echinulin |  | 3.90 | Vero E6 cells | Alhadrami et al. (2022) |
| 29 | EGCG |  | 4.24 | Vero E6 cells | Chiou et al. (2022) |
| 30 | Ellagic acid |  | 11.8 | Vero E6 cells | Bahun et al. (2022) |
| 31 | (-)-Gallocatechin gallate |  | 5.77 | Vero E6 Cells | Xiao et al. (2021) |
| 32 | Glabridin |  | 2.5 | Vero E6 cells | Ngwe Tun et al. (2022) |
| 33 | Hesperidin | 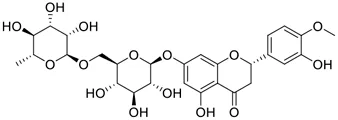 | 51.5 | Vero E6 cells | Huang et al. (2022) |
| 34 | Isopetasin |  | 0.37 | Vero E6 cells | Urda et al. (2022) |
| 35 | Ivermectin |  | 0.55 | Vero E6 cells | Chable-Bessia et al. (2022) |
| 36 | Licoflavone C |  | 1.34 | Vero E6 cells | Corona et al. (2022) |
| 37 | LPC (14:0/0:0) |  | 0.92 | Vero E6 cells | Du et al. (2022) |
| 38 | LPC (16:0/0:0) |  | 1.48 | Vero E6 cells | Du et al. (2022) |
| 39 | LPC (16:0/18:1) |  | 0.14 | Vero E6 cells | Du et al. (2022) |
| 40 | Myricetin |  | 0.63 | Vero E6 cells | Kato et al. (2021), Su et al. (2021) |
| 41 | Neferine |  | 0.36 | HEK293/hACE2 cells | Yang et al. (2021b) |
| 42 | (+)-Neoechinulin A |  | 0.47 | Vero E6 cells | Alhadrami et al. (2022) |
| 43 | Neoechinulin B |  | 32.9 | Vero E6 cells | Nishiuchi et al. (2022) |
| 44 | Neopetasin |  | 1.26 | Vero E6 cells | Urda et al. (2022) |
| 45 | Oridonin |  | 2.16 | Vero E6 cells | Zhong et al. (2022) |
| 46 | Petasin |  | 10.79 | Vero E6 cells | Urda et al. (2022) |
| 47 | PGG |  | 3.66 | Vero E6 cells | Chiou et al. (2022) |
| 48 | Piniterpenoid A |  | 64.5 | Vero E6 cells | Li et al. (2021b) |
| 49 | Piniterpenoid C |  | 76.1 | Vero E6 cells | Li et al. (2021b) |
| 50 | Plitidepsin |  | 0.0043 | Vero E6 cells | Guisado-Vasco et al. (2022), Sachse et al. (2022) |
| 51 | Punicalagin |  | 6.19 | Vero E6 cells | Saadh et al. (2021)Suručić et al. (2021) |
| 52 | Resibufogenin |  | 1.606 | Vero E6 cells | Jin et al. (2021) |
| 53 | Salvianolic acid A |  | 2.49 | Vero E6 cells | Zhong et al. (2022) |
| 54 | Sangivamycin |  | 0.015 | Vero E6 cells | Bennett et al. (2022) |
| 55 | Scutellarein |  | 5.68 | E. coli BL21 cells | Wu et al. (2022c) |
| 56 | (+)-Shikonin |  | 4.38 | Vero E6 cells | Zhao et al. (2021), Ma et al. (2022) |
| 57 | Shikonin |  | 4.50 | Vero E6 cells | Cui and Jia (2021), Zhao et al. (2021) |
| 58 | Sulforaphane |  | 2.40 | Caco-2 cells | Ordonez et al. (2022) |
| 59 | Tanshinone IIA |  | 7.82 μg/mL | Vero E6 cells | Elebeedy et al. (2022) |
| 60 | Telocinobufagin |  | 0.142 | Vero E6 cells | Jin et al. (2021) |
| 61 | Thymohydroquinone |  | 0.023 μg/mL | Vero E6 cells | Esharkawy et al. (2022) |
| 62 | Tubercidin |  | 0.05 | Calu-3 cells | Schultz et al. (2022) |
| 63 | Ugonin J |  | 2.38 | Vero E6 Cells | Chiou et al. (2021) |
| 64 | 5,3′,4′-trihydroxyflavone |  | 8.22 | Vero E6 cells | Zhao et al. (2021) |
| 65 | 7-OH-Cannabidiol |  | 3.60 | A549-ACE2 cells | Nguyen et al. (2022) |
Other promising natural products for treating SARS-CoV-2 infection in vitro.
4 Conclusion and outlook
The devastating SARS-CoV-2 variants have caused over six million deaths worldwide. Natural products and small-molecule inhibitors have been widely studied (in in vitro studies, animal models, and clinical trials) and play an essential function in treating COVID-19. Drug research and development is a highly time‐consuming process. To date, Gilead’s controversial Veklury® (Remdesivir, RdRp inhibitor) was conditionally approved to combat the outbreak. (Kalil et al., 2021; Wang and Yang, 2022a). Pfizer’s oral broad‐spectrum candidate Paxlovid® (PF‐07321332, Mpro inhibitor) and Merck’s oral prodrug Lagevrio® (Molnupiravir, RdRp inhibitor) raise new hope for a COVID‐19 cure. (Cully, 2022; Wang and Yang, 2022b). Promising clinical results have occurred, while small-molecule inhibitors still have a long way to go.
The substantial progress in treating COVID‐19 patients is not sufficient. Multiple factors must be considered. The first feasible factor, optimized drug combination therapy (such as gallinamide A + remdesivir, licorice-saponin A3 + PF‐07321332, telocinobufagin + molnupiravir, and cordycepin + tylophorine), targeting multiple targets, could not only enhance synergistic efficacy but also reduce drug resistance and toxicity. However, any potential combination would need to be tested in vitro and in vivo to verify the anticipated synergistic or additive effect. The second workable approach is natural product-based nanomedicines therapy. For example, the tylophorine-based lung-targeted liposome LP-NK007 could inhibit SARS-CoV-2 replication with a higher EC50 value via improving the accumulation and efficient delivery in the lung. Third, natural product-based lead optimization offers a valuable reference for enhancing anti‐SARS‐CoV‐2 potency and pharmacokinetic parameters. For example, taking gallinamide A as the lead, Payne et al. (Ashhurst et al., 2022) synthesized two highly selective SARS-CoV-2 cathepsin L inhibitors with nanomolar EC50 values. Taken together, we hope natural products (with the help of natural product-based nanomedicines therapy, lead optimization, and drug combination) prove to be a compelling direction in COVID-19 therapy.
Statements
Author contributions
ZW conceived the review. NW, LY, and XS collected the literatures. ZW and LY wrote the manuscript. ZW and XS edited the manuscript. All authors read and approved the final version of the manuscript.
Funding
This work was supported by the project of the PhD research start-up fund of Qufu Normal University, China (Grant No. 614901 and 615201).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1
AbdelmohsenU. R.BalasubramanianS.OelschlaegerT. A.GrkovicT.PhamN. B.QuinnR. J.et al (2017). Potential of marine natural products against drug-resistant fungal, viral, and parasitic infections. Lancet. Infect. Dis.17 (2), e30–e41. 10.1016/S1473-3099(16)30323-1
2
AlamS.SarkerM.RahmanM.AfrinS.RichiF. T.ZhaoC.et al (2021). Traditional herbal medicines, bioactive metabolites, and plant products against COVID-19: Update on clinical trials and mechanism of actions. Front. Pharmacol.12, 671498. 10.3389/fphar.2021.671498
3
AlhadramiH. A.BurgioG.ThisseraB.OrfaliR.JiffriS. E.YaseenM.et al (2022). Neoechinulin A as a promising SARS-CoV-2 Mpro inhibitor: In vitro and in silico study showing the ability of simulations in discerning active from inactive enzyme inhibitors. Mar. Drugs20 (3), 163. 10.3390/md20030163
4
AlqathamaA. A.AhmadR.AlsaediR. B.AlghamdiR. A.AbkarE. H.AlrehalyR. H.et al (2022). The vital role of animal, marine, and microbial natural products against COVID-19. Pharm. Biol.60 (1), 509–524. 10.1080/13880209.2022.2039215
5
AnT. Y.HuangR. Q.YangZ.ZhangD. K.LiG. R.YaoY. C.et al (2001). Alkaloids from Cynanchum komarovii with inhibitory activity against the tobacco mosaic virus. Phytochemistry58 (8), 1267–1269. 10.1016/s0031-9422(01)00382-x
6
AndrewsN.StoweJ.KirsebomF.ToffaS.RickeardT.GallagherE.et al (2022). Covid-19 vaccine effectiveness against the omicron (B.1.1.529) variant. N. Engl. J. Med.386, 1532–1546. 10.1056/NEJMoa2119451
7
AshhurstA. S.TangA. H.FajtováP.YoonM. C.AggarwalA.BeddingM. J.et al (2022). Potent anti-SARS-CoV-2 activity by the natural product gallinamide A and analogues via inhibition of cathepsin L. J. Med. Chem.65 (4), 2956–2970. 10.1021/acs.jmedchem.1c01494
8
BahunM.JukićM.OblakD.KranjcL.BajcG.ButalaM.et al (2022). Inhibition of the SARS-CoV-2 3CLpro main protease by plant polyphenols. Food Chem.373, 131594. 10.1016/j.foodchem.2021.131594
9
Barbosa Da SilvaE.SharmaV.Hernandez-AlvarezL.TangA. H.StoyeA.O’DonoghueA. J.et al (2022). Intramolecular interactions enhance the potency of gallinamide A analogues against Trypanosoma cruzi. J. Med. Chem.65 (5), 4255–4269. 10.1021/acs.jmedchem.1c02063
10
BennettR. P.PostnikovaE. N.EatonB. P.CaiY.YuS.SmithC. O.et al (2022). Sangivamycin is highly effective against SARS-CoV-2 in vitro and has favorable drug properties. JCI insight7 (1), e153165. 10.1172/jci.insight.153165
11
BibiS.HasanM. M.WangY. B.PapadakosS. P.YuH. (2022). Cordycepin as a promising inhibitor of SARS-CoV-2 RNA dependent RNA polymerase (RdRp). Curr. Med. Chem.29 (1), 152–162. 10.2174/0929867328666210820114025
12
CuiJ.JiaJ. (2021). Discovery of juglone and its derivatives as potent SARS-CoV-2 main proteinase inhibitors. Eur. J. Med. Chem.225, 113789. 10.1016/j.ejmech.2021.113789
13
CaohuyH.EidelmanO.ChenT.LiuS.YangQ.BeraA.et al (2021). Common cardiac medications potently inhibit ACE2 binding to the SARS-CoV-2 Spike, and block virus penetration and infectivity in human lung cells. Sci. Rep.11, 22195. 10.1038/s41598-021-01690-9
14
CaohuyH.EidelmanO.ChenT.YangQ.WaltonN. I.PollardH. B.et al (2022). Inflammation in the COVID-19 airway is due to inhibition of CFTR signaling by the SARS-CoV-2 Spike protein. bioRxiv[Preprint]. 10.1101/2022.01.18.476803
15
Chable-BessiaC.BoulléC.NeyretA.SwainJ.HénautM.MeridaP.et al (2022). Low selectivity indices of ivermectin and macrocyclic lactones on SARS-CoV-2 replication in vitro. COVID2, 60–75. 10.3390/covid2010005
16
ChandrashekarA.YuJ.McMahanK.Jacob-DolanC.LiuJ.HeX.et al (2022). Vaccine protection against the SARS-CoV-2 Omicron variant in macaques. Cell.185 (9), 1549–1555.e11. e11. 10.1016/j.cell.2022.03.024
17
ChenZ. H.HanY. Y.ShangY. J.ZhuangS. Y.HuangJ. N.WuB. Y.et al (2021). Cordycepin ameliorates synaptic dysfunction and dendrite morphology damage of hippocampal CA1 via A1R in cerebral ischemia. Front. Cell. Neurosci.15, 783478. 10.3389/fncel.2021.783478
18
ChhetriB. K.TedburyP. R.Sweeney-JonesA. M.ManiL.SoapiK.ManfrediC.et al (2022). Marine natural products as leads against SARS-CoV-2 infection. J. Nat. Prod.85, 657–665. 10.1021/acs.jnatprod.2c00015
19
ChiouW. C.ChenJ. C.ChenY. T.YangJ. M.HwangL. H.LyuY. S.et al (2022). The inhibitory effects of PGG and EGCG against the SARS-CoV-2 3C-like protease. Biochem. Biophys. Res. Commun.591, 130–136. 10.1016/j.bbrc.2020.12.106
20
ChiouW. C.LuH. F.HsuN. Y.ChangT. Y.ChinY. F.LiuP. C.et al (2021). Ugonin J acts as a SARS-CoV-2 3C-like protease inhibitor and exhibits anti-inflammatory properties. Front. Pharmacol.12, 720018. 10.3389/fphar.2021.720018
21
ChoiS. J.RyuE.LeeS.HuhS.ShinY. S.KangB. W.et al (2019). Adenosine induces EBV lytic reactivation through ADORA1 in EBV-associated gastric carcinoma. Int. J. Mol. Sci.20 (6), 1286. 10.3390/ijms20061286
22
CoronaA.WyciskK.TalaricoC.ManelfiC.MiliaJ.CannalireR.et al (2022). Natural compounds inhibit SARS-CoV-2 nsp13 unwinding and ATPase enzyme activities. ACS Pharmacol. Transl. Sci.5, 226–239. 10.1021/acsptsci.1c00253
23
CorpettiC.Del ReA.SeguellaL.PalencaI.RurgoS.De ConnoB.et al (2021). Cannabidiol inhibits SARS‐Cov‐2 spike (S) protein‐induced cytotoxicity and inflammation through a PPARγ‐dependent TLR4/NLRP3/Caspase‐1 signaling suppression in Caco‐2 cell line. Phytother. Res.35 (12), 6893–6903. 10.1002/ptr.7302
24
CuiH. R.ChenK. D.ZhangX. Y.ShangH. C. (2021). Network pharmacology-based analysis on bioactive compounds and mechanisms in Yiqifumai formula in the treatment of heart failure. TMR Mod. Herb. Med.4 (4), 27. 10.53388/mhm2021a1017001
25
CullyM. (2022). A tale of two antiviral targets—And the COVID-19 drugs that bind them. Nat. Rev. Drug Discov.21 (1), 3–5. 10.1038/d41573-021-00202-8
26
CunninghamK. G.MansonW. I. L. L. I. A. M.SpringF. S.HutchinsonS. A. (1950). Cordycepin, a metabolic product isolated from cultures of Cordyceps militaris (Linn.) Link. Nature166 (4231), 949. 10.1038/166949a0
27
De JinX. A.ZhangY.ZhaoS.DuanL.DuanY.LianF.et al (2021). Potential mechanism prediction of herbal medicine for pulmonary fibrosis associated with SARS-CoV-2 infection based on network analysis and molecular docking. Front. Pharmacol.12, 602218. 10.3389/fphar.2021.602218
28
Del RioC.OmerS. B.MalaniP. N. (2022). Winter of omicron-the evolving COVID-19 pandemic.JAMA327 (4), 319–320. 10.1001/jama.2021.24315
29
DuR.CooperL.ChenZ.LeeH.RongL.CuiQ.et al (2021). Discovery of chebulagic acid and punicalagin as novel allosteric inhibitors of SARS-CoV-2 3CLpro. Antivir. Res.190, 105075. 10.1016/j.antiviral.2021.105075
30
DuXinyiXuLongxinMaYimingLuShuaiyaoTangKegongQiaoXiangyuet al (2022). Herbal inhibitors of SARS-CoV-2 Mpro effectively ameliorate acute lung injury in mice. IUBMB Life74, 532–542. 10.1002/iub.2616
31
ElebeedyD.BadawyI.ElmaatyA. A.SalehM. M.KandeilA.GhanemA.et al (2022). In vitro and computational insights revealing the potential inhibitory effect of Tanshinone IIA against influenza A virus. Comput. Biol. Med.141, 105149. 10.1016/j.compbiomed.2021.105149
32
EsharkawyE. R.AlmalkiF.HaddaT. B. (2022). In vitro potential antiviral SARS-CoV-19-activity of natural product thymohydroquinone and dithymoquinone from Nigela sativia. Bioorg. Chem.120, 105587. 10.1016/j.bioorg.2021.105587
33
GaoX.QinB.ChenP.ZhuK.HouP.WojdylaJ. A.et al (2021). Crystal structure of SARS-CoV-2 papain-like protease. Acta Pharm. Sin. B11 (1), 237–245. 10.1016/j.apsb.2020.08.014
34
Guisado-VascoP.Carralón-GonzálezM. M.Aguareles-GorinesJ.Martí-BallesterosE. M.Sánchez-ManzanoM. D.Carnevali-RuizD.et al (2022). Plitidepsin as a successful rescue treatment for prolonged viral SARS-CoV-2 replication in a patient with previous anti-CD20 monoclonal antibody-mediated B cell depletion and chronic lymphocytic leukemia. J. Hematol. Oncol.15, 4. 10.1186/s13045-021-01220-0
35
HillenH. S.KokicG.FarnungL.DienemannC.TegunovD.CramerP.et al (2020). Structure of replicating SARS-CoV-2 polymerase. Nature584 (7819), 154–156. 10.1038/s41586-020-2368-8
36
HoffmannM.Kleine-WeberH.SchroederS.KrügerN.HerrlerT.ErichsenS.et al (2020). SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell.181 (2), 271–280. e8. 10.1016/j.cell.2020.02.052
37
HossainR.SarkarC.HassanS. M. H.KhanR. A.ArmanM.RayP.et al (2022). In silico screening of natural products as potential inhibitors of SARS-CoV-2 using molecular docking simulation. Chin. J. Integr. Med.28 (3), 249–256. 10.1007/s11655-021-3504-5
38
HouB.ZhangY. M.LiaoH. Y.FuL. F.LiD. D.ZhaoX.et al (2022). Target-based virtual screening and LC/MS-guided isolation procedure for identifying phloroglucinol-terpenoid inhibitors of SARS-CoV-2. J. Nat. Prod.85 (2), 327–336. 10.1021/acs.jnatprod.1c00805
39
HuangY.ZhouW.SunJ.OuG.ZhongN. S.LiuZ.et al (2022). Exploring the potential pharmacological mechanism of hesperidin and glucosyl hesperidin against COVID-19 based on bioinformatics analyses and antiviral assays. Am. J. Chin. Med.50 (2), 351–369. 10.1142/S0192415X22500148
40
IshidaJ.WangH. K.OyamaM.CosentinoM. L.HuC. Q.LeeK. H.et al (2001). Anti-AIDS agents. 46.1 Anti-HIV activity of harman, an anti-HIV principle from Symplocos setchuensis, and its derivatives. J. Nat. Prod.64 (7), 958–960. 10.1021/np0101189
41
JinY. H.JeonS.LeeJ.KimS.JangM. S.ParkC. M.et al (2021). Broad spectrum antiviral properties of cardiotonic steroids used as potential therapeutics for emerging coronavirus infections. Pharmaceutics13 (11), 1839. 10.3390/pharmaceutics13111839
42
JinZ.DuX.XuY.DengY.LiuM.ZhaoY.et al (2020). Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature582 (7811), 289–293. 10.1038/s41586-020-2223-y
43
KalilA. C.MehtaA. K.PattersonT. F.ErdmannN.GomezC. A.JainM. K.et al (2021). Efficacy of interferon beta-1a plus remdesivir compared with remdesivir alone in hospitalised adults with COVID-19: A double-bind, randomised, placebo-controlled, phase 3 trial. Lancet. Respir. Med.9 (12), 1365–1376. 10.1016/S2213-2600(21)00384-2
44
KatoY.HigashiyamaA.TakaokaE.NishikawaM.IkushiroS. (2021). Food phytochemicals, epigallocatechin gallate and myricetin, covalently bind to the active site of the coronavirus main protease in vitro. Adv. Redox Res.3, 100021. 10.1016/j.arres.2021.100021
45
KozlovM. (2022). Merck's COVID pill loses its lustre: What that means for the pandemic. Nature[Epub ahead of print]. 10.1038/d41586-021-03667-0
46
LewisD. S.HoJ.WillsS.KawallA.SharmaA.ChavadaK.et al (2022). Aloin isoforms (A and B) selectively inhibits proteolytic and deubiquitinating activity of papain like protease (PLpro) of SARS-CoV-2 in vitro. Sci. Rep.12, 2145. 10.1038/s41598-022-06104-y
47
LiX.GaoJ.LiM.CuiH.JiangW.TuZ. C.et al (2021a). Aromatic cadinane sesquiterpenoids from the fruiting bodies of Phellinus pini block SARS-CoV-2 Spike–ACE2 interaction. J. Nat. Prod.84 (8), 2385–2389. 10.1021/acs.jnatprod.1c00426
48
LiY.LiB.WangP.WangQ. (2021b). Traditional Chinese medicine, Qingfei Paidu decoction and xuanfei baidu decoction, inhibited cytokine production via NF-κB signaling pathway in macrophages: Implications for coronavirus disease 2019 (COVID-19) therapy. Front. Pharmacol.12, 722126. 10.3389/fphar.2021.722126
49
LiangX. X.ZhangX. J.ZhaoY. X.FengJ.ZengJ. C.ShiQ. Q.et al (2022). Aspulvins A–H, aspulvinone analogues with SARS-CoV-2 Mpro inhibitory and anti-inflammatory activities from an endophytic Cladosporium sp. J. Nat. Prod.85 (4), 878–887. 10.1021/acs.jnatprod.1c01003
50
LiningtonR. G.ClarkB. R.TrimbleE. E.AlmanzaA.UreñaL. D.KyleD. E.et al (2009). Antimalarial peptides from marine cyanobacteria: Isolation and structural elucidation of gallinamide A. J. Nat. Prod.72 (1), 14–17. 10.1021/np8003529
51
LiuG.DuW.SangX.TongQ.WangY.ChenG.et al (2022). RNA G-quadruplex in TMPRSS2 reduces SARS-CoV-2 infection. Nat. Commun.13, 1444. 10.1038/s41467-022-29135-5
52
LiuN.LiS.FanK.LuT.LiT. (2020). The prevention and treatment of COVID-19 with Qingfei Paidu decoction in Shanxi China. TMR Mod. Herb. Med.3 (3), 1–5.
53
LyuM.FanG.XiaoG.WangT.XuD.GaoJ.et al (2021). Traditional Chinese medicine in COVID-19. Acta Pharm. Sin. B11 (11), 3337–3363. 10.1016/j.apsb.2021.09.008
54
MaC.TanH.ChozaJ.WangY.WangJ. (2022). Validation and invalidation of SARS-CoV-2 main protease inhibitors using the Flip-GFP and Protease-Glo luciferase assays. Acta Pharm. Sin. B12 (4), 1636–1651. 10.1016/j.apsb.2021.10.026
55
MaC.WangJ. (2022). Validation and invalidation of SARS-CoV-2 papain-like protease inhibitors. ACS Pharmacol. Transl. Sci.5 (2), 102–109. 10.1021/acsptsci.1c00240
56
MengistH. M.FanX.JinT. (2020). Designing of improved drugs for COVID-19: Crystal structure of SARS-CoV-2 main protease Mpro. Signal Transduct. Target. Ther.5, 67. 10.1038/s41392-020-0178-y
57
MillerB.FriedmanA. J.ChoiH.HoganJ.McCammonJ. A.HookV.et al (2014). The marine cyanobacterial metabolite gallinamide A is a potent and selective inhibitor of human cathepsin L. J. Nat. Prod.77 (1), 92–99. 10.1021/np400727r
58
NasreenS.ChungH.HeS.BrownK. A.GubbayJ. B.BuchanS. A.et al (2022). Effectiveness of COVID-19 vaccines against symptomatic SARS-CoV-2 infection and severe outcomes with variants of concern in Ontario. Nat. Microbiol.7 (3), 379–385. 10.1038/s41564-021-01053-0
59
NguyenL. C.YangD.NicolaescuV.BestT. J.GulaH.SaxenaD.et al (2022). Cannabidiol inhibits SARS-CoV-2 replication through induction of the host ER stress and innate immune responses. Sci. Adv.8, eabi6110. 10.1126/sciadv.abi6110
60
Ngwe TunM. M.ToumeK.LuvaiE.NweK. M.MizukamiS.HirayamaK.et al (2022). The discovery of herbal drugs and natural compounds as inhibitors of SARS-CoV-2 infection in vitro. J. Nat. Med.76, 402–409. 10.1007/s11418-021-01596-w
61
NishiuchiK.OhashiH.NishiokaK.YamasakiM.FurutaM.MashikoT.et al (2022). Synthesis and antiviral activities of neoechinulin B and its derivatives. J. Nat. Prod.85 (1), 284–291. 10.1021/acs.jnatprod.1c01120
62
OrdonezA. A.BullenC. K.Villabona-RuedaA. F.ThompsonE. A.TurnerM. L.MerinoV. F.et al (2022). Sulforaphane exhibits antiviral activity against pandemic SARS-CoV-2 and seasonal HCoV-OC43 coronaviruses in vitro and in mice. Commun. Biol.5, 242. 10.1038/s42003-022-03189-z
63
PanggabeanJ. A.AdigunaS. B. P.RahmawatiS. I.AhmadiP.ZainuddinE. N.BayuA.et al (2022). Antiviral activities of algal-based sulfated polysaccharides. Molecules27 (4), 1178. 10.3390/molecules27041178
64
PanyaA.SongprakhonP.PanwongS.JantakeeK.KaewkodT.TragoolpuaY.et al (2021). Cordycepin inhibits virus replication in dengue virus-infected Vero cells. Molecules26 (11), 3118. 10.3390/molecules26113118
65
PokharkarO.LakshmananH.ZyryanovG.TsurkanM. (2022). In silico evaluation of antifungal compounds from marine sponges against COVID-19-associated mucormycosis. Mar. Drugs20 (3), 215. 10.3390/md20030215
66
PrassasI.DiamandisE. P. (2008). Novel therapeutic applications of cardiac glycosides. Nat. Rev. Drug Discov.7 (11), 926–935. 10.1038/nrd2682
67
QuintanaV. M.PicciniL. E.ZénereJ. D. P.DamonteE. B.PonceM. A.CastillaV.et al (2016). Antiviral activity of natural and synthetic β-carbolines against dengue virus. Antivir. Res.134, 26–33. 10.1016/j.antiviral.2016.08.018
68
RabieA. M. (2022). Potent inhibitory activities of the adenosine analogue cordycepin on SARS-CoV-2 replication. ACS Omega7, 2960–2969. 10.1021/acsomega.1c05998
69
ReisG.SilvaE. A.SilvaD. C.ThabaneL.MilagresA. C.FerreiraT. S.et al (2022). Effect of early treatment with ivermectin among patients with Covid-19. N. Engl. J. Med.386, 1721–1731. 10.1056/NEJMoa2115869
70
RodonJ.Muñoz-BasagoitiJ.Perez-ZsoltD.Noguera-JulianM.ParedesR.MateuL.et al (2021). Identification of plitidepsin as potent inhibitor of SARS-CoV-2-induced cytopathic effect after a drug repurposing screen. Front. Pharmacol.12, 646676. 10.3389/fphar.2021.646676
71
Sa-NgiamsuntornK.SuksatuA.PewkliangY.ThongsriP.KanjanasiriratP.ManopwisedjaroenS.et al (2021). Anti-SARS-CoV-2 activity of Andrographis paniculata extract and its major component Andrographolide in human lung epithelial cells and cytotoxicity evaluation in major organ cell representatives. J. Nat. Prod.84, 1261–1270. 10.1021/acs.jnatprod.0c01324
72
SaadhM. J.AlmaaytahA. M.AlarajM.DababnehM. F.Sa'adehI.AldalaenS. M.et al (2021). Punicalagin and zinc (II) ions inhibit the activity of SARS-CoV-2 3CL-protease in vitro. Eur. Rev. Med. Pharmacol. Sci.25 (10), 3908–3913. 10.26355/eurrev_202105_25958
73
SachseM.TenorioR.de CastroI. F.Muñoz-BasagoitiJ.Perez-ZsoltD.Raïch-ReguéD.et al (2022). Unraveling the antiviral activity of plitidepsin against SARS-CoV-2 by subcellular and morphological analysis. Antivir. Res.200, 105270. 10.1016/j.antiviral.2022.105270
74
SahooA.FuloriaS.SwainS. S.PandaS. K.SekarM.SubramaniyanV.et al (2021). Potential of marine terpenoids against SARS-CoV-2: An in silico drug development approach. Biomedicines9 (11), 1505. 10.3390/biomedicines9111505
75
SchulteB.KönigM.EscherB. I.WittenburgS.ProjM.WolfV.et al (2022). Andrographolide derivatives target the KEAP1/NRF2 axis and possess potent anti‐SARS‐CoV‐2 activity. ChemMedChem17 (5), e202100732. 10.1002/cmdc.202100732
76
SchultzD. C.JohnsonR. M.AyyanathanK.MillerJ.WhigK.KamaliaB.et al (2022). Pyrimidine inhibitors synergize with nucleoside analogues to block SARS-CoV-2. Nature604, 134–140. 10.1038/s41586-022-04482-x
77
ScottL.HsiaoN. Y.MoyoS.SinghL.TegallyH.DorG.et al (2021). Track Omicron’s spread with molecular data. Science374 (6574), 1454–1455. 10.1126/science.abn4543
78
SharmaV.SharmaA.BharateS. B. (2021). Natural products in mitigation of SARS-CoV infections. Curr. Med. Chem.28 (22), 4454–4483. 10.2174/0929867327666201027153940
79
ShenY.CaiH.MaS.ZhuW.ZhaoH.LiJ.et al (2022). Telocinobufagin has antitumor effects in non-small-cell lung cancer by inhibiting STAT3 signaling. J. Nat. Prod.85 (4), 765–775. 10.1021/acs.jnatprod.1c00761
80
SocolskyC.DomínguezL.AsakawaY.BardónA. (2012). Unusual terpenylated acylphloroglucinols from Dryopteris wallichiana. Phytochemistry80, 115–122. 10.1016/j.phytochem.2012.04.017
81
SourimantJ.LieberC. M.AggarwalM.CoxR. M.WolfJ. D.YoonJ. J.et al (2022). 4′-Fluorouridine is an oral antiviral that blocks respiratory syncytial virus and SARS-CoV-2 replication. Science375 (6577), 161–167. 10.1126/science.abj5508
82
SuH.YaoS.ZhaoW.ZhangY.LiuJ.ShaoQ.et al (2021). Identification of pyrogallol as a warhead in design of covalent inhibitors for the SARS-CoV-2 3CL protease. Nat. Commun.12, 3623. 10.1038/s41467-021-23751-3
83
SuručićR.TravarM.PetkovićM.TubićB.StojiljkovićM. P.GrabežM.et al (2021). Pomegranate peel extract polyphenols attenuate the SARS-CoV-2 S-glycoprotein binding ability to ACE2 receptor: In silico and in vitro studies. Bioorg. Chem.114, 105145. 10.1016/j.bioorg.2021.105145
84
TaoriK.LiuY.PaulV. J.LueschH. (2009). Combinatorial strategies by marine cyanobacteria: Symplostatin 4, an antimitotic natural dolastatin 10/15 hybrid that synergizes with the coproduced HDAC inhibitor largazole. ChemBioChem10 (10), 1634–1639. 10.1002/cbic.200900192
85
ThisseraB.SayedA. M.HassanM. H.AbdelwahabS. F.AmaezeN.SemlerV. T.et al (2021). Bioguided isolation of cyclopenin analogues as potential SARS-CoV-2 Mpro inhibitors from Penicillium citrinum TDPEF34. Biomolecules11 (9), 1366. 10.3390/biom11091366
86
TianH. Y.RuanL. J.YuT.ZhengQ. F.ChenN. H.WuR. B.et al (2017). Bufospirostenin A and Bufogargarizin C, steroids with rearranged skeletons from the toad Bufo bufo gargarizans. J. Nat. Prod.80 (4), 1182–1186. 10.1021/acs.jnatprod.6b01018
87
ToelzerC.GuptaK.YadavS. K.BorucuU.DavidsonA. D.Kavanagh WilliamsonM.et al (2020). Free fatty acid binding pocket in the locked structure of SARS-CoV-2 spike protein. Science370 (6517), 725–730. 10.1126/science.abd3255
88
TsaiY. C.LeeC. L.YenH. R.ChangY. S.LinY. P.HuangS. H.et al (2020). Antiviral action of tryptanthrin isolated from Strobilanthes cusia leaf against human coronavirus NL63. Biomolecules10 (3), 366. 10.3390/biom10030366
89
UedaY.MoriK.SatohS.DansakoH.IkedaM.KatoN.et al (2014). Anti-HCV activity of the Chinese medicinal fungus Cordyceps militaris. Biochem. Biophys. Res. Commun.447 (2), 341–345. 10.1016/j.bbrc.2014.03.150
90
UrdaL.KreuterM. H.DreweJ.BoonenG.ButterweckV.KlimkaitT.et al (2022). The petasites hybridus CO2 extract (Ze 339) blocks SARS-CoV-2 replication in vitro. Viruses14, 106. 10.3390/v14010106
91
van BreemenR. B.MuchiriR. N.BatesT. A.WeinsteinJ. B.LeierH. C.FarleyS.et al (2022). Cannabinoids block cellular entry of SARS-CoV-2 and the emerging variants. J. Nat. Prod.85, 176–184. 10.1021/acs.jnatprod.1c00946
92
WallsA. C.ParkY. J.TortoriciM. A.WallA.McGuireA. T.VeeslerD.et al (2020). Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell.181 (2), 281–292. 10.1016/j.cell.2020.02.058
93
WangK.SuB. O.WangZ.WuM.LiZ.HuY.et al (2010). Synthesis and antiviral activities of phenanthroindolizidine alkaloids and their derivatives. J. Agric. Food Chem.58 (5), 2703–2709. 10.1021/jf902543r
94
WangZ. (2019). Advances in the asymmetric total synthesis of natural products using chiral secondary amine catalyzed reactions of α, β-unsaturated aldehydes. Molecules24 (18), 3412. 10.3390/molecules24183412
95
WangZ.YangL. (2022a). Broad‐spectrum prodrugs with anti‐SARS‐CoV‐2 activities: Strategies, benefits, and challenges. J. Med. Virol.94 (4), 1373–1390. 10.1002/jmv.27517
96
WangZ.YangL. (2021). Chinese herbal medicine: Fighting SARS-CoV-2 infection on all fronts. J. Ethnopharmacol.270, 113869. 10.1016/j.jep.2021.113869
97
WangZ.YangL. (2020a). GS-5734: A potentially approved drug by FDA against SARS-cov-2. New J. Chem.44 (29), 12417–12429. 10.1039/d0nj02656e
98
WangZ.YangL. (2022b). In the age of Omicron variant: Paxlovid raises new hopes of COVID‐19 recovery. J. Med. Virol.94 (5), 1766–1767. 10.1002/jmv.27540
99
WangZ.YangL. (2022c). Post-acute sequelae of SARS-CoV-2 infection: A neglected public health issue. Front. Public Health10, 908757. 10.3389/fpubh.2022.908757
100
WangZ.YangL. (2020b). Turning the tide: Natural products and natural-product-inspired chemicals as potential counters to SARS-CoV-2 infection. Front. Pharmacol.11, 1013. 10.3389/fphar.2020.01013
101
WangZ.YangL.ZhaoX. E. (2021a). Co-crystallization and structure determination: An effective direction for anti-SARS-CoV-2 drug discovery. Comput. Struct. Biotechnol. J.19, 4684–4701. 10.1016/j.csbj.2021.08.029
102
WangZ.YeF.FengY.XiaoW.SongH.ZhaoL.et al (2021b). Discovery and nanosized preparations of (S, R)-tylophorine malate as novel anti-SARS-CoV-2 agents. ACS Med. Chem. Lett.12 (11), 1840–1846. 10.1021/acsmedchemlett.1c00481
103
WeiP.WangK.LuoC.HuangY.MisilimuD.WenH.et al (2021). Cordycepin confers long-term neuroprotection via inhibiting neutrophil infiltration and neuroinflammation after traumatic brain injury. J. Neuroinflammation18 (1), 137. 10.1186/s12974-021-02188-x
104
WeiX. X.ZhaoM. Z.ZhaoC.ZhangX.QiuR.LinY.et al (2020). The global registry of COVID-19 clinical trials: Indicating the design of traditional Chinese medicine clinical trials. TMR Mod. Herb. Med.3 (3), 140–146.
105
WhiteK. M.RosalesR.YildizS.KehrerT.MiorinL.MorenoE.et al (2021). Plitidepsin has potent preclinical efficacy against SARS-CoV-2 by targeting the host protein eEF1A. Science371 (6532), 926–931. 10.1126/science.abf4058
106
World Health OrganizationWHO coronavirus (COVID-19) dashboard, 2022. Available at: https://covid19.who.int/(Assessed April 22, 2022).
107
WuH.ChengH.LuoS.PengC.ZhouA.ChenZ.et al (2022a). Use of cellular metabolomics and lipidomics to decipher the mechanism of Huachansu injection-based intervention against human hepatocellular carcinoma cells. J. Pharm. Biomed. Anal.212, 114654. 10.1016/j.jpba.2022.114654
108
WuQ.YanS.WangY.LiM.XiaoY.LiY.et al (2022b). Discovery of 4′-O-methylscutellarein as a potent SARS-CoV-2 main protease inhibitor. Biochem. Biophys. Res. Commun.604, 76–82. 10.1016/j.bbrc.2022.03.052
109
WuY.XuL.CaoG.MinL.DongT. (2022c). Effect and mechanism of Qingfei Paidu decoction in the management of pulmonary fibrosis and COVID-19. Am. J. Chin. Med.50 (1), 33–51. 10.1142/S0192415X22500021
110
XiaoT.CuiM.ZhengC.ZhangP.RenS.BaoJ.et al (2022). Both baicalein and gallocatechin gallate effectively inhibit SARS-CoV-2 replication by targeting Mpro and Sepsis in Mice. Inflammation45, 1076–1088. 10.1007/s10753-021-01602-z
111
XiaoT.WeiY.CuiM.LiX.RuanH.ZhangL.et al (2021). Effect of dihydromyricetin on SARS-CoV-2 viral replication and pulmonary inflammation and fibrosis. Phytomedicine.91, 153704. 10.1016/j.phymed.2021.153704
112
YanG.LiD.LinY.FuZ.QiH.LiuX.et al (2021). Development of a simple and miniaturized sandwich-like fluorescence polarization assay for rapid screening of SARS-CoV-2 main protease inhibitors. Cell. Biosci.11, 199. 10.1186/s13578-021-00720-3
113
YangL. J.ChenR. H.HamdounS.CoghiP.NgJ. P.ZhangD. W.et al (2021a). Corilagin prevents SARS-CoV-2 infection by targeting RBD-ACE2 binding. Phytomedicine.87, 153591. 10.1016/j.phymed.2021.153591
114
YangL.WangZ. (2021). Natural products, alone or in combination with FDA-approved drugs, to treat COVID-19 and lung cancer. Biomedicines9 (6), 689. 10.3390/biomedicines9060689
115
YangY.YangP.HuangC.WuY.ZhouZ.WangX.et al (2021b). Inhibitory effect on SARS‐CoV‐2 infection of neferine by blocking Ca2+‐dependent membrane fusion. J. Med. Virol.93 (10), 5825–5832. 10.1002/jmv.27117
116
YiY.LiJ.LaiX.ZhangM.KuangY.BaoY. O.et al (2022). Natural triterpenoids from licorice potently inhibit SARS-CoV-2 infection. J. Adv. Res.36, 201–210. 10.1016/j.jare.2021.11.012
117
YinW.MaoC.LuanX.ShenD. D.ShenQ.SuH.et al (2020). Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir. Science368 (6498), 1499–1504. 10.1126/science.abc1560
118
YuH.HongX.LiuL.WuY.XieX.FangG.et al (2021). Cordycepin decreases ischemia/reperfusion injury in diabetic hearts via upregulating AMPK/Mfn2-dependent mitochondrial fusion. Front. Pharmacol.12, 754005. 10.3389/fphar.2021.754005
119
ZhanY. Q.ChenR. F.MaQ. H.ZhengJ.DengX.YangW.et al (2022). Efficacy and safety of phillyrin (KD-1) capsule in the treatment of moderate COVID-19: Protocol for a randomized controlled trial. TMR Mod. Herb. Med.5 (1), 5. 10.53388/mhm2021p1204001
120
ZhangS.PeiR.LiM.SuH.SunH.DingY.et al (2022). Cocktail polysaccharides isolated from Ecklonia kurome against the SARS-CoV-2 infection. Carbohydr. Polym.275, 118779. 10.1016/j.carbpol.2021.118779
121
ZhaoJ.MaQ.ZhangB.GuoP.WangZ.LiuY.et al (2021). Exploration of SARS-CoV-2 3CLpro inhibitors by virtual screening methods, FRET detection, and CPE assay. J. Chem. Inf. Model.61 (12), 5763–5773. 10.1021/acs.jcim.1c01089
122
ZhongB.PengW.DuS.ChenB.FengY.HuX.et al (2022). Oridonin inhibits SARS‐CoV‐2 by targeting its 3C‐Like protease. Small Sci.2, 2100124. 10.1002/smsc.202100124
123
ZhouX.LuoL.DresselW.ShadierG.KrumbiegelD.SchmidtkeP.et al (2008). Cordycepin is an immunoregulatory active ingredient of Cordyceps sinensis. Am. J. Chin. Med.36 (05), 967–980. 10.1142/S0192415X08006387
Summary
Keywords
natural products, COVID-19, SARS-CoV-2, cordycepin, gallinamide A, plitidepsin, telocinobufagin, tylophorine
Citation
Wang Z, Wang N, Yang L and Song X (2022) Bioactive natural products in COVID-19 therapy. Front. Pharmacol. 13:926507. doi: 10.3389/fphar.2022.926507
Received
22 April 2022
Accepted
11 July 2022
Published
19 August 2022
Volume
13 - 2022
Edited by
Hidayat Hussain, Leibniz Institute of Plant Biochemistry, Germany
Reviewed by
Zeynab Fakhar, University of the Witwatersrand, South Africa
Ramendra K. Singh, Allahabad University, India
Updates
Copyright
© 2022 Wang, Wang, Yang and Song.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhonglei Wang, wangzl16@tsinghua.org.cn; Liyan Yang, yangly@iccas.ac.cn; Xian-qing Song, Song_xianqing@126.com
This article was submitted to Experimental Pharmacology and Drug Discovery, a section of the journal Frontiers in Pharmacology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.