- 1Interdisciplinary Gerontology Program, Department of Psychological Sciences, University of North Carolina at Charlotte, Charlotte, NC, United States
- 2Meharry-Vanderbilt Alliance, Vanderbilt University Medical Center, Nashville, TN, United States
- 3Sarah Cannon Research Institute, Sarah Cannon, Nashville, TN, United States
- 4National Health Care for the Homeless Council, Nashville, TN, United States
- 5Department of Medicine, Meharry Medical College, Nashville, TN, United States
Introduction: Innovative methods to increase awareness about clinical trials and address barriers associated with low participation among racial/ethnic minorities are desperately needed. African Americans comprise 5% of all clinical trial participants, and Hispanics make up 1%. Use of multimedia educational material has shown promise as an effective strategy to increase minority clinical trial enrollment. However, this approach has not been broadly implemented. We tested the effect of a video educational program on clinical trial knowledge and enrollment in a sample of oncology outpatients.
Methods: A randomized controlled trial was conducted with 63 oncology patients without previous history of clinical trial participation. Participants were randomly assigned to the intervention, to watch a clinical trial educational video in the office, or to the control group which did not receive in-office education. The Clinical Trial Knowledge survey was administered before the intervention and 1 week after the intervention. Participation in clinical trials was assessed 1-year post study participation. Results for white participants and ethnic minorities were compared. Ethnicity was self-reported through the electronic health record and confirmed by self-reporting on questionnaire.
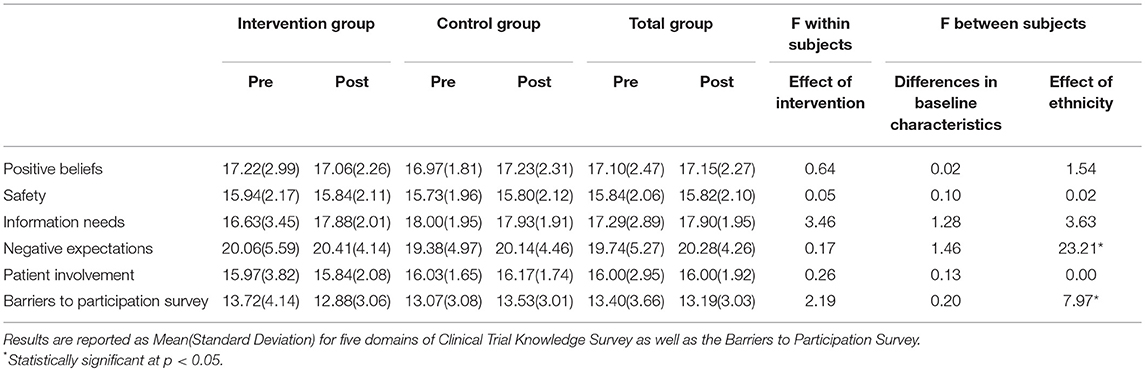
Results: Sixty-three participants were recruited in this study. At 1-year follow-up, 3 participants enrolled in clinical trials in the study group which had received office-based video intervention and 2 participants enrolled in the control group (Z = 0.39, p = 0.69). These results were not statistically significant. Impact of the intervention by ethnicity could not be assessed due to low total clinical trial enrollment. The video intervention did not change knowledge, attitudes, or barriers as measured by the Clinical Trial Knowledge Survey. Minority participants did report significantly more negative beliefs and barriers to participation than white participants.
Conclusions: Increasing awareness and knowledge about clinical trials in underrepresented communities is an important step to providing opportunities for participation. Future studies should focus on how to address the negative expectations of clinical trials and the greater information needs in minority populations. Tailored or personalized messaging may address negative perceptions of clinical trial participation.
Introduction
Clinical trial participation is low especially among racial/ethnic minorities. Studies show African Americans comprise 5% of all clinical trial participants, and Hispanics make up 1% (1). Only 3–5% of cancer patients enroll in clinical trials, with racial/ethnic minorities making up a small fraction of enrollees (2, 3). Cancer incidence rates are typically lower among racial/ethnic minorities than non-Hispanic Whites, yet, minorities have a higher risk of mortality and shorter survival than non-Hispanic Whites (4, 5). For this reason, minority participation in clinical trials has important implications for improving health equity and addressing ethnoracial health disparities (6).
Limited awareness and knowledge about clinical trials (7, 8) are key personal factors that impede a prospective participant's ability to decide on whether he/she would like to participate in a clinical trial (8). Studies show clinical trial awareness and knowledge is associated with sociodemographic and economic factors, such that younger individuals, Whites and persons of higher socioeconomic status (9) have greater clinical trial awareness and knowledge, and are more likely to participate in clinical trial than persons who do not fit this sociodemographic profile. As such, initiatives focused on clinical trial education (10) and increasing health and scientific literacy among minorities (11) may be particularly effective in reducing barriers to clinical trial participation in these groups. Video-based education may be especially effective in increasing knowledge about research (12), improving attitudes toward research (13), and increasing enrollment in research studies (1) among hard-to-reach populations. Clinical research education provided through multimedia may also be favored among those with limited health and scientific literacy (14, 15).
We undertook a study to (a) describe the utility of a clinical trial educational video in a diverse oncology patient population, and (b) examine the preliminary effect of an office-based clinical trial education video intervention on clinical trial knowledge, perceived barriers, and clinical trial enrollment. We also explored differences in clinical trial knowledge and barriers to participants between minority participants and White participants. The feasibility of implementing this intervention in a clinic setting was conducted using the Stages of Implementation Completion (SIC) measure (16). Findings from this study will add to the existing literature on effective strategies to increase minority group participation in clinical trial research.
Materials and Methods
Study Design
A randomized control trial design was conducted to determine preliminary effects of a clinical trial education video on clinical knowledge, perceptions of barriers to participating in clinical studies, willingness to participate in clinical trials and clinical trial enrollment. The Vanderbilt University Medical Center Institutional Review Board approved this study.
Participants and Setting
A power size calculation (power of 0.80, a 0.05 significance level) based on a previous study (17) with similar goals and methodology indicated a sample size of 40 (20 for each group) would provide confidence that the resulting effect size represents that which would be expected in a fully powered study. We sought to include in our sample a matched proportion of participants of racial/ethnic minorities. Inclusion criteria were a diagnosis of malignancy, age of 18 years or older, English proficiency, and no previous history of clinical trial participation. Prospective participants were recruited from urology, hematology and breast specialty clinics of Vanderbilt Ingram Cancer Center (VICC).
Intervention
Participants were randomized to the intervention (in-office video viewing group) or control, (DVD to take home, usual care group) using the web-based program Research Randomizer (4.0). Both groups received a clinical trial educational booklet and a copy of the DVD video on cancer clinical trials. The booklet provided a definition of clinical trial, descriptions of different types of clinical trials, and potential benefits and risks associated with participating in clinical trials. The video depicted oncology patient advocates sharing personal stories of participation in clinical trials and interviews with oncologists discussing the importance of cancer clinical trials. The video was created by the Vanderbilt-Ingram Cancer Center Office of Patient and Community Education and is distributed as an educational resource to new oncology patients. Using a tablet computer and headphones, the intervention (video viewing) group viewed the video while in the clinic. The control group was provided a copy of the booklet and video and given no further instruction.
Measures
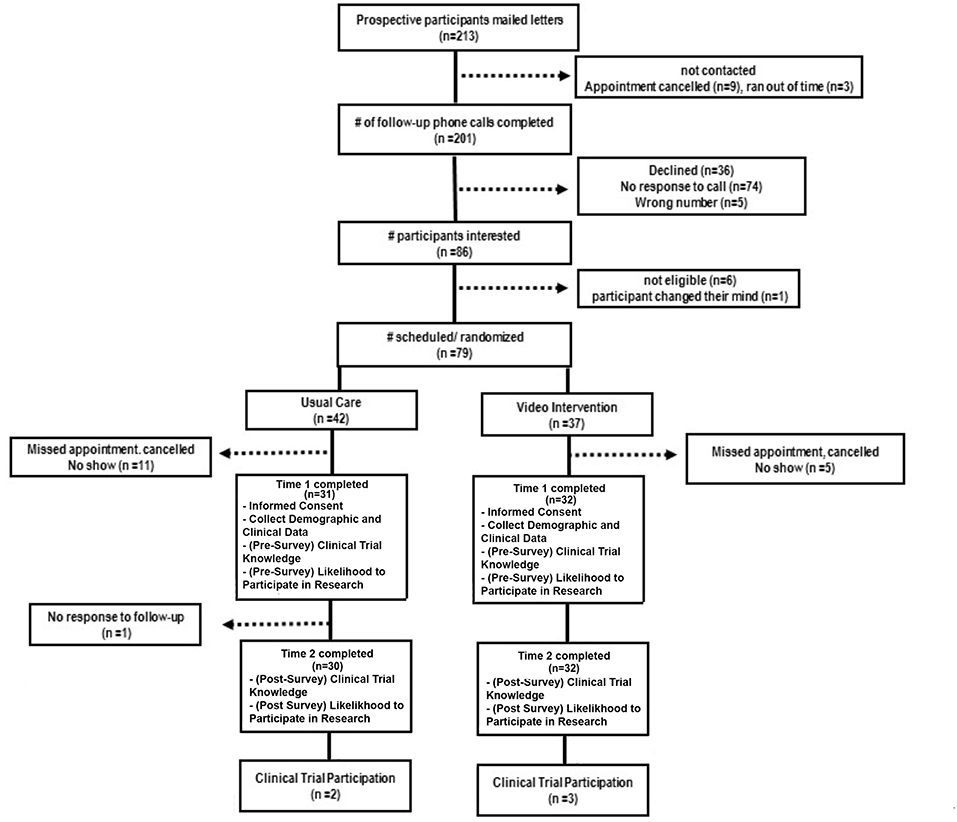
After randomization, all participants were asked to complete a pre-survey prior to receiving the educational resource intervention. A post-survey was conducted by phone 1 week later. A participant flow diagram is provided in Figure 1. The 22-item Clinical Trial Knowledge survey was used to assess participants' awareness about clinical trial research (18). Survey items span 5 independent subscales [positive beliefs (4-items), safety (4-items), information needs (4-items), negative expectations (6-items), and patient involvement (4-items)] and are measured on a 5-point Likert scale ranging from 1 (strongly disagree) to 5 (strongly agree). Perceived barriers to participation in clinical trials were assessed with 8-items on a 3-point scale ranging from 1 (this is not a reason for me), 2 (this is a minor reason for me), and 3 (this is a major reason for me) (unpublished observations, Patient-Centered Clinical Research Network, 2014). Participants were provided a list of reasons he/she may not want to participate in medical research studies and asked to indicate their feelings. Clinical trial knowledge and barriers to participation survey questions are provided in Supplementary Material. Enrollment in clinical trials was assessed 1-year post follow-up visit by review of VICC clinical trial participation electronic record. Study feasibility was measured using the Stages of Implementation Completion (SIC) measure an 8-stage, validated tool, measuring the implementation process across three phases (pre-implementation, implementation, and sustainability) (16).
Procedures
Potential participants were identified using Subject Locator, a recruitment tool to identify potential research participants based on discrete study inclusion/exclusion criteria available in Vanderbilt University Medical Center clinical systems. Study participant recruitment was streamlined by pre-screening patients from a list of upcoming appointments at the Vanderbilt Ingram Cancer Center. The resulting subset of clinic patients matched our study criteria and followed recruitment work flow.
To ensure sufficient representation of racial/ethnic minorities, the prospective participant pool was oversampled for racial/ethnic minorities at a ratio of 2:1. Prospective participants received a recruitment letter by mail and a follow-up phone call to determine interests and confirm eligibility for study participation. Study appointments were scheduled before or after the patient's next clinic visit. Once study appointments were scheduled, staff randomly assigned participants to the intervention or control condition.
All participants completed the pre-survey using android-based tablet computers equipped with Talking Survey™ software. Talking Survey™1 is an integrated patient surveying and healthcare education system. Key features include multilingual abilities, voice-over question administration, voice response option, and audio-to-text transcription. Participant data was automatically transmitted to the secure Research Electronic Data Capture (REDCap) database (19). Voice-over survey administration and touch screen response were used. After completing the pre-survey, participants in the video-viewing group watched the 10-min clinical trial education video. Participants in the control group were provided with the educational booklet and a DVD copy of the video after completing the survey. One week after completing the pre-survey, all participants completed the post-survey via a phone call. Approximately 1-year after participating in the study, participant's VICC clinical trial record system was queried to determine whether the participant enrolled in a VICC-affiliated clinical trial study. Participants received $50.00 after completing the post-survey.
Statistical Analyses
Results are reported using standard descriptive statistics. A mixed ANOVA was performed to assess the impact of the two interventions in the scores of the survey before and after the intervention. Race was converted into a dichotomous variable (whites/minorities) and included into the model as a covariate to assess its effects. Change scores from pre-survey to post-survey were calculated for each dependent variable and these values were used in the analyses. A series of one-way between-group analyses of variance were conducted to determine the impact of the experimental video vs. control on dependent variables. Between-group differences in clinical trial enrollment at 1-year post follow-up were analyzed using chi-square analysis. Significance was set at 0.05 level. Data was analyzed using Statistical Package for Social Sciences (version 23).
Results
Participants
Sixty-three participants were recruited. Baseline demographic characteristics are provided in Table 1. The intervention group and control group did not differ in age, gender makeup, level of education, or household income (p > 0.05). Hispanics were present in the video group but not the control group (p = 0.04). The majority of participants (53.2%) reported not reviewing either the educational booklet or video at home. There were no differences (Z = 0.39, p = 0.69) in the proportion of subjects who enrolled in a clinical trial after the video intervention (n = 3) and the control intervention (n = 2) (Table 2). This result could not be compared for minorities and whites due to low total clinical trial enrollment at the end of the study. We also studied the results of the survey by ethnic group to assess differences in clinical trial knowledge. Between groups analysis showed that the intervention did not have a significant impact on any of the domains assessed by the Clinical Trial Knowledge Survey or barriers to participation survey (Table 2). Within groups analysis did not demonstrate significant differences in baseline survey scores for the intervention and control groups. Within groups, minority participants were significantly more likely to harbor negative expectations of clinical trials (F = 23.21) and report higher barriers to participation (F = 7.97) irrespective of randomization arm (Figure 2 and Table 2).
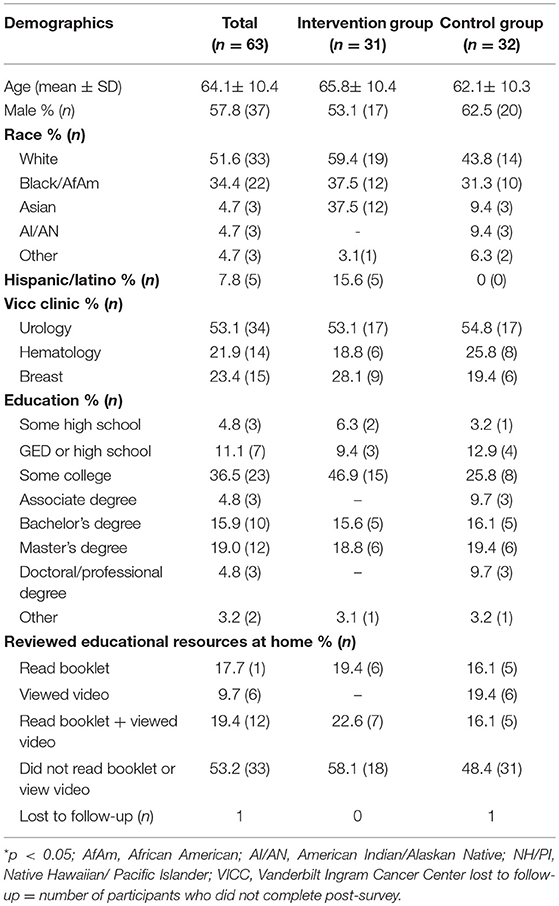
Table 1. Baseline demographic characteristics for the total study population, intervention group, and control group.
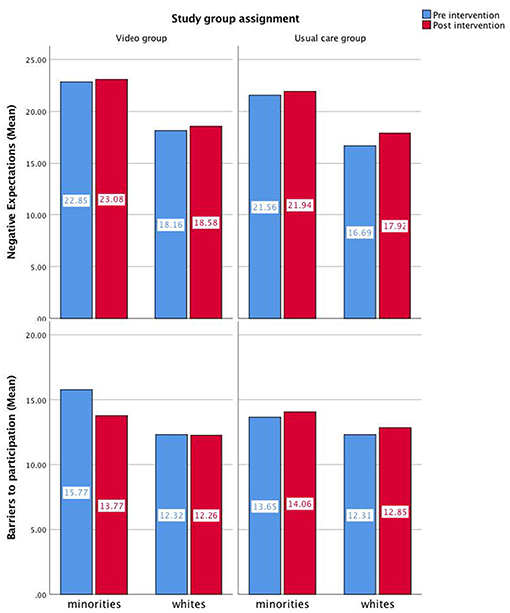
Figure 2. Pre and post intervention mean values of barriers to participation and negative expectations for each group of intervention by ethnicity.
Discussion
In this study we describe the utility of multimedia clinical trial educational resources and investigate the preliminary effect of a video educational program on clinical trial knowledge and subsequent enrollment in clinical trials in a diverse sample of oncology outpatients.
Our pilot randomized controlled trial revealed a null effect of video intervention on clinical trial knowledge or subsequent enrollment in clinical trials. In the control group, nearly half of participants reported not reading the educational booklet or watching the video provided to them, and only 19% in the video group reported reading the education booklet. These findings put into question the usefulness of multimedia technology as a method of health education and recruitment into clinical trials. Moreover, the study found that, within the randomization arms, minority participants were more likely to experience negative expectations of clinical trials and perceived more barriers to clinical trial participation than white participants which were not sufficiently addressed by the in-office video presentation. These findings align with prior work documenting minority group concerns about participation in clinical research, including clinical trials (1, 20–22).
These study findings suggest that alternative forms of communication be used to improve clinical trial knowledge and address barriers to participation which disproportionately affect minority groups (23). Some evidence suggests African Americans and Whites differ in their perceptions of effective communication channels for clinical trial information such that African Americans prefer to receive easy-to-understand clinical trial information through faith-based organizations and other in-person community-based channels, while Whites prefer to receive clinical trial information from doctors and print media (22). African Americans also express a preference for peer concordance representation in cancer information advertisements (24). Visual representation of extended family networks in cancer clinical trial educational videos provided to Hispanic cancer patients received greater clinical trial uptake, as it highlights cultural aspects of family input in patient decision-making central to Hispanic/Latino culture (20).
This study has several limitations. First, our sample size calculation based on previous studies
With similar goals and methodology indicated a sample size of 40, despite recruiting a total of 63 participants for this study. We did not account for stratification by ethnicity in our calculation of the sample size. This makes our results underpowered and hinders our ability to generalize findings, as studies with larger sample sizes are needed to confirm our results. Further studies should also apply stratified randomization and an adjusted sample size calculation to control for minority oversampling. Our results should be interpreted with caution as our study did not stratify randomization nor adjusted the sample size calculation by ethnicity.
To address the concerns about the combined analysis of the groups, we conducted between and within group analysis using ANOVA methods. Second, when assessing clinical trial enrollment 1-year post study participation, we did not query whether participants were asked to participate. It is possible some participants were never asked to participate in a clinical trial during the follow-up period. Despite these limitations, this study is unique in that it is one of the first to administer an interactive, tablet-based clinical trial educational video and survey in a clinic setting to a diverse patient population. Our use of interactive technology helps to circumvent research participation barriers related to lack of awareness about clinical trials, low literacy, and accessibility (25). Other strengths of our study include its RCT design and 50% minority group representation in the study population.
Interpersonal trust within the clinical relationship and medical establishment has been shown to be a significant factor in enhancing minority participation in clinical trials (26–28). A recent systematic review on barriers and facilitators to minority research participation recognized mistrust as a barrier to clinical trial participation (29). Despite expressions of mistrust, minority groups were willing to participate in clinical trials for altruistic reasons benefitting their family and community. Facilitators to clinical trial participation were illustrated as adapting culturally congruent practices such as addressing gaps in knowledge about research among a particular minority group (30, 31) translating study materials into appropriate languages and involvement of culturally and linguistically competent research staff (29, 32–35).
This study provides a guiding framework for future efforts to most effectively address and educate diverse patient populations about clinical trials for increased diversity in clinical research.
Ethics Statement
This study was carried out in accordance with the recommendations of the Vanderbilt University Institutional Review Board, Behavioral Sciences Committee, a sub-committee of the Institutional Review Board with written consent from all subjects. All subjects gave written informed consent in accordance with the Declaration of Helsinki. The protocol was approved by the Vanderbilt University Institutional Review Board, Behavioral Sciences Committee, a sub-committee of the Institutional Review Board.
Author Contributions
CW is the principal investigator of the study who designed the study and coordinated all aspects of the research including all steps of the manuscript preparation. She is responsible for the study concept, design, writing, reviewing, editing and approving the manuscript in its final form. JS and AH contributed in the study design and data collection. JS, AF, and AB contributed to the analysis and interpretation of data, drafting the work, writing the manuscript and reviewed and approved the manuscript. All authors read and approved the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Trial Registration: https://clinicaltrials.gov/ NCT02600533 registered 20 November 2015. Vanderbilt-Ingram Cancer Center and Cancer Center Support Grant (P30 CA068485), Research Electronic Database Capture (REDCap) and Subject Locator (UL1 TR000445) provided support for this research from NCATS/NIH. Thank you to the Vanderbilt University Information Technology Department, Vanderbilt University Research Informatics Core departments, Talking Survey™LLC, Middle Tennessee State University and the Tennessee Cancer Coalition Student Internship Program.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpubh.2019.00104/full#supplementary-material
Footnote
1. ^Talking Survey LLC: Talking Survey. Available online at: www.talkingsurvey.com.
References
1. Coakley M, Fadiran EO, Parrish LJ, Griffith RA, Weiss E, Carter C. (2012). Dialogues on diversifying clinical trials: successful strategies for engaging women and minorities in clinical trials. J Women's Health. 21:713–6. doi: 10.1089/jwh.2012.3733
2. Bell JA, Balneaves LG. Cancer patient decision making related to clinical trial participation: an integrative review with implications for patients' relational autonomy. Support Care Cancer. (2015) 23:1169–96. doi: 10.1007/s00520-014-2581-9
3. Unson CG, Dunbar N, Curry L, Kenyon L, Prestwood K. The effects of knowledge, attitudes, and significant others on decisions to enroll in a clinical trial on osteoporosis: implications for recruitment of older African-American women. J Natl Med Assoc. (2001) 93:392–401. discussion 402–394.
5. Byers TE, Wolf HJ, Bauer KR, Bolick-Aldrich S, Chen VW, Finch JL, et al. The impact of socioeconomic status on survival after cancer in the United States: findings from the National Program of Cancer Registries Patterns of Care Study. Cancer. (2008) 113:582–91. doi: 10.1002/cncr.23567
6. Fisher JA, Kalbaugh CA. Challenging assumptions about minority participation in US clinical research. Am J Public Health. (2011) 101:2217–22. doi: 10.2105/AJPH.2011.300279
7. Beachamp TL, Childress JF. Principles of Biomedical Ethics. New York, NY: Oxford University Press, (2012).
8. Ford JG, Howerton MW, Lai GY, Gary TL, Bolen S, Gibbons MC, et al. Barriers to recruiting underrepresented populations to cancer clinical trials: a systematic review. Cancer. (2008) 112:228–42. doi: 10.1002/cncr.23157
9. Sateren WB, Trimble EL, Abrams J, Brawley O, Breen N, Ford L, et al. How sociodemographics, presence of oncology specialists, and hospital cancer programs affect accrual to cancer treatment trials. J Clin Oncol. (2002) 20:2109–17. doi: 10.1200/JCO.2002.08.056
10. Caldwell PH, Hamilton S, Tan A, Craig JC. Strategies for increasing recruitment to randomised controlled trials: systematic review. PLoS Med. (2010) 7:e1000368. doi: 10.1371/journal.pmed.1000368
11. Evans KR, Lewis MJ, Hudson SV. The role of health literacy on African American and Hispanic/Latino perspectives on cancer clinical trials. J Cancer Educ. (2012) 27:299–305. doi: 10.1007/s13187-011-0300-5
12. Hutchison C, Cowan C, McMahon T, Paul J. A randomised controlled study of an audiovisual patient information intervention on informed consent and recruitment to cancer clinical trials. Br J Cancer. (2007) 97:705–11. doi: 10.1038/sj.bjc.6603943
13. Du W, Mood D, Gadgeel S, Simon MS. An educational video to increase clinical trials enrollment among lung cancer patients. J Thorac Oncol. (2008) 3:23–9. doi: 10.1097/JTO.0b013e31815e8bb2
14. Borrayo EA. Where's Maria? A video to increase awareness about breast cancer and mammography screening among low-literacy Latinas. Prevent Med. (2004) 39:99–110. doi: 10.1016/j.ypmed.2004.03.024
15. George S, Moran E, Duran N, Jenders RA. Using animation as an information tool to advance health research literacy among minority participants. AMIA Ann Symp Proc AMIA Symp. (2013) 2013:475–84.
16. Chamberlain P, Brown CH, Saldana L. Observational measure of implementation progress in community based settings: the Stages of Implementation Completion (SIC). Implement Sci. (2011) 6:116. doi: 10.1186/1748-5908-6-116
17. Ma GX, Tan Y, Blakeney NC, Seals BF, Ma XS, Zhai S, et al. The impact of a community-based clinical trial educational intervention among underrepresented Chinese Americans. Cancer Epidemiol Biomarkers Prevent. (2014) 23:424–32. doi: 10.1158/1055-9965.EPI-13-0773
18. Jenkinson C, Burton JS, Cartwright J, Magee H, Hall I, Alcock C, et al. Patient attitudes to clinical trials: development of a questionnaire and results from asthma and cancer patients. Health Expect. (2005) 8:244–52. doi: 10.1111/j.1369-7625.2005.00335.x
19. Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. (2009) 42:377–81. doi: 10.1016/j.jbi.2008.08.010
20. Quinn GP, McIntyre J, Gonzalez LE, Antonia TM, Antolino P, Wells KJ. Improving awareness of cancer clinical trials among Hispanic patients and families: audience segmentation decisions for a media intervention. J Health Commun. (2013) 18:1131–47. doi: 10.1080/10810730.2013.768723
21. Cook ED, Arnold KB, Hermos JA, McCaskill-Stevens W, Moody-Thomas S, Probstfield JL, et al. Impact of supplemental site grants to increase African American accrual for the Selenium and Vitamin E Cancer Prevention Trial. Clinical Trials. (2010) 7:90–9. doi: 10.1177/1470774509357227
22. Tanner A, Bergeron CD, Zheng Y, Friedman DB, Kim SH, Foster CB. Communicating effectively about clinical trials with african american communities: a comparison of african american and white information sources and needs. Health Promot Prac. (2016) 17:199–208. doi: 10.1177/1524839915621545
23. Germino BB, Mishel MH, Alexander GR, Jenerette C, Blyler D, Baker C, et al. Engaging African American breast cancer survivors in an intervention trial: culture, responsiveness and community. J Cancer Survivorship. (2011) 5:82–91. doi: 10.1007/s11764-010-0150-x
24. Munoz-Antonia T, Ung D, Montiel-Ishino FA, Nelson A, Canales J, Quinn GP. African Americans' and Hispanics' information needs about cancer care. J Cancer Educ. (2015) 30:327–32. doi: 10.1007/s13187-014-0721-z
25. Gysels M, Higginson IJ. Interactive technologies and videotapes for patient education in cancer care: systematic review and meta-analysis of randomised trials. Support Care Cancer. (2007) 15:7–20.
26. Corbie-Smith G, Thomas SB, Williams MV, Moody-Ayers S. Attitudes and beliefs of African Americans toward participation in medical research. J General Inter Med. (1999) 14:537–46.
27. Corbie-Smith G, Thomas SB, St George DMM. Distrust, race, and research. Arch Inter Med. (2002) 162:2458–63.
28. Shavers-Hornaday VL, Lynch CF, Burmeister LF, Torner JC. Why are African Americans under-represented in medical research studies? Impediments Participation Ethnicity Health. (1997) 2:31–45. doi: 10.1080/13557858.1997.9961813
29. George S, Duran N, Norris K. A systematic review of barriers and facilitators to minority research participation among African Americans, Latinos, Asian Americans, and Pacific Islanders. Am J Public Health. (2014) 104:e16–31. doi: 10.2105/AJPH.2013.301706
30. Banda DR, Libin AV, Wang H, Swain SM. A pilot study of a culturally targeted video intervention to increase participation of African American patients in cancer clinical trials. Oncologist. (2012) 17:708–14. doi: 10.1634/theoncologist.2011-0454
31. Lai GY, Gary TL, Tilburt J, Bolen S, Baffi C, Wilson RF, et al. Effectiveness of strategies to recruit underrepresented populations into cancer clinical trials. Clinical Trials. (2006) 3:133–41. doi: 10.1191/1740774506cn143oa
32. Larkey LK, Gonzalez JA, Mar LE, Glantz N. Latina recruitment for cancer prevention education via Community Based Participatory Research strategies. Contemp Clin Trials. (2009) 30:47–54. doi: 10.1016/j.cct.2008.08.003
33. Branson R D, Davis K Jr, Butler KL. African Americans' participation in clinical research: importance, barriers, and solutions. Am J Surg. (2007) 193:32–39; discussion 40. doi: 10.1016/j.amjsurg.2005.11.007
34. Rivers D, August EM, Sehovic I, Lee Green B, Quinn GP. A systematic review of the factors influencing African Americans' participation in cancer clinical trials. Contemp Clin Trials. (2013) 35:13–32. doi: 10.1016/j.cct.2013.03.007
Keywords: clinical trials, ethnic groups, minority groups, patients, cancer, education, multimedia
Citation: Skinner JS, Fair AM, Holman AS, Boyer AP and Wilkins CH (2019) The Impact of an Educational Video on Clinical Trial Enrollment and Knowledge in Ethnic Minorities: A Randomized Control Trial. Front. Public Health 7:104. doi: 10.3389/fpubh.2019.00104
Received: 07 January 2019; Accepted: 10 April 2019;
Published: 26 April 2019.
Edited by:
John D. Carpten, University of Southern California, United StatesReviewed by:
Giridhara R. Babu, Public Health Foundation of India, IndiaDanice Brown Greer, University of Texas at Tyler, United States
Copyright © 2019 Skinner, Fair, Holman, Boyer and Wilkins. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Consuelo H. Wilkins, Y29uc3VlbG8uaC53aWxraW5zQG1laGFycnktdmFuZGVyYmlsdC5vcmc=
†Deceased
 Jeannine S. Skinner1†
Jeannine S. Skinner1† Alecia M. Fair
Alecia M. Fair Consuelo H. Wilkins
Consuelo H. Wilkins