Abstract
Background:
Magnesium (Mg), an essential secondary macronutrient for plant growth and development, and its dose–response relationships with crop yield and quality remain to be systematically characterized.
Methods:
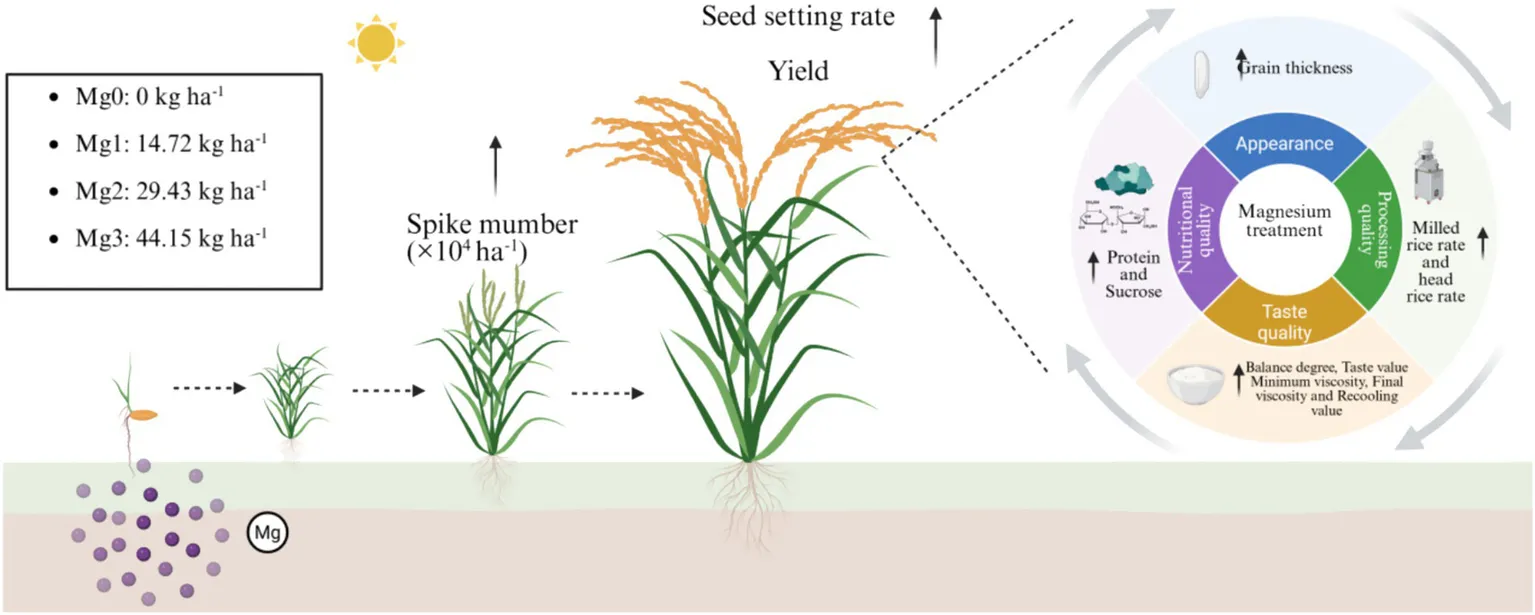
The experiment was conducted at Shenyang Agricultural University using the locally predominant rice cultivar “Liaojing 294.” A randomized block design was employed with four magnesium (Mg) treatment levels: Mg0 (no Mg), Mg1 (14.72 kg ha−1), Mg2 (29.43 kg ha−1), and Mg3 (44.15 kg ha−1). This study aimed to investigate the effects of Mg application rates on rice yield components, milling quality, cooking/eating quality, and nutritional composition.
Results:
(i) Mg application enhances yield per unit area by promoting tillering into panicles and grain filling; (ii) Mg improves rice pasting properties and eating quality; (iii) Moderate Mg application (29.43 kg ha−1) significantly increased grain protein content, while further increasing Mg rate (44.15 kg ha−1) resulted in decreased protein content but a significant rise in sucrose content; (iv) The pasting temperature and peak time were closely associated with soluble sugar content, exhibiting significant positive correlations with glucose and sucrose contents, respectively.
Conclusion:
When the Mg application rate was 29.43–44.15 kg ha−1, rice yield, processing quality, nutritional value, and palatability were significantly improved.
1 Introduction
As a staple food for over half of the global population, rice (Orica sativa L.) provides essential carbohydrates, proteins, vitamins, and minerals, which makes it a dietary cornerstone in many regions (Sen et al., 2020). Beyond caloric provision, rice serves as a critical vehicle for economic income and micronutrient delivery—particularly in developing countries (Muthayya et al., 2014). Magnesium (Mg), the fourth most essential macronutrient after nitrogen (N), phosphorus (P), and potassium (K), is frequently overlooked in agricultural production (Guo et al., 2016). Mg performs irreplaceable physiological functions in plants and enhances defense mechanisms against abiotic stresses (Cakmak, 2015; Senbayram et al., 2015; Ogura et al., 2020; Mengütay et al., 2013). Approximately 63.6% of cultivated land in China suffers from marginal or severe Mg deficiency (Chen et al., 2023), with this “hidden hunger” silently eroding crop production potential (Wang et al., 2020).
From molecular mechanisms to field phenotypes, Mg’s multidimensional functional network is progressively being unveiled. As the core metal ion in chlorophyll’s porphyrin ring, Mg not only determines the stable conformation of photosynthetic pigments (Humphrey, 2004) but also directly regulates photosynthetic carbon assimilation efficiency by activating key Calvin cycle enzymes like Rubisco (Hermans et al., 2005). Concurrently, Mg functions as a structural component of ribosomes, holding significant value for activating amino acid polymerization into polypeptide chains and protein biosynthesis (Wang et al., 2019). Physiological studies reveal Mg’s vital role in phloem loading and translocation of photoassimilates to sink organs, particularly during grain filling stages, which is crucial for grain yield formation (Ismail et al., 1994; Ogura et al., 2020).
In cereal crops such as rice, maize, and barley, magnesium fertilization significantly enhances grain yield (Liu et al., 2021; Khokhar et al., 2022). Notably, root crops like sugar beet exhibit even more dramatic responses, showing a fivefold increase in root dry biomass at 60 kg Mg ha−1 compared to Mg-deficient controls (D’Egidio et al., 2019). A meta-analysis of 99 research articles revealed that Mg fertilization increased crop yields across species by 6.9–2.5%, with no significant differences between rapidly available forms (e.g., MgSO4·7H2O) and slow-release forms (e.g., magnesium oxide and dolomite) in yield enhancement (Wang et al., 2020). Differential crop response patterns suggest that Mg regulation shares a common mechanism of improving photosynthetic product storage capacity through grain development indicators, while also diversifying into specialized adaptive strategies such as enhanced root morphological development. Excessive Mg, however, suppresses rice yield gains (Lamichhane et al., 2023; Yang et al., 2024), a phenomenon also observed in maize (Noor et al., 2015; Ertiftik and Zengin, 2017), indicating that optimal yield is achievable only within specific Mg application ranges.
Mg regulation exerts multidimensional effects on rice quality improvement. Studies show it significantly optimizes palatability by influencing starch metabolism (Ma et al., 2018) and mineral nutrient balance (Mg/K ratio), while promoting protein content enhancement (Li, 2002; Gransee and Führs, 2012; He et al., 2024). Mg fertilization also reduces rice chalkiness rate and degree (Zhang et al., 2004), increases grain Mg content, and modifies RVA profile parameters by elevating peak viscosity, breakdown value, and hot paste viscosity, while decreasing final viscosity, setback, and consistency (Yang et al., 2006). Yet, excessive Mg application leads to palatability decline (Wang et al., 2020), further emphasizing the existence of an optimal Mg threshold.
Soluble sugar content, a critical indicator of rice palatability, is closely linked to photosynthesis-mediated accumulation. It is reported that soluble sugars in outer rice layers are primary contributors to sweetness (Tran et al., 2005; Ma et al., 2020). Adequate Mg nutrition increases starch and sugar contents in rice stems and leaves, accelerating carbohydrate translocation from vegetative tissues to grains (Ding et al., 2006). Improved rice palatability likely arises from Mg-modulated regulation of sugar and starch accumulation and translocation. However, limited research exists on Mg’s impacts on palatability-related compounds (e.g., soluble sugars) and nutritional components in rice. Therefore, this study selects representative cultivars from Liaoning Province to address these gaps. We aimed to investigate the effects of magnesium application rates on rice yield components, milling quality, cooking/eating quality, and nutritional attributes. The findings provide theoretical and technical support for high-quality rice production.
2 Materials and methods
2.1 Experimental site
This study was conducted in 2017 at the rice cultivation experimental field of the South Campus of Shenyang Agricultural University (Shenyang, China; 123°34′E, 41°48′ N). The region is characterized by a temperate continental monsoon climate. The monthly average temperature and rainfall in the experimental area in 2017 are shown in Figure 1. The meteorological conditions during the experimental period were typical of the region’s weather characteristics. The baseline physicochemical properties of the experimental soil were as follows: alkali-hydrolyzable nitrogen (65.70 mg/kg), available phosphorus (13.59 mg/kg), available potassium (120.40 mg/kg), exchangeable Mg (0.43 g/kg), organic matter (25.63 g/kg), pH 5.53 (soil:water ratio 1:2.5), and electrical conductivity (EC) values of 274.70 μS/cm (soil:water ratio 1:5) and 294.70 μS/cm (soil:water ratio 1:2.5). Prior to sowing, composite soil samples (0–20 cm depth) were collected from three random points in each plot to verify the initial soil fertility.
Figure 1
2.2 Experimental design and planting material
Based on the recommended Mg application rate for rice proposed by Yang et al. (2024), a randomized complete block design (RCBD) was implemented with four Mg treatments: Mg sulfate heptahydrate (MgSO₄·7H₂O) was applied at 0, 90, 180, and 270 kg ha−1 (equivalent to MgO levels of 14.72, 29.43, and 44.15 kg ha−1 based on molecular weight conversion), designated as Mg0, Mg1, Mg2, and Mg3, respectively. Each treatment was replicated three times. Plots were physically separated by polyethylene dividers, with a 40 cm central access aisle reserved. Each plot measured 6 m in length × 1.5 m in width, configured with row spacing of 30 cm, plant spacing of 13.3 cm, and two seedlings transplanted per hill. Five rows were planted per plot, with border rows included around the experimental area. The japonica rice cultivar Liaojing 294, widely cultivated in Liaoning Province, was selected as the experimental material due to its high and stable yield, strong disease and lodging resistance, excellent grain quality, favorable palatability, and adaptability to most soils in the region.
2.3 Crop management
2.3.1 Measurement indicators and methods
Rice seeds were soaked for seedling raising in mid-April and transplanted to the main paddy field in late May. Mg fertilizer was applied as a single basal dose in the form of Mg sulfate heptahydrate (MgSO4·7H2O), with each treatment uniformly mixed with 1 kg of sieved fine soil before surface application. A compound fertilizer with an N–P2O5–K2O ratio of 13–17–15 was applied as basal fertilizer at 750 kg ha−1 across all plots. Field irrigation and pest/disease management followed local conventional practices.
2.3.2 Determination of appearance quality
Grain length and width were measured using ScanWizard software paired with a ScanMaker i800 Plus flatbed color scanner (MICROTEK, Shanghai). Grain thickness was measured with a vernier caliper. Each treatment group was measured 10 times, and average values were calculated.
2.3.3 Determination of milling quality
Brown rice rate, milled rice rate, and head rice rate were sequentially determined using an SY88-TH experimental huller and Yamamoto experimental rice polisher.
2.3.4 Determination of cooked rice palatability
Brown rice was milled to a precision of 90 ± 1% using a Yamamoto Vp-32 vertical rice polisher. After polishing, the appearance, hardness, viscosity, and degree of balance, and taste value of cooked rice were measured using the Satake Rice Taste Analyzer.
2.3.5 Determination of amylose content
Absorbance values were measured at a visible wavelength of 720 nm using a UV-2450 UV spectrophotometer, following the ISO 6647-2:2007 Amylose Content Determination Method. Calibration standards for high, medium, low, and waxy amylose contents were provided by the China National Rice Research Institute.
2.3.6 Determination of protein content
Protein content was measured using a FOSS Kjeltec 840 automatic Kjeldahl analyzer. Parallel dual samples were analyzed to minimize experimental error. Nitrogen content was multiplied by 5.95 to obtain protein content.
2.3.7 Determination of soluble sugar content
The determination method followed GB 5009.8-2006 for the Measurement of fructose, glucose, sucrose, maltose, and lactose in foods. Analysis was performed using a Waters Acquity H-Class UPLC system under the following chromatographic conditions: column: ACQUITY UPLC® CSHTMC18 (2.1 × 50 mm, 1.7 μm); mobile phase: acetonitrile:water = 75:25 (v/v); flow rate: 0.4 mL/min; column temperature: 40°C; injection volume: 2 μL. ELSD parameters were set as drift tube temperature 55°C, nitrogen pressure 30 psi, nebulizer temperature 30°C, and gain 500.
2.3.8 Determination of starch viscosity properties (RVA profile parameters)
Rice was milled to 80-mesh flour. Analysis was performed using a Perten RVA-TecMaster rapid visco analyzer (Sweden) with TCW (Thermal Cycle for Windows) software, following the International Association for Cereal Science and Technology standard method (ICC Standard No. 162).
2.4 Data analysis
Statistical analyses were conducted using SPSS 26.0 (SPSS Inc., Chicago, IL, United States). Differences between treatment groups were evaluated by one-way analysis of variance (ANOVA), followed by either the least significant difference (LSD) test for normally distributed datasets or the Kruskal–Wallis non-parametric test for non-normal distributions. Significance was defined at p < 0.05. Data visualization was performed using Origin 2023 (OriginLab, Northampton, MA, United States) to ensure clarity and precision in presenting research findings.
3 Results
3.1 Agronomic traits and yield components
Mg application significantly promoted rice growth, development, and yield formation. Specifically, Mg treatments increased average plant height by 2.78%. Concurrently, they enhanced grain filling rate by reducing the proportion of sterile grains (15.16, 17.86, and 16.39% improvements for Mg1, Mg2, and Mg3, respectively). Further analysis revealed that Mg application significantly increased panicle number per hectare, with yield increases of 7.83, 9.60, and 10.21% achieved through optimized tiller productivity (p < 0.05) (Figure 2).
Figure 2
3.2 Variations in appearance and milling quality
This study demonstrated that appropriate Mg application significantly improved rice grain morphology and milling quality, though effects varied among treatments. Specifically, the Mg2 treatment substantially increased grain thickness by 3.23%, while no significant changes were observed in grain length or width, indicating that moderate Mg enhanced grain plumpness. Mg application improved both milled rice rate and head rice rate, with the Mg3 treatment significantly increasing milled rice rate by 2.68% and head rice rate by 13.28% (p < 0.05) (Figure 3). These findings suggest that Mg application enhances rice processing performance.
Figure 3
3.3 Eating quality and starch gelatinization characteristics
Analysis revealed that Mg application moderately improved cooked rice appearance, viscosity, degree of balance, and taste value while reducing hardness. The taste value exhibited a trend of initial increase followed by decrease, with the Mg2 treatment maximizing rice degree of balance and taste value, which were significantly enhanced by 9.88 and 5.21%, respectively. The Mg3 treatment significantly increased rice viscosity by 11.59% (p < 0.05) (Figure 4A). These results indicate that appropriate Mg application improves cooked rice palatability.
Figure 4
Figure 4B radar charts illustrate differential RVA characteristics under magnesium treatments. Compared to the Mg0 control, magnesium application improved starch viscosity properties, with the Mg3 treatment significantly increasing minimum viscosity (↑5.36%), final viscosity (↑3.17%), and setback value (↑3.79%) (p < 0.05).
3.4 Variations in protein, amylose, glucose, and sucrose contents
Results showed that Mg application initially increased then decreased protein content. Specifically, the Mg2 and Mg3 treatments significantly enhanced grain protein content by 2.16 and 1.91%, respectively, but had no significant effect on amylose content. Mg application did not significantly alter glucose content, though sucrose content gradually increased with Mg dosage. The Mg3 treatment significantly increased sucrose content by 13.54% (p < 0.05).
3.5 Correlation analysis
Protein content was significantly positively correlated with cooked rice appearance, viscosity, degree of balance, and taste value, while also significantly influencing the breakdown value in the RVA profile. Amylose content exhibited a significant negative correlation with rice palatability parameters (excluding hardness) and RVA eigenvalues, and notably affected setback value changes. Sucrose effectively improved cooked rice palatability and gelatinization properties, showing a strong positive correlation with prolonged peak time (r = 0.79), whereas increased glucose content was significantly positively associated with elevated pasting temperature (r = 0.96). These results indicate that synergistic interactions among protein, starch components, and soluble sugars exert comprehensive regulatory effects on rice cooking quality and gelatinization characteristics (p < 0.05).
4 Discussion
4.1 Effects of magnesium on rice yield formation
This study revealed that Mg application significantly enhanced rice plant height, effective tillering rate, and grain filling rate (Figures 2A,B,F), with yield gains primarily attributed to Mg’s impact on population density. As the core of the chlorophyll molecule, Mg not only maintains the structural integrity of the photosystem reaction center but also optimizes electron transport chain efficiency to boost ATP synthesis capacity (Hawkesford et al., 2023), facilitating prioritized translocation of photosynthetic products to grain sinks during the grain-filling stage (Zhang et al., 2017). Additionally, Mg activates AGPase, a key enzyme in starch synthesis, significantly accelerating starch accumulation in endosperm cells and forming a positive feedback loop between efficient photosynthetic product conversion and grain filling (Farhat et al., 2016). Mg deficiency adversely affects grain size and starch content (Ceylan et al., 2016), causing carbohydrate storage in leaves rather than roots, thereby reducing root growth attributes and other plant characteristics, ultimately leading to yield losses (Gardner, 2003). This explains the improved grain filling degree and yield observed in our study. As Mg2+ application rates increase, the seed setting rate shows a declining trend. A possible explanation is the antagonistic interaction between Mg2+ and K+ cations. Excess Mg2+ inhibits root uptake of K+, leading to K+ deficiency during the grain filling stage. K+ acts as a critical activator for starch synthesis (via ADP-glucose pyrophosphorylase), and its deficiency directly reduces filling efficiency, thereby increasing the sterile grain rate (Dölger et al., 2024). However, the mechanistic interactions between Mg and K require further investigation.
Notably, Mg application exhibits a threshold effect. A study in Fujian rice-growing regions found that an optimal MgO rate of 31.6 kg ha−1 increased rice yield by 7.2%, mainly due to enhanced effective panicle number and filled grains per panicle (Yang et al., 2024). Our results showed that MgO application between 29.43–44.15 kg ha−1 benefited both population establishment and effective grain formation, consistent with Yang et al. (2024). Another study demonstrated that soil Mg application at 12 kg ha−1 significantly increased rice yield, with additive effects when combined with silicon (He et al., 2024), indicating Mg–Si interactive effects. Numerous studies have also explored K–Mg interactions in plants. For instance, high K concentrations in growth media inhibit root Mg uptake, causing antagonistic effects between the two elements (Ceylan et al., 2016). Conversely, low K concentrations promote Mg absorption, demonstrating synergistic effects (Xie et al., 2021). Furthermore, the increase of phosphorus content reduced Mg absorption (Houdegbe et al., 2022). Future research should investigate Mg interactions with other elements and their yield-boosting potentials.
4.2 Effects of magnesium on rice quality
Mg is an integral component of ribosomes, stabilizing ribosomal configurations essential for protein synthesis. Mg deficiency causes ribosomal dissociation into smaller ribosomal subunits (Wang et al., 2017). Appropriate Mg application promotes activation of numerous enzymes in nitrogen metabolism, enhancing nitrogenous compound synthesis (Grzebisz, 2013; Peng et al., 2020). Low Mg addition stimulates rice nitrogen uptake, increasing crude fat and protein contents, whereas excessive Mg inhibits nitrogen absorption (He et al., 2024), explaining the observed increase in grain protein content. Core components in rice endosperm closely related to palatability quality are amylose and protein. When amylose content is low, cooked rice palatability significantly improves (Li and Gilbert, 2018). Our study found Mg had no significant effect on amylose content, differing from Zhang et al. (2022) who reported reduced amylose levels. This discrepancy may arise from our basal Mg application versus foliar application in previous studies. Additionally, we observed Mg significantly increased minimum viscosity (+14.1%) and setback value (+19.8%) in the RVA profile, potentially due to moderate crosslinking between Mg2+ and starch acidic groups (especially carboxyl groups), forming bridging structures acting as “loading carriers” to promote effective mineral particle aggregation (Tang et al., 2024).
4.3 Mechanism of magnesium-regulated sugar metabolism influencing rice quality
Mg plays a critical role in phloem sucrose loading, influencing carbohydrate partitioning between source and sink tissues. Proton motive force generated by H+-pumping ATPases energizes H+-sucrose symporters to load sucrose into sieve tube cells (Hermans et al., 2004). Adequate Mg levels are also vital for sugar transport during seed germination and seedling development (Ceylan et al., 2016). Germinating seeds exhibit substantial sucrose transport from the scutellum to actively growing sink tissues like roots and shoots to sustain germination processes (Aoki et al., 2006; Scofield et al., 2007). Analogously, sufficient Mg participation is likely required for sugar translocation to sinks during seed maturation. Sucrose and glucose are the primary soluble sugars in rice grains (Zhao et al., 2020). As a non-reducing sugar, sucrose is the most abundant grain sugar, comprising approximately 90% of total sugars (Frank et al., 2012). Our study observed low grain glucose content under Mg-deficient conditions, with Mg application inducing an initial increase followed by a decrease, while sucrose content progressively increased with Mg dosage (Figure 4D). This may stem from Mg deficiency reducing photosynthetic efficiency in leaves, causing starch accumulation and decreased soluble sugar export. Conversely, Mg application enhances photosynthetic efficiency and starch mobilization, increasing leaf soluble sugar content, consistent with findings by Ma et al. (2018) and Zheng et al. (2019) (see Figure 5C,D).
Figure 5
Sucrose content was positively correlated with cooked rice palatability and RVA profile eigenvalues (Figure 6). This association arises from the dynamic equilibrium of multiple solutes (e.g., sugars and alcohols) during rice consumption. These solutes maintain protein conformational stability and membrane structural integrity through osmotic effects, regulate cellular turgor status, and directly influence the final viscoelastic properties of cooked rice (Zhao et al., 2020). However, glucose significantly elevated pasting temperature, potentially due to high temperatures promoting sucrose, oligosaccharide, and polysaccharide degradation, reducing sucrose levels while increasing glucose and fructose contents. Monosaccharides exhibit protective effects on starch crystalline structures during gelatinization (Renzetti and van der Sman, 2022). However, this study did not directly validate the specific molecular mechanisms underlying magnesium’s regulation of sugar transport (e.g., H+-ATPase activity and sucrose transporter function). Additionally, the experimental conclusions were drawn under specific environmental conditions, and the generalizability of magnesium fertilizer effects across diverse soil types, climatic zones, and rice cultivars requires further investigation.
Figure 6
5 Conclusion
This study demonstrates that Mg application elevated rice plant height and, by enhancing seed setting rate and tillering productivity leading to panicle formation, achieved significant increases in grain yield. When the application rate of MgO (magnesium oxide) is within the range of 29.43–44.15 kg ha−2, notable improvements are observed in grain thickness, rice eating quality, processing quality, starch pasting properties, as well as protein and sucrose contents. Collectively, basal application of MgSO₄·7H₂O at 180–270 kg ha−1 synergistically enhances both grain yield and quality parameters in rice. This study offers important theoretical support for precise Mg management and the development of efficient Mg fertilizer application techniques in rice production.
Statements
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
YW: Writing – original draft, Writing – review & editing. ZM: Supervision, Writing – review & editing. HC: Resources, Supervision, Writing – review & editing. WL: Supervision, Resources, Funding acquisition, Writing – review & editing, Project administration. DW: Writing – review & editing, Supervision, Resources.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was funded by the National Natural Science Foundation of China (32201892), Education Fund Item of Liaoning Province (LJKMZ20221016).
Acknowledgments
The test work of this experiment was completed in the Test Center of Agricultural College of Shenyang Agricultural University, for which the authors extend their thanks.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1
AokiN.ScofieldG. N.WangX. D.OfflerC. E.PatrickJ. W.FurbankR. T. (2006). Pathway of sugar transport in germinating wheat seeds. Plant Physiol.141, 1255–1263. doi: 10.1104/pp.106.082719
2
Cakmakİ. (2015). Magnesium in crop production, food quality, and human health. Crop Pasture Sci.66, i–ii. doi: 10.1071/CPv66n12_FO
3
CeylanY.KutmanÜ. B.MengütayM.Çakmakİ. (2016). Magnesium applications to growth medium and foliage affect the starch distribution, increase the grain size, and improve the seed germination in wheat. Plant Soil406, 145–156. doi: 10.1007/s11104-016-2871-8
4
ChenX. H.WangZ.MuneerM. A.MaC.HeD. D.WhiteP. J.et al. (2023). Short planks in the crop nutrient barrel theory of China are changing: evidence from 15 crops in 13 provinces. Food Energy Secur.12:e389. doi: 10.1002/fes3.389
5
D’EgidioS.GalieniA.StagnariF.PagnaniG.PisanteM. (2019). Quality and physiological traits of red beet under different magnesium nutrition and light intensity levels. Agronomy9:379. doi: 10.3390/agronomy9070379
6
DingY.LuoW.XuG. (2006). Characterisation of magnesium nutrition and interaction of magnesium and potassium in rice. Ann. Appl. Biol.149, 111–123. doi: 10.1111/j.1744-7348.2006.00080.x
7
DölgerJ. L.HenningsenJ. N.MühlingK. H. (2024). Antagonistic K/Mg ratios: is foliar application of MgSO4 a superior alternative to root resupply?Plant Soil505, 747–761. doi: 10.1007/s11104-024-06708-5
8
ErtiftikH.ZenginM. (2017). Response of maize for grain to potassium and magnesium fertilizers in soils with high lime contents. J. Plant Nutr.40, 93–103. doi: 10.1080/01904167.2016.1201493
9
FarhatN.ElkhouniA.ZorrigW.SmaouiA.AbdellyC.RabhiM. (2016). Effects of magnesium deficiency on photosynthesis and carbohydrate partitioning. Acta Physiol. Plant.38, 1427–1436. doi: 10.1007/s11738-016-2165-z
10
FrankT.ReichardtB.ShuQ.EngelK.-H. (2012). Metabolite profiling of colored rice (Oryza sativa L.) grains. J. Cereal Sci.55, 112–119. doi: 10.1016/j.jcs.2011.09.009
11
GardnerR. C. (2003). Genes for magnesium transport. Curr. Opin. Plant Biol.6, 263–267. doi: 10.1016/s1369-5266(03)00032-3
12
GranseeA.FührsH. (2012). Magnesium mobility in soils as a challenge for soil and plant analysis, magnesium fertilization, and root uptake under adverse growth conditions. Plant Soil368, 5–21. doi: 10.1007/s11104-012-1567-y
13
GrzebiszW. (2013). Crop response to magnesium fertilization as affected by nitrogen supply. Plant Soil368, 23–39. doi: 10.1007/s11104-012-1574-z
14
GuoW. L.NazimH.LiangZ. S.YangD. F. (2016). Magnesium deficiency in plants: an urgent problem. Crop J.4, 83–91. doi: 10.1016/j.cj.2015.11.003
15
HawkesfordM. J.CakmakI.CoskunetD.De KokL. J.LambersH.SchjoerringJ. K.et al. (2023). “Functions of macronutrients” in Marschner’s mineral nutrition of plants (London: Academic Press), 201–281.
16
HeZ.WangZ.HaoJ.WuY.LiuH. (2024). The use of magnesium fertilizer can improve the nutrient uptake, yield, and quality of rice in Liaoning Province. Agronomy14:639. doi: 10.3390/agronomy14030639
17
HermansC.BourgisF.FaucherM.StrasserR. J.DelrotS.VerbruggenN. (2005). Magnesium deficiency in sugar beets alters sugar partitioning and phloem loading in young mature leaves. Planta220, 541–549. doi: 10.1007/s00425-004-1376-5
18
HermansC.JohnsonG. N.StrasserR. J.VerbruggenN. (2004). Physiological characterization of magnesium deficiency in sugar beet: acclimation to low magnesium differentially affects photosystems I and II. Planta220, 344–355. doi: 10.1007/s00425-004-1340-4
19
HoudegbeA. C.Achigan-DakoE. G.SogbohossouE. O. D.SchranzM. E.OdindoA. O.SibiyaJ. (2022). Leaf elemental composition analysis in spider plant (Gynandropsis gynandra L. (Briq.)) differentiates three nutritional groups. Front. Plant Sci.13:841226. doi: 10.3389/fpls.2022.841226
20
HumphreyA. M. (2004). Chlorophyll as a color and functional ingredient. J. Food Sci.69, C422–C425. doi: 10.1111/j.1365-2621.2004.tb10710.x
21
IsmailC.ChristineH.HorstM. (1994). Changes in phloem export of sucrose in leaves in response to phosphorus, potassium, and magnesium deficiency in bean plants. J. Exp. Bot.45, 1251–1257. doi: 10.1093/jxb/45.9.1251
22
KhokharA.SharmaV.SinghM.YousufA.SandhuP.KumarV.et al. (2022). Effect of potassium and magnesium application on growth, yield, and nutrient uptake of rainfed maize in the sub-montaneous region of Punjab, India. J. Plant Nutr.45, 2202–2212. doi: 10.1080/01904167.2022.2063732
23
LamichhaneS.TarpleyL.DouF. G. (2023). Impact of excess magnesium salt supply on rice yield, physiological response, and grain mineral content. Sustainability15:15741. doi: 10.3390/su152215741
24
LiX. M. (2002). Effects of mineral magnesium on rice yield and quality. Chin. J. Plant Nutr. Fertil.8, 125–126. doi: 10.3321/j.issn:1008-505X.2002.01.025
25
LiH.GilbertR. G. (2018). Starch molecular structure: the basis for an improved understanding of cooked rice texture. Carbohyd. Polym.195, 9–17. doi: 10.1016/j.carbpol.2018.04.065
26
LiuZ.HuangQ.LiuX.LiP.NaseerM. R.CheY.et al. (2021). Magnesium fertilization affected rice yields in magnesium-sufficient soil in Heilongjiang Province, Northeast China. Front. Plant Sci.12:645806. doi: 10.3389/fpls.2021.645806
27
MaX. L.LiuX. F.YangM.YanQ. Y.YuanX. C.XiangP. W. (2018). Effects of magnesium fertilizer on sugar, starch, protein, and fruit quality in grape leaves. Chin. J. Soil Sci. Fertil.4, 114–120. doi: 10.11838/sfsc.20180418
28
MaZ. H.WangY. B.ChengH. T.ZhangG. C.LyuW. Y. (2020). Biochemical composition distribution in different grain layers is associated with the edible quality of rice cultivars. Food Chem.311, 125896–125810. doi: 10.1016/j.foodchem.2019.125896
29
MengütayM.CeylanY.KutmanÜ. B.Çakmakİ. (2013). Adequate magnesium nutrition mitigates adverse effects of heat stress on maize and wheat. Plant Soil368, 57–72. doi: 10.1007/s11104-013-1761-6
30
MuthayyaS.SugimotoJ. D.MontgomeryS.MaberlyG. F. (2014). An overview of global rice production, supply, trade, and consumption. Ann. N. Y. Acad. Sci.1324, 7–14. doi: 10.1111/nyas.12540
31
NoorS.AkhterS.IslamM. M.ShilN. C.KawocharM. A. (2015). Effect of magnesium on crop yields within maize-green manure-T. aman rice cropping pattern on acid soil. Arch. Agron. Soil Sci.61, 1381–1392. doi: 10.1080/03650340.2015.1011139
32
OguraT.KobayashiN. I.HermansC.IchihashiY.ShibataA.ShirasuK.et al. (2020). Short-term magnesium deficiency triggers nutrient retranslocation in Arabidopsis thaliana. Front. Plant Sci.11:563. doi: 10.3389/fpls.2020.00563
33
PengW. T.QiW. L.NieM. M.XiaoY. B.LiaoH.ChenZ. C. (2020). Magnesium supports nitrogen uptake through regulating NRT2.1/2.2 in soybean. Plant Soil457, 97–111. doi: 10.1007/s11104-019-04157-z
34
RenzettiS.van der SmanR. G. M. (2022). Food texture design in sugar reduced cakes: predicting batters rheology and physical properties of cakes from physicochemical principles. Food Hydrocoll.131:107795. doi: 10.1016/j.foodhyd.2022.107795
35
ScofieldG. N.AokiN.HiroseT.TakanoM.JenkinsC. L.FurbankR. T. (2007). The role of the sucrose transporter, OsSUT1, in germination and early seedling growth and development of rice plants. J. Exp. Bot.58, 483–495. doi: 10.1093/jxb/erl217
36
SenS.ChakrabortyR.KalitaP. (2020). Rice-not just a staple food: a comprehensive review on its phytochemicals and therapeutic potential. Trends Food Sci. Technol.97, 265–285. doi: 10.1016/j.tifs.2020.01.022
37
SenbayramM.GranseeA.WahleV.ThielH. (2015). Role of magnesium fertilizers in agriculture: plant–soil continuum. Crop Pasture Sci.66, 1219–1229. doi: 10.1071/CP15104
38
TangM.NiuX.WenS. (2024). Influences of magnesium ions in water on gelatinization characteristics of starch and its flocculation behaviors on particles. Physicochem. Probl. Miner. Process.60:178149. doi: 10.37190/ppmp/178149
39
TranT. U.SuzukiK.OkadomeH.IkezakiH.HommaS.OhtsuboK. (2005). Detection of changes in taste of japonica and indica brown and milled rice (Oryza sativa L.) during storage using physicochemical analyses and a taste sensing system. J. Agric. Food Chem.53, 1108–1118. doi: 10.1021/jf049064+
40
WangY. B.ChengH. T.MaZ. H.LüW. Y. (2020). Effects of magnesium application on quality of different japonica rice varieties with varying taste. J. Hebei Agric. Univ.43, 23–28. doi: 10.13320/j.cnki.jauh.2020.0047
41
WangY.HuaX.XuJ.ChenZ. C.FanT.ZengZ.et al. (2019). Comparative genomics revealed the gene evolution and functional divergence of magnesium transporter families in Saccharum. BMC Genomics20:83. doi: 10.1186/s12864-019-5437-3
42
WangH.YigX.OrrM. W.DambachM.CurtisR.StorzG. (2017). Increasing intracellular magnesium levels with the 31-amino acid MgtS protein. Proc. Natl. Acad. Sci. U.S.A.114, 5689–5694. doi: 10.1073/pnas.1703415114
43
XieK.Çakmakİ.WangS.ZhangF.GuoS. (2021). Synergistic and antagonistic interactions between potassium and magnesium in higher plants. Crop J.9, 249–256. doi: 10.1016/j.cj.2020.10.005
44
YangR. X.HuangW. Q.QiuC. Y.ZhangS. C.LiaoL. L.YanX. J.et al. (2024). Magnesium fertilizer efficiency and recommended application rate for rice in Fujian Province. Chin. J. Plant Nutr. Fertil.30, 1515–1528. doi: 10.11674/zwyf.2024084
45
YangW. X.WangQ. S.WangS. H.LiG. H.DingY. F. (2006). Effects of magnesium fertilizer on magnesium uptake, distribution, and rice palatability quality. Chin. J. Northwest Bot.26, 2473–2478. Available online at: http://xbzwxb.cnjournals.com/xbzwxb/article/abstract/2006012491?st=article_issue
46
ZhangH. P.ChenZ. Q.WangR.LuH.CuiP. Y.YangY. J.et al. (2022). Effects of nitrogen fertilizer combined with nano-magnesium on rice yield, quality, and nitrogen use efficiency. Crop J.4, 255–261. doi: 10.16035/j.issn.1001-7283.2022.04.036
47
ZhangS.ShaoY.-H.ShiZ. L.TianZ. W.JiangD.DaiT. B. (2017). Effects of magnesium application on dry matter accumulation, translocation, and grain filling in wheat under post-anthesis heat stress. Chin. J. Wheat Crops37, 963–969. doi: 10.7606/j.issn.1009-1041.2017.07.14
48
ZhangJ. Y.XiaoY. H.WanX. Y.LiuS. J.WangC. M.ChenL. M.et al. (2004). Stability analysis of appearance quality traits in rice varieties. Crop J.30, 548–554. Available at: https://zwxb.chinacrops.org/CN/Y2004/V30/I06/548
49
ZhaoQ.XueY.ShenQ. (2020). Changes in the major aroma-active compounds and taste components of Jasmine rice during storage. Food Res. Int.133:109160. doi: 10.1016/j.foodres.2020.109160
50
ZhengW.HassanM. U.NadeemF.WuL.ZhangF.LiX. (2019). Magnesium fertilization improves crop yield in most production systems: a meta-analysis. Front. Plant Sci.10:1727. doi: 10.3389/fpls.2019.01727
Summary
Keywords
magnesium fertilization, rice yield components, milling quality, nutritional quality, eating quality, starch gelatinization, japonica rice
Citation
Wang Y, Ma Z, Cheng H, Lyu W and Wang D (2025) Effects of moderate magnesium application on rice milling quality, nutritional value and eating quality. Front. Sustain. Food Syst. 9:1640845. doi: 10.3389/fsufs.2025.1640845
Received
05 June 2025
Accepted
28 July 2025
Published
18 August 2025
Volume
9 - 2025
Edited by
Matteo Balderacchi, Independent Researcher, Piacenza, Italy
Reviewed by
Gayatri Verma, Punjab Agricultural University, India
Marcin Różewicz, Institute of Soil Science and Plant Cultivation, Poland
Folasade Afolabi, Oyo State Technical University, Nigeria
Updates
Copyright
© 2025 Wang, Ma, Cheng, Lyu and Wang.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wenyan Lyu, lwyzxr@syau.edu.cn; Dong Wang, wangd@nwafu.edu.cn
†These authors have contributed equally to this work
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.