- 1Department of Zoology, Government Sadiq College Women University, Bahawalpur, Punjab, Pakistan
- 2Department of Entomology, Faculty of Agriculture and Environment, The Islamia University of Bahawalpur, Bahawalpur, Punjab, Pakistan
- 3School of Economics and Management, Beijing University of Technology, Beijing, China
- 4Department of Theriogenology, Faculty of Veterinary and Animal Sciences, Cholistan University of Veterinary and Animal Sciences, Bahawalpur, Punjab, Pakistan
- 5Key Laboratory of Plant-Soil Interactions of MOE, College of Resources and Environmental Sciences, National Academy of Agriculture Green Development, China Agricultural University, Beijing, China
- 6Department of Cell Biology, School of Life Sciences, Central South University, Changsha, China
- 7College of Life Science, Anhui Agricultural University, Hefei, Anhui, China
- 8Fisheries Faculty, Munzur University, Tunceli, Türkiye
- 9Department of Animal Nutrition and Forage Production, Faculty of AgriSciences, Mendel University in Brno, Brno, Czechia
- 10Institute of Scientific Instruments of the Czech Academy of Sciences, Brno, Czechia
- 11College of Agricultural Science and Engineering, Liaocheng University, Shangdong, China
Plastic pollution ranks among the most severe environmental disasters caused by humans, generating millions of tonnes of waste annually. The extensive and unregulated use of plastics has led to ecotoxicity and environmental imbalance. Microplastics (MPs) are prevalent in aquatic environments, and these MPs further degrade into even smaller particles known as nano-plastics (NPs). Both MPs and NPs impact the environment by readily absorbing organic pollutants and pathogens from their surroundings, owing to their bigger surface area to volume ratio. This review focuses on the source of origin, bioaccumulation, and potential impact of MPs and NPs on aquatic organisms and human health. Additionally, the review explores various methods employed for identification and quantification of these particles in aquatic ecosystems. Sufficient information is available on their characteristics, distributions, and effects on marine ecosystems compared with freshwater ecosystems. For plastic particles <10 μm, more toxicological effects were observed compared with larger size particles, in aquatic life. Understanding the mechanism of action and ecotoxicological effects of micro/nano-plastics on the health of aquatic life across various trophic levels, as well as human health, is of utmost importance. We address knowledge gaps and provide insights into future research approaches for a better understanding of the interactive mechanisms between binary pollutants.
1 Introduction
Plastic debris has emerged as a global environmental issue, and the improper handling of plastic waste has led to a rapid escalation of its presence in ecosystems (Oliveira et al., 2019; Yu et al., 2019) especially aquatic ecosystems (Han et al., 2024). The worldwide annual production of plastic materials now exceeds 320 million tonnes, with 40% dedicated to single-use packaging (Food and Agriculture Organization, 2013). A staggering 70% of plastic material, amounting to 5,800 million tonnes, has transformed into debris, and approximately 79% (4,900 million tonnes) has amassed in ecosystems or landfills as of 2015 (Geyer et al., 2017). The widespread use of plastics in various applications persists due to their cost-effective manufacturing, utility, and durability (Barría et al., 2020). Plastics have been the preferred material for many years owing to their versatility, ubiquity, lightness, durability, and adaptability (Nielsen et al., 2020). The use of plastics is also increasing every day in agriculture benefitting agricultural production. However, the misuse of plastics after agricultural operations can lead to plastic waste and consequent environmental contamination by plastic debris (Mongil-Manso et al., 2023; Kudzin et al., 2024). Unfortunately, due to careless and excessive use, improper management, and inadvertent disposal, a significant volume of plastics has amassed in aquatic systems (Peng et al., 2020). Thus, they can accumulate at higher trophic levels, infiltrate the food chain, and pose a potential risk to ecosystems, native and non-native species, and human health (Neves et al., 2024).
Plastics of various types are globally produced, with polyethylene, polyvinyl chloride, polystyrene, polypropylene, polyethylene terephthalate, and polyurethane identified as the most prevalent plastic varieties (Al-Thawadi, 2020). Through processes like mechanical abrasion and biological deterioration, plastics can undergo fragmentation, resulting in the formation of secondary microplastics (MPs) and nano-plastics (NPs), (Alimi et al., 2018; Oliveira et al., 2019). Micro/nano-plastics (MNPs), owing to their capacity to absorb and accumulate co-contaminants, exert a physical and chemical impact on the environment. The attachment of metallic/organic toxins to MNPs and their subsequent transport into animal bodies depend on sorption mechanisms primarily influenced by the physico-chemical characteristics of MNPs and the type of pollutants (Thiagarajan et al., 2021). Nanoplastics and MPs are categorized based on their size, with NPs measuring less than 1000 nm and MPs being less than 5 mm (Frias and Nash, 2019). Although there is currently no formal definition for NPs, they are generally considered to share the same origin and composition as MPs but with a size of less than 1,000 nm (Gigault et al., 2018; Ferreira et al., 2019; Barría et al., 2020). Generally, MNPs are classified into primary and secondary MNPs. Examples of primary MNPs include synthetic fibers, cosmetics, pharmaceuticals, and raw materials (Li et al., 2018; Wang et al., 2018; Wang et al., 2020). Primary MNPs, being smaller in size, have a larger surface area, facilitating the adsorption of hydrophobic constituents from marine systems, such as polycyclic aromatic hydrocarbons (PAHs), perfluorooctanoic acid (PFOA), dichlorodiphenyltrichloroethane (DDT), polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs), and metals (Li et al., 2018; Ferreira et al., 2019).
Micro and nano plastics have caused significant pollution in water bodies including drinking water (Li et al., 2023; Brancaleone et al., 2023). Moreover, aquatic organisms are regularly being exposed to pharmaceuticals nanomaterials (PC/NM prevalent in industrial and urban areas (Naz et al., 2021; Fernandes et al., 2023). Wastewater treatment plants appear to be a major source of contamination in the aquatic ecosystem (Vaid et al., 2021; Gagné et al., 2023). Consequently, investigations into the interactions between MNPs and PC/NM, along with their ecotoxicological effects on aquatic biota, have been conducted. Fish easily ingest microplastic particles, both unintentionally due to their small size and deliberately, due to resemblance to food sources (Zubair et al., 2020; Naz et al., 2022). A study by Wang et al. (Wang et al., 2020) revealed the presence of microplastics in over 150 fish species in aquatic environments. In the Gorgan Bay of the Caspian Sea, various types of microplastics, including polypropylene, polyester, nylon, and polystyrene, were detected in sediment, fishes, and benthic organisms, ranging from 80 to 105 MP/kg (Bagheri et al., 2020). Given that fish is a significant protein source for humans, the existence of microplastics in fish and their ecotoxicological effects could have adverse consequences for both aquatic food sources and human health (Barboza et al., 2018). There is an urgent need to find or develop various methods like the use of microorganisms (Herrera et al., 2023) or the use of non-toxic, novel agglomerate (Peller et al., 2024) for degradation of micro and nano plastics for sustainable plastic waste management. Furthermore, social responsibility and a shift in consumer behaviours and habits in adopting low-risk products should also be encouraged (Rashed et al., 2023). Despite an abundance of research on the ingestion and consequences of MNPs, there has been a scarcity of review publications on this topic until recently. Therefore, this review specifically focuses on a multidisciplinary approach, drawing upon insights from environmental science, ecology, toxicology, and public health. It covers various types of micro and nano-plastics, including microbeads, microfibers, and nanoplastics, and their interactions with different aquatic organisms ranging from plankton to fish. Furthermore, the review considers diverse aquatic environments such as oceans, rivers, lakes, and estuaries, acknowledging the variability in plastic pollution levels and ecological dynamics across these habitats. Additionally, the review highlights uncertainties and information gaps in understanding the fate, distribution, and harmful mechanisms of MNPs and PC/NM to aquatic organisms.
2 Toxic effects of MNPs on aquatic organisms
Microplastics (MPs) may have detrimental effects on aquatic ecosystems, impacting various organisms such as phytoplankton, invertebrates, mollusks, and fish, as they enter freshwater networks in substantial quantities (0.12–387 items/m3) (Brandts et al., 2018; Triebskorn et al., 2019). Numerous studies have been conducted to investigate the toxic effects of MNPs on water-dwelling organisms. A study conducted by Chae et al. (Chae et al., 2018) observed the trophic transfer and effects of 51 nm polystyrene nano-plastics (PS-NPs) on four freshwater species, including the alga Chlamydomonas reinhardtii. Despite exposure to concentrations as high as 100 mg/L resulting in little to no mortality, confocal laser microscopy revealed the attachment of NPs to the zoospores’ surface and outer layer penetration during cell division. Nano-plastics also led to reduced locomotor activity and induced histological abnormalities in the livers of fish directly exposed to them. Furthermore, the study observed that NPs could pass through embryonic walls and persist in hatched larvae yolk. In another investigation (Sökmen et al., 2020), the effects of short-term (24 h) exposure to negatively charged fluorescent PS-NPs (50 nm), aggregated with gold nanoparticles (Au ions), were explored in Danio rerio. Comparing the impacts of individual exposure to PS-NPs and Au ions, the study found increased mortality and deformation rates in the exposed organisms. Additionally, there was a stimulated immunological response, indicated by elevated expression of IL-6 and IL-1 β. Exposure to PS NPs or Au ions individually resulted in higher levels of reactive oxygen species (ROS), formation of intracellular vacuoles, and mitochondrial damage (Lee et al., 2019).
Exposure to 45 nm polymethyl methacrylate nanoparticles (PMMA-NPs) at concentrations of ≤20 mg/L was found to affect the immune system of fish, with an observed increase in mRNA transcripts associated with lipid metabolism (Brandts et al., 2018). In Sebastes schlegelii samples exposed to 0.5 and 15 μm PS-NPs (190 μg/L) exhibited clustering, reduced swimming speed, increased oxygen consumption, and ammonia excretion, as well as lower protein and lipid contents (Yin et al., 2019; Jiang et al., 2023a). Despite ingesting more than 90% of microalgae containing polystyrene nanoparticles (PS-NPs), brine shrimp (Artemia franciscana) did not show any significant effects (Sendra et al., 2020). Zebrafish exposed to secondary nanoparticles showed a 54% increase in cell death through skin diffusion compared to microplastics (Enfrin et al., 2020; Jiang et al., 2023b). Sökmen et al. (Sökmen et al., 2020) explored the impacts of NPs on zebrafish (D. rerio), revealing that 20 nm diameter PS-NPs reached and accumulated in the zebrafish brain, causing oxidative DNA damage. Other organs were also reported to be affected by NPs, establishing zebrafish as a valuable model for studying NP toxicity (Bhagat et al., 2020a; Sarasamma et al., 2020). The hydrophobicity of tetracycline-incubated NPs contributed to variations in toxic effects observed in the marine microalgae Skeletonema costatum (Feng et al., 2020a). Nano-plastics adsorption on microalgae has been documented in several studies, with some cases showing a reduction in algal growth while others did not (Bergami et al., 2017; Heinlaan et al., 2020).
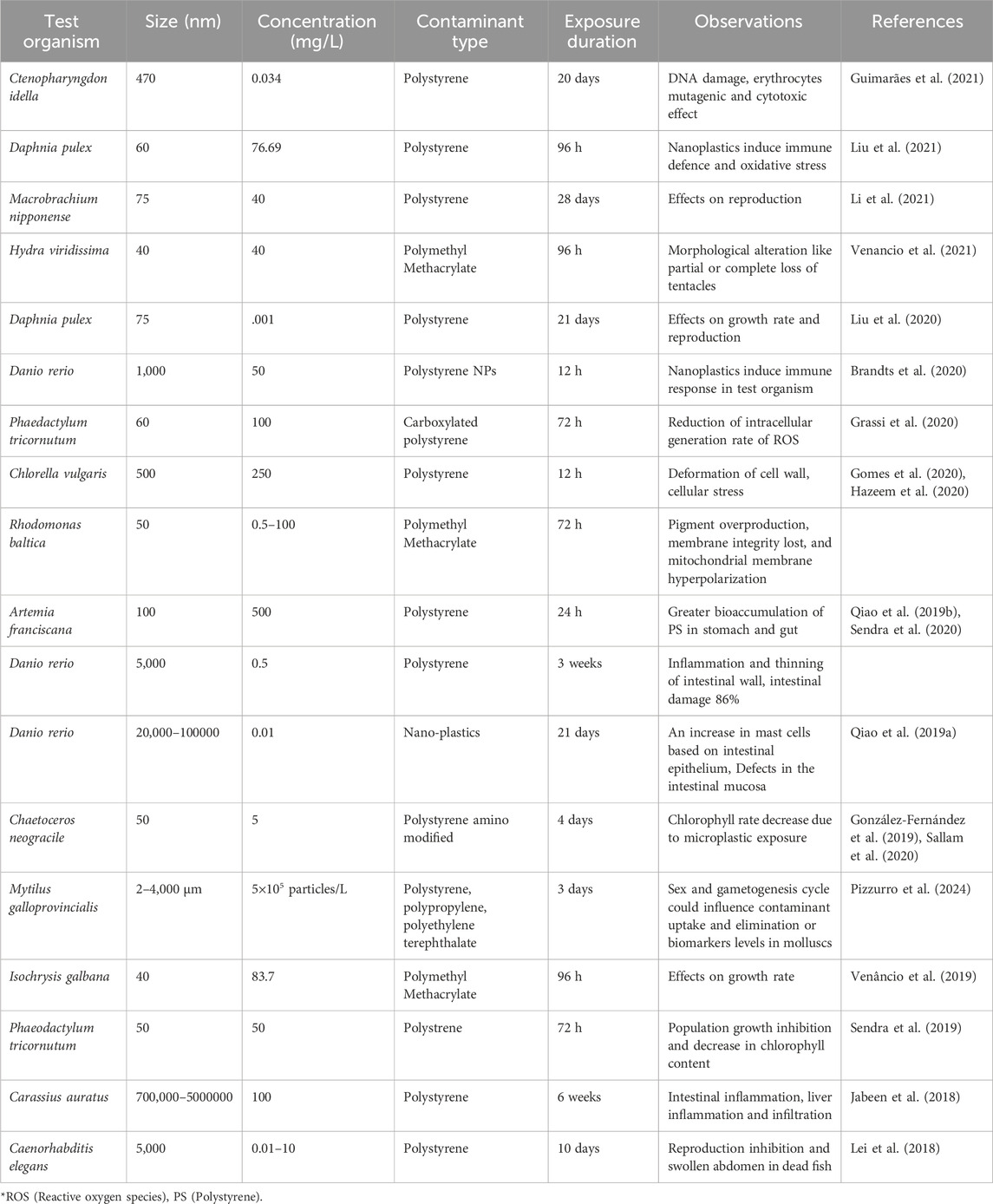
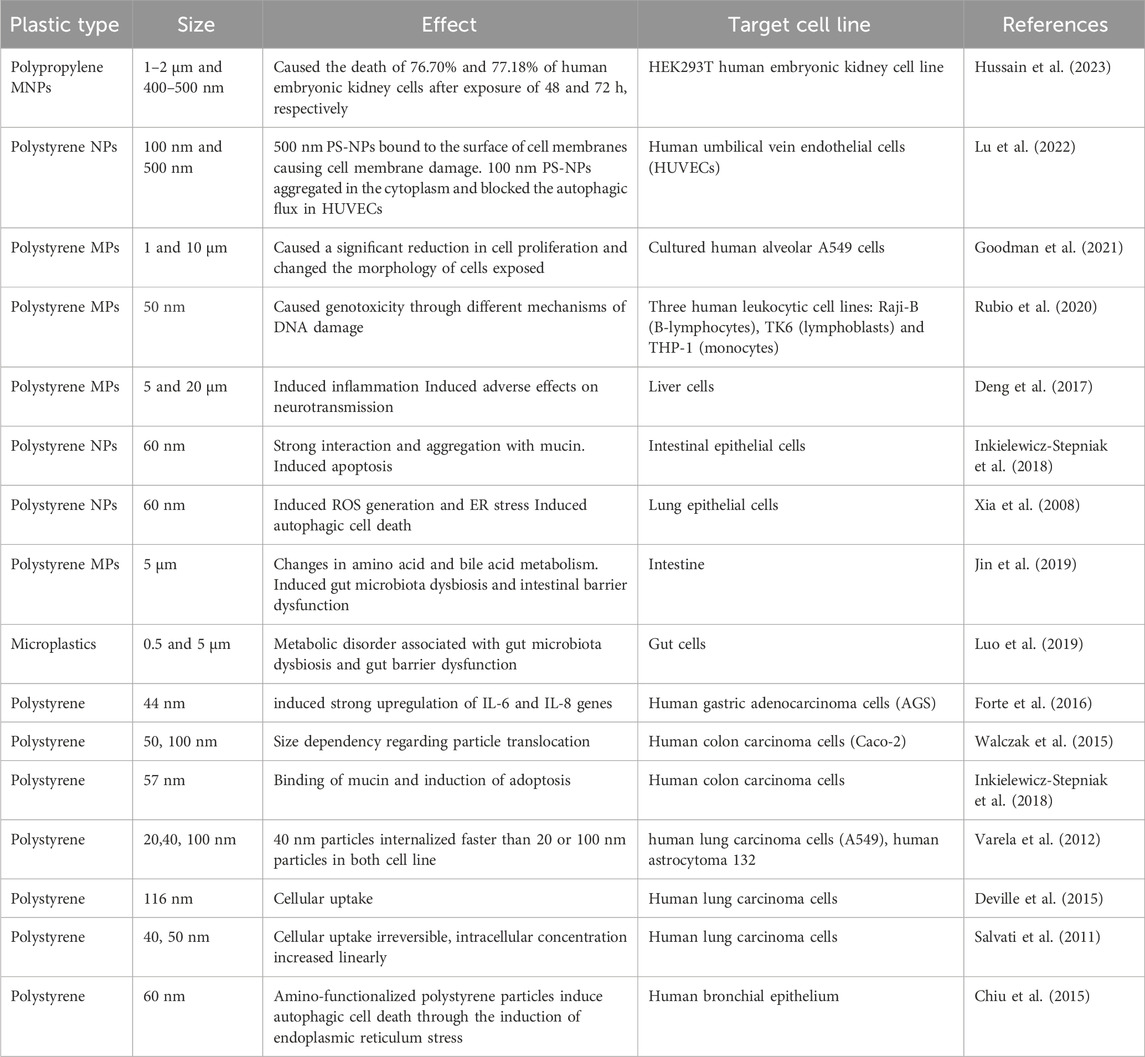
The aggregation behaviour of globular PS-NPs is influenced by the chemical conditions of the solution, which may be enhanced by increasing ionic strength and electrolyte valence (Cai et al., 2021). In freshwater biofilms, PS-NPs (positively charged amide-modified) are more hazardous to photosynthesis and extracellular enzymatic activity than negatively charged particles (Miao et al., 2019). Eutrophication may be aggravated by freshwater NPs and marine rotifer Brachionus koreanus showed elevated stress effects from NPs, and the related oxidative stress caused damage to the lipid membranes (Jeong et al., 2018; Feng et al., 2020b). Since their ingestion has been seen in numerous aquatic species (marine mammals, turtles, and fish) as well as invertebrates (zooplankton, bivalves, and crustaceans), plastic particles have raised some serious environmental concerns (Botterell et al., 2019; Wang et al., 2019; Huang et al., 2020; Zitouni et al., 2020; Naz et al., 2023a). Aside from particle features, the environment also has an impact on how NP pollution affects aquatic species. Exopolymeric substances (EPS) are the aggregation agents produced by microorganisms; nevertheless, when synthesized by diatoms and algae, they have been proven to inhibit NP harmful effects (Grassi et al., 2020; Mao et al., 2020). Apart from that various toxicological effects of MNPs are also reported in different species (Table 1).
3 Ecological toxicity and human health risk
3.1 Effect on organisms
In addition to their small size, physical and chemical properties of M NPs, can have a significant impact on aquatic species and human health. Adsorption of harmful chemicals on the MNPs raises concerns about how various lethal chemicals may interact with these particles, desorbing into animal tissues and causing harmful effects (Yu et al., 2019; Zhang et al., 2020). Nano-plastics have a greater surface area than MPs, allowing them to adsorb contaminants such as hazardous compounds or heavy metals at higher concentrations (Al-Thawadi, 2020; Naz et al., 2023b). These can be ingested by organisms and then transported and accumulated in their different organs. Aquatic life at all trophic levels, including bacteria, bivalves, algae, echinoderms, rotifers, arthropods, and fish, can be affected by NPs in terms of reproduction, mortality, multiple molting, growth, feeding, immunological responses, and antioxidation (Liu et al., 2019; Bibi et al., 2023). Once NPs enter the aquatic environment, they are easily transported down the food chain, posing a major threat to the ecological environment’s long-term growth, as well as food safety and human health (Zhang F. et al., 2020; Shi et al., 2020).
The interaction of NPs with heavy metals, polycyclic aromatic hydrocarbons, medicines, organic halogens, and pesticides, has become a major concern of environmental risks (Jacob et al., 2020). Extensive research has been conducted on the ecological toxicity of NPs, but few have been conducted on the combined toxicity induced by compound pollution (Bhagat et al., 2020b; Zhu et al., 2020). Interactions with co-pollutants can modify the uptake and accumulation of plastics and/or contaminants in exposed organisms, causing significant changes in the surface characteristics of plastics (Ghaffar et al., 2018; Zhang et al., 2020). The toxicity of MPs to organisms is determined by their aggregate size (Zhang et al., 2019). Because particle toxicity was inversely related to size in general, the aggregated MPs could be less bioavailable to aquatic organisms (Wang et al., 2020; Choi et al., 2020). Outside the organisms, MPs aggregates may have a harmful effect. MPs aggregates, for example, impeded photosynthesis and limited the transfer of nutrients and energy by microalgae in marine ecosystems. Furthermore, MP-biota hetero-aggregates may cause physical harm to organisms, such as splits and oxidative stress (Wu et al., 2019; Zhu et al., 2019; Choi et al., 2020).
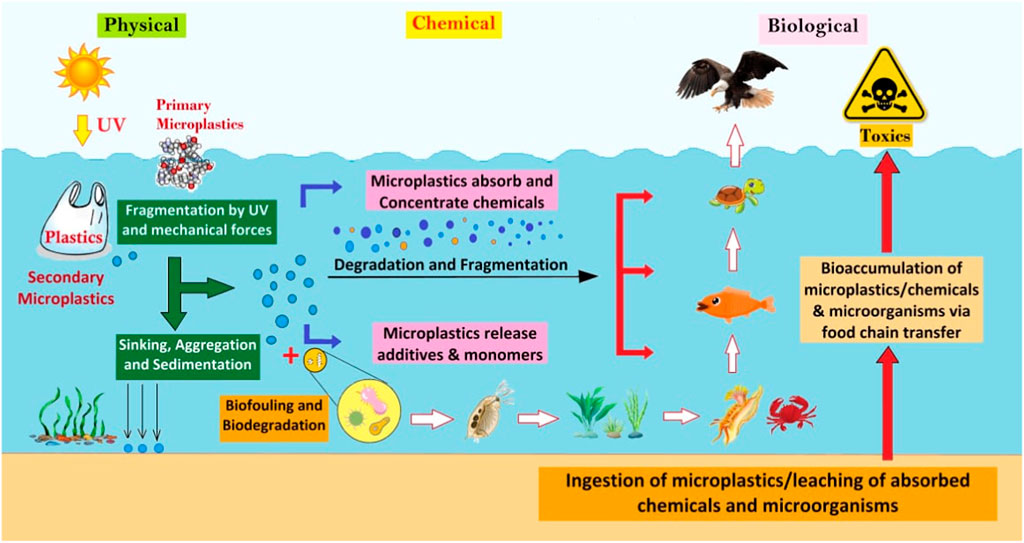
There is still a lack of knowledge about the hazardous contaminants, additives, and infections found in fish and shellfish, as well as their potential consequences on human health. According to the Food and Agriculture Organization (FAO) essential food risk evaluations are lacking, with no information on metabolism and nothing on the excretion of MPs and NPs after intake (Al-Thawadi, 2020). Accumulation and biomagnification of hazardous compounds connected with MPs in marine trophic webs is another harmful impact (Figure 1). When top predators and humans consume species polluted with MPs or chemicals released from these particles after ingestion, this magnification raises the danger of harmful effects of these chemicals (Gallo et al., 2018; Vedolin et al., 2018). As a result, it is been suggested that plastic debris raises the global risk of human and animal diseases by creating new contamination/infection pathways, introducing pathogens through the environmental spread of MPs, or migrating organisms contaminated with MPs linked to pathogens (Bhagat et al., 2020a; Al-Thawadi, 2020; Haroon et al., 2022).
3.1.1 Effects on mammals
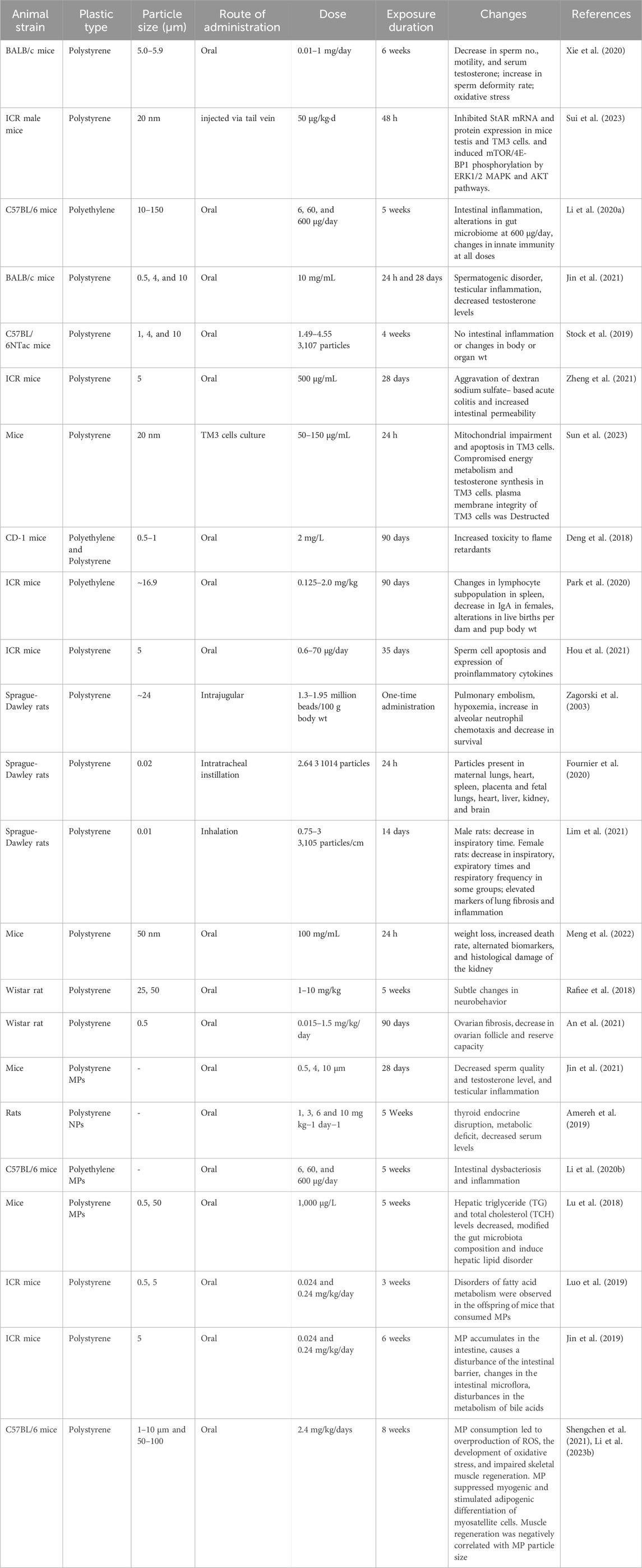
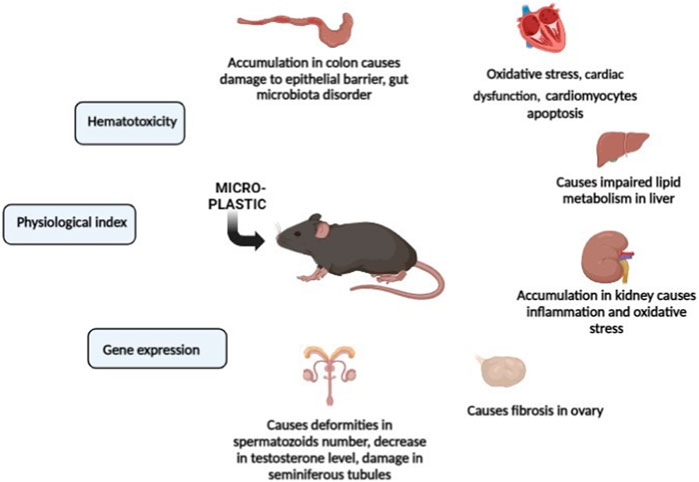
One of the most prominent classes of non-natural products made by humans that have pervaded earth’s surface environment is plastics, so much so that these durable synthetic organic polymers are heralded as a defining stratigraphic marker for the Anthropocene (Zalasiewicz et al., 2016). Geyer and colleagues (Geyer et al., 2017) recently estimated that 8.3 billion metric tons of virgin plastics have been produced up to the year 2017, and with the continuation of current production and waste management practices, about 12 billion tons of plastic waste would be found in landfills and the natural environment by 2050. Plastic wastes are persistent environmental pollutants. Larger pieces of plastic waste present well-publicized ecological problems in terms of physical entanglement and entrapment (Gündoğdu et al., 2019). In the past 3 years, a good number of studies have examined the effect of pristine MNPs in mammalian models (largely mice). These studies are summarized in Table 2 and are broadly recapped below. In mice, ingested MNPs could be found in the gut (Deng et al., 2017), liver and kidney (Yang et al., 2019). Pathological changes to the gut include a reduction in mucus secretion, gut barrier dysfunction (Jin et al., 2019), intestinal inflammation, and gut microbiota dysbiosis (Lu et al., 2018; Li B. et al., 2020). Figure 2 shows the effects of microplastic on mammalian model species (mouse).
3.2 Effects on human health
Studies on the toxic effects of M NPs on human health are mainly focused on gastrointestinal and pulmonary toxicity, which includes oxidative stress, metabolic problems, and inflammatory reactions. Furthermore, it is crucial to know whether MPs can be destroyed further after ingestion in the gut’s acidic environment or inside cells’ lysosomes. As a result, greater research into the long-term fate of ingested MPs and NPs in the human body is required (Yee et al., 2021).
Micro-plastics have been found in a variety of seafood species, including bivalves, fish, and shrimp as well as in sea salt and food packaging (Peixoto et al., 2019; Li et al., 2020; Jacob et al., 2020). These are thought to be bio-persistent, causing unfavourable biological responses in humans such as oxidative stress, inflammation, cell apoptosis, genotoxicity, and tissue necrosis, as well as localized cell and tissue damage, fibrosis, and even carcinogenesis (Peixoto et al., 2019). Ingestion, oral inhalation, or skin contact with NPs may occur as a result of the usage of plastic items or through unintended methods (Lehner et al., 2019). As a result, human exposure to NPs has been attributed to the ingestion of NP particles, which can be easily ingested through the consumption of contaminated seafood or water. If NPs enter the gastrointestinal tract, they can cause tissue inflammation or enter the circulatory system via the mesenteric lymph, where they can build up in the liver. Furthermore, oxidative stress, the gut microbiome, and lipid metabolism have all shown significant modifications. As a result, NPs may affect the central nervous system in humans (Mattsson et al., 2017). Most of the reported studies used polystyrene due to its ease of synthesis and processing into nanoparticles, whereas polyurethanes, polyolefins (e.g., polyethylene and polypropylene), polyesters, and are the most often used commercial plastics (Gunasekaran et al., 2020). The hazardous effects of different forms of MNPs on human health are mainly unknown due to variations in the shape, particle size, and chemical composition of plastics (Leslie and Depledge, 2020; Khan et al., 2023). Table 3 shows various studies related to the effect of micro and nano-plastics on human beings. Recent studies showed that various types of MNPs can affect the survival of human foetus during early embryonic development (Hussain et al., 2023). Likewise, the MNPs can cause severe damage to cell membrane (Lu et al., 2022), alter the morphology of the exposed human alveolar cells (Goodman et al., 2021) and cause genotoxicity in human blood cells (Rubio et al., 2020).
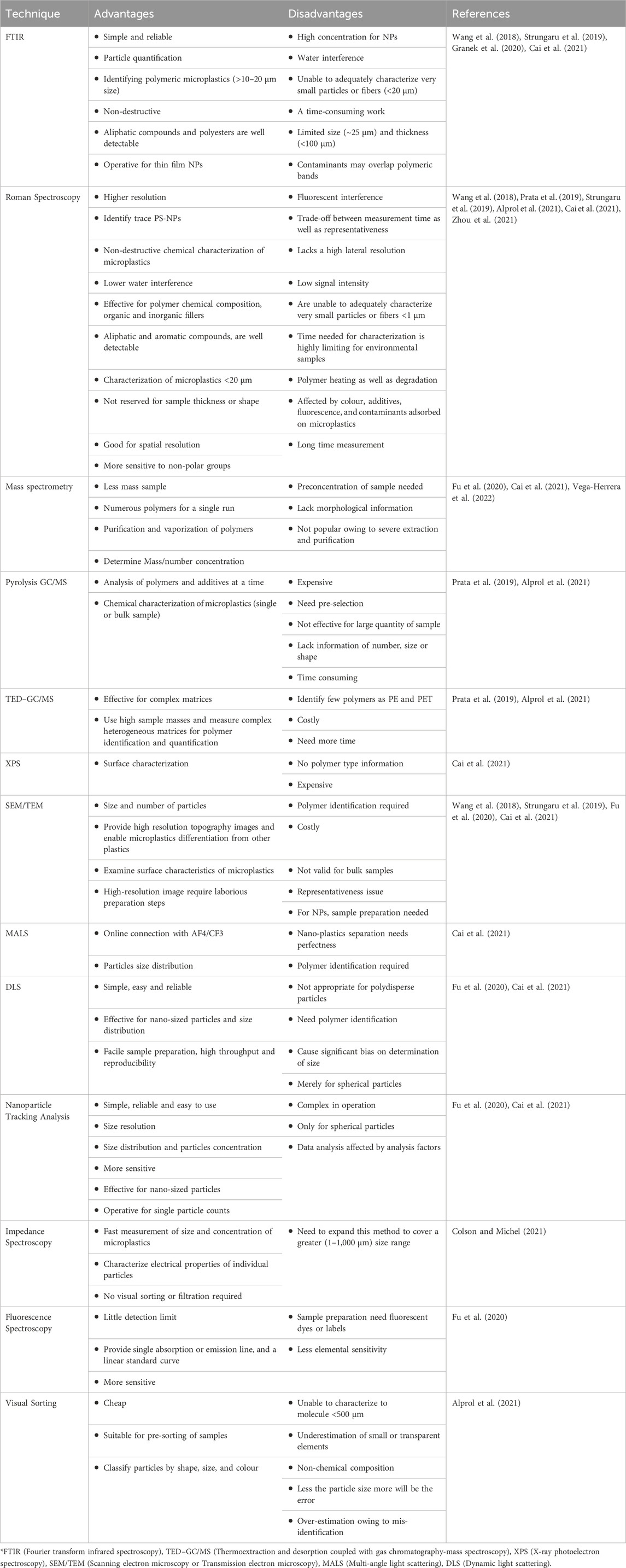
As a result, we recommend that future research needs focus on determining the potential risks associated with chronic exposure to various M NPs at appropriate concentrations. Unfortunately, the assessment of human exposure to NPs is still a scientific challenge owing to inappropriate methods, practiced reference materials, and standard analytical techniques (Brachner et al., 2020; Paul et al., 2020). Some common techniques used for the identification of M NPs are listed in table (Table 4).
4 Conclusion
Micro and nano-plastics are significant sources of plastic contamination in marine ecosystems and the production of M NPs has increased due to biodegradation, thermo-oxidative degradation, thermal and hydrolysis processes, and also photodegradation. The effects of MPs on marine life are well explored. However, their effects on freshwater species have very little literature as data on freshwater species is insufficient. So, freshwater systems are suffering from severe contamination compared with marine systems and the ecotoxicological effects of M NPs on freshwater species need more research efforts. The development of analytical methods for M NPs, as well as their standardization, is becoming more important to allow the detection, identification, and quantification of polymers in environmental matrices. While research on micro and nano-plastics is advancing rapidly, several significant limitations and gaps like lack of standardized methods for detection and characterization, limited understanding of fate and behavior of MNPs, ecological effects of MNPs on different trophic levels, long-term effects of MNPs, and ingestion and trophic transfer of MNPs still exist. Addressing these limitations and filling these knowledge gaps is essential for developing effective mitigation strategies, informing policy decisions, and safeguarding both aquatic ecosystems and human health from the impacts of micro and nano-plastic pollution. Furthermore, New ways to study the impacts of MNPs on the biota and humans (in vitro) are also required.
Author contributions
SN: Conceptualization, Data curation, Investigation, Methodology, Resources, Software, Validation, Writing–original draft, Writing–review and editing. AC: Conceptualization, Data curation, Investigation, Methodology, Writing–original draft, Writing–review and editing. NK: Conceptualization, Data curation, Investigation, Writing–review and editing. QU: Conceptualization, Project administration, Supervision, Validation, Visualization, Writing–original draft, Writing–review and editing. FZ: Conceptualization, Data curation, Investigation, Writing–review and editing. AQ: Data curation, Methodology, Writing–review and editing. IM: Conceptualization, Data curation, Writing–review and editing. AK: Data curation, Investigation, Methodology, Writing–review and editing. SS: Writing–review and editing. SB: Writing–review and editing. MK: Conceptualization, Data curation, Visualization, Writing–review and editing. PH: Funding acquisition, Investigation, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing–original draft, Writing–review and editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study was funded under project TAČR SS06020224 Development of an analytical platform for monitoring microplastic circulation in agricultural production.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Alimi, O. S., Farner Budarz, J., Hernandez, L. M., and Tufenkji, N. (2018). Microplastics and nanoplastics in aquatic environments: aggregation, deposition, and enhanced contaminant transport. Environ. Sci. Technol. 52 (4), 1704–1724. doi:10.1021/acs.est.7b05559
Alprol, A. E., Gaballah, M. S., and Hassaan, M. A. (2021). Micro and nanoplastics analysis: focus on their classification, sources, and impacts in marine environment. Reg. Stud. Mar. Sci. 42, 101625. doi:10.1016/j.rsma.2021.101625
Al-Thawadi, S. (2020). Microplastics and nanoplastics in aquatic environments: challenges and threats to aquatic organisms. Arab. J. Sci. Eng. 45 (6), 4419–4440. doi:10.1007/s13369-020-04402-z
Amereh, F., Eslami, A., Fazelipour, S., Rafiee, M., Zibaii, M. I., and Babaei, M. (2019). Thyroid endocrine status and biochemical stress responses in adult male Wistar rats chronically exposed to pristine polystyrene nanoplastics. Toxicol. Res. 8, 953–963. doi:10.1039/c9tx00147f
An, R., Wang, X., Yang, L., Zhang, J., Wang, N., Xu, F., et al. (2021). Polystyrene microplastics cause granulosa cells apoptosis and fibrosis in ovary through oxidative stress in rats. Toxicology 449, 152665. doi:10.1016/j.tox.2020.152665
Bagheri, T., Gholizadeh, M., Zakeri, M., Hedayati, A., Rabaniha, M., Aghaeimoghadam, A., et al. (2020). Microplastics distribution, abundance and composition in sediment, fishes and benthic organisms of the gorgan Bay, Caspian Sea. Chemosphere 257, 127201. doi:10.1016/j.chemosphere.2020.127201
Barboza, L. G. A., Vethaak, A. D., Lavorante, B. R., Lundebye, A. K., and Guilhermino, L. (2018). Marine microplastic debris: an emerging issue for food security, food safety and human health. Mar. Pollut. Bull. 133, 336–348. doi:10.1016/j.marpolbul.2018.05.047
Barría, C., Brandts, I., Tort, L., Oliveira, M., and Teles, M. (2020). Effect of nanoplastics on fish health and performance: a review. Mar. Pollut. Bull. 151, 110791. doi:10.1016/j.marpolbul.2019.110791
Bergami, E., Pugnalini, S., Vannuccini, M. L., Manfra, L., Faleri, C., Savorelli, F., et al. (2017). Long-term toxicity of surface-charged polystyrene nanoplastics to marine planktonic species Dunaliella tertiolecta and Artemia franciscana. Aquat. Toxicol. 189, 159–169. doi:10.1016/j.aquatox.2017.06.008
Bhagat, J., Nishimura, N., and Shimada, Y. (2020a). Toxicological interactions of microplastics/nanoplastics and environmental contaminants: current knowledge and future perspectives. J. Hazard. Mat. 405, 123913. doi:10.1016/j.jhazmat.2020.123913
Bhagat, J., Zang, L., Nishimura, N., and Shimada, Y. (2020b). Zebrafish: an emerging model to study microplastic and nanoplastic toxicity. Sci. Total Environ. 728, 138707. doi:10.1016/j.scitotenv.2020.138707
Bibi, S., Abbas, G., Khan, M. Z., Nawaz, T., Ullah, Q., Uddin, A., et al. (2023). The mutational analysis of mitochondrial DNA in maternal inheritance of polycystic ovarian syndrome. Front. Endocrinol. 14, 1093353. doi:10.3389/fendo.2023.1093353
Botterell, Z. L., Beaumont, N., Dorrington, T., Steinke, M., Thompson, R. C., and Lindeque, P. K. (2019). Bioavailability and effects of microplastics on marine zooplankton: a review. Environ. Pollut. 245, 98–110. doi:10.1016/j.envpol.2018.10.065
Brachner, A., Fragouli, D., Duarte, I. F., Farias, P., Dembski, S., Ghosh, M., et al. (2020). Assessment of human health risks posed by nano-and microplastics is currently not feasible. Int. J. Environ. Res. Public Health 17 (23), 8832. doi:10.3390/ijerph17238832
Brancaleone, E., Mattei, D., Fuscoletti, V., Lucentini, L., Favero, G., Frugis, A., et al. (2023). “Microplastic in drinking water: a pilot study,” in Proceedings of the 3rd international conference on microplastic pollution in the mediterranean sea (Cham: Springer International Publishing), 165–172.
Brandts, I., Garcia-Ordoñez, M., Tort, L., Teles, M., and Roher, N. (2020). Polystyrene nanoplastics accumulate in ZFL cell lysosomes and in zebrafish larvae after acute exposure, inducing a synergistic immune response in vitro without affecting larval survival in vivo. Environ. Sci. Nano 7 (8), 2410–2422. doi:10.1039/d0en00553c
Brandts, I., Teles, M., Tvarijonaviciute, A., Pereira, M. L., Martins, M. A., Tort, L., et al. (2018). Effects of polymethylmethacrylate nanoplastics on Dicentrarchus labrax. Genom 110 (6), 435–441. doi:10.1016/j.ygeno.2018.10.006
Cai, H., Xu, E. G., Du, F., Li, R., Liu, J., and Shi, H. (2021). Analysis of environmental nanoplastics: progress and challenges. Chem. Eng. J. 410, 128208. doi:10.1016/j.cej.2020.128208
Chae, Y., Kim, D., Kim, S. W., and An, Y. J. (2018). Trophic transfer and individual impact of nano-sized polystyrene in a four-species freshwater food chain. Sci. Rep. 8 (1), 284–311. doi:10.1038/s41598-017-18849-y
Chiu, H. W., Xia, T., Lee, Y. H., Chen, C. W., Tsai, J. C., and Wang, Y. J. (2015). Cationic polystyrene nanospheres induce autophagic cell death through the induction of endoplasmic reticulum stress. J. Nanobiotechnology 13, 76. doi:10.1039/c4nr05509h
Choi, J. S., Hong, S. H., and Park, J. W. (2020). Evaluation of microplastic toxicity in accordance with different sizes and exposure times in the marine copepod Tigriopus japonicus. Mar. Environ. Res. 153, 104838. doi:10.1016/j.marenvres.2019.104838
Colson, B. C., and Michel, A. P. (2021). Flow-through quantification of microplastics using impedance spectroscopy. ACS Sens. 6 (1), 238–244. doi:10.1021/acssensors.0c02223
Deng, Y., Zhang, Y., Lemos, B., and Ren, H. (2017). Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci. Rep. 7, 46687. doi:10.1038/srep46687
Deng, Y., Zhang, Y., Lemos, B., Ren, H., Yang, X., et al. (2018). Evidence that microplastics aggravate the toxicity of organophosphorus flame retardants in mice (Mus musculus). J. Hazard Mater 357, 348–354. doi:10.1016/j.jhazmat.2018.06.017
Deville, S., Penjweini, R., Smisdom, N., Notelaers, K., Nelissen, I., Hooyberghs, J., et al. (2015). Intracellular dynamics and fate of polystyrene nanoparticles in A549 lung epithelial cells monitored by image (cross-)correlation spectroscopy and single particle tracking. Biochim. Biophys. Acta Mol. Cell Res. 1853, 2411–2419. doi:10.1016/j.bbamcr.2015.07.004
Enfrin, M., Lee, J., Gibert, Y., Basheer, F., Kong, L., and Dumée, L. F. (2020). Release of hazardous nanoplastic contaminants due to microplastics fragmentation under shear stress forces. J. Hazard. Mat. 384, 121393. doi:10.1016/j.jhazmat.2019.121393
Feng, L. J., Shi, Y., Li, X. Y., Sun, X. D., Xiao, F., Sun, J. W., et al. (2020b). Nanoplastics promote microcystin synthesis and release from cyanobacterial Microcystis aeruginosa. Environ. Sci. Technol. 54 (6), 3386–3394. doi:10.1021/acs.est.9b06085
Feng, L. J., Sun, X. D., Zhu, F. P., Feng, Y., Duan, J. L., Xiao, F., et al. (2020a). Behavior of tetracycline and polystyrene nanoparticles in estuaries and their joint toxicity on marine microalgae Skeletonema costatum. Environ. Pollut. 263, 114453. doi:10.1016/j.envpol.2020.114453
Fernandes, V. C., Boesmans, D., Domingues, V. F., and Delerue-Matos, C. (2023). Evaluating contaminants in fish: plastic additives and pesticides in the context of food safety. Biol. Life Sci. Forum. doi:10.3390/Foods2023-15052
Ferreira, I., Venâncio, C., Lopes, I., and Oliveira, M. (2019). Nanoplastics and marine organisms: what has been studied? Environ. Toxicol. Pharmacol. 67, 1–7. doi:10.1016/j.etap.2019.01.006
Food and Agriculture Organization. The state of food insecurity in the world 2013: the multiple dimensions of food security. Rome: FAO; 2013. Available at: https://www.fao.org/news/archive/news-by-date/2013/en/(accessed on (accessed on 23 February 2024).
Forte, M., Iachetta, G., Tussellino, M., Carotenuto, R., Prisco, M., De Falco, M., et al. (2016). Polystyrene nanoparticles internalization in human gastric adenocarcinoma cells. Toxicol Vitro 31, 126–136. doi:10.1016/j.tiv.2015.11.006
Fournier, S. B., D'Errico, J. N., Adler, D. S., Kollontzi, S., Goedken, M. J., Fabris, L., et al. (2020). Nanopolystyrene translocation and fetal deposition after acute lung exposure during late-stage pregnancy. Part Fibre Toxicol. 17, 55. doi:10.1186/s12989-020-00385-9
Frias, JPGL, and Nash, R. (2019). Microplastics: finding a consensus on the definition. Mar. Pollut. Bull. 138, 145–147. doi:10.1016/j.marpolbul.2018.11.022
Fu, W., Min, J., Jiang, W., Li, Y., and Zhang, W. (2020). Separation, characterization and identification of microplastics and nanoplastics in the environment. Sci. Total Environ. 721, 137561. doi:10.1016/j.scitotenv.2020.137561
Gagné, F., Roubeau-Dumont, E., André, C., and Auclair, J. (2023). Micro and nanoplastic contamination and its effects on freshwater mussels caged in an urban area. J. Xenobiotics 13, 761–774. doi:10.3390/jox13040048
Gallo, F., Fossi, C., Weber, R., Santillo, D., Sousa, J., Ingram, I., et al. (2018). Marine litter plastics and microplastics and their toxic chemicals components: the need for urgent preventive measures. Environ. Sci. Eur. 30 (1), 13–14. doi:10.1186/s12302-018-0139-z
Geyer, R., Jambeck, J. R., and Law, K. L. (2017). Production, use, and fate of all plastics ever made. Sci. Adv. 3, e1700782. doi:10.1126/sciadv.1700782
Ghaffar, A., Jamil, H., Zubair, M., Farooq, M., Murtaza, A., and Ullah, Q. (2018). Effect of dietary supplementation with propylene glycol on blood metabolites and hormones of nili-ravi Buffalo heifers: effect of dietary supplementation on blood metabolites. Proc. Pak. Acad. Sci. B Life Environ. Sci. 55 (3), 55–59. Available at: https://ppaspk.org/index.php/PPAS-B/article/view/164
Gigault, J., Ter Halle, A., Baudrimont, M., Pascal, P. Y., Gauffre, F., Phi, T. L., et al. (2018). Current opinion: what is a nanoplastic? Environ. Pollut. 235, 1030–1034. doi:10.1016/j.envpol.2018.01.024
Gomes, T., Almeida, A. C., and Georgantzopoulou, A. (2020). Characterization of cell responses in Rhodomonas baltica exposed to PMMA nanoplastics. Sci. Total Environ. 726, 138547. doi:10.1016/j.scitotenv.2020.138547
González-Fernández, C., Toullec, J., Lambert, C., Le Goïc, N., Seoane, M., Moriceau, B., et al. (2019). Do transparent exopolymeric particles (TEP) affect the toxicity of nanoplastics on Chaetoceros neogracile? Environ. Pollut. 250, 873–882. doi:10.1016/j.envpol.2019.04.093
Goodman, K. E., Hare, J. T., Khamis, Z. I., Hua, T., and Sang, Q. X. A. (2021). Exposure of human lung cells to polystyrene microplastics significantly retards cell proliferation and triggers morphological changes. Chem. Res. Toxicol. 34, 1069–1081. doi:10.1021/acs.chemrestox.0c00486
Granek, E. F., Brander, S., and Holland, E. B. (2020). Microplastics in aquatic organisms: improving understanding and identifying research directions for the next decade. Limnol. Oceanogr. Lett. 5, 1–4. doi:10.1002/lol2.10145
Grassi, G., Gabellieri, E., Cioni, P., Paccagnini, E., Faleri, C., Lupetti, P., et al. (2020). Interplay between extracellular polymeric substances (EPS) from a marine diatom and model nanoplastic through eco-corona formation. Sci. Total Environ. 725, 138457. doi:10.1016/j.scitotenv.2020.138457
Guimarães, A. T. B., Estrela, F. N., Pereira, P. S., Vieira, J. E. A., Rodrigues, A. S. L., Silva, F. G., et al. (2021). Toxicity of polystyrene nanoplastics in Ctenopharyngodon idella juveniles: a genotoxic, mutagenic and cytotoxic perspective. Sci. Total Environ. 752, 141937. doi:10.1016/j.scitotenv.2020.141937
Gunasekaran, D., Chandrasekaran, N., Jenkins, D., and Mukherjee, A. (2020). Plain polystyrene microplastics reduce the toxic effects of ZnO particles on marine microalgae Dunaliella salina. J. Environ. Chem. Eng. 8 (5), 104250. doi:10.1016/j.jece.2020.104250
Gündoğdu, S., Yeşilyurt, İ. N., and Erbaş, C. (2019). Potential interaction between plastic litter and green turtle Chelonia mydas during nesting in an extremely polluted beach. Mar. Pollut. Bull. 140, 138–145. doi:10.1016/j.marpolbul.2019.01.032
Han, B., Yacoub, M., Li, A., Nicholson, K., Gruver, J., Neumann, K., et al. (2024). Human activities increased microplastics contamination in the himalaya mountains. Hydrology 11, 4. doi:10.3390/hydrology11010004
Haroon, M., Jamil, H., Ullah, Q., Farooq, Q., and Inamullah, M. (2022). Cholesterol and serum minerals profile in the pregnancy and peurperium period of Beetal Goats. Biosci. Res. 19 (1), 381–385.
Hazeem, L. J., Yesilay, G., Bououdina, M., Perna, S., Cetin, D., Suludere, Z., et al. (2020). Investigation of the toxic effects of different polystyrene micro-and nanoplastics on microalgae Chlorella vulgaris by analysis of cell viability, pigment content, oxidative stress and ultrastructural changes. Mar. Pollut. Bull. 156, 111278. doi:10.1016/j.marpolbul.2020.111278
Heinlaan, M., Kasemets, K., Aruoja, V., Blinova, I., Bondarenko, O., Lukjanova, A., et al. (2020). Hazard evaluation of polystyrene nanoplastic with nine bioassays did not show particle-specific acute toxicity. Sci. Total Environ. 707, 136073. doi:10.1016/j.scitotenv.2019.136073
Herrera, D. A. G., Mojicevic, M., Pantelic, B., Joshi, A., Collins, C., Batista, M., et al. (2023). Exploring microorganisms from plastic-polluted sites: unveiling plastic degradation and PHA production potential. Microorganisms 11, 2914. doi:10.3390/microorganisms11122914
Hou, B., Wang, F., Liu, T., and Wang, Z. (2021). Reproductive toxicity of polystyrene microplastics: in vivo experimental study on testicular toxicity in mice. J. Hazard Mater 405, 124028. doi:10.1016/j.jhazmat.2020.124028
Huang, D., Tao, J., Cheng, M., Deng, R., Chen, S., Yin, L., et al. (2020). Microplastics and nanoplastics in the environment: macroscopic transport and effects on creatures. J. Hazard. Mat. 407, 124399. doi:10.1016/j.jhazmat.2020.124399
Hussain, K. A., Romanova, S., Okur, I., Zhang, D., Kuebler, J., Huang, X., et al. (2023). Assessing the release of microplastics and nanoplastics from plastic containers and reusable food pouches: implications for human health. Environ. Sci. Technol. 57, 9782–9792. doi:10.1021/acs.est.3c01942
Inkielewicz-Stepniak, I., Tajber, L., Behan, G., Zhang, H., Radomski, M. W., Medina, C., et al. (2018). The role of mucin in the toxicological impact of polystyrene nanoparticles. Materials 11, 724. doi:10.3390/ma11050724
Jabeen, K., Li, B., Chen, Q., Su, L., Wu, C., Hollert, H., et al. (2018). Effects of virgin microplastics on goldfish (Carassius auratus). Chemosphere 213, 323–332. doi:10.1016/j.chemosphere.2018.09.031
Jacob, H., Besson, M., Swarzenski, P. W., Lecchini, D., and Metian, M. (2020). Effects of virgin micro-and nanoplastics on fish: trends, meta-analysis, and perspectives. Environ. Sci. Technol. 54 (8), 4733–4745. doi:10.1021/acs.est.9b05995
Jeong, C. B., Kang, H. M., Lee, Y. H., Kim, M. S., Lee, J. S., Seo, J. S., et al. (2018). Nanoplastic ingestion enhances toxicity of persistent organic pollutants (POPs) in the monogonont rotifer Brachionus koreanus via multixenobiotic resistance (MXR) disruption. Environ. Sci. Technol. 52 (19), 11411–11418. doi:10.1021/acs.est.8b03211
Jiang, N., Luo, C., Shao, M., Zheng, Z., Ullah, Q., Khan, M. Z., et al. (2023a). Uncovering the role of ribosomal protein L8 in milk fat synthesis mechanisms in yak mammary epithelial cells. Pak. J. Zool., 1–16. doi:10.17582/journal.pjz/20230415100435
Jiang, N., Naz, S., Ma, Y., Ullah, Q., Khan, M. Z., Wang, J., et al. (2023b). An overview of comet assay application for detecting DNA damage in aquatic animals. Agriculture 13 (3), 623. doi:10.3390/agriculture13030623
Jin, H., Ma, T., Sha, X., Liu, Z., Zhou, Y., Meng, X., et al. (2021). Polystyrene microplastics induced male reproductive toxicity in mice. J. Hazard Mater 401, 123430. doi:10.1016/j.jhazmat.2020.123430
Jin, Y., Lu, L., Tu, W., Luo, T., and Fu, Z. (2019). Impacts of polystyrene microplastic on the gut barrier, microbiota, and metabolism of mice. Sci. Total Environ. 649, 308–317. doi:10.1016/j.scitotenv.2018.08.353
Khan, R. U., Khan, M., Ullah, Q., Khan, M. Z., Sohail, A., Islam, R., et al. (2023). In vitro and in vivo effects of conventional and chitosan nanoparticle-encapsulated miltefosine drug for treatment of cutaneous leishmaniasis. Med. Sci. Forum 21 (1), 19. doi:10.3390/ECB2023-14334
Kudzin, M. H., Piwowarska, D., Festinger, N., and Chruściel, J. J. (2024). Risks associated with the presence of polyvinyl chloride in the environment and methods for its disposal and utilization. Materials 17, 173. [Online]. doi:10.3390/ma17010173
Lee, W. S., Cho, H. J., Kim, E., Huh, Y. H., Kim, H. J., Kim, B., et al. (2019). Bioaccumulation of polystyrene nanoplastics and their effect on the toxicity of Au ions in zebrafish embryos. Nanoscale 11 (7), 3173–3185. doi:10.1039/c8nr09321k
Lehner, R., Weder, C., Petri-Fink, A., and Rothen-Rutishauser, B. (2019). Emergence of nanoplastic in the environment and possible impact on human health. Environ. Sci. Technol. 53 (4), 1748–1765. doi:10.1021/acs.est.8b05512
Lei, L., Wu, S., Lu, S., Liu, M., Song, Y., Fu, Z., et al. (2018). Microplastic particles cause intestinal damage and other adverse effects in zebrafish Danio rerio and nematode Caenorhabditis elegans. Sci. Total Environ. 619-620, 1–8. doi:10.1016/j.scitotenv.2017.11.103
Leslie, H. A., and Depledge, M. H. (2020). Where is the evidence that human exposure to microplastics is safe? Environ. Int. 142, 105807. doi:10.1016/j.envint.2020.105807
Li, B., Ding, Y., Cheng, X., Sheng, D., Xu, Z., Rong, Q., et al. (2020a). Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 244, 125492. doi:10.1016/j.chemosphere.2019.125492
Li, B., Khan, M. Z., Khan, I. M., Ullah, Q., Cisang, Z. M., Zhang, N., et al. (2023a). Genetics, environmental stress, and amino acid supplementation affect lactational performance via mTOR signaling pathway in bovine mammary epithelial cells. Front. Genet. 14, 1195774. doi:10.3389/fgene.2023.1195774
Li, S., Liu, H., Gao, R., Abdurahman, A., Dai, J., and Zeng, F. (2018). Aggregation kinetics of microplastics in aquatic environment: complex roles of electrolytes, pH, and natural organic matter. Environ. Pollut. 237, 126–132. doi:10.1016/j.envpol.2018.02.042
Li, Y., Du, X., Liu, Z., Zhang, M., Huang, Y., Tian, J., et al. (2021). Two genes related to reproductive development in the juvenile prawn, Macrobrachium nipponense: molecular characterization and transcriptional response to nanoplastic exposure. Chemosphere 281, 130827. doi:10.1016/j.chemosphere.2021.130827
Li, Y., Meng, Y., Qin, L., Shen, M., Qin, T., Chen, X., et al. (2023b). Occurrence and removal efficiency of microplastics in four drinking water treatment plants in zhengzhou, China. China. Water 16, 131. doi:10.3390/w16010131
Li, Z., Feng, C., Wu, Y., and Guo, X. (2020b). Impacts of nanoplastics on bivalve: fluorescence tracing of organ accumulation, oxidative stress and damage. J. Hazard Mater 392, 122418. doi:10.1016/j.jhazmat.2020.122418
Lim, D., Jeong, J., Song, K. S., Sung, J. H., Oh, S. M., and Choi, J. (2021). Inhalation toxicity of polystyrene micro(nano)plastics using modified OECD TG 412. Chemosphere 262, 128330. doi:10.1016/j.chemosphere.2020.128330
Liu, Z., Cai, M., Wu, D., Yu, P., Jiao, Y., Jiang, Q., et al. (2020). Effects of nanoplastics at predicted environmental concentration on Daphnia pulex after exposure through multiple generations. Environ. Pollut. 256, 113506. doi:10.1016/j.envpol.2019.113506
Liu, Z., Li, Y., Pérez, E., Jiang, Q., Chen, Q., Jiao, Y., et al. (2021). Polystyrene nanoplastic induces oxidative stress, immune defense, and glycometabolism change in Daphnia pulex: application of transcriptome profiling in risk assessment of nanoplastics. J. Hazard Mat. 402, 123778. doi:10.1016/j.jhazmat.2020.123778
Liu, Z., Yu, P., Cai, M., Wu, D., Zhang, M., Huang, Y., et al. (2019). Polystyrene nanoplastic exposure induces immobilization, reproduction, and stress defense in the freshwater cladoceran Daphnia pulex. Chemosphere 215, 74–81. doi:10.1016/j.chemosphere.2018.09.176
Lu, L., Wan, Z., Luo, T., Fu, Z., and Jin, Y. (2018). Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 631-632, 449–458. doi:10.1016/j.scitotenv.2018.03.051
Lu, Y. Y., Li, H., Ren, H., Zhang, X., Huang, F., Zhang, D., et al. (2022). Size-dependent effects of polystyrene nanoplastics on autophagy response in human umbilical vein endothelial cells. J. Hazard Mater 421, 126770. doi:10.1016/j.jhazmat.2021.126770
Luo, T., Zhang, Y., Wang, C., Wang, X., Zhou, J., Shen, M., et al. (2019). Maternal exposure to different sizes of polystyrene microplastics during gestation causes metabolic disorders in their offspring. Environ. Pollut. 255, 113122. doi:10.1016/j.envpol.2019.113122
Mao, Y., Li, H., Huangfu, X., Liu, Y., and He, Q. (2020). Nanoplastics display strong stability in aqueous environments: insights from aggregation behaviour and theoretical calculations. Environ. Pollut. 258, 113760. doi:10.1016/j.envpol.2019.113760
Mattsson, K., Johnson, E. V., Malmendal, A., Linse, S., Hansson, L. A., and Cedervall, T. (2017). Brain damage and behavioural disorders in fish induced by plastic nanoparticles delivered through the food chain. Sci. Rep. 7 (1), 11452–11457. doi:10.1038/s41598-017-10813-0
Meng, X., Zhang, J., Wang, W., Gonzalez-Gil, G., Vrouwenvelder, J. S., and Li, Z. (2022). Effects of nano- and microplastics on kidney: physicochemical properties, bioaccumulation, oxidative stress and immunoreaction. Chemosphere 288, 132631. doi:10.1016/j.chemosphere.2021.132631
Miao, L., Hou, J., You, G., Liu, Z., Liu, S., Li, T., et al. (2019). Acute effects of nanoplastics and microplastics on periphytic biofilms depending on particle size, concentration and surface modification. Environ. Pollut. 255, 113300. doi:10.1016/j.envpol.2019.113300
Mongil-Manso, J., Jiménez-Ballesta, R., Trujillo-González, J. M., San José Wery, A., and Díez Méndez, A. (2023). A comprehensive review of plastics in agricultural soils: a case study of castilla y león (Spain) farmlands. Land 12, 1888. doi:10.3390/land12101888
Naz, S., Chatha, A., Ullah, Q., Maqbool, B., Iqbal, S., Khan, A., et al. (2023a). Impact of chronic exposure to heavy metal mixtures on selected biological parameters of freshwater fish species. J. Anim. Plant Sci. 33, 2023. doi:10.36899/JAPS.2023.6.0677
Naz, S., Chatha, A. M. M., Téllez-Isaías, G., Ullah, S., Ullah, Q., Khan, M. Z., et al. (2023b). A comprehensive review on metallic trace elements toxicity in fishes and potential remedial measures. Water 15 (16), 3017. doi:10.3390/w15163017
Naz, S., Hussain, R., Ullah, Q., Chatha, A. M. M., Shaheen, A., and Khan, R. U. (2021). Toxic effect of some heavy metals on hematology and histopathology of major carp (catla catla). Environ. Sci. Pollut. Res. 28, 6533–6539. doi:10.1007/s11356-020-10980-0
Naz, S., Mansouri, B., Chatha, A. M. M., Ullah, Q., Abadeen, Z. U., Khan, M. Z., et al. (2022). Water quality and health risk assessment of trace elements in surface water at punjnad headworks, Punjab, Pakistan. Environ. Sci. Pollut. Res. 29 (40), 61457–61469. doi:10.1007/s11356-022-20210-4
Neves, R. A. F., Guimarães, T. B., and Santos, L. N. (2024). First record of microplastic contamination in the non-native dark false mussel mytilopsis leucophaeata (Bivalvia: Dreissenidae) in a coastal urban lagoon. Int. J. Environ. Res. Public Health 21, 44. doi:10.3390/ijerph21010044
Nielsen, T. D., Hasselbalch, J., Holmberg, K., and Stripple, J. (2020). Politics and the plastic crisis: a review throughout the plastic life cycle. Wiley Interdiscip. Rev. Energy Environ. 9 (1), e360. doi:10.1002/wene.360
Oliveira, M., Almeida, M., and Miguel, I. A. (2019). A micro (nano) plastic boomerang tale: a never ending story? Trac. Trends Anal. Chem. 112, 196–200. doi:10.1016/j.trac.2019.01.005
Park, E. J., Han, J. S., Park, E. J., Seong, E., Lee, G. H., Kim, D. W., et al. (2020). Repeated-oral dose toxicity of polyethylene microplastics and the possible implications on reproduction and development of the next generation. Toxicol. Lett. 324, 75–85. doi:10.1016/j.toxlet.2020.01.008
Paul, M. B., Stock, V., Cara-Carmona, J., Lisicki, E., Shopova, S., Fessard, V., et al. (2020). Micro-and nanoplastics–current state of knowledge with the focus on oral uptake and toxicity. Nanoscale Adv. 2 (10), 4350–4367. doi:10.1039/d0na00539h
Peixoto, D., Pinheiro, C., Amorim, J., Oliva-Teles, L., Guilhermino, L., and Vieira, M. N. (2019). Microplastic pollution in commercial salt for human consumption: a review. Estuar. Coast Shelf Sci. 219, 161–168. doi:10.1016/j.ecss.2019.02.018
Peller, J. R., Tabor, G., Davis, C., Iceman, C., Nwachukwu, O., Doudrick, K., et al. (2024). Distribution and fate of polyethylene microplastics released by a portable toilet manufacturer into a freshwater wetland and lake. Water 16, 11. doi:10.3390/w16010011
Peng, L., Fu, D., Qi, H., Lan, C. Q., Yu, H., and Ge, C. (2020). Micro- and nano-plastics in marine environment: source, distribution and threats- A review. Sci. Total Environ. 698, 134254. doi:10.1016/j.scitotenv.2019.134254
Pizzurro, F., Nerone, E., Ancora, M., Di Domenico, M., Mincarelli, L. F., Cammà, C., et al. (2024). Exposure of Mytilus galloprovincialis to microplastics: accumulation, depuration and evaluation of the expression levels of a selection of molecular biomarkers. Animals 14, 4. doi:10.3390/ani14010004
Prata, J. C., da Costa, J. P., Duarte, A. C., and Rocha-Santos, T. (2019). Methods for sampling and detection of microplastics in water and sediment: a critical review. Trac. Trends Anal. Chem. 110, 150–159. doi:10.1016/j.trac.2018.10.029
Qiao, R., Deng, Y., Zhang, S., Wolosker, M. B., Zhu, Q., Ren, H., et al. (2019a). Accumulation of different shapes of microplastics initiates intestinal injury and gut microbiota dysbiosis in the gut of zebrafish. Chemosphere 236, 124334. doi:10.1016/j.chemosphere.2019.07.065
Qiao, R., Sheng, C., Lu, Y., Zhang, Y., Ren, H., and Lemos, B. (2019b). Microplastics induce intestinal inflammation, oxidative stress, and disorders of metabolome and microbiome in zebrafish. Sci. Total Environ. 662, 246–253. doi:10.1016/j.scitotenv.2019.01.245
Rafiee, M., Dargahi, L., Eslami, A., Beirami, E., Jahangiri-Rad, M., Sabour, S., et al. (2018). Neurobehavioral assessment of rats exposed to pristine polystyrene nanoplastics upon oral exposure. Chemosphere 193, 745–753. doi:10.1016/j.chemosphere.2017.11.076
Rashed, A. H., Yesilay, G., Hazeem, L., Rashdan, S., Almealla, R., Kilinc, Z., et al. (2023). Micro- and nano-plastics contaminants in the environment: sources, fate, toxicity, detection, remediation, and sustainable perspectives. Water 15, 3535. doi:10.3390/w15203535
Rubio, L., Barguilla, I., Domenech, J., Marcos, R., and Hernández, A. (2020). Biological effects, including oxidative stress and genotoxic damage, of polystyrene nanoparticles in different human hematopoietic cell lines. J. Hazard Mater 398, 122900. doi:10.1016/j.jhazmat.2020.122900
Sallam, M. A., Zubair, M., Gul, S. T., Ullah, Q., and Idrees, M. (2020). Evaluating the protective effects of vitamin E and selenium on hematology and liver, lung and uterus histopathology of rabbits with cypermethrin toxicity. Toxin Rev. 39 (3), 236–241. doi:10.1080/15569543.2018.1518335
Salvati, A., Åberg, C., Dos Santos, T., Varela, J., Pinto, P., Lynch, I., et al. (2011). Experimental and theoretical comparison of intracellular import of polymeric nanoparticles and small molecules: toward models of uptake kinetics. Nanomedicine 7, 818–826. doi:10.1016/j.nano.2011.03.005
Sarasamma, S., Audira, G., Siregar, P., Malhotra, N., Lai, Y. H., Liang, S. T., et al. (2020). Nanoplastics cause neurobehavioral impairments, reproductive and oxidative damages, and biomarker responses in zebrafish: throwing up alarms of wide spread health risk of exposure. Int. J. Mol. Sci. 21 (4), 1410. doi:10.3390/ijms21041410
Sendra, M., Sparaventi, E., Blasco, J., Moreno-Garrido, I., and Araujo, C. V. (2020). Ingestion and bioaccumulation of polystyrene nanoplastics and their effects on the microalgal feeding of Artemia franciscana. Ecotoxicol. Environ. Saf. 188, 109853. doi:10.1016/j.ecoenv.2019.109853
Sendra, M., Staffieri, E., Yeste, M. P., Moreno-Garrido, I., Gatica, J. M., Corsi, I., et al. (2019). Are the primary characteristics of polystyrene nanoplastics responsible for toxicity and ad/absorption in the marine diatom Phaeodactylum tricornutum? Environ. Pollut. 249, 610–619. doi:10.1016/j.envpol.2019.03.047
Shengchen, W., Jing, L., Yujie, Y., Yue, W., and Shiwon, X. (2021). Polystyrene microplastics-induced ROS overproduction disrupts the skeletal muscle regeneration by converting myoblasts into adipocytes. J. Hazard Mater 417, 125962. doi:10.1016/j.jhazmat.2021.125962
Shi, W., Han, Y., Sun, S., Tang, Y., Zhou, W., Du, X., et al. (2020). Immunotoxicities of microplastics and sertraline, alone and in combination, to a bivalve species: size-dependent interaction and potential toxication mechanism. J. Hazard Mat. 396, 122603. doi:10.1016/j.jhazmat.2020.122603
Sökmen, T. Ö., Sulukan, E., Türkoğlu, M., Baran, A., Özkaraca, M., and Ceyhun, S. B. (2020). Polystyrene nanoplastics (20 nm) are able to bioaccumulate and cause oxidative DNA damages in the brain tissue of zebrafish embryo (Danio rerio). NeuroToxicology 77, 51–59. doi:10.1016/j.neuro.2019.12.010
Stock, V., Böhmert, L., Lisicki, E., Block, R., Cara-Carmona, J., Pack, L. K., et al. (2019). Uptake and effects of orally ingested polystyrene microplastic particles in vitro and in vivo. Arch. Toxicol. 93 (7), 1817–1833. doi:10.1007/s00204-019-02478-7
Strungaru, S. A., Jijie, R., Nicoara, M., Plavan, G., and Faggio, C. (2019). Micro-(nano) plastics in freshwater ecosystems: abundance, toxicological impact and quantification methodology. Trac. Trends Anal. Chem. 110, 116–128. doi:10.1016/j.trac.2018.10.025
Sui, A., Yao, C., Chen, Y., Li, Y., Yu, S., Qu, J., et al. (2023). Polystyrene nanoplastics inhibit StAR expression by activating HIF-1α via ERK1/2 MAPK and AKT pathways in TM3 Leydig cells and testicular tissues of mice. Food Chem. Toxicol. 173, 113634. doi:10.1016/j.fct.2023.113634
Sun, Z., Wen, Y., Zhang, F., Fu, Z., Yuan, Y., Kuang, H., et al. (2023). Exposure to nanoplastics induces mitochondrial impairment and cytomembrane destruction in Leydig cells. Ecotoxicol. Environ. Saf. 255, 114796. doi:10.1016/j.ecoenv.2023.114796
Thiagarajan, V., Alex, S. A., Seenivasan, R., Chandrasekaran, N., and Mukherjee, A. (2021). Interactive effects of micro/nanoplastics and nanomaterials/pharmaceuticals: their ecotoxicological consequences in the aquatic systems. Aquat. Toxicol. 232, 105747. doi:10.1016/j.aquatox.2021.105747
Triebskorn, R., Braunbeck, T., Grummt, T., Hanslik, L., Huppertsberg, S., Jekel, M., et al. (2019). Relevance of nano- and microplastics for freshwater ecosystems: a critical review. Trac. Trends Anal. Chem. 110, 375–392. doi:10.1016/j.trac.2018.11.023
Vaid, M., Sarma, K., and Gupta, A. (2021). Microplastic pollution in aquatic environments with special emphasis on riverine systems: current understanding and way forward. J. Environ. Manage. 293, 112860. doi:10.1016/j.jenvman.2021.112860
Varela, J. A., Bexiga, M. G., Åberg, C., Simpson, J. C., and Dawson, K. A. (2012). Quantifying size-dependent interactions between fluorescently labeled polystyrene nanoparticles and mammalian cells. J. Nanobiotechnol 10, 39. doi:10.1186/1477-3155-10-39
Vedolin, M. C., Teophilo, C. Y. S., Turra, A., and Figueira, R. C. L. (2018). Spatial variability in the concentrations of metals in beached microplastics. Mar. Pollut. Bull. 129 (2), 487–493. doi:10.1016/j.marpolbul.2017.10.019
Vega-Herrera, A., Llorca, M., Borrell-Diaz, X., Redondo-Hasselerharm, P. E., Abad, E., Villanueva, C. M., et al. (2022). Polymers of micro(nano) plastic in household tap water of the Barcelona Metropolitan Area. Water Res. 220, 118645. doi:10.1016/j.watres.2022.118645
Venâncio, C., Ferreira, I., Martins, M. A., Soares, A. M., Lopes, I., and Oliveira, M. (2019). The effects of nanoplastics on marine plankton: a case study with polymethylmethacrylate. Ecotoxicol. Environ. Saf. 184, 109632. doi:10.1016/j.ecoenv.2019.109632
Venancio, C., Savuca, A., Oliveira, M., Martins, M. A., and Lopes, I. (2021). Polymethylmethacrylate nanoplastics effects on the freshwater cnidarian Hydra viridissima. J. Hazard Mat. 402, 123773. doi:10.1016/j.jhazmat.2020.123773
Walczak, A. P., Kramer, E., Hendriksen, P. J. M., Tromp, P., Helsper, J. P. F., van der Zande, M., et al. (2015). Translocation of differently sized and charged polystyrene nanoparticles in in vitro intestinal cell models of increasing complexity. Nanotoxicology 9, 453–461. doi:10.3109/17435390.2014.944599
Wang, F., Wang, B., Qu, H., Zhao, W., Duan, L., Zhang, Y., et al. (2020a). The influence of nanoplastics on the toxic effects, bioaccumulation, biodegradation and enantioselectivity of ibuprofen in freshwater algae chlorella pyrenoidosa. Environ. Pollut. 263, 114593. doi:10.1016/j.envpol.2020.114593
Wang, T., Zou, X., Li, B., Yao, Y., Li, J., Hui, H., et al. (2018). Microplastics in a wind farm area: a case study at the rudong offshore wind farm, yellow sea, China. Mar. Pollut. Bull. 128, 466–474. doi:10.1016/j.marpolbul.2018.01.050
Wang, W., Ge, J., and Yu, X. (2020b). Bioavailability and toxicity of microplastics to fish species: a review. Ecotoxicol. Environ. Saf. 189, 109913. doi:10.1016/j.ecoenv.2019.109913
Wang, Y., Mao, Z., Zhang, M., Ding, G., Sun, J., Du, M., et al. (2019). The uptake and elimination of polystyrene microplastics by the brine shrimp, Artemia parthenogenetica, and its impact on its feeding behavior and intestinal histology. Chemosphere 234, 123–131. doi:10.1016/j.chemosphere.2019.05.267
Wu, J., Jiang, R., Lin, W., and Ouyang, G. (2019). Effect of salinity and humic acid on the aggregation and toxicity of polystyrene nanoplastics with different functional groups and charges. Environ. Pollut. 245, 836–843. doi:10.1016/j.envpol.2018.11.055
Xia, T., Kovochich, M., Liong, M., Zink, J. I., and Nel, A. E. (2008). Cationic polystyrene nanosphere toxicity depends on cell-specific endocytic and mitochondrial injury pathways. ACS Nano 2, 85–96. doi:10.1021/nn700256c
Xie, X., Deng, T., Duan, J., Xie, J., Yuan, J., and Chen, M. (2020). Exposure to polystyrene microplastics causes reproductive toxicity through oxidative stress and activation of the p38 MAPK signaling pathway. Ecotoxicol. Environ. Saf. 190, 110133. doi:10.1016/j.ecoenv.2019.110133
Yang, Y. F., Chen, C. Y., Lu, T. H., and Liao, C. M. (2019). Toxicity-based toxicokinetic/toxicodynamic assessment for bioaccumulation of polystyrene microplastics in mice. J. Hazard Mater 366, 703–713. doi:10.1016/j.jhazmat.2018.12.048
Yee, M. S. L., Hii, L. W., Looi, C. K., Lim, W. M., Wong, S. F., Kok, Y. Y., et al. (2021). Impact of microplastics and nanoplastics on human health. Nanomaterials 11 (2), 496. doi:10.3390/nano11020496
Yin, L., Liu, H., Cui, H., Chen, B., Li, L., and Wu, F. (2019). Impacts of polystyrene microplastics on the behavior and metabolism in a marine demersal teleost, black rockfish (Sebastes schlegelii). J. Hazard. Mat. 380, 120861. doi:10.1016/j.jhazmat.2019.120861
Yu, F., Yang, C., Zhu, Z., Bai, X., and Ma, J. (2019). Adsorption behavior of organic pollutants and metals on micro/nanoplastics in the aquatic environment. Sci. Total Environ. 694, 133643. doi:10.1016/j.scitotenv.2019.133643
Zagorski, J., Debelak, J., Gellar, M., Watts, J. A., and Kline, J. A. (2003). Chemokines accumulate in the lungs of rats with severe pulmonary embolism induced by polystyrene microspheres. J. Immunol. 171 (10), 5529–5536. doi:10.4049/jimmunol.171.10.5529
Zalasiewicz, J., Waters, C. N., Ivar do Sul, J. A., Corcoran, P. L., Barnosky, A. D., Cearreta, A., et al. (2016). The geological cycle of plastics and their use as a stratigraphic indicator of the Anthropocene. Anthropocene 13, 4–17. doi:10.1016/j.ancene.2016.01.002
Zhang, F., Wang, Z., Song, L., Fang, H., and Wang, D. G. (2020a). Aquatic toxicity of iron-oxide-doped microplastics to Chlorella pyrenoidosa and Daphnia magna. Environ. Pollut. 257, 113451. doi:10.1016/j.envpol.2019.113451
Zhang, F., Wang, Z., Wang, S., Fang, H., and Wang, D. (2019). Aquatic behavior and toxicity of polystyrene nanoplastic particles with different functional groups: complex roles of pH, dissolved organic carbon and divalent cations. Chemosphere 228, 195–203. doi:10.1016/j.chemosphere.2019.04.115
Zhang, Y., Pu, S., Lv, X., Gao, Y., and Ge, L. (2020). Global trends and prospects in microplastics research: a bibliometric analysis. J. Hazard. Mat. 400, 123110. doi:10.1016/j.jhazmat.2020.123110
Zheng, H., Wang, J., Wei, X., Chang, L., and Liu, S. (2021). Proinflammatory properties and lipid disturbance of polystyrene microplastics in the livers of mice with acute colitis. Sci. Total Environ. 750, 143085. doi:10.1016/j.scitotenv.2020.143085
Zhou, X. X., Liu, R., Hao, L. T., and Liu, J. F. (2021). Identification of polystyrene nanoplastics using surface enhanced Raman spectroscopy. Talanta 221, 121552. doi:10.1016/j.talanta.2020.121552
Zhu, X., Zhao, W., Chen, X., Zhao, T., Tan, L., and Wang, J. (2020). Growth inhibition of the microalgae Skeletonema costatum under copper nanoparticles with microplastic exposure. Mar. Environ. Res. 158, 105005. doi:10.1016/j.marenvres.2020.105005
Zhu, Z. L., Wang, S. C., Zhao, F. F., Wang, S. G., Liu, F. F., and Liu, G. Z. (2019). Joint toxicity of microplastics with triclosan to marine microalgae Skeletonema costatum. Environ. Pollut. 246, 509–517. doi:10.1016/j.envpol.2018.12.044
Zitouni, N., Bousserrhine, N., Belbekhouche, S., Missawi, O., Alphonse, V., Boughatass, I., et al. (2020). First report on the presence of small microplastics (≤3 μm) in tissue of the commercial fish Serranus scriba (Linnaeus, 1758) from Tunisian Coasts and associated cellular alterations. Environ. Pollut. 263, 114576. doi:10.1016/j.envpol.2020.114576
Zubair, M., Ahmad, M., Saleemi, M. K., Gul, S. T., Ahmad, M., Martyniuk, C. J., et al. (2020). Sodium arsenite toxicity on hematology indices and reproductive parameters in teddy goat bucks and their amelioration with vitamin C. Environ. Sci. Pollut. Res. 27, 15223–15232. doi:10.1007/s11356-020-08049-z
Keywords: plastics, marine ecosystems, pollution, aquatic organism, public health, toxicity
Citation: Naz S, Chatha AMM, Khan NA, Ullah Q, Zaman F, Qadeer A, Khan IM, Danabas D, Kiran A, Skalickova S, Bernatova S, Khan MZ and Horky P (2024) Unraveling the ecotoxicological effects of micro and nano-plastics on aquatic organisms and human health. Front. Environ. Sci. 12:1390510. doi: 10.3389/fenvs.2024.1390510
Received: 23 February 2024; Accepted: 19 March 2024;
Published: 08 April 2024.
Edited by:
Divya Pal, Stockholm University, SwedenReviewed by:
Isha Burman, Indian Institute of Technology Dhanbad, IndiaAndrey E. Krauklis, University of Latvia, Latvia
Copyright © 2024 Naz, Chatha, Khan, Ullah, Zaman, Qadeer, Khan, Danabas, Kiran, Skalickova, Bernatova, Khan and Horky. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Qudrat Ullah, cXVkcmF0LnVsbGFoQHVhZC5lZHUucGs=; Pavel Horky, cGF2ZWwuaG9ya3lAbWVuZGVsdS5jeg==
 Saima Naz
Saima Naz Ahmad Manan Mustafa Chatha2
Ahmad Manan Mustafa Chatha2 Qudrat Ullah
Qudrat Ullah Abdul Qadeer
Abdul Qadeer Ibrar Muhammad Khan
Ibrar Muhammad Khan Azka Kiran
Azka Kiran Sylvie Skalickova
Sylvie Skalickova Silvie Bernatova
Silvie Bernatova Muhammad Zahoor Khan
Muhammad Zahoor Khan Pavel Horky
Pavel Horky