- 1Department of Agricultural Sciences, Biotechnology and Food Science, Cyprus University of Technology, Lemesos, Cyprus
- 2Hellenic Agricultural Organization ‘Demeter’, Institute of Plant Breeding and Genetic Resources, Thessaloniki, Greece
- 3Hellenic Agricultural Organization ‘Demeter’, Department of Deciduous Fruit Trees, Institute of Plant Breeding and Genetic Resources, Naoussa, Greece
Horticultural commodities (fruit and vegetables) are the major dietary source of several bioactive compounds of high nutraceutical value for humans, including polyphenols, carotenoids and vitamins. The aim of the current review was dual. Firstly, toward the eventual enhancement of horticultural crops with bio-functional compounds, the natural genetic variation in antioxidants found in different species and cultivars/genotypes is underlined. Notably, some landraces and/or traditional cultivars have been characterized by substantially higher phytochemical content, i.e., small tomato of Santorini island (cv. “Tomataki Santorinis”) possesses appreciably high amounts of ascorbic acid (AsA). The systematic screening of key bioactive compounds in a wide range of germplasm for the identification of promising genotypes and the restoration of key gene fractions from wild species and landraces may help in reducing the loss of agro-biodiversity, creating a healthier “gene pool” as the basis of future adaptation. Toward this direction, large scale comparative studies in different cultivars/genotypes of a given species provide useful insights about the ones of higher nutritional value. Secondly, the advancements in the employment of analytical techniques to determine the antioxidant potential through a convenient, easy and fast way are outlined. Such analytical techniques include electron paramagnetic resonance (EPR) and infrared (IR) spectroscopy, electrochemical, and chemometric methods, flow injection analysis (FIA), optical sensors, and high resolution screening (HRS). Taking into consideration that fruits and vegetables are complex mixtures of water- and lipid-soluble antioxidants, the exploitation of chemometrics to develop “omics” platforms (i.e., metabolomics, foodomics) is a promising tool for researchers to decode and/or predict antioxidant activity of fresh produce. For industry, the use of optical sensors and IR spectroscopy is recommended to estimate the antioxidant activity rapidly and at low cost, although legislation does not allow its correlation with health claims.
Introduction
The increased dietary intake of fresh produce has been a fundamental element of public health policy for the past years, as has been long considered to provide protection against a wide range of oxidative-stress related diseases, such as cancer, stroke, diabetes, Alzheimer's, cataract, and age-related function decline (Arts and Hollman, 2005). All recent studies support that a diet rich in fruit and vegetables is beneficial for human health, suggesting that these key bioactive compounds should be a clear target for enhancement of nutritional value of fresh produce.
Low amounts of reactive oxygen species (ROS) are continuously generated in cells of aerobic organisms (Kitsati et al., 2012), and play, as unequivocally accepted, important physiological roles. However, when ROS levels increase, an unregulated oxidation of cell components may be induced, a fact that is directly linked with the initiation and/or progression of an array of pathological conditions. Such conditions can be combated or delayed through the consumption of exogenous protective compounds (Kitsati et al., 2012). Epidemiological studies also support that the consumption of fresh produce, containing phenolic and non-phenolic antioxidants is inversely associated with risk of chronic diseases (Naska and Trichopoulou, 2014). In this context, the antioxidant potency per se is a well-established biomarker, which indicates beneficial biological effects of fresh produce.
In the sections below, the significance of exploiting the natural genetic variation, including landraces, traditional and commercial cultivars, for the enhancement of horticultural crops with bio-functional compounds is discussed. Considering the vast amount of genotypes for a given horticultural commodity and their different behavior under diverse ecological environments, there is an urgent need to screen and precisely evaluate their antioxidant capacity and overall phytochemical content in a compatible manner with up-to-date analytical approaches. Toward this aim and in view of the fact that a great number of methods for valorizing antioxidant capacity has been proposed over the recent years, this review focuses in techniques that can fulfill the requirements of horticulturists and food scientists for rapid, easy and low cost analytical tools.
Exploitation of Genotype Variation to Enhance Bio-functional Compounds
A wide diversity in the concentrations of key bioactive compounds (especially ascorbic acid (AsA), phenypropanoids and carotenoids) among cultivars within the same species has been recorded (reviewed in Kanellis and Manganaris, 2014). It is therefore conceivable that screening studies on genotypic diversity are of prime importance, especially in horticultural crops where the production is derived from a significant number of cultivars, i.e., peach, sweet cherry, apple. Although factors, such as environmental conditions (Dumas et al., 2003), developmental/ripening stage (Hancock et al., 2007; Ioannidi et al., 2009; Drogoudi and Pantelidis, 2011; Ilahy et al., 2011; Liu et al., 2015), tissue type (Drogoudi et al., 2007; Bulley et al., 2009), and pre- and post-harvest treatments (Ioannidi et al., 2009) play a pivotal role in regulating the accumulation of bioactive molecules in fresh commodities, genetic control appears to be by far the dominant source of variation within a species. As traits linked to nutritional quality are usually under polygenic control and quantitative inheritance, understanding the existing natural diversity, including indigenous and/or traditional cultivars at the genetic level is a great challenge in modern breeding programs. Early domestication and modern breeding strategies has led to the loss of certain genome regions linked to stress resistance and/or nutritional value, usually in favor of crop yield (Fernie et al., 2006). Ascorbic acid, polyphenols and carotenoids constitutes main antioxidant classes and are analyzed in detail in the following sections.
Ascorbic Acid
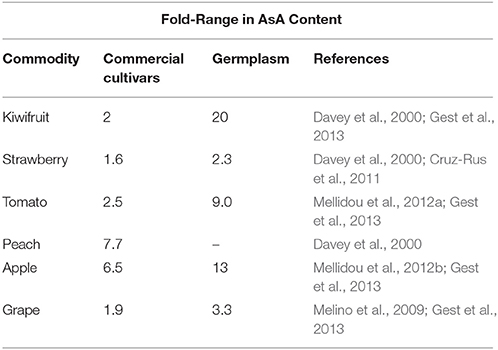
Horticultural commodities, such as pepper, strawberry, kiwifruit and citrus possess significant amounts of AsA, whereas other popular in human diet species, such as tomato and apple contain moderate amounts. The accumulation of AsA may vary not only within different species, but also among different cultivars (Bulley et al., 2009; Mellidou et al., 2012a,b; Gest et al., 2013), tissue types, with photosynthetically-active tissues containing higher levels than heterotrophic ones (Bulley et al., 2009) and developmental stage (Hancock et al., 2007; Bulley et al., 2009; Ioannidi et al., 2009; Mellidou et al., 2012b) within the same species. Indicatively, AsA content of edible parts of commercial cultivars may substantially vary both within commercial cultivars and germplasm (Table 1). Intra-variety diversity in AsA content due to population × year interaction or due to the different sampling locations may also be critical in several fruit species, such as tomato (Cortés-Olmos et al., 2014) and apple (Pissard et al., 2012), pointing out the urgent need to develop robust protocols to analyze fruit quality.
An excellent example to explore genotype variation and to elucidate how the differences in AsA metabolic pathways and the genes involved therein govern the AsA pool is the landrace “Santorini” a drought-tolerant tomato cultivar with high fruit AsA content, indigenous to Santorini island and protected designation of origin (PDO) product in Greece (Mellidou et al., 2012b; Koutsika-Sotiriou et al., 2016). Integration of metabolite analyses, non-labeled, and radio-labeled substrate feeding experiments, enzyme activity and transcript level profiling showed that enhanced AsA recycling activities were responsible for AsA accumulation during the last stages of ripening (Mellidou et al., 2012b).
Apart from differences between commercial cultivars, an interesting observation is that AsA concentrations tend to be higher in wild accessions compared to modern cultivars (Table 1). For example, within tomato germplasm, Solanum pennellii, and S. lycopersicum cv cerasiforme contain 5- and 2-fold more AsA, respectively, than the traditional cultivars of S. lycopersicum (Stevens et al., 2007), while natural diversity may reach up to 9-fold (Gest et al., 2013). Presumably, this is due to the fact that during plant evolution, traits, such as fruit size and total plant yield have been selected at the cost of other parameters, such as vitamin C, as modern crops are usually cultivated in favorable environments at the absence of stress conditions. Indeed, comparative studies between S. lycopersicum and S. pennellii, revealed that the wild accession responds much more efficiently to low-water conditions (Stevens et al., 2008). Cherry tomatoes are considered as intermediate genotypes both genetically and phenotypically between wild and cultivated tomatoes, and are richer in AsA than their large fruited counterparts. Additionally to the dilution effect that leads the decrease of AsA pool size, there seems to be a clear genetic linkage between fruit size and AsA concentrations, as revealed by the collocation of major Quantitative Trait Locus (QTL) for AsA contents with QTLs for fruit weight in both apple (Mellidou et al., 2012a) and tomato (Stevens et al., 2007). Whether domestication, that systematically favored growth at the expense of accumulating other necessary metabolites, has also influenced AsA metabolic pathways at the protein (structure) or gene (expression patterns) or gene cellular compartmentation level (Gest et al., 2013), remains an open question that merits further investigation.
Such differences within a species may be primarily attributed to higher rates of net biosynthesis, recycling, and/or intercellular and intracellular transport, or decreased turnover capacity, and feedback regulation of AsA pool through inhibition of the activity of the last enzyme in the biosynthetic pathway. Although numerous studies on genetic factors demonstrated that AsA accumulation showed a relatively high heritability, it also seems to be highly responsive to environmental stimuli and post-harvest treatments (Mellidou and Kanellis, 2017). The pathway derived from L-galactose is generally considered as the dominant route of AsA biosynthesis in Arabidopsis (Bulley et al., 2009; Yoshimura et al., 2014), tomato (Gilbert et al., 2009; Ioannidi et al., 2009; Bulley et al., 2012; Mellidou et al., 2012b; Wang et al., 2013), kiwifruit (Bulley et al., 2009; Li et al., 2010), black currant (Hancock et al., 2007), citrus (Alós et al., 2014), blueberry (Liu et al., 2015), and potato tuber (Bulley et al., 2012).
Several lines of evidence suggest GDP-L-galactose phosphorylase (GGP), the enzyme catalyzing the first committed step of the pathway is the key control point in the AsA biosynthetic pathway, and its transcription has been correlated with the AsA pool size in several species (Bulley and Laing, 2016; Mellidou and Kanellis, 2017). Although the transcriptional control of GGP in regulating AsA pool size is well-established, it is only recently that its translational regulation under unfavorable conditions has been also elucidated (Laing et al., 2015). The proposed model is based on the identification of a highly conserved non-canonical upstream open reading frame (uORF) in a wide range of species that allows the feedback regulation of GGP translation under rapidly changing conditions, without the need of gene transcription modifications. Allelic associations studies in a mapping population and 22 commercial cultivars of apple reinforce the notion that SNPs found in GGP coding sequence are rather linked to polymorphisms in the promoter region that alter allele expression, than to altered protein function (Mellidou et al., 2012a).
The genetic regulation of AsA accumulation may also vary depending on the tissue or developmental stage considered. Toward this end, in red ripe tomato fruit, the alternative routes proceeding either via D-galacturonate (Badejo et al., 2012) or via myo-inositol (Mellidou et al., 2012b) may also contribute to an enhanced AsA pool size in certain genotypes, presumably as a tool to support biosynthesis via the main route under unfavorable conditions. Furthermore, the alternative biosynthetic pathway via uronic acids has been suggested to exert a major role in regulating fruit AsA content in strawberry (Agius et al., 2003; Cruz-Rus et al., 2011), citrus (Xu et al., 2012), and rose (Li et al., 2017). Apart from changes in AsA biosynthetic capacity, perturbations in the AsA recycling pathway by altering AsA redox homeostasis may also alter not only AsA concentrations, but also plant responses to various environmental stimuli (Foyer and Noctor, 2011; Fotopoulos and Kanellis, 2013). Although a plethora of classical and modern breeding strategies have been employed to unravel the genetic mechanisms underlying AsA accumulation, a lot of effort is still required to restore its levels to cultivated crops toward producing high-quality and sustainable horticultural commodities for future generations.
Polyphenols
Polyphenols are the most abundant bioactive compounds with antioxidant activity, and consist of a wide range of biomolecules, such as simple phenols, phenolic acids, flavonoids, polymeric phenolics, and anthocyanins (Kanellis and Manganaris, 2014). Based on the structure of their basic skeleton, the main phenolic compounds are phenolic acids, flavonoids and anthocyanins (Lima et al., 2014). Phenol-explore database summarizes more than 35,000 content values for 500 different polyphenols in over 400 foods (http://phenol-explorer.eu/). Anthocyanins are vacuolar pigments usually present in the skin of fruits responsible for fruit color in several species (e.g., berries, grape, cherry, pomegranate, plum, apple) giving their characteristic red, blue or purple pigmentation. Other compounds of this category that are less studied in horticultural crops are lignans, stilbenes, tannins, coumarins, and lignins. In line with findings on AsA, landraces and unexplored cultivars accumulate such compounds at high levels providing a valuable tool toward sustainable breeding for the development of nutritionally-enriched cultivars. On the other hand, high concentrations of TPs may result to unpalatable bitter or astringent tastes (Lesschaeve and Noble, 2005) that may or not be related to browning coloration in fruit and vegetables (Cantín et al., 2011; Saeed et al., 2014). In particular, soft fruits (strawberry, blackberry, blueberry, raspberry) are generally characterized by appreciably high antioxidant potential but such fruit cannot be widely available due to short shelf-life or consumption is limited due to undesirable organoleptic characteristics being bitter, sour or astringent (i.e., aronia, cornelian cherry, or hyppophaes) (reviewed in Manganaris et al., 2014).
Apple (Malus × domestica) is the most important temperate fruit crop. Notably, high genetic variability in polyphenolic compounds within apple germplasm exist. Comparing wild germplasm with commercial cultivars, total polyphenolic content was found to vary significantly within Malus sieversii and Malus × domestica germplasm (up to 9-fold in the flesh, and up to 4- in the peel), with the wild genotypes of Malus sieversii generally containing higher concentrations in individual polyphenol groups (Volz and McGhie, 2011). Breeding and germplasm conservation strategies aim to improve polyphenolic for enhancing quality traits related to phenylpropanoid pathway (color, aroma, disease resistance, and browning disorders) of elite modern cultivars (Wojdyło et al., 2008).
The availability of the draft apple genome sequence (Velasco et al., 2010) caused a boost in apple genetics providing new tools for candidate gene detection. Over 50 metabolites, peel- and flesh-related, from the phenylpropanoid pathway were found to collocate with a certain QTL hotspot in linkage group 16, that contains a structural gene of the pathway, leucoanthocyanidin reductase (LAR), indicating that this gene may play a pivotal role in regulating the pathway (Khan et al., 2012). A feedback regulation of proanthocyanidin (PA) biosynthesis has been proposed, according to which low PA levels may enhance the transcription of structural biosynthetic genes of the pathways, such as LAR (Henry-Kirk et al., 2012).
Notably, over the last decade, red pigmentation of apples has received considerable attention and new red-fleshed apple cultivars have been released into the market, being considered of higher nutritional value (high anthocyanin levels). Cyanidin 3-O-galactoside is the dominant anthocyanin in both flesh and skin (Volz et al., 2014). Based on recent advances, anthocyanin accumulation can be regulated by three main mechanisms: (1) the anthocyanin biosynthetic pathway, (2) the transcription regulation through the MYB protein that affects anthocyanin biosynthesis during fruit growth and development, and (3) the anthocyanin transport from cytosol where it is synthesized to the vacuole where it is stored (Espley et al., 2009; Hu et al., 2016). Stable QTLs for polyphenolic content have been identified in apples and SNP-derived markers have been developed for polyphenolic-rich cultivars (Chagné et al., 2012, 2016). Using a transgenic/cisgenic approach, the integration of the dominant red-flesh MYB allele of the transcription factor MYB10 resulted in apples with significantly higher levels of foliar, flower and fruit (especially in the peel) anthocyanins in the transgenic red-fleshed apples (Espley et al., 2013).
Except for apple, a significant diversity in phenolic compounds among different genotypes of commodities of high nutritional value, such as soft fruits (Stephens et al., 2009; Stevenson and Scalzo, 2012) and pomegranates has been recorded. “Wonderful” and “Hicaznar” global pomegranate cultivars possessed higher total phenolic concentrations compared to local cultivars, grown in Spain (Legua et al., 2016). This is the case also for other commercially important fruit commodities, as apricot (Sochor et al., 2011), and peach (Kwon et al., 2015). Notably, the reference nectarine cultivar “Big Top,” that is highly appreciated by the consumers due to exceptional qualitative attributes, was characterized by low antioxidant capacity (Drogoudi et al., 2016). Late-harvested compared to early- harvested cultivars, tended to be characterized by higher polyphenolic contents (Font i Forcada et al., 2014; Drogoudi et al., 2016).
Carotenoids
Carotenoids are lipophilic molecules responsible for the yellow, orange and red pigmentation of several fruit. There are two main groups of carotenoids, one being carotenes [α-carotene, β-carotene (the precursor of vitamin A) and lycopene the most powerful antioxidant among this group], the other being oxygenated derivatives known as xanthophylls (lutein, cryptoxanthin, violaxnthin, and zeaxanthin). Generally, carrots, sweet potato, pineapple, mango, peach, and tomato are rich in carotenoids. The latter, taking additionally into account its increased consumption, is considered as a valuable source of lycopene and β-carotene in the human diet (Fraser and Bramley, 2004). Furthermore, tomato is widely used as a model species for carotenoid studies due to its diverse fruit pigmentation (yellow, orange, orange-red, and red) with different carotenoid profiles (Yuan et al., 2015). For instance, red tomatoes are rich in lycopene, while orange fruits of Beta mutants contain high β-carotene levels but at the expense of lycopene. Similar to findings on AsA, carotenoids are highly accumulated in wild species, such as S. pennellii compared to their domesticated relatives (Fernie et al., 2006). A dominant allele from wild species has been found to remarkably increase β-carotene in the fruit via higher β-cyclase activity (Ronen et al., 2000). Several studies on tomato natural mutants and transgenic plants with altered carotenoid accumulation allowed to decipher the complete carotenoid biosynthetic pathway in tomato (reviewed in Martí et al., 2016).
As far as other horticultural crops are concerned, Citrus spp. have the greater variation in carotenoid contents among different species and cultivars. For example orange, mandarin and clementine fruit contain high amounts of β-cryptoxanthin, violaxanthin, lutein and zeaxanthin, while lemon, and lime are relatively poor in carotenoids (Yuan et al., 2015). A wide diversity (up to 25-fold) between commercial cultivars and/or accessions has also been reported in Musa spp. (Davey et al., 2009; Borges et al., 2014). The commercially important bananas of the Cavendish group do not contain high levels of carotenoids, whilst other genotypes identified in banana germplasm contain significant amounts of these compounds (Amorim et al., 2009; Davey et al., 2009). Although factors, such as the degree of maturation, the environment, the type of soil, and storage conditions may be important in regulating carotenoids contents in banana, the most significant factors appears to be the genotype and the genetic origin (Davey et al., 2007; Amorim et al., 2009). Generally, genotypes from the genomic group AAB are rich in carotenoid levels compared to those from the AAA group (i.e., Cavendish), probably due to the presence of the B genome (Davey et al., 2007), indicating the potential of carotenoid compounds to be successfully bio-fortified in breeding programs.
Multidisciplinary research programs currently focus on bio-fortification of carotenoids in fruits and vegetables, mainly targeting on β-carotene, in an effort to tackle vitamin A deficiency (Farré et al., 2011). Therefore, understanding the genetic mechanisms underlying carotenoid accumulation responsible for the genetic diversity across and within species is a great challenge. Carotenoids and especially those having pro-vitamin A activity are quantitative traits, and their contents are regulated by the activity of multiple gene products (Davey et al., 2009). Carotenoid biosynthesis is now well-established with most of the genes being characterized in many species (Lu and Li, 2008; Cazzonelli and Pogson, 2010; Martí et al., 2016). Overall, precursor substrate availability (isopentenyl diphosphate and dimethylallyl-diphosphate), as well as the accumulation of phytoene via phytoene synthase (PSY) and the phytoene desaturase, are considered as key limiting factors in regulating carotenoid contents in various species (Rodríguez-Villalón et al., 2009; Ravel et al., 2013; Jourdan et al., 2015). Increasing the expression of PSY and lycopene β-cyclase (LYCB) in tomato has led to significant enhancement of the level of β-carotene and lycopene, respectively (Fraser et al., 2002; D'Ambrosio et al., 2004). Interestingly, when LYCB is overexpressed, the levels of β-carotene are also increased as seen in Golden Rice (Ye et al., 2000), due to the shift of the metabolic balance from the α to the β branch of carotenoid pathway at the expense of α-carotene and lutein (D'Ambrosio et al., 2004; Farré et al., 2011). In carrot, polymorphism associated with zeaxanthin epoxidase (ZEP) was associated with carotenoid and phytoene contents, indicating that this is a key gene governing carotenoid accumulation in carrot roots. Toward this end, the high-pigment 3 tomato mutants (mutation occurring on ZEP gene) had 30% more carotenoids in mature fruit (Galpaz et al., 2008). There is also compelling evidence that several regulatory genes and proteins as well as a large number of transcription factors affect the hormonal control of carotenoid accumulation at diverse levels through regulation of fruit ripening. In this regard, tomato mutants, such as rin and nor deficient in ethylene synthesis or perception not only show delayed normal ripening but also modified carotenoid content (Martí et al., 2016). Consistently, transcript levels of PSY appeared to be regulated by ethylene, whilst the ethylene-response transcription factor ERF6 plays a vital role in carotenoid accumulation in tomato fruit (Lee et al., 2012).
Advanced Analytical Techniques to Determine Antioxidant Potential
Bond dissociation energy and ionization potential are two major factors that determine the mechanism and the efficiency of antioxidants (Karadag et al., 2009). Thus, hydrogen atom transfer (HAT) and electron transfer (ET) reactions are the chemical principles of most widely employed antioxidant activity assays. Furthermore, antioxidant activity analyses can be also classified into redox potential and reactive species scavenging assays. In this context, a great number of simple spectrophotometric tests have been developed. The most commonly applied are 1,1-diphenyl-2-picryl-hydrazyl (DPPH) assay, ferric reducing/antioxidant power (FRAP), ABTS radical cation decolorization, oxygen radical absorbance capacity (ORAC), phosphomolybdenum, cupric ion reducing antioxidant capacity (CUPRAC) and superoxide radical scavenging activity. Due to great importance of antioxidants for human health, a panel of methods to determine the antioxidant activity of fresh produce in biological systems has been also applied, such as inhibition of LDL-cholesterol and of DNA oxidation as well as DNA nicking, haemolysis inhibition and cellular antioxidant capacity (Shahidi and Zhong, 2015).
Taking into consideration that fruit and vegetables are complex mixtures of natural antioxidants, many analytical techniques has been developed to assess their antioxidant activity. In particular, spectroscopy, and electrochemistry are widely used for the determination of antioxidant activity of horticultural produce. Further, flow injection analysis (FIA), optical sensors and high resolution screenings (HRS) allow the rapid estimation of antioxidant activity; whereas chemometrics predict the antioxidant activity extracting data from classical analytical chemistry. All these approaches pursue to reduce time of analysis and the use of harmful reagents and to automate the determinations. The main drawback of these methods as for simple colorimetric assays is the need to prepare extracts in order to measure antioxidant activity. Table 2 summarizes the aforementioned analytical methods, providing their application potential for the determination of antioxidant activity in fresh produce.
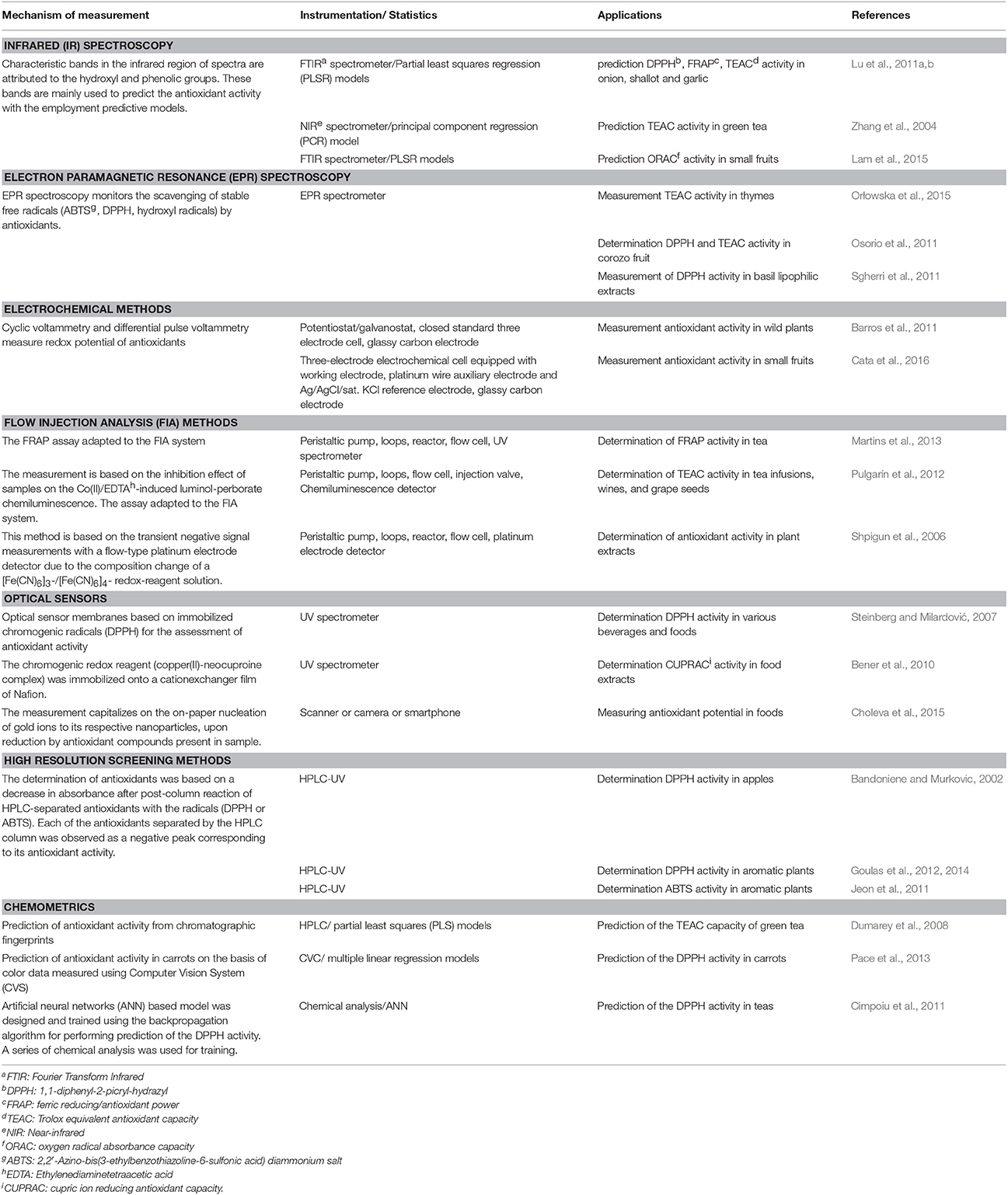
Table 2. A brief overview of analytical methods for the determination of antioxidant activity in fresh produce.
Infrared (IR) Spectroscopy
Infrared (IR) spectroscopy is widely used for analysis, quality control and authentication in food research and industry. In general, it is a fast, accurate, easy and non-destructive technique that can potentially replace the classical chemical analyses. For quantitative purposes, a reliable calibration model needs to be developed (Zhang et al., 2004), followed by a partial least-squares regression calculations to predict the antioxidant activity of a given plant material; while the use of multiple linear regression, artificial neural network, and least squares support vector machine can be also applied (Lu and Rasco, 2012; Wu et al., 2012). Regarding band assignments in the IR region of spectra, hydroxyl and phenolic functional groups were most closely correlated with antioxidant capacity. Further, a close correlation exists between IR estimated values and in vitro assays, such as DPPH, FRAP, ABTS, and ORAC that has been applied to predict antioxidant activity in fruit (Lam et al., 2015), onion, shallot, garlic (Lu et al., 2011a,b) and medicinal plants (Bunaciu et al., 2012).
Electron Paramagnetic Resonance (EPR) Spectroscopy
Electron paramagnetic resonance (EPR) spectroscopy is the only analytical technique that can directly detect and quantify free radicals generated by ex vivo and in vivo chemical reactions. An EPR spectrum is the first derivative of the absorbance peaks when chemical species having unpaired electrons absorb radiation into the microwave range in a magnetic field, and the integration of the area under curve correlates with the number of radicals. Thus, the time-dependent scavenging of free radicals by the antioxidants can be monitored (Osorio et al., 2011). The antioxidant status of fresh produce can be effectively monitored through the elimination of stable free radicals (i.e., ABTS∙+, DPPH∙, or hydroxyl radicals (OH∙) added to the sample. Among the main advantages of EPR are a) the high sensitivity, allowing the detection at the submicromolar level, and b) the possibility to analyze turbid or highly colored samples (Sgherri et al., 2011). Opposite to colorimetric assays, EPR is more specific due to the formation of characteristic signals (Osorio et al., 2011).
Nowadays, EPR has been successfully applied to assess the radical scavenging activity of different foodstuffs rich in phenolic compounds. It can be also used to study the ability of hydrophilic as well as lipophilic antioxidants to scavenge free radicals. In particular, such applications of EPR have been described for fruit (blueberry, apple, corozo), aromatic plants (thyme, basil), tea and coffee samples (Morsy and Khaled, 2002; Pérez-Martínez et al., 2010; Osorio et al., 2011; Sgherri et al., 2011; Mirosavljević et al., 2015; Orłowska et al., 2015).
Electrochemical Methods
Electrochemical properties of pure antioxidants and/or plant foods can be exploited for the evaluation of their antioxidant activity. Electrochemical methods are based on the direct relation between expected antioxidant activity and electric oxidation potential. Among the electrochemical methods, the cyclic voltammetry (CV) technique has been adapted to determine the antioxidant activity (reducing capacity) of low molecular weight antioxidants (Magalhães et al., 2008). CV tracings are usually recorded from − 0.5 to + 1.5 V vs. the reference electrode at a scan rate of 100 or 400 mV s−1 (Chevion et al., 2000).
CV techniques allow rapid screening of the antioxidant activity of complex mixtures as foods, fruit and vegetables. In addition, the determination of antioxidant activity in aqueous medium as well as in organic solvents is feasible. Therefore, it is a useful and powerful tool to detect water and lipid soluble antioxidants (Chevion et al., 2000). Moreover, turbid or intensely colored samples can be measured without prior sample preparation, such as discoloration, centrifugation etc (Magalhães et al., 2008). In the last decade, numerous studies described the application of CV techniques to detect the antioxidant activity in horticultural commodities, such as herbs (Cosio et al., 2006), wild plants (Barros et al., 2011), berries (Cata et al., 2016), mushrooms (Barros et al., 2008), and onions (Sun-Waterhouse et al., 2008).
Flow Injection Analysis (FIA) Methods
Researchers also pay attention to FIA systems as these convert classic antioxidant assays to automated techniques. Their advantages are the ease of operation, the potential of miniaturization, the low consumption of the sample and reagents, the versatility and the high analytical frequency (Martins et al., 2013). FIA involves the rapid injection of a sample into a non-segmented, continuous carrier stream. Regarding instrumentation, the simplest set-up is a single line and merging zones that can be controlled manually, whereas the most advanced ones involve a multi-syringe configuration and/or computer-controlled sequential injection (Magalhães et al., 2007, 2009).
In vitro assays (i.e., ABTS, DPPH, and FRAP) and chemiluminescence have been adopted to FIA systems. The literature includes several interesting FIA systems to monitor the antioxidant activity in plant extracts, pure antioxidants and agricultural products like wine and honey (Shpigun et al., 2006; Blasco et al., 2007; Alvarez-Suarez et al., 2010; Pulgarín et al., 2012).
Optical Sensors
Being rapid, sensitive and low-cost screening applications, the optical sensors have recently become very popular (Steinberg and Milardović, 2007). They are based on the immobilization of chromogenic reagents (colorful radicals, reducing compounds and nanoparticles) onto suitable membranes, such as plasticized PVC films, Nafion membrane, paper and multilayer membranes (Çekiç et al., 2015; Choleva et al., 2015; Li et al., 2016). A number of sensors has been proposed to evaluate the intensity of coloration and to calculate thereafter the antioxidant activity in foods, including orange, apricot and peach juices, green and black tea, olive oil, coffee etc., (Steinberg and Milardović, 2007; Bener et al., 2010).
High Resolution Screening (HRS) Methods
High resolution screening (HRS) methods combine a chromatographic technique, such as HPLC or GC with a post-column (bio)chemical detection apparatus i.e., DAD, MS, and NMR. A major concern in HRS methods is to find an assay that is compatible in terms of time-scale and (bio)chemical conditions with HPLC separations (Niederländer et al., 2008). HRS methods produce a high resolution biochromatogram that can pinpoint the antioxidants of fresh produce. They are rapid, since fractionation and/or isolation procedures are omitted. Among these methods, HPLC-DPPH/ABTS assays have been successfully applied to study the antioxidant composition of apples (Bandoniene and Murkovic, 2002), aromatic plants (Goulas et al., 2012, 2014) and edible flowers (Jeon et al., 2011).
Chemometrics
These methods are suitable to predict the antioxidant activity of complex matrices usually encountered in horticultural produce. Interestingly, the coupling of chromatographic fingerprints with chemometrics was applied to predict successfully the antioxidant activity in green tea and damiana herb (Dumarey et al., 2008; Lucio-Gutiérrez et al., 2012). Artificial neural networks were also employed to predict the antioxidant activity of herbs of known chemical composition (Cabrera and Prieto, 2010; Cimpoiu et al., 2011), while Pace et al. (2013) reported the combined use of multiple regression models with computer vision systems to predict antioxidant activity in carrots.
Conclusions
The promotion of selected cultivars of high antioxidant potential for a given species and directing breeding strategies toward the development of high nutritional value cultivars is expected to increase the consumption of horticultural commodities. It should be also taken into consideration the fact that the massive use of commercial cultivars with improved yield attributes has possibly resulted in the loss of some quality traits, including antioxidant compounds, found in landraces and unexplored traditional cultivars with local interest. Moreover, marketing strategies should also give an extra value to growers by directing the health-concerned consumers to consume products of high antioxidant content.
The evaluation of antioxidant properties of fresh produce is usually performed by simple spectrophotometric assays. Furthermore, advanced analytical methods based on IR and EPR spectroscopy and electrochemistry have been developed, while HRS methods allow the determination of antioxidant activity of individual phytochemical in a complex matrix like horticultural produce. IR spectroscopy and optical sensors are recommended to estimate the antioxidant activity for industrial purposes as rapid and low cost procedures that can be automated.
Author Contributions
GAM organized and drafted this manuscript. IM, VG, and PD wrote selected parts of this review. All authors read and approved the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Publication fees were covered by the Open Access Fund of Cyprus University of Technology.
References
Agius, F., González-Lamothe, R., Caballero, J. L., Muñoz-Blanco, J., Botella, M. A., and Valpuesta, V. (2003). Engineering increased vitamin C levels in plants by overexpression of a D-galacturonic acid reductase. Nat. Biotechnol. 21, 177–181. doi: 10.1038/nbt777
Alós, E., Rodrigo, M. J., and Zacarías, L. (2014). Differential transcriptional regulation of l-ascorbic acid content in peel and pulp of citrus fruits during development and maturation. Planta 239, 1113–1128. doi: 10.1007/s00425-014-2044-z
Alvarez-Suarez, J. M., González- Paramás, A. M., Santos-Buelga, C., and Battino, M. (2010). Antioxidant characterization of native monofloral Cuban honeys. J. Agric. Food Chem. 58, 9817–9824. doi: 10.1021/jf1018164
Amorim, E. P., Vilarinhos, A. D., Cohen, K. O., Amorim, V. B., Dos Santos-Serejo, J. A., Silva, S. O. E., et al. (2009). Genetic diversity of carotenoid-rich bananas evaluated by diversity arrays technology (DArT). Genet. Mol. Biol. 32, 96–103. doi: 10.1590/S1415-47572009005000024
Arts, I. C. W., and Hollman, P. C. H. (2005). Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 81, 317S–325S.
Badejo, A. A., Wada, K., Gao, Y., Maruta, T., Sawa, Y., Shigeoka, S., et al. (2012). Translocation and the alternative D-galacturonate pathway contribute to increasing the ascorbate level in ripening tomato fruits together with the D-mannose/L-galactose pathway. J. Exp. Bot. 63, 229–239. doi: 10.1093/jxb/err275
Bandoniene, D., and Murkovic, M. (2002). On-line HPLC-DPPH screening method for evaluation of radical scavenging phenols extracted from apples (Malus domestica L.). J. Agric. Food Chem. 50, 2482–2487. doi: 10.1021/jf011475s
Barros, L., Cabrita, L., Boas, M. V., Carvalho, A. M., and Ferreira, I. C. F. R. (2011). Chemical, biochemical and electrochemical assays to evaluate phytochemicals and antioxidant activity of wild plants. Food Chem. 127, 1600–1608. doi: 10.1016/j.foodchem.2011.02.024
Barros, L., Falcão, S., Baptista, P., Freire, C., Vilas-Boas, M., and Ferreira, I. C. F. R. (2008). Antioxidant activity of Agaricus sp. mushrooms by chemical, biochemical and electrochemical assays. Food Chem. 111, 61–66. doi: 10.1016/j.foodchem.2008.03.033
Bener, M., Ozyürek, M., Güçlü, K., and Apak, R. (2010). Development of a low-cost optical sensor for cupric reducing antioxidant capacity measurement of food extracts. Anal. Chem. 82, 4252–4258. doi: 10.1021/ac100646k
Blasco, A. J., Crevillén, A. G., González, M. C., and Escarpa, A. (2007). Direct electrochemical sensing and detection of natural antioxidants and antioxidant capacity in vitro systems. Electroanalysis 19, 2275–2286. doi: 10.1002/elan.200704004
Borges, C. V., Amorim, V. B., Ramlov, F., Ledo, C. A., Donato, M., Maraschin, M., et al. (2014). Characterisation of metabolic profile of banana genotypes, aiming at biofortified Musa spp. cultivars. Food Chem. 145, 496–504. doi: 10.1016/j.foodchem.2013.08.041
Bulley, S., and Laing, W. (2016). The regulation of ascorbate biosynthesis. Curr. Opin. Plant Biol. 33, 15–22. doi: 10.1016/j.pbi.2016.04.010
Bulley, S. M., Rassam, M., Hoser, D., Otto, W., Schünemann, N., Wright, M., et al. (2009). Gene expression studies in kiwifruit and gene over-expression in Arabidopsis indicates that GDP-L-galactose guanyltransferase is a major control point of vitamin C biosynthesis. J. Exp. Bot. 60, 765–778. doi: 10.1093/jxb/ern327
Bulley, S., Wright, M., Rommens, C., Yan, H., Rassam, M., Lin-Wang, K., et al. (2012). Enhancing ascorbate in fruits and tubers through over-expression of the l-galactose pathway gene GDP-l-galactose phosphorylase. Plant Biotechnol. J. 10, 390–397. doi: 10.1111/j.1467-7652.2011.00668.x
Bunaciu, A. A., Aboul-Enein, H. Y., and Fleschin, S. (2012). FTIR spectrophotometric methods used for antioxidant activity assay in medicinal plants. Appl. Spectrosc. Rev. 47, 245–255. doi: 10.1080/05704928.2011.645260
Cabrera, A. C., and Prieto, J. M. (2010). Application of artificial neural networks to the prediction of the antioxidant activity of essential oils in two experimental in vitro models. Food Chem. 118, 141–146. doi: 10.1016/j.foodchem.2009.04.070
Cantín, C. M., Tian, L., Qin, X., and Crisosto, C. H. (2011). Copigmentation triggers the development of skin burning disorder on peach and nectarine fruit [Prunus persica (L.) Batsch]. J. Agric. Food Chem. 59, 2393–2402. doi: 10.1021/jf104497s
Cata, A., Stefanut, M. N., Pop, R., Tanasie, C., Moşoarca, C., and Zamfir, A. D. (2016). Evaluation of antioxidant activities of some small fruits containing anthocyanins using electrochemical and chemical methods. Croat. Chem. Acta 89, 37–48. doi: 10.5562/cca2656
Cazzonelli, C. I., and Pogson, B. J. (2010). Source to sink: regulation of carotenoid biosynthesis in plants. Trends Plant Sci. 15, 266–274. doi: 10.1016/j.tplants.2010.02.003
Çekiç, S. D., Avan, A. N., Uzunboy, S., and Apak, R. (2015). A colourimetric sensor for the simultaneous determination of oxidative status and antioxidant activity on the same membrane: N,N-Dimethyl-p-phenylene diamine (DMPD) on nafion. Anal. Chim. Acta 885, 60–70. doi: 10.1016/j.aca.2015.01.041
Chagné, D., Kirk, C., How, N., Whitworth, C., Fontic, C., Reig, G., et al. (2016). A functional genetic marker for apple red skin coloration across different environments. Tree Genet. Genomes 12:67. doi: 10.1007/s11295-016-1025-8
Chagné, D., Krieger, C., Rassam, M., Sullivan, M., Fraser, J., André, C., et al. (2012). QTL and candidate gene mapping for polyphenolic composition in apple fruit. BMC Plant Biol. 12:12. doi: 10.1186/1471-2229-12-12
Chevion, S., Roberts, M. A., and Chevion, M. (2000). The use of cyclic voltammetry for the evaluation of antioxidant capacity. Free Radic. Biol. Med. 28, 860–870. doi: 10.1016/S0891-5849(00)00178-7
Choleva, T. G., Kappi, F. A., Giokas, D. L., and Vlessidis, A. G. (2015). Paper-based assay of antioxidant activity using analyte-mediated on-paper nucleation of gold nanoparticles as colorimetric probes. Anal. Chim. Acta 860, 61–69. doi: 10.1016/j.aca.2014.12.025
Cimpoiu, C., Cristea, V.-M., Hosu, A., Sandru, M., and Seserman, L. (2011). Antioxidant activity prediction and classification of some teas using artificial neural networks. Food Chem. 127, 1323–1328. doi: 10.1016/j.foodchem.2011.01.091
Cortés-Olmos, C., Leiva-Brondo, M., Roselló, J., Raigón, M. D., and Cebolla-Cornejo, J. (2014). The role of traditional varieties of tomato as sources of functional compounds. J. Sci. Food Agric. 94, 2888–2904. doi: 10.1002/jsfa.6629
Cosio, M. S., Buratti, S., Mannino, S., and Benedetti, S. (2006). Use of an electrochemical method to evaluate the antioxidant activity of herb extracts from the Labiatae family. Food Chem. 97, 725–731. doi: 10.1016/j.foodchem.2005.05.043
Cruz-Rus, E., Amaya, I., Sánchez-Sevilla, J. F., Botella, M. A., and Valpuesta, V. (2011). Regulation of L-ascorbic acid content in strawberry fruits. J. Exp. Bot. 62, 4191–4201. doi: 10.1093/jxb/err122
D'Ambrosio, C., Giorio, G., Marino, I., Merendino, A., Petrozza, A., Salfi, L., et al. (2004). Virtually complete conversion of lycopene into β-carotene in fruits of tomato plants transformed with the tomato lycopene β-cyclase (tlcy-b) cDNA. Plant Sci. 166, 207–214. doi: 10.1016/j.plantsci.2003.09.015
Davey, M. W., Mellidou, I., and Keulemans, W. (2009). Considerations to prevent the breakdown and loss of fruit carotenoids during extraction and analysis in Musa. J. Chromatogr. A 1216, 5759–5762. doi: 10.1016/j.chroma.2009.06.022
Davey, M. W., Van Montagu, M., Inzé, D., Sanmartin, M., Kanellis, A., Smirnoff, N., et al. (2000). Plant L-ascorbic acid: chemistry, function, metabolism, bioavailability and effects of processing. J. Sci. Food Agric. 80, 825–860. doi: 10.1002/(SICI)1097-0010(20000515)80:7<825::AID-JSFA598>3.0.CO;2-6
Davey, M. W., Stals, E., Ngoh-Newilah, G., Tomekpe, K., Lusty, C., Markham, R., et al. (2007). Sampling strategies and variability in fruit pulp micronutrient contents of west and central african bananas and plantains (Musa species). J. Agric. Food Chem. 55, 2633–2644. doi: 10.1021/jf063119l
Drogoudi, P. D., Michailidis, Z., and Pantelidis, G. (2007). Peel and flesh antioxidant content and harvest quality characteristics of seven apple cultivars. Sci. Hort. 115, 149–153. doi: 10.1016/j.scienta.2007.08.010
Drogoudi, P. D., and Pantelidis, G. (2011). Effects of position on canopy and harvest time on fruit physico-chemical and antioxidant properties in different apple cultivars. Sci. Hort. 129, 752–760. doi: 10.1016/j.scienta.2011.05.036
Drogoudi, P., Pantelidis, G. E., Goulas, V., Manganaris, G. A., Ziogas, V., and Manganaris, A. (2016). The appraisal of qualitative parameters and antioxidant contents during post-harvest peach fruit ripening underlines the genotype significance. Postharvest Biol. Technol. 115, 142–150. doi: 10.1016/j.postharvbio.2015.12.002
Dumarey, M., van Nederkassel, A. M., Deconinck, E., and Vander Heyden, Y. (2008). Exploration of linear multivariate calibration techniques to predict the total antioxidant capacity of green tea from chromatographic fingerprints. J. Chromatogr. A 1192, 81–88. doi: 10.1016/j.chroma.2008.03.052
Dumas, Y., Dadomo, M., Di Lucca, G., and Grolier, P. (2003). Effects of environmental factors and agricultural techniques on antioxidant content of tomatoes. J. Sci. Food Agric. 83, 369–382. doi: 10.1002/jsfa.1370
Espley, R. V., Bovy, A., Bava, C., Jaeger, S. R., Tomes, S., Norling, C., et al. (2013). Analysis of genetically modified red-fleshed apples reveals effects on growth and consumer attributes. Plant Biotechnol. J. 11, 408–419. doi: 10.1111/pbi.12017
Espley, R. V., Brendolise, C., Chagné, D., Kutty-Amma, S., Green, S., Volz, R., et al. (2009). Multiple repeats of a promoter segment causes transcription factor autoregulation in red apples. Plant Cell 21, 168–183. doi: 10.1105/tpc.108.059329
Farré, G., Bai, C., Twyman, R. M., Capell, T., Christou, P., and Zhu, C. (2011). Nutritious crops producing multiple carotenoids - a metabolic balancing act. Trends Plant Sci. 16, 532–540. doi: 10.1016/j.tplants.2011.08.001
Fernie, A. R., Tadmor, Y., and Zamir, D. (2006). Natural genetic variation for improving crop quality. Curr. Opin. Plant Biol. 9, 196–202. doi: 10.1016/j.pbi.2006.01.010
Font i Forcada, C., Gradziel, T. M., Gogorcena, Y., and Moreno, M. A. (2014). Phenotypic diversity among local Spanish and foreign peach and nectarine [Prunus persica (L.) Batsch] accessions. Euphytica 197, 261–277. doi: 10.1007/s10681-014-1065-9
Fotopoulos, V., and Kanellis, A. K. (2013). Altered apoplastic ascorbate redox state in tobacco plants via ascorbate oxidase overexpression results in delayed dark-induced senescence in detached leaves. Plant Physiol. Biochem. 73, 154–160. doi: 10.1016/j.plaphy.2013.09.002
Foyer, C. H., and Noctor, G. (2011). Ascorbate and glutathione: the heart of the redox hub. Plant Physiol. 155, 2–18. doi: 10.1104/pp.110.167569
Fraser, P. D., and Bramley, P. M. (2004). The biosynthesis and nutritional uses of carotenoids. Prog. Lipid Res. 43, 228–265. doi: 10.1016/j.plipres.2003.10.002
Fraser, P. D., Romer, S., Shipton, C. A., Mills, P. B., Kiano, J. W., Misawa, N., et al. (2002). Evaluation of transgenic tomato plants expressing an additional phytoene synthase in a fruit-specific manner. Proc. Natl. Acad. Sci. U.S.A. 99, 1092–1097. doi: 10.1073/pnas.241374598
Galpaz, N., Wang, Q., Menda, N., Zamir, D., and Hirschberg, J. (2008). Abscisic acid deficiency in the tomato mutant high-pigment 3 leading to increased plastid number and higher fruit lycopene content. Plant J. 53, 717–730. doi: 10.1111/j.1365-313X.2007.03362.x
Gest, N., Gautier, H., and Stevens, R. (2013). Ascorbate as seen through plant evolution: the rise of a successful molecule? J. Exp. Bot. 64, 33–53. doi: 10.1093/jxb/ers297
Gilbert, L., Alhagdow, M., Nunes-Nesi, A., Quemener, B., Guillon, F., Bouchet, B., et al. (2009). GDP-d-mannose 3,5-epimerase (GME) plays a key role at the intersection of ascorbate and non-cellulosic cell-wall biosynthesis in tomato. Plant J. 60, 499–508. doi: 10.1111/j.1365-313X.2009.03972.x
Goulas, V., Exarchou, V., Kanetis, L., and Gerothanassis, I. P. (2014). Evaluation of the phytochemical content, antioxidant activity and antimicrobial properties of mountain tea (Sideritis syriaca) decoction. J. Funct. Food 6, 248–258. doi: 10.1016/j.jff.2013.10.014
Goulas, V., Gomez-Caravaca, A. M., Exarchou, V., Gerothanassis, I. P., Segura-Carretero, A., and Gutiérrez, A. F. (2012). Exploring the antioxidant potential of Teucrium polium extracts by HPLC-SPE-NMR and on-line radical-scavenging activity detection. LWT-Food Sci. Technol. 46, 104–109. doi: 10.1016/j.lwt.2011.10.019
Hancock, R. D., Walker, P. G., Pont, S. D. A., Marquis, N., Vivera, S., Gordon, S. L., et al. (2007). L-Ascorbic acid accumulation in fruit of Ribes nigrum occurs by in situ biosynthesis via the L-galactose pathway. Funct. Plant Biol. 34:1080. doi: 10.1071/FP07221
Henry-Kirk, R. A., McGhie, T. K., Andre, C. M., Hellens, R. P., and Allan, A. C. (2012). Transcriptional analysis of apple fruit proanthocyanidin biosynthesis. J. Exp. Bot. 63, 5437–5450. doi: 10.1093/jxb/ers193
Hu, D.-G., Sun, C.-H., Ma, Q.-J., You, C.-X., Cheng, L., and Hao, Y.-J. (2016). MdMYB1 regulates anthocyanin and malate accumulation by directly facilitating their transport into vacuoles in apples. Plant Physiol. 170, 1315–1330. doi: 10.1104/pp.15.01333
Ilahy, R., Hdider, C., Lenucci, M. S., Tlili, I., and Dalessandro, G. (2011). Antioxidant activity and bioactive compound changes during fruit ripening of high-lycopene tomato cultivars. J. Food Comp. Anal. 24, 588–595. doi: 10.1016/j.jfca.2010.11.003
Ioannidi, E., Kalamaki, M. S., Engineer, C., Pateraki, I., Alexandrou, D., Mellidou, I., et al. (2009). Expression profiling of ascorbic acid-related genes during tomato fruit development and ripening and in response to stress conditions. J. Exp. Bot. 60, 663–678. doi: 10.1093/jxb/ern322
Jeon, J. S., Kim, H. G., Um, B. H., and Kim, C. Y. (2011). Rapid detection of antioxidant flavonoids in Azalea (Rhododendron mucronulatum) flowers using on-line HPLC-ABTS system and preparative isolation of three flavonoids by centrifugal partition chromatography. Sep. Sci. Technol. 46, 518–524. doi: 10.1080/01496395.2010.520296
Jourdan, M., Gagné, S., Dubois-Laurent, C., Maghraoui, M., Huet, S., Suel, A., et al. (2015). Carotenoid content and root color of cultivated carrot: a candidate-gene association study using an original broad unstructured population. PLoS ONE 10:e116674. doi: 10.1371/journal.pone.0116674
Kanellis, A., and Manganaris, G. (2014). “Antioxidants and bioactive compounds in fruits,” in Fruit Ripening: Physiology, Signalling and Genomics, eds P. Nath, M. Bouzayen, A. K. Mattoo, and J. C. Pech (CABI), 99–126.
Karadag, A., Ozcelik, B., and Saner, S. (2009). Review of methods to determine antioxidant capacities. Food Anal. Methods 2, 41–60. doi: 10.1007/s12161-008-9067-7
Khan, S. A., Chibon, P.-Y., de Vos, R. C., Schipper, B. A., Walraven, E., Beekwilder, J., et al. (2012). Genetic analysis of metabolites in apple fruits indicates an mQTL hotspot for phenolic compounds on linkage group 16. J. Exp. Bot. 63, 2895–2908. doi: 10.1093/jxb/err464
Kitsati, N., Fokas, D., Ouzouni, M. D., Mantzaris, M. D., Barbouti, A., and Galaris, D. (2012). Lipophilic caffeic acid derivatives protect cells against H2O2-induced DNA damage by chelating intracellular labile Iron. J. Agric. Food Chem. 60, 7873–7879. doi: 10.1021/jf301237y
Koutsika-Sotiriou, M., Mylonas, I., Tsivelikas, A., and Traka-Mavrona, E. (2016). Compensation studies on the tomato landrace ‘Tomataki Santorinis’. Sci. Hortic. 198, 78–85. doi: 10.1016/j.scienta.2015.11.006
Kwon, J. H., Jun, J. H., Nam, E. Y., Chung, K. H., Hong, S. S., Yoon, I. K., et al. (2015). Profiling diversity and comparison of eastern and western cultivars of Prunus persica based on phenotypic traits. Euphytica 206, 401–415. doi: 10.1007/s10681-015-1494-0
Laing, W. A., Martínez-Sánchez, M., Wright, M. A., Bulley, S. M., Brewster, D., Dare, A. P., et al. (2015). An upstream open reading frame is essential for feedback regulation of ascorbate biosynthesis in Arabidopsis. Plant Cell 27, 772–786. doi: 10.1105/tpc.114.133777
Lam, H. S., Proktor, A., Howard, L., and Cho, M. J. (2015). Rapid fruit extracts antioxidant capacity determination by fourier transform infrared spectroscopy. J. Food Sci. 70, C545–C549. doi: 10.1111/j.1365-2621.2005.tb08303.x
Lee, J. M., Joung, J.-G., McQuinn, R., Chung, M.-Y., Fei, Z., Tieman, D., et al. (2012). Combined transcriptome, genetic diversity and metabolite profiling in tomato fruit reveals that the ethylene response factor SlERF6 plays an important role in ripening and carotenoid accumulation. Plant J. 70, 191–204. doi: 10.1111/j.1365-313X.2011.04863.x
Legua, P., Forner-Giner, M. Á., Nuncio-Jáuregui, N., and Hernández, F. (2016). Polyphenolic compounds, anthocyanins and antioxidant activity of nineteen pomegranate fruits: a rich source of bioactive compounds. J. Funct. Foods 23, 628–636. doi: 10.1016/j.jff.2016.01.043
Lesschaeve, I., and Noble, A. C. (2005). Polyphenols: factors influencing their sensory properties and their effects on food and beverage preferences. Am. J. Clin. Nutr. 81, 330S–335S.
Li, J., Fu, B., Hou, C., Huo, D., Yang, M., Shen, C., et al. (2016). A colorimetric multilayer sensor for discriminating red wine and green tea by measurement of antioxidant activity. Anal. Meth. 8, 3345–3352. doi: 10.1039/C5AY03360H
Li, L., Lu, M., and An, H. (2017). Expression profiles of the genes involved in l-ascorbic acid biosynthesis and recycling in Rosa roxburghii leaves of various ages. Acta Physiol. Plant. 39, 44–53. doi: 10.1007/s11738-016-2346-9
Li, M., Ma, F., Liang, D., Li, J., and Wang, Y. (2010). Ascorbate biosynthesis during early fruit development is the main reason for its accumulation in kiwi. PLoS ONE 5:e14281. doi: 10.1371/journal.pone.0014281
Lima, G. P. P., Vianello, F., Corrêa, C. R., Campos, R. A., da, S., and Borguini, M. G. (2014). Polyphenols in fruits and vegetables and its effect on human health. Food Nutr. Sci. 5, 1065–1082. doi: 10.4236/fns.2014.511117
Liu, H., Cao, J., and Jiang, W. (2015). Changes in phenolics and antioxidant property of peach fruit during ripening and responses to 1-methylcyclopropene. Postharvest Biol. Technol. 108, 111–118. doi: 10.1016/j.postharvbio.2015.06.012
Lu, S., and Li, L. (2008). Carotenoid metabolism: biosynthesis, regulation, and beyond. J. Integr. Plant Biol. 50, 778–785. doi: 10.1111/j.1744-7909.2008.00708.x
Lu, X., and Rasco, B. A. (2012). Determination of antioxidant content and antioxidant activity in foods using infrared spectroscopy and chemometrics: a review. Crit. Rev. Food Sci. Nutr. 52, 853–875. doi: 10.1080/10408398.2010.511322
Lu, X., Wang, J., Al-Qadiri, H. M., Ross, C. F., Powers, J. R., Tang, J., et al. (2011a). Determination of total phenolic content and antioxidant capacity of onion (Allium cepa) and shallot (Allium oschaninii) using infrared spectroscopy. Food Chem. 129, 637–644. doi: 10.1016/j.foodchem.2011.04.105
Lu, X., Ross, C. F., Powers, J. R., Aston, D. E., and Rasco, B. A. (2011b). Determination of total phenolic content and antioxidant activity of garlic (Allium sativum) and elephant garlic (Allium ampeloprasum) by attenuated total reflectance-Fourier Transformed Infrared Spectroscopy. J. Agric. Food Chem. 59, 5215–5221. doi: 10.1021/jf201254f
Lucio-Gutiérrez, J. R., Garza-Juárez, A., Coello, J., Maspoch, S., Salazar-Cavazos, M. L., Salazar-Aranda, R., et al. (2012). Multi-wavelength high-performance liquid chromatographic fingerprints and chemometrics to predict the antioxidant activity of Turnera diffusa as part of its quality control. J. Chromatogr. A 1235, 68–76. doi: 10.1016/j.chroma.2012.02.042
Magalhães, L. M., Ribeiro, J. P. N., Segundo, M. A., Reis, S., and Lima, J. L. F. C. (2009). Multi-syringe flow-injection systems improve antioxidant assessment. Trends Anal. Chem. 28, 952–960. doi: 10.1016/j.trac.2009.04.016
Magalhães, L. M., Segundo, M. A., Reis, S., and Lima, J. L. F. C. (2008). Methodological aspects about in vitro evaluation of antioxidant properties. Anal. Chim. Acta 613, 1–19. doi: 10.1016/j.aca.2008.02.047
Magalhães, L. M., Segundo, M. A., Reis, S., Lima, J. L. F. C., Tóth, I. V., and Rangel, A. O. S. S. (2007). Automatic flow system for sequential determination of ABTS + scavenging capacity and Folin-Ciocalteu index: a comparative study in food products. Anal. Chim. Acta 592, 193–201. doi: 10.1016/j.aca.2007.04.028
Manganaris, G. A., Goulas, V., Vicente, A. R., and Terry, L. A. (2014). Berry antioxidants: small fruits providing large benefits. J. Sci. Food Agric. 94, 825–833. doi: 10.1002/jsfa.6432
Martins, A. C., Bukman, L., Vargas, A. M. M., Barizão, É. O., Moraes, J. C. G., Visentainer, J. V., et al. (2013). The antioxidant activity of teas measured by the FRAP method adapted to the FIA system: optimizing the conditions using the response surface methodology. Food Chem. 138, 574–580. doi: 10.1016/j.foodchem.2012.10.143
Martí, R., Roselló, S., and Cebolla-Cornejo, J. (2016). Tomato as a source of carotenoids and polyphenols targeted to cancer prevention. Cancers (Basel) 8:E58. doi: 10.3390/cancers8060058
Melino, V. J., Soole, K. L., and Ford, C. M. (2009). Ascorbate metabolism and the developmental demand for tartaric and oxalic acids in ripening grape berries. BMC Plant Biol. 9:145. doi: 10.1186/1471-2229-9-145
Mellidou, I., Chagné, D., Laing, W., Keulemans, J., and Davey, M. W. (2012a). Allelic variation in paralogs of GDP-L-galactose phosphorylase is a major determinant of vitamin C concentrations in apple fruit. Plant Physiol. 160, 1613–1629. doi: 10.1104/pp.112.203786
Mellidou, I., and Kanellis, A. K. (2017). Genetic control of ascorbic acid biosynthesis and recycling in horticultural crops. Front. Chem. 5:50. doi: 10.3389/fchem.2017.00050
Mellidou, I., Keulemans, J., Kanellis, A. K., and Davey, M. W. (2012b). Regulation of fruit ascorbic acid concentrations during ripening in high and low vitamin C tomato cultivars. BMC Plant Biol. 12:239. doi: 10.1186/1471-2229-12-239
Mirosavljević, K., Bilić, B., and Benković-Lačić, T. (2015). Antioxidant activity of selected apple cultivars studied by electron paramagnetic resonance. Acta Biol. Cracov. Bot. 57, 125–129. doi: 10.1515/abcsb-2015-0001
Morsy, M. A., and Khaled, M. M. (2002). Novel EPR characterization of the antioxidant activity of tea leaves. Spectrochim. Acta A 58, 1271–1277. doi: 10.1016/S1386-1425(01)00716-8
Naska, A., and Trichopoulou, A. (2014). Back to the future: the Mediterranean diet paradigm. Nutr. Metab. Cardiovasc. Dis. 24, 216–219. doi: 10.1016/j.numecd.2013.11.007
Niederländer, H. A. G., van Beek, T. A., Bartasiute, A., and Koleva, I. I. (2008). Antioxidant activity assays on-line with liquid chromatography. J. Chromatography A 1210, 121–134. doi: 10.1016/j.chroma.2008.09.061
Orłowska, M., Kowalska, T., Sajewicz, M., Pytlakowska, K., Bartoszek, M., Polak, J., et al. (2015). Antioxidant activity of selected thyme (Thymus L.) species and study of the equivalence of different measuring methodologies. J. AOAC Int. 4, 876–882. doi: 10.5740/jaoacint.SGE6-Orlowska
Osorio, C., Carriazo, J. G., and Almanza, O. (2011). Antioxidant activity of corozo (Bactris guineensis) fruit by electron paramagnetic resonance (EPR) spectroscopy. Eur. Food Res. Technol. 233, 103–108. doi: 10.1007/s00217-011-1499-4
Pace, B., Cefola, M., Renna, F., Renna, M., Serio, F., and Attolico, G. (2013). Multiple regression models and computer vision systems to predict antioxidant activity and total phenols in pigmented carrots. J. Food Eng. 117, 74–81. doi: 10.1016/j.jfoodeng.2013.02.005
Pérez-Martínez, M., Caemmerer, B., De Peña, M. P., Concepción, C., and Kroh, L. W. (2010). Influence of brewing method and acidity regulators on the antioxidant capacity of coffee brews. J. Agric. Food Chem. 58, 2958–2965. doi: 10.1021/jf9037375
Pissard, A., Baeten, V., Romnée, J., Dupont, P., Mouteau, A., and Lateur, M. (2012). Classical and NIR measurements of the quality and nutritional parameters of apples: a methodological study of intra-fruit variability. Biotechnol. Agron. Soc. Environ. 16, 294–306.
Pulgarín, J. A. M., Bermejo, L. F. G., and Durán, A. C. (2012). Use of the attenuation of luminol-perborate chemiluminescence with flow injection analysis for the total antioxidant activity in tea Infusions, wines, and grape seeds. Food Anal. Method. 5, 366–372. doi: 10.1007/s12161-011-9246-9
Ravel, C., Dardevet, M., Leenhardt, F., Bordes, J., Joseph, J. L., Perretant, M. R., et al. (2013). Improving the yellow pigment content of bread wheat flour by selecting the three homoeologous copies of Psy1. Mol. Breed. 31, 87–99. doi: 10.1007/s11032-012-9772-1
Rodríguez-Villalón, A., Gas, E., and Rodríguez-Concepción, M. (2009). Phytoene synthase activity controls the biosynthesis of carotenoids and the supply of their metabolic precursors in dark-grown Arabidopsis seedlings. Plant J. 60, 424–435. doi: 10.1111/j.1365-313X.2009.03966.x
Ronen, G., Carmel-Goren, L., Zamir, D., and Hirschberg, J. (2000). An alternative pathway to β-carotene formation in plant chromoplasts discovered by map-based cloning of beta and old-gold color mutations in tomato. Proc. Natl. Acad. Sci. U.S.A. 97, 11102–11107. doi: 10.1073/pnas.190177497
Saeed, M., Brewer, L., Johnston, J., McGhie, T. K., Gardiner, S. E., Heyes, J. A., et al. (2014). Genetic, metabolite and developmental determinism of fruit friction discolouration in pear. BMC Plant Biol. 14:241. doi: 10.1186/s12870-014-0241-3
Sgherri, C., Pinzino, C., Navari-Izzo, F., and Izzo, R. (2011). Contribution of major lipophilic antioxidants to the antioxidant activity of basil extracts: an EPR study. J. Sci. Food Agric. 91, 1128–1134. doi: 10.1002/jsfa.4296
Shahidi, F., and Zhong, Y. (2015). Measurement of antioxidant activity. J. Funct. Foods 18, 757–781. doi: 10.1016/j.jff.2015.01.047
Shpigun, L. K., Arharova, M. A., Brainina, K. Z., and Ivanova, A. V. (2006). Flow injection potentiometric determination of total antioxidant activity of plant extracts. Anal. Chim. Acta 573, 419–426. doi: 10.1016/j.aca.2006.03.094
Sochor, J., Skutkova, H., Babula, P., Zitka, O., Cernei, N., Rop, O., et al. (2011). Mathematical evaluation of the amino acid and polyphenol content and antioxidant activities of fruits from different apricot cultivars. Molecules 16, 7428–7457. doi: 10.3390/molecules16097428
Steinberg, I. M., and Milardović, S. (2007). Chromogenic radical based optical sensor membrane for screening of antioxidant activity. Talanta 71, 1782–1787. doi: 10.1016/j.talanta.2006.08.015
Stephens, M. J., Scalzo, J., Alspach, P. A., Beatson, R. A., and Conner, A. M. (2009). Genetic variation and covariation of yield and phytochemical traits in a red raspberry factorial study. J. Am. Soc. Hortic. Sci. 134, 445–452.
Stevens, R., Buret, M., Duffé, P., Garchery, C., Baldet, P., Rothan, C., et al. (2007). Candidate genes and quantitative trait loci affecting fruit ascorbic acid content in three tomato populations. Plant Physiol. 143, 1943–1953. doi: 10.1104/pp.106.091413
Stevens, R., Page, D., Gouble, B., Garchery, C., Zamir, D., and Causse, M. (2008). Tomato fruit ascorbic acid content is linked with monodehydroascorbate reductase activity and tolerance to chilling stress. Plant Cell Environ. 31, 1086–1096. doi: 10.1111/j.1365-3040.2008.01824.x
Stevenson, D., and Scalzo, J. (2012). Anthocyanin composition and content of blueberries for the world. J. Berry Res. 2, 179–189. doi: 10.3233/JBR-2012-038
Sun-Waterhouse, D., Smith, B. G., O'Connor, C. J., and Melton, L. D. (2008). Effect of raw and cooked onion dietary fibre on the antioxidant activity of ascorbic acid and quercetin. Food Chem. 111, 580–585. doi: 10.1016/j.foodchem.2008.04.023
Velasco, R., Zharkikh, A., Affourtit, J., Dhingra, A., Cestaro, A., Kalyanaraman, A., et al. (2010). The genome of the domesticated apple (Malus × domestica Borkh.). Nat. Genet. 42, 833–839. doi: 10.1038/ng.654
Volz, R. K., and McGhie, T. K. (2011). Genetic variability in apple fruit polyphenol composition in Malus × domestica and Malus sieversii germplasm grown in New Zealand. J. Agric. Food Chem. 59, 11509–11521. doi: 10.1021/jf202680h
Volz, R. K., McGhie, T. K., and Kumar, S. (2014). Variation and genetic parameters of fruit colour and polyphenol composition in an apple seedling population segregating for red leaf. Tree Genet. Gen. 10, 953–964. doi: 10.1007/s11295-014-0734-0
Wang, L.-Y., Li, D., Deng, Y.-S., Lv, W., and Meng, Q.-W. (2013). Antisense-mediated depletion of tomato GDP-l-galactose phosphorylase increases susceptibility to chilling stress. J. Plant Physiol. 170, 303–314. doi: 10.1016/j.jplph.2012.10.015
Wojdyło, A., Oszmianski, J., and Laskowski, P. (2008). Polyphenolic compounds and antioxidant activity of new and old apple varieties. J. Agric. Food Chem. 56, 6520–6530. doi: 10.1021/jf800510j
Wu, D., Chen, J., Lu, B., Xiong, L., He, Y., and Zhang, Y. (2012). Application of near infrared spectroscopy for the rapid determination of antioxidant activity of bamboo leaf extract. Food Chem. 135, 2147–2156. doi: 10.1016/j.foodchem.2012.07.011
Xu, Q., Chen, L.-L., Ruan, X., Chen, D., Zhu, A., Chen, C., et al. (2012). The draft genome of sweet orange (Citrus sinensis). Nat. Genet. 45, 59–66. doi: 10.1038/ng.2472
Ye, X., Al-Babili, S., Klöti, A., Zhang, J., Lucca, P., Beyer, P., et al. (2000). Engineering the provitamin A (β-Carotene) biosynthetic pathway into (Carotenoid-Free) rice endosperm. Science 287, 303–305. doi: 10.1126/science.287.5451.303
Yoshimura, K., Nakane, T., Kume, S., Shiomi, Y., Maruta, T., Ishikawa, T., et al. (2014). Transient expression analysis revealed the importance of VTC2 expression level in light/dark regulation of ascorbate biosynthesis in Arabidopsis. Biosci. Biotechnol. Biochem. 78, 60–66. doi: 10.1080/09168451.2014.877831
Yuan, H., Zhang, J., Nageswaran, D., and Li, L. (2015). Carotenoid metabolism and regulation in horticultural crops. Hort. Res. 2:15036. doi: 10.1038/hortres.2015.36
Keywords: ascorbic acid, carotenoids, polyphenols, phytochemicals, reactive oxygen species, spectroscopic methods, landrace, traditional cultivars
Citation: Manganaris GA, Goulas V, Mellidou I and Drogoudi P (2018) Antioxidant Phytochemicals in Fresh Produce: Exploitation of Genotype Variation and Advancements in Analytical Protocols. Front. Chem. 5:95. doi: 10.3389/fchem.2017.00095
Received: 11 August 2017; Accepted: 24 October 2017;
Published: 06 February 2018.
Edited by:
Matteo Balderacchi, Independent Researcher, ItalyReviewed by:
Tahira Fatima, Purdue University, United StatesEmmanuel Geoffriau, Agrocampus Ouest, France
Copyright © 2018 Manganaris, Goulas, Mellidou and Drogoudi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: George A. Manganaris, Z2VvcmdlLm1hbmdhbmFyaXNAY3V0LmFjLmN5
 George A. Manganaris
George A. Manganaris Vlasios Goulas
Vlasios Goulas Ifigeneia Mellidou
Ifigeneia Mellidou Pavlina Drogoudi
Pavlina Drogoudi