Abstract
Fluorine magnetic resonance spectroscopy (MRS) and magnetic resonance imaging (MRI) of chemical and physiological processes is becoming more widespread. The strength of this technique comes from the negligible background signal in in vivo19F MRI and the large chemical shift window of 19F that enables it to image concomitantly more than one marker. These same advantages have also been successfully exploited in the design of responsive 19F probes. Part of the recent growth of this technique can be attributed to novel designs of 19F probes with improved imaging parameters due to the incorporation of paramagnetic metal ions. In this review, we provide a description of the theories and strategies that have been employed successfully to improve the sensitivity of 19F probes with paramagnetic metal ions. The Bloch-Wangsness-Redfield theory accurately predicts how molecular parameters such as internuclear distance, geometry, rotational correlation times, as well as the nature, oxidation state, and spin state of the metal ion affect the sensitivity of the fluorine-based probes. The principles governing the design of responsive 19F probes are subsequently described in a “how to” guide format. Examples of such probes and their advantages and disadvantages are highlighted through a synopsis of the literature.
Fluorine MRS and MRI
Fluorine magnetic resonance imaging (MRI) was first reported in 1977, just four years after the development of 1H MRI, by Holland who acquired phantom images of NaF and perfluorotributylamine (Holland et al., 1977). In 1985, McFarland obtained the first 19F in vivo MR images of a rat using a fluorinated probe which accumulates in the liver, Fluosol-DA (McFarland et al., 1985). These pioneering studies demonstrated the key advantages of fluorine nuclei for magnetic resonance spectroscopy (MRS) and imaging (MRI). Today, these advantages are still exploited for monitoring a variety of biological analytes and processes.
The fluorine nuclei (19F, I = ) is attractive due to its 100% abundance and high receptivity (83% that of 1H). These attributes make it comparable to the 1H nuclei (Knight et al., 2011; Ruiz-Cabello et al., 2011). Further, the similar gyromagnetic ratios (γ) of 19F and 1H allow images to be collected on broadband 1H MRI scanners provided broadband amplifiers and dedicated radiofrequency coils are employed to accommodate the slower Larmor frequency of 19F (Stares et al., 2018). The primary advantage of 19F probes over 1H MRI contrast agents is that the background signal in 19F MRI is negligible. The body contains low amounts of fluorine that are primarily embedded in the solid matrices of bones and teeth. As a result, those fluorines have very short transverse relaxation times (T2) and very broad signals that are easily removed with appropriate pulse sequences (Yu et al., 2013a). On the other hand, T1 (longitudinal relaxation times) and T2-based 1H MRI contrast agents, which are primarily gadolinium- and iron oxide nanoparticle-based, generate contrast by modulating the relaxation rates of the naturally-occurring water molecules. The significant background from the endogenous water can render certain imaging more difficult. In addition, 19F nuclei have a large chemical shift range, >300 ppm, that readily allows for the design of fluorine probes featuring two distinct resonances that can be independently imaged if they are sufficiently separated. As will be discussed later, this property is advantageous in the design of ratiometric responsive 19F MRI probes. Ratiometric probes can independently map the distribution of the probe and target analyte in vivo and enable tracking of different cell types or monitoring of multiple markers simultaneously.
Improving the sensitivity of 19F probes
Despite the advantages of 19F MRS and MRI, this field is limited by its low sensitivity that requires the use of high concentrations of probe, typically between 10 and 50 mM. In comparison, Gd-based T1 contrast agents can be readily detected in vivo at substantially lower concentrations (0.1 μM) (Helm et al., 2018). This low sensitivity is due in part to the fact that only the 19F nuclei of the probe are detected. Those nuclei are inherently less concentrated than the 1H of H2O used in 1H MRI, hence the lower sensitivity. This issue is usually addressed by increasing the local concentration of 19F nuclei. A second underlying problem of 19F probes, and particularly diamagnetic ones, is the long T1 relaxation times of 19F, typically 0.5–3 s for small diamagnetic compounds, which necessitate long image acquisition times in order to obtain sufficient signal-to-noise ratio (SNR). This issue is best addressed via the incorporation of appropriate paramagnetic metal ions that shorten the relaxation rates of the 19F nuclei.
Increasing sensitivity by increasing the density of 19F nuclei
The most straightforward approach to increasing 19F MRS/MRI signal intensity is simply to increase the local concentration of 19F nuclei. This is most often accomplished by increasing the number of 19F nuclei on the probe. Complications of this approach are attributed to the hydrophobicity of fluorine which decreases the solubility of a probe in water and affects biodistribution and clearance. Nonetheless, many perfluorocarbons have successfully been used as 19F MRS/MRI oximetry sensors and 19F cell tracking agents provided they are injected as stable emulsions formulated to optimize clearance (Janjic and Ahrens, 2009; Ruiz-Cabello et al., 2011). Highly fluorinated molecules are mostly inert and considered non-toxic, which has facilitated their use in vivo and translation to humans. For instance, fluorinated nano-emulsions are undergoing a phase I clinical trial for 19F MRI cell tracking applications (Ahrens and Zhong, 2013; Ahrens et al., 2014). However, not all fluorinated compounds can be assumed to be entirely non-toxic, especially at the high concentrations required for 19F MRS/MRI. For instance, perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) are both known to affect the function of the pancreas, thyroid, and liver (Chang et al., 2014; Kamendulis et al., 2014).
An often overlooked complication of perfluorinated probes arise when their 19F nuclei are not chemically equivalent. Non-equivalent 19F nuclei of a probe that have small frequency differences between the 19F resonances result in blurry MR images that are the result of incomplete overlap of the images resulting from each 19F resonance (Janjic et al., 2008). This substantially complicates image acquisition, can lead to artifacts, and decreases SNR. It is thus best to increase the number of fluorines in such a way that they remain chemically equivalent.
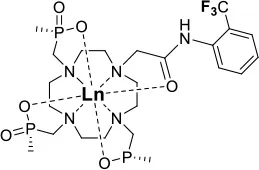
One of the most efficient ways to ensure that each 19F nuclei is chemically equivalent is to incorporate high molecular symmetry in the design. Transition metal and lanthanide complexes with C3 or C4 symmetry are ideally suited for this application if the ligands are fluorinated appropriately. The macrocyclic DOTA (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid) ligand, for instance, adopts a pseudo C4 symmetry in which the acetate arms are arranged in a propeller-like fashion above the plane of the coordinating nitrogen atoms. Four possible isomers exist for this system based on the combination of two macrocyclic ring configurations (square prismatic or square antiprismatic) and the two possible arrangements of the acetate arms (Caravan et al., 1999; Benetollo et al., 2003). The substituents on the acetate arms and the LnIII ion both influence the ratio of isomers observed by NMR such that one isomer can be favored over the others (Parker et al., 2002). The judicial choice of the macrocycle's arms has a notable impact on the 19F NMR spectra of this class of macrocycles and on their effectiveness to function as 19F MRS/MRI probes. Ln(F-DOTPME)− (1,4,7,10,-tetraazacyclododecane-1,4,7,10-tetrakis(methanephosphonic acid mono(2′,2′,2′-trifluoroethyl) ester)), the lanthanide complex of the tetra trifluoroethyl-substituted DOTMP (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetrayl-tetrakis(methylphosphonic acid)) analog (1, Figure 1; Kim et al., 1997), for instance, exists in solution at room temperature as a mixture of all eight isomers, which results in the presence of eight distinct 19F peaks within a 12 (YbIII) to 70 ppm (TmIII) range. As discussed above, the presence of these different isomers limits the efficacy of these complexes to function as 19F MR probes. The metal complexes of the tetra substituted DOTA with aryl-CF3 groups (11, Figure 1; Chalmers et al., 2010) exist primarily as one isomer, whose proportion depends on the nature of the lanthanide ion. This ligand is therefore better suited for 19F MRI applications. The problem of multiple isomers is not limited to tetra-substituted macrocyclic derivatives. In fact, many DOTA analogs with only one or two fluorinated arms are present in solution as more than one isomer (2–4), the percentage of which varies according to the lanthanide (Kenwright et al., 2008; Chalmers et al., 2010; Placidi et al., 2013; Cakic et al., 2016).
Figure 1
Substitution of one or more carboxylate arms with bulky phosphinate groups or the introduction of substituent at the α-position to the ring nitrogen affects this dynamic process. For instance, Chalmers et al. reported two DOTA derivatives incorporating two trifluoroaryl groups and either two carboxylic acid or two glutaric acid substituents at the α-position to the ring nitrogen (5 and 6, Figure 1) (Chalmers et al., 2011a,b). In solution the carboxylic acid derivative, unlike the glutamic acid one, favors one major isomer which accounts for 80% of the 19F NMR spectrum. The selectivity for one isomer can also be improved via the use of a phosphinate arm. For instance, all lanthanide complexes of 7 exist primarily (>87%) as one isomer. In extreme cases, a single isomer of a DOTA analog fluorinated on a single arm can be obtained, as for compounds 8–10 (Figure 1) (Jiang et al., 2011; Davies et al., 2015).
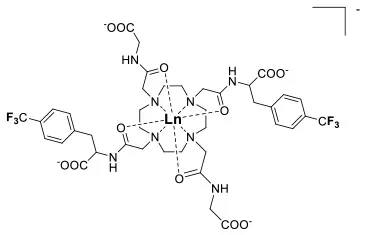
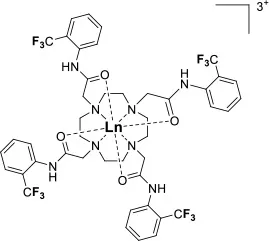
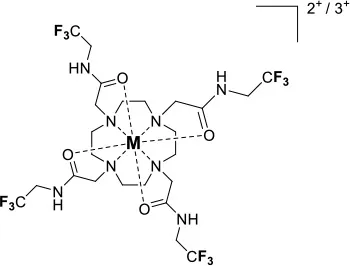
A single isomer for a tetra-substituted DOTA complex which exploits the symmetry of the ligand in order to maximize the number of equivalent fluorines can nonetheless be obtained. Examples include M-DOTAm-F12 (2,2′,2″,2‴-(1,4,7,10-tetraazacyclododecane-1,4,7,10-tetrayl)tetrakis(N-(2,2,2-trifluoroethyl)acetamide)) (12, Figure 1; Srivastava et al., 2017a,b) and more recently the redox-responsive Eu-complex 39 (Figure 9) (Basal et al., 2017). Both of these complexes contain 12 19F nuclei linked to the macrocycle via four amide arms. Importantly, they both exist as a single isomer for which all 12 19F nuclei remain chemically equivalent. This increases the sensitivity of the 19F MR probe and the resolution of the images that can be obtained with it. The high symmetry of the DOTA scaffold can also be exploited with certain transition metals. The unusual but stable 8-coordinate structure of the FeII-DOTAm-F12 maintains a high symmetry and chemical equivalency for its 12 fluorine nuclei (Srivastava et al., 2017a). The higher solubility in water of the M-DOTAm-F12, >100 mM, renders it particularly promising for further development and imaging applications. In summary, the C3 or C4 symmetry of metal complexes can be exploited to increase the number of chemically equivalent 19F nuclei on a probe, but the drawback of multiple stable isomers in solution remains a challenge.
Increasing sensitivity with paramagnetic metal ions
Increasing the number of 19F nuclei per agent can only go so far in terms of increasing the sensitivity of 19F probes. Ultimately, there is a limit to the number of 19F nuclei that can be added to a molecule in such a way that they are chemically equivalent all the while maintaining sufficient water solubility for in vivo applications. A complementary approach to increasing the number of 19F nuclei that is often used simultaneously is to decrease the T1 of the 19F nuclei. Diamagnetic molecules typically have T1 of 19F nuclei in the range of 0.5–3 s. For in vivo imaging applications, that are limited by time, the long T1 of diamagnetic fluorine probes hinders their sensitivity (Harvey et al., 2012b). It is thus preferential to decrease the T1 of the 19F nuclei of fluorine probes. This is best achieved by incorporating paramagnetic metal ions such as lanthanides(III) or iron(II). The metal should be selected with care as different paramagnetic metal ions have different effects on the relaxation of nearby nuclei.
The effect of a paramagnetic metal ion on the T1 and T2 of a 19F nuclei is described by the Bloch-Wangsness-Redfield theory. A 19F nuclei adjacent to a paramagnetic center in a magnetic field can relax via five different mechanisms: (1) chemical shift anisotropy, (2) inter-nuclear dipole-dipole interaction, (3) electron-nucleus contact interaction, (4) electron-nucleus dipole-dipole interaction, and (5) Curie relaxation (Chalmers et al., 2010). Of these, chemical shift anisotropy is negligible for a single 19F nuclei or CF3 group, and inter-nuclear dipole-dipole mechanisms are minimal, accounting for <1% of the total relaxation. The electron-nucleus contact interaction is also negligible since ligand geometries generally locate the 19F nuclei further than 4.5–7.5 Å away from the metal ion, resulting in coupling values of zero. Thus, only the electron-nucleus dipole-dipole and Curie relaxation processes contribute significantly to the relaxation of the 19F nuclei. Given this, the longitudinal (R1 = 1/T1) and transverse (R2 = 1/T2) relaxation rates of 19F nuclei in paramagnetic metal complexes are described by Equations (1) and (2), respectively (Chalmers et al., 2010).
where
In these equations, d is the distance separating the 19F nuclei from the paramagnetic metal ion, μ0 is the permeability of vacuum, γF is the gyromagnetic ratio of the 19F nuclei, T is the temperature in K, k is Boltzmann's constant, and ωF is the Larmor angular frequency of 19F. The effective magnetic moment, μeff, is proportional to the effective electron g-factor (gJ), the Bohr magneton (μB), and the electron angular momentum, J(J+1) according to Equation (3). The electron angular frequency (ωe) is a function of the magnetic field strength (B0) according to Equation (4) in which ℏ is the reduced Planck's constant. The τR+e term is dependent on the rotational correlation time (τR) and the electron spin longitudinal relaxation time (T1e) according to Equation (5).
In Equations (1) and (2), the first term represents the dipolar relaxation characterized by the stochastic electron magnetization of the electron-nucleus dipole-dipole interaction. The second term is the Curie relaxation arising from the interaction between the fluorine nuclear spin and the magnetic dipole induced by the applied magnetic field. Together, they relate R1 and R2 to the effective magnetic moment (μeff) of the paramagnetic metal, the 19F–metal distance (d), the rotational correlation time (τR), the applied magnetic field strength (B0), and the temperature (T). Optimizing the sensitivity of 19F MRS/MRI probes requires understanding each of these relationships.
Dependence on the nature of the metal ion
The effective magnetic moment (μeff) of the paramagnetic metal affects both the electron-nucleus dipole-dipole interaction and, to a greater extent, the Curie relaxation mechanism. The effective magnetic moment of transition metal complexes is a function of both their oxidation state and their ligand fields. Each lanthanide ion has a characteristic μeff value (Table 1).
Table 1
| Lanthanide ion | Ground state term | μeff (B.M.)a | μeff (exp)b | Bleany Constant (Cj)c | Electron relaxation time (T1e)/10−13 sd |
|---|---|---|---|---|---|
| CeIII | 2F5/2 | 2.56 | 2.55 | −6.3 | 0.90 |
| PrIII | 3H4 | 3.62 | 3.47 | −11.0 | 0.57 |
| NdIII | 4I9/2 | 3.68 | 3.69 | −4.2 | 1.15 |
| PmIII | 5I4 | 2.68 | 2.41 | 2.0 | Unknown |
| SmIII | 6H5/2 | 1.55–1.65 | 1.58 | −0.7 | 0.45 |
| EuIII | 7F0 | 3.40–3.51 | 3.4 | 4.0 | 0.09 |
| EuII | 8S7/2 | 7.6–8.0 | 0 | 104e | |
| GdIII | 8S7/2 | 7.94 | 7.63 | 0 | 104-105f |
| TbIII | 7F6 | 9.7 | 9.8 | −86 | 2.03 |
| DyIII | 6H15/2 | 10.6 | 10.3 | −100 | 2.99 |
| HoIII | 5I8 | 10.6 | 10.4 | −39 | 1.94 |
| ErIII | 4H15/2 | 9.6 | 9.4 | 33 | 2.38 |
| TmIII | 3H6 | 7.6 | 7.6 | 53 | 3.69 |
| YbIII | 2F7/2 | 4.5 | 4.3 | 22 | 1.37 |
Magnetic and relaxation properties of lanthanide(III) ions.
The effect of the nature of the transition or lanthanide ion on the relaxation rates of the 19F nuclei of fluorinated complexes and the sensitivity of the resulting fluorine probe has been explored via a series of non-responsive probes. Several fluorinated ligands have been investigated, which enables a more in-depth evaluation of the influence of the structure of the ligand on the 19F relaxation rates of the probe (Figure 1, Table 2). Studies by Parker and coworkers focused on a DOPA ([(4,7,-di{[hydroxyl(methyl)phosphoryl]methyl}-10-({[2-(trifluoromethyl)phenyl]carbamoyl}methyl)-1,4,7,10-tetraazacyclododecan-1-yl)methyl](methyl)phosphinic acid) ligand monosubstituted with ortho aryl-CF3 (7) and DOTA ligand scaffold tetra substituted with aryl-CF3 groups (11) (Chalmers et al., 2010, 2011a). In this scaffold, ortho substitution of the CF3 group lead to a greater decrease in relaxation times due to the shorter distance separating the 19F nuclei from the lanthanide ion (Senanayake et al., 2007; Chalmers et al., 2010). In agreement with the Bloch-Wangsness-Redfield theory, this first study demonstrated that paramagnetic lanthanide ions can increase the R1 by up to two orders of magnitude, with the greatest change observed with those metal ions with the highest μeff, notably, TbIII, DyIII, HoIII, and ErIII (Table 2). Unfortunately, the poor solubility of the tetra substituted complexes in water limited further in vivo studies.
Table 2
| Ligand (B0) | LnIII | δ (ppm) | Δδ (ppm) | R1 (Hz) | T1 (ms) | R2 (Hz) | T2 (ms) | T2/T1 |
|---|---|---|---|---|---|---|---|---|
| 2 (7.0 T)a | EuIII | −61.8 | n.d. | 1.4 | 714 | 18h | 56 | 0.078 |
 | −61.4 | 1.4 | 714 | 19i | 53 | 0.074 | ||
| GdIII | −61.6 | n.d. | 182 | 5.5 | 385 | 2.6 | 0.47 | |
| TbIII | −65.2 | n.d. | 32h | 31 | 48h | 21 | 0.68 | |
| −55.6 | 12i | 83 | 28i | 36 | 0.43 | |||
| 3 (7.0 T)b | Ligand | −61.24 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
 | GdIII | −60.45 | n.d. | 322.6 | 3.1 | 500 | 2.0 | 0.65 |
| YbIII | −60.54 | n.d. | 3.7 | 273 | 238 | 4.2 | 0.015 | |
| 4 (9.4 T)c* | YIII | −62 | 0 | n.d. | n.d. | n.d. | n.d. | n.d. |
 | TbIII | −51.9 | 10.1 | 116 | 8.62 | 119 | 8.4 | 0.975 |
| DyIII | −64.9 | −2.9 | 139 | 7.19 | 220 | 4.55 | 0.632 | |
| HoIII | −64.2 | −2.2 | 109 | 9.17 | 151 | 6.62 | 0.722 | |
| ErIII | −64.8 | −2.8 | 65 | 15.38 | 157 | 6.37 | 0.414 | |
| TmIII | −77.4 | −15.4 | 51 | 19.61 | 91 | 10.99 | 0.56 | |
| 5 (9.4 T)c* | TbIII | −61.9 | n.d. | 2.8 | 357.14 | 132 | 7.58 | 0.021 |
 | DyIII | −99.8 | n.d. | 158 | 6.33 | 213 | 4.69 | 0.742 |
| HoIII | −57.7 | n.d. | 91 | 10.99 | 159 | 6.29 | 0.572 | |
| ErIII | −58.6 | n.d. | 81 | 12.35 | 164 | 6.10 | 0.494 | |
| TmIII | −68.2 | n.d. | 56 | 17.86 | 102 | 9.80 | 0.549 | |
| 7 (9.4 T)c | YIII | −61.2 | 0 | 15 | 66.7 | |||
 | TbIII | −47.7 | 13.5 | 147 | 6.8 | 267 | 3.75 | 0.55 |
| DyIII | −63.6 | −2.4 | 185 | 5.41 | 251 | 3.98 | 0.737 | |
| HoIII | −61.5 | −0.3 | 120 | 8.33 | 143 | 6.99 | 0.839 | |
| ErIII | −72.6 | −11.4 | 109 | 9.17 | 138 | 7.25 | 0.79 | |
| TmIII | −89.5 | −28.3 | 63 | 15.87 | 84 | 11.9 | 0.75 | |
| 10 (11.7 T)d | ligand | −71.2 | 0.752 | 1330 | 1.43 | 701 | 0.53 | |
 | YIII | −71.1 | 0 | 0.66 | 1514 | 1.28 | 783 | 0.52 |
| GdIII | −66.6 | −4.5 | 236 | 4.24 | 602 | 1.66 | 0.39 | |
| TbIII | −63.3 | −7.8 | 13.8 | 72.6 | 24.0 | 41.7 | 0.57 | |
| 11 (9.4 T)e | YIII | −61.6 | 0 | 0.78 | 1282 | 2.9 | 345 | 0.27 |
 | TbIII | −53.9 | 7.7 | 185 | 5.41 | n.d. | n.d. | n.d. |
| HoIII | −59 | 2.6 | 192 | 5.21 | n.d. | n.d. | n.d. | |
| ErIII | −63.5 | −1.9 | 109 | 9.17 | n.d. | n.d. | n.d. | |
| TmIII | −65.1 | −3.5 | 59.7 | 16.8 | n.d. | n.d. | n.d. | |
| 12 (7.0 T)f | ligand | −72.7 | 1.1 | 880 | 1.5 | 680 | 0.77 | |
 | LaIII | −72.1 | 0 | 1.8 | 570 | 2.5 | 400 | 0.70 |
| EuIII | −72.4 | −0.3 | 2.8 | 360 | 24 | 41 | 0.11 | |
| GdIII | −72.0 | 0.1 | 83 | 12 | 7100 | 0.14 | 0.01 | |
| TbIII | −54.1 | 18.0 | 160 | 6.3 | 770 | 1.3 | 0.21 | |
| DyIII | −52.4 | 19.7 | 170 | 5.9 | 450 | 2.2 | 0.37 | |
| HoIII | −61.8 | 10.3 | 130 | 7.6 | 190 | 5.4 | 0.71 | |
| ErIII | −76.5 | −4.4 | 71 | 14 | 110 | 8.8 | 0.63 | |
| TmIII | −83.3 | −11.2 | 39 | 26 | 63 | 16 | 0.62 | |
| YbIII | −75.9 | −3.8 | 7.7 | 130 | 18 | 55 | 0.42 | |
| FeII | −70.1 | 2.0 | 180 | 5.7 | 180 | 5.6 | 0.98 | |
| 13 (7.0 T)g | ligand | −68.9 | 2000 | 0.5 | 20 | 50 | 0.1 | |
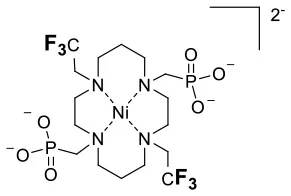 | NiII | −26 | n.d. | 357 | 2.8 | 1111 | 0.9 | 0.32 |
19F chemical shift and relaxation properties for non-responsive paramagnetic fluorine probes.
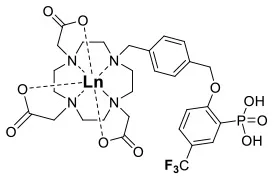
Recently, our lab reported the relaxation properties of the FeII and LnIII complexes of another macrocyclic ligand, DOTAm-F12 (12) (Srivastava et al., 2017a,b). Complexes of this series, including the FeII complex, have a number of advantageous properties for 19F imaging. The complexes each contain 12 fluorine nuclei that are chemically equivalent, resulting in a single 19F resonance. As discussed above, a high number of fluorine nuclei and their equivalency are two necessary components in the design of sensitive 19F probes for MR imaging. The complexes are also highly soluble in water; they can be dissolved in concentrations of up to 100 mM, a necessity for any biological applications. The FeII complex is notable as it is stable both in air and water and is a rare example of an eight coordinate iron complex (Srivastava et al., 2017a). Importantly, for both the FeII and the LnIII complexes, the structure of the macrocycle places the fluorine nuclei from 5.22 to 6.84 Å from the metal ion. As will be discussed later, this is an optimum distance since it minimizes the effect of the shortened T2 of the 19F nuclei. As in Parker's study, the paramagnetic metal ions decrease the T1 of the 19F nuclei by up to two orders of magnitude with the greatest decrease observed with those metal ions having the highest μeff, namely TbIII, DyIII, HoIII, and FeII. Paramagnetic NiII cyclam complexes featuring trans-trifluoroethyl groups were also recently reported by Blahut et al. (2016, 2017). The trans-[Ni(te2f2p)]2− (13, Figure 1) with phosphate ligands has a 19F–Ni distance of ~ 5 Å which facilitates a nearly 180-fold reduction in T1 vs. the free ligand (Table 2). This NiII complex and FeII-DOTAm-F12 (12) exemplify that the diverse electronic and structural parameters of transition metal complexes can also be exploited to optimize 19F paramagnetic relaxation enhancement (PRE).
The sensitivity of an agent, estimated by comparing SNR of solutions of complexes at the same concentration, is dependent not only on T1 but also on T2. The line-broadening characteristic of the shorter T2 of the fluorines of certain paramagnetic complexes significantly affect the sensitivity of the probe. An extreme example being the GdIII complex of DOTAm-F12 that has such a short T2 that the complex could not be observed by MRI. Note that for such complexes with rapid transverse relaxation, the use ultrashort TE (UTE) and zero TE (ZTE) acquisition pulse sequences can sometimes increase the SNR in 19F-MRI (Kislukhin et al., 2016).
Not all paramagnetic metals affect T2 to the same degree as T1. Therefore, the most sensitive metal-based probes are not those whose metal ions have the highest μeff, but those whose 19F have the highest T2/T1 ratio. For M-DOTAm-F12 in water, these are FeII, HoIII, TmIII, and YbIII. Importantly, the media also has a substantial effect on the relaxation times, and particularly on the transverse one. The T2 of all M-DOTAm-F12 complexes decrease substantially in blood. This affects the sensitivity of all metal complexes. HoIII-DOTAm-F12, for instance, is the most sensitive probe in water, yet it could not be detected in blood. The lower T2 value of M-DOTAm-F12 complexes in blood in comparison to water could be due to different parameters. Coordination to albumin or other serum proteins and the resulting increase in rotational correlation time can affect both T1 and T2. Blood is also more viscous than water, and this increase in viscosity also affects τR, T1, and T2. One should therefore not assume that a probe that is sensitive in water is necessarily appropriate for in vivo applications where it will accumulate in blood or tissues. It is therefore recommended that the sensitivity of probes be evaluated not only in water but also in the media in which they are intended to be used. Also note that the greatest effect is not necessarily obtained with lanthanide ions. In blood, the most sensitive probe is FeII-DOTAm-F12 whose limit of detection (300 μM) is more than an order of magnitude lower than that of small diamagnetic probes.
Similar trends have been observed with other lanthanide complexes, such as 2–5, 7, and 10–12 (Figure 1, Table 2). In general, the stronger relaxing metals with the higher μeff near 10, TbIII, DyIII, and HoIII decrease both T1 and T2 the most. Lanthanides such as ErIII, TmIII, and in the extreme case GdIII, have lower T2/T1 ratio and hence lower sensitivity (Table 2) (Chalmers et al., 2010, 2011a,b; Jiang et al., 2011; Harvey et al., 2012b; Placidi et al., 2013; Blahut et al., 2016; Cakic et al., 2016; Srivastava et al., 2017b). Do note that the PRE effect is determined by other parameters beyond the μeff of the metal ion; it is also dependent on the magnetic field strength and on the distance separating the 19F nuclei and the paramagnetic metal ion (Equation 1–5). As mentioned above, transition metals such as FeII (μeff ~ 5) and NiII (μeff ~ 3) (complexes 12 and 13, respectively) can also generate high 19F MRI SNR ratios of 20–30 despite their lower magnetic moments.
Even at greater 19F–M distances, the effect of the paramagnetic metal ion on the relaxation rates of the fluorine nuclei can be notable. The fluorinated complex of Jiang (10, Figure 1) is a DOTA-based complex containing three equivalent trifluoromethyl groups separated by a polyethylene glycol linker that positions the 19F nuclei more than 10 Å away from the paramagnetic metal ion. Despite this great distance, paramagnetic metals such as TbIII, DyIII, HoIII, ErIII, GdIII, FeIII, NiII, and CuII reduce T1 and T2 by >95% (Jiang et al., 2011). A noteworthy conclusion of this study is that the 19F relaxation times of these paramagnetic metal complexes are relatively constant (<5% change) with respect to environmental fluctuations in pH, temperature, and the concentration of either O2 or bovine serum albumin (BSA) (Jiang et al., 2011). It is thus the identity of the paramagnetic metal ion that is the primary contributor to both the chemical shift and the relaxation rates of the fluorines. This observation is important for the development of responsive probes (vide infra).
Dependence on the 19F-metal distance
From the Bloch-Wangsness-Redfield Equations (1–5) given above, both the R1 and the R2 of the fluorine nuclei have a steep dependence on the distance d separating the 19F nuclei from the metal ion. The 19F–M distance is a key parameter in determining the sensitivity of metal-based fluorine probes. The effect of the paramagnetic metal ion on R2 extends to longer distances than that of R1. Both too long and too short a distance can be detrimental. Too long a 19F–M distance and the effect of the paramagnetic metal ion on the relaxation times of the fluorine nuclei is severely diminished. Too short a distance, and the substantial shortening of T2 decreases the sensitivity of the fluorine probe. A higher T2/T1 ratio and thus a more sensitive fluorine probe is obtained if the 19F nuclei are positioned between 4.5 and 7.5 Å from the metal ion (Harvey et al., 2012b).
Within this range, even minor changes in 19F–M ion distance can have an impact on the relaxation rates of fluorine nuclei and the sensitivity of the agent. One such example are the lanthanide complexes containing a single aryl-CF3 moiety positioned either on an acetate-based DOTA (4, Figure 1) or a phosphinate-based DOPA (7, Figure 1) scaffold. The DOTA complexes (4) exists in solution as a mixture of multiple isomers with the primary species representing only 50% of the fluorine signal intensity by NMR. On the other hand, the principal isomer of the phosphinate analog (7) accounts for 87% of the total 19F signal intensity (Chalmers et al., 2011a). The R1 and R2 of lanthanide complexes of these two ligands are different. The R1 of complexes of DOPA scaffold are 10–67% greater than those of the DOTA scaffold (Table 2). However, the R2 of complexes (HoIII, ErIII, and TmIII) of DOPA scaffold are 5–12% smaller than those of DOTA scaffold with an exception of TbIII and DyIII being 14–124% greater than those of DOTA complexes. Global fitting of the relaxation rates collected at multiple field strengths to the Bloch-Wangsness-Redfield theory determined that the differences in R1 and R2 are due to a 0.3 Å shorter 19F–Ln distance in the DOPA complex than in the DOTA one (Chalmers et al., 2010, 2011a). Interestingly, the highest T2/T1 ratio with the DOTA-based scaffold was obtained with TbIII. For the DOPA-based scaffold, it was instead HoIII (0.84), ErIII (0.79), Tm (0.75), and Dy (0.74) that yielded the highest T2/T1 ratio. As is apparent from this example, the best metal ion for one fluorinated ligand is not necessarily the same for another one.
The effect that such minor modifications of the ligand and the structure of the complex have on the T2/T1 ratio and the sensitivity of the probe has been observed with other complexes. The YbIII -DO3A (1,4,7,10-tetraazacyclododecane-1,4,7-tris(acetic acid))-based complex with a fluorinated aryl phosphonate of Angelovski, (3, Figure 1) positions the 19F nuclei 7 Å from the metal, a 19F–Yb distance 0.7 Å greater than in Yb-DOTAm-F12 (Placidi et al., 2013). At 7 T, the T1 of 273 ms is nearly twice that in YbIII-DOTAm-F12. More noticeably, the very short T2 of the fluorines of this complex, 4.2 ms, results in a much smaller T2/T1 ratio and hence a much less sensitive probe than YbIII-DOTAm-F12.
Supramolecular self-assembly enables more facile synthesis of paramagnetic fluorine probes via the formation of ternary complexes. Given the hardness of lanthanide ions, fluorinated carboxylate ligands are particularly well-suited for this approach (8 and 9, Figure 1) (Davies et al., 2015). These assemblies also decrease the relaxation times of the fluorine to a similar degree as observed in the complexes discussed above, although the relatively weak affinity of the carboxylate ligand for the lanthanide (log Ka ~ 4.9) may result in decomplexation of the fluorinated moiety under biological conditions.
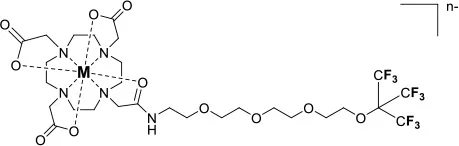
Similarly, paramagnetic metals have also been employed to increase the relaxation rates of fluorine nuclei in perfluorinated emulsions. Perfluorocarbon-based emulsions present an advantage over monomolecular complexes in that they contain a higher density of fluorine nuclei which increases their sensitivity. The design and application of perfluorocarbon tracers for 19F MRI have been reviewed elsewhere. (Knight et al., 2011; Tirotta et al., 2015) Here, the role of GdIII and FeIII in enhancing the relaxation rates of fluorine nuclei in nanoemulsions is explored. Neubauer et al. first reported a paramagnetic fluorinated emulsion containing a liquid perfluoro-15-crown-5 ether core encased in a lipid layer surface-coated with Gd-DTPA-BOA (6,9-bis(carboxymethyl)-3-(2-(((E)-octadec-9-en-1-yl)amino)-2-oxoethyl)-11-oxo-3,6,9,12-tetraazatriacont-21-enoic acid) complexes (14, Figure 2) (Neubauer et al., 2008). The relaxation rates of the fluorine nuclei in this paramagnetic emulsion increases according to an outer-sphere mechanism which requires the perfluorocarbons within the core of the nanoparticle to diffuse close to the GdIII ions on the surface of the lipid monolayer. In this 200 nm assembly, the R1 and R2 of the fluorine nuclei located ~ 15 Å away from the GdIII ion increase 4- and 8-fold, respectively. This, in turn, increases the sensitivity of the emulsion 2-fold (Neubauer et al., 2008). Further calculations on how the size of the nanoparticles, the 19F–Gd distance (d), and the diffusion coefficient of the perfluorocarbon affect the R1 and R2 of the 19F enabled further improvement of the assembly (Hu et al., 2011). A 4-fold relaxation enhancement was observed with a perfluoro-15-crown-5 ether and Gd-DTPA-BOA (14) compared to non-metallated perfluorocarbon. Additionally, a shorter linker between the Gd and the perfluorocarbon core of the DTPA complexes (14 and 15) vs. the Gd-DOTA chelate (16) further increased the fluorine relaxation rates (Hu et al., 2011). Subsequent work by de Vries et al. compared assemblies comprising DOTA-based GdIII complexes to those containing GdIII DTPA analogs (de Vries et al., 2014). Interestingly, Gd-DTPA-BSA (6,9-bis(carboxymethyl)-3-(2-(octadecylamino)-2-oxoethyl)-11-oxo-3,6,9,12-tetraazatriacontanoic acid) (17, R1 = 1.45 s−1) had a greater impact on the relaxation rates of the fluorines than Gd-DOTA-DSPE (2,2′,2″-(10-(2-((2-((((R)-2,3-bis(palmitoyloxy)propoxy)(hydroxy)phosphoryl)oxy)ethyl)amino)-2-oxoethyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triyl)triacetic acid)(18,R1 = 1.05 s−1)and Gd-DOTA-C6-DSPE(2,2′,2″-(10-(2-((6-((2-((((R)-2,3-bis(palmitoyloxy)propoxy)(hydroxy)phosphoryl)oxy)-ethyl)amino)-6-oxohexyl)amino)-2-oxoethyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triyl)triaceticacid)(19, Figure 2). This difference was attributed to a shorter 19F–Gd distance in the emulsions containing the Gd-DTPA complex. As predicted from the Bloch-Wangsness-Redfield theory, GdIII has a limited effect on R1 of nearby fluorines above 6 T. This highlights the limitations of paramagnetic complexes at high field (Neubauer et al., 2008; Hu et al., 2011; de Vries et al., 2014).
Figure 2
These previous examples positioned the paramagnetic metal ions on the surface of the nanoparticles and, thus, a significant distance away from the 19F nuclei. To resolve this issue Kislukhin et al. designed nanoparticles that incorporated the paramagnetic metal ions directly into the fluorous core, thereby decreasing the 19F–M distance (Kislukhin et al., 2016). Despite the electronegativity of the fluorine atoms that reduce the basicity of the ligand, fluorinated β-diketone ligands form neutral, hydrophobic complexes that are sufficiently stable and dispersible in the perfluorocarbon core (20, Figure 2). Three different paramagnetic metal ions were evaluated: FeIII, MnII, and GdIII. Of these, FeIII had the greater impact on the R1 of the 19F nuclei, increasing it 13-fold, and the sensitivity of the nanoparticles, increasing it 5-fold. In comparison, MnII and GdIII increased R1 7- and 5-fold, respectively. Both MnII and GdIII induced severe line broadening which decreased the sensitivity of the assembly. Advantageously, these nanoparticles are readily taken up by glioma cells. Since the nanoparticles also have low cytotoxicity (they reduce the viability of glioma cells by only 20%), they can be used to track cells in vivo 24 h post injection in mice. Do note, though, that it is not possible to distinguish between live and dead cells labeled with fluorine probes 24 h post injection, especially if the cells are engrafted subcutaneously.
Dependence on the magnetic field strength
As the Bloch-Wangsness-Redfield theory indicates, both the R1 and R2 of fluorine nuclei depend on the strength of the magnetic field (Figures 3A,B, respectively). The magnetic field dependencies of R1 and R2 are also a function of the μeff of the metal ion and as such, different metal ions are better suited for either low field or high field experiments. A steeper increase in R1 with respect to magnetic field strength is observed for DyIII and HoIII complexes of para-hydroxyl derivative of 4 than for its TmIII and TbIII, and ErIII complexes (Chalmers et al., 2010; Harvey et al., 2012b). For this ligand scaffold, the stronger relaxing ions such as HoIII are thus more appropriate for experiments performed at lower magnetic field strengths, whereas the weaker TmIII and ErIII provide adequate relaxation enhancement above 7 T (Chalmers et al., 2010; Harvey et al., 2012b).
Figure 3
The higher relaxation rates of fluorine nuclei of paramagnetic complexes allow for the collection of more data per unit time. Consequently, PRE increases both the spectral sensitivity and the SNR of paramagnetic fluorine agents compared to their diamagnetic analogs. Chalmers et al. demonstrated that the DyIII of fluorinated DOPA complex (7) increased the image SNR ~ 13-fold compared to the diamagnetic YIII analog (Chalmers et al., 2011a,b). The improved SNR correlates with a T2/T1 ratio closer to l. With such a ratio, the benefit of increased R1 is not diminished by the increased signal broadening due to rapid transverse relaxation. Similar conclusions were drawn with complexes of DOTAm-F12 (Srivastava et al., 2017b). In theory, sensitivity gains of 15–20 times that of diamagnetic analogs can be achieved with fluorinated lanthanide complexes between 1.5 and 9.4 T, the magnetic fields of clinical relevance (Harvey et al., 2012b). The use of paramagnetic metal ions is thus the most effective strategy to increase the sensitivity of probes for 19F MRS/MRI, enabling imaging of sub-millimolar concentrations of probe in <15 min with adequate SNR (>4) (Chalmers et al., 2011a,b).
The effect of the rotational correlation time, τR
Both the R1 and R2 of fluorine nuclei are affected by their rotational correlation time (τR) as well as by the field strength of the instrument (B0) and the temperature of the sample. Both R1 and R2 increase as the magnetic field strength (B0) increases. This increase is far more significant if the rotational correlation time, τR, is also optimized. Macromolecules, which tumble more slowly (longer τR), have higher R2. As can be seen in Figure 3B, R2 increases drastically for 19F nuclei with τR > 1,000 ps, especially at high magnetic field (>7 T). The resulting extreme line broadening explains why fluorines embedded in bones and teeth in the body are not visible by MRI. The effect of τR on R1 is more complicated (Figure 3A). R1 increases to a maximum when the tumbling rate of the M-19F vector () equals the Larmor frequency of the 19F nuclei; i.e., when = 1 (Modo and Bulte, 2007). Monomolecular lanthanide complexes, such as those discussed in this review, have rotational correlation times on the order of 200–350 ps (Chalmers et al., 2010; Harvey et al., 2012b). Unlike for R2, there is an optimal τR beyond which R1 decreases sharply especially at high magnetic field. The optimal τR is a function of the μeff of the paramagnetic metal ion. For Tm, an optimal τR of 200 ps increases R1 substantially, particularly at high magnetic field. Unlike for GdIII-based T1 contrast agents (Caravan et al., 1999), there is not much advantage to macromolecular paramagnetic fluorine probes with longer τR.
There are limited examples in the literature of macromolecular fluorinated probes that feature a paramagnetic center. They are mostly used to increase the number of 19F nuclei per unit. For example, the polymeric DyIII-chitosan conjugate, 21 (Figure 4), increases the relaxation rates of the fluorines via the PRE effect (De Luca et al., 2014). As for the small molecules described earlier in this section, the aryl trifluoro groups are advantageously positioned close, but not too close (6.5 Å) from the lanthanide ion. This macromolecular complex features 10 trifluoro groups of equivalent 19F nuclei appended to glycol chitosan, a linear polysaccharide selected for its biocompatibility. The increase in τR from 83 ps to 8 ns due to polymerization of the chitosan was evaluated with GdIII-complexes similarly conjugated to the chitosan monomer (22) and polymer (23) (De Luca et al., 2014). In this case, the R1 of the fluorine nuclei of the Dy chitosan conjugate 21 remained constant before and after polymerization (186 s−1 at 9.4 T), whereas the R2 decreased from 440 to 367 s−1, which is unusual for a polymer (De Luca et al., 2014). Of note, this polymer is a rare example of a metallic fluorine probe that was successfully used in vivo to image tumors in mice. Although a dose of 0.54 g/kg was used in this study—a dose higher than typically used with Gd-based contrast agents—the sensitivity of the probe remained poor and a very long imaging time of 7 h was required to have sufficient SNR (De Luca et al., 2014). Although this probe was more sensitive than small molecule fluorine probes, the very long imaging time remains a significant hurdle to the application of 19F probes.
Figure 4
One should note that although macromolecular paramagnetic fluorine probes are rare, their diamagnetic analogs are more common. Several macromolecular structures based on hyperbranched polymers, dendrimers, or nanoparticles that incorporate a high number of 19F nuclei have been described (Du et al., 2008; Criscione et al., 2009; Jiang et al., 2009; Peng et al., 2009; Ogawa et al., 2010a,b; Thurecht et al., 2010), and recently reviewed (Yu, 2013; Zhu et al., 2013). A macromolecular architecture is more advantageous for diamagnetic fluorine probes than for paramagnetic ones: the increase in τR decreases T1, and, if designed correctly, can increase the solubility of the probe in water as well as its in vivo retention time.
Exploiting the metal ion to design responsive fluorine probes
Applications of the bloch-wangsness-redfield theory to the design of responsive fluorine probes
Advantageously, a paramagnetic metal ion that alters T1 and T2 of the fluorine nuclei can also modulate their chemical shift (19FΔδ). Since variations in each of these three variables can readily be observed both by MRS and by MRI, paramagnetic fluorine complexes offer unique mechanisms for the development of responsive MR probes that often yield much greater response than their purely organic analogs (Yu et al., 2013a). Two different effects of paramagnetic metals can be exploited to design responsive molecular probes. The first is the PRE effect. It is governed by the Bloch-Wangsness-Redfield theory, affects both relaxation times (T1 and T2) and is a function primarily of the nature of the paramagnetic metal ion and the distance d separating it from the fluorine nuclei. The second is the lanthanide induced shift (LIS) effect. It is just as readily be observed with 19F than with 1H nuclei and is governed by the McConnell-Robertson theory. Unlike PRE, LIS is affected not only by the nature of the paramagnetic lanthanide ion and the distance d separating it from the fluorine nuclei, but also by the second order crystal field coefficient of the complex, , and the angle θ between the metal-fluorine vector and the principle magnetic dipolar axis of the lanthanide. Coupling either of these effects with the extensive range of chemical shifts available to 19F offers valuable opportunity to design ratiometric responsive MR probes that can distinguish between changes in signal intensity due to the presence of an analyte vs. that due to a change in the concentration of the probe.
The PRE effect described in the previous section of this review is the basis for the improved sensitivity on a per 19F basis of paramagnetic probes as compared to their organic counterparts. It is the substantial shorter T1 and higher T2/T1 ratio of paramagnetic fluorinated complexes that enables more scans to be acquired in the same amount of time thereby yielding higher SNR both by 19F MRS and MRI. Analysis of the Bloch-Wangsness-Redfield Equations (1–5) indicate that at constant applied magnetic field (the same scanner) and for the same metal ion, T1 and T2 are a function of primarily one parameter: the distance d separating the 19F nuclei from the metal ion. This parameter can be altered significantly by binding to or reacting with a substrate. Herein lies a substantial advantage of paramagnetic metal ions in fluorine probes: they readily enable the design of responsive and ratiometric molecular probes.
The dependence of T1 and T2 on the distance separating the fluorine nuclei from the lanthanide ion has already been applied to the design of responsive paramagnetic fluorine probes. As depicted in Figure 5, this distance can be altered either by reacting with a target or by induced conformational change upon binding to the desired analyte. The former approach yields an irreversible response, although one that can enable the detection of targets such as enzymes that are present at lower concentrations. The latter approach can lead to a reversible response but given the enduring limits in the sensitivity of fluorine probes, it is more appropriate for biological analytes present at substantially higher concentrations such as pH or certain small molecules or metal ions.
Figure 5
Current examples of this class of responsive probes use for the most part GdIII. As described in the previous section, GdIII has a notable effect on the T1 of nearby nuclei, and, importantly, a substantially more pronounced one on T2. Indeed, among the lanthanides, GdIII typically offers the lowest T2/T1 ratio (Table 2). As a result of this very low ratio, the peaks of 19F nuclei near GdIII ions broaden to the point of no longer being observable neither by MRS nor by MRI. Increasing the distance between the 19F nuclei and the GdIII increases T2; this enables the 19F signal to reappear.
A greater effect will be observed if the linker separating the 19F nuclei from the GdIII ion is completely cleaved by the target (Figure 5A). As the fluorinated moiety diffuses away from the GdIII complex, its relaxation times increase by orders of magnitude and, consequently, so does the 19F signal intensity. This approach is particularly well-suited for the detection of enzymes that cleave peptides or sugars. Indeed, the first example reported by Kikuchi and coworkers targeted caspase 3 (24, Figure 6). Adroit positioning of the 19F nuclei on the amino acid substrate of the enzyme that was directly tethered to the GdIII complex resulted in a complete off/on response (Mizukami et al., 2008). This first probe, and its dual 19F MRI/fluorescence analog (25, Figure 6) (Mizukami et al., 2009), exemplified a distinct advantage of 19F molecular probes over both GdIII- and nanoparticle-based responsive contrast agents: a substantial response with zero background such that presence of a signal can only result from co-localization of both the MR probe and the enzyme target. In comparison, 1H MRI contrast agents have to contend with the intrinsic high 1H background signal due to the high concentration of water of the biological milieu. With these agents, an increase in signal intensity does not necessarily correlate with the presence of the target; an identical response can simply result from higher local concentration of the contrast agent. Do note, however, that since such agents are not ratiometric they do not eliminate the possibility of false negative. That is, they cannot distinguish between a lack of signal due to a lack of enzyme as opposed to a lack of 19F probe.
Figure 6
This approach was recently extended by both the Kikuchi and the Engelmann groups to other enzymes, β-galactosidase (26 and 27) (Keliris et al., 2011, 2012; Mizukami et al., 2011), β-lactamase (28) (Matsushita et al., 2012), and matrix metalloprotease-2 (29, Figure 6; Yue et al., 2014, 2017) thereby opening the possibility of directly imaging gene expression by MRI. In each case, hydrolysis of the enzyme's substrate generates a self-immolative linker that releases the fluorinated moiety from the GdIII probe thereby turning on the 19F MR signal. This approach is not limited to enzymes and cleavable linkers. Similar responses can be obtained with an analyte-induced conformational changes that either increase (turn on) or decrease (turn off) the 19F–Gd distance (Figure 5B,C, respectively). Fujimoto and coworkers used this approach to the PRE effect to extend the traditional molecular beacon motif commonly used to detect DNA (or RNA) by luminescence to that by 19F NMR (30, Figure 6) (Sakamoto et al., 2011). Hybridization with the target nucleotide sequence causes the stem loop of the probe to unfold, thereby increasing d and thus also the T2 and signal intensity of the 19F nuclei.
Similar responses can be obtained with paramagnetic transition metals. Yu and Mason reported three fluorinated probes for β-galactosidase that use the same principle but in the opposite direction (31–33, Figure 7) (Yu et al., 2012, 2013b). Hydrolysis of the sugar moiety by β-galactosidase releases a fluorinated FeIII chelator. The T2 of this ligand decreases substantially upon subsequent coordination to the transition metal. As opposed to Kikuchi's and Engelman's examples, in this case the 19F signal of the probe is turned off by the enzyme. The practicality of such probes for in vivo imaging, however, is likely limited given that iron is tightly regulated in the body and is essentially unavailable (Raymond et al., 2003).
Figure 7
The same approach readily leads to the development of fluorine probes for paramagnetic metal ions. An example for MnII is 34 developed by Datta and coworkers (Figure 7B) (Sarkar et al., 2017). Coordination of MnII results in a 101-fold decrease of T2 and, therefore, significant line broadening which decreases the signal intensity of the 19F nuclei.
By extension, this strategy can readily be adjusted to design probes that signal the redox potential of their surroundings. If the metal is chosen correctly, a change in its oxidation state enables it to switch between a diamagnetic and a paramagnetic state (Figure 8). In the paramagnetic state, the metal attenuates the 19F signal by substantially decreasing their T2. Conversion to the diamagnetic state lengthens T2 resulting in a “turn on” response. Early examples that utilize metal redox state to modulate the relaxation of 19F nuclei, and in turn signal intensity, were reported by Chujo's lab (Tanaka et al., 2009; Kitamura et al., 2012). Their probes, Fc-POSS (35) and Cu-POSS (36) contain either a redox sensitive ferrocene or a Cu complex with a bis(2-pyridylmethyl)amino ligand (PMEA) along with trifluoromethyl groups appended to a cubic polyhedral oligomeric silsesquioxane (POSS) for water solubility (Figure 9A). The oxidation of FeII in 35 reduces the 19F signal intensity 3-fold due to the shortened relaxation time. The nitroxyl (HNO) mediated reduction of paramagnetic CuII in 36 to diamagnetic CuI modestly increased T1 (from 1180 ms to 1460 ms) and T2 (from 420 to 540 ms) causing a 3-fold increase in signal intensity. Thus, their work demonstrates the ability to apply PRE to the design of 19F redox sensitive responsive probes. Que's laboratory utilized this strategy and the PRE of CuII to detect cellular hypoxia by 19F MR (Xie et al., 2016). Reduction of CuATSM-F3 (37, Figure 9A) from CuII (d9, paramagnetic) to CuI (d10, diamagnetic) and subsequent ligand dissociation increases T2 of the 19F and restores their signal intensity. A second generation version of this probe (38) was designed to be more sensitive (Xie et al., 2017). It includes a higher density of 19F nuclei, a polyethylene glycol linker that increases the hydrophilicity of the probe and positions the 19F nuclei at an optimal distance from the copper ion. With a 19F–Cu distance of ~ 18 Å, the 19F signal is attenuated but remains visible by NMR when the probe is in the CuII state. Reduction to CuI generates a sharp signal which is 6-fold more intense, leading to a 4.5-fold increase in SNR in the 19F MRI images obtained at 7 T. The large differences in relaxation times between the two species allow them to be independently imaged using short (TE = 10 ms for CuII) or long (TE = 100 ms for CuI) echo pulse sequences (Xie et al., 2017).
Figure 8
Figure 9
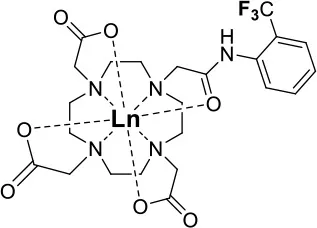
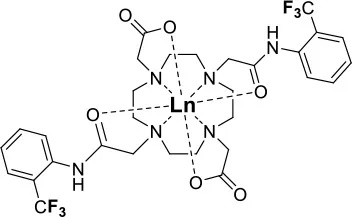
Allen's laboratory used an analogous approach with Eu in the design of their temperature and redox responsive 19F probe (39, Figure 9A). This Eu-DOTAm (1,4,7,10-tetrakis(carbamoylmethyl)-1,4,7,10-tetraazacyclododecane) complex with a p-trifluoromethylphenyl arm, has 12 equivalent fluorine nuclei (Basal et al., 2017). In its +2 oxidation state, Eu significantly shortens the T2 of the 19F and hence quenches their signal. In its +3 oxidation state, EuIII has a much lower μeff (3.40–3.51 B.M.) than EuII (7.6–8.0 B.M.; Tilley et al., 1980; Garcia and Allen, 2012) (Table 1) and thus a limited effect on the relaxation times of the fluorine which consequently reappear. In 19F MRI images, oxidation of EuII to EuIII results in a 6-fold increase in SNR. This responsive probe was successfully used to image non-hypoxic regions in vivo by 19F MRI.
This approach also enables direct detection of reactive oxygen species. Oxidation of Co-NODA-CF3 (2,2′-(7-(2-oxo-2-((2,2,2-trifluoroethyl)amino)ethyl)-1,4,7-triazonane-1,4-diyl)diacetic acid) (40, Figure 9A) with H2O2 from paramagnetic CoII (d7) to diamagnetic CoIII (d6) shifts the 19F signal by 3.6 ppm and increases the T1 and T2 of the 19F nuclei 60- and 121-fold, respectively (Yu et al., 2016). This, in turn, results in a 23-fold increase in signal intensity in spin-density weighted 19F MR images. Although the oxidation of the probe is reversible, the long (2 h) reaction time and the low sensitivity of the probe compared to in vivo levels of H2O2 still limit the application of this probe to in vivo imaging.
There are two main disadvantages to using paramagnetic metals that primarily affect T2 in the design of responsive fluorine probes. The first, as mentioned above, is that such probes are not ratiometric. A lack of signal can be attributed both to a lack of target and to a lack of probe. Similarly, signal intensity is not a direct measure of the concentration of the target as it is equally affected by the concentration of the probe. The second disadvantage is that upon release, the organic fluorine moiety regains its very long T1. Such probes are thus plagued by the same sensitivity issue as the purely organic fluorinated probes (Yu et al., 2005, 2013a). Their T1 of 0.4–5 s are10- to 500-fold longer than their paramagnetic counterparts, which greatly limits the number of scans that can be acquired per amount of time. The low sensitivity of such probes practically restricts the markers that they could measure in vivo. In theory, both of these problems can be resolved with the use of a paramagnetic ion that has a more limited effect on T2 and maintain as high a T2/T1 ratio as possible. FeII, TmIII, and HoIII are particularly well-suited for this approach (Srivastava et al., 2017b), but examples are not represented in the literature.
Applications of the mcconnell-robertson theory to the design of responsive fluorine probes
One of the first applications of lanthanides in NMR spectroscopy was as induced shift reagents (Sanders et al., 1972). This application has surprisingly rarely been applied to 19F NMR spectroscopy. Yet the same theory and equations developed for 1H apply to 19F and as such they can readily be exploited for the rational design of paramagnetic 19F shift reagents. Lanthanide induced shift (LIS) is traditionally defined as the difference in chemical shift, Δδ, between a paramagnetic complex and its diamagnetic analog (Δδ = δpara – δdia). In terms of responsive paramagnetic fluorine probes, the induced shift, 19FLIS, is defined as the difference between the chemical shift of a 19F nuclei in the presence and absence of the probe's analyte (Equation 6).
This shift is the sum of the contributions from the contact shift, δc, and the pseudocontact shift, δpc (Peters et al., 1996; Allegrozzi et al., 2000; Harvey et al., 2012b). If the fluorine nuclei is kept 5 Å or more from the paramagnetic metal center, the contribution from the contact shift is negligible, and the observed 19FLIS is primarily due to the pseudocontact shift. This pseudocontact shift, δpc, is the result of the dipolar interaction between the 19F nuclei and the unpaired electrons on the metal ion that experiences a Curie magnetization in the applied magnetic field. Thus, an anisotropic metal ion can amplify the chemical shift inequivalence between a fluorine nucleus in the presence and absence of the probe's analyte, resulting in greater resonance shift that are ultimately necessary for each nuclei to be imaged independently by MRI. Under the condition that the 19F nuclei is at least 5 Å away from the metal ion, the pseudocontact shift is a function of the nature of the metal, its coordination environment, and the metal ion (Figure 10). This relationship is defined by the McConnell-Robertson equation (Equation 7) (Harvey et al., 2012b)
Figure 10
Where d is the 19F–metal distance, θ is the angle between the principal magnetic dipolar axis of the metal ion and 19F nuclei, is the second order crystal field coefficient that is dependent on the coordination environment of the metal ion, and Cj is the Bleaney coefficient for the specific metal. Both the direction and magnitude of the shift are dependent on the identity of the metal ion as denoted by its Bleaney coefficient (Table 1). One can thus rank metals by relative pseudocontact shift (PCS) strength, which in turn indicates their efficacy as 19FLIS agents. DyIII and TbIII are undeniably excellent shift agents. However, given the necessity to maintain as high a T2/T1 ratio as possible to maximize 19F sensitivity (see above), TmIII and HoIII are the best choices in the design of 19FLIS probes for MRI.
According to the McConnell-Robertson equation, the design of responsive fluorinated lanthanide MR probes can be based on three parameters: the distance separating the 19F nuclei from the metal ion, d (Figure 5), the angle θ between the main dipolar magnetic axis of the metal and the metal—19F vector (Figure 11A), and the second order crystal field coefficient, , which is a function of the direct coordination environment of the metal (Figures 11B,C).
Figure 11
The second order crystal field coefficient, , which is a function of the direct coordination environment of the lanthanide or transition metal, has been used in the design of responsive fluorine probes. Advantageously, this approach enables the use of ligands of similar chemical structure as those used with gadolinium-based responsive contrast agents. Swapping GdIII for TmIII, HoIII, or DyIII and adding a fluorinated moiety within 4–7 Å of the metal is often all that is necessary. For example, the Parker group developed citrate-responsive 19F probes, which are essentially fluorinated versions of Ln-DO3A (43 and 44, Figure 12A) (Harvey et al., 2012a). Coordination of citrate to a TmIII complex results in the displacement of the two inner-sphere water molecules; the resulting change in causes a 19FLIS of 4.7 ppm. Such 19F probes are unfortunately marked with the same selectivity problem as the gadolinium responsive contrast agents that function by varying the number of inner-sphere water molecules (q-based responsive contrast agents) (Pierre et al., 2018). The selectivity of 43 and 44 for citrate over other hard anions that also have good affinity for the oxophilic lanthanides, such as bicarbonate, phosphate and lactate, is rather poor. The efficacy of the probe in blood and in cells is thus likely limited.
Figure 12
The same approach was used to design the CaII responsive fluorine probes 45 and 46 (Figure 12A). These probes are derivatives of a GdIII-based responsive contrast agent which displays a change in longitudinal proton relaxivity, r1, in the presence of calcium (Harvey et al., 2012a). For 45 and 46 to bind CaII, one of the amide arms of the macrocycle must release the lanthanide. In doing so, it is replaced by a water molecule. The resulting change in causes a 19FLIS of 3.0 or 3.9 ppm depending on the lanthanide ion (DyIII or TmIII, respectively) (Harvey et al., 2012a). Seemingly minor modifications on the structure of the probe can thus result in a substantial difference in its response.
The Parker group used a similar approach to develop four pH responsive probes. In 47 and 48 (Figure 12B) (Senanayake et al., 2007; Kenwright et al., 2008), protonation of a sulfonamide group coordinating the lanthanide at pH <5 breaks the N-Ln bond, which opens up a coordination site for a water molecule. The resulting change in due to the differing coordination environment of the lanthanide ion, and in the angle θ due to conformational change through rotation of the arm, cause a 19FLIS of −6 and +2 ppm between pH 8.5 and 4.2 for the two inequivalent 19F in the EuIII complex 47. The 19F resonance of the related Ho probe, 48, shifts upfield by 40 ppm upon deprotonation (Chalmers et al., 2010). Note the greater 19FLIS for HoIII, which has a Bleany's coefficient of greater magnitude, than for EuIII.
The other two pH probes, 49 and 50, also rely on protonation of a coordinating arm, but in these cases the aryl amide does not release the lanthanide at acidic pH (Figure 12B) (Kenwright et al., 2008; Chalmers et al., 2010). Instead, deprotonation of the amide changes both and θ between the Ln-19F vector and the principal magnetic dipolar axis of the LnIII ion. The latter is due to the differing conformation of the arm. The combination of these two effects result in a substantial 19FLIS of 18 and 23 ppm for 49 and 50, respectively.
The chemical shift of fluorine nuclei can also be modulated by altering the spin state of the metal ion. A recent example exploits the temperature induced transition of FeII from low spin (S = 0) to high spin (S = 2) in hexadentate triazacyclonone ligands with fluorinated 2-picolyl substituents (41 and 42, Figure 9B) to image temperature by 19F MRI (Thorarinsdottir et al., 2017). In these probes, the 19F are positioned at an optimal distance away from the FeII center so as to maximize 19FΔδ. Increasing the temperature from 4 to 60°C, causes the 19F resonances of 41 to shift downfield 28.3–24.6 ppm. The single resonance of 42 instead shifts 12 ppm upfield. On a per °C basis, 41 is therefore a more sensitive probe than 42. Importantly, the response of these two probes to temperature is nearly identical in water and fetal bovine serum (FBS), which bodes well for future in vivo applications.
The standard approaches that are used in the design of diamagnetic fluorine probes can also be applied to their paramagnetic analogs. An example is the α-chymotrypsin esterase probe 51 (Figure 12C). Hydrolysis of the ester, followed by self-immolation of the linker, yields a carboxylated fluorine moiety whose 19F nuclei signal is positioned 6.2 ppm away from the starting material (Harvey et al., 2012a). These probes function essentially like their purely organic analogs recently reviewed by Mason (Yu et al., 2005). The advantage of the paramagnetic version resides in the sensitivity of the probe (see section Improving the Sensitivity of 19F Probes). In this case, the lanthanide ion functions only to increase the sensitivity of the probe. HoIII shortens the T1 and T2 of the fluorine, thereby significantly increasing the number of scans that can be recorded in a given amount of time, and thus improving the SNR of the probe.
Although this review has focused on 19F MR imaging, a significant advantage of 19F probes is their ability to also function in other modalities. Fluorinated polyaminocarboxylate-based complexes, for instance, can also be effective chemical exchange saturation transfer (CEST) MR contrast agents. Such dual-modalities are particularly well-suited for the design of ratiometric agents. False negatives are more readily avoided if one of the two modalities is responsive to the targeted marker while the other one is not. This enables the distribution of the contrast agent to be mapped independently of that of the marker. Two examples of dual 19F-CEST agents have been reported. A fluorinated Eu-DOTAm-Gly complex with two trans trifluoromethyl groups (2, Figure 1) generated sufficient 19F and CEST contrast in vitro despite multiple isomers in solution (Cakic et al., 2016). Unfortunately, the contrast agent was not detected in ex vivo by 19F MRI, possibly due to interactions with tissues that substantially reduced T2. This first dual-modality 19F contrast agent is not responsive.
Fe-DOTAm-F12 which was developed in our lab, has 12 equivalent fluorine nuclei and one isomer in solution (12, Figure 1). It functions both as a 19F MR probe and a paraCEST contrast agent. Importantly, whereas the 19F signal intensity is independent of pH, the CEST signal is modulated by pH. The % saturation transfer of the complexes increases 5-fold between pH 4 and 6.2. The complex accurately determines the pH independently of the concentration of the contrast agent between pH 6.9 and 7.4, a range that is relevant to cancer diagnosis. Advantageously, the similar sensitivity of the agent in both modalities facilitates ratiometric determination of pH.
Outlook
The development of paramagnetic fluorine probes over the last decade mirrors, in many ways, that of paraCEST agents, the paramagnetic derivatives of CEST contrast agents. The introduction of paramagnetic metal ions in the design of fluorine probes, most notably lanthanides, iron, and cobalt, offer several advantages. Those include increased sensitivity of the fluorine probes due to decreased R1 of the fluorine nuclei and increased chemical shift spectral window due to the paramagnetic induced shift.
Advantageously, both the effects on relaxation rates and chemical shift offer new approaches to the development of responsive fluorine probes that are not available with their diamagnetic counterparts, and these strategies lead to a significantly greater response. Indeed, paramagnetic fluorinated complexes can be readily designed such that the chemical shifts and/or the relaxation times T1 and T2 of the fluorine nuclei change substantially upon binding to or reacting with a desired analyte. This causes either the appearance, disappearance or notable chemical shift of the 19F peaks. Moreover, the absence of background signal in 19F MRI is an added advantage of this class of probes over the more common Gd-based and iron oxide nanoparticle-based contrast agents which have to contend with the signal of endogenous water. The recent work published on paramagnetic fluorine probes, backed by accurate theoretical models, enable scientists to design molecules with maximum sensitivity by optimizing the nature, specially the oxidation and spin state, of the paramagnetic ion and its distance from the fluorine nuclei all while maximizing the number of chemically equivalent 19F nuclei. Those same parameters are used in the design and optimization of responsive probes. Notably, the large spectral windows of paramagnetic fluorine probes enable the design of ratiometric responsive probes which can independently report on both the distribution of a probe and that of its targeted analyte. Paramagnetic fluorine probes are also uniquely suited to multicolor, or multifrequency, imaging, which enables the tracking of different types of cells.
Paramagnetic fluorine probes have drawbacks too that should be kept in mind. The increase in sensitivity due to lower R1 induced by the paramagnetic ion is minimal at high magnetic fields. These probes are better suited for use with scanners of low and medium magnetic field strengths, below 6 T. The structure and conformation of the probe in solution, parameters that can be more difficult to predict, have a significant impact on the properties of the probe. Complexes existing as a single isomer in solution with all fluorine nuclei chemically equivalent are more sensitive and easier to image, but many lanthanide complexes exist in solution as interconverting isomers. The potential toxicity of the complex should also be kept in mind, especially given the high concentrations of probes required for in vivo imaging. Although the toxicity and pharmacological properties of this new class of probes has not yet been evaluated, conclusions drawn from Gd-based contrast agents are likely to extend to paramagnetic fluorine probes. Kinetically labile lanthanide complexes, for instance, are expected to present higher toxicity. Nonetheless, we foresee that these first generation of paramagnetic fluorine probes will open avenues for many more applications for in vivo imaging. In particular, the ability of these probes to track different cells and biomarkers, and their response to different stimuli, render them particularly promising for molecular imaging.
Statements
Author contributions
VP, KP, and KS: searched and analyzed the literature; VP, KP, and KS: co-wrote the manuscript.
Acknowledgments
This work was supported by the National Science Foundation Grant CAREER 1151665 and NSF INFEWS N/P/H2O:SusChEM CHE-1610832.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1
AhrensE. T.HelferB. M.O'HanlonC. F.SchirdaC. (2014). Clinical cell therapy imaging using a perfluorocarbon tracer and fluorine-19 MRI. Magn. Reson. Med.72, 1696–1701. 10.1002/mrm.25454
2
AhrensE. T.ZhongJ. (2013). In vivo MRI cell tracking using perfluorocarbon probes and fluorine-19 detection. NMR Biomed.26, 860–871. 10.1002/nbm.2948
3
AllegrozziM.BertiniI.JanikM. B. L.LeeY. M.LinG. H.LuchinatC. (2000). Lanthanide-induced pseudocontact shifts for solution structure refinements of macromolecules in shells up to 40 angstrom from the metal ion. J. Am. Chem. Soc.122, 4154–4161. 10.1021/ja993691b
4
AlsaadiB. M.RossottiF. J. C.WilliamsR. J. P. (1980). Hydration of complexone complexes of lanthanide cations. J. Chem. Soc. Dalton Trans. 2151–2154. 10.1039/dt9800002151
5
BasalL. A.BaileyM. D.RomeroJ.AliM.KurenbekovaL.YusteinJ.et al. (2017). Fluorinated EuII-based multimodal contrast agent for temperature- and redox-responsive magnetic resonance imaging. Chem. Sci.8, 8345–8350. 10.1039/C7SC03142D
6
BenetolloF.BombieriG.CalabiL.AimeS.BottaM. (2003). Structural variations across the lanthanide series of macrocyclic DOTA complexes: insights into the design of contrast agents for magnetic resonance imaging. Inorg. Chem.42, 148–157. 10.1021/ic025790n
7
BertiniI.CapozziF.LuchinatC.NicastroG.XiaZ. C. (1993a). Water proton relaxation for some lanthanide aqua ions in solution. J. Phys. Chem.97, 6351–6354.
8
BertiniI.TuranoP.VilaA. J. (1993b). Nuclear Magnetic resonance of paramagnetic metalloproteins. Chem. Rev.93, 2833–2932.
9
BlahutJ.BernášekK.GálisováA.HerynekV.CísarováI.KotekJ.et al. (2017). Paramagnetic 19F relaxation enhancement in nickel(II) complexes of N-trifluoroethyl cyclam derivatives and cell labeling for 19F MRI. Inorg. Chem.56, 13337–13348. 10.1021/acs.inorgchem.7b02119
10
BlahutJ.HermannP.GalisovaA.HerynekV.CisarovaI.TosnerZ.et al. (2016). Nickel(ii) complexes of N-CH2CF3 cyclam derivatives as contrast agents for 19F magnetic resonance imaging. Dalton Trans.45, 474–478. 10.1039/C5DT04138D
11
BleaneyB. (1972). Nuclear magnetic resonance shifts in solution due to lanthanide ions. J. Magn. Reson.8, 91–100. 10.1016/0022-2364(72)90027-3
12
BuraiL.TóthÉ.MoreauG.SourA.ScopellitiR.MerbachA. E. (2003). Novel macrocyclic EuII complexes: fast water exchange related to an extreme M?Owater distance. Chem. Eur. J.9, 1394–1404. 10.1002/chem.200390159
13
CakicN.SavicT.Stricker-ShaverJ.TruffaultV.Platas-IglesiasC.MirkesC.et al. (2016). Paramagnetic lanthanide chelates for multicontrast MRI. Chem. Commun.52, 9224–9227. 10.1039/C6CC04011J
14
CaravanP.EllisonJ. J.McMurryT. J.LaufferR. B. (1999). Gadolinium(III) chelates as MRI contrast agents: structure, dynamics, and applications. Chem. Rev.99, 2293–2352. 10.1021/cr980440x
15
ChalmersK. H.BottaM.ParkerD. (2011a). Strategies to enhance signal intensity with paramagnetic fluorine-labelled lanthanide complexes as probes for F-19 magnetic resonance. Dalton Trans.40, 904–913. 10.1039/C0DT01232G
16
ChalmersK. H.De LucaE.HoggN. H. M.KenwrightA. M.KuprovI.ParkerD.et al. (2010). Design principles and theory of paramagnetic fluorine-labelled lanthanide complexes as probes for F-19 magnetic resonance: a proof-of-concept study. Chem. Eur. J.16, 134–148. 10.1002/chem.200902300
17
ChalmersK. H.KenwrightA. M.ParkerD.BlamireA. M. (2011b). 19F-lanthanide complexes with increased sensitivity for 19F-MRI: optimization of the MR acquisition. Magn. Reson. Med.66, 931–936. 10.1002/mrm.22881
18
ChangE. T.AdamiH.-O.BoffettaP.ColeP.StarrT. B.MandelJ. S. (2014). A critical review of perfluorooctanoate and perfluorooctanesulfonate exposure and cancer risk in humans. Crit. Rev. Toxicol.44, 1–81. 10.3109/10408444.2014.905767
19
CriscioneJ. M.LeB. L.SternE.BrennanM.RahnerC.PapademetrisX.et al. (2009). Self-assembly of pH-responsive fluorinated dendrimer-based particulates for drug delivery and noninvasive imaging. Biomaterials30, 3946–3955. 10.1016/j.biomaterials.2009.04.014
20
DaviesG.-L.BrownA.BlackburnO.TropianoM.FaulknerS.BeerP. D.et al. (2015). Ligation driven 19F relaxation enhancement in self-assembled Ln(iii) complexes. Chem. Commun.51, 2918–2920. 10.1039/C4CC09952D
21
De LucaE.HarveyP.ChalmersK.MishraA.SenanayakeP. K.WilsonJ. I.et al. (2014). Characterisation and evaluation of paramagnetic fluorine labelled glycol chitosan conjugates for 19F and 1H magnetic resonance imaging. J. Biol. Inorg. Chem.19, 215–227. 10.1007/s00775-013-1028-y
22
de VriesA.MoonenR.YildirimM.LangereisS.LamerichsR.PikkemaatJ. A.et al. (2014). Relaxometric studies of gadolinium-functionalized perfluorocarbon nanoparticles for MR imaging. Contrast Media Mol. Imaging9, 83–91. 10.1002/cmmi.1541
23
DuW.NystromA. M.ZhangL.PowellK. T.LiY.ChengC.et al. (2008). Amphiphilic hyperbranched fluoropolymers as nanoscopic (19)F magnetic resonance imaging agent assemblies. Biomacromolecules9, 2826–2833. 10.1021/bm800595b
24
GarciaJ.AllenM. J. (2012). Developments in the coordination chemistry of europium(II). Eur. J. Inorg. Chem.2012, 4550–4563. 10.1002/ejic.201200159
25
GyslingH.TsutsuiM. (1971). Organolanthanides and organoactinides. Adv. Organomet. Chem.9, 361–395. 10.1016/S0065-3055(08)60054-4
26
HarveyP.ChalmersK. H.De LucaE.MishraA.ParkerD. (2012a). Paramagnetic 19F chemical shift probes that respond selectively to calcium or citrate levels and signal ester hydrolysis. Chem. Eur. J.18, 8748–8757. 10.1002/chem.201200737
27
HarveyP.KuprovI.ParkerD. (2012b). Lanthanide complexes as paramagnetic probes for 19F magnetic resonance. Eur. J. Inorg. Chem.2012, 2015–2022. 10.1002/ejic.201100894
28
HelmL.MorrowJ. R.BondC. J.CarniatoF.BottaM.BraunM.et al. (2018). Chapter 2: Gadolinium-based contrast agents, in Contrast Agents for MRI: Experimental Methods, eds PierreV. C.AllenM. J. (Croydon: The Royal Society of Chemistry), 121–242.
29
HollandG. N.BottomleyP. A.HinshawW. S. (1977). 19F magnetic resonance imaging. J. Magn. Reson.28, 133–136. 10.1016/0022-2364(77)90263-3
30
HuL.ZhangL.ChenJ.LanzaG. M.WicklineS. A. (2011). Diffusional mechanisms augment the fluorine MR relaxation in paramagnetic perfluorocarbon nanoparticles that provides a “relaxation switch” for detecting cellular endosomal activation. J. Magn. Reson. Imaging34, 653–661. 10.1002/jmri.22656
31
JanjicJ. M.AhrensE. T. (2009). Fluorine-containing nanoemulsions for MRI cell tracking. Wiley Interdiscipl. Rev. Nanomed. Nanobiotechnol.1, 492–501. 10.1002/wnan.35
32
JanjicJ. M.SrinivasM.KadayakkaraD. K. K.AhrensE. T. (2008). Self-delivering nanoemulsions for dual fluorine-19 MRI and fluorescence detection. J. Am. Chem. Soc.130, 2832–2841. 10.1021/ja077388j
33
JiangZ.-X.FengY.YuY. B. (2011). Fluorinated paramagnetic chelates as potential multi-chromic 19F tracer agents. Chem. Commun.47, 7233–7235. 10.1039/c1cc11150g
34
JiangZ.-X.LiuX.JeongE.-K.YuY. B. (2009). Symmetry-guided design and fluorous synthesis of a stable and rapidly excreted imaging tracer for (19)F MRI. Angew. Chem. Int. Ed.48, 4755–4758. 10.1002/anie.200901005
35
KamendulisL. M.WuQ.SanduskyG. E.HocevarB. A. (2014). Perfluorooctanoic acid exposure triggers oxidative stress in the mouse pancreas. Toxicol. Rep.1, 513–521. 10.1016/j.toxrep.2014.07.015
36
KelirisA.MamedovI.HagbergG. E.LogothetisN. K.SchefflerK.EngelmannJ. (2012). A smart 19F and 1H MRI probe with self-immolative linker as a versatile tool for detection of enzymes. Contrast Media Mol. Imaging7, 478–483. 10.1002/cmmi.1470
37
KelirisA.ZieglerT.MishraR.PohmannR.SauerM. G.UgurbilK.et al. (2011). Synthesis and characterization of a cell-permeable bimodal contrast agent targeting beta-galactosidase. Bioorg. Med. Chem.19, 2529–2540. 10.1016/j.bmc.2011.03.023
38
KenwrightA. M.KuprovI.De LucaE.ParkerD.PandyaS. U.SenanayakeP. K.et al. (2008). (19)F NMR based pH probes: lanthanide(III) complexes with pH-sensitive chemical shifts. Chem. Commun. 2514–2516. 10.1039/b802838a
39
KimW. D.KieferG. E.HuskensJ.SherryA. D. (1997). NMR studies of the lanthanide(III) complexes of 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetrakis(methanephosphonic acid mono(2′,2′,2′-trifluoroethyl) ester). Inorg. Chem.36, 4128–4134. 10.1021/ic970169m
40
KislukhinA. A.XuH.AdamsS. R.NarsinhK. H.TsienR. Y.AhrensE. T. (2016). Paramagnetic fluorinated nanoemulsions for sensitive cellular fluorine-19 magnetic resonance imaging. Nat. Mater.15, 662–668. 10.1038/nmat4585
41
KitamuraN.HiraokaT.TanakaK.ChujoY. (2012). Reduced glutathione-resisting 19F NMR sensors for detecting HNO. Bioorg. Med. Chem.20, 4668–4674. 10.1016/j.bmc.2012.06.013
42
KnightJ. C.EdwardsP. G.PaiseyS. J. (2011). Fluorinated contrast agents for magnetic resonance imaging; a review of recent developments. RSC Adv.1, 1415–1425. 10.1039/c1ra00627d
43
MatsushitaH.MizukamiS.MoriY.SugiharaF.ShirakawaM.YoshiokaY.et al. (2012). F-19 MRI monitoring of gene expression in living cells through cell-surface beta-lactamase activity. ChemBioChem13, 1579–1583. 10.1002/cbic.201200331
44
McFarlandE.KoutcherJ. A.RosenB. R.TeicherB.BradyT. J. (1985). In vivo 19F NMR imaging. J. Comput. Assist. Tomogr.9, 8–15. 10.1097/00004728-198501000-00002
45
MizukamiS.MatsushitaH.TakikawaR.SugiharaF.ShirakawaM.KikuchiK. (2011). 19F MRI detection of [small beta]-galactosidase activity for imaging of gene expression. Chem. Sci.2, 1151–1155. 10.1039/c1sc00071c
46
MizukamiS.TakikawaR.SugiharaF.HoriY.TochioH.WälchliM.et al. (2008). Paramagnetic relaxation-based 19F MRI probe to detect protease activity. J. Am. Chem. Soc.130, 794–795. 10.1021/ja077058z
47
MizukamiS.TakikawaR.SugiharaF.ShirakawaM.KikuchiK. (2009). Dual-function probe to detect protease activity for fluorescence measurement and 19F MRI. Angew. Chem. Int. Ed.48, 3641–3643. 10.1002/anie.200806328
48
ModoM. M. J.BulteJ. W. M. (2007). Molecular and Cellular MR Imaging.Boca Raton, FL: CRC Press.
49
NeubauerA. M.MyersonJ.CaruthersS. D.HockettF. D.WinterP. M.ChenJ.et al. (2008). Gadolinium-modulated 19F signals from perfluorocarbon nanoparticles as a new strategy for molecular imaging. Magn. Reson. Med.60, 1066–1072. 10.1002/mrm.21750
50
OgawaM.NitaharaS.AokiH.ItoS.NarazakiM.MatsudaT. (2010a). Fluorinated polymer nanoparticles as a Novel F-19 MRI contrast agent prepared by dendrimer-initiated living radical polymerization. Macromol. Chem. Phys.211, 1369–1376. 10.1002/macp.200900672
51
OgawaM.NitaharaS.AokiH.ItoS.NarazakiM.MatsudaT. (2010b). Synthesis and evaluation of water-soluble fluorinated dendritic block-copolymer nanoparticles as a F-19-MRI contrast agent. Macromol. Chem. Phys.211, 1602–1609. 10.1002/macp.201000100
52
ParkerD.DickinsR. S.PuschmannH.CrosslandC.HowardJ. A. K. (2002). Being excited by lanthanide coordination complexes: aqua species, chirality, excited-state chemistry, and exchange dynamics. Chem. Rev.102, 1977–2010. 10.1021/cr010452+
53
PengH.BlakeyI.DargavilleB.RasoulF.RoseS.WhittakerA. K. (2009). Synthesis and evaluation of partly fluorinated block copolymers as MRI imaging agents. Biomacromolecules10, 374–381. 10.1021/bm801136m
54
PetersJ. A.HuskensJ.RaberD. J. (1996). Lanthanide induced shifts and relaxation rate enhancements. Prog. Nucl. Magn. Reson. Spectrosc.28, 283–350. 10.1016/0079-6565(95)01026-2
55
PierreV. C.HarrisS. M.PaillouxS. L. (2018). Comparing strategies in the design of responsive contrast agents for magnetic resonance imaging: a case study with copper and zinc. Acc. Chem. Res. 51, 342–351. 10.1021/acs.accounts.7b00301
56
PlacidiM. P.BottaM.KálmánF. K.HagbergG. E.BaranyaiZ.KrenzerA.et al. (2013). Aryl-phosphonate lanthanide complexes and their fluorinated derivatives: investigation of their unusual relaxometric behavior and potential application as dual frequency 1H/19F MRI probes. Chem. Eur. J.19, 11644–11660. 10.1002/chem.201300763
57
RaymondK. N.DertzE. A.KimS. S. (2003). Enterobactin: an archetype for microbial iron transport. Proc. Natl. Acad. Sci. U.S.A.100, 3584–3588. 10.1073/pnas.0630018100
58
Ruiz-CabelloJ.BarnettB. P.BottomleyP. A.BulteJ. W. M. (2011). Fluorine (F-19) MRS and MRI in biomedicine. NMR Biomed.24, 114–129. 10.1002/nbm.1570
59
SakamotoT.ShimizuY. K.SasakiJ.HayakawaH.FujimotoK. (2011). Signal turn-on probe for nucleic acid detection based on 19F nuclear magnetic resonance. Bioorg. Med. Chem. Lett.21, 303–306. 10.1016/j.bmcl.2010.11.013
60
SandersJ. K. M.HansonS. W.WilliamsD. H. (1972). Paramagnetic shift reagents. Nature of the interactions. J. Am. Chem. Soc.94, 5325–5335. 10.1021/ja00770a031
61
SarkarA.BitonI. E.NeemanM.DattaA. (2017). A macrocyclic F-19-MR based probe for Mn2+ sensing. Inorg. Chem. Commun.78, 21–24. 10.1016/j.inoche.2017.02.008
62
SenanayakeP. K.KenwrightA. M.ParkerD.van der HoornS. K. (2007). Responsive fluorinated lanthanide probes for 19F magnetic resonance spectroscopy. Chem. Commun. 2923–2925. 10.1039/b705844f
63
SrivastavaK.FerrautoG.YoungV. G.AimeS.PierreV. C. (2017a). Eight-coordinate, stable Fe(II) complex as a dual 19F and CEST contrast agent for ratiometric pH imaging. Inorg. Chem.56, 12206–12213. 10.1021/acs.inorgchem.7b01629
64
SrivastavaK.WeitzE. A.PetersonK. L.MarjanskaM.PierreV. C. (2017b). Fe- and Ln-DOTAm-F12 are effective paramagnetic fluorine contrast agents for MRI in water and blood. Inorg. Chem.56, 1546–1557. 10.1021/acs.inorgchem.6b02631
65
StaresE.RhoJ.AhrensE. T.FosterP.LiA.BarthaR. (2018). Chapter 6: Fluorine-based contrast agents, in Contrast Agents for MRI: Experimental Methods, eds PierreV. C.AllenM. J. (Croydon: The Royal Society of Chemistry), 479–498.
66
TanakaK.KitamuraN.TakahashiY.ChujoY. (2009). Reversible signal regulation system of 19F NMR by redox reactions using a metal complex as a switching module. Bioorg. Med. Chem.17, 3818–3823. 10.1016/j.bmc.2009.04.039
67
ThorarinsdottirA. E.GaudetteA. I.HarrisT. D. (2017). Spin-crossover and high-spin iron(ii) complexes as chemical shift 19F magnetic resonance thermometers. Chem. Sci.8, 2448–2456. 10.1039/C6SC04287B
68
ThurechtK. J.BlakeyI.PengH.SquiresO.HsuS.AlexanderC.et al. (2010). Functional hyperbranched polymers: toward targeted in vivo 19F magnetic resonance imaging using designed macromolecules. J. Am. Chem. Soc.132, 5336–5337. 10.1021/ja100252y
69
TilleyT. D.AndersenR. A.SpencerB.RubenH.ZalkinA.TempletonD. H. (1980). Divalent lanthanide chemistry. Bis (pentamethylcyclopentadienyl) europium(II) and -ytterbium(II) derivatives: crystal structure of bis (pentamethylcyclopentadienyl) (tetrahydrofuran ytterbium(II) -hemitoluene at 176 K. Inorg. Chem.19, 2999–3003.
70
TirottaI.DichiaranteV.PigliacelliC.CavalloG.TerraneoG.BombelliF. B.et al. (2015). 19F magnetic resonance imaging (MRI): from design of materials to clinical applications. Chem. Rev.115, 1106–1129. 10.1021/cr500286d
71
XieD.KimS.KohliV.BanerjeeA.YuM.EnriquezJ. S.et al. (2017). Hypoxia-responsive F-19 MRI probes with improved redox properties and biocompatibility. Inorg. Chem.56, 6429–6437. 10.1021/acs.inorgchem.7b00500
72
XieD.KingT. L.BanerjeeA.KohliV.QueE. L. (2016). Exploiting copper redox for 19F magnetic resonance-based detection of cellular hypoxia. J. Am. Chem. Soc.138, 2937–2940. 10.1021/jacs.5b13215
73
YuJ.-X.HallacR. R.ChiguruS.MasonR. P. (2013a). New frontiers and developing applications in F-19 NMR. Prog. Nucl. Magn. Reson. Spectrosc.70, 25–49. 10.1016/j.pnmrs.2012.10.001
74
YuJ. X.KodibagkarV. D.CuiW. N.MasonR. P. (2005). F-19: a versatile reporter for non-invasive physiology and pharmacology using magnetic resonance. Curr. Med. Chem.12, 819–848. 10.2174/0929867053507342
75
YuJ.-X.KodibagkarV. D.HallacR. R.LiuL.MasonR. P. (2012). Dual 19F/1H MR gene reporter molecules for in vivo detection of β-galactosidase. Bioconjugate Chem.23, 596–603. 10.1021/bc200647q
76
YuJ.-X.KodibagkarV. D.LiuL.ZhangZ.LiuL.MagnussonJ.et al. (2013b). 19F-MRS/1H-MRI dual-function probe for detection of [small beta]-galactosidase activity. Chem. Sci.4, 2132–2142. 10.1039/C3SC21099E
77
YuM.XieD.PhanK. P.EnriquezJ. S.LuciJ. J.QueE. L. (2016). A CoII complex for 19F MRI-based detection of reactive oxygen species. Chem. Commun.52, 13885–13888. 10.1039/C6CC08207F
78
YuY. B. (2013). Fluorinated dendrimers as imaging agents for F-19 MRI. Wiley Interdiscipl. Rev. Nanomed. Nanobiotechnol.5, 646–661. 10.1002/wnan.1239
79
YueX.WangZ.ZhuL.WangY.QianC.MaY.et al. (2014). Novel 19F activatable probe for the detection of matrix metalloprotease-2 activity by MRI/MRS. Mol. Pharm.11, 4208–4217. 10.1021/mp500443x
80
YueX.WangZ.ZhuL.WangY.QianC.MaY.et al. (2017). Correction to “Novel 19F activatable probe for the detection of matrix metalloprotease-2 activity by MRI/MRS. Mol. Pharm.14, 1317–1318. 10.1021/acs.molpharmaceut.7b00150
81
ZhuQ.QiuF.ZhuB.ZhuX. (2013). Hyperbranched polymers for bioimaging. RSC Adv.3, 2071–2083. 10.1039/C2RA22210H
Summary
Keywords
fluorine, lanthanide, iron, magnetic resonance imaging, magnetic resonance spectroscopy, contrast agent, molecular probe, responsive probe
Citation
Peterson KL, Srivastava K and Pierre VC (2018) Fluorinated Paramagnetic Complexes: Sensitive and Responsive Probes for Magnetic Resonance Spectroscopy and Imaging. Front. Chem. 6:160. doi: 10.3389/fchem.2018.00160
Received
27 February 2018
Accepted
20 April 2018
Published
23 May 2018
Volume
6 - 2018
Edited by
Lorenzo Tei, Università degli Studi del Piemonte Orientale, Italy
Reviewed by
Francesca Garello, Università degli Studi di Torino, Italy; Amnon Bar-Shir, Weizmann Institute of Science, Israel; Lothar Helm, École Polytechnique Fédérale de Lausanne, Switzerland
Updates
Copyright
© 2018 Peterson, Srivastava and Pierre.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Valérie C. Pierre pierre@umn.edu
This article was submitted to Inorganic Chemistry, a section of the journal Frontiers in Chemistry
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.