Abstract
Introduction:
Dietary patterns were shown to be closely related to inflammation, which was independently associated with cognitive impairment (CI) in patients undergoing hemodialysis (HD). However, it remains unclear the influence of dietary patterns derived from inflammation on CI in this population. This study aimed to examine the association between dietary patterns derived from C-reactive protein (CRP) and interleukin-6 (IL-6) and CI in patients undergoing HD.
Methods:
Dietary intake was obtained from the simplified quantitative food frequency questionnaire. Reduced rank regression (RRR) was used to extract two dietary patterns, with IL-6 and CRP as response variables. Cognitive function was examined by the Montreal Cognitive Assessment (Beijing version). Venous blood was drawn for measuring IL-6 and CRP levels. Multivariable logistic regression was used to investigate the association between dietary patterns and CI.
Results:
Dietary pattern derived from IL-6 was not significantly associated with CI. The third quartile of dietary pattern, which used CRP as the response variable, significantly contributed to the increased risk of CI (AOR 8.62, 95% CI 1.47–50.67) after controlling age, sex, education level, marital status, and residential pattern (p-for-trend = 0.028). After considering hypertension and diabetes, physical activity level, anxiety and depression, smoking and drinking status, social support, energy intake, and the dietary pattern derived from IL-6 (p-for-trend = 0.026), the relationship between the dietary pattern derived from CRP and CI remained significant (AOR 14.54, 95% CI 1.40–151.13).
Conclusion:
Dietary pattern associated with high CRP level, including high intake of rice, liquor, fruit, tea and coffee and low intake of dark vegetables and juice, contributed to the increased risk of CI. The association between the consumption of seafood, sweet beverages, and alcohol and CI is yet to be established. However, they may be dietary contributing factors to inflammation in patients undergoing HD.
Introduction
Hemodialysis (HD) is the most widely available form of maintaining life for patients with end-stage kidney disease. Approximately 2.62 million people worldwide depend on HD treatment for survival (1). Cognitive impairment (CI) has been reported to be prevalent in 87% of individuals receiving HD globally (2). CI seriously affects the prognosis, hospitalization, and complications, thus decreasing patients' long-term quality of life and increasing the burden on families and society (3). Notably, the mortality rate of patients undergoing HD with CI was 1.7–2.5 times higher than that of patients without CI (4). However, the influencing factors that cause CI remained unclear (5). Increasing evidence has shown that diet is potentially associated with cognitive function in this population group (6, 7). The Mediterranean diet (MD), which mainly includes vegetables, fruits, seafood, beans, and nuts, has shown a promising association with a slow rate of cognitive decline and a reduced risk of CI (8). A randomized controlled trial (RCT) that included 447 participants has shown that MD was associated with the improvement of cognitive function (9). A Longitudinal neuroimaging study has also reported that high adherence to MD was associated with a slow rate of β-amyloid, which was closely related to cognitive function (10). Additionally, a prospective study that involved 3,831 participants over 65 years and followed up for 11 years has suggested that the Dietary Approaches to Stop Hypertension (DASH) dietary pattern was correlated with a reduced risk of cognitive function decline (11).
Low-grade chronic inflammation is common in patients undergoing HD. With the decline in renal clearance rate, the levels of circulating proinflammatory cytokines increase gradually (12). Oxidative and carbonyl stresses promoted by the uremic environment are highly proinflammatory (13). The microbiological quality and impurities in the dialysate may also lead to inflammation (14). In addition, a significant association between diet and inflammation has been presented in previous studies as well (15). A 5-year, single-blind, multicenter, controlled feeding trial randomized 66 participants into three groups: MD plus extra virgin olive oil, MD plus nuts and a low-fat diet (LFD). Compared with LFD, both MD groups had lower levels of interleukin-6 (IL-6) (16). In a meta-analysis of RCTs, DASH played an important role in decreasing the C-reactive protein (CRP) concentration (17). Plenty of studies have revealed that inflammation was a potential risk factor influencing cognitive function (18–20). A systematic review including 170 studies has revealed that patients in the CI group had higher levels of IL-6, compared to the control group (21). In a cohort study followed up for 6 years, higher levels of CRP, IL-1β, and fibrinogen were correlated with a higher risk of impaired attention (22). Therefore, dietary patterns leading to elevated inflammatory biomarkers may contribute to the development of CI. However, scientific evidence demonstrating the influence of dietary pattern related to inflammatory biomarkers on CI in patients undergoing HD is scarce. This study aimed to examine the association between dietary patterns derived from inflammatory biomarkers and cognitive impairment in patients undergoing HD.
Methods
Study design and participants
This cross-sectional study was conducted among patients undergoing HD using convenient sampling, with patient recruitment and data collection taking place between June and October 2022. All participants were recruited from a hemodialysis unit within a tertiary hospital in Nantong, in the Jiangsu province of China. Patients were eligible for the study if they were 18 years of age or older and undergoing HD for more than three months. Patients were excluded if they had psychiatric diseases, poor comprehension and hearing, and had abused alcohol and drugs for a long time. Patients who had taken antibiotics within 1 month or had severe inflammation within 6 months were also excluded. This study was reviewed and approved by the Institutional Review Board at Nantong University (2021-48), and all participants provided informed consent prior to participation.
The influencing factors of CI in patients undergoing HD were collected and analyzed based on the health ecology model in the study. According to previous publications, 15 influencing factors from five dimensions, including biological indicators, behavioral lifestyles, psychological factors, disease-related problems, and social environmental problems, were collected (23, 24). Following the calculation of sample size in the multifactor statistical analysis proposed by Kendall (25), the recommended sample size is usually 5–10 times the number of variables. A loss-to-follow-up rate of 20% has been considered, resulting in a required sample size between 90 and 180 patients. The collected influencing factors were included as covariates in the analysis to adjust for confounding effects on the relationship between dietary patterns derived from inflammatory biomarkers and cognitive impairment.
Dietary intake
Dietary intake was assessed using a quantitative simplified food frequency questionnaire (FFQ). It has been reported that FFQ had great reliability (0.54–0.77) and validity (0.54–0.87) (26). The FFQ is divided into 25 food groups, such as rice, gruel, noodle and their products, dessert, fried foods, and so on. Participants were asked to choose the frequency of consumption for each food item from nine possible frequency responses, ranging from “never” to “three times a day”. In addition, the intake of juice, tea, and coffee was also collected. Face-to-face interviews were conducted with patients during their HD sessions. Researchers printed and completed the FFQ based on patients' responses. Standard portions of food were photographed using a palm-sized reference object to facilitate participants' understanding and accurately estimate their dietary intake. Patients who had unreliable daily energy intake (< 800 kcal/day or >4,000 kcal/day for men; < 500 kcal/day or >3,500 kcal/day for women) were excluded from the analysis.
Cognitive function
Cognitive function was examined by the Montreal Cognitive Assessment Beijing version (MoCA-BJ), which is the most widely used screening tool to assess cognitive function. It has been reported that MoCA-BJ had high sensitivity (92.4%) and specificity (88.4%) in the identification of CI among the Chinese population (27). The MoCA-BJ includes eight cognitive fields: attention and concentration, executive function, memory, language, visuospatial construction, abstract thinking, computation, and orientation function. The total MoCA-BJ scores range from 0 to 30. The total scores would be increased by one point if participants were illiterate, primary, middle, or high school graduates (≤ 12 years of education). Participants with a score < 26 were defined as having CI (28). During dialysis sessions, executive and visuospatial construction sections in the questionnaire were filled out by patients themselves, and other sections were completed by researchers according to patients' responses.
CRP and IL-6
A volume of 1.5 ml blood (fasting) was taken from the participants for measuring the concentration of IL-6 and high-sensitivity CRP using an enzyme-linked immunosorbent assay (ELISA) before hemodialysis. When the IL-6 concentration is >7 pg/ml or the concentration of CRP is >5 mg/ml, inflammatory responses are observed in the body.
Demographics
Age, vintage, body mass index (BMI), and comorbidities, including hypertension and diabetes, were extracted from participants' medical records. Education level, drinking and smoking status, marital status, and residential pattern (living alone) were obtained from the demographic questionnaire. Education level was categorized as ≤ 12 years of education and >12 years of education. Marriage status was categorized as married, divorced, and unmarried.
Questionnaire
All the questionnaires, which were each marked with a unique non-identifiable participant study code, were administered to the patients by the researcher during their dialysis sessions. The hospital anxiety and depression scale (HADS) and social support rate scale (SSRS) were used to examine patients' anxiety and depression and the characteristics of social support for patients, respectively. The General Practice Physical Activity Questionnaire (GPPAQ) is utilized for assessing physical activity (PA) levels and identifying those with “Inactive” PA. It was designed to take less than 1 min to complete and the patients can be categorized into one of four “Physical Activity Index” (PAI) categories, namely “Inactive”, “Moderately Inactive”, “Moderately Active”, and “Active” based on the results.
Statistical analyses
Continuous variables that meet and do not meet the normal distribution were expressed as the mean ± standard deviation (SD), and median and interquartile range (IQR), respectively. Dichotomous variables are presented as numbers and percentages. An independent-sample t-test, one-way analysis of variance, and Mann-Whitney U test were performed to assess the distributions of general characteristics between CI and non-CI groups.
Reduced rank regression (RRR) analysis was used to extract two dietary patterns, with IL-6 and CRP as response variables, respectively. The RRR package was adopted to derive the dietary patterns by using the STATA statistical software version 16.0 (29). Factor loadings were analyzed to evaluate the association between 27 food groups and two dietary patterns. Food groups with factor loadings of ≥0.1 were positively correlated with the dietary pattern, while those with ≤ -0.1 were negatively correlated.
Logistic regression was used to examine the effect of dietary patterns on the risk of CI with odds ratios and 95% confidence intervals. The dietary pattern scores were calculated from the sum of the standardized intake of each food group multiplied by its factor loading, and the scores of dietary patterns were categorized into four groups based on the quartiles. The first quartile was used as the reference. In the multivariable-adjusted model, age, sex, education level, marital status, and residential pattern were considered in model 1, and model 2 was further adjusted for hypertension, diabetes, physical activity level, anxiety and depression, smoking and drinking status, social support scores, energy intake, and other dietary patterns. Moreover, the linear trend tests were conducted by incorporating the median value of four groups of dietary patterns as a continuous variable in the models. These statistical analyses were performed using the SPSS statistical software version 26.0. P < 0.05 were statistically significant.
Results
A total of 240 adult patients who had been undergoing HD for >3 months at the HD unit were preliminarily identified by unit staff. The unit staff further excluded patients who did not meet the eligible criteria and briefly explained the study to the potential patients during routine dialysis sessions. A total of 60 patients were excluded for the following reasons: poor hearing (n = 2), antibiotics usage within 1 month (n = 34), and severe infection within 6 months (n = 24). A total of 180 patients who expressed an interest in participating in the study were provided with the patient information sheets and discussed the study in more detail with the researchers. At least 48 h were given to the patients to decide whether to participate. If they were interested in the study after consideration, written consent was obtained from all participants prior to entering the study. A total of 175 patients ultimately participated in the study and signed the informed consent. Patients completed two separate interviews lasting about 20 min each and consisting of questionnaires and cognitive tests. During the study process, a patient who passed away during the study was excluded. Two patients were excluded due to missing or insufficient blood samples for analysis. The data of these three patients was completely missing at random, indicating that the probability of data being missed was entirely independent of its assumptions and other variables. Therefore, the study excluded the data and estimated any missing values from known variables. Finally, 172 patients were included in the analysis. A flowchart of included and excluded participants is shown in Figure 1.
Figure 1
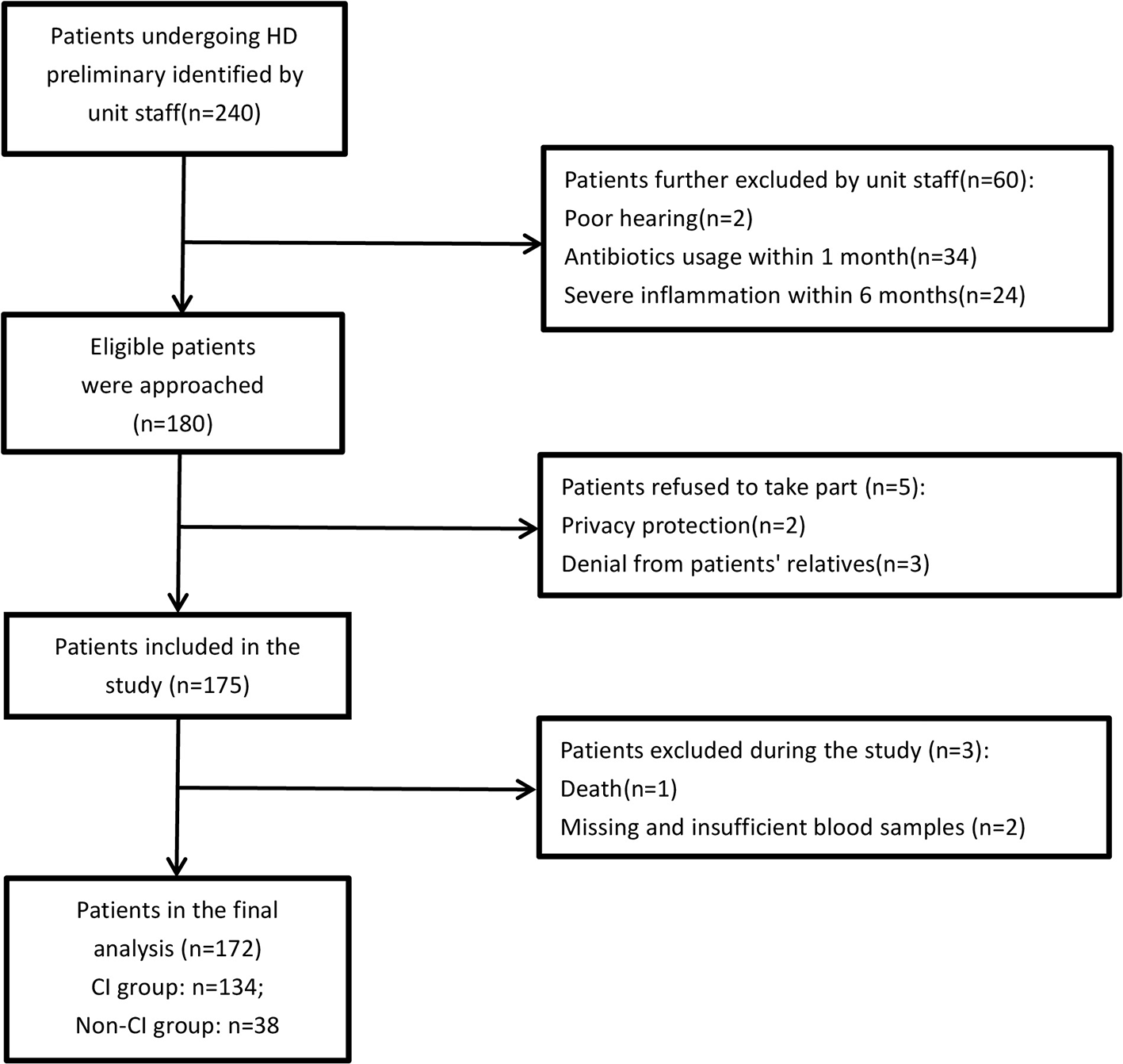
Flowchart of patients' recruitment. HD: hemodialysis; CI: cognitive impairment.
A total of 134 (77.91%) participants whose score of MoCA-BJ lower than 26 were grouped into the CI group, and the remaining 38 (22.09%) participants entered the non-CI group. Among all the included participants, 40.12% were women and 59.88% were men. The comparison of general characteristics between CI and non-CI groups is shown in Table 1. There were significant differences in gender, age, education level, residential pattern, and marital status between the two groups. Compared to the non-CI group, the participants in the CI group tended to be older (42.24 ± 1.61 vs. 57.37 ± 0.98 years), fewer educated, and more women (18.42% vs. 46.26%).
Table 1
| Characteristics | MoCA-BJ | P-value | ||
|---|---|---|---|---|
| Non-CI (n = 38) | CI (n = 134) | |||
| Biological indicators | Women | 7 (18.42) | 62 (46.26) | 0.002 a |
| Age (years) | 42.24 ± 1.61 | 57.37 ± 0.98 | < 0.001 a | |
| Vintage (years) | 2.00 (8.25) | 4.50 (9.00) | 0.39 | |
| BMI (kg/m2) | 22.29 (5.56) | 21.99 (4.99) | 0.82 | |
| Education level | < 0.001 a | |||
| • ≤ 12 years | 21 (55.26) | 116 (86.57) | ||
| •>12 years | 17 (44.74) | 18 (13.43) | ||
| Social environmental factors | Marital status | |||
| •Married | 27 (71.05) | 128(95.52) | < 0.001 a | |
| •Divorce | 1 (2.63) | 4 (2.99) | ||
| •Never married | 10 (26.32) | 2 (1.49) | ||
| Residential pattern (living alone) | 8 (21.05) | 10 (7.46) | 0.034 a | |
| Social support | 40.11 ± 0.44 | 39.85 ± 0.49 | 0.87 | |
| Disease-related indicators | Hypertension | 30 (78.94) | 86 (64.18) | 0.086 |
| Diabetes | 4 (10.53) | 30 (22.39) | 0.11 | |
| Behavioral lifestyle | Physical activity | 0.61 | ||
| •Inactive | 2 (5.26) | 4 (2.99) | ||
| •Moderately inactive | 5 (13.16) | 23 (17.16) | ||
| •Moderately active | 12 (31.58) | 32 (23.88) | ||
| •Active | 19 (50.00) | 75 (55.97) | ||
| Currently smoking | 7 (18.42) | 10 (7.46) | 0.91 | |
| Currently drinking | 5 (13.16) | 19 (14.18) | 0.87 | |
| Energy intake (kcal/kg/day) | 24.32 ± 1.62 | 21.00 ± 0.66 | 0.06 | |
| Psychological factors | Anxiety/depression | 2 (5.26) | 5 (3.73) | 0.68 |
Comparison of general characteristics between CI and non-CI groups.
ap < 0.05.
MoCA-BJ, Montreal Cognitive Assessment Beijing version; CI, cognitive impairment; BMI, body mass index.
RRR was used to extract two dietary patterns and explained 14.6% of the total variation of the response variables (IL-6 and CRP). Dietary pattern 1 (DP1) accounted for 8.2% of the variation of IL-6, and dietary pattern 2 (DP2) accounted for 6.4% of the variation of CRP. Factor loadings were used to describe the magnitudes and directions of the contributions of 27 food groups in two dietary patterns. In the DP1, food groups with factor loadings ≥0.1 were seafood, liquor, and sweet beverages, while those with factor loadings ≤ -0.1 were noodles, nuts, dumplings, and coarse cereals. As shown in Table 2, food groups with positive factor loadings greater than 0.1 included rice, fruit, tea and coffee, and liquor, while dark vegetables and juice had factor loadings < -0.1 in DP2.
Table 2
| Food/food groups | Dietary pattern 1 | Dietary pattern 2 | |
|---|---|---|---|
| Rice | −0.028 | 0.215‡ | |
| Gruel | −0.027 | 0.040 | |
| Noodle and their products | −0.149‡ | 0.031 | |
| Dessert | 0.041 | −0.037 | |
| Fried foods | −0.030 | −0.004 | |
| Dumplings | −0.192‡ | −0.090 | |
| Coarse cereals | −0.121‡ | −0.051 | |
| Tuber | 0.011 | 0.022 | |
| Milk | −0.070 | −0.018 | |
| Eggs | 0.090 | −0.088 | |
| Red meat | 0.069 | 0.063 | |
| Poultry | −0.001 | −0.080 | |
| Processed meat products | 0.003 | 0.030 | |
| Fish | −0.045 | −0.056 | |
| Seafood products | 0.207‡ | −0.082 | |
| Bean products | −0.099 | −0.034 | |
| Nuts | −0.110‡ | −0.071 | |
| Dark vegetables | −0.031 | −0.145‡ | |
| Light vegetables | 0.019 | −0.029 | |
| Mushrooms | 0.064 | −0.083 | |
| Fruits | −0.058 | 0.193‡ | |
| Sweet beverages | 0.196‡ | −0.039 | |
| Beer | −0.061 | −0.016 | |
| Rice wine | −0.020 | 0.003 | |
| Liquor | 0.105‡ | 0.101‡ | |
| Tea and coffee | 0.062 | 0.104‡ | |
| Juice | 0.067 | −0.145‡ | |
| Explained variation(%) | DP1 | DP2 | Total |
| Food/food groups | 3.1 | 3.2 | 6.3 |
| Predictor | 8.2 | 6.4 | 14.6 |
Factor loadings of food groups associated with dietary patterns 1, 2.
‡The absolute value of factor loadings >0.1.
DP1, dietary pattern 1 (dietary pattern derived from IL-6); DP2, dietary pattern 2 (dietary pattern derived from CRP).
The scores of dietary patterns were categorized into four groups based on the quartiles. General characteristics of patients undergoing HD in the first and fourth quartiles of two dietary patterns are presented in Table 3. Compared with patients in the first quartile, fewer patients in the fourth quartile of the DP1 were married. In the DP2, more patients in the fourth quartile were men, younger, current smokers, and lived alone. Meanwhile, fewer patients suffered from diabetes compared to those in the first quartile.
Table 3
| Characteristics | DP1 | DP2 | |||||
|---|---|---|---|---|---|---|---|
| Q1 | Q4 | P-value | Q1 | Q4 | P-value | ||
| Biological indicators | Women | 11 (22.58) | 18 (41.86) | 0.11 | 22 (51.16) | 9 (20.93) | 0.004 a |
| Age (years) | 55.6 ± 1.93 | 50.12 ± 2.06 | 0.06 | 57.42 ± 2.02 | 49.95 ± 1.73 | 0.006 a | |
| Vintage (years) | 2.00 (8.50) | 7.00 (9.50) | 0.09 | 3.50 (7.80) | 2.50 (11.00) | 0.87 | |
| BMI (kg/m2) | 22.82 (5.60) | 22.68 ± 0.57 | 0.60 | 22.01 ± 0.48 | 22.69 ± 0.48 | 0.32 | |
| Education level | 0.25 | 0.17 | |||||
| • ≤ 12 years | 21 (48.84) | 28 (65.12) | 33 (76.74) | 35 (81.4) | |||
| •>12 years | 22 (51.16) | 15 (34.88) | 10 (23.26) | 8 (18.6) | |||
| Social environmental factors | Marital status | 0.033 a | 0.18 | ||||
| •Married | 42 (97.67) | 35 (81.40) | 39 (90.70) | 38 (88.37) | |||
| •Divorce | 0(0.00) | 2 (4.65) | 0 (0.00) | 3 (6.98) | |||
| •Never married | 1 (2.33) | 6 (13.95) | 4 (9.30) | 2 (4.65) | |||
| Residential pattern (living alone) | 6 (13.95) | 8 (18.60) | 0.56 | 2 (4.65) | 10 (23.26) | 0.01 a | |
| Social support | 41.79 ± 0.82 | 39.26 ± 1.18 | 0.08 | 39.91 ± 1.04 | 39.07 ± 1.08 | 0.58 | |
| Disease-related indicators | Hypertension | 27 (62.79) | 30 (69.77) | 0.49 | 25 (58.14) | 30 (69.77) | 0.26 |
| Diabetes | 7 (16.28) | 5 (11.63) | 0.53 | 11 (25.58) | 4 (9.3) | 0.047 a | |
| Behavioral lifestyle | Physical activity | 0.53 | 0.68 | ||||
| •Inactive | 1 (2.33) | 2 (4.65) | 1 (2.33) | 2 (4.65) | |||
| •Moderately inactive | 7 (16.28) | 9 (20.93) | 8 (18.60) | 5 (11.63) | |||
| •Moderately active | 9 (20.93) | 13 (30.23) | 11 (25.58) | 9 (20.93) | |||
| •Active | 26 (60.47) | 19 (44.19) | 23 (53.49) | 27 (62.79) | |||
| Currently smoking | 4 (9.34) | 6 (13.95) | 0.50 | 2 (4.65) | 9 (20.93) | 0.02 a | |
| Currently drinking | 8 (18.60) | 7 (16.28) | 0.78 | 5 (11.63) | 10 (23.26) | 0.16 | |
| Energy intake (kcal/kg) | 24.53 ± 1.20 | 23.19 ± 1.45 | 0.48 | 20.11 (11.29) | 23.87 (9.68) | 0.19 | |
| Psychological factors | Anxiety/depression | 3 (6.98) | 0 (0.00) | 0.24 | 1 (2.33) | 1 (2.33) | 1.00 |
Characteristics of patients in different quartiles of dietary patterns scores.
ap < 0.05.
Q1, first quartile; Q4, fourth quartile; DP1, dietary pattern 1 (dietary pattern derived from IL-6); DP2, dietary pattern 2 (dietary pattern derived from CRP); BMI, body mass index.
Adjusted odds ratios (AOR) and the 95% confidence intervals for CI across the quartiles of each dietary pattern are presented in Table 4. DP1 was not significantly associated with the risk of CI. In model 1, the third quartile of DP2 significantly increased the risk of CI (AOR 8.62, 95% CI 1.47–50.67) after adjusting for age, sex, education level, marriage status, and residential pattern (p-for-trend = 0.028). Model 2 was further adjusted for hypertension and diabetes, physical activity level, anxiety and depression, smoking and drinking status, social support, energy intake, and dietary pattern derived from IL-6 (p-for-trend = 0.026), and it was found that the third quartile of DP2 significantly increased the CI risk compared to patients in the first quartile of DP2 (AOR 14.54, 95% CI 1.40–151.13).
Table 4
| Quartile of dietary pattern | DP1 | DP2 | ||
|---|---|---|---|---|
| Model 1 | Model 2 | Model 1 | Model 2 | |
| Q1 | 1.00 | 1.00 | 1.00 | 1.00 |
| Q2 | 2.01 (0.40–10.04) | 1.39 (0.14–13.71) | 1.06 (0.24–4.72) | 0.63 (0.15–6.20) |
| Q3 | 0.95 (0.22–4.08) | 0.34 (0.41–2.75) | 8.62 (1.47–50.67) a | 14.54 (1.40–151.13) a |
| Q4 | 0.80 (0.21–3.07) | 0.81 (0.14–4.77) | 4.44 (0.85–23.29) | 5.92 (0.61–57.11) |
| P for trend | 0.64 | 0.55 | 0.028 a | 0.026 a |
Logistic regression on the relationship between dietary pattern and cognitive impairment.
ap < 0.05.
Q1, first quartile; Q2, second quartile; Q3, third quartile; Q4, fourth quartile; DP1, dietary pattern 1 (dietary pattern derived from IL-6); DP2, dietary pattern 2 (dietary pattern derived from CRP).
Model 1: Adjusted for sex, age, education level, marital status, and residential pattern; Model 2: Adjusted for hypertension and diabetes, physical activity level, anxiety and depression, history of smoking and drinking, social support, energy intake, and dietary pattern derived from IL-6.
Discussion
The existing evidence from epidemiological, cross-sectional, and neuroimaging research has shown that diet plays a potential role in improving cognitive function. The significant association between diet and inflammation has been presented in previous studies as well. Additionally, as we know, inflammation is a potential risk factor influencing cognitive function. However, evidence examining the association between dietary patterns, inflammation, and cognitive function in patients undergoing HD is scarce. There were two main findings in this study. Two dietary patterns with IL-6 and CRP as response variables were identified to show the relationship between diet and inflammation. The dietary pattern that accounted for the variation in IL-6 was characterized by a high intake of seafood, sweet beverages, and liquor and a low intake of noodles, nuts, coarse cereals, and dumplings. The dietary pattern that explained the variation in CRP was characterized by a high intake of rice, tea and coffee, liquor, and fruit and a low intake of dark vegetables and juice. Additionally, the study showed that dietary patterns utilizing CRP as the response variable significantly contributed to the increased risk of CI, while dietary patterns derived from IL-6 did not.
CRP and IL-6 were used as the response variables to understand the dietary pattern in this study, which is in agreement with some previous studies aiming to examine the influence of diet on inflammation. The MONICA/KORA Augsburg cohort study extracted the dietary pattern with IL-6, IL-18, and CRP as the response in 981 middle-aged men (30), in which the dietary pattern related to inflammation was characterized by a high intake of meat, sweet beverages, and liquor. The lack of association between inflammation and meat consumption in the study may be attributed to the fact that patients undergoing HD were less inclined to consume meat due to concerns regarding high phosphorus intake (31). Different from the previous studies showing that long-chain n-3 polyunsaturated fatty acids in seafood decreased inflammation (32), our findings have shown that the intake of seafood increased the concentration of IL-6. The processing and storage of seafood may lead to an increase in inflammation due to the formation of dietary cholesterol oxidation products (COPs), which are heterogeneous compounds that occur during the processing and storage of cholesterol-rich seafood (33). COPs are linked to the induction of oxidative stress and subsequent inflammation (34). Similar to the previous study, sweet beverages were associated with an increasing level of inflammation in the study. Compared to rats fed with an ad libitum chow diet only, those supplemented with sweetened beverages exhibited elevated levels of tumor necrosis factor (TNF) and interleukin-1β (35). The sugar in sweet beverages resulted in a reduction of probiotics within the intestines and induced inflammation. Juice was found to be negatively correlated with inflammation in this study. It is consistent with several studies where researchers have observed a reduction in CRP level following juice consumption (36, 37). Juice is rich in anthocyanins, flavonoids, polyphenols, and vitamin C, which possess antioxidant and anti-inflammatory properties (38). Similar to our finding, high alcohol consumption was found to be positively correlated with elevated levels of CRP and IL-6 (39), although a study conducted on rat models demonstrated that alcohol could promote anti-inflammatory processes (40). A national health survey carried out in former West Germany suggested that non-drinkers and heavy drinkers had higher CRP concentrations than moderate drinkers (41). Alcohol consumption showed a U-shaped association with mean CRP values. Dosage may play an important role in the effect of liquor on inflammation.
A systematic review including 46 studies reported that vegetable-based and fruit-based dietary patterns were negatively associated with low-grade chronic inflammation (42). Similarly, in our study, dark vegetables were confirmed to be beneficial in reducing CRP concentrations. Vegetables have strong antioxidant and anti-inflammatory properties, which reduce the levels of inflammatory factors by inhibiting the production of free radicals (43). Furthermore, vegetables are rich in fiber that positively influences gut microbiota, which may contribute to the mitigation of inflammatory responses (44). However, the aforementioned findings on fruits were incongruous with our discovery that fruit consumption led to an elevation in CRP level. The potential reason for this discrepancy may lie in the patients' preference for high-sugar fruits, such as grapes and peaches, as reported by many participants. Low-sugar fruits, like oranges and kiwifruit, were often rejected by patients undergoing HD due to their high potassium content. It has been known that foods rich in sugar lead to an inflammatory reaction (45). The overconsumption of fructose in fruits or starch in rice provokes metabolic changes that result in low-grade chronic inflammation (46). Fructose consumption induces leptin resistance, resulting in elevated levels of leptin. However, excessive leptin levels can trigger adipocyte inflammation (47). In addition, leptin contributes to the release of reactive oxygen species and the recruitment of monocytes, which subsequently trigger an inflammatory response (48).
The study revealed that the consumption of tea and coffee was positively associated with inflammation. Results on the impact of tea and coffee on inflammation exhibit inconsistency. The presence of numerous polyphenolic compounds in tea and coffee is generally attributed to their anti-inflammatory properties (49). However, the processing technology of tea may affect its effect on inflammation. Fermented tea is commonly believed to be more effective in reducing lipids, while unfermented tea alleviates inflammation (50). A randomized study has indicated that the consumption of both fermented and unfermented tea does not have a significant impact on reducing CRP and IL-6 levels (51). Furthermore, a systematic review of RCTs suggested that green tea (unfermented tea) may not be effective in altering the levels of CRP and IL-6, particularly in cases with low inflammation (52). A cross-sectional study including 3,042 participants showed that coffee consumption was related to an increase in inflammatory markers (53). An RCT also demonstrated that patients who consumed caffeinated coffee exhibited higher levels of inflammation compared to those who abstained from coffee consumption (54). Caffeine may play an important role in increasing the levels of inflammatory markers (55).
The study further revealed that dietary pattern with CRP as a response variable was associated with an increased risk of CI. Numerous studies have consistently demonstrated that a diet with pro-inflammatory properties is significantly associated with an elevated risk of CI (56–60). However, the finding is inconsistent with the results of the Nurses' Health Study, which discovered no correlation between a pro-inflammatory diet and cognitive function in a vast population primarily consisting of Caucasian and highly educated elderly women (61). Further research may be necessary to confirm the relationship between pro-inflammatory diets and cognitive function in men and other races. The consumption of tea and coffee was observed in the dietary pattern related to CI in the study. Noguchi-Shinohara's study has revealed that black tea intake is associated with an increasing risk of CI compared to green tea (62), indicating patients in the study may prefer the consumption of black tea. Catechin in green tea increased the expression of immediate-early genes in the hippocampus and prevented the decline of cognitive function (63). The consumption of green tea has also been shown to decrease Alzheimer's disease (AD)-related pathology and enhance anti-oxidative stress capacity, thereby achieving the objective of reducing CI risk (64). As mentioned before, the association between coffee consumption and cognitive function remains controversial. Some studies have suggested that coffee may improve cognitive decline in individuals with impaired cognition (65). Coffee has been shown to decrease lipid peroxidation in brain membranes, increase antioxidant levels, and enhance long-term memory (66). Additionally, coffee may reduce the risk of CI by decreasing pathological cerebral amyloid-beta (Aβ) deposition, which is a characteristic of AD (67). However, a meta-analysis including up to 415,530 participants did not find evidence for the effects of coffee consumption on global cognition or memory (68). In fact, higher intake of coffee was cross-sectionally associated with smaller hippocampal volume and poorer memory function (69). The association between coffee and CI in patients undergoing HD may depend on the amount of coffee consumed, and further investigation is warranted.
The “white rice” pattern, characterized by high consumption of white rice, flour-based food, and alcohol, was found to have a positive association with CI (70). Our result suggested that a high intake of white rice and liquor was associated with an increased risk of CI. The association between white rice and CI may be explained by low intake of micronutrients (71), as many studies have indicated associations between CI and antioxidant vitamins (vitamin E, vitamin C, and β-carotene) and B vitamins (vitamin B6 and folate) deficiency (72). The association between alcohol consumption and CI may be influenced by dosage. Long-term drinking has been linked to an increased risk of cognitive decline (73). A dose-response meta-analysis has demonstrated that low intake of alcohol intake (< 11 g/day) is associated with a reduced risk of CI (74), while individuals who abstain from drinking or consuming high levels of liquor have an elevated risk (75). Compared to non-drinkers, moderate lifetime alcohol consumption is associated with lower Aβ deposition (76). The findings in the study are consistent with previous research (77), which also suggested that the consumption of vegetables and juice is beneficial for cognition function. Similarly, our study found that dark vegetables and juice specifically have a positive impact on CI. Polyphenols found in vegetables and juice are associated with improved cognition (78). However, we observed an increased risk of CI associated with fruit consumption. Fruit with a high glycemic index or load may disrupt insulin signaling, impair glucose metabolism, and consequently result in neuronal loss, reduced cortical thickness, and CI (79).
The ultra-processed food (UPF) was not considered a distinct group in the study. However, three food groups, namely, desserts, processed meat products, and sweet beverages, belong to the UPFs. Weinstein and colleagues noted in a recent study that the consumption of ultra-processed meat, oils/spreads, and dairy products has been positively associated with cognitive decline in elderly individuals with type-2 diabetes, particularly among women and obese individuals (80). The UPFs are characterized by high levels of starches, fats, oils, sugars, emulsifiers, and food additives that have a direct link to CI (81). The underlying mechanisms for the impact of UPF on cognitive function may include low-grade chronic inflammation (82, 83). Desserts and processed meat products groups were not significantly correlated with CRP or IL-6 in the study. However, sweet beverages exhibited a positive correlation with IL-6 levels in patients undergoing HD, which potentially corroborates the relationship between the UPFs and CI. The UPF groups in the study failed to show a significant association with CI; this finding is inconsistent with previous research indicating that consumption of sugary beverages increases the risk of Alzheimer's disease (84). The inconsistency observed in the study may be attributed to the limited consumption of sweetened beverages reported by patients. Moreover, the link between UPFs and CI could be mediated through chronic conditions such as diabetes, cardiovascular disease, and obesity. Studies are needed to further examine the relationship between UPF and CI in patients undergoing HD.
Strengths and limitations
Our study has several strengths. To the best of our knowledge, no previous studies have explored the association between dietary patterns, inflammatory biomarkers, and CI in patients undergoing HD. Our study was of great significance from the perspective of public health. A cost-effective strategy was given to patients undergoing HD to prevent the decline of cognitive function. However, several limitations should be considered. First, the causal relationship between dietary patterns derived from inflammation and CI was difficult to examine due to the cross-sectional study design. Second, dietary intake was obtained from FFQ, and there may be some recall bias. Finally, the dietary pattern derived from RRR explains the variation of response variables to the greatest extent, instead of the variation of food. A dietary pattern for dietary guidance should be obtained through factor analysis.
Practical application
Encouraging and supervising more dark vegetables and low-sugar juice consumption may be one method of improving cognitive impairment by relieving inflammation in patients undergoing HD. However, the consumption of rice, tea, coffee, liquor, and high-sugar fruits should be prudent in order to alleviate inflammation and reduce cognitive decline. The intake of fruits and juice for patients undergoing HD may require the guidance of nephrologists and other healthcare professionals due to their sugar and potassium content. Additionally, the types of teas and coffees, as well as the method of cooking rice, may be contributing factors to inflammation reactions. Further exploration is necessary to clarify their relationship with cognitive function in patients undergoing HD. Meanwhile, strategies to improve inflammation control in individuals receiving HD should concentrate on the dietary restriction of seafood, sweet beverages, and liquor. However, the association between alcohol intake and inflammation requires further examination.
Conclusion
This study provides patients undergoing HD with potential nonpharmacological, low cost dietary advice to improve cognitive function. The study suggests that dietary pattern, with CRP as a response variable, which is characterized by a high intake of rice, tea and coffee, liquor, and fruit and a low intake of dark vegetables and juice, is significantly associated with an increased risk of CI in patients undergoing HD. Consumption of seafood, sweet beverages, and liquor may be contributing factors to inflammation in patients; however, their association with CI has not yet been found.
Statements
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Ethics statement
This study was reviewed and approved by the Institutional Review Board at Nantong University (2021-48) and all participants provided informed consent prior to participation. The patients/participants provided their written informed consent to participate in this study.
Author contributions
Conceptualization, methodology, writing—original draft preparation, and writing—review and editing: YZ, XW, and YS. Software: YZ. Validation: XW. Formal analysis: YZ and XW. Investigation, resources, and data curation: YZ, XW, XuZ, QF, and XiZ. Visualization, supervision, project administration, and funding acquisition: YS. All authors contributed important intellectual content during manuscript drafting or revision, accepts personal accountability for the author's own contributions, and agrees to ensure that questions pertaining to the accuracy or integrity of any portion of the study are appropriately investigated and resolved.
Funding
This study was supported by the Social Livelihood Science and Technology Fund of Nantong (MS22022064).
Acknowledgments
We thank all the interviewers and everyone else who assisted with the survey.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1.
Zhou H Al-Ali F Kang GE Hamad A Ibrahim RA Talal T et al . Application of wearables to facilitate virtually supervised intradialytic exercise for reducing depression symptoms. Sensors (Basel). (2020) 20:1571. 10.3390/s20061571
2.
Murray AM Tupper DE Knopman DS Gilbertson DT Pederson SL Li S et al . Cognitive impairment in hemodialysis patients is common. Neurology. (2006) 67:216–23. 10.1212/01.wnl.0000225182.15532.40
3.
Xiaoyu L Gexia Z Fangqiang C Qian L Xue Z . Progress in Research of Cognitive Function in Hemodialysis Patients. China Continuing Med Educ. (2022) 14:194–8.
4.
Angermann S Schier J Baumann M Steubl D Hauser C Lorenz G et al . Cognitive impairment is associated with mortality in hemodialysis patients. J Alzheimers Dis. (2018) 66:1529–37. 10.3233/JAD-180767
5.
Noble JM Scarmeas N Papapanou PN . Poor oral health as a chronic, potentially modifiable dementia risk factor: review of the literature. Curr Neurol Neurosci Rep. (2013) 13:384. 10.1007/s11910-013-0384-x
6.
Lu R Fang Y Zhou Y Che M Shen J Liu Q et al . A pilot study of thiamin and folic acid in hemodialysis patients with cognitive impairment. Ren Fail. (2021) 43:766–73. 10.1080/0886022X.2021.1914656
7.
Nikić PM Andrić BR Stojimirović BB Trbojevic-Stanković J Bukumirić Z . Habitual coffee consumption enhances attention and vigilance in hemodialysis patients. Biomed Res Int. (2014) 2014:707460. 10.1155/2014/707460
8.
Solfrizzi V Panza F . Mediterranean diet and cognitive decline. A lesson from the whole-diet approach: what challenges lie ahead?J Alzheimers Dis. (2014) 39:283–6. 10.3233/JAD-130831
9.
Valls-Pedret C Sala-Vila A Serra-Mir M Corella D Torre Rd Martínez-González MÁ et al . Mediterranean diet and age-related cognitive decline: a randomized clinical trial. JAMA Intern Med. (2015) 175:1094–103. 10.1001/jamainternmed.2015.1668
10.
Berti V Walters M Sterling J Quinn CG Logue M Andrews R et al . Mediterranean diet and 3-year alzheimer brain biomarker changes in middle-aged adults. Neurology. (2018) 90:e1789–98. 10.1212/WNL.0000000000005527
11.
Wengreen H Munger RG Cutler A Quach A Bowles A Corcoran C et al . Prospective study of dietary approaches to stop hypertension- and mediterranean-style dietary patterns and age-related cognitive change: the cache county study on memory, health and aging. Am J Clin Nutr. (2013) 98:1263–71. 10.3945/ajcn.112.051276
12.
Carrero JJ Stenvinkel P . Persistent inflammation as a catalyst for other risk factors in chronic kidney disease: a hypothesis proposal. Clin J Am Soc Nephrol. (2009) 4:S49–55. 10.2215/CJN.02720409
13.
Aveles PR Criminácio CR Gonçalves S Bignelli AT Claro LM Siqueira SS et al . Association between biomarkers of carbonyl stress with increased systemic inflammatory response in different stages of chronic kidney disease and after renal transplantation. Nephron Clin Pract. (2010) 116:c294–9. 10.1159/000318792
14.
Santoro A Mancini E . Is hemodiafiltration the technical solution to chronic inflammation affecting hemodialysis patients?Kidney Int. (2014) 86:235–7. 10.1038/ki.2014.81
15.
Galland L . Diet and inflammation. Nutr Clin Pract. (2010) 25:634–40. 10.1177/0884533610385703
16.
Casas R Urpi-Sardà M Sacanella E Arranz S Corella D Castner O et al . Anti-inflammatory effects of the mediterranean diet in the early and late stages of atheroma plaque development. Mediators Inflamm. (2017) 2017:3674390. 10.1155/2017/3674390
17.
Soltani S Chitsazi MJ Salehi-Abargouei A . The effect of dietary approaches to stop hypertension (dash) on serum inflammatory markers: a systematic review and meta-analysis of randomized trials. Clin Nutr. (2018) 37:542–50. 10.1016/j.clnu.2017.02.018
18.
Adesso S Magnus T Cuzzocrea S Campolo M Rissiek B Paciello O et al . Indoxyl sulfate affects glial function increasing oxidative stress and neuroinflammation in chronic kidney disease: interaction between astrocytes and microglia. Front Pharmacol. (2017) 8:370. 10.3389/fphar.2017.00370
19.
Assem M Lando M Grissi M Kamel S Massy ZA Chillon JM et al . The impact of uremic toxins on cerebrovascular and cognitive disorders. Toxins (Basel). (2018) 10:303. 10.3390/toxins10070303
20.
Nowak KL Chonchol M . Does inflammation affect outcomes in dialysis patients?Semin Dial. (2018) 31:388–97. 10.1111/sdi.12686
21.
Shen X-N Niu L-D Wang Y-J Cao X-P Liu Q Tan L et al . Inflammatory markers in Alzheimer's disease and mild cognitive impairment: a meta-analysis and systematic review of 170 studies. J Neurol Neurosurg Psychiatry. (2019) 90:590–8. 10.1136/jnnp-2018-319148
22.
Tamura MK Tam K Vittinghoff E Raj D Sozio SM Rosas SE et al . Inflammatory markers and risk for cognitive decline in chronic kidney disease: the cric study. Kidney Int Rep. (2017) 2:192–200. 10.1016/j.ekir.2016.10.007
23.
Liu H Song Y Zhao D Zhan M . Effect of exercise on cognitive impairment in patients undergoing haemodialyses: a systematic review and meta-analysis of randomised controlled trials. J Ren Care. (2022) 48:243–52. 10.1111/jorc.12420
24.
Karakizlis H Bohl K Ziemek J Dodel R Hoyer J . Assessment of cognitive impairment and related risk factors in hemodialysis patients. J Nephrol. (2022) 35:931–42. 10.1007/s40620-021-01170-3
25.
Li A . Development and Preliminary Application of the Fear of Disease Progression Scale for Patients with Acute Myocardial Infaction. Zhenjiang: JiangSu University (2021).
26.
Jian G Jia-qing F Li-jing J Wen-qian Y Bing L Hong-wei G . Assessment of thereproducibility and validity of a simple food-frequency questionnaire used in dietary patterns studies. Acta Nutrimenta Sinica. (2011) 33:452–6. 10.13325/j.cnki.acta.nutr.sin.2011.05.012
27.
Hong-bo W Zhen-xin Z Fu-sheng N Li L . The application of montreal cognitive assessment in urban mhinese residents of Beijing. Chinese J Internal Medi. (2008) 2008:36–9. 10.3321/j.issn:0578-1426.2008.01.012
28.
Feiyun H Yanhong W Juanjuan L Li W Yu J Susu L . Diagnostic value of montreal cognitive assessment for mild cognitive impairment in chinese middle-aged adults: a meta-analysis. Chin J Evid-Based Med. (2017) 17:450–7. 10.7507/1672-2531.201609052
29.
Zheng B Liu Q Lyu J Yu CQ . Introduction of reduced rank regression and development of a user-written stata package. Chin J Epidemiol. (2022) 43:403–8.
30.
Meyer J Döring A Herder C Roden M Koenig W Thorand B . Dietary patterns, subclinical inflammation, incident coronary heart disease and mortality in middle-aged men from the monica/kora augsburg cohort study. Eur J Clin Nutr. (2011) 65:800–7. 10.1038/ejcn.2011.37
31.
Cupisti A Kalantar-Zadeh K . Management of natural and added dietary phosphorus burden in kidney disease. Semin Nephrol. (2013) 33:180–90. 10.1016/j.semnephrol.2012.12.018
32.
Mendivil CO . Dietary fish, fish nutrients, and immune function: a review. Front Nutr. (2020) 7:617652. 10.3389/fnut.2020.617652
33.
Liu Y Yang X Xiao F Jie F Zhang Q Liu Y et al . Dietary cholesterol oxidation products: perspectives linking food processing and storage with health implications. Compr Rev Food Sci Food Saf. (2022) 21:738–79. 10.1111/1541-4337.12880
34.
Gamba P Testa G Gargiulo S Staurenghi E Poli G Leonarduzzi G . Oxidized cholesterol as the driving force behind the development of Alzheimer's disease. Front Aging Neurosci. (2015) 7:119. 10.3389/fnagi.2015.00119
35.
Kjaergaard M Nilsson C Nielsen MO Grove K Raun K . Hypothalamic oxidative stress and inflammation, and peripheral glucose homeostasis in sprague-dawley rat offspring exposed to maternal and postnatal chocolate and soft drink. Nutr Diabetes. (2018) 8:44. 10.1038/s41387-018-0051-z
36.
Chai SC Davis K Zhang Z Zha L Kirschner KF . Effects of tart cherry juice on biomarkers of inflammation and oxidative stress in older adults. Nutrients. (2019) 11:11020228. 10.3390/nu11020228
37.
Miles EA Calder PC . Effects of citrus fruit juices and their bioactive components on inflammation and immunity: a narrative review. Front Immunol. (2021) 12:712608. 10.3389/fimmu.2021.712608
38.
Ou B Bosak KN Brickner PR Iezzoni DG Seymour EM . Processed tart cherry products–comparative phytochemical content, in vitro antioxidant capacity and in vitro anti-inflammatory activity. J Food Sci. (2012) 77:H105–12. 10.1111/j.1750-3841.2012.02681.x
39.
Shi H Ter Horst R Nielen S Bloemendaal M Jaeger M Joosten I et al . The gut microbiome as mediator between diet and its impact on immune function. Sci Rep. (2022) 12:5149. 10.1038/s41598-022-08544-y
40.
Terasaki LS Schwarz JM . Effects of moderate prenatal alcohol exposure during early gestation in rats on inflammation across the maternal-fetal-immune interface and later-life immune function in the offspring. J Neuroimmune Pharmacol. (2016) 11:680–92. 10.1007/s11481-016-9691-8
41.
Imhof A Froehlich M Brenner H Boeing H Pepys MB Koenig W . Effect of alcohol consumption on systemic markers of inflammation. Lancet. (2001) 357:763–7. 10.1016/S0140-6736(00)04170-2
42.
Barbaresko J Koch M Schulze MB Nöthlings U . Dietary pattern analysis and biomarkers of low-grade inflammation: a systematic literature review. Nutr Rev. (2013) 71:511–27. 10.1111/nure.12035
43.
Salminen LE Paul RH . Oxidative stress and genetic markers of suboptimal antioxidant defense in the aging brain: a theoretical review. Rev Neurosci. (2014) 25:805–19. 10.1515/revneuro-2014-0046
44.
Gehlich KH Beller J Lange-Asschenfeldt B Köcher W Meinke MC Lademann J . Fruit and vegetable consumption is associated with improved mental and cognitive health in older adults from non-western developing countries. Public Health Nutr. (2019) 22:689–96. 10.1017/S1368980018002525
45.
Proctor C Thiennimitr P Chattipakorn N Chattipakorn S . Diet, gut microbiota and cognition. Metab Brain Dis. (2017) 32:1–17. 10.1007/s11011-016-9917-8
46.
DiNicolantonio JJ Mehta V Onkaramurthy N O'Keefe JH . Fructose-induced inflammation and increased cortisol: a new mechanism for how sugar induces visceral adiposity. Prog Cardiovasc Dis. (2018) 61:3–9. 10.1016/j.pcad.2017.12.001
47.
Shapiro A Tümer N Gao Y Cheng K-Y Scarpace PJ . Prevention and reversal of diet-induced leptin resistance with a sugar-free diet despite high fat content. Br J Nutr. (2011) 106:390–7. 10.1017/S000711451100033X
48.
Fantuzzi G Faggioni R . Leptin in the regulation of immunity, inflammation, and hematopoiesis. J Leukoc Biol. (2000) 68:437–46. 10.1189/jlb.68.4.437
49.
P LI Bukowska B . Tea and coffee polyphenols and their biological properties based on the latest in vitro investigations. Ind Crops Prod. (2022) 175:114265. 10.1016/j.indcrop.2021.114265
50.
Deng X Hou Y Zhou H Yi L Xue Z Xue X et al . Hypolipidemic, anti-inflammatory, and anti-atherosclerotic effects of tea before and after microbial fermentation. Food Sci Nutr. (2021) 9:1160–70. 10.1002/fsn3.2096
51.
de Maat MP Pijl H Kluft C Princen HM . Consumption of black and green tea had no effect on inflammation, haemostasis and endothelial markers in smoking healthy individuals. Eur J Clin Nutr. (2000) 54:757–63. 10.1038/sj.ejcn.1601084
52.
Haghighatdoost F Hariri M . The effect of green tea on inflammatory mediators: a systematic review and meta-analysis of randomized clinical trials. Phytother Res. (2019) 33:2274–87. 10.1002/ptr.6432
53.
Zampelas A Panagiotakos DB Pitsavos C Chrysohoou C Stefanadis C . Associations between coffee consumption and inflammatory markers in healthy persons: the attica study. Am J Clin Nutr. (2004) 80:862–7. 10.1093/ajcn/80.4.862
54.
Wedick NM Brennan AM Sun Q Hu FB Mantzoros CS van Dam RM . Effects of caffeinated and decaffeinated coffee on biological risk factors for type 2 diabetes: a randomized controlled trial. Nutr J. (2011) 10:93. 10.1186/1475-2891-10-93
55.
Paiva C Beserra B Reis C Dorea JG Da Costa T Amato AA . Consumption of coffee or caffeine and serum concentration of inflammatory markers: a systematic review. Crit Rev Food Sci Nutr. (2019) 59:652–63. 10.1080/10408398.2017.1386159
56.
Ozawa M Shipley M Kivimaki M Singh-Manoux A Brunner EJ . Dietary pattern, inflammation and cognitive decline: the whitehall ii prospective cohort study. Clin Nutr. (2017) 36:506–12. 10.1016/j.clnu.2016.01.013
57.
Fan Y-C Chou C-C Bintoro BS Pan W-H Bai C-H . Combined effect of inflammation and hyperglycemia on mild cognitive impairment and associated dietary patterns in an older taiwanese population. Front Nutr. (2022) 9:791929. 10.3389/fnut.2022.791929
58.
Kesse-Guyot E Assmann KE Andreeva VA Touvier M Neufcout L Shivappa N et al . Long-term association between the dietary inflammatory index and cognitive functioning: findings from the Su.Vi.Max study.Eur J Nutr. (2017) 56:1647–55. 10.1007/s00394-016-1211-3
59.
Jia Y Yan S Sun M Yang Y Wang L Wu C et al . Association between dietary inflammatory index and cognitive impairment: a meta-analysis. Front Aging Neurosci. (2022) 14:1007629. 10.3389/fnagi.2022.1007629
60.
Gu Y Manly JJ Mayeux RP Brickman AM . An inflammation-related nutrient pattern is associated with both brain and cognitive measures in a multiethnic elderly population. Curr Alzheimer Res. (2018) 15:493–501. 10.2174/1567205015666180101145619
61.
Melo van Lent D Samieri C Grodstein F Seshadri S . Empirical dietary inflammatory pattern scores are not associated with worse cognitive performance in the nurses' health study. J Nutr. (2022) 152:2526–33. 10.1093/jn/nxac157
62.
Noguchi-Shinohara M Yuki S Dohmoto C Ikeda Y Samuraki M Iwasa K et al . Consumption of green tea, but not black tea or coffee, is associated with reduced risk of cognitive decline. PLoS ONE. (2014) 9:e96013. 10.1371/journal.pone.0096013
63.
Unno K Pervin M Taguchi K Konishi T Nakamura Y . Green tea catechins trigger immediate-early genes in the hippocampus and prevent cognitive decline and lifespan shortening. Molecules. (2020) 25:1484. 10.3390/molecules25071484
64.
Zhang R Zhang L Li Z Zhang P Song H Yao D-A et al . Green tea improves cognitive function through reducing ad-pathology and improving anti-oxidative stress capacity in Chinese middle-aged and elderly people. Front Aging Neurosci. (2022) 14:919766. 10.3389/fnagi.2022.919766
65.
Nehlig A . Effects of coffee/caffeine on brain health and disease: what should i tell my patients?Pract Neurol. (2016) 16:89–95. 10.1136/practneurol-2015-001162
66.
Abreu RV Silva-Oliveira EM Moraes MFD Pereira GS Moraes-Santos T . Chronic coffee and caffeine ingestion effects on the cognitive function and antioxidant system of rat brains. Pharmacol Biochem Behav. (2011) 99:659–64. 10.1016/j.pbb.2011.06.010
67.
Kim JW Byun MS Yi D Lee JH Jeon SY Jung G et al . Coffee intake and decreased amyloid pathology in human brain. Transl Psychiatry. (2019) 9:270. 10.1038/s41398-019-0604-5
68.
Zhou A Taylor AE Karhunen V Zhan Y Rovio SP Lahtl J et al . Habitual coffee consumption and cognitive function: a mendelian randomization meta-analysis in up to 415,530 participants. Sci Rep. (2018) 8:7526. 10.1038/s41598-018-25919-2
69.
Araújo LF Mirza SS Bos D Niessen WJ Barreto SM van der Lugt A et al . Association of coffee consumption with mri markers and cognitive function: a population-based study. J Alzheimers Dis. (2016) 53:451–61. 10.3233/JAD-160116
70.
Kim KY Yun JM . Dietary patterns and mild cognitive impairment risk in Korean adults over 50 years old. Prev Nutr Food Sci. (2021) 26:132–45. 10.3746/pnf.2021.26.2.132
71.
Kim J Yu A Choi BY Nam J Kim MK Oh DH . Dietary patterns and cognitive function in Korean older adults. Eur J Nutr. (2015) 54:309–18. 10.1007/s00394-014-0713-0
72.
Vogel T Dali-Youcef N Kaltenbach G et al . Homocysteine, vitamin B12, folate and cognitive functions: a systematic and critical review of the literature. Int J Clin Pract. (2009) 63:1061–7. 10.1111/j.1742-1241.2009.02026.x
73.
Han L Jia J . Long-term effects of alcohol consumption on cognitive function in seniors: a cohort study in China. BMC Geriatr. (2021) 21:699. 10.1186/s12877-021-02606-y
74.
Ran LS Liu WH Fang YY Xu XB Li J Luo X et al . Alcohol, coffee and tea intake and the risk of cognitive deficits: a dose-response meta-analysis. Epidemiol Psychiatr Sci. (2021) 30:e13. 10.1017/S2045796020001183
75.
Lee Y Back JH Kim J Kim S-H Na DL Cheong H-K et al . Systematic review of health behavioral risks and cognitive health in older adults. Int Psychogeriatr. (2010) 22:174–87. 10.1017/S1041610209991189
76.
Kim JW Byun MS Yi D Lee JH Ko K Jeon SY et al . Association of moderate alcohol intake with in vivo amyloid-beta deposition in human brain: a cross-sectional study. PLoS Med. (2020) 17:e1003022. 10.1371/journal.pmed.1003022
77.
Lamport DJ Saunders C Butler LT Pe Spencer J . Fruits, vegetables, 100% juices, and cognitive function. Nutr Rev. (2014) 72:774–89. 10.1111/nure.12149
78.
Carrillo J Zafrilla MP Marhuenda J . Cognitive function and consumption of fruit and vegetable polyphenols in a young population: is there a relationship?Foods. (2019) 8:507. 10.3390/foods8100507
79.
Staubo SC Aakre JA Vemuri P Syrjanen JA Mielke MM Geda YE et al . Mediterranean diet, micronutrients and macronutrients, and mri measures of cortical thickness. Alzheimers Dement. (2017) 13:168–77. 10.1016/j.jalz.2016.06.2359
80.
Weinstein G Vered S Ivancovsky-Wajcman D Ravona-Springer R Heymann A Zelber-Sagi S et al . Consumption of ultra-processed food and cognitive decline among older adults with type-2 diabetes. J Gerontol A Biol Sci Med Sci. (2023) 78:134–42. 10.1093/gerona/glac070
81.
Barbaresko J Lellmann AW Schmidt A Lehmann A Amini AM Egert S et al . Dietary factors and neurodegenerative disorders: an umbrella review of meta-analyses of prospective studies. Adv Nutr. (2020) 11:1161–73. 10.1093/advances/nmaa053
82.
Noble EE Hsu TM Kanoski SE . Gut to brain dysbiosis: mechanisms linking western diet consumption, the microbiome, and cognitive impairment. Front Behav Neurosci. (2017) 11:9. 10.3389/fnbeh.2017.00009
83.
Wieckowska-Gacek A Mietelska-Porowska A Wydrych M Wojda U . Western diet as a trigger of Alzheimer's disease: from metabolic syndrome and systemic inflammation to neuroinflammation and neurodegeneration. Ageing Res Rev. (2021) 70:101397. 10.1016/j.arr.2021.101397
84.
Yian Gu JJM Schupf N Mayeux R . Sugary beverage consumption and risk of Alzheimer's disease in a community-based multiethnic population. Alzheimer Dement. (2018) 14:645. 10.1016/j.jalz.2018.06.2696
Summary
Keywords
dietary pattern, cognitive impairment, inflammation, hemodialysis, end-stage kidney disease
Citation
Zhuang Y, Wang X, Zhang X, Fang Q, Zhang X and Song Y (2023) The relationship between dietary patterns derived from inflammation and cognitive impairment in patients undergoing hemodialysis. Front. Nutr. 10:1218592. doi: 10.3389/fnut.2023.1218592
Received
13 May 2023
Accepted
13 July 2023
Published
03 August 2023
Volume
10 - 2023
Edited by
Aurora Serralde, National Institute of Medical Sciences and Nutrition Salvador Zubirán, Mexico
Reviewed by
Paola Vanessa Miranda Alatriste, National Institute of Medical Sciences and Nutrition Salvador Zubirán, Mexico; Samuel Ramos-Acevedo, National Institute of Medical Sciences and Nutrition Salvador Zubirán, Mexico
Updates
Copyright
© 2023 Zhuang, Wang, Zhang, Fang, Zhang and Song.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yan Song yansong@ntu.edu.cn
†These authors have contributed equally to this work and share first authorship
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.