Abstract
Based on existing systematic reviews and meta-analyse we conducted this comprehensive review to evaluate the quality, effectiveness, and bias of evidence regarding the relationship between probiotic intake and improved constipation outcomes in children. A total of nine meta-analyses and systematic reviews were extracted from 628 articles, summarizing seven effectiveness indicators and the incidence of adverse reactions in the treatment of constipation. According to the results, our study revealed that the intake of probiotics in children with FC significantly improved treatment success rate and defecation frequency, while decreased the recurrence rate of constipation. However, no significant association was detected between probiotics intake and frequency of abdominal pain, stool consistency, frequency of defecation pain, frequency of fecal incontinence of children with FC. The intake of probiotics did not increase the incidence of adverse reactions and demonstrated good safety.
Introduction
Constipation is a common childhood symptom, with functional constipation (FC) accounting for over 95% of all constipation cases (1). According to the latest diagnostic standard of the Rome IV criteria, FC is characterized by difficulty in defecation, reduced frequency of defecation, or a feeling of incomplete defecation (fecal incontinence), accompanied by abdominal pain, abdominal distension, etc. (2). With an increased fat intake and changes in people’s dietary structure, the prevalence of constipation in children has gradually increased. According to statistics, the global total prevalence of FC in children is about 9.5% (3). The occurrence of FC is related to various factors, including dietary fiber intake, water intake, the level of physical activity, and defecation training (1, 3). Functional constipation affects children’s lives and health. Currently, the etiology and pathogenesis underlying FC have not been elucidated in clinical studies, and there is no consensus on its treatment. Functional constipation not only causes distressing physical, emotional, and social effects in affected children but also negatively affects their families, increasing their medical burden and ultimately damaging the health-related quality of life of the child (2, 4). Relevant studies have also shown that about half of children with constipation remain symptomatic into adulthood, which has a serious impact on their education and daily life, and it has caused significant health problems (5). Therefore, new and more effective treatments for constipation in children are very important for healthy development in childhood.
The gut microbiota is crucial to human health, and its condition is related to many childhood diseases (6). Probiotics are a combination of living bacteria and yeast, including the well-known strains of Lactobacillus acidophilus, Bifidobacterium lactis, and L. brevis (4, 7). Probiotics are now defined as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host by the Food and Agriculture Organization of the United Nations and the World Health Organization (FAO/WHO) (8). This definition is inclusive of a broad range of microbes and applications, while capturing the essence of probiotics (microbial, viable and beneficial to health). The distinction between commensal microorganisms and probiotics is also inferred from this definition. Although commensals in the gut are often the source of probiotic strains, until these strains are isolated, characterized and a credible case presented for their health effects, they cannot be called ‘probiotics’ (8). The use of probiotics the probiotic may change the gut microbial composition and structure and thus can be applied to the clinical treatment of constipation, diarrhea, irritable bowel syndrome, etc., (9). The beneficial effects of probiotics on the intestine may be related to the following aspects: improving gastrointestinal peristalsis and decreasing intestinal transit time; competing with other harmful microorganisms in the intestine for nutrition; adhering to endothelial cells, preventing pathogens from entering the body through intestinal epithelial cells, and stimulating phagocytosis through lymphocyte activation (9, 10). Furthermore, probiotics can stimulate the production of cytokines (such as IgA and INF) and regulate cellular and humoral immunity, thereby affecting the body’s specific and non-specific immunity (2, 11). Their metabolites can lead to a decrease in intestinal PH and the acidification of intestinal contents, thus inhibiting the growth of certain pathogens (12).
Although probiotics can regulate intestinal microbiota and the microenvironment of the intestine, some studies suggest that currently there is insufficient evidence to support the use of probiotic for the treatment of FC in children (13, 14). Clinical randomized double blind controlled trials have shown insufficient evidence to support the use of probiotics for the treatment of FC in children (15–17). However, some studies suggest that the use of probiotic preparations is effective for children with FC. The randomized double-blind controlled trials of Lactobacillus and Bifidobacterium suggest effectiveness in treating chronic constipation in children (18, 19). A prospective, multicenter study conducted suggests that probiotic formulations can promote average daily bowel movements (20). Therefore, probiotic preparations have an improvement effect on FC in children theoretically. However, there is still controversy internationally about whether probiotic preparations are needed for children with FC. Therefore, we conducted this study to comprehensively review the association between probiotic intake and constipation improvement in children (as reported in our systematic review and meta-analysis) and assess the effectiveness of the existing evidence.
Materials and methods
Search strategy
We searched systematic reviews and meta-analyses of observational studies and interventional studies from the databases of PubMed, Embase, Cochrane Library, and China National Knowledge Infrastructure from their inception to January 6, 2023. The search strategy used a combination of the following terms: constipation AND probiotic, prebiotics, synbiotics, Lactobacillus, L. GG, L. acidophilus, L. rhamnosus, L. plantarum, L. casei, L. gasseri, L. reuteri, L. lactis, Bifidobacterium, B. breve, B. longum, B. infantis, B. adolescentis, B. lactis, Bacillus, Clostridium butyricum, Streptococcus thermophilus, Escherichia coli, Propionibacterium freundendsreichii, Enterococcus SF68, Enterococcus SF68, Enterococcus faecalis, Saccharomyces boulardi, and VSL#3 AND systematic review, meta-analysis and review. No restrictions or filters were applied to the search process. We also manually searched the cited references of the retrieved articles and reviews. Two authors (DMY and WS) independently conducted the literature search. Any disagreements between the two researchers in terms of article selection were resolved by a third investigator (CPJ). Details of the search strategy are provided in Supplementary Table S1.
Eligibility criteria
Meta-analyses and systematic reviews evaluating probiotic intake and constipation in children with outcomes in children were included regardless of the race, gender, country, or region of the participants. Based on these studies, indicators of effectiveness and safety were summarized. If two or more constipation outcomes existed in a single article, the data of each outcome were extracted separately. If one outcome was assessed in more than one study, articles with the largest number of participants were included. Research with unrelated research content, design that was not meet standards, or incomplete research data were excluded.
Data extraction
Two authors (DMY and WYT) independently extracted data, and disagreements were resolved by consensus. The following data were extracted from the eligible studies: name of the first author; journal; year of publication; type of comparisons; type of studies included (RCT, or non-RCT); number of primary studies; follow-up time; number of participants in each study; estimated summary effect and corresponding 95% confidence intervals (CIs); outcome. Any difference was resolved by the third investigator (CPJ).
Assessment of methodological quality of included studies and quality of evidence
The methodological quality of each article was evaluated based on AMSTAR-2 (A Measurement Tool to Assess Systematic Reviews 2) items, which is a reliable strategy for evaluating the quality of systematic reviews and meta-analyses (21). The GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) approach was used to evaluate the strength of evidence for each outcome proposed in the meta-analysis and to classify the evidence into “high,” “moderate,” “low,” and “very low” quality to enable recommendations to be made (22).
Statistical analysis
We extracted data on probiotic intake for improving constipation in children and estimated the overall efficacy using the 95% CI reported in each study (if available). Heterogeneity between studies was assessed using I2 statistics. Values <50% indicated acceptable heterogeneity, values >50% suggested moderate heterogeneity, and values >75% were indicative of high heterogeneity (23). Egger’s regression asymmetry test was used to calculate an estimate of publication bias for any re-analysis that included at least 10 studies, which was considered indicative of small-study effects. A value of p < 0.1 was considered statistically significant according to Egger’s test (24).In addition, p < 0.05 was regarded as significant for other tests.
Results
Characteristics of the included meta-analyses
The detailed process of literature retrieval and selection is presented in Figure 1. We searched 628 articles and ultimately identified nine meta-analyses based on the inclusion and exclusion criteria. The effectiveness and safety indicators of using probiotics in children with constipation were summarized. Seven indicators of constipation were used as a reference for evaluating the effectiveness of probiotic therapy and were extracted from all eligible studies. In terms of safety, information on whether probiotics increased the incidence of adverse reactions in children was extracted. The effectiveness and safety results of probiotic intake in children with constipation are shown in Table 1. The median number of primary studies was six (interquartile range: 2–7), and the median number of cases was 467 (interquartile range: 111–467).
Figure 1
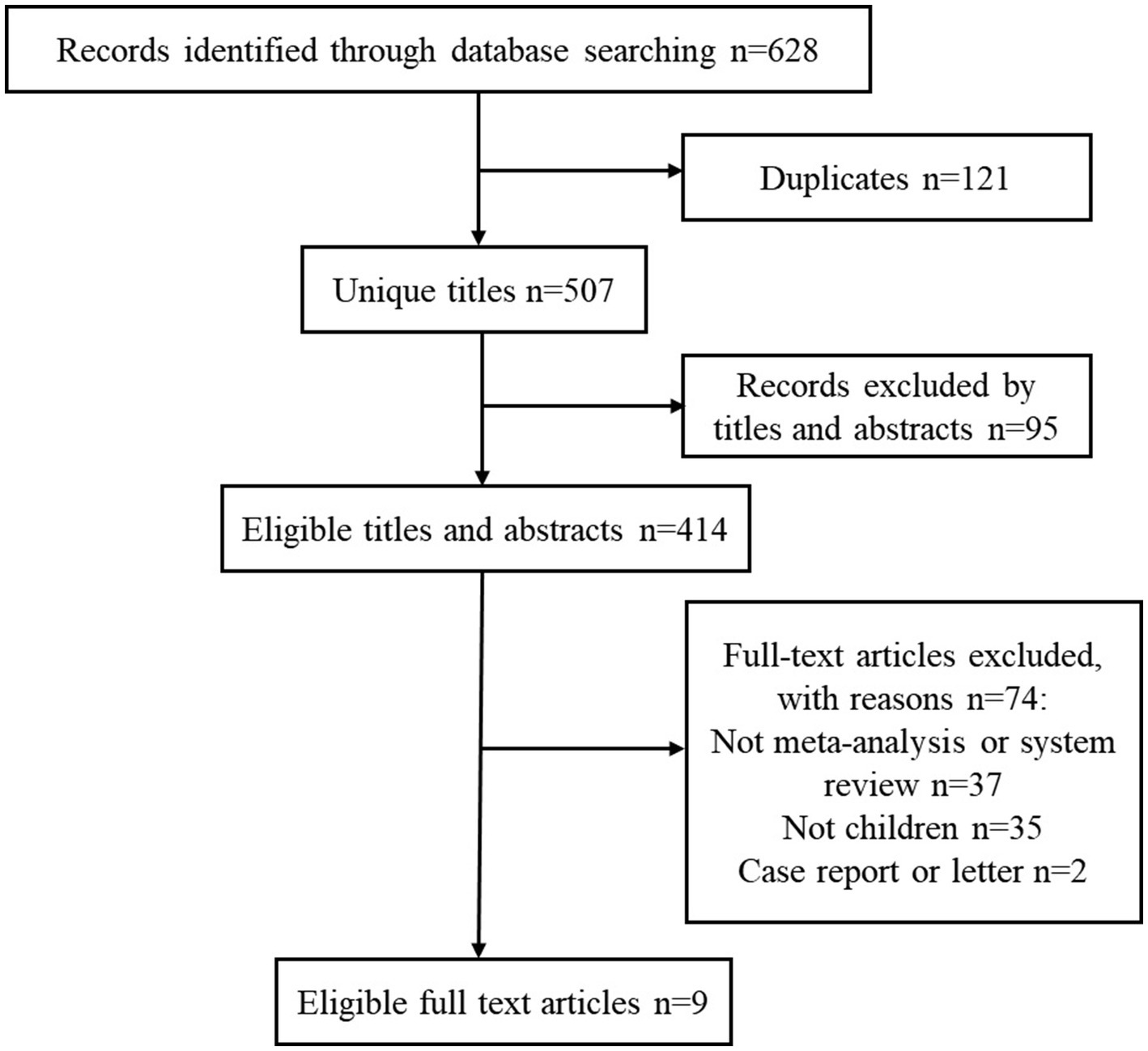
Flowchart of the systematic search and selection process.
Table 1
| Source | Item 1 | Item 2 | Item 3 | Item 4 | Item 5 | Item 6 | Item 7 | Item 8 | Item 9 | Item 10 | Item 11 | Item 12 | Item 13 | Item 14 | Item 15 | Item 16 | Overall quality |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Junli Wang (25) | Y | PY | Y | PY | Y | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | N | Moderate |
| Hao Li (26) | Y | PY | Y | PY | Y | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | N | Moderate |
| Tabbers (3) | Y | PY | Y | PY | Y | Y | N | Y | N | N | Y | N | N | Y | N | N | Critically Low |
| Wojtyniak (27) | Y | Y | Y | PY | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | High |
| Jin (28) | Y | PY | Y | Y | Y | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | N | Moderate |
| Gomes (29) | Y | PY | Y | Y | Y | Y | Y | Y | N | Y | Y | N | N | Y | N | Y | Critically Low |
| Huang (30) | Y | PY | Y | PY | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | High |
| Wegh (31) | Y | PY | Y | PY | Y | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | N | Moderate |
| Chmielewska (32) | Y | PY | Y | PY | Y | Y | Y | Y | N | N | Y | Y | N | Y | N | N | Critically Low |
The methodological quality of included meta-analysis using AMSTAR-2.
Rationale for selection of items:
1. Did the research of questions and inclusion criteria for the review include the components of PICO?.
2. Did the report of the review contain an explicit statement that the review methods were established prior to the conduct of the review and did the report justify any significant deviations from the protocol?.
3. Did the review authors explain their selection of the study designs for inclusion in the review?.
4. Did the review authors use a comprehensive literature search strategy?.
5. Did the review authors perform study selection in duplicate?.
6. Did the review authors perform data extraction in duplicate?
Di7. d the review authors provide a list of excluded studies and justify the exclusions?.
8. Did the review authors describe the included studies in adequate detail?.
9. Did the review authors use a satisfactory technique for assessing the risk of bias (RoB) in individual studies that were included in the review?.
10. Did the review authors report on the sources of funding for the studies included in the review?.
11. If meta-analysis was performed, did the review authors use appropriate methods for statistical combination of results?.
12. If meta-analysis was performed, did the review authors assess the potential impact of RoB in individual studies on the results of the meta-analysis or other evidence synthesis?.
13. Did the review authors account for RoB in primary studies when interpreting discussing the results of the review?.
14. Did the review authors provide a satisfactory explanation for, and discussion of, any heterogeneity observed in the results of the review?.
15. If the performed quantitative synthesis did the review authors carry out an adequate investigation of publication bias (small study bias) and discuss its likely impact on the results of the review?.
16. Did the review authors report any potential sources of conflict of interest, including any funding they received for conducting the review?.
Y, yes; N, no; PY, partial yes.
Description and summary of associations
The associations analyzed included seven clinical effect outcomes and one adverse event. The effectiveness indicators of probiotic preparations in the treatment of FC in children were reported in the nine included articles and included the following: treatment success rate, defecation frequency, frequency of abdominal pain, stool consistency, frequency of defecation pain, frequency of fecal incontinence, and recurrence rate. The incidence of adverse reactions was analyzed in terms of safety (Table 1).
Treatment success rate
Treatment success is usually defined as defecation ≥3 bowel movements per week. According to six studies (3, 26, 27, 29, 31, 32), probiotic preparations could significantly improve the level of treatment success compared with placebos or other interventions. There was significant difference in the treatment success of probiotics and placebos (OR = 4.81, 95% CI: 2.32–9.97, p < 0.0001) (26). Most of the RCTs are based on single-strain or mix-strain studies. Research has shown that the combination of Lactobacillus and Enterococcus can improve the success rate of treatment,and the average effective cumulative dose for children under 1 year old needs to reach 1.05 × 109 CFU can improve the success rate of FC treatment in children, while the average effective cumulative dose for children over 1 year old needs to reach 1.89 × 109 CFU (26). Two RCT reported that L. casei rhamnosus Lcr35, but not L. rhamnosus GG, showed a beneficial effect in children (18, 33).
Defecation frequency
Seven studies (3, 25, 27–31) showed that the use of probiotics in children with constipation could significantly increase the frequency of defecation. There was significant difference in terms of defecation frequency improvement between probiotic treatment groups and control groups (MD = 0.73, 95% CI: 0.14–1.31, p = 0.02), however, there was significant heterogeneity (I2 = 80%, p = 0.02). Subgroup assessments showed that Asian children had a significantly higher stool frequency with probiotic treatment (30). The RCTs found that L reuteri and Bifidobacterium longum could significantly increase the frequency of bowel movements compared with placebo (19, 34).
Recurrence rate
Only one study showed that compared with the use of a placebo, the administration of probiotics significantly reduced the recurrence rate of FC, and the heterogeneity was considerable (OR = 0.19, 95% CI: 0.05–0.68, p = 0.01) (26). Therefore, probiotic treatment may reduce the recurrence rate of functional constipation in children.
Frequency of abdominal pain
Six studies (3, 25, 27–29, 31) reported different results for reducing the frequency of abdominal pain and improving the symptoms of abdominal pain in children with FC after using probiotics. Tabbers et al. (3) found that probiotics significantly reduced abdominal pain compared with osmotic laxatives (1.9 episodes with probiotics vs. 4.8 episodes with osmotic laxatives; p = 0.04). Gomes et al. (29) showed that in the first week after probiotic intervention, a lower frequency (p = 0.017) of abdominal pain was found in a treatment group compared with that in a control group. The remaining four studies showed that there was no significant difference in terms of reducing the incidence of abdominal pain between probiotic treatment groups and control groups. There was no significant difference between the control and probiotic groups in the frequency of abdominal pain at the end of the intervention (OR = 1.09, 95% CI: 0.65–1.82) (25).
Stool consistency
Three studies (29–31) showed that probiotics had no significant effect on improving stool consistency in children. Stool consistency was not significantly different between probiotic groups and control groups (MD = −0.07, 95% CI: −0.21–0.06, p = 0.27) (30).
Frequency of defecation pain
Three studies (28, 29, 31) showed that compared with the results in a control group, the symptoms and frequency of defecation pain in children in a probiotic group did not improve (RR = 1.16, 95% CI: 0.81–1.66, p = 0.41) (28). Pooled results of two RCTs (n = 108) (5, 33) showed no difference between the L. casei rhamnosus Lcr35 and control groups in the frequency of abdominal pain (RR = 2.08, 95%: CI 0.19–23.37), heterogeneity was considerable (χ2 = 18.16; p < 0.0001; I2 = 94%) (18, 33).
Frequency of fecal incontinence
The frequency of fecal incontinence was reported in three studies (27–29). There was no significant effect with probiotics compared with placebos on the frequency of fecal incontinence at the end of intervention (MD = −0.05, 95% CI: −0.63–0.53), and no significant heterogeneity was found (χ2 = 0.32; p = 0.57; I2 = 0%) (27). Based on the pooled results of two RCTs, there was no significant effect of L. casei rhamnosus Lcr35 compared with placebo on the frequency of fecal incontinence at the end of intervention (MD = 0.05, 95% CI: −0.63–0.53); no significant heterogeneity was found (χ2 = 0.32; p = 0.57; I2 = 0%) (18, 33).
Adverse reaction rate
Four studies (3, 27, 28, 32) showed no significant difference in the incidence of adverse reactions in the digestive system between probiotic treatment groups and control groups. Chmielewska (32) demonstrated that the probiotics formulation used was tolerable in children, and no adverse event related to it was reported in any trial.
Adverse events were similar in the experimental and control groups (RR 0.58, 95% CI 0.25 to 1.31). No significant heterogeneity was found (χ2 = 1.01; p = 0.6; I2 = 0%). The most frequently occurring adverse events were abdominal pain, vomiting, and gastroenteritis.
Heterogeneity and publication bias of included studies
Of all 8 associations, 4 had acceptable heterogeneity (<50%), and another 4 had significant heterogeneity (>50%). Four studies of included studies were reported to have significant publication bias, whereas this was not detected in the other five studies.
Methodological quality
The AMSTAR 2 results for each study are presented in Table 2. Among the nine studies, four (44.4%) were identified as medium in the methodological quality assessment, and three were identified as extremely low [two studies (22.2%)] or low [one study (11.1%)]. The most common key flaws were the absence of detailed literature exclusion lists and funding sources and the failure to consider the risks of bias and heterogeneity when preparing conclusions and recommendations. Evidence of frequency of fecal incontinence and adverse events incidence showed “high” quality according to the GRADE classification, and the others were classified as “moderate” quality. No “low” quality was observed.
Table 2
| Outcome | Source | Year | No.of primary studies | Duration | No. of cases/total | Metric | Estimates | 95%CI | value of p | I2 | GRADE rating | AMSTAR-2 rating |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Significant associations | ||||||||||||
| Treatment success | Li | 2016 | 9 | 2–8 wks | 345/67 | OR | 4.81 | 2.32–9.97 | <0.0001 | 66% | Moderate | Moderate |
| Defecation frequency | Huang | 2017 | 6 | 3–12 wks | 231/444 | MD | 0.73 | 0.14–1.31 | 0.02 | 84% | Moderate | High |
| Recurrence rate | Li | 2016 | 4 | 2–8 wks | 144/243 | OR | 0.19 | 0.05–0.68 | 0.01 | 62% | Moderate | Moderate |
| Non-significant associations | ||||||||||||
| Stool consistency | Huang | 2017 | 3 | 3–8 wks | 133/267 | MD | −0.07 | −0.21-0.06 | 0.27 | 92% | Moderate | High |
| Frequency of abdominal pain | Wang | 2014 | 3 | 3–8 wks | 152/293 | OR | 1.09 | 0.65–1.82 | 0.75 | 0% | Moderate | Moderate |
| Frequency of defecation pain | Wang | 2014 | 2 | 3–8 wks | 108/209 | OR | 1.16 | 0.79–2.72 | 0.23 | 0% | Moderate | Moderate |
| Frequency of fecal incontinence | Wojtyniak | 2017 | 5 | 3–12 wks | 212/462 | MD | −0.05 | −0.63-0.53 | 0.57 | 0% | High | High |
| Adverse events | Wojtyniak | 2017 | 6 | 3–12 wks | 234/462 | RR | 0.58 | 0.83–1.62 | 0.6 | 0% | High | High |
Associations between probiotics intake and constipation in children outcome.
Discussion
Probiotics are often used in the clinical treatment of constipation in children, and many meta-analyses and systematic reviews of their efficacy have been conducted. On this basis, we conducted an umbrella review to analyze whether probiotics can improve constipation in children. We analyzed a total of nine meta-analyses and systematic reviews and summarized seven indicators of effectiveness in treating constipation. According to the results, our study revealed that the intake of probiotics in children with FC significantly improved treatment success rate and defecation frequency, while decreased the recurrence rate of constipation. However, no significant association was detected between probiotics intake and frequency of abdominal pain, stool consistency, frequency of defecation pain, frequency of fecal incontinence of children with FC. The incidence of adverse reactions was low and the safety was good when consuming probiotics. These results suggest that probiotic intake did improve constipation in children, although they cannot improve some symptoms of constipation in children.
As we know, probiotics can regulate the gut microbiota. Intestinal microecology consists of intestinal microbiota, intestinal epithelial cells, and intestinal mucosal immune system (35). The gut microbiome is comprised of the collective genome of microbes inhabiting the gut including bacteria, archaea, viruses, and fungi, including the genes and genomes of the microbiota, as well as the products of the microbiota and the host environment. Microbiome comprises all of the genetic material within a microbiota (36). The difference between the intestinal microbiota of the population in general may be the cause of constipation. The composition of intestinal microbiota varies from person to person and is affected by a range of factors, including diet, living environment, and drugs (34). And there are also differences among different populations.
The pivotal role of intestinal microbiota in the occurrence and development of constipation has prompted a shift in therapeutic options toward microecological intervention, especially probiotics, which has gradually replaced the traditional approaches for treating constipation (37). Those most widely studies are organisms within the genera Bifidobacterium and Lactobacillus. There is still controversy internationally about whether probiotics are needed for children with FC, and some research results indicate that probiotic preparations have an improvement effect on children with FC. In the study of probiotic therapy for FC in children, most studies have heterogeneity in terms of study population, probiotic strains, dosage, study duration, and follow-up. Tabbers et al. (15) indicated that both fermented dairy product containing Bifidobacterium lactis DN-173010 and control product could improve stool frequency from baseline to after 3 weeks, with no significant difference between both, and that no serious adverse events were observed in children with FC. Wojtyniak et al. (33) suggested that Lactobacillus casei rhamnosus was not associated with significant improvement in symptoms in children with FC aged less than 5 years and did not recommend the use of probiotics in children with FC. While the administration of L. casei rhamnosus Lcr35 augmented the number of stools and reduced the number of hard stools. Again, although the results were statistically signifcant, the overall effects were clinically modest. All of the conclusions are based on single studies, some of which had a very small number of participants and methodological limitations. The conclusions should be interpreted with great caution. Repeat studies with the probiotic strains that have been proven effective are needed. A paucity of data did not allow us to conclude whether any particular probiotic is more effective than another. In terms of microbial alterations in adults, the analysis of the difference in efficacy of probiotic subgroups shows that B.lactis of Bifidobacterium can significantly improve the rectosigmoid transit time, defecation frequency, hard stools, flatulence in patients with chronic constipation, while L.casei Shirota of Lactobacillus has no obvious therapeutic effect (38, 39). It is worth noting that the analysis of this research for different microbiota subgroups has high heterogeneity. However, another meta-analysis of a controlled study on adults with FC found that probiotic therapy can significantly improve symptoms in patients with FC, but the impact of B. lactis on treatment efficacy is not significant. In addition, the study also pointed out that increasing the variety of microbiota can significantly improve the symptoms of FC (40). This suggests that multi-strain probiotic mixtures may be more effective in treating FC than single-strain probiotic. Due to the differences in function and immunogenicity of the microbiota, the impact on the host may not be the same among different strains of the same species, and there are significant differences in the therapeutic effects of different strains of the same species on FC.
There are significant differences in the dosage of probiotic therapy among various researches. A RCT targeting constipation patients administered two different doses of B. lactis and placebo, after 2 weeks of treatment, probiotics showed a dose-dependent reduction in the whole gut transit time compared to placebo, indicating that high-dose probiotic therapy is more beneficial for constipation patients (41).The results of probiotic treatment are based on occupancy effects, only when a sufficient dose of probiotics is given can the beneficial microbiota be effectively restored. In addition, due to the limited colonization sites in the intestine, excess probiotic cannot be fully colonized. Therefore, even if excessive probiotic treatment is used, its beneficial effects on host health are very limited. There are alsodifferences in the treatment course of probiotics for FC among different researches, ranging from 2 to 12 weeks. The current research on whether different intervention times have an impact on the effectiveness of probiotics in treating FC is not clear, which requires further research to answer.
The gut microbiota may be involved in the production or aggravation of constipation. In adults, experimental studies have shown that constipation is often associated with gut microbiota dysbiosis, consisting of the modified abundance of certain taxa of the colonic microbiome (39). For example, some data have suggested the decreased abundance of Bifidobacteria, Lactobacillus, Bacteroides, and Prevotella (42). Research has shown that there is an increasing trend in the relative abundance of Bifidobacteria and Lactobacilli in adolescent FC patients (43). In children, one recent study showed that in those with FC, the most discriminative species were Bacteroides fragilis, Bacteroides ovatus, Bifidobacterium longum, Parabacteroides species (increased), and Alistipes finegoldii (decreased) (44). Although research has found that probiotics do not significantly alleviate symptoms of FC in children, meta-analysis of adult FC patients has shown that probiotics treatment can effectively reduce the whole gut transit time and rectosigmoid transit time, increase defecation frequency, reduce difficulty with evacuation, bloating, abdominal pain or discomfort and hard stools (38). Therefore, we speculate that the difference in efficacy may be related to different microecological imbalances in adult and child FC patients. Studies have shown that probiotics have a certain role in diseases such as diarrhea, constipation, inflammatory bowel disease, and irritable bowel syndrome (45, 46). Probiotics can effectively regulate multiple aspects of the pathogenesis of FC. First, probiotics can regulate the composition of the gut microbiota, which can be divided into three categories based on physiological functions: 1. physiological microbiota, mainly including obligate anaerobes, such as Bifidobacterium and Lactobacillus, which are the dominant gut microbiota and play a protective role in the normal physiological function of the intestine; 2. opportunistic pathogens, including E. coli, Enterococcus, and Clostridium, are mostly obligate anaerobes that only affect the host under certain special conditions; and 3. pathogenic bacteria, mostly Proteus, can cause human diseases if they are colonized in the intestinal tract for a long time and multiply in large numbers (47–49). On the one hand, probiotics compete for nutrients to produce metabolites and soluble factors, such as lactic acid, short-chain fatty acids (bacteria), and hydrogen peroxide, which affect the growth of pathogenic bacteria. On the other hand, by promoting the production of mucoprotein and reducing the adhesion of pathogenic bacteria, probiotics have a protective effect on pathogens and improve the intestinal ecosystem (50, 51). Second, constipation is associated with impaired intestinal motility and gas retention, while probiotics may improve irregular intestinal peristalsis and flatulence (52). The mechanism by which the gut microbiota promotes intestinal peristalsis is not yet clear and may be related to the regulatory effects of probiotics on SCFAs and 5-HT (53, 54). Opportunistic pathogens, such as Bifidobacterium and Lactobacillus, can ferment and degrade in the intestine, reduce intestinal pH, and produce metabolites, including SCFAs. Mainly, SCFAs include acetic acid, propanoic acid, butyric acid, etc. (55). SCFAs can reduce intestinal pH, promote colonic peristalsis, and effectively reduce the retention time of feces in the colon. A low-PH environment can also promote the setting of obligate anaerobes in the intestinal tract via positive feedback regulation, enabling SCFAs to competitively bind to enterocyte or receptor binding sites on the mucosa (56, 57). This improves colonization resistance, inhibits colonization and reproduction by opportunistic pathogens and pathogenic bacteria, improves intestinal microbiota disorder in patients with constipation, maintains a normal intestinal microbiota structure, and helps prevent and alleviate constipation and symptoms caused by intestinal microbiota disorder (58, 59).As far as we know, the role of probiotics in treating childhood constipation is uncertain due to conflicting research results. Whether probiotics can improve constipation in children is currently controversial; It is worth noting that the present study is the first comprehensive summary and evaluation based on the available evidence as to whether probiotic intake can improve constipation in children. Standard tools were used to assess the methodological quality (AMSTAR) and strength of evidence (GRADE) of the included literature (17, 18). However, although methodological patterns were used correctly, selection bias may still exist. To minimize this bias to the greatest extent possible, two authors applied the above methods. According to our systematic analysis of the research, the use of probiotics can significantly improve constipation in children,although probiotics do not significantly improve some symptoms of FC. Moreover, the safety of probiotics is tolerable for children.
Several limitations exist in our study. First, only two of the included studies were classified as high quality according to the AMSTAR-2 method due to most meta-analyses being based on observational studies. Second, differences in the type, dosage, and duration of probiotic use may have affected the research results. Third, the diet and physical condition of children may affect the therapeutic effect of probiotics. Finally, the simultaneous use of other drugs can also affect the outcome of treatment. In studies on probiotic therapy in children, the sample size is generally not large enough, which may also have affected our conclusions. Considering this study’s shortcomings, further high-quality research on this topic is needed.
Funding
This study was supported by 345 Talent Project of Shengjing Hospital of China Medical University.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Statements
Author contributions
MD and YW conducted this research and wrote the paper. SW and MZ designed the study and had primary responsibility for final content. PC and ZZ provided essential materials and analyzed data. All authors contributed to the article and approved the submitted version.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fnut.2023.1218909/full#supplementary-material
References
1.
MugieSMDi LorenzoCBenningaMA. Constipation in childhood. Nat Rev Gastroenterol Hepatol. (2011) 8:502–11. doi: 10.1038/nrgastro.2011.130
2.
KhanL. Constipation Management in Pediatric Primary Care. Pediatr Ann. (2018) 47:e180–4. doi: 10.3928/19382359-20180426-02
3.
TabbersMMBenningaMA. Constipation in children: fibre and probiotics. BMJ Clin Evid. (2015) 2015:0303.
4.
SadeghzadehMRabieefarAKhoshnevisaslPMousavinasabNEftekhariK. The effect of probiotics on childhood constipation: a randomized controlled double blind clinical trial. Int J Pediatr. (2014) 2014:937212. doi: 10.1155/2014/937212
5.
HawaKJanseSLeeJBaliNVazKYacobDet al. Providing a constipation action plan to families of children with constipation decreases health care utilization. J Pediatr Gastroenterol Nutr. (2022) 75:589–94. doi: 10.1097/MPG.0000000000003593
6.
HojsakI. Probiotics in functional gastrointestinal disorders. Adv Exp Med Biol. (2019) 1125:121–37. doi: 10.1007/5584_2018_321
7.
SasaranMOMargineanCOAdumitrachioaieiHMelitLE. Pathogen-specific benefits of probiotic and Synbiotic use in childhood acute gastroenteritis: an updated review of the literature. Nutrients. (2023) 15:15030643. doi: 10.3390/nu15030643
8.
HillCGuarnerFReidGGibsonGRMerensteinDJPotBet al. Expert consensus document. The international scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. (2014) 11:506–14. doi: 10.1038/nrgastro.2014.66
9.
BauMMorettiABertoniEVazzolerVLuiniCAgostiMet al. Risk and protective factors for gastrointestinal symptoms associated with antibiotic treatment in children: a population study. Pediatr Gastroenterol Hepatol Nutr. (2020) 23:35–48. doi: 10.5223/pghn.2020.23.1.35
10.
CaoLJiZZhangPWangJ. Epidemiology and mortality predictors for severe childhood community-acquired pneumonia in ICUs: a retrospective observational study. Front Pediatr. (2023) 11:1031423. doi: 10.3389/fped.2023.1031423
11.
KubotaMItoKTomimotoKKanazakiMTsukiyamaKKubotaAet al. Lactobacillus reuteri DSM 17938 and magnesium oxide in children with functional chronic constipation: a double-blind and randomized clinical trial. Nutrients. (2020) 12:12010225. doi: 10.3390/nu12010225
12.
SchwengerEMTejaniAMLoewenPS. Probiotics for preventing urinary tract infections in adults and children. Cochrane Database Syst Rev. (2015) 2015:CD008772. doi: 10.1002/14651858.CD008772.pub2
13.
Alvarez-CalatayudGPerez-MorenoJTolinMSanchezC. Clinical applications of the use of probiotics in pediatrics. Nutr Hosp. (2013) 28:564–74. doi: 10.3305/nh.2013.28.3.6603
14.
TabbersMMDiLorenzoCBergerMYFaureCLangendamMWNurkoSet al. Evaluation and treatment of functional constipation in infants and children: evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr. (2014) 58:258–74. doi: 10.1097/MPG.0000000000000266
15.
TabbersMMChmielewskaARoseboomMGCrastesNPerrinCReitsmaJBet al. Fermented milk containing Bifidobacterium lactis DN-173 010 in childhood constipation: a randomized, double-blind, controlled trial. Pediatrics. (2011) 127:e1392–9. doi: 10.1542/peds.2010-2590
16.
BanaszkiewiczASzajewskaH. Ineffectiveness of Lactobacillus GG as an adjunct to lactulose for the treatment of constipation in children: a double-blind, placebo-controlled randomized trial. J Pediatr. (2005) 146:364–9. doi: 10.1016/j.jpeds.2004.10.022
17.
TabbersMMde MillianoIRoseboomMGBenningaMA. Is Bifidobacterium breve effective in the treatment of childhood constipation? Results from a pilot study. Nutr J. (2011) 10:19. doi: 10.1186/1475-2891-10-19
18.
BuLNChangMHNiYHChenHLChengCC. Lactobacillus casei rhamnosus Lcr35 in children with chronic constipation. Pediatr Int. (2007) 49:485–90. doi: 10.1111/j.1442-200X.2007.02397.x
19.
GuerraPVLimaLNSouzaTCMazochiVPennaFJSilvaAMet al. Pediatric functional constipation treatment with Bifidobacterium-containing yogurt: a crossover, double-blind, controlled trial. World J Gastroenterol. (2011) 17:3916–21. doi: 10.3748/wjg.v17.i34.3916
20.
IndrioFDi MauroARiezzoG. Prophylactic use of a probiotic in the prevention of colic, regurgitation, and functional constipation--reply. JAMA Pediatr. (2014) 168:778. doi: 10.1001/jamapediatrics.2014.368
21.
SheaBJReevesBCWellsGThukuMHamelCMoranJet al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. (2017) 358:j4008. doi: 10.1136/bmj.j4008
22.
GuyattGHOxmanADVistGEKunzRFalck-YtterYAlonso-CoelloPet al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. (2008) 336:924–6. doi: 10.1136/bmj.39489.470347.AD
23.
ZhuJYuXZhengYLiJWangYLinYet al. Association of glucose-lowering medications with cardiovascular outcomes: an umbrella review and evidence map. Lancet Diabetes Endocrinol. (2020) 8:192–205. doi: 10.1016/S2213-8587(19)30422-X
24.
DemurtasJCelottoSBeaudartCSanchez-RodriguezDBalciCSoysalPet al. The efficacy and safety of influenza vaccination in older people: an umbrella review of evidence from meta-analyses of both observational and randomized controlled studies. Ageing Res Rev. (2020) 62:101118. doi: 10.1016/j.arr.2020.101118
25.
JunliWTaoCFengL. Meta analysis of the therapeutic effect of probiotics on functional constipation in children and adolescents. J Educ Bio. (2014) 2:174–9. doi: 10.3969/j.issn.2095-4301.2014.03.006
26.
HaoLYongzhiY. Yang Rong, et al Meta analysis of the clinical efficacy of bifidobacterium triple viable capsules/powders in the treatment of functional constipation in children. Chinese. J Microbiol. (2016) 28:1034–9. doi: 10.12281/cnki.cjm.201600007
27.
WojtyniakKSzajewskaH. Systematic review: probiotics for functional constipation in children. Eur J Pediatr. (2017) 176:1155–62. doi: 10.1007/s00431-017-2972-2
28.
JinLDengLWuWWangZShaoWLiuJ. Systematic review and meta-analysis of the effect of probiotic supplementation on functional constipation in children. Medicine (Baltimore). (2018) 97:e12174. doi: 10.1097/MD.0000000000012174
29.
GomesDMoraisMB. Gut microbiota and the use of probiotics in constipation in children and adolescents: systematic review. Rev Paul Pediatr. (2020) 38:e2018123. doi: 10.1590/1984-0462/2020/38/2018123
30.
HuangRHuJ. Positive effect of probiotics on constipation in children: a systematic review and Meta-analysis of six randomized controlled trials. Front Cell Infect Microbiol. (2017) 7:153. doi: 10.3389/fcimb.2017.00153
31.
WeghCAMBenningaMATabbersMM. Effectiveness of probiotics in children with functional abdominal pain disorders and functional constipation: a systematic review. J Clin Gastroenterol. (2018) 52 Proceedings from the 9th Probiotics, Prebiotics and New Foods, Nutraceuticals and Botanicals for Nutrition & Human and Microbiota Health Meeting, held in Rome, Italy from September 10 to 12, 2017:S10–26. doi: 10.1097/MCG.0000000000001054
32.
ChmielewskaASzajewskaH. Systematic review of randomised controlled trials: probiotics for functional constipation. World J Gastroenterol. (2010) 16:69–75. doi: 10.3748/wjg.v16.i1.69
33.
WojtyniakKHorvathADziechciarzPSzajewskaH. Lactobacillus casei rhamnosus Lcr35 in the Management of Functional Constipation in children: a randomized trial. J Pediatr. (2017) 184:e1:101–105.e1. doi: 10.1016/j.jpeds.2017.01.068
34.
CoccorulloPStrisciuglioCMartinelliMMieleEGrecoLStaianoA. Lactobacillus reuteri (DSM 17938) in infants with functional chronic constipation: a double-blind, randomized, placebo-controlled study. J Pediatr. (2010) 157:598–602. doi: 10.1016/j.jpeds.2010.04.066
35.
FuQSongTMaXCuiJ. Research progress on the relationship between intestinal microecology and intestinal bowel disease. Animal Model Exp Med. (2022) 5:297–310. doi: 10.1002/ame2.12262
36.
BergGRybakovaDFischerDCernavaTVergesMCCharlesTet al. Microbiome definition re-visited: old concepts and new challenges. Microbiome. (2020) 8:103. doi: 10.1186/s40168-020-00875-0
37.
PanRWangLXuXChenYWangHWangGet al. Crosstalk between the gut microbiome and colonic motility in chronic constipation: potential mechanisms and microbiota modulation. Nutrients. (2022) 14:14183704. doi: 10.3390/nu14183704
38.
DimidiEChristodoulidesSFragkosKCScottSMWhelanK. The effect of probiotics on functional constipation in adults: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. (2014) 100:1075–84. doi: 10.3945/ajcn.114.089151
39.
AttaluriAJacksonMValestinJRaoSS. Methanogenic flora is associated with altered colonic transit but not stool characteristics in constipation without IBS. Am J Gastroenterol. (2010) 105:1407–11. doi: 10.1038/ajg.2009.655
40.
ZhangCJiangJTianFZhaoJZhangHZhaiQet al. Meta-analysis of randomized controlled trials of the effects of probiotics on functional constipation in adults. Clin Nutr. (2020) 39:2960–9. doi: 10.1016/j.clnu.2020.01.005
41.
WallerPAGopalPKLeyerGJOuwehandACReiferCStewartMEet al. Dose-response effect of Bifidobacterium lactis HN019 on whole gut transit time and functional gastrointestinal symptoms in adults. Scand J Gastroenterol. (2011) 46:1057–64. doi: 10.3109/00365521.2011.584895
42.
KhalifILQuigleyEMKonovitchEAMaximovaID. Alterations in the colonic flora and intestinal permeability and evidence of immune activation in chronic constipation. Dig Liver Dis. (2005) 37:838–49. doi: 10.1016/j.dld.2005.06.008
43.
ZhuLLiuWAlkhouriRBakerRDBardJEQuigleyEMet al. Structural changes in the gut microbiome of constipated patients. Physiol Genomics. (2014) 46:679–86. doi: 10.1152/physiolgenomics.00082.2014
44.
de MeijTGde GrootEFEckABuddingAEKneepkensCMBenningaMAet al. Characterization of microbiota in children with chronic functional constipation. PLoS One. (2016) 11:e0164731. doi: 10.1371/journal.pone.0164731
45.
YueYYeKLuJWangXZhangSLiuLet al. Probiotic strain Lactobacillus plantarum YYC-3 prevents colon cancer in mice by regulating the tumour microenvironment. Biomed Pharmacother. (2020) 127:110159. doi: 10.1016/j.biopha.2020.110159
46.
WangTWangPGeWShiCXiaoGWangXet al. The probiotic Companilactobacillus crustorum MN047 alleviates colitis-associated tumorigenesis via modulating the intestinal microenvironment. Food Funct. (2021) 12:11331–42. doi: 10.1039/d1fo01531a
47.
CatassiCKryszakDLouis-JacquesODuerksenDRHillICroweSEet al. Detection of celiac disease in primary care: a multicenter case-finding study in North America. Am J Gastroenterol. (2007) 102:1454–60. doi: 10.1111/j.1572-0241.2007.01173.x
48.
ChenBKnowlesCHScottMAnandPWilliamsNSMilbrandtJet al. Idiopathic slow transit constipation and megacolon are not associated with neurturin mutations. Neurogastroenterol Motil. (2002) 14:513–7. doi: 10.1046/j.1365-2982.2002.00354.x
49.
DengQZhangLLiuXKangLYiJRenJet al. COF-based artificial probiotic for modulation of gut microbiota and immune microenvironment in inflammatory bowel disease. Chem Sci. (2023) 14:1598–605. doi: 10.1039/d2sc04984h
50.
de MillianoITabbersMMvan der PostJABenningaMA. Is a multispecies probiotic mixture effective in constipation during pregnancy? 'A pilot study'. Nutr J. (2012) 11:80. doi: 10.1186/1475-2891-11-80
51.
MezzasalmaVManfriniEFerriESandionigiALa FerlaBSchianoIet al. A randomized, double-blind, placebo-controlled trial: the efficacy of multispecies probiotic supplementation in alleviating symptoms of irritable bowel syndrome associated with constipation. Biomed Res Int. (2016) 2016:4740907–10. doi: 10.1155/2016/4740907
52.
IndrioFDi MauroARiezzoGCivardiEIntiniCCorvagliaLet al. Prophylactic use of a probiotic in the prevention of colic, regurgitation, and functional constipation: a randomized clinical trial. JAMA Pediatr. (2014) 168:228–33. doi: 10.1001/jamapediatrics.2013.4367
53.
BotelhoPBFerreiraMVRAraujoAMMendesMMNakanoEY. Effect of multispecies probiotic on gut microbiota composition in individuals with intestinal constipation: a double-blind, placebo-controlled randomized trial. Nutrition. (2020) 78:110890. doi: 10.1016/j.nut.2020.110890
54.
EorJYTanPLLimSMChoiDHYoonSMYangSYet al. Laxative effect of probiotic chocolate on loperamide-induced constipation in rats. Food Res Int. (2019) 116:1173–82. doi: 10.1016/j.foodres.2018.09.062
55.
AraujoAMBotelhoPBRibeiroDJSMagalhaesKGNakanoEYArrudaSF. A multiple-strain probiotic product provides a better enzymatic antioxidant response in individuals with constipation in a double-blind randomized controlled trial. Nutrition. (2021) 89:111225. doi: 10.1016/j.nut.2021.111225
56.
GanDChenJTangXXiaoLMartoniCJLeyerGet al. Impact of a probiotic chewable tablet on stool habits and microbial profile in children with functional constipation: a randomized controlled clinical trial. Front Microbiol. (2022) 13:985308. doi: 10.3389/fmicb.2022.985308
57.
HeYZhuLChenJTangXPanMYuanWet al. Efficacy of probiotic compounds in relieving constipation and their colonization in gut microbiota. Molecules. (2022) 27:27(3). doi: 10.3390/molecules27030666
58.
MaTYangNXieYLiYXiaoQLiQet al. Effect of the probiotic strain, Lactiplantibacillus plantarum P9, on chronic constipation: a randomized, double-blind, placebo-controlled study. Pharmacol Res. (2023) 191:106755. doi: 10.1016/j.phrs.2023.106755
59.
WeiYHuangNYeXLiuMWeiMHuangY. The postbiotic of hawthorn-probiotic ameliorating constipation caused by loperamide in elderly mice by regulating intestinal microecology. Front Nutr. (2023) 10:1103463. doi: 10.3389/fnut.2023.1103463
Summary
Keywords
probiotics, constipation, functional constipation, children, umbrella review
Citation
Dong M, Wu Y, Zhang M, Chen P, Zhang Z and Wang S (2023) Effect of probiotics intake on constipation in children: an umbrella review. Front. Nutr. 10:1218909. doi: 10.3389/fnut.2023.1218909
Received
08 May 2023
Accepted
22 August 2023
Published
01 September 2023
Volume
10 - 2023
Edited by
Arun K. B, Christ University, India
Reviewed by
Hui Jia, Shenyang Medical College, China; Amel Taibi, University of Toronto, Canada
Updates
Copyright
© 2023 Dong, Wu, Zhang, Chen, Zhang and Wang.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Shu Wang, 95154317@qq.com
†These authors have contributed equally to this work
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.