Abstract
The association between microbiota and physical activity is currently a key focus in sports performance research, and the effects of probiotics administration on athletes represent a relatively new area of research. While existing research highlights the promising potential of probiotics, our understanding of how they benefit highly active individuals remains incomplete. Nonetheless, it appears that probiotics have a beneficial effect on mental health, cognitive functions, sleep, gastrointestinal, and upper respiratory symptoms in adult humans. Additionally, the probiotic supplementation and their performance effects of different types of exercise are crucial when building a training program. In most cases, probiotic supplementation is effective in two major types of exercise: probiotics show strain and duration-specific effects both on endurance-based and intermittent-exercise associated sport. The supplementation can reduce inflammatory process activity and stress-related factors, e.g., anxiety, depression, in intermittent exercise-associated sports. In endurance-based sports, probiotics enhanced lipid metabolites, including short-chain and polyunsaturated fatty acids, modulated the maximal oxygen capacity, and reduced gastrointestinal symptoms. Exploring the relationship between probiotics, microbiome, and exercise performance could offer valuable insights for optimizing training techniques and strategies for professional athletes.
Created via Canva.com.
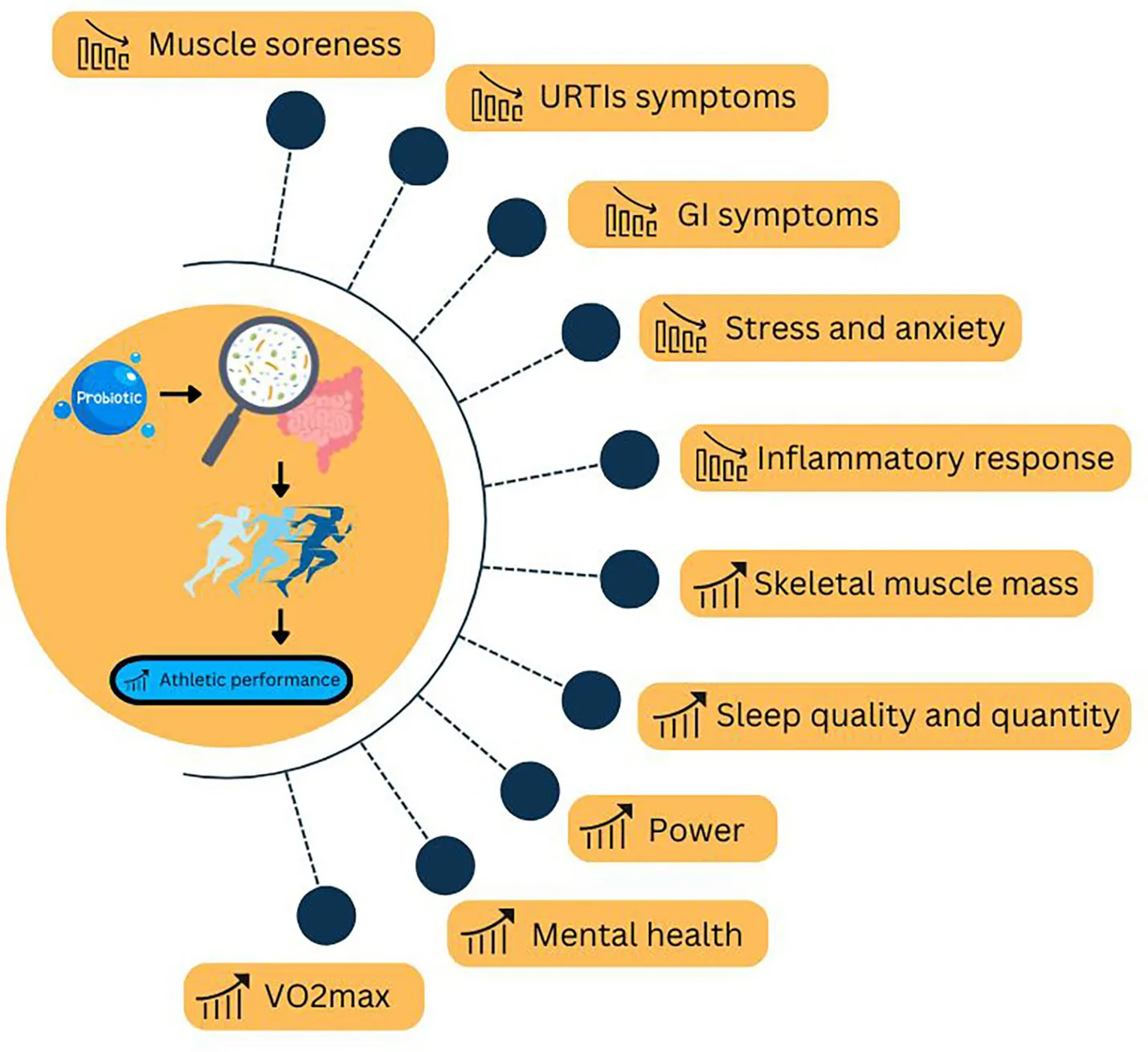
1 Introduction
Interest in and knowledge of the gut microbiome have increased drastically in the past 10 years (1, 2). The human gut microbiome represents a complex ecosystem that contributes essential functions to its host (3, 4). Recent large-scale experimental studies have provided evidence of its functional potential. The intricate communities of microorganisms in the human gastrointestinal tract are increasingly recognized as crucial contributors to human health and disease. Some human and in vitro studies have demonstrated that the gut microbiome can rapidly respond and change to the diet (5–8). The significance of the gut microbiome is underscored by several vital functions it performs for its human host. These functions include fermenting indigestible food components into absorbable metabolites, synthesizing essential vitamins, detoxifying harmful compounds, outcompeting pathogenic microbes, enhancing the intestinal barrier, and stimulating and regulating the immune system (9–14). In addition, the human gut microbiome plays an important role in biological processes associated with aging (15). Previously described that the microbiome has a crucial role in mental health, and there are correlations between the microbiome and anxiety and trauma-related disorders, and neurodegenerative disorders such as Alzheimer’s (AD) and Parkinson’s diseases (PD) (16–19). Our laboratory previously demonstrated that the microbiome has a critical role in AD, especially in the development of the disease, the mitochondrial function the metabolism-related molecular pathways in the liver (20–22). There is evidence that microbiomes might be a potential therapeutic target for liver diseases (23) and neurodevelopmental disorders, i.e., autism spectrum disorders (23–25).
Probiotics are live microorganisms that provide health benefits to the host when taken in sufficient quantities. Probiotic supplementation has been the research focus for years because it can modify the gut microbiota composition, increasing microbial diversity and supporting the growth and reproduction of health-promoting species (26, 27). While many probiotics can promote overall gastrointestinal, immune system, and brain health, the specific mechanisms through which they act—such as producing bioactive compounds, preventing pathogen adhesion, enhancing gut barrier function, modulating the immune response, and increasing brainwave components—can vary significantly between strains, even within the same bacterial species (28–32). The impact of probiotics on the gut microbiota composition is well-published (33–35). Despite growing evidence, the strain-specific effects of probiotics on athletic performance remain underexplored, necessitating a synthesis to guide personalized interventions.
This review (1) summarizes recent evidence on exercise-induced microbiome changes and (2) evaluates probiotic supplementation’s effects on athletic performance across exercise modalities. To identify relevant literature, studies were sourced from PubMed and Scopus (2015–2024). The search included various keywords and their combinations, such as “probiotics,” “microbiome,” “exercise performance,” and “wheelchair athletes,” focusing on human and animal trials. Articles were selected based on the relevance of their titles, abstracts, and full texts.
2 Associations between exercise and microbiome
The microbiota-physical activity association is currently the focus of converging interest in sports performance (36–40). Regular physical exercise provides many health benefits including cardiorespiratory fitness, and it can modify gut microbiota diversity (41, 42). O’Donovan et al. (42) studied 37 athletes from 16 different sports and categorized them based on peak static and dynamic components: low, moderate, and high static components; low, moderate, and high dynamic components. The authors found differences in microbiome and metabolome using shotgun sequencing. In the other study, the 12-week moderate-intensity aerobic exercise (running) showed no significant beneficial effect on the gut microbiota in 224 clinically well adolescents (43). In obese female humans, 6 weeks of cycle ergometer or treadmill training altered gut microbiota composition (44). In another clinical trial, exercise training improves gut microbiota profiles in sedentary people with prediabetes (45). In this study, subjects were randomized to two training modules: sprint and moderate-intensity continuous training for only 2 weeks, and they found training-specific gut microbiota changes. In contrast, short-term high-intensity interval training (HIIT) on a cycle ergometer did not impact the gut bacterial community in overweight men (46). Long-term, chronic exercise improved gut microflora in humans with type 2 diabetes (47). Similar results were observed in the other randomized clinical trial, where the 8-week high-intensity training (HIIT). The relative abundance of Bifidobacterium, A. municiphila, and butyrate-producing bacteria such as Lachnospira eligens, Enterococcus spp., and Clostridium Cluster IV was higher following lower-intensity exercise. In contrast, other butyrate producers (belonging to Eryspelothrichales and Oscillospirales) and the methane producer Methanobrevibacter smithii were more abundant after higher-intensity exercise (48). In young adolescents, 3 months of moderate-intensity exercise improved depressive symptoms. They increased the relative abundance of Coprococcus, Blautia, Dorea, Tyzzerella at the genus level, as well as Tyzzerella nexilis, Ruminococcus obeum at the species level (49). In contrast, 6 months of exercise induced subtle changes to the gut microbiota in humans with overweight and obesity (50). In elderly men, a 5-week endurance exercise program also affected the microbiome including a decreased relative abundance of Clostridium difficile, and increased Oscillospira. These results were correlated with the changes in several cardiometabolic risk factors, such as systolic and diastolic blood pressure (51). Changes in gut microbiome metabolites and redox homeostasis with exercise have been demonstrated in late middle-aged adults with familial and genetic risk for AD (52). In women, Bressa et al. (53) found differences in microbiota profiles between active and passive women. In active women, an increased abundance of health-promoting bacteria (Bifidobacterium spp., R. hominis, A. muciniphila, and F. prausnitzii) was present in the microbiota. In older women, 8 weeks of exercise increased the abundance of bacteria associated with an anti-inflammation pathway (54). Our laboratory previously demonstrated that gut microbiome alterations are associated with epigenetic age acceleration and physical fitness (55). We propose an overview of the exercise method or model and the physiological effects with the current evidence (Table 1).
Table 1
| Authors | Physical exercise | Physiological effect |
|---|---|---|
| O’Donovan et al. (42) | Classification of sports based on peak static and dynamic components | Different microbial diversity in different sports, increased estimated percent of maximal oxygen uptake, increased cardiac output, increased blood pressure load |
| Wang et al. (43) | 12-week running exercise at a moderate intensity of 50–70% HRmax for 30 min, 4 days/week | No significant effects |
| Allen et al. (44) | 6-week cycle ergometer or treadmill exercise at a moderate-to-vigorous intensity at 60–75% HRmax for 30 to 60 min, 3 days/week | Modulated composition and metabolic capacity of the gut microbiota |
| Motiani et al. (45) | Sprint and moderate-intensity continuous training for 2 weeks, 3 days/week | Improved VO2 peak after sprint, Decreased fatty acid uptake after moderate-intensity continuous training, Reduced systemic and intestinal inflammatory markers; modified microbiota profile in both training modules |
| Rettedal et al. (46) | Nine sessions of cycle ergometer HIIT on nonconsecutive days over 3 weeks | No significant effects |
| Pasini et al. (47) | 6-months of endurance, resistance, and flexibility training | Modified intestinal microbiota composition and gut barrier function |
| Wang et al. (49) | 3-months moderate intensity for 30 min, 4 days/week | Increased the relative abundance of microbiota genus and species levels, improved depressive symptoms |
| Torquati et al. (48) | 8 weeks of combined aerobic and resistance moderate-intensity continuous training or combined aerobic and resistance high-intensity interval training | Higher relative abundance of Bifidobacterium, and few butyrate producers at lower exercise intensity; Higher relative abundance of other butyrate producers and methane producers at higher exercise intensity |
| Kern et al. (50) | 6 months of bike or leisure-time exercise of either moderate (50% of VO2peak-reserve) or vigorous intensity (VIG, 70% of VO2peak-reserve), 5 days/week | No significant differences between alpha diversity and phenotypical outcomes, Beta diversity changed in all exercise groups |
| Taniguchi et al. (51) | 5-week aerobic exercise on an ergometer, 3 days/week | Changes in the relative abundance of microbiota and correlated with the changes in several cardiometabolic risk factors |
| Gaitán et al. (52) | 26-week treadmill exercise at moderate-to-vigorous intensity, less than 150 min per week | Increased levels of polyunsaturated free fatty acids (PUFAs) and changes in gut microbiome metabolites and redox homeostasis |
| Bressa et al. (53) | 7 days of accelerometer monitoring | Modulated microbiota profile in the active participants: increased abundance of health-promoting bacteria |
| Zhong et al. (54) | 8-week aerobic and resistance exercise for 60 min | Reduced abundance of bacteria associated with pro-inflammation |
Summary of associations between physical exercise and microbiome.
In animal studies, 12 weeks of resistance training enhanced the diversity of the gut microbiota in rats (56). In the same species, the wheel running exercise for 6 weeks was more effective in early life compared to adult animals (57). In mice, the moderate intensity of physical exercise affected the microbiome profile after 2, 6, 10, and 14 weeks of exercise (58). A similar duration of training (6 weeks) increased Bifidobacterium spp. level in exercised mice (59). Another type of exercise, namely swimming exercise, also modulated the relative abundance of the genus Desulfovibrio, genus Streptococcus, and genus p-75-a5 in depressed mice after the 5-week training program (60). The impact of physical activity has been demonstrated in neurodegenerative diseases, such as Parkinson’s and Alzheimer’s diseases; the relative abundance of the Bacteroidetes was decreased, while Firmicutes, Actinobacteria, Lactobacillaceae, Streptococcaceae, Lactobacillus, Streptococcus, Lactococcus, Lysinibacillus, Pelomonas, and Prevotellaceae_UCG-001 was increased in PD mice (61). In the APP/PS1 transgenic AD mice, 12 weeks of treadmill exercise effectively modulated the gut microbiome profile (62). Yu et al. demonstrated that exercise promoted the growth of butyrate-producing bacteria in the gut and enhanced butyrate production, which in turn enhanced lipid metabolism via the butyrate-SESN2/CRTC2 pathway (63).
3 Impact of probiotic supplementation on performance in healthy humans
A few important factors significantly determine sports performance, next to the training methods and strategies, such as sleep quality and quantity, mental health, stress, body composition, gastrointestinal symptoms, URTIs (upper respiratory tract infections) symptoms, and inflammatory responses. As we know, there is a relationship between cognitive function, sport-specific motor skills, and performance (64). Higher cognitive ability is associated with lower psychological distress (65). There is increasing evidence that the gut microbiome correlates with cognitive performance, and probiotic supplementation can improve cognitive functions and performance in healthy adults (33, 66). Probiotic administration mediates neuroprotective effects in healthy elderly (67). In addition, supplementation of Bifidobacterium longum BB68S improved cognitive functions such as immediate memory, visual–spatial/constructional memory, language, attention, and delayed memory in healthy older adults (68).
Stress, including anxiety, has been identified as a crucial factor in sport (65, 69). Previously described that sleep affects physical and mental performance, injury risk, recovery, and mental health (70, 71). 4-week probiotic supplementation reduced stress in healthy adults (72). In contrast, Morales-Torres et al. (73) did not find a significant effect after 4 weeks of Lactobacillus helveticus R0052 and Bifidobacterium longum R0175. The balance of circadian rhythm, i.e., sleep quality and quantity, is important in sports performance (74, 75). In healthy adults, probiotic supplementation is beneficial for cognitive function, mental health, and sleep (33, 66, 76).
Lactobacillus plantarum TWK10 significantly elevated exercise performance in a dose-dependent manner and improved the fatigue-associated features correlated with better physiological adaptation in healthy humans (77). Previously described probiotic supplementation showed both a systemic and local reduction of the inflammatory response (78, 79). These results might be important in the reduction of inflammatory responses expected after exercise or exercise-based injury.
URTIs are the most prevalent illnesses among athletes, leading to missed training sessions and competitions. The URTI symptoms, such as runny nose, nasal congestion, sneezing, and sore throat scores can decrease physiological performance (80, 81). Probiotic supplementation can suppress the symptoms of URTIs in adults (82, 83). Relatively short-term Streptococcus salivarius K12 supplementation can support the mucosal immune function of active young subjects (84). In contrast, there were no significant changes after probiotic administration in a meta-analysis that included more than 1,500 participants (85).
Endurance sports frequently cause exercise-induced gastrointestinal symptoms, i.e., abdominal bloating, heartburn, and diarrhea in endurance athletes, which may impact the physical and psychological performance (86). In healthy adults, daily supplementation of Bacillus subtilis BS50 alleviated gas-related gastrointestinal symptoms after 6 weeks (87). We propose an overview of probiotics and their physiological effects with the current evidence (Table 2).
Table 2
| Authors | Probiotics | Performance enhancement effect | Physiological effect | Population |
|---|---|---|---|---|
| Aljumaah et al. (66) | Lactobacillus rhamnosus GG | Cognitive funcions | Prevotella ruminicola, Bacteroides thetaiotaomicron, and Bacteroides xylanisolvens as taxa correlated with MCI (mild cognitive impairment) | healthy elderly |
| Kim et al. (33) | Bifidobacterium bifidum BGN4 and Bifidobacterium longum BORI | Mental flexibility and alleviating stress | Inflammation-causing gut bacteria were significantly reduced, and increased serum BDNF level | healthy elderly |
| Kim et al. (67) | Bifidobacterium bifidum BGN4 and Bifidobacterium longum BORI | Neuroprotective effects | Changes in the microbiota-related bile acid metabolism, which can reduce neuroinflammation in microglial cells | healthy elderly |
| Shi et al. (68) | Bifidobacterium longum BB68S | Cognitive functions | Significantly decreased the relative abundances of inflammation-related Solobacterium and Oribacterium, the relative abundance of Bifidobacterium increased markedly, changes of BRANS total score, including 5 domains such as immediate memory, visual–spatial, language, attention, and delayed memory | older adults |
| Boehme et al. (72) | Bifidobacterium longum NCC3001 | Stress relief | Improved sleep quality, reduction in anxiety, depression, and cortisol awakening response | adults |
| Morales-Torres et al. (73) | Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 | No significant effect | No significant effect | adults |
| Lee et al. (76) | Lactobacillus reuteri NK33 and Bifidobacterium adolescentis NK98 | Mental health and sleep | Reduced depressive symptoms, anxiety, and improved sleep quality; increased Bifidobacteriaceae and Lactobacillacea | adults |
| Huang et al. (77) | Lactobacillus plantarum TWK10 | Exercise performance | Decreased body fat significantly and increased muscle mass significantly | adults |
| Aida et al. (82) | Heyndrickxia coagulans strain SANK70258 | Upper respiratory tract infection | Induced anti-inflammatory effects (decreased IL-6 and TNFα level) via increased intestinal butyrate levels | adults |
| Altadill et al. (83) | Lactoplantibacillus plantarum DR7 | Upper respiratory tract infection | Reduced the proportion of patient days of URTI and of fever | adults |
| Bertuccioli et al. (84) | Streptococcus salivarius K12 | No effect for upper respiratory tract infection | Increased sIgA levels | adults |
| Garvey et al. (87) | Bacillus subtilis BS50 | Gastrointestinal symptoms | Improvement of burping and bloating | adults |
Summary of the physiological and performance enhancement effects of probiotic supplementation.
Since there is increasing evidence that exercise can modify gut microbiome profile, the following chapters focus on the other perspective, an overview of current knowledge from the past 5 years regarding exercise performance changes related to probiotics and different types of exercise in humans.
4 Endurance-based sports
The study investigated road cyclists who were supplemented with probiotics, and it demonstrated that there are no effects on the body composition of the athletes, except for an elevated muscle mass after a 4-month supplementation period. Furthermore, the long-term probiotics supplementation increased levels of the athlete’s aerobic capacity and positively affected the oxidative stress markers such as total oxidative status (TOS), TNF-α (tumor necrosis factor-alpha), and IL-6 (interleukin 6) related to the exercise capacity of competitive road cyclists (88). The elite road cyclist who received a multi-strain probiotic supplementation for 90 days reported a significantly lower incidence of GI symptoms compared to the placebo group, mean rate of perceived exertion values during the TTF (time-to-fatigue) was lower in the supplemented group, however, the authors did not find differences in VO2max and TTF values (89).
Eight weeks of Bifidobacterium lactis BL-99 administration showed an improvement in lipid metabolism markers such as DHA (docosahexaenoic acid), adrenic acid, linoleic acid, and acetic acid, and decreases in glycocholic acid and glycodeoxycholic acid; furthermore, the probiotics supplementation improved the VO2max and the knee-joint extensor strength in cross-country skiers (90).
URTIs in runners can present symptoms like a sore throat, coughing, congestion, a runny nose, mild fever, and fatigue. These infections, often caused by viruses like the common cold, can impair breathing, reduce oxygen uptake, and increase perceived exertion, making it harder for athletes to train or compete effectively. For athletes, especially endurance runners, URTIs are significant because even mild symptoms can lead to a decline in performance. A 6-week intervention with Lactobacillus helveticus Lafti L10 did not change time to exhaustion and the GI and cold/flu-like symptoms in exercise endurance among non-elite athletes (91). In contrast, the URTI symptoms were lower before the marathon, and the multi-strain probiotic administration affects cytokine production by monocytes after the competition in runners (92). In a longer supplementation study, 3 months of multi-strain probiotic administration increased serum HDL cholesterol and decreased LDL cholesterol and triglyceride levels, and the runner participants reported an improvement in general health (93). In endurance runners, a probiotic cocktail containing P. acidilactici and L. plantarum proved to be safe and did not affect gut or immune-associated parameters or intestinal symptoms after 4 weeks of supplementation (94). In long-distance male runners, an increase in lean body and skeletal muscle mass was demonstrated, while in the group of women taking the multi-strain probiotic for 1 month, a decrease in the content of total body fat and visceral fat was observed and the VO2max increased in both the women and men (95). The other study, which investigated long-distance runners, demonstrated that the Bifidobacterium longum subsp. longum Olympic No. 1 supplementation for 5 weeks significantly changed the 12-min Cooper’s test running distance and the abundance of gut microbiota (96). In marathon runners, pro-inflammatory cytokine production by stimulated lymphocytes decreased after 30 days of probiotic supplementation (97). We propose an overview of the probiotics, dosage, duration of supplementation, and their effects on performance in Table 3.
Table 3
| Authors | Sport | Probiotic supplementation | CFU | Duration | Effects for performance |
|---|---|---|---|---|---|
| Batatinha et al. (97) | Running | Bifidobacterium-animalis-subsp.-Lactis, Lactobacillus-Acidophilus | 10 × 109 | 30 days | ↓ pro-inflammatory cytokine production, maintained CD8 T cell and effector memory cell population |
| Lennon et al. (94) | Running | Pediococcus acidilactici, Lactobacillus plantarum | 3 × 109 | 4 weeks | No effects for GI symptoms |
| Lin et al. (96) | Running | Bifidobacterium longum subsp. longum OLP-01 | 1.5 × 1010 | 5 weeks | ↑ running distance ↑ gut microbiota abundance |
| Mazur-Kurach et al. (88) | Road cycle | Lactobacillus plantarum, Lactobacillus casei, Lactobacillus rhamnosus, Bifidobacterium breve, Lactobacillus acidophilus, Bifidobacterium longum, Bifidobacterium bifidum, Bifidobacterium infantis, Lactobacillus helveticus, Lactobacillus fermentum, Lactobacillus bulgaricus, Lactococcus lactis, Streptococcus thermophilus | 1 × 1011 | 4, 12 and 16 weeks | ↑ level of aerobic capacity ↑ magnitude of maximal oxygen uptake ↑ duration of exercise to failure ↓ heart rates |
| McDermott et al. (91) | Running | Lactobacillus helveticus Lafti L10 | 5 × 109 | 6 weeks | ↓ time-to-exhaustion |
| Schreiber et al. (89) | Cycle | Lactobacillus helveticus Lafti L10, Bifidobacterium animalis ssp. lactis Lafti B94, Enterococcus faecium R0026, Bifidobacterium longum R0175, Bacillus subtilis R0179 | 4.3 × 109 ≥4.3 × 109 ≥3.9 × 109 ≥2.1 × 109 ≥0.4 × 109 | 90 days | ↓ GI symptoms No significant effect in VO2max and time to fatigue |
| Smarkusz-Zarzecka et al. (95) | Running | Bifidobacterium lactis W52, Lactobacillus brevis W63, Lactobacillus casei W56, Lactococcus lactis W19, Lactococcus lactis W58, Lactobacillus acidophilus W37, Bifidobacterium bifidum W23 and Lactobacillus salivarius W24 | 2.5 × 109 | 12 weeks | ↑ VO2max ↑ minute ventilation ↑ functional capacity ↑ breathing reserve ↑ exercise capacity |
| Smarkusz-Zarzecka et al. (93) | Running | Bifidobacterium lactis W52, Levilactobacillus brevis W63, Lactobacillus casei W56, Lactococcus lactis W19, Lactobacillus lactis W58, Lactobacillus acidophilus W37, Bifidobacterium bifidum W23, Ligilactobacillus salivarius W24 | 2.5 × 109 | 12 weeks | ↓ GI symptoms |
| Li et al. (90) | Skiing | Bifidobacterium lactis BL-99 | 1 × 109 | 8 weeks | ↑ SCFAs level ↑ PUFAs level ↑ bile acids ↑ 180°/s knee joint extensor strength ↑ 60°/s knee joint extensor strength ↑ VO2max |
| Tavares-Silva et al. (92) | Running | Bifidobacterium-animalis-subsp.-Lactis, Lactobacillus-Acidophilus | 10 × 109 | 30 days | ↓ pro-inflammatory cytokine production ↑ numbers of naïve CD8 + T cells |
Summary of probiotic strains, dosing strategies, supplementation duration, and their reported effects on performance in endurance-based sports.
5 Intermittent exercise-associated sports
Salleh et al. (98) presented that 6 weeks of Lactobacillus casei supplementation improved aerobic capacity and reduced anxiety and stress in badminton players. Multi-strain probiotics in combination with dietary fiber for 23 days showed a reduction in inflammatory process activity and peripheral blood lymphocyte apoptosis in basketball athletes (99).
In professional soccer players, 1 month of synbiotic administration improved physical activity, sleep quality, and perceived general health, stress, and anxiety levels. Furthermore, the synbiotics induced an immunophysiological bioregulatory effect in the athletes (100). The Lactobacillus casei Shirota strain showed positive effects on anxiety-induced physiological parameters in football players (101). 6-week synbiotics supplementation, which is a combination of probiotics, significantly reduced the URTI symptoms, the incidence, and the duration of the symptoms in football players. In addition, the HRmax (maximal heart rate) and ER (lactic acid elimination rate) were markedly increased compared to the basal level during the recovery period after exercise in the symbiotic-supplemented group (102). In contrast, there were no significant changes in pain and fatigue in dancers after 12 weeks of Lactobacillus helveticus Rosell-52 and Bifidobacterium longum Rosell-17 supplementation (103). Furthermore, the probiotic mix capsule (containing eight different strains) did not change the TTE (time-to-exhaustion), RCP (respiratory compensation point), time-HHV (hypocapnic hyperventilation area time), and VO2max levels after 4 weeks in soccer players (104). We propose an overview of the probiotics, dosage, duration of supplementation, and their effects on performance in Table 4.
Table 4
| Authors | Sport | Probiotic supplementation | CFU | Duration | Effects for performance |
|---|---|---|---|---|---|
| Adikari et al. (101) | Football | Lactobacillus Casei Shirota strain | 3 × 1010 | 8 weeks | ↑ cognitive test reaction time (digit vigilance test) |
| Imanian et al. (104) | Soccer | Lactiplantibacillus plantarum BP06, Lacticaseibacillus casei BP07, Lactobacillus acidophilus BA05, Lactobacillus delbrueckii BD08 bulgaricus, Bifidobacterium infantis BI04, Bifidobacterium longum BL03, Bifidobacterium breve BB02 and Streptococcus salivarius thermophilus BT01 | 4.5 × 1011 | 4 weeks | No significant effect in TTE (time-to-exhaustion), RCP (respiratory compensation point), time-HHV (hypocapnic hyperventilation area time), and VO2max levels |
| Zhang et al. (102) | Football | Lactobacillus casei Zhang, Bifidobacterium lactis V9, Lactobacillus plantarum P-8 | ≥8 × 109 ≥8 × 109 ≥6 × 109 | 6 weeks | ↓ URTI symptoms ↑ SIgA level ↓ inflammatory factors ↓ HRmax ↓ ER (Lactic Acid Elimination Rate) |
| Quero et al. (100) | Soccer | Bifidobacterium lactis CBP-001010, Lactobacillus rhamnosus CNCM I-4036, Bifidobacterium longum ES1 | ≥1 × 109 | 1 month | ↓ stress level ↓ anxiety level ↓ depression level ↑ post-exercise dopamine concentration |
| Salleh et al. (98) | Badminton | Lactobacillus casei Shirota | 3 × 1010 | 6 weeks | ↓ stress levels and anxiety ↑ aerobic capacity |
| Trushina et al. (99) | Basketball | 10 of probiotic strains of Bifidobacteria and Lactobacilli | ≥1.25 × 1,010 | 23 days | ↓ inflammatory process activity ↓ peripheral blood lymphocyte apoptosis |
| Wiącek et al. (103) | Dance | Lactobacillus helveticus Rosell-52, Bifidobacterium longum Rosell-17 | 3 × 109 | 12 weeks | No significant changes in pain and fatigue |
Summary of probiotic strains, dosing strategies, supplementation duration, and their reported effects on performance in intermittent exercise-associated sports.
6 Resistance training-associated sports
Research on strength and power-based sports within this field is currently limited, resulting in a scarcity of published articles addressing this type of sport. In resistance-trained males, 30 and 60 days of Bacillus coagulans Unique IS-2 supplementation significantly increased the BCAA absorption and improved leg press and vertical jump power (105). In addition, Bacillus subtilis supplementation (5 billion CFU/day) improved the body composition of Division I female athletes in the 10-week resistance training program, where the athletes completed 3–4 workouts per week of upper- and lower-body exercises and sport-specific training (106). This study observed significant effects for improved squat, deadlift, and bench press 1 repetition maximum. The body composition analysis showed changes in body fat, muscle thickness, and rectus femoris after 10 weeks of supplementation. In rugby players, a shorter, 17-week daily probiotics (Lactobacillus acidophilus, Bifidobacterium bifidum, B. animalis ssp. Lactis) administration improved sleep quality and decreased muscle soreness and leg heaviness scores (107).
7 Wheelchair athletes
Gut disorders are a major contributor to morbidity among wheelchair athletes. The impact of probiotics on these athletes is not well-published. Nevertheless, there is only evidence of whether probiotics can improve the health and quality of life of wheelchair athletes. After a 12-week of probiotic administration, it has been confirmed that probiotics decreased inflammatory markers and improved the diversity of the gut microbiome (108). The probiotic contained Bifidobacterium lactis W51, Bifidobacterium lactis W52, Enterococcus faecium W54, Lactobacillus acidophilus W22, Lactobacillus paracasei W20, Lactobacillus plantarum W21, Lactobacillus salivarius W24, Lactococcus lactis W19. On the other hand, a total of 8 weeks of freeze-dried multispecies probiotic Bactosan pro FOS supplementation did not significantly reduce the GI symptoms (109).
8 Discussion
As we know, many factors affect the physiological performance of athletes, namely infections (i.e., URTIs), circadian rhythm, cognitive functions, stress, mental health, inflammatory responses, changes in skeletal muscle or body lean mass, gastrointestinal symptoms, etc. Technological advances have expanded knowledge of the gut microbiome from earlier beliefs. The topics covered in this review are new perspectives of what is currently understood regarding how the human gut microbiome changes after exercise and probiotics, and how probiotic administration impacts sports performance-related factors. The role of the endocrine and nervous routes in improving sleep efficiency was explained by previous studies that have reported the influence of probiotics on psycho-neuro-endocrine-immune activity via signals from the GI tract (110). Certain strains of bacteria produce substrates such as lactate and acetate for butyrate production, and it was associated with better cognitive function via modulation of the gut-brain axis (111, 112). Ameliorated lipid metabolism through SCFAs and PUFAs might have resulted from activated AMPK signaling pathway, which modulates fat synthesis. Another potential perspective is that a combination of exercise and probiotics has a positive effect on intestinal barrier function and systemic inflammatory response. These signaling pathways can amplify the gut-liver axis, which regulates metabolites in the intestinal area (113). Both lipid metabolism and the gut–liver axis are linked to athletic performance. Previous studies demonstrated that the gut–liver communication can significantly influence energy availability, inflammation, and recovery processes in human participants (114, 115). In addition, the probiotic supplementation might improve glucose metabolism, anti-inflammatory signaling activities through immunomodulation, and the balance of microbiome in the gut, thereby promoting an increase in VO2max (116). Optimizing probiotic intake, strains, timing (i.e., duration), and amount (colony-forming units, CFU) can help athletes improve their physiological performance. The impact of probiotics on athletes is a relatively new research field, with only a small number of studies conducted so far. While these studies have shown significant potential, there is still a limited understanding of the benefits of probiotics for highly active individuals and whether they gain from them.
9 Conclusion
Importantly, the effectiveness of probiotic supplementation on performance-related outcomes such as sleep quality and quantity, well-being, concentration, power, quickness, GI symptoms, heart rate, etc. is determined by multiple factors, including the specific probiotic strains and species, the colony-forming units (CFU), and the dosage protocol and strategy—encompassing both the duration and frequency of intake. These variables may influence the physiological impact of the supplementation either independently of, or in interaction with, exercise protocols, and might result in different effects. Furthermore, the positive effects depend on the type of sports. Probiotics show promise for enhancing both endurance and intermittent exercise performance, primarily through mechanisms such as the reduction of gastrointestinal symptoms and systemic inflammation. These effects may contribute to enhancing immune function, nutrient absorption, and overall performance and training capacity. However, current evidence supporting their efficacy in resistance-based sports remains limited and inconclusive. To establish more definitive conclusions, future research could focus on standardizing methodologies and assessing the physiological impacts of probiotics on elite athletes, including wheelchair athletes, focusing on consistent protocols and strain-specific interventions. Learning more about how probiotics and the microbiome change in exercise performance may provide new insights for new training techniques and strategies for professional athletes.
Statements
Author contributions
TT: Conceptualization, Writing – original draft. ZR: Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. Open-access funding is provided by the Hungarian University of Sports Science. This research was supported by the New National Excellence Program, and the Scientific Excellence Program, TKP2021-EGA-37, at the Hungarian University Sport Science, Innovation and Technology Ministry, Hungary, and by a grant from the National Science and Research Foundation, Hungary (OTKA 142192) to ZR. This research was funded by the NVKP_16-1–2016-0004 grant of the Hungarian National Research, Development and Innovation Office (NKFIH), VEKOP-2.3.2–162016-00002, VEKOP-2.3.3–15-2017–00016, the Therapeutic Thematic Programme TKP2021-EGA-23. This study was also supported by the grants RRF-2.3.121–2022-00003 (National Cardiovascular Laboratory Program) and 2019–2.1.7-ERA-NET-2021–00015, and the EU’s Horizon 2020 Research and Innovation Programme grant No. 739593. The project was implemented with the support from the National Research, Development and Innovation Fund of the Ministry of Culture and Innovation under the National Laboratories Program [National Tumor Biology Laboratory (2022–2.1.1-NL-2022–00010)] and the Hungarian Thematic Excellence Program (under project TKP2021-EGA-44) Grant Agreements with the National Research, Development and Innovation Office. This project has received funding from the HUN-REN Hungarian Research Network (grant No. 1500207).
Acknowledgments
The graphical abstract was drawn by Canva.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Gen AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
AD, Alzheimer’s disease; BCAA, Branched-chain amino acids; DHA, Docosahexaenoic acid; GI, Gastrointestinal; HDL, High-density lipoproteins; HIIT, High-intensity interval training; IL-6, Interleukin 6; LDL, Low-density lipoproteins; PD, Parkinson’s disease; RCP, Respiratory compensation point; time-HHV, Hypocapnic hyperventilation area time; TNF-a, Tumor necrosis factor-alpha; TTE, Time-to-exhaustion; TTF, Time-to-fatigue; URTIs, Upper respiratory tract infections.
References
1.
KraimiNRossTPujoJDe PalmaG. The gut microbiome in disorders of gut-brain interaction. Gut Microbes. (2024) 16:233. doi: 10.1080/19490976.2024.2360233
2.
KuzielGARakoff-NahoumS. The gut microbiome. Curr Biol. (2022) 32:R257–64. doi: 10.1016/j.cub.2022.02.023
3.
CresciGABawdenE. Gut microbiome: what we do and Don’t know. Nutr Clin Pract. (2015) 30:734–46. doi: 10.1177/0884533615609899
4.
Heintz-BuschartAWilmesP. Human gut microbiome: function matters. Trends Microbiol. (2018) 26:563–74. doi: 10.1016/j.tim.2017.11.002
5.
AguirreMEckAKoenenMESavelkoulPHMBuddingAEVenemaK. Diet drives quick changes in the metabolic activity and composition of human gut microbiota in a validated in vitro gut model. Res Microbiol. (2016) 167:114–25. doi: 10.1016/j.resmic.2015.09.006
6.
KlimenkoNSTyakhtAVPopenkoASVasilievASAltukhovIAIschenkoDSet al. Microbiome responses to an uncontrolled short-term diet intervention in the frame of the citizen science project. Nutrients. (2018) 10:576. doi: 10.3390/nu10050576
7.
QuerciaSTurroniSFioriJSoveriniMRampelliSBiagiEet al. Gut microbiome response to short-term dietary interventions in reactive hypoglycemia subjects. Diabetes Metab Res Rev. (2017) 33:2927. doi: 10.1002/DMRR.2927
8.
SangXLiSGuoRYanQLiuCZhangYet al. Dynamics and ecological reassembly of the human gut microbiome and the host metabolome in response to prolonged fasting. Front Microbiol. (2023) 14:1265425. doi: 10.3389/fmicb.2023.1265425
9.
ChenFDaiXZhouCCLiKXZhangYJLouXYet al. Integrated analysis of the faecal metagenome and serum metabolome reveals the role of gut microbiome-associated metabolites in the detection of colorectal cancer and adenoma. Gut. (2022) 71:1315–25. doi: 10.1136/gutjnl-2020-323476
10.
KarlJPMargolisLMMadslienEHMurphyNECastellaniJWGundersenYet al. Changes in intestinal microbiota composition and metabolism coincide with increased intestinal permeability in young adults under prolonged physiological stress. Am J Physiol Gastrointest Liver Physiol. (2017) 312:G559–71. doi: 10.1152/ajpgi.00066.2017
11.
PhamVTFehlbaumSSeifertNRichardNBruinsMJSybesmaWet al. Effects of colon-targeted vitamins on the composition and metabolic activity of the human gut microbiome- a pilot study. Gut Microbes. (2021) 13:1–20. doi: 10.1080/19490976.2021.1875774
12.
ShiNLiNDuanXNiuH. Interaction between the gut microbiome and mucosal immune system. Mil Med Res. (2017) 4:14. doi: 10.1186/s40779-017-0122-9
13.
UebansoTShimohataTMawatariKTakahashiA. Functional roles of B-vitamins in the gut and gut microbiome. Mol Nutr Food Res. (2020) 64:426. doi: 10.1002/MNFR.202000426
14.
WuJWangKWangXPangYJiangC. The role of the gut microbiome and its metabolites in metabolic diseases. Protein Cell. (2021) 12:360–73. doi: 10.1007/s13238-020-00814-7
15.
BradleyEHaranJ. The human gut microbiome and aging. Gut Microbes. (2024) 16:9677. doi: 10.1080/19490976.2024.2359677
16.
DuYLiYXuXLiRZhangMCuiYet al. Probiotics for constipation and gut microbiota in Parkinson’s disease. Parkinsonism Relat Disord. (2022) 103:92–7. doi: 10.1016/j.parkreldis.2022.08.022
17.
Malan-MullerSValles-ColomerMRaesJLowryCASeedatSHemmingsSMJ. The gut microbiome and mental health: implications for anxiety- and trauma-related disorders. OMICS. (2018) 22:90–107. doi: 10.1089/omi.2017.0077
18.
SassoJMAmmarRMTenchovRLemmelSKelberOGrieswelleMet al. Gut microbiome-brain Alliance: a landscape view into mental and gastrointestinal health and disorders. ACS Chem Neurosci. (2023) 14:1717–63. doi: 10.1021/acschemneuro.3c00127
19.
ZhangSLuJJinZXuHZhangDChenJet al. Gut microbiota metabolites: potential therapeutic targets for Alzheimer’s disease?Front Pharmacol. (2024) 15:1459655. doi: 10.3389/FPHAR.2024.1459655
20.
AbrahamDFeherJScuderiGLSzaboDDobolyiACservenakMet al. Exercise and probiotics attenuate the development of Alzheimer’s disease in transgenic mice: role of microbiome. Exp Gerontol. (2019) 115:122–31. doi: 10.1016/j.exger.2018.12.005
21.
KolonicsABoriZTormaFAbrahamDFehérJRadakZ. Exercise combined with postbiotics treatment results in synergistic improvement of mitochondrial function in the brain of male transgenic mice for Alzheimer’s disease. BMC Neurosci. (2023) 24:68. doi: 10.1186/S12868-023-00836-X
22.
TéglásTÁbrahámDJókaiMKondoSMohammadiRFehérJet al. Exercise combined with a probiotics treatment alters the microbiome, but moderately affects signalling pathways in the liver of male APP/PS1 transgenic mice. Biogerontology. (2020) 21:807–15. doi: 10.1007/s10522-020-09895-7
23.
SinghTPKadyanSDeviHParkGNagpalR. Gut microbiome as a therapeutic target for liver diseases. Life Sci. (2023) 322:121685. doi: 10.1016/j.lfs.2023.121685
24.
ChernikovaMAFloresGDKilroyELabusJSMayerEAAziz-ZadehL. The brain-gut-microbiome system: pathways and implications for autism Spectrum disorder. Nutrients. (2021) 13:4497. doi: 10.3390/nu13124497
25.
TaniyaMAChungHJAl MamunAAlamSAzizMAEmonNUet al. Role of gut microbiome in autism Spectrum disorder and its therapeutic regulation. Front Cell Infect Microbiol. (2022) 12:915701. doi: 10.3389/fcimb.2022.915701
26.
KurdiMBajwaSJSSharmaRChoudharyR. Gut microbiota and probiotics in perioperative management: a narrative review. Cureus. (2024) 16:e68404. doi: 10.7759/cureus.68404
27.
Van HulMCaniPDPetitfilsCDe VosWMTilgHEl-OmarEM. What defines a healthy gut microbiome?Gut. (2024) 73:1893–908. doi: 10.1136/gutjnl-2024-333378
28.
AhmadARJariahMNZulkifliNAMGhafarH. A review of nutritional ergogenic aids on physiological responses and skilled performance among football players. Ann Appl Sport Sci. (2024) 13:1458. doi: 10.52547/aassjournal.1458
29.
CaoFJinLGaoYDingYWenHQianZet al. Artificial-enzymes-armed Bifidobacterium longum probiotics for alleviating intestinal inflammation and microbiota dysbiosis. Nat Nanotechnol. (2023) 18:617–27. doi: 10.1038/s41565-023-01346-x
30.
ReikvamDHMeyer-MyklestadMHTrøseidMStiksrudB. Probiotics to manage inflammation in HIV infection. Curr Opin Infect Dis. (2020) 33:34–43. doi: 10.1097/QCO.0000000000000612
31.
SahaBNSarkarMAMHasanMNKamrul-HasanABDasSCAlamMRet al. Efficacy of probiotics as adjuvant to the standard triple therapy for the treatment of Helicobacter pylori-associated peptic ulcer disease: a randomized-control trial. Mymensingh Med J. (2024) 33:1166–71. PMID:
32.
ZhouJLiMChenQLiXChenLDongZet al. Programmable probiotics modulate inflammation and gut microbiota for inflammatory bowel disease treatment after effective oral delivery. Nat Commun. (2022) 13:3432. doi: 10.1038/S41467-022-31171-0
33.
KimCSChaLSimMJungSChunWYBaikHWet al. Probiotic supplementation improves cognitive function and mood with changes in gut microbiota in community-dwelling older adults: a randomized, double-blind, placebo-controlled, multicenter trial. J Gerontol A Biol Sci Med Sci. (2021) 76:32–40. doi: 10.1093/GERONA/GLAA090
34.
LaiHLiYHeYChenFMiBLiJet al. Effects of dietary fibers or probiotics on functional constipation symptoms and roles of gut microbiota: a double-blinded randomized placebo trial. Gut Microbes. (2023) 15:7837. doi: 10.1080/19490976.2023.2197837
35.
WangYLiNYangJJZhaoDMChenBZhangGQet al. Probiotics and fructo-oligosaccharide intervention modulate the microbiota-gut brain axis to improve autism spectrum reducing also the hyper-serotonergic state and the dopamine metabolism disorder. Pharmacol Res. (2020) 157:104784. doi: 10.1016/j.phrs.2020.104784
36.
AyaVFlórezAPerezLRamírezJD. Association between physical activity and changes in intestinal microbiota composition: a systematic review. PLoS One. (2021) 16:e0247039. doi: 10.1371/journal.pone.0247039
37.
CerdáBPérezMPérez-SantiagoJDTornero-AguileraJFGonzález-SolteroRLarrosaM. Gut microbiota modification: another piece in the puzzle of the benefits of physical exercise in health?Front Physiol. (2016) 7:51. doi: 10.3389/fphys.2016.00051
38.
ClarkAMachN. The crosstalk between the gut microbiota and mitochondria during exercise. Front Physiol. (2017) 8:319. doi: 10.3389/FPHYS.2017.00319
39.
MachNFuster-BotellaD. Endurance exercise and gut microbiota: a review. J Sport Health Sci. (2017) 6:179–97. doi: 10.1016/j.jshs.2016.05.001
40.
MohrAEJägerRCarpenterKCKerksickCMPurpuraMTownsendJRet al. The athletic gut microbiota. J Int Soc Sports Nutr. (2020) 17:24. doi: 10.1186/S12970-020-00353-W
41.
EstakiMPitherJBaumeisterPLittleJPGillSKGhoshSet al. Cardiorespiratory fitness as a predictor of intestinal microbial diversity and distinct metagenomic functions. Microbiome. (2016) 4:42. doi: 10.1186/S40168-016-0189-7
42.
O’DonovanCMMadiganSMGarcia-PerezIRankinAO’ SullivanOCotterPD. Distinct microbiome composition and metabolome exists across subgroups of elite Irish athletes. J Sci Med Sport. (2020) 23:63–8. doi: 10.1016/j.jsams.2019.08.290
43.
WangRCaiYLiJYauSLuWStubbsBet al. Effects of aerobic exercise on gut microbiota in adolescents with subthreshold mood syndromes and healthy adolescents: a 12-week, randomized controlled trial. J Affect Disord. (2021) 293:363–72. doi: 10.1016/j.jad.2021.06.025
44.
AllenJMMailingLJNiemiroGMMooreRCookMDWhiteBAet al. Exercise alters gut microbiota composition and function in lean and obese humans. Med Sci Sports Exerc. (2018) 50:747–57. doi: 10.1249/MSS.0000000000001495
45.
MotianiKKColladoMCEskelinenJJVirtanenKALöyttyniemiESalminenSet al. Exercise training modulates gut microbiota profile and improves Endotoxemia. Med Sci Sports Exerc. (2020) 52:94–104. doi: 10.1249/MSS.0000000000002112
46.
RettedalEACreeJMEAdamsSEMacRaeCSkidmorePMLCameron-SmithDet al. Short-term high-intensity interval training exercise does not affect gut bacterial community diversity or composition of lean and overweight men. Exp Physiol. (2020) 105:1268–79. doi: 10.1113/EP088744
47.
PasiniECorsettiGAssanelliDTestaCRomanoCDioguardiFSet al. Effects of chronic exercise on gut microbiota and intestinal barrier in human with type 2 diabetes. Minerva Med. (2019) 110:3–11. doi: 10.23736/S0026-4806.18.05589-1
48.
TorquatiLGajanandTCoxERWillisCRGZauggJKeatingSEet al. Effects of exercise intensity on gut microbiome composition and function in people with type 2 diabetes. Eur J Sport Sci. (2023) 23:530–41. doi: 10.1080/17461391.2022.2035436
49.
WangRCaiYLuWZhangRShaoRYauSYet al. Exercise effect on the gut microbiota in young adolescents with subthreshold depression: a randomized psychoeducation-controlled trial. Psychiatry Res. (2023) 319:115005. doi: 10.1016/j.psychres.2022.115005
50.
KernTBlondMBHansenTHRosenkildeMQuistJSGramASet al. Structured exercise alters the gut microbiota in humans with overweight and obesity-a randomized controlled trial. Int J Obes. (2020) 44:125–35. doi: 10.1038/s41366-019-0440-y
51.
TaniguchiHTanisawaKSunXKuboTHoshinoYHosokawaMet al. Effects of short-term endurance exercise on gut microbiota in elderly men. Phys Rep. (2018) 6:e13935. doi: 10.14814/phy2.13935
52.
GaitánJMMoonHYStremlauMDubalDBCookDBOkonkwoOCet al. Effects of aerobic exercise training on systemic biomarkers and cognition in late middle-aged adults at risk for Alzheimer’s disease. Front Endocrinol (Lausanne). (2021) 12:660181. doi: 10.3389/FENDO.2021.660181
53.
BressaCBailén-AndrinoMPérez-SantiagoJGonzález-SolteroRPérezMMontalvo-LomincharMGet al. Differences in gut microbiota profile between women with active lifestyle and sedentary women. PLoS One. (2017) 12:e0171352. doi: 10.1371/journal.pone.0171352
54.
ZhongFWenXYangMLaiHYMommaHChengLet al. Effect of an 8-week exercise training on gut microbiota in physically inactive older women. Int J Sports Med. (2021) 42:610–23. doi: 10.1055/a-1301-7011
55.
TormaFKerepesiCJókaiMBabszkiGKoltaiELigetiBet al. Alterations of the gut microbiome are associated with epigenetic age acceleration and physical fitness. Aging Cell. (2024) 23:e14101. doi: 10.1111/ACEL.14101
56.
CastroAPSilvaKKSMedeirosCSAAlvesFAraujoRCAlmeidaJA. Effects of 12 weeks of resistance training on rat gut microbiota composition. J Exp Biol. (2021) 224:8954. doi: 10.1242/jeb.242543
57.
MikaAVan TreurenWGonzálezAHerreraJJKnightRFleshnerM. Exercise is more effective at altering gut microbial composition and producing stable changes in lean mass in juvenile versus adult male F344 rats. PLoS One. (2015) 10:e0125889. doi: 10.1371/journal.pone.0125889
58.
YangWLiuYYangGMengBYiZYangGet al. Moderate-intensity physical exercise affects the exercise performance and gut microbiota of mice. Front Cell Infect Microbiol. (2021) 11:712381. doi: 10.3389/FCIMB.2021.712381
59.
LambertJEMyslickiJPBomhofMRBelkeDDShearerJReimerRA. Exercise training modifies gut microbiota in normal and diabetic mice. Appl Physiol Nutr Metab. (2015) 40:749–52. doi: 10.1139/apnm-2014-0452
60.
XieYWuZZhouLSunLXiaoLWangG. Swimming exercise modulates gut microbiota in CUMS-induced depressed mice. Neuropsychiatr Dis Treat. (2022) 18:749–60. doi: 10.2147/NDT.S355723
61.
FanTLiXZhangXZhangJSunLChenJet al. Influence of aerobic exercise training on mice gut microbiota in Parkinson’s disease. Turk J Biol. (2022) 46:288–97. doi: 10.55730/1300-0152.2617
62.
YuanSYangJJianYLeiYYaoSHuZet al. Treadmill exercise modulates intestinal microbes and suppresses LPS displacement to alleviate Neuroinflammation in the brains of APP/PS1 mice. Nutrients. (2022) 14:4134. doi: 10.3390/nu14194134
63.
YuCLiuSChenLShenJNiuYWangTet al. Effect of exercise and butyrate supplementation on microbiota composition and lipid metabolism. J Endocrinol. (2019) 243:125–35. doi: 10.1530/JOE-19-0122
64.
ScharfenHEMemmertD. The relationship between cognitive functions and sport-specific motor skills in elite youth soccer players. Front Psychol. (2019) 10:817. doi: 10.3389/FPSYG.2019.00817
65.
JokelaM. Why is cognitive ability associated with psychological distress and wellbeing? Exploring psychological, biological, and social mechanisms. Personal Individ Differ. (2022) 192:111592. doi: 10.1016/j.paid.2022.111592
66.
AljumaahMRBhatiaURoachJGunstadJAzcarate PerilMA. The gut microbiome, mild cognitive impairment, and probiotics: a randomized clinical trial in middle-aged and older adults. Clin Nutr. (2022) 41:2565–76. doi: 10.1016/j.clnu.2022.09.012
67.
KimCSJungSHwangGSShinDM. Gut microbiota indole-3-propionic acid mediates neuroprotective effect of probiotic consumption in healthy elderly: a randomized, double-blind, placebo-controlled, multicenter trial and in vitro study. Clin Nutr. (2023) 42:1025–33. doi: 10.1016/j.clnu.2023.04.001
68.
ShiSZhangQSangYGeSWangQWangRet al. Probiotic Bifidobacterium longum BB68S improves cognitive functions in healthy older adults: a randomized, double-blind, placebo-controlled trial. Nutrients. (2022) 15:51. doi: 10.3390/nu15010051
69.
TossiciGZurloniVNitriA. Stress and sport performance: a PNEI multidisciplinary approach. Front Psychol. (2024) 15:1358771. doi: 10.3389/fpsyg.2024.1358771
70.
CharestJGrandnerMA. Sleep and athletic performance: impacts on physical performance, mental performance, injury risk and recovery, and mental health. Sleep Med Clin. (2020) 15:41–57. doi: 10.1016/j.jsmc.2019.11.005
71.
MassarSAALimJHuettelSA. Sleep deprivation, effort allocation and performance. Prog Brain Res. (2019) 246:1–26. doi: 10.1016/BS.PBR.2019.03.007
72.
BoehmeMRémond-DerbezNLerondCLavalleLKeddaniSSteinmannMet al. Bifidobacterium longum subsp. longum reduces perceived psychological stress in healthy adults: an exploratory clinical trial. Nutrients. (2023) 15:3122. doi: 10.3390/nu15143122
73.
Morales-TorresRCarrasco-GubernatisCGrasso-CladeraACosmelliDParadaFJPalacios-GarcíaI. Psychobiotic effects on anxiety are modulated by lifestyle behaviors: a randomized placebo-controlled trial on healthy adults. Nutrients. (2023) 15:1706. doi: 10.3390/nu15071706
74.
FullagarHHKVincentGEMcCulloughMHalsonSFowlerP. Sleep and sport performance. J Clin Neurophysiol. (2023) 40:408–16. doi: 10.1097/WNP.0000000000000638
75.
KhanMAAl-JahdaliH. The consequences of sleep deprivation on cognitive performance. Neurosciences (Riyadh). (2023) 28:91–9. doi: 10.17712/nsj.2023.2.20220108
76.
LeeHJHongJKKimJKKimDHJangSWHanSWet al. Effects of probiotic NVP-1704 on mental health and sleep in healthy adults: an 8-week randomized, double-blind, placebo-controlled trial. Nutrients. (2021) 13:2660. doi: 10.3390/nu13082660
77.
HuangWCLeeMCLeeCCNgKSHsuYJTsaiTYet al. Effect of Lactobacillus plantarum TWK10 on exercise physiological adaptation, performance, and body composition in healthy humans. Nutrients. (2019) 11:2836. doi: 10.3390/nu11112836
78.
LiXHuSYinJPengXKingLLiLet al. Effect of synbiotic supplementation on immune parameters and gut microbiota in healthy adults: a double-blind randomized controlled trial. Gut Microbes. (2023) 15:7025. doi: 10.1080/19490976.2023.2247025
79.
SteneCRömeAPalmquistILinningeCMolinGAhrnéSet al. Administration of probiotics to healthy volunteers: effects on reactivity of intestinal mucosa and systemic leukocytes. BMC Gastroenterol. (2022) 22:100. doi: 10.1186/S12876-022-02185-1
80.
CicchellaAStefanelliCMassaroM. Upper respiratory tract infections in sport and the immune system response. A review. Biology (Basel). (2021) 10:362. doi: 10.3390/BIOLOGY10050362
81.
KaulbackKPyneDBHullJHSnydersCSewryNSchwellnusM. The effects of acute respiratory illness on exercise and sports performance outcomes in athletes – a systematic review by a subgroup of the IOC consensus group on “acute respiratory illness in the athlete.”. Eur J Sport Sci. (2023) 23:1356–74. doi: 10.1080/17461391.2022.2089914
82.
AidaMTogawaNMizuyamaKAokiYSuehiroSSakamotoAet al. Heyndrickxia coagulans strain SANK70258 suppresses symptoms of upper respiratory tract infection via immune modulation: a randomized, double-blind, placebo-controlled, parallel-group, comparative study. Front Immunol. (2024) 15:1389920. doi: 10.3389/FIMMU.2024.1389920
83.
AltadillTEspadaler-MazoJLiongMT. Effects of a lactobacilli probiotic on reducing duration of URTI and fever, and use of URTI-associated medicine: a re-analysis of a randomized, placebo-controlled study. Microorganisms. (2021) 9:1–10. doi: 10.3390/MICROORGANISMS9030528
84.
BertuccioliAGervasiMAnnibaliniGBinatoBPerroniFRocchiMBLet al. Use of Streptococcus salivarius K12 in supporting the mucosal immune function of active young subjects: a randomised double-blind study. Front Immunol. (2023) 14:1129060. doi: 10.3389/FIMMU.2023.1129060
85.
LiLHongKKSunQXiaoHLaiLMingMet al. Probiotics for preventing upper respiratory tract infections in adults: a systematic review and Meta-analysis of randomized controlled trials. Evid Based Complement Alternat Med. (2020) 2020:4140. doi: 10.1155/2020/8734140
86.
RibichiniEScaleseGCesariniAMocciCPallottaNSeveriCet al. Exercise-induced gastrointestinal symptoms in endurance sports: a review of pathophysiology, symptoms, and nutritional management. Dietetics. (2023) 2:289–307. doi: 10.3390/dietetics2030021
87.
GarveySMMahEBlonquistTMKadenVNSpearsJL. The probiotic Bacillus subtilis BS50 decreases gastrointestinal symptoms in healthy adults: a randomized, double-blind, placebo-controlled trial. Gut Microbes. (2022) 14:2668. doi: 10.1080/19490976.2022.2122668
88.
Mazur-KurachPFrączekBKlimekAT. Does multi-strain probiotic supplementation impact the effort capacity of competitive road cyclists?Int J Environ Res Public Health. (2022) 19:2205. doi: 10.3390/IJERPH191912205
89.
SchreiberCTamirSGolanRWeinsteinAWeinsteinY. The effect of probiotic supplementation on performance, inflammatory markers and gastro-intestinal symptoms in elite road cyclists. J Int Soc Sports Nutr. (2021) 18:36. doi: 10.1186/S12970-021-00432-6
90.
LiTRuiZMaoLChangYShaoJChenYet al. Eight weeks of Bifidobacterium lactis BL-99 supplementation improves lipid metabolism and sports performance through short-chain fatty acids in cross-country skiers: a preliminary study. Nutrients. (2023) 15:4554. doi: 10.3390/NU15214554
91.
McDermottCEJudkinsTCVincentHKCulpepperTColeeJNievesCet al. Impact of probiotic supplementation on exercise endurance among nonelite athletes: a randomized, placebo-controlled, double-blind, clinical trial. Appl Physiol Nutr Metab. (2024) 49:–142. doi: 10.1139/apnm-2024-0142
92.
Tavares-SilvaECarisAVSantosSARavacciGRThomatieli-SantosRV. Effect of multi-strain probiotic supplementation on URTI symptoms and cytokine production by monocytes after a marathon race: a randomized, double-blind, placebo study. Nutrients. (2021) 13:1478. doi: 10.3390/NU13051478
93.
Smarkusz-ZarzeckaJOstrowskaLLeszczyńskaJCwalinaU. Effect of a multi-strain probiotic supplement on gastrointestinal symptoms and serum biochemical parameters of long-distance runners: a randomized controlled trial. Int J Environ Res Public Health. (2022) 19:9363. doi: 10.3390/ijerph19159363
94.
LennonSLackieTMiltkoAKearnsZCPaquetteMRBloomerRJet al. Safety and efficacy of a probiotic cocktail containing P. acidilactici and L. plantarum for gastrointestinal discomfort in endurance runners: randomized double-blinded crossover clinical trial. Appl Physiol Nutr Metab. (2024) 49:890–903. doi: 10.1139/apnm-2023-0449
95.
Smarkusz-ZarzeckaJOstrowskaLLeszczyńskaJOrywalKCwalinaUPogodzińskiD. Analysis of the impact of a multi-strain probiotic on body composition and cardiorespiratory fitness in long-distance runners. Nutrients. (2020) 12:1–13. doi: 10.3390/NU12123758
96.
LinCLHsuYJHoHHChangYCKuoYWYehYTet al. Bifidobacterium longum subsp. longum OLP-01 supplementation during endurance running training improves exercise performance in middle- and long-distance runners: a double-blind controlled trial. Nutrients. (2020) 12:1–14. doi: 10.3390/NU12071972
97.
BatatinhaHTavares-SilvaELeiteGSFResendeASAlbuquerqueJATArslanianCet al. Probiotic supplementation in marathonists and its impact on lymphocyte population and function after a marathon: a randomized placebo-controlled double-blind study. Sci Rep. (2020) 10:18777. doi: 10.1038/S41598-020-75464-0
98.
SallehRMKuanGAzizMNARahimMRARahayuTSulaimanSet al. Effects of probiotics on anxiety, stress, mood and fitness of badminton players. Nutrients. (2021) 13:1783. doi: 10.3390/nu13061783
99.
TrushinaENRigerNAMustafinaOKTimoninANSolntsevaTNZilovaISet al. Multi-strain probiotic combined with dietary fiber is an effective factor in the nutritional support of immunity in athletes. Vopr Pitan. (2024) 93:19–30. doi: 10.33029/0042-8833-2024-93-2-19-30
100.
QueroCDManonellesPFernándezMAbellán-AynésOLópez-PlazaDAndreu-CaravacaLet al. Differential health effects on inflammatory, immunological and stress parameters in professional soccer players and sedentary individuals after consuming a Synbiotic. A triple-blinded, randomized, placebo-controlled pilot study. Nutrients. (2021) 13:1321. doi: 10.3390/nu13041321
101.
AdikariAMGCPAppukuttyMKuanG. Effects of daily probiotics supplementation on anxiety induced physiological parameters among competitive football players. Nutrients. (2020) 12:1–17. doi: 10.3390/NU12071920
102.
ZhangLXiaoHZhaoLLiuZChenLLiuC. Comparison of the effects of prebiotics and Synbiotics supplementation on the immune function of male university football players. Nutrients. (2023) 15:1158. doi: 10.3390/nu15051158
103.
WiącekJPodgórskiTKusyKŁoniewskiISkonieczna-ŻydeckaKKarolkiewiczJ. Evaluating the impact of probiotic therapy on the endocannabinoid system, pain, sleep and fatigue: a randomized, double-blind, placebo-controlled trial in dancers. Int J Mol Sci. (2024) 25:5611. doi: 10.3390/ijms25115611
104.
ImanianBHemmatinafarMDaryanooshFKoureshfardNSadeghiRNiknamAet al. The effect of probiotics and casein supplementation on aerobic capacity parameters of male soccer players. J Int Soc Sports Nutr. (2024) 21:2165. doi: 10.1080/15502783.2024.2382165
105.
TarikMRamakrishnanLBhatiaNGoswamiRKandasamyDRoyAet al. The effect of Bacillus coagulans unique IS-2 supplementation on plasma amino acid levels and muscle strength in resistance trained males consuming whey protein: a double-blind, placebo-controlled study. Eur J Nutr. (2022) 61:2673–85. doi: 10.1007/s00394-022-02844-9
106.
TooheyJCTownsendJRJohnsonSBToyAMVantreaseWCBenderDet al. Effects of probiotic (Bacillus subtilis) supplementation during offseason resistance training in female division I athletes. J Strength Cond Res. (2020) 34:3173–81. doi: 10.1519/JSC.0000000000002675
107.
HarnettJEPyneDBMcKuneAJPenmJPumpaKL. Probiotic supplementation elicits favourable changes in muscle soreness and sleep quality in rugby players. J Sci Med Sport. (2021) 24:195–9. doi: 10.1016/j.jsams.2020.08.005
108.
ValidoECaposselaSGlisicMHertig-GodeschalkABertoloAStuckiGet al. Gut microbiome and inflammation among athletes in wheelchair in a crossover randomized pilot trial of probiotic and prebiotic interventions. Sci Rep. (2024) 14:12838. doi: 10.1038/S41598-024-63163-Z
109.
Hertig-GodeschalkAGlisicMRuettimannBValidoECaposselaSStoyanovJet al. The feasibility of a randomized controlled crossover trial to assess the effect of probiotic and prebiotic supplementation on the health of elite wheelchair athletes. Pilot Feasibility Stud. (2023) 9:339. doi: 10.1186/s40814-023-01339-6
110.
CaniPDKnaufC. How gut microbes talk to organs: the role of endocrine and nervous routes. Mol Metab. (2016) 5:743–52. doi: 10.1016/j.molmet.2016.05.011
111.
MishraSPJainSWangBWangSMillerBCLeeJYet al. Abnormalities in microbiota/butyrate/FFAR3 signaling in aging gut impair brain function. JCI Insight. (2024) 9:8443. doi: 10.1172/JCI.INSIGHT.168443
112.
TuJZhangJChenG. Higher dietary butyrate intake is associated with better cognitive function in older adults: evidence from a cross-sectional study. Front Aging Neurosci. (2025) 17:1522498. doi: 10.3389/fnagi.2025.1522498
113.
WangYAiZXingXFanYZhangYNanBet al. The ameliorative effect of probiotics on diet-induced lipid metabolism disorders: a review. Crit Rev Food Sci Nutr. (2024) 64:3556–72. doi: 10.1080/10408398.2022.2132377
114.
SabakaPKruzliakPBalazDKomornikovaACelovskaDCammarotaGet al. Effect of short term aerobic exercise on fasting and postprandial lipoprotein subfractions in healthy sedentary men. Lipids Health Dis. (2015) 14:1–8. doi: 10.1186/S12944-015-0148-5
115.
SuWLiuJWangAZhangHSunYYanZet al. Distinct lipidomic profiles but similar improvements in aerobic capacity following sprint interval training versus moderate-intensity continuous training in male adolescents. Front Physiol. (2025) 16:1475391. doi: 10.3389/fphys.2025.1475391
116.
ShirkoohiNMMohammadiHGallalyDQDjafarianK. The effects of probiotic supplementation on body composition, recovery following exercise-induced muscle damage, and exercise performance: a systematic review and meta-analysis of clinical trials. Phys Rep. (2025) 13:e70288. doi: 10.14814/PHY2.70288
Summary
Keywords
microbiome, sport, performance, probiotics, exercise
Citation
Teglas T and Radak Z (2025) Probiotic supplementation for optimizing athletic performance: current evidence and future perspectives for microbiome-based strategies. Front. Nutr. 12:1572687. doi: 10.3389/fnut.2025.1572687
Received
07 February 2025
Accepted
26 June 2025
Published
15 July 2025
Volume
12 - 2025
Edited by
Xin Zhang, Ningbo University, China
Reviewed by
Seyed Morteza Tayebi, Allameh Tabataba’i University, Iran
Heinz Kleinöder, German Sport University Cologne, Germany
Updates
Copyright
© 2025 Teglas and Radak.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zsolt Radak, radak.zsolt@tf.hu
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.