- 1School of Chemistry and Materials Science, Jiangsu Key Laboratory of Green Synthetic Chemistry for Functional Materials, Jiangsu Normal University, Xuzhou, China
- 2Institute of Chemistry and BioMedical Sciences, Collaborative Innovation Center of Chemistry for Life Sciences, Nanjing University, Nanjing, China
- 3Department of Chemistry and Biochemistry, Texas Tech University, Lubbock, TX, United States
A new Fe(III)-catalyzed bicyclization reaction of yne-allenones with indoles has been established, enabling the direct construction of cyclobuta[a]naphthalen-4-ols with an all-carbon quaternary center in good to excellent yields. This reaction was performed by using low-cost FeCl3 as the catalyst and EtOH as the environmentally benign solvent, providing a green protocol for constructing the cyclobutarene framework with a high degree of atom economy and functional group compatibility. The reaction mechanism was proposed to proceed through a [2 + 2] cycloaddition/1,6-conjugate addition cascade.
Introduction
Development of practical and sustainable synthetic methods for the rapid construction of valuable cyclic target molecules, along with minimum environmental impacts, represents an endeavor of utmost importance in both academia, and industry (Anastas and Warner, 1998; Bruckmann et al., 2008; Martins et al., 2009; Jiang et al., 2010; Huang et al., 2018a). In this context, chemical transformations following the principles of atom-economy are generally believed to be green since such reactions enable different molecular fragments into integrated cyclic frameworks by recombining chemical bonds with maximum atom utilization and minimum generation of the chemical waste (Trost, 1995, 2002; Trost et al., 2003; Banert and Plefka, 2011; Kotha et al., 2013). The key to realize this goal is to implement reaction cascades, which allow the direct formation of multiple chemical bonds in a one-pot operation and can lead to a remarkable increase in resource efficiency for the overall process (Barluenga et al., 2009; Fuerstner, 2009; Tietze et al., 2009; Jones et al., 2010; Wang et al., 2015; Sugimoto and Matsuya, 2017; Zhang et al., 2017). Specifically, bicyclization cascades have emerged as an important platform for the synthesis of bioactive small-molecule libraries for their SAR studies (Dömling et al., 2012; Brauch et al., 2013; Vlaar et al., 2013; Koopmanschap et al., 2014; Rotstein et al., 2014; Huang et al., 2018b). Due to their annulation efficiency, economic and environmental aspects, and ease of operation as well as diminished waste disposals (Jia et al., 2014; Su et al., 2014; Tian et al., 2015; Chen et al., 2017; Huang et al., 2017; Liu et al., 2017b,c; Wang L. et al., 2017). In view of the environmental awareness of the chemical community, the combination of the presented bicyclization strategy and the use of environmentally benign solvents will furnish the transformations under avoidance of potential pollutants (Bihani et al., 2013; Wang J.-Y. et al., 2017; Sha et al., 2018a). Nevertheless, the design and development of environmentally compatible bicyclization cascades without generation of toxic waste and by-products holds considerable challenges.
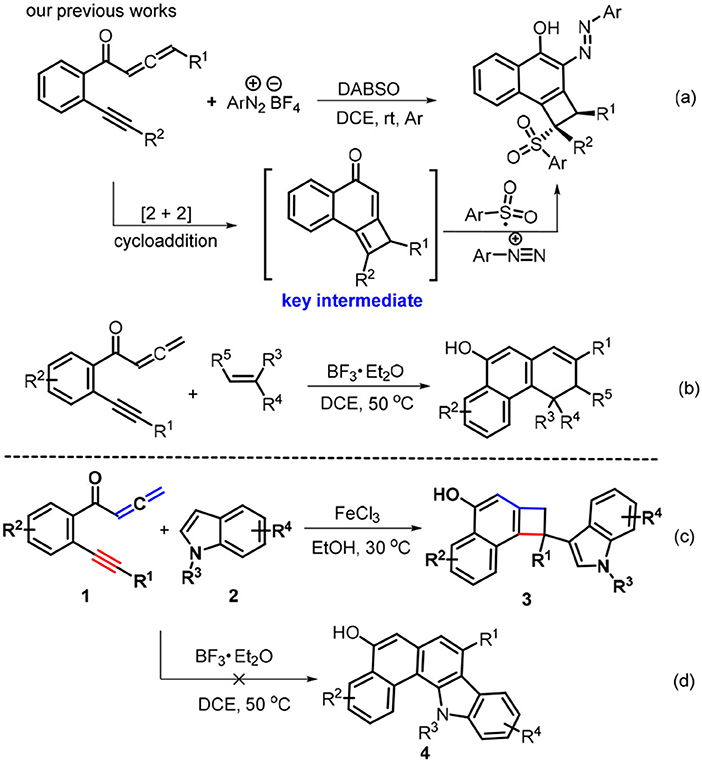
Cyclobutarenes are a class of structurally unique bicarbocyclic molecules which show a wide spectrum of biological activity (Christophe et al., 1998; Sadana et al., 2003). Due to the thermodynamic stability associated with the aromatic system and the kinetic reactivity of the strained cyclobutene ring (Cava and Napier, 1956; Mehta and Kotha, 2001), these molecules behave as reliable and synthetically useful feedstocks (Christophe et al., 1998) and have been extensively applied in natural product syntheses (Funk and Vollhardt, 1977, 1979; Grieco et al., 1980; Taber et al., 1987; Nemoto et al., 1995; Michellys et al., 2001). With these contributions in mind, great efforts to establish synthetic protocols for cyclobutarene synthesis have been developed which include 1,4-elimination-cycloaddition of functionalized arenes (Gray et al., 1978; Schirch et al., 1979; Sekine et al., 1979; Lenihan and Shechter, 1994, 1998; Chou et al., 1995), Parham cyclization (Bradsher and Hunt, 1981; Buchwald et al., 1987a,b; Beak and Selling, 1989; Aidhen and Ahuja, 1992), photo-induced cycloadditions (Parham et al., 1976; Kaneko and Naito, 1979; Neckers and Wagenaar, 1981; Kaneko et al., 1982; Kanao et al., 1983; Sato et al., 1987; Hoffmann and Pete, 1996), thermal extrusion reactions (Toda et al., 1988; D'Andrea et al., 1990; Hickman et al., 1991; Shimada et al., 1993; Andersen et al., 1996; Craig et al., 1998), intramolecular addition of carbanions to benzynes (Bunnett and Skorcz, 1962; Krohn et al., 1978; Gowland and Durst, 1979), [2 + 2 + 2] cycloadditions of 1,5-hexadiyne (Peter and Vollhardt, 1977, 1984; Funk and Vollhardt, 1980; McNichols and Stang, 1992), ring expansion of cycloproparenes (Birch et al., 1964; Iskander and Stansfield, 1965; Buckland et al., 1987; Kagabu and Saito, 1988; Müller et al., 1989), and [2 + 2] cycloadditions of allene precursors (Inanaga et al., 1992; Ezcurra and Moore, 1993; Toda et al., 1994) and other methods (Markgraf et al., 1969; Garratt and Nicolaides, 1972, 1974; Bilyard et al., 1979; Warrener et al., 1993). However, these methods encounter some drawbacks such as high temperatures (400°C-800°C), strong bases (n-BuLi and NaNH2), multiple steps, or a narrow substrate range. Moreover, indole derivatives stand for another important class of heterocyclic compounds present in a myriad of bioactive substances and natural products. Therefore, the development of general and sustainable entries toward cyclobutarene-indole pairs in atom- and pot-economic manner is of potential significance. Recently, we reported the combination of [2 + 2] cycloaddition with 1,4-radical addition reaction by treating yne-allenones with aryldiazonium salts and DABCO-bis(sulfur dioxide) (DABSO), affording functional cyclobuta[a]naphthalen-4-ols (Scheme 1a, Liu et al., 2017a). Subsequently, we developed a BF3•Et2O-catalyzed double [2 + 2] cycloaddition relay between yne-allenones and unactivated alkenes, enabling C-C triple bond cleavage to access phenanthren-9-ols (Scheme 1b, Li et al., 2018a). To continue our efforts in this project (Liu et al., 2017a; Wang J.-Y. et al., 2017; Li et al., 2018a,b; Sha et al., 2018b; Wang et al., 2018), we attempted to employ indoles 2 to be subjected with the reaction of yne-allenones 3 under our previous conditions (Li et al., 2018a) to assemble naphtho[1,2-a]carbazol-5-ols 4 (Scheme 1d), owing to indoles with C2 and C3 reactive sites could acted as C2 synthons for the synthesis of fused indoles (Haibach et al., 2011; Li et al., 2015; Liu et al., 2016; Ozaki et al., 2017). Unexpectedly, a double [2 + 2] cycloaddition relay did not occur. Instead, the reaction involved another [2 + 2] cycloaddition /1,6-addtition cascade to furnish 3-indolyl substituted cyclobuta[a]naphthalen-4-ols 3 by suitably adjusting the catalysts and solvents (Scheme 1c). Notably, the current green protocol represents an atom-economic and eco-friendly entry to structurally unique cyclobutarene-indole pairs through the combination of [2 + 2] cycloaddition with 1,6-conjugate addition by FeCl3 as a low-cost catalyst and EtOH as an environmentally benign solvent. Herein, we elaborate this attractive and benign transformation.
Results and Discussion
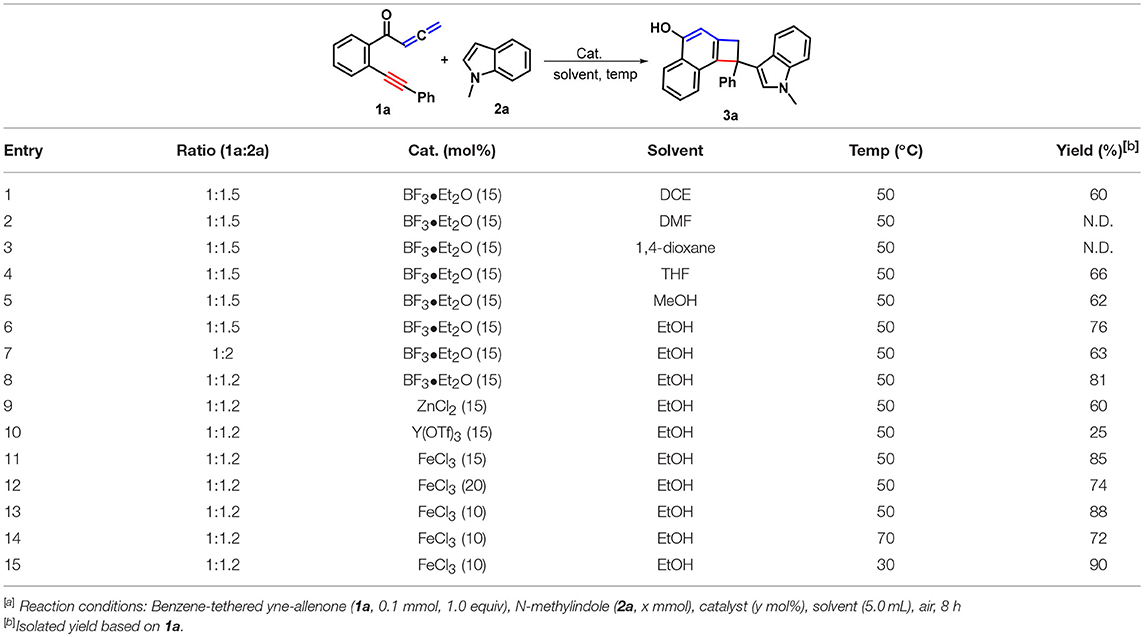
At the beginning of our studies, yne-allenone 1a and N-methylindole (2a) were chosen as the model substrates to explore the feasibility of double [2 + 2] cycloaddition relay reaction with our previous conditions (Table 1, entry 1). Instead of the expected naphtho[1,2-a]carbazol-5-ol 4a, 3-indolyl substituted cyclobuta[a]naphthalen-4-ol 3a was obtained in 60% yield. The following screening of solvents, such as N,N-dimethylformamide (DMF), 1,4-dioxane, tetrahydrofuran (THF), MeOH, and EtOH, showed that use of DMF and 1,4-dioxane as reaction media completely suppressed the reaction process (entries 2–3) whereas the latter three all made the transformations work more efficiently (entries 4–6). Among these, EtOH proved to be the best choice, providing the product 3a with the highest yield of 76% (entry 6). Increasing the component ratio to 1:2 is not beneficial for this transformation as a lower conversion was observed (63%, entry 7). In contrast, fine-tuning the component ratio to 1:1.2 could improve the reaction efficiency, resulting in a higher yield of 3a (81%, entry 8). As the next optimization step, we conducted the screening of a variety of Lewis acid catalysts, such as ZnCl2, Y(OTf)3 and FeCl3 that are often employed in the catalytic transformations, for this cyclization-addition cascade by using EtOH as the reaction media. The former two led to remarkably lower conversions (entries 9–10). Delightingly, the latter one showed the best catalytic performance in this transformation, delivering higher yield of 3a as compared with BF3•Et2O (85%, entry 11 vs. 8). It is found that the reaction efficiency was proven to display an important dependence on the loading of the Fe-catalyst. An increase in the FeCl3 loading had a detrimental impact on the reaction yield (entry 12) whereas reducing the catalytic amount of FeCl3 to 10 mol% could accelerate the conversion into 3a in an increased the yield to 88%. When the reaction temperature was elevated to 70°C, the reaction process was inhibited in some extent (entry 14). On the contrary, decreasing the reaction temperature to 30°C facilitated the current transformation and gave a higher yield of 90% (entry 15).
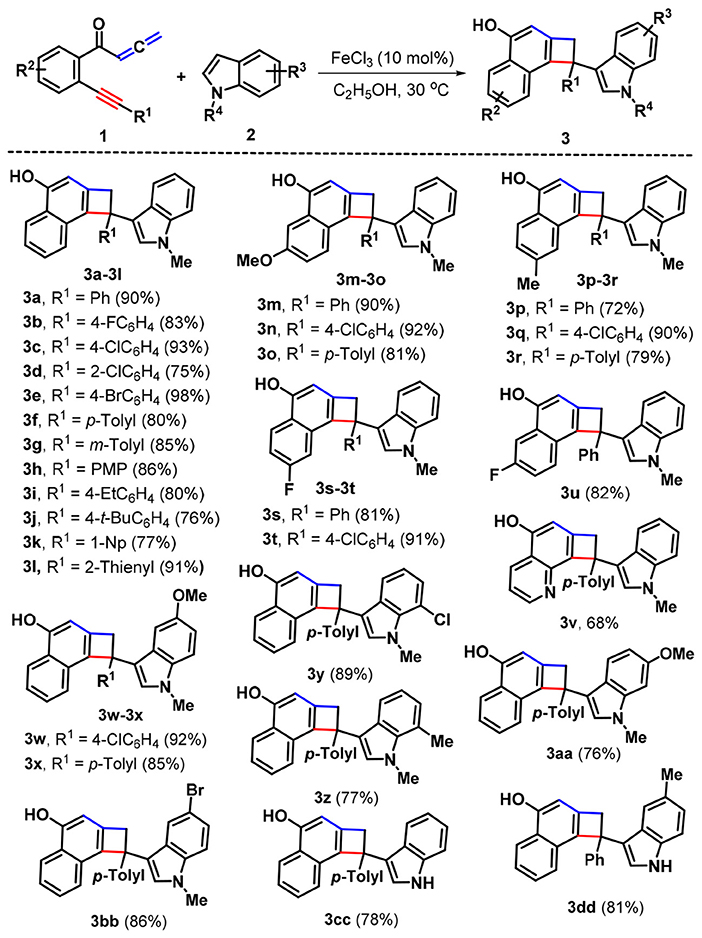
With these optimal conditions in hand (Table 1, entry 15), we set out to examine the scope of this Fe-catalyzed [2 + 2] cycloaddition /1,6-addition cascade by using a variety of yne-allenones and indoles. As depicted in Scheme 2, N-methylindole (2a) was first selected to evaluate the influence of substituents (R1) in the arylalkynyl moiety of yne-allenone 1. Both electron-poor and electron-rich groups at different positions of the arylalkynyl moiety (R1) can all tolerate this catalytic system, efficiently accessing the corresponding products 3b-3j in 75–98% yields. Diverse substituents, such as fluoro (1b), chloro (1c and 1d), bromo (1e), methyl (1f and 1g), methoxy (PMP = p-methoxyphenyl, 1h), ethyl(1i), t-butyl (1j) were suitable for this transformation. The presence of soft electron-withdrawing substituents (chloro, 1c and bromo, 1e) at the para-positions seemed to result in higher reactivity than that of electron-donating counterparts (3c and 3e vs. 3f and 3h-3j). Moreover, a sterically encumbered 1-naphthyl (1-Np) analog 1k was an effective candidate, which proceeded through a similar cyclization-addition process to give the corresponding product 3k in 77% yield. Besides, 2-thienyl counterpart 1l still showed high reactivity, delivering 2-thienyl product 3l in 91% yield. Next, we placed different functional groups (R2) including methoxy, methyl, and fluoro into the C4 or C5 position of the internal arene ring of substrates 1 and explored the synthetic utility of these substrates. Satisfyingly, all those substituents (1m−1u) would be compatible in the present reaction protocol, and the corresponding functionalized cyclobuta[a]naphthalen-4-ols 3m-3u in 72–92% yields were produced. Interestingly, the pyridine-tethered yne-allenone 1v could be successfully converted into cyclobutarene product 3v in 68% yield.
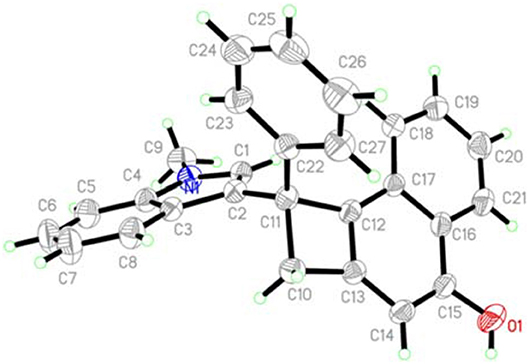
Next, the scope with respect to indoles components was evaluated. As anticipated, the different substituents including methoxy (2b), chloro (1c and 1d), bromo (1e), methyl (1f and 1g), located at different positions of the indole ring would be accommodated, confirming the reaction efficiency, as the cyclobuta[a]naphthalen-4-ol products 3w-3bb were offered in 76–92% yields. Finally, the free indole turned out to be a suitable reaction partner, leading to the formation of products 3cc and 3dd in 78 and 81% yields, respectively. Products 3 were fully characterized by their NMR and HR-MS spectral analysis. In the case of product 3a, its structure was further confirmed by X-ray crystallography (Figure 1).
Mechanism
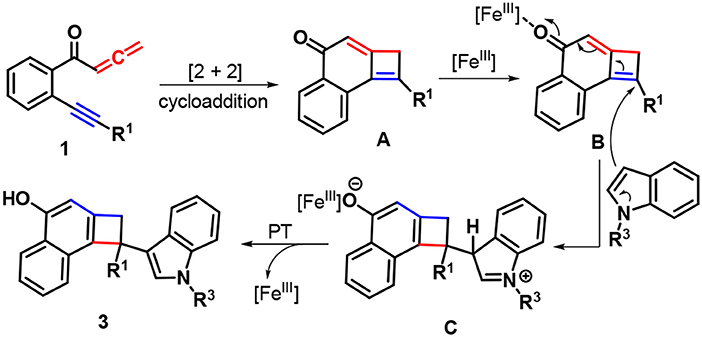
Based on the above experimental observations and literature reports (Haibach et al., 2011; Li et al., 2015, 2018a; Liu et al., 2016, 2017a; Ozaki et al., 2017; Sha et al., 2018b), a feasible mechanism for forming products 3 was proposed in Scheme 3. Initially, the intramolecular [2 + 2] cycloaddition of yne-allenones 1 rapidly occurs to yield cyclobutene intermediate A. In the presence of Fe-catalyst, 1,6-addition of indoles into intermediate B gives intermediate C, which converts into the final products 3 through proton transfer (PT), together with the regeneration of Fe-catalyst.
Conclusion
In summary, starting from readily available yne-allenones and indoles, we have established a new Fe-catalyzed [2 + 2] cycloaddition/1,6-conjugate addition cascade for the high-efficient and benign synthesis of a variety of 3-indolyl cyclobuta[a]naphthalen-4-ols with good to excellent yields. The current green protocol has the advantages of broad scope of substrates, good tolerance of functional group and high atom utilization as well as mild reaction conditions. Further application of the resulting cyclobutarenes is underway in our laboratory.
Materials and Methods
General
All melting points are uncorrected. The NMR spectra were recorded in CDCl3 or DMSO-d6 on a 400 MHz instrument with TMS as internal standard. Chemical shifts (δ) were reported in ppm with respect to TMS. Data are represented as follows: chemical shift, mutiplicity (s = singlet, d = doublet, t = triplet, m = multiplet), coupling constant (J, Hz) and integration. HRMS analyses were carried out using a TOF-MS instrument with an ESI source. X-Ray crystallographic analysis was performed with a SMART CCD and a P4 diffractometer (the copies of NMR see Supplementary Material). All yne-allenones 1 are known compounds and their preparation followed the previously reported procedures (Wei et al., 2009; Liu et al., 2017a; Li et al., 2018b).
General Procedure for the Synthesis of 3
Example for the Synthesis of 3a
1-(2-(Phenylethynyl)phenyl)buta-2,3-dien-1-one (1a, 0.3 mmol, 73.2 mg) was added to a 10-mL reaction tubing under the air conditions. Then, N-methylindole (2a, 0.36 mmol, 47.2 mg) and EtOH (5 mL) were continuously added into the above reaction mixture. Subsequently, FeCl3 (10 mol%, 4.8 mg) was added to the reaction system. Then the mixture was stirred at 30°C for 8 h until complete consumption of 1a as monitored by TLC analysis. After the reaction was finished, the reaction mixture was concentrated in vacuum, and the resulting residue was purified by column chromatography on silica gel (eluent, petroleum ether/ethyl acetate = 20:1) to afford the desired product 3a as a white solid.
1-(1-methyl-1H-indol-3-yl)-1-phenyl-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3a)
White solid, 102 mg, 90% yield; mp 179-181°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.23 (s, 1H), 8.24 (d, J = 8.0 Hz, 1H), 7.51 (d, J = 7.6 Hz, 1H), 7.38 (m, 5H), 7.29 (m, 2H), 7.21 (m, 1H), 7.07 (m, 1H), 6.91-6.78 (m, 4H), 3.88 (m, 2H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.8, 146.1, 140.3, 137.7, 137.0, 130.1, 128.5, 128.1, 127.5, 127.4, 126.8, 126.5, 125.2, 124.4, 123.9, 122.4, 121.5, 120.1, 119.0, 118.8, 110.3, 104.9, 54.4, 47.7, 32.7; IR (KBr, ν, cm−1) 3341, 3044, 1579, 1474, 1228, 1181, 1024, 905, 806, 738; HRMS (APCI-TOF) m/z calcd for C27H20NO [M-H]− 374.1545; found 374.1546.
1-(4-fluorophenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3b)
White solid, 97 mg, 83% yield; mp 160-162°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.24 (s, 1H), 8.25 (d, J = 8.0 Hz, 1H), 7.49 (d, J = 8.0 Hz, 1H), 7.43-7.34 (m, 5H), 7.11 (m, 3H), 6.87 (m, 4H), 3.88 (m, 2H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 162.2 (1JCF = 240.5 Hz), 159.8, 154.9, 142.3 (4JCF = 2.9 Hz), 142.2, 140.3, 137.7, 136.8, 129.9, 129.4 (3JCF = 7.9 Hz), 129.3, 128.1, 127.5, 126.7, 125.2, 124.4, 123.9, 122.2, 121.6, 120.0, 119.1, 118.6, 115.3 (2JCF = 21.0 Hz), 115.1, 110.4, 105.0, 53.7, 47.9, 32.7; IR (KBr, ν, cm−1) 3399, 3051, 1573, 1464, 1226, 1188, 1014, 904, 808, 748; HRMS (APCI-TOF) m/z calcd for C27H19FNO [M-H]− 392.1451; found 392.1453.
1-(4-chlorophenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3c)
White solid, 114 mg, 93% yield; mp 170-172°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.27 (s, 1H), 8.24 (d, J = 8.0 Hz, 1H), 7.47 (d, J = 7.6 Hz, 1H), 7.44-7.31 (m, 7H), 7.12-7.06 (m, 1H), 6.93 (s, 1H), 6.87 (m, 3H), 3.82 (m, 2H), 3.69 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.9, 145.1, 140.3, 137.7, 136.5, 131.0, 129.9, 129.3, 128.5, 128.2, 127.6, 126.7, 125.2, 124.4, 124.0, 122.1, 121.6, 120.0, 119.1, 118.2, 110.4, 104.9, 53.7, 47.8, 32.7; IR (KBr, ν, cm−1) 3407, 3031, 1563, 1441, 1223, 1103, 1012, 909, 838, 741; HRMS (APCI-TOF) m/z calcd for C27H19ClNO [M-H]− 408.1156; found 408.1143.
1-(2-chlorophenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3d)
White solid, 92 mg, 75% yield; mp 177-179°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.31 (s, 1H), 8.30-8.19 (m, 1H), 7.65 (d, J = 7.6 Hz, 1H), 7.53 (d, J = 7.6 Hz, 1H), 7.44-7.28 (m, 6H), 6.99 (m, 1H), 6.93 (s, 1H), 6.79 (s, 1H), 6.69 (m, 1H), 6.60 (d, J = 8.0 Hz, 1H), 4.32 (d, J = 14.4 Hz, 1H), 3.71 (s, 1H), 3.66 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 155.2, 143.4, 140.3, 137.7, 135.9, 134.0, 131.0, 130.5, 130.1, 128.9, 127.9, 127.6, 127.4, 126.2, 125.2, 124.4, 123.9, 122.2, 121.1, 119.3, 118.9, 116.1, 110.2, 104.7, 55.3, 47.5, 32.7; IR (KBr, ν, cm−1) 3442, 3022, 1533, 1421, 1203, 1123, 1010, 903, 834, 721; HRMS (APCI-TOF) m/z calcd for C27H19ClNO [M-H]− 408.1156; found 408.1162.
1-(4-bromophenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3e)
White solid, 133 mg, 98% yield; mp 166-168°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.26 (s, 1H), 8.24 (d, J = 8.0 Hz, 1H), 7.48 (d, J = 8.4 Hz, 3H), 7.43-7.34 (m, 3H), 7.30 (d, J = 8.4 Hz, 2H), 7.09 (m, 1H), 6.93 (s, 1H), 6.92-6.83 (m, 3H), 3.81 (m, 2H), 3.69 (s, 3H). 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.9, 145.5, 140.3, 137.7, 136.5, 131.5, 129.9, 129.7, 128.2, 127.6, 126.7, 125.3, 124.4, 124.0, 122.1, 121.6, 120.0, 119.6, 119.1, 118.1, 110.4, 104.9, 53.8, 47.8, 32.7. IR (KBr, ν, cm−1) 3412, 3021, 1538, 1391, 1202, 1129, 1017, 906, 818, 733; HRMS (APCI-TOF) m/z calcd for C27H19BrNO [M-H]− 452.0651; found 452.0634.
1-(1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3f)
White solid, 94 mg, 80% yield; mp 155-157°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.20 (s, 1H), 8.23 (d, J = 8.0 Hz, 1H), 7.49 (d, J = 8.0 Hz, 1H), 7.42-7.34 (m, 3H), 7.26 (d, J = 8.0 Hz, 2H), 7.08 (d, J = 8.0 Hz, 3H), 6.92-6.79 (m, 4H), 3.80 (m, 2H), 3.68 (s, 3H), 2.26 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.7, 143.0, 140.3, 137.7, 137.2, 135.3, 130.0, 129.1, 128.0, 127.4, 127.3, 126.8, 125.2, 124.3, 123.8, 122.4, 121.5, 120.2, 119.0, 118.9, 110.3, 105.0, 54.0, 47.8, 32.7, 21.1; IR (KBr, ν, cm−1) 3418, 3009, 1541, 1401, 1192, 1121, 1012, 916, 813, 730; HRMS (APCI-TOF) m/z calcd for C28H22NO [M-H]− 388.1702; found 388.1723.
1-(1-methyl-1H-indol-3-yl)-1-(m-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3g)
White solid, 99 mg, 85% yield; mp 149-151°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.20 (s, 1H), 8.23 (d, J = 8.4 Hz, 1H), 7.51 (d, J = 8.0 Hz, 1H), 7.38 (m, 3H), 7.23 (s, 1H), 7.18 (d, J = 5.6 Hz, 2H), 7.09-7.01 (m, 2H), 6.85 (m, 4H), 3.82 (m, 2H), 3.68 (s, 3H), 2.22 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.7, 146.0, 140.3, 137.7, 137.4, 137.1, 128.4, 128.0, 127.4, 127.2, 126.8, 125.2, 124.8, 124.3, 123.8, 120.1, 118.9, 110.3, 105.0, 54.3, 47.7, 32.7, 21.8; IR (KBr, ν, cm−1) 3411, 2966, 1511, 1406, 1162, 1091, 1015, 911, 833, 727; HRMS (APCI-TOF) m/z calcd for C28H22NO [M-H]− 388.1702; found 388.1720.
1-(4-methoxyphenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3h)
White solid, 105 mg, 86% yield; mp 167-169°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.19 (s, 1H), 8.25 (d, J = 8.0 Hz, 1H), 7.50 (d, J = 8.0 Hz, 1H), 7.42-7.33 (m, 3H), 7.29 (d, J = 8.8 Hz, 2H), 7.07 (m, 1H), 6.91-6.81 (m, 6H), 3.79 (m, 2H), 3.70 (s, 3H), 3.67 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 157.9, 154.7, 140.3, 138.1, 137.7, 137.3, 130.0, 128.5, 128.0, 127.3, 126.8, 125.2, 124.4, 123.8, 122.4, 121.5, 120.2, 119.2, 118.9, 113.8, 110.3, 105.0, 55.4, 53.7, 47.9, 32.7; IR (KBr, ν, cm−1) 3417, 2996, 1517, 1403, 1177, 1096, 1013, 914, 845, 720; HRMS (APCI-TOF) m/z calcd for C28H22NO2 [M-H]− 404.1651; found 404.1638.
1-(4-ethylphenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3i)
White solid, 97 mg, 80% yield; mp 165-167°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.19 (s, 1H), 8.24 (d, J = 8.0 Hz, 1H), 7.50 (d, J = 8.0 Hz, 1H), 7.42-7.33 (m, 3H), 7.29 (d, J = 8.0 Hz, 2H), 7.13-7.04 (m, 3H), 6.90-6.77 (m, 4H), 3.80 (m, 2H), 3.68 (s, 3H), 2.56 (m, 2H), 1.15 (m, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.7, 143.3, 141.7, 140.3, 137.7, 137.2, 130.0, 128.0, 127.9, 127.4, 127.4, 126.8, 125.2, 124.3, 123.8, 122.4, 121.5, 120.2, 119.0, 118.9, 110.3, 105.0, 54.0, 47.8, 32.7, 28.2, 16.0; IR (KBr, ν, cm−1) 3387, 3013, 1510, 1406, 1167, 1091, 1012, 918, 832, 722; HRMS (APCI-TOF) m/z calcd for C29H24NO [M-H]− 402.1858; found 402.1874.
1-(4-(tert-butyl)phenyl)-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3j)
White solid, 104 mg, 80% yield; mp 178-180°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.19 (s, 1H), 8.24 (d, J = 8.4 Hz, 1H), 7.53 (d, J = 8.0 Hz, 1H), 7.35 (m, 7H), 7.07 (m, 1H), 6.93-6.78 (m, 4H), 3.81 (m, 2H), 3.68 (s, 3H), 1.25 (s, 9H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.7, 148.5, 143.0, 140.3, 137.7, 137.1, 130.1, 128.0, 127.4, 127.2, 126.8, 125.3, 125.2, 124.3, 123.8, 122.5, 121.5, 120.2, 119.0, 118.9, 110.3, 104.9, 54.0, 47.7, 34.5, 32.7, 31.6; IR (KBr, ν, cm−1) 3402, 3010, 1512, 1423, 1177, 1093, 1018, 933, 814, 711; HRMS (APCI-TOF) m/z calcd for C31H28NO [M-H]− 430.2171; found 430.2187.
1-(1-methyl-1H-indol-3-yl)-1-(naphthalen-1-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3k)
White solid, 98 mg, 77% yield; mp 162-164°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.25 (s, 1H), 8.27 (d, J = 8.0 Hz, 1H), 7.89-7.81 (m, 3H), 7.71 (d, J = 7.2 Hz, 1H), 7.57 (m, 2H), 7.47-7.34 (m, 5H), 7.08 (m, 1H), 6.95 (s, 1H), 6.91 (d, J = 6.0 Hz, 2H), 6.81 (m, 1H), 3.93 (s, 2H), 3.69 (m, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.9, 143.7, 140.4, 137.8, 137.1, 133.3, 132.1, 130.1, 128.2, 128.1, 127.8, 127.5, 126.9, 126.6, 126.5, 126.0, 125.3, 125.2, 124.4, 123.9, 122.3, 121.5, 120.1, 119.0, 118.6, 110.4, 105.1, 54.5, 47.5, 32.7; IR (KBr, ν, cm−1) 3422, 3014, 1510, 1413, 1171, 1088, 1015, 937, 825, 727; HRMS (APCI-TOF) m/z calcd for C31H22NO [M-H]− 424.1702; found 424.1713.
1-(1-methyl-1H-indol-3-yl)-1-(thiophen-2-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3l)
White solid, 104 mg, 91% yield; mp 158-160°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.26 (s, 1H), 8.26 (d, J = 8.4 Hz, 1H), 7.71 (d, J = 8.4 Hz, 1H), 7.48 (m, 1H), 7.43-7.36 (m, 2H), 7.31 (d, J = 4.8 Hz, 1H), 7.12 (m, 2H), 6.99 (d, J = 5.6 Hz, 2H), 6.92 (m, 2H), 6.85 (s, 1H), 3.98 (d, J = 13.6 Hz, 1H), 3.77 (d, J = 13.6 Hz, 1H), 3.69 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 155.0, 151.2, 140.1, 137.6, 136.8, 129.5, 127.5, 126.9, 126.6, 125.3, 124.6, 124.5, 124.4, 124.0, 122.4, 121.6, 120.2, 119.1, 119.0, 110.4, 105.0, 50.9, 48.9, 32.7; IR (KBr, ν, cm−1) 3427, 3050, 1517, 1421, 1178, 1068, 1020, 936, 821, 734; HRMS (APCI-TOF) m/z calcd for C25H18NOS [M-H]− 380.1110; found 380.1108.
6-methoxy-1-(1-methyl-1H-indol-3-yl)-1-phenyl-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3m)
White solid, 110 mg, 90% yield; mp 172-174°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.11 (s, 1H), 7.58 (d, J = 2.4 Hz, 1H), 7.43 (d, J = 8.8 Hz, 1H), 7.37 (m, 3H), 7.29 (m, 2H), 7.20 (m, 1H), 7.11-7.05 (m, 2H), 6.88-6.79 (m, 4H), 3.87 (d, J = 13.2 Hz, 1H), 3.84 (s, 3H), 3.76 (d, J = 13.6 Hz, 1H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 156.1, 153.7, 146.1, 137.7, 137.3, 137.2, 128.5, 128.1, 127.5, 126.8, 126.4, 126.2, 125.5, 124.0, 121.5, 120.1, 119.6, 118.9, 110.3, 105.4, 103.2, 55.5, 54.3, 47.7, 32.7; IR (KBr, ν, cm−1) 3387, 3045, 1507, 1422, 1172, 1058, 1011, 932, 827, 739; HRMS (APCI-TOF) m/z calcd for C28H22NO2 [M-H]− 404.1651; found 404.1630.
1-(4-chlorophenyl)-6-methoxy-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3n)
White solid, 121 mg, 92% yield; mp 175-177°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.15 (s, 1H), 7.59 (d, J = 2.4 Hz, 1H), 7.44-7.31 (m, 6H), 7.13-7.05 (m, 2H), 6.94-6.81 (m, 4H), 3.85 (s, 3H), 3.83-3.72 (m, 2H), 3.68 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 156.2, 153.9, 145.1, 137.7, 137.2, 136.7, 131.0, 129.3, 128.5, 128.1, 126.7, 126.3, 125.3, 123.8, 121.6, 120.0, 119.7, 119.1, 118.3, 110.4, 105.4, 103.2, 55.5, 53.7, 47.7, 32.7. IR (KBr, ν, cm−1) 3382, 3025, 1523, 1421, 1192, 1074, 1008, 934, 828, 736. HRMS (APCI-TOF) m/z calcd for C28H21NClO2 [M-H]− 438.1261; found 438.1277.
6-methoxy-1-(1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3o)
White solid, 102 mg, 81% yield; mp 170-172°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.09 (s, 1H), 7.59 (d, J = 2.4 Hz, 1H), 7.43 (d, J = 9.2 Hz, 1H), 7.35 (d, J = 8.0 Hz, 1H), 7.26 (d, J = 8.0 Hz, 2H), 7.07 (m, 4H), 6.89-6.80 (m, 4H), 3.84 (s, 3H), 3.83-3.72 (m, 2H), 3.67 (s, 3H), 2.26 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 156.1, 153.6, 143.1, 137.7, 137.4, 137.3, 135.3, 129.1, 128.0, 127.4, 126.8, 126.2, 125.5, 124.0, 121.5, 120.2, 119.5, 119.1, 118.9, 110.2, 105.4, 103.1, 55.5, 54.0, 47.7, 32.6, 21.1; IR (KBr, ν, cm−1) 3387, 3022, 1521, 1425, 1202, 1094, 1016, 942, 835, 721; HRMS (APCI-TOF) m/z calcd for C29H24NO2 [M-H]− 418.1808; found 418.1822.
7-methyl-1-(1-methyl-1H-indol-3-yl)-1-phenyl-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3p)
White solid, 84 mg, 72% yield; mp 147-149°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.12 (s, 1H), 8.14 (d, J = 8.8 Hz, 1H), 7.38 (m, 3H), 7.32-7.26 (m, 3H), 7.20 (m, 2H), 7.08 (m, 1H), 6.88 (d, J = 4.4 Hz, 2H), 6.86-6.77 (m, 2H), 3.86 (d, J = 13.6 Hz, 1H), 3.73 (d, J = 13.6 Hz, 1H), 3.69 (s, 3H), 2.32 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.8, 146.2, 140.5, 137.7, 136.6, 136.3, 130.4, 128.5, 128.1, 127.5, 126.8, 126.4, 126.0, 124.3, 123.5, 121.5, 121.3, 120.2, 118.9, 118.9, 110.3, 104.2, 54.4, 47.9, 32.7, 22.0; IR (KBr, ν, cm−1) 3507, 3052, 1522, 1420, 1201, 1098, 1015, 944, 841, 727; HRMS (APCI-TOF) m/z calcd for C28H22NO [M-H]− 388.1702; found 388.1727.
1-(4-chlorophenyl)-7-methyl-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3q)
White solid, 114 mg, 90% yield; mp 152-154°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.16 (s, 1H), 8.14 (d, J = 8.4 Hz, 1H), 7.40-7.33 (m, 5H), 7.25 (s, 1H), 7.19 (d, J = 8.8 Hz, 1H), 7.09 (m, 1H), 6.94-6.83 (m, 3H), 6.79 (s, 1H), 3.85 (d, J = 13.6 Hz, 1H), 3.73 (s, 1H), 3.70 (s, 3H), 2.33 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.9, 145.2, 140.4, 137.7, 136.7, 135.9, 131.0, 130.2, 129.4, 128.5, 128.2, 126.7, 126.1, 124.4, 123.5, 121.6, 121.1, 120.1, 119.1, 118.3, 110.4, 104.2, 53.7, 47.9, 32.7, 22.0; IR (KBr, ν, cm−1) 3495, 3042, 1520, 1402, 1221, 1084, 1016, 947, 831, 709; HRMS (APCI-TOF) m/z calcd for C28H21ClNO [M-H]− 422.1312; found 422.1314.
7-methyl-1-(1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3r)
White solid, 96 mg, 79% yield; mp 153-155°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.10 (s, 1H), 8.14 (d, J = 8.4 Hz, 1H), 7.36 (d, J = 8.4 Hz, 1H), 7.26 (d, J = 8.0 Hz, 3H), 7.18 (d, J = 8.8 Hz, 1H), 7.08 (m, 3H), 6.90 (d, J = 8.8 Hz, 2H), 6.83 (m, 1H), 6.79 (s, 1H), 3.88-3.69 (m, 2H), 3.68 (s, 3H), 2.32 (s, 3H), 2.26 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.7, 143.1, 140.4, 137.7, 136.5, 136.5, 135.3, 130.3, 129.1, 128.0, 127.4, 126.9, 126.0, 124.3, 123.4, 121.5, 121.3, 120.3, 119.0, 118.9, 110.3, 104.2, 54.0, 47.9, 32.7, 22.0, 21.1; IR (KBr, ν, cm−1) 3490, 3031, 1500, 1422, 1213, 1090, 1012, 948, 843, 719; HRMS (APCI-TOF) m/z calcd for C29H24NO [M-H]− 402.1858; found 402.1844.
7-fluoro-1-(1-methyl-1H-indol-3-yl)-1-phenyl-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3s)
White solid, 96 mg, 81% yield; mp 144-146°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.36 (s, 1H), 7.87 (m, 1H), 7.55 (m, 1H), 7.33 (m, 6H), 7.21 (m, 1H), 7.08 (m, 1H), 6.93 (s, 1H), 6.91-6.77 (m, 3H), 3.84 (m, 2H), 3.68 (s, 3H). 13C NMR (100 MHz, DMSO-d6; δ, ppm) 160.3 (1JCF = 239.4 Hz), 157.9, 154.2, 154.1, 145.8, 139.7 (6JCF = 2.4 Hz), 139.6, 137.7, 137.3, 128.6, 128.1, 127.5, 127.2, 126.8, 126.7 (3JCF = 19.7 Hz), 125.9 (5JCF = 8.1 Hz), 125.8, 125.1 (4JCF = 8.4 Hz), 125.0, 121.5, 120.0, 119.0, 118.6, 117.5, 117.3, 110.3, 108.0 (2JCF = 21.7 Hz), 107.8, 106.0, 54.4, 47.8, 32.7; IR (KBr, ν, cm−1) 3501, 3021, 1502, 1421, 1215, 1099, 1014, 962, 883, 712; HRMS (APCI-TOF) m/z calcd for C27H19FNO [M-H]− 392.1451; found 392.1459.
1-(4-chlorophenyl)-7-fluoro-1-(1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3t)
White solid, 117 mg, 91% yield; mp 149-151°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.44 (s, 1H), 8.30 (m, 1H), 7.41-7.32 (m, 5H), 7.24 (m, 1H), 7.12-7.04 (m, 2H), 6.98 (s, 1H), 6.86 (d, J = 13.2 Hz, 3H), 3.82 (m, 2H), 3.71 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 160.0 (1JCF = 231.6 Hz), 157.1, 155.3, 144.8, 142.4, 137.8, 136.2 (6JCF = 3.4 Hz), 136.1, 131.1, 130.5 (5JCF = 9.6 Hz), 130.4, 129.3, 128.6, 128.2, 127.8 (4JCF = 9.9 Hz), 126.6, 122.4, 121.6, 119.9, 119.2, 117.8, 113.9 (2JCF = 24.1 Hz), 113.6, 110.5, 105.5 (3JCF = 20.3 Hz), 105.3, 104.5, 53.7, 47.7, 32.7; IR (KBr, ν, cm−1) 3512, 3020, 1505, 1411, 1210, 1095, 1010, 966, 861, 740; HRMS (APCI-TOF) m/z calcd for C27H18ClFNO [M-H]− 426.1061; found 426.1066.
6-fluoro-1-(1-methyl-1H-indol-3-yl)-1-phenyl-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3u)
White solid, 97 mg, 82% yield; mp 148-150°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.41 (s, 1H), 8.30 (m, 1H), 7.38 (d, J = 8.8 Hz, 3H), 7.30 (m, 2H), 7.24 (m, 2H), 7.08 (m, 2H), 6.94 (s, 1H), 6.90-6.79 (m, 3H), 3.84 (m, 2H), 3.70 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 162.3 (1JCF = 243.8 Hz), 160.0, 155.1, 145.8, 142.4, 137.8, 136.7 (6JCF = 5.1 Hz), 130.7 (5JCF = 9.3 Hz), 130.6, 128.6, 128.1, 127.8, 127.7 (4JCF = 9.6 Hz), 127.4, 126.7, 126.6, 122.4, 121.5, 120.0, 119.0, 118.4, 113.8 (2JCF = 24.8 Hz), 113.5, 110.4, 105.7 (3JCF = 20.1 Hz), 105.5, 104.6, 54.3, 47.7, 32.7; IR (KBr, ν, cm−1) 3502, 3047, 1502, 1422, 1235, 1099, 1015, 946, 851, 733; HRMS (APCI-TOF) m/z calcd for C27H19FNO [M-H]− 392.1451; found 392.1450.
8-(1-methyl-1H-indol-3-yl)-8-(p-tolyl)-7,8-dihydrocyclobuta[h]quinolin-5-ol (3v)
White solid, 80 mg, 68% yield; mp 162-164°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.56 (s, 1H), 8.88 (m, 1H), 8.60 (d, J = 8.4 Hz, 1H), 7.72 (d, J = 8.0 Hz, 2H), 7.41 (dd, J = 8.4, 4.2 Hz, 1H), 7.32 (d, J = 8.0 Hz, 1H), 7.23 (d, J = 8.0 Hz, 1H), 7.07 (d, J = 8.0 Hz, 3H), 7.01 (s, 1H), 6.90 (s, 1H), 6.85 (m, 1H), 3.91 (m, 2H), 3.64 (s, 3H), 2.23 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 155.0, 151.2, 145.1, 143.9, 137.5, 137.1, 135.0, 132.7, 128.8, 128.3, 128.1, 126.5, 121.3, 120.4, 120.3, 120.1, 119.3, 118.8, 110.1, 105.3, 55.2, 46.1, 32.6, 21.1; IR (KBr, ν, cm−1) 3545, 3067, 1552, 1421, 1230, 1129, 1012, 949, 840, 730; HRMS (APCI-TOF) m/z calcd for C27H21N2O [M-H]− 389.1654; found 389.1657.
1-(4-chlorophenyl)-1-(5-methoxy-1-methyl-1H-indol-3-yl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3w)
White solid, 121 mg, 92% yield; mp 166-168°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.27 (s, 1H), 8.27 (d, J = 8.0 Hz, 1H), 7.48 (d, J = 8.0 Hz, 1H), 7.44-7.33 (m, 6H), 7.27 (d, J = 8.8 Hz, 1H), 6.88 (d, J = 10.8 Hz, 2H), 6.74 (m, 1H), 6.24 (d, J = 2.0 Hz, 1H), 3.81 (m, 2H), 3.64 (s, 3H), 3.49 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 155.0, 153.3, 145.0, 140.5, 136.5, 133.1, 131.1, 130.0, 129.4, 128.7, 128.5, 127.6, 127.0, 125.3, 124.4, 124.0, 122.2, 117.6, 111.0, 110.9, 104.8, 102.4, 55.5, 53.8, 47.7, 32.8; IR (KBr, ν, cm−1) 3504, 2997, 1534, 1401, 1233, 1149, 1010, 942, 847, 736; HRMS (APCI-TOF) m/z calcd for C28H21NClO2 [M-H]− 438.1261; found 438.1267.
1-(5-methoxy-1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3x)
White solid, 107 mg, 85% yield; mp 170-172°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.21 (s, 1H), 8.25 (d, J = 8.0 Hz, 1H), 7.49 (d, J = 8.0 Hz, 1H), 7.43-7.33 (m, 2H), 7.29 (d, J = 8.0 Hz, 2H), 7.25 (d, J = 8.8 Hz, 1H), 7.11 (d, J = 7.6 Hz, 2H), 6.88 (s, 1H), 6.82 (s, 1H), 6.72 (m, 1H), 6.22 (d, J = 2.0 Hz, 1H), 3.79 (m, 2H), 3.63 (s, 3H), 3.47 (s, 3H), 2.27 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.8, 153.1, 143.0, 140.5, 137.1, 135.3, 133.1, 130.2, 129.1, 128.6, 127.5, 127.4, 127.1, 125.2, 124.4, 123.8, 122.4, 118.4, 110.8, 110.7, 104.9, 102.6, 55.5, 54.1, 47.7, 32.8, 21.1; IR (KBr, ν, cm−1) 3487, 3022, 1520, 1422, 1200, 1097, 1012, 944, 833, 725; HRMS (APCI-TOF) m/z calcd for C29H24NO2 [M-H]− 418.1808; found 418.1838.
1-(7-chloro-1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3y)
White solid, 113 mg, 89% yield; mp 163-165°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.24 (s, 1H), 8.24 (d, J = 8.0 Hz, 1H), 7.48 (d, J = 7.6 Hz, 1H), 7.38 (m, 2H), 7.23 (d, J = 8.0 Hz, 2H), 7.06 (d, J = 8.4 Hz, 2H), 6.93 (s, 1H), 6.88-6.76 (m, 4H), 3.99 (s, 3H), 3.78 (s, 2H), 2.24 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.8, 142.5, 140.2, 136.9, 135.5, 132.6, 131.2, 130.2, 129.9, 129.2, 127.4, 127.3, 125.2, 124.4, 123.9, 123.0, 122.2, 120.1, 119.6, 119.3, 116.5, 104.9, 53.6, 47.6, 36.5, 21.0; IR (KBr, ν, cm−1) 3505, 3022, 1524, 1400, 1209, 1082, 1012, 945, 833, 719; HRMS (APCI-TOF) m/z calcd for C28H21ClNO [M-H]− 422.1312; found 422.1304.
1-(1,7-dimethyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3z)
White solid, 93 mg, 77% yield; mp 165-167°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.17 (s, 1H), 8.23 (d, J = 8.0 Hz, 1H), 7.48 (d, J = 8.0 Hz, 1H), 7.37 (m, 2H), 7.22 (d, J = 8.0 Hz, 2H), 7.06 (d, J = 8.0 Hz, 2H), 6.85 (s, 1H), 6.79-6.64 (m, 4H), 3.93 (s, 3H), 3.77 (s, 2H), 2.68 (s, 3H), 2.25 (s, 3H). 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.6, 143.0, 140.3, 137.4, 136.3, 135.3, 129.9, 129.6, 129.1, 128.0, 127.3, 125.2, 124.3, 124.0, 123.8, 122.4, 121.8, 119.1, 118.5, 118.4(5), 118.3(9), 105.0, 53.8, 47.6, 36.6, 21.1, 19.7; IR (KBr, ν, cm−1) 3468, 3056, 1480, 1424, 1203, 1095, 1010, 958, 844, 722; HRMS (APCI-TOF) m/z calcd for C29H24NO [M-H]− 402.1858; found 402.1844.
1-(6-methoxy-1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3aa)
White solid, 96 mg, 76% yield; mp 171-173°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.18 (s, 1H), 8.23 (d, J = 8.0 Hz, 1H), 7.48 (d, J = 8.0 Hz, 1H), 7.41-7.32 (m, 2H), 7.26 (d, J = 8.0 Hz, 2H), 7.08 (d, J = 8.0 Hz, 2H), 6.89 (d, J = 2.0 Hz, 1H), 6.85 (s, 1H), 6.70 (d, J = 10.4 Hz, 2H), 6.48 (m, 1H), 3.81 (m, 2H), 3.75 (s, 3H), 3.63 (s, 3H), 2.26 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 156.0, 154.6, 143.1, 140.3, 138.5, 137.3, 135.3, 130.0, 129.1, 127.4, 127.3, 126.8, 125.2, 124.3, 123.8, 122.4, 121.1, 120.7, 119.1, 109.0, 105.0, 93.6, 55.7, 54.0, 47.8, 32.7, 21.1; IR (KBr, ν, cm−1) 3487, 3020, 1521, 1420, 1212, 1091, 1011, 944, 836, 729; HRMS (APCI-TOF) m/z calcd for C29H24NO2 [M-H]− 418.1808; found 418.1822.
1-(5-bromo-1-methyl-1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3bb)
White solid, 121 mg, 76% yield; mp 175-177°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.22 (s, 1H), 8.25 (d, J = 8.0 Hz, 1H), 7.46 (d, J = 8.0 Hz, 1H), 7.40 (m, 3H), 7.25 (d, J = 8.4 Hz, 2H), 7.20 (d, J = 8.0 Hz, 1H), 7.10 (d, J = 8.0 Hz, 2H), 6.95 (s, 1H), 6.93 (s, 1H), 6.86 (s, 1H), 3.84-3.74 (m, 2H), 3.69 (s, 3H), 2.27 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.8, 142.5, 140.3, 136.8, 136.5, 135.5, 129.9, 129.6, 129.2, 128.4, 127.5, 127.3, 125.2, 124.4, 124.0, 122.2, 118.8, 112.6, 111.6, 104.9, 53.7, 47.8, 32.9, 21.1; IR (KBr, ν, cm−1) 3531, 3050, 1522, 1404, 1242, 1121, 1017, 945, 836, 720; HRMS (APCI-TOF) m/z calcd for C28H21BrNO [M-H]− 466.0807; found 466.0815.
1-(1H-indol-3-yl)-1-(p-tolyl)-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3cc)
White solid, 88 mg, 78% yield; mp 156-158°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.86 (s, 1H), 10.20 (s, 1H), 8.26 (d, J = 8.4 Hz, 1H), 7.52 (d, J = 8.0 Hz, 1H), 7.38 (m, 3H), 7.27 (d, J = 8.4 Hz, 2H), 7.08 (d, J = 8.0 Hz, 2H), 7.02 (m, 1H), 6.96-6.88 (m, 3H), 6.81 (m, 1H), 3.82 (m, 2H), 2.25 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.6, 143.2, 140.3, 137.4, 137.4, 135.3, 130.0, 129.1, 127.4, 127.3, 126.6, 125.2, 124.4, 123.8, 123.7, 122.4, 122.0, 121.3, 120.0, 119.7, 118.8, 112.1, 105.0, 54.1, 47.6, 21.1; IR (KBr, ν, cm−1) 3501, 3408, 3051, 1520, 1402, 1240, 1118, 1010, 943, 816, 723; HRMS (APCI-TOF) m/z calcd for C27H20NO [M-H]− 374.1545; found 374.1529.
1-(6-methyl-1H-indol-3-yl)-1-phenyl-1,2-dihydrocyclobuta[a]naphthalen-4-ol (3dd)
White solid, 91 mg, 81% yield; mp 172-174°C; 1H NMR (400 MHz, DMSO-d6; δ, ppm) 10.72 (s, 1H), 10.18 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 7.52 (d, J = 8.0 Hz, 1H), 7.44-7.34 (m, 4H), 7.26 (m, 3H), 7.18 (m, 1H), 6.86 (d, J = 6.8 Hz, 3H), 6.79 (s, 1H), 3.83 (s, 2H), 2.22 (s, 3H); 13C NMR (100 MHz, DMSO-d6; δ, ppm) 154.6, 146.2, 140.4, 137.5, 135.7, 129.9, 128.5, 127.3, 127.1, 126.9, 126.3, 125.2, 124.4, 123.8, 123.0, 122.4, 119.6, 119.0, 111.9, 105.0, 54.2, 47.5, 21.9; IR (KBr, ν, cm−1) 3518, 3401, 3050, 1570, 1392, 1242, 1110, 1023, 941, 826, 721; HRMS (APCI-TOF) m/z calcd for C27H20NO [M-H]− 374.1545; found 374.1553.
X-Ray Structure of Product 3a (CCDC 1867087)
The crystal of compound 3a belongs to Triclinic, space group P-1 with a = 8.5599(7) Å, b = 12.1512(11) Å, c = 12.5112(12) Å, α = 100.943(2)°, β = 94.2510(10)°, γ = 106.823(3)°, V = 1211.40(19) Å3, Mr = 433.53, Z = 2, Dc = 1.743 g/cm3, μ(MoKα) = 0.074 mm−1, F(000) = 460, the final R = 0.0495 and wR = 0.1118.
Author Contributions
HL, BJ, and GL designed the project. HL performed the experiments. HL, W-JH, and S-JT analyzed the data. HL, BJ, and GL wrote the manuscript.
Funding
We are grateful for financial support from the NSFC (Nos. 21332005, 21472071 and 21871112), the Outstanding Youth Fund of JSNU (YQ2015003) and Robert A. Welch Foundation (D-1361, USA).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fchem.2018.00599/full#supplementary-material
References
Aidhen, I. S., and Ahuja, J. R. (1992). A novel synthesis of benzocyclobutenones. Tetrahedron Lett. 33, 5431–5432. doi: 10.1016/S0040-4039(00)79113-1
Anastas, P. T., and Warner, J. C. (1998). Green Chemistry: Theory and Practice. New York, NY: Oxford Science Publications.
Andersen, N. G., Maddaford, S. P., and Keay, B. A. (1996). Synthesis of functionalized naphthalenes from substituted 1-methoxybenzocyclobutenes. J. Org. Chem. 61, 2885–2887. doi: 10.1021/jo951978v
Banert, K., and Plefka, O. (2011). Synthesis with perfect atom economy: Generation of diazo ketones by 1,3-dipolar cycloaddition of nitrous oxide at cyclic alkynes under mild conditions. Angew. Chem. Int. Ed. 50, 6171–6174. doi: 10.1002/anie.201101326
Barluenga, J., Rodriguez, F., and Fananas, F. J. (2009). Recent advances in the synthesis of indole and quinoline derivatives through cascade reactions. Chem. Asian J. 4, 1036–1048. doi: 10.1002/asia.200900018
Beak, P., and Selling, G. W. (1989). Displacements at the nitrogen of lithioalkoxylamides by organometallic reagents. J. Org. Chem. 54, 5574–5580. doi: 10.1021/jo00284a034
Bihani, M., Bora, P. P., Bez, G., and Askari, H. (2013). Amberlyst A21 catalyzed chromatography-free method for multicomponent synthesis of dihydropyrano [2,3-c] pyrazoles in ethanol. ACS Sustainable Chem. Eng. 1, 440–447. doi: 10.1021/sc300173z
Bilyard, K. G., Garratt, P. J., Underwood, A. J., and Zahler, R. (1979). Vicinal diester dianions. Annelation with allylic and benzylic αω-dihalides. Synthesis of tricyclobuta[1,2:3,4:6,7]naphthalene. Tetrahedron Lett. 20, 1815–1818. doi: 10.1016/S0040-4039(01)86226-2
Birch, A. J., Brown, J. M., and Stansfield, F. (1964). Reactions of cyclohexadienes. IV. Some transformations of bisdihalocarbene adducts. J. Chem. Soc. 5343–5348. doi: 10.1039/jr9640005343
Bradsher, C. K., and Hunt, D. A. (1981). Novel synthesis of benzo[1,2:4,5]dicyclobutene via a dual Parham cyclialkylation. J. Org. Chem. 46, 4608–4610. doi: 10.1021/jo00335a072
Brauch, S., Van Berkel, S.-S., and Westermann, B. (2013). Higher-order multicomponent reactions: beyond four reactants. Chem. Soc. Rev. 42, 4948–4962. doi: 10.1039/c3cs35505e
Bruckmann, A., Krebs, A., and Bolm, C. (2008). Organocatalytic reactions. Effects of ball milling, microwave and ultrasound irradiation. Green Chem. 10, 1131–1141. doi: 10.1039/b812536h
Buchwald, S. L., Lucas, E. A., and Dewan, J. C. (1987a). Synthesis, structure, and reactions of a zirconocene-benzdiyne complex. J. Am. Chem, Soc. 109, 4396–4397. doi: 10.1021/ja00248a046
Buchwald, S. L., Watson, B. T., Lum, R. T., and Nugent, W. A. (1987b). A general method for the preparation of zirconocene complexes of substituted benzynes: in situ generation, coupling reactions, and use in the synthesis of polyfunctionalized aromatic compounds. J. Am. Chem. Soc. 109, 7137–7141. doi: 10.1021/ja00257a038
Buckland, S. J., Halton, B., Mei, Q., and Stang, P. J. (1987). Studies in the cycloproparene series: reactions of alkylidenecycloproparenes with electrophiles. Aust. J. Chem. 40, 1375–1387. doi: 10.1071/CH9871375
Bunnett, J. F., and Skorcz, J. A. (1962). Homocyclic ring closures via benzyne intermediates. A new synthesis of 1-substituted benzocyclobutenes. J. Org. Chem. 27, 3836–3843. doi: 10.1021/jo01058a019
Cava, M. P., and Napier, D. R. (1956). Benzocyclobutene and benzocyclobutadiene dimer. J. Am. Chem. Soc. 78:500. doi: 10.1021/ja01583a071
Chen, K., Liu, S., Wang, D., Hao, W.-J., Zhou, P., Tu, S.-J., et al. (2017). Silver/scandium-cocatalyzed bicyclization of β-Alkynyl ketones leading to benzo[c]xanthenes and naphtho[1,2-b]benzofurans. J. Org. Chem. 82, 11524–11530. doi: 10.1021/acs.joc.7b02134
Chou, C.-H., Wu, C.-C., and Chen, W.-K. (1995). Synthesis of Pyrido[b]cyclobuten-5-one and 1-azafulvenallene by flash vacuum pyrolysis of 3-chloroformyl-2-methylpyridine. Tetrahedron Lett. 36:5065. doi: 10.1016/0040-4039(95)00940-E
Christophe, S., Kuehm-Caubere, C., Renard, P., Pfeiffer, B., and Caubere, P. (1998). Design of new anticancer drugs. II. Easy arynic access to benzocyclobutacarbazoles, a new family of antitumor agents. Tetrahedron Lett. 39, 7284–7286. doi: 10.1016/S0040-4039(98)02186-8
Craig, D., Robson, M. J., and Shaw, S. J. (1998). Traceless linkers for solid-phase synthesis. Homo-and hetero-Diels-Alder reactions of o-quinodimethanes. Synlett 1998, 1381–1383. doi: 10.1055/s-1998-1968
D'Andrea, S. V., Freeman, J. P., and Szmuszkovicz, J. (1990). Diels-Alder cycloaddition reactions of isobenzofuran and o-quinodimethane with 1, 2-diheteroethylenes. J. Org. Chem. 55, 4356–4358. doi: 10.1021/jo00301a028
Dömling, A., Wang, W., and Wang, K. (2012). Chemistry and biology of multicomponent reactions. Chem. Rev. 112, 3083–3135. doi: 10.1021/cr100233r
Ezcurra, J. E., and Moore, H. W. (1993). Synthesis of o-quinodimethanes and benzocyclobutenes from dimethyl squarate. Tetrahedron Lett. 34, 6177–6180. doi: 10.1016/S0040-4039(00)73703-8
Fuerstner, A. (2009). Gold and platinum catalysis-a convenient tool for generating molecular complexity. Chem. Soc. Rev. 38, 3208–3221. doi: 10.1039/b816696j
Funk, R. L., and Vollhardt, K. P. C. (1977). A cobalt-catalyzed steroid synthesis. J. Am. Chem. Soc. 99, 5483–5484. doi: 10.1021/ja00458a044
Funk, R. L., and Vollhardt, K. P. C. (1979). The cobalt way to dl-estrone, a highly regiospecific functionalization of 2, 3-bis (trimethylsilyl) estratrien-17-one. J. Am. Chem. Soc. 101, 215–217. doi: 10.1021/ja00495a035
Funk, R. L., and Vollhardt, K. P. C. (1980). Cooligomerizations of 3-substituted 1, 5-hexadiynes with bis(trimethylsilyl)acetylene catalyzed by cobalt. A general synthesis of tricyclic ring systems from acyclic precursors. J. Am. Chem. Soc. 102, 5245–5253. doi: 10.1021/ja00536a022
Garratt, P. J., and Nicolaides, D. N. (1972). 3-Thiabicyclo[3.2.0]hepta-1, 4-diene. J. Chem. Soc. Chem. Commun. 1014–1015. doi: 10.1039/c39720001014
Garratt, P. J., and Nicolaides, D. N. (1974). Synthesis and some reactions of 3-thiabicyclo [3.2.0] hepta-1, 4-diene. Case for revival of the Mills-Nixon effect. J. Org. Chem. 39, 2222–2224. doi: 10.1021/jo00929a018
Gowland, B. D., and Durst, T. (1979). Benzocyclobutyl phenyl sulfone. An evaluation of its potential as a precursor to substituted benzocyclobutenes and ortho-quinodimethanes. Can. J. Chem. 57, 1462–1467. doi: 10.1139/v79-239
Gray, R., Harruff, L. G., Krymowski, J., Peterson, J., and Boekelheide, V. (1978). Syntheses of benzo[1,2:4,5]dicyclobutene derivatives. J. Am. Chem. Soc. 100, 2892–2893. doi: 10.1021/ja00477a054
Grieco, P. A., Takigawa, T., and Schillinger, W. J. (1980). Bicyclo[2.2.1]heptanes as intermediates in the synthesis of steroids. Total synthesis of estrone. J. Org. Chem. 45, 2247–2251. doi: 10.1021/jo01299a042
Haibach, M. C., Deb, I., De, C. K., and Seidel, D. (2011). Redox-neutral indole annulation cascades. J. Am. Chem. Soc. 133, 2100–2103. doi: 10.1021/ja110713k
Hickman, D. N., Wallace, T. W., and Wardleworth, J. M. (1991). The preparation of α-tetralones from benzocyclobutenones via sequential thermal electrocyclic reactions. Tetrahedron Lett. 32, 819–822. doi: 10.1016/S0040-4039(00)74896-9
Hoffmann, N., and Pete, J.-P. (1996). Acid catalyzed intramolecular photochemical reactions of 3-alkenyloxyphenols. Tetrahedron Lett. 37, 2027–2030. doi: 10.1016/0040-4039(96)00190-6
Huang, M.-H., Hao, W.-J., and Jiang, B. (2018a). Radical enabled bicyclization and annulation with 1,n-bifunctionalization: recent advances. Chem. Asian J. 13, 2958–2977. doi: 10.1002/asia.201801119
Huang, M.-H., Hao, W.-J., Li, G., Tu, S.-J., and Jiang, B. (2018b). Recent advances in radical transformations of internal alkynes. Chem. Commun. 54, 10791–10811. doi: 10.1039/C8CC04618B
Huang, M.-H., Zhu, Y.-L., Hao, W.-J., Wang, A.-F., Wang, D.-C., Liu, F., et al. (2017). Visible-light photocatalytic bicyclization of 1,7-enynes toward functionalized sulfone-containing benzo[a]fluoren-5-ones. Adv. Synth. Catal. 359, 2229–2234. doi: 10.1002/adsc.201700124
Inanaga, J., Sugimoto, Y., and Hanamoto, T. (1992). A novel method for the generation of 2, 3-naphthoquinodimethanes utilizing samarium (II) iodide-promoted allene synthesis. Tetrahedron Lett. 33, 7035–7038. doi: 10.1016/S0040-4039(00)60926-7
Iskander, G. M., and Stansfield, F. (1965). 4-Hydroxybenzocyclobuten-I-one and its derivatives. J. Chem. Soc. 1390–1392. doi: 10.1039/jr9650001390
Jia, S., Su, S., Li, C., Jia, X., and Li, J. (2014). Multicomponent cascade cycloaddition involving tropone, allenoate, and isocyanide: a rapid access to a 7, 6, 5-fused tricyclic skeleton. Org. Lett. 16, 5604–5607. doi: 10.1021/ol502656g
Jiang, B., Rajale, T., Wever, W., Tu, S.-J., and Li, G. (2010). Multicomponent reactions for the synthesis of heterocycles. Chem. Asian J. 5, 2318–2335. doi: 10.1002/asia.201000310
Jones, A. C., May, J. A., Sarpong, R., and Stoltz, B. M. (2010). Toward a symphony of reactivity: cascades involving catalysis and sigmatropic rearrangements. Angew. Chem. Int. Ed. 53, 2556–2591. doi: 10.1002/anie.201302572
Kagabu, S., and Saito, K. (1988). Dihalocarbene addition to benzocyclopropene. Tetrahedron Lett. 29, 675–676. doi: 10.1016/S0040-4039(00)80181-1
Kanao, Y., Iyoda, M., and Oda, M. (1983). The effect of cyclobutene ring annulation on p-benzoquinone: synthesis and properties of 1,2,3,4,5,6-hexahydrobenzo[1,2:4,5]dicyclobutene-3,6-dione([2,3:5,6]dicyclobuta-p-benzoquinone). Tetrahedron Lett. 24, 1727–1730. doi: 10.1016/S0040-4039(00)81755-4
Kaneko, C., and Naito, T. (1979). Intermolecular photochemical cycloaddition of 4-methoxy-2-quinolone with olefins: a regioselective synthesis of 5-substituted cyclobuta[c]-2-quinolones. Chem. Pharm. Bull. 27, 2254–2256. doi: 10.1248/cpb.27.2254
Kaneko, C., Naito, T., Momose, Y., Fujii, H., Nakayama, N., and Koizumi, I. (1982). Cycloadditions in syntheses. VIII. Synthesis of 1,2-dihydrocyclobuta[c]-pyridine and-quinoline and their 3-substituted derivatives. Chem. Pharm. Bull. 30, 519–525. doi: 10.1248/cpb.30.519
Koopmanschap, G., Ruijter, E., and Orru, R. V. A. (2014). Isocyanide-based multicomponent reactions towards cyclic constrained peptidomimetics. J. Org. Chem. 10, 544–598. doi: 10.3762/bjoc.10.50
Kotha, S., Goyal, D., and Chavan, A. S. (2013). Diversity-oriented approaches to unusual α-amino acids and peptides: step economy, atom economy, redox economy, and beyond. J. Org. Chem. 78, 12288–12313. doi: 10.1021/jo4020722
Krohn, K., Carboo, D., and Puttfarcker, U. (1978). 1-Phenyl-1,2-dihydrobenzazetes from N-phenylbenzylamines by aryne cyclization. Liebigs Ann. Chem. 1978, 608–616. doi: 10.1002/jlac.197819780411
Lenihan, B. D., and Shechter, H. (1994). Substituted o-[(Trimethylsilyl) methyl] benzyl p-tolyl sulfones: practical reagents for preparing cycloadducts of o-quinodimethanes. Tetrahedron Lett. 35, 7505–7508. doi: 10.1016/S0040-4039(00)78329-8
Lenihan, B. D., and Shechter, H. (1998). Chemistry of Conversions of [o-[1-Halo-1-(p-tolylsulfonyl)alkyl]benzyl]trimethylsilanes to o-Quinodimethanes and Benzocyclobutenes. J. Org. Chem. 63, 2086–2093. doi: 10.1021/jo9705935
Li, H., Hao, W.-J., Wang, M., Qin, X., Tu, S.-J., Zhou, P., et al. (2018a). Catalytic double [2 + 2] cycloaddition relay enabled C–C triple bond cleavage of Yne–Allenones. Org. Lett. 20, 4362–4366. doi: 10.1021/acs.orglett.8b01841
Li, H., Zhou, P., Xie, F., Hu, J.-Q., Yang, S.-Z., Wang, Y.-J., et al. (2018b). I2-Mediated iodobenzannulation of yne-allenones toward 1-naphthols and their synthetic application. J. Org. Chem. 83, 13335–13343. doi: 10.1021/acs.joc.8b02108
Li, X., Li, H., Song, W., Tseng, P.-S., Liu, L., Guzei, I. A., et al. (2015). Divergent reactivity of rhodium (I) carbenes derived from indole annulations. Angew. Chem. Int. Ed. 54, 12905–12908. doi: 10.1002/anie.201505329
Liu, C., Zhou, L., Huang, W., Wang, M., and Gu, Y. (2016). Synthesis of dihydrocarbazoles via (4 + 2) annulation of donor-acceptor cyclopropanes to indoles. Tetrahedron 72, 563–570. doi: 10.1016/j.tet.2015.12.023
Liu, F., Wang, J.-Y., Zhou, P., Li, G., Hao, W.-J., Tu, S.-J., et al. (2017a). Merging [2 + 2] cycloaddition with radical 1,4-addition: metal-free access to functionalized cyclobuta[a]naphthalen-4-ols. Angew. Chem. Int. Ed. 56, 15570–15574. doi: 10.1002/anie.201707615
Liu, S., Chen, K., Lan, X.-C., Hao, W.-J., Li, G., Tu, S.-J., et al. (2017b). Synergistic silver/scandium catalysis for divergent synthesis of skeletally diverse chromene derivatives. Chem. Commun. 53, 10692–10695. doi: 10.1039/C7CC05563C
Liu, S., Lan, X.-C., Chen, K., Hao, W.-J., Li, G., Tu, S.-J., et al. (2017c). Ag/Brønsted Acid Co-Catalyzed Spiroketalization of β-Alkynyl Ketones toward Spiro [chromane-2, 1′-isochromene] Derivatives. Org. Lett. 19, 3831–3834. doi: 10.1021/acs.orglett.7b01705
Markgraf, J. H., Katt, R. J., Scott, W. L., and Shefrin, R. N. (1969). Strained heterocyclic systems. IV. 1, 2-Dihydrocyclobuta [b] quinoline and derivatives. J. Org. Chem. 34, 4131–4133. doi: 10.1021/jo01264a081
Martins, M. A. P., Frizzo, C. P., Moreira, D. N., Buriol, L., and Machado, P. (2009). Solvent-free heterocyclic synthesis. Chem. Rev. 109, 4140–4182. doi: 10.1021/cr9001098
McNichols, A. T., and Stang, P. J. (1992). An improved synthesis of cyclopropa[4,5]benzocyclobutene. Synlett 1992, 971–972. doi: 10.1055/s-1992-21549
Mehta, G., and Kotha, S. (2001). Recent chemistry of benzocyclobutenes. Tetrahedron 57, 625–659. doi: 10.1016/S0040-4020(00)00958-3
Michellys, P.-Y., Maurin, P., Toupet, L., Pellissier, H., and Santelli, M. (2001). A very short synthesis of steroids from 1, 3-butadiene and benzocyclobutenes. J. Org. Chem. 66, 115–122. doi: 10.1021/jo001106f
Müller, P., Bernardinelli, G., Jacquier, Y., and Ricca, A. (1989). Tricarbonyl-chromium complexes of benzannelated cycloproparenes. Helv. Chim. Acta 72, 1618–1626. doi: 10.1002/hlca.19890720722
Neckers, D. C., and Wagenaar, F. L. (1981). Synthesis of 3, 4-benzo-2-thiabicyclo[3.2.0]hepta-1,3-diene. J. Org. Chem. 46, 3939–3940. doi: 10.1021/jo00332a046
Nemoto, H., Satoh, A., Fukumoto, K., and Kabuto, C. (1995). A rapid access to both enantiomers of 1,2,3,4-tetranor B-trienic 18,18,18-trifluorosteroids. The first enantiocontrolled total synthesis of 18,18,18-trifluorosteroids. J. Org. Chem. 60, 594–600. doi: 10.1021/jo00108a022
Ozaki, K., Matsuoka, W., Ito, H., and Itami, K. (2017). Annulative π-extension (APEX) of heteroarenes with dibenzosiloles and dibenzogermoles by palladium/o-chloranil catalysis. Org. Lett. 19, 1930–1933. doi: 10.1021/acs.orglett.7b00684
Parham, W. E., Jones, L. D., and Sayed, Y. A. (1976). Selective halogen-lithium exchange in bromophenylalkyl halides. J. Org. Chem. 41, 1184–1186. doi: 10.1021/jo00869a022
Peter, K., and Vollhardt, C. (1977). Transition-metal-catalyzed acetylene cyclizations in organic synthesis. Acc. Chem. Res. 10, 1–8. doi: 10.1021/ar50109a001
Peter, K., and Vollhardt, C. (1984). Cobalt-mediated [2 + 2 + 2]-cycloadditions: a maturing synthetic strategy [new synthetic methods (43)]. Angew. Chem. Int. Ed. 23, 539–556. doi: 10.1002/anie.198405393
Rotstein, B.-H., Zaretsky, S., Rai, V., and Yudin, A.-K. (2014). Small heterocycles in multicomponent reactions. Chem. Rev. 114, 8323–9359. doi: 10.1021/cr400615v
Sadana, A. K., Saini, R. K., and Billups, W. E. (2003). Cyclobutarenes and related compounds. Chem. Rev. 103, 1539–1602. doi: 10.1021/cr010022j
Sato, M., Suzuki, T., Morisawa, H., Fujita, S., Inukai, N., and Kaneko, C. (1987). Cycloadditions in syntheses. XXXV. 2-Cyano-1,2-dihydrocyclobuta[a]naphthalene and Its derivatives: synthesis and reaction with olefins. Chem. Pharm. Bull. 35, 3647–3657. doi: 10.1248/cpb.35.3647
Schirch, P. F. T., Boekelheide, V., and Cyclophane, M. (1979). J. Am. Chem. Soc. 101, 3125–3126. doi: 10.1021/ja00505a052
Sekine, Y., Brown, M., and Boekelheide, V. (1979). [2.2.2.2.2.2](1,2,3,4,5,6) Cyclophane: superphane. J. Am. Chem. Soc. 101, 3126–3127. doi: 10.1021/ja00505a053
Sha, H.-K., Liu, F., Lu, J., Liu, Z.-Q., Hao, W.-J., Tang, J.-L., et al. (2018a). Metal-free benzannulation of yne-allenone esters for atom-economic synthesis of functionalized 1-naphthols. Green Chem. 20, 3476–3485 doi: 10.1039/C8GC01034J
Sha, H.-K., Xu, T., Liu, F., Tang, B.-Z., Hao, W.-J., Tu, S.-J., et al. (2018b). Metal-free naphthannulation reactions of yne-allenone esters for accessing polycyclic aromatic hydrocarbons. Chem. Commun. 54, 10415–10418. doi: 10.1039/C8CC05018J
Shimada, S., Osoda, K., and Narasaka, K. (1993). Boron compound as a trapping reagent of α-hydroxy o-quinodimethanes in the diels–alder reaction. Bull. Chem. Soc. Jpn. 66, 1254–1257. doi: 10.1246/bcsj.66.1254
Su, S., Li, C., Jia, X., and Li, J. (2014). Isocyanide-based multicomponent reactions: concise synthesis of spirocyclic oxindoles with molecular complexity by using a [1,5]-hydrogen shift as the key step. Chem. Eur. J. 20, 5905–5909. doi: 10.1002/chem.201402576
Sugimoto, K., and Matsuya, Y. (2017). Recent applications of gold-catalyzed cascade reactions in total synthesis of natural product. Tetrahedron Lett. 58, 4420–4426. doi: 10.1016/j.tetlet.2017.10.029
Taber, D. F., Raman, K., and Gaul, M. D. (1987). Enantioselective ring construction: synthesis of (+)-estrone methyl ether. J. Org. Chem. 52, 28–34. doi: 10.1021/jo00377a005
Tian, Y., Tian, L., He, X., Li, C.-J., Jia, X., and Li, J. (2015). Indium (III) chloride-catalyzed isocyanide insertion reaction to construct complex spirooxindole. Org. Lett. 17, 4874–4877. doi: 10.1021/acs.orglett.5b02432
Tietze, L. F., Kinzel, T., and Brazel, C. C. (2009). The domino multicomponent allylation reaction for the stereoselective synthesis of homoallylic alcohols. Acc. Chem. Res. 42, 367–378. doi: 10.1021/ar800170y
Toda, F., Tanaka, K., Sano, I., and Isozaki, T. (1994). A new synthetic route to 1, 2-dihydrocyclobutaarenes. Angew. Chem. Int. Ed. 33, 1757–1758. doi: 10.1002/anie.199417571
Toda, M., Okada, K., and Oda, M. (1988). Synthesis of 3, 4; 7, 8; 9, 10-tribenzobicyclo [4.2.2] deca-1,3,7,9-tetraene: a new strained bridgehead olefin. Tetrahedron Lett. 29, 2329–2332. doi: 10.1016/S0040-4039(00)86051-7
Trost, B. M. (1995). Atom economy - a challenge for organic synthesis: homogeneous catalysis leads the way. Angew. Chem. Int. Ed. 34, 259–281. doi: 10.1002/anie.199502591
Trost, B. M. (2002). On inventing reactions for atom economy. Acc. Chem. Res. 35, 695–705 doi: 10.1021/ar010068z
Trost, B. M., Toste, F. D., and Greenman, K. (2003). Atom economy. palladium-catalyzed formation of coumarins by addition of phenols and alkynoates via a Net C-H insertion. J. Am. Chem. Soc. 125, 4518–4526. doi: 10.1021/ja0286573
Vlaar, T., Maes, B.-U., Ruijter, E., and Orru, R.-V. (2013). Palladium-catalyzed migratory insertion of isocyanides: an emerging platform in cross-coupling chemistry. Angew. Chem. Int. Ed. 52, 7084–7097. doi: 10.1002/anie.201300942
Wang, J.-Y., Xie, F.-L., Hu, J.-Q., Yang, S.-Z., Wang, Y.-J., Hao, W.-J., et al. (2018). Atom-economic synthesis of cyclobuta[a]naphthalen-4-ols via base-promoted [2 + 2] cycloaddition/1,6-nucleophilic addition cascades. Org. Biomol. Chem. 16, 7104–7108. doi: 10.1039/C8OB02042F
Wang, J.-Y., Zhou, P., Li, G., Hao, W.-J., Tu, S.-J., and Jiang, B. (2017). Synthesis of functionalized benzo[g]indoles and 1-naphthols via carbon-carbon triple bond breaking/rearranging. Org. Lett. 19, 6682–6685. doi: 10.1021/acs.orglett.7b03410
Wang, L., Shi, L.-X., Liu, L., Li, Z.-X., Xu, T., Hao, W.-J., et al. (2017). Synthesis of diastereoenriched oxazolo[5,4-b]indoles via catalyst-free multicomponent bicyclizations. J. Org. Chem. 82, 3605–3611. doi: 10.1021/acs.joc.7b00129
Wang, Y., Lu, H., and Xu, P.-F. (2015). Asymmetric catalytic cascade reactions for constructing diverse scaffolds and complex molecules. Acc. Chem. Res. 48, 1832–1844. doi: 10.1021/acs.accounts.5b00217
Warrener, R. N., Pitt, I. G., and Russell, R. A. (1993). Reaction of dienophiles with a 5, 6-bismethylenenorborn-2-en-7-one: the synthesis of a stable norborna-2, 5-dien-7-one. Aust. J. Chem. 46, 1009–1019. doi: 10.1071/CH9931009
Wei, H., Zhai, H., and Xu, P.-F. (2009). Novel platinum-catalyzed tandem reaction: an efficient approach to construct naphtho[1,2-b]furan. J. Org. Chem. 74, 2224–2226. doi: 10.1021/jo802645s
Keywords: Fe(III)-catalysis, bicyclization, 1,6-addition, yne-allenones, cyclobutarenes
Citation: Li H, Hao W-J, Li G, Tu S-J and Jiang B (2018) Fe(III)-Catalyzed Bicyclization of Yne-Allenones With Indoles for the Atom-Economic Synthesis of 3-Indolyl Cyclobutarenes. Front. Chem. 6:599. doi: 10.3389/fchem.2018.00599
Received: 12 September 2018; Accepted: 19 November 2018;
Published: 04 December 2018.
Edited by:
Oliver Reiser, University of Regensburg, GermanyReviewed by:
Federica Valentini, Università di Roma Tor Vergata, ItalyTill Opatz, Johannes Gutenberg University Mainz, Germany
Copyright © 2018 Li, Hao, Li, Tu and Jiang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guigen Li, Z3VpZ2VuLmxpQHR0dS5lZHU=
Bo Jiang, amlhbmdjaGVtQGpzbnUuZWR1LmNu
 Heng Li1
Heng Li1 Guigen Li
Guigen Li Bo Jiang
Bo Jiang