Abstract
Copper, a cofactor for many enzymes, is a bioelement that is involved in many main biochemical processes; although high levels of copper promote the proliferation of cancer cells. Further development of radiopharmaceuticals based on copper radioisotopes depend on understanding and taking advantage of its biochemical pathways in oncogenesis. As with other radiometals used in molecular imaging and/or targeted therapy, biological vectors are employed to transport copper radioisotopes to a target, aiming for high specific uptake at tumor sites and precise delivery of ionizing radiation. Evidence of the clinical utility of copper radioisotopes in the ionic form CuCl2 were also proven in an in vivo study of the copper metabolism, guiding personalized copper-chelating treatment in cancer patients and in imaging pathological sites associated with copper imbalance. Five of the copper radioisotopes have gained interest for nuclear medicine applications, based on their emissions, energies, and half-lives, as they can be produced with pharmaceutical-grade quality. The uptake mechanism, kinetics, and metabolic parameters are important findings in molecular imaging, which are decisive when designing individualized targeted radiotherapy for dose calculations of high linear energy transfer Auger electrons and β− emissions of 64Cu and 67Cu. As radiation deposits a high amount of energy within the intra-cellular space, the biochemical involvement of copper determines targets in drug design and validation. The biochemical pathways depict copper metabolism in normal cells and highlight its increased activity in tumor progression and angiogenesis. The avid uptake of copper into inter- and intra-mitochondrial spaces, as constituents of cytochrome C oxidase, substantiate the selection of 64/67CuCl2 as theranostic agents.
Introduction
The natural occurrence of copper (69.17% 63Cu, 30.83% 65Cu [1]), either in metallic form or as a mineral, confers a wide exposure to humans. Its inorganic salts are highly toxic but its varied coordination complexes have gained a lot of interest in drug design, as they are selective and also exhibit convenient pharmacokinetics and pharmacodynamics.
Copper is an essential microelement involved in important biochemical processes, such as: homeostasis, iron transport, respiration, and metabolism, as a result of its redox abilities in the biological environment: reversible translation between oxidized form (cupric ion, Cu2+) and the reduced form (cuprous ion, Cu+). It is a transition metal with 29 isotopes, out of which 27 are radioactive [2].
Along with the progress of nuclear medicine practices and technology, approaching molecular imaging and personalized treatment, five of the copper radioisotopes have gained interest for medical applications, considering their emissions, energies, production route, and availability, with half-lives ranging from 9.7 min (62Cu) to 2.6 days (67Cu) [2, 3]. Especially, 64Cu and 67Cu were intensively investigated as medically emergent radioisotopes for theranostic applications and therapy, respectively. Still, there is a need for more data regarding the production of 60Cu, 62Cu, and particularly 67Cu in medical small cyclotrons.
Recent studies demonstrated the usefulness of 64/67Cu agents, containing biological vectors to carry radioisotopes to target, aiming for high specific uptake at tumor sites, and precise delivery of ionizing radiation, such as peptides, antibodies, or other biologically active small molecules [2, 3]. Besides using such carriers, the clinical utility of copper radioisotopes in their most simple chemical form, copper chloride, was also proven, either for an in vivo study into the copper metabolism, guiding personalized copper-chelating treatment in cancer patients, or to image pathological sites associated with copper imbalance in inflammation, tumor angiogenesis, and metastasis.
As many of these findings are evidence-based and sourced directly from clinical practice (e.g., the significantly higher copper levels measured in serum and tumor cells of patients with cancer compared to normal subjects [4]), there is a need for an in-depth biological evaluation of the involved mechanisms and quantification. Therefore, we reviewed the relevant literature regarding the biological and biochemical pathways of copper, to substantiate the use of copper radioisotopes in oncology and promoting its further development.
Biological Pathways of Copper in Humans
Copper Bioavailability and Dietary Interactions
Humans are exposed to environmental copper from water, food, and tools or household goods, therefore the World Health Organization (WHO) defined a safe range for copper intake and acknowledged its effects, either positive or negative, on human health [1]. In an adult organism there is approximately 1.5 copper mg/kg bw, still up to 2.2 mg/kg bw is considered acceptable in the physiological range. Foods most abundant in copper are seafood, dry nuts and seeds, dark chocolate, and mushrooms [5]. A high nutritional intake does not represent any risk considering copper toxicity, as the human organism has a dynamic mechanism of homeostasis.
Copper bioavailability is fairly affected by dietary factors, such as carbohydrate, iron, zinc, molybdenum, and ascorbic acid co-ingestion. Large quantities of dietary zinc can decrease copper absorption and induce the symptoms of systemic copper deficiency. Also, an increased molybdenum intake drives the organism toward secondary copper deficiency, which can be rapidly corrected by copper supplementation. On the other hand, iron-copper interactions in the intestines conduct the regulation of copper transport modulation by the iron levels. Reduced levels of copper lead to a series of physiological changes, inducing pathological conditions, while high intake of copper, found as chronic or acute exposure, can result in liver damage [1].
Copper Metabolism and Physiological Role
The intestines are the main absorption site, the process being conducted by the enterocytes, with the participation of copper permease and human copper transporter-1 (hCTR1) [1–7]. Dietary Cu2+ is reduced to Cu+ by reductases, prior to being transported through the brush border membrane of the enterocytes by hCTR1 [1, 5, 8], yet the mechanisms for selective permeation of Cu+ ions across cell membranes are unknown [9]. After absorption, copper, bound to metallochaperone proteins, is delivered to the mitochondrion [10, 11] by the SLC25A3 inner membrane transporter [11, 12], which is required for the metalation of enzymes within the mitochondrial inter-membrane space [13]. The exceeding amount of copper can be deposited in an inert form in metallothionein, the main intracellular copper storage protein. Subsequently, it is released under the influence of ATP7A. At the end of the process, Cu+ is effluxed from enterocytes, chemically reconverted to Cu2+, and is thus able to bind to the transport proteins, albumin and alpha-2-macroglobulin. The carrier proteins deliver copper to the hepatic tissue, from where it is subsequently redirected to the target sites; therefore liver is the main organ that controls copper homeostasis mechanisms [1, 5]. Copper is distributed mostly in the bone and muscle tissues (up to 67%), but also in the liver, brain, and heart [1, 8].
Copper is further transferred to the cytoplasm, in inter-mitochondrial and intra-mitochondrial spaces, where it becomes a constituent of cytochrome c oxidase (CcO) and superoxide dismutase-1 (SOD1) [10–12]. Under normal circumstances, copper is transferred into the trans-Golgi network, where it is used for the synthesis of other cuproenzymes (ceruloplasmin, lysis oxidase, peptidylglycine alpha-amidating monooxygenase, and dopamine beta-hydroxylase) [1]. In the case of high intracellular copper influx, the same transporters will move to the cell surface, where they will mediate the efflux of excessed copper to the plasma (ATP7A) or bile (ATP7B) [1, 8]. Copper excretion is mainly achieved through bile, in the form of bile salts; the urinary excretion is rather insignificant [1, 8]. The ubiquitarian role of copper derives from its structural importance in a wide array of functional and modulatory proteins that are deeply involved in physiological and pathological mechanisms [13–15] (Supplementary Table S1). Copper is an enzymatic cofactor, an essential component of Cu-dependent enzymes: ceruloplasmin, cytochrome C oxidase, metallothionein, Cu/Zn superoxide dismutase-1, amine oxidases, lysyl oxidase, tyrosinase, zyklopten, and mono-oxygenases, and also represents an up-regulating trigger for a series of redox status modulatory enzymes: catalase, glutathione peroxidase, hepaestin, cartilage matrix glycoprotein, and Protein-6-lysine oxidase [1, 13]. Reduced activity of these enzymatic proteins is found in copper deficiency states [13, 16].
Copper Deficiency and Pathological Implications
Reduced or minimal activity of copper-dependent enzymes results in symptoms that may include hypochromic anemia, neutropenia, thrombocytopenia, and hypopigmentation, bone, cardiovascular, and neurological abnormalities, as well as immune system depression [1, 8]. Copper-related genetic diseases include Menkes syndrome (a mutation of ATP7A gene) which is expressed by reduced intestinal copper absorption and Wilson’s disease (a mutation of the ATP7B gene), when copper accumulates in excess in different organs (liver, brain, cornea) [1, 5, 8]. Wilson’s disease is caused by the cerebral and hepatic tissue accumulation of copper, leading to neurologic and psychiatric symptoms, and liver impairment [16, 17].
Children can develop potentially fatal idiopathic copper toxicosis when drinking contaminated water or food [8, 18, 19]. Correlations with Alzheimer’s disease have also been observed; elevated levels of free (unbound) copper in the blood were present, as well as high copper levels in amyloid senile plaque deposits [18, 20]. Diabetic patients exhibit elevated plasma copper levels [19, 21]. Copper deficiency is also associated with cardiovascular diseases [22].
The proliferation of cancer cells is promoted by high levels of copper [8, 23]. Elevated copper levels were found in different types of tumors while cancer growth was minimized when copper was chelated [5, 24]. Considering its redox properties, copper is a source for reactive oxygen species [1].
Medical Radioisotopes of Copper
Molecular Imaging, Targeted Therapy, and Theranostic Role of Radio-Copper
Molecular imaging allows for the quantification of functional parameters of an organ or process; moreover the interactions of a drug with its desired target can be analyzed, side effects can be determined, and the delivery, absorption, distribution, metabolism, and elimination in a living system can be precisely evaluated [25–27]. Among the molecular imaging techniques, positron emission tomography (PET) is most often used to tailor and deliver personalized treatment, as a result of receptor identification and mapping their density to a tissue or organ of interest, or by exploiting the imbalanced metabolism in different stages of pathological processes.
The positron-emitting radionuclide is customarily selected taking into account several factors, such as: the half-life of the radionuclide (this should match with the vector pharmacokinetics to allow optimal uptake), the energy of the positron emission (which determines the precision and image resolution), and the availability and cost of the production. Moreover, the specific/molar activity and carrier-free specifications, as quality parameters, become tremendously important when associated with molecular term (either imaging or therapy), together with radiobiological parameters, mainly the affinity, uptake, and retention profiles (radio)toxicity, blood clearance, and elimination route.
FIGURE 1
Five radioisotopes of copper (Table 1) can be produced at a cyclotron, with characteristics required for clinical use [28–31]. Based on their radioisotope emissions, 60Cu, 61Cu, 62Cu, and 64Cu are suitable for molecular imaging applications, while 64Cu and 67Cu are selected for targeted radionuclide therapy [30–32]. Due to their short half-lives, they are used in ionic form (as chlorides) or in combination with fast kinetic peptides. While the radiopharmaceuticals based on longer-lived radionuclides, such as 177Lu, 89Zr, or 90Y enable the investigation of the biological processes over a number of hours, which is often demanded by the study or imposed by slow kinetics of the vector [33–35], copper-64 is a theranostic radionuclide of particular interest due to its simultaneous emission of both β+ (17.52%) and β− (38.48%) particles [36–44]. The positron emission allows for high resolution PET imaging, while low abundance gamma emissions do not affect the imaging process compared to other positron emitters [30, 45]. It decays also through electron capture (EC 43.53%), when high linear energy transfer Auger electrons are emitted. When this happens in the close vicinity of a cancerous cell nucleus, it may cause DNA damage, eventually triggering cell death and thus, achieving a therapeutic effect. Taking advantage of the positron emissions, real-time therapy follow-up can be performed by PET imaging, presumably at any time point during therapy.
TABLE 1
| Radioisotope and half-life | Decay mode and energy | Most intense γ emissions | Nuclear reaction and cross-section data [74] |
|---|---|---|---|
| 67Cu 61.8 h | β− (100%) 121 keV (57%) 154 keV (22%) 189 keV (20%) | 91.2 keV (7%) 93.3 keV (16.1%) 184.6 keV (48.7%) | 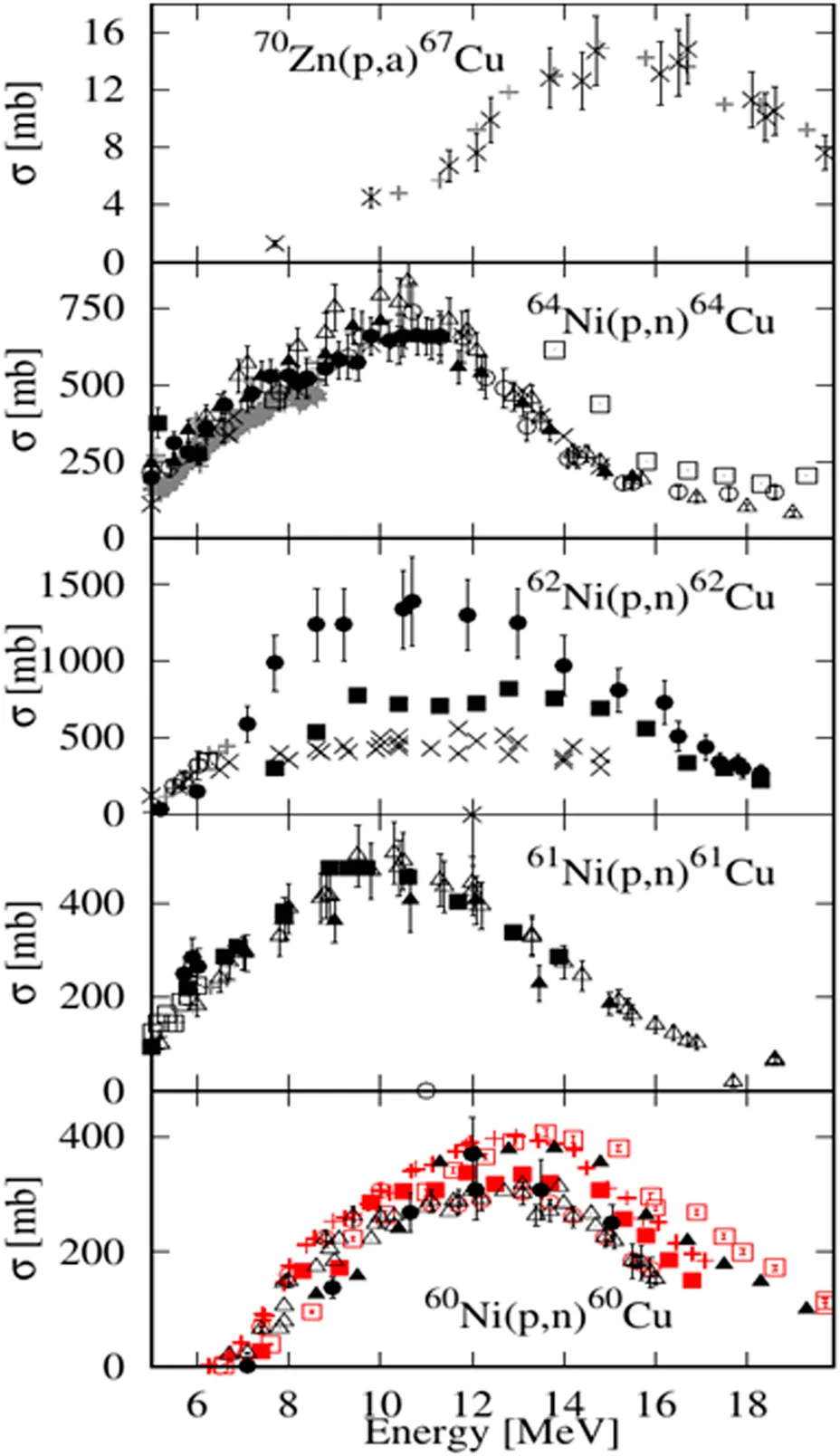 |
| 64Cu 12.7 h | β− (38.5%) 191 keV (38.5%) | - | |
| EC and β+ (61.5%) 278.2 keV (17.6%) | 1,345.77 keV (0.475%) | ||
| 62Cu 9.7 min | EC and β+ (100%) 1,321 keV (98) | 875.7 keV (0.15%) 1,173 keV (0.342%) | |
| 61Cu 3.32 h | EC and β+ (100%) 524 keV (51%) | 282.9 keV (12.2%) 656.0 keV (10.8%) 1,185.2 keV (3.7%) | |
| 60Cu 23.7 min | EC and β+ (100%) 872 keV (49%) | 1,332.4 keV (88%) 1791.6 keV (45.4%) |
Radioisotopes of copper produced in medium energy cyclotrons.
Production of Medical Radioisotopes of Copper, with Particular Interest on 64Cu
Researchers are investigating different routes to produce carrier-free and high specific activity copper radioisotopes [29]. Copper-64 can be produced in a reactor by (n,γ) and (n,p) reactions, on enriched targets [30], at thermal neutron fluxes (6-7⋅1012 n*cm−2*s−1). The average specific activity of 64Cu obtained was 2.4 TBq/g Cu, at the end of irradiation. Using this route, radionuclide impurities 65Zn and 60Co are co-produced and should be eliminated by radiochemical processing, using an anion exchange separator [31]. Higher specific activity can be achieved when fast neutron reactions are employed, but thermal neutron reactions also occur, leading to high amounts of long-lived radionuclide impurities, such as 65Zn (T1/2 = 245 days) [29].
The most common way to produce 64Cu is by using a small/medium energy cyclotron [32, 36–49]. Several nuclear reactions can be triggered on nickel or zinc targets by proton beams: 64Ni(p,n)64Cu, natZn(p,xn)64Cu or 68Zn(p,αn) [49–57], but also deuterons induced reactions: 64Ni(d,2n), 66Zn(d,α), 64Zn(d,2p) [52–54, 56]. Good yields of 64Cu production, with low radionuclide impurities, were obtained using enriched 64Ni or 64/66/68Zn targets [49]. The irradiation of natZn targets is a less expensive method to conveniently obtain lower activities of 64Cu, while the use of deuteron beams on these targets requires energies above 20 MeV to obtain reasonable yields [56].
The 64Ni(p,n)64Cu reaction is used at large scale for the production of 64Cu, although bearing the disadvantage of costly target material, this route is preferred for the high yields that can be achieved, even at small medical cyclotrons [54, 55, 58]. Using a 12 MeV cyclotron, specific activity of >87 × 104 GBq/g and an irradiation yield of >111 MBq/µAh were reported [42]. 64Ni (99.5% enrichment) is electrodeposited from the 64Ni(NO3)2 solution, resulting in a64Ni solid target [46]. Alternatively, liquid targets consisting of solutions of 64Ni(NO3)2 are conveniently used, with lower production yield [47]. The irradiation process parameters are tuned for best yields, according to the experimental set-up and needs of a site; optimal parameters: 2–4 h irradiation time, 40–50 mg 64Ni on target, lead to 9.99–18.5 GBq of 64Cu and high specific activity (11.47 × 106 GBq/g Cu). The production yield, on 15–55 mg of enriched 64Ni targets, ranges from 82.9 to 185 MBq/μAh [48]. Separation of Cu from the Ni targets employs ion-exchange chromatography, using a cation exchanger column (AG1-X8). Enriched 64Ni can be recovered up to 95% [42]. During proton irradiation of enriched 64Ni, 61Co is produced as a contaminant, which can be separated with 4M HCl as an eluent [49].
64CuCl2 as Radiopharmaceutical and/or Precursor
64CuCl2 is used either as a radiopharmaceutical or as a precursor for radiolabeling specific carriers, such as monoclonal antibodies, peptides, amino acids, hormones, nanoparticles, or small molecules, using chelating agents [58–60]. This is also the case for all the other copper radionuclides. Various cold copper complexes were studied and also used as anticancer agents [60].
After IV administration, 64CuCl2 accumulates in the liver (uptake fraction 0.65), brain (uptake fraction 0.1), kidney (uptake fraction 0.01), and pancreas (uptake fraction 0.0002). Based on preclinical studies, the calculated effective dose (ED) is 70 mSv for the whole body of a 70 kg adult, after the intravenous injection of 925 MBq of 64CuCl2 [61].
The chelators used for binding radio-copper to biomolecules (Figure 1) should have high thermodynamic stability; compact structures of macrocyclic or macro-bicyclic ligands with increased kinetic stability are preferred [62–71]. When dissociated from the complexes, Cu2+ is reduced to Cu+ and binds to SOD in high concentrations [63]. DOTA have been used for chelating 64Cu, however, its ability to bind many different metal ions, trans-chelation to liver proteins, and its decreased stability compared to TETA/CB-TE2A make it less attractive [64, 65]. By comparison, NOTA and the hexaamino sarcophagine ligands demonstrate ease of conjugation, high radiolabeling yields, and in vivo stability [58, 66]. They also achieve better clearance from the blood, liver, and kidneys [65, 66]. The kinetic stability of copper (II) cross-bridged cyclam complexes is superior to those of the TETA and DOTA complexes [63], while 64Cu-CB-TE2A proved to be the most stable, when compared to CB-cyclam, CB-DO2A, DOTA, and TETA, respectively [62].
Comparing the biodistribution, at 24 h p.i., of 64Cu-CB-DO2A, 64Cu-CB-TE2A, 64Cu-DOTA, and 64Cu-TETA, a larger amount of 64Cu-labeled cross-bridged chelates was cleared form the blood, liver, and kidney than the non cross-bridged analogues; moreover, 64Cu-CB-TE2A was the most resistant to trans-chelation in rat liver [65]. Hexaaza macrobicyclic sarcophagines (Sar) are very compact structures, acting like a “cage” around Cu2+, which increases the thermodynamic and kinetic stability, leading to low accumulation at non-targeted tissues. Evaluating the biodistribution data of 64Cu-Sar, 64Cu-diamSar, and 64Cu-SarAr in balb/c mice, it was found that all three complexes had been cleared from the blood rapidly, while the uptake was low in bone, heart, stomach, spleen, muscle, lungs, and the gastrointestinal tract [66].
64Cu as Radioisotope Contained in Theranostic Agents Intended for Different Tumors
64CuCl2 shows an increased and specific uptake in melanoma expressing high hCTR1: 12.7% ± 0.26 in B16F10 cells and 4.6% ± 0.04 in A375M cells, the tumor-to-muscle ratio was 4.11 ± 0.07 for B16F10 and 3.46 ± 1.25 for A375M. During 64CuCl2 treatment, tumor growth in both melanoma models was slower than without treatment, suggesting that 64CuCl2 radiotherapy is effective for hCTR1 high-expressing tumors [67].
In a xenograft model of glioblastoma multiforme (GBM) U87MG, the biodistribution of 64CuCl2 indicated no brain uptake, while PET images showed an uptake in glioma cells; a decrease of the tumor volume with more than 68% was noticed, raising the survival rate of the treated mice [68]. SI113 inhibits SGK1, a protein with increased the expression of glioblastoma. The combination of SI113 and 64CuCl2 has a synergistic effect and affects cell viability, triggering apoptosis, and necrosis. The inhibitory dose, tested in three cell lines in glioblastoma ((LI)PARI, ADF, and T98G) with different mutational status for p53, was 40 MBq [68].
In a study using the hypoxia-selective agent 64Cu-ATSM on hamsters implanted with GW39 (human colorectal carcinoma), the inhibition of tumor growth was observed for a 220 MBq injected dose; the animals presented an increased rate of survival with no acute toxicity. After administration, PET scans revealed that 64Cu-ATSM was localized in the GW39 tumor and PET imaging could be performed regularly [69].
Administration of 555 MBq of 64Cu-TETA-Y3-TATE in a single dose to CA20948 rats, a model of somatostatin receptor-positive pancreatic cancer, decreased the tumor volume (29–73%) and inhibited its growth. The multiple dose radiotherapy study (3 × 370 MBq) decreased the tumor volume (36–81%) and provided a tolerable radiation exposure level over an extended period [70].
64Cu-ATSM (64Cu-diacetyl-bis(N-4-methylthiosemicarbazone) showed a high cytotoxic effect, decreasing the clonogenic survival of LL/2 cells (Mouse Lewis Lung carcinoma cells) in a dose dependent manner; the uptake of 1.50 Bq/cell of 64Cu killed 99% of the cells. Under hypoxic conditions, 64Cu was accumulated in the cells and produced DNA damage, detected by comet assay and Annexin V-FITC and propidium iodide staining methods [71].
DU-145 human prostate cancer xenografts were visualized by PET using 64CuCl2, the cellular uptake was mediated by hCTR1, demonstrated by negative control PC-3 prostate cancer cells. Knockdown of hCTRl reflected the decreased cellular uptake and inhibition of tumor growth [72]. After 64CuCl2 administration, a rapid uptake in the PCa lesions reached the maximum value in 1 h [73].
Conclusions
The biochemical pathways show copper metabolism in normal cells and highlight its increased activity in human cancer cells, at a higher metabolic rate. Its involvement in tumor progression and angiogenesis and its pivotal role in preserving the intracellular homeostasis are particular indicators used in functional imaging. Thus, specific processes are targeted by radio-copper chloride, but also specific vectors radiolabeled with copper radioisotopes are used. Moreover, the copper presence in intermitochondrial and intramitochondrial spaces, as constituents of cytochrome c oxidase, substantiates the selection of 64Cu, a short range high LET emitter (Auger electrons), as a therapeutic agent, in a bioavailable chemical form, 64CuCl2.
The uptake mechanism, kinetics, and metabolic parameters are very important findings for PET imaging using 60Cu, 61Cu, 62Cu, or 64Cu which are decisive when designing an individualized targeted therapy and, also, for dose calculations of high LET Auger electrons and β− emissions of 64Cu and 67Cu. The concept of theranostic applications applies perfectly to copper radioisotopes, by matching pairs for diagnostics and therapy (e.g., 61Cu and 67Cu) or by taking advantage of the dual emissions of 64Cu for both purposes. In this latter case, a real-time therapy follow-up brings important benefits for patients.
Funding
The work has been funded by the UEFISCDI, Romanian Ministry of Education and Research, under contract 64PCCDI/2017. The work of Ramona Dusman, PhD student, has been funded by the Operational Program Human Capital of the Ministry of European Funds through the Financial Agreement 51668/09.07.2019, SMIS code 124705.
Statements
Author contributions
DN, RD, and RL reviewed the data regarding copper radioisotopes production and radiochemistry and edited the article. LC, RS, and DAN reviewed the data regarding biological assessment of 64Cu-labelled biomolecules, CD, AN, ID, and DD reviewed pharmaceutical and pharmacological data.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphy.2020.568296/full#supplementary-material.
References
1.
BertinatoJCaballeroBFinglasPMToldráF.“Copper: physiology” in: reference module in food science, encyclopedia of food and health: Waltham: Academic Press (2016) p. 321–6.
2.
AhmedovaATodorovBBurdzhievNGozeC.Copper radiopharmaceuticals for theranostic applications. Eur J Med Chem (2018) 157:1406–25. 10.1016/j.ejmech.2018.08.051
3.
WadasTJWongEHWeismanGRAndersonCJ.Copper chelation chemistry and its role in copper radiopharmaceuticals. Curr Pharmaceut Des (2007) 13(1):3–16. 10.2174/138161207779313768
4.
EvangelistaLMansiLCasciniGL.New issues for copper-64: from precursor to innovative pet tracers in clinical oncology. Curr. Rad (2013) 6(3):117–23. 10.2174/18744710113069990020
5.
LatorreMTroncosoRUauyR.Biological aspects of copper. In Clinical and translational perspectives on WILSON DISEASE: Waltham: Academic Press (2019) p. 25–31.
6.
MaryonEBMolloySAIvyKYuHKaplanJH.Rate and regulation of copper transport by human copper transporter 1 (hCTR1). J. Biol. Chem (2013) 288(25):18035–46. 10.1074/jbc.M112.442426
7.
SpreckelmeyerSvan der ZeeMBertrandBBodioESturupSCasiniA.Relevance of copper and organic cation transporters in the activity and transport mechanisms of an anticancer cyclometallated gold (III) compound in comparison to cisplatin. Front. Chem (2018) 6:377. 10.3389/fchem.2018.00377
8.
HordyjewskaAPopiołekŁKocotJ.The many “faces” of copper in medicine and treatment. Biometals (2014) 27:611–21. 10.1007/s10534-014-9736-5
9.
RenFLogemanBLZhangXLiuYThieleDJYuanP.X-ray structures of the high-affinity copper transporter Ctr1. Nat. Commun (2019) 10:1086. 10.1038/s41467-019-09376-7
10.
BakerZNCobinePALearySC.The mitochondrion: a central architect of copper homeostasis. Metallomics (2017) 9(11):1501–12. 10.1039/c7mt00221a
11.
WingeDR.Filling the mitochondrial copper pool. J. Biol. Chem (2018) 293:1897–8. 10.1074/jbc.H118.001457
12.
BouletAVestKEMaynardMKGammonMGRussellACMathewsATet alCopper trafficking to the mitochondrion ansd assembly of copper metalloenzymes. Biochim. Biophys. Acta (2006) 1763(7):759–72. 10.1016/j.bbamcr.2006.03.002
13.
TapieroHTownsendDMTewKD.Trace elements in human physiology and pathology. copper. Biomed. Pharmacother (2003) 57(9):386–98. 10.1016/S0753-3322(03)00012-X
14.
Martínez-GonzálezJVaronaSCañesLGalánMBrionesAMCachofeiroVet alEmerging roles of lysyl oxidases in the cardiovascular system: new concepts and therapeutic challenges. Biomolecules (2019) 9(10):610. 10.3390/biom9100610
15.
ChenHAttiehZKSyedBAKuoYMStevensYFuquaBKet alIdentification of zyklopen, a new member of the vertebrate multicopper ferroxidase family and characterization in rodents and human cells. J. Nutr (2010) 140(10):1728–35. 10.3945/jn.109.117531
16.
BorchardSBorkFRiederTEberhagenCPopperBLichtmanneggerJ.The exceptional sensitivity of brain mitochondria to copper. Toxicol. In Vitro (2018) 51:11–22. 10.1016/j.tiv.2018.04.012
17.
DrăgoiCMMoroşanEDumitrescuIBNicolaeACArseneALDrăgănescuDet alInsights into chrononutrition: the innermost interplay amongst nutrition, metabolism and the circadian clock, in the context of epigenetic reprogramming. Farmacia (2019) 67(4):557–71. 10.31925/farmacia.2019.4.2
18.
BanciLBertiniICiofi-BaffoniSKozyrevaTZovoKPalumaaP.Affinity gradients drive copper to cellular destinations. Nature (2010) 465(7298):645–8. 10.1038/nature09018
19.
BlockhuysSCelauroEHildesjoCFeiziAStalOFierro-GonzalezJC.Defining the human copper proteome and analysis of its expression variation in cancers. Metallomics (2017) 9(2):112–23. 10.1039/c6mt00202a
20.
MursaJRobienKHamackLJParkKJacobsDR.Dietary supplements and mortality rate in older women: the iowa women’s health study. Arch. Intern. Med (2011) 171(18):1625–33. 10.1001/archinternmed.2011.445
21.
DrăgoiCMNicolaeACDumitrescuIBPopaDERitivoiuMArseneAL.DNA targeting as a molecular mechanism underlying endogenous indoles biological effects. Farmacia (2019) 67(2):367–77. 10.31925/farmacia.2019.2.24
22.
KlevayLM.Is the western diet adequate in copper?. J. Trace Elem. Med. Biol (2011) 25(4):204–12. 10.1016/j.jtemb.2011.08.146
23.
MorrisMCEvansDATangneyCCBieniasJLSchneiderJAWilsonRSet alDietary copper and high saturated and trans fat intakes associated with cognitive decline. Arch. Neurol (2006) 63(8):1085–8. 10.1001/archneur.63.8.1085
24.
BarrosMHJohnsonATzagoloffA.COX23, a homologue of COX17, is required for cytochrome oxidase assembly. J. Biol. Chem (2004) 279:31943–7. 10.1074/jbc.M405014200
25.
WadasJWongEHWeismanGRAndersonCJ.Copper chelation chemistry and its role in copper radiopharmaceuticals. Curr. Pharm. Des (2007) 13(1):3–16. 10.2174/138161207779313768
26.
RowlandDJLewisJSWelchMJ.Molecular imaging: the application of small animal positron emission tomography. J. Cell Biochem. Suppl (2002) 39:110–5. 10.1002/jcb.10417
27.
KnowlesRFriedrichFFischerCPaechDLaddME.Beyond T2 and 3T: new MRI techniques for clinicians. Clin. Transl. Radiat. Oncol (2019) 18:87–97. 10.1016/j.ctro.2019.04.009
28.
CoenenHHGeeADAdamMAntoniGCutlerCSFujibayashiYet alConsensus nomenclature rules for radiopharmaceutical chemistry–setting the record straight. Nucl. Med. Biol (2017) 55:v–xi. 10.1016/j.nucmedbio.2017.09.004
29.
SunXAndersonCJ.Production and applications of copper-64 radiopharmaceuticals. Methods Enzymol (2004) 386:237–61. 10.1016/S0076-6879(04)86011-7
30.
Manual for reactor produced radioisotopes. Viena: IAEA – International Atomic Energy Agency (2003) 257 p.
31.
VimalnathKVRajeswariAChirayilVSharadPLJagadeesanKCJoshiPVet alStudies on preparation of 64Cu using (n,γ) route of reactor production using medium ux research reactor in india. J. Radioanal. Nucl. Chem (2011) 290:221–5. 10.1007/s10967-011-1301-x
32.
KastleinerSCoenenHHQaimSM.Possibility of production of 67Cu at a small-sized cyclotron via the (p,α)-Reaction on enriched 70Zn. Radiochim. Acta (1999) 84:107–10. 10.1524/ract.1999.84.2.107
33.
NiculaeDLunguVAnghelRGruiaIIliescuM.Labelling of anti-epidermal growth factor monoclonal antibody with 177Lu: radiochemical and biological evaluation. J. Labelled Cmpds. Radiopharmaceuticals (2010) 53:355–9. 10.1002/jlcr.1771
34.
JauwYWSMenke-van der Houven van OordtCWHoekstraOSHendrikseNHVugtsDJZijlstraJMet alImmuno-positron emission tomography with zirconium-89-labeled monoclonal antibodies in oncology: what can we learn from initial clinical trials?. Front. Pharmacol (2016) 7:131. 10.3389/fphar.2016.00131
35.
SgourosGBodeiLMcDevittMR.Radiopharmaceutical therapy in cancer: clinical advances and challenges. Nat. Rev. Drug Discov (2020) 19:589–608. 10.1038/s41573-020-0073-9
36.
SzelecsenyiFBlessingGQaimSM.Excitation functions of proton induced nuclear reaction on enriched Ni-61 and Ni-64: possibility of production of no-carrier-added Cu-61 and Cu-64 at a small cyclotron. Appl. Radiat. Isot (1993) 44:575–80. 10.1016/0969-8043(93)90172-7
37.
Adam RebelesRVan den WinkelPHermanneATarkanyiF.New measurement and evaluation of the excitation function of 64Ni(p,n) reaction for the production of 64Cu. Nucl. Instrum. Methods Phys. Res. B (2009) 267:457–61. 10.1016/j.nimb.2008.11.038
38.
UddinMChakrabortyASpellerbergSShariffMDasSRashidMet alExperimental determination of proton induced reaction cross sections on natNi near threshold energy. Radiochim. Acta (2016) 104(5):305–14. 10.1515/ract-2015-2527
39.
PielHQaimSMStöcklinG.Excitation functions of (p,xn)-Reactions on natNi and highly enriched 62Ni: possibility of production of medically important radioisotope 62Cu at a small cyclotron. Radiochim. Acta (1992) 57(1):1–6. 10.1524/ract.1992.57.1.1
40.
SinghBPSharmaMKMusthafaMMBhardwajHDPrasadR.A study of pre-equilibrium emission in some proton- and alpha-induced reactions. Nucl. Instrum. Methods Phys Res A (2006) 562(2):717–20. 10.1016/j.nima.2006.02.030
41.
AmjedNTárkányiFHermanneADitróiFTakácsSHussainM.Activation cross-sections of proton induced reactions on natural Ni up to 65MeV. Appl. Radiat. Isot (2014) 92:73–84. 10.1016/j.apradiso.2014.06.008
42.
ObataAKasamatsuSMcCarthyDWWelchMJSajiHYonekuraYet alProduction of therapeutic quantities of 64Cu using a 12 MeV cyclotron. Nucl. Med. Biol (2003) 30:535–9. 10.1016/S0969-8051(03)00024-6
43.
SynowieckiMAPerkLRNijsenJFW.Production of novel diagnostic radionuclides in small medical cyclotrons. EJNMMI Radiopharm. Chem (2018) 3(1):3. 10.1186/s41181-018-0038-z
44.
KozempelJAbbasKSimonelliFZampeseMHolzwarthUGibsonNet alA novel method for n.c.a. 64Cu production by the 64Zn(d,2p)64Cu reaction and dual ion-exchange column chromatography. Radiochim. Acta (2007) 95:75–80. 10.1524/ract.2007.95.2.75
45.
AsabellaNCasciniGLAltiniCPaparellaDNotaristefanoARubinG.The copper radioisotopes: a systematic review with special interest to 64Cu. BioMed Res. Int (2014) 2014:786463. 10.1155/2014/786463
46.
NiculaeDIlieSLeonteRChilugLCraciunL.Automated production and purification of copper medical radioisotopes in a variable energy cyclotron using solid targetsThe 23rd International Symposium on Radiopharmaceutical Sciences (ISRS 2019). J. Label Compd. Radiopharm (2019) 62(S1):S301–S302. 10.1002/jlcr.3725
47.
do CarmoSJCScottPJHAlvesF.Production of radiometals in liquid targets. EJNMMI Radiopharm. Chem (2020) 5:2. 10.1186/s41181-019-0088-x
48.
McCarthyDWSheferREKlinkowsteinREBassLAMargeneauWHCutlerCSet alEfficient production of high specific activity 64Cu using A biomedical cyclotron. Nucl. Med. Biol (1997) 24:35–43. 10.1016/s0969-8051(96)00157-6
49.
Avila-RodriguezMANyebJANicklesRJ.Simultaneous production of high specific activity 64Cu and 61Co with 11.4MeV protons on enriched 64Ni nuclei. Appl. Radiat. Isot (2007) 65:1115–20. 10.1016/j.apradiso.2007.05.012
50.
SoLVPellegriniPKatsifisAHowseJGreguricI.Radiochemical separation and quality assessment for the 68Zn target based 64Cu radioisotope production. J. Radioanal. Nucl. Chem (2008) 277(2):451–66. 10.1007/s10967-007-7143-x
51.
WilliamsHARobinsonSJulyanPZweitJHastingsD.A comparison of PET imaging characteristics of various copper radioisotopes. Eur. J. Nucl. Med. Mol. Imag (2005) 32(12):1473–80. 10.1007/s00259-005-1906-9
52.
SmithSVWatersDJBartoloND.Separation of Cu from 67Ga waste products using anion exchange and low acid aqueous/organic mixtures. Radiochim. Acta (1996) 75:65–8. 10.1524/ract.1996.75.2.65
53.
MettlerFAJrGuiberteauMJ.Radioactivity, radionuclides and radiopharmaceuticals. In: MettlerMiltonFAGuiberteauJ., editors Esentials of nuclear medicine Imaging. 6th ed:Philadelphia: W.B. Saunders (2012). p. 1–21.
54.
ZweitJSmithAMDowneySSharmaHL.Excitation functions for deuteron induced reactions in natural nickel: production of No-carrier-added 64Cu from enriched 64Ni targets for positron tomography emission. Appl. Radiat (1991) 42(2):193–7. 10.1016/0883-2889(91)90073-a
55.
AlvesFAlvesVHPdo CarmoSJCNevesACBSilvaMAbrunhosaAJ.Production of copper-64 and gallium-68 with a medical cyclotron using liquid targets. Mod. Phys. Lett (2017) 32 (17). 10.1142/S0217732317400132
56.
AbbasKKozempelJBonardiMGroppiFAlfaranoAHolzwarthUet alCyclotron production of 64Cu by deuteron irradiation of 64Zn. Appl. Radiat. Isot (2006) 64:1001–5. 10.1016/j.apradiso.2005.12.021
57.
FerrariCAsabellaANVillanoCGiacobbiBCoccettiDPanichelliPet alCopper-64 dichloride as theranostic agent for glioblastoma multiforme: a preclinical study. BioMed Res. Int (2015). 10.1155/2015/129764
58.
JalilianAROssoJJrThe current status and future of theranostic copper-64 radiopharmaceuticals. Iran. J. Nucl. Med (2017) 25(1):1–10.
59.
MaMTDonnellyPS.Peptide targeted copper-64 radiopharmaceuticals. Curr. Top. Med. Chem (2011) 11:500–20. 10.2174/156802611794785172
60.
SantiniCPelleiMGandinVPorchiaMTisatoFMarzanoC.Advances in copper complexes as anticancer agents. Chem. Rev (2014) 114(1):815–62. 10.1021/cr400135x
61.
EvangelistaLMansiLCasciniGL.New issues for copper-64: from precursor to innovative pet tracers in clinical oncology. Curr. Rad (2013) 6(3):117–23. 10.2174/18744710113069990020
62.
AndersonCJFerdaniR.Copper-64 radiopharmaceuticals for PET imaging of cancer: advances in preclinical and clinical research. Cancer Biother. Radiopharm (2009) 24(4):379–93. 10.1089/cbr.2009.0674
63.
BassLAWangMWelchMJAndersonCJ.In Vivo transchelation of copper-64 from TETA-octreotide to superoxide dismutase in rat liver. Bioconjug. Chem (2000) 11(4):527–32. 10.1021/bc990167l
64.
CaiZAndersonCJ.Chelators for copper radionuclides in positron emission tomography radiopharmaceuticals. J. Label Compd. Radiopharm (2014) 57(4):224–30. 10.1002/jlcr.3165
65.
BoswellCASunXNiuWWeismanGRWongEHRheingoldALet alComparative in Vivo stability of copper-64-labeled cross-bridged and conventional tetraazamacrocyclic complexes. J. Med. Chem (2004) 47(6):1465–74. 10.1021/jm030383m
66.
WeiLYeaYWadasTJLewisJSWelchMJAchilefuSet al64Cu-labeled CB-TE2A and diamsar-conjugated RGD peptide analogs for targeting angiogenesis: comparison of their biological activity. Nucl. Med. Biol (2009) 36:277–85. 10.1016/j.nucmedbio.2008.12.008
67.
QinCLiuHChenKHuXMaXLanXet alTheranostics of malignant melanoma with 64CuCl2. J. Nucl. Med (2014) 55(5):812–7. 10.2967/jnumed.113.133850
68.
CatalognaGTalaricoCDattiloVGangemiVCalabriaFD’AntonaLet alThe SGK1 kinase inhibitor SI113 sensitizes theranostic effects of the 64CuCl2 in human glioblastoma multiforme cells. Cell Physiol. Biochem (2017) 43:108–19. 10.1159/000480328
69.
LewisJSLaforestRBuettnerTLSongSKFujibayashiYConnettJMet alCopper-64-diacetyl-bis(N4-methylthiosemicarbazone): an agent for radiotherapy. Proc. Natl. Acad. Sci. U.S.A (2001) 98(3):1206–11. 10.1073/pnas.98.3.1206
70.
LewisJSLewisMRCutlerPDSrinivasanASchmidtMASchwarzSWet alRadiotherapy and dosimetry of 64Cu-TETA-Tyr3-Octreotate in a somatostatin receptor-positive, tumor-bearing rat model. Clin. Cancer Res (1999) 5(11):3608–16.
71.
ObataAKasamatsuSLewisJS.Basic characterization of 64Cu-ATSM as a radiotherapy agent. Nucl. Med. Biol (2005) 32(1):21–8. 10.1016/j.nucmedbio.2004.08.012
72.
CaiHWuJSMuzikOHsiehJTLeeRJPengF.Reduced 64Cu uptake and tumor growth inhibition by knockdown of human copper transporter 1 in xenograft mouse model of prostate cancer. J Nucl. Med (2014) 55(4):622–8. 10.2967/jnumed.113.126979
73.
RighiSUgoliniMBottoniG.Biokinetic and dosimetric aspects of 64CuCl2 in human prostate cancer: possible theranostic implications. EJNMMI Res (2018) 8(1):18. 10.1186/s13550-018-0373-9
74.
Experimental nuclear reaction data (EXFOR) database. IAEA – International Atomic Energy Agency. Available from: https://www-nds.iaea.org/exfor/ (Accessed May 30, 2020).
Summary
Keywords
molecular imaging, theranostics (combined therapeutic and diagnostic technology), copper, emergent radioisotopes, cancer, Cu-64/67
Citation
Niculae D, Dusman R, Leonte RA, Chilug LE, Dragoi CM, Nicolae A, Serban RM, Niculae DA, Dumitrescu IB and Draganescu D (2021) Biological Pathways as Substantiation of the Use of Copper Radioisotopes in Cancer Theranostics. Front. Phys. 8:568296. doi: 10.3389/fphy.2020.568296
Received
31 May 2020
Accepted
27 November 2020
Published
14 January 2021
Volume
8 - 2020
Edited by
Yolanda Prezado, INSERM U1021 Signalisation normale et pathologique de l’embryon aux thérapies innovantes des cancers, France
Reviewed by
Rosa Maria Moresco, University of Milano-Bicocca, Italy
Shi-Ying Li, Guangzhou Medical University, China
Updates
Copyright
© 2021 Niculae, Dusman, Leonte, Chilug, Dragoi, Nicolae, Serban, Niculae, Dumitrescu and Draganescu.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ramona Dusman, ramona.dusman@nipne.ro; Dana Niculae, dana.niculae@nipne.ro
This article was submitted to Medical Physics and Imaging, a section of the journal Frontiers in Physics
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.