Abstract
Pharmaceutical and non-pharmaceutical interventions (NPIs) have been crucial for controlling COVID-19. They are complemented by voluntary health-protective behavior, building a complex interplay between risk perception, behavior, and disease spread. We studied how voluntary health-protective behavior and vaccination willingness impact the long-term dynamics. We analyzed how different levels of mandatory NPIs determine how individuals use their leeway for voluntary actions. If mandatory NPIs are too weak, COVID-19 incidence will surge, implying high morbidity and mortality before individuals react; if they are too strong, one expects a rebound wave once restrictions are lifted, challenging the transition to endemicity. Conversely, moderate mandatory NPIs give individuals time and room to adapt their level of caution, mitigating disease spread effectively. When complemented with high vaccination rates, this also offers a robust way to limit the impacts of the Omicron variant of concern. Altogether, our work highlights the importance of appropriate mandatory NPIs to maximise the impact of individual voluntary actions in pandemic control.
1 Introduction
During the COVID-19 pandemic, the virus has played a central role in people’s day-to-day conversations and the information they search for and consume [1]. The growing amount of news and specialized literature on COVID-19 can inform individual decisions in a wide range of situations and on various timescales [2]. For example, people decide multiple times every day how closely they follow mask-wearing regulations or meeting restrictions. However, if hesitant, they might take weeks or months to decide whether to accept a vaccine. These decisions impact the spreading dynamics of COVID-19 and ultimately determine the effectiveness of interventions and how smoothly we transit to SARS-CoV-2 endemicity.
While typical models of disease spread consider that individual behavior affects the spreading dynamics of an infectious disease, they often neglect that there is also a relation in the opposite causal direction. This feedback loop comprises that, e.g., mass media regularly updates individuals on the latest local developments of the pandemic, such as the current occupancy of intensive care units (ICUs). This information affects individuals’ opinions and risk perceptions and, thus ultimately their actions [3]. For example, given high perceived risk, individuals reduce their non-essential contacts beyond existing regulations and increase their willingness to accept vaccine offers accordingly, an effect observed in empirical research conducted with routine surveys in Germany [4] and other parts of the world [5–8]. However, to quantify the effect of individual voluntary actions on the dynamics of COVID-19, two questions remain open: 1) What is the relationship between risk perception and voluntary action, on the one hand, and the spread of the disease, on the other hand; and 2) what is the relative contribution of voluntary action when mandatory restrictions are in place?
In this work, we aim to quantify the impact of voluntary actions on disease spread while studying the questions mentioned above for the COVID-19 pandemic. 1) We analyze survey and COVID-19 vaccination data in European countries to uncover the relationship between the occupancy of ICUs—which determines the perceived risk—and voluntary immediate health-protective behavior as well as the willingness to get vaccinated. We then incorporate these effective feedback loops into a deterministic compartmental model (Figure 1A). 2) We decompose the overall contact structure into contextual contacts (Figure 1B) and for each context define a range in which voluntary action can be adapted according to individual risk-perception, given the level of mandatory non-pharmaceutical interventions (NPIs). To that end, we use the functional form identified in 1) (Figure 2). We explore different intervention scenarios in the face of adverse seasonality [9–11], using as reference the winter 2021/2022 in central Europe. Our analysis confirms that both extremes (“freedom day” or stringent measures throughout) bear large harms in the long run. However, when measures leave space for voluntary actions, people’s adaptive behavior can efficiently contribute to breaking the wave and change the course of the pandemic.
FIGURE 1
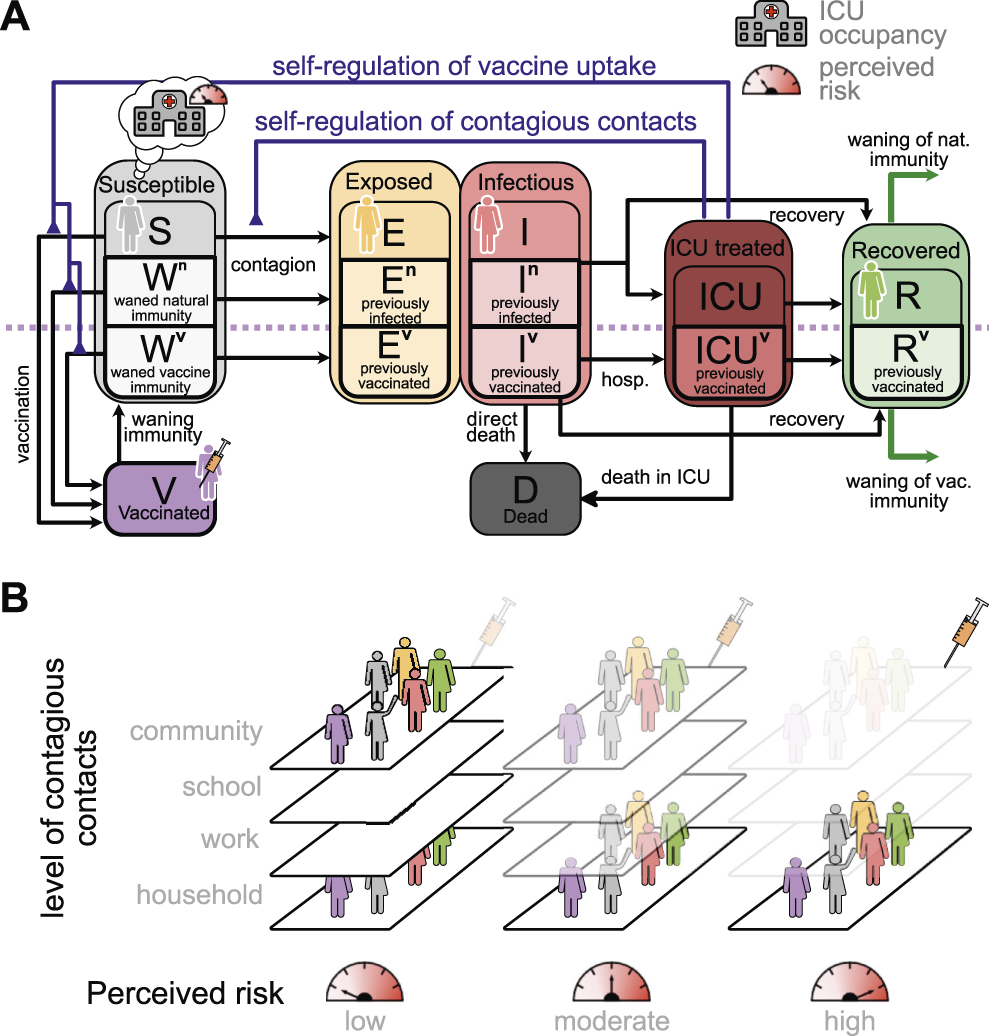
Interplay between risk perception and voluntary health-protective behavior. (A): Sketch of the proposed age-stratified compartmental model of disease spread, which incorporates different stages for disease progression and immunological conditions of the susceptible population with their respective chances of being infected and developing a severe course (Supplementary Figure S1, Supplementary Information, for full model). The behavioral feedback (blue lines) changes individuals’ contagious contact behavior, as well as their willingness to get vaccinated, and hence the effective spreading rate. (B): We use the contact matrix of [12], which yields the contact rates at home, school, work and in the community for each age-group. For the subsequent scenarios, we adapt these contexts of contacts separately. Some of the contacts are by definition hard to reduce voluntarily (e.g., household contacts), while others (at school and work) strongly depend on current mandatory non-pharmaceutical interventions (Supplementary Figure S3 for details).
FIGURE 2
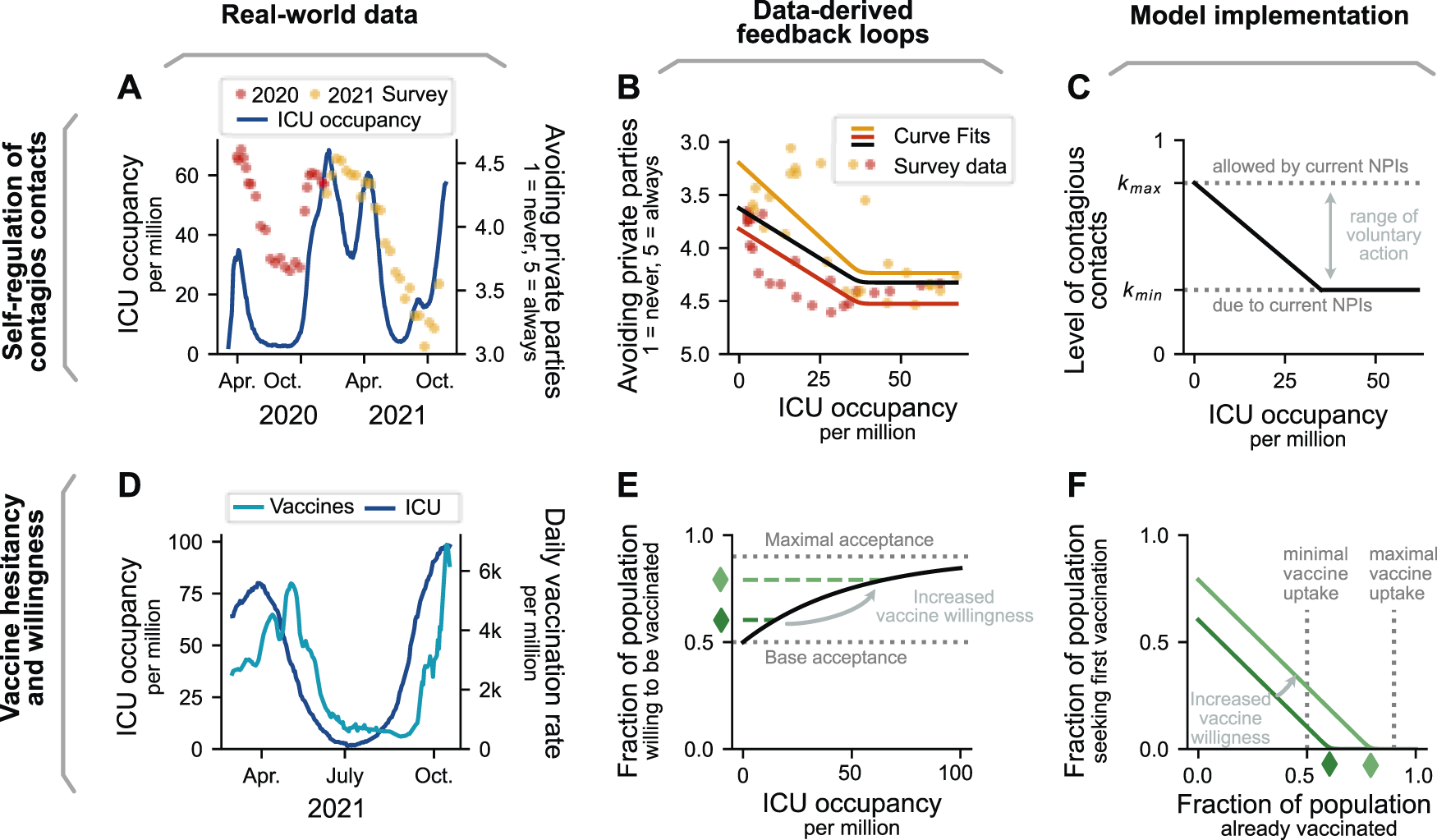
Data-derived formulation of behavioral feedback loops. (A): Reported contact reductions follow intensive care unit (ICU) occupancy in Germany. Survey participants were asked how likely they were to avoid private parties over the course of the pandemic on a discrete scale from 1 (never) to 5 (always) [4]. To decouple the effect of vaccination availability, we present 2020 (red) and 2021 (yellow) data separately. Ticks indicate the middle of the month. (B): The survey data on contact reduction and the ICU occupancy are related. The piece-wise linear relationship shows the reduction of contacts with increasing ICU occupancy, and for even higher ICU occupancy a saturation. Red, yellow, and black represent fits to the data from 2020, 2021, and overall, respectively. (C): In the model, the contact reduction and its dependency on ICU occupancy is implemented as a multiplicative reduction factor k that weighs the age-dependent contextual contact matrices (Figure 1B). (D): Vaccine uptake increases with ICU occupancy in Romania (shown here) and other European countries (Supplementary Figure S4). (E): Willingness to accept a vaccine offer is modeled using an exponentially-saturating function, ranging between a lower and upper bound of acceptance depending on ICU occupancy. The bounds represent that a fraction of people is willing to be vaccinated even at no immediate threat (no ICU occupancy), and another fraction is not willing or able to get vaccinated no matter the threat. (F): Vaccines are delivered at a rate proportional to the number of people seeking a vaccine, i.e., the difference between the number of people willing to be vaccinated and those already vaccinated. Thus, when the number of already vaccinated equals the number of people willing to get vaccinated, no more vaccinations are carried out. The same functional shape describes the booster uptake.
2 Results
2.1 Data-Derived Behavioral Feedback Loops
Throughout this manuscript, we investigate how the interplay between information about the COVID-19 pandemic and its spreading dynamics is mediated by the perception of risk. Risk perception modulates both, 1) people’s immediate voluntary health-protective behavior, e.g., their level of contacts and their adherence to mask-wearing and hygiene recommendations, and 2) their willingness (or hesitancy) to receive vaccination (Figure 1). Individuals constantly receive information on the current COVID-19 incidence, ICU occupancy, and deaths (which are all closely related [13–15]) either via news outlets or because of reports about COVID-19 cases in their social circles. Hence, the risk they perceive depends on this evolving trend over time.
We tailor our approach to the situation of the COVID-19 pandemic, i.e., to a disease having the following characteristics: 1) high transmissibility, 2) relatively low infection fatality rate, 3) widespread vaccine hesitancy, 4) waning immunity, and 5) public attention and coverage. We differentiate from the approaches of [16–18] as we neither model the contagion of fear explicitly nor a direct coupling between incidence and fear. Instead, we assume that individuals build their perception of risk based on the ICU occupancy over time using a memory function, similar to the theoretical approach in [19, 20]. This is a sensible choice, as ICU occupancy signals 1) how likely governmental bodies are to re-implement emergency NPIs to prevent overwhelming healthcare facilities (and thereby limit individual freedoms), and 2) how likely it is that an individual’s close contacts (or their contacts) would have been severely ill. Besides, our modeling framework constitutes a methodological advancement from that presented in [17], as we provide a detailed description of all epidemiologically relevant disease states and several external effects influencing its spread, such as seasonality, contextual contact networks and NPIs.
We assume that individuals base their decisions about heath-protective behavior on the recent developments of the pandemic. Following the ideas of Zauberman et al. about perception of time in decision-making [21], we consider that when individuals decide about behavior that only has immediate protective effects, they consider only the current risk-level. For instance, when deciding whether or not to wear a mask in the supermarket on a given day, they only consider the most recently reported ICU occupancy. Decisions with longer-term protection, in contrast, are also based on a longer-term risk-assessment. When deciding whether or not to get a booster vaccine, for example, individuals do not only take into account the ICU-occupancy on the day of the decision but they are looking back at a longer period. We detail the assumptions about the perceived risk-level and the resulting health-protective behavior in the Methods section. In the following, we sketch the derivation of the feedback loops from this perceived risk to people’s immediate voluntary health-protective behavior and willingness to get vaccinated.
2.1.1 Feedback on Health-Protective Behavior
To determine the explicit relationship between the perceived level of risk and immediate voluntary health-protective behavior—which presents one of the feedback loops in our model—we exploit results from the German COSMO study, a periodic survey where participants are asked about their opinions and behavior regarding the COVID-19 pandemic and NPIs [4]. Their answers on adhering to health-protective behavior recommendations (avoiding private parties in this case) correlate with the ICU occupancy in Germany at the time (Figure 2A). However, at very high ICU occupancy, adoption of health-protective behavior seems to reach a plateau (Figure 2B); no further adoption seems to be feasible, arguably because those individuals willing to engage in health-protective behavior have done so already as far as they can, and those unwilling are insensitive to higher burden on ICUs. Hence, we fit a piece-wise linear function (with a rounded edge at the transition—called a softplus) to the COSMO data [Pearson correlation coefficient r = 0.64 for 2020–2021 (black), r = 0.81 for 2020 (red) and r = 0.53 for 2021 (yellow)] and use it for the feedback between information in terms of ICU occupancy and voluntary health-protective behavior (Figure 2C and Methods for details).
2.1.2 Feedback on Vaccination Behavior
The second feedback loop in our model describes the relationship between the level of perceived risk and vaccine hesitancy. To quantify it, we study the vaccination trends in different European countries and compare them with the trends in ICU occupancy (Supplementary Figure S4, Supplementary Information). The case of Romania (Figure 2D) illustrates the relation very clearly: Vaccination rates follow the ICU occupancy with a delay of a few weeks. By analyzing the correlation between vaccination rate and ICU occupancy with a variable delay, we reach the highest Pearson correlation coefficient (0.96) with a delay of 25 days. However, the specific reaction delay and magnitude of the effect differs between countries (Supplementary Figure S4). In our model, we propose that as ICU occupancy increases so does the willingness to get vaccinated (i.e., higher probability of accepting a vaccine offer when ICU occupancy is high). As not everybody in the population is willing to accept a vaccine offer, the willing fraction of the population is a function that saturates below 1 (Figure 2E). With this formulation, vaccinations are only carried out if the fraction of the population willing to get vaccinated is larger than the fraction of currently vaccinated (Figure 2F and Methods for details).
Our model can capture two features observed in real-world vaccination programs. First, when case numbers are low and vaccine uptake high, rational agents might have insufficient incentives for getting vaccinated. Assuming a high perceived risk of vaccine side effects, the agents would thus decline vaccination when offered. The above is known as the free-rider problem in game theory and economics [22]. Second, the two feedback loops in our model and the incorporation of waning immunity allows us to observe different incidence curve shapes and replicate recurrent waves of infections. The above is a necessary validity check, as real-world outbreaks exhibit a large variety of incidence curve shapes [23]. These may ultimately unveil universal patterns of disease spread that are consistent across countries [24].
2.2 Behavioral Feedback Loops Yield More Realistic Results than Classical Models
Classical SEIR-like compartmental models have found wide application in the first stages of the COVID-19 pandemic. In these models, the different stages of disease progression are represented by separate compartments and individuals transit from one to another at a given (and typically constant) transition rate. In that way, an infectious disease outbreak will proliferate if the spreading rate of the disease is larger than the recovery rate and if a large-enough fraction of the population is susceptible to being infected. However, these simple models often tend to overestimate the size of an infectious disease outbreak or all possible trajectories for the incidence trends [23], as they do not incorporate mechanisms of dynamical adaptation of restrictions [25] or, as studied in this paper, behavior.
We observe that including the feedback loops described above reduces the peak in incidences and hospitalizations while keeping the timing of the wave almost unchanged (see Figure 3). More generally, these feedback loops break increasing and declining trends, resulting in long but flat infection plateaus or multiple waves. Compared to classical SEIR-like models, where two dynamical regimes are possible—exponential growth or decay of case numbers, when neglecting waning immunity—, our model captures a broader spectrum of dynamics by linking ICU occupancy with individuals’ health-protective voluntary behavior and vaccine uptake.
FIGURE 3
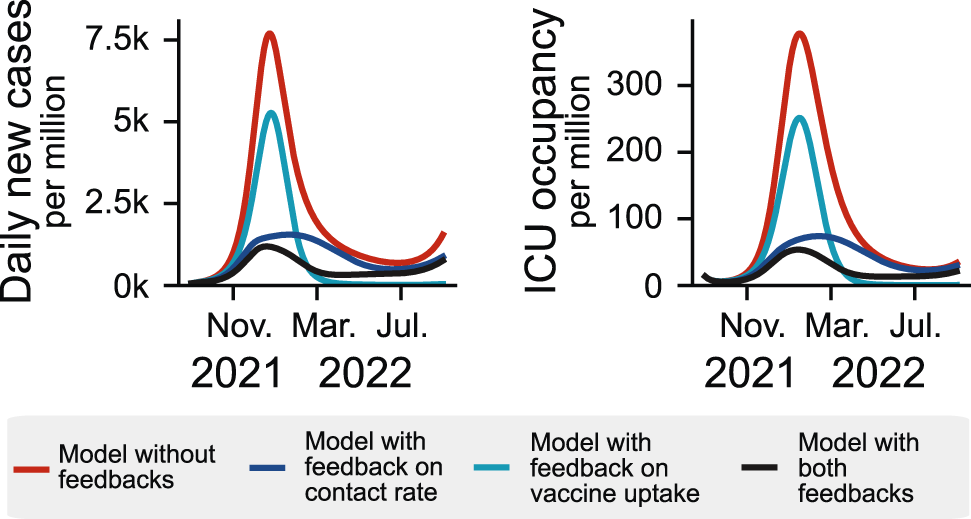
Incorporating behavioral feedback loops in compartmental models broadens the dynamic range of the solutions and yields more realistic results. Different variations of a compartmental model are displayed to show the effect of the two feedback loops used in our model: When ICU occupancy increases, individuals increase their health-protective behavior and are more willing to be vaccinated. This dynamical adaptation can break a wave at lower case numbers and lead to extended infection plateaus (blue curves), which a classic compartment model is unable to reproduce as it does not incorporate the population’s reaction to the disease (red curve).
2.3 Policies With Either too Weak or too Strong Interventions Throughout Winter Bear Higher Levels of Mortality and Morbidity
Using parameters obtained from surveys and other data sources (Supplementary Table S3, Supplementary Information), we analyze five scenarios of mandatory NPIs throughout winter (for all age-stratified results see Supplementary Material): 1) no NPIs at all, 2)-4) moderate NPIs and 5) strong NPIs (Methods for details). The stringency of the scenarios and the seasonal effects are depicted in Figures 4A,B and Figures 5A,B. As an example case, we assume a country with a total vaccination rate of 60% and a recovered fraction of 20%. Note that we include the possibility of overlaps between vaccinated and recovered. Thus, the total fraction of immune individuals does not add up to 80% but 68%. For more detail on the initial conditions, see Supplementary Material, Supplementary Section S3.1.
FIGURE 4
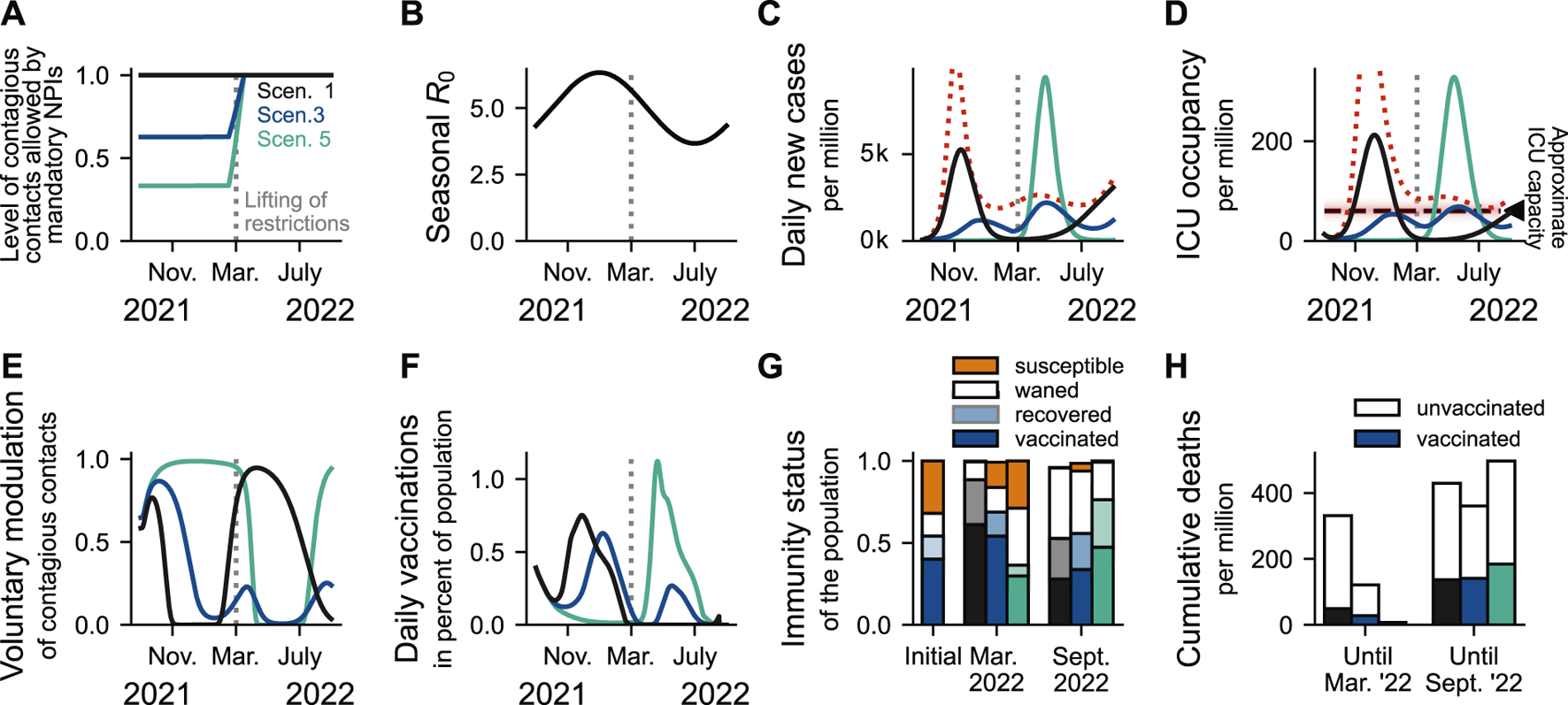
Maintaining moderate contact restrictions throughout winter outperforms extreme scenarios in balancing the burden on ICUs by allowing people the freedom to act according to their risk perception. The level of mandatory NPIs sustained throughout winter 2021/2022, together with people’s voluntary preventive actions, determines case numbers and ICU occupancy over winter and beyond. Ticks are set on the first day of the month. (A): The three displayed scenarios of mandatory NPI stringency in winter reflect “freedom-day” with only basic hygiene measures (black), considerable contact reduction and protective measures (e.g., mandatory masks) in school, at the workplace and in the community (blue), and strong contact reduction and partial school closure (mint). All measures are gradually lifted centred around 1 March 2022, over the course of 4 weeks. (B): The seasonality of the basic reproduction number R0. (C,D): Scenario 1 (black): Without mandatory restrictions, incidence and ICU occupancy increase steeply; this increases voluntary health-protective behavior and vaccine uptake in the population (E,F), and leads to higher rates of naturally acquired immunity (G), but also high mortality and morbidity in winter (H). Note that disproportionally more vaccinated individuals die after March 2022 because, at this point, most of the population is vaccinated. A “full wave” is added in (C,D) (red dotted line), depicting the development of case numbers and ICU occupancy in the absence of behavioral feedback mechanisms. Scenario 3 (blue): Maintaining moderate restrictions would prevent overwhelming ICUs while allowing for higher vaccine uptakes and rates of post-infection immunity. Scenario 5 (mint): Maintaining strong restrictions would minimize COVID-19 cases and hospitalizations in winter, generating a perception of safety across the population. However, this perceived safety is expected to lower the incentives to get vaccinated. Furthermore, immunity of all kinds will wane over winter. Altogether, this can cause a severe rebound wave if restrictions are completely lifted in March. Furthermore, in all scenarios where ICU capacity is exceeded, we would in reality expect either disproportionally higher mortality due to the burden on the health system or a change in mandatory NPIs.
FIGURE 5
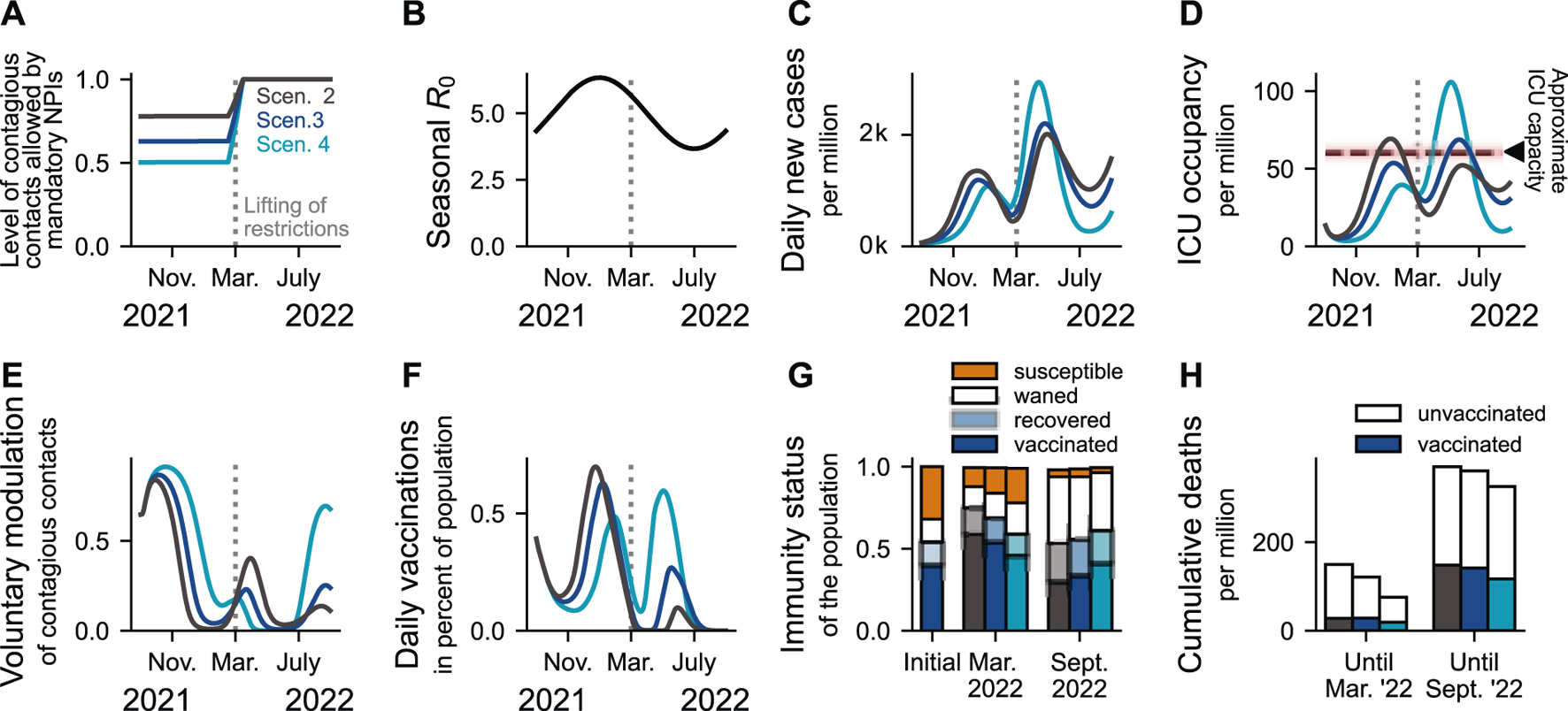
Moderate restrictions leave enough room for effective adaptation of behavior to perceived risk. (A): We explore three scenarios with similar levels of moderate mandatory NPIs sustained throughout winter, the period of adverse seasonality (B). Considering Scenario 3 as reference, moderate restrictions seem to be robust against relaxations of NPIs, as both morbidity and mortality are similar to that of Scenario 2 (C,D,H). However, a perturbation with less strength in the opposite direction (Scenario 4, increasing mandatory NPIs) has a disproportional effect on ICU occupancy. These differences are based on the modulation of voluntary contacts (E) and vaccine uptake (F). Thus, when leaving room for adaption of health-protective measures to perceived risk, people’s behavior will stabilize moderate scenarios where mandatory NPIs are strong enough to prevent a major surge, but not over-protective, so individuals find it rewarding to be vaccinated and to adapt their level of contacts. Note that disproportionally more vaccinated individuals die after March 2022 because, at this point, most of the population is vaccinated (G).
Without any mandatory NPIs throughout winter (Scenario 1, Figure 4, black lines), case numbers and hospitalizations will show a steep rise (Figures 4C,D). As a consequence, individuals voluntarily adapt their health-protective behavior and are more inclined to accept a vaccine offer (Figures 4E–G). Although this scenario features unrealistically high mortality and morbidity, modeling results in the absence of any behavior feedback mechanisms yield even higher levels (cf. Figures 4C,D, dotted red line).
In contrast, suppressing the seasonal wave through strong mandatory NPIs (Scenario 5, Figure 4, mint lines) and thereby maintaining low case numbers through winter only delays the wave to a later but inevitable date once restrictions are lifted (Figures 4C,D). Low COVID-19 incidence throughout winter implies 1) low post-infection immunity, 2) little incentives for first or booster vaccination, 3) waning immunity, and 4) lower rates of “naturally” boosting immune memory upon re-exposure to the virus [26]. The resulting low immunity levels (Figure 4G) then fuel a higher rebound wave when restrictions are lifted in March 2022, despite favorable seasonality. Similar rebound waves have been observed for other seasonal respiratory viruses [27, 28].
Interestingly, the middle strategy, namely moderate NPIs during winter, prevents the high wave in winter as well as the rebound wave in spring that characterize the scenarios with no or with strong NPIs, respectively (Scenario 3, Figure 4, dark blue). Unlike in the extreme scenarios, the ICU capacity in Scenario 3 is not exceeded in any season, hence avoiding reduced health care quality and strong burden to health care workers. Figure 4H shows that the death toll in Scenario 3 is lower than in the other scenarios. In reality however, this difference would be much larger because Scenarios 1 and 5 surpass the assumed ICU capacity by far; that would imply disproportionally higher mortality, an effect we did not quantify in our model. Alternatively, emergency mandatory NPIs would be introduced, which we do not model here.
2.4 Voluntary Actions can Dampen the Wave if Restrictions are Moderate
As presented in the previous section, extreme scenarios (Scenarios 1 and 5) bear high levels of morbidity and mortality. However, in scenarios with intermediate restriction levels (Scenarios 2–4, Figure 5A), voluntary preventive actions (Figure 5E) can compensate for slightly too low levels of mandatory NPIs, provided that these NPIs are strong enough to prevent a surge in COVID-19 incidence that might be too sudden or strong for individuals to voluntarily adopt health-protective behavior (Figures 5C,D). For example, while having different levels of mandatory NPIs, Scenarios 2 and 3 reach similar peaks in ICU occupancy (Figure 5D). Conversely, despite considering a proportional increase in the strength of NPIs (comparable to that from Scenario 2 to 3, Figure 5A), Scenario 4 is too protective: there are too few incentives to get vaccinated (Figure 5F) due to the low risk perception as well as too few infections (Figure 5C) and, hence, appropriate immunity levels are not reached (Figure 5G). As a consequence, a disproportionally larger off-seasonal wave in spring overwhelms ICUs (Figure 5D). Noteworthy, even though the nominal mortality is the lowest for Scenario 4 (Figure 5H), this value does not account for triage-induced over-mortality or novel necessary NPIs that would be likely be imposed and is thus invalid.
2.5 Case Study: Emergence of the Omicron Variant of Concern and its Effect on Case Numbers
A risk that cannot be neglected is the emergence of SARS-CoV-2 variants of concern (VOC), such as the Omicron VOC. This variant is rapidly replacing the Delta VOC, thus posing an imminent risk. Although there is substantial uncertainty about its epidemiological features, preliminary evidence shows: Compared to the Delta VOC, Omicron exhibits 1) an increased risk of reinfection or break-through infection [29–31], 2) a substantial reduction in antibody neutralization [32–38], 3) a reduction in vaccine effectiveness against infection [31, 37, 39–44], and 4) faster spread [30, 31, 45, 46] mainly due to immune escape [47].
Given this evidence, we analyze the impacts of a potential full replacement of the dominant Delta VOC by the Omicron VOC by 15th of January 2022. We incorporate the protection against infection by booster doses. As example scenario, we start with Scenario 3 (moderate mandatory NPIs), as it resembles a typical development in Europe. We then analyze four different possible reactions to the Omicron VOC, i.e., starting to switch from Scenario 3 to Scenarios 1, 3, 4, or 5 before it takes over (Figure 6A). We evaluate three possibilities regarding the booster vaccine-protection against infection, 50, 65, and 80% (relative to the protection granted for Delta). This is consistent with available evidence suggesting Omicron’s immune escape to reduce vaccine effectiveness against symptomatic disease to about 73% for freshly mRNA-boosted individuals [32]. Furthermore, we explore two possibilities of severity of infections after previous immunization: Either efficacy against severe course remains the same as with Delta, both for the immunized and immune-naive persons (Figures 6B,E,H), or protection is five times better for the immunized (Figures 6C,F,I).
FIGURE 6
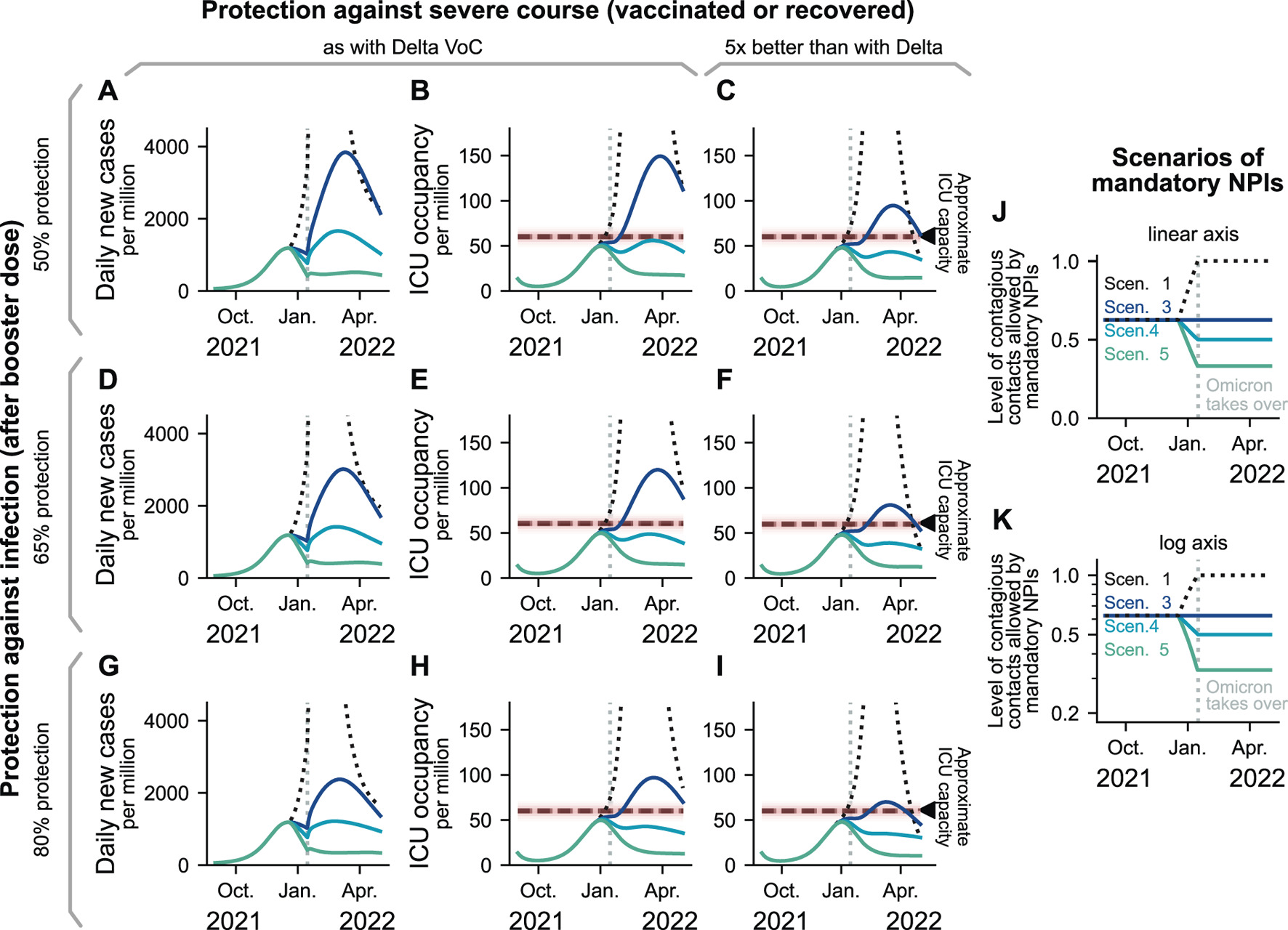
Development of the pandemic under the emergence of the Omicron VOC. Assuming a full replacement of Delta by the Omicron VOC on 15th of January 2022, we model three different possibilities for vaccine-protection against infection, and two levels of long-lasting vaccine- or post-infection protection against severe course (A–I). In color, we display four scenarios that are derived from the previously studied ones (J,K). All scenarios share moderate mandatory NPIs until mid December 2021, where we evaluate different possibilities for policy adaptation to mitigate the spread of the Omicron VOC. (A,B,D,E,G,H): Case numbers and ICU occupancy while assuming that a protection against hospitalization (once infected despite previous immunization) is similar to the protection against Delta. (C, D, I): ICU occupancy while assuming a protection against hospitalization (once infected and after previous immunization) five times better than the protection regarding Delta.
As expected, the enhanced transmissibility resulting from the partial escape of the Omicron VOC breaks the decreasing trend in case numbers observed for Scenarios 3, 4, and 5 from the moment where the replacement takes place (Figures 6A,D,G). This results in a substantial surge in daily new cases in all scenarios except for Scenario 5 (most restrictive). Regarding ICU occupancy, our results depend strongly on the assumed protection against infection by recent vaccination or boosters. When the protection against infection granted by recently administered vaccines is above 50%, both Scenarios 4 (which has a more strict testing policy and further reduced contacts compared to Scenario 3) and 5 (in addition, group sizes in school are reduced) yield optimistic results for ICU occupancy. If Omicron infections lead to much less severe course of the disease for immunized or convalescent individuals, then even Scenario 3 can avoid severely overfilling intensive care units. We have represented Scenario 1 (lifting all mandatory NPIs) with dashed lines, as it yields unrealistic results: Stricter NPIs would probably be reinstated if ICU occupancy becomes too high. The scenarios end in April, where we expect that an updated booster vaccine is developed and distributed. In that phase, lifting restrictions at the pace of vaccination and aiming for low case numbers would maximize freedom while minimizing mortality and morbidity [25, 48–50].
3 Discussion
Modeling the interplay of human behavior and disease spread is one of the grand challenges of infectious disease modeling. While not being the first to model behavioral adaptation [17, 51–55], we incorporate data-driven insights into our modeling framework, inspiring the explicit functional dependency between risk and health-protective behavior as well as vaccine hesitancy in the context of the COVID-19 pandemic. Thereby, we can incorporate self-regulation mechanisms into our scenario analysis, which best qualitatively describe what is to be expected in the future or in the event of the emergence of novel SARS-CoV-2 VOCs, such as the Omicron variant. We hence take a further step towards more empirically-grounded mathematical models.
Within our framework, a smooth transition to SARS-CoV-2 endemicity requires, besides a working and accepted vaccine, two ingredients. First, mandatory NPI levels should be high enough to prevent a surge in case numbers so fast that individuals could not react on time to prevent overwhelming ICUs. Second, mandatory NPIs should leave enough room so that individuals can effectively adopt voluntary preventive actions as a response to an increased perception of risk. Hence, governments must guarantee that the decision to, e.g., attend non-essential face-to-face activities that could be carried out remotely remains in the individual’s hands. Under such circumstances, voluntary actions can dampen the wave and prevent overwhelming ICUs (Scenarios 2 and 3, Figure 5). Otherwise, irresponsible or overprotective measures would result in a wave that could surpass the healthcare capacity in the short term or when lifting all measures (Scenarios 1, 4, and 5, Figures 4, 5). In any case, people’s awareness about the danger of a disease should ideally be driven by trust in scientific and governmental bodies instead of by the current burden to the healthcare system. Hence, it is crucial during a disease outbreak to engage in extensive, expert-guided, and audience-tailored risk communication [56] and to prevent the spread of mis- and dis-information that could damage general trust [57, 58].
Despite the empirical basis of our approach, the functional shape of the feedback mechanisms remains one of the main uncertainties in our model. The voluntary adoption of health-protective measures was inspired by survey data [4], and is thus bound to its limitations. Additionally, as ICU capacity was never extremely overwhelmed in Germany in the time frame of the COSMO survey, the study does not provide information on how people would act at very high levels of ICU occupancy; in principle, such emergency situations would trigger even stronger reactions in the population, and certainly also a change in NPI stringency (which we assumed to be constant throughout). Furthermore, when extrapolating our results to other countries, one should consider cultural differences or varying levels of trust in governmental bodies. Therefore, more empirical research to inform model assumptions and parameters remains crucial.
Vaccine uptake and coverage are critical parameters that determine mortality and morbidity levels. In line with what has been observed in high-income countries, we assume that vaccination rates are mostly limited by vaccine hesitancy instead of vaccine stocks or logistics. In that way, we can deal with emergent VOCs (as Omicron) with a healthy combination of mandatory NPIs aiming for low-case numbers while a working vaccine is developed and coverage is insufficient [25, 48] and by letting individuals decide on their own when the roll-out is complete. However, the core problem remains latent; wealthy countries concentrate resources while some countries cannot afford enough vaccines to protect even their population at risk [59]. As the latter countries are forced into accepting high-case numbers in order to keep their economies running, there are increased risks of breeding variants that could escape natural or vaccine-elicited protection [60]. Therefore, vaccine policy planning from an international perspective is critical for a smooth transition to SARS-CoV-2 endemicity.
Modeling the introduction and spread of different SARS-CoV-2 variants in a population is challenging. At the very least, modeling these dynamics would require having separate compartments for all the disease states of all circulating variants, disproportionally increasing the complexity of our model. In our approach, we take advantage of the extensive immune escape of the Omicron VOC to natural and vaccine-elicited neutralization [29, 31, 32, 45, 47], and assume that the replacement of Delta VOC occures very quickly (i.e., basically instantaneously) in mid-January. This simplification is not too distant from reality; replacement of Delta and other predominant sublineages for Omicron took only a few weeks in several countries [61]. For the spread of Omicron, we use the same basic reproduction number as for Delta but instead consider most individuals previously immunized to have lost protection against infection, i.e., they are moved to the susceptible pool (Methods for details). Thereby, we can capture the explosive spread of Omicron VOC without increasing the base transmissibility. We furthermore include that those people having received a booster vaccine maintain some protection against infection with Omicron, which, however, also wanes. These assumptions are consistent with a large Danish cohort of households, where the secondary attack rate among unvaccinated was slightly higher for Delta infections than for Omicron [47], and with extensive experimental and observational studies [32, 38, 62, 63]. Despite the approximation we did for the transition to the Omicron variant, the mid- and long-term dynamics of the Omicron VOC should be reflected well.
In our work, the level of mandatory NPIs dictates the minimum and maximum level of voluntary health-protective behavior that individuals may adapt. For each scenario, we assume one specific, static level of mandatory NPIs, which best resembles real-world observations on compulsory measures aiming to reduce the probability of contagion (i.e., mask-wearing mandates, immunity passports, meeting restrictions, among others) and testing policy (as described in Methods). However, this static level can lead to unrealistically high waves of incidence and ICU occupancy, which 1) have not been seen so far and 2) would undoubtedly trigger the implementation of additional restrictions to prevent a major collapse in the health system. Nonetheless, we decided to incorporate this static mandatory NPI level because it illustrates a worst-case trajectory of each scenario. Besides, due to pandemic fatigue [64], we would expect the effectiveness of interventions and thus the imposed change in health-protective behavior in the different mandatory NPI scenarios to decay over time.
In summary, the way governments approach a pandemic situation when vaccines are available will shape long-term transmission dynamics by influencing the magnitude of information-behavior feedback loops. We show that the latter play a major role during the transition from epidemicity to endemicity. Thus most importantly, the challenge for authorities is to find ways to engage individuals with vaccination programs and health-protective behavior without requiring high case numbers for that. Here, clear communication and trust continues to be essential [65].
4 Methods
4.1 Model Overview
We use an age-stratified compartmental model with compartments for susceptible-exposed-infected-recovered (SEIR) as well as for fatalities (D), receiving treatment in an ICU (ICU), and vaccination (first time and booster vaccines) (V) (Supplementary Figure S1). We also include waning immunity and seasonality effects (Figures 4, 5B). To account for behavioral change induced by perceived risk of infection, we include a feedback loop between ICU occupancy, voluntary health-protective behavior and willingness to receive vaccination (Figure 2 and Supplementary Material). Explicitly, we assume that increases in ICU occupancy 1) decrease the contact rates among the population and thus the spreading rate of COVID-19 [4–7], and 2) increase vaccine acceptance among hesitant individuals [4, 8]. For the first feedback loop (voluntary health protective behavior), we assume that individuals adapt their contacts in different contexts depending on the risk they have perceived recently. The level of potentially contagious contacts is multiplied by a factor k that decreases with ICU occupancy between the minimum and maximum allowed by current mandatory NPIs (Figure 2C). Regarding the second feedback loop (related to vaccine uptake), we assume that a fraction of the population will always accept a vaccination offer, despite current ICU occupancy. From this minimum onward, vaccination willingness monotonically increases with ICU occupancy and saturates towards a maximum, accounting for a fraction of the population that will never accept the vaccine (Figure 2E). This means that we assume that there is a fraction in the population that is certainly not able or willing to be vaccinated. Given a fraction of people willing to be vaccinated, we determine the speed of the vaccination program using a linearly increasing function (Figure 2F). We model these two feedback loops to act on different timescales, as individuals can, e.g., decrease the number of contacts and contact intensity on a daily basis, while getting vaccinated takes longer. To capture this, we explicitly include memory kernels accounting for how individuals subjectively weigh events happening on different timescales when forming their perception of risk [21].
4.2 Memory on Perceived Risk
We assume that perceived risk regarding the disease depends on information about ICU occupancy that reaches individuals via media or affected social contacts. This perception of risk builds over time; people are not only aware of the occupancy numbers at the present moment but also of those in the recent past. To incorporate this into our model, we calculate the convolution of the ICU occupancy with a Gamma distribution (Supplementary Figure S2, Supplementary Information), effectively “weighting” the ICU occupancy numbers with their recency into a variable of risk perception which we call HR. As a result, ICU occupancy numbers from a few days ago weigh more in people’s memory and thus influence voluntary health-protective behavior at the present moment more than ICU occupancy that lies further in the past. We use this concept of ICU occupancy “with memory” to design the functions of the feedback loops (Figures 2B,C,E,F). The effect of the parameters chosen for the Gamma distribution on the model results as well as of all other model parameters is quantified in the sensitivity analysis, Supplementary Section S4, Supplementary Information.
4.3 NPI- and Risk-Induced Change in Health-Protective Behavior
When analyzing the joint effect of mandatory NPIs and voluntary measures to mitigate the spread of COVID-19, we find a strong overlap between them; mandatory NPIs limit the range of the measures that individuals could voluntarily take to protect themselves and their loved ones. For example, when large private gatherings are officially forbidden, individuals cannot voluntarily choose not to meet. Additionally, when the engagement of the population in voluntary protective measures is very large, certain mandatory NPIs would not be required. We model the combined effect of mandatory NPIs and voluntary adoption of health-protective behavior as a function kNPI, self (HR). Using the baseline of mandatory NPIs as an input, this function calculates the level of voluntary preventive action in dependence of the perceived risk HR. To be precise, the value of kNPI, self (HR) ∈ [0, 1] represents the level to which (potentially contagious) contacts of an average individual are reduced (Figure 2C), a factor that is multiplied onto the entries of a contact matrix separated by contexts (Supplementary Figure S3, Supplementary Information). For example, adaption of voluntary mask-wearing or a direct reduction of gatherings decreases the level of potentially contagious contacts and, thereby, kNPI, self (HR). Furthermore, we distinguish between contacts made at home, in schools, in workplaces or during communal activities. We weight all the interactions with different with that act on contextual contact matrices , see Supplementary Section S1.2 and Figure 1.
Inspired by the COSMO survey data [4] (Figure 2B), we suggest the following shape for : The level of (potentially) contagious contacts decreases linearly upon increases in the ICU-mediated perception of risk HR below a threshold HR = Hmax, from which point on no further reduction is possible (Figure 2C). This might represent 1) a fraction of the population agnostic to measures or unwilling to comply, or 2) limitations of voluntary preventive action imposed by practical constraints related to the current level of imposed restrictions, for example, having to make contacts in one’s own household or having to go to work or school. We implement as a softplus function, having a differentiable transition at Hmax. Each function (for each scenario) is defined by 3 parameters Hmax, , and . Hmax = 37 is obtained by the fit to the COSMO data shown in Figure 2 (black line) and used for the two other fits shown in Figure 2 (red and yellow lines) as well as for the behavior parametrizations for the different scenarios (Supplementary Figure S3, Supplementary Information).
4.4 Different Mandatory NPI Scenarios
We choose to simulate five different scenarios, each having a different level of overall stringency. In the following we briefly describe the scenarios:
Scenario 1 (“Freedom day”): All mandatory restrictions are lifted, resulting in a factor of . However, if ICU occupancy increases, we leave room for individuals’ voluntary action based on perceived risk to reduce viral transmission: . We assume that communal activities and workplaces leave more room for voluntary preventive action than households and schools because of the possibility of working from home, avoiding non-essential gatherings etc. This difference is depicted in Supplementary Figure S3.
Scenario 2 (Moderate NPIs A): Easy-to-follow measures are kept in place and potentially contagious contacts at school are reduced to .
Scenario 3 (Moderate NPIs B): Further measures at work (e.g., home office or testing) reduce .
Scenario 4 (Moderate NPIs C): Further reduction in potentially contagious school contacts and restrictions affecting communal contacts reduce and .
Scenario 5 (Strong NPIs): Communal activities are further reduced to .
Table 1 lists all values for the different scenarios and contexts of interaction between individuals. The reduction of household contacts is assumed to remain the same for all scenarios. Note that, as the stringency of measures increases, room for voluntary adoption of health-protective behavior usually decreases: To give an example, without mandatory measures the level of contact reduction in communal activities lies in the range 1−0.6, whereas in a scenario with strong mandatory NPIs it lies in the range 0.2−0.1. The difference between the two bounds effectively measures the room for voluntary actions (0.4 for freedom day vs. 0.1 for strong NPIs). An exception are school contacts in which moderate restriction scenarios (2 and 3) display a wider range of possible voluntary action than the freedom day scenario. As health-protective behavior among children could be encouraged but not imposed, their adherence to rules constitutes a voluntary act.
TABLE 1
| Sc. | Name | Description of measures | kHouseholds | kSchools | kWorkplaces | kCommunities |
|---|---|---|---|---|---|---|
| 1 | “freedom day” | no mandatory measures | 0.8–1 | 0.8–1 | 0.6–1 | 0.6–1 |
| 2 | moderate NPIs A | increased stringency affecting risk of transmission in schools | 0.8–1 | 0.25–0.5 | 0.5–0.9 | 0.5–0.9 |
| 3 | moderate NPIs B | mild NPIs + reduction of transmission at workplaces | 0.8–1 | 0.25–0.5 | 0.25–0.5 | 0.5–0.9 |
| 4 | moderate NPIs C | moderate NPIs + enforcement of restrictions in communal activities | 0.8–1 | 0.1–0.25 | 0.25–0.5 | 0.25–0.5 |
| 5 | strong NPIs | strong NPIs + further restrictions wherever possible | 0.8–1 | 0.1–0.25 | 0.25–0.5 | 0.1–0.2 |
Different scenarios of mandatory NPIs. Listed are descriptions of the general measures imposed in each scenario as well as the input parameters to the function that modulates the spread. The parameters act as multiplicative factors onto infection terms in our model, thus high parameter values (close to 1) translate to little reduction in infections and low parameters (close to 0) translate to strong reductions in infections. For each cell, the first parameter translates to a reduction at high ICUs and the second parameter to the corresponding reduction at empty ICUs , between which we linearly interpolate (Supplementary Figure S3).
4.5 Modeling the Introduction and Spread of the Omicron VOC
Modeling the introduction and spread of the Omicron VOC requires modifications to the model compartments, transition rates, and parameters. In particular, these modifications allow us to explore the effects of Omicron’s 1) extensive immune escape and 2) potential reduced risk for severe course of the disease. We implemented the introduction of Omicron VOC as a total replacement of the previously dominating Delta VOC on 15 Jan 2022. At that moment, we rearrange the distribution of individuals between the “waned” and “immune” compartments, increase the rate of waning immunity to account for Omicron’s immune escape, and reduce the probability of having a severe course. Explicitly, before the introduction of the Omicron VOC, the immune population is tracked in additional pseudo-compartments Vo, Ro, Rv,o with a faster waning rate. In that way, there are always less individuals in Vo, Ro, Rv,o than in V, R, Rv. At the time of variant replacement, V − Vo, R − Ro, Rv − Rv,o individuals are moved from the vaccinated and recovered compartments to the respective waned compartments; individuals previously protected against Delta would now be susceptible to Omicron. We model booster-vaccination protection against infection following a leaky scheme, thus boostered individuals have a probability of η of being entirely protected. With probability 1 − η, individuals remain in their current compartment but are tracked as if the vaccine had worked successfully.
Statements
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Source code for data generation and analysis is available online on GitHub https://github.com/Priesemann-Group/covid19_infoXpand_feedbackloop. Further inquiries can be directed to the corresponding author.
Author contributions
Conceptualization: PD, JW, SC, SB, JD, VP. Methodology: PD, JW, SC, SB, SM, EI, VP. Software: PD, JW. Formal analysis: PD, JW, SC, SB. Writing—Original Draft: all authors. Writing—Review and Editing: all authors. Visualization: PD, JW, SC. Supervision: SC, MK, MM, KN, AC, VP.
Funding
Open Access publication has been enabled by the Max-Planck-Society. Authors with affiliation “1” acknowledge support from the Max-Planck-Society. SC acknowledges support from the Centre for Biotechnology and Bioengineering—CeBiB (PIA project FB0001, ANID, Chile). SB and SM were financially supported by the German Federal Ministry of Education and Research (BMBF) as part of the Network University Medicine (NUM), project egePan, funding code: 01KX2021. AC recieved funding from the Digital Society research program funded by the Ministry of Culture and Science of the German State of North Rhine-Westphalia. MK acknowledges support from the Netherlands Organisation for Health Research and Development (ZonMw), funding code: 91216062, and from the European Union’s Horizon 2020 research and innovation program under grant agreement No 101003480 (Project CORESMA). KN acknowledges support by the German Federal Ministry of Education and Research (BMBF) for the MODUS-COVID project, funding code: 01KX2022A.
Acknowledgments
We thank Prof. Dr. Cornelia Betsch, her colleagues and her team for publicly sharing survey data of the COVID-19 Snapshot Monitoring (COSMO) study, which enabled a quantitative approach otherwise not possible.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The handling editor declared a past co-authorship with several of the authors EI, SB, SM, AV, MK, VP.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphy.2022.842180/full#supplementary-material
References
1.
Casero-RipollesA. Impact of COVID-19 on the media System. Communicative and Democratic Consequences of News Consumption during the Outbreak. Epi (2020) 29. 10.3145/epi.2020.mar.23
2.
KimHKAhnJAtkinsonLKahlorLA. Effects of COVID-19 Misinformation on Information Seeking, Avoidance, and Processing: A Multicountry Comparative Study. Sci Commun (2020) 42:586–615. 10.1177/1075547020959670
3.
FerrerRAKleinWM. Risk Perceptions and Health Behavior. Curr Opin Psychol (2015) 5:85–9. 10.1016/j.copsyc.2015.03.012
4.
BetschCWielerLHHabersaatK. Monitoring Behavioural Insights Related to Covid-19. The Lancet (2020) 395:1255–6. 10.1016/s0140-6736(20)30729-7
5.
ImbrianoGLarsenEMMackinDMAnAKLuhmannCCMohantyAet alOnline Survey of the Impact of Covid-19 Risk and Cost Estimates on Worry and Health Behavior Compliance in Young Adults. Front Public Health (2021) 9:157. 10.3389/fpubh.2021.612725
6.
PerrottaDGrowARampazzoFCimentadaJDel FavaEGil-ClavelSet alBehaviours and Attitudes in Response to the Covid-19 Pandemic: Insights from a Cross-National Facebook Survey. EPJ Data Sci (2021) 10:17–3. 10.1140/epjds/s13688-021-00270-1
7.
DruckmanJNKlarSKrupnikovYLevenduskyMRyanJB. Affective Polarization, Local Contexts and Public Opinion in america. Nat Hum Behav (2021) 5:28–38. 10.1038/s41562-020-01012-5
8.
SalaliGDUysalMS. Effective Incentives for Increasing Covid-19 Vaccine Uptake. Psychol Med (2021) 1–3. 10.1017/s0033291721004013
9.
GavenčiakTMonradJTLeechGSharmaMMindermannSBraunerJMet al Seasonal Variation in Sars-Cov-2 Transmission in Temperate Climates. medRxiv (2021). 10.1101/2021.06.10.21258647
10.
MoriyamaMHugentoblerWJIwasakiA. Seasonality of Respiratory Viral Infections. Annu Rev Virol (2020) 7:83–101. 10.1146/annurev-virology-012420-022445
11.
SajadiMMHabibzadehPVintzileosAShokouhiSMiralles-WilhelmFAmorosoA. Temperature and Latitude Analysis to Predict Potential Spread and Seasonality for COVID-19. Available at SSRN 3550308 (2020).
12.
MistryDLitvinovaMy PionttiAPChinazziMFumanelliLGomesMFet alInferring High-Resolution Human Mixing Patterns for Disease Modeling. Nat Commun (2021) 12:1–12. 10.1038/s41467-020-20544-y
13.
OlivieriAPalùGSebastianiG. Covid-19 Cumulative Incidence, Intensive Care, and Mortality in Italian Regions Compared to Selected European Countries. Int J Infect Dis (2021) 102:363–8. 10.1016/j.ijid.2020.10.070
14.
BravataDMPerkinsAJMyersLJArlingGZhangYZillichAJet alAssociation of Intensive Care Unit Patient Load and Demand with Mortality Rates in Us Department of Veterans Affairs Hospitals during the Covid-19 Pandemic. JAMA Netw Open (2021) 4:e2034266. 10.1001/jamanetworkopen.2020.34266
15.
LindenMMohrSBDehningJMohringJMeyer-HermannMPigeotIet alCase Numbers beyond Contact Tracing Capacity Are Endangering the Containment of COVID-19. Dtsch Arztebl Int (2020) 117:790–1. 10.3238/arztebl.2020.0790
16.
EpsteinJMParkerJCummingsDHammondRA. Coupled Contagion Dynamics of Fear and Disease: Mathematical and Computational Explorations. PLoS One (2008) 3:e3955. 10.1371/journal.pone.0003955
17.
EpsteinJMHatnaECrodelleJ. Triple Contagion: a Two-Fears Epidemic Model. J R Soc Interf (2021) 18:20210186. 10.1098/rsif.2021.0186
18.
BauchCT. Imitation Dynamics Predict Vaccinating Behaviour. Proc R Soc B (2005) 272:1669–75. 10.1098/rspb.2005.3153
19.
d’OnofrioAManfrediP. Information-related Changes in Contact Patterns May Trigger Oscillations in the Endemic Prevalence of Infectious Diseases. J Theor Biol (2009) 256:473–8.
20.
d’OnofrioAManfrediPSalinelliE. Vaccinating Behaviour, Information, and the Dynamics of Sir Vaccine Preventable Diseases. Theor Popul Biol (2007) 71:301–17.
21.
ZaubermanGKimBKMalkocSABettmanJR. Discounting Time and Time Discounting: Subjective Time Perception and Intertemporal Preferences. J Marketing Res (2009) 46:543–56. 10.1509/jmkr.46.4.543
22.
BauchCTBhattacharyyaS. Evolutionary Game Theory and Social Learning Can Determine How Vaccine Scares Unfold. Plos Comput Biol (2012) 8:e1002452. 10.1371/journal.pcbi.1002452
23.
TkachenkoAVMaslovSWangTElbanaAWongGNGoldenfeldN. Stochastic Social Behavior Coupled to Covid-19 Dynamics Leads to Waves, Plateaus, and an Endemic State. eLife (2021) 10:e68341. 10.7554/eLife.68341
24.
DankulovMMTadicBMelnikR. Worldwide Clustering and Infection Cycles as Universal Features of Multiscale Stochastic Processes in the Sars-Cov-2 Pandemic. medRxiv (2021). 10.1101/2021.12.20.21268095
25.
BauerSContrerasSDehningJLindenMIftekharEMohrSBet alRelaxing Restrictions at the Pace of Vaccination Increases freedom and Guards against Further Covid-19 Waves. Plos Comput Biol (2021) 17:e1009288. 10.1371/journal.pcbi.1009288
26.
BrownELEssigmannHT. Original Antigenic Sin: the Downside of Immunological Memory and Implications for Covid-19. Msphere (2021) 6:e00056–21. 10.1128/mSphere.00056-21
27.
GomezGBMahéCChavesSS. Uncertain Effects of the Pandemic on Respiratory Viruses. Science (2021) 372:1043–4. 10.1126/science.abh3986
28.
Sanz-MuñozITamames-GómezSCastrodeza-SanzJEiros-BouzaJMde Lejarazu-LeonardoRO. Social Distancing, Lockdown and the Wide Use of Mask; a Magic Solution or a Double-Edged Sword for Respiratory Viruses Epidemiology?Vaccines (2021) 9:595. 10.3390/vaccines9060595
29.
VianaRMoyoSAmoakoDGTegallyHScheepersCLessellsRJet alRapid Epidemic Expansion of the Sars-Cov-2 Omicron Variant in Southern Africa. Nature (2021). 10.1038/d41586-021-03832-5
30.
PulliamJRvan SchalkwykCGovenderNvon GottbergACohenCGroomeMJet alIncreased Risk of Sars-Cov-2 Reinfection Associated with Emergence of the Omicron Variant in south africa. medRxiv (2021). 10.1101/2021.11.11.21266068
31.
FergusonNGhaniACoriAHoganAHinsleyWVolzE. Report 49: Growth, Population Distribution and Immune Escape of the Omicron in england. Imperial College London (2021). 10.25561/93038
32.
CeleSJacksonLKhouryDSKhanKMoyo-GweteTTegallyHet alOmicron Extensively but Incompletely Escapes Pfizer Bnt162b2 Neutralization. Nature (2021). 10.1038/s41586-021-04387-1
33.
WilhelmAWideraMGrikscheitKToptanTSchenkBPallasCet alReduced Neutralization of Sars-Cov-2 Omicron Variant by Vaccine Sera and Monoclonal Antibodies. medRxiv (2021). 10.1101/2021.12.07.21267432
34.
CameroniEBowenJERosenLESalibaCZepedaSKCulapKet alBroadly Neutralizing Antibodies Overcome Sars-Cov-2 Omicron Antigenic Shift. Nature (2021) 1–9. 10.1038/s41586-021-04386-2
35.
RoesslerARieplerLBanteDvon LaerDKimpelJ. Sars-cov-2 B. 1.1. 529 Variant (Omicron) Evades Neutralization by Sera from Vaccinated and Convalescent Individuals. medRxiv (2021). 10.1101/2021.12.08.21267491
36.
HoffmannMKrügerNSchulzSCossmannARochaCKempfAet alThe Omicron Variant Is Highly Resistant against Antibody-Mediated Neutralization–Implications for Control of the Covid-19 Pandemic. Cell (2021). 10.1016/j.cell.2021.12.032
37.
GardnerBJKilpatrickAM. Estimates of Reduced Vaccine Effectiveness against Hospitalization, Infection, Transmission and Symptomatic Disease of a New Sars-Cov-2 Variant, Omicron (B. 1.1. 529), Using Neutralizing Antibody Titers. medRxiv (2021). 10.1101/2021.12.10.21267594
38.
Pérez-ThenELucasCMonteiroVSMiricMBracheVCochonLet alImmunogenicity of Heterologous Bnt162b2 Booster in Fully Vaccinated Individuals with Coronavac against Sars-Cov-2 Variants delta and Omicron: The dominican republic Experience. medRxiv (2021). 10.1101/2021.12.27.21268459
39.
GruellHVanshyllaKTober-LauPHillusDSchommersPLehmannCet alMrna Booster Immunization Elicits Potent Neutralizing Serum Activity against the Sars-Cov-2 Omicron Variant. Nat. Med. (2022). 10.1038/s41591-021-01676-0
40.
KuhlmannCMayerCKClaassenMMapongaTGSutherlandADSulimanTet alBreakthrough Infections with Sars-Cov-2 Omicron Variant Despite Booster Dose of Mrna Vaccine. Available at SSRN 3981711 (2021).
41.
NemetIKlikerLLustigYZuckermanNErsterOCohenCet alThird Bnt162b2 Vaccination Neutralization of SARS-CoV-2 Omicron Infection. New Engl J Med (2021). 10.1056/nejmc2119358
42.
BasileKRockettRJMcPhieKFennellMJohnson-MackinnonJAgiusJet alImproved Neutralization of the Sars-Cov-2 Omicron Variant after Pfizer-Biontech Bnt162b2 Covid-19 Vaccine Boosting. bioRxiv (2021). 10.1101/2021.12.12.472252
43.
Garcia-BeltranWFSt DenisKJHoelzemerALamECNitidoADSheehanMLet almRNA-based COVID-19 Vaccine Boosters Induce Neutralizing Immunity Against SARS-CoV-2 Omicron Variant. Cell (2022). 10.1016/j.cell.2021.12.033
44.
AndrewsNStoweJKirsebomFToffaSRickeardTGallagherEet alEffectiveness of Covid-19 Vaccines against the Omicron (B. 1.1. 529) Variant of Concern. medRxiv (2021). 10.1101/2021.12.14.21267615
45.
TorjesenI. Covid Restrictions Tighten as Omicron Cases Double Every Two to Three Days. BMJ (2021) 375. 10.1136/bmj.n3051
46.
BarnardRCDaviesNGPearsonCAJitMJohnWEdmundsWJ. Projected epidemiological consequences of the Omicron SARS-CoV-2 variant in England. medRxiv (2021). 10.1101/2021.12.15.21267858
47.
LyngseFPMortensenLHDenwoodMJChristiansenLEMøllerCHSkovRLet alSars-cov-2 Omicron Voc Transmission in Danish Households. medRxiv (2021). 10.1101/2021.12.27.21268278
48.
ContrerasSDehningJMohrSBBauerSSpitznerFPPriesemannV. Low Case Numbers Enable Long-Term Stable Pandemic Control without Lockdowns. Sci Adv (2021) 7:eabg2243. 10.1126/sciadv.abg2243
49.
Oliu-BartonMPradelskiBSRAghionPArtusPKickbuschILazarusJVet alSARS-CoV-2 Elimination, Not Mitigation, Creates Best Outcomes for Health, the Economy, and Civil Liberties. The Lancet (2021) 397:2234–6. 10.1016/s0140-6736(21)00978-8
50.
CzypionkaTIftekharENPrainsackBPriesemannVBauerSCalero ValdezAet alThe Benefits, Costs and Feasibility of a Low Incidence Covid-19 Strategy. The Lancet Reg Health - Europe (2022) 13:100294. 10.1016/j.lanepe.2021.100294
51.
FunkSSalathéMJansenVAA. Modelling the Influence of Human Behaviour on the Spread of Infectious Diseases: a Review. J R Soc Interf (2010) 7:1247–56. 10.1098/rsif.2010.0142
52.
VerelstFWillemLBeutelsP. Behavioural Change Models for Infectious Disease Transmission: a Systematic Review (2010-2015). J R Soc Interf (2016) 13:20160820. 10.1098/rsif.2016.0820
53.
WestonDHauckKAmlôtR. Infection Prevention Behaviour and Infectious Disease Modelling: a Review of the Literature and Recommendations for the Future. BMC public health (2018) 18:336–16. 10.1186/s12889-018-5223-1
54.
BedsonJSkripLAPediDAbramowitzSCarterSJallohMFet alA Review and Agenda for Integrated Disease Models Including Social and Behavioural Factors. Nat Hum Behav (2021) 1–13. 10.1038/s41562-021-01136-2
55.
BuonomoBDella MarcaR. Effects of Information-Induced Behavioural Changes during the Covid-19 Lockdowns: the Case of italy. R Soc Open Sci (2020) 7:201635. 10.1098/rsos.201635
56.
PriesemannVBallingRBauerSBeutelsPValdezACCuschieriSet alTowards a European Strategy to Address the Covid-19 Pandemic. The Lancet (2021) 398:838–9. 10.1016/s0140-6736(21)01808-0
57.
CinelliMQuattrociocchiWGaleazziAValensiseCMBrugnoliESchmidtALet alThe Covid-19 Social media Infodemic. Sci Rep (2020) 10:16598–10. 10.1038/s41598-020-73510-5
58.
BanerjeeDMeenaK. Covid-19 as an “Infodemic” in Public Health: Critical Role of the Social media. Front Public Health (2021) 9:231. 10.3389/fpubh.2021.610623
59.
ContrerasSOlivera-NappaÁPriesemannV. Rethinking Covid-19 Vaccine Allocation: it Is Time to Care about Our Neighbours. The Lancet Reg Health–Europe (2022) 12, 100277. 10.1016/j.lanepe.2021.100277
60.
ThompsonRNHillEMGogJR. SARS-CoV-2 Incidence and Vaccine Escape. Lancet Infect Dis (2021) 21:913–4. 10.1016/s1473-3099(21)00202-4
61.
RitchieHMathieuERodés-GuiraoLAppelCGiattinoCOrtiz-OspinaEet alCoronavirus Pandemic (Covid-19). Our World in Data (2020). Available at: Https://ourworldindata.org/coronavirus.
62.
VianaJvan DorpCHNunesAGomesMCvan BovenMKretzschmarMEet alControlling the Pandemic during the SARS-CoV-2 Vaccination Rollout. Nat Commun (2021) 12:3674–15. 10.1038/s41467-021-23938-8
63.
LuLMokBW-YChenL-LChanJM-CTsangOT-YLamBH-Set alNeutralization of SARS-CoV-2 Omicron Variant by Sera from BNT162b2 or Coronavac Vaccine Recipients. Clinical Infectious Diseases (2021). 10.1093/cid/ciab1041
64.
PetherickAGoldszmidtRAndradeEBFurstRHaleTPottAet alA Worldwide Assessment of Changes in Adherence to Covid-19 Protective Behaviours and Hypothesized Pandemic Fatigue. Nat Hum Behav (2021) 5:1145–60. 10.1038/s41562-021-01181-x
65.
IftekharENPriesemannVBallingRBauerSBeutelsPCalero ValdezAet alA Look into the Future of the COVID-19 Pandemic in Europe: an Expert Consultation. Lancet Reg Health - Europe (2021) 8:100185. 10.1016/j.lanepe.2021.100185
Summary
Keywords
COVID-19, disease modeling, infodemic, human behavior, self-regulation, vaccine hesitancy, health policy and practice, Omicron variant (SARS-CoV-2)
Citation
Dönges P, Wagner J, Contreras S, Iftekhar EN, Bauer S, Mohr SB, Dehning J, Calero Valdez A, Kretzschmar M, Mäs M, Nagel K and Priesemann V (2022) Interplay Between Risk Perception, Behavior, and COVID-19 Spread. Front. Phys. 10:842180. doi: 10.3389/fphy.2022.842180
Received
23 December 2021
Accepted
20 January 2022
Published
15 February 2022
Volume
10 - 2022
Edited by
Matjaž Perc, University of Maribor, Slovenia
Reviewed by
Gui-Quan Sun, North University of China, China
Bosiljka Tadic, Institut Jožef Stefan (IJS), Slovenia
Updates
Copyright
© 2022 Dönges, Wagner, Contreras, Iftekhar, Bauer, Mohr, Dehning, Calero Valdez, Kretzschmar, Mäs, Nagel and Priesemann.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Viola Priesemann, viola.priesemann@ds.mpg.de
†These authors have contributed equally to this work
This article was submitted to Social Physics, a section of the journal Frontiers in Physics
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.