- 1Department of Plant Breeding, Swedish University of Agricultural Sciences, Alnarp, Sweden
- 2Department of Plant Protection Biology, Swedish University of Agricultural Sciences, Alnarp, Sweden
Neoplasm formation, a non-meristematic tissue growth on young field pea (Pisum sativum L.) pods is triggered in the absence of UV light and/or in response to oviposition by pea weevil (Bruchus pisorum L.). This trait is expressed in some genotypes [neoplastic (Np) genotypes] of P. sativum and has the capacity to obstruct pea weevil larval entry into developing seeds. In the present study, 26% of the tested accessions depicted the trait when grown under greenhouse conditions. However, UV light inhibits full expression of this trait and subsequently it is inconspicuous at the field level. In order to investigate UV light impact on the expression of neoplasm, particular Np genotypes were subjected to UV lamp light exposure in the greenhouse and sunlight at the field level. Under these different growing conditions, the highest mean percentage of Np pods was in the control chamber in the greenhouse (36%) whereas in single and double UV lamp chambers, the percentage dropped to 10 and 15%, respectively. Furthermore, when the same Np genotypes were grown in the field, the percentage of Np pods dropped significantly (7%). In order to enhance Np expression at the field level, intercropping of Np genotypes with sorghum was investigated. As result, the percentage of Np pods was threefold in intercropped Np genotypes as compared to those without intercropping. Therefore, intercropping Np genotypes with other crops such as sorghum and maize can facilitate neoplasm formation, which in turn can minimize the success rate of pea weevil larvae entry into developing seeds. Greenhouse artificial infestation experiments showed that pea weevil damage in Np genotypes is lower in comparison to wild type genotypes. Therefore, promoting Np formation under field conditions via intercropping can serve as part of an integrated pea weevil management strategy especially for small scale farming systems.
Introduction
Abiotic factors like light, water and nutrients have a major influence on the phenotype of crops which in turn influences the multi-trophic interactions of the crop with ecological and economic implications (Dicke and Hilker, 2003). Some genotypes of field pea (Pisum sativum ssp. sativum L.) produce neoplasm on its pods when grown under greenhouse conditions. This phenomenon is a non-meristematic tissue growth on the surface of young pods in response to absence of UV light (Nuttall and Lyall, 1964; Dodds and Matthews, 1966). This trait can also be triggered by pea weevil (Bruchus pisorum L.) as a direct response to oviposition (Berdnikov et al., 1992; Doss et al., 2000). However, according to Doss et al. (1995), neoplastic (Np) tissue growth triggered by pea weevil oviposition is morphologically different from that caused by absence of UV light. Neoplasm in field pea is a result of mutation of a gene at Np (neoplasm) locus, in which a mutant allele (Np) is dominant over a wild type allele (np) (Nuttall and Lyall, 1964). Despite the fact that the expression of this trait is controlled by the dominant (Np) allele, its penetrance is influenced by the genotype (homozygosity: Np/Np vs. heterozygosity: Np/np), and the level of UV light intensity or humidity (Nuttall and Lyall, 1964; Burgess and Fleming, 1973; Doss et al., 2000). This trait has also been reported in other Pisum species like P. elatius and P. humile under greenhouse conditions (Dodds and Matthews, 1966).
Organic compounds extracted from pea weevil that were referred to as bruchins were reported to trigger neoplasm formation when applied on young pea pods of particular genotypes (Doss et al., 1995; Oliver et al., 2000; Doss, 2005). Doss (2005) reported that treating Np pea pods with bruchins lead to upregulation of genes that are known to be involved in defense metabolic pathways. In general, most studies conducted to date show that this trait is an induced response triggered by both abiotic and biotic stresses. Comparison of Np and wild type genotypes in their resistance to pea weevil revealed a lower average pea weevil damage in neoplasm producing genotypes under field (Doss et al., 2000) and greenhouse conditions (Teshome et al., 2015), which suggests the importance of this treat in pea weevil managment.
Pea weevil is the major menace in field pea production in Ethiopia and elsewhere (Pesho et al., 1977; Clement et al., 2002; Seyoum et al., 2012). Currently, chemical pesticide spraying and after harvest fumigation are the only options for pea weevil containment (Horne and Bailey, 1991; Baker, 1998). Despite the potential role of neoplasm formation in pea weevil management, little attention has been given to it until now. This is partly due to the fact that the penetrance of the trait under field conditions is inconspicuous and/or inconsistent (Nuttall and Lyall, 1964; Doss et al., 2000). In the present study, the influence of UV light on the expression of neoplasm under greenhouse conditions was studied in parental and F1 hybrids of Np and wild type (non-Np) genotypes. In addition, intercropping Np genotypes was investigated, if the shade provided by intercropping could enhance Np expression in Np genotypes under field conditions. Furthermore, both Np and wild type genotypes were screened for pea weevil resistance under greenhouse conditions.
Materials and Methods
Impact of UV Light on Neoplasm Formation
Neoplasm producing genotypes were identified from various field pea accessions during greenhouse screening experiments for pea weevil resistance in 2012 and 2013 (Teshome et al., 2015). The Np genotypes used in the present study are hereafter referred to as Np genotypes. The Np genotypes used in this study were 32433A, 203084A, 235899A, 237065A, 226037A, 226037B, 226037C, 226037D, and 226037E. Two separate experiments were carried out to study the effect of UV light on neoplasm formation. Primarily, Np genotypes were tested both under greenhouse and field conditions. Additionally, F1 hybrids produced from crosses of wild type genotype pollen recipient and Np pollen donor parents were tested for neoplasm formation under greenhouse conditions.
All plants were grown in 2 l plastic pots in a greenhouse chamber at 22°C and a minimum of 12 h light. Before flowering, plants were moved into chambers (2.54 m2) covered with a light-proof plastic sheet. Five chambers were used for this experiment with all having two cool white Fluorescent lamps, Sylvania Luxline plus 58W. One of the five chambers was used as a control chamber. The remaining four chambers had UV lamps, two of which had a single UV lamp (3 U 15 W UV light bulb) and the remaining two having double UV lamps (2 × 3 U 15 W UV light bulbs) each. The plants in the experimental chambers were exposed to UV light for 12 h from 6:00 pm to 6:00 am. Each genotype was represented by a minimum of three and a maximum of six replicates. Pods were harvested at maturity and individual pods of each genotype were assessed for neoplasm formation. Based on the level of neoplasm formation, pods from each plant were categorized into two different groups, low and high. The low score was given when there was sparse coverage of Np tissue on both sides of the pods. A high score was given when there was a conspicuous neoplasm formation that covered most of the pod. A similar protocol was used for the F1 hybrids as for the parental Np genotypes although they were only tested in double UV lamp and control chambers.
Intercropping of Np Genotypes with Sorghum
Field experiments were carried out at Alnarp, Sweden in 2013 and 2014 to investigate the effect of shading provided by the canopy of sorghum on neoplasm expression. In this experiment, Np genotypes were intercropped with sorghum (Sorghum bicolor L. Moench.). Sorghum and pea genotypes were planted in different rows, in which each pea row had adjacent rows of sorghum on both sides. The distance between pea plants in a row was 5 cm and likewise in-between sorghum plants. The distance between rows was 10 cm. The total area of the plot was 2 m2 with two replications. Sorghum plants were also grown along the borders of the plots. There were five blocks in total with two blocks with intercropping and another two without intercropping and the last block with shading but without intercropping.
Scanning Electron Microscopy (SEM)
Morphological and anatomical characteristics of Np pods, which were harvested at maturity from plants grown in the greenhouse, were examined under SEM. Small pieces of the pods from genotype 226037B were fixed with 2.5% glutaraldehyde in 0.1 M Na-phosphate buffer pH 7.2 overnight at +4°C, washed with the same buffer 3 × 15 min, dehydrated in graded series of ethanol and critical-point dried (CPD 020, Balzers, Lichtenstein). The samples were attached on the sample stubs with double-sided tape and sputter coated with gold and palladium 3:2 mix (JFC-1100, JOEL, Tokyo, Japan), and examined in a SEM (435 VP, LEO Electron Microscopy Ltd, Cambridge, UK) with 10 kV.
Greenhouse Screening of Np and Wild Type Genotypes for Pea Weevil Resistance
Artificial infestation was done in insect rearing cages (60 cm × 60 cm × 120 cm, MegaView Science Co Ltd, Taiwan). The plants were moved into cages when they started to flower. Six plants were placed in each cage and each genotype was intermixed with different genotypes in consecutive experiments. Newly emerged adult pea weevils from seeds of a previous pea weevil screening study were used for artificial infestation. The weevils were kept at 4°C until they were released. In order to balance the sex ratio and ensure successful mating, the sex of the weevils was determined as described by Bousquet (1990) ahead of release. Twenty-five pairs of naive male and female pea weevils were released in each cage as soon as the first flower was detected. Pods were harvested at maturity and stored at room temperature. Three months after harvest, damage assessment was carried out on seeds of each genotype. Percent seed damage (PSD) was calculated based on pea weevil damage symptoms as described in Teshome et al. (2015). All plants used in this experiment were grown in a similar manner as in the UV light experiment.
Data Analysis
All percentages of Np pods and PSD were arcsine transformed to homogenize variances and ensure normal distribution. A one way analysis of variance model was used to compare the proportion of neoplasm formation in Np genotypes when grown under different greenhouse and field conditions. In addition, pair-wise comparisons between the control, i.e., greenhouse normal lamp condition and all other conditions were carried out based on post-hoc t-test with P-values adjusted using the single-step method (Hothorn et al., 2008). A significance level of 5% was used for ANOVA and multiple comparison test. All analysis were carried on R version 3.2.2 (R Core Team, 2015).
Results
Among 19 accessions used for resistance screening against pea weevil in the greenhouse, five accessions showed consistent neoplasm expression (Table 1). Neoplasm formation in these genotypes was clear and distinct (Figure 1A). The scanning electron micrographs also revealed a distinct outgrowth on the outer surface of pods of Np genotypes (Figure 1B). The remaining 15 field pea accessions as well as P. fulvum did not show any Np growth in repeated greenhouse experiments (Table 1).
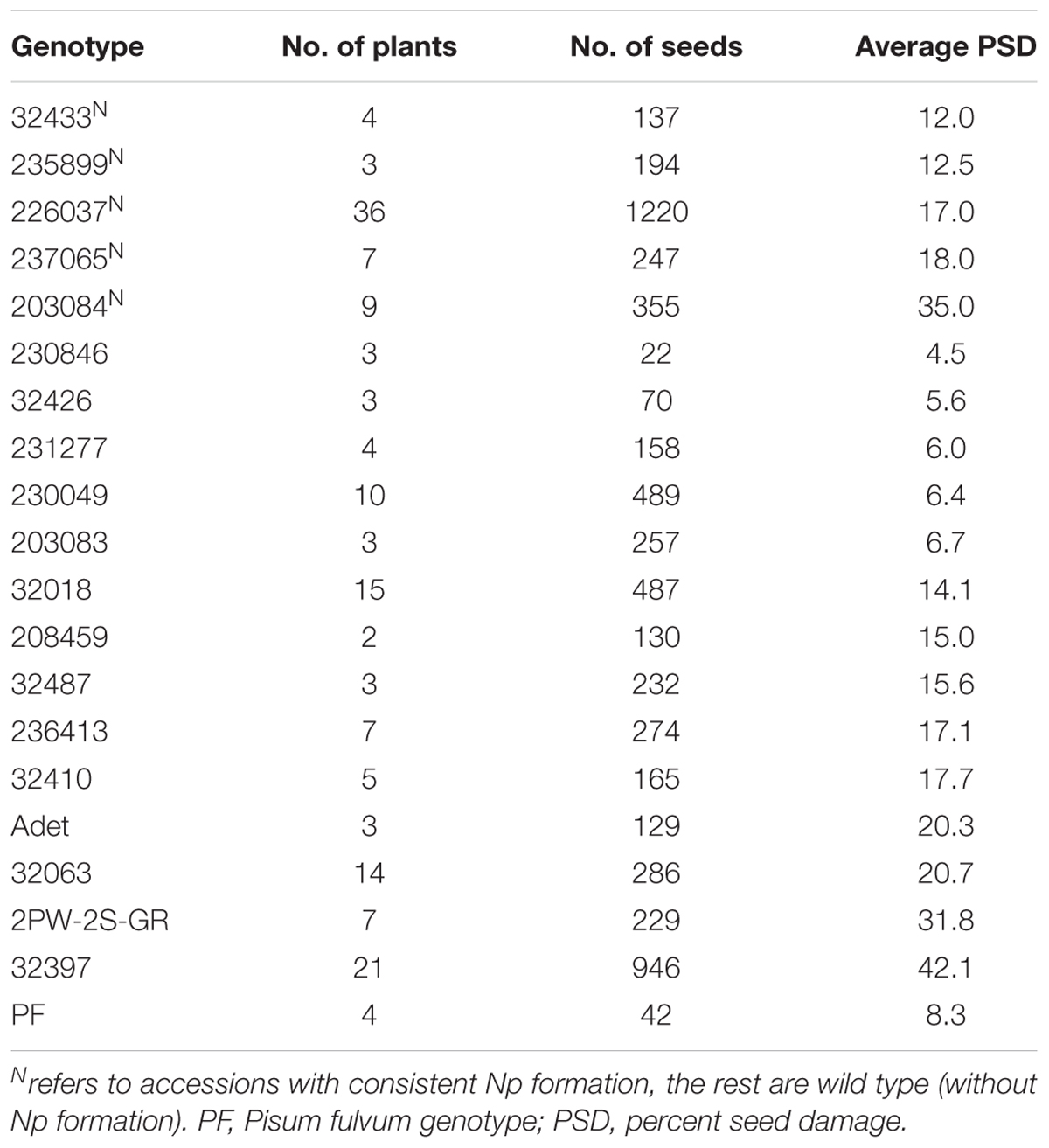
TABLE 1. Comparison of average performance of neoplastic (Np) and non-Np genotypes originating from different accessions against pea weevil attack in greenhouse experiments.
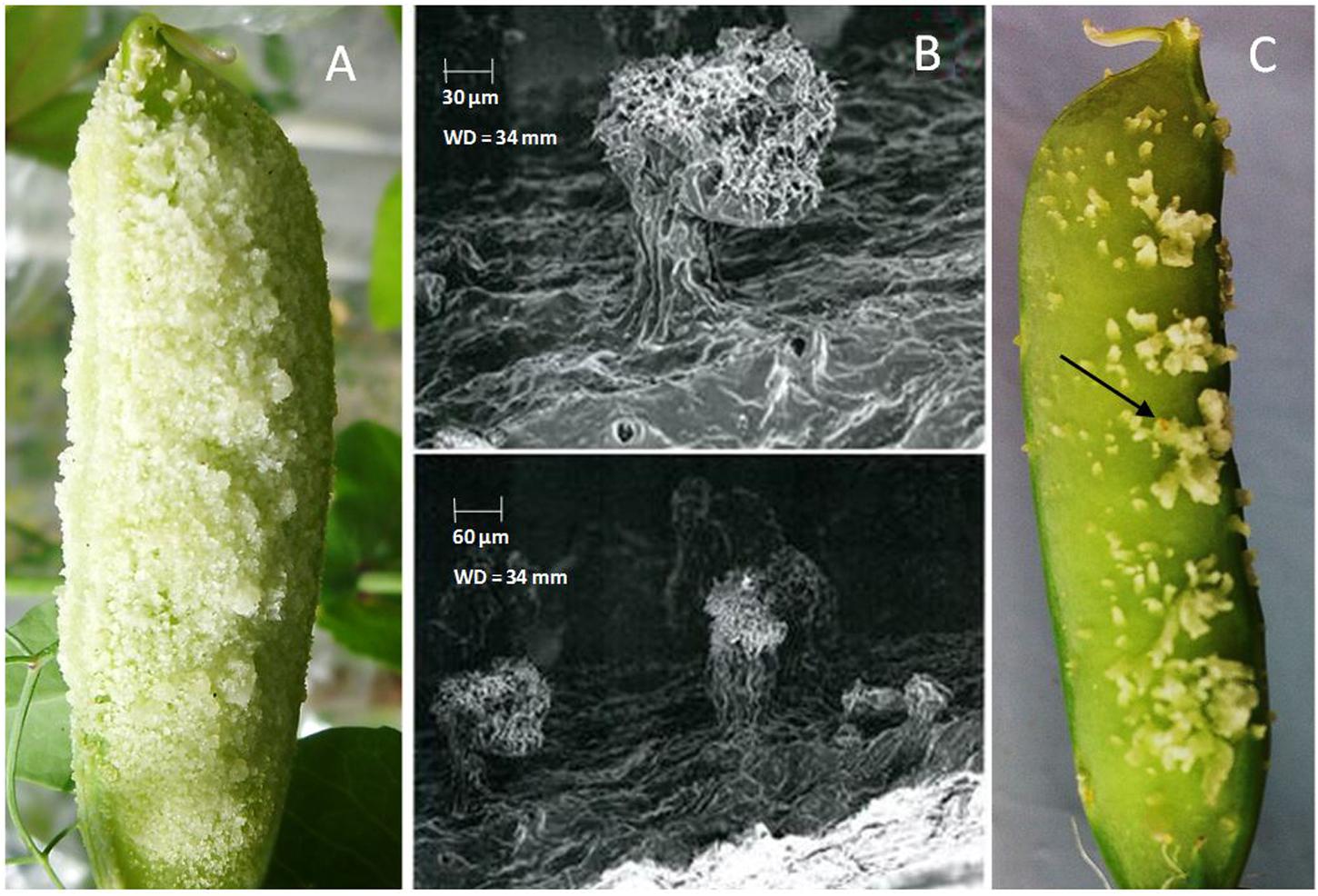
FIGURE 1. (A) Neoplasm formation in genotype 226037B pod in the control chamber (no UV light); (B) scanning electron micrographs of neoplasm formation on genotype 226037B; (C) pea weevil eggs oviposited on neoplastic (Np) pod in greenhouse screening.
Particular Np genotypes that have shown consistent Np formation in repeated greenhouse trials were exposed to UV light to investigate UV influence on neoplasm formation. The highest percentage of Np pods, 36%, was recorded when genotypes were grown in the control chamber under greenhouse conditions and the least, 7%, when the replicates were grown in the field without intercropping. The mean percentage of Np pods in the field with intercropping was threefold of the mean Np pods without intercropping (Table 2). In addition, the median of the percentage of Np pods with intercropping was also higher than the median of the percentages recorded for the single and double UV light exposed chambers under greenhouse conditions (Figure 2).

TABLE 2. Pair-wise comparison of percent neoplasm formation on pods of selected genotypes grown under different greenhouse and field conditions with percent neoplasm formation on pods of same genotypes grown under normal light greenhouse condition (control).
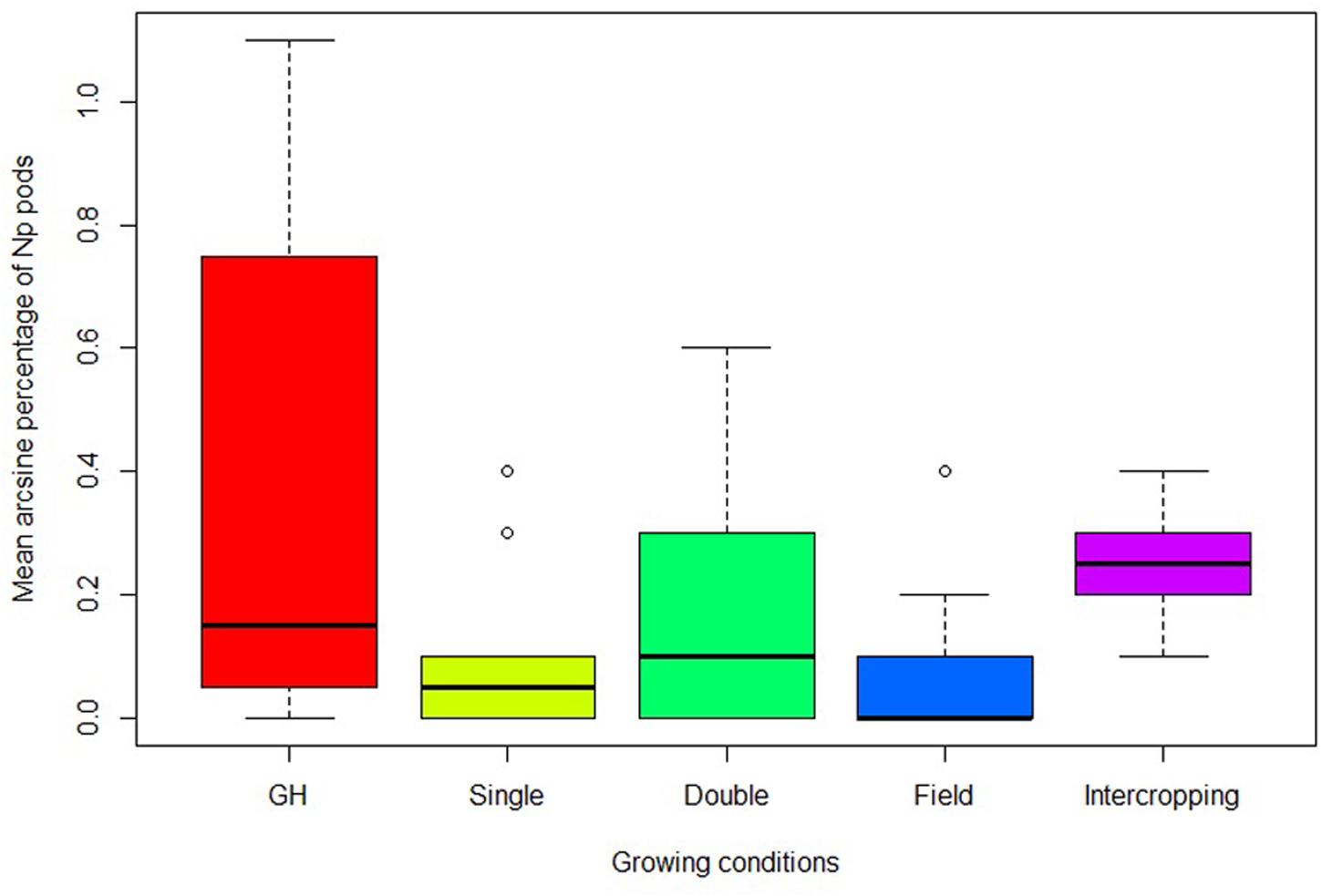
FIGURE 2. Change in proportion of Np pods in different growing conditions in the greenhouse and at the field with and without intercropping. Growing conditions; GH, control chamber; Single, single UV lamp; Double, double UV lamps; Field, field without intercropping; Intercropping, field with intercropping.
The comparison of the mean percentage of Np pods under greenhouse UV light exposure and field conditions revealed a marginally significant difference (P = 0.05). Further post-hoc test revealed that the mean percentage of Np pods without intercropping was significantly different from the control group in the greenhouse (Table 2). The Np genotypes grown under single or double UV lamps in the greenhouse or intercropped in the field were not significantly different from the control group in their mean percentage of Np pods. Under intercropping conditions, three genotypes 235899A, 237065A, and 22603B scored 30% or higher percent of Np pods. Interestingly, 203084A scored the highest percentage of Np pods (42.9%) in the field with intercropping but produced low percentage of Np pods in the control chamber (data not shown).
All F1 hybrids produced by crossing non-Np (used as pollen recipient) and Np (used as pollen donors) produced neoplasm under greenhouse conditions (Figure 3). In both control and UV chambers, the highest percentage of Np pods was recorded for the F1 hybrid 32018-20 × 226037-2S with 100 and 77.8% Np pods, respectively. The least percentage of Np pods was scored for the 32397-6 × 226037D hybrid which was 50% in the control chamber. In most F1 hybrids, the percentage of Np pods decreased significantly when grown under double UV lamp conditions (Figure 3).
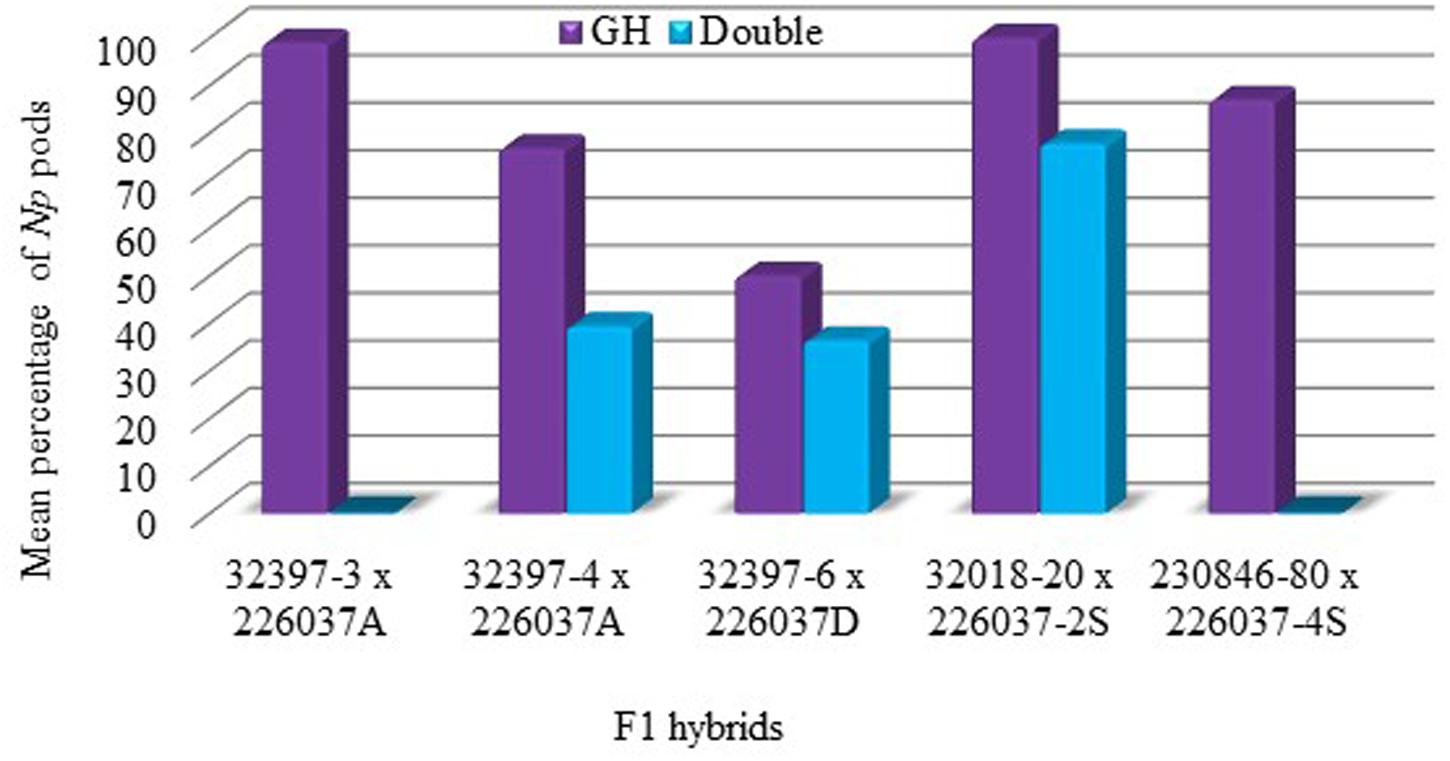
FIGURE 3. Mean percentage of Np pods for F1 hybrids in the greenhouse. Growing conditions; GH, control chamber; Double, double UV lamps.
In general, Np accessions scored low average PSD in comparison to susceptible checks. The least average PSD among Np accessions was recorded for genotype 32433 which was 12% and the highest, 35%, for 203084 (Table 1). Among the wild type accessions, 231277, 32426, and 208459 recorded low PSD whereas 32397 (susceptible check) scored the highest average PSD (42%). After the initial screening that included both Np and wild type accessions, selected Np genotypes, a P. fulvum accession from NordGen and a Np F1 hybrid were tested for pea weevil resistance under greenhouse conditions. The Np genotypes scored less PSD in three consecutive greenhouse experiments with few notable exceptions (Table 4). The PSD of most Np genotypes was lower than the average seed damage all of plants in the same cage. For example, genotype 237065A scored 30% PSD at the first screening despite the mean PSD per cage and percent of plants with infested seeds per cage was 51 and 100%, respectively. Relatively lower PSD values were also observed for this genotype in the second and third round of screenings using seeds from the same generation. The Np F1 hybrid of 32397-3 × 226037A also scored a relatively low PSD in two consecutive experiments. On the contrary, 203084A, which is an Np genotype, scored high PSD values in all experiments. This genotype scored low percentage of Np pods in a control chamber in the greenhouse (Table 3). The P. fulvum genotype scored no seed damage in two consecutive experiments (Table 4). Analysis of variance (ANOVA) of PSD of Np genotypes, F1 hybrids and the P. fulvum line gave a highly significant variation, P = 0.001 (Table 4).
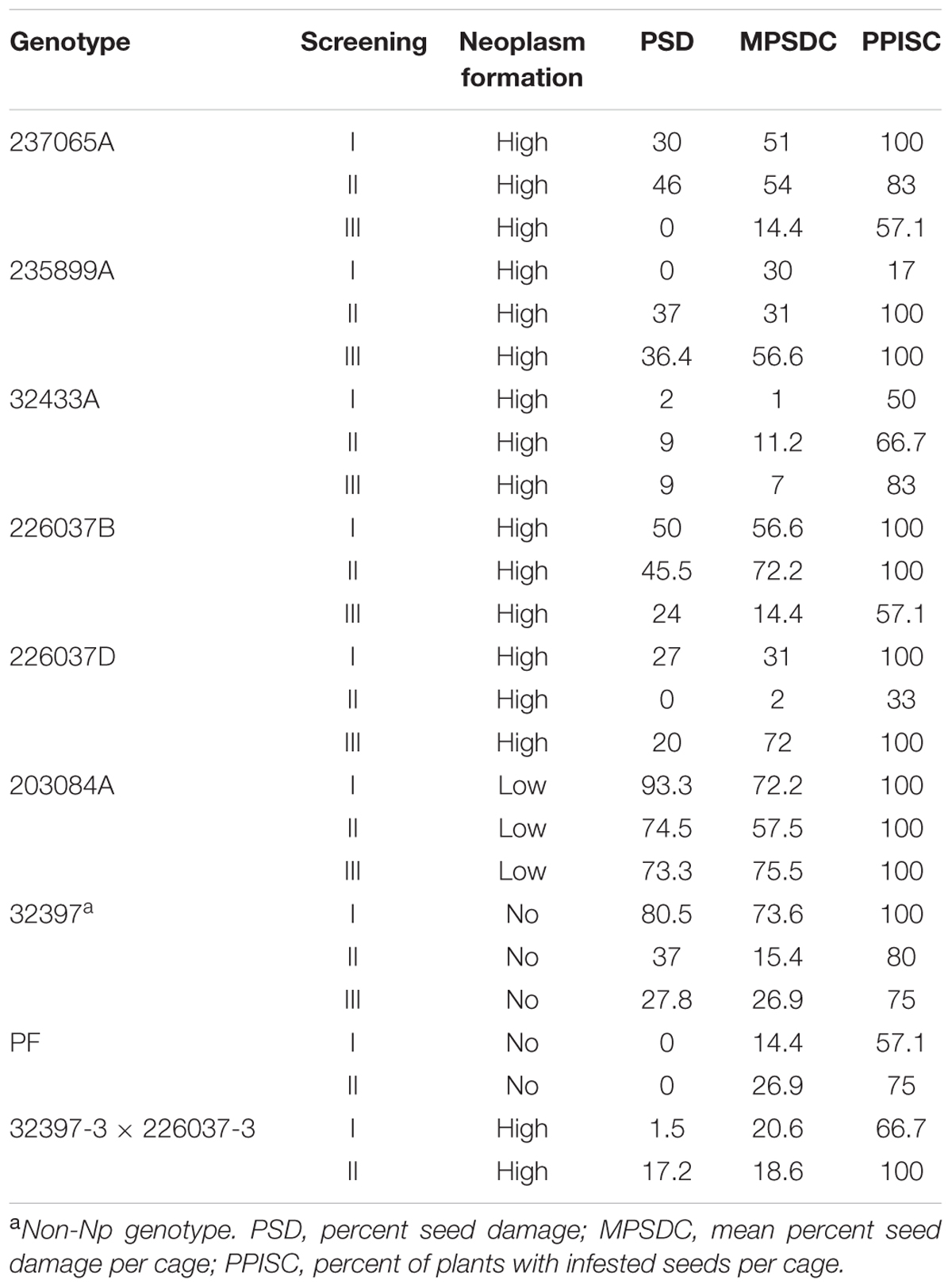
TABLE 3. Percentage of seed damage (PSD) of selected individual Np and wild type genotypes in three separate greenhouse experiments
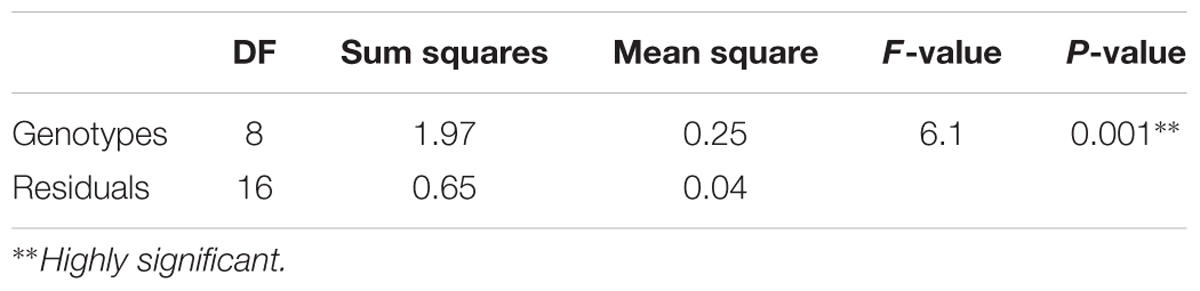
TABLE 4. ANOVA comparison of mean arcsine transformed PSD of genotypes tested in greenhouse screening.
Discussion
Neoplasm formation is an infrequent phenomenon that occurs in certain genotypes of P. sativum in the absence of UV light, for example under greenhouse conditions (Nuttall and Lyall, 1964). Figure 1A shows Np tissue growth in Np genotype 226037B when grown under greenhouse conditions. In the present study, 26% of the tested P. sativum accessions depicted the trait (Table 1). According to Berdnikov et al. (1992), only 2.3% of the assessed Ethiopian germplasm collections showed neoplasm formation. The high percentage of Np accessions observed in the present study is most likely due to preselection of these accessions from a pool of collections used for resistance screening against pea weevil (Teshome et al., 2015).
The present study clearly showed that neoplasm formation on Np genotypes is conspicuous. However, the level of neoplasm varies among Np genotypes and growing conditions. A similar trend was reported in an oviposition preference study by Mendesil et al. (2016), where the level of neoplasm formation was different among Np genotypes. This study revealed that the highest proportion of pods with neoplasm was recorded when Np genotypes were grown in the control chamber under greenhouse conditions. In the control chamber, 36% of the pods showed neoplasm formation. On the contrary, when the same genotypes were exposed to single and double UV lamps, the proportion of Np pods was reduced. Furthermore, when these genotypes were grown under field conditions, the percentage of Np pods dropped to only 7%. This result is consistent with previous findings that reported a negative influence of UV light on neoplasm formation (Nuttall and Lyall, 1964; Dodds and Matthews, 1966).
Previous studies showed that the oviposition of female pea weevil on pods of Np genotypes triggers the expression of Np gene (Berdnikov et al., 1992; Hardie, 1992; Doss et al., 2000). However, the type of neoplasm formed on Np pods in the absence of UV light or upon oviposition by pea weevil is morphologically different (Figures 1A,C). Host plants usually trigger a series of responses to either prevent further oviposition or to reduce the success rate of deposited eggs (Hilker and Meiners, 2002; Fatouros et al., 2005; Meiners et al., 2005). According to Doss et al. (2000), neoplasm formation triggered by oviposition can impede the entry of newly hatched larvae and hence minimize infestation rate. Furthermore, the additional mass of cells on the epidermal layer of Np pods could potentially upset the behavior of the gravid female pea weevil when choosing site of oviposition. Oviposition preference experiments showed that Np genotypes had a reduced rate of oviposition as compared to wild type genotypes (Mendesil et al., 2016). Despite this trait being pertinent in pea weevil resistance, its expression is attenuated by UV light and hence less effective against pea weevil under field conditions (Nuttall and Lyall, 1964; Snoad and Matthews, 1969; Doss et al., 1995).
Nuttall and Lyall (1964) and Doss et al. (1995) detected neoplasm formation on shaded pods grown in the field. The present study has also showed an increase in neoplasm formation when the pods are shaded from direct sunlight at the field level (data not shown). However, mechanical shading is inconvenient for periodic application and resource and time consuming in the case of small-scale farming systems. On the other hand, the idea to shade the pods of Np genotypes with the canopy of taller and branching sorghum plants resulted in a significant increase in neoplasm formation. The proportion of Np pods with intercropping was three fold higher than without it at the field level (Table 2). The fact that neoplasm formation can be enhanced with intercropping, as shown in this study, indicates that intercropping can be implemented as part of an integrated pest management approach against pea weevil.
The fact that Np genotypes scored a relatively low PSD in three consecutive experiments under greenhouse conditions suggests intercropping as a viable approach in pea weevil management. Doss et al. (2000) reported that Np genotypes are less susceptible to pea weevil in comparison with wild type genotypes under field conditions. Similar results were also reported by Teshome et al. (2015) in an experiment conducted for screening field pea germplasm for resistance against pea weevil. Hence, enhancing Np formation with intercropping could be a way forward to minimize pea weevil damage at field level. Intercropping could also result in release of non-host volatiles that can adversely affect the pea weevil’s capability to locate its host and oviposit. According to Ali et al. (2007), intercropping field pea with different crops reduces susceptibility to Ascochyta blight and pea aphid (Acyrthosiphon pisum) infestation. Hence, intercropping is a silver bullet management option in field pea production that is environmentally benign, cost effective and requires minimum skill-set for application.
In order to successfully use neoplasm formation in field pea as part of integrated pea weevil management, the trait needs to be bred into locally adapted varieties. The experiment conducted to determine the heritability of neoplasm in field pea in the present study through crossing Np genotypes with wild type genotypes showed that all F1 hybrids produced neoplasm under greenhouse conditions suggesting that the Np allele at Np locus is dominant over wild type and the inheritance of the trait is according to the principle of Mendelian genetics, in line with previous studies (Nuttall and Lyall, 1964; Dodds and Matthews, 1966). However, similar to what was observed in the parental Np genotypes, the exposure of the F1 hybrids to double UV lamps results in a significant reduction in neoplasm formation (Figure 3), which signifies the importance of avoiding direct sunlight for effective expression of this trait under field conditions. The interspecific hybrids of Np field pea genotypes and P. fulvum also depicted neoplasm formation under greenhouse conditions. P. fulvum is known to have enhanced resistance against pea weevil as compared to cultivated P. sativum varieties (Hardie et al., 1995; Clement et al., 2002; Byrne et al., 2008). In the present study, both the Np genotypes and the P. fulvum line included in the artificial infestation experiment scored comparatively low PSD (Table 4). Therefore, developing field pea varieties through crossing Np genotypes with P. fulvum could result in pyramiding of resistance genes with different modes of action against pea weevil. Such varieties could have sustainable resistance and could easily be augmented by integrated pest management techniques like intercropping and trap cropping.
Author Contributions
TB and MG secured the funding. AT and MG conceived and designed the study. AT, MG, EM, and SM collected the data. AT and MG performed data analysis. AT wrote the manuscript with help of MG, TB, EM, and SM.
Funding
The authors would like to thank the Swedish International Development Agency (Sida) for funding this research project. We would also like to acknowledge Ethiopian Institute of Biodiversity and NordGen, for providing field pea accessions used in the present study.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We would also like to thank Tiny Motlhaodi for providing the sorghum accessions used in the intercropping trial and Kerstin Brismar for her help with microscopy.
References
Ali, K., Louw, S. V., and Swart, W. J. (2007). Effect of mixed cropping of Pisum sativum L. on Acyrthosiphon pisum (Harris) infestation and ascochyta blight in Ethiopia. Pest. Manage. J. Eth. 11, 69–79.
Baker, G. J. (1998). Pea Weevil. Fact Sheet. Adelaide, SA: Primary Industries and Resources SA and the South Australian Research and Development Institute.
Berdnikov, V. A., Trusov, Y. A., Bogdanova, V. S., Kosterin, O. E., Rozov, S. M., Nedel’kina, S. V., et al. (1992). The neoplastic pod gene (Np) may be a factor for resistance to the pest Bruchus pisorum L. Pisum Genet. 24, 37–39.
Bousquet, Y. (1990). Beetles Associated with Stored Products in Canada: An Identification Guide, Publication 1837. Ottawa: Agriculture Canada.
Burgess, J., and Fleming, E. N. (1973). The structure and development of a genetic tumour of the pea. Protoplasma 76, 315–325. doi: 10.1007/BF01279129
Byrne, O. M., Hardie, D. C., Khan, T. N., Speijers, J. A., and Yan, G. (2008). Genetic analysis of pod and seed resistance to pea weevil in a Pisum sativum x P. fulvum interspecific cross. Aust. J. Agric. Res. 59, 854–862. doi: 10.1071/AR07353
Clement, S. L., Hardie, D. C., and Elberson, L. R. (2002). Variation among accessions of Pisum fulvum for resistance to pea weevil. Crop Sci. 42, 2167–2173. doi: 10.2135/cropsci2002.2167
Dicke, M., and Hilker, M. (2003). Induced plant defences: from molecular biology to evolutionary ecology. Basic Appl. Ecol. 4, 3–14. doi: 10.1078/1439-1791-00131
Doss, R. P. (2005). Treatment of pea pods with Bruchin B results in up-regulation of a gene similar to MtN19. Plant Physol. Biochem. 43, 225–231. doi: 10.1016/j.plaphy.2005.01.016
Doss, R. P., Oliver, J. E., Proebsting, W. M., Potter, S. W., Kuy, S., Clement, S. L., et al. (2000). Bruchins: insect-derived plant regulators that stimulate neoplasm formation. Proc. Natl. Acad. Sci. U.S.A. 97, 6218–6223. doi: 10.1073/pnas.110054697
Doss, R. P., Proebsting, W. M., Potter, S. W., and Clement, S. L. (1995). Response of Np mutant of pea (Pisum sativum L.) to pea weevil (Bruchus pisorum L.) oviposition and extracts. J. Chem. Ecol. 21, 1–10. doi: 10.1007/BF02033665
Fatouros, N. E., Bukovinszkine’Kiss, G., Kalkers, L. A., Gamborena, R. S., Dicke, M., and Hilker, M. (2005). Oviposition-induced plant cues: do they arrest Trichogramma wasps during host location? Entomol. Exp. Appl. 115, 207–215. doi: 10.1007/s10886-011-0047-9
Hardie, D. (1992). Resistance to the Pea Weevil in Pisum species. Ph.D. thesis, The Unviersity of Adelaide, Adelaide, SA.
Hardie, D. C., Baker, G. J., and Marshall, D. R. (1995). Field screening of Pisum accessions to evaluate their susceptibility to the pea weevil (Coleoptera: Bruchidae). Euphytica 84, 155–165. doi: 10.1007/BF01677954
Hilker, M., and Meiners, T. (2002). Induction of plant responses to oviposition and feeding by herbivorous arthropods: a comparison. Entomol. Exp. Appl. 104, 181–192. doi: 10.1046/j.1570-7458.2002.01005.x
Horne, J., and Bailey, P. (1991). Bruchus pisorum L. (Coleoptera, Bruchidae) control by a knockdown pyrethroid in field peas. Crop Prot. 10, 53–56. doi: 10.1016/0261-2194(91)90026-N
Hothorn, T., Bretz, F., and Westfall, P. (2008). Simultaneous inference in general parametric models. Biom. J. 50, 346–363. doi: 10.1002/bimj.200810425
Meiners, T., Hacker, N. K., Anderson, P., and Hilker, M. (2005). Response of the elm leaf beetle to host plants induced by oviposition and feeding: the infestation rate matters. Entomol. Exp. Appl. 115, 171–177. doi: 10.1111/j.1570-7458.2005.00280.x
Mendesil, E., Rämert, B., Marttila, S., Hillbur, Y., and Andersson, P. (2016). Oviposition preference of pea weevil, Bruchus pisorum L. among host and non-host plants and its implications for pest management. Front. Plant Sci. 6:1186. doi: 10.3389/fpls.2015.01186
Nuttall, V. W., and Lyall, L. H. (1964). Inheritance of neoplastic pod in the pea. J. Hered. 55, 184–186.
Oliver, J. E., Doss, R. P., Williamson, R. T., Carney, J. R., and DeVilbiss, E. D. (2000). Bruchins-mitogenic 3-(hydroxypropanoyl) esters of long chain diols from weevils of the Bruchidae. Tetrahedron 56, 7633–7641. doi: 10.1016/S0040-4020(00)00650-5
Pesho, G. R., Muehlbauer, F. J., and Harberts, W. H. (1977). Resistance of pea inroductions to pea weevil. J. Econ. Entomol. 70, 30–33. doi: 10.1093/jee/70.1.30
R Core Team (2015). A Language and Environment for Statistical Computing. Vienna: R Foundation for Statistical Computing. Available at: https://www.r-project.org/
Seyoum, E., Damte, T., Bejiga, G., and Tesfaye, A. (2012). “The status of pea weevil, Bruchus pisorum (Coleoptera: Chrysomelidae) in ethiopia,” in Proceedings of the 17th Annual Conference. 26-27 November 2010 Invasive Plant Pests Threatening Ethiopian Agriculture, Ethiopianed, ed. B. Mulatu (Addis Ababa: Plant Protection Society of Ethiopia), 52–66.
Snoad, B., and Matthews, P. (1969). “Neoplasm of the pea pod,” in Chromosomes Today, eds C. D. Darlingron and K. R. Lewis (Edinburgh, TX: Oliver & Boyd Ltd), 126–131.
Teshome, A., Mendesil, E., Geleta, M., Andargie, D., Anderson, P., Rämert, B., et al. (2015). Screening the primary gene pool of field pea (Pisum sativum L. var. sativum) in Ethiopia for resistance against pea weevil (Bruchus pisorum L.). Genet. Resour. Crop Evol. 62, 525–538. doi: 10.1007/s10722-014-0178-2
Keywords: Bruchus pisorum, field pea, intercropping, neoplasm, pea weevil, Pisum sativum
Citation: Teshome A, Bryngelsson T, Mendesil E, Marttila S and Geleta M (2016) Enhancing Neoplasm Expression in Field Pea (Pisum sativum) via Intercropping and Its Significance to Pea Weevil (Bruchus pisorum) Management. Front. Plant Sci. 7:654. doi: 10.3389/fpls.2016.00654
Received: 25 February 2016; Accepted: 28 April 2016;
Published: 18 May 2016.
Edited by:
Gero Benckiser, Justus-Liebig-Universität Gießen, GermanyReviewed by:
Oswaldo Valdes-Lopez, National Autonomus University of Mexico, MexicoVijai Kumar Gupta, NUI Galway, Ireland
Copyright © 2016 Teshome, Bryngelsson, Mendesil, Marttila and Geleta. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Abel Teshome, YWJlbGl0by50ZXNob21lQGdtYWlsLmNvbQ==; YWJlbC50ZXNob21lQHNsdS5zZQ==
 Abel Teshome
Abel Teshome Tomas Bryngelsson1
Tomas Bryngelsson1 Esayas Mendesil
Esayas Mendesil Salla Marttila
Salla Marttila Mulatu Geleta
Mulatu Geleta