- 1 Functional Brain Mapping Laboratory, University of Geneva Medical School, University of Geneva, Genève, Switzerland
- 2 Ecole de Traduction et d’Interprétation, University of Geneva, Genève, Switzerland
In this review we will focus on delineating the neural substrates of the executive control of language in the bilingual brain, based on the existing neuroimaging, intracranial, transcranial magnetic stimulation, and neuropsychological evidence. We will also offer insights from ongoing brain-imaging studies into the development of expertise in multilingual language control. We will concentrate specifically on evidence regarding how the brain selects and controls languages for comprehension and production. This question has been addressed in a number of ways and using various tasks, including language switching during production or perception, translation, and interpretation. We will attempt to synthesize existing evidence in order to bring to light the neural substrates that are crucial to executive control of language.
Introduction
There is a considerable behavioral literature demonstrating that multilingualism1 has benefits in domains extending beyond language (Diamond, 2010). For example, these studies have shown greater cognitive flexibility and control (Bialystok and Senman, 2004; Bialystok and Depape, 2009; Adi-Japha et al., 2010), superior performance on non-verbal switching tasks (Garbin et al., 2010; Prior and MacWhinney, 2010), and advantages on tests of attentional control and flexibility (Costa et al., 2008, 2009; Hernandez et al., 2010) in multilingual compared to monolingual children and adults. Several studies have also used functional brain-imaging to examine the advantages conferred by bilingualism on executive control (Bialystok et al., 2005; Garbin et al., 2010).
On the basis of experiments on bilingual speech production, Abutalebi and Green(2007, 2008) propose a model of language control in the multilingual brain whereby a left hemisphere cortico-subcortical loop comprising the anterior cingulate and prefrontal cortices alongside the left caudate nucleus work to control linguistic functions subtended by a left fronto-parietal network.
In the present paper, we will first briefly describe neuropsychological, intracranial stimulation, and transcranial magnetic stimulation (TMS) studies that contribute to elucidating the neural bases of language control. We will then review functional neuroimaging studies using various paradigms and linguistic stimuli to examine aspects of language control in bilingualism.
Neuropsychological Evidence
While neuroimaging studies provide correlational evidence for the engagement of brain regions during certain processes, more direct evidence for the involvement of a given structure is provided by neuropsychological reports, by intracranial stimulation studies, and by TMS studies which can directly show that a region is critically involved in a given process. Such lines of evidence are less frequently encountered than functional imaging. Here, we will briefly examine two reports of different speech pathologies, as well as the limited evidence from direct electrical brain stimulation studies. We will also describe a few relevant TMS case studies. Much of the evidence presented here focuses on language switching, i.e., the process of changing the output language. Healthy bilinguals normally select and switch languages as a function of the linguistic knowledge of the interlocutor. This normal process may, however, be disrupted after brain lesions.
Aphasia is a disorder of speech in which comprehension or production of speech is impaired, an extensive discussion of which is beyond the scope of this review. However, aphasia in bilingual patients is of interest in so far as some cases show a loss of appropriate control of language in one or both languages, thereby providing evidence for the involvement of particular brain areas for language control.
A psycholinguistic model of bilingual speech production worth considering in this context is Paradis’ activation threshold hypothesis (Paradis, 1993, 2001). It suggests that at any given moment, a stored item in a multilingual lexicon requires a certain amount of activation (its threshold) in order to be accessed. When the threshold is reached, all other alternatives are inhibited (their thresholds are raised). However, thresholds are never raised so high as to totally inhibit the language. Every time a particular item is activated its threshold decreases, but if it is not used for a while, its threshold increases. It has been suggested (Paradis, 1996) that lesions can alter the threshold of languages, which could therefore explain asymmetric impairments in languages in bilingual aphasia, as well as unequal recovery.
Pathological mixing, which refers to the intermingling of languages within a single utterance, has been reported after left temporo-parietal lesions (Fabbro, 1999), suggesting a crucial role for this region in maintaining the appropriate “language set” for output. A related, but different pathology is pathological switching, in which a patient alternates the language of utterance between self-contained speech segments. Fabbro et al. (2000) report a case of pathological language switching following a lesion of the left (and partly of the right) anterior cingulate gyrus, and of the white matter underlying the left inferior, middle, and superior frontal gyri. The patient displayed no aphasic symptoms in either language but was found to switch between his L1 and L2 despite instructions to speak only one language, and despite displaying awareness of the switches.
Abutalebi et al. (2000) report a case of pathological language mixing after a lesion incorporating the head of the left caudate nucleus. They note that the patient always produced output in which noun phrases were complete, and morphological markers were always used appropriately, suggesting that the impairment involved a stage of lexical selection subsequent to specification of syntactic and semantic information.
Marien et al. (2005) report a rare case of bilingual subcortical aphasia in a child. Following a left thalamic hemorrhage, the patient displayed global aphasia in L1 and L2. A few days later the patient displayed fluent aphasia equally affecting L1 and L2, prominently featuring spontaneous pathological language mixing and switching. The patient also displayed significant translation difficulties. Investigation with SPECT2 showed hypoperfusion in left fronto-parietal and temporal regions as well as the left caudate nucleus. Six months later pathological mixing and switching had remitted but translation difficulties and fluent L1 and L2 aphasia had not. Follow-up SPECT imaging showed relative increases in perfusion in the left frontal regions and left caudate nucleus, but not the left temporal or parietal regions. The pattern of recovery in this case provides compelling evidence for the involvement of a left fronto-subcortical network in language selection.
Abutalebi et al. (2009) studied the recovery of a patient afflicted by bilingual aphasia after a left basal ganglia hemorrhage implicating the globus pallidus and putamen. Initially the patient showed global aphasia in L1 and L2, which changed to fluent aphasia with anomia equally affecting both languages after a few days. The patient was then treated with intensive speech therapy in L2 only, which significantly improved the aphasic symptoms in L2, but not L1. A dynamic causal modeling analysis of functional magnetic resonance imaging (fMRI) data obtained during a bilingual picture naming task showed that improvement in naming performance after treatment was associated with increased functional coupling between a fronto-subcortical network (the “control network”) and a fronto-temporal one (the “language network”).
Aglioti and Fabbro (1993), followed-up by Aglioti et al. (1996) report a case of subcortical bilingual aphasia in which a lesion of the left basal ganglia asymmetrically more severely impaired the patient’s most used language. The impairment included increased difficulty translating into this language. They ascribed this asymmetrical outcome to the role of the basal ganglia in controlling automatized motor and cognitive tasks (for review, see Takakusaki et al., 2004) and in managing behavioral patterns (cf. Graybiel, 1997), arguing that the most used language is more automated than a less used one.
These case studies heavily implicate the basal ganglia in the control of language output in multilingual individuals. However, one report contradicts this view. Fabbro et al. (2000) report a case of pathological language switching following a lesion of the left (and partly of the right) anterior cingulate gyrus, and of the white matter underlying the left inferior, middle, and superior frontal gyri. The patient displayed no aphasic symptoms in either language but was found to switch between his L1 and L2 despite instructions to speak only one language, and despite displaying awareness of the switches. Although Fabbro et al. (2000) argue that language switching is controlled by mechanisms generally involved in task switching (that is, a fronto-parietal network) the published MRI images of the lesion also show damage to the left striatum (see Marien et al., 2005), which could be the cause of the patient’s pathological switching. This reinterpretation is consistent with other reports of pathological language mixing and switching in polyglot aphasia following subcortical damage.
Intracranial Electrical Stimulation and Transcranial Magnetic Stimulation Evidence
Less direct evidence for the involvement of the left dorsolateral prefrontal cortex (DLPFC) in language switching comes from a TMS study by Holtzheimer et al. (2005). They reported involuntary language switching in two patients who were treated for drug-resistant major depressive disorder using repetitive TMS to temporarily inhibit various cortical regions. In the first case, an English–German bilingual patient, whose primary spoken and written language was English, reported “thinking in German” after a session of rTMS over the left DLPFC. A second patient, an English–Spanish bilingual, similarly reported the “thinking in Spanish” and the impulse to speak to the tester in Spanish after rTMS of the left DLPFC. Such evidence is not entirely conclusive, since the mechanism of action is unknown, but it further bolsters the evidence that the left DLPFC can play a role in language switching.
Nardone et al. (2011) have recently reported that excitatory TMS of the left DLPFC transiently alleviated pathological language switching in a bilingual patient who had suffered a left frontal stroke.
The most direct evidence for brain regions implicated in language control in bilinguals come from intracranial stimulation studies, although such studies are very rare. Moritz-Gasser and Duffau (2009b) used direct electrical stimulation to map a language switching network in a bilingual patient. They demonstrated that stimulation of multiple sites could induce language switching, namely stimulation of the left posterior superior temporal sulcus and subcortical stimulation of the left superior longitudinal fasciculus (a white matter tract which connects the left inferior frontal and posterior superior temporal cortices). In an earlier intraoperative study, Kho et al. (2007) induced an involuntary shift from French (L1) to Chinese (L2) during a counting task by stimulating a site in the left inferior frontal gyrus. In another patient they reported involuntary language switching during a Wada test in which the left hemisphere was anesthetized.
Taken together, these reports point to the involvement of a left-lateralized fronto-temporal network in regulating language switching. We suggest that this can be reconciled with the apparently contradictory neuropsychological evidence in the following way. These studies, which show that stimulation or inhibition of cortical areas can lead to language switching, do not necessarily prove that these regions are involved in language selection processes. There is evidence that different languages may be represented in different portions of cortex in multilingual brains (Fabbro, 2001; Sebastian-Galles et al., 2006; Leonard et al., 2010), although this may be a function of proficiency or age of acquisition of L2 (Dehaene et al., 1997; Kim et al., 1997; Golestani et al., 2006). If this holds for the participants of these investigations then it may be the case that these investigations have differentially inhibited or excited the representation of a given language over another, and it is the consequent facilitation or impairment of access that leads to the language switching behavior, without selection mechanisms necessarily being involved. Under such a schema, the subcortical regions implicated by the neuropsychological evidence are likely to be involved in the management of cortical representation for appropriate behavioral output. Such an architecture is in line with much existing data on executive control in other domains. However, the cases reported by Kho et al. (2007) do suggest that a left-lateralized cortical network is part of the switching mechanism.
By examining the functional neuroimaging literature alongside these case reports, we can further delineate the role of these brain areas, in vivo, in healthy volunteers.
Neuroimaging Evidence
We will focus principally on fMRI studies as they are the most informative with respect to localization of language control processes. We will also review studies that have employed alternative imaging techniques, such as positron emission tomography (PET) and optical imaging (near infrared spectroscopy, NIRS). Numerous electroencephalography (EEG) studies have been carried out to explore these questions. However, these studies mainly focus on the temporal dynamics of language control rather than its localization, and we will therefore address them briefly in a separate section.
Language Switching
Language switching tasks can provide direct insight into the substrates of controlling language. Behavioral evidence (Meuter and Allport, 1999) shows that switching between languages is associated with a cost, as manifested by slowed reaction times. The neural manifestations of this cost have been investigated using a variety of tasks in which participants are required either to comprehend or to produce stimuli in multiple languages.
Receptive tasks have included listening to a series of words in either of two languages [Price et al., 1999 (PET); Rodriguez-Fornells et al., 2002 (fMRI)], and listening to sentences with a language switch midway through [Abutalebi et al., 2007 (fMRI)].
Crinion et al. (2006) used a task involving covert reading of words in alternating languages [2006 (PET)]. Explicit, or overt production tasks that have been employed include naming pictures in alternating languages [Hernandez et al., 2000 (fMRI); Hernandez et al., 2001 (fMRI); Khateb et al., 2007 (EEG); Abutalebi et al., 2008 (fMRI); Costa et al., 2009 (fMRI)], digit naming in alternating languages [Wang et al., 2009 (fMRI)], language switching during verbal fluency tasks [Hirshorn and Thompson-Schill, 2006 (fMRI)], and language switching during alternate translation from L1 → L2 and L2 → L1 [Quaresima et al., 2002 (NIRS)].
Neuroimaging evidence for the neural substrates of language switching has implicated a network of predominantly left-hemisphere lateralized cortical regions, including the superior temporal sulcus (Rodriguez-Fornells et al., 2002; Abutalebi et al., 2007, 2008), the superior and inferior parietal lobule (Price et al., 1999; Rodriguez-Fornells et al., 2002; Hirshorn and Thompson-Schill, 2006; Khateb et al., 2007; Costa et al., 2009; Wang et al., 2009), the supplementary motor area (SMA; Wang et al., 2007; Abutalebi et al., 2008), the DLPFC (Hernandez et al., 2000, 2001; Hirshorn and Thompson-Schill, 2006; Khateb et al., 2007; Abutalebi et al., 2008; Wang et al., 2009), the inferior frontal gyrus (Price et al., 1999; Hernandez et al., 2001; Quaresima et al., 2002; Rodriguez-Fornells et al., 2002; Hirshorn and Thompson-Schill, 2006; Abutalebi et al., 2007, 2008), the precentral gyrus (Khateb et al., 2007; Wang et al., 2009), and the right anterior cingulate cortex (Abutalebi et al., 2008). Other right hemisphere activations are reported in the DLPFC, the precentral gyrus, and the SMA by Hernandez (2009) for switching versus not switching during picture naming. Despite the heterogeneity of paradigms used, a consensus does seem to emerge, implicating the left inferior frontal gyrus, left DLPFC, and the left parietal lobule during language switching, consistent with the evidence from TMS and direct stimulation studies presented above. The SMA and precentral gyrus may additionally be engaged in tasks that involve productive switches.
The above described fronto-parietal network overlaps considerably with that ascribed to general executive control which is implicated in diverse processes such as inhibition of prepotent responses, initiation of behavior, planning of action, judgment and decision making, and feedback management (e.g., Collette et al., 2005, 2006; Schumacher et al., 2007; Nagel et al., 2008). Considerable attention has been devoted to the differences between language switching and more general task switching (for discussion see Moritz-Gasser and Duffau, 2009a), and the extent of such differences remains a matter of debate.
Translation Tasks
Translation requires rapid access to representations of lexical items in two languages. It therefore demands a different type of language control compared to that required during language switching: selection is still essential, but simply favoring one language over another will not enable faithful translation, beyond the case of isolated words.
Two of the studies described above also included translation tasks (silently mouthing translations of visually presented words: Price et al., 1999; or overtly producing translations of them: Quaresima et al., 2002) in the context of language switching paradigms. Further studies have focused more explicitly on the process of translation. Klein et al. (1995) recorded brain activity using PET while bilingual participants overtly translated single auditorily presented words. Lehtonen et al. (2005) conducted an fMRI investigation in which they asked bilingual individuals to silently translate visually presented sentences. This latter study is of particular interest as it is the only one in which participants are required to tap supra-lexical levels of the speech system in order to successfully carry out the translation task.
Price et al. (1999) showed involvement of the anterior cingulate cortex, the putamen and head of the caudate nucleus, the SMA, the left anterior insula, and the cerebellum bilaterally during silent mouthing of translations. Quaresima et al. (2002) examined only the anterior portion of the left hemisphere during overt translation, and found activation of this region during task performance. Klein et al. (1995) demonstrated engagement of left-lateralized inferior frontal, dorsolateral prefrontal, and inferior temporal cortices, as well as (specifically for translation from L2 → L1) activation of the left putamen. Lehtonen et al. (2005) reported significant activation of the left inferior frontal gyrus and putamen for translation from L2 → L1. We propose that these activations arise from two processes: semantic retrieval in the left inferior frontal gyrus, and control of output in the basal ganglia.
Interpretation Tasks
Simultaneous interpretation places even heavier demands upon the executive control of language than does translation of isolated words or sentences. It requires not just the ongoing retrieval of lexical, terminological, and phraseological units in the appropriate language, but also the maintenance of information in verbal working memory and the continuous monitoring of input and output streams, while constantly executing language and modality switches (Moser, 1978; Moser-Mercer et al., 2000; Christoffels et al., 2006)
There are substantial difficulties in examining overt interpretation of sentences using most imaging techniques as they are highly susceptible to the artifacts arising from speech-related movements. Thus, very few studies have attempted to investigate simultaneous interpretation. Rinne et al. (2000) carried out a PET investigation of eight professional simultaneous interpreters, using overt production. They found that the left premotor and ventrolateral prefrontal cortices were engaged during interpretation both from L2 to L1 and from L1 to L2. In addition, interpreting into L2 engaged the left inferior temporal cortex and the right cerebellum.
In an ongoing fMRI study of the neural substrates of simultaneous interpretation (Hervais-Adelman and colleagues, in preparation), 34 multilingual participants were asked to listen to sentences in a highly proficient second language, and to either shadow (simultaneously repeat) or simultaneously interpret sentences into their L1. Shadowing speech calls for simultaneous speech production and perception, as well as for the simultaneous processing of two streams of speech (one being the sentence being heard, and the other being the feedback from the participants’ own output) in a single language, whereas simultaneous interpretation calls for the simultaneous processing of two languages, with the input and output streams being different languages. Comparison of activations arising from these two reveals the substrates underlying the simultaneous processing of two languages during simultaneous interpretation. Preliminary results provide evidence for the engagement of the left premotor and ventrolateral prefrontal cortices, alongside the pre-SMA and caudate nucleus for interpretation into L1 (Figure 1). The pattern of the preliminary results is consistent with much of the evidence presented above for the role of these regions in language control.
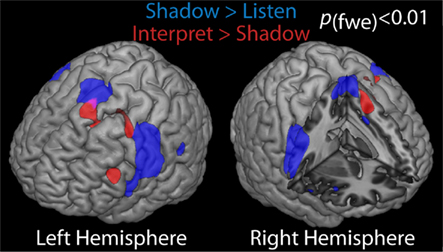
Figure 1. Significant differences in activation levels in 34 non-experts, rendered on canonical single-subject brain. Contrasts shown are speech shadowing in L2 versus listening to L2 (blue) and simultaneous interpretation into L1 versus shadowing (red), at a family wise error corrected significance level of p < 0.01.
We have also recently found evidence for brain structural plasticity in individuals training to become simultaneous language interpreters as they develop expertise in this skill. We found that in interpretation students, but not in matched controls, there is an increase in gray matter volume over the course of a 15-month training program in brain regions known to be involved not only in semantic processing but also in aspects of executive function and error monitoring (Figure 2; Golestani and colleagues, in preparation). These preliminary results constitute direct, longitudinal evidence for experience-dependent plasticity. These results, coupled with other functional imaging results, lend further credibility to the hypothesis of a left-lateralized fronto-parieto-subcortical mechanism for controlling language output and comprehension in multilingual individuals.
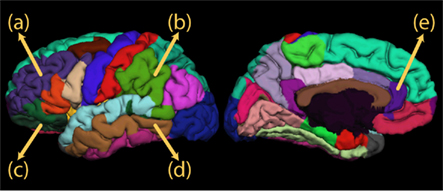
Figure 2. Regions in which we found longitudinal evidence for brain structural plasticity in simultaneous language interpreters: (A) left middle frontal gyrus, (B) left supramarginal gyrus, (C) left pars orbitalis, (D) left middle temporal gyrus, (E) rostral anterior cingulate.
Electroencephalography
The literature on event-related potentials (ERPs) in the study of bilingualism has been thoroughly reviewed by Moreno et al. (2008). ERPs reflect underlying neural activity with a high degree of temporal resolution but in themselves do not provide information about the location of that activity in the brain. Although it is possible to localize the sources of ERPs with an adequate degree of spatial resolution, the articles described in the following section describe only analyses of the temporal dynamics of the neural responses, with varying degrees of topographic accuracy. Even though they do not provide information relating to localization of relevant functions, the information they provide about the time-course of processing is nevertheless illuminating.
In speech production, there is plentiful evidence (see, for example, reviews by Costa, 2005; and Kroll et al., 2008) that languages are simultaneously activated and the inappropriate one suppressed, as a function of task. By providing information at a higher temporal resolution than other imaging modalities, ERP studies can directly address questions such as the psycholinguistic stages of representation and selection at which language interference occurs. For example, Hoshino and Thierry (2011) used EEG in an interference paradigm to determine the timing of language selection in a production task, and showed parallel activation of languages even beyond lexical selection. In comprehension, Van Heuven and Dijkstra (2010) have reviewed the EEG and MRI evidence for various psycholinguistic models of bilingual word recognition, and have found that the evidence favors their bilingual interactive activation+ (BIA+) model, which posits an integrated bilingual lexicon that is accessed in a language non-selective manner (for details of the model see Dijkstra and Van Heuven, 2002).
We will here look at two paradigms that have been used to explore ERPs of language control. These are go/no-go tasks and language switching tasks. We begin by discussing go/no-go tasks.
Go/no-go Tasks
In a go/no-go task, participants are required to respond only if certain conditions are met (“go” trials) or otherwise to make no response (“no-go” trials). The magnitude of an ERP component known as the N200 during no-go trials is thought to reflect the control processes relating to suppression of responses. The N200 (or N2) is a negativity observed approximately 200 ms after stimulus onset. The exact role of the N200 is debated; Nieuwenhuis et al. (2003) argue that its presence reflects response inhibition, while Donkers and van Boxtel (2004) argue that it reflects conflict-monitoring. Enhancement of N200 components has been interpreted as reflecting interference effects in bilingual tasks, and has been fairly widely observed (see Rodriguez-Fornells et al., 2006 for review). However, more recent evidence (Huster et al., 2010) suggests that the N200 may in fact reflect response selection, and that a later component, the P300, may reflect inhibitory cognitive components. Nevertheless, the N200 is closely associated with some aspect of response–suppression, in linguistic and non-linguistic tasks. Nieuwenhuis et al. (2003) localized the source of the N200 as the anterior cingulate cortex, and Huster et al. (2010) attributed its source to the left anterior middle cingulate cortex. These localizations are consistent with the neuroimaging evidence presented above.
Moreno et al. (2008) also describe a later ERP component that is systematically greater in amplitude in bilingual than monolingual participants during no-go trials, this being a mid-frontal negativity observed between 400 and 800 ms post-stimulus onset (also reviewed in Rodriguez-Fornells et al., 2006). They suggest that this component reflects enhanced cognitive control mechanisms related to the day-to-day demands of bilingualism.
Language Switching Tasks
Language switching is a task that directly calls upon language selection and control mechanisms, and has been extensively used in the study of bilingual control. However, the paradigms and results are rather heterogeneous across studies, and as such, the typical ERP components of language switching during speech production have not been well characterized. Nevertheless, over the studies they review, Moreno et al. (2008) conclude that the data indicate that language switching in production requires active inhibition of a non-target language, and that the ERPs related to language switching and to withholding responses during non-linguistic go/no-go tasks are substantially similar.
For switching during receptive tasks, the data are likewise inconsistent and seem to vary depending on the paradigm. Paradigms requiring participants to make semantic judgments appear to elicit enhanced N400 components for switch trials (e.g., Alvarez et al., 2003; Proverbio et al., 2004). Although there is an ongoing controversy about the exact functional interpretation of the N400, it is generally accepted that the amplitude of the N400 component is sensitive to semantic aspects of word processing, particularly to the cloze probability of a word as it is greater in the case of unexpected words (Kutas et al., 2006; Steinhauer and Connolly, 2008; Friederici and Wartenburger, 2010). While Alvarez et al. (2003) found that N400 was specifically enhanced for L1 to L2 switches, Chauncey et al. (2008) found the reverse. They used masked-priming to examine the ERP correlates of language switching without an overt language switch (the primes were largely invisible) and found enhancements of N250 and N400 components for switch trials. The N400 component was particularly enhanced for L2 to L1 switches and the N250 component was particularly enhanced for L1 to L2 switches.
Code switches are a particular form of language switching, whereby multilingual speakers electively employ words from alternative languages within utterances, while respecting the syntactic structure of the carrier language. It may be expected that listening to such switches might elicit similar ERPs to those described above. However, Moreno et al. (2002) found that code switches within sentences did not elicit enhanced N400 effects while lexical switches did. Instead, the code switch trials produced an enhanced posterior late positivity component (LPC), which is generally observed in response to unexpected or improbable task-relevant events (see, e.g., reviews by Donchin and Coles, 1988; Picton, 1992; Polich and Kok, 1995; Polich, 2007).
A recent study by Kuipers and Thierry (2010) sought to examine the time-course of neural events related to the detection of language changes using an auditory oddball paradigm. They compared ERPs elicited by rare language switch events with those elicited by frequent no-switch trials. They found that bilingual participants showed a response to language switches as early as 200 ms, followed by an N400, while monolingual participants showed only an enhanced N400 in response to switches, suggesting a fundamental difference in the early processing of words in bilinguals. There was also a group difference in the P600 component, which was enhanced for switch trials in bilinguals but not monolinguals. The P600 is associated with stimulus re-evaluation (Osterhout and Holcomb, 1992; Hahne and Friederici, 1999), implying that the bilinguals and not the monolinguals engaged in a process of reinterpreting the stimuli after a switch. The data suggest that bilinguals have a mechanism for rapidly detecting and adapting to language switches.
Overall, the existing work using ERPs to investigate the neural substrates of language control reveals several similarities between bilingual language control and control of other executive functions. Although it is difficult to draw conclusions about the localization of the functions tapped by the variety of tasks and paradigms that have been employed, the findings are complementary to those revealed using methods that offer higher spatial resolution. Additionally, ERP findings contribute to a better understanding of the stages of processing involved in bilingual language control.
Conclusion
We have described a number of studies from functional neuroimaging, direct brain stimulation, TMS, and neuropsychology that outline the neural bases of the executive control of language. Beyond the domain of multilingual language control, a fronto-basal-ganglia network has been implicated in the inhibitory control of action and cognition (Aron et al., 2007), and this appears to converge with part of the putative bilingual language control network outlined here. In the context of language switching tasks, the evidence points mainly to a cortical network incorporating the parietal lobe, the posterior superior temporal sulcus and the left inferior frontal gyrus. Tasks involving the conversion of content from one language to another (i.e., translation and interpretation) mainly engage a left-lateralized cortico-subcortical circuit, including the basal ganglia, inferior frontal gyrus, and DLPFC. There is strong anatomical support for functional links between these regions.
We propose that the evidence suggests the presence of two distinct networks contributing to the executive control of language. Although perturbing either may have superficially similar behavioral consequences, they are likely to have differing roles. It seems likely that a fronto-basal-ganglia loop is implicated in the inhibition of inappropriate languages during production. The basal ganglia also play an apparently crucial role in enabling access to translation equivalents, which may reflect inhibitory processes that allow the selection of a term in one language rather than another. Alongside this network, there appears to be another, cortical, fronto-parietal network that sustains more general switching mechanisms. This system, like the fronto-basal-ganglia system delineated above, has a role in other executive functions. These two systems, working in concert with language-specific brain areas, likely manage both inhibitory control as well as language selection, both of which are necessary for the effective management of language in bilingual brains.
The critical components underlying the executive control of language in the multilingual brain seem well delineated, but the exact functional roles of these components and their interactions remain to be fully described. Ongoing work on the acquisition of expertise in interpretation, which is a highly demanding linguistic task involving rapid language switching and handling multiple simultaneous linguistic streams, will shed further light on the executive control of language in the multilingual brain.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
- ^We use the terms bilingual and multilingual to refer to individuals who have attained a fluent level of speech in more than one language. We do not attempt to distinguish between individuals who acquired their second (or additional) languages early as opposed to late in life, although we acknowledge that this has a significant impact on language processing. In this paper we use the terms L1 to refer to an individual’s first-acquired language and L2 to their second, irrespective of the fluency level of the two languages.
- ^ Single-photon emission computed tomography, a technique allowing imaging of brain metabolic activity.
References
Abutalebi, J., Annoni, J. M., Zimine, I., Pegna, A. J., Seghier, M. L., Lee-Jahnke, H., Lazeyras, F., Cappa, S. F., and Khateb, A. (2008). Language control and lexical competition in bilinguals: an event-related fMRI study. Cereb. Cortex 18, 1496–1505.
Abutalebi, J., Brambati, S. M., Annoni, J. M., Moro, A., Cappa, S. F., and Perani, D. (2007). The neural cost of the auditory perception of language switches: an event-related functional magnetic resonance imaging study in bilinguals. J. Neurosci. 27, 13762–13769.
Abutalebi, J., and Green, D. (2007). Bilingual language production: The neurocognition of language representation and control. J. Neurolinguistics 20, 242–275.
Abutalebi, J., and Green, D. (2008). Control mechanisms in bilingual language production: neural evidence from language switching studies. Lang. Cogn. Process. 23, 557–582.
Abutalebi, J., Miozzo, A., and Cappa, S. F. (2000). Do subcortical structures control “language selection” in polyglots? Evidence from pathological language mixing. Neurocase 6, 51–56.
Abutalebi, J., Rosa, P. A., Tettamanti, M., Green, D. W., and Cappa, S. F. (2009). Bilingual aphasia and language control: a follow-up fMRI and intrinsic connectivity study. Brain Lang. 109, 141–156.
Adi-Japha, E., Berberich-Artzi, J., and Libnawi, A. (2010). Cognitive flexibility in drawings of bilingual children. Child Dev. 81, 1356–1366.
Aglioti, S., Beltramello, A., Girardi, F., and Fabbro, F. (1996). Neurolinguistic and follow-up study of an unusual pattern of recovery from bilingual subcortical aphasia. Brain 119, 1551.
Aglioti, S., and Fabbro, F. (1993). Paradoxical selective recovery in a bilingual aphasic following subcortical lesions. Neuroreport 4, 1359.
Alvarez, R. P., Holcomb, P. J., and Grainger, J. (2003). Accessing word meaning in two languages: an event-related brain potential study of beginning bilinguals. Brain Lang. 87, 290–304.
Aron, A. R., Durston, S., Eagle, D. M., Logan, G. D., Stinear, C. M., and Stuphorn, V. (2007). Converging evidence for a fronto-basal-ganglia network for inhibitory control of action and cognition. J. Neurosci. 27, 11860–11864.
Bialystok, E., Craik, F. I. M., Grady, C., Chau, W., Ishii, R., Gunji, A., and Pantev, C. (2005). Effect of bilingualism on cognitive control in the Simon task: evidence from MEG. Neuroimage 24, 40–49.
Bialystok, E., and Depape, A. M. (2009). Musical expertise, bilingualism, and executive functioning. J. Exp. Psychol. Hum. Percept. Perform. 35, 565–574.
Bialystok, E., and Senman, L. (2004). Executive processes in appearance-reality tasks: the role of inhibition of attention and symbolic representation. Child Dev. 75, 562–579.
Chauncey, K., Grainger, J., and Holcomb, P. J. (2008). Code-switching effects in bilingual word recognition: a masked priming study with event-related potentials. Brain Lang. 105, 161–174.
Christoffels, I. K., De Groot, A., and Kroll, J. F. (2006). Memory and language skills in simultaneous interpreters: the role of expertise and language proficiency. J. Mem. Lang. 54, 324–345.
Collette, F., Hogge, M., Salmon, E., and Van Der Linden, M. (2006). Exploration of the neural substrates of executive functioning by functional neuroimaging. Neuroscience 139, 209–221.
Collette, F., Van Der Linden, M., Laureys, S., Delfiore, G., Degueldre, C., Luxen, A., and Salmon, E. (2005). Exploring the unity and diversity of the neural substrates of executive functioning. Hum. Brain Mapp. 25, 409–423.
Costa, A. (2005). “Lexical access in bilingual production,” in Handbook of Bilingualism: Psycholinguistic Approaches, eds J. Kroll, and A. M. B. De Groot (Oxford: Oxford University Press), 308–325.
Costa, A., Hernandez, M., Costa-Faidella, J., and Sebastian-Galles, N. (2009). On the bilingual advantage in conflict processing: now you see it, now you don’t. Cognition 113, 135–149.
Costa, A., Hernandez, M., and Sebastian-Galls, N. (2008). Bilingualism aids conflict resolution: evidence from the ANT task. Cognition 106, 59–86.
Crinion, J., Turner, R., Grogan, A., Hanakawa, T., Noppeney, U., Devlin, J. T., Aso, T., Urayama, S., Fukuyama, H., Stockton, K., Usui, K., Green, D. W., and Price, C. J. (2006). Language control in the bilingual brain. Science 312, 1537–1540.
Dehaene, S., Dupoux, E., Mehler, J., Cohen, L., Paulesu, E., Perani, D., Van De Moortele, P. F., Lehèricy, S., and Le Bihan, D. (1997). Anatomical variability in the cortical representation of first and second language. Neuroreport 8, 3809.
Dijkstra, T., and Van Heuven, W. J. B. (2002). The architecture of the bilingual word recognition system: from identification to decision. Biling. Lang. Cogn. 5, 175–197.
Donchin, E., and Coles, M. G. H. (1988). Is the P300 component a manifestation of context updating? Behav. Brain Sci. 11, 357–374.
Donkers, F. C. L., and van Boxtel, G. J. M. (2004). The N2 in go/no-go tasks reflects conflict monitoring not response inhibition. Brain Cogn. 56, 165–176.
Fabbro, F. (2001). The bilingual brain: cerebral representation of languages. Brain Lang. 79, 211–222.
Fabbro, F. (ed.). (1999). The Neurolinguistics of Bilingualism: An Introduction. Hove: Psychology Press.
Fabbro, F., Skrap, M., and Aglioti, S. (2000). Pathological switching between languages after frontal lesions in a bilingual patient. J. Neurol. Neurosurg. Psychiatr. 68, 650.
Friederici, A. D., and Wartenburger, I. (2010). Language and brain. Wiley Interdiscip. Rev. Cogn. Sci. 1, 150–159.
Garbin, G., Sanjuan, A., Forn, C., Bustamante, J. C., Rodriguez-Pujadas, A., Belloch, V., Hernandez, M., Costa, A., and Avila, C. (2010). Bridging language and attention: brain basis of the impact of bilingualism on cognitive control. Neuroimage 53, 1272–1278.
Golestani, N., Alario, F. X., Meriaux, S., Le Bihan, D., Dehaene, S., and Pallier, C. (2006). Syntax production in bilinguals. Neuropsychologia 44, 1029–1040.
Graybiel, A. M. (1997). The basal ganglia and cognitive pattern generators. Schizophr. Bull. 23, 459–469.
Hahne, A., and Friederici, A. D. (1999). Electrophysiological evidence for two steps in syntactic analysis. Early automatic and late controlled processes. J. Cogn. Neurosci. 11, 194–205.
Hernandez, A., Martinez, A., and Kohnert, K. (2000). In search of the language switch: an fMRI study of picture naming in Spanish-English bilinguals. Brain Lang. 73, 421–431.
Hernandez, A. E. (2009). Language switching in the bilingual brain: what’s next? Brain Lang. 109, 133–140.
Hernandez, A. E., Dapretto, M., Mazziotta, J., and Bookheimer, S. (2001). Language switching and language representation in Spanish-English bilinguals: an fMRI study. Neuroimage 14, 510–520.
Hernandez, M., Costa, A., Fuentes, L. J., Vivas, A. B., and Sebastian-Galles, N. (2010). The impact of bilingualism on the executive control and orienting networks of attention. Biling. Lang. Cogn. 13, 315–325.
Hirshorn, E. A., and Thompson-Schill, S. L. (2006). Role of the left inferior frontal gyrus in covert word retrieval: neural correlates of switching during verbal fluency. Neuropsychologia 44, 2547–2557.
Holtzheimer, P., Fawaz, W., Wilson, C., and Avery, D. (2005). Repetitive transcranial magnetic stimulation may induce language switching in bilingual patients. Brain Lang. 94, 274–277.
Hoshino, N., and Thierry, G. (2011). Language selection in bilingual word production: electrophysiological evidence for cross-language competition. Brain Res. 1371, 100–109.
Huster, R. J., Westerhausen, R., Pantev, C., and Konrad, C. (2010). The role of the cingulate cortex as neural generator of the N200 and P300 in a tactile response inhibition task. Hum. Brain Mapp. 31, 1260–1271.
Khateb, A., Abutalebi, J., Michel, C. M., Pegna, A. J., Lee-Jahnke, H., and Annoni, J. M. (2007). Language selection in bilinguals: a spatio-temporal analysis of electric brain activity. Int. J. Psychophysiol. 65, 201–213.
Kho, K. H., Duffau, H., Gatignol, P., Leijten, F. S. S., Ramsey, N. F., Van Rijen, P. C., and Rutten, G. J. M. (2007). Involuntary language switching in two bilingual patients during the Wada test and intraoperative electrocortical stimulation. Brain Lang. 101, 31–37.
Kim, K. H. S., Relkin, N. R., Lee, K. M., and Hirsch, J. (1997). Distinct cortical areas associated with native and second languages. Nature 388, 171–174.
Klein, D., Milner, B., Zatorre, R. J., Meyer, E., and Evans, A. C. (1995). The neural substrates underlying word generation – a bilingual functional-imaging study. Proc. Natl. Acad. Sci. U.S.A. 92, 2899–2903.
Kroll, J. F., Bobb, S. C., Misra, M., and Guo, T. (2008). Language selection in bilingual speech: evidence for inhibitory processes. Acta Psychol. (Amst.) 128, 416–430.
Kuipers, J. R., and Thierry, G. (2010). Event-related brain potentials reveal the time-course of language change detection in early bilinguals. Neuroimage 50, 1633–1638.
Kutas, M., Van Petten, C., and Kluender, R. (2006). “Psycholinguistics electrified II: 1994–2005,” in Handbook of Psycholinguistics, 2nd Edn, eds M. A. Gernsbacher, and M. Traxler (New York, NY: Elsevier Press), 659–724.
Lehtonen, M. H., Laine, M., Niemi, J., Thomsen, T., Vorobyev, V. A., and Hugdahl, K. (2005). Brain correlates of sentence translation in Finnish-Norwegian bilinguals. Neuroreport 16, 607–610.
Leonard, M. K., Brown, T. T., Travis, K. E., Gharapetian, L., Hagler, D. J. Jr., Dale, A. M., Elman, J. L., and Halgren, E. (2010). Spatiotemporal dynamics of bilingual word processing. Neuroimage 49, 3286–3294.
Marien, P., Abutalebi, J., Engelborghs, S., and De Deyn, P. P. (2005). Pathophysiology of language switching and mixing in an early bilingual child with subcortical aphasia. Neurocase 11, 385–398.
Meuter, R. F. I., and Allport, A. (1999). Bilingual language switching in naming: asymmetrical costs of language selection. J. Mem. Lang. 40, 25–40.
Moreno, E. M., Federmeier, K. D., and Kutas, M. (2002). Switching languages, switching palabras (words): an electrophysiological study of code switching. Brain Lang. 80, 188–207.
Moreno, E. M., Rodríguez-Fornells, A., and Laine, M. (2008). Event-related potentials (ERPs) in the study of bilingual language processing. J. Neurolinguistics 21, 477–508.
Moritz-Gasser, S., and Duffau, H. (2009a). Cognitive processes and neural basis of language switching: proposal of a new model. Neuroreport 20, 1577–1580.
Moritz-Gasser, S., and Duffau, H. (2009b). Evidence of a large-scale network underlying language switching: a brain stimulation study. J. Neurosurg. 111, 729–732.
Moser, B. (1978). “Simultaneous interpretation: a hypothetical model and its practical application,” in Language, Interpretation and Communication, eds D. Gerver, and H. W. Sinaiko (New York, NY: Plenum Press), 353–368.
Moser-Mercer, B., Frauenfelder, U. H., Casado, B., and Kunzli, A. (2000). “Searching to define expertise in interpreting,” in Language Processing and Simultaneous Interpreting: Interdisciplinary Perspectives, eds B. E. Dimitrova, and K. Hyltenstam (Amsterdam: John Benjamins), 107–132.
Nagel, I. E., Schumacher, E. H., Goebel, R., and D’esposito, M. (2008). Functional MRI investigation of verbal selection mechanisms in lateral prefrontal cortex. Neuroimage 43, 801–807.
Nardone, R., De Blasi, P., Bergmann, J., Caleri, F., Tezzon, F., Ladurner, G., Golaszewski, S., and Trinka, E. (2011). Theta burst stimulation of dorsolateral prefrontal cortex modulates pathological language switching: a case report. Neurosci. Lett. 487, 378–382.
Nieuwenhuis, S., Yeung, N., Van Den Wildenberg, W., and Ridderinkhof, K. R. (2003). Electrophysiological correlates of anterior cingulate function in a go/no-go task: effects of response conflict and trial type frequency. Cogn. Affect. Behav. Neurosci. 3, 17–26.
Osterhout, L., and Holcomb, P. J. (1992). Event-related brain potentials elicited by syntactic anomaly. J. Mem. Lang. 31, 785–806.
Paradis, M. (1993). Linguistic, psycholinguistic, and neurolinguistic aspects of “interference” in bilingual speakers: the activation threshold hypothesis. Int. J. Psycholinguistics 9, 133–145.
Paradis, M. (1996). Selective deficit in one language is not a demonstration of different anatomical representation: comments on Gomez-Tortosa et al. (1995). Brain Lang. 54, 170–173; discussion 174–175.
Paradis, M. (2001). An integrated neurolinguistic theory of bilingualism (1976–2000). Paper Presented at the LACUS Forum 27, Houston, TX.
Picton, T. W. (1992). The P300 wave of the human event-related potential. J. Clin. Neurophysiol. 9, 456.
Polich, J. (2007). Updating P300: an integrative theory of P3a and P3b. Clin. Neurophysiol. 118, 2128–2148.
Polich, J., and Kok, A. (1995). Cognitive and biological determinants of P300: an integrative review. Biol. Psychol. 41, 103–146.
Price, C. J., Green, D. W., and Von Studnitz, R. (1999). A functional imaging study of translation and language switching. Brain 122(Pt 12), 2221–2235.
Prior, A., and MacWhinney, B. (2010). A bilingual advantage in task switching. Biling. Lang. Cogn. 13, 253–262.
Proverbio, A. M., Leoni, G., and Zani, A. (2004). Language switching mechanisms in simultaneous interpreters: an ERP study. Neuropsychologia 42, 1636–1656.
Quaresima, V., Ferrari, M., Van Der Sluijs, M. C. P., Menssen, J., and Colier, W. (2002). Lateral frontal cortex oxygenation changes during translation and language switching revealed by non-invasive near-infrared multi-point measurements. Brain Res. Bull. 59, 235–243.
Rinne, J. O., Tommola, J., Laine, M., Krause, B. J., Schmidt, D., Kaasinen, V., Teras, M., Sipila, H., and Sunnari, M. (2000). The translating brain: cerebral activation patterns during simultaneous interpreting. Neurosci. Lett. 294, 85–88.
Rodriguez-Fornells, A., Balaguer, R. D. D., and Münte, T. F. (2006). Executive control in bilingual language processing. Lang. Learn. 56, 133–190.
Rodriguez-Fornells, A., Rotte, M., Heinze, H. J., Nösselt, T., and Münte, T. F. (2002). Brain potential and functional MRI evidence for how to handle two languages with one brain. Nature 415, 1026–1029.
Schumacher, E. H., Cole, M. W., and D’esposito, M. (2007). Selection and maintenance of stimulus-response rules during preparation and performance of a spatial choice-reaction task. Brain Res. 1136, 77–87.
Sebastian-Galles, N., Rodriguez-Fornells, A., De Diego-Balaguer, R., and Diaz, B. (2006). First- and second-language phonological representations in the mental lexicon. J. Cogn. Neurosci. 18, 1277–1291.
Steinhauer, K., and Connolly, J. F. (2008). “Event-related potentials in the study of language,” in Handbook of the Neuroscience of Language, eds B. Stemmer, and H. Whitaker (New York, NY: Elsevier), 91–104.
Takakusaki, K., Saitoh, K., Harada, H., and Kashiwayanagi, M. (2004). Role of basal ganglia-brainstem pathways in the control of motor behaviors. Neurosci. Res. 50, 137–151.
Van Heuven, W., and Dijkstra, T. (2010). Language comprehension in the bilingual brain: fMRI and ERP support for psycholinguistic models. Brain Res. Rev. 64, 104–122.
Wang, Y., Kuhl, P. K., Chen, C., and Dong, Q. (2009). Sustained and transient language control in the bilingual brain. Neuroimage 47, 414–422.
Keywords: executive control, bilingualism, dorsolateral prefrontal cortex, parietal lobe, basal ganglia, anterior cingulate cortex, language switching, simultaneous interpretation
Citation: Hervais-Adelman AG, Moser-Mercer B and Golestani N (2011) Executive control of language in the bilingual brain: integrating the evidence from neuroimaging to neuropsychology. Front. Psychology 2:234. doi: 10.3389/fpsyg.2011.00234
Received: 08 March 2011; Accepted: 29 August 2011;
Published online: 15 September 2011.
Edited by:
Ingrid Christoffels, University of Leiden, NetherlandsReviewed by:
Jubin Abutalebi, Vita Salute San Raffaele University, ItalySylvain Moreno, The Rotman Research Institute, Canada
Copyright: © 2011 Hervais-Adelman, Moser-Mercer and Golestani. This is an open-access article subject to a non-exclusive license between the authors and Frontiers Media SA, which permits use, distribution and reproduction in other forums, provided the original authors and source are credited and other Frontiers conditions are complied with.
*Correspondence: Alexis Georges Hervais-Adelman, Functional Brain Mapping Laboratory, University of Geneva Medical School, rue Michel-Servet 1, CH-1211 Genève 4, Switzerland. e-mail:YWxleGlzLmFkZWxtYW5AdW5pZ2UuY2g=


