- 1 Program in Behavioral and Cognitive Neuroscience, Department of Psychology, University of Florida, Gainesville, FL, USA
- 2 Center for Smell and Taste, University of Florida, Gainesville, FL, USA
- 3 Department of Otolaryngology-Head and Neck Surgery, University of Florida, Gainesville, FL, USA
- 4 Department of Surgery, Duke University Medical Center, Durham, NC, USA
- 5 NIMH Center for the Study of Emotion and Attention, University of Florida, Gainesville, FL, USA
Recent studies lead to the conclusion that focused attention, through the activity of corticofugal and medial olivocochlear (MOC) efferent pathways, modulates activity at the most peripheral aspects of the auditory system within the cochlea. In two experiments, we investigated the effects of different intermodal attention manipulations on the response of outer hair cells (OHCs), and the control exerted by the MOC efferent system. The effect of the MOCs on OHC activity was characterized by measuring the amplitude and rapid adaptation time course of distortion product otoacoustic emissions (DPOAEs). In the first, DPOAE recordings were compared while participants were reading a book and counting the occurrence of the letter “a” (auditory-ignoring) and while counting either short- or long-duration eliciting tones (auditory-attending). In the second, DPOAEs were recorded while subjects watched muted movies with subtitles (auditory-ignoring/visual distraction) and were compared with DPOAEs recorded while subjects counted the same tones (auditory-attending) as in Experiment 1. In both Experiments 1 and 2, the absolute level of the averaged DPOAEs recorded during the auditory-ignoring condition was statistically higher than that recorded in the auditory-attending condition. Efferent-induced rapid adaptation was evident in all DPOAE contours, under all attention conditions, suggesting that two medial efferent processes act independently to determine rapid adaptation, which is unaffected by attention, and the overall DPOAE level, which is significantly affected by changes in the focus of attention.
Introduction
It is well established that attending to a stimulus – visual or auditory – results in relatively larger sensory responses in the corresponding cortical areas than when the same signal is ignored (c.f., Woldorf et al., 1987; Johnson and Zatorre, 2005; Kauramäki et al., 2007), yet the initial level within the central nervous system at which auditory attention operates remains a matter of some debate.
Electrophysiological investigations in humans have led to the conclusion that selective attention mechanisms do not affect subcortical processing, but, instead, modulate the initial stages of cortical input above the thalamus (Woldorf et al., 1987; Hillyard, 1993; Saupe et al., 2009). Consistent with this view, studies of shorter-latency potentials have failed to demonstrate an attentional modulation of either cochlear or brainstem responses (Hackley et al., 1990; Hirschhorn and Michie, 1990).
By contrast, recent work using cross-modal attention manipulations in experimental animals and humans has forced the conclusion that peripheral and pre-cortical stages of processing are affected by attention tasks. Studies in human and non-human animal subjects have shown that peripheral responses, including brainstem and cochlear evoked potentials, as well as otoacoustic emissions (OAEs) are decreased when ignoring auditory probes and attending to visual (c.f., Picton et al., 1971; Lukas, 1980, 1981; Puel et al., 1988; Méric and Collet, 1992, 1994; Giard et al., 1994, 2000; Maison et al., 2001; Delano et al., 2007). The observed decrease in auditory responses when attending to visual stimuli is, of course, appealing, as the same effect is observed cortically. Avan and Bonfils (1992) and Michie et al. (1996), on the other hand, reported that attending to visual tasks produced no consistent change in distortion product OAEs (DPOAEs), which may even show increases when ignoring auditory stimuli.
Physiological studies indicate that stimulation of the corticofugal pathways within the cortex can, through the medial olivocochlear (MOC) efferent tracts innervating outer hair cells (OHCs), alter cochlear activity in both humans and non-human animals (Xiao and Suga, 2002; Perrot et al., 2006; Delano et al., 2007; Liu et al., 2010), describing the neural basis for attentional modulation of the auditory receptors (Fritz et al., 2007; Knudsen, 2007; Suga, 2008). Manipulating laterality, de Boer and Thornton (2007) and Harkrider and Bowers (2009) showed that attending to the eliciting stimulus reduces the magnitude of MOC suppression of click-evoked OAEs produced by contralateral noise; evidence of a release from MOC inhibition when attending to the ipsilateral ear.
While it is apparent that a variety of different attention manipulations do influence cochlear function, the nature of this influence remains unclear. Measurement of the rapid adaptation of DPOAEs offers a valuable, non-invasive measure of corticofugal and MOC control of OHCs and cochlear function (Liberman et al., 1996; Kim et al., 2001; Bassim et al., 2003). In two experiments, we compared DPOAE rapid adaptation magnitudes and absolute levels under different conditions of auditory or visual attention in human listeners.
Materials and Methods
Subjects
Thirty-three human subjects participated in Experiment 1 and 12 subjects in Experiment 2; no subject participated in both experiments. A brief history was taken from each subject to document ear-related complaints and history of ear infections, ear surgery, ototoxic medication use, noise exposure, and chronic medication use. The subjects were college students between the ages of 18 and 22 years and most were females (76% in Experiment 1 and 75% in Experiment 2).
All experiments were approved by the Institutional Review Boards of Duke University Medical Center and the University of Florida.
Instrumentation, Stimulus Parameters, and Testing Paradigms
The equipment, stimuli, and DPOAE recording procedures are the same as those employed in our previous work (c.f., Bassim et al., 2003). Each ear was fitted with two transducers; the two primary tones were generated digitally (Tucker-Davis Technologies, Gainesville, FL, USA) and fed individually to each transducer (Etymotic Research, Elk Grove Village, IL, USA). The primaries and emissions were measured in the ear canal with a low-noise microphone probe (Etymotic Research), sampled continuously at a rate of 40.9 kHz and amplified (40 dB), digitized (Tucker-Davis Technologies), and stored to the hard drive. At the end of the session, emission strength was analyzed using the heterodyne method as described by Kim et al. (2001).
The primary tone parameters used were f2/f1 = 1.21, f1 level (L1) = 70 dB SPL, and f2 level (L2) = 65 dB SPL. DPOAE levels were measured as the f2 frequency was stepped between 1.0 and 10.0 kHz and the f2 frequency producing the largest emission in either ear was selected for further study in a given session; the same stimuli that produced the largest, chosen DPOAE were presented binaurally to produce the largest DPOAE in the selected ear (the ear with the largest DPOAE). Responses from the non-selected ear, though collected, were not further analyzed. Duration of the primary tones for the reading and counting sessions was the same for both conditions tested and was either 3 or 6 s and, in all cases, the inter-stimulus interval was 3.8 s. Using long-duration tones, as opposed to transient or click stimuli, to measure DPOAEs offers the advantage of characterizing the MOC onset time course. The rise/fall times of all stimuli were zero, with the primaries beginning at 0° of phase in order to minimize the effects of frequency splatter. This splatter was further minimized because the DPOAE amplitude was measured as the 2f1 − f2 frequency peak in the Fast Fourier Transform (11.92 Hz bandwidth).
Once the ear tip transducers were fully seated within the ear canal, the acoustic system output was calibrated in the subject’s ear at the start of each session and calibration tests were repeated throughout the test session to detect the emergence of small changes in probe placement or orientation. Additionally, the measurement system monitored average primary tone levels on a stimulus-by-stimulus basis and any sudden change in the levels, usually indicating a displacement of the probe/microphone system in the ear canal, resulted in the session being interrupted. In these situations, the earphones were re-positioned and another calibration was performed.
DPOAE Analysis
A real time spectral analysis was performed during each stimulus presentation to monitor the level of the primaries as well as for the presence of the 2f1 − f2 DPOAE, which, depending on subject noise levels, could be observed in most FFT traces. Estimation of the changes in DPOAE amplitude as a function of time was done by fitting a two-component exponential to the DPOAE adaptation curves. The use of a two-component exponential fit is based on previous studies in animals and humans (Kim et al., 2001; Bassim et al., 2003), and was fit using the MatLab curve-fitting tool. A participant’s data were included in the overall analysis only if the magnitude of the observed rapid adaptation was larger than the observed noise (i.e., the variation in DPOAE amplitude with time) in the adaptation contour. Overall DPOAE levels across attention conditions were estimated by averaging the last 15% of the data points in each trace (i.e., the data points from 2550 to 3000 ms). The results obtained for each subject under the testing conditions were compared statistically using the analyses of variance (ANOVA) with alpha set at 5%.
Experiment 1
Methods
Thirty-three subjects participated in this experiment. The amplitudes of DPOAEs were measured in both ears as the f2 frequency was stepped between 1.0 and 10.0 kHz (20 frequencies in a geometric progression), and, in order to maximize the response signal-to-noise ratio, the f2 frequency producing the largest emission was selected for further study in a given session; only these chosen responses, and not those from the opposite ear, were further analyzed. At least 128 stimuli were presented in each condition and the responses from the selected ear, the ear with the selected DPOAE, were averaged across subjects. To minimize the influence of any systematic effects such as sequence of presentation or fatigue, each session was divided into blocks of 128 runs and the order of the attention/distractor conditions was randomly varied across subjects.
Adaptation of the DPOAE was measured under two different conditions of attention/distraction. In the first condition, the participants were instructed to attend to the eliciting tones (auditory-attending condition). The tones were either 3 or 6 s in duration and the listener was asked to count the occurrences of the shorter-duration tones. An equal number of 3 and 6 s tones were presented in all attending conditions. In the second, auditory-ignoring condition, the subject was instructed to ignore the tones (the same 3- and 6-s tones described in the attending condition) and to read a passage in a book or magazine of their choice, counting the occurrences of the letter “a.” Given the exploratory nature of the present study, intermodal target detection tasks were selected to maximize the potential effects of attention focus in this first study of DPOAEs and attention. Thus, we compared an auditory task condition (tone-counting) with a visual task condition (letter counting), rather than with a no-task condition. The DPOAEs to at least 128-tone presentations were averaged in each condition (using only the first 3 s of the 6-s tones).
Results
Distortion product otoacoustic emissions measured under the auditory-attending (counting tones; red trace) and auditory-ignoring (reading text; blue trace) conditions, averaged across subject, are presented in the upper panel of Figure 1. Each trace is fitted with a two-component exponential for use in estimating the magnitude and time constants of the MOC-mediated rapid adaptation response. Both DPOAE curves show the characteristic MOC-mediated rapid adaptation response after primary tone onset (Liberman et al., 1996; Kim et al., 2001; Bassim et al., 2003). The absolute level of the DPOAE recorded during the auditory-ignoring condition, however, is significantly higher in level when compared with the DPOAE recorded while the subjects were counting (auditory-attending) the 3-s tones (p < 0.05; paired t-test). The difference in the level of the two contours was ∼0.3 dB.
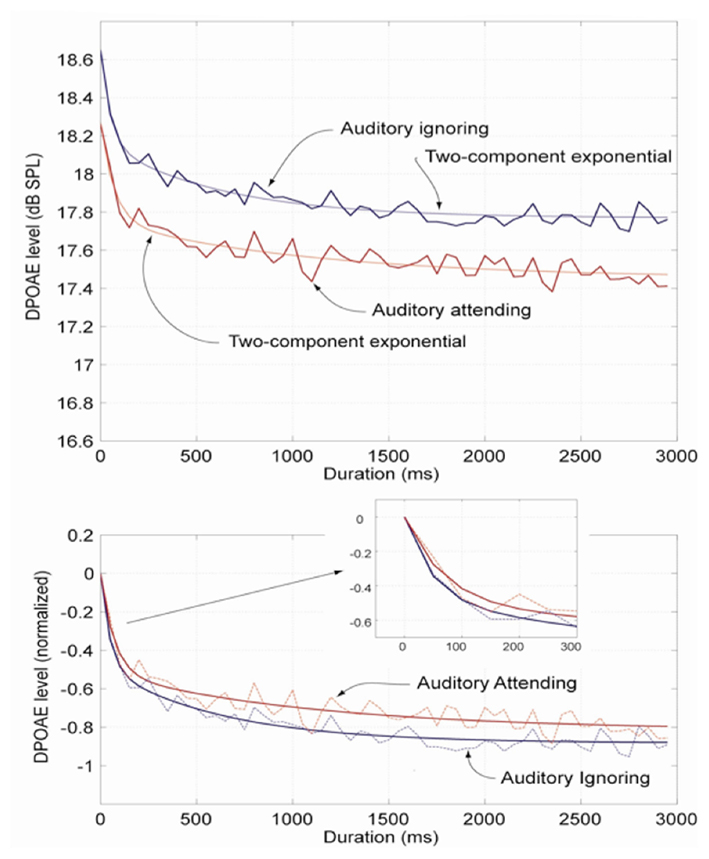
Figure 1. Averaged group DPOAE traces, Experiment 1 (top panel). The mean DPOAE trace measured in the auditory-ignoring (text reading) condition is shown in blue and the mean DPOAE trace recorded in the auditory-attending (tone-counting) condition is shown in red. Each trace is fitted with a two-component exponential for use in estimating the magnitude and time constants of the MOC-mediated rapid adaptation response. When the two DPOAE traces are normalized to the DPOAE onset level (bottom panel, and inset), it can be seen that the onset slope is relatively steeper and there is relatively more rapid adaptation for the auditory-ignoring condition (blue) compared with the auditory-attending (red) condition. The estimated time constants are 49 ms for the auditory-ignoring condition and 66 ms for the auditory-attending condition, differences that are statistically indistinguishable (paired t-test). The estimated difference in overall DPOAE level is ∼0.3 dB.
When the two mean DPOAE traces are normalized to initial onset level (lower panel, Figure 1), it can be seen that the magnitude of the rapid adaptation is relatively greater for the auditory-ignoring DPOAE; although, when compared, the magnitude of adaptation, ∼0.8 dB for both functions, is not statistically different (paired t-test). Likewise, when the normalized contour onsets are compared (Figure 1, lower panel inset), it can also be seen that the onset of adaptation is relatively steeper, the rapid adaptation time constant being relatively shorter (i.e., the decreasing adaptation slope is relatively steeper) for the auditory-ignoring condition compared with the auditory-attending DPOAE trace. The measured rapid adaptation time constants are 49 ms for the auditory-ignoring (reading text) condition compared with 66 ms for the auditory-attending condition. A statistical comparison showed that the two time constants were statistically indistinguishable (paired t-test).
Figure 2 presents a comparison of the absolute levels of the DPOAEs, for both Experiment 1 attending conditions, for each individual subject. Of the 33 subjects, 18 showed relatively higher overall DPOAE levels for the auditory-ignoring condition, eight had relatively lower absolute DPOAE levels recorded during the auditory-attending condition, and seven were approximately the same.
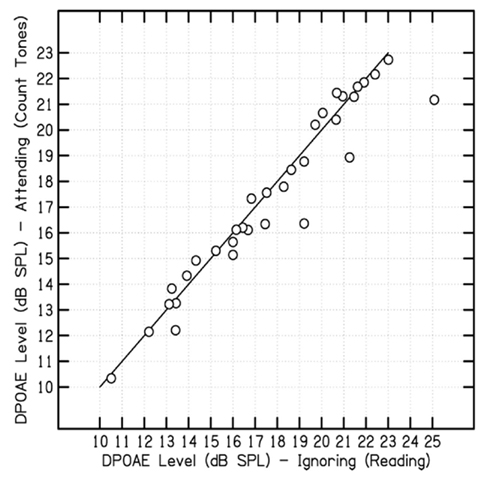
Figure 2. Scatter plot comparison of the absolute DPOAE level measured for each subject during the auditory-attending condition (counting tones) and DPOAE levels obtained during the auditory-ignoring condition (reading text) in Experiment 1.
Experiment 2
Methods
Twelve subjects, not involved in Experiment 1, participated in this experiment. As in Experiment 1, the primary tones were presented simultaneously to both ears to maximize the magnitude of the adaptation response (Liberman et al., 1996; Bassim et al., 2003). In this experiment all stimuli and conditions were similar, except the participants were instructed to read the subtitles on a (muted) movie of their choice in the auditory-ignoring condition. The DVD movie was played on a television in the sound room. In the auditory-attending (tone-counting) condition, in order to be certain that neither the television nor DVD player interfered with the recordings, both devices were left running, but were completely covered from view. The order of the two conditions was randomized across subjects, but both conditions were measured in the same recording session. The DPOAEs to at least 128-tone presentations were averaged in each condition (using only the first 3 s of the 6-s tones) and the results for all participants averaged.
Results
Distortion product otoacoustic emission curves measured in Experiment 2 are presented in the top panel in Figure 3. The DPOAE trace recorded in the auditory-ignoring (movie/subtitle viewing) condition is shown in green and the DPOAE trace recorded in the auditory-attending (tone-counting) condition is shown in red. Each trace is fitted with a two-component exponential.
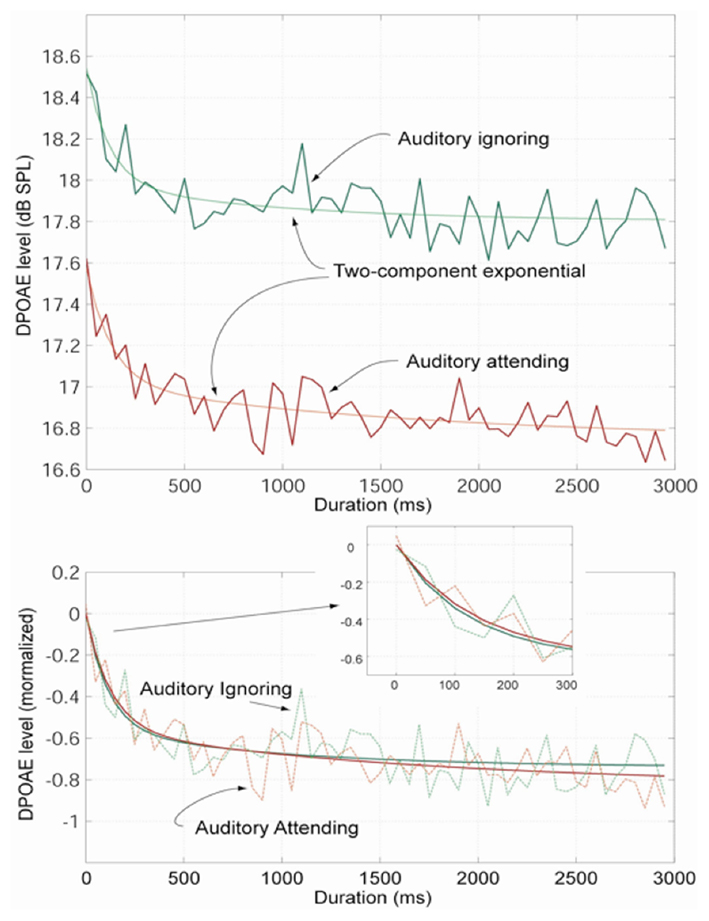
Figure 3. Distortion product otoacoustic emission traces, averaged across participants, from Experiment 2 (top panel). The mean DPOAE trace recorded in the auditory-ignoring (movie/subtitle viewing) condition is shown in green, and the mean DPOAE trace recorded in the auditory-attending (tone-counting) condition is shown in red. Each trace is fitted with a two-component exponential. The two DPOAE traces normalized to onset level are shown in the lower panel (and inset). Consistent with the findings of Experiment 1, the DPOAE adaptation curve recorded during the auditory-ignoring condition is significantly higher (p < 0.001) in overall level compared with that recorded during the auditory-attending condition (red). The calculated time constants are 114 ms for the auditory-ignoring condition and 127 ms for the auditory-attending condition. The absolute difference between the two traces is ∼1 dB.
Consistent with the findings of Experiment 1, the average DPOAE curve recorded during the auditory-ignoring condition is significantly higher (p < 0.001) in overall level compared with that recorded during the auditory-attending condition. The absolute difference between the two curves is ∼1 dB.
When the two DPOAE curves are normalized to onset level (Figure 3, lower panel and inset), it can be seen that there is a small, statistically insignificant (paired t-test) difference in the rapid adaptation onset time constants, with the auditory-ignoring adaptation onset (green) being relatively steeper. The time constants are 114 ms for the auditory-ignoring condition and 127 ms for the auditory-attending condition.
Figure 4 presents a scatter plot of the DPOAE absolute levels for the auditory-attending and auditory-ignoring conditions from Experiment 2. Of the DPOAEs recorded from 12 subjects in this experiment, 11 showed DPOAEs with absolute levels that were relatively higher under the auditory-ignoring condition, compared with the level of the DPOAE recorded in the same session when the subjects were attending to the DPOAE-eliciting tones.
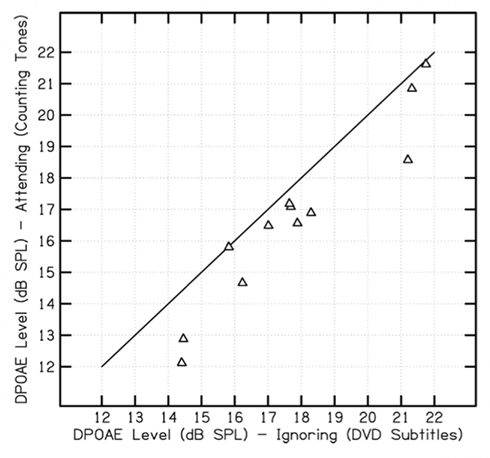
Figure 4. Scatter plot comparison of absolute DPOAE levels recorded from individual participants during auditory-attending (counting tones) and auditory-ignoring conditions (watching muted DVD, reading subtitles) conditions in Experiment 2.
The substantial difference in the absolute level of the DPOAEs recorded under the same auditory-attending conditions in Experiments 1 and 2 highlights the inter-subject variability evident in the recordings and illustrates the need to compare across conditions within a subject and given test session. This finding is consistent with reports from other investigators (c.f., Meric et al., 1996).
Discussion
Given the known innervation pattern of the mammalian cochlea, the only neural pathway by which changes in attention can influence OHC receptor function is through the corticofugal pathway and MOC fibers innervating the base of OHCs. MOC neurons exert remarkable control over the response of the cochlea to sound (c.f., Guinan, 1996, 2006, 2010; Robertson, 2009). The time course of MOC modulation of OHC responses can be estimated non-invasively by recording DPOAEs to long-duration tones (Liberman et al., 1996; Kim et al., 2001; Bassim et al., 2003). Using this measure to characterize the effects of different task manipulations, we show that changing cognitive task demands produces significant alterations in responding at the most peripheral aspects of the auditory system – in the response of the OHC receptors.
In both experiments, when DPOAE adaptation contours are compared across experimental conditions, they are significantly lower in overall level when participants attended to the eliciting tones (auditory-attending condition), compared with when listeners are asked to ignore the eliciting tones (e.g., when participants are asked to read a book or watch a muted movie); when reading, the mean DPOAE was approximately 0.3 dB higher than when participants were counting the brief tones (Figure 1, upper panel), and the group mean DPOAE level was 1.0 dB higher when participants were watching muted movies and reading subtitles (Figure 3, upper panel).
One obvious conclusion from this finding is that directing top-down attentional resources to a given modality (auditory, visual) may cause changes in the sensory periphery through descending pathways. Alternative interpretations are also possible however: participants may have found the tasks used to be different in difficulty, resulting in systematic alterations of effort or arousal. It is well conceivable that such confounding variables are at the core of the robust effects seen in our experiments. These alternative explanations cannot be ruled out at this point and require additional experimentation, now under way in our laboratory.
It is interesting to note that, although significant changes in the absolute level of the DPOAEs were observed, we found no significant corresponding changes in either the time constant or the magnitude of the rapid adaptation process, both of which are known to be under MOC control (Liberman et al., 1996): changes in the onset slope of the adaptation are apparent (see inset panels, Figures 1 and 3), but these were small and did not reach statistical significance. This result was unexpected because our initial hypothesis was, like the more central and cortical reports, that the MOC system mediates a peripheral selective attention mechanism in the cochlea via the suppression of ignored stimuli. Indeed, that hypothesis drove the use of the DPOAE rapid adaptation recording technique in this study. This finding might indicate, as in the rat, that the rapid adaptation response is produced predominantly by the middle ear acoustic reflex, and not by the MOC system (Relkin et al., 2005). In our earlier report, however, we argued against the involvement of the middle ear muscles by demonstrating that the stimuli employed in these measures were below thresholds for activation of the acoustic reflex (Bassim et al., 2003). However unlikely, we cannot emphatically rule out an MEM involvement in the present results because we did not directly measure middle ear responses as was done in our original report.
Our present results, showing that rapid adaptation is still apparent in the DPOAE responses to experimental conditions, suggests that those MOC processes influencing the overall level of the DPOAE contour and those mediated rapid adaptation mechanism may operate separately and simultaneously. From these data it can be argued that the MOC tracts serve two different, but related functions, with both serving to increase the signal-to-noise ratio of the cochlea. The first, rapid adaptation, suppresses the response of OHCs to sustained or repeated stimulation and, as a consequence, increases responding to new or transient signals (Liberman et al., 1996). The second component, via task-related top-down signals, may for instance increase the salience of attended signals at the expense of ignored stimuli. These conjectures are subject to ongoing research in our laboratory. If one embraces an attentional explanation for the present data, it is important to point out that, given the suppressive nature of MOC action on OHCs (Guinan, 1996, 2006, 2010; Robertson, 2009), our data argue that the MOC is actively, in some manner, suppressing the OHC responses to the attended auditory signals.
The observed increase in the amplitude of cochlear responses to ignored auditory stimuli is consistent with the findings from several studies by Michie et al. (1996). In their work, Michie and colleagues attempted to replicate several findings from groups using different attention and stimulus paradigms – all of which reported a relative decrease in the amplitude of ignored responses (Puel et al., 1988; Giard et al., 1994, 2000); this was the expected peripheral consequence of ignoring a stimulus as this effect is the generally accepted effect observed cortically (Woldorf et al., 1987; Johnson and Zatorre, 2005; Kauramäki et al., 2007). Interestingly, in four of five experiments, they demonstrated statistical increases in the level of evoked OAE responses to ignored auditory stimuli.
Our observation of a relative increase in response magnitude when participants are instructed to ignore the auditory stimulus, however, is in apparent disagreement with previous results from a number of investigators employing different cognitive paradigms and varied physiological measures (c.f., Oatman, 1971, 1976; Oatman and Anderson, 1977; Lukas, 1980, 1981; Puel et al., 1988; Giard et al., 1994; Maison et al., 2001; Delano et al., 2007). In an effort to maximize task-related differences, the present study compared visual attending directly with different auditory-attending tasks, as described above. The potential disadvantages of this approach are mentioned above. Several previous studies that may inform the present research however employed paradigms that compared attention conditions against a no-task condition, resulting in similar limitations: Oatman (1971, 1976) and Oatman and Anderson (1977) compared auditory evoked potentials to tone bursts in cats while they were focusing attention on a visual task with responses to the same tones when they were performing no-task; Cortical and cochlear nucleus responses were relatively smaller when recorded during the visual attention task. Using the same stimulus paradigm in humans, Lukas (1980) likewise reported that auditory evoked potentials in humans were relatively lower in level when subjects performed a visual task (and ignored the auditory stimuli), compared to when they performed no attentional task during the auditory recordings. Because of concerns over the differential effects of arousal on the active visual task, compared with the passive auditory condition, Lukas (1981) repeated their earlier work, with the added auditory condition that participants were required to count tones varying in frequency, and showed a similar, relatively smaller physiological response to ignored auditory stimuli. Delano et al. (2007), employed a intermodal paradigm, similar to that used by Lukas (1981), in behaviorally trained chinchillas and also showed consistently smaller, longer-latency compound action potentials, to ignored tone bursts.
At this time, the reasons for the differing findings, whether ignored responses are relatively lower (Lukas, 1981; Delano et al., 2007) or relatively higher (Michie et al., 1996; present data) are uncertain. At least two issues, however, are worthy of additional study. The first is that our study and that of Michie et al. (1996) employed OAE measures, while Lukas (1981) and Delano et al. (2007) both used compound action potentials to assess changes in sensitivity. Both measures have been shown to be sensitive to efferent influence (c.f., Guinan, 1996, 2006), so it is unclear how this measurement technique, alone, might explain the observed differences.
The second issue is likely more important, both our study and Michie et al. (1996), the only studies showing an increase in the amplitude of responses to ignored stimuli, employed the same stimulus as both the target and for generating the physiological measure, whereas the two studies that showed a relative decrease in the amplitude of ignored stimuli used different target and response-evoking stimuli. Delano et al. (2007) used a 100-μs click to evoke the compound action potentials and 100–200 ms-long tones (1, 2, 4, 6, 8 kHz) to evoke the cochlear microphonics, while their target stimuli were 3810 ms-long tones of 1200 and 3600 Hz. The other study, Lukas (1981), employed two different click stimuli, varying in duration and, as a consequence, having differing frequency spectra. There are data from multiple studies showing that the effects of attention are significantly different at frequencies away from the expected, or target frequency (Greenberg and Larkin, 1968; Dai et al., 1991; Strickland and Viemeister, 1995; Scharf et al., 1997), compared with the response to the target frequency. In this case, when the target and evoking stimulus are different, the effects of the descending input on OHC sensitivity may be different at frequencies away from the attended signal, and the effect of attention may not be evident in the measurements. Indeed, Delano et al. (2007) suggest this is why they might have failed to show an effect of attention on their auditory task condition.
As stated above, another methodological question relates to the fact that comparisons between auditory-attending (counting) tones on one hand and counting visual letters or being distracted by visual media with subtitles might be confounded by factors other than attention. Most importantly, it is conceivable that overall non-specific arousal states of an individual can affect MOC and OHC activity. Because it cannot be ruled out that conditions of tone-counting, letter counting, and movie watching differ along dimensions of arousal or task engagement, we conducted an additional control experiment (Smith, unpublished observations) in which participants counted tones with and without distraction by strong emotional visual stimuli taken from the International Affective Picture System (IAPS). These data replicate the present findings, but show no effect of emotional engagement or physiological arousal as induced by the IAPS, on DPOAEs.
There has long been a debate about the ability of the MOC to influence the operating point of the receptor (c.f., Wiederhold and Kiang, 1970; Guinan and Gifford, 1988; Rajan et al., 1990) although, with the possible exception of this report, few data support this suggestion (Xie and Henson, 1998). At this juncture, it is indisputable that cognitive task demands can have significant effects on OHC function and, as a consequence, on the responses of the auditory system as a whole. Such effects are mediated through the corticofugal pathways, with the last relay via the MOC efferent tracts. How the changes in sensitivity in the cochlea with cognitive state are reflected in, or account for, the changes reported at higher, cortical centers remains to be determined.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We are indebted to Miriam and Bill Henson, Jean-Marie Aran, and E. Christopher Kirk for insightful discussions concerning the role of the medial efferents in hearing and techniques for measuring efferent function. We are also grateful to Roger Miller for development of the DPOAE recording facilities and Sridhar Srinivasan for assistance with the data analysis. Present address for Rony K. Aouad is Department of Otolaryngology, University of Kentucky Medical Center, Lexington, Kentucky, 40536. This work was supported in part by NIH NIDCD DC02832 to David W. Smith.
Abbreviations
DPOAE, distortion product otoacoustic emissions; MOC, medial olivocochlear efferent; OAE, otoacoustic emission; OHC, outer hair cell.
References
Avan, P., and Bonfils, P. (1992). Analysis of possible interactions of an attentional task with cochlear mechanics. Hear. Res. 57, 269–275.
Bassim, M. K., Miller, R. L., Buss, E., and Smith, D. W. (2003). Rapid adaptation of the cubic distortion tone emission in humans: monaural, binaural and contralateral stimulation effects. Hear. Res. 182, 140–152.
Dai, H., Scharf, B., and Buus, S. (1991). Effective attenuation of signals in noise under focused attention. J. Acoust. Soc. Am. 89, 2837–2842.
de Boer, J., and Thornton, A. R. D. (2007). Effect of subject task on contralateral suppression of click evoked otoacoustic emissions. Hear. Res. 233, 117–123.
Delano, P. H., Elgueda, D., Hamame, C. M., and Robles, L. (2007). Selective attention to visual stimuli reduces cochlear sensitivity in chinchillas. J. Neurosci. 27, 4146–4153.
Fritz, J. B., Elhilalia, M., David, S. V., and Shamma, S. A. (2007). Auditory attention – focusing the searchlight on sound. Curr. Opin. Neurobiol. 17, 437–455.
Giard, M. H., Collet, L., Bouchet, P., and Pernier, J. (1994). Auditory selective attention in the human cochlea. Brain Res. 633, 353–356.
Giard, M. H., Fort, A., Mouchetant-Rostaing, Y., and Pernier, J. (2000). Neurophysiological mechanisms of auditory selective attention in humans. Front. Biosci. 5, 84–94.
Greenberg, G. Z., and Larkin, W. D. (1968). Frequency-response characteristic of auditory observers detecting signals of a single frequency in noise: the probe-signal method. J. Acoust. Soc. Am. 44, 1513–1523.
Guinan, J. J. Jr. (1996). “Physiology of olivocochlear efferents,” in The Cochlea, eds P. Dallos, A. N. Popper, and R. R. Fay (New York: Springer-Verlag), 435–502.
Guinan, J. J. Jr. (2006). Olivocochlear efferents: anatomy, physiology, function, and the measurement of efferent effects in humans. Ear Hear. 27, 589–607.
Guinan, J. J. Jr. (2010). Cochlear efferent innervation and function. Curr. Opin. Otolaryngol. Head Neck Surg. 18, 447–453.
Guinan, J. J. Jr., and Gifford, M. L. (1988). Effects of electrical stimulation of efferent olivocochlear neurons on cat auditory-nerve fibers. I. Rate-level functions. Hear. Res. 33, 97–114.
Hackley, S. A., Woldorf, M., and Hillyard, S. A. (1990). Cross-modal selective attention effects on retinal, myogenic, brainstem and cerebral evoked potentials. Psychophysiology 27, 195–208.
Harkrider, A. W., and Bowers, C. D. (2009). Evidence for a cortically mediated release from inhibition in the human cochlea. J. Am. Acad. Audiol. 20, 208–215.
Hillyard, S. A. (1993). Electrical and magnetic brain recordings: contributions to cognitive neuroscience. Curr. Opin. Neurobiol. 3, 217–224.
Hirschhorn, T. N., and Michie, P. T. (1990). Brainstem auditory evoked potentials (BAEPs) and selective attention revisited. Psychophysiology 27, 495–512.
Johnson, J. A., and Zatorre, R. J. (2005). Attention to simultaneous unrelated auditory and visual events: behavioral and neural correlates. Cereb. Cortex 15, 1609–1620.
Kauramäki, J., Jääskeläinen, I. P., and Sams, M. (2007). Selective attention increases both gain and feature selectivity of the human auditory cortex. PLoS ONE 2, e909. doi: 10.1371/journal.pone.0000909
Kim, D. O., Dorn, P. A., Neely, S. T., and Gorga, M. P. (2001). Adaptation of distortion product otoacoustic emissions in humans. J. Assoc. Res. Otolaryngol. 2, 31–40.
Liberman, M. C., Puria, S., and Guinan, J. J. Jr. (1996). The ipsilaterally evoked olivocochlear reflex causes rapid adaptation of the 2f1-f2 distortion product otoacoustic emission. J. Acoust. Soc. Am. 99, 2572–2584.
Liu, X., Yan, Y., Wang, Y., and Yan, J. (2010). Corticofugal modulation of initial neural processing of sound information from the ipsilateral ear in the mouse. PLoS ONE 5, e14038. doi: 10.1371/journal.pone.0014038
Lukas, J. H. (1980). Human auditory attention: the olivocochlear bundle may function as a peripheral filter. Psychophysiology 17, 444–452.
Lukas, J. H. (1981). The role of efferent inhibition in human auditory attention: an examination of the auditory brainstem potentials. Int. J. Neurosci. 12, 137–145.
Maison, S., Micheyl, C., and Collet, L. (2001). Influence of focused auditory attention on cochlear activity in humans. Psychophysiology 38, 35–40.
Méric, C., and Collet, L. (1992). Visual attention and evoked otoacoustic emissions: a slight but real effect. Int. J. Psychophysiol. 12, 233–235.
Méric, C., and Collet, L. (1994). Differential effects of visual attention on spontaneous and evoked otoacoustic emissions. Int. J. Psychophysiol. 17, 281–289.
Meric, C., Micheyl, C., and Collet, L. (1996). Attention and evoked otoacoustic emissions: attempts at characterization of intersubject variation. Physiol. Behav. 59, 1–9.
Michie, P. T., LePage, E. L., Solowij, N., Haller, M., and Terry, L. (1996). Evoked otoacoustic emissions and auditory selective attention. Hear. Res. 98, 54–67.
Oatman, L. C. (1971). Role of visual attention on auditory evoked potentials in unanesthetized cats. Exp. Neurol. 32, 341–356.
Oatman, L. C. (1976). Effects of visual attention on the intensity of auditory evoked potentials. Exp. Neurol. 51, 41–53.
Oatman, L. C., and Anderson, B. W. (1977). Effects of visual attention on tone burst evoked auditory potentials. Exp. Neurol. 57, 200–211.
Perrot, X., Ryvlin, P., Isnard, J., Guénot, M., Catenoix, H., Fischer, C., Mauguière, F., and Collet, L. (2006). Evidence for corticofugal modulation of peripheral auditory activity in humans. Cereb. Cortex 16, 941–948.
Picton, T. W., Hillyard, S. A., Galambos, R., and Schiff, M. (1971). Human auditory attention: a central or peripheral process? Science 173, 351–353.
Puel, J. L., Bonfils, P., and Pujol, R. (1988). Selective attention modifies the active micromechanical properties of the cochlea. Brain Res. 44, 380–383.
Rajan, R., Robertson, D., and Johnstone, B. M. (1990). Absence of tonic activity of the crossed olivocochlear bundle in determining compound action potential thresholds, amplitudes and masking phenomena in anaesthetised guinea pigs with normal hearing sensitivities. Hear. Res. 44, 195–207.
Relkin, E. M., Sterns, A., Azeredo, W., Prieve, B. A., and Woods, C. I. (2005). Physiological mechanisms of onset adaptation and contralateral suppression of DPOAEs in the rat. J. Assoc. Res. Otolaryngol. 6, 119–135.
Robertson, D. (2009). Centrifugal control in mammalian hearing. Clin. Exp. Pharmacol. Physiol. 36, 603–611.
Saupe, K., Schröger, E., Andersen, S. K., and Müller, M. M. (2009). Neural mechanisms of intermodal sustained selective attention with concurrently presented auditory and visual stimuli. Front. Hum. Neurosci. 3:58. doi: 10.3389/neuro.09.058.2009
Scharf, B., Magnan, J., and Chays, A. (1997). On the role of the olivocochlear bundle in hearing: 16 case studies. Hear. Res. 103, 101–122.
Strickland, E. A., and Viemeister, N. F. (1995). An attempt to find psychophysical evidence for efferent action in humans. Abstr. Midwinter Res. Meet. Assoc. Res. Otolaryngol. 18, 173.
Suga, N. (2008). Role of corticofugal feedback in hearing. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 194, 169–183.
Wiederhold, M. L., and Kiang, N. Y. S. (1970). Effects of electrical stimulation of the crossed olivocochlear efferent bundle on single auditory-nerve fibers in the cat. J. Acoust. Soc. Am. 48, 950–965.
Woldorf, M. G., Hansen, J. C., and Hillyard, S. A. (1987). “Evidence for effects of selective attention in the mid-latency range of the human auditory event-related potential,” in Current Trends in Event Related Potential Research (EEG Suppl. 40), eds R. Jr. Johnson, J. W. Rohrbaugh, and R. Parasuraman (Amsterdam: Elsevier Science Publishers), 146–154.
Xiao, Z., and Suga, N. (2002). Modulation of cochlear hair cells by the auditory cortex in the mustached bat. Nat. Neurosci. 5, 57–63.
Keywords: corticofugal pathways, medial olivocochlear efferents, MOC, selective auditory attention, distortion product otoacoustic emission, DPOAE, human
Citation: Smith DW, Aouad RK and Keil A (2012) Cognitive task demands modulate the sensitivity of the human cochlea. Front. Psychology 3:30. doi: 10.3389/fpsyg.2012.00030
Received: 30 September 2011; Accepted: 24 January 2012;
Published online: 13 February 2012.
Edited by:
Andrew J. Oxenham, University of Minnesota, USAReviewed by:
Andrew J. Oxenham, University of Minnesota, USAGlenis Long, Graduate Center of the City University of New York, USA
John J. Guinan, Harvard Medical School, USA
Copyright: © 2012 Smith, Aouad and Keil. This is an open-access article distributed under the terms of the Creative Commons Attribution Non Commercial License, which permits non-commercial use, distribution, and reproduction in other forums, provided the original authors and source are credited.
*Correspondence: David W. Smith, Program in Behavioral and Cognitive Neuroscience, Department of Psychology, Box 112250, University of Florida, Gainesville, FL 32611, USA. e-mail:ZHdzbWl0aEB1ZmwuZWR1.


