- 1Department of Psychology, Concordia University, Montreal, QC, Canada
- 2Department of Psychology, University of York, York, UK
- 3School of Optometry and Vision Science, University of Waterloo, Waterloo, ON, Canada
- 4School of Optometry and Vision Science, The University of Auckland, Auckland, New Zealand
- 5Department of Psychology and Neuroscience Program, Colgate University, Hamilton, NY, USA
Transcranial Direct Current Stimulation (tDCS) has recently been employed in traditional psychophysical paradigms in an effort to measure direct manipulations on spatial frequency channel operations in the early visual system. However, the effects of tDCS on contrast sensitivity have only been measured at a single spatial frequency and orientation. Since contrast sensitivity is known to depend on spatial frequency and orientation, we ask how the effects of anodal and cathodal tDCS may vary according to these dimensions. We measured contrast sensitivity with sinusoidal gratings at four different spatial frequencies (0.5, 4, 8, and 12 cycles/°), two orientations (45° Oblique and Horizontal), and for two stimulus size conditions [fixed size (3°) and fixed period (1.5 cycles)]. Only contrast sensitivity measured with a 45° oblique grating with a spatial frequency of 8 cycles/° (period = 1.5 cycles) demonstrated clear polarity specific effects of tDCS, whereby cathodal tDCS increased and anodal tDCS decreased contrast sensitivity. Overall, effects of tDCS were largest for oblique stimuli presented at high spatial frequencies (i.e., 8 and 12 cycles/°), and were small or absent at lower spatial frequencies, other orientations and stimulus size. Thus, the impact of tDCS on contrast sensitivity, and therefore on spatial frequency channel operations, is opposite in direction to other behavioral effects of tDCS, and only measurable in stimuli that generally elicit lower contrast sensitivity (e.g., oblique gratings with period of 1.5 cycles at spatial frequencies above the peak of the contrast sensitivity function).
Introduction
Neuro-stimulation techniques have recently been combined with traditional psychophysical paradigms in an effort to obtain a measure of direct manipulation on spatial frequency channel operations in the early visual system (review: Antal et al., 2006). One technique that is gaining popularity due to its affordability and simplicity is transcranial Direct Current Stimulation (tDCS), a non-invasive brain stimulation technique that transiently modulates excitation and inhibition in the human brain via alterations in the membrane potential of neurons (Antal et al., 2001, 2006; Nitsche et al., 2008; Stagg et al., 2009; Stagg and Nitsche, 2011). The technique involves a stimulating device that delivers a mild direct current (DC) between two electrodes (anode and cathode) placed on the scalp of an observer, which creates a resistive DC circuit that induces a mild intra-cerebral electrical current from the anode where current enters cortex, to the cathode where current exits the cortex. The direction of current flow determines the effect of tDCS. Specifically, anodal stimulation (a-tDCS) generates a sub-threshold depolarization, while cathodal (c-tDCS) stimulation hyperpolarizes the membrane potential of neurons (Radman et al., 2009; Reato et al., 2010; Paulus, 2011; Stagg and Nitsche, 2011; Pellicciari et al., 2013; Rahman et al., 2013). Polarity specific behavioral effects of tDCS are well established in motor cortex (e.g., Jacobson et al., 2012). However, in primary visual cortex, it is typical to find either facilitatory or inhibitory effects due to a-tDCS or c-tDCS, but not both. Also, the polarity specific facilitation and inhibitory effects of tDCS may be opposite to those reported in motor cortex (Antal et al., 2001; Accornero et al., 2007; Lang et al., 2007; Chaieb et al., 2008; Spiegel et al., 2012; Peters et al., 2013; Pirulli et al., 2014). Part of the variability in tDCS effects for different cortical loci can be attributed to structural (e.g., cell type and morphology and the direction of current flow in relation to the somatodendritic axis), or functional differences between stimulated areas (Rushton, 1927; Ward and Weiskrantz, 1969; Shipp, 2005; Radman et al., 2009; Reato et al., 2010; Bikson et al., 2013). Given that the visual cortex is both structurally and functionally different from motor cortex, it should come as no surprise that the effects of tDCS over the visual cortex are less clear.
The application of a-tDCS over primary visual cortex has been shown to enhance contrast sensitivity in amblyopic persons (Spiegel et al., 2013) at spatial frequencies above the peak of the contrast sensitivity function (CSF) and near the peak of the CSF (Kraft et al., 2010) while inhibitory effects of c-tDCS (Antal et al., 2001; Chaieb et al., 2008) on contrast sensitivity have been found for spatial frequencies above the peak of the CSF. However, all previous studies of tDCS on contrast sensitivity presented a single spatial frequency to observers, and thus, the effect of tDCS on the shape of the CSF (Campbell et al., 1966; Graham, 1989; Peli et al., 1993), which involves multiple spatial frequencies, is currently unknown1. Furthermore, the influence of stimulus orientation on tDCS induced changes in contrast sensitivity has not been investigated.
The goal of the current study was to assess how the effects of tDCS vary according to the stimulus dimensions (spatial frequency and orientation) used to measure contrast sensitivity. Given the known functional organization of the early visual system, and the properties of the DC circuit generated by tDCS, certain predictions as to the interaction of tDCS and stimulus dimension can be made. First, the effects of tDCS on contrast sensitivity should be greatest at higher spatial frequencies, and diminish with decreasing spatial frequency. This is because tDCS exerts its greatest effect at cortical sites closet to the skull (Miranda et al., 2006, 2013; Rahman et al., 2013) and V1 neurons at the occipital pole (close to the skull) have higher preferred spatial frequencies than those located deeper within the calcarine sulcus (Tootell et al., 1981, 1988; De Valois et al., 1982; Foster et al., 1985; Engel et al., 1997; Horton, 2006; Henriksson et al., 2008; Yu et al., 2010). Cells further from the occipital pole have receptive fields located peripherally in the visual field, which means that stimuli presented further than 2° eccentricity from fovea may not be affected as strongly by tDCS than stimuli presented in the central visual field (Kraft et al., 2010; but see Costa et al., 2015 for a contrasting view). Stimulus orientation may also influence the effect of tDCS on contrast sensitivity. Contrast sensitivity to oblique gratings is lower than that to horizontal gratings (the “Oblique Effect”; Campbell et al., 1966; Appelle, 1972; Essock, 1980). Therefore, contrast sensitivity to oblique gratings may be more susceptible to the facilitatory effects of a-tDCS whereas horizontal gratings may be more susceptible to the inhibitory effects of c-tDCS. This, in essence, should decrease the magnitude of the “Oblique Effect”. Thus, we measured changes in contrast sensitivity from a non-stimulation baseline under both a-tDCS and c-tDCS to gratings of four different spatial frequencies that spanned the CSF (0.5, 4, 8, and 12 cycles/°) and two stimulus orientations (45° oblique or Horizontal).
Materials and Methods
Participants
Twenty-six undergraduate students participated at baseline, out of which 20 continued onto the tDCS portion of this study. All observers but two were naïve to the goals of the experiment. Observers were prevented from moving onto the tDCS sessions when their contrast detection thresholds measured just prior to the application of tDCS exceeded 2 SDs of their average thresholds measured at baseline. Participants that continued onto the tDCS sessions were separated into two groups; 10 (Nfemale = 7, Mage = 20.2) participants were presented with oblique gratings while the other 10 (Nfemale = 5, Mage = 20.5) saw horizontal gratings. Two of the participants in the oblique orientation group completed the experiment at Concordia University (Montreal, QC, Canada), while data for all other participants in this study were collected at Colgate University (Hamilton, NY, USA). All participants had normal, or corrected-to-normal visual acuity (Snellen cutoff = 20/25) and no astigmatism. Written informed consent was obtained from all participants and all were treated in accordance to the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (Medical Research Council of Canada, 2003) and the ethical standards of the Federal Code of Regulations Title 45 (Public Welfare) and Department of Health and Humans Services, Part 46 (Protection of Human Subjects). All participants were compensated financially for their time.
Apparatus
All stimuli were presented on 22.5″ Viewsonic (G225fB) monitors driven by a dual core Intel® Xeon® processor (1.60 GHz x2) equipped with 4GB RAM and a 256MB PCIe x16 ATI FireGL V7200 dual DVI/VGA graphics card with 8-bit grayscale resolution at Colgate University and an Apple Mac Pro (2x 2.66 GHz processor) equipped with 8GB of RAM and a 1GB PCIe x16 ATI Radeon HD 5770 Graphics card with 8-bit grayscale resolution. The color management settings for the graphics card (i.e., 3D display settings) were adjusted such that the luminance “gain” of the green gun was twice that of the red gun, which was set to twice that of the blue gun. A bit-stealing algorithm (Tyler, 1997; Bex et al., 2007) was employed to yield 10.8 bits of luminance (i.e., grayscale) resolution (i.e., 1785 unique levels) distributed evenly across a 0–255 scale. Stimuli were displayed using a linearized look-up table, generated by calibrating with a Color-Vision Spyder3 Pro sensor. Maximum luminance output of both display monitors was 100 cd/m2 (50 cd/m2 mean luminance after calibration). The frame refresh rate was set to 85 Hz (100 Hz at Concordia), and the resolution was set to 1600 × 1200 pixels (1024 × 768 pixels at Concordia). Single pixels subtended 0.0134° (0.0381° at Concordia) of visual angle, i.e., 0.80 arc min. (2.28 arc min at Concordia) as viewed from 1.0 m. Head position was maintained with a chin rest. Participants viewed the display monitor from 2 m in a dark room through an aperture (16° of visual angle in diameter) of a large black circular mask that was fit to the monitor bezel in order to obscure any monitor or room orientation cues.
Transcranial Direct Current was generated with a 9V battery driven direct current stimulator (Chattanooga Ionto, USA) and delivered via a pair of carbon-rubber electrodes (The Magstim Company Ltd., UK). The electrodes were encased in potassium chloride soaked Spontex sponge pockets (The Magstim Company Ltd., UK). The size of the stimulating electrode was 6 cm × 8 cm, and the size of the reference electrode was 12 cm × 8 cm. The larger size of the reference electrode renders it inert due to low current density (Nitsche et al., 2007; Spiegel et al., 2012). Both electrodes were held in place with four Magstim rubber headbands (The Magstim Company Ltd., UK), applied in a manner that maximized complete electrode sponge surface contact over the targeted scalp regions.
Stimuli
Stimuli consisted of foveally presented sinusoidal gratings generated at one of two orientations: either oblique (45°) or horizontal (90°). All gratings were windowed by a 2D Gaussian, which ramped down the contrast to mean luminance. Stimulus spatial frequency was 0.5, 4, 8, or 12 cycles/°, with a period of 1.5 cycles (fixed period condition). The electrical field generated by tDCS is prominently focused onto the surface of the visual cortex, which limits the spatial extent of the visual field modulated by tDCS to the central 1–2° of the visual field (Kraft et al., 2010)2. As the effects of tDCS change as both a function of spatial frequency and stimulus area, we added a second stimulus condition and measured contrast sensitivity with a fixed stimulus size (3°), and adjusted the period of the stimulus with spatial frequency (fixed stimulus size condition). All stimuli were surrounded by a low contrast ring (Michelson Contrast = 10%) 1 pixel in size, 0.78° away from the border of the grating, and paired with a low frequency tone; both served to minimize participant doubt as to the location and/or presence of the stimulus on the screen. Stimulus contrast was expressed as Michelson contrast = [(Lmax - Lmin)/(Lmax + Lmin)] scaled to have zero mean and then normalized to 1.0.
Psychophysical Procedure
The within-subject stimulus conditions for this experiment consisted of four spatial frequencies (0.5, 4, 8, and 12 cycles/°), and two period conditions (fixed period and fixed size). Observers were grouped according to the stimulus orientation (45° oblique or horizontal). The psychophysical procedure for both the training and test phases were identical. The stimulus presentation consisted of a 2-Interval Force Choice (2-IFC) procedure where participants had to indicate the interval, either the first or the second, which contained the target. Target contrast was controlled by a 2-up, 1-down staircase setup and controlled by the PAL_AMUD_setupUD and the PAL_AMUD_updateUD functions from the Palamedes toolbox for MATLAB (Prins and Kingdom, 2009; Kingdom and Prins, 2010). Threshold was approached from above with a target contrast step size of 0.05% Michelson contrast. Each staircase ran until 12 reversals were observed and the averaged target contrast value of the last five reversals was used as an estimate of target contrast threshold (70.71% correct on the psychometric function).
All staircases completed by observers began with an instruction screen that informed them of the spatial frequency and size condition of the stimulus (orientation never changed within observers). Each trial began with a black fixation dot (0.1°) presented at the center of the screen. The fixation dot served both to remind the observer a stimulus will appear shortly and the location of said stimulus. The fixation screen (300 ms) was followed by a blank screen (150 ms) set to mean luminance, followed by the first stimulus interval (onset followed a square-wave function) presented for 150 ms. This sequence was repeated for the second stimulus interval (see Figure 1). One interval contained the stimulus, surrounded by a low-contrast ring, while the other interval contained only a low-contrast ring. Participants indicated, via keyboard press, the interval that they believed contained the target. The duration of the response interval was unlimited, and participants received no feedback on their accuracy.
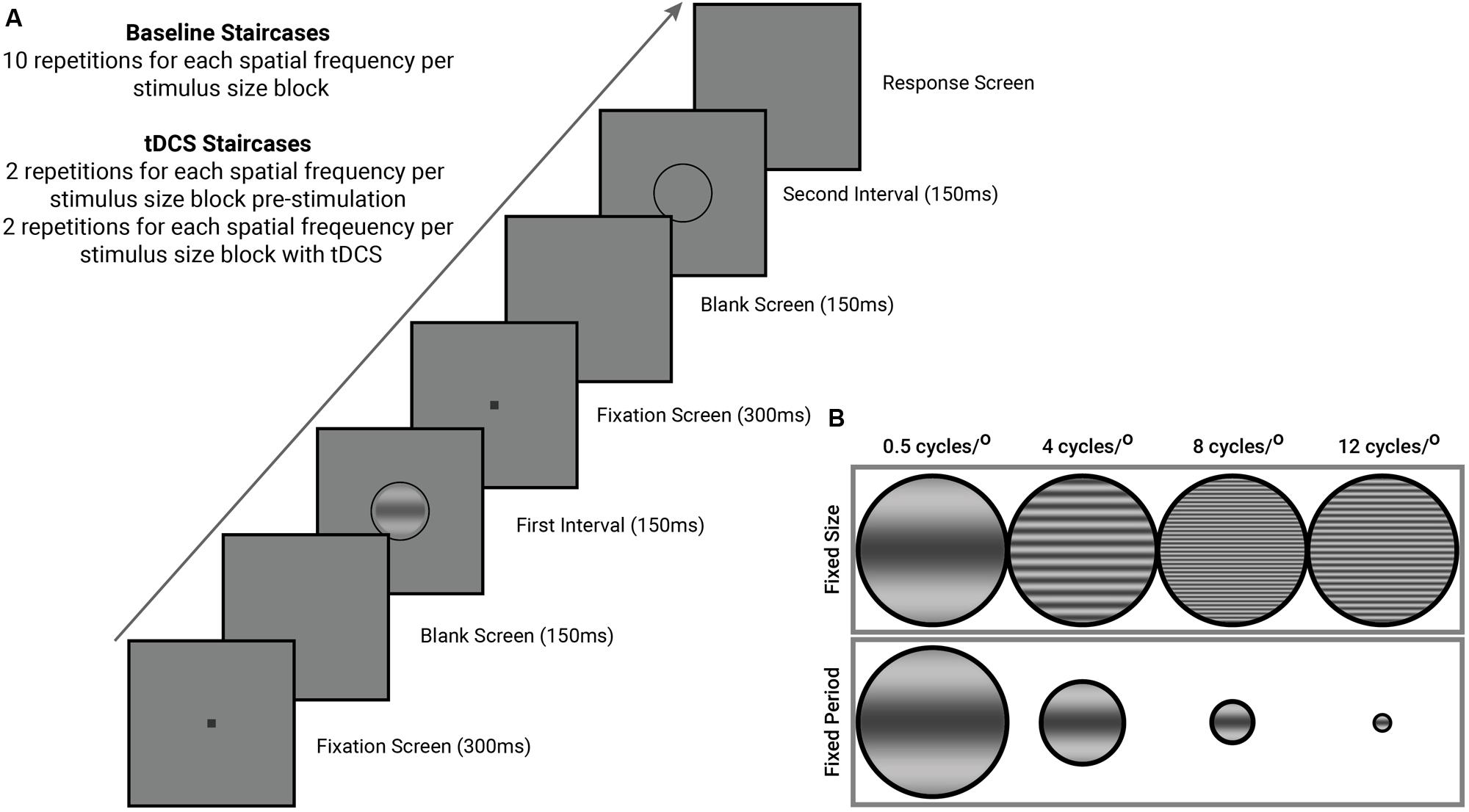
FIGURE 1. General psychophysical procedures completed by all observers in this study. (A) Stimulus presentation sequence (see text for details). (B) Contrast sensitivity was measured for both stimuli of a fixed size and fixed period, at four different spatial frequencies (0.5, 4, 8, and 12 cycles/°). Groups (n = 10 per group) were split according to stimulus orientation (45° oblique, and horizontal). Stimuli in the fixed period condition do not represent the actual change in size of our stimuli during the staircase, and are a graphical representation of the different stimulus dimensions used in this study. Stimuli of a fixed size subtended 3° of visual angle while stimuli of a fixed period had a period of 1.5 cycles.
Each spatial frequency by stimulus size block was repeated 10 times by observers in the baseline portion of the study (total of 80 staircase blocks), which approximately took 5 h to complete over multiple 1-h sessions completed on different days (approximately five sessions over 2 weeks). All staircase blocks were randomly interleaved for each observer, and only the final eight stimulus blocks were stored for data analysis. The contrast sensitivity of observers across each sequential measurement for all spatial frequency and stimulus size conditions is shown in Figure A1 (see Supplementary Material A), separated by orientation group. The 20 observers (10 per orientation group) that continued onto the tDCS portion of this study showed no statistically significant increment or decrement in contrast sensitivity across the final eight stimulus blocks completed during baseline (the slope of the line of best fit across all eight stimulus blocks was not statistically different from 0, all ps > 0.05). This is consistent with other studies that have shown either small (Sowden et al., 2002; Li et al., 2009), or no change in the CSF over sequential measurements in healthy adults (Dorais and Sagi, 1997; Adini et al., 2002, 2004; Maehara and Goryo, 2007).
tDCS Procedure
Transcranial Direct Current Stimulation is known to be a safe neuro-stimulation technique with no long lasting negative side effects, it is nevertheless important to limit the duration of stimulation to no more than 30–35 min (Nitsche et al., 2003b; Poreisz et al., 2007; Bikson et al., 2009; Russo et al., 2013; Fertonani et al., 2015). In order to meet this time restriction, the number of repetitions for each spatial frequency by stimulus size block was set to two. The total number of staircases completed by observers while receiving tDCS was 16 (four spatial frequencies by two stimulus size conditions by two repetitions). Prior to receiving either a-tDCS or c-tDCS, participants completed two staircases for each spatial frequency by stimulus size blocks, which were combined with the eight stimulus blocks from the baseline portion of this study and used as a pre-stimulation baseline (see Supplementary Material A, Figure A2). If contrast detection thresholds exceed their average baseline thresholds by at least 2 SDs, participants were asked to repeat the pre-stimulation baseline measurements. If thresholds following the repetition remained 2 SDs away from average thresholds, participants were excused from the study.
Immediately following baseline measurements, participants repeated the 16 staircases while receiving tDCS (time to complete: M = 21.05 min, SD = 2.74). All observers completed two stimulation sessions (anodal and cathodal, counterbalanced across participants) with no less than 48 hours between sessions. As both a-tDCS and c-tDCS have been shown to produce differential effects on contrast detection performance (see Antal et al., 2001; Kraft et al., 2010; Jacobson et al., 2012; Spiegel et al., 2012), we used both stimulation conditions to serve as a control of the other. Specifically, we prioritize any relative effects whereby tDCS polarity differentially modulated contrast sensitivity for a particular stimulus dimension within our observers. This allowed us to avoid certain confounds that have been associated with sham in neurostimulation designs (for review: Duecker and Sack, 2015). Specifically, while observers are typically unable to differentiate between a-tDCS and c-tDCS, they have been shown to easily detect the sham condition, which may alter their response pattern and thus, serves as a poor control for neurostimulation (Minhas et al., 2011; Kessler et al., 2012; O’Connell et al., 2012).
Injecting current was set to 2 mA, which yielded a stimulation current density of 0.042 mA/cm2 over primary visual cortex. The stimulation and reference electrode were positioned over Oz and Cz, respectively, in accordance with the 10–20 EEG system (Chatrian et al., 1985; Antal et al., 2004a). The current was initially ramped up, over a period of 30 s and participants waited for a minute once the current ramped-up so the experimenter could verify comfort levels. When participants completed the 16 staircases, the current was ramped back down to zero over a period of 30 s. Once the experimental session was completed, participants completed a post-stimulation checklist to verify for any minor side-effects (Nitsche et al., 2008) – none were reported.
Statistical Analyses
Contrast detection thresholds (cthreshold) were transformed to dB sensitivity units Contrast Sensitivity db = 20log 10(1/cthreshold) prior to analyses. The first statistical analysis conducted for all stimulus block conditions (stimulus orientation by stimulus period condition), was a 2 (tDCS polarity) × 4 (spatial frequency) repeated measures ANOVA on the difference contrast sensitivity values (stimulation – pre-stimulation), which tested for any spatial frequency dependent or polarity specific effect of tDCS on contrast sensitivity. All statistically significant interactions were followed by simple effect analyses. ANOVA output tables for all analyses are reported in Supplementary Material B.
Additionally, this study was designed to serve as a potential reference for future experiments that aim to use contrast sensitivity as a dependent measure of tDCS effects, but direct comparison between studies is complicated when only p-values are reported (see Kline, 2004 - Chapter 3 – for an in-depth description of the issues associated with null-hypothesis significance testing and p-values). Thus, we report an additional effect size analysis, which measured the magnitude of effects both at the group level (Hedge’s g) and at the case level (e.g., Left Tail Ratios, LTRs). The advantage of effect size measures is that their expected values are independent of sample size and thus they simplify the interpretation of results (particularly in regards to comparisons with other studies) and promote replication. The magnitude of an effect size should be interpreted in context to the relevant literature (Cohen, 1988). Thus, we interpret effect size magnitude according to the meta-analysis findings of Jacobson et al. (2012). They reported average effect sizes (g) of approximately 1.11 (CI [0.53 – 2.04]) of a-tDCS and 0.56 (CI [0.04 – 1.22]) of c-tDCS in cognitive studies (i.e., studies that measured the impact of tDCS on language, attention/perception, executive function, and memory). Any effect size that exceeds the average effect of either a-tDCS or c-tDCS is considered large, while effect sizes below the average values are moderate or small. LTRs are a case level analysis designed to assess the relative proportion of contrast sensitivity measurements recorded during stimulation to those of pre-stimulation in the left-tail of the combined distribution (see Supplementary Material B). Under assumptions of normality, homogeneity of variance, and large and equal group sizes, case-level proportions are functions of the magnitude of effect size at the group-level (Kline, 2004). However, when these assumptions are not met, group-level and case-level analyses will both offer separate information on the obtained effects. Given that the current that enters cortex with tDCS is several orders of magnitude less than what is required to elicit action potentials, any influence of tDCS on psychophysical performance will be relatively small, and may only be large enough in a sub-group of our sample (see Spiegel et al., 2013). Thus, the combination of group-level and case-level analyses offer a thorough descriptive approach of the data by quantifying effects in both central tendency and spread of the distribution of contrast sensitivity values. LTRs are calculated with the largest proportion as the numerator (regardless of time-point affiliation); values marked by an asterisk (∗) indicate that the pre-stimulation contrast sensitivity values were over-represented in the left tail of the combined distribution. Finally, interval estimates reported for Hedge’s g effect size measures are exact 95% confidence intervals calculate from the non-central t distribution (see Supplementary Material B; Cumming and Finch, 2001; Kline, 2004). Interval estimates for variance accounted for effect sizes are not reported, as their distribution in correlated designs are complex and do not follow a central nor a non-central distribution (Cumming and Finch, 2001; Kline, 2004).
Results
Two observers in the oblique condition completed the study at Concordia University, and thus, we first verified that their contrast sensitivity values were similar to those of the Colgate University sample (see Figure 2). We report U1 (see Supplementary Material B; Cohen, 1988), a statistic of overlap with range [0–1]: values of 0 indicate complete overlap between both samples, while values of 1 indicate no overlap whatsoever. At baseline, there was significant overlap between contrast sensitivity measures collected at both testing facilities (U1 never exceeded 0.27). Both a-tDCS and c-tDCS measures showed similar results to those of baseline, except for the fixed size stimuli with spatial frequency of 4 cycles/°, U1 = 0.87. This shows little overlap between scores from the Colgate and Concordia samples. However, given that contrast sensitivity values were discrepant for a single stimulus condition block, we average contrast sensitivity values collected at both testing locations for all subsequent analyses.
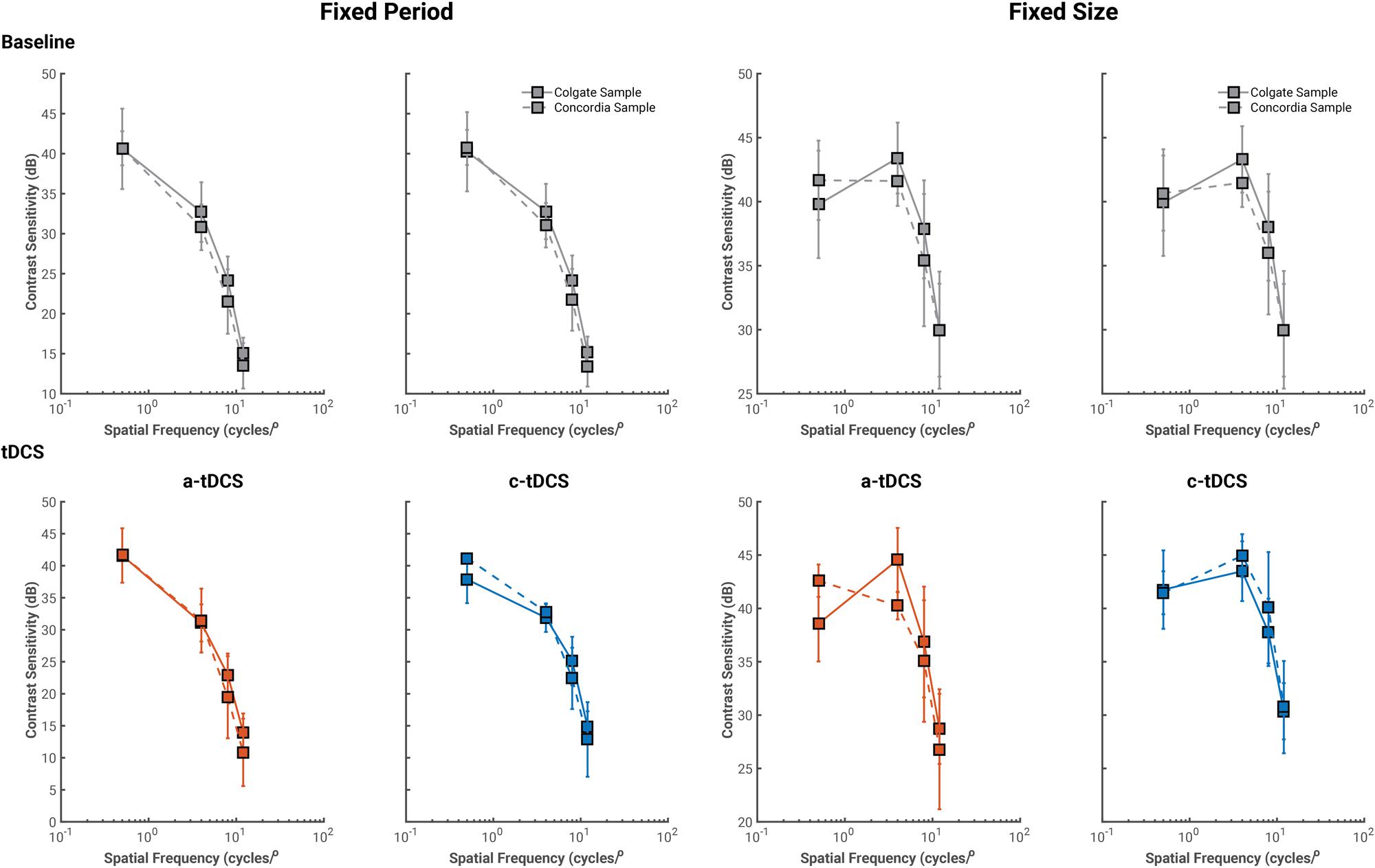
FIGURE 2. Average contrast sensitivity values collected from the Colgate University (solid lines) and Concordia University (dashed lines) at baseline (gray) and tDCS sessions. For all conditions, contrast sensitivity values from both samples overlapped significantly and thus, were averaged for all subsequent analyses.
Fixed Period Oblique and Horizontal Stimuli
The average effects of both a-tDCS and c-tDCS on fixed period oblique and horizontal gratings are shown in Figure 3. Contrast sensitivity measured with oblique fixed period gratings showed a statistically significant interaction between tDCS polarity and spatial frequency, F(3,27) = 8.10, p < 0.001, = 0.474, which stemmed from a contrast sensitivity decrease under a-tDCS and increase under c-tDCS at a spatial frequency of 8 cycles/°, F(1,9) = 20.79, p < 0.001, = 0.698. There was no statistically significant interaction between spatial frequency and tDCS type on contrast sensitivity measured with horizontal fixed period gratings, F(3,27) = 1.97, p = 0.585, = 0.179.
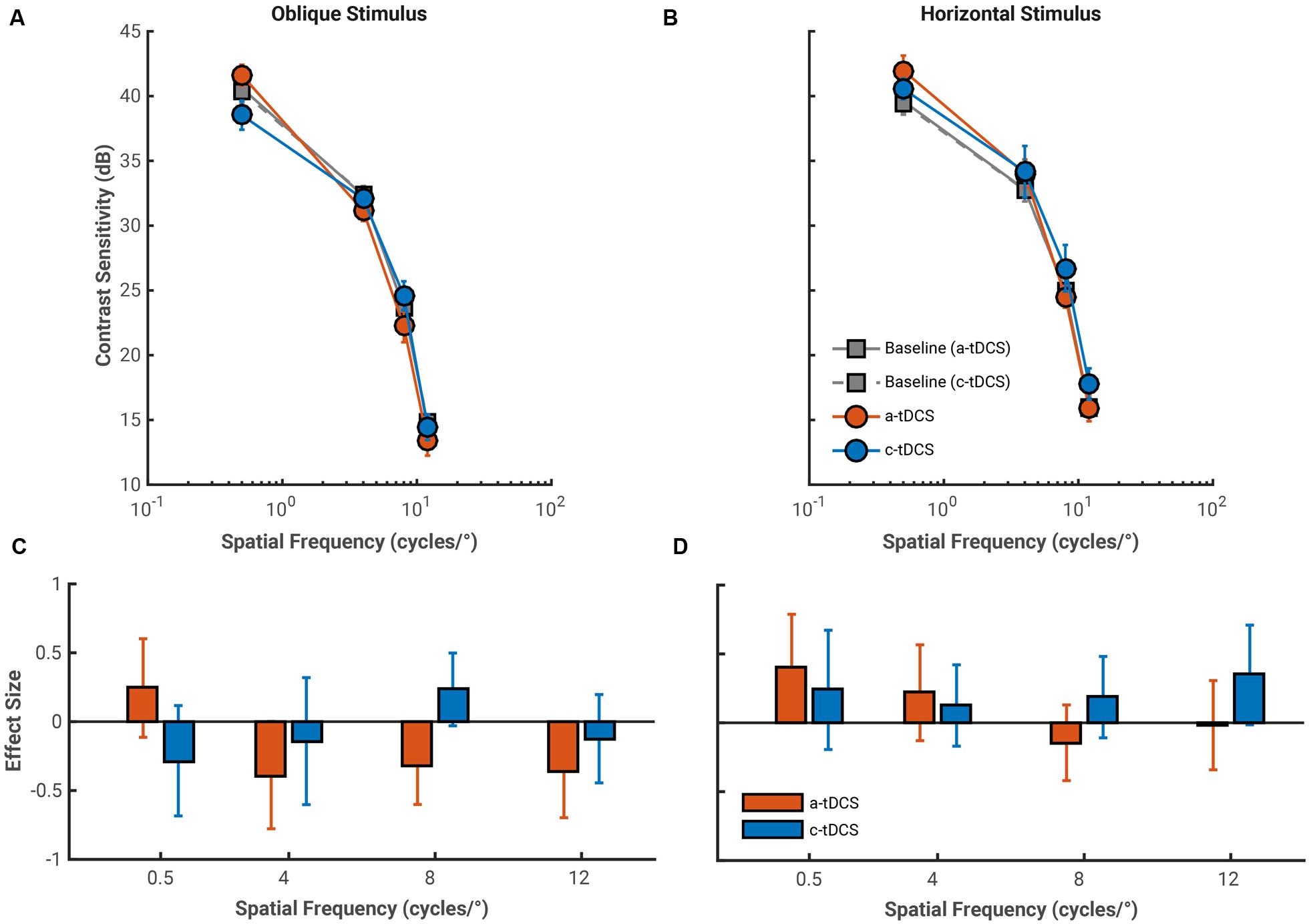
FIGURE 3. Average pre-stimulation (gray) and stimulation contrast sensitivity functions (CSFs) for both a-tDCS (red) and c-tDCS (blue) measured with the oblique (A) and horizontal (B) fixed period gratings (at spatial frequencies of 0.5, 4, 8, and 12 cycles/°). Contrast sensitivity is presented in decibels (dB). Error bars represent the standard error of the mean difference calculated across observers. (C,D) The effect sizes of the mean difference contrast sensitivity measured at stimulation and at pre-stimulation for oblique and horizontal conditions, respectively. For oblique gratings, contrast sensitivity measured at 8 cycles/° showed a polarity specific effect of tDCS, whereby a-tDCS decreased and c-tDCS increased contrast sensitivity. Error bars represent the exact 95% confidence interval of the effect size. We used error bar overlap to assess the magnitude of tDCS effects on contrast sensitivity. Thus, error bars that do not contain 0 and do not overlap with changes in contrast sensitivity with the other tDCS polarity are considered “significant”.
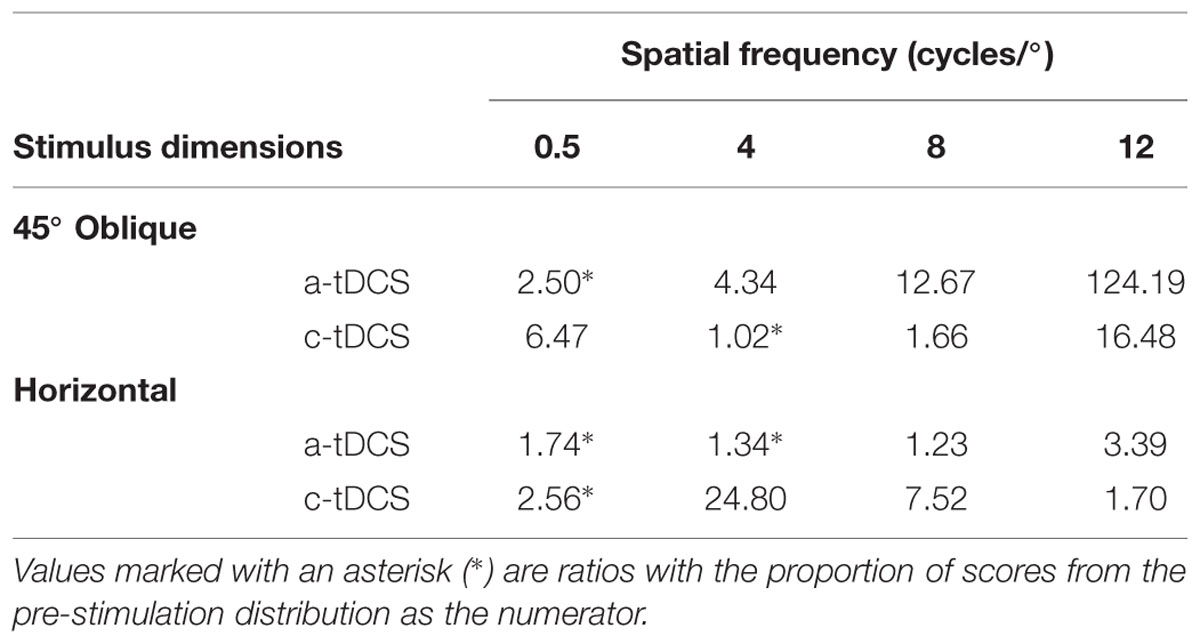
The effect size analysis also showed the polarity specific effect of tDCS on contrast sensitivity measured to an 8 cycles/° oblique grating (Figure 3C). Contrast sensitivity decreased by a third of a standard deviation under a-tDCS (8 cycles/°: g = –0.32, 95% CI [–0.60 –0.03]) while it increased by a quarter of a standard deviation under c-tDCS (g = 0.24, 95% CI [–0.03 0.50]). Additionally, we found a-tDCS to decrease contrast sensitivity by a similar amount at spatial frequencies of 4 cycles/° (g = –0.40, 95% CI [–0.78 –0.03]) and 12 cycles/° (g = –0.36, 95% CI [–0.70 –0.01]). At the group level, a-tDCS induced decreases in contrast sensitivity remained stable across spatial frequency, but at the case-level, we found that observers were progressively more likely to have contrast sensitivity values 1 SD below the grand mean than pre-stimulation contrast sensitivity values as spatial frequency increased. This would suggest that these decrements in contrast sensitivity under a-tDCS are accentuated with spatial frequency (see Table 1). Thus, the effects of a-tDCS may be spatial frequency dependent, and increase in magnitude in accordance with an increase in spatial frequency.
The effects of a-tDCS and c-tDCS on horizontal fixed period gratings were small in comparison to those of its oblique counterpart. We did find a moderate increment in contrast sensitivity under c-tDCS at a spatial frequency of 12 cycles/° (g = 0.35, 95% CI [–0.02 0.71]). This effect may be spatial frequency dependent, as the both the effect size and LTRs (see Table 1) showed that the benefit of c-tDCS on contrast sensitivity increased with spatial frequency: from 4 cycles/° (g = 0.13, 95% CI [–0.17 0.42]) and 8 cycles/° (g = 0.19, 95% CI [–0.11 0.48]), which reached significance at 12 cycles/°. Thus, the results of the fixed period condition show that the effects of a-tDCS may be most pronounced on oblique gratings while those of c-tDCS on horizontal gratings, both for spatial frequencies above the peak of the CSF.
Fixed Size Oblique and Horizontal Stimuli
The average effects of both a-tDCS and c-tDCS on oblique gratings of a fixed size are shown in Figure 4. There were no statistically significant interactions between spatial frequency and tDCS polarity for contrast sensitivity measure with either oblique, F(3,27) = 0.65, p = 0.585, = 0.068, or horizontal, F(3,27) = 2.83, p = 0.057, = 0.239, gratings. There was a main effect of tDCS polarity on contrast sensitivity measured to oblique gratings, F(1,9) = 9.23, p = 0.014, = 0.506. Anodal tDCS decreased and c-tDCS increased contrast sensitivity for all spatial frequencies. Effects of tDCS collapsed across spatial frequency are not particularly informative, and thus, we turn to our effect size analysis to measure if any changes in contrast sensitivity can attributed to tDCS.
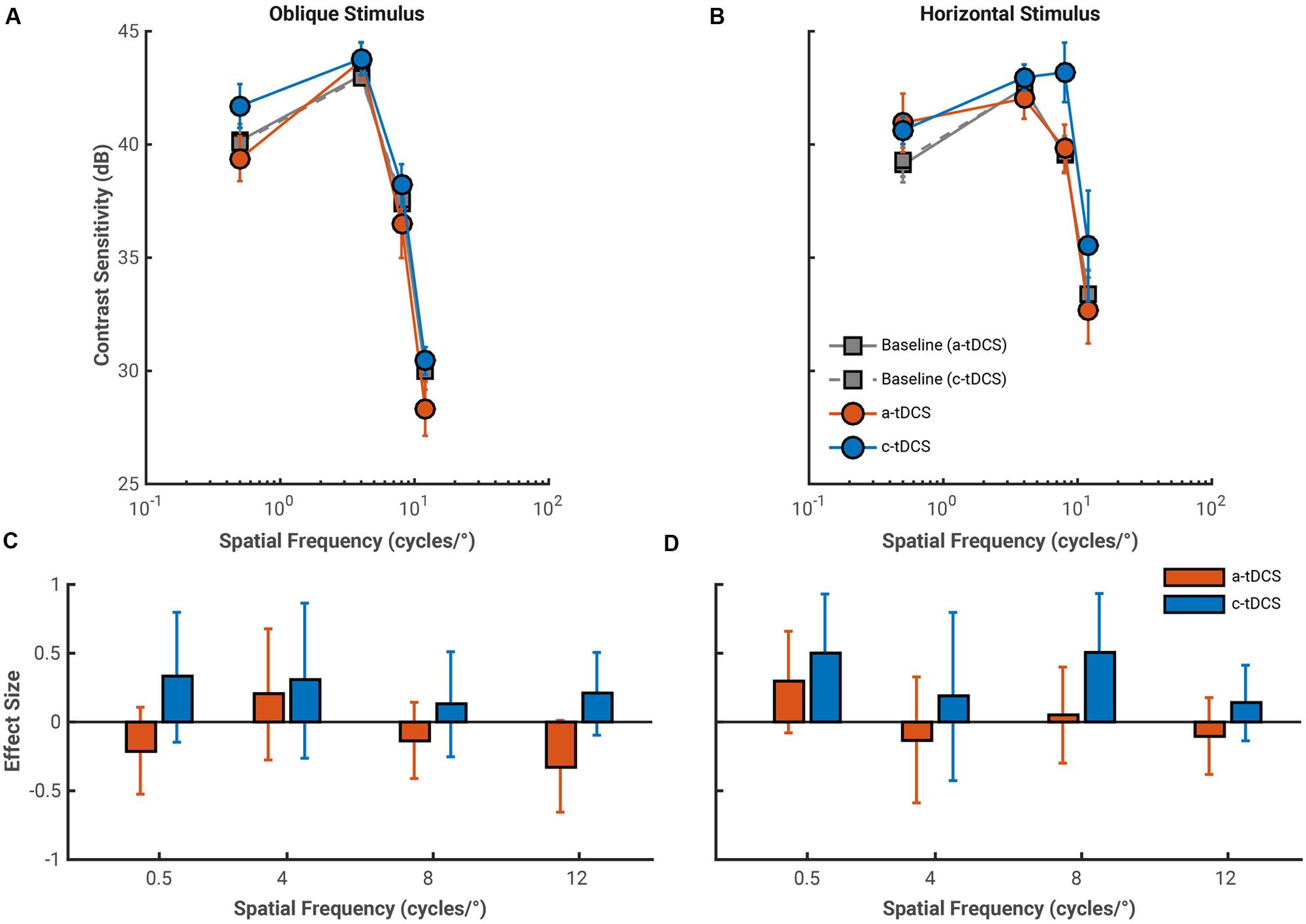
FIGURE 4. Average pre-stimulation (gray) and stimulation CSFs for both a-tDCS (red) and c-tDCS (blue) measured with the oblique (A) and horizontal (B) fixed size gratings (at spatial frequencies of 0.5, 4, 8, and 12 cycles/°). Contrast sensitivity is presented in decibels (dB). Error bars represent the standard error of the mean difference calculated across observers. (C,D) The effect sizes of the mean difference contrast sensitivity measured at stimulation and at pre-stimulation for oblique and horizontal conditions, respectively. We found a large increase in contrast sensitivity measured with the 8 cycles/° horizontal, fixed size grating under c-tDCS, and a potential polarity specific effect of tDCS on contrast sensitivity measured to oblique gratings at a spatial frequency of 12 cycles/°. Error bars represent the exact 95% confidence interval of the effect size. As in figure, we used error bar overlap to assess the magnitude of tDCS effects on contrast sensitivity.
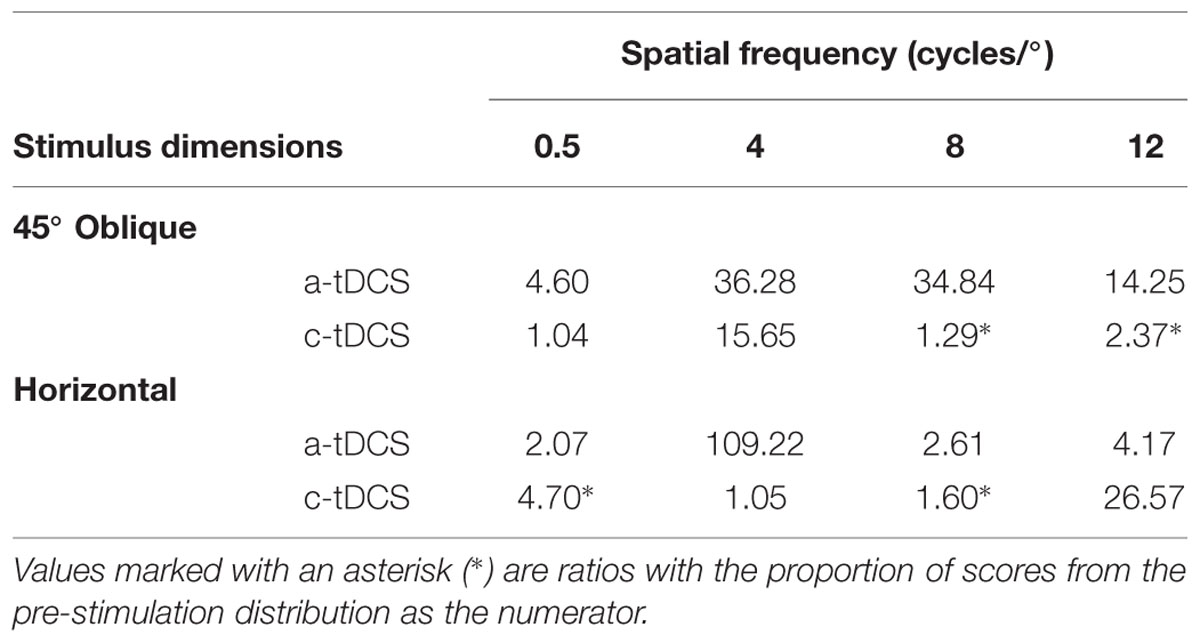
Overall, effect sizes in the fixed size condition were small and had large confidence intervals. There is an indication of a polarity specific effect of tDCS on contrast sensitivity measured to an oblique grating at 12 cycles/°. This effect has a similar direction to the polarity specific effect obtain in the fixed period condition: a-tDCS decreased contrast sensitivity (g = –0.33, 95% CI [–0.65 0.01]) while c-tDCS increased sensitivity (g = 0.21, 95% CI [–0.10 0.51]). The influence of a-tDCS here does not seem to increase with spatial frequency. LTRs were similar for both 4 and 8 cycles/° conditions, and decreased slightly at 12 cycles/°, which suggest a narrowing of the contrast sensitivity distribution of a-tDCS (see Table 2). We found no meaningful effects of a-tDCS on contrast sensitivity measured with horizontal gratings, but did find an abnormal increase in contrast sensitivity under c-tDCS to a horizontal grating of 8 cycles/° (g = 0.51, 95% CI [0.06 0.93]). While this may be indicative of an actual facilitation in contrast sensitivity, the effects of c-tDCS in this stimulus condition seem independent of spatial frequency. Additionally, the LTR value for this condition was small in comparison to the magnitude of the effect size, which should be considered when interpreting this result.
Orientation Dependent Effects of tDCS
Given that the effects of tDCS reported above varied according to the orientation of the stimulus, we opted compared the these effects directly by calculating effect size measures for the difference in contrast sensitivity between horizontal and oblique gratings for all stimulus and stimulation conditions (see Figure 5). Baseline contrast sensitivity, in both stimulus size conditions followed the well-defined “Oblique Effect” (Campbell et al., 1966; Appelle, 1972). Horizontal contrast sensitivity exceeded that of oblique at higher spatial frequencies in the fixed period (8 cycles/°: 12 cycles/°: g = 0.62, 95% CI [–0.29 1.51]) and fixed size conditions (8 cycles/°: g = 0.90, 95% CI [–0.04 1.81]; 12 cycles/°: g = 1.16, 95% CI [0.20 2.10]). However, the overlap between confidence intervals for baseline and tDCS suggest tDCS had no measurable impact on the magnitude of the Oblique Effect. Thus, while the effects of tDCS are orientation dependent (as shown above), they do not influence contrast sensitivity sufficiently to diminish or increase the magnitude of the Oblique Effect.
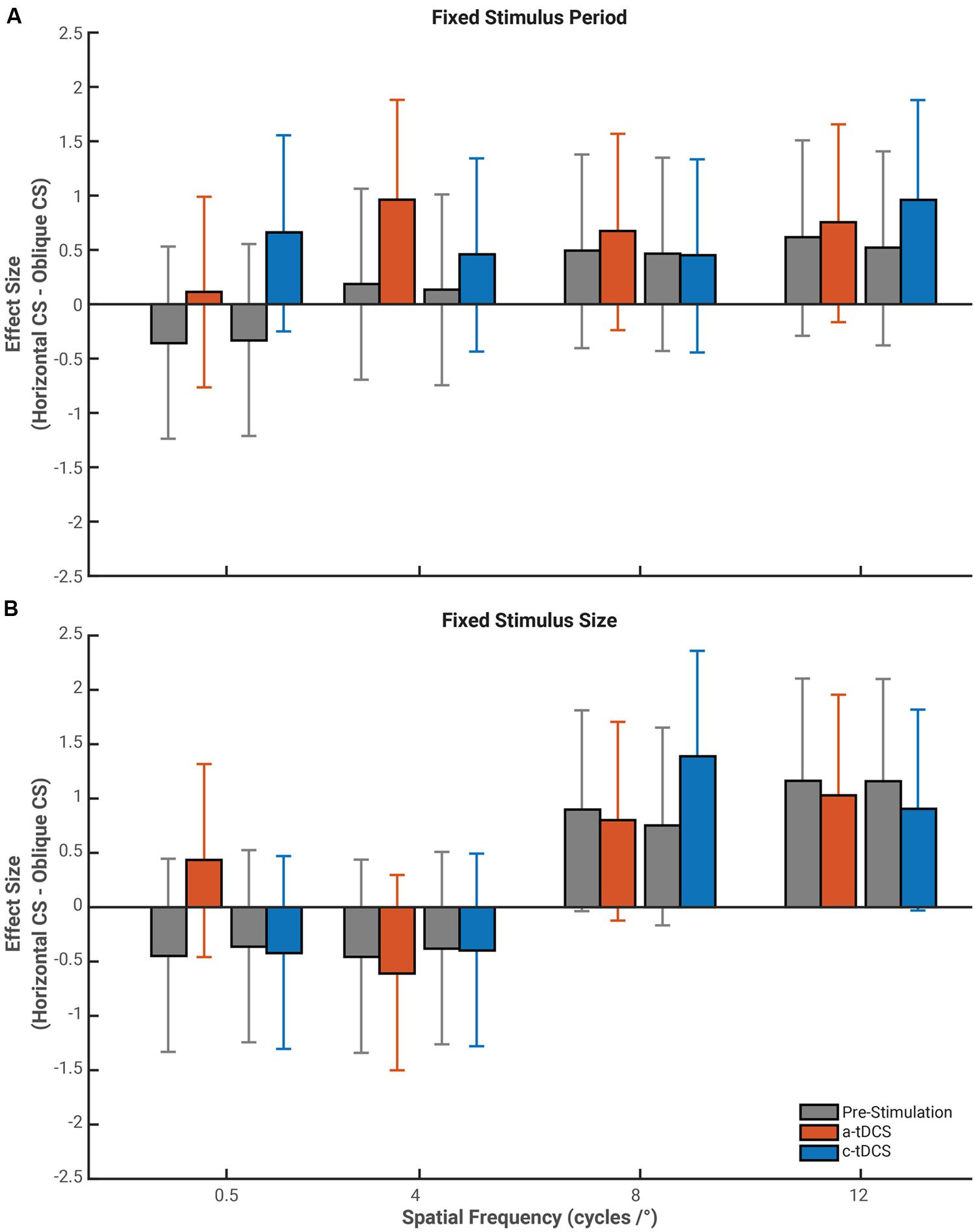
FIGURE 5. Effect size of the mean difference between contrast sensitivity measured with horizontally orientated gratings and oblique orientated gratings. Gray bars represent the respective pre-stimulation baseline for either a-tDCS (red) or c-tDCS (blue) contrast sensitivity difference between horizontal and oblique gratings for stimuli of a fixed period (A) and fixed size (B). We do find a-tDCS to increase the difference between contrast sensitivity measured to horizontal gratings and that of oblique gratings at a spatial frequency of 4 cycles/° and for c-tDCS to have a similar effect at a spatial frequency of 12 cycles/°. Error bars represent the exact 95% confidence interval for the mean difference effect size.
Effects of tDCS on Low Spatial Frequency Contrast Sensitivity
Finally, we note that while contrast sensitivity to a grating with a spatial frequency of 0.5 cycles/° can be affected by tDCS, these effects are unlikely to be indicative of a true modulation. The 0.5 cycles/° grating were identical in both the fixed period and fixed size condition, and attributing contrast sensitivity to either condition was arbitrary in our analysis. When contrast sensitivity values from both stimulus size conditions (fixed period and fixed size) were combined, and the effects of tDCS reanalyzed, we find that both a-tDCS (g = 0.46, 95% CI [0.05 0.85], LTR = 1.61∗) and c-tDCS (g = 0.44, 95% CI [0.02 0.85], LTR = 4.44∗) increased contrast sensitivity from baseline equally. As both a-tDCS and c-tDCS had an identical influence on contrast sensitivity values, neither can serve as a control for the other, which clouds any meaningful effects we may have obtained at lower spatial frequencies. We had not anticipated any modulation of contrast sensitivity under tDCS for our lowest spatial frequency grating as it differed from all others used in this study. At 0.5 cycles/°, a grating is part of the low spatial frequency rollover in the CSF, and is presumably subject to additional inhibition than the other gratings (Webster and Miyahara, 1997; Meese and Hess, 2004). If the application of tDCS over primary visual cortex creates an imbalance in the interactive properties of neurons (i.e., excitatory and inhibitory interactions), regardless of polarity, then contrast sensitivity to low spatial frequency gratings may be affected differently by the current generated with tDCS than to high spatial frequencies. Our findings here suggest that the application of a current, regardless of polarity, will increase contrast sensitivity to low spatial frequencies. Why this is, however, remains unclear.
Discussion
The goal of the current study was to assess whether the stimulus dimensions of gratings (spatial frequency, and orientation) could modulate the influence of tDCS on contrast sensitivity. We observe that the effects of both a-tDCS and c-tDCS were most pronounced on contrast sensitivity to obliquely oriented gratings of higher spatial frequency (i.e., above the peak of the CSF), and were absent at spatial frequencies below the peak the CSF. Generally, we found that a-tDCS decreased contrast sensitivity, while c-tDCS increased contrast sensitivity. However, these effects were small, and varied greatly across both stimulus spatial frequency, orientation and size conditions. In all but one stimulus condition, we found the influences of tDCS to be selective for polarity; only a-tDCS or c-tDCS had a large enough effect to influence contrast sensitivity. That said, when measured with an 8 cycles/° oblique grating (fixed period condition), contrast sensitivity was affected differently according to tDCS polarity: a-tDCS decreased while c-tDCS increased contrast sensitivity. Thus, while polarity specific effects of tDCS may be uncommon in vision studies (Antal et al., 2001; Accornero et al., 2007; Lang et al., 2007; Chaieb et al., 2008; Spiegel et al., 2012; Peters et al., 2013; Pirulli et al., 2014), we found that polarity specific influences of tDCS can be obtained under certain stimulus conditions (e.g., high frequency oblique gratings with small periods). Moreover, the effects of a-tDCS and c-tDCS on contrast sensitivity measured with fixed period gratings seem tied to orientation. Contrast sensitivity measured with oblique gratings was most subject to the influence of a-tDCS, while contrast sensitivity measured with horizontal gratings was most influenced by c-tDCS. While this did not affect the magnitude of the “Oblique Effect” (Campbell et al., 1966; Appelle, 1972; Essock, 1980), it may be indicative of an anisotropy of tDCS effects in vision, similar to the reported effects of Hansen et al. (2015).
The behavioral effects of tDCS result from an interaction between the electrical components of stimulation (Miranda et al., 2006; Paulus, 2011), the neuroanatomy of the stimulated area (Shipp, 2005; Radman et al., 2009; Bikson et al., 2013), the task completed by observers (Lapenta et al., 2013), and their cognitive state (Miniussi et al., 2010). While this allows for the broad acting effects of tDCS on cortex to be narrowed, or guided by the task, it also emphasizes that stimulus design should take into consideration the cortical area stimulated by tDCS. In primary visual cortex, the superficial layers near the apex of the calcarine sulcus contain neurons with higher preferred spatial frequencies than cells further from the apex (Tootell et al., 1981, 1988; De Valois et al., 1982; Foster et al., 1985; Engel et al., 1997; Horton, 2006; Henriksson et al., 2008; Yu et al., 2010). Additionally, the magnitude of the electric field generated by tDCS is greater at the cortical surface (Miranda et al., 2006; Nitsche et al., 2007; Bikson et al., 2013). Thus, it is plausible the effects of tDCS on contrast sensitivity were greatest when higher spatial frequency gratings were used as neurons with higher preferred spatial frequencies would be most influenced by tDCS. Likewise, the peak in current density at the apex of the primary visual cortex suggest the effects of tDCS may be restricted to the central visual field, which is retinotopically mapped to the apex of the calcarine sulcus (Tootell et al., 1988; Engel et al., 1997; Grill-Spector and Malach, 2004; Horton, 2006). There is a study that corroborates this hypothesis (Kraft et al., 2010), however, other factors may influence the localization of tDCS effects in the visual field, as a recent study by Costa et al. (2015) has failed to replicate the findings of Kraft et al. (2010). Nevertheless, if the effects of tDCS are greatest within the central 2° of the visual field, as proposed by Kraft et al., (2010), it may explain why contrast sensitivity to fixed size gratings, which extend beyond the area affected by tDCS, was only mildly altered by tDCS. Additional psychophysical mechanisms (e.g., summation effects; Graham et al., 1978; Legge, 1978; Peli et al., 1993; Meese and Summers, 2007) may have contributed to the lack of tDCS influence on contrast sensitivity to large gratings of high spatial frequency, as they also raise contrast sensitivity and potentially restricts any measurable influence of tDCS.
Changes in the stimulus characteristics presented to observers can have large contrasting tDCS effects on the same psychophysical measure. We opted to represent this with effect sizes to characterize changes in central tendency, and LTR, to define changes in the tail of the distribution (Feingold, 1995). While these may be considered uncommon statistical approaches, they are ideally suited to infer the meaningfulness of a change in behavior attributed to tDCS. For example, effects of tDCS in the tails of a distribution are to be expected as not all observers are affected equally by tDCS (Wagner et al., 2007; Datta et al., 2009; Spiegel et al., 2013). Thus, we used LTR to better define our dataset and characterized not only average effects (group-level) but also account for individual differences. Our analyses demonstrated that while the changes in contrast sensitivity induced by tDCS were sufficiently large to shift the central tendency of a distribution, certain effects were most apparent in the tails of the distribution. The decrease in contrast sensitivity under a-tDCS to fixed period gratings was of a similar magnitude for spatial frequencies of 4, 8, and 12 cycles/°, but the proportion of contrast sensitivity values in the left tail of the distribution increased with spatial frequency. This suggests observer contrast sensitivity, generally, was much more likely to show an influence of a-tDCS in higher spatial frequency conditions than when the spatial frequency neared the peak of the CSF. Furthermore, we calculated 95% confidence intervals of effect size measures to obtain an estimate of the sampling error in our effects. While most effect sizes were of moderate size, many had large confidence intervals that contained both positive and negative values. As 95% of all confidence intervals calculated in this way will contain the true effect size of a-tDCS and c-tDCS on contrast sensitivity measurements, both increments and decrements in contrast sensitivity appear equally valid directions in many conditions evaluated here. Hence, the expected directionality of tDCS polarity – a-tDCS excites while c-tDCS inhibits – which stems predominantly from findings in motor cortex (Nitsche et al., 2003a, 2007; Stagg et al., 2009; Jacobson et al., 2012; Pellicciari et al., 2013), should be disregarded for cortical areas that are functionally and structurally different (Shipp, 2005, 2007).
tDCS Polarity and Psychophysical Performance
We found facilitatory and inhibitory effects of tDCS on low-level visual function, but our findings contrast those of other, similar studies (Antal et al., 2001; Chaieb et al., 2008; Kraft et al., 2010; Peters et al., 2013; Spiegel et al., 2013). It well established that the a-tDCS excitatory, c-tDCS inhibitory effect is only truly valid when measured in motor cortex, while in visual cortex the behavioral outcome of tDCS cannot necessarily be predicted by its polarity (Antal et al., 2004a; Accornero et al., 2007; Miniussi et al., 2013; Pirulli et al., 2014; Hansen et al., 2015). There are many factors that contribute to the net influence of current on cell activity that may explain the different outcomes between stimulation in motor and primary visual cortex (e.g., neuroanatomy and functional anatomy; Radman et al., 2009; Peterchev et al., 2012; Bikson et al., 2013; Rahman et al., 2013). Still, if cells in primary cortex are similarly influenced by tDCS as those of motor cortex, an additional mechanism must be defined to account for the variability in behavioral outcomes of tDCS in vision studies. For tasks that involve the detection of a stimulus, facilitatory effects of c-tDCS may stem from an increase in signal-to-noise ratios that result from a decrease in cell excitability (Antal et al., 2004b; Miniussi et al., 2013; Pirulli et al., 2014). An increase in the signal-to-noise ratio could minimize stimulus uncertainty (Pelli, 1985), which will increase the detectability of the stimulus. Similarly, a-tDCS could worsen performance by injecting additional noise and decreasing the signal-to-noise ratio. That said, tDCS is a continuous neurostimulation procedure and its effects on neuronal behavior cannot be as simple as an increment in excitability under a-tDCS and decrement in excitability under c-tDCS (Miniussi et al., 2013; Pirulli et al., 2014). The continuous current generated by tDCS may instead alter the balance of excitation and inhibition in neurons affected by the current (Pirulli et al., 2014). Balance of excitation and inhibition is a known neuro-mechanism responsible for the tuning characteristics of visually responsive cells (it serves to narrow the bandwidth of tuning curves and regulates their responses to contrast; Rose and Blakemore, 1974; Blin et al., 1993; Ferster and Miller, 2000; Li et al., 2008; Edden et al., 2009; Katzner et al., 2011). Thus, the psychophysical performance change under tDCS obtained in vision studies, such as the one presented here, may lie in low-level gain mechanisms that adjust the responses of a cell to a given level of contrast.
Limitations
Our tDCS stimulation protocol used large electrodes (48 cm2 over Oz and 96 cm2 over Cz), which most likely covered both primary visual and secondary visual cortical areas. As these areas differ in their cortical folding (Rosa et al., 1997a,b; Horton, 2006), the alignment between the current generated by tDCS to the somatodendritic axis of the cell will vary and potentially alter the polarizing effects of tDCS (Rushton, 1927; Radman et al., 2009; Rahman et al., 2013). It is unclear how the stimulation of both primary and secondary visual cortex may have impacted our findings here, however, more focal approaches that use smaller electrodes (HD-tDCS; Miranda et al., 2013; Rahman et al., 2013), may help prevent the simultaneous stimulation of multiple visually responsive cortical sites in future studies.
Conclusion
The effects of tDCS on contrast sensitivity are largest when measured with high spatial frequency oblique oriented gratings of a fixed period (1.5 cycles). Additionally, we found that the magnitude of a-tDCS and c-tDCS effects may be anisotropic, as c-tDCS generally elicited larger effects with horizontal gratings, while a-tDCS with oblique gratings. Finally, the overall magnitude of tDCS effects on contrast sensitivity were small, and spatial frequency dependent effects vanished when contrast sensitivity was measured with larger gratings of variable period. The effects of tDCS on low-level visual function is evidently subject to the particular stimulus attributes presented to observers, and further demonstrates the susceptability of this stimulation technique to the activity of cells within the cortical area it stimulates. In regards to contrast sensitivity, we find that under certain stimulus condition, tDCS effects may be facilitatory or inhibitory within a particular group of observers, regardless of stimulation polarity. Consequently, careful use of stimuli that reliably elicit tDCS polarity specific effects should be favored when implementing tDCS in vision studies.
Funding
Portions of the current study were funded by a discovery grant from the Natural Sciences and Engineering Research Council (NSERC) to AJ, and by the Colgate Research Council Grant to BH. BR was supported by the Fonds de Recherche du Quebec – Nature et Technologie (FQRNT), and a bursary from the Ministère de l’Éducation, de l’Enseignement supérieur et de la Recherche (MEESR).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgment
The authors of this paper would like to acknowledge the contribution of Kristin Andres with data collection.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpsyg.2015.01784
Footnotes
- ^ The contrast sensitivity function is an index of sensitivity to contrast across multiple spatial frequencies, but previous studies that have used contrast sensitivity as a dependent measure of tDCS have restricted their stimulus to a single spatial frequency, orientation, and size. This can alter contrast sensitivity and thus alter tDCS effects. For example, CSFs measured with full-field gratings (well localized in Fourier space) generally have narrower bandpass shape and peak at a higher spatial frequency (∼4 cycles/°) than CSFs measured with gratings localized both in spatial frequency and space (i.e., Gabors), which peak at about 1 cycle/° (Peli et al., 1993). Similarly, CSFs measured with cardinally oriented gratings have higher contrast sensitivity values at spatial frequencies above the peak of the CSF than when measured with obliquely oriented gratings (Campbell et al., 1966).
- ^ We note that these results stem from a single study, which has yet to have been replicated.
References
Accornero, N., Li Voti, P., La Riccia, M., and Gregori, B. (2007). Visual evoked potentials modulation during direct current cortical polarization. Exp. Brain Res. 178, 261–266. doi: 10.1007/s00221-006-0733-y
Adini, Y., Sagi, D., and Tsodyks, M. (2002). Context-enabled learning in the human visual system. Nature 415, 790–793. doi: 10.1038/415790a
Adini, Y., Wilkonsky, A., Haspel, R., Tsodyks, M., and Sagi, D. (2004). Perceptual learning in contrast discrimination: the effect of contrast uncertainty. J. Vis. 4, 993–1005. doi: 10.1167/4.12.2
Antal, A., Kincses, T. Z., Nitsche, M. A., Bartfai, O., and Paulus, W. (2004a). Excitability changes induced in the human primary visual cortex by transcranial direct current stimulation: direct electrophysiological evidence. Invest. Ophthalmol. Vis. Sci. 45, 702–707. doi: 10.1167/iovs.03-0688
Antal, A., Nitsche, M. A., Kruse, W., Kincses, T. Z., Hoffmann, K.-P., and Paulus, W. (2004b). Direct current stimulation over V5 enhances visuomotor coordination by improving motion perception in humans. J. Cogn. Neurosci. 16, 521–527. doi: 10.1162/089892904323057263
Antal, A., Nitsche, M. A., and Paulus, W. (2001). External modulation of visual perception in humans. Neuroreport 12, 3553–3555. doi: 10.1097/00001756-200111160-00036
Antal, A., Nitsche, M. A., and Paulus, W. (2006). Transcranial direct current stimulation and the visual cortex. Brain Res. Bull. 68, 459–463. doi: 10.1016/j.brainresbull.2005.10.006
Appelle, S. (1972). Perception and discrimination as a function of stimulus orientation: the “oblique effect” in man and animals. Psychol. Bull. 78, 266–278. doi: 10.1037/h0033117
Bex, P. J., Mareschal, I., and Dakin, S. C. (2007). Contrast gain control in natural scenes. J. Vis. 7, 1–12. doi: 10.1167/7.11.12
Bikson, M., Datta, A., and Elwassif, M. (2009). Establishing safety limits for transcranial direct current stimulation. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 120, 1033–1034. doi: 10.1016/j.clinph.2009.03.018
Bikson, M., Name, A., and Rahman, A. (2013). Origins of specificity during tDCS: anatomical, activity-selective, and input-bias mechanisms. Front. Hum. Neurosci. 7:688. doi: 10.3389/fnhum.2013.00688
Blin, O., Mestre, D., Paut, O., Vercher, J. L., and Audebert, C. (1993). GABA-ergic control of visual perception in healthy volunteers: effects of midazolam, a benzodiazepine, on spatio-temporal contrast sensitivity. Br. J. Clin. Pharmacol. 36, 117–124. doi: 10.1111/j.1365-2125.1993.tb04206.x
Campbell, F. W., Kulikowski, J. J., and Levinson, J. (1966). The effect of orientation on the visual resolution of gratings. J. Physiol. 187, 427–436. doi: 10.1113/jphysiol.1966.sp008100
Chaieb, L., Antal, A., and Paulus, W. (2008). Gender-specific modulation of short-term neuroplasticity in the visual cortex induced by transcranial direct current stimulation. Vis. Neurosci. 25, 77–81. doi: 10.1017/S0952523808080097
Chatrian, G. E., Lettich, E., and Nelson, P. L. (1985). Ten Percent Electrode System for Topographic Studies of Spontaneous and Evoked EEG Activities. Am. J. EEG Technol. 25, 83–92. doi: 10.1080/00029238.1985.11080163
Cohen, J. (1988). Statistical Power Analysis for the Behavioral Sciences, 2nd Edn. New Jersey: Lawrence Erlbaum Associates.
Costa, T. L., Gualtieri, M., Barboni, M. T. S., Katayama, R. K., Boggio, P. S., and Ventura, D. F. (2015). Contrasting effects of transcranial direct current stimulation on central and peripheral visual fields. Exp. Brain Res 233, 1391–1397. doi: 10.1007/s00221-015-4213-0
Cumming, G., and Finch, S. (2001). A primer on the understanding, use, and calculation of confidence intervals that are based on central and noncentral distributions. Educ. Psychol. 61, 532–574. doi: 10.1177/0013164401614002
Datta, A., Bansal, V., Diaz, J., Patel, J., Reato, D., and Bikson, M. (2009). Gyri-precise head model of transcranial direct current stimulation: improved spatial focality using a ring electrode versus conventional rectangular pad. Brain Stimul 2, 201–207. doi: 10.1016/j.brs.2009.03.005
De Valois, R. L., Albrecht, D. G., and Thorell, L. G. (1982). Spatial frequency selectivity of cells in macaque visual cortex. Vision Res. 22, 545–559. doi: 10.1016/0042-6989(82)90113-4
Dorais, A., and Sagi, D. (1997). Contrast masking effects change with practice. Vision Res. 37, 1725–1733. doi: 10.1016/S0042-6989(96)00329-X
Duecker, F., and Sack, A. T. (2015). Rethinking the role of sham TMS. Front. Psychol. 6:210. doi: 10.3389/fpsyg.2015.00210
Edden, R. A. E., Muthukumaraswamy, S. D., Freeman, T. C., and Singh, K. D. (2009). Orientation discrimination performance is predicted by GABA concentration and gamma oscillation frequency in human primary visual cortex. J. Neurosci. 29, 15721–15726. doi: 10.1523/JNEUROSCI.4426-09.2009
Engel, S. A., Glover, G. H., and Wandell, B. A. (1997). Retinotopic organization in human visual cortex and the spatial precision of functional MRI. Cereb. Cortex 7, 181–192. doi: 10.1093/cercor/7.2.181
Essock, E. A. (1980). The oblique effect of stimulus identification considered with respect to two classes of oblique effects. Perception 9, 37–46. doi: 10.1068/p090037
Feingold, A. (1995). The additive effects of differences in central tendency and variability are important in comparisons between groups. Am. Psychol. 50, 5–13. doi: 10.1037/0003-066X.50.1.5
Ferster, D., and Miller, K. D. K. (2000). Neuron mechanisms of orientation selectivity in the visual cortex. Annu. Rev. Neurosci. 23, 441–471. doi: 10.1146/annurev.neuro.23.1.441
Fertonani, A., Ferrari, C., and Miniussi, C. (2015). What do you feel if I apply transcranial electric stimulation? Safety, sensations and secondary induced effects. Clin. Neurophysiol 126, 2181–2188. doi: 10.1016/j.clinph.2015.03.015
Foster, K. H., Gaska, J. P., Nagler, M., and Pollen, D. A. (1985). Spatial and temporal frequency selectivity of neurones in visual cortical areas V1 and V2 of the macaque monkey. J. Physiol. 365, 331–363. doi: 10.1113/jphysiol.1985.sp015776
Graham, N. V., Robson, J. G., and Nachmias, J. (1978). Grating summation in fovea and periphery. Vision Res. 18, 815–825. doi: 10.1016/0042-6989(78)90122-0
Grill-Spector, K., and Malach, R. (2004). The human visual cortex. Annu. Rev. Neurosci. 27, 649–677. doi: 10.1146/annurev.neuro.27.070203.144220
Hansen, B. C., Richard, B., Andres, K., Johnson, A. P., Thompson, B., and Essock, E. A. (2015). A cortical locus for anisotropic overlay suppression of stimuli presented at fixation. Vis. Neurosci. 32, E023. doi: 10.1017/S0952523815000255
Henriksson, L., Nurminen, L., Hyvärinen, A., and Vanni, S. (2008). Spatial frequency tuning in human retinotopic visual areas. J. Vis. 8, 1–13. doi: 10.1167/8.10.5
Horton, J. C. (2006). Ocular integration in the human visual cortex. Can. J. Ophthalmol. 41, 584–593. doi: 10.1016/S0008-4182(06)80027-X
Jacobson, L., Koslowsky, M., and Lavidor, M. (2012). TDCS polarity effects in motor and cognitive domains: a meta-analytical review. Exp. Brain Res. 216, 1–10. doi: 10.1007/s00221-011-2891-9
Katzner, S., Busse, L., and Carandini, M. (2011). GABAA inhibition controls response gain in visual cortex. J. Neurosci. 31, 5931–5941. doi: 10.1523/JNEUROSCI.5753-10.2011
Kessler, S. K., Turkeltaub, P. E., Benson, J. G., and Hamilton, R. H. (2012). Differences in the experience of active and sham transcranial direct current stimulation. Brain Stimul. 5, 155–162. doi: 10.1016/j.brs.2011.02.007
Kingdom, F. A. A., and Prins, N. (2010). PsychophysicsA Practical Introduction. 1st Edn. London: Elsevier.
Kline, R. B. (2004). Beyond Significance Testing: Reforming Data Analysis Methods in Behavioral Research. Washington, DC: American Psychological Association.
Kraft, A., Roehmel, J., Olma, M. C., Schmidt, S., Irlbacher, K., and Brandt, S. A. (2010). Transcranial direct current stimulation affects visual perception measured by threshold perimetry. Exp. Brain Res. 207, 283–290. doi: 10.1007/s00221-010-2453-6
Lang, N., Siebner, H. R., Chadaide, Z., Boros, K., Nitsche, M. A., Rothwell, J. C., et al. (2007). Bidirectional modulation of primary visual cortex excitability: a combined tDCS and rTMS study. Investig. Ophthalmol. Vis. Sci. 48, 5782–5787. doi: 10.1167/iovs.07-0706
Lapenta, O. M., Minati, L., Fregni, F., and Boggio, P. S. (2013). Je pense donc je fais: transcranial direct current stimulation modulates brain oscillations associated with motor imagery and movement observation. Front. Hum. Neurosci. 7:256. doi: 10.3389/fnhum.2013.00256
Legge, G. E. (1978). Space domain properties of a spatial frequency channel in human vision. Vision Res. 18, 959–969. doi: 10.1016/0042-6989(78)90024-X
Li, G., Yang, Y., Liang, Z., Xia, J., Yang, Y., and Zhou, Y. (2008). GABA-mediated inhibition correlates with orientation selectivity in primary visual cortex of cat. Neuroscience 155, 914–922. doi: 10.1016/j.neuroscience.2008.06.032
Li, R., Polat, U., Makous, W., and Bavelier, D. (2009). Enhancing the contrast sensitivity function through video game training. Nat. Neurosci. 12, 549–551. doi: 10.1038/nn.2296
Maehara, G., and Goryo, K. (2007). Perceptual learning in monocular pattern masking: experiments and explanations by the twin summation gain control model of contrast processing. Percept. Psychophys. 69, 1009–1021. doi: 10.3758/BF03193939
Meese, T. S., and Hess, R. F. (2004). Low spatial frequencies are suppressively masked across spatial scale, orientation, field position, and eye of origin. J. Vis. 4, 843–859. doi: 10.1167/4.10.2
Meese, T. S., and Summers, R. J. (2007). Area summation in human vision at and above detection threshold. Proc. Biol. Sci. 274, 2891–2900. doi: 10.1098/rspb.2008.3002
Minhas, P., Datta, A., and Bikson, M. (2011). Cutaneous perception during tDCS: role of electrode shape and sponge salinity. Clin. Neurophysiol. 122, 637–638. doi: 10.1016/j.clinph.2010.09.023
Miniussi, C., Harris, J. A., and Ruzzoli, M. (2013). Modelling non-invasive brain stimulation in cognitive neuroscience. Neurosci. Biobehav. Rev 37, 1702–1712. doi: 10.1016/j.neubiorev.2013.06.014
Miniussi, C., Ruzzoli, M., and Walsh, V. (2010). The mechanism of transcranial magnetic stimulation in cognition. Cortex 46, 128–130. doi: 10.1016/j.cortex.2009.03.004
Miranda, P. C., Lomarev, M., and Hallett, M. (2006). Modeling the current distribution during transcranial direct current stimulation. Clin. Neurophysiol. 117, 1623–1629. doi: 10.1016/j.clinph.2006.04.009
Miranda, P. C., Mekonnen, A., Salvador, R., and Ruffini, G. (2013). The electric field in the cortex during transcranial current stimulation. Neuroimage 70, 48–58. doi: 10.1016/j.neuroimage.2012.12.034
Nitsche, M. A., Cohen, L. G., Wassermann, E. M., Priori, A., Lang, N., Antal, A., et al. (2008). Transcranial direct current stimulation: state of the art 2008. Brain Stimul. 1, 206–223. doi: 10.1016/j.brs.2008.06.004
Nitsche, M. A., Doemkes, S., Karaköse, T., Antal, A., Liebetanz, D., Lang, N., et al. (2007). Shaping the effects of transcranial direct current stimulation of the human motor cortex. J. Neurophysiol. 97, 3109–3117. doi: 10.1152/jn.01312.2006
Nitsche, M. A., Fricke, K., Henschke, U., Schlitterlau, A., Liebetanz, D., Lang, N., et al. (2003a). Pharmacological modulation of cortical excitability shifts induced by transcranial direct current stimulation in humans. J. Physiol. 553, 293–301. doi: 10.1113/jphysiol.2003.049916
Nitsche, M. A., Liebetanz, D., Lang, N., Antal, A., Tergau, F., Paulus, W., et al. (2003b). Safety criteria for transcranial direct current stimulation (tDCS) in humans [1] (multiple letters). Clin. Neurophysiol. 114, 2220–2223. doi: 10.1016/S1388-2457(03)00235-9
O’Connell, N. E., Cossar, J., Marston, L., Wand, B. M., Bunce, D., Moseley, G. L., et al. (2012). Rethinking Clinical Trials of Transcranial Direct Current Stimulation: participant and Assessor Blinding Is Inadequate at Intensities of 2mA. PLoS ONE 7:e47514. doi: 10.1371/journal.pone.0047514
Paulus, W. (2011). Transcranial electrical stimulation (tES – tDCS; tRNS, tACS) methods. Neuropsychol. Rehabil. 21, 602–617. doi: 10.1080/09602011.2011.557292
Peli, E., Arend, L. E., Young, G. M., and Goldstein, R. B. (1993). Contrast sensitivity to patch stimuli: effects of spatial bandwidth and temporal presentation. Spat. Vis. 7, 1–14. doi: 10.1163/156856893X00018
Pelli, D. G. (1985). Uncertainty explains many aspects of visual contrast detection and discrimination. J. Opt. Soc. Am. A 2, 1508. doi: 10.1364/JOSAA.2.001508
Pellicciari, M. C. M., Brignani, D., and Miniussi, C. (2013). Excitability modulation of the motor system induced by transcranial direct current stimulation: a multimodal approach. Neuroimage 83, 569–580. doi: 10.1016/j.neuroimage.2013.06.076
Peterchev, A. V., Wagner, T. A., Miranda, P. C., Nitsche, M. A., Paulus, W., Lisanby, S. H., et al. (2012). Fundamentals of transcranial electric and magnetic stimulation dose: definition, selection, and reporting practices. Brain Stimul. 5, 435–453. doi: 10.1016/j.brs.2011.10.001
Peters, M. A. K., Thompson, B., Merabet, L. B., Wu, A. D., and Shams, L. (2013). Anodal tDCS to V1 blocks visual perceptual learning consolidation. Neuropsychologia 51, 1234–1239. doi: 10.1016/j.neuropsychologia.2013.03.013
Pirulli, C., Fertonani, A., and Miniussi, C. (2014). Is neural hyperpolarization by cathodal stimulation always detrimental at the behavioral level? Front. Behav. Neurosci. 8:226. doi: 10.3389/fnbeh.2014.00226
Poreisz, C., Boros, K., Antal, A., and Paulus, W. (2007). Safety aspects of transcranial direct current stimulation concerning healthy subjects and patients. Brain Res. Bull. 72, 208–214. doi: 10.1016/j.brainresbull.2007.01.004
Prins, N., and Kingdom, F. A. A. (2009). Palamedes: Matlab Routines for Analyzing Psychophysical Data. Available at: http://www.palamedestoolbox.org/
Radman, T., Ramos, R. L., Brumberg, J. C., and Bikson, M. (2009). Role of cortical cell type and morphology in sub-and suprathreshold uniform electric field stimulation. Brain Stimul. 2, 215–228. doi: 10.1016/j.brs.2009.03.007
Rahman, A., Reato, D., Arlotti, M., Gasca, F., Datta, A., Parra, L. C., et al. (2013). Cellular effects of acute direct current stimulation: somatic and synaptic terminal effects. J. Physiol. 591, 2563–2578. doi: 10.1113/jphysiol.2012.247171
Reato, D., Rahman, A., Bikson, M., and Parra, L. C. (2010). Low-intensity electrical stimulation affects network dynamics by modulating population rate and spike timing. J. Neurosci. 30, 15067–15079. doi: 10.1523/JNEUROSCI.2059-10.2010
Rosa, M. G. P., Casagrande, V. A., Preuss, T., and Kaas, J. H. (1997a). Visual field representation in striate and prestriate cortices of a prosimian primate (Galago garnetti). J. Neurophysiol. 77, 3193–3217.
Rosa, M. G. P., Fritsches, K. A., and Elston, G. N. (1997b). The second visual area in the marmoset monkey: visuotopic organisation, magnification factors, architectonical boundaries, and modularity. J. Comp. Neurol. 387, 547–567. doi: 10.1002/(SICI)1096-9861(19971103)387:4<547::AID-CNE6>3.0.CO;2-2
Rose, D., and Blakemore, C. (1974). Effects of bicuculline on functions of inhibition in visual cortex. Nature 249, 375–377. doi: 10.1038/249375a0
Rushton, W. A. H. (1927). THe effect upon the threshold for nervous excitation of the length of nerve exposed, and the angle between current and nerve. J. Physiol. 63, 357–377. doi: 10.1113/jphysiol.1927.sp002409
Russo, R., Wallace, D., Fitzgerald, P. B., and Cooper, N. R. (2013). Perception of comfort during active and sham transcranial direct current stimulation: a double blind study. Brain Stimul. 6, 946–951. doi: 10.1016/j.brs.2013.05.009
Shipp, S. (2005). The importance of being agranular: a comparative account of visual and motor cortex. Philos. Trans. R. Soc. B Biol. Sci. 360, 797–814. doi: 10.1098/rstb.2005.1630
Shipp, S. (2007). Structure and function of the cerebral cortex. Curr. Biol. 17, 443–449. doi: 10.1016/j.cub.2007.03.044
Sowden, P. T., Rose, D., and Davies, I. R. L. (2002). Perceptual learning of luminance contrast detection: specific for spatial frequency and retinal location but not orientation. Vision Res. 42, 1249–1258. doi: 10.1016/S0042-6989(02)00019-6
Spiegel, D. P., Byblow, W. D., Hess, R. F., and Thompson, B. (2013). Anodal transcranial direct current stimulation transiently improves contrast sensitivity and normalizes visual cortex activation in individuals with amblyopia. Neurorehabil. Neural Repair 27, 760–769. doi: 10.1177/1545968313491006
Spiegel, D. P., Hansen, B. C., Byblow, W. D., and Thompson, B. (2012). Anodal transcranial direct current stimulation reduces psychophysically measured surround suppression in the human visual cortex. PLoS ONE 7:e36220. doi: 10.1371/journal.pone.0036220
Stagg, C. J., Best, J. G., Stephenson, M. C., O’Shea, J., Wylezinska, M., Kincses, Z. T., et al. (2009). Polarity-Sensitive Modulation of Cortical Neurotransmitters by Transcranial Stimulation. J. Neurosci. 29, 5202–5206. doi: 10.1523/JNEUROSCI.4432-08.2009
Stagg, C. J., and Nitsche, M. A. (2011). Physiological Basis of Transcranial Direct Current Stimulation. Neuroscientist 17, 37–53. doi: 10.1177/1073858410386614
Tootell, R. B., Silverman, M. S., and De Valois, R. L. (1981). Spatial frequency columns in primary visual cortex. Science 214, 813–815. doi: 10.1126/science.7292014
Tootell, R. B., Switkes, E., Silverman, M. S., and Hamilton, S. L. (1988). Functional anatomy of macaque striate cortex. II. Retinotopic organization. J. Neurosci. 8, 1531–1568.
Tyler, C. W. (1997). Colour bit-stealing to enhance the luminance resolution of digital displays on a single pixel basis. Spat. Vis. 10, 369–377. doi: 10.1163/156856897X00294
Wagner, T., Fregni, F., Fecteau, S., Grodzinsky, A., Zahn, M., and Pascual-Leone, A. (2007). Transcranial direct current stimulation: a computer-based human model study. Neuroimage 35, 1113–1124. doi: 10.1016/j.neuroimage.2007.01.027
Ward, R., and Weiskrantz, L. (1969). Impaired Discrimination Following Polarisation of the Striate Cortex. Exp. Brain Res. 356, 346–356. doi: 10.1007/BF00235243
Webster, M. A., and Miyahara, E. (1997). Contrast adaptation and the spatial structure of natural images. J. Opt. Soc. Am. A 14, 2355–2366. doi: 10.1364/JOSAA.14.002355
Yu, H.-H. H., Verma, R., Yang, Y., Tibballs, H. A., Lui, L. L., Reser, D. H., et al. (2010). Spatial and temporal frequency tuning in striate cortex: functional uniformity and specializations related to receptive field eccentricity. Eur. J. Neurosci. 31, 1043–1062. doi: 10.1111/j.1460-9568.2010.07118.x
Keywords: contrast sensitivity, transcranial Direct Current Stimulation (tDCS), spatial frequency, orientation, spatial vision
Citation: Richard B, Johnson AP, Thompson B and Hansen BC (2015) The Effects of tDCS Across the Spatial Frequencies and Orientations that Comprise the Contrast Sensitivity Function. Front. Psychol. 6:1784. doi: 10.3389/fpsyg.2015.01784
Received: 08 July 2015; Accepted: 05 November 2015;
Published: 27 November 2015.
Edited by:
Frédéric Gosselin, University of Montreal, CanadaReviewed by:
Andrew M. Haun, Harvard Medical School, USACarlo Miniussi, University of Brescia and IRCCS Centro San Giovanni di Dio Fatebenefratelli, Italy (Anna Fertonani contributed to the review of Carlo Miniussi)
Copyright © 2015 Richard, Johnson, Thompson and Hansen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bruno Richard, YnJ1bm8ucmljaGFyZEB5b3JrLmFjLnVr
 Bruno Richard
Bruno Richard Aaron P. Johnson
Aaron P. Johnson Benjamin Thompson
Benjamin Thompson Bruce C. Hansen
Bruce C. Hansen