- Department of Philosophy, Sociology, Pedagogy and Applied Psychology, University of Padova, Padova, Italy
Depression is widely seen as the result of difficulties in regulating emotions. Based on neuroimaging studies on voluntary emotion regulation, neurobiological models have focused on the concept of cognitive control, considering emotion regulation as a shift toward involving controlled processes associated with activation of the prefrontal and parietal executive areas, instead of responding automatically to emotional stimuli. According to such models, the weaker executive area activation observed in depressed patients is attributable to a lack of cognitive control over negative emotions. Going beyond the concept of cognitive control, psychodynamic models describe the development of individuals’ capacity to regulate their emotional states in mother-infant interactions during childhood, through the construction of the representation of the self, others, and relationships. In this mini-review, we link these psychodynamic models with recent findings regarding the abnormal functioning of the default system in depression. Consistently with psychodynamic models, psychological functions associated with the default system include self-related processing, semantic processes, and implicit forms of emotion regulation. The abnormal activation of the default system observed in depression may explain the dysfunctional aspects of emotion regulation typical of the condition, such as an exaggerated negative self-focus and rumination on self-esteem issues. We also discuss the clinical implications of these findings with reference to the therapeutic relationship as a key tool for revisiting impaired or distorted representations of the self and relational objects.
Introduction
Depression is generally considered as the outcome of difficulties in regulating emotions (for reviews see Campbell-Sills and Barlow, 2007; Aldao et al., 2010). When dealing with their own emotions, depressed individuals tend to ruminate on (Nolen-Hoeksema, 2000; Garnefski and Kraaij, 2006), avoid or suppress thoughts and emotions associated with negative events (Wenzlaff and Rude, 2002; Campbell-Sills et al., 2006), whereas a reappraisal of the event from a different perspective (Gross, 2002; Webb et al., 2012) or a non-judgmental acceptance (Liverant et al., 2008; Kohl et al., 2012) would be more effective for the purpose of containing negative emotional activation and its physiological correlates. Given its importance for psychological wellbeing, emotion regulation is attributed a key role in psychological treatments for depression (Messina et al., 2013; Grecucci et al., 2015).
Within the emerging field of affective neuroscience, the concept of voluntary emotion regulation has been widely used to explain the findings of functional neuroimaging studies conducted to elucidate the neural correlates of affective dysfunctions (for reviews, see Taylor and Liberzon, 2007; Menon, 2011). These studies have amply documented that individuals suffering from depression have a decreased activation of prefrontal cortex areas involved in executive control (including the dorsolateral prefrontal cortex – dlPFC –, and the dorsal anterior cingulate cortex – dACC), suggesting a weaker top–down control over their emotional reactivity deriving from the activation of limbic structures such as the amygdala (Drevets, 2001; Siegle et al., 2007). This interpretation is in line with fMRI studies concerning the neural correlates of emotion regulation that have revealed an increased activation of the prefrontal areas and a decreased activation of the amygdala in tasks involving the cognitive control of emotions by comparison with the spontaneous response to emotional stimuli (Ochsner and Gross, 2005; Buhle et al., 2014). Even if other authors have found that depressed individuals can display impaired emotion regulation despite preserved recruitment of dlPFC (Greening et al., 2014) and increased dlPFC recruitment during emotion regulation attempts (Johnstone et al., 2007), the emotion dysregulation seen in depression is consistently interpreted as a lack of cognitive control over emotional states (see Figure 1, the part in red, for a graphic representation of the executive areas involved in voluntary emotion regulation).
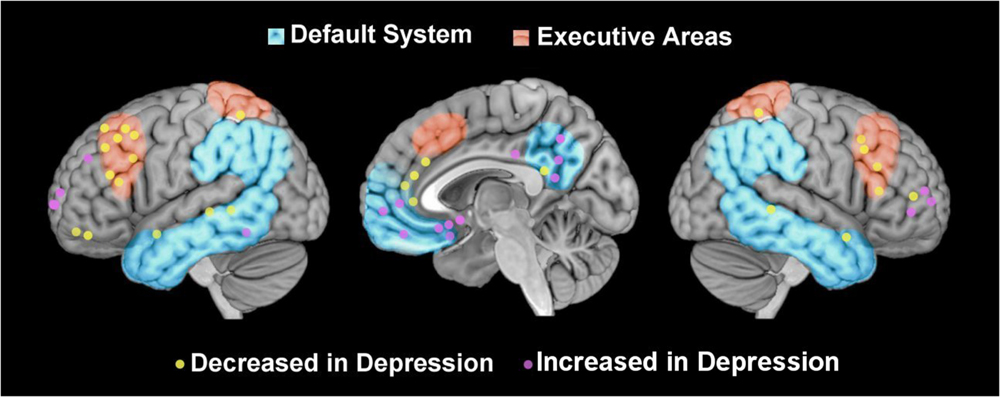
FIGURE 1. Graphical representation of foci of perfusion reported in association with depression. Graphical representation of foci of brain activity reported in studies that have compared depressed patients and healthy controls (in yellow decreased activations, in violet increased activations), located in executive areas [in red, retrieved from http://neurosynth.org/; (Yarkoni et al., 2011)], and default system (Raichle et al., 2001b).
In addition to investigating brain activity in response to stimuli or during cognitive tasks, neuroscientists have become increasingly interested in the brain’s intrinsic activity in resting state. Studying resting state activity has led to the identification of the “default system,” a set of regions – including the ventral medial prefrontal cortex (vmPFC), the posterior cingulate cortex (pCC), the posterior parietal lobe, and the lateral, inferior and medial temporal cortices (see Figure 1, in blue) – that are usually activated at rest and deactivated during cognitively effortful tasks (Raichle et al., 2001a; Raichle and Snyder, 2007). Research on resting state activity has also been applied to investigating emotional disorders, comparing the resting-state brain signals of patients and healthy subjects (Broyd et al., 2009; Whitfield-Gabrieli and Ford, 2012). This field of research is generating new lines of inquiry for neurobiological models of emotion regulation and their application to interpreting the brain correlates of emotional disorders. In the present mini-review, we address this issue by examining the findings on the abnormal functioning of resting-state brain activity in depressed patients. We specifically consider these findings in the light of clinical concept, coming from psychodynamic tradition, which underscore the role of internal representations of the self and others in emotion dysregulation.
Psychological Mechanisms: Internal Representations and Emotion Regulation
While the neurobiological models have conceptualized emotion regulation as a form of cognitive control, the psychodynamic tradition has concentrated more on investigating how individuals develop the ability to regulate their emotions in the course of their childhood, collecting evidence of the importance to emotion regulation of constructing a representation of the self and of relationships with others in their primary relationships. This interest is apparent in the works of Ferenczi (1933), Spitz (1945, 1965), Freud (1955), Bion (1959, 1967), Winnicott (1965), and Freud and Burlingham (1974), among others. These authors take the view that emotion regulation in infants depends on their caregivers’ contingent responses (on “good enough” parenting; Winnicott, 1965). For instance, Bion (1967) pointed out the caregivers’ important role in the infants’ acquisition of a comprehension and containment of their inner world through the transformation of the infant’s projected psychological experience in a “metabolized” form. Children can thus internalize the mother’s function and, with time, they learn to regulate their negative affective states.
Internalizing the mother’s function coincides with a self-building process described by Kernberg (1976) as the product of introjection and identification of the child’s significant relationship with the mother. Failure to achieve an interpersonal regulation in the mother-infant interaction may interfere with the construction of the self, and of its regulatory functions. Winnicott (1967) and Kohut (1971) used the term “weakened self” to describe the effects of a caregiver’s incongruous mirroring that oblige children to internalize distorted parental representations, which take the place of their subjective experience, preventing them from regulating that subjective experience. This is also implicit in Kernberg’s (1976) model, according to which a lack of differentiation between the internal representations of the self and others is an important factor contributing to emotional instability. With specific regard to depression, a mother’s inadequate mirroring function induces the child to internalize a rigid or even sadistic super-ego that implies a poor self-representation in which the subject feels helpless (Kohut, 1971, 1977; Kohut and Wolf, 1978).
The assumption that an individual’s internal representation of the self and others is important to emotion regulation has been the object of empirical investigation in the field of attachment theory (Bowlby, 1969, 1973, 1980). The attachment patterns characterizing the relationship between children and their caregivers can be seen as a particular form of dyadic regulation, in which the infant experiences the caregiver’s emotional availability (Cassidy, 1994). Within the primary relationship, adaptive emotion regulation strategies are learned by building “internal working models” (IWM; Bowlby, 1969), which are mental representations of the self and others based on the child’s daily experiences and expectations regarding a given caregiver’s response to attachment behavior. When the primary attachment figure is available and responsive to their needs, infants develop a sense of attachment security characterized by IWM that include positive beliefs regarding the self and others, the accessibility of others and their ability to alleviate distress, thus shaping affect regulation in the event of distress (Feeney and Cassidy, 2003; Fraley et al., 2006). But if infants have caregivers who are inconsistent and fail to respond adequately to their attachment needs, they develop a sense of attachment insecurity characterized by IWM that include negative representations of the self, low self-esteem and parenting self-esteem (Calvo and Bianco, 2015), difficulty in relying on others when their own emotional resources are insufficient, low dyadic adjustment, and loneliness (Shaver and Hazan, 1987; Kobak and Sceery, 1988; Sroufe, 2005). In recent years, insecure attachment has been quantitatively operationalized in terms of self-reported anxiety and avoidance in adult attachment relationships (Brennan et al., 1998), which imply two different maladaptive affect regulation strategies, called “secondary strategies” (Mikulincer et al., 2003; Shaver and Mikulincer, 2007): (i) strategies based on over-activation of the attachment system, including relational overdependence, desire to minimize physical, cognitive, and emotional distance from others; (ii) strategies based on deactivation of the attachment system, featuring the creation of emotional distance from others, avoidance of intimacy, and the suppression of negative feelings and memories. In the attachment literature, there is large evidence of depressive symptoms being associated with the chronic use of secondary emotion regulation strategies (Cole-Detke and Kobak, 1996; Rosenstein and Horowitz, 1996; Mickelson et al., 1997; Malik et al., 2015).
Recent contributions to this line of research have shown that the mirroring function of their attachment figures enables children to internalize the ability to think for themselves and perceive themselves as a thinking entity, a process called “mentalization” (Fonagy et al., 2007, 2011). In conditions of attachment security, children’s affective states are accurately (but not overwhelmingly) reflected back to them, whereas attachment insecurity and a lack of mirroring interfere with this mentalization process, giving rise to emotional disorders (Fonagy and Target, 1997; Bateman and Fonagy, 2004).
Neural Mechanisms: The Default System in Emotion Regulation
While psychodynamic models of emotion regulation emphasize the importance of the internal representation of the self and others in explaining emotional disorders, neuroscience has concentrated more on emotion regulation as a form of cognitive control, neglecting the importance of the semantic representations on which controlling processes may act. Considering the default system in association with emotion dysregulation can compensate for this shortcoming.
Default System Functions Linked to Emotion Regulation
The main feature of the default system is that it is activated during rest (Raichle et al., 2001a; Raichle and Snyder, 2007). It has been amply documented that, in the absence of a task that demands voluntary attention, the mind usually tends to wander (Mason et al., 2007). Activation of the default system is generally anti-correlated with activation of the executive areas (Fox et al., 2005), a situation reflecting the opposition between internal (default system) and external (executive functions) addressed attention.
In task-related studies, activation of the default system has been associated with cognitive tasks that include aspects of self-representation (Northoff et al., 2006) and self-projection, described as the capacity to project oneself mentally from the present into the past or future, but also from other people’s perspectives (Buckner and Carroll, 2007; Spreng et al., 2009). Self-projection underlies several processes that may be associated with emotional dysregulation, such as access to autobiographical memories (self-projection into the past) or future plans (self-projection into the future), but also with empathy and theory of mind (self-projection from others’ perspectives). In line with psychodynamic theory and research showing the importance of self-representation for emotion regulation, the evidence of the default system being activated in self-related processes suggests its involvement in emotion regulation. Intriguingly, the overlap between brain structures that are activated for self-representation and in theory of mind (Saxe et al., 2006) seems to confirm the psychodynamic view of a common source for the representations of the self and others constructed in infants’ primary relationships with their caregivers.
Another point in common with the psychodynamic models is the similarity between the default system and the semantic system, a set of regions activated by the retrieval and manipulation of conceptual knowledge gained from capturing regularities in the outside world (Binder et al., 2009). Semantic memory may include more complex representations of clinical relevance, however, such as those of relationships governing social interactions (Zahn et al., 2007), and representations of the self (Lou et al., 2004). In this setting, IWM can be seen as an example of representations that capture regularities in interpersonal relationships. Indeed, semantic areas are modulated by the exposition to attachment-related experimental stimuli, such as familiar faces (Gobbini et al., 2004) or of attachment eliciting pictures (Buchheim et al., 2006).
The activation of semantic areas is commonly described in voluntary emotion regulation studies, such as reappraisal (Buhle et al., 2014; Messina et al., 2015), but also in association with implicit forms of regulation in which executive functions are not involved, such as spontaneous avoidance (Benelli et al., 2012). Psychotherapeutic action has also been found associated with changes in semantic area activation (Messina et al., 2013). Going beyond the concept of emotion regulation as a form of cognitive control, recent recent contributions have begun to take into consideration the importance of semantic representation on which controlling processes may act (Blair and Mitchell, 2009; Buhle et al., 2014; Greening et al., 2014). According to recent models, semantic areas may play a key role in emotion regulation by conveying information about motivational priorities and facilitating processing of behaviorally relevant inputs (see Viviani, 2013, 2014; Messina et al., 2015).
Abnormal Default System Activity in Depression
Due to the default system’s involvement in the self-related processing and semantic representation of repeated past experiences, an abnormal default system activity should be expected in depressed patients. Several studies have tried to clarify the specificity of resting-state brain functioning in depression, using positron emission tomography (PET) or the perfusion MRI technique known as arterial spin labeling (ASL). The methodological features of these studies are listed in Table 1.
Both PET and ASL studies have produced evidence of less activity in the frontal executive areas of the brains of depressed patients compared with healthy controls, especially in the dlPFC (Mayberg et al., 1997; Kennedy et al., 2001; Lui et al., 2009; Monkul et al., 2012), but also in the dACC (Mayberg et al., 1997; Monkul et al., 2012) (see Figure 1). A greater activation has also often been reported in subcortical areas, including the amygdala, in depressed patients (Drevets, 2001; Lui et al., 2009; Duhameau et al., 2010). These results are consistent with the neurobiological model of emotion regulation that postulates a weaker cognitive control of emotions in depression.
In addition to the prefrontal-subcortical network of voluntary emotion regulation, studies on resting-state brain activity have reported foci of increased activation located in the anterior part of the default system, extending from the subgenual ACC to the anterior portion of the ACC or the vmPFC (Drevets, 2001; Kennedy et al., 2001; Duhameau et al., 2010), and also to posterior portions of the default system such as the PCC and precuneus (Lui et al., 2009; Monkul et al., 2012). Judging from evidence of how the default system functions, these results may suggest a greater negative self-focus and attention to internal thoughts in depressed patients than in controls, coinciding with the rumination processes characteristic of depression. Consistently with this hypothesis, increased default system activation has been associated to individual differences in maladaptive rumination (Hamilton et al., 2011). In psychodynamic terms, this rumination can be described as an “internal dialog”: if they have failed to mentalize some emotional states (which are often relational needs that have been not mirrored in primary relationships), individuals use forms of emotion regulation based on suppression or avoidance (“defense mechanisms” in psychodynamic terms–Freud, 1967), instead of mentalizing their emotional responses flexibly to suit their relational needs.
Clinical Implications: Therapy and Changing Internal Representations
If representations of the self and others are constructed within a close relationship, these internal representations may also be changed by means of a close relationship. The therapeutic relationship can be seen as a relational context in which patients act out their attachment patterns and can make changes to the internal representations of the self and others (Lyddon and Sherry, 2001; Dales, 2008). Researchers have provided strong empirical evidence of the outcome of psychotherapy being mediated by the quality of the therapeutic relationship (Horvath, 2002), with includes the therapist’s empathy (Burns and Nolen-Hoeksema, 1992; Elliott et al., 2011) and ability to engage the patient in a therapeutic alliance (Martin et al., 2000). The importance of therapeutic relationships is a factor spanning all psychotherapy techniques (Meares et al., 2005; Norcross and Lambert, 2011).
In psychodynamic psychotherapy, the work on internal representations of the self and others is also expressed in specific techniques that aim to explore how patients’ internal models influence their relationship with the therapist in order to arrive at cognitive but also emotional insights on the influence of their primary relationships on their current relationships. The most popular example is transference interpretation (Freud, 1912). An evolution of transference interpretation is the “triangle of insight” (Menninger, 1958), widely used in brief psychodynamic psychotherapy (Malan, 1976; Davanloo, 1980), but with potential applications also in other psychotherapeutic approaches. Using the triangle of insight, therapists and patients observe together how past experiences (the first vertex of the triangle) influence current life experiences (the second vertex), and are manifested in the here-and-now of the therapeutic relationship (the third vertex). In the following transcript of psychotherapy intervention, we provide an example of the triangle of insight in Intensive Transactional Analysis Therapy:
T: What’s the problem? (current problem)
P: Well… I have a lot of anxiety when I have to speak in front of lots of people
T: Even now? (Therapist links patient’s current life experience with the here-and-now of the therapeutic relationship)
P: Yes
T: Okay! Where is this anxiety?
P: Here (points to chest). My heart is racing and I am sweating
T: Your heart is racing…and…is something else that is happening?
P: Eh… I have confusion in my head
T: Do you have confusion now?
P: (nods)
T: Can you explain what this confusion is… (Therapist continues the exploration of third vertex of triangle of insight and patient’s emotion regulation)
Conclusion, Outstanding Questions, and Future Directions
The core idea emerging from the present mini-review is that the default system is abnormally activated in patients with depression, consistently with the observation of negative self-focus and rumination in such patients. In line with clinical models coming from psychodynamic theory, these difficulties in emotion regulation can be seen as associated with the existence of rigid, negative internal representations of the self and others. Considering such processes in neurobiological models of emotion dysregulation helps us to build bridges between the theories behind clinical psychology and neuroscience. Several gaps remain, however, in this attempt at integration. One question remaining to be answered concerns how processes of cognitive control and internal representation of the self and others interact in engendering rumination and avoidance instead of adaptive strategies for regulating emotions. With this in mind, future studies should clarify how individual differences in default system activation, and this system’s correlations with other brain networks are associated with the complexity and flexibility of internal representation.
Author Contributions
IM conceived the review and wrote the sections “Introduction,” “Neural mechanisms,” and “Conclusion” of the manuscript. FB wrote “Psychological mechanisms” and MC wrote “Clinical implications”. VC and MS supervised the manuscript. All authors listed, have made substantial, direct and intellectual contribution to the work, and approved it for publication.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Aldao, A., Nolen-Hoeksema, S., and Schweizer, S. (2010). Emotion-regulation strategies across psychopathology: a meta-analytic review. Clin. Psychol. Rev. 30, 217–237. doi: 10.1016/j.cpr.2009.11.004
Bateman, A. W., and Fonagy, P. (2004). Mentalization-based treatment of BPD. J. Personal. Disord. 18, 36–51. doi: 10.1521/pedi.18.1.36.32772
Beck, A. T., Ward, C., and Mendelson, M. (1961). An inventory for measuring depression. Arch. Gen. Psychiatry 4, 561–571. doi: 10.1001/archpsyc.1961.01710120031004
Benelli, E., Mergenthaler, E., Walter, S., Messina, I., Sambin, M., Buchheim, A., et al. (2012). Emotional and cognitive processing of narratives and individual appraisal styles: Recruitment of cognitive control networks vs. modulation of deactivations. Front. Hum. Neurosci. 6:239. doi: 10.3389/fnhum.2012.00239
Binder, J. R., Desai, R. H., Graves, W. W., and Conant, L. L. (2009). Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb. Cortex 19, 2767–2796. doi: 10.1093/cercor/bhp055
Blair, R., and Mitchell, D. (2009). Psychopathy, attention and emotion. Psychol. Med. 39, 543–555. doi: 10.1017/S0033291708003991
Brennan, K. A., Clark, C. L., and Shaver, P. R. (1998). “Self-report measurement of adult attachment: an integrative overview,” in Attachment Theory and Close Relationships, eds J. A. Simpson and W. S. Rholes (New York, NY: Guilford Press), 46–76.
Brink, T. L., Yesavage, J. A., Lum, O., Heersema, P. H., Adey, M. B., and Rose, T. L. (1982). Screening tests for geriatric depression. Clin. Gerontol. 1, 37–43. doi: 10.1300/J018v01n01_06
Brody, A. L., Saxena, S., Stoessel, P., Gillies, L. A., Fairbanks, L. A., Alborzian, S., et al. (2001). Regional brain metabolic changes in patients with major depression treated with either paroxetine or interpersonal therapy: preliminary findings. Arch. Gen. Psychiatry 58, 631–640. doi: 10.1001/archpsyc.58.7.631
Broyd, S. J., Demanuele, C., Debener, S., Helps, S. K., James, C. J., and Sonuga-Barke, E. J. (2009). Default-mode brain dysfunction in mental disorders: a systematic review. Neurosci. Biobehav. Rev. 33, 279–296. doi: 10.1016/j.neubiorev.2008.09.002
Buchheim, A., Erk, S., George, C., Kächele, H., Ruchsow, M., Spitzer, M., et al. (2006). Measuring attachment representation in an fMRI environment: a pilot study. Psychopathology 39, 144–152. doi: 10.1159/000091799
Buckner, R. L., and Carroll, D. C. (2007). Self-projection and the brain. Trends Cogn. Sci. 11, 49–57. doi: 10.1016/j.tics.2006.11.004
Buhle, J. T., Silvers, J. A., Wager, T. D., Lopez, R., Onyemekwu, C., Kober, H., et al. (2014). Cognitive reappraisal of emotion: a meta-analysis of human neuroimaging studies. Cereb. Cortex 24, 2981–2990. doi: 10.1093/cercor/bht154
Burns, D. D., and Nolen-Hoeksema, S. (1992). Therapeutic empathy and recovery from depression in cognitive-behavioral therapy: a structural equation model. J. Consult. Clin. Psychol. 60, 441–449. doi: 10.1037/0022-006X.60.3.441
Calvo, V., and Bianco, F. (2015). Influence of adult attachment insecurities on parenting self-esteem: the mediating role of dyadic adjustment. Front. Psychol. 6:1461. doi: 10.3389/fpsyg.2015.01461
Campbell-Sills, L., and Barlow, D. H. (2007). “Incorporating emotion regulation into conceptualizations and treatments of anxiety and mood disorders,” in Handbook of Emotion Regulation, ed. J. J. Gross (New York, NY: Guilford Press).
Campbell-Sills, L., Barlow, D. H., Brown, T. A., and Hofmann, S. G. (2006). Effects of suppression and acceptance on emotional responses of individuals with anxiety and mood disorders. Behav. Res. Ther. 44, 1251–1263. doi: 10.1016/j.brat.2005.10.001
Cassidy, J. (1994). Emotion regulation: Influences of attachment relationships. Monogr. Soc. Res. Child Dev. 59, 228–249. doi: 10.2307/1166148
Cole-Detke, H., and Kobak, R. (1996). Attachment processes in eating disorder and depression. J. Consult. Clin. Psychol. 64, 282–290. doi: 10.1037/0022-006X.64.2.282
Dales, S. (2008). Attachment, affect regulation and mutual synchrony in adult psychotherapy. Am. J. Psychother. 62, 283–312.
Dotson, V. M., Beason-Held, L., Kraut, M. A., and Resnick, S. M. (2009). Longitudinal study of chronic depressive symptoms and regional cerebral blood flow in older men and women. Int. J. Geriatr. Psychiatry 24, 809–819. doi: 10.1002/gps.2298
Drevets, W. C. (2001). Neuroimaging and neuropathological studies of depression: Implications for the cognitive-emotional features of mood disorders. Curr. Opin. Neurobiol. 11, 240–249. doi: 10.1016/S0959-4388(00)00203-8
Drevets, W. C., Videen, T. O., Price, J. L., Preskorn, S. H., Carmichael, S. T., and Raichle, M. E. (1992). A functional anatomical study of unipolar depression. J. Neurosci. 12, 3628–3641.
Duhameau, B., Ferré, J., Jannin, P., Gauvrit, J., Vérin, M., Millet, B., et al. (2010). Chronic and treatment-resistant depression: a study using arterial spin labeling perfusion MRI at 3Tesla. Psychiatry Res. Neuroimag. 182, 111–116. doi: 10.1016/j.pscychresns.2010.01.009
Elliott, R., Bohart, A. C., Watson, J. C., and Greenberg, L. S. (2011). Empathy. Psychotherapy (Chicago, Ill.) 48, 43–49. doi: 10.1037/a0022187
Feeney, B. C., and Cassidy, J. (2003). Reconstructive memory related to adolescent-parent conflict interactions: the influence of attachment-related representations on immediate perceptions and changes in perceptions over time. J. Pers. Soc. Psychol. 85, 945. doi: 10.1037/0022-3514.85.5.945
Ferenczi, S. (1933). Final Contributions to the Problems and Methods of Psychoanalysis (trans. Eric Mosbacher and others), ed. Karnac Books (New York, NY: Michael Balint).
Fonagy, P., Bateman, A., and Bateman, A. (2011). The widening scope of mentalizing: a discussion. Psychol. Psychother. Theor. Res. Pract. 84, 98–110. doi: 10.1111/j.2044-8341.2010.02005.x
Fonagy, P., Gergely, G., and Target, M. (2007). The parent–infant dyad and the construction of the subjective self. J. Child Psychol. Psychiatry 48, 288–328. doi: 10.1111/j.1469-7610.2007.01727.x
Fonagy, P., and Target, M. (1997). Attachment and reflective function: their role in self-organization. Dev. Psychopathol. 9, 679–700. doi: 10.1017/S0954579497001399
Fox, M. D., Snyder, A. Z., Vincent, J. L., Corbetta, M., Van Essen, D. C., and Raichle, M. E. (2005). The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc. Natl. Acad. Sci. U.S.A. 102, 9673–9678. doi: 10.1073/pnas.0504136102
Fraley, C. R., Niedenthal, P. M., Marks, M., Brumbaugh, C., and Vicary, A. (2006). Adult attachment and the perception of emotional expressions: probing the hyperactivating strategies underlying anxious attachment. J. Pers. 74, 1163–1190. doi: 10.1111/j.1467-6494.2006.00406.x
Freud, A. (1955). Safeguarding the Emotional Health of Our Children: An Inquiry into the Concept of the Rejecting Mother. New York, NY: Child Welfare League of America.
Freud, S. (1912). “The dynamics of transference,” in Classics in Psychoanalytic Techniques, ed. R.Langs (New york: Rowman & Littlefield Publishers, Inc), 3.
Freud, A., and Burlingham, D. (1974). Infants Without Families and Reports on the Hampstead Nurseries, 1939-1945 / by Anna Freud, in Collaboration with Dorothy Burlingham. London: Hogarth Press.
Garnefski, N., and Kraaij, V. (2006). Relationships between cognitive emotion regulation strategies and depressive symptoms: a comparative study of five specific samples. Pers. Individ. Diff. 40, 1659–1669. doi: 10.1016/j.paid.2005.12.009
Gobbini, M. I., Leibenluft, E., Santiago, N., and Haxby, J. V. (2004). Social and emotional attachment in the neural representation of faces. Neuroimage 22, 1628–1635. doi: 10.1016/j.neuroimage.2004.03.049
Grecucci, A., Theuninck, A., Frederickson, J., and Job, R. (2015). “Mechanisms of social emotion regulation: From neuroscience to psychotherapy,” in Handbook of Emotion Regulation: Processes, Cognitive Effects and Social Consequences, ed. AA.VV (Hauppauge, NY: Nova Publishing), 57–84.
Greening, S. G., Osuch, E. A., Williamson, P. C., and Mitchell, D. G. (2014). The neural correlates of regulating positive and negative emotions in medication-free major depression. Soc. Cogn. Affect. Neurosci. 9, 628–637. doi: 10.1093/scan/nst027
Gross, J. J. (2002). Emotion regulation: affective, cognitive, and social consequences. Psychophysiology 39, 281–291. doi: 10.1017/S0048577201393198
Guy, W. (ed.). (1976). “Clinical Global Impressions,” in ECDEU Assessment Manual for Psychopharmacology. Rockville, MD: National Institute of Mental Health.
Hamilton, J. P., Furman, D. J., Chang, C., Thomason, M. E., Dennis, E., and Gotlib, I. H. (2011). Default-mode and task-positive network activity in major depressive disorder: implications for adaptive and maladaptive rumination. Biol. Psychiatry 70, 327–333. doi: 10.1016/j.biopsych.2011.02.003
Hamilton, M. (1960). A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 23, 56–62. doi: 10.1136/jnnp.23.1.56
Horvath, A. O., Del Re, A. C., Flückiger, C., and Symonds, D. (2002). “Alliance in individual psychotherapy,” in Psychotherapy Relationships that Work: Therapist Contributions and Responsiveness to Patients, ed. J. C. Norcross (New York: Oxford University Press), 37–70.
Johnstone, T., van Reekum, C. M., Urry, H. L., Kalin, N. H., and Davidson, R. J. (2007). Failure to regulate: counterproductive recruitment of top-down prefrontal-subcortical circuitry in major depression. J. Neurosci. Official J. Soc. Neurosci. 27, 8877–8884. doi: 10.1523/JNEUROSCI.2063-07.2007
Kennedy, S. H., Evans, K. R., Krüger, S., Mayberg, H. S., Meyer, J. H., McCann, S., et al. (2001). Changes in regional brain glucose metabolism measured with positron emission tomography after paroxetine treatment of major depression. Am. J. Psychiatry 158, 899–905. doi: 10.1176/appi.ajp.158.6.899
Kernberg, O. (1976). Object-Relations Theory and Clinical Psychoanalysis. New York, NY: Jason Aronson.
Kobak, R. R., and Sceery, A. (1988). Attachment in late adolescence: working models, affect regulation, and representations of self and others. Child Dev. 59, 135–146. doi: 10.2307/1130395
Kohl, A., Rief, W., and Glombiewski, J. A. (2012). How effective are acceptance strategies? A meta-analytic review of experimental results. J. Behav. Ther. Exp. Psychiatry 43, 988–1001. doi: 10.1016/j.jbtep.2012.03.004
Kohut, H., and Wolf, E. (1978). The disorders of the self and their treatment: an outline. Int. J. Psychoanal. 60, 3–27.
Liverant, G. I., Brown, T. A., Barlow, D. H., and Roemer, L. (2008). Emotion regulation in unipolar depression: the effects of acceptance and suppression of subjective emotional experience on the intensity and duration of sadness and negative affect. Behav. Res. Ther. 46, 1201–1209. doi: 10.1016/j.brat.2008.08.001
Lou, H. C., Luber, B., Crupain, M., Keenan, J. P., Nowak, M., Kjaer, T. W., et al. (2004). Parietal cortex and representation of the mental self. Proc. Natl. Acad. Sci. U.S.A. 101, 6827–6832. doi: 10.1073/pnas.0400049101
Lui, S., Parkes, L. M., Huang, X., Zou, K., Chan, R. C., Yang, H., et al. (2009). Depressive disorders: focally altered cerebral perfusion measured with arterial spin-labeling MR imaging 1. Radiology 251, 476–484. doi: 10.1148/radiol.2512081548
Lyddon, W. J., and Sherry, A. (2001). Developmental personality styles: an attachment theory conceptualization of personality disorders. J. Counsel. Dev. 79, 405–414. doi: 10.1002/j.1556-6676.2001.tb01987.x
Malan, D. H. (1976). The Frontier of Brief Psychotherapy: An Example of the Convergence of Research and Clinical Practice. New York, NY: Plenum Medical Book.
Malik, S., Wells, A., and Wittkowski, A. (2015). Emotion regulation as a mediator in the relationship between attachment and depressive symptomatology: a systematic review. J. Affect. Disord. 172, 428–444. doi: 10.1016/j.jad.2014.10.007
Martin, D. J., Garske, J. P., and Davis, M. K. (2000). Relation of the therapeutic alliance with outcome and other variables: a meta-analytic review. J. Consult. Clin. Psychol. 68, 438–450. doi: 10.1037/0022-006X.68.3.438
Mason, M. F., Norton, M. I., Van Horn, J. D., Wegner, D. M., Grafton, S. T., and Macrae, C. N. (2007). Wandering minds: the default network and stimulus-independent thought. Science (New York) 315, 393–395. doi: 10.1126/science.1131295
Mayberg, H. S., Brannan, S. K., Mahurin, R. K., Jerabek, P. A., Brickman, J. S., Tekell, J. L., et al. (1997). Cingulate function in depression: a potential predictor of treatment response. Neuroreport 8, 1057–1061. doi: 10.1097/00001756-199703030-00048
Meares, R., Butt, D., Henderson-Brooks, C., and Samir, H. (2005). A poetics of change. Psychoanal. Dialogues 15, 661–680. doi: 10.1080/10481881509348858
Menon, V. (2011). Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn. Sci. 15, 483–506. doi: 10.1016/j.tics.2011.08.003
Messina, I., Bianco, S., Sambin, M., and Viviani, R. (2015). Executive and semantic processes in reappraisal of negative stimuli: insights from a meta-analisys of neuroimaging studiES. Front. Psychol. 6:956. doi: 10.3389/fpsyg.2015.00956
Messina, I., Sambin, M., Palmieri, A., and Viviani, R. (2013). Neural correlates of psychotherapy in anxiety and depression: a meta-analysis. PLoS ONE 8:e74657. doi: 10.1371/journal.pone.0074657
Mickelson, K. D., Kessler, R. C., and Shaver, P. R. (1997). Adult attachment in a nationally representative sample. J. Pers. Soc. Psychol. 73, 1092–1106. doi: 10.1037/0022-3514.73.5.1092
Mikulincer, M., Shaver, P. R., and Pereg, D. (2003). Attachment theory and affect regulation: the dynamics, development, and cognitive consequences of attachment-related strategies. Motiv. Emot. 27, 77–102. doi: 10.1023/A:1024515519160
Monkul, E. S., Silva, L. A., Narayana, S., Peluso, M. A., Zamarripa, F., Nery, F. G., et al. (2012). Abnormal resting state corticolimbic blood flow in depressed unmedicated patients with major depression: a 15O-H2O PET study. Hum. Brain Mapp. 33, 272–279. doi: 10.1002/hbm.21212
Montgomery, S. A., Smeyatsky, N., Ruiter, M., and Montgomery, D. (1985). Profiles of antidepressant activity with the Montgomery-Asberg depression rating scale. Acta Psychiatr. Scand. 72, 38–42. doi: 10.1111/j.1600-0447.1985.tb08073.x
Nolen-Hoeksema, S. (2000). The role of rumination in depressive disorders and mixed anxiety/depressive symptoms. J. Abnorm. Psychol. 109, 504–511. doi: 10.1037/0021-843X.109.3.504
Norcross, J. C., and Lambert, M. J. (2011). Evidence-based therapy relationships. Psychother. Relationsh. Evid. Based Responsiveness 2, 3–23. doi: 10.1093/acprof:oso/9780199737208.003.0001
Northoff, G., Heinzel, A., de Greck, M., Bermpohl, F., Dobrowolny, H., and Panksepp, J. (2006). Self-referential processing in our brain–a meta-analysis of imaging studies on the self. Neuroimage 31, 440–457. doi: 10.1016/j.neuroimage.2005.12.002
Ochsner, K. N., and Gross, J. J. (2005). The cognitive control of emotion. Trends Cogn. Sci. 9, 242–249. doi: 10.1016/j.tics.2005.03.010
Orosz, A., Jann, K., Federspiel, A., Horn, H., Höfle, O., Dierks, T., et al. (2012). Reduced cerebral blood flow within the default-mode network and within total gray matter in major depression. Brain Connect. 2, 303–310. doi: 10.1089/brain.2012.0101
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., and Shulman, G. L. (2001a). A default mode of brain function. Proc. Natl. Acad. Sci. U.S.A. 98, 676–682. doi: 10.1073/pnas.98.2.676
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., and Shulman, G. L. (2001b). A default mode of brain function. Proc. Natl. Acad. Sci. U.S.A. 98, 676–682. doi: 10.1073/pnas.98.2.676
Raichle, M. E., and Snyder, A. Z. (2007). A default mode of brain function: a brief history of an evolving idea. Neuroimage 37, 1083–1090. doi: 10.1016/j.neuroimage.2007.02.041 ; discussion 1097–1099.
Roffman, J. L., Witte, J. M., Tanner, A. S., Ghaznavi, S., Abernethy, R. S., Crain, L. D., et al. (2014). Neural predictors of successful brief psychodynamic psychotherapy for persistent depression. Psychother. Psychosom. 83, 364–370. doi: 10.1159/000364906
Rosenstein, D. S., and Horowitz, H. A. (1996). Adolescent attachment and psychopathology. J. Consult. Clin. Psychol. 64, 244–253. doi: 10.1037/0022-006X.64.2.244
Saxe, R., Moran, J. M., Scholz, J., and Gabrieli, J. (2006). Overlapping and non-overlapping brain regions for theory of mind and self reflection in individual subjects. Soc. Cogn. Affect. Neurosci. 1, 229–234. doi: 10.1093/scan/nsl034
Saxena, S., Brody, A. L., Ho, M. L., Alborzian, S., Ho, M. K., Maidment, K. M., et al. (2001). Cerebral metabolism in major depression and obsessive-compulsive disorder occurring separately and concurrently. Biol. Psychiatry 50, 159–170. doi: 10.1016/S0006-3223(01)01123-4
Shaver, P. R., and Mikulincer, M. (2007). “Adult attachment strategies and the regulation of emotion,” in Handbook of Emotion Regulation, ed. J. J. Gross (New York, NY: Guilford Press), 446–465.
Siegle, G. J., Thompson, W., Carter, C. S., Steinhauer, S. R., and Thase, M. E. (2007). Increased amygdala and decreased dorsolateral prefrontal BOLD responses in unipolar depression: Related and independent features. Biol. Psychiatry 61, 198–209. doi: 10.1016/j.biopsych.2006.05.048
Spitz, R. A. (1945). Hospitalism; an inquiry into the genesis of psychiatric conditions in early childhood. Psychoanal. Study Child 1, 53–74.
Spitz, R. A. (1965). The First Year of Life: A Psychoanalytic Study of Normal and Deviant Development of Object Relations. Madison: International Universities Press.
Spreng, R. N., Mar, R. A., and Kim, A. S. (2009). The common neural basis of autobiographical memory, prospection, navigation, theory of mind, and the default mode: a quantitative meta-analysis. J. Cogn. Neurosci. 21, 489–510. doi: 10.1162/jocn.2008.21029
Sroufe, L. A. (2005). Attachment and development: a prospective, longitudinal study from birth to adulthood. Attach. Hum. Dev. 7, 349–367. doi: 10.1080/14616730500365928
Taylor, S. F., and Liberzon, I. (2007). Neural correlates of emotion regulation in psychopathology. Trends Cogn. Sci. 11, 413–418. doi: 10.1016/j.tics.2007.08.006
Videbech, P., Ravnkilde, B., Pedersen, A. R., Egander, A., Landbo, B., Rasmussen, N., et al. (2001). The danish PET/depression project: PET findings in patients with major depression. Psychol. Med. 31, 1147–1158. doi: 10.1017/S0033291701004469
Viviani, R. (2013). Emotion regulation, attention to emotion, and the ventral attentional network. Front. Hum. Neurosci. 7:746. doi: 10.3389/fnhum.2013.00746
Viviani, R. (2014). Neural correlates of emotion regulation in the ventral prefrontal cortex and the encoding of subjective value and economic utility. Front. Psychiatry 5:123. doi: 10.3389/fpsyt.2014.00123
Webb, T. L., Miles, E., and Sheeran, P. (2012). Dealing with feeling: a meta-analysis of the effectiveness of strategies derived from the process model of emotion regulation. Psychol. Bull. 138, 775–808. doi: 10.1037/a0027600
Wenzlaff, R. M., and Rude, S. S. (2002). Cognitive vulnerability to depression: the role of thought suppression and attitude certainty. Cogn. Emot. 16, 533–548. doi: 10.1080/02699930143000338
Whitfield-Gabrieli, S., and Ford, J. M. (2012). Default mode network activity and connectivity in psychopathology. Annu. Rev. Clin. Psychol. 8, 49–76. doi: 10.1146/annurev-clinpsy-032511-143049
Winnicott, D. (1967). The Predicament of the Family: A Psycho-Analytical Symposium. London: Hogarth Press and The Institute of Psycho-Analysis.
Winnicott, D. W. (1965). “Studies in the theory of emotional development,” in The Maturational Processes and the Facilitating Environment, ed. M. M. R. Khan (London: Hogarth Press), 37–55.
Yarkoni, T., Poldrack, R. A., Nichols, T. E., Van Essen, D. C., and Wager, T. D. (2011). Large-scale automated synthesis of human functional neuroimaging data. Nat. Methods 8, 665–670. doi: 10.1038/nmeth.1635
Keywords: depression, default system, emotion regulation, self, psychodynamic, psychotherapy, neuroimaging
Citation: Messina I, Bianco F, Cusinato M, Calvo V and Sambin M (2016) Abnormal Default System Functioning in Depression: Implications for Emotion Regulation. Front. Psychol. 7:858. doi: 10.3389/fpsyg.2016.00858
Received: 30 March 2016; Accepted: 24 May 2016;
Published: 10 June 2016.
Edited by:
Alessandro Grecucci, University of Trento, ItalyReviewed by:
Eric S. Allard, Cleveland State University, USASteven Grant Greening, Louisiana State University, USA
Copyright © 2016 Messina, Bianco, Cusinato, Calvo and Sambin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Irene Messina, aXJlbmUtbWVzc2luYUBob3RtYWlsLmNvbQ==
 Irene Messina
Irene Messina Francesca Bianco
Francesca Bianco Maria Cusinato
Maria Cusinato Vincenzo Calvo
Vincenzo Calvo Marco Sambin
Marco Sambin