- Department of Plant Biology and Crop Science, Rothamsted Research, Harpenden, UK
New pathogen-host interaction mechanisms can be revealed by integrating mutant phenotype data with genetic information. PHI-base is a multi-species manually curated database combining peer-reviewed published phenotype data from plant and animal pathogens and gene/protein information in a single database.
PHI-base is a multi-species knowledge database capturing the phenotypes available on forward and reverse mutants from 231 pathogenic organisms described in the literature. Plant pathogens represent 60% of the species within PHI-base. Simple and advanced search tools, available at www.phi-base.org, allow users to query PHI-base directly. Flat file downloads enable larger comparative biology studies, systems biology approaches and a richer annotation of genomes, transcriptomes and proteome data sets. Since 2014, phenotype information from PHI-base is directly displayed in pathogen genome browsers accessible at www.phytopathdb.org (Kersey et al., 2014). PHI-base regularly interacts with the international community to provide researchers with effective query tools and new data types to study pathogen-host interactions.
Available online since 2005, PHI-base catalogs experimentally verified pathogenicity, virulence and effector genes from fungal, protist, and bacterial pathogens which infect animal, plant, fish, insect, and/or fungal hosts (Urban et al., 2015). PHI-base is a database devoted to the identification and presentation of information on pathogenicity and effector genes and their host interactions. PHI-base was developed out of a need for a knowledge database enabling the discovery of candidate targets in medically and agronomically important species for intervention with chemistries and/or host modifications. Recent bioinformatics studies enabled by whole-database downloads of PHI-base, include comparative analyses, genome/transcript and proteome annotations, and system biology approaches (Hu et al., 2014; Zhang et al., 2014). PHI-base has been cited in 122 published articles including genetics, genomics and bioinformatics research and review articles (for an up-to-date list, see the “About” page of the PHI-base website). In 2014, the web site had more than 6000 visits and the entire content was downloaded >300 times. Phenotypic outcome data from PHI-base are also displayed directly in genome browsers as permanent tracks in public genome sequence resources such as Ensembl Fungi (Figure 1). Through a simple system of color coding and using nine high level PHI-base phenotypes (Urban et al., 2015), genomic features such as pathogenicity islands can directly be investigated.
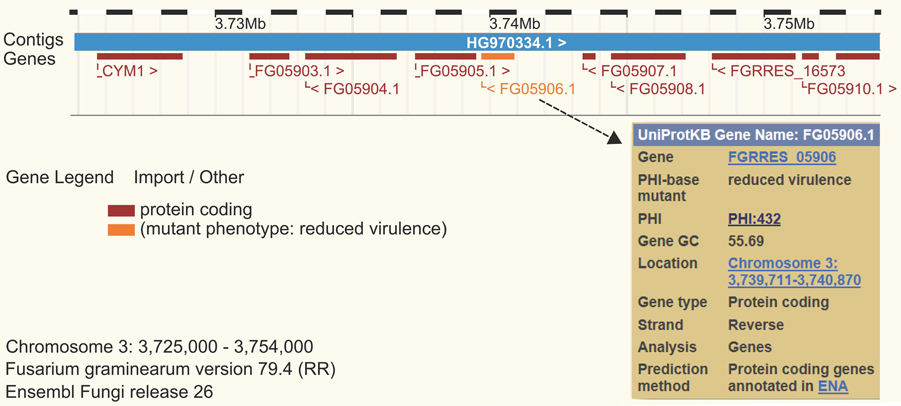
Figure 1. Ensembl genome browser view for Fusarium graminearum. The website at http://fungi.ensembl.org/ Fusarium_graminearum was searched for the gene id FGSG_05906 encoding the secreted lipase gene Fgfgl1. The PHI-base phenotype of the mutant is displayed and color coded in orange as “reduced virulence.”
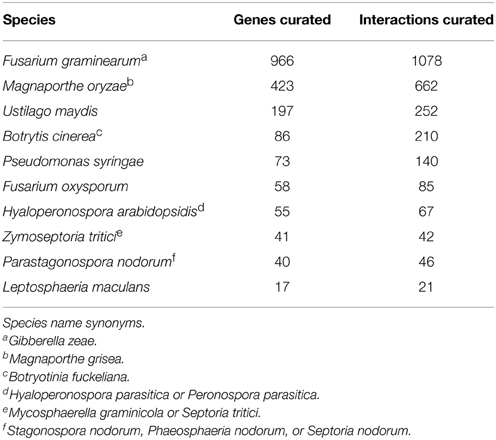
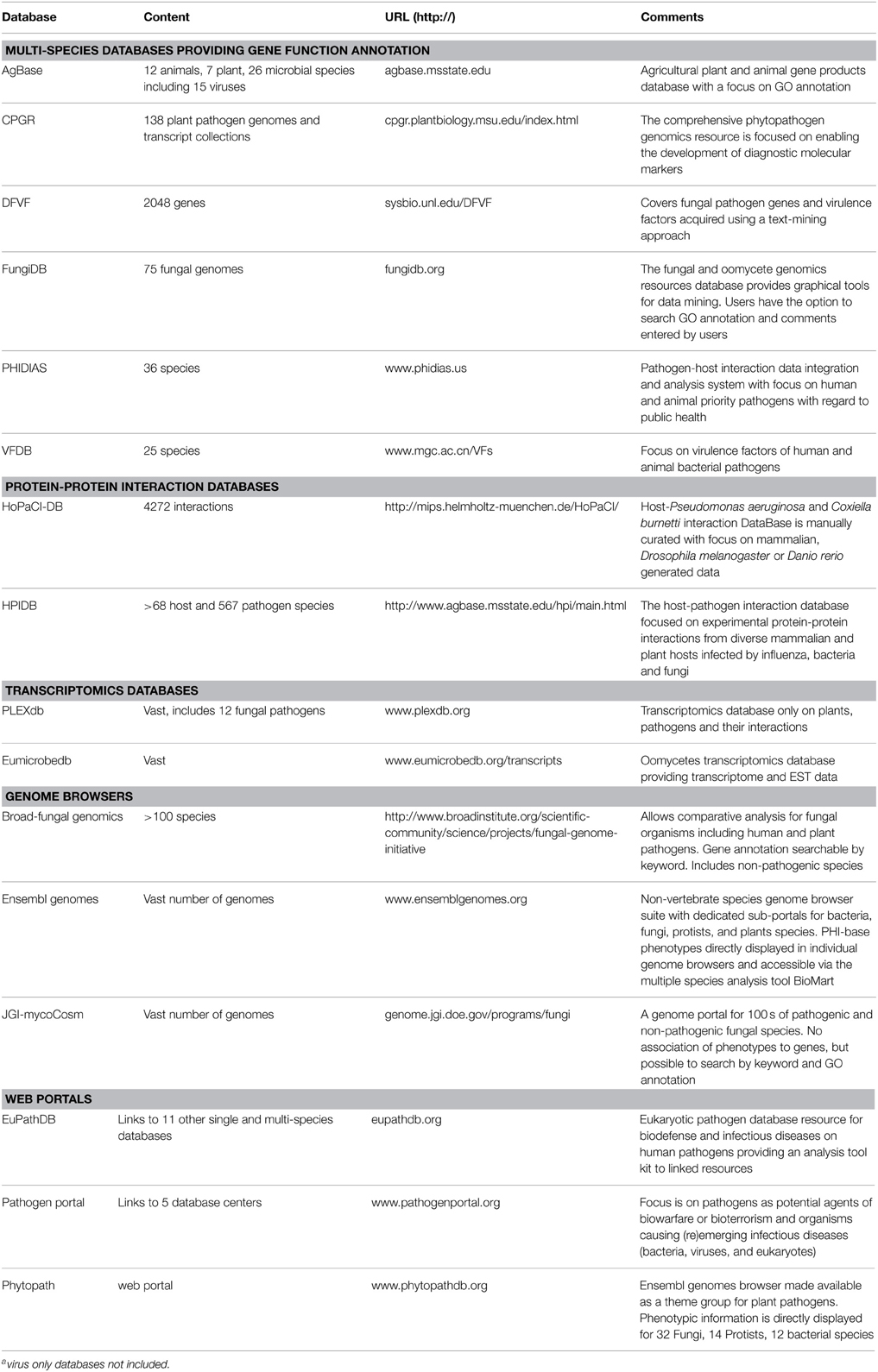
The latest PHI-base release, version 3.8, contains a total of 3562 pathogen genes tested in 3697 plant- and 1257 animal-pathogen interactions. The top 10 plant pathogens are listed in Table 1. The data in PHI-base is obtained by biocuration scientists who extract the relevant information from peer-reviewed published articles in a manual curation workflow that includes the evaluation of full text, figures and tables, to create computable data records using controlled vocabularies and ontologies. This approach generates a unique level of detail and breadth compared to automated approaches and thus provides instant access to a catalog of gold standard curated gene/protein function and host phenotypic information. Various complementary multi-species databases on pathogens exist that provide gene function annotation. Each specializes in particular species/pathogen groups and/or uses only automated approaches to knowledge acquisition (Table 2). Other resources are more geared to the analysis of host-pathogen interactions by providing protein-protein interaction (PPI) data, transcriptomics and genome assembly datasets or provide WEB portal linking to multiple databases and providing advanced analysis tools.
Future plans for PHI-base include the development of an online tool to allow author curation of published pathogen-host interactions from any pathogenic species. This new feature will be based on the Canto curation tool for PomBase (Rutherford et al., 2014). A refined PHI-base website will become available in 2015 to allow the display of additional manually curated information, including data on host target genes/proteins.
Funding
This work is supported by the UK Biotechnology and Biological Sciences Research Council (BBSRC) (BB/I001077/1, BB/I000488/1, BB/K020056/1). PHI-base receives additional support from the BBSRC as a National Capability (BB/J004383/1). Funding for the open access charge was obtained from the Research Councils UK Open Access Fund.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to thank all contributing scientists, who provided expert knowledge by reviewing PHI-base data and suggesting articles for curation. We thank Drs Helder Pedro, Paul Kersey, Uma Maheswari, and Dan Staines at the European Bioinformatics Institute (Cambridge, UK) for discussions and significantly improving the pathogen species content within Ensembl Genomes. Rashmi Pant and Arathi Raghunath at Molecular Connections (Bangalore, India) are thanked for expert data capture.
References
Hu, X., Xiao, G., Zheng, P., Shang, Y., Su, Y., Zhang, X., et al. (2014). Trajectory and genomic determinants of fungal-pathogen speciation and host adaptation. Proc. Natl. Acad. Sci. U.S.A. 111, 16796–16801. doi: 10.1073/pnas.1412662111
Kersey, P. J., Allen, J. E., Christensen, M., Davis, P., Falin, L. J., Grabmueller, C., et al. (2014). Ensembl Genomes 2013: scaling up access to genome-wide data. Nucleic Acids Res. 42, D546–D552. doi: 10.1093/nar/gkt979
Rutherford, K. M., Harris, M. A., Lock, A., Oliver, S. G., and Wood, V. (2014). Canto: an online tool for community literature curation. Bioinformatics 30, 1791–1792. doi: 10.1093/bioinformatics/btu103
Urban, M., Pant, R., Raghunath, A., Irvine, A. G., Pedro, H., and Hammond-Kosack, K. E. (2015). The pathogen-host interactions database (PHI-base): additions and future developments. Nucleic Acids Res. 43, D645–D655. doi: 10.1093/nar/gku1165
Keywords: gene regulatory networks, plant diseases, protein interaction mapping, genetic recombination, comparative genomics, horizontal gene transfer, phytopathogens, emerging diseases
Citation: Urban M, Irvine AG, Cuzick A and Hammond-Kosack KE (2015) Using the pathogen-host interactions database (PHI-base) to investigate plant pathogen genomes and genes implicated in virulence. Front. Plant Sci. 6:605. doi: 10.3389/fpls.2015.00605
Received: 11 May 2015; Accepted: 22 July 2015;
Published: 06 August 2015.
Edited by:
Dan MacLean, The Sainsbury Laboratory, UKReviewed by:
Ana Elena Dorantes-Acosta, Universidad Veracruzana, MexicoOliver Furzer, University of East Anglia, UK
Copyright © 2015 Urban, Irvine, Cuzick and Hammond-Kosack. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Martin Urban, Department of Plant Biology and Crop Science, Rothamsted Research, West Common, Harpenden, Herts AL5 2JQ, UK,bWFydGluLnVyYmFuQHJvdGhhbXN0ZWQuYWMudWs=
 Martin Urban
Martin Urban Alistair G. Irvine
Alistair G. Irvine Alayne Cuzick
Alayne Cuzick Kim E. Hammond-Kosack
Kim E. Hammond-Kosack