Abstract
Heavy metal contamination of soil and water causing toxicity/stress has become one important constraint to crop productivity and quality. This situation has further worsened by the increasing population growth and inherent food demand. It has been reported in several studies that counterbalancing toxicity due to heavy metal requires complex mechanisms at molecular, biochemical, physiological, cellular, tissue, and whole plant level, which might manifest in terms of improved crop productivity. Recent advances in various disciplines of biological sciences such as metabolomics, transcriptomics, proteomics, etc., have assisted in the characterization of metabolites, transcription factors, and stress-inducible proteins involved in heavy metal tolerance, which in turn can be utilized for generating heavy metal-tolerant crops. This review summarizes various tolerance strategies of plants under heavy metal toxicity covering the role of metabolites (metabolomics), trace elements (ionomics), transcription factors (transcriptomics), various stress-inducible proteins (proteomics) as well as the role of plant hormones. We also provide a glance of some strategies adopted by metal-accumulating plants, also known as “metallophytes.”
Introduction
During the last few decades, increased anthropogenic activities, rapid industrialization, and modern agricultural practices have resulted in increased heavy metal contamination in the environment, which causes toxicity to the living organisms (Eapen and D'souza, 2005; Kavamura and Esposito, 2010; Miransari, 2011). Large areas of land have been contaminated with heavy metals due to the use of pesticides, fertilizers, municipal and compost wastes, and also due to heavy metal release from smelting industries and metalliferous mines (Yang et al., 2005). Although many heavy metals occur naturally in the earth's crust at various levels, the problem arises when they are released in excess into the environment due to natural and/or anthropogenic activities. The 53 elements belonging to the d-block have been categorized as “heavy metals” based on their density (>5 g/cm3) (Jarup, 2003). During evolution of angiosperms, only 19 elements such as C, O, H, Mg, S, N, Ca, P, and K (macronutrients) and Cu, Zn, Mn, Fe, Mo, B, Ni, Co, Cl, and B (micronutrients) were selected for basic metabolism (Ernst, 2006). In addition, Si is also considered as a beneficial element, and it has been reported to be involved in the maintenance of plant structures in some plants (Epstein, 1999). Macro and micronutrients play an important role in physiological and biochemical processes of plants such as chlorophyll biosynthesis, photosynthesis, DNA synthesis, protein modifications, redox reactions in the chloroplast and the mitochondrion, sugar metabolism, and nitrogen fixation. For example, Zn is a cofactor for more than 300 enzymes and 200 transcription factors associated with the maintenance of membrane integrity, auxin metabolism, and reproduction (Marschner, 1995; Barker and Pilbeam, 2007; Briat et al., 2007; Williams and Pittman, 2010; Prasad, 2012; Ricachenevsky et al., 2013). However, at elevated concentrations, heavy metals produce severe toxicity symptoms in plants, and therefore, their uptake and utilization are tightly controlled by the plant cells (Janicka-Russak et al., 2008; Saito et al., 2010; Singh et al., 2012; Srivastava et al., 2012; DalCorso et al., 2013a; Farias et al., 2013; Fidalgo et al., 2013). Some heavy metals, such as Cd, Cr, Pb, Al, Hg, etc., although being non-essential and without physiological function, are very toxic even at very low concentrations (Ernst et al., 2008; Janicka-Russak et al., 2008; Garzón et al., 2011; Hayat et al., 2012; Shahid et al., 2012; Chong-qing et al., 2013; Gill et al., 2013). Essential and non-essential heavy metals generally produce common toxic effects on plants, such as low biomass accumulation, chlorosis, inhibition of growth and photosynthesis, altered water balance and nutrient assimilation, and senescence, which ultimately cause plant death.
In addition to adverse impacts on plants, heavy metals pose threat to human health due to their persistence in nature. For instance, Pb is one of the most toxic heavy metals that has soil retention time of 150–5000 years and reported to maintain its concentration high for as long as 150 years (NandaKumar et al., 1995; Yang et al., 2005). Plants growing in heavy metal-contaminated sites generally accumulate higher amounts of heavy metals, and thus, contamination of food chain occurs. Contaminated food chain acts as a primary route for the entry of heavy metals into animal and human tissues, making them prone to several diseases that range from dermatitis to various types of cancers (McLaughlin et al., 1999). This problem might become even worse if sufficient measures are not taken at the right time. Therefore, research in this area is driven by the hope to decrease the entry of heavy metals in crop plants, thereby reducing the risk of contamination in animals and human beings.
Abiotic stresses are estimated to be the main cause for global crop yield reduction of ca. 70%, and thus, are considered a great constraint to crop production (Acquaah, 2007; Jewell et al., 2010). This situation has worsened due to disturbed equilibrium between crop productivity and population growth. Therefore, it is especially important to understand plants' responses to such stressors, particularly heavy metals, in order to find new methods for improving crops quantitatively and qualitatively. Currently, studies are being performed to address the above mentioned problems and have majorly focused on “omic” tools that take into consideration of ionomics (trace elements), metabolomics (metabolome), transcriptomics (transcriptome), and proteomics (proteome). The data obtained will provide insights that might help in enhancing stress tolerance and be employed in breeding and engineering programs aiming at developing plants with new and desired agronomical traits (Lee et al., 2007; Atkinson and Urwin, 2012). In this context, this review is focused on several aspects, from plant responses to heavy metals (considering sensitive as well as metallophytes) to the role of ionomics, metabolomics, transcriptomics, and proteomics in the regulation of heavy metal tolerance (Figure 1).
Figure 1
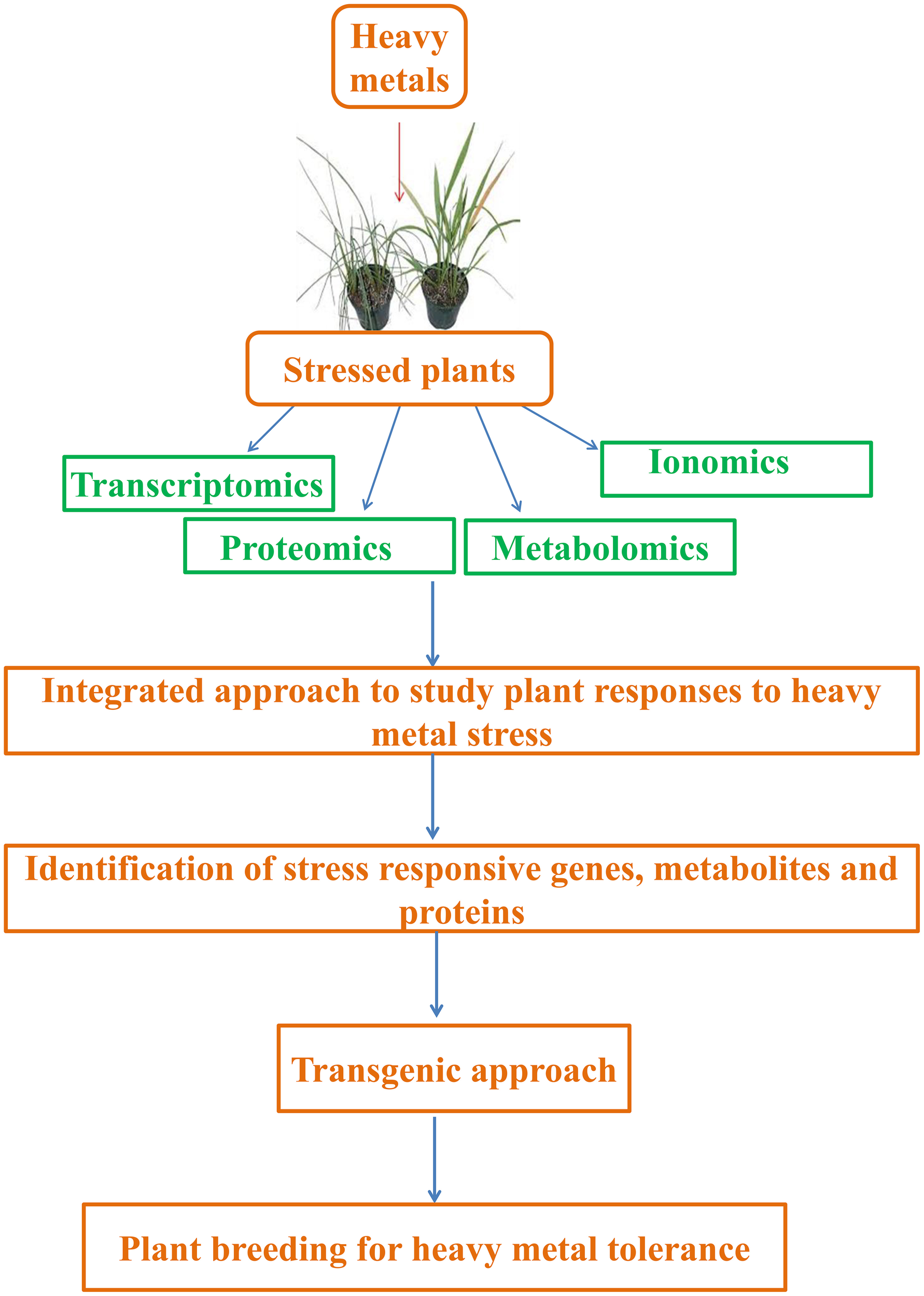
Integrated approach to study plant responses to heavy metal stress. Transcriptomics, proteomics, metabolomics, and ionomics are useful tools that can help us to decipher and analyze active regulatory networks controlling heavy metal stress responses and tolerance.
Plant responses to heavy metal stress
Being sessile organisms, plants cannot escape unwanted changes in the environment. Exposure to heavy metals triggers a wide range of physiological and biochemical alterations, and plants have to develop and/or adopt a series of strategies that allow them to cope with the negative consequences of heavy metal toxicity. Plants respond to external stimuli including heavy metal toxicity via several mechanisms. These include (i) sensing of external stress stimuli, (ii) signal transduction and transmission of a signal into the cell, and (iii) triggering appropriate measures to counterbalance the negative effects of stress stimuli by modulating the physiological, biochemical, and molecular status of the cell. At the whole plant level, it is difficult to measure sensing and changes in the signal transduction after exposing plants to heavy metal stress. However, monitoring early responses, such as oxidative stress, transcriptomic and proteomic changes, or accumulation of metabolites, might be useful to study sensing and signal transduction changes that take place after plants' exposure to stress. For instance, Tamás et al. (2010) reported that early signs of metal toxicity in barley were similar to water deficiency signs, and thus, overexpression of genes related to dehydration stress in barley was found after exposure to Cd and Hg. Similar to this, Hernandez et al. (2012) reported oxidative stress and glutathione depletion in alfalfa roots as early signs of sensing and signal transduction after exposure to heavy metals. In another study by Zhang et al. (2002), seed germination and seedling growth of wheat was found to be inhibited due to high concentration of As. Similarly, Imran et al. (2013) reported reduction in plumule and radicle length of Helainthus annuus L. seedlings when exposed to As. In addition, As has also been reported to decrease the photosynthetic pigment, damage chloroplast membrane, and decrease enzyme activity by reacting with the sulfhydryl group of proteins and also reported to alter nutrient balance and protein metabolism (Li et al., 2006; Singh et al., 2009; Ahsan et al., 2010).
Heavy metals exert toxicities in plants through four proposed mechanisms. These include (i) similarities with the nutrient cations, which result into a competition for absorption at root surface; for example, As and Cd compete with P and Zn, respectively, for their absorption; (ii) direct interaction of heavy metals with sulfhydryl group (-SH) of functional proteins, which disrupts their structure and function, and thus, renders them inactive; (iii) displacement of essential cations from specific binding sites that lead to a collapse of function; and (iv) generation of reactive oxygen species (ROS), which consequently damages the macromolecules (Sharma and Dietz, 2009; DalCorso et al., 2013a).
The roots of sessile plants are the first organ that encounters heavy metals, and thus, roots have been widely studied to assess the impact of a stressor. Plants growing on heavy metal-rich soils suffer from both decreased growth and yield (Keunen et al., 2011), indicating an implication of heavy metal toxicity in hampering the overall growth performance of the stressed plants (Kikui et al., 2005; Panda et al., 2009; Buendía-González et al., 2010; Gangwar et al., 2010, 2011; Gangwar and Singh, 2011; Eleftheriou et al., 2012; Hayat et al., 2012; Silva, 2012; Anjum et al., 2014). Root growth is a combination of cell division and elongation. In this context, a decrease in mitotic activity has been reported in several plant species after exposure to heavy metals, which consequently results into a suppressed root growth (Fontes and Cox, 1998; Doncheva et al., 2005; Sundaramoorthy et al., 2010; Hossain et al., 2012a,b; Thounaojam et al., 2012). A study by Liu et al. (1992) showed that Cr(VI) has greater toxic effect on cell division than Cr(III). Furthermore, Sundaramoorthy et al. (2010) have also observed that Cr(VI) caused an extension in cell cycle that leads to the inhibition in cell division, thereby reducing root growth.
Pena et al. (2012) have reported that Cd toxicity affects the cell cycle G1/S transition and progression through S phase via decreased expression of a cyclin-dependent kinase (CDK), suggesting that ROS might be involved in such alterations. Yuan et al. (2013) have reported that excess Cu affects both elongation and meristem zones by altering auxin distribution through PINFORMED1 (PIN1) protein, and that Cu-mediated auxin redistribution is responsible for Cu-mediated inhibition of primary root elongation. Similarly, Petö et al. (2011) have also demonstrated that excess Cu inhibits root length and alters morphology by inducing alterations in auxin levels, which antagonizes nitric oxide function. It has also been demonstrated that inhibition in root growth is accompanied by an increase in root diameter, suggesting that plant cytoskeleton might also be a target of heavy metal toxicity (Zobel et al., 2007). Therefore, these studies suggest that heavy metals might cause an inhibition in root growth that alters water balance and nutrient absorption, thereby affecting their transportation to the aboveground plant parts and thus negatively affecting shoot growth and ultimately decreasing biomass accumulation. Roots utilize several mechanisms such as synthesis and deposition of callose to reduce and/or avoid heavy metal toxicity. These mechanisms create a barrier for the entry of heavy metals and enhance plasticity of root anatomy. Apart from barricading the entry of heavy metals, roots also allow their transportation to aboveground plant parts (in the case of metallophytes or hyperaccumulator plants: plants that can grow in heavy metal-contaminated soil; Fahr et al., 2013) for sequestration into the vacuoles rendering them inactive, and thus non-reactive.
Plasma membranes serve as a highly regulated checkpoint for an entry of unwanted substances inside the cell and protect the cell from negative consequences of many stressors. It has been reported that Arabidopsis halleri and Arabidopsis arenosa were more tolerant to heavy metal stress than Arabidopsis thaliana due to the lowest membrane depolarization, indicating that rapid membrane voltage changes might be an excellent tool for monitoring the effects of heavy metal toxicity (Kenderešová et al., 2012). Once inside the cell, heavy metals alter metabolism that results into a reduction of growth and lower biomass accumulation (Nagajyoti et al., 2010). Heavy metal toxicity might also cause stunted stem and root length, and chlorosis in younger leaves that can extend to the older leaves after prolonged exposure (Israr et al., 2006; Guo et al., 2008a,b; Warne et al., 2008; Gangwar and Singh, 2011; Gangwar et al., 2011; Srivastava et al., 2012). At the cellular and molecular levels, heavy metal toxicity affects plants in many ways. For instance, it alters the key physiological and biochemical processes such as seed germination, pigment synthesis, photosynthesis, gas exchanges, respiration, inactivation and denaturation of enzymes, blocks functional groups of metabolically important molecules, hormonal balance, nutrient assimilation, protein synthesis, and DNA replication (Nagajyoti et al., 2010; Yadav, 2010; Keunen et al., 2011; He et al., 2012; Hossain et al., 2012a,b; Silva, 2012; Wani et al., 2012; Singh et al., 2013). Under Cd stress, severe deleterious effects on various photosynthetic indices such as photosynthetic rate (Pn) and intracellular CO2 concentration (Ci) have been reported in tomato seedlings (Dong et al., 2005). Maleva et al. (2012) have observed that Mn, Cu, Cd, Zn, and Ni caused a significant decline in the levels of chlorophyll contents, accompanied by a decrease in the photochemical efficiency of photosystem II (PS II) in Elodea densa. Similarly, Li et al. (2012) have also reported that Cu, Zn, Pb, and Cd depressed chlorophyll and carotenoids levels and the quantum yield of PS II in Thalassia hemprichii, indicating that heavy metals have negative consequences on photosynthesis. Apart from affecting light reactions, heavy metals decrease CO2 assimilation by either diminishing RUBP carboxylase activity or by reacting with the thiol group of RUBISCO. For instance, Zn has been reported to inhibit RUBISCO activity in Phaseolus vulgaris by replacing Zn+2 for Mg+2, as both are bivalent cations (Monnet et al., 2001). In another study on Erythrina variegate by Muthuchelian et al. (2001), decreased RUBISCO activity was observed under Cd stress, and this decrease in RUBISCO activity might be due to the formation of mercaptide by Cd with thiol group of RUBISCO (Siborova, 1988). These researchers also reported decreased CO2 fixation, which is possibly due to a decrease in ATP and reductant pool (Husaini and Rai, 1991), as Cd ions decrease the proton source for reduction reactions (Ferretti et al., 1993). Similarly, Cu, a well-known inhibitor of carboxylase and oxygenase activities of RUBISCO (Lidon and Henriques, 1991), was found to decrease RUBISCO activity in Chenopodium rubrum (Schafer et al., 1992) by interacting with the essential cysteine residue of the enzyme (Siborova, 1988). Such reduction in pigments, photosynthetic rate, quantum yield of PS II, gas exchange, stomatal conductance, and CO2 assimilation might be linked to the ultrastructural changes (changes at cellular and tissue levels) induced by heavy metal stress. The effects arising due to changes in ultrastructures of membranes have been reported in several studies (Azzarello et al., 2012; Basile et al., 2012; Esposito et al., 2012; He et al., 2012; Sánchez-Pardo et al., 2012; Ali et al., 2013a,b). Moreover, heavy metals have been reported to affect another key physiological process, i.e., nitrogen metabolism, which is involved in plant function, from metabolism to allocation of resources, thereby regulating plant growth and development. Heavy metals have been found to enhance protease activity (Chaffei et al., 2003), and thus, reducing the activity of enzymes involved in nitrate (Nitrate reductase; NR and Nitrite reductase; NiR) and ammonia (Glutamine synthetase; GS, Glutamine oxoglutarate aminotransferase; GOGAT and Glutamate dehydrogenase; GDH) assimilation. The heavy metal Cd has been reported to affect nitrogen metabolism by inhibiting nitrate uptake and transportation, nitrate reductase, and GS activity (Hernández et al., 1997; Lea and Miflin, 2004), thereby affecting primary N assimilation processes.
Heavy metal-mediated alteration in hormonal balance correlates with their toxicities in plants (Petö et al., 2011; Wilkinson et al., 2012). For instance, in Brassica juncea, As causes toxicity by changing the levels of the auxins:indole-3-acetic acid (IAA), indole-3- butyric acid (IBA), and naphthalene acetic acid (NAA) and altering the expression of about 69 microRNAs (Srivastava et al., 2013). However, exogenous supply of IAA improves the growth of B. juncea under As stress, suggesting an implication of the regulation of the hormone level in the management of As stress.
Metallophytes under heavy metal stress
Metallophytes, also known as hyperaccumulators, have the ability to uptake large amounts of heavy metals from the soil, and this property makes them unique to be utilized in technologies such as biogeochemical and biogeobotanical prospection and phytoremediation. The absorbed heavy metals from the soil by these hyperaccumulators are not retained in the roots but are translocated to the shoots and accumulated in the aboveground organs at concentrations 100–1000-fold higher than the observed in non-hyperaccumulating species (Figures 2ia,b). However, this high concentration does not pose any toxic effect on plants (Rascio, 1997; Reeves, 2006; Prasad et al., 2010). With significant advances in our understanding of the mechanisms adopted by hyperaccumulators, there has been implication of three hallmarks that distinguish them from non-hyperaccumultors. These are (i) greater capability of heavy metal uptake, (ii) root-to-shoot translocation of heavy metal, and (iii) detoxification and sequestration of heavy metal (Figures 2iia–c). Studies on Thlaspi caerulescens and A. halleri, model plants for studying heavy metal tolerance strategies, have been done (Milner and Kochian, 2008; Singh et al., 2009; Frérot et al., 2010; Krämer, 2010). The studies have revealed that hyperaccumulation is not due to the presence of a novel gene, but it arises only from differential expression of genes that are common to hyperaccumulators and non-hyperaccumulators (Verbruggen et al., 2009). Hyperaccumulation of heavy metal includes three complex phenomena discussed below:
Figure 2
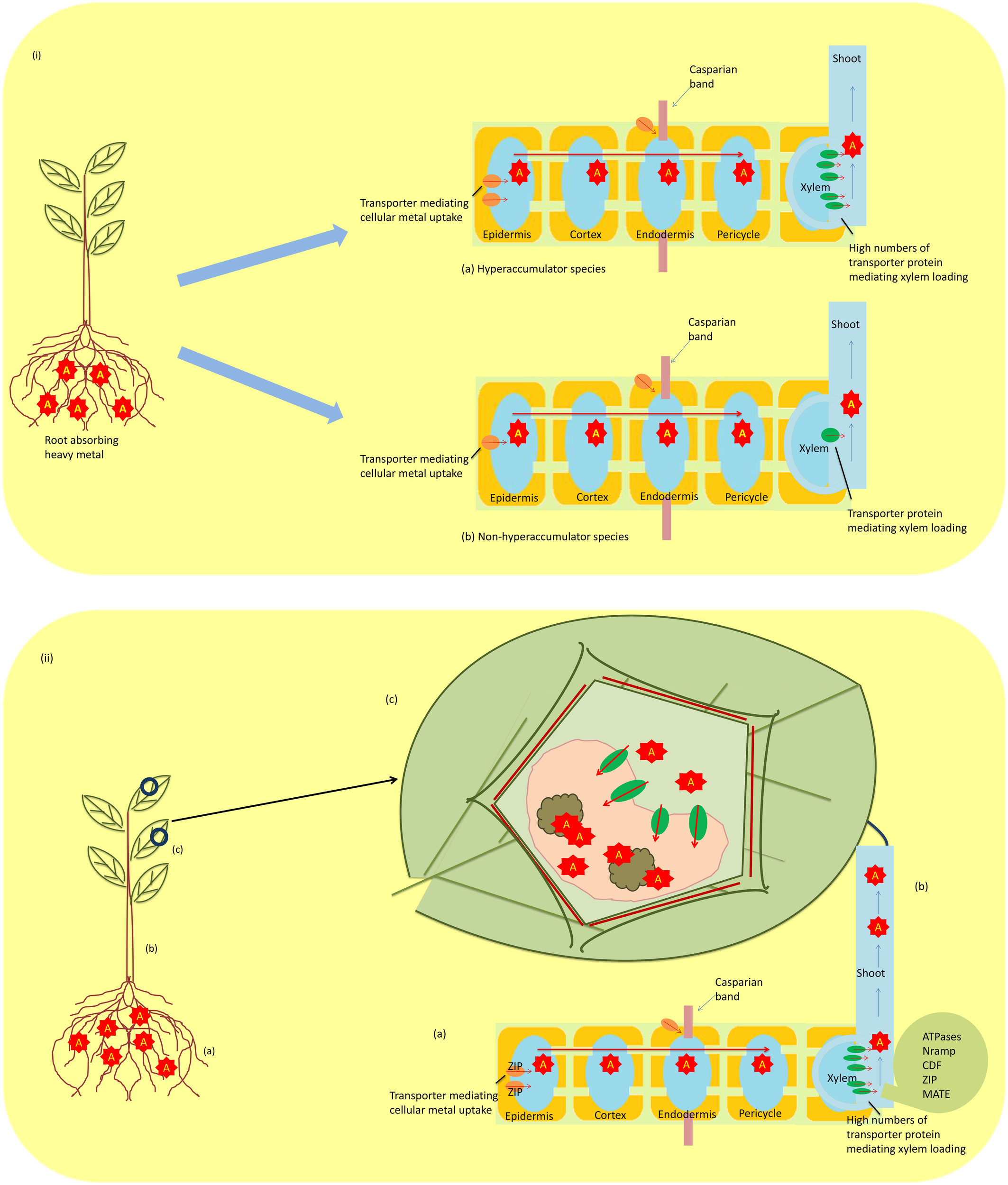
(i) A schematic diagram showing differential expression of constitutive gene in regulating transporters in hyperaccumulating (a) and non-hyperaccumulating (b) species (A; heavy metal). (ii) Mechanism of metal tolerance in hyperaccumulators (a) metal uptake by roots; ZIP (zinc-regulated transporter iron-regulated transporter proteins) (b) translocation of metal from root to shoot; ATPases (or CPx-type, P1B-type), Nramp (natural resistance-associated macrophage protein), CDF (cation diffusion facilitator family proteins), zinc–iron permease (ZIP) family proteins, MATE (Multidrug and Toxin Efflux) family, and (c) sequestration of metal in vacuole. A, metal; B, organic acid.
Heavy metal uptake
Hyperaccumulators have an extraordinary ability to absorb heavy metals from the soil under varying concentration of heavy metals (Ma et al., 2001; Yang et al., 2002). Although heavy metals are taken up by hyperaccumulators, their uptake is affected by several factors such as pH, water content, organic substances, etc. Moreover, heavy metal uptake requires a suitable transporting system to enter the plant (Figure 2i). Several researchers have reported that pH affects (i) proton secretion by roots that further acidify rhizosphere, thus enhancing metal dissolution, and (ii) the growth of metal-accumulating plant species (Bernal et al., 1994; Peng et al., 2005; Kuriakose and Prasad, 2008). Apart from pH, organic substances released from the roots affect growth in hyperaccumulating plants. Krishnamurti et al. (1997) have reported that organic acids released influence Cd solubility by forming Cd complexes. Therefore, pH and organic substances released from the rhizosphere of a hyperaccumulator mobilize heavy metal and enhance absorption (Krishnamurti et al., 1997; Peng et al., 2005). High uptake of heavy metal has also been associated with enhanced root proliferation (Whiting et al., 2000). Furthermore, constitutive overexpression of genes also attributes to enhanced heavy metal uptake. To pinpoint the genes involved in overexpression, several comparative studies have been performed in hyperaccumulating Arabidopsis halleri and Thlapsi caerulescens with that of congener non-hyperaccumulating species. Studies on T. caerulescens and A. halleri have revealed that increased Zn uptake is due to overexpression of genes belonging to the ZIP (Zinc-regulated transporter Iron-regulated transporter proteins) family encoding plasma membrane located transporters (Assunção et al., 2001): ZTN1 and ZTN2 in T. caerulescens and ZIP6 and ZIP9 in A. halleri. The decreased uptake of Cd under increasing Zn concentration was noticed in both genera, and it clearly demonstrated that expression of ZIP genes is Zn regulated (Assunção et al., 2010) and Cd influx is mainly due to Zn transporters having strong preference for Zn over Cd (Weber et al., 2006). Evidence exists that As being a chemical analog of phosphate enters the plant cell via phosphate transporters (Meharg and Hartley-Whitaker, 2002; Kanoun-Boulé et al., 2009). Similarly, a study on the As hyperaccumulator Pteris vittata and non-hyperaccumulator Pteris tremula has shown that plasma membranes of root cells of P. vittata had high density of phosphate/arsenate transporters than P. tremula (Caille et al., 2005), possibly due to constitutive gene overexpression. In addition, a study on the Se hyperaccumulators Astragalus bisulcatus (Fabaceae) and Stanleya pinnata (Brassicaceae) revealed that there was a higher Se/S ratio in the shoots of these species in comparison to the non-hyperaccumulator sister species, and this observation also supported the fact that an enhanced Se uptake was through sulfate transporters (Galeas et al., 2007).
Root-to-shoot translocation of heavy metals
Unlike non-hyperaccumulator plants, hyperaccumulators do not retain the heavy metal absorbed from roots but translocate them into shoots via xylem and several classes of proteins are involved in this translocation. The proteins involved are heavy metal-transporting ATPases (or CPx-type, P1B-type), natural resistance-associated macrophage proteins (Nramp), cation diffusion facilitator (CDF) family proteins, zinc–iron permease (ZIP) family proteins, and MATE (Multidrug And Toxin Efflux) protein family. The CPx-type ATPases are involved in transporting toxic metals like Cu, Zn, Cd, and Pb using ATP across cell membranes (Williams et al., 2000). The P1B-type ATPases also have the similar role of transporting heavy metal, but they also regulate metal homeostasis as well as tolerance (Axelsen and Palmgren, 1998). These heavy metal ATPases (HMAs) overexpressed in roots and shoots of hyperaccumulators suggest their upregulation in hyperaccumulators in comparison to non-hyperaccumulators (Papoyan and Kochian, 2004). Nramp is another class of protein family that has been found to be involved in transporting heavy metal ions, and genes coding for these proteins are termed as Nramp genes. Studies on rice revealed three homologs of this protein, namely OsNramp1, OsNramp2, and OsNramp3, and these proteins expressed in different tissues of rice transport distinct but related ions (Belouchi et al., 1997). Another class of proteins (CDF) have been found to be involved in transporting Zn, Co, and Cd, and regulate effluxing of cation out of the cytoplasmic compartment, and therefore, they are termed as “cation efflux transporters” (Mäser et al., 2001). A related Zn transporter (ZNT1) from T. caerulescens was reported by Pence et al. (2000), which belongs to a superfamily known as ZIP gene family, and was found to be expressed at high levels in roots and shoots. Another Zn transporter ZAT1 was also reported in Arabidopsis that was highly expressed in root tissues (van der Zaal et al., 1999). The transporter protein MATE is also involved in heavy metal translocation; FDR3, a protein of this family, was found to be expressed in roots of T. caerulescens and A. halleri, and the gene encoding this protein FDR3 plays a role in translocation of heavy metal (Talke et al., 2006; van de Mortel et al., 2006; Krämer et al., 2007). Therefore, the abovementioned studies provide strong evidence that multiple transporter proteins are involved in the translocation of heavy metal.
Detoxification/sequestration of heavy metal
After translocating, hyperaccumulators sequestrate and then detoxify the heavy metal, a process that allows them to survive under metal-contaminated areas without suffering from any toxic effect (Figure 2ii). The process of detoxification/sequestration occurs in the vacuole of plants (Vögeli-Lange and Wagner, 1990; Kanoun-Boulé et al., 2009; Singh et al., 2011a) and several transporter families are involved in this process, namelyABC, CDF, HMA, and NRAMP transporters. The ABC transporters are involved in transporting heavy metal into the vacuole and mainly two subfamilies (MRP and PDR) are active. The HMT1, first vacuolar ABC transporter reported in Schizosaccharomyces pombe, localized in the tonoplast aids in transporting PC–Cd (phytochelatins–cadmium) complexes formed in the cytosol (Ortiz et al., 1992, 1995; Kuriakose and Prasad, 2008). Later on, a functional homolog of HMT1 has been reported in Caenorhabditis elegans and Drosophila (Vatamaniuk et al., 2005; Sooksa-Nguan et al., 2009); however, no such homolog was studied in plants. Studies in A. thaliana have revealed two transporters AtMRP1 and AtMRP2 in transporting PC–Cd complexes into the vacuole (Lu et al., 1997, 1998), and these transporters confer the metal tolerance. The CDF transporter family, also named “metal tolerance protein (MTP),” is also involved in transporting metal cations such as Zn2+, Cd2+, Co2+, Ni2+, or Mn2+ from the cytosol to the vacuole (Krämer et al., 2007; Montanini et al., 2007). They have been categorized into two of four distinct groups of which groups I and III are the most important (Blaudez et al., 2003). Comparative studies in A. halleri and T. caerulescens with those of non-hyperaccumulators have shown higher expression of MTP1 (group III), MTP8 (group I), and MTP11 (group I) (Becher et al., 2004; Talke et al., 2006; van de Mortel et al., 2006). Similarly, AhMTP1 protein also showed a constitutive higher expression in leaves of A. halleri under exogenous supply of Zn (Dräger et al., 2004). The MTP11 and MTP8 were found to be close homologs of ShMTP8 (formerly ShMTP1) and confirmed Mn tolerance in A. thaliana (Delhaize et al., 2003), thus suggesting a role of these proteins in metal tolerance. Likewise, other transporter proteins such as HMA and NRAMP are also involved in transporting the metal from the cytosol to the vacuole. However, HMAs are thought to be involved in detoxification mechanisms due to their overexpression, as reported in A. thaliana (Morel et al., 2009).
Apart from the role of transporter proteins, organic acids are also involved in detoxification mechanisms, as they help in entrapping the metal ion and chelating them. For instance, citrate binds with Ni in leaves of Thlaspi goesingense, enabling formation of metal–organic acid complex for chelation (Krämer et al., 2000). Similarly, malate binds with Zn in A. halleri and Cd in T. caerulescens (Salt et al., 1999; Sarret et al., 2002). The role of amino acids in hyperaccumulator has been found to be important due to the formation of stable complexes with bivalent cations (Callahan et al., 2006), thus helping largely in sequestrating metal cations. For example, histidine (His) is involved in Ni hyperaccumulation, and a high concentration of His has been reported in the roots of Ni hyperaccumulators (Assuncão et al., 2003). The mechanism of heavy metal detoxification in hyperaccumulators also relies on the overexpression of genes related with antioxidant activity such as reduced glutathione (GSH), cysteine and O-acetylserine (Anjum et al., 2014). Studies have revealed that upstream signaling of salicylic acid results in increased serine acetyltransferase (SAT) activity and higher GSH level (Freeman et al., 2005a). Similarly, overexpression of NgSAT in Noccaea goesingense resulted in enhanced levels of GSH that resulted in Ni, Co, Zn, and to a small extent Cd tolerance (Freeman et al., 2004; Freeman and Salt, 2007).
Heavy metal and reactive oxygen species (ROS) production
Another negative consequence of heavy metal accumulation is the generation of ROS. In plants, ROS accumulation depends upon the balance between ROS production and ROS scavenging (Mittler et al., 2004), which in turn also depends on growth conditions such as temperature, light intensity, presence of heavy metal, etc. For instance, the presence of excess heavy metals results into a limitation of CO2 fixation in the chloroplasts, which coupled with an over reduction of the photosynthetic electron transport chain serves as a major site of ROS production (Mittler et al., 2004). Over reduction of the electron transport chains in the mitochondria is also a major site of ROS generation (Davidson and Schiestl, 2001; Keunen et al., 2011). Møller et al. (2007) reported that 1–5% of O2 consumed by the isolated mitochondria converts into ROS. ROS hydrogen peroxide (H2O2) is produced in the peroxisomes after glycolate is oxidized to glyoxylic acid during photorespiration (Mittler et al., 2004). Therefore, ROS such as singlet oxygen (1O2), superoxide anion (), H2O2, and hydroxyl radicals (∙OH) are produced in these organelles because of spin inversion and one-two and three-electron transfer reactions to O2, respectively, during functioning of the electron transport chains (Sharma and Dietz, 2009). The redox active heavy metals such as Cu, Cd Fe, and Zn can induce ROS formation directly by participating in Haber–Weiss and Fenton reactions or indirectly by inhibiting the functioning of enzymes in the cellular antioxidant defense network (Schützendübel and Polle, 2002; Halliwell, 2006; Keunen et al., 2011).
ROS are unstable, highly reactive, and thus, promptly react with other macromolecules to generate more free radicals because unpaired electrons tend to pair and give rise to two stable electron bonds (Foyer and Halliwell, 1976). Being extremely reactive in nature, ROS can interact with macromolecules such as DNA, pigments, proteins, lipids, and other essential cellular molecules depending on the properties like chemical reactivity, redox potential, half-life, and mobility within the cellular system, ultimately leading to a series of destructive processes collectively termed as “oxidative stress” (Mittler, 2002; Sharma and Dietz, 2009; Hossain et al., 2012a,b). Among ROS, ∙OH is the most reactive, highly damaging, and short-lived (1 ns), and can oxidize macromolecules within a diffusion distance. Therefore, ROS might induce reversible as well as irreversible modifications in lipids, proteins, and nucleic acids; however, most of these ROS effects are damaging and irreversible.
Conversely, ROS also act as signaling molecules involved in the regulation of many key physiological processes such as root hair growth, stomatal movement, cell growth, and cell differentiation when finely tuned and regulated by an antioxidative defense system (Foreman et al., 2003; Kwak et al., 2006; Tsukagoshi et al., 2010). It has been shown in several studies that ROS generated by NADPH oxidases during stress are channeled by the plant to serve as a stress signal to activate acclimation and defense mechanisms, which in turn counteract oxidative stress (Mittler et al., 2004; Davletova et al., 2005; Miller et al., 2008, 2010). Therefore, the fate of ROS (i.e., whether it will act as signaling molecule or damaging one) in the cellular system depends upon the output of many complex processes that involve in antioxidative system, signaling cascades, redox alterations, etc. When the generation of ROS exceeds that of the scavenging potential of antioxidants, oxidative stress occurs (Figure 3).
Figure 3
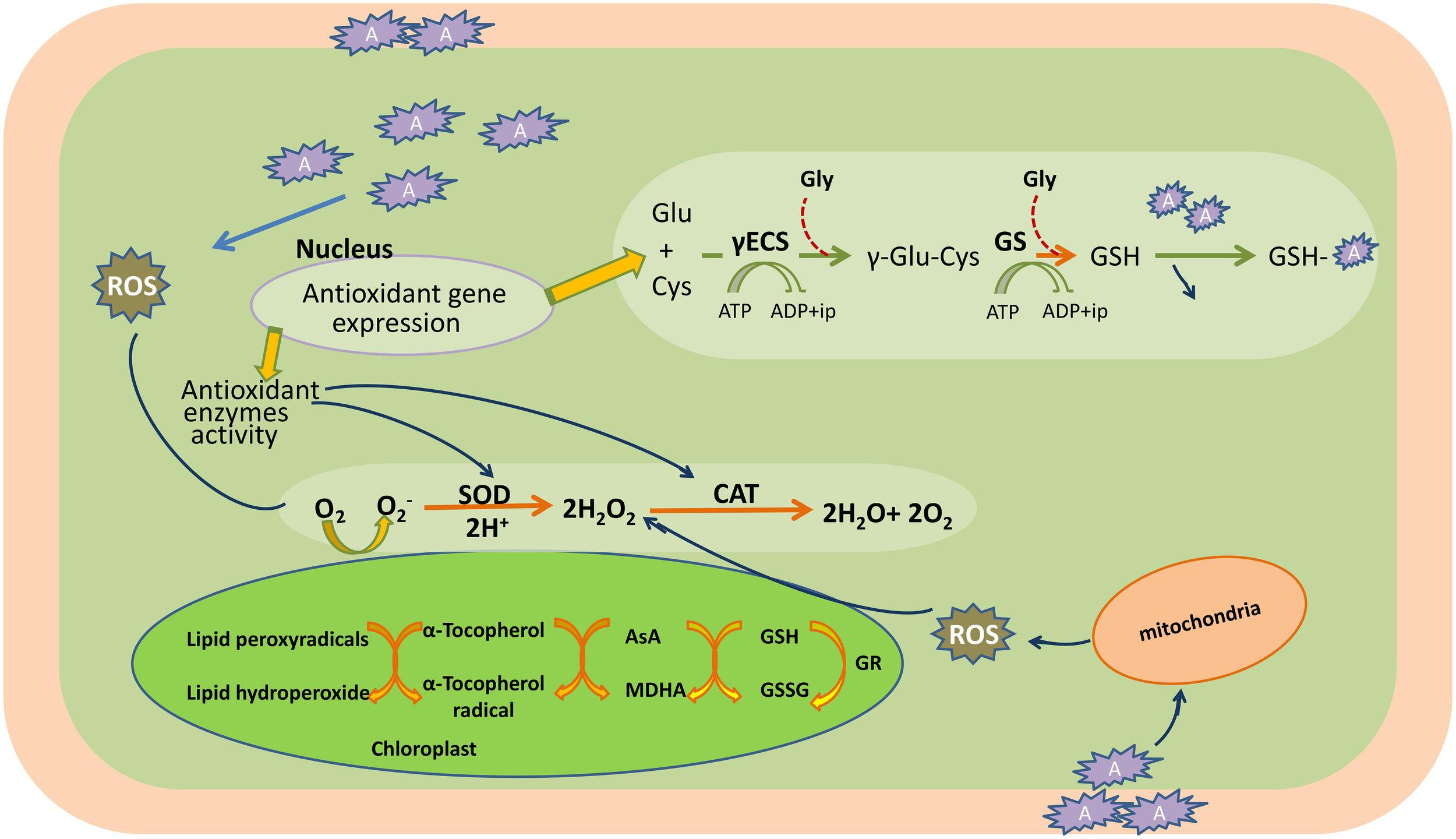
Heavy metal induced-oxidative stress, tolerance, and detoxification mechanisms in the plant cell. AsA, ascorbic acid; CAT, catalase; Cys, cysteine; c-ECS, c-glutamylcysteinesynthetase; Glu, glutamine; Gly, glycine; GR, glutathione reductase; GS, glutathione synthetase; GSH, glutathione (reduced); GSSG, oxidized glutathione; H2O2, hydrogen peroxide; MDHA, monodehydroascorbate; O2, oxygen molecule; , superoxide radicals; ROS, reactive oxygen species; SOD, superoxide dismutase; A, heavy metal.
Plants possess a specific mechanism to keep the routinely formed ROS at physiological limit, preventing them from exceeding toxic threshold levels, thereby playing an important role in the acclimation process against an imposed stress (Mittler et al., 2004). This mechanism is known as the plant antioxidant defense system that regulates ROS levels in the cellular system at a particular time (Figure 3). An antioxidant system comprises two types of components: enzymatic and non-enzymatic. The enzymatic antioxidants include superoxide dismutase (SOD; EC 1.15.1.1), catalase (CAT; EC 1.11.1.6), ascorbate peroxidase (APX; EC 1.11.1.11), glutathione reductase (GR; EC 1.6.4.2), monodehydroascorbate reductase (MDHAR; EC 1.6.5.4), dehydroascorbate reductase (DHAR; EC 1.8.5.1), glutathione peroxidase (GPX; EC 1.11.1.9), and glutathione-S-transferase (GST; EC 2.5.1.18), whereas non-enzymatic antioxidants include water-soluble compounds such as ascorbate, glutathione, proline, and α-tocopherol (Apel and Hirt, 2004; Sharma and Dietz, 2009; Hossain et al., 2011, 2012a,b). Although ascorbate and glutathione both function as cofactors of enzymatic antioxidants, both can also directly quench ROS and regulate the gene expression associated with biotic and abiotic stress responses (Hossain et al., 2012a,b). The importance of antioxidants is based on the facts that their increased and/or decreased levels are generally related to an enhanced or declined stress tolerance of stressed plants. Since the evolution of O2, antioxidants play an important role in sustaining ROS concentration at an appropriate level that can promote plant development and reinforce resistance to stressors by modulating the expression of a set of genes and redox signaling pathways (Neill et al., 2002). Keeping into consideration the importance of antioxidants in managing ROS levels and oxidative stress, the responses of transgenic plants and/or organisms overexpressing antioxidant and/or its biosynthetic pathway gene(s) against heavy metal stress are listed in Table 1.
Table 1
| Antioxidant and/or its biosynthetic pathway gene(s) | Source | Target transgenic | Response of transgenic plants and/or organisms | References |
|---|---|---|---|---|
| CAT3 | Brassica juncea | Nicotiana tabacum | Cd stress tolerance, better seedling growth, and longer roots | Gichner et al., 2004 |
| CAT | Brassica juncea | Nicotiana tabacum | Zn and Cd stress tolerance, 2.0-fold higher CAT activity than wild type, lower H2O2 level, and cell death | Guan et al., 2009 |
| CAT1 and CAT2 | Brassica oleracea | Arabidopsis | Low level of H2O2 and enhanced stress tolerance | Chiang et al., 2013 |
| Cu/ZnSOD and/or CAT | Zea mays | Brassica campestris | Less reduction in photosynthetic activity than wild type under SO2 stress | Tseng et al., 2007 |
| MnSOD | Triticum aestivum | Brassica napus | SOD activity was 1.5–2.5-fold greater than wild type and enhanced Al tolerance | Basu et al., 2001 |
| Cu/ZnSOD and APX | – | Festuca arundinacea | Increased tolerance against Cu, Cd, and As due to depressed oxidative stress | Lee et al., 2007 |
| cytGR/cpGR | Bacterial | Brassica juncea | cpGR transgenic showed lower Cd accumulation and 50 times higher GR activity than wild type plants | Pilon-Smits et al., 2000 |
| GR | Brassica rapa | Escherichia coli | Increased tolerance against H2O2 induced by Cd, Zn, and Al due to an enhanced GR activity | Kim et al., 2009 |
| DHAR/GR/GST | Escherichia coli | Nicotiana tabacum | Overexpression enhanced metal tolerance due to maintained redox couples such as ascorbate and glutathione | Le Martret et al., 2011 |
| DHAR | Oryza sativa | Escherichia coli | DHAR-overexpressing E. coli strain was more tolerant to oxidant and metal-mediated stress conditions than the control E. coli strain | Shin et al., 2008 |
| MDHAR/DHAR | Arabidopsis | Nicotiana tabacum | DHAR but not MDHAR enhanced Al tolerance by maintaining ascorbate level | Yin et al., 2010 |
| GST | Trichoderma virens | Nicotiana tabacum | Enhanced Cd tolerance simultaneously no Cd accumulation, increased activity of SOD, CAT, GST, APX, and GPX than wild type | Dixit et al., 2011 |
| Sulfite oxidase (SO) | Zea mays | Nicotiana tabacum | Increased tolerance against S due to enhanced CAT-mediated H2O2 scavenging | Xia et al., 2012 |
| TcPCS1 | Thlaspi caerulescens | Saccharomyces cerevisiae and Nicotiana tabacum | Increased tolerance to Cd due to the decreased lipid peroxidation and enhanced activities of SOD, POD, and CAT | Liu et al., 2011 |
| Serine acetyltransferase | Thlaspi goesingense | Escherichia coli | Imparts Ni and Co tolerance due to involvement of glutathione | Freeman et al., 2005a |
| gshII | Escherichia coli | Brassica juncea | Transgenic plants had higher level of glutathione, phytochelatin, and thiols and thus showed enhanced Cd tolerance | Liang Zhu et al., 1999 |
| AsPCS1/GSH1 | Allium sativum/Saccharomyces cerevisiae | Arabidopsis | Elevated production of phytochelatin and glutathione that imparts Cd and As tolerance | Guo et al., 2008a |
| APS1 | Arabidopsis | Brassica juncea | Increased Se tolerance due to its rapid reduction | Pilon-Smits et al., 1999 |
| MTH1745 | Methanothermobacter thermoautotrophicum | Oryza sativa | Increased Hg tolerance, higher photosynthesis, SOD and POD activity, and lower superoxide radicals, H2O2, and lipid peroxidation than wild type | Chen et al., 2012b |
| PCs | Arabidopsis | Nicotiana tabacum | Enhanced Cd tolerance and hampers root-to-shoot Cd transport | Pomponi et al., 2006 |
| PCS1 | Arabidopsis | Arabidopsis | Enhanced As tolerance but increased Cd hypersensitivity | Li et al., 2004 |
| PCS | Anabaena sp. PCC 7120 | Escherichia coli | Enhanced tolerance against multiple stresses such as Cd and Cu by increasing phytochelatin production | Chaurasia et al., 2008 |
| MT1 | Mus musculus | Nicotiana tabacum | Enhanced Hg accumulation and tolerance | Ruiz et al., 2011 |
| MT1 | Paxillus involutus | Hebeloma cylindrosporum | Increased Cu and Cd tolerance | Bellion et al., 2007 |
| Δ1-pyrroline-5-carboxylate synthetase | Vigna aconitifolia | Chlamydomonas reinhardtii | Transgenic grows rapidly in toxic Cd concentration (100 μM), and bind four-fold more Cd than wild-type cells. Proline likely acts as an antioxidant in Cd-stressed cells and thus increases Cd tolerance | Siripornadulsil et al., 2002 |
| Alkyl hydroperoxide reductase | Anabaena sp. PCC 7120 | Escherichia coli | Enhanced tolerance against Cu and Cd by enhancing scavenging of H2O2 and reactive sulfur species | Mishra et al., 2009b |
Summary of transgenic plants over-expressing gene(s) of enzymatic and non-enzymatic antioxidants and their performance against heavy metal stress.
Plant antioxidant defense system
The term “antioxidant” refers to a class of compounds that protect cells from damage caused by exposure to certain highly reactive species like ROS. The network and coordination of antioxidants are solely responsible for removing, neutralizing, and scavenging ROS. SOD is an enzyme involved in dismutating superoxide radicals generated by oxidation of molecular oxygen into H2O2 and O2 in all the cellular compartments (Fridovich, 1989).
H2O2 produced by the action of SOD is quite dangerous as it can diffuse through the membrane very easily and damage other cellular components, and thus, metabolites (ascorbate and glutathione) and enzymes (monodehydroascorbate reductase; MDHAR, dehydroascorbate reductase; DHAR and glutathione reductase; GR) are implicated in scavenging of H2O2(Foyer et al., 1997). Three types of SODs have been reported in plants on the basis of the metal containing (1) the chloroplastic or cytosolic Cu–Zn SOD; the cytosolic Cu–Zn SOD is referred to as Cu–Zn SOD I, whereas the chloroplastic one is referred to as Cu–Zn SOD II; (2) Mitochondrial Mn SOD, and (3) the chloroplastic Fe SOD. APX is regarded as a housekeeping protein in the cytosol and chloroplast, and is involved in scavenging of H2O2. The substrate for this enzyme is ascorbate and the product, which is a radical, is reduced to dehydroascorbate by an enzyme MDHAR in the presence of an electron donor NADPH (Asada, 1992, 1996). CAT is an important oxidoreductase enzyme that catalyzes decomposition of H2O2 into H2O and O2, and it is found in most plants and is localized in the peroxisome. CAT is a key enzyme involved in detoxifying peroxides generated during photorespiration (Morita et al., 1994; Lin and Kao, 2000). Although APX and CAT serves the same function of detoxifying, different affinities (on the basis of Km values) of APX and CAT depict the role of APX in modulating H2O2 for signaling and CAT in detoxifying excess H2O2 during stress (Mittler, 2002).
The above mentioned enzymatic components play a relevant role in mitigating heavy metal stress. Several studies have revealed that treatment of heavy metal enhances ROS formation, and thus, substantial increase in the activities of SOD, CAT, and APX was observed (Bharwana et al., 2013; Bashri and Prasad, 2015). A study by Wang et al. (2004) revealed a considerable increase in the activities of POD, APX, and SOD under Cu stress in B. juncea seedlings. Similarly, Bharwana et al. (2013) showed that under Pb treatment, there was an appreciable rise in SOD, guaiacol peroxidase, APX and CAT activities, and their activities were further enhanced with the rising concentration of Pb from 50 to 100 μM. Similar to this, Singh et al. (2013) reported increased activity of SOD and CAT under As exposure (5 and 50 μM). These results suggest that cooperative action of antioxidants is required for a detoxification mechanism under heavy metal stress.
“OMICS” tools
Metabolomics
Metabolomics refers to the identification and quantification of all low-molecular weight metabolites required by the organisms during developmental stages (Arbona et al., 2013), and some metabolites have been reported to be involved under heavy metal stress tolerance strategies. In the following section, we discuss the role of metabolomics under heavy metal stress.
Amino acids and amines
Amino acids and their derivatives have been reported to chelate metal ions, thus conferring metal tolerance to plants. Amino acids, particularly proline and histidine, have been found to chelate metal ions in cells as well as in the xylem sap (Rai, 2002; Sharma and Dietz, 2006). Proline has been reported to accumulate under heavy metal stress (Talanova et al., 2000; Yusuf et al., 2012a). A study on microalgae has demonstrated an increased level of proline under Cd stress (Siripornadulsil et al., 2002). The mechanism of action of increased levels of proline is not sequestration, but it reduces the formation of free radicals and also maintains reducing environment by enhancing the level of GSH (Siripornadulsil et al., 2002). Histidine, another important amino acid, has been found to play an important role under heavy metal stress. Krämer et al. (1996) reported increased histidine levels in the xylem sap of Alyssum lesbiacum (Ni hyperaccumulator) under Ni stress. Similarly, Kerkeb and Krämer (2003) reported simultaneous uptake of Ni and Histidine in B. juncea. Changes in the histidine content have functional significance in metal stress tolerance (Sharma and Dietz, 2006). NA (aminocarboxylate), an amino acid derivative synthesized by condensation process of three S-adenosyl-L-methionine, has been also reported to chelate metal ions. They have been found to be involved in the movement of mineral nutrients (Stephan and Scholz, 1993). The physiological role of NA has been confirmed by studying the tomato mutant lacking NA synthase, an enzyme involved in catalyzing formation of NA, which showed accumulation of Fe and Cu (Scholz et al., 1985; Herbik et al., 1996). Apart from its chelating action, NA has been reported to be precursor of phytosiderophore mugineic acid involved in binding metals such as Zn, Cu, and Fe (Treeby et al., 1989).
Organic acids
Organic acids such as malate, citrate, and oxalate have been reported to transport metals through xylem and are involved in sequestrating ions in vacuole (Rauser, 1999). Citrate, synthesized from citrate synthase, has been shown to have high affinity for Fe, Ni, and Cd, but it is majorly involved in chelating Fe (Cataldo et al., 1988). Malate has been reported to chelate Zn and is mainly involved in chelating cytosolic ions (Mathys, 1977).
Glutathione and α-tocopherol
Glutathione (GSH) is a water-soluble tripeptide thiol having low molecular weight (c-Glu-Cys-Gly) and plays a role in the cellular defense against the toxic actions of heavy metals (Meister and Anderson, 1983). Glutathione reductase (GR) readily converts an oxidized glutathione (GSSG) form to reduced form of GSH. GR contains a conserved disulfide bridge that breaks off under metal stress (Creissen et al., 1992; Lee et al., 1998) and plays an important role in defense by reducing GSSG, thus allowing a high GSH/GSSG ratio to be maintained. Studies on Luffa seedlings showed an increasing trend in GR activity with an increasing concentration of As (Singh et al., 2015). GR-catalyzed reduction of glutathione disulfide (GSSG) to glutathione (GSH) is NADPH dependent, and to maintain the proper ratio of GSH/GSSG, GSH biosynthesis must be initiated with rapid reduction in GSSG by GR (Kumar et al., 2012).
Alpha-tocopherol is the most active form of vitamin E and is synthesized in the plastids of higher plants. It is found to be involved in scavenging ROS and lipid peroxides (Munne-Bosch, 2005) by quenching 1O2 in the chloroplast and thus, prevents cell membrane from damage under stress. Several studies have reported changes in the levels of α-tocopherol under heavy metal stress (Collin et al., 2008; Yusuf et al., 2010; Kumar et al., 2012; Lushchak and Semchuk, 2012). A study by Collin et al. (2008) reported an increased concentration of α-tocopherol in Arabidopsis under Cd treatment, and the authors suggested that there is an upregulation of genes related to its biosynthesis (Figure 3).
Phenols
Synthesis of phenolic compounds under heavy metal stress is due to their high tendency to chelate metals, which is due to the presence of hydroxyl and carboxyl groups that bind to metal ions particularly iron and copper (Jun et al., 2003). Winkel-Shirley (2002) reported induction of phenolic compounds in maize under aluminum exposure. Similarly, Diáz et al. (2001) reported accumulation of phenols in leaves of P. vulgaris when exposed to Cu stress. This increase in phenolics is correlated with increased activity of enzymes involved in biosynthesis of phenols under heavy metal stress. Phenols have been reported to be directly involved in chelating Fe ions and thus, suppressing Fenton's reaction, which is the important source of ROS production. Stimulation of CHS (Chalcone synthase) and PAL (phenylalanine ammonia-lyase) activities has been reported in several plants exposed to Cu, Cd, Al, Pb, and Ni (Babu et al., 2003; Sobkowiak and Deckert, 2006; Kováčik and Klejdus, 2008; Kováčik et al., 2009; Pawlak-Sprada et al., 2011). Lavid et al. (2001) reported that tea plants rich in tannin are tolerant to Mn and prevent from Mn toxicity by directly chelating the Mn.
Ionome and ionomics
Ionome includes the role of mineral nutrients, namely nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), sulfur (S) and magnesium (Mg) and trace metals namely iron (Fe), copper (Cu), manganese (Mn), molybdenum (Mo), cobalt (Co), and zinc (Zn) in alleviating heavy metal toxicity. Although all the mineral nutrients and trace elements are essential for growth and development processes of plants, concentration greater than the required level becomes toxic to the plants. Apart from posing toxicity at higher concentration, nutrients under safe limit play important role in alleviating toxicity induced by heavy metals.
Nitrogen is the most essential nutrient as it is the major constituent of proteins, nucleic acids, vitamins, and hormones. It has the potentiality of alleviating heavy metal toxicity, as it enhances the photosynthetic capacity by increasing chlorophyll synthesis, often synthesizes N-containing metabolites like proline, GSH, etc. and by enhancing the activity of antioxidant enzymes (Sharma and Dietz, 2006; Lin et al., 2011). In a study by Pankovic et al. (2000), it has been shown that supplementing 7.5 mM (optimal level) of N to sunflower reduced the inhibitory effect of Cd on photosynthesis by enhancing Rubisco activity and by increasing protein content. In another study by Zhu et al. (2011), it has been shown that supplementing N fertilizer in the form of 16 mM (NH4)2SO4, alleviated Cd-induced toxicity in Sedum. The alleviating potential not only depends on the supplemented level of N but also on the source of N. For instance, when N was applied in the form of -N, it reduced the Cd concentration in leaves of rice plants that was found to be below 100 mg kg−1 (Jalloh et al., 2009), but when supplemented as N, it increased the Cd concentration, which suggests antagonistic behavior of - while synergistic of toward Cd. Another mineral nutrient, phosphorus (P) is the major constituent of cell membrane and nucleic acid, and majorly required for phosphorylation reaction. It has also been reported in alleviating metal-induced toxicity either by diluting the metal or by decreasing the mobility of the metal by forming metal–phosphate complex (Sarwar et al., 2010). In addition, P can also increase GSH content and prevent membrane damage, thereby conferring tolerance to plants (Wang et al., 2009).
Potassium (K) ion is required by the plant to maintain anion–cation balance in cells and plays important regulatory role in protein synthesis and enzyme activation. By improving nutritional status of K, condition of oxidative stress in plants can be minimized (Shen et al., 2000). Supplementation of K at 60 mg kg−1 alleviated the toxicity induced by Cd at 25 mg kg−1 by increasing the content of AsA and GSH. Similar to nitrogen, K source may also play an important role in alleviating toxicity. A study by Zhao et al. (2004) clearly demonstrated that application of KNO3, K2SO4, and KCl at the rate of 55, 110, and 166 mg.K.kg−1, respectively, to the soil has differential effect on Cd (concentration 15 mg Kg−1) accumulation. When KCl and K2SO4 were applied in increasing concentration from 0 to 55 mg kg−1, there was 60–90% increase in Cd accumulation in shoots, whereas similar increasing concentrations of KNO3 increased the Cd content very marginally, suggesting its protective action against Cd stress.
Sulfur (S), another mineral nutrient, serves as an important constituent of several coenzymes, vitamins, and ferredoxin. Wangeline et al. (2004) reported that Cd toxicity could be alleviated by the upregulation of S-assimilation pathway, thus suggesting toward alleviating role of S under heavy metal toxicity. Studies on Triticum aestivum (Khan et al., 2007), B. juncea (Wangeline et al., 2004), and Arabidopsis (Howarth et al., 2003) have shown increased ATP-sulfurylase (ATPS) and serine acetyl transferase (SAT) activities under Cd stress, and thereby conferring tolerance to these plants. As ATPS activity helps in maintaining GSH level required for regulating Ascorbate (AsA)–GSH cycle (Khan et al., 2009), it has been reported that S at 40 mg.S.Kg−1 enhanced the AsA–GSH cycle, thereby reducing Cd-induced toxicity in mustard (Anjum et al., 2008). Thus, indicating toward the possibility that S supplementation to soil system might enhance the formation of S-containing defense compounds such as GSH and phytochelatins. Study by Astolfi et al. (2004) has shown that Cd (100 μM) exposure enhanced the ATPS, O-acetyl serine (OAS) thiol lyase activity, which is related to the production of phytochelatins that play the most effective detoxifying mechanism in plants (Zhang et al., 2010). Apart from enhancing the formation of phytochelatins, S also regulates ethylene signaling and thereby helping under heavy metal stress (Masood et al., 2012). Calcium (Ca) is majorly involved in activating the enzymes and also plays an important role in regulating metabolic activities. Due to chemical similarity as well as due to same channels and intracellular Ca-binding sites (Lauer Júnior et al., 2008) of Ca+2 and Cd+2, Cd present in external medium, replaces the Ca, and thereby affects the growth of plant. However, Ca has been shown to decrease the heavy metal-induced toxicity (Suzuki, 2005; Farzadfar et al., 2013). It has been reported that 30 mM Ca reduced the Cd content from 46.7 to 17.4 μg in Arabidopsis seedlings (Suzuki, 2005). Similar to this, Zhenyan et al. (2005) reported enhanced Cd (concentration 0.5 mM) tolerance in Lactuca sativa when supplied with 4 mM CaCl2, which was due to enhanced expression of phytochelatin synthase. Ca reduces heavy metal-induced toxicity by reducing their uptake, influencing physiological processes, or activating expressions of other defense compounds.
Magnesium (Mg), an important constituent for chlorophyll biosynthesis, plays an essential role under heavy metal toxicity. Abul Kashem and Kawai (2007) reported that Cd (0.25 μM) -induced toxicity in Japanese mustard spinach was alleviated by Mg at 10 mM, and Cd accumulation was reduced by 40%. Mg-induced alleviation is not due to inhibition in uptake but due to enhanced antioxidant enzymes (Chou et al., 2011). Moreover, Mg-induced alleviation has been also correlated with expression of some genes OsIRT1, OsZIP1, and OsZIP3 of rice.
Trace elements are required in lesser amount for the biological system, which include iron (Fe), copper (Cu), manganese (Mn), molybdenum (Mo), cobalt (Co), and zinc (Zn), and their high levels could be toxic. The essentialities of these trace metals are due to their active participation in the redox reactions as well as because of their roles as enzyme cofactors (Sanita di Toppi and Gabbrielli, 1999). However, apart from their roles in biological system, they have been reported to play a crucial role in alleviating metal toxicity. Several trace elements have direct as well as indirect effects on heavy metal availability and toxicity (Sarwar et al., 2010). Direct effects include lowered solubility of heavy metals in the soil (Hart et al., 2005; Shi et al., 2005; Matusik et al., 2008), competition between heavy metals and trace elements for the same membrane transporters (Baszynski et al., 1980; Qiu et al., 2005), and heavy metal sequestration in the vacuoles (Salt and Rauser, 1995; Zaccheo et al., 2006). Indirect effects include dilution of heavy metal concentration by increasing plant biomass (dilution effect) and alleviation of heavy metal stress by increasing antioxidant defense system (Hassan et al., 2005; Suzuki, 2005; Jalloh et al., 2009). Zn, being an important group of metal transporter family, has been suggested to prevent damage caused by Cd toxicity. As reported in the case of Thalpsi violacea, plants supplied with 2 mgL−1 Cd showed 48.5 mg Kg−1 Zn accumulation than that of control (16.8 mgKg−1), whereas when the plant was supplemented with 5 mgL−1 Cd, Zn accumulation decreased upto 12.8 mg Kg−1, suggesting Cd/Zn antagonism (Street et al., 2010). Furthermore, Zn also enhances the activities of antioxidant enzymes and competes with Cd to bind with the membrane protein in order to protect plant (Wu and Zhang, 2002). Other trace metal Fe, under Cd stress, showed reduced Cd uptake and translocation, thus increasing plant growth. Study by Qureshi et al. (2010) revealed that exogenous application of 40 μM Fe reduces the condition of oxidative stress by stabilizing the thylakoid complex under Cd stress. It was also reported that at Fe concentrations of 1.89 mg L−1 (moderate) and 16.8 mg L−1 (high), under low level of Cd (0.1 mM), plant height showed increment (Nada et al., 2007).
Similarly, studies have also revealed the protective roles of trace elements in ameliorating toxic effects of heavy metals by protecting photosynthetic tissue and increasing antioxidant capacity (Zornoza et al., 2010; Tkalec et al., 2014). Pa1'ove-Balang et al. (2006) have shown that Mn-mediated amelioration of Cd toxicity was associated with a decreased Cd uptake. Apart from the beneficial role played by trace metals, there are some beneficial nutrients like selenium (Se), and silicon (Si) that also play a major role under heavy metal stress. Earlier, selenium (Se) was considered as toxic element but later on Schwarz and Foltz (1957) confirmed it to be an essential one. Studies on lettuce grown under Pb and Cd toxicity supplemented with Se showed a decrease in heavy metal accumulation as well as enhanced uptake of essential nutrients (He et al., 2004). Belokobylsky et al. (2004) and Feng and Wei (2012) have found that Se level up to 5 mg L−1 has beneficial effects on Spirulina platensis and P. vittata, respectively. Filek et al. (2008) have shown that exogenous application of Se alleviates toxic effects of Cd by enhancing the activities of antioxidant enzymes such as SOD, CAT, GPX, and APX. Several reports have revealed that appropriate dose of Se can protect plants against damage by heavy metals such as Hg, Pb, Cd, Cr, and Sb (Khattak et al., 1991; Shanker et al., 1996; Belokobylsky et al., 2004; He et al., 2004; Feng et al., 2011). Role of Si under heavy metal stress is also well established (Singh et al., 2011b; Dragišić Maksimović et al., 2012; Tripathi et al., 2012). Study by Song et al. (2009) has shown that supplementation of Si under Cd stress decreased an uptake and root to shoot translocation of Cd as well as enhanced the activities of enzymes of the defense machinery in B. chinensis. Similarly, study by Bharwana et al. (2013) revealed that Si application reduces Pb uptake and enhances the activities of antioxidants viz., SOD, GPX, APX, and CAT.
The measurement of elemental composition and their changes as a response to some stimuli in living organisms comes under the study of ionomics. Alteration in ionome could be direct or indirect. Direct one includes the changes in nutrient level in soil or due to impairment of ion transporter, whereas indirect changes might be due to changes in cell wall structure (Salt et al., 2008). Heavy metals due to their interaction with nutrient elements affect the uptake and distribution of these elements and may result in deficiency of minerals thus affecting the growth. Sarwar et al. (2010) suggested that Cd affects the permeability of plasma membrane and thus interferes with the nutrient uptake. However, there exists both antagonistic as well as synergistic interaction between heavy metals and micronutrient uptake, which could be due to differences in plant species and nutrient concentration. Likewise, a study by Cataldo et al. (1983) reported antagonistic interaction between Cd and Fe, Zn, Cu, and Mn in soybean plants, whereas Nan et al. (2002) reported synergistic interaction between Cd and Zn in wheat and corn. In a study by Yang et al. (1998), decreased accumulation of Fe, Mn, and Cu in ryegrass, maize, cabbage, and white clover was observed after Cd exposure, whereas there was increased P accumulation. Similarly, Cui et al. (2008) reported decrease in Fe and Zn uptake in rice after Cd treatment in hydroponic system. A study by Safarzadeh et al. (2013) determined the effect of different doses (0, 45, and 90 mg kg soil) of Cd on uptake of Fe, Zn, Cu, and Mn in seven rice cultivar and reported decrease in Zn, Fe, Mn, and Cu uptake. Not only the uptake decreased but also there was decrease in the translocation of these minerals as Cu and Fe contents found to be greater in roots than in shoots that indicate toward impairment of ions transporters.
Similar to Cd, As has also been reported to influence nutrient uptake and their distribution in plants. Meharg and Hartley-Whitaker (2002) reported As-induced decrease in P uptake is due to chemical similarity between P and arsenate and due to which arsenate enters the plant via the phosphate transport systems. However, the concentration of As also plays an important role in P uptake. Burló et al. (1999) reported higher uptake of P at lower level of As in tomato plants. Similarly, Carbonell et al. (1998) reported increased P uptake in tomato plant when exposed to low level of As. As not only influences P uptake but also affects the uptake of other nutrients like N, P, K, Ca, etc. A study by Carbonell-Barrachina et al. (1997) observed increased concentration of N, P, K, Ca, and Mg in P. vulgaris L. plants when exposed to arsenite. Similarly, Carbonell-Barrachina et al. (1994) reported decreased uptake of K, Ca, and Mg (macronutrinets), B, Cu, Mn, and Zn (micronutrinets) in Lycopersicum esculentum Mill. The effect of As concentration on nutrient level of hyperaccumulator P. vittata L. had also been studied by Tu and Ma (2005), and the authors reported that both micro- and macronutrients were in the range of normal concentration as in non-hyperaccumulators. However, there was enhancement in P and K contents in the fronds of P. vittata L. at lower level of As. They reported molar ratio of P/As to be 1.0 in fronds of P. vittata L., which is the threshold value for normal growth of plants.
Heavy metal ions such as Cu+2, Zn+2, Mn+2, and Fe+2 are essential for plant metabolism but when they are present in excess amount become highly toxic. For instance, Zn and Mn when present in excess impairs growth and compete with Fe. Excess Fe in the plant system participates in the fenton reaction, thereby creating a condition of oxidative stress (Williams and Pittman, 2010; Shanmugam et al., 2011). In order to avoid toxicity induced by mineral elements and trace elements, these are chelated by low molecular weight compounds and sequestrated in vacuoles or excluded to extracellular spaces by transporters situated in the tonoplast or plasma membrane, which plays central role in maintaining metal homeostasis under safe limit. These transporters belong to (1) P1B-ATPase or CPx-type ATPase, (2) Cation Diffusion Facilitator (CDF) also known as Metal Tolerance Proteins (MTPs), (3) Natural Resistance-Associated Macrophage Proteins (NRAMPs), and (4) ZRT–IRT-like Protein (ZIP) transporters.
P1B-ATPases (also known as heavy metal ATPases: HMAs)
P1B-ATPases (also known as Heavy Metal ATPases: HMAs) are found in a wide range of organisms ranging from prokaryotes to eukaryotes including yeasts, insect, mammals, and plants. Being energized by ATP hydrolysis, they translocate heavy metals (Zn, Co, Cu, Cd, and Pb) out of cytoplasm (to plasma membrane and into vacuole) and thus play important roles in their transport, compartmentalization, and detoxification (Williams et al., 2000; Grennan, 2009). HMA members (HMA2, HMA3, and HMA4) export Zn and Cd. For instance, HMA4 protein that plays a role in nutrition and transport of Zn from root to shoot also protects plants from Cd via its efflux (Mills et al., 2005). Hussain et al. (2004) demonstrated that though HMA2 and HMA4 are essential for Zn homeostasis in Arabidopsis, double mutants of HMA2 and HMA4 (hma2 and hma4) exhibited increased sensitivity of plant to Cd, suggesting that they may also play a role in Cd detoxification. Similarly, loss of function in HMA2 and HMA4 has been shown to increase Cd sensitivity in Arabidopsis under phytochelatins deficient, cad1-3 as well as CAD1 backgrounds (Wong and Cobbett, 2009). A recent study on 349 wild varieties of A. thaliana with combined approach of genome-wide association mapping, linkage mapping, and transgenic complementation revealed that HMA3 is solely responsible for variation in amount of Cd accumulated. Varieties with high Cd accumulation indicate toward reduced HMA3 function (Chao et al., 2012). Similarly, Song et al. (2014) found expression of C-type ATP-binding cassette (ABC) transporter (OsABCC) family in Oryza sativa and reported its involvement in detoxifying and reducing As accumulation in grains. They reported higher expression of these transporters under higher level of As.
Cation diffusion facilitators (CDFs) or metal tolerance proteins (MTPs)
Cation Diffusion Facilitators (CDFs), also known as Metal Tolerance Proteins (MTPs) family, were first reported by Nies and Silver (1995) and found in diverse group of organisms such as bacteria, fungi, animals, and plants. Members of this family are involved in cellular heavy metals homeostasis with principal selectivity for Zn2+, Mn2+, and Fe2+ (Podar et al., 2012). Despite their specificities for Zn2+, Mn2+, and Fe2+, many CDFs may also transport other heavy metals such as Co2+, Ni2+, and Cd2+ (Ricachenevsky et al., 2013). CDFs transporters are involved in heavy metals efflux from the cytoplasm either to the extracellular space or into the organelles (Haney et al., 2005; Peiter et al., 2007; Ricachenevsky et al., 2013). Twelve MTP genes have been recognized so far in A. thaliana and 10 in O. sativa (Gustin et al., 2011). In A. thaliana, the first CDF gene was characterized as the Zinc Transporter 1 gene (ZAT1) and later renamed as METAL TOLERANCE PROTEIN 1 (AtMTP1) (van der Zaal et al., 1999; Delhaize et al., 2003). The AtMTP1 gene is expressed constitutively in roots as well as in shoots, and when overexpressed in Arabidopsis, it enhances Zn tolerance (van der Zaal et al., 1999). However, RNA interference (RNAi)-mediated silencing (Desbrosses-Fonrouge et al., 2005) or T-DNA insertion mutation (Kobae et al., 2004) of this gene increases Zn sensitivity, indicating its important role in regulation of Zn homeostasis. In A. halleri, a Zn hyperaccumulator plant, AhMTP1 gene is believed to have a role in Zn hypertolerance (Shahzad et al., 2010). Unlike AtMTP1 gene, AtMTP3 is expressed predominantly in roots and reported to be engaged in maintenance of Zn homeostasis by excluding Zn under Zn oversupply (Arrivault et al., 2006). Another member of MTPs family, AtMTP11, has been reported to transport as well as provide Mn tolerance (Delhaize et al., 2007; Peiter et al., 2007). In rice, an ortholog of MTPs, OsMTP1, has been characterized and supposed to be located on chromosome 5 and highly expressed in mature leaves and stem (Lan et al., 2012; Yuan et al., 2012).
Menguer et al. (2013) demonstrated that OsMTP1 gene localized on tonoplast, and when heterologously expressed in the yeast-mutant zrc1 and cot1, complemented Zn hypersensitivity. Besides, its expression also alleviated Co sensitivity, rescued Fe hypersensitivity of the ccc1 mutant, and restored growth of the Cd-hypersensitive mutant ycf1, indicating potential role of this gene in possible biotechnological applications, such as bio fortification and phytoremediation.
Natural resistance-associated macrophage proteins (NRAMPs) transporters
Nramp is a highly conserved family of integral membrane proteins that are conserved in different species and located in the plasma membrane of root apical cells (Simões et al., 2012). They are involved in proton-coupled active transport of various heavy metals(Fe2+, Zn2+, Mn2+, Co2+, Cd2+, Cu2+, Ni2+, and Pb2+) in wide range of organisms including bacteria, fungi, animals, and plants (Hall and Williams, 2003; Cailliatte et al., 2009). However, the physiological role of NRAMP was primarily related with Fe and to a lesser extent for Mn transport (Cailliatte et al., 2009). AtNRAMP1 and 6, forms the first group, and AtNRAMP2–5 constitute the second group (Mäser et al., 2001). Of these, AtNRAMP1, 3, 4, and 6 have been shown to encode functional plant heavy metal transporters (Krämer et al., 2007; Cailliatte et al., 2009). Yeast mutants defective in heavy metals uptake have been utilized to investigate transport specificities of plant Nramps. Study revealed that AtNRAMP1 can complement the fet3fet4 yeast mutant that is defective in both low and high-affinity Fe transporters, whereas overexpression of AtNRAMP1 in Arabidopsis increases plant resistance to toxic Fe concentrations (Curie et al., 2000). Furthermore, AtNRAMP3 and AtNRAMP4 have been shown to mediate the remobilization of Fe from the vacuolar store and are essential for seed germination under low Fe conditions (Thomine et al., 2003; Lanquar et al., 2005), indicating a role of Nramps in Fe homeostasis.
Since the roles of NRAMPs family proteins were previously related with Fe uptake and transport in biological systems, however, increasing numbers of studies indicated that members of this family have wide range of specificities for pumping cations inside and/or outside the cell. Cailliatte et al. (2010) demonstrated that NRAMP1, localized on plasma membrane, restores the capacity of the iron-regulated transporter1 (a ZIP family metal transporter) mutant to take up Fe and Co, indicating that NRAMP1 has a broad selectivity for heavy metals in-vivo. An AtNRAMP4 homolog, TjNRAMP4, was cloned from the Ni hyperaccumulator Thlaspi japonicum, and its expression increased Ni2+ sensitivity of wild-type yeast due to elevated Ni accumulation, indicating that this protein might transport Ni into the cytoplasm (Mizuno et al., 2005). Besides regulating uptake and distribution of essential heavy metals, Nramps have also been found to be involved in the transport of non-essential heavy metals. In Arabidopsis, AtNRAMP3 disruption increases Cd2+ resistance, whereas overexpression of this gene confers increased Cd2+ sensitivity, indicating that it plays a role in Cd2+ transport and sensitivity in plant (Thomine et al., 2000; Mäser et al., 2001).
A O. sativa Nrat1 (OsNrat1) gene, a Nramp aluminum transporter and localized at all cells of root tips, when expressed in yeast transports only Al3+ but not the Mn, Fe, and Cd, indicating that this transporter gene specifically transports only Al (Xia et al., 2011). Furthermore, in knockout of Nrat1, Al sensitivity increased, whereas in wild type, its expression is up-regulated by Al in root that is believed to be required for a prior step of final Al detoxification through sequestration of Al into vacuoles. Study of Cailliatte et al. (2009) demonstrated that Arabidopsis transgenic plants overexpressing AtNRAMP6 gene were hypersensitive to Cd, although plant Cd content remained unchanged, thereby indicating that modification in expression pattern of AtNRAMP6 affects distribution and availability of Cd within the cell. However, Sano et al. (2012) have shown that Nicotiana tabacum NRAMP1 gene (NtNRAMP1), a plasma membrane transporter, when overexpressed in tobacco BY-2 cells increases resistance of the cells to both Fe and Cd, and suggested that NtNRAMP1 moderates Fe-uptake and prevents toxicity resulting from excess Fe or Cd application. Tiwari et al. (2014) also demonstrated that OsNRAPM1, localized on plasma membrane of endodermis and pericycle cell, when expressed in Arabidopsis provides tolerance against As and Cd with their enhanced accumulation in root and shoot, and proposed that modification in this gene may be helpful in reducing the risk of food chain contamination by these toxic heavy metals. These studies clearly indicate that NRAMP genes are able to encode multi-specific heavy metals transporters. In recent years, a new Nramp5 belonging to rice (Os Nramp5) has been characterized, which is responsible for accumulation of Mn in rice and has been reported to encode proteins localized on plasma membrane, thus suggesting that Nramp5 is a major transporter responsible for transport of Mn and Cd (Sasaki et al., 2012). To gain deep insights into the roles of NRAMP genes transporter in heavy metals uptake and homeostasis in plants, a more systematic characterization of the different members of the NRAMP family is further required.
ZRT, IRT-like proteins (ZIP) transporters
Members of the ZIP family named on the first member identified ZRT IRT- like Protein in Arabidopsis, expressed in roots of iron deficient plants and found to be capable of transporting various heavy metals such as Fe, Zn, Mn, Cd and Ni within cellular systems (Mäser et al., 2001). The key feature of the ZIP family is that these proteins can transport heavy metals from the extracellular space or from organelles lumen into the cytoplasm. In Arabidopsis, 15 genes viz. ZIP1-12, IRT1, IRT2, and IRT3 of the ZIP family are reported (Milner et al., 2013). Among these members, AtIRT1, AtIRT2, and AtIRT3 transporters are well characterized, with AtIRT1 being the most studied (Eide et al., 1996; Lin et al., 2009; Vert et al., 2009) for their involvement in regulation of Zn and Fe homeostasis in plants. Rest of the ZIP family members has been studied for their membrane localization and heavy metals they transport into or outside of a specific organelle (Milner et al., 2013). In a model legume Medicago truncatula, six ZIP family transporters MtZIP1, MtZIP3, MtZIP4, MtZIP5, MtZIP6, and MtZIP7 have been tested for their ability to complement yeast heavy metals uptake mutants, and each family member was able to rescue the growth of Zn, Mn, and Fe uptake mutants, indicating their function in heavy metals transport (López-Millán et al., 2004).
Apart from the abovementioned transporters, recently another transporter arsenate reductase (ACR) has been characterized in yeast Saccharomyces cerevisiae, a model system for As resistance. It was reported that a 4.2-kb region conferred arsenite (AsIII) resistance in S. cerevisiae; they found three ACR genes, namely ACR1, ACR2, and ACR3 (Bobrowicz et al., 1997). These authors also reported that ACR1 regulates ACR2 and ACR3 by transcriptional factor and any loss in ACR1 function yeast conferred arsenite and arsenate hypersensitivities (Bobrowicz et al., 1997; Ghosh et al., 1999). Later on, Landrieu et al. (2004a,b) reported that ACR2 represents arsenate reductase that showed homology to yeast ASCR2 (ScACR2). Similarly, Ellis et al. (2006) reported other transporter PvACR2 from P. vittata and OsACR2.1 and OsACR2.3 from O. sativa (Duan et al., 2007). Earlier, ACR2 (called as CDC25) was thought to be involved in As metabolism in A. thaliana. Recent studies on A. thaliana have revealed the involvement of new arsenate reductase (ACR), namely HAC1 (Chao et al., 2014) or ATQ1 (Sánchez-Bermejo et al., 2014). Chao et al. (2014) reported that loss of function of HAC1 in A. thaliana resulted in decreased As accumulation in roots, and thus, there was diminished As efflux to external medium. Another transporter, OsABCC1 localized in phloem cells of O. sativa, has been reported to be involved in sequestration of As to vacuole (Song et al., 2014). However, in anaerobic paddy fields, As (mainly Arsenite) uptake is regulated by transporters of Si, namely Lsi1 (low silicon 1; influx transporter) and Lsi2 (low silicon 2; efflux transporters) (Ma et al., 2008). Apart from these transporters, there are some other transporters as well that transfer arsenate and arsenite. Likewise, a transporter from P. vittata, PvACR3 has been reported to compartmentalize As into the vacuoles and loss in its function results in As hypersensitivity (Indriolo et al., 2010).
Transcriptomics
Investigations on the basic mechanisms of heavy metal tolerance and adaptation are the area of great scientific interest and an intensive research. Various stressors induce an expression of a set of genes in plants (Nakashima et al., 2009).
At molecular level, the regulation of gene expression is very important for the biological processes, which determines the fate of plant development as well as tolerance to heavy metal stress. Stressors trigger large number of genes and several proteins in order to link the signaling pathways that confer stress tolerance (Umezawa et al., 2006; Valliyodan and Nguyen, 2006; Manavalan et al., 2009; Tran et al., 2010). These genes are classified into two groups: the regulatory genes and the functional genes (Tran et al., 2010). The genes of regulatory group encode various transcription factors (TFs), which can regulate various stress-responsive genes cooperatively and/or separately and thus, constitute a gene network. However, the genes of functional group encode metabolic compounds such as amines, alcohols, and sugars, which play a crucial role in heavy metal stress tolerance. The TFs, which are reported to be master regulators, control an expression of gene clusters and usually members of multigene families. Studies reveal that a single TF can control the expression of many target genes via specific binding of the TF to the cis-acting element in the promoters of its target genes (Wray et al., 2003; Nakashima et al., 2009). Most of the TFs contain a DNA-binding domain that interacts with cis-regulatory elements in the promoters of its target genes and via a protein–protein interaction domain that helps in oligomerization of TFs with other regulators (Wray et al., 2003; Shiu et al., 2005). This type of transcriptional regulatory system is referred as “regulon” (Nakashima et al., 2009). Various TFs families such as AREB/ABF, MYB, AP2/EREBP, WRKY, bHLH, bZIP, MYC, HSF, DREB1/CBF, NAC, HB, ARID, EMF1, CCAAT-HAP2, CCAAT-DR1, CCAAT-HAP3, CCAAT-HAP5, C2H2, C3H, C2C2-Dof, C2C2-YABBY, C2C2-CO-like, C2C2-Gata, E2F-DP, ABI3VP1, ARF, AtSR, CPP, E2F-DP, SBP, MADS, TUB, etc. are known to influence stress response in plants (Singh et al., 2002; Shiu et al., 2005; Shameer et al., 2009). LeDuc et al. (2006), in a transcriptome analysis on plants, reported that plants treated with heavy metals could induce transcription factors that regulate corresponding transcriptional processes.
Liang et al. (2013) reported first FER regulatory gene involved in Fe uptake in tomato, and the functional analog of FER is FER-like Deficiency Induced Transcripition Factor (FIT) that has been conferred to play an important role under Fe deficiency in Arabidopsis (Yuan et al., 2005). In addition to this, there are several other subgroups of bHLH family viz., AtbHLH38, AtbHLH39, AtbHLH100, and AtbHLH101 that have been shown to be upregulated under Fe deficiency in roots and leaves of Arabidopsis (Wang et al., 2007; Yuan et al., 2008). Later, several researchers proposed that AtbHLH38 or AtbHLH39 interacts with FIT and forms heterodimers and directly activates transcription factors for ferric chelate reductase and ferrous transporters, which are the two major genes regulating Fe uptake under deficient condition (Varotto et al., 2002; Vert et al., 2002; Yuan et al., 2008). In Arabidopsis, IRT1 has been reported to be the most essential ferrous transporter. Beside transporting Fe, it can also transport Zn, Mn, Co, Ni, and Cd, and thus, these metals get accumulated under Fe deficiency (Vert et al., 2002; Schaaf et al., 2006). A recent study by Wu et al. (2012) in Arabidopsis revealed that expression of FIT with AtbHLH38 or AtbHLH39 further activates expression of several other transporters viz., HMA3, (MTP3), Iron Regulated Transporter2 (IRT2) that play regulatory role in maintaining Fe content under Cd exposure.
Transcriptome analysis in A. thaliana and B. juncea exposed to Cd stress has revealed the induction of basic region leucine zipper (bZIP) and zinc finger transcription factors (Ramos et al., 2007). ERF1 and ERF5, two transcription factors belonging to AP2/ERF superfamily (characterized by AP2/ERF domain; Nakano et al., 2006), have been reported to be induced when A. thaliana was exposed to Cd (Herbette et al., 2006). Similar induction of TFs has been reported in A. halleri under Cd stress (Weber et al., 2006). Differential expression of ERF factors under Cd indicates toward their responses to various levels of Cd stress. A study by Nakashima and Yamaguchi-Shinozaki (2006) reported down-regulation of dehydration-responsive element-binding protein (DREB) transcription factor (involved in cold and osmotic stress responsive genes) in roots of A. thaliana under heavy metal treatment and suggested it could be acclimation response and DREB might have helped in normalizing osmotic potential, so that flow of heavy metal-contaminated water could be reduced, thus helping plants to avoid toxic effects of heavy metal. Therefore, acquiring a deep knowledge of the interrelated mechanisms, which regulate the expression of these genes, is a crucial issue in plant biology and necessary to generate genetically improved crop plants for extreme environments like heavy metal stress (Umezawa et al., 2006; Valliyodan and Nguyen, 2006; Nakashima et al., 2009). Summary of an involvement of TFs in conferring heavy metal and other abiotic stresses tolerance is given in Table 2.
Table 2
| Name of TF | Family of TF | Studied plant | Plant response | References |
|---|---|---|---|---|
| WRKY6 | WRKY | Arabidopsis thaliana | Plant exhibits dual WRKY-dependent signaling mechanism that modulates Asv uptake and transposon expression and provides a coordinated strategy for Asv tolerance and transposon gene silencing | Castrillo et al., 2013 |
| WRKY22, WRKY25, and WRKY29 | WRKY | Arabidopsis thaliana | TFs induced by Cu and Cd involve in stress response via MAPK and oxylipin signaling | Opdenakker et al., 2012 |
| WRKY45 | WRKY | Arabidopsis spp. | Involved in Zn and Fe stress response and homeostasis | van de Mortel et al., 2006 |
| ART1 | C2H2 | Oryza sativa | Constitutively expressed in roots and regulates genes related to Al tolerance and thus increases Al tolerance | Yamaji et al., 2009 |
| ASR5 | – | Oryza sativa | Overexpression enhanced Al tolerance. Authors suggested that this protein is localized in nucleus and acts as a transcription factor to regulate the expression of different genes that collectively protect rice cells from Al-induced stress | Arenhart et al., 2013 |
| ZIP39 | bZIP | Oryza sativa | Overexpression regulates endoplasmic reticulum (ER) stress-responsive genes and thus regulates ER stress response | Takahashi et al., 2012 |
| HsfA4a | HSF | Oryza sativa | Expression of this TF increases Cd tolerance by inducing up-regulation of MT gene expression | Shim et al., 2009 |
| Hsfs | HSF | Arabidopsis spp. | TF up-regulated by Cd stress and plays a role in Cd stress tolerance | Herbette et al., 2006; Weber et al., 2006 |
| CaPF1 | AP2/EREBP | Pinus Virginiana Mill. | Overexpression of TF enhanced production on of APX, GR, and SOD which confer tolerance against oxidative stress induced by Cd, Cu, and Zn | Tang et al., 2005 |
| OXS2 | C2-H2 ZF | Arabidopsis thaliana | Enhanced Cd tolerance | Blanvillain et al., 2011 |
| ACEl | – | Saccharomyces cerevisiae | TF binds metal-regulatory elements (MREs) upstream promoter of target gene for induction of MT which plays a role in Cu homeostasis | Fürst et al., 1988 |
| ACE1 | – | Arabidopsis thaliana | Overexpression protects plant against Cu stress by inducing activity of SOD and POD, and suppressing inhibition in chlorophyll biosynthesis | Xu et al., 2009 |
| ACE1 | – | Saccharomyces cerevisiae | TF binds MREs upstream promoter of target gene for induction of MT which plays a role in Cu homeostasis | Dixon et al., 1996 |
| ACP1 | AP2/EREBP | Physcomitrella patens | Expression of this gene enhances metal responding genes which confer tolerance against Cd and Cu | Cho et al., 2007 |
| OSISAP1 | Zinc-finger protein | Nicotiana tabacum | Overexpression enhances tolerance against various abiotic stresses including heavy metal like Cu, Cd, Mn, Ca, Zn, and Li | Mukhopadhyay et al., 2004 |
| STOP1 | C2-H2 ZF | Arabidopsis thaliana | Expression protects plants from Al toxicity by proton pump regulation | Iuchi et al., 2007 |
| bHLH38 and bHLH39 | bHLH | Arabidopsis thaliana | Overexpression enhanced Cd tolerance by increased Cd sequestration in roots and also improved Fe homeostasis in shoots | Wu et al., 2012 |
| bHLH100 | bHLH | Arabidopsis spp. | Involved in Zn and Fe stress response and homeostasis | van de Mortel et al., 2006 |
| – | MYB, bHLH, bZIP | Sedum alfredii | These TFs families were up-regulated by Cd and involved in Cd hyperaccumulation and tolerance | Gao et al., 2013 |
| PYE | bHLH | Arabidopsis thaliana | Expression is implicated in regulating plant growth response against Fe deficiency | Long et al., 2010 |
Summary of transcription factors (TFs) whose overexpression in plants confers heavy metal stress tolerance.
Mitogen-activated protein kinase MAPK cascade
MAPK cascade are activated in response by plants when exposed to heavy metal stresses. This cascade has its significance in activation of signal transduction pathway used in hormone synthesis (Jonak et al., 2002). This cascade involves three kinases viz., MAPK kinase kinase (MAPKKK), the MAPK kinase (MAPKK), and the MAPK, which are activated by phosphorylation process. The finally formed phosphorylated MAPK cascade phosphorylates substrates in cells including transcription factors in nucleus. Therefore, MAPK regulates the transduction of information downstream. Jonak et al. (2004) have shown four isoforms of MAPK that are activated under Cu or Cd stress in Medicago sativa. All these pathways finally lead to regulation of transcription factors that in turn activate genes for activation of metal transporters, biosynthesis of chelating compounds, and other defensing compounds.
Proteomics
Proteomics is a well-established technique in the post-genomic era (Liu et al., 2013). Proteomics deals with the study of large-scale expression of proteins in an organism encoded by its genome (Anderson and Anderson, 1998). Proteomics not only serves as a powerful tool for describing complete protein changes in any organisms but it can also be used to compare variation in protein profiles at organ, tissue, cell and organelle levels under various stress conditions including heavy metal stress (Ahsan et al., 2009). Although genomic analysis has enhanced our understanding regarding plants' response to heavy metal toxicity, transcriptomic changes in the genome are not always reflected at protein level (Gygi et al., 1999; Hossain and Komatsu, 2013). For instance, putative Zn and Mg transporter protein MHX was more abundant in Arabidopsis even though its corresponding transcript level was not different (Elbaz et al., 2006). This suggests that transcription of any gene is not a guaranty that gene would be translated into a functional protein. This may occur due to the potential impact of post-transcriptional and translational modifications, protein folding, stability and localization, protein–protein interactions, which are considered important determinants of a protein function (Dalcorso et al., 2013b). Therefore, depth analyses of proteomics offer a new platform for identifying target proteins, which take part in heavy metal detoxification, and in studying complex biological processes and interactions among the possible pathways that involve a network of proteins (Ahsan et al., 2009).
Furthermore, it is known that proteins directly take part in plant stress responses, and plant adaptations to heavy metal stress are always accompanied with deep proteomic changes. Therefore, technique of proteomics can be exploited for deciphering the possible relationships between proteins abundance and plant stress adaptation. It can contribute to better understanding of physiological mechanisms under heavy metal stress such as perception of stress and further signaling cascade that leads to changes in the expression of huge numbers of genes at transcriptional level and in metabolite profile, which could be used for an acquisition of an enhanced plant tolerance under heavy metal toxicity (Kosová et al., 2011). Studies have revealed that an abundance of defense proteins was increased for scavenging of ROS, and molecular chaperones play a role in re-establishing the conformation of a functional protein that contributes in helping heavy metal stressed plants to maintain the redox homeostasis (Zhao et al., 2011; Sharmin et al., 2012; Wang et al., 2012). Under heavy metal stress, modulations of various metabolic pathways occur such as photosynthesis, respiration, nitrogen metabolism, sulfur metabolism, etc. particularly in photosynthesis and mitochondrial respiration that help stressed plants to produce more reducing power such as NADPH, NADH, and FADH2 and assimilatory power ATP to compensate high energy demand of heavy metal-challenged plants (Hossain and Komatsu, 2013). For example, an increased abundance of RUBISCO large sub unit (LSU)-binding proteins, oxygen-evolving enhancer protein 1 and 2, NAD(P)H-dependent oxido-reductase, and photosystem I and II-related proteins is an adaptive feature to withstand heavy metal stress (Semane et al., 2010). The cellular mechanism of stress sensing and further transduction of signals into the cell appear to be the first reactions in the plant cell against heavy metal. Furthermore, an intracellular communication of stress signals plays a fundamental role in signal transduction pathways under stress, which ultimately activate defense-related genes and thus signaling cascades (Hossain et al., 2012c). Therefore, to decipher an underlying molecular mechanism of alterations in the protein signature of a plant cell in order to withstand stress, a deep study on the cellular as well as organelle proteomics would be of great importance in developing heavy metal-tolerant crops. Alterations in protein profile under heavy metal stress, which could be utilized for developing heavy metal-tolerant plants, are given in Table 3.
Table 3
| Metal | Technique(s) used | Plant species | Alterations in protein(s) expression profile | Plant response | References |
|---|---|---|---|---|---|
| Cd | 2DE, MALDI-TOF-MS, LC–ESI-QTOF-MS | Arabidopsis thaliana | ~1100 Spots reported, 41 spots showed significant changes including phytochelatins, glutathione-S-transferases, ATP sulfurylase, glycine hydroxymethyl transferase, trehalose-6-phosphate phosphatase | Alterations in these proteins in plant roots help to withstand Cd stress via modulating S assimilation | Roth et al., 2006 |
| 2DE, MALDI-TOF/TOF-MS | Phytolacca americana | 32 Proteins are differentially expressed, 14 enhanced, and 11 reduced under Cd treatment. Major changes were in photosynthetic pathway, S and GSH metabolism, transcription, translation and chaperones, 2 cys-peroxiadse and oxido-reductases proteins | These alterations play a key role in enhancing Cd hypertolerance in plant | Zhao et al., 2011 | |
| 2DE, MALDI-TOF/TOF-MS | Arabis paniculata | 18 Proteins differentially expressed upon Cd treatment which were mainly related with photosynthetic pathway and antioxidant defense system such as ribulose-5-phosphate 3-epimerase (RPE), RuBisCO activase, Protein thylakoid formation 1 (THF1), Mn-SOD, APX, GST | Plant adopted alterations mainly in antioxidative/xenobiotic defense and hence exhibited increased Cd tolerance | Zheng et al., 2011 | |
| 2-D DIGE, MALDI-TOF/TOF | Populus sp. | A number of changes in the expression of proteins with various functions were identified; in particular a decreased abundance of oxidative stress regulating proteins, whereas pathogenesis-related proteins showed a drastic increase in abundance. Furthermore, a large number of proteins involved in carbon metabolism showed a decrease in abundance, while proteins involved in remobilizing carbon from other energy sources were up-regulated | Due to deep proteomic changes, plant experienced lesser negative impact of Cd on physiological parameters and hence plant showed Cd tolerance | Kieffer et al., 2008 | |
| 2DE, MALDI-TOF-MS | Oryza sativa | 36 Proteins either up-and/or down-regulated by Cd treatment. Most of the proteins were related to oxidative stress and antioxidative system | Antioxidative system related proteins play a role in Cd tolerance | Lee et al., 2010 | |
| 2DE | Thlaspi caerulescens | 48 Tentatively spots identified which represent core metabolic functions, e.g., photosynthesis, nitrogen assimilation, carbohydrate metabolism as well as putative signaling and regulatory functions | The possible roles of some of the proteins were related with metal accumulation and tolerance | Tuomainen et al., 2006 | |
| As | 2DE, MALDI-TOF-MS | Oryza sativa | 23 Proteins up-regulated related with defense proteins like S-adenosylmethionine synthetase (SAMS), GSTs, cysteine synthase (CS), GST-tau, and tyrosine-specific protein phosphatase proteins (TSPP), and an omega domain containing GST | SAMS, CS, GSTs, and GR presumably work synchronously and GSH plays a key role in protecting rice roots against As stress | Ahsan et al., 2008 |
| IPG, 2-DE, MALDI-TOFMS, ESI-MS/MS | Oryza sativa | 12 Proteins differentially expressed related with energy production and metabolism. RuBisCO large subunit and chloroplast 29 kDa ribonucleoproteins were decreased | Reduction in photosynthetic machinery proteins was related with As toxicity | Ahsan et al., 2010 | |
| 2DE, MALDI-TOF-MS, LC-MS/MS | Chlamydomonas reinhardtii | 15 Proteins overexpressed like oxygen-evolving enhancer protein, rubisco small subunit 1, chaperones, Fe-SOD, Mn-SOD, and heat shock like proteins | Organism exhibited time course acclimation against As stress by modulating protein signatures | Walliwalagedara et al., 2012 | |
| Hg | 2-DE, MALDI-TOF-TOF-MS | Suaeda salsa | 43 Proteins with significant changes reported. They include proteins related to metabolic processes, photosynthesis, stress response, protein fate, energy metabolism, signaling pathways, and immunosuppression | Alterations in these proteins was linked with Hg toxicity | Liu et al., 2013 |
| 2DE, ESI-MS/MS | Oryza sativa | 25 Proteins differentially expressed by Hg involved in cellular functions including the redox and hormone homeostasis, chaperone activity, metabolism, and transcription regulation | Plant exhibited Hg toxicity due to alterations in these proteins | Chen et al., 2012a | |
| 2DE | Oryza sativa | 33 Proteins were highly reproducible. Most of the proteins showed homology to RuBisCO protein, and some to defense/stress-related proteins, like the pathogenesis related class 5 protein (OsPR5), the probenazole-inducible protein (referred to as the OsPR10), SOD, and the oxygen evolving protein | Severe fragmentation of ribulose-1,5-bisphosphate carboxylase/oxygenase and induction of stress-related proteins causes Hg toxicity | Hajduch et al., 2001 | |
| Cr | 2DE, MALDI-TOF, MALDI-TOF-TOF | Miscanthus sinensis | 36 Proteins differentially expressed. The identified proteins included: heavy metal-inducible proteins such as carbohydrate and nitrogen metabolism, molecular chaperone proteins, and novel proteins such as inositol monophosphatase, nitrate reductase, adenine phosphoribosyl transferase, formate dehydrogenase, and a putative dihydrolipoamide dehydrogenase | Miscanthus plant experienced Cr toxicity due to altered vacuole Cr sequestration, nitrogen metabolism, and lipid peroxidation in roots | Sharmin et al., 2012 |
| 2DE, MALDI-TOF-MS-MS | Zea mays | 58 Proteins identified related with photosynthesis and chloroplast organization, the redox homeostasis and defense response, RNA processing, protein synthesis and folding, DNA damage response, mitochondrial oxidative phosphorylation, and miscellaneous with unknown function | Plant exhibited Cr toxicity due to the deep changes in proteomics | Wang et al., 2013 | |
| Cu | 2DE | Oryza sativa | Changes RuBisCO, defense/stress-related proteins, like the pathogenesis related class 5 protein (OsPR5), the probenazole-inducible protein (referred to as the OsPR10), and SOD | Alterations in these protein resulting in Cu stress | Hajduch et al., 2001 |
| SDS-PAGE and 2DE | Oryza sativa | 13 Proteins identified such as metallothionein-like protein, membrane-associated protein-like protein, putative wall-associated protein kinase, pathogenesis-related proteins, and the putative small GTP-binding protein Rab2 which were up regulated by Cu stress. Three proteins, a putative small cytochrome P450 (CYP90D2), a putative thioredoxin and a putative GTPase, were down regulated by Cu stress | Plant experienced Cu toxicity due to a decline in thioredoxin and CYP90D2 and thus engineering of this protein may enhance Cu tolerance | Zhang et al., 2009a,b | |
| 2DE-MS | Populus sp. | 450 Proteins were reproducibly separated, including metabolic processes proteins such as photosynthesis, S assimilation, sugar metabolism, chaperones, and defense related proteins such as GST, DHAR, APX | Plant adjusts its metabolism against Cu stress by changing protein expression. These proteomic temporal features should be taken into account for the future development of metal tolerant plants | Lingua et al., 2012 | |
| IPG, 2-DE, MALDI-TOF-MS | Ectocarpus siliculosus | Up-regulation of photosynthesis (PSII Mn-stabilizing protein of OEC33), glycolysis, and pentose phosphate metabolism; higher accumulation of HSP70 and vBPO | Cu stress leads to up-regulation of certain proteins such as HSP70 and vBPO for proper protein folding and ROS detoxification, respectively | Ritter et al., 2010 | |
| IPG, 2-DE, LC-MS/MS | Cannabis sativa | Induced aldo/keto reductase, PCs expression, suppression/no change in ROS scavenging enzymes | Cu induced aldo/keto reductase acts as a Cu chaperone reduce Cu ions to Cu(I), promote PCs-mediated vacuolar transport in order to reduce Cu toxicity | Bona et al., 2007 | |
| SDS-PAGE and 2DE | Oryza sativa | 25 Protein spots were differentially expressed in Cu-treated samples. Among them, 18 protein spots were up-regulated, and 7 protein spots were down-regulated. Antioxidants proteins such as glyoxalase I, peroxiredoxin, aldose reductase, and DnaK-type molecular chaperone up-regulated. Moreover, down-regulation of key metabolic enzymes like alpha-amylase or enolase revealed also observed | Plant showed physiological alterations under Cu stress due to the change in metabolic pathway related proteins | Ahsan et al., 2007 | |
| Zn | iTRAQ | Arabidopsis thaliana | 521 Proteins identified. Among them, several were membrane proteins. IRT1, an iron and zinc transporter, and FRO2, a ferric-chelate reductase, increased greatly in response to excess Zn | Plant exhibits Zn stress in which V-ATPase activity might play a central role | Fukao et al., 2011 |
| 2DE-MS | Populus sp. | 450 Proteins were reproducibly separated, including metabolic processes proteins such as photosynthesis, S assimilation, sugar metabolism, chaperones, and defense related proteins such as GST, DHAR, APX | Plant adjusts its metabolism against Zn stress by changing protein expression | Lingua et al., 2012 | |
| Ni | 2DE, MALDI-TOF-MS | Brassica juncea | 61 Proteins differentially expressed. The majority of proteins were found to be involved in S metabolism and protection against oxidative stress. The induced expression of photosynthesis and ATP generation-related proteins were also observed | An increased expression of defense proteins and those related with energy metabolism suggesting the Ni tolerance in plant is an energy-demanding process | Wang et al., 2012 |
| 2-DE, LC-MS/MS | Alyssum lesbiacum | 12 Proteins differentially expressed. They include proteins of S metabolism, antioxidants, heat shock | Modulation in S metabolic and defense related proteins enhanced Ni tolerance of plant | Ingle et al., 2005 | |
| Mn | IPG, 2-DE, Nano-LC-MS/MS, ESI MS/MS | Vigna unguiculata | 8 Differentially expressed proteins indentified involved in CO2 fixation, stabilization of the Mn cluster of the photosystem II, pathogenesis-response reactions, and protein degradation | Coordinated interplay of apoplastic and symplastic reactions help plant to withstand Mn toxicity | Führs et al., 2008 |
| 2DIEF/SDS-PAGE, 2D Blue native BN/SDS-PAGE | Hordeum vulgare | A range of proteins differentially expressed in response to Mn. A putative inorganic pyrophosphatase, a probenazole-inducible protein (PBZ1), a protein belonging to a universal stress protein (Usp) family, a chloroplast translational elongation factor (Tu) and the 50S ribosomal protein L11 | In young leaves toxicity resulted due to Mn-induced Mg and Fe deficiencies | Führs et al., 2010 |
Summary of heavy metal-induced changes in protein expressions and their potential uses in developing heavy metal tolerant plants.
Apart from inducing synthesis of amino acids (proline and histidine), amines, organic acids, and plant antioxidant α-tocopherol and glutathione, some nitrogen containing metabolites like some peptides (phytochelatins, metallothioneins, and ferritins) have been reported to play an important role under heavy metal stress. In the following section, we will discuss about the roles of peptides in heavy metal tolerance.
Peptides
Phytochelatins (PCs) have been the best-characterized chelators in plant systems. PCs belong to a family of metal-binding protein having general structure (c-Glu-Cys)nGly (n = 2–11) (Cobbett and Goldsbrough, 2002). These are synthesized by the transpeptidation of the γ-Glu-Cys moiety of GSH, and the transpeptidation reactions are carried out by enzyme named phytochelatin synthase (PCS). It has been reported that PCS were activated under heavy metal exposure (Rauser, 1995; Cobbett, 2000), and similar PC synthase activity has been observed in several other crops (Klapheck et al., 1995; Chen et al., 1997; Mishra et al., 2009a). Loeffler et al. (1989) confirmed that metals induce PC synthesis, in in-vivo as well as in in-vitro cultures, and were continuously synthesized until activated metal ions chelated. Haag-Kerwer et al. (1999) reported induction of PCs in B. juncea after the accumulation of Cd in the cells, and thus plays important role in detoxifying heavy metals (Hirata et al., 2005). Due to the presence of thiol group, they have the capability of chelating metals and forming complexes (Cobbett, 2000), which are then sequestrated in the vacuole. From the preceding discussion, it is clear that chelation by PCs is not a simple process but involves a complex molecular mechanism, where firstly, the PCS gets activated by metal ion and biosynthesis of PCs takes place; secondly, formation of complexes and sequestration in vacuole; thirdly, more complexation with the sulfides or organic acids in the vacuole, and finally detoxified. Besides detoxifying heavy metals, PCs also play a major role in metal ion homeostasis and thus regulating the metal ion availability in plant cells (Guo et al., 2008b).
Like PCs, metallothioneins (MTs) are synthesized and activated under heavy metal toxicity. They belong to a family of low molecular weight protein having cysteine-rich metal binding peptide. Due to the presence of mercaptides, they have the ability of binding metal ions. Metal-binding activities of MTs have been expressed in Escherichia coli in presence of Cd, Zn, and Cu (Tommey et al., 1991). In addition, Zhou and Goldsbrough (1994) reported restoration in Cu tolerance ability of MT-deficient yeast strains, when provided with the Arabidopsis MTs. Similar to this, Zhigang et al. (2006) conferred increased tolerance of A. thaliana to Cd and Cu, when ectopically substituted with B. juncea MT. Moreover, comparative study of mutant and wild-type A. thaliana has clearly revealed that MT mutant was hypersensitive to Cd and accumulated much lower amount of Cd than wild type, thus conferring role of MTs in both heavy metal tolerance as well as accumulation (Zimeri et al., 2005). In terms of transcript amount, expression of MT genes varies during different developmental stages of plant as well as under varying environmental condition (Rauser, 1999). Beside, chelating metal ions MTs can also catalyze antioxidant protection mechanism as well as plasma membrane repair (Hamer, 1986).
Ferritins are other multimeric proteins that could accumulate iron atom (Harrison and Arosio, 1996). However, animal ferritins have been reported to store other metals like Cu, Zn, Cd, etc., whereas plants ferritin could store only Fe. These are synthesized in plants when there is excess Fe in the surroundings and thus represents first-line defense against Fe-induced oxidative stress (Ravet et al., 2009). These are not only involved in storing or releasing Fe but also involved in scavenging free reactive iron (Ravet et al., 2009).
Plant growth hormones
In spite of five classical plant hormones, i.e., gibberellins (GAs), cytokinins (CKs), auxins, abscisic acid (ABA), and ethylene, jasmonate (JA), brassinosteroids (BR), and salicylic acid (SA) are also well known for regulating many physiological processes and heavy metal stress tolerance (Freeman et al., 2005b; Gangwar et al., 2010; Gangwar and Singh, 2011; Peleg and Blumwald, 2011; Choudhary et al., 2012a,b; Vriet et al., 2012). Furthermore, it is also expected that some more growth hormones are yet to be discovered in future. In laboratory as well as filed studies, two strategies have been used for plant hormone-mediated increase in stress tolerance as well as crop yield. These strategies include exogenous application of plant hormones and genetic manipulation of their endogenous contents. Both approaches have given promising results for increasing crop yield and enhancing stress tolerance in a variety of crop species (Vriet et al., 2012). Although SA and GAs both are cost effective and can easily be availed for their exogenous application in crop fields under stress conditions, high cost of synthetic BRs and the variability of the results have discouraged the use of exogenous BRs in agriculture and horticulture (Khripach et al., 2000; Gomes, 2011). In this context, modulation of endogenous BRs levels by genetic engineering has emerged an efficient strategy for enhancing crop yield under normal as well as adverse growth conditions (Divi and Krishna, 2009). Herein, we have summarized recent advances made in enhancing heavy metal tolerance as well as achieving high yield with desired agronomic traits by using salicylic acid (SA), brassinosteroids (BRs), and gibberellins (GA).
Salicylic acid (SA)
In recent years, SA has gained much scientific attention due to its function as an endogenous signaling molecule conveying local and systemic plant–pathogen defense responses. Besides this, it has been reported that SA also plays a role in plant response against abiotic stresses such as heavy metal toxicities, chilling, drought, osmotic stress, and heat. In this sense, SA appears to be an “effective therapeutic agent” for plants as in the case of mammals (Rivas-San Vicente and Plasencia, 2011). Salicylic acid is a phenolic compound biosynthesized in all the plant kingdoms through the phenylpropanoid pathway (Métraux, 2002).
Being well characterized and studied role of SA in pathogen resistance, an exogenous application of SA could also provide protection against several types of abiotic stresses such as heavy metals, high or low temperature, salinity, radiation, etc. (Horváth et al., 2007; Hayat et al., 2010). Since under stress condition, reduced plant growth could result from an altered hormonal status, and thus, an exogenous application of plant hormones like SA has been an attractive approach to attenuate heavy metal stress. Studies carried out so far demonstrated that SA treatment to plants evoke acclimatization effect, which causes an enhanced tolerance toward heavy metal stress primarily due to the adjustment of metabolic processes such as enhanced antioxidative capacity. In one of the first works, it was demonstrated that SA may induce protective effects against Cu toxicity in tobacco and cucumber (Strobel and Kuc, 1995). Later, an increasing numbers of studies have demonstrated SA-mediated amelioration of toxicities produced by various heavy metals. Zhou et al. (2009) reported that 0.2 mM of SA ameliorates Hg toxicity in alfalfa by increasing activity of APX, POD, and NADPH oxidase, and amounts of ascorbate, glutathione, and proline, and decreased lipid peroxidation, and an increase in NADPH oxidase activity. It indicates a role of ROS signaling in such an amelioration process.
In maize plant, Cd declined the growth by inhibiting chlorophyll synthesis, ribulose 1,5-bisphosphate carboxylase and phosphoenolpyruvate carboxylase, and enhancing oxidative damage such as lipid peroxidation and electrolyte leakage, whereas SA pretreatment of seeds reversed these toxic effects (Krantev et al., 2008). In cucumber, an exogenous application of SA has also been reported to enhance Mn tolerance by modulating nutrients' statuses and antioxidant defense system (Shi and Zhu, 2008). Similarly, in pea seedlings, Cd toxicity caused decline in growth due to an inhabited photosynthetic process and enhanced oxidative damage, whereas SA pretreatment alleviated damaging consequences of Cd on growth and photosynthesis (Popova et al., 2009). Moreover, Guo et al. (2009) have demonstrated that SA pretreatment alleviated Cd toxicity in rice by enhancing antioxidant components such as SOD, CAT, POD, glutathione, and non-protein thiols, which in turn depressed oxidative damage induced by Cd. Conversely, Metwally et al. (2003) reported that SA down-regulates activities of antioxidant enzymes such as CAT and APX under Cd stress and concluded that SA alleviates Cd toxicity not at the level of antioxidant defense system but by affecting other mechanisms of Cd detoxification. Contrary to this, SA at higher concentration may also cause tissue damage and cell death by inducing oxidative stress (Horváth et al., 2007). For instance, SA has been shown to potentiate generation of ROS in photosynthetic tissue under abiotic stresses and thus causes tissue damage (Borsani et al., 2001). Therefore, it can be concluded that the concentration of SA appears to be important in regulating stress responses. The SA-mediated alterations in genes that are involved in mediating stress tolerance are listed in Table 4.
Table 4
| Plant hormone | Alteration in gene(s) | Studied plant | Response | References |
|---|---|---|---|---|
| Salicylic acid | Heam oxygenase-1 (HO-1) | Medicago sativa | Alleviation of Cd-triggered oxidative stress by re-establishing redox homeostasis | Cui et al., 2012b |
| Serine acetyltransferase | Thlaspi spp. | Elevated level of glutathione and increased Ni tolerance | Freeman et al., 2005b | |
| Citrate synthase | Cassia tora | Enhanced Al tolerance through an efflux of citrate | Yang et al., 2003 | |
| SR3 | Phaseolus vulgaris | This gene up-regulated by SA and provides resistance against Hg, Cd, As, and Cu | Zhang et al., 2006 | |
| gsh1, gsh2, or gr1 and gst | Arabidopsis thaliana | SA did not influence expression of these genes except gst and thus did not affect Cu and Cd tolerance | Xiang and Oliver, 1998 | |
| MT1 and MT2 | Arabidopsis thaliana | SA did not alter expression of these genes hence did not impart Cu tolerance | Murphy and Taiz, 1995 | |
| Brassinosteroids | Antioxidant defense related genes | Raphanus sativus | Increased resistance against Cr toxicity due to diminished production of ROS and an enhanced defense system | Choudhary et al., 2012b |
| Fe-SOD, CAT1, APX, GST1, GR, POD, GSH1, PAL, PPO, SKDH, and CAD | Solanum lycopersicum | Alleviates Cd-induced inhibition on photosynthesis by up-regulating defense system and decreasing oxidative stress | Ahammed et al., 2013 | |
| Genes encoding polyamines, IAA and ABA metabolic genes, and Cu homeostasis | Raphanus sativus | Lower ion leakage due to a maintenance of Cu homeostasis and hence an enhanced Cu tolerance | Choudhary et al., 2012a | |
| HSP83, HAT2, GH3.9, SAL2, NIA1, GAS4, SAUR36, DWARF1, DWARF4, and BR6OX | Arabidopsis thaliana | BR-exhibited synergistic effect with Cd and increased Cd sensitivity of plants | Villiers et al., 2012 | |
| NADPH oxidase and RBOH, MAPK1, and MAPK3 | Cucumis sativus | BR-mediated production of H2O2via NADPH oxidase increased stress tolerance in cooperation with kinases | Xia et al., 2009 | |
| Induced NO production that up-regulates ABA biosynthetic gene vp14 | Zea mays | BR-induced NO production that up-regulates ABA biosynthesis gene vp14 and thus confers stress tolerance | Zhang et al., 2011 | |
| Set of stress marker genes | Brassica napus | Increased tolerance against abiotic stresses such as drought and cold | Kagale et al., 2007 | |
| UBC32, a stress-induced functional ubiquitin conjugation enzyme (E2) | Arabidopsis thaliana | Protects plants from abiotic stress through endoplasmic reticulum (ER)-associated protein degradation (ERAD) component and UBC32 plays a crucial role in such protection | Cui et al., 2012a | |
| Gibberellic acid | IRT1 | Arabidopsis thaliana | GA-suppressed up-regulation of IRT1 and enhanced accumulation of NO that enhanced Cd tolerance | Zhu et al., 2012 |
| CAX2 | Nicotiana tabacum | GA did not influence expression of this gene and did not alter Mn and Cd tolerance | Hirschi et al., 2000 | |
| GA-biosynthesis and redox genes | Glycine max | Increased Cu tolerance due to decreased oxidative damage and enhanced antioxidant defense system | Khan and Lee, 2013 | |
| adenosine 5′-phosphosulfate reductase (APR) | Arabidopsis thaliana | GA plays a role in abiotic stress tolerance via regulating S assimilation pathway | Koprivova et al., 2008 |
Summary of plant hormone-mediated alterations in genes and their relation with an increased heavy metal stress tolerance.
It is known that SA also involves in the regulation of oxidative stress caused by various stress factors (Yang et al., 2004). An enhanced level of SA under heavy metal stress suggests a connection between the extent of plant tolerance to heavy metal, which is mediated by the SA signal and the redox balance (Metwally et al., 2003; Sharma and Dietz, 2009). In the SA signaling under heavy metal stress, several signaling molecules such as nitric oxide (NO), H2O2, Ca+2, etc. and their interactions have been reported (Rodríguez-Serrano et al., 2009; Xu et al., 2014). Moreover, Cui et al. (2012b) have reported a cross-talk of haem oxygenase-1 and SA in alleviation of Cd stress in M. sativa. In spite of considerable progress in the understanding of SA signaling, molecular events, which are involved in the SA signaling in order to alleviate heavy metal stress, are still poorly known (Figure 4).
Figure 4
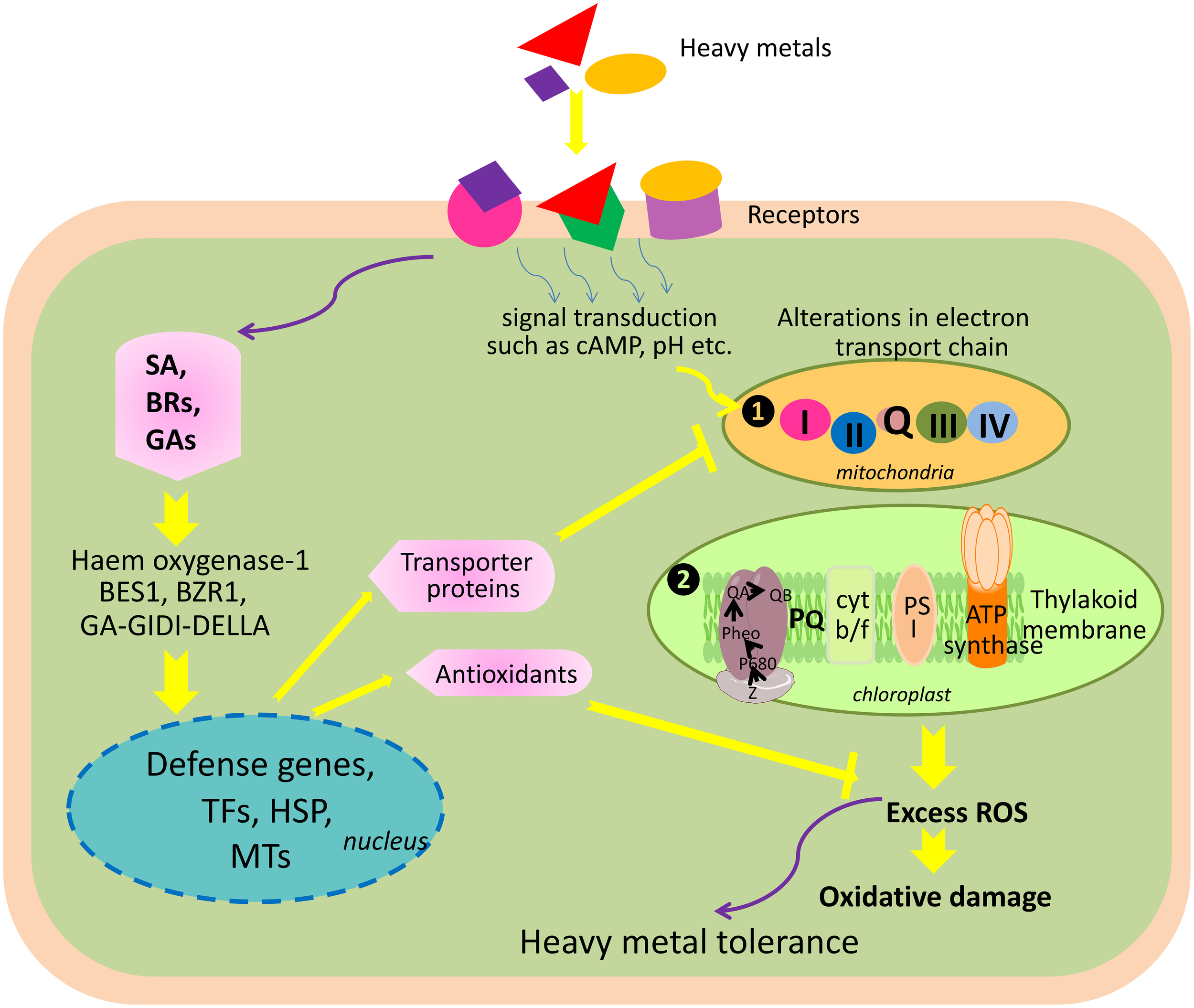
Schematic representation of plant hormone-mediated alleviation of heavy metal toxicity in plants. Heavy metals' signals are perceived by receptors, and receptors transduce signals via cAMP, pH, etc. causing alterations in electron transport systems of the cell, which results into an excess production of reactive oxygen species (ROS). ROS cause damage to macromolecules and thus create oxidative stress inside the cell. On the other hand, in the presence of plant hormones, signals received by them initiate a cascade of signal transduction involving haem oxygenase, two transcription factors induced by brassinosteroids (BES1 and BZR1) and a gibberellic acid-mediated GA-GID1-DELLA signaling pathway. These factors, in turn, initiate expression of the nuclear genes encoding defense proteins, transcription factors (TFs), heat shock proteins (HSP), and metal transporter proteins (MTs). MTs protect electron transport chains against heavy metals by regulating their uptake. Defense proteins protect plant against ROS under heavy metal stress (Numbers 1 and 2 designated to chloroplast and mitochondria show the sources of ROS in cell).
Brassinosteroids (BRs)
Brassinosteroids are group of hormones having ability of regulating ion uptake in plant cells and very effectively reducing the heavy metal accumulation in plants. BRs can also impart plant stress tolerance against variety of biotic and abiotic stresses such as heavy metal, salinity, drought, low and high temperatures, and pathogen attack (Bajguz and Hayat, 2009; Hao et al., 2013). An increasing numbers of studies have shown that an exogenous application of BRs is widely used in order to improve crop yield as well as stress tolerance in various plant species (Divi and Krishna, 2009; Peleg and Blumwald, 2011; Li et al., 2013). Cadmium, a heavy metal, very toxic even when present in trace amount, have been found to retard chlorophyll biosynthesis, activity of several enzymes, and inhibit light and dark reactions of photosynthesis by limiting the energy/reducing sources (Vassilev and Yordanov, 1997). However, it has been reported that Cd-induced toxicity can be lowered with BR. For instance, Janeckzo et al. (2005) reported that Cd-induced inhibition in pigments content, cotyledon growth could be minimized with exogenous epibrassinolide (EPL: another BR). Hayat et al. (2007) have verified the role of HBL under Cd stress in B. juncea. In Vigna radiata L. Wilczek, Al stress caused a reduction in length, fresh and dry mass of root and shoot; activity of carbonic anhydrase; water use efficiency; relative water content; chlorophyll content; and the rate of photosynthesis, whereas addition of BR reversed these toxic effects and protected the plants via elevated level of proline in an association with an antioxidant defense system which at least in part was responsible for the amelioration of Al stress (Ali et al., 2008). In B. juncea, BR alleviates Cd toxicity through enhanced level of antioxidants (Hayat et al., 2007). The Cr, a known toxic metal, reduced the growth performance of Raphanus sativus L., whereas BR protects plants from adverse consequences of Cr toxicity by regulating antioxidant defense system (Sharma et al., 2011). Micronutrients such as Cu and Ni are essential for growth and development, but in excess, they cause severe toxic effects. Cu, which has increasingly attained interest due to its use in fungicides, fertilizers, and pesticides, is also highly toxic to plants, but when seeds of B. juncea primed with epibrassinolide (a form of BR) were exposed to Cu stress, improvement in shoot emergence and biomass accumulation, along with reduced Cu uptake and accumulation, was noticed (Sharma and Bhardwaj, 2007). Similar protective responses of exogenous BR on B. juncea and V. radiata under Cu and Ni toxicities have been reported (Alam et al., 2007; Sharma et al., 2008; Fariduddin et al., 2009; Yusuf et al., 2012b). Besides higher plants, BR has also been found to be effective in alleviating heavy metals such as Cu, Pb, and Cd toxicities in algae, Chlorella vulgaris, through the regulation of antioxidant defense system (Bajguz, 2010). The BR-mediated alterations in the gene expressions and their roles in stress tolerance are listed in Table 2.
Gibberellic acid (GA)
The gibberellins (GAs) are a large family of tetracyclic diterpenoid plant growth hormone associated with the plant growth and developmental processes (Matsuoka, 2003). To alleviate deleterious effects of stress, different types of plant hormones have been used that might complement decreased and/or imbalanced hormone level during exposure of stress. Of these, GA has been a focus of plant scientists (Hisamatsu et al., 2000; Iqbal et al., 2011; Zhu et al., 2012).
Several studies revealed that GA alleviates various abiotic stresses including heavy metal toxicity. In A. thaliana, GA (5 μM) is reported to ameliorate Cd toxicity by reducing Cd uptake and lipid peroxidation (Zhu et al., 2012). Furthermore, authors demonstrated that GA reduces NO level which in turn down-regulates expression of IRT1 gene, a Fe transporter (might be involved in Cd absorption) as indicated by no effect of GA in reduction of Cd uptake in an IRT1 knockout mutant irt1. It is reported that an exogenous addition of GA reprograms the growth of soybean under stress conditions by enhancing the levels of daidzein and genistein contents, suggesting protective role of GA in mitigating adverse consequences of stressors (Hamayun et al., 2010). In wheat seedlings, Ni (50 mM) has been shown to decline growth, chlorophyll content, and carbonic anhydrase activity by enhancing oxidative stress, whereas an addition of GA ameliorates Ni-induced toxic effects (Siddiqui et al., 2011). Gangwar et al. (2011) have also reported that an exogenous addition of GA ameliorates toxic effects of Cr (50–250 μM) on growth and ammonium assimilation of pea seedlings by regulating oxidative stress and an antioxidant system. In B. napus L., GA (50 μM) has been shown to alleviate Cd (10–400 μM)-induced negative impact on seed germination and growth by regulating oxidative stress and damage (Meng et al., 2009). It has been observed that Pb and Zn affect seed germination in Cicer arietinum cv. Aziziye-94 by altering hormonal balance, and an exogenous application of GA reverses the toxic effect of heavy metals (Atici et al., 2005). Furthermore, Sharaf et al. (2009) have also reported that GA mitigates detrimental effects of Cd and Pb on broad bean and lupin plants by regulating activities of proteases, CAT, and POD. These studies clearly indicate that GA plays an important role in protecting plant metabolism against various stresses; however, this may occur via various routes suggesting complex GA signaling during plant acclimation against stresses. The GA-mediated alterations in genes and their relation with stress tolerance are summarized in Table 4.
Conclusion and future prospects
Around 3.1 billion people from developing countries live in rural areas, and out of this population, ~2.5 billion people depend on agricultural practices for their livelihood, which contributes 30% to economic growth because of the gross domestic products obtained from agriculture (FAO, 2012). It is expected that world population will be about 10 billion by the middle of the twenty-first century, and we will witness serious food shortages (Smith et al., 2010; Naika et al., 2013). Furthermore, the situation will likely to be severe due to increased anthropogenic activities that have resulted into unwanted changes in the environment such as soil, air, and water pollution with various factors including heavy metal. These situations (pollution and population) are posing a continuously increasing burden on global crop productivity, and hence, there are demands for crop varieties that should be adaptive and resistant to various stresses.
In contrast to biotic stress, which is under the control of monogenic trait, abiotic stress tolerance is a genetically complex process that involves many components of signaling pathways, multigenic in nature, and thus, comparatively more difficult to control and engineer (Vinocur and Altman, 2005). Therefore, plant-engineering strategies for heavy metal tolerance depend on the expression of gene(s) whose product(s) are involved either in signaling and regulatory pathways or in the synthesis of functional and structural proteins and metabolites that confer heavy metal stress tolerance. Recently, several efforts are being made to improve heavy metal stress tolerance capacity through genetic engineering with several achievements; however, the genetically complex mechanisms of heavy metal stress tolerance and transfer of technology to field conditions make it difficult. Advances in various functional tools, resources, and “omics” have helped in the molecular characterization of the genes, metabolites, and proteins involved in heavy metal stress tolerance. Furthermore, genetic engineering of heavy metal-responsive genes (particularly TFs), metabolites, and proteins has shown surprising results but its full potential remains to be exploited. The design of future experiments that use a multidisciplinary approach with well-integrated “omics,” i.e., transcritomics, metabolomics, proteomics, etc. ultimately required to significantly improve heavy metal tolerance as well as tolerance to other abiotic stresses in economically important crop plants.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Statements
Acknowledgments
The University Grants Commission, Central Regional Office, Bhopal and New Delhi are greatly acknowledged for providing financial support to Dr. VS (PI-UGC Minor research Project, File No. MS-27/201022/XII/13-14/CRO) and Dr. SP (PI- UGC Major research Project no: 41-460/2012(SR)) respectively. We are also very grateful to Department of Botany, University of Allahabad for providing necessary lab facilities and also to University Grant Commission for providing financial support to PP and RS as research fellow to carry out this work.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1
Abul KashemM. D. A.KawaiS. (2007). Alleviation of cadmium phytotoxicity by magnesium in Japanese mustard spinach. Soil Sci. Plant Nutr.53, 246–251. 10.1111/j.1747-0765.2007.00129.x
2
AcquaahG. (2007). Principles of Plant Genetics and Breeding. Oxford, UK: Blackwell.
3
AhammedG. J.ChoudharyS. P.ChenS.XiaX.ShiK.ZhouY.et al. (2013). Role of brassinosteroids in alleviation of phenanthrene-cadmium co-contamination-induced photosynthetic inhibition and oxidative stress in tomato. J. Exp. Bot.64, 199–213. 10.1093/jxb/ers323
4
AhsanN.LeeD. G.AlamI.KimP. J.LeeJ. J.AhnY. O.et al. (2008). Comparative proteomic study of arsenic-induced differentially expressed proteins in rice roots reveals glutathione plays a central role during As stress. Proteomics8, 3561–3576. 10.1002/pmic.200701189
5
AhsanN.LeeD. G.KimK. H.AlamI.LeeS. H.LeeK. W.et al. (2010). Analysis of arsenic stress-induced differentially expressed proteins in rice leaves by two-dimensional gel electrophoresis coupled with mass spectrometry. Chemosphere78, 224–231. 10.1016/j.chemosphere.2009.11.004
6
AhsanN.LeeD. G.LeeS. H.KangK. Y.LeeJ. J.KimP. J.et al. (2007). Excess copper induced physiological and proteomic changes in germinating rice seeds. Chemosphere67, 1182–1193. 10.1016/j.chemosphere.2006.10.075
7
AhsanN.RenautJ.KomatsuS. (2009). Recent developments in the application of proteomics to the analysis of plant responses to heavy metal. Proteomics9, 2602–2621. 10.1002/pmic.200800935
8
AlamM. M.HayatS.AliB.AhmadA. (2007). Effect of 28-homobrassinolide treatment on nickel toxicity in Brassica juncea. Photosynthetica45, 139–142. 10.1007/s11099-007-0022-4
9
AliB.HasanS. A.HayatS.HayatQ.YadavS.FariduddinQ.et al. (2008). A role for brassinosteroids in the amelioration of aluminium stress through antioxidant system in mung bean (Vigna radiata L. Wilczek). Environ. Exp. Bot.62, 153–159. 10.1016/j.envexpbot.2007.07.014
10
AliB.QianP.JinR.AliS.KhanM.AzizR.et al. (2013a). Physiological and ultra-structural changes in Brassica napus seedlings induced by cadmium stress. Biol. Plant.58, 131–138. 10.1007/s10535-013-0358-5
11
AliB.WangB.AliS.GhaniM. A.HayatM. T.YangC.et al. (2013b). 5-Aminolevulinic acid ameliorates the growth, photosynthetic gas exchange capacity, and ultrastructural changes under cadmium stress in Brassica napus L. J. Plant Growth Regul.32, 604–614. 10.1007/s00344-013-9328-6
12
AndersonN. L.AndersonN. G. (1998). Proteome and proteomics: new technologies, new concepts, and new words. Electrophoresis19, 1853–1861. 10.1002/elps.1150191103
13
AnjumN. A.GillS. S.GillR.HasanuzzamanM.DuarteA. C.PereiraE.et al. (2014). Metal/metalloid stress tolerance in plants: role of ascorbate, its redox couple, and associated enzymes. Protoplasma251, 1265–1283. 10.1007/s00709-014-0636-x
14
AnjumN. A.UmarS.AhmadA.IqbalM.KhanN. A. (2008). Sulphur protects Mustard (Brassica campestris L.) from cadmium toxicity by improving leaf ascorbate and glutathione. Plant Growth Regul.54, 271–279. 10.1007/s10725-007-9251-6
15
ApelK.HirtH. (2004). Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol.55, 373–399. 10.1146/annurev.arplant.55.031903.141701
16
ArbonaV.ManziM.OllasC. D.Gonez-CadenaoA. (2013). Metabolites as a tool to investigate abiotic stress tolerance in plants. Int. J. Mol. Sci. 14, 4885–4911. 10.3390/ijms14034885
17
ArenhartR. A.De LimaJ. C.PedronM.CarvalhoF. E. L.Da SilveiraJ. A. G.RosaS. B.et al. (2013). Involvement of ASR genes in aluminium tolerance mechanisms in rice. Plant Cell Environ.36, 52–67. 10.1111/j.1365-3040.2012.02553.x
18
ArrivaultS.SengerT.KrämerU. (2006). The Arabidopsis metal tolerance protein AtMTP3 maintains metal homeostasis by mediating Zn exclusion from the shoot under Fe deficiency and Zn oversupply. Plant J.46, 861–879. 10.1111/j.1365-313X.2006.02746.x
19
AsadaK. (1992). Ascorbate peroxidase: a hydrogen peroxide scavenging enzyme in plants. Plant Physiol.85, 235–241. 10.1111/j.1399-3054.1992.tb04728.x
20
AsadaK. (1996). Radical production and scavenging in chloroplasts, in Photosynthesis and the Environment, ed BakerN. (Dordrecht: Kluwer; Atlantic Canada Society for Microbial Ecology; Halifax), 123–150.
21
AssuncãoA. G. L.SchatH.AartsM. G. M. (2003). Thlaspi caerulescens, an attractive model species to study heavy metal hyperaccumulation in plants. New Phytol.159, 351–360. 10.1046/j.1469-8137.2003.00820.x
22
AssunçãoA. G.Da Costa MartinsP.De FolterS.VooijsR.SchatH.AartsM. G. M. (2001). Elevated expression of metal transporter genes in three accessions of the metal hyperaccumulator Thlaspi caerulescens. Plant Cell Environ.24, 217–226. 10.1111/j.1365-3040.2001.00666.x
23
AssunçãoA. G.HerreroE.LinY. F.HuettelB.TalukdarS.SmaczniakC.et al. (2010). Arabidopsis thaliana transcription factors bZIP19 and bZIP23 regulate the adaptation to zinc deficiency. Proc. Natl. Acad. Sci. U.S.A.107, 10296–10301. 10.1073/pnas.1004788107
24
AstolfiS.ZuchiS.PasseraC. (2004). Role of sulphur availability on cadmium-induced changes of nitrogen and sulphur metabolism in maize (Zea mays L.) Leaves. J. Plant Physiol.161, 795–802. 10.1016/j.jplph.2003.11.005
25
AticiÖ.AğarG.BattalP. (2005). Changes in phytohormone contents in chickpea seeds germinating under lead or zinc stress. Biol. Plant.49, 215–222. 10.1007/s10535-005-5222-9
26
AtkinsonN. J.UrwinP. E. (2012). The interaction of plant biotic and abiotic stresses: from genes to the field. J. Exp. Bot.63, 3523–3544. 10.1093/jxb/ers100
27
AxelsenK. B.PalmgrenM. G. (1998). Evolution of substrate specificities in the P-type ATPase superfamily. J. Mol. Evol.46, 84–101. 10.1007/PL00006286
28
AzzarelloE.PandolfiC.GiordanoC.RossiM.MugnaiS.MancusoS. (2012). Ultramorphological and physiological modifications induced by high zinc levels in Paulownia tomentosa. Environ. Exp. Bot.81, 11–17. 10.1016/j.envexpbot.2012.02.008
29
BabuT. S.AkhtarT. A.LampiM. A.TripuranthakamS.DixonD. G.GreenbergB. M. (2003). Similar stress responses are elicited by copper and ultraviolet radiation in the aquatic plant Lemnagibba: implication of reactive oxygen species as common signals. Plant Cell Phys.44, 1320–1329. 10.1093/pcp/pcg160
30
BajguzA. (2010). An enhancing effect of exogenous brassinolide on the growth and antioxidant activity in Chlorella vulgaris cultures under heavy metal stress. Environ. Exp. Bot.68, 175–179. 10.1016/j.envexpbot.2009.11.003
31
BajguzA.HayatS. (2009). Effects of brassinosteroids on the plant responses to environmental stresses. Plant Physiol. Biochem.47, 1–8. 10.1016/j.plaphy.2008.10.002
32
BarkerA. V.PilbeamD. J. (2007). Hand Book of Plant Nutrition. Boca Raton, FL: Taylor and Francis.
33
BashriG.PrasadS. M. (2015). Indole acetic acid modulates changes in growth, chlorophyll a fluorescence and antioxidant potential of Trigonella foenum-graecum L. grown under cadmium stress. Acta Physiol. Plant.37:1745. 10.1007/s11738-014-1745-z
34
BasileA.SorboS.PisaniT.PaoliL.MunziS.LoppiS. (2012). Bioacumulation and ultrastructural effects of Cd, Cu, Pb and Zn in the moss Scorpiurum circinatum (Brid.) Fleisch & Loeske. Environ. Pollut.166, 208–211. 10.1016/j.envpol.2012.03.018
35
BasuU.GoodA. G.TaylorG. J. (2001). Transgenic Brassica napus plants overexpressing aluminium-induced mitochondrial manganese superoxide dismutase cDNA are resistant to aluminium. Plant Cell Environ.24, 1278–1269. 10.1046/j.0016-8025.2001.00783.x
36
BaszynskiT.WajdaL.KrolM.WolinskaD.KrupaZ.TukendorfA. (1980). Photosynthetic activities of cadmium-treated tomato plants. Physiol. Plant.48, 365–370. 10.1111/j.1399-3054.1980.tb03269.x
37
BecherM.TalkeI. N.KrallL.KrämerU. (2004). Cross-species microarray transcript profiling reveals high constitutive expression of metal homeostasis genes in shoots of the zinc hyperaccumulator Arabidopsis halleri. Plant J.37, 251–268. 10.1046/j.1365-313X.2003.01959.x
38
BellionM.CourbotM.JacobC.GuinetF.BlaudezD.ChalotM. (2007). Metal induction of a Paxillus involutus metallothionein and its heterologous expression in Hebeloma cylindrosporum. New Phytol.174, 151–158. 10.1111/j.1469-8137.2007.01973.x
39
BelokobylskyA. I.GinturiE. I.KuchavaN. E.KirkesaliE. I.MosulishviliL. M.FrontasyevaM. V.et al. (2004). Accumulation of selenium and chromium in the growth dynamics of Spirulina platensis. J. Radioanal. Nucl. Chem.259, 65–68. 10.1023/B:JRNC.0000015807.53132.c0
40
BelouchiA.KwanT.GrosP. (1997). Cloning and characterization of the OsNramp family from Oryza sativa, a new family of membrane proteins possibly implicated in the transport of metal ions. Plant Mol. Biol.33, 1085–1092. 10.1023/A:1005723304911
41
BernalM. P.McGrathS. P.MillerA. J.BakerA. J. M. (1994). Comparison of the chemical changes in the rhizosphere of the nickel hyperaccumulator Alyssum murale with the non-accumulator Raphanus sativus. Plant Soil64, 251–259. 10.1007/BF00010077
42
BharwanaS. A.AliS.FarooqM. A.IqbalN.AbbasF.AhmadM. S. A. (2013). Alleviation of lead toxicity by silicon is related to elevated photosynthesis, antioxidant enzymes suppressed lead uptake and oxidative stress in cotton. J. Bioremed. Biodeg.4, 4. 10.4172/2155-6199.1000187
43
BlanvillainR.WeiS.WeiP.KimJ. H.OwD. W. (2011). Stress tolerance to stress escape in plants: role of the OXS2 zinc-finger transcription factor family. EMBO J.30, 3812–3822. 10.1038/emboj.2011.270
44
BlaudezD.KohlerA.MartinF.SandersD.ChalotM. (2003). Poplar metal tolerance protein 1 (MTP1) confers zinc tolerance and is an oligomeric vacuolar zinc transporter with an essential leucine zipper motif. Plant Cell15, 2911–2928. 10.1105/tpc.017541
45
BobrowiczP.WysockiR.OwsianikG.GoffeauA.UlaszewskiS. (1997). Isolation of three contiguous genes, ACR1, ACR2 and ACR3, involved in resistance to arsenic compounds in the yeast Saccharomyces cerevisiae. Yeast13, 819–828.
46
BonaE.MarsanoF.CavalettoM.BertaG. (2007). Proteomic characterization of copper stress response in Cannabis sativa roots. Proteomics7, 1121–1130. 10.1002/pmic.200600712
47
BorsaniO.ValpuestaV.BotellaM. A. (2001). Evidence for a role of salicylic acid in the oxidativedamage generated by NaCl and osmotic stress in Arabidopsis seedlings. Plant Physiol.126, 1024–1030. 10.1104/pp.126.3.1024
48
BriatJ. F.CurieC.GaymardF. (2007). Iron utilization and metabolism in plants. Curr. Opin. Plant Biol.10, 276–282. 10.1016/j.pbi.2007.04.003
49
Buendía-GonzálezL.Orozco-VillafuerteJ.Cruz-SosaF.Barrera-DíazC. E.Vernon-CarterE. J. (2010). Prosopis laevigata a potential chromium (VI) and cadmium (II) hyperaccumulator desert plant. Biores. Technol.101, 5862–5867. 10.1016/j.biortech.2010.03.027
50
BurlóF.GuijarroI.Carbonell-BarrachinaA. A.VlaeroD.Martínez-SánchezF. (1999). Arsenic species: effects on and accumulation by tomato plants. J. Agric. Food Chem.47, 1247–1253. 10.1021/jf9806560
51
CailleN.ZhaoF. J.McGrathS. P. (2005). Comparison of root absorption, translocation and tolerance of arsenic in the hyperaccumulator Pteris vittata and the nonhyperaccumulator Pteris tremula. New Phytol.165, 755–761. 10.1111/j.1469-8137.2004.01239.x
52
CailliatteR.LapeyreB.BriatJ. F.MariS.CurieC. (2009). The NRAMP6 metal transporter contributes to cadmium toxicity. Biochem. J.422, 217–228. 10.1042/BJ20090655
53
CailliatteR.SchikoraA.BriatJ. F.MariS.CurieC. (2010). High-affinity manganese uptake by the metal transporter NRAMP1 is essential for Arabidopsis growth in low manganese conditions. Plant Cell22, 904–917. 10.1105/tpc.109.073023
54
CallahanD. L.BakerA. J. M.KolevS. D.WeddA. G. (2006). Metal ion ligands in hyperaccumulating plants. J. Biol. Inorg. Chem.11, 2–12. 10.1007/s00775-005-0056-7
55
CarbonellA. A.AarabiM. A.DeLauneR. D.GambrellR. P.PatrickW. H.Jr. (1998). Arsenic in wetland vegetation: availability, phytotoxicity, uptake and effects on plant growth and nutrition. Sci. Total Environ. 217, 189–199. 10.1016/S0048-9697(98)00195-8
56
Carbonell-BarrachinaA. A.Burló -CarbonellF.Mataix-BeneytoJ. (1997). Effect of sodium arsenite and sodium chloride on bean plant nutrition (macronutrients). J. Plant Nutr.20, 1617–1633. 10.1080/01904169709365361
57
Carbonell-BarrachinaA.Burló -CarbonellF.Mataix-BeneytoJ. (1994). Effect of arsenite on the concentration of micronutrients in tomato plants grown in hydroponic culture. J. Plant Nutr.17, 1887–1903. 10.1080/01904169409364853
58
CastrilloG.Sánchez-BermejoE.de LorenzoL.CrevillénP.Fraile-EscancianoA.TcM.et al. (2013). WRKY6 Transcription factor restricts arsenate uptake and transposon activation in Arabidopsis. Plant Cell25, 2944–2957. 10.1105/tpc.113.114009
59
CataldoD. A.GarlandT. R.WildungR. E. (1983). Cadmium uptake kinetics in intact soybean plants. Plant Physiol. 73, 844–848. 10.1104/pp.73.3.844
60
CataldoD. A.McFaddenK. M.GarlandT. R.WildungR. E. (1988). Organic constituents and complexation of nickel(II), iron(III), cadmium(II), and plutonium(IV) in soybean xylem exudates. Plant Physiol.86, 734–739. 10.1104/pp.86.3.734
61
ChaffeiC.GouiaH.GhorbelM. H. (2003). Nitrogen metabolism in tomato plants under cadmium stress. J. Plant Nutr. 26, 1617–1634. 10.1081/PLN-120022372
62
ChaoD. Y.ChenY.ChenJ.ShiS.ChenZ.WangC.et al. (2014). Genome-wide association mapping identifies a new arsenate reductase enzyme critical for limiting arsenic accumulation in plants. PLoS Biol. 12:e1002009. 10.1371/journal.pbio.1002009
63
ChaoD.-Y.SilvaA.BaxterI.HuangY. S.NordborgM.DankuJ.et al. (2012). Genome-wide association studies identify heavy metal ATPase3 as the primary determinant of natural variation in leaf cadmium in Arabidopsis thaliana. PLoS Biol. 8:e1002923. 10.1371/journal.pgen.1002923
64
ChaurasiaN.MishraY.RaiL. C. (2008). Cloning expression and analysis of phytochelatin synthase (pcs) gene from Anabaena sp. PCC 7120 offering multiple stress tolerance in Escherichia coli. Biochem. Biophys. Res. Commun.376, 225–230. 10.1016/j.bbrc.2008.08.129
65
ChenJ.ZhouJ.GoldsbroughP. B. (1997). Characterization of phytochelatin synthase from tomato. Physiol. Plant.101, 165–172. 10.1111/j.1399-3054.1997.tb01833.x
66
ChenY. A.ChiW. C.HuangT. L.LinC. Y.Quynh NguyehT. T.HsuingY. C.et al. (2012a). Mercury-induced biochemical and proteomic changes in rice roots. Plant Physiol. Biochem.55, 23–32. 10.1016/j.plaphy.2012.03.008
67
ChenZ.PanY.WangS.DingY.YangW.ZhuC. (2012b). Overexpression of a protein disulfide isomerase-like protein from Methanothermobacter thermoautotrophicum enhances mercury tolerance in transgenic rice. Plant Sci.197, 10–20. 10.1016/j.plantsci.2012.08.005
68
ChiangC. M.ChenS. P.ChenL. F. O.ChiangM. C.ChienH. L.LinK. H. (2013). Expression of the broccoli catalase gene (BoCAT) enhances heat tolerance in transgenic Arabidopsis. J. Plant Biochem. Biotechnol.23, 266–277. 10.1007/s13562-013-0210-1
69
ChoS. H.HoangQ.PheeJ. W.KimY. Y.ShinH. Y.ShinJ. S. (2007). Modified suppression subtractive hybridization identifies an AP2-containing protein involved in metal responses in Physcomitrella patens. Mol. Cells23, 100–107.
70
Chong-qingW.TaoW.PingM.Zi-chaoL.LingY. (2013). Quantitative trait loci for mercury tolerance in rice seedlings. Rice Sci.20, 238–242. 10.1016/S1672-6308(13)60124-9
71
ChouT. S.ChaoY. Y.HuangW. D.HongC. Y.KaoC. H. (2011). Effect of magnesium deficiency on antioxidant status and cadmium toxicity in rice seedlings. J. Plant Physiol.168, 1021–1030. 10.1016/j.jplph.2010.12.004
72
ChoudharyS. P.KanwarM.BhardwajR.YuJ. Q.TranL. S. P. (2012b). Chromium stress mitigation by polyamine-brassinosteroid application involves phytohormonal and physiological strategies in Raphanus sativus L. PLoS ONE7:e33210. 10.1371/journal.pone.0033210
73
ChoudharyS. P.OralH. V.BhardwajR.YuJ. Q.TranL. S. P. (2012a). Interaction of brassinosteroids and polyamines enhances copper stress tolerance in Raphanus sativus. J. Exp. Bot.63, 5659–5675. 10.1093/jxb/ers219
74
CobbettC. S. (2000). Phytochelatin biosynthesis and function in heavy-metal detoxification. Curr. Opin. Plant Biol.3, 211–216. 10.1016/S1369-5266(00)00066-2
75
CobbettC.GoldsbroughP. (2002). Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu. Rev. Plant Biol.53, 159–182. 10.1146/annurev.arplant.53.100301.135154
76
CollinV. C.EymeryF.GentyB.ReyP.HavauxM. (2008). Vitamin E is essential for the tolerance of Arabidopsis thaliana to metal induced oxidative stress. Plant Cell Environ.31, 244–257. 10.1111/j.1365-3040.2007.01755.x
77
CreissenG.EdwardsE. A.EnardC.WellburnA.MullineauxP. (1992). Molecular characterisation of glutathione reductase cDNAs from pea (Pisum sativum L.). Plant J.2, 129–131.
78
CuiY.ZhangX.ZhuY. (2008). Does copper reduce cadmium uptake by different rice genotypes?J. Environ. Sci. 20, 332–338. 10.1016/S1001-0742(08)60052-2
79
CuiF.LiuL.ZhaoQ.ZhangZ.LiQ.LinB.et al. (2012a). Arabidopsis ubiquitin conjugase UBC32 is an ERAD component that functions in brassinosteroid-mediated salt stress tolerance. Plant Cell24, 233–244. 10.1105/tpc.111.093062
80
CuiW.LiL.GaoZ.WuH.XieY.ShenW. (2012b). Haem oxygenase-1 is involved in salicylic acid-induced alleviation of oxidative stress due to cadmium stress in Medicago sativa. J. Exp. Bot.63, 5521–5534. 10.1093/jxb/ers201
81
CurieC.AlonsoJ. M.Le JeanM.EckerJ. R.BriatJ. F. (2000). Involvement of NRAMP1 from Arabidopsis thaliana in iron transport. Biochem. J.347, 749–755. 10.1042/bj3470749
82
DalcorsoG.FasaniE.FuriniA. (2013b). Recent advances in the analysis of metal hyperaccumulation and hypertolerance in plants using proteomics. Front. Plant Sci.4:280. 10.3389/fpls.2013.00280
83
DalCorsoG.ManaraA.FuriniA. (2013a). An overview of heavy metal challenge in plants: from roots to shoots. Metallomics5, 1117–1132. 10.1039/c3mt00038a
84
DavidsonJ. F.SchiestlR. H. (2001). Mitochondrial respiratory electron carriers are involved in oxidative stress during heat stress in Saccharomyces cerevisiae. Mol. Cell. Biol.21, 8483–8489. 10.1128/MCB.21.24.8483-8489.2001
85
DavletovaS.RizhskyL.LiangH.ShengqiangZ.OliverD. J.CoutuJ.et al. (2005). Cytosolic ascorbate peroxidase 1 is a central component of the reactive oxygen gene network of Arabidopsis. Plant Cell17, 268–281. 10.1105/tpc.104.026971
86
DelhaizeE.GruberB. D.PittmanJ. K.WhiteR. G.LeungH.MiaoY.et al. (2007). A role for the AtMTP11 gene of Arabidopsis in manganese transport and tolerance. Plant J.51, 198–210. 10.1111/j.1365-313X.2007.03138.x
87
DelhaizeE.KataokaT.HebbD. M.WhiteR. G.RyanP. R. (2003). Genes encoding proteins of the cation diffusion facilitator family that confer manganese tolerance. Plant Cell15, 1131–1142. 10.1105/tpc.009134
88
Desbrosses-FonrougeA. G.VoigtK.SchröderA.ArrivaultS.ThomineS.KrämerU. (2005). Arabidopsis thaliana MTP1 is a Zn transporter in the vacuolar membrane which mediates Zn detoxification and drives leaf Zn accumulation. FEBS Lett.579, 4165–4174. 10.1016/j.febslet.2005.06.046
89
DiázJ.BernalA.Po MarF.MerinoF. (2001). Induction of shikimate dehydrogenase and peroxidase in pepper (Capsicum annum L.) seedlings in response to copper stress and its relation to lignification. Plant Sci. 161, 179. 10.1016/S0168-9452(01)00410-1
90
DiviU. K.KrishnaP. (2009). Brassinosteroid: a biotechnological target for enhancing crop yield and stress tolerance. New Biotechnol.26, 131–136. 10.1016/j.nbt.2009.07.006
91
DixitP.MukherjeeP. K.RamachandranV.EapenS. (2011). Glutathione transferase from Trichoderma virens enhances cadmium tolerance without enhancing its accumulation in transgenic Nicotiana tabacum. PLoS ONE6:e16360. 10.1371/journal.pone.0016360
92
DixonW. J.InouyeC.KarinM.TulliusT. D. (1996). CUP2 binds in a bipartite manner to upstream activation sequence c in the promoter of the yeast copper metallothionein gene. J. Biol. Inorg. Chem.1, 451–459. 10.1007/s007750050078
93
DonchevaS.AmeńosM.PoschenriederC.BarceĺoJ. (2005). Root cell patterning: a primary target for aluminium toxicity in maize. J. Exp. Bot.56, 1213–1220. 10.1093/jxb/eri115
94
DongJ.WuF. B.ZhangG. P. (2005). Effect of cadmium on growth and photosynthesis of tomato seedlings. J. Zhejiang Univ. Sci. B6, 974–980. 10.1631/jzus.2005.B0974
95
DrägerD. B.Desbrosses-FonrougeA. G.KrachC.ChardonnensA. N.MeyerR. C.Saumitou-LapradeP.et al. (2004). Two genes encoding Arabidopsis halleri MTP1 metal transport proteins co-segregate with zinc tolerance and account for high MTP1 transcript levels. Plant J.39, 425–439. 10.1111/j.1365-313X.2004.02143.x
96
Dragišić MaksimovićJ.MojovićM.MaksimovićV.RömheldV.NikolicM. (2012). Silicon ameliorates manganese toxicity in cucumber by decreasing hydroxyl radical accumulation in the leaf apoplast. J. Exp. Bot.63, 2411–2420. 10.1093/jxb/err359
97
DuanG.-L.ZhouY.TongY.-P.MukhopadhyayR.RosenB. P.ZhuY.-G. (2007). A CDC25 homologue from rice functions as an arsenate reductase. New Phytol. 174, 311–321. 10.1111/j.1469-8137.2007.02009.x
98
EapenS.D'souzaS. F. (2005). Prospects of genetic engineering of plants for phytoremediation of toxic metal. Biotechnol. Adv.23, 97–114. 10.1016/j.biotechadv.2004.10.001
99
EideD.BroderiusM.FettJ.GuerinotM. L. (1996). A novel iron-regulated metal transporter from plants identified by functional expression in yeast. Proc. Natl. Acad. Sci. U.S.A.93, 5624–5628. 10.1073/pnas.93.11.5624
100
ElbazB.Shoshani-KnaaniN.David-AssaelO.Mizrachy-DagriT.MizrahiK.SaulH.et al. (2006). High expression in leaves of the zinc hyperaccumulator Arabidopsis halleri of AhMHX, a homolog of an Arabidopsis thaliana vacuolar metal/protonexchanger. Plant Cell Environ.29, 1179–1190. 10.1111/j.1365-3040.2006.01500.x
101
EleftheriouE. P.AdamakisI. D.MelissaP. (2012). Effects of hexavalent chromium on microtubule organization, ER distribution and callose deposition in root tip cells of Allium cepa L. Protoplasma249, 401–416. 10.1007/s00709-011-0292-3
102
EllisD. R.GumaeliusL.IndrioloE.PickeringI. J.BanksJ. A.SaltD. E. (2006). A novel arsenate reductase from the arsenic hyperaccumulating fern Pteris vittata. Plant Physiol. 141, 1544–1554. 10.1104/pp.106.084079
103
EpsteinE. (1999). Silicon. Annu. Rev. Plant Physiol. Plant Mol. Biol.50, 641–664. 10.1146/annurev.arplant.50.1.641
104
ErnstW. H. O. (2006). Evolution of metal tolerance in higher plants. For. Snow Landsc. Res.80, 251–274. Available online at: http://www.wsl.ch/dienstleistungen/publikationen/pdf/7764.pdf
105
ErnstW. H. O.KraussG. J.VerkleijJ. A. C.WesenbergD. (2008). Interaction of heavy metal with the sulphur metabolism in angiosperms from an ecological point of view. Plant Cell Environ.31, 123–143. 10.1111/j.1365-3040.2007.01746.x
106
EspositoS.SorboS.ConteB.BasileA. (2012). Effects of heavy metal on ultrastructure and HSP70s induction in the aquatic moss Leptodictyum riparium Hedw. Int. J. Phytoremediation14, 443–455. 10.1080/15226514.2011.620904
107
FahrM.LaplazeL.BendaouN.HocherV.MzibriM. E.BoguszD.et al. (2013). Effect of lead on root growth. Front. Plant Sci.4:175. 10.3389/fpls.2013.00175
108
FAO (2012). Statistical Yearbook Viale delle Terme di Caracalla. Rome.
109
FariasJ. G.AntesF. L. G.NunesP. A. A.NunesS. T.SchaichG.RossatoL. V.et al. (2013). Effects of excess copper in vineyard soils on the mineral nutrition of potato genotypes. Food Energy Security2, 49–69. 10.1002/fes3.16
110
FariduddinQ.YusufM.HayatS.AhmadA. (2009). Effect of 28-homobrassinolide on antioxidant capacity and photosynthesis in Brassica juncea plants exposed to different levels of copper. Environ. Exp. Bot.66, 418–424. 10.1016/j.envexpbot.2009.05.001
111
FarzadfarS.ZarinkamarF.Modarres-SanavyS. A. M.HojatiM. (2013). Exogenously applied calcium alleviates cadmium toxicity in Matricaria chamomilla L. Plants Environ. Sci. Pollut. Res. 20, 1413–1422. 10.1007/s11356-012-1181-9
112
FengR. W.WeiC. Y. (2012). Antioxidative mechanisms on selenium accumulation in Pteris vittata L., a potential selenium phytoremediation plant. Plant Soil Environ.58, 105–110. Available online at: http://www.agriculturejournals.cz/publicFiles/61470.pdf
113
FengR. W.WeiC. Y.TuS. X.TangS. R.WuF. C. (2011). Detoxification of antimony by selenium and their interaction in paddy rice under hydroponic conditions. Microchem. J.97, 57–61. 10.1016/j.microc.2010.06.003
114
FerrettiM.GhisiR.MerloL.Dalla VecchiaF.PasseraC. (1993). Effect of cadmium on photosynthesis and enzymes of photosynthetic sulfate and nitrate assimilation pathways in maize (Zea mays L.). Photosynthetica29, 49–54.
115
FidalgoF.AzenhaM.SilvaA. F.de SousaA.SantiagoA.FerrazP.et al. (2013). Copper-induced stress in Solanum nigrum L. and antioxidant defense system responses. Food Energy Security2, 70–80. 10.1002/fes3.20
116
FilekM.KeskinenR.HartikainenH.SzarejkoI.JaniakA.MiszalskiZ.et al. (2008). The protective role of selenium in rape seedlings subjected to cadmium stress. J. Plant Physiol.165, 833–844. 10.1016/j.jplph.2007.06.006
117
FontesR. L. S.CoxF. R. (1998). Zinc toxicity in soybean grown at high iron concentration in nutrient solution. J. Plant Nutr.21, 1723–1730. 10.1080/01904169809365517
118
ForemanJ.DemidchikV.BothwellJ. H.MylonaP.MiedemaH.TorresM. A.et al. (2003). Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature422, 442–446. 10.1038/nature01485
119
FoyerC. H.HalliwellB. (1976). Presence of glutathione and glutathione reductase in chloroplasts: a proposed role in ascorbic acid metabolism. Planta133, 21–25. 10.1007/BF00386001
120
FoyerC. H.Lopez-DelgadoH.DatJ. F.ScottI. M. (1997). Hydrogen peroxide and glutathione-associated mechanisms of acclamatory stress tolerance and signalling. Physiol. Plant.100, 241–254. 10.1111/j.1399-3054.1997.tb04780.x
121
FreemanJ. L.SaltD. E. (2007). The metal tolerance profile of Thlaspi goesingense is mimicked in Arabidopsis thaliana heterologously expressing serine acetyl-transferase. BMC Plant Biol.7:63. 10.1186/1471-2229-7-63
122
FreemanJ. L.GarciaD.KimD.HopfA.SaltD. E. (2005a). Constitutively elevated salicylic acid signals glutathione-mediated nickel tolerance in Thlaspi nickel hyperaccumulators. Plant Physiol.137, 1082–1091. 10.1104/pp.104.055293
123
FreemanJ. L.PersansM. W.NiemanK.AlbrechtC.PeerW.PickeringI. J.et al. (2004). Increased glutathione biosynthesis plays a role in nickel tolerance in Thlaspi nickel hyperaccumulators. Plant Cell16, 2176–2191. 10.1105/tpc.104.023036
124
FreemanJ. L.PersansM. W.NiemanK.SaltD. E. (2005b). Nickel and cobalt resistance engineered in Escherichia coli by overexpression of serine acetyltransferase from the nickel hyperaccumulator plant Thlaspi goesingense. Appl. Environ. Microbiol.71, 8627–8633. 10.1128/AEM.71.12.8627-8633.2005
125
FrérotH.FauconM. P.WillemsG.GodéC.CourseauxA.DarracqA.et al. (2010). Genetic architecture of zinc hyperaccumulation in Arabidopsis halleri: the essential role of QTLx environment interactions. New Phytol.187, 355–367. 10.1111/j.1469-8137.2010.03295.x
126
FridovichI. (1989). Superoxide dismutases: an adaptation to paramagnetic gas. J. Biol. Chem.264, 7761–7764.
127
FührsH.BehrensC.GallienS.HeintzD.Van DorsselaerA.BraunH. P.et al. (2010). Physiological and proteomic characterization of manganese sensitivity and tolerance in rice (Oryza sativa) in comparison with barley (Hordeum vulgare). Ann. Bot.105, 1129–1140. 10.1093/aob/mcq046
128
FührsH.HartwigM.MolinaL. E.HeintzD.Van DorsselaerA.BraunH. P.et al. (2008). Early manganese-toxicity response in Vigna unguiculata L. a proteomic and transcriptomic study. Proteomics8, 149–159. 10.1002/pmic.200700478
129
FukaoY.FerjaniA.TomiokaR.NagasakiN.KurataR.NishimoriY.et al. (2011). iTRAQ analysis reveals mechanisms of growth defects due to excess zinc in Arabidopsis. Plant Physiol.155, 1893–1907. 10.1104/pp.110.169730
130
FürstP.HuS.HackettR.HamerD. (1988). Copper activates metallothionein gene transcription by altering the conformation of a specific DNA binding protein. Cell55, 705–717. 10.1016/0092-8674(88)90229-2
131
GaleasM. L.ZhangL. H.FreemanJ. L.WegnerM.Pilon-SmitsE. A. H. (2007). Seasonal fluctuations of selenium and sulphur accumulation in selenium hyperaccumulators and related nonaccumulators. New Phytol.173, 517–525. 10.1111/j.1469-8137.2006.01943.x
132
GangwarS.SinghV. P. (2011). Indole acetic acid differently changes growth and nitrogen metabolism in Pisum sativum L. seedlings under chromium (VI) phytotoxicity: implication of oxidative stress. Sci. Hortic.129, 321–328. 10.1016/j.scienta.2011.03.026
133
GangwarS.SinghV. P.PrasadS. M.MauryaJ. N. (2010). Modulation of manganese toxicity in Pisum sativum L. seedlings by kinetin. Sci. Hortic.126, 467–474. 10.1016/j.scienta.2010.08.013
134
GangwarS.SinghV. P.SrivastavaP. K.MauryaJ. N. (2011). Modification of chromium (VI) phytotoxicity by exogenous gibberellic acid application in Pisum sativum (L.) seedlings. Acta Physiol. Plant.33, 1385–1397. 10.1007/s11738-010-0672-x
135
GaoJ.SunL.YangX.LiuJ. X. (2013). Transcriptomic analysis of cadmium stress response in the heavy metal hyperaccumulator Sedum alfredii Hance. PLoS ONE8:e64643. 10.1371/journal.pone.0064643
136
GarzónT.GunséB.MorenoA. R.TomosA. D.BarcelóJ.PoschenriederC. (2011). Aluminium-induced alteration of ion homeostasis in root tip vacuoles of two maize varieties differing in Al tolerance. Plant Sci.180, 709–715. 10.1016/j.plantsci.2011.01.022
137
GhoshM.ShenJ.RosenB. P. (1999). Pathways of As(III) detoxification in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. U.S.A. 96, 5001–5006. 10.1073/pnas.96.9.5001
138
GichnerT.PatkovaZ.SzakovaJ.DemnerovaK. (2004). Cadmium induces DNA damages in tobacco roots, but no DNA damage, somatic mutations orhomologous recombinations in tobacco leaves. Mutat. Res. Genet. Toxicol. Environ. Mut.559, 49–57. 10.1016/j.mrgentox.2003.12.008
139
GillS. S.HasanuzzamanM.NaharK.MacoveiA.TutejaN. (2013). Importance of nitric oxide in cadmium stress tolerance in crop plants. Plant Physiol. Biochem.63, 254–261. 10.1016/j.plaphy.2012.12.001
140
GomesM. M. A. (2011). Physiological effects related to brassinosteroid application in plants, in Brassinosteroids: A Class of Plant Hormone, eds HayatS.AhmadA. (Dordrecht: Springer), 193–242. 10.1007/978-94-007-0189-2_7
141
GrennanA. K. (2009). Identification of genes involved in metal transport in plants. Plant Physiol. 149, 1623–1624. 10.1104/pp.109.900287
142
GuanZ.ChaiT.ZhangY.XuJ.WeiW. (2009). Enhancement of Cd tolerance in transgenic tobacco plants overexpressing a Cd-induced catalase cDNA. Chemosphere76, 623–630. 10.1016/j.chemosphere.2009.04.047
143
GuoB.LiangY.ZhuY. (2009). Does salicylic acid regulate antioxidant defense system, cell death, cadmium uptake and partitioning to acquire cadmium tolerance in rice?J. Plant Physiol.166, 20–31. 10.1016/j.jplph.2008.01.002
144
GuoJ.DaiX.XuW.MaM. (2008a). Overexpressing GSHI and AsPCSI simultaneously increases the tolerance and accumulation of cadmium and arsenic in Arabidopsis thaliana. Chemosphere72, 1020–1026. 10.1016/j.chemosphere.2008.04.018
145
GuoW. J.MeetamM.GoldsbroughP. (2008b). Examining the specific contributions of individual Arabidopsis metallothioneins to copper distribution and metal tolerance. Plant Physiol.164, 1697–1706. 10.1104/pp.108.115782
146
GustinJ. L.ZanisM. J.SaltD. E. (2011). Structure and evolution of the plant cation diffusion facilitator family of ion transporters. BMC Evol. Biol.11:76. 10.1186/1471-2148-11-76
147
GygiS. P.RochonY.FranzaB. R.AebersoldR. (1999). Correlation between protein and mRNA abundance in yeast. Mol. Cell. Biol. 19, 1720–1730. 10.1128/MCB.19.3.1720
148
Haag-KerwerA.SchäferH. J.HeissS.WalterC.RauschT. (1999). Cadmium exposure in Brassica juncea causes a decline in transpiration rate and leaf expansion without effect on photosynthesis. J. Exp. Bot.50, 1827–1835. 10.1093/jxb/50.341.1827
149
HajduchM.RakwalR.AgrawalG. K.YonekuraM.PretovaA. (2001). High-resolution two-dimensional electrophoresis separation of proteins from metal-stressed rice (Oryza sativa L.) leaves: drastic reductions/fragmentation of ribulose-1,5-bisphosphate carboxylase/oxygenase and induction of stress-related proteins. Electrophoresis22, 2824–2831. 10.1002/1522-2683(200108)22:13<2824::AID-ELPS2824>3.0.CO;2-C
150
HallJ. L.WilliamsL. E. (2003). Transition metal transporters in plants. J. Exp. Bot.54, 2601–2613. 10.1093/jxb/erg303
151
HalliwellB. (2006). Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol.141, 312–322. 10.1104/pp.106.077073
152
HamayunM.KhanS. A.KhanA. L.ShinJ. H.AhmadB.ShinD. H. (2010). Exogenous gibberellic acid reprograms soybean to higher growth and salt stress tolerance. J. Agric. Food Chem.58, 7226–7232. 10.1021/jf101221t
153
HamerD. H. (1986). Metallothionein. Annu. Rev. Biochem.55, 913–951. 10.1146/annurev.bi.55.070186.004405
154
HaneyC. J.GrassG.FrankeS.RensingC. (2005). New developments in the understanding of the cation diffusion facilitator family. J. Ind. Microbiol. Biotechnol.32, 215–226. 10.1007/s10295-005-0224-3
155
HaoJ.YinY.FeiS. Z. (2013). Brassinosteroid signaling network: implications on yield and stress tolerance. Plant Cell Rep.32, 1017–1030. 10.1007/s00299-013-1438-x
156
HarrisonP. M.ArosioP. (1996). The ferritins: molecular properties, iron storage function and cellular regulation. Biochim. Biophys. Acta275, 161–203. 10.1016/0005-2728(96)00022-9
157
HartJ. J.WelchR. M.NorvellW. A.ClarkeJ. M.KochianL. V. (2005). Zinc effects on cadmium accumulation and partitioning in near isogenic lines of durum wheat that differ in grain cadmium concentration. New Phystol.167, 391–401. 10.1111/j.1469-8137.2005.01416.x
158
HassanM. J.ZhangG.WuF.WeiK.ChenZ. (2005). Zinc alleviates growth inhibition and oxidative stress caused by cadmium in rice. J. Plant Nutr. Soil Sci.168, 255–261. 10.1002/jpln.200420403
159
HayatQ.HayatS.IrfanM.AhmadA. (2010). Effect of exogenous salicylic acid under changing environment: a review. Environ. Exp. Bot.68, 14–25. 10.1016/j.envexpbot.2009.08.005
160
HayatS.AliB.Aiman HasanS.AhmadA. (2007). Brassinosteroid enhanced the level of antioxidants under cadmium stress in Brassica juncea. Environ. Exp. Bot.60, 33–41. 10.1016/j.envexpbot.2006.06.002
161
HayatS.KhaliqueG.IrfanM.WaniA. S.TripathiB. N.AhmadA. (2012). Physiological changes induced by chromium stress in plants: an overview. Protoplasma249, 599–611. 10.1007/s00709-011-0331-0
162
HeH.ZhanJ.HeL.GuM. (2012). Nitric oxide signaling in aluminum stress in plants. Protoplasma249, 483–492. 10.1007/s00709-011-0310-5
163
HeP. P.LvX. Z.WangG. Y. (2004). Effects of Se and Zn supplementation on the antagonism against Pb and Cd in vegetables. Environ. Int.30, 167–172. 10.1016/S0160-4120(03)00167-3
164
HerbetteS.TaconnatL.HugouvieuxV.PietteL.MagnietteM. L.CuineS.et al. (2006). Genome-wide transcriptome profiling of the early cadmium response of Arabidopsis roots and shoots. Biochimie88, 1751–1765. 10.1016/j.biochi.2006.04.018
165
HerbikA.GiritchA.HorstmannC.BeckerR.BalzerH. J.BäumleinH.et al. (1996). Iron and copper nutrition-dependent changes in protein expression in a tomato wild type and the nicotianamine-free mutant chloronerva. Plant Physiol.111, 533–540. 10.1104/pp.111.2.533
166
HernándezL. E.GarateA.Carpena-RuizR. (1997). Effects of cadmium on the uptake, distribution and assimilation of nitrate in Pisum sativum. Plant Soil. 189, 97–106.
167
HernandezL. E.VillasanteC. O.Montero-PalmeroM. B.EscobarC.CarpenaR. O. (2012). Heavy metal perception in a microscale environment: a model system using high doses of pollutants, in Metal Toxicity in Plants: Perception, Signaling and Remediation, eds GuptaD. K.SandalioL. M. (Berlin; Heidelberg: Springer-Verlag), 23–37. 10.1007/978-3-642-22081-4_2
168
HirataK.TsujiN.MiyamotoK. (2005). Biosynthetic regulation of phytochelatins, heavy metal-binding peptides. J. Biosci. Bioeng.100, 593–599. 10.1263/jbb.100.593
169
HirschiK. D.KorenkovV. D.WilganowskiN. L.WagnerG. J. (2000). Expression of Arabidopsis CAX2 in tobacco. Altered metal accumulation and increased manganese tolerance. Plant Physiol.124, 125–133. 10.1104/pp.124.1.125
170
HisamatsuT.KoshiokaM.KubotaS.FujimeY.KingR. W.ManderL. N. (2000). The role of gibberellin in the control of growth and flowering in Matthiola incana. Physiol. Plant.109, 97–105. 10.1034/j.1399-3054.2000.100114.x
171
HorváthE.SzalaiG.JandaT. (2007). Induction of abiotic stress tolerance by salicylic acid signaling. J. Plant Growth Regul.26, 290–300. 10.1007/s00344-007-9017-4
172
HossainM. A.HasanuzzamanM.FujitaM. (2011). Coordinate induction of antioxidant defense and glyoxalase system by exogenous proline and glycinebetaineis correlated with salt tolerance in mung bean. Front. Agric. China5, 1–14. 10.1007/s11703-010-1070-2
173
HossainM. A.HossainM. D.RohmanM. M.da SilvaJ. A. T.FujitaM. (2012b). Onion major compounds (flavonoids, organosulfurs) and highly expressed glutathione-related enzymes: possible physiological interaction, gene cloning and abiotic stress response, in Onion Consumption and Health, eds AguirreC. B.JaramilloL. M. (New York, NY: Nova Science Publishers), 49–90.
174
HossainM. A.PiyatidaP.da SilvaJ. A. T.FujitaM. (2012a). Molecular mechanism of heavy metal toxicity and tolerance in plants: central role of glutathione in detoxification of reactive oxygen species and methylglyoxal and in heavy metal chelation. J. Bot.2012:872875. 10.1155/2012/872875
175
HossainZ.KomatsuS. (2013). Contribution of proteomic studies towards understanding plant heavy metal tress response. Front. Plant Sci.3:310. 10.3389/fpls.2012.00310
176
HossainZ.NouriM. Z.KomatsuS. (2012c). Plant cell organelle proteomics in response to abiotic stress. J. Proteome Res.11, 37–48. 10.1021/pr200863r
177
HowarthJ. R.Domínguez-SolísJ. R.Gutíerrez-AlcaláG.WrayJ. L.RomeroL. C.GotorC. (2003). The serine acetyltransferase gene family in Arabidopsis thaliana and the regulation of its expression by cadmium. Plant Mol. Biol.51, 589–598. 10.1023/A:1022349623951
178
HusainiY.RaiL. C. (1991). Studies on nitrogen and phosphorus metabolism and the photosynthetic electron transport system of Nostoc linckia under cadmium stress. J. Plant Physiol.138, 429–435. 10.1016/S0176-1617(11)80518-3
179
HussainD.HaydonM. J.WangY.WongE.ShersonS. M.YoungJ.et al. (2004). P-Type ATPase heavy metal transporters with roles in essential zinc homeostasis in Arabidopsis. Plant Cell16, 1327–1339. 10.1105/tpc.020487
180
ImranM. A.ChM. N.KhanR. M.AliZ.MahmoodT. (2013). Toxicity of arsenic (As) on seed germination of sunflower (Helianthus annuus L.). Int. J. Phys. Sci. 8, 840–847. 10.5897/IJPS2013.3894
181
IndrioloE.NaG.EllisD.SaltD. E.BanksJ. A. (2010). A vacuolar arsenite transporter necessary for arsenic tolerance in the arsenic hyperaccumulating fern Pteris vittata is missing in flowering plants. Plant Cell22, 2045–2057. 10.1105/tpc.109.069773
182
IngleR. A.SmithJ. A.SweetloveL. J. (2005). Responses to nickel in the proteome of the hyperaccumulator plant Alyssum lesbiacum. Biometal18, 627–641. 10.1007/s10534-005-2999-0
183
IqbalN.NazarR.KhanM. I. R.MasoodA.KhanN. A. (2011). Role of gibberellins in regulation of source-sink relations under optimal and limiting environmental conditions. Curr. Sci.100, 998–1007. Available online at: http://www.currentscience.ac.in/Volumes/100/07/0998.pdf
184
IsrarM.SahiS.DattaR.SarkarD. (2006). Bioaccumulation and physiological effects of mercury in Sesbania drummonii. Chemosphere65, 591–598. 10.1016/j.chemosphere.2006.02.016
185
IuchiS.KoyamaH.IuchiA.KobayashiY.KitabayashiS.KobayashiY.et al. (2007). Zinc finger protein STOP1 is critical for proton tolerance in Arabidopsis and coregulates a key gene in aluminum tolerance. Proc. Natl. Acad. Sci. U.S.A.104, 9900–9905. 10.1073/pnas.0700117104
186
JallohM. A.ChenJ.ZhenF.ZhangG. (2009). Effect of different N fertilizer forms on anti-oxidant capacity and grain yield of rice growing under Cd stress. J. Hazard. Mater.162, 1081–1085. 10.1016/j.jhazmat.2008.05.146
187
JaneckzoA.KoscielniakJ.PilipowiczM.Szarek-LukaszewskaG.SkoczowskiA. (2005). Protection of winter rape photosystem 2 by 24-epibrassinolide under cadmium stress. Photosynthetica43, 293–298. 10.1007/s11099-005-0048-4
188
Janicka-RussakM.KabałaK.BurzyńskiM.KłobusG. (2008). Response of plasma membrane H+-ATPase to heavy metal stress in Cucumis sativus roots. J. Exp. Bot.59, 3721–3728. 10.1093/jxb/ern219
189
JarupL. (2003). Hazards of heavy metal contamination. Br. Med. Bull.68, 167–182. 10.1093/bmb/ldg032
190
JewellM. C.CampbellB. C.GodwinI. D. (2010). Transgenic plants for abiotic stress resistance, in Transgenic Crop Plants, eds KoleC.MichlerC. H.AbbottA. G.HallT. C. (Berlin; Heidelberg: Springer-Verlag), 67–31. 10.1007/978-3-642-04812-8_2
191
JonakC.NakagamiH.HirtH. (2004). Heavy metal stress. Activation of distinct mitogen-activated protein kinase pathways by copper and cadmium. Plant Physiol.136, 3276–3283. 10.1104/pp.104.045724
192
JonakC.OkrészL.BögreL.HirtH. (2002). Complexity, cross talk and integration of plant MAP kinase signalling. Curr. Opin. Plant Biol.5, 415–424. 10.1016/S1369-5266(02)00285-6
193
JunM.FuH. Y.HongJ.WanX.YangC. S.HoC. T. (2003). Comparison ofantioxidant activities of isoflavones from kudzu root (PuerarialobataOhwi). J. Food Sci. 68, 2117–2122. 10.1111/j.1365-2621.2003.tb07029.x
194
KagaleS.DiviU. K.KrochkoJ. E.KellerW. A.KrishnaP. (2007). Brassinosteroid confers tolerance in Arabidopsis thaliana and Brassica napus to a range of abiotic stresses. Planta225, 353–364. 10.1007/s00425-006-0361-6
195
Kanoun-BouléM.VicenteJ. A.NabaisC.PrasadM. N. V.FreitasH. (2009). Ecophysiological tolerance of duckweeds exposed to copper. Aquat. Toxicol.91, 1–9. 10.1016/j.aquatox.2008.09.009
196
KavamuraV. N.EspositoE. (2010). Biotechnological strategies applied to the decontamination of soils polluted with heavy metal. Biotechnol. Adv.28, 61–69. 10.1016/j.biotechadv.2009.09.002
197
KenderešováL.StaňováA.PavlovkinJ.ĎurišováE.NadubinskáM.CiamporováM.et al. (2012). Early Zn2+-induced effects on membrane potential account for primary heavy metal susceptibility in tolerant and sensitive Arabidopsis species. Ann. Bot.110, 445–459. 10.1093/aob/mcs111
198
KerkebL.KrämerU. (2003). The role of free histidine in xylem loading of nickel in Alyssum lesbiacum and Brassica juncea. Plant Physiol.131, 716–724. 10.1104/pp102.010686
199
KeunenE.RemansT.BohlerS.VangronsveldJ.CuypersA. (2011). Metal-induced oxidative stress and plant mitochondria. Int. J. Mol. Sci.12, 6894–6918. 10.3390/ijms12106894
200
KhanA. L.LeeI. J. (2013). Endophytic Penicillium funiculosum LHL06 secretes gibberellin that reprograms Glycine max L. growth during copper stress. BMC Plant Biol.13:86. 10.1186/1471-2229-13-86
201
KhanN. A.AnjumN. A.NazarR.IqbalN. (2009). Increased activity of ATP-sulfurylase and increased contents of cysteine and glutathione reduce high cadmium- induced oxidative stress in mustard cultivar with high photosynthetic potential. Russ. J. Plant Physiol.56, 670–677. 10.1134/S1021443709050136
202
KhanN. A.Samiullah SinghS.NazarR. (2007). Activities of antioxidative enzymes, sulphur assimilation, photosynthetic activity and growth of wheat (Triticum aestivum) cultivars differing in yield potential under cadmium stress. J. Agron. Crop Sci.193, 435–444. 10.1111/j.1439-037X.2007.00272.x
203
KhattakR. A.PageA. L.ParkerD. R.BakhtarD. (1991). Accumulation and interactions of arsenic, selenium, molybdenum and phosphorus in Alfalfa. J. Environ. Qual.20, 165–168. 10.2134/jeq1991.00472425002000010026x
204
KhripachV.ZhabinskiiV.De GrootA. (2000). Twenty years of brassinosteroids: steroidal plant hormones warrant better crops for the XXI century. Ann. Bot.86, 441–447. 10.1006/anbo.2000.1227
205
KiefferP.DommesJ.HoffmannL.HausmanJ. F.RenautJ. (2008). Quantitative changes in protein expression of cadmium-exposed poplar plants. Proteomics8, 2514–2530. 10.1002/pmic.200701110
206
KikuiS.SasakiT.MaekawaM.MiyaoA.HirochikaH.MatsumotoH.et al. (2005). Physiological and genetic analyses of aluminium tolerance in rice, focusing on root growth during germination. J. Inorg. Biochem.99, 1837–1844. 10.1016/j.jinorgbio.2005.06.031
207
KimI. S.ShinS. Y.KimY. S.KimH. Y.YoonH. S. (2009). Expression of a glutathione reductase from Brassica rapa subsp. pekinensis enhanced cellular redox homeostasis by modulating antioxidant proteins in Escherichia coli. Mol. Cells28, 479–487. 10.1007/s10059-009-0168-y
208
KlapheckS.SchlunzS.BergmannL. (1995). Synthesis of phytochelatins and homo-phytochelatins in Pisum sativum L. Plant Physiol.107, 515–521.
209
KobaeY.UemuraT.SatoM. H.OhnishiM.MimuraT.NakagawaT.et al. (2004). Zinc transporter of Arabidopsis thaliana AtMTP1 is localized to vacuolar membranes and implicated in zinc homeostasis. Plant Cell Physiol.45, 1749–1758. 10.1093/pcp/pci015
210
KoprivovaA.NorthK. A.KoprivaS. (2008). Complex signaling network in regulation of adenosine 5′-phosphosulfate reductase by salt stress in Arabidopsis roots. Plant Physiol.146, 1408–1420. 10.1104/pp.107.113175
211
KosováK.VítámvásP.PrášilI. T.RenautJ. (2011). Plant proteome changes under abiotic stress- contribution of proteomics studies to understanding plant stress response. J. Proteomics74, 1301–1322. 10.1016/j.jprot.2011.02.006
212
KováčikJ.KlejdusB. (2008). Dynamics of phenolic acids and lignin accumulation in metal-treated Matricariachamomilla roots. Plant Cell Rep. 27, 605–615. 10.1007/s00299-007-0490-9
213
KováčikJ.KlejdusB.BačkorM. (2009). Phenolic metabolism of Matricariachamomilla plants exposed to nickel. J. Plant Physiol. 166, 1460–1464. 10.1016/j.jplph.2009.03.002
214
KrämerU. (2010). Metal hyperaccumulation in plants. Annu. Rev. Plant Biol.61, 517–534. 10.1146/annurev-arplant-042809-112156
215
KrämerU.Cotter-HowellsJ. D.CharnockJ. M.BakerA. J. M.SmithJ. A. C. (1996). Free histidine as a metal chelator in plants that accumulate nickel. Nature379, 635–638. 10.1038/379635a0
216
KrämerU.PickeringI. J.PrinceR. C.RaskinI.SaltD. E. (2000). Subcellular localization and speciation of nickel in hyperaccumulator and non-accumulator Thlaspi species. Plant Physiol.122, 1343–1354. 10.1104/pp.122.4.1343
217
KrämerU.TalkeI. N.HanikenneM. (2007). Transition metal transport. FEBS Lett.581, 2263–2272. 10.1016/j.febslet.2007.04.010
218
KrantevA.YordanovaR.JandaT.SzalaiG.PopovaL. (2008). Treatment with salicylic acid decreases the effect of cadmium on photosynthesis in maize plants. J. Plant Physiol.165, 920–931. 10.1016/j.jplph.2006.11.014
219
KrishnamurtiG. S. R.CieslinskiG.HuangP. M.Van ReesK. C. J. (1997). Kinetics of cadmium release from soils as influenced by organic acids: implication in cadmium availability. J. Environ. Qual.26, 271–277. 10.2134/jeq1997.00472425002600010038x
220
KumarA.PrasadM. N. V.SytarO. (2012). Lead toxicity, defense strategies and associated indicative biomarkers in Talinumtriangularegrown hydroponically. Chemosphere89, 1056–1165. 10.1016/j.chemosphere.2012.05.070
221
KuriakoseS. V.PrasadM. N. V. (2008). Cadmium stress affects seed germination and seedling growth in Sorghum bicolor (L.) Moench by changing the activities of hydrolyzing enzymes. Plant Growth Regul.54, 143–156. 10.1007/s10725-007-9237-4
222
KwakJ. M.NguyenV.SchroederJ. I. (2006). The role of reactive oxygen species in hormonal responses. Plant Physiol.141, 323–329. 10.1104/pp.106.079004
223
LanH. X.WangZ. F.WangQ. H.WangM. M.BaoY. M.HuangJ.et al. (2012). Characterization of a vacuolar zinc transporter OZT1 in rice (Oryza sativa L.). Mol. Biol. Rep.40, 1201–1210. 10.1007/s11033-012-2162-2
224
LandrieuI.da CostaM.De VeylderL.DewitteF.VandepoeleK.HassanS.et al. (2004a). A small CDC25 dual-specificity tyrosine-phosphatase isoform in Arabidopsis thaliana. Proc. Natl. Acad. Sci. U.S.A. 101, 13380–13385. 10.1073/pnas.0405248101
225
LandrieuI.HassanS.SautyM.DewitteF.WieruszeskiJ. M.InzéD.et al. (2004b). Characterization of the Arabidopsis thaliana Arath;CDC25 dualspecificity tyrosine phosphatase. Biochem. Biophys. Res. Commun. 322, 734–739. 10.1016/j.bbrc.2004.07.182
226
LanquarV.LelièvreF.BolteS.HamèsC.AlconC.NeumannD.et al. (2005). Mobilization of vacuolar iron by AtNRAMP3 and AtNRAMP4 is essential for seed germination on low iron. EMBO J.24, 4041–4051. 10.1038/sj.emboj.7600864
227
Lauer JúniorC. M.BonattoD.Mielniczki-PereiraA. A.SchuchA. Z.DiasJ. F.YoneamaM. L.et al. (2008). The Pmr1 protein, the major yeast Ca2+-ATPase in the grefecvolgi, regulates intracellular levels of the cadmium ion. FEMS Microbiol. Lett.285, 79–88. 10.1111/j.1574-6968.2008.01214.x
228
LavidN.SchwartzA.Yar DenO.Tel-OrE. (2001). The involvement of polyphenols and peroxidase acitivities in heavy metal accumulation by epidermal glands of water lily (Nymphaeceaea). Planta212, 323. 10.1007/s004250000400
229
LeaP. J.MiflinB. J. (2004). Glutamate synthase and the synthesis of glutamate in plants. Plant Physiol. Biochem.41, 555–564. 10.1016/S0981-9428(03)00060-3
230
Le MartretB.PoageM.ShielK.NugentG. D.DixP. J. (2011). Tobacco chloroplast transformants expressing genes encoding dehydroascorbate reductase, glutathione reductase, and glutathione-S-transferase, exhibit altered anti-oxidant metabolism and improved abiotic stress tolerance. Plant Biotechnol. J.9, 661–673. 10.1111/j.1467-7652.2011.00611.x
231
LeDucD. L.AbdelSamieM.Móntes-BayonM.WuC. P.ReisingerS. J.TerryN. (2006). Overexpressing both ATP sulfurylase and selenocysteine methyltransferase enhances selenium phytoremediation traits in Indian mustard. Environ. Poll.144, 70–76. 10.1016/j.envpol.2006.01.008
232
LeeH.JoJ.SonD. (1998). Molecular cloning and characterization of the gene encoding glutathione reductase in Brassica campestris. Biochim. Biophys. Acta1395, 309–314. 10.1016/S0167-4781(97)00198-X
233
LeeK.BaeD. W.KimS. H.HanH. J.LiuX.ParkH. C.et al. (2010). Comparative proteomic analysis of the short-term responses of rice roots and leaves to cadmium. J. Plant Physiol.167, 161–168. 10.1016/j.jplph.2009.09.006
234
LeeS. H.AhsanN.LeeK. W.KimD. H.LeeD. G.KwakS. S.et al. (2007). Simultaneous overexpression of both CuZn superoxide dismutase and ascorbate peroxidase in transgenic tall fescue plants confers increased tolerance to a wide range of abiotic stresses. J. Plant Physiol.164, 1626–1638. 10.1016/j.jplph.2007.01.003
235
LiL.HuangX.BorthakurD.NiH. (2012). Photosynthetic activity and antioxidative response of seagrass Thalassia hemprichii to trace metal stress. Acta Oceanol. Sin.31, 98–108. 10.1007/s13131-012-0210-3
236
LiP.ChenL.ZhouY.XiaX.ShiK.ChenZ.et al. (2013). Brassinosteroids-induced systemic stress tolerance was associated with increased transcripts of several defence-related genes in the phloem in Cucumis sativus. PLoS ONE8:e66582. 10.1371/journal.pone.0066582
237
LiW. X.ChenT. B.HuangZ. C.LeiM.LiaoX. Y. (2006). Effect of arsenic on chloroplast ultrastructure and calcium distribution in arsenic hyperaccumulator Pteris vittata L. Chemosphere62, 803–809. 10.1016/j.chemosphere.2005.04.055
238
LiY.DhankherO. P.CarreiraL.LeeD.ChenA.SchroederJ. I.et al. (2004). Overexpression of phytochelatin synthase in Arabidopsis leads to enhanced arsenic tolerance and cadmium hypersensitivity. Plant Cell Physiol.45, 1787–1797. 10.1093/pcp/pch202
239
LiangW. H.LiL.ZhangF.LiuY. X.LiM. M.ShiH. H.et al. (2013). Effects of abiotic stress, light, phytochromes and phytohormones on the expression of OsAQP, a rice aquaporin gene. Plant Growth Regul.69, 21–27. 10.1007/s10725-012-9743-x
240
Liang ZhuY.Pilon-SmitsE. A. H.JouaninL.TerryN. (1999). Overexpression of glutathione synthetase in Indian mustard enhances cadmium cccumulation and tolerance. Plant Physiol.119, 173–180.
241
LidonF. C.HenriquesF. S. (1991). Limiting step on photosynthesis of rice plants treated with varying copper levels. J. Plant Physiol. 138, 115–118.
242
LinC. C.KaoC. H. (2000). Effect of NaCl stress on H2O2 metabolism in rice leaves. J. Plant Growth Regul.30, 151–155. 10.1023/A:1006345126589
243
LinT.ZhuX.ZhangF.WanX. (2011). The detoxification effect of cadmium stress in Populus yunnanensis. Res. J. Bot.4, 13–19. 10.3923/brj.2011.13.19
244
LinY. F.LiangH. M.YangS. Y.BochA.ClemensS.ChenC. C.et al. (2009). Arabidopsis IRT3 is a zinc-regulated and plasma membrane localized zinc/iron transporter. New Phytol.182, 392–404. 10.1111/j.1469-8137.2009.02766.x
245
LinguaG.BonaE.TodeschiniV.CattaneoC.MarsanoF.BertaG.et al. (2012). Effects of heavy metal and arbuscular mycorrhiza on the leaf proteome of a selected Poplar clone: A time course analysis. PLoS ONE7:e38662. 10.1371/journal.pone.0038662
246
LiuD.JiangW.LiM. (1992). Effects of trivalent and hexavalent chromium on root growth and cell division of Allium cepa. Hereditas117, 23–29. 10.1111/j.1601-5223.1992.tb00003.x
247
LiuG. Y.ZhangY. X.ChaiT. Y. (2011). Phytochelatin synthase of Thlaspi caerulescens enhanced tolerance and accumulation of heavy metal when expressed in yeast and tobacco. Plant Cell Rep.30, 1067–1076. 10.1007/s00299-011-1013-2
248
LiuX.WuH.JiC.WeiL.ZhaoJ.YuJ. (2013). An integrated proteomic and metabolomic study on the chronic effects of mercury in Suaeda salsa under an environmentally relevant salinity. PLoS ONE8:e64041. 10.1371/journal.pone.0064041
249
LoefflerS.HochbergerA.GrillE.WinnackerE.-L.ZenkM.-H. (1989). Termination of the phytochelatin synthase reaction through sequestration of heavy metals by the reaction product. FEBS Lett.258, 42–46. 10.1016/0014-5793(89)81611-4
250
LongT. A.TsukagoshiH.BuschW.LahnerB.SaltD. E.BenfeyP. N. (2010). The bHLH transcription factor POPEYE regulates response to iron deficiency in Arabidopsis roots. Plant Cell22, 2219–2236. 10.1105/tpc.110.074096
251
López-MillánA. F.EllisD. R.GrusakM. A. (2004). Identification and characterization of several new members of the ZIP family of metal ion transporters in Medicagotruncatula. Plant Mol. Biol.54, 583–596. 10.1023/B:PLAN.0000038271.96019.aa
252
LuY. P.LiZ. S.ReaP. A. (1997). AtMRP1 gene of Arabidopsis encodes a glutathione S-conjugate pump: isolation and functional definition of a plant ATP-binding cassette transporter gene. Proc. Natl. Acad. Sci. U.S.A.94, 8243–8248. 10.1073/pnas.94.15.8243
253
LuY. P.LiZ. S.DrozdowiczY. M.HortensteinerS.MartinoiaE.ReaP. A. (1998). AtMRP2, an Arabidopsis ATP binding cassette transporter able to transport glutathione S-conjugates and chlorophyll catabolites: functional comparisons with AtMRP1. Plant Cell10, 267–282.
254
LushchakV. I.SemchukN. M. (2012). Tocopherol biosynthesis: chemistry, regulation and effects of environmental factors. Acta Physiol. Plant. 34, 1607–1628. 10.1007/s11738-012-0988-9
255
MaJ. F.YamajiN.MitaniN.XuX. Y.SuY. H.McGrathS. P.et al. (2008). Transporters of arsenite in rice and their role in arsenic accumulation in rice grain. Proc. Natl. Acad. Sci. U.S.A.105, 9931–9935. 10.1073/pnas.0802361105
256
MaL. Q.KomarK. M.TuC.ZhangW.CaiY.KennellyE. D. (2001). A fern that hyperaccumulates arsenic. Nature409, 579. 10.1038/35054664
257
MalevaM. G.NekrasovaG. F.BorisovaG. G.ChukinaN. V.UshakovaO. S. (2012). Effect of heavy metal on photosynthetic apparatus and antioxidant status of elodea. Russ. J. Plant Physiol.59, 190–197. 10.1134/S1021443712020069
258
ManavalanL. P.GuttikondaS. K.TranL.-S. P.NguyenH. T. (2009). Physiological and molecular approaches to improve drought resistance in soybean. Plant Cell Physiol.50, 1260–1276. 10.1093/pcp/pcp082
259
MarschnerH. (1995). Mineral Nutrition of Higher Plants. Boston, MA: Academic Press.
260
MäserP.ThomineS.SchroederJ. I.WardJ. M.HirschiK.SzeH.et al. (2001). Phylogenetic relationships within cation transporter families of Arabidopsis. Plant Physiol.126, 1646–1667. 10.1104/pp.126.4.1646
261
MasoodA.IqbalN.KhanN. A. (2012). Role of ethylene in alleviation of cadmium-induced photosynthetic capacity inhibition by sulfur in mustard. Plant Cell Environ.35, 524–533. 10.1111/j.1365-3040.2011.02432.x
262
MathysW. (1977). The role of malate, oxalate, and mustard oil glucosides in the evolution of zinc resistance in herbage plants. Physiol. Plant.40, 130–136. 10.1111/j.1399-3054.1977.tb01509.x
263
MatsuokaM. (2003). Gibberellin signaling: how do plant cells respond to GA signals?J. Plant Growth Regul.22, 123–125. 10.1007/s00344-003-0039-2
264
MatusikJ.BajdaT.ManeckiM. (2008). Immobilization of aqueous cadmium by addition of phosphates. J. Hazard. Mater.152, 1332–1339. 10.1016/j.jhazmat.2007.08.010
265
McLaughlinM. J.ParkerD. R.ClarkJ. M. (1999). Metal and micronutrients-food safety issues. Field Crops Res.60, 143–163. 10.1016/S0378-4290(98)00137-3
266
MehargA. A.Hartley-WhitakerJ. (2002). Arsenic uptake and metabolism in arsenic resistant and non resistant plant species. New Phytol.154, 29–43. 10.1046/j.1469-8137.2002.00363.x
267
MeisterA.AndersonM. E. (1983). Glutathione. Annu. Rev. Biochem.52, 711–760. 10.1146/annurev.bi.52.070183.003431
268
MengH.HuaS.ShamsiI. H.JilaniG.LiY.JiangL. (2009). Cadmium-induced stress on the seed germination and seedling growth of Brassica napus L., and its alleviation through exogenous plant growth regulators. Plant Growth Regul.58, 47–59. 10.1007/s10725-008-9351-y
269
MenguerP. K.FarthingE.PeastonK. A.RicachenevskyF. K.FettJ. P.WilliamsL. E. (2013). Functional analysis of the rice vacuolar zinc transporter OsMTP1. J. Exp. Bot.64, 2871–2883. 10.1093/jxb/ert136
270
MétrauxJ. P. (2002). Recent breakthroughs in the study of salicylic acid biosynthesis. Trends Plant. Sci.7, 332–334. 10.1016/S1360-1385(02)02313-0
271
MetwallyA.FinkemeierI.GeorgiM.DietzK. J. (2003). Salicylic acid alleviates the cadmium toxicity in barley seedlings. Plant Physiol.132, 272–281. 10.1104/pp.102.018457
272
MillerG.ShulaevV.MittlerR. (2008). Reactive oxygen signaling and abiotic stress. Physiol. Plant133, 481–489. 10.1111/j.1399-3054.2008.01090.x
273
MillerG.SuzukiN.Ciftci-YilmazS.MittlerR. (2010). Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant Cell Environ.33, 453–467. 10.1111/j.1365-3040.2009.02041.x
274
MillsR. F.FranciniA.da RochaP. S. C. F.BaccariniP. J.AylettM.KrijgerG. C.et al. (2005). The plant P1B-type ATPase AtHMA4 transports Zn and Cd and plays a role in detoxification of transition metals supplied at elevated levels. FEBS Lett.579, 783–791. 10.1016/j.febslet.2004.12.040
275
MilnerM. J.KochianL. V. (2008). Investigating heavy-metal hyperaccumulation using Thlaspi caerulescens as a model system. Ann. Bot.102, 3–13. 10.1093/aob/mcn063
276
MilnerM. J.SeamonJ.CraftE.KochianL. V. (2013). Transport properties of members of the ZIP family in plants and their role in Zn and Mn homeostasis. J. Exp. Bot.64, 369–381. 10.1093/jxb/ers315
277
MiransariM. (2011). Hyperaccumulators, arbuscular mycorrhizal fungi and stress of heavy metal. Biotechnol. Adv.29, 645–653. 10.1016/j.biotechadv.2011.04.006
278
MishraS.TripathiR. D.SrivastavaS.DwivediS.TrivediP. K.DhankherO. P.et al. (2009a). Thiol metabolism play significant role during Cd detoxification by Ceratophyllum demersum L. Biores. Tech.100, 2155–2161. 10.1016/j.biortech.2008.10.041
279
MishraY.ChaurasiaN.RaiL. C. (2009b). AhpC (alkyl hydroperoxide reductase) from Anabaena sp. PCC 7120 protects Escherichia coli from multiple abiotic stresses. Biochem. Biophys. Res. Commun.381, 606–611. 10.1016/j.bbrc.2009.02.100
280
MittlerR. (2002). Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci.7, 405–410. 10.1016/S1360-1385(02)02312-9
281
MittlerR.VanderauweraS.GolleryM.Van BreusegemF. (2004). Reactive oxygen gene network of plants. Trends Plant Sci.9, 490–498. 10.1016/j.tplants.2004.08.009
282
MizunoT.UsuiK.HorieK.NosakaS.MizunoN.ObataH. (2005). Cloning of three ZIP/Nramp transporter genes from a Ni hyperaccumulator plant Thlaspi japonicum and their Ni2+-transport abilities. Plant Physiol. Biochem.43, 793–801. 10.1016/j.plaphy.2005.07.006
283
MøllerI. M.JensenP. E.HanssonA. (2007). Oxidative modifications to cellular components in plants. Annu. Rev. Plant Biol.58, 459–481. 10.1146/annurev.arplant.58.032806.103946
284
MonnetF.VaillantN.VernayP.CoudretA.SallanonH.HitmiA. (2001). Relationship between PSII activity, CO2 fixation, and Zn, Mn and Mg contents of Lolium perenne under zinc stress. J. Plant Physiol.158, 1137–1144. 10.1078/S0176-1617(04)70140-6
285
MontaniniB.BlaudezD.JeandrozS.SandersD.ChalotM. (2007). Phylogenetic and functional analysis of the Cation Diffusion Facilitator (CDF) family. Improved signature and prediction of substrate specificity. BMC Genomics8:107. 10.1186/1471-2164-8-107
286
MorelM.CrouzetJ.GravotA.AuroyP.LeonhardtN.VavasseurA.et al. (2009). AtHMA3, a P1B-ATPase allowing Cd/Zn/Co/Pb vacuolar storage in Arabidopsis. Plant Physiol.149, 894–904. 10.1104/pp.108.130294
287
MoritaS.TasakaM.FujisawaH.UshimaruT.TsujiH. (1994). A cDNA clone encoding a rice catalase isozyme. Plant Physiol.105, 1015–1016. 10.1104/pp.105.3.1015
288
MukhopadhyayA.VijS.TyagiA. K. (2004). Overexpression of a zinc-finger protein gene from rice confers tolerance to cold, dehydration, and salt stress in transgenic tobacco. Proc. Natl. Acad. Sci. U.S.A.101, 6309–6314. 10.1073/pnas.0401572101
289
Munne-BoschS. (2005). The role of a9-tocopherol in plant stress tolerance. J. Plant Physiol.162, 743–748. 10.1016/j.jplph.2005.04.022
290
MurphyA.TaizL. (1995). Comparison of metallothionein gene expression and nonprotein thiols in ten Arabidopsis ecotypes. Plant Physiol.109, 945–954. 10.1104/pp.109.3.945
291
MuthuchelianK.BertaminiM.NedunchezhianN. (2001). Triacontanol can protect Erythrina variegata from cadmium toxicity. J. Plant Physiol.158, 1487–1490. 10.1078/0176-1617-00627
292
NadaE.FerjaniB. A.AliR.ImedB. R. B. M.MakkiB. (2007). Cadmium-induced growth inhibition and alteration of biochemical parameters in almond seedlings grown in solution culture. Acta Physil. Plant.29, 57–62. 10.1007/s11738-006-0009-y
293
NagajyotiP. C.LeeK. D.SreekanthT. V. M. (2010). Heavy metal, occurrence and toxicity for plants: a review. Environ. Chem. Lett.8, 199–216. 10.1007/s10311-010-0297-8
294
NaikaM.ShameerK.MathewO. K.GowdaR.SowdhaminiR. (2013). STIFDB2: an updated version of plant stress-responsive transcription factor database with additional stress signals, stress-responsive transcription factor binding sites and stress-responsive genes in Arabidopsis and rice. Plant Cell Physiol.54:e8. 10.1093/pcp/pcs185
295
NakanoT.SuzukiK.FujimuraT.ShinshiH. (2006). Genomewide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol.140, 411–432. 10.1104/pp.105.073783
296
NakashimaK.Yamaguchi-ShinozakiK. (2006). Regulons involved in osmotic stress-responsive and cold stress-responsive gene expression in plants. Physiol. Plant.126, 62–71. 10.1111/j.1399-3054.2005.00592.x
297
NakashimaK.ItoY.Yamaguchi-ShinozakiK. (2009). Transcriptional regulatory networks in response to abiotic stresses in Arabidopsis and grasses. Plant Physiol.149, 88–95. 10.1104/pp.108.129791
298
NanZ.LiJ.ZhangJ.ChengG. (2002). Cadmium and zinc interactions and their transfer in soil–crop system under actual field conditions. Sci. Total Environ. 285, 187–195. 10.1016/S0048-9697(01)00919-6
299
NandaKumarP. B. A.DushenkovV.MottoH.RaskinI. (1995). Phytoextraction: the use of plants to remove heavy metal from soils. Environ. Sci. Technol.29, 1232–1238. 10.1021/es00005a014
300
NeillS.DesikanR.HancockJ. (2002). Hydrogen peroxide signaling. Curr. Opin. Plant Biol.5, 388–395. 10.1016/S1369-5266(02)00282-0
301
NiesD. H.SilverS. (1995). Ion efflux systems involved in bacterial metal resistances. J. Ind. Microbiol.14, 186–199. 10.1007/BF01569902
302
OpdenakkerK.RemansT.KeunenE.VangronsveldJ.CuypersA. (2012). Exposure of Arabidopsis thaliana to Cd or Cu excess leads to oxidative stress mediated alterations in MAPKinase transcript levels. Environ. Exp. Bot.83, 53–61. 10.1016/j.envexpbot.2012.04.003
303
OrtizD. F.KreppelL.SpeiserD. M.ScheelG.McDonaldG.OwD. W. (1992). Heavy metal tolerance in the fission yeast requires an ATP-binding cassette-type vacuolar membrane transporter. EMBO J.11, 3491–3499.
304
OrtizD. F.RuscittiT.McCueK. F.OwD. W. (1995). Transport of metal-binding peptides by HMT1, a fission Yeast ABC-type vacuolar membrane protein. J. Biol. Chem.270, 4721–4728. 10.1074/jbc.270.9.4721
305
Pal'ove-BalangP.KisováA.PavlovkinJ.MistríkI. (2006). Effect of manganese on cadmium toxicity in maize seedlings. Plant Soil Environ.52, 143–149. Available online at: http://www.agriculturejournals.cz/publicFiles/50533.pdf
306
PandaS. K.BaluskaF.MatsumotoH. (2009). Aluminum stress signaling in plants. Plant Signal. Behav.4, 592–597. 10.4161/psb.4.7.8903
307
PankovicD.PlesnicarM.Arsenijeevic-MaksimovicI.PetrovicN.SakacZ.KastoriR. (2000). Effects of nitrogen nutrition on photosynthesis in Cd-treated sunflower plants. Ann. Bot.86, 841–847. 10.1006/anbo.2000.1250
308
PapoyanA.KochianL. V. (2004). Identification of Thlaspi caerulescens genes that may be involved in heavy metal hyperaccumulationand tolerance. Characterization of a novel heavy metal transporting ATPase. Plant Physiol.136, 3814–3823. 10.1104/pp.104.044503
309
Pawlak-SpradaS.Arasimowicz-JelonekM.PodgórskaM.DeckertJ. (2011). Activation of phenylopropanoid pathway in legume plants exposed to heavy metals. Part I. Effects of cadmium and lead on phenylalanine ammonia-lyase gene expression, enzyme activity and lignin content. Acta. Biochim. Pol.58, 211–216.
310
PeiterE.MontaniniB.GobertA.PedasP.HustedS.MaathuisF. J. M.et al. (2007). A secretory pathway-localized cation diffussion facilitator confers plant manganese tolerance. Proc. Natl. Acad. Sci. U.S.A.104, 8532–8537. 10.1073/pnas.0609507104
311
PelegZ.BlumwaldE. (2011). Hormone balance and abiotic stress tolerance in crop plants. Curr. Opin. Plant Biol.14, 290–295. 10.1016/j.pbi.2011.02.001
312
PenaL. B.BarciaR. A.AzpilicuetaC. E.MéndezA. A.GallegoS. M. (2012). Oxidative post translational modifications of proteins related to cell cycle are involved in cadmium toxicity in wheat seedlings. Plant Sci.196, 1–7. 10.1016/j.plantsci.2012.07.008
313
PenceN. S.LarsenP. B.EbbsS. D.LasatM. M.LethamD. L. D.GarvinD. F.et al. (2000). The molecular basis for heavy metal hyperaccumulation in Thlaspi caerulescens. Proc. Natl. Acad. Sci. U.S.A.97, 4956–4960. 10.1073/pnas.97.9.4956
314
PengH. Y.YangX. E.JiangL. Y. (2005). Copper phytoavailability and uptake by Elsholtzi asplendens from contaminated soil as affected by soil amendments. J. Environ. Sci. Health40, 839–856. 10.1081/ESE-200048283
315
PetöA.LehotaiN.Lozano-JusteJ.LeónJ.TariI.ErdeiL.et al. (2011). Involvement of nitric oxide and auxin in signal transduction of copper-induced morphological responses in Arabidopsis seedlings. Ann. Bot.108, 449–457. 10.1093/aob/mcr176
316
Pilon-SmitsE. A. H.HwangS.LytleC. M.ZhuY.TaiJ. C.BravoR. C.et al. (1999). Overexpression of ATP sulfurylase in indian mustard leads to increased selenate uptake, reduction, and tolerance. Plant Physiol.119, 1123–1132. 10.1104/pp.119.1.123
317
Pilon-SmitsE. A. H.ZhuY. L.SearsT.TerryN. (2000). Overexpression of glutathione reductase in Brassica juncea: effects on cadmium accumulation and tolerance. Physiol. Plant.110, 455–460. 10.1111/j.1399-3054.2000.1100405.x
318
PodarD.SchererJ.NoordallyZ.HerzykP.NiesD.SandersD. (2012). Metal selectivity determinants in a family of transition metal transporters. J. Biol. Chem.287, 3185–3196. 10.1074/jbc.m111.305649
319
PomponiM.CensiV.Di GirolamoV.De PaolisA.di ToppiL. S.AromoloR.et al. (2006). Overexpression of Arabidopsis phytochelatin synthase in tobacco plants enhances Cd(2+) tolerance and accumulation but not translocation to the shoot. Planta223, 180–190. 10.1007/s00425-005-0073-3
320
PopovaL. P.MaslenkovaL. T.YordanovaR. Y.IvanovaA. P.KrantevA. P.SzalaiG.et al. (2009). Exogenous treatment with salicylic acid attenuates cadmium toxicity in pea seedlings. Plant Physiol. Biochem.47, 224–231. 10.1016/j.plaphy.2008.11.007
321
PrasadA. S. (2012). Discovery of human zinc deficiency: 50 years later. J. Trace Elem. Med. Biol.26, 66–69. 10.1016/j.jtemb.2012.04.004
322
PrasadM. N. V.FreitasH.FraenzleS.WuenschmannS.MarkertB. (2010). Knowledge explosion in phytotechnologies for environmental solutions. Environ. Pollut.158, 18–23. 10.1016/j.envpol.2009.07.038
323
QiuZ. Z.GuanZ. Y.LongC. Y. (2005). Effect of zinc on cadmium uptake by spring wheat (Triticum aestivum L.): long time hydroponic study and short time 109Cd tracing study. J. Zhejiang Univ. Sci.6, 643–648. 10.1007/BF02856167
324
QureshiM. I.D'AmiciG. M.FagioniM.RinalducciS.ZollaL. (2010). Iron stabilizes thylakoid protein-pigment complexes in indian mustard during Cd-phytoremediation as revealed by BN-SDS-PAGE and ESI-MS/MS. J. Plant Physiol.167, 761–770. 10.1016/j.jplph.2010.01.017
325
RaiV. K. (2002). Role of amino acids in plant responses to stress. Biol. Plant.45, 481–487. 10.1023/A:1022308229759
326
RamosJ.ClementeM. R.NayaL.LoscosJ.Perez-RontomeC.SatoS.et al. (2007). Phytochelatin synthases of the model legume Lotus japonicus. A small multigene family with different responses to cadmium and alternatively spiced variants. Plant Physiol.143, 110–118. 10.1104/pp.106.090894
327
RascioN. (1997). Metal accumulation by some plants growing on zinc-mine deposits. Oikos29, 250–253. 10.2307/3543610
328
RauserW. E. (1995). Phytochelatins and related peptides. Structure, biosynthesis, and function. Plant Physiol.109, 1141–1149. 10.1104/pp.109.4.1141
329
RauserW. E. (1999). Structure and function of metal chelators produced by plants. The case for organic acids, amino acids, phytin and metallothioneins. Cell Biochem. Biophys.31, 19–48. 10.1007/BF02738153
330
RavetK.TouraineB.BoucherezJ.BriatJ. F.GaymardF.CellierF. (2009). Ferritins control interaction between iron homeostasis and oxidative stress in Arabidopsis. Plant J.57, 400–412. 10.1111/j.1365-313X.2008.03698.x
331
ReevesR. D. (2006). Hyperaccumulation of trace elements by plants, in Phytoremediation of Metal-Contaminated Soils, NATO Science Series: IV: Earth and Environmental Sciences, eds MorelJ. L.EchevarriaG.GoncharovaN. (New York, NY: Springer), 25–52.
332
RicachenevskyF. K.MenguerP. K.SperottoR. A.WilliamsL. E.FettJ. P. (2013). Roles of plant metal tolerance proteins (MTP) in metal storage and potential use in biofortification strategies. Front. Plant Sci4:144. 10.3389/fpls.2013.00144
333
RitterA.UbertiniM.RomacS.GaillardF.DelageL.MannA.et al. (2010). Copper stress proteomics highlights local adaptation of two strains of the model brown alga Ectocarpus siliculosus. Proteomics10, 2074–2088. 10.1002/pmic.200900004
334
Rivas-San VicenteM.PlasenciaJ. (2011). Salicylic acid beyond defence: its role in plant growth and development. J. Exp. Bot.62, 3321–3338. 10.1093/jxb/err031
335
Rodríguez-SerranoM.Romero-PuertasM. C.PazmiňoD. M.TestillanoP. S.RisueňoM. C.Del RíoL. A.et al. (2009). Cellular response of pea plants to cadmium toxicity: cross talk between reactive oxygen species, nitric oxide, and calcium. Plant Physiol.150, 229–243. 10.1104/pp.108.131524
336
RothU.von Roepenack-LahayeE.ClemensS. (2006). Proteome changes in Arabidopsis thaliana roots upon exposure to Cd2+. J. Exp. Bot.57, 4003–4013. 10.1093/jxb/erl170
337
RuizO. N.AlvarezD.TorresC.RomanL.DaniellH. (2011). Metallothionein expression in chloroplasts enhances mercury accumulation and phytoremediation capability. Plant Biotechnol. J.9, 609–617. 10.1111/j.1467-7652.2011.00616.x
338
SafarzadehS.RonaghiA.KarimianN. (2013). Effect of cadmium toxicity on micronutrient concentration, uptake and partitioning in seven rice cultivars. Arch. Agron. Soil Sci.59, 231–245. 10.1080/03650340.2011.622752
339
SaitoA.SaitoM.IchikawaY.YoshibaM.TadanoT.MiwaE.et al. (2010). Difference in the distribution and speciation of cellular nickel between nickel-tolerant and non-tolerant Nicotiana tabacum L. cv. BY-2 cells. Plant Cell Environ.33, 174–187. 10.1111/j.1365-3040.2009.02068.x
340
SaltD. E.RauserW. E. (1995). MgATP-dependent transport of phytochelatins across the tonoplast of oat roots. Plant Physiol.107, 1293–1301.
341
SaltD. E.BaxterI.LahnerB. (2008). Ionomics and the study of the plant ionome. Annu. Rev. Plant Biol. 59, 709–733. 10.1146/annurev.arplant.59.032607.092942
342
SaltD. E.PrinceR. C.BakerA. J. M.RaskinI.PickeringI. J. (1999). Zinc ligands in the metal accumulator Thlaspi caerulescensas determined using X-ray absorption spectroscopy. Environ. Sci. Technol.33, 713–717. 10.1021/es980825x
343
Sánchez-BermejoE.CastrilloG.del LlanoB.NavarroC.Zarco-FernándezS.Martinez- HerreraD. J.et al. (2014). Natural variation in arsenate tolerance identifies an arsenate reductase in Arabidopsis thaliana. Nat. Commun. 5:4617. 10.1038/ncomms5617
344
Sánchez-PardoB.Fernández-PascualM.ZornozaP. (2012). Copper microlocalisation, ultrastructural alterations and antioxidant responses in the nodules of white lupin and soybeanplants grown under conditions of copper excess. Environ. Exp. Bot.84, 52–60. 10.1016/j.envexpbot.2012.04.017
345
Sanita di ToppiL.GabbrielliR. (1999). Response to cadmium in higher plants. Environ. Exp. Bot.41, 105–130. 10.1016/S0098-8472(98)00058-6
346
SanoT.YoshiharaT.HandaK.SatoM. H.NagataT.HasezawaS. (2012). Metal ion homeostasis mediated by NRAMP transporters in plant cells-focused on increased resistance to iron and cadmium ion, in Crosstalk and Integration of Membrane Trafficking Pathways, ed WeigertR. (InTech), 213–228. Available online at: http://www.intechopen.com/books/crosstalk-and-integration-of-membrane-trafficking-pathways/metal-ion-homeostasis-mediated-by-nramp-transporters-in-plant-cells-focused-on-increased-resistance-
347
SarretG.Saumitou-LapradeP.BertV.ProuxO.HazemannJ. L.TraverseA.et al. (2002). Forms of zinc accumulated in the hyperaccumulator Arabidopsis halleri. Plant Physiol.130, 1815–1826. 10.1104/pp.007799
348
SarwarN.SaifullahMalhiS. S.ZiaM. H.NaeemA.BibiaS.FaridaG. (2010). Role of mineral nutrition in minimizing cadmium accumulation by plants. J. Sci. Food Agric. 90, 925–937. 10.1002/jsfa.3916
349
SasakiA.YamajiN.YokoshoK.MaJ. F. (2012). Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell. 24, 2155–2167. 10.1105/tpc.112.096925
350
SchaferC.SimperH.HofmannB. (1992). Glucose feeding results in coordinated changes of chlorophyll content, ribulose-1,5-biphosphate carboxylase-oxygenase activity and photosynthetic potential photoautotrophic suspension cultured cells of Chenopodium ruburum. Plant Cell Environ. 15, 343–350.
351
SchaafG.HonsbeinA.MedaA. R.KirchnerS.WipfD.von WirénN. (2006). AtIREG2 encodes a tonoplast transport protein involved in iron-dependent nickel detoxification in Arabidopsis thaliana roots. J. Biol. Chem.281, 25532–25540. 10.1074/jbc.M601062200
352
ScholzG.SchlesierG.SeifertK. (1985). Effect of nicotianamine on iron uptake by the tomato mutant ‘chloronerva’. Physiol. Plant.63, 99–104. 10.1111/j.1399-3054.1985.tb02825.x
353
SchützendübelA.PolleA. (2002). Plant responses to abiotic stresses: heavy metal-induced oxidative stress and protection by mycorrhization. J. Exp. Bot.53, 1351–1365. 10.1093/jexbot/53.372.1351
354
SchwarzK.FoltzC. M. (1957). Selenium as an integral part of factor 3 against dietary necrotic liver degeneration. J. Am. Chem. Soc.70, 3292–3293. 10.1021/ja01569a087
355
SemaneB.DupaeJ.CuypersA.NobenJ. P.TuomainenM.TervahautaA.et al. (2010). Leaf proteome responses of Arabidopsis thaliana exposed to mild cadmium stress. J. Plant Physiol.167, 247–254. 10.1016/j.jplph.2009.09.015
356
ShahidM.PinelliE.DumatC. (2012). Review of Pb availability and toxicity to plants in relation with metal speciation; role of synthetic and natural organic ligands. J. Hazard. Mater. 219–220, 1–12. 10.1016/j.jhazmat.2012.01.060
357
ShahzadZ.GostiF.FrerotH.LacombeE.RoosensN.Saumitou- LapradeP.et al. (2010). The five AhMTP1 zinc transporters undergo different evolutionary fates towards adaptive evolution to zinc tolerance in Arabidopsis halleri. PLoS Genet.6:e1000911. 10.1371/journal.pgen.1000911
358
ShameerK.AmbikaS.VargheseS. M.KarabaN.UdayakumarM.SowdhaminiR. (2009). STIFDB–Arabidopsis stress-responsive transcription factor DataBase. Int. J. Plant Genomics2009:583429. 10.1155/2009/583429
359
ShankerK.MishraS.SrivastavaS.SrivastavaR.DaasS.PrakashS.et al. (1996). Effect of selenite and selenate on plant uptake and translocation of mercury by tomato (Lycopersicum esculentum). Plant Soil183, 233–238. 10.1007/BF00011438
360
ShanmugamV.LoJ. C.WuC. L.WangS. L.LaiC. C.ConnollyE. L.et al. (2011). Differential expression and regulation of iron regulated metal transporters in Arabidopsis halleri and Arabidopsis thaliana—the role in zinc tolerance. New Phytol.190, 125–137. 10.1111/j.1469-8137.2010.03606.x
361
SharafA. E. M. M.FarghalI. I.SofyM. R. (2009). Role of gibberellic acid in abolishing the detrimental effects of Cd and Pb on broad bean and Lupin plants. Res. J. Agric. Biol. Sci.5, 668–673. Available online at: https://www.researchgate.net/publication/242631571
362
SharmaI.PatiP. K.BhardwajR. (2011). Effect of 28-homobrassinolide on antioxidant defence system in Raphanus sativus L. under chromium toxicity. Ecotoxicology20, 862–874. 10.1007/s10646-011-0650-0
363
SharmaP.BhardwajR. (2007). Effects of 24-epibrassinolide on growth and metal uptake Brassica juncea L. under copper metal stress. Acta Physiol. Plant.29, 259–263. 10.1007/s11738-007-0032-7
364
SharmaP.BhardwajR.AroraN.AroraH. K.KumarA. (2008). Effects of 28-homobrassinolide on nickel uptake, protein content and antioxidative defence system in Brassica juncea. Biol. Plant52, 767–770. 10.1007/s10535-008-0149-6
365
SharmaS. S.DietzK. J. (2009). The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci.14, 43–50. 10.1016/j.tplants.2008.10.007
366
SharmaS. S.DietzK. J. (2006). The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J. Exp. Bot.57, 711–726. 10.1093/jxb/erj073
367
SharminS. A.AlamI.KimK. H.KimY. G.KimP. J.BahkJ. D.et al. (2012). Chromium-induced physiological and proteomic alterations in roots of Miscanthus sinensis. Plant Sci.187, 113–126. 10.1016/j.plantsci.2012.02.002
368
ShenW.NadaK.TachibanaS. (2000). Involvement of polyamines in the chilling tolerance of cucumber cultivars. Plant Physiol.124, 431–439. 10.1104/pp.124.1.431
369
ShiQ.ZhuZ. (2008). Effects of exogenous salicylic acid on manganese toxicity, element contents and antioxidative system in cucumber. Environ. Exp. Bot.63, 317–326. 10.1016/j.envexpbot.2007.11.003
370
ShiX. H.ZhangC. H.WangH.ZhangF. S. (2005). Effect of Si on the distribution of Cd in rice seedlings. Plant Soil272, 53. 10.1007/s11104-004-3920-2
371
ShimD.HwangJ. U.LeeJ.LeeS.ChoiY.AnG.et al. (2009). Orthologs of the class A4 heat shock transcription factor HsfA4a confer cadmium tolerance in wheat and rice. Plant Cell21, 4031–4043. 10.1105/tpc.109.066902
372
ShinS. Y.KimI. S.KimY. H.ParkH. M.LeeJ. Y.KangH. G.et al. (2008). Scavenging reactive oxygen species by rice dehydroascorbate reductase alleviates oxidative stresses in Escherichia coli. Mol. Cells26, 616–620.
373
ShiuS. H.ShihM. C.LiW. H. (2005). Transcription factor families have much higher expansion rates in plants than in animals. Plant Physiol.139, 18–26. 10.1104/pp.105.065110
374
SiborovaM. (1988). Cd2+ ions affect the quaternary structure of ribulose-1,5-bisphosphate carboxylase from barley leaves. Biochem. Physiol. Pflanzen183, 371–378. 10.1016/S0015-3796(88)80045-3
375
SiddiquiM. H.Al-WhaibiM. H.BasalahM. O. (2011). Interactive effect of calcium and gibberellin on nickel tolerance in relation to antioxidant systems in Triticum aestivum L. Protoplasma248, 503–511. 10.1007/s00709-010-0197-6
376
SilvaS. (2012). Aluminium toxicity targets in plants. J. Bot.2012:219462. 10.1155/2012/219462
377
SimõesC. C.MeloJ. O.MagalhaesJ. V.GuimarãesC. T. (2012). Genetic and molecular mechanisms of aluminum tolerance in plants. Genet. Mol. Res.11, 1949–1957. 10.4238/2012.July.19.14
378
SinghK.FoleyR. C.Oñate-SánchezL. (2002). Transcription factors in plant defense and stress responses. Curr. Opin. Plant Biol.5, 430–436. 10.1016/S1369-5266(02)00289-3
379
SinghN.MaL. Q.VuJ. C.RajA. (2009). Effects of arsenic on nitrate metabolism in arsenic hyperaccumulating and non-hyperaccumulating ferns. Environ. Pollut. 157, 2300–2305. 10.1016/j.envpol.2009.03.036
380
SinghR. K.AnandhanS.SinghS.PatadeV. Y.AhmedZ.PandeV. (2011a). Metallothionein-like gene from Cicer microphyllumis regulated by multiple abiotic stresses. Protoplasma248, 839–847. 10.1007/s00709-010-0249-y
381
SinghV. P.SinghS.KumarJ.PrasadS. M. (2015). Investigating the roles of ascorbate-glutathione cycle and thiol metabolism in arsenate tolerance in ridged Luffa seedlings. Protoplasma252, 1217–1229. 10.1007/s00709-014-0753-6
382
SinghV. P.SrivastavaP. K.PrasadS. M. (2012). Differential effect of UV-B radiation on growth, oxidative stress and ascorbate-glutathione cycle in two cyanobacteria under copper toxicity. Plant Physiol. Biochem.61, 61–70. 10.1016/j.plaphy.2012.09.005
383
SinghV. P.SrivatavaP. K.PrasadS. M. (2013). Nitric oxide alleviates arsenic-induced toxic effects in ridged Luffa seedlings. Plant. Physiol. Biochem.71, 155–163. 10.1016/j.plaphy.2013.07.003
384
SinghV. P.TripathiD. K.KumarD.ChauhanD. K. (2011b). Influence of exogenous silicon addition on aluminium tolerance in rice seedlings. Biol. Trace Elem. Res.144, 1260–1274. 10.1007/s12011-011-9118-6
385
SiripornadulsilS.TrainaS.VermaD. P. S.SayreR. T. (2002). Molecular mechanisms of proline-mediated tolerance to toxic heavy metal in transgenic microalgae. Plant Cell14, 2837–2847. 10.1105/tpc.004853
386
SmithP.GregoryP. J.van VuurenD.ObersteinerM.HavlíkP.RounsevellM.et al. (2010). Competition for land. Philos. Trans R. Soc. B Biol. Sci.365, 2941–2957. 10.1098/rstb.2010.0127
387
SobkowiakR.DeckertJ. (2006). Proteins induced by cadmium in soybean cells. J. Plant Physiol. 163, 1203–1206. 10.1016/j.jplph.2005.08.017
388
SongA.LiZ.ZhangJ.XueG.FanF.LiangY. (2009). Silicon-enhanced resistance to cadmium toxicity in Brassica chinensis L. is attributed to Si-suppressed cadmium uptake and transport and Si-enhanced antioxidant defense capacity. J. Hazard. Mater.172, 74–83. 10.1016/j.jhazmat.2009.06.143
389
SongW.-Y.YamakiT.YamajiN.KoD.JungK. H.Fujii-KashinoM.et al. (2014). A rice ABC transporter, OsABCC1, reduces arsenic accumulation in the grain. PNAS111, 15699–15704. 10.1073/pnas.1414968111
390
Sooksa-NguanT.YakubovB.KozlovskyyV. I.BarkumeC. M.HoweK. J.ThannhauserT. W.et al. (2009). Drosophila ABC transporter, DmHMT-1, confers tolerance to cadmium. DmHMT-1 and its yeast homolog, SpHMT-1, are not essential for vacuolar phytochelatin sequestration. J. Biol. Chem.284, 354–362. 10.1074/jbc.M806501200
391
SrivastavaG.KumarS.DubeyG.MishraV.PrasadS. M. (2012). Nickel and ultraviolet-B stresses induce differential growth and photosynthetic responses in Pisum sativum L. seedlings. Biol. Trace Elem. Res.149, 86–96. 10.1007/s12011-012-9406-9
392
SrivastavaS.SrivastavaA. K.SuprasannaP.D'souzaS. F. (2013). Identification and profiling of arsenic stress-induced microRNAs in Brassica juncea. J. Exp. Bot.64, 303–315. 10.1093/jxb/ers333
393
StephanU. W.ScholzG. (1993). Nicotianamine: mediator of transport of iron and heavy metals in the phloem?Physiol. Plant.88, 522–529.
394
StreetR. A.KulkarniM. G.StirkW. A.SouthwayC.Van StadenJ. (2010). Effect of cadmium on growth and micronutrient distribution in wild garlic (Tulbaghia violacea). South Afr. J. Bot.76, 332–336. 10.1016/j.sajb.2009.12.006
395
StrobelN. E.KucA. (1995). Chemical and biological inducers ofsystemic acquired resistance to pathogens protect cucumber andtobacco from damage caused by paraquat and cupric chloride. Phytopathol85, 1306–1310. 10.1094/Phyto-85-1306
396
SundaramoorthyP.ChidambaramA.GaneshK. S.UnnikannanP.BaskaranL. (2010). Chromium stress in paddy: (i) nutrient status of paddy under chromium stress; (ii) phytoremediation of chromium by aquatic and terrestrial weeds. C. R. Biol.333, 597–607. 10.1016/j.crvi.2010.03.002
397
SuzukiN. (2005). Alleviation by calcium of cadmium induced root growth inhibition in Arabidopsis seedlings. Plant Biotechnol.22, 19–25. 10.5511/plantbiotechnology.22.19
398
TakahashiH.KawakatsuT.WakasaY.HayashiS.TakaiwaF. (2012). A rice transmembrane bZIP transcription factor, OsbZIP39, regulates the endoplasmic reticulum stress response. Plant Cell Physiol.53, 144–153. 10.1093/pcp/pcr157
399
TalanovaV. V.TitovA. F.BoevaN. P. (2000). Effect of increasing concentration of lead and cadmium on cucumber seedlings. Biol. Plant.43, 441–444. 10.1023/A:1026735603890
400
TalkeI. N.HanikenneM.KrämerU. (2006). Zinc-dependent global transcriptional control, transcriptional deregulation, and higher gene copy number for genes in metal homeostasis of the hyperaccumulator Arabidopsis halleri. Plant Physiol.142, 148–167. 10.1104/pp.105.076232
401
TamásL.MistríkI.HuttováJ.HaluskováL.ValentovicováK.ZelinováV. (2010). Role of reactive oxygen species-generating enzymes and hydrogen peroxide during cadmium, mercury and osmotic stresses in barley root tip. Planta231, 221–231. 10.1007/s00425-009-1042-z
402
TangW.CharlesT. M.NewtonR. J. (2005). Overexpression of the pepper transcription factor CaPF1 in transgenic Virginia pine (Pinus Virginiana Mill.) confers multiple stress tolerance and enhances organ growth. Plant Mol. Biol.59, 603–617. 10.1007/s11103-005-0451-z
403
ThomineS.LelièvreF.DebarbieuxE.SchroederJ. I.Barbier-BrygooH. (2003). AtNRAMP3, a multispecificvacuolar metal transporter involved in plant responses to iron deficiency. Plant J.34, 685–695. 10.1046/j.1365-313X.2003.01760.x
404
ThomineS.WangR.WardJ. M.CrawfordN. M.SchroederJ. I. (2000). Cadmium and iron transport by members of a plant transporter gene family in Arabidopsis with homology to NRAMP genes. Proc. Nat. Acad. Sci. U.S.A.97, 4991–4996. 10.1073/pnas.97.9.4991
405
ThounaojamT. C.PandaP.MazumdarP.KumarD.SharmaG. D.SahooL.et al. (2012). Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol. Biochem.53, 33–39. 10.1016/j.plaphy.2012.01.006
406
TiwariM.SharmaD.DwivediS.SinghM.TripathiR. D.TrivediP. K. (2014). Expression in Arabidopsis and cellular localization reveal involvement of rice NRAMP, OsNRAMP1, in arsenic transport and tolerance. Plant Cell Environ. 37, 140–152. 10.1111/pce.12138
407
TkalecM.ŠtefanićP. P.CvjetkoP.ŠikićS.PavlicaM.BalenB. (2014). The effects of cadmium-zinc interactions on biochemical responses in tobacco seedlings and adult plants. PLoS ONE9:e87582. 10.1371/journal.pone.0087582
408
TommeyA. M.ShiJ.LindsayW. P.UrwinP. E.RobinsonN. J. (1991). Expression of the pea gene PsMTA in E. coli. Metal binding properties of the expressed protein. FEBS Lett.292, 48–52. 10.1016/0014-5793(91)80831-M
409
TranL. S. P.NishiyamaR.Yamaguchi-ShinozakiK.ShinozakiK. (2010). Potential utilization of NAC transcription factors to enhance abiotic stress tolerance in plants by biotechnological approach. GM Crops1, 32–39. 10.4161/gmcr.1.1.10569
410
TreebyM.MarschnerH.RömheldV. (1989). Mobilization of iron and other micronutrient cations from a calcareous soil by plant-borne, microbial, and synthetic metal chelators. Plant Soil114, 217–226. 10.1007/BF02220801
411
TripathiD. K.SinghV. P.KumarD.ChauhanD. K. (2012). Impact of exogenous silicon addition on chromium uptake, growth, mineral elements, oxidative stress, antioxidant capacity, and leaf and root structures in rice seedlings exposed to hexavalent chromium. Acta Physiol. Plant.34, 279–289. 10.1007/s11738-011-0826-5
412
TsengM. J.LiuC. W.YiuJ. C. (2007). Enhanced tolerance to sulfur dioxide and salt stress of transgenic Chinese cabbage plants expressing both superoxide dismutase and catalase in chloroplasts. Plant Physiol. Biochem.45, 822–833. 10.1016/j.plaphy.2007.07.011
413
TsukagoshiH.BuschW.BenfeyP. N. (2010). Transcriptional regulation of ROS controls transition from proliferation to differentiation in the root. Cell143, 606–616. 10.1016/j.cell.2010.10.020
414
TuC.MaL. Q. (2005). Effects of arsenic on concentration and distribution of nutrients in the fronds of the arsenic hyperaccumulator Pteris vittata L. Environ. Pollut.135, 333–340. 10.1016/j.envpol.2004.03.026
415
TuomainenM. H.NunanN.LehesrantaS. J.TervahautaA. I.HassinenV. H.SchatH.et al. (2006). Multivariate analysis of protein profiles of metal hyperaccumulator Thlaspi caerulescens accessions. Proteomics6, 3696–3706. 10.1002/pmic.200501357
416
UmezawaT.FujitaM.FujitaY.Yamaguchi-ShinozakiK.ShinozakiK. (2006). Engineering drought tolerance in plants: discovering and tailoring genes to unlock the future. Curr. Opin. Biotechnol.17, 113–122. 10.1016/j.copbio.2006.02.002
417
ValliyodanB.NguyenH. T. (2006). Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr. Opin. Plant Biol.9, 189–195. 10.1016/j.pbi.2006.01.019
418
van de MortelJ. E.VillanuevaL. A.SchatH.KwekkeboomJ.CoughlanS.MoerlandP. D.et al. (2006). Large expression differences in genes for iron and zinc homeostasis, stress response, and lignin biosynthesis distinguish roots of Arabidopsis thaliana and the related metal hyperaccumulator Thlaspi caerulescens. Plant Physiol.142, 1127–1134. 10.1104/pp.106.082073
419
van der ZaalB. J.NeuteboomL. W.PinasJ. E.ChardonnensA. N.SchatH.VerkleijJ. A.et al. (1999). Overexpression of a novel Arabidopsis gene related to putative zinc-transporter genes from animals can lead to enhanced zinc resistance and accumulation. Plant Physiol.119, 1047–1055. 10.1104/pp.119.3.1047
420
VarottoC.MaiwaldD.PesaresiP.JahnsP.SalaminiF.LeisterD. (2002). The metal ion transporter IRT1 is necessary for iron homeostasis and efficient photosynthesis in Arabidopsis thaliana. Plant J.31, 589–599. 10.1046/j.1365-313X.2002.01381.x
421
VassilevA.YordanovI. (1997). Reductive analysis of factors limiting growth of cadmium-treated plants: a review. Bulg. J. Plant Physiol.23, 114–133.
422
VatamaniukO. K.BucherE. A.SundaramM. V.ReaP. A. (2005). CeHMT-1, a putative phytochelatin transporter, is required for cadmium tolerance in Caenorhabditis elegans. J. Biol. Chem.280, 23684–23690. 10.1074/jbc.M503362200
423
VerbruggenN.HermansC.SchatH. (2009). Molecular mechanisms of metal hyperaccumulation in plants. New Phytol.181, 759–776. 10.1111/j.1469-8137.2008.02748.x
424
VertG.BarberonM.ZelaznyE.SéguélaM.BriatJ. F.CurieC. (2009). Arabidopsis IRT2 cooperates with the high-affinity iron uptake system to maintain iron homeostasis in root epidermal cells. Planta229, 1171–1179. 10.1007/s00425-009-0904-8
425
VertG.GrotzN.DédaldéchampF.GaymardF.GuerinotM. L.BriatJ. F.et al. (2002). IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell14, 1223–1233. 10.1105/tpc.001388
426
VilliersF.JourdainA.BastienO.LeonhardtN.FujiokaS.TichtinckyG.et al. (2012). Evidence for functional interaction between brassinosteroids and cadmium response in Arabidopsis thaliana. J. Exp. Bot.63, 1185–1200. 10.1093/jxb/err335
427
VinocurB.AltmanA. (2005). Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Curr. Opin. Biotechnol.16, 123–132. 10.1016/j.copbio.2005.02.001
428
Vögeli-LangeR.WagnerG. J. (1990). Subcellular localization of cadmium and cadmium-binding peptides in tobacco leaves. Implication of a transport function for cadmium-binding peptides. Plant Physiol.92, 1086–1093. 10.1104/pp.92.4.1086
429
VrietC.RussinovaE.ReuzeauC. (2012). Boosting crop yields with plant steroids. Plant Cell24, 842–857. 10.1105/tpc.111.094912
430
WalliwalagedaraC.van KeulenH.WillardB.WeiR. (2012). Differential proteome analysis of Chlamydomonas reinhardtii response to arsenic exposure. Am. J. Plant Sci.3, 764–772. 10.4236/ajps.2012.36092
431
WangH. Y.KlatteM.JakobyM.BäumleinH.WeisshaarB.BauerP. (2007). Iron deficiency-mediated stress regulation of four subgroup Ib BHLH genes in Arabidopsis thaliana. Planta226, 897–908. 10.1007/s00425-007-0535-x
432
WangH.ZhaoS. C.LiuR. L.ZhouW.JinJ. Y. (2009). Changes of photosynthetic activities of maize (Zea mays L.) seedlings in response to cadmium stress. Photosynthetica47, 277–283. 10.1007/s11099-009-0043-2
433
WangR.GaoF.GuoB. G.HuangJ. C.WangL.ZhouY. J. (2013). Short-term chromium-stress-induced alterations in the maize leaf proteome. Int. J. Mol. Sci.14, 11125–11144. 10.3390/ijms140611125
434
WangS. H.YangZ. M.YangH.LuB.LiS. Q.LuY. P. (2004). Copper induced stress and antioxidative responses in roots of Brassica juncea L. Bot. Bull. Acad. Sin.45, 203–212. Available online at: http://ejournal.sinica.edu.tw/bbas/content/2004/3/Bot453-04.pdf
435
WangY.HuH.ZhuL. Y.LiX. X. (2012). Response to nickel in the proteome of the metal accumulator plant Brassica juncea. J. Plant Interact.7, 230–237. 10.1080/17429145.2011.603060
436
WangelineA. L.BurkheadJ. L.HaleK. L.LindblomS. D.TerryN.PilonM.et al. (2004). Over- expression of ATP sulfurylase in indian mustard: effects on tolerance and accumulation of twelve metals. J. Environ. Qual.33, 54–60. 10.2134/jeq2004.5400
437
WaniP. A.KhanM. S.ZaidiA. (2012). Toxic effects of heavy metal on germination and physiological processes of plants, in Toxicity of Heavy Metal to Legumes and Bioremediation, eds ZaidiA.WaniP. A.KhanM. S. (Springer-Verlag Wien), 45–66.
438
WarneM. S.HeemsbergenD.StevensD.McLaughlinM.CozensG.WhatmuffM.et al. (2008). Modeling the toxicity of copper and zinc salts to wheat in 14 soils. Environ. Toxicol. Chem.27, 786–792. 10.1897/07-294.1
439
WeberM.TrampczynskaA.ClemensS. (2006). Comparative transcriptome analysis of toxic metal responses in Arabidopsis thaliana and the Cd2+-hypertolerant facultative metallophyte Arabidopsis halleri. Plant Cell Environ.29, 950–963. 10.1111/j.1365-3040.2005.01479.x
440
WhitingS. N.LeakeJ. R.McGrathS. P.BakerA. J. M. (2000). Positive responses to zinc and cadmium by roots of the hyperaccumulator Thlaspi caerulescens. New Phytol.145, 199–210. 10.1046/j.1469-8137.2000.00570.x
441
WilkinsonS.KudoyarovaG. R.VeselovD. S.ArkhipovaT. N.DaviesW. J. (2012). Plant hormone interactions: innovative targets for crop breeding an and management. J. Exp. Bot.63, 3499–3509. 10.1093/jxb/ers148
442
WilliamsL. E.PittmanJ. K. (2010). Dissecting pathways involved in manganese homeostasis and stress in higher plants, in Cell Biology of Metals and Nutrients, Plant Cell Monographs, Vol. 17, eds HellR.MendalR. R. (Berlin, Heidelberg: Springer-Verlag), 95–117.
443
WilliamsL. E.PittmanJ. K.HallJ. L. (2000). Emerging mechanisms for heavy metal transport in plants. Biochim. Biophys. Acta1465, 104–126. 10.1016/S0005-2736(00)00133-4
444
Winkel-ShirleyB. (2002). Biosynthesis of flavonoids and effects of stress. Curr. Opin. Plant Biol. 5, 218. 10.1016/S1369-5266(02)00256-X
445
WongC. K. E.CobbettC. S. (2009). HMA P-Type ATPases are the major mechanism for root-to-shoot Cd translocation in Arabidopsis thaliana. New Phytol.181, 71–78. 10.1111/j.1469-8137.2008.02638.x
446
WrayG. A.HahnM. W.AbouheifE.BalhoffJ. P.PizerM.RockmanM. V.et al. (2003). The evolution of transcriptional regulation in eukaryotes. Mol. Biol. Evol.20, 1377–1419. 10.1093/molbev/msg140
447
WuF. B.ZhangG. P. (2002). Genotypic variation in kernel heavy metal concentrations in barley and as affected by soil factors. J. Plant Nutr.25, 1163–1173. 10.1081/PLN-120004380
448
WuH.ChenC.DuJ.LiuH.CuiY.ZhangY.et al. (2012). Co-overexpression FIT with AtbHLH38 or AtbHLH39 in Arabidopsis-enhanced cadmium tolerance via increased cadmium sequestration in roots and improved iron homeostasis of shoots. Plant Physiol.158, 790–800. 10.1104/pp.111.190983
449
XiaX. J.WangY. J.ZhouY. H.TaoY.MaoW. H.ShiK.et al. (2009). Reactive oxygen species are involved in brassinosteroid-induced stress tolerance in cucumber. Plant Physiol.150, 801–814. 10.1104/pp.109.138230
450
XiaX. J.ZhouY. H.DingJ.ShiK.AsamiT.ChenZ.et al. (2011). Induction of systemic stress tolerance by brassinosteroid in Cucumis sativus. New Phytol.191, 706–720. 10.1111/j.1469-8137.2011.03745.x
451
XiaZ.SunK.WangM.WuK.ZhangH.WuJ. (2012). Overexpression of a maize sulfite oxidase gene in tobacco enhances tolerance to sulfite stress via sulfite oxidation and CAT-mediated H2O2 scavenging. PLoS ONE7:e37383. 10.1371/journal.pone.0037383
452
XiangC.OliverD. J. (1998). Glutathione metabolic genes coordinately respond to heavy metal and jasmonic acid in Arabidopsis. Plant Cell10, 1539–1550. 10.1105/tpc.10.9.1539
453
XuJ.TianY. S.PengR. H.XiongA. S.ZhuB.JinX. F.et al. (2009). Yeast copper-dependent transcription factor ACE1 enhanced copper stress tolerance in Arabidopsis. BMB Rep.42, 752–757. 10.5483/BMBRep.2009.42.11.752
454
XuL. L.FanZ. Y.DongY. J.KongJ.BaiX. Y. (2014). Effects of exogenous salicylic acid and nitric oxide on physiological characteristics of two peanut cultivars under cadmium stress. Biol. Plant.59, 171–182. 10.1007/s10535-014-0475-9
455
YadavS. K. (2010). Heavy metal toxicity in plants: an overview on the role of glutathione and phytochelatins in heavy metal stress tolerance of plants. South Afr. J. Bot.76, 167–179. 10.1016/j.sajb.2009.10.007
456
YamajiN.HuangC. F.NagaoS.YanoM.SatoY.NagamuraY.et al. (2009). A zinc finger transcription factor ART1 regulates multiple genes implicated in aluminum tolerance in rice. Plant Cell21, 3339–3349. 10.1105/tpc.109.070771
457
YangM. J.LinX. Y.YangX. E. (1998). Impact of Cd on growth and nutrient accumulation of different plant species. Chin. J. Appl. Ecol. 9, 89–94.
458
YangX. E.LongX. X.NiW. Z. (2002). Physiological and molecular mechanisms of heavy metal uptake by hyperaccumulating plant species. J. Plant Nutr. Fert.8, 8–15. Available online at: http://www.ipublishing.co.in/ijesarticles/thirteen/articles/volthree/EIJES31135.pdf
459
YangX.FengY.HeZ.StoffellP. J. (2005). Molecular mechanisms of heavy metal hyperaccumulation and phytoremediation. J. Trace Elem. Med. Biol.18, 339–353. 10.1016/j.jtemb.2005.02.007
460
YangY.QiM.MeiC. (2004). Endogenous salicylic acid protects rice plants from oxidative damage caused by aging as well as biotic and abiotic stress. Plant J.40, 909–919. 10.1111/j.1365-313X.2004.02267.x
461
YangZ. M.WangJ.WangS. H.XuL. L. (2003). Salicylic acid-induced aluminum tolerance by modulation of citrate efflux from roots of Cassia tora L. Planta217, 168–174. 10.1007/s00425-003-0980-0
462
YinL.WangS.EltayebA. E.UddinM. I.YamamotoY.TsujiW.et al. (2010). Overexpression of dehydroascorbate reductase, but not monodehydroascorbate reductase, confers tolerance to aluminum stress in transgenic tobacco. Planta231, 609–621. 10.1007/s00425-009-1075-3
463
YuanH. M.XuH. H.LiuW. C.LuY. T. (2013). Copper regulates primary root elongation through PIN1-mediated auxin redistribution. Plant Cell Physiol.54, 766–778. 10.1093/pcp/pct030
464
YuanL.YuanY.DuJ.SunJ.GuoS. (2012). Effects of 24-epibrassinolide on nitrogen metabolism in cucumber seedlings under Ca(NO3)2 stress. Plant Physiol. Biochem.61, 29–35. 10.1016/j.plaphy.2012.09.004
465
YuanY. X.ZhangJ.WangD. W.LingH.-Q. (2005). AtbHLH29 of Arabidopsis thaliana is a functional ortholog of tomato FER involved in controlling iron acquisition in strategy I plants. Cell Res.15, 613–621. 10.1038/sj.cr.7290331
466
YuanY.WuH.WangN.LiJ.ZhaoW.DuJ.et al. (2008). FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Res.18, 385–397. 10.1038/cr.2008.26
467
YusufM. A.KumarD.RajwanshiR.StrasserR. J.Tsimilli-MichaelM.Govindjeeet al. (2010). Overexpression of c-tocopherol methyl transferase gene in transgenic Brassica juncea plants alleviates abiotic stress: physiological and chlorophyll a fluorescence measurements. Biochim. Biophys. Acta1797, 1428–1438. 10.1016/j.bbabio.2010.02.002l
468
YusufM.FariduddinQ.AhmadA. (2012a). 24-Epibrassinolide modulates growth, nodulation, antioxidant system, and osmolyte in tolerant and sensitive varieties of Vigna radiata under different levels of nickel: a shotgun approach. Plant Physiol. Biochem.57, 143–153. 10.1016/j.plaphy.2012.05.004
469
YusufM.FariduddinQ.VarshneyP.AhmadA. (2012b). Salicylic acid minimizes nickel and/or salinity-induced toxicity in Indian mustard (Brassica juncea) through an improved antioxidant system. Environ. Sci. Pollut. Res.19, 8–18. 10.1007/s11356-011-0531-3
470
ZaccheoP.LauraC.ValeriaD. M. P. (2006). Ammonium nutrition as a strategy for cadmium metabolisation in the rhizosphere of sunflower. Plant Soil283, 43–56. 10.1007/s11104-005-4791-x
471
ZhangA.ZhangJ.ZhangJ.YeN.ZhangH.TanM.et al. (2011). Nitric oxide mediates brassinosteroid-induced ABA biosynthesis involved in oxidative stress tolerance in maize leaves. Plant Cell Physiol.52, 181–192. 10.1093/pcp/pcq187
472
ZhangG.ChenM.LiL.XuZ.ChenX.GuoJ.et al. (2009a). Overexpression of the soybean GmERF3 gene, an AP2/ERF type transcription factor for increased tolerances to salt, drought, and diseases in transgenic tobacco. J. Exp. Bot.60, 3781–3796. 10.1093/jxb/erp214
473
ZhangH.LianC.ShenZ. (2009b). Proteomic identification of small, copper-responsive proteins in germinating embryos of Oryza sativa. Ann. Bot.103, 923–930. 10.1093/aob/mcp012
474
ZhangY.LiJ.YuF.CongL.WangL.BurkardG.et al. (2006). Cloning and expression analysis of SKn-type dehydrin gene from bean in response to heavy metal. Mol. Biotechnol.32, 205–217. 10.1385/MB:32:3:205
475
ZhangY.YuZ.FuX.LiangC. (2002). Noc3p, a bHLH protein, plays an integral role in the initiation of DNA replication in budding yeast. Cell109, 849–860. 10.1016/S0092-8674(02)00805-X
476
ZhangZ. C.ChenB. X.QiuB. S. (2010). Phytochelatin synthesis plays a similar role in shoots of the cadmium hyperaccumulator Sedum alfredii as in non-resistant plants. Plant Cell Environ.33, 1248–1255. 10.1111/j.1365-3040.2010.02144.x
477
ZhaoL.SunY. L.CuiS. X.ChenM.YangH. M.LiuH. M.et al. (2011). Cd-induced changes in leaf proteome of the hyperaccumulator plant Phytolacca americana. Chemosphere85, 56–66. 10.1016/j.chemosphere.2011.06.029
478
ZhaoZ. Q.ZhuY. G.LiH. Y.SmithS. E.SmithF. A. (2004). Effects of forms and rates of potassium fertilizers on cadmium uptake by two cultivars of spring wheat (Triticum aestivum L.). Environ. Int.29, 973–978. 10.1016/S0160-4120(03)00081-3
479
ZhengX. W.QiuR. L.YingR. R.TangY. T.TangL.FangX. H. (2011). The differentially-expressed proteome in Zn/Cd hyperaccumulator Arabis paniculata Franch. in response to Zn and Cd. Chemosphere82, 321–328. 10.1016/j.chemosphere.2010.10.030
480
ZhenyanH. E.JiangchuanL. I.ZhangH.MaM. I. (2005). Different effects of calcium and lanthanum on the expression of phytochelatin synthase gene and cadmium absorption in Lactuca sativa. Plant Sci.168, 309–318. 10.1016/j.plantsci.2004.07.001
481
ZhigangA.CuijieL.YuangangZ.YejieD.WachterA.GromesR. (2006). Expression of BjMT2, a metallothionein 2 from Brassica juncea, increases copper and cadmium tolerance in Escherichia coli and Arabidopsis thaliana, but inhibits root elongation in Arabidopsis thaliana seedlings. J. Exp. Bot.57, 3575–3582. 10.1093/jxb/erl102
482
ZhouJ.GoldsbroughP. B. (1994). Functional homologs of fungal metallothionein genes from Arabidopsis. Plant Cell6, 875–884. 10.1105/tpc.6.6.875
483
ZhouZ. S.GuoK.ElbazA. A.YangZ. M. (2009). Salicylic acid alleviates mercury toxicity by preventing oxidative stress in roots of Medicago sativa. Environ. Exp. Bot.65, 27–34. 10.1016/j.envexpbot.2008.06.001
484
ZhuE.LiuD.LiJ. G.LiT. Q.YangX. E.HeZ. L.et al. (2011). Effect of nitrogen fertilizer on growth and cadmium accumulation in Sedum alfredii Hance. J. Plant Nutr.34, 115–126. 10.1080/01904167.2011.531363
485
ZhuX. F.JiangT.WangZ. W.LeiG. J.ShiY. Z.LiG. X.et al. (2012). Gibberellic acid alleviates cadmium toxicity by reducing nitric oxide accumulation and expression of IRT1 in Arabidopsis thaliana. J. Hazard. Mater. 239–240, 302–307. 10.1016/j.jhazmat.2012.08.077
486
ZimeriA. M.DhankherO. P.McCaigB.MeagherR. B. (2005). The plant MT1 metallothioneins are stabilized by binding cadmium and are required for cadmium tolerance and accumulation. Plant Mol. Biol.58, 839–855. 10.1007/s11103-005-8268-3
487
ZobelR. W.KinraideT. B.BaligarV. C. (2007). Fine root diameters can change in response to changes in nutrient concentrations. Plant Soil297, 243–254. 10.1007/s11104-007-9341-2
488
ZornozaP.Sánchez-PardoB.CarpenaR. R. O. (2010). Interaction and accumulation of manganese and cadmium in the manganese accumulator Lupinus albus. J. Plant Physiol.167, 1027–1032. 10.1016/j.jplph.2010.02.011
Summary
Keywords
crop, heavy metal, ionomics, metabolomics, metallophytes, proteomics, transcriptomics, yield
Citation
Singh S, Parihar P, Singh R, Singh VP and Prasad SM (2016) Heavy Metal Tolerance in Plants: Role of Transcriptomics, Proteomics, Metabolomics, and Ionomics. Front. Plant Sci. 6:1143. doi: 10.3389/fpls.2015.01143
Received
14 August 2015
Accepted
02 December 2015
Published
08 February 2016
Volume
6 - 2015
Edited by
Zuhua He, Shanghai Institute for Biological Sciences, China
Reviewed by
Carla Antonio, Instituto de Tecnologia Química e Biológica, Portugal; Dai-Yin Chao, Shanghai Institute of Plant Physiology and Ecology, China
Updates
Copyright
© 2016 Singh, Parihar, Singh, Singh and Prasad.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Vijay P. Singh vijaypratap.au@gmail.com;
This article was submitted to Plant Physiology, a section of the journal Frontiers in Plant Science
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.