- 1Plant Molecular Biology, Department of Biology, Ludwig-Maximilians-University Munich, Munich, Germany
- 2Department of Plant and Environmental Sciences, Copenhagen Plant Science Centre, University of Copenhagen, Copenhagen, Denmark
Construction of a functional Photosystem II (PSII) in cyanobacteria and chloroplasts depends on the action of auxiliary factors, which transiently interact with PSII intermediates during assembly. In addition to a common PSII structure and a conserved set of PSII assembly factors, cyanobacteria, and higher plants have evolved additional, clade-specific assembly factors. Most such factors in cyanobacteria and chloroplasts have been identified by “top-down” approaches (forward and reverse genetics), which involved genetic disruption of individual components in the assembly process and subsequent characterization of the ensuing phenotypic effects on the respective mutant lines/strains. In contrast, a “bottom-up” strategy, based on the engineering of a synthetic bacterium with a plant-type PSII, has the potential to identify all assembly factors sufficient to make a functional plant PSII.
Photosystem II (PSII) is a water-plastoquinone photo-oxidoreductase, which is found in cyanobacteria and their endosymbiotic descendants, the chloroplasts. Light-driven water splitting and subsequent electron transfer steps are carried out with the assistance of non-proteinaceous cofactors. Thus, the PSII monomer harbors a Mn4CaO5 cluster, chloride, bicarbonate, 1-2 hemes, 1 nonheme iron, 35 chlorophyll a molecules, 2 pheophytins, 11 β-carotenes, and 2 plastoquinones (Umena et al., 2011), all of which are embedded in a shell made up of at least 20 proteins (Shen, 2015) that determine their correct positioning and relative orientation. Several PSII-associated lipids have been identified in crystal structures and might also be important for functionality (Mizusawa and Wada, 2012; Kansy et al., 2014). The structural core of PSII is conserved between chloroplasts and cyanobacteria (Allen et al., 2011). However, the oxygen-evolving complex in cyanobacteria contains subunits U and V, which are replaced by Q, R, P, and Tn in higher plants (Bricker et al., 2012). Furthermore, in contrast to the soluble, peripherally attached phycobilisomes found in cyanobacteria, green photosynthetic eukaryotes have evolved integrated light-harvesting complexes and lack phycobilisomes (Hohmann-Marriott and Blankenship, 2011; Figure 1A).
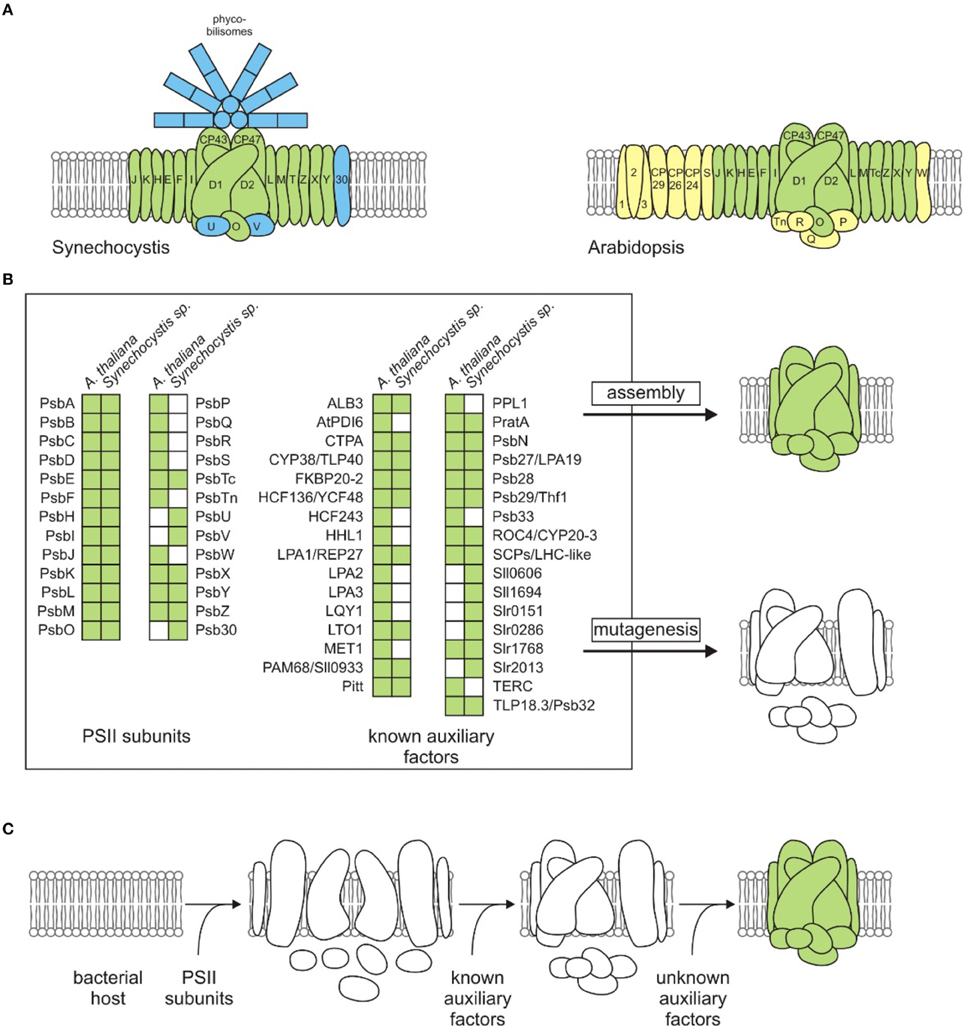
Figure 1. Strategies for identifying auxiliary factors in PSII assembly. (A) Subunit compositions of cyanobacterial (Synechocystis) and plant thylakoid (Arabidopsis) PSII complexes. Components shown in blue or yellow are only found in Synechocystis or Arabidopsis, respectively. Light-harvesting proteins Lhcb1, 2 and 3 are labeled with numbers (1, 2, and 3). (B) PSII subunits and known PSII auxiliary factors. The list of auxiliary factors has been adapted from Nickelsen and Rengstl (2013) and Järvi et al. (2015), and the recently identified auxiliary factors MET1 (Bhuiyan et al., 2015) and Slr0151 (Yang et al., 2014) have been added. Note that for simplicity, no distinction has been made between factors necessary for assembly, repair or supercomplex formation, and Deg/FtsH proteases are not listed. The classical strategy for study of the assembly process is based on mutagenesis of single components in an intact system. Subsequent analyses focus on the identification of a specific loss of function in a photosynthetic organism. Green and white squares indicate presence and absence of a component listed for each organism, respectively. (C) Generation of a synthetic, bacterial host strain carrying a functional plant-type PSII. The design of the synthetic approach largely depends on the choice of the host organism. In the case of a cyanobacterial host, endogenous PSII subunits or auxiliary factors must be deleted or replaced by the plant-type subunits/factors, whereas the presence of essential cofactors (see text), in addition to subunits and auxiliary factors, must be assured for the assembly of a functional, plant-type PSII in a non-photosynthetic bacterium. White and green PSII cartoons in (B,C) indicate un- or partially assembled and fully assembled PSII complexes, respectively.
In accordance with its structural complexity, the assembly of PSII is an elaborate and highly coordinated process, which depends on the action of a network of assembly factors and the fabrication of distinct metastable modules during the course of assembly (see for reviews on this topic: Nixon et al., 2010; Nickelsen and Rengstl, 2013; and papers in this special issue of Frontiers in Plant Science). Several modules common to cyanobacterial and chloroplast PSII assembly processes have been described and are characterized by transient binding of specific assembly factors. An additional level of complexity arises from the fact that PSII is susceptible to photodamage, with subunit D1 being the primary target (Kato and Sakamoto, 2009), as replacement of damaged D1 entails partial disassembly of PSII and reassembly of a functional complex. This repair mechanism features some distinct intermediates, but otherwise involves steps and assembly factors that are shared with the de novo assembly pathway (Järvi et al., 2015).
It has become increasingly clear in recent years that the set of assembly factors is largely conserved between cyanobacteria and chloroplasts (Nickelsen and Rengstl, 2013). However, higher plants have extended their inventory during evolution, giving rise to new, plant-specific factors, such as the D1 stabilization factor HCF243 (Zhang et al., 2011) or the repair factors PPL1 and LQY1 (Ishihara et al., 2007; Lu et al., 2011; Figure 1B). Furthermore, the consequences of disruption of conserved auxiliary factors sometimes differ between the two systems. For instance, Arabidopsis PAM68 and its cyanobacterial counterpart (Armbruster et al., 2010), as well as HCF136 (Meurer et al., 1998) and the cyanobacterial homolog YCF48 (Komenda et al., 2008), participate in the early steps of PSII assembly, but their absence has more severe effects on photosynthesis in plants. Thus, to understand the evolutionary diversification of assembly factors, it is crucial to study chloroplast and cyanobacterial PSII assembly in parallel.
Classical or “Top-down” Methods of Identifying PSII Assembly Factors
Extensive efforts have been made over the past several decades to identify auxiliary factors involved in PSII assembly, and a large number have been found by screening Synechocystis sp. PCC 6803 (hereafter Synechocystis), Chlamydomonas reinhardtii, or Arabidopsis thaliana (hereafter Arabidopsis) mutant collections for PSII-defective mutants. This type of genetic approach has emerged as a powerful strategy, since single components of an initially intact system can be functionally deleted without directly altering other elements in the assembly process. Subsequent studies then allow the in-depth characterisation of the effects of the loss of a specific function in the assembly process.
Forward Genetic Screens
A classical way to screen for mutants defective in PSII is based on the observation of a high-chlorophyll fluorescence (HCF) phenotype in mutagenized Arabidopsis plants, which can be recognized when leaves are exposed to UV light in the dark (Meurer et al., 1996). Several assembly factors, including HCF136 and HCF243 (stabilization of D1 and assembly of the reaction center), LPA1 (D1 integration), LPA2 and LPA3 (integration of CP43) were identified in this manner (Meurer et al., 1998; Peng et al., 2006; Ma et al., 2007; Cai et al., 2010; Zhang et al., 2011). The sensitivity of this screening method for PSII mutants can be further increased by measuring photosynthetic parameters of large mutant collections under varying growth conditions using automatic pulse-amplitude modulation (PAM) of chlorophyll fluorescence (Varotto et al., 2000) or imaging PAM technologies (Ajjawi et al., 2010). This type of approach has led to the identification of several novel factors such as LPA19, PAM68, LQY1, HHL1, and PSB33 (Armbruster et al., 2010; Wei et al., 2010; Lu et al., 2011; Jin et al., 2014; Fristedt et al., 2015). However, forward screens are both labor-intensive and time-consuming, and more than 90% of nuclear genes in Arabidopsis have at least one homolog (Armisén et al., 2008). In many cases, duplicated genes code for proteins with redundant functions, and for that reason are inaccessible to classical forward screening techniques.
Reverse Genetic Screens
The increasing accumulation of information on entire genomes, transcriptomes, and proteomes in public databases, together with the availability of indexed libraries of Arabidopsis mutant lines with mutations in almost every gene, and the establishment of efficient methods for gene silencing, and genetic engineering by means of homologous recombination in cyanobacteria, have made it possible to apply reverse genetic strategies to both cyanobacterial and eukaryotic photosynthetic model organisms and selectively knock out whole gene families. In one such case, the Synechocystis genome database was searched for TPR (tetratricopeptide repeat) genes (Klinkert et al., 2004) and the ORFs were systematically mutagenized. Of the 22 putative TPR proteins identified, PratA and Pitt were shown to be important for Mn2+ transport to D1 (Stengel et al., 2012) and for early steps in photosynthetic pigment-protein complex formation (Schottkowski et al., 2009), respectively. Interestingly, the green lineage-specific TPR-domain-containing protein MET1 was recently shown to assist in PSII supercomplex formation and in PSII repair (Bhuiyan et al., 2015). A further example of the power of reverse genetics is the screen of the lumenal immunophilin family in Arabidopsis, which comprises at least 16 FKBP (FK-506 binding proteins) and cyclophilins that are known to function as protein chaperones or foldases. Two of these, CYP38/TLP40 and FKBP20-2, were found to be involved in early PSII biogenesis and PSII-LHCII assembly, respectively (Lima et al., 2006; Fu et al., 2007; Sirpiö et al., 2008).
Potential and Limitations of Top-down Approaches
It is obvious that additional PSII assembly auxiliary factors will be identified in forward as well as reverse genetic screens. But the latter are becoming more important owing to major advances in rates of data generation and data mining brought by “omics” technologies. Additionally, methods for large-scale quantification of transient protein interactions of PSII subunits or known PSII auxiliary factors with unknown proteins (Braun et al., 2013) will provide a more thorough understanding of the components and their function in the assembly process.
However, an inherent limitation of top-down approaches is that they allow only those factors that are required for a given process to be identified. To define the suite of assembly factors and additional auxiliary factors sufficient for PSII assembly, an entirely different strategy is needed, in which all structural PSII proteins and auxiliary factors are brought together and the final outcome of their interplay (PSII assembly) can be monitored. This “bottom-up” concept cannot be optimally implemented in commonly used model organisms like Arabidopsis, maize, barley or tobacco, as “brute force” genetic approaches (like shot-gun complementation) are not feasible in these organisms and non-photosynthetic propagation of mutants is difficult to achieve, if at all. Consequently, new tactics have to be adopted to speed up and complement research on PSII assembly. Such efforts must be directed at the discovery of the complete set of auxiliary factors sufficient to make a functional plant-type photosystem and are outlined below.
A Novel “Bottom-up” Approach: Generation of a Synthetic Bacterium with a Functional Plant-type PSII
In this context, microbial cell systems possess unsurpassed advantages over plants in several respects. They are fast growing (with doubling times as short as 20 min), they have small genomes that are easy to manipulate, and do not show genetic compartmentalization. The ideal host system for study of the assembly of a plant-type PSII via a “bottom-up” approach should fulfill the following requirements: (i) be able to synthesize such essential non-proteinaceous cofactors as Chl a, pheophytin, heme and β-carotene, (ii) possess an appropriate electron acceptor for PSII, and (iii) a lipid composition similar to that found in thylakoids, (iv) be capable of heterotrophic growth, and (v) intermediates should accumulate in mutants that are dysfunctional in the assembly process. Notably several cyanobacteria meet virtually all these criteria, which is a reflection of their evolutionary relationship to chloroplasts. However, in the case of a cyanobacterial host like Synechocystis endogenous PSII subunits and known, conserved auxiliary factors must be deleted, because they could interfere with the plant-type PSII subunits or auxiliary factors to be tested. This would entail the use of either a sequential, marker-less deletion strategy (Viola et al., 2014 and references within) or a time-saving, multiplex editing strategy recently described for bacterial strains, but not yet established in polyploid cyanobacteria (Ramey et al., 2015). Alternatively, cyanobacterial PSII genes can be directly replaced by their (appropriately optimized) plant counterparts in order to keep untranslated regulatory regions and operon structures intact (Viola et al., 2014). A plant-type PSII can then be constructed stepwise by mimicking the chloroplast assembly process. Known and normally short-lived assembled modules, for instance the D2-Cyt b559 complex, the reaction-center complex lacking both CP47 and CP43 or the reaction-center complex lacking CP43 alone (Müller and Eichacker, 1999; Komenda et al., 2004; Dobáková et al., 2007; Nixon et al., 2010), can serve as checkpoints after introducing known plant-type assembly factors into a cyanobacterial strain expressing the respective plant-type subunits found in each module. As the ultimate goal, unknown factors required for the building of each PSII module can be identified by complementation assays with cDNA libraries derived from higher plants (Figure 1C). An initial benchmark for this kind of approach would be the successful assembly of the minimal PSII core of the five subunits D1, D2, α, and β subunits of Cyt b559 and PsbI, which is able to perform charge separation (Nanba and Satoh, 1987).
Critical Aspects
The bottom-up strategy is inherently appealing, but there are significant uncertainties. While top-down strategies rely on the disruption of a single component of a complex system followed by the characterisation of the effects attributable to loss of that component, the success of the bottom-up strategy critically depends on the ability to monitor the completion of each step in the assembly process. This is the vital prerequisite for the identification of novel assembly factors introduced by shot-gun complementation approaches by perhaps only incremental advances in the assembly process. Nevertheless, in light of the tremendously fast progress in sequencing, gene-synthesis and gene-editing technologies, such synthetic biology-related approaches will sooner or later enrich the field of thylakoid assembly research.
Funding
This work was funded by a grant from the Ludwig-Maximilians-University (LMUexcellent) and by the German Science Foundation (DFG, grant LE 1265/20-1).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Paul Hardy for critical comments on the manuscript.
References
Ajjawi, I., Lu, Y., Savage, L. J., Bell, S. M., and Last, R. L. (2010). Large-scale reverse genetics in Arabidopsis: case studies from the Chloroplast 2010 Project. Plant Physiol. 152, 529–540. doi: 10.1104/pp.109.148494
Allen, J. F., de Paula, W. B. M., Puthiyaveetil, S., and Nield, J. (2011). A structural phylogenetic map for chloroplast photosynthesis. Trends Plant Sci. 16, 645–655. doi: 10.1016/j.tplants.2011.10.004
Armbruster, U., Zühlke, J., Rengstl, B., Kreller, R., Makarenko, E., Rühle, T., et al. (2010). The Arabidopsis thylakoid protein PAM68 is required for efficient D1 biogenesis and Photosystem II assembly. Plant Cell 22, 3439–3460. doi: 10.1105/tpc.110.077453
Armisén, D., Lecharny, A., and Aubourg, S. (2008). Unique genes in plants: specificities and conserved features throughout evolution. BMC Evol. Biol. 8:280. doi: 10.1186/1471-2148-8-280
Bhuiyan, N. H., Friso, G., Poliakov, A., Ponnala, L., and Van Wijk, K. J. (2015). MET1 is a thylakoid-associated TPR protein involved in Photosystem II supercomplex formation and repair in Arabidopsis. Plant Cell 27, tpc.114.132787. doi: 10.1105/tpc.114.132787
Braun, P., Aubourg, S., Van Leene, J., De Jaeger, G., and Lurin, C. (2013). Plant protein interactomes. Annu. Rev. Plant Biol. 64, 161–187. doi: 10.1146/annurev-arplant-050312-120140
Bricker, T. M., Roose, J. L., Fagerlund, R. D., Frankel, L. K., and Eaton-Rye, J. J. (2012). The extrinsic proteins of Photosystem II. Biochim. Biophys. Acta 1817, 121–142. doi: 10.1016/j.bbabio.2011.7.006
Cai, W., Ma, J., Chi, W., Zou, M., Guo, J., Lu, C., et al. (2010). Cooperation of LPA3 and LPA2 is essential for Photosystem II assembly in Arabidopsis. Plant Physiol. 154, 109–120. doi: 10.1104/pp.110.159558
Dobáková, M., Tichy, M., and Komenda, J. (2007). Role of the PsbI protein in Photosystem II assembly and repair in the cyanobacterium Synechocystis sp. PCC 6803. Plant Physiol. 145, 1681–1691. doi: 10.1104/pp.107.107805
Fristedt, R., Herdean, A., Blaby-Haas, C. E., Mamedov, F., Merchant, S. S., Last, R. L., et al. (2015). PHOTOSYSTEM II PROTEIN33, a protein conserved in the plastid lineage, is associated with the chloroplast thylakoid membrane and provides stability to Photosystem II supercomplexes in Arabidopsis. Plant Physiol. 167, 481–492. doi: 10.1104/pp.114.253336
Fu, A., He, Z., Cho, H. S., Lima, A., Buchanan, B. B., and Luan, S. (2007). A chloroplast cyclophilin functions in the assembly and maintenance of Photosystem II in Arabidopsis thaliana. Proc. Natl. Acad. Sci. U.S.A. 104, 15947–15952. doi: 10.1073/pnas.0707851104
Hohmann-Marriott, M. F., and Blankenship, R. E. (2011). Evolution of photosynthesis. Annu. Rev. Plant Biol. 62, 515–548. doi: 10.1146/annurev-arplant-042110-103811
Ishihara, S., Takabayashi, A., Ido, K., Endo, T., Ifuku, K., and Sato, F. (2007). Distinct functions for the two PsbP-like proteins PPL1 and PPL2 in the chloroplast thylakoid lumen of Arabidopsis. Plant Physiol. 145, 668–679. doi: 10.1104/pp.107.105866
Järvi, S., Suorsa, M., and Aro, E.-M. (2015). Photosystem II repair in plant chloroplasts—Regulation, assisting proteins and shared components with Photosystem II biogenesis. Biochim. Biophys. Acta 1847, 900–909. doi: 10.1016/j.bbabio.2015.01.006
Jin, H., Liu, B., Luo, L., Feng, D., Wang, P., Liu, J., et al. (2014). HYPERSENSITIVE TO HIGH LIGHT1 Interacts with LOW QUANTUM YIELD OF PHOTOSYSTEM II1 and Functions in Protection of Photosystem II from Photodamage in Arabidopsis. Plant Cell 26, 1213–1229. doi: 10.1105/tpc.113.122424
Kansy, M., Wilhelm, C., and Goss, R. (2014). Influence of thylakoid membrane lipids on the structure and function of the plant Photosystem II core complex. Planta 240, 781–796. doi: 10.1007/s00425-014-2130-2
Kato, Y., and Sakamoto, W. (2009). Protein quality control in chloroplasts: a current model of D1 protein degradation in the Photosystem II repair cycle. J. Biochem. 146, 463–469. doi: 10.1093/jb/mvp073
Klinkert, B., Ossenbühl, F., Sikorski, M., Berry, S., Eichacker, L., and Nickelsen, J. (2004). PratA, a periplasmic tetratricopeptide repeat protein involved in biogenesis of Photosystem II in Synechocystis sp. PCC 6803. J. Biol. Chem. 279, 44639–44644. doi: 10.1074/jbc.M405393200
Komenda, J., Nickelsen, J., Tichı, M., Prášil, O., Eichacker, L. A., and Nixon, P. J. (2008). The cyanobacterial homologue of HCF136/YCF48 is a component of an early Photosystem II assembly complex and is important for both the efficient assembly and repair of Photosystem II in Synechocystis sp. PCC 6803. J. Biol. Chem. 283, 22390–22399. doi: 10.1074/jbc.M801917200
Komenda, J., Reisinger, V., Müller, B. C., Dobáková, M., Granvogl, B., and Eichacker, L. A. (2004). Accumulation of the D2 Protein is a key regulatory step for assembly of the Photosystem II reaction center complex in Synechocystis PCC 6803. J. Biol. Chem. 279, 48620–48629. doi: 10.1074/jbc.M405725200
Lima, A., Lima, S., Wong, J. H., Phillips, R. S., Buchanan, B. B., and Luan, S. (2006). A redox-active FKBP-type immunophilin functions in accumulation of the Photosystem II supercomplex in Arabidopsis thaliana. Proc. Natl. Acad. Sci. U.S.A. 103, 12631–12636. doi: 10.1073/pnas.0605452103
Lu, Y., Hall, D. A., and Last, R. L. (2011). A small zinc finger thylakoid protein plays a role in maintenance of Photosystem II in Arabidopsis thaliana. Plant Cell 23, 1861–1875. doi: 10.1105/tpc.111.085456
Ma, J., Peng, L., Guo, J., Lu, Q., Lu, C., and Zhang, L. (2007). LPA2 is required for efficient assembly of Photosystem II in Arabidopsis thaliana. Plant Cell 19, 1980–1993. doi: 10.1105/tpc.107.050526
Meurer, J., Meierhoff, K., and Westhoff, P. (1996). Isolation of high-chlorophyll-fluorescence mutants of Arabidopsis thaliana and their characterisation by spectroscopy, immunoblotting and northern hybridisation. Planta 198, 385–396. doi: 10.1007/BF00620055
Meurer, J., Plücken, H., Kowallik, K. V., and Westhoff, P. (1998). A nuclear-encoded protein of prokaryotic origin is essential for the stability of Photosystem II in Arabidopsis thaliana. EMBO J. 17, 5286–5297. doi: 10.1093/emboj/17.18.5286
Mizusawa, N., and Wada, H. (2012). The role of lipids in Photosystem II. Biochim. Biophys. Acta 1817, 194–208. doi: 10.1016/j.bbabio.2011.04.008
Müller, B., and Eichacker, L. A. (1999). Assembly of the D1 precursor in monomeric Photosystem II reaction center precomplexes precedes chlorophyll a-triggered accumulation of reaction center II in barley etioplasts. Plant Cell 11, 2365–2377. doi: 10.1105/tpc.11.12.2365
Nanba, O., and Satoh, K. (1987). Isolation of a Photosystem II reaction center consisting of D-1 and D-2 polypeptides and cytochrome b-559. Proc. Natl. Acad. Sci. U.S.A. 84, 109–112. doi: 10.1073/pnas.84.1.109
Nickelsen, J., and Rengstl, B. (2013). Photosystem II assembly: from cyanobacteria to plants. Annu. Rev. Plant Biol. 64, 609–635. doi: 10.1146/annurev-arplant-050312-120124
Nixon, P. J., Michoux, F., Yu, J., Boehm, M., and Komenda, J. (2010). Recent advances in understanding the assembly and repair of Photosystem II. Ann. Bot. 106, 1–16. doi: 10.1093/aob/mcq059
Peng, L., Ma, J., Chi, W., Guo, J., Zhu, S., Lu, Q., et al. (2006). LOW PSII ACCUMULATION1 is involved in efficient assembly of Photosystem II in Arabidopsis thaliana. Plant Cell 18, 955–969. doi: 10.1105/tpc.105.037689
Ramey, C. J., Barón-Sola, Á., Aucoin, H. R., and Boyle, N. R. (2015). Genome engineering in cyanobacteria: where we are and where we need to go. ACS Synth. Biol. 4, 1186–1196. doi: 10.1021/acssynbio.5b00043
Schottkowski, M., Ratke, J., Oster, U., Nowaczyk, M., and Nickelsen, J. (2009). Pitt, a novel tetratricopeptide repeat protein involved in light-dependent chlorophyll biosynthesis and thylakoid membrane biogenesis in Synechocystis sp. PCC 6803. Mol. Plant 2, 1289–1297. doi: 10.1093/mp/ssp075
Shen, J.-R. (2015). The structure of Photosystem II and the mechanism of water oxidation in photosynthesis. Annu. Rev. Plant Biol. 66, 23–48. doi: 10.1146/annurev-arplant-050312-120129
Sirpiö, S., Khrouchtchova, A., Allahverdiyeva, Y., Hansson, M., Fristedt, R., Vener, A. V., et al. (2008). AtCYP38 ensures early biogenesis, correct assembly and sustenance of Photosystem II. Plant J. 55, 639–651. doi: 10.1111/j.1365-313X.2008.03532.x
Stengel, A., Gügel, I. L., Hilger, D., Rengstl, B., Jung, H., and Nickelsen, J. (2012). Initial steps of Photosystem II de novo assembly and preloading with manganese take place in biogenesis centers in Synechocystis. Plant Cell 24, 660–675. doi: 10.1105/tpc.111.093914
Umena, Y., Kawakami, K., Shen, J.-R., and Kamiya, N. (2011). Crystal structure of oxygen-evolving Photosystem II at a resolution of 1.9Å. Nature 473, 55–60. doi: 10.1038/nature09913
Varotto, C., Pesaresi, P., Maiwald, D., Kurth, J., Salamini, F., and Leister, D. (2000). Identification of photosynthetic mutants of Arabidopsis by automatic screening for altered effective quantum yield of Photosystem 2. Photosynthetica 38, 497–504. doi: 10.1023/A:1012445020761
Viola, S., Rühle, T., and Leister, D. (2014). A single vector-based strategy for marker-less gene replacement in Synechocystis sp. PCC 6803. Microb. Cell Fact. 13:4. doi: 10.1186/1475-2859-13-4
Wei, L., Guo, J., Ouyang, M., Sun, X., Ma, J., Chi, W., et al. (2010). LPA19, a Psb27 homolog in Arabidopsis thaliana, facilitates D1 protein precursor processing during PSII biogenesis. J. Biol. Chem. 285, 21391–21398. doi: 10.1074/jbc.M110.105064
Yang, H., Liao, L., Bo, T., Zhao, L., Sun, X., Lu, X., et al. (2014). Slr0151 in Synechocystis sp. PCC 6803 is required for efficient repair of Photosystem II under high-light condition. J. Integr. Plant Biol. 56, 1136–1150. doi: 10.1111/jipb.12275
Keywords: PSII assembly, PSII, PSII complex, assembly factor, synthetic bacterium, forward genetic screen, reverse genetic, chloroplast
Citation: Rühle T and Leister D (2016) Photosystem II Assembly from Scratch. Front. Plant Sci. 6:1234. doi: 10.3389/fpls.2015.01234
Received: 28 October 2015; Accepted: 19 December 2015;
Published: 12 January 2016.
Edited by:
Julian Eaton-Rye, University of Otago, New ZealandReviewed by:
Eva-Mari Aro, University of Turku, FinlandJohnna Roose, Louisiana State University, USA
Copyright © 2016 Rühle and Leister. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dario Leister, bGVpc3RlckBsbXUuZGU=
 Thilo Rühle
Thilo Rühle Dario Leister
Dario Leister