- 1College of Resources and Environmental Sciences, China Agricultural University, Beijing, China
- 2Center for Training, Consultancy, and Technology Transfer, Vietnam Academy of Science and Technology, Hanoi, Vietnam
- 3Stockbridge School of Agriculture, University of Massachusetts Amherst, Amherst, MA, USA
- 4Institute of Resource and Regional Planning, Chinese Academy of Agricultural Sciences, Beijing, China
As the demands for nanotechnology and nanoparticle (NP) applications in agriculture increase, the ecological risk has drawn more attention because of the unpredictable results of interactions between NPs and transgenic crops. In this study, we investigated the effects of various concentrations of Fe2O3 NPs on Bt-transgenic cotton in comparison with conventional cotton for 10 days. Each treatment was conducted in triplicate, and each experiment was repeated three times. Results demonstrated that Fe2O3 NPs inhibited the plant height and root length of Bt-transgenic cotton and promoted root hairs and biomass of non-transgenic cotton. Nutrients such as Na and K in Bt-transgenic cotton roots increased, while Zn contents decreased with Fe2O3 NPs. Most hormones in the roots of Bt-transgenic cotton increased at low Fe2O3 NP exposure (100 mg⋅L-1) but decreased at high concentrations of Fe2O3 NPs (1000 mg⋅L-1). Fe2O3 NPs increased the Bt-toxin in leaves and roots of Bt-transgenic cotton. Fe2O3 NPs were absorbed into roots, then transported to the shoots of both Bt-transgenic and non-transgenic cottons. The bioaccumulation of Fe2O3 NPs in plants might be a potential risk for agricultural crops and affect the environment and human health.
Introduction
Iron oxide (Fe2O3), the most common oxide of iron, has important magnetic properties. Iron (III) oxide is a convenient compound for the general study of polymorphism and the magnetic and structural phase transitions of NPs. Fe2O3 NPs can be applied in the fields of photoelectrochemistry (such as solar energy conversion and water splitting) and photocatalysts for the removal of organic and inorganic species from aqueous or gas phases (Chirita and Grozescu, 2009). Ali et al. (2012) reported that the addition of Fe2O3 NPs in cement could improve the strength and water permeability of the specimens. Introducing Fe2O3 NPs into soil could significantly increase root elongation and photosynthesis rate in soybean as compared with bulk Fe2O3. Similar results have also been reported for rice seedlings (Oryza sativa L. var. Koshihikari) treated with 500, 1000, and 2000 mg⋅L-1γ-Fe2O3 NPs when compared with the control and bulk treatments (Alidoust and Isoda, 2014). Thus, nano-size effects might enhance plant growth relative to bulk metal. With the development of nanotechnology, a rapidly growing body of concerns has been raised regarding the potential risks and negative effects of NPs on the environment and human health.
The toxicity of metal NPs is not clearly known to living organisms, but previous studies have shown that nanotoxicity is generally affected by unique properties (particle size, shape, and surface properties) (Crane et al., 2008; Navarro et al., 2008). Nanotoxicity has been tested using various organisms, including bacteria, algae, protozoa, plants, and fish (U.S. EPA, 1993). The metabolic processes in living organisms have been investigated to assess its toxicity to the environment (Bitton, 1999). However, in the plant kingdom, investigation of phenotypic differences, seed germination, and plant biomass in response to NP exposure could be one of the most effective ways to assess NP toxicity (Wang and Liu, 2001; Boutin et al., 2004; Di Salvatore M, 2008; Parsons et al., 2010). For example, alumina (Al2O3) NPs can cause phytotoxicity by inhibiting root elongation in corn, cucumber, soybean, cabbage, and carrot (Yang and Watts, 2005). This phytotoxicity is also evident for other plant species, including radish, rape canola, ryegrass, lettuce, corn, and cucumber, when treated with multiwall carbon nanotubes, aluminum (Al), alumina (Al2O3), zinc (Zn), and zinc oxide (ZnO) NPs (Lin and Xing, 2007). In addition to NP-induced toxicity in plants, different plant species could respond differently in the same NP exposure. In a study by Li et al. (2014), CeO2 NPs were revealed to have toxic effects on the root biomass of Bt-transgenic cotton under 100 and 500 mg⋅L-1 exposures, but to have no effects on conventional cotton. In addition, Le et al. (2014) reported that SiO2 NPs negatively affected the activities of CAT in the roots of both Bt-transgenic and non-transgenic cottons.
Insect pests can significantly reduce crop yields and subsequently cause economic losses in agriculture all over the world. At present, chemical pesticides are still the major method to control pest damage; however, the disadvantages of chemical pesticide usage in agriculture include the fact that pests becomes resistant to the chemicals and overuse of pesticides can cause serious risks to the environment and human health (Liu et al., 2015). As such, transgenic plants have been widely applied in agriculture for the purpose of controlling pest damage. For example, over expression of Bacillus thuringiensis (Bt) insecticidal protein in cotton can significantly enhance the resistance of cotton to insects and control insect damage (Wang, 2007; Huang et al., 2010; Yu et al., 2014). Currently, Bt-transgenic cotton is widely used in agriculture, and China is the largest cotton producing country in the world (Du, 2001). However, as demand for nanotechnology and NP applications in agriculture increases, the ecological risks are drawing attention because of the unpredictable results in interactions between NPs and transgenic crops.
To our knowledge, this is the first study on the effects of Fe2O3 NPs on Bt-transgenic cotton. In this study, the toxic effects of Fe2O3 NPs on both conventional and Bt-transgenic cottons were investigated from the aspects of plant growth, nutrient levels, hormone levels, and the changes to Bt-toxic protein in Bt-transgenic cotton in the presence of Fe2O3 NPs. Fe2O3 NP accumulation and distribution were also assayed to reveal how NPs are distributed inside cotton. TEM images showed that Fe2O3 NPs were found in the roots of both conventional and transgenic cotton.
Materials and Methods
Characterization of Fe2O3 Nanoparticles
Fe2O3 NPs were purchased from Shanghai Hufeng Bioscience Technology Company (Shanghai City, China). Scanning electron microscopy (JEOL JSM 5600, Japan) was used to determine the morphology of Fe2O3 NPs. TEM images were obtained (JEM 200CX, Japan) at 200 kV. Following Amrut et al. (2010), the samples were prepared by dispersing drops of the colloid on a copper grid, which was then covered with a carbon film, and the solvent was evaporated. Fe2O3 NP suspensions were prepared at a concentration of 2 mg⋅L-1 for measurement of hydrodynamic size and zeta potential (Nicomp 380 DLS Zeta potential/Particle system, Santa Barbara, CA, USA).
Experimental Exposure
Bt-transgenic cotton (Bt-29317) and conventional cotton (Jihe 321) were purchased from the Chinese Academy of Agricultural Sciences, China Agricultural University. Four cotton plants were allowed to acclimatize in a pot containing 2.0 L of nutrient solution for 4 days and then exposed to 0, 100, or 1000 mg⋅L-1 Fe2O3 NPs for 10 days. The nutrient solution was made following previous reports by Li et al. (2014). Each treatment was performed in triplicate, and each experiment was repeated three times. Fe2O3 NPs were dispersed in DI H2O for 30 min using an ultra-sonicator (KQ3200DE) (Le et al., 2014; Li et al., 2014). The experiments were conducted in a greenhouse with natural light, humidity, and temperature at China Agricultural University.
Measurement of Biomass, Plant Height, Root Length, and Root Hairs
The plant height (mm) and root length (mm) were measured from the growth point in shoot to the cotyledon node and from the growth point to the root point, respectively, after 10 days of exposure to Fe2O3 NPs (Le et al., 2014). The root hairs (root) were counted one by one in each cotton plant.
The samples were collected at day 10. The shoots and roots were rinsed with both tap water and DI H2O for 5 min three times and then dried separately at 80°C for 24–36 h until a constant dry weight was obtained. The dry weight was used to determine the effects of the Fe2O3 NPs on plant growth.
Measurement of Nutrient Contents
Nutrient contents were determined using inductivelycoupled plasma (ICP) mass spectrometry (DRC-II) and ICP atomic emission spectroscopy (iCap 6000) (Li et al., 2014). Fine powders (20–30 mg) of oven-dried shoots and roots were digested in 5 mL of HNO3 at room temperature for 24 h; then, 3 mL of H2O2 was added to each sample to further digest samples at 180°C for 4–5 h.
Determination of Hormone Concentration
Extraction and purification of ABA, IAA, ZR, and GA were performed with enzyme-linked immune absorbent assay (ELISA) kits using monoclonal antibodies (Phytodetek, Agdia, Elkhart, IN, USA) as described in He et al. (2005) and Dong et al. (2008). All samples were measured at 405 nm after the purification step (Wang et al., 2012).
Determination of Bt Toxin
Approximately 0.5-g samples of roots and leaves of Bt-transgenic cotton were harvested and stored at -40°C until analysis. The samples were placed in 9.5 mL of extraction solution (1.33 g of Na2CO3, 1.46 g of NaCl, 0.5 g of vitamin C, and 0.25 g of dithiothreitol in 250 mL of DI H2O). The mixture was gently shaken for 30 min before being centrifuged at 4,000 rpm for 15 min. The supernatants were used to analyze the levels of the Bt toxin protein using an ELISA procedure (Rui et al., 2005).
Transmission Electron Microscopy Observation
Fe2O3 NP-treated shoot and root samples of both conventional and Bt-transgenic cotton were harvested and washed with DI H2O at day 10. Each sample was prefixed in 2.5% glutaraldehyde, then washed in 0.1 mol⋅L-1 pH 7.0 phosphate buffer mixed with 1% osmium tetroxide for 2 h before being dehydrated in a graded ethanol series and finally embedded in epoxy resin. The samples for TEM observation were sectioned using the procedure described in Zhang et al. (2012).
Data Analysis
The results are presented as the mean ± SD. One-way analysis of variance was used to calculate statistical analysis (SPSS 22.0 software). A confidence interval of 95% (p < 0.05) was considered significant in all cases.
Results and Discussion
Characterizations of Fe2O3 NPs
Fe2O3 NPs were purchased from Sigma Inc. The advertised diameter was smaller than 50 nm, the specific surface area was from 50 from 245 m2g-1, and the density was 5.25 g⋅cm3; the crystal phase was cubic. Fe2O3 NPs were characterized by scanning electron microscopy (JEOL JSM 5600, Japan) and dynamic light scattering (DLS). In addition, Fe2O3 NPs were prepared at concentration of 2 mg⋅L-1 for measurement of hydrodynamic size (154.3 nm) and zeta potential (-9.27 mV; Nicomp 380 DLS Zeta potential/Particle system, Santa Barbara, CA, USA). Figure 1 gives the SEM image; the diameter was larger than the advertised data.
Effects of Fe2O3 NPs on the Growth of Cotton Plants
In control groups, no significant difference in plant height was observed between conventional and Bt-transgenic cotton. However, a decrease in plant height for Bt-transgenic cotton was shown with both 100 and 1000 mg⋅L exposure doses of Fe2O3 NPs, while similar phenotypic difference was only observed in 1000 mg⋅L Fe2O3 NP-treated conventional cotton (Figure 2A). Although root length in conventional cotton is approximately 12.5% longer than in Bt-transgenic cotton without the Fe2O3 NP exposure, a significant decrease in the root length in 100 mg⋅L Fe2O3 NP-treated transgenic cotton was evident. The opposite phenomenon in Fe2O3 NP-treated conventional cotton was attained (Figure 2B). Similarly, a low exposure dose of Fe2O3 NPs could stimulate development of root hairs in conventional cotton as compared with its respective control group, while no difference was found in transgenic cotton among the treatments in Figure 2C. Regarding fresh biomass of separated shoots and roots in both types of cottons, shoot biomass in conventional cotton was similar among treatments; however, root biomass in Fe2O3 NP-treated conventional cotton was 30.8–41.2% higher relative to its control group (Figures 2D,E). Analysis of shoots and roots biomass in Bt-transgenic cotton showed that Fe2O3 NPs seemed to have no impact on biomass. These results suggest that Bt-transgenic cotton is more sensitive to NP exposure than conventional cotton. A similar result was reported by Li et al. (2014), in which 100 and 500 mg⋅L-1 CeO2 NPs could cause more toxicity to Bt-transgenic cotton by reducing root biomass, while no impact was found in non-transgenic cotton. However, phytotoxicity might be dependent on NP species. For example, as SiO2 NP concentration increased up to 2000 mg⋅L-1, the plant height and biomasses of both conventional and Bt-transgenic cottons significantly decreased (Le et al., 2014). Other NPs could also impact plant biomass, such as ZnO and CuO NPs. Ryegrass (Loliumperenne) biomass was significantly reduced by different concentrations (0–1000 mg⋅L-1) of ZnO NPs. Wang et al. (2015) demonstrated that the root length and biomass of the transgenic rice O. sativa (OsCDC2 and OsCYCD genes, which play important roles in controlling the cell cycle during plant development) were inhibited by 5 mg⋅L-1 CuO NPs and 5 mg⋅L-1CuO bulk particles after 72-h exposure.
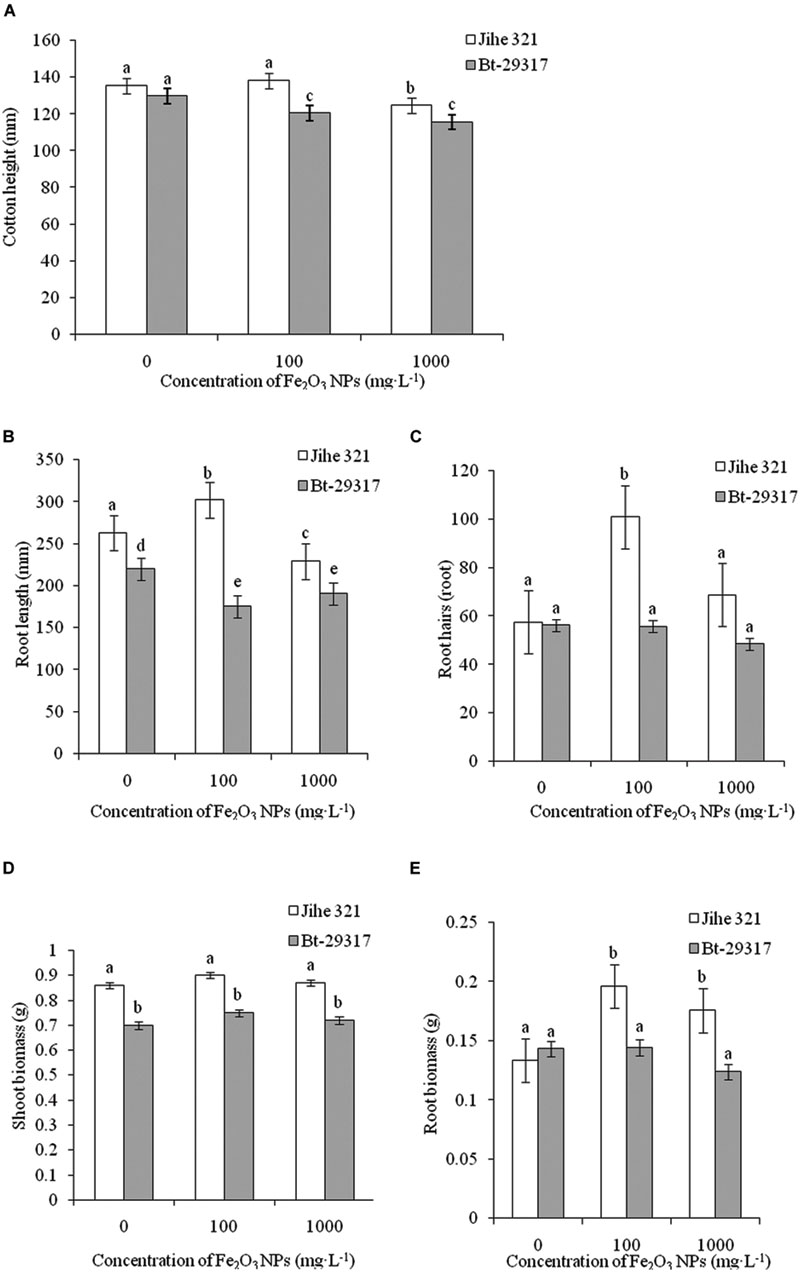
FIGURE 2. Effects of Fe2O3 NPs on the growth of cotton plants. (A–E) Represents plant height, root length, root hairs, shoot biomass, and root biomass, respectively. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposures, and different small letters for the same Fe2O3 NP concentration indicate a significant difference at p < 0.05 level between Bt-transgenic and non-transgenic cotton.
Effects of Fe2O3 NPs on Nutrient Contents in Cotton Plants
Nutrient contents (macronutrients and micronutrients) in both types of cotton treated by Fe2O3 NPs were investigated. K is one of the most important micro nutrients in plants. K contents in both types of cottons were increased in the 100 mg⋅L-1 Fe2O3 NP treatment, although this increase was not statistically significant in Bt-transgenic cotton compared with its corresponding control (Figure 3A). Similar trends were also observed for Ca contents in shoots (Figure 3C). Fe2O3 NPs inhibited Na uptake in shoots of conventional cotton at both 100 mg⋅L-1 and 1000 mg⋅L-1, whereas an increase in Na in the shoots was found for 100 mg⋅L-1 Fe2O3 NP-treated Bt-transgenic cotton (Figure 3B). Cotton type could also determine the nutrient content. For example, in the absence of Fe2O3 NPs, Mg content in conventional cotton was approximately half that of transgenic cotton. When treated with various concentrations of Fe2O3 NPs, increases in Mg were observed in conventional cotton shoots treated with Fe2O3 NPs, whereas a decrease in Mg in 100 mg⋅L Fe2O3 NP-treated Bt-transgenic shoots was found (Figure 3D). Similar results were also found in B content in both conventional and transgenic cottons (Figure 3I). Fe2O3 NPs significantly decreased Mo contents in shoots of both conventional and Bt-transgenic cottons (Figure 3G). Other elemental contents, including Mn, Zn, P, and Cu (Figures 3E,F,H,J), showed no change in either type of plant in the presence of Fe2O3 NPs. These results suggest that Fe2O3 NPs could more seriously interrupt nutrient uptake in Bt-transgenic cotton as compared with conventional cotton.
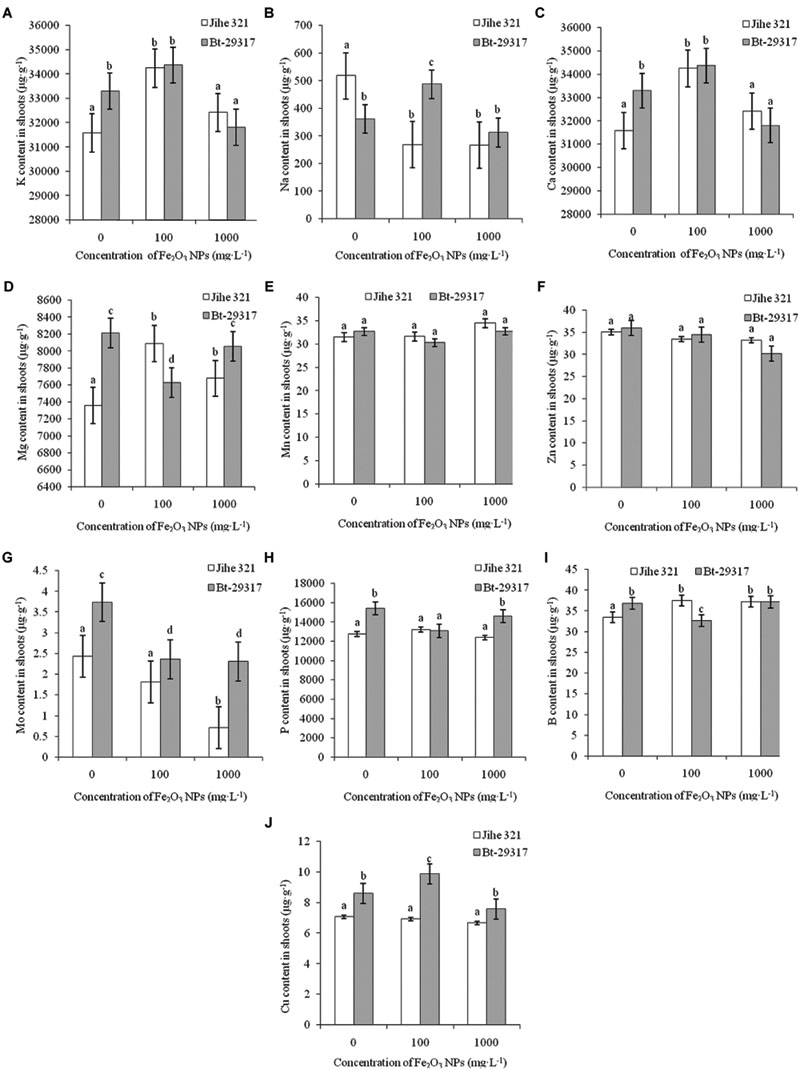
FIGURE 3. Effects of Fe2O3 NPs on nutrient contents in the shoots of cotton plants. (A–J) Shows the levels of K, Na, Ca, Mg, Mn, Zn, Mo, P, B, and Cu in Fe2O3 NPs treated shoots of the conventional and the Bt-transgenic cottons, respectively. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposures, and different small letters for the same Fe2O3 NP concentration indicate a significant difference at p < 0.05 level between Bt-transgenic and non-transgenic cotton.
Figure 4 shows the trend of nutrient uptake in roots of both types of cotton. Fe2O3 NPs significantly enhanced K uptake in the roots of both Bt-transgenic and non-transgenic cotton (Figure 4A). A significant increase in Na content was only observed in the roots of Bt-transgenic cotton in Figure 4B. Fe2O3 NPs only caused a decrease in Ca uptake in conventional cotton, whereas 100 mg⋅L-1 Fe2O3 NPs enhanced Ca content in Bt-transgenic root (Figure 4D). The opposite results were found for Mg content in both types of cotton in Figure 4D. In conventional cotton, Fe2O3 NPs significantly stimulated Mg uptake at both concentrations, while a decrease in Mg content was evident for Bt-transgenic cotton relative to its corresponding control. No significant effects of Fe2O3 NPs were seen for Mn content in roots, regardless of type of cotton, although conventional cotton could take up more Mn than Bt-transgenic cotton in the absence of Fe2O3 NPs (Figure 4E). Fe2O3 NPs decreased the Zn content in the roots of Bt-transgenic cotton, while no significant difference was reported in Zn content for conventional cotton roots (Figure 4F). The presence of 100 mg⋅L-1 of Fe2O3 NPs could enhance Cu uptake in the roots of both types of cottons, and Cu contents in both plants slightly decreased at 1000 mg⋅L-1 (Figure 4J). Fe2O3 NPs decreased Mo contents in the roots of both conventional and Bt-transgenic cottons at 1000 mg⋅L-1 (Figure 4G). P contents were not affected by Fe2O3 NPs (Figure 4H), except for 1000 mg⋅L-1 Fe2O3 NP-treated conventional cotton, whose P content was slightly lower relative to its corresponding control group. Fe2O3 NPs had no impact on B content in roots, regard less of plant types (Figure 4I).
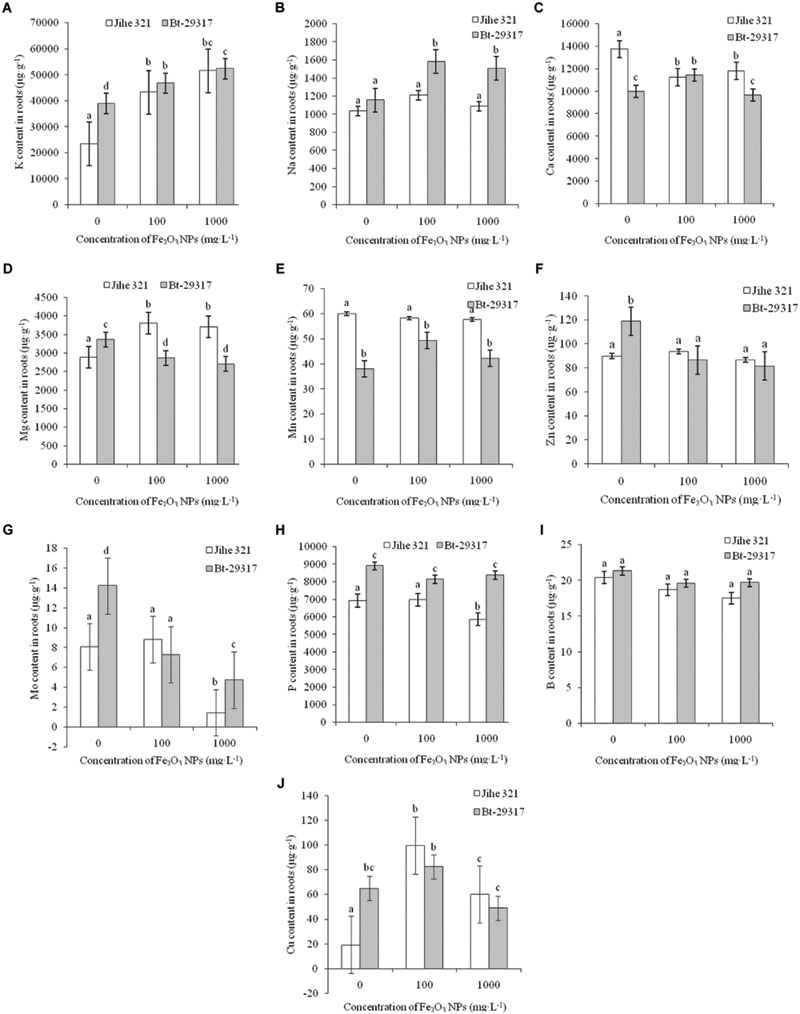
FIGURE 4. Effects of Fe2O3 NPs on nutrient contents in the roots of cotton plants. (A–J) Shows the levels of K, Na, Ca, Mg, Mn, Zn, Mo, P, B, and Cu in Fe2O3 NPs treated roots of the conventional and the Bt-transgenic cottons, respectively. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposures, and different small letters for the same Fe2O3 NP concentration indicate a significant difference at p < 0.05 level between Bt-transgenic and non-transgenic cotton.
Effects of Fe2O3 NPs on Hormone Concentration in Cotton Plants
The effects of Fe2O3 NPs on plant hormones in conventional and Bt-transgenic cottons are shown in Figures 5 and 6. IAA contents in leaves of both types of cottons significantly increased (p < 0.05) at an exposure dose of 100 mg⋅L-1 Fe2O3 NPs, but no change was found at 1000 mg⋅L-1Fe2O3 NP treatment (Figure 5A). A decrease in ABA concentration was only found in the Fe2O3 NP-treated conventional cotton, while Fe2O3 NPs had no impact on ABA concentrations in the leaves of Bt-transgenic cotton. In addition, no significant difference in ABA concentration was seen between Bt-transgenic and non-transgenic cotton leaves under Fe2O3 NP exposure (Figure 5B). Similar results were found for ZR concentration in the leaves of both cotton plants in Figure 5D. The GA content in the leaves of conventional cotton declined for both Fe2O3 NPs treatments, whereas it significantly increased in the leaves of Bt-transgenic cotton (Figure 5C).
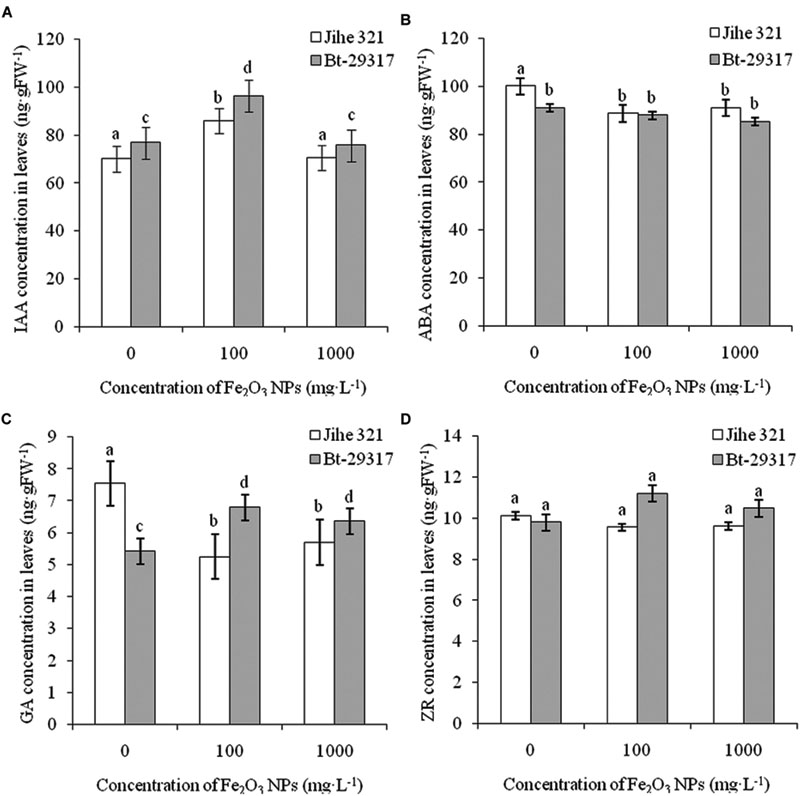
FIGURE 5. Effects of Fe2O3 NPs on hormone concentration in leaves of cotton plants. The concentrations of IAA, AA, GA, and ZR in Fe2O3 NPs treated shoots are shown in A–D, respectively. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposures, and different small letters for the same Fe2O3 NP concentration indicate a significant difference at p < 0.05 level between Bt-transgenic and non-transgenic cotton.
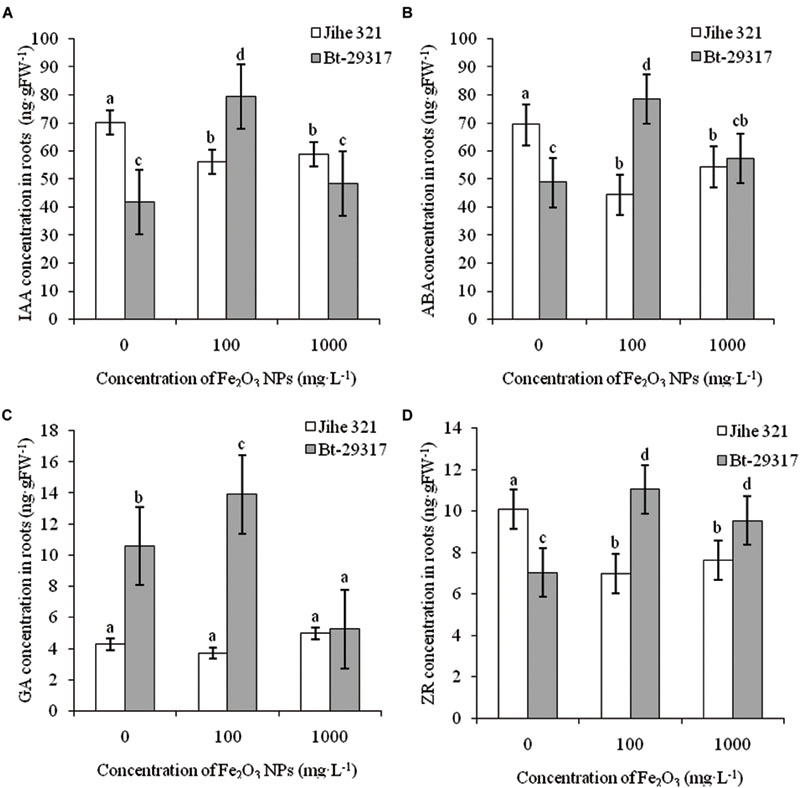
FIGURE 6. Effects of Fe2O3 NPs on hormone concentration in roots of cotton plants. The concentrations of IAA, AA, GA, and ZR in Fe2O3 NPs treated roots are shown in A–D, respectively. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposures, and different small letters for the same Fe2O3 NP concentration indicate a significant difference at p < 0.05 level between Bt-transgenic and non-transgenic cotton.
Fe2O3 NPs enhanced the concentrations of all four plant hormones (including IAA, ABA, GA, and ZR) in the roots of Bt-transgenic cotton, especially at 100 mg⋅L-1 Fe2O3 NP exposure. However, IAA, ABA, and ZR concentrations in the roots of Fe2O3 NP-treated conventional cotton significantly decreased. In addition, Fe2O3 NPs had no impact on GA concentration in the roots of conventional cotton (Figure 6). This suggests that the root hormone concentrations in conventional and Bt-transgenic cotton had different responses to Fe2O3 NP exposure. Le et al. (2014, 2015) reported that IAA and ABA concentrations in the roots of conventional and Bt-transgenic cottons were altered by SiO2 and CeO2 NPs. Gui et al. (2015) reported that phytohormones such as IAA and ABA intransgenic and non-transgenic rice (O. sativa) were affected by 2, 20, and 200 mg⋅L-1Fe2O3 NP exposures. IAA stimulates growth processes such as cell elongation and division, whereas ABA controls plant senescence and responses to stress (Davies, 1995; Mok and Mok, 2001). Thus, our results further demonstrate that NPs could have negative effects on the plant growth process, development, and senescence by manipulating the phytohormone concentration in plants.
Fe contents in the Shoots and Roots of Cotton Plants
Figure 7 shows the total Fe contents in shoots and roots of both conventional and Bt-transgenic cotton treated with two concentrations of Fe2O3 NPs. No difference was seen in Fe contents in both cotton shoots for treatment with 100 mg⋅L-1 Fe2O3 NPs when compared with the control group. However, as the concentration of Fe2O3 NPs increased to 1000 mg⋅L-1, Fe contents in both cotton varieties were significantly higher than the control groups, and Fe content in the Bt-transgenic cotton was significantly higher than that in conventional cotton (Figure 7A). Fe contents in roots of both Fe2O3 NP-treated cottons are shown in Figure 7B. Fe content in the roots of Bt-transgenic cotton treated with 1000 mg⋅L-1 Fe2O3 NPs was 5.30 times higher than in its corresponding control, while this value was 2.8 times for 1000 mg⋅L-1 Fe2O3 NP-treated conventional cotton as compared with its corresponding control. In addition, Fe content in Bt-transgenic cotton roots was 1.27 times higher than that of conventional cotton at 1000 mg⋅L-1 Fe2O3 NP treatment. This indicates that the Bt-transgenic cotton was capable of taking up more Fe relative to the conventional cotton, which agrees with a previous study (Li et al., 2014) in which CeO2 NPs aggregates more easily penetrated into the roots of Bt-transgenic cotton than conventional cotton. A small amount of Fe2O3 NPs was transported to the shoots. Our results are in agreement with previous studies that showed that CuO NPs were taken up by maize through the root system (Wang et al., 2012) and CeO2 and SiO2 NPs were transported to the shoots from the cotton root system (Le et al., 2014; Li et al., 2014).
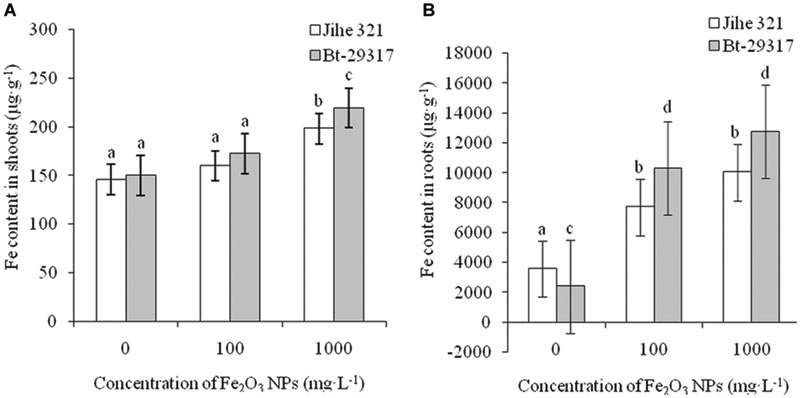
FIGURE 7. Fe content in the shoots and roots of cotton plants. The Fe contents in the shoots and roots of both conventional and Bt-transgenic cottons are shown in A,B, respectively. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposures, and different small letters for the same Fe2O3 NP concentration indicate a significant difference at p < 0.05 level between Bt-transgenic and non-transgenic cotton.
Transmission electron microscopy images of the root sections of Bt-transgenic and non-transgenic cotton show the presence of dark dots (particles) primarily localized in the endodermis and vascular cylinder with 1000 mg⋅L-1 Fe2O3 NP exposure (Figure 8). The absorption of Fe2O3 NPs and their aggregation in the roots of both conventional and Bt-transgenic cottons are evident. Most Fe2O3 NPs were found in the root outer epidermis, and only a few NPs were localized in the intercellular spaces. Thus, it can be concluded that the Fe2O3 NPs could enter the endodermis and vascular cylinder of both Bt-transgenic and non-transgenic cotton. Previous studies also demonstrated that SiO2 and CeO2 NPs were localized in the intercellular spaces of both types of cotton (Le et al., 2014; Li et al., 2014). ZnO NPs were also observed in the endodermis and vascular cylinder of ryegrass roots (Lin and Xing, 2008).
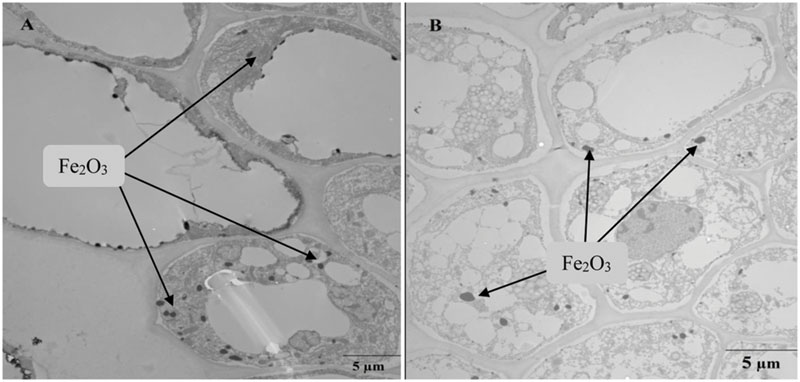
FIGURE 8. Transmission electron microscopy images of root sections of non-transgenic cotton (A) and Bt-transgenic cotton (B) plants after 10 days of treatment with Fe2O3 NPs.
Effects of Fe2O3 NPs on Bt Toxin in Bt-Transgenic Cotton
Adamczyk and Sumerford (2001) reported that the performance of Bt genes for controlling target insect pests varies according to cotton variety, age of plant (Wan et al., 2005), part of plant (Abel and Adamczyk, 2004), type of gene, and insertion site of the gene into the DNA of target plants (Gore et al., 2001; Gore and Adamczyk, 2004; Jackson et al., 2004). Bt toxin is the product of an exogenous Bt gene, whose concentration in shoots is the most important index for evaluating the insect resistant ability of Bt cotton; however, if the root expresses more Bt toxin, it can negatively affect the soil ecological system (Saxena et al., 1999; Obrycki et al., 2001; Fu et al., 2012). Figure 9 shows the expression of Bt toxin in leaves and roots of Bt-transgenic cotton in the presence of Fe2O3 NPs. A significant difference in the contents of Bt toxin was seen in both leaves and roots of Bt-transgenic cotton treated with various concentrations of Fe2O3 NPs as compared with the control group. Bt toxin concentrations in leaves and roots were 845.89 and 886.94 ng⋅g-1, respectively, at the exposure dose of 100 mg⋅L-1 Fe2O3 NPs, which were 1.61 and 1.36 times higher than their respective control group. Upon exposure to 1000 mg⋅L-1 Fe2O3 NPs, the Bt toxin in Bt-transgenic cotton leaves and roots were still greater (p < 0.05) than that of the control group, although they were lower than in the lower exposure dose of Fe2O3 NP-treated Bt-transgenic cotton. The expression of Bt toxin in Bt-transgenic cotton leaves increased, especially at 100 mg⋅L-1 Fe2O3 NPs, which could play an important role in resisting insect damage. To reduce the risk of resistance development in target insect pests against Bt cotton and agricultural crops, there is a need to understand variations in the efficiency of Bt genes and the application of NPs in agriculture.
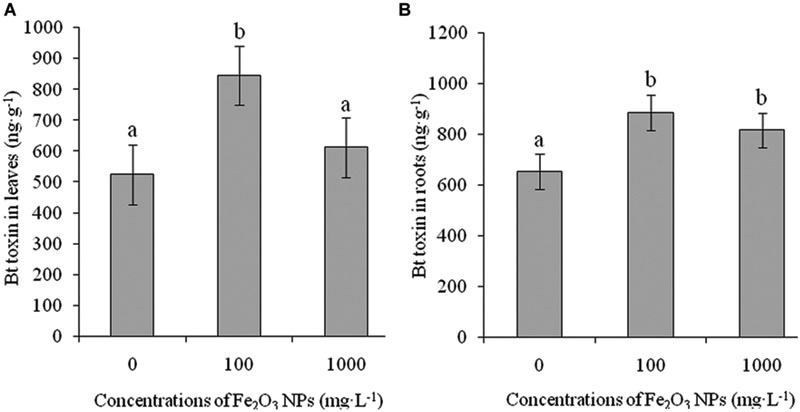
FIGURE 9. Effects of Fe2O3 NPs on Bt toxin in leaves and roots of Bt-transgenic cotton. (A,B) Represents the levels of Bt toxin in the shoots and roots of transgenic cotton. The values are presented as means ± SD. Different small letters for the same cultivar indicate a significant difference at p < 0.05 level between control and Fe2O3 NP exposure.
Conclusion
The present study demonstrated that Fe2O3 NPs could inhibit the plant height and root length of Bt-transgenic cotton, as well as promote the root hairs and biomass of conventional cotton. The effects of Fe2O3 NPs on nutrient contents in the shoots and roots of both types of cotton were investigated. Fe2O3 NPs enhanced Na content in the roots of Bt-transgenic plants, and similar results were observed for K contents in the roots of both Bt-transgenic and non-transgenic cotton. Zn contents in the roots of Bt-transgenic cotton decreased upon Fe2O3 NP exposure, while Cu contents in the roots of both types of cotton increased upon exposure to 100 mg⋅L-1Fe2O3 NPs and decreased at 1000 mg⋅L-1 Fe2O3 NPs. Responses of hormone concentrations in the presence of Fe2O3 NPs differed between the leaves and roots of both types of cottons. Most hormones in the roots of Bt-transgenic cotton increased at low Fe2O3 NP exposure (100 mg⋅L-1), but decreased at a high concentration of Fe2O3 NPs (1000 mg⋅L-1). In addition, Bt-toxin in the leaves and roots of both Bt-transgenic and non-transgenic cotton increased upon NP exposure. TEM images shows that Fe2O3 NPs were evident in the root sections of both Bt-transgenic and non-transgenic cotton, and Fe contents in the shoots and roots increased with increasing exposure doses of Fe2O3 NPs. The present study illustrates the bioaccumulation of Fe2O3 NPs in plants, which might have potential risks for agricultural crops and affect the environment and human health.
Author Contributions
LN and YR conceived and designed the experiment. CM provided scientific expertise. LN, CM, YR, WC, YD, LL, and BX performed the experiments and analyzed the data. LN, CM, and YR wrote and revised the paper. All authors have read and approved the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The project was supported by the National Natural Science Foundation of China (No U1401234, 41130526, and 41371471). The authors gratefully acknowledge the help of Ouyang Li from Peking University in determining nutrients by ICP-MS – We also acknowledge assistance with plant hormone determination provided by Professor Wang Baomin of the college of Agriculture and Biotechnology, China Agricultural University.
Abbreviations
ABA, abscisic acid; DI H2O, deionized water; GA, gibberellic acid; h, hours; IAA, indole-3-acetic acid; NPs, Nanoparticles; SD, standard deviation; TEM, transmission electron microscopy; ZR, trans-zeatin riboside.
References
Abel, C. A., and Adamczyk, J. J. Jr. (2004). Relative concentration of cry1a in maize leaves and cotton bolls with diverse chlorophyll content and corresponding larval development of fall armyworm (lepidoptera: noctuidae) and southwestern corn borer (lepidoptera: crambidae) on maize whorl leaf profiles. J. Econ. Entomol. 97, 1737–1744.
Adamczyk, J. J. Jr., and Sumerford, D. V. (2001). Potential factors impacting season-long expression of Cry1Ac in 13 commercial varieties of Bollgard® cotton. J. Insect Sci. 1:13.
Ali, K., Nazarib, A., and Khalaja, G. (2012). Effects of Fe2O3 nanoparticles on water permeability and strength assessments of high strength self-compacting concrete. J. Mater. Sci. Technol. 28, 73–82. doi: 10.1016/S1005-0302(12)60026-7
Alidoust, D., and Isoda, A. (2014). Phytotoxicity assessment of c-Fe2O3 nanoparticles on root elongation and growth of rice plant. Environ. Earth Sci. 71, 5173–5182. doi: 10.1007/s12665-013-2920-z
Amrut, S. L., Satish, J. S., Ramchandara, B. P., and Raghumani, S. N. (2010). Synthesis and optical characterization of copper oxide nanoparticles. Adv. Appl. Sci. Res. 1, 36–40.
Boutin, C., Elmegaard, N., and Kjaer, C. (2004). Toxicity testing of fifteen noncrop plant species with six herbicides in a greenhouse experiment: implications for risk assessment. Ecotoxicology 13, 349–369. doi: 10.1023/B:ECTX.0000033092.82507.f3
Chirita, M., and Grozescu, I. (2009). Fe2O3 – Nanoparticles, Physical Properties and their Photochemical and Photoelectrochemical Applications. Romania: Chemical Bulletin of Politehnica University of Timisoara, 54.
Crane, M., Handy, R. D., Garrod, J., and Owen, R. (2008). Ecotoxicity test methods and environmental hazard assessment for engineered nanoparticles. Ecotoxicology 17, 421–437. doi: 10.1007/s10646-008-0215-z
Davies, J. (1995). “The plant hormones: their nature, occurrence, and functions,” in Plant Hormones: Physiology, Biochemistry and Molecular Biology, ed. P. J. Davies (Dordrecht: Kluwer Academic Publishers), 1–12.
Di Salvatore M, Carafa, A. M., and Carratù, G. (2008). Assessment of heavy metals phytotoxicity using seed germination and root elongation tests: a comparison of two growth substrates. Chemosphere 73, 1461–1464. doi: 10.1016/j.chemosphere.2008.07.061
Dong, H., Niu, Y., Li, W., and Zhang, D. (2008). Effects of cotton rootstock on endogenous cytokinins and abscisic acid in xylem sap and leaves in relation to leaf senescence. J. Exp. Bot. 59, 1295–1304. doi: 10.1093/jxb/ern035
Du, M. (2001). Bt transgenic cotton in China: present and prospect. Paper Presented at the 2001 China International Cotton Conference, Guilin.
Fu, Q., Zhang, Y., Huang, W., Hu, H., Chen, D., and Yang, C. (2012). Remaining dynamics of Cry1Ab proteins from transgenic Bt corn in soil. J. Food Agric. Environ. 10, 294–298.
Gore, J., and Adamczyk, J. J. Jr. (2004). Impact of bollworms [Helicoverpazea (Boddie)] on maturity and yield of bollgard cotton. J. Cotton Sci. 8, 223–229.
Gore, J., Leonard, B. R., and Adamczyk, J. J. (2001). Bollworm (Lepidoptera: Noctuidae) survival on ‘Bollgard’ and ‘BollgardII’ cotton flower bud and flower components. J. Econ. Entomol. 94, 1445–1451.
Gui, X., Deng, Y., Rui, Y., Gao, B., Luo, W., Chen, S., et al. (2015). Response difference of transgenic and conventional rice (Oryza sativa) to nanoparticles (γFe2O3). Environ. Sci. Pollut. Res. 22, 17716–17723. doi: 10.1007/s11356-015-4976-7
He, J. T., Shi, Z. H., Yan, J., Zhao, M. P., Guo, Z. Q., and Chang, W. B. (2005). Biotin-avidin amplified enzyme-linked immunosorbent assay for determination of isoflavonedaidzein. Talanta 65, 621–626. doi: 10.1016/j.talanta.2004.07.027
Huang, J., Mi, J., Lin, H., Wang, Z., Chen, R., Hu, R., et al. (2010). A decade of Bt cotton in Chinese fields: assessing the direct effects and indirect extanilities of Bt cotton adoption in China. Sci. China Life Sci. 53, 981–991. doi: 10.1007/s11427-010-4036-y
Jackson, R. E., Bradley, J. R. Jr., Van Duyn, J. W., and Gould, F. (2004). Comparative production of Helicoverpa zea (Lepidoptera: Noctuidae) from transgenic cotton expressing either one or two Bacillus thuringiensis proteins with and without insecticide oversprays. J. Econ. Entomol. 97, 1719–1725. doi: 10.1603/0022-0493-97.5.1719
Le, V. N., Ma, C., Rui, Y., Liu, S., Li, X., Xing, B., et al. (2015). Phytotoxic mechanism of nanoparticles: destruction of chloroplasts and vascular bundles and alteration of nutrient absorption. Sci. Rep. 5:11618. doi: 10.1038/srep11618
Le, V. N., Rui, Y., Gui, X., Li, X., Liu, S., and Han, Y. (2014). Uptake, transport, distribution and Bio-effects of SiO2 nanoparticles in Bt-transgenic cotton. J. Nanobiotechnology 12, 50. doi: 10.1186/s12951-014-0050-8
Li, X., Gui, X., Rui, Y., Ji, W., Van Nhan L, Yu, Z., et al. (2014). Bt-transgenic cotton is more sensitive to CeO2 nanoparticles than its parental non-transgenic cotton. J. Hazard. Mater. 274, 173–180. doi: 10.1016/j.jhazmat.2014.04.025
Lin, D., and Xing, B. (2007). Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ. Pollut. 150, 243–250. doi: 10.1016/j.envpol.2007.01.016
Lin, D., and Xing, B. (2008). Root uptake and phytotoxicity of ZnO nanoparticles. Environ. Sci. Technol. 42, 5580–5585. doi: 10.1021/es800422x
Liu, F., Wang, X. D., Zhao, Y. Y., Li, Y. J., Liu, Y. C., and Sun, J. (2015). Silencing the HaAK gene by transgenic plant-mediated RNAi impairs larval growth of Helicoverpa armigera. Int. J. Biol. Sci. 11, 67–74. doi: 10.7150/ijbs.10468
Mok, W. S., and Mok, M. C. (2001). Cytokinin metabolism and action. Annual Review of Plant Physiology and Plant Molecular Biology. Plant Mol. Biol. 52, 89–118. doi: 10.1146/annurev.arplant.52.1.89
Navarro, E., Baun, A., Behra, R., Hartmann, N. B., Filser, J., Miao, A. J., et al. (2008). Environmental behavior and exotoxicity of engineered nanoparticles to algae, plants, and fungi. Ecotoxicology 17, 372–386. doi: 10.1007/s10646-008-0214-0
Obrycki, J. J., Losey, J. E., Taylor, O. R., and Jesse, L. C. H. (2001). Transgenic insecticidal corn: beyond insecticidal toxicity to ecological complexity. Biosience 51, 353–361. doi: 10.1641/0006-3568(2001)051[0353:TICBIT]2.0.CO;2
Parsons, J. G., Lopez, M. L., Gonzalez, C. M., Peralta-Videa, J. R., and Gardea-Torresdey, J. L. (2010). Toxicity and biotransformation of uncoated and coated nickel hydroxide nanoparticles on mesquite plants. Environ. Toxicol. Chem. 29, 1146–1154. doi: 10.1002/etc.146
Rui, Y.-K., Yi, G.-X., Zhao, J., Wang, B.-M., Li, Z.-H., Zhai, Z.-X., et al. (2005). Changes of Bt toxin in the rhizosphere of transgenic Bt cotton and its influence on soil functional bacteria. World J. Microbiol. Biotechnol. 21, 1279–1284. doi: 10.1007/s11274-005-2303-z
Saxena, D., Flores, S., and Stotzk, G. (1999). Transgenic plants – Insecticidal toxin in root exudates from Bt corn. Nature 402, 480–480. doi: 10.1038/44999
U.S. EPA (1993). Methods for Measuring the Acute Toxicity of Effluents and Receiving Waters to Freshwater and Marine Organisms, 4th Edn. EPA/600/4-90/027F. Washington, DC: U.S. Environmental Protection Agency.
Wan, P., Zhang, Y., Wu, K., and Huang, M. (2005). Seasonal expression profiles of insecticidal protein and control efficacy against Helicoverpa armigera for Bt cotton in the Yangtze River valley of China. J. Econ. Entomol. 98, 195–201. doi: 10.1093/jee/98.1.195
Wang, D. P. (2007). Current status and future strategies for development of transgenic plants in China. J. Integr. Plant Biol. 49, 1281–1283. doi: 10.1007/s00122-014-2371-2
Wang, S., Liu, H., Zhang, Y., and Xin, H. (2015). The effect of CuO NPs on reactive oxygen species and cell cycle gene expression in roots of rice. Environ. Toxicol. Chem. 34, 554–561. doi: 10.1002/etc.2826
Wang, X., and Liu, Y. (2001). Validation of germination rate and root elongation as an indicator to assess phytotoxicity with Cucumis sativus. Chemosphere 44, 1711–1721. doi: 10.1016/S0045-6535(00)00520-8
Wang, Y., Li, B., Du, M., Eneji, A. E., Wang, B., Duan, L., et al. (2012). Mechanism of phytohormone involvement in feedback regulation of cotton leaf senescence induced by potassium deficiency. J. Exp. Bot. 63, 5887–5901. doi: 10.1093/jxb/ers238
Yang, L., and Watts, D. (2005). Particle surface characteristics may play an important role in phytotoxicity of alumina nanoparticles. Toxicol. Lett. 158, 122–132. doi: 10.1016/j.toxlet.2005.03.003
Yu, R., Xu, X., Liang, Y., Tian, H., Pan, Z., Jin, S., et al. (2014). The insect Ecdysone receptor is a good potential target for RNAi-based pest control. Int. J. Biol. Sci. 10, 1171–1180. doi: 10.7150/ijbs.9598
Keywords: fate, phytotoxicity, Fe2O3 nanoparticles, insecticide activity, Bt-transgenic cotton
Citation: Van Nhan L, Ma C, Rui Y, Cao W, Deng Y, Liu L and Xing B (2016) The Effects of Fe2O3 Nanoparticles on Physiology and Insecticide Activity in Non-Transgenic and Bt-Transgenic Cotton. Front. Plant Sci. 6:1263. doi: 10.3389/fpls.2015.01263
Received: 13 May 2015; Accepted: 24 December 2015;
Published: 22 January 2016.
Edited by:
Burkhard Schulz, University of Maryland, USAReviewed by:
Uener Kolukisaoglu, University of Tuebingen, GermanyNaser A. Anjum, University of Aveiro, Portugal
Copyright © 2016 Van Nhan, Ma, Rui, Cao, Deng, Liu and Xing. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yukui Rui, cnVpeXVrdWlAMTYzLmNvbQ==; eXVrdWlydWlAdW1hc3MuY29t
 Le Van Nhan
Le Van Nhan Chuanxin Ma
Chuanxin Ma Yukui Rui
Yukui Rui Weidong Cao4
Weidong Cao4