- Molecular Genetics Lab, Biotechnology Department, Institute of Vegetables and Flowers, Chinese Academy of Agricultural Sciences, Beijing, China
Alterations in leaf adaxial-abaxial (ad-ab) polarity are one of the main factors that influence leaf curvature. In Chinese cabbage, leaf incurvature is an essential prerequisite to the formation of a leafy head. Identifying ad-ab patterning genes and investigating their genetic variation may facilitate elucidation of the mechanisms underlying leaf incurvature during head formation. Comparative genomic analysis of 45 leaf ad-ab patterning genes in Brassica rapa based on 26 homologs of Arabidopsis thaliana indicated that these genes underwent expansion and were retained after whole genome triplication (WGT). We also assessed the nucleotide diversity and selection footprints of these 45 genes in a collection of 94 Brassica rapa accessions that were composed of heading and non-heading morphotypes. Six of the 45 genes showed significant negative Tajima's D indices and nucleotide diversity reduction in heading accessions compared to those in non-heading accessions, indicating that they underwent purifying selection. Further testing of the BrARF3.1 gene, which was one of the selection signals from a larger collection, confirmed that purifying selection did occur. Our results provide genetic evidence that ad-ab patterning genes are involved in leaf incurvature, which is associated with formation of a leafy head, as well as promote an understanding of the genetic mechanism underlying leafy head formation in Chinese cabbage.
Introduction
Chinese cabbage (Brassica rapa L. ssp. pekinensis) is widely cultivated and one of the most consumed vegetable crops in Asia. It has a uniform, compact leafy head composed of several enlarged, winged, and crinkly incurved leaves that have been utilized as highly nutritious food. Because head traits such as size, shape, and weight contribute to yield and quality, these features are considered to be of high commercial value (Yu et al., 2013; Mao et al., 2014).
To produce a leafy head, Chinese cabbage undergoes three developmental stages, namely, seedling, rosette, and heading (Yu et al., 2013). The seedling leaves and rosette leaves are flat, whereas the head leaves within the leafy head show incurvature with a larger abaxial (bottom) surface. In addition, compared to other non-heading B. rapa, Chinese cabbage also shows significant differences in leaf morphology such as leaf shape, size, bulges, lobes, petioles, and blade base angles (Figure 1), indicating that leaf heading is a complex quantitative trait influenced by multiple genes (Yu et al., 2013; Wang et al., 2014). Quantitative trait locus (QTL) mapping of leafy heads using a recombinant inbred line (RIL) population previously identified three candidate genes, BrpGL1, BrpESR1, and BrpSAW1, which regulate development of trichome, petiole, serration, and cell division and possibly contribute to the formation of leafy heads (Yu et al., 2013). The global analysis of the transcriptomes from rosette leaves and folding leaves in Chinese cabbage using RNA-sequencing (RNA-Seq) revealed that the regulation of transcription factors, protein kinases and calcium may play critical roles in controlling leafy head development besides some stimuli, such as carbohydrate levels, light intensity and endogenous hormones (Wang et al., 2012). In addition, because leaf incurvature is an essential prerequisite to leafy head formation (Mao et al., 2014), genetic studies involving leaf incurvature may lead to a better understanding of the molecular mechanism underlying leafy head formation in Chinese cabbage.

Figure 1. Leaf morphology of heading and non-heading B. rapa in mature stage. (A) The leafy head of heading B. rapa (Chinese cabbage), and its incurved leaves from inside to outside. (B) The non-heading B. rapa (pak choi) with flat leaves from inside to outside. Scale bars: 10 cm.
Most plant species have evolved flat leaves that further differentiate into two specialized leaf domains, namely, the adaxial (top) and abaxial surfaces. The adaxial side of the leaf originates closest to the meristem and develops a tightly packed layer of palisade mesophyll cells that facilitate in the capture of light, whereas the abaxial side contains a loosely packed spongy mesophyll layer and a higher density of stomatal pores that assist in gas exchange and regulate transpiration (Braybrook and Kuhlemeier, 2010; Moon and Hake, 2011; Yamaguchi et al., 2012). The developmental mechanism behind leaf ad-ab polarity specification and flat lamina formation has always been of interest to biologists. Several regulators involved in leaf adaxial-abaxial (ad-ab) polarity specification have now been identified. Further studies have also demonstrated that mutual antagonistic interactions between adaxial and abaxial determinants are not only responsible for ad-ab polarity specification, but also define the boundary between the ad-ab domain, in which subsequent flat lamina growth is promoted (Yamaguchi et al., 2012). Loss- or gain-of-function mutations in adaxial or abaxial polarity genes generally result in loss of lamina flattening and an upward- or downward-curling leaf phenotype due to alterations in the ad-ab patterning system (Kidner and Timmermans, 2007, 2010; Yamaguchi et al., 2012).
The inward-curled rosette leaf phenotype of Arabidopsis abaxial polarity mutants exactly resembles the head leaf incurvature underlying the leafy head formation in Chinese cabbage. The HYL1 gene is responsible for the leaf abaxial determinant-miR166 (Han et al., 2004; Wu et al., 2007), and the rosette leaves of Arabidopsis hyl1 mutants are curved inward (Wu et al., 2004; Liu et al., 2011). BcpLH, which is the homolog of the Arabidopsis HYL1 gene that was isolated from Chinese cabbage, has been shown to affect the inward curvature of folding leaves (Yu et al., 2000). Moreover, unlike flat seedling leaves and rosette leaves that mainly function in photosynthesis and respiration, head leaves generally serve as storage organs. These findings prompted us to hypothesize that the incurvature of head leaves is associated with alterations in the leaf ad-ab patterning system.
Leaf ad-ab patterning is regulated by several distinct transcription factor (TF) families, of which some are regulated by two kinds of small RNAs, namely, microRNAs (miR165/166) and trans-acting short interfering RNAs (TAS3-derived tasiR-ARF). In angiosperm models, adaxial polarity is specified by genes of the class III homeodomain-leucine zipper (HD-ZIPIII) family (REV, PHB, PHV, and ATHB8) (McConnell et al., 2001; Emery et al., 2003), Myb and LOB domain transcription factors ASYMMETRIC LEAVES1 (AS1) and AS2 (Lin et al., 2003; Iwakawa et al., 2007), and TAS3-derived tasiR-ARF (Hunter et al., 2006; Chitwood et al., 2007, 2009). On the other hand, KANADI family genes (KAN1–3) (Eshed et al., 2001, 2004; Kerstetter et al., 2001), AUXIN RESPONSE FACTORS (ARF) ETTIN (ETT/ARF3) and ARF4 (Pekker et al., 2005), and miR165/166 small RNAs (Kidner and Martienssen, 2004) are abaxial-specific. YABBY family genes (YAB1–3) were first proposed as abaxial determinants (Stahle et al., 2009). However, recent studies have shown that YABBY genes and WUSCHEL-RELATED HOMEOBOX genes (WOX1 and WOX3) are middle domain-specific and involved not only in adaxial/abaxial patterning and but also in subsequent flat lamina growth (Kidner and Timmermans, 2007; Vandenbussche et al., 2009; Nakata et al., 2012). The production of microRNAs and tasiRNAs requires a number of proteins, which include AGO1, AGO7, SGS3, RDR6, DCL1, DCL4, and SE (reviewed by Moon and Hake, 2011). The regulatory network controlling ad-ab polarity is based on the mutual antagonistic interaction between adaxial and abaxial determinants. The KAN genes are key regulators of abaxial polarity of leaves, whereas HD-ZIPIII genes are major adaxial determinants. KAN and HD-ZIPIII are thought to act antagonistically (Eshed et al., 2004; Huang et al., 2014), whereas AS2 and KAN negatively regulate each other (Lin et al., 2003; Wu et al., 2008). In addition, HD-ZIPIII genes are the targets of miR165/166-mediated gene regulation, and its expression is repressed at the adaxial side. Similar to the HD-ZIPIII genes, the expression of ARF3/4 abaxial genes is regulated by tasiR-ARF (Fahlgren et al., 2006; Kidner and Timmermans, 2010).
Chinese cabbage is a subspecies of B. rapa (AA, 2n = 2x = 20). Both B. rapa and A. thaliana belong to the cruciferae family. B. rapa has undergone additional whole genome triplication (WGT) since its divergence from A. thaliana, followed by extensive gene loss and divergence of three subgenomes (Wang et al., 2011; Cheng et al., 2012a). Although the leaf ad-ab patterning mechanism in A. thaliana has been extensively studied, that in Chinese cabbage and its association with head leaf incurvature have not been investigated. The present study has identified genes correlated to leaf ad-ab polarity establishment in Chinese cabbage by conducting comparative genomic analysis of B. rapa and A. thaliana, as well as analyzed the retention of all ad-ab patterning genes in B. rapa after WGT. We investigated the genetic variation in 45 leaf ad-ab patterning genes in a 94-accession collection, including heading and non-heading morphotypes of B. rapa. We further assessed their footprints of selection by using Tajima's D index and nucleotide diversity (π) among various heading and non-heading accessions. The results of the present study may contribute to the establishment of the genetic mechanism underlying leafy head formation in Chinese cabbage.
Materials and Methods
Database for Ad-Ab Patterning Genes in B. rapa
The sequences of A. thaliana genes that are involved in the ad-ab patterning pathway were downloaded from the TAIR database (http://www.arabidopsis.org/). The B. rapa genome (version 1.5) and gene sequences from BRAD (http://brassicadb.org/brad/) (Cheng et al., 2011) were used to identify the ad-ab patterning genes in B. rapa.
Identification of Homologous Genes Between B. rapa and A. thaliana
The ad-ab patterning gene DNA and protein sequences of A. thaliana and B. rapa were respectively aligned using BLASTN and BLASTP using a cut off E-value of ≤1E-10 and coverage of ≥0.75. Syntenic orthologs between A. thaliana and B. rapa from BRAD (http://brassicadb.org/brad/) were determined based on sequence similarity (cutoff: E ≤ 10-20) and collinearity of flanking genes (Cheng et al., 2012b).
Selection of Accessions
To represent the maximum morphotype diversity of B. rapa, a total of 94 B. rapa accessions were selected from a larger collection that was used in a resequencing project involving SNP genotyping and phenotyping (Table S3). The selected accessions were also classified into two groups based on their phenotypes: 47 heading B. rapa (H-Br) and 47 non-heading B. rapa (NH-Br). The phylogenetic relationship of the 94 accessions has been presented in Zhao et al. (2005). Another 150 H-Br and 150 NH-Br accessions were further collected to confirm the purifying selection signal of BrARF3.1.
π Assessment and Neutrality Testing
By using resequencing and genotyping data from the 94 accessions, genetic diversity indices were calculated. Using the Variscan software (Hutter et al., 2006), neutrality (Tajima's D) (Tajima, 1989) was initially estimated for the entire genome by 10-kb sliding window analysis of all three groups (All, NH-Br, and H-Br), with test statistic values at the negative and positive 5 percentiles of the genome-wide distribution set as thresholds. Then, the π and Tajima's D index of the coding regions of the 45 candidate genes were estimated in all three groups using Variscan.
Availability of Supporting Data
All the 26 genes of A. thaliana referred in this report were retrieved from the TAIR database.
The B. rapa genome sequence (version 1.5), as well as the coding and protein sequences of the 45 ad-ab patterning genes were acquired from the Brassica database (BRAD).
Results
Identification of Leaf Ad-Ab Patterning Genes in B. rapa
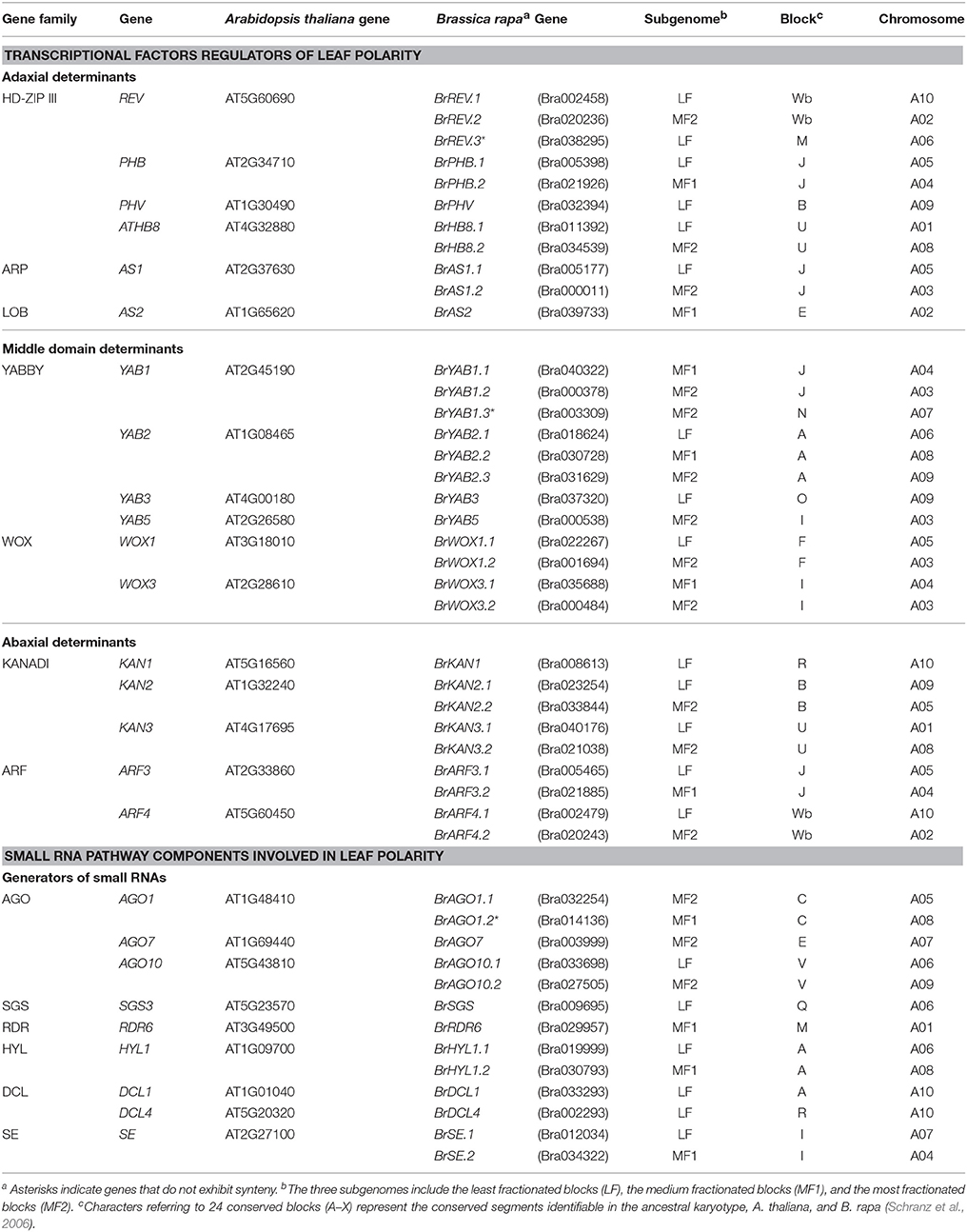
A total of 26 genes involved in leaf ad-ab patterning have been identified in A. thaliana, which include 17 genes encoding for seven TF families of leaf polarity determinants, namely, 6 adaxial determinants, 6 middle domain determinants, and 5 abaxial determinants; the remaining 9 genes are involved in the production and activity of small RNA determinants (miR165/166 and ta-siRNA) (Table 1). By using a combination of syntenic and non-syntenic homology analyses (Guo et al., 2014) and the 26 homologs in A. thaliana, we identified 45 leaf ad-ab patterning genes in the draft genome of B. rapa (v1.5), which consists of a total of 41,020 annotated genes (Wang et al., 2011). Thirty-two of the 45 genes were TF determinants, whereas 13 were responsible for small RNA pathway determinants. Each gene in B. rapa was assigned a name based on its A. thaliana homolog.
Comparative analysis of syntenic orthologs of A. thaliana and B. rapa showed that 42 of the 45 genes were syntenic to 26 genes in A. thaliana. No tandem clusters involving these genes were identified. Wang et al. (2011) inferred three subgenomes in B. rapa, namely, the least fractionated blocks (LF), the medium fractionated blocks (MF1), and the most fractionated blocks (MF2). Based on this subgenome information, 21, 9, and 15 genes were distributed across LF, MF1, and MF2, respectively. Of the 42 syntenic orthologs, 20 were on LF, 8 were on MF1, and 14 were on MF2. The number of syntenic orthologs distributed across LF was more than those in MF1 and MF2, respectively, indicating that the subgenomic distribution of the ad-ab patterning genes is consistent with the gene fractionation status at the whole-genome level.
Expansion of Leaf Ad-Ab Patterning Genes in B. rapa
Only 62% of the A. thaliana orthologs harbored more than one paralogous copy in B. rapa, which was suggestive of the deletion of one or more paralogs via diploidization after WGT. To further observe the retention status of the leaf ad-ab patterning genes after WGT of the B. rapa genome, we investigated the number and ratio of single-copy to multiple-copy paralogous genes. We also used the ratio of the total number of single- to multiple-copy (two and three) paralogous genes (genes in the same tandem array were only counted once) of the entire B. rapa genome as background (1.342; 9175/6836) (Li et al., 2014) to calculate the P-value using Fisher's t-test. The P-value of the TF pathway ad-ab patterning genes (0.42; 5/12) was 0.026, indicating that these were significantly lower than that of the background, whereas no differences between the small RNA pathway ad-ab patterning genes (1.25; 5/4) and total ad-ab patterning genes (0.63; 10/16) and the background were observed (P > 0.05; Table 2). These results were suggestive of over-retention of TF pathway ad-ab patterning genes, but not total or small RNA pathway ad-ab patterning genes in B. rapa.
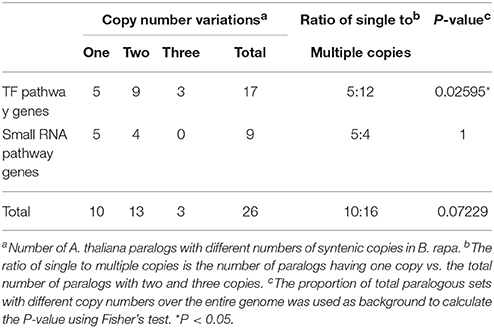
Table 2. Number and ratio of single-copy to multiple-copy paralogs of Arabidopsis ad-ab patterning genes.
Polymorphisms in B. rapa Leaf Ad-Ab Patterning Genes
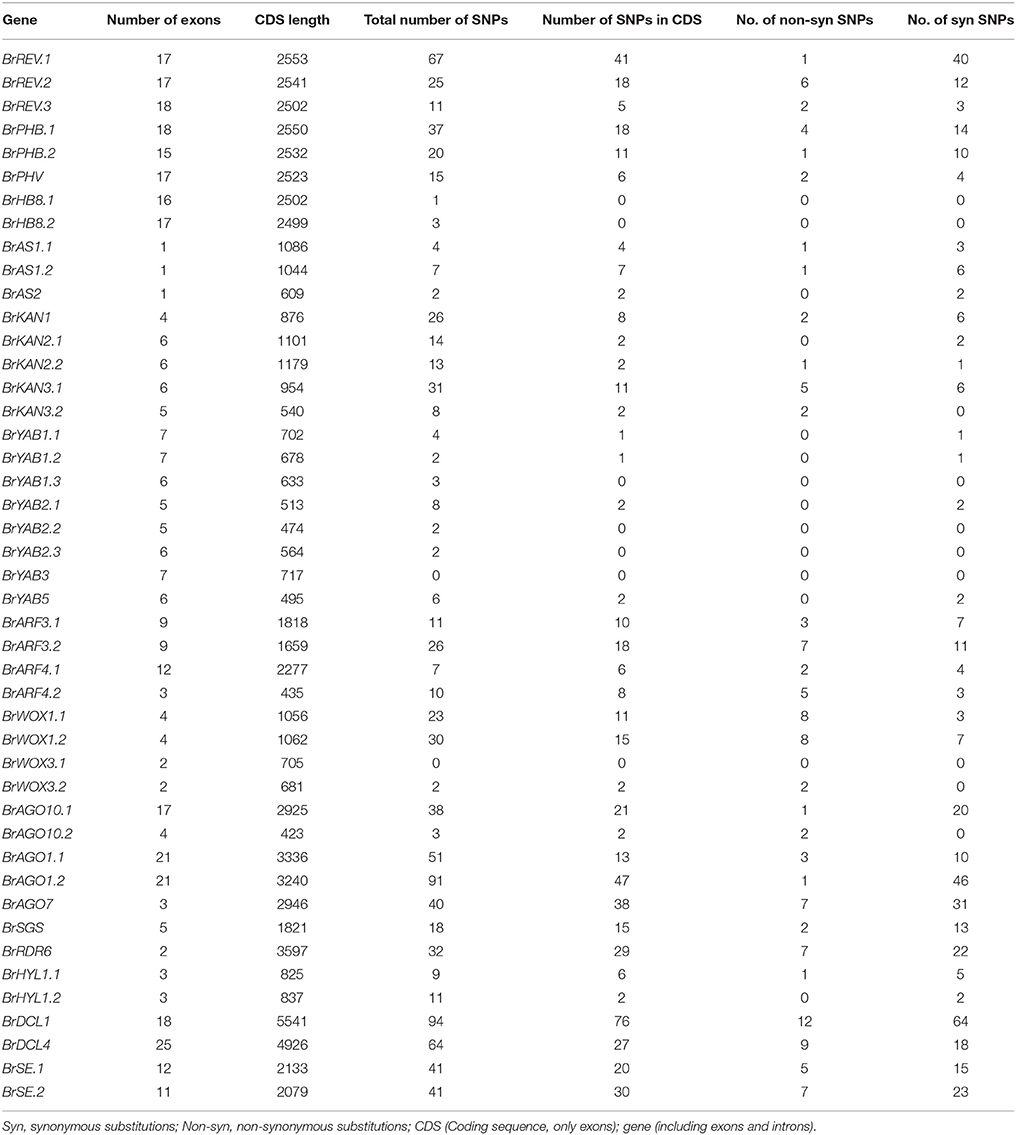
We estimated the genetic variation of 45 ad-ab patterning genes by examining our resequencing data set, which consisted of 94 double haploid (DH) accessions that represented different geographic origins and all known B. rapa morphotypes selected from a larger sample collection used in a whole-genome resequencing project (data not published). We called out all sequence variations within these 45 genes (including exons and introns). A total of 953 SNPs were detected. A total of 539 SNPs were identified within coding sequences (CDS), which included 419 synonymous and 120 non-synonymous SNPs. Details on the distribution of SNPs are presented in Table 3.
Nucleotide diversity (π) significantly varied among the 45 candidate genes. The genes encoding for TF pathway determinants exhibited a lower π than those involved in small RNA pathway determinants (Table S1). For the TF pathway genes, the YABBY gene family showed the lowest number of nucleotide substitutions (π range: 0.000233–0.001081), followed by the KAN family genes (π range: 0.000957–0.001993), whereas the WOX gene family showed higher variability (π range: 0.000924–0.00628). By comparison, the genes involved in small RNA pathways showed a higher number of polymorphisms (π range: 0.001704–0.007578). More specifically, two genes (BrWOX3.1 and BrYAB3) show no variations in gene regions, whereas 14 genes (BrAS2, BrHB8.1, BrHB8.2, BrHYL1.2, BrKAN2.1, BrWOX3.1, and all the eight YABBY family genes) did not harbor any non-synonymous mutations within coding regions.
To detect the correlation between the pattern of nucleotide diversity among leaf ad-ab pattering genes and the leaf morphotype in B. rapa, nucleotide variation and diversity of the 45 candidate genes were separately estimated by grouping the 94 accessions into two different morphotype classes: 47 were Chinese cabbage as heading B. rapa (H-Br), whereas the other 47 were non-heading B. rapa (NH-Br). Approximately 10 genes (BrARF3.1, BrARF4.1, BrKAN1, BrKAN2.1, BrKAN2.2, BrKAN3.1, BrHYL1.1, BrRDR6, BrYAB1.1, and BrWOX1.1) exhibited lower π in H-Br compared to those of NH-Br, whereas BrSE.2 and BrHYL1.2 showed lower π in NH-Br than that observed in H-Br (Figure 2A). However, the remaining 33 genes in the NH-Br and H-Br groups did not exhibit substantial differences in diversity.
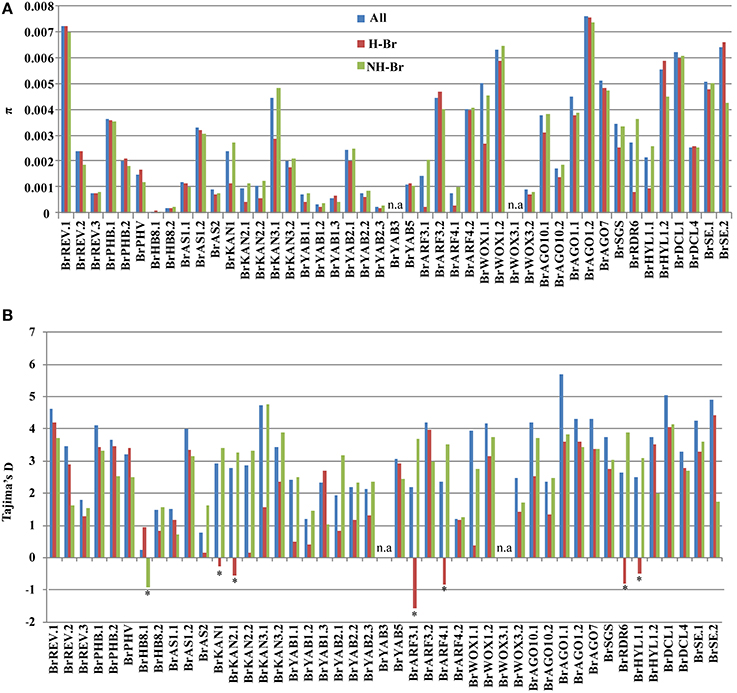
Figure 2. Nucleotide diversity (π) (A) and Tajima's D (B) indices in 45 candidate genes of three groups: whole collection (All), and H-Br and NH-Br groups. The genes for which Tajima's D is significant in the 94 accessions collection are indicated (*P < 0.05), as well as in All, H-Br, and NH-Br subgroups.
Inferred Selection Patterns of the Ad-Ab Patterning Genes across Various Genetic Groups
To determine whether the reduction in nucleotide diversity of the candidate genes were caused by artificial selection, Tajima's D test was used to determine the departure from neutrality of the 45 candidate gene regions in all three groups (All, HBr, and NH-Br). Neutrality was first estimated for the entire genome using sliding window analysis of all three groups. Figure S1 shows that these distributions substantially deviated in all three groups. Based on this finding, we set the test statistic values at the negative and positive 5 percentiles of the genome-wide distribution as thresholds. Neutrality test statistics values for the 45 candidate genes were considered significant when these exceeded the threshold value (Table S2).
Six of the 45 genes underwent negative selection in the H-Br group. All 44 candidate genes, except for BrAGO1.1, did not show any significant negative or positive Tajima's D value in the entire collection (Table S1), whereas BrAGO1.1 showed a significant positive Tajima's D value. Although 10 genes showed significantly reduced π in the H-Br group compared to that in the NH-Br group (Figure 2A), six of these (BrARF3.1, BrARF4.1, BrKAN1, BrKAN2.1, BrRDR6, and BrHYL1.1) yielded significant negative Tajima's D indices in the H-Br group. On the other hand, positive yet not significant indices were obtained in the NH-Br group (Figure 2B). These findings suggest that purifying selection had acted on the six genes in the H-Br group. In addition, one gene (BrHB8.1) showed a significant negative Tajima's D value, although no differences in π between the NH-Br and H-Br groups were observed. The remaining 38 genes showed positive Tajima's D values between the H-Br and NH-Br groups, although not statistically significant (Figure 2B), indicating that these did not significantly differ from neutral expectations. Combined π analysis and neutrality testing suggested that 6 of the 45 genes (BrARF3.1, BrARF4.1, BrKAN1, BrKAN2.1, BrRDR6, and BrHYL1.1) probably underwent purifying selection in the H-Br group.
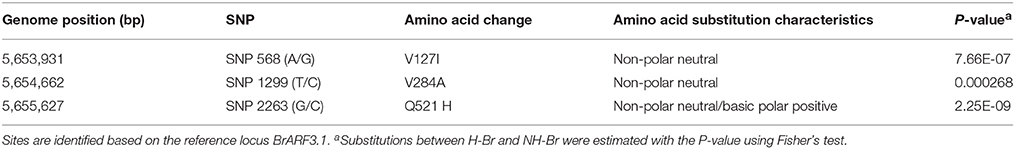
To verify the reliability of the selection signals, we further examined BrARF3.1 using an expanded collection consisting of 300 accessions (Table S4). Three non-synonymous substitutions were detected in the coding region of BrARF3.1 (Table S5). The A/G SNP at position 568 and the T/C SNP at position 1299 was predicted to result in an amino acid change of Val/Ile and Val/Ala, respectively, but affected neither the charge of the amino acid R-chain nor the polarity of the amino acid. The G/C SNP in exon 9 at gene position 2263 (CDS position: 1563 bp) was predicted to result in an amino acid change from an uncharged Gln to a positively charged His (Figure S2), which results in the alteration of protein function, as well as a significant difference between the H-Br and NH-Br groups (P = 2.25E-9) by using the Fisher's test (Table 4). We then designed a CAPS marker to genotype the G/C mutation in an extended germplasm collection consisting of 300 accessions, which included 150 heading and 150 non-heading cultivars (Table S3). Our results showed that 91.3% of the heading accessions were of the G genotype, and only 8.7% of the accessions were of the C genotype. On the other hand, in the non-heading group, 71.3% of the accessions were of the C genotype, whereas 26% were of the G genotype, and Fisher's exact test showed a P-value of 6.93E-32 (Figure 3). These results confirmed that this substitution clearly differentiated H-Br from NH-Br, thus validating our hypothesis that the BrARF3.1 gene in the H-Br group underwent purifying selection.
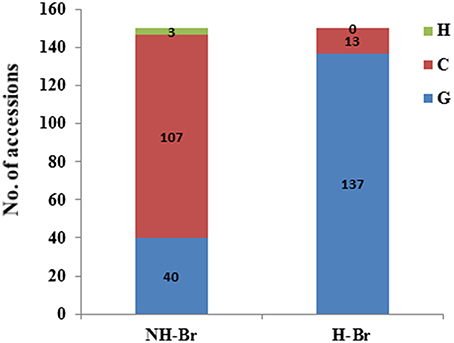
Figure 3. Nucleotide bias distribution in the BrARF3.1 gene between heading and non-heading B. rapa accessions. A non-synonymous mutation (G to C) in the BrARF3.1 gene was observed in a larger B. rapa germplasm collection consisting of 300 accessions; C, G, and H represent the genotype of C, G and hybrid, respectively.
Differentiation of Leaf Ad-Ab Patterning Genes in B. rapa
B. rapa underwent WGT after its divergence from A. thaliana. Whole-genome comparative analysis using 26 homologs in A. thaliana identified 45 ad-ab patterning genes in B. rapa, which suggested that the ad-ab patterning genes in B. rapa underwent duplication after the WGT event. The sequences of these 45 genes exhibited significant variation relative to the 94 B. rapa collections. Moreover, a higher level of sequence variation was observed among the paralogous copies. The KAN and ARF gene families are key regulators of abaxial polarity in leaves, and the absence of KAN or ARF function resulted in leaf curvature in A. thaliana, similar to that of Chinese cabbage. Taking BrKAN and BrARF genes as examples, five BrKAN genes as homologs of three AtKAN genes exhibited significant differences in nucleotide diversities within all 94 collections (Table S2, Figure 2). The highest diversity was observed in BrKAN3.1. Nucleotide diversity within the BrKAN3.1 gene was 10-fold higher than that observed in BrKAN2.1 and BrKAN2.2. Moreover, all the BrKAN genes, instead of only the BrKAN3.2 gene, showed lower π values in the H-Br group compared to that in the NH-Br group. However, only BrKAN1 and BrKAN2.1 showed significant negative Tajima D values, whereas the other BrKAN genes showed positive Tajima D values, although not statistically significant. The ARF gene family also showed different π values and selection patterns. The BrARF3.1 gene showed a lower degree of π than BrARF3.2, and comparison of these two paralogous genes indicated that the BrARF3.1 gene had reduced π and a significant negative Tajima's D value in the H-Br group, but not in the NH-Br group. On the other hand, no differences in π and Tajima's D value for the BrARF3.2 gene were observed between the H-Br and NH-Br groups. Similar findings were obtained from the paralogous pair BrARF4.1 and BrARF4.2. These results suggested that each of the BrKAN and BrARF orthologous genes has a different number of duplicated copies, which in turn results in variable π values and selection patterns. In general, duplications are assumed to be essential to the generation of novel gene functions, as well as to the alterations in patterns of expression (Lynch and Conery, 2000). However, several duplicated genes may have experienced relaxed selection, but when duplicates acquire different functions, these are potentially exposed to gradually increasing selective constraints (Lynch and Conery, 2000). In the present study, the duplicated paralogous genes might have undergone functional divergence during the leaf ad-ab patterning pathway in B. rapa.
Discussion
Leaf Ad-Ab Patterning and Leaf Morphology Diversity
Angiosperm leaves show remarkable morphological diversity, and evolutionary studies on various leaf forms such as unifacial, peltate, and compound leaves have suggested that alterations in the ad-ab patterning mechanism causes dramatic modifications in leaf morphology and thus, could be a major driving force in the generation of diverse leaf forms (see review Yamaguchi et al., 2012). B. rapa comprises a variety of vegetables with rich leaf morphological diversity. In Chinese cabbage, leaf morphology is highly variable, which include seedling leaf, rosette leaf, and head leaf. Seedling leaves and rosette leaves are flat and function in photosynthesis and respiration, whereas head leaves show incurvature and are responsible for the formation of a leafy head. In the present study, the observed diversity in leaf morphology in Chinese cabbage might have been caused by alterations in leaf ad-ab patterning. In addition, previous studies on leaf ad-ab patterning mutants present the leaf curling phenotype (Yamaguchi et al., 2012), and leaf incurvature is essential for leafy head formation (Mao et al., 2014), further indicating that leaf ad-ab patterning could be involved in head leaf incurvature and subsequent leafy head formation. However, studies on leaf ad-ab patterning in B. rapa are limited. The aim of the present study was to identify the ad-ab patterning genes in B. rapa and to investigate whether these were involved in leafy head formation in Chinese cabbage.
Six Leaf Ad-Ab Patterning Genes are under Selection in Chinese Cabbage
Comparative analysis using A. thaliana has identified 45 ad-ab patterning genes in B. rapa. Our establishment of a set of ad-ab patterning genes in B. rapa will not only provide a valuable resource for the study of the leaf ad-ab patterning system of B. rapa, but also will facilitate in elucidating its role in leafy head formation in Chinese cabbage.
We also assessed intraspecific nucleotide and modes of selection for the 45 ad-ab polarity genes in a collection of 94 accessions that included the heading and non-heading morphotype. All 45 genes exhibited significant sequence variation; however, these did not follow any common pattern and was strongly dependent on its gene family. Moreover, members of the same gene family or even paralogous copies exhibited significant variation. However, most of the genes exhibited similar π values and did not show significant selection signals between the NH-Br and H-Br groups. Only six genes (BrARF3.1, BrARF4.1, BrKAN1, BrKAN2.1, BrRDR6, and BrHYL1.1) showed significant negative Tajima's D values that were in agreement with the observed reduction in π value in the H-Br group, which was suggestive of purifying selection. However, by using these analytical methods, we could not avoid acquiring false positive selection or false negative signals. Such reduced diversity could also be explained by background selection, genetic hitchhiking, or by an extension of the effective population size following a bottleneck. Despite these limitations, the present study highlighted that purifying selection of these six genes in the H-Br group could not be ruled out, and the selection signal of the BrARF3.1 gene was subsequently confirmed in a larger group of accessions. We thus postulated that these six genes could serve as candidate genes that are responsible for leafy head formation in Chinese cabbage and its functional characterization should be investigated in future studies.
Leaf Ad-Ab Patterning Genes are Probably Linked to Leafy Head Formation in Chinese Cabbage
The present study has determined that 6 of the 45 ad-ab patterning genes in the H-Br group underwent strong negative selection. Of these six genes, BrARF3.1, BrARF4.1, BrKAN1, and BrKAN2.1 were identified as homologs of Arabidopsis abaxial polarity genes, and BrHYL1.1 is the homolog of Arabidopsis HYL, which is involved in the biogenesis of the abaxial determinant, miRNA166. On the other hand, the RDR6 gene encodes for small RNA determinants, including adaxial and abaxial determinants. These findings indicated that all six genes, except for BrRDR6, were involved in leaf abaxial polarity. Previous studies on Arabidopsis have suggested that ARF3 and ARF4 act together with KANs in the determination of leaf abaxial fate (Pekker et al., 2005). Individual loss-of-function mutants of ARF (ARF3 and ARF4) or KAN (KAN1–KAN3) show weak or no phenotypes in leaves; however, double mutants of arf3arf4 develop leaves that are curled up, resembling the phenotype of kan1 kan2 leaves (Eshed et al., 2001, 2004; Pekker et al., 2005), thereby indicating an overlap in the function of leaf abaxial polarity. As expected, the homologous genes of ARF3, ARF4, KAN1, and KAN2 in B. rapa were all included in the candidate gene list for leaf incurvature formation. Therefore, our results suggest that leaf ad-ab patterning is associated to leaf incurvature within the leafy head of Chinese cabbage. This hypothesis is supported by the evidence that BcpLH (also known as BrHYL) plays a role in the inward orientation of head leaves of Chinese cabbage.
Taken together, our present study has provided a set of candidate genes that are responsible for leafy head formation in Chinese cabbage. Functional analysis of these candidate genes and anatomy analysis of ad-ab patterning in heading leaves compared to those of flat rosette leaves may improve our understanding of the mechanism underlying leafy head formation.
Author Contributions
XW and FC designed the research. JL and BL performed the research and analyzed the data. FC and JW contributed computational tools and data. JL wrote the article. XW, JW and FC reviewed the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer Stefan De Folter and Handling Editor declared their shared affiliation, and the Handling Editor states that the process nevertheless met the standards of a fair and objective review.
Acknowledgments
The National Natural Science Foundation of China (NSFC Grant No. 31301784), the National Program Fund for Key Basic Research Projects (the 973 Program: 2013CB127000 and 2012CB113900), the National High Technology R&D Program of China (2012AA100101), NSFC grants 31301771 and 31272179, and the Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences supported this study. This research work was conducted at the Key Laboratory of Biology and Genetic Improvement of Horticultural Crops, Ministry of Agriculture, China.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fpls.2016.00094
References
Braybrook, S. A., and Kuhlemeier, C. (2010). How a plant builds leaves. Plant Cell 22, 1006–1018. doi: 10.1105/tpc.110.073924
Cheng, F., Liu, S., Wu, J., Fang, L., Sun, S., Liu, B., et al. (2011). BRAD, the genetics and genomics database for Brassica plants. BMC Plant Biol. 11:136. doi: 10.1186/1471-2229-11-136
Cheng, F., Wu, J., Fang, L., Sun, S., Liu, B., Lin, K., et al. (2012a). Biased gene fractionation and dominant gene expression among the subgenomes of Brassica rapa. PLoS ONE 7:e36442. doi: 10.1371/journal.pone.0036442
Cheng, F., Wu, J., Fang, L., and Wang, X. (2012b). Syntenic gene analysis between Brassica rapa and other Brassicaceae species. Front. Plant Sci. 3:198. doi: 10.3389/fpls.2012.00198
Chitwood, D. H., Guo, M., Nogueira, F. T., and Timmermans, M. C. (2007). Establishing leaf polarity: the role of small RNAs and positional signals in the shoot apex. Development 134, 813–823. doi: 10.1242/dev.000497
Chitwood, D. H., Nogueira, F. T., Howell, M. D., Montgomery, T. A., Carrington, J. C., and Timmermans, M. C. (2009). Pattern formation via small RNA mobility. Genes Dev. 23, 549–554. doi: 10.1101/gad.1770009
Emery, J. F., Floyd, S. K., Alvarez, J., Eshed, Y., Hawker, N. P., Izhaki, A., et al. (2003). Radial patterning of Arabidopsis shoots by class III HD-ZIP and KANADI genes. Curr. Biol. 13, 1768–1774. doi: 10.1016/j.cub.2003.09.035
Eshed, Y., Baum, S. F., Perea, J. V., and Bowman, J. L. (2001). Establishment of polarity in lateral organs of plants. Curr. Biol. 11, 1251–1260. doi: 10.1016/S0960-9822(01)00392-X
Eshed, Y., Izhaki, A., Baum, S. F., Floyd, S. K., and Bowman, J. L. (2004). Asymmetric leaf development and blade expansion in Arabidopsis are mediated by KANADI and YABBY activities. Development 131, 2997–3006. doi: 10.1242/dev.01186
Fahlgren, N., Montgomery, T. A., Howell, M. D., Allen, E., Dvorak, S. K., Alexander, A. L., et al. (2006). Regulation of AUXIN RESPONSE FACTOR3 by TAS3 ta-siRNA affects developmental timing and patterning in Arabidopsis. Curr. Biol. 16, 939–944. doi: 10.1016/j.cub.2006.03.065
Guo, N., Cheng, F., Wu, J., Liu, B., Zheng, S., Liang, J., et al. (2014). Anthocyanin biosynthetic genes in Brassica rapa. BMC Genomics 15:426. doi: 10.1186/1471-2164-15-426
Han, M. H., Goud, S., Song, L., and Fedoroff, N. (2004). The Arabidopsis double-stranded RNA-binding protein HYL1 plays a role in microRNA-mediated gene regulation. Proc. Natl. Acad. Sci. U.S.A. 101, 1093–1098. doi: 10.1073/pnas.0307969100
Huang, T., Harrar, Y., Lin, C., Reinhart, B., Newell, N. R., Talavera-Rauh, F., et al. (2014). Arabidopsis KANADI1 acts as a transcriptional repressor by interacting with a specific cis-element and regulates auxin biosynthesis, transport, and signaling in opposition to HD-ZIPIII factors. Plant Cell 26, 246–262. doi: 10.1105/tpc.113.111526
Hunter, C., Willmann, M. R., Wu, G., Yoshikawa, M., de la Luz Gutiérrez-Nava, M., and Poethig, S. R. (2006). Trans-acting siRNA-mediated repression of ETTIN and ARF4 regulates heteroblasty in Arabidopsis. Development 133, 2973–2981. doi: 10.1242/dev.02491
Hutter, S., Vilella, A. J., and Rozas, J. (2006). Genome-wide DNA polymorphism analyses using VariScan. BMC Bioinformatics 7:409. doi: 10.1186/1471-2105-7-409
Iwakawa, H., Iwasaki, M., Kojima, S., Ueno, Y., Soma, T., Tanaka, H., et al. (2007). Expression of the ASYMMETRIC LEAVES2 gene in the adaxial domain of Arabidopsis leaves represses cell proliferation in this domain and is critical for the development of properly expanded leaves. Plant J. 51, 173–184. doi: 10.1111/j.1365-313X.2007.03132.x
Kerstetter, R. A., Bollman, K., Taylor, R. A., Bomblies, K., and Poethig, R. S. (2001). KANADI regulates organ polarity in Arabidopsis. Nature 411, 706–709. doi: 10.1038/35079629
Kidner, C. A., and Martienssen, R. A. (2004). Spatially restricted microRNA directs leaf polarity through ARGONAUTE1. Nature 428, 81–84. doi: 10.1038/nature02366
Kidner, C. A., and Timmermans, M. C. (2007). Mixing and matching pathways in leaf polarity. Curr. Opin. Plant Biol. 10, 13–20. doi: 10.1016/j.pbi.2006.11.013
Kidner, C. A., and Timmermans, M. C. P. (2010). Signaling Sides: adaxial-abaxial patterning in leaves. Curr. Top. Dev. Biol. 91, 141–168. doi: 10.1016/S0070-2153(10)91005-3
Li, J., Liu, B., Cheng, F., Wang, X., Aarts, M. G., and Wu, J. (2014). Expression profiling reveals functionally redundant multiple-copy genes related to zinc, iron and cadmium responses in Brassica rapa. New Phytol. 203, 182–194. doi: 10.1111/nph.12803
Lin, W.-C., Shuai, B., and Springer, P. S. (2003). The Arabidopsis LATERAL ORGAN BOUNDARIES-domain gene ASYMMETRIC LEAVES2 functions in the repression of KNOX gene expression and in adaxial-abaxial patterning. Plant Cell 15, 2241–2252. doi: 10.1105/tpc.014969
Liu, Z., Jia, L., Wang, H., and He, Y. (2011). HYL1 regulates the balance between adaxial and abaxial identity for leaf flattening via miRNA-mediated pathways. J. Exp. Bot. 62, 4367–4381. doi: 10.1093/jxb/err167
Lynch, M., and Conery, J.S. (2000). The evolutionary fate and consequences of duplicate genes. Science 290, 1151-1155. doi: 10.1126/science.290.5494.1151
Mao, Y., Wu, F., Yu, X., Bai, J., Zhong, W., and He, Y. (2014). MicroRNA319a-targeted Brassica rapa ssp. pekinensis TCP genes modulate head shape in chinese cabbage by differential cell division arrest in leaf regions. Plant Physiol. 164, 710–720. doi: 10.1104/pp.113.228007
McConnell, J. R., Emery, J., Eshed, Y., Bao, N., Bowman, J., and Barton, M. K. (2001). Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411, 709–713. doi: 10.1038/35079635
Moon, J., and Hake, S. (2011). How a leaf gets its shape. Curr. Opin. Plant Biol. 14, 24–30. doi: 10.1016/j.pbi.2010.08.012
Nakata, M., Matsumoto, N., Tsugeki, R., Rikirsch, E., Laux, T., and Okada, K. (2012). Roles of the middle domain-specific WUSCHEL-RELATED HOMEOBOX genes in early development of leaves in Arabidopsis. Plant Cell 24, 519–535. doi: 10.1105/tpc.111.092858
Pekker, I., Alvarez, J. P., and Eshed, Y. (2005). Auxin response factors mediate Arabidopsis organ asymmetry via modulation of KANADI activity. Plant Cell 17, 2899–2910. doi: 10.1105/tpc.105.034876
Schranz, M. E., Lysak, M. A., and Mitchell-Olds, T. (2006). The ABC's of comparative genomics in the Brassicaceae: building blocks of crucifer genomes. Trends Plant Sci. 11, 535–542. doi: 10.1016/j.tplants.2006.09.002
Stahle, M. I., Kuehlich, J., Staron, L., von Arnim, A. G., and Golz, J. F. (2009). YABBYs and the transcriptional corepressors LEUNIG and LEUNIG_HOMOLOG maintain leaf polarity and meristem activity in Arabidopsis. Plant Cell 21, 3105–3118. doi: 10.1105/tpc.109.070458
Tajima, F. (1989). Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123, 585–595.
Vandenbussche, M., Horstman, A., Zethof, J., Koes, R., Rijpkema, A. S., and Gerats, T. (2009). Differential recruitment of WOX transcription factors for lateral development and organ fusion in Petunia and Arabidopsis. Plant Cell 21, 2269–2283. doi: 10.1105/tpc.109.065862
Wang, F., Li, L., Li, H., Liu, L., Zhang, Y., Gao, J., et al. (2012). Transcriptome analysis of rosette and folding leaves in Chinese cabbage using high-throughput RNA sequencing. Genomics 99, 299–307. doi: 10.1016/j.ygeno.2012.02.005
Wang, X., Wang, H., Wang, J., Sun, R., Wu, J., Liu, S., et al. (2011). The genome of the mesopolyploid crop species Brassica rapa. Nat. Genet. 43, 1035–1039. doi: 10.1038/ng.919
Wang, Y., Wu, F., Bai, J., and He, Y. (2014). BrpSPL9 (Brassica rapa ssp. pekinensis SPL9) controls the earliness of heading time in Chinese cabbage. Plant Biotechnol. J. 12, 312–321. doi: 10.1111/pbi.12138
Wu, F., Yu, L., Cao, W., Mao, Y., Liu, Z., and He, Y. (2007). The N-terminal double-stranded RNA binding domains of Arabidopsis HYPONASTIC LEAVES1 are sufficient for pre-microRNA processing. Plant Cell 19, 914–925. doi: 10.1105/tpc.106.048637
Wu, G., Lin, W.-C., Huang, T., Poethig, R. S., Springer, P. S., and Kerstetter, R. A. (2008). KANADI1 regulates adaxial-abaxial polarity in Arabidopsis by directly repressing the transcription of ASYMMETRIC LEAVES2. Proc. Natl. Acad. Sci. U.S.A. 105, 16392–16397. doi: 10.1073/pnas.0803997105
Wu, H., Yu, L., Tang, X.-R., Shen, R.-J., and He, Y.-K. (2004). Leaf downward curvature and delayed flowering caused by AtLH overexpression in Arabidopsis thaliana. Acta Bot. Sin. 46, 1106–1113.
Yamaguchi, T., Nukazuka, A., and Tsukaya, H. (2012). Leaf adaxial-abaxial polarity specification and lamina outgrowth: evolution and development. Plant Cell Physiol. 53, 1180–1194. doi: 10.1093/pcp/pcs074
Yu, X., Peng, J., Feng, X., Yang, S., Zheng, Z., Tang, X., et al. (2000). Cloning and structural and expressional characterization of BcpLH gene preferentially expressed in folding leaf of Chinese cabbage. Sci. China Ser. C Life Sci. 43, 321–329. doi: 10.1007/BF02879292
Yu, X., Wang, H., Zhong, W., Bai, J., Liu, P., and He, Y. (2013). QTL mapping of leafy heads by genome resequencing in the RIL population of Brassica rapa. PLoS ONE 8:e76059. doi: 10.1371/journal.pone.0076059
Keywords: Chinese cabbage, Brassica rapa, adaxial-abaxial polarity, genetic variation, purifying selection, leafy head
Citation: Liang J, Liu B, Wu J, Cheng F and Wang X (2016) Genetic Variation and Divergence of Genes Involved in Leaf Adaxial-Abaxial Polarity Establishment in Brassica rapa. Front. Plant Sci. 7:94. doi: 10.3389/fpls.2016.00094
Received: 08 September 2015; Accepted: 18 January 2016;
Published: 09 February 2016.
Edited by:
Stewart Gillmor, Centro de Investigación y de Estudios Avanzados del Instituto Politécnico Nacional, MexicoReviewed by:
Stefan De Folter, Centro de Investigación y de Estudios Avanzados del Instituto Politécnico Nacional, MexicoLi Yang, University of North Carolina Chapel Hill, USA
Copyright © 2016 Liang, Liu, Wu, Cheng and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiaowu Wang, d2FuZ3hpYW93dUBjYWFzLmNu
†These authors have contributed equally to this work.
 Jianli Liang†
Jianli Liang† Xiaowu Wang
Xiaowu Wang